User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Tastier chocolate may be healthier chocolate
Chocolate: Now part of a well-balanced diet
Asking if someone loves chocolate is like asking if they love breathing. It’s really not a question that needs to be asked. The thing with chocolate, however, is that most people who love chocolate actually love sugar, since your typical milk chocolate contains only about 30% cacao. The rest, of course, is sugar.
Now, dark chocolate is actually kind of good for you since it contains beneficial flavonoids and less sugar. But that healthiness comes at a cost: Dark chocolate is quite bitter, and gets more so as the cacao content rises, to the point where 100% cacao chocolate is very nearly inedible. That’s the chocolate conundrum, the healthier it is, the worse it tastes. But what if there’s another way? What if you can have tasty chocolate that’s good for you?
That’s the question a group of researchers from Penn State University dared to ask. The secret, they discovered, is to subject the cacao beans to extra-intense roasting. We’re not sure how screaming insults at a bunch of beans will help, but if science says so ... YOU USELESS LUMP OF BARELY EDIBLE FOOD! HOW DARE YOU EXIST!
Oh, not that kind of roasting. Oops.
For their study, the researchers made 27 unsweetened chocolates, prepared using various cacao bean roasting times and temperatures, and served them to volunteers. Those volunteers reported that chocolates made with cacao beans roasted more intensely (such as 20 minutes at 340° F, 80 min at 275° F, and 54 min at 304° F) were far more acceptable than were chocolates prepared with raw or lightly roasted cacao beans.
The implications of healthy yet tasty chocolate are obvious: Master the chocolate and you’ll make millions. Imagine a future where parents say to their kids: “Don’t forget to eat your chocolate.” So, we’re off to do some cooking. Don’t want Hershey to make all the money off of this revelation.
The villain hiding in dairy for some MS patients
For some of us, lactose can be a real heartbreaker when it comes to dairy consumption, but for people with multiple sclerosis (MS) there’s another villain they may also have to face that can make their symptoms worse.
Physicians at the Institute of Anatomy at University Hospital Bonn (Germany) were getting so many complaints from patients with MS about how much worse they felt about after having cheese, yogurt, and milk that they decided to get to the bottom of it. The culprit, it seems, is casein, a protein specifically found in cow’s milk.
The researchers injected mice with various proteins found in cow’s milk and found perforated myelin sheaths in those given casein. In MS, the patient’s own immune system destroys that sheath, which leads to paresthesia, vision problems, and movement disorders.
“The body’s defenses actually attack the casein, but in the process they also destroy proteins involved in the formation of myelin, “ said Rittika Chunder, a postdoctoral fellow at the University of Bonn. How? Apparently it’s all a big misunderstanding.
While looking at molecules needed for myelin production, the researchers came across MAG, which is very similar to casein, which is a problem when patients with MS are allergic to casein. After they have dairy products, the B-cell squad gets called in to clean up the evil twin, casein, but can’t differentiate it from the good twin, MAG, so it all gets a wash and the myelin sheath suffers.
Since this happens only to patients with MS who have a casein allergy, the researchers advise them to stay away from milk, yogurt, or cottage cheese while they work on a self-test to check if patients carry the antibodies.
A small price to pay, perhaps, to stop a villainous evil twin.
You would even say it glows
If you’re anything like us – and we think you are since you’re reading this – you’ve been asking yourself: Are there any common medications in my house that will make good radiation sensors?
Not that anyone needs to worry about excess radiation or anything. Far from it. We were just wondering.
It just so happens that Anna Mrozik and Paweł Bilski, both of the Institute of Nuclear Physics Polish Academy of Sciences (IFJ PAN) in Kraków, Poland, were wondering the same thing: “During an uncontrolled release of radiation, it is highly unlikely that members of the public will be equipped with personal radiation dose monitors.”
People would need to use something they had lying around the house. A smartphone would work, the investigators explained in a statement from the IFJ PAN, but the process of converting one to radiation-sensor duty, which involves dismantling it and breaking the display glass, “is laborious and time-consuming [and] the destruction of a valuable and useful device does not seem to be the optimal solution.”
Naturally, they turned to drugs. The key, in this case, is optically stimulated luminescence. They needed to find materials that would glow with greater intensity as the radiation dose increased. Turns out that ibuprofen- and paracetamol-based painkillers fit the bill quite nicely, although aspirin also works.
It’s not known exactly which substance is causing the luminescence, but rest assured, the “physicists from the IFJ PAN intend to identify it.”
This is why you don’t interrupt someone using headphones
There’s nothing like taking a nice relaxing walk with your headphones. Whether you’re listening to a podcast or a song or talking on the phone, it’s an escape from reality that makes you feel like you’re completely in tune with what you’re listening to.
According to a new study, headphones, as opposed to speakers, make people feel more connected to what they are listening to. Data collected from more than 4,000 people showed that listening with headphones makes more of an impact than listening to speakers.
“Headphones produce a phenomenon called in-head localization, which makes the speaker sound as if they’re inside your head,” study coauthor On Amir of the University of California, San Diego, said in a statement. Because of this, people feel like the speakers are close to them and there’s more of a sense of empathy for the speakers and the listener is more likely to be swayed toward the ideas of the speaker.
These findings could lead to more efficient training programs, online work, and advertising, the investigators suggested.
We now finally understand why people get so mad when they have to take out their headphones to answer or talk to us. We ruined a satisfying moment going on in their brains.
Chocolate: Now part of a well-balanced diet
Asking if someone loves chocolate is like asking if they love breathing. It’s really not a question that needs to be asked. The thing with chocolate, however, is that most people who love chocolate actually love sugar, since your typical milk chocolate contains only about 30% cacao. The rest, of course, is sugar.
Now, dark chocolate is actually kind of good for you since it contains beneficial flavonoids and less sugar. But that healthiness comes at a cost: Dark chocolate is quite bitter, and gets more so as the cacao content rises, to the point where 100% cacao chocolate is very nearly inedible. That’s the chocolate conundrum, the healthier it is, the worse it tastes. But what if there’s another way? What if you can have tasty chocolate that’s good for you?
That’s the question a group of researchers from Penn State University dared to ask. The secret, they discovered, is to subject the cacao beans to extra-intense roasting. We’re not sure how screaming insults at a bunch of beans will help, but if science says so ... YOU USELESS LUMP OF BARELY EDIBLE FOOD! HOW DARE YOU EXIST!
Oh, not that kind of roasting. Oops.
For their study, the researchers made 27 unsweetened chocolates, prepared using various cacao bean roasting times and temperatures, and served them to volunteers. Those volunteers reported that chocolates made with cacao beans roasted more intensely (such as 20 minutes at 340° F, 80 min at 275° F, and 54 min at 304° F) were far more acceptable than were chocolates prepared with raw or lightly roasted cacao beans.
The implications of healthy yet tasty chocolate are obvious: Master the chocolate and you’ll make millions. Imagine a future where parents say to their kids: “Don’t forget to eat your chocolate.” So, we’re off to do some cooking. Don’t want Hershey to make all the money off of this revelation.
The villain hiding in dairy for some MS patients
For some of us, lactose can be a real heartbreaker when it comes to dairy consumption, but for people with multiple sclerosis (MS) there’s another villain they may also have to face that can make their symptoms worse.
Physicians at the Institute of Anatomy at University Hospital Bonn (Germany) were getting so many complaints from patients with MS about how much worse they felt about after having cheese, yogurt, and milk that they decided to get to the bottom of it. The culprit, it seems, is casein, a protein specifically found in cow’s milk.
The researchers injected mice with various proteins found in cow’s milk and found perforated myelin sheaths in those given casein. In MS, the patient’s own immune system destroys that sheath, which leads to paresthesia, vision problems, and movement disorders.
“The body’s defenses actually attack the casein, but in the process they also destroy proteins involved in the formation of myelin, “ said Rittika Chunder, a postdoctoral fellow at the University of Bonn. How? Apparently it’s all a big misunderstanding.
While looking at molecules needed for myelin production, the researchers came across MAG, which is very similar to casein, which is a problem when patients with MS are allergic to casein. After they have dairy products, the B-cell squad gets called in to clean up the evil twin, casein, but can’t differentiate it from the good twin, MAG, so it all gets a wash and the myelin sheath suffers.
Since this happens only to patients with MS who have a casein allergy, the researchers advise them to stay away from milk, yogurt, or cottage cheese while they work on a self-test to check if patients carry the antibodies.
A small price to pay, perhaps, to stop a villainous evil twin.
You would even say it glows
If you’re anything like us – and we think you are since you’re reading this – you’ve been asking yourself: Are there any common medications in my house that will make good radiation sensors?
Not that anyone needs to worry about excess radiation or anything. Far from it. We were just wondering.
It just so happens that Anna Mrozik and Paweł Bilski, both of the Institute of Nuclear Physics Polish Academy of Sciences (IFJ PAN) in Kraków, Poland, were wondering the same thing: “During an uncontrolled release of radiation, it is highly unlikely that members of the public will be equipped with personal radiation dose monitors.”
People would need to use something they had lying around the house. A smartphone would work, the investigators explained in a statement from the IFJ PAN, but the process of converting one to radiation-sensor duty, which involves dismantling it and breaking the display glass, “is laborious and time-consuming [and] the destruction of a valuable and useful device does not seem to be the optimal solution.”
Naturally, they turned to drugs. The key, in this case, is optically stimulated luminescence. They needed to find materials that would glow with greater intensity as the radiation dose increased. Turns out that ibuprofen- and paracetamol-based painkillers fit the bill quite nicely, although aspirin also works.
It’s not known exactly which substance is causing the luminescence, but rest assured, the “physicists from the IFJ PAN intend to identify it.”
This is why you don’t interrupt someone using headphones
There’s nothing like taking a nice relaxing walk with your headphones. Whether you’re listening to a podcast or a song or talking on the phone, it’s an escape from reality that makes you feel like you’re completely in tune with what you’re listening to.
According to a new study, headphones, as opposed to speakers, make people feel more connected to what they are listening to. Data collected from more than 4,000 people showed that listening with headphones makes more of an impact than listening to speakers.
“Headphones produce a phenomenon called in-head localization, which makes the speaker sound as if they’re inside your head,” study coauthor On Amir of the University of California, San Diego, said in a statement. Because of this, people feel like the speakers are close to them and there’s more of a sense of empathy for the speakers and the listener is more likely to be swayed toward the ideas of the speaker.
These findings could lead to more efficient training programs, online work, and advertising, the investigators suggested.
We now finally understand why people get so mad when they have to take out their headphones to answer or talk to us. We ruined a satisfying moment going on in their brains.
Chocolate: Now part of a well-balanced diet
Asking if someone loves chocolate is like asking if they love breathing. It’s really not a question that needs to be asked. The thing with chocolate, however, is that most people who love chocolate actually love sugar, since your typical milk chocolate contains only about 30% cacao. The rest, of course, is sugar.
Now, dark chocolate is actually kind of good for you since it contains beneficial flavonoids and less sugar. But that healthiness comes at a cost: Dark chocolate is quite bitter, and gets more so as the cacao content rises, to the point where 100% cacao chocolate is very nearly inedible. That’s the chocolate conundrum, the healthier it is, the worse it tastes. But what if there’s another way? What if you can have tasty chocolate that’s good for you?
That’s the question a group of researchers from Penn State University dared to ask. The secret, they discovered, is to subject the cacao beans to extra-intense roasting. We’re not sure how screaming insults at a bunch of beans will help, but if science says so ... YOU USELESS LUMP OF BARELY EDIBLE FOOD! HOW DARE YOU EXIST!
Oh, not that kind of roasting. Oops.
For their study, the researchers made 27 unsweetened chocolates, prepared using various cacao bean roasting times and temperatures, and served them to volunteers. Those volunteers reported that chocolates made with cacao beans roasted more intensely (such as 20 minutes at 340° F, 80 min at 275° F, and 54 min at 304° F) were far more acceptable than were chocolates prepared with raw or lightly roasted cacao beans.
The implications of healthy yet tasty chocolate are obvious: Master the chocolate and you’ll make millions. Imagine a future where parents say to their kids: “Don’t forget to eat your chocolate.” So, we’re off to do some cooking. Don’t want Hershey to make all the money off of this revelation.
The villain hiding in dairy for some MS patients
For some of us, lactose can be a real heartbreaker when it comes to dairy consumption, but for people with multiple sclerosis (MS) there’s another villain they may also have to face that can make their symptoms worse.
Physicians at the Institute of Anatomy at University Hospital Bonn (Germany) were getting so many complaints from patients with MS about how much worse they felt about after having cheese, yogurt, and milk that they decided to get to the bottom of it. The culprit, it seems, is casein, a protein specifically found in cow’s milk.
The researchers injected mice with various proteins found in cow’s milk and found perforated myelin sheaths in those given casein. In MS, the patient’s own immune system destroys that sheath, which leads to paresthesia, vision problems, and movement disorders.
“The body’s defenses actually attack the casein, but in the process they also destroy proteins involved in the formation of myelin, “ said Rittika Chunder, a postdoctoral fellow at the University of Bonn. How? Apparently it’s all a big misunderstanding.
