User login
Massive liver metastasis from colon adenocarcinoma causing cardiac tamponade
Colorectal cancer is the third most commonly diagnosed cancer in the United States.1 About 5% of Americans will be diagnosed with colorectal cancer in their lifetime, of which 20% will present with distant metastasis.2 The most common sites of metastasis are regional lymph nodes, liver, lung and peritoneum, and patients may present with signs or symptoms related to disease burden at any of these organs.
Case presentation and summary
A 55-year-old man had presented to an outside hospital in August of 2014 with 6 months of hematochezia and a 40-lb weight loss. He was found to be severely anemic on admission (hemoglobin, 4.9 g/dL [normal, 13-17 g/dL], hematocrit, 16% [normal, 35%-45%]). A computed-tomography (CT) scan of the abdomen and pelvis with contrast revealed a mass of 6.9 x 4.7 x 6.3 cm in the rectosigmoid colon and a mass of 10.0 x 12.0 x 10.7 cm in the right hepatic lobe consistent with metastatic disease. The patient was taken to the operating room where the rectosigmoid mass was resected completely. The liver mass was deemed unresectable because of its large size, and surgically directed therapy could not be performed. Pathology was consistent with a T3N1 moderately differentiated adenocarcinoma 11 cm from the anal verge. Further molecular tumor studies revealed wild type KRAS and NRAS, as well as a BRAF mutation.
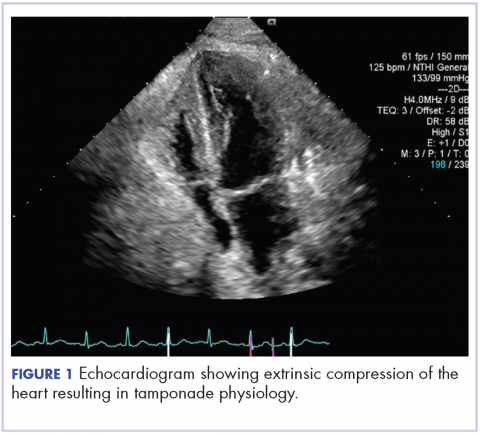
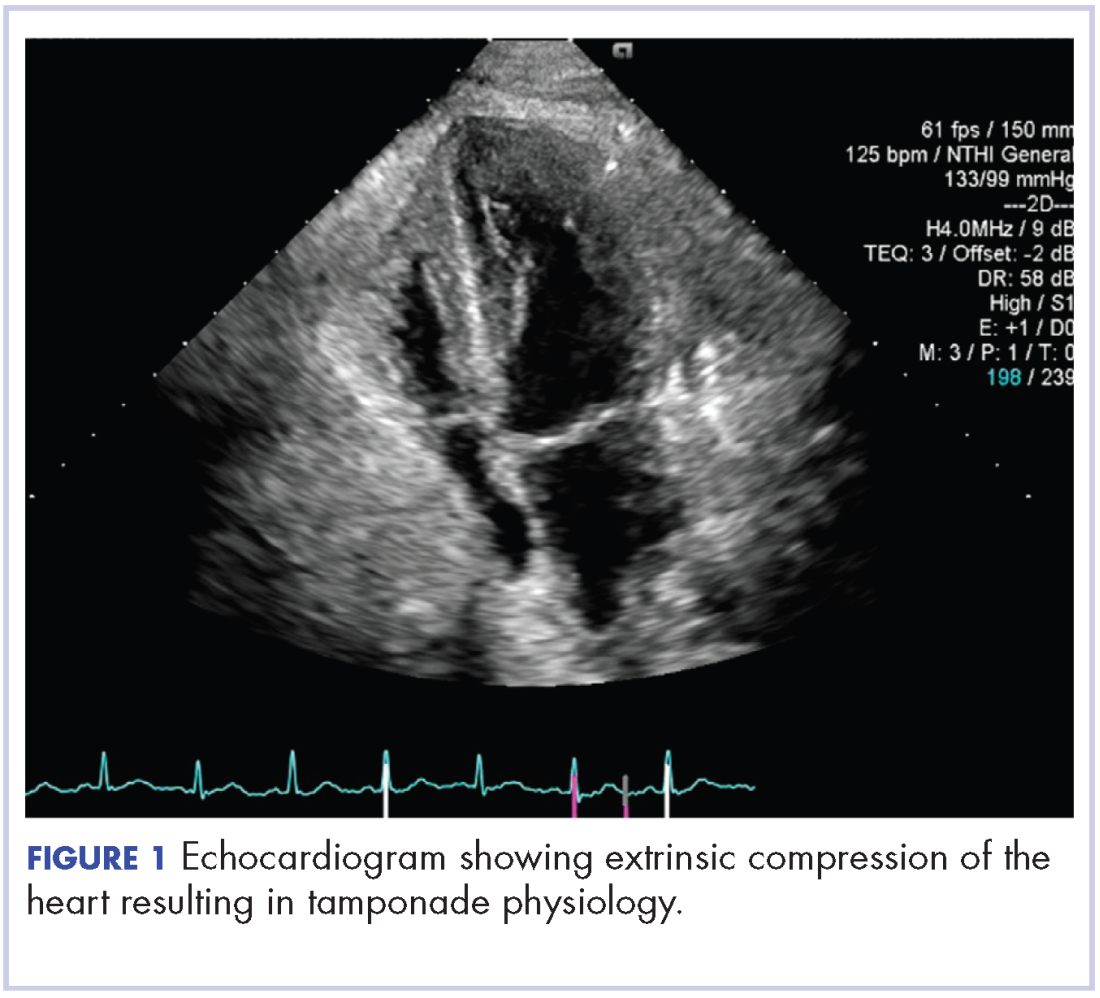
About 4 weeks after the surgery, the patient was seen at our institution for an initial consultation and was noted to have significant anasarca, including 4+ pitting lower extremity edema and scrotal edema. He complained of dyspnea on exertion, which he attributed to deconditioning. His resting heart rate was found to be 123 beats per minute (normal, 60-100 bpm). Jugular venous distention was present. The patient was sent for an urgent echocardiogram, which showed external compression of the right atrium and ventricle by his liver metastasis resulting in tamponade physiology without the presence of any pericardial effusion (Figure 1).
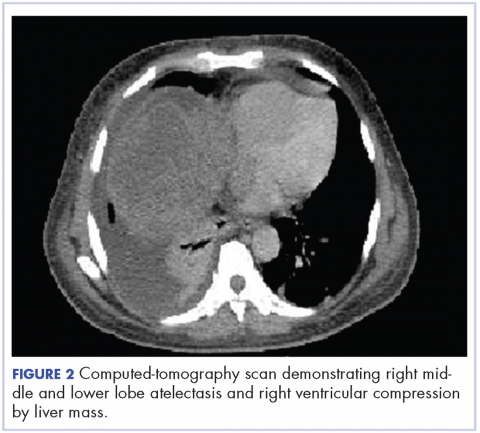
A CT of the abdomen and pelvis at that time showed that the liver mass had increased to 17.6 x 12.1 x 16.1 cm, exerting pressure on the heart and causing atelectasis of the right middle and lower lung lobes (Figure 2).
Treatment plan
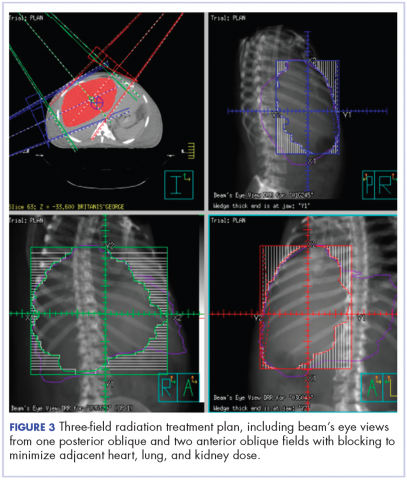
The patient was evaluated by surgical oncology for resection, but his cardiovascular status placed him at high risk for perioperative complications, so such surgery was not pursued. Radioembolization was considered but not pursued because the process needed to evaluate, plan, and treat was not considered sufficiently timely. We consulted with our radiation oncology colleagues about external beam radiotherapy (EBRT) for rapid palliation. They evaluated the patient and recommended the EBRT, and the patient signed consent for treatment. We performed a CT-based simulation and generated an external beam, linear-accelerator–based treatment plan. The plan consisted of three 15-megavoltage photon fields delivering 3,000 cGy in 10 fractions to the whole liver, with appropriate multileaf collimation blocking to minimize dose to adjacent heart, right lung, and bilateral kidneys (Figure 3).
Before initiation of the EBRT, the patient received systemic chemotherapy with a dose-adjusted FOLFOX regimen (5-FU bolus 200 mg/m2, leucovorin 200 mg/m2, oxaliplatin 85 mg/m2, with infusional 5-FU 2,400 mg/m2 over 46 hours). After completing 1 dose of modified FOLFOX, he completed 10 fractions of whole liver radiotherapy with the aforementioned plan. He tolerated the initial treatment well and his subjective symptoms improved. The patient then proceeded to further systemic therapy. After recent data demonstrated improved median progression-free survival and response rates with FOLFOXIRI plus bevacizumab (infusional 5-FU 3200 mg/m2, leucovorin 200 mg/m2, irinotecan 165 mg/m2, and oxaliplatin 85 mg/m2, bevacizumab 5 mg/kg) versus FOLFIRI plus bevacizumab,3 we decided to modify his systemic therapy to FOLFOXIRI with bevacizumab to induce a better response.
Treatment response
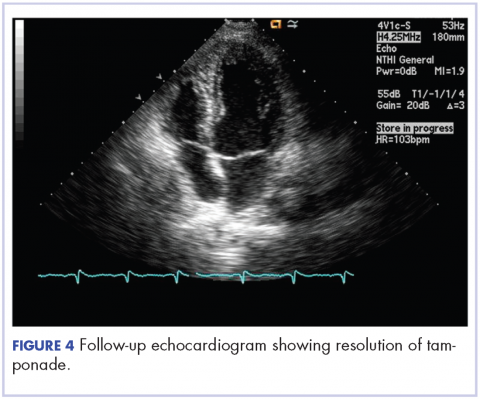
After 2 doses of chemotherapy and completion of radiotherapy, the edema and shortness of breath improved. A follow-up echocardiogram performed a month after completion of EBRT, 1 dose of FOLFOX, and 1 dose of FOLFOXIRI showed resolution of the cardiac compression (Figure 4).
A CT scan of the abdomen and pelvis obtained after 3 cycles of FOLFOXIRI showed marked decrease in the size of the right lobe hepatic mass from 17.6 x 12.1 cm to 12.0 x 8.0 cm. Given the survival benefit of VEGF inhibition in colon cancer, bevacizumab (5 mg/kg) was added to the FOLFOXIRI regimen with cycle 4. Unfortunately, after the 5th cycle, a CT scan of the abdomen showed an increase in size of the hepatic lesions. At this time, FOLFOXIRI and bevacizumab were stopped, and given the tumor’s KRAS/NRAS wild type status, systemic therapy was changed to panitumumab (6 mg/kg). The patient initially tolerated treatment well, but after 9 cycles, the total bilirubin started to increase. CT abdomen at this point was consistent with progression of disease. The patient was not eligible for a clinical trial targeting BRAF mutation given the elevated bilirubin. Regorafanib (80 mg daily for 3 weeks on and 1 week off) was started. After the first cycle, the total bilirubin increased further and the regorafanib was dose reduced to 40 mg daily. Unfortunately, a repeat CT scan of the abdomen demonstrated progression of disease, and given that he developed a progressive transaminitis and hyperbilirubinemia, hospice care was recommended. The patient died shortly thereafter, about 15 months after his initial diagnosis.
Discussion
Massive liver metastasis in the setting of disseminated cancer is not an uncommon manifestation of advanced cancer that can have life-threatening consequences. In te present case, a bulky liver metastasis caused extrinsic compression of the right atrium, resulting in obvious clinical and echocardiogram-proven cardiac tamponade physiology. To our knowledge, this is the first reported case of the treatment of a bulky hepatic metastasis causing cardiac tamponade. In this patient’s case, both radiotherapy and chemotherapy were given safely in rapid sequence resulting in quick resolution of the patient’s symptoms and echocardiogram findings. The presence of a BRAF mutation conferred a poor prognosis and poor response to systemic chemotherapy. Nevertheless, the patient showed good response to a FOLFOXIRI regimen, chosen in this emergent situation given its significantly higher response rates compared with the standard FOLFIRI regimen, which was tolerated well with minimal adverse effects.
Findings from randomized controlled trials examining the role of palliative radiotherapy for metastatic liver disease have suggested that dose escalation above 30 Gy to the whole liver may lead to unacceptably high rates of radiation-induced liver disease, which typically leads to mortality.4-8 Two prospective trials comparing twice daily with daily fractionation have shown no benefit to hyperfractionation, with possibly increased rates of acute toxicity in the setting of hepatocellular carcinoma.9,10 There is emerging evidence that partial liver irradiation, in the appropriate setting in the form of boost after whole-liver RT or stereotactic body radiotherapy, may allow for further dose escalation while avoiding clinical hepatitis.11 Although there is no clear consensus about optimal RT dose and fractionation, the aforementioned studies show that dose and fractionation schemes ranging between 21 Gy and 30 Gy in 1.5 Gy to 3 Gy daily fractions likely provide the best therapeutic ratio for whole-liver irradiation.
In conclusion, this case demonstrates the resolution of cardiac tamponade from a massive liver colorectal metastasis after chemoradiation and illustrates the potential utility of adding radiotherapy to chemotherapy in an urgent scenario where the former might not typically be considered.
1. American Cancer Society. Cancer Facts & Figures 2015. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2015.html. Published 2015. Accessed October 10, 2017.
2. Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin. 2014;64(2):104-117.
3. Loupakis F, Cremolini C, Masi G, et al. Initial therapy with FOLFOXIRI and bevacizumab for metastatic colorectal cancer. N Engl J Med. 2014;371:1609-1618.
4. Russell AH, Clyde C, Wasserman TH, Turner SS, Rotman M. Accelerated hyperfractionated hepatic irradiation in the management of patients with liver metastases: results of the RTOG dose escalating protocol. Int J Radiat Oncol Biol Phys. 1993;27(1):117-123.
5. Turek-Maischeider M, Kazem I. Palliative irradiation for liver metastases. JAMA. 1975;232(6):625-628.
6. Sherman DM, Weichselbaum R, Order SE, Cloud L, Trey C, Piro AJ. Palliation of hepatic metastasis. Cancer. 1978;41(5):2013-2017.
7. Prasad B, Lee MS, Hendrickson FR. Irradiation of hepatic metastases. Int J Radiat Oncol Biol Phys. 1977;2:129-132.
8. Borgelt BB, Gelber R, Brady LW, Griffin T, Hendrickson FR. The palliation of hepatic metastases: results of the Radiation Therapy Oncology Group pilot study. Int J Radiat Oncol Biol Phys. 1981;7(5):587-591.
9. Raju PI, Maruyama Y, DeSimone P, MacDonald J. Treatment of liver metastases with a combination of chemotherapy and hyperfractionated external radiation therapy. Am J Clin Oncol. 1987;10(1):41-43.
10. Stillwagon GB, Order SE, Guse C, et al. 194 hepatocellular cancers treated by radiation and chemotherapy combinations: toxicity and response: a Radiation Therapy Oncology Group Study. Int J Radiat Oncol Biol Phys. 1989;17(6):1223-1229.
11. Mohiuddin M, Chen E, Ahmad N. Combined liver radiation and chemotherapy for palliation of hepatic metastases from colorectal cancer. J Clin Oncol. 1996;14(3):722-728.
Colorectal cancer is the third most commonly diagnosed cancer in the United States.1 About 5% of Americans will be diagnosed with colorectal cancer in their lifetime, of which 20% will present with distant metastasis.2 The most common sites of metastasis are regional lymph nodes, liver, lung and peritoneum, and patients may present with signs or symptoms related to disease burden at any of these organs.
Case presentation and summary
A 55-year-old man had presented to an outside hospital in August of 2014 with 6 months of hematochezia and a 40-lb weight loss. He was found to be severely anemic on admission (hemoglobin, 4.9 g/dL [normal, 13-17 g/dL], hematocrit, 16% [normal, 35%-45%]). A computed-tomography (CT) scan of the abdomen and pelvis with contrast revealed a mass of 6.9 x 4.7 x 6.3 cm in the rectosigmoid colon and a mass of 10.0 x 12.0 x 10.7 cm in the right hepatic lobe consistent with metastatic disease. The patient was taken to the operating room where the rectosigmoid mass was resected completely. The liver mass was deemed unresectable because of its large size, and surgically directed therapy could not be performed. Pathology was consistent with a T3N1 moderately differentiated adenocarcinoma 11 cm from the anal verge. Further molecular tumor studies revealed wild type KRAS and NRAS, as well as a BRAF mutation.
About 4 weeks after the surgery, the patient was seen at our institution for an initial consultation and was noted to have significant anasarca, including 4+ pitting lower extremity edema and scrotal edema. He complained of dyspnea on exertion, which he attributed to deconditioning. His resting heart rate was found to be 123 beats per minute (normal, 60-100 bpm). Jugular venous distention was present. The patient was sent for an urgent echocardiogram, which showed external compression of the right atrium and ventricle by his liver metastasis resulting in tamponade physiology without the presence of any pericardial effusion (Figure 1).
A CT of the abdomen and pelvis at that time showed that the liver mass had increased to 17.6 x 12.1 x 16.1 cm, exerting pressure on the heart and causing atelectasis of the right middle and lower lung lobes (Figure 2).
Treatment plan
The patient was evaluated by surgical oncology for resection, but his cardiovascular status placed him at high risk for perioperative complications, so such surgery was not pursued. Radioembolization was considered but not pursued because the process needed to evaluate, plan, and treat was not considered sufficiently timely. We consulted with our radiation oncology colleagues about external beam radiotherapy (EBRT) for rapid palliation. They evaluated the patient and recommended the EBRT, and the patient signed consent for treatment. We performed a CT-based simulation and generated an external beam, linear-accelerator–based treatment plan. The plan consisted of three 15-megavoltage photon fields delivering 3,000 cGy in 10 fractions to the whole liver, with appropriate multileaf collimation blocking to minimize dose to adjacent heart, right lung, and bilateral kidneys (Figure 3).
Before initiation of the EBRT, the patient received systemic chemotherapy with a dose-adjusted FOLFOX regimen (5-FU bolus 200 mg/m2, leucovorin 200 mg/m2, oxaliplatin 85 mg/m2, with infusional 5-FU 2,400 mg/m2 over 46 hours). After completing 1 dose of modified FOLFOX, he completed 10 fractions of whole liver radiotherapy with the aforementioned plan. He tolerated the initial treatment well and his subjective symptoms improved. The patient then proceeded to further systemic therapy. After recent data demonstrated improved median progression-free survival and response rates with FOLFOXIRI plus bevacizumab (infusional 5-FU 3200 mg/m2, leucovorin 200 mg/m2, irinotecan 165 mg/m2, and oxaliplatin 85 mg/m2, bevacizumab 5 mg/kg) versus FOLFIRI plus bevacizumab,3 we decided to modify his systemic therapy to FOLFOXIRI with bevacizumab to induce a better response.
Treatment response
After 2 doses of chemotherapy and completion of radiotherapy, the edema and shortness of breath improved. A follow-up echocardiogram performed a month after completion of EBRT, 1 dose of FOLFOX, and 1 dose of FOLFOXIRI showed resolution of the cardiac compression (Figure 4).
A CT scan of the abdomen and pelvis obtained after 3 cycles of FOLFOXIRI showed marked decrease in the size of the right lobe hepatic mass from 17.6 x 12.1 cm to 12.0 x 8.0 cm. Given the survival benefit of VEGF inhibition in colon cancer, bevacizumab (5 mg/kg) was added to the FOLFOXIRI regimen with cycle 4. Unfortunately, after the 5th cycle, a CT scan of the abdomen showed an increase in size of the hepatic lesions. At this time, FOLFOXIRI and bevacizumab were stopped, and given the tumor’s KRAS/NRAS wild type status, systemic therapy was changed to panitumumab (6 mg/kg). The patient initially tolerated treatment well, but after 9 cycles, the total bilirubin started to increase. CT abdomen at this point was consistent with progression of disease. The patient was not eligible for a clinical trial targeting BRAF mutation given the elevated bilirubin. Regorafanib (80 mg daily for 3 weeks on and 1 week off) was started. After the first cycle, the total bilirubin increased further and the regorafanib was dose reduced to 40 mg daily. Unfortunately, a repeat CT scan of the abdomen demonstrated progression of disease, and given that he developed a progressive transaminitis and hyperbilirubinemia, hospice care was recommended. The patient died shortly thereafter, about 15 months after his initial diagnosis.
Discussion
Massive liver metastasis in the setting of disseminated cancer is not an uncommon manifestation of advanced cancer that can have life-threatening consequences. In te present case, a bulky liver metastasis caused extrinsic compression of the right atrium, resulting in obvious clinical and echocardiogram-proven cardiac tamponade physiology. To our knowledge, this is the first reported case of the treatment of a bulky hepatic metastasis causing cardiac tamponade. In this patient’s case, both radiotherapy and chemotherapy were given safely in rapid sequence resulting in quick resolution of the patient’s symptoms and echocardiogram findings. The presence of a BRAF mutation conferred a poor prognosis and poor response to systemic chemotherapy. Nevertheless, the patient showed good response to a FOLFOXIRI regimen, chosen in this emergent situation given its significantly higher response rates compared with the standard FOLFIRI regimen, which was tolerated well with minimal adverse effects.
Findings from randomized controlled trials examining the role of palliative radiotherapy for metastatic liver disease have suggested that dose escalation above 30 Gy to the whole liver may lead to unacceptably high rates of radiation-induced liver disease, which typically leads to mortality.4-8 Two prospective trials comparing twice daily with daily fractionation have shown no benefit to hyperfractionation, with possibly increased rates of acute toxicity in the setting of hepatocellular carcinoma.9,10 There is emerging evidence that partial liver irradiation, in the appropriate setting in the form of boost after whole-liver RT or stereotactic body radiotherapy, may allow for further dose escalation while avoiding clinical hepatitis.11 Although there is no clear consensus about optimal RT dose and fractionation, the aforementioned studies show that dose and fractionation schemes ranging between 21 Gy and 30 Gy in 1.5 Gy to 3 Gy daily fractions likely provide the best therapeutic ratio for whole-liver irradiation.
In conclusion, this case demonstrates the resolution of cardiac tamponade from a massive liver colorectal metastasis after chemoradiation and illustrates the potential utility of adding radiotherapy to chemotherapy in an urgent scenario where the former might not typically be considered.
Colorectal cancer is the third most commonly diagnosed cancer in the United States.1 About 5% of Americans will be diagnosed with colorectal cancer in their lifetime, of which 20% will present with distant metastasis.2 The most common sites of metastasis are regional lymph nodes, liver, lung and peritoneum, and patients may present with signs or symptoms related to disease burden at any of these organs.
Case presentation and summary
A 55-year-old man had presented to an outside hospital in August of 2014 with 6 months of hematochezia and a 40-lb weight loss. He was found to be severely anemic on admission (hemoglobin, 4.9 g/dL [normal, 13-17 g/dL], hematocrit, 16% [normal, 35%-45%]). A computed-tomography (CT) scan of the abdomen and pelvis with contrast revealed a mass of 6.9 x 4.7 x 6.3 cm in the rectosigmoid colon and a mass of 10.0 x 12.0 x 10.7 cm in the right hepatic lobe consistent with metastatic disease. The patient was taken to the operating room where the rectosigmoid mass was resected completely. The liver mass was deemed unresectable because of its large size, and surgically directed therapy could not be performed. Pathology was consistent with a T3N1 moderately differentiated adenocarcinoma 11 cm from the anal verge. Further molecular tumor studies revealed wild type KRAS and NRAS, as well as a BRAF mutation.
About 4 weeks after the surgery, the patient was seen at our institution for an initial consultation and was noted to have significant anasarca, including 4+ pitting lower extremity edema and scrotal edema. He complained of dyspnea on exertion, which he attributed to deconditioning. His resting heart rate was found to be 123 beats per minute (normal, 60-100 bpm). Jugular venous distention was present. The patient was sent for an urgent echocardiogram, which showed external compression of the right atrium and ventricle by his liver metastasis resulting in tamponade physiology without the presence of any pericardial effusion (Figure 1).
A CT of the abdomen and pelvis at that time showed that the liver mass had increased to 17.6 x 12.1 x 16.1 cm, exerting pressure on the heart and causing atelectasis of the right middle and lower lung lobes (Figure 2).
Treatment plan
The patient was evaluated by surgical oncology for resection, but his cardiovascular status placed him at high risk for perioperative complications, so such surgery was not pursued. Radioembolization was considered but not pursued because the process needed to evaluate, plan, and treat was not considered sufficiently timely. We consulted with our radiation oncology colleagues about external beam radiotherapy (EBRT) for rapid palliation. They evaluated the patient and recommended the EBRT, and the patient signed consent for treatment. We performed a CT-based simulation and generated an external beam, linear-accelerator–based treatment plan. The plan consisted of three 15-megavoltage photon fields delivering 3,000 cGy in 10 fractions to the whole liver, with appropriate multileaf collimation blocking to minimize dose to adjacent heart, right lung, and bilateral kidneys (Figure 3).
Before initiation of the EBRT, the patient received systemic chemotherapy with a dose-adjusted FOLFOX regimen (5-FU bolus 200 mg/m2, leucovorin 200 mg/m2, oxaliplatin 85 mg/m2, with infusional 5-FU 2,400 mg/m2 over 46 hours). After completing 1 dose of modified FOLFOX, he completed 10 fractions of whole liver radiotherapy with the aforementioned plan. He tolerated the initial treatment well and his subjective symptoms improved. The patient then proceeded to further systemic therapy. After recent data demonstrated improved median progression-free survival and response rates with FOLFOXIRI plus bevacizumab (infusional 5-FU 3200 mg/m2, leucovorin 200 mg/m2, irinotecan 165 mg/m2, and oxaliplatin 85 mg/m2, bevacizumab 5 mg/kg) versus FOLFIRI plus bevacizumab,3 we decided to modify his systemic therapy to FOLFOXIRI with bevacizumab to induce a better response.
Treatment response
After 2 doses of chemotherapy and completion of radiotherapy, the edema and shortness of breath improved. A follow-up echocardiogram performed a month after completion of EBRT, 1 dose of FOLFOX, and 1 dose of FOLFOXIRI showed resolution of the cardiac compression (Figure 4).
