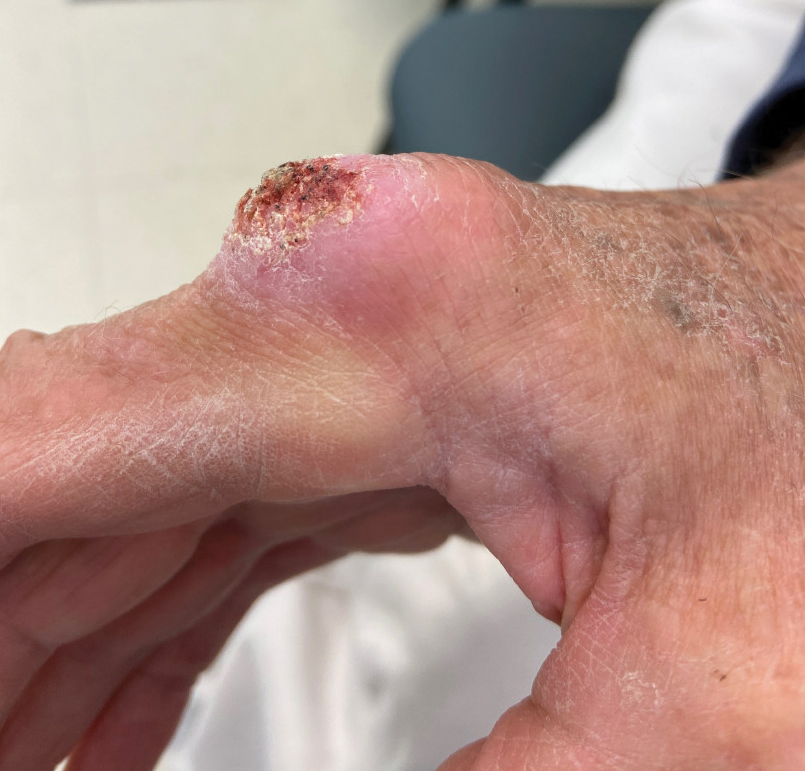
User login
Kaposi’s sarcoma: Antiretroviral-related improvements in survival measured
than their uninfected counterparts, based on the first such analysis of the American College of Surgeons’ National Cancer Database.
One-year overall survival for all patients with Kaposi’s sarcoma (KS), 74.9% in 2004-2007, rose by 6.4 percentage points to 81.3% in 2016-2018, with the use of ART for HIV starting in 2008. Two-year survival was up by an even larger 8.3 percentage points: 68.0% to 76.3%, said Amar D. Desai of New Jersey Medical School, Newark, and Shari R. Lipner, MD, of Weill Cornell Medicine, New York.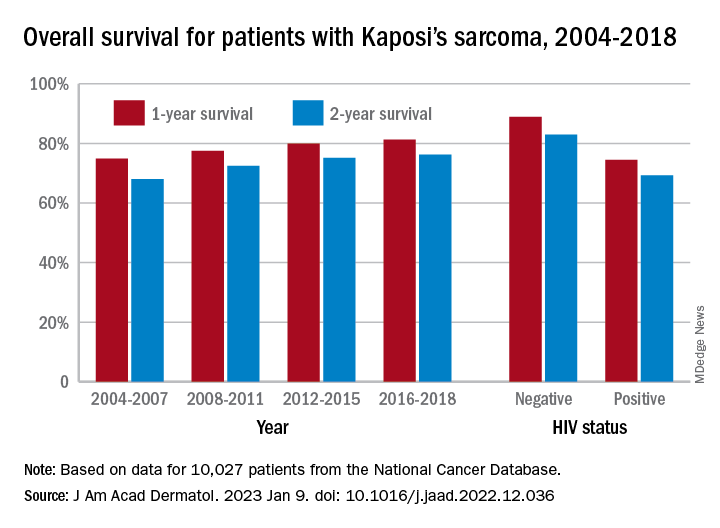
Since HIV-infected patients represented a much lower 46.7% of the Kaposi’s population in 2016-2018 than in 2004-2007 (70.5%), “better outcomes for all KS patients likely reflects advancements in ART, preventing many HIV+ patients from progressing to AIDS, changes in clinical practice with earlier treatment start, and more off-label treatments,” they wrote in the Journal of the American Academy of Dermatology.
Overall survival rates for the 10,027 patients with KS with data available in the National Cancer Database were 77.9% at 1 year and 72.4% at 2 years. HIV status had a significant (P < .0074) effect over the entire study period: One-year survival rates were 88.9% for HIV-negative and 74.5% for HIV-positive patients, and 2-year rates were 83.0% (HIV-negative) and 69.3% (HIV-positive), the investigators reported in what they called “the largest analysis since the advent of antiretroviral therapy for HIV in 2008.”
The improvement in overall survival, along with the continued differences in survival between HIV infected and noninfected patients, indicate that “dermatologists, as part of a multidisciplinary team including oncologists and infectious disease physicians, can play significant roles in early KS diagnosis,” Mr. Desai and Dr. Lipner said.
Mr. Desai had no conflicts of interest to report. Dr. Lipner has served as a consultant for Ortho-Dermatologics, Hoth Therapeutics, and BelleTorus Corporation.
than their uninfected counterparts, based on the first such analysis of the American College of Surgeons’ National Cancer Database.
One-year overall survival for all patients with Kaposi’s sarcoma (KS), 74.9% in 2004-2007, rose by 6.4 percentage points to 81.3% in 2016-2018, with the use of ART for HIV starting in 2008. Two-year survival was up by an even larger 8.3 percentage points: 68.0% to 76.3%, said Amar D. Desai of New Jersey Medical School, Newark, and Shari R. Lipner, MD, of Weill Cornell Medicine, New York.
Since HIV-infected patients represented a much lower 46.7% of the Kaposi’s population in 2016-2018 than in 2004-2007 (70.5%), “better outcomes for all KS patients likely reflects advancements in ART, preventing many HIV+ patients from progressing to AIDS, changes in clinical practice with earlier treatment start, and more off-label treatments,” they wrote in the Journal of the American Academy of Dermatology.
Overall survival rates for the 10,027 patients with KS with data available in the National Cancer Database were 77.9% at 1 year and 72.4% at 2 years. HIV status had a significant (P < .0074) effect over the entire study period: One-year survival rates were 88.9% for HIV-negative and 74.5% for HIV-positive patients, and 2-year rates were 83.0% (HIV-negative) and 69.3% (HIV-positive), the investigators reported in what they called “the largest analysis since the advent of antiretroviral therapy for HIV in 2008.”
The improvement in overall survival, along with the continued differences in survival between HIV infected and noninfected patients, indicate that “dermatologists, as part of a multidisciplinary team including oncologists and infectious disease physicians, can play significant roles in early KS diagnosis,” Mr. Desai and Dr. Lipner said.
Mr. Desai had no conflicts of interest to report. Dr. Lipner has served as a consultant for Ortho-Dermatologics, Hoth Therapeutics, and BelleTorus Corporation.
than their uninfected counterparts, based on the first such analysis of the American College of Surgeons’ National Cancer Database.
One-year overall survival for all patients with Kaposi’s sarcoma (KS), 74.9% in 2004-2007, rose by 6.4 percentage points to 81.3% in 2016-2018, with the use of ART for HIV starting in 2008. Two-year survival was up by an even larger 8.3 percentage points: 68.0% to 76.3%, said Amar D. Desai of New Jersey Medical School, Newark, and Shari R. Lipner, MD, of Weill Cornell Medicine, New York.
Since HIV-infected patients represented a much lower 46.7% of the Kaposi’s population in 2016-2018 than in 2004-2007 (70.5%), “better outcomes for all KS patients likely reflects advancements in ART, preventing many HIV+ patients from progressing to AIDS, changes in clinical practice with earlier treatment start, and more off-label treatments,” they wrote in the Journal of the American Academy of Dermatology.
Overall survival rates for the 10,027 patients with KS with data available in the National Cancer Database were 77.9% at 1 year and 72.4% at 2 years. HIV status had a significant (P < .0074) effect over the entire study period: One-year survival rates were 88.9% for HIV-negative and 74.5% for HIV-positive patients, and 2-year rates were 83.0% (HIV-negative) and 69.3% (HIV-positive), the investigators reported in what they called “the largest analysis since the advent of antiretroviral therapy for HIV in 2008.”
The improvement in overall survival, along with the continued differences in survival between HIV infected and noninfected patients, indicate that “dermatologists, as part of a multidisciplinary team including oncologists and infectious disease physicians, can play significant roles in early KS diagnosis,” Mr. Desai and Dr. Lipner said.
Mr. Desai had no conflicts of interest to report. Dr. Lipner has served as a consultant for Ortho-Dermatologics, Hoth Therapeutics, and BelleTorus Corporation.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Measles
I received a call late one night from a colleague in the emergency department of the children’s hospital. “This 2-year-old has a fever, cough, red eyes, and an impressive rash. I’ve personally never seen a case of measles, but I’m worried given that this child has never received the MMR vaccine.”
By the end of the call, I was worried too. Measles is a febrile respiratory illness classically accompanied by cough, coryza, conjunctivitis, and a characteristic maculopapular rash that begins on the face and spreads to the trunk and limbs. It is also highly contagious: 90% percent of susceptible, exposed individuals become infected.
Admittedly, measles is rare. Just 118 cases were reported in the United States in 2022, but 83 of those were in Columbus just 3 hours from where my colleague and I live and work. According to City of Columbus officials, the outbreak occurred almost exclusively in unimmunized children, the majority of whom were 5 years and younger. An unexpectedly high number of children were hospitalized. Typically, one in five people with measles will require hospitalization. In this outbreak, 33 children have been hospitalized as of Jan. 10.
Public health experts warn that 2023 could be much worse unless we increase measles immunization rates in the United States and globally. Immunization of around 95% of eligible people with two doses of measles-containing vaccine is associated with herd immunity. Globally, we’re falling short. Only 81% of the world’s children have received their first measle vaccine dose and only 71% have received the second dose. These are the lowest coverage rates for measles vaccine since 2008.
A 2022 joint press release from the Centers for Disease Control and Prevention and the World Health Organization noted that “measles anywhere is a threat everywhere, as the virus can quickly spread to multiple communities and across international borders.” Some prior measles outbreaks in the United States have started with a case in an international traveler or a U.S. resident who contracted measles during travel abroad.
In the United States, the number of children immunized with multiple routine vaccines has fallen in the last couple of years, in part because of pandemic-related disruptions in health care delivery. Increasing vaccine hesitancy, fueled by debates over the COVID-19 vaccine, may be slowing catch-up immunization in kids who fell behind.
Investigators from Emory University, Atlanta, and Marshfield Clinic Research Institute recently estimated that 9,145,026 U.S. children are susceptible to measles. If pandemic-level immunization rates continue without effective catch-up immunization, that number could rise to more than 15 million.
School vaccination requirements support efforts to ensure that kids are protected against vaccine-preventable diseases, but some data suggest that opposition to requiring MMR vaccine to attend public school is growing. According to a 2022 Kaiser Family Foundation Vaccine Monitor survey, 28% of U.S. adults – and 35% of parents of children under 18 – now say that parents should be able to decide to not vaccinate their children for measles, mumps, and rubella. That’s up from 16% of adults and 23% of parents in a 2019 Pew Research Center poll.
Public confidence in the benefits of MMR has also dropped modestly. About 85% of adults surveyed said that the benefits of MMR vaccine outweigh the risk, down from 88% in 2019. Among adults not vaccinated against COVID-19, only 70% said that benefits of these vaccines outweigh the risks.
While the WHO ramps up efforts to improve measles vaccination globally, pediatric clinicians can take steps now to mitigate the risk of measles outbreaks in their own communities. Query health records to understand how many eligible children in your practice have not yet received MMR vaccine. Notify families that vaccination is strongly recommended and make scheduling an appointment to receive vaccine easy. Some practices may have the bandwidth to offer evening and weekend hours for vaccine catch-up visits.
Curious about immunization rates in your state? The American Academy of Pediatrics has an interactive map that reports immunization coverage levels by state and provides comparisons to national rates and goals.
Prompt recognition and isolation of individuals with measles, along with prophylaxis of susceptible contacts, can limit community transmission. Measles can resemble other illnesses associated with fever and rash. Washington state has developed a screening tool to assist with recognition of measles. The CDC also has a measles outbreak toolkit that includes resources that outline clinical features and diagnoses, as well as strategies for talking to parents about vaccines.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant disclosed that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected].
