User login
Flu, RSV infecting children at staggering rates
There has been no ebb in the flurry of respiratory illnesses infecting America’s youngest children. More pediatric wards across the country are announcing crises as beds reach capacity, the pediatric death toll jumped significantly in the past week, and sometimes children are being infected with more than one virus at a time.
In Oregon, for example, the governor announced an official state of emergency to help hospitals deal with the surge of respiratory viruses. Doernbecher Children’s Hospital in Portland, which houses half of all pediatric ICU beds statewide, moved to “crisis mode” and said every pediatric ICU bed was full, Oregon Public Broadcasting reported.
Last month, pediatricians nationwide called for a similar emergency declaration from the federal government to help them respond to the wave of illnesses, which include influenza and respiratory syncytial virus (RSV).
“What’s concerning to us are not only the number of infections, but the severity of these infections, leading to a high number of emergency room visits and hospitalizations,” pediatric infectious disease expert Oscar G. Gómez-Duarte, MD, PhD, said in a news release.
Five more pediatric deaths due to the flu were logged by the CDC in the past week, bringing the total for pediatric flu deaths this season to 12. Nearly 21 per 100,000 children ages 4 and under are being hospitalized for the flu, which is double the rate for children ages 5-17. Last year at this time, fewer than 1 child under age 4 per 100,000 were being hospitalized for the flu.
RSV rates are also stunningly high.
“If we look at CDC data, the RSV hospitalization rate is 10 times higher than usual for this point in the season,” American Medical Association Vice President Andrea Garcia, JD, said in this week’s AMA podcast. “And 171 out of every 100,000 infants younger than 6 months were hospitalized with RSV for the week ending Nov. 12. That is more than double the RSV hospitalization rate for newborns last year and seven times the rate in 2018, which is the last complete season we saw before the pandemic.”
Dr. Gómez-Duarte said hospitals are admitting children with respiratory illnesses who had otherwise been healthy, and sometimes they are even seeing patients with more than one illness.
“Yes, some children are getting what we call coinfections, where they become infected with more than one virus at a time. In some instances, a child becomes initially infected with flu, begins to recover, and subsequently comes down with rhinovirus (a common cold virus), RSV, or any other respiratory virus,” he said. “These coinfections tend to be more severe than when the child just has one infection.”
A version of this article first appeared on Medscape.com.
There has been no ebb in the flurry of respiratory illnesses infecting America’s youngest children. More pediatric wards across the country are announcing crises as beds reach capacity, the pediatric death toll jumped significantly in the past week, and sometimes children are being infected with more than one virus at a time.
In Oregon, for example, the governor announced an official state of emergency to help hospitals deal with the surge of respiratory viruses. Doernbecher Children’s Hospital in Portland, which houses half of all pediatric ICU beds statewide, moved to “crisis mode” and said every pediatric ICU bed was full, Oregon Public Broadcasting reported.
Last month, pediatricians nationwide called for a similar emergency declaration from the federal government to help them respond to the wave of illnesses, which include influenza and respiratory syncytial virus (RSV).
“What’s concerning to us are not only the number of infections, but the severity of these infections, leading to a high number of emergency room visits and hospitalizations,” pediatric infectious disease expert Oscar G. Gómez-Duarte, MD, PhD, said in a news release.
Five more pediatric deaths due to the flu were logged by the CDC in the past week, bringing the total for pediatric flu deaths this season to 12. Nearly 21 per 100,000 children ages 4 and under are being hospitalized for the flu, which is double the rate for children ages 5-17. Last year at this time, fewer than 1 child under age 4 per 100,000 were being hospitalized for the flu.
RSV rates are also stunningly high.
“If we look at CDC data, the RSV hospitalization rate is 10 times higher than usual for this point in the season,” American Medical Association Vice President Andrea Garcia, JD, said in this week’s AMA podcast. “And 171 out of every 100,000 infants younger than 6 months were hospitalized with RSV for the week ending Nov. 12. That is more than double the RSV hospitalization rate for newborns last year and seven times the rate in 2018, which is the last complete season we saw before the pandemic.”
Dr. Gómez-Duarte said hospitals are admitting children with respiratory illnesses who had otherwise been healthy, and sometimes they are even seeing patients with more than one illness.
“Yes, some children are getting what we call coinfections, where they become infected with more than one virus at a time. In some instances, a child becomes initially infected with flu, begins to recover, and subsequently comes down with rhinovirus (a common cold virus), RSV, or any other respiratory virus,” he said. “These coinfections tend to be more severe than when the child just has one infection.”
A version of this article first appeared on Medscape.com.
There has been no ebb in the flurry of respiratory illnesses infecting America’s youngest children. More pediatric wards across the country are announcing crises as beds reach capacity, the pediatric death toll jumped significantly in the past week, and sometimes children are being infected with more than one virus at a time.
In Oregon, for example, the governor announced an official state of emergency to help hospitals deal with the surge of respiratory viruses. Doernbecher Children’s Hospital in Portland, which houses half of all pediatric ICU beds statewide, moved to “crisis mode” and said every pediatric ICU bed was full, Oregon Public Broadcasting reported.
Last month, pediatricians nationwide called for a similar emergency declaration from the federal government to help them respond to the wave of illnesses, which include influenza and respiratory syncytial virus (RSV).
“What’s concerning to us are not only the number of infections, but the severity of these infections, leading to a high number of emergency room visits and hospitalizations,” pediatric infectious disease expert Oscar G. Gómez-Duarte, MD, PhD, said in a news release.
Five more pediatric deaths due to the flu were logged by the CDC in the past week, bringing the total for pediatric flu deaths this season to 12. Nearly 21 per 100,000 children ages 4 and under are being hospitalized for the flu, which is double the rate for children ages 5-17. Last year at this time, fewer than 1 child under age 4 per 100,000 were being hospitalized for the flu.
RSV rates are also stunningly high.
“If we look at CDC data, the RSV hospitalization rate is 10 times higher than usual for this point in the season,” American Medical Association Vice President Andrea Garcia, JD, said in this week’s AMA podcast. “And 171 out of every 100,000 infants younger than 6 months were hospitalized with RSV for the week ending Nov. 12. That is more than double the RSV hospitalization rate for newborns last year and seven times the rate in 2018, which is the last complete season we saw before the pandemic.”
Dr. Gómez-Duarte said hospitals are admitting children with respiratory illnesses who had otherwise been healthy, and sometimes they are even seeing patients with more than one illness.
“Yes, some children are getting what we call coinfections, where they become infected with more than one virus at a time. In some instances, a child becomes initially infected with flu, begins to recover, and subsequently comes down with rhinovirus (a common cold virus), RSV, or any other respiratory virus,” he said. “These coinfections tend to be more severe than when the child just has one infection.”
A version of this article first appeared on Medscape.com.
Buzzy Lancet long COVID paper under investigation for ‘data errors’
An editorial that accompanied the paper when it was published in January of last year described it as “the first large cohort study with 6-months’ follow-up” of people hospitalized with COVID-19. The article has received plenty of attention since then.
Titled “6-month consequences of COVID-19 in patients discharged from hospital: a cohort study,” the paper has been cited nearly 1,600 times, according to Clarivate’s Web of Science. Altmetric finds references to it in multiple documents from the World Health Organization.
According to the expression of concern, dated November 24, a reader found inconsistencies between the data in the article and a later paper describing the same cohort of patients after a year of follow-up. That discovery sparked an investigation that is still ongoing:
- On Jan 8, 2021, The Lancet published an Article, 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study, by Chaolin Huang and colleagues. 1 On Aug 28, 2021, The Lancet published an Article, 1-year outcomes in hospital survivors with COVID-19: a longitudinal cohort study, by Lixue Huang and colleagues. 2 We received an inquiry from a researcher on data inconsistencies between these two Articles, and we sought an explanation from the corresponding author of the two papers. On Nov 7, 2022, Lancet editors were informed that inconsistencies between the 6-month and the 1-year data were due to “some variables in the dataset used for the 6-month paper were mistakenly disrupted in order”. In view of the extent of these data errors, we now issue an Expression of Concern about the 6-month paper 1 while we investigate further, including further statistical and clinical review of the corrected data. We will update this notice as soon as we have further information.
The corresponding author of both papers, Bin Cao of China’s National Center for Respiratory Medicine and the China-Japan Friendship Hospital in Beijing, has not responded to our request for comment.
A profile of Cao published in Lancet Infectious Diseases last March described him as “a leading researcher in pneumonia and influenza” who “has been instrumental in increasing knowledge about COVID-19.” In addition to the follow-up study of hospitalized COVID patients:
- Cao’s seminal papers during the COVID-19 pandemic include the first report of the clinical characteristics of COVID-19 patients in Wuhan, the description of the risk factors for mortality for adult inpatients, and the results of trials testing the use of antiviral drugs, including lopinavir-ritonavir, to treat COVID-19 in China.
We reached out to The Lancet’s press office and Richard Horton, the journal’s editor-in-chief, and received this statement:
- The Lancet Group treats all communications between editors and authors or readers as confidential. Investigations are continuing, and the Expression of Concern will be updated as soon as we have further information to share. More information about our policies is available here:
This year, The Lancet overtook the New England Journal of Medicine as the medical journal with the highest impact factor, in large part due to the papers it published about COVID-19.
