User login
Advanced Melanoma: First-line Therapy
Malignant melanoma is the most serious form of primary skin cancer and one of the only malignancies in which the incidence rate has been rising. It is estimated that in 2018 there were 91,270 newly diagnosed cases and 9320 deaths from advanced melanoma in the United States. Melanoma is the fifth most common cancer type in males and the sixth most common in females. Despite rising incidence rates, improvement in the treatment of advanced melanoma has resulted in declining death rates over the past decade.1 Although most melanoma is diagnosed at an early stage and can be cured with surgical excision, the prognosis for metastatic melanoma had been historically poor prior to recent advancements in treatment. Conventional chemotherapy treatment with dacarbazine or temozolomide resulted in response rates ranging from 7.5% to 12.1%, but without much impact on median overall survival (OS), with reported OS ranging from 6.4 to 7.8 months. Combination approaches with interferon alfa-2B and low-dose interleukin-2 resulted in improved response rates compared with traditional chemotherapy, but again without survival benefit.2
Immunotherapy in the form of high-dose interleukin-2 emerged as the first therapy to alter the natural history of advanced melanoma, with both improved response rates (objective response rate [ORR], 16%) and median OS (2 months), with some patients achieving durable responses lasting more than 30 months. However, significant systemic toxicity limited its application to carefully selected patients.3 The past decade has brought rapid advancements in treatment with immune checkpoint inhibitors and molecularly targeted agents, which have significantly improved ORRs, progression-free survival (PFS), and OS for patients with metastatic melanoma.4-8
This review is the first of 2 articles focusing on the treatment and sequencing of therapies in advanced melanoma. Here, we review the selection of first-line therapy for metastatic melanoma. Current evidence for immune checkpoint blockade and molecularly targeted agents in the treatment of metastatic melanoma after progression on first-line therapy is discussed in a separate article.
Pathogenesis
The incidence of melanoma is strongly associated with ultraviolet light–mediated DNA damage related to sun exposure. Specifically, melanoma is associated to a greater degree with intense intermittent sun exposure and sunburn, but not associated with higher occupational exposure.9 Ultraviolet radiation can induce DNA damage by a number of mechanisms, and deficient DNA repair leads to somatic mutations that drive the progression from normal melanocyte to melanoma.10
The most commonly identified genetic mutations in cutaneous melanomas are alterations in the mitogen-activated protein kinase (MAPK) pathway. Typically, an extracellular growth factor causes dimerization of the growth factor receptor, which activates the intracellular RAS GTPase protein. Subsequently BRAF is phosphorylated within the kinase domain, which leads to downstream activation of the MEK and ERK kinases through phosphorylation. Activated ERK leads to phosphorylation of various cytoplasmic and nuclear targets, and the downstream effects of these changes promote cellular proliferation. While activation of this pathway usually requires phosphorylation of BRAF by RAS, mutations placing an acidic amino acid near the kinase domain mimics phosphorylation and leads to constitutive activation of the BRAF serine/threonine kinase in the absence of upstream signaling from extracellular growth factors mediated through RAS.11 One study of tumor samples of 71 patients with cutaneous melanoma detected NRAS mutations in 30% and BRAF mutations in 59% of all tumors tested. Of the BRAF mutation–positive tumors, 88% harbored the Val599Glu mutation, now commonly referred to as the BRAF V600E mutation. The same study demonstrated that the vast majority of BRAF mutations were seen in the primary tumor and were preserved when metastases were analyzed. Additionally, both NRAS and BRAF mutations were detected in the radial growth phase of the melanoma tumor. These findings indicate that alterations in the MAPK pathway occur early in the pathogenesis of advanced melanoma.11 Another group demonstrated that 66% of malignant melanoma tumor samples harbored BRAF mutations, of which 80% were specifically the V600E mutation. In vitro assays showed that the BRAF V600E–mutated kinase had greater than 10-fold kinase activity compared to wild-type BRAF, and that this kinase enhanced cellular proliferation even when upstream NRAS signaling was inhibited.12
The Cancer Genome Atlas Network performed a large analysis of tumor samples from 331 different melanoma patients and studied variations at the DNA, RNA, and protein levels. The study established a framework of 4 notable genomic subtypes, including mutant BRAF (52%), mutant RAS (28%), mutant NF1 (14%), and triple wild-type (6%). Additionally, mRNA transcriptomic analysis of overexpressed genes identified 3 different subclasses, which were labeled as “immune,” “keratin,” and “MITF-low.” The immune subclass was characterized by increased expression of proteins found in immune cells, immune signaling molecules, immune checkpoint proteins, cytokines, and chemokines, and correlated with increased lymphocyte invasion within the tumor. Interestingly, in the post-accession survival analysis, the “immune” transcriptomic subclass was statistically correlated with an improved prognosis.13 Having an understanding of the molecular pathogenesis of advanced melanoma helps to create a framework for understanding the mechanisms of current standard of care therapies for the disease.
Case Presentation
A 62-year-old Caucasian man with a history of well-controlled type 2 diabetes mellitus and hypertension is being followed by his dermatologist for surveillance of melanocytic nevi. On follow-up he is noted to have an asymmetrical melanocytic lesion over the right scalp with irregular borders and variegated color. He is asymptomatic and the remainder of physical examination is unremarkable, as he has no other concerning skin lesions and no cervical, axillary, or inguinal lymphadenopathy.
How is melanoma diagnosed?
Detailed discussion about diagnosis and staging will be deferred in this review of treatment of advanced melanoma. In brief, melanoma is best diagnosed by excisional biopsy and histopathology. Staging of melanoma is done according to the American Joint Committee on Cancer’s (AJCC) Cancer Staging Manual, 8th edition, using a TNM staging system that incorporates tumor thickness (Breslow depth); ulceration; number of involved regional lymph nodes; presence of in-transit, satellite, and/or microsatellite metastases; distant metastases; and serum lactate dehydrogenase level.14
Case Continued
The patient undergoes a wide excisional biopsy of the right scalp lesion, which is consistent with malignant melanoma. Pathology demonstrates a Breslow depth of 2.6 mm, 2 mitotic figures/mm2, and no evidence of ulceration. He subsequently undergoes wide local excision with 0/3 sentinel lymph nodes positive for malignancy. His final staging is consistent with pT3aN0M0, stage IIA melanoma.
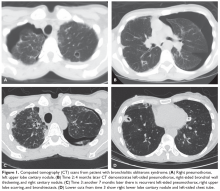
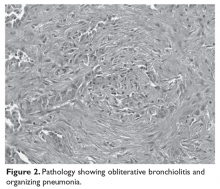
He is seen in follow-up with medical oncology for the next 3.5 years without any evidence of disease recurrence. He then develops symptoms of vertigo, diplopia, and recurrent falls, prompting medical attention. Magnetic resonance imaging (MRI) brain reveals multiple supratentorial and infratentorial lesions concerning for intracranial metastases. Further imaging with computed tomography (CT) chest/abdomen/pelvis reveals a right lower lobe pulmonary mass with right hilar and subcarinal lymphadenopathy. He is admitted for treatment with intravenous dexamethasone and further evaluation with endobronchial ultrasound-guided fine-needle aspiration of the right lower lobe mass, which reveals metastatic melanoma. Given the extent of his intracranial metastases, he is treated with whole brain radiation therapy for symptomatic relief prior to initiating systemic therapy.
What is the general approach to first-line treatment for metastatic melanoma?
The past decade has brought an abundance of data supporting the use of immunotherapy with immune checkpoint inhibitors or molecularly targeted therapy with combined BRAF/MEK inhibitors in the first-line setting.4-8 After the diagnosis of metastatic melanoma has been made, molecular testing is recommended to determine the BRAF status of the tumor. Immunotherapy is the clear choice for first-line therapy in the absence of an activating BRAF V600 mutation. When a BRAF V600 mutation is present, current evidence supports the use of either immunotherapy or molecularly targeted therapy as first-line therapy.
To date, there have been no prospective clinical trials comparing the sequencing of immunotherapy and molecularly targeted therapy in the first-line setting. An ongoing clinical trial (NCT02224781) is comparing dabrafenib and trametinib followed by ipilimumab and nivolumab at time of progression to ipilimumab and nivolumab followed by dabrafenib and trametinib in patients with newly diagnosed stage III/IV BRAF V600 mutation–positive melanoma. The primary outcome measure is 2-year OS. Until completion of that trial, current practice regarding which type of therapy to use in the first-line setting is based on a number of factors including clinical characteristics and provider preferences.
Data suggest that immunotherapies can produce durable responses, especially after treatment completion or discontinuation, albeit at the expense of taking a longer time to achieve clinical benefit and the risk of potentially serious immune-related adverse effects. This idea of a durable, off-treatment response is highlighted by a study that followed 105 patients who had achieved a complete response (CR) and found that 24-month disease-free survival from the time of CR was 90.9% in all patients and 89.9% in the 67 patients who had discontinued pembrolizumab after attaining CR.15 BRAF/MEK inhibition has the potential for rapid clinical responses, though concerns exist about the development of resistance to therapy. The following sections explore the evidence supporting the use of these therapies.
Immunotherapy with Immune Checkpoint Inhibitors
Immunotherapy via immune checkpoint blockade has revolutionized the treatment of many solid tumors over the past decade. The promise of immunotherapy revolves around the potential for achieving a dynamic and durable systemic response against cancer by augmenting the antitumor effects of the immune system. T-cells are central to mounting a systemic antitumor response, and, in addition to antigen recognition, their function depends heavily on fine tuning between co-stimulatory and co-inhibitory signaling. The cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) expressed on T-cells was the first discovered co-inhibitory receptor of T-cell activation.16 Later, it was discovered that the programmed cell death 1 receptor (PD-1), expressed on T-cells, and its ligands PD-L1 and PD-L2, expressed on antigen presenting cells, tumor cells, or other cells in the tumor microenvironment, also served as a potent negative regulator of T-cell function.17
Together, these 2 signaling pathways help to maintain peripheral immune tolerance, whereby autoreactive T-cells that have escaped from the thymus are silenced to prevent autoimmunity. However, these pathways can also be utilized by cancer cells to escape immune surveillance. Monoclonal antibodies that inhibit the aforementioned co-inhibitory signaling pathways, and thus augment the immune response, have proven to be an effective anticancer therapy capable of producing profound and durable responses in certain malignancies.16,17
Ipilimumab
Ipilimumab is a monoclonal antibody that inhibits the function of the CTLA-4 co-inhibitory immune checkpoint. In a phase 3 randomized controlled trial of 676 patients with previously treated metastatic melanoma, ipilimumab at a dose of 3 mg/kg every 3 weeks for 4 cycles, with or without a gp100 peptide vaccine, resulted in an improved median OS of 10.0 and 10.1 months, respectively, compared to 6.4 months in those receiving the peptide vaccine alone, meeting the primary endpoint.4 Subsequently, a phase 3 trial of 502 patients with untreated metastatic melanoma compared ipilimumab at a dose of 10 mg/kg every 3 weeks for 4 cycles plus dacarbazine to dacarbazine plus placebo and found a significant increase in median OS (11.2 months vs 9.1 months), with no additive benefit of chemotherapy. There was a higher reported rate of grade 3 or 4 adverse events in this trial with ipilimumab dosed at 10 mg/kg, which was felt to be dose-related.18 These trials were the first to show improved OS with any systemic therapy in metastatic melanoma and led to US Food and Drug Administration approval of ipilimumab for this indication in 2011.
PD-1 Inhibitor Monotherapy
The PD-1 inhibitors nivolumab and pembrolizumab were initially approved for metastatic melanoma after progression on ipilimumab. In the phase 1 trial of patients with previously treated metastatic melanoma, nivolumab therapy resulted in an ORR of 28%.19 The subsequent phase 2 trial conducted in pretreated patients, including patients who had progressed on ipilimumab, confirmed a similar ORR of 31%, as well as a median PFS of 3.7 months and a median OS of 16.8 months. The estimated response duration in patients who did achieve a response to therapy was 2 years.20 A phase 3 trial (CheckMate 037) comparing nivolumab (n = 120) to investigator’s choice chemotherapy (n = 47) in those with melanoma refractory to ipilimumab demonstrated that nivolumab was superior for the primary endpoint of ORR (31.7% vs 10.6%), had less toxicity (5% rate of grade 3 or 4 adverse events versus 9%), and increased median duration of response (32 months vs 13 months).21
The phase 1 trial (KEYNOTE-001) testing the efficacy of pembrolizumab demonstrated an ORR of 33% in the total population of patients treated and an ORR of 45% in those who were treatment-naive. Additionally, the median OS was 23 months for the total population and 31 months for treatment-naive patients, with only 14% of patients experiencing a grade 3 or 4 adverse event.22 The KEYNOTE-002 phase 2 trial compared 2 different pembrolizumab doses (2 mg/kg and 10 mg/kg every 3 weeks) to investigator’s choice chemotherapy (paclitaxel plus carboplatin, paclitaxel, carboplatin, dacarbazine, or oral temozolomide) in 540 patients with advanced melanoma with documented progression on ipilimumab with or without prior progression on molecularly targeted therapy if positive for a BRAF V600 mutation. The final analysis demonstrated significantly improved ORR with pembrolizumab (22% at 2 mg/kg vs 26% at 10 mg/kg vs 4% chemotherapy) and significantly improved 24-month PFS (16% vs 22% vs 0.6%, respectively). There was a nonstatistically significant improvement in median OS (13.4 months vs 14.7 months vs 10 months), although 55% of the patients initially assigned to the chemotherapy arm crossed over and received pembrolizumab after documentation of progressive disease.23,24
Because PD-1 inhibition improved efficacy with less toxicity than chemotherapy when studied in progressive disease, subsequent studies focused on PD-1 inhibition in the frontline setting. CheckMate 066 was a phase 3 trial comparing nivolumab to dacarbazine as first-line therapy for 418 patients with untreated metastatic melanoma who did not have a BRAF mutation. For the primary end point of 1-year OS, nivolumab was superior to dacarbazine (72.9% vs 42.1%; hazard ratio [HR], 0.42; P < 0.001). Treatment with nivolumab also resulted in superior ORR (40% vs 14%) and PFS (5.1 months vs 2.2 months). Additionally, nivolumab therapy had a lower rate of grade 3 or 4 toxicity compared to dacarbazine (11.7% vs 17.6%).25
The KEYNOTE-006 trial compared 2 separate dosing schedules of pembrolizumab (10 mg/kg every 2 weeks versus every 3 weeks) to ipilimumab (3 mg/kg every 3 weeks for 4 cycles) in a 1:1:1 ratio in 834 patients with metastatic melanoma who had received up to 1 prior systemic therapy, but no prior CTLA-4 or PD-1 inhibitors. The first published data reported statistically significant outcomes for the co-primary end points of 6-month PFS (47.3% for pembrolizumab every 2 weeks vs 46.4% for pembrolizumab every 3 weeks vs 26.5% for ipilimumab; HR, 0.58 for both pembrolizumab groups compared to ipilimumab; P < 0.001) and 12-month OS (74.1% vs 68.4% vs 58.2%) with pembrolizumab compared to ipilimumab. Compared to ipilimumab, pembrolizumab every 2 weeks had a hazard ratio of 0.63 (P = 0.0005) and pembrolizumab every 3 weeks had a hazard ratio of 0.69 (P = 0.0036). The pembrolizumab groups was also had lower rates of grade 3 to 5 toxicity (13.3% vs 10.1% vs 19.9%).5 Updated outcomes demonstrated improved ORR compared to the first analysis (37% vs 36% vs 13%), and improved OS (median OS, not reached for the pembrolizumab groups vs 16.0 months for the ipilimumab group; HR, 0.68, P = 0.0009 for pembrolizumab every 2 weeks versus HR 0.68, P = 0.0008 for pembrolizumab every 3 weeks).26 In addition, 24-month OS was 55% in both pembrolizumab groups compared to 43% in the ipilimumab group. Grade 3 or 4 toxicity occurred less frequently with pembrolizumab (17% vs 17% vs 20%).
Further analysis from the KEYNOTE-006 trial data demonstrated improved ORR, PFS, and OS with pembrolizumab compared to ipilimumab in tumors positive for PD-L1 expression. For PD-L1-negative tumors, response rate was higher, and PFS and OS rates were similar with pembrolizumab compared to ipilimumab. Given that pembrolizumab was associated with similar survival outcomes in PD-L1-negative tumors and with less toxicity than ipilimumab, the superiority of PD-L1 inhibitors over ipilimumab was further supported, regardless of tumor PD-L1 status.27
In sum, PD-1 inhibition should be considered the first-line immunotherapy in advanced melanoma, either alone or in combination with ipilimumab, as discussed in the following section. There is no longer a role for ipilimumab monotherapy in the first-line setting, based on evidence from direct comparison to single-agent PD-1 inhibition in clinical trials that demonstrated superior efficacy and less serious toxicity with PD-1 inhibitors.5,26 The finding that ORR and OS outcomes with single-agent PD-1 inhibitors are higher in treatment-naive patients compared to those receiving prior therapies also supports this approach.22
Combination CTLA-4 and PD-1 Therapy
Despite the potential for durable responses, the majority of patients fail to respond to single-agent PD-1 therapy. Given that preclinical data had suggested the potential for synergy between dual inhibition of CTLA-4 and PD-1, clinical trials were designed to test this approach. The first randomized phase 2 trial that established superior efficacy with combination therapy was the CheckMate 069 trial comparing nivolumab plus ipilimumab to ipilimumab monotherapy. Combination therapy resulted in increased ORR (59% vs 11%), median PFS (not reached vs 3.0 months), 2-year PFS (51.3% vs 12.0%), and 2-year OS (63.8% vs 53.6%).28 Similarly, a phase 1b trial of pembrolizumab plus reduced-dose ipilimumab demonstrated an ORR of 61%, with a 1-year PFS of 69% and 1-year OS of 89%.29
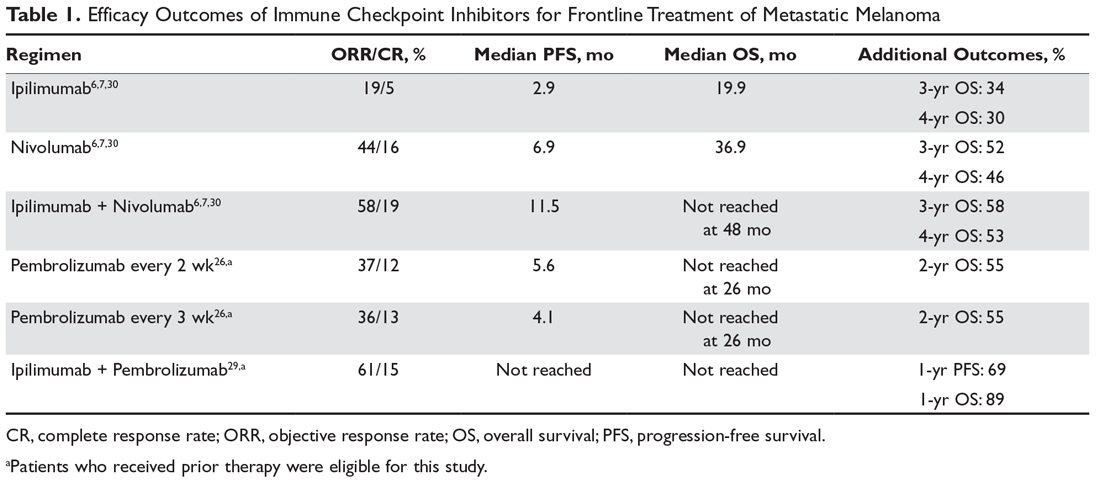
The landmark phase 3 CheckMate 067 trial analyzed efficacy outcomes for 3 different treatment regimens including nivolumab plus ipilimumab, nivolumab monotherapy, and ipilimumab monotherapy in previously untreated patients with unresectable stage III or IV melanoma. The trial was powered to compare survival outcomes for both the combination therapy arm against ipilimumab and the nivolumab monotherapy arm against ipilimumab, but not to compare combination therapy to nivolumab monotherapy. The initial analysis demonstrated a median PFS of 11.5 months with combination therapy versus 6.9 months with nivolumab and 2.9 months with ipilimumab, as well as an ORR of 58% versus 44% and 19%, respectively (Table 1).6 The updated 3-year survival outcomes from CheckMate 067 were notable for superior median OS with combination therapy (not reached in combination vs 37.6 months for nivolumab vs 19.9 months ipilimumab), improved 3-year OS (58% vs 52% vs 34%), and improved 3-year PFS (39% vs 32% vs 10%).7 In the reported 4-year survival outcomes, median OS was not reached in the combination therapy group, and was 36.9 months in the nivolumab monotherapy group and 19.9 months in the ipilimumab monotherapy group. Rates of grade 3 or 4 adverse events were significantly higher in the combination therapy group, at 59% compared to 22% with nivolumab monotherapy and 28% with ipilimumab alone.30 The 3- and 4-year OS outcomes (58% and 54%, respectively) with combination therapy were the highest seen in any phase 3 trial for treatment of advanced melanoma, supporting its use as the best approved first-line therapy in those who can tolerate the potential toxicity of combination therapy7,30 The conclusions from this landmark trial were that both combination therapy and nivolumab monotherapy resulted in statistically significant improvement in OS compared to ipilimumab.
Toxicity Associated with Immune Checkpoint Inhibitors
While immune checkpoint inhibitors have revolutionized the treatment of many solid tumor malignancies, this new class of cancer therapy has brought about a new type of toxicity for clinicians to be aware of, termed immune-related adverse events (irAEs). As immune checkpoint inhibitors amplify the immune response against malignancy, they also increase the likelihood that autoreactive T-cells persist and proliferate within the circulation. Therefore, these therapies can result in almost any type of autoimmune side effect. The most commonly reported irAEs in large clinical trials studying CTLA-4 and PD-1 inhibitors include rash/pruritus, diarrhea/colitis, hepatitis, endocrinopathies (thyroiditis, hypophysitis, adrenalitis), and pneumonitis. Other more rare toxicities include pancreatitis, autoimmune hematologic toxicities, cardiac toxicity (myocarditis, heart failure), and neurologic toxicities (neuropathies, myasthenia gravis-like syndrome, Guillain-Barré syndrome). It has been observed that PD-1 inhibitors have a lower incidence of irAEs than CTLA-4 inhibitors, and that the combined use of PD-1 and CTLA-4 inhibitors is associated with a greater incidence of irAEs compared to monotherapy with either agent.31 Toxicities associated with ipilimumab have been noted to be dose dependent.18 Generally, these toxicities are treated with immunosuppression in the form of glucocorticoids and are often reversible.31 There are several published guidelines that include algorithms for the management of irAEs by organizations such as the National Comprehensive Cancer Network.32
For example, previously untreated patients treated with ipilimumab plus dacarbazine as compared to dacarbazine plus placebo had greater grade 3 or 4 adverse events (56.3% vs 27.5%), and 77.7% of patients experiencing an irAE of any grade.18 In the CheckMate 066 trial comparing frontline nivolumab to dacarbazine, nivolumab had a lower rate of grade 3 or 4 toxicity (11.7% vs 17.6%) and irAEs were relatively infrequent, with diarrhea and elevated alanine aminotransferase level each being the most prominent irAE (affecting 1.0% of patients).25 In the KEYNOTE-006 trial, irAEs seen in more than 1% of patients treated with pembrolizumab included colitis, hepatitis, hypothyroidism, and hyperthyroidism, whereas those occurring in more than 1% of patients treated with ipilimumab included colitis and hypophysitis. Overall, there were lower rates of grade 3 to 5 toxicity with the 2 pembrolizumab doses compared to ipilimumab (13.3% pembrolizumab every 2 weeks vs 10.1% pembrolizumab every 3 weeks vs 19.9% ipilimumab).5 In the CheckMate 067 trial comparing nivolumab plus ipilimumab, nivolumab monotherapy, and ipilimumab monotherapy, rates of treatment-related adverse events of any grade were higher in the combination group (96% combination vs 86% nivolumab vs 86% ipilimumab), as were rates of grade 3 or 4 adverse events (59% vs 21% vs 28%, respectively). The irAE profile was similar to that demonstrated in prior studies: rash/pruritus were the most common, and diarrhea/colitis, elevated aminotransferases, and endocrinopathies were among the more common irAEs.7
Alternative dosing strategies have been investigated in an effort to preserve efficacy and minimize toxicity. A phase 1b trial of pembrolizumab plus reduced-dose ipilimumab demonstrated an ORR of 61%, with a 1-year PFS of 69% and a 1-year OS of 89%. This combination led to 45% of patients having a grade 3 or 4 adverse event, 60% having irAEs of any grade, and only 27% having grade 3 or 4 irAEs.29 The CheckMate 067 trial studied the combination of nivolumab 1 mg/kg plus ipilimumab 3 mg/kg.6 The CheckMate 511 trial compared different combination dosing strategies (nivolumab 3 mg/kg + ipilimumab 1 mg/kg versus nivolumab 1 mg/kg + ipilimumab 3 mg/kg) to assess for safety benefit. In the results published in abstract form, the reduced ipilimumab dose (nivolumab 3 mg/kg + ipilimumab 1 mg/kg arm) resulted in significantly decreased grade 3 to 5 adverse events (33.9% vs 48.3%) without significant differences in ORR, PFS, or OS.33
The question about the efficacy of checkpoint inhibitors in patients who discontinue treatment due to irAEs has been raised, as one hypothesis suggests that such toxicities may also indicate that the antitumor immune response has been activated. In a retrospective pooled analysis of phase 2 and 3 trials where patients received combination therapy with ipilimumab and nivolumab and discontinued therapy during the induction phase due to irAEs, outcomes did not appear to be inferior. Median PFS was 8.4 months in those who discontinued therapy compared to 10.8 months in those who continued therapy, but this did not reach statistical significance. Median OS had not been reached in either group and ORR was actually higher in those who discontinued due to adverse events (58.3% vs 50.2%). While this retrospective analysis needs to be validated, it does suggest that patients likely derive antitumor benefit from immunotherapy even if they have to discontinue therapy due to irAEs. Of note, patients in this analysis were not trialed on nivolumab monotherapy after receiving immunosuppressive treatment for toxicity related to combination therapy, which has since been deemed a reasonable treatment option.34
Molecularly Targeted Therapy for Metastatic Melanoma
As previously mentioned, the MAPK pathway is frequently altered in metastatic melanoma and thus serves as a target for therapy. Mutations in BRAF can cause constitutive activation of the protein’s kinase function, which subsequently phosphorylates/activates MEK in the absence of extracellular growth signals and causes increased cellular proliferation. For the roughly half of patients diagnosed with metastatic melanoma who harbor a BRAF V600 mutation, molecularly targeted therapy with BRAF/MEK inhibitors has emerged as a standard of care treatment option. As such, all patients with advanced disease should be tested for BRAF mutations.
After early phase 1 studies of the BRAF inhibitor vemurafenib demonstrated successful inhibition of mutated BRAF,35 subsequent studies confirmed the benefit of BRAF targeted therapy. In the phase 3 randomized controlled BRIM-3 trial comparing vemurafenib with dacarbazine for treatment of 675 patients with previously untreated metastatic melanoma positive for a BRAF V600E mutation, the vemurafenib group had superior ORR and 6-month OS during the first analysis.36 In a subsequent analysis, median PFS and median OS were also superior with vemurafenib compared to dacarbazine, as vemurafenib had a median OS of 13.6 months compared to 9.7 months with dacarbazine (HR, 0.70; P = 0.0008).37 Dabrafenib was the next BRAF inhibitor to demonstrate clinical efficacy with superior PFS compared to dacarbazine.38
Despite tumor shrinkage in the majority of patients, the development of resistance to therapy was an issue early on. The development of acquired resistance emerged as a heterogeneous process, though many of the identified resistance mechanisms involved reactivation of the MAPK pathway.39 A phase 3 trial of 322 patients with metastatic melanoma comparing the MEK inhibitor trametinib as monotherapy against chemotherapy demonstrated a modest improvement in both median PFS and OS.40 As a result, subsequent efforts focused on a strategy of concurrent MEK inhibition as a means to overcome resistance to molecularly targeted monotherapy
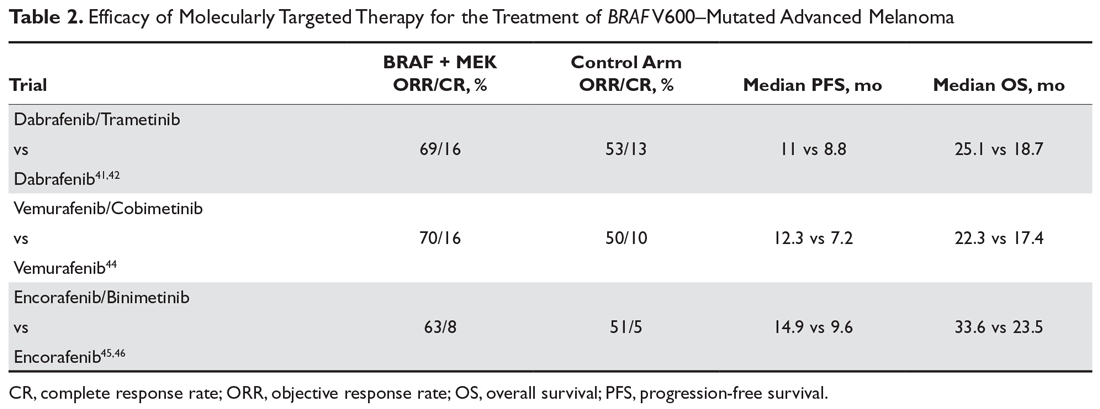
At least 4 large phase 3 randomized controlled trials of combination therapy with BRAF plus MEK inhibitors showed an improved ORR, PFS, and OS when compared to BRAF inhibition alone. The COMBI-d trial comparing dabrafenib plus trametinib versus dabrafenib alone was the first to demonstrate the superiority of combined BRAF/MEK inhibition and made combination therapy the current standard of care for patients with metastatic melanoma and a BRAF V600 mutation. In the final analysis of this trial, 3-year PFS was 22% with combination therapy compared to 12% with dabrafenib alone, and 3-year OS was 44% compared to 32%.8,41,42 A second trial with the combination of dabrafenib and trametinib (COMBI-V) also demonstrated superior efficacy when compared to single-agent vemurafenib without increased toxicity.43 Subsequently, the combination of vemurafenib with MEK inhibitor cobimetinib demonstrated superiority compared to vemurafenib alone,44 followed by the newest combination encorafenib (BRAF inhibitor) and binimetinib (MEK inhibitor) proving superior to either vemurafenib or encorafenib alone.45,46
It is important to note that there have been no studies directly comparing the efficacy of the 3 approved BRAF/MEK inhibitor combinations, but the 3 different regimens have some differences in their toxicity profiles (Table 2). Of note, single-agent BRAF inhibition was associated with increased cutaneous toxicity, including secondary squamous cell carcinoma and keratoacanthoma,47 which was demonstrated to be driven by paradoxical activation of the MAPK pathway.48 The concerning cutaneous toxicities such as squamous cell carcinoma were substantially reduced by combination BRAF/MEK inhibitor therapy.47 Collectively, the higher efficacy along with manageable toxicity profile established combination BRAF/MEK inhibition as the preferred regimen for patients with BRAF-mutated metastatic melanoma who are being considered for molecularly targeted therapy. BRAF inhibitor monotherapy should only be used when there is a specific concern regarding the use of a MEK inhibitor in certain clinical circumstances.
Other driver mutations associated with metastatic melanoma such as NRAS-mutated tumors have proven more difficult to effectively treat with molecularly targeted therapy, with one study showing that the MEK inhibitor binimetinib resulted in a modest improvement in ORR and median PFS without OS benefit compared to dacarbazine.49 Several phase 2 trials involving metastatic melanoma harboring a c-Kit alteration have demonstrated some efficacy with the tyrosine kinase inhibitor imatinib. The largest phase 2 trial of 43 patients treated with imatinib resulted in a 53.5% disease control rate (23.3% partial response and 30.2% stable disease), with 9 of the 10 patients who achieved partial response having a mutation in either exon 11 or 13. Median PFS was 3.5 months and 1-year OS was 51.0%.50
Case Conclusion
Prior to initiation of systemic therapy, the patient’s melanoma is tested and is found to be positive for a BRAF V600K mutation. At his follow-up appointment, the patient continues to endorse generalized weakness, fatigue, issues with balance, and residual pulmonary symptoms after being treated for post-obstructive pneumonia. Given his current symptoms and extent of metastatic disease, immunotherapy is deferred and he is started on combination molecularly targeted therapy with dabrafenib and trametinib. He initially does well, with a partial response noted by resolution of symptoms and decreased size of his intracranial metastases and decreased size of the right lower lobe mass. Further follow-up of this patient is presented in the second article in this 2-part review of advanced melanoma.
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2018. CA Cancer J Clin. 2018;68:7-30.
2. Ives NJ, Stowe RL, Lorigan P, Wheatley K. Chemotherapy compared with biochemotherapy for the treatment of metastatic melanoma: a meta-analysis of 18 trials involving 2621 patients. J Clin Oncol. 2007;25:5426-34.
3. Atkins MB, Lotze MT, Dutcher JP, et al. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J Clin Oncol. 1999;17:2105-16.
4. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-23.
5. Robert C, Schachter J, Long GV, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372:2522-2532.
6. Larkin J, Chiarion-Sileni V, Gonazalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373:23-34.
7. Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377:1345-1356.
8. Long GV, Stroyakovskiy D, Gogas H, et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med. 2014;371:1877-1888.
9. Elwood JM, Jopson J. Melanoma and sun exposure: an overview of published studies. Int J Cancer. 1997;73:198-203.
10. Gilchrest BA, Eller MS, Geller AC, Yaar M. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 199;340:1341-1348.
11. Omholt K, Platz A, Kanter L, et al. NRAS and BRAF mutations arise early during melanoma pathogenesis and are preserved throughout tumor progression. Clin Cancer Res. 2003;9:6483-8.
12. Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-54.
13. Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 2015;161:1681-96.
14. Gershenwald JE, Scolyer RA, Hess KR, et al. Melanoma staging: evidence-based changes in the American Joint Committee on Cancer Eighth Edition Cancer Staging Manual. CA Cancer J Clin. 2017;67:472-492.
15. Robert C, Ribas A, Hamid O, et al. Durable complete response after discontinuation of pembrolizumab in patients with metastatic melanoma. J Clin Oncol. 2018;36:1668-1674.
16. Salama AKS, Hodi FS. Cytotoxic T-lymphocyte-associated antigen-4. Clin Cancer Res. 2011;17:4622-8.
17. Boussiotis VA. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med. 2016;375:1767-1778.
18. Robert C, Thomas L, Bondarenko I, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364:2517-2526.
19. Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454.
20. Topalian S, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020-30.
21. Weber JS, D’Angelo SP, Minor D, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16:375-84.
22. Ribas A, Hamid O, Daud A, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600-1609.
23. Ribas A, Puzanov I, Dummer R, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16:908-18.
24. Hamid O, Puzanov I, Dummer R, et al. Final analysis of a randomised trial comparing pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory advanced melanoma. Eur J Cancer. 2017;86:37-45.
25. Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
26. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicenter, randomised, open-label phase 3 study (KEYNOTE-006). Lancet Oncol. 2017;390:1853-1862.
27. Carlino MS, Long GV, Schadendorf D, et al. Outcomes by line of therapy and programmed death ligand 1 expression in patients with advanced melanoma treated with pembrolizumab or ipilimumab in KEYNOTE-006. A randomised clinical trial. Eur J Cancer. 2018;101:236-243.
28. Hodi FS, Chesney J, Pavlick AC, et al. Combined nivolumab and ipilimumab versus ipilimumab alone in patients with advanced melanoma: 2-year overall survival outcomes in a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol. 2016;17:1558-1568.
29. Long GV, Atkinson V, Cebon JS, et al. Standard-dose pembrolizumab in combination with reduced-dose ipilimumab for patients with advanced melanoma (KEYNOTE-029): an open-label, phase 1b trial. Lancet Oncol. 2017;18:1202-10.
30. Hodi FS, Chiarion-Sileni V, Gonzalez R, et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018;19:1480-1492.
31. Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the immune-related adverse effects of immune checkpoint inhibitors: a review. JAMA Oncol. 2016;2:1346-1353.
32. National Comprehensive Cancer Network. Management of immunotherapy-related toxicities (version 2.2019). www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed April 8, 2019.
33. Lebbé C, Meyer N, Mortier L, et al. Initial results from a phase IIIb/IV study evaluating two dosing regimens of nivolumab (NIVO) in combination with ipilimumab (IPI) in patients with advanced melanoma (CheckMate 511) [Abstract LBA47]. Ann Oncol. 2018;29:mdy424.057.
34. Schadendorf D, Wolchok JD, Hodi FS, et al. Efficacy and safety outcomes in patients with advanced melanoma who discontinued treatment with nivolumab and ipilimumab because of adverse events: a pooled analysis of randomized phase ii and iii trials. J Clin Oncol. 2017;35:3807-3814.
35. Flaherty KT, Puzanov I, Kim KB, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809-819.
36. Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
37. McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAFV600E and BRAFV600K mutation-positive melanoma (BRIM-3): extended follow up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15:323-332.
38. Hauschild A, Grob JJ, Demidov LV, et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicenter, open-label, phase 3 randomised controlled trial. Lancet Oncol. 2012;380:358-365.
39. Rizos H, Menzies AM, Pupo GM, et al. BRAF inhibitor resistance mechanisms in metastatic melanoma: spectrum and clinical impact. Clin Cancer Res. 2014;20:1965-1977.
40. Flaherty KT, Robert C, Hersey P, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367:107-114.
41. Long GV, Stroyakovskiy D, Gogas H, et al. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: a multicenter, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2015;386:444-451.
42. Long GV, Flaherty KT, Stroyakovskiy D, et al. Dabrafenib plus trametinib versus dabrafenib monotherapy in patients with metastatic BRAF V600E/K-mutant melanoma: long-term survival and safety analysis of a phase 3 study. Ann Oncol. 2017;28:1631-1639.
43. Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30-39.
44. Ascierto PA, McArthur GA, Dréno B, et al. Cobimetinib combined with vemurafenib in advanced BRAFV600-mutant melanoma (coBRIM): updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016;17:1248-260.
45. Dummer R, Ascierto PA, Gogas HJ, et al. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): a multicenter, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:603-615.
46. Dummer R, Ascierto PA, Gogas HJ, et al. Overall survival in patients with BRAF-mutant melanoma receiving encorafenib plus binimetinib versus vemurafenib or encorafenib (COLUMBUS): a multicenter, open-label, randomised, phase 3 trial. Lancet Oncol. 2018;19:1315-1327.
47. Carlos G, Anforth R, Clements A, et al. Cutaneous toxic effects of BRAF inhibitors alone and in combination with MEK inhibitors for metastatic melanoma. JAMA. Dermatol 2015;151:1103-1109.
48. Su F, Viros A, Milagre C, et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N Engl J Med. 2012;366:207-215.
49. Dummer R, Schadendorf D, Ascierto P, et al. Binimetinib versus dacarbazine in patients with advanced NRAS-mutant melanoma (NEMO): a multicenter, open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18:435-445.
50. Guo J, Si L, Kong Y, et al. Phase II, open-label, single-arm trial of imatinib mesylate in patients with metastatic melanoma harboring c-Kit mutation or amplification. J Clin Oncol. 2011;29:2904-2909.
Malignant melanoma is the most serious form of primary skin cancer and one of the only malignancies in which the incidence rate has been rising. It is estimated that in 2018 there were 91,270 newly diagnosed cases and 9320 deaths from advanced melanoma in the United States. Melanoma is the fifth most common cancer type in males and the sixth most common in females. Despite rising incidence rates, improvement in the treatment of advanced melanoma has resulted in declining death rates over the past decade.1 Although most melanoma is diagnosed at an early stage and can be cured with surgical excision, the prognosis for metastatic melanoma had been historically poor prior to recent advancements in treatment. Conventional chemotherapy treatment with dacarbazine or temozolomide resulted in response rates ranging from 7.5% to 12.1%, but without much impact on median overall survival (OS), with reported OS ranging from 6.4 to 7.8 months. Combination approaches with interferon alfa-2B and low-dose interleukin-2 resulted in improved response rates compared with traditional chemotherapy, but again without survival benefit.2
Immunotherapy in the form of high-dose interleukin-2 emerged as the first therapy to alter the natural history of advanced melanoma, with both improved response rates (objective response rate [ORR], 16%) and median OS (2 months), with some patients achieving durable responses lasting more than 30 months. However, significant systemic toxicity limited its application to carefully selected patients.3 The past decade has brought rapid advancements in treatment with immune checkpoint inhibitors and molecularly targeted agents, which have significantly improved ORRs, progression-free survival (PFS), and OS for patients with metastatic melanoma.4-8
This review is the first of 2 articles focusing on the treatment and sequencing of therapies in advanced melanoma. Here, we review the selection of first-line therapy for metastatic melanoma. Current evidence for immune checkpoint blockade and molecularly targeted agents in the treatment of metastatic melanoma after progression on first-line therapy is discussed in a separate article.
Pathogenesis
The incidence of melanoma is strongly associated with ultraviolet light–mediated DNA damage related to sun exposure. Specifically, melanoma is associated to a greater degree with intense intermittent sun exposure and sunburn, but not associated with higher occupational exposure.9 Ultraviolet radiation can induce DNA damage by a number of mechanisms, and deficient DNA repair leads to somatic mutations that drive the progression from normal melanocyte to melanoma.10
The most commonly identified genetic mutations in cutaneous melanomas are alterations in the mitogen-activated protein kinase (MAPK) pathway. Typically, an extracellular growth factor causes dimerization of the growth factor receptor, which activates the intracellular RAS GTPase protein. Subsequently BRAF is phosphorylated within the kinase domain, which leads to downstream activation of the MEK and ERK kinases through phosphorylation. Activated ERK leads to phosphorylation of various cytoplasmic and nuclear targets, and the downstream effects of these changes promote cellular proliferation. While activation of this pathway usually requires phosphorylation of BRAF by RAS, mutations placing an acidic amino acid near the kinase domain mimics phosphorylation and leads to constitutive activation of the BRAF serine/threonine kinase in the absence of upstream signaling from extracellular growth factors mediated through RAS.11 One study of tumor samples of 71 patients with cutaneous melanoma detected NRAS mutations in 30% and BRAF mutations in 59% of all tumors tested. Of the BRAF mutation–positive tumors, 88% harbored the Val599Glu mutation, now commonly referred to as the BRAF V600E mutation. The same study demonstrated that the vast majority of BRAF mutations were seen in the primary tumor and were preserved when metastases were analyzed. Additionally, both NRAS and BRAF mutations were detected in the radial growth phase of the melanoma tumor. These findings indicate that alterations in the MAPK pathway occur early in the pathogenesis of advanced melanoma.11 Another group demonstrated that 66% of malignant melanoma tumor samples harbored BRAF mutations, of which 80% were specifically the V600E mutation. In vitro assays showed that the BRAF V600E–mutated kinase had greater than 10-fold kinase activity compared to wild-type BRAF, and that this kinase enhanced cellular proliferation even when upstream NRAS signaling was inhibited.12
The Cancer Genome Atlas Network performed a large analysis of tumor samples from 331 different melanoma patients and studied variations at the DNA, RNA, and protein levels. The study established a framework of 4 notable genomic subtypes, including mutant BRAF (52%), mutant RAS (28%), mutant NF1 (14%), and triple wild-type (6%). Additionally, mRNA transcriptomic analysis of overexpressed genes identified 3 different subclasses, which were labeled as “immune,” “keratin,” and “MITF-low.” The immune subclass was characterized by increased expression of proteins found in immune cells, immune signaling molecules, immune checkpoint proteins, cytokines, and chemokines, and correlated with increased lymphocyte invasion within the tumor. Interestingly, in the post-accession survival analysis, the “immune” transcriptomic subclass was statistically correlated with an improved prognosis.13 Having an understanding of the molecular pathogenesis of advanced melanoma helps to create a framework for understanding the mechanisms of current standard of care therapies for the disease.
Case Presentation
A 62-year-old Caucasian man with a history of well-controlled type 2 diabetes mellitus and hypertension is being followed by his dermatologist for surveillance of melanocytic nevi. On follow-up he is noted to have an asymmetrical melanocytic lesion over the right scalp with irregular borders and variegated color. He is asymptomatic and the remainder of physical examination is unremarkable, as he has no other concerning skin lesions and no cervical, axillary, or inguinal lymphadenopathy.
How is melanoma diagnosed?
Detailed discussion about diagnosis and staging will be deferred in this review of treatment of advanced melanoma. In brief, melanoma is best diagnosed by excisional biopsy and histopathology. Staging of melanoma is done according to the American Joint Committee on Cancer’s (AJCC) Cancer Staging Manual, 8th edition, using a TNM staging system that incorporates tumor thickness (Breslow depth); ulceration; number of involved regional lymph nodes; presence of in-transit, satellite, and/or microsatellite metastases; distant metastases; and serum lactate dehydrogenase level.14
Case Continued
The patient undergoes a wide excisional biopsy of the right scalp lesion, which is consistent with malignant melanoma. Pathology demonstrates a Breslow depth of 2.6 mm, 2 mitotic figures/mm2, and no evidence of ulceration. He subsequently undergoes wide local excision with 0/3 sentinel lymph nodes positive for malignancy. His final staging is consistent with pT3aN0M0, stage IIA melanoma.
He is seen in follow-up with medical oncology for the next 3.5 years without any evidence of disease recurrence. He then develops symptoms of vertigo, diplopia, and recurrent falls, prompting medical attention. Magnetic resonance imaging (MRI) brain reveals multiple supratentorial and infratentorial lesions concerning for intracranial metastases. Further imaging with computed tomography (CT) chest/abdomen/pelvis reveals a right lower lobe pulmonary mass with right hilar and subcarinal lymphadenopathy. He is admitted for treatment with intravenous dexamethasone and further evaluation with endobronchial ultrasound-guided fine-needle aspiration of the right lower lobe mass, which reveals metastatic melanoma. Given the extent of his intracranial metastases, he is treated with whole brain radiation therapy for symptomatic relief prior to initiating systemic therapy.
What is the general approach to first-line treatment for metastatic melanoma?
The past decade has brought an abundance of data supporting the use of immunotherapy with immune checkpoint inhibitors or molecularly targeted therapy with combined BRAF/MEK inhibitors in the first-line setting.4-8 After the diagnosis of metastatic melanoma has been made, molecular testing is recommended to determine the BRAF status of the tumor. Immunotherapy is the clear choice for first-line therapy in the absence of an activating BRAF V600 mutation. When a BRAF V600 mutation is present, current evidence supports the use of either immunotherapy or molecularly targeted therapy as first-line therapy.
To date, there have been no prospective clinical trials comparing the sequencing of immunotherapy and molecularly targeted therapy in the first-line setting. An ongoing clinical trial (NCT02224781) is comparing dabrafenib and trametinib followed by ipilimumab and nivolumab at time of progression to ipilimumab and nivolumab followed by dabrafenib and trametinib in patients with newly diagnosed stage III/IV BRAF V600 mutation–positive melanoma. The primary outcome measure is 2-year OS. Until completion of that trial, current practice regarding which type of therapy to use in the first-line setting is based on a number of factors including clinical characteristics and provider preferences.
Data suggest that immunotherapies can produce durable responses, especially after treatment completion or discontinuation, albeit at the expense of taking a longer time to achieve clinical benefit and the risk of potentially serious immune-related adverse effects. This idea of a durable, off-treatment response is highlighted by a study that followed 105 patients who had achieved a complete response (CR) and found that 24-month disease-free survival from the time of CR was 90.9% in all patients and 89.9% in the 67 patients who had discontinued pembrolizumab after attaining CR.15 BRAF/MEK inhibition has the potential for rapid clinical responses, though concerns exist about the development of resistance to therapy. The following sections explore the evidence supporting the use of these therapies.
Immunotherapy with Immune Checkpoint Inhibitors
Immunotherapy via immune checkpoint blockade has revolutionized the treatment of many solid tumors over the past decade. The promise of immunotherapy revolves around the potential for achieving a dynamic and durable systemic response against cancer by augmenting the antitumor effects of the immune system. T-cells are central to mounting a systemic antitumor response, and, in addition to antigen recognition, their function depends heavily on fine tuning between co-stimulatory and co-inhibitory signaling. The cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) expressed on T-cells was the first discovered co-inhibitory receptor of T-cell activation.16 Later, it was discovered that the programmed cell death 1 receptor (PD-1), expressed on T-cells, and its ligands PD-L1 and PD-L2, expressed on antigen presenting cells, tumor cells, or other cells in the tumor microenvironment, also served as a potent negative regulator of T-cell function.17
Together, these 2 signaling pathways help to maintain peripheral immune tolerance, whereby autoreactive T-cells that have escaped from the thymus are silenced to prevent autoimmunity. However, these pathways can also be utilized by cancer cells to escape immune surveillance. Monoclonal antibodies that inhibit the aforementioned co-inhibitory signaling pathways, and thus augment the immune response, have proven to be an effective anticancer therapy capable of producing profound and durable responses in certain malignancies.16,17
Ipilimumab
Ipilimumab is a monoclonal antibody that inhibits the function of the CTLA-4 co-inhibitory immune checkpoint. In a phase 3 randomized controlled trial of 676 patients with previously treated metastatic melanoma, ipilimumab at a dose of 3 mg/kg every 3 weeks for 4 cycles, with or without a gp100 peptide vaccine, resulted in an improved median OS of 10.0 and 10.1 months, respectively, compared to 6.4 months in those receiving the peptide vaccine alone, meeting the primary endpoint.4 Subsequently, a phase 3 trial of 502 patients with untreated metastatic melanoma compared ipilimumab at a dose of 10 mg/kg every 3 weeks for 4 cycles plus dacarbazine to dacarbazine plus placebo and found a significant increase in median OS (11.2 months vs 9.1 months), with no additive benefit of chemotherapy. There was a higher reported rate of grade 3 or 4 adverse events in this trial with ipilimumab dosed at 10 mg/kg, which was felt to be dose-related.18 These trials were the first to show improved OS with any systemic therapy in metastatic melanoma and led to US Food and Drug Administration approval of ipilimumab for this indication in 2011.
PD-1 Inhibitor Monotherapy
The PD-1 inhibitors nivolumab and pembrolizumab were initially approved for metastatic melanoma after progression on ipilimumab. In the phase 1 trial of patients with previously treated metastatic melanoma, nivolumab therapy resulted in an ORR of 28%.19 The subsequent phase 2 trial conducted in pretreated patients, including patients who had progressed on ipilimumab, confirmed a similar ORR of 31%, as well as a median PFS of 3.7 months and a median OS of 16.8 months. The estimated response duration in patients who did achieve a response to therapy was 2 years.20 A phase 3 trial (CheckMate 037) comparing nivolumab (n = 120) to investigator’s choice chemotherapy (n = 47) in those with melanoma refractory to ipilimumab demonstrated that nivolumab was superior for the primary endpoint of ORR (31.7% vs 10.6%), had less toxicity (5% rate of grade 3 or 4 adverse events versus 9%), and increased median duration of response (32 months vs 13 months).21
The phase 1 trial (KEYNOTE-001) testing the efficacy of pembrolizumab demonstrated an ORR of 33% in the total population of patients treated and an ORR of 45% in those who were treatment-naive. Additionally, the median OS was 23 months for the total population and 31 months for treatment-naive patients, with only 14% of patients experiencing a grade 3 or 4 adverse event.22 The KEYNOTE-002 phase 2 trial compared 2 different pembrolizumab doses (2 mg/kg and 10 mg/kg every 3 weeks) to investigator’s choice chemotherapy (paclitaxel plus carboplatin, paclitaxel, carboplatin, dacarbazine, or oral temozolomide) in 540 patients with advanced melanoma with documented progression on ipilimumab with or without prior progression on molecularly targeted therapy if positive for a BRAF V600 mutation. The final analysis demonstrated significantly improved ORR with pembrolizumab (22% at 2 mg/kg vs 26% at 10 mg/kg vs 4% chemotherapy) and significantly improved 24-month PFS (16% vs 22% vs 0.6%, respectively). There was a nonstatistically significant improvement in median OS (13.4 months vs 14.7 months vs 10 months), although 55% of the patients initially assigned to the chemotherapy arm crossed over and received pembrolizumab after documentation of progressive disease.23,24
Because PD-1 inhibition improved efficacy with less toxicity than chemotherapy when studied in progressive disease, subsequent studies focused on PD-1 inhibition in the frontline setting. CheckMate 066 was a phase 3 trial comparing nivolumab to dacarbazine as first-line therapy for 418 patients with untreated metastatic melanoma who did not have a BRAF mutation. For the primary end point of 1-year OS, nivolumab was superior to dacarbazine (72.9% vs 42.1%; hazard ratio [HR], 0.42; P < 0.001). Treatment with nivolumab also resulted in superior ORR (40% vs 14%) and PFS (5.1 months vs 2.2 months). Additionally, nivolumab therapy had a lower rate of grade 3 or 4 toxicity compared to dacarbazine (11.7% vs 17.6%).25
The KEYNOTE-006 trial compared 2 separate dosing schedules of pembrolizumab (10 mg/kg every 2 weeks versus every 3 weeks) to ipilimumab (3 mg/kg every 3 weeks for 4 cycles) in a 1:1:1 ratio in 834 patients with metastatic melanoma who had received up to 1 prior systemic therapy, but no prior CTLA-4 or PD-1 inhibitors. The first published data reported statistically significant outcomes for the co-primary end points of 6-month PFS (47.3% for pembrolizumab every 2 weeks vs 46.4% for pembrolizumab every 3 weeks vs 26.5% for ipilimumab; HR, 0.58 for both pembrolizumab groups compared to ipilimumab; P < 0.001) and 12-month OS (74.1% vs 68.4% vs 58.2%) with pembrolizumab compared to ipilimumab. Compared to ipilimumab, pembrolizumab every 2 weeks had a hazard ratio of 0.63 (P = 0.0005) and pembrolizumab every 3 weeks had a hazard ratio of 0.69 (P = 0.0036). The pembrolizumab groups was also had lower rates of grade 3 to 5 toxicity (13.3% vs 10.1% vs 19.9%).5 Updated outcomes demonstrated improved ORR compared to the first analysis (37% vs 36% vs 13%), and improved OS (median OS, not reached for the pembrolizumab groups vs 16.0 months for the ipilimumab group; HR, 0.68, P = 0.0009 for pembrolizumab every 2 weeks versus HR 0.68, P = 0.0008 for pembrolizumab every 3 weeks).26 In addition, 24-month OS was 55% in both pembrolizumab groups compared to 43% in the ipilimumab group. Grade 3 or 4 toxicity occurred less frequently with pembrolizumab (17% vs 17% vs 20%).
Further analysis from the KEYNOTE-006 trial data demonstrated improved ORR, PFS, and OS with pembrolizumab compared to ipilimumab in tumors positive for PD-L1 expression. For PD-L1-negative tumors, response rate was higher, and PFS and OS rates were similar with pembrolizumab compared to ipilimumab. Given that pembrolizumab was associated with similar survival outcomes in PD-L1-negative tumors and with less toxicity than ipilimumab, the superiority of PD-L1 inhibitors over ipilimumab was further supported, regardless of tumor PD-L1 status.27
In sum, PD-1 inhibition should be considered the first-line immunotherapy in advanced melanoma, either alone or in combination with ipilimumab, as discussed in the following section. There is no longer a role for ipilimumab monotherapy in the first-line setting, based on evidence from direct comparison to single-agent PD-1 inhibition in clinical trials that demonstrated superior efficacy and less serious toxicity with PD-1 inhibitors.5,26 The finding that ORR and OS outcomes with single-agent PD-1 inhibitors are higher in treatment-naive patients compared to those receiving prior therapies also supports this approach.22
Combination CTLA-4 and PD-1 Therapy
Despite the potential for durable responses, the majority of patients fail to respond to single-agent PD-1 therapy. Given that preclinical data had suggested the potential for synergy between dual inhibition of CTLA-4 and PD-1, clinical trials were designed to test this approach. The first randomized phase 2 trial that established superior efficacy with combination therapy was the CheckMate 069 trial comparing nivolumab plus ipilimumab to ipilimumab monotherapy. Combination therapy resulted in increased ORR (59% vs 11%), median PFS (not reached vs 3.0 months), 2-year PFS (51.3% vs 12.0%), and 2-year OS (63.8% vs 53.6%).28 Similarly, a phase 1b trial of pembrolizumab plus reduced-dose ipilimumab demonstrated an ORR of 61%, with a 1-year PFS of 69% and 1-year OS of 89%.29
The landmark phase 3 CheckMate 067 trial analyzed efficacy outcomes for 3 different treatment regimens including nivolumab plus ipilimumab, nivolumab monotherapy, and ipilimumab monotherapy in previously untreated patients with unresectable stage III or IV melanoma. The trial was powered to compare survival outcomes for both the combination therapy arm against ipilimumab and the nivolumab monotherapy arm against ipilimumab, but not to compare combination therapy to nivolumab monotherapy. The initial analysis demonstrated a median PFS of 11.5 months with combination therapy versus 6.9 months with nivolumab and 2.9 months with ipilimumab, as well as an ORR of 58% versus 44% and 19%, respectively (Table 1).6 The updated 3-year survival outcomes from CheckMate 067 were notable for superior median OS with combination therapy (not reached in combination vs 37.6 months for nivolumab vs 19.9 months ipilimumab), improved 3-year OS (58% vs 52% vs 34%), and improved 3-year PFS (39% vs 32% vs 10%).7 In the reported 4-year survival outcomes, median OS was not reached in the combination therapy group, and was 36.9 months in the nivolumab monotherapy group and 19.9 months in the ipilimumab monotherapy group. Rates of grade 3 or 4 adverse events were significantly higher in the combination therapy group, at 59% compared to 22% with nivolumab monotherapy and 28% with ipilimumab alone.30 The 3- and 4-year OS outcomes (58% and 54%, respectively) with combination therapy were the highest seen in any phase 3 trial for treatment of advanced melanoma, supporting its use as the best approved first-line therapy in those who can tolerate the potential toxicity of combination therapy7,30 The conclusions from this landmark trial were that both combination therapy and nivolumab monotherapy resulted in statistically significant improvement in OS compared to ipilimumab.
Toxicity Associated with Immune Checkpoint Inhibitors
While immune checkpoint inhibitors have revolutionized the treatment of many solid tumor malignancies, this new class of cancer therapy has brought about a new type of toxicity for clinicians to be aware of, termed immune-related adverse events (irAEs). As immune checkpoint inhibitors amplify the immune response against malignancy, they also increase the likelihood that autoreactive T-cells persist and proliferate within the circulation. Therefore, these therapies can result in almost any type of autoimmune side effect. The most commonly reported irAEs in large clinical trials studying CTLA-4 and PD-1 inhibitors include rash/pruritus, diarrhea/colitis, hepatitis, endocrinopathies (thyroiditis, hypophysitis, adrenalitis), and pneumonitis. Other more rare toxicities include pancreatitis, autoimmune hematologic toxicities, cardiac toxicity (myocarditis, heart failure), and neurologic toxicities (neuropathies, myasthenia gravis-like syndrome, Guillain-Barré syndrome). It has been observed that PD-1 inhibitors have a lower incidence of irAEs than CTLA-4 inhibitors, and that the combined use of PD-1 and CTLA-4 inhibitors is associated with a greater incidence of irAEs compared to monotherapy with either agent.31 Toxicities associated with ipilimumab have been noted to be dose dependent.18 Generally, these toxicities are treated with immunosuppression in the form of glucocorticoids and are often reversible.31 There are several published guidelines that include algorithms for the management of irAEs by organizations such as the National Comprehensive Cancer Network.32
For example, previously untreated patients treated with ipilimumab plus dacarbazine as compared to dacarbazine plus placebo had greater grade 3 or 4 adverse events (56.3% vs 27.5%), and 77.7% of patients experiencing an irAE of any grade.18 In the CheckMate 066 trial comparing frontline nivolumab to dacarbazine, nivolumab had a lower rate of grade 3 or 4 toxicity (11.7% vs 17.6%) and irAEs were relatively infrequent, with diarrhea and elevated alanine aminotransferase level each being the most prominent irAE (affecting 1.0% of patients).25 In the KEYNOTE-006 trial, irAEs seen in more than 1% of patients treated with pembrolizumab included colitis, hepatitis, hypothyroidism, and hyperthyroidism, whereas those occurring in more than 1% of patients treated with ipilimumab included colitis and hypophysitis. Overall, there were lower rates of grade 3 to 5 toxicity with the 2 pembrolizumab doses compared to ipilimumab (13.3% pembrolizumab every 2 weeks vs 10.1% pembrolizumab every 3 weeks vs 19.9% ipilimumab).5 In the CheckMate 067 trial comparing nivolumab plus ipilimumab, nivolumab monotherapy, and ipilimumab monotherapy, rates of treatment-related adverse events of any grade were higher in the combination group (96% combination vs 86% nivolumab vs 86% ipilimumab), as were rates of grade 3 or 4 adverse events (59% vs 21% vs 28%, respectively). The irAE profile was similar to that demonstrated in prior studies: rash/pruritus were the most common, and diarrhea/colitis, elevated aminotransferases, and endocrinopathies were among the more common irAEs.7
Alternative dosing strategies have been investigated in an effort to preserve efficacy and minimize toxicity. A phase 1b trial of pembrolizumab plus reduced-dose ipilimumab demonstrated an ORR of 61%, with a 1-year PFS of 69% and a 1-year OS of 89%. This combination led to 45% of patients having a grade 3 or 4 adverse event, 60% having irAEs of any grade, and only 27% having grade 3 or 4 irAEs.29 The CheckMate 067 trial studied the combination of nivolumab 1 mg/kg plus ipilimumab 3 mg/kg.6 The CheckMate 511 trial compared different combination dosing strategies (nivolumab 3 mg/kg + ipilimumab 1 mg/kg versus nivolumab 1 mg/kg + ipilimumab 3 mg/kg) to assess for safety benefit. In the results published in abstract form, the reduced ipilimumab dose (nivolumab 3 mg/kg + ipilimumab 1 mg/kg arm) resulted in significantly decreased grade 3 to 5 adverse events (33.9% vs 48.3%) without significant differences in ORR, PFS, or OS.33
The question about the efficacy of checkpoint inhibitors in patients who discontinue treatment due to irAEs has been raised, as one hypothesis suggests that such toxicities may also indicate that the antitumor immune response has been activated. In a retrospective pooled analysis of phase 2 and 3 trials where patients received combination therapy with ipilimumab and nivolumab and discontinued therapy during the induction phase due to irAEs, outcomes did not appear to be inferior. Median PFS was 8.4 months in those who discontinued therapy compared to 10.8 months in those who continued therapy, but this did not reach statistical significance. Median OS had not been reached in either group and ORR was actually higher in those who discontinued due to adverse events (58.3% vs 50.2%). While this retrospective analysis needs to be validated, it does suggest that patients likely derive antitumor benefit from immunotherapy even if they have to discontinue therapy due to irAEs. Of note, patients in this analysis were not trialed on nivolumab monotherapy after receiving immunosuppressive treatment for toxicity related to combination therapy, which has since been deemed a reasonable treatment option.34
Molecularly Targeted Therapy for Metastatic Melanoma
As previously mentioned, the MAPK pathway is frequently altered in metastatic melanoma and thus serves as a target for therapy. Mutations in BRAF can cause constitutive activation of the protein’s kinase function, which subsequently phosphorylates/activates MEK in the absence of extracellular growth signals and causes increased cellular proliferation. For the roughly half of patients diagnosed with metastatic melanoma who harbor a BRAF V600 mutation, molecularly targeted therapy with BRAF/MEK inhibitors has emerged as a standard of care treatment option. As such, all patients with advanced disease should be tested for BRAF mutations.
After early phase 1 studies of the BRAF inhibitor vemurafenib demonstrated successful inhibition of mutated BRAF,35 subsequent studies confirmed the benefit of BRAF targeted therapy. In the phase 3 randomized controlled BRIM-3 trial comparing vemurafenib with dacarbazine for treatment of 675 patients with previously untreated metastatic melanoma positive for a BRAF V600E mutation, the vemurafenib group had superior ORR and 6-month OS during the first analysis.36 In a subsequent analysis, median PFS and median OS were also superior with vemurafenib compared to dacarbazine, as vemurafenib had a median OS of 13.6 months compared to 9.7 months with dacarbazine (HR, 0.70; P = 0.0008).37 Dabrafenib was the next BRAF inhibitor to demonstrate clinical efficacy with superior PFS compared to dacarbazine.38
Despite tumor shrinkage in the majority of patients, the development of resistance to therapy was an issue early on. The development of acquired resistance emerged as a heterogeneous process, though many of the identified resistance mechanisms involved reactivation of the MAPK pathway.39 A phase 3 trial of 322 patients with metastatic melanoma comparing the MEK inhibitor trametinib as monotherapy against chemotherapy demonstrated a modest improvement in both median PFS and OS.40 As a result, subsequent efforts focused on a strategy of concurrent MEK inhibition as a means to overcome resistance to molecularly targeted monotherapy
At least 4 large phase 3 randomized controlled trials of combination therapy with BRAF plus MEK inhibitors showed an improved ORR, PFS, and OS when compared to BRAF inhibition alone. The COMBI-d trial comparing dabrafenib plus trametinib versus dabrafenib alone was the first to demonstrate the superiority of combined BRAF/MEK inhibition and made combination therapy the current standard of care for patients with metastatic melanoma and a BRAF V600 mutation. In the final analysis of this trial, 3-year PFS was 22% with combination therapy compared to 12% with dabrafenib alone, and 3-year OS was 44% compared to 32%.8,41,42 A second trial with the combination of dabrafenib and trametinib (COMBI-V) also demonstrated superior efficacy when compared to single-agent vemurafenib without increased toxicity.43 Subsequently, the combination of vemurafenib with MEK inhibitor cobimetinib demonstrated superiority compared to vemurafenib alone,44 followed by the newest combination encorafenib (BRAF inhibitor) and binimetinib (MEK inhibitor) proving superior to either vemurafenib or encorafenib alone.45,46
It is important to note that there have been no studies directly comparing the efficacy of the 3 approved BRAF/MEK inhibitor combinations, but the 3 different regimens have some differences in their toxicity profiles (Table 2). Of note, single-agent BRAF inhibition was associated with increased cutaneous toxicity, including secondary squamous cell carcinoma and keratoacanthoma,47 which was demonstrated to be driven by paradoxical activation of the MAPK pathway.48 The concerning cutaneous toxicities such as squamous cell carcinoma were substantially reduced by combination BRAF/MEK inhibitor therapy.47 Collectively, the higher efficacy along with manageable toxicity profile established combination BRAF/MEK inhibition as the preferred regimen for patients with BRAF-mutated metastatic melanoma who are being considered for molecularly targeted therapy. BRAF inhibitor monotherapy should only be used when there is a specific concern regarding the use of a MEK inhibitor in certain clinical circumstances.
Other driver mutations associated with metastatic melanoma such as NRAS-mutated tumors have proven more difficult to effectively treat with molecularly targeted therapy, with one study showing that the MEK inhibitor binimetinib resulted in a modest improvement in ORR and median PFS without OS benefit compared to dacarbazine.49 Several phase 2 trials involving metastatic melanoma harboring a c-Kit alteration have demonstrated some efficacy with the tyrosine kinase inhibitor imatinib. The largest phase 2 trial of 43 patients treated with imatinib resulted in a 53.5% disease control rate (23.3% partial response and 30.2% stable disease), with 9 of the 10 patients who achieved partial response having a mutation in either exon 11 or 13. Median PFS was 3.5 months and 1-year OS was 51.0%.50
Case Conclusion
Prior to initiation of systemic therapy, the patient’s melanoma is tested and is found to be positive for a BRAF V600K mutation. At his follow-up appointment, the patient continues to endorse generalized weakness, fatigue, issues with balance, and residual pulmonary symptoms after being treated for post-obstructive pneumonia. Given his current symptoms and extent of metastatic disease, immunotherapy is deferred and he is started on combination molecularly targeted therapy with dabrafenib and trametinib. He initially does well, with a partial response noted by resolution of symptoms and decreased size of his intracranial metastases and decreased size of the right lower lobe mass. Further follow-up of this patient is presented in the second article in this 2-part review of advanced melanoma.
Malignant melanoma is the most serious form of primary skin cancer and one of the only malignancies in which the incidence rate has been rising. It is estimated that in 2018 there were 91,270 newly diagnosed cases and 9320 deaths from advanced melanoma in the United States. Melanoma is the fifth most common cancer type in males and the sixth most common in females. Despite rising incidence rates, improvement in the treatment of advanced melanoma has resulted in declining death rates over the past decade.1 Although most melanoma is diagnosed at an early stage and can be cured with surgical excision, the prognosis for metastatic melanoma had been historically poor prior to recent advancements in treatment. Conventional chemotherapy treatment with dacarbazine or temozolomide resulted in response rates ranging from 7.5% to 12.1%, but without much impact on median overall survival (OS), with reported OS ranging from 6.4 to 7.8 months. Combination approaches with interferon alfa-2B and low-dose interleukin-2 resulted in improved response rates compared with traditional chemotherapy, but again without survival benefit.2
Immunotherapy in the form of high-dose interleukin-2 emerged as the first therapy to alter the natural history of advanced melanoma, with both improved response rates (objective response rate [ORR], 16%) and median OS (2 months), with some patients achieving durable responses lasting more than 30 months. However, significant systemic toxicity limited its application to carefully selected patients.3 The past decade has brought rapid advancements in treatment with immune checkpoint inhibitors and molecularly targeted agents, which have significantly improved ORRs, progression-free survival (PFS), and OS for patients with metastatic melanoma.4-8
This review is the first of 2 articles focusing on the treatment and sequencing of therapies in advanced melanoma. Here, we review the selection of first-line therapy for metastatic melanoma. Current evidence for immune checkpoint blockade and molecularly targeted agents in the treatment of metastatic melanoma after progression on first-line therapy is discussed in a separate article.
Pathogenesis
The incidence of melanoma is strongly associated with ultraviolet light–mediated DNA damage related to sun exposure. Specifically, melanoma is associated to a greater degree with intense intermittent sun exposure and sunburn, but not associated with higher occupational exposure.9 Ultraviolet radiation can induce DNA damage by a number of mechanisms, and deficient DNA repair leads to somatic mutations that drive the progression from normal melanocyte to melanoma.10
The most commonly identified genetic mutations in cutaneous melanomas are alterations in the mitogen-activated protein kinase (MAPK) pathway. Typically, an extracellular growth factor causes dimerization of the growth factor receptor, which activates the intracellular RAS GTPase protein. Subsequently BRAF is phosphorylated within the kinase domain, which leads to downstream activation of the MEK and ERK kinases through phosphorylation. Activated ERK leads to phosphorylation of various cytoplasmic and nuclear targets, and the downstream effects of these changes promote cellular proliferation. While activation of this pathway usually requires phosphorylation of BRAF by RAS, mutations placing an acidic amino acid near the kinase domain mimics phosphorylation and leads to constitutive activation of the BRAF serine/threonine kinase in the absence of upstream signaling from extracellular growth factors mediated through RAS.11 One study of tumor samples of 71 patients with cutaneous melanoma detected NRAS mutations in 30% and BRAF mutations in 59% of all tumors tested. Of the BRAF mutation–positive tumors, 88% harbored the Val599Glu mutation, now commonly referred to as the BRAF V600E mutation. The same study demonstrated that the vast majority of BRAF mutations were seen in the primary tumor and were preserved when metastases were analyzed. Additionally, both NRAS and BRAF mutations were detected in the radial growth phase of the melanoma tumor. These findings indicate that alterations in the MAPK pathway occur early in the pathogenesis of advanced melanoma.11 Another group demonstrated that 66% of malignant melanoma tumor samples harbored BRAF mutations, of which 80% were specifically the V600E mutation. In vitro assays showed that the BRAF V600E–mutated kinase had greater than 10-fold kinase activity compared to wild-type BRAF, and that this kinase enhanced cellular proliferation even when upstream NRAS signaling was inhibited.12
The Cancer Genome Atlas Network performed a large analysis of tumor samples from 331 different melanoma patients and studied variations at the DNA, RNA, and protein levels. The study established a framework of 4 notable genomic subtypes, including mutant BRAF (52%), mutant RAS (28%), mutant NF1 (14%), and triple wild-type (6%). Additionally, mRNA transcriptomic analysis of overexpressed genes identified 3 different subclasses, which were labeled as “immune,” “keratin,” and “MITF-low.” The immune subclass was characterized by increased expression of proteins found in immune cells, immune signaling molecules, immune checkpoint proteins, cytokines, and chemokines, and correlated with increased lymphocyte invasion within the tumor. Interestingly, in the post-accession survival analysis, the “immune” transcriptomic subclass was statistically correlated with an improved prognosis.13 Having an understanding of the molecular pathogenesis of advanced melanoma helps to create a framework for understanding the mechanisms of current standard of care therapies for the disease.
Case Presentation
A 62-year-old Caucasian man with a history of well-controlled type 2 diabetes mellitus and hypertension is being followed by his dermatologist for surveillance of melanocytic nevi. On follow-up he is noted to have an asymmetrical melanocytic lesion over the right scalp with irregular borders and variegated color. He is asymptomatic and the remainder of physical examination is unremarkable, as he has no other concerning skin lesions and no cervical, axillary, or inguinal lymphadenopathy.
How is melanoma diagnosed?
Detailed discussion about diagnosis and staging will be deferred in this review of treatment of advanced melanoma. In brief, melanoma is best diagnosed by excisional biopsy and histopathology. Staging of melanoma is done according to the American Joint Committee on Cancer’s (AJCC) Cancer Staging Manual, 8th edition, using a TNM staging system that incorporates tumor thickness (Breslow depth); ulceration; number of involved regional lymph nodes; presence of in-transit, satellite, and/or microsatellite metastases; distant metastases; and serum lactate dehydrogenase level.14
Case Continued
The patient undergoes a wide excisional biopsy of the right scalp lesion, which is consistent with malignant melanoma. Pathology demonstrates a Breslow depth of 2.6 mm, 2 mitotic figures/mm2, and no evidence of ulceration. He subsequently undergoes wide local excision with 0/3 sentinel lymph nodes positive for malignancy. His final staging is consistent with pT3aN0M0, stage IIA melanoma.
He is seen in follow-up with medical oncology for the next 3.5 years without any evidence of disease recurrence. He then develops symptoms of vertigo, diplopia, and recurrent falls, prompting medical attention. Magnetic resonance imaging (MRI) brain reveals multiple supratentorial and infratentorial lesions concerning for intracranial metastases. Further imaging with computed tomography (CT) chest/abdomen/pelvis reveals a right lower lobe pulmonary mass with right hilar and subcarinal lymphadenopathy. He is admitted for treatment with intravenous dexamethasone and further evaluation with endobronchial ultrasound-guided fine-needle aspiration of the right lower lobe mass, which reveals metastatic melanoma. Given the extent of his intracranial metastases, he is treated with whole brain radiation therapy for symptomatic relief prior to initiating systemic therapy.
What is the general approach to first-line treatment for metastatic melanoma?
The past decade has brought an abundance of data supporting the use of immunotherapy with immune checkpoint inhibitors or molecularly targeted therapy with combined BRAF/MEK inhibitors in the first-line setting.4-8 After the diagnosis of metastatic melanoma has been made, molecular testing is recommended to determine the BRAF status of the tumor. Immunotherapy is the clear choice for first-line therapy in the absence of an activating BRAF V600 mutation. When a BRAF V600 mutation is present, current evidence supports the use of either immunotherapy or molecularly targeted therapy as first-line therapy.
To date, there have been no prospective clinical trials comparing the sequencing of immunotherapy and molecularly targeted therapy in the first-line setting. An ongoing clinical trial (NCT02224781) is comparing dabrafenib and trametinib followed by ipilimumab and nivolumab at time of progression to ipilimumab and nivolumab followed by dabrafenib and trametinib in patients with newly diagnosed stage III/IV BRAF V600 mutation–positive melanoma. The primary outcome measure is 2-year OS. Until completion of that trial, current practice regarding which type of therapy to use in the first-line setting is based on a number of factors including clinical characteristics and provider preferences.