While looking at molecules needed for myelin production, the researchers came across MAG, which is very similar to casein, which is a problem when patients with MS are allergic to casein. After they have dairy products, the B-cell squad gets called in to clean up the evil twin, casein, but can’t differentiate it from the good twin, MAG, so it all gets a wash and the myelin sheath suffers.
Since this happens only to patients with MS who have a casein allergy, the researchers advise them to stay away from milk, yogurt, or cottage cheese while they work on a self-test to check if patients carry the antibodies.
A small price to pay, perhaps, to stop a villainous evil twin.
You would even say it glows
If you’re anything like us – and we think you are since you’re reading this – you’ve been asking yourself: Are there any common medications in my house that will make good radiation sensors?
Not that anyone needs to worry about excess radiation or anything. Far from it. We were just wondering.
It just so happens that Anna Mrozik and Paweł Bilski, both of the Institute of Nuclear Physics Polish Academy of Sciences (IFJ PAN) in Kraków, Poland, were wondering the same thing: “During an uncontrolled release of radiation, it is highly unlikely that members of the public will be equipped with personal radiation dose monitors.”
People would need to use something they had lying around the house. A smartphone would work, the investigators explained in a statement from the IFJ PAN, but the process of converting one to radiation-sensor duty, which involves dismantling it and breaking the display glass, “is laborious and time-consuming [and] the destruction of a valuable and useful device does not seem to be the optimal solution.”
Naturally, they turned to drugs. The key, in this case, is optically stimulated luminescence. They needed to find materials that would glow with greater intensity as the radiation dose increased. Turns out that ibuprofen- and paracetamol-based painkillers fit the bill quite nicely, although aspirin also works.
It’s not known exactly which substance is causing the luminescence, but rest assured, the “physicists from the IFJ PAN intend to identify it.”
This is why you don’t interrupt someone using headphones
There’s nothing like taking a nice relaxing walk with your headphones. Whether you’re listening to a podcast or a song or talking on the phone, it’s an escape from reality that makes you feel like you’re completely in tune with what you’re listening to.
According to a new study, headphones, as opposed to speakers, make people feel more connected to what they are listening to. Data collected from more than 4,000 people showed that listening with headphones makes more of an impact than listening to speakers.
“Headphones produce a phenomenon called in-head localization, which makes the speaker sound as if they’re inside your head,” study coauthor On Amir of the University of California, San Diego, said in a statement. Because of this, people feel like the speakers are close to them and there’s more of a sense of empathy for the speakers and the listener is more likely to be swayed toward the ideas of the speaker.
These findings could lead to more efficient training programs, online work, and advertising, the investigators suggested.
We now finally understand why people get so mad when they have to take out their headphones to answer or talk to us. We ruined a satisfying moment going on in their brains.
Azithromycin doesn’t prevent recurrent wheezing after acute infant RSV
Azithromycin administered for severe early-life respiratory syncytial virus (RSV) bronchiolitis did not prevent recurrent wheezing in affected children over the next 2-4 years, a randomized, single-center study found.
Antibiotics are frequently given to patients with RSV bronchiolitis, although this practice is not supported by American Academy of Pediatrics clinical guidelines. Many doctors will prescribe them anyway if they see redness in the ears or other signs of infection, lead author Avraham Beigelman, MD, a pediatric allergist and immunologist at Washington University in St. Louis, said in an interview.
The double-blind, placebo-controlled trial, presented at the 2022 meeting of the American Academy of Allergy, Asthma & Immunology in Phoenix, was simultaneously published online Feb. 27, 2022, in the New England Journal of Medicine–Evidence.
Since azithromycin has shown anti-inflammatory benefit in chronic lung diseases and is a mainstay of care in cystic fibrosis and had shown previous effects in RSV patients, this trial examined its potential for preventing future recurrent wheezing in infants hospitalized with RSV who are at risk for developing asthma later. About half of children admitted to the hospital for RSV will develop asthma by age 7, Dr. Beigelman said.
“We were very surprised that azithromycin didn’t help in this trial given our previous findings,” Dr. Beigelman said.
And while those given azithromycin versus those given a placebo showed no significant decrease in recurrent wheezing, there was a slight suggestion that treatment with antibiotics of any kind may increase the risk of later wheezing in infants hospitalized with the virus.
“The study was not designed to tease at the effects of different antibiotics or combinations of antibiotics, so we have to be very cautious about this trend,” Dr. Beigelman said. “There may be short-term effects and long-term effects. Certain antibiotics may affect the infant microbiome in other parts of the body, such as the gut, [in] a way that may predispose to asthma. But all these associations suggest that early-life antibiotics for viral infections are not good for you.”
He pointed to the longstanding question among clinicians whether it is the antibiotic that’s increasing the risk of the harm or the condition for which the antibiotic is prescribed. These exploratory data, however, suggest that antibiotics for RSV may be causing harm.
In pursuit of that hypothesis, his group has collected airway microbiome samples from these infants and plan to investigate whether bacteria colonizing the airway may interact with the antibiotics to increase wheezing. The researchers will analyze stool samples from the babies to see whether the gut microbiome may also play a role in wheezing and the subsequent risk of developing childhood asthma.
Study details
The trial prospectively enrolled 200 otherwise healthy babies aged 1-18 months who were hospitalized at St. Louis Children’s Hospital for acute RSV bronchiolitis. Although RSV is a very common pediatric virus, only bout 3% of babies will require hospitalization in order to receive oxygen, Dr. Beigelman said.
Babies were randomly assigned to receive placebo or oral azithromycin at 10 mg/kg daily for 7 days, followed by 5 mg/kg daily for 7 days. Randomization was stratified by recent open-label antibiotic use. The primary outcome was recurrent wheeze, defined as a third episode of post-RSV wheeze over the following 2-4 years.
The biologic activity of azithromycin was clear since nasal-wash interleukin at day 14 after randomization was lower in azithromycin-treated infants. But despite evidence of activity, the risk of post-RSV recurrent wheeze was similar in both arms: 47% in the azithromycin group versus 36% in the placebo group, for an adjusted hazard ratio of 1.45 (95% confidence interval, 0.92-2.29; P = .11).
Nor did azithromycin lower the risk of recurrent wheeze in babies already receiving other antibiotics at the time of enrollment (HR, 0.94; 95% CI, 0.43-2.07). As for antibiotic-naive participants receiving azithromycin, there was a slight signal of potential increased risk of developing recurrent wheezing (HR, 1.79; 95% CI, 1.03-3.1).
The bottom line? The findings support current clinical guidelines recommending against the use of antibiotics for RSV. “At the very least, azithromycin and antibiotics in general have no benefit in preventing recurrent wheeze, and there is a possibility they may be harmful,” Dr. Beigelman said.
This trial is funded by the National Heart, Lung, and Blood Institute. Dr. Beigelman reported relationships with AstraZeneca, Novartis, and Sanofi. Two study coauthors disclosed various ties to industry.
Azithromycin administered for severe early-life respiratory syncytial virus (RSV) bronchiolitis did not prevent recurrent wheezing in affected children over the next 2-4 years, a randomized, single-center study found.
Antibiotics are frequently given to patients with RSV bronchiolitis, although this practice is not supported by American Academy of Pediatrics clinical guidelines. Many doctors will prescribe them anyway if they see redness in the ears or other signs of infection, lead author Avraham Beigelman, MD, a pediatric allergist and immunologist at Washington University in St. Louis, said in an interview.
The double-blind, placebo-controlled trial, presented at the 2022 meeting of the American Academy of Allergy, Asthma & Immunology in Phoenix, was simultaneously published online Feb. 27, 2022, in the New England Journal of Medicine–Evidence.
Since azithromycin has shown anti-inflammatory benefit in chronic lung diseases and is a mainstay of care in cystic fibrosis and had shown previous effects in RSV patients, this trial examined its potential for preventing future recurrent wheezing in infants hospitalized with RSV who are at risk for developing asthma later. About half of children admitted to the hospital for RSV will develop asthma by age 7, Dr. Beigelman said.
“We were very surprised that azithromycin didn’t help in this trial given our previous findings,” Dr. Beigelman said.
And while those given azithromycin versus those given a placebo showed no significant decrease in recurrent wheezing, there was a slight suggestion that treatment with antibiotics of any kind may increase the risk of later wheezing in infants hospitalized with the virus.
“The study was not designed to tease at the effects of different antibiotics or combinations of antibiotics, so we have to be very cautious about this trend,” Dr. Beigelman said. “There may be short-term effects and long-term effects. Certain antibiotics may affect the infant microbiome in other parts of the body, such as the gut, [in] a way that may predispose to asthma. But all these associations suggest that early-life antibiotics for viral infections are not good for you.”
He pointed to the longstanding question among clinicians whether it is the antibiotic that’s increasing the risk of the harm or the condition for which the antibiotic is prescribed. These exploratory data, however, suggest that antibiotics for RSV may be causing harm.
In pursuit of that hypothesis, his group has collected airway microbiome samples from these infants and plan to investigate whether bacteria colonizing the airway may interact with the antibiotics to increase wheezing. The researchers will analyze stool samples from the babies to see whether the gut microbiome may also play a role in wheezing and the subsequent risk of developing childhood asthma.
Study details
The trial prospectively enrolled 200 otherwise healthy babies aged 1-18 months who were hospitalized at St. Louis Children’s Hospital for acute RSV bronchiolitis. Although RSV is a very common pediatric virus, only bout 3% of babies will require hospitalization in order to receive oxygen, Dr. Beigelman said.
Babies were randomly assigned to receive placebo or oral azithromycin at 10 mg/kg daily for 7 days, followed by 5 mg/kg daily for 7 days. Randomization was stratified by recent open-label antibiotic use. The primary outcome was recurrent wheeze, defined as a third episode of post-RSV wheeze over the following 2-4 years.
The biologic activity of azithromycin was clear since nasal-wash interleukin at day 14 after randomization was lower in azithromycin-treated infants. But despite evidence of activity, the risk of post-RSV recurrent wheeze was similar in both arms: 47% in the azithromycin group versus 36% in the placebo group, for an adjusted hazard ratio of 1.45 (95% confidence interval, 0.92-2.29; P = .11).
Nor did azithromycin lower the risk of recurrent wheeze in babies already receiving other antibiotics at the time of enrollment (HR, 0.94; 95% CI, 0.43-2.07). As for antibiotic-naive participants receiving azithromycin, there was a slight signal of potential increased risk of developing recurrent wheezing (HR, 1.79; 95% CI, 1.03-3.1).
The bottom line? The findings support current clinical guidelines recommending against the use of antibiotics for RSV. “At the very least, azithromycin and antibiotics in general have no benefit in preventing recurrent wheeze, and there is a possibility they may be harmful,” Dr. Beigelman said.
This trial is funded by the National Heart, Lung, and Blood Institute. Dr. Beigelman reported relationships with AstraZeneca, Novartis, and Sanofi. Two study coauthors disclosed various ties to industry.
Azithromycin administered for severe early-life respiratory syncytial virus (RSV) bronchiolitis did not prevent recurrent wheezing in affected children over the next 2-4 years, a randomized, single-center study found.
Antibiotics are frequently given to patients with RSV bronchiolitis, although this practice is not supported by American Academy of Pediatrics clinical guidelines. Many doctors will prescribe them anyway if they see redness in the ears or other signs of infection, lead author Avraham Beigelman, MD, a pediatric allergist and immunologist at Washington University in St. Louis, said in an interview.
The double-blind, placebo-controlled trial, presented at the 2022 meeting of the American Academy of Allergy, Asthma & Immunology in Phoenix, was simultaneously published online Feb. 27, 2022, in the New England Journal of Medicine–Evidence.
Since azithromycin has shown anti-inflammatory benefit in chronic lung diseases and is a mainstay of care in cystic fibrosis and had shown previous effects in RSV patients, this trial examined its potential for preventing future recurrent wheezing in infants hospitalized with RSV who are at risk for developing asthma later. About half of children admitted to the hospital for RSV will develop asthma by age 7, Dr. Beigelman said.
“We were very surprised that azithromycin didn’t help in this trial given our previous findings,” Dr. Beigelman said.
And while those given azithromycin versus those given a placebo showed no significant decrease in recurrent wheezing, there was a slight suggestion that treatment with antibiotics of any kind may increase the risk of later wheezing in infants hospitalized with the virus.
“The study was not designed to tease at the effects of different antibiotics or combinations of antibiotics, so we have to be very cautious about this trend,” Dr. Beigelman said. “There may be short-term effects and long-term effects. Certain antibiotics may affect the infant microbiome in other parts of the body, such as the gut, [in] a way that may predispose to asthma. But all these associations suggest that early-life antibiotics for viral infections are not good for you.”
He pointed to the longstanding question among clinicians whether it is the antibiotic that’s increasing the risk of the harm or the condition for which the antibiotic is prescribed. These exploratory data, however, suggest that antibiotics for RSV may be causing harm.
In pursuit of that hypothesis, his group has collected airway microbiome samples from these infants and plan to investigate whether bacteria colonizing the airway may interact with the antibiotics to increase wheezing. The researchers will analyze stool samples from the babies to see whether the gut microbiome may also play a role in wheezing and the subsequent risk of developing childhood asthma.