I received a call late one night from a colleague in the emergency department of the children’s hospital. “This 2-year-old has a fever, cough, red eyes, and an impressive rash. I’ve personally never seen a case of measles, but I’m worried given that this child has never received the MMR vaccine.”
By the end of the call, I was worried too. Measles is a febrile respiratory illness classically accompanied by cough, coryza, conjunctivitis, and a characteristic maculopapular rash that begins on the face and spreads to the trunk and limbs. It is also highly contagious: 90% percent of susceptible, exposed individuals become infected.
Admittedly, measles is rare. Just 118 cases were reported in the United States in 2022, but 83 of those were in Columbus just 3 hours from where my colleague and I live and work. According to City of Columbus officials, the outbreak occurred almost exclusively in unimmunized children, the majority of whom were 5 years and younger. An unexpectedly high number of children were hospitalized. Typically, one in five people with measles will require hospitalization. In this outbreak, 33 children have been hospitalized as of Jan. 10.
Public health experts warn that 2023 could be much worse unless we increase measles immunization rates in the United States and globally. Immunization of around 95% of eligible people with two doses of measles-containing vaccine is associated with herd immunity. Globally, we’re falling short. Only 81% of the world’s children have received their first measle vaccine dose and only 71% have received the second dose. These are the lowest coverage rates for measles vaccine since 2008.
A 2022 joint press release from the Centers for Disease Control and Prevention and the World Health Organization noted that “measles anywhere is a threat everywhere, as the virus can quickly spread to multiple communities and across international borders.” Some prior measles outbreaks in the United States have started with a case in an international traveler or a U.S. resident who contracted measles during travel abroad.
In the United States, the number of children immunized with multiple routine vaccines has fallen in the last couple of years, in part because of pandemic-related disruptions in health care delivery. Increasing vaccine hesitancy, fueled by debates over the COVID-19 vaccine, may be slowing catch-up immunization in kids who fell behind.
Investigators from Emory University, Atlanta, and Marshfield Clinic Research Institute recently estimated that 9,145,026 U.S. children are susceptible to measles. If pandemic-level immunization rates continue without effective catch-up immunization, that number could rise to more than 15 million.
School vaccination requirements support efforts to ensure that kids are protected against vaccine-preventable diseases, but some data suggest that opposition to requiring MMR vaccine to attend public school is growing. According to a 2022 Kaiser Family Foundation Vaccine Monitor survey, 28% of U.S. adults – and 35% of parents of children under 18 – now say that parents should be able to decide to not vaccinate their children for measles, mumps, and rubella. That’s up from 16% of adults and 23% of parents in a 2019 Pew Research Center poll.
Public confidence in the benefits of MMR has also dropped modestly. About 85% of adults surveyed said that the benefits of MMR vaccine outweigh the risk, down from 88% in 2019. Among adults not vaccinated against COVID-19, only 70% said that benefits of these vaccines outweigh the risks.
While the WHO ramps up efforts to improve measles vaccination globally, pediatric clinicians can take steps now to mitigate the risk of measles outbreaks in their own communities. Query health records to understand how many eligible children in your practice have not yet received MMR vaccine. Notify families that vaccination is strongly recommended and make scheduling an appointment to receive vaccine easy. Some practices may have the bandwidth to offer evening and weekend hours for vaccine catch-up visits.
Curious about immunization rates in your state? The American Academy of Pediatrics has an interactive map that reports immunization coverage levels by state and provides comparisons to national rates and goals.
Prompt recognition and isolation of individuals with measles, along with prophylaxis of susceptible contacts, can limit community transmission. Measles can resemble other illnesses associated with fever and rash. Washington state has developed a screening tool to assist with recognition of measles. The CDC also has a measles outbreak toolkit that includes resources that outline clinical features and diagnoses, as well as strategies for talking to parents about vaccines.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant disclosed that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected].
I received a call late one night from a colleague in the emergency department of the children’s hospital. “This 2-year-old has a fever, cough, red eyes, and an impressive rash. I’ve personally never seen a case of measles, but I’m worried given that this child has never received the MMR vaccine.”
By the end of the call, I was worried too. Measles is a febrile respiratory illness classically accompanied by cough, coryza, conjunctivitis, and a characteristic maculopapular rash that begins on the face and spreads to the trunk and limbs. It is also highly contagious: 90% percent of susceptible, exposed individuals become infected.
Admittedly, measles is rare. Just 118 cases were reported in the United States in 2022, but 83 of those were in Columbus just 3 hours from where my colleague and I live and work. According to City of Columbus officials, the outbreak occurred almost exclusively in unimmunized children, the majority of whom were 5 years and younger. An unexpectedly high number of children were hospitalized. Typically, one in five people with measles will require hospitalization. In this outbreak, 33 children have been hospitalized as of Jan. 10.
Public health experts warn that 2023 could be much worse unless we increase measles immunization rates in the United States and globally. Immunization of around 95% of eligible people with two doses of measles-containing vaccine is associated with herd immunity. Globally, we’re falling short. Only 81% of the world’s children have received their first measle vaccine dose and only 71% have received the second dose. These are the lowest coverage rates for measles vaccine since 2008.
A 2022 joint press release from the Centers for Disease Control and Prevention and the World Health Organization noted that “measles anywhere is a threat everywhere, as the virus can quickly spread to multiple communities and across international borders.” Some prior measles outbreaks in the United States have started with a case in an international traveler or a U.S. resident who contracted measles during travel abroad.
In the United States, the number of children immunized with multiple routine vaccines has fallen in the last couple of years, in part because of pandemic-related disruptions in health care delivery. Increasing vaccine hesitancy, fueled by debates over the COVID-19 vaccine, may be slowing catch-up immunization in kids who fell behind.
Investigators from Emory University, Atlanta, and Marshfield Clinic Research Institute recently estimated that 9,145,026 U.S. children are susceptible to measles. If pandemic-level immunization rates continue without effective catch-up immunization, that number could rise to more than 15 million.
School vaccination requirements support efforts to ensure that kids are protected against vaccine-preventable diseases, but some data suggest that opposition to requiring MMR vaccine to attend public school is growing. According to a 2022 Kaiser Family Foundation Vaccine Monitor survey, 28% of U.S. adults – and 35% of parents of children under 18 – now say that parents should be able to decide to not vaccinate their children for measles, mumps, and rubella. That’s up from 16% of adults and 23% of parents in a 2019 Pew Research Center poll.
Public confidence in the benefits of MMR has also dropped modestly. About 85% of adults surveyed said that the benefits of MMR vaccine outweigh the risk, down from 88% in 2019. Among adults not vaccinated against COVID-19, only 70% said that benefits of these vaccines outweigh the risks.
While the WHO ramps up efforts to improve measles vaccination globally, pediatric clinicians can take steps now to mitigate the risk of measles outbreaks in their own communities. Query health records to understand how many eligible children in your practice have not yet received MMR vaccine. Notify families that vaccination is strongly recommended and make scheduling an appointment to receive vaccine easy. Some practices may have the bandwidth to offer evening and weekend hours for vaccine catch-up visits.
Curious about immunization rates in your state? The American Academy of Pediatrics has an interactive map that reports immunization coverage levels by state and provides comparisons to national rates and goals.
Prompt recognition and isolation of individuals with measles, along with prophylaxis of susceptible contacts, can limit community transmission. Measles can resemble other illnesses associated with fever and rash. Washington state has developed a screening tool to assist with recognition of measles. The CDC also has a measles outbreak toolkit that includes resources that outline clinical features and diagnoses, as well as strategies for talking to parents about vaccines.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant disclosed that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected].
New Omicron subvariant is ‘crazy infectious,’ COVID expert warns
“It’s crazy infectious,” said Paula Cannon, PhD, a virologist at the University of Southern California, Los Angeles. “All the things that have protected you for the past couple of years, I don’t think are going to protect you against this new crop of variants.”
XBB.1.5 is spreading quickly in the United States. It accounted for 27.6% of cases in the country in the week ending on Jan. 7, up from about 1% of cases at one point in December, according to the Centers for Disease Control and Prevention. It’s especially prevalent in the Northeast, now accounting for more than 70% of the cases in that region.
It’s spreading across the globe, too. Maria Van Kerkhove, PhD, technical lead of the World Health Organization, has called XBB.1.5 is “the most transmissible subvariant that has been detected yet.”
Ashish Jha, MD, the White House COVID-19 response coordinator, tweeted a few days ago that the spread of XBB.1.5 is “stunning” but cautioned that it’s unclear if the symptoms of infection will be more severe than for previous variants.
“Whether we’ll have an XBB.1.5 wave (and if yes, how big) will depend on many factors including immunity of the population, people’s actions, etc.,” he tweeted.
He urged people to get up to date on their boosters, wear a snug-fitting mask, and avoid crowded indoor spaces. He noted that people who haven’t been infected recently or haven’t gotten the bivalent booster likely have little protection against infection.
The symptoms for XBB.1.5 appear to be the same as for other versions of COVID-19. However, it’s less common for people infected with XBB.1.5 to report losing their sense of taste and smell, USA Today reported.
A version of this article first appeared on WebMD.com.
“It’s crazy infectious,” said Paula Cannon, PhD, a virologist at the University of Southern California, Los Angeles. “All the things that have protected you for the past couple of years, I don’t think are going to protect you against this new crop of variants.”
XBB.1.5 is spreading quickly in the United States. It accounted for 27.6% of cases in the country in the week ending on Jan. 7, up from about 1% of cases at one point in December, according to the Centers for Disease Control and Prevention. It’s especially prevalent in the Northeast, now accounting for more than 70% of the cases in that region.
It’s spreading across the globe, too. Maria Van Kerkhove, PhD, technical lead of the World Health Organization, has called XBB.1.5 is “the most transmissible subvariant that has been detected yet.”
Ashish Jha, MD, the White House COVID-19 response coordinator, tweeted a few days ago that the spread of XBB.1.5 is “stunning” but cautioned that it’s unclear if the symptoms of infection will be more severe than for previous variants.
“Whether we’ll have an XBB.1.5 wave (and if yes, how big) will depend on many factors including immunity of the population, people’s actions, etc.,” he tweeted.
He urged people to get up to date on their boosters, wear a snug-fitting mask, and avoid crowded indoor spaces. He noted that people who haven’t been infected recently or haven’t gotten the bivalent booster likely have little protection against infection.
The symptoms for XBB.1.5 appear to be the same as for other versions of COVID-19. However, it’s less common for people infected with XBB.1.5 to report losing their sense of taste and smell, USA Today reported.
A version of this article first appeared on WebMD.com.
“It’s crazy infectious,” said Paula Cannon, PhD, a virologist at the University of Southern California, Los Angeles. “All the things that have protected you for the past couple of years, I don’t think are going to protect you against this new crop of variants.”
XBB.1.5 is spreading quickly in the United States. It accounted for 27.6% of cases in the country in the week ending on Jan. 7, up from about 1% of cases at one point in December, according to the Centers for Disease Control and Prevention. It’s especially prevalent in the Northeast, now accounting for more than 70% of the cases in that region.
It’s spreading across the globe, too. Maria Van Kerkhove, PhD, technical lead of the World Health Organization, has called XBB.1.5 is “the most transmissible subvariant that has been detected yet.”
Ashish Jha, MD, the White House COVID-19 response coordinator, tweeted a few days ago that the spread of XBB.1.5 is “stunning” but cautioned that it’s unclear if the symptoms of infection will be more severe than for previous variants.
“Whether we’ll have an XBB.1.5 wave (and if yes, how big) will depend on many factors including immunity of the population, people’s actions, etc.,” he tweeted.
He urged people to get up to date on their boosters, wear a snug-fitting mask, and avoid crowded indoor spaces. He noted that people who haven’t been infected recently or haven’t gotten the bivalent booster likely have little protection against infection.
The symptoms for XBB.1.5 appear to be the same as for other versions of COVID-19. However, it’s less common for people infected with XBB.1.5 to report losing their sense of taste and smell, USA Today reported.
A version of this article first appeared on WebMD.com.
Autopsies show COVID virus invades entire body
A study on the subject was published in the journal Nature. The researchers completed autopsies from April 2020 to March 2021 of 44 unvaccinated people who had severe COVID-19. The median age was 62.5 years old, and 30% were female. Extensive brain sampling was done for 11 cases.
