User login
Two Brain Stim Methods Better Than One for Depression?
TOPLINE:
a new study showed.
METHODOLOGY:
- Researchers conducted a double-blind, sham-controlled randomized clinical trial from 2021 to 2023 at three hospitals in China with 240 participants with MDD (mean age, 32.5 years; 58% women).
- Participants received active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, or sham tDCS + sham rTMS with treatments administered five times per week for 2 weeks.
- tDCS was administered in 20-minute sessions using a 2-mA direct current stimulator, whereas rTMS involved 1600 pulses of 10-Hz stimulation targeting the left dorsolateral prefrontal cortex. Sham treatments used a pseudostimulation coil and only emitted sound.
- The primary outcome was change in the 24-item Hamilton Depression Rating Scale (HDRS-24) total score from baseline to week 2.
- Secondary outcomes included HDRS-24 total score change at week 4, remission rate (HDRS-24 total score ≤ 9), response rate (≥ 50% reduction in HDRS-24 total score), and adverse events.
TAKEAWAY:
- The active tDCS + active rTMS group demonstrated the greatest reduction in mean HDRS-24 score (18.33 ± 5.39) at week 2 compared with sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS (P < .001).
- Response rates at week 2 were notably higher in the active tDCS + active rTMS group (85%) than in the active tDCS + sham rTMS (30%) and sham tDCS + sham rTMS groups (32%).
- The remission rate at week 4 reached 83% in the active tDCS + active rTMS group, which was significantly higher than the remission rates with the other interventions (P < .001).
- The treatments were well tolerated, with no serious adverse events, seizures, or manic symptoms reported across all intervention groups.
IN PRACTICE:
This trial “was the first to evaluate the safety, feasibility, and efficacy of combining tDCS and rTMS in treating depression. Future studies should focus on investigating the mechanism of this synergistic effect and improving the stimulation parameters to optimize the therapeutic effect,” the investigators wrote.
SOURCE:
This study was led by Dongsheng Zhou, MD, Ningbo Kangning Hospital, Ningbo, China. It was published online in JAMA Network Open.
LIMITATIONS:
The brief treatment duration involving 10 sessions may have been insufficient for tDCS and rTMS to demonstrate their full antidepressant potential. The inability to regulate participants’ antidepressant medications throughout the study period presented another limitation. Additionally, the lack of stratified randomization and adjustment for center effects may have introduced variability in the results.
DISCLOSURES:
This study received support from multiple grants, including from the Natural Science Foundation of Zhejiang Province, Basic Public Welfare Research Project of Zhejiang Province, Ningbo Medical and Health Brand Discipline, Ningbo Clinical Medical Research Centre for Mental Health, Ningbo Top Medical and Health Research Program, and the Zhejiang Medical and Health Science and Technology Plan Project. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
a new study showed.
METHODOLOGY:
- Researchers conducted a double-blind, sham-controlled randomized clinical trial from 2021 to 2023 at three hospitals in China with 240 participants with MDD (mean age, 32.5 years; 58% women).
- Participants received active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, or sham tDCS + sham rTMS with treatments administered five times per week for 2 weeks.
- tDCS was administered in 20-minute sessions using a 2-mA direct current stimulator, whereas rTMS involved 1600 pulses of 10-Hz stimulation targeting the left dorsolateral prefrontal cortex. Sham treatments used a pseudostimulation coil and only emitted sound.
- The primary outcome was change in the 24-item Hamilton Depression Rating Scale (HDRS-24) total score from baseline to week 2.
- Secondary outcomes included HDRS-24 total score change at week 4, remission rate (HDRS-24 total score ≤ 9), response rate (≥ 50% reduction in HDRS-24 total score), and adverse events.
TAKEAWAY:
- The active tDCS + active rTMS group demonstrated the greatest reduction in mean HDRS-24 score (18.33 ± 5.39) at week 2 compared with sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS (P < .001).
- Response rates at week 2 were notably higher in the active tDCS + active rTMS group (85%) than in the active tDCS + sham rTMS (30%) and sham tDCS + sham rTMS groups (32%).
- The remission rate at week 4 reached 83% in the active tDCS + active rTMS group, which was significantly higher than the remission rates with the other interventions (P < .001).
- The treatments were well tolerated, with no serious adverse events, seizures, or manic symptoms reported across all intervention groups.
IN PRACTICE:
This trial “was the first to evaluate the safety, feasibility, and efficacy of combining tDCS and rTMS in treating depression. Future studies should focus on investigating the mechanism of this synergistic effect and improving the stimulation parameters to optimize the therapeutic effect,” the investigators wrote.
SOURCE:
This study was led by Dongsheng Zhou, MD, Ningbo Kangning Hospital, Ningbo, China. It was published online in JAMA Network Open.
LIMITATIONS:
The brief treatment duration involving 10 sessions may have been insufficient for tDCS and rTMS to demonstrate their full antidepressant potential. The inability to regulate participants’ antidepressant medications throughout the study period presented another limitation. Additionally, the lack of stratified randomization and adjustment for center effects may have introduced variability in the results.
DISCLOSURES:
This study received support from multiple grants, including from the Natural Science Foundation of Zhejiang Province, Basic Public Welfare Research Project of Zhejiang Province, Ningbo Medical and Health Brand Discipline, Ningbo Clinical Medical Research Centre for Mental Health, Ningbo Top Medical and Health Research Program, and the Zhejiang Medical and Health Science and Technology Plan Project. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
a new study showed.
METHODOLOGY:
- Researchers conducted a double-blind, sham-controlled randomized clinical trial from 2021 to 2023 at three hospitals in China with 240 participants with MDD (mean age, 32.5 years; 58% women).
- Participants received active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, or sham tDCS + sham rTMS with treatments administered five times per week for 2 weeks.
- tDCS was administered in 20-minute sessions using a 2-mA direct current stimulator, whereas rTMS involved 1600 pulses of 10-Hz stimulation targeting the left dorsolateral prefrontal cortex. Sham treatments used a pseudostimulation coil and only emitted sound.
- The primary outcome was change in the 24-item Hamilton Depression Rating Scale (HDRS-24) total score from baseline to week 2.
- Secondary outcomes included HDRS-24 total score change at week 4, remission rate (HDRS-24 total score ≤ 9), response rate (≥ 50% reduction in HDRS-24 total score), and adverse events.
TAKEAWAY:
- The active tDCS + active rTMS group demonstrated the greatest reduction in mean HDRS-24 score (18.33 ± 5.39) at week 2 compared with sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS (P < .001).
- Response rates at week 2 were notably higher in the active tDCS + active rTMS group (85%) than in the active tDCS + sham rTMS (30%) and sham tDCS + sham rTMS groups (32%).
- The remission rate at week 4 reached 83% in the active tDCS + active rTMS group, which was significantly higher than the remission rates with the other interventions (P < .001).
- The treatments were well tolerated, with no serious adverse events, seizures, or manic symptoms reported across all intervention groups.
IN PRACTICE:
This trial “was the first to evaluate the safety, feasibility, and efficacy of combining tDCS and rTMS in treating depression. Future studies should focus on investigating the mechanism of this synergistic effect and improving the stimulation parameters to optimize the therapeutic effect,” the investigators wrote.
SOURCE:
This study was led by Dongsheng Zhou, MD, Ningbo Kangning Hospital, Ningbo, China. It was published online in JAMA Network Open.
LIMITATIONS:
The brief treatment duration involving 10 sessions may have been insufficient for tDCS and rTMS to demonstrate their full antidepressant potential. The inability to regulate participants’ antidepressant medications throughout the study period presented another limitation. Additionally, the lack of stratified randomization and adjustment for center effects may have introduced variability in the results.
DISCLOSURES:
This study received support from multiple grants, including from the Natural Science Foundation of Zhejiang Province, Basic Public Welfare Research Project of Zhejiang Province, Ningbo Medical and Health Brand Discipline, Ningbo Clinical Medical Research Centre for Mental Health, Ningbo Top Medical and Health Research Program, and the Zhejiang Medical and Health Science and Technology Plan Project. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Daytime Sleepiness May Flag Predementia Risk
TOPLINE:
a new study shows.
METHODOLOGY:
- Researchers included 445 older adults without dementia (mean age, 76 years; 57% women).
- Sleep components were assessed, and participants were classified as poor or good sleepers using the Pittsburgh Sleep Quality Index questionnaire.
- The primary outcome was incidence of MCR syndrome.
- The mean follow-up duration was 2.9 years.
TAKEAWAY:
- During the study period, 36 participants developed MCR syndrome.
- Poor sleepers had a higher risk for incident MCR syndrome, compared with good sleepers, after adjustment for age, sex, and educational level (adjusted hazard ratio [aHR], 2.6; 95% CI, 1.3-5.0; P < .05). However, this association was no longer significant after further adjustment for depressive symptoms.
- Sleep-related daytime dysfunction, defined as excessive sleepiness and lower enthusiasm for activities, was the only sleep component linked to a significant risk for MCR syndrome in fully adjusted models (aHR, 3.3; 95% CI, 1.5-7.4; P < .05).
- Prevalent MCR syndrome was not significantly associated with poor sleep quality (odds ratio, 1.1), suggesting that the relationship is unidirectional.
IN PRACTICE:
“Establishing the relationship between sleep dysfunction and MCR [syndrome] risk is important because early intervention may offer the best hope for preventing dementia,” the investigators wrote.
“Our findings emphasize the need for screening for sleep issues. There’s potential that people could get help with their sleep issues and prevent cognitive decline later in life,” lead author Victoire Leroy, MD, PhD, Albert Einstein College of Medicine, New York City, added in a press release.
SOURCE:
The study was published online in Neurology.
LIMITATIONS:
Study limitations included the lack of objective sleep measurements and potential recall bias in self-reported sleep complaints, particularly among participants with cognitive issues. In addition, the relatively short follow-up period may have resulted in a lower number of incident MCR syndrome cases. The sample population was also predominantly White (80%), which may have limited the generalizability of the findings to other populations.
DISCLOSURES:
The study was funded by the National Institute on Aging. No conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
a new study shows.
METHODOLOGY:
- Researchers included 445 older adults without dementia (mean age, 76 years; 57% women).
- Sleep components were assessed, and participants were classified as poor or good sleepers using the Pittsburgh Sleep Quality Index questionnaire.
- The primary outcome was incidence of MCR syndrome.
- The mean follow-up duration was 2.9 years.
TAKEAWAY:
- During the study period, 36 participants developed MCR syndrome.
- Poor sleepers had a higher risk for incident MCR syndrome, compared with good sleepers, after adjustment for age, sex, and educational level (adjusted hazard ratio [aHR], 2.6; 95% CI, 1.3-5.0; P < .05). However, this association was no longer significant after further adjustment for depressive symptoms.
- Sleep-related daytime dysfunction, defined as excessive sleepiness and lower enthusiasm for activities, was the only sleep component linked to a significant risk for MCR syndrome in fully adjusted models (aHR, 3.3; 95% CI, 1.5-7.4; P < .05).
- Prevalent MCR syndrome was not significantly associated with poor sleep quality (odds ratio, 1.1), suggesting that the relationship is unidirectional.
IN PRACTICE:
“Establishing the relationship between sleep dysfunction and MCR [syndrome] risk is important because early intervention may offer the best hope for preventing dementia,” the investigators wrote.
“Our findings emphasize the need for screening for sleep issues. There’s potential that people could get help with their sleep issues and prevent cognitive decline later in life,” lead author Victoire Leroy, MD, PhD, Albert Einstein College of Medicine, New York City, added in a press release.
SOURCE:
The study was published online in Neurology.
LIMITATIONS:
Study limitations included the lack of objective sleep measurements and potential recall bias in self-reported sleep complaints, particularly among participants with cognitive issues. In addition, the relatively short follow-up period may have resulted in a lower number of incident MCR syndrome cases. The sample population was also predominantly White (80%), which may have limited the generalizability of the findings to other populations.
DISCLOSURES:
The study was funded by the National Institute on Aging. No conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
a new study shows.
METHODOLOGY:
- Researchers included 445 older adults without dementia (mean age, 76 years; 57% women).
- Sleep components were assessed, and participants were classified as poor or good sleepers using the Pittsburgh Sleep Quality Index questionnaire.
- The primary outcome was incidence of MCR syndrome.
- The mean follow-up duration was 2.9 years.
TAKEAWAY:
- During the study period, 36 participants developed MCR syndrome.
- Poor sleepers had a higher risk for incident MCR syndrome, compared with good sleepers, after adjustment for age, sex, and educational level (adjusted hazard ratio [aHR], 2.6; 95% CI, 1.3-5.0; P < .05). However, this association was no longer significant after further adjustment for depressive symptoms.
- Sleep-related daytime dysfunction, defined as excessive sleepiness and lower enthusiasm for activities, was the only sleep component linked to a significant risk for MCR syndrome in fully adjusted models (aHR, 3.3; 95% CI, 1.5-7.4; P < .05).
- Prevalent MCR syndrome was not significantly associated with poor sleep quality (odds ratio, 1.1), suggesting that the relationship is unidirectional.
IN PRACTICE:
“Establishing the relationship between sleep dysfunction and MCR [syndrome] risk is important because early intervention may offer the best hope for preventing dementia,” the investigators wrote.
“Our findings emphasize the need for screening for sleep issues. There’s potential that people could get help with their sleep issues and prevent cognitive decline later in life,” lead author Victoire Leroy, MD, PhD, Albert Einstein College of Medicine, New York City, added in a press release.
SOURCE:
The study was published online in Neurology.
LIMITATIONS:
Study limitations included the lack of objective sleep measurements and potential recall bias in self-reported sleep complaints, particularly among participants with cognitive issues. In addition, the relatively short follow-up period may have resulted in a lower number of incident MCR syndrome cases. The sample population was also predominantly White (80%), which may have limited the generalizability of the findings to other populations.
DISCLOSURES:
The study was funded by the National Institute on Aging. No conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Impact and Recovery of VHA Epilepsy Care Services During the COVID-19 Pandemic
The COVID-19 pandemic affected diverse workplaces globally, leading to temporary and permanent changes across the health care landscape. Included among the impacted areas of care were epilepsy and electroencephalogram (EEG) clinicians and services. Surveys among epilepsy specialists and neurophysiologists conducted at the onset of the pandemic to evaluate working conditions include analyses from the American Epilepsy Society (AES), the National Association of Epilepsy Centers (NAEC), the International League Against Epilepsy, and an Italian national survey.1-4 These investigations revealed reductions in epilepsy monitoring unit (EMU) admissions (23% decline), epilepsy surgery (6% decline), inpatient EEG (22% of respondents reported decline), and patients having difficulty accessing epilepsy professionals (28% of respondents reported decline) or obtaining medications (20% of respondents reported decline).1-3
While such research provided evidence for changes to epilepsy care in 2020, there are limited data on subsequent adaptations during the pandemic. These studies did not incorporate data on the spread of COVID-19 or administrative workload numbers to analyze service delivery beyond self reports. This study aimed to address this gap in the literature by highlighting results from longitudinal national surveys conducted at the Epilepsy Centers of Excellence (ECoE), a specialty care service within the Veterans Health Administration (VHA), which annually serves > 9 million veterans.5 The ECoE represents epileptologists and neurophysiologists across the United States at the 17 primary facilities that were established at the time of this survey (2 ECoEs have been added since survey completion) in 4 geographical regions and for which other regional facilities refer patients for diagnostic services or specialty care.6
National surveys were conducted among the ECoE directors regarding adaptations made from May 2020 to June 2022 to provide a comprehensive account of limitations they experienced and how adjustments have been made to improve patient care. Survey responses were compared to administrative workload numbers and COVID-19 spread data from the Centers for Disease Control and Prevention (CDC) to provide a comprehensive analysis of performance during the pandemic.
METHODS
Data were collected as part of a quality improvement initiative by the VHA ECoE; institutional review board approval was not required. An 18-item survey covering 5 broad domains was sent to ECoE directors 4 separate times to accumulate data from 4 time periods: May to June 2020 (T1); December 2020 to February 2021 (T2); July to August 2021 (T3); and June to July 2022 (T4). These periods correspond to the following phases of the pandemic: T1, onset of pandemic; T2, vaccine availability; T3, Delta variant predominant; T4, Omicron variant predominant.
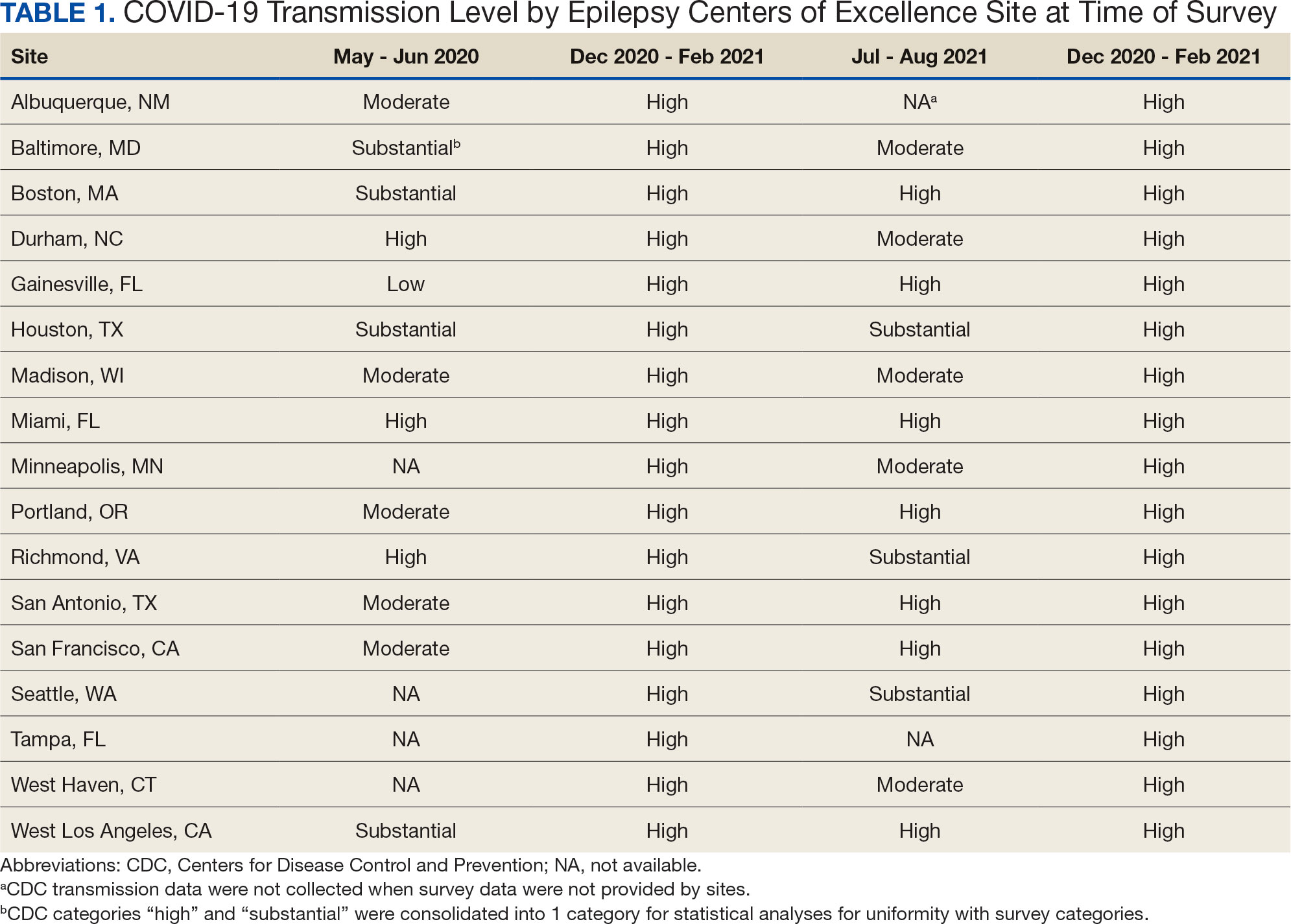
Data on the spread of COVID-19 were collected from the CDC archived dataset, US COVID-19 County Level of Community Transmission Historical Changes (Table 1).7 Administrative workload (patient counts) for EEG, EMU, and outpatient clinics were extracted from VHA administrative databases for the participating sites for the months prior to each survey: T1, April 2020; T2, November 2020; T3, June 2021; and T4, May 2022 (Table 2).