Data suggest that immunotherapies can produce durable responses, especially after treatment completion or discontinuation, albeit at the expense of taking a longer time to achieve clinical benefit and the risk of potentially serious immune-related adverse effects. This idea of a durable, off-treatment response is highlighted by a study that followed 105 patients who had achieved a complete response (CR) and found that 24-month disease-free survival from the time of CR was 90.9% in all patients and 89.9% in the 67 patients who had discontinued pembrolizumab after attaining CR.15 BRAF/MEK inhibition has the potential for rapid clinical responses, though concerns exist about the development of resistance to therapy. The following sections explore the evidence supporting the use of these therapies.
Immunotherapy with Immune Checkpoint Inhibitors
Immunotherapy via immune checkpoint blockade has revolutionized the treatment of many solid tumors over the past decade. The promise of immunotherapy revolves around the potential for achieving a dynamic and durable systemic response against cancer by augmenting the antitumor effects of the immune system. T-cells are central to mounting a systemic antitumor response, and, in addition to antigen recognition, their function depends heavily on fine tuning between co-stimulatory and co-inhibitory signaling. The cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) expressed on T-cells was the first discovered co-inhibitory receptor of T-cell activation.16 Later, it was discovered that the programmed cell death 1 receptor (PD-1), expressed on T-cells, and its ligands PD-L1 and PD-L2, expressed on antigen presenting cells, tumor cells, or other cells in the tumor microenvironment, also served as a potent negative regulator of T-cell function.17
Together, these 2 signaling pathways help to maintain peripheral immune tolerance, whereby autoreactive T-cells that have escaped from the thymus are silenced to prevent autoimmunity. However, these pathways can also be utilized by cancer cells to escape immune surveillance. Monoclonal antibodies that inhibit the aforementioned co-inhibitory signaling pathways, and thus augment the immune response, have proven to be an effective anticancer therapy capable of producing profound and durable responses in certain malignancies.16,17
Ipilimumab
Ipilimumab is a monoclonal antibody that inhibits the function of the CTLA-4 co-inhibitory immune checkpoint. In a phase 3 randomized controlled trial of 676 patients with previously treated metastatic melanoma, ipilimumab at a dose of 3 mg/kg every 3 weeks for 4 cycles, with or without a gp100 peptide vaccine, resulted in an improved median OS of 10.0 and 10.1 months, respectively, compared to 6.4 months in those receiving the peptide vaccine alone, meeting the primary endpoint.4 Subsequently, a phase 3 trial of 502 patients with untreated metastatic melanoma compared ipilimumab at a dose of 10 mg/kg every 3 weeks for 4 cycles plus dacarbazine to dacarbazine plus placebo and found a significant increase in median OS (11.2 months vs 9.1 months), with no additive benefit of chemotherapy. There was a higher reported rate of grade 3 or 4 adverse events in this trial with ipilimumab dosed at 10 mg/kg, which was felt to be dose-related.18 These trials were the first to show improved OS with any systemic therapy in metastatic melanoma and led to US Food and Drug Administration approval of ipilimumab for this indication in 2011.
PD-1 Inhibitor Monotherapy
The PD-1 inhibitors nivolumab and pembrolizumab were initially approved for metastatic melanoma after progression on ipilimumab. In the phase 1 trial of patients with previously treated metastatic melanoma, nivolumab therapy resulted in an ORR of 28%.19 The subsequent phase 2 trial conducted in pretreated patients, including patients who had progressed on ipilimumab, confirmed a similar ORR of 31%, as well as a median PFS of 3.7 months and a median OS of 16.8 months. The estimated response duration in patients who did achieve a response to therapy was 2 years.20 A phase 3 trial (CheckMate 037) comparing nivolumab (n = 120) to investigator’s choice chemotherapy (n = 47) in those with melanoma refractory to ipilimumab demonstrated that nivolumab was superior for the primary endpoint of ORR (31.7% vs 10.6%), had less toxicity (5% rate of grade 3 or 4 adverse events versus 9%), and increased median duration of response (32 months vs 13 months).21
The phase 1 trial (KEYNOTE-001) testing the efficacy of pembrolizumab demonstrated an ORR of 33% in the total population of patients treated and an ORR of 45% in those who were treatment-naive. Additionally, the median OS was 23 months for the total population and 31 months for treatment-naive patients, with only 14% of patients experiencing a grade 3 or 4 adverse event.22 The KEYNOTE-002 phase 2 trial compared 2 different pembrolizumab doses (2 mg/kg and 10 mg/kg every 3 weeks) to investigator’s choice chemotherapy (paclitaxel plus carboplatin, paclitaxel, carboplatin, dacarbazine, or oral temozolomide) in 540 patients with advanced melanoma with documented progression on ipilimumab with or without prior progression on molecularly targeted therapy if positive for a BRAF V600 mutation. The final analysis demonstrated significantly improved ORR with pembrolizumab (22% at 2 mg/kg vs 26% at 10 mg/kg vs 4% chemotherapy) and significantly improved 24-month PFS (16% vs 22% vs 0.6%, respectively). There was a nonstatistically significant improvement in median OS (13.4 months vs 14.7 months vs 10 months), although 55% of the patients initially assigned to the chemotherapy arm crossed over and received pembrolizumab after documentation of progressive disease.23,24
Because PD-1 inhibition improved efficacy with less toxicity than chemotherapy when studied in progressive disease, subsequent studies focused on PD-1 inhibition in the frontline setting. CheckMate 066 was a phase 3 trial comparing nivolumab to dacarbazine as first-line therapy for 418 patients with untreated metastatic melanoma who did not have a BRAF mutation. For the primary end point of 1-year OS, nivolumab was superior to dacarbazine (72.9% vs 42.1%; hazard ratio [HR], 0.42; P < 0.001). Treatment with nivolumab also resulted in superior ORR (40% vs 14%) and PFS (5.1 months vs 2.2 months). Additionally, nivolumab therapy had a lower rate of grade 3 or 4 toxicity compared to dacarbazine (11.7% vs 17.6%).25
The KEYNOTE-006 trial compared 2 separate dosing schedules of pembrolizumab (10 mg/kg every 2 weeks versus every 3 weeks) to ipilimumab (3 mg/kg every 3 weeks for 4 cycles) in a 1:1:1 ratio in 834 patients with metastatic melanoma who had received up to 1 prior systemic therapy, but no prior CTLA-4 or PD-1 inhibitors. The first published data reported statistically significant outcomes for the co-primary end points of 6-month PFS (47.3% for pembrolizumab every 2 weeks vs 46.4% for pembrolizumab every 3 weeks vs 26.5% for ipilimumab; HR, 0.58 for both pembrolizumab groups compared to ipilimumab; P < 0.001) and 12-month OS (74.1% vs 68.4% vs 58.2%) with pembrolizumab compared to ipilimumab. Compared to ipilimumab, pembrolizumab every 2 weeks had a hazard ratio of 0.63 (P = 0.0005) and pembrolizumab every 3 weeks had a hazard ratio of 0.69 (P = 0.0036). The pembrolizumab groups was also had lower rates of grade 3 to 5 toxicity (13.3% vs 10.1% vs 19.9%).5 Updated outcomes demonstrated improved ORR compared to the first analysis (37% vs 36% vs 13%), and improved OS (median OS, not reached for the pembrolizumab groups vs 16.0 months for the ipilimumab group; HR, 0.68, P = 0.0009 for pembrolizumab every 2 weeks versus HR 0.68, P = 0.0008 for pembrolizumab every 3 weeks).26 In addition, 24-month OS was 55% in both pembrolizumab groups compared to 43% in the ipilimumab group. Grade 3 or 4 toxicity occurred less frequently with pembrolizumab (17% vs 17% vs 20%).
Further analysis from the KEYNOTE-006 trial data demonstrated improved ORR, PFS, and OS with pembrolizumab compared to ipilimumab in tumors positive for PD-L1 expression. For PD-L1-negative tumors, response rate was higher, and PFS and OS rates were similar with pembrolizumab compared to ipilimumab. Given that pembrolizumab was associated with similar survival outcomes in PD-L1-negative tumors and with less toxicity than ipilimumab, the superiority of PD-L1 inhibitors over ipilimumab was further supported, regardless of tumor PD-L1 status.27
In sum, PD-1 inhibition should be considered the first-line immunotherapy in advanced melanoma, either alone or in combination with ipilimumab, as discussed in the following section. There is no longer a role for ipilimumab monotherapy in the first-line setting, based on evidence from direct comparison to single-agent PD-1 inhibition in clinical trials that demonstrated superior efficacy and less serious toxicity with PD-1 inhibitors.5,26 The finding that ORR and OS outcomes with single-agent PD-1 inhibitors are higher in treatment-naive patients compared to those receiving prior therapies also supports this approach.22
Combination CTLA-4 and PD-1 Therapy
Despite the potential for durable responses, the majority of patients fail to respond to single-agent PD-1 therapy. Given that preclinical data had suggested the potential for synergy between dual inhibition of CTLA-4 and PD-1, clinical trials were designed to test this approach. The first randomized phase 2 trial that established superior efficacy with combination therapy was the CheckMate 069 trial comparing nivolumab plus ipilimumab to ipilimumab monotherapy. Combination therapy resulted in increased ORR (59% vs 11%), median PFS (not reached vs 3.0 months), 2-year PFS (51.3% vs 12.0%), and 2-year OS (63.8% vs 53.6%).28 Similarly, a phase 1b trial of pembrolizumab plus reduced-dose ipilimumab demonstrated an ORR of 61%, with a 1-year PFS of 69% and 1-year OS of 89%.29
The landmark phase 3 CheckMate 067 trial analyzed efficacy outcomes for 3 different treatment regimens including nivolumab plus ipilimumab, nivolumab monotherapy, and ipilimumab monotherapy in previously untreated patients with unresectable stage III or IV melanoma. The trial was powered to compare survival outcomes for both the combination therapy arm against ipilimumab and the nivolumab monotherapy arm against ipilimumab, but not to compare combination therapy to nivolumab monotherapy. The initial analysis demonstrated a median PFS of 11.5 months with combination therapy versus 6.9 months with nivolumab and 2.9 months with ipilimumab, as well as an ORR of 58% versus 44% and 19%, respectively (Table 1).6 The updated 3-year survival outcomes from CheckMate 067 were notable for superior median OS with combination therapy (not reached in combination vs 37.6 months for nivolumab vs 19.9 months ipilimumab), improved 3-year OS (58% vs 52% vs 34%), and improved 3-year PFS (39% vs 32% vs 10%).7 In the reported 4-year survival outcomes, median OS was not reached in the combination therapy group, and was 36.9 months in the nivolumab monotherapy group and 19.9 months in the ipilimumab monotherapy group. Rates of grade 3 or 4 adverse events were significantly higher in the combination therapy group, at 59% compared to 22% with nivolumab monotherapy and 28% with ipilimumab alone.30 The 3- and 4-year OS outcomes (58% and 54%, respectively) with combination therapy were the highest seen in any phase 3 trial for treatment of advanced melanoma, supporting its use as the best approved first-line therapy in those who can tolerate the potential toxicity of combination therapy7,30 The conclusions from this landmark trial were that both combination therapy and nivolumab monotherapy resulted in statistically significant improvement in OS compared to ipilimumab.
Toxicity Associated with Immune Checkpoint Inhibitors
While immune checkpoint inhibitors have revolutionized the treatment of many solid tumor malignancies, this new class of cancer therapy has brought about a new type of toxicity for clinicians to be aware of, termed immune-related adverse events (irAEs). As immune checkpoint inhibitors amplify the immune response against malignancy, they also increase the likelihood that autoreactive T-cells persist and proliferate within the circulation. Therefore, these therapies can result in almost any type of autoimmune side effect. The most commonly reported irAEs in large clinical trials studying CTLA-4 and PD-1 inhibitors include rash/pruritus, diarrhea/colitis, hepatitis, endocrinopathies (thyroiditis, hypophysitis, adrenalitis), and pneumonitis. Other more rare toxicities include pancreatitis, autoimmune hematologic toxicities, cardiac toxicity (myocarditis, heart failure), and neurologic toxicities (neuropathies, myasthenia gravis-like syndrome, Guillain-Barré syndrome). It has been observed that PD-1 inhibitors have a lower incidence of irAEs than CTLA-4 inhibitors, and that the combined use of PD-1 and CTLA-4 inhibitors is associated with a greater incidence of irAEs compared to monotherapy with either agent.31 Toxicities associated with ipilimumab have been noted to be dose dependent.18 Generally, these toxicities are treated with immunosuppression in the form of glucocorticoids and are often reversible.31 There are several published guidelines that include algorithms for the management of irAEs by organizations such as the National Comprehensive Cancer Network.32
For example, previously untreated patients treated with ipilimumab plus dacarbazine as compared to dacarbazine plus placebo had greater grade 3 or 4 adverse events (56.3% vs 27.5%), and 77.7% of patients experiencing an irAE of any grade.18 In the CheckMate 066 trial comparing frontline nivolumab to dacarbazine, nivolumab had a lower rate of grade 3 or 4 toxicity (11.7% vs 17.6%) and irAEs were relatively infrequent, with diarrhea and elevated alanine aminotransferase level each being the most prominent irAE (affecting 1.0% of patients).25 In the KEYNOTE-006 trial, irAEs seen in more than 1% of patients treated with pembrolizumab included colitis, hepatitis, hypothyroidism, and hyperthyroidism, whereas those occurring in more than 1% of patients treated with ipilimumab included colitis and hypophysitis. Overall, there were lower rates of grade 3 to 5 toxicity with the 2 pembrolizumab doses compared to ipilimumab (13.3% pembrolizumab every 2 weeks vs 10.1% pembrolizumab every 3 weeks vs 19.9% ipilimumab).5 In the CheckMate 067 trial comparing nivolumab plus ipilimumab, nivolumab monotherapy, and ipilimumab monotherapy, rates of treatment-related adverse events of any grade were higher in the combination group (96% combination vs 86% nivolumab vs 86% ipilimumab), as were rates of grade 3 or 4 adverse events (59% vs 21% vs 28%, respectively). The irAE profile was similar to that demonstrated in prior studies: rash/pruritus were the most common, and diarrhea/colitis, elevated aminotransferases, and endocrinopathies were among the more common irAEs.7
Alternative dosing strategies have been investigated in an effort to preserve efficacy and minimize toxicity. A phase 1b trial of pembrolizumab plus reduced-dose ipilimumab demonstrated an ORR of 61%, with a 1-year PFS of 69% and a 1-year OS of 89%. This combination led to 45% of patients having a grade 3 or 4 adverse event, 60% having irAEs of any grade, and only 27% having grade 3 or 4 irAEs.29 The CheckMate 067 trial studied the combination of nivolumab 1 mg/kg plus ipilimumab 3 mg/kg.6 The CheckMate 511 trial compared different combination dosing strategies (nivolumab 3 mg/kg + ipilimumab 1 mg/kg versus nivolumab 1 mg/kg + ipilimumab 3 mg/kg) to assess for safety benefit. In the results published in abstract form, the reduced ipilimumab dose (nivolumab 3 mg/kg + ipilimumab 1 mg/kg arm) resulted in significantly decreased grade 3 to 5 adverse events (33.9% vs 48.3%) without significant differences in ORR, PFS, or OS.33
The question about the efficacy of checkpoint inhibitors in patients who discontinue treatment due to irAEs has been raised, as one hypothesis suggests that such toxicities may also indicate that the antitumor immune response has been activated. In a retrospective pooled analysis of phase 2 and 3 trials where patients received combination therapy with ipilimumab and nivolumab and discontinued therapy during the induction phase due to irAEs, outcomes did not appear to be inferior. Median PFS was 8.4 months in those who discontinued therapy compared to 10.8 months in those who continued therapy, but this did not reach statistical significance. Median OS had not been reached in either group and ORR was actually higher in those who discontinued due to adverse events (58.3% vs 50.2%). While this retrospective analysis needs to be validated, it does suggest that patients likely derive antitumor benefit from immunotherapy even if they have to discontinue therapy due to irAEs. Of note, patients in this analysis were not trialed on nivolumab monotherapy after receiving immunosuppressive treatment for toxicity related to combination therapy, which has since been deemed a reasonable treatment option.34
Molecularly Targeted Therapy for Metastatic Melanoma
As previously mentioned, the MAPK pathway is frequently altered in metastatic melanoma and thus serves as a target for therapy. Mutations in BRAF can cause constitutive activation of the protein’s kinase function, which subsequently phosphorylates/activates MEK in the absence of extracellular growth signals and causes increased cellular proliferation. For the roughly half of patients diagnosed with metastatic melanoma who harbor a BRAF V600 mutation, molecularly targeted therapy with BRAF/MEK inhibitors has emerged as a standard of care treatment option. As such, all patients with advanced disease should be tested for BRAF mutations.
After early phase 1 studies of the BRAF inhibitor vemurafenib demonstrated successful inhibition of mutated BRAF,35 subsequent studies confirmed the benefit of BRAF targeted therapy. In the phase 3 randomized controlled BRIM-3 trial comparing vemurafenib with dacarbazine for treatment of 675 patients with previously untreated metastatic melanoma positive for a BRAF V600E mutation, the vemurafenib group had superior ORR and 6-month OS during the first analysis.36 In a subsequent analysis, median PFS and median OS were also superior with vemurafenib compared to dacarbazine, as vemurafenib had a median OS of 13.6 months compared to 9.7 months with dacarbazine (HR, 0.70; P = 0.0008).37 Dabrafenib was the next BRAF inhibitor to demonstrate clinical efficacy with superior PFS compared to dacarbazine.38
Despite tumor shrinkage in the majority of patients, the development of resistance to therapy was an issue early on. The development of acquired resistance emerged as a heterogeneous process, though many of the identified resistance mechanisms involved reactivation of the MAPK pathway.39 A phase 3 trial of 322 patients with metastatic melanoma comparing the MEK inhibitor trametinib as monotherapy against chemotherapy demonstrated a modest improvement in both median PFS and OS.40 As a result, subsequent efforts focused on a strategy of concurrent MEK inhibition as a means to overcome resistance to molecularly targeted monotherapy
At least 4 large phase 3 randomized controlled trials of combination therapy with BRAF plus MEK inhibitors showed an improved ORR, PFS, and OS when compared to BRAF inhibition alone. The COMBI-d trial comparing dabrafenib plus trametinib versus dabrafenib alone was the first to demonstrate the superiority of combined BRAF/MEK inhibition and made combination therapy the current standard of care for patients with metastatic melanoma and a BRAF V600 mutation. In the final analysis of this trial, 3-year PFS was 22% with combination therapy compared to 12% with dabrafenib alone, and 3-year OS was 44% compared to 32%.8,41,42 A second trial with the combination of dabrafenib and trametinib (COMBI-V) also demonstrated superior efficacy when compared to single-agent vemurafenib without increased toxicity.43 Subsequently, the combination of vemurafenib with MEK inhibitor cobimetinib demonstrated superiority compared to vemurafenib alone,44 followed by the newest combination encorafenib (BRAF inhibitor) and binimetinib (MEK inhibitor) proving superior to either vemurafenib or encorafenib alone.45,46
It is important to note that there have been no studies directly comparing the efficacy of the 3 approved BRAF/MEK inhibitor combinations, but the 3 different regimens have some differences in their toxicity profiles (Table 2). Of note, single-agent BRAF inhibition was associated with increased cutaneous toxicity, including secondary squamous cell carcinoma and keratoacanthoma,47 which was demonstrated to be driven by paradoxical activation of the MAPK pathway.48 The concerning cutaneous toxicities such as squamous cell carcinoma were substantially reduced by combination BRAF/MEK inhibitor therapy.47 Collectively, the higher efficacy along with manageable toxicity profile established combination BRAF/MEK inhibition as the preferred regimen for patients with BRAF-mutated metastatic melanoma who are being considered for molecularly targeted therapy. BRAF inhibitor monotherapy should only be used when there is a specific concern regarding the use of a MEK inhibitor in certain clinical circumstances.
Other driver mutations associated with metastatic melanoma such as NRAS-mutated tumors have proven more difficult to effectively treat with molecularly targeted therapy, with one study showing that the MEK inhibitor binimetinib resulted in a modest improvement in ORR and median PFS without OS benefit compared to dacarbazine.49 Several phase 2 trials involving metastatic melanoma harboring a c-Kit alteration have demonstrated some efficacy with the tyrosine kinase inhibitor imatinib. The largest phase 2 trial of 43 patients treated with imatinib resulted in a 53.5% disease control rate (23.3% partial response and 30.2% stable disease), with 9 of the 10 patients who achieved partial response having a mutation in either exon 11 or 13. Median PFS was 3.5 months and 1-year OS was 51.0%.50
Case Conclusion
Prior to initiation of systemic therapy, the patient’s melanoma is tested and is found to be positive for a BRAF V600K mutation. At his follow-up appointment, the patient continues to endorse generalized weakness, fatigue, issues with balance, and residual pulmonary symptoms after being treated for post-obstructive pneumonia. Given his current symptoms and extent of metastatic disease, immunotherapy is deferred and he is started on combination molecularly targeted therapy with dabrafenib and trametinib. He initially does well, with a partial response noted by resolution of symptoms and decreased size of his intracranial metastases and decreased size of the right lower lobe mass. Further follow-up of this patient is presented in the second article in this 2-part review of advanced melanoma.
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2018. CA Cancer J Clin. 2018;68:7-30.
2. Ives NJ, Stowe RL, Lorigan P, Wheatley K. Chemotherapy compared with biochemotherapy for the treatment of metastatic melanoma: a meta-analysis of 18 trials involving 2621 patients. J Clin Oncol. 2007;25:5426-34.
3. Atkins MB, Lotze MT, Dutcher JP, et al. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J Clin Oncol. 1999;17:2105-16.
4. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-23.
5. Robert C, Schachter J, Long GV, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372:2522-2532.
6. Larkin J, Chiarion-Sileni V, Gonazalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373:23-34.
7. Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377:1345-1356.
8. Long GV, Stroyakovskiy D, Gogas H, et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med. 2014;371:1877-1888.
9. Elwood JM, Jopson J. Melanoma and sun exposure: an overview of published studies. Int J Cancer. 1997;73:198-203.
10. Gilchrest BA, Eller MS, Geller AC, Yaar M. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 199;340:1341-1348.
11. Omholt K, Platz A, Kanter L, et al. NRAS and BRAF mutations arise early during melanoma pathogenesis and are preserved throughout tumor progression. Clin Cancer Res. 2003;9:6483-8.
12. Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-54.
13. Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 2015;161:1681-96.
14. Gershenwald JE, Scolyer RA, Hess KR, et al. Melanoma staging: evidence-based changes in the American Joint Committee on Cancer Eighth Edition Cancer Staging Manual. CA Cancer J Clin. 2017;67:472-492.
15. Robert C, Ribas A, Hamid O, et al. Durable complete response after discontinuation of pembrolizumab in patients with metastatic melanoma. J Clin Oncol. 2018;36:1668-1674.
16. Salama AKS, Hodi FS. Cytotoxic T-lymphocyte-associated antigen-4. Clin Cancer Res. 2011;17:4622-8.
17. Boussiotis VA. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med. 2016;375:1767-1778.
18. Robert C, Thomas L, Bondarenko I, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364:2517-2526.
19. Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454.
20. Topalian S, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020-30.
21. Weber JS, D’Angelo SP, Minor D, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16:375-84.
22. Ribas A, Hamid O, Daud A, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600-1609.
23. Ribas A, Puzanov I, Dummer R, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16:908-18.
24. Hamid O, Puzanov I, Dummer R, et al. Final analysis of a randomised trial comparing pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory advanced melanoma. Eur J Cancer. 2017;86:37-45.
25. Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
26. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicenter, randomised, open-label phase 3 study (KEYNOTE-006). Lancet Oncol. 2017;390:1853-1862.
27. Carlino MS, Long GV, Schadendorf D, et al. Outcomes by line of therapy and programmed death ligand 1 expression in patients with advanced melanoma treated with pembrolizumab or ipilimumab in KEYNOTE-006. A randomised clinical trial. Eur J Cancer. 2018;101:236-243.
28. Hodi FS, Chesney J, Pavlick AC, et al. Combined nivolumab and ipilimumab versus ipilimumab alone in patients with advanced melanoma: 2-year overall survival outcomes in a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol. 2016;17:1558-1568.
29. Long GV, Atkinson V, Cebon JS, et al. Standard-dose pembrolizumab in combination with reduced-dose ipilimumab for patients with advanced melanoma (KEYNOTE-029): an open-label, phase 1b trial. Lancet Oncol. 2017;18:1202-10.
30. Hodi FS, Chiarion-Sileni V, Gonzalez R, et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018;19:1480-1492.
31. Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the immune-related adverse effects of immune checkpoint inhibitors: a review. JAMA Oncol. 2016;2:1346-1353.
32. National Comprehensive Cancer Network. Management of immunotherapy-related toxicities (version 2.2019). www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed April 8, 2019.
33. Lebbé C, Meyer N, Mortier L, et al. Initial results from a phase IIIb/IV study evaluating two dosing regimens of nivolumab (NIVO) in combination with ipilimumab (IPI) in patients with advanced melanoma (CheckMate 511) [Abstract LBA47]. Ann Oncol. 2018;29:mdy424.057.
34. Schadendorf D, Wolchok JD, Hodi FS, et al. Efficacy and safety outcomes in patients with advanced melanoma who discontinued treatment with nivolumab and ipilimumab because of adverse events: a pooled analysis of randomized phase ii and iii trials. J Clin Oncol. 2017;35:3807-3814.
35. Flaherty KT, Puzanov I, Kim KB, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809-819.
36. Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
37. McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAFV600E and BRAFV600K mutation-positive melanoma (BRIM-3): extended follow up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15:323-332.
38. Hauschild A, Grob JJ, Demidov LV, et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicenter, open-label, phase 3 randomised controlled trial. Lancet Oncol. 2012;380:358-365.
39. Rizos H, Menzies AM, Pupo GM, et al. BRAF inhibitor resistance mechanisms in metastatic melanoma: spectrum and clinical impact. Clin Cancer Res. 2014;20:1965-1977.
40. Flaherty KT, Robert C, Hersey P, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367:107-114.
41. Long GV, Stroyakovskiy D, Gogas H, et al. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: a multicenter, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2015;386:444-451.
42. Long GV, Flaherty KT, Stroyakovskiy D, et al. Dabrafenib plus trametinib versus dabrafenib monotherapy in patients with metastatic BRAF V600E/K-mutant melanoma: long-term survival and safety analysis of a phase 3 study. Ann Oncol. 2017;28:1631-1639.
43. Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30-39.
44. Ascierto PA, McArthur GA, Dréno B, et al. Cobimetinib combined with vemurafenib in advanced BRAFV600-mutant melanoma (coBRIM): updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016;17:1248-260.
45. Dummer R, Ascierto PA, Gogas HJ, et al. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): a multicenter, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:603-615.
46. Dummer R, Ascierto PA, Gogas HJ, et al. Overall survival in patients with BRAF-mutant melanoma receiving encorafenib plus binimetinib versus vemurafenib or encorafenib (COLUMBUS): a multicenter, open-label, randomised, phase 3 trial. Lancet Oncol. 2018;19:1315-1327.
47. Carlos G, Anforth R, Clements A, et al. Cutaneous toxic effects of BRAF inhibitors alone and in combination with MEK inhibitors for metastatic melanoma. JAMA. Dermatol 2015;151:1103-1109.
48. Su F, Viros A, Milagre C, et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N Engl J Med. 2012;366:207-215.
49. Dummer R, Schadendorf D, Ascierto P, et al. Binimetinib versus dacarbazine in patients with advanced NRAS-mutant melanoma (NEMO): a multicenter, open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18:435-445.
50. Guo J, Si L, Kong Y, et al. Phase II, open-label, single-arm trial of imatinib mesylate in patients with metastatic melanoma harboring c-Kit mutation or amplification. J Clin Oncol. 2011;29:2904-2909.
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2018. CA Cancer J Clin. 2018;68:7-30.
2. Ives NJ, Stowe RL, Lorigan P, Wheatley K. Chemotherapy compared with biochemotherapy for the treatment of metastatic melanoma: a meta-analysis of 18 trials involving 2621 patients. J Clin Oncol. 2007;25:5426-34.
3. Atkins MB, Lotze MT, Dutcher JP, et al. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J Clin Oncol. 1999;17:2105-16.
4. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-23.
5. Robert C, Schachter J, Long GV, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372:2522-2532.
6. Larkin J, Chiarion-Sileni V, Gonazalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373:23-34.
7. Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377:1345-1356.
8. Long GV, Stroyakovskiy D, Gogas H, et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med. 2014;371:1877-1888.
9. Elwood JM, Jopson J. Melanoma and sun exposure: an overview of published studies. Int J Cancer. 1997;73:198-203.
10. Gilchrest BA, Eller MS, Geller AC, Yaar M. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 199;340:1341-1348.
11. Omholt K, Platz A, Kanter L, et al. NRAS and BRAF mutations arise early during melanoma pathogenesis and are preserved throughout tumor progression. Clin Cancer Res. 2003;9:6483-8.
12. Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-54.
13. Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 2015;161:1681-96.
14. Gershenwald JE, Scolyer RA, Hess KR, et al. Melanoma staging: evidence-based changes in the American Joint Committee on Cancer Eighth Edition Cancer Staging Manual. CA Cancer J Clin. 2017;67:472-492.
15. Robert C, Ribas A, Hamid O, et al. Durable complete response after discontinuation of pembrolizumab in patients with metastatic melanoma. J Clin Oncol. 2018;36:1668-1674.
16. Salama AKS, Hodi FS. Cytotoxic T-lymphocyte-associated antigen-4. Clin Cancer Res. 2011;17:4622-8.
17. Boussiotis VA. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med. 2016;375:1767-1778.
18. Robert C, Thomas L, Bondarenko I, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364:2517-2526.
19. Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454.
20. Topalian S, Sznol M, McDermott DF, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020-30.
21. Weber JS, D’Angelo SP, Minor D, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16:375-84.
22. Ribas A, Hamid O, Daud A, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315:1600-1609.
23. Ribas A, Puzanov I, Dummer R, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16:908-18.
24. Hamid O, Puzanov I, Dummer R, et al. Final analysis of a randomised trial comparing pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory advanced melanoma. Eur J Cancer. 2017;86:37-45.
25. Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
26. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicenter, randomised, open-label phase 3 study (KEYNOTE-006). Lancet Oncol. 2017;390:1853-1862.
27. Carlino MS, Long GV, Schadendorf D, et al. Outcomes by line of therapy and programmed death ligand 1 expression in patients with advanced melanoma treated with pembrolizumab or ipilimumab in KEYNOTE-006. A randomised clinical trial. Eur J Cancer. 2018;101:236-243.
28. Hodi FS, Chesney J, Pavlick AC, et al. Combined nivolumab and ipilimumab versus ipilimumab alone in patients with advanced melanoma: 2-year overall survival outcomes in a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol. 2016;17:1558-1568.
29. Long GV, Atkinson V, Cebon JS, et al. Standard-dose pembrolizumab in combination with reduced-dose ipilimumab for patients with advanced melanoma (KEYNOTE-029): an open-label, phase 1b trial. Lancet Oncol. 2017;18:1202-10.
30. Hodi FS, Chiarion-Sileni V, Gonzalez R, et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018;19:1480-1492.
31. Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the immune-related adverse effects of immune checkpoint inhibitors: a review. JAMA Oncol. 2016;2:1346-1353.
32. National Comprehensive Cancer Network. Management of immunotherapy-related toxicities (version 2.2019). www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed April 8, 2019.
33. Lebbé C, Meyer N, Mortier L, et al. Initial results from a phase IIIb/IV study evaluating two dosing regimens of nivolumab (NIVO) in combination with ipilimumab (IPI) in patients with advanced melanoma (CheckMate 511) [Abstract LBA47]. Ann Oncol. 2018;29:mdy424.057.
34. Schadendorf D, Wolchok JD, Hodi FS, et al. Efficacy and safety outcomes in patients with advanced melanoma who discontinued treatment with nivolumab and ipilimumab because of adverse events: a pooled analysis of randomized phase ii and iii trials. J Clin Oncol. 2017;35:3807-3814.
35. Flaherty KT, Puzanov I, Kim KB, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809-819.
36. Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
37. McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAFV600E and BRAFV600K mutation-positive melanoma (BRIM-3): extended follow up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15:323-332.
38. Hauschild A, Grob JJ, Demidov LV, et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicenter, open-label, phase 3 randomised controlled trial. Lancet Oncol. 2012;380:358-365.
39. Rizos H, Menzies AM, Pupo GM, et al. BRAF inhibitor resistance mechanisms in metastatic melanoma: spectrum and clinical impact. Clin Cancer Res. 2014;20:1965-1977.
40. Flaherty KT, Robert C, Hersey P, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367:107-114.
41. Long GV, Stroyakovskiy D, Gogas H, et al. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: a multicenter, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2015;386:444-451.
42. Long GV, Flaherty KT, Stroyakovskiy D, et al. Dabrafenib plus trametinib versus dabrafenib monotherapy in patients with metastatic BRAF V600E/K-mutant melanoma: long-term survival and safety analysis of a phase 3 study. Ann Oncol. 2017;28:1631-1639.
43. Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30-39.
44. Ascierto PA, McArthur GA, Dréno B, et al. Cobimetinib combined with vemurafenib in advanced BRAFV600-mutant melanoma (coBRIM): updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016;17:1248-260.
45. Dummer R, Ascierto PA, Gogas HJ, et al. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): a multicenter, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:603-615.
46. Dummer R, Ascierto PA, Gogas HJ, et al. Overall survival in patients with BRAF-mutant melanoma receiving encorafenib plus binimetinib versus vemurafenib or encorafenib (COLUMBUS): a multicenter, open-label, randomised, phase 3 trial. Lancet Oncol. 2018;19:1315-1327.
47. Carlos G, Anforth R, Clements A, et al. Cutaneous toxic effects of BRAF inhibitors alone and in combination with MEK inhibitors for metastatic melanoma. JAMA. Dermatol 2015;151:1103-1109.
48. Su F, Viros A, Milagre C, et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N Engl J Med. 2012;366:207-215.
49. Dummer R, Schadendorf D, Ascierto P, et al. Binimetinib versus dacarbazine in patients with advanced NRAS-mutant melanoma (NEMO): a multicenter, open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18:435-445.
50. Guo J, Si L, Kong Y, et al. Phase II, open-label, single-arm trial of imatinib mesylate in patients with metastatic melanoma harboring c-Kit mutation or amplification. J Clin Oncol. 2011;29:2904-2909.
Management of Late Pulmonary Complications After Hematopoietic Stem Cell Transplantation
Hematopoietic stem cell transplantation (HSCT) is increasingly being used to treat hematologic malignancies as well as nonmalignant diseases and solid tumors. Over the past 2 decades overall survival following transplant and transplant-related mortality have improved.1 With this increased survival, there is a need to focus on late complications after transplantation. Pulmonary complications are a common but sometimes underrecognized cause of late morbidity and mortality in HSCT patients. This article, the second of 2 articles on post-HSCT pulmonary complications, reviews late-onset complications, with a focus on the evaluation and treatment of bronchiolitis obliterans syndrome (BOS), one of the most common and serious late pulmonary complications in HSCT patients. The first article reviewed the management of early-onset pulmonary complications and included a basic overview of stem cell transplantation, discussion of factors associated with pulmonary complications, and a review of methods for assessing pretransplant risk for pulmonary complications in patients undergoing HSCT.2
Case Presentation
A 40-year-old white woman with a history of acute myeloid leukemia status post peripheral blood stem cell transplant presents with dyspnea on exertion, which she states started about 1 month ago and now is limiting her with even 1 flight of stairs. She also complains of mild dry cough and a 4- to 5-lb weight loss over the past 1 to 2 months. She has an occasional runny nose, but denies gastroesophageal reflux, fevers, chills, or night sweats. She has a history of matched related sibling donor transplant with busulfan and cyclophosphamide conditioning 1 year prior to presentation. She has had significant graft-versus-host disease (GVHD), affecting the liver, gastrointestinal tract, skin, and eyes.
On physical examination, heart rate is 110 beats/min, respiratory rate is 16 breaths/min, blood pressure is 92/58 mm Hg, and the patient is afebrile. Eye exam reveals scleral injection, mouth shows dry mucous membranes with a few white plaques, and the skin has chronic changes with a rash over both arms. Cardiac exam reveals tachycardia but regular rhythm and there are no murmurs, rubs, or gallops. Lungs are clear bilaterally and abdomen shows no organomegaly.
Laboratory exam shows a white blood cell count of 7800 cells/μL, hemoglobin level of 12.4 g/dL, and platelet count of 186 × 103/μL. Liver enzymes are mildly elevated. Chest radiograph shows clear lung fields bilaterally.
- What is the differential in this patient with dyspnea 1 year after transplantation?
Late pulmonary complications are generally accepted as those occurring more than 100 days post transplant. This period of time is characterized by chronic GVHD and impaired cellular and humoral immunity. Results of longitudinal studies of infections in adult HSCT patients suggest that special attention should be paid to allogeneic HSCT recipients for post-engraftment infectious pulmonary complications.3 Encapsulated bacteria such as Haemophilus influenzae and Streptococcus pneumoniae are the most frequent bacterial organisms causing late infectious pulmonary complications. Nontuberculous mycobacteria and Nocardia should also be considered. Depending upon geographic location, social and occupational risk factors, and prevalence, tuberculosis should also enter the differential.
There are many noninfectious late-onset pulmonary complications after HSCT. Unfortunately, the literature has divided pulmonary complications after HSCT using a range of criteria and classifications based upon timing, predominant pulmonary function test (PFT) findings, and etiology. These include early versus late, obstructive versus restrictive, and infectious versus noninfectious, which makes a comprehensive literature review of late pulmonary complications difficult. The most common noninfectious late-onset complications are bronchiolitis obliterans, cryptogenic organizing pneumonia (previously referred to as bronchiolitis obliterans organizing pneumonia, or BOOP), and interstitial pneumonia. Other rarely reported complications include eosinophilic pneumonia, pulmonary alveolar proteinosis, air leak syndrome, and pulmonary hypertension.
Case Continued
Because the patient does not have symptoms of infection, PFTs are obtained. Pretransplant PFTs and current PFTs are shown in Table 1.
- What is the diagnosis in this case?
Bronchiolitis Obliterans
BOS is one of the most common and most serious late-onset pulmonary diseases after allogeneic transplantation. It is considered the pulmonary form of chronic GVHD. BOS was first described in 1982 in patients with chronic GVHD after bone marrow transplantation.4 Many differing definitions of bronchiolitis obliterans have been described in the literature. A recent review of the topic cites 10 different published sets of criteria for the diagnosis of bronchiolitis obliterans.5 Traditionally, bronchiolitis obliterans was thought to occur in 2% to 8% of patients undergoing allogeneic HSCT, but these findings were from older studies that used a diagnosis based on very specific pathology findings. When more liberal diagnostic criteria are used, the incidence may be as high as 26% of allogeneic HSCT patients.6
Bronchiolitis obliterans is a progressive lung disease characterized by narrowing of the terminal airways and obliteration of the terminal bronchi. Pathology may show constrictive bronchiolitis but can also show lymphocytic bronchiolitis, which may be associated with a better outcome.7 As noted, bronchiolitis obliterans has traditionally been considered a pathologic diagnosis. Current diagnostic criteria have evolved based upon the difficulty in obtaining this diagnosis through transbronchial biopsy given the patchy nature of the disease.8 The gold standard of open lung biopsy is seldom pursued in the post-HSCT population as the procedure continues to carry a worrisome risk-benefit profile.
The 2005 National Institutes of Health (NIH) consensus development project on criteria for clinical trials in chronic GVHD developed a clinical strategy for diagnosing BOS using the following criteria: absence of active infection, decreased forced expiratory volume in 1 second (FEV1) < 75%, FEV1/forced vital capacity (FVC) ratio of < 70%, and evidence of air trapping on high-resolution computed tomography (HRCT) or PFTs (residual volume > 120%). These diagnostic criteria were applied to a small series of patients with clinically identified bronchiolitis obliterans or biopsy-proven bronchiolitis obliterans. Only 18% of these patients met the requirements for the NIH consensus definition.5 A 2011 study that applied the NIH criteria found an overall prevalence of 5.5% among all transplant recipients but a prevalence of 14% in patients with GVHD.9 In 2014, the NIH consensus development group updated their recommendations. The new criteria for diagnosis of BOS require the presence of airflow obstruction (FEV1/FVC < 70% or 5th percentile of predicted), FEV1 < 75% predicted with a ≥ 10% decline in fewer than 2 years, absence of infection, and presence of air trapping (by expiratory computed tomography [CT] scan or PFT with residual volume >120% predicted) (Table 2).
Some issues must be considered when determining airflow obstruction. The 2005 NIH working group recommends using Crapo as the reference set,11 but the National Health and Nutrition Examination Survey (NHANES) III reference values are the preferred reference set at this time12 and should be used in the United States. A recent article showed that the NHANES values were superior to older reference sets (however, they did not use Crapo as the comparison), although this study used the lower limit of normal as compared with the fixed 70% ratio.13 The 2014 NIH consensus group does not recommend a specific reference set and recognizes an FEV1/FVC ratio of 70% or less than the lower limit of normal as the cutoff value for airflow obstruction.10
Another issue in PFT interpretation is the finding of a decrease in FEV1 and FVC and normal total lung capacity, which is termed a nonspecific pattern. This pattern has been reported to occur in 9% of all PFTs and usually is associated with obstructive lung disease or obesity.14 A 2013 study described the nonspecific pattern as a BOS subgroup occurring in up to 31% of bronchiolitis obliterans patients.15
- What are the radiographic findings of BOS?
Chest radiograph is often normal in BOS. As discussed, air trapping can be documented using HRCT, according to the NIH clinical definition of bronchiolitis obliterans.16 A study that explored findings and trends seen on HRCT in HSCT patients with BOS found that the syndrome in these patients is characterized by central airway dilatation.17 Expiratory airway trapping on HRCT is the main finding, and this is best demonstrated on HRCT during inspiratory and expiratory phases.18 Other findings are bronchial wall thickening, parenchymal hypoattenuation, bronchiectasis, and centrilobular nodules.19
Galbán and colleagues developed a new technique called parametric response mapping that uses CT scanners to quantify normal parenchyma, functional small airway disease, emphysema, and parenchymal disease as relative lung volumes.20 This technique can detect airflow obstruction and small airway disease and was found to be a good method for detecting BOS after HSCT. In their study of parametric response mapping, the authors found that functional small airway disease affecting 28% or more of the total lung was highly indicative of bronchiolitis obliterans.20
- What therapies are used to treat BOS?
Traditionally, BOS has been treated with systemic immunosuppression. The recommended treatment had been systemic steroids at approximately 1 mg/ kg. However, it is increasingly recognized that BOS responds poorly to systemic steroids, and systemic steroids may actually be harmful and associated with increased mortality.15,21 The chronic GVHD recommendations from 2005 recommend ancillary therapy with inhaled corticosteroids and pulmonary rehabilitation.11 The updated 2011 German consensus statement lays out a clear management strategy for mild and moderate-severe disease with monitoring recommendations.22 The 2014 NIH chronic GVHD working group recommends fluticasone, azithromycin, and montelukast (ie, the FAM protocol) for treating BOS.23 FAM therapy in BOS may help lower the systemic steroid dose.24,25 Montelukast is not considered a treatment mainstay for BOS after lung transplant, but there is a study showing possible benefit in chronic GVHD.26 An evaluation of the natural history of a cohort of BOS patients treated with FAM therapy showed a rapid decline of FEV1 in the 6 months prior to diagnosis and treatment of BOS and subsequent stabilization following diagnosis and treatment.27 The benefit of high-dose inhaled corticosteroids or the combination of inhaled corticosteroids and long-acting beta-agonists has been demonstrated in small studies, which showed that these agents stabilized FEV1 and avoided the untoward side effects of systemic corticosteroids.28–30
Macrolide antibiotics have been explored as a treatment for BOS post HSCT because pilot studies suggested that azithromycin improved or stabilized FEV1 in patients with BOS after lung transplant or HSCT.31–33 Other studies of azithromycin have not shown benefit in the HSCT population after 3 months of therapy.34 A recent meta-analysis could neither support or refute the benefit of azithromycin for BOS after HSCT.35 In the lung transplant population, a study showed that patients who were started on azithromycin after transplant and continued on it 3 times a week had improved FEV1; these patients also had a reduced rate of BOS and improved overall and BOS-free survival 2 years after transplant.36 However, these benefits of azithromycin have not been observed in patients after HSCT. In fact, the ALLOZITHRO trial was stopped early because prophylactic azithromycin started at the time of the conditioning regimen with HSCT was associated with increased hematologic disease relapse, a decrease in airflow-decline-free survival, and reduced 2-year survival.30
Azithromycin is believed to exert an effect by its anti-inflammatory properties and perhaps by decreasing lung neutrophilia (it may be most beneficial in the subset of patients with high neutrophilia on bronchoalveolar lavage [BAL]).30 Adverse effects of chronic azithromycin include QT prolongation, cardiac arrhythmia, hearing loss, and antibiotic-resistant organism colonization.37,38
Other therapies include pulmonary rehabilitation, which may improve health-related quality of life and 6-minute walk distance,39 extracorporeal photopheresis,40 immunosuppression with calcineurin inhibitors or mycophenolate mofetil,21,41 and lung transplantation.42–44 A study with imatinib for the treatment of lung disease in steroid-refractory GVHD has shown promising results, but further validation with larger clinical trials is required.45
Case Continued
The patient is diagnosed with BOS and is treated for several months with prednisone 40 mg/day weaned over 3 months. She is started on inhaled corticosteroids, a proton pump inhibitor, and azithromycin 3 times per week, but she has a progressive decline in FEV1. She starts pulmonary rehabilitation but continues to functionally decline. Over the next year she develops bilateral pneumothoraces and bilateral cavitary nodules (Figure 1).
- What is causing this decline and the radiographic abnormalities?
Spontaneous air leak syndrome has been described in a little more than 1% of patients undergoing HSCT and has included pneumothorax and mediastinal and subcutaneous emphysema.46 It appears that air leak syndrome is more likely to occur in patients with chronic GVHD.47 The association between chronic GVHD and air leak syndrome could explain this patient’s recurrent pneumothoraces. The recurrent cavitary nodules are suspicious for infectious etiologies such as nontuberculous mycobacteria, tuberculosis, and fungal infections.
Case Continued
During an episode of pneumothorax, the patient undergoes chest tube placement, pleurodesis, and lung biopsy. Pathology reveals bronchiolitis obliterans as well as organizing pneumonia (Figure 2). No organisms are seen on acid-fast bacilli or GMS stains.
- What are the other late-onset noninfectious pulmonary complications?
Definitions of other late noninfectious pulmonary complications following HSCT are shown in Table 3.
Interstitial pneumonias may represent COP or may be idiopathic pneumonia syndrome with a later onset or a nonspecific interstitial pneumonia. This syndrome is poorly defined, with a number of differing definitions of the syndrome published in the literature.50–55
A rare pulmonary complication after HSCT is pulmonary veno-occlusive disease (PVOD). Pulmonary hypertension has been reported after HSCT,56 but PVOD is a subset of pulmonary hypertension. It is associated with pleural effusions and volume overload on chest radiography.57,58 It may present early or late after transplant and is poorly understood.
Besides obstructive and restrictive PFT abnormalities, changes in small airway function59 after transplant and loss in diffusing capacity of the lungs for carbon monoxide (D
Case Conclusion
The patient’s lung function continues to worsen, but no infectious etiologies are discovered. Ultimately, she dies of respiratory failure caused by progressive bronchiolitis obliterans.
Conclusion
Late pulmonary complications occur frequently in patients who have undergone HSCT. These complications can be classified as infectious versus noninfectious etiologies. Late-onset complications are more common in allogeneic transplantations because they are associated with chronic GVHD. These complications can be manifestations of pulmonary GHVD or can be infectious complications associated with prolonged immunosuppression. Appropriate monitoring for the development of BOS is essential. Early and aggressive treatment of respiratory infections and diagnostic bronchoscopy with BAL can help elucidate most infectious causes. Still, diagnostic challenges remain and multiple causes of respiratory deterioration can be present concurrently in the post-HSCT patient. Steroid therapy remains the mainstay treatment for most noninfectious pulmonary complications and should be strongly considered once infection is effectively ruled out.
1. Remberger M, Ackefors M, Berglund S, et al. Improved survival after allogeneic hematopoietic stem cell transplantation in recent years. A single-center study. Biol Blood Marrow Transplant 2011;17:1688–97.
2. Wood KL, Esguerra VG. Management of late pulmonary complications after hematopoietic stem cell transplantation. Hosp Phys Hematology-Oncology Board Review Manual 2018;13(1):36–48.
3. Ninin E, Milpied N, Moreau P, et al. Longitudinal study of bacterial, viral, and fungal infections in adult recipients of bone marrow transplants. Clin Infect Dis 2001;33:41–7.
4. Roca J, Granena A, Rodriguez-Roisin R, et al. Fatal airway disease in an adult with chronic graft-versus-host disease. Thorax 1982;37:77–8.
5. Williams KM, Chien JW, Gladwin MT, Pavletic SZ. Bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. JAMA 2009;302:306–14.
6. Chien JW, Martin PJ, Gooley TA, et al. Airflow obstruction after myeloablative allogeneic hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2003;168:208–14.
7. Holbro A, Lehmann T, Girsberger S, et al. Lung histology predicts outcome of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013;19:973–80.
8. Chamberlain D, Maurer J, Chaparro C, Idolor L. Evaluation of transbronchial lung biopsy specimens in the diagnosis of bronchiolitis obliterans after lung transplantation. J Heart Lung Transplant 1994;13:963–71.
9. Au BK, Au MA, Chien JW. Bronchiolitis obliterans syndrome epidemiology after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2011;17:1072–8.
10. Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 2015;21:389–401.
11. Couriel D, Carpenter PA, Cutler C, et al. Ancillary therapy and supportive care of chronic graft-versus-host disease: national institutes of health consensus development project on criteria for clinical trials in chronic Graft-versus-host disease: V. Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant 2006;12:375–96.
12. Pellegrino R, Viegi G, Brusasco V, et al. Interpretative strategies for lung function tests. Eur Respir J 2005;26:948–68.
13. Williams KM, Hnatiuk O, Mitchell SA, et al. NHANES III equations enhance early detection and mortality prediction of bronchiolitis obliterans syndrome after hematopoietic SCT. Bone Marrow Transplant 2014;49:561–6.
14. Hyatt RE, Cowl CT, Bjoraker JA, Scanlon PD. Conditions associated with an abnormal nonspecific pattern of pulmonary function tests. Chest 2009;135:419–24.
15. Bergeron A, Godet C, Chevret S, et al. Bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: phenotypes and prognosis. Bone Marrow Transplant 2013;48:819–24.
16. Filipovich AH, Weisdorf D, Pavletic S, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant 2005;11:945–56.
17. Gazourian L, Coronata AM, Rogers AJ, et al. Airway dilation in bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. Respir Med 2013;107:276–83.
18. Gunn ML, Godwin JD, Kanne JP, et al. High-resolution CT findings of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. J Thorac Imaging 2008;23:244–50.
19. Sargent MA, Cairns RA, Murdoch MJ, et al. Obstructive lung disease in children after allogeneic bone marrow transplantation: evaluation with high-resolution CT. AJR Am J Roentgenol 1995;164:693–6.
20. Galban CJ, Boes JL, Bule M, et al. Parametric response mapping as an indicator of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20:1592–8.
21. Meyer KC, Raghu G, Verleden GM, et al. An international ISHLT/ATS/ERS clinical practice guideline: diagnosis and management of bronchiolitis obliterans syndrome. Eur Respir J 2014;44:1479–1503.
22. Hildebrandt GC, Fazekas T, Lawitschka A, et al. Diagnosis and treatment of pulmonary chronic GVHD: report from the consensus conference on clinical practice in chronic GVHD. Bone Marrow Transplant 2011;46:1283–95.
23. Carpenter PA, Kitko CL, Elad S, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: V. The 2014 Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant 2015;21:1167–87.
24. Norman BC, Jacobsohn DA, Williams KM, et al. Fluticasone, azithromycin and montelukast therapy in reducing corticosteroid exposure in bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: a case series of eight patients. Bone Marrow Transplant 2011;46:1369–73.
25. Williams KM, Cheng GS, Pusic I, et al. Fluticasone, azithromycin, and montelukast treatment for new-onset bronchiolitis obliterans syndrome after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2016;22:710–6.
26. Or R, Gesundheit B, Resnick I, et al. Sparing effect by montelukast treatment for chronic graft versus host disease: a pilot study. Transplantation 2007;83:577–81.
27. Cheng GS, Storer B, Chien JW, et al. Lung function trajectory in bronchiolitis obliterans syndrome after allogeneic hematopoietic cell transplant. Ann Am Thorac Soc 2016;13:1932–9.
28. Bergeron A, Belle A, Chevret S, et al. Combined inhaled steroids and bronchodilatators in obstructive airway disease after allogeneic stem cell transplantation. Bone Marrow Transplant 2007;39:547–53.
29. Bashoura L, Gupta S, Jain A, et al. Inhaled corticosteroids stabilize constrictive bronchiolitis after hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;41:63–7.
30. Bergeron A, Chevret S, Granata A, et al. Effect of azithromycin on airflow decline-free survival after allogeneic hematopoietic stem cell transplant: the ALLOZITHRO randomized clinical trial. JAMA 2017;318:557–66.
31. Gerhardt SG, McDyer JF, Girgis RE, et al. Maintenance azithromycin therapy for bronchiolitis obliterans syndrome: results of a pilot study. Am J Respir Crit Care Med 2003;168:121–5.
32. Khalid M, Al Saghir A, Saleemi S, et al. Azithromycin in bronchiolitis obliterans complicating bone marrow transplantation: a preliminary study. Eur Respir J 2005;25:490–3.
33. Maimon N, Lipton JH, Chan CK, Marras TK. Macrolides in the treatment of bronchiolitis obliterans in allograft recipients. Bone Marrow Transplant 2009;44:69–73.
34. Lam DC, Lam B, Wong MK, et al. Effects of azithromycin in bronchiolitis obliterans syndrome after hematopoietic SCT--a randomized double-blinded placebo-controlled study. Bone Marrow Transplant 2011;46:1551–6.
35. Yadav H, Peters SG, Keogh KA, et al. Azithromycin for the treatment of obliterative bronchiolitis after hematopoietic stem cell transplantation: a systematic review and meta-analysis. Biol Blood Marrow Transplant 2016;22:2264–9.
36. Vos R, Vanaudenaerde BM, Verleden SE, et al. A randomised controlled trial of azithromycin to prevent chronic rejection after lung transplantation. Eur Respir J 2011;37:164–72.
37. Svanstrom H, Pasternak B, Hviid A. Use of azithromycin and death from cardiovascular causes. N Engl J Med 2013;368:1704–12.
38. Albert RK, Connett J, Bailey WC, et al. Azithromycin for prevention of exacerbations of COPD. N Engl J Med 2011;365:689–98.
39. Tran J, Norder EE, Diaz PT, et al. Pulmonary rehabilitation for bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2012;18:1250–4.
40. Lucid CE, Savani BN, Engelhardt BG, et al. Extracorporeal photopheresis in patients with refractory bronchiolitis obliterans developing after allo-SCT. Bone Marrow Transplant 2011;46:426–9.
41. Hostettler KE, Halter JP, Gerull S, et al. Calcineurin inhibitors in bronchiolitis obliterans syndrome following stem cell transplantation. Eur Respir J 2014;43:221–32.
42. Holm AM, Riise GC, Brinch L, et al. Lung transplantation for bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation: unresolved questions. Transplantation 2013;96:e21–22.
43. Cheng GS, Edelman JD, Madtes DK, et al. Outcomes of lung transplantation after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20:1169–75.
44. Okumura H, Ohtake S, Ontachi Y, et al. Living-donor lobar lung transplantation for broncho-bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation: does bronchiolitis obliterans recur in transplanted lungs? Int J Hematol 2007;86:369–73.
45. Olivieri A, Cimminiello M, Corradini P, et al. Long-term outcome and prospective validation of NIH response criteria in 39 patients receiving imatinib for steroid-refractory chronic GVHD. Blood 2013;122:4111–8.
46. Rahmanian S, Wood KL. Bronchiolitis obliterans and the risk of pneumothorax after transbronchial biopsy. Respiratory Medicine CME 2010;3:87–9.
47. Sakai R, Kanamori H, Nakaseko C, et al. Air-leak syndrome following allo-SCT in adult patients: report from the Kanto Study Group for Cell Therapy in Japan. Bone Marrow Transplant 2011;46:379–84.
48. Visscher DW, Myers JL. Histologic spectrum of idiopathic interstitial pneumonias. Proc Am Thorac Soc 2006;3:322–9.
49. Cordier JF. Cryptogenic organising pneumonia. Eur Respir J 2006;28:422–46.
50. Nishio N, Yagasaki H, Takahashi Y, et al. Late-onset non-infectious pulmonary complications following allogeneic hematopoietic stem cell transplantation in children. Bone Marrow Transplant 2009;44:303–8.
51. Ueda K, Watadani T, Maeda E, et al. Outcome and treatment of late-onset noninfectious pulmonary complications after allogeneic haematopoietic SCT. Bone Marrow Transplant 2010;45:1719–27.
52. Schlemmer F, Chevret S, Lorillon G, et al. Late-onset noninfectious interstitial lung disease after allogeneic hematopoietic stem cell transplantation. Respir Med 2014;108:1525–33.
53. Palmas A, Tefferi A, Myers JL, et al. Late-onset noninfectious pulmonary complications after allogeneic bone marrow transplantation. Br J Haematol 1998;100:680–7.
54. Sakaida E, Nakaseko C, Harima A, et al. Late-onset noninfectious pulmonary complications after allogeneic stem cell transplantation are significantly associated with chronic graft-versus-host disease and with the graft-versus-leukemia effect. Blood 2003;102:4236–42.
55. Solh M, Arat M, Cao Q, et al. Late-onset noninfectious pulmonary complications in adult allogeneic hematopoietic cell transplant recipients. Transplantation 2011;91:798–803.
56. Dandoy CE, Hirsch R, Chima R, et al. Pulmonary hypertension after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013;19:1546–56.
57. Bunte MC, Patnaik MM, Pritzker MR, Burns LJ. Pulmonary veno-occlusive disease following hematopoietic stem cell transplantation: a rare model of endothelial dysfunction. Bone Marrow Transplant 2008;41:677–86.
58. Troussard X, Bernaudin JF, Cordonnier C, et al. Pulmonary veno-occlusive disease after bone marrow transplantation. Thorax 1984;39:956–7.
59. Lahzami S, Schoeffel RE, Pechey V, et al. Small airways function declines after allogeneic haematopoietic stem cell transplantation. Eur Respir J 2011;38:1180–8.
60. Jain NA, Pophali PA, Klotz JK, et al. Repair of impaired pulmonary function is possible in very-long-term allogeneic stem cell transplantation survivors. Biol Blood Marrow Transplant 2014;20:209–13.
61. Barisione G, Bacigalupo A, Crimi E, et al. Changes in lung volumes and airway responsiveness following haematopoietic stem cell transplantation. Eur Respir J 2008;32:1576–82.
62. Kovalszki A, Schumaker GL, Klein A, et al. Reduced respiratory and skeletal muscle strength in survivors of sibling or unrelated donor hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;41:965–9.
63. Mathiesen S, Uhlving HH, Buchvald F, et al. Aerobic exercise capacity at long-term follow-up after paediatric allogeneic haematopoietic SCT. Bone Marrow Transplant 2014;49:1393–9.
Hematopoietic stem cell transplantation (HSCT) is increasingly being used to treat hematologic malignancies as well as nonmalignant diseases and solid tumors. Over the past 2 decades overall survival following transplant and transplant-related mortality have improved.1 With this increased survival, there is a need to focus on late complications after transplantation. Pulmonary complications are a common but sometimes underrecognized cause of late morbidity and mortality in HSCT patients. This article, the second of 2 articles on post-HSCT pulmonary complications, reviews late-onset complications, with a focus on the evaluation and treatment of bronchiolitis obliterans syndrome (BOS), one of the most common and serious late pulmonary complications in HSCT patients. The first article reviewed the management of early-onset pulmonary complications and included a basic overview of stem cell transplantation, discussion of factors associated with pulmonary complications, and a review of methods for assessing pretransplant risk for pulmonary complications in patients undergoing HSCT.2
Case Presentation
A 40-year-old white woman with a history of acute myeloid leukemia status post peripheral blood stem cell transplant presents with dyspnea on exertion, which she states started about 1 month ago and now is limiting her with even 1 flight of stairs. She also complains of mild dry cough and a 4- to 5-lb weight loss over the past 1 to 2 months. She has an occasional runny nose, but denies gastroesophageal reflux, fevers, chills, or night sweats. She has a history of matched related sibling donor transplant with busulfan and cyclophosphamide conditioning 1 year prior to presentation. She has had significant graft-versus-host disease (GVHD), affecting the liver, gastrointestinal tract, skin, and eyes.
On physical examination, heart rate is 110 beats/min, respiratory rate is 16 breaths/min, blood pressure is 92/58 mm Hg, and the patient is afebrile. Eye exam reveals scleral injection, mouth shows dry mucous membranes with a few white plaques, and the skin has chronic changes with a rash over both arms. Cardiac exam reveals tachycardia but regular rhythm and there are no murmurs, rubs, or gallops. Lungs are clear bilaterally and abdomen shows no organomegaly.
Laboratory exam shows a white blood cell count of 7800 cells/μL, hemoglobin level of 12.4 g/dL, and platelet count of 186 × 103/μL. Liver enzymes are mildly elevated. Chest radiograph shows clear lung fields bilaterally.
- What is the differential in this patient with dyspnea 1 year after transplantation?
Late pulmonary complications are generally accepted as those occurring more than 100 days post transplant. This period of time is characterized by chronic GVHD and impaired cellular and humoral immunity. Results of longitudinal studies of infections in adult HSCT patients suggest that special attention should be paid to allogeneic HSCT recipients for post-engraftment infectious pulmonary complications.3 Encapsulated bacteria such as Haemophilus influenzae and Streptococcus pneumoniae are the most frequent bacterial organisms causing late infectious pulmonary complications. Nontuberculous mycobacteria and Nocardia should also be considered. Depending upon geographic location, social and occupational risk factors, and prevalence, tuberculosis should also enter the differential.
There are many noninfectious late-onset pulmonary complications after HSCT. Unfortunately, the literature has divided pulmonary complications after HSCT using a range of criteria and classifications based upon timing, predominant pulmonary function test (PFT) findings, and etiology. These include early versus late, obstructive versus restrictive, and infectious versus noninfectious, which makes a comprehensive literature review of late pulmonary complications difficult. The most common noninfectious late-onset complications are bronchiolitis obliterans, cryptogenic organizing pneumonia (previously referred to as bronchiolitis obliterans organizing pneumonia, or BOOP), and interstitial pneumonia. Other rarely reported complications include eosinophilic pneumonia, pulmonary alveolar proteinosis, air leak syndrome, and pulmonary hypertension.
Case Continued
Because the patient does not have symptoms of infection, PFTs are obtained. Pretransplant PFTs and current PFTs are shown in Table 1.
- What is the diagnosis in this case?
Bronchiolitis Obliterans
BOS is one of the most common and most serious late-onset pulmonary diseases after allogeneic transplantation. It is considered the pulmonary form of chronic GVHD. BOS was first described in 1982 in patients with chronic GVHD after bone marrow transplantation.4 Many differing definitions of bronchiolitis obliterans have been described in the literature. A recent review of the topic cites 10 different published sets of criteria for the diagnosis of bronchiolitis obliterans.5 Traditionally, bronchiolitis obliterans was thought to occur in 2% to 8% of patients undergoing allogeneic HSCT, but these findings were from older studies that used a diagnosis based on very specific pathology findings. When more liberal diagnostic criteria are used, the incidence may be as high as 26% of allogeneic HSCT patients.6
Bronchiolitis obliterans is a progressive lung disease characterized by narrowing of the terminal airways and obliteration of the terminal bronchi. Pathology may show constrictive bronchiolitis but can also show lymphocytic bronchiolitis, which may be associated with a better outcome.7 As noted, bronchiolitis obliterans has traditionally been considered a pathologic diagnosis. Current diagnostic criteria have evolved based upon the difficulty in obtaining this diagnosis through transbronchial biopsy given the patchy nature of the disease.8 The gold standard of open lung biopsy is seldom pursued in the post-HSCT population as the procedure continues to carry a worrisome risk-benefit profile.
The 2005 National Institutes of Health (NIH) consensus development project on criteria for clinical trials in chronic GVHD developed a clinical strategy for diagnosing BOS using the following criteria: absence of active infection, decreased forced expiratory volume in 1 second (FEV1) < 75%, FEV1/forced vital capacity (FVC) ratio of < 70%, and evidence of air trapping on high-resolution computed tomography (HRCT) or PFTs (residual volume > 120%). These diagnostic criteria were applied to a small series of patients with clinically identified bronchiolitis obliterans or biopsy-proven bronchiolitis obliterans. Only 18% of these patients met the requirements for the NIH consensus definition.5 A 2011 study that applied the NIH criteria found an overall prevalence of 5.5% among all transplant recipients but a prevalence of 14% in patients with GVHD.9 In 2014, the NIH consensus development group updated their recommendations. The new criteria for diagnosis of BOS require the presence of airflow obstruction (FEV1/FVC < 70% or 5th percentile of predicted), FEV1 < 75% predicted with a ≥ 10% decline in fewer than 2 years, absence of infection, and presence of air trapping (by expiratory computed tomography [CT] scan or PFT with residual volume >120% predicted) (Table 2).
Some issues must be considered when determining airflow obstruction. The 2005 NIH working group recommends using Crapo as the reference set,11 but the National Health and Nutrition Examination Survey (NHANES) III reference values are the preferred reference set at this time12 and should be used in the United States. A recent article showed that the NHANES values were superior to older reference sets (however, they did not use Crapo as the comparison), although this study used the lower limit of normal as compared with the fixed 70% ratio.13 The 2014 NIH consensus group does not recommend a specific reference set and recognizes an FEV1/FVC ratio of 70% or less than the lower limit of normal as the cutoff value for airflow obstruction.10
Another issue in PFT interpretation is the finding of a decrease in FEV1 and FVC and normal total lung capacity, which is termed a nonspecific pattern. This pattern has been reported to occur in 9% of all PFTs and usually is associated with obstructive lung disease or obesity.14 A 2013 study described the nonspecific pattern as a BOS subgroup occurring in up to 31% of bronchiolitis obliterans patients.15
- What are the radiographic findings of BOS?
Chest radiograph is often normal in BOS. As discussed, air trapping can be documented using HRCT, according to the NIH clinical definition of bronchiolitis obliterans.16 A study that explored findings and trends seen on HRCT in HSCT patients with BOS found that the syndrome in these patients is characterized by central airway dilatation.17 Expiratory airway trapping on HRCT is the main finding, and this is best demonstrated on HRCT during inspiratory and expiratory phases.18 Other findings are bronchial wall thickening, parenchymal hypoattenuation, bronchiectasis, and centrilobular nodules.19
Galbán and colleagues developed a new technique called parametric response mapping that uses CT scanners to quantify normal parenchyma, functional small airway disease, emphysema, and parenchymal disease as relative lung volumes.20 This technique can detect airflow obstruction and small airway disease and was found to be a good method for detecting BOS after HSCT. In their study of parametric response mapping, the authors found that functional small airway disease affecting 28% or more of the total lung was highly indicative of bronchiolitis obliterans.20
- What therapies are used to treat BOS?
Traditionally, BOS has been treated with systemic immunosuppression. The recommended treatment had been systemic steroids at approximately 1 mg/ kg. However, it is increasingly recognized that BOS responds poorly to systemic steroids, and systemic steroids may actually be harmful and associated with increased mortality.15,21 The chronic GVHD recommendations from 2005 recommend ancillary therapy with inhaled corticosteroids and pulmonary rehabilitation.11 The updated 2011 German consensus statement lays out a clear management strategy for mild and moderate-severe disease with monitoring recommendations.22 The 2014 NIH chronic GVHD working group recommends fluticasone, azithromycin, and montelukast (ie, the FAM protocol) for treating BOS.23 FAM therapy in BOS may help lower the systemic steroid dose.24,25 Montelukast is not considered a treatment mainstay for BOS after lung transplant, but there is a study showing possible benefit in chronic GVHD.26 An evaluation of the natural history of a cohort of BOS patients treated with FAM therapy showed a rapid decline of FEV1 in the 6 months prior to diagnosis and treatment of BOS and subsequent stabilization following diagnosis and treatment.27 The benefit of high-dose inhaled corticosteroids or the combination of inhaled corticosteroids and long-acting beta-agonists has been demonstrated in small studies, which showed that these agents stabilized FEV1 and avoided the untoward side effects of systemic corticosteroids.28–30
Macrolide antibiotics have been explored as a treatment for BOS post HSCT because pilot studies suggested that azithromycin improved or stabilized FEV1 in patients with BOS after lung transplant or HSCT.31–33 Other studies of azithromycin have not shown benefit in the HSCT population after 3 months of therapy.34 A recent meta-analysis could neither support or refute the benefit of azithromycin for BOS after HSCT.35 In the lung transplant population, a study showed that patients who were started on azithromycin after transplant and continued on it 3 times a week had improved FEV1; these patients also had a reduced rate of BOS and improved overall and BOS-free survival 2 years after transplant.36 However, these benefits of azithromycin have not been observed in patients after HSCT. In fact, the ALLOZITHRO trial was stopped early because prophylactic azithromycin started at the time of the conditioning regimen with HSCT was associated with increased hematologic disease relapse, a decrease in airflow-decline-free survival, and reduced 2-year survival.30
Azithromycin is believed to exert an effect by its anti-inflammatory properties and perhaps by decreasing lung neutrophilia (it may be most beneficial in the subset of patients with high neutrophilia on bronchoalveolar lavage [BAL]).30 Adverse effects of chronic azithromycin include QT prolongation, cardiac arrhythmia, hearing loss, and antibiotic-resistant organism colonization.37,38
Other therapies include pulmonary rehabilitation, which may improve health-related quality of life and 6-minute walk distance,39 extracorporeal photopheresis,40 immunosuppression with calcineurin inhibitors or mycophenolate mofetil,21,41 and lung transplantation.42–44 A study with imatinib for the treatment of lung disease in steroid-refractory GVHD has shown promising results, but further validation with larger clinical trials is required.45
Case Continued
The patient is diagnosed with BOS and is treated for several months with prednisone 40 mg/day weaned over 3 months. She is started on inhaled corticosteroids, a proton pump inhibitor, and azithromycin 3 times per week, but she has a progressive decline in FEV1. She starts pulmonary rehabilitation but continues to functionally decline. Over the next year she develops bilateral pneumothoraces and bilateral cavitary nodules (Figure 1).
- What is causing this decline and the radiographic abnormalities?
Spontaneous air leak syndrome has been described in a little more than 1% of patients undergoing HSCT and has included pneumothorax and mediastinal and subcutaneous emphysema.46 It appears that air leak syndrome is more likely to occur in patients with chronic GVHD.47 The association between chronic GVHD and air leak syndrome could explain this patient’s recurrent pneumothoraces. The recurrent cavitary nodules are suspicious for infectious etiologies such as nontuberculous mycobacteria, tuberculosis, and fungal infections.
Case Continued
During an episode of pneumothorax, the patient undergoes chest tube placement, pleurodesis, and lung biopsy. Pathology reveals bronchiolitis obliterans as well as organizing pneumonia (Figure 2). No organisms are seen on acid-fast bacilli or GMS stains.
- What are the other late-onset noninfectious pulmonary complications?
Definitions of other late noninfectious pulmonary complications following HSCT are shown in Table 3.
Interstitial pneumonias may represent COP or may be idiopathic pneumonia syndrome with a later onset or a nonspecific interstitial pneumonia. This syndrome is poorly defined, with a number of differing definitions of the syndrome published in the literature.50–55
A rare pulmonary complication after HSCT is pulmonary veno-occlusive disease (PVOD). Pulmonary hypertension has been reported after HSCT,56 but PVOD is a subset of pulmonary hypertension. It is associated with pleural effusions and volume overload on chest radiography.57,58 It may present early or late after transplant and is poorly understood.
Besides obstructive and restrictive PFT abnormalities, changes in small airway function59 after transplant and loss in diffusing capacity of the lungs for carbon monoxide (D
Case Conclusion
The patient’s lung function continues to worsen, but no infectious etiologies are discovered. Ultimately, she dies of respiratory failure caused by progressive bronchiolitis obliterans.
Conclusion
Late pulmonary complications occur frequently in patients who have undergone HSCT. These complications can be classified as infectious versus noninfectious etiologies. Late-onset complications are more common in allogeneic transplantations because they are associated with chronic GVHD. These complications can be manifestations of pulmonary GHVD or can be infectious complications associated with prolonged immunosuppression. Appropriate monitoring for the development of BOS is essential. Early and aggressive treatment of respiratory infections and diagnostic bronchoscopy with BAL can help elucidate most infectious causes. Still, diagnostic challenges remain and multiple causes of respiratory deterioration can be present concurrently in the post-HSCT patient. Steroid therapy remains the mainstay treatment for most noninfectious pulmonary complications and should be strongly considered once infection is effectively ruled out.
Hematopoietic stem cell transplantation (HSCT) is increasingly being used to treat hematologic malignancies as well as nonmalignant diseases and solid tumors. Over the past 2 decades overall survival following transplant and transplant-related mortality have improved.1 With this increased survival, there is a need to focus on late complications after transplantation. Pulmonary complications are a common but sometimes underrecognized cause of late morbidity and mortality in HSCT patients. This article, the second of 2 articles on post-HSCT pulmonary complications, reviews late-onset complications, with a focus on the evaluation and treatment of bronchiolitis obliterans syndrome (BOS), one of the most common and serious late pulmonary complications in HSCT patients. The first article reviewed the management of early-onset pulmonary complications and included a basic overview of stem cell transplantation, discussion of factors associated with pulmonary complications, and a review of methods for assessing pretransplant risk for pulmonary complications in patients undergoing HSCT.2
Case Presentation
A 40-year-old white woman with a history of acute myeloid leukemia status post peripheral blood stem cell transplant presents with dyspnea on exertion, which she states started about 1 month ago and now is limiting her with even 1 flight of stairs. She also complains of mild dry cough and a 4- to 5-lb weight loss over the past 1 to 2 months. She has an occasional runny nose, but denies gastroesophageal reflux, fevers, chills, or night sweats. She has a history of matched related sibling donor transplant with busulfan and cyclophosphamide conditioning 1 year prior to presentation. She has had significant graft-versus-host disease (GVHD), affecting the liver, gastrointestinal tract, skin, and eyes.
On physical examination, heart rate is 110 beats/min, respiratory rate is 16 breaths/min, blood pressure is 92/58 mm Hg, and the patient is afebrile. Eye exam reveals scleral injection, mouth shows dry mucous membranes with a few white plaques, and the skin has chronic changes with a rash over both arms. Cardiac exam reveals tachycardia but regular rhythm and there are no murmurs, rubs, or gallops. Lungs are clear bilaterally and abdomen shows no organomegaly.
Laboratory exam shows a white blood cell count of 7800 cells/μL, hemoglobin level of 12.4 g/dL, and platelet count of 186 × 103/μL. Liver enzymes are mildly elevated. Chest radiograph shows clear lung fields bilaterally.
- What is the differential in this patient with dyspnea 1 year after transplantation?
Late pulmonary complications are generally accepted as those occurring more than 100 days post transplant. This period of time is characterized by chronic GVHD and impaired cellular and humoral immunity. Results of longitudinal studies of infections in adult HSCT patients suggest that special attention should be paid to allogeneic HSCT recipients for post-engraftment infectious pulmonary complications.3 Encapsulated bacteria such as Haemophilus influenzae and Streptococcus pneumoniae are the most frequent bacterial organisms causing late infectious pulmonary complications. Nontuberculous mycobacteria and Nocardia should also be considered. Depending upon geographic location, social and occupational risk factors, and prevalence, tuberculosis should also enter the differential.