Because of its nature as a respiratory illness, SARS-CoV-2 was most widespread in the respiratory system such as in the lungs. But it was also found in 79 other body locations, including the heart, kidneys, liver, muscles, nerves, reproductive tract, and eyes.
The researchers said their work shows the SARS-CoV-2 “is capable of infecting and replicating within the human brain.” They also said their results indicate the virus spreads via the blood early during infection, which “seeds the virus throughout the body following infection of the respiratory tract.”
The authors noted that, while the virus was found outside the respiratory tract, they did not find signs of inflammation beyond the respiratory system.
The results will help narrow down treatments for long COVID, and particularly support the idea of using the antiviral drug Paxlovid to treat long COVID, according to a blog post from the National Institute of Allergy and Infectious Diseases. A clinical trial is already underway examining the treatment, and results are expected in January 2024.
A version of this article first appeared on WebMD.com.
A study on the subject was published in the journal Nature. The researchers completed autopsies from April 2020 to March 2021 of 44 unvaccinated people who had severe COVID-19. The median age was 62.5 years old, and 30% were female. Extensive brain sampling was done for 11 cases.
Because of its nature as a respiratory illness, SARS-CoV-2 was most widespread in the respiratory system such as in the lungs. But it was also found in 79 other body locations, including the heart, kidneys, liver, muscles, nerves, reproductive tract, and eyes.
The researchers said their work shows the SARS-CoV-2 “is capable of infecting and replicating within the human brain.” They also said their results indicate the virus spreads via the blood early during infection, which “seeds the virus throughout the body following infection of the respiratory tract.”
The authors noted that, while the virus was found outside the respiratory tract, they did not find signs of inflammation beyond the respiratory system.
The results will help narrow down treatments for long COVID, and particularly support the idea of using the antiviral drug Paxlovid to treat long COVID, according to a blog post from the National Institute of Allergy and Infectious Diseases. A clinical trial is already underway examining the treatment, and results are expected in January 2024.
A version of this article first appeared on WebMD.com.
A study on the subject was published in the journal Nature. The researchers completed autopsies from April 2020 to March 2021 of 44 unvaccinated people who had severe COVID-19. The median age was 62.5 years old, and 30% were female. Extensive brain sampling was done for 11 cases.
Because of its nature as a respiratory illness, SARS-CoV-2 was most widespread in the respiratory system such as in the lungs. But it was also found in 79 other body locations, including the heart, kidneys, liver, muscles, nerves, reproductive tract, and eyes.
The researchers said their work shows the SARS-CoV-2 “is capable of infecting and replicating within the human brain.” They also said their results indicate the virus spreads via the blood early during infection, which “seeds the virus throughout the body following infection of the respiratory tract.”
The authors noted that, while the virus was found outside the respiratory tract, they did not find signs of inflammation beyond the respiratory system.
The results will help narrow down treatments for long COVID, and particularly support the idea of using the antiviral drug Paxlovid to treat long COVID, according to a blog post from the National Institute of Allergy and Infectious Diseases. A clinical trial is already underway examining the treatment, and results are expected in January 2024.
A version of this article first appeared on WebMD.com.
FROM NATURE
Advanced Primary Care program boosts COVID-19 results
The better outcomes were seen in higher vaccination rates and fewer infections, hospitalizations, and deaths from the disease, according to study authors, led by Emily Gruber, MBA, MPH, with the Maryland Primary Care Program, Maryland Department of Health in Baltimore.
The results were published online in JAMA Network Open.
The study population was divided into MDPCP participants (n = 208,146) and a matched cohort (n = 37,203) of beneficiaries not attributed to MDPCP practices but who met eligibility criteria for study participation from Jan. 1, 2020, through Dec. 31, 2021.
More vaccinations, more antibody treatments
Researchers broke down the comparisons of better outcomes: 84.47% of MDPCP beneficiaries were fully vaccinated vs. 77.93% of nonparticipating beneficiaries (P less than .001). COVID-19–positive program beneficiaries also received monoclonal antibody treatment more often (8.45% vs. 6.11%; P less than .001).
Plus, program participants received more care via telehealth (62.95% vs. 54.53%; P less than .001) compared with those not participating.
Regarding secondary outcomes, MDPCP beneficiaries had lower rates of COVID cases (6.55% vs. 7.09%; P less than .001), lower rates of COVID-19 hospitalizations (1.81% vs. 2.06%; P = .001), and lower rates of death due to COVID-19 (0.56% vs. 0.77%; P less than .001).
Program components
Enrollment in the MDPCP is voluntary, and primary care practices can apply each year to be part of the program.
The model integrates primary care and public health in the pandemic response. It was created by the Maryland Department of Health (MDH) and the Centers for Medicare & Medicaid Services (CMS).
It expands the role of primary care to include services such as expanded care management, integrated behavioral health, data-driven care, and screenings and referrals to address social needs.
Coauthor Howard Haft, MD, MMM, with the Maryland Department of Public Health, said in an interview that among the most important factors in the program’s success were giving providers vaccines to distribute and then giving providers data on how many patients are vaccinated, and who’s not vaccinated but at high risk, and how those rates compare to other practices.
As to whether this could be a widespread model, Dr. Haft said, “It’s highly replicable.”
“Every state in the nation overall has all of these resources. It’s a matter of having the operational and political will to put those resources together. Almost every state has the technological ability to use their health information exchange to help tie pieces together.”
Vaccines and testing made available to providers
Making ample vaccines and testing available to providers in their offices helped patients get those services in a place they trust, Dr. Haft said.
The model also included a payment system for providers that included a significant amount of non–visit-based payments when many locations were closed in the height of the pandemic.
“That helped financially,” as did providing free telehealth platforms to practices with training on how to use them, Dr. Haft said.
‘Innovative and important’
Renu Tipirneni, MD, an assistant professor of internal medicine at the University of Michigan and at the Institute for Healthcare Policy and Innovation in Ann Arbor, said Maryland is out front putting into practice what practices nationwide aspire to do – coordinating physical and mental health and social needs and integrating primary and public health. Dr. Tipirneni, who was not involved with the study, said she was impressed the researchers were able to show statistically significant improvement with COVID-19 outcomes in the first 2 years.
“In terms of health outcomes, we often have to wait longer to see good outcomes,” she said. “It’s a really innovative and important model.”
She said states can learn from each other and this model is an example.
Integrating primary care and public health and addressing social needs may be the biggest challenges for states, she said, as those realms typically have been siloed.
“But they may be the key components to achieving these outcomes,” she said.
Take-home message
The most important benefit of the program is that data suggest it saves lives, according to Dr. Haft. While the actual difference between COVID deaths in the program and nonprogram groups was small, multiplying that savings across the nation shows substantial potential benefit, he explained.
“At a time when we were losing lives at an unconscionable rate, we were able to make a difference in saving lives,” Dr. Haft said.
Authors report no relevant financial disclosures.
The study received financial support from the Maryland Department of Health.
Dr. Tiperneni is helping evaluate Michigan’s Medicaid contract.
The better outcomes were seen in higher vaccination rates and fewer infections, hospitalizations, and deaths from the disease, according to study authors, led by Emily Gruber, MBA, MPH, with the Maryland Primary Care Program, Maryland Department of Health in Baltimore.
The results were published online in JAMA Network Open.
The study population was divided into MDPCP participants (n = 208,146) and a matched cohort (n = 37,203) of beneficiaries not attributed to MDPCP practices but who met eligibility criteria for study participation from Jan. 1, 2020, through Dec. 31, 2021.
More vaccinations, more antibody treatments
Researchers broke down the comparisons of better outcomes: 84.47% of MDPCP beneficiaries were fully vaccinated vs. 77.93% of nonparticipating beneficiaries (P less than .001). COVID-19–positive program beneficiaries also received monoclonal antibody treatment more often (8.45% vs. 6.11%; P less than .001).
Plus, program participants received more care via telehealth (62.95% vs. 54.53%; P less than .001) compared with those not participating.
Regarding secondary outcomes, MDPCP beneficiaries had lower rates of COVID cases (6.55% vs. 7.09%; P less than .001), lower rates of COVID-19 hospitalizations (1.81% vs. 2.06%; P = .001), and lower rates of death due to COVID-19 (0.56% vs. 0.77%; P less than .001).
Program components
Enrollment in the MDPCP is voluntary, and primary care practices can apply each year to be part of the program.
The model integrates primary care and public health in the pandemic response. It was created by the Maryland Department of Health (MDH) and the Centers for Medicare & Medicaid Services (CMS).
It expands the role of primary care to include services such as expanded care management, integrated behavioral health, data-driven care, and screenings and referrals to address social needs.
Coauthor Howard Haft, MD, MMM, with the Maryland Department of Public Health, said in an interview that among the most important factors in the program’s success were giving providers vaccines to distribute and then giving providers data on how many patients are vaccinated, and who’s not vaccinated but at high risk, and how those rates compare to other practices.
As to whether this could be a widespread model, Dr. Haft said, “It’s highly replicable.”
“Every state in the nation overall has all of these resources. It’s a matter of having the operational and political will to put those resources together. Almost every state has the technological ability to use their health information exchange to help tie pieces together.”
Vaccines and testing made available to providers
Making ample vaccines and testing available to providers in their offices helped patients get those services in a place they trust, Dr. Haft said.
The model also included a payment system for providers that included a significant amount of non–visit-based payments when many locations were closed in the height of the pandemic.
“That helped financially,” as did providing free telehealth platforms to practices with training on how to use them, Dr. Haft said.
‘Innovative and important’
Renu Tipirneni, MD, an assistant professor of internal medicine at the University of Michigan and at the Institute for Healthcare Policy and Innovation in Ann Arbor, said Maryland is out front putting into practice what practices nationwide aspire to do – coordinating physical and mental health and social needs and integrating primary and public health. Dr. Tipirneni, who was not involved with the study, said she was impressed the researchers were able to show statistically significant improvement with COVID-19 outcomes in the first 2 years.
“In terms of health outcomes, we often have to wait longer to see good outcomes,” she said. “It’s a really innovative and important model.”
She said states can learn from each other and this model is an example.
Integrating primary care and public health and addressing social needs may be the biggest challenges for states, she said, as those realms typically have been siloed.
“But they may be the key components to achieving these outcomes,” she said.
Take-home message
The most important benefit of the program is that data suggest it saves lives, according to Dr. Haft. While the actual difference between COVID deaths in the program and nonprogram groups was small, multiplying that savings across the nation shows substantial potential benefit, he explained.
“At a time when we were losing lives at an unconscionable rate, we were able to make a difference in saving lives,” Dr. Haft said.
Authors report no relevant financial disclosures.
The study received financial support from the Maryland Department of Health.
Dr. Tiperneni is helping evaluate Michigan’s Medicaid contract.
The better outcomes were seen in higher vaccination rates and fewer infections, hospitalizations, and deaths from the disease, according to study authors, led by Emily Gruber, MBA, MPH, with the Maryland Primary Care Program, Maryland Department of Health in Baltimore.
The results were published online in JAMA Network Open.
The study population was divided into MDPCP participants (n = 208,146) and a matched cohort (n = 37,203) of beneficiaries not attributed to MDPCP practices but who met eligibility criteria for study participation from Jan. 1, 2020, through Dec. 31, 2021.
More vaccinations, more antibody treatments
Researchers broke down the comparisons of better outcomes: 84.47% of MDPCP beneficiaries were fully vaccinated vs. 77.93% of nonparticipating beneficiaries (P less than .001). COVID-19–positive program beneficiaries also received monoclonal antibody treatment more often (8.45% vs. 6.11%; P less than .001).
Plus, program participants received more care via telehealth (62.95% vs. 54.53%; P less than .001) compared with those not participating.
Regarding secondary outcomes, MDPCP beneficiaries had lower rates of COVID cases (6.55% vs. 7.09%; P less than .001), lower rates of COVID-19 hospitalizations (1.81% vs. 2.06%; P = .001), and lower rates of death due to COVID-19 (0.56% vs. 0.77%; P less than .001).
Program components
Enrollment in the MDPCP is voluntary, and primary care practices can apply each year to be part of the program.
The model integrates primary care and public health in the pandemic response. It was created by the Maryland Department of Health (MDH) and the Centers for Medicare & Medicaid Services (CMS).
It expands the role of primary care to include services such as expanded care management, integrated behavioral health, data-driven care, and screenings and referrals to address social needs.