We’ve counted retractions for three of those papers, most notably a paper about the use of the drug hydroxychloroquine that claimed to use medical data from a company called Surgisphere. As Retraction Watch readers may remember, the article was retracted after sleuths questioned if the data were real, and the company would not produce it for review.
This article first appeared on Retraction Watch.
An editorial that accompanied the paper when it was published in January of last year described it as “the first large cohort study with 6-months’ follow-up” of people hospitalized with COVID-19. The article has received plenty of attention since then.
Titled “6-month consequences of COVID-19 in patients discharged from hospital: a cohort study,” the paper has been cited nearly 1,600 times, according to Clarivate’s Web of Science. Altmetric finds references to it in multiple documents from the World Health Organization.
According to the expression of concern, dated November 24, a reader found inconsistencies between the data in the article and a later paper describing the same cohort of patients after a year of follow-up. That discovery sparked an investigation that is still ongoing:
- On Jan 8, 2021, The Lancet published an Article, 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study, by Chaolin Huang and colleagues. 1 On Aug 28, 2021, The Lancet published an Article, 1-year outcomes in hospital survivors with COVID-19: a longitudinal cohort study, by Lixue Huang and colleagues. 2 We received an inquiry from a researcher on data inconsistencies between these two Articles, and we sought an explanation from the corresponding author of the two papers. On Nov 7, 2022, Lancet editors were informed that inconsistencies between the 6-month and the 1-year data were due to “some variables in the dataset used for the 6-month paper were mistakenly disrupted in order”. In view of the extent of these data errors, we now issue an Expression of Concern about the 6-month paper 1 while we investigate further, including further statistical and clinical review of the corrected data. We will update this notice as soon as we have further information.
The corresponding author of both papers, Bin Cao of China’s National Center for Respiratory Medicine and the China-Japan Friendship Hospital in Beijing, has not responded to our request for comment.
A profile of Cao published in Lancet Infectious Diseases last March described him as “a leading researcher in pneumonia and influenza” who “has been instrumental in increasing knowledge about COVID-19.” In addition to the follow-up study of hospitalized COVID patients:
- Cao’s seminal papers during the COVID-19 pandemic include the first report of the clinical characteristics of COVID-19 patients in Wuhan, the description of the risk factors for mortality for adult inpatients, and the results of trials testing the use of antiviral drugs, including lopinavir-ritonavir, to treat COVID-19 in China.
We reached out to The Lancet’s press office and Richard Horton, the journal’s editor-in-chief, and received this statement:
- The Lancet Group treats all communications between editors and authors or readers as confidential. Investigations are continuing, and the Expression of Concern will be updated as soon as we have further information to share. More information about our policies is available here:
This year, The Lancet overtook the New England Journal of Medicine as the medical journal with the highest impact factor, in large part due to the papers it published about COVID-19.
We’ve counted retractions for three of those papers, most notably a paper about the use of the drug hydroxychloroquine that claimed to use medical data from a company called Surgisphere. As Retraction Watch readers may remember, the article was retracted after sleuths questioned if the data were real, and the company would not produce it for review.
This article first appeared on Retraction Watch.
An editorial that accompanied the paper when it was published in January of last year described it as “the first large cohort study with 6-months’ follow-up” of people hospitalized with COVID-19. The article has received plenty of attention since then.
Titled “6-month consequences of COVID-19 in patients discharged from hospital: a cohort study,” the paper has been cited nearly 1,600 times, according to Clarivate’s Web of Science. Altmetric finds references to it in multiple documents from the World Health Organization.
According to the expression of concern, dated November 24, a reader found inconsistencies between the data in the article and a later paper describing the same cohort of patients after a year of follow-up. That discovery sparked an investigation that is still ongoing:
- On Jan 8, 2021, The Lancet published an Article, 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study, by Chaolin Huang and colleagues. 1 On Aug 28, 2021, The Lancet published an Article, 1-year outcomes in hospital survivors with COVID-19: a longitudinal cohort study, by Lixue Huang and colleagues. 2 We received an inquiry from a researcher on data inconsistencies between these two Articles, and we sought an explanation from the corresponding author of the two papers. On Nov 7, 2022, Lancet editors were informed that inconsistencies between the 6-month and the 1-year data were due to “some variables in the dataset used for the 6-month paper were mistakenly disrupted in order”. In view of the extent of these data errors, we now issue an Expression of Concern about the 6-month paper 1 while we investigate further, including further statistical and clinical review of the corrected data. We will update this notice as soon as we have further information.
The corresponding author of both papers, Bin Cao of China’s National Center for Respiratory Medicine and the China-Japan Friendship Hospital in Beijing, has not responded to our request for comment.
A profile of Cao published in Lancet Infectious Diseases last March described him as “a leading researcher in pneumonia and influenza” who “has been instrumental in increasing knowledge about COVID-19.” In addition to the follow-up study of hospitalized COVID patients:
- Cao’s seminal papers during the COVID-19 pandemic include the first report of the clinical characteristics of COVID-19 patients in Wuhan, the description of the risk factors for mortality for adult inpatients, and the results of trials testing the use of antiviral drugs, including lopinavir-ritonavir, to treat COVID-19 in China.
We reached out to The Lancet’s press office and Richard Horton, the journal’s editor-in-chief, and received this statement:
- The Lancet Group treats all communications between editors and authors or readers as confidential. Investigations are continuing, and the Expression of Concern will be updated as soon as we have further information to share. More information about our policies is available here:
This year, The Lancet overtook the New England Journal of Medicine as the medical journal with the highest impact factor, in large part due to the papers it published about COVID-19.
We’ve counted retractions for three of those papers, most notably a paper about the use of the drug hydroxychloroquine that claimed to use medical data from a company called Surgisphere. As Retraction Watch readers may remember, the article was retracted after sleuths questioned if the data were real, and the company would not produce it for review.
This article first appeared on Retraction Watch.
Covid vax prevents death in children regardless of variant
The vaccine’s effectiveness against infection in the short term has been established, as has the waning effectiveness of the vaccine over time, wrote Juan Manuel Castelli, MD, of the Ministry of Health of Argentina, Buenos Aires, and colleagues, in the British Medical Journal.
However, data on the impact of vaccine effectiveness on mortality in children and adolescents are limited, especially during periods of omicron variant dominance, the researchers said.
In their new study, the researchers reviewed data from 844,460 children and adolescents aged 3-17 years from the National Surveillance System and the Nominalized Federal Vaccination Registry of Argentina, during a time that included a period of omicron dominance.
Argentina began vaccinating adolescents aged 12-17 years against COVID-19 in August 2021 and added children aged 3-11 years in October 2021. Those aged 12-17 years who were considered fully vaccinated received two doses of either Pfizer-BioNTech and/or Moderna vaccines, and fully-vaccinated 3- to 11-year-olds received two doses of Sinopharm vaccine.
The average time from the second vaccine dose to a COVID-19 test was 66 days for those aged 12-17 years and 54 days for 3- to 11-year-olds. The researchers matched COVID-19 cases with uninfected controls, and a total of 139,321 cases were included in the analysis.
Overall, the estimated vaccine effectiveness against COVID-19 was 64.2% during a period of delta dominance (61.2% in children aged 3-11 years and 66.8% in adolescents aged 12-17 years).
During a period of omicron dominance, estimated vaccine effectiveness was 19.9% across all ages (15.9% and 26.0% for younger and older age groups, respectively).
Effectiveness of the vaccine decreased over time, regardless of the dominant variant, but the decline was greater during the omicron dominant period, the researchers noted. During the omicron period, effectiveness in children aged 3-11 years decreased from 37.6% at 15-30 days postvaccination to 2.0% at 60 days or longer after vaccination. In adolescents aged 12-17 years, vaccine effectiveness during the omicron period decreased from 55.8% at 15-30 days postvaccination to 12.4% at 60 days or longer after vaccination.
Despite the waning protection against infection, the vaccine’s effectiveness against death from COVID-19 was 66.9% in children aged 3-11 years and 97.6% in adolescents aged 12-17 during the period of omicron dominance, the researchers noted.
The results are consistent with similar studies showing a decreased vaccine effectiveness against infection but a persistent effectiveness against deaths over time, the researchers wrote in the discussion section of their paper.
“Our results suggest that the primary vaccination schedule is effective in preventing mortality in children and adolescents with COVID-19 regardless of the circulating SARS-CoV-2 variant,” the researchers said.
Study limitations and strengths
The study was limited by several factors including the incomplete data on symptoms and hospital admissions, the possible impact of unmeasured confounding variables, and the observational design that prevents conclusions of causality, the researchers noted. However, the results were strengthened by the large sample size and access to detailed vaccination records, they said.
Both heterologous and homologous mRNA vaccine schedules showed similar effectiveness in preventing short-term infection and mortality from COVID-19 during periods of differing dominant variants, they noted.
The study findings support the vaccination of children against COVID-19 as an important public health measure to prevent mortality in children and adolescents, they concluded.
