User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
New measles outbreaks reported in Los Angeles and El Paso
according to the Centers for Disease Control and Prevention.
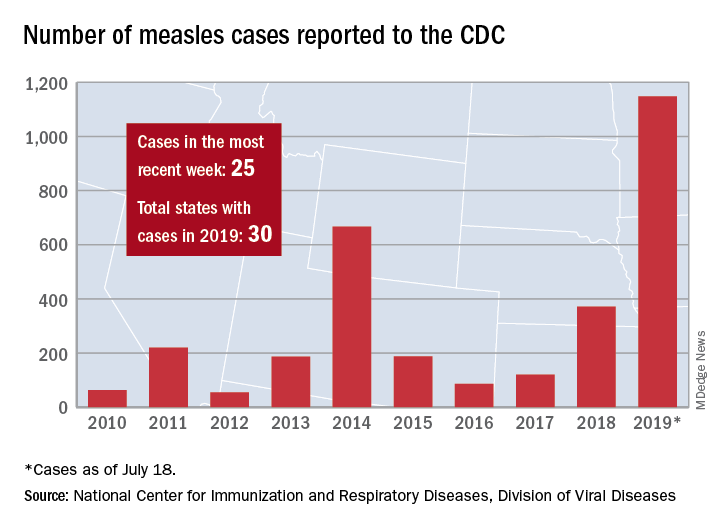
The total number of confirmed cases of measles in the United States is now up to 1,148 for the year, which is 25 more than the previous week, the CDC said on July 22. The highest 1-week total for the year was the 90 cases reported during the week of April 11.
The number of outbreaks is back up to five as California returned to the list after a 1-week absence and El Paso, Tex., made its first appearance of the year. The current outbreak in California – the state’s fifth – is occurring in Los Angeles, which is now up to 16 total cases in 2019. El Paso just reported its fourth case on July 17, and the city’s health department noted that “it had been more than 25 years since El Paso saw its last case of measles before these four recent cases.” Outbreaks also are ongoing in Rockland County, N.Y.; New York City; and three counties in Washington State.
States that joined the ranks of the measles-infected during this most recent reporting week were Alaska and Ohio, which brings the total number to 30 for the year, the CDC said.
The Alaska Department of Health and Social Services said that it “has confirmed a single case of measles in an unvaccinated teenager from the Kenai Peninsula who recently traveled out of state to Arizona via Seattle.” The Ohio case is a “young adult from Stark County [who] recently traveled to a state with confirmed measles cases,” according to the state’s health department.
according to the Centers for Disease Control and Prevention.

The total number of confirmed cases of measles in the United States is now up to 1,148 for the year, which is 25 more than the previous week, the CDC said on July 22. The highest 1-week total for the year was the 90 cases reported during the week of April 11.
The number of outbreaks is back up to five as California returned to the list after a 1-week absence and El Paso, Tex., made its first appearance of the year. The current outbreak in California – the state’s fifth – is occurring in Los Angeles, which is now up to 16 total cases in 2019. El Paso just reported its fourth case on July 17, and the city’s health department noted that “it had been more than 25 years since El Paso saw its last case of measles before these four recent cases.” Outbreaks also are ongoing in Rockland County, N.Y.; New York City; and three counties in Washington State.
States that joined the ranks of the measles-infected during this most recent reporting week were Alaska and Ohio, which brings the total number to 30 for the year, the CDC said.
The Alaska Department of Health and Social Services said that it “has confirmed a single case of measles in an unvaccinated teenager from the Kenai Peninsula who recently traveled out of state to Arizona via Seattle.” The Ohio case is a “young adult from Stark County [who] recently traveled to a state with confirmed measles cases,” according to the state’s health department.
according to the Centers for Disease Control and Prevention.

The total number of confirmed cases of measles in the United States is now up to 1,148 for the year, which is 25 more than the previous week, the CDC said on July 22. The highest 1-week total for the year was the 90 cases reported during the week of April 11.
The number of outbreaks is back up to five as California returned to the list after a 1-week absence and El Paso, Tex., made its first appearance of the year. The current outbreak in California – the state’s fifth – is occurring in Los Angeles, which is now up to 16 total cases in 2019. El Paso just reported its fourth case on July 17, and the city’s health department noted that “it had been more than 25 years since El Paso saw its last case of measles before these four recent cases.” Outbreaks also are ongoing in Rockland County, N.Y.; New York City; and three counties in Washington State.
States that joined the ranks of the measles-infected during this most recent reporting week were Alaska and Ohio, which brings the total number to 30 for the year, the CDC said.
The Alaska Department of Health and Social Services said that it “has confirmed a single case of measles in an unvaccinated teenager from the Kenai Peninsula who recently traveled out of state to Arizona via Seattle.” The Ohio case is a “young adult from Stark County [who] recently traveled to a state with confirmed measles cases,” according to the state’s health department.
Atherosclerotic disease risk persists decades after smoking cessation
Adults who quit smoking reduced their risk for peripheral artery disease in the short term, but remained at increased risk for up to 30 years, compared with never-smokers, based on data from more than 13,000 adults in a community-based study.

Most reports on the impact of smoking cessation on cardiovascular disease have focused on coronary heart disease (CHD), and stroke, while data on the effects of smoking cessation on peripheral artery disease (PAD) are limited, wrote Ning Ding, MBBS, SCM, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, Md., and colleagues.
To compare the impact of smoking on PAD, CHD, and stroke, the researchers used data from the Atherosclerosis Risk in Communities (ARIC) study, which included 15,792 adults aged 45-64 years in four communities. The findings were published in the Journal of the American College of Cardiology.
The study population of 13,355 individuals had no baseline history of PAD, CHD, or stroke. Over a median 26 years of follow-up, the researchers identified 492 cases of PAD, 1,798 cases of CHD, and 1,106 cases of stroke.
The risk of all three conditions began to decline within 5 years of smoking cessation, which could be encouraging to smokers who wish to quit, the researchers noted. In addition, the longer the duration of smoking cessation, the lower the risk for all three conditions (See central illustration).
However, a significantly elevated risk remained for PAD for up to 30 years after smoking cessation and for CHD for up to 20 years after smoking cessation, compared with never-smokers.
The researchers also found a roughly fourfold increased risk for PAD for smokers who smoked for 40 or more pack-years, compared with never-smokers, which was greater than the 2.1 hazard ratio for CHD and 1.8 HR for stroke. In addition, current smokers of at least one pack per day had a significantly greater risk of PAD, compared with never-smokers (HR, 5.36) that was higher than the risk for CHD or stroke (HR, 2.38 and HR, 1.88, respectively).
The study findings were limited by several factors including the reliance on self-reports, potential misclassification of data, and the potential exclusion of mild PAD cases that did not require hospitalization, the researchers noted. However, the results support the value of encouraging smokers to quit and support the need to include PAD risk in public health information, they said. “Although public statements about smoking and [cardiovascular disease] have been focusing on CHD and stroke, our results indicate the need to take account of PAD as well for comprehensively acknowledging the effect of smoking on overall cardiovascular health,” they added.
