User login
Higher dietary vitamin A linked to lower SCC risk
in a large prospective study published in JAMA Dermatology.
There was also an inverse association between intake of carotenoids and risk of cutaneous SCC over the follow-up period of 26-28 years. The results of the study support “the protective role of vitamin A against SCC development,” wrote Jongwoo Kim, MD, of Brown University, Providence, R.I., and Inje University, Seoul, South Korea, and coauthors. “Our data further support the contention that supplemental and dietary vitamin A may be beneficial in preventing SCC,” they added.
The study evaluated intake of vitamin A and carotenoids and SCC risk with data from the Health Professionals Follow-Up Study (1986-2012) of 48,400 men, and the Nurses’ Health Study (1984-2012) of 75,170 women. Participants in those studies completed questionnaires based on lifestyle and medical history. Only white participants were included, because of the low number of SCC cases and low SCC risk in nonwhite participants, and participants who did not report diet and those who had a history of melanoma, SCC, or other cancer diagnoses at baseline were excluded.
Over the follow-up of 26-28 years, a total of 3,978 SCC cases were confirmed using pathological records. The investigators used different quintiles based on the median total amount of vitamin A intake. Using quintile 1 (lowest intake) as a reference, the pooled multivariate hazard ratios of vitamin A intake were 0.97, 0.97, 0.93, and 0.83 for quintiles 2, 3, 4, and 5, respectively (P less than .001 for the trend, in order of increasing quintiles).
In addition, they reported that greater intakes of retinol and several carotenoids were also significantly associated with a lower SCC risk.
The results were “generally consistent between men and women,” and “the inverse associations appeared to be more prominent among those with moles and those with burn or blistering sunburn reaction as children or adolescents,” they wrote.
The large sample size, prospective design, and confirmation of SCC cases by histology are among the strengths of the study, while a key limitation of the study was the homogeneous nature of the study population, which “may limit the generalizability of our findings,” the authors wrote.
The study was funded by the National Institutes of Health and Inje University (South Korea). One author reported serving as a consultant for AbbVie, Amgen, the Centers for Disease Control and Prevention, Janssen, Merck, Novartis, and Pfizer; and as a compensated investigator for Amgen, Regeneron, and Sanofi. Dr. Kim and the remaining three authors reported no disclosures.
SOURCE: Kim J et al. JAMA Dermatol. 2019 Jul 31. doi: 10.1001/jamadermatol.2019.1937.
in a large prospective study published in JAMA Dermatology.
There was also an inverse association between intake of carotenoids and risk of cutaneous SCC over the follow-up period of 26-28 years. The results of the study support “the protective role of vitamin A against SCC development,” wrote Jongwoo Kim, MD, of Brown University, Providence, R.I., and Inje University, Seoul, South Korea, and coauthors. “Our data further support the contention that supplemental and dietary vitamin A may be beneficial in preventing SCC,” they added.
The study evaluated intake of vitamin A and carotenoids and SCC risk with data from the Health Professionals Follow-Up Study (1986-2012) of 48,400 men, and the Nurses’ Health Study (1984-2012) of 75,170 women. Participants in those studies completed questionnaires based on lifestyle and medical history. Only white participants were included, because of the low number of SCC cases and low SCC risk in nonwhite participants, and participants who did not report diet and those who had a history of melanoma, SCC, or other cancer diagnoses at baseline were excluded.
Over the follow-up of 26-28 years, a total of 3,978 SCC cases were confirmed using pathological records. The investigators used different quintiles based on the median total amount of vitamin A intake. Using quintile 1 (lowest intake) as a reference, the pooled multivariate hazard ratios of vitamin A intake were 0.97, 0.97, 0.93, and 0.83 for quintiles 2, 3, 4, and 5, respectively (P less than .001 for the trend, in order of increasing quintiles).
In addition, they reported that greater intakes of retinol and several carotenoids were also significantly associated with a lower SCC risk.
The results were “generally consistent between men and women,” and “the inverse associations appeared to be more prominent among those with moles and those with burn or blistering sunburn reaction as children or adolescents,” they wrote.
The large sample size, prospective design, and confirmation of SCC cases by histology are among the strengths of the study, while a key limitation of the study was the homogeneous nature of the study population, which “may limit the generalizability of our findings,” the authors wrote.
The study was funded by the National Institutes of Health and Inje University (South Korea). One author reported serving as a consultant for AbbVie, Amgen, the Centers for Disease Control and Prevention, Janssen, Merck, Novartis, and Pfizer; and as a compensated investigator for Amgen, Regeneron, and Sanofi. Dr. Kim and the remaining three authors reported no disclosures.
SOURCE: Kim J et al. JAMA Dermatol. 2019 Jul 31. doi: 10.1001/jamadermatol.2019.1937.
in a large prospective study published in JAMA Dermatology.
There was also an inverse association between intake of carotenoids and risk of cutaneous SCC over the follow-up period of 26-28 years. The results of the study support “the protective role of vitamin A against SCC development,” wrote Jongwoo Kim, MD, of Brown University, Providence, R.I., and Inje University, Seoul, South Korea, and coauthors. “Our data further support the contention that supplemental and dietary vitamin A may be beneficial in preventing SCC,” they added.
The study evaluated intake of vitamin A and carotenoids and SCC risk with data from the Health Professionals Follow-Up Study (1986-2012) of 48,400 men, and the Nurses’ Health Study (1984-2012) of 75,170 women. Participants in those studies completed questionnaires based on lifestyle and medical history. Only white participants were included, because of the low number of SCC cases and low SCC risk in nonwhite participants, and participants who did not report diet and those who had a history of melanoma, SCC, or other cancer diagnoses at baseline were excluded.
Over the follow-up of 26-28 years, a total of 3,978 SCC cases were confirmed using pathological records. The investigators used different quintiles based on the median total amount of vitamin A intake. Using quintile 1 (lowest intake) as a reference, the pooled multivariate hazard ratios of vitamin A intake were 0.97, 0.97, 0.93, and 0.83 for quintiles 2, 3, 4, and 5, respectively (P less than .001 for the trend, in order of increasing quintiles).
In addition, they reported that greater intakes of retinol and several carotenoids were also significantly associated with a lower SCC risk.
The results were “generally consistent between men and women,” and “the inverse associations appeared to be more prominent among those with moles and those with burn or blistering sunburn reaction as children or adolescents,” they wrote.
The large sample size, prospective design, and confirmation of SCC cases by histology are among the strengths of the study, while a key limitation of the study was the homogeneous nature of the study population, which “may limit the generalizability of our findings,” the authors wrote.
The study was funded by the National Institutes of Health and Inje University (South Korea). One author reported serving as a consultant for AbbVie, Amgen, the Centers for Disease Control and Prevention, Janssen, Merck, Novartis, and Pfizer; and as a compensated investigator for Amgen, Regeneron, and Sanofi. Dr. Kim and the remaining three authors reported no disclosures.
SOURCE: Kim J et al. JAMA Dermatol. 2019 Jul 31. doi: 10.1001/jamadermatol.2019.1937.
FROM JAMA DERMATOLOGY
Metastatic Adamantinoma Presenting as a Cutaneous Papule
To the Editor:
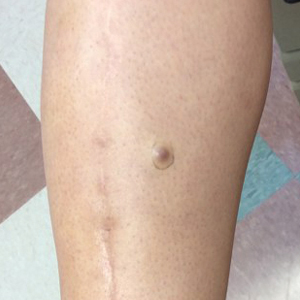
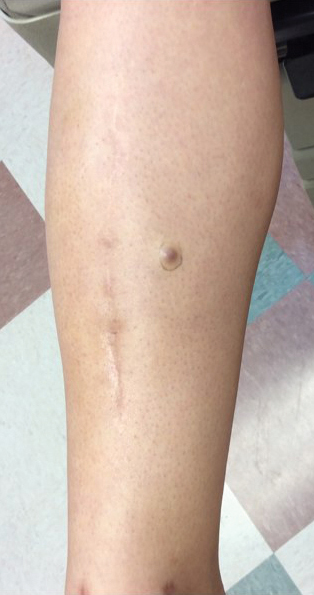
A 34-year-old woman with a history of adamantinoma of the right tibia that had been surgically resected with tibial reconstruction 5 years prior presented with a mildly tender, enlarging lesion on the right distal shin of 6 months’ duration that had started to change color. Review of systems was otherwise negative. Physical examination revealed an 8-mm, slightly tender, rubbery, pink papule adjacent to the surgical scar over the right tibia (Figure 1). Given the rapid growth of the lesion and its proximity to the surgical site, a punch biopsy was performed.
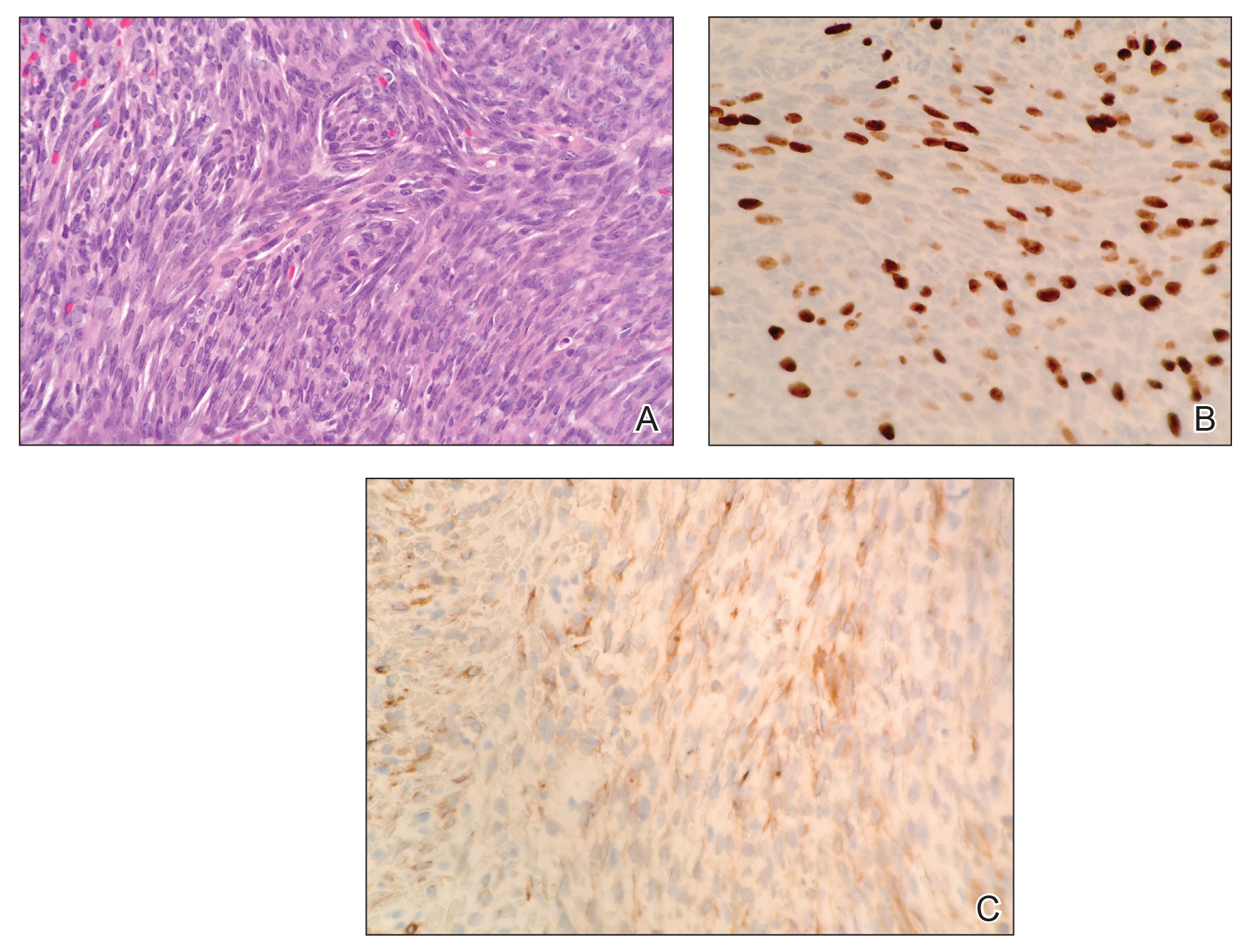
Histopathologic examination demonstrated a densely cellular dermal tumor composed of spindle cells with large hyperchromatic nuclei, numerous mitotic figures, and minimal eosinophilic cytoplasm (Figure 2A). Immunohistochemical studies revealed that approximately 40% of the tumor nuclei were immunoreactive to Ki-67 (Figure 2B), and total cytokeratin was focally positive (Figure 2C). A diagnosis of metastatic adamantinoma was made. Positron emission tomography and magnetic resonance imaging revealed new lytic lesions involving the T10 and L2 vertebrae (without frank spinal cord compression) and the right superior sacrum. Additionally, a small pulmonary nodule on the left upper lobe was noted on positron emission tomography, but it was below the size threshold for reliable detection. A computed tomography–guided biopsy of the T10 lesion demonstrated metastatic adamantinoma. The patient underwent a spinal stabilization procedure and discussed options regarding further oncologic and palliative management.
Adamantinoma is an extremely rare primary malignant bone tumor that typically involves the anterior portion of the tibial metaphysis or diaphysis in approximately 90% of cases. Young adults most commonly are affected in the third or fourth decades of life.1 Although the histogenesis is not clearly understood, experts have theorized that fetal implantation during embryogenesis or traumatic implantation of epithelial cells may be causes of this tumor and may explain the close pathologic similarity to basal cell carcinoma.2
Adamantinomas are slow growing, and as a result, patients often present with gradual onset of pain and swelling that persists for years.3,4 Metastasis occurs in 10% to 30% of patients, typically located in regional lymph nodes, the lungs, and distant bone.1,4 Our case represents a rare instance of adamantinoma metastasis to the skin. Although primary adamantinomas consist of both epithelial and stromal components, the typical metastatic lesions of adamantinomas are solely epithelial (often in a spindle-cell pattern),1 as was seen in our patient.