Data support value of vaccination, outside experts say
“COVID vaccines may not be as effective over time as the gene variants in the SARS-CoV-2 virus change,” Adrienne G. Randolph, MD, a pediatrician at Harvard Medical School and Boston Children’s Hospital, said in an interview. “Therefore, it is essential to assess vaccine effectiveness over time to look at effectiveness against variants and duration of effectiveness.” Dr. Randolph, who was not involved in the study, said she was not surprised by the findings, which she described as consistent with data from the United States. “COVID vaccines are very effective against preventing life-threatening disease, but the effectiveness against less severe illness for COVID vaccines is not as effective against Omicron,” she noted.
The take-home message for clinicians is that it’s important to get children vaccinated against COVID to prevent severe and life-threatening illness, said Dr. Randolph. “Although these cases are uncommon in children, it is not possible to predict which children will be the most severely affected by COVID,” she emphasized.
However, “we need more data on the new COVID booster vaccines in children that are designed to be more effective against Omicron’s newer variants,” Dr. Randolph said in an interview. “We also need more data on COVID vaccine effectiveness in the youngest children, under 5 years of age, and data on vaccinating mothers to prevent COVID in infants,” she said.
Tim Joos, MD, a Seattle-based clinician who practices a combination of internal medicine and pediatrics, agreed that future research should continue to assess how the new COVID boosters are faring against new variants, noting that the current study did not include data from children who received the new bivalent vaccine.
“The methodology of this study uses a test negative case control design which is common for estimating vaccine effectiveness post-release of a vaccine, but is subject to biases,” Dr. Joos explained. “These are not the clean effectiveness numbers of the prospective randomized control trials that we are used to hearing about when a vaccine is first being approved.”
“Nevertheless, the study reinforces the initial manufacturers’ studies that the vaccines are effective at preventing infection in the pediatric population,” Dr. Joos said in an interview. The current study also reinforces the effectiveness of vaccines in preventing “the rare but devastating mortality from COVID-19 in the pediatric population.”
Commenting on other research showing an increasing ratio of COVID deaths among vaccinated individuals compared to total COVID deaths, he noted that this finding is “likely reflecting a denominator effect of rapidly declining COVID deaths overall,” partly from the vaccines and partly from immunity after previous natural infection.
The study received no outside funding. The researchers, Dr. Randolph, and Dr. Joos had no financial conflicts to disclose. Dr. Joos serves on the Editorial Advisory Board of Pediatric News.
The vaccine’s effectiveness against infection in the short term has been established, as has the waning effectiveness of the vaccine over time, wrote Juan Manuel Castelli, MD, of the Ministry of Health of Argentina, Buenos Aires, and colleagues, in the British Medical Journal.
However, data on the impact of vaccine effectiveness on mortality in children and adolescents are limited, especially during periods of omicron variant dominance, the researchers said.
In their new study, the researchers reviewed data from 844,460 children and adolescents aged 3-17 years from the National Surveillance System and the Nominalized Federal Vaccination Registry of Argentina, during a time that included a period of omicron dominance.
Argentina began vaccinating adolescents aged 12-17 years against COVID-19 in August 2021 and added children aged 3-11 years in October 2021. Those aged 12-17 years who were considered fully vaccinated received two doses of either Pfizer-BioNTech and/or Moderna vaccines, and fully-vaccinated 3- to 11-year-olds received two doses of Sinopharm vaccine.
The average time from the second vaccine dose to a COVID-19 test was 66 days for those aged 12-17 years and 54 days for 3- to 11-year-olds. The researchers matched COVID-19 cases with uninfected controls, and a total of 139,321 cases were included in the analysis.
Overall, the estimated vaccine effectiveness against COVID-19 was 64.2% during a period of delta dominance (61.2% in children aged 3-11 years and 66.8% in adolescents aged 12-17 years).
During a period of omicron dominance, estimated vaccine effectiveness was 19.9% across all ages (15.9% and 26.0% for younger and older age groups, respectively).
Effectiveness of the vaccine decreased over time, regardless of the dominant variant, but the decline was greater during the omicron dominant period, the researchers noted. During the omicron period, effectiveness in children aged 3-11 years decreased from 37.6% at 15-30 days postvaccination to 2.0% at 60 days or longer after vaccination. In adolescents aged 12-17 years, vaccine effectiveness during the omicron period decreased from 55.8% at 15-30 days postvaccination to 12.4% at 60 days or longer after vaccination.
Despite the waning protection against infection, the vaccine’s effectiveness against death from COVID-19 was 66.9% in children aged 3-11 years and 97.6% in adolescents aged 12-17 during the period of omicron dominance, the researchers noted.
The results are consistent with similar studies showing a decreased vaccine effectiveness against infection but a persistent effectiveness against deaths over time, the researchers wrote in the discussion section of their paper.
“Our results suggest that the primary vaccination schedule is effective in preventing mortality in children and adolescents with COVID-19 regardless of the circulating SARS-CoV-2 variant,” the researchers said.
Study limitations and strengths
The study was limited by several factors including the incomplete data on symptoms and hospital admissions, the possible impact of unmeasured confounding variables, and the observational design that prevents conclusions of causality, the researchers noted. However, the results were strengthened by the large sample size and access to detailed vaccination records, they said.
Both heterologous and homologous mRNA vaccine schedules showed similar effectiveness in preventing short-term infection and mortality from COVID-19 during periods of differing dominant variants, they noted.
The study findings support the vaccination of children against COVID-19 as an important public health measure to prevent mortality in children and adolescents, they concluded.
Data support value of vaccination, outside experts say
“COVID vaccines may not be as effective over time as the gene variants in the SARS-CoV-2 virus change,” Adrienne G. Randolph, MD, a pediatrician at Harvard Medical School and Boston Children’s Hospital, said in an interview. “Therefore, it is essential to assess vaccine effectiveness over time to look at effectiveness against variants and duration of effectiveness.” Dr. Randolph, who was not involved in the study, said she was not surprised by the findings, which she described as consistent with data from the United States. “COVID vaccines are very effective against preventing life-threatening disease, but the effectiveness against less severe illness for COVID vaccines is not as effective against Omicron,” she noted.
The take-home message for clinicians is that it’s important to get children vaccinated against COVID to prevent severe and life-threatening illness, said Dr. Randolph. “Although these cases are uncommon in children, it is not possible to predict which children will be the most severely affected by COVID,” she emphasized.
However, “we need more data on the new COVID booster vaccines in children that are designed to be more effective against Omicron’s newer variants,” Dr. Randolph said in an interview. “We also need more data on COVID vaccine effectiveness in the youngest children, under 5 years of age, and data on vaccinating mothers to prevent COVID in infants,” she said.
Tim Joos, MD, a Seattle-based clinician who practices a combination of internal medicine and pediatrics, agreed that future research should continue to assess how the new COVID boosters are faring against new variants, noting that the current study did not include data from children who received the new bivalent vaccine.
“The methodology of this study uses a test negative case control design which is common for estimating vaccine effectiveness post-release of a vaccine, but is subject to biases,” Dr. Joos explained. “These are not the clean effectiveness numbers of the prospective randomized control trials that we are used to hearing about when a vaccine is first being approved.”
“Nevertheless, the study reinforces the initial manufacturers’ studies that the vaccines are effective at preventing infection in the pediatric population,” Dr. Joos said in an interview. The current study also reinforces the effectiveness of vaccines in preventing “the rare but devastating mortality from COVID-19 in the pediatric population.”
Commenting on other research showing an increasing ratio of COVID deaths among vaccinated individuals compared to total COVID deaths, he noted that this finding is “likely reflecting a denominator effect of rapidly declining COVID deaths overall,” partly from the vaccines and partly from immunity after previous natural infection.
The study received no outside funding. The researchers, Dr. Randolph, and Dr. Joos had no financial conflicts to disclose. Dr. Joos serves on the Editorial Advisory Board of Pediatric News.
The vaccine’s effectiveness against infection in the short term has been established, as has the waning effectiveness of the vaccine over time, wrote Juan Manuel Castelli, MD, of the Ministry of Health of Argentina, Buenos Aires, and colleagues, in the British Medical Journal.
However, data on the impact of vaccine effectiveness on mortality in children and adolescents are limited, especially during periods of omicron variant dominance, the researchers said.
In their new study, the researchers reviewed data from 844,460 children and adolescents aged 3-17 years from the National Surveillance System and the Nominalized Federal Vaccination Registry of Argentina, during a time that included a period of omicron dominance.
Argentina began vaccinating adolescents aged 12-17 years against COVID-19 in August 2021 and added children aged 3-11 years in October 2021. Those aged 12-17 years who were considered fully vaccinated received two doses of either Pfizer-BioNTech and/or Moderna vaccines, and fully-vaccinated 3- to 11-year-olds received two doses of Sinopharm vaccine.
The average time from the second vaccine dose to a COVID-19 test was 66 days for those aged 12-17 years and 54 days for 3- to 11-year-olds. The researchers matched COVID-19 cases with uninfected controls, and a total of 139,321 cases were included in the analysis.
Overall, the estimated vaccine effectiveness against COVID-19 was 64.2% during a period of delta dominance (61.2% in children aged 3-11 years and 66.8% in adolescents aged 12-17 years).