The ARIC study was funded by the National Heart, Lung, and Blood Institute, National Institutes of Health. Lead author Dr. Ding had no financial conflicts to disclose; coauthors disclosed relationships with Bristol-Myers Squibb and Fukuda Denshi.
SOURCE: Ding N et al. J Am Coll Cardiol. 2019 Jul 22;74:498-507. doi: 10.1016/j.jacc.2019.06.003.
Although the pathophysiology of smoking and cardiovascular disease has yet to be teased out, the current study findings support the public health message that any and all smokers can improve their health by quitting any time: “It is never too early or too late to benefit from quitting,” wrote Nancy A. Rigotti, MD, and Mary M. McDermott, MD, in an accompanying editorial. The editorialists questioned whether the findings were generalizable to patients with mild PAD or those who are not hospitalized. However, they found the data consistent with previous studies suggesting that atherosclerosis is not homogeneous. “Differences in shear stress and hemodynamic forces among the femoral, coronary, and carotid arterial beds may also explain variability in associations of smoking and smoking cessation with the incidence of PAD versus myocardial infarction or stroke,” they said.
The findings also support the need to emphasize PAD in public health messages and provide an opportunity to educate patients about the risks of limb loss and impaired mobility associated with PAD, they said.
Many clinicians put a low priority on smoking cessation, the editorialists wrote, but “long-term tobacco abstinence is achievable using a chronic disease management approach resembling the strategies used to manage other risk factors,” they said. They cited the American College of Cardiology’s recently released “Expert Consensus Decision Pathway on Tobacco Cessation Treatment.” The pathway outlines advice for clinicians, including how to provide a brief intervention and resources along with advice to quit smoking.
Dr. Rigotti is affiliated with Harvard Medical School, Boston. Dr. McDermott is affiliated with Northwestern University, Chicago. Dr. Rigotti disclosed royalties from UpToDate, serving as a consultant for Achieve Life Sciences, and travel expenses from Pfizer for unpaid consulting. Dr. McDermott disclosed research funding from Regeneron, the National Heart, Lung, and Blood Institute, the National Institute on Aging, and the American Heart Association, plus research support from Chromadex, ReserveAge, Hershey, and ViroMed.
Although the pathophysiology of smoking and cardiovascular disease has yet to be teased out, the current study findings support the public health message that any and all smokers can improve their health by quitting any time: “It is never too early or too late to benefit from quitting,” wrote Nancy A. Rigotti, MD, and Mary M. McDermott, MD, in an accompanying editorial. The editorialists questioned whether the findings were generalizable to patients with mild PAD or those who are not hospitalized. However, they found the data consistent with previous studies suggesting that atherosclerosis is not homogeneous. “Differences in shear stress and hemodynamic forces among the femoral, coronary, and carotid arterial beds may also explain variability in associations of smoking and smoking cessation with the incidence of PAD versus myocardial infarction or stroke,” they said.
The findings also support the need to emphasize PAD in public health messages and provide an opportunity to educate patients about the risks of limb loss and impaired mobility associated with PAD, they said.
Many clinicians put a low priority on smoking cessation, the editorialists wrote, but “long-term tobacco abstinence is achievable using a chronic disease management approach resembling the strategies used to manage other risk factors,” they said. They cited the American College of Cardiology’s recently released “Expert Consensus Decision Pathway on Tobacco Cessation Treatment.” The pathway outlines advice for clinicians, including how to provide a brief intervention and resources along with advice to quit smoking.
Dr. Rigotti is affiliated with Harvard Medical School, Boston. Dr. McDermott is affiliated with Northwestern University, Chicago. Dr. Rigotti disclosed royalties from UpToDate, serving as a consultant for Achieve Life Sciences, and travel expenses from Pfizer for unpaid consulting. Dr. McDermott disclosed research funding from Regeneron, the National Heart, Lung, and Blood Institute, the National Institute on Aging, and the American Heart Association, plus research support from Chromadex, ReserveAge, Hershey, and ViroMed.
Although the pathophysiology of smoking and cardiovascular disease has yet to be teased out, the current study findings support the public health message that any and all smokers can improve their health by quitting any time: “It is never too early or too late to benefit from quitting,” wrote Nancy A. Rigotti, MD, and Mary M. McDermott, MD, in an accompanying editorial. The editorialists questioned whether the findings were generalizable to patients with mild PAD or those who are not hospitalized. However, they found the data consistent with previous studies suggesting that atherosclerosis is not homogeneous. “Differences in shear stress and hemodynamic forces among the femoral, coronary, and carotid arterial beds may also explain variability in associations of smoking and smoking cessation with the incidence of PAD versus myocardial infarction or stroke,” they said.
The findings also support the need to emphasize PAD in public health messages and provide an opportunity to educate patients about the risks of limb loss and impaired mobility associated with PAD, they said.
Many clinicians put a low priority on smoking cessation, the editorialists wrote, but “long-term tobacco abstinence is achievable using a chronic disease management approach resembling the strategies used to manage other risk factors,” they said. They cited the American College of Cardiology’s recently released “Expert Consensus Decision Pathway on Tobacco Cessation Treatment.” The pathway outlines advice for clinicians, including how to provide a brief intervention and resources along with advice to quit smoking.
Dr. Rigotti is affiliated with Harvard Medical School, Boston. Dr. McDermott is affiliated with Northwestern University, Chicago. Dr. Rigotti disclosed royalties from UpToDate, serving as a consultant for Achieve Life Sciences, and travel expenses from Pfizer for unpaid consulting. Dr. McDermott disclosed research funding from Regeneron, the National Heart, Lung, and Blood Institute, the National Institute on Aging, and the American Heart Association, plus research support from Chromadex, ReserveAge, Hershey, and ViroMed.
Adults who quit smoking reduced their risk for peripheral artery disease in the short term, but remained at increased risk for up to 30 years, compared with never-smokers, based on data from more than 13,000 adults in a community-based study.

Most reports on the impact of smoking cessation on cardiovascular disease have focused on coronary heart disease (CHD), and stroke, while data on the effects of smoking cessation on peripheral artery disease (PAD) are limited, wrote Ning Ding, MBBS, SCM, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, Md., and colleagues.
To compare the impact of smoking on PAD, CHD, and stroke, the researchers used data from the Atherosclerosis Risk in Communities (ARIC) study, which included 15,792 adults aged 45-64 years in four communities. The findings were published in the Journal of the American College of Cardiology.
The study population of 13,355 individuals had no baseline history of PAD, CHD, or stroke. Over a median 26 years of follow-up, the researchers identified 492 cases of PAD, 1,798 cases of CHD, and 1,106 cases of stroke.
The risk of all three conditions began to decline within 5 years of smoking cessation, which could be encouraging to smokers who wish to quit, the researchers noted. In addition, the longer the duration of smoking cessation, the lower the risk for all three conditions (See central illustration).