Operative removal via amputation or en bloc resection with limb salvage is the current treatment of choice. Adamantinomas are highly radioresistant, and chemotherapy has shown minimal efficacy.3,5
In conclusion, the presence of cutaneous metastasis from an adamantinoma is rare. Our case emphasizes this tumor’s potential for late metastasis as well as late recurrence.3,6 Most importantly, dermatologists should be made aware of this rare bone tumor and its unusual presentation, as early detection can aid in prognosis.
- Schowinsky JT, Ormond DR, Kleinschmidt-DeMasters BK. Tibial adamantinoma: late metastasis to the brain. J Neuropathol Exp Neurol. 2015;74:95-97.
- Jain D, Jain VK, Vasishta RK, et al. Adamantinoma: a clinicopathological review and update. Diagn Pathol. 2008;3:8.
- Qureshi AA, Shott S, Mallin BA, et al. Current trends in the management of adamantinoma of long bones. an international study. J Bone Joint Surg Am. 2000;82-A:1122-1131.
- Desai SS, Jambhekar N, Agarwal M, et al. Adamantinoma of tibia: a study of 12 cases. J Surg Oncol. 2006;93:429-433.
- Weiss SW, Dorfman HD. Adamantinoma of long bone. an analysis of nine new cases with emphasis on metastasizing lesions and fibrous dysplasia-like changes. Hum Pathol. 1977;8:141-153.
- Szendroi M, Antal I, Arató G. Adamantinoma of long bones: a long-term follow-up study of 11 cases. Pathol Oncol Res. 2009;15:209-216.
To the Editor:
A 34-year-old woman with a history of adamantinoma of the right tibia that had been surgically resected with tibial reconstruction 5 years prior presented with a mildly tender, enlarging lesion on the right distal shin of 6 months’ duration that had started to change color. Review of systems was otherwise negative. Physical examination revealed an 8-mm, slightly tender, rubbery, pink papule adjacent to the surgical scar over the right tibia (Figure 1). Given the rapid growth of the lesion and its proximity to the surgical site, a punch biopsy was performed.

Histopathologic examination demonstrated a densely cellular dermal tumor composed of spindle cells with large hyperchromatic nuclei, numerous mitotic figures, and minimal eosinophilic cytoplasm (Figure 2A). Immunohistochemical studies revealed that approximately 40% of the tumor nuclei were immunoreactive to Ki-67 (Figure 2B), and total cytokeratin was focally positive (Figure 2C). A diagnosis of metastatic adamantinoma was made. Positron emission tomography and magnetic resonance imaging revealed new lytic lesions involving the T10 and L2 vertebrae (without frank spinal cord compression) and the right superior sacrum. Additionally, a small pulmonary nodule on the left upper lobe was noted on positron emission tomography, but it was below the size threshold for reliable detection. A computed tomography–guided biopsy of the T10 lesion demonstrated metastatic adamantinoma. The patient underwent a spinal stabilization procedure and discussed options regarding further oncologic and palliative management.

Adamantinoma is an extremely rare primary malignant bone tumor that typically involves the anterior portion of the tibial metaphysis or diaphysis in approximately 90% of cases. Young adults most commonly are affected in the third or fourth decades of life.1 Although the histogenesis is not clearly understood, experts have theorized that fetal implantation during embryogenesis or traumatic implantation of epithelial cells may be causes of this tumor and may explain the close pathologic similarity to basal cell carcinoma.2
Adamantinomas are slow growing, and as a result, patients often present with gradual onset of pain and swelling that persists for years.3,4 Metastasis occurs in 10% to 30% of patients, typically located in regional lymph nodes, the lungs, and distant bone.1,4 Our case represents a rare instance of adamantinoma metastasis to the skin. Although primary adamantinomas consist of both epithelial and stromal components, the typical metastatic lesions of adamantinomas are solely epithelial (often in a spindle-cell pattern),1 as was seen in our patient.
Operative removal via amputation or en bloc resection with limb salvage is the current treatment of choice. Adamantinomas are highly radioresistant, and chemotherapy has shown minimal efficacy.3,5
In conclusion, the presence of cutaneous metastasis from an adamantinoma is rare. Our case emphasizes this tumor’s potential for late metastasis as well as late recurrence.3,6 Most importantly, dermatologists should be made aware of this rare bone tumor and its unusual presentation, as early detection can aid in prognosis.
To the Editor:
A 34-year-old woman with a history of adamantinoma of the right tibia that had been surgically resected with tibial reconstruction 5 years prior presented with a mildly tender, enlarging lesion on the right distal shin of 6 months’ duration that had started to change color. Review of systems was otherwise negative. Physical examination revealed an 8-mm, slightly tender, rubbery, pink papule adjacent to the surgical scar over the right tibia (Figure 1). Given the rapid growth of the lesion and its proximity to the surgical site, a punch biopsy was performed.

Histopathologic examination demonstrated a densely cellular dermal tumor composed of spindle cells with large hyperchromatic nuclei, numerous mitotic figures, and minimal eosinophilic cytoplasm (Figure 2A). Immunohistochemical studies revealed that approximately 40% of the tumor nuclei were immunoreactive to Ki-67 (Figure 2B), and total cytokeratin was focally positive (Figure 2C). A diagnosis of metastatic adamantinoma was made. Positron emission tomography and magnetic resonance imaging revealed new lytic lesions involving the T10 and L2 vertebrae (without frank spinal cord compression) and the right superior sacrum. Additionally, a small pulmonary nodule on the left upper lobe was noted on positron emission tomography, but it was below the size threshold for reliable detection. A computed tomography–guided biopsy of the T10 lesion demonstrated metastatic adamantinoma. The patient underwent a spinal stabilization procedure and discussed options regarding further oncologic and palliative management.

Adamantinoma is an extremely rare primary malignant bone tumor that typically involves the anterior portion of the tibial metaphysis or diaphysis in approximately 90% of cases. Young adults most commonly are affected in the third or fourth decades of life.1 Although the histogenesis is not clearly understood, experts have theorized that fetal implantation during embryogenesis or traumatic implantation of epithelial cells may be causes of this tumor and may explain the close pathologic similarity to basal cell carcinoma.2
Adamantinomas are slow growing, and as a result, patients often present with gradual onset of pain and swelling that persists for years.3,4 Metastasis occurs in 10% to 30% of patients, typically located in regional lymph nodes, the lungs, and distant bone.1,4 Our case represents a rare instance of adamantinoma metastasis to the skin. Although primary adamantinomas consist of both epithelial and stromal components, the typical metastatic lesions of adamantinomas are solely epithelial (often in a spindle-cell pattern),1 as was seen in our patient.
Operative removal via amputation or en bloc resection with limb salvage is the current treatment of choice. Adamantinomas are highly radioresistant, and chemotherapy has shown minimal efficacy.3,5
In conclusion, the presence of cutaneous metastasis from an adamantinoma is rare. Our case emphasizes this tumor’s potential for late metastasis as well as late recurrence.3,6 Most importantly, dermatologists should be made aware of this rare bone tumor and its unusual presentation, as early detection can aid in prognosis.
- Schowinsky JT, Ormond DR, Kleinschmidt-DeMasters BK. Tibial adamantinoma: late metastasis to the brain. J Neuropathol Exp Neurol. 2015;74:95-97.
- Jain D, Jain VK, Vasishta RK, et al. Adamantinoma: a clinicopathological review and update. Diagn Pathol. 2008;3:8.
- Qureshi AA, Shott S, Mallin BA, et al. Current trends in the management of adamantinoma of long bones. an international study. J Bone Joint Surg Am. 2000;82-A:1122-1131.
- Desai SS, Jambhekar N, Agarwal M, et al. Adamantinoma of tibia: a study of 12 cases. J Surg Oncol. 2006;93:429-433.
- Weiss SW, Dorfman HD. Adamantinoma of long bone. an analysis of nine new cases with emphasis on metastasizing lesions and fibrous dysplasia-like changes. Hum Pathol. 1977;8:141-153.
- Szendroi M, Antal I, Arató G. Adamantinoma of long bones: a long-term follow-up study of 11 cases. Pathol Oncol Res. 2009;15:209-216.
- Schowinsky JT, Ormond DR, Kleinschmidt-DeMasters BK. Tibial adamantinoma: late metastasis to the brain. J Neuropathol Exp Neurol. 2015;74:95-97.
- Jain D, Jain VK, Vasishta RK, et al. Adamantinoma: a clinicopathological review and update. Diagn Pathol. 2008;3:8.
- Qureshi AA, Shott S, Mallin BA, et al. Current trends in the management of adamantinoma of long bones. an international study. J Bone Joint Surg Am. 2000;82-A:1122-1131.
- Desai SS, Jambhekar N, Agarwal M, et al. Adamantinoma of tibia: a study of 12 cases. J Surg Oncol. 2006;93:429-433.
- Weiss SW, Dorfman HD. Adamantinoma of long bone. an analysis of nine new cases with emphasis on metastasizing lesions and fibrous dysplasia-like changes. Hum Pathol. 1977;8:141-153.
- Szendroi M, Antal I, Arató G. Adamantinoma of long bones: a long-term follow-up study of 11 cases. Pathol Oncol Res. 2009;15:209-216.
Practice Points
- Metastatic adamantinoma of the skin is a rare clinical scenario.
- Dermatologists should be made aware of this rare bone tumor and its unusual presentation, as early detection can aid in prognosis.
Skin safety gap divides white, older from nonwhite, younger
of passersby and skin-cancer screening attendees in Washington, D.C.
“These findings highlight the importance of tailoring free skin cancer screening events for nonwhite and younger populations,” senior author Adam Friedman, MD, professor and interim chair of dermatology at George Washington University, Washington, said in a statement provided by the institution. “While free screening events are important, we also have to think about comprehensive, community-based solutions that reach broader demographic populations than skin cancer screenings alone.”
For the new study, which appears in the July 2019 issue of Journal of Drugs in Dermatology, researchers led by Emily C. Murphy, BS, of George Washington University, sought to better understand skin safety precautions in the population beyond those who attend skin cancer screenings (J Drugs Dermatol. 2019;18[7]:649-53).
The study authors surveyed 285 passersby at six locations in Washington, D.C. (65% were female, 47% were under age 31, 48% were white, and 29% were black) and 144 attendees at a free skin cancer screening at George Washington University (70% were female, 16% were under 31, 44% were over 60, 73% were white, and 14% were black).
The attendees at the screening event were much more likely to engage in sun safety habits that are linked to lower risk of squamous cell carcinoma, basal cell carcinoma, and melanoma: 34% always used sunscreen vs. 19% of the public group (P = .001), and 52% always sought shade vs. 32% of the public group (P = .002). Seventeen percent of the public group never used sunscreen compared with 8% of the screening group.
Whites and older subjects were more likely to embrace sun-safety practices. When the groups were combined, 84% of whites and 52% of blacks always or sometimes used sunscreen (P less than .0001). Those over 60 were much more likely to always seek shade than were those under 31 (53% vs. 24%, P less than .0001).
“The screening group was older and included more individuals with fair skin, highlighting the need to target younger and nonwhite populations for sun safety education,” the researchers wrote. “Encouraging sun safety in younger populations will decrease the risk of skin cancer for patients now and later in their lives. That said, educating populations who seek skin cancer screenings is still important given 22% of our screening cohort reported rarely or never wearing sunscreen, underscoring this program’s value.”
The researchers noted that the study’s limitations include its small size, the possibility that the public group had higher education rates because they were near a university, and the lack of insight into whether the public group represented the general population.
No study funding was reported. The study authors reported no relevant disclosures.
of passersby and skin-cancer screening attendees in Washington, D.C.
“These findings highlight the importance of tailoring free skin cancer screening events for nonwhite and younger populations,” senior author Adam Friedman, MD, professor and interim chair of dermatology at George Washington University, Washington, said in a statement provided by the institution. “While free screening events are important, we also have to think about comprehensive, community-based solutions that reach broader demographic populations than skin cancer screenings alone.”
For the new study, which appears in the July 2019 issue of Journal of Drugs in Dermatology, researchers led by Emily C. Murphy, BS, of George Washington University, sought to better understand skin safety precautions in the population beyond those who attend skin cancer screenings (J Drugs Dermatol. 2019;18[7]:649-53).
The study authors surveyed 285 passersby at six locations in Washington, D.C. (65% were female, 47% were under age 31, 48% were white, and 29% were black) and 144 attendees at a free skin cancer screening at George Washington University (70% were female, 16% were under 31, 44% were over 60, 73% were white, and 14% were black).
The attendees at the screening event were much more likely to engage in sun safety habits that are linked to lower risk of squamous cell carcinoma, basal cell carcinoma, and melanoma: 34% always used sunscreen vs. 19% of the public group (P = .001), and 52% always sought shade vs. 32% of the public group (P = .002). Seventeen percent of the public group never used sunscreen compared with 8% of the screening group.
Whites and older subjects were more likely to embrace sun-safety practices. When the groups were combined, 84% of whites and 52% of blacks always or sometimes used sunscreen (P less than .0001). Those over 60 were much more likely to always seek shade than were those under 31 (53% vs. 24%, P less than .0001).
“The screening group was older and included more individuals with fair skin, highlighting the need to target younger and nonwhite populations for sun safety education,” the researchers wrote. “Encouraging sun safety in younger populations will decrease the risk of skin cancer for patients now and later in their lives. That said, educating populations who seek skin cancer screenings is still important given 22% of our screening cohort reported rarely or never wearing sunscreen, underscoring this program’s value.”