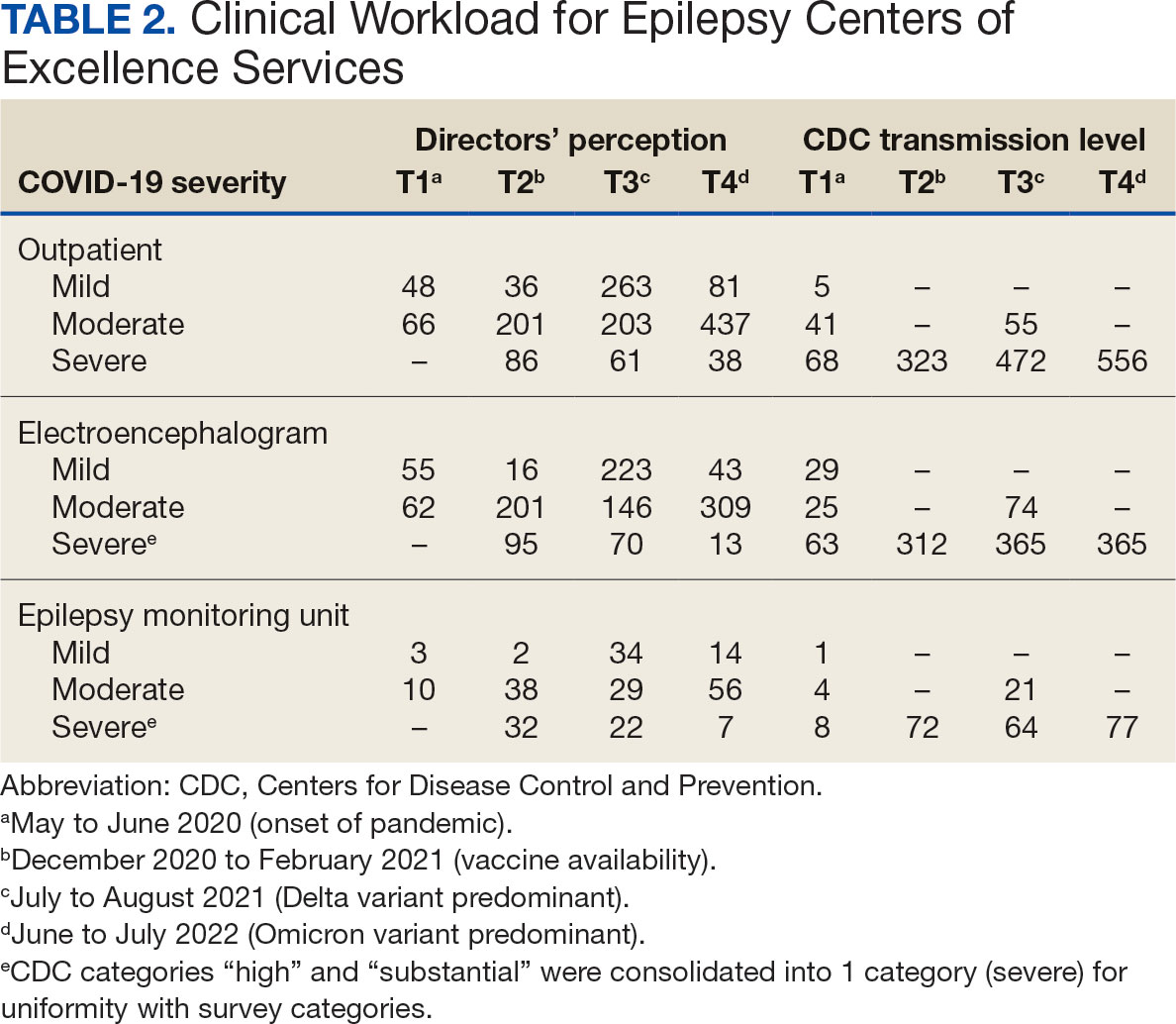
Survey Structure and Content
The survey was developed by the ECoE and was not validated prior to its use due to the time-sensitive nature of gathering information during the pandemic. The first survey (T1) was an emailed spreadsheet with open-ended questions to gauge availability of services (ie, outpatient clinic, EEG, EMU), assess whether safety precautions were being introduced, and understand whether national or local guidelines were thought to be helpful. Responses from this and subsequent surveys were standardized into yes/no and multiple choice formats. Subsequent surveys were administered online using a Research Electronic Data Capture tool.8,9
Availability of outpatient epilepsy services across the 4 time periods were categorized as unlimited (in-person with no restrictions), limited (in-person with restrictions), planned (not currently performed but scheduled for the near future), and unavailable (no in-person services offered) (eAppendices 1-6, available in article PDF).
Statistical Analyses
Analyses were performed to compare survey responses to workload and CDC data on COVID-19 community spread. The following associations were examined: (1) CDC COVID-19 spread vs respondents’ perception of spread; (2) respondents’ perception of spread vs availability of services; (3) CDC COVID-19 spread vs availability of services; (4) respondents’ perception of spread vs workload; and (5) CDC COVID-19 spread vs workload. Availability of services was dichotomized for analyses, with limited or fully available services classified as available. As services were mostly open at T3 regardless of the spread of the virus, and the CDC COVID-19 spread classification for all sites was severe or high at T2 and T4, corresponding associations were not tested at these time points. For associations 1 through 3, Fisher exact tests were used; for associations 4 and 5, Mann-Whitney U tests (where the COVID-19 spread fell into 2 categories) and Kruskal-Wallis tests (for 3 categories of COVID-19 spread) were performed. All tests were 2-tailed and performed at 0.05 error rate. Bonferroni corrections were applied to adjust P values for multiple hypotheses tests.
RESULTS
From the 17 sites invited, responses at each time point were obtained from 13 (T1),17 (T2), 15 (T3), and 16 (T4) centers. There was no significant association between self-reported COVID-19 spread and CDC classification of COVID spread. There were no associations between COVID-19 community spread (respondent reported or CDC severity level) and outpatient clinic availability (self-reported or workload captured). At T3, a positive association was found between the CDC spread level and workload (P = .008), but this was not significant after Bonferroni correction (P = .06).
EEG availability surpassed EMU availability at all time points, although EMU services made some recovery at T3 and T4. No associations were found between COVID-19 community spread (self-reported or CDC severity level) and outpatient EEG or EMU availability (self-reported or workload captured). At T3, there was a positive association between EEG workload and CDC COVID-19 severity level (P = .04), but this was not significant after Bonferroni correction (P = .30).
For outpatient EEG, staff and patient mask use were universally implemented by T2, while the use of full personal protective equipment (PPE) occurred at a subset of sites (T2, 6/17 [35%]; T3, 3/15 [20%]; T4: 4/16 [25%]). COVID-19 testing was rarely implemented prior to outpatient EEG (T1, 0 sites; T2, 1 site; T3, 1 site; T4, 0 sites). Within the EMU, safety precautions including COVID-19 testing, patient mask usage, staff mask usage, and aerosolization demonstrated a sustained majority usage across the 4 surveys.
National and Local Guidelines
The open-ended survey at T1 asked site directors, “Should there be national recommendations on how EEGs and related procedures should be done during the pandemic or should this be left to local conditions?” Responses were mixed, with 5 respondents desiring a national standard, 4 respondents favoring a local response, and 4 respondents believing a national standard should be in place but with modifications based on local outbreak levels and needs.
Surveys performed at T2 through T4 asked, “Which of the following do you feel was/will be helpful in adapting to COVID-19–related changes?” Overall, there was substantial agreement that guidelines were helpful. Most sites anticipated permanent changes in enhanced safety precautions and telehealth.
DISCUSSION
This longitudinal study across 4 time points describes how epilepsy services within the VHA and ECoE adapted to the COVID-19 pandemic. The first survey, conducted 2 months after COVID-19 was declared a pandemic, allowed a comparison with other concurrent US national surveys.1,2,10 The subsequent surveys describe longitudinal adaptations to balance patient and staff safety with service availability and is a unique feature of the current report. Results demonstrate flexibility and adaptability by the ECoEs surveyed, which surprisingly did not show significant associations between CDC COVID-19 spread data and administrative workload data.
Trends in Availability of Services
The most significant impact of COVID-19 restrictions was during T1. There were no significant relationships between service availability/workload and objective CDC COVID-19 spread levels or subjective self-reported COVID-19 spread. Respondents’ perceptions of local COVID-19 spread showed no association with CDC COVID-19 spread data. It appears that subjective perception of spread may be unreliable and factors other than actual or perceived COVID-19 spread were likely driving patterns for service availability.
In-person outpatient visits were most impacted at T1, similar to other civilian surveys, with only 1 site reporting in-person outpatient visits without limitations.1,2 These numbers significantly changed by T2, with all sites offering either limited or unlimited in-person visits. While the surveys did not evaluate factors leading to this rapid recovery, it may be related to the availability of COVID-19 vaccinations within the VHA during this time.11 The US Department of Veterans Affairs was the first federal agency to mandate employee vaccination.12 By the most recent time point (T4), all responding sites offered outpatient visits. Outpatient EEGs followed a similar trend, with T1 being the most restrictive and full, unrestricted outpatient EEGs available by T3.
Fiscal year (FY) trends from ECoE annual reports suggest that encounters slowly recovered over the course of the pandemic. In FY 2019 there were 13,143 outpatient encounters and 6394 EEGs, which dropped to 8097 outpatient encounters and 4432 EEGs in FY 2020 before rising to 8489 outpatient encounters and 5604 EEGs in FY 2021 and 9772 outpatient encounters and 5062 EEGs in FY 2022. Thus, while clinicians described availability of services, patients may have remained hesitant or were otherwise unable to fulfill in-person appointments. The increased availability of home EEG (145 encounter days in 2021 and 436 encounter days in 2022) may be filling this gap.
In contrast to outpatient clinics and EEG, EMU availability showed relatively slower reimplementation. In the last survey, about 30% of sites were still not offering EMU or had limited services. Early trends regarding reduced staffing and patient reluctance for elective admission cited in other surveys may have also affected EMU availability within the VHA.2,13 Consistent with trends in availability, ECoE annual report data suggest EMU patient participation was about one-half of prepandemic rates: 3069 encounters in FY 2019 dropped to 1614 encounters in 2020. By 2021, rates were about two-thirds of prepandemic rates with 2058 encounters in 2021 and 2101 encounters in 2022.
Early survey results (T1) from this study echo trends from other surveys. In the AES survey (April to June 2020), about a quarter of respondents (22%) reported doing fewer EEG studies than usual. The Italian national survey (April 2020) revealed reduced presurgical evaluations (81%), ambulatory EEG (78%), standard EEG (5%) and long-term EEG (32%).4 In the NAEC survey (end of 2020)—which roughly corresponded to T2—outpatient EEGs were still < 75% of pre-COVID levels in one-half of the centers.
National and Local Guidelines
Both national and local guidelines were perceived as useful by most respondents, with national guidelines being more beneficial. This aligns with the NAEC survey, where there was a perceived need for detailed recommendations for PPE and COVID-19 testing of patients, visitors, and staff. Based on national and local guidelines, ECoE implemented safety procedures, as reflected in responses. Staff masking procedures appeared to be the most widely adopted for all services, while the use of full PPE waned as the pandemic progressed. COVID-19 testing was rarely used for routine outpatient visits but common in EMU admissions. This is similar to a survey conducted by the American Academy of Neurology which found full PPE implementation intermittently in outpatient settings and more frequently in inpatient settings.14
Telehealth Attitudes
While most sites anticipated permanent implementation of safety precautions and telehealth, the latter was consistently reported as more likely to be sustained. The VHA had a large and well-developed system of telehealth services that considerably predated the pandemic.15,16 Through this established infrastructure, remote services were quickly increased across the VHA.17-19 This telehealth structure was supplemented by the ability of VHA clinicians to practice across state lines, following a 2018 federal rule.20 The AES survey noted the VHA ECoE's longstanding experience with telehealth as a model for telemedicine use in providing direct patient care, remote EEG analysis, and clinician-to-clinician consultation.1
Trends in the number of telehealth patients seen, observed through patterns in ECoE annual reports are consistent with positive views toward this method of service provision. Specifically, these annual reports capture trends in Video Telehealth Clinic (local station), Video Telehealth Clinic (different station), Home Video Telehealth, Telephone Clinic, and eConsults. Though video telehealth at in-person stations had a precipitous drop in 2020 that continued to wane in subsequent years (898 encounters in 2019; 455 encounters in 2020; 90 encounters in 2021; 88 encounters in 2022), use of home video telehealth rose over time (143 encounters in 2019; 1003 encounters in 2020; 3206 encounters in 2021; 3315 encounters in 2022). Use of telephone services rose drastically in 2020 but has since become a less frequently used service method (2636 in 2019; 5923 in 2020; 5319 in 2021; 3704 in 2022).
Limitations
While the survey encouraged a high response rate, this limited its scope and interpretability. While the availability of services was evaluated, the underlying reasons were not queried. Follow-up questions about barriers to reopening may have allowed for a better understanding of why some services, such as EMU, continued to operate suboptimally later in the pandemic. Similarly, asking about unique strategies or barriers for telehealth would have allowed for a better understanding of its current and future use. We hypothesize that staffing changes during the pandemic may have influenced the availability of services, but changes to staffing were not assessed via the survey and were not readily available via other sources (eg, ECoE annual reports) at the time of publication. An additional limitation is the lack of comparable surveys in the literature for time points T2 to T4, as most analogous surveys were performed early in 2020.
Conclusions
This longitudinal study performed at 4 time points during the COVID-19 pandemic is the first to offer a comprehensive picture of changes to epilepsy and EEG services over time, given that other similar surveys lacked follow-up. Results reveal a significant limitation of services at VHA ECoE shortly after the onset of the pandemic, with return to near-complete operational status 2 years later. While safety precautions and telehealth are predicted to continue, telehealth is perceived as a more permanent change in services.
Albert DVF, Das RR, Acharya JN, et al. The impact of COVID-19 on epilepsy care: a survey of the American Epilepsy Society membership. Epilepsy Curr. 2020;20(5):316-324. doi:10.1177/1535759720956994
Ahrens SM, Ostendorf AP, Lado FA, et al. Impact of the COVID-19 pandemic on epilepsy center practice in the United States. Neurology. 2022;98(19):e1893-e1901. doi:10.1212/WNL.0000000000200285
Cross JH, Kwon CS, Asadi-Pooya AA, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332. doi:10.1111/epi.17045
Assenza G, Lanzone J, Ricci L, et al. Electroencephalography at the time of Covid-19 pandemic in Italy. Neurol Sci. 2020;41(8):1999-2004. doi:10.1007/s10072-020-04546-8
US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Veteran population. Updated September 7, 2022. Accessed October 25, 2024. https://www.va.gov/vetdata/veteran_population.asp
US Department of Veterans Affairs, Veterans Health Administration. Epilepsy Centers of Excellence (ECoE). Annual report fiscal year 2019. Accessed October 25, 2024. https://www.epilepsy.va.gov/docs/FY19AnnualReport-VHAEpilepsyCentersofExcellence.pdf
Centers for Disease Control and Prevention. United States COVID-19 county level of community transmission historical changes – ARCHIVED. Updated February 20, 2024. Accessed October 25, 2024. https://data.cdc.gov/Public-Health-Surveillance/United-States-COVID-19-County-Level-of-Community-T/nra9-vzzn
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381. doi:10.1016/j.jbi.2008.08.010
Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
World Health Organization. Rolling updates on coronavirus disease (COVID-19). Updated July 31, 2020. Accessed October 25, 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen
US Department of Veterans Affairs. VA announces initial plans for COVID-19 vaccine distribution. News release. December 10, 2020. Accessed October 25, 2024. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=5580
Steinhauer J. V.A. Issues Vaccine Mandate for Health Care Workers, a First for a Federal Agency. The New York Times. August 16, 2021. Accessed October 25, 2024. https://www.nytimes.com/2021/07/26/us/politics/veterans-affairs-coronavirus-covid-19.html
Zafar SF, Khozein RJ, LaRoche SM, Westover MB, Gilmore EJ. Impact of the COVID-19 pandemic on continuous EEG utilization. J Clin Neurophysiol. 2022;39(7):567-574. doi:10.1097/WNP.0000000000000802
Qureshi AI, Rheaume C, Huang W, et al. COVID-19 exposure during neurology practice. Neurologist. 2021;26(6):225-230. doi:10.1097/NRL.0000000000000346
Darkins A, Cruise C, Armstrong M, Peters J, Finn M. Enhancing access of combat-wounded veterans to specialist rehabilitation services: the VA Polytrauma Telehealth Network. Arch Phys Med Rehabil. 2008;89(1):182-187. doi:10.1016/j.apmr.2007.07.027
Darkins A, Ryan P, Kobb R, et al. Care coordination/home telehealth: the systematic implementation of health informatics, home telehealth, and disease management to support the care of veteran patients with chronic conditions. Telemed J E Health. 2008;14(10):1118-1126. doi:10.1089/tmj.2008.0021
Gentry MT, Puspitasari AJ, McKean AJ, et al. Clinician satisfaction with rapid adoption and implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2021;27(12):1385-1392. doi:10.1089/tmj.2020.0575
Connolly SL, Stolzmann KL, Heyworth L, et al. Patient and provider predictors of telemental health use prior to and during the COVID-19 pandemic within the Department of Veterans Affairs. Am Psychol. 2022;77(2):249-261. doi:10.1037/amp0000895
Shelton CJ, Kim A, Hassan AM, Bhat A, Barnello J, Castro CA. System-wide implementation of telehealth to support military veterans and their families in response to COVID-19: a paradigm shift. J Mil Veteran Fam Health. 2020;6(S2):50-57. doi:10.3138/jmvfh-CO19-0003
VA expands telehealth by allowing health care providers to treat patients across state lines. News release. US Dept of Veterans Affairs. May 11, 2018. Accessed October 25, 2024. https://news.va.gov/press-room/va-expands-telehealth-by-allowing-health-care-providers-to-treat-patients-across-state-lines/
The COVID-19 pandemic affected diverse workplaces globally, leading to temporary and permanent changes across the health care landscape. Included among the impacted areas of care were epilepsy and electroencephalogram (EEG) clinicians and services. Surveys among epilepsy specialists and neurophysiologists conducted at the onset of the pandemic to evaluate working conditions include analyses from the American Epilepsy Society (AES), the National Association of Epilepsy Centers (NAEC), the International League Against Epilepsy, and an Italian national survey.1-4 These investigations revealed reductions in epilepsy monitoring unit (EMU) admissions (23% decline), epilepsy surgery (6% decline), inpatient EEG (22% of respondents reported decline), and patients having difficulty accessing epilepsy professionals (28% of respondents reported decline) or obtaining medications (20% of respondents reported decline).1-3
While such research provided evidence for changes to epilepsy care in 2020, there are limited data on subsequent adaptations during the pandemic. These studies did not incorporate data on the spread of COVID-19 or administrative workload numbers to analyze service delivery beyond self reports. This study aimed to address this gap in the literature by highlighting results from longitudinal national surveys conducted at the Epilepsy Centers of Excellence (ECoE), a specialty care service within the Veterans Health Administration (VHA), which annually serves > 9 million veterans.5 The ECoE represents epileptologists and neurophysiologists across the United States at the 17 primary facilities that were established at the time of this survey (2 ECoEs have been added since survey completion) in 4 geographical regions and for which other regional facilities refer patients for diagnostic services or specialty care.6
National surveys were conducted among the ECoE directors regarding adaptations made from May 2020 to June 2022 to provide a comprehensive account of limitations they experienced and how adjustments have been made to improve patient care. Survey responses were compared to administrative workload numbers and COVID-19 spread data from the Centers for Disease Control and Prevention (CDC) to provide a comprehensive analysis of performance during the pandemic.
METHODS
Data were collected as part of a quality improvement initiative by the VHA ECoE; institutional review board approval was not required. An 18-item survey covering 5 broad domains was sent to ECoE directors 4 separate times to accumulate data from 4 time periods: May to June 2020 (T1); December 2020 to February 2021 (T2); July to August 2021 (T3); and June to July 2022 (T4). These periods correspond to the following phases of the pandemic: T1, onset of pandemic; T2, vaccine availability; T3, Delta variant predominant; T4, Omicron variant predominant.