A CT scan of the abdomen and pelvis obtained after 3 cycles of FOLFOXIRI showed marked decrease in the size of the right lobe hepatic mass from 17.6 x 12.1 cm to 12.0 x 8.0 cm. Given the survival benefit of VEGF inhibition in colon cancer, bevacizumab (5 mg/kg) was added to the FOLFOXIRI regimen with cycle 4. Unfortunately, after the 5th cycle, a CT scan of the abdomen showed an increase in size of the hepatic lesions. At this time, FOLFOXIRI and bevacizumab were stopped, and given the tumor’s KRAS/NRAS wild type status, systemic therapy was changed to panitumumab (6 mg/kg). The patient initially tolerated treatment well, but after 9 cycles, the total bilirubin started to increase. CT abdomen at this point was consistent with progression of disease. The patient was not eligible for a clinical trial targeting BRAF mutation given the elevated bilirubin. Regorafanib (80 mg daily for 3 weeks on and 1 week off) was started. After the first cycle, the total bilirubin increased further and the regorafanib was dose reduced to 40 mg daily. Unfortunately, a repeat CT scan of the abdomen demonstrated progression of disease, and given that he developed a progressive transaminitis and hyperbilirubinemia, hospice care was recommended. The patient died shortly thereafter, about 15 months after his initial diagnosis.
Discussion
Massive liver metastasis in the setting of disseminated cancer is not an uncommon manifestation of advanced cancer that can have life-threatening consequences. In te present case, a bulky liver metastasis caused extrinsic compression of the right atrium, resulting in obvious clinical and echocardiogram-proven cardiac tamponade physiology. To our knowledge, this is the first reported case of the treatment of a bulky hepatic metastasis causing cardiac tamponade. In this patient’s case, both radiotherapy and chemotherapy were given safely in rapid sequence resulting in quick resolution of the patient’s symptoms and echocardiogram findings. The presence of a BRAF mutation conferred a poor prognosis and poor response to systemic chemotherapy. Nevertheless, the patient showed good response to a FOLFOXIRI regimen, chosen in this emergent situation given its significantly higher response rates compared with the standard FOLFIRI regimen, which was tolerated well with minimal adverse effects.
Findings from randomized controlled trials examining the role of palliative radiotherapy for metastatic liver disease have suggested that dose escalation above 30 Gy to the whole liver may lead to unacceptably high rates of radiation-induced liver disease, which typically leads to mortality.4-8 Two prospective trials comparing twice daily with daily fractionation have shown no benefit to hyperfractionation, with possibly increased rates of acute toxicity in the setting of hepatocellular carcinoma.9,10 There is emerging evidence that partial liver irradiation, in the appropriate setting in the form of boost after whole-liver RT or stereotactic body radiotherapy, may allow for further dose escalation while avoiding clinical hepatitis.11 Although there is no clear consensus about optimal RT dose and fractionation, the aforementioned studies show that dose and fractionation schemes ranging between 21 Gy and 30 Gy in 1.5 Gy to 3 Gy daily fractions likely provide the best therapeutic ratio for whole-liver irradiation.
In conclusion, this case demonstrates the resolution of cardiac tamponade from a massive liver colorectal metastasis after chemoradiation and illustrates the potential utility of adding radiotherapy to chemotherapy in an urgent scenario where the former might not typically be considered.
1. American Cancer Society. Cancer Facts & Figures 2015. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2015.html. Published 2015. Accessed October 10, 2017.
2. Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin. 2014;64(2):104-117.
3. Loupakis F, Cremolini C, Masi G, et al. Initial therapy with FOLFOXIRI and bevacizumab for metastatic colorectal cancer. N Engl J Med. 2014;371:1609-1618.
4. Russell AH, Clyde C, Wasserman TH, Turner SS, Rotman M. Accelerated hyperfractionated hepatic irradiation in the management of patients with liver metastases: results of the RTOG dose escalating protocol. Int J Radiat Oncol Biol Phys. 1993;27(1):117-123.
5. Turek-Maischeider M, Kazem I. Palliative irradiation for liver metastases. JAMA. 1975;232(6):625-628.
6. Sherman DM, Weichselbaum R, Order SE, Cloud L, Trey C, Piro AJ. Palliation of hepatic metastasis. Cancer. 1978;41(5):2013-2017.
7. Prasad B, Lee MS, Hendrickson FR. Irradiation of hepatic metastases. Int J Radiat Oncol Biol Phys. 1977;2:129-132.
8. Borgelt BB, Gelber R, Brady LW, Griffin T, Hendrickson FR. The palliation of hepatic metastases: results of the Radiation Therapy Oncology Group pilot study. Int J Radiat Oncol Biol Phys. 1981;7(5):587-591.
9. Raju PI, Maruyama Y, DeSimone P, MacDonald J. Treatment of liver metastases with a combination of chemotherapy and hyperfractionated external radiation therapy. Am J Clin Oncol. 1987;10(1):41-43.
10. Stillwagon GB, Order SE, Guse C, et al. 194 hepatocellular cancers treated by radiation and chemotherapy combinations: toxicity and response: a Radiation Therapy Oncology Group Study. Int J Radiat Oncol Biol Phys. 1989;17(6):1223-1229.
11. Mohiuddin M, Chen E, Ahmad N. Combined liver radiation and chemotherapy for palliation of hepatic metastases from colorectal cancer. J Clin Oncol. 1996;14(3):722-728.
1. American Cancer Society. Cancer Facts & Figures 2015. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2015.html. Published 2015. Accessed October 10, 2017.
2. Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin. 2014;64(2):104-117.
3. Loupakis F, Cremolini C, Masi G, et al. Initial therapy with FOLFOXIRI and bevacizumab for metastatic colorectal cancer. N Engl J Med. 2014;371:1609-1618.
4. Russell AH, Clyde C, Wasserman TH, Turner SS, Rotman M. Accelerated hyperfractionated hepatic irradiation in the management of patients with liver metastases: results of the RTOG dose escalating protocol. Int J Radiat Oncol Biol Phys. 1993;27(1):117-123.
5. Turek-Maischeider M, Kazem I. Palliative irradiation for liver metastases. JAMA. 1975;232(6):625-628.
6. Sherman DM, Weichselbaum R, Order SE, Cloud L, Trey C, Piro AJ. Palliation of hepatic metastasis. Cancer. 1978;41(5):2013-2017.
7. Prasad B, Lee MS, Hendrickson FR. Irradiation of hepatic metastases. Int J Radiat Oncol Biol Phys. 1977;2:129-132.
8. Borgelt BB, Gelber R, Brady LW, Griffin T, Hendrickson FR. The palliation of hepatic metastases: results of the Radiation Therapy Oncology Group pilot study. Int J Radiat Oncol Biol Phys. 1981;7(5):587-591.
9. Raju PI, Maruyama Y, DeSimone P, MacDonald J. Treatment of liver metastases with a combination of chemotherapy and hyperfractionated external radiation therapy. Am J Clin Oncol. 1987;10(1):41-43.
10. Stillwagon GB, Order SE, Guse C, et al. 194 hepatocellular cancers treated by radiation and chemotherapy combinations: toxicity and response: a Radiation Therapy Oncology Group Study. Int J Radiat Oncol Biol Phys. 1989;17(6):1223-1229.
11. Mohiuddin M, Chen E, Ahmad N. Combined liver radiation and chemotherapy for palliation of hepatic metastases from colorectal cancer. J Clin Oncol. 1996;14(3):722-728.
Cardiac pleomorphic sarcoma after placement of Dacron graft
Primary cardiac tumors, either benign or malignant, are very rare. The combined incidence is 0.002% on pooled autopsy series.1 The benign tumors account for 63% of primary cardiac tumors and include myxoma, the most common, and followed by papillary fibroelastoma, fibroma, and hemangioma. The remaining 37% are malignant tumors, essentially predominated by sarcomas.1
Although myxoma is the most common tumor arising in the left atrium, we present a case that shows that sarcoma can also arise from the same chamber. In fact, sarcomas could mimic cardiac myxoma.2 The cardiac sarcomas can have similar clinical presentation and more importantly can share similar histopathological features. Sarcomas may have myxoid features.2 Cases diagnosed as cardiac myxomas should be diligently worked up to rule out the presence of sarcomas with myxoid features. In addition, foreign bodies have been found to induce sarcomas in experimental animals.3,4 In particular, 2 case reports have described sarcomas arising in association with Dacron vascular prostheses in humans.5,6 We present here the case of a patient who was diagnosed with cardiac pleomorphic sarcoma 8 years after the placement of a Dacron graft.
Case presentation and summary
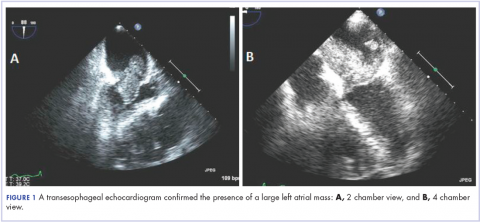
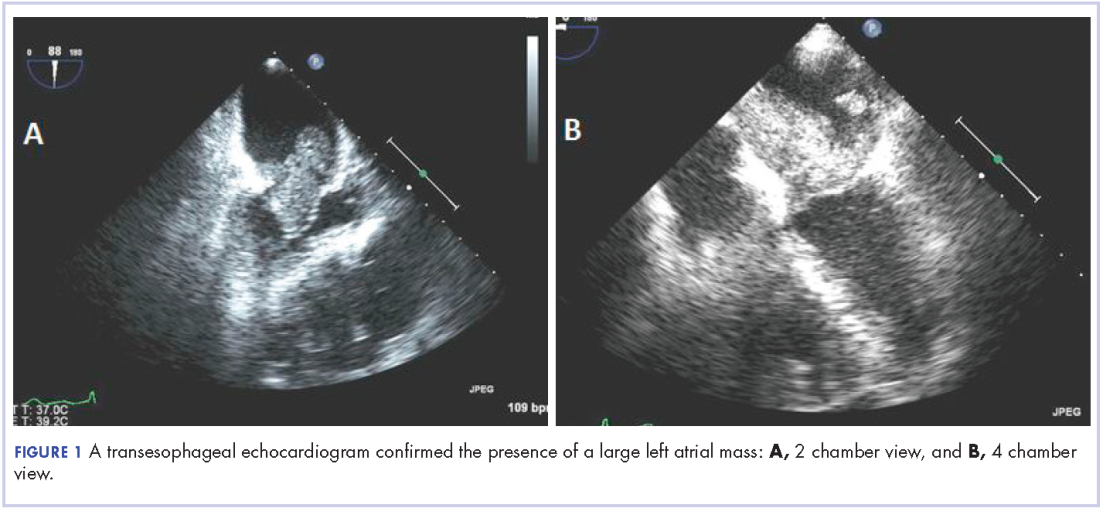
A 56-year-old woman with history of left atrial myxoma status after resection in 2005 and placement of a Dacron graft, morbid obesity, hypertension, and asthma presented to the emergency department with progressively worsening shortness of breath and blurry vision over period of 2 months. Acute coronary syndrome was ruled out by electrocardiogram and serial biomarkers. A computed-tomography angiogram was pursued because of her history of left atrial myxoma, and the results suggested the presence of a left atrial tumor. She underwent a transesophageal echocardiogram, which confirmed the presence of a large left atrial mass that likely was attached to the interatrial septum prolapsing across the mitral valve and was suggestive for recurrent left atrial myxoma (Figure 1). The results of a cardiac catheterization showed normal coronaries.
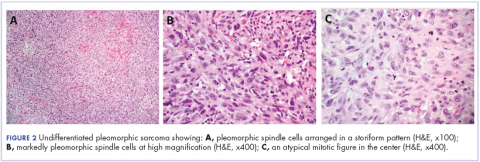
The patient subsequently underwent an excision of the left atrial tumor with profound internal and external myocardial cooling using antegrade blood cardioplegia under mildly hypothermic cardiopulmonary bypass. Frozen sections showed high-grade malignancy in favor of sarcoma. The hematoxylin and eosin stained permanent sections showed sheets of malignant pleomorphic spindle cells focally arranged in a storiform pattern. There were areas of necrosis and abundant mitotic activity. By immunohistochemical (IHC) stains, the tumor cells were diffusely positive for vimentin, and negative for pan-cytokeratin antibody (AE1/AE3), S-100 protein, Melan-A antibody, HMB45, CD34, CD31, myogenin, and MYOD1. IHC stains for CK-OSCAR, desmin, and smooth muscle actin were focally positive, and a ki-67 stain showed a proliferation index of about 80%. The histologic and IHC findings were consistent with a final diagnosis of high-grade undifferentiated pleomorphic sarcoma (Figure 2).
A positron emission tomography scan performed November 2013 did not show any other activity. The patient was scheduled for chemotherapy with adriamycin and ifosfamide with a plan for total of 6 cycles. Before her admission for the chemotherapy, the patient was admitted to the hospital for atrial fibrillation with rapid ventricular response and had multiple complications requiring prolonged hospitalization and rehabilitation. Repeat imaging 2 months later showed diffuse metastatic disease. However, her performance status had declined and she was not eligible for chemotherapy. She was placed under hospice care.
Discussion
This case demonstrates development of a cardiac pleomorphic sarcoma, a rare tumor, after placement of a Dacron graft. Given that foreign bodies have been found to induce sarcomas in experimental animals,3,4 and a few case reports have described sarcomas arising in association with Dacron vascular prostheses, 5-10 it seems that an exuberant host response around the foreign body might represent an important intermediate step in the development of the sarcoma.
There is no clearly defined pathogenesis that explains the link between a Dacron graft and sarcomas. In 1950s, Oppenheimer and colleagues described the formation of malignant tumors by various types of plastics, including Dacron, that were embedded in rats. 3,4 Most of the tumors were some form of sarcomas. It was inferred that physical properties of the plastics may have some role in tumor development. Plastics in sheet form or film that remained in situ for more than 6 months induced significant number of tumors compared with other forms such as sponges, films with holes, or powders.3,4 The 3-dimensional polymeric structure of the Dacron graft seems to play a role in induction of sarcoma as well. A pore diameter of less than 0.4 mm may increase tumorigenicity.11 The removal of the material before the 6-months mark does not lead to malignant tumors, which further supports the link between Dacron graft and formation of tumor. A pocket is formed around the foreign material after a certain period, as has been shown in histologic studies as the site of tumor origin.9,10
At the molecular level, the MDM-2/p53 pathway has been cited as possible mechanism for pathogenesis of intimal sarcoma.12,13 It has been suggested that endothelial dysplasia occurs as a precursor lesion in these sarcomas.14 The Dacron graft may cause a dysplastic effect on the endothelium leading to this precursor lesion and in certain cases transforming into sarcoma. Further definitive studies are required.
The primary treatment for cardiac sarcoma is surgical removal, although it is not always feasible. Findings in a Mayo clinic study showed that the median survival was 17 months for patients who underwent complete surgical excision, compared with 6 months for those who complete resection was not possible.15 In addition, a 10% survival rate at 1 year has been reported in primary cardiac sarcomas that are treated without any type of surgery.16
There is no clear-cut evidence supporting or refuting adjuvant chemotherapy for cardiac sarcoma. Some have inferred a potential benefit of adjuvant chemotherapy although definitive conclusions cannot be drawn. The median survival was 16.5 months in a case series of patients who received adjuvant chemotherapy, compared with 9 months and 11 months in 2 other case series.17,18,19 Multiple chemotherapy regimens have been used in the past for treatment. A retrospective s
Radiation showed some benefit in progression-free survival in a French retrospective study.21 Radiation therapies have been tried in other cases, as well in addition to chemotherapy. However, there is not enough data to support or refute it at this time.15,17,20 Several sporadic cases reported show benefit of cardiac transplantation.21,22
Conclusion
In consideration of the placement of the Dacron graft 8 years before the tumor occurrence, the anatomic proximity of the tumor to the Dacron graft, and the association between sarcoma with Dacron in medical literature, it seems logical to infer that this unusual malignancy in our patient is associated with the Dacron prosthesis.
1. Patil HR, Singh D, Hajdu M. Cardiac sarcoma presenting as heart failure and diagnosed as recurrent myxoma by echocardiogram. Eur J Echocardiogr. 2010;11(4):E12.
2. Awamleh P, Alberca MT, Gamallo C, Enrech S, Sarraj A. Left atrium myxosarcoma: an exceptional cardiac malignant primary tumor. Clin Cardiol. 2007;30(6):306-308.
3. Oppenheimer BS, Oppenheimer ET, Stout AP, Danishefsky I. Malignant tumors resulting from embedding plastics in rodents. Science. 1953;118:305-306.
4. Oppenheimer BS, Oppenheimer ET, Stout AP, Willhite M, Danishefski, I. The latent period in carcinogenesis by plastics in rats and its relation to the presarcomatous stage. Cancer. 1958;11(1):204-213.
5. Almeida NJ, Hoang P, Biddle P, Arouni A, Esterbrooks D. Primary cardiac angiosarcoma: in a patient with a Dacron aortic prosthesis. Tex Heart Inst J. 2011;38(1):61-65; discussion 65.
6. Stewart B, Manglik N, Zhao B, et al. Aortic intimal sarcoma: report of two cases with immunohistochemical analysis for pathogenesis. Cardiovasc Pathol. 2013;22(5):351-356.
7. Umscheid TW, Rouhani G, Morlang T, et al. Hemangiosarcoma after endovascular aortic aneurysm repair. J Endovasc Ther. 2007;14(1):101-105.
8. Ben-Izhak O, Vlodavsky E, Ofer A, Engel A, Nitecky S, Hoffman A. Epithelioid angiosarcoma associated with a Dacron vascular graft. Am J Surg Pathol. 1999;23(11):1418-1422.
9. Fyfe BS, Quintana CS, Kaneko M, Griepp RB. Aortic sarcoma four years after Dacron graft insertion. Ann Thorac Surg. 1994;58(6):1752-1754.
10. O’Connell TX, Fee HJ, Golding A. Sarcoma associated with Dacron prosthetic material: case report and review of the literature. J Thorac Cardiovasc Surg. 1976;72(1):94-96.
11. Karp RD, Johnson KH, Buoen LC, et al. Tumorogenesis by millipore filters in mice: histology and ultastructure of tissue reactions, as related to pore size. J Natl Cancer Inst. 1973;51:1275-1285.
12. Bode-Lesniewska B, Zhao J, Speel EJ, et al. Gains of 12q13-14 and overexpression of mdm2 are frequent findings in intimal sarcomas of the pulmonary artery. Virchows Arch. 2001;438:57-65.
13. Zeitz C, Rossle M, Haas C, et al. MDM-2 oncoprotein overexpression, p53 gene mutation, and VEGF up-regulation in angiosarcomas. Am J Surg Pathol. 1998;153:1425-1433.
14. Haber LM, Truong L. Immunohistochemical demonstration of the endothelialnature of aortic intimal sarcoma. Am J Surg Pathol. 1988 Oct;12(10):798-802. PubMed PMID: 3138923.
15. Simpson L, Kumar SK, Okuno SH, et al. Malignant primary cardiac tumors: review of a single institution experience. Cancer. 2008;112(11):2440-2446.
16. Leja MJ, Shah DJ, Reardon MJ. Primary cardiac tumors. Tex Heart Inst J. 2011;38(3):261-262.
17. Donsbeck AV, Ranchere D, Coindre JM, Le Gall F, Cordier JF, Loire R. Primary cardiac sarcomas: an immunohistochemical and grading study with long-term follow-up of 24 cases. Histopathology. 1999;34(4):295-304.
18. Putnam JB, Sweeney MS, Colon R, Lanza LA, Frazier OH, Cooley DC. Primary cardiac sarcomas. Ann Thorac Surg. 1990; 51; 906-910.
19. Murphy WR, Sweeney MS, Putnam JB et al. Surgical treatment of cardiac tumors: a 25-year experience. Ann Thorac Surg. 1990;49;612-618.
20. Llombart-Cussac A, Pivot X, Contesso G, et al. Adjuvant chemotherapy for primary cardiac sarcomas: the IGR experience. Br J Cancer. 1998;78(12):1624-1628.
21. Isambert N, Ray-Coquard I, Italiano A, et al. Primary cardiac sarcomas: a retrospective study of the French Sarcoma Group. Eur J Cancer. 2014;50(1):128-136.
22. Agaimy A, Rösch J, Weyand M, Strecker T. Primary and metastatic cardiac sarcomas: a 12-year experience at a German heart center. Int J Clin Exp Pathol. 2012;5(9):928-938.
Primary cardiac tumors, either benign or malignant, are very rare. The combined incidence is 0.002% on pooled autopsy series.1 The benign tumors account for 63% of primary cardiac tumors and include myxoma, the most common, and followed by papillary fibroelastoma, fibroma, and hemangioma. The remaining 37% are malignant tumors, essentially predominated by sarcomas.1
Although myxoma is the most common tumor arising in the left atrium, we present a case that shows that sarcoma can also arise from the same chamber. In fact, sarcomas could mimic cardiac myxoma.2 The cardiac sarcomas can have similar clinical presentation and more importantly can share similar histopathological features. Sarcomas may have myxoid features.2 Cases diagnosed as cardiac myxomas should be diligently worked up to rule out the presence of sarcomas with myxoid features. In addition, foreign bodies have been found to induce sarcomas in experimental animals.3,4 In particular, 2 case reports have described sarcomas arising in association with Dacron vascular prostheses in humans.5,6 We present here the case of a patient who was diagnosed with cardiac pleomorphic sarcoma 8 years after the placement of a Dacron graft.
Case presentation and summary
A 56-year-old woman with history of left atrial myxoma status after resection in 2005 and placement of a Dacron graft, morbid obesity, hypertension, and asthma presented to the emergency department with progressively worsening shortness of breath and blurry vision over period of 2 months. Acute coronary syndrome was ruled out by electrocardiogram and serial biomarkers. A computed-tomography angiogram was pursued because of her history of left atrial myxoma, and the results suggested the presence of a left atrial tumor. She underwent a transesophageal echocardiogram, which confirmed the presence of a large left atrial mass that likely was attached to the interatrial septum prolapsing across the mitral valve and was suggestive for recurrent left atrial myxoma (Figure 1). The results of a cardiac catheterization showed normal coronaries.
The patient subsequently underwent an excision of the left atrial tumor with profound internal and external myocardial cooling using antegrade blood cardioplegia under mildly hypothermic cardiopulmonary bypass. Frozen sections showed high-grade malignancy in favor of sarcoma. The hematoxylin and eosin stained permanent sections showed sheets of malignant pleomorphic spindle cells focally arranged in a storiform pattern. There were areas of necrosis and abundant mitotic activity. By immunohistochemical (IHC) stains, the tumor cells were diffusely positive for vimentin, and negative for pan-cytokeratin antibody (AE1/AE3), S-100 protein, Melan-A antibody, HMB45, CD34, CD31, myogenin, and MYOD1. IHC stains for CK-OSCAR, desmin, and smooth muscle actin were focally positive, and a ki-67 stain showed a proliferation index of about 80%. The histologic and IHC findings were consistent with a final diagnosis of high-grade undifferentiated pleomorphic sarcoma (Figure 2).
A positron emission tomography scan performed November 2013 did not show any other activity. The patient was scheduled for chemotherapy with adriamycin and ifosfamide with a plan for total of 6 cycles. Before her admission for the chemotherapy, the patient was admitted to the hospital for atrial fibrillation with rapid ventricular response and had multiple complications requiring prolonged hospitalization and rehabilitation. Repeat imaging 2 months later showed diffuse metastatic disease. However, her performance status had declined and she was not eligible for chemotherapy. She was placed under hospice care.
Discussion
This case demonstrates development of a cardiac pleomorphic sarcoma, a rare tumor, after placement of a Dacron graft. Given that foreign bodies have been found to induce sarcomas in experimental animals,3,4 and a few case reports have described sarcomas arising in association with Dacron vascular prostheses, 5-10 it seems that an exuberant host response around the foreign body might represent an important intermediate step in the development of the sarcoma.
There is no clearly defined pathogenesis that explains the link between a Dacron graft and sarcomas. In 1950s, Oppenheimer and colleagues described the formation of malignant tumors by various types of plastics, including Dacron, that were embedded in rats. 3,4 Most of the tumors were some form of sarcomas. It was inferred that physical properties of the plastics may have some role in tumor development. Plastics in sheet form or film that remained in situ for more than 6 months induced significant number of tumors compared with other forms such as sponges, films with holes, or powders.3,4 The 3-dimensional polymeric structure of the Dacron graft seems to play a role in induction of sarcoma as well. A pore diameter of less than 0.4 mm may increase tumorigenicity.11 The removal of the material before the 6-months mark does not lead to malignant tumors, which further supports the link between Dacron graft and formation of tumor. A pocket is formed around the foreign material after a certain period, as has been shown in histologic studies as the site of tumor origin.9,10
At the molecular level, the MDM-2/p53 pathway has been cited as possible mechanism for pathogenesis of intimal sarcoma.12,13 It has been suggested that endothelial dysplasia occurs as a precursor lesion in these sarcomas.14 The Dacron graft may cause a dysplastic effect on the endothelium leading to this precursor lesion and in certain cases transforming into sarcoma. Further definitive studies are required.
The primary treatment for cardiac sarcoma is surgical removal, although it is not always feasible. Findings in a Mayo clinic study showed that the median survival was 17 months for patients who underwent complete surgical excision, compared with 6 months for those who complete resection was not possible.15 In addition, a 10% survival rate at 1 year has been reported in primary cardiac sarcomas that are treated without any type of surgery.16
There is no clear-cut evidence supporting or refuting adjuvant chemotherapy for cardiac sarcoma. Some have inferred a potential benefit of adjuvant chemotherapy although definitive conclusions cannot be drawn. The median survival was 16.5 months in a case series of patients who received adjuvant chemotherapy, compared with 9 months and 11 months in 2 other case series.17,18,19 Multiple chemotherapy regimens have been used in the past for treatment. A retrospective s
Radiation showed some benefit in progression-free survival in a French retrospective study.21 Radiation therapies have been tried in other cases, as well in addition to chemotherapy. However, there is not enough data to support or refute it at this time.15,17,20 Several sporadic cases reported show benefit of cardiac transplantation.21,22
Conclusion
In consideration of the placement of the Dacron graft 8 years before the tumor occurrence, the anatomic proximity of the tumor to the Dacron graft, and the association between sarcoma with Dacron in medical literature, it seems logical to infer that this unusual malignancy in our patient is associated with the Dacron prosthesis.
Primary cardiac tumors, either benign or malignant, are very rare. The combined incidence is 0.002% on pooled autopsy series.1 The benign tumors account for 63% of primary cardiac tumors and include myxoma, the most common, and followed by papillary fibroelastoma, fibroma, and hemangioma. The remaining 37% are malignant tumors, essentially predominated by sarcomas.1
Although myxoma is the most common tumor arising in the left atrium, we present a case that shows that sarcoma can also arise from the same chamber. In fact, sarcomas could mimic cardiac myxoma.2 The cardiac sarcomas can have similar clinical presentation and more importantly can share similar histopathological features. Sarcomas may have myxoid features.2 Cases diagnosed as cardiac myxomas should be diligently worked up to rule out the presence of sarcomas with myxoid features. In addition, foreign bodies have been found to induce sarcomas in experimental animals.3,4 In particular, 2 case reports have described sarcomas arising in association with Dacron vascular prostheses in humans.5,6 We present here the case of a patient who was diagnosed with cardiac pleomorphic sarcoma 8 years after the placement of a Dacron graft.