However, a significantly elevated risk remained for PAD for up to 30 years after smoking cessation and for CHD for up to 20 years after smoking cessation, compared with never-smokers.
The researchers also found a roughly fourfold increased risk for PAD for smokers who smoked for 40 or more pack-years, compared with never-smokers, which was greater than the 2.1 hazard ratio for CHD and 1.8 HR for stroke. In addition, current smokers of at least one pack per day had a significantly greater risk of PAD, compared with never-smokers (HR, 5.36) that was higher than the risk for CHD or stroke (HR, 2.38 and HR, 1.88, respectively).
The study findings were limited by several factors including the reliance on self-reports, potential misclassification of data, and the potential exclusion of mild PAD cases that did not require hospitalization, the researchers noted. However, the results support the value of encouraging smokers to quit and support the need to include PAD risk in public health information, they said. “Although public statements about smoking and [cardiovascular disease] have been focusing on CHD and stroke, our results indicate the need to take account of PAD as well for comprehensively acknowledging the effect of smoking on overall cardiovascular health,” they added.
The ARIC study was funded by the National Heart, Lung, and Blood Institute, National Institutes of Health. Lead author Dr. Ding had no financial conflicts to disclose; coauthors disclosed relationships with Bristol-Myers Squibb and Fukuda Denshi.
SOURCE: Ding N et al. J Am Coll Cardiol. 2019 Jul 22;74:498-507. doi: 10.1016/j.jacc.2019.06.003.
Adults who quit smoking reduced their risk for peripheral artery disease in the short term, but remained at increased risk for up to 30 years, compared with never-smokers, based on data from more than 13,000 adults in a community-based study.

Most reports on the impact of smoking cessation on cardiovascular disease have focused on coronary heart disease (CHD), and stroke, while data on the effects of smoking cessation on peripheral artery disease (PAD) are limited, wrote Ning Ding, MBBS, SCM, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, Md., and colleagues.
To compare the impact of smoking on PAD, CHD, and stroke, the researchers used data from the Atherosclerosis Risk in Communities (ARIC) study, which included 15,792 adults aged 45-64 years in four communities. The findings were published in the Journal of the American College of Cardiology.
The study population of 13,355 individuals had no baseline history of PAD, CHD, or stroke. Over a median 26 years of follow-up, the researchers identified 492 cases of PAD, 1,798 cases of CHD, and 1,106 cases of stroke.
The risk of all three conditions began to decline within 5 years of smoking cessation, which could be encouraging to smokers who wish to quit, the researchers noted. In addition, the longer the duration of smoking cessation, the lower the risk for all three conditions (See central illustration).
However, a significantly elevated risk remained for PAD for up to 30 years after smoking cessation and for CHD for up to 20 years after smoking cessation, compared with never-smokers.
The researchers also found a roughly fourfold increased risk for PAD for smokers who smoked for 40 or more pack-years, compared with never-smokers, which was greater than the 2.1 hazard ratio for CHD and 1.8 HR for stroke. In addition, current smokers of at least one pack per day had a significantly greater risk of PAD, compared with never-smokers (HR, 5.36) that was higher than the risk for CHD or stroke (HR, 2.38 and HR, 1.88, respectively).
The study findings were limited by several factors including the reliance on self-reports, potential misclassification of data, and the potential exclusion of mild PAD cases that did not require hospitalization, the researchers noted. However, the results support the value of encouraging smokers to quit and support the need to include PAD risk in public health information, they said. “Although public statements about smoking and [cardiovascular disease] have been focusing on CHD and stroke, our results indicate the need to take account of PAD as well for comprehensively acknowledging the effect of smoking on overall cardiovascular health,” they added.
The ARIC study was funded by the National Heart, Lung, and Blood Institute, National Institutes of Health. Lead author Dr. Ding had no financial conflicts to disclose; coauthors disclosed relationships with Bristol-Myers Squibb and Fukuda Denshi.
SOURCE: Ding N et al. J Am Coll Cardiol. 2019 Jul 22;74:498-507. doi: 10.1016/j.jacc.2019.06.003.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Adjuvanted flu vaccine performs better than others in young children
according to an industry-funded synthesis of six studies.
The vaccine “offers significant advances over conventional inactivated influenza vaccines and presents an acceptable safety profile in children 6 months through 5 years of age,” Sanjay S. Patel, PhD, of Novartis Vaccines and Diagnostics, Cambridge, Mass., and associates wrote in the analysis, published in the International Journal of Infectious Diseases. “The noteworthy increases in antibody responses and decreases in influenza cases following vaccination suggest an alternative for use in a population that is heavily impacted by influenza disease.”
Children are, of course, vulnerable to flu. The Centers for Disease Control and Prevention reported that 186 children died of flu during the landmark 2017-2018 flu season. That’s the highest number of pediatric flu deaths since they became a notifiable condition in 2004 (exclusive of the 2009 pandemic, when 358 pediatric deaths were reported from April 15, 2009, to October 2, 2010).The CDC said the vaccine during that flu season had an overall effectiveness level of 40%. According to research of others, however, flu vaccines are less effective in younger children than in adolescents and adults (Vaccine. 2014;32[31]:3886-94; Cochrane Database Syst Rev. 2008. doi: 10.1002/14651858.CD004879.pub3).
Fluad – a MF59-adjuvanted inactivated trivalent seasonal influenza vaccine – is used in adults over 65 in the United States and 29 other countries, and it is approved for children aged 6 months through 23 months in Canada.
Dr. Patel and associates examined the results of six studies – one phase 1b, three phase 2, and two phase 3 – that tested Fluad with or without other vaccines in 11,942 children aged 6 months to 5 years. The studies, mostly multicenter, were conducted in various countries, mainly in Europe and South and Central America, from 2006 to 2012.
In general, children in the intervention groups in the studies received two doses of the Fluad vaccine 4 weeks apart: two 0.25-mL doses for children aged 6-35 months and two 0.5-mL doses for those aged 3 years or older. In most of the studies, parallel control groups received nonadjuvanted trivalent or quadrivalent influenza vaccines.
Most participants (93%-94%) completed the studies. Solicited adverse effects were common in all groups (72% in the Fluad group vs. 67% who received IIV3 vaccines), and generally mild to moderate and resolved in 1-3 days. Unsolicited adverse effects were similar (55% and 62%, respectively) in the two flu vaccine groups. The authors wrote that “these data reflect a safety profile consistent with other licensed inactivated influenza vaccines administered to children.”
As for results, Dr. Patel and colleagues said, “HI [hemagglutination inhibition] antibody responses to both homologous and heterologous influenza strains are higher following vaccination with aIIV3, and this increase in immunogenicity is observed across all age subgroups in children aged 6 months through 5 years, and most profound in the children 6 to 36 months.”