Study details
The trial prospectively enrolled 200 otherwise healthy babies aged 1-18 months who were hospitalized at St. Louis Children’s Hospital for acute RSV bronchiolitis. Although RSV is a very common pediatric virus, only bout 3% of babies will require hospitalization in order to receive oxygen, Dr. Beigelman said.
Babies were randomly assigned to receive placebo or oral azithromycin at 10 mg/kg daily for 7 days, followed by 5 mg/kg daily for 7 days. Randomization was stratified by recent open-label antibiotic use. The primary outcome was recurrent wheeze, defined as a third episode of post-RSV wheeze over the following 2-4 years.
The biologic activity of azithromycin was clear since nasal-wash interleukin at day 14 after randomization was lower in azithromycin-treated infants. But despite evidence of activity, the risk of post-RSV recurrent wheeze was similar in both arms: 47% in the azithromycin group versus 36% in the placebo group, for an adjusted hazard ratio of 1.45 (95% confidence interval, 0.92-2.29; P = .11).
Nor did azithromycin lower the risk of recurrent wheeze in babies already receiving other antibiotics at the time of enrollment (HR, 0.94; 95% CI, 0.43-2.07). As for antibiotic-naive participants receiving azithromycin, there was a slight signal of potential increased risk of developing recurrent wheezing (HR, 1.79; 95% CI, 1.03-3.1).
The bottom line? The findings support current clinical guidelines recommending against the use of antibiotics for RSV. “At the very least, azithromycin and antibiotics in general have no benefit in preventing recurrent wheeze, and there is a possibility they may be harmful,” Dr. Beigelman said.
This trial is funded by the National Heart, Lung, and Blood Institute. Dr. Beigelman reported relationships with AstraZeneca, Novartis, and Sanofi. Two study coauthors disclosed various ties to industry.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE–EVIDENCE
Robust immune response after COVID-19 boosters in those with IBD
Of the study participants, 93% had detectable antibodies after their initial vaccination series, which increased to 99.5% following an additional dose.
“Most IBD patients, including those who are immune suppressed and/or did not have detectable humoral immune responses following the initial mRNA COVID-19 vaccine series, demonstrate strong immune responses to additional doses of mRNA vaccines,” Michael D. Kappelman, MD, a pediatric gastroenterologist at the University of North Carolina at Chapel Hill, told this news organization.
“These data support an additional vaccine dose of mRNA vaccine in patients at risk for an inadequate response to the initial series,” he said.
Dr. Kappelman presented these findings on behalf of the PREVENT-COVID Study Group as an e-poster at the 17th congress of the European Crohn’s and Colitis Organisation.
A study design to measure boosters’ benefits
For people with Crohn’s disease or ulcerative colitis who are taking immunosuppressants, boosters are generally recommended, Dr. Kappelman and colleagues noted. However, “real-world data on the effectiveness and safety of additional vaccine doses are lacking.”
They studied 659 people with IBD (mean age, 45 years; 72% female), of whom 72% had Crohn’s disease and 27% had ulcerative colitis/unclassified IBD.
Of these participants, 63% received Pfizer/BioNTech vaccine and 37% received the Moderna vaccine. Five participants received the Johnson & Johnson vaccine. In 98% of cases, people who received an mRNA vaccine initially also received the same type for the additional dose.
Participants completed baseline and follow-up surveys. Their blood work was obtained and evaluated 8 weeks after completion of the initial vaccine series and 6 weeks after a booster to measure anti–receptor binding domain IgG antibody levels specific to SARS-CoV-2.
Mean increase in antibody levels was 61 µg/mL in the Pfizer vaccine group and 78 µg/mL in the Moderna vaccine group following the booster shot.
Of the 47 patients without initial antibody response, 45 (96%) had detectable antibodies following an additional dose.
Serious adverse events (AEs) associated with the booster were rare, Dr. Kappelman said. Among participants, 44% reported no AEs, 24% mild AEs, 25% moderate AEs, and 6% reported serious AEs.
“These data can be used to inform vaccine decisions in patients with a broad array of immune-medicated conditions frequently managed by immunosuppression,” the investigators note.
A ‘reassuring’ finding
“This abstract [gives us] an important understanding about how patients with inflammatory bowel disease respond to COVID-19 vaccination. There have been mixed reports in the prior studies regarding how well patients with IBD respond to vaccination,” Jason Ken Hou, MD, said when asked to comment on the research.
The main findings that 99.5% of patients had detectable antibodies after an additional dose “is reassuring, as prior studies have suggested some patients did not develop antibodies after the [initial series],” added Dr. Hou, associate professor of medicine-gastroenterology at Baylor College of Medicine in Houston.
The researchers conducted the study within a previously established, well-known Internet-based cohort of IBD patients, Dr. Hou said. Although the researchers collected information on the IBD medications that patients were taking at the time of vaccination, the analyses that were presented did not compare antibody response rates based on medication.
“Further study is still required, as there is more to vaccination response than detectable antibody alone,” he added.
A version of this article first appeared on Medscape.com.
Of the study participants, 93% had detectable antibodies after their initial vaccination series, which increased to 99.5% following an additional dose.
“Most IBD patients, including those who are immune suppressed and/or did not have detectable humoral immune responses following the initial mRNA COVID-19 vaccine series, demonstrate strong immune responses to additional doses of mRNA vaccines,” Michael D. Kappelman, MD, a pediatric gastroenterologist at the University of North Carolina at Chapel Hill, told this news organization.
“These data support an additional vaccine dose of mRNA vaccine in patients at risk for an inadequate response to the initial series,” he said.
Dr. Kappelman presented these findings on behalf of the PREVENT-COVID Study Group as an e-poster at the 17th congress of the European Crohn’s and Colitis Organisation.
A study design to measure boosters’ benefits
For people with Crohn’s disease or ulcerative colitis who are taking immunosuppressants, boosters are generally recommended, Dr. Kappelman and colleagues noted. However, “real-world data on the effectiveness and safety of additional vaccine doses are lacking.”
They studied 659 people with IBD (mean age, 45 years; 72% female), of whom 72% had Crohn’s disease and 27% had ulcerative colitis/unclassified IBD.
Of these participants, 63% received Pfizer/BioNTech vaccine and 37% received the Moderna vaccine. Five participants received the Johnson & Johnson vaccine. In 98% of cases, people who received an mRNA vaccine initially also received the same type for the additional dose.
Participants completed baseline and follow-up surveys. Their blood work was obtained and evaluated 8 weeks after completion of the initial vaccine series and 6 weeks after a booster to measure anti–receptor binding domain IgG antibody levels specific to SARS-CoV-2.
Mean increase in antibody levels was 61 µg/mL in the Pfizer vaccine group and 78 µg/mL in the Moderna vaccine group following the booster shot.
Of the 47 patients without initial antibody response, 45 (96%) had detectable antibodies following an additional dose.
Serious adverse events (AEs) associated with the booster were rare, Dr. Kappelman said. Among participants, 44% reported no AEs, 24% mild AEs, 25% moderate AEs, and 6% reported serious AEs.
“These data can be used to inform vaccine decisions in patients with a broad array of immune-medicated conditions frequently managed by immunosuppression,” the investigators note.
A ‘reassuring’ finding
“This abstract [gives us] an important understanding about how patients with inflammatory bowel disease respond to COVID-19 vaccination. There have been mixed reports in the prior studies regarding how well patients with IBD respond to vaccination,” Jason Ken Hou, MD, said when asked to comment on the research.
The main findings that 99.5% of patients had detectable antibodies after an additional dose “is reassuring, as prior studies have suggested some patients did not develop antibodies after the [initial series],” added Dr. Hou, associate professor of medicine-gastroenterology at Baylor College of Medicine in Houston.
The researchers conducted the study within a previously established, well-known Internet-based cohort of IBD patients, Dr. Hou said. Although the researchers collected information on the IBD medications that patients were taking at the time of vaccination, the analyses that were presented did not compare antibody response rates based on medication.
“Further study is still required, as there is more to vaccination response than detectable antibody alone,” he added.
A version of this article first appeared on Medscape.com.
Of the study participants, 93% had detectable antibodies after their initial vaccination series, which increased to 99.5% following an additional dose.
“Most IBD patients, including those who are immune suppressed and/or did not have detectable humoral immune responses following the initial mRNA COVID-19 vaccine series, demonstrate strong immune responses to additional doses of mRNA vaccines,” Michael D. Kappelman, MD, a pediatric gastroenterologist at the University of North Carolina at Chapel Hill, told this news organization.
“These data support an additional vaccine dose of mRNA vaccine in patients at risk for an inadequate response to the initial series,” he said.
Dr. Kappelman presented these findings on behalf of the PREVENT-COVID Study Group as an e-poster at the 17th congress of the European Crohn’s and Colitis Organisation.
A study design to measure boosters’ benefits
For people with Crohn’s disease or ulcerative colitis who are taking immunosuppressants, boosters are generally recommended, Dr. Kappelman and colleagues noted. However, “real-world data on the effectiveness and safety of additional vaccine doses are lacking.”
They studied 659 people with IBD (mean age, 45 years; 72% female), of whom 72% had Crohn’s disease and 27% had ulcerative colitis/unclassified IBD.
Of these participants, 63% received Pfizer/BioNTech vaccine and 37% received the Moderna vaccine. Five participants received the Johnson & Johnson vaccine. In 98% of cases, people who received an mRNA vaccine initially also received the same type for the additional dose.
Participants completed baseline and follow-up surveys. Their blood work was obtained and evaluated 8 weeks after completion of the initial vaccine series and 6 weeks after a booster to measure anti–receptor binding domain IgG antibody levels specific to SARS-CoV-2.
Mean increase in antibody levels was 61 µg/mL in the Pfizer vaccine group and 78 µg/mL in the Moderna vaccine group following the booster shot.
Of the 47 patients without initial antibody response, 45 (96%) had detectable antibodies following an additional dose.
Serious adverse events (AEs) associated with the booster were rare, Dr. Kappelman said. Among participants, 44% reported no AEs, 24% mild AEs, 25% moderate AEs, and 6% reported serious AEs.
“These data can be used to inform vaccine decisions in patients with a broad array of immune-medicated conditions frequently managed by immunosuppression,” the investigators note.
A ‘reassuring’ finding
“This abstract [gives us] an important understanding about how patients with inflammatory bowel disease respond to COVID-19 vaccination. There have been mixed reports in the prior studies regarding how well patients with IBD respond to vaccination,” Jason Ken Hou, MD, said when asked to comment on the research.
The main findings that 99.5% of patients had detectable antibodies after an additional dose “is reassuring, as prior studies have suggested some patients did not develop antibodies after the [initial series],” added Dr. Hou, associate professor of medicine-gastroenterology at Baylor College of Medicine in Houston.
The researchers conducted the study within a previously established, well-known Internet-based cohort of IBD patients, Dr. Hou said. Although the researchers collected information on the IBD medications that patients were taking at the time of vaccination, the analyses that were presented did not compare antibody response rates based on medication.
“Further study is still required, as there is more to vaccination response than detectable antibody alone,” he added.
A version of this article first appeared on Medscape.com.
FROM ECCO 2022
First possible case of deer-to-human COVID transmission identified
according a new preprint study that hasn’t yet been peer-reviewed.
Typically, humans spread the virus to deer, and then deer spread it to other deer. But new evidence suggests that the virus could spill over from deer into humans. The researchers identified a COVID-19 case in someone from Ontario who had recently been in contact with deer.
“This particular case, while raising a red flag, doesn’t seem to be hugely alarming,” Finlay Maguire, PhD, one of the study authors and an epidemiologist at Dalhousie University, told CBC News.
“While we haven’t seen [transmission from deer to humans] happen directly, we sampled from the human case around the same time we sampled from the deer, and we sampled from around the same location,” he said. “There is also a plausible link by which it could have happened, in that the individual involved is known to have had considerable contact with deer.”
Dr. Maguire and colleagues have been monitoring the spread of the coronavirus among animals. They analyzed nasal swabs and lymph node samples taken from hundreds of deer that were killed by hunters in fall 2021 in southwestern and eastern Ontario. Among 298 sampled deer, 17 tested positive -- all from southwestern Ontario.
During the analysis, they found a “highly divergent” coronavirus lineage, which means a cluster of samples with many mutations. Around the same time, they found a genetically similar version in a person from the same region.
The study points to the need for better surveillance of the coronavirus, Dr. Maguire told CBC News, including in humans, animals, plants, and the broader environment. Researchers aren’t quite sure how deer contract the virus from humans, but it could happen through contaminated water, direct contact, food, farming, or other animals such as mink.
The coronavirus lineage identified in the study is different from what’s circulating among humans now, and it’s not related to the Delta or Omicron variants. The closest genetic relative came from samples taken from humans and mink in Michigan in 2020, which means the divergent lineage mutated and evolved over time.
“It’s reassuring that we found no evidence of further transmission, during a time when we were doing a lot of sampling and a lot of sequencing,” Samira Mubareka, MD, one of the study authors and a virologist at Sunnybrook Health Sciences Centre, told CBC News.
“If we continue to do this surveillance, we’ll get a much better sense of what the actual risk is,” she said.
