User login
Richard Quinn is an award-winning journalist with 15 years’ experience. He has worked at the Asbury Park Press in New Jersey and The Virginian-Pilot in Norfolk, Va., and currently is managing editor for a leading commercial real estate publication. His freelance work has appeared in The Jewish State, The Hospitalist, The Rheumatologist, ACEP Now, and ENT Today. He lives in New Jersey with his wife and three cats.
A Deadly Month
The results of a new Journal of General Internal Medicine study that associates a 10% increase in medication-error-related deaths in teaching hospitals in July should not be ignored by hospitalists, says an associate residency program director.
Majid E. Cina, MD, FACP, a hospitalist and associate director at the University of Maryland School of Medicine in Baltimore, says the implication is that errors made by new medical interns are responsible for the spike, but HM leaders in academic settings shouldn’t draw too solid a conclusion. The spike could be tied to interns across a variety of departments, or the result of a series of other factors outside the scope of researchers.
“Correlations made by such studies are inherently flawed, with no certain cause-effect relationship established despite all attempts to control variables,” Dr. Cina says. “Still, these data cannot be ignored. ... Program directors should take heed.”
Researchers reported that inside medical institutions, in counties containing teaching hospitals, fatal medication errors spiked 10% in July and in no other month (JR=1.10; [1.06-1.14]) (J Gen Intern Med. 2010 Aug;25(8):774-779). Counties without teaching hospitals in the region had no spikes, and the greater the concentration of teaching hospitals in a region, the greater the July spike (R=0.80; P=0.005).
“After assessing competing explanations, we concluded that the July mortality spike results at least partly from changes associated with the arrival of new medical residents,” the study authors wrote.
Dr. Cina says his institution prioritizes the care of patients in July. Additional staff is put in place at the beginning of internships, which start a week early to allow for a smooth transition period. Senior staff also work longer hours in July, in close contact with residents on “a steep learning curve,” he says. “I recognized my work days will be much longer in July,” Dr. Cina adds.
As an academic hospitalist, Dr. Cina noted that new rules proposed by the Accreditation Council for Graduate Medical Education (ACGME) to reduce resident work hours might help mitigate medication-error-related deaths. Those regulations could go into effect in July 2011.
The results of a new Journal of General Internal Medicine study that associates a 10% increase in medication-error-related deaths in teaching hospitals in July should not be ignored by hospitalists, says an associate residency program director.
Majid E. Cina, MD, FACP, a hospitalist and associate director at the University of Maryland School of Medicine in Baltimore, says the implication is that errors made by new medical interns are responsible for the spike, but HM leaders in academic settings shouldn’t draw too solid a conclusion. The spike could be tied to interns across a variety of departments, or the result of a series of other factors outside the scope of researchers.
“Correlations made by such studies are inherently flawed, with no certain cause-effect relationship established despite all attempts to control variables,” Dr. Cina says. “Still, these data cannot be ignored. ... Program directors should take heed.”
Researchers reported that inside medical institutions, in counties containing teaching hospitals, fatal medication errors spiked 10% in July and in no other month (JR=1.10; [1.06-1.14]) (J Gen Intern Med. 2010 Aug;25(8):774-779). Counties without teaching hospitals in the region had no spikes, and the greater the concentration of teaching hospitals in a region, the greater the July spike (R=0.80; P=0.005).
“After assessing competing explanations, we concluded that the July mortality spike results at least partly from changes associated with the arrival of new medical residents,” the study authors wrote.
Dr. Cina says his institution prioritizes the care of patients in July. Additional staff is put in place at the beginning of internships, which start a week early to allow for a smooth transition period. Senior staff also work longer hours in July, in close contact with residents on “a steep learning curve,” he says. “I recognized my work days will be much longer in July,” Dr. Cina adds.
As an academic hospitalist, Dr. Cina noted that new rules proposed by the Accreditation Council for Graduate Medical Education (ACGME) to reduce resident work hours might help mitigate medication-error-related deaths. Those regulations could go into effect in July 2011.
The results of a new Journal of General Internal Medicine study that associates a 10% increase in medication-error-related deaths in teaching hospitals in July should not be ignored by hospitalists, says an associate residency program director.
Majid E. Cina, MD, FACP, a hospitalist and associate director at the University of Maryland School of Medicine in Baltimore, says the implication is that errors made by new medical interns are responsible for the spike, but HM leaders in academic settings shouldn’t draw too solid a conclusion. The spike could be tied to interns across a variety of departments, or the result of a series of other factors outside the scope of researchers.
“Correlations made by such studies are inherently flawed, with no certain cause-effect relationship established despite all attempts to control variables,” Dr. Cina says. “Still, these data cannot be ignored. ... Program directors should take heed.”
Researchers reported that inside medical institutions, in counties containing teaching hospitals, fatal medication errors spiked 10% in July and in no other month (JR=1.10; [1.06-1.14]) (J Gen Intern Med. 2010 Aug;25(8):774-779). Counties without teaching hospitals in the region had no spikes, and the greater the concentration of teaching hospitals in a region, the greater the July spike (R=0.80; P=0.005).
“After assessing competing explanations, we concluded that the July mortality spike results at least partly from changes associated with the arrival of new medical residents,” the study authors wrote.
Dr. Cina says his institution prioritizes the care of patients in July. Additional staff is put in place at the beginning of internships, which start a week early to allow for a smooth transition period. Senior staff also work longer hours in July, in close contact with residents on “a steep learning curve,” he says. “I recognized my work days will be much longer in July,” Dr. Cina adds.
As an academic hospitalist, Dr. Cina noted that new rules proposed by the Accreditation Council for Graduate Medical Education (ACGME) to reduce resident work hours might help mitigate medication-error-related deaths. Those regulations could go into effect in July 2011.
OB-GYN Society Takes Step Toward HM Model
The American College of Obstetricians and Gynecologists (ACOG) has given a preliminary imprimatur to the HM model, whose practitioners are known in maternity circles as laborists.
ACOG’s Committee on Patient Safety and Quality released an internally published opinion this month describing the structure as “one potential solution to the achieving increased professional and patient satisfaction.” The committee, however, stopped short of formally approving the laborist movement, its chair says.
“ACOG is not officially endorsing this as a standard and a must,” says Patrice Weiss, MD, chair of the Carilion Clinic’s OB-GYN department in Roanoke, Va. “This is an option for communities or hospitals in which there may be a manpower issue with obstetricians.”
The college’s qualified blessing sees advantages in the “obstetric-gynecologic hospitalist,” including the relieved pressure of not running a private practice, more predictable schedules, competitive compensation, paid benefits, and guaranteed time off. Internal-medicine hospitalist programs routinely tout those same benefits when advertising for openings. The committee opinion adds that the laborist model also delivers benefits to a hospital, including enhanced patient safety, increased levels of nursing satisfaction, and, potentially, improved outcomes.
Dr. Weiss cautions that while the laborist approach is “gaining popularity and momentum,” institutions must safeguard against potential issues, such as arguments with PCPs over delivery fees and potential complications on handoffs. While internal-medicine HM groups have long dealt with potential frictions between PCPs and hospitalists, the conflicts are still developing in the laborist model.
“For such a program to be implemented, clear, concise communication between the providers, patients, and hospitals is key,” Dr. Weiss says. “Up-front communication will prevent surprises.”
The American College of Obstetricians and Gynecologists (ACOG) has given a preliminary imprimatur to the HM model, whose practitioners are known in maternity circles as laborists.
ACOG’s Committee on Patient Safety and Quality released an internally published opinion this month describing the structure as “one potential solution to the achieving increased professional and patient satisfaction.” The committee, however, stopped short of formally approving the laborist movement, its chair says.
“ACOG is not officially endorsing this as a standard and a must,” says Patrice Weiss, MD, chair of the Carilion Clinic’s OB-GYN department in Roanoke, Va. “This is an option for communities or hospitals in which there may be a manpower issue with obstetricians.”
The college’s qualified blessing sees advantages in the “obstetric-gynecologic hospitalist,” including the relieved pressure of not running a private practice, more predictable schedules, competitive compensation, paid benefits, and guaranteed time off. Internal-medicine hospitalist programs routinely tout those same benefits when advertising for openings. The committee opinion adds that the laborist model also delivers benefits to a hospital, including enhanced patient safety, increased levels of nursing satisfaction, and, potentially, improved outcomes.
Dr. Weiss cautions that while the laborist approach is “gaining popularity and momentum,” institutions must safeguard against potential issues, such as arguments with PCPs over delivery fees and potential complications on handoffs. While internal-medicine HM groups have long dealt with potential frictions between PCPs and hospitalists, the conflicts are still developing in the laborist model.
“For such a program to be implemented, clear, concise communication between the providers, patients, and hospitals is key,” Dr. Weiss says. “Up-front communication will prevent surprises.”
The American College of Obstetricians and Gynecologists (ACOG) has given a preliminary imprimatur to the HM model, whose practitioners are known in maternity circles as laborists.
ACOG’s Committee on Patient Safety and Quality released an internally published opinion this month describing the structure as “one potential solution to the achieving increased professional and patient satisfaction.” The committee, however, stopped short of formally approving the laborist movement, its chair says.
“ACOG is not officially endorsing this as a standard and a must,” says Patrice Weiss, MD, chair of the Carilion Clinic’s OB-GYN department in Roanoke, Va. “This is an option for communities or hospitals in which there may be a manpower issue with obstetricians.”