Coauthor Howard Haft, MD, MMM, with the Maryland Department of Public Health, said in an interview that among the most important factors in the program’s success were giving providers vaccines to distribute and then giving providers data on how many patients are vaccinated, and who’s not vaccinated but at high risk, and how those rates compare to other practices.
As to whether this could be a widespread model, Dr. Haft said, “It’s highly replicable.”
“Every state in the nation overall has all of these resources. It’s a matter of having the operational and political will to put those resources together. Almost every state has the technological ability to use their health information exchange to help tie pieces together.”
Vaccines and testing made available to providers
Making ample vaccines and testing available to providers in their offices helped patients get those services in a place they trust, Dr. Haft said.
The model also included a payment system for providers that included a significant amount of non–visit-based payments when many locations were closed in the height of the pandemic.
“That helped financially,” as did providing free telehealth platforms to practices with training on how to use them, Dr. Haft said.
‘Innovative and important’
Renu Tipirneni, MD, an assistant professor of internal medicine at the University of Michigan and at the Institute for Healthcare Policy and Innovation in Ann Arbor, said Maryland is out front putting into practice what practices nationwide aspire to do – coordinating physical and mental health and social needs and integrating primary and public health. Dr. Tipirneni, who was not involved with the study, said she was impressed the researchers were able to show statistically significant improvement with COVID-19 outcomes in the first 2 years.
“In terms of health outcomes, we often have to wait longer to see good outcomes,” she said. “It’s a really innovative and important model.”
She said states can learn from each other and this model is an example.
Integrating primary care and public health and addressing social needs may be the biggest challenges for states, she said, as those realms typically have been siloed.
“But they may be the key components to achieving these outcomes,” she said.
Take-home message
The most important benefit of the program is that data suggest it saves lives, according to Dr. Haft. While the actual difference between COVID deaths in the program and nonprogram groups was small, multiplying that savings across the nation shows substantial potential benefit, he explained.
“At a time when we were losing lives at an unconscionable rate, we were able to make a difference in saving lives,” Dr. Haft said.
Authors report no relevant financial disclosures.
The study received financial support from the Maryland Department of Health.
Dr. Tiperneni is helping evaluate Michigan’s Medicaid contract.
FROM JAMA NETWORK OPEN
Pediatric vaccination rates have failed to recover
I guess we shouldn’t be surprised that vaccination rates in this country fell during the frenzy created by the COVID pandemic. We had a lot on our plates. Schools closed and many of us retreated into what seemed to be the safety of our homes. Parents were reluctant to take their children anywhere, let alone a pediatrician’s office. State health agencies wisely focused on collecting case figures and then shepherding the efforts to immunize against SARS-CoV-2 once vaccines were available. Tracking and promoting the existing children’s vaccinations fell off the priority list, even in places with exemplary vaccination rates.
Whether or not the pandemic is over continues to be a topic for debate, but there is clearly a general shift toward a new normalcy. However, vaccination rates of our children have not rebounded to prepandemic levels. In fact, in some areas they are continuing to fall.
In a recent guest essay in the New York Times, Ezekiel J. Emmanuel, MD, PhD, a physician and professor of medical ethics and health policy at the University of Pennsylvania, and Matthew Guido, his research assistant, explore the reasons for this lack of a significant rebound. The authors cite recent outbreaks of measles in Ohio and polio in New York City as examples of the peril we are facing if we fail to reverse the trend. In some areas measles vaccine rates alarmingly have dipped below the threshold for herd immunity.
While Dr. Emmanuel and Mr. Guido acknowledge that the pandemic was a major driver of the falling vaccination rates they lay blame on the persistent decline on three factors that they view as correctable: nonmedical exemptions, our failure to vigorously enforce existing vaccine requirements, and inadequate public health campaigns.
The authors underestimate the lingering effect of the pandemic on parents’ vaccine hesitancy. As a septuagenarian who often hangs out with other septuagenarians I view the rapid development and effectiveness of the COVID vaccine as astounding and a boost for vaccines in general. However, were I much younger I might treat the vaccine’s success with a shrug. After some initial concern, the younger half of the population didn’t seem to see the illness as much of a threat to themselves or their peers. This attitude was reinforced by the fact that few of their peers, including those who were unvaccinated, were getting seriously ill. Despite all the hype, most parents and their children never ended up getting seriously ill.
You can understand why many parents might be quick to toss what you and I consider a successful COVID vaccine onto what they view as a growing pile of vaccines for diseases that in their experience have never sickened or killed anyone they have known.
Let’s be honest: Over the last half century we have produced several generations of parents who have little knowledge and certainly no personal experience with a childhood disease on the order or magnitude of polio. The vaccines that we have developed during their lifetimes have been targeted at diseases such as haemophilus influenzae meningitis that, while serious and anxiety provoking for pediatricians, occur so sporadically that most parents have no personal experience to motivate them to vaccinate their children.
Dr. Emmanuel and Mr. Guido are correct in advocating for the broader elimination of nonmedical exemptions and urging us to find the political will to vigorously enforce the vaccine requirements we have already enacted. I agree that our promotional campaigns need to be more robust. But, this will be a difficult challenge unless we can impress our audience with our straight talk and honesty. We must acknowledge and then explain why all vaccines are not created equal and that some are of more critical importance than others.
We are slowly learning that education isn’t the cure-all for vaccine hesitancy we once thought it was. And using scare tactics can backfire and create dysfunctional anxiety. We must choosing our words and target audience carefully. And ... having the political will to force parents into doing the right thing will be critical if we wish to restore our vaccination rates.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
I guess we shouldn’t be surprised that vaccination rates in this country fell during the frenzy created by the COVID pandemic. We had a lot on our plates. Schools closed and many of us retreated into what seemed to be the safety of our homes. Parents were reluctant to take their children anywhere, let alone a pediatrician’s office. State health agencies wisely focused on collecting case figures and then shepherding the efforts to immunize against SARS-CoV-2 once vaccines were available. Tracking and promoting the existing children’s vaccinations fell off the priority list, even in places with exemplary vaccination rates.
Whether or not the pandemic is over continues to be a topic for debate, but there is clearly a general shift toward a new normalcy. However, vaccination rates of our children have not rebounded to prepandemic levels. In fact, in some areas they are continuing to fall.
In a recent guest essay in the New York Times, Ezekiel J. Emmanuel, MD, PhD, a physician and professor of medical ethics and health policy at the University of Pennsylvania, and Matthew Guido, his research assistant, explore the reasons for this lack of a significant rebound. The authors cite recent outbreaks of measles in Ohio and polio in New York City as examples of the peril we are facing if we fail to reverse the trend. In some areas measles vaccine rates alarmingly have dipped below the threshold for herd immunity.
While Dr. Emmanuel and Mr. Guido acknowledge that the pandemic was a major driver of the falling vaccination rates they lay blame on the persistent decline on three factors that they view as correctable: nonmedical exemptions, our failure to vigorously enforce existing vaccine requirements, and inadequate public health campaigns.
The authors underestimate the lingering effect of the pandemic on parents’ vaccine hesitancy. As a septuagenarian who often hangs out with other septuagenarians I view the rapid development and effectiveness of the COVID vaccine as astounding and a boost for vaccines in general. However, were I much younger I might treat the vaccine’s success with a shrug. After some initial concern, the younger half of the population didn’t seem to see the illness as much of a threat to themselves or their peers. This attitude was reinforced by the fact that few of their peers, including those who were unvaccinated, were getting seriously ill. Despite all the hype, most parents and their children never ended up getting seriously ill.
You can understand why many parents might be quick to toss what you and I consider a successful COVID vaccine onto what they view as a growing pile of vaccines for diseases that in their experience have never sickened or killed anyone they have known.
Let’s be honest: Over the last half century we have produced several generations of parents who have little knowledge and certainly no personal experience with a childhood disease on the order or magnitude of polio. The vaccines that we have developed during their lifetimes have been targeted at diseases such as haemophilus influenzae meningitis that, while serious and anxiety provoking for pediatricians, occur so sporadically that most parents have no personal experience to motivate them to vaccinate their children.
Dr. Emmanuel and Mr. Guido are correct in advocating for the broader elimination of nonmedical exemptions and urging us to find the political will to vigorously enforce the vaccine requirements we have already enacted. I agree that our promotional campaigns need to be more robust. But, this will be a difficult challenge unless we can impress our audience with our straight talk and honesty. We must acknowledge and then explain why all vaccines are not created equal and that some are of more critical importance than others.
We are slowly learning that education isn’t the cure-all for vaccine hesitancy we once thought it was. And using scare tactics can backfire and create dysfunctional anxiety. We must choosing our words and target audience carefully. And ... having the political will to force parents into doing the right thing will be critical if we wish to restore our vaccination rates.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
I guess we shouldn’t be surprised that vaccination rates in this country fell during the frenzy created by the COVID pandemic. We had a lot on our plates. Schools closed and many of us retreated into what seemed to be the safety of our homes. Parents were reluctant to take their children anywhere, let alone a pediatrician’s office. State health agencies wisely focused on collecting case figures and then shepherding the efforts to immunize against SARS-CoV-2 once vaccines were available. Tracking and promoting the existing children’s vaccinations fell off the priority list, even in places with exemplary vaccination rates.
Whether or not the pandemic is over continues to be a topic for debate, but there is clearly a general shift toward a new normalcy. However, vaccination rates of our children have not rebounded to prepandemic levels. In fact, in some areas they are continuing to fall.
In a recent guest essay in the New York Times, Ezekiel J. Emmanuel, MD, PhD, a physician and professor of medical ethics and health policy at the University of Pennsylvania, and Matthew Guido, his research assistant, explore the reasons for this lack of a significant rebound. The authors cite recent outbreaks of measles in Ohio and polio in New York City as examples of the peril we are facing if we fail to reverse the trend. In some areas measles vaccine rates alarmingly have dipped below the threshold for herd immunity.
While Dr. Emmanuel and Mr. Guido acknowledge that the pandemic was a major driver of the falling vaccination rates they lay blame on the persistent decline on three factors that they view as correctable: nonmedical exemptions, our failure to vigorously enforce existing vaccine requirements, and inadequate public health campaigns.
The authors underestimate the lingering effect of the pandemic on parents’ vaccine hesitancy. As a septuagenarian who often hangs out with other septuagenarians I view the rapid development and effectiveness of the COVID vaccine as astounding and a boost for vaccines in general. However, were I much younger I might treat the vaccine’s success with a shrug. After some initial concern, the younger half of the population didn’t seem to see the illness as much of a threat to themselves or their peers. This attitude was reinforced by the fact that few of their peers, including those who were unvaccinated, were getting seriously ill. Despite all the hype, most parents and their children never ended up getting seriously ill.
You can understand why many parents might be quick to toss what you and I consider a successful COVID vaccine onto what they view as a growing pile of vaccines for diseases that in their experience have never sickened or killed anyone they have known.
Let’s be honest: Over the last half century we have produced several generations of parents who have little knowledge and certainly no personal experience with a childhood disease on the order or magnitude of polio. The vaccines that we have developed during their lifetimes have been targeted at diseases such as haemophilus influenzae meningitis that, while serious and anxiety provoking for pediatricians, occur so sporadically that most parents have no personal experience to motivate them to vaccinate their children.
Dr. Emmanuel and Mr. Guido are correct in advocating for the broader elimination of nonmedical exemptions and urging us to find the political will to vigorously enforce the vaccine requirements we have already enacted. I agree that our promotional campaigns need to be more robust. But, this will be a difficult challenge unless we can impress our audience with our straight talk and honesty. We must acknowledge and then explain why all vaccines are not created equal and that some are of more critical importance than others.
We are slowly learning that education isn’t the cure-all for vaccine hesitancy we once thought it was. And using scare tactics can backfire and create dysfunctional anxiety. We must choosing our words and target audience carefully. And ... having the political will to force parents into doing the right thing will be critical if we wish to restore our vaccination rates.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Berdazimer gel under review at FDA for treating molluscum contagiosum
, the manufacturer announced.
If the submission is accepted by the FDA, the topical product could be approved in the first quarter of 2024, according to a press release from Novan, the manufacturer. If approved, it would be the first-in-class topical treatment for MC, the common, contagious viral skin infection that affects approximately six million individuals in the United States each year, most of them children aged 1-14 years, the statement noted. No FDA-approved therapies currently exist for the condition, which causes unsightly lesions on the face, trunk, limbs, and axillae that may persist untreated for a period of years.