So far, the coronavirus has been found in wild white-tailed deer in the northeastern United States and central Canadian provinces.
Other known cases of transmission from animals to humans have been identified in farmed mink and potentially hamsters, the news outlet reported. But for the most part, humans transmit the virus to animals and are most likely to catch the virus from other people.
At the same time, the Public Health Agency of Canada has issued guidance for hunters, trappers, and those who handle wild deer. People should wear gloves, goggles, and a mask when they could be exposed to respiratory tissues and fluids, especially indoors.
Coronaviruses are killed by normal cooking temperatures, the agency said, and there has been no evidence that cooked venison can spread the virus.
A version of this article first appeared on WebMD.com.
according a new preprint study that hasn’t yet been peer-reviewed.
Typically, humans spread the virus to deer, and then deer spread it to other deer. But new evidence suggests that the virus could spill over from deer into humans. The researchers identified a COVID-19 case in someone from Ontario who had recently been in contact with deer.
“This particular case, while raising a red flag, doesn’t seem to be hugely alarming,” Finlay Maguire, PhD, one of the study authors and an epidemiologist at Dalhousie University, told CBC News.
“While we haven’t seen [transmission from deer to humans] happen directly, we sampled from the human case around the same time we sampled from the deer, and we sampled from around the same location,” he said. “There is also a plausible link by which it could have happened, in that the individual involved is known to have had considerable contact with deer.”
Dr. Maguire and colleagues have been monitoring the spread of the coronavirus among animals. They analyzed nasal swabs and lymph node samples taken from hundreds of deer that were killed by hunters in fall 2021 in southwestern and eastern Ontario. Among 298 sampled deer, 17 tested positive -- all from southwestern Ontario.
During the analysis, they found a “highly divergent” coronavirus lineage, which means a cluster of samples with many mutations. Around the same time, they found a genetically similar version in a person from the same region.
The study points to the need for better surveillance of the coronavirus, Dr. Maguire told CBC News, including in humans, animals, plants, and the broader environment. Researchers aren’t quite sure how deer contract the virus from humans, but it could happen through contaminated water, direct contact, food, farming, or other animals such as mink.
The coronavirus lineage identified in the study is different from what’s circulating among humans now, and it’s not related to the Delta or Omicron variants. The closest genetic relative came from samples taken from humans and mink in Michigan in 2020, which means the divergent lineage mutated and evolved over time.
“It’s reassuring that we found no evidence of further transmission, during a time when we were doing a lot of sampling and a lot of sequencing,” Samira Mubareka, MD, one of the study authors and a virologist at Sunnybrook Health Sciences Centre, told CBC News.
“If we continue to do this surveillance, we’ll get a much better sense of what the actual risk is,” she said.
So far, the coronavirus has been found in wild white-tailed deer in the northeastern United States and central Canadian provinces.
Other known cases of transmission from animals to humans have been identified in farmed mink and potentially hamsters, the news outlet reported. But for the most part, humans transmit the virus to animals and are most likely to catch the virus from other people.
At the same time, the Public Health Agency of Canada has issued guidance for hunters, trappers, and those who handle wild deer. People should wear gloves, goggles, and a mask when they could be exposed to respiratory tissues and fluids, especially indoors.
Coronaviruses are killed by normal cooking temperatures, the agency said, and there has been no evidence that cooked venison can spread the virus.
A version of this article first appeared on WebMD.com.
according a new preprint study that hasn’t yet been peer-reviewed.
Typically, humans spread the virus to deer, and then deer spread it to other deer. But new evidence suggests that the virus could spill over from deer into humans. The researchers identified a COVID-19 case in someone from Ontario who had recently been in contact with deer.
“This particular case, while raising a red flag, doesn’t seem to be hugely alarming,” Finlay Maguire, PhD, one of the study authors and an epidemiologist at Dalhousie University, told CBC News.
“While we haven’t seen [transmission from deer to humans] happen directly, we sampled from the human case around the same time we sampled from the deer, and we sampled from around the same location,” he said. “There is also a plausible link by which it could have happened, in that the individual involved is known to have had considerable contact with deer.”
Dr. Maguire and colleagues have been monitoring the spread of the coronavirus among animals. They analyzed nasal swabs and lymph node samples taken from hundreds of deer that were killed by hunters in fall 2021 in southwestern and eastern Ontario. Among 298 sampled deer, 17 tested positive -- all from southwestern Ontario.
During the analysis, they found a “highly divergent” coronavirus lineage, which means a cluster of samples with many mutations. Around the same time, they found a genetically similar version in a person from the same region.
The study points to the need for better surveillance of the coronavirus, Dr. Maguire told CBC News, including in humans, animals, plants, and the broader environment. Researchers aren’t quite sure how deer contract the virus from humans, but it could happen through contaminated water, direct contact, food, farming, or other animals such as mink.
The coronavirus lineage identified in the study is different from what’s circulating among humans now, and it’s not related to the Delta or Omicron variants. The closest genetic relative came from samples taken from humans and mink in Michigan in 2020, which means the divergent lineage mutated and evolved over time.
“It’s reassuring that we found no evidence of further transmission, during a time when we were doing a lot of sampling and a lot of sequencing,” Samira Mubareka, MD, one of the study authors and a virologist at Sunnybrook Health Sciences Centre, told CBC News.
“If we continue to do this surveillance, we’ll get a much better sense of what the actual risk is,” she said.
So far, the coronavirus has been found in wild white-tailed deer in the northeastern United States and central Canadian provinces.
Other known cases of transmission from animals to humans have been identified in farmed mink and potentially hamsters, the news outlet reported. But for the most part, humans transmit the virus to animals and are most likely to catch the virus from other people.
At the same time, the Public Health Agency of Canada has issued guidance for hunters, trappers, and those who handle wild deer. People should wear gloves, goggles, and a mask when they could be exposed to respiratory tissues and fluids, especially indoors.
Coronaviruses are killed by normal cooking temperatures, the agency said, and there has been no evidence that cooked venison can spread the virus.
A version of this article first appeared on WebMD.com.
EMA recommends PreHevbri hepatitis B vaccine for approval
The European Medicines Agency’s (EMA’s) human medicines committee has recommended approval of a hepatitis B vaccine for adults.
The agency’s Committee for Medicinal Products for Human Use (CHMP) granted a positive opinion for PreHevbri on Feb. 24 for active immunization against hepatitis B virus (HBV) infection. PreHevbri (PreHevBrio in the United States and Sci-B-Vac in Israel) received approval from the Food and Drug Administration on Nov. 30, 2021. The vaccine is produced by VBI Vaccines (Delaware) Inc., based in Cambridge, Mass.
The World Health Organization estimates that more than 290 million people globally are infected with HBV. HBV is the leading cause of liver disease, and an estimated 900,000 people die every year from complications from chronic HBV infection, according to a VBI Vaccine press release. A 2019 report from the European Centre for Disease Prevention and Control found that adults in the European Union aged 35-44 had the highest rates of acute infections with HBV, and people aged 25-34 had the highest rate of chronic HBV infections. Vaccination programs are key interventions in preventing transmission of the virus, the report noted.
PreHevbri is a hepatitis B vaccine composed of three surface antigens of the hepatitis B virus. The vaccine is administered via injection in three doses on a 0-, 1-, and 6-month schedule and is indicated for use in adults aged 18 years and older.
The CHMP recommendation was based on data from a safety and immunogenicity study, which included 1,607 participants aged 18 and older, and a lot-to-lot study, which included 2,838 adults aged 18-45, according the VBI vaccine press release.
The recommendation will now be reviewed by the European Commission. If approved, PreHevbri will be the only three-antigen HBV vaccine for adults approved in the European Union.
A version of this article first appeared on Medscape.com.
The European Medicines Agency’s (EMA’s) human medicines committee has recommended approval of a hepatitis B vaccine for adults.
The agency’s Committee for Medicinal Products for Human Use (CHMP) granted a positive opinion for PreHevbri on Feb. 24 for active immunization against hepatitis B virus (HBV) infection. PreHevbri (PreHevBrio in the United States and Sci-B-Vac in Israel) received approval from the Food and Drug Administration on Nov. 30, 2021. The vaccine is produced by VBI Vaccines (Delaware) Inc., based in Cambridge, Mass.
The World Health Organization estimates that more than 290 million people globally are infected with HBV. HBV is the leading cause of liver disease, and an estimated 900,000 people die every year from complications from chronic HBV infection, according to a VBI Vaccine press release. A 2019 report from the European Centre for Disease Prevention and Control found that adults in the European Union aged 35-44 had the highest rates of acute infections with HBV, and people aged 25-34 had the highest rate of chronic HBV infections. Vaccination programs are key interventions in preventing transmission of the virus, the report noted.
PreHevbri is a hepatitis B vaccine composed of three surface antigens of the hepatitis B virus. The vaccine is administered via injection in three doses on a 0-, 1-, and 6-month schedule and is indicated for use in adults aged 18 years and older.
The CHMP recommendation was based on data from a safety and immunogenicity study, which included 1,607 participants aged 18 and older, and a lot-to-lot study, which included 2,838 adults aged 18-45, according the VBI vaccine press release.
The recommendation will now be reviewed by the European Commission. If approved, PreHevbri will be the only three-antigen HBV vaccine for adults approved in the European Union.
A version of this article first appeared on Medscape.com.
The European Medicines Agency’s (EMA’s) human medicines committee has recommended approval of a hepatitis B vaccine for adults.
The agency’s Committee for Medicinal Products for Human Use (CHMP) granted a positive opinion for PreHevbri on Feb. 24 for active immunization against hepatitis B virus (HBV) infection. PreHevbri (PreHevBrio in the United States and Sci-B-Vac in Israel) received approval from the Food and Drug Administration on Nov. 30, 2021. The vaccine is produced by VBI Vaccines (Delaware) Inc., based in Cambridge, Mass.
The World Health Organization estimates that more than 290 million people globally are infected with HBV. HBV is the leading cause of liver disease, and an estimated 900,000 people die every year from complications from chronic HBV infection, according to a VBI Vaccine press release. A 2019 report from the European Centre for Disease Prevention and Control found that adults in the European Union aged 35-44 had the highest rates of acute infections with HBV, and people aged 25-34 had the highest rate of chronic HBV infections. Vaccination programs are key interventions in preventing transmission of the virus, the report noted.
PreHevbri is a hepatitis B vaccine composed of three surface antigens of the hepatitis B virus. The vaccine is administered via injection in three doses on a 0-, 1-, and 6-month schedule and is indicated for use in adults aged 18 years and older.
The CHMP recommendation was based on data from a safety and immunogenicity study, which included 1,607 participants aged 18 and older, and a lot-to-lot study, which included 2,838 adults aged 18-45, according the VBI vaccine press release.
The recommendation will now be reviewed by the European Commission. If approved, PreHevbri will be the only three-antigen HBV vaccine for adults approved in the European Union.
A version of this article first appeared on Medscape.com.
Some physicians still lack access to COVID-19 vaccines
It would be overused and trite to say that the pandemic has drastically altered all of our lives and will cause lasting impact on how we function in society and medicine for years to come. While it seems that the current trend of the latest Omicron variant is on the downslope, the path to get to this point has been fraught with challenges that have struck at the very core of our society. As a primary care physician on the front lines seeing COVID patients, I have had to deal with not only the disease but the politics around it. I practice in Florida, and I still cannot give COVID vaccines in my office.
I am a firm believer in the ability for physicians to be able to give all the necessary adult vaccines and provide them for their patients. The COVID vaccine exacerbated a majorly flawed system that further increased the health care disparities in the country. The current vaccine system for the majority of adult vaccines involves the physician’s being able to directly purchase supplies from the vaccine manufacturer, administer them to the patients, and be reimbursed.
Third parties can purchase vaccines at lower rates than those for physicians
The Affordable Care Act mandates that all vaccines approved by the Advisory Committee on Immunization Practices (ACIP) at the Centers for Disease Control and Prevention must be covered. This allows for better access to care as physicians will be able to purchase, store, and deliver vaccines to their patients. The fallacy in this system is that third parties get involved and rebates or incentives are given to these groups to purchase vaccines at a rate lower than those for physicians.
In addition, many organizations can get access to vaccines before physicians and at a lower cost. That system was flawed to begin with and created a deterrent for access to care and physician involvement in the vaccination process. This was worsened by different states being given the ability to decide how vaccines would be distributed for COVID.
Many pharmacies were able to give out COVID vaccines while many physician offices still have not received access to any of the vaccines. One of the major safety issues with this is that no physicians were involved in the administration of the vaccine, and it is unclear what training was given to the individuals injecting that vaccine. Finally, different places were interpreting the recommendations from ACIP on their own and not necessarily following the appropriate guidelines. All of these factors have further widened the health care disparity gap and made it difficult to provide the COVID vaccines in doctors’ offices.
Recommended next steps, solutions to problem
The question is what to do about this. The most important thing is to get the vaccines in arms so they can save lives. In addition, doctors need to be able to get the vaccines in their offices.
Many patients trust their physicians to advise them on what to do regarding health care. The majority of patients want to know if they should get the vaccine and ask for counseling. Physicians answering patients’ questions about vaccines is an important step in overcoming vaccine hesitancy.