Data on the spread of COVID-19 were collected from the CDC archived dataset, US COVID-19 County Level of Community Transmission Historical Changes (Table 1).7 Administrative workload (patient counts) for EEG, EMU, and outpatient clinics were extracted from VHA administrative databases for the participating sites for the months prior to each survey: T1, April 2020; T2, November 2020; T3, June 2021; and T4, May 2022 (Table 2).

Survey Structure and Content
The survey was developed by the ECoE and was not validated prior to its use due to the time-sensitive nature of gathering information during the pandemic. The first survey (T1) was an emailed spreadsheet with open-ended questions to gauge availability of services (ie, outpatient clinic, EEG, EMU), assess whether safety precautions were being introduced, and understand whether national or local guidelines were thought to be helpful. Responses from this and subsequent surveys were standardized into yes/no and multiple choice formats. Subsequent surveys were administered online using a Research Electronic Data Capture tool.8,9
Availability of outpatient epilepsy services across the 4 time periods were categorized as unlimited (in-person with no restrictions), limited (in-person with restrictions), planned (not currently performed but scheduled for the near future), and unavailable (no in-person services offered) (eAppendices 1-6, available in article PDF).
Statistical Analyses
Analyses were performed to compare survey responses to workload and CDC data on COVID-19 community spread. The following associations were examined: (1) CDC COVID-19 spread vs respondents’ perception of spread; (2) respondents’ perception of spread vs availability of services; (3) CDC COVID-19 spread vs availability of services; (4) respondents’ perception of spread vs workload; and (5) CDC COVID-19 spread vs workload. Availability of services was dichotomized for analyses, with limited or fully available services classified as available. As services were mostly open at T3 regardless of the spread of the virus, and the CDC COVID-19 spread classification for all sites was severe or high at T2 and T4, corresponding associations were not tested at these time points. For associations 1 through 3, Fisher exact tests were used; for associations 4 and 5, Mann-Whitney U tests (where the COVID-19 spread fell into 2 categories) and Kruskal-Wallis tests (for 3 categories of COVID-19 spread) were performed. All tests were 2-tailed and performed at 0.05 error rate. Bonferroni corrections were applied to adjust P values for multiple hypotheses tests.
RESULTS
From the 17 sites invited, responses at each time point were obtained from 13 (T1),17 (T2), 15 (T3), and 16 (T4) centers. There was no significant association between self-reported COVID-19 spread and CDC classification of COVID spread. There were no associations between COVID-19 community spread (respondent reported or CDC severity level) and outpatient clinic availability (self-reported or workload captured). At T3, a positive association was found between the CDC spread level and workload (P = .008), but this was not significant after Bonferroni correction (P = .06).
EEG availability surpassed EMU availability at all time points, although EMU services made some recovery at T3 and T4. No associations were found between COVID-19 community spread (self-reported or CDC severity level) and outpatient EEG or EMU availability (self-reported or workload captured). At T3, there was a positive association between EEG workload and CDC COVID-19 severity level (P = .04), but this was not significant after Bonferroni correction (P = .30).
For outpatient EEG, staff and patient mask use were universally implemented by T2, while the use of full personal protective equipment (PPE) occurred at a subset of sites (T2, 6/17 [35%]; T3, 3/15 [20%]; T4: 4/16 [25%]). COVID-19 testing was rarely implemented prior to outpatient EEG (T1, 0 sites; T2, 1 site; T3, 1 site; T4, 0 sites). Within the EMU, safety precautions including COVID-19 testing, patient mask usage, staff mask usage, and aerosolization demonstrated a sustained majority usage across the 4 surveys.
National and Local Guidelines
The open-ended survey at T1 asked site directors, “Should there be national recommendations on how EEGs and related procedures should be done during the pandemic or should this be left to local conditions?” Responses were mixed, with 5 respondents desiring a national standard, 4 respondents favoring a local response, and 4 respondents believing a national standard should be in place but with modifications based on local outbreak levels and needs.
Surveys performed at T2 through T4 asked, “Which of the following do you feel was/will be helpful in adapting to COVID-19–related changes?” Overall, there was substantial agreement that guidelines were helpful. Most sites anticipated permanent changes in enhanced safety precautions and telehealth.
DISCUSSION
This longitudinal study across 4 time points describes how epilepsy services within the VHA and ECoE adapted to the COVID-19 pandemic. The first survey, conducted 2 months after COVID-19 was declared a pandemic, allowed a comparison with other concurrent US national surveys.1,2,10 The subsequent surveys describe longitudinal adaptations to balance patient and staff safety with service availability and is a unique feature of the current report. Results demonstrate flexibility and adaptability by the ECoEs surveyed, which surprisingly did not show significant associations between CDC COVID-19 spread data and administrative workload data.
Trends in Availability of Services
The most significant impact of COVID-19 restrictions was during T1. There were no significant relationships between service availability/workload and objective CDC COVID-19 spread levels or subjective self-reported COVID-19 spread. Respondents’ perceptions of local COVID-19 spread showed no association with CDC COVID-19 spread data. It appears that subjective perception of spread may be unreliable and factors other than actual or perceived COVID-19 spread were likely driving patterns for service availability.
In-person outpatient visits were most impacted at T1, similar to other civilian surveys, with only 1 site reporting in-person outpatient visits without limitations.1,2 These numbers significantly changed by T2, with all sites offering either limited or unlimited in-person visits. While the surveys did not evaluate factors leading to this rapid recovery, it may be related to the availability of COVID-19 vaccinations within the VHA during this time.11 The US Department of Veterans Affairs was the first federal agency to mandate employee vaccination.12 By the most recent time point (T4), all responding sites offered outpatient visits. Outpatient EEGs followed a similar trend, with T1 being the most restrictive and full, unrestricted outpatient EEGs available by T3.
Fiscal year (FY) trends from ECoE annual reports suggest that encounters slowly recovered over the course of the pandemic. In FY 2019 there were 13,143 outpatient encounters and 6394 EEGs, which dropped to 8097 outpatient encounters and 4432 EEGs in FY 2020 before rising to 8489 outpatient encounters and 5604 EEGs in FY 2021 and 9772 outpatient encounters and 5062 EEGs in FY 2022. Thus, while clinicians described availability of services, patients may have remained hesitant or were otherwise unable to fulfill in-person appointments. The increased availability of home EEG (145 encounter days in 2021 and 436 encounter days in 2022) may be filling this gap.
In contrast to outpatient clinics and EEG, EMU availability showed relatively slower reimplementation. In the last survey, about 30% of sites were still not offering EMU or had limited services. Early trends regarding reduced staffing and patient reluctance for elective admission cited in other surveys may have also affected EMU availability within the VHA.2,13 Consistent with trends in availability, ECoE annual report data suggest EMU patient participation was about one-half of prepandemic rates: 3069 encounters in FY 2019 dropped to 1614 encounters in 2020. By 2021, rates were about two-thirds of prepandemic rates with 2058 encounters in 2021 and 2101 encounters in 2022.
Early survey results (T1) from this study echo trends from other surveys. In the AES survey (April to June 2020), about a quarter of respondents (22%) reported doing fewer EEG studies than usual. The Italian national survey (April 2020) revealed reduced presurgical evaluations (81%), ambulatory EEG (78%), standard EEG (5%) and long-term EEG (32%).4 In the NAEC survey (end of 2020)—which roughly corresponded to T2—outpatient EEGs were still < 75% of pre-COVID levels in one-half of the centers.
National and Local Guidelines
Both national and local guidelines were perceived as useful by most respondents, with national guidelines being more beneficial. This aligns with the NAEC survey, where there was a perceived need for detailed recommendations for PPE and COVID-19 testing of patients, visitors, and staff. Based on national and local guidelines, ECoE implemented safety procedures, as reflected in responses. Staff masking procedures appeared to be the most widely adopted for all services, while the use of full PPE waned as the pandemic progressed. COVID-19 testing was rarely used for routine outpatient visits but common in EMU admissions. This is similar to a survey conducted by the American Academy of Neurology which found full PPE implementation intermittently in outpatient settings and more frequently in inpatient settings.14
Telehealth Attitudes
While most sites anticipated permanent implementation of safety precautions and telehealth, the latter was consistently reported as more likely to be sustained. The VHA had a large and well-developed system of telehealth services that considerably predated the pandemic.15,16 Through this established infrastructure, remote services were quickly increased across the VHA.17-19 This telehealth structure was supplemented by the ability of VHA clinicians to practice across state lines, following a 2018 federal rule.20 The AES survey noted the VHA ECoE's longstanding experience with telehealth as a model for telemedicine use in providing direct patient care, remote EEG analysis, and clinician-to-clinician consultation.1
Trends in the number of telehealth patients seen, observed through patterns in ECoE annual reports are consistent with positive views toward this method of service provision. Specifically, these annual reports capture trends in Video Telehealth Clinic (local station), Video Telehealth Clinic (different station), Home Video Telehealth, Telephone Clinic, and eConsults. Though video telehealth at in-person stations had a precipitous drop in 2020 that continued to wane in subsequent years (898 encounters in 2019; 455 encounters in 2020; 90 encounters in 2021; 88 encounters in 2022), use of home video telehealth rose over time (143 encounters in 2019; 1003 encounters in 2020; 3206 encounters in 2021; 3315 encounters in 2022). Use of telephone services rose drastically in 2020 but has since become a less frequently used service method (2636 in 2019; 5923 in 2020; 5319 in 2021; 3704 in 2022).
Limitations
While the survey encouraged a high response rate, this limited its scope and interpretability. While the availability of services was evaluated, the underlying reasons were not queried. Follow-up questions about barriers to reopening may have allowed for a better understanding of why some services, such as EMU, continued to operate suboptimally later in the pandemic. Similarly, asking about unique strategies or barriers for telehealth would have allowed for a better understanding of its current and future use. We hypothesize that staffing changes during the pandemic may have influenced the availability of services, but changes to staffing were not assessed via the survey and were not readily available via other sources (eg, ECoE annual reports) at the time of publication. An additional limitation is the lack of comparable surveys in the literature for time points T2 to T4, as most analogous surveys were performed early in 2020.
Conclusions
This longitudinal study performed at 4 time points during the COVID-19 pandemic is the first to offer a comprehensive picture of changes to epilepsy and EEG services over time, given that other similar surveys lacked follow-up. Results reveal a significant limitation of services at VHA ECoE shortly after the onset of the pandemic, with return to near-complete operational status 2 years later. While safety precautions and telehealth are predicted to continue, telehealth is perceived as a more permanent change in services.
The COVID-19 pandemic affected diverse workplaces globally, leading to temporary and permanent changes across the health care landscape. Included among the impacted areas of care were epilepsy and electroencephalogram (EEG) clinicians and services. Surveys among epilepsy specialists and neurophysiologists conducted at the onset of the pandemic to evaluate working conditions include analyses from the American Epilepsy Society (AES), the National Association of Epilepsy Centers (NAEC), the International League Against Epilepsy, and an Italian national survey.1-4 These investigations revealed reductions in epilepsy monitoring unit (EMU) admissions (23% decline), epilepsy surgery (6% decline), inpatient EEG (22% of respondents reported decline), and patients having difficulty accessing epilepsy professionals (28% of respondents reported decline) or obtaining medications (20% of respondents reported decline).1-3
While such research provided evidence for changes to epilepsy care in 2020, there are limited data on subsequent adaptations during the pandemic. These studies did not incorporate data on the spread of COVID-19 or administrative workload numbers to analyze service delivery beyond self reports. This study aimed to address this gap in the literature by highlighting results from longitudinal national surveys conducted at the Epilepsy Centers of Excellence (ECoE), a specialty care service within the Veterans Health Administration (VHA), which annually serves > 9 million veterans.5 The ECoE represents epileptologists and neurophysiologists across the United States at the 17 primary facilities that were established at the time of this survey (2 ECoEs have been added since survey completion) in 4 geographical regions and for which other regional facilities refer patients for diagnostic services or specialty care.6
National surveys were conducted among the ECoE directors regarding adaptations made from May 2020 to June 2022 to provide a comprehensive account of limitations they experienced and how adjustments have been made to improve patient care. Survey responses were compared to administrative workload numbers and COVID-19 spread data from the Centers for Disease Control and Prevention (CDC) to provide a comprehensive analysis of performance during the pandemic.
METHODS
Data were collected as part of a quality improvement initiative by the VHA ECoE; institutional review board approval was not required. An 18-item survey covering 5 broad domains was sent to ECoE directors 4 separate times to accumulate data from 4 time periods: May to June 2020 (T1); December 2020 to February 2021 (T2); July to August 2021 (T3); and June to July 2022 (T4). These periods correspond to the following phases of the pandemic: T1, onset of pandemic; T2, vaccine availability; T3, Delta variant predominant; T4, Omicron variant predominant.

Data on the spread of COVID-19 were collected from the CDC archived dataset, US COVID-19 County Level of Community Transmission Historical Changes (Table 1).7 Administrative workload (patient counts) for EEG, EMU, and outpatient clinics were extracted from VHA administrative databases for the participating sites for the months prior to each survey: T1, April 2020; T2, November 2020; T3, June 2021; and T4, May 2022 (Table 2).

Survey Structure and Content
The survey was developed by the ECoE and was not validated prior to its use due to the time-sensitive nature of gathering information during the pandemic. The first survey (T1) was an emailed spreadsheet with open-ended questions to gauge availability of services (ie, outpatient clinic, EEG, EMU), assess whether safety precautions were being introduced, and understand whether national or local guidelines were thought to be helpful. Responses from this and subsequent surveys were standardized into yes/no and multiple choice formats. Subsequent surveys were administered online using a Research Electronic Data Capture tool.8,9
Availability of outpatient epilepsy services across the 4 time periods were categorized as unlimited (in-person with no restrictions), limited (in-person with restrictions), planned (not currently performed but scheduled for the near future), and unavailable (no in-person services offered) (eAppendices 1-6, available in article PDF).
Statistical Analyses
Analyses were performed to compare survey responses to workload and CDC data on COVID-19 community spread. The following associations were examined: (1) CDC COVID-19 spread vs respondents’ perception of spread; (2) respondents’ perception of spread vs availability of services; (3) CDC COVID-19 spread vs availability of services; (4) respondents’ perception of spread vs workload; and (5) CDC COVID-19 spread vs workload. Availability of services was dichotomized for analyses, with limited or fully available services classified as available. As services were mostly open at T3 regardless of the spread of the virus, and the CDC COVID-19 spread classification for all sites was severe or high at T2 and T4, corresponding associations were not tested at these time points. For associations 1 through 3, Fisher exact tests were used; for associations 4 and 5, Mann-Whitney U tests (where the COVID-19 spread fell into 2 categories) and Kruskal-Wallis tests (for 3 categories of COVID-19 spread) were performed. All tests were 2-tailed and performed at 0.05 error rate. Bonferroni corrections were applied to adjust P values for multiple hypotheses tests.
RESULTS
From the 17 sites invited, responses at each time point were obtained from 13 (T1),17 (T2), 15 (T3), and 16 (T4) centers. There was no significant association between self-reported COVID-19 spread and CDC classification of COVID spread. There were no associations between COVID-19 community spread (respondent reported or CDC severity level) and outpatient clinic availability (self-reported or workload captured). At T3, a positive association was found between the CDC spread level and workload (P = .008), but this was not significant after Bonferroni correction (P = .06).
EEG availability surpassed EMU availability at all time points, although EMU services made some recovery at T3 and T4. No associations were found between COVID-19 community spread (self-reported or CDC severity level) and outpatient EEG or EMU availability (self-reported or workload captured). At T3, there was a positive association between EEG workload and CDC COVID-19 severity level (P = .04), but this was not significant after Bonferroni correction (P = .30).
For outpatient EEG, staff and patient mask use were universally implemented by T2, while the use of full personal protective equipment (PPE) occurred at a subset of sites (T2, 6/17 [35%]; T3, 3/15 [20%]; T4: 4/16 [25%]). COVID-19 testing was rarely implemented prior to outpatient EEG (T1, 0 sites; T2, 1 site; T3, 1 site; T4, 0 sites). Within the EMU, safety precautions including COVID-19 testing, patient mask usage, staff mask usage, and aerosolization demonstrated a sustained majority usage across the 4 surveys.
National and Local Guidelines
The open-ended survey at T1 asked site directors, “Should there be national recommendations on how EEGs and related procedures should be done during the pandemic or should this be left to local conditions?” Responses were mixed, with 5 respondents desiring a national standard, 4 respondents favoring a local response, and 4 respondents believing a national standard should be in place but with modifications based on local outbreak levels and needs.
Surveys performed at T2 through T4 asked, “Which of the following do you feel was/will be helpful in adapting to COVID-19–related changes?” Overall, there was substantial agreement that guidelines were helpful. Most sites anticipated permanent changes in enhanced safety precautions and telehealth.
DISCUSSION
This longitudinal study across 4 time points describes how epilepsy services within the VHA and ECoE adapted to the COVID-19 pandemic. The first survey, conducted 2 months after COVID-19 was declared a pandemic, allowed a comparison with other concurrent US national surveys.1,2,10 The subsequent surveys describe longitudinal adaptations to balance patient and staff safety with service availability and is a unique feature of the current report. Results demonstrate flexibility and adaptability by the ECoEs surveyed, which surprisingly did not show significant associations between CDC COVID-19 spread data and administrative workload data.
Trends in Availability of Services
The most significant impact of COVID-19 restrictions was during T1. There were no significant relationships between service availability/workload and objective CDC COVID-19 spread levels or subjective self-reported COVID-19 spread. Respondents’ perceptions of local COVID-19 spread showed no association with CDC COVID-19 spread data. It appears that subjective perception of spread may be unreliable and factors other than actual or perceived COVID-19 spread were likely driving patterns for service availability.
In-person outpatient visits were most impacted at T1, similar to other civilian surveys, with only 1 site reporting in-person outpatient visits without limitations.1,2 These numbers significantly changed by T2, with all sites offering either limited or unlimited in-person visits. While the surveys did not evaluate factors leading to this rapid recovery, it may be related to the availability of COVID-19 vaccinations within the VHA during this time.11 The US Department of Veterans Affairs was the first federal agency to mandate employee vaccination.12 By the most recent time point (T4), all responding sites offered outpatient visits. Outpatient EEGs followed a similar trend, with T1 being the most restrictive and full, unrestricted outpatient EEGs available by T3.
Fiscal year (FY) trends from ECoE annual reports suggest that encounters slowly recovered over the course of the pandemic. In FY 2019 there were 13,143 outpatient encounters and 6394 EEGs, which dropped to 8097 outpatient encounters and 4432 EEGs in FY 2020 before rising to 8489 outpatient encounters and 5604 EEGs in FY 2021 and 9772 outpatient encounters and 5062 EEGs in FY 2022. Thus, while clinicians described availability of services, patients may have remained hesitant or were otherwise unable to fulfill in-person appointments. The increased availability of home EEG (145 encounter days in 2021 and 436 encounter days in 2022) may be filling this gap.
In contrast to outpatient clinics and EEG, EMU availability showed relatively slower reimplementation. In the last survey, about 30% of sites were still not offering EMU or had limited services. Early trends regarding reduced staffing and patient reluctance for elective admission cited in other surveys may have also affected EMU availability within the VHA.2,13 Consistent with trends in availability, ECoE annual report data suggest EMU patient participation was about one-half of prepandemic rates: 3069 encounters in FY 2019 dropped to 1614 encounters in 2020. By 2021, rates were about two-thirds of prepandemic rates with 2058 encounters in 2021 and 2101 encounters in 2022.
Early survey results (T1) from this study echo trends from other surveys. In the AES survey (April to June 2020), about a quarter of respondents (22%) reported doing fewer EEG studies than usual. The Italian national survey (April 2020) revealed reduced presurgical evaluations (81%), ambulatory EEG (78%), standard EEG (5%) and long-term EEG (32%).4 In the NAEC survey (end of 2020)—which roughly corresponded to T2—outpatient EEGs were still < 75% of pre-COVID levels in one-half of the centers.