The college’s qualified blessing sees advantages in the “obstetric-gynecologic hospitalist,” including the relieved pressure of not running a private practice, more predictable schedules, competitive compensation, paid benefits, and guaranteed time off. Internal-medicine hospitalist programs routinely tout those same benefits when advertising for openings. The committee opinion adds that the laborist model also delivers benefits to a hospital, including enhanced patient safety, increased levels of nursing satisfaction, and, potentially, improved outcomes.
Dr. Weiss cautions that while the laborist approach is “gaining popularity and momentum,” institutions must safeguard against potential issues, such as arguments with PCPs over delivery fees and potential complications on handoffs. While internal-medicine HM groups have long dealt with potential frictions between PCPs and hospitalists, the conflicts are still developing in the laborist model.
“For such a program to be implemented, clear, concise communication between the providers, patients, and hospitals is key,” Dr. Weiss says. “Up-front communication will prevent surprises.”
HM to the Rescue
In the day-to-day grind of practicing medicine, it might be easy to lose perspective, but three weeks ago, hospitalist Rohini Noronha was unexpectedly reminded of the value of her training.
Dr. Noronha, MD, MBBS, MB, program director of Apogee Physicians hospitalist group at Wilkes-Barre General Hospital in Pennsylvania, was aboard a US Airways flight from Philadelphia to Dallas on May 20 when a voice on the public-address system asked for a doctor. She responded and was taken to a man who wasn’t breathing—and instinct and muscle memory took over.
“As a hospitalist, we are always in charge of running all the cardiac codes and arrests in the hospital,” Dr. Noronha says. “That prepared me. If you’re in private practice, you don’t see all those codes.”
Dr. Noronha, a native of India who came to the U.S. about eight years ago, performed CPR and, with the aid of two jolts from an automated external cardiac defibrillator, resuscitated the man. It was a heroic tale of a hospitalist in the right time at the right place—and one that has shined a positive spotlight on HM.
“I was pretty shocked it got so much attention,” Dr. Noronha adds. “This is just what we do. If someone is sick, it’s part of our training to go and help.”
Dr. Noronha’s training began in Mumbai, where she went to medical school, continued with another year of schooling at the University of Massachusetts, and finished with residency at Good Samaritan Hospital in Baltimore. She has worked with Apogee since 2006. Although she had a brief stint in India in private practice, she chose HM for the pace, the interaction with patients, and the ability to see immediate results—all traits that were on display on May 20. Her patient not only survived, but he was talking when paramedics arrived to ferry him off the plane.
Dr. Noronha eventually took off on her way to a conference. She conducted interviews and politely recounted her story for anyone who asked. She returned to work the next week, where cardiac codes are more routine and her first thought was a guilty pleasure.
“This is so much easier,” she says.
In the day-to-day grind of practicing medicine, it might be easy to lose perspective, but three weeks ago, hospitalist Rohini Noronha was unexpectedly reminded of the value of her training.
Dr. Noronha, MD, MBBS, MB, program director of Apogee Physicians hospitalist group at Wilkes-Barre General Hospital in Pennsylvania, was aboard a US Airways flight from Philadelphia to Dallas on May 20 when a voice on the public-address system asked for a doctor. She responded and was taken to a man who wasn’t breathing—and instinct and muscle memory took over.
“As a hospitalist, we are always in charge of running all the cardiac codes and arrests in the hospital,” Dr. Noronha says. “That prepared me. If you’re in private practice, you don’t see all those codes.”
Dr. Noronha, a native of India who came to the U.S. about eight years ago, performed CPR and, with the aid of two jolts from an automated external cardiac defibrillator, resuscitated the man. It was a heroic tale of a hospitalist in the right time at the right place—and one that has shined a positive spotlight on HM.
“I was pretty shocked it got so much attention,” Dr. Noronha adds. “This is just what we do. If someone is sick, it’s part of our training to go and help.”
Dr. Noronha’s training began in Mumbai, where she went to medical school, continued with another year of schooling at the University of Massachusetts, and finished with residency at Good Samaritan Hospital in Baltimore. She has worked with Apogee since 2006. Although she had a brief stint in India in private practice, she chose HM for the pace, the interaction with patients, and the ability to see immediate results—all traits that were on display on May 20. Her patient not only survived, but he was talking when paramedics arrived to ferry him off the plane.
Dr. Noronha eventually took off on her way to a conference. She conducted interviews and politely recounted her story for anyone who asked. She returned to work the next week, where cardiac codes are more routine and her first thought was a guilty pleasure.
“This is so much easier,” she says.
In the day-to-day grind of practicing medicine, it might be easy to lose perspective, but three weeks ago, hospitalist Rohini Noronha was unexpectedly reminded of the value of her training.
Dr. Noronha, MD, MBBS, MB, program director of Apogee Physicians hospitalist group at Wilkes-Barre General Hospital in Pennsylvania, was aboard a US Airways flight from Philadelphia to Dallas on May 20 when a voice on the public-address system asked for a doctor. She responded and was taken to a man who wasn’t breathing—and instinct and muscle memory took over.
“As a hospitalist, we are always in charge of running all the cardiac codes and arrests in the hospital,” Dr. Noronha says. “That prepared me. If you’re in private practice, you don’t see all those codes.”
Dr. Noronha, a native of India who came to the U.S. about eight years ago, performed CPR and, with the aid of two jolts from an automated external cardiac defibrillator, resuscitated the man. It was a heroic tale of a hospitalist in the right time at the right place—and one that has shined a positive spotlight on HM.
“I was pretty shocked it got so much attention,” Dr. Noronha adds. “This is just what we do. If someone is sick, it’s part of our training to go and help.”
Dr. Noronha’s training began in Mumbai, where she went to medical school, continued with another year of schooling at the University of Massachusetts, and finished with residency at Good Samaritan Hospital in Baltimore. She has worked with Apogee since 2006. Although she had a brief stint in India in private practice, she chose HM for the pace, the interaction with patients, and the ability to see immediate results—all traits that were on display on May 20. Her patient not only survived, but he was talking when paramedics arrived to ferry him off the plane.
Dr. Noronha eventually took off on her way to a conference. She conducted interviews and politely recounted her story for anyone who asked. She returned to work the next week, where cardiac codes are more routine and her first thought was a guilty pleasure.
“This is so much easier,” she says.
What Is the Most Cost- Effective Evaluation for a First Syncopal Episode?
Case
A 71-year-old woman is admitted after losing consciousness and falling at home. Her history is significant for hypertension, bilateral internal carotid artery stenoses, chronic kidney disease, and diabetes. Resting vital signs are normal. Cardiac, pulmonary, and neurologic exams are unremarkable, as is an electrocardiogram (ECG). She was noted to have a small scalp laceration. Noncontrast CT of the head demonstrates a small occipital subdural bleed thought to be a result of her fall.
What is the most cost-effective evaluation for this patient admitted with suspected first syncopal episode?
Overview
Syncope is defined as sudden, self-terminating loss of consciousness. The final common pathway of all causes of syncope is global cerebral hypoperfusion—specifically, hypoperfusion of the reticular activating system. The differential diagnosis of syncopal loss of consciousness includes neurally mediated (e.g., vasovagal) syncope, orthostatic hypotension, cardiac arrhythmias, structural heart disease, and cerebrovascular disease.
Among young, otherwise healthy people, neurally mediated syncope, which has a relatively benign prognosis, is by far the most common etiology, while in older patients, primary cardiac causes are more common. Nonsyncopal mechanisms, such as seizure and hypoglycemia, should also be considered in the differential diagnosis of transient loss of consciousness (see Table 1, p. 19).1
Syncope is a common problem, accounting for 1% to 2% of ED visits in the U.S.2 The primary objective for evaluation is identification of individuals at increased risk of death due to associated conditions, especially cardiac conditions such as structural heart disease; myocardial ischemia and infarction (MI); Wolff-Parkinson-White, Brugada, or long QT syndromes; and polymorphic ventricular tachycardia.3 True syncope can be associated with other concerning causes, such as aortic stenosis, aortic dissection, and massive pulmonary embolus, as well as arrhythmias from underlying cardiac disease.4
Review of the Data
History: A detailed history and physical examination reveals the cause in 50% of syncopal episodes. Key factors include the account of third-party observers, although it is important to note that tonic-clonic movements can be associated with the global cerebral hypoperfusion of syncope as well as with seizure.4 History of dyspnea, chest pain, or palpitations argue for a primary cardiac or pulmonary cause.
Among patients who endorse a history of palpitations, the sensation of rapid and regular pulsations in the neck points very strongly toward an AV-nodal re-entrant tachycardia, because the carotid pulse and cannon A wave arrive simultaneously in the neck.5 Postictal confusion and focal neurologic exam findings suggest a neurologic rather than syncopal cause, although there are exceptions, and the relationship between seizure and impaired myocardial perfusion is complex.
One factor shown to be significantly associated with a cardiac cause for episodic syncope is family history of sudden cardiac death.4
Physical exam: A thorough general and neurologic exam is critical in evaluation of a first syncopal episode. Supine and standing blood pressure and heart rate should be measured, waiting at least one minute (and by some protocols up to three minutes) after the patient stands up to record standing vitals. Measurement of sitting vitals is not necessary. Neither postural hypotension nor tachycardia are sensitive for hypovolemia in normal volunteers, but a 30-point increase in heart rate is more than 99% specific; a 20-point drop in systolic blood pressure (SBP) is less so.5 A difference of more than 20 mmHg between systolic arm pressures in an individual with chest pain suggests aortic dissection.