Case presentation and summary
A 56-year-old woman with history of left atrial myxoma status after resection in 2005 and placement of a Dacron graft, morbid obesity, hypertension, and asthma presented to the emergency department with progressively worsening shortness of breath and blurry vision over period of 2 months. Acute coronary syndrome was ruled out by electrocardiogram and serial biomarkers. A computed-tomography angiogram was pursued because of her history of left atrial myxoma, and the results suggested the presence of a left atrial tumor. She underwent a transesophageal echocardiogram, which confirmed the presence of a large left atrial mass that likely was attached to the interatrial septum prolapsing across the mitral valve and was suggestive for recurrent left atrial myxoma (Figure 1). The results of a cardiac catheterization showed normal coronaries.
The patient subsequently underwent an excision of the left atrial tumor with profound internal and external myocardial cooling using antegrade blood cardioplegia under mildly hypothermic cardiopulmonary bypass. Frozen sections showed high-grade malignancy in favor of sarcoma. The hematoxylin and eosin stained permanent sections showed sheets of malignant pleomorphic spindle cells focally arranged in a storiform pattern. There were areas of necrosis and abundant mitotic activity. By immunohistochemical (IHC) stains, the tumor cells were diffusely positive for vimentin, and negative for pan-cytokeratin antibody (AE1/AE3), S-100 protein, Melan-A antibody, HMB45, CD34, CD31, myogenin, and MYOD1. IHC stains for CK-OSCAR, desmin, and smooth muscle actin were focally positive, and a ki-67 stain showed a proliferation index of about 80%. The histologic and IHC findings were consistent with a final diagnosis of high-grade undifferentiated pleomorphic sarcoma (Figure 2).
A positron emission tomography scan performed November 2013 did not show any other activity. The patient was scheduled for chemotherapy with adriamycin and ifosfamide with a plan for total of 6 cycles. Before her admission for the chemotherapy, the patient was admitted to the hospital for atrial fibrillation with rapid ventricular response and had multiple complications requiring prolonged hospitalization and rehabilitation. Repeat imaging 2 months later showed diffuse metastatic disease. However, her performance status had declined and she was not eligible for chemotherapy. She was placed under hospice care.
Discussion
This case demonstrates development of a cardiac pleomorphic sarcoma, a rare tumor, after placement of a Dacron graft. Given that foreign bodies have been found to induce sarcomas in experimental animals,3,4 and a few case reports have described sarcomas arising in association with Dacron vascular prostheses, 5-10 it seems that an exuberant host response around the foreign body might represent an important intermediate step in the development of the sarcoma.
There is no clearly defined pathogenesis that explains the link between a Dacron graft and sarcomas. In 1950s, Oppenheimer and colleagues described the formation of malignant tumors by various types of plastics, including Dacron, that were embedded in rats. 3,4 Most of the tumors were some form of sarcomas. It was inferred that physical properties of the plastics may have some role in tumor development. Plastics in sheet form or film that remained in situ for more than 6 months induced significant number of tumors compared with other forms such as sponges, films with holes, or powders.3,4 The 3-dimensional polymeric structure of the Dacron graft seems to play a role in induction of sarcoma as well. A pore diameter of less than 0.4 mm may increase tumorigenicity.11 The removal of the material before the 6-months mark does not lead to malignant tumors, which further supports the link between Dacron graft and formation of tumor. A pocket is formed around the foreign material after a certain period, as has been shown in histologic studies as the site of tumor origin.9,10
At the molecular level, the MDM-2/p53 pathway has been cited as possible mechanism for pathogenesis of intimal sarcoma.12,13 It has been suggested that endothelial dysplasia occurs as a precursor lesion in these sarcomas.14 The Dacron graft may cause a dysplastic effect on the endothelium leading to this precursor lesion and in certain cases transforming into sarcoma. Further definitive studies are required.
The primary treatment for cardiac sarcoma is surgical removal, although it is not always feasible. Findings in a Mayo clinic study showed that the median survival was 17 months for patients who underwent complete surgical excision, compared with 6 months for those who complete resection was not possible.15 In addition, a 10% survival rate at 1 year has been reported in primary cardiac sarcomas that are treated without any type of surgery.16
There is no clear-cut evidence supporting or refuting adjuvant chemotherapy for cardiac sarcoma. Some have inferred a potential benefit of adjuvant chemotherapy although definitive conclusions cannot be drawn. The median survival was 16.5 months in a case series of patients who received adjuvant chemotherapy, compared with 9 months and 11 months in 2 other case series.17,18,19 Multiple chemotherapy regimens have been used in the past for treatment. A retrospective s
Radiation showed some benefit in progression-free survival in a French retrospective study.21 Radiation therapies have been tried in other cases, as well in addition to chemotherapy. However, there is not enough data to support or refute it at this time.15,17,20 Several sporadic cases reported show benefit of cardiac transplantation.21,22
Conclusion
In consideration of the placement of the Dacron graft 8 years before the tumor occurrence, the anatomic proximity of the tumor to the Dacron graft, and the association between sarcoma with Dacron in medical literature, it seems logical to infer that this unusual malignancy in our patient is associated with the Dacron prosthesis.
1. Patil HR, Singh D, Hajdu M. Cardiac sarcoma presenting as heart failure and diagnosed as recurrent myxoma by echocardiogram. Eur J Echocardiogr. 2010;11(4):E12.
2. Awamleh P, Alberca MT, Gamallo C, Enrech S, Sarraj A. Left atrium myxosarcoma: an exceptional cardiac malignant primary tumor. Clin Cardiol. 2007;30(6):306-308.
3. Oppenheimer BS, Oppenheimer ET, Stout AP, Danishefsky I. Malignant tumors resulting from embedding plastics in rodents. Science. 1953;118:305-306.
4. Oppenheimer BS, Oppenheimer ET, Stout AP, Willhite M, Danishefski, I. The latent period in carcinogenesis by plastics in rats and its relation to the presarcomatous stage. Cancer. 1958;11(1):204-213.
5. Almeida NJ, Hoang P, Biddle P, Arouni A, Esterbrooks D. Primary cardiac angiosarcoma: in a patient with a Dacron aortic prosthesis. Tex Heart Inst J. 2011;38(1):61-65; discussion 65.
6. Stewart B, Manglik N, Zhao B, et al. Aortic intimal sarcoma: report of two cases with immunohistochemical analysis for pathogenesis. Cardiovasc Pathol. 2013;22(5):351-356.
7. Umscheid TW, Rouhani G, Morlang T, et al. Hemangiosarcoma after endovascular aortic aneurysm repair. J Endovasc Ther. 2007;14(1):101-105.
8. Ben-Izhak O, Vlodavsky E, Ofer A, Engel A, Nitecky S, Hoffman A. Epithelioid angiosarcoma associated with a Dacron vascular graft. Am J Surg Pathol. 1999;23(11):1418-1422.
9. Fyfe BS, Quintana CS, Kaneko M, Griepp RB. Aortic sarcoma four years after Dacron graft insertion. Ann Thorac Surg. 1994;58(6):1752-1754.
10. O’Connell TX, Fee HJ, Golding A. Sarcoma associated with Dacron prosthetic material: case report and review of the literature. J Thorac Cardiovasc Surg. 1976;72(1):94-96.
11. Karp RD, Johnson KH, Buoen LC, et al. Tumorogenesis by millipore filters in mice: histology and ultastructure of tissue reactions, as related to pore size. J Natl Cancer Inst. 1973;51:1275-1285.
12. Bode-Lesniewska B, Zhao J, Speel EJ, et al. Gains of 12q13-14 and overexpression of mdm2 are frequent findings in intimal sarcomas of the pulmonary artery. Virchows Arch. 2001;438:57-65.
13. Zeitz C, Rossle M, Haas C, et al. MDM-2 oncoprotein overexpression, p53 gene mutation, and VEGF up-regulation in angiosarcomas. Am J Surg Pathol. 1998;153:1425-1433.
14. Haber LM, Truong L. Immunohistochemical demonstration of the endothelialnature of aortic intimal sarcoma. Am J Surg Pathol. 1988 Oct;12(10):798-802. PubMed PMID: 3138923.
15. Simpson L, Kumar SK, Okuno SH, et al. Malignant primary cardiac tumors: review of a single institution experience. Cancer. 2008;112(11):2440-2446.
16. Leja MJ, Shah DJ, Reardon MJ. Primary cardiac tumors. Tex Heart Inst J. 2011;38(3):261-262.
17. Donsbeck AV, Ranchere D, Coindre JM, Le Gall F, Cordier JF, Loire R. Primary cardiac sarcomas: an immunohistochemical and grading study with long-term follow-up of 24 cases. Histopathology. 1999;34(4):295-304.
18. Putnam JB, Sweeney MS, Colon R, Lanza LA, Frazier OH, Cooley DC. Primary cardiac sarcomas. Ann Thorac Surg. 1990; 51; 906-910.
19. Murphy WR, Sweeney MS, Putnam JB et al. Surgical treatment of cardiac tumors: a 25-year experience. Ann Thorac Surg. 1990;49;612-618.
20. Llombart-Cussac A, Pivot X, Contesso G, et al. Adjuvant chemotherapy for primary cardiac sarcomas: the IGR experience. Br J Cancer. 1998;78(12):1624-1628.
21. Isambert N, Ray-Coquard I, Italiano A, et al. Primary cardiac sarcomas: a retrospective study of the French Sarcoma Group. Eur J Cancer. 2014;50(1):128-136.
22. Agaimy A, Rösch J, Weyand M, Strecker T. Primary and metastatic cardiac sarcomas: a 12-year experience at a German heart center. Int J Clin Exp Pathol. 2012;5(9):928-938.
1. Patil HR, Singh D, Hajdu M. Cardiac sarcoma presenting as heart failure and diagnosed as recurrent myxoma by echocardiogram. Eur J Echocardiogr. 2010;11(4):E12.
2. Awamleh P, Alberca MT, Gamallo C, Enrech S, Sarraj A. Left atrium myxosarcoma: an exceptional cardiac malignant primary tumor. Clin Cardiol. 2007;30(6):306-308.
3. Oppenheimer BS, Oppenheimer ET, Stout AP, Danishefsky I. Malignant tumors resulting from embedding plastics in rodents. Science. 1953;118:305-306.
4. Oppenheimer BS, Oppenheimer ET, Stout AP, Willhite M, Danishefski, I. The latent period in carcinogenesis by plastics in rats and its relation to the presarcomatous stage. Cancer. 1958;11(1):204-213.
5. Almeida NJ, Hoang P, Biddle P, Arouni A, Esterbrooks D. Primary cardiac angiosarcoma: in a patient with a Dacron aortic prosthesis. Tex Heart Inst J. 2011;38(1):61-65; discussion 65.
6. Stewart B, Manglik N, Zhao B, et al. Aortic intimal sarcoma: report of two cases with immunohistochemical analysis for pathogenesis. Cardiovasc Pathol. 2013;22(5):351-356.
7. Umscheid TW, Rouhani G, Morlang T, et al. Hemangiosarcoma after endovascular aortic aneurysm repair. J Endovasc Ther. 2007;14(1):101-105.
8. Ben-Izhak O, Vlodavsky E, Ofer A, Engel A, Nitecky S, Hoffman A. Epithelioid angiosarcoma associated with a Dacron vascular graft. Am J Surg Pathol. 1999;23(11):1418-1422.
9. Fyfe BS, Quintana CS, Kaneko M, Griepp RB. Aortic sarcoma four years after Dacron graft insertion. Ann Thorac Surg. 1994;58(6):1752-1754.
10. O’Connell TX, Fee HJ, Golding A. Sarcoma associated with Dacron prosthetic material: case report and review of the literature. J Thorac Cardiovasc Surg. 1976;72(1):94-96.
11. Karp RD, Johnson KH, Buoen LC, et al. Tumorogenesis by millipore filters in mice: histology and ultastructure of tissue reactions, as related to pore size. J Natl Cancer Inst. 1973;51:1275-1285.
12. Bode-Lesniewska B, Zhao J, Speel EJ, et al. Gains of 12q13-14 and overexpression of mdm2 are frequent findings in intimal sarcomas of the pulmonary artery. Virchows Arch. 2001;438:57-65.
13. Zeitz C, Rossle M, Haas C, et al. MDM-2 oncoprotein overexpression, p53 gene mutation, and VEGF up-regulation in angiosarcomas. Am J Surg Pathol. 1998;153:1425-1433.
14. Haber LM, Truong L. Immunohistochemical demonstration of the endothelialnature of aortic intimal sarcoma. Am J Surg Pathol. 1988 Oct;12(10):798-802. PubMed PMID: 3138923.
15. Simpson L, Kumar SK, Okuno SH, et al. Malignant primary cardiac tumors: review of a single institution experience. Cancer. 2008;112(11):2440-2446.
16. Leja MJ, Shah DJ, Reardon MJ. Primary cardiac tumors. Tex Heart Inst J. 2011;38(3):261-262.
17. Donsbeck AV, Ranchere D, Coindre JM, Le Gall F, Cordier JF, Loire R. Primary cardiac sarcomas: an immunohistochemical and grading study with long-term follow-up of 24 cases. Histopathology. 1999;34(4):295-304.
18. Putnam JB, Sweeney MS, Colon R, Lanza LA, Frazier OH, Cooley DC. Primary cardiac sarcomas. Ann Thorac Surg. 1990; 51; 906-910.
19. Murphy WR, Sweeney MS, Putnam JB et al. Surgical treatment of cardiac tumors: a 25-year experience. Ann Thorac Surg. 1990;49;612-618.
20. Llombart-Cussac A, Pivot X, Contesso G, et al. Adjuvant chemotherapy for primary cardiac sarcomas: the IGR experience. Br J Cancer. 1998;78(12):1624-1628.
21. Isambert N, Ray-Coquard I, Italiano A, et al. Primary cardiac sarcomas: a retrospective study of the French Sarcoma Group. Eur J Cancer. 2014;50(1):128-136.
22. Agaimy A, Rösch J, Weyand M, Strecker T. Primary and metastatic cardiac sarcomas: a 12-year experience at a German heart center. Int J Clin Exp Pathol. 2012;5(9):928-938.
Patient navigators’ personal experiences with cancer: does it have an impact on treatment?
Patient navigation has emerged in the past decade as a strategy to decrease cancer disparities among low-income, minority populations. Patient navigators help individuals who face personal and systemtic barriers to gaining access to care.1 Their role is to help patients find their way through a complex health care system,2,3 including logistic support of rescheduling appointments, assistance with transportation, and child care needs. They provide personal support, including coaching patients on their clinical visits, educating them about the cancer treatment process, and addressing their fears of diagnosis and treatment. Patient navigation has shown improvement in cancer screening rates, time to diagnostic resolution for those patients who have abnormal cancer screening tests, and quality of cancer care.4,5
In hiring patient navigators, it is not clear which professional training and skill sets and what personal experiences are most useful to becoming an effective navigator. Personal cancer experience can include a personal diagnosis, the experience of serving as a primary caregiver for a patient during treatment, or having a family member or close friend with cancer. Several current support programs specifically recruit cancer survivors on the assumption that their cancer treatment experience can provide helpful insights to a current patient for both emotional and logistical support.6 In this paper, we sought to address whether patient navigation promotes more timely diagnostic care if the navigator has experience with cancer.
Methods
This is a secondary analysis of the patients with abnormal cancer screening in the navigation arm of the national Patient Navigation Research Program (PNRP) study,1, 5 a collaborative effort across 10 centers to investigate the efficacy of patient navigation on improving patient-level outcomes for those who have abnormal results from a breast, cervical, colorectal, or prostate cancer screening test. The study demonstrated that patient navigation was effective in reducing delays in diagnosis and treatment5 and resulting in a higher quality of care,4 especially among vulnerable populations.7 The Institutional Review Board of each respective institution approved the research.
All of the patient navigators were paid employees with a minimum high-school diploma or equivalent. Navigators’ activities were standardized across centers through a national training program.8 Navigators used the care management model to identify and address barriers to care and to track participants throughout the course of their diagnostic evaluation,9 with the primary aim of timely diagnostic resolution. Most navigation programs were embedded within the clinical care system and interacted with patients through mail, by phone, and face-to-face contact.1
Data collection
Each center used agreed-upon inclusion and exclusion criteria and collected and coded the same patient-level data. Medical records were abstracted for pertinent clinical data on patients. Demographic data were collected through a patient survey or extracted from medical record registration. The central data coordinating center collected navigator information including demographic characteristics and experience with cancer.
We created a new variable, Personal experience with cancer. Personal experience with cancer was based on three questions asked of navigators: whether they were a cancer survivor; whether they were the primary caregiver to a family member or close friend with cancer; and whether they had a family member with cancer. Because of the small sample size, responses from navigators who were cancer survivors (n = 6) or primary caregivers to a family member with cancer (n = 4) were collapsed into a single category, referred to as personal experience with cancer, to compare with navigators who had no personal experience with cancer, which included those who reported a family member with cancer but who were not serving as a primary caregiver.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Each clinical center received approved from their institution’s human subjects review board. Informed consent was obtained from all patient navigator participants included in the study. Participating patients completed informed consent at some centers. At other centers where the study design was an implementation of a system intervention, a waiver of informed consent was approved by the Institutional Review Board.
Data analysis
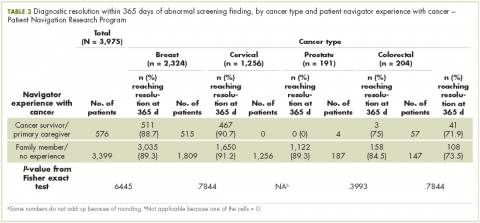
The primary outcome variable was time to diagnostic resolution. We included only participants supported by a single navigator. A Fisher exact test by cancer type was used to compare the two groups (personal experience vs none) in the proportion of patients who achieved diagnostic resolution by 365 days. We reviewed the percentage of patients resolved for the total population as well as stratified by cancer site (breast, cervical, prostate, and colorectal), owing to the known mean differences in time to diagnostic resolution by type of cancer.
Cox proportional hazard models and adjusted hazard ratios were developed and calculated to examine the impact of navigator’s personal experience with cancer on time to resolution, controlling for patient gender, race, age, and cancer type in the models. The analysis controlled for the individual effect of navigators through clustering. We used P < .05 as the cut-off for significance, and used Stata 10.1 (StataCorp, College Station Texas 77845) for all analyses.
Results
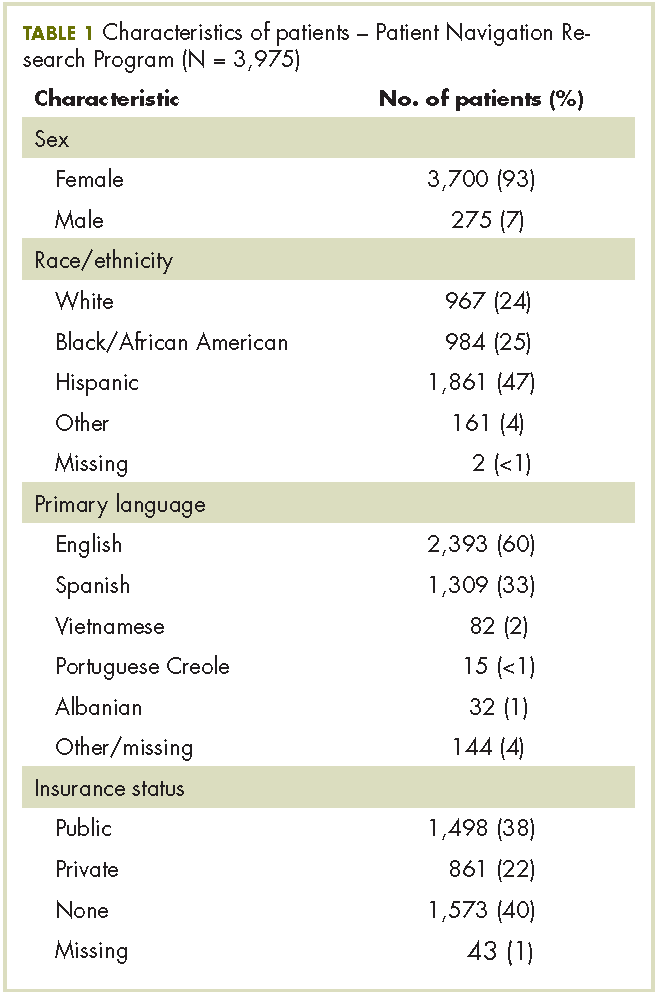
Our analytic sample included the 3,975 patients with only 1 navigator over the course of the study, 79% of the navigation (n = 5,063) arm. Most of the patients were women (93%), and most were from racial and ethnic minority communities. Most patients spoke English (60%), with Spanish (33%) as the next most common language. Most patients were publically insured (38%) or uninsured (40%) (Table 1).
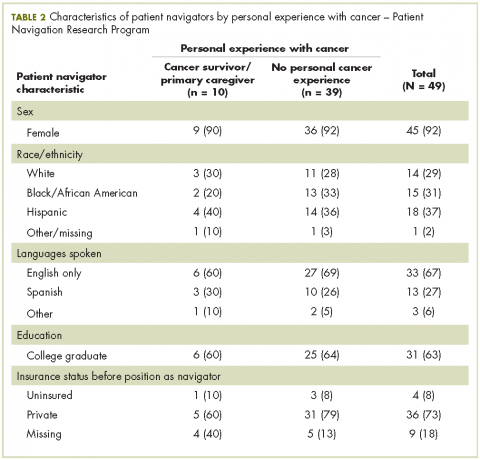
Of the total 49 navigators, 6 were cancer survivors and 4 were primary caregivers to a family member with cancer; an additional 19 reported that they had family members with cancer (Table 2). Most of the navigators were women. The racial/ethnic distribution mirrored the populations they served: white (29%); black or African American (31%); and Hispanic (37%). English was the only spoken language of 67% of the navigators; 27% spoke Spanish, and 6% reported speaking another language. Most had a college degree (63%).
The unadjusted bivariate comparison of patients who achieved diagnostic resolution within 365 days, by navigator experience with cancer, are shown in Table 3. We found no difference in time to diagnostic resolution for those patients for whom navigators had personal experience with cancer compared with those whose navigators had no experience. When stratified by type of cancer screening abnormality (breast, cervical, prostate, or colorectal), the results also did not reveal a significant difference in the proportion of patients achieving diagnostic resolution by 365 days by navigator experience with cancer.
In the Cox proportional hazard model adjusting for patient gender, age, race/ethnicity, cancer type, and adjusting for navigator using clustering, there was no difference between patients whose navigators had experience with cancer care, and those who did not (adjusted hazard ratio, 1.03; 95% confidence interval, .83-1.3). The level of education of navigators was not significantly associated with time to diagnostic resolution for patients.
Discussion
Although several cancer support programs have explicitly used cancer survivors as patient navigators or other supports for patients in active cancer care, there are scant data on whether this expertise improves care. Our study was not able to identify that navigators with previous experience with cancer care, either as a patient or as the primary caregiver, was associated with improved time to diagnostic resolution.
As patient navigation has become the standard of cancer diagnostic and treatment practices, there is a need to develop competencies and standards for hiring and training navigators. Part of this hiring process is to determine what past experience and training are relevant for effective navigation. There is little previous research on relevant skills of navigators, with only one study having demonstrated that language and racial/ ethnic concordance between patients and navigators was associated with more timely care. The national PNRP program hired mostly lay navigators with minimal medical experience, but with affiliations to the communities of the patients receiving care. Our program has demonstrated that lay individuals can be trained in the logistic aspects of navigation.5 Although it may seem intuitive that the experience of being a cancer survivor may make a navigator more empathetic, it is also possible that being too close to the experience of survivorship can also pose challenges to a navigator. Alternatively, navigation may be equally effective with proper training regardless of previous experience with cancer.
Our study is limited to addressing the outcome of timely resolution in the diagnostic phase of care after abnormal cancer screening. It is possible that past experience with cancer care will be beneficial when providing navigation for cancer care. While this study represents one of the largest groups of navigators who have been studied, the small sample may have limited our ability to detect differences. Our study has the benefit of a diverse group of navigators from a nationally representative, multi-site study. We suggest that prior experience with cancer care is not a prerequisite to supporting diagnostic care after abnormal cancer screening. Providing appropriate training to navigators may be sufficient to ensure effective and appropriate care is provided by patient navigators.
Acknowledgments
The authors acknowledge the contributions of the following members of the Patient Navigation Research Program:Clinical centers Boston Medical Center and Boston University: Karen M Freund (principal investigator [PI]), Tracy A Battaglia (co-PI); Denver Health and Hospital Authority: Peter Raich (PI), Elizabeth Whitley (co-PI); George Washington University Cancer Institute: Steven R Patierno (PI), Lisa M Alexander, Paul H Levine, Heather A Young, Heather J Hoffman, Nancy L LaVerda; H Lee Moffitt Cancer Center and Research Institute: Richard G Roetzheim (PI), Cathy Meade, Kristen J Wells; Northwest Portland Area Indian Health Board: Victoria Warren-Mears (PI); Northwestern University Robert H Lurie Comprehensive Cancer Center: Steven Rosen (PI), Melissa Simon; The Ohio State University Comprehensive Cancer Center: Electra Paskett (PI); University of Illinois at Chicago and Access Community Health Center: Elizabeth Calhoun (PI), Julie Darnell. University of Rochester: Kevin Fiscella (PI), Samantha Hendren; University of Texas Health Science Center at San Antonio Cancer Therapy and Research Center: Donald Dudley (PI), Kevin Hall, Anand Karnard, Amelie Ramirez. Program office National Cancer Institute, Center to Reduce Cancer Health Disparities: Martha Hare, Mollie Howerton, Ken Chu, Emmanuel Taylor, Mary Ann Van Dyun. Evaluation contractor NOVA Research Company: Paul Young, Frederick Snyder
6. Macvean ML, White VM, Sanson-Fisher R. One-to-one volunteer support programs for people with cancer: a review of the literature. Patient Educ Couns. 2008;70:10-24.
7. Rodday AM, Parsons SK, Snyder F, et al. The impact of patient navigation in eliminating economic disparities in cancer care. Cancer. 2015;121(22):4025-4034.
10. Charlot M, Santana MC, Chen CA, et al. Impact of patient and navigator race and language concordance on care after cancer screening abnormalities. Cancer. 2015;121(9):1477-1483.