National and Local Guidelines
Both national and local guidelines were perceived as useful by most respondents, with national guidelines being more beneficial. This aligns with the NAEC survey, where there was a perceived need for detailed recommendations for PPE and COVID-19 testing of patients, visitors, and staff. Based on national and local guidelines, ECoE implemented safety procedures, as reflected in responses. Staff masking procedures appeared to be the most widely adopted for all services, while the use of full PPE waned as the pandemic progressed. COVID-19 testing was rarely used for routine outpatient visits but common in EMU admissions. This is similar to a survey conducted by the American Academy of Neurology which found full PPE implementation intermittently in outpatient settings and more frequently in inpatient settings.14
Telehealth Attitudes
While most sites anticipated permanent implementation of safety precautions and telehealth, the latter was consistently reported as more likely to be sustained. The VHA had a large and well-developed system of telehealth services that considerably predated the pandemic.15,16 Through this established infrastructure, remote services were quickly increased across the VHA.17-19 This telehealth structure was supplemented by the ability of VHA clinicians to practice across state lines, following a 2018 federal rule.20 The AES survey noted the VHA ECoE's longstanding experience with telehealth as a model for telemedicine use in providing direct patient care, remote EEG analysis, and clinician-to-clinician consultation.1
Trends in the number of telehealth patients seen, observed through patterns in ECoE annual reports are consistent with positive views toward this method of service provision. Specifically, these annual reports capture trends in Video Telehealth Clinic (local station), Video Telehealth Clinic (different station), Home Video Telehealth, Telephone Clinic, and eConsults. Though video telehealth at in-person stations had a precipitous drop in 2020 that continued to wane in subsequent years (898 encounters in 2019; 455 encounters in 2020; 90 encounters in 2021; 88 encounters in 2022), use of home video telehealth rose over time (143 encounters in 2019; 1003 encounters in 2020; 3206 encounters in 2021; 3315 encounters in 2022). Use of telephone services rose drastically in 2020 but has since become a less frequently used service method (2636 in 2019; 5923 in 2020; 5319 in 2021; 3704 in 2022).
Limitations
While the survey encouraged a high response rate, this limited its scope and interpretability. While the availability of services was evaluated, the underlying reasons were not queried. Follow-up questions about barriers to reopening may have allowed for a better understanding of why some services, such as EMU, continued to operate suboptimally later in the pandemic. Similarly, asking about unique strategies or barriers for telehealth would have allowed for a better understanding of its current and future use. We hypothesize that staffing changes during the pandemic may have influenced the availability of services, but changes to staffing were not assessed via the survey and were not readily available via other sources (eg, ECoE annual reports) at the time of publication. An additional limitation is the lack of comparable surveys in the literature for time points T2 to T4, as most analogous surveys were performed early in 2020.
Conclusions
This longitudinal study performed at 4 time points during the COVID-19 pandemic is the first to offer a comprehensive picture of changes to epilepsy and EEG services over time, given that other similar surveys lacked follow-up. Results reveal a significant limitation of services at VHA ECoE shortly after the onset of the pandemic, with return to near-complete operational status 2 years later. While safety precautions and telehealth are predicted to continue, telehealth is perceived as a more permanent change in services.
Albert DVF, Das RR, Acharya JN, et al. The impact of COVID-19 on epilepsy care: a survey of the American Epilepsy Society membership. Epilepsy Curr. 2020;20(5):316-324. doi:10.1177/1535759720956994
Ahrens SM, Ostendorf AP, Lado FA, et al. Impact of the COVID-19 pandemic on epilepsy center practice in the United States. Neurology. 2022;98(19):e1893-e1901. doi:10.1212/WNL.0000000000200285
Cross JH, Kwon CS, Asadi-Pooya AA, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332. doi:10.1111/epi.17045
Assenza G, Lanzone J, Ricci L, et al. Electroencephalography at the time of Covid-19 pandemic in Italy. Neurol Sci. 2020;41(8):1999-2004. doi:10.1007/s10072-020-04546-8
US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Veteran population. Updated September 7, 2022. Accessed October 25, 2024. https://www.va.gov/vetdata/veteran_population.asp
US Department of Veterans Affairs, Veterans Health Administration. Epilepsy Centers of Excellence (ECoE). Annual report fiscal year 2019. Accessed October 25, 2024. https://www.epilepsy.va.gov/docs/FY19AnnualReport-VHAEpilepsyCentersofExcellence.pdf
Centers for Disease Control and Prevention. United States COVID-19 county level of community transmission historical changes – ARCHIVED. Updated February 20, 2024. Accessed October 25, 2024. https://data.cdc.gov/Public-Health-Surveillance/United-States-COVID-19-County-Level-of-Community-T/nra9-vzzn
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381. doi:10.1016/j.jbi.2008.08.010
Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
World Health Organization. Rolling updates on coronavirus disease (COVID-19). Updated July 31, 2020. Accessed October 25, 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen
US Department of Veterans Affairs. VA announces initial plans for COVID-19 vaccine distribution. News release. December 10, 2020. Accessed October 25, 2024. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=5580
Steinhauer J. V.A. Issues Vaccine Mandate for Health Care Workers, a First for a Federal Agency. The New York Times. August 16, 2021. Accessed October 25, 2024. https://www.nytimes.com/2021/07/26/us/politics/veterans-affairs-coronavirus-covid-19.html
Zafar SF, Khozein RJ, LaRoche SM, Westover MB, Gilmore EJ. Impact of the COVID-19 pandemic on continuous EEG utilization. J Clin Neurophysiol. 2022;39(7):567-574. doi:10.1097/WNP.0000000000000802
Qureshi AI, Rheaume C, Huang W, et al. COVID-19 exposure during neurology practice. Neurologist. 2021;26(6):225-230. doi:10.1097/NRL.0000000000000346
Darkins A, Cruise C, Armstrong M, Peters J, Finn M. Enhancing access of combat-wounded veterans to specialist rehabilitation services: the VA Polytrauma Telehealth Network. Arch Phys Med Rehabil. 2008;89(1):182-187. doi:10.1016/j.apmr.2007.07.027
Darkins A, Ryan P, Kobb R, et al. Care coordination/home telehealth: the systematic implementation of health informatics, home telehealth, and disease management to support the care of veteran patients with chronic conditions. Telemed J E Health. 2008;14(10):1118-1126. doi:10.1089/tmj.2008.0021
Gentry MT, Puspitasari AJ, McKean AJ, et al. Clinician satisfaction with rapid adoption and implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2021;27(12):1385-1392. doi:10.1089/tmj.2020.0575
Connolly SL, Stolzmann KL, Heyworth L, et al. Patient and provider predictors of telemental health use prior to and during the COVID-19 pandemic within the Department of Veterans Affairs. Am Psychol. 2022;77(2):249-261. doi:10.1037/amp0000895
Shelton CJ, Kim A, Hassan AM, Bhat A, Barnello J, Castro CA. System-wide implementation of telehealth to support military veterans and their families in response to COVID-19: a paradigm shift. J Mil Veteran Fam Health. 2020;6(S2):50-57. doi:10.3138/jmvfh-CO19-0003
VA expands telehealth by allowing health care providers to treat patients across state lines. News release. US Dept of Veterans Affairs. May 11, 2018. Accessed October 25, 2024. https://news.va.gov/press-room/va-expands-telehealth-by-allowing-health-care-providers-to-treat-patients-across-state-lines/
Albert DVF, Das RR, Acharya JN, et al. The impact of COVID-19 on epilepsy care: a survey of the American Epilepsy Society membership. Epilepsy Curr. 2020;20(5):316-324. doi:10.1177/1535759720956994
Ahrens SM, Ostendorf AP, Lado FA, et al. Impact of the COVID-19 pandemic on epilepsy center practice in the United States. Neurology. 2022;98(19):e1893-e1901. doi:10.1212/WNL.0000000000200285
Cross JH, Kwon CS, Asadi-Pooya AA, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332. doi:10.1111/epi.17045
Assenza G, Lanzone J, Ricci L, et al. Electroencephalography at the time of Covid-19 pandemic in Italy. Neurol Sci. 2020;41(8):1999-2004. doi:10.1007/s10072-020-04546-8
US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. Veteran population. Updated September 7, 2022. Accessed October 25, 2024. https://www.va.gov/vetdata/veteran_population.asp
US Department of Veterans Affairs, Veterans Health Administration. Epilepsy Centers of Excellence (ECoE). Annual report fiscal year 2019. Accessed October 25, 2024. https://www.epilepsy.va.gov/docs/FY19AnnualReport-VHAEpilepsyCentersofExcellence.pdf
Centers for Disease Control and Prevention. United States COVID-19 county level of community transmission historical changes – ARCHIVED. Updated February 20, 2024. Accessed October 25, 2024. https://data.cdc.gov/Public-Health-Surveillance/United-States-COVID-19-County-Level-of-Community-T/nra9-vzzn
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-381. doi:10.1016/j.jbi.2008.08.010
Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi:10.1016/j.jbi.2019.103208
World Health Organization. Rolling updates on coronavirus disease (COVID-19). Updated July 31, 2020. Accessed October 25, 2024. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen
US Department of Veterans Affairs. VA announces initial plans for COVID-19 vaccine distribution. News release. December 10, 2020. Accessed October 25, 2024. https://www.va.gov/opa/pressrel/pressrelease.cfm?id=5580
Steinhauer J. V.A. Issues Vaccine Mandate for Health Care Workers, a First for a Federal Agency. The New York Times. August 16, 2021. Accessed October 25, 2024. https://www.nytimes.com/2021/07/26/us/politics/veterans-affairs-coronavirus-covid-19.html
Zafar SF, Khozein RJ, LaRoche SM, Westover MB, Gilmore EJ. Impact of the COVID-19 pandemic on continuous EEG utilization. J Clin Neurophysiol. 2022;39(7):567-574. doi:10.1097/WNP.0000000000000802
Qureshi AI, Rheaume C, Huang W, et al. COVID-19 exposure during neurology practice. Neurologist. 2021;26(6):225-230. doi:10.1097/NRL.0000000000000346
Darkins A, Cruise C, Armstrong M, Peters J, Finn M. Enhancing access of combat-wounded veterans to specialist rehabilitation services: the VA Polytrauma Telehealth Network. Arch Phys Med Rehabil. 2008;89(1):182-187. doi:10.1016/j.apmr.2007.07.027
Darkins A, Ryan P, Kobb R, et al. Care coordination/home telehealth: the systematic implementation of health informatics, home telehealth, and disease management to support the care of veteran patients with chronic conditions. Telemed J E Health. 2008;14(10):1118-1126. doi:10.1089/tmj.2008.0021
Gentry MT, Puspitasari AJ, McKean AJ, et al. Clinician satisfaction with rapid adoption and implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2021;27(12):1385-1392. doi:10.1089/tmj.2020.0575
Connolly SL, Stolzmann KL, Heyworth L, et al. Patient and provider predictors of telemental health use prior to and during the COVID-19 pandemic within the Department of Veterans Affairs. Am Psychol. 2022;77(2):249-261. doi:10.1037/amp0000895
Shelton CJ, Kim A, Hassan AM, Bhat A, Barnello J, Castro CA. System-wide implementation of telehealth to support military veterans and their families in response to COVID-19: a paradigm shift. J Mil Veteran Fam Health. 2020;6(S2):50-57. doi:10.3138/jmvfh-CO19-0003
VA expands telehealth by allowing health care providers to treat patients across state lines. News release. US Dept of Veterans Affairs. May 11, 2018. Accessed October 25, 2024. https://news.va.gov/press-room/va-expands-telehealth-by-allowing-health-care-providers-to-treat-patients-across-state-lines/
Managing Diabetes and Dementia in Long-Term Care
VANCOUVER, BRITISH COLUMBIA — Conditions like diabetes and dementia are common in patients who are admitted to long-term care facilities, but aggressive management of these conditions in long-term care residents is not recommended, according to a presentation given at the Family Medicine Forum (FMF) 2024.
Hospitalizations for hypoglycemia are risky for patients with diabetes who are residents of long-term care facilities, particularly those aged 75 years or older, said Adam Gurau, MD, a family physician in Toronto. Gurau completed a fellowship in care of the elderly at the University of Toronto, in Ontario, Canada.
“A lot of studies have shown diabetes-related hospitalizations,” said Gurau. He cited a 2014 study that found that hypoglycemia hospitalization rates were twice as high in older patients (age, 75 years or older) as in younger patients (age, 65-74 years).
“It is important to keep in mind that our residents in long-term care are at increasing risk for hypoglycemia, and we really should try to reduce [this risk] and not use dangerous medications or potentially dangerous [means of] diabetes management,” said Gurau.
A Canadian study that examined the composite risk for emergency department visits, hospitalizations, or death within 30 days of reaching intensive glycemic control with high-risk agents (such as insulin or sulfonylureas) suggested little benefit and possible harm in using these agents in adults aged 75 years or older.
In addition, current guidelines on diabetes management encourage a different approach. “Looking at some of the more recent North American guidelines, many of them actually now recommend relaxing glycemic targets to reduce overtreatment and prevent hypoglycemia,” said Gurau.
Deprescribing Medications
Medication reviews present opportunities for taking a global view of a patient’s treatments and determining whether any drug can be removed from the list. “What we want to do is optimize medications,” said Gurau. “We’re not talking about adding medications. We’re talking about removing medications, which is, I think, what we should be doing.”
Some research suggests that patients are open to deprescribing. One survey examined older adults (mean age, 79.1 years) with three or more chronic conditions who had been prescribed at least five medications. The researchers found that most participants (77%) were willing to deprescribe one or more medicines if a doctor advised that it was possible. “General practitioners may be able to increase deprescribing by building trust with their patients and communicating evidence about the risks of medication use,” the researchers wrote.
About 62% of seniors living in a residential care home have a diagnosis of Alzheimer’s disease or another dementia, according to the Alzheimer Society of Canada. Evidence suggests that nonpharmacologic approaches, such as massage and touch therapy and music, can manage neuropsychiatric symptoms, such as aggression and agitation, that are associated with dementia in older adults, noted Gurau.
“We want to focus on nonpharmacologic approaches for many of these [long-term care] residents,” said Gurau. “We have to do as much as we can to exhaust all the nonpharmacologic approaches.”
Preventing Hospitalizations
Another challenge to tackle in long-term care is the unnecessary transfer of residents to hospital emergency departments, according to Gurau. “In many situations, it’s worth trying as hard as we can to treat them in the nursing home, as opposed to having them go to hospital.”
Researchers estimated that 25% of the transfers from long-term care facilities in Canada to hospital emergency departments in 2014 were potentially preventable.
Urinary tract infections accounted for 30% of hospital emergency department visits for potentially preventable conditions by older patients who are residents in long-term care, according to 2013-2014 data from the Canadian Institute for Health Information.
“There are lots of downsides to going to the hospital [from long-term care],” Gurau told this news organization. “There are risks for infections, risks for increasing delirium and agitation [in patients with dementia], and risks for other behavior that can really impact somebody’s life.”
Gurau reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER, BRITISH COLUMBIA — Conditions like diabetes and dementia are common in patients who are admitted to long-term care facilities, but aggressive management of these conditions in long-term care residents is not recommended, according to a presentation given at the Family Medicine Forum (FMF) 2024.
Hospitalizations for hypoglycemia are risky for patients with diabetes who are residents of long-term care facilities, particularly those aged 75 years or older, said Adam Gurau, MD, a family physician in Toronto. Gurau completed a fellowship in care of the elderly at the University of Toronto, in Ontario, Canada.
“A lot of studies have shown diabetes-related hospitalizations,” said Gurau. He cited a 2014 study that found that hypoglycemia hospitalization rates were twice as high in older patients (age, 75 years or older) as in younger patients (age, 65-74 years).
“It is important to keep in mind that our residents in long-term care are at increasing risk for hypoglycemia, and we really should try to reduce [this risk] and not use dangerous medications or potentially dangerous [means of] diabetes management,” said Gurau.
A Canadian study that examined the composite risk for emergency department visits, hospitalizations, or death within 30 days of reaching intensive glycemic control with high-risk agents (such as insulin or sulfonylureas) suggested little benefit and possible harm in using these agents in adults aged 75 years or older.
In addition, current guidelines on diabetes management encourage a different approach. “Looking at some of the more recent North American guidelines, many of them actually now recommend relaxing glycemic targets to reduce overtreatment and prevent hypoglycemia,” said Gurau.
Deprescribing Medications
Medication reviews present opportunities for taking a global view of a patient’s treatments and determining whether any drug can be removed from the list. “What we want to do is optimize medications,” said Gurau. “We’re not talking about adding medications. We’re talking about removing medications, which is, I think, what we should be doing.”
Some research suggests that patients are open to deprescribing. One survey examined older adults (mean age, 79.1 years) with three or more chronic conditions who had been prescribed at least five medications. The researchers found that most participants (77%) were willing to deprescribe one or more medicines if a doctor advised that it was possible. “General practitioners may be able to increase deprescribing by building trust with their patients and communicating evidence about the risks of medication use,” the researchers wrote.
About 62% of seniors living in a residential care home have a diagnosis of Alzheimer’s disease or another dementia, according to the Alzheimer Society of Canada. Evidence suggests that nonpharmacologic approaches, such as massage and touch therapy and music, can manage neuropsychiatric symptoms, such as aggression and agitation, that are associated with dementia in older adults, noted Gurau.
“We want to focus on nonpharmacologic approaches for many of these [long-term care] residents,” said Gurau. “We have to do as much as we can to exhaust all the nonpharmacologic approaches.”
Preventing Hospitalizations
Another challenge to tackle in long-term care is the unnecessary transfer of residents to hospital emergency departments, according to Gurau. “In many situations, it’s worth trying as hard as we can to treat them in the nursing home, as opposed to having them go to hospital.”
Researchers estimated that 25% of the transfers from long-term care facilities in Canada to hospital emergency departments in 2014 were potentially preventable.
Urinary tract infections accounted for 30% of hospital emergency department visits for potentially preventable conditions by older patients who are residents in long-term care, according to 2013-2014 data from the Canadian Institute for Health Information.
“There are lots of downsides to going to the hospital [from long-term care],” Gurau told this news organization. “There are risks for infections, risks for increasing delirium and agitation [in patients with dementia], and risks for other behavior that can really impact somebody’s life.”
Gurau reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER, BRITISH COLUMBIA — Conditions like diabetes and dementia are common in patients who are admitted to long-term care facilities, but aggressive management of these conditions in long-term care residents is not recommended, according to a presentation given at the Family Medicine Forum (FMF) 2024.
Hospitalizations for hypoglycemia are risky for patients with diabetes who are residents of long-term care facilities, particularly those aged 75 years or older, said Adam Gurau, MD, a family physician in Toronto. Gurau completed a fellowship in care of the elderly at the University of Toronto, in Ontario, Canada.
“A lot of studies have shown diabetes-related hospitalizations,” said Gurau. He cited a 2014 study that found that hypoglycemia hospitalization rates were twice as high in older patients (age, 75 years or older) as in younger patients (age, 65-74 years).
“It is important to keep in mind that our residents in long-term care are at increasing risk for hypoglycemia, and we really should try to reduce [this risk] and not use dangerous medications or potentially dangerous [means of] diabetes management,” said Gurau.
A Canadian study that examined the composite risk for emergency department visits, hospitalizations, or death within 30 days of reaching intensive glycemic control with high-risk agents (such as insulin or sulfonylureas) suggested little benefit and possible harm in using these agents in adults aged 75 years or older.
In addition, current guidelines on diabetes management encourage a different approach. “Looking at some of the more recent North American guidelines, many of them actually now recommend relaxing glycemic targets to reduce overtreatment and prevent hypoglycemia,” said Gurau.
Deprescribing Medications
Medication reviews present opportunities for taking a global view of a patient’s treatments and determining whether any drug can be removed from the list. “What we want to do is optimize medications,” said Gurau. “We’re not talking about adding medications. We’re talking about removing medications, which is, I think, what we should be doing.”
Some research suggests that patients are open to deprescribing. One survey examined older adults (mean age, 79.1 years) with three or more chronic conditions who had been prescribed at least five medications. The researchers found that most participants (77%) were willing to deprescribe one or more medicines if a doctor advised that it was possible. “General practitioners may be able to increase deprescribing by building trust with their patients and communicating evidence about the risks of medication use,” the researchers wrote.
About 62% of seniors living in a residential care home have a diagnosis of Alzheimer’s disease or another dementia, according to the Alzheimer Society of Canada. Evidence suggests that nonpharmacologic approaches, such as massage and touch therapy and music, can manage neuropsychiatric symptoms, such as aggression and agitation, that are associated with dementia in older adults, noted Gurau.