The researchers noted that the study’s limitations include its small size, the possibility that the public group had higher education rates because they were near a university, and the lack of insight into whether the public group represented the general population.
No study funding was reported. The study authors reported no relevant disclosures.
of passersby and skin-cancer screening attendees in Washington, D.C.
“These findings highlight the importance of tailoring free skin cancer screening events for nonwhite and younger populations,” senior author Adam Friedman, MD, professor and interim chair of dermatology at George Washington University, Washington, said in a statement provided by the institution. “While free screening events are important, we also have to think about comprehensive, community-based solutions that reach broader demographic populations than skin cancer screenings alone.”
For the new study, which appears in the July 2019 issue of Journal of Drugs in Dermatology, researchers led by Emily C. Murphy, BS, of George Washington University, sought to better understand skin safety precautions in the population beyond those who attend skin cancer screenings (J Drugs Dermatol. 2019;18[7]:649-53).
The study authors surveyed 285 passersby at six locations in Washington, D.C. (65% were female, 47% were under age 31, 48% were white, and 29% were black) and 144 attendees at a free skin cancer screening at George Washington University (70% were female, 16% were under 31, 44% were over 60, 73% were white, and 14% were black).
The attendees at the screening event were much more likely to engage in sun safety habits that are linked to lower risk of squamous cell carcinoma, basal cell carcinoma, and melanoma: 34% always used sunscreen vs. 19% of the public group (P = .001), and 52% always sought shade vs. 32% of the public group (P = .002). Seventeen percent of the public group never used sunscreen compared with 8% of the screening group.
Whites and older subjects were more likely to embrace sun-safety practices. When the groups were combined, 84% of whites and 52% of blacks always or sometimes used sunscreen (P less than .0001). Those over 60 were much more likely to always seek shade than were those under 31 (53% vs. 24%, P less than .0001).
“The screening group was older and included more individuals with fair skin, highlighting the need to target younger and nonwhite populations for sun safety education,” the researchers wrote. “Encouraging sun safety in younger populations will decrease the risk of skin cancer for patients now and later in their lives. That said, educating populations who seek skin cancer screenings is still important given 22% of our screening cohort reported rarely or never wearing sunscreen, underscoring this program’s value.”
The researchers noted that the study’s limitations include its small size, the possibility that the public group had higher education rates because they were near a university, and the lack of insight into whether the public group represented the general population.
No study funding was reported. The study authors reported no relevant disclosures.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
How to recognize pediatric leukemia cutis
Researchers have characterized the clinical presentation, progression, and prognosis of leukemia cutis in a pediatric population, according to findings from a retrospective case series.
“To our knowledge, this is the largest reported case series of pediatric leukemia cutis,” wrote Elena Corina Andriescu of the University of Texas, Houston, and colleagues. The results were published in Pediatric Dermatology.
The study included 31 children with histologically confirmed leukemia cutis at one of two pediatric institutions. The researchers reviewed medical records to distinguish common features among patients.
Various clinical data, including disease subtype, related symptoms, management, and prognosis, were collected from January 1993 to March 2014. The children in the case series ranged in age up to 19 years with a median age at diagnosis of 26.8 months.
After analysis, the researchers reported that the magnitude and morphology of disease lesions differed among pediatric patients, with the most common sites being the lower extremities and head. The most common morphologies were nodules and papules. Additionally, the researchers found that lesions were often erythematous, violaceous, or both colors.
The majority of patients (65%) presented with concomitant systemic leukemia and leukemia cutis. The most common types of leukemia associated with the skin condition were acute myeloid leukemia (in 74% of cases) and acute lymphoblastic leukemia (in 16% of cases). The researchers saw no significant differences in leukemia cutis morphology or distribution based on the leukemia diagnosis.
“Most cases of leukemia cutis arose during initial leukemia episodes, rather than with relapsed leukemia,” they added.
Because of an insufficiency of specific genetic data, investigators were unable to make prognostic inferences in the majority of participants.
Two key limitations of the study were the small sample size and retrospective design. As a result, the investigators were unable to prospectively classify skin findings in a systematic manner. Despite these limitations, the authors noted that these findings add to the present knowledge of leukemia cutis in pediatric patients.
“Importantly, the presence of [leukemia cutis] changed the management of systemic leukemia in one‐third of patients,” the researchers wrote. “The potential for major changes in treatment plans such as adding radiation therapy and deferring hematopoietic stem cell transplantation underscores the importance of diagnosing [leukemia cutis].”
No funding sources were reported. The authors did not report conflicts of interest.
SOURCE: Andriescu EC et al. Pediatr Dermatol. 2019 Jul 5. doi: 10.1111/pde.13864.
Researchers have characterized the clinical presentation, progression, and prognosis of leukemia cutis in a pediatric population, according to findings from a retrospective case series.
“To our knowledge, this is the largest reported case series of pediatric leukemia cutis,” wrote Elena Corina Andriescu of the University of Texas, Houston, and colleagues. The results were published in Pediatric Dermatology.
The study included 31 children with histologically confirmed leukemia cutis at one of two pediatric institutions. The researchers reviewed medical records to distinguish common features among patients.
Various clinical data, including disease subtype, related symptoms, management, and prognosis, were collected from January 1993 to March 2014. The children in the case series ranged in age up to 19 years with a median age at diagnosis of 26.8 months.
After analysis, the researchers reported that the magnitude and morphology of disease lesions differed among pediatric patients, with the most common sites being the lower extremities and head. The most common morphologies were nodules and papules. Additionally, the researchers found that lesions were often erythematous, violaceous, or both colors.
The majority of patients (65%) presented with concomitant systemic leukemia and leukemia cutis. The most common types of leukemia associated with the skin condition were acute myeloid leukemia (in 74% of cases) and acute lymphoblastic leukemia (in 16% of cases). The researchers saw no significant differences in leukemia cutis morphology or distribution based on the leukemia diagnosis.
“Most cases of leukemia cutis arose during initial leukemia episodes, rather than with relapsed leukemia,” they added.
Because of an insufficiency of specific genetic data, investigators were unable to make prognostic inferences in the majority of participants.
Two key limitations of the study were the small sample size and retrospective design. As a result, the investigators were unable to prospectively classify skin findings in a systematic manner. Despite these limitations, the authors noted that these findings add to the present knowledge of leukemia cutis in pediatric patients.
“Importantly, the presence of [leukemia cutis] changed the management of systemic leukemia in one‐third of patients,” the researchers wrote. “The potential for major changes in treatment plans such as adding radiation therapy and deferring hematopoietic stem cell transplantation underscores the importance of diagnosing [leukemia cutis].”
No funding sources were reported. The authors did not report conflicts of interest.
SOURCE: Andriescu EC et al. Pediatr Dermatol. 2019 Jul 5. doi: 10.1111/pde.13864.
Researchers have characterized the clinical presentation, progression, and prognosis of leukemia cutis in a pediatric population, according to findings from a retrospective case series.
“To our knowledge, this is the largest reported case series of pediatric leukemia cutis,” wrote Elena Corina Andriescu of the University of Texas, Houston, and colleagues. The results were published in Pediatric Dermatology.
The study included 31 children with histologically confirmed leukemia cutis at one of two pediatric institutions. The researchers reviewed medical records to distinguish common features among patients.
Various clinical data, including disease subtype, related symptoms, management, and prognosis, were collected from January 1993 to March 2014. The children in the case series ranged in age up to 19 years with a median age at diagnosis of 26.8 months.
After analysis, the researchers reported that the magnitude and morphology of disease lesions differed among pediatric patients, with the most common sites being the lower extremities and head. The most common morphologies were nodules and papules. Additionally, the researchers found that lesions were often erythematous, violaceous, or both colors.
The majority of patients (65%) presented with concomitant systemic leukemia and leukemia cutis. The most common types of leukemia associated with the skin condition were acute myeloid leukemia (in 74% of cases) and acute lymphoblastic leukemia (in 16% of cases). The researchers saw no significant differences in leukemia cutis morphology or distribution based on the leukemia diagnosis.
“Most cases of leukemia cutis arose during initial leukemia episodes, rather than with relapsed leukemia,” they added.
Because of an insufficiency of specific genetic data, investigators were unable to make prognostic inferences in the majority of participants.
Two key limitations of the study were the small sample size and retrospective design. As a result, the investigators were unable to prospectively classify skin findings in a systematic manner. Despite these limitations, the authors noted that these findings add to the present knowledge of leukemia cutis in pediatric patients.
“Importantly, the presence of [leukemia cutis] changed the management of systemic leukemia in one‐third of patients,” the researchers wrote. “The potential for major changes in treatment plans such as adding radiation therapy and deferring hematopoietic stem cell transplantation underscores the importance of diagnosing [leukemia cutis].”
No funding sources were reported. The authors did not report conflicts of interest.
SOURCE: Andriescu EC et al. Pediatr Dermatol. 2019 Jul 5. doi: 10.1111/pde.13864.
FROM PEDIATRIC DERMATOLOGY
Racial, ethnic minorities often don’t practice sun protective behaviors
Despite higher rates of skin cancer morbidity and mortality among racial and ethnic minorities, affected adults often are not recognizing their risks or taking preventive measures, said Costner McKenzie, BA, and Roopal V. Kundu, MD of Northwestern University, Chicago.
In a multivariable logistic regression analysis, Mr. Costner and Dr. Kundu sampled data of 33,672 adults included in the 2015 National Health Interview Survey. Data from the 2010 U.S. Census Bureau also were used to develop sample weights representative of the U.S. population. There was a survey of a smaller sample of adults who were determined to have sun-sensitive skin. The findings were published in the Journal of the American Academy of Dermatology.
Sun sensitivity was determined by skin reaction to 1 hour of unprotected sun exposure. Those who self-reported severe sunburn with blisters or moderate sunburn with peeling were determined to be sun sensitive.
The sample surveyed comprised 3,665 women (41%) and 5,287 men (59%). Of these, 82% were white non-Hispanic, 3% black non-Hispanic, 3% Asian non-Hispanic, 11% Hispanic, and 1% other non-Hispanic.
Mr. McKenzie and Dr. Kundu found that (adjusted odds ratio [aOR], 0.43, 0.54, and 0.70, respectively). Non-Hispanic blacks and Hispanics also were less likely to use sunscreen greater than SPF 15 (a0R, 0.39 and 0.64, respectively). Non-Hispanic blacks, non-Hispanic Asians, and Hispanics were less likely to have ever had a total body skin examination (aOR, 0.29, 0.21, and 0.39, respectively).
Yet these same three groups were more likely to wear long sleeves outside (non-Hispanic blacks aOR, 1.96, non-Hispanic Asians aOR, 2.09, and Hispanics aOR, 2.29). In addition, non-Hispanic Asians and Hispanics were more likely to shelter in the shade on warm, sunny days (aOR, 1.63 and 1.85, respectively).
Citing recent literature, the authors noted that although skin cancer is the most commonly diagnosed cancer, it is not typically thought of as a disease that afflicts minority populations, especially among minorities themselves, who do not generally recognize their own risk (Arch Dermatol. 2009;145[2]:207-8). In fact, morbidity and mortality from skin cancer actually are greater in racial and ethnic minorities (J Am Acad Dermatol. 2016;75[5]:983-91; J Am Acad Dermatol. 2006;55[5]:741-60), despite greater incidence of skin cancer among white adults.
“This study highlights the impact of race and ethnicity on sun protective behaviors,” said Mr. McKenzie and Dr. Kundu. Cultural beliefs, stigma, personal preferences, as well as a lack of “knowledge-based interventions” specifically intended for minorities could be responsible for the observed differences between population groups, they speculated.
The primary limitations of the study were its cross-sectional design and the use of self-reported data, the authors noted.
Additional research is needed to fully examine the reasons behind these differences as well as to identify appropriate interventions that promote sun protection, they added.
There was no external funding and the authors had no conflicts of interest to disclose.
SOURCE: McKenzie C and Kundu RV. J Am Acad Dermatol. 2019 Jun 19. doi: 10.1016/j.jaad.2019.06.1306.
Despite higher rates of skin cancer morbidity and mortality among racial and ethnic minorities, affected adults often are not recognizing their risks or taking preventive measures, said Costner McKenzie, BA, and Roopal V. Kundu, MD of Northwestern University, Chicago.
In a multivariable logistic regression analysis, Mr. Costner and Dr. Kundu sampled data of 33,672 adults included in the 2015 National Health Interview Survey. Data from the 2010 U.S. Census Bureau also were used to develop sample weights representative of the U.S. population. There was a survey of a smaller sample of adults who were determined to have sun-sensitive skin. The findings were published in the Journal of the American Academy of Dermatology.
Sun sensitivity was determined by skin reaction to 1 hour of unprotected sun exposure. Those who self-reported severe sunburn with blisters or moderate sunburn with peeling were determined to be sun sensitive.
The sample surveyed comprised 3,665 women (41%) and 5,287 men (59%). Of these, 82% were white non-Hispanic, 3% black non-Hispanic, 3% Asian non-Hispanic, 11% Hispanic, and 1% other non-Hispanic.
Mr. McKenzie and Dr. Kundu found that (adjusted odds ratio [aOR], 0.43, 0.54, and 0.70, respectively). Non-Hispanic blacks and Hispanics also were less likely to use sunscreen greater than SPF 15 (a0R, 0.39 and 0.64, respectively). Non-Hispanic blacks, non-Hispanic Asians, and Hispanics were less likely to have ever had a total body skin examination (aOR, 0.29, 0.21, and 0.39, respectively).