Other findings useful in diagnosis of syncope include signs of aortic stenosis, pulmonary hypertension, and myxoma. Absent aortic component of S2, a late-peaking or prolonged murmur, sustained apical impulse, and delayed carotid upstroke (“pulsus tardus”) strongly support the presence of severe aortic stenosis. Absence of a palpable pulmonic component of S2 argues strongly against significant pulmonary hypertension.
There are few physical exam findings reliably seen in pulmonary embolism, but the presence of tachycardia is reliably seen about 80% of the time.4 Although atrial myxoma is associated with a characteristic “tumor plop” heard in early diastole, the finding is not common.5
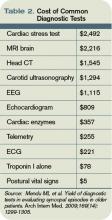
Diagnostic studies: In a recent observational study of more than 2,000 adults older than 65 hospitalized after a syncopal episode, cardiac enzymes, electroencephalography, CT scan of the head, and carotid ultrasonography determined the etiology of syncope in less than 1% of cases. Inpatient monitoring on telemetry was helpful about 5% of the time. On the other hand, postural vital signs contributed to the diagnosis more than 20% of the time, at a fraction of the cost of these other diagnostics (see Table 2, above).6
The role of transthoracic echocardiogram (echo) is more controversial. It does seem clear, however, that echo is more likely to be diagnostic in adults older than 60, in the presence of a heart murmur on physical exam or with an abnormal ECG.7 Although it demonstrated an arrhythmia that determined the etiology of syncope in only 3% of cases in this study, ECG is still routinely recommended; it is relatively inexpensive, risk-free, and can reveal abnormalities, including bundle-branch block, prior MI, and nonsustained ventricular tachycardia, which may be associated with cardiovascular comorbidities.4
Although the diagnostic tests above are of limited utility in uncovering the cause of syncopal episodes, they may be helpful when history or physical examination suggests a specific cause. For example, in individuals presenting with syncope who have lateralizing neurologic signs or symptoms or carotid bruits, carotid ultrasound is an appropriate diagnostic tool. In a retrospective analysis of 140 older adults who underwent carotid ultrasonography as part of a syncope evaluation, carotid lesions were identified in just 2% of subjects.8 These lesions were not thought to have been the primary etiology of syncope but did prompt additional evaluation or intervention.
Among older individuals or individuals with known heart disease, longer-term cardiac monitoring might be appropriate. The method of cardiac monitoring should be guided by the frequency of episodes. For events occurring daily, Holter monitoring is appropriate. For events occurring at least monthly, an event monitor is appropriate.
For less frequent events, the clinician can consider an implantable loop recorder (ILR).3 In a study of 167 individuals without a clear cause of syncope after initial evaluation, diagnosis was achieved in 90% of patients after one year of monitoring by ILR.9
Among individuals in whom the etiology remains unclear, tilt-table testing is often considered. This modality remains controversial and is unlikely to establish a diagnosis in individuals with an otherwise normal evaluation.3 Electrophysiologic testing is of similarly low yield in individuals with otherwise normal evaluation and is generally not recommended, except in individuals with known heart disease, including history of MI, congestive heart failure (CHF), and pre-excitation.10
Diagnostic algorithms: Algorithm-driven diagnostic protocols for evaluation of syncope do exist, but they are generally based on expert consensus opinion rather than large-scale studies. There are evidence-based syncope risk scores under development, but definitive validation is forthcoming. Examination of two such protocols is provided here.
The San Francisco Syncope Rule is among the most well-known algorithms, and predicts adverse outcomes at seven days. The study cohort included 684 patients presenting with syncope to an academic ED. Adverse outcomes, including death, myocardial infarction, arrhythmia, pulmonary embolus, stroke, subarachnoid hemorrhage, ED return, or hospitalization at seven days, were identified. History of CHF, hematocrit less than 30%, ECG abnormality, shortness of breath, and SBP less than 90 mmHg at presentation were associated with increased risk of an adverse outcome. If any of these findings is present, a patient is considered at high risk for adverse outcome at one week.2 The rule is simple to use; however, external validation has been controversial.
In another risk-prediction study—a large multicenter study of patients older than 60—age greater than 90 years, male sex, history of arrhythmia, SBP greater than 160 mm Hg, ECG abnormality, and elevated troponin I were used to construct a score for risk stratification.11 Specific ECG abnormalities included nonsinus rhythm, heart rate less than 40 beats per minute, evidence of acute or chronic ischemic heart disease, prolonged QRS or QT, left or right ventricular hypertrophy, left-axis deviation, and bundle-branch block. Notably, in this older cohort, CHF (specifically, systolic dysfunction with ejection fraction less than 40%) was not significantly associated with risk of adverse event at 30 days. Study authors stratified participants into low- (score ≤0), intermediate- (score 1-2), and high-risk groups (score >2), with 30-day risk of an adverse event ranging from 2.5% to 20%.
One caveat to the interpretation of these data is the fact that even in the “low risk” group, risk of adverse event was still 2.5%, a figure that many clinicians might consider intolerably high.11 This risk score has not been externally validated.
Back to the Case
Our patient was admitted to the inpatient medicine service. She was monitored overnight on telemetry without evidence of arrhythmia. Collateral history revealed new use of multiple antihypertensives prescribed by outside providers, including both atenolol and propranolol. Her subdural hematoma was managed conservatively and she remained free of neurologic deficits. On discharge, her hypertension regimen was simplified. She was referred for outpatient stress echocardiogram.
Bottom Line
Detailed history and physical exam, including postural vital signs, should form the backbone of the routine evaluation of syncope. An ECG is a critical—and inexpensive—initial diagnostic test, while inpatient telemetry, although a routine component of inpatient evaluation, is expensive and relatively low-yield. Risk prediction rules might ultimately help guide admission decisions and inpatient workup, but definitive external validation of these rules has yet to be accomplished. TH
Dr. Wander is a resident in the Department of Medicine at the University of Washington School of Medicine in Seattle. Dr. Best is an assistant professor of medicine in the Division of General Internal Medicine at the University of Washington School of Medicine.
References
- Jhanjee R, Can I, Benditt DG. Syncope. Dis Mon. 2009;55(9):532-585.
- Quinn JV, Stiell IG, McDermott DA, Sellers KL, Kohn MA, Wells GA. Derivation of the San Francisco Syncope Rule to predict patients with short-term serious outcomes. Ann Emerg Med. 2004;43(2):224-232.
- Strickberger SA, Benson DW, Biaggioni I, et al. AHA/ACCF Scientific Statement on the evaluation of syncope: from the American Heart Association Councils on Clinical Cardiology, Cardiovascular Nursing, Cardiovascular Disease in the Young, and Stroke, and the Quality of Care and Outcomes Research Interdisciplinary Working Group; and the American College of Cardiology Foundation: in collaboration with the Heart Rhythm Society: endorsed by the American Autonomic Society. Circulation. 2006;113(2):316-327.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 1: Value of history, physical examination, and electrocardiography. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;126(12):989-996.
- McGee S. Evidence-Based Physical Diagnosis. Saunders; 2001.
- Mendu ML, McAvay G, Lampert R, Stoehr J, Tinetti ME. Yield of diagnostic tests in evaluating syncopal episodes in older patients. Arch Intern Med. 2009;169(14):1299-1305.
- Panther R, Mahmood S, Gal R. Echocardiography in the diagnostic evaluation of syncope. J Am Soc Echocardiogr. 1998;11(3):294-298.
- Schnipper JL, Ackerman RH, Krier JB, Honour M. Diagnostic yield and utility of neurovascular ultrasonography in the evaluation of patients with syncope. Mayo Clin Proc. 2005;80(4):480-488.
- Assar MD, Krahn AD, Klein GJ, Yee R, Skanes AC. Optimal duration of monitoring in patients with unexplained syncope. Am J Cardiol. 2003;92(10):1231-1233.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 2: Unexplained syncope. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;127(1):76-86.
- Sun BC, Derose SF, Liang LJ, et al. Predictors of 30-day serious events in older patients with syncope. Ann Emerg Med. 2009;54(6):769-778.e1-5.
Case
A 71-year-old woman is admitted after losing consciousness and falling at home. Her history is significant for hypertension, bilateral internal carotid artery stenoses, chronic kidney disease, and diabetes. Resting vital signs are normal. Cardiac, pulmonary, and neurologic exams are unremarkable, as is an electrocardiogram (ECG). She was noted to have a small scalp laceration. Noncontrast CT of the head demonstrates a small occipital subdural bleed thought to be a result of her fall.
What is the most cost-effective evaluation for this patient admitted with suspected first syncopal episode?
Overview
Syncope is defined as sudden, self-terminating loss of consciousness. The final common pathway of all causes of syncope is global cerebral hypoperfusion—specifically, hypoperfusion of the reticular activating system. The differential diagnosis of syncopal loss of consciousness includes neurally mediated (e.g., vasovagal) syncope, orthostatic hypotension, cardiac arrhythmias, structural heart disease, and cerebrovascular disease.
Among young, otherwise healthy people, neurally mediated syncope, which has a relatively benign prognosis, is by far the most common etiology, while in older patients, primary cardiac causes are more common. Nonsyncopal mechanisms, such as seizure and hypoglycemia, should also be considered in the differential diagnosis of transient loss of consciousness (see Table 1, p. 19).1
Syncope is a common problem, accounting for 1% to 2% of ED visits in the U.S.2 The primary objective for evaluation is identification of individuals at increased risk of death due to associated conditions, especially cardiac conditions such as structural heart disease; myocardial ischemia and infarction (MI); Wolff-Parkinson-White, Brugada, or long QT syndromes; and polymorphic ventricular tachycardia.3 True syncope can be associated with other concerning causes, such as aortic stenosis, aortic dissection, and massive pulmonary embolus, as well as arrhythmias from underlying cardiac disease.4
Review of the Data
History: A detailed history and physical examination reveals the cause in 50% of syncopal episodes. Key factors include the account of third-party observers, although it is important to note that tonic-clonic movements can be associated with the global cerebral hypoperfusion of syncope as well as with seizure.4 History of dyspnea, chest pain, or palpitations argue for a primary cardiac or pulmonary cause.