Patient navigation has emerged in the past decade as a strategy to decrease cancer disparities among low-income, minority populations. Patient navigators help individuals who face personal and systemtic barriers to gaining access to care.1 Their role is to help patients find their way through a complex health care system,2,3 including logistic support of rescheduling appointments, assistance with transportation, and child care needs. They provide personal support, including coaching patients on their clinical visits, educating them about the cancer treatment process, and addressing their fears of diagnosis and treatment. Patient navigation has shown improvement in cancer screening rates, time to diagnostic resolution for those patients who have abnormal cancer screening tests, and quality of cancer care.4,5
In hiring patient navigators, it is not clear which professional training and skill sets and what personal experiences are most useful to becoming an effective navigator. Personal cancer experience can include a personal diagnosis, the experience of serving as a primary caregiver for a patient during treatment, or having a family member or close friend with cancer. Several current support programs specifically recruit cancer survivors on the assumption that their cancer treatment experience can provide helpful insights to a current patient for both emotional and logistical support.6 In this paper, we sought to address whether patient navigation promotes more timely diagnostic care if the navigator has experience with cancer.
Methods
This is a secondary analysis of the patients with abnormal cancer screening in the navigation arm of the national Patient Navigation Research Program (PNRP) study,1, 5 a collaborative effort across 10 centers to investigate the efficacy of patient navigation on improving patient-level outcomes for those who have abnormal results from a breast, cervical, colorectal, or prostate cancer screening test. The study demonstrated that patient navigation was effective in reducing delays in diagnosis and treatment5 and resulting in a higher quality of care,4 especially among vulnerable populations.7 The Institutional Review Board of each respective institution approved the research.
All of the patient navigators were paid employees with a minimum high-school diploma or equivalent. Navigators’ activities were standardized across centers through a national training program.8 Navigators used the care management model to identify and address barriers to care and to track participants throughout the course of their diagnostic evaluation,9 with the primary aim of timely diagnostic resolution. Most navigation programs were embedded within the clinical care system and interacted with patients through mail, by phone, and face-to-face contact.1
Data collection
Each center used agreed-upon inclusion and exclusion criteria and collected and coded the same patient-level data. Medical records were abstracted for pertinent clinical data on patients. Demographic data were collected through a patient survey or extracted from medical record registration. The central data coordinating center collected navigator information including demographic characteristics and experience with cancer.
We created a new variable, Personal experience with cancer. Personal experience with cancer was based on three questions asked of navigators: whether they were a cancer survivor; whether they were the primary caregiver to a family member or close friend with cancer; and whether they had a family member with cancer. Because of the small sample size, responses from navigators who were cancer survivors (n = 6) or primary caregivers to a family member with cancer (n = 4) were collapsed into a single category, referred to as personal experience with cancer, to compare with navigators who had no personal experience with cancer, which included those who reported a family member with cancer but who were not serving as a primary caregiver.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Each clinical center received approved from their institution’s human subjects review board. Informed consent was obtained from all patient navigator participants included in the study. Participating patients completed informed consent at some centers. At other centers where the study design was an implementation of a system intervention, a waiver of informed consent was approved by the Institutional Review Board.
Data analysis
The primary outcome variable was time to diagnostic resolution. We included only participants supported by a single navigator. A Fisher exact test by cancer type was used to compare the two groups (personal experience vs none) in the proportion of patients who achieved diagnostic resolution by 365 days. We reviewed the percentage of patients resolved for the total population as well as stratified by cancer site (breast, cervical, prostate, and colorectal), owing to the known mean differences in time to diagnostic resolution by type of cancer.
Cox proportional hazard models and adjusted hazard ratios were developed and calculated to examine the impact of navigator’s personal experience with cancer on time to resolution, controlling for patient gender, race, age, and cancer type in the models. The analysis controlled for the individual effect of navigators through clustering. We used P < .05 as the cut-off for significance, and used Stata 10.1 (StataCorp, College Station Texas 77845) for all analyses.
Results
Our analytic sample included the 3,975 patients with only 1 navigator over the course of the study, 79% of the navigation (n = 5,063) arm. Most of the patients were women (93%), and most were from racial and ethnic minority communities. Most patients spoke English (60%), with Spanish (33%) as the next most common language. Most patients were publically insured (38%) or uninsured (40%) (Table 1).
Of the total 49 navigators, 6 were cancer survivors and 4 were primary caregivers to a family member with cancer; an additional 19 reported that they had family members with cancer (Table 2). Most of the navigators were women. The racial/ethnic distribution mirrored the populations they served: white (29%); black or African American (31%); and Hispanic (37%). English was the only spoken language of 67% of the navigators; 27% spoke Spanish, and 6% reported speaking another language. Most had a college degree (63%).
The unadjusted bivariate comparison of patients who achieved diagnostic resolution within 365 days, by navigator experience with cancer, are shown in Table 3. We found no difference in time to diagnostic resolution for those patients for whom navigators had personal experience with cancer compared with those whose navigators had no experience. When stratified by type of cancer screening abnormality (breast, cervical, prostate, or colorectal), the results also did not reveal a significant difference in the proportion of patients achieving diagnostic resolution by 365 days by navigator experience with cancer.
In the Cox proportional hazard model adjusting for patient gender, age, race/ethnicity, cancer type, and adjusting for navigator using clustering, there was no difference between patients whose navigators had experience with cancer care, and those who did not (adjusted hazard ratio, 1.03; 95% confidence interval, .83-1.3). The level of education of navigators was not significantly associated with time to diagnostic resolution for patients.
Discussion
Although several cancer support programs have explicitly used cancer survivors as patient navigators or other supports for patients in active cancer care, there are scant data on whether this expertise improves care. Our study was not able to identify that navigators with previous experience with cancer care, either as a patient or as the primary caregiver, was associated with improved time to diagnostic resolution.
As patient navigation has become the standard of cancer diagnostic and treatment practices, there is a need to develop competencies and standards for hiring and training navigators. Part of this hiring process is to determine what past experience and training are relevant for effective navigation. There is little previous research on relevant skills of navigators, with only one study having demonstrated that language and racial/ ethnic concordance between patients and navigators was associated with more timely care. The national PNRP program hired mostly lay navigators with minimal medical experience, but with affiliations to the communities of the patients receiving care. Our program has demonstrated that lay individuals can be trained in the logistic aspects of navigation.5 Although it may seem intuitive that the experience of being a cancer survivor may make a navigator more empathetic, it is also possible that being too close to the experience of survivorship can also pose challenges to a navigator. Alternatively, navigation may be equally effective with proper training regardless of previous experience with cancer.
Our study is limited to addressing the outcome of timely resolution in the diagnostic phase of care after abnormal cancer screening. It is possible that past experience with cancer care will be beneficial when providing navigation for cancer care. While this study represents one of the largest groups of navigators who have been studied, the small sample may have limited our ability to detect differences. Our study has the benefit of a diverse group of navigators from a nationally representative, multi-site study. We suggest that prior experience with cancer care is not a prerequisite to supporting diagnostic care after abnormal cancer screening. Providing appropriate training to navigators may be sufficient to ensure effective and appropriate care is provided by patient navigators.
Acknowledgments
The authors acknowledge the contributions of the following members of the Patient Navigation Research Program:Clinical centers Boston Medical Center and Boston University: Karen M Freund (principal investigator [PI]), Tracy A Battaglia (co-PI); Denver Health and Hospital Authority: Peter Raich (PI), Elizabeth Whitley (co-PI); George Washington University Cancer Institute: Steven R Patierno (PI), Lisa M Alexander, Paul H Levine, Heather A Young, Heather J Hoffman, Nancy L LaVerda; H Lee Moffitt Cancer Center and Research Institute: Richard G Roetzheim (PI), Cathy Meade, Kristen J Wells; Northwest Portland Area Indian Health Board: Victoria Warren-Mears (PI); Northwestern University Robert H Lurie Comprehensive Cancer Center: Steven Rosen (PI), Melissa Simon; The Ohio State University Comprehensive Cancer Center: Electra Paskett (PI); University of Illinois at Chicago and Access Community Health Center: Elizabeth Calhoun (PI), Julie Darnell. University of Rochester: Kevin Fiscella (PI), Samantha Hendren; University of Texas Health Science Center at San Antonio Cancer Therapy and Research Center: Donald Dudley (PI), Kevin Hall, Anand Karnard, Amelie Ramirez. Program office National Cancer Institute, Center to Reduce Cancer Health Disparities: Martha Hare, Mollie Howerton, Ken Chu, Emmanuel Taylor, Mary Ann Van Dyun. Evaluation contractor NOVA Research Company: Paul Young, Frederick Snyder
Patient navigation has emerged in the past decade as a strategy to decrease cancer disparities among low-income, minority populations. Patient navigators help individuals who face personal and systemtic barriers to gaining access to care.1 Their role is to help patients find their way through a complex health care system,2,3 including logistic support of rescheduling appointments, assistance with transportation, and child care needs. They provide personal support, including coaching patients on their clinical visits, educating them about the cancer treatment process, and addressing their fears of diagnosis and treatment. Patient navigation has shown improvement in cancer screening rates, time to diagnostic resolution for those patients who have abnormal cancer screening tests, and quality of cancer care.4,5
In hiring patient navigators, it is not clear which professional training and skill sets and what personal experiences are most useful to becoming an effective navigator. Personal cancer experience can include a personal diagnosis, the experience of serving as a primary caregiver for a patient during treatment, or having a family member or close friend with cancer. Several current support programs specifically recruit cancer survivors on the assumption that their cancer treatment experience can provide helpful insights to a current patient for both emotional and logistical support.6 In this paper, we sought to address whether patient navigation promotes more timely diagnostic care if the navigator has experience with cancer.
Methods
This is a secondary analysis of the patients with abnormal cancer screening in the navigation arm of the national Patient Navigation Research Program (PNRP) study,1, 5 a collaborative effort across 10 centers to investigate the efficacy of patient navigation on improving patient-level outcomes for those who have abnormal results from a breast, cervical, colorectal, or prostate cancer screening test. The study demonstrated that patient navigation was effective in reducing delays in diagnosis and treatment5 and resulting in a higher quality of care,4 especially among vulnerable populations.7 The Institutional Review Board of each respective institution approved the research.
All of the patient navigators were paid employees with a minimum high-school diploma or equivalent. Navigators’ activities were standardized across centers through a national training program.8 Navigators used the care management model to identify and address barriers to care and to track participants throughout the course of their diagnostic evaluation,9 with the primary aim of timely diagnostic resolution. Most navigation programs were embedded within the clinical care system and interacted with patients through mail, by phone, and face-to-face contact.1
Data collection
Each center used agreed-upon inclusion and exclusion criteria and collected and coded the same patient-level data. Medical records were abstracted for pertinent clinical data on patients. Demographic data were collected through a patient survey or extracted from medical record registration. The central data coordinating center collected navigator information including demographic characteristics and experience with cancer.
We created a new variable, Personal experience with cancer. Personal experience with cancer was based on three questions asked of navigators: whether they were a cancer survivor; whether they were the primary caregiver to a family member or close friend with cancer; and whether they had a family member with cancer. Because of the small sample size, responses from navigators who were cancer survivors (n = 6) or primary caregivers to a family member with cancer (n = 4) were collapsed into a single category, referred to as personal experience with cancer, to compare with navigators who had no personal experience with cancer, which included those who reported a family member with cancer but who were not serving as a primary caregiver.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Each clinical center received approved from their institution’s human subjects review board. Informed consent was obtained from all patient navigator participants included in the study. Participating patients completed informed consent at some centers. At other centers where the study design was an implementation of a system intervention, a waiver of informed consent was approved by the Institutional Review Board.
Data analysis
The primary outcome variable was time to diagnostic resolution. We included only participants supported by a single navigator. A Fisher exact test by cancer type was used to compare the two groups (personal experience vs none) in the proportion of patients who achieved diagnostic resolution by 365 days. We reviewed the percentage of patients resolved for the total population as well as stratified by cancer site (breast, cervical, prostate, and colorectal), owing to the known mean differences in time to diagnostic resolution by type of cancer.
Cox proportional hazard models and adjusted hazard ratios were developed and calculated to examine the impact of navigator’s personal experience with cancer on time to resolution, controlling for patient gender, race, age, and cancer type in the models. The analysis controlled for the individual effect of navigators through clustering. We used P < .05 as the cut-off for significance, and used Stata 10.1 (StataCorp, College Station Texas 77845) for all analyses.
Results
Our analytic sample included the 3,975 patients with only 1 navigator over the course of the study, 79% of the navigation (n = 5,063) arm. Most of the patients were women (93%), and most were from racial and ethnic minority communities. Most patients spoke English (60%), with Spanish (33%) as the next most common language. Most patients were publically insured (38%) or uninsured (40%) (Table 1).
Of the total 49 navigators, 6 were cancer survivors and 4 were primary caregivers to a family member with cancer; an additional 19 reported that they had family members with cancer (Table 2). Most of the navigators were women. The racial/ethnic distribution mirrored the populations they served: white (29%); black or African American (31%); and Hispanic (37%). English was the only spoken language of 67% of the navigators; 27% spoke Spanish, and 6% reported speaking another language. Most had a college degree (63%).
The unadjusted bivariate comparison of patients who achieved diagnostic resolution within 365 days, by navigator experience with cancer, are shown in Table 3. We found no difference in time to diagnostic resolution for those patients for whom navigators had personal experience with cancer compared with those whose navigators had no experience. When stratified by type of cancer screening abnormality (breast, cervical, prostate, or colorectal), the results also did not reveal a significant difference in the proportion of patients achieving diagnostic resolution by 365 days by navigator experience with cancer.
In the Cox proportional hazard model adjusting for patient gender, age, race/ethnicity, cancer type, and adjusting for navigator using clustering, there was no difference between patients whose navigators had experience with cancer care, and those who did not (adjusted hazard ratio, 1.03; 95% confidence interval, .83-1.3). The level of education of navigators was not significantly associated with time to diagnostic resolution for patients.
Discussion
Although several cancer support programs have explicitly used cancer survivors as patient navigators or other supports for patients in active cancer care, there are scant data on whether this expertise improves care. Our study was not able to identify that navigators with previous experience with cancer care, either as a patient or as the primary caregiver, was associated with improved time to diagnostic resolution.
As patient navigation has become the standard of cancer diagnostic and treatment practices, there is a need to develop competencies and standards for hiring and training navigators. Part of this hiring process is to determine what past experience and training are relevant for effective navigation. There is little previous research on relevant skills of navigators, with only one study having demonstrated that language and racial/ ethnic concordance between patients and navigators was associated with more timely care. The national PNRP program hired mostly lay navigators with minimal medical experience, but with affiliations to the communities of the patients receiving care. Our program has demonstrated that lay individuals can be trained in the logistic aspects of navigation.5 Although it may seem intuitive that the experience of being a cancer survivor may make a navigator more empathetic, it is also possible that being too close to the experience of survivorship can also pose challenges to a navigator. Alternatively, navigation may be equally effective with proper training regardless of previous experience with cancer.
Our study is limited to addressing the outcome of timely resolution in the diagnostic phase of care after abnormal cancer screening. It is possible that past experience with cancer care will be beneficial when providing navigation for cancer care. While this study represents one of the largest groups of navigators who have been studied, the small sample may have limited our ability to detect differences. Our study has the benefit of a diverse group of navigators from a nationally representative, multi-site study. We suggest that prior experience with cancer care is not a prerequisite to supporting diagnostic care after abnormal cancer screening. Providing appropriate training to navigators may be sufficient to ensure effective and appropriate care is provided by patient navigators.
Acknowledgments
The authors acknowledge the contributions of the following members of the Patient Navigation Research Program:Clinical centers Boston Medical Center and Boston University: Karen M Freund (principal investigator [PI]), Tracy A Battaglia (co-PI); Denver Health and Hospital Authority: Peter Raich (PI), Elizabeth Whitley (co-PI); George Washington University Cancer Institute: Steven R Patierno (PI), Lisa M Alexander, Paul H Levine, Heather A Young, Heather J Hoffman, Nancy L LaVerda; H Lee Moffitt Cancer Center and Research Institute: Richard G Roetzheim (PI), Cathy Meade, Kristen J Wells; Northwest Portland Area Indian Health Board: Victoria Warren-Mears (PI); Northwestern University Robert H Lurie Comprehensive Cancer Center: Steven Rosen (PI), Melissa Simon; The Ohio State University Comprehensive Cancer Center: Electra Paskett (PI); University of Illinois at Chicago and Access Community Health Center: Elizabeth Calhoun (PI), Julie Darnell. University of Rochester: Kevin Fiscella (PI), Samantha Hendren; University of Texas Health Science Center at San Antonio Cancer Therapy and Research Center: Donald Dudley (PI), Kevin Hall, Anand Karnard, Amelie Ramirez. Program office National Cancer Institute, Center to Reduce Cancer Health Disparities: Martha Hare, Mollie Howerton, Ken Chu, Emmanuel Taylor, Mary Ann Van Dyun. Evaluation contractor NOVA Research Company: Paul Young, Frederick Snyder
6. Macvean ML, White VM, Sanson-Fisher R. One-to-one volunteer support programs for people with cancer: a review of the literature. Patient Educ Couns. 2008;70:10-24.
7. Rodday AM, Parsons SK, Snyder F, et al. The impact of patient navigation in eliminating economic disparities in cancer care. Cancer. 2015;121(22):4025-4034.
10. Charlot M, Santana MC, Chen CA, et al. Impact of patient and navigator race and language concordance on care after cancer screening abnormalities. Cancer. 2015;121(9):1477-1483.
6. Macvean ML, White VM, Sanson-Fisher R. One-to-one volunteer support programs for people with cancer: a review of the literature. Patient Educ Couns. 2008;70:10-24.
7. Rodday AM, Parsons SK, Snyder F, et al. The impact of patient navigation in eliminating economic disparities in cancer care. Cancer. 2015;121(22):4025-4034.
10. Charlot M, Santana MC, Chen CA, et al. Impact of patient and navigator race and language concordance on care after cancer screening abnormalities. Cancer. 2015;121(9):1477-1483.
Sexual aids not available to cancer survivors despite recommendations
ORLANDO – Therapeutic aids for sexual rehabilitation were not available at most major cancer centers, according to results of a structured telephone survey presented at the Cancer Survivorship Symposium.
Of the centers reached, 87% said they had no sexual aids available for men, and 72% said they had no such aids for women, said lead study author Sharon Bober, PhD, a psychologist at the Dana-Farber Cancer Institute in Boston, Massachusetts.
“I think the scarcity of all of these products really underscores the cultural taboos around sexual dysfunction, as did some of the discomfort of the staff responding to our calls,” Dr. Bober said in a press conference at the symposium, which was sponsored by the American Academy of Family Physicians, the American College of Physicians, and the American Society of Clinical Oncology.
Cancer treatment guidelines from the National Comprehensive Cancer Network (NCCN) recommend therapeutic aids for sexual health rehabilitation including vaginal dilators, moisturizers, and vacuum erection devices, Dr. Bober said.
Dr. Bober and her colleagues surveyed 25 NCI-designated Cancer Centers/National Comprehensive Cancer Network–member institutions about on-site availability of sexual aids and resources for cancer survivors.
After conducting internet searches and phone calls designed to identify potential sources of sexual aids at each center, study staff posed as relatives of patients and used a structured script to query cancer center staff about on-site availability of sexual aids.
Separate calls were conducted to query on availability of men and women’s sexual aids.
Of 23 centers that responded about men, 87% reported having no sexual aids, and of 22 centers that responded about women, 72% reported having no sexual aids, Dr. Bober reported at the symposium.
The lack of sexual aids was particularly notable given the wide availability of wigs, prosthetics, sunscreen, and other cancer care products at leading cancer centers, she added.
“Only one center of the 25 had an extensive list of products and resources for both men and women, which may well serve as a model when we think about the needs for cancer survivors in general,” said Dr. Bober.
These results suggest that leading cancer centers are not meeting the needs of cancer survivors in terms of recommended sexual therapeutic aids and informational resources, according to Timothy Gilligan, MD, an American Society of Clinical Oncology expert and member of the Cancer Survivorship news planning team.
“You sort of wonder where a cancer patient’s supposed to go to get this information if not at the Cancer Center,” said Dr. Gilligan, who moderated the press conference. “We’re really kind of leaving them shortchanged here, and the good news is I think we could easily do better if we just decide that we want to.”
The study was funded by Dana-Farber Cancer Institute. Dr. Bober reported research funding from Apex Neuro.
SOURCE: Bober S. et al. Cancer Survivorship Symposium Abstract #134
ORLANDO – Therapeutic aids for sexual rehabilitation were not available at most major cancer centers, according to results of a structured telephone survey presented at the Cancer Survivorship Symposium.
Of the centers reached, 87% said they had no sexual aids available for men, and 72% said they had no such aids for women, said lead study author Sharon Bober, PhD, a psychologist at the Dana-Farber Cancer Institute in Boston, Massachusetts.
“I think the scarcity of all of these products really underscores the cultural taboos around sexual dysfunction, as did some of the discomfort of the staff responding to our calls,” Dr. Bober said in a press conference at the symposium, which was sponsored by the American Academy of Family Physicians, the American College of Physicians, and the American Society of Clinical Oncology.
Cancer treatment guidelines from the National Comprehensive Cancer Network (NCCN) recommend therapeutic aids for sexual health rehabilitation including vaginal dilators, moisturizers, and vacuum erection devices, Dr. Bober said.
Dr. Bober and her colleagues surveyed 25 NCI-designated Cancer Centers/National Comprehensive Cancer Network–member institutions about on-site availability of sexual aids and resources for cancer survivors.
After conducting internet searches and phone calls designed to identify potential sources of sexual aids at each center, study staff posed as relatives of patients and used a structured script to query cancer center staff about on-site availability of sexual aids.
Separate calls were conducted to query on availability of men and women’s sexual aids.
Of 23 centers that responded about men, 87% reported having no sexual aids, and of 22 centers that responded about women, 72% reported having no sexual aids, Dr. Bober reported at the symposium.
The lack of sexual aids was particularly notable given the wide availability of wigs, prosthetics, sunscreen, and other cancer care products at leading cancer centers, she added.
“Only one center of the 25 had an extensive list of products and resources for both men and women, which may well serve as a model when we think about the needs for cancer survivors in general,” said Dr. Bober.
These results suggest that leading cancer centers are not meeting the needs of cancer survivors in terms of recommended sexual therapeutic aids and informational resources, according to Timothy Gilligan, MD, an American Society of Clinical Oncology expert and member of the Cancer Survivorship news planning team.
“You sort of wonder where a cancer patient’s supposed to go to get this information if not at the Cancer Center,” said Dr. Gilligan, who moderated the press conference. “We’re really kind of leaving them shortchanged here, and the good news is I think we could easily do better if we just decide that we want to.”
The study was funded by Dana-Farber Cancer Institute. Dr. Bober reported research funding from Apex Neuro.
SOURCE: Bober S. et al. Cancer Survivorship Symposium Abstract #134
ORLANDO – Therapeutic aids for sexual rehabilitation were not available at most major cancer centers, according to results of a structured telephone survey presented at the Cancer Survivorship Symposium.
Of the centers reached, 87% said they had no sexual aids available for men, and 72% said they had no such aids for women, said lead study author Sharon Bober, PhD, a psychologist at the Dana-Farber Cancer Institute in Boston, Massachusetts.
“I think the scarcity of all of these products really underscores the cultural taboos around sexual dysfunction, as did some of the discomfort of the staff responding to our calls,” Dr. Bober said in a press conference at the symposium, which was sponsored by the American Academy of Family Physicians, the American College of Physicians, and the American Society of Clinical Oncology.
Cancer treatment guidelines from the National Comprehensive Cancer Network (NCCN) recommend therapeutic aids for sexual health rehabilitation including vaginal dilators, moisturizers, and vacuum erection devices, Dr. Bober said.
Dr. Bober and her colleagues surveyed 25 NCI-designated Cancer Centers/National Comprehensive Cancer Network–member institutions about on-site availability of sexual aids and resources for cancer survivors.
After conducting internet searches and phone calls designed to identify potential sources of sexual aids at each center, study staff posed as relatives of patients and used a structured script to query cancer center staff about on-site availability of sexual aids.
Separate calls were conducted to query on availability of men and women’s sexual aids.
Of 23 centers that responded about men, 87% reported having no sexual aids, and of 22 centers that responded about women, 72% reported having no sexual aids, Dr. Bober reported at the symposium.
The lack of sexual aids was particularly notable given the wide availability of wigs, prosthetics, sunscreen, and other cancer care products at leading cancer centers, she added.
“Only one center of the 25 had an extensive list of products and resources for both men and women, which may well serve as a model when we think about the needs for cancer survivors in general,” said Dr. Bober.
These results suggest that leading cancer centers are not meeting the needs of cancer survivors in terms of recommended sexual therapeutic aids and informational resources, according to Timothy Gilligan, MD, an American Society of Clinical Oncology expert and member of the Cancer Survivorship news planning team.
“You sort of wonder where a cancer patient’s supposed to go to get this information if not at the Cancer Center,” said Dr. Gilligan, who moderated the press conference. “We’re really kind of leaving them shortchanged here, and the good news is I think we could easily do better if we just decide that we want to.”
The study was funded by Dana-Farber Cancer Institute. Dr. Bober reported research funding from Apex Neuro.
SOURCE: Bober S. et al. Cancer Survivorship Symposium Abstract #134
FROM THE CSC 2018
Key clinical point: Therapeutic aids for sexual health rehabilitation were not available at most leading cancer centers, despite clinical practice guidelines recommending their use.
Major finding: Of the centers reached, 87% said they had no sexual aids available for men, and 72% said they had no aids for women.
Data source: Analysis of responses from cancer center staff at 25 NCI-designated cancer centers to telephone queries that used a structured script.
Disclosures: Study funding came from Dana-Farber Cancer Institute. Dr. Bober reported research funding from Apex Neuro.
Source: Bober S. et al. Cancer Survivorship Symposium, Abstract #134.
Exercise during chemotherapy may yield long-term physical benefits
ORLANDO – Physical exercise during adjuvant chemotherapy may lead to improved physical activity and decreased fatigue years after the treatment is completed, results of a recent analysis suggest.
Four years after participating in an exercise program that took place during cancer treatment, patients reported more moderate-to-vigorous activity and less fatigue, compared with patients who did not participate in the program, according to long-term follow-up results presented at the Cancer Survivorship Symposium.
“We think that offering exercise during cancer treatment, including chemotherapy, is recommended and has beneficial short- and long-term effects on health,” said Anne M. May, PhD, of University Medical Center, Utrecht, the Netherlands.
Speaking in a press conference at the symposium, Dr. May described results of the analysis, which included 128 patients who had previously participated in PACT, a 237-patient randomized controlled trial evaluating a supervised exercise program versus usual care in patients undergoing adjuvant treatment for breast or colon cancer.
The 18-week exercise program included moderate- to high-intensity aerobic and strength training under physical therapist supervision for 60 minutes twice weekly, plus home-based physical activity for 30 minutes three times weekly.
At 4 years’ follow-up, patients in the exercise group reported on average 90 minutes of moderate-to-vigorous exercise per week, compared to an average of 70 minutes per week in the usual care group (P less than .05), Dr. May reported at the symposium, which was sponsored by the American Academy of Family Physicians, the American College of Physicians, and the American Society of Clinical Oncology.