The active ingredient in berdazimer gel 10.3% is berdazimer sodium, a nitric oxide–releasing agent. A 3.4% formulation is in development for the topical treatment of acne, according to the company.
The submission for FDA approval is based on data from the B-SIMPLE4 study, a phase 3 randomized trial of nearly 900 individuals with MC aged 6 months and older (mean age, 6.6 years), with 3-70 raised lesions. Participants were randomized to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily for 12 weeks. The results were published in JAMA Dermatology.
The primary outcome was complete clearance of all lesions. At 12 weeks, 32.4% of patients in the berdazimer group achieved this outcome vs. 19.7% of those in the vehicle group (P < .001). Overall adverse event rates were low in both groups; 4.1% of patients on berdazimer and 0.7% of those on the vehicle experienced adverse events that led to discontinuation of treatment. The most common adverse events across both groups were application-site pain and erythema, and most of these were mild or moderate.
, the manufacturer announced.
If the submission is accepted by the FDA, the topical product could be approved in the first quarter of 2024, according to a press release from Novan, the manufacturer. If approved, it would be the first-in-class topical treatment for MC, the common, contagious viral skin infection that affects approximately six million individuals in the United States each year, most of them children aged 1-14 years, the statement noted. No FDA-approved therapies currently exist for the condition, which causes unsightly lesions on the face, trunk, limbs, and axillae that may persist untreated for a period of years.
The active ingredient in berdazimer gel 10.3% is berdazimer sodium, a nitric oxide–releasing agent. A 3.4% formulation is in development for the topical treatment of acne, according to the company.
The submission for FDA approval is based on data from the B-SIMPLE4 study, a phase 3 randomized trial of nearly 900 individuals with MC aged 6 months and older (mean age, 6.6 years), with 3-70 raised lesions. Participants were randomized to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily for 12 weeks. The results were published in JAMA Dermatology.
The primary outcome was complete clearance of all lesions. At 12 weeks, 32.4% of patients in the berdazimer group achieved this outcome vs. 19.7% of those in the vehicle group (P < .001). Overall adverse event rates were low in both groups; 4.1% of patients on berdazimer and 0.7% of those on the vehicle experienced adverse events that led to discontinuation of treatment. The most common adverse events across both groups were application-site pain and erythema, and most of these were mild or moderate.
, the manufacturer announced.
If the submission is accepted by the FDA, the topical product could be approved in the first quarter of 2024, according to a press release from Novan, the manufacturer. If approved, it would be the first-in-class topical treatment for MC, the common, contagious viral skin infection that affects approximately six million individuals in the United States each year, most of them children aged 1-14 years, the statement noted. No FDA-approved therapies currently exist for the condition, which causes unsightly lesions on the face, trunk, limbs, and axillae that may persist untreated for a period of years.
The active ingredient in berdazimer gel 10.3% is berdazimer sodium, a nitric oxide–releasing agent. A 3.4% formulation is in development for the topical treatment of acne, according to the company.
The submission for FDA approval is based on data from the B-SIMPLE4 study, a phase 3 randomized trial of nearly 900 individuals with MC aged 6 months and older (mean age, 6.6 years), with 3-70 raised lesions. Participants were randomized to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily for 12 weeks. The results were published in JAMA Dermatology.
The primary outcome was complete clearance of all lesions. At 12 weeks, 32.4% of patients in the berdazimer group achieved this outcome vs. 19.7% of those in the vehicle group (P < .001). Overall adverse event rates were low in both groups; 4.1% of patients on berdazimer and 0.7% of those on the vehicle experienced adverse events that led to discontinuation of treatment. The most common adverse events across both groups were application-site pain and erythema, and most of these were mild or moderate.
Children and COVID: New cases fell as the old year ended
The end of 2022 saw a drop in new COVID-19 cases in children, even as rates of emergency department visits continued upward trends that began in late October.
New cases for the week of Dec. 23-29 fell for the first time since late November, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The AAP/CHA analysis of publicly available state data differs somewhat from figures reported by the Centers for Disease Control and Prevention, which has new cases for the latest available week, Dec.18-24, at just over 27,000 after 3 straight weeks of declines from a count of almost 63,000 for the week ending Nov. 26. The CDC, however, updates previously reported data on a regular basis, so that 27,000 is likely to increase in the coming weeks.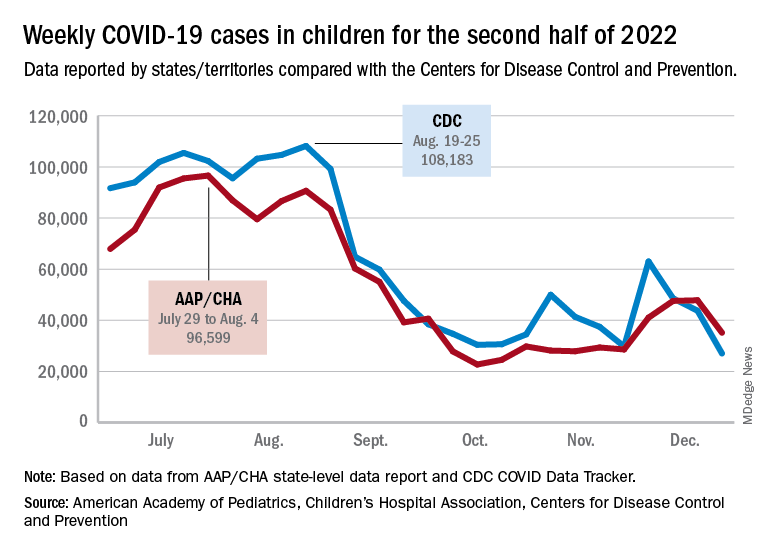
The CDC line on the graph also shows a peak for the week of Oct. 30 to Nov. 5 when new cases reached almost 50,000, compared with almost 30,000 reported for the week of Oct. 28 to Nov. 3 by the AAP and CHA in their report of state-level data. The AAP and CHA put the total number of child COVID cases since the start of the pandemic at 15.2 million as of Dec. 29, while the CDC reports 16.2 million cases as of Dec. 28.
There have been 1,975 deaths from COVID-19 in children aged 0-17 years, according to the CDC, which amounts to just over 0.2% of all COVID deaths for which age group data were available.
CDC data on emergency department visits involving diagnosed COVID-19 have been rising since late October. In children aged 0-11 years, for example, COVID was involved in 1.0% of ED visits (7-day average) as late as Nov. 4, but by Dec. 27 that rate was 2.6%. Children aged 12-15 years went from 0.6% on Oct. 28 to 1.5% on Dec. 27, while 16- to 17-year-olds had ED visit rates of 0.6% on Oct. 19 and 1.7% on Dec. 27, the CDC said on its COVID Data Tracker.
New hospital admissions with diagnosed COVID, which had been following the same upward trend as ED visits since late October, halted that rise in children aged 0-17 years and have gone no higher than 0.29 per 100,000 population since Dec. 9, the CDC data show.
The end of 2022 saw a drop in new COVID-19 cases in children, even as rates of emergency department visits continued upward trends that began in late October.
New cases for the week of Dec. 23-29 fell for the first time since late November, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The AAP/CHA analysis of publicly available state data differs somewhat from figures reported by the Centers for Disease Control and Prevention, which has new cases for the latest available week, Dec.18-24, at just over 27,000 after 3 straight weeks of declines from a count of almost 63,000 for the week ending Nov. 26. The CDC, however, updates previously reported data on a regular basis, so that 27,000 is likely to increase in the coming weeks.
The CDC line on the graph also shows a peak for the week of Oct. 30 to Nov. 5 when new cases reached almost 50,000, compared with almost 30,000 reported for the week of Oct. 28 to Nov. 3 by the AAP and CHA in their report of state-level data. The AAP and CHA put the total number of child COVID cases since the start of the pandemic at 15.2 million as of Dec. 29, while the CDC reports 16.2 million cases as of Dec. 28.
There have been 1,975 deaths from COVID-19 in children aged 0-17 years, according to the CDC, which amounts to just over 0.2% of all COVID deaths for which age group data were available.
CDC data on emergency department visits involving diagnosed COVID-19 have been rising since late October. In children aged 0-11 years, for example, COVID was involved in 1.0% of ED visits (7-day average) as late as Nov. 4, but by Dec. 27 that rate was 2.6%. Children aged 12-15 years went from 0.6% on Oct. 28 to 1.5% on Dec. 27, while 16- to 17-year-olds had ED visit rates of 0.6% on Oct. 19 and 1.7% on Dec. 27, the CDC said on its COVID Data Tracker.
New hospital admissions with diagnosed COVID, which had been following the same upward trend as ED visits since late October, halted that rise in children aged 0-17 years and have gone no higher than 0.29 per 100,000 population since Dec. 9, the CDC data show.
The end of 2022 saw a drop in new COVID-19 cases in children, even as rates of emergency department visits continued upward trends that began in late October.
New cases for the week of Dec. 23-29 fell for the first time since late November, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The AAP/CHA analysis of publicly available state data differs somewhat from figures reported by the Centers for Disease Control and Prevention, which has new cases for the latest available week, Dec.18-24, at just over 27,000 after 3 straight weeks of declines from a count of almost 63,000 for the week ending Nov. 26. The CDC, however, updates previously reported data on a regular basis, so that 27,000 is likely to increase in the coming weeks.
The CDC line on the graph also shows a peak for the week of Oct. 30 to Nov. 5 when new cases reached almost 50,000, compared with almost 30,000 reported for the week of Oct. 28 to Nov. 3 by the AAP and CHA in their report of state-level data. The AAP and CHA put the total number of child COVID cases since the start of the pandemic at 15.2 million as of Dec. 29, while the CDC reports 16.2 million cases as of Dec. 28.
There have been 1,975 deaths from COVID-19 in children aged 0-17 years, according to the CDC, which amounts to just over 0.2% of all COVID deaths for which age group data were available.
CDC data on emergency department visits involving diagnosed COVID-19 have been rising since late October. In children aged 0-11 years, for example, COVID was involved in 1.0% of ED visits (7-day average) as late as Nov. 4, but by Dec. 27 that rate was 2.6%. Children aged 12-15 years went from 0.6% on Oct. 28 to 1.5% on Dec. 27, while 16- to 17-year-olds had ED visit rates of 0.6% on Oct. 19 and 1.7% on Dec. 27, the CDC said on its COVID Data Tracker.
New hospital admissions with diagnosed COVID, which had been following the same upward trend as ED visits since late October, halted that rise in children aged 0-17 years and have gone no higher than 0.29 per 100,000 population since Dec. 9, the CDC data show.
Latent TB: The case for vigilance
The US Preventive Services Task Force (USPSTF) recently released draft recommendations on screening for tuberculosis (TB).1 The USPSTF continues to recommend screening for latent TB infection (LTBI) in those at high risk.
Why is this important? Up to one-quarter of the world’s population has been infected with TB, according to World Health Organization (WHO) estimates. In 2021, active TB was diagnosed in 10.6 million people, and it caused 1.6 million deaths.2 Worldwide, TB is still a major cause of mortality: It is the 13th leading cause of death and is the leading cause of infectious disease mortality in non-COVID years.
Although the rate of active TB in the United States has been declining for decades (from 30.7/100,000 in 1960 to 2.4/100,000 in 2021), 7882 cases were reported in 2021, and an estimated 13 million people in the United States have LTBI.3 If not treated, 5% to 10% of LTBI cases will progress to active TB. This risk is higher in those with certain medical conditions.3 People born outside the United States currently account for 71.4% of reported TB cases in the United States.3
To reduce the morbidity and mortality of TB, the Centers for Disease Control and Prevention (CDC), WHO, and USPSTF all recommend screening for and treating LTBI. An effective approach to TB control also includes early detection and completion of treatment for active TB, as well as testing contacts of active TB cases.
Who should be screened? Those at high risk for LTBI include those who were born in, or who have resided in, countries with high rates of TB (eg, Latin America, the Caribbean, Africa, Asia, Eastern Europe, and Russia); those who have lived in a correctional facility or homeless shelter; household and other close contacts of active TB cases; and health care workers who provide care to patients with TB.
Some chronic medical conditions can increase risk for progression to active TB in those with LTBI. Patients who should be tested for LTBI as part of their routine care include those who are HIV positive; are receiving immunosuppressive therapy (chemotherapy, biological immune suppressants); have received an organ transplant; have silicosis; use illicit injected drugs; and/or have had a gastrectomy or jejunoileal bypass.