There are many noninfectious late-onset pulmonary complications after HSCT. Unfortunately, the literature has divided pulmonary complications after HSCT using a range of criteria and classifications based upon timing, predominant pulmonary function test (PFT) findings, and etiology. These include early versus late, obstructive versus restrictive, and infectious versus noninfectious, which makes a comprehensive literature review of late pulmonary complications difficult. The most common noninfectious late-onset complications are bronchiolitis obliterans, cryptogenic organizing pneumonia (previously referred to as bronchiolitis obliterans organizing pneumonia, or BOOP), and interstitial pneumonia. Other rarely reported complications include eosinophilic pneumonia, pulmonary alveolar proteinosis, air leak syndrome, and pulmonary hypertension.
Case Continued
Because the patient does not have symptoms of infection, PFTs are obtained. Pretransplant PFTs and current PFTs are shown in Table 1.
- What is the diagnosis in this case?
Bronchiolitis Obliterans
BOS is one of the most common and most serious late-onset pulmonary diseases after allogeneic transplantation. It is considered the pulmonary form of chronic GVHD. BOS was first described in 1982 in patients with chronic GVHD after bone marrow transplantation.4 Many differing definitions of bronchiolitis obliterans have been described in the literature. A recent review of the topic cites 10 different published sets of criteria for the diagnosis of bronchiolitis obliterans.5 Traditionally, bronchiolitis obliterans was thought to occur in 2% to 8% of patients undergoing allogeneic HSCT, but these findings were from older studies that used a diagnosis based on very specific pathology findings. When more liberal diagnostic criteria are used, the incidence may be as high as 26% of allogeneic HSCT patients.6
Bronchiolitis obliterans is a progressive lung disease characterized by narrowing of the terminal airways and obliteration of the terminal bronchi. Pathology may show constrictive bronchiolitis but can also show lymphocytic bronchiolitis, which may be associated with a better outcome.7 As noted, bronchiolitis obliterans has traditionally been considered a pathologic diagnosis. Current diagnostic criteria have evolved based upon the difficulty in obtaining this diagnosis through transbronchial biopsy given the patchy nature of the disease.8 The gold standard of open lung biopsy is seldom pursued in the post-HSCT population as the procedure continues to carry a worrisome risk-benefit profile.
The 2005 National Institutes of Health (NIH) consensus development project on criteria for clinical trials in chronic GVHD developed a clinical strategy for diagnosing BOS using the following criteria: absence of active infection, decreased forced expiratory volume in 1 second (FEV1) < 75%, FEV1/forced vital capacity (FVC) ratio of < 70%, and evidence of air trapping on high-resolution computed tomography (HRCT) or PFTs (residual volume > 120%). These diagnostic criteria were applied to a small series of patients with clinically identified bronchiolitis obliterans or biopsy-proven bronchiolitis obliterans. Only 18% of these patients met the requirements for the NIH consensus definition.5 A 2011 study that applied the NIH criteria found an overall prevalence of 5.5% among all transplant recipients but a prevalence of 14% in patients with GVHD.9 In 2014, the NIH consensus development group updated their recommendations. The new criteria for diagnosis of BOS require the presence of airflow obstruction (FEV1/FVC < 70% or 5th percentile of predicted), FEV1 < 75% predicted with a ≥ 10% decline in fewer than 2 years, absence of infection, and presence of air trapping (by expiratory computed tomography [CT] scan or PFT with residual volume >120% predicted) (Table 2).
Some issues must be considered when determining airflow obstruction. The 2005 NIH working group recommends using Crapo as the reference set,11 but the National Health and Nutrition Examination Survey (NHANES) III reference values are the preferred reference set at this time12 and should be used in the United States. A recent article showed that the NHANES values were superior to older reference sets (however, they did not use Crapo as the comparison), although this study used the lower limit of normal as compared with the fixed 70% ratio.13 The 2014 NIH consensus group does not recommend a specific reference set and recognizes an FEV1/FVC ratio of 70% or less than the lower limit of normal as the cutoff value for airflow obstruction.10
Another issue in PFT interpretation is the finding of a decrease in FEV1 and FVC and normal total lung capacity, which is termed a nonspecific pattern. This pattern has been reported to occur in 9% of all PFTs and usually is associated with obstructive lung disease or obesity.14 A 2013 study described the nonspecific pattern as a BOS subgroup occurring in up to 31% of bronchiolitis obliterans patients.15
- What are the radiographic findings of BOS?
Chest radiograph is often normal in BOS. As discussed, air trapping can be documented using HRCT, according to the NIH clinical definition of bronchiolitis obliterans.16 A study that explored findings and trends seen on HRCT in HSCT patients with BOS found that the syndrome in these patients is characterized by central airway dilatation.17 Expiratory airway trapping on HRCT is the main finding, and this is best demonstrated on HRCT during inspiratory and expiratory phases.18 Other findings are bronchial wall thickening, parenchymal hypoattenuation, bronchiectasis, and centrilobular nodules.19
Galbán and colleagues developed a new technique called parametric response mapping that uses CT scanners to quantify normal parenchyma, functional small airway disease, emphysema, and parenchymal disease as relative lung volumes.20 This technique can detect airflow obstruction and small airway disease and was found to be a good method for detecting BOS after HSCT. In their study of parametric response mapping, the authors found that functional small airway disease affecting 28% or more of the total lung was highly indicative of bronchiolitis obliterans.20
- What therapies are used to treat BOS?
Traditionally, BOS has been treated with systemic immunosuppression. The recommended treatment had been systemic steroids at approximately 1 mg/ kg. However, it is increasingly recognized that BOS responds poorly to systemic steroids, and systemic steroids may actually be harmful and associated with increased mortality.15,21 The chronic GVHD recommendations from 2005 recommend ancillary therapy with inhaled corticosteroids and pulmonary rehabilitation.11 The updated 2011 German consensus statement lays out a clear management strategy for mild and moderate-severe disease with monitoring recommendations.22 The 2014 NIH chronic GVHD working group recommends fluticasone, azithromycin, and montelukast (ie, the FAM protocol) for treating BOS.23 FAM therapy in BOS may help lower the systemic steroid dose.24,25 Montelukast is not considered a treatment mainstay for BOS after lung transplant, but there is a study showing possible benefit in chronic GVHD.26 An evaluation of the natural history of a cohort of BOS patients treated with FAM therapy showed a rapid decline of FEV1 in the 6 months prior to diagnosis and treatment of BOS and subsequent stabilization following diagnosis and treatment.27 The benefit of high-dose inhaled corticosteroids or the combination of inhaled corticosteroids and long-acting beta-agonists has been demonstrated in small studies, which showed that these agents stabilized FEV1 and avoided the untoward side effects of systemic corticosteroids.28–30
Macrolide antibiotics have been explored as a treatment for BOS post HSCT because pilot studies suggested that azithromycin improved or stabilized FEV1 in patients with BOS after lung transplant or HSCT.31–33 Other studies of azithromycin have not shown benefit in the HSCT population after 3 months of therapy.34 A recent meta-analysis could neither support or refute the benefit of azithromycin for BOS after HSCT.35 In the lung transplant population, a study showed that patients who were started on azithromycin after transplant and continued on it 3 times a week had improved FEV1; these patients also had a reduced rate of BOS and improved overall and BOS-free survival 2 years after transplant.36 However, these benefits of azithromycin have not been observed in patients after HSCT. In fact, the ALLOZITHRO trial was stopped early because prophylactic azithromycin started at the time of the conditioning regimen with HSCT was associated with increased hematologic disease relapse, a decrease in airflow-decline-free survival, and reduced 2-year survival.30
Azithromycin is believed to exert an effect by its anti-inflammatory properties and perhaps by decreasing lung neutrophilia (it may be most beneficial in the subset of patients with high neutrophilia on bronchoalveolar lavage [BAL]).30 Adverse effects of chronic azithromycin include QT prolongation, cardiac arrhythmia, hearing loss, and antibiotic-resistant organism colonization.37,38
Other therapies include pulmonary rehabilitation, which may improve health-related quality of life and 6-minute walk distance,39 extracorporeal photopheresis,40 immunosuppression with calcineurin inhibitors or mycophenolate mofetil,21,41 and lung transplantation.42–44 A study with imatinib for the treatment of lung disease in steroid-refractory GVHD has shown promising results, but further validation with larger clinical trials is required.45
Case Continued
The patient is diagnosed with BOS and is treated for several months with prednisone 40 mg/day weaned over 3 months. She is started on inhaled corticosteroids, a proton pump inhibitor, and azithromycin 3 times per week, but she has a progressive decline in FEV1. She starts pulmonary rehabilitation but continues to functionally decline. Over the next year she develops bilateral pneumothoraces and bilateral cavitary nodules (Figure 1).
- What is causing this decline and the radiographic abnormalities?
Spontaneous air leak syndrome has been described in a little more than 1% of patients undergoing HSCT and has included pneumothorax and mediastinal and subcutaneous emphysema.46 It appears that air leak syndrome is more likely to occur in patients with chronic GVHD.47 The association between chronic GVHD and air leak syndrome could explain this patient’s recurrent pneumothoraces. The recurrent cavitary nodules are suspicious for infectious etiologies such as nontuberculous mycobacteria, tuberculosis, and fungal infections.
Case Continued
During an episode of pneumothorax, the patient undergoes chest tube placement, pleurodesis, and lung biopsy. Pathology reveals bronchiolitis obliterans as well as organizing pneumonia (Figure 2). No organisms are seen on acid-fast bacilli or GMS stains.
- What are the other late-onset noninfectious pulmonary complications?
Definitions of other late noninfectious pulmonary complications following HSCT are shown in Table 3.
Interstitial pneumonias may represent COP or may be idiopathic pneumonia syndrome with a later onset or a nonspecific interstitial pneumonia. This syndrome is poorly defined, with a number of differing definitions of the syndrome published in the literature.50–55
A rare pulmonary complication after HSCT is pulmonary veno-occlusive disease (PVOD). Pulmonary hypertension has been reported after HSCT,56 but PVOD is a subset of pulmonary hypertension. It is associated with pleural effusions and volume overload on chest radiography.57,58 It may present early or late after transplant and is poorly understood.
Besides obstructive and restrictive PFT abnormalities, changes in small airway function59 after transplant and loss in diffusing capacity of the lungs for carbon monoxide (D
Case Conclusion
The patient’s lung function continues to worsen, but no infectious etiologies are discovered. Ultimately, she dies of respiratory failure caused by progressive bronchiolitis obliterans.
Conclusion
Late pulmonary complications occur frequently in patients who have undergone HSCT. These complications can be classified as infectious versus noninfectious etiologies. Late-onset complications are more common in allogeneic transplantations because they are associated with chronic GVHD. These complications can be manifestations of pulmonary GHVD or can be infectious complications associated with prolonged immunosuppression. Appropriate monitoring for the development of BOS is essential. Early and aggressive treatment of respiratory infections and diagnostic bronchoscopy with BAL can help elucidate most infectious causes. Still, diagnostic challenges remain and multiple causes of respiratory deterioration can be present concurrently in the post-HSCT patient. Steroid therapy remains the mainstay treatment for most noninfectious pulmonary complications and should be strongly considered once infection is effectively ruled out.
1. Remberger M, Ackefors M, Berglund S, et al. Improved survival after allogeneic hematopoietic stem cell transplantation in recent years. A single-center study. Biol Blood Marrow Transplant 2011;17:1688–97.
2. Wood KL, Esguerra VG. Management of late pulmonary complications after hematopoietic stem cell transplantation. Hosp Phys Hematology-Oncology Board Review Manual 2018;13(1):36–48.
3. Ninin E, Milpied N, Moreau P, et al. Longitudinal study of bacterial, viral, and fungal infections in adult recipients of bone marrow transplants. Clin Infect Dis 2001;33:41–7.
4. Roca J, Granena A, Rodriguez-Roisin R, et al. Fatal airway disease in an adult with chronic graft-versus-host disease. Thorax 1982;37:77–8.
5. Williams KM, Chien JW, Gladwin MT, Pavletic SZ. Bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. JAMA 2009;302:306–14.
6. Chien JW, Martin PJ, Gooley TA, et al. Airflow obstruction after myeloablative allogeneic hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2003;168:208–14.
7. Holbro A, Lehmann T, Girsberger S, et al. Lung histology predicts outcome of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013;19:973–80.
8. Chamberlain D, Maurer J, Chaparro C, Idolor L. Evaluation of transbronchial lung biopsy specimens in the diagnosis of bronchiolitis obliterans after lung transplantation. J Heart Lung Transplant 1994;13:963–71.
9. Au BK, Au MA, Chien JW. Bronchiolitis obliterans syndrome epidemiology after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2011;17:1072–8.
10. Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 2015;21:389–401.
11. Couriel D, Carpenter PA, Cutler C, et al. Ancillary therapy and supportive care of chronic graft-versus-host disease: national institutes of health consensus development project on criteria for clinical trials in chronic Graft-versus-host disease: V. Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant 2006;12:375–96.
12. Pellegrino R, Viegi G, Brusasco V, et al. Interpretative strategies for lung function tests. Eur Respir J 2005;26:948–68.
13. Williams KM, Hnatiuk O, Mitchell SA, et al. NHANES III equations enhance early detection and mortality prediction of bronchiolitis obliterans syndrome after hematopoietic SCT. Bone Marrow Transplant 2014;49:561–6.
14. Hyatt RE, Cowl CT, Bjoraker JA, Scanlon PD. Conditions associated with an abnormal nonspecific pattern of pulmonary function tests. Chest 2009;135:419–24.
15. Bergeron A, Godet C, Chevret S, et al. Bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: phenotypes and prognosis. Bone Marrow Transplant 2013;48:819–24.
16. Filipovich AH, Weisdorf D, Pavletic S, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant 2005;11:945–56.
17. Gazourian L, Coronata AM, Rogers AJ, et al. Airway dilation in bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. Respir Med 2013;107:276–83.
18. Gunn ML, Godwin JD, Kanne JP, et al. High-resolution CT findings of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. J Thorac Imaging 2008;23:244–50.
19. Sargent MA, Cairns RA, Murdoch MJ, et al. Obstructive lung disease in children after allogeneic bone marrow transplantation: evaluation with high-resolution CT. AJR Am J Roentgenol 1995;164:693–6.
20. Galban CJ, Boes JL, Bule M, et al. Parametric response mapping as an indicator of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20:1592–8.
21. Meyer KC, Raghu G, Verleden GM, et al. An international ISHLT/ATS/ERS clinical practice guideline: diagnosis and management of bronchiolitis obliterans syndrome. Eur Respir J 2014;44:1479–1503.
22. Hildebrandt GC, Fazekas T, Lawitschka A, et al. Diagnosis and treatment of pulmonary chronic GVHD: report from the consensus conference on clinical practice in chronic GVHD. Bone Marrow Transplant 2011;46:1283–95.
23. Carpenter PA, Kitko CL, Elad S, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: V. The 2014 Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant 2015;21:1167–87.
24. Norman BC, Jacobsohn DA, Williams KM, et al. Fluticasone, azithromycin and montelukast therapy in reducing corticosteroid exposure in bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: a case series of eight patients. Bone Marrow Transplant 2011;46:1369–73.
25. Williams KM, Cheng GS, Pusic I, et al. Fluticasone, azithromycin, and montelukast treatment for new-onset bronchiolitis obliterans syndrome after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2016;22:710–6.
26. Or R, Gesundheit B, Resnick I, et al. Sparing effect by montelukast treatment for chronic graft versus host disease: a pilot study. Transplantation 2007;83:577–81.
27. Cheng GS, Storer B, Chien JW, et al. Lung function trajectory in bronchiolitis obliterans syndrome after allogeneic hematopoietic cell transplant. Ann Am Thorac Soc 2016;13:1932–9.
28. Bergeron A, Belle A, Chevret S, et al. Combined inhaled steroids and bronchodilatators in obstructive airway disease after allogeneic stem cell transplantation. Bone Marrow Transplant 2007;39:547–53.
29. Bashoura L, Gupta S, Jain A, et al. Inhaled corticosteroids stabilize constrictive bronchiolitis after hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;41:63–7.
30. Bergeron A, Chevret S, Granata A, et al. Effect of azithromycin on airflow decline-free survival after allogeneic hematopoietic stem cell transplant: the ALLOZITHRO randomized clinical trial. JAMA 2017;318:557–66.
31. Gerhardt SG, McDyer JF, Girgis RE, et al. Maintenance azithromycin therapy for bronchiolitis obliterans syndrome: results of a pilot study. Am J Respir Crit Care Med 2003;168:121–5.
32. Khalid M, Al Saghir A, Saleemi S, et al. Azithromycin in bronchiolitis obliterans complicating bone marrow transplantation: a preliminary study. Eur Respir J 2005;25:490–3.
33. Maimon N, Lipton JH, Chan CK, Marras TK. Macrolides in the treatment of bronchiolitis obliterans in allograft recipients. Bone Marrow Transplant 2009;44:69–73.
34. Lam DC, Lam B, Wong MK, et al. Effects of azithromycin in bronchiolitis obliterans syndrome after hematopoietic SCT--a randomized double-blinded placebo-controlled study. Bone Marrow Transplant 2011;46:1551–6.
35. Yadav H, Peters SG, Keogh KA, et al. Azithromycin for the treatment of obliterative bronchiolitis after hematopoietic stem cell transplantation: a systematic review and meta-analysis. Biol Blood Marrow Transplant 2016;22:2264–9.
36. Vos R, Vanaudenaerde BM, Verleden SE, et al. A randomised controlled trial of azithromycin to prevent chronic rejection after lung transplantation. Eur Respir J 2011;37:164–72.
37. Svanstrom H, Pasternak B, Hviid A. Use of azithromycin and death from cardiovascular causes. N Engl J Med 2013;368:1704–12.
38. Albert RK, Connett J, Bailey WC, et al. Azithromycin for prevention of exacerbations of COPD. N Engl J Med 2011;365:689–98.
39. Tran J, Norder EE, Diaz PT, et al. Pulmonary rehabilitation for bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2012;18:1250–4.
40. Lucid CE, Savani BN, Engelhardt BG, et al. Extracorporeal photopheresis in patients with refractory bronchiolitis obliterans developing after allo-SCT. Bone Marrow Transplant 2011;46:426–9.
41. Hostettler KE, Halter JP, Gerull S, et al. Calcineurin inhibitors in bronchiolitis obliterans syndrome following stem cell transplantation. Eur Respir J 2014;43:221–32.
42. Holm AM, Riise GC, Brinch L, et al. Lung transplantation for bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation: unresolved questions. Transplantation 2013;96:e21–22.
43. Cheng GS, Edelman JD, Madtes DK, et al. Outcomes of lung transplantation after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20:1169–75.
44. Okumura H, Ohtake S, Ontachi Y, et al. Living-donor lobar lung transplantation for broncho-bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation: does bronchiolitis obliterans recur in transplanted lungs? Int J Hematol 2007;86:369–73.
45. Olivieri A, Cimminiello M, Corradini P, et al. Long-term outcome and prospective validation of NIH response criteria in 39 patients receiving imatinib for steroid-refractory chronic GVHD. Blood 2013;122:4111–8.
46. Rahmanian S, Wood KL. Bronchiolitis obliterans and the risk of pneumothorax after transbronchial biopsy. Respiratory Medicine CME 2010;3:87–9.
47. Sakai R, Kanamori H, Nakaseko C, et al. Air-leak syndrome following allo-SCT in adult patients: report from the Kanto Study Group for Cell Therapy in Japan. Bone Marrow Transplant 2011;46:379–84.
48. Visscher DW, Myers JL. Histologic spectrum of idiopathic interstitial pneumonias. Proc Am Thorac Soc 2006;3:322–9.
49. Cordier JF. Cryptogenic organising pneumonia. Eur Respir J 2006;28:422–46.
50. Nishio N, Yagasaki H, Takahashi Y, et al. Late-onset non-infectious pulmonary complications following allogeneic hematopoietic stem cell transplantation in children. Bone Marrow Transplant 2009;44:303–8.
51. Ueda K, Watadani T, Maeda E, et al. Outcome and treatment of late-onset noninfectious pulmonary complications after allogeneic haematopoietic SCT. Bone Marrow Transplant 2010;45:1719–27.
52. Schlemmer F, Chevret S, Lorillon G, et al. Late-onset noninfectious interstitial lung disease after allogeneic hematopoietic stem cell transplantation. Respir Med 2014;108:1525–33.
53. Palmas A, Tefferi A, Myers JL, et al. Late-onset noninfectious pulmonary complications after allogeneic bone marrow transplantation. Br J Haematol 1998;100:680–7.
54. Sakaida E, Nakaseko C, Harima A, et al. Late-onset noninfectious pulmonary complications after allogeneic stem cell transplantation are significantly associated with chronic graft-versus-host disease and with the graft-versus-leukemia effect. Blood 2003;102:4236–42.
55. Solh M, Arat M, Cao Q, et al. Late-onset noninfectious pulmonary complications in adult allogeneic hematopoietic cell transplant recipients. Transplantation 2011;91:798–803.
56. Dandoy CE, Hirsch R, Chima R, et al. Pulmonary hypertension after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013;19:1546–56.
57. Bunte MC, Patnaik MM, Pritzker MR, Burns LJ. Pulmonary veno-occlusive disease following hematopoietic stem cell transplantation: a rare model of endothelial dysfunction. Bone Marrow Transplant 2008;41:677–86.
58. Troussard X, Bernaudin JF, Cordonnier C, et al. Pulmonary veno-occlusive disease after bone marrow transplantation. Thorax 1984;39:956–7.
59. Lahzami S, Schoeffel RE, Pechey V, et al. Small airways function declines after allogeneic haematopoietic stem cell transplantation. Eur Respir J 2011;38:1180–8.
60. Jain NA, Pophali PA, Klotz JK, et al. Repair of impaired pulmonary function is possible in very-long-term allogeneic stem cell transplantation survivors. Biol Blood Marrow Transplant 2014;20:209–13.
61. Barisione G, Bacigalupo A, Crimi E, et al. Changes in lung volumes and airway responsiveness following haematopoietic stem cell transplantation. Eur Respir J 2008;32:1576–82.
62. Kovalszki A, Schumaker GL, Klein A, et al. Reduced respiratory and skeletal muscle strength in survivors of sibling or unrelated donor hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;41:965–9.
63. Mathiesen S, Uhlving HH, Buchvald F, et al. Aerobic exercise capacity at long-term follow-up after paediatric allogeneic haematopoietic SCT. Bone Marrow Transplant 2014;49:1393–9.
1. Remberger M, Ackefors M, Berglund S, et al. Improved survival after allogeneic hematopoietic stem cell transplantation in recent years. A single-center study. Biol Blood Marrow Transplant 2011;17:1688–97.
2. Wood KL, Esguerra VG. Management of late pulmonary complications after hematopoietic stem cell transplantation. Hosp Phys Hematology-Oncology Board Review Manual 2018;13(1):36–48.
3. Ninin E, Milpied N, Moreau P, et al. Longitudinal study of bacterial, viral, and fungal infections in adult recipients of bone marrow transplants. Clin Infect Dis 2001;33:41–7.
4. Roca J, Granena A, Rodriguez-Roisin R, et al. Fatal airway disease in an adult with chronic graft-versus-host disease. Thorax 1982;37:77–8.
5. Williams KM, Chien JW, Gladwin MT, Pavletic SZ. Bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. JAMA 2009;302:306–14.
6. Chien JW, Martin PJ, Gooley TA, et al. Airflow obstruction after myeloablative allogeneic hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2003;168:208–14.
7. Holbro A, Lehmann T, Girsberger S, et al. Lung histology predicts outcome of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013;19:973–80.
8. Chamberlain D, Maurer J, Chaparro C, Idolor L. Evaluation of transbronchial lung biopsy specimens in the diagnosis of bronchiolitis obliterans after lung transplantation. J Heart Lung Transplant 1994;13:963–71.
9. Au BK, Au MA, Chien JW. Bronchiolitis obliterans syndrome epidemiology after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2011;17:1072–8.
10. Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 2015;21:389–401.
11. Couriel D, Carpenter PA, Cutler C, et al. Ancillary therapy and supportive care of chronic graft-versus-host disease: national institutes of health consensus development project on criteria for clinical trials in chronic Graft-versus-host disease: V. Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant 2006;12:375–96.
12. Pellegrino R, Viegi G, Brusasco V, et al. Interpretative strategies for lung function tests. Eur Respir J 2005;26:948–68.
13. Williams KM, Hnatiuk O, Mitchell SA, et al. NHANES III equations enhance early detection and mortality prediction of bronchiolitis obliterans syndrome after hematopoietic SCT. Bone Marrow Transplant 2014;49:561–6.
14. Hyatt RE, Cowl CT, Bjoraker JA, Scanlon PD. Conditions associated with an abnormal nonspecific pattern of pulmonary function tests. Chest 2009;135:419–24.
15. Bergeron A, Godet C, Chevret S, et al. Bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: phenotypes and prognosis. Bone Marrow Transplant 2013;48:819–24.
16. Filipovich AH, Weisdorf D, Pavletic S, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant 2005;11:945–56.
17. Gazourian L, Coronata AM, Rogers AJ, et al. Airway dilation in bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. Respir Med 2013;107:276–83.
18. Gunn ML, Godwin JD, Kanne JP, et al. High-resolution CT findings of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. J Thorac Imaging 2008;23:244–50.
19. Sargent MA, Cairns RA, Murdoch MJ, et al. Obstructive lung disease in children after allogeneic bone marrow transplantation: evaluation with high-resolution CT. AJR Am J Roentgenol 1995;164:693–6.
20. Galban CJ, Boes JL, Bule M, et al. Parametric response mapping as an indicator of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20:1592–8.
21. Meyer KC, Raghu G, Verleden GM, et al. An international ISHLT/ATS/ERS clinical practice guideline: diagnosis and management of bronchiolitis obliterans syndrome. Eur Respir J 2014;44:1479–1503.
22. Hildebrandt GC, Fazekas T, Lawitschka A, et al. Diagnosis and treatment of pulmonary chronic GVHD: report from the consensus conference on clinical practice in chronic GVHD. Bone Marrow Transplant 2011;46:1283–95.
23. Carpenter PA, Kitko CL, Elad S, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: V. The 2014 Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant 2015;21:1167–87.
24. Norman BC, Jacobsohn DA, Williams KM, et al. Fluticasone, azithromycin and montelukast therapy in reducing corticosteroid exposure in bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: a case series of eight patients. Bone Marrow Transplant 2011;46:1369–73.
25. Williams KM, Cheng GS, Pusic I, et al. Fluticasone, azithromycin, and montelukast treatment for new-onset bronchiolitis obliterans syndrome after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2016;22:710–6.
26. Or R, Gesundheit B, Resnick I, et al. Sparing effect by montelukast treatment for chronic graft versus host disease: a pilot study. Transplantation 2007;83:577–81.
27. Cheng GS, Storer B, Chien JW, et al. Lung function trajectory in bronchiolitis obliterans syndrome after allogeneic hematopoietic cell transplant. Ann Am Thorac Soc 2016;13:1932–9.
28. Bergeron A, Belle A, Chevret S, et al. Combined inhaled steroids and bronchodilatators in obstructive airway disease after allogeneic stem cell transplantation. Bone Marrow Transplant 2007;39:547–53.
29. Bashoura L, Gupta S, Jain A, et al. Inhaled corticosteroids stabilize constrictive bronchiolitis after hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;41:63–7.
30. Bergeron A, Chevret S, Granata A, et al. Effect of azithromycin on airflow decline-free survival after allogeneic hematopoietic stem cell transplant: the ALLOZITHRO randomized clinical trial. JAMA 2017;318:557–66.
31. Gerhardt SG, McDyer JF, Girgis RE, et al. Maintenance azithromycin therapy for bronchiolitis obliterans syndrome: results of a pilot study. Am J Respir Crit Care Med 2003;168:121–5.
32. Khalid M, Al Saghir A, Saleemi S, et al. Azithromycin in bronchiolitis obliterans complicating bone marrow transplantation: a preliminary study. Eur Respir J 2005;25:490–3.
33. Maimon N, Lipton JH, Chan CK, Marras TK. Macrolides in the treatment of bronchiolitis obliterans in allograft recipients. Bone Marrow Transplant 2009;44:69–73.
34. Lam DC, Lam B, Wong MK, et al. Effects of azithromycin in bronchiolitis obliterans syndrome after hematopoietic SCT--a randomized double-blinded placebo-controlled study. Bone Marrow Transplant 2011;46:1551–6.
35. Yadav H, Peters SG, Keogh KA, et al. Azithromycin for the treatment of obliterative bronchiolitis after hematopoietic stem cell transplantation: a systematic review and meta-analysis. Biol Blood Marrow Transplant 2016;22:2264–9.
36. Vos R, Vanaudenaerde BM, Verleden SE, et al. A randomised controlled trial of azithromycin to prevent chronic rejection after lung transplantation. Eur Respir J 2011;37:164–72.
37. Svanstrom H, Pasternak B, Hviid A. Use of azithromycin and death from cardiovascular causes. N Engl J Med 2013;368:1704–12.
38. Albert RK, Connett J, Bailey WC, et al. Azithromycin for prevention of exacerbations of COPD. N Engl J Med 2011;365:689–98.
39. Tran J, Norder EE, Diaz PT, et al. Pulmonary rehabilitation for bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2012;18:1250–4.
40. Lucid CE, Savani BN, Engelhardt BG, et al. Extracorporeal photopheresis in patients with refractory bronchiolitis obliterans developing after allo-SCT. Bone Marrow Transplant 2011;46:426–9.
41. Hostettler KE, Halter JP, Gerull S, et al. Calcineurin inhibitors in bronchiolitis obliterans syndrome following stem cell transplantation. Eur Respir J 2014;43:221–32.
42. Holm AM, Riise GC, Brinch L, et al. Lung transplantation for bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation: unresolved questions. Transplantation 2013;96:e21–22.
43. Cheng GS, Edelman JD, Madtes DK, et al. Outcomes of lung transplantation after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014;20:1169–75.
44. Okumura H, Ohtake S, Ontachi Y, et al. Living-donor lobar lung transplantation for broncho-bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation: does bronchiolitis obliterans recur in transplanted lungs? Int J Hematol 2007;86:369–73.
45. Olivieri A, Cimminiello M, Corradini P, et al. Long-term outcome and prospective validation of NIH response criteria in 39 patients receiving imatinib for steroid-refractory chronic GVHD. Blood 2013;122:4111–8.
46. Rahmanian S, Wood KL. Bronchiolitis obliterans and the risk of pneumothorax after transbronchial biopsy. Respiratory Medicine CME 2010;3:87–9.
47. Sakai R, Kanamori H, Nakaseko C, et al. Air-leak syndrome following allo-SCT in adult patients: report from the Kanto Study Group for Cell Therapy in Japan. Bone Marrow Transplant 2011;46:379–84.
48. Visscher DW, Myers JL. Histologic spectrum of idiopathic interstitial pneumonias. Proc Am Thorac Soc 2006;3:322–9.
49. Cordier JF. Cryptogenic organising pneumonia. Eur Respir J 2006;28:422–46.
50. Nishio N, Yagasaki H, Takahashi Y, et al. Late-onset non-infectious pulmonary complications following allogeneic hematopoietic stem cell transplantation in children. Bone Marrow Transplant 2009;44:303–8.
51. Ueda K, Watadani T, Maeda E, et al. Outcome and treatment of late-onset noninfectious pulmonary complications after allogeneic haematopoietic SCT. Bone Marrow Transplant 2010;45:1719–27.
52. Schlemmer F, Chevret S, Lorillon G, et al. Late-onset noninfectious interstitial lung disease after allogeneic hematopoietic stem cell transplantation. Respir Med 2014;108:1525–33.
53. Palmas A, Tefferi A, Myers JL, et al. Late-onset noninfectious pulmonary complications after allogeneic bone marrow transplantation. Br J Haematol 1998;100:680–7.
54. Sakaida E, Nakaseko C, Harima A, et al. Late-onset noninfectious pulmonary complications after allogeneic stem cell transplantation are significantly associated with chronic graft-versus-host disease and with the graft-versus-leukemia effect. Blood 2003;102:4236–42.
55. Solh M, Arat M, Cao Q, et al. Late-onset noninfectious pulmonary complications in adult allogeneic hematopoietic cell transplant recipients. Transplantation 2011;91:798–803.
56. Dandoy CE, Hirsch R, Chima R, et al. Pulmonary hypertension after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013;19:1546–56.
57. Bunte MC, Patnaik MM, Pritzker MR, Burns LJ. Pulmonary veno-occlusive disease following hematopoietic stem cell transplantation: a rare model of endothelial dysfunction. Bone Marrow Transplant 2008;41:677–86.
58. Troussard X, Bernaudin JF, Cordonnier C, et al. Pulmonary veno-occlusive disease after bone marrow transplantation. Thorax 1984;39:956–7.
59. Lahzami S, Schoeffel RE, Pechey V, et al. Small airways function declines after allogeneic haematopoietic stem cell transplantation. Eur Respir J 2011;38:1180–8.
60. Jain NA, Pophali PA, Klotz JK, et al. Repair of impaired pulmonary function is possible in very-long-term allogeneic stem cell transplantation survivors. Biol Blood Marrow Transplant 2014;20:209–13.
61. Barisione G, Bacigalupo A, Crimi E, et al. Changes in lung volumes and airway responsiveness following haematopoietic stem cell transplantation. Eur Respir J 2008;32:1576–82.
62. Kovalszki A, Schumaker GL, Klein A, et al. Reduced respiratory and skeletal muscle strength in survivors of sibling or unrelated donor hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;41:965–9.
63. Mathiesen S, Uhlving HH, Buchvald F, et al. Aerobic exercise capacity at long-term follow-up after paediatric allogeneic haematopoietic SCT. Bone Marrow Transplant 2014;49:1393–9.
Translating AHA/ACC cholesterol guidelines into meaningful risk reduction
A new cholesterol guideline1 builds on the 2013 American College of Cardiology (ACC)/American Heart Association (AHA) cholesterol guidelines,2 which were a major paradigm shift in the evaluation and management of blood cholesterol levels and risk for atherosclerotic cardiovascular disease (ASCVD). The work was presented (and simultaneously published) on November 10, 2018, at the annual AHA Scientific Sessions in Chicago. Full text,1 an executive summary,3 and accompanying systematic review of evidence4 are available online.
The 2018 AHA/ACC cholesterol guideline represents a step forward in ASCVD prevention—especially in primary prevention, where it provides guidance for risk refinement and personalization. In this article, we mine the details of what has changed and what is new in this guideline so that you can prepare to adopt the recommendations in your practice.
2013 and 2018 guidelines: Similarities, differences
As in earlier iterations, the 2018 guideline emphasizes healthy lifestyle across the life-course as the basis of ASCVD prevention—as elaborated in the 2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk.5 In contrast to the 2013 guidelines,2 the 2018 guideline is more comprehensive and more personalized, focusing on risk assessment for individual patients, rather than simply providing population-based approaches. Moreover, the guideline isn’t limited to adults: It makes recommendations pertaining to children and adolescents.1
TABLE 11,2 compares the most important differences between the 2013 and 2018 guidelines.
The 2013 ACC/AHA guidelines eliminated low-density lipoprotein cholesterol (LDL-C) and non-high-density lipoprotein cholesterol (non-HDL-C)a goals of therapy and replaced them with the concept of 4 “statin benefit groups”—that is, patient populations for which clear evidence supports the role of statin therapy.4 In the 2018 guideline, statin benefit groups have been maintained, although without explicit use of this term.1
Primary prevention. Although no major changes in statin indications are made for patients with (1) established ASCVD (ie, for secondary prevention), (2) diabetes mellitus (DM) and who are 40 to 75 years of age, or (3) a primary LDL-C elevation ≥ 190 mg/dL, significant changes were made for primary prevention patients ages 40 to 75 years.1 ASCVD risk calculation using the 2013 pooled cohort equations (PCE) is still recommended4; however, risk estimation is refined by the use of specific so-called risk-enhancing factors (TABLE 21). In cases in which the risk decision remains uncertain, obtaining the coronary artery calcium (CAC) score (which we’ll describe shortly) using specialized computed tomography (CT) is advised to facilitate the shared physician–patient decision-making process.1
LDL-C and non-HDL-C thresholds. Although LDL-C and non-HDL-C goals are not overtly brought back from the 2002 National Cholesterol Education Program/Adult Treatment Panel guidelines,6 the new guideline does introduce LDL-C and non-HDL-C thresholds—levels at which adding nonstatin therapy can be considered, in contrast to previous goals to which therapy was titrated. Definitions of statin intensity remain the same: Moderate-intensity statin therapy is expected to reduce the LDL-C level by 30% to 50%; high-intensity statin therapy, by ≥ 50%.1 The intensity of statin therapy has been de-escalated in the intermediate-risk group, where previous guidelines advised high-intensity statin therapy,4 and replaced with moderate-intensity statin therapy (similar to 2016 US Preventive Services Task Force [USPSTF] recommendations7).
[polldaddy:10312157]
Continue to: Fasting vs nonfasting lipid profiles
Fasting vs nonfasting lipid profiles. In contrast to previous guidelines,2,8 which used fasting lipid profiles, nonfasting lipid profiles are now recommended for establishing a baseline LDL-C level and for ASCVD risk estimation for most patients—as long as the triglycerides (TG) level is < 400 mg/dL. When the calculated LDL-C level is < 70 mg/dL using the standard Friedewald formula, obtaining a direct LDL-C or a modified LDL-C estimate9 is deemed reasonable to improve accuracy. (The modified LDL-C can be estimated using The Johns Hopkins Hospital’s free “LDL Cholesterol Calculator” [www.hopkinsmedicine.org/apps/all-apps/ldl-cholesterol-calculator]).
A fasting lipid profile is still preferred for patients who have a family history of a lipid disorder. The definition of hypertriglyceridemia has been revised from a fasting TG level ≥ 150 mg/dL to a nonfasting or fasting TG level ≥ 175 mg/dL.1
Nonstatin add-on therapy. The new guideline supports the addition of nonstatin therapies to maximally tolerated statin therapy in patients who have established ASCVD or a primary LDL-C elevation ≥ 190 mg/dL when (1) the LDL-C level has not been reduced by the expected percentage (≥ 50% for high-intensity statin therapy) or (2) explicit LDL-C level thresholds have been met.1
The principal 2 groups of recommended nonstatins for which there is randomized, controlled trial evidence of cardiovascular benefit are (1) the cholesterol-absorbing agent ezetimibe10 and (2) the proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors evolocumab11 and alirocumab.12
AAFP’s guarded positions on the 2013 and 2018 guidelines
The American Academy of Family Physicians (AAFP) welcomed the patient-centered and outcome-oriented aspects of the 2013 ACC/AHA guidelines, endorsing them with 3 qualifications.13
- Many of the recommendations were based on expert opinion, not rigorous research results—in particular, not on the findings of randomized controlled trials (although key points are based on high-quality evidence).
- There were conflicts of interest disclosed for 15 members of the guidelines panel, including a vice chair.
- Validation of the PCE risk estimation tool was lacking.
Continue to: AAFP announced...
AAFP announced in March that it does not endorse the 2018 AHA/ACC guideline, asserting that (1) only a small portion of the recommendations, primarily focused on the addition of nonstatin therapy, were addressed by an independent systematic review and (2) many of the guideline recommendations are based on low-quality or insufficient evidence. AAFP nevertheless bestowed an “affirmation of value” designation on the guideline—meaning that it provides some benefit for family physicians’ practice without fulfilling all criteria for full endorsement.14
Detailed recommendations from the 2018 guideline
Lifestyle modification
When talking about ASCVD risk with patients, it is important to review current lifestyle habits (eg, diet, physical activity, weight or body mass index, and tobacco use). Subsequent to that conversation, a healthy lifestyle should be endorsed and relevant advice provided. In addition, patient-directed materials (eg, ACC’s CardioSmart [www.cardiosmart.org]; AHA’s Life’s Simple 7 [www.heart.org/en/professional/workplace-health/lifes-simple-7]; and the National Lipid Association’s Patient Tear Sheets [www.lipid.org/practicetools/tools/tearsheets] and Clinicians’ Lifestyle Modification Toolbox [www.lipid.org/CLMT]) and referrals (eg, to cardiac rehabilitation, a dietitian, a smoking-cessation program) should be provided.1
Primary prevention of ASCVD
Risk assessment for primary prevention is now approached as a process, rather than the simple risk calculation used in the 2013 ACC/AHA guidelines.2 Assessment involves risk estimation followed by risk personalization, which, in some cases, is followed by risk reclassification using CAC scoring.1
Patients are classified into 1 of 4 risk groups, based on the PCE1:
- low (< 5%)
- borderline (5%-7.5%)
- intermediate (7.5%-19.9%)
- high (≥ 20%).
However, the PCE-based risk score is a population-based tool, which might not reflect the actual risk of individual patients. In some populations, PCE underestimates ASCVD risk; in others, it overestimates risk. A central tenet of the new guideline is personalization of risk, taking into account the unique circumstances of each patient. Moreover, the new guideline provides guidance on how to interpret the PCE risk score for several different ethnic and racial groups.1
Continue to: Medical therapy
Medical therapy. The decision to start lipid-lowering therapy should be made after a physician–patient discussion that considers costs of therapy as well as patient preferences and values in the context of shared decision-making. Discussion should include a review of major risk factors (eg, cigarette smoking, elevated blood pressure, and the LDL-C level), the PCE risk score, the presence of risk-enhancing factors (TABLE 21), potential benefits of lifestyle changes and statin therapy, and the potential for adverse drug effects and drug–drug interactions.1
If the estimated ASCVD risk is 7.5%-19.9%, starting moderate-intensity statin therapy is recommended. Risk-enhancing factors favor initiation of statin therapy, even in patients at borderline risk (5%-7.5%). If risk is uncertain, the CAC score can be used to facilitate shared decision-making.1 The use of CAC is in agreement with the USPSTF statement that CAC can moderately improve discrimination and reclassification, but has an unclear effect on downstream health care utilization.15 Importantly, CAC should not be measured routinely in patients already taking a statin because its primary role is to facilitate shared decision-making regarding initiation of statin therapy.16
If the 10-year ASCVD risk is ≥ 20%, high-intensity statin therapy is advised, without need to obtain the CAC score. If high-intensity statin therapy is advisable but not acceptable to, or tolerated by, the patient, it might be reasonable to add a nonstatin drug (ezetimibe or a bile-acid sequestrant) to moderate-intensity statin therapy.1
Risk-enhancing factors (TABLE 21) apply to intermediate- and borderline-risk patients. Importantly, these factors include membership in specific ethnic groups, conditions specific to females, and male–female distinctions in risk. Risk-enhancing factors also incorporate biomarkers that are often measured by lipid specialists, such as lipoprotein(a) (Lp[a]) and apolipoprotein B (ApoB).1
Lp(a) is an atherogenic particle, akin to an LDL particle, that consists of a molecule of apolipoprotein (a) (a nonfunctional mimic of a portion of plasminogen) covalently bound to ApoB, like the one found on the LDL particle. Lp(a) is proportionally associated with an increased risk for ASCVD and aortic stenosis at a level > 50 mg/dL.17 A family history of premature ASCVD is a relative indication for measuring Lp(a).1
Continue to: When and why to measure CAC
When and why to measure CAC
If the decision to initiate statin therapy is still uncertain after risk estimation and personalization, or when a patient is undecided about committing to lifelong lipid-lowering therapy, the new guideline recommends obtaining a CAC score to inform the shared decision-making process.1,18 Measurement of CAC is obtained by noncontrast, electrocardiographic-gated CT that can be performed in 10 to 15 minutes, requiring approximately 1 millisievert of radiation (equivalent of the approximate dose absorbed during 2 mammograms). Although measurement of the CAC score is generally not covered by insurance, its cost ($50-$450) nationwide makes it accessible.19
CAC measures the presence (or absence) of subclinical atherosclerosis by detecting calcified plaque in coronary arteries. The absolute CAC score is expressed in Agatston units; an age–gender population percentile is also provided. Keep in mind that the presence of any CAC (ie, a score > 0) is abnormal and demonstrates the presence of subclinical coronary artery disease. The prevalence of CAC > 0 increases with age, but a significant percentage of older people have a CAC score = 0. When CAC > 0, additional information is provided by the distribution of plaque burden among the different coronary arteries.20
Among intermediate-risk patients, 50% have CAC = 0 and, therefore, a very low event rate over the ensuing 10 years, which allows statin therapy to be safely deferred unless certain risk factors are present (eg, family history, smoking, DM).1,18 It is reasonable to repeat CAC testing in 5 to 10 years to assess whether subclinical atherosclerosis has developed. The 2018 guideline emphasizes that, when the CAC score is > 0 but < 100 Agatston units, statin therapy is favored, especially in patients > 55 years of age; when the CAC score is ≥ 100 Agatston units or at the ≥ 75th percentile, statin therapy is indicated regardless of age.1
Patients who might benefit from knowing their CAC score include those who are:
- reluctant to initiate statin therapy but who want to understand their risk and potential for benefit more precisely
- concerned about the need to reinstitute statin therapy after discontinuing it because of statin-associated adverse effects
- older (men, 55-80 years; women, 60-80 years) who have a low burden of risk factors and who question whether they would benefit from statin therapy
- middle-aged (40-55 years) and who have a PCE-calculated risk of 5% to < 7.5% for ASCVD and factors that increase their risk for ASCVD, even though they are in a borderline-risk group.1
Primary prevention in special populations
Older patients. In adults ≥ 75 years who have an LDL-C level 70 to 189 mg/dL, initiating a moderate-intensity statin might be reasonable; however, it might also be reasonable to stop treatment in this population when physical or cognitive decline, multiple morbidities, frailty, or reduced life expectancy limits the potential benefit of statin therapy. It might be reasonable to use the CAC score in adults 76 to 80 years of age who have an LDL-C level of 70 to 189 mg/dL to reclassify those whose CAC score = 0, so that they can avoid statin therapy.1
Continue to: Children and adolescents
Children and adolescents. In alignment with current pediatric guidelines,21 but in contrast to USPSTF reccomendations,22 the 2018 ACC/AHA guideline endorses universal lipid screening for pediatric patients (see TABLE W11,21,22). It is reasonable to obtain a fasting lipid profile or nonfasting non-HDL-C in all children and adolescents who have neither cardiovascular risk factors nor a family history of early cardiovascular disease to detect moderate-to-severe lipid abnormalities. Screening should be done once at 9 to 11 years of age and again at 17 to 21 years.1
A screening test as early as 2 years of age to detect familial hypercholesterolemia (FH) is reasonable when a family history of either early CVD or significant hypercholesterolemia is present. The guideline endorses reverse cascade screening for detection of FH in family members of children and adolescents who have severe hypercholesterolemia.1
In children and adolescents with a lipid abnormality, especially when associated with the metabolic syndrome, lifestyle counseling is beneficial for lowering the LDL-C level. In children and adolescents ≥ 10 years of age with (1) an LDL-C level persistently ≥ 190 mg/dL or (2) an LDL level ≥ 160 mg/dL plus a clinical presentation consistent with FH, it is reasonable to initiate statin therapy if they do not respond adequately to 3 to 6 months of lifestyle therapy.1
Ethnicity as a risk-modifying factor. The PCE distinguishes between US adults of European ancestry and African ancestry, but no other ethnic groups are distinguished.4 The new guideline advocates for the use of PCE in other populations; however, it states that, for clinical decision-making purposes, it is reasonable, in adults of different races and ethnicities, for the physician to review racial and ethnic features that can influence ASCVD risk to allow adjustment of the choice of statin or intensity of treatment. Specifically, South Asian ancestry is now treated as a risk-enhancing factor, given the high prevalence of premature and extensive ASCVD in this patient population.1
Concerns specific to women. Considering conditions specific to women as potential risk-enhancing factors is advised when discussing lifestyle intervention and the potential for benefit from statin therapy—in particular, (1) in the setting of premature menopause (< 40 years) and (2) when there is a history of a pregnancy-associated disorder (eg, hypertension, preeclampsia, gestational DM, a small-for-gestational-age infant, and preterm delivery). If the decision is made to initiate statin therapy in women of childbearing age who are sexually active, there is a guideline mandate to counsel patients on using reliable contraception. When pregnancy is planned, statin therapy should be discontinued 1 to 2 months before pregnancy is attempted; when pregnancy occurs while a patient is taking a statin, therapy should be stopped as soon as the pregnancy is discovered.1
Continue to: Adults with chronic kidney disease
Adults with chronic kidney disease. Chronic kidney disease that is not treated with dialysis or kidney transplantation is considered a risk-enhancing factor; initiation of a moderate-intensity statin or a moderate-intensity statin plus ezetimibe can be useful in patients with chronic kidney disease who are 40 to 75 years of age and have an LDL-C level of 70 to 189 mg/dL and a PCE-calculated risk ≥ 7.5%. In adults with advanced kidney disease that requires dialysis who are already taking a statin, it may be reasonable to continue the statin; however, initiation of a statin in adults with advanced kidney disease who require dialysis is not recommended because of an apparent lack of benefit.1
Adults with a chronic inflammatory disorder or human immunodeficiency virus infection. Any of these conditions are treated as risk-enhancing factors; in a risk discussion with affected patients, therefore, moderate-intensity statin therapy or high-intensity statin therapy is favored for those 40 to 75 years of age who have an LDL-C level of 70 to 189 mg/dL and PCE-calculated risk ≥ 7.5%. A fasting lipid profile and assessment of ASCVD risk factors for these patients can be useful (1) as a guide to the potential benefit of statin therapy and (2) for monitoring or adjusting lipid-lowering drug therapy before, and 4 to 12 weeks after, starting inflammatory disease-modifying therapy or antiretroviral therapy.
In adults with rheumatoid arthritis who undergo ASCVD risk assessment with a lipid profile, it can be useful to recheck lipid values and other major ASCVD risk factors 2 to 4 months after the inflammatory disease has been controlled.1
Primary hypercholesterolemia
The diagnosis and management of heterozygous or homozygous familial hypercholesterolemia (HeFH or HoFH) is beyond the scope of the 2018 ACC/AHA cholesterol guidelines; instead, the 2015 AHA Scientific Statement, “The Agenda for Familial Hypercholesterolemia,” provides a contemporary review of these topics.23 However, the 2018 cholesterol guideline does acknowledge that an LDL-C level ≥ 190 mg/dL often corresponds to primary (ie, genetic) hypercholesterolemia.
In patients 20 to 75 years of age who have a primary elevation of LDL-C level ≥ 190 mg/dL, the guideline recommends initiation of high-intensity statin therapy without calculating ASCVD risk using the PCE. If a > 50% LDL-C reduction is not achieved, or if the LDL-C level on maximally tolerated statin therapy remains ≥ 100 mg/dL, adding ezetimibe is considered reasonable. If there is < 50% reduction in the LDL-C level while taking maximally tolerated statin and ezetimibe therapy, adding a bile-acid sequestrant can be considered, as long as the TG level is not > 300 mg/dL (ie, bile-acid sequestrants can elevate the TG level significantly).
Continue to: In patients 30 to 75 years of age...
In patients 30 to 75 years of age who have a diagnosis of HeFH and an LDL-C level ≥ 100 mg/dL while taking maximally tolerated statin and ezetimibe therapy, the addition of a PCSK9 inhibitor can be considered. Regardless of whether there is a diagnosis of HeFH, addition of a PCSK9 inhibitor can be considered in patients 40 to 75 years of age who have a baseline LDL-C level ≥ 220 mg/dL and who achieve an on-treatment LDL-C level ≥ 130 mg/dL while receiving maximally tolerated statin therapy and ezetimibe.1
Diabetes mellitus
In patients with DM who are 40 to 75 years of age, moderate-intensity statin therapy is recommended without calculating the 10-year ASCVD risk. When the LDL-C level is 70 to 189 mg/dL, however, it is reasonable to use the PCE to assess 10-year ASCVD risk to facilitate risk stratification.
In patients with DM who are at higher risk, especially those who have multiple risk factors or are 50 to 75 years of age, it is reasonable to use a high-intensity statin to reduce the LDL-C level by ≥ 50 %. In adults > 75 years of age with DM who are already on statin therapy, it is reasonable to continue statin therapy; for those that age who are not on statin therapy, it might be reasonable to initiate statin therapy after a physician–patient discussion of potential benefits and risks.
In adults with DM and PCE-calculated risk ≥ 20%, it might be reasonable to add ezetimibe to maximally tolerated statin therapy to reduce the LDL-C level by ≥ 50%. In adults 20 to 39 years of age with DM of long duration (≥ 10 years of type 2 DM, ≥ 20 years of type 1 DM), albuminuria (≥ 30 μg of albumin/mg creatinine), estimated glomerular filtration rate < 60 mL/min/1.73 m2, retinopathy, neuropathy, or ankle-brachial index < 0.9, it might be reasonable to initiate statin therapy.1
Secondary prevention
Presence of clinical ASCVD. In patients with clinical ASCVD who are ≤ 75 years of age, high-intensity statin therapy should be initiated or continued, with the aim of achieving ≥ 50% reduction in the LDL-C level. When high-intensity statin therapy is contraindicated or if a patient experiences statin-associated adverse effects, moderate-intensity statin therapy should be initiated or continued with the aim of achieving a 30% to 49% reduction in the LDL-C level.
Continue to: In patients...
In patients > 75 years of age with clinical ASCVD, it is reasonable to initiate or continue moderate- or high-intensity statin therapy after evaluation of the potential for ASCVD risk reduction, adverse effects, and drug–drug interactions, as well as patient frailty and patient preference.1
Very high risk. In patients at very high risk (this includes a history of multiple major ASCVD events or 1 major ASCVD event plus multiple high-risk conditions), maximally tolerated LDL-C-lowering therapy should include maximally tolerated statin therapy and ezetimibe before considering a PCSK9 inhibitor. An LDL-C level ≥ 70 mg/dL or a non-HDL-C level ≥ 100 mg/dL is considered a reasonable threshold for adding a PCSK9 inhibitor to background lipid-lowering therapy1 (TABLE 31).
Heart failure. In patients with heart failure who have (1) a reduced ejection fraction attributable to ischemic heart disease, (2) a reasonable life expectancy (3-5 years), and (3) are not already on a statin because of ASCVD, consider initiating moderate-intensity statin therapy to reduce the risk for an ASCVD event.1
Reduction of elevated triglycerides
The guideline defines moderate hypertriglyceridemia as a nonfasting or fasting TG level of 175 to 499 mg/dL. Such a finding is considered a risk-enhancing factor and is 1 of 5 components of the metabolic syndrome. Three independent measurements are advised to diagnose primary moderate hypertriglyceridemia. Severe hypertriglyceridemia is diagnosed when the fasting TG level is ≥ 500 mg/dL.1
In moderate hypertriglyceridemia, most TGs are carried in very-low-density lipoprotein particles; in severe hypertriglyceridemia, on the other hand, chylomicrons predominate, raising the risk for pancreatitis. In adults with severe hypertriglyceridemia, therefore—especially when the fasting TG level is ≥ 1000 mg/dL—it is reasonable to identify and address other causes of hypertriglyceridemia. If TGs are persistently elevated or increasing, levels should be reduced to prevent acute pancreatitis with a very low-fat diet and by avoiding refined carbohydrates and alcohol; consuming omega-3 fatty acids; and, if necessary, taking a fibrate.1
Continue to: In adults...
In adults ≥ 20 years of age with moderate hypertriglyceridemia, lifestyle factors (eg, obesity, metabolic syndrome), secondary factors (eg, DM, chronic liver or kidney disease, nephrotic syndrome, hypothyroidism), and medications that increase the TG level need to be addressed first. In adults 40 to 75 years of age with moderate or severe hypertriglyceridemia and a PCE-calculated ASCVD risk ≥ 7.5%, it is reasonable to reevaluate risk after lifestyle and secondary factors are addressed and to consider a persistently elevated TG level as a factor favoring initiation or intensification of statin therapy. In adults 40 to 75 years of age with severe hypertriglyceridemia and ASCVD risk ≥ 7.5%, it is reasonable to address reversible causes of a high TG level and to initiate statin therapy.1
Other considerations in cholesterol management
Tools to assess adherence
The response to lifestyle and statin therapy should be evaluated by the percentage reduction in the LDL-C level compared with baseline, not by assessment of the absolute LDL-C level. When seeing a patient whose treatment is ongoing, a baseline level can be estimated using a desktop LDL-calculator app.
Adherence and percentage response to LDL-C–lowering medications and lifestyle changes should be evaluated with repeat lipid measurement 4 to 12 weeks after either a statin is initiated or the dosage is adjusted, and repeated every 3 to 12 months as needed. In patients with established ASCVD who are at very high risk, triggers for adding nonstatin therapy are defined by a threshold LDL-C level ≥ 70 mg/dL on maximal statin therapy.1
Interventions focused on improving adherence to prescribed therapy are recommended for management of adults with an elevated cholesterol level. These interventions include telephone reminders, calendar reminders, integrated multidisciplinary educational activities, and pharmacist-led interventions, such as simplification of the medication regimen to once-daily dosing.1
Statin safety and associated adverse effects
A physician–patient risk discussion is recommended before initiating statin therapy to review net clinical benefit, during which the 2 parties weigh the potential for ASCVD risk reduction against the potential for statin-associated adverse effects, statin–drug interactions, and safety, with the physician emphasizing that adverse effects can be addressed successfully.
Continue to: Statins are one of...
Statins are one of the safest classes of medication, with an excellent risk-benefit ratio. However, there are myriad confusing media reports regarding potential adverse effects and safety of the statin class—reports that often lead patients to discontinue or refuse statins.
Statin-associated adverse effects include the common statin-associated muscle symptoms (SAMS), new-onset DM, cognitive effects, and hepatic injury. The frequency of new-onset DM depends on the population exposed to statins, with a higher incidence of new-onset DM found in patients who are already predisposed, such as those with obesity, prediabetes, and metabolic syndrome. Cognitive effects are rare and difficult to interpret; they were not reported in the large statin mega-trials but have been described in case reports. Significant transaminase elevations > 3 times the upper limit of normal are infrequent; hepatic failure with statins is extremely rare and found at the same incidence in the general population.1
SAMS include (in order of decreasing prevalence)24:
- myalgias with a normal creatine kinase (CK) level
- conditions such as myositis or myopathy (elevated CK level)
- rhabdomyolysis (CK level > 10 times the upper limit of normal, plus renal injury)
- extremely rare statin-associated autoimmune myopathy, with detectable 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase antibodies.
In patients with SAMS, thorough assessment of symptoms is recommended, in addition to evaluation for nonstatin causes and predisposing factors. Identification of potential SAMS-predisposing factors is recommended before initiation of treatment, including demographics (eg, East-Asian ancestry), comorbid conditions (eg, hypothyroidism and vitamin D deficiency), and use of medications adversely affecting statin metabolism (eg, cyclosporine).
In patients with statin-associated adverse effects that are not severe, it is recommended to reassess and rechallenge to achieve a maximal lowering of the LDL-C level by a modified dosing regimen or an alternate statin or by combining a statin with nonstatin therapy. In patients with increased risk for DM or new-onset DM, it is recommended to continue statin therapy.
Continue to: Routine CK and liver function testing...
Routine CK and liver function testing is not useful in patients treated with statins; however, it is recommended that CK be measured in patients with severe SAMS or objective muscle weakness, or both, and to measure liver function if symptoms suggest hepatotoxicity. In patients at increased risk for ASCVD who have chronic, stable liver disease (including non-alcoholic fatty liver disease), it is reasonable, when appropriately indicated, to use statins after obtaining baseline measurements and determining a schedule of monitoring and safety checks.
In patients at increased risk for ASCVD who have severe or recurrent SAMS after appropriate statin rechallenge, it is reasonable to use nonstatin therapy that is likely to provide net clinical benefit. The guideline does not recommend routine use of coenzyme Q10 supplementation for the treatment or prevention of SAMS.1
Guideline criticism
Guideline development is challenging on multiple levels, including balancing perspectives from multiple stakeholders. Nevertheless, the 2018 AHA/ACC cholesterol guideline builds nicely on progress made since its 2013 predecessor was released.4 This document was developed with the participation of representatives from 10 professional societies in addition to the ACC and AHA—notably, the National Lipid Association and American Society for Preventive Cardiology.1
To refine risk estimation and facilitate shared decision-making, the new guideline introduced so-called risk-enhancing factors and use of the CAC.1 However, some potential risk-enhancing factors were left out: erectile dysfunction, for example, often a marker of increased cardiovascular risk in men < 50 years of age.25 In addition, although pretreatment ApoB was introduced as a risk-enhancing factor,1 no recommendation is given to measure ApoB after initiation of therapy for evaluation of residual cardiovascular risk, as endorsed in other guidelines.26,27
Moreover, the guideline does not include the “extreme risk” category in the guideline developed by the American Association of Clinical Endocrinologists (AACE).28 Although the 2018 AHA/ACC guideline introduces < 70 mg/dL and < 100 mg/dL LDL-C thresholds,1 the < 55 mg/dL LDL-C threshold used for patients in the AACE/American College of Endocrinology extreme-risk category is not mentioned.26 This omission might leave patients who are at extreme ASCVD risk without optimal lipid-lowering therapy. Similarly, the guideline does not elaborate on the diagnosis and treatment of HoFH and HeFH.1 The age cutoff of 30 years for the recommendation to consider PCSK9 inhibitors in patients with HeFH appears arbitrary and excludes younger FH patients who have an extreme LDL-C elevation from potentially important therapy.23
Continue to: Guidelines are dynamic instruments...
Guidelines are dynamic instruments that require constant updating, given the production of new evidence. In fact, the results of the Reduction of Cardiovascular Events With Icosapent Ethyl-Intervention Trial (REDUCE-IT) were presented at the same meeting at which this guideline was unveiled.29 REDUCE-IT demonstrated an astonishing highly significant 25% reduction in the composite primary major adverse cardiovascular event outcome in patients with an LDL-C level of 44 to 100 mg/dL on statin therapy, who had a TG level of 135 to 499 mg/dL and had been treated for a median of 4.9 years with 4 g of pure eicosapentaenoic acid.
In addition, the guideline’s value statements, which address the need to consider the cost of drugs in determining most appropriate treatment, are no longer accurate because the price of PCSK9 inhibitors has dropped by more than half since the guideline was issued.30
An upward climb to clinical payoff
Even after close study of the 2018 AHA/ACC cholesterol guideline, implementing it in practice might remain a challenge to clinicians who are inexperienced in ordering lipid markers such as Lp(a) and interpreting the CAC score. Moreover, initiating and monitoring nonstatin therapies will be a demanding task—especially with PCSK9 inhibitors, which present access difficulties because they are relatively expensive (even after the recent price cut). That’s why, when there is doubt in the mind of the physician or other provider, we will likely see more referrals to specialists in lipid management and ASCVD risk estimation to optimize preventive therapy.31
CORRESPONDENCE
Cezary Wójcik, MD, PhD, FNLA, Oregon Health & Science University, 3181 SW Sam Jackson Park Rd, Portland, OR 97239; [email protected]
1. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 8. pii: S0735-1097(18)39034-X. [Epub ahead of print]
2. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S1-S45.
3. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. pii: S0735-1097(18)39033-8. [Epub ahead of print]
4. Wilson PWF, Polonsky TS, Miedema MD, et al. Systematic review for the 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. pii: S0735-1097(18)39035-1. [Epub ahead of print]
5. Eckel RH, Jakicic JM, Ard JD, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2960-2984.
6. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (adult treatment panel III): final report. Circulation. 2002;106:3143-3421.
7. US Preventive Services Task Force, Bibbins-Domingo K, Grossman DC, et al. Statin use for the primary prevention of cardiovascular disease in adults: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:1997-2007.
8. National Cholesterol Education Program. Second report of the Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (adult treatment panel II). Circulation. 1994;89:1333-1445.
9. Martin SS, Giugliano RP, Murphy SA, et al. Comparison of low-density lipoprotein cholesterol assessment by Martin/Hopkins estimation, Friedewald estimation, and preparative ultracentrifugation: insights from the FOURIER trial. JAMA Cardiol. 2018;3:749-753.
10. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
11. Sabatine MS, Giugliano RP, Keech AC, et al; FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-1722.
12. Szarek M, White HD, Schwartz GG, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab reduces total nonfatal cardiovascular and fatal events in the ODYSSEY OUTCOMES trial. J Am Coll Cardiol. 2019;73:387-396.
13. Crawford C. AAFP endorses ACC/AHA cholesterol management guidelines with qualifications. Leawood, KS: American Academy of Family Physicians; 2014 June 18. www.aafp.org/news/health-of-the-public/20140618cholesterolgdlnendorse.html. Accessed March 20, 2019.
14. Crawford C. AAFP News. AAFP affirms value of new cholesterol management guideline. March 20, 2019. www.aafp.org/news/health-of-the-public/20190320acc-ahacholguidln.html?cmpid=em_AP_20190320. Accessed April 1, 2019.
15. Lin JS, Evans CV, Johnson E, et al. Nontraditional Risk Factors in Cardiovascular Disease Risk Assessment: A Systematic Evidence Report for the U.S. Preventive Services Task Force. Evidence Synthesis, No. 166. Rockville, MD: Agency for Healthcare Research and Quality (US); 2018 Jul. Report No.: 17-05225-EF-1.
16. Puri R, Nicholls SJ, Shao M, et al. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol. 2015;65:1273-1282.
17. Gencer B, Kronenberg F, Stroes ES, et al. Lipoprotein(a): the revenant. Eur Heart J. 2017;38:1553-1560.
18. Michos ED, Blaha MJ, Blumenthal RS. Use of the coronary artery calcium score in discussion of initiation of statin therapy in primary prevention. Mayo Clin Proc. 2017;92:1831-1841.
19. MDsave. Cardiac CT calcium scoring. www.mdsave.com/procedures/cardiac-ct-calcium-scoring/d785f4cf. Accessed Aprl 1, 2019.
20. Blaha MJ, Budoff MJ, Tota-Maharaj R, et al. Improving the CAC score by addition of regional measures of calcium distribution: multi-ethnic study of atherosclerosis. JACC Cardiovasc Imaging. 2016;9:1407-1416.
21. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents: summary report. Pediatrics. 2011;128(Suppl 5):S213-S256.
22. US Preventive Services Task Force, Bibbins-Domingo K, Grossman DC, et al. Screening for lipid disorders in children and adolescents: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:625-633.
23. Gidding SS, Champagne MA, de Ferranti SD, et al; American Heart Association Atherosclerosis, Hypertension, and Obesity in Young Committee of Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and Council on Lifestyle and Cardiometabolic Health. The agenda for familial hypercholesterolemia: a scientific statement from the American Heart Association. Circulation. 2015;132:2167-2192.
24. Newman CB, Preiss D, Tobert JA, et al; American Heart Association Clinical Lipidology, Lipoprotein, Metabolism and Thrombosis Committee, a Joint Committee of the Council on Atherosclerosis, Thrombosis and Vascular Biology and Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular Disease in the Young; Council on Clinical Cardiology; and Stroke Council. Statin safety and associated adverse events: a scientific statement from the American Heart Association. Arterioscler Thromb Vasc Biol. 2019;39:e38-e81.
25. Miner M, Parish SJ, Billups KL, et al. Erectile dysfunction and subclinical cardiovascular disease. Sex Med Rev. 2018 Jan 27. pii: S2050-0521(18)30009-X. [Epub ahead of print]
26. Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1-87.
27. Anderson TJ, Grégoire J, Pearson GJ, et al. 2016 Canadian Cardiovascular Society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2016;32:1263-1282.
28. Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1-87.
29. Bhatt DL, Steg PG, Miller M, et al; REDUCE-IT Investigators. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22.
30. Dangi-Garimella S. Amgen announces 60% reduction in list price of PCSK9 inhibitor evolocumab. AJMC Managed Markets Network. October 24, 2018. https://www.ajmc.com/newsroom/amgen-announces-60-reduction-in-list-price-of-pcsk9-inhibitor-evolocumab. Accessed April 12, 2019.
31. Kaufman TM, Duell PB, Purnell JQ, et al. Application of PCSK9 inhibitors in practice: challenges and opportunities. Circ Res. 2017;121:499-501.
A new cholesterol guideline1 builds on the 2013 American College of Cardiology (ACC)/American Heart Association (AHA) cholesterol guidelines,2 which were a major paradigm shift in the evaluation and management of blood cholesterol levels and risk for atherosclerotic cardiovascular disease (ASCVD). The work was presented (and simultaneously published) on November 10, 2018, at the annual AHA Scientific Sessions in Chicago. Full text,1 an executive summary,3 and accompanying systematic review of evidence4 are available online.
The 2018 AHA/ACC cholesterol guideline represents a step forward in ASCVD prevention—especially in primary prevention, where it provides guidance for risk refinement and personalization. In this article, we mine the details of what has changed and what is new in this guideline so that you can prepare to adopt the recommendations in your practice.
2013 and 2018 guidelines: Similarities, differences
As in earlier iterations, the 2018 guideline emphasizes healthy lifestyle across the life-course as the basis of ASCVD prevention—as elaborated in the 2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk.5 In contrast to the 2013 guidelines,2 the 2018 guideline is more comprehensive and more personalized, focusing on risk assessment for individual patients, rather than simply providing population-based approaches. Moreover, the guideline isn’t limited to adults: It makes recommendations pertaining to children and adolescents.1
TABLE 11,2 compares the most important differences between the 2013 and 2018 guidelines.
The 2013 ACC/AHA guidelines eliminated low-density lipoprotein cholesterol (LDL-C) and non-high-density lipoprotein cholesterol (non-HDL-C)a goals of therapy and replaced them with the concept of 4 “statin benefit groups”—that is, patient populations for which clear evidence supports the role of statin therapy.4 In the 2018 guideline, statin benefit groups have been maintained, although without explicit use of this term.1
Primary prevention. Although no major changes in statin indications are made for patients with (1) established ASCVD (ie, for secondary prevention), (2) diabetes mellitus (DM) and who are 40 to 75 years of age, or (3) a primary LDL-C elevation ≥ 190 mg/dL, significant changes were made for primary prevention patients ages 40 to 75 years.1 ASCVD risk calculation using the 2013 pooled cohort equations (PCE) is still recommended4; however, risk estimation is refined by the use of specific so-called risk-enhancing factors (TABLE 21). In cases in which the risk decision remains uncertain, obtaining the coronary artery calcium (CAC) score (which we’ll describe shortly) using specialized computed tomography (CT) is advised to facilitate the shared physician–patient decision-making process.1
LDL-C and non-HDL-C thresholds. Although LDL-C and non-HDL-C goals are not overtly brought back from the 2002 National Cholesterol Education Program/Adult Treatment Panel guidelines,6 the new guideline does introduce LDL-C and non-HDL-C thresholds—levels at which adding nonstatin therapy can be considered, in contrast to previous goals to which therapy was titrated. Definitions of statin intensity remain the same: Moderate-intensity statin therapy is expected to reduce the LDL-C level by 30% to 50%; high-intensity statin therapy, by ≥ 50%.1 The intensity of statin therapy has been de-escalated in the intermediate-risk group, where previous guidelines advised high-intensity statin therapy,4 and replaced with moderate-intensity statin therapy (similar to 2016 US Preventive Services Task Force [USPSTF] recommendations7).
[polldaddy:10312157]
Continue to: Fasting vs nonfasting lipid profiles
Fasting vs nonfasting lipid profiles. In contrast to previous guidelines,2,8 which used fasting lipid profiles, nonfasting lipid profiles are now recommended for establishing a baseline LDL-C level and for ASCVD risk estimation for most patients—as long as the triglycerides (TG) level is < 400 mg/dL. When the calculated LDL-C level is < 70 mg/dL using the standard Friedewald formula, obtaining a direct LDL-C or a modified LDL-C estimate9 is deemed reasonable to improve accuracy. (The modified LDL-C can be estimated using The Johns Hopkins Hospital’s free “LDL Cholesterol Calculator” [www.hopkinsmedicine.org/apps/all-apps/ldl-cholesterol-calculator]).
A fasting lipid profile is still preferred for patients who have a family history of a lipid disorder. The definition of hypertriglyceridemia has been revised from a fasting TG level ≥ 150 mg/dL to a nonfasting or fasting TG level ≥ 175 mg/dL.1
Nonstatin add-on therapy. The new guideline supports the addition of nonstatin therapies to maximally tolerated statin therapy in patients who have established ASCVD or a primary LDL-C elevation ≥ 190 mg/dL when (1) the LDL-C level has not been reduced by the expected percentage (≥ 50% for high-intensity statin therapy) or (2) explicit LDL-C level thresholds have been met.1
The principal 2 groups of recommended nonstatins for which there is randomized, controlled trial evidence of cardiovascular benefit are (1) the cholesterol-absorbing agent ezetimibe10 and (2) the proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors evolocumab11 and alirocumab.12
AAFP’s guarded positions on the 2013 and 2018 guidelines
The American Academy of Family Physicians (AAFP) welcomed the patient-centered and outcome-oriented aspects of the 2013 ACC/AHA guidelines, endorsing them with 3 qualifications.13
- Many of the recommendations were based on expert opinion, not rigorous research results—in particular, not on the findings of randomized controlled trials (although key points are based on high-quality evidence).
- There were conflicts of interest disclosed for 15 members of the guidelines panel, including a vice chair.
- Validation of the PCE risk estimation tool was lacking.
Continue to: AAFP announced...
AAFP announced in March that it does not endorse the 2018 AHA/ACC guideline, asserting that (1) only a small portion of the recommendations, primarily focused on the addition of nonstatin therapy, were addressed by an independent systematic review and (2) many of the guideline recommendations are based on low-quality or insufficient evidence. AAFP nevertheless bestowed an “affirmation of value” designation on the guideline—meaning that it provides some benefit for family physicians’ practice without fulfilling all criteria for full endorsement.14
Detailed recommendations from the 2018 guideline
Lifestyle modification
When talking about ASCVD risk with patients, it is important to review current lifestyle habits (eg, diet, physical activity, weight or body mass index, and tobacco use). Subsequent to that conversation, a healthy lifestyle should be endorsed and relevant advice provided. In addition, patient-directed materials (eg, ACC’s CardioSmart [www.cardiosmart.org]; AHA’s Life’s Simple 7 [www.heart.org/en/professional/workplace-health/lifes-simple-7]; and the National Lipid Association’s Patient Tear Sheets [www.lipid.org/practicetools/tools/tearsheets] and Clinicians’ Lifestyle Modification Toolbox [www.lipid.org/CLMT]) and referrals (eg, to cardiac rehabilitation, a dietitian, a smoking-cessation program) should be provided.1
Primary prevention of ASCVD
Risk assessment for primary prevention is now approached as a process, rather than the simple risk calculation used in the 2013 ACC/AHA guidelines.2 Assessment involves risk estimation followed by risk personalization, which, in some cases, is followed by risk reclassification using CAC scoring.1
Patients are classified into 1 of 4 risk groups, based on the PCE1:
- low (< 5%)
- borderline (5%-7.5%)
- intermediate (7.5%-19.9%)
- high (≥ 20%).