Also, doctors need to be informed and supportive of the vaccine process.
The next step is the governmental aspect with those in power making sure that vaccines are accessible to all. Even if the vaccine cannot be given in the office, doctors should still be recommending that patients receive them. Plus, doctors should take every opportunity to ask about what vaccines their patients have received and encourage their patients to get vaccinated.
The COVID-19 vaccines are safe and effective and have been monitored for safety more than any other vaccine. There are multiple systems in place to look for any signals that could indicate an issue was caused by a COVID-19 vaccine. These vaccines can be administered with other vaccines, and there is a great opportunity for physicians to encourage patients to receive these life-saving vaccines.
While it may seem that the COVID-19 case counts are on the downslope, the importance of continuing to vaccinate is predicated on the very real concern that the disease is still circulating and the unvaccinated are still at risk for severe infection.
Dr. Goldman is immediate past governor of the Florida chapter of the American College of Physicians, a regent for the American College of Physicians, vice-president of the Florida Medical Association, and president of the Florida Medical Association Political Action Committee. You can reach Dr. Goldman at [email protected].
It would be overused and trite to say that the pandemic has drastically altered all of our lives and will cause lasting impact on how we function in society and medicine for years to come. While it seems that the current trend of the latest Omicron variant is on the downslope, the path to get to this point has been fraught with challenges that have struck at the very core of our society. As a primary care physician on the front lines seeing COVID patients, I have had to deal with not only the disease but the politics around it. I practice in Florida, and I still cannot give COVID vaccines in my office.
I am a firm believer in the ability for physicians to be able to give all the necessary adult vaccines and provide them for their patients. The COVID vaccine exacerbated a majorly flawed system that further increased the health care disparities in the country. The current vaccine system for the majority of adult vaccines involves the physician’s being able to directly purchase supplies from the vaccine manufacturer, administer them to the patients, and be reimbursed.
Third parties can purchase vaccines at lower rates than those for physicians
The Affordable Care Act mandates that all vaccines approved by the Advisory Committee on Immunization Practices (ACIP) at the Centers for Disease Control and Prevention must be covered. This allows for better access to care as physicians will be able to purchase, store, and deliver vaccines to their patients. The fallacy in this system is that third parties get involved and rebates or incentives are given to these groups to purchase vaccines at a rate lower than those for physicians.
In addition, many organizations can get access to vaccines before physicians and at a lower cost. That system was flawed to begin with and created a deterrent for access to care and physician involvement in the vaccination process. This was worsened by different states being given the ability to decide how vaccines would be distributed for COVID.
Many pharmacies were able to give out COVID vaccines while many physician offices still have not received access to any of the vaccines. One of the major safety issues with this is that no physicians were involved in the administration of the vaccine, and it is unclear what training was given to the individuals injecting that vaccine. Finally, different places were interpreting the recommendations from ACIP on their own and not necessarily following the appropriate guidelines. All of these factors have further widened the health care disparity gap and made it difficult to provide the COVID vaccines in doctors’ offices.
Recommended next steps, solutions to problem
The question is what to do about this. The most important thing is to get the vaccines in arms so they can save lives. In addition, doctors need to be able to get the vaccines in their offices.
Many patients trust their physicians to advise them on what to do regarding health care. The majority of patients want to know if they should get the vaccine and ask for counseling. Physicians answering patients’ questions about vaccines is an important step in overcoming vaccine hesitancy.
Also, doctors need to be informed and supportive of the vaccine process.
The next step is the governmental aspect with those in power making sure that vaccines are accessible to all. Even if the vaccine cannot be given in the office, doctors should still be recommending that patients receive them. Plus, doctors should take every opportunity to ask about what vaccines their patients have received and encourage their patients to get vaccinated.
The COVID-19 vaccines are safe and effective and have been monitored for safety more than any other vaccine. There are multiple systems in place to look for any signals that could indicate an issue was caused by a COVID-19 vaccine. These vaccines can be administered with other vaccines, and there is a great opportunity for physicians to encourage patients to receive these life-saving vaccines.
While it may seem that the COVID-19 case counts are on the downslope, the importance of continuing to vaccinate is predicated on the very real concern that the disease is still circulating and the unvaccinated are still at risk for severe infection.
Dr. Goldman is immediate past governor of the Florida chapter of the American College of Physicians, a regent for the American College of Physicians, vice-president of the Florida Medical Association, and president of the Florida Medical Association Political Action Committee. You can reach Dr. Goldman at [email protected].
It would be overused and trite to say that the pandemic has drastically altered all of our lives and will cause lasting impact on how we function in society and medicine for years to come. While it seems that the current trend of the latest Omicron variant is on the downslope, the path to get to this point has been fraught with challenges that have struck at the very core of our society. As a primary care physician on the front lines seeing COVID patients, I have had to deal with not only the disease but the politics around it. I practice in Florida, and I still cannot give COVID vaccines in my office.
I am a firm believer in the ability for physicians to be able to give all the necessary adult vaccines and provide them for their patients. The COVID vaccine exacerbated a majorly flawed system that further increased the health care disparities in the country. The current vaccine system for the majority of adult vaccines involves the physician’s being able to directly purchase supplies from the vaccine manufacturer, administer them to the patients, and be reimbursed.
Third parties can purchase vaccines at lower rates than those for physicians
The Affordable Care Act mandates that all vaccines approved by the Advisory Committee on Immunization Practices (ACIP) at the Centers for Disease Control and Prevention must be covered. This allows for better access to care as physicians will be able to purchase, store, and deliver vaccines to their patients. The fallacy in this system is that third parties get involved and rebates or incentives are given to these groups to purchase vaccines at a rate lower than those for physicians.
In addition, many organizations can get access to vaccines before physicians and at a lower cost. That system was flawed to begin with and created a deterrent for access to care and physician involvement in the vaccination process. This was worsened by different states being given the ability to decide how vaccines would be distributed for COVID.
Many pharmacies were able to give out COVID vaccines while many physician offices still have not received access to any of the vaccines. One of the major safety issues with this is that no physicians were involved in the administration of the vaccine, and it is unclear what training was given to the individuals injecting that vaccine. Finally, different places were interpreting the recommendations from ACIP on their own and not necessarily following the appropriate guidelines. All of these factors have further widened the health care disparity gap and made it difficult to provide the COVID vaccines in doctors’ offices.
Recommended next steps, solutions to problem
The question is what to do about this. The most important thing is to get the vaccines in arms so they can save lives. In addition, doctors need to be able to get the vaccines in their offices.
Many patients trust their physicians to advise them on what to do regarding health care. The majority of patients want to know if they should get the vaccine and ask for counseling. Physicians answering patients’ questions about vaccines is an important step in overcoming vaccine hesitancy.
Also, doctors need to be informed and supportive of the vaccine process.
The next step is the governmental aspect with those in power making sure that vaccines are accessible to all. Even if the vaccine cannot be given in the office, doctors should still be recommending that patients receive them. Plus, doctors should take every opportunity to ask about what vaccines their patients have received and encourage their patients to get vaccinated.
The COVID-19 vaccines are safe and effective and have been monitored for safety more than any other vaccine. There are multiple systems in place to look for any signals that could indicate an issue was caused by a COVID-19 vaccine. These vaccines can be administered with other vaccines, and there is a great opportunity for physicians to encourage patients to receive these life-saving vaccines.
While it may seem that the COVID-19 case counts are on the downslope, the importance of continuing to vaccinate is predicated on the very real concern that the disease is still circulating and the unvaccinated are still at risk for severe infection.
Dr. Goldman is immediate past governor of the Florida chapter of the American College of Physicians, a regent for the American College of Physicians, vice-president of the Florida Medical Association, and president of the Florida Medical Association Political Action Committee. You can reach Dr. Goldman at [email protected].
GI involvement may signal risk for MIS-C after COVID
While evaluating an adolescent who had endured a several-day history of vomiting and diarrhea, I mentioned the likelihood of a viral causation, including SARS-CoV-2 infection. His well-informed mother responded, “He has no respiratory symptoms. Does COVID cause GI disease?”
Indeed, not only is the gastrointestinal tract a potential portal of entry of the virus but it may well be the site of mediation of both local and remote injury and thus a harbinger of more severe clinical phenotypes.
As we learn more about the clinical spectrum of COVID, it is becoming increasingly clear that certain features of GI tract involvement may allow us to establish a timeline of the clinical course and perhaps predict the outcome.
The GI tract’s involvement isn’t surprising
The ways in which the GI tract serves as a target organ of SARS-CoV-2 have been postulated in the literature. In part, this is related to the presence of abundant receptors for SARS-CoV-2 cell binding and internalization. The virus uses angiotensin-converting enzyme 2 receptors to enter various cells. These receptors are highly expressed on not only lung cells but also enterocytes. Binding of SARS-CoV-2 to ACE2 receptors allows GI involvement, leading to microscopic mucosal inflammation, increased permeability, and altered intestinal absorption.
The clinical GI manifestations of this include anorexia, nausea, vomiting, diarrhea, and abdominal pain, which may be the earliest, or sole, symptoms of COVID-19, often noted before the onset of fever or respiratory symptoms. In fact, John Ong, MBBS, and colleagues, in a discussion about patients with primary GI SARS-CoV-2 infection and symptoms, use the term “GI-COVID.”
Clinical course of GI manifestations
After SARS-CoV-2 exposure, adults most commonly present with respiratory symptoms, with GI symptoms reported in 10%-15% of cases. However, the overall incidence of GI involvement during SARS-CoV-2 infection varies according to age, with children more likely than adults to manifest intestinal symptoms.
There are also differences in incidence reported when comparing hospitalized with nonhospitalized individuals. In early reports from the onset of the COVID-19 pandemic, 11%-43% of hospitalized adult patients manifested GI symptoms. Of note, the presence of GI symptoms was associated with more severe disease and thus predictive of outcomes in those admitted to hospitals.
In a multicenter study that assessed pediatric inpatients with COVID-19, GI manifestations were present in 57% of patients and were the first manifestation in 14%. Adjusted by confounding factors, those with GI symptoms had a higher risk for pediatric intensive care unit admission. Patients admitted to the PICU also had higher serum C-reactive protein and aspartate aminotransferase values.
Emerging data on MIS-C
In previously healthy children and adolescents, the severe, life-threatening complication of multisystem inflammatory syndrome in children (MIS-C) may present 2-6 weeks after acute infection with SARS-CoV-2. MIS-C appears to be an immune activation syndrome and is presumed to be the delayed immunologic sequelae of mild/asymptomatic SARS-CoV-2 infection. This response manifests as hyperinflammation in conjunction with a peak in antibody production a few weeks later.
One report of 186 children with MIS-C in the United States noted that the involved organ system included the GI tract in 92%, followed by cardiovascular in 80%, hematologic in 76%, mucocutaneous in 74%, and respiratory in 70%. Affected children were hospitalized for a median of 7 days, with 80% requiring intensive care, 20% receiving mechanical ventilation, and 48% receiving vasoactive support; 2% died. In a similar study of patients hospitalized in New York, 88% had GI symptoms (abdominal pain, vomiting, and/or diarrhea). A retrospective chart review of patients with MIS-C found that the majority had GI symptoms with any portion of the GI tract potentially involved, but ileal and colonic inflammation predominated.
Elizabeth Whittaker, MD, and colleagues described the clinical characteristics of children in eight hospitals in England who met criteria for MIS-C that were temporally associated with SARS-CoV-2. At presentation, all of the patients manifested fever and nonspecific GI symptoms, including vomiting (45%), abdominal pain (53%), and diarrhea (52%). During hospitalization, 50% developed shock with evidence of myocardial dysfunction.
Ermias D. Belay, MD, and colleagues described the clinical characteristics of a large cohort of patients with MIS-C that were reported to the U.S. Centers for Disease Control and Prevention. Of 1,733 patients identified, GI symptoms were reported in 53%-67%. Over half developed hypotension or shock and were admitted for intensive care. Younger children more frequently presented with abdominal pain in contrast with adolescents, who more frequently manifest respiratory symptoms.
In a multicenter retrospective study of Italian children with COVID-19 that was conducted from the onset of the pandemic to early 2021, GI symptoms were noted in 38%. These manifestations were mild and self-limiting, comparable to other viral intestinal infections. However, a subset of children (9.5%) had severe GI manifestations of MIS-C, defined as a medical and/or radiologic diagnosis of acute abdomen, appendicitis, intussusception, pancreatitis, abdominal fluid collection, or diffuse adenomesenteritis requiring surgical consultation. Overall, 42% of this group underwent surgery. The authors noted that the clinical presentation of abdominal pain, lymphopenia, and increased C-reactive protein and ferritin levels were associated with a 9- to 30-fold increased probability of these severe sequelae. In addition, the severity of the GI manifestations was correlated with age (5-10 years: overall response, 8.33; >10 years: OR, 6.37). Again, the presence of GI symptoms was a harbinger of hospitalization and PICU admission.
Given that GI symptoms are a common presentation of MIS-C, its diagnosis may be delayed as clinicians first consider other GI/viral infections, inflammatory bowel disease, or Kawasaki disease. Prompt identification of GI involvement and awareness of the potential outcomes may guide the management and improve the outcome.