Yet these same three groups were more likely to wear long sleeves outside (non-Hispanic blacks aOR, 1.96, non-Hispanic Asians aOR, 2.09, and Hispanics aOR, 2.29). In addition, non-Hispanic Asians and Hispanics were more likely to shelter in the shade on warm, sunny days (aOR, 1.63 and 1.85, respectively).
Citing recent literature, the authors noted that although skin cancer is the most commonly diagnosed cancer, it is not typically thought of as a disease that afflicts minority populations, especially among minorities themselves, who do not generally recognize their own risk (Arch Dermatol. 2009;145[2]:207-8). In fact, morbidity and mortality from skin cancer actually are greater in racial and ethnic minorities (J Am Acad Dermatol. 2016;75[5]:983-91; J Am Acad Dermatol. 2006;55[5]:741-60), despite greater incidence of skin cancer among white adults.
“This study highlights the impact of race and ethnicity on sun protective behaviors,” said Mr. McKenzie and Dr. Kundu. Cultural beliefs, stigma, personal preferences, as well as a lack of “knowledge-based interventions” specifically intended for minorities could be responsible for the observed differences between population groups, they speculated.
The primary limitations of the study were its cross-sectional design and the use of self-reported data, the authors noted.
Additional research is needed to fully examine the reasons behind these differences as well as to identify appropriate interventions that promote sun protection, they added.
There was no external funding and the authors had no conflicts of interest to disclose.
SOURCE: McKenzie C and Kundu RV. J Am Acad Dermatol. 2019 Jun 19. doi: 10.1016/j.jaad.2019.06.1306.
Despite higher rates of skin cancer morbidity and mortality among racial and ethnic minorities, affected adults often are not recognizing their risks or taking preventive measures, said Costner McKenzie, BA, and Roopal V. Kundu, MD of Northwestern University, Chicago.
In a multivariable logistic regression analysis, Mr. Costner and Dr. Kundu sampled data of 33,672 adults included in the 2015 National Health Interview Survey. Data from the 2010 U.S. Census Bureau also were used to develop sample weights representative of the U.S. population. There was a survey of a smaller sample of adults who were determined to have sun-sensitive skin. The findings were published in the Journal of the American Academy of Dermatology.
Sun sensitivity was determined by skin reaction to 1 hour of unprotected sun exposure. Those who self-reported severe sunburn with blisters or moderate sunburn with peeling were determined to be sun sensitive.
The sample surveyed comprised 3,665 women (41%) and 5,287 men (59%). Of these, 82% were white non-Hispanic, 3% black non-Hispanic, 3% Asian non-Hispanic, 11% Hispanic, and 1% other non-Hispanic.
Mr. McKenzie and Dr. Kundu found that (adjusted odds ratio [aOR], 0.43, 0.54, and 0.70, respectively). Non-Hispanic blacks and Hispanics also were less likely to use sunscreen greater than SPF 15 (a0R, 0.39 and 0.64, respectively). Non-Hispanic blacks, non-Hispanic Asians, and Hispanics were less likely to have ever had a total body skin examination (aOR, 0.29, 0.21, and 0.39, respectively).
Yet these same three groups were more likely to wear long sleeves outside (non-Hispanic blacks aOR, 1.96, non-Hispanic Asians aOR, 2.09, and Hispanics aOR, 2.29). In addition, non-Hispanic Asians and Hispanics were more likely to shelter in the shade on warm, sunny days (aOR, 1.63 and 1.85, respectively).
Citing recent literature, the authors noted that although skin cancer is the most commonly diagnosed cancer, it is not typically thought of as a disease that afflicts minority populations, especially among minorities themselves, who do not generally recognize their own risk (Arch Dermatol. 2009;145[2]:207-8). In fact, morbidity and mortality from skin cancer actually are greater in racial and ethnic minorities (J Am Acad Dermatol. 2016;75[5]:983-91; J Am Acad Dermatol. 2006;55[5]:741-60), despite greater incidence of skin cancer among white adults.
“This study highlights the impact of race and ethnicity on sun protective behaviors,” said Mr. McKenzie and Dr. Kundu. Cultural beliefs, stigma, personal preferences, as well as a lack of “knowledge-based interventions” specifically intended for minorities could be responsible for the observed differences between population groups, they speculated.
The primary limitations of the study were its cross-sectional design and the use of self-reported data, the authors noted.
Additional research is needed to fully examine the reasons behind these differences as well as to identify appropriate interventions that promote sun protection, they added.
There was no external funding and the authors had no conflicts of interest to disclose.
SOURCE: McKenzie C and Kundu RV. J Am Acad Dermatol. 2019 Jun 19. doi: 10.1016/j.jaad.2019.06.1306.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Grouped Erythematous Papules and Plaques on the Trunk
The Diagnosis: Cutaneous B-Cell Lymphoma, Follicle Center Subtype
A 4-mm punch biopsy through the center of the largest lesion on the right posterior shoulder demonstrated a superficial and deep dermal atypical lymphoid infiltrate composed predominantly of small mature lymphocytes with interspersed intermediate-sized cells with irregular to cleaved nuclei, dispersed chromatin, one or more distinct nucleoli, occasional mitoses, and small amounts of cytoplasm (Figure, A). Immunoperoxidase studies showed the infiltrate to be a mixture of CD3+ T cells and CD20+ B cells (Figure, B). The B cells coexpressed B-cell lymphoma (Bcl) 6 protein (Figure, C) but were negative for multiple myeloma 1/interferon regulatory factor 4 and CD10; Bcl2 protein was positive in T cells but inconclusive for staining in B cells. Very few plasma cells were seen with CD138 stain. Fluorescence in situ hybridization studies were negative for IgH and BCL2 gene rearrangement. Molecular diagnostic studies for IgH and κ light chain gene rearrangement were positive for a clonal population. A clonal T-cell receptor γ chain gene rearrangement was not identified. The overall morphologic, immunophenotypic, and molecular findings were consistent with cutaneous involvement by a B-cell lymphoproliferative disorder, favoring primary cutaneous follicle center lymphoma (PCFCL).

The patient was referred to our cancer center for further workup consisting of a complete blood cell count with differential; comprehensive metabolic panel; lactate dehydrogenase; serum protein electrophoresis; peripheral blood flow cytometry; and computed tomography of the chest, abdomen, and pelvis. The analysis was unremarkable, supporting primary cutaneous disease. Additional studies suggested in the National Comprehensive Cancer Network (NCCN) Guidelines for primary cutaneous B-cell lymphomas include hepatitis B testing if the patient is being considered for immunotherapy and/or chemotherapy due to risk of reactivation, pregnancy testing in women of childbearing age, and human immunodeficiency virus testing.1 These tests were not performed in our patient because he did not have any risk factors for hepatitis B or human immunodeficiency virus.
Primary cutaneous B-cell lymphomas originate in the skin without evidence of extracutaneous disease at presentation. They account for approximately 25% of primary cutaneous lymphomas in the United States, with primary cutaneous T-cell lymphoma being most common.2 The revised 2017 World Health Organization classification system defines 3 major subtypes of primary cutaneous B-cell lymphoma (Table).3-9 Primary cutaneous follicle center lymphoma is the most common subtype, accounting for approximately 60% of cases. In Europe, an association with Borrelia burgdorferi has been reported.10 The extent of skin involvement determines the T portion of TNM staging for PCFCL. It is based on the size and location of affected body regions that are delineated, such as the head and neck, chest, abdomen/genitalia, upper back, lower back/buttocks, each upper arm, each lower arm/hand, each upper leg, and each lower leg/foot. T1 is for solitary skin involvement in which the lesion is 5 cm or less in diameter (T1a) or greater than 5 cm (T1b). T2 is for regional skin involvement limited to 1 or 2 contiguous body regions, whereas T2a has all lesions confined to an area 15 cm or less in diameter, T2b has lesions confined to an area greater than 15 cm up to 30 cm in diameter, and the area for T2c is greater than 30 cm in diameter. Finally, T3 is generalized skin involvement, whereas T3a has multiple lesions in 2 noncontiguous body regions, and T3b has multiple lesions on 3 or more regions.11 At presentation, our patient was considered T2cN0M0, as his lesions were present on only 2 contiguous regions extending beyond 30 cm without any evidence of lymph node involvement or metastasis.
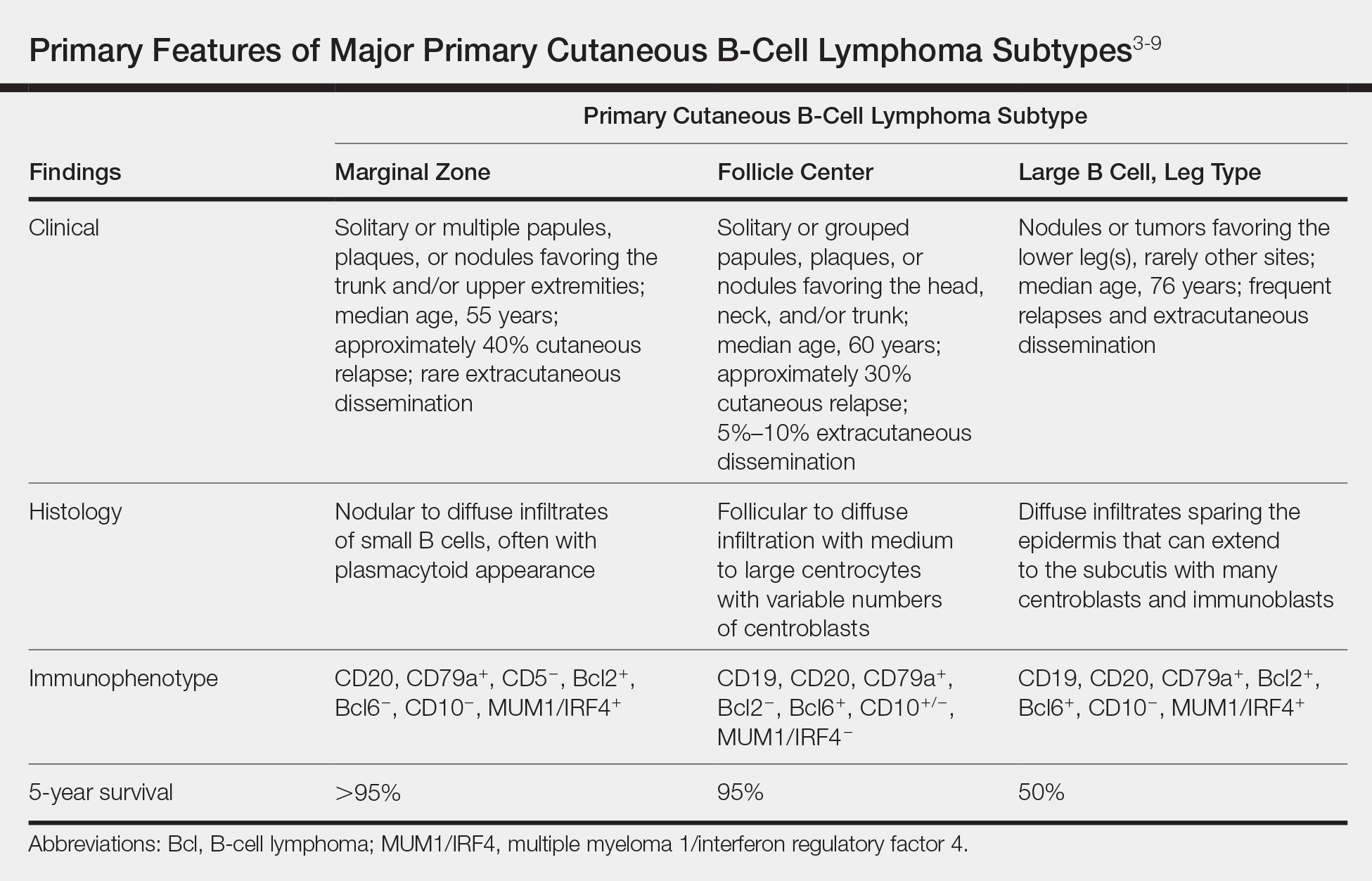
Treatment of PCFCL is tailored to each case, as there is a paucity of randomized data in this rare entity. It is guided by the number and location of cutaneous lesions, associated skin symptoms, age of the patient, and performance status. Local disease can be treated with intralesional corticosteroids, excision, or close monitoring if the patient is asymptomatic. Low-dose radiation therapy may be used as primary treatment or for local recurrence.12 Patients with more extensive skin lesions can relapse after clearing; those with refractory disease can be managed with single-agent rituximab.13 Our patient underwent low-dose radiation therapy with good response and has not experienced recurrence.
Lymphocytoma cutis, also known as benign reactive lymphoid hyperplasia, can be idiopathic or can arise after arthropod assault, penetrative skin trauma, drugs, or infections. In granuloma annulare, small dermal papules may present in isolation or coalesce to form annular plaques. It is a benign inflammatory disorder of unknown cause, can have mild pruritus, and usually is self-limited. Pyogenic granuloma is a benign vascular proliferation of unknown etiology. Sarcoidosis is an immune-mediated systemic disorder with granuloma formation that has a predilection for the lungs and the skin.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Primary Cutaneous B-Cell Lymphomas. Version 2.2018. https://oncolife.com.ua/doc/nccn/Primary_Cutaneous_B-Cell_Lymphomas.pdf. Published January 10, 2018. Accessed June 21, 2019.
- Dores GM, Anderson WF, Devesa SS. Cutaneous lymphomas reported to the National Cancer Institute's surveillance, epidemiology, and end results program: applying the new WHO-European Organisation for Research and Treatment of Cancer classification system. J Clin Oncol. 2005;23:7246-7248.