In addition, local communities may have populations or geographic regions in which TB rates are high. Family physicians can obtain this information from their state or local health departments.
There are 2 screening tests for LTBI: TB blood tests (interferon-gamma release assays [IGRAs]) and the Mantoux tuberculin skin test (TST). Two TB blood tests are available in the United States: QuantiFERON-TB Gold Plus (QFT-Plus) and T-SPOT.TB test (T-Spot).
There are advantages and disadvantages to both types of tests. A TST requires accurate administration and interpretation and 2 clinic visits, 48 to 72 hours apart. The cutoff on a positive test (5, 10, or 15 mm) depends on the patient’s age and risk.4 An IGRA should be processed within 8 to 32 hours and is more expensive. However, a major advantage is that it is more specific, because it is unaffected by previous vaccination with bacille Calmette-Guérin or by most nontuberculous mycobacteria infections.
To rule out active TB ... If a TB screening test is positive, the recommended work-up is to ask about TB symptoms and perform a chest x-ray to rule out active pulmonary TB. Sputum collection for acid-fast smear and culture should be ordered for anyone with a suspicious chest x-ray, respiratory symptoms consistent with TB, or HIV infection.
Treatment for LTBI markedly reduces the risk for active TB. There are 4 options:
- Isoniazid (INH) plus rifapentine (RPT) once per week for 3 months.
- Rifampin (RIF) daily for 4 months.
- INH plus RIF daily for 3 months.
- INH daily for 6 or 9 months.
Details about the variables to consider in choosing a regimen are described on the CDC website.4,5
Know your resources. Local and state public health departments should have TB control programs and are sources of information on TB diagnosis and treatment; they also can assist with follow-up of TB contacts.6 Although LTBI is a reportable condition only in young children, any suspicion of community spread of active TB should be reported to the public health department.
1. USPSTF. Latent tuberculosis infection in adults: screening. Draft recommendation statement. Published November 22, 2022. Accessed December 14, 2022. www.uspreventiveservicestaskforce.org/uspstf/draft-recommendation/latent-tuberculosis-infection-adults
2. WHO. Tuberculosis: key facts. Updated October 27, 2022. Accessed December 14, 2022. www.who.int/news-room/fact-sheets/detail/tuberculosis
3. CDC. Tuberculosis: data and statistics. Updated November 29, 2022. Accessed December 14, 2022. www.cdc.gov/tb/statistics/default.htm
4. CDC. Latent TB infection: a guide for primary health care providers. Updated February 3, 2021. Accessed December 14, 2022. www.cdc.gov/tb/publications/ltbi/pdf/LTBIbooklet508.pdf
5. CDC. Treatment regimens for latent TB infection. Updated February 13, 2020. Accessed December 14, 2022. www.cdc.gov/tb/topic/treatment/ltbi.htm
6. CDC. TB control offices. Updated March 28, 2022. Accessed December 14, 2022. www.cdc.gov/tb/links/tboffices.htm
The US Preventive Services Task Force (USPSTF) recently released draft recommendations on screening for tuberculosis (TB).1 The USPSTF continues to recommend screening for latent TB infection (LTBI) in those at high risk.
Why is this important? Up to one-quarter of the world’s population has been infected with TB, according to World Health Organization (WHO) estimates. In 2021, active TB was diagnosed in 10.6 million people, and it caused 1.6 million deaths.2 Worldwide, TB is still a major cause of mortality: It is the 13th leading cause of death and is the leading cause of infectious disease mortality in non-COVID years.
Although the rate of active TB in the United States has been declining for decades (from 30.7/100,000 in 1960 to 2.4/100,000 in 2021), 7882 cases were reported in 2021, and an estimated 13 million people in the United States have LTBI.3 If not treated, 5% to 10% of LTBI cases will progress to active TB. This risk is higher in those with certain medical conditions.3 People born outside the United States currently account for 71.4% of reported TB cases in the United States.3
To reduce the morbidity and mortality of TB, the Centers for Disease Control and Prevention (CDC), WHO, and USPSTF all recommend screening for and treating LTBI. An effective approach to TB control also includes early detection and completion of treatment for active TB, as well as testing contacts of active TB cases.
Who should be screened? Those at high risk for LTBI include those who were born in, or who have resided in, countries with high rates of TB (eg, Latin America, the Caribbean, Africa, Asia, Eastern Europe, and Russia); those who have lived in a correctional facility or homeless shelter; household and other close contacts of active TB cases; and health care workers who provide care to patients with TB.
Some chronic medical conditions can increase risk for progression to active TB in those with LTBI. Patients who should be tested for LTBI as part of their routine care include those who are HIV positive; are receiving immunosuppressive therapy (chemotherapy, biological immune suppressants); have received an organ transplant; have silicosis; use illicit injected drugs; and/or have had a gastrectomy or jejunoileal bypass.
In addition, local communities may have populations or geographic regions in which TB rates are high. Family physicians can obtain this information from their state or local health departments.
There are 2 screening tests for LTBI: TB blood tests (interferon-gamma release assays [IGRAs]) and the Mantoux tuberculin skin test (TST). Two TB blood tests are available in the United States: QuantiFERON-TB Gold Plus (QFT-Plus) and T-SPOT.TB test (T-Spot).
There are advantages and disadvantages to both types of tests. A TST requires accurate administration and interpretation and 2 clinic visits, 48 to 72 hours apart. The cutoff on a positive test (5, 10, or 15 mm) depends on the patient’s age and risk.4 An IGRA should be processed within 8 to 32 hours and is more expensive. However, a major advantage is that it is more specific, because it is unaffected by previous vaccination with bacille Calmette-Guérin or by most nontuberculous mycobacteria infections.
To rule out active TB ... If a TB screening test is positive, the recommended work-up is to ask about TB symptoms and perform a chest x-ray to rule out active pulmonary TB. Sputum collection for acid-fast smear and culture should be ordered for anyone with a suspicious chest x-ray, respiratory symptoms consistent with TB, or HIV infection.
Treatment for LTBI markedly reduces the risk for active TB. There are 4 options:
- Isoniazid (INH) plus rifapentine (RPT) once per week for 3 months.
- Rifampin (RIF) daily for 4 months.
- INH plus RIF daily for 3 months.
- INH daily for 6 or 9 months.
Details about the variables to consider in choosing a regimen are described on the CDC website.4,5
Know your resources. Local and state public health departments should have TB control programs and are sources of information on TB diagnosis and treatment; they also can assist with follow-up of TB contacts.6 Although LTBI is a reportable condition only in young children, any suspicion of community spread of active TB should be reported to the public health department.
The US Preventive Services Task Force (USPSTF) recently released draft recommendations on screening for tuberculosis (TB).1 The USPSTF continues to recommend screening for latent TB infection (LTBI) in those at high risk.
Why is this important? Up to one-quarter of the world’s population has been infected with TB, according to World Health Organization (WHO) estimates. In 2021, active TB was diagnosed in 10.6 million people, and it caused 1.6 million deaths.2 Worldwide, TB is still a major cause of mortality: It is the 13th leading cause of death and is the leading cause of infectious disease mortality in non-COVID years.
Although the rate of active TB in the United States has been declining for decades (from 30.7/100,000 in 1960 to 2.4/100,000 in 2021), 7882 cases were reported in 2021, and an estimated 13 million people in the United States have LTBI.3 If not treated, 5% to 10% of LTBI cases will progress to active TB. This risk is higher in those with certain medical conditions.3 People born outside the United States currently account for 71.4% of reported TB cases in the United States.3
To reduce the morbidity and mortality of TB, the Centers for Disease Control and Prevention (CDC), WHO, and USPSTF all recommend screening for and treating LTBI. An effective approach to TB control also includes early detection and completion of treatment for active TB, as well as testing contacts of active TB cases.
Who should be screened? Those at high risk for LTBI include those who were born in, or who have resided in, countries with high rates of TB (eg, Latin America, the Caribbean, Africa, Asia, Eastern Europe, and Russia); those who have lived in a correctional facility or homeless shelter; household and other close contacts of active TB cases; and health care workers who provide care to patients with TB.
Some chronic medical conditions can increase risk for progression to active TB in those with LTBI. Patients who should be tested for LTBI as part of their routine care include those who are HIV positive; are receiving immunosuppressive therapy (chemotherapy, biological immune suppressants); have received an organ transplant; have silicosis; use illicit injected drugs; and/or have had a gastrectomy or jejunoileal bypass.
In addition, local communities may have populations or geographic regions in which TB rates are high. Family physicians can obtain this information from their state or local health departments.
There are 2 screening tests for LTBI: TB blood tests (interferon-gamma release assays [IGRAs]) and the Mantoux tuberculin skin test (TST). Two TB blood tests are available in the United States: QuantiFERON-TB Gold Plus (QFT-Plus) and T-SPOT.TB test (T-Spot).
There are advantages and disadvantages to both types of tests. A TST requires accurate administration and interpretation and 2 clinic visits, 48 to 72 hours apart. The cutoff on a positive test (5, 10, or 15 mm) depends on the patient’s age and risk.4 An IGRA should be processed within 8 to 32 hours and is more expensive. However, a major advantage is that it is more specific, because it is unaffected by previous vaccination with bacille Calmette-Guérin or by most nontuberculous mycobacteria infections.
To rule out active TB ... If a TB screening test is positive, the recommended work-up is to ask about TB symptoms and perform a chest x-ray to rule out active pulmonary TB. Sputum collection for acid-fast smear and culture should be ordered for anyone with a suspicious chest x-ray, respiratory symptoms consistent with TB, or HIV infection.
Treatment for LTBI markedly reduces the risk for active TB. There are 4 options:
- Isoniazid (INH) plus rifapentine (RPT) once per week for 3 months.
- Rifampin (RIF) daily for 4 months.
- INH plus RIF daily for 3 months.
- INH daily for 6 or 9 months.
Details about the variables to consider in choosing a regimen are described on the CDC website.4,5
Know your resources. Local and state public health departments should have TB control programs and are sources of information on TB diagnosis and treatment; they also can assist with follow-up of TB contacts.6 Although LTBI is a reportable condition only in young children, any suspicion of community spread of active TB should be reported to the public health department.
1. USPSTF. Latent tuberculosis infection in adults: screening. Draft recommendation statement. Published November 22, 2022. Accessed December 14, 2022. www.uspreventiveservicestaskforce.org/uspstf/draft-recommendation/latent-tuberculosis-infection-adults
2. WHO. Tuberculosis: key facts. Updated October 27, 2022. Accessed December 14, 2022. www.who.int/news-room/fact-sheets/detail/tuberculosis
3. CDC. Tuberculosis: data and statistics. Updated November 29, 2022. Accessed December 14, 2022. www.cdc.gov/tb/statistics/default.htm
4. CDC. Latent TB infection: a guide for primary health care providers. Updated February 3, 2021. Accessed December 14, 2022. www.cdc.gov/tb/publications/ltbi/pdf/LTBIbooklet508.pdf
5. CDC. Treatment regimens for latent TB infection. Updated February 13, 2020. Accessed December 14, 2022. www.cdc.gov/tb/topic/treatment/ltbi.htm
6. CDC. TB control offices. Updated March 28, 2022. Accessed December 14, 2022. www.cdc.gov/tb/links/tboffices.htm
1. USPSTF. Latent tuberculosis infection in adults: screening. Draft recommendation statement. Published November 22, 2022. Accessed December 14, 2022. www.uspreventiveservicestaskforce.org/uspstf/draft-recommendation/latent-tuberculosis-infection-adults
2. WHO. Tuberculosis: key facts. Updated October 27, 2022. Accessed December 14, 2022. www.who.int/news-room/fact-sheets/detail/tuberculosis
3. CDC. Tuberculosis: data and statistics. Updated November 29, 2022. Accessed December 14, 2022. www.cdc.gov/tb/statistics/default.htm
4. CDC. Latent TB infection: a guide for primary health care providers. Updated February 3, 2021. Accessed December 14, 2022. www.cdc.gov/tb/publications/ltbi/pdf/LTBIbooklet508.pdf
5. CDC. Treatment regimens for latent TB infection. Updated February 13, 2020. Accessed December 14, 2022. www.cdc.gov/tb/topic/treatment/ltbi.htm
6. CDC. TB control offices. Updated March 28, 2022. Accessed December 14, 2022. www.cdc.gov/tb/links/tboffices.htm
Atypical Keratotic Nodule on the Knuckle
The Diagnosis: Atypical Mycobacterial Infection
The history of rapid growth followed by shrinkage as well as the craterlike clinical appearance of our patient’s lesion were suspicious for the keratoacanthoma variant of squamous cell carcinoma (SCC). Periodic acid–Schiff green staining was negative for fungal or bacterial organisms, and the biopsy findings of keratinocyte atypia and irregular epidermal proliferation seemed to confirm our suspicion for well-differentiated SCC (Figure 1). Our patient subsequently was scheduled for Mohs micrographic surgery. Fortunately, a sample of tissue had been sent for panculture—bacterial, fungal, and mycobacterial—to rule out infectious etiologies, given the history of possible traumatic inoculation, and returned positive for Mycobacterium marinum infection prior to the surgery. Mohs surgery was canceled, and he was referred to an infectious disease specialist who started antibiotic treatment with azithromycin, ethambutol, and rifabutin. After 1 month of treatment the lesion substantially improved (Figure 2), further supporting the diagnosis of M marinum infection over SCC.