“We want to focus on nonpharmacologic approaches for many of these [long-term care] residents,” said Gurau. “We have to do as much as we can to exhaust all the nonpharmacologic approaches.”
Preventing Hospitalizations
Another challenge to tackle in long-term care is the unnecessary transfer of residents to hospital emergency departments, according to Gurau. “In many situations, it’s worth trying as hard as we can to treat them in the nursing home, as opposed to having them go to hospital.”
Researchers estimated that 25% of the transfers from long-term care facilities in Canada to hospital emergency departments in 2014 were potentially preventable.
Urinary tract infections accounted for 30% of hospital emergency department visits for potentially preventable conditions by older patients who are residents in long-term care, according to 2013-2014 data from the Canadian Institute for Health Information.
“There are lots of downsides to going to the hospital [from long-term care],” Gurau told this news organization. “There are risks for infections, risks for increasing delirium and agitation [in patients with dementia], and risks for other behavior that can really impact somebody’s life.”
Gurau reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM FMF 2024
First Phase 3 Drug Trial in IgG4-Related Disease Has Success
WASHINGTON — The B cell–depleting agent inebilizumab (Uplizna) dramatically reduced the risk of flares and increased year-long remission of IgG4-related disease (RD), new research has found.
In a phase 3, multicenter, double-blind, randomized, placebo-controlled trial of 135 adults with active IgG4-RD, treatment with inebilizumab resulted in a significant 87% reduction in flare risk and nearly fivefold greater likelihood of flare-free remission at 1 year. The results were published online November 14 in The New England Journal of Medicine and were presented at the annual meeting of the American College of Rheumatology (ACR).
The drug’s manufacturer, Amgen, released top-line results of the trial, called MITIGATE, in June 2024.
Until now, the mainstay of management for the chronic multiorgan disease IgG4-RD has been glucocorticoids, which can cause numerous adverse effects. “It is hoped that inebilizumab can be used as an important steroid-sparing medication in this disease to reduce steroid toxicity,” lead author John H. Stone, MD, professor of medicine at Harvard Medical School, Boston, Massachusetts, said in an interview, noting that it may not entirely eliminate the need for steroid treatment, but for many, it appears to work after the remission induction period as a monotherapy without steroids.
Asked to comment, Leonard H. Calabrese, DO, head of the Section of Clinical Immunology and manager of the Clinical Immunology Clinic at the Cleveland Clinic, Ohio, said: “There has been anecdotal or observational evidence for some effect with other immunosuppressive agents, including rituximab, but no robust clinical trial until this study. This clearly has demonstrated efficacy by reducing the risk of flares. And most importantly, putting people into remission means no active disease in any given organ. ... This gives us another tool in the toolbox to attack B cell–directed diseases, and I think it really makes a lot of sense.”
Calabrese cautioned, though, that “this is a disease that extends over many years. This is just a 1-year study. Label extensions will be important.”
And several questions remain, Calabrese noted: “How long do patients need to remain on drug? What will happen when the drug is stopped? Can they be retreated? These are the natural questions that arise in any sentinel study like this. But this is extremely encouraging. And I think it’s great for patients. I also think it’s a clarion call to increase awareness about this disease since there’s now strong evidence of effective treatment.”
Underrecognized, Often Misdiagnosed as Cancer
Indeed, IgG4-RD, a chronic, relapsing, autoimmune, fibro-inflammatory multiorgan disease, was only first described in Japan in 2003. Since then, it has been reported all over the world yet remains vastly underrecognized. It is often misdiagnosed as cancer because it produces lesions in multiple organs. It received an ICD-10 code only about a year ago. A previous study estimated a prevalence of about 5.3 persons per 100,000 but that is likely to be a three- to fourfold underestimate, said Stone, who is also executive chairman of the IgG4ward! Foundation.
“Nobody had heard of the disease until about 20 years ago. ... And there are many people in the world who have still not heard of it despite the fact that it is a multiorgan autoimmune disease and is probably as common, or more common, than many other diseases that rheumatologists spend a lot of time thinking about, such as scleroderma.”
While knowledge about the disease is increasing in rheumatology circles, it’s less well-recognized among many of the specialties where patients present, depending on the location of their lesions. These include gastroenterology, ophthalmology, pulmonary medicine, neurology, and nephrology. “All would be likely to see this disease,” Stone said.
The disease can be mistaken for tumors in many of those locations and even as metastatic cancer, he noted, adding that “any time a patient has a mass lesion in a typical organ, the pancreas, the major salivary glands, the lungs, or the kidneys, this should be on the differential diagnosis.”
The diagnosis of IgG4-RD is a clinical one, involving “quadrangulation between clinical features, serological findings, IgG4 levels in the blood, radiology studies, and then pathology biopsies when those are available,” Stone said.
Calabrese characterized the current situation as “we’re all blind men on the elephant. To the neurologist or the neurosurgeon, it’s a mass in the brain. It could present to the ophthalmologist as an [eye] tumor. It can be thyroid gland failure, pulmonary disease, retroperitoneal fibrosis, hepatobiliary disease, and beyond. So, whoever sees that patient, there’s often a long lag time in recognizing it.”
And interestingly, Stone noted that unlike other autoimmune diseases, IgG4-RD primarily affects middle-aged men rather than younger-to-middle-aged women. And when IgG4-RD is diagnosed, glucocorticoid treatment can be particularly toxic when the pancreas is involved, heightening the risk for hyperglycemia and potentially causing diabetes.
Dramatic Improvement in Flares, Remission Achievement
MITIGATE is a phase 3, multicenter, double-blind, randomized, placebo-controlled trial in which 135 adults (mean age 58.2 years, 88 men) with active IgG4-RD were randomized 1:1 to receive 300-mg intravenous infusions of inebilizumab or placebo on days 1 and 15, and again at week 26. At baseline, 62 (45.9%) participants had newly diagnosed IgG4-RD and 73 (54.1%) had recurrent disease.
Both groups received identical glucocorticoid tapers. Overall, 127 (94.1%) completed the 52 weeks of treatment.
By 52 weeks, only seven patients in the inebilizumab group (10%) had experienced disease flares vs 40 (60%) in the placebo group, a significant difference with a hazard ratio of 0.13 (P < .001).
The percentage of participants achieving flare-free, treatment-free complete remission was 59 with inebilizumab (57%), compared with just 15 (22%) in the placebo group (odds ratio [OR], 4.68; P < .001). And for flare-free, glucocorticoid-free complete remission, those proportions were 40 (59%) vs 15 (22%), respectively (OR, 4.96; P < .001).
Excluding the 8-week glucocorticoid taper period, mean total glucocorticoid use was 1264.2 mg less in the inebilizumab than the placebo group, a significant reduction. Overall, 61 participants (90%) were able to entirely discontinue glucocorticoids during the trial, compared with just 25 (37%) in the placebo group.
Adverse events of grade 3 or higher occurred in 12 participants (18%) in the inebilizumab group and 8 (12%) in the placebo group; serious adverse events occurred in 12 (18%) and 6 (9%), respectively. However, no serious adverse event occurred in more than one participant, and there were no deaths. Adverse events led to withdrawal from the trial in six patients (9%) in the inebilizumab group and three patients (4%) in the placebo group.
Adverse events that occurred in more than 10% of participants in the inebilizumab group were COVID-19 in 16 participants (24%), lymphopenia in 11 (16%), and urinary tract infection in 8 (12%).
Importantly, Stone noted, B-cell depletion can reduce responses to vaccines, so patients should receive all recommended vaccinations, including COVID-19, influenza, respiratory syncytial virus, and others, prior to initiating therapy.
Uplizna (inebilizumab-cdon) was approved by the Food and Drug Administration (FDA) for the treatment of neuromyelitis optica spectrum disorder in 2020. In October 2024, the FDA granted Amgen breakthrough therapy designation for use in IgG4-RD. The company is also developing the drug for use in myasthenia gravis.
The study was funded by Amgen. Stone has reported being a consultant for Amgen, Zenas, Argenx, Bristol Myers Squibb, Novartis, Sanofi, and Horizon Pharma. Calabrese has reported being a consultant and/or speaker for Amgen, AstraZeneca, Jansen, Sanofi, and UCB.
A version of this article first appeared on Medscape.com.
WASHINGTON — The B cell–depleting agent inebilizumab (Uplizna) dramatically reduced the risk of flares and increased year-long remission of IgG4-related disease (RD), new research has found.
In a phase 3, multicenter, double-blind, randomized, placebo-controlled trial of 135 adults with active IgG4-RD, treatment with inebilizumab resulted in a significant 87% reduction in flare risk and nearly fivefold greater likelihood of flare-free remission at 1 year. The results were published online November 14 in The New England Journal of Medicine and were presented at the annual meeting of the American College of Rheumatology (ACR).
The drug’s manufacturer, Amgen, released top-line results of the trial, called MITIGATE, in June 2024.
Until now, the mainstay of management for the chronic multiorgan disease IgG4-RD has been glucocorticoids, which can cause numerous adverse effects. “It is hoped that inebilizumab can be used as an important steroid-sparing medication in this disease to reduce steroid toxicity,” lead author John H. Stone, MD, professor of medicine at Harvard Medical School, Boston, Massachusetts, said in an interview, noting that it may not entirely eliminate the need for steroid treatment, but for many, it appears to work after the remission induction period as a monotherapy without steroids.
Asked to comment, Leonard H. Calabrese, DO, head of the Section of Clinical Immunology and manager of the Clinical Immunology Clinic at the Cleveland Clinic, Ohio, said: “There has been anecdotal or observational evidence for some effect with other immunosuppressive agents, including rituximab, but no robust clinical trial until this study. This clearly has demonstrated efficacy by reducing the risk of flares. And most importantly, putting people into remission means no active disease in any given organ. ... This gives us another tool in the toolbox to attack B cell–directed diseases, and I think it really makes a lot of sense.”
Calabrese cautioned, though, that “this is a disease that extends over many years. This is just a 1-year study. Label extensions will be important.”
And several questions remain, Calabrese noted: “How long do patients need to remain on drug? What will happen when the drug is stopped? Can they be retreated? These are the natural questions that arise in any sentinel study like this. But this is extremely encouraging. And I think it’s great for patients. I also think it’s a clarion call to increase awareness about this disease since there’s now strong evidence of effective treatment.”
Underrecognized, Often Misdiagnosed as Cancer
Indeed, IgG4-RD, a chronic, relapsing, autoimmune, fibro-inflammatory multiorgan disease, was only first described in Japan in 2003. Since then, it has been reported all over the world yet remains vastly underrecognized. It is often misdiagnosed as cancer because it produces lesions in multiple organs. It received an ICD-10 code only about a year ago. A previous study estimated a prevalence of about 5.3 persons per 100,000 but that is likely to be a three- to fourfold underestimate, said Stone, who is also executive chairman of the IgG4ward! Foundation.
“Nobody had heard of the disease until about 20 years ago. ... And there are many people in the world who have still not heard of it despite the fact that it is a multiorgan autoimmune disease and is probably as common, or more common, than many other diseases that rheumatologists spend a lot of time thinking about, such as scleroderma.”
While knowledge about the disease is increasing in rheumatology circles, it’s less well-recognized among many of the specialties where patients present, depending on the location of their lesions. These include gastroenterology, ophthalmology, pulmonary medicine, neurology, and nephrology. “All would be likely to see this disease,” Stone said.
The disease can be mistaken for tumors in many of those locations and even as metastatic cancer, he noted, adding that “any time a patient has a mass lesion in a typical organ, the pancreas, the major salivary glands, the lungs, or the kidneys, this should be on the differential diagnosis.”
The diagnosis of IgG4-RD is a clinical one, involving “quadrangulation between clinical features, serological findings, IgG4 levels in the blood, radiology studies, and then pathology biopsies when those are available,” Stone said.
Calabrese characterized the current situation as “we’re all blind men on the elephant. To the neurologist or the neurosurgeon, it’s a mass in the brain. It could present to the ophthalmologist as an [eye] tumor. It can be thyroid gland failure, pulmonary disease, retroperitoneal fibrosis, hepatobiliary disease, and beyond. So, whoever sees that patient, there’s often a long lag time in recognizing it.”
And interestingly, Stone noted that unlike other autoimmune diseases, IgG4-RD primarily affects middle-aged men rather than younger-to-middle-aged women. And when IgG4-RD is diagnosed, glucocorticoid treatment can be particularly toxic when the pancreas is involved, heightening the risk for hyperglycemia and potentially causing diabetes.
Dramatic Improvement in Flares, Remission Achievement
MITIGATE is a phase 3, multicenter, double-blind, randomized, placebo-controlled trial in which 135 adults (mean age 58.2 years, 88 men) with active IgG4-RD were randomized 1:1 to receive 300-mg intravenous infusions of inebilizumab or placebo on days 1 and 15, and again at week 26. At baseline, 62 (45.9%) participants had newly diagnosed IgG4-RD and 73 (54.1%) had recurrent disease.
Both groups received identical glucocorticoid tapers. Overall, 127 (94.1%) completed the 52 weeks of treatment.
By 52 weeks, only seven patients in the inebilizumab group (10%) had experienced disease flares vs 40 (60%) in the placebo group, a significant difference with a hazard ratio of 0.13 (P < .001).
The percentage of participants achieving flare-free, treatment-free complete remission was 59 with inebilizumab (57%), compared with just 15 (22%) in the placebo group (odds ratio [OR], 4.68; P < .001). And for flare-free, glucocorticoid-free complete remission, those proportions were 40 (59%) vs 15 (22%), respectively (OR, 4.96; P < .001).
Excluding the 8-week glucocorticoid taper period, mean total glucocorticoid use was 1264.2 mg less in the inebilizumab than the placebo group, a significant reduction. Overall, 61 participants (90%) were able to entirely discontinue glucocorticoids during the trial, compared with just 25 (37%) in the placebo group.
Adverse events of grade 3 or higher occurred in 12 participants (18%) in the inebilizumab group and 8 (12%) in the placebo group; serious adverse events occurred in 12 (18%) and 6 (9%), respectively. However, no serious adverse event occurred in more than one participant, and there were no deaths. Adverse events led to withdrawal from the trial in six patients (9%) in the inebilizumab group and three patients (4%) in the placebo group.
Adverse events that occurred in more than 10% of participants in the inebilizumab group were COVID-19 in 16 participants (24%), lymphopenia in 11 (16%), and urinary tract infection in 8 (12%).
Importantly, Stone noted, B-cell depletion can reduce responses to vaccines, so patients should receive all recommended vaccinations, including COVID-19, influenza, respiratory syncytial virus, and others, prior to initiating therapy.
Uplizna (inebilizumab-cdon) was approved by the Food and Drug Administration (FDA) for the treatment of neuromyelitis optica spectrum disorder in 2020. In October 2024, the FDA granted Amgen breakthrough therapy designation for use in IgG4-RD. The company is also developing the drug for use in myasthenia gravis.
The study was funded by Amgen. Stone has reported being a consultant for Amgen, Zenas, Argenx, Bristol Myers Squibb, Novartis, Sanofi, and Horizon Pharma. Calabrese has reported being a consultant and/or speaker for Amgen, AstraZeneca, Jansen, Sanofi, and UCB.
A version of this article first appeared on Medscape.com.
WASHINGTON — The B cell–depleting agent inebilizumab (Uplizna) dramatically reduced the risk of flares and increased year-long remission of IgG4-related disease (RD), new research has found.
In a phase 3, multicenter, double-blind, randomized, placebo-controlled trial of 135 adults with active IgG4-RD, treatment with inebilizumab resulted in a significant 87% reduction in flare risk and nearly fivefold greater likelihood of flare-free remission at 1 year. The results were published online November 14 in The New England Journal of Medicine and were presented at the annual meeting of the American College of Rheumatology (ACR).
The drug’s manufacturer, Amgen, released top-line results of the trial, called MITIGATE, in June 2024.
Until now, the mainstay of management for the chronic multiorgan disease IgG4-RD has been glucocorticoids, which can cause numerous adverse effects. “It is hoped that inebilizumab can be used as an important steroid-sparing medication in this disease to reduce steroid toxicity,” lead author John H. Stone, MD, professor of medicine at Harvard Medical School, Boston, Massachusetts, said in an interview, noting that it may not entirely eliminate the need for steroid treatment, but for many, it appears to work after the remission induction period as a monotherapy without steroids.
Asked to comment, Leonard H. Calabrese, DO, head of the Section of Clinical Immunology and manager of the Clinical Immunology Clinic at the Cleveland Clinic, Ohio, said: “There has been anecdotal or observational evidence for some effect with other immunosuppressive agents, including rituximab, but no robust clinical trial until this study. This clearly has demonstrated efficacy by reducing the risk of flares. And most importantly, putting people into remission means no active disease in any given organ. ... This gives us another tool in the toolbox to attack B cell–directed diseases, and I think it really makes a lot of sense.”
Calabrese cautioned, though, that “this is a disease that extends over many years. This is just a 1-year study. Label extensions will be important.”
And several questions remain, Calabrese noted: “How long do patients need to remain on drug? What will happen when the drug is stopped? Can they be retreated? These are the natural questions that arise in any sentinel study like this. But this is extremely encouraging. And I think it’s great for patients. I also think it’s a clarion call to increase awareness about this disease since there’s now strong evidence of effective treatment.”
Underrecognized, Often Misdiagnosed as Cancer
Indeed, IgG4-RD, a chronic, relapsing, autoimmune, fibro-inflammatory multiorgan disease, was only first described in Japan in 2003. Since then, it has been reported all over the world yet remains vastly underrecognized. It is often misdiagnosed as cancer because it produces lesions in multiple organs. It received an ICD-10 code only about a year ago. A previous study estimated a prevalence of about 5.3 persons per 100,000 but that is likely to be a three- to fourfold underestimate, said Stone, who is also executive chairman of the IgG4ward! Foundation.
“Nobody had heard of the disease until about 20 years ago. ... And there are many people in the world who have still not heard of it despite the fact that it is a multiorgan autoimmune disease and is probably as common, or more common, than many other diseases that rheumatologists spend a lot of time thinking about, such as scleroderma.”
While knowledge about the disease is increasing in rheumatology circles, it’s less well-recognized among many of the specialties where patients present, depending on the location of their lesions. These include gastroenterology, ophthalmology, pulmonary medicine, neurology, and nephrology. “All would be likely to see this disease,” Stone said.
The disease can be mistaken for tumors in many of those locations and even as metastatic cancer, he noted, adding that “any time a patient has a mass lesion in a typical organ, the pancreas, the major salivary glands, the lungs, or the kidneys, this should be on the differential diagnosis.”
The diagnosis of IgG4-RD is a clinical one, involving “quadrangulation between clinical features, serological findings, IgG4 levels in the blood, radiology studies, and then pathology biopsies when those are available,” Stone said.
Calabrese characterized the current situation as “we’re all blind men on the elephant. To the neurologist or the neurosurgeon, it’s a mass in the brain. It could present to the ophthalmologist as an [eye] tumor. It can be thyroid gland failure, pulmonary disease, retroperitoneal fibrosis, hepatobiliary disease, and beyond. So, whoever sees that patient, there’s often a long lag time in recognizing it.”
And interestingly, Stone noted that unlike other autoimmune diseases, IgG4-RD primarily affects middle-aged men rather than younger-to-middle-aged women. And when IgG4-RD is diagnosed, glucocorticoid treatment can be particularly toxic when the pancreas is involved, heightening the risk for hyperglycemia and potentially causing diabetes.
Dramatic Improvement in Flares, Remission Achievement
MITIGATE is a phase 3, multicenter, double-blind, randomized, placebo-controlled trial in which 135 adults (mean age 58.2 years, 88 men) with active IgG4-RD were randomized 1:1 to receive 300-mg intravenous infusions of inebilizumab or placebo on days 1 and 15, and again at week 26. At baseline, 62 (45.9%) participants had newly diagnosed IgG4-RD and 73 (54.1%) had recurrent disease.
Both groups received identical glucocorticoid tapers. Overall, 127 (94.1%) completed the 52 weeks of treatment.
By 52 weeks, only seven patients in the inebilizumab group (10%) had experienced disease flares vs 40 (60%) in the placebo group, a significant difference with a hazard ratio of 0.13 (P < .001).