However, the PCE-based risk score is a population-based tool, which might not reflect the actual risk of individual patients. In some populations, PCE underestimates ASCVD risk; in others, it overestimates risk. A central tenet of the new guideline is personalization of risk, taking into account the unique circumstances of each patient. Moreover, the new guideline provides guidance on how to interpret the PCE risk score for several different ethnic and racial groups.1
Continue to: Medical therapy
Medical therapy. The decision to start lipid-lowering therapy should be made after a physician–patient discussion that considers costs of therapy as well as patient preferences and values in the context of shared decision-making. Discussion should include a review of major risk factors (eg, cigarette smoking, elevated blood pressure, and the LDL-C level), the PCE risk score, the presence of risk-enhancing factors (TABLE 21), potential benefits of lifestyle changes and statin therapy, and the potential for adverse drug effects and drug–drug interactions.1
If the estimated ASCVD risk is 7.5%-19.9%, starting moderate-intensity statin therapy is recommended. Risk-enhancing factors favor initiation of statin therapy, even in patients at borderline risk (5%-7.5%). If risk is uncertain, the CAC score can be used to facilitate shared decision-making.1 The use of CAC is in agreement with the USPSTF statement that CAC can moderately improve discrimination and reclassification, but has an unclear effect on downstream health care utilization.15 Importantly, CAC should not be measured routinely in patients already taking a statin because its primary role is to facilitate shared decision-making regarding initiation of statin therapy.16
If the 10-year ASCVD risk is ≥ 20%, high-intensity statin therapy is advised, without need to obtain the CAC score. If high-intensity statin therapy is advisable but not acceptable to, or tolerated by, the patient, it might be reasonable to add a nonstatin drug (ezetimibe or a bile-acid sequestrant) to moderate-intensity statin therapy.1
Risk-enhancing factors (TABLE 21) apply to intermediate- and borderline-risk patients. Importantly, these factors include membership in specific ethnic groups, conditions specific to females, and male–female distinctions in risk. Risk-enhancing factors also incorporate biomarkers that are often measured by lipid specialists, such as lipoprotein(a) (Lp[a]) and apolipoprotein B (ApoB).1
Lp(a) is an atherogenic particle, akin to an LDL particle, that consists of a molecule of apolipoprotein (a) (a nonfunctional mimic of a portion of plasminogen) covalently bound to ApoB, like the one found on the LDL particle. Lp(a) is proportionally associated with an increased risk for ASCVD and aortic stenosis at a level > 50 mg/dL.17 A family history of premature ASCVD is a relative indication for measuring Lp(a).1
Continue to: When and why to measure CAC
When and why to measure CAC
If the decision to initiate statin therapy is still uncertain after risk estimation and personalization, or when a patient is undecided about committing to lifelong lipid-lowering therapy, the new guideline recommends obtaining a CAC score to inform the shared decision-making process.1,18 Measurement of CAC is obtained by noncontrast, electrocardiographic-gated CT that can be performed in 10 to 15 minutes, requiring approximately 1 millisievert of radiation (equivalent of the approximate dose absorbed during 2 mammograms). Although measurement of the CAC score is generally not covered by insurance, its cost ($50-$450) nationwide makes it accessible.19
CAC measures the presence (or absence) of subclinical atherosclerosis by detecting calcified plaque in coronary arteries. The absolute CAC score is expressed in Agatston units; an age–gender population percentile is also provided. Keep in mind that the presence of any CAC (ie, a score > 0) is abnormal and demonstrates the presence of subclinical coronary artery disease. The prevalence of CAC > 0 increases with age, but a significant percentage of older people have a CAC score = 0. When CAC > 0, additional information is provided by the distribution of plaque burden among the different coronary arteries.20
Among intermediate-risk patients, 50% have CAC = 0 and, therefore, a very low event rate over the ensuing 10 years, which allows statin therapy to be safely deferred unless certain risk factors are present (eg, family history, smoking, DM).1,18 It is reasonable to repeat CAC testing in 5 to 10 years to assess whether subclinical atherosclerosis has developed. The 2018 guideline emphasizes that, when the CAC score is > 0 but < 100 Agatston units, statin therapy is favored, especially in patients > 55 years of age; when the CAC score is ≥ 100 Agatston units or at the ≥ 75th percentile, statin therapy is indicated regardless of age.1
Patients who might benefit from knowing their CAC score include those who are:
- reluctant to initiate statin therapy but who want to understand their risk and potential for benefit more precisely
- concerned about the need to reinstitute statin therapy after discontinuing it because of statin-associated adverse effects
- older (men, 55-80 years; women, 60-80 years) who have a low burden of risk factors and who question whether they would benefit from statin therapy
- middle-aged (40-55 years) and who have a PCE-calculated risk of 5% to < 7.5% for ASCVD and factors that increase their risk for ASCVD, even though they are in a borderline-risk group.1
Primary prevention in special populations
Older patients. In adults ≥ 75 years who have an LDL-C level 70 to 189 mg/dL, initiating a moderate-intensity statin might be reasonable; however, it might also be reasonable to stop treatment in this population when physical or cognitive decline, multiple morbidities, frailty, or reduced life expectancy limits the potential benefit of statin therapy. It might be reasonable to use the CAC score in adults 76 to 80 years of age who have an LDL-C level of 70 to 189 mg/dL to reclassify those whose CAC score = 0, so that they can avoid statin therapy.1
Continue to: Children and adolescents
Children and adolescents. In alignment with current pediatric guidelines,21 but in contrast to USPSTF reccomendations,22 the 2018 ACC/AHA guideline endorses universal lipid screening for pediatric patients (see TABLE W11,21,22). It is reasonable to obtain a fasting lipid profile or nonfasting non-HDL-C in all children and adolescents who have neither cardiovascular risk factors nor a family history of early cardiovascular disease to detect moderate-to-severe lipid abnormalities. Screening should be done once at 9 to 11 years of age and again at 17 to 21 years.1
A screening test as early as 2 years of age to detect familial hypercholesterolemia (FH) is reasonable when a family history of either early CVD or significant hypercholesterolemia is present. The guideline endorses reverse cascade screening for detection of FH in family members of children and adolescents who have severe hypercholesterolemia.1
In children and adolescents with a lipid abnormality, especially when associated with the metabolic syndrome, lifestyle counseling is beneficial for lowering the LDL-C level. In children and adolescents ≥ 10 years of age with (1) an LDL-C level persistently ≥ 190 mg/dL or (2) an LDL level ≥ 160 mg/dL plus a clinical presentation consistent with FH, it is reasonable to initiate statin therapy if they do not respond adequately to 3 to 6 months of lifestyle therapy.1
Ethnicity as a risk-modifying factor. The PCE distinguishes between US adults of European ancestry and African ancestry, but no other ethnic groups are distinguished.4 The new guideline advocates for the use of PCE in other populations; however, it states that, for clinical decision-making purposes, it is reasonable, in adults of different races and ethnicities, for the physician to review racial and ethnic features that can influence ASCVD risk to allow adjustment of the choice of statin or intensity of treatment. Specifically, South Asian ancestry is now treated as a risk-enhancing factor, given the high prevalence of premature and extensive ASCVD in this patient population.1
Concerns specific to women. Considering conditions specific to women as potential risk-enhancing factors is advised when discussing lifestyle intervention and the potential for benefit from statin therapy—in particular, (1) in the setting of premature menopause (< 40 years) and (2) when there is a history of a pregnancy-associated disorder (eg, hypertension, preeclampsia, gestational DM, a small-for-gestational-age infant, and preterm delivery). If the decision is made to initiate statin therapy in women of childbearing age who are sexually active, there is a guideline mandate to counsel patients on using reliable contraception. When pregnancy is planned, statin therapy should be discontinued 1 to 2 months before pregnancy is attempted; when pregnancy occurs while a patient is taking a statin, therapy should be stopped as soon as the pregnancy is discovered.1
Continue to: Adults with chronic kidney disease
Adults with chronic kidney disease. Chronic kidney disease that is not treated with dialysis or kidney transplantation is considered a risk-enhancing factor; initiation of a moderate-intensity statin or a moderate-intensity statin plus ezetimibe can be useful in patients with chronic kidney disease who are 40 to 75 years of age and have an LDL-C level of 70 to 189 mg/dL and a PCE-calculated risk ≥ 7.5%. In adults with advanced kidney disease that requires dialysis who are already taking a statin, it may be reasonable to continue the statin; however, initiation of a statin in adults with advanced kidney disease who require dialysis is not recommended because of an apparent lack of benefit.1
Adults with a chronic inflammatory disorder or human immunodeficiency virus infection. Any of these conditions are treated as risk-enhancing factors; in a risk discussion with affected patients, therefore, moderate-intensity statin therapy or high-intensity statin therapy is favored for those 40 to 75 years of age who have an LDL-C level of 70 to 189 mg/dL and PCE-calculated risk ≥ 7.5%. A fasting lipid profile and assessment of ASCVD risk factors for these patients can be useful (1) as a guide to the potential benefit of statin therapy and (2) for monitoring or adjusting lipid-lowering drug therapy before, and 4 to 12 weeks after, starting inflammatory disease-modifying therapy or antiretroviral therapy.
In adults with rheumatoid arthritis who undergo ASCVD risk assessment with a lipid profile, it can be useful to recheck lipid values and other major ASCVD risk factors 2 to 4 months after the inflammatory disease has been controlled.1
Primary hypercholesterolemia
The diagnosis and management of heterozygous or homozygous familial hypercholesterolemia (HeFH or HoFH) is beyond the scope of the 2018 ACC/AHA cholesterol guidelines; instead, the 2015 AHA Scientific Statement, “The Agenda for Familial Hypercholesterolemia,” provides a contemporary review of these topics.23 However, the 2018 cholesterol guideline does acknowledge that an LDL-C level ≥ 190 mg/dL often corresponds to primary (ie, genetic) hypercholesterolemia.
In patients 20 to 75 years of age who have a primary elevation of LDL-C level ≥ 190 mg/dL, the guideline recommends initiation of high-intensity statin therapy without calculating ASCVD risk using the PCE. If a > 50% LDL-C reduction is not achieved, or if the LDL-C level on maximally tolerated statin therapy remains ≥ 100 mg/dL, adding ezetimibe is considered reasonable. If there is < 50% reduction in the LDL-C level while taking maximally tolerated statin and ezetimibe therapy, adding a bile-acid sequestrant can be considered, as long as the TG level is not > 300 mg/dL (ie, bile-acid sequestrants can elevate the TG level significantly).
Continue to: In patients 30 to 75 years of age...
In patients 30 to 75 years of age who have a diagnosis of HeFH and an LDL-C level ≥ 100 mg/dL while taking maximally tolerated statin and ezetimibe therapy, the addition of a PCSK9 inhibitor can be considered. Regardless of whether there is a diagnosis of HeFH, addition of a PCSK9 inhibitor can be considered in patients 40 to 75 years of age who have a baseline LDL-C level ≥ 220 mg/dL and who achieve an on-treatment LDL-C level ≥ 130 mg/dL while receiving maximally tolerated statin therapy and ezetimibe.1
Diabetes mellitus
In patients with DM who are 40 to 75 years of age, moderate-intensity statin therapy is recommended without calculating the 10-year ASCVD risk. When the LDL-C level is 70 to 189 mg/dL, however, it is reasonable to use the PCE to assess 10-year ASCVD risk to facilitate risk stratification.
In patients with DM who are at higher risk, especially those who have multiple risk factors or are 50 to 75 years of age, it is reasonable to use a high-intensity statin to reduce the LDL-C level by ≥ 50 %. In adults > 75 years of age with DM who are already on statin therapy, it is reasonable to continue statin therapy; for those that age who are not on statin therapy, it might be reasonable to initiate statin therapy after a physician–patient discussion of potential benefits and risks.
In adults with DM and PCE-calculated risk ≥ 20%, it might be reasonable to add ezetimibe to maximally tolerated statin therapy to reduce the LDL-C level by ≥ 50%. In adults 20 to 39 years of age with DM of long duration (≥ 10 years of type 2 DM, ≥ 20 years of type 1 DM), albuminuria (≥ 30 μg of albumin/mg creatinine), estimated glomerular filtration rate < 60 mL/min/1.73 m2, retinopathy, neuropathy, or ankle-brachial index < 0.9, it might be reasonable to initiate statin therapy.1
Secondary prevention
Presence of clinical ASCVD. In patients with clinical ASCVD who are ≤ 75 years of age, high-intensity statin therapy should be initiated or continued, with the aim of achieving ≥ 50% reduction in the LDL-C level. When high-intensity statin therapy is contraindicated or if a patient experiences statin-associated adverse effects, moderate-intensity statin therapy should be initiated or continued with the aim of achieving a 30% to 49% reduction in the LDL-C level.
Continue to: In patients...
In patients > 75 years of age with clinical ASCVD, it is reasonable to initiate or continue moderate- or high-intensity statin therapy after evaluation of the potential for ASCVD risk reduction, adverse effects, and drug–drug interactions, as well as patient frailty and patient preference.1
Very high risk. In patients at very high risk (this includes a history of multiple major ASCVD events or 1 major ASCVD event plus multiple high-risk conditions), maximally tolerated LDL-C-lowering therapy should include maximally tolerated statin therapy and ezetimibe before considering a PCSK9 inhibitor. An LDL-C level ≥ 70 mg/dL or a non-HDL-C level ≥ 100 mg/dL is considered a reasonable threshold for adding a PCSK9 inhibitor to background lipid-lowering therapy1 (TABLE 31).
Heart failure. In patients with heart failure who have (1) a reduced ejection fraction attributable to ischemic heart disease, (2) a reasonable life expectancy (3-5 years), and (3) are not already on a statin because of ASCVD, consider initiating moderate-intensity statin therapy to reduce the risk for an ASCVD event.1
Reduction of elevated triglycerides
The guideline defines moderate hypertriglyceridemia as a nonfasting or fasting TG level of 175 to 499 mg/dL. Such a finding is considered a risk-enhancing factor and is 1 of 5 components of the metabolic syndrome. Three independent measurements are advised to diagnose primary moderate hypertriglyceridemia. Severe hypertriglyceridemia is diagnosed when the fasting TG level is ≥ 500 mg/dL.1
In moderate hypertriglyceridemia, most TGs are carried in very-low-density lipoprotein particles; in severe hypertriglyceridemia, on the other hand, chylomicrons predominate, raising the risk for pancreatitis. In adults with severe hypertriglyceridemia, therefore—especially when the fasting TG level is ≥ 1000 mg/dL—it is reasonable to identify and address other causes of hypertriglyceridemia. If TGs are persistently elevated or increasing, levels should be reduced to prevent acute pancreatitis with a very low-fat diet and by avoiding refined carbohydrates and alcohol; consuming omega-3 fatty acids; and, if necessary, taking a fibrate.1
Continue to: In adults...
In adults ≥ 20 years of age with moderate hypertriglyceridemia, lifestyle factors (eg, obesity, metabolic syndrome), secondary factors (eg, DM, chronic liver or kidney disease, nephrotic syndrome, hypothyroidism), and medications that increase the TG level need to be addressed first. In adults 40 to 75 years of age with moderate or severe hypertriglyceridemia and a PCE-calculated ASCVD risk ≥ 7.5%, it is reasonable to reevaluate risk after lifestyle and secondary factors are addressed and to consider a persistently elevated TG level as a factor favoring initiation or intensification of statin therapy. In adults 40 to 75 years of age with severe hypertriglyceridemia and ASCVD risk ≥ 7.5%, it is reasonable to address reversible causes of a high TG level and to initiate statin therapy.1
Other considerations in cholesterol management
Tools to assess adherence
The response to lifestyle and statin therapy should be evaluated by the percentage reduction in the LDL-C level compared with baseline, not by assessment of the absolute LDL-C level. When seeing a patient whose treatment is ongoing, a baseline level can be estimated using a desktop LDL-calculator app.
Adherence and percentage response to LDL-C–lowering medications and lifestyle changes should be evaluated with repeat lipid measurement 4 to 12 weeks after either a statin is initiated or the dosage is adjusted, and repeated every 3 to 12 months as needed. In patients with established ASCVD who are at very high risk, triggers for adding nonstatin therapy are defined by a threshold LDL-C level ≥ 70 mg/dL on maximal statin therapy.1
Interventions focused on improving adherence to prescribed therapy are recommended for management of adults with an elevated cholesterol level. These interventions include telephone reminders, calendar reminders, integrated multidisciplinary educational activities, and pharmacist-led interventions, such as simplification of the medication regimen to once-daily dosing.1
Statin safety and associated adverse effects
A physician–patient risk discussion is recommended before initiating statin therapy to review net clinical benefit, during which the 2 parties weigh the potential for ASCVD risk reduction against the potential for statin-associated adverse effects, statin–drug interactions, and safety, with the physician emphasizing that adverse effects can be addressed successfully.
Continue to: Statins are one of...
Statins are one of the safest classes of medication, with an excellent risk-benefit ratio. However, there are myriad confusing media reports regarding potential adverse effects and safety of the statin class—reports that often lead patients to discontinue or refuse statins.
Statin-associated adverse effects include the common statin-associated muscle symptoms (SAMS), new-onset DM, cognitive effects, and hepatic injury. The frequency of new-onset DM depends on the population exposed to statins, with a higher incidence of new-onset DM found in patients who are already predisposed, such as those with obesity, prediabetes, and metabolic syndrome. Cognitive effects are rare and difficult to interpret; they were not reported in the large statin mega-trials but have been described in case reports. Significant transaminase elevations > 3 times the upper limit of normal are infrequent; hepatic failure with statins is extremely rare and found at the same incidence in the general population.1
SAMS include (in order of decreasing prevalence)24:
- myalgias with a normal creatine kinase (CK) level
- conditions such as myositis or myopathy (elevated CK level)
- rhabdomyolysis (CK level > 10 times the upper limit of normal, plus renal injury)
- extremely rare statin-associated autoimmune myopathy, with detectable 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase antibodies.
In patients with SAMS, thorough assessment of symptoms is recommended, in addition to evaluation for nonstatin causes and predisposing factors. Identification of potential SAMS-predisposing factors is recommended before initiation of treatment, including demographics (eg, East-Asian ancestry), comorbid conditions (eg, hypothyroidism and vitamin D deficiency), and use of medications adversely affecting statin metabolism (eg, cyclosporine).
In patients with statin-associated adverse effects that are not severe, it is recommended to reassess and rechallenge to achieve a maximal lowering of the LDL-C level by a modified dosing regimen or an alternate statin or by combining a statin with nonstatin therapy. In patients with increased risk for DM or new-onset DM, it is recommended to continue statin therapy.
Continue to: Routine CK and liver function testing...
Routine CK and liver function testing is not useful in patients treated with statins; however, it is recommended that CK be measured in patients with severe SAMS or objective muscle weakness, or both, and to measure liver function if symptoms suggest hepatotoxicity. In patients at increased risk for ASCVD who have chronic, stable liver disease (including non-alcoholic fatty liver disease), it is reasonable, when appropriately indicated, to use statins after obtaining baseline measurements and determining a schedule of monitoring and safety checks.
In patients at increased risk for ASCVD who have severe or recurrent SAMS after appropriate statin rechallenge, it is reasonable to use nonstatin therapy that is likely to provide net clinical benefit. The guideline does not recommend routine use of coenzyme Q10 supplementation for the treatment or prevention of SAMS.1
Guideline criticism
Guideline development is challenging on multiple levels, including balancing perspectives from multiple stakeholders. Nevertheless, the 2018 AHA/ACC cholesterol guideline builds nicely on progress made since its 2013 predecessor was released.4 This document was developed with the participation of representatives from 10 professional societies in addition to the ACC and AHA—notably, the National Lipid Association and American Society for Preventive Cardiology.1
To refine risk estimation and facilitate shared decision-making, the new guideline introduced so-called risk-enhancing factors and use of the CAC.1 However, some potential risk-enhancing factors were left out: erectile dysfunction, for example, often a marker of increased cardiovascular risk in men < 50 years of age.25 In addition, although pretreatment ApoB was introduced as a risk-enhancing factor,1 no recommendation is given to measure ApoB after initiation of therapy for evaluation of residual cardiovascular risk, as endorsed in other guidelines.26,27
Moreover, the guideline does not include the “extreme risk” category in the guideline developed by the American Association of Clinical Endocrinologists (AACE).28 Although the 2018 AHA/ACC guideline introduces < 70 mg/dL and < 100 mg/dL LDL-C thresholds,1 the < 55 mg/dL LDL-C threshold used for patients in the AACE/American College of Endocrinology extreme-risk category is not mentioned.26 This omission might leave patients who are at extreme ASCVD risk without optimal lipid-lowering therapy. Similarly, the guideline does not elaborate on the diagnosis and treatment of HoFH and HeFH.1 The age cutoff of 30 years for the recommendation to consider PCSK9 inhibitors in patients with HeFH appears arbitrary and excludes younger FH patients who have an extreme LDL-C elevation from potentially important therapy.23
Continue to: Guidelines are dynamic instruments...
Guidelines are dynamic instruments that require constant updating, given the production of new evidence. In fact, the results of the Reduction of Cardiovascular Events With Icosapent Ethyl-Intervention Trial (REDUCE-IT) were presented at the same meeting at which this guideline was unveiled.29 REDUCE-IT demonstrated an astonishing highly significant 25% reduction in the composite primary major adverse cardiovascular event outcome in patients with an LDL-C level of 44 to 100 mg/dL on statin therapy, who had a TG level of 135 to 499 mg/dL and had been treated for a median of 4.9 years with 4 g of pure eicosapentaenoic acid.
In addition, the guideline’s value statements, which address the need to consider the cost of drugs in determining most appropriate treatment, are no longer accurate because the price of PCSK9 inhibitors has dropped by more than half since the guideline was issued.30
An upward climb to clinical payoff
Even after close study of the 2018 AHA/ACC cholesterol guideline, implementing it in practice might remain a challenge to clinicians who are inexperienced in ordering lipid markers such as Lp(a) and interpreting the CAC score. Moreover, initiating and monitoring nonstatin therapies will be a demanding task—especially with PCSK9 inhibitors, which present access difficulties because they are relatively expensive (even after the recent price cut). That’s why, when there is doubt in the mind of the physician or other provider, we will likely see more referrals to specialists in lipid management and ASCVD risk estimation to optimize preventive therapy.31
CORRESPONDENCE
Cezary Wójcik, MD, PhD, FNLA, Oregon Health & Science University, 3181 SW Sam Jackson Park Rd, Portland, OR 97239; [email protected]
A new cholesterol guideline1 builds on the 2013 American College of Cardiology (ACC)/American Heart Association (AHA) cholesterol guidelines,2 which were a major paradigm shift in the evaluation and management of blood cholesterol levels and risk for atherosclerotic cardiovascular disease (ASCVD). The work was presented (and simultaneously published) on November 10, 2018, at the annual AHA Scientific Sessions in Chicago. Full text,1 an executive summary,3 and accompanying systematic review of evidence4 are available online.
The 2018 AHA/ACC cholesterol guideline represents a step forward in ASCVD prevention—especially in primary prevention, where it provides guidance for risk refinement and personalization. In this article, we mine the details of what has changed and what is new in this guideline so that you can prepare to adopt the recommendations in your practice.
2013 and 2018 guidelines: Similarities, differences
As in earlier iterations, the 2018 guideline emphasizes healthy lifestyle across the life-course as the basis of ASCVD prevention—as elaborated in the 2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk.5 In contrast to the 2013 guidelines,2 the 2018 guideline is more comprehensive and more personalized, focusing on risk assessment for individual patients, rather than simply providing population-based approaches. Moreover, the guideline isn’t limited to adults: It makes recommendations pertaining to children and adolescents.1
TABLE 11,2 compares the most important differences between the 2013 and 2018 guidelines.
The 2013 ACC/AHA guidelines eliminated low-density lipoprotein cholesterol (LDL-C) and non-high-density lipoprotein cholesterol (non-HDL-C)a goals of therapy and replaced them with the concept of 4 “statin benefit groups”—that is, patient populations for which clear evidence supports the role of statin therapy.4 In the 2018 guideline, statin benefit groups have been maintained, although without explicit use of this term.1
Primary prevention. Although no major changes in statin indications are made for patients with (1) established ASCVD (ie, for secondary prevention), (2) diabetes mellitus (DM) and who are 40 to 75 years of age, or (3) a primary LDL-C elevation ≥ 190 mg/dL, significant changes were made for primary prevention patients ages 40 to 75 years.1 ASCVD risk calculation using the 2013 pooled cohort equations (PCE) is still recommended4; however, risk estimation is refined by the use of specific so-called risk-enhancing factors (TABLE 21). In cases in which the risk decision remains uncertain, obtaining the coronary artery calcium (CAC) score (which we’ll describe shortly) using specialized computed tomography (CT) is advised to facilitate the shared physician–patient decision-making process.1
LDL-C and non-HDL-C thresholds. Although LDL-C and non-HDL-C goals are not overtly brought back from the 2002 National Cholesterol Education Program/Adult Treatment Panel guidelines,6 the new guideline does introduce LDL-C and non-HDL-C thresholds—levels at which adding nonstatin therapy can be considered, in contrast to previous goals to which therapy was titrated. Definitions of statin intensity remain the same: Moderate-intensity statin therapy is expected to reduce the LDL-C level by 30% to 50%; high-intensity statin therapy, by ≥ 50%.1 The intensity of statin therapy has been de-escalated in the intermediate-risk group, where previous guidelines advised high-intensity statin therapy,4 and replaced with moderate-intensity statin therapy (similar to 2016 US Preventive Services Task Force [USPSTF] recommendations7).
[polldaddy:10312157]
Continue to: Fasting vs nonfasting lipid profiles
Fasting vs nonfasting lipid profiles. In contrast to previous guidelines,2,8 which used fasting lipid profiles, nonfasting lipid profiles are now recommended for establishing a baseline LDL-C level and for ASCVD risk estimation for most patients—as long as the triglycerides (TG) level is < 400 mg/dL. When the calculated LDL-C level is < 70 mg/dL using the standard Friedewald formula, obtaining a direct LDL-C or a modified LDL-C estimate9 is deemed reasonable to improve accuracy. (The modified LDL-C can be estimated using The Johns Hopkins Hospital’s free “LDL Cholesterol Calculator” [www.hopkinsmedicine.org/apps/all-apps/ldl-cholesterol-calculator]).
A fasting lipid profile is still preferred for patients who have a family history of a lipid disorder. The definition of hypertriglyceridemia has been revised from a fasting TG level ≥ 150 mg/dL to a nonfasting or fasting TG level ≥ 175 mg/dL.1
Nonstatin add-on therapy. The new guideline supports the addition of nonstatin therapies to maximally tolerated statin therapy in patients who have established ASCVD or a primary LDL-C elevation ≥ 190 mg/dL when (1) the LDL-C level has not been reduced by the expected percentage (≥ 50% for high-intensity statin therapy) or (2) explicit LDL-C level thresholds have been met.1
The principal 2 groups of recommended nonstatins for which there is randomized, controlled trial evidence of cardiovascular benefit are (1) the cholesterol-absorbing agent ezetimibe10 and (2) the proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors evolocumab11 and alirocumab.12
AAFP’s guarded positions on the 2013 and 2018 guidelines
The American Academy of Family Physicians (AAFP) welcomed the patient-centered and outcome-oriented aspects of the 2013 ACC/AHA guidelines, endorsing them with 3 qualifications.13
- Many of the recommendations were based on expert opinion, not rigorous research results—in particular, not on the findings of randomized controlled trials (although key points are based on high-quality evidence).
- There were conflicts of interest disclosed for 15 members of the guidelines panel, including a vice chair.
- Validation of the PCE risk estimation tool was lacking.
Continue to: AAFP announced...
AAFP announced in March that it does not endorse the 2018 AHA/ACC guideline, asserting that (1) only a small portion of the recommendations, primarily focused on the addition of nonstatin therapy, were addressed by an independent systematic review and (2) many of the guideline recommendations are based on low-quality or insufficient evidence. AAFP nevertheless bestowed an “affirmation of value” designation on the guideline—meaning that it provides some benefit for family physicians’ practice without fulfilling all criteria for full endorsement.14
Detailed recommendations from the 2018 guideline
Lifestyle modification
When talking about ASCVD risk with patients, it is important to review current lifestyle habits (eg, diet, physical activity, weight or body mass index, and tobacco use). Subsequent to that conversation, a healthy lifestyle should be endorsed and relevant advice provided. In addition, patient-directed materials (eg, ACC’s CardioSmart [www.cardiosmart.org]; AHA’s Life’s Simple 7 [www.heart.org/en/professional/workplace-health/lifes-simple-7]; and the National Lipid Association’s Patient Tear Sheets [www.lipid.org/practicetools/tools/tearsheets] and Clinicians’ Lifestyle Modification Toolbox [www.lipid.org/CLMT]) and referrals (eg, to cardiac rehabilitation, a dietitian, a smoking-cessation program) should be provided.1
Primary prevention of ASCVD
Risk assessment for primary prevention is now approached as a process, rather than the simple risk calculation used in the 2013 ACC/AHA guidelines.2 Assessment involves risk estimation followed by risk personalization, which, in some cases, is followed by risk reclassification using CAC scoring.1
Patients are classified into 1 of 4 risk groups, based on the PCE1:
- low (< 5%)
- borderline (5%-7.5%)
- intermediate (7.5%-19.9%)
- high (≥ 20%).
However, the PCE-based risk score is a population-based tool, which might not reflect the actual risk of individual patients. In some populations, PCE underestimates ASCVD risk; in others, it overestimates risk. A central tenet of the new guideline is personalization of risk, taking into account the unique circumstances of each patient. Moreover, the new guideline provides guidance on how to interpret the PCE risk score for several different ethnic and racial groups.1
Continue to: Medical therapy
Medical therapy. The decision to start lipid-lowering therapy should be made after a physician–patient discussion that considers costs of therapy as well as patient preferences and values in the context of shared decision-making. Discussion should include a review of major risk factors (eg, cigarette smoking, elevated blood pressure, and the LDL-C level), the PCE risk score, the presence of risk-enhancing factors (TABLE 21), potential benefits of lifestyle changes and statin therapy, and the potential for adverse drug effects and drug–drug interactions.1
If the estimated ASCVD risk is 7.5%-19.9%, starting moderate-intensity statin therapy is recommended. Risk-enhancing factors favor initiation of statin therapy, even in patients at borderline risk (5%-7.5%). If risk is uncertain, the CAC score can be used to facilitate shared decision-making.1 The use of CAC is in agreement with the USPSTF statement that CAC can moderately improve discrimination and reclassification, but has an unclear effect on downstream health care utilization.15 Importantly, CAC should not be measured routinely in patients already taking a statin because its primary role is to facilitate shared decision-making regarding initiation of statin therapy.16
If the 10-year ASCVD risk is ≥ 20%, high-intensity statin therapy is advised, without need to obtain the CAC score. If high-intensity statin therapy is advisable but not acceptable to, or tolerated by, the patient, it might be reasonable to add a nonstatin drug (ezetimibe or a bile-acid sequestrant) to moderate-intensity statin therapy.1
Risk-enhancing factors (TABLE 21) apply to intermediate- and borderline-risk patients. Importantly, these factors include membership in specific ethnic groups, conditions specific to females, and male–female distinctions in risk. Risk-enhancing factors also incorporate biomarkers that are often measured by lipid specialists, such as lipoprotein(a) (Lp[a]) and apolipoprotein B (ApoB).1
Lp(a) is an atherogenic particle, akin to an LDL particle, that consists of a molecule of apolipoprotein (a) (a nonfunctional mimic of a portion of plasminogen) covalently bound to ApoB, like the one found on the LDL particle. Lp(a) is proportionally associated with an increased risk for ASCVD and aortic stenosis at a level > 50 mg/dL.17 A family history of premature ASCVD is a relative indication for measuring Lp(a).1
Continue to: When and why to measure CAC
When and why to measure CAC
If the decision to initiate statin therapy is still uncertain after risk estimation and personalization, or when a patient is undecided about committing to lifelong lipid-lowering therapy, the new guideline recommends obtaining a CAC score to inform the shared decision-making process.1,18 Measurement of CAC is obtained by noncontrast, electrocardiographic-gated CT that can be performed in 10 to 15 minutes, requiring approximately 1 millisievert of radiation (equivalent of the approximate dose absorbed during 2 mammograms). Although measurement of the CAC score is generally not covered by insurance, its cost ($50-$450) nationwide makes it accessible.19
CAC measures the presence (or absence) of subclinical atherosclerosis by detecting calcified plaque in coronary arteries. The absolute CAC score is expressed in Agatston units; an age–gender population percentile is also provided. Keep in mind that the presence of any CAC (ie, a score > 0) is abnormal and demonstrates the presence of subclinical coronary artery disease. The prevalence of CAC > 0 increases with age, but a significant percentage of older people have a CAC score = 0. When CAC > 0, additional information is provided by the distribution of plaque burden among the different coronary arteries.20
Among intermediate-risk patients, 50% have CAC = 0 and, therefore, a very low event rate over the ensuing 10 years, which allows statin therapy to be safely deferred unless certain risk factors are present (eg, family history, smoking, DM).1,18 It is reasonable to repeat CAC testing in 5 to 10 years to assess whether subclinical atherosclerosis has developed. The 2018 guideline emphasizes that, when the CAC score is > 0 but < 100 Agatston units, statin therapy is favored, especially in patients > 55 years of age; when the CAC score is ≥ 100 Agatston units or at the ≥ 75th percentile, statin therapy is indicated regardless of age.1
Patients who might benefit from knowing their CAC score include those who are:
- reluctant to initiate statin therapy but who want to understand their risk and potential for benefit more precisely
- concerned about the need to reinstitute statin therapy after discontinuing it because of statin-associated adverse effects
- older (men, 55-80 years; women, 60-80 years) who have a low burden of risk factors and who question whether they would benefit from statin therapy
- middle-aged (40-55 years) and who have a PCE-calculated risk of 5% to < 7.5% for ASCVD and factors that increase their risk for ASCVD, even though they are in a borderline-risk group.1
Primary prevention in special populations
Older patients. In adults ≥ 75 years who have an LDL-C level 70 to 189 mg/dL, initiating a moderate-intensity statin might be reasonable; however, it might also be reasonable to stop treatment in this population when physical or cognitive decline, multiple morbidities, frailty, or reduced life expectancy limits the potential benefit of statin therapy. It might be reasonable to use the CAC score in adults 76 to 80 years of age who have an LDL-C level of 70 to 189 mg/dL to reclassify those whose CAC score = 0, so that they can avoid statin therapy.1
Continue to: Children and adolescents
Children and adolescents. In alignment with current pediatric guidelines,21 but in contrast to USPSTF reccomendations,22 the 2018 ACC/AHA guideline endorses universal lipid screening for pediatric patients (see TABLE W11,21,22). It is reasonable to obtain a fasting lipid profile or nonfasting non-HDL-C in all children and adolescents who have neither cardiovascular risk factors nor a family history of early cardiovascular disease to detect moderate-to-severe lipid abnormalities. Screening should be done once at 9 to 11 years of age and again at 17 to 21 years.1
A screening test as early as 2 years of age to detect familial hypercholesterolemia (FH) is reasonable when a family history of either early CVD or significant hypercholesterolemia is present. The guideline endorses reverse cascade screening for detection of FH in family members of children and adolescents who have severe hypercholesterolemia.1
In children and adolescents with a lipid abnormality, especially when associated with the metabolic syndrome, lifestyle counseling is beneficial for lowering the LDL-C level. In children and adolescents ≥ 10 years of age with (1) an LDL-C level persistently ≥ 190 mg/dL or (2) an LDL level ≥ 160 mg/dL plus a clinical presentation consistent with FH, it is reasonable to initiate statin therapy if they do not respond adequately to 3 to 6 months of lifestyle therapy.1
Ethnicity as a risk-modifying factor. The PCE distinguishes between US adults of European ancestry and African ancestry, but no other ethnic groups are distinguished.4 The new guideline advocates for the use of PCE in other populations; however, it states that, for clinical decision-making purposes, it is reasonable, in adults of different races and ethnicities, for the physician to review racial and ethnic features that can influence ASCVD risk to allow adjustment of the choice of statin or intensity of treatment. Specifically, South Asian ancestry is now treated as a risk-enhancing factor, given the high prevalence of premature and extensive ASCVD in this patient population.1
Concerns specific to women. Considering conditions specific to women as potential risk-enhancing factors is advised when discussing lifestyle intervention and the potential for benefit from statin therapy—in particular, (1) in the setting of premature menopause (< 40 years) and (2) when there is a history of a pregnancy-associated disorder (eg, hypertension, preeclampsia, gestational DM, a small-for-gestational-age infant, and preterm delivery). If the decision is made to initiate statin therapy in women of childbearing age who are sexually active, there is a guideline mandate to counsel patients on using reliable contraception. When pregnancy is planned, statin therapy should be discontinued 1 to 2 months before pregnancy is attempted; when pregnancy occurs while a patient is taking a statin, therapy should be stopped as soon as the pregnancy is discovered.1
Continue to: Adults with chronic kidney disease
Adults with chronic kidney disease. Chronic kidney disease that is not treated with dialysis or kidney transplantation is considered a risk-enhancing factor; initiation of a moderate-intensity statin or a moderate-intensity statin plus ezetimibe can be useful in patients with chronic kidney disease who are 40 to 75 years of age and have an LDL-C level of 70 to 189 mg/dL and a PCE-calculated risk ≥ 7.5%. In adults with advanced kidney disease that requires dialysis who are already taking a statin, it may be reasonable to continue the statin; however, initiation of a statin in adults with advanced kidney disease who require dialysis is not recommended because of an apparent lack of benefit.1
Adults with a chronic inflammatory disorder or human immunodeficiency virus infection. Any of these conditions are treated as risk-enhancing factors; in a risk discussion with affected patients, therefore, moderate-intensity statin therapy or high-intensity statin therapy is favored for those 40 to 75 years of age who have an LDL-C level of 70 to 189 mg/dL and PCE-calculated risk ≥ 7.5%. A fasting lipid profile and assessment of ASCVD risk factors for these patients can be useful (1) as a guide to the potential benefit of statin therapy and (2) for monitoring or adjusting lipid-lowering drug therapy before, and 4 to 12 weeks after, starting inflammatory disease-modifying therapy or antiretroviral therapy.
In adults with rheumatoid arthritis who undergo ASCVD risk assessment with a lipid profile, it can be useful to recheck lipid values and other major ASCVD risk factors 2 to 4 months after the inflammatory disease has been controlled.1
Primary hypercholesterolemia
The diagnosis and management of heterozygous or homozygous familial hypercholesterolemia (HeFH or HoFH) is beyond the scope of the 2018 ACC/AHA cholesterol guidelines; instead, the 2015 AHA Scientific Statement, “The Agenda for Familial Hypercholesterolemia,” provides a contemporary review of these topics.23 However, the 2018 cholesterol guideline does acknowledge that an LDL-C level ≥ 190 mg/dL often corresponds to primary (ie, genetic) hypercholesterolemia.
In patients 20 to 75 years of age who have a primary elevation of LDL-C level ≥ 190 mg/dL, the guideline recommends initiation of high-intensity statin therapy without calculating ASCVD risk using the PCE. If a > 50% LDL-C reduction is not achieved, or if the LDL-C level on maximally tolerated statin therapy remains ≥ 100 mg/dL, adding ezetimibe is considered reasonable. If there is < 50% reduction in the LDL-C level while taking maximally tolerated statin and ezetimibe therapy, adding a bile-acid sequestrant can be considered, as long as the TG level is not > 300 mg/dL (ie, bile-acid sequestrants can elevate the TG level significantly).
Continue to: In patients 30 to 75 years of age...
In patients 30 to 75 years of age who have a diagnosis of HeFH and an LDL-C level ≥ 100 mg/dL while taking maximally tolerated statin and ezetimibe therapy, the addition of a PCSK9 inhibitor can be considered. Regardless of whether there is a diagnosis of HeFH, addition of a PCSK9 inhibitor can be considered in patients 40 to 75 years of age who have a baseline LDL-C level ≥ 220 mg/dL and who achieve an on-treatment LDL-C level ≥ 130 mg/dL while receiving maximally tolerated statin therapy and ezetimibe.1
Diabetes mellitus
In patients with DM who are 40 to 75 years of age, moderate-intensity statin therapy is recommended without calculating the 10-year ASCVD risk. When the LDL-C level is 70 to 189 mg/dL, however, it is reasonable to use the PCE to assess 10-year ASCVD risk to facilitate risk stratification.
In patients with DM who are at higher risk, especially those who have multiple risk factors or are 50 to 75 years of age, it is reasonable to use a high-intensity statin to reduce the LDL-C level by ≥ 50 %. In adults > 75 years of age with DM who are already on statin therapy, it is reasonable to continue statin therapy; for those that age who are not on statin therapy, it might be reasonable to initiate statin therapy after a physician–patient discussion of potential benefits and risks.
In adults with DM and PCE-calculated risk ≥ 20%, it might be reasonable to add ezetimibe to maximally tolerated statin therapy to reduce the LDL-C level by ≥ 50%. In adults 20 to 39 years of age with DM of long duration (≥ 10 years of type 2 DM, ≥ 20 years of type 1 DM), albuminuria (≥ 30 μg of albumin/mg creatinine), estimated glomerular filtration rate < 60 mL/min/1.73 m2, retinopathy, neuropathy, or ankle-brachial index < 0.9, it might be reasonable to initiate statin therapy.1
Secondary prevention
Presence of clinical ASCVD. In patients with clinical ASCVD who are ≤ 75 years of age, high-intensity statin therapy should be initiated or continued, with the aim of achieving ≥ 50% reduction in the LDL-C level. When high-intensity statin therapy is contraindicated or if a patient experiences statin-associated adverse effects, moderate-intensity statin therapy should be initiated or continued with the aim of achieving a 30% to 49% reduction in the LDL-C level.
Continue to: In patients...
In patients > 75 years of age with clinical ASCVD, it is reasonable to initiate or continue moderate- or high-intensity statin therapy after evaluation of the potential for ASCVD risk reduction, adverse effects, and drug–drug interactions, as well as patient frailty and patient preference.1
Very high risk. In patients at very high risk (this includes a history of multiple major ASCVD events or 1 major ASCVD event plus multiple high-risk conditions), maximally tolerated LDL-C-lowering therapy should include maximally tolerated statin therapy and ezetimibe before considering a PCSK9 inhibitor. An LDL-C level ≥ 70 mg/dL or a non-HDL-C level ≥ 100 mg/dL is considered a reasonable threshold for adding a PCSK9 inhibitor to background lipid-lowering therapy1 (TABLE 31).
Heart failure. In patients with heart failure who have (1) a reduced ejection fraction attributable to ischemic heart disease, (2) a reasonable life expectancy (3-5 years), and (3) are not already on a statin because of ASCVD, consider initiating moderate-intensity statin therapy to reduce the risk for an ASCVD event.1
Reduction of elevated triglycerides
The guideline defines moderate hypertriglyceridemia as a nonfasting or fasting TG level of 175 to 499 mg/dL. Such a finding is considered a risk-enhancing factor and is 1 of 5 components of the metabolic syndrome. Three independent measurements are advised to diagnose primary moderate hypertriglyceridemia. Severe hypertriglyceridemia is diagnosed when the fasting TG level is ≥ 500 mg/dL.1
In moderate hypertriglyceridemia, most TGs are carried in very-low-density lipoprotein particles; in severe hypertriglyceridemia, on the other hand, chylomicrons predominate, raising the risk for pancreatitis. In adults with severe hypertriglyceridemia, therefore—especially when the fasting TG level is ≥ 1000 mg/dL—it is reasonable to identify and address other causes of hypertriglyceridemia. If TGs are persistently elevated or increasing, levels should be reduced to prevent acute pancreatitis with a very low-fat diet and by avoiding refined carbohydrates and alcohol; consuming omega-3 fatty acids; and, if necessary, taking a fibrate.1
Continue to: In adults...
In adults ≥ 20 years of age with moderate hypertriglyceridemia, lifestyle factors (eg, obesity, metabolic syndrome), secondary factors (eg, DM, chronic liver or kidney disease, nephrotic syndrome, hypothyroidism), and medications that increase the TG level need to be addressed first. In adults 40 to 75 years of age with moderate or severe hypertriglyceridemia and a PCE-calculated ASCVD risk ≥ 7.5%, it is reasonable to reevaluate risk after lifestyle and secondary factors are addressed and to consider a persistently elevated TG level as a factor favoring initiation or intensification of statin therapy. In adults 40 to 75 years of age with severe hypertriglyceridemia and ASCVD risk ≥ 7.5%, it is reasonable to address reversible causes of a high TG level and to initiate statin therapy.1
Other considerations in cholesterol management
Tools to assess adherence
The response to lifestyle and statin therapy should be evaluated by the percentage reduction in the LDL-C level compared with baseline, not by assessment of the absolute LDL-C level. When seeing a patient whose treatment is ongoing, a baseline level can be estimated using a desktop LDL-calculator app.
Adherence and percentage response to LDL-C–lowering medications and lifestyle changes should be evaluated with repeat lipid measurement 4 to 12 weeks after either a statin is initiated or the dosage is adjusted, and repeated every 3 to 12 months as needed. In patients with established ASCVD who are at very high risk, triggers for adding nonstatin therapy are defined by a threshold LDL-C level ≥ 70 mg/dL on maximal statin therapy.1
Interventions focused on improving adherence to prescribed therapy are recommended for management of adults with an elevated cholesterol level. These interventions include telephone reminders, calendar reminders, integrated multidisciplinary educational activities, and pharmacist-led interventions, such as simplification of the medication regimen to once-daily dosing.1
Statin safety and associated adverse effects
A physician–patient risk discussion is recommended before initiating statin therapy to review net clinical benefit, during which the 2 parties weigh the potential for ASCVD risk reduction against the potential for statin-associated adverse effects, statin–drug interactions, and safety, with the physician emphasizing that adverse effects can be addressed successfully.
Continue to: Statins are one of...
Statins are one of the safest classes of medication, with an excellent risk-benefit ratio. However, there are myriad confusing media reports regarding potential adverse effects and safety of the statin class—reports that often lead patients to discontinue or refuse statins.
Statin-associated adverse effects include the common statin-associated muscle symptoms (SAMS), new-onset DM, cognitive effects, and hepatic injury. The frequency of new-onset DM depends on the population exposed to statins, with a higher incidence of new-onset DM found in patients who are already predisposed, such as those with obesity, prediabetes, and metabolic syndrome. Cognitive effects are rare and difficult to interpret; they were not reported in the large statin mega-trials but have been described in case reports. Significant transaminase elevations > 3 times the upper limit of normal are infrequent; hepatic failure with statins is extremely rare and found at the same incidence in the general population.1
SAMS include (in order of decreasing prevalence)24:
- myalgias with a normal creatine kinase (CK) level
- conditions such as myositis or myopathy (elevated CK level)
- rhabdomyolysis (CK level > 10 times the upper limit of normal, plus renal injury)
- extremely rare statin-associated autoimmune myopathy, with detectable 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase antibodies.
In patients with SAMS, thorough assessment of symptoms is recommended, in addition to evaluation for nonstatin causes and predisposing factors. Identification of potential SAMS-predisposing factors is recommended before initiation of treatment, including demographics (eg, East-Asian ancestry), comorbid conditions (eg, hypothyroidism and vitamin D deficiency), and use of medications adversely affecting statin metabolism (eg, cyclosporine).
In patients with statin-associated adverse effects that are not severe, it is recommended to reassess and rechallenge to achieve a maximal lowering of the LDL-C level by a modified dosing regimen or an alternate statin or by combining a statin with nonstatin therapy. In patients with increased risk for DM or new-onset DM, it is recommended to continue statin therapy.
Continue to: Routine CK and liver function testing...
Routine CK and liver function testing is not useful in patients treated with statins; however, it is recommended that CK be measured in patients with severe SAMS or objective muscle weakness, or both, and to measure liver function if symptoms suggest hepatotoxicity. In patients at increased risk for ASCVD who have chronic, stable liver disease (including non-alcoholic fatty liver disease), it is reasonable, when appropriately indicated, to use statins after obtaining baseline measurements and determining a schedule of monitoring and safety checks.
In patients at increased risk for ASCVD who have severe or recurrent SAMS after appropriate statin rechallenge, it is reasonable to use nonstatin therapy that is likely to provide net clinical benefit. The guideline does not recommend routine use of coenzyme Q10 supplementation for the treatment or prevention of SAMS.1
Guideline criticism
Guideline development is challenging on multiple levels, including balancing perspectives from multiple stakeholders. Nevertheless, the 2018 AHA/ACC cholesterol guideline builds nicely on progress made since its 2013 predecessor was released.4 This document was developed with the participation of representatives from 10 professional societies in addition to the ACC and AHA—notably, the National Lipid Association and American Society for Preventive Cardiology.1
To refine risk estimation and facilitate shared decision-making, the new guideline introduced so-called risk-enhancing factors and use of the CAC.1 However, some potential risk-enhancing factors were left out: erectile dysfunction, for example, often a marker of increased cardiovascular risk in men < 50 years of age.25 In addition, although pretreatment ApoB was introduced as a risk-enhancing factor,1 no recommendation is given to measure ApoB after initiation of therapy for evaluation of residual cardiovascular risk, as endorsed in other guidelines.26,27
Moreover, the guideline does not include the “extreme risk” category in the guideline developed by the American Association of Clinical Endocrinologists (AACE).28 Although the 2018 AHA/ACC guideline introduces < 70 mg/dL and < 100 mg/dL LDL-C thresholds,1 the < 55 mg/dL LDL-C threshold used for patients in the AACE/American College of Endocrinology extreme-risk category is not mentioned.26 This omission might leave patients who are at extreme ASCVD risk without optimal lipid-lowering therapy. Similarly, the guideline does not elaborate on the diagnosis and treatment of HoFH and HeFH.1 The age cutoff of 30 years for the recommendation to consider PCSK9 inhibitors in patients with HeFH appears arbitrary and excludes younger FH patients who have an extreme LDL-C elevation from potentially important therapy.23
Continue to: Guidelines are dynamic instruments...
Guidelines are dynamic instruments that require constant updating, given the production of new evidence. In fact, the results of the Reduction of Cardiovascular Events With Icosapent Ethyl-Intervention Trial (REDUCE-IT) were presented at the same meeting at which this guideline was unveiled.29 REDUCE-IT demonstrated an astonishing highly significant 25% reduction in the composite primary major adverse cardiovascular event outcome in patients with an LDL-C level of 44 to 100 mg/dL on statin therapy, who had a TG level of 135 to 499 mg/dL and had been treated for a median of 4.9 years with 4 g of pure eicosapentaenoic acid.
In addition, the guideline’s value statements, which address the need to consider the cost of drugs in determining most appropriate treatment, are no longer accurate because the price of PCSK9 inhibitors has dropped by more than half since the guideline was issued.30
An upward climb to clinical payoff
Even after close study of the 2018 AHA/ACC cholesterol guideline, implementing it in practice might remain a challenge to clinicians who are inexperienced in ordering lipid markers such as Lp(a) and interpreting the CAC score. Moreover, initiating and monitoring nonstatin therapies will be a demanding task—especially with PCSK9 inhibitors, which present access difficulties because they are relatively expensive (even after the recent price cut). That’s why, when there is doubt in the mind of the physician or other provider, we will likely see more referrals to specialists in lipid management and ASCVD risk estimation to optimize preventive therapy.31
CORRESPONDENCE
Cezary Wójcik, MD, PhD, FNLA, Oregon Health & Science University, 3181 SW Sam Jackson Park Rd, Portland, OR 97239; [email protected]
1. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 8. pii: S0735-1097(18)39034-X. [Epub ahead of print]
2. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S1-S45.
3. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. pii: S0735-1097(18)39033-8. [Epub ahead of print]
4. Wilson PWF, Polonsky TS, Miedema MD, et al. Systematic review for the 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. pii: S0735-1097(18)39035-1. [Epub ahead of print]
5. Eckel RH, Jakicic JM, Ard JD, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2960-2984.
6. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (adult treatment panel III): final report. Circulation. 2002;106:3143-3421.
7. US Preventive Services Task Force, Bibbins-Domingo K, Grossman DC, et al. Statin use for the primary prevention of cardiovascular disease in adults: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:1997-2007.
8. National Cholesterol Education Program. Second report of the Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (adult treatment panel II). Circulation. 1994;89:1333-1445.
9. Martin SS, Giugliano RP, Murphy SA, et al. Comparison of low-density lipoprotein cholesterol assessment by Martin/Hopkins estimation, Friedewald estimation, and preparative ultracentrifugation: insights from the FOURIER trial. JAMA Cardiol. 2018;3:749-753.
10. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
11. Sabatine MS, Giugliano RP, Keech AC, et al; FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-1722.
12. Szarek M, White HD, Schwartz GG, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab reduces total nonfatal cardiovascular and fatal events in the ODYSSEY OUTCOMES trial. J Am Coll Cardiol. 2019;73:387-396.
13. Crawford C. AAFP endorses ACC/AHA cholesterol management guidelines with qualifications. Leawood, KS: American Academy of Family Physicians; 2014 June 18. www.aafp.org/news/health-of-the-public/20140618cholesterolgdlnendorse.html. Accessed March 20, 2019.
14. Crawford C. AAFP News. AAFP affirms value of new cholesterol management guideline. March 20, 2019. www.aafp.org/news/health-of-the-public/20190320acc-ahacholguidln.html?cmpid=em_AP_20190320. Accessed April 1, 2019.
15. Lin JS, Evans CV, Johnson E, et al. Nontraditional Risk Factors in Cardiovascular Disease Risk Assessment: A Systematic Evidence Report for the U.S. Preventive Services Task Force. Evidence Synthesis, No. 166. Rockville, MD: Agency for Healthcare Research and Quality (US); 2018 Jul. Report No.: 17-05225-EF-1.
16. Puri R, Nicholls SJ, Shao M, et al. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol. 2015;65:1273-1282.
17. Gencer B, Kronenberg F, Stroes ES, et al. Lipoprotein(a): the revenant. Eur Heart J. 2017;38:1553-1560.
18. Michos ED, Blaha MJ, Blumenthal RS. Use of the coronary artery calcium score in discussion of initiation of statin therapy in primary prevention. Mayo Clin Proc. 2017;92:1831-1841.
19. MDsave. Cardiac CT calcium scoring. www.mdsave.com/procedures/cardiac-ct-calcium-scoring/d785f4cf. Accessed Aprl 1, 2019.
20. Blaha MJ, Budoff MJ, Tota-Maharaj R, et al. Improving the CAC score by addition of regional measures of calcium distribution: multi-ethnic study of atherosclerosis. JACC Cardiovasc Imaging. 2016;9:1407-1416.
21. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents: summary report. Pediatrics. 2011;128(Suppl 5):S213-S256.
22. US Preventive Services Task Force, Bibbins-Domingo K, Grossman DC, et al. Screening for lipid disorders in children and adolescents: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:625-633.
23. Gidding SS, Champagne MA, de Ferranti SD, et al; American Heart Association Atherosclerosis, Hypertension, and Obesity in Young Committee of Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and Council on Lifestyle and Cardiometabolic Health. The agenda for familial hypercholesterolemia: a scientific statement from the American Heart Association. Circulation. 2015;132:2167-2192.
24. Newman CB, Preiss D, Tobert JA, et al; American Heart Association Clinical Lipidology, Lipoprotein, Metabolism and Thrombosis Committee, a Joint Committee of the Council on Atherosclerosis, Thrombosis and Vascular Biology and Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular Disease in the Young; Council on Clinical Cardiology; and Stroke Council. Statin safety and associated adverse events: a scientific statement from the American Heart Association. Arterioscler Thromb Vasc Biol. 2019;39:e38-e81.
25. Miner M, Parish SJ, Billups KL, et al. Erectile dysfunction and subclinical cardiovascular disease. Sex Med Rev. 2018 Jan 27. pii: S2050-0521(18)30009-X. [Epub ahead of print]
26. Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1-87.
27. Anderson TJ, Grégoire J, Pearson GJ, et al. 2016 Canadian Cardiovascular Society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2016;32:1263-1282.
28. Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1-87.
29. Bhatt DL, Steg PG, Miller M, et al; REDUCE-IT Investigators. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22.
30. Dangi-Garimella S. Amgen announces 60% reduction in list price of PCSK9 inhibitor evolocumab. AJMC Managed Markets Network. October 24, 2018. https://www.ajmc.com/newsroom/amgen-announces-60-reduction-in-list-price-of-pcsk9-inhibitor-evolocumab. Accessed April 12, 2019.
31. Kaufman TM, Duell PB, Purnell JQ, et al. Application of PCSK9 inhibitors in practice: challenges and opportunities. Circ Res. 2017;121:499-501.
1. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 8. pii: S0735-1097(18)39034-X. [Epub ahead of print]
2. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S1-S45.
3. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. pii: S0735-1097(18)39033-8. [Epub ahead of print]
4. Wilson PWF, Polonsky TS, Miedema MD, et al. Systematic review for the 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. pii: S0735-1097(18)39035-1. [Epub ahead of print]
5. Eckel RH, Jakicic JM, Ard JD, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2960-2984.
6. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (adult treatment panel III): final report. Circulation. 2002;106:3143-3421.
7. US Preventive Services Task Force, Bibbins-Domingo K, Grossman DC, et al. Statin use for the primary prevention of cardiovascular disease in adults: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:1997-2007.
8. National Cholesterol Education Program. Second report of the Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (adult treatment panel II). Circulation. 1994;89:1333-1445.
9. Martin SS, Giugliano RP, Murphy SA, et al. Comparison of low-density lipoprotein cholesterol assessment by Martin/Hopkins estimation, Friedewald estimation, and preparative ultracentrifugation: insights from the FOURIER trial. JAMA Cardiol. 2018;3:749-753.
10. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397.
11. Sabatine MS, Giugliano RP, Keech AC, et al; FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376:1713-1722.
12. Szarek M, White HD, Schwartz GG, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab reduces total nonfatal cardiovascular and fatal events in the ODYSSEY OUTCOMES trial. J Am Coll Cardiol. 2019;73:387-396.
13. Crawford C. AAFP endorses ACC/AHA cholesterol management guidelines with qualifications. Leawood, KS: American Academy of Family Physicians; 2014 June 18. www.aafp.org/news/health-of-the-public/20140618cholesterolgdlnendorse.html. Accessed March 20, 2019.
14. Crawford C. AAFP News. AAFP affirms value of new cholesterol management guideline. March 20, 2019. www.aafp.org/news/health-of-the-public/20190320acc-ahacholguidln.html?cmpid=em_AP_20190320. Accessed April 1, 2019.
15. Lin JS, Evans CV, Johnson E, et al. Nontraditional Risk Factors in Cardiovascular Disease Risk Assessment: A Systematic Evidence Report for the U.S. Preventive Services Task Force. Evidence Synthesis, No. 166. Rockville, MD: Agency for Healthcare Research and Quality (US); 2018 Jul. Report No.: 17-05225-EF-1.
16. Puri R, Nicholls SJ, Shao M, et al. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol. 2015;65:1273-1282.
17. Gencer B, Kronenberg F, Stroes ES, et al. Lipoprotein(a): the revenant. Eur Heart J. 2017;38:1553-1560.
18. Michos ED, Blaha MJ, Blumenthal RS. Use of the coronary artery calcium score in discussion of initiation of statin therapy in primary prevention. Mayo Clin Proc. 2017;92:1831-1841.
19. MDsave. Cardiac CT calcium scoring. www.mdsave.com/procedures/cardiac-ct-calcium-scoring/d785f4cf. Accessed Aprl 1, 2019.
20. Blaha MJ, Budoff MJ, Tota-Maharaj R, et al. Improving the CAC score by addition of regional measures of calcium distribution: multi-ethnic study of atherosclerosis. JACC Cardiovasc Imaging. 2016;9:1407-1416.
21. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute. Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents: summary report. Pediatrics. 2011;128(Suppl 5):S213-S256.
22. US Preventive Services Task Force, Bibbins-Domingo K, Grossman DC, et al. Screening for lipid disorders in children and adolescents: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:625-633.
23. Gidding SS, Champagne MA, de Ferranti SD, et al; American Heart Association Atherosclerosis, Hypertension, and Obesity in Young Committee of Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and Council on Lifestyle and Cardiometabolic Health. The agenda for familial hypercholesterolemia: a scientific statement from the American Heart Association. Circulation. 2015;132:2167-2192.
24. Newman CB, Preiss D, Tobert JA, et al; American Heart Association Clinical Lipidology, Lipoprotein, Metabolism and Thrombosis Committee, a Joint Committee of the Council on Atherosclerosis, Thrombosis and Vascular Biology and Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular Disease in the Young; Council on Clinical Cardiology; and Stroke Council. Statin safety and associated adverse events: a scientific statement from the American Heart Association. Arterioscler Thromb Vasc Biol. 2019;39:e38-e81.
25. Miner M, Parish SJ, Billups KL, et al. Erectile dysfunction and subclinical cardiovascular disease. Sex Med Rev. 2018 Jan 27. pii: S2050-0521(18)30009-X. [Epub ahead of print]
26. Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1-87.
27. Anderson TJ, Grégoire J, Pearson GJ, et al. 2016 Canadian Cardiovascular Society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2016;32:1263-1282.
28. Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of dyslipidemia and prevention of cardiovascular disease. Endocr Pract. 2017;23(Suppl 2):1-87.
29. Bhatt DL, Steg PG, Miller M, et al; REDUCE-IT Investigators. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22.
30. Dangi-Garimella S. Amgen announces 60% reduction in list price of PCSK9 inhibitor evolocumab. AJMC Managed Markets Network. October 24, 2018. https://www.ajmc.com/newsroom/amgen-announces-60-reduction-in-list-price-of-pcsk9-inhibitor-evolocumab. Accessed April 12, 2019.
31. Kaufman TM, Duell PB, Purnell JQ, et al. Application of PCSK9 inhibitors in practice: challenges and opportunities. Circ Res. 2017;121:499-501.
PRACTICE RECOMMENDATIONS
› Reduce the low-density lipoprotein cholesterol (LDL-C) level in patients with clinical atherosclerotic cardiovascular disease (ASCVD) using high-intensity statin therapy or maximally tolerated statin therapy. A
› Use an LDL-C threshold of 70 mg/dL to prompt consideration of adding nonstatin therapy in patients who have very high-risk ASCVD. A
› Start high-intensity statin therapy in patients who have primary hypercholesterolemia (LDL-C level ≥ 190 mg/dL) without calculating the 10-year ASCVD risk. A
› Begin moderate-intensity statin therapy in patients 40 to 75 years of age who have diabetes mellitus and an LDL-C level ≥ 70 mg/dL without calculating 10-year ASCVD risk. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Energy-based therapies in female genital cosmetic surgery: Hype, hope, and a way forward
Energy-based therapy use in gynecology dates back to the early 1970s, when ablative carbon dioxide (C02) lasers were employed to treat cervical erosions.1 Soon after, reports were published on laser treatment for diethylstilbestrol-associated vaginal adenosis, laser laparoscopy for adhesiolysis, laser hysteroscopy, and laser genital wart ablation.2 Starting around 2011, the first articles were published on the use of fractional C02 laser treatment for vulvovaginal atrophy.3,4 Use of laser and light-based therapies to treat “vaginal rejuvenation” is now increasing at an annual rate of 26%. In a few years, North America is expected to be the largest market for vaginal laser rejuvenation. In 2016, more than 500,000 feminine rejuvenation procedures were performed in the United States, and it is estimated that more than 27,000 energy-based devices will be in operation by 2021.5
Clearly, there is considerable public interest and intrigue in office-based female genital cosmetic procedures. In 2018, the US Food and Drug Administration contacted 7 manufacturers of energy-based devices to request revision and clarification for marketing of these devices, since these technologies are neither cleared nor approved for cosmetic vulvovaginal conditions.6 The companies responded within 30 days.
In this article, we appraise the existing literature regarding the mechanism of action of energy-based therapies used in gynecology and review outcomes of their use in female genital cosmetic surgery.
Laser technology devices and how they work
LASER is an acronym for Light Amplification by Stimulated Emission of Radiation. Laser devices are composed of 1) an excitable medium (gas, liquid, solid) needed to emit light, 2) an energy source to excite the medium, 3) mirrors to bounce the light back and forth, and 4) a delivery and cooling system (FIGURE 1).
The electromagnetic spectrum is the range of all the wavelengths of light, including visible light, radio waves, infrared light, ultraviolet light, x-rays, and gamma rays (FIGURE 2). Most lasers used for the treatment of vulvovaginal disorders, typically C02 and erbium:yttrium aluminum garnet (Er:YAG) lasers, involve the infrared wavelengths.
The basic principle of laser treatment is to match the wavelength of the laser with the absorption spectrum of the desired target—a chromophore such as hemoglobin, melanin, or water (FIGURE 3). In essence, light is absorbed by the chromophore (which in vulvar and vaginal tissues is mostly water) and transformed into heat, leading to target destruction. In a fractionated (or fractional) laser beam, the laser is broken up into many smaller beams that treat only portions of the treatment area, with areas of intact epithelium in between the treated areas. At appropriately low thermal denaturation temperatures (45° to 50°C), tissue regeneration can occur through activation of heat shock proteins and tissue growth factors, creating neocollagenesis and neovascularization.
The concept of ablative resurfacing versus fractional resurfacing is borrowed from dermatology (FIGURE 4), understanding that tissue ablation and thermal denaturation occur at temperatures greater than 100°C, as occurs with carbonization of vulvar condylomata.
Continue to: In dermatology, fractionated lasers...
In dermatology, fractionated lasers have been used in the treatment of hair removal, vascular and pigmented lesions, scars, wound healing, tattoo removal, warts, and actinic keratoses. For these conditions, the targeted chromophores are water, hemoglobin, melanosomes, and tattoo ink. The laser pulses must be shorter than the target tissue thermal relaxation times in order to avoid excess heating and collateral tissue damage. Choosing appropriate settings is critical to achieve selective heating, or destruction, of the target tissue. These settings include appropriate wavelengths, pulse durations, and fluence, which is energy delivered per unit area (typically, joules per square centimeter).
For gynecologic conditions, the lasers used are most often CO2, Er:YAG, and hybrid (which include ablative and nonablative wavelengths) devices. In the epithelium of the vagina and vulva, these lasers generally have a very shallow depth of optical penetration, on the order of 10 to 200 µm.
Radiofrequency-based devices emit focused electromagnetic waves
Radiofrequency systems use a wand to deliver radiofrequency energy to create heat within the subepithelial layers of vulvar and vaginal tissues, while the surface remains cool. These devices can use monopolar or bipolar energy (current) to create a reverse thermal gradient designed to heat the deeper tissues transepithelially at a higher temperature while a coolant protects the surface epithelium. Some wand technologies require multiple treatments, while others require only a single treatment.
The TABLE lists currently available energy-based technologies.
Therapeutic uses for energy-based devices
Investigators have studied laser devices for treating various gynecologic conditions, including vulvovaginal atrophy, stress urinary incontinence (UI), vaginal laxity, lichen sclerosus, and vulvodynia.
Vulvovaginal atrophy
Genitourinary syndrome of menopause (GSM) includes symptoms of vulvovaginal irritation, burning, itching, discharge, dyspareunia, lower urinary tract symptoms such as frequency and urinary tract infections, and vaginal dryness or vulvovaginal atrophy.7 First-line treatment for vulvovaginal atrophy includes the use of nonhormonal lubricants for intercourse and vaginal moisturizers, which temporarily moisten the vaginal epithelium. Low-dose vaginal estrogen is a second-line therapy for symptomatic vulvovaginal atrophy; newer pharmacologic options include dehydroepiandrosterone (DHEA) suppositories (prasterone), solubilized estradiol capsules, and the selective estrogen receptor modulator (SERM) ospemifene.
Fractionated CO2, Erb:YAG, and hybrid lasers also have been used to treat women with symptomatic vulvovaginal atrophy and GSM through similar mechanisms described in dermatologic conditions with low-temperature laser activation of tissue proteins and growth factors creating new connective tissue and angiogenesis. A number of landmark studies have been published detailing patient outcomes with energy-based treatments for these symptoms.
Three-arm trial. Cruz and colleagues conducted a double-blind randomized trial to evaluate the efficacy of fractional CO2 laser vaginal treatment compared with local estriol therapy and the combination of laser plus estriol.8 The investigators randomly assigned 45 postmenopausal women to treatment with fractional CO2 laser with placebo vaginal cream, estriol with sham laser, or laser plus estriol. Treatment consisted of 2 sessions 4 weeks apart, with 20 consecutive weeks of estriol or placebo 3 times per week.
At weeks 8 and 20, the Vaginal Health Index (VHI) average score was significantly higher in all study arms. At week 20, the laser plus estriol group also showed incremental improvement in the VHI score (P = .01). The laser and the laser plus estriol groups had significant improvement in dyspareunia, burning, and dryness, while the estriol group improved only in dryness (P<.001). The laser plus estriol group had significant improvement in the total Female Sexual Function Index (FSFI) score (P = .02) and in the individual domains of pain, desire, and lubrication. Although the laser-alone group had significant worsening in the FSFI pain domain (P = .04), all treatment arms had comparable FSFI total scores at week 20. No adverse events were recorded during the study period.
Continue to: Retrospective study...
Retrospective study. To assess the efficacy of 3, 4, or 5 treatments with microablative fractional CO2 laser therapy for symptoms of GSM, Athanasiou and colleagues studied outcomes in 94 postmenopausal women.9 The intensity or bothersomeness of GSM symptoms as well as sexual function significantly improved in this cohort. The intensity of dyspareunia and dryness decreased from a median of 9 (minimum–maximum, 5–10) and 8 (0–10), respectively, at baseline to 0 (0–6) and 0 (0–8) at 1 month after the last laser therapy (P<.001 for all). The FSFI score and the frequency of sexual intercourse rose from 10.8 (2–26.9) and 1 (0–8) at baseline to 27.8 (15.2–35.4) and 4 (2–8) at 1 month after the last laser therapy (P<.001 for all).
The positive effects of laser therapy were unchanged throughout the 12 months of follow-up, and the pattern was the same for symptom-free rates. No adverse events were recorded during the study period.
The investigators noted that, based on short- and long-term follow-up, 4 or 5 laser treatments may be superior to 3 treatments for lowering the intensity of GSM symptoms. They found no differences in outcomes between 4 and 5 laser treatments.
Prospective comparative cohort study. Gaspar and colleagues recruited 50 postmenopausal women with GSM and assigned 25 participants to 2 weeks of pretreatment with estriol ovules 3 times per week (for epithelial hydration) followed by 3 sessions of Er:YAG nonablative laser treatments; 25 women in the active control group received treatment with estriol ovules over 8 weeks.10 Pre- and posttreatment biopsies, maturation index, maturation value, pH, and VAS symptom analysis were recorded up to 18 months after treatment.
Up to the 6-month follow-up, both treatment groups had a statistically significant reduction of all GSM symptoms. At all follow-ups, however, symptom relief was more prominent in the laser-treated group. In addition, the effects of the laser therapy remained statistically significant at the 12- and 18-month follow-ups, while the treatment effects of estriol were diminished at 12 months and, at 18 months, this group had some symptoms that were significantly worse than before treatment.
Overall, adverse effects were minimal and transient in both groups, affecting 4% of participants in the laser group, and 12% in the estriol group.
Long-term effectiveness evaluation. To assess the long-term efficacy and acceptability of vaginal laser treatment for the management of GSM, Gambacciani and colleagues treated 205 postmenopausal women with an Er:YAG laser for 3 applications every 30 days, with evaluations performed after 1, 3, 6, 12, 18, and 24 months from the last laser treatment.11 An active control group (n = 49) received 3 months of local treatment with either hormonal (estriol gel twice weekly) or nonhormonal (hyaluronic acid-based preparations or moisturizers and lubricants) agents.
Treatment with the ER:YAG laser induced a significant decrease (P<.01) in scores of the Visual Analog Scale (VAS) for vulvovaginal atrophy symptoms for vaginal dryness and dyspareunia and an increase in the VHI score (P<.01) up to 12 months after the last treatment. After 18 and 24 months, values returned to levels similar to those at baseline.
Women who also had stress UI (n = 114) received additional laser treatment of the anterior vaginal wall specifically designed for UI, with assessment based on the International Consultation on Incontinence Questionnaire–Urinary Incontinence Short Form (ICIQ-UI SF). Laser treatment induced a significant decrease (P<.05) in ICIQ-UI SF scores compared with baseline values, and scores remained lower than baseline values after 1, 2, 3, 6, and 12 months after the last laser treatment. Values measured after 18 and 24 months, however, did not differ significantly from baseline.
In the control group, the VAS score showed a similar decrease and comparable pattern during the treatment period. However, after the end of the treatment period, the control group’s VAS scores for vaginal dryness and dyspareunia showed a progressive increase, and after 6 months, the values were significantly different from corresponding values measured in the laser therapy group. The follow-up period in the control group ended after 6 months, because almost all patients started a new local or systemic treatment for their GSM symptoms. No adverse events related to treatment were recorded throughout the study period.
In an earlier pilot study by the same authors, 19 women with GSM who also had mild to moderate stress UI were treated with a vaginal Er:YAG laser.12 Compared with vaginal estriol treatment in the active control group, laser treatment was associated with a significant improvement (P<.01) in ICIQ-SF scores, with rapid and long-lasting effects that persisted up to week 24 of the observation period.
Continue to: Urinary incontinence...
Urinary incontinence
The cause of UI is considered to be multifactorial, including disruption in connective tissue supports of the urethrovesical junction leading to urethral hypermobility, pelvic floor muscle weakness, nerve damage to the urethral rhabdosphincter related to pudendal neuropathy or pelvic plexopathy, and atrophic changes of the urethra mucosa and submucosa. Purported mechanisms of action for energy-based therapies designed for treatment of UI relate to direct effects on connective tissue, blood vessels, and possibly nerves.
In 3 clinical trials designed specifically to treat UI with an Er:YAG laser, women showed subjective symptomatic improvement.
Ogrinc and colleagues followed 175 pre- and postmenopausal women with stress UI or mixed UI in a prospective nonrandomized study.13 They treated women with an Er:YAG laser for an average of 2.5 (0.5) procedures separated by a 2-month period and performed follow-up assessments at 2, 6, and 12 months after treatment.
After treatment, 77% of women with stress UI had significant improvement in symptoms based on the ICIQ SF and the Incontinence Severity Index (ISI), while only 34% of those with mixed UI had no symptoms at 1-year follow-up. No major adverse effects were noted in either group.
Okui compared the effects of Er:YAG laser treatment with those of tension-free vaginal tape (TVT) or transobturator tape (TOT) sling procedures (n = 50 in each group) in women with stress UI or mixed UI.14 At 12 months after treatment, all 3 treatments demonstrated comparable improvements in the women with stress UI. Some patients with mixed UI in the TVT and TOT groups showed exacerbation, while all women in the laser-treated group tended to have symptom improvement.
In another recent study, Blaganje and colleagues randomly assigned 114 premenopausal parous women with stress UI to an Er:YAG laser procedure or sham treatment.15 Three months after treatment, ICIQ-UI SF scores were significantly more improved (P<.001) in the laser-treated group than in the sham group. In addition, 21% of laser-treated patients were dry at follow-up compared with 4% of the sham-treated group.
Key takeaway. While these studies showed promising short-term results for laser treatment of UI, they need to be replicated in appropriately powered clinical trials that include critical subjective and objective outcomes as well as longer-term follow-up for both effectiveness and safety.
Vaginal laxity/pre-prolapse
Vaginal laxity is defined as the symptom of excessive vaginal looseness.16 Also referred to as “pre-prolapse,” this subjective symptom generally refers to a widened vaginal opening (genital hiatus) but with pelvic organ prolapse that is within the vagina or hymen.17 Notably, the definition is ambiguous, and rigorous clinical data based on validated outcomes and prolapse grading are lacking.
Krychman and colleagues conducted the first randomized controlled study comparing monopolar radiofrequency at the vaginal introitus with sham therapy for vaginal laxity in 174 premenopausal women, known as the VIVEVE I trial.18 The primary outcome, the proportion of women reporting no vaginal laxity at 6 months after treatment, was assessed using a vaginal laxity questionnaire, a 7-point rating scale for laxity or tightness ranging from very loose to very tight. With a single radiofrequency treatment, 43.5% of the active group and 19.6% (P = .002) of the sham group obtained the primary outcome.
There were also statistically significant improvements in overall sexual function and decreased sexual distress. The adjusted odds ratio (OR, 3.39; 95% confidence interval, 1.54–7.45) showed that the likelihood of no vaginal laxity at 6 months was more than 3 times greater for women who received the active treatment compared with those who received sham treatment. Adverse events were mild, resolved spontaneously, and were similar in the 2 groups.
Continue to: Outlook for energy-based...