There was also a trend toward decreased fatigue reported in the exercise vs. usual care group, though the finding did not reach statistical significance, she said.
It is “encouraging” to see that this exercise program had a long-term impact on patients’ physical activity levels, said ASCO expert Timothy Gilligan, MD, MSc.
“I think the public sometimes gets jaded because the nutritional recommendations seem to change every year, but if you look at the research on exercise in health … it’s interesting how consistent the data is that exercise really is good for us – if we can only get people to do it,” said Dr. Gilligan, who moderated the press conference.
Dr. May said she had no disclosures to report for the study, which was supported by grants from the Dutch Cancer Society, the Dutch Pink Ribbon Foundation, and the Netherlands Organization for Health Research.
SOURCE: May AM et al. Cancer Survivorship Symposium, Abstract 99.
ORLANDO – Physical exercise during adjuvant chemotherapy may lead to improved physical activity and decreased fatigue years after the treatment is completed, results of a recent analysis suggest.
Four years after participating in an exercise program that took place during cancer treatment, patients reported more moderate-to-vigorous activity and less fatigue, compared with patients who did not participate in the program, according to long-term follow-up results presented at the Cancer Survivorship Symposium.
“We think that offering exercise during cancer treatment, including chemotherapy, is recommended and has beneficial short- and long-term effects on health,” said Anne M. May, PhD, of University Medical Center, Utrecht, the Netherlands.
Speaking in a press conference at the symposium, Dr. May described results of the analysis, which included 128 patients who had previously participated in PACT, a 237-patient randomized controlled trial evaluating a supervised exercise program versus usual care in patients undergoing adjuvant treatment for breast or colon cancer.
The 18-week exercise program included moderate- to high-intensity aerobic and strength training under physical therapist supervision for 60 minutes twice weekly, plus home-based physical activity for 30 minutes three times weekly.
At 4 years’ follow-up, patients in the exercise group reported on average 90 minutes of moderate-to-vigorous exercise per week, compared to an average of 70 minutes per week in the usual care group (P less than .05), Dr. May reported at the symposium, which was sponsored by the American Academy of Family Physicians, the American College of Physicians, and the American Society of Clinical Oncology.
There was also a trend toward decreased fatigue reported in the exercise vs. usual care group, though the finding did not reach statistical significance, she said.
It is “encouraging” to see that this exercise program had a long-term impact on patients’ physical activity levels, said ASCO expert Timothy Gilligan, MD, MSc.
“I think the public sometimes gets jaded because the nutritional recommendations seem to change every year, but if you look at the research on exercise in health … it’s interesting how consistent the data is that exercise really is good for us – if we can only get people to do it,” said Dr. Gilligan, who moderated the press conference.
Dr. May said she had no disclosures to report for the study, which was supported by grants from the Dutch Cancer Society, the Dutch Pink Ribbon Foundation, and the Netherlands Organization for Health Research.
SOURCE: May AM et al. Cancer Survivorship Symposium, Abstract 99.
ORLANDO – Physical exercise during adjuvant chemotherapy may lead to improved physical activity and decreased fatigue years after the treatment is completed, results of a recent analysis suggest.
Four years after participating in an exercise program that took place during cancer treatment, patients reported more moderate-to-vigorous activity and less fatigue, compared with patients who did not participate in the program, according to long-term follow-up results presented at the Cancer Survivorship Symposium.
“We think that offering exercise during cancer treatment, including chemotherapy, is recommended and has beneficial short- and long-term effects on health,” said Anne M. May, PhD, of University Medical Center, Utrecht, the Netherlands.
Speaking in a press conference at the symposium, Dr. May described results of the analysis, which included 128 patients who had previously participated in PACT, a 237-patient randomized controlled trial evaluating a supervised exercise program versus usual care in patients undergoing adjuvant treatment for breast or colon cancer.
The 18-week exercise program included moderate- to high-intensity aerobic and strength training under physical therapist supervision for 60 minutes twice weekly, plus home-based physical activity for 30 minutes three times weekly.
At 4 years’ follow-up, patients in the exercise group reported on average 90 minutes of moderate-to-vigorous exercise per week, compared to an average of 70 minutes per week in the usual care group (P less than .05), Dr. May reported at the symposium, which was sponsored by the American Academy of Family Physicians, the American College of Physicians, and the American Society of Clinical Oncology.
There was also a trend toward decreased fatigue reported in the exercise vs. usual care group, though the finding did not reach statistical significance, she said.
It is “encouraging” to see that this exercise program had a long-term impact on patients’ physical activity levels, said ASCO expert Timothy Gilligan, MD, MSc.
“I think the public sometimes gets jaded because the nutritional recommendations seem to change every year, but if you look at the research on exercise in health … it’s interesting how consistent the data is that exercise really is good for us – if we can only get people to do it,” said Dr. Gilligan, who moderated the press conference.
Dr. May said she had no disclosures to report for the study, which was supported by grants from the Dutch Cancer Society, the Dutch Pink Ribbon Foundation, and the Netherlands Organization for Health Research.
SOURCE: May AM et al. Cancer Survivorship Symposium, Abstract 99.
REPORTING FROM CSC 2018
Key clinical point: Exercise during adjuvant chemotherapy may lead to lower levels of fatigue and higher levels of physical activity years after treatment is completed.
Major finding: Four years after participating in an 18-week exercise program during cancer treatment, patients reported an average of 90 minutes of moderate-to-vigorous physical activity per day, compared with 70 minutes for patients who did not participate (P less than .05).
Study details: Long-term follow-up of 128 patients who had participated in the PACT study, a randomized controlled trial of a supervised exercise program versus usual care in breast and colon cancer patients undergoing adjuvant treatment including chemotherapy.
Disclosures: The authors reported no disclosures relevant to the study, which was supported by grants from the Dutch Cancer Society, the Dutch Pink Ribbon Foundation, and the Netherlands Organization for Health Research.
Source: May AM et al. Cancer Survivorship Symposium, Abstract 99.
From angst to analytics: lessons learned from an oncology care model internal pilot
In March 2016, 13 practices affiliated with the US Oncology Network (USON) were invited to participate in the Oncology Care Model (OCM) proposed by the Center for Medicare and Medicaid Services (CMS) and Center for Medicare and Medicaid Innovation (CMMI). The OCM, a novel value-based care model, was designed to provide higher-quality and better-coordinated oncology care at a lower cost to CMS.1 Of the 13 practices, 12 agreed to participate with a start date for the program of July 1, 2016. At least 40% of the practices’ patients were insured by Medicare, and any eligible patients with active cancer were offered an opportunity to enter the program. USON practices treat more than 25,000 patients with a qualifying episode per year and the overall OCM program sees more than 150,000 beneficiaries per year,2 so we anticipated that the OCM would have a substantial impact on each of the 12 practices on USON.
Faced with the scenario of having only 3 months between notification of approval and launch of the OCM, it was imperative that all the practices be proactive in planning and preparing to launch the OCM. With this goal in mind, representatives from all OCM candidate practices convened to anticipate the needs of the OCM and chart out a program to meet those needs. In this article, we discuss the requirements and scope of the OCM, the development of an internal pilot project, the anticipated gains from the pilot, and the results and findings from the pilot, both expected and unexpected.
The road to the Oncology Care Model
The government and oncology practices have been on separate trajectories to the OCM. In the last 15 years, the major intersections of these trajectories had to do with price and not patient outcomes. In 2003, the Medicare Prescription Drug Improvement and Modernization Act (MMA) focused on drug price reductions from an average wholesale price–based schedule to an average sales price–based schedule.3 There was the sequester in 2013,4 and more recently a proposal to restructure the payment for Part B drugs. In the background, recurrent negotiations to fix the calculation for the sustainable growth rate allowed for periodic draconian cuts to the prices of services. The cumulative effect of these price reductions has been to put economic pressure on community oncologists such that many have moved to a hospital environment.5
This contentious relationship with community oncology began to change with the passage of the Affordable Care Act (ACA) in 2010.6 Section 3022 of the ACA established the Medicare Shared Savings Program (MSSP) with the charge to create a new type of health care entity that was responsible for achieving the triple aim of improving population health, improving individual patient care, and bending the cost curve.7 Additional programs, such as the Pioneer Accountable Care Organization (ACO) program and the Comprehensive Primary Care Initiative were established to test alternative payment models.8-10
The ACA also funded the CMMI with a mandate to “test innovative payment and service delivery models” to achieve the triple aim; US$10 billion were appropriated for the years 2011-2019 for this purpose. The CMMI funded a pilot project for cancer care, the COME HOME [Community Oncology MEdical HOME] initiative, to test whether some aspects of care could be transformed or augmented to reduce overall costs or at least reduce the rate of increase. Findings from COME HOME have helped inform the OCM program.11
Over the same period, practices belonging to the USON were paving a path toward value. An electronic health record (EHR) for the entire network was adopted in 2005. A pathways program in which chemotherapy regimens were assessed on cost as well as benefits and toxicity, was started in 2006. Higher-cost regimens with no additional benefits comparable with other evidence-based regimens were deselected for initial treatment choices at the time of initial decision support. This process was streamlined using web-based technology that improved pathways compliance and tracking of off-pathways exceptions.12 Retrospective studies indicated that pathways had the potential to bend the cost curve by reducing drug spending.13,14 USON and its practices also tested a nurse call system (Innovent Oncology) funded by a monthly management fee. This program guided patients through chemotherapy with regular telephonic symptom assessment and discussion of patient-centered values and advance care planning. Results of these programs indicated relative reductions in both drug and hospital expenses.15
Additional experience has come from participation in the United Healthcare Episodes of Care (EOC) initiative, which eliminated the chemotherapy drug incentives, compensating physicians on a per-episode basis instead. This study showed a significant reduction in the total cost of cancer therapy after modifying the fee-for-service system and incorporating feedback data and financial incentives to reward improved outcomes and cost efficiency.16
The Oncology Care Model represents a convergence of purchaser demand and provider readiness. The purchaser holds providers accountable for cost and quality. The data on outcomes and costs will provide an extensive database that can be analyzed by the participating practices to address variations and reduce unnecessary care and preventable costs. Best practices are rewarded.17
The OCM and practice readiness
As a part of the CMS proposal process, practices were required to submit implementation plans by June 30, 2015. The purpose of the implementation plan was to define how a practice could transform over 6 broad domains: 24/7 coverage; EHR certification; navigation and care coordination; continuous quality improvement; incorporation of the Institute of Medicine’s (IOM’s) Care Management Plan; and adherence to nationally recognized guidelines. The periods of patient eligibility for the program were 6-month treatment episodes triggered by a cancer diagnosis, a provider encounter claim, and a Part B or D drug claim specifically identified as a cancer treatment. The episodes could be repeated if the 3 criteria continued to be met. All charges continued to be billed as fee-for-service as before, but in addition, participating practices could bill a monthly enhanced oncology services (MEOS) payment for the duration of an episode. Reducing the total cost of care while meeting performance metrics thresholds would also qualify a practice for performance-based payments.
Of the primary components, EHR certification and adherence to guidelines had been addressed previously, but the other domains represented significant challenges. Although 24/7 physician coverage with access to an EHR is standard for all practices, most practice sites do not have an insight into the frequency of hospital admissions, the ability to efficiently add sick patients to the daily schedule, or a routine call system to assess chemotherapy toxicity.
The OCM proposes 10 potential navigation/care coordination functions (Table 1) and does not consider those functions to be the role of one person, but rather a team responsibility. Most of our practices perform at least some of these functions, but they are not formally designated, coordinated, or recorded. A similar condition exists for the IOM care plan, which includes recommendations for treatment and adverse event management (Table 2). The prognosis, toxicity, quality-of-life, and goals-of-care requirements are often found in the physician notes, but not systematically documented or searchable.
Similar challenges have been observed for continuous quality improvement programs. Although the data are available, they are often not easy to search and, therefore, are difficult to retrieve and report. The OCM, as with any transformational program, must always weigh the benefit of information with the burden of consumption of physician and staff time to collect and input these data.18
Prepilot project work
In October 2015, lead physicians and managers from the 12 participating practices were brought together with analytic, technical, process management, and business experts from USON and McKesson Specialty Health. The objective of the meeting was to define the areas of greatest need for day 1 of the OCM and to be prepared. The challenges were to identify the changes needed to meet the requirements of the OCM while improving the patient experience, sustaining the viability of the community oncology practices, creating teams to deliver more effective care, and using data to bend the cost curve. Accordingly, 4 work streams were created: Care and Support; Content; Technology; and Communications, Revenue Cycle and Incentives.
Care and Support
The key tasks of the Care and Support team were to define the workflows for navigation and the IOM care plan. As a patient’s journey through the clinic was mapped out, it became clear that although multiple personnel could participate in the navigation and care plans, there was no systematic way to organize and record the components of successful navigation. The goals for the pilot were to test various options for navigation and to identify best practices that could be translated into standard operating procedures.
Content
The Content team was charged with identifying available programs that would fit into the OCM requirements. These included advance care planning, survivorship, chemotherapy teaching, risk assessment, pathways, and symptom assessment. A longer-term goal was the development of care paths, a more comprehensive map of the patient’s journey that would include consultations, coordinated care, imaging, labs, and other services.
Technology
The task for the Technology work stream was to identify processes of care that required documentation and to evaluate current and future technology solutions to improve efficiencies. The electronic medical record satisfied for the input of data with relevant clinical details, demographics, disease types, and staging. A web-based pathways tool supported clinical decision-making, as well as compliance to pathways. The Medicare quality metric programs set the stage for development of capture and reporting tools for data from many sources. The pilot would indicate the adequacy of these tools and the need for expansion or development of new functions or programs. Of particular importance was recording the IOM care plan and navigation functions in a searchable format. As care paths are developed, risk prediction, palliative care, and other services need to be encompassed. Finally, technology will support the identification and enrollment of eligible patients, and billing activities.
Communications, Revenue Cycle, and Incentives
The final work stream was Communications, Revenue Cycle, and Incentives. For the pilot, the focus was on revenue cycle. A new category of patient needed to be identified, enrolled, and billed to CMS for services. At the outset, the technology did not address the identification of patients receiving only oral drugs. The office visit, the diagnosis, and the drug claim all had to be aligned for enrollment and billing. It was critical to understand the workload by patient and total volume to estimate the technology and personnel needs to meet the initial number of new OCM patients. Communication refers to both internal and external parties. Education of the entire practice staff regarding transformation will be critical for success.
Of the 12 participating practices, 3 practice sites were selected for the pilot program. Each had fewer than 10 medical oncologists and at least 1 radiation oncologist. Each site had a physician champion and an administrative lead. All of the sites were part of a larger regional oncology practice. A fourth site had independently started a pilot and that experience was shared with the larger group as well. The sites were distributed across the country in 4 different time zones.
The pilot experience
The pilot experience yielded important findings, some expected and some unexpected. The challenges of navigation, the treatment plan, and team building were anticipated. We were surprised at the sheer number of potential candidates and the difficulty in finding eligible candidates. Not to be overlooked was a need for continued and possibly increased emphasis on adherence to pathways and process changes to reduce hospitalizations and emergency department (ED) visits.
Navigation
At the outset, none of the pilot practices had formal navigation processes as outlined in Table 1. Many of the processes, such as coordinating appointments and facilitating follow-up services and financial support, were provided by the practice, but were not identified or coordinated as navigation. The practices, as a first step, defined who was responsible for those services and identified 1 person who would be responsible for their completion. It was agreed that navigation was a process shared by a team and not an individual responsibility, yet the person who would monitor the completion of the tasks was not identified. It soon became apparent that true navigation included more tasks than initially outlined.
Additional tasks included appropriate patient education regarding treatment toxicities, follow-up after chemotherapy or a hospitalization, and coordination of other aspects of the IOM care plan, such as survivorship and advance care planning. Each of the practices recruited staff internally to assume the navigator role, and standard operating procedures were developed for completing and documenting this expanded responsibility. True navigation, however, depends on building the team character while still having 1 or 2 members of the team identified as being responsible for following and documenting the patient’s journey through an episode. To meet those needs, navigators developed ad hoc methods, such as spreadsheets, to track patients. The technology team developed drop-down check lists within the EHR, but the burden of documentation continued. Lastly, an ongoing challenge is how best to designate responsibility and assess how many additional staffers are needed.
IOM Care Management Plan
Before initiation of the pilot project, no practice was providing patients with a comprehensive, written treatment plan. Considerably more than half of the members of the work-stream teams believed that would be difficult to implement. However, the members of the Care and Support work stream made some fundamental assumptions to make the care plan workable: first, all aspects of the plan did not occur at the same time, and were not completed by the same person; second, and critically, items 2-9 of Table 2 could be completed at one time during the early conversations between the physician and patient about the goals of treatment. Diagnosis, prognosis, treatment intent, response rate, quality of life, and toxicities were included in the treatment plan, and the remaining IOM care plan could be discussed, or at least identified, as issues for further discussion with other team members. These components were incorporated into a 1-page document that was either typed into the record and printed out or handwritten and copied for the patient. This became the treatment plan and was ready for use at the start of the pilot.
The physician response to the treatment plan was mixed. One site adopted it enthusiastically and quickly moved to use the plan for all patients. Other sites had variable uptake. One hurdle was defining response rate to therapy and prognosis. Data were provided but they often did not match the conditions of individual patients. Some physicians were uncomfortable with the process. Documentation was difficult because the plans had to be scanned into the EHR. Patients generally responded favorably to the plans and would bring them to teaching or chemotherapy sessions.
As with navigation, the treatment plan challenges pointed the technology team toward the development and implementation of an electronic version of the plan. The pilot allowed members of the technology team to visit the clinics, to evaluate workflows and make assumptions on how to structure a treatment plan electronically.
Team-based care
None of the pilot sites had a formalized structure for team-based care. Team huddles were developed and weekly and daily huddles were encouraged. The weekly huddles took about 15-30 minutes, during which patients scheduled for the coming week were reviewed. All personnel who saw patients were invited – benefit counselors, advanced practice providers, schedulers, lab technicians, medical assistants, office and infusion nurses, social workers, pharmacists, physicians, and lead administrative staff. The daily huddle was smaller and generally included a nurse, a medical assistant, and a physician, at a minimum, to review the patients in the hospital, those to be seen in the clinic that day, and any follow-up information based on scheduled contact following recent treatments or events. In some sites, these huddles were uniformly endorsed, in others, not at all. Although many physicians felt that the functions were being handled informally and the additional time commitment would not improve the process, once they began to attend the meetings, they appreciated the value of the huddles and continued attending them. As the complexity of delivery and documentation becomes more apparent, these will prove indispensable to coordinated care.
24/7 access
Hospitalization is one of the chief drivers of the total cost of care, so the pilot sites were concerned that more needed to be done to reduce unnecessary hospitalizations. One site surveyed the patients coming into the clinic about their previous ED visits. Many of the visits had been for noncancer-related events and the clinic was not aware of many of the many of the visits. These findings prompted a number of changes. Open slots were created daily for patients who needed to be worked in for any areas of concern. The on-call physician, triage nurse, or navigation lead could fill these slots. All patients discharged from the hospital were called within 48 hours after discharge and scheduled for a clinic visit within 1 week. Night and weekend call logs were scrutinized each morning and patients’ calls were returned for any issues related to symptom or toxicity management. At one site, patients were given wallet cards with the clinic number, the treatment regimen, and, on the reverse, all symptoms that would justify calling the clinic. The patients were encouraged to call the clinic earlier rather than later in the day. On the back end, the clinics were to have processes in place so that patient calls would be answered quickly to facilitate same-day evaluations in the clinic.
Enrollment and revenue cycle
The most intractable problem was the identification and enrollment of OCM patients. As already noted, 3 components were necessary for enrollment: a drug charge for Part B or D Medicare, a provider visit, and an approved cancer diagnosis. To identify those patients, the claims system would churn out a weekly list of all eligible patients. However, the claims system had no mechanism to pick up Part D claims for oral medications. This meant that any patient with a provider visit and an appropriate diagnosis was potentially eligible for enrollment. At one site, the list of potential patients was 2-3 times the number of actual candidates. It took 6 weeks of manual chart review to resolving the list. Collectively, the 12 practices could have as many as 20,000 patients eligible for the July 1 enrollment. The pilot allowed the practices to get an early start on recruitment of business office staff and plans to address the backlog of potentially eligible patients. The process of identifying eligible patients for the OCM still needs a better solution because finding the appropriate patients is a critical first step in this model.
Underlying all of these initiatives is communication, both internal and external. We have to select and celebrate best practices. We have to educate our staffs. We will have to demonstrate that we are giving better care to our patients by using patient and provider testimonials and data.
From angst to analytics
The challenges of practice transformation can be daunting. It will be difficult to formalize processes and document data in ways that were untested before the pilot program was set up. However, the pilot accomplished 2 things: it identified additional areas that needed improvement and it demonstrated that the most challenging aspects of the OCM were feasible. Navigation and the IOM care plan were broken down into parts; each component was separately addressed, and programs were put in place to make the pieces manageable and part of an overall movement toward team-based care. The addition of a technology platform has been a key factor for the success of the value-based care initiative. Additional technology support has been enlisted to facilitate the processes, and an electronic version of the treatment plan is being tested. More difficult will be efforts to address the cultural resistance to change, which we hope to do by using data and outcomes from the CMS claims data files. The OCM represents an unprecedented opportunity for measurement of the quality of care we deliver.
We are now well und
Acknowledgment
The authors thank Supriya Srinivasan, PhD, for editorial support.
1. Oncology Care Model. Centers for Medicare and Medicaid Services 2016. https://innovation.cms.gov/initiatives/oncology-care/. Last updated November 14, 2017. Accessed November 16 2016.
2. Mortimer L, Strawbridge L, Lukens E, et al. CMS’ Oncology Care Model: delivering higher value cancer care. Clin Pharmacol Ther. 2017.
3. Medicare Prescription Drug, Improvement and Modernization Act of 2003, Pub Law No. 108-173; 2003.
4. Mathews D. The sequester: absolutely everything you could possibly need to know, in one FAQ. https://www.washingtonpost.com/news/wonk/wp/2013/02/20/the-sequester-absolutely-everything-you-could-possibly-need-to-know-in-one-faq/?utm_term=.a0f3a768399b. Published February 20, 2013. Accessed December 4, 2017.
5. Community Oncology Alliance. 2016 community oncology practice impact report: tracking the changing landscape of cancer care. https://www.communityoncology.org/wp-content/uploads/2016/09/PracticeImpactReport-2016-Report.pdf. Issued October 4, 2016. Accessed April 10, 2017.
6. The Patient Protection and Affordable Care Act, Pub Law No. 111-148; 2010.
7. Centers for Medicare and Medicaid Services. Shared Savings Program 2016. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/sharedsavingsprogram/index.html?redirect=/sharedsavingsprogram/. Last modified October 12, 2017. Accessed November 16 2016.
8. Dale SB, Ghosh A, Peikes DN, et al. Two-year costs and quality in the comprehensive primary care initiative. N Engl J Med. 2016;374:2345-2356.
9. McWilliams JM, Chernew ME, Landon BE, Schwartz AL. Performance differences in year 1 of pioneer accountable care organizations. N Engl J Med. 2015;372:1927-1936.
10. Rajkumar R, Press MJ, Conway PH. The CMS Innovation Center – a five-year self-assessment. N Engl J Med. 2015;372:1981-1983.
11. Waters TM, Webster JA, Stevens LA, et al. Community oncology medical homes: physician-driven change to improve patient care and reduce costs. J Oncol Pract. 2015;11(6):462-467.
12. Wilfong LS, Hoverman JR, Gosse N, Neubauer MA, Torres V. Changing physician compensation and implementing new technology to enhance pathways compliance. J Clin Oncol. 2016;34(Suppl 7S):Abstr 187.
13. Hoverman JR, Cartwright TH, Patt DA, et al. Pathways, outcomes, and costs in colon cancer: retrospective evaluations in two distinct databases. J Oncol Pract. 2011;7(3 Suppl):52s-59s.
14. Neubauer MA, Hoverman JR, Kolodziej M, et al. Cost effectiveness of evidence-based treatment guidelines for the treatment of non-small-cell lung cancer in the community setting. J Oncol Pract. 2010;6(1):12-18.
15. Hoverman JR, Klein I, Harrison DW, et al. Opening the black box: the impact of an oncology management program consisting of level I pathways and an outbound nurse call system. J Oncol Pract. 2014;10(1):63-67.
16. Newcomer LN, Gould B, Page RD, Donelan SA, Perkins M. Changing physician incentives for affordable, quality cancer care: results of an episode payment model. J Oncol Pract. 2014;10(5):322-326.
17. Meyer JA, Rybowski L, Eichler R. Theory and reality of value-based purchasing: lessons from the pioneers. Rockville, MD.: Agency for Health Care Policy and Research; 1997.
18. Stein CM. Academic clinical research: death by a thousand clicks. Sci Transl Med. 2015;7:318fs349.
19. Churchill W. The end of the beginning. 1st ed. Boston, MA: Little Brown & Co; 1943.
In March 2016, 13 practices affiliated with the US Oncology Network (USON) were invited to participate in the Oncology Care Model (OCM) proposed by the Center for Medicare and Medicaid Services (CMS) and Center for Medicare and Medicaid Innovation (CMMI). The OCM, a novel value-based care model, was designed to provide higher-quality and better-coordinated oncology care at a lower cost to CMS.1 Of the 13 practices, 12 agreed to participate with a start date for the program of July 1, 2016. At least 40% of the practices’ patients were insured by Medicare, and any eligible patients with active cancer were offered an opportunity to enter the program. USON practices treat more than 25,000 patients with a qualifying episode per year and the overall OCM program sees more than 150,000 beneficiaries per year,2 so we anticipated that the OCM would have a substantial impact on each of the 12 practices on USON.
Faced with the scenario of having only 3 months between notification of approval and launch of the OCM, it was imperative that all the practices be proactive in planning and preparing to launch the OCM. With this goal in mind, representatives from all OCM candidate practices convened to anticipate the needs of the OCM and chart out a program to meet those needs. In this article, we discuss the requirements and scope of the OCM, the development of an internal pilot project, the anticipated gains from the pilot, and the results and findings from the pilot, both expected and unexpected.