These studies provide a clear picture of the differential presenting features of COVID-19 and MIS-C. Although there may be other environmental/genetic factors that govern the incidence, impact, and manifestations, COVID’s status as an ongoing pandemic gives these observations worldwide relevance. This is evident in a recent report documenting pronounced GI symptoms in African children with COVID-19.
It should be noted, however, that the published data cited here reflect the impact of the initial variants of SARS-CoV-2. The GI binding, effects, and aftermath of infection with the Delta and Omicron variants is not yet known.
Cause and effect, or simply coincidental?
Some insight into MIS-C pathogenesis was provided by Lael M. Yonker, MD, and colleagues in their analysis of biospecimens from 100 children: 19 with MIS-C, 26 with acute COVID-19, and 55 controls. They demonstrated that in children with MIS-C the prolonged presence of SARS-CoV-2 in the GI tract led to the release of zonulin, a biomarker of intestinal permeability, with subsequent trafficking of SARS-CoV-2 antigens into the bloodstream, leading to hyperinflammation. They were then able to decrease plasma SARS-CoV-2 spike antigen levels and inflammatory markers, with resulting clinical improvement after administration of larazotide, a zonulin antagonist.
These observations regarding the potential mechanism and triggers of MIS-C may offer biomarkers for early detection and/or strategies for prevention and treatment of MIS-C.
Bottom line
The GI tract is the target of an immune-mediated inflammatory response that is triggered by SARS-CoV-2, with MIS-C being the major manifestation of the resultant high degree of inflammation. These observations will allow an increased awareness of nonrespiratory symptoms of SARS-CoV-2 infection by clinicians working in emergency departments and primary care settings.
Clues that may enhance the ability of pediatric clinicians to recognize the potential for severe GI involvement include the occurrence of abdominal pain, leukopenia, and elevated inflammatory markers. Their presence should raise suspicion of MIS-C and lead to early evaluation.
Of note, COVID-19 mRNA vaccination is associated with a lower incidence of MIS-C in adolescents. This underscores the importance of COVID vaccination for all eligible children. Yet, we clearly have our work cut out for us. Of 107 children with MIS-C who were hospitalized in France, 31% were adolescents eligible for vaccination; however, none had been fully vaccinated. At the end of 2021, CDC data noted that less than 1% of vaccine-eligible children (12-17 years) were fully vaccinated.
The Pfizer-BioNTech vaccine is now authorized for receipt by children aged 5-11 years, the age group that is at highest risk for MIS-C. However, despite the approval of vaccines for these younger children, there is limited access in some parts of the United States at a time of rising incidence.
We look forward to broad availability of pediatric vaccination strategies. In addition, with the intense focus on safe and effective therapeutics for SARS-CoV-2 infection, we hope to soon have strategies to prevent and/or treat the life-threatening manifestations and long-term consequences of MIS-C. For example, the recently reported central role of the gut microbiota in immunity against SARS-CoV-2 infection offer the possibility that “microbiota modulation” may both reduce GI injury and enhance vaccine efficacy.
Dr. Balistreri has disclosed no relevant financial relationships.
William F. Balistreri, MD, is the Dorothy M.M. Kersten Professor of Pediatrics; director emeritus, Pediatric Liver Care Center; medical director emeritus, liver transplantation; and professor, University of Cincinnati College of Medicine, department of pediatrics, Cincinnati Children’s Hospital Medical Center. He has served as director of the division of gastroenterology, hepatology, and nutrition at Cincinnati Children’s for 25 years and frequently covers gastroenterology, liver, and nutrition-related topics for this news organization. Dr Balistreri is currently editor-in-chief of the Journal of Pediatrics, having previously served as editor-in-chief of several journals and textbooks. He also became the first pediatrician to act as president of the American Association for the Study of Liver Diseases. In his spare time, he coaches youth lacrosse.
A version of this article first appeared on Medscape.com.
While evaluating an adolescent who had endured a several-day history of vomiting and diarrhea, I mentioned the likelihood of a viral causation, including SARS-CoV-2 infection. His well-informed mother responded, “He has no respiratory symptoms. Does COVID cause GI disease?”
Indeed, not only is the gastrointestinal tract a potential portal of entry of the virus but it may well be the site of mediation of both local and remote injury and thus a harbinger of more severe clinical phenotypes.
As we learn more about the clinical spectrum of COVID, it is becoming increasingly clear that certain features of GI tract involvement may allow us to establish a timeline of the clinical course and perhaps predict the outcome.
The GI tract’s involvement isn’t surprising
The ways in which the GI tract serves as a target organ of SARS-CoV-2 have been postulated in the literature. In part, this is related to the presence of abundant receptors for SARS-CoV-2 cell binding and internalization. The virus uses angiotensin-converting enzyme 2 receptors to enter various cells. These receptors are highly expressed on not only lung cells but also enterocytes. Binding of SARS-CoV-2 to ACE2 receptors allows GI involvement, leading to microscopic mucosal inflammation, increased permeability, and altered intestinal absorption.
The clinical GI manifestations of this include anorexia, nausea, vomiting, diarrhea, and abdominal pain, which may be the earliest, or sole, symptoms of COVID-19, often noted before the onset of fever or respiratory symptoms. In fact, John Ong, MBBS, and colleagues, in a discussion about patients with primary GI SARS-CoV-2 infection and symptoms, use the term “GI-COVID.”
Clinical course of GI manifestations
After SARS-CoV-2 exposure, adults most commonly present with respiratory symptoms, with GI symptoms reported in 10%-15% of cases. However, the overall incidence of GI involvement during SARS-CoV-2 infection varies according to age, with children more likely than adults to manifest intestinal symptoms.
There are also differences in incidence reported when comparing hospitalized with nonhospitalized individuals. In early reports from the onset of the COVID-19 pandemic, 11%-43% of hospitalized adult patients manifested GI symptoms. Of note, the presence of GI symptoms was associated with more severe disease and thus predictive of outcomes in those admitted to hospitals.
In a multicenter study that assessed pediatric inpatients with COVID-19, GI manifestations were present in 57% of patients and were the first manifestation in 14%. Adjusted by confounding factors, those with GI symptoms had a higher risk for pediatric intensive care unit admission. Patients admitted to the PICU also had higher serum C-reactive protein and aspartate aminotransferase values.
Emerging data on MIS-C
In previously healthy children and adolescents, the severe, life-threatening complication of multisystem inflammatory syndrome in children (MIS-C) may present 2-6 weeks after acute infection with SARS-CoV-2. MIS-C appears to be an immune activation syndrome and is presumed to be the delayed immunologic sequelae of mild/asymptomatic SARS-CoV-2 infection. This response manifests as hyperinflammation in conjunction with a peak in antibody production a few weeks later.
One report of 186 children with MIS-C in the United States noted that the involved organ system included the GI tract in 92%, followed by cardiovascular in 80%, hematologic in 76%, mucocutaneous in 74%, and respiratory in 70%. Affected children were hospitalized for a median of 7 days, with 80% requiring intensive care, 20% receiving mechanical ventilation, and 48% receiving vasoactive support; 2% died. In a similar study of patients hospitalized in New York, 88% had GI symptoms (abdominal pain, vomiting, and/or diarrhea). A retrospective chart review of patients with MIS-C found that the majority had GI symptoms with any portion of the GI tract potentially involved, but ileal and colonic inflammation predominated.
Elizabeth Whittaker, MD, and colleagues described the clinical characteristics of children in eight hospitals in England who met criteria for MIS-C that were temporally associated with SARS-CoV-2. At presentation, all of the patients manifested fever and nonspecific GI symptoms, including vomiting (45%), abdominal pain (53%), and diarrhea (52%). During hospitalization, 50% developed shock with evidence of myocardial dysfunction.
Ermias D. Belay, MD, and colleagues described the clinical characteristics of a large cohort of patients with MIS-C that were reported to the U.S. Centers for Disease Control and Prevention. Of 1,733 patients identified, GI symptoms were reported in 53%-67%. Over half developed hypotension or shock and were admitted for intensive care. Younger children more frequently presented with abdominal pain in contrast with adolescents, who more frequently manifest respiratory symptoms.
In a multicenter retrospective study of Italian children with COVID-19 that was conducted from the onset of the pandemic to early 2021, GI symptoms were noted in 38%. These manifestations were mild and self-limiting, comparable to other viral intestinal infections. However, a subset of children (9.5%) had severe GI manifestations of MIS-C, defined as a medical and/or radiologic diagnosis of acute abdomen, appendicitis, intussusception, pancreatitis, abdominal fluid collection, or diffuse adenomesenteritis requiring surgical consultation. Overall, 42% of this group underwent surgery. The authors noted that the clinical presentation of abdominal pain, lymphopenia, and increased C-reactive protein and ferritin levels were associated with a 9- to 30-fold increased probability of these severe sequelae. In addition, the severity of the GI manifestations was correlated with age (5-10 years: overall response, 8.33; >10 years: OR, 6.37). Again, the presence of GI symptoms was a harbinger of hospitalization and PICU admission.
Given that GI symptoms are a common presentation of MIS-C, its diagnosis may be delayed as clinicians first consider other GI/viral infections, inflammatory bowel disease, or Kawasaki disease. Prompt identification of GI involvement and awareness of the potential outcomes may guide the management and improve the outcome.
These studies provide a clear picture of the differential presenting features of COVID-19 and MIS-C. Although there may be other environmental/genetic factors that govern the incidence, impact, and manifestations, COVID’s status as an ongoing pandemic gives these observations worldwide relevance. This is evident in a recent report documenting pronounced GI symptoms in African children with COVID-19.
It should be noted, however, that the published data cited here reflect the impact of the initial variants of SARS-CoV-2. The GI binding, effects, and aftermath of infection with the Delta and Omicron variants is not yet known.
Cause and effect, or simply coincidental?
Some insight into MIS-C pathogenesis was provided by Lael M. Yonker, MD, and colleagues in their analysis of biospecimens from 100 children: 19 with MIS-C, 26 with acute COVID-19, and 55 controls. They demonstrated that in children with MIS-C the prolonged presence of SARS-CoV-2 in the GI tract led to the release of zonulin, a biomarker of intestinal permeability, with subsequent trafficking of SARS-CoV-2 antigens into the bloodstream, leading to hyperinflammation. They were then able to decrease plasma SARS-CoV-2 spike antigen levels and inflammatory markers, with resulting clinical improvement after administration of larazotide, a zonulin antagonist.
These observations regarding the potential mechanism and triggers of MIS-C may offer biomarkers for early detection and/or strategies for prevention and treatment of MIS-C.
Bottom line
The GI tract is the target of an immune-mediated inflammatory response that is triggered by SARS-CoV-2, with MIS-C being the major manifestation of the resultant high degree of inflammation. These observations will allow an increased awareness of nonrespiratory symptoms of SARS-CoV-2 infection by clinicians working in emergency departments and primary care settings.
Clues that may enhance the ability of pediatric clinicians to recognize the potential for severe GI involvement include the occurrence of abdominal pain, leukopenia, and elevated inflammatory markers. Their presence should raise suspicion of MIS-C and lead to early evaluation.
Of note, COVID-19 mRNA vaccination is associated with a lower incidence of MIS-C in adolescents. This underscores the importance of COVID vaccination for all eligible children. Yet, we clearly have our work cut out for us. Of 107 children with MIS-C who were hospitalized in France, 31% were adolescents eligible for vaccination; however, none had been fully vaccinated. At the end of 2021, CDC data noted that less than 1% of vaccine-eligible children (12-17 years) were fully vaccinated.
The Pfizer-BioNTech vaccine is now authorized for receipt by children aged 5-11 years, the age group that is at highest risk for MIS-C. However, despite the approval of vaccines for these younger children, there is limited access in some parts of the United States at a time of rising incidence.
We look forward to broad availability of pediatric vaccination strategies. In addition, with the intense focus on safe and effective therapeutics for SARS-CoV-2 infection, we hope to soon have strategies to prevent and/or treat the life-threatening manifestations and long-term consequences of MIS-C. For example, the recently reported central role of the gut microbiota in immunity against SARS-CoV-2 infection offer the possibility that “microbiota modulation” may both reduce GI injury and enhance vaccine efficacy.
Dr. Balistreri has disclosed no relevant financial relationships.
William F. Balistreri, MD, is the Dorothy M.M. Kersten Professor of Pediatrics; director emeritus, Pediatric Liver Care Center; medical director emeritus, liver transplantation; and professor, University of Cincinnati College of Medicine, department of pediatrics, Cincinnati Children’s Hospital Medical Center. He has served as director of the division of gastroenterology, hepatology, and nutrition at Cincinnati Children’s for 25 years and frequently covers gastroenterology, liver, and nutrition-related topics for this news organization. Dr Balistreri is currently editor-in-chief of the Journal of Pediatrics, having previously served as editor-in-chief of several journals and textbooks. He also became the first pediatrician to act as president of the American Association for the Study of Liver Diseases. In his spare time, he coaches youth lacrosse.
A version of this article first appeared on Medscape.com.