Outlook for energy-based therapies: Cautiously optimistic
Preliminary outcome data on the use of energy-based therapies for female genital cosmetic surgery is largely positive for the treatment of vulvovaginal atrophy, but some case series suggest the potential for scarring, burning, and inefficacy. This prompted the FDA to send “It has come to our attention” letters to a number of device manufacturers in 2018.6
Supportive evidence is weak. Early data are encouraging regarding fractionated laser therapy for the treatment of vulvovaginal atrophy and stress UI and radiofrequency wand therapy for vaginal laxity and stress UI. Unfortunately, the level of evidence to support wide use of these technologies for all pelvic floor disorders is weak. A recent committee opinion from the International Urogynecology Association noted that only 8 studies (1 randomized trial and 7 observational studies) on these conditions fulfilled the criteria of good quality.19 The International Continence Society and the International Society for the Study of Vulvovaginal Disorders recently published a best practice consensus document declaring laser and energy-based treatments in gynecology and urology to be largely experimental.20
Questions persist. Knowledge gaps exist, and recommendations related to subspecialty training—who should perform these procedures (gynecologists, plastic surgeons, urologists, dermatologists, family practitioners) and the level of training needed to safely perform them—are lacking. Patient selection and physician knowledge and experience related to female genital anatomy, female sexual function and dysfunction, multidisciplinary treatment options for various pelvic support problems and UI, as well as psychologic screening for body dysmorphic disorders, need to be considered in terms of treating both the functional and aesthetic aspects related to cosmetic and reconstructive gynecologic surgery.
Special considerations. The use of energy-based therapies in special populations, such as survivors of breast cancer or other gynecologic cancers, as well as women undergoing chemotherapy, radiation therapy, and hormonal manipulation (particularly with antiestrogenic SERMs and aromatase inhibitors) has not been adequately evaluated. A discussion of the risks, benefits, alternatives, and limited long-term outcome data for energy-based therapies in cancer survivors, as for all patients, must be included for adequate informed consent prior to undertaking these treatments.
Guidelines for appropriate tissue priming, laser settings, and concomitant energy-based technology with local hormone treatment (also known as laser-augmented drug delivery) need to be developed. Comparative long-term studies are needed to determine the safety and effectiveness of these technologies.
Caution advised. Given the lack of long-term safety and effectiveness data on energy-based therapies for the vague indications of vaginal laxity, and even for the well-defined conditions of stress UI and vulvovaginal atrophy, clinicians should exercise caution before promoting treatment, which can be expensive and is not without potential complications, such as vaginal pain, adhesive agglutination, and persistent dryness and dyspareunia.21
Fortunately, many randomized trials on various energy-based devices for gynecologic indications (GSM, stress UI, vaginal laxity, lichen sclerosus) are underway, and results from these studies will help inform future clinical practice and guideline development.
- Kaplan I, Goldman J, Ger R. The treatment of erosions of the uterine cervix by means of the CO2 laser. Obstet Gynecol. 1973;41:795-796.
- Tadir Y, Gaspar A, Lev-Sagie A, et al. Light and energy-based therapeutics for genitourinary syndrome of menopause: consensus and controversies. Lasers Surg Med. 2017;49:137-159.
- Gaspar A, Addamo G, Brandi H. Vaginal fractional CO2 laser: a minimally invasive option for vaginal rejuvenation. Am J Cosmetic Surg. 2011;28:156-162.
- Salvatore S, Leone Roberti Maggiore U, Athanasiou S, et al. Histological study on the effects of microablative fractional CO2 laser on atrophic vaginal tissue: an ex vivo study. Menopause. 2015;22:845-849.
- Benedetto AV. What's new in cosmetic dermatology. Dermatol Clin. 2019;37:117-128.
- US Food and Drug Administration. FDA warns against use of energy-based devices to perform vaginal rejuvenation or vaginal cosmetic procedures: FDA safety communication. https://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm615013.htm. Accessed April 8, 2019.
- Portman DJ, Gass ML; Vulvovaginal Atrophy Terminology Consensus Conference Panel. Genitourinary syndrome of menopause: new terminology for vulvovaginal atrophy from the International Society for the Study of Women's Sexual Health and the North American Menopause Society. Menopause. 2014;21:1063-1068.
- Cruz VL, Steiner ML, Pompei LM, et al. Randomized, double-blind, placebo-controlled clinical trial for evaluating the efficacy of fractional CO2 laser compared with topical estriol in the treatment of vaginal atrophy in postmenopausal women. Menopause. 2018;25:21-28.
- Athanasiou S, Pitsouni E, Grigoradis T, et al. Microablative fractional CO2 laser for the genitourinary syndrome of menopause: up to 12-month results. Menopause. 2019;26:248-255.
- Gaspar A, Brandi H, Gomez V, et al. Efficacy of Erbium:YAG laser treatment compared to topical estriol treatment for symptoms of genitourinary syndrome of menopause. Lasers Surg Med. 2017;49:160-168.
- Gambacciani M, Levancini M, Russo E, et al. Long-term effects of vaginal erbium laser in the treatment of genitourinary syndrome of menopause. Climacteric. 2018;21:148-152.
- Gambacciani M, Levancini M, Cervigni M. Vaginal erbium laser: the second-generation thermotherapy for the genitourinary syndrome of menopause. Climacteric. 2015;18:757-763.
- Ogrinc UB, Sencar S, Lenasi H. Novel minimally invasive laser treatment of urinary incontinence in women. Lasers Surg Med. 2015;47:689-697.
- Okui N. Comparison between erbium-doped yttrium aluminum garnet laser therapy and sling procedures in the treatment of stress and mixed urinary incontinence. World J Urol. 2018. doi:10.1007/s00345-018-2445-x.
- Blaganje M, Scepanovic D, Zgur L, et al. Non-ablative Er:YAG laser therapy effect on stress urinary incontinence related to quality of life and sexual function: a randomized controlled trial. Eur J Obstet Gynecol Reprod Biol. 2018;224:153-158.
- Haylen BT, Maher CF, Barber MD, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic organ prolapse (POP). Int Urogynecologic J. 2016;27:165-194.
- Garcia B, Pardo J. Academic cosmetic gynecology and energy-based therapies: ambiguities, explorations, and the FDA advisories. Int Urogynecol J. 2019;30:1-2.
- Krychman M, Rowan CG, Allan BB, et al. Effect of single-treatment, surface-cooled radiofrequency therapy on vaginal laxity and female sexual function: the VIVEVE I randomized controlled trial. J Sex Med. 2017;14:215-225.
- Shobeiri SA, Kerkhof MH, Minassian VA, et al; IUGA Research and Development Committee. IUGA committee opinion: laser-based vaginal devices for treatment of stress urinary incontinence, genitourinary syndrome of menopause, and vaginal laxity. Int Urogynecol J. 2019;30:371-376.
- Preti M, Vieira-Baptista P, Digesu GA, et al. The clinical role of LASER for vulvar and vaginal treatments in gynecology and female urology: an ICS/ISSVD best practice consensus document. Neurourol Urodyn. 2019;38:1009-1023.
- Gordon C, Gonzales S, Krychman ML. Rethinking the techno vagina: a case series of patient complications following vaginal laser treatment for atrophy. Menopause. 2019;26:423-427.
Energy-based therapy use in gynecology dates back to the early 1970s, when ablative carbon dioxide (C02) lasers were employed to treat cervical erosions.1 Soon after, reports were published on laser treatment for diethylstilbestrol-associated vaginal adenosis, laser laparoscopy for adhesiolysis, laser hysteroscopy, and laser genital wart ablation.2 Starting around 2011, the first articles were published on the use of fractional C02 laser treatment for vulvovaginal atrophy.3,4 Use of laser and light-based therapies to treat “vaginal rejuvenation” is now increasing at an annual rate of 26%. In a few years, North America is expected to be the largest market for vaginal laser rejuvenation. In 2016, more than 500,000 feminine rejuvenation procedures were performed in the United States, and it is estimated that more than 27,000 energy-based devices will be in operation by 2021.5
Clearly, there is considerable public interest and intrigue in office-based female genital cosmetic procedures. In 2018, the US Food and Drug Administration contacted 7 manufacturers of energy-based devices to request revision and clarification for marketing of these devices, since these technologies are neither cleared nor approved for cosmetic vulvovaginal conditions.6 The companies responded within 30 days.
In this article, we appraise the existing literature regarding the mechanism of action of energy-based therapies used in gynecology and review outcomes of their use in female genital cosmetic surgery.
Laser technology devices and how they work
LASER is an acronym for Light Amplification by Stimulated Emission of Radiation. Laser devices are composed of 1) an excitable medium (gas, liquid, solid) needed to emit light, 2) an energy source to excite the medium, 3) mirrors to bounce the light back and forth, and 4) a delivery and cooling system (FIGURE 1).
The electromagnetic spectrum is the range of all the wavelengths of light, including visible light, radio waves, infrared light, ultraviolet light, x-rays, and gamma rays (FIGURE 2). Most lasers used for the treatment of vulvovaginal disorders, typically C02 and erbium:yttrium aluminum garnet (Er:YAG) lasers, involve the infrared wavelengths.
The basic principle of laser treatment is to match the wavelength of the laser with the absorption spectrum of the desired target—a chromophore such as hemoglobin, melanin, or water (FIGURE 3). In essence, light is absorbed by the chromophore (which in vulvar and vaginal tissues is mostly water) and transformed into heat, leading to target destruction. In a fractionated (or fractional) laser beam, the laser is broken up into many smaller beams that treat only portions of the treatment area, with areas of intact epithelium in between the treated areas. At appropriately low thermal denaturation temperatures (45° to 50°C), tissue regeneration can occur through activation of heat shock proteins and tissue growth factors, creating neocollagenesis and neovascularization.
The concept of ablative resurfacing versus fractional resurfacing is borrowed from dermatology (FIGURE 4), understanding that tissue ablation and thermal denaturation occur at temperatures greater than 100°C, as occurs with carbonization of vulvar condylomata.
Continue to: In dermatology, fractionated lasers...
In dermatology, fractionated lasers have been used in the treatment of hair removal, vascular and pigmented lesions, scars, wound healing, tattoo removal, warts, and actinic keratoses. For these conditions, the targeted chromophores are water, hemoglobin, melanosomes, and tattoo ink. The laser pulses must be shorter than the target tissue thermal relaxation times in order to avoid excess heating and collateral tissue damage. Choosing appropriate settings is critical to achieve selective heating, or destruction, of the target tissue. These settings include appropriate wavelengths, pulse durations, and fluence, which is energy delivered per unit area (typically, joules per square centimeter).
For gynecologic conditions, the lasers used are most often CO2, Er:YAG, and hybrid (which include ablative and nonablative wavelengths) devices. In the epithelium of the vagina and vulva, these lasers generally have a very shallow depth of optical penetration, on the order of 10 to 200 µm.
Radiofrequency-based devices emit focused electromagnetic waves
Radiofrequency systems use a wand to deliver radiofrequency energy to create heat within the subepithelial layers of vulvar and vaginal tissues, while the surface remains cool. These devices can use monopolar or bipolar energy (current) to create a reverse thermal gradient designed to heat the deeper tissues transepithelially at a higher temperature while a coolant protects the surface epithelium. Some wand technologies require multiple treatments, while others require only a single treatment.
The TABLE lists currently available energy-based technologies.
Therapeutic uses for energy-based devices
Investigators have studied laser devices for treating various gynecologic conditions, including vulvovaginal atrophy, stress urinary incontinence (UI), vaginal laxity, lichen sclerosus, and vulvodynia.
Vulvovaginal atrophy
Genitourinary syndrome of menopause (GSM) includes symptoms of vulvovaginal irritation, burning, itching, discharge, dyspareunia, lower urinary tract symptoms such as frequency and urinary tract infections, and vaginal dryness or vulvovaginal atrophy.7 First-line treatment for vulvovaginal atrophy includes the use of nonhormonal lubricants for intercourse and vaginal moisturizers, which temporarily moisten the vaginal epithelium. Low-dose vaginal estrogen is a second-line therapy for symptomatic vulvovaginal atrophy; newer pharmacologic options include dehydroepiandrosterone (DHEA) suppositories (prasterone), solubilized estradiol capsules, and the selective estrogen receptor modulator (SERM) ospemifene.
Fractionated CO2, Erb:YAG, and hybrid lasers also have been used to treat women with symptomatic vulvovaginal atrophy and GSM through similar mechanisms described in dermatologic conditions with low-temperature laser activation of tissue proteins and growth factors creating new connective tissue and angiogenesis. A number of landmark studies have been published detailing patient outcomes with energy-based treatments for these symptoms.
Three-arm trial. Cruz and colleagues conducted a double-blind randomized trial to evaluate the efficacy of fractional CO2 laser vaginal treatment compared with local estriol therapy and the combination of laser plus estriol.8 The investigators randomly assigned 45 postmenopausal women to treatment with fractional CO2 laser with placebo vaginal cream, estriol with sham laser, or laser plus estriol. Treatment consisted of 2 sessions 4 weeks apart, with 20 consecutive weeks of estriol or placebo 3 times per week.
At weeks 8 and 20, the Vaginal Health Index (VHI) average score was significantly higher in all study arms. At week 20, the laser plus estriol group also showed incremental improvement in the VHI score (P = .01). The laser and the laser plus estriol groups had significant improvement in dyspareunia, burning, and dryness, while the estriol group improved only in dryness (P<.001). The laser plus estriol group had significant improvement in the total Female Sexual Function Index (FSFI) score (P = .02) and in the individual domains of pain, desire, and lubrication. Although the laser-alone group had significant worsening in the FSFI pain domain (P = .04), all treatment arms had comparable FSFI total scores at week 20. No adverse events were recorded during the study period.
Continue to: Retrospective study...
Retrospective study. To assess the efficacy of 3, 4, or 5 treatments with microablative fractional CO2 laser therapy for symptoms of GSM, Athanasiou and colleagues studied outcomes in 94 postmenopausal women.9 The intensity or bothersomeness of GSM symptoms as well as sexual function significantly improved in this cohort. The intensity of dyspareunia and dryness decreased from a median of 9 (minimum–maximum, 5–10) and 8 (0–10), respectively, at baseline to 0 (0–6) and 0 (0–8) at 1 month after the last laser therapy (P<.001 for all). The FSFI score and the frequency of sexual intercourse rose from 10.8 (2–26.9) and 1 (0–8) at baseline to 27.8 (15.2–35.4) and 4 (2–8) at 1 month after the last laser therapy (P<.001 for all).
The positive effects of laser therapy were unchanged throughout the 12 months of follow-up, and the pattern was the same for symptom-free rates. No adverse events were recorded during the study period.
The investigators noted that, based on short- and long-term follow-up, 4 or 5 laser treatments may be superior to 3 treatments for lowering the intensity of GSM symptoms. They found no differences in outcomes between 4 and 5 laser treatments.
Prospective comparative cohort study. Gaspar and colleagues recruited 50 postmenopausal women with GSM and assigned 25 participants to 2 weeks of pretreatment with estriol ovules 3 times per week (for epithelial hydration) followed by 3 sessions of Er:YAG nonablative laser treatments; 25 women in the active control group received treatment with estriol ovules over 8 weeks.10 Pre- and posttreatment biopsies, maturation index, maturation value, pH, and VAS symptom analysis were recorded up to 18 months after treatment.
Up to the 6-month follow-up, both treatment groups had a statistically significant reduction of all GSM symptoms. At all follow-ups, however, symptom relief was more prominent in the laser-treated group. In addition, the effects of the laser therapy remained statistically significant at the 12- and 18-month follow-ups, while the treatment effects of estriol were diminished at 12 months and, at 18 months, this group had some symptoms that were significantly worse than before treatment.
Overall, adverse effects were minimal and transient in both groups, affecting 4% of participants in the laser group, and 12% in the estriol group.
Long-term effectiveness evaluation. To assess the long-term efficacy and acceptability of vaginal laser treatment for the management of GSM, Gambacciani and colleagues treated 205 postmenopausal women with an Er:YAG laser for 3 applications every 30 days, with evaluations performed after 1, 3, 6, 12, 18, and 24 months from the last laser treatment.11 An active control group (n = 49) received 3 months of local treatment with either hormonal (estriol gel twice weekly) or nonhormonal (hyaluronic acid-based preparations or moisturizers and lubricants) agents.
Treatment with the ER:YAG laser induced a significant decrease (P<.01) in scores of the Visual Analog Scale (VAS) for vulvovaginal atrophy symptoms for vaginal dryness and dyspareunia and an increase in the VHI score (P<.01) up to 12 months after the last treatment. After 18 and 24 months, values returned to levels similar to those at baseline.
Women who also had stress UI (n = 114) received additional laser treatment of the anterior vaginal wall specifically designed for UI, with assessment based on the International Consultation on Incontinence Questionnaire–Urinary Incontinence Short Form (ICIQ-UI SF). Laser treatment induced a significant decrease (P<.05) in ICIQ-UI SF scores compared with baseline values, and scores remained lower than baseline values after 1, 2, 3, 6, and 12 months after the last laser treatment. Values measured after 18 and 24 months, however, did not differ significantly from baseline.
In the control group, the VAS score showed a similar decrease and comparable pattern during the treatment period. However, after the end of the treatment period, the control group’s VAS scores for vaginal dryness and dyspareunia showed a progressive increase, and after 6 months, the values were significantly different from corresponding values measured in the laser therapy group. The follow-up period in the control group ended after 6 months, because almost all patients started a new local or systemic treatment for their GSM symptoms. No adverse events related to treatment were recorded throughout the study period.
In an earlier pilot study by the same authors, 19 women with GSM who also had mild to moderate stress UI were treated with a vaginal Er:YAG laser.12 Compared with vaginal estriol treatment in the active control group, laser treatment was associated with a significant improvement (P<.01) in ICIQ-SF scores, with rapid and long-lasting effects that persisted up to week 24 of the observation period.
Continue to: Urinary incontinence...
Urinary incontinence
The cause of UI is considered to be multifactorial, including disruption in connective tissue supports of the urethrovesical junction leading to urethral hypermobility, pelvic floor muscle weakness, nerve damage to the urethral rhabdosphincter related to pudendal neuropathy or pelvic plexopathy, and atrophic changes of the urethra mucosa and submucosa. Purported mechanisms of action for energy-based therapies designed for treatment of UI relate to direct effects on connective tissue, blood vessels, and possibly nerves.
In 3 clinical trials designed specifically to treat UI with an Er:YAG laser, women showed subjective symptomatic improvement.
Ogrinc and colleagues followed 175 pre- and postmenopausal women with stress UI or mixed UI in a prospective nonrandomized study.13 They treated women with an Er:YAG laser for an average of 2.5 (0.5) procedures separated by a 2-month period and performed follow-up assessments at 2, 6, and 12 months after treatment.
After treatment, 77% of women with stress UI had significant improvement in symptoms based on the ICIQ SF and the Incontinence Severity Index (ISI), while only 34% of those with mixed UI had no symptoms at 1-year follow-up. No major adverse effects were noted in either group.
Okui compared the effects of Er:YAG laser treatment with those of tension-free vaginal tape (TVT) or transobturator tape (TOT) sling procedures (n = 50 in each group) in women with stress UI or mixed UI.14 At 12 months after treatment, all 3 treatments demonstrated comparable improvements in the women with stress UI. Some patients with mixed UI in the TVT and TOT groups showed exacerbation, while all women in the laser-treated group tended to have symptom improvement.
In another recent study, Blaganje and colleagues randomly assigned 114 premenopausal parous women with stress UI to an Er:YAG laser procedure or sham treatment.15 Three months after treatment, ICIQ-UI SF scores were significantly more improved (P<.001) in the laser-treated group than in the sham group. In addition, 21% of laser-treated patients were dry at follow-up compared with 4% of the sham-treated group.
Key takeaway. While these studies showed promising short-term results for laser treatment of UI, they need to be replicated in appropriately powered clinical trials that include critical subjective and objective outcomes as well as longer-term follow-up for both effectiveness and safety.
Vaginal laxity/pre-prolapse
Vaginal laxity is defined as the symptom of excessive vaginal looseness.16 Also referred to as “pre-prolapse,” this subjective symptom generally refers to a widened vaginal opening (genital hiatus) but with pelvic organ prolapse that is within the vagina or hymen.17 Notably, the definition is ambiguous, and rigorous clinical data based on validated outcomes and prolapse grading are lacking.
Krychman and colleagues conducted the first randomized controlled study comparing monopolar radiofrequency at the vaginal introitus with sham therapy for vaginal laxity in 174 premenopausal women, known as the VIVEVE I trial.18 The primary outcome, the proportion of women reporting no vaginal laxity at 6 months after treatment, was assessed using a vaginal laxity questionnaire, a 7-point rating scale for laxity or tightness ranging from very loose to very tight. With a single radiofrequency treatment, 43.5% of the active group and 19.6% (P = .002) of the sham group obtained the primary outcome.
There were also statistically significant improvements in overall sexual function and decreased sexual distress. The adjusted odds ratio (OR, 3.39; 95% confidence interval, 1.54–7.45) showed that the likelihood of no vaginal laxity at 6 months was more than 3 times greater for women who received the active treatment compared with those who received sham treatment. Adverse events were mild, resolved spontaneously, and were similar in the 2 groups.
Continue to: Outlook for energy-based...
Outlook for energy-based therapies: Cautiously optimistic
Preliminary outcome data on the use of energy-based therapies for female genital cosmetic surgery is largely positive for the treatment of vulvovaginal atrophy, but some case series suggest the potential for scarring, burning, and inefficacy. This prompted the FDA to send “It has come to our attention” letters to a number of device manufacturers in 2018.6
Supportive evidence is weak. Early data are encouraging regarding fractionated laser therapy for the treatment of vulvovaginal atrophy and stress UI and radiofrequency wand therapy for vaginal laxity and stress UI. Unfortunately, the level of evidence to support wide use of these technologies for all pelvic floor disorders is weak. A recent committee opinion from the International Urogynecology Association noted that only 8 studies (1 randomized trial and 7 observational studies) on these conditions fulfilled the criteria of good quality.19 The International Continence Society and the International Society for the Study of Vulvovaginal Disorders recently published a best practice consensus document declaring laser and energy-based treatments in gynecology and urology to be largely experimental.20
Questions persist. Knowledge gaps exist, and recommendations related to subspecialty training—who should perform these procedures (gynecologists, plastic surgeons, urologists, dermatologists, family practitioners) and the level of training needed to safely perform them—are lacking. Patient selection and physician knowledge and experience related to female genital anatomy, female sexual function and dysfunction, multidisciplinary treatment options for various pelvic support problems and UI, as well as psychologic screening for body dysmorphic disorders, need to be considered in terms of treating both the functional and aesthetic aspects related to cosmetic and reconstructive gynecologic surgery.
Special considerations. The use of energy-based therapies in special populations, such as survivors of breast cancer or other gynecologic cancers, as well as women undergoing chemotherapy, radiation therapy, and hormonal manipulation (particularly with antiestrogenic SERMs and aromatase inhibitors) has not been adequately evaluated. A discussion of the risks, benefits, alternatives, and limited long-term outcome data for energy-based therapies in cancer survivors, as for all patients, must be included for adequate informed consent prior to undertaking these treatments.
Guidelines for appropriate tissue priming, laser settings, and concomitant energy-based technology with local hormone treatment (also known as laser-augmented drug delivery) need to be developed. Comparative long-term studies are needed to determine the safety and effectiveness of these technologies.
Caution advised. Given the lack of long-term safety and effectiveness data on energy-based therapies for the vague indications of vaginal laxity, and even for the well-defined conditions of stress UI and vulvovaginal atrophy, clinicians should exercise caution before promoting treatment, which can be expensive and is not without potential complications, such as vaginal pain, adhesive agglutination, and persistent dryness and dyspareunia.21
Fortunately, many randomized trials on various energy-based devices for gynecologic indications (GSM, stress UI, vaginal laxity, lichen sclerosus) are underway, and results from these studies will help inform future clinical practice and guideline development.
Energy-based therapy use in gynecology dates back to the early 1970s, when ablative carbon dioxide (C02) lasers were employed to treat cervical erosions.1 Soon after, reports were published on laser treatment for diethylstilbestrol-associated vaginal adenosis, laser laparoscopy for adhesiolysis, laser hysteroscopy, and laser genital wart ablation.2 Starting around 2011, the first articles were published on the use of fractional C02 laser treatment for vulvovaginal atrophy.3,4 Use of laser and light-based therapies to treat “vaginal rejuvenation” is now increasing at an annual rate of 26%. In a few years, North America is expected to be the largest market for vaginal laser rejuvenation. In 2016, more than 500,000 feminine rejuvenation procedures were performed in the United States, and it is estimated that more than 27,000 energy-based devices will be in operation by 2021.5
Clearly, there is considerable public interest and intrigue in office-based female genital cosmetic procedures. In 2018, the US Food and Drug Administration contacted 7 manufacturers of energy-based devices to request revision and clarification for marketing of these devices, since these technologies are neither cleared nor approved for cosmetic vulvovaginal conditions.6 The companies responded within 30 days.
In this article, we appraise the existing literature regarding the mechanism of action of energy-based therapies used in gynecology and review outcomes of their use in female genital cosmetic surgery.
Laser technology devices and how they work
LASER is an acronym for Light Amplification by Stimulated Emission of Radiation. Laser devices are composed of 1) an excitable medium (gas, liquid, solid) needed to emit light, 2) an energy source to excite the medium, 3) mirrors to bounce the light back and forth, and 4) a delivery and cooling system (FIGURE 1).
The electromagnetic spectrum is the range of all the wavelengths of light, including visible light, radio waves, infrared light, ultraviolet light, x-rays, and gamma rays (FIGURE 2). Most lasers used for the treatment of vulvovaginal disorders, typically C02 and erbium:yttrium aluminum garnet (Er:YAG) lasers, involve the infrared wavelengths.
The basic principle of laser treatment is to match the wavelength of the laser with the absorption spectrum of the desired target—a chromophore such as hemoglobin, melanin, or water (FIGURE 3). In essence, light is absorbed by the chromophore (which in vulvar and vaginal tissues is mostly water) and transformed into heat, leading to target destruction. In a fractionated (or fractional) laser beam, the laser is broken up into many smaller beams that treat only portions of the treatment area, with areas of intact epithelium in between the treated areas. At appropriately low thermal denaturation temperatures (45° to 50°C), tissue regeneration can occur through activation of heat shock proteins and tissue growth factors, creating neocollagenesis and neovascularization.
The concept of ablative resurfacing versus fractional resurfacing is borrowed from dermatology (FIGURE 4), understanding that tissue ablation and thermal denaturation occur at temperatures greater than 100°C, as occurs with carbonization of vulvar condylomata.
Continue to: In dermatology, fractionated lasers...
In dermatology, fractionated lasers have been used in the treatment of hair removal, vascular and pigmented lesions, scars, wound healing, tattoo removal, warts, and actinic keratoses. For these conditions, the targeted chromophores are water, hemoglobin, melanosomes, and tattoo ink. The laser pulses must be shorter than the target tissue thermal relaxation times in order to avoid excess heating and collateral tissue damage. Choosing appropriate settings is critical to achieve selective heating, or destruction, of the target tissue. These settings include appropriate wavelengths, pulse durations, and fluence, which is energy delivered per unit area (typically, joules per square centimeter).
For gynecologic conditions, the lasers used are most often CO2, Er:YAG, and hybrid (which include ablative and nonablative wavelengths) devices. In the epithelium of the vagina and vulva, these lasers generally have a very shallow depth of optical penetration, on the order of 10 to 200 µm.
Radiofrequency-based devices emit focused electromagnetic waves
Radiofrequency systems use a wand to deliver radiofrequency energy to create heat within the subepithelial layers of vulvar and vaginal tissues, while the surface remains cool. These devices can use monopolar or bipolar energy (current) to create a reverse thermal gradient designed to heat the deeper tissues transepithelially at a higher temperature while a coolant protects the surface epithelium. Some wand technologies require multiple treatments, while others require only a single treatment.
The TABLE lists currently available energy-based technologies.
Therapeutic uses for energy-based devices
Investigators have studied laser devices for treating various gynecologic conditions, including vulvovaginal atrophy, stress urinary incontinence (UI), vaginal laxity, lichen sclerosus, and vulvodynia.
Vulvovaginal atrophy
Genitourinary syndrome of menopause (GSM) includes symptoms of vulvovaginal irritation, burning, itching, discharge, dyspareunia, lower urinary tract symptoms such as frequency and urinary tract infections, and vaginal dryness or vulvovaginal atrophy.7 First-line treatment for vulvovaginal atrophy includes the use of nonhormonal lubricants for intercourse and vaginal moisturizers, which temporarily moisten the vaginal epithelium. Low-dose vaginal estrogen is a second-line therapy for symptomatic vulvovaginal atrophy; newer pharmacologic options include dehydroepiandrosterone (DHEA) suppositories (prasterone), solubilized estradiol capsules, and the selective estrogen receptor modulator (SERM) ospemifene.
Fractionated CO2, Erb:YAG, and hybrid lasers also have been used to treat women with symptomatic vulvovaginal atrophy and GSM through similar mechanisms described in dermatologic conditions with low-temperature laser activation of tissue proteins and growth factors creating new connective tissue and angiogenesis. A number of landmark studies have been published detailing patient outcomes with energy-based treatments for these symptoms.
Three-arm trial. Cruz and colleagues conducted a double-blind randomized trial to evaluate the efficacy of fractional CO2 laser vaginal treatment compared with local estriol therapy and the combination of laser plus estriol.8 The investigators randomly assigned 45 postmenopausal women to treatment with fractional CO2 laser with placebo vaginal cream, estriol with sham laser, or laser plus estriol. Treatment consisted of 2 sessions 4 weeks apart, with 20 consecutive weeks of estriol or placebo 3 times per week.
At weeks 8 and 20, the Vaginal Health Index (VHI) average score was significantly higher in all study arms. At week 20, the laser plus estriol group also showed incremental improvement in the VHI score (P = .01). The laser and the laser plus estriol groups had significant improvement in dyspareunia, burning, and dryness, while the estriol group improved only in dryness (P<.001). The laser plus estriol group had significant improvement in the total Female Sexual Function Index (FSFI) score (P = .02) and in the individual domains of pain, desire, and lubrication. Although the laser-alone group had significant worsening in the FSFI pain domain (P = .04), all treatment arms had comparable FSFI total scores at week 20. No adverse events were recorded during the study period.
Continue to: Retrospective study...
Retrospective study. To assess the efficacy of 3, 4, or 5 treatments with microablative fractional CO2 laser therapy for symptoms of GSM, Athanasiou and colleagues studied outcomes in 94 postmenopausal women.9 The intensity or bothersomeness of GSM symptoms as well as sexual function significantly improved in this cohort. The intensity of dyspareunia and dryness decreased from a median of 9 (minimum–maximum, 5–10) and 8 (0–10), respectively, at baseline to 0 (0–6) and 0 (0–8) at 1 month after the last laser therapy (P<.001 for all). The FSFI score and the frequency of sexual intercourse rose from 10.8 (2–26.9) and 1 (0–8) at baseline to 27.8 (15.2–35.4) and 4 (2–8) at 1 month after the last laser therapy (P<.001 for all).
The positive effects of laser therapy were unchanged throughout the 12 months of follow-up, and the pattern was the same for symptom-free rates. No adverse events were recorded during the study period.
The investigators noted that, based on short- and long-term follow-up, 4 or 5 laser treatments may be superior to 3 treatments for lowering the intensity of GSM symptoms. They found no differences in outcomes between 4 and 5 laser treatments.
Prospective comparative cohort study. Gaspar and colleagues recruited 50 postmenopausal women with GSM and assigned 25 participants to 2 weeks of pretreatment with estriol ovules 3 times per week (for epithelial hydration) followed by 3 sessions of Er:YAG nonablative laser treatments; 25 women in the active control group received treatment with estriol ovules over 8 weeks.10 Pre- and posttreatment biopsies, maturation index, maturation value, pH, and VAS symptom analysis were recorded up to 18 months after treatment.
Up to the 6-month follow-up, both treatment groups had a statistically significant reduction of all GSM symptoms. At all follow-ups, however, symptom relief was more prominent in the laser-treated group. In addition, the effects of the laser therapy remained statistically significant at the 12- and 18-month follow-ups, while the treatment effects of estriol were diminished at 12 months and, at 18 months, this group had some symptoms that were significantly worse than before treatment.
Overall, adverse effects were minimal and transient in both groups, affecting 4% of participants in the laser group, and 12% in the estriol group.
Long-term effectiveness evaluation. To assess the long-term efficacy and acceptability of vaginal laser treatment for the management of GSM, Gambacciani and colleagues treated 205 postmenopausal women with an Er:YAG laser for 3 applications every 30 days, with evaluations performed after 1, 3, 6, 12, 18, and 24 months from the last laser treatment.11 An active control group (n = 49) received 3 months of local treatment with either hormonal (estriol gel twice weekly) or nonhormonal (hyaluronic acid-based preparations or moisturizers and lubricants) agents.
Treatment with the ER:YAG laser induced a significant decrease (P<.01) in scores of the Visual Analog Scale (VAS) for vulvovaginal atrophy symptoms for vaginal dryness and dyspareunia and an increase in the VHI score (P<.01) up to 12 months after the last treatment. After 18 and 24 months, values returned to levels similar to those at baseline.
Women who also had stress UI (n = 114) received additional laser treatment of the anterior vaginal wall specifically designed for UI, with assessment based on the International Consultation on Incontinence Questionnaire–Urinary Incontinence Short Form (ICIQ-UI SF). Laser treatment induced a significant decrease (P<.05) in ICIQ-UI SF scores compared with baseline values, and scores remained lower than baseline values after 1, 2, 3, 6, and 12 months after the last laser treatment. Values measured after 18 and 24 months, however, did not differ significantly from baseline.
In the control group, the VAS score showed a similar decrease and comparable pattern during the treatment period. However, after the end of the treatment period, the control group’s VAS scores for vaginal dryness and dyspareunia showed a progressive increase, and after 6 months, the values were significantly different from corresponding values measured in the laser therapy group. The follow-up period in the control group ended after 6 months, because almost all patients started a new local or systemic treatment for their GSM symptoms. No adverse events related to treatment were recorded throughout the study period.
In an earlier pilot study by the same authors, 19 women with GSM who also had mild to moderate stress UI were treated with a vaginal Er:YAG laser.12 Compared with vaginal estriol treatment in the active control group, laser treatment was associated with a significant improvement (P<.01) in ICIQ-SF scores, with rapid and long-lasting effects that persisted up to week 24 of the observation period.
Continue to: Urinary incontinence...
Urinary incontinence
The cause of UI is considered to be multifactorial, including disruption in connective tissue supports of the urethrovesical junction leading to urethral hypermobility, pelvic floor muscle weakness, nerve damage to the urethral rhabdosphincter related to pudendal neuropathy or pelvic plexopathy, and atrophic changes of the urethra mucosa and submucosa. Purported mechanisms of action for energy-based therapies designed for treatment of UI relate to direct effects on connective tissue, blood vessels, and possibly nerves.
In 3 clinical trials designed specifically to treat UI with an Er:YAG laser, women showed subjective symptomatic improvement.
Ogrinc and colleagues followed 175 pre- and postmenopausal women with stress UI or mixed UI in a prospective nonrandomized study.13 They treated women with an Er:YAG laser for an average of 2.5 (0.5) procedures separated by a 2-month period and performed follow-up assessments at 2, 6, and 12 months after treatment.
After treatment, 77% of women with stress UI had significant improvement in symptoms based on the ICIQ SF and the Incontinence Severity Index (ISI), while only 34% of those with mixed UI had no symptoms at 1-year follow-up. No major adverse effects were noted in either group.
Okui compared the effects of Er:YAG laser treatment with those of tension-free vaginal tape (TVT) or transobturator tape (TOT) sling procedures (n = 50 in each group) in women with stress UI or mixed UI.14 At 12 months after treatment, all 3 treatments demonstrated comparable improvements in the women with stress UI. Some patients with mixed UI in the TVT and TOT groups showed exacerbation, while all women in the laser-treated group tended to have symptom improvement.
In another recent study, Blaganje and colleagues randomly assigned 114 premenopausal parous women with stress UI to an Er:YAG laser procedure or sham treatment.15 Three months after treatment, ICIQ-UI SF scores were significantly more improved (P<.001) in the laser-treated group than in the sham group. In addition, 21% of laser-treated patients were dry at follow-up compared with 4% of the sham-treated group.
Key takeaway. While these studies showed promising short-term results for laser treatment of UI, they need to be replicated in appropriately powered clinical trials that include critical subjective and objective outcomes as well as longer-term follow-up for both effectiveness and safety.
Vaginal laxity/pre-prolapse
Vaginal laxity is defined as the symptom of excessive vaginal looseness.16 Also referred to as “pre-prolapse,” this subjective symptom generally refers to a widened vaginal opening (genital hiatus) but with pelvic organ prolapse that is within the vagina or hymen.17 Notably, the definition is ambiguous, and rigorous clinical data based on validated outcomes and prolapse grading are lacking.
Krychman and colleagues conducted the first randomized controlled study comparing monopolar radiofrequency at the vaginal introitus with sham therapy for vaginal laxity in 174 premenopausal women, known as the VIVEVE I trial.18 The primary outcome, the proportion of women reporting no vaginal laxity at 6 months after treatment, was assessed using a vaginal laxity questionnaire, a 7-point rating scale for laxity or tightness ranging from very loose to very tight. With a single radiofrequency treatment, 43.5% of the active group and 19.6% (P = .002) of the sham group obtained the primary outcome.
There were also statistically significant improvements in overall sexual function and decreased sexual distress. The adjusted odds ratio (OR, 3.39; 95% confidence interval, 1.54–7.45) showed that the likelihood of no vaginal laxity at 6 months was more than 3 times greater for women who received the active treatment compared with those who received sham treatment. Adverse events were mild, resolved spontaneously, and were similar in the 2 groups.
Continue to: Outlook for energy-based...
Outlook for energy-based therapies: Cautiously optimistic
Preliminary outcome data on the use of energy-based therapies for female genital cosmetic surgery is largely positive for the treatment of vulvovaginal atrophy, but some case series suggest the potential for scarring, burning, and inefficacy. This prompted the FDA to send “It has come to our attention” letters to a number of device manufacturers in 2018.6
Supportive evidence is weak. Early data are encouraging regarding fractionated laser therapy for the treatment of vulvovaginal atrophy and stress UI and radiofrequency wand therapy for vaginal laxity and stress UI. Unfortunately, the level of evidence to support wide use of these technologies for all pelvic floor disorders is weak. A recent committee opinion from the International Urogynecology Association noted that only 8 studies (1 randomized trial and 7 observational studies) on these conditions fulfilled the criteria of good quality.19 The International Continence Society and the International Society for the Study of Vulvovaginal Disorders recently published a best practice consensus document declaring laser and energy-based treatments in gynecology and urology to be largely experimental.20
Questions persist. Knowledge gaps exist, and recommendations related to subspecialty training—who should perform these procedures (gynecologists, plastic surgeons, urologists, dermatologists, family practitioners) and the level of training needed to safely perform them—are lacking. Patient selection and physician knowledge and experience related to female genital anatomy, female sexual function and dysfunction, multidisciplinary treatment options for various pelvic support problems and UI, as well as psychologic screening for body dysmorphic disorders, need to be considered in terms of treating both the functional and aesthetic aspects related to cosmetic and reconstructive gynecologic surgery.
Special considerations. The use of energy-based therapies in special populations, such as survivors of breast cancer or other gynecologic cancers, as well as women undergoing chemotherapy, radiation therapy, and hormonal manipulation (particularly with antiestrogenic SERMs and aromatase inhibitors) has not been adequately evaluated. A discussion of the risks, benefits, alternatives, and limited long-term outcome data for energy-based therapies in cancer survivors, as for all patients, must be included for adequate informed consent prior to undertaking these treatments.
Guidelines for appropriate tissue priming, laser settings, and concomitant energy-based technology with local hormone treatment (also known as laser-augmented drug delivery) need to be developed. Comparative long-term studies are needed to determine the safety and effectiveness of these technologies.
Caution advised. Given the lack of long-term safety and effectiveness data on energy-based therapies for the vague indications of vaginal laxity, and even for the well-defined conditions of stress UI and vulvovaginal atrophy, clinicians should exercise caution before promoting treatment, which can be expensive and is not without potential complications, such as vaginal pain, adhesive agglutination, and persistent dryness and dyspareunia.21
Fortunately, many randomized trials on various energy-based devices for gynecologic indications (GSM, stress UI, vaginal laxity, lichen sclerosus) are underway, and results from these studies will help inform future clinical practice and guideline development.
- Kaplan I, Goldman J, Ger R. The treatment of erosions of the uterine cervix by means of the CO2 laser. Obstet Gynecol. 1973;41:795-796.
- Tadir Y, Gaspar A, Lev-Sagie A, et al. Light and energy-based therapeutics for genitourinary syndrome of menopause: consensus and controversies. Lasers Surg Med. 2017;49:137-159.
- Gaspar A, Addamo G, Brandi H. Vaginal fractional CO2 laser: a minimally invasive option for vaginal rejuvenation. Am J Cosmetic Surg. 2011;28:156-162.
- Salvatore S, Leone Roberti Maggiore U, Athanasiou S, et al. Histological study on the effects of microablative fractional CO2 laser on atrophic vaginal tissue: an ex vivo study. Menopause. 2015;22:845-849.
- Benedetto AV. What's new in cosmetic dermatology. Dermatol Clin. 2019;37:117-128.
- US Food and Drug Administration. FDA warns against use of energy-based devices to perform vaginal rejuvenation or vaginal cosmetic procedures: FDA safety communication. https://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm615013.htm. Accessed April 8, 2019.
- Portman DJ, Gass ML; Vulvovaginal Atrophy Terminology Consensus Conference Panel. Genitourinary syndrome of menopause: new terminology for vulvovaginal atrophy from the International Society for the Study of Women's Sexual Health and the North American Menopause Society. Menopause. 2014;21:1063-1068.
- Cruz VL, Steiner ML, Pompei LM, et al. Randomized, double-blind, placebo-controlled clinical trial for evaluating the efficacy of fractional CO2 laser compared with topical estriol in the treatment of vaginal atrophy in postmenopausal women. Menopause. 2018;25:21-28.
- Athanasiou S, Pitsouni E, Grigoradis T, et al. Microablative fractional CO2 laser for the genitourinary syndrome of menopause: up to 12-month results. Menopause. 2019;26:248-255.
- Gaspar A, Brandi H, Gomez V, et al. Efficacy of Erbium:YAG laser treatment compared to topical estriol treatment for symptoms of genitourinary syndrome of menopause. Lasers Surg Med. 2017;49:160-168.
- Gambacciani M, Levancini M, Russo E, et al. Long-term effects of vaginal erbium laser in the treatment of genitourinary syndrome of menopause. Climacteric. 2018;21:148-152.
- Gambacciani M, Levancini M, Cervigni M. Vaginal erbium laser: the second-generation thermotherapy for the genitourinary syndrome of menopause. Climacteric. 2015;18:757-763.
- Ogrinc UB, Sencar S, Lenasi H. Novel minimally invasive laser treatment of urinary incontinence in women. Lasers Surg Med. 2015;47:689-697.
- Okui N. Comparison between erbium-doped yttrium aluminum garnet laser therapy and sling procedures in the treatment of stress and mixed urinary incontinence. World J Urol. 2018. doi:10.1007/s00345-018-2445-x.
- Blaganje M, Scepanovic D, Zgur L, et al. Non-ablative Er:YAG laser therapy effect on stress urinary incontinence related to quality of life and sexual function: a randomized controlled trial. Eur J Obstet Gynecol Reprod Biol. 2018;224:153-158.
- Haylen BT, Maher CF, Barber MD, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic organ prolapse (POP). Int Urogynecologic J. 2016;27:165-194.
- Garcia B, Pardo J. Academic cosmetic gynecology and energy-based therapies: ambiguities, explorations, and the FDA advisories. Int Urogynecol J. 2019;30:1-2.
- Krychman M, Rowan CG, Allan BB, et al. Effect of single-treatment, surface-cooled radiofrequency therapy on vaginal laxity and female sexual function: the VIVEVE I randomized controlled trial. J Sex Med. 2017;14:215-225.
- Shobeiri SA, Kerkhof MH, Minassian VA, et al; IUGA Research and Development Committee. IUGA committee opinion: laser-based vaginal devices for treatment of stress urinary incontinence, genitourinary syndrome of menopause, and vaginal laxity. Int Urogynecol J. 2019;30:371-376.
- Preti M, Vieira-Baptista P, Digesu GA, et al. The clinical role of LASER for vulvar and vaginal treatments in gynecology and female urology: an ICS/ISSVD best practice consensus document. Neurourol Urodyn. 2019;38:1009-1023.
- Gordon C, Gonzales S, Krychman ML. Rethinking the techno vagina: a case series of patient complications following vaginal laser treatment for atrophy. Menopause. 2019;26:423-427.
- Kaplan I, Goldman J, Ger R. The treatment of erosions of the uterine cervix by means of the CO2 laser. Obstet Gynecol. 1973;41:795-796.
- Tadir Y, Gaspar A, Lev-Sagie A, et al. Light and energy-based therapeutics for genitourinary syndrome of menopause: consensus and controversies. Lasers Surg Med. 2017;49:137-159.
- Gaspar A, Addamo G, Brandi H. Vaginal fractional CO2 laser: a minimally invasive option for vaginal rejuvenation. Am J Cosmetic Surg. 2011;28:156-162.
- Salvatore S, Leone Roberti Maggiore U, Athanasiou S, et al. Histological study on the effects of microablative fractional CO2 laser on atrophic vaginal tissue: an ex vivo study. Menopause. 2015;22:845-849.
- Benedetto AV. What's new in cosmetic dermatology. Dermatol Clin. 2019;37:117-128.
- US Food and Drug Administration. FDA warns against use of energy-based devices to perform vaginal rejuvenation or vaginal cosmetic procedures: FDA safety communication. https://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm615013.htm. Accessed April 8, 2019.
- Portman DJ, Gass ML; Vulvovaginal Atrophy Terminology Consensus Conference Panel. Genitourinary syndrome of menopause: new terminology for vulvovaginal atrophy from the International Society for the Study of Women's Sexual Health and the North American Menopause Society. Menopause. 2014;21:1063-1068.
- Cruz VL, Steiner ML, Pompei LM, et al. Randomized, double-blind, placebo-controlled clinical trial for evaluating the efficacy of fractional CO2 laser compared with topical estriol in the treatment of vaginal atrophy in postmenopausal women. Menopause. 2018;25:21-28.
- Athanasiou S, Pitsouni E, Grigoradis T, et al. Microablative fractional CO2 laser for the genitourinary syndrome of menopause: up to 12-month results. Menopause. 2019;26:248-255.
- Gaspar A, Brandi H, Gomez V, et al. Efficacy of Erbium:YAG laser treatment compared to topical estriol treatment for symptoms of genitourinary syndrome of menopause. Lasers Surg Med. 2017;49:160-168.
- Gambacciani M, Levancini M, Russo E, et al. Long-term effects of vaginal erbium laser in the treatment of genitourinary syndrome of menopause. Climacteric. 2018;21:148-152.
- Gambacciani M, Levancini M, Cervigni M. Vaginal erbium laser: the second-generation thermotherapy for the genitourinary syndrome of menopause. Climacteric. 2015;18:757-763.
- Ogrinc UB, Sencar S, Lenasi H. Novel minimally invasive laser treatment of urinary incontinence in women. Lasers Surg Med. 2015;47:689-697.
- Okui N. Comparison between erbium-doped yttrium aluminum garnet laser therapy and sling procedures in the treatment of stress and mixed urinary incontinence. World J Urol. 2018. doi:10.1007/s00345-018-2445-x.
- Blaganje M, Scepanovic D, Zgur L, et al. Non-ablative Er:YAG laser therapy effect on stress urinary incontinence related to quality of life and sexual function: a randomized controlled trial. Eur J Obstet Gynecol Reprod Biol. 2018;224:153-158.
- Haylen BT, Maher CF, Barber MD, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic organ prolapse (POP). Int Urogynecologic J. 2016;27:165-194.
- Garcia B, Pardo J. Academic cosmetic gynecology and energy-based therapies: ambiguities, explorations, and the FDA advisories. Int Urogynecol J. 2019;30:1-2.
- Krychman M, Rowan CG, Allan BB, et al. Effect of single-treatment, surface-cooled radiofrequency therapy on vaginal laxity and female sexual function: the VIVEVE I randomized controlled trial. J Sex Med. 2017;14:215-225.
- Shobeiri SA, Kerkhof MH, Minassian VA, et al; IUGA Research and Development Committee. IUGA committee opinion: laser-based vaginal devices for treatment of stress urinary incontinence, genitourinary syndrome of menopause, and vaginal laxity. Int Urogynecol J. 2019;30:371-376.
- Preti M, Vieira-Baptista P, Digesu GA, et al. The clinical role of LASER for vulvar and vaginal treatments in gynecology and female urology: an ICS/ISSVD best practice consensus document. Neurourol Urodyn. 2019;38:1009-1023.
- Gordon C, Gonzales S, Krychman ML. Rethinking the techno vagina: a case series of patient complications following vaginal laser treatment for atrophy. Menopause. 2019;26:423-427.
Assessing and treating sexual function after vaginal surgery
Sexual dysfunction is challenging for patients and clinicians. Just as sexual function is multidimensional—with physical and psychosocial elements—sexual dysfunction can likewise have multiple contributing factors, and is often divided into dysfunction of desire, arousal, orgasm, and sex-related pain. Addressing each of these dimensions of sexual dysfunction in relationship to pelvic reconstructive surgery is beyond the scope of this article. Here, we focus on aspects of sexual dysfunction most likely to be reported by patients after surgery for pelvic organ prolapse (POP) or urinary incontinence, or for both. We discuss what is known about why sexual dysfunction develops after these procedures; how to assess symptoms when sexual dysfunction occurs; and how best to treat these difficult problems.
CASE Postoperative sexual concerns
Your 62-year-old patient presents 2 weeks after vaginal hysterectomy, uterosacral vault suspension, anterior and posterior colporrhaphy, and retropubic midurethral polypropylene sling placement. She reports feeling tired but otherwise doing well.
The patient returns 8 weeks postoperatively, having just resumed her customary exercise routine, and reports that she is feeling well. Upon questioning, she says that she has not yet attempted to have sexual intercourse with her 70-year-old husband.
The patient returns 6 months later and reports that, although she is doing well overall, she is unable to have sexual intercourse.
How can you help this patient? What next steps in evaluation are indicated? Then, with an understanding of her problem in hand, what treatment options can you offer to her?
Surgery for pelvic-floor disorders and sexual function
The impact of surgery on sexual function is important to discuss with patients preoperatively and postoperatively. Because patients with POP and urinary incontinence have a higher rate of sexual dysfunction at baseline, it is important to know how surgery to correct these conditions can affect sexual function.1 Regrettably, many studies of surgical procedures for POP and urinary incontinence either do not include sexual function outcomes or are not powered to detect differences in these outcomes.
Native-tissue repair. A 2015 systematic review looked at studies of women undergoing native-tissue repair for POP without mesh placement of any kind, including a midurethral sling.2 Based on 9 studies that reported validated sexual function questionnaire scores, investigators determined that sexual function scores generally improved following surgery. Collectively, for studies included in this review that specifically reported the rate of dyspareunia before and after surgery, 47% of women reported improvement in dyspareunia; 39% reported no change; 18% reported deterioration in dyspareunia; and only 4% had de novo dyspareunia.
Colporrhaphy. Posterior colporrhaphy, commonly performed to correct posterior vaginal prolapse, can narrow vaginal caliber and the introitus, potentially causing dyspareunia. Early description of posterior colporrhaphy technique included plication of the levator ani muscles, which was associated with significant risk of dyspareunia postoperatively.3 However, posterior colporrhaphy that involves standard plication of the rectovaginal muscularis or site-specific repair has been reported to have a dyspareunia rate from 7% to 20%.4,5 It is generally recommended, therefore, that levator muscle plication during colporrhaphy be avoided in sexually active women.
Continue to: Vaginal mesh...
Vaginal mesh. Mesh has been used in various surgical procedures to correct pelvic floor disorders. Numerous randomized trials have comparatively evaluated the use of transvaginal polypropylene mesh and native tissue for POP repair, and many of these studies have assessed postoperative sexual function. In a 2013 systematic review on sexual function after POP repair, the authors found no significant difference in postoperative sexual function scores or the dyspareunia rate after vaginal mesh repair (14%) and after native-tissue repair (12%).6
Ask; then ask again
· Talk about sexual function before and after surgery
Remember the basics
· A thorough history and physical exam are paramount
Ask in a different way
· Any of several validated questionnaires can be a valuable adjunct to the history and physical exam
Individualize treatment
· Many patients respond to nonsurgical treatment, but surgical management is necessary in some cases
Studies of postsurgical sexual function are lacking
Important aspects of sexual function—orgasm, arousal, desire, lubrication, sexual satisfaction, effects on the partner—lack studies. A study of 71 sexually active couples assessed sexual function with questionnaires before and after vaginal native-tissue repair and found that, except for orgasm, all domains improved in female questionnaires. In male partners, interest, sexual drive, and overall satisfaction improved, whereas erection, ejaculation, and orgasm remained unchanged.7
Urinary incontinence during sexual intercourse affects approximately 30% of women with overactive bladder or stress incontinence.8 Several reviews have analyzed data on overall sexual function following urinary incontinence surgery:
- After stress incontinence surgery, the rate of coital incontinence was found to be significantly lower (odds ratio, 0.11).9 In this review, 18 studies, comprising more than 1,500 women, were analyzed, with most participants having undergone insertion of a midurethral mesh sling. Most women (55%) reported no change in overall sexual function, based on validated sexual questionnaire scores; 32% reported improvement; and 13% had deterioration in sexual function.
- As for type of midurethral sling, 2 reviews concluded that there is no difference in sexual function outcomes between retropubic and trans‑obturator sling routes.9,10
Although most studies that have looked at POP and incontinence surgeries show either improvement or no change in sexual function, we stress that sexual function is a secondary outcome in most of those studies, which might not be appropriately powered to detect differences in outcomes. Furthermore, although studies describe dyspareunia and overall sexual function in validated questionnaire scores, most do not evaluate other specific domains of sexual function. It remains unclear, therefore, how POP and incontinence surgeries affect orgasm, desire, arousal, satisfaction, and partner sexual domains; more studies are needed to focus on these areas of female sexual function.
How do we assess these patients?
We do know that sexual function is important to women undergoing gynecologic surgery: In a recent qualitative study of women undergoing pelvic reconstruction, patients rated lack of improvement in sexual function following surgery a “very severe” adverse event.11 Unfortunately, however, sexual activity and function is not always measured before gynecologic surgery. Although specific reporting guidelines do not exist for routine gynecologic surgery, a terminology report from the International Urogynecologic Association/International Continence Society (IUGA/ICS) recommends that sexual activity and partner status be evaluated prior to and following surgical treatment as essential outcomes.12 In addition, the report recommends that sexual pain be assessed prior to and following surgical procedures.12
Ascertain sexual health. First, asking your patients simple questions about sexual function, pain, and bother before and after surgery opens the door to dialogue that allows them, and their partner, to express concerns to you in a safe environment. It also allows you to better understand the significant impact of your surgical interventions on their sexual health.
Questionnaires. Objective measures of vaginal blood flow and engorgement exist, but assessment of sexual activity in the clinical setting is largely limited to self-assessment with questionnaires. Incorporating simple questions, such as “Are you sexually active?,” “Do you have any problems with sexual activity?,” and “Do you have pain with activity?” are likely to be as effective as a more detailed interview and can identify women with sexual concerns.13 Many clinicians are put at a disadvantage, however, because they are faced with the difficult situation of addressing postoperative sexual problems without knowing whether the patient had such reports prior to surgery.
Continue to: Aside from simple screening tools...
Aside from simple screening tools, a number of sexual function questionnaires have been developed. Some are generic, and others are condition-specific:
- Generic questionnaires are typically designed to address the function of a range of women. For example, the Female Sexual Function Index comprises 19 questions. Domains include orgasm, desire, arousal, lubrication, pain and satisfaction.14
- Condition-specific questionnaires of sexual function each have been validated in their target population so that they measure nuances in sexual health relevant to that population. The Pelvic Organ Prolapse/Incontinence Sexual Questionnaire—IUGA-Revised includes questions about the domains listed for the generic Index (above) plus questions about the impact of coital incontinence or bulge symptoms on sexual function.12
History-taking. If a woman identifies a problem with sexual function, a thorough history helps elicit whether the condition is lifelong or acquired, situational or general, and, most important, whether or not it is bothersome to her.14,15 It is important not to make assumptions when pursuing this part of the history, and to encourage patients to be candid about how they have sex and with whom.
Physical examination. The patient should undergo a complete physical exam, including 1) a detailed pelvic exam assessing the vulva, vagina, and pelvic-floor musculature, and 2) estrogenization of the tissue.
Partner concerns. For women who have a partner, addressing the concerns of that partner following gynecologic surgery can be useful to the couple: The partner might be concerned about inflicting pain or doing damage during sex after gynecologic surgery.
CASE Informative discussion
While ascertaining her sexual symptoms, your patient reveals to you that she has attempted sexual intercourse on 3 occasions; each time, penetration was too painful to continue. She tells you she did not have this problem before surgery.
The patient says that she has tried water-based lubricants and is using vaginal estrogen 3 times per week, but “nothing helps.” She reports that she is arousable and has been able to achieve orgasm with clitoral stimulation, but would like to have vaginal intercourse. Her husband does have erectile dysfunction, which, she tells you, can make penetration difficult.
On physical examination, you detect mild atrophy. Vaginal length is 9 cm; no narrowing or scarring of the vaginal introitus or canal is seen. No mesh is visible or palpable. The paths of the midurethral sling arms are nontender. However, levator muscles are tender and tense bilaterally.
Given these findings on examination, what steps can you take to relieve your patient’s pain?
What can we offer these patients?
Treating sexual dysfunction after pelvic reconstructive surgery must, as emphasized earlier, be guided by a careful history and physical exam. Doing so is critical to determining the underlying cause. Whenever feasible, offer the least invasive treatment.
The IUGA/ICS terminology report describes several symptoms of postoperative sexual dysfunction12:
- de novo sexual dysfunction
- de novo dyspareunia
- shortened vagina
- tight vagina (introital or vaginal narrowing, or both)
- scarred vagina (including mesh-related problems)
- hispareunia (pain experienced by a male partner after intercourse).
Of course, any one or combination of these symptoms can be present in a given patient. Furthermore, de novo sexual dysfunction, de novo dyspareunia, and hispareunia can have various underlying causes—again, underscoring the importance of the history and exam in determining treatment.
Continue to: Nonsurgical treatment...
Nonsurgical treatment
Nonhormonal vaginal lubricants and moisturizers; vaginal estrogen therapy. Although, in older women, vaginal atrophy is often not a new diagnosis postsurgically, the condition might have been untreated preoperatively and might therefore come into play in sexual dysfunction postoperatively. If a patient reports vaginal dryness or pain upon penetration, assess for vaginal atrophy and, if present, treat accordingly.
Vaginal dilation and physical therapy. A shortened, tight, or scarred vagina might be amenable to therapy with vaginal dilators and physical therapy, but might ultimately require surgery.
Pelvic-floor myalgia or spasm can develop after surgery or, as with atrophy, might have existed preoperatively but was left untreated. Pelvic-floor myalgia should be suspected if the patient describes difficult penetration or a feeling of tightness, even though scarring or constriction of the vagina is not seen on examination. Physical therapy with a specialist in pelvic floor treatment is a first-line treatment for pelvic-floor myalgia,16 and is likely to be a helpful adjunct in many situations, including mesh-related sexual problems.17
Oral or vaginal medications to relax pelvic-floor muscle spasm are an option, although efficacy data are limited. If pain is of longstanding duration and is thought to have a neuropathic component, successful use of tricyclic antidepressants, neuroleptics, and serotonin–norepinephrine reuptake inhibitors has been reported.18
Surgery
Data are sparse regarding surgical treatment of female sexual dysfunction after pelvic reconstructive surgery. Again, it is clear, however, that the key is carefully assessing each patient and then individualizing treatment. Patients can have any type of dysfunction that a patient who hasn’t had surgery can—but is also at risk of conditions directly related to surgery.
In any patient who has had mesh placed as part of surgery, thorough examination is necessary to determine whether or not the implant is involved in sexual dysfunction. If the dysfunction is an apparent result of surgery performed by another surgeon, make every effort to review the operative report to determine which material was implanted and how it was placed.
Trigger-point injection can be attempted in a patient who has site-specific tenderness that is not clearly associated with tissue obstruction of the vagina or mesh erosion.12,19 Even in areas of apparent banding or scarring related to mesh, trigger-point injection can be attempted to alleviate pain. How often trigger-point injections should be performed is understudied.
If, on examination, tenderness that replicates the dyspareunia is elicited when palpating the levator or obturator internus muscle, pelvic-floor muscle trigger-point injection can be offered (although physical therapy is first-line treatment). Trigger-point injection also can be a useful adjunct in women who have another identified cause of pain but also have developed pelvic-floor muscle spasm.
Not addressing concomitant pelvic-floor myalgia could prevent successful treatment of pain. Inclusion of a pudendal block also might help to alleviate pain.
Continue to: Surgical resection...
Surgical resection. If a skin bridge is clearly observed at the introitus, or if the introitus has been overly narrowed by perineorrhaphy but the remainder of the vagina has adequate length and caliber, surgical resection of the skin bridge might relieve symptoms of difficult penetration. In the case of obstructive perineorrhaphy, an attempt at reversal can be made by incising the perineum vertically but then reapproximating the edges transversely—sometimes referred to as reverse perineorrhaphy.
If scar tissue found elsewhere in the vagina might obstruct penetration, this condition might also be amenable to resection. When scarring is annular, relaxing incisions can be made bilaterally to relieve tension on that tissue; alternatively, it might be necessary to perform a Z-plasty. Nearly always, severe scarring is accompanied by levator myalgia, and a combined approach of surgery and physical therapy is necessary.
Neovagina. It is possible to find vaginal stenosis or shortening, to a varying degree, after surgical prolapse repair, with or without mesh or graft. As discussed, vaginal dilation should be offered but, if this is ineffective, the patient might be a candidate for surgical creation of a neovagina. Numerous techniques have been described for patients with congenital vaginal agenesis, with a few reports of similar techniques used to treat iatrogenic vaginal stenosis or obliteration.
The general principle of all neovagina procedures is to create a space between bladder and rectum of adequate caliber and length for desired sexual function. Reported techniques include a thigh or buttock skin graft, use of bowel or peritoneum, and, recently, a buccal mucosa graft.20,21
Resection or excision of mesh. In patients who develop sexual dysfunction after mesh placement, the problem can be caused by exposure of the mesh in the vagina or erosion into another organ, but can also arise in the absence of exposure or erosion. Patients might have tenderness to palpation at points where the mesh is palpable through the mucosa but not exposed.
Again, complete investigation is necessary to look for mesh involvement in the vagina and, depending on the type of implant, other adjacent organs. Assessing partner symptoms, such as pain and scratches, also can be telling.
If there is palpable tenderness on vaginal examination of the mesh, resection of the vaginal portion might be an option.17 Complete excision of mesh implants can be morbid, however, and might not provide a better outcome than partial excision. The risk of morbidity from complete mesh excision must be weighed against the likelihood that partial excision will not resolve pain and that the patient will require further excision subsequently.17,22 Excising fragmented mesh can be difficult; making every attempt to understand the contribution of mesh to sexual dysfunction is therefore critical to determining how, and how much of, the mesh comes out at the first attempt.
Last, for any woman who opts for surgical intervention to treat pain, you should engage in a discussion to emphasize the multidimensional nature of sexual function and the fact that any surgical intervention might not completely resolve her dysfunction.
Continue to: CASE Discussing options...
CASE Discussing options, choosing an intervention
You discuss the examination findings (no shortening or narrowing of the vagina) with the patient. She is relieved but puzzled as to why she cannot have intercourse. You discuss the tension and t
At 3-month follow-up, she reports great improvement. She is able to have intercourse, although she says she still has discomfort sometimes. She continues to work with the pelvic floor physical therapist and feels optimistic. You plan to see her in 6 months but counsel her to call if symptoms are not improving or are worsening.
It is difficult to counsel patients about sexual function after pelvic reconstructive surgery because data that could guide identification of problems (and how to treat them) are incomplete. Assessingsexual function preoperatively and having an open conversation about risks and benefits of surgery, with specific mention of its impact on sexual health, are critical (see “Key touchpoints in managing sexual dysfunction after pelvic reconstructive surgery”).
It is also crucial to assess sexual function postoperatively as a matter of routine. Validated questionnaires can be a useful adjunct to a thorough history and physical exam, and can help guide your discussions.
Treatment of postop sexual dysfunction must, first, account for the complex nature of sexual function and, second, be individualized, starting with the least invasive options, when feasible.
- Rogers RG. Sexual function in women with pelvic floor disorders. Can Urol Assoc J. 2013;7:S199-S201.
- Jha S, Gray T. A systematic review and meta-analysis of the impact of native tissue repair for pelvic organ prolapse on sexual function. Int Urogynecol J. 2015;26:321-327.
- Thompson JC, Rogers RG. Surgical management for pelvic organ prolapse and its impact on sexual function. Sex Med Rev. 2016;4:213-220.
- Sung VW, Rardin CR, Raker CA, et al. Porcine subintestinal submucosal graft augmentation for rectocele repair: a randomized controlled trial. Obstet Gynecol. 2012;119:125-133.
- Paraiso MF, Barber MD, Muir TW, et al. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
- Dietz V, Maher C. Pelvic organ prolapse and sexual function. Int Urogynecol J. 2013;24:1853-1857.
- Kuhn A, Brunnmayr G, Stadlmayr W, et al. Male and female sexual function after surgical repair of female organ prolapse. J Sex Med. 2009;6:1324-1334.
- Gray T, Li W, Campbell P, et al. Evaluation of coital incontinence by electronic questionnaire: prevalence, associations and outcomes in women attending a urogynaecology clinic. Int Urogynecol J. 2018;29:969-978.
- Jha S, Ammenbal M, Metwally M. Impact of incontinence surgery on sexual function: a systematic review and meta-analysis. J Sex Med. 2012;9:34-43.
- Schimpf MO, Rahn DD, Wheeler TL, et al; Society of Gynecologic Surgeons Systematic Review Group. Sling surgery for stress urinary incontinence in women: a systematic review and metaanalysis. Am J Obstet Gynecol. 2014;211:71.e1-e71.e27.
- Dunivan GC, Sussman AL, Jelovsek JE, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Gaining the patient perspective on pelvic floor disorders’ surgical adverse events. Am J Obstet Gynecol. 2019;220:185.e1-e185.e10.
- Rogers RG, Pauls RN, Thakar R, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for the assessment of sexual health of women with pelvic floor dysfunction. Int Urogynecol J. 2018;29:647-666.
- Plouffe L Jr. Screening for sexual problems through a simple questionnaire. Am J Obstet Gynecol. 1985;151:166-169.
- Hatzichristou D, Rosen RC, Derogatis LR, et al. Recommendations for the clinical evaluation of men and women with sexual dysfunction. J Sex Med. 2010;7:337-348.
- McCabe MP, Sharlip ID, Atalla E, et al. Definition of sexual dysfunctions in women and men: a consensus statement from the Fourth International Consultation of Sexual Medicine 2015. J Sex Med. 2015;13:135-143.
- Berghmans B. Physiotherapy for pelvic pain and female sexual dysfunction: an untapped resource. Int Urogynecol J. 2018;29:631-638.
- Cundiff GW, Quinlan DJ, van Rensburg JA, et al. Foundation for an evidence-informed algorithm for treating pelvic floor mesh complications: a review. BJOG. 2018;125:1026-1037.
- Steege JF, Siedhoff MT. Chronic pelvic pain. Obstet Gynecol. 2014;124:616-629.
- Wehbe SA, Whitmore K, Kellogg-Spadt S. Urogenital complaints and female sexual dysfunction (part 1). J Sex Med. 2010;7:1704-1713.
- Grimsby GM, Bradshaw K, Baker LA. Autologous buccal mucosa graft augmentation for foreshortened vagina. Obstet Gynecol. 2014;123:947-950.
- Morley GW, DeLancey JO. Full-thickness skin graft vaginoplasty for treatment of the stenotic or foreshortened vagina. Obstet Gynecol. 1991;77:485-489.
- Pickett SD, Barenberg B, Quiroz LH, et al. The significant morbidity of removing pelvic mesh from multiple vaginal compartments. Obstet Gynecol. 2015;125:1418-1422.
Sexual dysfunction is challenging for patients and clinicians. Just as sexual function is multidimensional—with physical and psychosocial elements—sexual dysfunction can likewise have multiple contributing factors, and is often divided into dysfunction of desire, arousal, orgasm, and sex-related pain. Addressing each of these dimensions of sexual dysfunction in relationship to pelvic reconstructive surgery is beyond the scope of this article. Here, we focus on aspects of sexual dysfunction most likely to be reported by patients after surgery for pelvic organ prolapse (POP) or urinary incontinence, or for both. We discuss what is known about why sexual dysfunction develops after these procedures; how to assess symptoms when sexual dysfunction occurs; and how best to treat these difficult problems.
CASE Postoperative sexual concerns
Your 62-year-old patient presents 2 weeks after vaginal hysterectomy, uterosacral vault suspension, anterior and posterior colporrhaphy, and retropubic midurethral polypropylene sling placement. She reports feeling tired but otherwise doing well.
The patient returns 8 weeks postoperatively, having just resumed her customary exercise routine, and reports that she is feeling well. Upon questioning, she says that she has not yet attempted to have sexual intercourse with her 70-year-old husband.
The patient returns 6 months later and reports that, although she is doing well overall, she is unable to have sexual intercourse.
How can you help this patient? What next steps in evaluation are indicated? Then, with an understanding of her problem in hand, what treatment options can you offer to her?
Surgery for pelvic-floor disorders and sexual function
The impact of surgery on sexual function is important to discuss with patients preoperatively and postoperatively. Because patients with POP and urinary incontinence have a higher rate of sexual dysfunction at baseline, it is important to know how surgery to correct these conditions can affect sexual function.1 Regrettably, many studies of surgical procedures for POP and urinary incontinence either do not include sexual function outcomes or are not powered to detect differences in these outcomes.
Native-tissue repair. A 2015 systematic review looked at studies of women undergoing native-tissue repair for POP without mesh placement of any kind, including a midurethral sling.2 Based on 9 studies that reported validated sexual function questionnaire scores, investigators determined that sexual function scores generally improved following surgery. Collectively, for studies included in this review that specifically reported the rate of dyspareunia before and after surgery, 47% of women reported improvement in dyspareunia; 39% reported no change; 18% reported deterioration in dyspareunia; and only 4% had de novo dyspareunia.
Colporrhaphy. Posterior colporrhaphy, commonly performed to correct posterior vaginal prolapse, can narrow vaginal caliber and the introitus, potentially causing dyspareunia. Early description of posterior colporrhaphy technique included plication of the levator ani muscles, which was associated with significant risk of dyspareunia postoperatively.3 However, posterior colporrhaphy that involves standard plication of the rectovaginal muscularis or site-specific repair has been reported to have a dyspareunia rate from 7% to 20%.4,5 It is generally recommended, therefore, that levator muscle plication during colporrhaphy be avoided in sexually active women.
Continue to: Vaginal mesh...
Vaginal mesh. Mesh has been used in various surgical procedures to correct pelvic floor disorders. Numerous randomized trials have comparatively evaluated the use of transvaginal polypropylene mesh and native tissue for POP repair, and many of these studies have assessed postoperative sexual function. In a 2013 systematic review on sexual function after POP repair, the authors found no significant difference in postoperative sexual function scores or the dyspareunia rate after vaginal mesh repair (14%) and after native-tissue repair (12%).6
Ask; then ask again
· Talk about sexual function before and after surgery
Remember the basics
· A thorough history and physical exam are paramount
Ask in a different way
· Any of several validated questionnaires can be a valuable adjunct to the history and physical exam
Individualize treatment
· Many patients respond to nonsurgical treatment, but surgical management is necessary in some cases
Studies of postsurgical sexual function are lacking
Important aspects of sexual function—orgasm, arousal, desire, lubrication, sexual satisfaction, effects on the partner—lack studies. A study of 71 sexually active couples assessed sexual function with questionnaires before and after vaginal native-tissue repair and found that, except for orgasm, all domains improved in female questionnaires. In male partners, interest, sexual drive, and overall satisfaction improved, whereas erection, ejaculation, and orgasm remained unchanged.7
Urinary incontinence during sexual intercourse affects approximately 30% of women with overactive bladder or stress incontinence.8 Several reviews have analyzed data on overall sexual function following urinary incontinence surgery:
- After stress incontinence surgery, the rate of coital incontinence was found to be significantly lower (odds ratio, 0.11).9 In this review, 18 studies, comprising more than 1,500 women, were analyzed, with most participants having undergone insertion of a midurethral mesh sling. Most women (55%) reported no change in overall sexual function, based on validated sexual questionnaire scores; 32% reported improvement; and 13% had deterioration in sexual function.
- As for type of midurethral sling, 2 reviews concluded that there is no difference in sexual function outcomes between retropubic and trans‑obturator sling routes.9,10
Although most studies that have looked at POP and incontinence surgeries show either improvement or no change in sexual function, we stress that sexual function is a secondary outcome in most of those studies, which might not be appropriately powered to detect differences in outcomes. Furthermore, although studies describe dyspareunia and overall sexual function in validated questionnaire scores, most do not evaluate other specific domains of sexual function. It remains unclear, therefore, how POP and incontinence surgeries affect orgasm, desire, arousal, satisfaction, and partner sexual domains; more studies are needed to focus on these areas of female sexual function.
How do we assess these patients?
We do know that sexual function is important to women undergoing gynecologic surgery: In a recent qualitative study of women undergoing pelvic reconstruction, patients rated lack of improvement in sexual function following surgery a “very severe” adverse event.11 Unfortunately, however, sexual activity and function is not always measured before gynecologic surgery. Although specific reporting guidelines do not exist for routine gynecologic surgery, a terminology report from the International Urogynecologic Association/International Continence Society (IUGA/ICS) recommends that sexual activity and partner status be evaluated prior to and following surgical treatment as essential outcomes.12 In addition, the report recommends that sexual pain be assessed prior to and following surgical procedures.12
Ascertain sexual health. First, asking your patients simple questions about sexual function, pain, and bother before and after surgery opens the door to dialogue that allows them, and their partner, to express concerns to you in a safe environment. It also allows you to better understand the significant impact of your surgical interventions on their sexual health.
Questionnaires. Objective measures of vaginal blood flow and engorgement exist, but assessment of sexual activity in the clinical setting is largely limited to self-assessment with questionnaires. Incorporating simple questions, such as “Are you sexually active?,” “Do you have any problems with sexual activity?,” and “Do you have pain with activity?” are likely to be as effective as a more detailed interview and can identify women with sexual concerns.13 Many clinicians are put at a disadvantage, however, because they are faced with the difficult situation of addressing postoperative sexual problems without knowing whether the patient had such reports prior to surgery.
Continue to: Aside from simple screening tools...
Aside from simple screening tools, a number of sexual function questionnaires have been developed. Some are generic, and others are condition-specific:
- Generic questionnaires are typically designed to address the function of a range of women. For example, the Female Sexual Function Index comprises 19 questions. Domains include orgasm, desire, arousal, lubrication, pain and satisfaction.14
- Condition-specific questionnaires of sexual function each have been validated in their target population so that they measure nuances in sexual health relevant to that population. The Pelvic Organ Prolapse/Incontinence Sexual Questionnaire—IUGA-Revised includes questions about the domains listed for the generic Index (above) plus questions about the impact of coital incontinence or bulge symptoms on sexual function.12
History-taking. If a woman identifies a problem with sexual function, a thorough history helps elicit whether the condition is lifelong or acquired, situational or general, and, most important, whether or not it is bothersome to her.14,15 It is important not to make assumptions when pursuing this part of the history, and to encourage patients to be candid about how they have sex and with whom.
Physical examination. The patient should undergo a complete physical exam, including 1) a detailed pelvic exam assessing the vulva, vagina, and pelvic-floor musculature, and 2) estrogenization of the tissue.