Among patients who endorse a history of palpitations, the sensation of rapid and regular pulsations in the neck points very strongly toward an AV-nodal re-entrant tachycardia, because the carotid pulse and cannon A wave arrive simultaneously in the neck.5 Postictal confusion and focal neurologic exam findings suggest a neurologic rather than syncopal cause, although there are exceptions, and the relationship between seizure and impaired myocardial perfusion is complex.
One factor shown to be significantly associated with a cardiac cause for episodic syncope is family history of sudden cardiac death.4
Physical exam: A thorough general and neurologic exam is critical in evaluation of a first syncopal episode. Supine and standing blood pressure and heart rate should be measured, waiting at least one minute (and by some protocols up to three minutes) after the patient stands up to record standing vitals. Measurement of sitting vitals is not necessary. Neither postural hypotension nor tachycardia are sensitive for hypovolemia in normal volunteers, but a 30-point increase in heart rate is more than 99% specific; a 20-point drop in systolic blood pressure (SBP) is less so.5 A difference of more than 20 mmHg between systolic arm pressures in an individual with chest pain suggests aortic dissection.
Other findings useful in diagnosis of syncope include signs of aortic stenosis, pulmonary hypertension, and myxoma. Absent aortic component of S2, a late-peaking or prolonged murmur, sustained apical impulse, and delayed carotid upstroke (“pulsus tardus”) strongly support the presence of severe aortic stenosis. Absence of a palpable pulmonic component of S2 argues strongly against significant pulmonary hypertension.
There are few physical exam findings reliably seen in pulmonary embolism, but the presence of tachycardia is reliably seen about 80% of the time.4 Although atrial myxoma is associated with a characteristic “tumor plop” heard in early diastole, the finding is not common.5
Diagnostic studies: In a recent observational study of more than 2,000 adults older than 65 hospitalized after a syncopal episode, cardiac enzymes, electroencephalography, CT scan of the head, and carotid ultrasonography determined the etiology of syncope in less than 1% of cases. Inpatient monitoring on telemetry was helpful about 5% of the time. On the other hand, postural vital signs contributed to the diagnosis more than 20% of the time, at a fraction of the cost of these other diagnostics (see Table 2, above).6
The role of transthoracic echocardiogram (echo) is more controversial. It does seem clear, however, that echo is more likely to be diagnostic in adults older than 60, in the presence of a heart murmur on physical exam or with an abnormal ECG.7 Although it demonstrated an arrhythmia that determined the etiology of syncope in only 3% of cases in this study, ECG is still routinely recommended; it is relatively inexpensive, risk-free, and can reveal abnormalities, including bundle-branch block, prior MI, and nonsustained ventricular tachycardia, which may be associated with cardiovascular comorbidities.4
Although the diagnostic tests above are of limited utility in uncovering the cause of syncopal episodes, they may be helpful when history or physical examination suggests a specific cause. For example, in individuals presenting with syncope who have lateralizing neurologic signs or symptoms or carotid bruits, carotid ultrasound is an appropriate diagnostic tool. In a retrospective analysis of 140 older adults who underwent carotid ultrasonography as part of a syncope evaluation, carotid lesions were identified in just 2% of subjects.8 These lesions were not thought to have been the primary etiology of syncope but did prompt additional evaluation or intervention.
Among older individuals or individuals with known heart disease, longer-term cardiac monitoring might be appropriate. The method of cardiac monitoring should be guided by the frequency of episodes. For events occurring daily, Holter monitoring is appropriate. For events occurring at least monthly, an event monitor is appropriate.
For less frequent events, the clinician can consider an implantable loop recorder (ILR).3 In a study of 167 individuals without a clear cause of syncope after initial evaluation, diagnosis was achieved in 90% of patients after one year of monitoring by ILR.9
Among individuals in whom the etiology remains unclear, tilt-table testing is often considered. This modality remains controversial and is unlikely to establish a diagnosis in individuals with an otherwise normal evaluation.3 Electrophysiologic testing is of similarly low yield in individuals with otherwise normal evaluation and is generally not recommended, except in individuals with known heart disease, including history of MI, congestive heart failure (CHF), and pre-excitation.10
Diagnostic algorithms: Algorithm-driven diagnostic protocols for evaluation of syncope do exist, but they are generally based on expert consensus opinion rather than large-scale studies. There are evidence-based syncope risk scores under development, but definitive validation is forthcoming. Examination of two such protocols is provided here.
The San Francisco Syncope Rule is among the most well-known algorithms, and predicts adverse outcomes at seven days. The study cohort included 684 patients presenting with syncope to an academic ED. Adverse outcomes, including death, myocardial infarction, arrhythmia, pulmonary embolus, stroke, subarachnoid hemorrhage, ED return, or hospitalization at seven days, were identified. History of CHF, hematocrit less than 30%, ECG abnormality, shortness of breath, and SBP less than 90 mmHg at presentation were associated with increased risk of an adverse outcome. If any of these findings is present, a patient is considered at high risk for adverse outcome at one week.2 The rule is simple to use; however, external validation has been controversial.
In another risk-prediction study—a large multicenter study of patients older than 60—age greater than 90 years, male sex, history of arrhythmia, SBP greater than 160 mm Hg, ECG abnormality, and elevated troponin I were used to construct a score for risk stratification.11 Specific ECG abnormalities included nonsinus rhythm, heart rate less than 40 beats per minute, evidence of acute or chronic ischemic heart disease, prolonged QRS or QT, left or right ventricular hypertrophy, left-axis deviation, and bundle-branch block. Notably, in this older cohort, CHF (specifically, systolic dysfunction with ejection fraction less than 40%) was not significantly associated with risk of adverse event at 30 days. Study authors stratified participants into low- (score ≤0), intermediate- (score 1-2), and high-risk groups (score >2), with 30-day risk of an adverse event ranging from 2.5% to 20%.
One caveat to the interpretation of these data is the fact that even in the “low risk” group, risk of adverse event was still 2.5%, a figure that many clinicians might consider intolerably high.11 This risk score has not been externally validated.
Back to the Case
Our patient was admitted to the inpatient medicine service. She was monitored overnight on telemetry without evidence of arrhythmia. Collateral history revealed new use of multiple antihypertensives prescribed by outside providers, including both atenolol and propranolol. Her subdural hematoma was managed conservatively and she remained free of neurologic deficits. On discharge, her hypertension regimen was simplified. She was referred for outpatient stress echocardiogram.
Bottom Line
Detailed history and physical exam, including postural vital signs, should form the backbone of the routine evaluation of syncope. An ECG is a critical—and inexpensive—initial diagnostic test, while inpatient telemetry, although a routine component of inpatient evaluation, is expensive and relatively low-yield. Risk prediction rules might ultimately help guide admission decisions and inpatient workup, but definitive external validation of these rules has yet to be accomplished. TH
Dr. Wander is a resident in the Department of Medicine at the University of Washington School of Medicine in Seattle. Dr. Best is an assistant professor of medicine in the Division of General Internal Medicine at the University of Washington School of Medicine.
References
- Jhanjee R, Can I, Benditt DG. Syncope. Dis Mon. 2009;55(9):532-585.
- Quinn JV, Stiell IG, McDermott DA, Sellers KL, Kohn MA, Wells GA. Derivation of the San Francisco Syncope Rule to predict patients with short-term serious outcomes. Ann Emerg Med. 2004;43(2):224-232.
- Strickberger SA, Benson DW, Biaggioni I, et al. AHA/ACCF Scientific Statement on the evaluation of syncope: from the American Heart Association Councils on Clinical Cardiology, Cardiovascular Nursing, Cardiovascular Disease in the Young, and Stroke, and the Quality of Care and Outcomes Research Interdisciplinary Working Group; and the American College of Cardiology Foundation: in collaboration with the Heart Rhythm Society: endorsed by the American Autonomic Society. Circulation. 2006;113(2):316-327.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 1: Value of history, physical examination, and electrocardiography. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;126(12):989-996.
- McGee S. Evidence-Based Physical Diagnosis. Saunders; 2001.
- Mendu ML, McAvay G, Lampert R, Stoehr J, Tinetti ME. Yield of diagnostic tests in evaluating syncopal episodes in older patients. Arch Intern Med. 2009;169(14):1299-1305.
- Panther R, Mahmood S, Gal R. Echocardiography in the diagnostic evaluation of syncope. J Am Soc Echocardiogr. 1998;11(3):294-298.
- Schnipper JL, Ackerman RH, Krier JB, Honour M. Diagnostic yield and utility of neurovascular ultrasonography in the evaluation of patients with syncope. Mayo Clin Proc. 2005;80(4):480-488.
- Assar MD, Krahn AD, Klein GJ, Yee R, Skanes AC. Optimal duration of monitoring in patients with unexplained syncope. Am J Cardiol. 2003;92(10):1231-1233.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 2: Unexplained syncope. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;127(1):76-86.
- Sun BC, Derose SF, Liang LJ, et al. Predictors of 30-day serious events in older patients with syncope. Ann Emerg Med. 2009;54(6):769-778.e1-5.