While evaluating an adolescent who had endured a several-day history of vomiting and diarrhea, I mentioned the likelihood of a viral causation, including SARS-CoV-2 infection. His well-informed mother responded, “He has no respiratory symptoms. Does COVID cause GI disease?”
Indeed, not only is the gastrointestinal tract a potential portal of entry of the virus but it may well be the site of mediation of both local and remote injury and thus a harbinger of more severe clinical phenotypes.
As we learn more about the clinical spectrum of COVID, it is becoming increasingly clear that certain features of GI tract involvement may allow us to establish a timeline of the clinical course and perhaps predict the outcome.
The GI tract’s involvement isn’t surprising
The ways in which the GI tract serves as a target organ of SARS-CoV-2 have been postulated in the literature. In part, this is related to the presence of abundant receptors for SARS-CoV-2 cell binding and internalization. The virus uses angiotensin-converting enzyme 2 receptors to enter various cells. These receptors are highly expressed on not only lung cells but also enterocytes. Binding of SARS-CoV-2 to ACE2 receptors allows GI involvement, leading to microscopic mucosal inflammation, increased permeability, and altered intestinal absorption.
The clinical GI manifestations of this include anorexia, nausea, vomiting, diarrhea, and abdominal pain, which may be the earliest, or sole, symptoms of COVID-19, often noted before the onset of fever or respiratory symptoms. In fact, John Ong, MBBS, and colleagues, in a discussion about patients with primary GI SARS-CoV-2 infection and symptoms, use the term “GI-COVID.”
Clinical course of GI manifestations
After SARS-CoV-2 exposure, adults most commonly present with respiratory symptoms, with GI symptoms reported in 10%-15% of cases. However, the overall incidence of GI involvement during SARS-CoV-2 infection varies according to age, with children more likely than adults to manifest intestinal symptoms.
There are also differences in incidence reported when comparing hospitalized with nonhospitalized individuals. In early reports from the onset of the COVID-19 pandemic, 11%-43% of hospitalized adult patients manifested GI symptoms. Of note, the presence of GI symptoms was associated with more severe disease and thus predictive of outcomes in those admitted to hospitals.
In a multicenter study that assessed pediatric inpatients with COVID-19, GI manifestations were present in 57% of patients and were the first manifestation in 14%. Adjusted by confounding factors, those with GI symptoms had a higher risk for pediatric intensive care unit admission. Patients admitted to the PICU also had higher serum C-reactive protein and aspartate aminotransferase values.
Emerging data on MIS-C
In previously healthy children and adolescents, the severe, life-threatening complication of multisystem inflammatory syndrome in children (MIS-C) may present 2-6 weeks after acute infection with SARS-CoV-2. MIS-C appears to be an immune activation syndrome and is presumed to be the delayed immunologic sequelae of mild/asymptomatic SARS-CoV-2 infection. This response manifests as hyperinflammation in conjunction with a peak in antibody production a few weeks later.
One report of 186 children with MIS-C in the United States noted that the involved organ system included the GI tract in 92%, followed by cardiovascular in 80%, hematologic in 76%, mucocutaneous in 74%, and respiratory in 70%. Affected children were hospitalized for a median of 7 days, with 80% requiring intensive care, 20% receiving mechanical ventilation, and 48% receiving vasoactive support; 2% died. In a similar study of patients hospitalized in New York, 88% had GI symptoms (abdominal pain, vomiting, and/or diarrhea). A retrospective chart review of patients with MIS-C found that the majority had GI symptoms with any portion of the GI tract potentially involved, but ileal and colonic inflammation predominated.
Elizabeth Whittaker, MD, and colleagues described the clinical characteristics of children in eight hospitals in England who met criteria for MIS-C that were temporally associated with SARS-CoV-2. At presentation, all of the patients manifested fever and nonspecific GI symptoms, including vomiting (45%), abdominal pain (53%), and diarrhea (52%). During hospitalization, 50% developed shock with evidence of myocardial dysfunction.
Ermias D. Belay, MD, and colleagues described the clinical characteristics of a large cohort of patients with MIS-C that were reported to the U.S. Centers for Disease Control and Prevention. Of 1,733 patients identified, GI symptoms were reported in 53%-67%. Over half developed hypotension or shock and were admitted for intensive care. Younger children more frequently presented with abdominal pain in contrast with adolescents, who more frequently manifest respiratory symptoms.
In a multicenter retrospective study of Italian children with COVID-19 that was conducted from the onset of the pandemic to early 2021, GI symptoms were noted in 38%. These manifestations were mild and self-limiting, comparable to other viral intestinal infections. However, a subset of children (9.5%) had severe GI manifestations of MIS-C, defined as a medical and/or radiologic diagnosis of acute abdomen, appendicitis, intussusception, pancreatitis, abdominal fluid collection, or diffuse adenomesenteritis requiring surgical consultation. Overall, 42% of this group underwent surgery. The authors noted that the clinical presentation of abdominal pain, lymphopenia, and increased C-reactive protein and ferritin levels were associated with a 9- to 30-fold increased probability of these severe sequelae. In addition, the severity of the GI manifestations was correlated with age (5-10 years: overall response, 8.33; >10 years: OR, 6.37). Again, the presence of GI symptoms was a harbinger of hospitalization and PICU admission.
Given that GI symptoms are a common presentation of MIS-C, its diagnosis may be delayed as clinicians first consider other GI/viral infections, inflammatory bowel disease, or Kawasaki disease. Prompt identification of GI involvement and awareness of the potential outcomes may guide the management and improve the outcome.
These studies provide a clear picture of the differential presenting features of COVID-19 and MIS-C. Although there may be other environmental/genetic factors that govern the incidence, impact, and manifestations, COVID’s status as an ongoing pandemic gives these observations worldwide relevance. This is evident in a recent report documenting pronounced GI symptoms in African children with COVID-19.
It should be noted, however, that the published data cited here reflect the impact of the initial variants of SARS-CoV-2. The GI binding, effects, and aftermath of infection with the Delta and Omicron variants is not yet known.
Cause and effect, or simply coincidental?
Some insight into MIS-C pathogenesis was provided by Lael M. Yonker, MD, and colleagues in their analysis of biospecimens from 100 children: 19 with MIS-C, 26 with acute COVID-19, and 55 controls. They demonstrated that in children with MIS-C the prolonged presence of SARS-CoV-2 in the GI tract led to the release of zonulin, a biomarker of intestinal permeability, with subsequent trafficking of SARS-CoV-2 antigens into the bloodstream, leading to hyperinflammation. They were then able to decrease plasma SARS-CoV-2 spike antigen levels and inflammatory markers, with resulting clinical improvement after administration of larazotide, a zonulin antagonist.
These observations regarding the potential mechanism and triggers of MIS-C may offer biomarkers for early detection and/or strategies for prevention and treatment of MIS-C.
Bottom line
The GI tract is the target of an immune-mediated inflammatory response that is triggered by SARS-CoV-2, with MIS-C being the major manifestation of the resultant high degree of inflammation. These observations will allow an increased awareness of nonrespiratory symptoms of SARS-CoV-2 infection by clinicians working in emergency departments and primary care settings.
Clues that may enhance the ability of pediatric clinicians to recognize the potential for severe GI involvement include the occurrence of abdominal pain, leukopenia, and elevated inflammatory markers. Their presence should raise suspicion of MIS-C and lead to early evaluation.
Of note, COVID-19 mRNA vaccination is associated with a lower incidence of MIS-C in adolescents. This underscores the importance of COVID vaccination for all eligible children. Yet, we clearly have our work cut out for us. Of 107 children with MIS-C who were hospitalized in France, 31% were adolescents eligible for vaccination; however, none had been fully vaccinated. At the end of 2021, CDC data noted that less than 1% of vaccine-eligible children (12-17 years) were fully vaccinated.
The Pfizer-BioNTech vaccine is now authorized for receipt by children aged 5-11 years, the age group that is at highest risk for MIS-C. However, despite the approval of vaccines for these younger children, there is limited access in some parts of the United States at a time of rising incidence.
We look forward to broad availability of pediatric vaccination strategies. In addition, with the intense focus on safe and effective therapeutics for SARS-CoV-2 infection, we hope to soon have strategies to prevent and/or treat the life-threatening manifestations and long-term consequences of MIS-C. For example, the recently reported central role of the gut microbiota in immunity against SARS-CoV-2 infection offer the possibility that “microbiota modulation” may both reduce GI injury and enhance vaccine efficacy.
Dr. Balistreri has disclosed no relevant financial relationships.
William F. Balistreri, MD, is the Dorothy M.M. Kersten Professor of Pediatrics; director emeritus, Pediatric Liver Care Center; medical director emeritus, liver transplantation; and professor, University of Cincinnati College of Medicine, department of pediatrics, Cincinnati Children’s Hospital Medical Center. He has served as director of the division of gastroenterology, hepatology, and nutrition at Cincinnati Children’s for 25 years and frequently covers gastroenterology, liver, and nutrition-related topics for this news organization. Dr Balistreri is currently editor-in-chief of the Journal of Pediatrics, having previously served as editor-in-chief of several journals and textbooks. He also became the first pediatrician to act as president of the American Association for the Study of Liver Diseases. In his spare time, he coaches youth lacrosse.
A version of this article first appeared on Medscape.com.
Children and COVID: New cases down to pre-Omicron level
New cases of COVID-19 in U.S. children dropped for the fifth consecutive week, but the rate of decline slowed considerably, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The national count of new cases has now fallen for five straight weeks since peaking Jan. 14-20, and this week’s figure is the lowest since the pre-Omicron days of mid-November, based on data collected by the AAP and CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.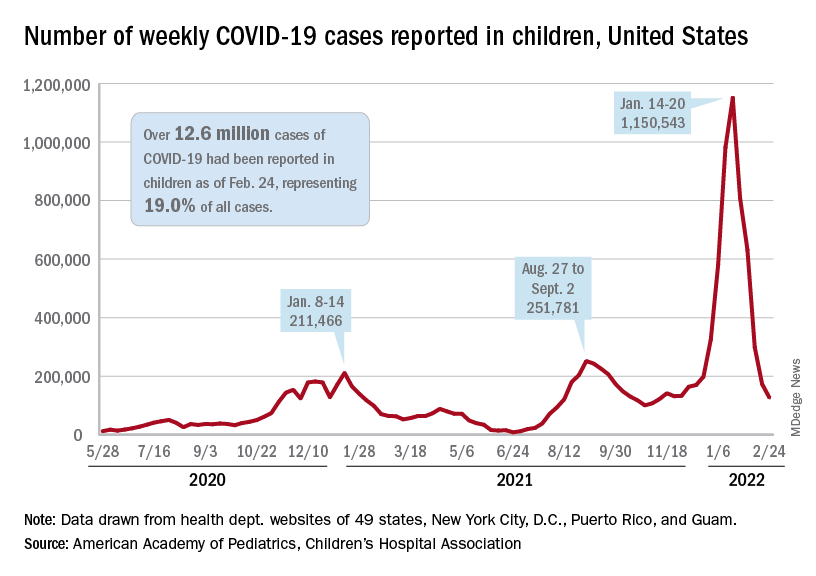
Over 12.6 million pediatric cases have been reported by those jurisdictions since the start of the pandemic, representing 19.0% of all cases in the United States, the AAP and CHA said in their weekly COVID report.
The highest cumulative rate among the states, 27.5%, can be found in Vermont, followed by New Hampshire (26.7%) and Alaska (26.6%). Alabama’s 12.1% is lower than any other jurisdiction, but the state stopped reporting during the summer of 2021, just as the Delta surge was beginning. The next two lowest states, Florida (12.8%) and Utah (13.9%), both define children as those aged 0-14 years, so the state with the lowest rate and no qualifiers is Idaho at 14.3%, the AAP/CHA data show.
The downward trend in new cases is reflected in other national measures. The daily rate of new hospital admissions for children aged 0-17 years was 0.32 per 100,000 population on Feb. 26, which is a drop of 75% since admissions peaked at 1.25 per 100,000 on Jan. 15, according to the Centers for Disease Control and Prevention.
The most recent 7-day average (Feb. 20-26) for child admissions with confirmed COVID-19 was 237 per day, compared with 914 per day during the peak week of Jan. 10-16. Emergency department visits with diagnosed COVID, measured as a percentage of all ED visits by age group, are down even more. The 7-day average was 1.2% on Feb. 25 for children aged 0-11 years, compared with a peak of 13.9% in mid-January, the CDC said on its COVID Data Tracker. The current rates for older children are even lower.
The decline of the Omicron surge over the last few weeks is allowing states to end mask mandates in schools around the country. The governors of California, Oregon, and Washington just announced that their states will be lifting their mask requirements on March 11, and New York State will end its mandate on March 2, while New York City is scheduled to go mask-free as of March 7, according to District Administration.
Those types of government moves, however, do not seem to be entirely supported by the public. In a survey conducted Feb. 9-21 by the Kaiser Family Foundation, 43% of the 1,502 respondents said that all students and staff should be required to wear masks in schools, while 40% said that there should be no mask requirements at all.