Partner concerns. For women who have a partner, addressing the concerns of that partner following gynecologic surgery can be useful to the couple: The partner might be concerned about inflicting pain or doing damage during sex after gynecologic surgery.
CASE Informative discussion
While ascertaining her sexual symptoms, your patient reveals to you that she has attempted sexual intercourse on 3 occasions; each time, penetration was too painful to continue. She tells you she did not have this problem before surgery.
The patient says that she has tried water-based lubricants and is using vaginal estrogen 3 times per week, but “nothing helps.” She reports that she is arousable and has been able to achieve orgasm with clitoral stimulation, but would like to have vaginal intercourse. Her husband does have erectile dysfunction, which, she tells you, can make penetration difficult.
On physical examination, you detect mild atrophy. Vaginal length is 9 cm; no narrowing or scarring of the vaginal introitus or canal is seen. No mesh is visible or palpable. The paths of the midurethral sling arms are nontender. However, levator muscles are tender and tense bilaterally.
Given these findings on examination, what steps can you take to relieve your patient’s pain?
What can we offer these patients?
Treating sexual dysfunction after pelvic reconstructive surgery must, as emphasized earlier, be guided by a careful history and physical exam. Doing so is critical to determining the underlying cause. Whenever feasible, offer the least invasive treatment.
The IUGA/ICS terminology report describes several symptoms of postoperative sexual dysfunction12:
- de novo sexual dysfunction
- de novo dyspareunia
- shortened vagina
- tight vagina (introital or vaginal narrowing, or both)
- scarred vagina (including mesh-related problems)
- hispareunia (pain experienced by a male partner after intercourse).
Of course, any one or combination of these symptoms can be present in a given patient. Furthermore, de novo sexual dysfunction, de novo dyspareunia, and hispareunia can have various underlying causes—again, underscoring the importance of the history and exam in determining treatment.
Continue to: Nonsurgical treatment...
Nonsurgical treatment
Nonhormonal vaginal lubricants and moisturizers; vaginal estrogen therapy. Although, in older women, vaginal atrophy is often not a new diagnosis postsurgically, the condition might have been untreated preoperatively and might therefore come into play in sexual dysfunction postoperatively. If a patient reports vaginal dryness or pain upon penetration, assess for vaginal atrophy and, if present, treat accordingly.
Vaginal dilation and physical therapy. A shortened, tight, or scarred vagina might be amenable to therapy with vaginal dilators and physical therapy, but might ultimately require surgery.
Pelvic-floor myalgia or spasm can develop after surgery or, as with atrophy, might have existed preoperatively but was left untreated. Pelvic-floor myalgia should be suspected if the patient describes difficult penetration or a feeling of tightness, even though scarring or constriction of the vagina is not seen on examination. Physical therapy with a specialist in pelvic floor treatment is a first-line treatment for pelvic-floor myalgia,16 and is likely to be a helpful adjunct in many situations, including mesh-related sexual problems.17
Oral or vaginal medications to relax pelvic-floor muscle spasm are an option, although efficacy data are limited. If pain is of longstanding duration and is thought to have a neuropathic component, successful use of tricyclic antidepressants, neuroleptics, and serotonin–norepinephrine reuptake inhibitors has been reported.18
Surgery
Data are sparse regarding surgical treatment of female sexual dysfunction after pelvic reconstructive surgery. Again, it is clear, however, that the key is carefully assessing each patient and then individualizing treatment. Patients can have any type of dysfunction that a patient who hasn’t had surgery can—but is also at risk of conditions directly related to surgery.
In any patient who has had mesh placed as part of surgery, thorough examination is necessary to determine whether or not the implant is involved in sexual dysfunction. If the dysfunction is an apparent result of surgery performed by another surgeon, make every effort to review the operative report to determine which material was implanted and how it was placed.
Trigger-point injection can be attempted in a patient who has site-specific tenderness that is not clearly associated with tissue obstruction of the vagina or mesh erosion.12,19 Even in areas of apparent banding or scarring related to mesh, trigger-point injection can be attempted to alleviate pain. How often trigger-point injections should be performed is understudied.
If, on examination, tenderness that replicates the dyspareunia is elicited when palpating the levator or obturator internus muscle, pelvic-floor muscle trigger-point injection can be offered (although physical therapy is first-line treatment). Trigger-point injection also can be a useful adjunct in women who have another identified cause of pain but also have developed pelvic-floor muscle spasm.
Not addressing concomitant pelvic-floor myalgia could prevent successful treatment of pain. Inclusion of a pudendal block also might help to alleviate pain.
Continue to: Surgical resection...
Surgical resection. If a skin bridge is clearly observed at the introitus, or if the introitus has been overly narrowed by perineorrhaphy but the remainder of the vagina has adequate length and caliber, surgical resection of the skin bridge might relieve symptoms of difficult penetration. In the case of obstructive perineorrhaphy, an attempt at reversal can be made by incising the perineum vertically but then reapproximating the edges transversely—sometimes referred to as reverse perineorrhaphy.
If scar tissue found elsewhere in the vagina might obstruct penetration, this condition might also be amenable to resection. When scarring is annular, relaxing incisions can be made bilaterally to relieve tension on that tissue; alternatively, it might be necessary to perform a Z-plasty. Nearly always, severe scarring is accompanied by levator myalgia, and a combined approach of surgery and physical therapy is necessary.
Neovagina. It is possible to find vaginal stenosis or shortening, to a varying degree, after surgical prolapse repair, with or without mesh or graft. As discussed, vaginal dilation should be offered but, if this is ineffective, the patient might be a candidate for surgical creation of a neovagina. Numerous techniques have been described for patients with congenital vaginal agenesis, with a few reports of similar techniques used to treat iatrogenic vaginal stenosis or obliteration.
The general principle of all neovagina procedures is to create a space between bladder and rectum of adequate caliber and length for desired sexual function. Reported techniques include a thigh or buttock skin graft, use of bowel or peritoneum, and, recently, a buccal mucosa graft.20,21
Resection or excision of mesh. In patients who develop sexual dysfunction after mesh placement, the problem can be caused by exposure of the mesh in the vagina or erosion into another organ, but can also arise in the absence of exposure or erosion. Patients might have tenderness to palpation at points where the mesh is palpable through the mucosa but not exposed.
Again, complete investigation is necessary to look for mesh involvement in the vagina and, depending on the type of implant, other adjacent organs. Assessing partner symptoms, such as pain and scratches, also can be telling.
If there is palpable tenderness on vaginal examination of the mesh, resection of the vaginal portion might be an option.17 Complete excision of mesh implants can be morbid, however, and might not provide a better outcome than partial excision. The risk of morbidity from complete mesh excision must be weighed against the likelihood that partial excision will not resolve pain and that the patient will require further excision subsequently.17,22 Excising fragmented mesh can be difficult; making every attempt to understand the contribution of mesh to sexual dysfunction is therefore critical to determining how, and how much of, the mesh comes out at the first attempt.
Last, for any woman who opts for surgical intervention to treat pain, you should engage in a discussion to emphasize the multidimensional nature of sexual function and the fact that any surgical intervention might not completely resolve her dysfunction.
Continue to: CASE Discussing options...
CASE Discussing options, choosing an intervention
You discuss the examination findings (no shortening or narrowing of the vagina) with the patient. She is relieved but puzzled as to why she cannot have intercourse. You discuss the tension and t
At 3-month follow-up, she reports great improvement. She is able to have intercourse, although she says she still has discomfort sometimes. She continues to work with the pelvic floor physical therapist and feels optimistic. You plan to see her in 6 months but counsel her to call if symptoms are not improving or are worsening.
It is difficult to counsel patients about sexual function after pelvic reconstructive surgery because data that could guide identification of problems (and how to treat them) are incomplete. Assessingsexual function preoperatively and having an open conversation about risks and benefits of surgery, with specific mention of its impact on sexual health, are critical (see “Key touchpoints in managing sexual dysfunction after pelvic reconstructive surgery”).
It is also crucial to assess sexual function postoperatively as a matter of routine. Validated questionnaires can be a useful adjunct to a thorough history and physical exam, and can help guide your discussions.
Treatment of postop sexual dysfunction must, first, account for the complex nature of sexual function and, second, be individualized, starting with the least invasive options, when feasible.
Sexual dysfunction is challenging for patients and clinicians. Just as sexual function is multidimensional—with physical and psychosocial elements—sexual dysfunction can likewise have multiple contributing factors, and is often divided into dysfunction of desire, arousal, orgasm, and sex-related pain. Addressing each of these dimensions of sexual dysfunction in relationship to pelvic reconstructive surgery is beyond the scope of this article. Here, we focus on aspects of sexual dysfunction most likely to be reported by patients after surgery for pelvic organ prolapse (POP) or urinary incontinence, or for both. We discuss what is known about why sexual dysfunction develops after these procedures; how to assess symptoms when sexual dysfunction occurs; and how best to treat these difficult problems.
CASE Postoperative sexual concerns
Your 62-year-old patient presents 2 weeks after vaginal hysterectomy, uterosacral vault suspension, anterior and posterior colporrhaphy, and retropubic midurethral polypropylene sling placement. She reports feeling tired but otherwise doing well.
The patient returns 8 weeks postoperatively, having just resumed her customary exercise routine, and reports that she is feeling well. Upon questioning, she says that she has not yet attempted to have sexual intercourse with her 70-year-old husband.
The patient returns 6 months later and reports that, although she is doing well overall, she is unable to have sexual intercourse.
How can you help this patient? What next steps in evaluation are indicated? Then, with an understanding of her problem in hand, what treatment options can you offer to her?
Surgery for pelvic-floor disorders and sexual function
The impact of surgery on sexual function is important to discuss with patients preoperatively and postoperatively. Because patients with POP and urinary incontinence have a higher rate of sexual dysfunction at baseline, it is important to know how surgery to correct these conditions can affect sexual function.1 Regrettably, many studies of surgical procedures for POP and urinary incontinence either do not include sexual function outcomes or are not powered to detect differences in these outcomes.
Native-tissue repair. A 2015 systematic review looked at studies of women undergoing native-tissue repair for POP without mesh placement of any kind, including a midurethral sling.2 Based on 9 studies that reported validated sexual function questionnaire scores, investigators determined that sexual function scores generally improved following surgery. Collectively, for studies included in this review that specifically reported the rate of dyspareunia before and after surgery, 47% of women reported improvement in dyspareunia; 39% reported no change; 18% reported deterioration in dyspareunia; and only 4% had de novo dyspareunia.
Colporrhaphy. Posterior colporrhaphy, commonly performed to correct posterior vaginal prolapse, can narrow vaginal caliber and the introitus, potentially causing dyspareunia. Early description of posterior colporrhaphy technique included plication of the levator ani muscles, which was associated with significant risk of dyspareunia postoperatively.3 However, posterior colporrhaphy that involves standard plication of the rectovaginal muscularis or site-specific repair has been reported to have a dyspareunia rate from 7% to 20%.4,5 It is generally recommended, therefore, that levator muscle plication during colporrhaphy be avoided in sexually active women.
Continue to: Vaginal mesh...
Vaginal mesh. Mesh has been used in various surgical procedures to correct pelvic floor disorders. Numerous randomized trials have comparatively evaluated the use of transvaginal polypropylene mesh and native tissue for POP repair, and many of these studies have assessed postoperative sexual function. In a 2013 systematic review on sexual function after POP repair, the authors found no significant difference in postoperative sexual function scores or the dyspareunia rate after vaginal mesh repair (14%) and after native-tissue repair (12%).6
Ask; then ask again
· Talk about sexual function before and after surgery
Remember the basics
· A thorough history and physical exam are paramount
Ask in a different way
· Any of several validated questionnaires can be a valuable adjunct to the history and physical exam
Individualize treatment
· Many patients respond to nonsurgical treatment, but surgical management is necessary in some cases
Studies of postsurgical sexual function are lacking
Important aspects of sexual function—orgasm, arousal, desire, lubrication, sexual satisfaction, effects on the partner—lack studies. A study of 71 sexually active couples assessed sexual function with questionnaires before and after vaginal native-tissue repair and found that, except for orgasm, all domains improved in female questionnaires. In male partners, interest, sexual drive, and overall satisfaction improved, whereas erection, ejaculation, and orgasm remained unchanged.7
Urinary incontinence during sexual intercourse affects approximately 30% of women with overactive bladder or stress incontinence.8 Several reviews have analyzed data on overall sexual function following urinary incontinence surgery:
- After stress incontinence surgery, the rate of coital incontinence was found to be significantly lower (odds ratio, 0.11).9 In this review, 18 studies, comprising more than 1,500 women, were analyzed, with most participants having undergone insertion of a midurethral mesh sling. Most women (55%) reported no change in overall sexual function, based on validated sexual questionnaire scores; 32% reported improvement; and 13% had deterioration in sexual function.
- As for type of midurethral sling, 2 reviews concluded that there is no difference in sexual function outcomes between retropubic and trans‑obturator sling routes.9,10
Although most studies that have looked at POP and incontinence surgeries show either improvement or no change in sexual function, we stress that sexual function is a secondary outcome in most of those studies, which might not be appropriately powered to detect differences in outcomes. Furthermore, although studies describe dyspareunia and overall sexual function in validated questionnaire scores, most do not evaluate other specific domains of sexual function. It remains unclear, therefore, how POP and incontinence surgeries affect orgasm, desire, arousal, satisfaction, and partner sexual domains; more studies are needed to focus on these areas of female sexual function.
How do we assess these patients?
We do know that sexual function is important to women undergoing gynecologic surgery: In a recent qualitative study of women undergoing pelvic reconstruction, patients rated lack of improvement in sexual function following surgery a “very severe” adverse event.11 Unfortunately, however, sexual activity and function is not always measured before gynecologic surgery. Although specific reporting guidelines do not exist for routine gynecologic surgery, a terminology report from the International Urogynecologic Association/International Continence Society (IUGA/ICS) recommends that sexual activity and partner status be evaluated prior to and following surgical treatment as essential outcomes.12 In addition, the report recommends that sexual pain be assessed prior to and following surgical procedures.12
Ascertain sexual health. First, asking your patients simple questions about sexual function, pain, and bother before and after surgery opens the door to dialogue that allows them, and their partner, to express concerns to you in a safe environment. It also allows you to better understand the significant impact of your surgical interventions on their sexual health.
Questionnaires. Objective measures of vaginal blood flow and engorgement exist, but assessment of sexual activity in the clinical setting is largely limited to self-assessment with questionnaires. Incorporating simple questions, such as “Are you sexually active?,” “Do you have any problems with sexual activity?,” and “Do you have pain with activity?” are likely to be as effective as a more detailed interview and can identify women with sexual concerns.13 Many clinicians are put at a disadvantage, however, because they are faced with the difficult situation of addressing postoperative sexual problems without knowing whether the patient had such reports prior to surgery.
Continue to: Aside from simple screening tools...
Aside from simple screening tools, a number of sexual function questionnaires have been developed. Some are generic, and others are condition-specific:
- Generic questionnaires are typically designed to address the function of a range of women. For example, the Female Sexual Function Index comprises 19 questions. Domains include orgasm, desire, arousal, lubrication, pain and satisfaction.14
- Condition-specific questionnaires of sexual function each have been validated in their target population so that they measure nuances in sexual health relevant to that population. The Pelvic Organ Prolapse/Incontinence Sexual Questionnaire—IUGA-Revised includes questions about the domains listed for the generic Index (above) plus questions about the impact of coital incontinence or bulge symptoms on sexual function.12
History-taking. If a woman identifies a problem with sexual function, a thorough history helps elicit whether the condition is lifelong or acquired, situational or general, and, most important, whether or not it is bothersome to her.14,15 It is important not to make assumptions when pursuing this part of the history, and to encourage patients to be candid about how they have sex and with whom.
Physical examination. The patient should undergo a complete physical exam, including 1) a detailed pelvic exam assessing the vulva, vagina, and pelvic-floor musculature, and 2) estrogenization of the tissue.
Partner concerns. For women who have a partner, addressing the concerns of that partner following gynecologic surgery can be useful to the couple: The partner might be concerned about inflicting pain or doing damage during sex after gynecologic surgery.
CASE Informative discussion
While ascertaining her sexual symptoms, your patient reveals to you that she has attempted sexual intercourse on 3 occasions; each time, penetration was too painful to continue. She tells you she did not have this problem before surgery.
The patient says that she has tried water-based lubricants and is using vaginal estrogen 3 times per week, but “nothing helps.” She reports that she is arousable and has been able to achieve orgasm with clitoral stimulation, but would like to have vaginal intercourse. Her husband does have erectile dysfunction, which, she tells you, can make penetration difficult.
On physical examination, you detect mild atrophy. Vaginal length is 9 cm; no narrowing or scarring of the vaginal introitus or canal is seen. No mesh is visible or palpable. The paths of the midurethral sling arms are nontender. However, levator muscles are tender and tense bilaterally.
Given these findings on examination, what steps can you take to relieve your patient’s pain?
What can we offer these patients?
Treating sexual dysfunction after pelvic reconstructive surgery must, as emphasized earlier, be guided by a careful history and physical exam. Doing so is critical to determining the underlying cause. Whenever feasible, offer the least invasive treatment.
The IUGA/ICS terminology report describes several symptoms of postoperative sexual dysfunction12:
- de novo sexual dysfunction
- de novo dyspareunia
- shortened vagina
- tight vagina (introital or vaginal narrowing, or both)
- scarred vagina (including mesh-related problems)
- hispareunia (pain experienced by a male partner after intercourse).
Of course, any one or combination of these symptoms can be present in a given patient. Furthermore, de novo sexual dysfunction, de novo dyspareunia, and hispareunia can have various underlying causes—again, underscoring the importance of the history and exam in determining treatment.
Continue to: Nonsurgical treatment...
Nonsurgical treatment
Nonhormonal vaginal lubricants and moisturizers; vaginal estrogen therapy. Although, in older women, vaginal atrophy is often not a new diagnosis postsurgically, the condition might have been untreated preoperatively and might therefore come into play in sexual dysfunction postoperatively. If a patient reports vaginal dryness or pain upon penetration, assess for vaginal atrophy and, if present, treat accordingly.
Vaginal dilation and physical therapy. A shortened, tight, or scarred vagina might be amenable to therapy with vaginal dilators and physical therapy, but might ultimately require surgery.
Pelvic-floor myalgia or spasm can develop after surgery or, as with atrophy, might have existed preoperatively but was left untreated. Pelvic-floor myalgia should be suspected if the patient describes difficult penetration or a feeling of tightness, even though scarring or constriction of the vagina is not seen on examination. Physical therapy with a specialist in pelvic floor treatment is a first-line treatment for pelvic-floor myalgia,16 and is likely to be a helpful adjunct in many situations, including mesh-related sexual problems.17
Oral or vaginal medications to relax pelvic-floor muscle spasm are an option, although efficacy data are limited. If pain is of longstanding duration and is thought to have a neuropathic component, successful use of tricyclic antidepressants, neuroleptics, and serotonin–norepinephrine reuptake inhibitors has been reported.18
Surgery
Data are sparse regarding surgical treatment of female sexual dysfunction after pelvic reconstructive surgery. Again, it is clear, however, that the key is carefully assessing each patient and then individualizing treatment. Patients can have any type of dysfunction that a patient who hasn’t had surgery can—but is also at risk of conditions directly related to surgery.
In any patient who has had mesh placed as part of surgery, thorough examination is necessary to determine whether or not the implant is involved in sexual dysfunction. If the dysfunction is an apparent result of surgery performed by another surgeon, make every effort to review the operative report to determine which material was implanted and how it was placed.
Trigger-point injection can be attempted in a patient who has site-specific tenderness that is not clearly associated with tissue obstruction of the vagina or mesh erosion.12,19 Even in areas of apparent banding or scarring related to mesh, trigger-point injection can be attempted to alleviate pain. How often trigger-point injections should be performed is understudied.
If, on examination, tenderness that replicates the dyspareunia is elicited when palpating the levator or obturator internus muscle, pelvic-floor muscle trigger-point injection can be offered (although physical therapy is first-line treatment). Trigger-point injection also can be a useful adjunct in women who have another identified cause of pain but also have developed pelvic-floor muscle spasm.
Not addressing concomitant pelvic-floor myalgia could prevent successful treatment of pain. Inclusion of a pudendal block also might help to alleviate pain.
Continue to: Surgical resection...
Surgical resection. If a skin bridge is clearly observed at the introitus, or if the introitus has been overly narrowed by perineorrhaphy but the remainder of the vagina has adequate length and caliber, surgical resection of the skin bridge might relieve symptoms of difficult penetration. In the case of obstructive perineorrhaphy, an attempt at reversal can be made by incising the perineum vertically but then reapproximating the edges transversely—sometimes referred to as reverse perineorrhaphy.
If scar tissue found elsewhere in the vagina might obstruct penetration, this condition might also be amenable to resection. When scarring is annular, relaxing incisions can be made bilaterally to relieve tension on that tissue; alternatively, it might be necessary to perform a Z-plasty. Nearly always, severe scarring is accompanied by levator myalgia, and a combined approach of surgery and physical therapy is necessary.
Neovagina. It is possible to find vaginal stenosis or shortening, to a varying degree, after surgical prolapse repair, with or without mesh or graft. As discussed, vaginal dilation should be offered but, if this is ineffective, the patient might be a candidate for surgical creation of a neovagina. Numerous techniques have been described for patients with congenital vaginal agenesis, with a few reports of similar techniques used to treat iatrogenic vaginal stenosis or obliteration.
The general principle of all neovagina procedures is to create a space between bladder and rectum of adequate caliber and length for desired sexual function. Reported techniques include a thigh or buttock skin graft, use of bowel or peritoneum, and, recently, a buccal mucosa graft.20,21
Resection or excision of mesh. In patients who develop sexual dysfunction after mesh placement, the problem can be caused by exposure of the mesh in the vagina or erosion into another organ, but can also arise in the absence of exposure or erosion. Patients might have tenderness to palpation at points where the mesh is palpable through the mucosa but not exposed.
Again, complete investigation is necessary to look for mesh involvement in the vagina and, depending on the type of implant, other adjacent organs. Assessing partner symptoms, such as pain and scratches, also can be telling.
If there is palpable tenderness on vaginal examination of the mesh, resection of the vaginal portion might be an option.17 Complete excision of mesh implants can be morbid, however, and might not provide a better outcome than partial excision. The risk of morbidity from complete mesh excision must be weighed against the likelihood that partial excision will not resolve pain and that the patient will require further excision subsequently.17,22 Excising fragmented mesh can be difficult; making every attempt to understand the contribution of mesh to sexual dysfunction is therefore critical to determining how, and how much of, the mesh comes out at the first attempt.
Last, for any woman who opts for surgical intervention to treat pain, you should engage in a discussion to emphasize the multidimensional nature of sexual function and the fact that any surgical intervention might not completely resolve her dysfunction.
Continue to: CASE Discussing options...
CASE Discussing options, choosing an intervention
You discuss the examination findings (no shortening or narrowing of the vagina) with the patient. She is relieved but puzzled as to why she cannot have intercourse. You discuss the tension and t
At 3-month follow-up, she reports great improvement. She is able to have intercourse, although she says she still has discomfort sometimes. She continues to work with the pelvic floor physical therapist and feels optimistic. You plan to see her in 6 months but counsel her to call if symptoms are not improving or are worsening.
It is difficult to counsel patients about sexual function after pelvic reconstructive surgery because data that could guide identification of problems (and how to treat them) are incomplete. Assessingsexual function preoperatively and having an open conversation about risks and benefits of surgery, with specific mention of its impact on sexual health, are critical (see “Key touchpoints in managing sexual dysfunction after pelvic reconstructive surgery”).
It is also crucial to assess sexual function postoperatively as a matter of routine. Validated questionnaires can be a useful adjunct to a thorough history and physical exam, and can help guide your discussions.
Treatment of postop sexual dysfunction must, first, account for the complex nature of sexual function and, second, be individualized, starting with the least invasive options, when feasible.
- Rogers RG. Sexual function in women with pelvic floor disorders. Can Urol Assoc J. 2013;7:S199-S201.
- Jha S, Gray T. A systematic review and meta-analysis of the impact of native tissue repair for pelvic organ prolapse on sexual function. Int Urogynecol J. 2015;26:321-327.
- Thompson JC, Rogers RG. Surgical management for pelvic organ prolapse and its impact on sexual function. Sex Med Rev. 2016;4:213-220.
- Sung VW, Rardin CR, Raker CA, et al. Porcine subintestinal submucosal graft augmentation for rectocele repair: a randomized controlled trial. Obstet Gynecol. 2012;119:125-133.
- Paraiso MF, Barber MD, Muir TW, et al. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
- Dietz V, Maher C. Pelvic organ prolapse and sexual function. Int Urogynecol J. 2013;24:1853-1857.
- Kuhn A, Brunnmayr G, Stadlmayr W, et al. Male and female sexual function after surgical repair of female organ prolapse. J Sex Med. 2009;6:1324-1334.
- Gray T, Li W, Campbell P, et al. Evaluation of coital incontinence by electronic questionnaire: prevalence, associations and outcomes in women attending a urogynaecology clinic. Int Urogynecol J. 2018;29:969-978.
- Jha S, Ammenbal M, Metwally M. Impact of incontinence surgery on sexual function: a systematic review and meta-analysis. J Sex Med. 2012;9:34-43.
- Schimpf MO, Rahn DD, Wheeler TL, et al; Society of Gynecologic Surgeons Systematic Review Group. Sling surgery for stress urinary incontinence in women: a systematic review and metaanalysis. Am J Obstet Gynecol. 2014;211:71.e1-e71.e27.
- Dunivan GC, Sussman AL, Jelovsek JE, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Gaining the patient perspective on pelvic floor disorders’ surgical adverse events. Am J Obstet Gynecol. 2019;220:185.e1-e185.e10.
- Rogers RG, Pauls RN, Thakar R, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for the assessment of sexual health of women with pelvic floor dysfunction. Int Urogynecol J. 2018;29:647-666.
- Plouffe L Jr. Screening for sexual problems through a simple questionnaire. Am J Obstet Gynecol. 1985;151:166-169.
- Hatzichristou D, Rosen RC, Derogatis LR, et al. Recommendations for the clinical evaluation of men and women with sexual dysfunction. J Sex Med. 2010;7:337-348.
- McCabe MP, Sharlip ID, Atalla E, et al. Definition of sexual dysfunctions in women and men: a consensus statement from the Fourth International Consultation of Sexual Medicine 2015. J Sex Med. 2015;13:135-143.
- Berghmans B. Physiotherapy for pelvic pain and female sexual dysfunction: an untapped resource. Int Urogynecol J. 2018;29:631-638.
- Cundiff GW, Quinlan DJ, van Rensburg JA, et al. Foundation for an evidence-informed algorithm for treating pelvic floor mesh complications: a review. BJOG. 2018;125:1026-1037.
- Steege JF, Siedhoff MT. Chronic pelvic pain. Obstet Gynecol. 2014;124:616-629.
- Wehbe SA, Whitmore K, Kellogg-Spadt S. Urogenital complaints and female sexual dysfunction (part 1). J Sex Med. 2010;7:1704-1713.
- Grimsby GM, Bradshaw K, Baker LA. Autologous buccal mucosa graft augmentation for foreshortened vagina. Obstet Gynecol. 2014;123:947-950.
- Morley GW, DeLancey JO. Full-thickness skin graft vaginoplasty for treatment of the stenotic or foreshortened vagina. Obstet Gynecol. 1991;77:485-489.
- Pickett SD, Barenberg B, Quiroz LH, et al. The significant morbidity of removing pelvic mesh from multiple vaginal compartments. Obstet Gynecol. 2015;125:1418-1422.
- Rogers RG. Sexual function in women with pelvic floor disorders. Can Urol Assoc J. 2013;7:S199-S201.
- Jha S, Gray T. A systematic review and meta-analysis of the impact of native tissue repair for pelvic organ prolapse on sexual function. Int Urogynecol J. 2015;26:321-327.
- Thompson JC, Rogers RG. Surgical management for pelvic organ prolapse and its impact on sexual function. Sex Med Rev. 2016;4:213-220.
- Sung VW, Rardin CR, Raker CA, et al. Porcine subintestinal submucosal graft augmentation for rectocele repair: a randomized controlled trial. Obstet Gynecol. 2012;119:125-133.
- Paraiso MF, Barber MD, Muir TW, et al. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
- Dietz V, Maher C. Pelvic organ prolapse and sexual function. Int Urogynecol J. 2013;24:1853-1857.
- Kuhn A, Brunnmayr G, Stadlmayr W, et al. Male and female sexual function after surgical repair of female organ prolapse. J Sex Med. 2009;6:1324-1334.
- Gray T, Li W, Campbell P, et al. Evaluation of coital incontinence by electronic questionnaire: prevalence, associations and outcomes in women attending a urogynaecology clinic. Int Urogynecol J. 2018;29:969-978.
- Jha S, Ammenbal M, Metwally M. Impact of incontinence surgery on sexual function: a systematic review and meta-analysis. J Sex Med. 2012;9:34-43.
- Schimpf MO, Rahn DD, Wheeler TL, et al; Society of Gynecologic Surgeons Systematic Review Group. Sling surgery for stress urinary incontinence in women: a systematic review and metaanalysis. Am J Obstet Gynecol. 2014;211:71.e1-e71.e27.
- Dunivan GC, Sussman AL, Jelovsek JE, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Gaining the patient perspective on pelvic floor disorders’ surgical adverse events. Am J Obstet Gynecol. 2019;220:185.e1-e185.e10.
- Rogers RG, Pauls RN, Thakar R, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for the assessment of sexual health of women with pelvic floor dysfunction. Int Urogynecol J. 2018;29:647-666.
- Plouffe L Jr. Screening for sexual problems through a simple questionnaire. Am J Obstet Gynecol. 1985;151:166-169.
- Hatzichristou D, Rosen RC, Derogatis LR, et al. Recommendations for the clinical evaluation of men and women with sexual dysfunction. J Sex Med. 2010;7:337-348.
- McCabe MP, Sharlip ID, Atalla E, et al. Definition of sexual dysfunctions in women and men: a consensus statement from the Fourth International Consultation of Sexual Medicine 2015. J Sex Med. 2015;13:135-143.
- Berghmans B. Physiotherapy for pelvic pain and female sexual dysfunction: an untapped resource. Int Urogynecol J. 2018;29:631-638.
- Cundiff GW, Quinlan DJ, van Rensburg JA, et al. Foundation for an evidence-informed algorithm for treating pelvic floor mesh complications: a review. BJOG. 2018;125:1026-1037.
- Steege JF, Siedhoff MT. Chronic pelvic pain. Obstet Gynecol. 2014;124:616-629.
- Wehbe SA, Whitmore K, Kellogg-Spadt S. Urogenital complaints and female sexual dysfunction (part 1). J Sex Med. 2010;7:1704-1713.
- Grimsby GM, Bradshaw K, Baker LA. Autologous buccal mucosa graft augmentation for foreshortened vagina. Obstet Gynecol. 2014;123:947-950.
- Morley GW, DeLancey JO. Full-thickness skin graft vaginoplasty for treatment of the stenotic or foreshortened vagina. Obstet Gynecol. 1991;77:485-489.
- Pickett SD, Barenberg B, Quiroz LH, et al. The significant morbidity of removing pelvic mesh from multiple vaginal compartments. Obstet Gynecol. 2015;125:1418-1422.
2019 Update on cervical disease
Cervical cancer rates remain low in the United States, with the incidence having plateaued for decades. And yet, in 2019, more than 13,000 US women will be diagnosed with cervical cancer.1 Globally, in 2018 almost 600,000 women were diagnosed with cervical cancer2; it is the fourth most frequent cancer in women. This is despite the fact that we have adequate primary and secondary prevention tools available to minimize—and almost eliminate—cervical cancer. We must continue to raise the bar for preventing, screening for, and managing this disease.
Human papillomavirus (HPV) vaccines provide a highly effective primary prevention strategy, but we need to improve our ability to identify and diagnose dysplastic lesions prior to the development of cervical cancer. Highly sensitive HPV testing and cytology is a powerful secondary prevention approach that enables us to assess a woman’s risk of having precancerous cells both now and in the near future. These modalities have been very successful in decreasing the incidence of cervical cancer in the United States and other areas with organized screening programs. In low- and middle-income countries, however, access to, availability of, and performance with these modalities is not optimal. Innovative strategies and new technologies are being evaluated to overcome these limitations.
Advances in radiation and surgical technology have enabled us to vastly improve cervical cancer treatment. Women with early-stage cervical cancer are candidates for surgical management, which frequently includes a radical hysterectomy and lymph node dissection. While these surgeries traditionally have been performed via an exploratory laparotomy, minimally invasive techniques (laparoscopic and robot-assisted surgical techniques) have decreased the morbidity with these surgeries. Notable new studies have shed light on the comparative effectiveness of minimally invasive technologies and have shown us that new is not always better.
The US Preventive Services Task Force (USPSTF) recently released its updated cervical cancer screening guidelines. The suggested approach to screening differs from previous recommendations. HPV testing as a primary test (that is, HPV testing alone or followed by cytology) takes the spotlight now, according to the analysis by the Task Force.
In this Update, we highlight important studies published in the past year that address these issues.
Continue to: New tech's potential to identify high-grade...
New tech's potential to identify high-grade cervical dysplasia may be a boon to low-resource settings
Hu L, Bell D, Antani S, et al. An observational study of deep learning and automated evaluation of cervical images for cancer screening. J Natl Cancer Inst. 2019;doi:10.1093/jnci/djy225.
When cervical screening tests like cytology and HPV testing show abnormal results, colposcopy often is recommended. The goal of colposcopy is to identify the areas that might harbor a high-grade precancerous lesion or worse. The gold standard in this case, however, is histology, not colposcopic impression, as many studies have shown that colposcopy without biopsies is limited and that performance is improved with more biopsies.3,4
Visual inspection with acetic acid (VIA) is an approach used often in low-resource settings where visual impression is the gold standard. However, as with colposcopy, a visual evaluation without histology does not perform well, and often women are overtreated. Many attempts have been made with new technologies to overcome the limitations of time, cost, and workforce required for cytology and histology services. New disruptive technologies may be able to surmount human limitations and improve on not only VIA but also the need for histology.
Novel technology uses images to develop algorithm with predictive ability
In a recent observational study, Hu and colleagues used images that were collected during a large population study in Guanacaste, Costa Rica.5 More than 9,000 women were followed for up to 7 years, and cervical photographs (cervigrams) were obtained. Well-annotated histopathology results were obtained for women with abnormal screening, and 279 women had a high-grade dysplastic lesion or cancer.
Cervigrams from women with high-grade lesions and matched controls were collected, and a deep learning-based algorithm using artificial intelligence technology was developed using 70% of the images. The remaining 30% of images were used as a validation set to test the algorithm's ability to "predict" high-grade dysplasia without knowing the final result.
Findings. Termed automated visual evaluation (AVE), this new technology demonstrated a very accurate ability to identify high-grade dysplasia or worse, with an area under the curve (AUC) of 0.91 from merely a cervicogram (FIGURE). This outperformed conventional Pap smears (AUC, 0.71), liquid-based cytology (AUC, 0.79) and, surprisingly, highly sensitive HPV testing (AUC, 0.82) in women in the prime of their screening ages (>25 years of age).
Colposcopy remains the gold standard for evaluating abnormal cervical cancer screening tests in the United States. But can we do better for our patients using new technologies like AVE? If validated in large-scale trials, AVE has the potential to revolutionize cervical cancer screening in low-resource settings where follow-up and adequate histology services are limited or nonexistent. Future large studies are necessary to evaluate the role of AVE alone versus in combination with other diagnostic testing (such as HPV testing) to detect cervical lesions globally.
Continue to: Data offer persuasive evidence...
Data offer persuasive evidence to abandon minimally invasive surgery in management of early-stage cervical cancer
Melamed A, Margul DJ, Chen L, et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med. 2018;379:1905-1914.
Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379:1895-1904.
Over the past decade, gynecologic cancer surgery has shifted from what routinely were open procedures to the adoption of minimally invasive techniques. Recently, a large, well-designed prospective study and a large retrospective study both demonstrated worse outcomes with minimally invasive radical hysterectomy (MIRH) as compared with traditional open radical abdominal hysterectomy (RAH). These 2 landmark studies, initially presented at the Society of Gynecologic Oncology's 2018 annual meeting and later published in the New England Journal of Medicine, have really affected the gynecologic oncology community.
Shorter overall survival in women who had MIRH
Melamed and colleagues conducted a large, retrospective US-based study to evaluate all-cause mortality in women with cervical cancer who underwent MIRH compared with those who had RAH.6 The authors also sought to evaluate national trends in 4-year relative survival rates after minimally invasive surgery was adopted.
The study included 2,461 women who met the inclusion criteria; 49.8% (1,225) underwent MIRH procedures and, of those, 79.8% (978) had robot-assisted laparoscopy. Most women had stage IB1 tumors (88%), and most carcinomas were squamous cell (61%); 40.6% of tumors were less than 2 cm in size. There were no differences between the 2 groups with respect to rates of positive parametria, surgical margins, and lymph node involvement. Administration of adjuvant therapy, in those who qualified, was also similar between groups.
Results. At a median follow-up of 45 months, 94 deaths occurred in the minimally invasive group and 70 in the open surgery group. The risk of death at 4 years was 9.1% in the minimally invasive group versus 5.3% in the open surgery group, with a 65% higher risk of death from any cause, which was highly statistically significant.
Prospective trial showed MIRH was associated with lower survival rates
From 2008 to 2017, Ramirez and colleagues conducted a phase 3, multicenter, randomized controlled trial to prospectively establish the noninferiority of MIRH compared with RAH.7 The study included 631 women from 33 centers. The prespecified expected disease-free survival rate was 90% at 4.5 years.
To be included as a site, centers were required to submit details from 10 minimally invasive cases as well as 2 unedited videos for review by the trial management committee. In contrast to Melamed and colleagues' retrospective study, of the 319 procedures that were classified as minimally invasive, only 15.6% were robotically assisted. Similarly, most women had stage IB1 tumors (91.9%), and most were squamous cell carcinomas (67%). There were also no differences in the postoperative pathology findings or the need for adjuvant therapy administered between groups. The median follow-up was 2.5 years.
Results. At that time there were 27 recurrences in the MIRH group and 7 in the RAH group; there were also 19 deaths after MIRH and 3 after RAH. Disease-free survival at 4.5 years was 86% with MIRH versus 96.5% with RAH. Reported 3-year disease-free survival and overall survival were also significantily lower in the minimally invasive subgroup (91.2% vs 97.1%, 93.8% vs 99.0%, respectively).
Study limitations. Criticisms of this trial are that noninferiority could not be declared; in addition, the investigators were unable to complete enrollment secondary to early enrollment termination after the data and safety monitoring board raised survival concerns.
Many argue that subgroup analyses suggest a lower risk of poor outcomes in patients with smaller tumors (<2 cm); however, it is critical to note that this study was not powered to detect these differences.
The evidence is compelling and demonstrates potentially worse disease-related outcomes using MIRH when compared to traditional RAH with respect to cervical cancer recurrence, rates of death, and disease-free and overall survival. Several hypotheses have been proposed, and future research is needed to elucidate the differences in variables responsible for the outcomes demonstrated in these studies. Although there has been no ban on robot-assisted surgical devices or traditional minimally invasive techniques, the National Comprehensive Cancer Network has updated its recommendations to include careful counseling of patients who require a surgical approach for the management of early-stage cervical cancer.
Continue to: USPSTF updated guidance on cervical cancer screening...
USPSTF updated guidance on cervical cancer screening
Melnikow J, Henderson JT, Burda BU, et al. Screening for cervical cancer with high-risk human papillomavirus testing: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320:687-705.
US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
Past guidelines for cervical cancer screening have included testing for high-risk HPV (hrHPV) as a cotest with cytology or for triage of atypical squamous cells of undetermined significance (ASCUS) in women aged 30 to 65 years.8 The American Society for Colposcopy and Cervical Pathology and the Society of Gynecologic Oncology, with other stakeholder organizations, issued interim guidance for primary HPV testing--that is, HPV test first and, in the case of non-16/18 hrHPV types, cytology as a triage. The most recent evidence report and systematic review by Melnikow and colleagues for the USPSTF offers an in-depth analysis of risks, benefits, harms, and value of cotesting and other management strategies.9
Focus on screening effectiveness
Large trials of cotesting were conducted in women aged 25 to 65.10-13 These studies all consistently showed that primary hrHPV screening led to a statistically significant increased detection of cervical intraepithelial neoplasia (CIN) 3+ in the initial round of screening, with a relative risk of detecting CIN 3+ ranging from 1.61 to 7.46 compared with cytology alone.
Four additional studies compared cotesting with conventional cytology for the detection of CIN 3+. None of these trials demonstrated a significantly higher detection rate of CIN 3+ with cotesting compared with conventional cytology testing alone. Notably, the studies reviewed were performed in European countries that had organized screening programs in place and a nationalized health care system. Thus, these data may not be as applicable to women in the United States, particularly to women who have limited health care access.
Risks of screening
In the same studies reviewed for screening effectiveness, the investigators found that overall, screening with hrHPV primary or cotesting was associated with more false-positive results and higher colposcopy rates. Women screened with hrHPV alone had a 7.9% referral rate to colposcopy, while those screened with cytology had a 2.8% referral rate to colposcopy. Similarly, the rate of biopsy was higher in the hrHPV-only group (3.2% vs 1.3%).
Overall, while cotesting might have some improvement in performance compared with hrHPV as a single modality, there might be risks of overreferral to colposcopy and overtreatment with additional cytology over hrHPV testing alone.
This evidence review also included an analysis of more potential harms. Very limited evidence suggests that positive hrHPV test results may be associated with greater psychological harm, including decreased sexual satisfaction, increased anxiety and distress, and worse feelings about sexual partners, than abnormal cytology results. These were assessed, however, 1 to 2 weeks after the test results were provided to the patients, and long-term assessment was not done.
New recommendations from the USPSTF
Based on these data, the USPSTF issued new recommendations regarding screening (TABLE).14 For women aged 21 to 29, cytology alone should be used for screening every 3 years. Women aged 30 to 65 can be screened with cytology alone every 3 years, with hrHPV testing alone every 5 years, or with cotesting every 5 years.
Primary screening with hrHPV is more effective in diagnosing a CIN 3+ than cytology alone. Cotesting with cytology and hrHPV testing appears to have limited performance improvement, with potential harm, compared with hrHPV testing alone in diagnosing CIN 3+. The Task Force recommendation is hrHPV testing alone or cotesting every 5 years.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
- World Health Organization website. Cervical cancer. https://www.who.int/cancer/prevention/diagnosis-screening/cervical-cancer/en/. Accessed April 17, 2019.
- Wentzensen N, Walker JL, Gold MA, et al. Multiple biopsies and detection of cervical cancer precursors at colposcopy. J Clin Oncol. 2015;33:83-89.
- Gage JC, Hanson VW, Abbey K, et al. Number of cervical biopsies and sensitivity of colposcopy. Obstet Gynecol. 2006;108:264-272.
- Hu L, Bell D, Antani S, et al. An observational study of deep learning and automated evaluation of cervical images for cancer screening. J Natl Cancer Inst. 2019;doi:10.1093/jnci/djy225.
- Melamed A, Margul DJ, Chen L, et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med. 2018;379:1905-1914.
- Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379:1895-1904.
- Saslow D, Solomon D, Lawson HW, et al; ACS-ASCCP-ASCP Cervical Cancer Guideline Committee. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62:147-172.
- Melnikow J, Henderson JT, Burda BU, et al. Screening for cervical cancer with high-risk human papillomavirus testing: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320:687-705.
- Canfell K, Caruana M, Gebski V, et al. Cervical screening with primary HPV testing or cytology in a population of women in which those aged 33 years or younger had previously been offered HPV vaccination: results of the Compass pilot randomised trial. PLoS Med. 2017;14:e1002388. doi:10.1371/journal.pmed.1002388.
- Leinonen MK, Nieminen P, Lonnberg S, et al. Detection rates of precancerous and cancerous cervical lesions within one screening round of primary human papillomavirus DNA testing: prospective randomised trial in Finland. BMJ. 2012;345:e7789.
- Ogilvie GS, van Niekerk D, Krajden M, et al. Effect of screening with primary cervical HPV testing vs cytology testing on high-grade cervical intraepithelial neoplasia at 48 months: the HPV FOCAL randomized clinical trial. JAMA. 2018;320:43-52.
- Ronco G, Fioprgi-Rossi P, Carozzi F, et al; New Technologies for Cervical Cancer screening (NTCC) Working Group. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomised controlled trial. Lancet Oncol. 2010;11:249-257.
- US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
Cervical cancer rates remain low in the United States, with the incidence having plateaued for decades. And yet, in 2019, more than 13,000 US women will be diagnosed with cervical cancer.1 Globally, in 2018 almost 600,000 women were diagnosed with cervical cancer2; it is the fourth most frequent cancer in women. This is despite the fact that we have adequate primary and secondary prevention tools available to minimize—and almost eliminate—cervical cancer. We must continue to raise the bar for preventing, screening for, and managing this disease.
Human papillomavirus (HPV) vaccines provide a highly effective primary prevention strategy, but we need to improve our ability to identify and diagnose dysplastic lesions prior to the development of cervical cancer. Highly sensitive HPV testing and cytology is a powerful secondary prevention approach that enables us to assess a woman’s risk of having precancerous cells both now and in the near future. These modalities have been very successful in decreasing the incidence of cervical cancer in the United States and other areas with organized screening programs. In low- and middle-income countries, however, access to, availability of, and performance with these modalities is not optimal. Innovative strategies and new technologies are being evaluated to overcome these limitations.
Advances in radiation and surgical technology have enabled us to vastly improve cervical cancer treatment. Women with early-stage cervical cancer are candidates for surgical management, which frequently includes a radical hysterectomy and lymph node dissection. While these surgeries traditionally have been performed via an exploratory laparotomy, minimally invasive techniques (laparoscopic and robot-assisted surgical techniques) have decreased the morbidity with these surgeries. Notable new studies have shed light on the comparative effectiveness of minimally invasive technologies and have shown us that new is not always better.
The US Preventive Services Task Force (USPSTF) recently released its updated cervical cancer screening guidelines. The suggested approach to screening differs from previous recommendations. HPV testing as a primary test (that is, HPV testing alone or followed by cytology) takes the spotlight now, according to the analysis by the Task Force.
In this Update, we highlight important studies published in the past year that address these issues.
Continue to: New tech's potential to identify high-grade...
New tech's potential to identify high-grade cervical dysplasia may be a boon to low-resource settings
Hu L, Bell D, Antani S, et al. An observational study of deep learning and automated evaluation of cervical images for cancer screening. J Natl Cancer Inst. 2019;doi:10.1093/jnci/djy225.
When cervical screening tests like cytology and HPV testing show abnormal results, colposcopy often is recommended. The goal of colposcopy is to identify the areas that might harbor a high-grade precancerous lesion or worse. The gold standard in this case, however, is histology, not colposcopic impression, as many studies have shown that colposcopy without biopsies is limited and that performance is improved with more biopsies.3,4
Visual inspection with acetic acid (VIA) is an approach used often in low-resource settings where visual impression is the gold standard. However, as with colposcopy, a visual evaluation without histology does not perform well, and often women are overtreated. Many attempts have been made with new technologies to overcome the limitations of time, cost, and workforce required for cytology and histology services. New disruptive technologies may be able to surmount human limitations and improve on not only VIA but also the need for histology.
Novel technology uses images to develop algorithm with predictive ability
In a recent observational study, Hu and colleagues used images that were collected during a large population study in Guanacaste, Costa Rica.5 More than 9,000 women were followed for up to 7 years, and cervical photographs (cervigrams) were obtained. Well-annotated histopathology results were obtained for women with abnormal screening, and 279 women had a high-grade dysplastic lesion or cancer.
Cervigrams from women with high-grade lesions and matched controls were collected, and a deep learning-based algorithm using artificial intelligence technology was developed using 70% of the images. The remaining 30% of images were used as a validation set to test the algorithm's ability to "predict" high-grade dysplasia without knowing the final result.
Findings. Termed automated visual evaluation (AVE), this new technology demonstrated a very accurate ability to identify high-grade dysplasia or worse, with an area under the curve (AUC) of 0.91 from merely a cervicogram (FIGURE). This outperformed conventional Pap smears (AUC, 0.71), liquid-based cytology (AUC, 0.79) and, surprisingly, highly sensitive HPV testing (AUC, 0.82) in women in the prime of their screening ages (>25 years of age).

Colposcopy remains the gold standard for evaluating abnormal cervical cancer screening tests in the United States. But can we do better for our patients using new technologies like AVE? If validated in large-scale trials, AVE has the potential to revolutionize cervical cancer screening in low-resource settings where follow-up and adequate histology services are limited or nonexistent. Future large studies are necessary to evaluate the role of AVE alone versus in combination with other diagnostic testing (such as HPV testing) to detect cervical lesions globally.
Continue to: Data offer persuasive evidence...
Data offer persuasive evidence to abandon minimally invasive surgery in management of early-stage cervical cancer
Melamed A, Margul DJ, Chen L, et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med. 2018;379:1905-1914.
Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379:1895-1904.
Over the past decade, gynecologic cancer surgery has shifted from what routinely were open procedures to the adoption of minimally invasive techniques. Recently, a large, well-designed prospective study and a large retrospective study both demonstrated worse outcomes with minimally invasive radical hysterectomy (MIRH) as compared with traditional open radical abdominal hysterectomy (RAH). These 2 landmark studies, initially presented at the Society of Gynecologic Oncology's 2018 annual meeting and later published in the New England Journal of Medicine, have really affected the gynecologic oncology community.
Shorter overall survival in women who had MIRH
Melamed and colleagues conducted a large, retrospective US-based study to evaluate all-cause mortality in women with cervical cancer who underwent MIRH compared with those who had RAH.6 The authors also sought to evaluate national trends in 4-year relative survival rates after minimally invasive surgery was adopted.
The study included 2,461 women who met the inclusion criteria; 49.8% (1,225) underwent MIRH procedures and, of those, 79.8% (978) had robot-assisted laparoscopy. Most women had stage IB1 tumors (88%), and most carcinomas were squamous cell (61%); 40.6% of tumors were less than 2 cm in size. There were no differences between the 2 groups with respect to rates of positive parametria, surgical margins, and lymph node involvement. Administration of adjuvant therapy, in those who qualified, was also similar between groups.
Results. At a median follow-up of 45 months, 94 deaths occurred in the minimally invasive group and 70 in the open surgery group. The risk of death at 4 years was 9.1% in the minimally invasive group versus 5.3% in the open surgery group, with a 65% higher risk of death from any cause, which was highly statistically significant.
Prospective trial showed MIRH was associated with lower survival rates
From 2008 to 2017, Ramirez and colleagues conducted a phase 3, multicenter, randomized controlled trial to prospectively establish the noninferiority of MIRH compared with RAH.7 The study included 631 women from 33 centers. The prespecified expected disease-free survival rate was 90% at 4.5 years.
To be included as a site, centers were required to submit details from 10 minimally invasive cases as well as 2 unedited videos for review by the trial management committee. In contrast to Melamed and colleagues' retrospective study, of the 319 procedures that were classified as minimally invasive, only 15.6% were robotically assisted. Similarly, most women had stage IB1 tumors (91.9%), and most were squamous cell carcinomas (67%). There were also no differences in the postoperative pathology findings or the need for adjuvant therapy administered between groups. The median follow-up was 2.5 years.
Results. At that time there were 27 recurrences in the MIRH group and 7 in the RAH group; there were also 19 deaths after MIRH and 3 after RAH. Disease-free survival at 4.5 years was 86% with MIRH versus 96.5% with RAH. Reported 3-year disease-free survival and overall survival were also significantily lower in the minimally invasive subgroup (91.2% vs 97.1%, 93.8% vs 99.0%, respectively).
Study limitations. Criticisms of this trial are that noninferiority could not be declared; in addition, the investigators were unable to complete enrollment secondary to early enrollment termination after the data and safety monitoring board raised survival concerns.
Many argue that subgroup analyses suggest a lower risk of poor outcomes in patients with smaller tumors (<2 cm); however, it is critical to note that this study was not powered to detect these differences.
The evidence is compelling and demonstrates potentially worse disease-related outcomes using MIRH when compared to traditional RAH with respect to cervical cancer recurrence, rates of death, and disease-free and overall survival. Several hypotheses have been proposed, and future research is needed to elucidate the differences in variables responsible for the outcomes demonstrated in these studies. Although there has been no ban on robot-assisted surgical devices or traditional minimally invasive techniques, the National Comprehensive Cancer Network has updated its recommendations to include careful counseling of patients who require a surgical approach for the management of early-stage cervical cancer.
Continue to: USPSTF updated guidance on cervical cancer screening...
USPSTF updated guidance on cervical cancer screening
Melnikow J, Henderson JT, Burda BU, et al. Screening for cervical cancer with high-risk human papillomavirus testing: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320:687-705.
US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
Past guidelines for cervical cancer screening have included testing for high-risk HPV (hrHPV) as a cotest with cytology or for triage of atypical squamous cells of undetermined significance (ASCUS) in women aged 30 to 65 years.8 The American Society for Colposcopy and Cervical Pathology and the Society of Gynecologic Oncology, with other stakeholder organizations, issued interim guidance for primary HPV testing--that is, HPV test first and, in the case of non-16/18 hrHPV types, cytology as a triage. The most recent evidence report and systematic review by Melnikow and colleagues for the USPSTF offers an in-depth analysis of risks, benefits, harms, and value of cotesting and other management strategies.9
Focus on screening effectiveness
Large trials of cotesting were conducted in women aged 25 to 65.10-13 These studies all consistently showed that primary hrHPV screening led to a statistically significant increased detection of cervical intraepithelial neoplasia (CIN) 3+ in the initial round of screening, with a relative risk of detecting CIN 3+ ranging from 1.61 to 7.46 compared with cytology alone.
Four additional studies compared cotesting with conventional cytology for the detection of CIN 3+. None of these trials demonstrated a significantly higher detection rate of CIN 3+ with cotesting compared with conventional cytology testing alone. Notably, the studies reviewed were performed in European countries that had organized screening programs in place and a nationalized health care system. Thus, these data may not be as applicable to women in the United States, particularly to women who have limited health care access.
Risks of screening
In the same studies reviewed for screening effectiveness, the investigators found that overall, screening with hrHPV primary or cotesting was associated with more false-positive results and higher colposcopy rates. Women screened with hrHPV alone had a 7.9% referral rate to colposcopy, while those screened with cytology had a 2.8% referral rate to colposcopy. Similarly, the rate of biopsy was higher in the hrHPV-only group (3.2% vs 1.3%).
Overall, while cotesting might have some improvement in performance compared with hrHPV as a single modality, there might be risks of overreferral to colposcopy and overtreatment with additional cytology over hrHPV testing alone.
This evidence review also included an analysis of more potential harms. Very limited evidence suggests that positive hrHPV test results may be associated with greater psychological harm, including decreased sexual satisfaction, increased anxiety and distress, and worse feelings about sexual partners, than abnormal cytology results. These were assessed, however, 1 to 2 weeks after the test results were provided to the patients, and long-term assessment was not done.
New recommendations from the USPSTF
Based on these data, the USPSTF issued new recommendations regarding screening (TABLE).14 For women aged 21 to 29, cytology alone should be used for screening every 3 years. Women aged 30 to 65 can be screened with cytology alone every 3 years, with hrHPV testing alone every 5 years, or with cotesting every 5 years.

Primary screening with hrHPV is more effective in diagnosing a CIN 3+ than cytology alone. Cotesting with cytology and hrHPV testing appears to have limited performance improvement, with potential harm, compared with hrHPV testing alone in diagnosing CIN 3+. The Task Force recommendation is hrHPV testing alone or cotesting every 5 years.
Cervical cancer rates remain low in the United States, with the incidence having plateaued for decades. And yet, in 2019, more than 13,000 US women will be diagnosed with cervical cancer.1 Globally, in 2018 almost 600,000 women were diagnosed with cervical cancer2; it is the fourth most frequent cancer in women. This is despite the fact that we have adequate primary and secondary prevention tools available to minimize—and almost eliminate—cervical cancer. We must continue to raise the bar for preventing, screening for, and managing this disease.
Human papillomavirus (HPV) vaccines provide a highly effective primary prevention strategy, but we need to improve our ability to identify and diagnose dysplastic lesions prior to the development of cervical cancer. Highly sensitive HPV testing and cytology is a powerful secondary prevention approach that enables us to assess a woman’s risk of having precancerous cells both now and in the near future. These modalities have been very successful in decreasing the incidence of cervical cancer in the United States and other areas with organized screening programs. In low- and middle-income countries, however, access to, availability of, and performance with these modalities is not optimal. Innovative strategies and new technologies are being evaluated to overcome these limitations.
Advances in radiation and surgical technology have enabled us to vastly improve cervical cancer treatment. Women with early-stage cervical cancer are candidates for surgical management, which frequently includes a radical hysterectomy and lymph node dissection. While these surgeries traditionally have been performed via an exploratory laparotomy, minimally invasive techniques (laparoscopic and robot-assisted surgical techniques) have decreased the morbidity with these surgeries. Notable new studies have shed light on the comparative effectiveness of minimally invasive technologies and have shown us that new is not always better.
The US Preventive Services Task Force (USPSTF) recently released its updated cervical cancer screening guidelines. The suggested approach to screening differs from previous recommendations. HPV testing as a primary test (that is, HPV testing alone or followed by cytology) takes the spotlight now, according to the analysis by the Task Force.
In this Update, we highlight important studies published in the past year that address these issues.
Continue to: New tech's potential to identify high-grade...
New tech's potential to identify high-grade cervical dysplasia may be a boon to low-resource settings
Hu L, Bell D, Antani S, et al. An observational study of deep learning and automated evaluation of cervical images for cancer screening. J Natl Cancer Inst. 2019;doi:10.1093/jnci/djy225.
When cervical screening tests like cytology and HPV testing show abnormal results, colposcopy often is recommended. The goal of colposcopy is to identify the areas that might harbor a high-grade precancerous lesion or worse. The gold standard in this case, however, is histology, not colposcopic impression, as many studies have shown that colposcopy without biopsies is limited and that performance is improved with more biopsies.3,4
Visual inspection with acetic acid (VIA) is an approach used often in low-resource settings where visual impression is the gold standard. However, as with colposcopy, a visual evaluation without histology does not perform well, and often women are overtreated. Many attempts have been made with new technologies to overcome the limitations of time, cost, and workforce required for cytology and histology services. New disruptive technologies may be able to surmount human limitations and improve on not only VIA but also the need for histology.
Novel technology uses images to develop algorithm with predictive ability
In a recent observational study, Hu and colleagues used images that were collected during a large population study in Guanacaste, Costa Rica.5 More than 9,000 women were followed for up to 7 years, and cervical photographs (cervigrams) were obtained. Well-annotated histopathology results were obtained for women with abnormal screening, and 279 women had a high-grade dysplastic lesion or cancer.
Cervigrams from women with high-grade lesions and matched controls were collected, and a deep learning-based algorithm using artificial intelligence technology was developed using 70% of the images. The remaining 30% of images were used as a validation set to test the algorithm's ability to "predict" high-grade dysplasia without knowing the final result.
Findings. Termed automated visual evaluation (AVE), this new technology demonstrated a very accurate ability to identify high-grade dysplasia or worse, with an area under the curve (AUC) of 0.91 from merely a cervicogram (FIGURE). This outperformed conventional Pap smears (AUC, 0.71), liquid-based cytology (AUC, 0.79) and, surprisingly, highly sensitive HPV testing (AUC, 0.82) in women in the prime of their screening ages (>25 years of age).

Colposcopy remains the gold standard for evaluating abnormal cervical cancer screening tests in the United States. But can we do better for our patients using new technologies like AVE? If validated in large-scale trials, AVE has the potential to revolutionize cervical cancer screening in low-resource settings where follow-up and adequate histology services are limited or nonexistent. Future large studies are necessary to evaluate the role of AVE alone versus in combination with other diagnostic testing (such as HPV testing) to detect cervical lesions globally.
Continue to: Data offer persuasive evidence...
Data offer persuasive evidence to abandon minimally invasive surgery in management of early-stage cervical cancer
Melamed A, Margul DJ, Chen L, et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med. 2018;379:1905-1914.
Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379:1895-1904.
Over the past decade, gynecologic cancer surgery has shifted from what routinely were open procedures to the adoption of minimally invasive techniques. Recently, a large, well-designed prospective study and a large retrospective study both demonstrated worse outcomes with minimally invasive radical hysterectomy (MIRH) as compared with traditional open radical abdominal hysterectomy (RAH). These 2 landmark studies, initially presented at the Society of Gynecologic Oncology's 2018 annual meeting and later published in the New England Journal of Medicine, have really affected the gynecologic oncology community.
Shorter overall survival in women who had MIRH
Melamed and colleagues conducted a large, retrospective US-based study to evaluate all-cause mortality in women with cervical cancer who underwent MIRH compared with those who had RAH.6 The authors also sought to evaluate national trends in 4-year relative survival rates after minimally invasive surgery was adopted.
The study included 2,461 women who met the inclusion criteria; 49.8% (1,225) underwent MIRH procedures and, of those, 79.8% (978) had robot-assisted laparoscopy. Most women had stage IB1 tumors (88%), and most carcinomas were squamous cell (61%); 40.6% of tumors were less than 2 cm in size. There were no differences between the 2 groups with respect to rates of positive parametria, surgical margins, and lymph node involvement. Administration of adjuvant therapy, in those who qualified, was also similar between groups.
Results. At a median follow-up of 45 months, 94 deaths occurred in the minimally invasive group and 70 in the open surgery group. The risk of death at 4 years was 9.1% in the minimally invasive group versus 5.3% in the open surgery group, with a 65% higher risk of death from any cause, which was highly statistically significant.
Prospective trial showed MIRH was associated with lower survival rates
From 2008 to 2017, Ramirez and colleagues conducted a phase 3, multicenter, randomized controlled trial to prospectively establish the noninferiority of MIRH compared with RAH.7 The study included 631 women from 33 centers. The prespecified expected disease-free survival rate was 90% at 4.5 years.
To be included as a site, centers were required to submit details from 10 minimally invasive cases as well as 2 unedited videos for review by the trial management committee. In contrast to Melamed and colleagues' retrospective study, of the 319 procedures that were classified as minimally invasive, only 15.6% were robotically assisted. Similarly, most women had stage IB1 tumors (91.9%), and most were squamous cell carcinomas (67%). There were also no differences in the postoperative pathology findings or the need for adjuvant therapy administered between groups. The median follow-up was 2.5 years.
Results. At that time there were 27 recurrences in the MIRH group and 7 in the RAH group; there were also 19 deaths after MIRH and 3 after RAH. Disease-free survival at 4.5 years was 86% with MIRH versus 96.5% with RAH. Reported 3-year disease-free survival and overall survival were also significantily lower in the minimally invasive subgroup (91.2% vs 97.1%, 93.8% vs 99.0%, respectively).
Study limitations. Criticisms of this trial are that noninferiority could not be declared; in addition, the investigators were unable to complete enrollment secondary to early enrollment termination after the data and safety monitoring board raised survival concerns.
Many argue that subgroup analyses suggest a lower risk of poor outcomes in patients with smaller tumors (<2 cm); however, it is critical to note that this study was not powered to detect these differences.
The evidence is compelling and demonstrates potentially worse disease-related outcomes using MIRH when compared to traditional RAH with respect to cervical cancer recurrence, rates of death, and disease-free and overall survival. Several hypotheses have been proposed, and future research is needed to elucidate the differences in variables responsible for the outcomes demonstrated in these studies. Although there has been no ban on robot-assisted surgical devices or traditional minimally invasive techniques, the National Comprehensive Cancer Network has updated its recommendations to include careful counseling of patients who require a surgical approach for the management of early-stage cervical cancer.
Continue to: USPSTF updated guidance on cervical cancer screening...
USPSTF updated guidance on cervical cancer screening
Melnikow J, Henderson JT, Burda BU, et al. Screening for cervical cancer with high-risk human papillomavirus testing: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320:687-705.
US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
Past guidelines for cervical cancer screening have included testing for high-risk HPV (hrHPV) as a cotest with cytology or for triage of atypical squamous cells of undetermined significance (ASCUS) in women aged 30 to 65 years.8 The American Society for Colposcopy and Cervical Pathology and the Society of Gynecologic Oncology, with other stakeholder organizations, issued interim guidance for primary HPV testing--that is, HPV test first and, in the case of non-16/18 hrHPV types, cytology as a triage. The most recent evidence report and systematic review by Melnikow and colleagues for the USPSTF offers an in-depth analysis of risks, benefits, harms, and value of cotesting and other management strategies.9
Focus on screening effectiveness
Large trials of cotesting were conducted in women aged 25 to 65.10-13 These studies all consistently showed that primary hrHPV screening led to a statistically significant increased detection of cervical intraepithelial neoplasia (CIN) 3+ in the initial round of screening, with a relative risk of detecting CIN 3+ ranging from 1.61 to 7.46 compared with cytology alone.
Four additional studies compared cotesting with conventional cytology for the detection of CIN 3+. None of these trials demonstrated a significantly higher detection rate of CIN 3+ with cotesting compared with conventional cytology testing alone. Notably, the studies reviewed were performed in European countries that had organized screening programs in place and a nationalized health care system. Thus, these data may not be as applicable to women in the United States, particularly to women who have limited health care access.
Risks of screening
In the same studies reviewed for screening effectiveness, the investigators found that overall, screening with hrHPV primary or cotesting was associated with more false-positive results and higher colposcopy rates. Women screened with hrHPV alone had a 7.9% referral rate to colposcopy, while those screened with cytology had a 2.8% referral rate to colposcopy. Similarly, the rate of biopsy was higher in the hrHPV-only group (3.2% vs 1.3%).
Overall, while cotesting might have some improvement in performance compared with hrHPV as a single modality, there might be risks of overreferral to colposcopy and overtreatment with additional cytology over hrHPV testing alone.
This evidence review also included an analysis of more potential harms. Very limited evidence suggests that positive hrHPV test results may be associated with greater psychological harm, including decreased sexual satisfaction, increased anxiety and distress, and worse feelings about sexual partners, than abnormal cytology results. These were assessed, however, 1 to 2 weeks after the test results were provided to the patients, and long-term assessment was not done.
New recommendations from the USPSTF
Based on these data, the USPSTF issued new recommendations regarding screening (TABLE).14 For women aged 21 to 29, cytology alone should be used for screening every 3 years. Women aged 30 to 65 can be screened with cytology alone every 3 years, with hrHPV testing alone every 5 years, or with cotesting every 5 years.

Primary screening with hrHPV is more effective in diagnosing a CIN 3+ than cytology alone. Cotesting with cytology and hrHPV testing appears to have limited performance improvement, with potential harm, compared with hrHPV testing alone in diagnosing CIN 3+. The Task Force recommendation is hrHPV testing alone or cotesting every 5 years.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
- World Health Organization website. Cervical cancer. https://www.who.int/cancer/prevention/diagnosis-screening/cervical-cancer/en/. Accessed April 17, 2019.
- Wentzensen N, Walker JL, Gold MA, et al. Multiple biopsies and detection of cervical cancer precursors at colposcopy. J Clin Oncol. 2015;33:83-89.
- Gage JC, Hanson VW, Abbey K, et al. Number of cervical biopsies and sensitivity of colposcopy. Obstet Gynecol. 2006;108:264-272.
- Hu L, Bell D, Antani S, et al. An observational study of deep learning and automated evaluation of cervical images for cancer screening. J Natl Cancer Inst. 2019;doi:10.1093/jnci/djy225.
- Melamed A, Margul DJ, Chen L, et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med. 2018;379:1905-1914.
- Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379:1895-1904.
- Saslow D, Solomon D, Lawson HW, et al; ACS-ASCCP-ASCP Cervical Cancer Guideline Committee. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62:147-172.
- Melnikow J, Henderson JT, Burda BU, et al. Screening for cervical cancer with high-risk human papillomavirus testing: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320:687-705.
- Canfell K, Caruana M, Gebski V, et al. Cervical screening with primary HPV testing or cytology in a population of women in which those aged 33 years or younger had previously been offered HPV vaccination: results of the Compass pilot randomised trial. PLoS Med. 2017;14:e1002388. doi:10.1371/journal.pmed.1002388.
- Leinonen MK, Nieminen P, Lonnberg S, et al. Detection rates of precancerous and cancerous cervical lesions within one screening round of primary human papillomavirus DNA testing: prospective randomised trial in Finland. BMJ. 2012;345:e7789.
- Ogilvie GS, van Niekerk D, Krajden M, et al. Effect of screening with primary cervical HPV testing vs cytology testing on high-grade cervical intraepithelial neoplasia at 48 months: the HPV FOCAL randomized clinical trial. JAMA. 2018;320:43-52.
- Ronco G, Fioprgi-Rossi P, Carozzi F, et al; New Technologies for Cervical Cancer screening (NTCC) Working Group. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomised controlled trial. Lancet Oncol. 2010;11:249-257.
- US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
- World Health Organization website. Cervical cancer. https://www.who.int/cancer/prevention/diagnosis-screening/cervical-cancer/en/. Accessed April 17, 2019.
- Wentzensen N, Walker JL, Gold MA, et al. Multiple biopsies and detection of cervical cancer precursors at colposcopy. J Clin Oncol. 2015;33:83-89.
- Gage JC, Hanson VW, Abbey K, et al. Number of cervical biopsies and sensitivity of colposcopy. Obstet Gynecol. 2006;108:264-272.
- Hu L, Bell D, Antani S, et al. An observational study of deep learning and automated evaluation of cervical images for cancer screening. J Natl Cancer Inst. 2019;doi:10.1093/jnci/djy225.
- Melamed A, Margul DJ, Chen L, et al. Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med. 2018;379:1905-1914.
- Ramirez PT, Frumovitz M, Pareja R, et al. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med. 2018;379:1895-1904.
- Saslow D, Solomon D, Lawson HW, et al; ACS-ASCCP-ASCP Cervical Cancer Guideline Committee. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62:147-172.
- Melnikow J, Henderson JT, Burda BU, et al. Screening for cervical cancer with high-risk human papillomavirus testing: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320:687-705.
- Canfell K, Caruana M, Gebski V, et al. Cervical screening with primary HPV testing or cytology in a population of women in which those aged 33 years or younger had previously been offered HPV vaccination: results of the Compass pilot randomised trial. PLoS Med. 2017;14:e1002388. doi:10.1371/journal.pmed.1002388.
- Leinonen MK, Nieminen P, Lonnberg S, et al. Detection rates of precancerous and cancerous cervical lesions within one screening round of primary human papillomavirus DNA testing: prospective randomised trial in Finland. BMJ. 2012;345:e7789.
- Ogilvie GS, van Niekerk D, Krajden M, et al. Effect of screening with primary cervical HPV testing vs cytology testing on high-grade cervical intraepithelial neoplasia at 48 months: the HPV FOCAL randomized clinical trial. JAMA. 2018;320:43-52.
- Ronco G, Fioprgi-Rossi P, Carozzi F, et al; New Technologies for Cervical Cancer screening (NTCC) Working Group. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomised controlled trial. Lancet Oncol. 2010;11:249-257.
- US Preventive Services Task Force, Curry SJ, Krist AH, et al. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
Cannabidiol (CBD) for schizophrenia: Promise or pipe dream?
Over the past few decades, it has become increasingly clear that cannabis use can increase the risk of developing a psychotic disorder and worsen the course of existing schizophrenia in a dose-dependent fashion.1-3 Beyond psychosis, although many patients with mental illness use cannabis for recreational purposes or as purported “self-medication,” currently available evidence suggests that marijuana is more likely to represent a harm than a benefit for psychiatric disorders4 (Box4-8). Our current state of knowledge therefore suggests that psychiatrists should caution their patients against using cannabis and prioritize interventions to reduce or discontinue use, especially among those with psychotic disorders.
Box
Data from California in 2006—a decade after the state’s legalization of “medical marijuana”—revealed that 23% of patients in a sample enrolled in medical marijuana clinics were receiving cannabis to treat a mental disorder.5 That was a striking statistic given the dearth of evidence to support a benefit of cannabis for psychiatric conditions at the time, leaving clinicians who provided the necessary recommendations to obtain medical marijuana largely unable to give informed consent about the risks and benefits, much less recommendations about specific products, routes of administration, or dosing. In 2019, we know considerably more about the interaction between cannabinoids and mental health, but research findings thus far warrant more caution than enthusiasm, with one recent review concluding that “whenever an association is observed between cannabis use and psychiatric disorders, the relationship is generally an adverse one.”4
Some critics have argued that the medical marijuana industry represents little more than a front for recreational use. In California and other states that have legalized recreational use, that claim has been rendered all but moot, although the public remains curious about the potential health benefits of cannabinoids and will likely continue to look to clinicians for advice. For those seeking guidance from evidence-based research, the existing state of knowledge can seem like a “Wild West” of anecdotal subjective reports, biased opinions, and uncontrolled clinical studies. Cannabis remains a Schedule I drug at the federal level, and quality clinical research has been limited to a relatively modest number of randomized controlled trials (RCTs), mostly involving FDA-approved cannabinoids rather than smoked cannabis. Randomized controlled trials that have involved smoked marijuana have generally involved low-potency delta-9-tetrahydrocannabinol (THC) cannabis that may not reflect the same therapeutic and adverse effects of the increasingly high potency cannabis now available on the street and in dispensaries.
In psychiatry, a few RCTs are underway exploring cannabis as a viable treatment for mental disorders (eg, posttraumatic stress disorder), but none have yet been completed or published. At best, retrospective studies to date have failed to support a consistent benefit of cannabis for any psychiatric disorder and at worst increasingly suggest a negative impact on psychotic, mood, and anxiety disorders.4,6 Meanwhile, synthetic cannabinoid receptor agonists (eg, “Spice” products) have come to represent a clear public health risk, with both medical and psychiatric toxicity.7
A more cautiously optimistic case for the therapeutic potential of cannabinoids in psychiatry could be made for cannabidiol (CBD), which may possess anxiolytic, antipsychotic, and neuroprotective properties.8 Based on its purported health benefits, it is possible that CBD may even gain widespread popularity as a food supplement. Because a pharmaceutically-manufactured form of CBD was recently FDA-approved for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome, off-label prescribing of CBD for psychiatric disorders can be anticipated. While there is not yet sufficient evidence about risks and benefits to justify CBD being recommended broadly in psychiatry, that same informational vacuum has not stopped eager patients from seeking approval for cannabis, and some physicians from providing it.
Despite that conclusion, because cannabis is classified as a Schedule I drug by the US Drug Enforcement Agency, clinical research investigating the risks and benefits of cannabis has been limited. It therefore remains possible that cannabis, or individual cannabinoids such as cannabidiol (CBD), may yet find a therapeutic niche in psychiatry. This article reviews evidence on CBD for the treatment of schizophrenia.
Cannabinergic drugs as potential antipsychotics
Although the bulk of evidence indicates a harmful effect of cannabis in individuals with or at risk for psychosis, there have been a few published cases of schizophrenia improving with dronabinol, an FDA-approved, synthetic form of delta-9-tetrahydrocannabinol (THC).9,10 THC is the constituent of cannabis that produces euphoric effects. These provocative findings have not been replicated in controlled clinical trials, but suggest at least the theoretical possibility of idiosyncratic benefits from THC for some individuals within the psychotic spectrum.
Still, given that most available evidence supports that THC has a harmful effect on psychosis and psychosis risk, researchers have instead performed randomized controlled trials (RCTs) to investigate a possible therapeutic role for medications that oppose the agonist effects of THC at cannabinoid type 1 (CB1) receptors. To date, 2 RCTs comparing rimonabant, a CB1 inverse agonist, with placebo (PLB) in patients with schizophrenia have failed to demonstrate any benefit for psychotic symptoms or cognitive deficits.11,12 A third trial examining rimonabant for people diagnosed with schizophrenia who were overweight found significant benefits for anxiety and depressive symptoms, but none for positive symptoms or the primary outcome of weight loss.13 While these results are discouraging, the role of THC in precipitating psychosis suggests that novel agents opposing the actions of THC on the cannabinoid system could have antipsychotic properties.14
Cannabidiol: An antipsychotic medication?