Case
A 71-year-old woman is admitted after losing consciousness and falling at home. Her history is significant for hypertension, bilateral internal carotid artery stenoses, chronic kidney disease, and diabetes. Resting vital signs are normal. Cardiac, pulmonary, and neurologic exams are unremarkable, as is an electrocardiogram (ECG). She was noted to have a small scalp laceration. Noncontrast CT of the head demonstrates a small occipital subdural bleed thought to be a result of her fall.
What is the most cost-effective evaluation for this patient admitted with suspected first syncopal episode?
Overview
Syncope is defined as sudden, self-terminating loss of consciousness. The final common pathway of all causes of syncope is global cerebral hypoperfusion—specifically, hypoperfusion of the reticular activating system. The differential diagnosis of syncopal loss of consciousness includes neurally mediated (e.g., vasovagal) syncope, orthostatic hypotension, cardiac arrhythmias, structural heart disease, and cerebrovascular disease.
Among young, otherwise healthy people, neurally mediated syncope, which has a relatively benign prognosis, is by far the most common etiology, while in older patients, primary cardiac causes are more common. Nonsyncopal mechanisms, such as seizure and hypoglycemia, should also be considered in the differential diagnosis of transient loss of consciousness (see Table 1, p. 19).1
Syncope is a common problem, accounting for 1% to 2% of ED visits in the U.S.2 The primary objective for evaluation is identification of individuals at increased risk of death due to associated conditions, especially cardiac conditions such as structural heart disease; myocardial ischemia and infarction (MI); Wolff-Parkinson-White, Brugada, or long QT syndromes; and polymorphic ventricular tachycardia.3 True syncope can be associated with other concerning causes, such as aortic stenosis, aortic dissection, and massive pulmonary embolus, as well as arrhythmias from underlying cardiac disease.4
Review of the Data
History: A detailed history and physical examination reveals the cause in 50% of syncopal episodes. Key factors include the account of third-party observers, although it is important to note that tonic-clonic movements can be associated with the global cerebral hypoperfusion of syncope as well as with seizure.4 History of dyspnea, chest pain, or palpitations argue for a primary cardiac or pulmonary cause.
Among patients who endorse a history of palpitations, the sensation of rapid and regular pulsations in the neck points very strongly toward an AV-nodal re-entrant tachycardia, because the carotid pulse and cannon A wave arrive simultaneously in the neck.5 Postictal confusion and focal neurologic exam findings suggest a neurologic rather than syncopal cause, although there are exceptions, and the relationship between seizure and impaired myocardial perfusion is complex.
One factor shown to be significantly associated with a cardiac cause for episodic syncope is family history of sudden cardiac death.4
Physical exam: A thorough general and neurologic exam is critical in evaluation of a first syncopal episode. Supine and standing blood pressure and heart rate should be measured, waiting at least one minute (and by some protocols up to three minutes) after the patient stands up to record standing vitals. Measurement of sitting vitals is not necessary. Neither postural hypotension nor tachycardia are sensitive for hypovolemia in normal volunteers, but a 30-point increase in heart rate is more than 99% specific; a 20-point drop in systolic blood pressure (SBP) is less so.5 A difference of more than 20 mmHg between systolic arm pressures in an individual with chest pain suggests aortic dissection.
Other findings useful in diagnosis of syncope include signs of aortic stenosis, pulmonary hypertension, and myxoma. Absent aortic component of S2, a late-peaking or prolonged murmur, sustained apical impulse, and delayed carotid upstroke (“pulsus tardus”) strongly support the presence of severe aortic stenosis. Absence of a palpable pulmonic component of S2 argues strongly against significant pulmonary hypertension.
There are few physical exam findings reliably seen in pulmonary embolism, but the presence of tachycardia is reliably seen about 80% of the time.4 Although atrial myxoma is associated with a characteristic “tumor plop” heard in early diastole, the finding is not common.5
Diagnostic studies: In a recent observational study of more than 2,000 adults older than 65 hospitalized after a syncopal episode, cardiac enzymes, electroencephalography, CT scan of the head, and carotid ultrasonography determined the etiology of syncope in less than 1% of cases. Inpatient monitoring on telemetry was helpful about 5% of the time. On the other hand, postural vital signs contributed to the diagnosis more than 20% of the time, at a fraction of the cost of these other diagnostics (see Table 2, above).6
The role of transthoracic echocardiogram (echo) is more controversial. It does seem clear, however, that echo is more likely to be diagnostic in adults older than 60, in the presence of a heart murmur on physical exam or with an abnormal ECG.7 Although it demonstrated an arrhythmia that determined the etiology of syncope in only 3% of cases in this study, ECG is still routinely recommended; it is relatively inexpensive, risk-free, and can reveal abnormalities, including bundle-branch block, prior MI, and nonsustained ventricular tachycardia, which may be associated with cardiovascular comorbidities.4
Although the diagnostic tests above are of limited utility in uncovering the cause of syncopal episodes, they may be helpful when history or physical examination suggests a specific cause. For example, in individuals presenting with syncope who have lateralizing neurologic signs or symptoms or carotid bruits, carotid ultrasound is an appropriate diagnostic tool. In a retrospective analysis of 140 older adults who underwent carotid ultrasonography as part of a syncope evaluation, carotid lesions were identified in just 2% of subjects.8 These lesions were not thought to have been the primary etiology of syncope but did prompt additional evaluation or intervention.
Among older individuals or individuals with known heart disease, longer-term cardiac monitoring might be appropriate. The method of cardiac monitoring should be guided by the frequency of episodes. For events occurring daily, Holter monitoring is appropriate. For events occurring at least monthly, an event monitor is appropriate.
For less frequent events, the clinician can consider an implantable loop recorder (ILR).3 In a study of 167 individuals without a clear cause of syncope after initial evaluation, diagnosis was achieved in 90% of patients after one year of monitoring by ILR.9
Among individuals in whom the etiology remains unclear, tilt-table testing is often considered. This modality remains controversial and is unlikely to establish a diagnosis in individuals with an otherwise normal evaluation.3 Electrophysiologic testing is of similarly low yield in individuals with otherwise normal evaluation and is generally not recommended, except in individuals with known heart disease, including history of MI, congestive heart failure (CHF), and pre-excitation.10
Diagnostic algorithms: Algorithm-driven diagnostic protocols for evaluation of syncope do exist, but they are generally based on expert consensus opinion rather than large-scale studies. There are evidence-based syncope risk scores under development, but definitive validation is forthcoming. Examination of two such protocols is provided here.
The San Francisco Syncope Rule is among the most well-known algorithms, and predicts adverse outcomes at seven days. The study cohort included 684 patients presenting with syncope to an academic ED. Adverse outcomes, including death, myocardial infarction, arrhythmia, pulmonary embolus, stroke, subarachnoid hemorrhage, ED return, or hospitalization at seven days, were identified. History of CHF, hematocrit less than 30%, ECG abnormality, shortness of breath, and SBP less than 90 mmHg at presentation were associated with increased risk of an adverse outcome. If any of these findings is present, a patient is considered at high risk for adverse outcome at one week.2 The rule is simple to use; however, external validation has been controversial.
In another risk-prediction study—a large multicenter study of patients older than 60—age greater than 90 years, male sex, history of arrhythmia, SBP greater than 160 mm Hg, ECG abnormality, and elevated troponin I were used to construct a score for risk stratification.11 Specific ECG abnormalities included nonsinus rhythm, heart rate less than 40 beats per minute, evidence of acute or chronic ischemic heart disease, prolonged QRS or QT, left or right ventricular hypertrophy, left-axis deviation, and bundle-branch block. Notably, in this older cohort, CHF (specifically, systolic dysfunction with ejection fraction less than 40%) was not significantly associated with risk of adverse event at 30 days. Study authors stratified participants into low- (score ≤0), intermediate- (score 1-2), and high-risk groups (score >2), with 30-day risk of an adverse event ranging from 2.5% to 20%.
One caveat to the interpretation of these data is the fact that even in the “low risk” group, risk of adverse event was still 2.5%, a figure that many clinicians might consider intolerably high.11 This risk score has not been externally validated.
Back to the Case
Our patient was admitted to the inpatient medicine service. She was monitored overnight on telemetry without evidence of arrhythmia. Collateral history revealed new use of multiple antihypertensives prescribed by outside providers, including both atenolol and propranolol. Her subdural hematoma was managed conservatively and she remained free of neurologic deficits. On discharge, her hypertension regimen was simplified. She was referred for outpatient stress echocardiogram.
Bottom Line
Detailed history and physical exam, including postural vital signs, should form the backbone of the routine evaluation of syncope. An ECG is a critical—and inexpensive—initial diagnostic test, while inpatient telemetry, although a routine component of inpatient evaluation, is expensive and relatively low-yield. Risk prediction rules might ultimately help guide admission decisions and inpatient workup, but definitive external validation of these rules has yet to be accomplished. TH
Dr. Wander is a resident in the Department of Medicine at the University of Washington School of Medicine in Seattle. Dr. Best is an assistant professor of medicine in the Division of General Internal Medicine at the University of Washington School of Medicine.
References
- Jhanjee R, Can I, Benditt DG. Syncope. Dis Mon. 2009;55(9):532-585.
- Quinn JV, Stiell IG, McDermott DA, Sellers KL, Kohn MA, Wells GA. Derivation of the San Francisco Syncope Rule to predict patients with short-term serious outcomes. Ann Emerg Med. 2004;43(2):224-232.