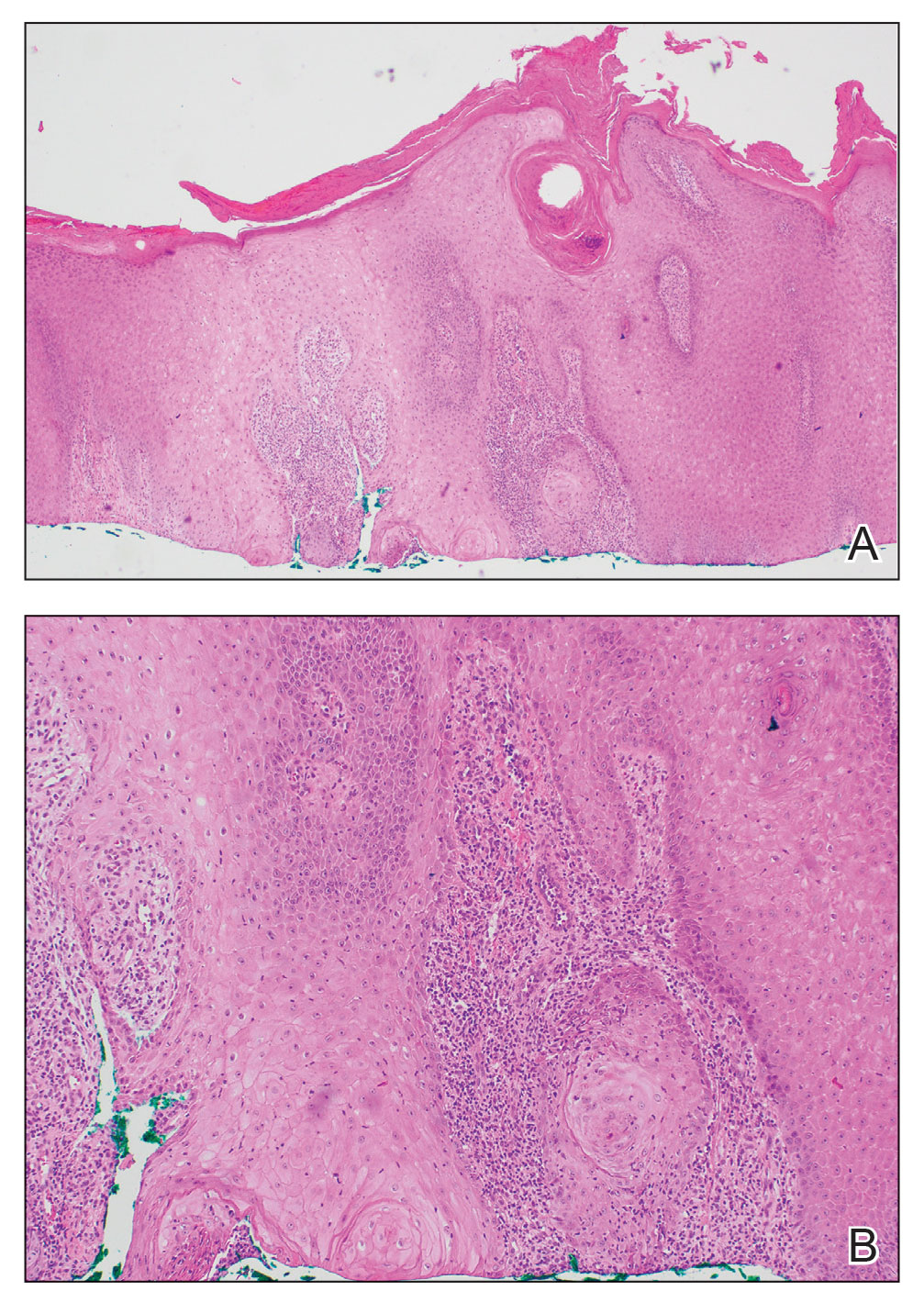
The differential diagnosis also included sporotrichosis, leishmaniasis, and chromoblastomycosis. Sporotrichosis lesions typically develop as multiple nodules and ulcers along a path of lymphatic drainage and can exhibit asteroid bodies and cigar-shaped yeast forms on histology. Chromoblastomycosis may display pseudoepitheliomatous hyperplasia and granulomatous inflammation; however, pathognomonic pigmented Medlar bodies also likely would be present.1 Leishmaniasis has a wide variety of presentations; however, it typically occurs in patients with exposure to endemic areas outside of the United States. Although leishmaniasis may demonstrate pseudoepitheliomatous hyperplasia, ulceration, and mixed inflammation on histology, it also likely would show amastigotes within dermal macrophages.2
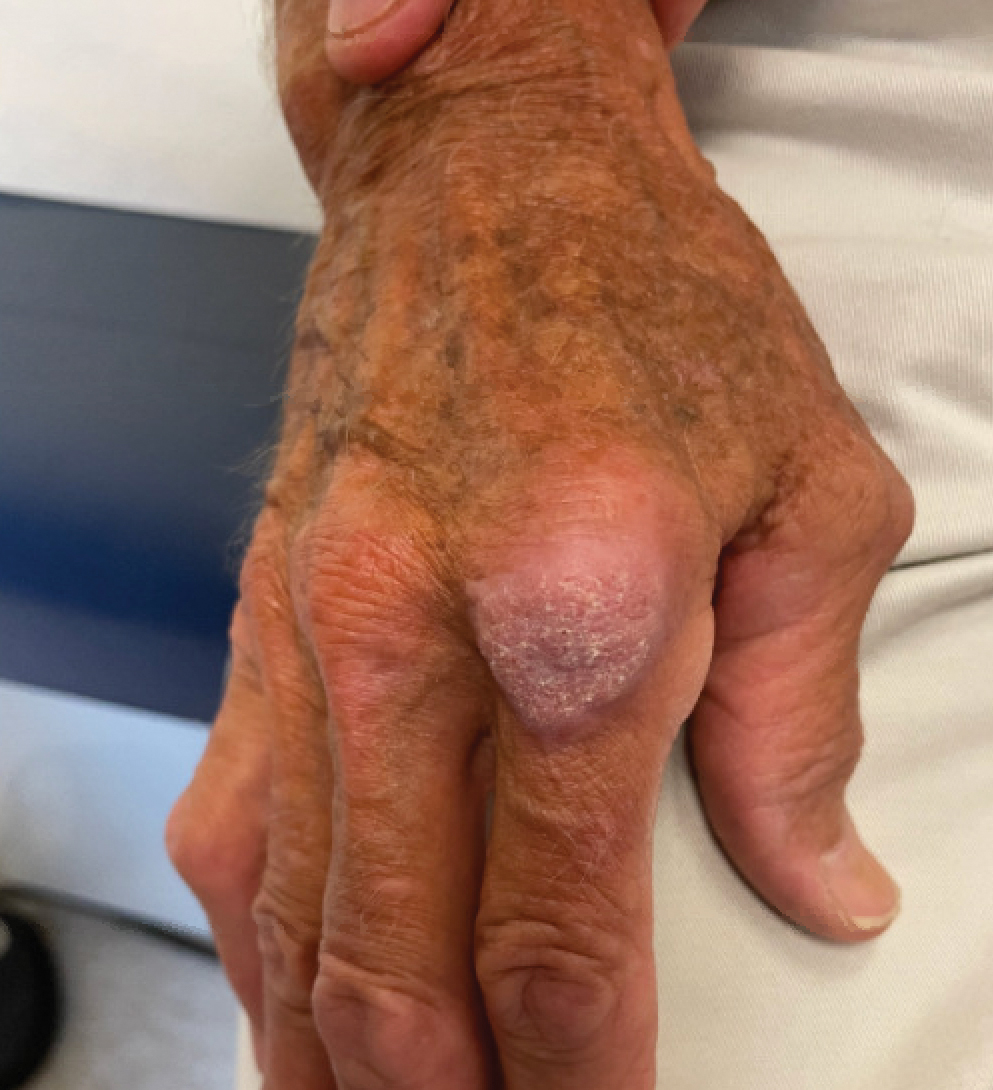
Atypical mycobacterial infections initially may be misdiagnosed as SCC due to their tendency to induce irregular acanthosis in the form of pseudoepitheliomatous hyperplasia as well as mild keratinocyte atypia secondary to inflammation.3,4 Our case is unique because it occurred with M marinum infection specifically. The histopathologic findings of M marinum infections are variable and may additionally include granulomas, most commonly suppurative; intraepithelial abscesses; small vessel proliferation; dermal fibrosis; multinucleated giant cells; and transepidermal elimination.4,5 Periodic acid–Schiff, Ziehl-Neelsen (acid-fast bacilli), and Fite staining may be used to distinguish M marinum infection from SCC but have low sensitivities (approximately 30%). Culture remains the most reliable test, with a sensitivity of nearly 80%.5-7 In our patient, a Periodic acid–Schiff stain was obtained prior to receiving culture results, and acid-fast bacilli and Fite staining were added after the culture returned positive; however, all 3 stains failed to highlight any mycobacteria.
The primary risk factor for infection with M marinum is contact with aquatic environments or marine animals, and most cases involve the fingers or the hand.6 After we reached the diagnosis and further discussed the patient’s history, he recalled fishing for and cleaning raw shrimp around the time that he had a splinter. The Infectious Diseases Society of America recommends a treatment course extending 1 to 2 months after clinical symptoms resolve with ethambutol in addition to clarithromycin or azithromycin.8 If the infection is near a joint, rifampin should be empirically added to account for a potentially deeper infection. Imaging should be obtained to evaluate for joint space involvement, with magnetic resonance imaging being the preferred modality. If joint space involvement is confirmed, surgical debridement is indicated. Surgical debridement also is indicated for infections that fail to respond to antibiotic therapy.8
This case highlights M marinum infection as a potential mimicker of SCC, particularly if the biopsy is relatively superficial, as often occurs when obtained via the common shave technique. The distinction is critical, as M marinum infection is highly treatable and inappropriate surgery on the typical hand and finger locations may subject patients to substantial morbidity, such as the need for a skin graft, reduced mobility from scarring, or risk for serious wound infection.9 For superficial biopsies of an atypical squamous process, pathologists also may consider routinely recommending tissue culture, especially for hand and finger locations or when a history of local trauma is reported, instead of recommending complete excision or repeat biopsy alone.
- Elewski BE, Hughey LC, Hunt KM, et al. Fungal diseases. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1329-1363.
- Bravo FG. Protozoa and worms. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1470-1502.