In contrast to THC, CBD has minimal euphorigenic properties and has recently been heralded in the popular press as a “miracle drug” with benefits for medical and psychiatric disorders alike.15 It has even been speculated that it could become a popular food supplement.16 In 2018, the FDA gave full approval to a pharmaceutically manufactured form of CBD (brand name: Epidiolex) as a novel treatment for 2 rare and severe forms of pediatric epilepsy, Lennox-Gastaut syndrome and Dravet syndrome,17 based on RCTs supporting its efficacy for these often refractory and life-threatening conditions.18-20
In psychiatry, there have not yet been enough robust clinical studies to support broad therapeutic claims for CBD as a treatment for any mental disorder.21 However, there is growing evidence that CBD has potential as an antipsychotic medication. In 1995, the first case report was published describing the efficacy of CBD, 1,500 mg/d, as standalone therapy in a single individual with schizophrenia.22 In 2006, the same research group followed up with a case series in which only 1 out of 3 patients with treatment-refractory schizophrenia improved with flexible dosing of CBD to a maximum dose of 1,280 mg/d.23
There have been 3 published RCTs exploring the efficacy of CBD in schizophrenia (Table24-26). The first study, published in 2012, included 39 adults with schizophrenia who were randomized to 800 mg/d of CBD or amisulpride (AMS), a second-generation antipsychotic that is popular in Europe but is not available in the United States.24 Over 4 weeks of randomized treatment, CBD resulted in as much improvement in overall symptoms and positive symptoms as AMS, and improvement of negative symptoms was significantly greater with CBD. Compared with patients treated with antipsychotic medication, patients who were treated with CBD had fewer extrapyramidal symptoms, less weight gain, and less prolactin elevation. This initial trial suggests that CBD might be as efficacious in schizophrenia as antipsychotic medication, without its burdensome adverse effects. However, this is the only RCT of CBD monotherapy published to date.
Continue to: Two other recently published RCTs...
Two other recently published RCTs compared CBD with PLB as add-on therapy to antipsychotics. McGuire et al25 compared CBD, 1,000 mg/d, to PLB over 6 weeks in 88 patients with schizophrenia. Positive symptom improvement was statistically greater with CBD than with PLB, although the magnitude of clinical change was modest (using the Positive and Negative Syndrome Scale [PANSS] positive symptom subscale: −3.2 points for CBD vs −1.7 points for PLB). Changes in PANSS total score and subscales for general and negative symptoms were not significantly different between treatment groups. There was also no significant difference in overall change in neurocognitive symptoms, although post-hoc analysis revealed significantly greater improvement in motor speed for patients treated with CBD. More than twice the number of patients treated with CBD were rated as “much improved” by the Clinical Global Impressions scale compared with patients treated with PLB, but this was not a statistically significant finding, and most patients experienced only “minimal” or “no improvement.” In terms of adverse events, there were no significant differences between patients in the CBD and PLB groups. Although this study is technically “positive” for CBD and suggests minimal adverse effects, it is not clear whether the statistically significant positive symptom improvements (+1.5 PANSS points for CBD over PLB) were clinically significant.
The most recently published placebo-controlled RCT of CBD as add-on therapy to antipsychotic medication included 36 patients with schizophrenia treated over 6 weeks.26 In this study, there was no benefit of CBD, 600 mg/d, on any PANSS score outcome (total, general, positive, or negative symptoms). For the primary outcome of the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery, there were no significant drug × time effects, and post-hoc analyses showed that only patients treated with PLB improved with time. Sedation was more common among patients treated with CBD compared with PLB.
Making sense of the data
There have been mixed results from the few case reports and 3 RCTs of patients with schizophrenia who were treated with CBD. How can we resolve these disparate findings? A few possible interpretations of the data that warrant clarification through additional research include:
Dosing. In the first case report with positive results, CBD was dosed at 1,500 mg/d,22 whereas in the subsequent case series with mixed results, the maximum allowable dose of CBD was 1,280 mg/d.23 Likewise, in the RCTs, positive results were found when CBD was dosed at 800 to 1,000 mg/d,24,25 but not at 600 mg/d.26 The efficacy of CBD for schizophrenia might depend on higher doses.
Treatment resistance. In the second case series in which only 1 out of 3 patients responded to treatment with CBD,23 the patients had demonstrated previous nonresponse to at least 2 first-generation antipsychotics (FGAs) and risperidone, 6 mg/d. In the RCTs, all patients were antipsychotic-responsive.24-26 Cannabidiol may not be as effective for patients with treatment-refractory schizophrenia as it is for patients with schizophrenia who respond to antipsychotics.
Continue to: Clinical stability
Clinical stability. Within the RCTs, the greatest response was observed in the study that enrolled patients who were hospitalized with acute symptoms of schizophrenia.23 In the 2 studies that found either modest or no benefit with CBD, the patients had been stabilized on antipsychotic medications prior to randomization. Cannabidiol may offer limited benefit as add-on therapy to patients who have already responded to antipsychotic treatment, where there is “less room” for additional improvement.
Monotherapy. Both the case reports22,23 and the RCT with the most robust positive findings24 involved treatment with CBD as monotherapy. For some patients with schizophrenia, CBD might be effective as standalone therapy as an alternative to antipsychotics that is better tolerated. Adding CBD to antipsychotic therapy might be redundant and therefore less effective.
Answering questions about CBD
Cannabidiol is becoming increasingly popular for its purported health benefits. The mixed results of the few studies published on CBD for schizophrenia place clinicians in a difficult position when attempting to answer questions about how cannabinoids might fit into treatment of patients with psychosis. Consider the following:
Is cannabis helpful for patients with schizophrenia? No. Aside from the few case reports suggesting that FDA-approved THC (dronabinol) can improve symptoms in some patients,9,10 most of the evidence from anecdotal reports and both experimental and observational studies indicate that cannabis, THC, and synthetic cannabinoids have a harmful effect in patients with or at risk for psychosis.1-3
If you are considering recommending some form of cannabis to patients with schizophrenia, what kind should you recommend? Recommending or encouraging cannabis use for patients with psychosis is ill-advised. Although certain types of cannabis might contain more THC (eg, Cannabis indica vs Cannabis sativa) or variable amounts of CBD, in general the amount of CBD in whole leaf cannabis is minimal, with the ratio of THC to CBD increasingly significantly over the past decade.3,27 Most forms of cannabis should therefore be avoided by individuals with or at risk for psychotic disorders.
Continue to: What about CBD oil and other CBD products sold in dispensaries?
What about CBD oil and other CBD products sold in dispensaries? Cannabidiol is increasingly available in various forms based on its ability to be designated as a legal hemp product (containing <0.3% THC) at the federal level or as a cannabinoid in states where cannabis is legal. However, several studies have now shown that cannabis products sold online or in dispensaries are often labeled inaccurately, with both under- and over-reporting of THC and CBD content.28-30 Some CBD products have been found to have almost no CBD at all.29,30 The unreliability of product labeling makes it difficult to predict the effects of CBD products that are not subject to FDA purity standards for medications or dietary supplements. It also raises questions about the sources of CBD and the reliability of dosing in the studies discussed above.
Why might CBD work as an antipsychotic? Although CBD has minimal affinity for cannabinoid receptors, it appears to act as a partial agonist of dopamine D2 receptors and an agonist at 5-HT1A receptors, with overall effects that decrease mesolimbic dopamine activity.31,32 In addition, CBD increases the availability of the endogenous cannabinoid anandamide, which may have antipsychotic properties.14,33
Now that the FDA has approved CBD manufactured by a pharmaceutical company, should it be prescribed “off-label” for patients with schizophrenia? This is the “million dollar question,” with insufficient evidence to provide a clear answer. It should now be possible to prescribe FDA-approved CBD for off-label purposes, including the treatment of schizophrenia and other psychiatric disorders. No doubt, some clinicians are already doing so. This will predictably yield more anecdotal evidence about efficacy and adverse effects in the future, but there is not yet adequate evidence to support an FDA indication for CBD in schizophrenia. Additional studies of CBD for schizophrenia are ongoing.
Bottom Line
Cannabidiol (CBD) is becoming increasingly popular based on its purported health benefits, but the evidence supporting a therapeutic role in psychiatry is preliminary at best. Although CBD is now available by prescription as an FDA-approved drug for the treatment of 2 rare forms of epilepsy, its benefits in patients with schizophrenia are uncertain based on mixed results in clinical trials.
Related Resources
- Clinicaltrials.gov. Studies of “cannabidiol” and “schizophrenia.” U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=Schizophrenia&term=cannabidiol.
- Grinspoon P. Cannabidiol (CBD) – what we know and what we don’t. Harvard Health Blog. https://www.health.harvard.edu/blog/cannabidiol-cbd-what-we-know-and-what-wedont-2018082414476. Published August 24, 2018.
Drug Brand Names
Cannabidiol • Epidiolex
Dronabinol • Marinol
Risperidone • Risperdal
1. Pierre JM. Cannabis, synthetic cannabinoids, and psychosis risk: what the evidence says. Current Psychiatry. 2011;10(9):49-58.
2. Radhakrishan R, Wilkinson ST, D’Souza DC. Gone to pot – a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54.
3. Pierre JM. Risks of increasingly potent cannabis: joint effects of potency and frequency. Current Psychiatry. 2016;16(2):14-20.
4. Hanna RC, Perez JM, Ghose S. Cannabis and development of dual diagnoses: a literature review. Am J Drug Alcohol Abuse. 2017;43(4):442-255.
5. Nunberg H, Kilmer B, Pacula RL, et al. An analysis of applicants presenting to a medical marijuana specialty practice in California. J Drug Policy Anal. 2011;4(1):1.
6. Wilkinson ST, Radhakrishnan, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064.
7. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: Update 2015. Subst Abus. 2016;38(3):344-366.
8. Crippa JA, Guimarães FS, Campos A, et al. Translational investigation of the therapeutic potential of cannabidiol (CBD): toward a new age. Front Immunol. 2018;9:2009.
9. Schwarz G, Karajgi B. Improvement in refractory psychosis with dronabinol: four case reports. J Clin Psychiatry. 2010;71(11):1552-1553.
10. Schwarz G, Karajgi B, McCarthy R. Synthetic delta-9-tetrahydrocannabinol (dronabinol) can improve the symptoms of schizophrenia. J Clin Psychopharmacol. 2009;29(3):255-258.
11. Meltzer HY, Arvanitis L, Bauer D, et al. Placebo-controlled evaluation of four novel compounds for the treatment of schizophrenia and schizoaffective disorder. Am J Psychiatry. 2004;161(6):975-984.
12. Boggs DL, Kelly DL, McMahon RP, et al. Rimonabant for neurocognition in schizophrenia: a 16-week double blind placebo controlled trial. Schizophr Res. 2012;134(2-3):207-210.
13. Kelly DL, Gorelick DA, Conley RR, et al. Effects of cannabinoid-1 receptor antagonist rimonabant on psychiatric symptoms in overweight people with schizophrenia: a randomized, double-blind, pilot study. J Clin Psychopharmacol. 2011;31(1):86-91.
14. Leweke FM, Mueller JK, Lange B, et al. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):604-612.
15. Halperin A. What is CBD? The ‘miracle’ cannabis compound that doesn’t get you high. The Guardian. https://www.theguardian.com/society/2018/may/28/what-is-cbd-cannabidiol-cannabis-medical-uses. Published May 28, 2018. Accessed April 3, 2019.
16. Pierre J. Coca, cola, and cannabis: psychoactive drugs as beverages. Psychology Today (blog) Psych Unseen. https://www.psychologytoday.com/us/blog/psych-unseen/201810/coca-cola-and-cannabis-psychoactive-drugs-beverages. Published October 1, 2018. Accessed April 3, 2019.
17. U.S. Food and Drug Administration. FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. FDA News Release. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm611046.htm. Published June 25, 2018. Accessed April 3, 2019.
18. Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011-2020.
19. Thiele EA, March ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
20. Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
21. Khoury JM, Neves MCLD, Rogue MAV, et al. Is there a role of cannabidiol in psychiatry? World J Biol Psychiatry. 2017:1-16.
22. Zuardi AW, Morais SL, Guimares FS, et al. Antipsychotic effect of cannabidiol. J Clin Psychiatry. 1995;56(10):485-486.
23. Zuardi AW, Hallak JEC, Dursun SM. Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol. 2006;20(5):683-686.
24. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2:e94. doi: 10.1038/tp.2012.15.
25. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225-231.
26. Boggs DL, Surti I, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacol. 2018;235(7):1923-1932.
27. ElSohly MA, Mehmedic Z, Foster S, et al. Changes in cannabis potency over the last 2 decades (1995-2014): analysis of current data in the United States. Biol Psychiatry. 2016; 79(7):613-619.
28. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491-2492.
29. Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, et al. Consistency of label claims of internet-purchased hemp oil and cannabis products as determined using IMS and LC-MS: a marketplace study. J Reg Sci. 2016;3:1-6.
30. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318(17):1708-1709.
31. Seeman P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl Psychiatry. 2016;6(10):e920. doi: 10.1038/tp.2016.195.
32. Renard J, Norris C, Rushlow W, et al. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;157-165.
33. Gururajan A, Malone DT. Does cannabidiol have a role in the treatment of schizophrenia? Schizophr Res. 2016;176(2-3):281-290.
Over the past few decades, it has become increasingly clear that cannabis use can increase the risk of developing a psychotic disorder and worsen the course of existing schizophrenia in a dose-dependent fashion.1-3 Beyond psychosis, although many patients with mental illness use cannabis for recreational purposes or as purported “self-medication,” currently available evidence suggests that marijuana is more likely to represent a harm than a benefit for psychiatric disorders4 (Box4-8). Our current state of knowledge therefore suggests that psychiatrists should caution their patients against using cannabis and prioritize interventions to reduce or discontinue use, especially among those with psychotic disorders.
Box
Data from California in 2006—a decade after the state’s legalization of “medical marijuana”—revealed that 23% of patients in a sample enrolled in medical marijuana clinics were receiving cannabis to treat a mental disorder.5 That was a striking statistic given the dearth of evidence to support a benefit of cannabis for psychiatric conditions at the time, leaving clinicians who provided the necessary recommendations to obtain medical marijuana largely unable to give informed consent about the risks and benefits, much less recommendations about specific products, routes of administration, or dosing. In 2019, we know considerably more about the interaction between cannabinoids and mental health, but research findings thus far warrant more caution than enthusiasm, with one recent review concluding that “whenever an association is observed between cannabis use and psychiatric disorders, the relationship is generally an adverse one.”4
Some critics have argued that the medical marijuana industry represents little more than a front for recreational use. In California and other states that have legalized recreational use, that claim has been rendered all but moot, although the public remains curious about the potential health benefits of cannabinoids and will likely continue to look to clinicians for advice. For those seeking guidance from evidence-based research, the existing state of knowledge can seem like a “Wild West” of anecdotal subjective reports, biased opinions, and uncontrolled clinical studies. Cannabis remains a Schedule I drug at the federal level, and quality clinical research has been limited to a relatively modest number of randomized controlled trials (RCTs), mostly involving FDA-approved cannabinoids rather than smoked cannabis. Randomized controlled trials that have involved smoked marijuana have generally involved low-potency delta-9-tetrahydrocannabinol (THC) cannabis that may not reflect the same therapeutic and adverse effects of the increasingly high potency cannabis now available on the street and in dispensaries.
In psychiatry, a few RCTs are underway exploring cannabis as a viable treatment for mental disorders (eg, posttraumatic stress disorder), but none have yet been completed or published. At best, retrospective studies to date have failed to support a consistent benefit of cannabis for any psychiatric disorder and at worst increasingly suggest a negative impact on psychotic, mood, and anxiety disorders.4,6 Meanwhile, synthetic cannabinoid receptor agonists (eg, “Spice” products) have come to represent a clear public health risk, with both medical and psychiatric toxicity.7
A more cautiously optimistic case for the therapeutic potential of cannabinoids in psychiatry could be made for cannabidiol (CBD), which may possess anxiolytic, antipsychotic, and neuroprotective properties.8 Based on its purported health benefits, it is possible that CBD may even gain widespread popularity as a food supplement. Because a pharmaceutically-manufactured form of CBD was recently FDA-approved for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome, off-label prescribing of CBD for psychiatric disorders can be anticipated. While there is not yet sufficient evidence about risks and benefits to justify CBD being recommended broadly in psychiatry, that same informational vacuum has not stopped eager patients from seeking approval for cannabis, and some physicians from providing it.
Despite that conclusion, because cannabis is classified as a Schedule I drug by the US Drug Enforcement Agency, clinical research investigating the risks and benefits of cannabis has been limited. It therefore remains possible that cannabis, or individual cannabinoids such as cannabidiol (CBD), may yet find a therapeutic niche in psychiatry. This article reviews evidence on CBD for the treatment of schizophrenia.
Cannabinergic drugs as potential antipsychotics
Although the bulk of evidence indicates a harmful effect of cannabis in individuals with or at risk for psychosis, there have been a few published cases of schizophrenia improving with dronabinol, an FDA-approved, synthetic form of delta-9-tetrahydrocannabinol (THC).9,10 THC is the constituent of cannabis that produces euphoric effects. These provocative findings have not been replicated in controlled clinical trials, but suggest at least the theoretical possibility of idiosyncratic benefits from THC for some individuals within the psychotic spectrum.
Still, given that most available evidence supports that THC has a harmful effect on psychosis and psychosis risk, researchers have instead performed randomized controlled trials (RCTs) to investigate a possible therapeutic role for medications that oppose the agonist effects of THC at cannabinoid type 1 (CB1) receptors. To date, 2 RCTs comparing rimonabant, a CB1 inverse agonist, with placebo (PLB) in patients with schizophrenia have failed to demonstrate any benefit for psychotic symptoms or cognitive deficits.11,12 A third trial examining rimonabant for people diagnosed with schizophrenia who were overweight found significant benefits for anxiety and depressive symptoms, but none for positive symptoms or the primary outcome of weight loss.13 While these results are discouraging, the role of THC in precipitating psychosis suggests that novel agents opposing the actions of THC on the cannabinoid system could have antipsychotic properties.14
Cannabidiol: An antipsychotic medication?
In contrast to THC, CBD has minimal euphorigenic properties and has recently been heralded in the popular press as a “miracle drug” with benefits for medical and psychiatric disorders alike.15 It has even been speculated that it could become a popular food supplement.16 In 2018, the FDA gave full approval to a pharmaceutically manufactured form of CBD (brand name: Epidiolex) as a novel treatment for 2 rare and severe forms of pediatric epilepsy, Lennox-Gastaut syndrome and Dravet syndrome,17 based on RCTs supporting its efficacy for these often refractory and life-threatening conditions.18-20
In psychiatry, there have not yet been enough robust clinical studies to support broad therapeutic claims for CBD as a treatment for any mental disorder.21 However, there is growing evidence that CBD has potential as an antipsychotic medication. In 1995, the first case report was published describing the efficacy of CBD, 1,500 mg/d, as standalone therapy in a single individual with schizophrenia.22 In 2006, the same research group followed up with a case series in which only 1 out of 3 patients with treatment-refractory schizophrenia improved with flexible dosing of CBD to a maximum dose of 1,280 mg/d.23
There have been 3 published RCTs exploring the efficacy of CBD in schizophrenia (Table24-26). The first study, published in 2012, included 39 adults with schizophrenia who were randomized to 800 mg/d of CBD or amisulpride (AMS), a second-generation antipsychotic that is popular in Europe but is not available in the United States.24 Over 4 weeks of randomized treatment, CBD resulted in as much improvement in overall symptoms and positive symptoms as AMS, and improvement of negative symptoms was significantly greater with CBD. Compared with patients treated with antipsychotic medication, patients who were treated with CBD had fewer extrapyramidal symptoms, less weight gain, and less prolactin elevation. This initial trial suggests that CBD might be as efficacious in schizophrenia as antipsychotic medication, without its burdensome adverse effects. However, this is the only RCT of CBD monotherapy published to date.
Continue to: Two other recently published RCTs...
Two other recently published RCTs compared CBD with PLB as add-on therapy to antipsychotics. McGuire et al25 compared CBD, 1,000 mg/d, to PLB over 6 weeks in 88 patients with schizophrenia. Positive symptom improvement was statistically greater with CBD than with PLB, although the magnitude of clinical change was modest (using the Positive and Negative Syndrome Scale [PANSS] positive symptom subscale: −3.2 points for CBD vs −1.7 points for PLB). Changes in PANSS total score and subscales for general and negative symptoms were not significantly different between treatment groups. There was also no significant difference in overall change in neurocognitive symptoms, although post-hoc analysis revealed significantly greater improvement in motor speed for patients treated with CBD. More than twice the number of patients treated with CBD were rated as “much improved” by the Clinical Global Impressions scale compared with patients treated with PLB, but this was not a statistically significant finding, and most patients experienced only “minimal” or “no improvement.” In terms of adverse events, there were no significant differences between patients in the CBD and PLB groups. Although this study is technically “positive” for CBD and suggests minimal adverse effects, it is not clear whether the statistically significant positive symptom improvements (+1.5 PANSS points for CBD over PLB) were clinically significant.
The most recently published placebo-controlled RCT of CBD as add-on therapy to antipsychotic medication included 36 patients with schizophrenia treated over 6 weeks.26 In this study, there was no benefit of CBD, 600 mg/d, on any PANSS score outcome (total, general, positive, or negative symptoms). For the primary outcome of the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery, there were no significant drug × time effects, and post-hoc analyses showed that only patients treated with PLB improved with time. Sedation was more common among patients treated with CBD compared with PLB.
Making sense of the data
There have been mixed results from the few case reports and 3 RCTs of patients with schizophrenia who were treated with CBD. How can we resolve these disparate findings? A few possible interpretations of the data that warrant clarification through additional research include:
Dosing. In the first case report with positive results, CBD was dosed at 1,500 mg/d,22 whereas in the subsequent case series with mixed results, the maximum allowable dose of CBD was 1,280 mg/d.23 Likewise, in the RCTs, positive results were found when CBD was dosed at 800 to 1,000 mg/d,24,25 but not at 600 mg/d.26 The efficacy of CBD for schizophrenia might depend on higher doses.
Treatment resistance. In the second case series in which only 1 out of 3 patients responded to treatment with CBD,23 the patients had demonstrated previous nonresponse to at least 2 first-generation antipsychotics (FGAs) and risperidone, 6 mg/d. In the RCTs, all patients were antipsychotic-responsive.24-26 Cannabidiol may not be as effective for patients with treatment-refractory schizophrenia as it is for patients with schizophrenia who respond to antipsychotics.
Continue to: Clinical stability
Clinical stability. Within the RCTs, the greatest response was observed in the study that enrolled patients who were hospitalized with acute symptoms of schizophrenia.23 In the 2 studies that found either modest or no benefit with CBD, the patients had been stabilized on antipsychotic medications prior to randomization. Cannabidiol may offer limited benefit as add-on therapy to patients who have already responded to antipsychotic treatment, where there is “less room” for additional improvement.
Monotherapy. Both the case reports22,23 and the RCT with the most robust positive findings24 involved treatment with CBD as monotherapy. For some patients with schizophrenia, CBD might be effective as standalone therapy as an alternative to antipsychotics that is better tolerated. Adding CBD to antipsychotic therapy might be redundant and therefore less effective.
Answering questions about CBD
Cannabidiol is becoming increasingly popular for its purported health benefits. The mixed results of the few studies published on CBD for schizophrenia place clinicians in a difficult position when attempting to answer questions about how cannabinoids might fit into treatment of patients with psychosis. Consider the following:
Is cannabis helpful for patients with schizophrenia? No. Aside from the few case reports suggesting that FDA-approved THC (dronabinol) can improve symptoms in some patients,9,10 most of the evidence from anecdotal reports and both experimental and observational studies indicate that cannabis, THC, and synthetic cannabinoids have a harmful effect in patients with or at risk for psychosis.1-3
If you are considering recommending some form of cannabis to patients with schizophrenia, what kind should you recommend? Recommending or encouraging cannabis use for patients with psychosis is ill-advised. Although certain types of cannabis might contain more THC (eg, Cannabis indica vs Cannabis sativa) or variable amounts of CBD, in general the amount of CBD in whole leaf cannabis is minimal, with the ratio of THC to CBD increasingly significantly over the past decade.3,27 Most forms of cannabis should therefore be avoided by individuals with or at risk for psychotic disorders.
Continue to: What about CBD oil and other CBD products sold in dispensaries?
What about CBD oil and other CBD products sold in dispensaries? Cannabidiol is increasingly available in various forms based on its ability to be designated as a legal hemp product (containing <0.3% THC) at the federal level or as a cannabinoid in states where cannabis is legal. However, several studies have now shown that cannabis products sold online or in dispensaries are often labeled inaccurately, with both under- and over-reporting of THC and CBD content.28-30 Some CBD products have been found to have almost no CBD at all.29,30 The unreliability of product labeling makes it difficult to predict the effects of CBD products that are not subject to FDA purity standards for medications or dietary supplements. It also raises questions about the sources of CBD and the reliability of dosing in the studies discussed above.
Why might CBD work as an antipsychotic? Although CBD has minimal affinity for cannabinoid receptors, it appears to act as a partial agonist of dopamine D2 receptors and an agonist at 5-HT1A receptors, with overall effects that decrease mesolimbic dopamine activity.31,32 In addition, CBD increases the availability of the endogenous cannabinoid anandamide, which may have antipsychotic properties.14,33
Now that the FDA has approved CBD manufactured by a pharmaceutical company, should it be prescribed “off-label” for patients with schizophrenia? This is the “million dollar question,” with insufficient evidence to provide a clear answer. It should now be possible to prescribe FDA-approved CBD for off-label purposes, including the treatment of schizophrenia and other psychiatric disorders. No doubt, some clinicians are already doing so. This will predictably yield more anecdotal evidence about efficacy and adverse effects in the future, but there is not yet adequate evidence to support an FDA indication for CBD in schizophrenia. Additional studies of CBD for schizophrenia are ongoing.
Bottom Line
Cannabidiol (CBD) is becoming increasingly popular based on its purported health benefits, but the evidence supporting a therapeutic role in psychiatry is preliminary at best. Although CBD is now available by prescription as an FDA-approved drug for the treatment of 2 rare forms of epilepsy, its benefits in patients with schizophrenia are uncertain based on mixed results in clinical trials.
Related Resources
- Clinicaltrials.gov. Studies of “cannabidiol” and “schizophrenia.” U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=Schizophrenia&term=cannabidiol.
- Grinspoon P. Cannabidiol (CBD) – what we know and what we don’t. Harvard Health Blog. https://www.health.harvard.edu/blog/cannabidiol-cbd-what-we-know-and-what-wedont-2018082414476. Published August 24, 2018.
Drug Brand Names
Cannabidiol • Epidiolex
Dronabinol • Marinol
Risperidone • Risperdal
Over the past few decades, it has become increasingly clear that cannabis use can increase the risk of developing a psychotic disorder and worsen the course of existing schizophrenia in a dose-dependent fashion.1-3 Beyond psychosis, although many patients with mental illness use cannabis for recreational purposes or as purported “self-medication,” currently available evidence suggests that marijuana is more likely to represent a harm than a benefit for psychiatric disorders4 (Box4-8). Our current state of knowledge therefore suggests that psychiatrists should caution their patients against using cannabis and prioritize interventions to reduce or discontinue use, especially among those with psychotic disorders.
Box
Data from California in 2006—a decade after the state’s legalization of “medical marijuana”—revealed that 23% of patients in a sample enrolled in medical marijuana clinics were receiving cannabis to treat a mental disorder.5 That was a striking statistic given the dearth of evidence to support a benefit of cannabis for psychiatric conditions at the time, leaving clinicians who provided the necessary recommendations to obtain medical marijuana largely unable to give informed consent about the risks and benefits, much less recommendations about specific products, routes of administration, or dosing. In 2019, we know considerably more about the interaction between cannabinoids and mental health, but research findings thus far warrant more caution than enthusiasm, with one recent review concluding that “whenever an association is observed between cannabis use and psychiatric disorders, the relationship is generally an adverse one.”4
Some critics have argued that the medical marijuana industry represents little more than a front for recreational use. In California and other states that have legalized recreational use, that claim has been rendered all but moot, although the public remains curious about the potential health benefits of cannabinoids and will likely continue to look to clinicians for advice. For those seeking guidance from evidence-based research, the existing state of knowledge can seem like a “Wild West” of anecdotal subjective reports, biased opinions, and uncontrolled clinical studies. Cannabis remains a Schedule I drug at the federal level, and quality clinical research has been limited to a relatively modest number of randomized controlled trials (RCTs), mostly involving FDA-approved cannabinoids rather than smoked cannabis. Randomized controlled trials that have involved smoked marijuana have generally involved low-potency delta-9-tetrahydrocannabinol (THC) cannabis that may not reflect the same therapeutic and adverse effects of the increasingly high potency cannabis now available on the street and in dispensaries.
In psychiatry, a few RCTs are underway exploring cannabis as a viable treatment for mental disorders (eg, posttraumatic stress disorder), but none have yet been completed or published. At best, retrospective studies to date have failed to support a consistent benefit of cannabis for any psychiatric disorder and at worst increasingly suggest a negative impact on psychotic, mood, and anxiety disorders.4,6 Meanwhile, synthetic cannabinoid receptor agonists (eg, “Spice” products) have come to represent a clear public health risk, with both medical and psychiatric toxicity.7
A more cautiously optimistic case for the therapeutic potential of cannabinoids in psychiatry could be made for cannabidiol (CBD), which may possess anxiolytic, antipsychotic, and neuroprotective properties.8 Based on its purported health benefits, it is possible that CBD may even gain widespread popularity as a food supplement. Because a pharmaceutically-manufactured form of CBD was recently FDA-approved for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome, off-label prescribing of CBD for psychiatric disorders can be anticipated. While there is not yet sufficient evidence about risks and benefits to justify CBD being recommended broadly in psychiatry, that same informational vacuum has not stopped eager patients from seeking approval for cannabis, and some physicians from providing it.
Despite that conclusion, because cannabis is classified as a Schedule I drug by the US Drug Enforcement Agency, clinical research investigating the risks and benefits of cannabis has been limited. It therefore remains possible that cannabis, or individual cannabinoids such as cannabidiol (CBD), may yet find a therapeutic niche in psychiatry. This article reviews evidence on CBD for the treatment of schizophrenia.
Cannabinergic drugs as potential antipsychotics
Although the bulk of evidence indicates a harmful effect of cannabis in individuals with or at risk for psychosis, there have been a few published cases of schizophrenia improving with dronabinol, an FDA-approved, synthetic form of delta-9-tetrahydrocannabinol (THC).9,10 THC is the constituent of cannabis that produces euphoric effects. These provocative findings have not been replicated in controlled clinical trials, but suggest at least the theoretical possibility of idiosyncratic benefits from THC for some individuals within the psychotic spectrum.
Still, given that most available evidence supports that THC has a harmful effect on psychosis and psychosis risk, researchers have instead performed randomized controlled trials (RCTs) to investigate a possible therapeutic role for medications that oppose the agonist effects of THC at cannabinoid type 1 (CB1) receptors. To date, 2 RCTs comparing rimonabant, a CB1 inverse agonist, with placebo (PLB) in patients with schizophrenia have failed to demonstrate any benefit for psychotic symptoms or cognitive deficits.11,12 A third trial examining rimonabant for people diagnosed with schizophrenia who were overweight found significant benefits for anxiety and depressive symptoms, but none for positive symptoms or the primary outcome of weight loss.13 While these results are discouraging, the role of THC in precipitating psychosis suggests that novel agents opposing the actions of THC on the cannabinoid system could have antipsychotic properties.14
Cannabidiol: An antipsychotic medication?
In contrast to THC, CBD has minimal euphorigenic properties and has recently been heralded in the popular press as a “miracle drug” with benefits for medical and psychiatric disorders alike.15 It has even been speculated that it could become a popular food supplement.16 In 2018, the FDA gave full approval to a pharmaceutically manufactured form of CBD (brand name: Epidiolex) as a novel treatment for 2 rare and severe forms of pediatric epilepsy, Lennox-Gastaut syndrome and Dravet syndrome,17 based on RCTs supporting its efficacy for these often refractory and life-threatening conditions.18-20
In psychiatry, there have not yet been enough robust clinical studies to support broad therapeutic claims for CBD as a treatment for any mental disorder.21 However, there is growing evidence that CBD has potential as an antipsychotic medication. In 1995, the first case report was published describing the efficacy of CBD, 1,500 mg/d, as standalone therapy in a single individual with schizophrenia.22 In 2006, the same research group followed up with a case series in which only 1 out of 3 patients with treatment-refractory schizophrenia improved with flexible dosing of CBD to a maximum dose of 1,280 mg/d.23
There have been 3 published RCTs exploring the efficacy of CBD in schizophrenia (Table24-26). The first study, published in 2012, included 39 adults with schizophrenia who were randomized to 800 mg/d of CBD or amisulpride (AMS), a second-generation antipsychotic that is popular in Europe but is not available in the United States.24 Over 4 weeks of randomized treatment, CBD resulted in as much improvement in overall symptoms and positive symptoms as AMS, and improvement of negative symptoms was significantly greater with CBD. Compared with patients treated with antipsychotic medication, patients who were treated with CBD had fewer extrapyramidal symptoms, less weight gain, and less prolactin elevation. This initial trial suggests that CBD might be as efficacious in schizophrenia as antipsychotic medication, without its burdensome adverse effects. However, this is the only RCT of CBD monotherapy published to date.
Continue to: Two other recently published RCTs...
Two other recently published RCTs compared CBD with PLB as add-on therapy to antipsychotics. McGuire et al25 compared CBD, 1,000 mg/d, to PLB over 6 weeks in 88 patients with schizophrenia. Positive symptom improvement was statistically greater with CBD than with PLB, although the magnitude of clinical change was modest (using the Positive and Negative Syndrome Scale [PANSS] positive symptom subscale: −3.2 points for CBD vs −1.7 points for PLB). Changes in PANSS total score and subscales for general and negative symptoms were not significantly different between treatment groups. There was also no significant difference in overall change in neurocognitive symptoms, although post-hoc analysis revealed significantly greater improvement in motor speed for patients treated with CBD. More than twice the number of patients treated with CBD were rated as “much improved” by the Clinical Global Impressions scale compared with patients treated with PLB, but this was not a statistically significant finding, and most patients experienced only “minimal” or “no improvement.” In terms of adverse events, there were no significant differences between patients in the CBD and PLB groups. Although this study is technically “positive” for CBD and suggests minimal adverse effects, it is not clear whether the statistically significant positive symptom improvements (+1.5 PANSS points for CBD over PLB) were clinically significant.
The most recently published placebo-controlled RCT of CBD as add-on therapy to antipsychotic medication included 36 patients with schizophrenia treated over 6 weeks.26 In this study, there was no benefit of CBD, 600 mg/d, on any PANSS score outcome (total, general, positive, or negative symptoms). For the primary outcome of the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery, there were no significant drug × time effects, and post-hoc analyses showed that only patients treated with PLB improved with time. Sedation was more common among patients treated with CBD compared with PLB.
Making sense of the data
There have been mixed results from the few case reports and 3 RCTs of patients with schizophrenia who were treated with CBD. How can we resolve these disparate findings? A few possible interpretations of the data that warrant clarification through additional research include:
Dosing. In the first case report with positive results, CBD was dosed at 1,500 mg/d,22 whereas in the subsequent case series with mixed results, the maximum allowable dose of CBD was 1,280 mg/d.23 Likewise, in the RCTs, positive results were found when CBD was dosed at 800 to 1,000 mg/d,24,25 but not at 600 mg/d.26 The efficacy of CBD for schizophrenia might depend on higher doses.
Treatment resistance. In the second case series in which only 1 out of 3 patients responded to treatment with CBD,23 the patients had demonstrated previous nonresponse to at least 2 first-generation antipsychotics (FGAs) and risperidone, 6 mg/d. In the RCTs, all patients were antipsychotic-responsive.24-26 Cannabidiol may not be as effective for patients with treatment-refractory schizophrenia as it is for patients with schizophrenia who respond to antipsychotics.
Continue to: Clinical stability
Clinical stability. Within the RCTs, the greatest response was observed in the study that enrolled patients who were hospitalized with acute symptoms of schizophrenia.23 In the 2 studies that found either modest or no benefit with CBD, the patients had been stabilized on antipsychotic medications prior to randomization. Cannabidiol may offer limited benefit as add-on therapy to patients who have already responded to antipsychotic treatment, where there is “less room” for additional improvement.
Monotherapy. Both the case reports22,23 and the RCT with the most robust positive findings24 involved treatment with CBD as monotherapy. For some patients with schizophrenia, CBD might be effective as standalone therapy as an alternative to antipsychotics that is better tolerated. Adding CBD to antipsychotic therapy might be redundant and therefore less effective.
Answering questions about CBD
Cannabidiol is becoming increasingly popular for its purported health benefits. The mixed results of the few studies published on CBD for schizophrenia place clinicians in a difficult position when attempting to answer questions about how cannabinoids might fit into treatment of patients with psychosis. Consider the following:
Is cannabis helpful for patients with schizophrenia? No. Aside from the few case reports suggesting that FDA-approved THC (dronabinol) can improve symptoms in some patients,9,10 most of the evidence from anecdotal reports and both experimental and observational studies indicate that cannabis, THC, and synthetic cannabinoids have a harmful effect in patients with or at risk for psychosis.1-3
If you are considering recommending some form of cannabis to patients with schizophrenia, what kind should you recommend? Recommending or encouraging cannabis use for patients with psychosis is ill-advised. Although certain types of cannabis might contain more THC (eg, Cannabis indica vs Cannabis sativa) or variable amounts of CBD, in general the amount of CBD in whole leaf cannabis is minimal, with the ratio of THC to CBD increasingly significantly over the past decade.3,27 Most forms of cannabis should therefore be avoided by individuals with or at risk for psychotic disorders.
Continue to: What about CBD oil and other CBD products sold in dispensaries?
What about CBD oil and other CBD products sold in dispensaries? Cannabidiol is increasingly available in various forms based on its ability to be designated as a legal hemp product (containing <0.3% THC) at the federal level or as a cannabinoid in states where cannabis is legal. However, several studies have now shown that cannabis products sold online or in dispensaries are often labeled inaccurately, with both under- and over-reporting of THC and CBD content.28-30 Some CBD products have been found to have almost no CBD at all.29,30 The unreliability of product labeling makes it difficult to predict the effects of CBD products that are not subject to FDA purity standards for medications or dietary supplements. It also raises questions about the sources of CBD and the reliability of dosing in the studies discussed above.
Why might CBD work as an antipsychotic? Although CBD has minimal affinity for cannabinoid receptors, it appears to act as a partial agonist of dopamine D2 receptors and an agonist at 5-HT1A receptors, with overall effects that decrease mesolimbic dopamine activity.31,32 In addition, CBD increases the availability of the endogenous cannabinoid anandamide, which may have antipsychotic properties.14,33
Now that the FDA has approved CBD manufactured by a pharmaceutical company, should it be prescribed “off-label” for patients with schizophrenia? This is the “million dollar question,” with insufficient evidence to provide a clear answer. It should now be possible to prescribe FDA-approved CBD for off-label purposes, including the treatment of schizophrenia and other psychiatric disorders. No doubt, some clinicians are already doing so. This will predictably yield more anecdotal evidence about efficacy and adverse effects in the future, but there is not yet adequate evidence to support an FDA indication for CBD in schizophrenia. Additional studies of CBD for schizophrenia are ongoing.
Bottom Line
Cannabidiol (CBD) is becoming increasingly popular based on its purported health benefits, but the evidence supporting a therapeutic role in psychiatry is preliminary at best. Although CBD is now available by prescription as an FDA-approved drug for the treatment of 2 rare forms of epilepsy, its benefits in patients with schizophrenia are uncertain based on mixed results in clinical trials.
Related Resources
- Clinicaltrials.gov. Studies of “cannabidiol” and “schizophrenia.” U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=Schizophrenia&term=cannabidiol.
- Grinspoon P. Cannabidiol (CBD) – what we know and what we don’t. Harvard Health Blog. https://www.health.harvard.edu/blog/cannabidiol-cbd-what-we-know-and-what-wedont-2018082414476. Published August 24, 2018.
Drug Brand Names
Cannabidiol • Epidiolex
Dronabinol • Marinol
Risperidone • Risperdal
1. Pierre JM. Cannabis, synthetic cannabinoids, and psychosis risk: what the evidence says. Current Psychiatry. 2011;10(9):49-58.
2. Radhakrishan R, Wilkinson ST, D’Souza DC. Gone to pot – a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54.
3. Pierre JM. Risks of increasingly potent cannabis: joint effects of potency and frequency. Current Psychiatry. 2016;16(2):14-20.
4. Hanna RC, Perez JM, Ghose S. Cannabis and development of dual diagnoses: a literature review. Am J Drug Alcohol Abuse. 2017;43(4):442-255.
5. Nunberg H, Kilmer B, Pacula RL, et al. An analysis of applicants presenting to a medical marijuana specialty practice in California. J Drug Policy Anal. 2011;4(1):1.
6. Wilkinson ST, Radhakrishnan, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064.
7. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: Update 2015. Subst Abus. 2016;38(3):344-366.
8. Crippa JA, Guimarães FS, Campos A, et al. Translational investigation of the therapeutic potential of cannabidiol (CBD): toward a new age. Front Immunol. 2018;9:2009.
9. Schwarz G, Karajgi B. Improvement in refractory psychosis with dronabinol: four case reports. J Clin Psychiatry. 2010;71(11):1552-1553.
10. Schwarz G, Karajgi B, McCarthy R. Synthetic delta-9-tetrahydrocannabinol (dronabinol) can improve the symptoms of schizophrenia. J Clin Psychopharmacol. 2009;29(3):255-258.
11. Meltzer HY, Arvanitis L, Bauer D, et al. Placebo-controlled evaluation of four novel compounds for the treatment of schizophrenia and schizoaffective disorder. Am J Psychiatry. 2004;161(6):975-984.
12. Boggs DL, Kelly DL, McMahon RP, et al. Rimonabant for neurocognition in schizophrenia: a 16-week double blind placebo controlled trial. Schizophr Res. 2012;134(2-3):207-210.
13. Kelly DL, Gorelick DA, Conley RR, et al. Effects of cannabinoid-1 receptor antagonist rimonabant on psychiatric symptoms in overweight people with schizophrenia: a randomized, double-blind, pilot study. J Clin Psychopharmacol. 2011;31(1):86-91.
14. Leweke FM, Mueller JK, Lange B, et al. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):604-612.
15. Halperin A. What is CBD? The ‘miracle’ cannabis compound that doesn’t get you high. The Guardian. https://www.theguardian.com/society/2018/may/28/what-is-cbd-cannabidiol-cannabis-medical-uses. Published May 28, 2018. Accessed April 3, 2019.
16. Pierre J. Coca, cola, and cannabis: psychoactive drugs as beverages. Psychology Today (blog) Psych Unseen. https://www.psychologytoday.com/us/blog/psych-unseen/201810/coca-cola-and-cannabis-psychoactive-drugs-beverages. Published October 1, 2018. Accessed April 3, 2019.
17. U.S. Food and Drug Administration. FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. FDA News Release. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm611046.htm. Published June 25, 2018. Accessed April 3, 2019.
18. Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011-2020.
19. Thiele EA, March ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
20. Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
21. Khoury JM, Neves MCLD, Rogue MAV, et al. Is there a role of cannabidiol in psychiatry? World J Biol Psychiatry. 2017:1-16.
22. Zuardi AW, Morais SL, Guimares FS, et al. Antipsychotic effect of cannabidiol. J Clin Psychiatry. 1995;56(10):485-486.
23. Zuardi AW, Hallak JEC, Dursun SM. Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol. 2006;20(5):683-686.
24. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2:e94. doi: 10.1038/tp.2012.15.
25. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225-231.
26. Boggs DL, Surti I, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacol. 2018;235(7):1923-1932.
27. ElSohly MA, Mehmedic Z, Foster S, et al. Changes in cannabis potency over the last 2 decades (1995-2014): analysis of current data in the United States. Biol Psychiatry. 2016; 79(7):613-619.
28. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491-2492.
29. Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, et al. Consistency of label claims of internet-purchased hemp oil and cannabis products as determined using IMS and LC-MS: a marketplace study. J Reg Sci. 2016;3:1-6.
30. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318(17):1708-1709.
31. Seeman P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl Psychiatry. 2016;6(10):e920. doi: 10.1038/tp.2016.195.
32. Renard J, Norris C, Rushlow W, et al. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;157-165.
33. Gururajan A, Malone DT. Does cannabidiol have a role in the treatment of schizophrenia? Schizophr Res. 2016;176(2-3):281-290.
1. Pierre JM. Cannabis, synthetic cannabinoids, and psychosis risk: what the evidence says. Current Psychiatry. 2011;10(9):49-58.
2. Radhakrishan R, Wilkinson ST, D’Souza DC. Gone to pot – a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54.
3. Pierre JM. Risks of increasingly potent cannabis: joint effects of potency and frequency. Current Psychiatry. 2016;16(2):14-20.
4. Hanna RC, Perez JM, Ghose S. Cannabis and development of dual diagnoses: a literature review. Am J Drug Alcohol Abuse. 2017;43(4):442-255.
5. Nunberg H, Kilmer B, Pacula RL, et al. An analysis of applicants presenting to a medical marijuana specialty practice in California. J Drug Policy Anal. 2011;4(1):1.
6. Wilkinson ST, Radhakrishnan, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064.
7. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: Update 2015. Subst Abus. 2016;38(3):344-366.
8. Crippa JA, Guimarães FS, Campos A, et al. Translational investigation of the therapeutic potential of cannabidiol (CBD): toward a new age. Front Immunol. 2018;9:2009.
9. Schwarz G, Karajgi B. Improvement in refractory psychosis with dronabinol: four case reports. J Clin Psychiatry. 2010;71(11):1552-1553.
10. Schwarz G, Karajgi B, McCarthy R. Synthetic delta-9-tetrahydrocannabinol (dronabinol) can improve the symptoms of schizophrenia. J Clin Psychopharmacol. 2009;29(3):255-258.
11. Meltzer HY, Arvanitis L, Bauer D, et al. Placebo-controlled evaluation of four novel compounds for the treatment of schizophrenia and schizoaffective disorder. Am J Psychiatry. 2004;161(6):975-984.
12. Boggs DL, Kelly DL, McMahon RP, et al. Rimonabant for neurocognition in schizophrenia: a 16-week double blind placebo controlled trial. Schizophr Res. 2012;134(2-3):207-210.
13. Kelly DL, Gorelick DA, Conley RR, et al. Effects of cannabinoid-1 receptor antagonist rimonabant on psychiatric symptoms in overweight people with schizophrenia: a randomized, double-blind, pilot study. J Clin Psychopharmacol. 2011;31(1):86-91.
14. Leweke FM, Mueller JK, Lange B, et al. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):604-612.
15. Halperin A. What is CBD? The ‘miracle’ cannabis compound that doesn’t get you high. The Guardian. https://www.theguardian.com/society/2018/may/28/what-is-cbd-cannabidiol-cannabis-medical-uses. Published May 28, 2018. Accessed April 3, 2019.
16. Pierre J. Coca, cola, and cannabis: psychoactive drugs as beverages. Psychology Today (blog) Psych Unseen. https://www.psychologytoday.com/us/blog/psych-unseen/201810/coca-cola-and-cannabis-psychoactive-drugs-beverages. Published October 1, 2018. Accessed April 3, 2019.
17. U.S. Food and Drug Administration. FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. FDA News Release. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm611046.htm. Published June 25, 2018. Accessed April 3, 2019.
18. Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011-2020.
19. Thiele EA, March ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
20. Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
21. Khoury JM, Neves MCLD, Rogue MAV, et al. Is there a role of cannabidiol in psychiatry? World J Biol Psychiatry. 2017:1-16.
22. Zuardi AW, Morais SL, Guimares FS, et al. Antipsychotic effect of cannabidiol. J Clin Psychiatry. 1995;56(10):485-486.
23. Zuardi AW, Hallak JEC, Dursun SM. Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol. 2006;20(5):683-686.
24. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2:e94. doi: 10.1038/tp.2012.15.
25. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225-231.
26. Boggs DL, Surti I, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacol. 2018;235(7):1923-1932.
27. ElSohly MA, Mehmedic Z, Foster S, et al. Changes in cannabis potency over the last 2 decades (1995-2014): analysis of current data in the United States. Biol Psychiatry. 2016; 79(7):613-619.
28. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491-2492.
29. Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, et al. Consistency of label claims of internet-purchased hemp oil and cannabis products as determined using IMS and LC-MS: a marketplace study. J Reg Sci. 2016;3:1-6.
30. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318(17):1708-1709.
31. Seeman P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl Psychiatry. 2016;6(10):e920. doi: 10.1038/tp.2016.195.
32. Renard J, Norris C, Rushlow W, et al. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;157-165.
33. Gururajan A, Malone DT. Does cannabidiol have a role in the treatment of schizophrenia? Schizophr Res. 2016;176(2-3):281-290.
Management of Early Pulmonary Complications After Hematopoietic Stem Cell Transplantation
Hematopoietic stem cell transplantation (HSCT) is widely used in the economically developed world to treat a variety of hematologic malignancies as well as nonmalignant diseases and solid tumors. An estimated 17,900 HSCTs were performed in 2011, and survival rates continue to increase.1 Pulmonary complications post HSCT are common, with rates ranging from 40% to 60%, and are associated with increased morbidity and mortality.2
Clinical diagnosis of pulmonary complications in the HSCT population has been aided by a previously well-defined chronology of the most common diseases.3 Historically, early pulmonary complications were defined as pulmonary complications occurring within 100 days of HSCT (corresponding to the acute graft-versus-host disease [GVHD] period). Late pulmonary complications are those that occur thereafter. This timeline, however, is now more variable given the increasing indications for HSCT, the use of reduced-intensity conditioning strategies, and varied individual immune reconstitution. This article discusses the management of early post-HSCT pulmonary complications; late post-HSCT pulmonary complications will be discussed in a separate follow-up article.
Transplant Basics
The development of pulmonary complications is affected by many factors associated with the transplant. Autologous transplantation involves the collection of a patient’s own stem cells, appropriate storage and processing, and re-implantation after induction therapy. During induction therapy, the patient undergoes high-dose chemotherapy or radiation therapy that ablates the bone marrow. The stem cells are then transfused back into the patient to repopulate the bone marrow. Allogeneic transplants involve the collection of stem cells from a donor. Donors are matched as closely as possible to the recipient’s histocompatibility antigen (HLA) haplotypes to prevent graft failure and rejection. The donor can be related or unrelated to the recipient. If there is not a possibility of a related match (from a sibling), then a national search is undertaken to look for a match through the National Marrow Donor Program. There are fewer transplant reactions and occurrences of GVHD if the major HLAs of the donor and recipient match. Table 1 reviews basic definitions pertaining to HSCT.
How the cells for transplantation are obtained is also an important factor in the rate of complications. There are 3 main sources: peripheral blood, bone marrow, and umbilical cord. Peripheral stem cell harvesting involves exposing the donor to granulocyte-colony stimulating factor (gCSF), which increases peripheral circulation of stem cells. These cells are then collected and infused into the recipient after the recipient has completed an induction regimen involving chemotherapy and/or radiation, depending on the protocol. This procedure is called peripheral blood stem cell transplant (PBSCT). Stem cells can also be directly harvested from bone marrow cells, which are collected from repeated aspiration of bone marrow from the posterior iliac crest.4 This technique is most common in children, whereas in adults peripheral blood stem cells are the most common source. Overall mortality does not differ based on the source of the stem cells. It is postulated that GVHD may be more common in patients undergoing PBSCT, but the graft failure rate may be lower.5
The third option is umbilical cord blood (UCB) as the source of stem cells. This involves the collection of umbilical cord blood that is prepared and frozen after birth. It has a smaller volume of cells, and although fewer cells are needed when using UCB, 2 separate donors may be required for a single adult recipient. The engraftment of the stem cells is slower and infections in the post-transplant period are more common. Prior reports indicate GVHD rates may be lower.4 While the use of UCB is not common in adults, the incidence has doubled over the past decade, increasing from 3% to 6%.
The conditioning regimen can influence pulmonary complications. Traditionally, an ablative transplant involves high-dose chemotherapy or radiation to eradicate the recipient’s bone marrow. This regimen can lead to many complications, especially in the immediate post-transplant period. In the past 10 years, there has been increasing interest in non-myeloablative, or reduced-intensity, conditioning transplants.6 These “mini transplants” involve smaller doses of chemotherapy or radiation, which do not totally eradicate the bone marrow; after the transplant a degree of chimerism develops where the donor and recipient stem cells coexist. The medications in the preparative regimen also should be considered because they can affect pulmonary complications after transplant. Certain chemotherapeutic agents such as carmustine, bleomycin, and many others can lead to acute and chronic presentations of pulmonary diseases such as hypersensitivity pneumonitis, pulmonary fibrosis, acute respiratory distress syndrome, and abnormal pulmonary function testing.
After the HSCT, GVHD can develop in more than 50% of allogeneic recipients.3 The incidence of GVHD has been reported to be increasing over the past 12 years.It is divided into acute GVHD (which traditionally happens in the first 100 days after transplant) and chronic GVHD (after day 100). This calendar-day–based system has been augmented based on a 2006 National Institutes of Health working group report emphasizing the importance of organ-specific features of chronic GVHD in the clinical presentation of GVHD.7 Histologic changes in chronic organ GVHD tend to include more fibrotic features, whereas in acute GVHD more inflammatory changes are seen. The NIH working group report also stressed the importance of obtaining a biopsy specimen for histopathologic review and interdisciplinary collaboration to arrive at a consensus diagnosis, and noted the limitations of using histologic changes as the sole determinant of a “gold standard” diagnosis.7 GVHD can directly predispose patients to pulmonary GVHD and indirectly predispose them to infectious complications because the mainstay of therapy for GVHD is increased immunosuppression.
Pretransplant Evaluation
Case Patient 1
A 56-year-old man is diagnosed with acute myeloid leukemia (AML) after presenting with signs and symptoms consistent with pancytopenia. He has a past medical history of chronic sinus congestion, arthritis, depression, chronic pain, and carpal tunnel surgery. He is employed as an oilfield worker and has a 40-pack-year smoking history, but he recently cut back to half a pack per day. He is being evaluated for allogeneic transplant with his brother as the donor and the planned conditioning regimen is total body irradiation (TBI), thiotepa, cyclophosphamide, and antithymocyte globulin with T-cell depletion. Routine pretransplant pulmonary function testing (PFT) reveals a restrictive pattern and he is sent for pretransplant pulmonary evaluation.
Physical exam reveals a chronically ill appearing man. He is afebrile, the respiratory rate is 16 breaths/min, blood pressure is 145/88 mm Hg, heart rate is 92 beats/min, and oxygen saturation is 95%. He is in no distress. Auscultation of the chest reveals slightly diminished breath sounds bilaterally but is clear and without wheezes, rhonchi, or rales. Heart exam shows regular rate and rhythm without murmurs, rubs, or gallops. Extremities reveal no edema or rashes. Otherwise, the remainder of the exam is normal. The patient’s PFT results are shown in Table 2.
- What aspects of this patient’s history put him at risk for pulmonary complications after transplantation?
Risk Factors for Pulmonary Complications
Predicting who is at risk for pulmonary complications is difficult. Complications are generally divided into infectious and noninfectious categories. Regardless of category, allogeneic HSCT recipients are at increased risk compared with autologous recipients, but even in autologous transplants, more than 25% of patients will develop pulmonary complications in the first year.8 Prior to transplant, patients undergo full PFT. Early on, many studies attempted to show relationships between various factors and post-transplant pulmonary complications. Factors that were implicated were forced expiratory volume in 1 second (FEV1), diffusing capacity of the lung for carbon monoxide (D
Another sometimes overlooked risk before transplantation is restrictive lung disease. One study showed a twofold increase in respiratory failure and mortality if there was pretransplant restriction based on TLC < 80%.16
An interesting study by one group in pretransplant evaluation found decreased muscle strength by maximal inspiratory muscle strength (PImax), maximal expiratory muscle strength (PEmax), dominant hand grip strength, and 6-minute walk test (6MWT) distance prior to allogeneic transplant, but did not find a relationship between these variables and mortality.17 While this study had a small sample size, these findings likely deserve continued investigation.18
- What methods are used to calculate risk for complications?
Risk Scoring Systems
Several pretransplantation risk scores have been developed. In a study that looked at more than 2500 allogeneic transplants, Parimon et al showed that risk of mortality and respiratory failure could be estimated prior to transplant using a scoring system—the Lung Function Score (LFS)—that combines the FEV1 and D
The Pretransplantation Assessment of Mortality score, initially developed in 2006, predicts mortality within the first 2 years after HSCT based on 8 clinical factors: disease risk, age at transplant, donor type, conditioning regimen, and markers of organ function (percentage of predicted FEV1, percentage of predicted D
- What other preoperative testing or interventions should be considered in this patient?
Since there is a high risk of infectious complications after transplant, the question of whether pretransplantation patients should undergo screening imaging may arise. There is no evidence that routine chest computed tomography (CT) reduces the risk of infectious complications after transplantation.26 An area that may be insufficiently addressed in the pretransplantation evaluation is smoking cessation counseling.27 Studies have shown an elevated risk of mortality in smokers.28-30 Others have found a higher incidence of respiratory failure but not an increased mortality.31 Overall, with the good rates of smoking cessation that can be accomplished, smokers should be counseled to quit before transplantation.
In summary, patients should undergo full PFTs prior to transplantation to help stratify risk for pulmonary complications and mortality and to establish a clinical baseline. The LFS (using FEV1 and D
Case Patient 1 Conclusion
The patient undergoes transplantation due to his lack of other treatment options. Evaluation prior to transplant, however, shows that he is at high risk for pulmonary complications. He has a LFS of 7 prior to transplant (using the D
Early Infectious Pulmonary Complications
Case Patient 2
A 27-year-old man with a medical history significant for AML and allogeneic HSCT presents with cough productive of a small amount of clear to white sputum, dyspnea on exertion, and fevers for 1 week. He also has mild nausea and a decrease in appetite. He underwent HSCT 2.5 months prior to admission, which was a matched unrelated bone marrow transplant with TBI and cyclophosphamide conditioning. His past medical history is significant only for exercise-induced asthma for which he takes a rescue inhaler infrequently prior to transplantation. His pretransplant PFTs showed normal spirometry with an FEV1 of 106% of predicted and D
Physical exam is notable for fever of 101.0°F, heart rate 80 beats/min, respiratory rate 16 breaths/ min, and blood pressure 142/78 mm Hg; an admission oxygen saturation is 93% on room air. Lungs show bibasilar crackles and the remainder of the exam is normal. Laboratory testing shows a white blood cell count of 2400 cells/μL, hemoglobin 7.6 g/dL, and platelet count 66 × 103/μL. Creatinine is 1.0 mg/dL. Chest radiograph shows ill-defined bilateral lower-lobe infiltrates. CT scans are shown in the Figure.
- For which infectious complications is this patient most at risk?
Pneumonia
A prospective trial in the HSCT population reported a pneumonia incidence rate of 68%, and pneumonia is more common in allogeneic HSCT with prolonged immunosuppressive therapy.32 Development of pneumonia within 100 days of transplant directly correlates with nonrelapsed mortality.33 Early detection is key, and bronchoscopy within the first 5 days of symptoms has been shown to change therapy in approximately 40% of cases but has not been shown to affect mortality.34 The clinical presentation of pneumonia in the HSCT population can be variable because of the presence of neutropenia and profound immunosuppression. Traditionally accepted diagnostic criteria of fevers, sputum production, and new infiltrates should be used with caution, and an appropriately high index of suspicion should be maintained. Progression to respiratory failure, regardless of causative organism of infection, portends a poor prognosis, with mortality rates estimated at 70% to 90%.35,36 Several transplant-specific factors may affect early infections. For instance, UCB transplants have been found to have a higher incidence of invasive aspergillosis and cytomegalovirus (CMV) infections but without higher mortality attributed to the infections.37
Bacterial Pneumonia
Bacterial pneumonia accounts for 20% to 50% of pneumonia cases in HSCT recipients.38 Gram-negative organisms, specifically Pseudomonas aeruginosa and Escherichia coli, were reported to be the most common pathologic bacteria in recent prospective trials, whereas previous retrospective trials showed that common community-acquired organisms were the most common cause of pneumonia in HSCT recipients.32,39 This underscores the importance of being aware of the clinical prevalence of microorganisms and local antibiograms, along with associated institutional susceptibility profiles. Initiation of immediate empiric broad-spectrum antibiotics is essential when bacterial pneumonia is suspected.
Viral Pneumonia
The prevalence of viral pneumonia in stem cell transplant recipients is estimated at 28%,32 with most cases being caused by community viral pathogens such as rhinovirus, respiratory syncytial virus (RSV), influenza A and B, and parainfluenza.39 The prevention, prophylaxis, and early treatment of viral pneumonias, specifically CMV infection, have decreased the mortality associated with early pneumonia after HSCT. Co-infection with bacterial organisms must be considered and has been associated with increased mortality in the intensive care unit setting.40
Supportive treatment with rhinovirus infection is sufficient as the disease is usually self-limited in immunocompromised patients. In contrast, infection with RSV in the lower respiratory tract is associated with increased mortality in prior reports, and recent studies suggest that further exploration of prophylaxis strategies is warranted.41 Treatment with ribavirin remains the backbone of therapy, but drug toxicity continues to limit its use. The addition of immunomodulators such as RSV immune globulin or palivizumab to ribavirin remains controversial, but a retrospective review suggests that early treatment may prevent progression to lower respiratory tract infection and lead to improved mortality.42 Infection with influenza A/B must be considered during influenza season. Treatment with oseltamivir may shorten the duration of disease when influenza A/B or parainfluenza are detected. Reactivation of latent herpes simplex virus during the pre-engraftment phase should also be considered. Treatment is similar to that in nonimmunocompromised hosts. When CMV pneumonia is suspected, careful history regarding compliance with prophylactic antivirals and CMV status of both the recipient and donor are key. A presumptive diagnosis can be made with the presence of appropriate clinical scenario, supportive radiographic images showing areas of ground-glass opacification or consolidation, and positive CMV polymerase chain reaction (PCR) assay. Visualization of inclusion bodies on lung biopsy tissue remains the gold standard for diagnosis. Treatment consists of CMV immunoglobulin and ganciclovir.
Fungal Pneumonia
Early fungal pneumonias have been associated with increased mortality in the HSCT population.43 Clinical suspicion should remain high and compliance with antifungal prophylaxis should be questioned thoroughly. Invasive aspergillosis (IA) remains the most common fungal infection. A bimodal distribution of onset of infection peaking on day 16 and again on day 96 has been described in the literature.44 Patients often present with classic pneumonia symptoms, but these may be accompanied by hemoptysis. Proven IA diagnosis requires visualization of fungal forms from biopsy or needle aspiration or a positive culture obtained in a sterile fashion.45 Most clinical data comes from experience with probable and possible diagnosis of IA. Bronchoalveolar lavage with testing with Aspergillus galactomannan assay has been shown to be clinically useful in establishing the clinical diagnosis in the HSCT population.46 Classic air-crescent findings on chest CT are helpful in establishing a possible diagnosis, but retrospective analysis reveals CT findings such as focal infiltrates and pulmonary nodular patterns are more common.47 First-line treatment with voriconazole has been shown to decrease short-term mortality attributable to IA but has not had an effect on long-term, all-cause mortality.48 Surgical resection is reserved for patients with refractory disease or patients presenting with massive hemoptysis.
Mucormycosis is an emerging disease with ever increasing prevalence in the HSCT population, reflecting the improved prophylaxis and treatment of IA. Initial clinical presentation is similar to IA, most commonly affecting the lung, although craniofacial involvement is classic for mucormycosis, especially in HSCT patients with diabetes.49Mucor infections can present with massive hemoptysis due to tissue invasion and disregard for tissue and fascial planes. Diagnosis of mucormycosis is associated with as much as a six-fold increase in risk for death. Diagnosis requires identification of the organism by examination or culture and biopsy is often necessary.50,51 Amphotericin B remains first-line therapy as mucormycosis is resistant to azole antifungals, with higher doses recommended for cerebral involvement.52
Candida pulmonary infections during the early HSCT period are becoming increasingly rare due to widespread use of fluconazole prophylaxis and early treatment of mucosal involvement during neutropenia. Endemic fungal infections such as blastomycosis, coccidioidomycosis, and histoplasmosis should be considered in patients inhabiting specific geographic areas or with recent travel to these areas.
- What test should be performed to evaluate for infectious causes of pneumonia?
Role of Flexible Fiberoptic Bronchoscopy
The utility of flexible fiberoptic bronchoscopy (FOB) in immune-compromised patients for the evaluation of pulmonary infiltrates is a frequently debated topic. Current studies suggest a diagnosis can be made in approximately 80% of cases in the immune-compromised population.32,53 Noninvasive testing such as urine and serum antigens, sputum cultures, Aspergillus galactomannan assays, viral nasal swabs, and PCR studies often lead to a diagnosis in appropriate clinical scenarios. Conservative management would dictate the use of noninvasive testing whenever possible, and randomized controlled trials have shown noninvasive testing to be noninferior to FOB in preventing need for mechanical ventilation, with no difference in overall mortality.54 FOB has been shown to be most useful in establishing a diagnosis when an infectious etiology is suspected.55 In multivariate analysis, a delay in the identification of the etiology of pulmonary infiltrate was associated with increased mortality.56 Additionally, early FOB was found to be superior to late FOB in revealing a diagnosis. 32,57 Despite its ability to detect the cause of pulmonary disease, direct antibiotic therapy, and possibly change therapy, FOB with diagnostic maneuvers has not been shown to affect mortality.58 In a large case series, FOB with bronchoalveolar lavage (BAL) revealed a diagnosis in approximately 30% to 50% of cases. The addition of transbronchial biopsy did not improve diagnostic utility.58 More recent studies have confirmed that the addition of transbronchial biopsy does not add to diagnostic yield and is associated with increased adverse events.59 The appropriate use of advanced techniques such as endobronchial ultrasound–guided transbronchial needle aspirations, endobronchial biopsy, and CT-guided navigational bronchoscopy has not been established and should be considered on a case-by-case basis. In summary, routine early BAL is the diagnostic test of choice, especially when infectious pulmonary complications are suspected.
Contraindications for FOB in this population mirror those in the general population. These include acute severe hypoxemic respiratory failure, myocardial ischemia or acute coronary syndrome within 2 weeks of procedure, severe thrombocytopenia, and inability to provide or obtain informed consent from patient or health care power of attorney. Coagulopathy and thrombocytopenia are common comorbid conditions in the HSCT population. A platelet count of < 20 × 103/µL has generally been used as a cut-off for routine FOB with BAL.60 Risks of the procedures should be discussed clearly with the patient, but simple FOB for airway evaluation and BAL is generally well tolerated even under these conditions.
Early Nonifectious Pulmonary Complications
Case Patient 2 Continued
Bronchoscopy with BAL performed the day after admission is unremarkable and stains and cultures are negative for viral, bacterial, and fungal organisms. The patient is initially started on broad-spectrum antibiotics, but his oxygenation continues to worsen to the point that he is placed on noninvasive positive pressure ventilation. He is started empirically on amphotericin B and eventually is intubated. VATS lung biopsy is ultimately performed and pathology is consistent with diffuse alveolar damage.
- Based on these biopsy findings, what is the diagnosis?
Based on the pathology consistent with diffuse alveolar damage, a diagnosis of idiopathic pneumonia syndrome (IPS) is made.
- What noninfectious pulmonary complications occur in the early post-transplant period?
The overall incidence of noninfectious pulmonary complications after HSCT is generally estimated at 20% to 30%.32 Acute pulmonary edema is a common very early noninfectious pulmonary complication and clinically the most straightforward to treat. Three distinct clinical syndromes—peri-engraftment respiratory distress syndrome (PERDS), diffuse alveolar hemorrhage (DAH), and IPS—comprise the remainder of the pertinent early noninfectious complications. Clinical presentation differs based upon the disease entity. Recent studies have evaluated the role of angiotensin-converting enzyme polymorphisms as a predictive marker for risk of developing early noninfectious pulmonary complications.61
Peri-Engraftment Respiratory Distress Syndrome
PERDS is a clinical syndrome comprising the cardinal features of erythematous rash and fever along with noncardiogenic pulmonary infiltrates and hypoxemia that occur in the peri-engraftment period, defined as recovery of absolute neutrophil count to > 500/μL on 2 consecutive days.62 PERDS occurs in the autologous HSCT population and may be a clinical correlate to early GVHD in the allogeneic HSCT population. It is hypothesized that the pathophysiology underlying PERDS is an autoimmune-related capillary leak caused by pro-inflammatory cytokine release.63 Treatment remains anecdotal and currently consists of supportive care and high-dose corticosteroids. Some have favored limiting the use of gCSF given its role in stimulating rapid white blood cell recovery.33 Prognosis is favorable, but progression to fulminant respiratory failure requiring mechanical ventilation portends a poor prognosis.
Diffuse Alveolar Hemorrhage
DAH is clinical syndrome consisting of diffuse alveolar infiltrates on pulmonary imaging combined with progressively bloodier return per aliquot during BAL in 3 different subsegments or more than 20% hemosiderin-laden macrophages on BAL fluid evaluation. Classically, DAH is defined in the absence of pulmonary infection or cardiac dysfunction. The pathophysiology is thought to be related to inflammation of pulmonary vasculature within the alveolar walls leading to alveolitis. Although no prospective trials exist, early use of high-dose corticosteroid therapy is thought to improve outcomes;64,65 a recent study, however, showed low-dose steroids may be associated with the lowest mortality.66 Mortality is directly linked to the presence of superimposed infection, need for mechanical ventilation, late onset, and development of multiorgan failure.67
Idiopathic Pneumonia Syndrome
IPS is a complex clinical syndrome whose pathology is felt to stem from a variety of possible lung insults such as direct myeloablative drug toxicity, occult pulmonary infection, or cytokine-driven inflammation. The ATS published an article further subcategorizing IPS as different clinical entities based upon whether the primary insult involves the vascular endothelium, interstitial tissue, and airway tissue, truly idiopathic, or unclassified.68 In clinical practice, IPS is defined as widespread alveolar injury in the absence of evidence of renal failure, heart failure, and excessive fluid resuscitation. In addition, negative testing for a variety of bacterial, viral, and fungal causes is also necessary.69 Clinical syndromes included within the IPS definition are ARDS, acute interstitial pneumonia, DAH, cryptogenic organizing pneumonia, and BOS.70 Risk factors for developing IPS include TBI, older age of recipient, acute GVHD, and underlying diagnosis of AML or myelodysplastic syndrome.12 In addition, it has been shown that risk for developing IPS is lower in patients undergoing allogeneic HSCT who receive non-myeloablative conditioning regimens.71 The pathologic finding in IPS is diffuse alveolar damage. A 2006 study in which investigators reviewed BAL samples from patients with IPS found that 3% of the patients had PCR evidence of human metapneumovirus infection, and a study in 2015 found PCR evidence of infection in 53% of BAL samples from patients diagnosed with IPS.72,73 This fuels the debate on whether IPS is truly an infection-driven process where the source of infection, pulmonary or otherwise, simply escapes detection. Various surfactant proteins, which play a role in decreasing surface tension within the alveolar interface and function as mediators within the innate immunity of the lung, have been studied in regard to development of IPS. Small retrospective studies have shown a trend toward lower pre-transplant serum protein surfactant D and the development of IPS.74
The diagnosis of IPS does not require pathologic diagnosis in most circumstances. The correct clinical findings in association with a negative infectious workup lead to a presumptive diagnosis of IPS. The extent of the infectious workup that must be completed to adequately rule out infection is often a difficult clinical question. Recent recommendations include BAL fluid evaluation for routine bacterial cultures, appropriate viral culture, and consideration of PCR testing to evaluate for Mycoplasma, Chlamydia, and Aspergillus antigens.75 Transbronchial biopsy continues to appear in recommendations, but is not routinely performed and should be completed as the patient’s clinical status permits.8,68 Table 3 reviews basic features of early noninfectious pulmonary complications.
Treatment of IPS centers around moderate to high doses of corticosteroids. Based on IPS experimental modes, tumor necrosis factor (TNF)-α has been implicated as an important mediator. Unfortunately, several studies evaluating etanercept have produced conflicting results, and this agent’s clinical effects on morbidity and mortality remain in question.76
- What treatment should be offered to the patient with diffuse alveolar damage on biopsy?
Treatment consists of supportive care and empiric broad-spectrum antibiotics with consideration of high-dose corticosteroids. Based upon early studies in murine models implicating TNF, pilot studies were performed evaluating etanercept as a possible safe and effective addition to high-dose systemic corticosteroids.77 Although these results were promising, data from a truncated randomized control clinical trial failed to show improvement in patient response in the adult population.76 More recent data from the same author suggests that pediatric populations with IPS are, however, responsive to etanercept and high-dose corticosteroid therapy.78 When IPS develops as a late complication, treatment with high-dose corticosteroids (2 mg/kg/day) and etanercept (0.4 mg/kg twice weekly) has been shown to improve 2-year survival.79
Case Patient 2 Conclusion
The patient is started on steroids and makes a speedy recovery. He is successfully extubated 5 days later.
Conclusion
Careful pretransplant evaluation, including a full set of pulmonary function tests, can help predict a patient’s risk for pulmonary complications after transplant, allowing risk factor modification strategies to be implemented prior to transplant, including smoking cessation. It also helps identify patients at high risk for complications who will require closer monitoring after transplantation. Early posttransplant complications include infectious and noninfectious entities. Bacterial, viral, and fungal pneumonias are in the differential of infectious pneumonia, and bronchoscopy can be helpful in establishing a diagnosis. A common, important noninfectious cause of early pulmonary complications is IPS, which is treated with steroids and sometimes anti-TNF therapy.
1. Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010;303:1617–24.
2. Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004;170:22–48.
4. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med 2006;354:1813–26.
5. Anasetti C, Logan BR, Lee SJ, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 2012;367:1487–96.
6. Giralt S, Ballen K, Rizzo D, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009;15:367–9.
7. Shulman HM, Kleiner D, Lee SJ, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant 2006;12:31–47.
8. Afessa B, Abdulai RM, Kremers WK, et al. Risk factors and outcome of pulmonary complications after autologous hematopoietic stem cell transplant. Chest 2012;141:442–50.
9. Bolwell BJ. Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003;32:853–61.
10. Carlson K, Backlund L, Smedmyr B, et al. Pulmonary function and complications subsequent to autologous bone marrow transplantation. Bone Marrow Transplant 1994;14:805–11.
11. Clark JG, Schwartz DA, Flournoy N, et al. Risk factors for airflow obstruction in recipients of bone marrow transplants. Ann Intern Med 1987;107:648–56.
12. Crawford SW, Fisher L. Predictive value of pulmonary function tests before marrow transplantation. Chest 1992; 101:1257–64.
13. Ghalie R, Szidon JP, Thompson L, et al. Evaluation of pulmonary complications after bone marrow transplantation: the role of pretransplant pulmonary function tests. Bone Marrow Transplant 1992;10:359–65.
14. Ho VT, Weller E, Lee SJ, et al. Prognostic factors for early severe pulmonary complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001;7:223–9.
15. Horak DA, Schmidt GM, Zaia JA, et al. Pretransplant pulmonary function predicts cytomegalovirus-associated interstitial pneumonia following bone marrow transplantation. Chest 1992;102:1484–90.
16. Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, et al. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transplant 2010;16:199–206.
17. White AC, Terrin N, Miller KB, Ryan HF. Impaired respiratory and skeletal muscle strength in patients prior to hematopoietic stem-cell transplantation. Chest 2005;128145–52.