- Strickberger SA, Benson DW, Biaggioni I, et al. AHA/ACCF Scientific Statement on the evaluation of syncope: from the American Heart Association Councils on Clinical Cardiology, Cardiovascular Nursing, Cardiovascular Disease in the Young, and Stroke, and the Quality of Care and Outcomes Research Interdisciplinary Working Group; and the American College of Cardiology Foundation: in collaboration with the Heart Rhythm Society: endorsed by the American Autonomic Society. Circulation. 2006;113(2):316-327.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 1: Value of history, physical examination, and electrocardiography. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;126(12):989-996.
- McGee S. Evidence-Based Physical Diagnosis. Saunders; 2001.
- Mendu ML, McAvay G, Lampert R, Stoehr J, Tinetti ME. Yield of diagnostic tests in evaluating syncopal episodes in older patients. Arch Intern Med. 2009;169(14):1299-1305.
- Panther R, Mahmood S, Gal R. Echocardiography in the diagnostic evaluation of syncope. J Am Soc Echocardiogr. 1998;11(3):294-298.
- Schnipper JL, Ackerman RH, Krier JB, Honour M. Diagnostic yield and utility of neurovascular ultrasonography in the evaluation of patients with syncope. Mayo Clin Proc. 2005;80(4):480-488.
- Assar MD, Krahn AD, Klein GJ, Yee R, Skanes AC. Optimal duration of monitoring in patients with unexplained syncope. Am J Cardiol. 2003;92(10):1231-1233.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 2: Unexplained syncope. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;127(1):76-86.
- Sun BC, Derose SF, Liang LJ, et al. Predictors of 30-day serious events in older patients with syncope. Ann Emerg Med. 2009;54(6):769-778.e1-5.
Belt Tightening
Let the debate formally begin.
Proposed regulations (PDF) from the Accreditation Council for Graduate Medical Education (ACGME) that limit first-year residents to 16 hours of duty will be seen either as an awakening or an abomination to educational leaders, according to the incoming president of the Association of Program Directors in Internal Medicine (APDIM).
“The draft ... will be welcomed by programs wishing to manage fatigue and will be seen as a threat by programs who have not yet accepted the need to reform graduate medical education,” says Ethan Fried, who takes over as APDIM president July 1.
The new changes come as no shock to academic hospitalists who have been waiting for the prescribed five-year update to the landmark 2003 duty-hour standards, especially after the recommendations published in the Institute of Medicine’s 2008 report “Resident Duty Hours: Enhancing Sleep, Supervision and Safety.” If approved, the new regulations will likely take effect in July 2011.
The data points of the rules will be debated thoroughly between now and then, but Dr. Fried views the recommendations as more than just tweaks to the existing infrastructure governing residency programs. He sees the suggestions as a sea change, particularly allowances for added duty time for second- and third-year residents, as well as situational exceptions that allow residents to work longer to ensure continuity of care.
“The draft turns the old concept of professionalism 180 degrees by telling residents that sleep deprivation is no longer a lifestyle choice,” adds Dr. Fried, MD, MS, FACP, assistant professor of clinical medicine at Columbia University, vice chair for education in the Department of Medicine and director of Graduate Medical Education at St. Luke's-Roosevelt in New York City. “Residents must explicitly believe that it is their personal responsibility to work rested and free of fatigue in most cases. Furthermore, the draft makes explicit the rare but real situation in which the care of an individual patient supersedes the duty hour restrictions.”
In an editorial, members of the ACGME Duty Hour Task Force also argue that their recommendations should be viewed as more than a singular recommendation on how many hours young doctors can work (10.1056/NEJMsb1005800).
“Although much of the debate has focused on establishing appropriate limits on resident hours,” the authors wrote, “the task force recognized that ensuring patient safety and providing an excellent teaching environment entail more than setting these limits.”
Let the debate formally begin.
Proposed regulations (PDF) from the Accreditation Council for Graduate Medical Education (ACGME) that limit first-year residents to 16 hours of duty will be seen either as an awakening or an abomination to educational leaders, according to the incoming president of the Association of Program Directors in Internal Medicine (APDIM).
“The draft ... will be welcomed by programs wishing to manage fatigue and will be seen as a threat by programs who have not yet accepted the need to reform graduate medical education,” says Ethan Fried, who takes over as APDIM president July 1.
The new changes come as no shock to academic hospitalists who have been waiting for the prescribed five-year update to the landmark 2003 duty-hour standards, especially after the recommendations published in the Institute of Medicine’s 2008 report “Resident Duty Hours: Enhancing Sleep, Supervision and Safety.” If approved, the new regulations will likely take effect in July 2011.
The data points of the rules will be debated thoroughly between now and then, but Dr. Fried views the recommendations as more than just tweaks to the existing infrastructure governing residency programs. He sees the suggestions as a sea change, particularly allowances for added duty time for second- and third-year residents, as well as situational exceptions that allow residents to work longer to ensure continuity of care.
“The draft turns the old concept of professionalism 180 degrees by telling residents that sleep deprivation is no longer a lifestyle choice,” adds Dr. Fried, MD, MS, FACP, assistant professor of clinical medicine at Columbia University, vice chair for education in the Department of Medicine and director of Graduate Medical Education at St. Luke's-Roosevelt in New York City. “Residents must explicitly believe that it is their personal responsibility to work rested and free of fatigue in most cases. Furthermore, the draft makes explicit the rare but real situation in which the care of an individual patient supersedes the duty hour restrictions.”
In an editorial, members of the ACGME Duty Hour Task Force also argue that their recommendations should be viewed as more than a singular recommendation on how many hours young doctors can work (10.1056/NEJMsb1005800).
“Although much of the debate has focused on establishing appropriate limits on resident hours,” the authors wrote, “the task force recognized that ensuring patient safety and providing an excellent teaching environment entail more than setting these limits.”
Let the debate formally begin.
Proposed regulations (PDF) from the Accreditation Council for Graduate Medical Education (ACGME) that limit first-year residents to 16 hours of duty will be seen either as an awakening or an abomination to educational leaders, according to the incoming president of the Association of Program Directors in Internal Medicine (APDIM).
“The draft ... will be welcomed by programs wishing to manage fatigue and will be seen as a threat by programs who have not yet accepted the need to reform graduate medical education,” says Ethan Fried, who takes over as APDIM president July 1.
The new changes come as no shock to academic hospitalists who have been waiting for the prescribed five-year update to the landmark 2003 duty-hour standards, especially after the recommendations published in the Institute of Medicine’s 2008 report “Resident Duty Hours: Enhancing Sleep, Supervision and Safety.” If approved, the new regulations will likely take effect in July 2011.
The data points of the rules will be debated thoroughly between now and then, but Dr. Fried views the recommendations as more than just tweaks to the existing infrastructure governing residency programs. He sees the suggestions as a sea change, particularly allowances for added duty time for second- and third-year residents, as well as situational exceptions that allow residents to work longer to ensure continuity of care.
“The draft turns the old concept of professionalism 180 degrees by telling residents that sleep deprivation is no longer a lifestyle choice,” adds Dr. Fried, MD, MS, FACP, assistant professor of clinical medicine at Columbia University, vice chair for education in the Department of Medicine and director of Graduate Medical Education at St. Luke's-Roosevelt in New York City. “Residents must explicitly believe that it is their personal responsibility to work rested and free of fatigue in most cases. Furthermore, the draft makes explicit the rare but real situation in which the care of an individual patient supersedes the duty hour restrictions.”
In an editorial, members of the ACGME Duty Hour Task Force also argue that their recommendations should be viewed as more than a singular recommendation on how many hours young doctors can work (10.1056/NEJMsb1005800).
“Although much of the debate has focused on establishing appropriate limits on resident hours,” the authors wrote, “the task force recognized that ensuring patient safety and providing an excellent teaching environment entail more than setting these limits.”
Save Time, Save Money
A new study that shows time and money could be saved by standardizing billing practices will likely find a supportive audience from HM groups and their business staffs, one hospitalist program executive says.
James Kodjababian, chief revenue officer and vice president of management services for Sound Physicians in Tacoma, Wash., says his firm is large enough that it has put in place systems and information technology (IT) to navigate the complex billing infrastructure that varies from carrier to carrier. But he thinks smaller HM groups likely struggle to deal with the labyrinthine codes and regulations that different insurance companies use.
“It’s like translating 43 different languages to consolidate it and manage it,” he says.
His sentiment is buttressed by “Saving Billions of Dollars—and Physician’s Time—By Streamlining Billing Practices,” which reported that standardized payment rules and claim forms “would translate into $7 million of savings annually in physician and clinical services” (doi: 10.1377/hlthaff.2009.0075). The study in Health Affairs also reported that four hours of physician time and five of support staff time could be saved each week.
Kodjababian acknowledges that to achieve such industrywide standardization, insurance companies would have to invest funding and man-hours. However, he says, the data that could be culled from an improved system would prove beneficial both to carriers and physicians.
“It’s time and money, but at the end of the day, you create a much better information set for people to benefit from,” Kodjababian says. “Right now, it’s very difficult to compare notes. If everybody is processing the same way, you can start to run statistics. You can start to see in a more macro perspective what things we should be doing.”
A new study that shows time and money could be saved by standardizing billing practices will likely find a supportive audience from HM groups and their business staffs, one hospitalist program executive says.
James Kodjababian, chief revenue officer and vice president of management services for Sound Physicians in Tacoma, Wash., says his firm is large enough that it has put in place systems and information technology (IT) to navigate the complex billing infrastructure that varies from carrier to carrier. But he thinks smaller HM groups likely struggle to deal with the labyrinthine codes and regulations that different insurance companies use.
“It’s like translating 43 different languages to consolidate it and manage it,” he says.