- Swerdlow SH, Campo E, Harris NL, et al, eds. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th ed. Lyon, France: IARC; 2017.
- Surveillance, Epidemiology, and End Results Program. National Cancer Institute website. https://seer.cancer.gov/. Accessed June 26, 2019.
- Cerroni L. B-cell lymphomas of the skin. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. China: Elsevier; 2018:2113-2126.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous follicle center lymphoma. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-follicle-center-lymphoma. Updated February 7, 2018. Accessed June 26, 2019.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous marginal zone lymphoma. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-marginal-zone-lymphoma. Updated March 6, 2019. Accessed June 26, 2019.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous large B cell lymphoma, leg type. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-large-b-cell-lymphoma-leg-type. Updated July 3, 2017. Accessed June 26, 2019.
- Suárez AL, Pulitzer M, Horwitz S, et al. Primary cutaneous B-cell lymphomas: part I. clinical features, diagnosis, and classification. J Am Acad Dermatol. 2013;69:329.e1-13; quiz 241-342.
- Goodlad JR, Davidson MM, Hollowood K, et al. Primary cutaneous B-cell lymphoma and Borrelia burgdorferi infection in patients from the Highlands of Scotand. Am J Surg Pathol. 2000;24:1279-1285.
- Kim YH, Willemze R, Pimpinelli N, et al. TNM classification system for primary cutaneous lymphomas other than mycosis fungoides and Sezary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110:479-484.
- Wilcon RA. Cutaneous B-cell lymphomas: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 2016;91:1052-1055.
- Morales AV, Advani R, Horwitz SM, et al. Indolent primary cutaneous B-cell lymphoma: experience using systemic rituximab. J Am Acad Dermatol. 2008;59:953-957.
The Diagnosis: Cutaneous B-Cell Lymphoma, Follicle Center Subtype
A 4-mm punch biopsy through the center of the largest lesion on the right posterior shoulder demonstrated a superficial and deep dermal atypical lymphoid infiltrate composed predominantly of small mature lymphocytes with interspersed intermediate-sized cells with irregular to cleaved nuclei, dispersed chromatin, one or more distinct nucleoli, occasional mitoses, and small amounts of cytoplasm (Figure, A). Immunoperoxidase studies showed the infiltrate to be a mixture of CD3+ T cells and CD20+ B cells (Figure, B). The B cells coexpressed B-cell lymphoma (Bcl) 6 protein (Figure, C) but were negative for multiple myeloma 1/interferon regulatory factor 4 and CD10; Bcl2 protein was positive in T cells but inconclusive for staining in B cells. Very few plasma cells were seen with CD138 stain. Fluorescence in situ hybridization studies were negative for IgH and BCL2 gene rearrangement. Molecular diagnostic studies for IgH and κ light chain gene rearrangement were positive for a clonal population. A clonal T-cell receptor γ chain gene rearrangement was not identified. The overall morphologic, immunophenotypic, and molecular findings were consistent with cutaneous involvement by a B-cell lymphoproliferative disorder, favoring primary cutaneous follicle center lymphoma (PCFCL).

The patient was referred to our cancer center for further workup consisting of a complete blood cell count with differential; comprehensive metabolic panel; lactate dehydrogenase; serum protein electrophoresis; peripheral blood flow cytometry; and computed tomography of the chest, abdomen, and pelvis. The analysis was unremarkable, supporting primary cutaneous disease. Additional studies suggested in the National Comprehensive Cancer Network (NCCN) Guidelines for primary cutaneous B-cell lymphomas include hepatitis B testing if the patient is being considered for immunotherapy and/or chemotherapy due to risk of reactivation, pregnancy testing in women of childbearing age, and human immunodeficiency virus testing.1 These tests were not performed in our patient because he did not have any risk factors for hepatitis B or human immunodeficiency virus.
Primary cutaneous B-cell lymphomas originate in the skin without evidence of extracutaneous disease at presentation. They account for approximately 25% of primary cutaneous lymphomas in the United States, with primary cutaneous T-cell lymphoma being most common.2 The revised 2017 World Health Organization classification system defines 3 major subtypes of primary cutaneous B-cell lymphoma (Table).3-9 Primary cutaneous follicle center lymphoma is the most common subtype, accounting for approximately 60% of cases. In Europe, an association with Borrelia burgdorferi has been reported.10 The extent of skin involvement determines the T portion of TNM staging for PCFCL. It is based on the size and location of affected body regions that are delineated, such as the head and neck, chest, abdomen/genitalia, upper back, lower back/buttocks, each upper arm, each lower arm/hand, each upper leg, and each lower leg/foot. T1 is for solitary skin involvement in which the lesion is 5 cm or less in diameter (T1a) or greater than 5 cm (T1b). T2 is for regional skin involvement limited to 1 or 2 contiguous body regions, whereas T2a has all lesions confined to an area 15 cm or less in diameter, T2b has lesions confined to an area greater than 15 cm up to 30 cm in diameter, and the area for T2c is greater than 30 cm in diameter. Finally, T3 is generalized skin involvement, whereas T3a has multiple lesions in 2 noncontiguous body regions, and T3b has multiple lesions on 3 or more regions.11 At presentation, our patient was considered T2cN0M0, as his lesions were present on only 2 contiguous regions extending beyond 30 cm without any evidence of lymph node involvement or metastasis.

Treatment of PCFCL is tailored to each case, as there is a paucity of randomized data in this rare entity. It is guided by the number and location of cutaneous lesions, associated skin symptoms, age of the patient, and performance status. Local disease can be treated with intralesional corticosteroids, excision, or close monitoring if the patient is asymptomatic. Low-dose radiation therapy may be used as primary treatment or for local recurrence.12 Patients with more extensive skin lesions can relapse after clearing; those with refractory disease can be managed with single-agent rituximab.13 Our patient underwent low-dose radiation therapy with good response and has not experienced recurrence.
Lymphocytoma cutis, also known as benign reactive lymphoid hyperplasia, can be idiopathic or can arise after arthropod assault, penetrative skin trauma, drugs, or infections. In granuloma annulare, small dermal papules may present in isolation or coalesce to form annular plaques. It is a benign inflammatory disorder of unknown cause, can have mild pruritus, and usually is self-limited. Pyogenic granuloma is a benign vascular proliferation of unknown etiology. Sarcoidosis is an immune-mediated systemic disorder with granuloma formation that has a predilection for the lungs and the skin.
The Diagnosis: Cutaneous B-Cell Lymphoma, Follicle Center Subtype
A 4-mm punch biopsy through the center of the largest lesion on the right posterior shoulder demonstrated a superficial and deep dermal atypical lymphoid infiltrate composed predominantly of small mature lymphocytes with interspersed intermediate-sized cells with irregular to cleaved nuclei, dispersed chromatin, one or more distinct nucleoli, occasional mitoses, and small amounts of cytoplasm (Figure, A). Immunoperoxidase studies showed the infiltrate to be a mixture of CD3+ T cells and CD20+ B cells (Figure, B). The B cells coexpressed B-cell lymphoma (Bcl) 6 protein (Figure, C) but were negative for multiple myeloma 1/interferon regulatory factor 4 and CD10; Bcl2 protein was positive in T cells but inconclusive for staining in B cells. Very few plasma cells were seen with CD138 stain. Fluorescence in situ hybridization studies were negative for IgH and BCL2 gene rearrangement. Molecular diagnostic studies for IgH and κ light chain gene rearrangement were positive for a clonal population. A clonal T-cell receptor γ chain gene rearrangement was not identified. The overall morphologic, immunophenotypic, and molecular findings were consistent with cutaneous involvement by a B-cell lymphoproliferative disorder, favoring primary cutaneous follicle center lymphoma (PCFCL).

The patient was referred to our cancer center for further workup consisting of a complete blood cell count with differential; comprehensive metabolic panel; lactate dehydrogenase; serum protein electrophoresis; peripheral blood flow cytometry; and computed tomography of the chest, abdomen, and pelvis. The analysis was unremarkable, supporting primary cutaneous disease. Additional studies suggested in the National Comprehensive Cancer Network (NCCN) Guidelines for primary cutaneous B-cell lymphomas include hepatitis B testing if the patient is being considered for immunotherapy and/or chemotherapy due to risk of reactivation, pregnancy testing in women of childbearing age, and human immunodeficiency virus testing.1 These tests were not performed in our patient because he did not have any risk factors for hepatitis B or human immunodeficiency virus.
Primary cutaneous B-cell lymphomas originate in the skin without evidence of extracutaneous disease at presentation. They account for approximately 25% of primary cutaneous lymphomas in the United States, with primary cutaneous T-cell lymphoma being most common.2 The revised 2017 World Health Organization classification system defines 3 major subtypes of primary cutaneous B-cell lymphoma (Table).3-9 Primary cutaneous follicle center lymphoma is the most common subtype, accounting for approximately 60% of cases. In Europe, an association with Borrelia burgdorferi has been reported.10 The extent of skin involvement determines the T portion of TNM staging for PCFCL. It is based on the size and location of affected body regions that are delineated, such as the head and neck, chest, abdomen/genitalia, upper back, lower back/buttocks, each upper arm, each lower arm/hand, each upper leg, and each lower leg/foot. T1 is for solitary skin involvement in which the lesion is 5 cm or less in diameter (T1a) or greater than 5 cm (T1b). T2 is for regional skin involvement limited to 1 or 2 contiguous body regions, whereas T2a has all lesions confined to an area 15 cm or less in diameter, T2b has lesions confined to an area greater than 15 cm up to 30 cm in diameter, and the area for T2c is greater than 30 cm in diameter. Finally, T3 is generalized skin involvement, whereas T3a has multiple lesions in 2 noncontiguous body regions, and T3b has multiple lesions on 3 or more regions.11 At presentation, our patient was considered T2cN0M0, as his lesions were present on only 2 contiguous regions extending beyond 30 cm without any evidence of lymph node involvement or metastasis.

Treatment of PCFCL is tailored to each case, as there is a paucity of randomized data in this rare entity. It is guided by the number and location of cutaneous lesions, associated skin symptoms, age of the patient, and performance status. Local disease can be treated with intralesional corticosteroids, excision, or close monitoring if the patient is asymptomatic. Low-dose radiation therapy may be used as primary treatment or for local recurrence.12 Patients with more extensive skin lesions can relapse after clearing; those with refractory disease can be managed with single-agent rituximab.13 Our patient underwent low-dose radiation therapy with good response and has not experienced recurrence.
Lymphocytoma cutis, also known as benign reactive lymphoid hyperplasia, can be idiopathic or can arise after arthropod assault, penetrative skin trauma, drugs, or infections. In granuloma annulare, small dermal papules may present in isolation or coalesce to form annular plaques. It is a benign inflammatory disorder of unknown cause, can have mild pruritus, and usually is self-limited. Pyogenic granuloma is a benign vascular proliferation of unknown etiology. Sarcoidosis is an immune-mediated systemic disorder with granuloma formation that has a predilection for the lungs and the skin.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Primary Cutaneous B-Cell Lymphomas. Version 2.2018. https://oncolife.com.ua/doc/nccn/Primary_Cutaneous_B-Cell_Lymphomas.pdf. Published January 10, 2018. Accessed June 21, 2019.
- Dores GM, Anderson WF, Devesa SS. Cutaneous lymphomas reported to the National Cancer Institute's surveillance, epidemiology, and end results program: applying the new WHO-European Organisation for Research and Treatment of Cancer classification system. J Clin Oncol. 2005;23:7246-7248.
- Swerdlow SH, Campo E, Harris NL, et al, eds. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th ed. Lyon, France: IARC; 2017.
- Surveillance, Epidemiology, and End Results Program. National Cancer Institute website. https://seer.cancer.gov/. Accessed June 26, 2019.
- Cerroni L. B-cell lymphomas of the skin. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. China: Elsevier; 2018:2113-2126.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous follicle center lymphoma. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-follicle-center-lymphoma. Updated February 7, 2018. Accessed June 26, 2019.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous marginal zone lymphoma. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-marginal-zone-lymphoma. Updated March 6, 2019. Accessed June 26, 2019.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous large B cell lymphoma, leg type. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-large-b-cell-lymphoma-leg-type. Updated July 3, 2017. Accessed June 26, 2019.
- Suárez AL, Pulitzer M, Horwitz S, et al. Primary cutaneous B-cell lymphomas: part I. clinical features, diagnosis, and classification. J Am Acad Dermatol. 2013;69:329.e1-13; quiz 241-342.
- Goodlad JR, Davidson MM, Hollowood K, et al. Primary cutaneous B-cell lymphoma and Borrelia burgdorferi infection in patients from the Highlands of Scotand. Am J Surg Pathol. 2000;24:1279-1285.
- Kim YH, Willemze R, Pimpinelli N, et al. TNM classification system for primary cutaneous lymphomas other than mycosis fungoides and Sezary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110:479-484.
- Wilcon RA. Cutaneous B-cell lymphomas: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 2016;91:1052-1055.
- Morales AV, Advani R, Horwitz SM, et al. Indolent primary cutaneous B-cell lymphoma: experience using systemic rituximab. J Am Acad Dermatol. 2008;59:953-957.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Primary Cutaneous B-Cell Lymphomas. Version 2.2018. https://oncolife.com.ua/doc/nccn/Primary_Cutaneous_B-Cell_Lymphomas.pdf. Published January 10, 2018. Accessed June 21, 2019.
- Dores GM, Anderson WF, Devesa SS. Cutaneous lymphomas reported to the National Cancer Institute's surveillance, epidemiology, and end results program: applying the new WHO-European Organisation for Research and Treatment of Cancer classification system. J Clin Oncol. 2005;23:7246-7248.
- Swerdlow SH, Campo E, Harris NL, et al, eds. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th ed. Lyon, France: IARC; 2017.