For example, in one of the phase 3 studies when the influenza viruses were antigenically matched (homologous) for A/H1N1 among the children aged 6-35 months seroconversion was 100% for allV3 (Fluad) and 38% for IIV3-1/IIV3-4 (trivalent/quadrivalent flu vaccines); among children aged 3-5 years seroconversion was 100% for allV3 and 82% for IIV3-1/IIV3-4. For AH3N2 homologous among children aged 6-35 months, seroconversion was 98% for allV3 and 44% for IIV3-1/IIV3-4. For the B strain homologous among children aged 6-35 months, seroconversion was 88% for allV3 and 19% for IIV3-1/IIV3-4; among children aged 3-5 years seroconversion for B was 99% for allV3 and 59% for IIV3-1/IIV3-4.
In the same study when the influenza viruses were antigenically mismatched (heterologous) for A/H1N1 among children of all ages 6 months to greater than 72 months, seroconversion was 96% for allV3 (Fluad) and 44% for IIV3-1/IIV3-4; for A/H3N2 it was 98% for allV3 and 49% for IIV3-1/IIV3-4, and for the B strain it was 10% for allV3 and 3% for IIV3-1/IIV3-4.
They added that “in addition, aIIV3 had the fastest onset of immunogenicity and longest persistence of immune response, which has implications for the real-world clinical setting, where the influenza season might start earlier than expected or last longer, and second (follow-up) vaccinations may be missed.”
Dr. Patel and associates said the MF59 adjuvant in Fluad “recruits immune cells (primarily monocytes, macrophages, neutrophils, and dendritic cells) at the site of injection and differentiates them into antigen-presenting cells. With an MF59-adjuvanted vaccine, more antigen is transported from the injection site to the draining lymph node, wherein MF59 leads to T-cell activation and an increased B-cell expansion and a greater number and diversity of antibodies.”
According to goodrx.com, one syringe of Fluad 0.5 mL costs $45-$74 with coupon. The same dose of Fluzone Quadrivalent, a flu vaccine recently approved by the Food and Drug Administration for use in young children aged 6-35 months, costs $31 with coupon.
The study was funded by Novartis Vaccines and Diagnostics and Seqirus (formerly part of Novartis Vaccines and Diagnostics). The study authors disclosed employment by Novartis and Seqirus.
SOURCE: Patel SS et al. Int J Infect Dis. 2019. doi: 10.1016/j.ijid.2019.05.009.
according to an industry-funded synthesis of six studies.
The vaccine “offers significant advances over conventional inactivated influenza vaccines and presents an acceptable safety profile in children 6 months through 5 years of age,” Sanjay S. Patel, PhD, of Novartis Vaccines and Diagnostics, Cambridge, Mass., and associates wrote in the analysis, published in the International Journal of Infectious Diseases. “The noteworthy increases in antibody responses and decreases in influenza cases following vaccination suggest an alternative for use in a population that is heavily impacted by influenza disease.”
Children are, of course, vulnerable to flu. The Centers for Disease Control and Prevention reported that 186 children died of flu during the landmark 2017-2018 flu season. That’s the highest number of pediatric flu deaths since they became a notifiable condition in 2004 (exclusive of the 2009 pandemic, when 358 pediatric deaths were reported from April 15, 2009, to October 2, 2010).The CDC said the vaccine during that flu season had an overall effectiveness level of 40%. According to research of others, however, flu vaccines are less effective in younger children than in adolescents and adults (Vaccine. 2014;32[31]:3886-94; Cochrane Database Syst Rev. 2008. doi: 10.1002/14651858.CD004879.pub3).
Fluad – a MF59-adjuvanted inactivated trivalent seasonal influenza vaccine – is used in adults over 65 in the United States and 29 other countries, and it is approved for children aged 6 months through 23 months in Canada.
Dr. Patel and associates examined the results of six studies – one phase 1b, three phase 2, and two phase 3 – that tested Fluad with or without other vaccines in 11,942 children aged 6 months to 5 years. The studies, mostly multicenter, were conducted in various countries, mainly in Europe and South and Central America, from 2006 to 2012.
In general, children in the intervention groups in the studies received two doses of the Fluad vaccine 4 weeks apart: two 0.25-mL doses for children aged 6-35 months and two 0.5-mL doses for those aged 3 years or older. In most of the studies, parallel control groups received nonadjuvanted trivalent or quadrivalent influenza vaccines.
Most participants (93%-94%) completed the studies. Solicited adverse effects were common in all groups (72% in the Fluad group vs. 67% who received IIV3 vaccines), and generally mild to moderate and resolved in 1-3 days. Unsolicited adverse effects were similar (55% and 62%, respectively) in the two flu vaccine groups. The authors wrote that “these data reflect a safety profile consistent with other licensed inactivated influenza vaccines administered to children.”
As for results, Dr. Patel and colleagues said, “HI [hemagglutination inhibition] antibody responses to both homologous and heterologous influenza strains are higher following vaccination with aIIV3, and this increase in immunogenicity is observed across all age subgroups in children aged 6 months through 5 years, and most profound in the children 6 to 36 months.”
For example, in one of the phase 3 studies when the influenza viruses were antigenically matched (homologous) for A/H1N1 among the children aged 6-35 months seroconversion was 100% for allV3 (Fluad) and 38% for IIV3-1/IIV3-4 (trivalent/quadrivalent flu vaccines); among children aged 3-5 years seroconversion was 100% for allV3 and 82% for IIV3-1/IIV3-4. For AH3N2 homologous among children aged 6-35 months, seroconversion was 98% for allV3 and 44% for IIV3-1/IIV3-4. For the B strain homologous among children aged 6-35 months, seroconversion was 88% for allV3 and 19% for IIV3-1/IIV3-4; among children aged 3-5 years seroconversion for B was 99% for allV3 and 59% for IIV3-1/IIV3-4.
In the same study when the influenza viruses were antigenically mismatched (heterologous) for A/H1N1 among children of all ages 6 months to greater than 72 months, seroconversion was 96% for allV3 (Fluad) and 44% for IIV3-1/IIV3-4; for A/H3N2 it was 98% for allV3 and 49% for IIV3-1/IIV3-4, and for the B strain it was 10% for allV3 and 3% for IIV3-1/IIV3-4.
They added that “in addition, aIIV3 had the fastest onset of immunogenicity and longest persistence of immune response, which has implications for the real-world clinical setting, where the influenza season might start earlier than expected or last longer, and second (follow-up) vaccinations may be missed.”
Dr. Patel and associates said the MF59 adjuvant in Fluad “recruits immune cells (primarily monocytes, macrophages, neutrophils, and dendritic cells) at the site of injection and differentiates them into antigen-presenting cells. With an MF59-adjuvanted vaccine, more antigen is transported from the injection site to the draining lymph node, wherein MF59 leads to T-cell activation and an increased B-cell expansion and a greater number and diversity of antibodies.”
According to goodrx.com, one syringe of Fluad 0.5 mL costs $45-$74 with coupon. The same dose of Fluzone Quadrivalent, a flu vaccine recently approved by the Food and Drug Administration for use in young children aged 6-35 months, costs $31 with coupon.