The road to the Oncology Care Model
The government and oncology practices have been on separate trajectories to the OCM. In the last 15 years, the major intersections of these trajectories had to do with price and not patient outcomes. In 2003, the Medicare Prescription Drug Improvement and Modernization Act (MMA) focused on drug price reductions from an average wholesale price–based schedule to an average sales price–based schedule.3 There was the sequester in 2013,4 and more recently a proposal to restructure the payment for Part B drugs. In the background, recurrent negotiations to fix the calculation for the sustainable growth rate allowed for periodic draconian cuts to the prices of services. The cumulative effect of these price reductions has been to put economic pressure on community oncologists such that many have moved to a hospital environment.5
This contentious relationship with community oncology began to change with the passage of the Affordable Care Act (ACA) in 2010.6 Section 3022 of the ACA established the Medicare Shared Savings Program (MSSP) with the charge to create a new type of health care entity that was responsible for achieving the triple aim of improving population health, improving individual patient care, and bending the cost curve.7 Additional programs, such as the Pioneer Accountable Care Organization (ACO) program and the Comprehensive Primary Care Initiative were established to test alternative payment models.8-10
The ACA also funded the CMMI with a mandate to “test innovative payment and service delivery models” to achieve the triple aim; US$10 billion were appropriated for the years 2011-2019 for this purpose. The CMMI funded a pilot project for cancer care, the COME HOME [Community Oncology MEdical HOME] initiative, to test whether some aspects of care could be transformed or augmented to reduce overall costs or at least reduce the rate of increase. Findings from COME HOME have helped inform the OCM program.11
Over the same period, practices belonging to the USON were paving a path toward value. An electronic health record (EHR) for the entire network was adopted in 2005. A pathways program in which chemotherapy regimens were assessed on cost as well as benefits and toxicity, was started in 2006. Higher-cost regimens with no additional benefits comparable with other evidence-based regimens were deselected for initial treatment choices at the time of initial decision support. This process was streamlined using web-based technology that improved pathways compliance and tracking of off-pathways exceptions.12 Retrospective studies indicated that pathways had the potential to bend the cost curve by reducing drug spending.13,14 USON and its practices also tested a nurse call system (Innovent Oncology) funded by a monthly management fee. This program guided patients through chemotherapy with regular telephonic symptom assessment and discussion of patient-centered values and advance care planning. Results of these programs indicated relative reductions in both drug and hospital expenses.15
Additional experience has come from participation in the United Healthcare Episodes of Care (EOC) initiative, which eliminated the chemotherapy drug incentives, compensating physicians on a per-episode basis instead. This study showed a significant reduction in the total cost of cancer therapy after modifying the fee-for-service system and incorporating feedback data and financial incentives to reward improved outcomes and cost efficiency.16
The Oncology Care Model represents a convergence of purchaser demand and provider readiness. The purchaser holds providers accountable for cost and quality. The data on outcomes and costs will provide an extensive database that can be analyzed by the participating practices to address variations and reduce unnecessary care and preventable costs. Best practices are rewarded.17
The OCM and practice readiness
As a part of the CMS proposal process, practices were required to submit implementation plans by June 30, 2015. The purpose of the implementation plan was to define how a practice could transform over 6 broad domains: 24/7 coverage; EHR certification; navigation and care coordination; continuous quality improvement; incorporation of the Institute of Medicine’s (IOM’s) Care Management Plan; and adherence to nationally recognized guidelines. The periods of patient eligibility for the program were 6-month treatment episodes triggered by a cancer diagnosis, a provider encounter claim, and a Part B or D drug claim specifically identified as a cancer treatment. The episodes could be repeated if the 3 criteria continued to be met. All charges continued to be billed as fee-for-service as before, but in addition, participating practices could bill a monthly enhanced oncology services (MEOS) payment for the duration of an episode. Reducing the total cost of care while meeting performance metrics thresholds would also qualify a practice for performance-based payments.
Of the primary components, EHR certification and adherence to guidelines had been addressed previously, but the other domains represented significant challenges. Although 24/7 physician coverage with access to an EHR is standard for all practices, most practice sites do not have an insight into the frequency of hospital admissions, the ability to efficiently add sick patients to the daily schedule, or a routine call system to assess chemotherapy toxicity.
The OCM proposes 10 potential navigation/care coordination functions (Table 1) and does not consider those functions to be the role of one person, but rather a team responsibility. Most of our practices perform at least some of these functions, but they are not formally designated, coordinated, or recorded. A similar condition exists for the IOM care plan, which includes recommendations for treatment and adverse event management (Table 2). The prognosis, toxicity, quality-of-life, and goals-of-care requirements are often found in the physician notes, but not systematically documented or searchable.
Similar challenges have been observed for continuous quality improvement programs. Although the data are available, they are often not easy to search and, therefore, are difficult to retrieve and report. The OCM, as with any transformational program, must always weigh the benefit of information with the burden of consumption of physician and staff time to collect and input these data.18
Prepilot project work
In October 2015, lead physicians and managers from the 12 participating practices were brought together with analytic, technical, process management, and business experts from USON and McKesson Specialty Health. The objective of the meeting was to define the areas of greatest need for day 1 of the OCM and to be prepared. The challenges were to identify the changes needed to meet the requirements of the OCM while improving the patient experience, sustaining the viability of the community oncology practices, creating teams to deliver more effective care, and using data to bend the cost curve. Accordingly, 4 work streams were created: Care and Support; Content; Technology; and Communications, Revenue Cycle and Incentives.
Care and Support
The key tasks of the Care and Support team were to define the workflows for navigation and the IOM care plan. As a patient’s journey through the clinic was mapped out, it became clear that although multiple personnel could participate in the navigation and care plans, there was no systematic way to organize and record the components of successful navigation. The goals for the pilot were to test various options for navigation and to identify best practices that could be translated into standard operating procedures.
Content
The Content team was charged with identifying available programs that would fit into the OCM requirements. These included advance care planning, survivorship, chemotherapy teaching, risk assessment, pathways, and symptom assessment. A longer-term goal was the development of care paths, a more comprehensive map of the patient’s journey that would include consultations, coordinated care, imaging, labs, and other services.
Technology
The task for the Technology work stream was to identify processes of care that required documentation and to evaluate current and future technology solutions to improve efficiencies. The electronic medical record satisfied for the input of data with relevant clinical details, demographics, disease types, and staging. A web-based pathways tool supported clinical decision-making, as well as compliance to pathways. The Medicare quality metric programs set the stage for development of capture and reporting tools for data from many sources. The pilot would indicate the adequacy of these tools and the need for expansion or development of new functions or programs. Of particular importance was recording the IOM care plan and navigation functions in a searchable format. As care paths are developed, risk prediction, palliative care, and other services need to be encompassed. Finally, technology will support the identification and enrollment of eligible patients, and billing activities.
Communications, Revenue Cycle, and Incentives
The final work stream was Communications, Revenue Cycle, and Incentives. For the pilot, the focus was on revenue cycle. A new category of patient needed to be identified, enrolled, and billed to CMS for services. At the outset, the technology did not address the identification of patients receiving only oral drugs. The office visit, the diagnosis, and the drug claim all had to be aligned for enrollment and billing. It was critical to understand the workload by patient and total volume to estimate the technology and personnel needs to meet the initial number of new OCM patients. Communication refers to both internal and external parties. Education of the entire practice staff regarding transformation will be critical for success.
Of the 12 participating practices, 3 practice sites were selected for the pilot program. Each had fewer than 10 medical oncologists and at least 1 radiation oncologist. Each site had a physician champion and an administrative lead. All of the sites were part of a larger regional oncology practice. A fourth site had independently started a pilot and that experience was shared with the larger group as well. The sites were distributed across the country in 4 different time zones.
The pilot experience
The pilot experience yielded important findings, some expected and some unexpected. The challenges of navigation, the treatment plan, and team building were anticipated. We were surprised at the sheer number of potential candidates and the difficulty in finding eligible candidates. Not to be overlooked was a need for continued and possibly increased emphasis on adherence to pathways and process changes to reduce hospitalizations and emergency department (ED) visits.
Navigation
At the outset, none of the pilot practices had formal navigation processes as outlined in Table 1. Many of the processes, such as coordinating appointments and facilitating follow-up services and financial support, were provided by the practice, but were not identified or coordinated as navigation. The practices, as a first step, defined who was responsible for those services and identified 1 person who would be responsible for their completion. It was agreed that navigation was a process shared by a team and not an individual responsibility, yet the person who would monitor the completion of the tasks was not identified. It soon became apparent that true navigation included more tasks than initially outlined.
Additional tasks included appropriate patient education regarding treatment toxicities, follow-up after chemotherapy or a hospitalization, and coordination of other aspects of the IOM care plan, such as survivorship and advance care planning. Each of the practices recruited staff internally to assume the navigator role, and standard operating procedures were developed for completing and documenting this expanded responsibility. True navigation, however, depends on building the team character while still having 1 or 2 members of the team identified as being responsible for following and documenting the patient’s journey through an episode. To meet those needs, navigators developed ad hoc methods, such as spreadsheets, to track patients. The technology team developed drop-down check lists within the EHR, but the burden of documentation continued. Lastly, an ongoing challenge is how best to designate responsibility and assess how many additional staffers are needed.
IOM Care Management Plan
Before initiation of the pilot project, no practice was providing patients with a comprehensive, written treatment plan. Considerably more than half of the members of the work-stream teams believed that would be difficult to implement. However, the members of the Care and Support work stream made some fundamental assumptions to make the care plan workable: first, all aspects of the plan did not occur at the same time, and were not completed by the same person; second, and critically, items 2-9 of Table 2 could be completed at one time during the early conversations between the physician and patient about the goals of treatment. Diagnosis, prognosis, treatment intent, response rate, quality of life, and toxicities were included in the treatment plan, and the remaining IOM care plan could be discussed, or at least identified, as issues for further discussion with other team members. These components were incorporated into a 1-page document that was either typed into the record and printed out or handwritten and copied for the patient. This became the treatment plan and was ready for use at the start of the pilot.
The physician response to the treatment plan was mixed. One site adopted it enthusiastically and quickly moved to use the plan for all patients. Other sites had variable uptake. One hurdle was defining response rate to therapy and prognosis. Data were provided but they often did not match the conditions of individual patients. Some physicians were uncomfortable with the process. Documentation was difficult because the plans had to be scanned into the EHR. Patients generally responded favorably to the plans and would bring them to teaching or chemotherapy sessions.
As with navigation, the treatment plan challenges pointed the technology team toward the development and implementation of an electronic version of the plan. The pilot allowed members of the technology team to visit the clinics, to evaluate workflows and make assumptions on how to structure a treatment plan electronically.
Team-based care
None of the pilot sites had a formalized structure for team-based care. Team huddles were developed and weekly and daily huddles were encouraged. The weekly huddles took about 15-30 minutes, during which patients scheduled for the coming week were reviewed. All personnel who saw patients were invited – benefit counselors, advanced practice providers, schedulers, lab technicians, medical assistants, office and infusion nurses, social workers, pharmacists, physicians, and lead administrative staff. The daily huddle was smaller and generally included a nurse, a medical assistant, and a physician, at a minimum, to review the patients in the hospital, those to be seen in the clinic that day, and any follow-up information based on scheduled contact following recent treatments or events. In some sites, these huddles were uniformly endorsed, in others, not at all. Although many physicians felt that the functions were being handled informally and the additional time commitment would not improve the process, once they began to attend the meetings, they appreciated the value of the huddles and continued attending them. As the complexity of delivery and documentation becomes more apparent, these will prove indispensable to coordinated care.
24/7 access
Hospitalization is one of the chief drivers of the total cost of care, so the pilot sites were concerned that more needed to be done to reduce unnecessary hospitalizations. One site surveyed the patients coming into the clinic about their previous ED visits. Many of the visits had been for noncancer-related events and the clinic was not aware of many of the many of the visits. These findings prompted a number of changes. Open slots were created daily for patients who needed to be worked in for any areas of concern. The on-call physician, triage nurse, or navigation lead could fill these slots. All patients discharged from the hospital were called within 48 hours after discharge and scheduled for a clinic visit within 1 week. Night and weekend call logs were scrutinized each morning and patients’ calls were returned for any issues related to symptom or toxicity management. At one site, patients were given wallet cards with the clinic number, the treatment regimen, and, on the reverse, all symptoms that would justify calling the clinic. The patients were encouraged to call the clinic earlier rather than later in the day. On the back end, the clinics were to have processes in place so that patient calls would be answered quickly to facilitate same-day evaluations in the clinic.
Enrollment and revenue cycle
The most intractable problem was the identification and enrollment of OCM patients. As already noted, 3 components were necessary for enrollment: a drug charge for Part B or D Medicare, a provider visit, and an approved cancer diagnosis. To identify those patients, the claims system would churn out a weekly list of all eligible patients. However, the claims system had no mechanism to pick up Part D claims for oral medications. This meant that any patient with a provider visit and an appropriate diagnosis was potentially eligible for enrollment. At one site, the list of potential patients was 2-3 times the number of actual candidates. It took 6 weeks of manual chart review to resolving the list. Collectively, the 12 practices could have as many as 20,000 patients eligible for the July 1 enrollment. The pilot allowed the practices to get an early start on recruitment of business office staff and plans to address the backlog of potentially eligible patients. The process of identifying eligible patients for the OCM still needs a better solution because finding the appropriate patients is a critical first step in this model.
Underlying all of these initiatives is communication, both internal and external. We have to select and celebrate best practices. We have to educate our staffs. We will have to demonstrate that we are giving better care to our patients by using patient and provider testimonials and data.
From angst to analytics
The challenges of practice transformation can be daunting. It will be difficult to formalize processes and document data in ways that were untested before the pilot program was set up. However, the pilot accomplished 2 things: it identified additional areas that needed improvement and it demonstrated that the most challenging aspects of the OCM were feasible. Navigation and the IOM care plan were broken down into parts; each component was separately addressed, and programs were put in place to make the pieces manageable and part of an overall movement toward team-based care. The addition of a technology platform has been a key factor for the success of the value-based care initiative. Additional technology support has been enlisted to facilitate the processes, and an electronic version of the treatment plan is being tested. More difficult will be efforts to address the cultural resistance to change, which we hope to do by using data and outcomes from the CMS claims data files. The OCM represents an unprecedented opportunity for measurement of the quality of care we deliver.
We are now well und
Acknowledgment
The authors thank Supriya Srinivasan, PhD, for editorial support.
In March 2016, 13 practices affiliated with the US Oncology Network (USON) were invited to participate in the Oncology Care Model (OCM) proposed by the Center for Medicare and Medicaid Services (CMS) and Center for Medicare and Medicaid Innovation (CMMI). The OCM, a novel value-based care model, was designed to provide higher-quality and better-coordinated oncology care at a lower cost to CMS.1 Of the 13 practices, 12 agreed to participate with a start date for the program of July 1, 2016. At least 40% of the practices’ patients were insured by Medicare, and any eligible patients with active cancer were offered an opportunity to enter the program. USON practices treat more than 25,000 patients with a qualifying episode per year and the overall OCM program sees more than 150,000 beneficiaries per year,2 so we anticipated that the OCM would have a substantial impact on each of the 12 practices on USON.
Faced with the scenario of having only 3 months between notification of approval and launch of the OCM, it was imperative that all the practices be proactive in planning and preparing to launch the OCM. With this goal in mind, representatives from all OCM candidate practices convened to anticipate the needs of the OCM and chart out a program to meet those needs. In this article, we discuss the requirements and scope of the OCM, the development of an internal pilot project, the anticipated gains from the pilot, and the results and findings from the pilot, both expected and unexpected.
The road to the Oncology Care Model
The government and oncology practices have been on separate trajectories to the OCM. In the last 15 years, the major intersections of these trajectories had to do with price and not patient outcomes. In 2003, the Medicare Prescription Drug Improvement and Modernization Act (MMA) focused on drug price reductions from an average wholesale price–based schedule to an average sales price–based schedule.3 There was the sequester in 2013,4 and more recently a proposal to restructure the payment for Part B drugs. In the background, recurrent negotiations to fix the calculation for the sustainable growth rate allowed for periodic draconian cuts to the prices of services. The cumulative effect of these price reductions has been to put economic pressure on community oncologists such that many have moved to a hospital environment.5
This contentious relationship with community oncology began to change with the passage of the Affordable Care Act (ACA) in 2010.6 Section 3022 of the ACA established the Medicare Shared Savings Program (MSSP) with the charge to create a new type of health care entity that was responsible for achieving the triple aim of improving population health, improving individual patient care, and bending the cost curve.7 Additional programs, such as the Pioneer Accountable Care Organization (ACO) program and the Comprehensive Primary Care Initiative were established to test alternative payment models.8-10
The ACA also funded the CMMI with a mandate to “test innovative payment and service delivery models” to achieve the triple aim; US$10 billion were appropriated for the years 2011-2019 for this purpose. The CMMI funded a pilot project for cancer care, the COME HOME [Community Oncology MEdical HOME] initiative, to test whether some aspects of care could be transformed or augmented to reduce overall costs or at least reduce the rate of increase. Findings from COME HOME have helped inform the OCM program.11
Over the same period, practices belonging to the USON were paving a path toward value. An electronic health record (EHR) for the entire network was adopted in 2005. A pathways program in which chemotherapy regimens were assessed on cost as well as benefits and toxicity, was started in 2006. Higher-cost regimens with no additional benefits comparable with other evidence-based regimens were deselected for initial treatment choices at the time of initial decision support. This process was streamlined using web-based technology that improved pathways compliance and tracking of off-pathways exceptions.12 Retrospective studies indicated that pathways had the potential to bend the cost curve by reducing drug spending.13,14 USON and its practices also tested a nurse call system (Innovent Oncology) funded by a monthly management fee. This program guided patients through chemotherapy with regular telephonic symptom assessment and discussion of patient-centered values and advance care planning. Results of these programs indicated relative reductions in both drug and hospital expenses.15
Additional experience has come from participation in the United Healthcare Episodes of Care (EOC) initiative, which eliminated the chemotherapy drug incentives, compensating physicians on a per-episode basis instead. This study showed a significant reduction in the total cost of cancer therapy after modifying the fee-for-service system and incorporating feedback data and financial incentives to reward improved outcomes and cost efficiency.16
The Oncology Care Model represents a convergence of purchaser demand and provider readiness. The purchaser holds providers accountable for cost and quality. The data on outcomes and costs will provide an extensive database that can be analyzed by the participating practices to address variations and reduce unnecessary care and preventable costs. Best practices are rewarded.17
The OCM and practice readiness
As a part of the CMS proposal process, practices were required to submit implementation plans by June 30, 2015. The purpose of the implementation plan was to define how a practice could transform over 6 broad domains: 24/7 coverage; EHR certification; navigation and care coordination; continuous quality improvement; incorporation of the Institute of Medicine’s (IOM’s) Care Management Plan; and adherence to nationally recognized guidelines. The periods of patient eligibility for the program were 6-month treatment episodes triggered by a cancer diagnosis, a provider encounter claim, and a Part B or D drug claim specifically identified as a cancer treatment. The episodes could be repeated if the 3 criteria continued to be met. All charges continued to be billed as fee-for-service as before, but in addition, participating practices could bill a monthly enhanced oncology services (MEOS) payment for the duration of an episode. Reducing the total cost of care while meeting performance metrics thresholds would also qualify a practice for performance-based payments.
Of the primary components, EHR certification and adherence to guidelines had been addressed previously, but the other domains represented significant challenges. Although 24/7 physician coverage with access to an EHR is standard for all practices, most practice sites do not have an insight into the frequency of hospital admissions, the ability to efficiently add sick patients to the daily schedule, or a routine call system to assess chemotherapy toxicity.
The OCM proposes 10 potential navigation/care coordination functions (Table 1) and does not consider those functions to be the role of one person, but rather a team responsibility. Most of our practices perform at least some of these functions, but they are not formally designated, coordinated, or recorded. A similar condition exists for the IOM care plan, which includes recommendations for treatment and adverse event management (Table 2). The prognosis, toxicity, quality-of-life, and goals-of-care requirements are often found in the physician notes, but not systematically documented or searchable.
Similar challenges have been observed for continuous quality improvement programs. Although the data are available, they are often not easy to search and, therefore, are difficult to retrieve and report. The OCM, as with any transformational program, must always weigh the benefit of information with the burden of consumption of physician and staff time to collect and input these data.18
Prepilot project work
In October 2015, lead physicians and managers from the 12 participating practices were brought together with analytic, technical, process management, and business experts from USON and McKesson Specialty Health. The objective of the meeting was to define the areas of greatest need for day 1 of the OCM and to be prepared. The challenges were to identify the changes needed to meet the requirements of the OCM while improving the patient experience, sustaining the viability of the community oncology practices, creating teams to deliver more effective care, and using data to bend the cost curve. Accordingly, 4 work streams were created: Care and Support; Content; Technology; and Communications, Revenue Cycle and Incentives.
Care and Support
The key tasks of the Care and Support team were to define the workflows for navigation and the IOM care plan. As a patient’s journey through the clinic was mapped out, it became clear that although multiple personnel could participate in the navigation and care plans, there was no systematic way to organize and record the components of successful navigation. The goals for the pilot were to test various options for navigation and to identify best practices that could be translated into standard operating procedures.
Content
The Content team was charged with identifying available programs that would fit into the OCM requirements. These included advance care planning, survivorship, chemotherapy teaching, risk assessment, pathways, and symptom assessment. A longer-term goal was the development of care paths, a more comprehensive map of the patient’s journey that would include consultations, coordinated care, imaging, labs, and other services.
Technology
The task for the Technology work stream was to identify processes of care that required documentation and to evaluate current and future technology solutions to improve efficiencies. The electronic medical record satisfied for the input of data with relevant clinical details, demographics, disease types, and staging. A web-based pathways tool supported clinical decision-making, as well as compliance to pathways. The Medicare quality metric programs set the stage for development of capture and reporting tools for data from many sources. The pilot would indicate the adequacy of these tools and the need for expansion or development of new functions or programs. Of particular importance was recording the IOM care plan and navigation functions in a searchable format. As care paths are developed, risk prediction, palliative care, and other services need to be encompassed. Finally, technology will support the identification and enrollment of eligible patients, and billing activities.
Communications, Revenue Cycle, and Incentives
The final work stream was Communications, Revenue Cycle, and Incentives. For the pilot, the focus was on revenue cycle. A new category of patient needed to be identified, enrolled, and billed to CMS for services. At the outset, the technology did not address the identification of patients receiving only oral drugs. The office visit, the diagnosis, and the drug claim all had to be aligned for enrollment and billing. It was critical to understand the workload by patient and total volume to estimate the technology and personnel needs to meet the initial number of new OCM patients. Communication refers to both internal and external parties. Education of the entire practice staff regarding transformation will be critical for success.
Of the 12 participating practices, 3 practice sites were selected for the pilot program. Each had fewer than 10 medical oncologists and at least 1 radiation oncologist. Each site had a physician champion and an administrative lead. All of the sites were part of a larger regional oncology practice. A fourth site had independently started a pilot and that experience was shared with the larger group as well. The sites were distributed across the country in 4 different time zones.
The pilot experience
The pilot experience yielded important findings, some expected and some unexpected. The challenges of navigation, the treatment plan, and team building were anticipated. We were surprised at the sheer number of potential candidates and the difficulty in finding eligible candidates. Not to be overlooked was a need for continued and possibly increased emphasis on adherence to pathways and process changes to reduce hospitalizations and emergency department (ED) visits.
Navigation
At the outset, none of the pilot practices had formal navigation processes as outlined in Table 1. Many of the processes, such as coordinating appointments and facilitating follow-up services and financial support, were provided by the practice, but were not identified or coordinated as navigation. The practices, as a first step, defined who was responsible for those services and identified 1 person who would be responsible for their completion. It was agreed that navigation was a process shared by a team and not an individual responsibility, yet the person who would monitor the completion of the tasks was not identified. It soon became apparent that true navigation included more tasks than initially outlined.
Additional tasks included appropriate patient education regarding treatment toxicities, follow-up after chemotherapy or a hospitalization, and coordination of other aspects of the IOM care plan, such as survivorship and advance care planning. Each of the practices recruited staff internally to assume the navigator role, and standard operating procedures were developed for completing and documenting this expanded responsibility. True navigation, however, depends on building the team character while still having 1 or 2 members of the team identified as being responsible for following and documenting the patient’s journey through an episode. To meet those needs, navigators developed ad hoc methods, such as spreadsheets, to track patients. The technology team developed drop-down check lists within the EHR, but the burden of documentation continued. Lastly, an ongoing challenge is how best to designate responsibility and assess how many additional staffers are needed.
IOM Care Management Plan
Before initiation of the pilot project, no practice was providing patients with a comprehensive, written treatment plan. Considerably more than half of the members of the work-stream teams believed that would be difficult to implement. However, the members of the Care and Support work stream made some fundamental assumptions to make the care plan workable: first, all aspects of the plan did not occur at the same time, and were not completed by the same person; second, and critically, items 2-9 of Table 2 could be completed at one time during the early conversations between the physician and patient about the goals of treatment. Diagnosis, prognosis, treatment intent, response rate, quality of life, and toxicities were included in the treatment plan, and the remaining IOM care plan could be discussed, or at least identified, as issues for further discussion with other team members. These components were incorporated into a 1-page document that was either typed into the record and printed out or handwritten and copied for the patient. This became the treatment plan and was ready for use at the start of the pilot.
The physician response to the treatment plan was mixed. One site adopted it enthusiastically and quickly moved to use the plan for all patients. Other sites had variable uptake. One hurdle was defining response rate to therapy and prognosis. Data were provided but they often did not match the conditions of individual patients. Some physicians were uncomfortable with the process. Documentation was difficult because the plans had to be scanned into the EHR. Patients generally responded favorably to the plans and would bring them to teaching or chemotherapy sessions.
As with navigation, the treatment plan challenges pointed the technology team toward the development and implementation of an electronic version of the plan. The pilot allowed members of the technology team to visit the clinics, to evaluate workflows and make assumptions on how to structure a treatment plan electronically.
Team-based care
None of the pilot sites had a formalized structure for team-based care. Team huddles were developed and weekly and daily huddles were encouraged. The weekly huddles took about 15-30 minutes, during which patients scheduled for the coming week were reviewed. All personnel who saw patients were invited – benefit counselors, advanced practice providers, schedulers, lab technicians, medical assistants, office and infusion nurses, social workers, pharmacists, physicians, and lead administrative staff. The daily huddle was smaller and generally included a nurse, a medical assistant, and a physician, at a minimum, to review the patients in the hospital, those to be seen in the clinic that day, and any follow-up information based on scheduled contact following recent treatments or events. In some sites, these huddles were uniformly endorsed, in others, not at all. Although many physicians felt that the functions were being handled informally and the additional time commitment would not improve the process, once they began to attend the meetings, they appreciated the value of the huddles and continued attending them. As the complexity of delivery and documentation becomes more apparent, these will prove indispensable to coordinated care.
24/7 access
Hospitalization is one of the chief drivers of the total cost of care, so the pilot sites were concerned that more needed to be done to reduce unnecessary hospitalizations. One site surveyed the patients coming into the clinic about their previous ED visits. Many of the visits had been for noncancer-related events and the clinic was not aware of many of the many of the visits. These findings prompted a number of changes. Open slots were created daily for patients who needed to be worked in for any areas of concern. The on-call physician, triage nurse, or navigation lead could fill these slots. All patients discharged from the hospital were called within 48 hours after discharge and scheduled for a clinic visit within 1 week. Night and weekend call logs were scrutinized each morning and patients’ calls were returned for any issues related to symptom or toxicity management. At one site, patients were given wallet cards with the clinic number, the treatment regimen, and, on the reverse, all symptoms that would justify calling the clinic. The patients were encouraged to call the clinic earlier rather than later in the day. On the back end, the clinics were to have processes in place so that patient calls would be answered quickly to facilitate same-day evaluations in the clinic.