- Surveillance, Epidemiology, and End Results Program. National Cancer Institute website. https://seer.cancer.gov/. Accessed June 26, 2019.
- Cerroni L. B-cell lymphomas of the skin. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. China: Elsevier; 2018:2113-2126.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous follicle center lymphoma. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-follicle-center-lymphoma. Updated February 7, 2018. Accessed June 26, 2019.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous marginal zone lymphoma. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-marginal-zone-lymphoma. Updated March 6, 2019. Accessed June 26, 2019.
- Jacobsen E, Freedman AS, Willemze R. Primary cutaneous large B cell lymphoma, leg type. UpToDate website. https://www.uptodate.com/contents/primary-cutaneous-large-b-cell-lymphoma-leg-type. Updated July 3, 2017. Accessed June 26, 2019.
- Suárez AL, Pulitzer M, Horwitz S, et al. Primary cutaneous B-cell lymphomas: part I. clinical features, diagnosis, and classification. J Am Acad Dermatol. 2013;69:329.e1-13; quiz 241-342.
- Goodlad JR, Davidson MM, Hollowood K, et al. Primary cutaneous B-cell lymphoma and Borrelia burgdorferi infection in patients from the Highlands of Scotand. Am J Surg Pathol. 2000;24:1279-1285.
- Kim YH, Willemze R, Pimpinelli N, et al. TNM classification system for primary cutaneous lymphomas other than mycosis fungoides and Sezary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110:479-484.
- Wilcon RA. Cutaneous B-cell lymphomas: 2016 update on diagnosis, risk-stratification, and management. Am J Hematol. 2016;91:1052-1055.
- Morales AV, Advani R, Horwitz SM, et al. Indolent primary cutaneous B-cell lymphoma: experience using systemic rituximab. J Am Acad Dermatol. 2008;59:953-957.
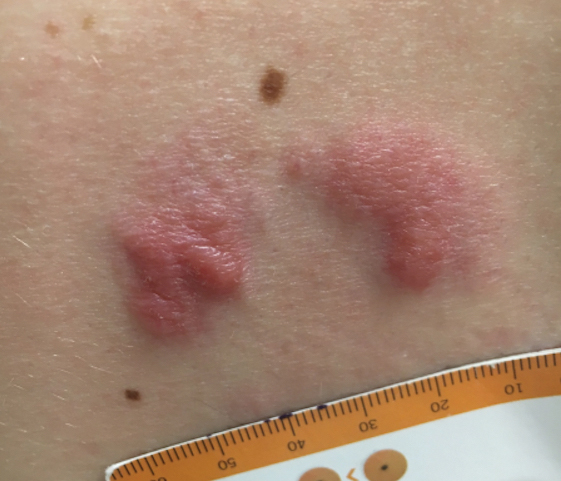
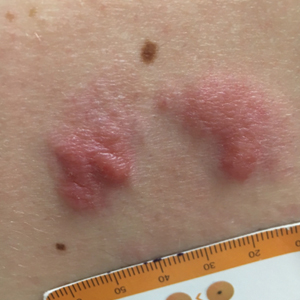
A 34-year-old man presented to the outpatient dermatology clinic with 3 groups of mildly pruritic, erythematous papules and plaques. The most prominent group appeared on the right posterior shoulder and had been slowly enlarging in size over the last 12 months (quiz image). A similar thinner group appeared on the left mid-back 6 months prior, and a third smaller group appeared over the left serratus anterior muscle 2 months prior. The patient reported having similar episodes dating back to his early 20s. In those instances, the lesions presented without an inciting incident, became more pronounced, and persisted for months to years before resolving. Previously affected areas included the upper and lateral back, flanks, and posterior upper arms. The patient used triamcinolone cream 0.1% up to 3 times daily on active lesions, which improved the pruritus and seemed to make the lesions resolve more quickly. He denied fever, chills, night sweats, anorexia, weight loss, fatigue, cough, and shortness of breath. His only medication was ranitidine 150 mg twice daily for gastroesophageal reflux disease. Physical examination revealed no palpable lymphadenopathy.
Smoking linked to increased complication risk after Mohs surgery
, based on data from a retrospective case-control study of 1,008 adult patients.
The increased risk of complications for smokers following many types of surgery is well documented; however, “the effect of smoking in the specific setting of cutaneous tissue transfer is not well characterized in the literature describing outcomes after Mohs reconstruction,” wrote Chang Ye Wang, MD, of St. Louis University, Missouri, and colleagues.
To determine the impact of smoking on acute and long-term complications, the researchers reviewed data from 1,008 adults (396 women and 612 men) who underwent Mohs surgery between July 1, 2012, and June 30, 2016, at a single center. The study population included 128 current smokers, 385 former smokers, and 495 never smokers. The age of the patients ranged from 21 years to 90 years, with a median of 70 years. The results were published in JAMA Facial Plastic Surgery.
The overall rate of acute complications was 4.1%, and the most common complication was infection, in 19 cases; others were 10 cases of flap or graft necrosis, 10 cases of wound dehiscence, and 6 of cases of hematoma or uncontrolled bleeding; some patients experienced more than one of these complications. The risk of acute complications increased for current smokers (odds ratio 9.58) and former smokers (OR, 3.64) in a multivariate analysis. Increased risk of acute complications also was associated with a larger defect (OR, 2.25) and use of free cartilage graft (OR, 8.19).
The researchers defined acute complications as “any postsurgical infection, dehiscence, hematoma, uncontrolled bleeding, and tissue necrosis that required medical counseling or intervention,” and long-term complications as “any postsurgical functional defect or unsatisfactory cosmesis that prompted the patient to request an additional procedural intervention or the surgeon to offer it.”
The overall rate of long-term complications was 7.4%. A procedure in the center of the face was associated with a 25% increased risk of long-term complications (OR, 25.4). Other factors associated with an increased risk of long-term complications were the use of interpolation flap or flap-graft combination (OR, 3.49), larger flaps (OR, 1.42), and presence of basal cell carcinomas or other basaloid tumors (OR, 3.43). Smoking was not associated with an increased risk of long-term complications, and an older age was associated with a decreased risk of long-term complications (OR, 0.66).
The findings were limited by the retrospective study design and unblinded data collection, as well as a lack of photographs of all patients at matching time points, the researchers said. However, the results are consistent with previous studies and “may allow the surgeon to better quantify the magnitude of risk and provide helpful information for patient counseling,” they added.
The researchers had no financial conflicts to disclose.
SOURCE: Wang CY et al. JAMA Facial Plast. Surg. 2019 June 13. doi: 10.1001/jamafacial.2019.0243.
, based on data from a retrospective case-control study of 1,008 adult patients.
The increased risk of complications for smokers following many types of surgery is well documented; however, “the effect of smoking in the specific setting of cutaneous tissue transfer is not well characterized in the literature describing outcomes after Mohs reconstruction,” wrote Chang Ye Wang, MD, of St. Louis University, Missouri, and colleagues.
To determine the impact of smoking on acute and long-term complications, the researchers reviewed data from 1,008 adults (396 women and 612 men) who underwent Mohs surgery between July 1, 2012, and June 30, 2016, at a single center. The study population included 128 current smokers, 385 former smokers, and 495 never smokers. The age of the patients ranged from 21 years to 90 years, with a median of 70 years. The results were published in JAMA Facial Plastic Surgery.
The overall rate of acute complications was 4.1%, and the most common complication was infection, in 19 cases; others were 10 cases of flap or graft necrosis, 10 cases of wound dehiscence, and 6 of cases of hematoma or uncontrolled bleeding; some patients experienced more than one of these complications. The risk of acute complications increased for current smokers (odds ratio 9.58) and former smokers (OR, 3.64) in a multivariate analysis. Increased risk of acute complications also was associated with a larger defect (OR, 2.25) and use of free cartilage graft (OR, 8.19).
The researchers defined acute complications as “any postsurgical infection, dehiscence, hematoma, uncontrolled bleeding, and tissue necrosis that required medical counseling or intervention,” and long-term complications as “any postsurgical functional defect or unsatisfactory cosmesis that prompted the patient to request an additional procedural intervention or the surgeon to offer it.”
The overall rate of long-term complications was 7.4%. A procedure in the center of the face was associated with a 25% increased risk of long-term complications (OR, 25.4). Other factors associated with an increased risk of long-term complications were the use of interpolation flap or flap-graft combination (OR, 3.49), larger flaps (OR, 1.42), and presence of basal cell carcinomas or other basaloid tumors (OR, 3.43). Smoking was not associated with an increased risk of long-term complications, and an older age was associated with a decreased risk of long-term complications (OR, 0.66).
The findings were limited by the retrospective study design and unblinded data collection, as well as a lack of photographs of all patients at matching time points, the researchers said. However, the results are consistent with previous studies and “may allow the surgeon to better quantify the magnitude of risk and provide helpful information for patient counseling,” they added.
The researchers had no financial conflicts to disclose.
SOURCE: Wang CY et al. JAMA Facial Plast. Surg. 2019 June 13. doi: 10.1001/jamafacial.2019.0243.
, based on data from a retrospective case-control study of 1,008 adult patients.
The increased risk of complications for smokers following many types of surgery is well documented; however, “the effect of smoking in the specific setting of cutaneous tissue transfer is not well characterized in the literature describing outcomes after Mohs reconstruction,” wrote Chang Ye Wang, MD, of St. Louis University, Missouri, and colleagues.
To determine the impact of smoking on acute and long-term complications, the researchers reviewed data from 1,008 adults (396 women and 612 men) who underwent Mohs surgery between July 1, 2012, and June 30, 2016, at a single center. The study population included 128 current smokers, 385 former smokers, and 495 never smokers. The age of the patients ranged from 21 years to 90 years, with a median of 70 years. The results were published in JAMA Facial Plastic Surgery.
The overall rate of acute complications was 4.1%, and the most common complication was infection, in 19 cases; others were 10 cases of flap or graft necrosis, 10 cases of wound dehiscence, and 6 of cases of hematoma or uncontrolled bleeding; some patients experienced more than one of these complications. The risk of acute complications increased for current smokers (odds ratio 9.58) and former smokers (OR, 3.64) in a multivariate analysis. Increased risk of acute complications also was associated with a larger defect (OR, 2.25) and use of free cartilage graft (OR, 8.19).
The researchers defined acute complications as “any postsurgical infection, dehiscence, hematoma, uncontrolled bleeding, and tissue necrosis that required medical counseling or intervention,” and long-term complications as “any postsurgical functional defect or unsatisfactory cosmesis that prompted the patient to request an additional procedural intervention or the surgeon to offer it.”
The overall rate of long-term complications was 7.4%. A procedure in the center of the face was associated with a 25% increased risk of long-term complications (OR, 25.4). Other factors associated with an increased risk of long-term complications were the use of interpolation flap or flap-graft combination (OR, 3.49), larger flaps (OR, 1.42), and presence of basal cell carcinomas or other basaloid tumors (OR, 3.43). Smoking was not associated with an increased risk of long-term complications, and an older age was associated with a decreased risk of long-term complications (OR, 0.66).
The findings were limited by the retrospective study design and unblinded data collection, as well as a lack of photographs of all patients at matching time points, the researchers said. However, the results are consistent with previous studies and “may allow the surgeon to better quantify the magnitude of risk and provide helpful information for patient counseling,” they added.
The researchers had no financial conflicts to disclose.
SOURCE: Wang CY et al. JAMA Facial Plast. Surg. 2019 June 13. doi: 10.1001/jamafacial.2019.0243.
FROM JAMA FACIAL PLASTIC SURGERY
Total margin control surgery warranted for high-risk keratinocyte carcinomas
MILAN – A recent meta-analysis found that total margin control surgery substantially cut the risk of recurrence in patients with high-risk keratinocyte carcinomas, Chrysalyne Schmults, MD, said at the World Congress of Dermatology.
While standard excision will cure the vast majority of low-risk basal cell carcinoma (BCC) or squamous cell carcinoma (SCC), those classified as high risk are best treated with the advanced surgical procedure, also referred to as circumferential peripheral and deep margin assessment (CCPDMA), said Dr. Schmults, director of the Mohs and Dermatologic Surgery Center at Brigham and Women’s Hospital in Boston.
For high-risk cases, CCPDMA has superior outcomes, compared with standard excision, according to results of the meta-analysis conducted by Dr. Schmults and colleagues. The meta-analysis has not yet been published.
The in the meta-analysis of medical literature from 1993 to 2017.
“This is a really big drop,” Dr. Schmults said. “If you do total margin control surgery on your perineural invasive cases, you’re going to drop your recurrence rate by two-thirds, and that’s a really big difference.”
For carcinomas without perineural invasion, the recurrence rate is about 4.5% for standard excision and about 2.0% for CCPDMA, the analysis showed. “That’s a difference, but it’s not a big enough difference that we’re going to start doing total margin control surgery on every single basal cell and squamous cell carcinoma that comes to us,” Dr. Schmults commented.
Using techniques such as Mohs surgery that allow for CCPDMA, nearly 100% of the surgical margin can be seen; by contrast, standard histology allows for visualization of only about 1% of the marginal surface, according to Dr. Schmults.
While CCPDMA may have superior outcomes for high-risk keratinocyte carcinomas, defining what constitutes high risk remains a challenge, particularly for patients with BCCs. “We don’t have good literature on which are the rare, bad basal cells,” she said.
But more clarity may be on the way. In a paper under review for publication on Brigham and Women’s Hospital (BWH) staging criteria for BCC, Dr. Schmults and colleagues describe a subset of patients with BCC at higher risk of metastasis and death based on specific high-risk tumor characteristics.