The percentage of participants achieving flare-free, treatment-free complete remission was 59 with inebilizumab (57%), compared with just 15 (22%) in the placebo group (odds ratio [OR], 4.68; P < .001). And for flare-free, glucocorticoid-free complete remission, those proportions were 40 (59%) vs 15 (22%), respectively (OR, 4.96; P < .001).
Excluding the 8-week glucocorticoid taper period, mean total glucocorticoid use was 1264.2 mg less in the inebilizumab than the placebo group, a significant reduction. Overall, 61 participants (90%) were able to entirely discontinue glucocorticoids during the trial, compared with just 25 (37%) in the placebo group.
Adverse events of grade 3 or higher occurred in 12 participants (18%) in the inebilizumab group and 8 (12%) in the placebo group; serious adverse events occurred in 12 (18%) and 6 (9%), respectively. However, no serious adverse event occurred in more than one participant, and there were no deaths. Adverse events led to withdrawal from the trial in six patients (9%) in the inebilizumab group and three patients (4%) in the placebo group.
Adverse events that occurred in more than 10% of participants in the inebilizumab group were COVID-19 in 16 participants (24%), lymphopenia in 11 (16%), and urinary tract infection in 8 (12%).
Importantly, Stone noted, B-cell depletion can reduce responses to vaccines, so patients should receive all recommended vaccinations, including COVID-19, influenza, respiratory syncytial virus, and others, prior to initiating therapy.
Uplizna (inebilizumab-cdon) was approved by the Food and Drug Administration (FDA) for the treatment of neuromyelitis optica spectrum disorder in 2020. In October 2024, the FDA granted Amgen breakthrough therapy designation for use in IgG4-RD. The company is also developing the drug for use in myasthenia gravis.
The study was funded by Amgen. Stone has reported being a consultant for Amgen, Zenas, Argenx, Bristol Myers Squibb, Novartis, Sanofi, and Horizon Pharma. Calabrese has reported being a consultant and/or speaker for Amgen, AstraZeneca, Jansen, Sanofi, and UCB.
A version of this article first appeared on Medscape.com.
FROM ACR 2024
The Use of Biomarkers for Alzheimer’s Disease in Primary Care
In our previous case-based review, I teased the opportunity to use biomarkers to increase the accuracy and expediency of the diagnosis of Alzheimer’s disease (AD). These tests are no longer confined to the research setting but are now available to specialists and primary care clinicians alike. Given that most cognitive disorders are first identified in primary care, however, I believe that their greatest impact will be in our clinical space.
The pathologic processes associated with AD can be detected approximately 2 decades before the advent of clinical symptoms, and the symptomatic period of cognitive impairment is estimated to occupy just the final third of the disease course of AD. Using imaging studies, primarily PET, as well as cerebrospinal fluid (CSF) and even blood biomarkers for beta amyloid and tau, the pathologic drivers of AD, clinicians can identify patients with AD pathology before any symptoms are present. Importantly for our present-day interventions, the application of biomarkers can also help to diagnose AD earlier.
Amyloid PET identifies one of the earliest markers of potential AD, but a barrier common to advanced diagnostic imaging has been cost. Medicare has now approved coverage for amyloid PET in cases of suspected cognitive impairment. In a large study of more than 16,000 older adults in the United States, PET scans were positive in 55.3% of cases with mild cognitive impairment (MCI). The PET positivity rate among adults with other dementia was 70.1%. The application of PET resulted in a change in care in more than 60% of patients with MCI and dementia. One quarter of participants had their diagnosis changed from AD to another form of dementia, and 10% were changed from a diagnosis of other dementia to AD.
Liquid biomarkers can involve either CSF or blood samples. To date, CSF testing has yielded more consistent results and has defined protocols for assessment. Still, collection of CSF is more challenging than collection of blood, and patients and their families may object to lumbar puncture. CSF assessment therefore remains generally in the province of specialists and research centers.
Primary care clinicians have been waiting for a reliable blood-based biomarker for AD, and that wait may be about to end. A study published in July 2024 included 1213 adults being evaluated for cognitive symptoms in Sweden. They completed a test measuring the ratio of phosphorylated tau 217 vs nonphosphorylated tau 217, with or without a test for serum amyloid ratios as well. These tests were compared with clinicians’ clinical diagnoses as well as CSF results, which were considered the gold standard.
Using only clinical tools, primary care clinicians’ and specialists’ diagnostic accuracy for MCI and dementia were just 61% and 73%, respectively. These values were substantially weaker vs the performance of either the serum tau or amyloid ratios (both 90% accurate). The authors concluded that serum testing has the potential to improve clinical care of patients with cognitive impairment.
Where does that leave us today? Commercially available blood biomarkers are available now which use different tests and cutoff values. These may be helpful but will probably be difficult to compare and interpret for primary care clinicians. In addition, insurance is less likely to cover these tests. Amyloid PET scans are a very reasonable option to augment clinician judgment of suspected cognitive impairment, but not all geographic areas will have ready access to this imaging study.
Still, it is an exciting time to have more objective tools at our disposal to identify MCI and AD. These tools can only be optimized by clinicians who recognize symptoms and perform the baseline testing necessary to determine pretest probability of MCI or dementia.
Charles P. Vega, Health Sciences Clinical Professor, Family Medicine, University of California, Irvine, has disclosed the following relevant financial relationships: Serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for McNeil Pharmaceuticals.
A version of this article first appeared on Medscape.com.
In our previous case-based review, I teased the opportunity to use biomarkers to increase the accuracy and expediency of the diagnosis of Alzheimer’s disease (AD). These tests are no longer confined to the research setting but are now available to specialists and primary care clinicians alike. Given that most cognitive disorders are first identified in primary care, however, I believe that their greatest impact will be in our clinical space.
The pathologic processes associated with AD can be detected approximately 2 decades before the advent of clinical symptoms, and the symptomatic period of cognitive impairment is estimated to occupy just the final third of the disease course of AD. Using imaging studies, primarily PET, as well as cerebrospinal fluid (CSF) and even blood biomarkers for beta amyloid and tau, the pathologic drivers of AD, clinicians can identify patients with AD pathology before any symptoms are present. Importantly for our present-day interventions, the application of biomarkers can also help to diagnose AD earlier.
Amyloid PET identifies one of the earliest markers of potential AD, but a barrier common to advanced diagnostic imaging has been cost. Medicare has now approved coverage for amyloid PET in cases of suspected cognitive impairment. In a large study of more than 16,000 older adults in the United States, PET scans were positive in 55.3% of cases with mild cognitive impairment (MCI). The PET positivity rate among adults with other dementia was 70.1%. The application of PET resulted in a change in care in more than 60% of patients with MCI and dementia. One quarter of participants had their diagnosis changed from AD to another form of dementia, and 10% were changed from a diagnosis of other dementia to AD.
Liquid biomarkers can involve either CSF or blood samples. To date, CSF testing has yielded more consistent results and has defined protocols for assessment. Still, collection of CSF is more challenging than collection of blood, and patients and their families may object to lumbar puncture. CSF assessment therefore remains generally in the province of specialists and research centers.
Primary care clinicians have been waiting for a reliable blood-based biomarker for AD, and that wait may be about to end. A study published in July 2024 included 1213 adults being evaluated for cognitive symptoms in Sweden. They completed a test measuring the ratio of phosphorylated tau 217 vs nonphosphorylated tau 217, with or without a test for serum amyloid ratios as well. These tests were compared with clinicians’ clinical diagnoses as well as CSF results, which were considered the gold standard.
Using only clinical tools, primary care clinicians’ and specialists’ diagnostic accuracy for MCI and dementia were just 61% and 73%, respectively. These values were substantially weaker vs the performance of either the serum tau or amyloid ratios (both 90% accurate). The authors concluded that serum testing has the potential to improve clinical care of patients with cognitive impairment.
Where does that leave us today? Commercially available blood biomarkers are available now which use different tests and cutoff values. These may be helpful but will probably be difficult to compare and interpret for primary care clinicians. In addition, insurance is less likely to cover these tests. Amyloid PET scans are a very reasonable option to augment clinician judgment of suspected cognitive impairment, but not all geographic areas will have ready access to this imaging study.
Still, it is an exciting time to have more objective tools at our disposal to identify MCI and AD. These tools can only be optimized by clinicians who recognize symptoms and perform the baseline testing necessary to determine pretest probability of MCI or dementia.
Charles P. Vega, Health Sciences Clinical Professor, Family Medicine, University of California, Irvine, has disclosed the following relevant financial relationships: Serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for McNeil Pharmaceuticals.
A version of this article first appeared on Medscape.com.
In our previous case-based review, I teased the opportunity to use biomarkers to increase the accuracy and expediency of the diagnosis of Alzheimer’s disease (AD). These tests are no longer confined to the research setting but are now available to specialists and primary care clinicians alike. Given that most cognitive disorders are first identified in primary care, however, I believe that their greatest impact will be in our clinical space.
The pathologic processes associated with AD can be detected approximately 2 decades before the advent of clinical symptoms, and the symptomatic period of cognitive impairment is estimated to occupy just the final third of the disease course of AD. Using imaging studies, primarily PET, as well as cerebrospinal fluid (CSF) and even blood biomarkers for beta amyloid and tau, the pathologic drivers of AD, clinicians can identify patients with AD pathology before any symptoms are present. Importantly for our present-day interventions, the application of biomarkers can also help to diagnose AD earlier.
Amyloid PET identifies one of the earliest markers of potential AD, but a barrier common to advanced diagnostic imaging has been cost. Medicare has now approved coverage for amyloid PET in cases of suspected cognitive impairment. In a large study of more than 16,000 older adults in the United States, PET scans were positive in 55.3% of cases with mild cognitive impairment (MCI). The PET positivity rate among adults with other dementia was 70.1%. The application of PET resulted in a change in care in more than 60% of patients with MCI and dementia. One quarter of participants had their diagnosis changed from AD to another form of dementia, and 10% were changed from a diagnosis of other dementia to AD.
Liquid biomarkers can involve either CSF or blood samples. To date, CSF testing has yielded more consistent results and has defined protocols for assessment. Still, collection of CSF is more challenging than collection of blood, and patients and their families may object to lumbar puncture. CSF assessment therefore remains generally in the province of specialists and research centers.
Primary care clinicians have been waiting for a reliable blood-based biomarker for AD, and that wait may be about to end. A study published in July 2024 included 1213 adults being evaluated for cognitive symptoms in Sweden. They completed a test measuring the ratio of phosphorylated tau 217 vs nonphosphorylated tau 217, with or without a test for serum amyloid ratios as well. These tests were compared with clinicians’ clinical diagnoses as well as CSF results, which were considered the gold standard.
Using only clinical tools, primary care clinicians’ and specialists’ diagnostic accuracy for MCI and dementia were just 61% and 73%, respectively. These values were substantially weaker vs the performance of either the serum tau or amyloid ratios (both 90% accurate). The authors concluded that serum testing has the potential to improve clinical care of patients with cognitive impairment.
Where does that leave us today? Commercially available blood biomarkers are available now which use different tests and cutoff values. These may be helpful but will probably be difficult to compare and interpret for primary care clinicians. In addition, insurance is less likely to cover these tests. Amyloid PET scans are a very reasonable option to augment clinician judgment of suspected cognitive impairment, but not all geographic areas will have ready access to this imaging study.
Still, it is an exciting time to have more objective tools at our disposal to identify MCI and AD. These tools can only be optimized by clinicians who recognize symptoms and perform the baseline testing necessary to determine pretest probability of MCI or dementia.
Charles P. Vega, Health Sciences Clinical Professor, Family Medicine, University of California, Irvine, has disclosed the following relevant financial relationships: Serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for McNeil Pharmaceuticals.
A version of this article first appeared on Medscape.com.
A New and Early Predictor of Dementia?
, in new findings that may provide a potential opportunity to identify high-risk populations for targeted enrollment in clinical trials of dementia prevention and treatment.
Results of an international study assessing frailty trajectories showed frailty levels notably increased in the 4-9 years before dementia diagnosis. Even among study participants whose baseline frailty measurement was taken prior to that acceleration period, frailty was still positively associated with dementia risk, the investigators noted.
“We found that with every four to five additional health problems, there is on average a 40% higher risk of developing dementia, while the risk is lower for people who are more physically fit,” said study investigator David Ward, PhD, of the Centre for Health Services Research, The University of Queensland, Brisbane, Australia.
The findings were published online in JAMA Neurology.
A Promising Biomarker
An accessible biomarker for both biologic age and dementia risk is essential for advancing dementia prevention and treatment strategies, the investigators noted, adding that growing evidence suggests frailty may be a promising candidate for this role.
To learn more about the association between frailty and dementia, Ward and his team analyzed data on 29,849 participants aged 60 years or above (mean age, 71.6 years; 62% women) who participated in four cohort studies: the English Longitudinal Study of Ageing (ELSA; n = 6771), the Health and Retirement Study (HRS; n = 9045), the Rush Memory and Aging Project (MAP; n = 1451), and the National Alzheimer’s Coordinating Center (NACC; n = 12,582).
The primary outcome was all-cause dementia. Depending on the cohort, dementia diagnoses were determined through cognitive testing, self- or family report of physician diagnosis, or a diagnosis by the study physician. Participants were excluded if they had cognitive impairment at baseline.
Investigators retrospectively determined frailty index scores by gathering information on health and functional outcomes for participants from each cohort. Only participants with frailty data on at least 30 deficits were included.
Commonly included deficits included high blood pressure, cancer, and chronic pain, as well as functional problems such as hearing impairment, difficulty with mobility, and challenges managing finances.
Investigators conducted follow-up visits with participants until they developed dementia or until the study ended, with follow-up periods varying across cohorts.
After adjustment for potential confounders, frailty scores were modeled using backward time scales.
Among participants who developed incident dementia (n = 3154), covariate-adjusted expected frailty index scores were, on average, higher in women than in men by 18.5% in ELSA, 20.9% in HRS, and 16.2% in MAP. There were no differences in frailty scores between sexes in the NACC cohort.
When measured on a timeline, as compared with those who didn’t develop dementia, frailty scores were significantly and consistently higher in the dementia groups 8-20 before dementia onset (20 years in HRS; 13 in MAP; 12 in ELSA; 8 in NACC).
Increases in the rates of frailty index scores began accelerating 4-9 years before dementia onset for the various cohorts, investigators noted.
In all four cohorts, each 0.1 increase in frailty scores was positively associated with increased dementia risk.
Adjusted hazard ratios [aHRs] ranged from 1.18 in the HRS cohort to 1.73 in the NACC cohort, which showed the strongest association.
In participants whose baseline frailty measurement was conducted before the predementia acceleration period began, the association of frailty scores and dementia risk was positive. These aHRs ranged from 1.18 in the HRS cohort to 1.43 in the NACC cohort.
The ‘Four Pillars’ of Prevention
The good news, investigators said, is that the long trajectory of frailty symptoms preceding dementia onset provides plenty of opportunity for intervention.
To slow the development of frailty, Ward suggested adhering to the “four pillars of frailty prevention and management,” which include good nutrition with plenty of protein, exercise, optimizing medications for chronic conditions, and maintaining a strong social network.
Ward suggested neurologists track frailty in their patients and pointed to a recent article focused on helping neurologists use frailty measures to influence care planning.
Study limitations include the possibility of reverse causality and the fact that investigators could not adjust for genetic risk for dementia.
Unclear Pathway
Commenting on the findings, Lycia Neumann, PhD, senior director of Health Services Research at the Alzheimer’s Association, noted that many studies over the years have shown a link between frailty and dementia. However, she cautioned that a link does not imply causation.
The pathway from frailty to dementia is not 100% clear, and both are complex conditions, said Neumann, who was not part of the study.
“Adopting healthy lifestyle behaviors early and consistently can help decrease the risk of — or postpone the onset of — both frailty and cognitive decline,” she said. Neumann added that physical activity, a healthy diet, social engagement, and controlling diabetes and blood pressure can also reduce the risk for dementia as well as cardiovascular disease.
The study was funded in part by the Deep Dementia Phenotyping Network through the Frailty and Dementia Special Interest Group. Ward and Neumann reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
, in new findings that may provide a potential opportunity to identify high-risk populations for targeted enrollment in clinical trials of dementia prevention and treatment.
Results of an international study assessing frailty trajectories showed frailty levels notably increased in the 4-9 years before dementia diagnosis. Even among study participants whose baseline frailty measurement was taken prior to that acceleration period, frailty was still positively associated with dementia risk, the investigators noted.
“We found that with every four to five additional health problems, there is on average a 40% higher risk of developing dementia, while the risk is lower for people who are more physically fit,” said study investigator David Ward, PhD, of the Centre for Health Services Research, The University of Queensland, Brisbane, Australia.
The findings were published online in JAMA Neurology.
A Promising Biomarker
An accessible biomarker for both biologic age and dementia risk is essential for advancing dementia prevention and treatment strategies, the investigators noted, adding that growing evidence suggests frailty may be a promising candidate for this role.
To learn more about the association between frailty and dementia, Ward and his team analyzed data on 29,849 participants aged 60 years or above (mean age, 71.6 years; 62% women) who participated in four cohort studies: the English Longitudinal Study of Ageing (ELSA; n = 6771), the Health and Retirement Study (HRS; n = 9045), the Rush Memory and Aging Project (MAP; n = 1451), and the National Alzheimer’s Coordinating Center (NACC; n = 12,582).
The primary outcome was all-cause dementia. Depending on the cohort, dementia diagnoses were determined through cognitive testing, self- or family report of physician diagnosis, or a diagnosis by the study physician. Participants were excluded if they had cognitive impairment at baseline.
Investigators retrospectively determined frailty index scores by gathering information on health and functional outcomes for participants from each cohort. Only participants with frailty data on at least 30 deficits were included.
Commonly included deficits included high blood pressure, cancer, and chronic pain, as well as functional problems such as hearing impairment, difficulty with mobility, and challenges managing finances.
Investigators conducted follow-up visits with participants until they developed dementia or until the study ended, with follow-up periods varying across cohorts.
After adjustment for potential confounders, frailty scores were modeled using backward time scales.
Among participants who developed incident dementia (n = 3154), covariate-adjusted expected frailty index scores were, on average, higher in women than in men by 18.5% in ELSA, 20.9% in HRS, and 16.2% in MAP. There were no differences in frailty scores between sexes in the NACC cohort.
When measured on a timeline, as compared with those who didn’t develop dementia, frailty scores were significantly and consistently higher in the dementia groups 8-20 before dementia onset (20 years in HRS; 13 in MAP; 12 in ELSA; 8 in NACC).
Increases in the rates of frailty index scores began accelerating 4-9 years before dementia onset for the various cohorts, investigators noted.
In all four cohorts, each 0.1 increase in frailty scores was positively associated with increased dementia risk.
Adjusted hazard ratios [aHRs] ranged from 1.18 in the HRS cohort to 1.73 in the NACC cohort, which showed the strongest association.
In participants whose baseline frailty measurement was conducted before the predementia acceleration period began, the association of frailty scores and dementia risk was positive. These aHRs ranged from 1.18 in the HRS cohort to 1.43 in the NACC cohort.
The ‘Four Pillars’ of Prevention
The good news, investigators said, is that the long trajectory of frailty symptoms preceding dementia onset provides plenty of opportunity for intervention.
To slow the development of frailty, Ward suggested adhering to the “four pillars of frailty prevention and management,” which include good nutrition with plenty of protein, exercise, optimizing medications for chronic conditions, and maintaining a strong social network.
Ward suggested neurologists track frailty in their patients and pointed to a recent article focused on helping neurologists use frailty measures to influence care planning.
Study limitations include the possibility of reverse causality and the fact that investigators could not adjust for genetic risk for dementia.
Unclear Pathway
Commenting on the findings, Lycia Neumann, PhD, senior director of Health Services Research at the Alzheimer’s Association, noted that many studies over the years have shown a link between frailty and dementia. However, she cautioned that a link does not imply causation.
The pathway from frailty to dementia is not 100% clear, and both are complex conditions, said Neumann, who was not part of the study.
“Adopting healthy lifestyle behaviors early and consistently can help decrease the risk of — or postpone the onset of — both frailty and cognitive decline,” she said. Neumann added that physical activity, a healthy diet, social engagement, and controlling diabetes and blood pressure can also reduce the risk for dementia as well as cardiovascular disease.