Enrollment and revenue cycle
The most intractable problem was the identification and enrollment of OCM patients. As already noted, 3 components were necessary for enrollment: a drug charge for Part B or D Medicare, a provider visit, and an approved cancer diagnosis. To identify those patients, the claims system would churn out a weekly list of all eligible patients. However, the claims system had no mechanism to pick up Part D claims for oral medications. This meant that any patient with a provider visit and an appropriate diagnosis was potentially eligible for enrollment. At one site, the list of potential patients was 2-3 times the number of actual candidates. It took 6 weeks of manual chart review to resolving the list. Collectively, the 12 practices could have as many as 20,000 patients eligible for the July 1 enrollment. The pilot allowed the practices to get an early start on recruitment of business office staff and plans to address the backlog of potentially eligible patients. The process of identifying eligible patients for the OCM still needs a better solution because finding the appropriate patients is a critical first step in this model.
Underlying all of these initiatives is communication, both internal and external. We have to select and celebrate best practices. We have to educate our staffs. We will have to demonstrate that we are giving better care to our patients by using patient and provider testimonials and data.
From angst to analytics
The challenges of practice transformation can be daunting. It will be difficult to formalize processes and document data in ways that were untested before the pilot program was set up. However, the pilot accomplished 2 things: it identified additional areas that needed improvement and it demonstrated that the most challenging aspects of the OCM were feasible. Navigation and the IOM care plan were broken down into parts; each component was separately addressed, and programs were put in place to make the pieces manageable and part of an overall movement toward team-based care. The addition of a technology platform has been a key factor for the success of the value-based care initiative. Additional technology support has been enlisted to facilitate the processes, and an electronic version of the treatment plan is being tested. More difficult will be efforts to address the cultural resistance to change, which we hope to do by using data and outcomes from the CMS claims data files. The OCM represents an unprecedented opportunity for measurement of the quality of care we deliver.
We are now well und
Acknowledgment
The authors thank Supriya Srinivasan, PhD, for editorial support.
1. Oncology Care Model. Centers for Medicare and Medicaid Services 2016. https://innovation.cms.gov/initiatives/oncology-care/. Last updated November 14, 2017. Accessed November 16 2016.
2. Mortimer L, Strawbridge L, Lukens E, et al. CMS’ Oncology Care Model: delivering higher value cancer care. Clin Pharmacol Ther. 2017.
3. Medicare Prescription Drug, Improvement and Modernization Act of 2003, Pub Law No. 108-173; 2003.
4. Mathews D. The sequester: absolutely everything you could possibly need to know, in one FAQ. https://www.washingtonpost.com/news/wonk/wp/2013/02/20/the-sequester-absolutely-everything-you-could-possibly-need-to-know-in-one-faq/?utm_term=.a0f3a768399b. Published February 20, 2013. Accessed December 4, 2017.
5. Community Oncology Alliance. 2016 community oncology practice impact report: tracking the changing landscape of cancer care. https://www.communityoncology.org/wp-content/uploads/2016/09/PracticeImpactReport-2016-Report.pdf. Issued October 4, 2016. Accessed April 10, 2017.
6. The Patient Protection and Affordable Care Act, Pub Law No. 111-148; 2010.
7. Centers for Medicare and Medicaid Services. Shared Savings Program 2016. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/sharedsavingsprogram/index.html?redirect=/sharedsavingsprogram/. Last modified October 12, 2017. Accessed November 16 2016.
8. Dale SB, Ghosh A, Peikes DN, et al. Two-year costs and quality in the comprehensive primary care initiative. N Engl J Med. 2016;374:2345-2356.
9. McWilliams JM, Chernew ME, Landon BE, Schwartz AL. Performance differences in year 1 of pioneer accountable care organizations. N Engl J Med. 2015;372:1927-1936.
10. Rajkumar R, Press MJ, Conway PH. The CMS Innovation Center – a five-year self-assessment. N Engl J Med. 2015;372:1981-1983.
11. Waters TM, Webster JA, Stevens LA, et al. Community oncology medical homes: physician-driven change to improve patient care and reduce costs. J Oncol Pract. 2015;11(6):462-467.
12. Wilfong LS, Hoverman JR, Gosse N, Neubauer MA, Torres V. Changing physician compensation and implementing new technology to enhance pathways compliance. J Clin Oncol. 2016;34(Suppl 7S):Abstr 187.
13. Hoverman JR, Cartwright TH, Patt DA, et al. Pathways, outcomes, and costs in colon cancer: retrospective evaluations in two distinct databases. J Oncol Pract. 2011;7(3 Suppl):52s-59s.
14. Neubauer MA, Hoverman JR, Kolodziej M, et al. Cost effectiveness of evidence-based treatment guidelines for the treatment of non-small-cell lung cancer in the community setting. J Oncol Pract. 2010;6(1):12-18.
15. Hoverman JR, Klein I, Harrison DW, et al. Opening the black box: the impact of an oncology management program consisting of level I pathways and an outbound nurse call system. J Oncol Pract. 2014;10(1):63-67.
16. Newcomer LN, Gould B, Page RD, Donelan SA, Perkins M. Changing physician incentives for affordable, quality cancer care: results of an episode payment model. J Oncol Pract. 2014;10(5):322-326.
17. Meyer JA, Rybowski L, Eichler R. Theory and reality of value-based purchasing: lessons from the pioneers. Rockville, MD.: Agency for Health Care Policy and Research; 1997.
18. Stein CM. Academic clinical research: death by a thousand clicks. Sci Transl Med. 2015;7:318fs349.
19. Churchill W. The end of the beginning. 1st ed. Boston, MA: Little Brown & Co; 1943.
1. Oncology Care Model. Centers for Medicare and Medicaid Services 2016. https://innovation.cms.gov/initiatives/oncology-care/. Last updated November 14, 2017. Accessed November 16 2016.
2. Mortimer L, Strawbridge L, Lukens E, et al. CMS’ Oncology Care Model: delivering higher value cancer care. Clin Pharmacol Ther. 2017.
3. Medicare Prescription Drug, Improvement and Modernization Act of 2003, Pub Law No. 108-173; 2003.
4. Mathews D. The sequester: absolutely everything you could possibly need to know, in one FAQ. https://www.washingtonpost.com/news/wonk/wp/2013/02/20/the-sequester-absolutely-everything-you-could-possibly-need-to-know-in-one-faq/?utm_term=.a0f3a768399b. Published February 20, 2013. Accessed December 4, 2017.
5. Community Oncology Alliance. 2016 community oncology practice impact report: tracking the changing landscape of cancer care. https://www.communityoncology.org/wp-content/uploads/2016/09/PracticeImpactReport-2016-Report.pdf. Issued October 4, 2016. Accessed April 10, 2017.
6. The Patient Protection and Affordable Care Act, Pub Law No. 111-148; 2010.
7. Centers for Medicare and Medicaid Services. Shared Savings Program 2016. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/sharedsavingsprogram/index.html?redirect=/sharedsavingsprogram/. Last modified October 12, 2017. Accessed November 16 2016.
8. Dale SB, Ghosh A, Peikes DN, et al. Two-year costs and quality in the comprehensive primary care initiative. N Engl J Med. 2016;374:2345-2356.
9. McWilliams JM, Chernew ME, Landon BE, Schwartz AL. Performance differences in year 1 of pioneer accountable care organizations. N Engl J Med. 2015;372:1927-1936.
10. Rajkumar R, Press MJ, Conway PH. The CMS Innovation Center – a five-year self-assessment. N Engl J Med. 2015;372:1981-1983.
11. Waters TM, Webster JA, Stevens LA, et al. Community oncology medical homes: physician-driven change to improve patient care and reduce costs. J Oncol Pract. 2015;11(6):462-467.
12. Wilfong LS, Hoverman JR, Gosse N, Neubauer MA, Torres V. Changing physician compensation and implementing new technology to enhance pathways compliance. J Clin Oncol. 2016;34(Suppl 7S):Abstr 187.
13. Hoverman JR, Cartwright TH, Patt DA, et al. Pathways, outcomes, and costs in colon cancer: retrospective evaluations in two distinct databases. J Oncol Pract. 2011;7(3 Suppl):52s-59s.
14. Neubauer MA, Hoverman JR, Kolodziej M, et al. Cost effectiveness of evidence-based treatment guidelines for the treatment of non-small-cell lung cancer in the community setting. J Oncol Pract. 2010;6(1):12-18.
15. Hoverman JR, Klein I, Harrison DW, et al. Opening the black box: the impact of an oncology management program consisting of level I pathways and an outbound nurse call system. J Oncol Pract. 2014;10(1):63-67.
16. Newcomer LN, Gould B, Page RD, Donelan SA, Perkins M. Changing physician incentives for affordable, quality cancer care: results of an episode payment model. J Oncol Pract. 2014;10(5):322-326.
17. Meyer JA, Rybowski L, Eichler R. Theory and reality of value-based purchasing: lessons from the pioneers. Rockville, MD.: Agency for Health Care Policy and Research; 1997.
18. Stein CM. Academic clinical research: death by a thousand clicks. Sci Transl Med. 2015;7:318fs349.
19. Churchill W. The end of the beginning. 1st ed. Boston, MA: Little Brown & Co; 1943.
Hodgkin lymphoma survivors are at an increased risk of subsequent ER-negative breast cancer
Young women with primary Hodgkin lymphoma had an increased relative risk of estrogen receptor–positive breast cancer if they received radiotherapy and, irrespective of the type of treatment they got, an elevated risk of ER-negative breast cancer, based on results of a study based on patient records from 12 U.S. National Cancer Institute Surveillance, Epidemiology, and End Results registries.
Of 7,355 women diagnosed with primary Hodgkin lymphoma during 1973-2009 and aged 10-39 years, 377 patients subsequently were diagnosed with breast cancer at a mean age of 45 years; 57% of the cancers were ER positive, 34% were ER negative, and 9% had unknown/borderline ER status, Diana R. Withrow, PhD, and her colleagues from the radiation epidemiology branch, division of cancer epidemiology and genetics, National Cancer Institute reported in JAMA Oncology.
Survivors of Hodgkin lymphoma had a greater relative risk of ER-negative (standardized incidence ratio, 5.8; 95% confidence interval, 4.8-6.9) than ER-positive breast cancer (SIR, 3.1; 95% CI, 2.7-3.5; P less than .001 for the difference), the researchers wrote.
For ER-positive disease, the increased SIR was observed only among women who had received radiotherapy for their Hodgkin lymphoma (SIR, 3.9; 95% CI, 3.4-4.5). In this group, the SIR for ER-positive disease was lower in the chemotherapy than in the no/unknown chemotherapy group (P = .04), said the researchers.
The authors acknowledged that lack of information on patient risk factors such as family history, reproductive factors, and hormone therapy, as well as detailed treatment information such as radiotherapy dose, fields, specific chemotherapeutic agents, and subsequent therapy is a limitation of the current study. Further research, including comprehensive treatment records, will lead to a better understanding of the association between treatment and breast cancer subtype in these patients, the researchers concluded.
None of the study authors reported any conflicts of interest.
SOURCE: Withrow D et al. doi: 10.1001/jamaoncol.2017.4887.
Young women with primary Hodgkin lymphoma had an increased relative risk of estrogen receptor–positive breast cancer if they received radiotherapy and, irrespective of the type of treatment they got, an elevated risk of ER-negative breast cancer, based on results of a study based on patient records from 12 U.S. National Cancer Institute Surveillance, Epidemiology, and End Results registries.
Of 7,355 women diagnosed with primary Hodgkin lymphoma during 1973-2009 and aged 10-39 years, 377 patients subsequently were diagnosed with breast cancer at a mean age of 45 years; 57% of the cancers were ER positive, 34% were ER negative, and 9% had unknown/borderline ER status, Diana R. Withrow, PhD, and her colleagues from the radiation epidemiology branch, division of cancer epidemiology and genetics, National Cancer Institute reported in JAMA Oncology.
Survivors of Hodgkin lymphoma had a greater relative risk of ER-negative (standardized incidence ratio, 5.8; 95% confidence interval, 4.8-6.9) than ER-positive breast cancer (SIR, 3.1; 95% CI, 2.7-3.5; P less than .001 for the difference), the researchers wrote.
For ER-positive disease, the increased SIR was observed only among women who had received radiotherapy for their Hodgkin lymphoma (SIR, 3.9; 95% CI, 3.4-4.5). In this group, the SIR for ER-positive disease was lower in the chemotherapy than in the no/unknown chemotherapy group (P = .04), said the researchers.
The authors acknowledged that lack of information on patient risk factors such as family history, reproductive factors, and hormone therapy, as well as detailed treatment information such as radiotherapy dose, fields, specific chemotherapeutic agents, and subsequent therapy is a limitation of the current study. Further research, including comprehensive treatment records, will lead to a better understanding of the association between treatment and breast cancer subtype in these patients, the researchers concluded.
None of the study authors reported any conflicts of interest.
SOURCE: Withrow D et al. doi: 10.1001/jamaoncol.2017.4887.
Young women with primary Hodgkin lymphoma had an increased relative risk of estrogen receptor–positive breast cancer if they received radiotherapy and, irrespective of the type of treatment they got, an elevated risk of ER-negative breast cancer, based on results of a study based on patient records from 12 U.S. National Cancer Institute Surveillance, Epidemiology, and End Results registries.
Of 7,355 women diagnosed with primary Hodgkin lymphoma during 1973-2009 and aged 10-39 years, 377 patients subsequently were diagnosed with breast cancer at a mean age of 45 years; 57% of the cancers were ER positive, 34% were ER negative, and 9% had unknown/borderline ER status, Diana R. Withrow, PhD, and her colleagues from the radiation epidemiology branch, division of cancer epidemiology and genetics, National Cancer Institute reported in JAMA Oncology.
Survivors of Hodgkin lymphoma had a greater relative risk of ER-negative (standardized incidence ratio, 5.8; 95% confidence interval, 4.8-6.9) than ER-positive breast cancer (SIR, 3.1; 95% CI, 2.7-3.5; P less than .001 for the difference), the researchers wrote.
For ER-positive disease, the increased SIR was observed only among women who had received radiotherapy for their Hodgkin lymphoma (SIR, 3.9; 95% CI, 3.4-4.5). In this group, the SIR for ER-positive disease was lower in the chemotherapy than in the no/unknown chemotherapy group (P = .04), said the researchers.
The authors acknowledged that lack of information on patient risk factors such as family history, reproductive factors, and hormone therapy, as well as detailed treatment information such as radiotherapy dose, fields, specific chemotherapeutic agents, and subsequent therapy is a limitation of the current study. Further research, including comprehensive treatment records, will lead to a better understanding of the association between treatment and breast cancer subtype in these patients, the researchers concluded.
None of the study authors reported any conflicts of interest.
SOURCE: Withrow D et al. doi: 10.1001/jamaoncol.2017.4887.
FROM JAMA ONCOLOGY
Key clinical point: .
Major finding: Survivors of Hodgkin lymphoma had a greater relative risk of ER-negative (standardized incidence ratio, 5.8) than ER-positive breast cancer (SIR, 3.1).
Study details: 7,355 women diagnosed with first primary Hodgkin lymphoma during 1973-2009, who were aged 10-39 years, and reported to 12 U.S. National Cancer Institute SEER registries.
Disclosures: None reported.
Source: Withrow D et al. doi: 10.1001/jamaoncolo.2017.4887.
Analysis of Twitter lung cancer content reveals opportunity for clinicians
Social media communication around lung cancer is focused primarily on cancer treatment and use of pharmaceutical and research interventions, followed closely by awareness, prevention, and risk topics, according to an analysis of Twitter conversation over a 10-day period.
Although awareness and risk prevention tweets were likely to contain cues toward action, “messages focused on treatment, end of life ... were significantly less likely to integrate cues for personal activity,” the investigators wrote. The report was published in Journal of the American College of Radiology.
The investigators collected 1.3 million unique Twitter messages between Sept. 30 and Oct. 9, 2016, that contained at least one of six keywords commonly used to describe cancer: cancer, chemo, tumor, malignant, biopsy, and metastasis. They then drew a random, proportional stratified sample of 3,000 messages (12.5%) for manual coding from the 23,926 messages posted that included keywords related to lung cancer. Tweets were examined by user type (individuals, media, and organizations) to identify content and structural message features.
Message content was most frequently related to treatment (32.1%), followed by awareness (22.9%), end of life (15.5%), prevention and risk information (13.3%), active cancer-unknown phase (7.6%), diagnosis (6.1%), early detection (2.7%), and survivorship (1%), Dr. Sutton and her colleagues reported.
“The large volume of messages containing content about pharmaceuticals suggests that Twitter is also a forum for sharing information and discussing emerging treatments. Importantly, treatment messages were shared primarily by individuals, suggesting that this online user community jointly includes members of the public as well as medical practitioners and companies who have an awareness of emerging treatment approaches, suggesting an opportunity for online engagement between these various groups (e.g., Lung Cancer Social Media #LCSM community and related chats),” the investigators wrote.
The National Science Foundation supported parts of this research. None of the authors reported any conflicts of interest.
SOURCE: Sutton J. et al., J Am Coll Radiol. 2018 Jan. doi: 10.1016/j.jacr.2017.09.043
Social media communication around lung cancer is focused primarily on cancer treatment and use of pharmaceutical and research interventions, followed closely by awareness, prevention, and risk topics, according to an analysis of Twitter conversation over a 10-day period.
Although awareness and risk prevention tweets were likely to contain cues toward action, “messages focused on treatment, end of life ... were significantly less likely to integrate cues for personal activity,” the investigators wrote. The report was published in Journal of the American College of Radiology.
The investigators collected 1.3 million unique Twitter messages between Sept. 30 and Oct. 9, 2016, that contained at least one of six keywords commonly used to describe cancer: cancer, chemo, tumor, malignant, biopsy, and metastasis. They then drew a random, proportional stratified sample of 3,000 messages (12.5%) for manual coding from the 23,926 messages posted that included keywords related to lung cancer. Tweets were examined by user type (individuals, media, and organizations) to identify content and structural message features.
Message content was most frequently related to treatment (32.1%), followed by awareness (22.9%), end of life (15.5%), prevention and risk information (13.3%), active cancer-unknown phase (7.6%), diagnosis (6.1%), early detection (2.7%), and survivorship (1%), Dr. Sutton and her colleagues reported.
“The large volume of messages containing content about pharmaceuticals suggests that Twitter is also a forum for sharing information and discussing emerging treatments. Importantly, treatment messages were shared primarily by individuals, suggesting that this online user community jointly includes members of the public as well as medical practitioners and companies who have an awareness of emerging treatment approaches, suggesting an opportunity for online engagement between these various groups (e.g., Lung Cancer Social Media #LCSM community and related chats),” the investigators wrote.
The National Science Foundation supported parts of this research. None of the authors reported any conflicts of interest.
SOURCE: Sutton J. et al., J Am Coll Radiol. 2018 Jan. doi: 10.1016/j.jacr.2017.09.043
Social media communication around lung cancer is focused primarily on cancer treatment and use of pharmaceutical and research interventions, followed closely by awareness, prevention, and risk topics, according to an analysis of Twitter conversation over a 10-day period.
Although awareness and risk prevention tweets were likely to contain cues toward action, “messages focused on treatment, end of life ... were significantly less likely to integrate cues for personal activity,” the investigators wrote. The report was published in Journal of the American College of Radiology.
The investigators collected 1.3 million unique Twitter messages between Sept. 30 and Oct. 9, 2016, that contained at least one of six keywords commonly used to describe cancer: cancer, chemo, tumor, malignant, biopsy, and metastasis. They then drew a random, proportional stratified sample of 3,000 messages (12.5%) for manual coding from the 23,926 messages posted that included keywords related to lung cancer. Tweets were examined by user type (individuals, media, and organizations) to identify content and structural message features.
Message content was most frequently related to treatment (32.1%), followed by awareness (22.9%), end of life (15.5%), prevention and risk information (13.3%), active cancer-unknown phase (7.6%), diagnosis (6.1%), early detection (2.7%), and survivorship (1%), Dr. Sutton and her colleagues reported.
“The large volume of messages containing content about pharmaceuticals suggests that Twitter is also a forum for sharing information and discussing emerging treatments. Importantly, treatment messages were shared primarily by individuals, suggesting that this online user community jointly includes members of the public as well as medical practitioners and companies who have an awareness of emerging treatment approaches, suggesting an opportunity for online engagement between these various groups (e.g., Lung Cancer Social Media #LCSM community and related chats),” the investigators wrote.
The National Science Foundation supported parts of this research. None of the authors reported any conflicts of interest.
SOURCE: Sutton J. et al., J Am Coll Radiol. 2018 Jan. doi: 10.1016/j.jacr.2017.09.043
FROM JOURNAL OF THE AMERICAN COLLEGE OF RADIOLOGY
Key clinical point: In a random sample of Twitter conversation related to lung cancer, message content was most frequently related to treatment.
Major finding: Majority of tweets evaluated focused on lung cancer treatment and the use of pharmaceutical and research interventions, followed by awareness, prevention, and risk topics.
Study details: Random sample of 3,000 tweets posted in a 10-day period between Sept. 30 and Oct. 9, 2016. Lung cancer–specific tweets by user type (individuals, media, and organizations) were examined to identify content and structural message features.
Disclosures: The National Science Foundation supported parts of this research. None of the authors reported any conflicts of interest.
Source: Sutton J. et al., J Am Coll Radiol. 2018 Jan. doi: 10.1016/j.jacr.2017.09.043.
Supportive oncodermatology: Cancer advances spawn new subspecialty
Not too long ago at the Dana-Farber/Brigham and Women’s Cancer Center in Boston, a woman with widely metastatic melanoma, who had been planning her own funeral, was surprised when she had a phenomenal response to immunotherapy.
She was shocked to learn that her cancer was almost completely gone after 12 weeks, but she was stunned when she developed a rash that made her oncologist think she needed to stop treatment.
With traditional cytotoxic chemotherapies, there were a few well-defined skin side effects that oncologists were comfortable managing on their own with steroids or by reducing or stopping treatment for a bit.
But over the last decade, new cancer options have become available, most notably immunotherapies and targeted biologics, which are keeping some people alive longer but also causing cutaneous side effects that have never been seen before in oncology and are being reported frequently.
An urgent need
Currently in the United States, there’s only a handful of dedicated supportive oncodermatology services, which can be found at major academic cancer centers such as Dana-Farber/Brigham and Women’s, but the residents and fellows being trained at these centers are starting to fan out across the country and set up new services.
One day, it’s likely that every major cancer institution will have “a toxicities team with expert dermatologists,” said Dr. LeBoeuf, who launched the supportive oncodermatology program at Dana-Farber in 2014 and who now runs it with a team of dermatologists and clinics every week. Dr. LeBoeuf is a leader in the field, like the other dermatologists interviewed for this story.
With all the new treatments and with even more on the way, “there’s an urgent need for dermatologists to be involved in care of cancer patients,” Dr. LeBoeuf said.
The problem
Immunotherapies like the PD-1 blocking agents pembrolizumab (Keytruda) and nivolumab (Opdivo) – both used for an ever-expanding list of tumors – amp up the immune system to fight cancer, but they also tend to cause adverse events that mimic autoimmune diseases such as lupus, psoriasis, lichen planus, and vitiligo. Dermatologists are familiar with those problems and how to manage them, but oncologists generally are not.
Meanwhile, the many targeted therapies approved over the past decade interfere with specific molecules needed for tumor growth, but they also are associated with a wide range of skin, hair, and nail side effects that include skin growths, itching, paronychia, and more.
Agents that target vascular endothelial growth factors, such as sorafenib (Nexavar) and bevacizumab (Avastin), can trigger a painful hand-foot skin reaction that’s different from the hand-foot syndrome reported with older cytotoxic agents.
Epidermal growth factor receptor (EGFR) inhibitors, such as erlotinib (Tarceva) or gefitinib (Iressa), often cause miserable acne-like eruptions, but that can mean the drug is working.
It’s hard for oncologists to know what’s life-threatening and what isn’t; that’s where dermatologists come in.
A solution
When problems come up, oncologists and patients need answers right away, she said. There’s no time to wait a month or two for a dermatology appointment to find out whether, for instance, a new mouth ulcer is a minor inconvenience or the first sign of Stevens-Johnson syndrome, and the last thing an exhausted cancer patient needs is to be told to go to yet another clinic for a dermatology consult.
For supportive oncodermatology, that means being where the patients are: in the cancer centers. “Our clinic is situated on the same cancer floor as all the other oncology clinics,” which means easy access for both patients and oncologists, Dr. Choi said. “They just come down the hall.”
Build it, and they will come
The Stanford (Calif.) Cancer Center is a good example of what happens once a supportive oncodermatology service is up and running.
The program there was the brainchild of dermatologist Bernice Kwong, MD, who helped launch it in 2012 with 2 half-day outpatient clinics per week.
“Once people knew we were there seeing patients, we needed to expand it to 3 half days, and within 6 months, we knew we had to be” in the cancer center daily, she said. “The oncologists felt we were helping them keep their patients on treatment longer; they didn’t have to stop therapy to sort out a rash.”
Currently, the clinic sees about 15 to 20 patients a day, but “we have more need than that,” said Dr. Kwong, who is trying to recruit more dermatologists to help.
“The need is huge. There’s so much room for growth,” she noted, but first, “you need the oncologists to be on board.”
Dermatologist Adam Friedman, MD, director of supportive oncodermatology at the George Washington University Cancer Center, Washington, says his program is on the other end of the growth curve since it was only launched in the spring of 2017. Only about 80 patients have been treated so far, and there’s one dedicated clinic day a month, although he is on call for urgent cases, as is the case for many of the other dermatologists interviewed for this story.
Dr. Friedman expects business will pick up soon once word gets out, just like at Dana-Farber/Brigham and Women’s, Stanford, and elsewhere. “The places with the greatest need are going to have these services first, and then you’ll see them pop up elsewhere. I think we are going to see more,” he said.
The birth of supportive oncodermatology
Dermatologist Mario Lacouture, MD, director of the oncodermatology program at Memorial Sloan Kettering Cancer Center, New York, is considered by many oncodermatologists to be the father of the field.
He started the very first program in 2005 at Northwestern University, Chicago, followed by the program at Sloan Kettering a few years later. He has helped train many of the leaders in the field and coined the phrase “supportive oncodermatology” as the senior author in the field’s seminal paper, published in 2011 (J Am Acad Dermatol. 2011 Sep;65[3]:624-35). That article, in turn, inspired at least a few young dermatologists to make supportive oncodermatology their career choice. Dr. Lacouture speaks regularly at oncology and dermatology meetings to raise awareness about how dermatologists can improve cancer care.