During a period of omicron dominance, estimated vaccine effectiveness was 19.9% across all ages (15.9% and 26.0% for younger and older age groups, respectively).
Effectiveness of the vaccine decreased over time, regardless of the dominant variant, but the decline was greater during the omicron dominant period, the researchers noted. During the omicron period, effectiveness in children aged 3-11 years decreased from 37.6% at 15-30 days postvaccination to 2.0% at 60 days or longer after vaccination. In adolescents aged 12-17 years, vaccine effectiveness during the omicron period decreased from 55.8% at 15-30 days postvaccination to 12.4% at 60 days or longer after vaccination.
Despite the waning protection against infection, the vaccine’s effectiveness against death from COVID-19 was 66.9% in children aged 3-11 years and 97.6% in adolescents aged 12-17 during the period of omicron dominance, the researchers noted.
The results are consistent with similar studies showing a decreased vaccine effectiveness against infection but a persistent effectiveness against deaths over time, the researchers wrote in the discussion section of their paper.
“Our results suggest that the primary vaccination schedule is effective in preventing mortality in children and adolescents with COVID-19 regardless of the circulating SARS-CoV-2 variant,” the researchers said.
Study limitations and strengths
The study was limited by several factors including the incomplete data on symptoms and hospital admissions, the possible impact of unmeasured confounding variables, and the observational design that prevents conclusions of causality, the researchers noted. However, the results were strengthened by the large sample size and access to detailed vaccination records, they said.
Both heterologous and homologous mRNA vaccine schedules showed similar effectiveness in preventing short-term infection and mortality from COVID-19 during periods of differing dominant variants, they noted.
The study findings support the vaccination of children against COVID-19 as an important public health measure to prevent mortality in children and adolescents, they concluded.
Data support value of vaccination, outside experts say
“COVID vaccines may not be as effective over time as the gene variants in the SARS-CoV-2 virus change,” Adrienne G. Randolph, MD, a pediatrician at Harvard Medical School and Boston Children’s Hospital, said in an interview. “Therefore, it is essential to assess vaccine effectiveness over time to look at effectiveness against variants and duration of effectiveness.” Dr. Randolph, who was not involved in the study, said she was not surprised by the findings, which she described as consistent with data from the United States. “COVID vaccines are very effective against preventing life-threatening disease, but the effectiveness against less severe illness for COVID vaccines is not as effective against Omicron,” she noted.
The take-home message for clinicians is that it’s important to get children vaccinated against COVID to prevent severe and life-threatening illness, said Dr. Randolph. “Although these cases are uncommon in children, it is not possible to predict which children will be the most severely affected by COVID,” she emphasized.
However, “we need more data on the new COVID booster vaccines in children that are designed to be more effective against Omicron’s newer variants,” Dr. Randolph said in an interview. “We also need more data on COVID vaccine effectiveness in the youngest children, under 5 years of age, and data on vaccinating mothers to prevent COVID in infants,” she said.
Tim Joos, MD, a Seattle-based clinician who practices a combination of internal medicine and pediatrics, agreed that future research should continue to assess how the new COVID boosters are faring against new variants, noting that the current study did not include data from children who received the new bivalent vaccine.
“The methodology of this study uses a test negative case control design which is common for estimating vaccine effectiveness post-release of a vaccine, but is subject to biases,” Dr. Joos explained. “These are not the clean effectiveness numbers of the prospective randomized control trials that we are used to hearing about when a vaccine is first being approved.”
“Nevertheless, the study reinforces the initial manufacturers’ studies that the vaccines are effective at preventing infection in the pediatric population,” Dr. Joos said in an interview. The current study also reinforces the effectiveness of vaccines in preventing “the rare but devastating mortality from COVID-19 in the pediatric population.”
Commenting on other research showing an increasing ratio of COVID deaths among vaccinated individuals compared to total COVID deaths, he noted that this finding is “likely reflecting a denominator effect of rapidly declining COVID deaths overall,” partly from the vaccines and partly from immunity after previous natural infection.
The study received no outside funding. The researchers, Dr. Randolph, and Dr. Joos had no financial conflicts to disclose. Dr. Joos serves on the Editorial Advisory Board of Pediatric News.
FROM THE BMJ
The surprising failure of vitamin D in deficient kids
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
U.S. flu activity already at mid-season levels
according to the Centers of Disease Control and Prevention.
Nationally, 6% of all outpatient visits were because of flu or flu-like illness for the week of Nov. 13-19, up from 5.8% the previous week, the CDC’s Influenza Division said in its weekly FluView report.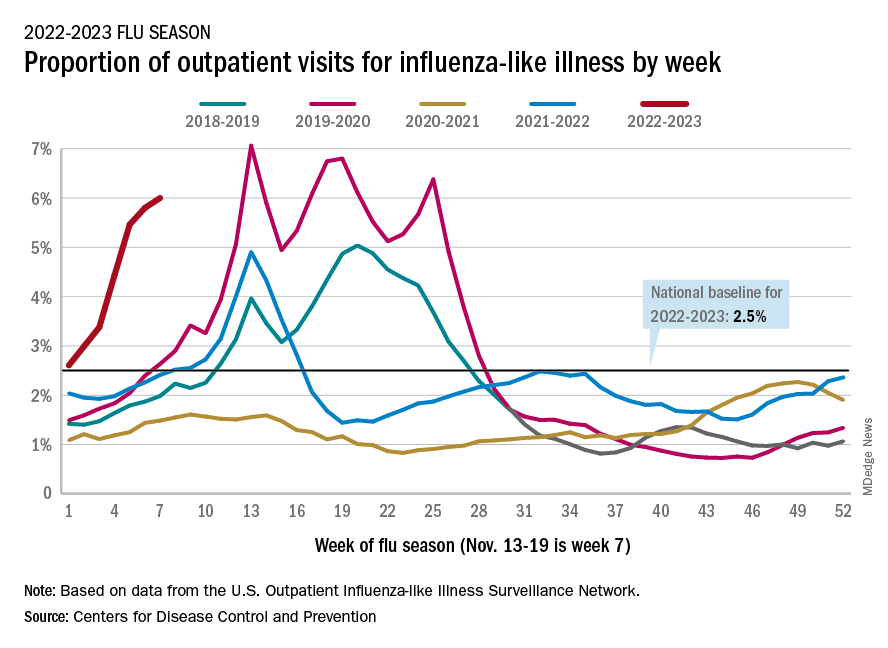
Those figures are the highest recorded in November since 2009, but the peak of the 2009-10 flu season occurred even earlier – the week of Oct. 18-24 – and the rate of flu-like illness had already dropped to just over 4.0% by Nov. 15-21 that year and continued to drop thereafter.
Although COVID-19 and respiratory syncytial virus (RSV) are included in the data from the CDC’s Outpatient Influenza-like Illness Surveillance Network, the agency did note that “seasonal influenza activity is elevated across the country” and estimated that “there have been at least 6.2 million illnesses, 53,000 hospitalizations, and 2,900 deaths from flu” during the 2022-23 season.
Total flu deaths include 11 reported in children as of Nov. 19, and children ages 0-4 had a higher proportion of visits for flu like-illness than other age groups.
The agency also said the cumulative hospitalization rate of 11.3 per 100,000 population “is higher than the rate observed in [the corresponding week of] every previous season since 2010-2011.” Adults 65 years and older have the highest cumulative rate, 25.9 per 100,000, for this year, compared with 20.7 for children 0-4; 11.1 for adults 50-64; 10.3 for children 5-17; and 5.6 for adults 18-49 years old, the CDC said.
A version of this article first appeared on WebMD.com.
according to the Centers of Disease Control and Prevention.
Nationally, 6% of all outpatient visits were because of flu or flu-like illness for the week of Nov. 13-19, up from 5.8% the previous week, the CDC’s Influenza Division said in its weekly FluView report.
Those figures are the highest recorded in November since 2009, but the peak of the 2009-10 flu season occurred even earlier – the week of Oct. 18-24 – and the rate of flu-like illness had already dropped to just over 4.0% by Nov. 15-21 that year and continued to drop thereafter.
Although COVID-19 and respiratory syncytial virus (RSV) are included in the data from the CDC’s Outpatient Influenza-like Illness Surveillance Network, the agency did note that “seasonal influenza activity is elevated across the country” and estimated that “there have been at least 6.2 million illnesses, 53,000 hospitalizations, and 2,900 deaths from flu” during the 2022-23 season.
Total flu deaths include 11 reported in children as of Nov. 19, and children ages 0-4 had a higher proportion of visits for flu like-illness than other age groups.
The agency also said the cumulative hospitalization rate of 11.3 per 100,000 population “is higher than the rate observed in [the corresponding week of] every previous season since 2010-2011.” Adults 65 years and older have the highest cumulative rate, 25.9 per 100,000, for this year, compared with 20.7 for children 0-4; 11.1 for adults 50-64; 10.3 for children 5-17; and 5.6 for adults 18-49 years old, the CDC said.
A version of this article first appeared on WebMD.com.
according to the Centers of Disease Control and Prevention.
Nationally, 6% of all outpatient visits were because of flu or flu-like illness for the week of Nov. 13-19, up from 5.8% the previous week, the CDC’s Influenza Division said in its weekly FluView report.