The study was funded in part by the Deep Dementia Phenotyping Network through the Frailty and Dementia Special Interest Group. Ward and Neumann reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
, in new findings that may provide a potential opportunity to identify high-risk populations for targeted enrollment in clinical trials of dementia prevention and treatment.
Results of an international study assessing frailty trajectories showed frailty levels notably increased in the 4-9 years before dementia diagnosis. Even among study participants whose baseline frailty measurement was taken prior to that acceleration period, frailty was still positively associated with dementia risk, the investigators noted.
“We found that with every four to five additional health problems, there is on average a 40% higher risk of developing dementia, while the risk is lower for people who are more physically fit,” said study investigator David Ward, PhD, of the Centre for Health Services Research, The University of Queensland, Brisbane, Australia.
The findings were published online in JAMA Neurology.
A Promising Biomarker
An accessible biomarker for both biologic age and dementia risk is essential for advancing dementia prevention and treatment strategies, the investigators noted, adding that growing evidence suggests frailty may be a promising candidate for this role.
To learn more about the association between frailty and dementia, Ward and his team analyzed data on 29,849 participants aged 60 years or above (mean age, 71.6 years; 62% women) who participated in four cohort studies: the English Longitudinal Study of Ageing (ELSA; n = 6771), the Health and Retirement Study (HRS; n = 9045), the Rush Memory and Aging Project (MAP; n = 1451), and the National Alzheimer’s Coordinating Center (NACC; n = 12,582).
The primary outcome was all-cause dementia. Depending on the cohort, dementia diagnoses were determined through cognitive testing, self- or family report of physician diagnosis, or a diagnosis by the study physician. Participants were excluded if they had cognitive impairment at baseline.
Investigators retrospectively determined frailty index scores by gathering information on health and functional outcomes for participants from each cohort. Only participants with frailty data on at least 30 deficits were included.
Commonly included deficits included high blood pressure, cancer, and chronic pain, as well as functional problems such as hearing impairment, difficulty with mobility, and challenges managing finances.
Investigators conducted follow-up visits with participants until they developed dementia or until the study ended, with follow-up periods varying across cohorts.
After adjustment for potential confounders, frailty scores were modeled using backward time scales.
Among participants who developed incident dementia (n = 3154), covariate-adjusted expected frailty index scores were, on average, higher in women than in men by 18.5% in ELSA, 20.9% in HRS, and 16.2% in MAP. There were no differences in frailty scores between sexes in the NACC cohort.
When measured on a timeline, as compared with those who didn’t develop dementia, frailty scores were significantly and consistently higher in the dementia groups 8-20 before dementia onset (20 years in HRS; 13 in MAP; 12 in ELSA; 8 in NACC).
Increases in the rates of frailty index scores began accelerating 4-9 years before dementia onset for the various cohorts, investigators noted.
In all four cohorts, each 0.1 increase in frailty scores was positively associated with increased dementia risk.
Adjusted hazard ratios [aHRs] ranged from 1.18 in the HRS cohort to 1.73 in the NACC cohort, which showed the strongest association.
In participants whose baseline frailty measurement was conducted before the predementia acceleration period began, the association of frailty scores and dementia risk was positive. These aHRs ranged from 1.18 in the HRS cohort to 1.43 in the NACC cohort.
The ‘Four Pillars’ of Prevention
The good news, investigators said, is that the long trajectory of frailty symptoms preceding dementia onset provides plenty of opportunity for intervention.
To slow the development of frailty, Ward suggested adhering to the “four pillars of frailty prevention and management,” which include good nutrition with plenty of protein, exercise, optimizing medications for chronic conditions, and maintaining a strong social network.
Ward suggested neurologists track frailty in their patients and pointed to a recent article focused on helping neurologists use frailty measures to influence care planning.
Study limitations include the possibility of reverse causality and the fact that investigators could not adjust for genetic risk for dementia.
Unclear Pathway
Commenting on the findings, Lycia Neumann, PhD, senior director of Health Services Research at the Alzheimer’s Association, noted that many studies over the years have shown a link between frailty and dementia. However, she cautioned that a link does not imply causation.
The pathway from frailty to dementia is not 100% clear, and both are complex conditions, said Neumann, who was not part of the study.
“Adopting healthy lifestyle behaviors early and consistently can help decrease the risk of — or postpone the onset of — both frailty and cognitive decline,” she said. Neumann added that physical activity, a healthy diet, social engagement, and controlling diabetes and blood pressure can also reduce the risk for dementia as well as cardiovascular disease.
The study was funded in part by the Deep Dementia Phenotyping Network through the Frailty and Dementia Special Interest Group. Ward and Neumann reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Vitamin K Supplementation Reduces Nocturnal Leg Cramps in Older Adults
TOPLINE:
Vitamin K supplementation significantly reduced the frequency, intensity, and duration of nocturnal leg cramps in older adults. No adverse events related to vitamin K were identified.
METHODOLOGY:
- Researchers conducted a multicenter, double-blind, placebo-controlled randomized clinical trial in China from September 2022 to December 2023.
- A total of 199 participants aged ≥ 65 years with at least two documented episodes of nocturnal leg cramps during a 2-week screening period were included.
- Participants were randomized in a 1:1 ratio to receive either 180 μg of vitamin K (menaquinone 7) or a placebo daily for 8 weeks.
- The primary outcome was the mean number of nocturnal leg cramps per week, while secondary outcomes were the duration and severity of muscle cramps.
- The ethics committees of Third People’s Hospital of Chengdu and Affiliated Hospital of North Sichuan Medical College approved the study, and all participants provided written informed consent.
TAKEAWAY:
- Vitamin K group experienced a significant reduction in the mean weekly frequency of cramps (mean difference, 2.60 [SD, 0.81] to 0.96 [SD, 1.41]) compared with the placebo group, which maintained a mean weekly frequency of 3.63 (SD, 2.20) (P < .001).
- The severity of nocturnal leg cramps decreased more in the vitamin K group (mean difference, −2.55 [SD, 2.12] points) than in the placebo group (mean difference, −1.24 [SD, 1.16] points).
- The duration of nocturnal leg cramps also decreased more in the vitamin K group (mean difference, −0.90 [SD, 0.88] minutes) than in the placebo group (mean difference, −0.32 [SD, 0.78] minutes).
- No adverse events related to vitamin K use were identified, indicating a good safety profile for the supplementation.
IN PRACTICE:
“Given the generally benign characteristics of NLCs, treatment modality must be both effective and safe, thus minimizing the risk of iatrogenic harm,” the study authors wrote.
SOURCE:
This study was led by Jing Tan, MD, the Third People’s Hospital of Chengdu in Chengdu, China. It was published online on October 28 in JAMA Internal Medicine.
LIMITATIONS:
This study did not investigate the quality of life or sleep, which could have provided additional insights into the impact of vitamin K on nocturnal leg cramps. The relatively mild nature of nocturnal leg cramps experienced by the participants may limit the generalizability of the findings to populations with more severe symptoms.
DISCLOSURES:
This study was supported by grants from China Health Promotion Foundation and the Third People’s Hospital of Chengdu Scientific Research Project. Tan disclosed receiving personal fees from BeiGene, AbbVie, Pfizer, Xian Janssen Pharmaceutical, and Takeda Pharmaceutical outside the submitted work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Vitamin K supplementation significantly reduced the frequency, intensity, and duration of nocturnal leg cramps in older adults. No adverse events related to vitamin K were identified.
METHODOLOGY:
- Researchers conducted a multicenter, double-blind, placebo-controlled randomized clinical trial in China from September 2022 to December 2023.
- A total of 199 participants aged ≥ 65 years with at least two documented episodes of nocturnal leg cramps during a 2-week screening period were included.
- Participants were randomized in a 1:1 ratio to receive either 180 μg of vitamin K (menaquinone 7) or a placebo daily for 8 weeks.
- The primary outcome was the mean number of nocturnal leg cramps per week, while secondary outcomes were the duration and severity of muscle cramps.
- The ethics committees of Third People’s Hospital of Chengdu and Affiliated Hospital of North Sichuan Medical College approved the study, and all participants provided written informed consent.
TAKEAWAY:
- Vitamin K group experienced a significant reduction in the mean weekly frequency of cramps (mean difference, 2.60 [SD, 0.81] to 0.96 [SD, 1.41]) compared with the placebo group, which maintained a mean weekly frequency of 3.63 (SD, 2.20) (P < .001).
- The severity of nocturnal leg cramps decreased more in the vitamin K group (mean difference, −2.55 [SD, 2.12] points) than in the placebo group (mean difference, −1.24 [SD, 1.16] points).
- The duration of nocturnal leg cramps also decreased more in the vitamin K group (mean difference, −0.90 [SD, 0.88] minutes) than in the placebo group (mean difference, −0.32 [SD, 0.78] minutes).
- No adverse events related to vitamin K use were identified, indicating a good safety profile for the supplementation.
IN PRACTICE:
“Given the generally benign characteristics of NLCs, treatment modality must be both effective and safe, thus minimizing the risk of iatrogenic harm,” the study authors wrote.
SOURCE:
This study was led by Jing Tan, MD, the Third People’s Hospital of Chengdu in Chengdu, China. It was published online on October 28 in JAMA Internal Medicine.
LIMITATIONS:
This study did not investigate the quality of life or sleep, which could have provided additional insights into the impact of vitamin K on nocturnal leg cramps. The relatively mild nature of nocturnal leg cramps experienced by the participants may limit the generalizability of the findings to populations with more severe symptoms.
DISCLOSURES:
This study was supported by grants from China Health Promotion Foundation and the Third People’s Hospital of Chengdu Scientific Research Project. Tan disclosed receiving personal fees from BeiGene, AbbVie, Pfizer, Xian Janssen Pharmaceutical, and Takeda Pharmaceutical outside the submitted work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Vitamin K supplementation significantly reduced the frequency, intensity, and duration of nocturnal leg cramps in older adults. No adverse events related to vitamin K were identified.
METHODOLOGY:
- Researchers conducted a multicenter, double-blind, placebo-controlled randomized clinical trial in China from September 2022 to December 2023.
- A total of 199 participants aged ≥ 65 years with at least two documented episodes of nocturnal leg cramps during a 2-week screening period were included.
- Participants were randomized in a 1:1 ratio to receive either 180 μg of vitamin K (menaquinone 7) or a placebo daily for 8 weeks.
- The primary outcome was the mean number of nocturnal leg cramps per week, while secondary outcomes were the duration and severity of muscle cramps.
- The ethics committees of Third People’s Hospital of Chengdu and Affiliated Hospital of North Sichuan Medical College approved the study, and all participants provided written informed consent.
TAKEAWAY:
- Vitamin K group experienced a significant reduction in the mean weekly frequency of cramps (mean difference, 2.60 [SD, 0.81] to 0.96 [SD, 1.41]) compared with the placebo group, which maintained a mean weekly frequency of 3.63 (SD, 2.20) (P < .001).
- The severity of nocturnal leg cramps decreased more in the vitamin K group (mean difference, −2.55 [SD, 2.12] points) than in the placebo group (mean difference, −1.24 [SD, 1.16] points).
- The duration of nocturnal leg cramps also decreased more in the vitamin K group (mean difference, −0.90 [SD, 0.88] minutes) than in the placebo group (mean difference, −0.32 [SD, 0.78] minutes).
- No adverse events related to vitamin K use were identified, indicating a good safety profile for the supplementation.
IN PRACTICE:
“Given the generally benign characteristics of NLCs, treatment modality must be both effective and safe, thus minimizing the risk of iatrogenic harm,” the study authors wrote.
SOURCE:
This study was led by Jing Tan, MD, the Third People’s Hospital of Chengdu in Chengdu, China. It was published online on October 28 in JAMA Internal Medicine.
LIMITATIONS:
This study did not investigate the quality of life or sleep, which could have provided additional insights into the impact of vitamin K on nocturnal leg cramps. The relatively mild nature of nocturnal leg cramps experienced by the participants may limit the generalizability of the findings to populations with more severe symptoms.
DISCLOSURES:
This study was supported by grants from China Health Promotion Foundation and the Third People’s Hospital of Chengdu Scientific Research Project. Tan disclosed receiving personal fees from BeiGene, AbbVie, Pfizer, Xian Janssen Pharmaceutical, and Takeda Pharmaceutical outside the submitted work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Scurvy: A Diagnosis Still Relevant Today
“Petechial rash often prompts further investigation into hematological, dermatological, or vasculitis causes. However, if the above investigations are negative and skin biopsy has not revealed a cause, there is a Renaissance-era diagnosis that is often overlooked but is easily investigated and treated,” wrote Andrew Dermawan, MD, and colleagues from Sir Charles Gairdner Hospital in Nedlands, Australia, in BMJ Case Reports. The diagnosis they highlight is scurvy, a disease that has faded from common medical concern but is reemerging, partly because of the rise in bariatric surgery.
Diagnosing Scurvy in the 2020s
In their article, Dermawan and colleagues present the case of a 50-year-old man with a bilateral petechial rash on his lower limbs, without any history of trauma. The patient, who exhibited no infectious symptoms, also had gross hematuria, microcytic anemia, mild neutropenia, and lymphopenia. Tests for autoimmune and hematological diseases were negative, as were abdominal and leg CT scans, ruling out abdominal hemorrhage and vasculitis. Additionally, a skin biopsy showed no causative findings.
The doctors noted that the patient had undergone sleeve gastrectomy, prompting them to inquire about his diet. They discovered that, because of financial difficulties, his diet primarily consisted of processed foods with little to no fruits or vegetables, and he had stopped taking supplements recommended by his gastroenterologist. Further tests revealed a vitamin D deficiency and a severe deficiency in vitamin C. With the diagnosis of scurvy confirmed, the doctors treated the patient with 1000 mg of ascorbic acid daily, along with cholecalciferol, folic acid, and a multivitamin complex, leading to a complete resolution of his symptoms.
Risk Factors Then and Now
It can cause mucosal and gastric hemorrhages, and if left untreated, it can lead to fatal bleeding.
Historically known as “sailors’ disease,” scurvy plagued men on long voyages who lacked access to fresh fruits or vegetables and thus did not get enough vitamin C. In 1747, James Lind, a British physician in the Royal Navy, demonstrated that the consumption of oranges and lemons could combat scurvy.
Today’s risk factors for scurvy include malnutrition, gastrointestinal disorders (eg, chronic inflammatory bowel diseases), alcohol and tobacco use, eating disorders, psychiatric illnesses, dialysis, and the use of medications that reduce the absorption of ascorbic acid (such as corticosteroids and proton pump inhibitors).
Scurvy remains more common among individuals with unfavorable socioeconomic conditions. The authors of the study emphasize how the rising cost of living — specifically in Australia but applicable elsewhere — is changing eating habits, leading to a high consumption of low-cost, nutritionally poor foods.
Poverty has always been a risk factor for scurvy, but today there may be an additional cause: bariatric surgery. Patients undergoing these procedures are at a risk for deficiencies in fat-soluble vitamins A, D, E, and K, and if their diet is inadequate, they may also experience a vitamin C deficiency. Awareness of this can facilitate the timely diagnosis of scurvy in these patients.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
“Petechial rash often prompts further investigation into hematological, dermatological, or vasculitis causes. However, if the above investigations are negative and skin biopsy has not revealed a cause, there is a Renaissance-era diagnosis that is often overlooked but is easily investigated and treated,” wrote Andrew Dermawan, MD, and colleagues from Sir Charles Gairdner Hospital in Nedlands, Australia, in BMJ Case Reports. The diagnosis they highlight is scurvy, a disease that has faded from common medical concern but is reemerging, partly because of the rise in bariatric surgery.
Diagnosing Scurvy in the 2020s
In their article, Dermawan and colleagues present the case of a 50-year-old man with a bilateral petechial rash on his lower limbs, without any history of trauma. The patient, who exhibited no infectious symptoms, also had gross hematuria, microcytic anemia, mild neutropenia, and lymphopenia. Tests for autoimmune and hematological diseases were negative, as were abdominal and leg CT scans, ruling out abdominal hemorrhage and vasculitis. Additionally, a skin biopsy showed no causative findings.
The doctors noted that the patient had undergone sleeve gastrectomy, prompting them to inquire about his diet. They discovered that, because of financial difficulties, his diet primarily consisted of processed foods with little to no fruits or vegetables, and he had stopped taking supplements recommended by his gastroenterologist. Further tests revealed a vitamin D deficiency and a severe deficiency in vitamin C. With the diagnosis of scurvy confirmed, the doctors treated the patient with 1000 mg of ascorbic acid daily, along with cholecalciferol, folic acid, and a multivitamin complex, leading to a complete resolution of his symptoms.
Risk Factors Then and Now
It can cause mucosal and gastric hemorrhages, and if left untreated, it can lead to fatal bleeding.
Historically known as “sailors’ disease,” scurvy plagued men on long voyages who lacked access to fresh fruits or vegetables and thus did not get enough vitamin C. In 1747, James Lind, a British physician in the Royal Navy, demonstrated that the consumption of oranges and lemons could combat scurvy.
Today’s risk factors for scurvy include malnutrition, gastrointestinal disorders (eg, chronic inflammatory bowel diseases), alcohol and tobacco use, eating disorders, psychiatric illnesses, dialysis, and the use of medications that reduce the absorption of ascorbic acid (such as corticosteroids and proton pump inhibitors).
Scurvy remains more common among individuals with unfavorable socioeconomic conditions. The authors of the study emphasize how the rising cost of living — specifically in Australia but applicable elsewhere — is changing eating habits, leading to a high consumption of low-cost, nutritionally poor foods.
Poverty has always been a risk factor for scurvy, but today there may be an additional cause: bariatric surgery. Patients undergoing these procedures are at a risk for deficiencies in fat-soluble vitamins A, D, E, and K, and if their diet is inadequate, they may also experience a vitamin C deficiency. Awareness of this can facilitate the timely diagnosis of scurvy in these patients.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
“Petechial rash often prompts further investigation into hematological, dermatological, or vasculitis causes. However, if the above investigations are negative and skin biopsy has not revealed a cause, there is a Renaissance-era diagnosis that is often overlooked but is easily investigated and treated,” wrote Andrew Dermawan, MD, and colleagues from Sir Charles Gairdner Hospital in Nedlands, Australia, in BMJ Case Reports. The diagnosis they highlight is scurvy, a disease that has faded from common medical concern but is reemerging, partly because of the rise in bariatric surgery.
Diagnosing Scurvy in the 2020s
In their article, Dermawan and colleagues present the case of a 50-year-old man with a bilateral petechial rash on his lower limbs, without any history of trauma. The patient, who exhibited no infectious symptoms, also had gross hematuria, microcytic anemia, mild neutropenia, and lymphopenia. Tests for autoimmune and hematological diseases were negative, as were abdominal and leg CT scans, ruling out abdominal hemorrhage and vasculitis. Additionally, a skin biopsy showed no causative findings.
The doctors noted that the patient had undergone sleeve gastrectomy, prompting them to inquire about his diet. They discovered that, because of financial difficulties, his diet primarily consisted of processed foods with little to no fruits or vegetables, and he had stopped taking supplements recommended by his gastroenterologist. Further tests revealed a vitamin D deficiency and a severe deficiency in vitamin C. With the diagnosis of scurvy confirmed, the doctors treated the patient with 1000 mg of ascorbic acid daily, along with cholecalciferol, folic acid, and a multivitamin complex, leading to a complete resolution of his symptoms.
Risk Factors Then and Now
It can cause mucosal and gastric hemorrhages, and if left untreated, it can lead to fatal bleeding.
Historically known as “sailors’ disease,” scurvy plagued men on long voyages who lacked access to fresh fruits or vegetables and thus did not get enough vitamin C. In 1747, James Lind, a British physician in the Royal Navy, demonstrated that the consumption of oranges and lemons could combat scurvy.
Today’s risk factors for scurvy include malnutrition, gastrointestinal disorders (eg, chronic inflammatory bowel diseases), alcohol and tobacco use, eating disorders, psychiatric illnesses, dialysis, and the use of medications that reduce the absorption of ascorbic acid (such as corticosteroids and proton pump inhibitors).
Scurvy remains more common among individuals with unfavorable socioeconomic conditions. The authors of the study emphasize how the rising cost of living — specifically in Australia but applicable elsewhere — is changing eating habits, leading to a high consumption of low-cost, nutritionally poor foods.