New cases of COVID-19 in U.S. children dropped for the fifth consecutive week, but the rate of decline slowed considerably, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The national count of new cases has now fallen for five straight weeks since peaking Jan. 14-20, and this week’s figure is the lowest since the pre-Omicron days of mid-November, based on data collected by the AAP and CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Over 12.6 million pediatric cases have been reported by those jurisdictions since the start of the pandemic, representing 19.0% of all cases in the United States, the AAP and CHA said in their weekly COVID report.
The highest cumulative rate among the states, 27.5%, can be found in Vermont, followed by New Hampshire (26.7%) and Alaska (26.6%). Alabama’s 12.1% is lower than any other jurisdiction, but the state stopped reporting during the summer of 2021, just as the Delta surge was beginning. The next two lowest states, Florida (12.8%) and Utah (13.9%), both define children as those aged 0-14 years, so the state with the lowest rate and no qualifiers is Idaho at 14.3%, the AAP/CHA data show.
The downward trend in new cases is reflected in other national measures. The daily rate of new hospital admissions for children aged 0-17 years was 0.32 per 100,000 population on Feb. 26, which is a drop of 75% since admissions peaked at 1.25 per 100,000 on Jan. 15, according to the Centers for Disease Control and Prevention.
The most recent 7-day average (Feb. 20-26) for child admissions with confirmed COVID-19 was 237 per day, compared with 914 per day during the peak week of Jan. 10-16. Emergency department visits with diagnosed COVID, measured as a percentage of all ED visits by age group, are down even more. The 7-day average was 1.2% on Feb. 25 for children aged 0-11 years, compared with a peak of 13.9% in mid-January, the CDC said on its COVID Data Tracker. The current rates for older children are even lower.
The decline of the Omicron surge over the last few weeks is allowing states to end mask mandates in schools around the country. The governors of California, Oregon, and Washington just announced that their states will be lifting their mask requirements on March 11, and New York State will end its mandate on March 2, while New York City is scheduled to go mask-free as of March 7, according to District Administration.
Those types of government moves, however, do not seem to be entirely supported by the public. In a survey conducted Feb. 9-21 by the Kaiser Family Foundation, 43% of the 1,502 respondents said that all students and staff should be required to wear masks in schools, while 40% said that there should be no mask requirements at all.
New cases of COVID-19 in U.S. children dropped for the fifth consecutive week, but the rate of decline slowed considerably, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The national count of new cases has now fallen for five straight weeks since peaking Jan. 14-20, and this week’s figure is the lowest since the pre-Omicron days of mid-November, based on data collected by the AAP and CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Over 12.6 million pediatric cases have been reported by those jurisdictions since the start of the pandemic, representing 19.0% of all cases in the United States, the AAP and CHA said in their weekly COVID report.
The highest cumulative rate among the states, 27.5%, can be found in Vermont, followed by New Hampshire (26.7%) and Alaska (26.6%). Alabama’s 12.1% is lower than any other jurisdiction, but the state stopped reporting during the summer of 2021, just as the Delta surge was beginning. The next two lowest states, Florida (12.8%) and Utah (13.9%), both define children as those aged 0-14 years, so the state with the lowest rate and no qualifiers is Idaho at 14.3%, the AAP/CHA data show.
The downward trend in new cases is reflected in other national measures. The daily rate of new hospital admissions for children aged 0-17 years was 0.32 per 100,000 population on Feb. 26, which is a drop of 75% since admissions peaked at 1.25 per 100,000 on Jan. 15, according to the Centers for Disease Control and Prevention.
The most recent 7-day average (Feb. 20-26) for child admissions with confirmed COVID-19 was 237 per day, compared with 914 per day during the peak week of Jan. 10-16. Emergency department visits with diagnosed COVID, measured as a percentage of all ED visits by age group, are down even more. The 7-day average was 1.2% on Feb. 25 for children aged 0-11 years, compared with a peak of 13.9% in mid-January, the CDC said on its COVID Data Tracker. The current rates for older children are even lower.
The decline of the Omicron surge over the last few weeks is allowing states to end mask mandates in schools around the country. The governors of California, Oregon, and Washington just announced that their states will be lifting their mask requirements on March 11, and New York State will end its mandate on March 2, while New York City is scheduled to go mask-free as of March 7, according to District Administration.
Those types of government moves, however, do not seem to be entirely supported by the public. In a survey conducted Feb. 9-21 by the Kaiser Family Foundation, 43% of the 1,502 respondents said that all students and staff should be required to wear masks in schools, while 40% said that there should be no mask requirements at all.
Elective surgery should be delayed 7 weeks after COVID-19 infection for unvaccinated patients, statement recommends
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
New studies indicate COVID pandemic began in Wuhan market
Two preprint studies released on Feb. 26 offer additional evidence that the coronavirus pandemic started at a market in Wuhan, China.
By analyzing data from several sources, scientists concluded that the virus came from animals and spread to humans in late 2019 at the Huanan Seafood Market. They added that no evidence supported a theory that the virus came from a laboratory in Wuhan.
“When you look at all the evidence together, it’s an extraordinarily clear picture that the pandemic started at the Huanan market,” Michael Worobey, D.Phil., a co-author on both studies and an evolutionary biologist at the University of Arizona, told the New York Times.
The two reports haven’t yet been peer-reviewed or published in a scientific journal. They were posted on Zenodo, an open-access research repository operated by CERN.
In one study, researchers used spatial analysis to show that the earliest COVID-19 cases, which were diagnosed in December 2019, were linked to the market. Researchers also found that environmental samples that tested positive for the SARS-CoV-2 virus were associated with animal vendors.
In another study, researchers found that two major viral lineages of the coronavirus resulted from at least two events when the virus spread from animals into humans. The first transmission most likely happened in late November or early December 2019, they wrote, and the other likely happened a few weeks later.
Several of the researchers behind the new studies also published a review last summer that said the pandemic originated in an animal, likely at a wildlife market. At that time, they said the first known case was a vendor at the Huanan market.
The new findings provide the strongest evidence yet that the pandemic had animal-related origins, Dr. Worobey told CNN. He called the results a “game, set and match” for the theory that the pandemic began in a lab.
“It’s no longer something that makes sense to imagine that this started any other way,” he said.
In a separate line of research, scientists at the Chinese CDC conducted a new analysis of samples collected at the market in January. They found that the samples included the two main lineages of the coronavirus. They posted the results in a report on the Research Square preprint server Feb. 26.
“The beauty of it is how simply it all adds up now,” Jeremy Kamil, a virologist at Louisiana State University Health Sciences, who wasn’t involved with the new studies, told the New York Times.
The initial spread of the coronavirus was like a firework, Dr. Worobey told CNN, starting at the market and exploding outward. The “overwhelming majority” of cases were specifically linked to the western section of the market, where most of the live-mammal vendors were located, the study authors wrote. Then COVID-19 cases spread into the community from there, and the pattern of transmission changed by January or February 2020.
When researchers tested surfaces at the market for coronavirus genetic material, one stall had the most positives, including a cage where raccoon dogs had been kept.
The study authors said the findings highlight the urgent need to pay attention to situations where wild animals and humans interact closely on a daily basis.
“We need to do a better job of farming and regulating these wild animals,” Robert Garry, one of the co-authors and a professor of microbiology and immunology at the Tulane University School of Medicine, told CNN.
That could include better infrastructure in places like markets where viruses spill over from animals to humans, he said. Surveillance is also key in preventing future pandemics by detecting new respiratory diseases in humans, isolating patients, and sequencing new virus strains.
“This is not the last time this happens,” he said.
A version of this article first appeared on WebMD.com.
Two preprint studies released on Feb. 26 offer additional evidence that the coronavirus pandemic started at a market in Wuhan, China.
By analyzing data from several sources, scientists concluded that the virus came from animals and spread to humans in late 2019 at the Huanan Seafood Market. They added that no evidence supported a theory that the virus came from a laboratory in Wuhan.
“When you look at all the evidence together, it’s an extraordinarily clear picture that the pandemic started at the Huanan market,” Michael Worobey, D.Phil., a co-author on both studies and an evolutionary biologist at the University of Arizona, told the New York Times.
The two reports haven’t yet been peer-reviewed or published in a scientific journal. They were posted on Zenodo, an open-access research repository operated by CERN.
In one study, researchers used spatial analysis to show that the earliest COVID-19 cases, which were diagnosed in December 2019, were linked to the market. Researchers also found that environmental samples that tested positive for the SARS-CoV-2 virus were associated with animal vendors.
In another study, researchers found that two major viral lineages of the coronavirus resulted from at least two events when the virus spread from animals into humans. The first transmission most likely happened in late November or early December 2019, they wrote, and the other likely happened a few weeks later.
Several of the researchers behind the new studies also published a review last summer that said the pandemic originated in an animal, likely at a wildlife market. At that time, they said the first known case was a vendor at the Huanan market.
The new findings provide the strongest evidence yet that the pandemic had animal-related origins, Dr. Worobey told CNN. He called the results a “game, set and match” for the theory that the pandemic began in a lab.
“It’s no longer something that makes sense to imagine that this started any other way,” he said.
In a separate line of research, scientists at the Chinese CDC conducted a new analysis of samples collected at the market in January. They found that the samples included the two main lineages of the coronavirus. They posted the results in a report on the Research Square preprint server Feb. 26.
“The beauty of it is how simply it all adds up now,” Jeremy Kamil, a virologist at Louisiana State University Health Sciences, who wasn’t involved with the new studies, told the New York Times.
The initial spread of the coronavirus was like a firework, Dr. Worobey told CNN, starting at the market and exploding outward. The “overwhelming majority” of cases were specifically linked to the western section of the market, where most of the live-mammal vendors were located, the study authors wrote. Then COVID-19 cases spread into the community from there, and the pattern of transmission changed by January or February 2020.
When researchers tested surfaces at the market for coronavirus genetic material, one stall had the most positives, including a cage where raccoon dogs had been kept.
The study authors said the findings highlight the urgent need to pay attention to situations where wild animals and humans interact closely on a daily basis.
“We need to do a better job of farming and regulating these wild animals,” Robert Garry, one of the co-authors and a professor of microbiology and immunology at the Tulane University School of Medicine, told CNN.
That could include better infrastructure in places like markets where viruses spill over from animals to humans, he said. Surveillance is also key in preventing future pandemics by detecting new respiratory diseases in humans, isolating patients, and sequencing new virus strains.
“This is not the last time this happens,” he said.
A version of this article first appeared on WebMD.com.
Two preprint studies released on Feb. 26 offer additional evidence that the coronavirus pandemic started at a market in Wuhan, China.
By analyzing data from several sources, scientists concluded that the virus came from animals and spread to humans in late 2019 at the Huanan Seafood Market. They added that no evidence supported a theory that the virus came from a laboratory in Wuhan.
“When you look at all the evidence together, it’s an extraordinarily clear picture that the pandemic started at the Huanan market,” Michael Worobey, D.Phil., a co-author on both studies and an evolutionary biologist at the University of Arizona, told the New York Times.
The two reports haven’t yet been peer-reviewed or published in a scientific journal. They were posted on Zenodo, an open-access research repository operated by CERN.
In one study, researchers used spatial analysis to show that the earliest COVID-19 cases, which were diagnosed in December 2019, were linked to the market. Researchers also found that environmental samples that tested positive for the SARS-CoV-2 virus were associated with animal vendors.
In another study, researchers found that two major viral lineages of the coronavirus resulted from at least two events when the virus spread from animals into humans. The first transmission most likely happened in late November or early December 2019, they wrote, and the other likely happened a few weeks later.
Several of the researchers behind the new studies also published a review last summer that said the pandemic originated in an animal, likely at a wildlife market. At that time, they said the first known case was a vendor at the Huanan market.
The new findings provide the strongest evidence yet that the pandemic had animal-related origins, Dr. Worobey told CNN. He called the results a “game, set and match” for the theory that the pandemic began in a lab.
“It’s no longer something that makes sense to imagine that this started any other way,” he said.
In a separate line of research, scientists at the Chinese CDC conducted a new analysis of samples collected at the market in January. They found that the samples included the two main lineages of the coronavirus. They posted the results in a report on the Research Square preprint server Feb. 26.
“The beauty of it is how simply it all adds up now,” Jeremy Kamil, a virologist at Louisiana State University Health Sciences, who wasn’t involved with the new studies, told the New York Times.
The initial spread of the coronavirus was like a firework, Dr. Worobey told CNN, starting at the market and exploding outward. The “overwhelming majority” of cases were specifically linked to the western section of the market, where most of the live-mammal vendors were located, the study authors wrote. Then COVID-19 cases spread into the community from there, and the pattern of transmission changed by January or February 2020.
When researchers tested surfaces at the market for coronavirus genetic material, one stall had the most positives, including a cage where raccoon dogs had been kept.
The study authors said the findings highlight the urgent need to pay attention to situations where wild animals and humans interact closely on a daily basis.
“We need to do a better job of farming and regulating these wild animals,” Robert Garry, one of the co-authors and a professor of microbiology and immunology at the Tulane University School of Medicine, told CNN.
That could include better infrastructure in places like markets where viruses spill over from animals to humans, he said. Surveillance is also key in preventing future pandemics by detecting new respiratory diseases in humans, isolating patients, and sequencing new virus strains.
“This is not the last time this happens,” he said.
A version of this article first appeared on WebMD.com.