Those figures are the highest recorded in November since 2009, but the peak of the 2009-10 flu season occurred even earlier – the week of Oct. 18-24 – and the rate of flu-like illness had already dropped to just over 4.0% by Nov. 15-21 that year and continued to drop thereafter.
Although COVID-19 and respiratory syncytial virus (RSV) are included in the data from the CDC’s Outpatient Influenza-like Illness Surveillance Network, the agency did note that “seasonal influenza activity is elevated across the country” and estimated that “there have been at least 6.2 million illnesses, 53,000 hospitalizations, and 2,900 deaths from flu” during the 2022-23 season.
Total flu deaths include 11 reported in children as of Nov. 19, and children ages 0-4 had a higher proportion of visits for flu like-illness than other age groups.
The agency also said the cumulative hospitalization rate of 11.3 per 100,000 population “is higher than the rate observed in [the corresponding week of] every previous season since 2010-2011.” Adults 65 years and older have the highest cumulative rate, 25.9 per 100,000, for this year, compared with 20.7 for children 0-4; 11.1 for adults 50-64; 10.3 for children 5-17; and 5.6 for adults 18-49 years old, the CDC said.
A version of this article first appeared on WebMD.com.
More vaccinated people dying of COVID as fewer get booster shots
“We can no longer say this is a pandemic of the unvaccinated,” Kaiser Family Foundation Vice President Cynthia Cox, who conducted the analysis, told The Washington Post.
People who had been vaccinated or boosted made up 58% of COVID-19 deaths in August, the analysis showed. The rate has been on the rise: 23% of coronavirus deaths were among vaccinated people in September 2021, and the vaccinated made up 42% of deaths in January and February 2022, the Post reported.
Research continues to show that people who are vaccinated or boosted have a lower risk of death. The rise in deaths among the vaccinated is the result of three factors, Ms. Cox said.
- A large majority of people in the United States have been vaccinated (267 million people, the said).
- People who are at the greatest risk of dying from COVID-19 are more likely to be vaccinated and boosted, such as the elderly.
- Vaccines lose their effectiveness over time; the virus changes to avoid vaccines; and people need to choose to get boosters to continue to be protected.
The case for the effectiveness of vaccines and boosters versus skipping the shots remains strong. People age 6 months and older who are unvaccinated are six times more likely to die of COVID-19, compared to those who got the primary series of shots, the Post reported. Survival rates were even better with additional booster shots, particularly among older people.
“I feel very confident that if people continue to get vaccinated at good numbers, if people get boosted, we can absolutely have a very safe and healthy holiday season,” Ashish Jha, White House coronavirus czar, said on Nov. 22.
The number of Americans who have gotten the most recent booster has been increasing ahead of the holidays. CDC data show that 12% of the U.S. population age 5 and older has received a booster.
A new study by a team of researchers from Harvard University and Yale University estimates that 94% of the U.S. population has been infected with COVID-19 at least once, leaving just 1 in 20 people who have never had the virus.
“Despite these high exposure numbers, there is still substantial population susceptibility to infection with an Omicron variant,” the authors wrote.
They said that if all states achieved the vaccination levels of Vermont, where 55% of people had at least one booster and 22% got a second one, there would be “an appreciable improvement in population immunity, with greater relative impact for protection against infection versus severe disease. This additional protection results from both the recovery of immunity lost due to waning and the increased effectiveness of the bivalent booster against Omicron infections.”
A version of this article first appeared on WebMD.com.
“We can no longer say this is a pandemic of the unvaccinated,” Kaiser Family Foundation Vice President Cynthia Cox, who conducted the analysis, told The Washington Post.
People who had been vaccinated or boosted made up 58% of COVID-19 deaths in August, the analysis showed. The rate has been on the rise: 23% of coronavirus deaths were among vaccinated people in September 2021, and the vaccinated made up 42% of deaths in January and February 2022, the Post reported.
Research continues to show that people who are vaccinated or boosted have a lower risk of death. The rise in deaths among the vaccinated is the result of three factors, Ms. Cox said.
- A large majority of people in the United States have been vaccinated (267 million people, the said).
- People who are at the greatest risk of dying from COVID-19 are more likely to be vaccinated and boosted, such as the elderly.
- Vaccines lose their effectiveness over time; the virus changes to avoid vaccines; and people need to choose to get boosters to continue to be protected.
The case for the effectiveness of vaccines and boosters versus skipping the shots remains strong. People age 6 months and older who are unvaccinated are six times more likely to die of COVID-19, compared to those who got the primary series of shots, the Post reported. Survival rates were even better with additional booster shots, particularly among older people.
“I feel very confident that if people continue to get vaccinated at good numbers, if people get boosted, we can absolutely have a very safe and healthy holiday season,” Ashish Jha, White House coronavirus czar, said on Nov. 22.
The number of Americans who have gotten the most recent booster has been increasing ahead of the holidays. CDC data show that 12% of the U.S. population age 5 and older has received a booster.
A new study by a team of researchers from Harvard University and Yale University estimates that 94% of the U.S. population has been infected with COVID-19 at least once, leaving just 1 in 20 people who have never had the virus.
“Despite these high exposure numbers, there is still substantial population susceptibility to infection with an Omicron variant,” the authors wrote.
They said that if all states achieved the vaccination levels of Vermont, where 55% of people had at least one booster and 22% got a second one, there would be “an appreciable improvement in population immunity, with greater relative impact for protection against infection versus severe disease. This additional protection results from both the recovery of immunity lost due to waning and the increased effectiveness of the bivalent booster against Omicron infections.”
A version of this article first appeared on WebMD.com.
“We can no longer say this is a pandemic of the unvaccinated,” Kaiser Family Foundation Vice President Cynthia Cox, who conducted the analysis, told The Washington Post.
People who had been vaccinated or boosted made up 58% of COVID-19 deaths in August, the analysis showed. The rate has been on the rise: 23% of coronavirus deaths were among vaccinated people in September 2021, and the vaccinated made up 42% of deaths in January and February 2022, the Post reported.
Research continues to show that people who are vaccinated or boosted have a lower risk of death. The rise in deaths among the vaccinated is the result of three factors, Ms. Cox said.
- A large majority of people in the United States have been vaccinated (267 million people, the said).
- People who are at the greatest risk of dying from COVID-19 are more likely to be vaccinated and boosted, such as the elderly.
- Vaccines lose their effectiveness over time; the virus changes to avoid vaccines; and people need to choose to get boosters to continue to be protected.
The case for the effectiveness of vaccines and boosters versus skipping the shots remains strong. People age 6 months and older who are unvaccinated are six times more likely to die of COVID-19, compared to those who got the primary series of shots, the Post reported. Survival rates were even better with additional booster shots, particularly among older people.
“I feel very confident that if people continue to get vaccinated at good numbers, if people get boosted, we can absolutely have a very safe and healthy holiday season,” Ashish Jha, White House coronavirus czar, said on Nov. 22.
The number of Americans who have gotten the most recent booster has been increasing ahead of the holidays. CDC data show that 12% of the U.S. population age 5 and older has received a booster.
A new study by a team of researchers from Harvard University and Yale University estimates that 94% of the U.S. population has been infected with COVID-19 at least once, leaving just 1 in 20 people who have never had the virus.
“Despite these high exposure numbers, there is still substantial population susceptibility to infection with an Omicron variant,” the authors wrote.
They said that if all states achieved the vaccination levels of Vermont, where 55% of people had at least one booster and 22% got a second one, there would be “an appreciable improvement in population immunity, with greater relative impact for protection against infection versus severe disease. This additional protection results from both the recovery of immunity lost due to waning and the increased effectiveness of the bivalent booster against Omicron infections.”
A version of this article first appeared on WebMD.com.
OSA overlap impairs functional performance in COPD
Obstructive sleep apnea (OSA) was associated with both impaired functional performance during exercise and overall worse outcomes in patients with chronic obstructive pulmonary disease (COPD), based on data from 34 adults.
Individuals with COPD are at increased risk for hospital readmissions and disease exacerbations, Patricia Faria Camargo, PhD, of Federal University of São Carlos (Brazil), and colleagues wrote. These patients often have concomitant obstructive sleep apnea, which itself can promote adverse cardiovascular events, but the impact of the overlap of these two conditions on clinical outcomes has not been explored.
In a study published in Heart & Lung, the researchers recruited 17 adults with COPD only and 17 with OSA and COPD. At baseline, patients underwent pulmonary function tests, echocardiography, and polysomnography to confirm their OSA and COPD diagnoses.
The primary endpoint was the impact of OSA on functional performance and cardiac autonomic control in COPD patients, based on measures of heart rate variability and the 6-minute walk test (6MWT). Participants were followed for 1 year, with telephone contacts every 3 months. A secondary endpoint was the number of exacerbations, hospitalizations, and deaths. At baseline, OSA-COPD patients had worse polysomnographic function, compared with COPD patients; they also tended to be older and have higher body mass index, but other demographics were similar between the groups.
(P = .003), as measured by the 6MWT. The OSA-COPD patients also showed significantly worse autonomic response during exercise, compared with the COPD group.