- Zayour M, Lazova R. Pseudoepitheliomatous hyperplasia: a review. Am J Dermatopathol. 2011;33:112-122; quiz 123-126. doi:10.1097 /DAD.0b013e3181fcfb47
- Li JJ, Beresford R, Fyfe J, et al. Clinical and histopathological features of cutaneous nontuberculous mycobacterial infection: a review of 13 cases. J Cutan Pathol. 2017;44:433-443. doi:10.1111/cup.12903
- Abbas O, Marrouch N, Kattar MM, et al. Cutaneous non-tuberculous mycobacterial infections: a clinical and histopathological study of 17 cases from Lebanon. J Eur Acad Dermatol Venereol. 2011;25:33-42. doi:10.1111/j.1468-3083.2010.03684.x
- Johnson MG, Stout JE. Twenty-eight cases of Mycobacterium marinum infection: retrospective case series and literature review. Infection. 2015;43:655-662. doi:10.1007/s15010-015-0776-8
- Aubry A, Mougari F, Reibel F, et al. Mycobacterium marinum. Microbiol Spectr. 2017;5. doi:10.1128/microbiolspec.TNMI7-0038-2016
- Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367-416. doi:10.1164/rccm.200604-571ST
- Alam M, Ibrahim O, Nodzenski M, et al. Adverse events associated with Mohs micrographic surgery: multicenter prospective cohort study of 20,821 cases at 23 centers. JAMA Dermatol. 2013;149:1378-1385. doi:10.1001/jamadermatol.2013.6255
The Diagnosis: Atypical Mycobacterial Infection
The history of rapid growth followed by shrinkage as well as the craterlike clinical appearance of our patient’s lesion were suspicious for the keratoacanthoma variant of squamous cell carcinoma (SCC). Periodic acid–Schiff green staining was negative for fungal or bacterial organisms, and the biopsy findings of keratinocyte atypia and irregular epidermal proliferation seemed to confirm our suspicion for well-differentiated SCC (Figure 1). Our patient subsequently was scheduled for Mohs micrographic surgery. Fortunately, a sample of tissue had been sent for panculture—bacterial, fungal, and mycobacterial—to rule out infectious etiologies, given the history of possible traumatic inoculation, and returned positive for Mycobacterium marinum infection prior to the surgery. Mohs surgery was canceled, and he was referred to an infectious disease specialist who started antibiotic treatment with azithromycin, ethambutol, and rifabutin. After 1 month of treatment the lesion substantially improved (Figure 2), further supporting the diagnosis of M marinum infection over SCC.

The differential diagnosis also included sporotrichosis, leishmaniasis, and chromoblastomycosis. Sporotrichosis lesions typically develop as multiple nodules and ulcers along a path of lymphatic drainage and can exhibit asteroid bodies and cigar-shaped yeast forms on histology. Chromoblastomycosis may display pseudoepitheliomatous hyperplasia and granulomatous inflammation; however, pathognomonic pigmented Medlar bodies also likely would be present.1 Leishmaniasis has a wide variety of presentations; however, it typically occurs in patients with exposure to endemic areas outside of the United States. Although leishmaniasis may demonstrate pseudoepitheliomatous hyperplasia, ulceration, and mixed inflammation on histology, it also likely would show amastigotes within dermal macrophages.2

Atypical mycobacterial infections initially may be misdiagnosed as SCC due to their tendency to induce irregular acanthosis in the form of pseudoepitheliomatous hyperplasia as well as mild keratinocyte atypia secondary to inflammation.3,4 Our case is unique because it occurred with M marinum infection specifically. The histopathologic findings of M marinum infections are variable and may additionally include granulomas, most commonly suppurative; intraepithelial abscesses; small vessel proliferation; dermal fibrosis; multinucleated giant cells; and transepidermal elimination.4,5 Periodic acid–Schiff, Ziehl-Neelsen (acid-fast bacilli), and Fite staining may be used to distinguish M marinum infection from SCC but have low sensitivities (approximately 30%). Culture remains the most reliable test, with a sensitivity of nearly 80%.5-7 In our patient, a Periodic acid–Schiff stain was obtained prior to receiving culture results, and acid-fast bacilli and Fite staining were added after the culture returned positive; however, all 3 stains failed to highlight any mycobacteria.
The primary risk factor for infection with M marinum is contact with aquatic environments or marine animals, and most cases involve the fingers or the hand.6 After we reached the diagnosis and further discussed the patient’s history, he recalled fishing for and cleaning raw shrimp around the time that he had a splinter. The Infectious Diseases Society of America recommends a treatment course extending 1 to 2 months after clinical symptoms resolve with ethambutol in addition to clarithromycin or azithromycin.8 If the infection is near a joint, rifampin should be empirically added to account for a potentially deeper infection. Imaging should be obtained to evaluate for joint space involvement, with magnetic resonance imaging being the preferred modality. If joint space involvement is confirmed, surgical debridement is indicated. Surgical debridement also is indicated for infections that fail to respond to antibiotic therapy.8
This case highlights M marinum infection as a potential mimicker of SCC, particularly if the biopsy is relatively superficial, as often occurs when obtained via the common shave technique. The distinction is critical, as M marinum infection is highly treatable and inappropriate surgery on the typical hand and finger locations may subject patients to substantial morbidity, such as the need for a skin graft, reduced mobility from scarring, or risk for serious wound infection.9 For superficial biopsies of an atypical squamous process, pathologists also may consider routinely recommending tissue culture, especially for hand and finger locations or when a history of local trauma is reported, instead of recommending complete excision or repeat biopsy alone.
The Diagnosis: Atypical Mycobacterial Infection
The history of rapid growth followed by shrinkage as well as the craterlike clinical appearance of our patient’s lesion were suspicious for the keratoacanthoma variant of squamous cell carcinoma (SCC). Periodic acid–Schiff green staining was negative for fungal or bacterial organisms, and the biopsy findings of keratinocyte atypia and irregular epidermal proliferation seemed to confirm our suspicion for well-differentiated SCC (Figure 1). Our patient subsequently was scheduled for Mohs micrographic surgery. Fortunately, a sample of tissue had been sent for panculture—bacterial, fungal, and mycobacterial—to rule out infectious etiologies, given the history of possible traumatic inoculation, and returned positive for Mycobacterium marinum infection prior to the surgery. Mohs surgery was canceled, and he was referred to an infectious disease specialist who started antibiotic treatment with azithromycin, ethambutol, and rifabutin. After 1 month of treatment the lesion substantially improved (Figure 2), further supporting the diagnosis of M marinum infection over SCC.

The differential diagnosis also included sporotrichosis, leishmaniasis, and chromoblastomycosis. Sporotrichosis lesions typically develop as multiple nodules and ulcers along a path of lymphatic drainage and can exhibit asteroid bodies and cigar-shaped yeast forms on histology. Chromoblastomycosis may display pseudoepitheliomatous hyperplasia and granulomatous inflammation; however, pathognomonic pigmented Medlar bodies also likely would be present.1 Leishmaniasis has a wide variety of presentations; however, it typically occurs in patients with exposure to endemic areas outside of the United States. Although leishmaniasis may demonstrate pseudoepitheliomatous hyperplasia, ulceration, and mixed inflammation on histology, it also likely would show amastigotes within dermal macrophages.2

Atypical mycobacterial infections initially may be misdiagnosed as SCC due to their tendency to induce irregular acanthosis in the form of pseudoepitheliomatous hyperplasia as well as mild keratinocyte atypia secondary to inflammation.3,4 Our case is unique because it occurred with M marinum infection specifically. The histopathologic findings of M marinum infections are variable and may additionally include granulomas, most commonly suppurative; intraepithelial abscesses; small vessel proliferation; dermal fibrosis; multinucleated giant cells; and transepidermal elimination.4,5 Periodic acid–Schiff, Ziehl-Neelsen (acid-fast bacilli), and Fite staining may be used to distinguish M marinum infection from SCC but have low sensitivities (approximately 30%). Culture remains the most reliable test, with a sensitivity of nearly 80%.5-7 In our patient, a Periodic acid–Schiff stain was obtained prior to receiving culture results, and acid-fast bacilli and Fite staining were added after the culture returned positive; however, all 3 stains failed to highlight any mycobacteria.
The primary risk factor for infection with M marinum is contact with aquatic environments or marine animals, and most cases involve the fingers or the hand.6 After we reached the diagnosis and further discussed the patient’s history, he recalled fishing for and cleaning raw shrimp around the time that he had a splinter. The Infectious Diseases Society of America recommends a treatment course extending 1 to 2 months after clinical symptoms resolve with ethambutol in addition to clarithromycin or azithromycin.8 If the infection is near a joint, rifampin should be empirically added to account for a potentially deeper infection. Imaging should be obtained to evaluate for joint space involvement, with magnetic resonance imaging being the preferred modality. If joint space involvement is confirmed, surgical debridement is indicated. Surgical debridement also is indicated for infections that fail to respond to antibiotic therapy.8
This case highlights M marinum infection as a potential mimicker of SCC, particularly if the biopsy is relatively superficial, as often occurs when obtained via the common shave technique. The distinction is critical, as M marinum infection is highly treatable and inappropriate surgery on the typical hand and finger locations may subject patients to substantial morbidity, such as the need for a skin graft, reduced mobility from scarring, or risk for serious wound infection.9 For superficial biopsies of an atypical squamous process, pathologists also may consider routinely recommending tissue culture, especially for hand and finger locations or when a history of local trauma is reported, instead of recommending complete excision or repeat biopsy alone.
- Elewski BE, Hughey LC, Hunt KM, et al. Fungal diseases. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1329-1363.
- Bravo FG. Protozoa and worms. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1470-1502.
- Zayour M, Lazova R. Pseudoepitheliomatous hyperplasia: a review. Am J Dermatopathol. 2011;33:112-122; quiz 123-126. doi:10.1097 /DAD.0b013e3181fcfb47
- Li JJ, Beresford R, Fyfe J, et al. Clinical and histopathological features of cutaneous nontuberculous mycobacterial infection: a review of 13 cases. J Cutan Pathol. 2017;44:433-443. doi:10.1111/cup.12903
- Abbas O, Marrouch N, Kattar MM, et al. Cutaneous non-tuberculous mycobacterial infections: a clinical and histopathological study of 17 cases from Lebanon. J Eur Acad Dermatol Venereol. 2011;25:33-42. doi:10.1111/j.1468-3083.2010.03684.x
- Johnson MG, Stout JE. Twenty-eight cases of Mycobacterium marinum infection: retrospective case series and literature review. Infection. 2015;43:655-662. doi:10.1007/s15010-015-0776-8
- Aubry A, Mougari F, Reibel F, et al. Mycobacterium marinum. Microbiol Spectr. 2017;5. doi:10.1128/microbiolspec.TNMI7-0038-2016
- Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367-416. doi:10.1164/rccm.200604-571ST
- Alam M, Ibrahim O, Nodzenski M, et al. Adverse events associated with Mohs micrographic surgery: multicenter prospective cohort study of 20,821 cases at 23 centers. JAMA Dermatol. 2013;149:1378-1385. doi:10.1001/jamadermatol.2013.6255
- Elewski BE, Hughey LC, Hunt KM, et al. Fungal diseases. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1329-1363.
- Bravo FG. Protozoa and worms. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1470-1502.
- Zayour M, Lazova R. Pseudoepitheliomatous hyperplasia: a review. Am J Dermatopathol. 2011;33:112-122; quiz 123-126. doi:10.1097 /DAD.0b013e3181fcfb47
- Li JJ, Beresford R, Fyfe J, et al. Clinical and histopathological features of cutaneous nontuberculous mycobacterial infection: a review of 13 cases. J Cutan Pathol. 2017;44:433-443. doi:10.1111/cup.12903
- Abbas O, Marrouch N, Kattar MM, et al. Cutaneous non-tuberculous mycobacterial infections: a clinical and histopathological study of 17 cases from Lebanon. J Eur Acad Dermatol Venereol. 2011;25:33-42. doi:10.1111/j.1468-3083.2010.03684.x
- Johnson MG, Stout JE. Twenty-eight cases of Mycobacterium marinum infection: retrospective case series and literature review. Infection. 2015;43:655-662. doi:10.1007/s15010-015-0776-8
- Aubry A, Mougari F, Reibel F, et al. Mycobacterium marinum. Microbiol Spectr. 2017;5. doi:10.1128/microbiolspec.TNMI7-0038-2016
- Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367-416. doi:10.1164/rccm.200604-571ST
- Alam M, Ibrahim O, Nodzenski M, et al. Adverse events associated with Mohs micrographic surgery: multicenter prospective cohort study of 20,821 cases at 23 centers. JAMA Dermatol. 2013;149:1378-1385. doi:10.1001/jamadermatol.2013.6255
A 75-year-old man presented with a lesion on the knuckle of 5 months’ duration. He reported that the lesion initially grew very quickly before shrinking down to its current size. He denied any bleeding or pain but thought he may have had a splinter in the area around the time the lesion appeared. He reported spending a lot of time outdoors and noted several recent insect and tick bites. He also owned a boat and frequently went fishing. He previously had been treated for actinic keratoses but had no history of skin cancer and no family history of melanoma. Physical examination revealed a 2-cm erythematous nodule with central hyperkeratosis overlying the metacarpophalangeal joint of the right index finger. A shave biopsy was performed.