The study was funded by Novartis Vaccines and Diagnostics and Seqirus (formerly part of Novartis Vaccines and Diagnostics). The study authors disclosed employment by Novartis and Seqirus.
SOURCE: Patel SS et al. Int J Infect Dis. 2019. doi: 10.1016/j.ijid.2019.05.009.
according to an industry-funded synthesis of six studies.
The vaccine “offers significant advances over conventional inactivated influenza vaccines and presents an acceptable safety profile in children 6 months through 5 years of age,” Sanjay S. Patel, PhD, of Novartis Vaccines and Diagnostics, Cambridge, Mass., and associates wrote in the analysis, published in the International Journal of Infectious Diseases. “The noteworthy increases in antibody responses and decreases in influenza cases following vaccination suggest an alternative for use in a population that is heavily impacted by influenza disease.”
Children are, of course, vulnerable to flu. The Centers for Disease Control and Prevention reported that 186 children died of flu during the landmark 2017-2018 flu season. That’s the highest number of pediatric flu deaths since they became a notifiable condition in 2004 (exclusive of the 2009 pandemic, when 358 pediatric deaths were reported from April 15, 2009, to October 2, 2010).The CDC said the vaccine during that flu season had an overall effectiveness level of 40%. According to research of others, however, flu vaccines are less effective in younger children than in adolescents and adults (Vaccine. 2014;32[31]:3886-94; Cochrane Database Syst Rev. 2008. doi: 10.1002/14651858.CD004879.pub3).
Fluad – a MF59-adjuvanted inactivated trivalent seasonal influenza vaccine – is used in adults over 65 in the United States and 29 other countries, and it is approved for children aged 6 months through 23 months in Canada.
Dr. Patel and associates examined the results of six studies – one phase 1b, three phase 2, and two phase 3 – that tested Fluad with or without other vaccines in 11,942 children aged 6 months to 5 years. The studies, mostly multicenter, were conducted in various countries, mainly in Europe and South and Central America, from 2006 to 2012.
In general, children in the intervention groups in the studies received two doses of the Fluad vaccine 4 weeks apart: two 0.25-mL doses for children aged 6-35 months and two 0.5-mL doses for those aged 3 years or older. In most of the studies, parallel control groups received nonadjuvanted trivalent or quadrivalent influenza vaccines.
Most participants (93%-94%) completed the studies. Solicited adverse effects were common in all groups (72% in the Fluad group vs. 67% who received IIV3 vaccines), and generally mild to moderate and resolved in 1-3 days. Unsolicited adverse effects were similar (55% and 62%, respectively) in the two flu vaccine groups. The authors wrote that “these data reflect a safety profile consistent with other licensed inactivated influenza vaccines administered to children.”
As for results, Dr. Patel and colleagues said, “HI [hemagglutination inhibition] antibody responses to both homologous and heterologous influenza strains are higher following vaccination with aIIV3, and this increase in immunogenicity is observed across all age subgroups in children aged 6 months through 5 years, and most profound in the children 6 to 36 months.”
For example, in one of the phase 3 studies when the influenza viruses were antigenically matched (homologous) for A/H1N1 among the children aged 6-35 months seroconversion was 100% for allV3 (Fluad) and 38% for IIV3-1/IIV3-4 (trivalent/quadrivalent flu vaccines); among children aged 3-5 years seroconversion was 100% for allV3 and 82% for IIV3-1/IIV3-4. For AH3N2 homologous among children aged 6-35 months, seroconversion was 98% for allV3 and 44% for IIV3-1/IIV3-4. For the B strain homologous among children aged 6-35 months, seroconversion was 88% for allV3 and 19% for IIV3-1/IIV3-4; among children aged 3-5 years seroconversion for B was 99% for allV3 and 59% for IIV3-1/IIV3-4.
In the same study when the influenza viruses were antigenically mismatched (heterologous) for A/H1N1 among children of all ages 6 months to greater than 72 months, seroconversion was 96% for allV3 (Fluad) and 44% for IIV3-1/IIV3-4; for A/H3N2 it was 98% for allV3 and 49% for IIV3-1/IIV3-4, and for the B strain it was 10% for allV3 and 3% for IIV3-1/IIV3-4.
They added that “in addition, aIIV3 had the fastest onset of immunogenicity and longest persistence of immune response, which has implications for the real-world clinical setting, where the influenza season might start earlier than expected or last longer, and second (follow-up) vaccinations may be missed.”
Dr. Patel and associates said the MF59 adjuvant in Fluad “recruits immune cells (primarily monocytes, macrophages, neutrophils, and dendritic cells) at the site of injection and differentiates them into antigen-presenting cells. With an MF59-adjuvanted vaccine, more antigen is transported from the injection site to the draining lymph node, wherein MF59 leads to T-cell activation and an increased B-cell expansion and a greater number and diversity of antibodies.”
According to goodrx.com, one syringe of Fluad 0.5 mL costs $45-$74 with coupon. The same dose of Fluzone Quadrivalent, a flu vaccine recently approved by the Food and Drug Administration for use in young children aged 6-35 months, costs $31 with coupon.
The study was funded by Novartis Vaccines and Diagnostics and Seqirus (formerly part of Novartis Vaccines and Diagnostics). The study authors disclosed employment by Novartis and Seqirus.
SOURCE: Patel SS et al. Int J Infect Dis. 2019. doi: 10.1016/j.ijid.2019.05.009.
FROM INTERNATIONAL JOURNAL OF INFECTIOUS DISEASES
Adjuvanted influenza vaccine appears safe for at-risk children
according to a study in the International Journal of Infectious Diseases.
Sanjay S. Patel, PhD, of Novartis Vaccines and Diagnostics, Cambridge, Mass., and colleagues performed a retrospective analysis on an integrated dataset that drew from six randomized clinical trials comparing aIIV3 with nonadjuvanted trivalent inactivated influenza vaccine (IIV3). The dataset comprised 10,794 patients aged 6 months through 5 years, of whom 373 (3%) were deemed at risk of influenza complications after review of their medical history for conditions such as heart disease, asthma, and endocrine disorders.
The rates of solicited adverse events (such as erythema, diarrhea, fever, and localized swelling) were 74% in the aIIV3 group and 73% in the IIV3 group. The rates for any unsolicited adverse events (such as upper respiratory tract infection) for aIIV3 and IIV3 were 54% and 59%, respectively (Int J Infect Dis. 2019. doi: 10.1016/j.ijid.2019.04.023).
One of the six studies included in the dataset randomized 2,655 children for immunogenicity analyses, of whom 103 (4%) were deemed at risk. Hemagglutination inhibition assay geometric mean titers against homologous A/H1N1, A/H3N2, and B strains 21 days after the second of two doses of vaccines were two to three times higher in the aIIV3 than in the IIV3 group, which suggests that aIIV3 is more immunogenic than IIV3. As the investigators noted, this is likely because the adjuvanted vaccine induces a greater magnitude of immune response to the vaccine, something already lower in children than in adults, as well as more breadth of response, meaning the response goes beyond strains included in the vaccines.