His sentiment is buttressed by “Saving Billions of Dollars—and Physician’s Time—By Streamlining Billing Practices,” which reported that standardized payment rules and claim forms “would translate into $7 million of savings annually in physician and clinical services” (doi: 10.1377/hlthaff.2009.0075). The study in Health Affairs also reported that four hours of physician time and five of support staff time could be saved each week.
Kodjababian acknowledges that to achieve such industrywide standardization, insurance companies would have to invest funding and man-hours. However, he says, the data that could be culled from an improved system would prove beneficial both to carriers and physicians.
“It’s time and money, but at the end of the day, you create a much better information set for people to benefit from,” Kodjababian says. “Right now, it’s very difficult to compare notes. If everybody is processing the same way, you can start to run statistics. You can start to see in a more macro perspective what things we should be doing.”
A new study that shows time and money could be saved by standardizing billing practices will likely find a supportive audience from HM groups and their business staffs, one hospitalist program executive says.
James Kodjababian, chief revenue officer and vice president of management services for Sound Physicians in Tacoma, Wash., says his firm is large enough that it has put in place systems and information technology (IT) to navigate the complex billing infrastructure that varies from carrier to carrier. But he thinks smaller HM groups likely struggle to deal with the labyrinthine codes and regulations that different insurance companies use.
“It’s like translating 43 different languages to consolidate it and manage it,” he says.
His sentiment is buttressed by “Saving Billions of Dollars—and Physician’s Time—By Streamlining Billing Practices,” which reported that standardized payment rules and claim forms “would translate into $7 million of savings annually in physician and clinical services” (doi: 10.1377/hlthaff.2009.0075). The study in Health Affairs also reported that four hours of physician time and five of support staff time could be saved each week.
Kodjababian acknowledges that to achieve such industrywide standardization, insurance companies would have to invest funding and man-hours. However, he says, the data that could be culled from an improved system would prove beneficial both to carriers and physicians.
“It’s time and money, but at the end of the day, you create a much better information set for people to benefit from,” Kodjababian says. “Right now, it’s very difficult to compare notes. If everybody is processing the same way, you can start to run statistics. You can start to see in a more macro perspective what things we should be doing.”
New Research Target
A report in this month’s Journal of Hospital Medicine shows macrolide and quinolone antibiotics are associated with similar rates of treatment failure in acute exacerbation of chronic pulmonary disease (AECOPD). The lead author says the study could be a precursor to, say, an intrepid HM researcher working on a randomized trial of the antibiotics’ effectiveness.
“It’s a perfect thing for hospitalists to study because they’re the ones treating it,” says Michael Rothberg, MD, MPH, associate professor of medicine at Tufts University School of Medicine in Boston, and lead author of "Comparative Effectiveness of Macrolides and Quinolones for Patients Hospitalized with Acute Exacerbations of Chronic Obstructive Pulmonary Disease (AECOPD)."
The retrospective cohort review reported that out of nearly 20,000 patients, 6,139 (31%) were treated initially with a macrolide and 13,469 (69%) with a quinolone. “Those who received macrolides had a lower risk of treatment failure (6.8% vs. 8.1%, p<0.01), a finding that was attenuated after multivariable adjustment (OR=0.89, 95% CI 0.78-1.01), and disappeared in a grouped-treatment analysis (OR=1.01, 95% CI 0.75-1.35),” the authors wrote. The study found no differences in adjusted length of stay or cost. However, antibiotic-associated diarrhea was more common with quinolones (1.2% vs. 0.6%, p<0.001).
Dr. Rothberg, who is affiliated with the Center for Quality of Care Research at Baystate Medical Center in Springfield, Mass., says the data, while a point in the right direction, should be viewed as a first step in doing more search to determine the best treatment for AECOPD.
“If you look at the guidelines, the recommendations are all over the map,” Dr. Rothberg says. “This is really because there are no randomized trials in COPD patients. … There are so many unanswered questions. There’s been so much focus on pneumonia, heart failure, and acute myocardial infarction. COPD kind of has a dearth of research.”
Dr. Rothberg hopes to further that research via the COPD Outcomes-Based Network for Clinical Effectiveness & Research Translation (CONCERT), a team of physicians and researchers from centers around the country who are advocating for improvements to COPD treatment. Baystate is one of CONCERT’s outposts.
A report in this month’s Journal of Hospital Medicine shows macrolide and quinolone antibiotics are associated with similar rates of treatment failure in acute exacerbation of chronic pulmonary disease (AECOPD). The lead author says the study could be a precursor to, say, an intrepid HM researcher working on a randomized trial of the antibiotics’ effectiveness.
“It’s a perfect thing for hospitalists to study because they’re the ones treating it,” says Michael Rothberg, MD, MPH, associate professor of medicine at Tufts University School of Medicine in Boston, and lead author of "Comparative Effectiveness of Macrolides and Quinolones for Patients Hospitalized with Acute Exacerbations of Chronic Obstructive Pulmonary Disease (AECOPD)."
The retrospective cohort review reported that out of nearly 20,000 patients, 6,139 (31%) were treated initially with a macrolide and 13,469 (69%) with a quinolone. “Those who received macrolides had a lower risk of treatment failure (6.8% vs. 8.1%, p<0.01), a finding that was attenuated after multivariable adjustment (OR=0.89, 95% CI 0.78-1.01), and disappeared in a grouped-treatment analysis (OR=1.01, 95% CI 0.75-1.35),” the authors wrote. The study found no differences in adjusted length of stay or cost. However, antibiotic-associated diarrhea was more common with quinolones (1.2% vs. 0.6%, p<0.001).
Dr. Rothberg, who is affiliated with the Center for Quality of Care Research at Baystate Medical Center in Springfield, Mass., says the data, while a point in the right direction, should be viewed as a first step in doing more search to determine the best treatment for AECOPD.
“If you look at the guidelines, the recommendations are all over the map,” Dr. Rothberg says. “This is really because there are no randomized trials in COPD patients. … There are so many unanswered questions. There’s been so much focus on pneumonia, heart failure, and acute myocardial infarction. COPD kind of has a dearth of research.”
Dr. Rothberg hopes to further that research via the COPD Outcomes-Based Network for Clinical Effectiveness & Research Translation (CONCERT), a team of physicians and researchers from centers around the country who are advocating for improvements to COPD treatment. Baystate is one of CONCERT’s outposts.
A report in this month’s Journal of Hospital Medicine shows macrolide and quinolone antibiotics are associated with similar rates of treatment failure in acute exacerbation of chronic pulmonary disease (AECOPD). The lead author says the study could be a precursor to, say, an intrepid HM researcher working on a randomized trial of the antibiotics’ effectiveness.
“It’s a perfect thing for hospitalists to study because they’re the ones treating it,” says Michael Rothberg, MD, MPH, associate professor of medicine at Tufts University School of Medicine in Boston, and lead author of "Comparative Effectiveness of Macrolides and Quinolones for Patients Hospitalized with Acute Exacerbations of Chronic Obstructive Pulmonary Disease (AECOPD)."
The retrospective cohort review reported that out of nearly 20,000 patients, 6,139 (31%) were treated initially with a macrolide and 13,469 (69%) with a quinolone. “Those who received macrolides had a lower risk of treatment failure (6.8% vs. 8.1%, p<0.01), a finding that was attenuated after multivariable adjustment (OR=0.89, 95% CI 0.78-1.01), and disappeared in a grouped-treatment analysis (OR=1.01, 95% CI 0.75-1.35),” the authors wrote. The study found no differences in adjusted length of stay or cost. However, antibiotic-associated diarrhea was more common with quinolones (1.2% vs. 0.6%, p<0.001).
Dr. Rothberg, who is affiliated with the Center for Quality of Care Research at Baystate Medical Center in Springfield, Mass., says the data, while a point in the right direction, should be viewed as a first step in doing more search to determine the best treatment for AECOPD.
“If you look at the guidelines, the recommendations are all over the map,” Dr. Rothberg says. “This is really because there are no randomized trials in COPD patients. … There are so many unanswered questions. There’s been so much focus on pneumonia, heart failure, and acute myocardial infarction. COPD kind of has a dearth of research.”
Dr. Rothberg hopes to further that research via the COPD Outcomes-Based Network for Clinical Effectiveness & Research Translation (CONCERT), a team of physicians and researchers from centers around the country who are advocating for improvements to COPD treatment. Baystate is one of CONCERT’s outposts.
New Resident Regulations on the Horizon
The Accreditation Council for Graduate Medical Education (ACGME) task force is close to offering revised standards for medical resident work hours—a decision that could significantly change the landscape for academic hospitalist programs.
While no date has been set for the unveiling, a May 4 letter written by ACGME CEO Thomas Nasca, MD, MACP, says “the work of the task force is nearly complete.” Many expect the rules will be offered as a draft for public comment in the coming weeks. If approved, the new regulations would probably take effect in July 2011. ACGME formed the task force more than a year ago as the prescribed five-year update to the landmark 2003 duty-hour standards.
Medical experts say the new rules will in many ways mirror the recommendations of the Institute of Medicine’s 2008 report “Resident Duty Hours: Enhancing Sleep, Supervision and Safety.” The oft-quoted report recommended residents only treat patients for up to 16 hours during their shift, down from the current recommendation of 24 hours. It also suggests residents take an uninterrupted five hours for a continuous sleep period between 10 p.m. and 8 a.m.
Many HM physicians expect the new ACGME rules will include a 60-hour workweek cap, part of a growing trend to try to balance the educational requirements of medical school with the need to expose residents to practical experience. Dr. Nasca gave no hint as to what ACGME’s recommendations will be.