A lower work load during exercise and the interaction between group and time factors suggests that OSA impacts the exercise capacity of COPD patients, the researchers said. Notably, however, neither age nor body mass index was associated with functional performance in the OSA-COPD group.
Patients in the OSA-COPD group also were significantly more likely to experience exacerbations during the study period, compared with the COPD-only group (67.4% vs. 23.5; P = .03). However, the severity of COPD was similar between the groups, which further illustrates that OSA can impair functional performance in COPD patients, the researchers said.
The findings were limited by several factors including the small sample size and restricted collection of follow-up data during the pandemic, the researchers noted. However, the results support previous studies, and suggest that overlapping OSA and COPD produces worse outcomes.
“Future studies can confirm our findings, providing new clinical evidences to the assessment of sleep quality in COPD patients and its implications for the general health status of these individuals, in addition to contributing to more assertive clinical and therapeutic alternative support the need for more research into the mechanisms behind this overlap in larger samples to develop treatment alternatives,” they concluded.
The study was supported by the Federal University of Sao Carlos. The researchers had no financial conflicts to disclose.
Obstructive sleep apnea (OSA) was associated with both impaired functional performance during exercise and overall worse outcomes in patients with chronic obstructive pulmonary disease (COPD), based on data from 34 adults.
Individuals with COPD are at increased risk for hospital readmissions and disease exacerbations, Patricia Faria Camargo, PhD, of Federal University of São Carlos (Brazil), and colleagues wrote. These patients often have concomitant obstructive sleep apnea, which itself can promote adverse cardiovascular events, but the impact of the overlap of these two conditions on clinical outcomes has not been explored.
In a study published in Heart & Lung, the researchers recruited 17 adults with COPD only and 17 with OSA and COPD. At baseline, patients underwent pulmonary function tests, echocardiography, and polysomnography to confirm their OSA and COPD diagnoses.
The primary endpoint was the impact of OSA on functional performance and cardiac autonomic control in COPD patients, based on measures of heart rate variability and the 6-minute walk test (6MWT). Participants were followed for 1 year, with telephone contacts every 3 months. A secondary endpoint was the number of exacerbations, hospitalizations, and deaths. At baseline, OSA-COPD patients had worse polysomnographic function, compared with COPD patients; they also tended to be older and have higher body mass index, but other demographics were similar between the groups.
(P = .003), as measured by the 6MWT. The OSA-COPD patients also showed significantly worse autonomic response during exercise, compared with the COPD group.
A lower work load during exercise and the interaction between group and time factors suggests that OSA impacts the exercise capacity of COPD patients, the researchers said. Notably, however, neither age nor body mass index was associated with functional performance in the OSA-COPD group.
Patients in the OSA-COPD group also were significantly more likely to experience exacerbations during the study period, compared with the COPD-only group (67.4% vs. 23.5; P = .03). However, the severity of COPD was similar between the groups, which further illustrates that OSA can impair functional performance in COPD patients, the researchers said.
The findings were limited by several factors including the small sample size and restricted collection of follow-up data during the pandemic, the researchers noted. However, the results support previous studies, and suggest that overlapping OSA and COPD produces worse outcomes.
“Future studies can confirm our findings, providing new clinical evidences to the assessment of sleep quality in COPD patients and its implications for the general health status of these individuals, in addition to contributing to more assertive clinical and therapeutic alternative support the need for more research into the mechanisms behind this overlap in larger samples to develop treatment alternatives,” they concluded.
The study was supported by the Federal University of Sao Carlos. The researchers had no financial conflicts to disclose.
Obstructive sleep apnea (OSA) was associated with both impaired functional performance during exercise and overall worse outcomes in patients with chronic obstructive pulmonary disease (COPD), based on data from 34 adults.
Individuals with COPD are at increased risk for hospital readmissions and disease exacerbations, Patricia Faria Camargo, PhD, of Federal University of São Carlos (Brazil), and colleagues wrote. These patients often have concomitant obstructive sleep apnea, which itself can promote adverse cardiovascular events, but the impact of the overlap of these two conditions on clinical outcomes has not been explored.
In a study published in Heart & Lung, the researchers recruited 17 adults with COPD only and 17 with OSA and COPD. At baseline, patients underwent pulmonary function tests, echocardiography, and polysomnography to confirm their OSA and COPD diagnoses.
The primary endpoint was the impact of OSA on functional performance and cardiac autonomic control in COPD patients, based on measures of heart rate variability and the 6-minute walk test (6MWT). Participants were followed for 1 year, with telephone contacts every 3 months. A secondary endpoint was the number of exacerbations, hospitalizations, and deaths. At baseline, OSA-COPD patients had worse polysomnographic function, compared with COPD patients; they also tended to be older and have higher body mass index, but other demographics were similar between the groups.
(P = .003), as measured by the 6MWT. The OSA-COPD patients also showed significantly worse autonomic response during exercise, compared with the COPD group.
A lower work load during exercise and the interaction between group and time factors suggests that OSA impacts the exercise capacity of COPD patients, the researchers said. Notably, however, neither age nor body mass index was associated with functional performance in the OSA-COPD group.
Patients in the OSA-COPD group also were significantly more likely to experience exacerbations during the study period, compared with the COPD-only group (67.4% vs. 23.5; P = .03). However, the severity of COPD was similar between the groups, which further illustrates that OSA can impair functional performance in COPD patients, the researchers said.
The findings were limited by several factors including the small sample size and restricted collection of follow-up data during the pandemic, the researchers noted. However, the results support previous studies, and suggest that overlapping OSA and COPD produces worse outcomes.
“Future studies can confirm our findings, providing new clinical evidences to the assessment of sleep quality in COPD patients and its implications for the general health status of these individuals, in addition to contributing to more assertive clinical and therapeutic alternative support the need for more research into the mechanisms behind this overlap in larger samples to develop treatment alternatives,” they concluded.
The study was supported by the Federal University of Sao Carlos. The researchers had no financial conflicts to disclose.
FROM HEART & LUNG
COPD care bundle curbs all-cause readmissions
at 30, 60, and 90 days, based on data from approximately 300 patients.
COPD remains a leading cause of mortality and a leading contributor to health care costs, but data suggest that adoption of an interdisciplinary care bundle could reduce hospital readmission for COPD patients, Sibyl Cherian, PharmD, BCPS, of Overlook Medical Center, Summit, N.J., and colleagues wrote. The Centers for Medicare & Medicaid Services has introduced both penalties and bundled payments for hospitals with excess all-cause readmission rates after hospitalizations, but more data are needed on the ability of a COPD care bundle to reduce readmission for COPD.
In a study published in the Journal of the American Pharmacists Association, the researchers assigned 127 individuals with COPD to a COPD care bundle arm and 189 to a control arm for treatment at a single center. The standard of care group was admitted between Jan. 1 and Dec. 31, 2017; the COPD care bundle group was admitted between Jan. 1 and Dec. 31, 2018. The mean age of the participants across both groups was 72 years, and more than 70% of patients in each group were White. The COPD care bundle was managed by a team including pulmonologists, hospitalists, care managers, advanced practice nurses, pharmacists, respiratory care practitioners, physical therapists, documentation specialists, quality improvement experts, social workers, and dietitians.
The primary outcome was 30-day all-cause readmission among adults with acute exacerbation of COPD.
Overall, the rate of 30-day all-cause readmissions was significantly lower in the COPD care bundle arm versus the control arm (11.8% vs. 21.7%; P = .017). Similar differences appeared between the care bundle group and control group for all-cause readmissions at 60 days (8.7% vs. 18%; P = .013) and 90 days (4.7% vs. 19.6%; P < .001).
Reasons for reduced readmissions after implementation of the COPD care bundle included pulmonary follow-up appointments of 7 days or less, significantly increased physical therapy consults, and significant escalation of COPD maintenance therapy, the researchers wrote.
Notably, pharmacists consulted with 68.5% of patients overall and assisted with access to outpatient medications for 45.7% of those in the care bundle arm, the researchers wrote. Patients in the COPD care bundle group were significantly more likely to have an escalation in maintenance therapy versus the control patients (44.9% vs. 22.2%; P < .001), which illustrates the importance of interventions by pharmacists in escalating therapy to reduce readmissions.
The study findings were limited by several factors including the retrospective design and use of data from a single center, the researchers noted. Other limitations included the lack of data on the need for therapy escalation in the control group and by the lack of controlling for socioeconomic status, which is a known risk factor for hospital readmission.
However, the results support the value of a COPD care bundle for reducing readmissions, and that such a bundle can be replicated at other hospitals, although more research is needed to evaluate the impact of other COPD care strategies, they emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose.
at 30, 60, and 90 days, based on data from approximately 300 patients.
COPD remains a leading cause of mortality and a leading contributor to health care costs, but data suggest that adoption of an interdisciplinary care bundle could reduce hospital readmission for COPD patients, Sibyl Cherian, PharmD, BCPS, of Overlook Medical Center, Summit, N.J., and colleagues wrote. The Centers for Medicare & Medicaid Services has introduced both penalties and bundled payments for hospitals with excess all-cause readmission rates after hospitalizations, but more data are needed on the ability of a COPD care bundle to reduce readmission for COPD.