High risk is better defined for SCCs. According to the BWH classification system for cutaneous SCCs, developed by Dr. Schmults and colleagues, high-risk features include larger tumor diameter, poorly differentiated histology, perineural invasion, and tumor invasion beyond subcutaneous fat or to bone.
Patients with SCC classified as high stage by the BWH system have about a 25% risk of metastasis or death. “These patients really need that total margin control surgery,” Dr. Schmults said.
High-stage SCC patients who did not get Mohs surgery in a study (J Clin Oncol. 2014 Feb 1;32[4]:327-34) conducted by Dr. Schmults and her associates, had a quadrupling in risk of death from disease, compared with those who did get the procedure in a more recent study (J Am Acad Dermatol. 2019 Mar;80[3]:633-8).
“These are fairly small studies,” she pointed out. But the large meta-analysis she presented at the meeting showed the same result, “that the higher the stage, the worse your tumor is, and whether it’s basal cell or squamous cell, the greater the advantage for total margin control surgery.”
Dr. Schmults reported that she is the panel chair for the National Comprehensive Cancer Network guidelines on nonmelanoma skin cancer, and that she was a cutaneous SCC committee member for the 8th edition of the American Joint Committee on Cancer cancer staging system. She also participated in the development of the BWH staging system for SCC.
MILAN – A recent meta-analysis found that total margin control surgery substantially cut the risk of recurrence in patients with high-risk keratinocyte carcinomas, Chrysalyne Schmults, MD, said at the World Congress of Dermatology.
While standard excision will cure the vast majority of low-risk basal cell carcinoma (BCC) or squamous cell carcinoma (SCC), those classified as high risk are best treated with the advanced surgical procedure, also referred to as circumferential peripheral and deep margin assessment (CCPDMA), said Dr. Schmults, director of the Mohs and Dermatologic Surgery Center at Brigham and Women’s Hospital in Boston.
For high-risk cases, CCPDMA has superior outcomes, compared with standard excision, according to results of the meta-analysis conducted by Dr. Schmults and colleagues. The meta-analysis has not yet been published.
The in the meta-analysis of medical literature from 1993 to 2017.
“This is a really big drop,” Dr. Schmults said. “If you do total margin control surgery on your perineural invasive cases, you’re going to drop your recurrence rate by two-thirds, and that’s a really big difference.”
For carcinomas without perineural invasion, the recurrence rate is about 4.5% for standard excision and about 2.0% for CCPDMA, the analysis showed. “That’s a difference, but it’s not a big enough difference that we’re going to start doing total margin control surgery on every single basal cell and squamous cell carcinoma that comes to us,” Dr. Schmults commented.
Using techniques such as Mohs surgery that allow for CCPDMA, nearly 100% of the surgical margin can be seen; by contrast, standard histology allows for visualization of only about 1% of the marginal surface, according to Dr. Schmults.
While CCPDMA may have superior outcomes for high-risk keratinocyte carcinomas, defining what constitutes high risk remains a challenge, particularly for patients with BCCs. “We don’t have good literature on which are the rare, bad basal cells,” she said.
But more clarity may be on the way. In a paper under review for publication on Brigham and Women’s Hospital (BWH) staging criteria for BCC, Dr. Schmults and colleagues describe a subset of patients with BCC at higher risk of metastasis and death based on specific high-risk tumor characteristics.
High risk is better defined for SCCs. According to the BWH classification system for cutaneous SCCs, developed by Dr. Schmults and colleagues, high-risk features include larger tumor diameter, poorly differentiated histology, perineural invasion, and tumor invasion beyond subcutaneous fat or to bone.
Patients with SCC classified as high stage by the BWH system have about a 25% risk of metastasis or death. “These patients really need that total margin control surgery,” Dr. Schmults said.
High-stage SCC patients who did not get Mohs surgery in a study (J Clin Oncol. 2014 Feb 1;32[4]:327-34) conducted by Dr. Schmults and her associates, had a quadrupling in risk of death from disease, compared with those who did get the procedure in a more recent study (J Am Acad Dermatol. 2019 Mar;80[3]:633-8).
“These are fairly small studies,” she pointed out. But the large meta-analysis she presented at the meeting showed the same result, “that the higher the stage, the worse your tumor is, and whether it’s basal cell or squamous cell, the greater the advantage for total margin control surgery.”
Dr. Schmults reported that she is the panel chair for the National Comprehensive Cancer Network guidelines on nonmelanoma skin cancer, and that she was a cutaneous SCC committee member for the 8th edition of the American Joint Committee on Cancer cancer staging system. She also participated in the development of the BWH staging system for SCC.
MILAN – A recent meta-analysis found that total margin control surgery substantially cut the risk of recurrence in patients with high-risk keratinocyte carcinomas, Chrysalyne Schmults, MD, said at the World Congress of Dermatology.
While standard excision will cure the vast majority of low-risk basal cell carcinoma (BCC) or squamous cell carcinoma (SCC), those classified as high risk are best treated with the advanced surgical procedure, also referred to as circumferential peripheral and deep margin assessment (CCPDMA), said Dr. Schmults, director of the Mohs and Dermatologic Surgery Center at Brigham and Women’s Hospital in Boston.
For high-risk cases, CCPDMA has superior outcomes, compared with standard excision, according to results of the meta-analysis conducted by Dr. Schmults and colleagues. The meta-analysis has not yet been published.
The in the meta-analysis of medical literature from 1993 to 2017.
“This is a really big drop,” Dr. Schmults said. “If you do total margin control surgery on your perineural invasive cases, you’re going to drop your recurrence rate by two-thirds, and that’s a really big difference.”
For carcinomas without perineural invasion, the recurrence rate is about 4.5% for standard excision and about 2.0% for CCPDMA, the analysis showed. “That’s a difference, but it’s not a big enough difference that we’re going to start doing total margin control surgery on every single basal cell and squamous cell carcinoma that comes to us,” Dr. Schmults commented.
Using techniques such as Mohs surgery that allow for CCPDMA, nearly 100% of the surgical margin can be seen; by contrast, standard histology allows for visualization of only about 1% of the marginal surface, according to Dr. Schmults.
While CCPDMA may have superior outcomes for high-risk keratinocyte carcinomas, defining what constitutes high risk remains a challenge, particularly for patients with BCCs. “We don’t have good literature on which are the rare, bad basal cells,” she said.
But more clarity may be on the way. In a paper under review for publication on Brigham and Women’s Hospital (BWH) staging criteria for BCC, Dr. Schmults and colleagues describe a subset of patients with BCC at higher risk of metastasis and death based on specific high-risk tumor characteristics.
High risk is better defined for SCCs. According to the BWH classification system for cutaneous SCCs, developed by Dr. Schmults and colleagues, high-risk features include larger tumor diameter, poorly differentiated histology, perineural invasion, and tumor invasion beyond subcutaneous fat or to bone.
Patients with SCC classified as high stage by the BWH system have about a 25% risk of metastasis or death. “These patients really need that total margin control surgery,” Dr. Schmults said.
High-stage SCC patients who did not get Mohs surgery in a study (J Clin Oncol. 2014 Feb 1;32[4]:327-34) conducted by Dr. Schmults and her associates, had a quadrupling in risk of death from disease, compared with those who did get the procedure in a more recent study (J Am Acad Dermatol. 2019 Mar;80[3]:633-8).
“These are fairly small studies,” she pointed out. But the large meta-analysis she presented at the meeting showed the same result, “that the higher the stage, the worse your tumor is, and whether it’s basal cell or squamous cell, the greater the advantage for total margin control surgery.”
Dr. Schmults reported that she is the panel chair for the National Comprehensive Cancer Network guidelines on nonmelanoma skin cancer, and that she was a cutaneous SCC committee member for the 8th edition of the American Joint Committee on Cancer cancer staging system. She also participated in the development of the BWH staging system for SCC.
REPORTING FROM WCD2019
Response endures in cemiplimab-treated patients with cutaneous SCC
MILAN – In an updated analysis of a pivotal phase 2 study, Michael R. Migden, MD, said at the World Congress of Dermatology.
Median duration of response was not reached at the time of the analysis, with probability of no progression or death above 80% at the 20-month mark, according to Dr. Migden, of the department of dermatology at the University of Texas MD Anderson Cancer Center, Houston.
The safety profile of cemiplimab in this study was comparable with what has been reported for other anti–programmed death agents, he said in an oral presentation at the meeting.
While the median time to response was less than 2 months, about one-fifth of patients with locally advanced disease had “unconventional” late responses, occurring up to 10 months after starting treatment, Dr. Migden said. “If you’re just putting someone on some agent like this for a few months, and say, ‘well, I don’t see anything improving,’ it could be one of these patients in this 20% that deserve a little bit longer therapy.”
Cemiplimab (Libtayo) is the only Food and Drug Administration–approved treatment for patients with locally advanced or metastatic cutaneous squamous cell carcinoma (CSCC) who are not suitable for curative surgery or radiation, Dr. Migden said in his presentation. That approval was based in part on previously reported results from the phase 2 study, known as EMPOWER-CSCC-1, which demonstrated that cemiplimab had substantial antitumor activity and durable responses.
In this update on EMPOWER-CSCC-1, Dr. Migden described results for 78 patients with locally advanced CSCC and 59 with metastatic CSCC who received weight-based intravenous cemiplimab for up to 96 weeks, with optional retreatment for those who had disease progression during the follow-up period. This was an older population, with a mean age of 72 years, and more than 80% were male. About one-third had prior systemic therapy, and the majority had prior cancer-related radiotherapy and surgery.
The objective response rate was 43.6% in the locally advanced group and 49.2% in the metastatic group; this numerical difference of less than 3 percentage points was not statistically significant, according to Dr. Migden.
More importantly, he said, the disease control rate (responses, stable disease, and noncomplete response/nonprogressive disease) was 79.5% in the locally advanced group and 71.2% in the metastatic group.
Time to response was “quite rapid” at a median of 1.9 months in both groups, though 7 of the 34 responders in the locally advanced group had unconventional late responses, taking 6-10 months to get to the point of response, Dr. Migden said.
The probability of being event free (such as no progression or death) has remained relatively flat, he added. In the metastatic cohort, event-free probability was 96.4% at 6 months, 88.9% at 12 months, and 82.5% at 20 months, with a 16.5-month median duration of follow-up, while in the locally advanced cohort, the event-free probability was 96.2% at 6 months, 87.8% at both 12 months and 20 months, with a median follow-up of 9.3 months.
Serious treatment-emergent adverse events were reported for 28.5% in these patients, though Dr. Migden noted that treatment emergent does not necessarily mean related to the study drug. Immune-related adverse events of grade 3 or greater were seen in 11.7% of patients, and adverse events leading to discontinuation were reported for 8.8%.
Dr. Migden reported disclosures related to Regeneron, Novartis, Genentech, Eli Lilly, and Sun Pharmaceutical.
MILAN – In an updated analysis of a pivotal phase 2 study, Michael R. Migden, MD, said at the World Congress of Dermatology.
Median duration of response was not reached at the time of the analysis, with probability of no progression or death above 80% at the 20-month mark, according to Dr. Migden, of the department of dermatology at the University of Texas MD Anderson Cancer Center, Houston.
The safety profile of cemiplimab in this study was comparable with what has been reported for other anti–programmed death agents, he said in an oral presentation at the meeting.
While the median time to response was less than 2 months, about one-fifth of patients with locally advanced disease had “unconventional” late responses, occurring up to 10 months after starting treatment, Dr. Migden said. “If you’re just putting someone on some agent like this for a few months, and say, ‘well, I don’t see anything improving,’ it could be one of these patients in this 20% that deserve a little bit longer therapy.”
Cemiplimab (Libtayo) is the only Food and Drug Administration–approved treatment for patients with locally advanced or metastatic cutaneous squamous cell carcinoma (CSCC) who are not suitable for curative surgery or radiation, Dr. Migden said in his presentation. That approval was based in part on previously reported results from the phase 2 study, known as EMPOWER-CSCC-1, which demonstrated that cemiplimab had substantial antitumor activity and durable responses.
In this update on EMPOWER-CSCC-1, Dr. Migden described results for 78 patients with locally advanced CSCC and 59 with metastatic CSCC who received weight-based intravenous cemiplimab for up to 96 weeks, with optional retreatment for those who had disease progression during the follow-up period. This was an older population, with a mean age of 72 years, and more than 80% were male. About one-third had prior systemic therapy, and the majority had prior cancer-related radiotherapy and surgery.
The objective response rate was 43.6% in the locally advanced group and 49.2% in the metastatic group; this numerical difference of less than 3 percentage points was not statistically significant, according to Dr. Migden.
More importantly, he said, the disease control rate (responses, stable disease, and noncomplete response/nonprogressive disease) was 79.5% in the locally advanced group and 71.2% in the metastatic group.
Time to response was “quite rapid” at a median of 1.9 months in both groups, though 7 of the 34 responders in the locally advanced group had unconventional late responses, taking 6-10 months to get to the point of response, Dr. Migden said.
The probability of being event free (such as no progression or death) has remained relatively flat, he added. In the metastatic cohort, event-free probability was 96.4% at 6 months, 88.9% at 12 months, and 82.5% at 20 months, with a 16.5-month median duration of follow-up, while in the locally advanced cohort, the event-free probability was 96.2% at 6 months, 87.8% at both 12 months and 20 months, with a median follow-up of 9.3 months.
Serious treatment-emergent adverse events were reported for 28.5% in these patients, though Dr. Migden noted that treatment emergent does not necessarily mean related to the study drug. Immune-related adverse events of grade 3 or greater were seen in 11.7% of patients, and adverse events leading to discontinuation were reported for 8.8%.
Dr. Migden reported disclosures related to Regeneron, Novartis, Genentech, Eli Lilly, and Sun Pharmaceutical.