Cancer survivors were also a concern. “Cancer treatment has improved so much that people are living longer, but the majority of survivors have either temporary or permanent cutaneous problems that would benefit from dermatologic care. However, the oncology community and patients are usually not aware that there are things we can do to help,” Dr. Lacouture said.
The message seems to have gotten out, however, among the hundreds of oncologists affiliated with Sloan Kettering. Dr. Lacouture needs a team of supportive oncodermatologists to meet the demand, with walk-in clinics every day and round-the-clock call.
He anticipates a day when visiting a supportive oncodermatologist will be routine, even before the start of cancer treatment, just as people visit a dentist before bone marrow transplants or radiation treatment to the head and neck. The idea would be to prevent cutaneous toxicity, something Dr. Lacouture and his team are already doing at Sloan Kettering. In time, supportive oncodermatology “is something that is going to be instituted early on” in treatment, he said.
“It’s important for dermatologists to reach out to their local oncologists; they will see there are many, many cancer patients and survivors who would benefit immensely from their care,” he said.
Dr. Lacouture is a consultant for Galderma, Janssen, and Johnson & Johnson. The other dermatologists interviewed for this story had no relevant industry disclosures. La Roche-Posay, a subsidiary of L’Oreal, is helping fund the supportive oncodermatology program at George Washington University. The company is interested in using cosmetics to camouflage cancer treatment skin lesions, Dr. Friedman said. Dr. Friedman is a member of the Dermatology News advisory board.
[email protected]
Not too long ago at the Dana-Farber/Brigham and Women’s Cancer Center in Boston, a woman with widely metastatic melanoma, who had been planning her own funeral, was surprised when she had a phenomenal response to immunotherapy.
She was shocked to learn that her cancer was almost completely gone after 12 weeks, but she was stunned when she developed a rash that made her oncologist think she needed to stop treatment.
With traditional cytotoxic chemotherapies, there were a few well-defined skin side effects that oncologists were comfortable managing on their own with steroids or by reducing or stopping treatment for a bit.
But over the last decade, new cancer options have become available, most notably immunotherapies and targeted biologics, which are keeping some people alive longer but also causing cutaneous side effects that have never been seen before in oncology and are being reported frequently.
An urgent need
Currently in the United States, there’s only a handful of dedicated supportive oncodermatology services, which can be found at major academic cancer centers such as Dana-Farber/Brigham and Women’s, but the residents and fellows being trained at these centers are starting to fan out across the country and set up new services.
One day, it’s likely that every major cancer institution will have “a toxicities team with expert dermatologists,” said Dr. LeBoeuf, who launched the supportive oncodermatology program at Dana-Farber in 2014 and who now runs it with a team of dermatologists and clinics every week. Dr. LeBoeuf is a leader in the field, like the other dermatologists interviewed for this story.
With all the new treatments and with even more on the way, “there’s an urgent need for dermatologists to be involved in care of cancer patients,” Dr. LeBoeuf said.
The problem
Immunotherapies like the PD-1 blocking agents pembrolizumab (Keytruda) and nivolumab (Opdivo) – both used for an ever-expanding list of tumors – amp up the immune system to fight cancer, but they also tend to cause adverse events that mimic autoimmune diseases such as lupus, psoriasis, lichen planus, and vitiligo. Dermatologists are familiar with those problems and how to manage them, but oncologists generally are not.
Meanwhile, the many targeted therapies approved over the past decade interfere with specific molecules needed for tumor growth, but they also are associated with a wide range of skin, hair, and nail side effects that include skin growths, itching, paronychia, and more.
Agents that target vascular endothelial growth factors, such as sorafenib (Nexavar) and bevacizumab (Avastin), can trigger a painful hand-foot skin reaction that’s different from the hand-foot syndrome reported with older cytotoxic agents.
Epidermal growth factor receptor (EGFR) inhibitors, such as erlotinib (Tarceva) or gefitinib (Iressa), often cause miserable acne-like eruptions, but that can mean the drug is working.
It’s hard for oncologists to know what’s life-threatening and what isn’t; that’s where dermatologists come in.
A solution
When problems come up, oncologists and patients need answers right away, she said. There’s no time to wait a month or two for a dermatology appointment to find out whether, for instance, a new mouth ulcer is a minor inconvenience or the first sign of Stevens-Johnson syndrome, and the last thing an exhausted cancer patient needs is to be told to go to yet another clinic for a dermatology consult.
For supportive oncodermatology, that means being where the patients are: in the cancer centers. “Our clinic is situated on the same cancer floor as all the other oncology clinics,” which means easy access for both patients and oncologists, Dr. Choi said. “They just come down the hall.”
Build it, and they will come
The Stanford (Calif.) Cancer Center is a good example of what happens once a supportive oncodermatology service is up and running.
The program there was the brainchild of dermatologist Bernice Kwong, MD, who helped launch it in 2012 with 2 half-day outpatient clinics per week.
“Once people knew we were there seeing patients, we needed to expand it to 3 half days, and within 6 months, we knew we had to be” in the cancer center daily, she said. “The oncologists felt we were helping them keep their patients on treatment longer; they didn’t have to stop therapy to sort out a rash.”
Currently, the clinic sees about 15 to 20 patients a day, but “we have more need than that,” said Dr. Kwong, who is trying to recruit more dermatologists to help.
“The need is huge. There’s so much room for growth,” she noted, but first, “you need the oncologists to be on board.”
Dermatologist Adam Friedman, MD, director of supportive oncodermatology at the George Washington University Cancer Center, Washington, says his program is on the other end of the growth curve since it was only launched in the spring of 2017. Only about 80 patients have been treated so far, and there’s one dedicated clinic day a month, although he is on call for urgent cases, as is the case for many of the other dermatologists interviewed for this story.
Dr. Friedman expects business will pick up soon once word gets out, just like at Dana-Farber/Brigham and Women’s, Stanford, and elsewhere. “The places with the greatest need are going to have these services first, and then you’ll see them pop up elsewhere. I think we are going to see more,” he said.
The birth of supportive oncodermatology
Dermatologist Mario Lacouture, MD, director of the oncodermatology program at Memorial Sloan Kettering Cancer Center, New York, is considered by many oncodermatologists to be the father of the field.
He started the very first program in 2005 at Northwestern University, Chicago, followed by the program at Sloan Kettering a few years later. He has helped train many of the leaders in the field and coined the phrase “supportive oncodermatology” as the senior author in the field’s seminal paper, published in 2011 (J Am Acad Dermatol. 2011 Sep;65[3]:624-35). That article, in turn, inspired at least a few young dermatologists to make supportive oncodermatology their career choice. Dr. Lacouture speaks regularly at oncology and dermatology meetings to raise awareness about how dermatologists can improve cancer care.
Cancer survivors were also a concern. “Cancer treatment has improved so much that people are living longer, but the majority of survivors have either temporary or permanent cutaneous problems that would benefit from dermatologic care. However, the oncology community and patients are usually not aware that there are things we can do to help,” Dr. Lacouture said.
The message seems to have gotten out, however, among the hundreds of oncologists affiliated with Sloan Kettering. Dr. Lacouture needs a team of supportive oncodermatologists to meet the demand, with walk-in clinics every day and round-the-clock call.
He anticipates a day when visiting a supportive oncodermatologist will be routine, even before the start of cancer treatment, just as people visit a dentist before bone marrow transplants or radiation treatment to the head and neck. The idea would be to prevent cutaneous toxicity, something Dr. Lacouture and his team are already doing at Sloan Kettering. In time, supportive oncodermatology “is something that is going to be instituted early on” in treatment, he said.
“It’s important for dermatologists to reach out to their local oncologists; they will see there are many, many cancer patients and survivors who would benefit immensely from their care,” he said.
Dr. Lacouture is a consultant for Galderma, Janssen, and Johnson & Johnson. The other dermatologists interviewed for this story had no relevant industry disclosures. La Roche-Posay, a subsidiary of L’Oreal, is helping fund the supportive oncodermatology program at George Washington University. The company is interested in using cosmetics to camouflage cancer treatment skin lesions, Dr. Friedman said. Dr. Friedman is a member of the Dermatology News advisory board.
[email protected]
Not too long ago at the Dana-Farber/Brigham and Women’s Cancer Center in Boston, a woman with widely metastatic melanoma, who had been planning her own funeral, was surprised when she had a phenomenal response to immunotherapy.
She was shocked to learn that her cancer was almost completely gone after 12 weeks, but she was stunned when she developed a rash that made her oncologist think she needed to stop treatment.
With traditional cytotoxic chemotherapies, there were a few well-defined skin side effects that oncologists were comfortable managing on their own with steroids or by reducing or stopping treatment for a bit.
But over the last decade, new cancer options have become available, most notably immunotherapies and targeted biologics, which are keeping some people alive longer but also causing cutaneous side effects that have never been seen before in oncology and are being reported frequently.
An urgent need
Currently in the United States, there’s only a handful of dedicated supportive oncodermatology services, which can be found at major academic cancer centers such as Dana-Farber/Brigham and Women’s, but the residents and fellows being trained at these centers are starting to fan out across the country and set up new services.
One day, it’s likely that every major cancer institution will have “a toxicities team with expert dermatologists,” said Dr. LeBoeuf, who launched the supportive oncodermatology program at Dana-Farber in 2014 and who now runs it with a team of dermatologists and clinics every week. Dr. LeBoeuf is a leader in the field, like the other dermatologists interviewed for this story.
With all the new treatments and with even more on the way, “there’s an urgent need for dermatologists to be involved in care of cancer patients,” Dr. LeBoeuf said.
The problem
Immunotherapies like the PD-1 blocking agents pembrolizumab (Keytruda) and nivolumab (Opdivo) – both used for an ever-expanding list of tumors – amp up the immune system to fight cancer, but they also tend to cause adverse events that mimic autoimmune diseases such as lupus, psoriasis, lichen planus, and vitiligo. Dermatologists are familiar with those problems and how to manage them, but oncologists generally are not.
Meanwhile, the many targeted therapies approved over the past decade interfere with specific molecules needed for tumor growth, but they also are associated with a wide range of skin, hair, and nail side effects that include skin growths, itching, paronychia, and more.
Agents that target vascular endothelial growth factors, such as sorafenib (Nexavar) and bevacizumab (Avastin), can trigger a painful hand-foot skin reaction that’s different from the hand-foot syndrome reported with older cytotoxic agents.
Epidermal growth factor receptor (EGFR) inhibitors, such as erlotinib (Tarceva) or gefitinib (Iressa), often cause miserable acne-like eruptions, but that can mean the drug is working.
It’s hard for oncologists to know what’s life-threatening and what isn’t; that’s where dermatologists come in.
A solution
When problems come up, oncologists and patients need answers right away, she said. There’s no time to wait a month or two for a dermatology appointment to find out whether, for instance, a new mouth ulcer is a minor inconvenience or the first sign of Stevens-Johnson syndrome, and the last thing an exhausted cancer patient needs is to be told to go to yet another clinic for a dermatology consult.
For supportive oncodermatology, that means being where the patients are: in the cancer centers. “Our clinic is situated on the same cancer floor as all the other oncology clinics,” which means easy access for both patients and oncologists, Dr. Choi said. “They just come down the hall.”
Build it, and they will come
The Stanford (Calif.) Cancer Center is a good example of what happens once a supportive oncodermatology service is up and running.
The program there was the brainchild of dermatologist Bernice Kwong, MD, who helped launch it in 2012 with 2 half-day outpatient clinics per week.
“Once people knew we were there seeing patients, we needed to expand it to 3 half days, and within 6 months, we knew we had to be” in the cancer center daily, she said. “The oncologists felt we were helping them keep their patients on treatment longer; they didn’t have to stop therapy to sort out a rash.”
Currently, the clinic sees about 15 to 20 patients a day, but “we have more need than that,” said Dr. Kwong, who is trying to recruit more dermatologists to help.
“The need is huge. There’s so much room for growth,” she noted, but first, “you need the oncologists to be on board.”
Dermatologist Adam Friedman, MD, director of supportive oncodermatology at the George Washington University Cancer Center, Washington, says his program is on the other end of the growth curve since it was only launched in the spring of 2017. Only about 80 patients have been treated so far, and there’s one dedicated clinic day a month, although he is on call for urgent cases, as is the case for many of the other dermatologists interviewed for this story.
Dr. Friedman expects business will pick up soon once word gets out, just like at Dana-Farber/Brigham and Women’s, Stanford, and elsewhere. “The places with the greatest need are going to have these services first, and then you’ll see them pop up elsewhere. I think we are going to see more,” he said.
The birth of supportive oncodermatology
Dermatologist Mario Lacouture, MD, director of the oncodermatology program at Memorial Sloan Kettering Cancer Center, New York, is considered by many oncodermatologists to be the father of the field.
He started the very first program in 2005 at Northwestern University, Chicago, followed by the program at Sloan Kettering a few years later. He has helped train many of the leaders in the field and coined the phrase “supportive oncodermatology” as the senior author in the field’s seminal paper, published in 2011 (J Am Acad Dermatol. 2011 Sep;65[3]:624-35). That article, in turn, inspired at least a few young dermatologists to make supportive oncodermatology their career choice. Dr. Lacouture speaks regularly at oncology and dermatology meetings to raise awareness about how dermatologists can improve cancer care.
Cancer survivors were also a concern. “Cancer treatment has improved so much that people are living longer, but the majority of survivors have either temporary or permanent cutaneous problems that would benefit from dermatologic care. However, the oncology community and patients are usually not aware that there are things we can do to help,” Dr. Lacouture said.
The message seems to have gotten out, however, among the hundreds of oncologists affiliated with Sloan Kettering. Dr. Lacouture needs a team of supportive oncodermatologists to meet the demand, with walk-in clinics every day and round-the-clock call.
He anticipates a day when visiting a supportive oncodermatologist will be routine, even before the start of cancer treatment, just as people visit a dentist before bone marrow transplants or radiation treatment to the head and neck. The idea would be to prevent cutaneous toxicity, something Dr. Lacouture and his team are already doing at Sloan Kettering. In time, supportive oncodermatology “is something that is going to be instituted early on” in treatment, he said.
“It’s important for dermatologists to reach out to their local oncologists; they will see there are many, many cancer patients and survivors who would benefit immensely from their care,” he said.
Dr. Lacouture is a consultant for Galderma, Janssen, and Johnson & Johnson. The other dermatologists interviewed for this story had no relevant industry disclosures. La Roche-Posay, a subsidiary of L’Oreal, is helping fund the supportive oncodermatology program at George Washington University. The company is interested in using cosmetics to camouflage cancer treatment skin lesions, Dr. Friedman said. Dr. Friedman is a member of the Dermatology News advisory board.
[email protected]
Immunotherapy with less intense chemotherapy safe and effective for older patients with ALL
according to the results of a single-arm phase 2 study.
After a median follow-up of 29 months, 2-year progression-free survival was 59% with a median of 25 months for 52 patients with newly diagnosed disease who were aged 60 years or older, Hagop Kantarjian, MD, and his associates reported online in the Lancet Oncology.
Nearly every patient experienced an overall response (98%). The treatment also appeared to be safe, Dr. Kantarjian and associates wrote.
They reported that no patient died within 4 weeks of treatment and that four patients experienced veno-occlusive disease. Common grade 3 or 4 side effects included thrombocytopenia (81%), consolidation chemotherapy (69%), hyperglycemia (54%), infections during induction (52%), hypokalemia (31%), increased aminotransferases (19%), hyperbilirubinemia (17%), and hemorrhage (15%). Six patients died from treatment-related side effects, five of whom died from sepsis and one of whom died as a result of veno-occlusive disease.
Inotuzumab ozogamicin is an anti-CD22 monoclonal antibody that is bound to the toxin calicheamicin. It has shown “substantial improvements” for some patient subgroups when added to hyper-CVAD chemotherapy (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and high-dose cytarabine); however, older patients have yet to experience a similar benefit, Dr. Kantarjian and his associates said.
Both CVAD and inotuzumab ozogamicin have shown to be quite toxic for older patients. So – in order limit increased toxicity – Dr. Kantarjian and his associates lowered the intensity of hyper-CVAD. They accomplished this by reducing some aspects of the regimen by up to 50% and eliminating anthracyclines altogether. The researchers referred to this regimen as “mini–hyper-CVD.” The mini–hyper-CVD did not appear to impact activity.
“In our study, activity did not appear to be compromised by the use of lower-intensity chemotherapy in combination with a novel monoclonal antibody,” they wrote.
Dr. Kantarjian and his associates noted that, while they observed fewer deaths compared with previously published data, the number of deaths they did observe in patients who achieved a complete response was high.
They suggested that modifications to the regimen could further improve safety and that such modifications required further research.
For the time being, however, the researchers noted that this low-intensity regimen, in combination in inotuzumab ozogamicin, is highly effective and that these data now require prospective confirmation in a randomized, phase 3 setting.
The study was funded by the MD Anderson Cancer Center. Researchers reported that Pfizer provided inotuzumab ozogamicin free of charge and that Dr. Kantarjian and two other researchers received grants from Pfizer.
SOURCE: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
The efficacy and safety of inotuzumab ozogamicin reported by Hagop Kantarjian, MD, and his colleagues are an encouraging building block for older patents with acute lymphoblastic leukemia, according to Carmelo Rizzari, MD.
“The authors have upgraded the valuable premises available from early inotuzumab ozogamicin studies into more concrete promises by rationally integrating the drug in a reduced-intensity chemotherapy regimen,” wrote Dr. Rizzari in an accompanying editorial published Lancet Oncology.
Adding inotuzumab ozogamicin to chemotherapy has shown efficacy and safety for both pediatric and adult patients with relapsed or refractory acute lymphoblastic leukemia and therefore deserves more investigation in the “neglected” subgroup of older patients, according to Dr. Rizzari.
“Monoclonal antibodies represent, in the wider settings of immunotherapy, viable options to improve the results obtained in different subsets of patients with acute lymphoblastic leukemia,” wrote Dr. Rizzari.
Echoing the researchers, Dr. Rizzari mentioned the importance of the next step – a phase 3 trial comparing inotuzumab ozogamicin with the current standard of care – but also mentioned that a specific, widely recognized standard of care for this patient subgroup is difficult to pin down.
Dr. Rizzari is with the Pediatric Hematology-Oncology Unit of the University of Milano-Bicocca in Milan. These comments are based on an accompanying editorial published online in Lancet Oncology (2018 Jan 15. doi: 10.1016/S1470-2045[18]30013-5 ). Dr. Rizzari declared no competing interests.
The efficacy and safety of inotuzumab ozogamicin reported by Hagop Kantarjian, MD, and his colleagues are an encouraging building block for older patents with acute lymphoblastic leukemia, according to Carmelo Rizzari, MD.
“The authors have upgraded the valuable premises available from early inotuzumab ozogamicin studies into more concrete promises by rationally integrating the drug in a reduced-intensity chemotherapy regimen,” wrote Dr. Rizzari in an accompanying editorial published Lancet Oncology.
Adding inotuzumab ozogamicin to chemotherapy has shown efficacy and safety for both pediatric and adult patients with relapsed or refractory acute lymphoblastic leukemia and therefore deserves more investigation in the “neglected” subgroup of older patients, according to Dr. Rizzari.
“Monoclonal antibodies represent, in the wider settings of immunotherapy, viable options to improve the results obtained in different subsets of patients with acute lymphoblastic leukemia,” wrote Dr. Rizzari.
Echoing the researchers, Dr. Rizzari mentioned the importance of the next step – a phase 3 trial comparing inotuzumab ozogamicin with the current standard of care – but also mentioned that a specific, widely recognized standard of care for this patient subgroup is difficult to pin down.
Dr. Rizzari is with the Pediatric Hematology-Oncology Unit of the University of Milano-Bicocca in Milan. These comments are based on an accompanying editorial published online in Lancet Oncology (2018 Jan 15. doi: 10.1016/S1470-2045[18]30013-5 ). Dr. Rizzari declared no competing interests.
The efficacy and safety of inotuzumab ozogamicin reported by Hagop Kantarjian, MD, and his colleagues are an encouraging building block for older patents with acute lymphoblastic leukemia, according to Carmelo Rizzari, MD.
“The authors have upgraded the valuable premises available from early inotuzumab ozogamicin studies into more concrete promises by rationally integrating the drug in a reduced-intensity chemotherapy regimen,” wrote Dr. Rizzari in an accompanying editorial published Lancet Oncology.
Adding inotuzumab ozogamicin to chemotherapy has shown efficacy and safety for both pediatric and adult patients with relapsed or refractory acute lymphoblastic leukemia and therefore deserves more investigation in the “neglected” subgroup of older patients, according to Dr. Rizzari.
“Monoclonal antibodies represent, in the wider settings of immunotherapy, viable options to improve the results obtained in different subsets of patients with acute lymphoblastic leukemia,” wrote Dr. Rizzari.
Echoing the researchers, Dr. Rizzari mentioned the importance of the next step – a phase 3 trial comparing inotuzumab ozogamicin with the current standard of care – but also mentioned that a specific, widely recognized standard of care for this patient subgroup is difficult to pin down.
Dr. Rizzari is with the Pediatric Hematology-Oncology Unit of the University of Milano-Bicocca in Milan. These comments are based on an accompanying editorial published online in Lancet Oncology (2018 Jan 15. doi: 10.1016/S1470-2045[18]30013-5 ). Dr. Rizzari declared no competing interests.
according to the results of a single-arm phase 2 study.
After a median follow-up of 29 months, 2-year progression-free survival was 59% with a median of 25 months for 52 patients with newly diagnosed disease who were aged 60 years or older, Hagop Kantarjian, MD, and his associates reported online in the Lancet Oncology.
Nearly every patient experienced an overall response (98%). The treatment also appeared to be safe, Dr. Kantarjian and associates wrote.
They reported that no patient died within 4 weeks of treatment and that four patients experienced veno-occlusive disease. Common grade 3 or 4 side effects included thrombocytopenia (81%), consolidation chemotherapy (69%), hyperglycemia (54%), infections during induction (52%), hypokalemia (31%), increased aminotransferases (19%), hyperbilirubinemia (17%), and hemorrhage (15%). Six patients died from treatment-related side effects, five of whom died from sepsis and one of whom died as a result of veno-occlusive disease.
Inotuzumab ozogamicin is an anti-CD22 monoclonal antibody that is bound to the toxin calicheamicin. It has shown “substantial improvements” for some patient subgroups when added to hyper-CVAD chemotherapy (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and high-dose cytarabine); however, older patients have yet to experience a similar benefit, Dr. Kantarjian and his associates said.
Both CVAD and inotuzumab ozogamicin have shown to be quite toxic for older patients. So – in order limit increased toxicity – Dr. Kantarjian and his associates lowered the intensity of hyper-CVAD. They accomplished this by reducing some aspects of the regimen by up to 50% and eliminating anthracyclines altogether. The researchers referred to this regimen as “mini–hyper-CVD.” The mini–hyper-CVD did not appear to impact activity.
“In our study, activity did not appear to be compromised by the use of lower-intensity chemotherapy in combination with a novel monoclonal antibody,” they wrote.
Dr. Kantarjian and his associates noted that, while they observed fewer deaths compared with previously published data, the number of deaths they did observe in patients who achieved a complete response was high.
They suggested that modifications to the regimen could further improve safety and that such modifications required further research.
For the time being, however, the researchers noted that this low-intensity regimen, in combination in inotuzumab ozogamicin, is highly effective and that these data now require prospective confirmation in a randomized, phase 3 setting.
The study was funded by the MD Anderson Cancer Center. Researchers reported that Pfizer provided inotuzumab ozogamicin free of charge and that Dr. Kantarjian and two other researchers received grants from Pfizer.
SOURCE: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
according to the results of a single-arm phase 2 study.
After a median follow-up of 29 months, 2-year progression-free survival was 59% with a median of 25 months for 52 patients with newly diagnosed disease who were aged 60 years or older, Hagop Kantarjian, MD, and his associates reported online in the Lancet Oncology.
Nearly every patient experienced an overall response (98%). The treatment also appeared to be safe, Dr. Kantarjian and associates wrote.
They reported that no patient died within 4 weeks of treatment and that four patients experienced veno-occlusive disease. Common grade 3 or 4 side effects included thrombocytopenia (81%), consolidation chemotherapy (69%), hyperglycemia (54%), infections during induction (52%), hypokalemia (31%), increased aminotransferases (19%), hyperbilirubinemia (17%), and hemorrhage (15%). Six patients died from treatment-related side effects, five of whom died from sepsis and one of whom died as a result of veno-occlusive disease.
Inotuzumab ozogamicin is an anti-CD22 monoclonal antibody that is bound to the toxin calicheamicin. It has shown “substantial improvements” for some patient subgroups when added to hyper-CVAD chemotherapy (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and high-dose cytarabine); however, older patients have yet to experience a similar benefit, Dr. Kantarjian and his associates said.
Both CVAD and inotuzumab ozogamicin have shown to be quite toxic for older patients. So – in order limit increased toxicity – Dr. Kantarjian and his associates lowered the intensity of hyper-CVAD. They accomplished this by reducing some aspects of the regimen by up to 50% and eliminating anthracyclines altogether. The researchers referred to this regimen as “mini–hyper-CVD.” The mini–hyper-CVD did not appear to impact activity.
“In our study, activity did not appear to be compromised by the use of lower-intensity chemotherapy in combination with a novel monoclonal antibody,” they wrote.
Dr. Kantarjian and his associates noted that, while they observed fewer deaths compared with previously published data, the number of deaths they did observe in patients who achieved a complete response was high.
They suggested that modifications to the regimen could further improve safety and that such modifications required further research.
For the time being, however, the researchers noted that this low-intensity regimen, in combination in inotuzumab ozogamicin, is highly effective and that these data now require prospective confirmation in a randomized, phase 3 setting.
The study was funded by the MD Anderson Cancer Center. Researchers reported that Pfizer provided inotuzumab ozogamicin free of charge and that Dr. Kantarjian and two other researchers received grants from Pfizer.
SOURCE: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.
FROM LANCET ONCOLOGY
Key clinical point: Inotuzumab ozogamicin with less intense chemotherapy is safe and effective for older patients with ALL.
Major finding: After a median follow-up of 29 months, 59% of patients experienced 2-year recurrence-free survival, 56% experienced 3-year overall survival and only four patients died within four weeks of treatment.
Data source: Single-arm phase 2 study of 52 patients aged 60 years or older with newly diagnosed Philadelphia chromosome–negative ALL.
Disclosures: The study was funded by a grant from the MD Anderson Cancer Center. Pfizer provided inotuzumab ozogamicin free of charge. Dr. Kantarjian and two other researchers reported receiving grants from Pfizer.
Source: Kantarjian H et al. Lancet Oncol. 2018 Jan 15. doi: 10.1016/S1470-2945(18)30011-1.