The small number of at-risk children in the study poses a limitation on its findings. Dr. Patel and associates said that, regardless, the results of immunogenicity analyses were strong. “Overall, this analysis indicates that aIIV3 has a similar safety profile in young children with underlying medical conditions, consistent with other licensed inactivated influenza vaccines.”
Novartis Vaccines and Diagnostics originally funded the study, but was later acquired by CSL Group and now operates as Seqirus, which continued funding for the study. The authors were employees of one or the other of these companies.
according to a study in the International Journal of Infectious Diseases.
Sanjay S. Patel, PhD, of Novartis Vaccines and Diagnostics, Cambridge, Mass., and colleagues performed a retrospective analysis on an integrated dataset that drew from six randomized clinical trials comparing aIIV3 with nonadjuvanted trivalent inactivated influenza vaccine (IIV3). The dataset comprised 10,794 patients aged 6 months through 5 years, of whom 373 (3%) were deemed at risk of influenza complications after review of their medical history for conditions such as heart disease, asthma, and endocrine disorders.
The rates of solicited adverse events (such as erythema, diarrhea, fever, and localized swelling) were 74% in the aIIV3 group and 73% in the IIV3 group. The rates for any unsolicited adverse events (such as upper respiratory tract infection) for aIIV3 and IIV3 were 54% and 59%, respectively (Int J Infect Dis. 2019. doi: 10.1016/j.ijid.2019.04.023).
One of the six studies included in the dataset randomized 2,655 children for immunogenicity analyses, of whom 103 (4%) were deemed at risk. Hemagglutination inhibition assay geometric mean titers against homologous A/H1N1, A/H3N2, and B strains 21 days after the second of two doses of vaccines were two to three times higher in the aIIV3 than in the IIV3 group, which suggests that aIIV3 is more immunogenic than IIV3. As the investigators noted, this is likely because the adjuvanted vaccine induces a greater magnitude of immune response to the vaccine, something already lower in children than in adults, as well as more breadth of response, meaning the response goes beyond strains included in the vaccines.
The small number of at-risk children in the study poses a limitation on its findings. Dr. Patel and associates said that, regardless, the results of immunogenicity analyses were strong. “Overall, this analysis indicates that aIIV3 has a similar safety profile in young children with underlying medical conditions, consistent with other licensed inactivated influenza vaccines.”
Novartis Vaccines and Diagnostics originally funded the study, but was later acquired by CSL Group and now operates as Seqirus, which continued funding for the study. The authors were employees of one or the other of these companies.
according to a study in the International Journal of Infectious Diseases.
Sanjay S. Patel, PhD, of Novartis Vaccines and Diagnostics, Cambridge, Mass., and colleagues performed a retrospective analysis on an integrated dataset that drew from six randomized clinical trials comparing aIIV3 with nonadjuvanted trivalent inactivated influenza vaccine (IIV3). The dataset comprised 10,794 patients aged 6 months through 5 years, of whom 373 (3%) were deemed at risk of influenza complications after review of their medical history for conditions such as heart disease, asthma, and endocrine disorders.
The rates of solicited adverse events (such as erythema, diarrhea, fever, and localized swelling) were 74% in the aIIV3 group and 73% in the IIV3 group. The rates for any unsolicited adverse events (such as upper respiratory tract infection) for aIIV3 and IIV3 were 54% and 59%, respectively (Int J Infect Dis. 2019. doi: 10.1016/j.ijid.2019.04.023).
One of the six studies included in the dataset randomized 2,655 children for immunogenicity analyses, of whom 103 (4%) were deemed at risk. Hemagglutination inhibition assay geometric mean titers against homologous A/H1N1, A/H3N2, and B strains 21 days after the second of two doses of vaccines were two to three times higher in the aIIV3 than in the IIV3 group, which suggests that aIIV3 is more immunogenic than IIV3. As the investigators noted, this is likely because the adjuvanted vaccine induces a greater magnitude of immune response to the vaccine, something already lower in children than in adults, as well as more breadth of response, meaning the response goes beyond strains included in the vaccines.
The small number of at-risk children in the study poses a limitation on its findings. Dr. Patel and associates said that, regardless, the results of immunogenicity analyses were strong. “Overall, this analysis indicates that aIIV3 has a similar safety profile in young children with underlying medical conditions, consistent with other licensed inactivated influenza vaccines.”
Novartis Vaccines and Diagnostics originally funded the study, but was later acquired by CSL Group and now operates as Seqirus, which continued funding for the study. The authors were employees of one or the other of these companies.
FROM THE INTERNATIONAL JOURNAL OF INFECTIOUS DISEASES
Smoking-cessation attempts changed little over 7-year span
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
FROM MMWR
New analysis challenges fluid resuscitation guidelines for patients in shock
Although guideline recommended, according to a detailed analysis of available data, including a randomized trial.
Several sets of guidelines for resuscitation of patients in shock have advocated volume expansion with bolus intravenous fluid, but that recommendation was based on expected physiologic benefits not a randomized trial. The only randomized trial associated this approach showed increased mortality, and a new analysis of these and other data appears to explain why.
According to the findings of a study lead by Michael Levin, MD, of the department of medicine at Imperial College London and colleagues, “volume resuscitation is associated with deterioration of respiratory function and neurological function in some patients.” Their study was published in Lancet Respiratory Medicine. The authors stated that saline-induced hyperchloremic acidosis appears to have been “a major contributor” to the observed increase in adverse outcomes.
The key take home message is that “normal saline and other unbuffered crystalloid solutions should be avoided in resuscitating seriously ill patients,” according to the authors, who believe the findings might be relevant to adults as well as children.
The controversy about the role of volume expansion for management of shock was ignited by a 2011 trial called FEAST (N Engl J Med. 2011;364:2483-95). That trial, which randomized African children with severe febrile illness to a bolus of 20-40 mg of 5% albumin solution, a bolus of 0.9% saline solution, or no bolus, was halted early when 48-hour mortality data showed a lower death rate in the no bolus group (7.3%) than either the albumin (10.6%) or saline (10.5%) bolus groups.
The FEAST result was unexpected and so contrary to accepted thinking that it prompted widespread debate, including whether findings in the resource-poor area of the world where the FEAST trial was conducted could be extrapolated to centers elsewhere in the world. Arguing for benefit, fluid resuscitation is known to increase pulse pressure and urinary output. Arguing against benefit, pulmonary edema is a known complication of bolus fluid replacement.
In an attempt to address and potentially resolve this controversy, data collected in the FEAST trial along with four other sets of data involving volume expansion in critically ill children were evaluated with a focus on changes in cardiovascular, neurological, and respiratory function. Analysis of blood biochemistry and blood oxygen transport were also conducted.