In a study published in the Journal of the American Pharmacists Association, the researchers assigned 127 individuals with COPD to a COPD care bundle arm and 189 to a control arm for treatment at a single center. The standard of care group was admitted between Jan. 1 and Dec. 31, 2017; the COPD care bundle group was admitted between Jan. 1 and Dec. 31, 2018. The mean age of the participants across both groups was 72 years, and more than 70% of patients in each group were White. The COPD care bundle was managed by a team including pulmonologists, hospitalists, care managers, advanced practice nurses, pharmacists, respiratory care practitioners, physical therapists, documentation specialists, quality improvement experts, social workers, and dietitians.
The primary outcome was 30-day all-cause readmission among adults with acute exacerbation of COPD.
Overall, the rate of 30-day all-cause readmissions was significantly lower in the COPD care bundle arm versus the control arm (11.8% vs. 21.7%; P = .017). Similar differences appeared between the care bundle group and control group for all-cause readmissions at 60 days (8.7% vs. 18%; P = .013) and 90 days (4.7% vs. 19.6%; P < .001).
Reasons for reduced readmissions after implementation of the COPD care bundle included pulmonary follow-up appointments of 7 days or less, significantly increased physical therapy consults, and significant escalation of COPD maintenance therapy, the researchers wrote.
Notably, pharmacists consulted with 68.5% of patients overall and assisted with access to outpatient medications for 45.7% of those in the care bundle arm, the researchers wrote. Patients in the COPD care bundle group were significantly more likely to have an escalation in maintenance therapy versus the control patients (44.9% vs. 22.2%; P < .001), which illustrates the importance of interventions by pharmacists in escalating therapy to reduce readmissions.
The study findings were limited by several factors including the retrospective design and use of data from a single center, the researchers noted. Other limitations included the lack of data on the need for therapy escalation in the control group and by the lack of controlling for socioeconomic status, which is a known risk factor for hospital readmission.
However, the results support the value of a COPD care bundle for reducing readmissions, and that such a bundle can be replicated at other hospitals, although more research is needed to evaluate the impact of other COPD care strategies, they emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose.
at 30, 60, and 90 days, based on data from approximately 300 patients.
COPD remains a leading cause of mortality and a leading contributor to health care costs, but data suggest that adoption of an interdisciplinary care bundle could reduce hospital readmission for COPD patients, Sibyl Cherian, PharmD, BCPS, of Overlook Medical Center, Summit, N.J., and colleagues wrote. The Centers for Medicare & Medicaid Services has introduced both penalties and bundled payments for hospitals with excess all-cause readmission rates after hospitalizations, but more data are needed on the ability of a COPD care bundle to reduce readmission for COPD.
In a study published in the Journal of the American Pharmacists Association, the researchers assigned 127 individuals with COPD to a COPD care bundle arm and 189 to a control arm for treatment at a single center. The standard of care group was admitted between Jan. 1 and Dec. 31, 2017; the COPD care bundle group was admitted between Jan. 1 and Dec. 31, 2018. The mean age of the participants across both groups was 72 years, and more than 70% of patients in each group were White. The COPD care bundle was managed by a team including pulmonologists, hospitalists, care managers, advanced practice nurses, pharmacists, respiratory care practitioners, physical therapists, documentation specialists, quality improvement experts, social workers, and dietitians.
The primary outcome was 30-day all-cause readmission among adults with acute exacerbation of COPD.
Overall, the rate of 30-day all-cause readmissions was significantly lower in the COPD care bundle arm versus the control arm (11.8% vs. 21.7%; P = .017). Similar differences appeared between the care bundle group and control group for all-cause readmissions at 60 days (8.7% vs. 18%; P = .013) and 90 days (4.7% vs. 19.6%; P < .001).
Reasons for reduced readmissions after implementation of the COPD care bundle included pulmonary follow-up appointments of 7 days or less, significantly increased physical therapy consults, and significant escalation of COPD maintenance therapy, the researchers wrote.
Notably, pharmacists consulted with 68.5% of patients overall and assisted with access to outpatient medications for 45.7% of those in the care bundle arm, the researchers wrote. Patients in the COPD care bundle group were significantly more likely to have an escalation in maintenance therapy versus the control patients (44.9% vs. 22.2%; P < .001), which illustrates the importance of interventions by pharmacists in escalating therapy to reduce readmissions.
The study findings were limited by several factors including the retrospective design and use of data from a single center, the researchers noted. Other limitations included the lack of data on the need for therapy escalation in the control group and by the lack of controlling for socioeconomic status, which is a known risk factor for hospital readmission.
However, the results support the value of a COPD care bundle for reducing readmissions, and that such a bundle can be replicated at other hospitals, although more research is needed to evaluate the impact of other COPD care strategies, they emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF THE AMERICAN PHARMACISTS ASSOCIATION
The right indoor relative humidity could ward off COVID
The “sweet spot” associated with reduced COVID-19 cases and deaths is 40%-60% indoor relative humidity, an MIT news release said. People who maintained indoor relative humidity outside those parameters had higher rates of catching COVID-19.
Most people are comfortable with 30%-50% relative humidity, researchers said. An airplane cabin has about 20% relative humidity.
Relative humidity is the amount of moisture in the air, compared with the total moisture the air can hold at a given temperature before saturating and forming condensation.
The study was published in The Journal of the Royal Society Interface. Researchers examined COVID-19 data and meteorological measurements from 121 countries from January 2020 through August 2020, before vaccines became available to the public.
“When outdoor temperatures were below the typical human comfort range, they assumed indoor spaces were heated to reach that comfort range. Based on the added heating, they calculated the associated drop in indoor relative humidity,” the MIT news release said.
The research teams found that when a region reported a rise in COVID-19 cases and deaths, the region’s estimated indoor relative humidity was either lower than 40% or higher than 60%, the release said.
“There’s potentially a protective effect of this intermediate indoor relative humidity,” said Connor Verheyen, the lead author and a PhD student in medical engineering and medical physics in the Harvard-MIT Program in Health Sciences and Technology.
Widespread use of the 40%-60% indoor humidity range could reduce the need for lockdowns and other widespread restrictions, the study concluded.
“Unlike measures that depend on individual compliance (for example, masking or hand-washing), indoor RH optimization would achieve high compliance because all occupants of a common indoor space would be exposed to similar ambient conditions,” the study said. “Compared to the long timelines and high costs of vaccine production and distribution, humidity control systems could potentially be implemented more quickly and cheaply in certain indoor settings.”
A version of this article first appeared on WebMD.com.
The “sweet spot” associated with reduced COVID-19 cases and deaths is 40%-60% indoor relative humidity, an MIT news release said. People who maintained indoor relative humidity outside those parameters had higher rates of catching COVID-19.
Most people are comfortable with 30%-50% relative humidity, researchers said. An airplane cabin has about 20% relative humidity.
Relative humidity is the amount of moisture in the air, compared with the total moisture the air can hold at a given temperature before saturating and forming condensation.
The study was published in The Journal of the Royal Society Interface. Researchers examined COVID-19 data and meteorological measurements from 121 countries from January 2020 through August 2020, before vaccines became available to the public.
“When outdoor temperatures were below the typical human comfort range, they assumed indoor spaces were heated to reach that comfort range. Based on the added heating, they calculated the associated drop in indoor relative humidity,” the MIT news release said.
The research teams found that when a region reported a rise in COVID-19 cases and deaths, the region’s estimated indoor relative humidity was either lower than 40% or higher than 60%, the release said.
“There’s potentially a protective effect of this intermediate indoor relative humidity,” said Connor Verheyen, the lead author and a PhD student in medical engineering and medical physics in the Harvard-MIT Program in Health Sciences and Technology.
Widespread use of the 40%-60% indoor humidity range could reduce the need for lockdowns and other widespread restrictions, the study concluded.
“Unlike measures that depend on individual compliance (for example, masking or hand-washing), indoor RH optimization would achieve high compliance because all occupants of a common indoor space would be exposed to similar ambient conditions,” the study said. “Compared to the long timelines and high costs of vaccine production and distribution, humidity control systems could potentially be implemented more quickly and cheaply in certain indoor settings.”
A version of this article first appeared on WebMD.com.
The “sweet spot” associated with reduced COVID-19 cases and deaths is 40%-60% indoor relative humidity, an MIT news release said. People who maintained indoor relative humidity outside those parameters had higher rates of catching COVID-19.
Most people are comfortable with 30%-50% relative humidity, researchers said. An airplane cabin has about 20% relative humidity.
Relative humidity is the amount of moisture in the air, compared with the total moisture the air can hold at a given temperature before saturating and forming condensation.
The study was published in The Journal of the Royal Society Interface. Researchers examined COVID-19 data and meteorological measurements from 121 countries from January 2020 through August 2020, before vaccines became available to the public.
“When outdoor temperatures were below the typical human comfort range, they assumed indoor spaces were heated to reach that comfort range. Based on the added heating, they calculated the associated drop in indoor relative humidity,” the MIT news release said.
The research teams found that when a region reported a rise in COVID-19 cases and deaths, the region’s estimated indoor relative humidity was either lower than 40% or higher than 60%, the release said.