In an article in this month’s The Hospitalist, academic and community hospitalists say they have been keeping on eye on how the newest rules will change their playing fields: Will a wave of academics flee teaching hospitals, as additional clinical duties become an intrusion? Will teaching hospitals face financial pressure as they struggle to replace the lower-cost labor force that residents represent? And—perhaps most importantly from a medical perspective—will graduate trainees be as prepared as their predecessors when they enter practice?
“Hospitalists will always be involved in teaching—it will never go away,” says Julia Wright, MD, FHM, a member of Team Hospitalist and clinical associate professor of medicine and director of hospital medicine at the University of Wisconsin School of Medicine and Public Health in Madison. “But it will be a very different balance, a different kind of feel.”
The Accreditation Council for Graduate Medical Education (ACGME) task force is close to offering revised standards for medical resident work hours—a decision that could significantly change the landscape for academic hospitalist programs.
While no date has been set for the unveiling, a May 4 letter written by ACGME CEO Thomas Nasca, MD, MACP, says “the work of the task force is nearly complete.” Many expect the rules will be offered as a draft for public comment in the coming weeks. If approved, the new regulations would probably take effect in July 2011. ACGME formed the task force more than a year ago as the prescribed five-year update to the landmark 2003 duty-hour standards.
Medical experts say the new rules will in many ways mirror the recommendations of the Institute of Medicine’s 2008 report “Resident Duty Hours: Enhancing Sleep, Supervision and Safety.” The oft-quoted report recommended residents only treat patients for up to 16 hours during their shift, down from the current recommendation of 24 hours. It also suggests residents take an uninterrupted five hours for a continuous sleep period between 10 p.m. and 8 a.m.
Many HM physicians expect the new ACGME rules will include a 60-hour workweek cap, part of a growing trend to try to balance the educational requirements of medical school with the need to expose residents to practical experience. Dr. Nasca gave no hint as to what ACGME’s recommendations will be.
In an article in this month’s The Hospitalist, academic and community hospitalists say they have been keeping on eye on how the newest rules will change their playing fields: Will a wave of academics flee teaching hospitals, as additional clinical duties become an intrusion? Will teaching hospitals face financial pressure as they struggle to replace the lower-cost labor force that residents represent? And—perhaps most importantly from a medical perspective—will graduate trainees be as prepared as their predecessors when they enter practice?
“Hospitalists will always be involved in teaching—it will never go away,” says Julia Wright, MD, FHM, a member of Team Hospitalist and clinical associate professor of medicine and director of hospital medicine at the University of Wisconsin School of Medicine and Public Health in Madison. “But it will be a very different balance, a different kind of feel.”
The Accreditation Council for Graduate Medical Education (ACGME) task force is close to offering revised standards for medical resident work hours—a decision that could significantly change the landscape for academic hospitalist programs.
While no date has been set for the unveiling, a May 4 letter written by ACGME CEO Thomas Nasca, MD, MACP, says “the work of the task force is nearly complete.” Many expect the rules will be offered as a draft for public comment in the coming weeks. If approved, the new regulations would probably take effect in July 2011. ACGME formed the task force more than a year ago as the prescribed five-year update to the landmark 2003 duty-hour standards.
Medical experts say the new rules will in many ways mirror the recommendations of the Institute of Medicine’s 2008 report “Resident Duty Hours: Enhancing Sleep, Supervision and Safety.” The oft-quoted report recommended residents only treat patients for up to 16 hours during their shift, down from the current recommendation of 24 hours. It also suggests residents take an uninterrupted five hours for a continuous sleep period between 10 p.m. and 8 a.m.
Many HM physicians expect the new ACGME rules will include a 60-hour workweek cap, part of a growing trend to try to balance the educational requirements of medical school with the need to expose residents to practical experience. Dr. Nasca gave no hint as to what ACGME’s recommendations will be.
In an article in this month’s The Hospitalist, academic and community hospitalists say they have been keeping on eye on how the newest rules will change their playing fields: Will a wave of academics flee teaching hospitals, as additional clinical duties become an intrusion? Will teaching hospitals face financial pressure as they struggle to replace the lower-cost labor force that residents represent? And—perhaps most importantly from a medical perspective—will graduate trainees be as prepared as their predecessors when they enter practice?
“Hospitalists will always be involved in teaching—it will never go away,” says Julia Wright, MD, FHM, a member of Team Hospitalist and clinical associate professor of medicine and director of hospital medicine at the University of Wisconsin School of Medicine and Public Health in Madison. “But it will be a very different balance, a different kind of feel.”
ONLINE EXCLUSIVE: Workforce Readiness
In the battle over how to best cope with the changing landscape of medical training, a key front could be job descriptions. That’s right: job descriptions.
“It behooves a program to define what and who they are, and follow it through in the hiring process,” says Ken Simone, DO, FHM, president of Hospitalist and Practice Solutions in Veazie, Maine, and author of the recently published Hospitalist Recruitment and Retention: Building a Hospital Medicine Program (Hoboken, N.J.: Wiley-Blackwell, 2010).
Dr. Simone, a member of Team Hospitalist, notes that the way any successful program—private, academic, or community—hires and keeps quality staff is to attract like-minded physicians.
For younger physicians, he says a major factor in job selection is the role of mentoring. And in the wake of new and potentially continuing changes to the training that residents are allowed to experience, that training role becomes even more important. “The good news is we can basically mold this person and we can support this person, so they become the provider we want them to be, as opposed to an experienced doctor who has habits they won’t change,” Dr. Simone says.
—Ken Simone, DO, FHM, president, Hospitalist and Practice Solutions, Veazie, Maine
And while much of the discussion about residency regulations focuses on potential downsides, Dr. Simone quickly points out that post-graduate physicians often are well versed in technology, evidence-based protocols, and other modern techniques that older physicians are reticent to adopt.
“Although we recognize that recruiting is challenging, that’s not been something that has prohibited [HM] from agreeing to be the solution here,” says Shaun Frost, MD, FACP, FHM, regional director for Cogent Healthcare in St. Paul, Minn., and an SHM board member. “With education, training, and mentoring, young hospitalists fresh out of their residencies can learn to practice as efficiently as so-called 20th-century residents.”
Richard Quinn is a freelance writer based in New Jersey.
In the battle over how to best cope with the changing landscape of medical training, a key front could be job descriptions. That’s right: job descriptions.
“It behooves a program to define what and who they are, and follow it through in the hiring process,” says Ken Simone, DO, FHM, president of Hospitalist and Practice Solutions in Veazie, Maine, and author of the recently published Hospitalist Recruitment and Retention: Building a Hospital Medicine Program (Hoboken, N.J.: Wiley-Blackwell, 2010).
Dr. Simone, a member of Team Hospitalist, notes that the way any successful program—private, academic, or community—hires and keeps quality staff is to attract like-minded physicians.
For younger physicians, he says a major factor in job selection is the role of mentoring. And in the wake of new and potentially continuing changes to the training that residents are allowed to experience, that training role becomes even more important. “The good news is we can basically mold this person and we can support this person, so they become the provider we want them to be, as opposed to an experienced doctor who has habits they won’t change,” Dr. Simone says.
—Ken Simone, DO, FHM, president, Hospitalist and Practice Solutions, Veazie, Maine
And while much of the discussion about residency regulations focuses on potential downsides, Dr. Simone quickly points out that post-graduate physicians often are well versed in technology, evidence-based protocols, and other modern techniques that older physicians are reticent to adopt.
“Although we recognize that recruiting is challenging, that’s not been something that has prohibited [HM] from agreeing to be the solution here,” says Shaun Frost, MD, FACP, FHM, regional director for Cogent Healthcare in St. Paul, Minn., and an SHM board member. “With education, training, and mentoring, young hospitalists fresh out of their residencies can learn to practice as efficiently as so-called 20th-century residents.”
Richard Quinn is a freelance writer based in New Jersey.
In the battle over how to best cope with the changing landscape of medical training, a key front could be job descriptions. That’s right: job descriptions.
“It behooves a program to define what and who they are, and follow it through in the hiring process,” says Ken Simone, DO, FHM, president of Hospitalist and Practice Solutions in Veazie, Maine, and author of the recently published Hospitalist Recruitment and Retention: Building a Hospital Medicine Program (Hoboken, N.J.: Wiley-Blackwell, 2010).
Dr. Simone, a member of Team Hospitalist, notes that the way any successful program—private, academic, or community—hires and keeps quality staff is to attract like-minded physicians.
For younger physicians, he says a major factor in job selection is the role of mentoring. And in the wake of new and potentially continuing changes to the training that residents are allowed to experience, that training role becomes even more important. “The good news is we can basically mold this person and we can support this person, so they become the provider we want them to be, as opposed to an experienced doctor who has habits they won’t change,” Dr. Simone says.
—Ken Simone, DO, FHM, president, Hospitalist and Practice Solutions, Veazie, Maine
And while much of the discussion about residency regulations focuses on potential downsides, Dr. Simone quickly points out that post-graduate physicians often are well versed in technology, evidence-based protocols, and other modern techniques that older physicians are reticent to adopt.
“Although we recognize that recruiting is challenging, that’s not been something that has prohibited [HM] from agreeing to be the solution here,” says Shaun Frost, MD, FACP, FHM, regional director for Cogent Healthcare in St. Paul, Minn., and an SHM board member. “With education, training, and mentoring, young hospitalists fresh out of their residencies can learn to practice as efficiently as so-called 20th-century residents.”
Richard Quinn is a freelance writer based in New Jersey.