MILAN – In an updated analysis of a pivotal phase 2 study, Michael R. Migden, MD, said at the World Congress of Dermatology.
Median duration of response was not reached at the time of the analysis, with probability of no progression or death above 80% at the 20-month mark, according to Dr. Migden, of the department of dermatology at the University of Texas MD Anderson Cancer Center, Houston.
The safety profile of cemiplimab in this study was comparable with what has been reported for other anti–programmed death agents, he said in an oral presentation at the meeting.
While the median time to response was less than 2 months, about one-fifth of patients with locally advanced disease had “unconventional” late responses, occurring up to 10 months after starting treatment, Dr. Migden said. “If you’re just putting someone on some agent like this for a few months, and say, ‘well, I don’t see anything improving,’ it could be one of these patients in this 20% that deserve a little bit longer therapy.”
Cemiplimab (Libtayo) is the only Food and Drug Administration–approved treatment for patients with locally advanced or metastatic cutaneous squamous cell carcinoma (CSCC) who are not suitable for curative surgery or radiation, Dr. Migden said in his presentation. That approval was based in part on previously reported results from the phase 2 study, known as EMPOWER-CSCC-1, which demonstrated that cemiplimab had substantial antitumor activity and durable responses.
In this update on EMPOWER-CSCC-1, Dr. Migden described results for 78 patients with locally advanced CSCC and 59 with metastatic CSCC who received weight-based intravenous cemiplimab for up to 96 weeks, with optional retreatment for those who had disease progression during the follow-up period. This was an older population, with a mean age of 72 years, and more than 80% were male. About one-third had prior systemic therapy, and the majority had prior cancer-related radiotherapy and surgery.
The objective response rate was 43.6% in the locally advanced group and 49.2% in the metastatic group; this numerical difference of less than 3 percentage points was not statistically significant, according to Dr. Migden.
More importantly, he said, the disease control rate (responses, stable disease, and noncomplete response/nonprogressive disease) was 79.5% in the locally advanced group and 71.2% in the metastatic group.
Time to response was “quite rapid” at a median of 1.9 months in both groups, though 7 of the 34 responders in the locally advanced group had unconventional late responses, taking 6-10 months to get to the point of response, Dr. Migden said.
The probability of being event free (such as no progression or death) has remained relatively flat, he added. In the metastatic cohort, event-free probability was 96.4% at 6 months, 88.9% at 12 months, and 82.5% at 20 months, with a 16.5-month median duration of follow-up, while in the locally advanced cohort, the event-free probability was 96.2% at 6 months, 87.8% at both 12 months and 20 months, with a median follow-up of 9.3 months.
Serious treatment-emergent adverse events were reported for 28.5% in these patients, though Dr. Migden noted that treatment emergent does not necessarily mean related to the study drug. Immune-related adverse events of grade 3 or greater were seen in 11.7% of patients, and adverse events leading to discontinuation were reported for 8.8%.
Dr. Migden reported disclosures related to Regeneron, Novartis, Genentech, Eli Lilly, and Sun Pharmaceutical.
REPORTING FROM WCD2019
Advanced basal cell carcinoma responds to pembrolizumab in proof-of-concept study
MILAN – A checkpoint inhibitor given with or without targeted therapy resulted in robust response rates in patients with advanced basal cell carcinoma, according to an investigator in a recent proof-of-concept study.
The side effect profile for the progressive death 1 (PD-1) inhibitor pembrolizumab (Keytruda), plus or minus the hedgehog pathway inhibitor vismodegib (Erivedge), was “not out of range” with what’s been observed in other cancer types, said Anne Lynn S. Chang, MD, a medical dermatologist at Stanford (Calif.) University.
Taken together, these safety and efficacy results suggest pembrolizumab could prove useful for patients with advanced basal cell carcinomas, Dr. Chang said in an oral presentation at the World Congress of Dermatology.
Unexpectedly, dual therapy with pembrolizumab and vismodegib was not clearly superior to pembrolizumab alone, though the two arms are not directly comparable in this nonrandomized, investigator-initiated study, according to Dr. Chang.
“That was a surprise for us, but this is a subjective and a small study,” she told attendees at the conference.
There is currently a multi-institutional clinical trial underway to evaluate another PD-1 inhibitor, cemiplimab, in patients with advanced basal cell carcinoma previously treated with a hedgehog pathway inhibitor, she said.
While a large fraction of basal cell carcinomas are cured by resection, those that are locally advanced or metastatic to lymph nodes or distant organs are challenging to manage. Hedgehog pathway inhibitors, which include vismodegib and sonidegib, are the only Food and Drug Administration–approved drug class in this setting, but more than half of patients do not have a response to treatment, and another 20% or so will initially respond but develop resistance, Dr. Chang said.
Other treatments that have been tried in advanced basal cell carcinomas include taxanes, platinum-based agents, 5-fluorouracil, and everolimus, but their efficacy is unclear, according to Dr. Chang, because of a lack of systematic studies.
Investigators thought PD-1 inhibitors might be promising in basal cell carcinoma for a few reasons, Dr. Chang said. In particular, PD-1 inhibitor treatment response has been linked to mutational burden and PD–ligand 1 expression, and advanced basal cell carcinomas have the largest mutational burden of all human cancers, a large proportion of which express PD-L1.
There are multiple case reports in the literature suggesting that advanced basal cell carcinoma is responsive to PD-1 inhibitors, she added.
In the study, nine patients received 200 mg infusion of pembrolizumab every 3 weeks, and seven received the pembrolizumab infusions plus oral vismodegib 150 mg per day.
The pembrolizumab monotherapy group included patients who had tumor progression on vismodegib, did not tolerate vismodegib, or had contraindications to vismodegib. The pembrolizumab-vismodegib regimen was used in patients who had prior stable or partial responses to hedgehog pathway inhibitors.
The overall response rate was 38%, or 6 of 16 patients, with a 70% probability of 1-year progression-free survival and 94% probability of 1-year overall survival, Dr. Chang reported.
The results did not make the case that pembrolizumab and vismodegib was superior to pembrolizumab alone, though as Dr. Chang noted, the numbers of patients were small. Response rates were 29% (two of seven patients) in the combination arm and 44% (four of nine patients) in the monotherapy arm.
There were 24 immune-related adverse events in the trial; the most serious was hyponatremia, which was reversible, according to Dr. Chang. The overall rates of grade 3-4 adverse events were 22% for pembrolizumab monotherapy and 17% for dual therapy.
A report on the study has also been published in the Journal of the American Academy of Dermatology.
Funding for the study was provided by Merck. Dr. Chang reported serving as a clinical investigator and advisory board member for studies sponsored by Genentech-Roche, Merck, Novartis, and Regeneron.
MILAN – A checkpoint inhibitor given with or without targeted therapy resulted in robust response rates in patients with advanced basal cell carcinoma, according to an investigator in a recent proof-of-concept study.
The side effect profile for the progressive death 1 (PD-1) inhibitor pembrolizumab (Keytruda), plus or minus the hedgehog pathway inhibitor vismodegib (Erivedge), was “not out of range” with what’s been observed in other cancer types, said Anne Lynn S. Chang, MD, a medical dermatologist at Stanford (Calif.) University.
Taken together, these safety and efficacy results suggest pembrolizumab could prove useful for patients with advanced basal cell carcinomas, Dr. Chang said in an oral presentation at the World Congress of Dermatology.
Unexpectedly, dual therapy with pembrolizumab and vismodegib was not clearly superior to pembrolizumab alone, though the two arms are not directly comparable in this nonrandomized, investigator-initiated study, according to Dr. Chang.
“That was a surprise for us, but this is a subjective and a small study,” she told attendees at the conference.
There is currently a multi-institutional clinical trial underway to evaluate another PD-1 inhibitor, cemiplimab, in patients with advanced basal cell carcinoma previously treated with a hedgehog pathway inhibitor, she said.
While a large fraction of basal cell carcinomas are cured by resection, those that are locally advanced or metastatic to lymph nodes or distant organs are challenging to manage. Hedgehog pathway inhibitors, which include vismodegib and sonidegib, are the only Food and Drug Administration–approved drug class in this setting, but more than half of patients do not have a response to treatment, and another 20% or so will initially respond but develop resistance, Dr. Chang said.
Other treatments that have been tried in advanced basal cell carcinomas include taxanes, platinum-based agents, 5-fluorouracil, and everolimus, but their efficacy is unclear, according to Dr. Chang, because of a lack of systematic studies.
Investigators thought PD-1 inhibitors might be promising in basal cell carcinoma for a few reasons, Dr. Chang said. In particular, PD-1 inhibitor treatment response has been linked to mutational burden and PD–ligand 1 expression, and advanced basal cell carcinomas have the largest mutational burden of all human cancers, a large proportion of which express PD-L1.
There are multiple case reports in the literature suggesting that advanced basal cell carcinoma is responsive to PD-1 inhibitors, she added.
In the study, nine patients received 200 mg infusion of pembrolizumab every 3 weeks, and seven received the pembrolizumab infusions plus oral vismodegib 150 mg per day.
The pembrolizumab monotherapy group included patients who had tumor progression on vismodegib, did not tolerate vismodegib, or had contraindications to vismodegib. The pembrolizumab-vismodegib regimen was used in patients who had prior stable or partial responses to hedgehog pathway inhibitors.
The overall response rate was 38%, or 6 of 16 patients, with a 70% probability of 1-year progression-free survival and 94% probability of 1-year overall survival, Dr. Chang reported.
The results did not make the case that pembrolizumab and vismodegib was superior to pembrolizumab alone, though as Dr. Chang noted, the numbers of patients were small. Response rates were 29% (two of seven patients) in the combination arm and 44% (four of nine patients) in the monotherapy arm.
There were 24 immune-related adverse events in the trial; the most serious was hyponatremia, which was reversible, according to Dr. Chang. The overall rates of grade 3-4 adverse events were 22% for pembrolizumab monotherapy and 17% for dual therapy.
A report on the study has also been published in the Journal of the American Academy of Dermatology.
Funding for the study was provided by Merck. Dr. Chang reported serving as a clinical investigator and advisory board member for studies sponsored by Genentech-Roche, Merck, Novartis, and Regeneron.
MILAN – A checkpoint inhibitor given with or without targeted therapy resulted in robust response rates in patients with advanced basal cell carcinoma, according to an investigator in a recent proof-of-concept study.
The side effect profile for the progressive death 1 (PD-1) inhibitor pembrolizumab (Keytruda), plus or minus the hedgehog pathway inhibitor vismodegib (Erivedge), was “not out of range” with what’s been observed in other cancer types, said Anne Lynn S. Chang, MD, a medical dermatologist at Stanford (Calif.) University.
Taken together, these safety and efficacy results suggest pembrolizumab could prove useful for patients with advanced basal cell carcinomas, Dr. Chang said in an oral presentation at the World Congress of Dermatology.
Unexpectedly, dual therapy with pembrolizumab and vismodegib was not clearly superior to pembrolizumab alone, though the two arms are not directly comparable in this nonrandomized, investigator-initiated study, according to Dr. Chang.
“That was a surprise for us, but this is a subjective and a small study,” she told attendees at the conference.
There is currently a multi-institutional clinical trial underway to evaluate another PD-1 inhibitor, cemiplimab, in patients with advanced basal cell carcinoma previously treated with a hedgehog pathway inhibitor, she said.
While a large fraction of basal cell carcinomas are cured by resection, those that are locally advanced or metastatic to lymph nodes or distant organs are challenging to manage. Hedgehog pathway inhibitors, which include vismodegib and sonidegib, are the only Food and Drug Administration–approved drug class in this setting, but more than half of patients do not have a response to treatment, and another 20% or so will initially respond but develop resistance, Dr. Chang said.
Other treatments that have been tried in advanced basal cell carcinomas include taxanes, platinum-based agents, 5-fluorouracil, and everolimus, but their efficacy is unclear, according to Dr. Chang, because of a lack of systematic studies.
Investigators thought PD-1 inhibitors might be promising in basal cell carcinoma for a few reasons, Dr. Chang said. In particular, PD-1 inhibitor treatment response has been linked to mutational burden and PD–ligand 1 expression, and advanced basal cell carcinomas have the largest mutational burden of all human cancers, a large proportion of which express PD-L1.
There are multiple case reports in the literature suggesting that advanced basal cell carcinoma is responsive to PD-1 inhibitors, she added.
In the study, nine patients received 200 mg infusion of pembrolizumab every 3 weeks, and seven received the pembrolizumab infusions plus oral vismodegib 150 mg per day.
The pembrolizumab monotherapy group included patients who had tumor progression on vismodegib, did not tolerate vismodegib, or had contraindications to vismodegib. The pembrolizumab-vismodegib regimen was used in patients who had prior stable or partial responses to hedgehog pathway inhibitors.
The overall response rate was 38%, or 6 of 16 patients, with a 70% probability of 1-year progression-free survival and 94% probability of 1-year overall survival, Dr. Chang reported.
The results did not make the case that pembrolizumab and vismodegib was superior to pembrolizumab alone, though as Dr. Chang noted, the numbers of patients were small. Response rates were 29% (two of seven patients) in the combination arm and 44% (four of nine patients) in the monotherapy arm.
There were 24 immune-related adverse events in the trial; the most serious was hyponatremia, which was reversible, according to Dr. Chang. The overall rates of grade 3-4 adverse events were 22% for pembrolizumab monotherapy and 17% for dual therapy.
A report on the study has also been published in the Journal of the American Academy of Dermatology.
Funding for the study was provided by Merck. Dr. Chang reported serving as a clinical investigator and advisory board member for studies sponsored by Genentech-Roche, Merck, Novartis, and Regeneron.
REPORTING FROM WCD2019