“There’s potentially a protective effect of this intermediate indoor relative humidity,” said Connor Verheyen, the lead author and a PhD student in medical engineering and medical physics in the Harvard-MIT Program in Health Sciences and Technology.
Widespread use of the 40%-60% indoor humidity range could reduce the need for lockdowns and other widespread restrictions, the study concluded.
“Unlike measures that depend on individual compliance (for example, masking or hand-washing), indoor RH optimization would achieve high compliance because all occupants of a common indoor space would be exposed to similar ambient conditions,” the study said. “Compared to the long timelines and high costs of vaccine production and distribution, humidity control systems could potentially be implemented more quickly and cheaply in certain indoor settings.”
A version of this article first appeared on WebMD.com.
FROM THE JOURNAL OF THE ROYAL SOCIETY INTERFACE
AAP issues guidelines to combat rise in respiratory illness
Updated guidance from the group outlines measures to optimize resources to manage a surge of patients filling hospital beds, emergency departments, and physicians’ practices.
A separate document from the AAP endorses giving extra doses of palivizumab, a monoclonal antibody used to prevent severe infection in infants at high risk of respiratory syncytial virus (RSV), as long as the illness is prevalent in the community.
Upticks in rates of RSV and influenza, along with a crisis in children’s mental health, prompted the AAP and the Children’s Hospital Association to petition the Biden administration on Nov. 14 to declare an emergency. Such a move would free up extra funding and waivers to allow physicians and hospitals to pool resources, the organizations said.
Despite those challenges, the AAP stressed in its new guidance that routine care, such as immunizations and chronic disease management, “cannot be neglected.”
Shifting resources
Officials at some children’s hospitals said that they have already implemented many of the AAP’s recommended measures for providing care during a surge, such as cross-training staff who usually treat adults, expanding telehealth and urgent care, and optimizing the use of ancillary care spaces.
“A lot of this is just reinforcing the things that I think children’s hospitals have been doing,” Lindsay Ragsdale, MD, chief medical officer for Kentucky Children’s Hospital, Lexington, said. “Can we shift adults around? Can we use an adult unit? Can we use an occupied space creatively? We’re really thinking outside the box.”
Andrew Pavia, MD, chief of the division of pediatric infectious diseases at University of Utah Health, Salt Lake City, said large children’s hospitals have been actively sharing practices for handling a surge through various channels, but the new guidance could be a useful “checklist” for small hospitals and physician practices that lack well-developed plans.
The AAP’s suggestions for pediatricians in outpatient settings include stocking up on personal protective equipment, using social media and office staff to increase communication with families, and keeping abreast of wait times at local emergency departments.
Addressing a subset of kids
In updated guidance for palivizumab, the AAP noted that earlier-than-usual circulation of RSV prompted pediatricians in some areas to begin administering the drug in the summer and early fall.
Palivizumab is typically given in five consecutive monthly intramuscular injections during RSV season, starting in November. Eligible infants and young children include those born prematurely or who have conditions such as chronic lung disease, hemodynamically significant congenital heart disease, or a suppressed immune system.
The AAP said it supports giving extra doses if RSV activity “persists at high levels in a given region through the fall and winter.” Published studies are sparse but contain “no evidence of increased frequency or severity of adverse events with later doses in a five-dose series nor with doses beyond five doses,” the group added.
The guidance may encourage payers to pick up the tab for extra doses, which are priced at more than $1,800 for cash customers, Dr. Pavia said. However, that recommendation addresses “a pretty small part of the problem overall because the injections are used for a very small subset of kids who are at the highest risk, and more than 80% of hospitalizations for RSV are among healthy kids,” he added.
Dr. Ragsdale and Dr. Pavia have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Updated guidance from the group outlines measures to optimize resources to manage a surge of patients filling hospital beds, emergency departments, and physicians’ practices.
A separate document from the AAP endorses giving extra doses of palivizumab, a monoclonal antibody used to prevent severe infection in infants at high risk of respiratory syncytial virus (RSV), as long as the illness is prevalent in the community.
Upticks in rates of RSV and influenza, along with a crisis in children’s mental health, prompted the AAP and the Children’s Hospital Association to petition the Biden administration on Nov. 14 to declare an emergency. Such a move would free up extra funding and waivers to allow physicians and hospitals to pool resources, the organizations said.
Despite those challenges, the AAP stressed in its new guidance that routine care, such as immunizations and chronic disease management, “cannot be neglected.”
Shifting resources
Officials at some children’s hospitals said that they have already implemented many of the AAP’s recommended measures for providing care during a surge, such as cross-training staff who usually treat adults, expanding telehealth and urgent care, and optimizing the use of ancillary care spaces.
“A lot of this is just reinforcing the things that I think children’s hospitals have been doing,” Lindsay Ragsdale, MD, chief medical officer for Kentucky Children’s Hospital, Lexington, said. “Can we shift adults around? Can we use an adult unit? Can we use an occupied space creatively? We’re really thinking outside the box.”
Andrew Pavia, MD, chief of the division of pediatric infectious diseases at University of Utah Health, Salt Lake City, said large children’s hospitals have been actively sharing practices for handling a surge through various channels, but the new guidance could be a useful “checklist” for small hospitals and physician practices that lack well-developed plans.
The AAP’s suggestions for pediatricians in outpatient settings include stocking up on personal protective equipment, using social media and office staff to increase communication with families, and keeping abreast of wait times at local emergency departments.
Addressing a subset of kids
In updated guidance for palivizumab, the AAP noted that earlier-than-usual circulation of RSV prompted pediatricians in some areas to begin administering the drug in the summer and early fall.
Palivizumab is typically given in five consecutive monthly intramuscular injections during RSV season, starting in November. Eligible infants and young children include those born prematurely or who have conditions such as chronic lung disease, hemodynamically significant congenital heart disease, or a suppressed immune system.
The AAP said it supports giving extra doses if RSV activity “persists at high levels in a given region through the fall and winter.” Published studies are sparse but contain “no evidence of increased frequency or severity of adverse events with later doses in a five-dose series nor with doses beyond five doses,” the group added.
The guidance may encourage payers to pick up the tab for extra doses, which are priced at more than $1,800 for cash customers, Dr. Pavia said. However, that recommendation addresses “a pretty small part of the problem overall because the injections are used for a very small subset of kids who are at the highest risk, and more than 80% of hospitalizations for RSV are among healthy kids,” he added.
Dr. Ragsdale and Dr. Pavia have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Updated guidance from the group outlines measures to optimize resources to manage a surge of patients filling hospital beds, emergency departments, and physicians’ practices.
A separate document from the AAP endorses giving extra doses of palivizumab, a monoclonal antibody used to prevent severe infection in infants at high risk of respiratory syncytial virus (RSV), as long as the illness is prevalent in the community.
Upticks in rates of RSV and influenza, along with a crisis in children’s mental health, prompted the AAP and the Children’s Hospital Association to petition the Biden administration on Nov. 14 to declare an emergency. Such a move would free up extra funding and waivers to allow physicians and hospitals to pool resources, the organizations said.
Despite those challenges, the AAP stressed in its new guidance that routine care, such as immunizations and chronic disease management, “cannot be neglected.”
Shifting resources
Officials at some children’s hospitals said that they have already implemented many of the AAP’s recommended measures for providing care during a surge, such as cross-training staff who usually treat adults, expanding telehealth and urgent care, and optimizing the use of ancillary care spaces.
“A lot of this is just reinforcing the things that I think children’s hospitals have been doing,” Lindsay Ragsdale, MD, chief medical officer for Kentucky Children’s Hospital, Lexington, said. “Can we shift adults around? Can we use an adult unit? Can we use an occupied space creatively? We’re really thinking outside the box.”
Andrew Pavia, MD, chief of the division of pediatric infectious diseases at University of Utah Health, Salt Lake City, said large children’s hospitals have been actively sharing practices for handling a surge through various channels, but the new guidance could be a useful “checklist” for small hospitals and physician practices that lack well-developed plans.
The AAP’s suggestions for pediatricians in outpatient settings include stocking up on personal protective equipment, using social media and office staff to increase communication with families, and keeping abreast of wait times at local emergency departments.
Addressing a subset of kids
In updated guidance for palivizumab, the AAP noted that earlier-than-usual circulation of RSV prompted pediatricians in some areas to begin administering the drug in the summer and early fall.
Palivizumab is typically given in five consecutive monthly intramuscular injections during RSV season, starting in November. Eligible infants and young children include those born prematurely or who have conditions such as chronic lung disease, hemodynamically significant congenital heart disease, or a suppressed immune system.
The AAP said it supports giving extra doses if RSV activity “persists at high levels in a given region through the fall and winter.” Published studies are sparse but contain “no evidence of increased frequency or severity of adverse events with later doses in a five-dose series nor with doses beyond five doses,” the group added.
The guidance may encourage payers to pick up the tab for extra doses, which are priced at more than $1,800 for cash customers, Dr. Pavia said. However, that recommendation addresses “a pretty small part of the problem overall because the injections are used for a very small subset of kids who are at the highest risk, and more than 80% of hospitalizations for RSV are among healthy kids,” he added.
Dr. Ragsdale and Dr. Pavia have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.