The cardiovascular, respiratory, and neurologic functions were scored on the basis of objective measurements, such as heart rate, respiratory rate, and blood pressure. These measures were evaluated prior to fluid administration and at 1 hour, 4 hours, 8 hours, 24 hours, and 48 hours after fluid administration. Odds ratio (OR) of an adverse outcome were evaluated in the context of each 10-unit change in these scores.
Relative to baseline, there was worsening respiratory and neurological function after fluid administration. Although cardiovascular function improved, hemoglobin concentrations were lower in those who received fluid than in those who did not. Fluid resuscitation was also associated with lower bicarbonate and increased base deficit and chloride at 24 hours.
Regression modeling with physiological variables suggests “that the increased mortality in FEAST can be explained by bolus-induced worsening in respiratory and neurological function, hemodilution, and hyperchloremic acidosis,” according to the authors.
Analyses of the four other sets of data, which included children treated for meningococcal sepsis in the United Kingdom, acutely ill with malaria treated in Malawi, and cohorts of children in South Africa and a London hospital for acute illnesses, provided supportive data.
Although this analysis does not address the value of administering buffered solutions in low volumes, the authors concluded that the data from the FEAST trial are generalizable. They challenge the routine use of bolus infusions of saline or albumin in the initial management of shock, which has been guideline recommended. The risks of fluid resuscitation might be particularly high among children who already have compromised respiratory or neurologic function.
SOURCE: Levin M et al. Lancet Respir Med. 2019;7:581-93.
Although guideline recommended, according to a detailed analysis of available data, including a randomized trial.
Several sets of guidelines for resuscitation of patients in shock have advocated volume expansion with bolus intravenous fluid, but that recommendation was based on expected physiologic benefits not a randomized trial. The only randomized trial associated this approach showed increased mortality, and a new analysis of these and other data appears to explain why.
According to the findings of a study lead by Michael Levin, MD, of the department of medicine at Imperial College London and colleagues, “volume resuscitation is associated with deterioration of respiratory function and neurological function in some patients.” Their study was published in Lancet Respiratory Medicine. The authors stated that saline-induced hyperchloremic acidosis appears to have been “a major contributor” to the observed increase in adverse outcomes.
The key take home message is that “normal saline and other unbuffered crystalloid solutions should be avoided in resuscitating seriously ill patients,” according to the authors, who believe the findings might be relevant to adults as well as children.
The controversy about the role of volume expansion for management of shock was ignited by a 2011 trial called FEAST (N Engl J Med. 2011;364:2483-95). That trial, which randomized African children with severe febrile illness to a bolus of 20-40 mg of 5% albumin solution, a bolus of 0.9% saline solution, or no bolus, was halted early when 48-hour mortality data showed a lower death rate in the no bolus group (7.3%) than either the albumin (10.6%) or saline (10.5%) bolus groups.
The FEAST result was unexpected and so contrary to accepted thinking that it prompted widespread debate, including whether findings in the resource-poor area of the world where the FEAST trial was conducted could be extrapolated to centers elsewhere in the world. Arguing for benefit, fluid resuscitation is known to increase pulse pressure and urinary output. Arguing against benefit, pulmonary edema is a known complication of bolus fluid replacement.
In an attempt to address and potentially resolve this controversy, data collected in the FEAST trial along with four other sets of data involving volume expansion in critically ill children were evaluated with a focus on changes in cardiovascular, neurological, and respiratory function. Analysis of blood biochemistry and blood oxygen transport were also conducted.
The cardiovascular, respiratory, and neurologic functions were scored on the basis of objective measurements, such as heart rate, respiratory rate, and blood pressure. These measures were evaluated prior to fluid administration and at 1 hour, 4 hours, 8 hours, 24 hours, and 48 hours after fluid administration. Odds ratio (OR) of an adverse outcome were evaluated in the context of each 10-unit change in these scores.
Relative to baseline, there was worsening respiratory and neurological function after fluid administration. Although cardiovascular function improved, hemoglobin concentrations were lower in those who received fluid than in those who did not. Fluid resuscitation was also associated with lower bicarbonate and increased base deficit and chloride at 24 hours.
Regression modeling with physiological variables suggests “that the increased mortality in FEAST can be explained by bolus-induced worsening in respiratory and neurological function, hemodilution, and hyperchloremic acidosis,” according to the authors.
Analyses of the four other sets of data, which included children treated for meningococcal sepsis in the United Kingdom, acutely ill with malaria treated in Malawi, and cohorts of children in South Africa and a London hospital for acute illnesses, provided supportive data.
Although this analysis does not address the value of administering buffered solutions in low volumes, the authors concluded that the data from the FEAST trial are generalizable. They challenge the routine use of bolus infusions of saline or albumin in the initial management of shock, which has been guideline recommended. The risks of fluid resuscitation might be particularly high among children who already have compromised respiratory or neurologic function.
SOURCE: Levin M et al. Lancet Respir Med. 2019;7:581-93.
Although guideline recommended, according to a detailed analysis of available data, including a randomized trial.
Several sets of guidelines for resuscitation of patients in shock have advocated volume expansion with bolus intravenous fluid, but that recommendation was based on expected physiologic benefits not a randomized trial. The only randomized trial associated this approach showed increased mortality, and a new analysis of these and other data appears to explain why.
According to the findings of a study lead by Michael Levin, MD, of the department of medicine at Imperial College London and colleagues, “volume resuscitation is associated with deterioration of respiratory function and neurological function in some patients.” Their study was published in Lancet Respiratory Medicine. The authors stated that saline-induced hyperchloremic acidosis appears to have been “a major contributor” to the observed increase in adverse outcomes.
The key take home message is that “normal saline and other unbuffered crystalloid solutions should be avoided in resuscitating seriously ill patients,” according to the authors, who believe the findings might be relevant to adults as well as children.
The controversy about the role of volume expansion for management of shock was ignited by a 2011 trial called FEAST (N Engl J Med. 2011;364:2483-95). That trial, which randomized African children with severe febrile illness to a bolus of 20-40 mg of 5% albumin solution, a bolus of 0.9% saline solution, or no bolus, was halted early when 48-hour mortality data showed a lower death rate in the no bolus group (7.3%) than either the albumin (10.6%) or saline (10.5%) bolus groups.
The FEAST result was unexpected and so contrary to accepted thinking that it prompted widespread debate, including whether findings in the resource-poor area of the world where the FEAST trial was conducted could be extrapolated to centers elsewhere in the world. Arguing for benefit, fluid resuscitation is known to increase pulse pressure and urinary output. Arguing against benefit, pulmonary edema is a known complication of bolus fluid replacement.
In an attempt to address and potentially resolve this controversy, data collected in the FEAST trial along with four other sets of data involving volume expansion in critically ill children were evaluated with a focus on changes in cardiovascular, neurological, and respiratory function. Analysis of blood biochemistry and blood oxygen transport were also conducted.