Poverty has always been a risk factor for scurvy, but today there may be an additional cause: bariatric surgery. Patients undergoing these procedures are at a risk for deficiencies in fat-soluble vitamins A, D, E, and K, and if their diet is inadequate, they may also experience a vitamin C deficiency. Awareness of this can facilitate the timely diagnosis of scurvy in these patients.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Experts Challenge New Diagnostic Criteria for Alzheimer’s disease
In a paper published online in JAMA Neurology, the International Working Group (IWG), which includes 46 experts from 17 countries, is recommending that the diagnosis of Alzheimer’s disease be limited to individuals with mild cognitive impairment or dementia and not be applied to cognitively normal individuals with Alzheimer’s disease biomarkers such as amyloid-beta 42/40 or p-tau.
Clinicians should be “very careful” about using the “A” word (Alzheimer’s) for cognitively unimpaired people with Alzheimer’s disease biomarkers, said the paper’s first author Bruno Dubois, MD, professor of neurology, Sorbonne University and Department of Neurology, Pitié-Salpêtrière Hospital, Paris, France.
Providing an Alzheimer’s disease diagnosis to those who have a high chance of never developing cognitive impairment can be psychologically harmful, said Dubois.
“It’s not something small like telling someone they have a fever. Just imagine you’re 65 years old and are amyloid positive, and you’re told you have Alzheimer’s disease. It affects the decisions you make for the rest of your life and changes your vision of your future, even though you may never develop the disease,” he added.
Divergent View
The IWG’s perspective on Alzheimer’s disease contrasts with a recent proposal from the Alzheimer’s Association. The Alzheimer’s Association criteria suggest that Alzheimer’s disease should be regarded solely as a biological entity, which could include cognitively normal individuals with one core Alzheimer’s disease biomarker.
The IWG noted that its concerns regarding the application of a purely biological definition of Alzheimer’s disease in clinical practice prompted the group to consider updating its guidelines, potentially offering “an alternative definitional view of Alzheimer’s disease as a clinical-biological construct for clinical use.”
The group conducted a PubMed search for relevant Alzheimer’s disease articles, and included references, published between July 2020 and March 2024. The research showed the majority of biomarker-positive, cognitively normal individuals will not become symptomatic during their lifetime.
The risk of a 55-year-old who is amyloid positive developing Alzheimer’s disease is not that much higher than that for an individual of a similar age who is amyloid negative, Dubois noted. “There’s an 83% chance that person will never develop Alzheimer’s disease.”
Disclosing a diagnosis of Alzheimer’s disease to cognitively normal people with only one core Alzheimer’s disease biomarker represents “the most problematic implication of a purely biological definition of the disease,” the authors noted.
“A biomarker is a marker of pathology, not a biomarker of disease,” said Dubois, adding that a person may have markers for several different brain diseases.
The IWG recommends the following nomenclature: At risk for Alzheimer’s disease for those with Alzheimer’s disease biomarkers but low lifetime risk and presymptomatic Alzheimer’s disease for those with Alzheimer’s disease biomarkers with a very high lifetime risk for progression such as individuals with autosomal dominant genetic mutations and other distinct biomarker profiles that put them at extremely high lifetime risk of developing the disease.
Dubois emphasized the difference between those showing typical Alzheimer’s disease symptoms with positive biomarkers who should be considered to have the disease and those with positive biomarkers but no typical Alzheimer’s disease symptoms who should be considered at risk.
This is an important distinction as it affects research approaches and assessment of risks, he said.
For low-risk asymptomatic individuals, the IWG does not recommend routine diagnostic testing outside of the research setting. “There’s no reason to send a 65-year-old cognitively normal subject off to collect biomarker information,” said Dubois.
He reiterated the importance of clinicians using appropriate and sensitive language surrounding Alzheimer’s disease when face to face with patients. This issue “is not purely semantic; this is real life.”
For these patients in the clinical setting, “we have to be very careful about proposing treatments that may have side effects,” he said.
However, this does not mean asymptomatic at-risk people should not be studied to determine what pharmacological interventions might prevent or delay the onset of clinical disease, he noted.
Presymptomatic individuals who are at a high risk of developing Alzheimer’s disease “should be the target for clinical trials in the future” to determine best ways to delay the conversion to Alzheimer’s disease, he said.
The main focus of such research should be to better understand the “biomarker pattern profile” that is associated with a high risk of developing Alzheimer’s disease, said Dubois.
Plea for Unity
In an accompanying editorial, Ronald C. Petersen, PhD, MD, director, Mayo Clinic Alzheimer’s Disease Research Center and Mayo Clinic Study of Aging, Rochester, Minnesota, and colleagues outline the difference between the IWG and Alzheimer’s Association positions.
As the IWG uses Alzheimer’s disease to define those with cognitive impairment and the Alzheimer’s Association group uses Alzheimer’s disease to define those with the pathology of the disease, the field is now at a crossroads. “Do we name the disease before clinical symptoms?” they asked.
They note that Alzheimer’s Association criteria distinguish between a disease and an illness, whereas the IWG does not. “As such, although the primary disagreement between the groups is semantic, the ramifications of the labeling can be significant.”
It is “incumbent” that the field “come together” on an Alzheimer’s disease definition, the editorial concluded. “Neither the Alzheimer’s Association or IWG documents are appropriate to serve as a guide for how to apply biomarkers in a clinical setting. Appropriate-use criteria are needed to form a bridge between biological frameworks and real-world clinical practice so we can all maximally help all of our patients with this disorder.”
In a comment, Reisa Sperling, MD, professor of neurology, Harvard Medical School, and director, Center for Alzheimer Research and Treatment, Brigham and Women’s Hospital and Massachusetts General Hospital, all in Boston, who is part of the Alzheimer’s Association work group that published the revised criteria for diagnosis and staging of Alzheimer’s disease, likened Alzheimer’s disease, which begins in the brain many years before dementia onset, to cardiovascular disease in that it involves multiple processes. She noted the World Health Organization classifies cardiovascular disease as a “disease” prior to clinical manifestations such as stroke and myocardial infarction.
“If someone has Alzheimer’s disease pathology in their brain, they are at risk for dementia or clinical manifestations of the disease — just like vascular disease quantifies the risk of stroke or heart attack, not risk of developing ‘vascular disease’ if the underlying vascular disease is already present,” said Sperling.
A large part of the controversy is related to terminology and the “stigma” of the “A” word in the same way there used to be fear around using the “C” word — cancer, said Sperling.
“Once people began talking about cancer publicly as a potentially treatable disease and began getting screened and diagnosed before symptoms of cancer were manifest, this has had a tremendous impact on public health.”
She clarified that her work group does not recommend screening asymptomatic people with Alzheimer’s disease biomarkers. “We actually need to prove that treating at the preclinical stage of the disease is able to prevent clinical impairment and dementia,” she said, adding “hopefully, we are getting closer to this.”
Dubois reported no relevant disclosures. Petersen reported receiving personal fees from Roche, Genentech, Eli Lilly and Company, Eisai, and Novo Nordisk outside the submitted work and royalties from Oxford University Press, UpToDate, and Medscape educational activities.
A version of this article appeared on Medscape.com.
In a paper published online in JAMA Neurology, the International Working Group (IWG), which includes 46 experts from 17 countries, is recommending that the diagnosis of Alzheimer’s disease be limited to individuals with mild cognitive impairment or dementia and not be applied to cognitively normal individuals with Alzheimer’s disease biomarkers such as amyloid-beta 42/40 or p-tau.
Clinicians should be “very careful” about using the “A” word (Alzheimer’s) for cognitively unimpaired people with Alzheimer’s disease biomarkers, said the paper’s first author Bruno Dubois, MD, professor of neurology, Sorbonne University and Department of Neurology, Pitié-Salpêtrière Hospital, Paris, France.
Providing an Alzheimer’s disease diagnosis to those who have a high chance of never developing cognitive impairment can be psychologically harmful, said Dubois.
“It’s not something small like telling someone they have a fever. Just imagine you’re 65 years old and are amyloid positive, and you’re told you have Alzheimer’s disease. It affects the decisions you make for the rest of your life and changes your vision of your future, even though you may never develop the disease,” he added.
Divergent View
The IWG’s perspective on Alzheimer’s disease contrasts with a recent proposal from the Alzheimer’s Association. The Alzheimer’s Association criteria suggest that Alzheimer’s disease should be regarded solely as a biological entity, which could include cognitively normal individuals with one core Alzheimer’s disease biomarker.
The IWG noted that its concerns regarding the application of a purely biological definition of Alzheimer’s disease in clinical practice prompted the group to consider updating its guidelines, potentially offering “an alternative definitional view of Alzheimer’s disease as a clinical-biological construct for clinical use.”
The group conducted a PubMed search for relevant Alzheimer’s disease articles, and included references, published between July 2020 and March 2024. The research showed the majority of biomarker-positive, cognitively normal individuals will not become symptomatic during their lifetime.
The risk of a 55-year-old who is amyloid positive developing Alzheimer’s disease is not that much higher than that for an individual of a similar age who is amyloid negative, Dubois noted. “There’s an 83% chance that person will never develop Alzheimer’s disease.”
Disclosing a diagnosis of Alzheimer’s disease to cognitively normal people with only one core Alzheimer’s disease biomarker represents “the most problematic implication of a purely biological definition of the disease,” the authors noted.
“A biomarker is a marker of pathology, not a biomarker of disease,” said Dubois, adding that a person may have markers for several different brain diseases.
The IWG recommends the following nomenclature: At risk for Alzheimer’s disease for those with Alzheimer’s disease biomarkers but low lifetime risk and presymptomatic Alzheimer’s disease for those with Alzheimer’s disease biomarkers with a very high lifetime risk for progression such as individuals with autosomal dominant genetic mutations and other distinct biomarker profiles that put them at extremely high lifetime risk of developing the disease.
Dubois emphasized the difference between those showing typical Alzheimer’s disease symptoms with positive biomarkers who should be considered to have the disease and those with positive biomarkers but no typical Alzheimer’s disease symptoms who should be considered at risk.
This is an important distinction as it affects research approaches and assessment of risks, he said.
For low-risk asymptomatic individuals, the IWG does not recommend routine diagnostic testing outside of the research setting. “There’s no reason to send a 65-year-old cognitively normal subject off to collect biomarker information,” said Dubois.
He reiterated the importance of clinicians using appropriate and sensitive language surrounding Alzheimer’s disease when face to face with patients. This issue “is not purely semantic; this is real life.”
For these patients in the clinical setting, “we have to be very careful about proposing treatments that may have side effects,” he said.
However, this does not mean asymptomatic at-risk people should not be studied to determine what pharmacological interventions might prevent or delay the onset of clinical disease, he noted.
Presymptomatic individuals who are at a high risk of developing Alzheimer’s disease “should be the target for clinical trials in the future” to determine best ways to delay the conversion to Alzheimer’s disease, he said.
The main focus of such research should be to better understand the “biomarker pattern profile” that is associated with a high risk of developing Alzheimer’s disease, said Dubois.
Plea for Unity
In an accompanying editorial, Ronald C. Petersen, PhD, MD, director, Mayo Clinic Alzheimer’s Disease Research Center and Mayo Clinic Study of Aging, Rochester, Minnesota, and colleagues outline the difference between the IWG and Alzheimer’s Association positions.
As the IWG uses Alzheimer’s disease to define those with cognitive impairment and the Alzheimer’s Association group uses Alzheimer’s disease to define those with the pathology of the disease, the field is now at a crossroads. “Do we name the disease before clinical symptoms?” they asked.
They note that Alzheimer’s Association criteria distinguish between a disease and an illness, whereas the IWG does not. “As such, although the primary disagreement between the groups is semantic, the ramifications of the labeling can be significant.”
It is “incumbent” that the field “come together” on an Alzheimer’s disease definition, the editorial concluded. “Neither the Alzheimer’s Association or IWG documents are appropriate to serve as a guide for how to apply biomarkers in a clinical setting. Appropriate-use criteria are needed to form a bridge between biological frameworks and real-world clinical practice so we can all maximally help all of our patients with this disorder.”
In a comment, Reisa Sperling, MD, professor of neurology, Harvard Medical School, and director, Center for Alzheimer Research and Treatment, Brigham and Women’s Hospital and Massachusetts General Hospital, all in Boston, who is part of the Alzheimer’s Association work group that published the revised criteria for diagnosis and staging of Alzheimer’s disease, likened Alzheimer’s disease, which begins in the brain many years before dementia onset, to cardiovascular disease in that it involves multiple processes. She noted the World Health Organization classifies cardiovascular disease as a “disease” prior to clinical manifestations such as stroke and myocardial infarction.
“If someone has Alzheimer’s disease pathology in their brain, they are at risk for dementia or clinical manifestations of the disease — just like vascular disease quantifies the risk of stroke or heart attack, not risk of developing ‘vascular disease’ if the underlying vascular disease is already present,” said Sperling.
A large part of the controversy is related to terminology and the “stigma” of the “A” word in the same way there used to be fear around using the “C” word — cancer, said Sperling.
“Once people began talking about cancer publicly as a potentially treatable disease and began getting screened and diagnosed before symptoms of cancer were manifest, this has had a tremendous impact on public health.”
She clarified that her work group does not recommend screening asymptomatic people with Alzheimer’s disease biomarkers. “We actually need to prove that treating at the preclinical stage of the disease is able to prevent clinical impairment and dementia,” she said, adding “hopefully, we are getting closer to this.”
Dubois reported no relevant disclosures. Petersen reported receiving personal fees from Roche, Genentech, Eli Lilly and Company, Eisai, and Novo Nordisk outside the submitted work and royalties from Oxford University Press, UpToDate, and Medscape educational activities.
A version of this article appeared on Medscape.com.
In a paper published online in JAMA Neurology, the International Working Group (IWG), which includes 46 experts from 17 countries, is recommending that the diagnosis of Alzheimer’s disease be limited to individuals with mild cognitive impairment or dementia and not be applied to cognitively normal individuals with Alzheimer’s disease biomarkers such as amyloid-beta 42/40 or p-tau.
Clinicians should be “very careful” about using the “A” word (Alzheimer’s) for cognitively unimpaired people with Alzheimer’s disease biomarkers, said the paper’s first author Bruno Dubois, MD, professor of neurology, Sorbonne University and Department of Neurology, Pitié-Salpêtrière Hospital, Paris, France.
Providing an Alzheimer’s disease diagnosis to those who have a high chance of never developing cognitive impairment can be psychologically harmful, said Dubois.
“It’s not something small like telling someone they have a fever. Just imagine you’re 65 years old and are amyloid positive, and you’re told you have Alzheimer’s disease. It affects the decisions you make for the rest of your life and changes your vision of your future, even though you may never develop the disease,” he added.
Divergent View
The IWG’s perspective on Alzheimer’s disease contrasts with a recent proposal from the Alzheimer’s Association. The Alzheimer’s Association criteria suggest that Alzheimer’s disease should be regarded solely as a biological entity, which could include cognitively normal individuals with one core Alzheimer’s disease biomarker.
The IWG noted that its concerns regarding the application of a purely biological definition of Alzheimer’s disease in clinical practice prompted the group to consider updating its guidelines, potentially offering “an alternative definitional view of Alzheimer’s disease as a clinical-biological construct for clinical use.”
The group conducted a PubMed search for relevant Alzheimer’s disease articles, and included references, published between July 2020 and March 2024. The research showed the majority of biomarker-positive, cognitively normal individuals will not become symptomatic during their lifetime.
The risk of a 55-year-old who is amyloid positive developing Alzheimer’s disease is not that much higher than that for an individual of a similar age who is amyloid negative, Dubois noted. “There’s an 83% chance that person will never develop Alzheimer’s disease.”
Disclosing a diagnosis of Alzheimer’s disease to cognitively normal people with only one core Alzheimer’s disease biomarker represents “the most problematic implication of a purely biological definition of the disease,” the authors noted.
“A biomarker is a marker of pathology, not a biomarker of disease,” said Dubois, adding that a person may have markers for several different brain diseases.
The IWG recommends the following nomenclature: At risk for Alzheimer’s disease for those with Alzheimer’s disease biomarkers but low lifetime risk and presymptomatic Alzheimer’s disease for those with Alzheimer’s disease biomarkers with a very high lifetime risk for progression such as individuals with autosomal dominant genetic mutations and other distinct biomarker profiles that put them at extremely high lifetime risk of developing the disease.
Dubois emphasized the difference between those showing typical Alzheimer’s disease symptoms with positive biomarkers who should be considered to have the disease and those with positive biomarkers but no typical Alzheimer’s disease symptoms who should be considered at risk.
This is an important distinction as it affects research approaches and assessment of risks, he said.
For low-risk asymptomatic individuals, the IWG does not recommend routine diagnostic testing outside of the research setting. “There’s no reason to send a 65-year-old cognitively normal subject off to collect biomarker information,” said Dubois.
He reiterated the importance of clinicians using appropriate and sensitive language surrounding Alzheimer’s disease when face to face with patients. This issue “is not purely semantic; this is real life.”
For these patients in the clinical setting, “we have to be very careful about proposing treatments that may have side effects,” he said.
However, this does not mean asymptomatic at-risk people should not be studied to determine what pharmacological interventions might prevent or delay the onset of clinical disease, he noted.
Presymptomatic individuals who are at a high risk of developing Alzheimer’s disease “should be the target for clinical trials in the future” to determine best ways to delay the conversion to Alzheimer’s disease, he said.
The main focus of such research should be to better understand the “biomarker pattern profile” that is associated with a high risk of developing Alzheimer’s disease, said Dubois.
Plea for Unity
In an accompanying editorial, Ronald C. Petersen, PhD, MD, director, Mayo Clinic Alzheimer’s Disease Research Center and Mayo Clinic Study of Aging, Rochester, Minnesota, and colleagues outline the difference between the IWG and Alzheimer’s Association positions.
As the IWG uses Alzheimer’s disease to define those with cognitive impairment and the Alzheimer’s Association group uses Alzheimer’s disease to define those with the pathology of the disease, the field is now at a crossroads. “Do we name the disease before clinical symptoms?” they asked.
They note that Alzheimer’s Association criteria distinguish between a disease and an illness, whereas the IWG does not. “As such, although the primary disagreement between the groups is semantic, the ramifications of the labeling can be significant.”
It is “incumbent” that the field “come together” on an Alzheimer’s disease definition, the editorial concluded. “Neither the Alzheimer’s Association or IWG documents are appropriate to serve as a guide for how to apply biomarkers in a clinical setting. Appropriate-use criteria are needed to form a bridge between biological frameworks and real-world clinical practice so we can all maximally help all of our patients with this disorder.”
In a comment, Reisa Sperling, MD, professor of neurology, Harvard Medical School, and director, Center for Alzheimer Research and Treatment, Brigham and Women’s Hospital and Massachusetts General Hospital, all in Boston, who is part of the Alzheimer’s Association work group that published the revised criteria for diagnosis and staging of Alzheimer’s disease, likened Alzheimer’s disease, which begins in the brain many years before dementia onset, to cardiovascular disease in that it involves multiple processes. She noted the World Health Organization classifies cardiovascular disease as a “disease” prior to clinical manifestations such as stroke and myocardial infarction.
“If someone has Alzheimer’s disease pathology in their brain, they are at risk for dementia or clinical manifestations of the disease — just like vascular disease quantifies the risk of stroke or heart attack, not risk of developing ‘vascular disease’ if the underlying vascular disease is already present,” said Sperling.
A large part of the controversy is related to terminology and the “stigma” of the “A” word in the same way there used to be fear around using the “C” word — cancer, said Sperling.
“Once people began talking about cancer publicly as a potentially treatable disease and began getting screened and diagnosed before symptoms of cancer were manifest, this has had a tremendous impact on public health.”
She clarified that her work group does not recommend screening asymptomatic people with Alzheimer’s disease biomarkers. “We actually need to prove that treating at the preclinical stage of the disease is able to prevent clinical impairment and dementia,” she said, adding “hopefully, we are getting closer to this.”
Dubois reported no relevant disclosures. Petersen reported receiving personal fees from Roche, Genentech, Eli Lilly and Company, Eisai, and Novo Nordisk outside the submitted work and royalties from Oxford University Press, UpToDate, and Medscape educational activities.
A version of this article appeared on Medscape.com.
From JAMA Neurology