The cardiovascular, respiratory, and neurologic functions were scored on the basis of objective measurements, such as heart rate, respiratory rate, and blood pressure. These measures were evaluated prior to fluid administration and at 1 hour, 4 hours, 8 hours, 24 hours, and 48 hours after fluid administration. Odds ratio (OR) of an adverse outcome were evaluated in the context of each 10-unit change in these scores.
Relative to baseline, there was worsening respiratory and neurological function after fluid administration. Although cardiovascular function improved, hemoglobin concentrations were lower in those who received fluid than in those who did not. Fluid resuscitation was also associated with lower bicarbonate and increased base deficit and chloride at 24 hours.
Regression modeling with physiological variables suggests “that the increased mortality in FEAST can be explained by bolus-induced worsening in respiratory and neurological function, hemodilution, and hyperchloremic acidosis,” according to the authors.
Analyses of the four other sets of data, which included children treated for meningococcal sepsis in the United Kingdom, acutely ill with malaria treated in Malawi, and cohorts of children in South Africa and a London hospital for acute illnesses, provided supportive data.
Although this analysis does not address the value of administering buffered solutions in low volumes, the authors concluded that the data from the FEAST trial are generalizable. They challenge the routine use of bolus infusions of saline or albumin in the initial management of shock, which has been guideline recommended. The risks of fluid resuscitation might be particularly high among children who already have compromised respiratory or neurologic function.
SOURCE: Levin M et al. Lancet Respir Med. 2019;7:581-93.
FROM THE LANCET RESPIRATORY MEDICINE
Little association found between in utero H1N1 vaccine and 5-year health outcomes
according to Laura K. Walsh of the University of Ottawa and associates.
The investigators conducted a population-based retrospective cohort study from November 2009 to October 2010 of all live births within the province of Ontario. Of the 104,249 eligible live births reported to the Ontario birth registry, 31,295 were exposed to the H1N1 vaccine in utero. After adjustment, there were no significant differences in the women who did and did not receive vaccines during pregnancy, according to the study, published in the BMJ.
After a median follow-up of 5 years, 14% of children received an asthma diagnosis, with a median age at diagnosis of 1.8 years. Children were more likely to receive an asthma diagnosis if their mothers had a preexisting condition or if they were born preterm. At follow-up, 34% of children had at least one upper respiratory tract infection. Sensory disorder, neoplasm, and pediatric complex chronic condition were rare, each occurring in less than 1% of the study cohort (BMJ. 2019 Jul 10. doi: 10.1136/bmj.l4151).
No significant association was found between prenatal exposure to the H1N1 vaccine and upper or lower respiratory infections, otitis media, all infections, neoplasms, sensory disorders, rates of urgent and inpatient health services use, pediatric complex chronic conditions, or mortality. A weak but significant association was observed for asthma (adjusted hazard ratio, 1.05; 95% confidence interval, 1.02-1.09), and a weak inverse association was found for gastrointestinal infections (adjusted incidence rate ratio, 0.94; 95% CI, 0.91-0.98).
“Although we observed a small, but statistically significant, increase in pediatric asthma and a reduction in gastrointestinal infections, we are not aware of any biologic mechanisms to explain these findings. Future studies in different settings and with different influenza vaccine formulations will be important for developing the evidence base on longer-term pediatric outcomes following influenza vaccination during pregnancy,” the investigators concluded.
The study was funded by grants from the Canadian Institutes of Health Research and the Institute for Clinical Evaluative Sciences.
according to Laura K. Walsh of the University of Ottawa and associates.
The investigators conducted a population-based retrospective cohort study from November 2009 to October 2010 of all live births within the province of Ontario. Of the 104,249 eligible live births reported to the Ontario birth registry, 31,295 were exposed to the H1N1 vaccine in utero. After adjustment, there were no significant differences in the women who did and did not receive vaccines during pregnancy, according to the study, published in the BMJ.
After a median follow-up of 5 years, 14% of children received an asthma diagnosis, with a median age at diagnosis of 1.8 years. Children were more likely to receive an asthma diagnosis if their mothers had a preexisting condition or if they were born preterm. At follow-up, 34% of children had at least one upper respiratory tract infection. Sensory disorder, neoplasm, and pediatric complex chronic condition were rare, each occurring in less than 1% of the study cohort (BMJ. 2019 Jul 10. doi: 10.1136/bmj.l4151).
No significant association was found between prenatal exposure to the H1N1 vaccine and upper or lower respiratory infections, otitis media, all infections, neoplasms, sensory disorders, rates of urgent and inpatient health services use, pediatric complex chronic conditions, or mortality. A weak but significant association was observed for asthma (adjusted hazard ratio, 1.05; 95% confidence interval, 1.02-1.09), and a weak inverse association was found for gastrointestinal infections (adjusted incidence rate ratio, 0.94; 95% CI, 0.91-0.98).
“Although we observed a small, but statistically significant, increase in pediatric asthma and a reduction in gastrointestinal infections, we are not aware of any biologic mechanisms to explain these findings. Future studies in different settings and with different influenza vaccine formulations will be important for developing the evidence base on longer-term pediatric outcomes following influenza vaccination during pregnancy,” the investigators concluded.
The study was funded by grants from the Canadian Institutes of Health Research and the Institute for Clinical Evaluative Sciences.
according to Laura K. Walsh of the University of Ottawa and associates.
The investigators conducted a population-based retrospective cohort study from November 2009 to October 2010 of all live births within the province of Ontario. Of the 104,249 eligible live births reported to the Ontario birth registry, 31,295 were exposed to the H1N1 vaccine in utero. After adjustment, there were no significant differences in the women who did and did not receive vaccines during pregnancy, according to the study, published in the BMJ.
After a median follow-up of 5 years, 14% of children received an asthma diagnosis, with a median age at diagnosis of 1.8 years. Children were more likely to receive an asthma diagnosis if their mothers had a preexisting condition or if they were born preterm. At follow-up, 34% of children had at least one upper respiratory tract infection. Sensory disorder, neoplasm, and pediatric complex chronic condition were rare, each occurring in less than 1% of the study cohort (BMJ. 2019 Jul 10. doi: 10.1136/bmj.l4151).
No significant association was found between prenatal exposure to the H1N1 vaccine and upper or lower respiratory infections, otitis media, all infections, neoplasms, sensory disorders, rates of urgent and inpatient health services use, pediatric complex chronic conditions, or mortality. A weak but significant association was observed for asthma (adjusted hazard ratio, 1.05; 95% confidence interval, 1.02-1.09), and a weak inverse association was found for gastrointestinal infections (adjusted incidence rate ratio, 0.94; 95% CI, 0.91-0.98).
“Although we observed a small, but statistically significant, increase in pediatric asthma and a reduction in gastrointestinal infections, we are not aware of any biologic mechanisms to explain these findings. Future studies in different settings and with different influenza vaccine formulations will be important for developing the evidence base on longer-term pediatric outcomes following influenza vaccination during pregnancy,” the investigators concluded.
The study was funded by grants from the Canadian Institutes of Health Research and the Institute for Clinical Evaluative Sciences.
FROM THE BMJ