18. Afessa B. Pretransplant pulmonary evaluation of the blood and marrow transplant recipient. Chest 2005;128:8–10.
19. Parimon T, Madtes DK, Au DH, et al. Pretransplant lung function, respiratory failure, and mortality after stem cell transplantation. Am J Respir Crit Care Med 2005;172:384–90.
20. Pavletic SZ, Martin P, Lee SJ, et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006;12:252–66.
21. Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med 2006;144:407–14.
22. Au BK, Gooley TA, Armand P, et al. Reevaluation of the pretransplant assessment of mortality score after allogeneic hematopoietic transplantation. Biol Blood Marrow Transplant 2015;21:848–54.
23. Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005;106:2912–9.
24. Chien JW, Sullivan KM. Carbon monoxide diffusion capacity: how low can you go for hematopoietic cell transplantation eligibility? Biol Blood Marrow Transplant 2009;15: 447–53.
25. Coffey DG, Pollyea DA, Myint H, et al. Adjusting DLCO for Hb and its effects on the Hematopoietic Cell Transplantation-specific Comorbidity Index. Bone Marrow Transplant 2013;48:1253–6.
26. Kasow KA, Krueger J, Srivastava DK, et al. Clinical utility of computed tomography screening of chest, abdomen, and sinuses before hematopoietic stem cell transplantation: the St. Jude experience. Biol Blood Marrow Transplant 2009;15:490–5.
27. Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45: 1259–68.
28. Savani BN, Montero A, Wu C, et al. Prediction and prevention of transplant-related mortality from pulmonary causes after total body irradiation and allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:223–30.
29. Ehlers SL, Gastineau DA, Patten CA, et al. The impact of smoking on outcomes among patients undergoing hematopoietic SCT for the treatment of acute leukemia. Bone Marrow Transplant 2011;46:285–90.
30. Marks DI, Ballen K, Logan BR, et al. The effect of smoking on allogeneic transplant outcomes. Biol Blood Marrow Transplant 2009;15:1277–87.
31. Tran BT, Halperin A, Chien JW. Cigarette smoking and outcomes after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2011;17:1004–11.
32. Lucena CM, Torres A, Rovira M, et al. Pulmonary complications in hematopoietic SCT: a prospective study. Bone Marrow Transplant 2014;49:1293–9.
33. Chi AK, Soubani AO, White AC, Miller KB. An update on pulmonary complications of hematopoietic stem cell transplantation. Chest 2013;144:1913–22.
34. Dunagan DP, Baker AM, Hurd DD, Haponik EF. Bronchoscopic evaluation of pulmonary infiltrates following bone marrow transplantation. Chest 1997;111:135–41.
35. Naeem N, Reed MD, Creger RJ, et al. Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006;37:119–33.
36. Afessa B, Tefferi A, Hoagland HC, et al. Outcome of recipients of bone marrow transplants who require intensive care unit support. Mayo Clin Proc 1992;67:117–22.
37. Parody R, Martino R, de la Camara R, et al. Fungal and viral infections after allogeneic hematopoietic transplantation from unrelated donors in adults: improving outcomes over time. Bone Marrow Transplant 2015;50:274–81.
38. Orasch C, Weisser M, Mertz D, et al. Comparison of infectious complications during induction/consolidation chemotherapy versus allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:521–6.
39. Aguilar-Guisado M, Jimenez-Jambrina M, Espigado I, et al. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 2011;25:E629–38.
40. Palacios G, Hornig M, Cisterna D, et al. Streptococcus pneumoniae coinfection is correlated with the severity of H1N1 pandemic influenza. PLoS One 2009;4:e8540.
41. Hynicka LM, Ensor CR. Prophylaxis and treatment of respiratory syncytial virus in adult immunocompromised patients. Ann Pharmacother 2012;46:558–66.
42. Shah JN, Chemaly RF. Management of RSV infections in adult recipients of hematopoietic stem cell transplantation. Blood 2011;2755–63.
43. Marr KA, Bowden RA. Fungal infections in patients undergoing blood and marrow transplantation. Transpl Infect Dis 1999;1:237–46.
44. Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J Infect Dis 1997;175:1459–66.
45. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7–14.
46. Fisher CE, Stevens AM, Leisenring W, et al. Independent contribution of bronchoalveolar lavage and serum galactomannan in the diagnosis of invasive pulmonary aspergillosis. Transpl Infect Dis 2014;16:505–10.
47. Kojima R, Tateishi U, Kami M, et al. Chest computed tomography of late invasive aspergillosis after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:506–11.
48. Salmeron G, Porcher R, Bergeron A, et al. Persistent poor long-term prognosis of allogeneic hematopoietic stem cell transplant recipients surviving invasive aspergillosis. Haematologica 2012;97:1357–63.
49. McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982;92(10 Pt 1):1140.
50. Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012;54 Suppl 1:S55–60.
51. Klingspor L, Saaedi B, Ljungman P, Szakos A. Epidemiology and outcomes of patients with invasive mould infections: a retrospective observational study from a single centre (2005-2009). Mycoses 2015;58:470–7.
52. Danion F, Aguilar C, Catherinot E, et al. Mucormycosis: new developments in a persistently devastating infection. Semin Respir Crit Care Med 2015;36:692–70.
53. Rano A, Agusti C, Jimenez P, et al. Pulmonary infiltrates in non-HIV immunocompromised patients: a diagnostic approach using non-invasive and bronchoscopic procedures. Thorax 2001;56:379–87.
54. Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med 2008;36:100–7.
55. Jain P, Sandur S, Meli Y, et al. Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 2004;125:712–22.
56. Rano A, Agusti C, Benito N, et al. Prognostic factors of non-HIV immunocompromised patients with pulmonary infiltrates. Chest 2002;122:253–61.
57. Shannon VR, Andersson BS, Lei X, et al. Utility of early versus late fiberoptic bronchoscopy in the evaluation of new pulmonary infiltrates following hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:647–55.
58. Patel NR, Lee PS, Kim JH, et al. The influence of diagnostic bronchoscopy on clinical outcomes comparing adult autologous and allogeneic bone marrow transplant patients. Chest 2005;127:1388–96.
59. Chellapandian D, Lehrnbecher T, Phillips B, et al. Bronchoalveolar lavage and lung biopsy in patients with cancer and hematopoietic stem-cell transplantation recipients: a systematic review and meta-analysis. J Clin Oncol 2015;33:501–9.
60. Carr IM, Koegelenberg CF, von Groote-Bidlingmaier F, et al. Blood loss during flexible bronchoscopy: a prospective observational study. Respiration 2012;84:312–8.
61. Miyamoto M, Onizuka M, Machida S, et al. ACE deletion polymorphism is associated with a high risk of non-infectious pulmonary complications after stem cell transplantation. Int J Hematol 2014;99:175–83.
62. Capizzi SA, Kumar S, Huneke NE, et al. Peri-engraftment respiratory distress syndrome during autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1299–303.
63. Spitzer TR. Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:893–8.
64. Wanko SO, Broadwater G, Folz RJ, Chao NJ. Diffuse alveolar hemorrhage: retrospective review of clinical outcome in allogeneic transplant recipients treated with aminocaproic acid. Biol Blood Marrow Transplant 2006;12:949–53.
65. Metcalf JP, Rennard SI, Reed EC, et al. Corticosteroids as adjunctive therapy for diffuse alveolar hemorrhage associated with bone marrow transplantation. University of Nebraska Medical Center Bone Marrow Transplant Group. Am J Med 1994;96:327–34.
66. Rathi NK, Tanner AR, Dinh A, et al. Low-, medium- and high-dose steroids with or without aminocaproic acid in adult hematopoietic SCT patients with diffuse alveolar hemorrhage. Bone Marrow Transplant 2015;50:420–6.
67. Afessa B, Tefferi A, Litzow MR, Peters SG. Outcome of diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med 2002;166:1364–8.
68. Panoskaltsis-Mortari A, Griese M, Madtes DK, et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am J Respir Crit Care Med 2011;183:1262–79.
69. Clark JG, Hansen JA, Hertz MI, Pet al. NHLBI workshop summary. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Resp Dis 1993;147:1601–6.
70. Vande Vusse LK, Madtes DK. Early onset noninfectious pulmonary syndromes after hematopoietic cell transplantation. Clin Chest Med 2017;38:233–48.
71. Fukuda T, Hackman RC, Guthrie KA, et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003;102:2777–85.
72. Englund JA, Boeckh M, Kuypers J, et al. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med 2006;144:344–9.
73. Seo S, Renaud C, Kuypers JM, et al. Idiopathic pneumonia syndrome after hematopoietic cell transplantation: evidence of occult infectious etiologies. Blood 2015;125:3789–97.
74. Nakane T, Nakamae H, Kamoi H, et al. Prognostic value of serum surfactant protein D level prior to transplant for the development of bronchiolitis obliterans syndrome and idiopathic pneumonia syndrome following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;42:43–9.
75. Gilbert CR, Lerner A, Baram M, Awsare BK. Utility of flexible bronchoscopy in the evaluation of pulmonary infiltrates in the hematopoietic stem cell transplant population—a single center fourteen year experience. Arch Bronconeumol 2013;49:189–95.
76. Yanik GA, Horowitz MM, Weisdorf DJ, et al. Randomized, double-blind, placebo-controlled trial of soluble tumor necrosis factor receptor: enbrel (etanercept) for the treatment of idiopathic pneumonia syndrome after allogeneic stem cell transplantation: blood and marrow transplant clinical trials network protocol. Biol Blood Marrow Transplant 2014;20:858–64.
77. Levine JE, Paczesny S, Mineishi S, et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008;111:2470–5.
78. Yanik GA, Grupp SA, Pulsipher MA, et al. TNF-receptor inhibitor therapy for the treatment of children with idiopathic pneumonia syndrome. A joint Pediatric Blood and Marrow Transplant Consortium and Children’s Oncology Group Study (ASCT0521). Biol Blood Marrow Transplant 2015;21:67–73.
79. Thompson J, Yin Z, D’Souza A, et al. Etanercept and corticosteroid therapy for the treatment of late-onset idiopathic pneumonia syndrome. Biol Blood Marrow Transplant J 2017; 23:1955–60.
Hematopoietic stem cell transplantation (HSCT) is widely used in the economically developed world to treat a variety of hematologic malignancies as well as nonmalignant diseases and solid tumors. An estimated 17,900 HSCTs were performed in 2011, and survival rates continue to increase.1 Pulmonary complications post HSCT are common, with rates ranging from 40% to 60%, and are associated with increased morbidity and mortality.2
Clinical diagnosis of pulmonary complications in the HSCT population has been aided by a previously well-defined chronology of the most common diseases.3 Historically, early pulmonary complications were defined as pulmonary complications occurring within 100 days of HSCT (corresponding to the acute graft-versus-host disease [GVHD] period). Late pulmonary complications are those that occur thereafter. This timeline, however, is now more variable given the increasing indications for HSCT, the use of reduced-intensity conditioning strategies, and varied individual immune reconstitution. This article discusses the management of early post-HSCT pulmonary complications; late post-HSCT pulmonary complications will be discussed in a separate follow-up article.
Transplant Basics
The development of pulmonary complications is affected by many factors associated with the transplant. Autologous transplantation involves the collection of a patient’s own stem cells, appropriate storage and processing, and re-implantation after induction therapy. During induction therapy, the patient undergoes high-dose chemotherapy or radiation therapy that ablates the bone marrow. The stem cells are then transfused back into the patient to repopulate the bone marrow. Allogeneic transplants involve the collection of stem cells from a donor. Donors are matched as closely as possible to the recipient’s histocompatibility antigen (HLA) haplotypes to prevent graft failure and rejection. The donor can be related or unrelated to the recipient. If there is not a possibility of a related match (from a sibling), then a national search is undertaken to look for a match through the National Marrow Donor Program. There are fewer transplant reactions and occurrences of GVHD if the major HLAs of the donor and recipient match. Table 1 reviews basic definitions pertaining to HSCT.
How the cells for transplantation are obtained is also an important factor in the rate of complications. There are 3 main sources: peripheral blood, bone marrow, and umbilical cord. Peripheral stem cell harvesting involves exposing the donor to granulocyte-colony stimulating factor (gCSF), which increases peripheral circulation of stem cells. These cells are then collected and infused into the recipient after the recipient has completed an induction regimen involving chemotherapy and/or radiation, depending on the protocol. This procedure is called peripheral blood stem cell transplant (PBSCT). Stem cells can also be directly harvested from bone marrow cells, which are collected from repeated aspiration of bone marrow from the posterior iliac crest.4 This technique is most common in children, whereas in adults peripheral blood stem cells are the most common source. Overall mortality does not differ based on the source of the stem cells. It is postulated that GVHD may be more common in patients undergoing PBSCT, but the graft failure rate may be lower.5
The third option is umbilical cord blood (UCB) as the source of stem cells. This involves the collection of umbilical cord blood that is prepared and frozen after birth. It has a smaller volume of cells, and although fewer cells are needed when using UCB, 2 separate donors may be required for a single adult recipient. The engraftment of the stem cells is slower and infections in the post-transplant period are more common. Prior reports indicate GVHD rates may be lower.4 While the use of UCB is not common in adults, the incidence has doubled over the past decade, increasing from 3% to 6%.
The conditioning regimen can influence pulmonary complications. Traditionally, an ablative transplant involves high-dose chemotherapy or radiation to eradicate the recipient’s bone marrow. This regimen can lead to many complications, especially in the immediate post-transplant period. In the past 10 years, there has been increasing interest in non-myeloablative, or reduced-intensity, conditioning transplants.6 These “mini transplants” involve smaller doses of chemotherapy or radiation, which do not totally eradicate the bone marrow; after the transplant a degree of chimerism develops where the donor and recipient stem cells coexist. The medications in the preparative regimen also should be considered because they can affect pulmonary complications after transplant. Certain chemotherapeutic agents such as carmustine, bleomycin, and many others can lead to acute and chronic presentations of pulmonary diseases such as hypersensitivity pneumonitis, pulmonary fibrosis, acute respiratory distress syndrome, and abnormal pulmonary function testing.
After the HSCT, GVHD can develop in more than 50% of allogeneic recipients.3 The incidence of GVHD has been reported to be increasing over the past 12 years.It is divided into acute GVHD (which traditionally happens in the first 100 days after transplant) and chronic GVHD (after day 100). This calendar-day–based system has been augmented based on a 2006 National Institutes of Health working group report emphasizing the importance of organ-specific features of chronic GVHD in the clinical presentation of GVHD.7 Histologic changes in chronic organ GVHD tend to include more fibrotic features, whereas in acute GVHD more inflammatory changes are seen. The NIH working group report also stressed the importance of obtaining a biopsy specimen for histopathologic review and interdisciplinary collaboration to arrive at a consensus diagnosis, and noted the limitations of using histologic changes as the sole determinant of a “gold standard” diagnosis.7 GVHD can directly predispose patients to pulmonary GVHD and indirectly predispose them to infectious complications because the mainstay of therapy for GVHD is increased immunosuppression.
Pretransplant Evaluation
Case Patient 1
A 56-year-old man is diagnosed with acute myeloid leukemia (AML) after presenting with signs and symptoms consistent with pancytopenia. He has a past medical history of chronic sinus congestion, arthritis, depression, chronic pain, and carpal tunnel surgery. He is employed as an oilfield worker and has a 40-pack-year smoking history, but he recently cut back to half a pack per day. He is being evaluated for allogeneic transplant with his brother as the donor and the planned conditioning regimen is total body irradiation (TBI), thiotepa, cyclophosphamide, and antithymocyte globulin with T-cell depletion. Routine pretransplant pulmonary function testing (PFT) reveals a restrictive pattern and he is sent for pretransplant pulmonary evaluation.
Physical exam reveals a chronically ill appearing man. He is afebrile, the respiratory rate is 16 breaths/min, blood pressure is 145/88 mm Hg, heart rate is 92 beats/min, and oxygen saturation is 95%. He is in no distress. Auscultation of the chest reveals slightly diminished breath sounds bilaterally but is clear and without wheezes, rhonchi, or rales. Heart exam shows regular rate and rhythm without murmurs, rubs, or gallops. Extremities reveal no edema or rashes. Otherwise, the remainder of the exam is normal. The patient’s PFT results are shown in Table 2.
- What aspects of this patient’s history put him at risk for pulmonary complications after transplantation?
Risk Factors for Pulmonary Complications
Predicting who is at risk for pulmonary complications is difficult. Complications are generally divided into infectious and noninfectious categories. Regardless of category, allogeneic HSCT recipients are at increased risk compared with autologous recipients, but even in autologous transplants, more than 25% of patients will develop pulmonary complications in the first year.8 Prior to transplant, patients undergo full PFT. Early on, many studies attempted to show relationships between various factors and post-transplant pulmonary complications. Factors that were implicated were forced expiratory volume in 1 second (FEV1), diffusing capacity of the lung for carbon monoxide (D
Another sometimes overlooked risk before transplantation is restrictive lung disease. One study showed a twofold increase in respiratory failure and mortality if there was pretransplant restriction based on TLC < 80%.16
An interesting study by one group in pretransplant evaluation found decreased muscle strength by maximal inspiratory muscle strength (PImax), maximal expiratory muscle strength (PEmax), dominant hand grip strength, and 6-minute walk test (6MWT) distance prior to allogeneic transplant, but did not find a relationship between these variables and mortality.17 While this study had a small sample size, these findings likely deserve continued investigation.18
- What methods are used to calculate risk for complications?
Risk Scoring Systems
Several pretransplantation risk scores have been developed. In a study that looked at more than 2500 allogeneic transplants, Parimon et al showed that risk of mortality and respiratory failure could be estimated prior to transplant using a scoring system—the Lung Function Score (LFS)—that combines the FEV1 and D
The Pretransplantation Assessment of Mortality score, initially developed in 2006, predicts mortality within the first 2 years after HSCT based on 8 clinical factors: disease risk, age at transplant, donor type, conditioning regimen, and markers of organ function (percentage of predicted FEV1, percentage of predicted D
- What other preoperative testing or interventions should be considered in this patient?
Since there is a high risk of infectious complications after transplant, the question of whether pretransplantation patients should undergo screening imaging may arise. There is no evidence that routine chest computed tomography (CT) reduces the risk of infectious complications after transplantation.26 An area that may be insufficiently addressed in the pretransplantation evaluation is smoking cessation counseling.27 Studies have shown an elevated risk of mortality in smokers.28-30 Others have found a higher incidence of respiratory failure but not an increased mortality.31 Overall, with the good rates of smoking cessation that can be accomplished, smokers should be counseled to quit before transplantation.
In summary, patients should undergo full PFTs prior to transplantation to help stratify risk for pulmonary complications and mortality and to establish a clinical baseline. The LFS (using FEV1 and D
Case Patient 1 Conclusion
The patient undergoes transplantation due to his lack of other treatment options. Evaluation prior to transplant, however, shows that he is at high risk for pulmonary complications. He has a LFS of 7 prior to transplant (using the D
Early Infectious Pulmonary Complications
Case Patient 2
A 27-year-old man with a medical history significant for AML and allogeneic HSCT presents with cough productive of a small amount of clear to white sputum, dyspnea on exertion, and fevers for 1 week. He also has mild nausea and a decrease in appetite. He underwent HSCT 2.5 months prior to admission, which was a matched unrelated bone marrow transplant with TBI and cyclophosphamide conditioning. His past medical history is significant only for exercise-induced asthma for which he takes a rescue inhaler infrequently prior to transplantation. His pretransplant PFTs showed normal spirometry with an FEV1 of 106% of predicted and D
Physical exam is notable for fever of 101.0°F, heart rate 80 beats/min, respiratory rate 16 breaths/ min, and blood pressure 142/78 mm Hg; an admission oxygen saturation is 93% on room air. Lungs show bibasilar crackles and the remainder of the exam is normal. Laboratory testing shows a white blood cell count of 2400 cells/μL, hemoglobin 7.6 g/dL, and platelet count 66 × 103/μL. Creatinine is 1.0 mg/dL. Chest radiograph shows ill-defined bilateral lower-lobe infiltrates. CT scans are shown in the Figure.
- For which infectious complications is this patient most at risk?
Pneumonia
A prospective trial in the HSCT population reported a pneumonia incidence rate of 68%, and pneumonia is more common in allogeneic HSCT with prolonged immunosuppressive therapy.32 Development of pneumonia within 100 days of transplant directly correlates with nonrelapsed mortality.33 Early detection is key, and bronchoscopy within the first 5 days of symptoms has been shown to change therapy in approximately 40% of cases but has not been shown to affect mortality.34 The clinical presentation of pneumonia in the HSCT population can be variable because of the presence of neutropenia and profound immunosuppression. Traditionally accepted diagnostic criteria of fevers, sputum production, and new infiltrates should be used with caution, and an appropriately high index of suspicion should be maintained. Progression to respiratory failure, regardless of causative organism of infection, portends a poor prognosis, with mortality rates estimated at 70% to 90%.35,36 Several transplant-specific factors may affect early infections. For instance, UCB transplants have been found to have a higher incidence of invasive aspergillosis and cytomegalovirus (CMV) infections but without higher mortality attributed to the infections.37
Bacterial Pneumonia
Bacterial pneumonia accounts for 20% to 50% of pneumonia cases in HSCT recipients.38 Gram-negative organisms, specifically Pseudomonas aeruginosa and Escherichia coli, were reported to be the most common pathologic bacteria in recent prospective trials, whereas previous retrospective trials showed that common community-acquired organisms were the most common cause of pneumonia in HSCT recipients.32,39 This underscores the importance of being aware of the clinical prevalence of microorganisms and local antibiograms, along with associated institutional susceptibility profiles. Initiation of immediate empiric broad-spectrum antibiotics is essential when bacterial pneumonia is suspected.
Viral Pneumonia
The prevalence of viral pneumonia in stem cell transplant recipients is estimated at 28%,32 with most cases being caused by community viral pathogens such as rhinovirus, respiratory syncytial virus (RSV), influenza A and B, and parainfluenza.39 The prevention, prophylaxis, and early treatment of viral pneumonias, specifically CMV infection, have decreased the mortality associated with early pneumonia after HSCT. Co-infection with bacterial organisms must be considered and has been associated with increased mortality in the intensive care unit setting.40
Supportive treatment with rhinovirus infection is sufficient as the disease is usually self-limited in immunocompromised patients. In contrast, infection with RSV in the lower respiratory tract is associated with increased mortality in prior reports, and recent studies suggest that further exploration of prophylaxis strategies is warranted.41 Treatment with ribavirin remains the backbone of therapy, but drug toxicity continues to limit its use. The addition of immunomodulators such as RSV immune globulin or palivizumab to ribavirin remains controversial, but a retrospective review suggests that early treatment may prevent progression to lower respiratory tract infection and lead to improved mortality.42 Infection with influenza A/B must be considered during influenza season. Treatment with oseltamivir may shorten the duration of disease when influenza A/B or parainfluenza are detected. Reactivation of latent herpes simplex virus during the pre-engraftment phase should also be considered. Treatment is similar to that in nonimmunocompromised hosts. When CMV pneumonia is suspected, careful history regarding compliance with prophylactic antivirals and CMV status of both the recipient and donor are key. A presumptive diagnosis can be made with the presence of appropriate clinical scenario, supportive radiographic images showing areas of ground-glass opacification or consolidation, and positive CMV polymerase chain reaction (PCR) assay. Visualization of inclusion bodies on lung biopsy tissue remains the gold standard for diagnosis. Treatment consists of CMV immunoglobulin and ganciclovir.
Fungal Pneumonia
Early fungal pneumonias have been associated with increased mortality in the HSCT population.43 Clinical suspicion should remain high and compliance with antifungal prophylaxis should be questioned thoroughly. Invasive aspergillosis (IA) remains the most common fungal infection. A bimodal distribution of onset of infection peaking on day 16 and again on day 96 has been described in the literature.44 Patients often present with classic pneumonia symptoms, but these may be accompanied by hemoptysis. Proven IA diagnosis requires visualization of fungal forms from biopsy or needle aspiration or a positive culture obtained in a sterile fashion.45 Most clinical data comes from experience with probable and possible diagnosis of IA. Bronchoalveolar lavage with testing with Aspergillus galactomannan assay has been shown to be clinically useful in establishing the clinical diagnosis in the HSCT population.46 Classic air-crescent findings on chest CT are helpful in establishing a possible diagnosis, but retrospective analysis reveals CT findings such as focal infiltrates and pulmonary nodular patterns are more common.47 First-line treatment with voriconazole has been shown to decrease short-term mortality attributable to IA but has not had an effect on long-term, all-cause mortality.48 Surgical resection is reserved for patients with refractory disease or patients presenting with massive hemoptysis.
Mucormycosis is an emerging disease with ever increasing prevalence in the HSCT population, reflecting the improved prophylaxis and treatment of IA. Initial clinical presentation is similar to IA, most commonly affecting the lung, although craniofacial involvement is classic for mucormycosis, especially in HSCT patients with diabetes.49Mucor infections can present with massive hemoptysis due to tissue invasion and disregard for tissue and fascial planes. Diagnosis of mucormycosis is associated with as much as a six-fold increase in risk for death. Diagnosis requires identification of the organism by examination or culture and biopsy is often necessary.50,51 Amphotericin B remains first-line therapy as mucormycosis is resistant to azole antifungals, with higher doses recommended for cerebral involvement.52
Candida pulmonary infections during the early HSCT period are becoming increasingly rare due to widespread use of fluconazole prophylaxis and early treatment of mucosal involvement during neutropenia. Endemic fungal infections such as blastomycosis, coccidioidomycosis, and histoplasmosis should be considered in patients inhabiting specific geographic areas or with recent travel to these areas.
- What test should be performed to evaluate for infectious causes of pneumonia?
Role of Flexible Fiberoptic Bronchoscopy
The utility of flexible fiberoptic bronchoscopy (FOB) in immune-compromised patients for the evaluation of pulmonary infiltrates is a frequently debated topic. Current studies suggest a diagnosis can be made in approximately 80% of cases in the immune-compromised population.32,53 Noninvasive testing such as urine and serum antigens, sputum cultures, Aspergillus galactomannan assays, viral nasal swabs, and PCR studies often lead to a diagnosis in appropriate clinical scenarios. Conservative management would dictate the use of noninvasive testing whenever possible, and randomized controlled trials have shown noninvasive testing to be noninferior to FOB in preventing need for mechanical ventilation, with no difference in overall mortality.54 FOB has been shown to be most useful in establishing a diagnosis when an infectious etiology is suspected.55 In multivariate analysis, a delay in the identification of the etiology of pulmonary infiltrate was associated with increased mortality.56 Additionally, early FOB was found to be superior to late FOB in revealing a diagnosis. 32,57 Despite its ability to detect the cause of pulmonary disease, direct antibiotic therapy, and possibly change therapy, FOB with diagnostic maneuvers has not been shown to affect mortality.58 In a large case series, FOB with bronchoalveolar lavage (BAL) revealed a diagnosis in approximately 30% to 50% of cases. The addition of transbronchial biopsy did not improve diagnostic utility.58 More recent studies have confirmed that the addition of transbronchial biopsy does not add to diagnostic yield and is associated with increased adverse events.59 The appropriate use of advanced techniques such as endobronchial ultrasound–guided transbronchial needle aspirations, endobronchial biopsy, and CT-guided navigational bronchoscopy has not been established and should be considered on a case-by-case basis. In summary, routine early BAL is the diagnostic test of choice, especially when infectious pulmonary complications are suspected.
Contraindications for FOB in this population mirror those in the general population. These include acute severe hypoxemic respiratory failure, myocardial ischemia or acute coronary syndrome within 2 weeks of procedure, severe thrombocytopenia, and inability to provide or obtain informed consent from patient or health care power of attorney. Coagulopathy and thrombocytopenia are common comorbid conditions in the HSCT population. A platelet count of < 20 × 103/µL has generally been used as a cut-off for routine FOB with BAL.60 Risks of the procedures should be discussed clearly with the patient, but simple FOB for airway evaluation and BAL is generally well tolerated even under these conditions.
Early Nonifectious Pulmonary Complications
Case Patient 2 Continued
Bronchoscopy with BAL performed the day after admission is unremarkable and stains and cultures are negative for viral, bacterial, and fungal organisms. The patient is initially started on broad-spectrum antibiotics, but his oxygenation continues to worsen to the point that he is placed on noninvasive positive pressure ventilation. He is started empirically on amphotericin B and eventually is intubated. VATS lung biopsy is ultimately performed and pathology is consistent with diffuse alveolar damage.
- Based on these biopsy findings, what is the diagnosis?
Based on the pathology consistent with diffuse alveolar damage, a diagnosis of idiopathic pneumonia syndrome (IPS) is made.
- What noninfectious pulmonary complications occur in the early post-transplant period?
The overall incidence of noninfectious pulmonary complications after HSCT is generally estimated at 20% to 30%.32 Acute pulmonary edema is a common very early noninfectious pulmonary complication and clinically the most straightforward to treat. Three distinct clinical syndromes—peri-engraftment respiratory distress syndrome (PERDS), diffuse alveolar hemorrhage (DAH), and IPS—comprise the remainder of the pertinent early noninfectious complications. Clinical presentation differs based upon the disease entity. Recent studies have evaluated the role of angiotensin-converting enzyme polymorphisms as a predictive marker for risk of developing early noninfectious pulmonary complications.61
Peri-Engraftment Respiratory Distress Syndrome
PERDS is a clinical syndrome comprising the cardinal features of erythematous rash and fever along with noncardiogenic pulmonary infiltrates and hypoxemia that occur in the peri-engraftment period, defined as recovery of absolute neutrophil count to > 500/μL on 2 consecutive days.62 PERDS occurs in the autologous HSCT population and may be a clinical correlate to early GVHD in the allogeneic HSCT population. It is hypothesized that the pathophysiology underlying PERDS is an autoimmune-related capillary leak caused by pro-inflammatory cytokine release.63 Treatment remains anecdotal and currently consists of supportive care and high-dose corticosteroids. Some have favored limiting the use of gCSF given its role in stimulating rapid white blood cell recovery.33 Prognosis is favorable, but progression to fulminant respiratory failure requiring mechanical ventilation portends a poor prognosis.
Diffuse Alveolar Hemorrhage
DAH is clinical syndrome consisting of diffuse alveolar infiltrates on pulmonary imaging combined with progressively bloodier return per aliquot during BAL in 3 different subsegments or more than 20% hemosiderin-laden macrophages on BAL fluid evaluation. Classically, DAH is defined in the absence of pulmonary infection or cardiac dysfunction. The pathophysiology is thought to be related to inflammation of pulmonary vasculature within the alveolar walls leading to alveolitis. Although no prospective trials exist, early use of high-dose corticosteroid therapy is thought to improve outcomes;64,65 a recent study, however, showed low-dose steroids may be associated with the lowest mortality.66 Mortality is directly linked to the presence of superimposed infection, need for mechanical ventilation, late onset, and development of multiorgan failure.67
Idiopathic Pneumonia Syndrome
IPS is a complex clinical syndrome whose pathology is felt to stem from a variety of possible lung insults such as direct myeloablative drug toxicity, occult pulmonary infection, or cytokine-driven inflammation. The ATS published an article further subcategorizing IPS as different clinical entities based upon whether the primary insult involves the vascular endothelium, interstitial tissue, and airway tissue, truly idiopathic, or unclassified.68 In clinical practice, IPS is defined as widespread alveolar injury in the absence of evidence of renal failure, heart failure, and excessive fluid resuscitation. In addition, negative testing for a variety of bacterial, viral, and fungal causes is also necessary.69 Clinical syndromes included within the IPS definition are ARDS, acute interstitial pneumonia, DAH, cryptogenic organizing pneumonia, and BOS.70 Risk factors for developing IPS include TBI, older age of recipient, acute GVHD, and underlying diagnosis of AML or myelodysplastic syndrome.12 In addition, it has been shown that risk for developing IPS is lower in patients undergoing allogeneic HSCT who receive non-myeloablative conditioning regimens.71 The pathologic finding in IPS is diffuse alveolar damage. A 2006 study in which investigators reviewed BAL samples from patients with IPS found that 3% of the patients had PCR evidence of human metapneumovirus infection, and a study in 2015 found PCR evidence of infection in 53% of BAL samples from patients diagnosed with IPS.72,73 This fuels the debate on whether IPS is truly an infection-driven process where the source of infection, pulmonary or otherwise, simply escapes detection. Various surfactant proteins, which play a role in decreasing surface tension within the alveolar interface and function as mediators within the innate immunity of the lung, have been studied in regard to development of IPS. Small retrospective studies have shown a trend toward lower pre-transplant serum protein surfactant D and the development of IPS.74
The diagnosis of IPS does not require pathologic diagnosis in most circumstances. The correct clinical findings in association with a negative infectious workup lead to a presumptive diagnosis of IPS. The extent of the infectious workup that must be completed to adequately rule out infection is often a difficult clinical question. Recent recommendations include BAL fluid evaluation for routine bacterial cultures, appropriate viral culture, and consideration of PCR testing to evaluate for Mycoplasma, Chlamydia, and Aspergillus antigens.75 Transbronchial biopsy continues to appear in recommendations, but is not routinely performed and should be completed as the patient’s clinical status permits.8,68 Table 3 reviews basic features of early noninfectious pulmonary complications.
Treatment of IPS centers around moderate to high doses of corticosteroids. Based on IPS experimental modes, tumor necrosis factor (TNF)-α has been implicated as an important mediator. Unfortunately, several studies evaluating etanercept have produced conflicting results, and this agent’s clinical effects on morbidity and mortality remain in question.76
- What treatment should be offered to the patient with diffuse alveolar damage on biopsy?
Treatment consists of supportive care and empiric broad-spectrum antibiotics with consideration of high-dose corticosteroids. Based upon early studies in murine models implicating TNF, pilot studies were performed evaluating etanercept as a possible safe and effective addition to high-dose systemic corticosteroids.77 Although these results were promising, data from a truncated randomized control clinical trial failed to show improvement in patient response in the adult population.76 More recent data from the same author suggests that pediatric populations with IPS are, however, responsive to etanercept and high-dose corticosteroid therapy.78 When IPS develops as a late complication, treatment with high-dose corticosteroids (2 mg/kg/day) and etanercept (0.4 mg/kg twice weekly) has been shown to improve 2-year survival.79
Case Patient 2 Conclusion
The patient is started on steroids and makes a speedy recovery. He is successfully extubated 5 days later.
Conclusion
Careful pretransplant evaluation, including a full set of pulmonary function tests, can help predict a patient’s risk for pulmonary complications after transplant, allowing risk factor modification strategies to be implemented prior to transplant, including smoking cessation. It also helps identify patients at high risk for complications who will require closer monitoring after transplantation. Early posttransplant complications include infectious and noninfectious entities. Bacterial, viral, and fungal pneumonias are in the differential of infectious pneumonia, and bronchoscopy can be helpful in establishing a diagnosis. A common, important noninfectious cause of early pulmonary complications is IPS, which is treated with steroids and sometimes anti-TNF therapy.
Hematopoietic stem cell transplantation (HSCT) is widely used in the economically developed world to treat a variety of hematologic malignancies as well as nonmalignant diseases and solid tumors. An estimated 17,900 HSCTs were performed in 2011, and survival rates continue to increase.1 Pulmonary complications post HSCT are common, with rates ranging from 40% to 60%, and are associated with increased morbidity and mortality.2
Clinical diagnosis of pulmonary complications in the HSCT population has been aided by a previously well-defined chronology of the most common diseases.3 Historically, early pulmonary complications were defined as pulmonary complications occurring within 100 days of HSCT (corresponding to the acute graft-versus-host disease [GVHD] period). Late pulmonary complications are those that occur thereafter. This timeline, however, is now more variable given the increasing indications for HSCT, the use of reduced-intensity conditioning strategies, and varied individual immune reconstitution. This article discusses the management of early post-HSCT pulmonary complications; late post-HSCT pulmonary complications will be discussed in a separate follow-up article.
Transplant Basics
The development of pulmonary complications is affected by many factors associated with the transplant. Autologous transplantation involves the collection of a patient’s own stem cells, appropriate storage and processing, and re-implantation after induction therapy. During induction therapy, the patient undergoes high-dose chemotherapy or radiation therapy that ablates the bone marrow. The stem cells are then transfused back into the patient to repopulate the bone marrow. Allogeneic transplants involve the collection of stem cells from a donor. Donors are matched as closely as possible to the recipient’s histocompatibility antigen (HLA) haplotypes to prevent graft failure and rejection. The donor can be related or unrelated to the recipient. If there is not a possibility of a related match (from a sibling), then a national search is undertaken to look for a match through the National Marrow Donor Program. There are fewer transplant reactions and occurrences of GVHD if the major HLAs of the donor and recipient match. Table 1 reviews basic definitions pertaining to HSCT.
How the cells for transplantation are obtained is also an important factor in the rate of complications. There are 3 main sources: peripheral blood, bone marrow, and umbilical cord. Peripheral stem cell harvesting involves exposing the donor to granulocyte-colony stimulating factor (gCSF), which increases peripheral circulation of stem cells. These cells are then collected and infused into the recipient after the recipient has completed an induction regimen involving chemotherapy and/or radiation, depending on the protocol. This procedure is called peripheral blood stem cell transplant (PBSCT). Stem cells can also be directly harvested from bone marrow cells, which are collected from repeated aspiration of bone marrow from the posterior iliac crest.4 This technique is most common in children, whereas in adults peripheral blood stem cells are the most common source. Overall mortality does not differ based on the source of the stem cells. It is postulated that GVHD may be more common in patients undergoing PBSCT, but the graft failure rate may be lower.5
The third option is umbilical cord blood (UCB) as the source of stem cells. This involves the collection of umbilical cord blood that is prepared and frozen after birth. It has a smaller volume of cells, and although fewer cells are needed when using UCB, 2 separate donors may be required for a single adult recipient. The engraftment of the stem cells is slower and infections in the post-transplant period are more common. Prior reports indicate GVHD rates may be lower.4 While the use of UCB is not common in adults, the incidence has doubled over the past decade, increasing from 3% to 6%.
The conditioning regimen can influence pulmonary complications. Traditionally, an ablative transplant involves high-dose chemotherapy or radiation to eradicate the recipient’s bone marrow. This regimen can lead to many complications, especially in the immediate post-transplant period. In the past 10 years, there has been increasing interest in non-myeloablative, or reduced-intensity, conditioning transplants.6 These “mini transplants” involve smaller doses of chemotherapy or radiation, which do not totally eradicate the bone marrow; after the transplant a degree of chimerism develops where the donor and recipient stem cells coexist. The medications in the preparative regimen also should be considered because they can affect pulmonary complications after transplant. Certain chemotherapeutic agents such as carmustine, bleomycin, and many others can lead to acute and chronic presentations of pulmonary diseases such as hypersensitivity pneumonitis, pulmonary fibrosis, acute respiratory distress syndrome, and abnormal pulmonary function testing.
After the HSCT, GVHD can develop in more than 50% of allogeneic recipients.3 The incidence of GVHD has been reported to be increasing over the past 12 years.It is divided into acute GVHD (which traditionally happens in the first 100 days after transplant) and chronic GVHD (after day 100). This calendar-day–based system has been augmented based on a 2006 National Institutes of Health working group report emphasizing the importance of organ-specific features of chronic GVHD in the clinical presentation of GVHD.7 Histologic changes in chronic organ GVHD tend to include more fibrotic features, whereas in acute GVHD more inflammatory changes are seen. The NIH working group report also stressed the importance of obtaining a biopsy specimen for histopathologic review and interdisciplinary collaboration to arrive at a consensus diagnosis, and noted the limitations of using histologic changes as the sole determinant of a “gold standard” diagnosis.7 GVHD can directly predispose patients to pulmonary GVHD and indirectly predispose them to infectious complications because the mainstay of therapy for GVHD is increased immunosuppression.
Pretransplant Evaluation
Case Patient 1
A 56-year-old man is diagnosed with acute myeloid leukemia (AML) after presenting with signs and symptoms consistent with pancytopenia. He has a past medical history of chronic sinus congestion, arthritis, depression, chronic pain, and carpal tunnel surgery. He is employed as an oilfield worker and has a 40-pack-year smoking history, but he recently cut back to half a pack per day. He is being evaluated for allogeneic transplant with his brother as the donor and the planned conditioning regimen is total body irradiation (TBI), thiotepa, cyclophosphamide, and antithymocyte globulin with T-cell depletion. Routine pretransplant pulmonary function testing (PFT) reveals a restrictive pattern and he is sent for pretransplant pulmonary evaluation.
Physical exam reveals a chronically ill appearing man. He is afebrile, the respiratory rate is 16 breaths/min, blood pressure is 145/88 mm Hg, heart rate is 92 beats/min, and oxygen saturation is 95%. He is in no distress. Auscultation of the chest reveals slightly diminished breath sounds bilaterally but is clear and without wheezes, rhonchi, or rales. Heart exam shows regular rate and rhythm without murmurs, rubs, or gallops. Extremities reveal no edema or rashes. Otherwise, the remainder of the exam is normal. The patient’s PFT results are shown in Table 2.
- What aspects of this patient’s history put him at risk for pulmonary complications after transplantation?
Risk Factors for Pulmonary Complications
Predicting who is at risk for pulmonary complications is difficult. Complications are generally divided into infectious and noninfectious categories. Regardless of category, allogeneic HSCT recipients are at increased risk compared with autologous recipients, but even in autologous transplants, more than 25% of patients will develop pulmonary complications in the first year.8 Prior to transplant, patients undergo full PFT. Early on, many studies attempted to show relationships between various factors and post-transplant pulmonary complications. Factors that were implicated were forced expiratory volume in 1 second (FEV1), diffusing capacity of the lung for carbon monoxide (D
Another sometimes overlooked risk before transplantation is restrictive lung disease. One study showed a twofold increase in respiratory failure and mortality if there was pretransplant restriction based on TLC < 80%.16
An interesting study by one group in pretransplant evaluation found decreased muscle strength by maximal inspiratory muscle strength (PImax), maximal expiratory muscle strength (PEmax), dominant hand grip strength, and 6-minute walk test (6MWT) distance prior to allogeneic transplant, but did not find a relationship between these variables and mortality.17 While this study had a small sample size, these findings likely deserve continued investigation.18
- What methods are used to calculate risk for complications?
Risk Scoring Systems
Several pretransplantation risk scores have been developed. In a study that looked at more than 2500 allogeneic transplants, Parimon et al showed that risk of mortality and respiratory failure could be estimated prior to transplant using a scoring system—the Lung Function Score (LFS)—that combines the FEV1 and D
The Pretransplantation Assessment of Mortality score, initially developed in 2006, predicts mortality within the first 2 years after HSCT based on 8 clinical factors: disease risk, age at transplant, donor type, conditioning regimen, and markers of organ function (percentage of predicted FEV1, percentage of predicted D
- What other preoperative testing or interventions should be considered in this patient?
Since there is a high risk of infectious complications after transplant, the question of whether pretransplantation patients should undergo screening imaging may arise. There is no evidence that routine chest computed tomography (CT) reduces the risk of infectious complications after transplantation.26 An area that may be insufficiently addressed in the pretransplantation evaluation is smoking cessation counseling.27 Studies have shown an elevated risk of mortality in smokers.28-30 Others have found a higher incidence of respiratory failure but not an increased mortality.31 Overall, with the good rates of smoking cessation that can be accomplished, smokers should be counseled to quit before transplantation.
In summary, patients should undergo full PFTs prior to transplantation to help stratify risk for pulmonary complications and mortality and to establish a clinical baseline. The LFS (using FEV1 and D
Case Patient 1 Conclusion
The patient undergoes transplantation due to his lack of other treatment options. Evaluation prior to transplant, however, shows that he is at high risk for pulmonary complications. He has a LFS of 7 prior to transplant (using the D
Early Infectious Pulmonary Complications
Case Patient 2
A 27-year-old man with a medical history significant for AML and allogeneic HSCT presents with cough productive of a small amount of clear to white sputum, dyspnea on exertion, and fevers for 1 week. He also has mild nausea and a decrease in appetite. He underwent HSCT 2.5 months prior to admission, which was a matched unrelated bone marrow transplant with TBI and cyclophosphamide conditioning. His past medical history is significant only for exercise-induced asthma for which he takes a rescue inhaler infrequently prior to transplantation. His pretransplant PFTs showed normal spirometry with an FEV1 of 106% of predicted and D
Physical exam is notable for fever of 101.0°F, heart rate 80 beats/min, respiratory rate 16 breaths/ min, and blood pressure 142/78 mm Hg; an admission oxygen saturation is 93% on room air. Lungs show bibasilar crackles and the remainder of the exam is normal. Laboratory testing shows a white blood cell count of 2400 cells/μL, hemoglobin 7.6 g/dL, and platelet count 66 × 103/μL. Creatinine is 1.0 mg/dL. Chest radiograph shows ill-defined bilateral lower-lobe infiltrates. CT scans are shown in the Figure.
- For which infectious complications is this patient most at risk?
Pneumonia
A prospective trial in the HSCT population reported a pneumonia incidence rate of 68%, and pneumonia is more common in allogeneic HSCT with prolonged immunosuppressive therapy.32 Development of pneumonia within 100 days of transplant directly correlates with nonrelapsed mortality.33 Early detection is key, and bronchoscopy within the first 5 days of symptoms has been shown to change therapy in approximately 40% of cases but has not been shown to affect mortality.34 The clinical presentation of pneumonia in the HSCT population can be variable because of the presence of neutropenia and profound immunosuppression. Traditionally accepted diagnostic criteria of fevers, sputum production, and new infiltrates should be used with caution, and an appropriately high index of suspicion should be maintained. Progression to respiratory failure, regardless of causative organism of infection, portends a poor prognosis, with mortality rates estimated at 70% to 90%.35,36 Several transplant-specific factors may affect early infections. For instance, UCB transplants have been found to have a higher incidence of invasive aspergillosis and cytomegalovirus (CMV) infections but without higher mortality attributed to the infections.37
Bacterial Pneumonia
Bacterial pneumonia accounts for 20% to 50% of pneumonia cases in HSCT recipients.38 Gram-negative organisms, specifically Pseudomonas aeruginosa and Escherichia coli, were reported to be the most common pathologic bacteria in recent prospective trials, whereas previous retrospective trials showed that common community-acquired organisms were the most common cause of pneumonia in HSCT recipients.32,39 This underscores the importance of being aware of the clinical prevalence of microorganisms and local antibiograms, along with associated institutional susceptibility profiles. Initiation of immediate empiric broad-spectrum antibiotics is essential when bacterial pneumonia is suspected.
Viral Pneumonia
The prevalence of viral pneumonia in stem cell transplant recipients is estimated at 28%,32 with most cases being caused by community viral pathogens such as rhinovirus, respiratory syncytial virus (RSV), influenza A and B, and parainfluenza.39 The prevention, prophylaxis, and early treatment of viral pneumonias, specifically CMV infection, have decreased the mortality associated with early pneumonia after HSCT. Co-infection with bacterial organisms must be considered and has been associated with increased mortality in the intensive care unit setting.40
Supportive treatment with rhinovirus infection is sufficient as the disease is usually self-limited in immunocompromised patients. In contrast, infection with RSV in the lower respiratory tract is associated with increased mortality in prior reports, and recent studies suggest that further exploration of prophylaxis strategies is warranted.41 Treatment with ribavirin remains the backbone of therapy, but drug toxicity continues to limit its use. The addition of immunomodulators such as RSV immune globulin or palivizumab to ribavirin remains controversial, but a retrospective review suggests that early treatment may prevent progression to lower respiratory tract infection and lead to improved mortality.42 Infection with influenza A/B must be considered during influenza season. Treatment with oseltamivir may shorten the duration of disease when influenza A/B or parainfluenza are detected. Reactivation of latent herpes simplex virus during the pre-engraftment phase should also be considered. Treatment is similar to that in nonimmunocompromised hosts. When CMV pneumonia is suspected, careful history regarding compliance with prophylactic antivirals and CMV status of both the recipient and donor are key. A presumptive diagnosis can be made with the presence of appropriate clinical scenario, supportive radiographic images showing areas of ground-glass opacification or consolidation, and positive CMV polymerase chain reaction (PCR) assay. Visualization of inclusion bodies on lung biopsy tissue remains the gold standard for diagnosis. Treatment consists of CMV immunoglobulin and ganciclovir.
Fungal Pneumonia
Early fungal pneumonias have been associated with increased mortality in the HSCT population.43 Clinical suspicion should remain high and compliance with antifungal prophylaxis should be questioned thoroughly. Invasive aspergillosis (IA) remains the most common fungal infection. A bimodal distribution of onset of infection peaking on day 16 and again on day 96 has been described in the literature.44 Patients often present with classic pneumonia symptoms, but these may be accompanied by hemoptysis. Proven IA diagnosis requires visualization of fungal forms from biopsy or needle aspiration or a positive culture obtained in a sterile fashion.45 Most clinical data comes from experience with probable and possible diagnosis of IA. Bronchoalveolar lavage with testing with Aspergillus galactomannan assay has been shown to be clinically useful in establishing the clinical diagnosis in the HSCT population.46 Classic air-crescent findings on chest CT are helpful in establishing a possible diagnosis, but retrospective analysis reveals CT findings such as focal infiltrates and pulmonary nodular patterns are more common.47 First-line treatment with voriconazole has been shown to decrease short-term mortality attributable to IA but has not had an effect on long-term, all-cause mortality.48 Surgical resection is reserved for patients with refractory disease or patients presenting with massive hemoptysis.
Mucormycosis is an emerging disease with ever increasing prevalence in the HSCT population, reflecting the improved prophylaxis and treatment of IA. Initial clinical presentation is similar to IA, most commonly affecting the lung, although craniofacial involvement is classic for mucormycosis, especially in HSCT patients with diabetes.49Mucor infections can present with massive hemoptysis due to tissue invasion and disregard for tissue and fascial planes. Diagnosis of mucormycosis is associated with as much as a six-fold increase in risk for death. Diagnosis requires identification of the organism by examination or culture and biopsy is often necessary.50,51 Amphotericin B remains first-line therapy as mucormycosis is resistant to azole antifungals, with higher doses recommended for cerebral involvement.52
Candida pulmonary infections during the early HSCT period are becoming increasingly rare due to widespread use of fluconazole prophylaxis and early treatment of mucosal involvement during neutropenia. Endemic fungal infections such as blastomycosis, coccidioidomycosis, and histoplasmosis should be considered in patients inhabiting specific geographic areas or with recent travel to these areas.
- What test should be performed to evaluate for infectious causes of pneumonia?
Role of Flexible Fiberoptic Bronchoscopy
The utility of flexible fiberoptic bronchoscopy (FOB) in immune-compromised patients for the evaluation of pulmonary infiltrates is a frequently debated topic. Current studies suggest a diagnosis can be made in approximately 80% of cases in the immune-compromised population.32,53 Noninvasive testing such as urine and serum antigens, sputum cultures, Aspergillus galactomannan assays, viral nasal swabs, and PCR studies often lead to a diagnosis in appropriate clinical scenarios. Conservative management would dictate the use of noninvasive testing whenever possible, and randomized controlled trials have shown noninvasive testing to be noninferior to FOB in preventing need for mechanical ventilation, with no difference in overall mortality.54 FOB has been shown to be most useful in establishing a diagnosis when an infectious etiology is suspected.55 In multivariate analysis, a delay in the identification of the etiology of pulmonary infiltrate was associated with increased mortality.56 Additionally, early FOB was found to be superior to late FOB in revealing a diagnosis. 32,57 Despite its ability to detect the cause of pulmonary disease, direct antibiotic therapy, and possibly change therapy, FOB with diagnostic maneuvers has not been shown to affect mortality.58 In a large case series, FOB with bronchoalveolar lavage (BAL) revealed a diagnosis in approximately 30% to 50% of cases. The addition of transbronchial biopsy did not improve diagnostic utility.58 More recent studies have confirmed that the addition of transbronchial biopsy does not add to diagnostic yield and is associated with increased adverse events.59 The appropriate use of advanced techniques such as endobronchial ultrasound–guided transbronchial needle aspirations, endobronchial biopsy, and CT-guided navigational bronchoscopy has not been established and should be considered on a case-by-case basis. In summary, routine early BAL is the diagnostic test of choice, especially when infectious pulmonary complications are suspected.
Contraindications for FOB in this population mirror those in the general population. These include acute severe hypoxemic respiratory failure, myocardial ischemia or acute coronary syndrome within 2 weeks of procedure, severe thrombocytopenia, and inability to provide or obtain informed consent from patient or health care power of attorney. Coagulopathy and thrombocytopenia are common comorbid conditions in the HSCT population. A platelet count of < 20 × 103/µL has generally been used as a cut-off for routine FOB with BAL.60 Risks of the procedures should be discussed clearly with the patient, but simple FOB for airway evaluation and BAL is generally well tolerated even under these conditions.
Early Nonifectious Pulmonary Complications
Case Patient 2 Continued
Bronchoscopy with BAL performed the day after admission is unremarkable and stains and cultures are negative for viral, bacterial, and fungal organisms. The patient is initially started on broad-spectrum antibiotics, but his oxygenation continues to worsen to the point that he is placed on noninvasive positive pressure ventilation. He is started empirically on amphotericin B and eventually is intubated. VATS lung biopsy is ultimately performed and pathology is consistent with diffuse alveolar damage.
- Based on these biopsy findings, what is the diagnosis?
Based on the pathology consistent with diffuse alveolar damage, a diagnosis of idiopathic pneumonia syndrome (IPS) is made.
- What noninfectious pulmonary complications occur in the early post-transplant period?
The overall incidence of noninfectious pulmonary complications after HSCT is generally estimated at 20% to 30%.32 Acute pulmonary edema is a common very early noninfectious pulmonary complication and clinically the most straightforward to treat. Three distinct clinical syndromes—peri-engraftment respiratory distress syndrome (PERDS), diffuse alveolar hemorrhage (DAH), and IPS—comprise the remainder of the pertinent early noninfectious complications. Clinical presentation differs based upon the disease entity. Recent studies have evaluated the role of angiotensin-converting enzyme polymorphisms as a predictive marker for risk of developing early noninfectious pulmonary complications.61
Peri-Engraftment Respiratory Distress Syndrome
PERDS is a clinical syndrome comprising the cardinal features of erythematous rash and fever along with noncardiogenic pulmonary infiltrates and hypoxemia that occur in the peri-engraftment period, defined as recovery of absolute neutrophil count to > 500/μL on 2 consecutive days.62 PERDS occurs in the autologous HSCT population and may be a clinical correlate to early GVHD in the allogeneic HSCT population. It is hypothesized that the pathophysiology underlying PERDS is an autoimmune-related capillary leak caused by pro-inflammatory cytokine release.63 Treatment remains anecdotal and currently consists of supportive care and high-dose corticosteroids. Some have favored limiting the use of gCSF given its role in stimulating rapid white blood cell recovery.33 Prognosis is favorable, but progression to fulminant respiratory failure requiring mechanical ventilation portends a poor prognosis.
Diffuse Alveolar Hemorrhage
DAH is clinical syndrome consisting of diffuse alveolar infiltrates on pulmonary imaging combined with progressively bloodier return per aliquot during BAL in 3 different subsegments or more than 20% hemosiderin-laden macrophages on BAL fluid evaluation. Classically, DAH is defined in the absence of pulmonary infection or cardiac dysfunction. The pathophysiology is thought to be related to inflammation of pulmonary vasculature within the alveolar walls leading to alveolitis. Although no prospective trials exist, early use of high-dose corticosteroid therapy is thought to improve outcomes;64,65 a recent study, however, showed low-dose steroids may be associated with the lowest mortality.66 Mortality is directly linked to the presence of superimposed infection, need for mechanical ventilation, late onset, and development of multiorgan failure.67
Idiopathic Pneumonia Syndrome
IPS is a complex clinical syndrome whose pathology is felt to stem from a variety of possible lung insults such as direct myeloablative drug toxicity, occult pulmonary infection, or cytokine-driven inflammation. The ATS published an article further subcategorizing IPS as different clinical entities based upon whether the primary insult involves the vascular endothelium, interstitial tissue, and airway tissue, truly idiopathic, or unclassified.68 In clinical practice, IPS is defined as widespread alveolar injury in the absence of evidence of renal failure, heart failure, and excessive fluid resuscitation. In addition, negative testing for a variety of bacterial, viral, and fungal causes is also necessary.69 Clinical syndromes included within the IPS definition are ARDS, acute interstitial pneumonia, DAH, cryptogenic organizing pneumonia, and BOS.70 Risk factors for developing IPS include TBI, older age of recipient, acute GVHD, and underlying diagnosis of AML or myelodysplastic syndrome.12 In addition, it has been shown that risk for developing IPS is lower in patients undergoing allogeneic HSCT who receive non-myeloablative conditioning regimens.71 The pathologic finding in IPS is diffuse alveolar damage. A 2006 study in which investigators reviewed BAL samples from patients with IPS found that 3% of the patients had PCR evidence of human metapneumovirus infection, and a study in 2015 found PCR evidence of infection in 53% of BAL samples from patients diagnosed with IPS.72,73 This fuels the debate on whether IPS is truly an infection-driven process where the source of infection, pulmonary or otherwise, simply escapes detection. Various surfactant proteins, which play a role in decreasing surface tension within the alveolar interface and function as mediators within the innate immunity of the lung, have been studied in regard to development of IPS. Small retrospective studies have shown a trend toward lower pre-transplant serum protein surfactant D and the development of IPS.74
The diagnosis of IPS does not require pathologic diagnosis in most circumstances. The correct clinical findings in association with a negative infectious workup lead to a presumptive diagnosis of IPS. The extent of the infectious workup that must be completed to adequately rule out infection is often a difficult clinical question. Recent recommendations include BAL fluid evaluation for routine bacterial cultures, appropriate viral culture, and consideration of PCR testing to evaluate for Mycoplasma, Chlamydia, and Aspergillus antigens.75 Transbronchial biopsy continues to appear in recommendations, but is not routinely performed and should be completed as the patient’s clinical status permits.8,68 Table 3 reviews basic features of early noninfectious pulmonary complications.
Treatment of IPS centers around moderate to high doses of corticosteroids. Based on IPS experimental modes, tumor necrosis factor (TNF)-α has been implicated as an important mediator. Unfortunately, several studies evaluating etanercept have produced conflicting results, and this agent’s clinical effects on morbidity and mortality remain in question.76
- What treatment should be offered to the patient with diffuse alveolar damage on biopsy?
Treatment consists of supportive care and empiric broad-spectrum antibiotics with consideration of high-dose corticosteroids. Based upon early studies in murine models implicating TNF, pilot studies were performed evaluating etanercept as a possible safe and effective addition to high-dose systemic corticosteroids.77 Although these results were promising, data from a truncated randomized control clinical trial failed to show improvement in patient response in the adult population.76 More recent data from the same author suggests that pediatric populations with IPS are, however, responsive to etanercept and high-dose corticosteroid therapy.78 When IPS develops as a late complication, treatment with high-dose corticosteroids (2 mg/kg/day) and etanercept (0.4 mg/kg twice weekly) has been shown to improve 2-year survival.79
Case Patient 2 Conclusion
The patient is started on steroids and makes a speedy recovery. He is successfully extubated 5 days later.
Conclusion
Careful pretransplant evaluation, including a full set of pulmonary function tests, can help predict a patient’s risk for pulmonary complications after transplant, allowing risk factor modification strategies to be implemented prior to transplant, including smoking cessation. It also helps identify patients at high risk for complications who will require closer monitoring after transplantation. Early posttransplant complications include infectious and noninfectious entities. Bacterial, viral, and fungal pneumonias are in the differential of infectious pneumonia, and bronchoscopy can be helpful in establishing a diagnosis. A common, important noninfectious cause of early pulmonary complications is IPS, which is treated with steroids and sometimes anti-TNF therapy.
1. Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010;303:1617–24.
2. Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004;170:22–48.
4. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med 2006;354:1813–26.
5. Anasetti C, Logan BR, Lee SJ, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 2012;367:1487–96.
6. Giralt S, Ballen K, Rizzo D, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009;15:367–9.
7. Shulman HM, Kleiner D, Lee SJ, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant 2006;12:31–47.
8. Afessa B, Abdulai RM, Kremers WK, et al. Risk factors and outcome of pulmonary complications after autologous hematopoietic stem cell transplant. Chest 2012;141:442–50.
9. Bolwell BJ. Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003;32:853–61.
10. Carlson K, Backlund L, Smedmyr B, et al. Pulmonary function and complications subsequent to autologous bone marrow transplantation. Bone Marrow Transplant 1994;14:805–11.
11. Clark JG, Schwartz DA, Flournoy N, et al. Risk factors for airflow obstruction in recipients of bone marrow transplants. Ann Intern Med 1987;107:648–56.
12. Crawford SW, Fisher L. Predictive value of pulmonary function tests before marrow transplantation. Chest 1992; 101:1257–64.
13. Ghalie R, Szidon JP, Thompson L, et al. Evaluation of pulmonary complications after bone marrow transplantation: the role of pretransplant pulmonary function tests. Bone Marrow Transplant 1992;10:359–65.
14. Ho VT, Weller E, Lee SJ, et al. Prognostic factors for early severe pulmonary complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001;7:223–9.
15. Horak DA, Schmidt GM, Zaia JA, et al. Pretransplant pulmonary function predicts cytomegalovirus-associated interstitial pneumonia following bone marrow transplantation. Chest 1992;102:1484–90.
16. Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, et al. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transplant 2010;16:199–206.
17. White AC, Terrin N, Miller KB, Ryan HF. Impaired respiratory and skeletal muscle strength in patients prior to hematopoietic stem-cell transplantation. Chest 2005;128145–52.
18. Afessa B. Pretransplant pulmonary evaluation of the blood and marrow transplant recipient. Chest 2005;128:8–10.
19. Parimon T, Madtes DK, Au DH, et al. Pretransplant lung function, respiratory failure, and mortality after stem cell transplantation. Am J Respir Crit Care Med 2005;172:384–90.
20. Pavletic SZ, Martin P, Lee SJ, et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006;12:252–66.
21. Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med 2006;144:407–14.
22. Au BK, Gooley TA, Armand P, et al. Reevaluation of the pretransplant assessment of mortality score after allogeneic hematopoietic transplantation. Biol Blood Marrow Transplant 2015;21:848–54.
23. Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005;106:2912–9.
24. Chien JW, Sullivan KM. Carbon monoxide diffusion capacity: how low can you go for hematopoietic cell transplantation eligibility? Biol Blood Marrow Transplant 2009;15: 447–53.
25. Coffey DG, Pollyea DA, Myint H, et al. Adjusting DLCO for Hb and its effects on the Hematopoietic Cell Transplantation-specific Comorbidity Index. Bone Marrow Transplant 2013;48:1253–6.
26. Kasow KA, Krueger J, Srivastava DK, et al. Clinical utility of computed tomography screening of chest, abdomen, and sinuses before hematopoietic stem cell transplantation: the St. Jude experience. Biol Blood Marrow Transplant 2009;15:490–5.
27. Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45: 1259–68.
28. Savani BN, Montero A, Wu C, et al. Prediction and prevention of transplant-related mortality from pulmonary causes after total body irradiation and allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:223–30.
29. Ehlers SL, Gastineau DA, Patten CA, et al. The impact of smoking on outcomes among patients undergoing hematopoietic SCT for the treatment of acute leukemia. Bone Marrow Transplant 2011;46:285–90.
30. Marks DI, Ballen K, Logan BR, et al. The effect of smoking on allogeneic transplant outcomes. Biol Blood Marrow Transplant 2009;15:1277–87.
31. Tran BT, Halperin A, Chien JW. Cigarette smoking and outcomes after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2011;17:1004–11.
32. Lucena CM, Torres A, Rovira M, et al. Pulmonary complications in hematopoietic SCT: a prospective study. Bone Marrow Transplant 2014;49:1293–9.
33. Chi AK, Soubani AO, White AC, Miller KB. An update on pulmonary complications of hematopoietic stem cell transplantation. Chest 2013;144:1913–22.
34. Dunagan DP, Baker AM, Hurd DD, Haponik EF. Bronchoscopic evaluation of pulmonary infiltrates following bone marrow transplantation. Chest 1997;111:135–41.
35. Naeem N, Reed MD, Creger RJ, et al. Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006;37:119–33.
36. Afessa B, Tefferi A, Hoagland HC, et al. Outcome of recipients of bone marrow transplants who require intensive care unit support. Mayo Clin Proc 1992;67:117–22.
37. Parody R, Martino R, de la Camara R, et al. Fungal and viral infections after allogeneic hematopoietic transplantation from unrelated donors in adults: improving outcomes over time. Bone Marrow Transplant 2015;50:274–81.
38. Orasch C, Weisser M, Mertz D, et al. Comparison of infectious complications during induction/consolidation chemotherapy versus allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:521–6.
39. Aguilar-Guisado M, Jimenez-Jambrina M, Espigado I, et al. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 2011;25:E629–38.
40. Palacios G, Hornig M, Cisterna D, et al. Streptococcus pneumoniae coinfection is correlated with the severity of H1N1 pandemic influenza. PLoS One 2009;4:e8540.
41. Hynicka LM, Ensor CR. Prophylaxis and treatment of respiratory syncytial virus in adult immunocompromised patients. Ann Pharmacother 2012;46:558–66.
42. Shah JN, Chemaly RF. Management of RSV infections in adult recipients of hematopoietic stem cell transplantation. Blood 2011;2755–63.
43. Marr KA, Bowden RA. Fungal infections in patients undergoing blood and marrow transplantation. Transpl Infect Dis 1999;1:237–46.
44. Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J Infect Dis 1997;175:1459–66.
45. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7–14.
46. Fisher CE, Stevens AM, Leisenring W, et al. Independent contribution of bronchoalveolar lavage and serum galactomannan in the diagnosis of invasive pulmonary aspergillosis. Transpl Infect Dis 2014;16:505–10.
47. Kojima R, Tateishi U, Kami M, et al. Chest computed tomography of late invasive aspergillosis after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:506–11.
48. Salmeron G, Porcher R, Bergeron A, et al. Persistent poor long-term prognosis of allogeneic hematopoietic stem cell transplant recipients surviving invasive aspergillosis. Haematologica 2012;97:1357–63.
49. McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982;92(10 Pt 1):1140.
50. Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012;54 Suppl 1:S55–60.
51. Klingspor L, Saaedi B, Ljungman P, Szakos A. Epidemiology and outcomes of patients with invasive mould infections: a retrospective observational study from a single centre (2005-2009). Mycoses 2015;58:470–7.
52. Danion F, Aguilar C, Catherinot E, et al. Mucormycosis: new developments in a persistently devastating infection. Semin Respir Crit Care Med 2015;36:692–70.
53. Rano A, Agusti C, Jimenez P, et al. Pulmonary infiltrates in non-HIV immunocompromised patients: a diagnostic approach using non-invasive and bronchoscopic procedures. Thorax 2001;56:379–87.
54. Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med 2008;36:100–7.
55. Jain P, Sandur S, Meli Y, et al. Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 2004;125:712–22.
56. Rano A, Agusti C, Benito N, et al. Prognostic factors of non-HIV immunocompromised patients with pulmonary infiltrates. Chest 2002;122:253–61.
57. Shannon VR, Andersson BS, Lei X, et al. Utility of early versus late fiberoptic bronchoscopy in the evaluation of new pulmonary infiltrates following hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:647–55.
58. Patel NR, Lee PS, Kim JH, et al. The influence of diagnostic bronchoscopy on clinical outcomes comparing adult autologous and allogeneic bone marrow transplant patients. Chest 2005;127:1388–96.
59. Chellapandian D, Lehrnbecher T, Phillips B, et al. Bronchoalveolar lavage and lung biopsy in patients with cancer and hematopoietic stem-cell transplantation recipients: a systematic review and meta-analysis. J Clin Oncol 2015;33:501–9.
60. Carr IM, Koegelenberg CF, von Groote-Bidlingmaier F, et al. Blood loss during flexible bronchoscopy: a prospective observational study. Respiration 2012;84:312–8.
61. Miyamoto M, Onizuka M, Machida S, et al. ACE deletion polymorphism is associated with a high risk of non-infectious pulmonary complications after stem cell transplantation. Int J Hematol 2014;99:175–83.
62. Capizzi SA, Kumar S, Huneke NE, et al. Peri-engraftment respiratory distress syndrome during autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1299–303.
63. Spitzer TR. Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:893–8.
64. Wanko SO, Broadwater G, Folz RJ, Chao NJ. Diffuse alveolar hemorrhage: retrospective review of clinical outcome in allogeneic transplant recipients treated with aminocaproic acid. Biol Blood Marrow Transplant 2006;12:949–53.
65. Metcalf JP, Rennard SI, Reed EC, et al. Corticosteroids as adjunctive therapy for diffuse alveolar hemorrhage associated with bone marrow transplantation. University of Nebraska Medical Center Bone Marrow Transplant Group. Am J Med 1994;96:327–34.
66. Rathi NK, Tanner AR, Dinh A, et al. Low-, medium- and high-dose steroids with or without aminocaproic acid in adult hematopoietic SCT patients with diffuse alveolar hemorrhage. Bone Marrow Transplant 2015;50:420–6.
67. Afessa B, Tefferi A, Litzow MR, Peters SG. Outcome of diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med 2002;166:1364–8.
68. Panoskaltsis-Mortari A, Griese M, Madtes DK, et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am J Respir Crit Care Med 2011;183:1262–79.
69. Clark JG, Hansen JA, Hertz MI, Pet al. NHLBI workshop summary. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Resp Dis 1993;147:1601–6.
70. Vande Vusse LK, Madtes DK. Early onset noninfectious pulmonary syndromes after hematopoietic cell transplantation. Clin Chest Med 2017;38:233–48.
71. Fukuda T, Hackman RC, Guthrie KA, et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003;102:2777–85.
72. Englund JA, Boeckh M, Kuypers J, et al. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med 2006;144:344–9.
73. Seo S, Renaud C, Kuypers JM, et al. Idiopathic pneumonia syndrome after hematopoietic cell transplantation: evidence of occult infectious etiologies. Blood 2015;125:3789–97.
74. Nakane T, Nakamae H, Kamoi H, et al. Prognostic value of serum surfactant protein D level prior to transplant for the development of bronchiolitis obliterans syndrome and idiopathic pneumonia syndrome following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;42:43–9.
75. Gilbert CR, Lerner A, Baram M, Awsare BK. Utility of flexible bronchoscopy in the evaluation of pulmonary infiltrates in the hematopoietic stem cell transplant population—a single center fourteen year experience. Arch Bronconeumol 2013;49:189–95.
76. Yanik GA, Horowitz MM, Weisdorf DJ, et al. Randomized, double-blind, placebo-controlled trial of soluble tumor necrosis factor receptor: enbrel (etanercept) for the treatment of idiopathic pneumonia syndrome after allogeneic stem cell transplantation: blood and marrow transplant clinical trials network protocol. Biol Blood Marrow Transplant 2014;20:858–64.
77. Levine JE, Paczesny S, Mineishi S, et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008;111:2470–5.
78. Yanik GA, Grupp SA, Pulsipher MA, et al. TNF-receptor inhibitor therapy for the treatment of children with idiopathic pneumonia syndrome. A joint Pediatric Blood and Marrow Transplant Consortium and Children’s Oncology Group Study (ASCT0521). Biol Blood Marrow Transplant 2015;21:67–73.
79. Thompson J, Yin Z, D’Souza A, et al. Etanercept and corticosteroid therapy for the treatment of late-onset idiopathic pneumonia syndrome. Biol Blood Marrow Transplant J 2017; 23:1955–60.
1. Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010;303:1617–24.
2. Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004;170:22–48.
4. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med 2006;354:1813–26.
5. Anasetti C, Logan BR, Lee SJ, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 2012;367:1487–96.
6. Giralt S, Ballen K, Rizzo D, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009;15:367–9.
7. Shulman HM, Kleiner D, Lee SJ, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant 2006;12:31–47.
8. Afessa B, Abdulai RM, Kremers WK, et al. Risk factors and outcome of pulmonary complications after autologous hematopoietic stem cell transplant. Chest 2012;141:442–50.
9. Bolwell BJ. Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003;32:853–61.
10. Carlson K, Backlund L, Smedmyr B, et al. Pulmonary function and complications subsequent to autologous bone marrow transplantation. Bone Marrow Transplant 1994;14:805–11.
11. Clark JG, Schwartz DA, Flournoy N, et al. Risk factors for airflow obstruction in recipients of bone marrow transplants. Ann Intern Med 1987;107:648–56.
12. Crawford SW, Fisher L. Predictive value of pulmonary function tests before marrow transplantation. Chest 1992; 101:1257–64.
13. Ghalie R, Szidon JP, Thompson L, et al. Evaluation of pulmonary complications after bone marrow transplantation: the role of pretransplant pulmonary function tests. Bone Marrow Transplant 1992;10:359–65.
14. Ho VT, Weller E, Lee SJ, et al. Prognostic factors for early severe pulmonary complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001;7:223–9.
15. Horak DA, Schmidt GM, Zaia JA, et al. Pretransplant pulmonary function predicts cytomegalovirus-associated interstitial pneumonia following bone marrow transplantation. Chest 1992;102:1484–90.
16. Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, et al. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transplant 2010;16:199–206.
17. White AC, Terrin N, Miller KB, Ryan HF. Impaired respiratory and skeletal muscle strength in patients prior to hematopoietic stem-cell transplantation. Chest 2005;128145–52.
18. Afessa B. Pretransplant pulmonary evaluation of the blood and marrow transplant recipient. Chest 2005;128:8–10.
19. Parimon T, Madtes DK, Au DH, et al. Pretransplant lung function, respiratory failure, and mortality after stem cell transplantation. Am J Respir Crit Care Med 2005;172:384–90.
20. Pavletic SZ, Martin P, Lee SJ, et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006;12:252–66.
21. Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med 2006;144:407–14.
22. Au BK, Gooley TA, Armand P, et al. Reevaluation of the pretransplant assessment of mortality score after allogeneic hematopoietic transplantation. Biol Blood Marrow Transplant 2015;21:848–54.
23. Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005;106:2912–9.
24. Chien JW, Sullivan KM. Carbon monoxide diffusion capacity: how low can you go for hematopoietic cell transplantation eligibility? Biol Blood Marrow Transplant 2009;15: 447–53.
25. Coffey DG, Pollyea DA, Myint H, et al. Adjusting DLCO for Hb and its effects on the Hematopoietic Cell Transplantation-specific Comorbidity Index. Bone Marrow Transplant 2013;48:1253–6.
26. Kasow KA, Krueger J, Srivastava DK, et al. Clinical utility of computed tomography screening of chest, abdomen, and sinuses before hematopoietic stem cell transplantation: the St. Jude experience. Biol Blood Marrow Transplant 2009;15:490–5.
27. Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45: 1259–68.
28. Savani BN, Montero A, Wu C, et al. Prediction and prevention of transplant-related mortality from pulmonary causes after total body irradiation and allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:223–30.
29. Ehlers SL, Gastineau DA, Patten CA, et al. The impact of smoking on outcomes among patients undergoing hematopoietic SCT for the treatment of acute leukemia. Bone Marrow Transplant 2011;46:285–90.
30. Marks DI, Ballen K, Logan BR, et al. The effect of smoking on allogeneic transplant outcomes. Biol Blood Marrow Transplant 2009;15:1277–87.
31. Tran BT, Halperin A, Chien JW. Cigarette smoking and outcomes after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2011;17:1004–11.
32. Lucena CM, Torres A, Rovira M, et al. Pulmonary complications in hematopoietic SCT: a prospective study. Bone Marrow Transplant 2014;49:1293–9.
33. Chi AK, Soubani AO, White AC, Miller KB. An update on pulmonary complications of hematopoietic stem cell transplantation. Chest 2013;144:1913–22.
34. Dunagan DP, Baker AM, Hurd DD, Haponik EF. Bronchoscopic evaluation of pulmonary infiltrates following bone marrow transplantation. Chest 1997;111:135–41.
35. Naeem N, Reed MD, Creger RJ, et al. Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006;37:119–33.
36. Afessa B, Tefferi A, Hoagland HC, et al. Outcome of recipients of bone marrow transplants who require intensive care unit support. Mayo Clin Proc 1992;67:117–22.
37. Parody R, Martino R, de la Camara R, et al. Fungal and viral infections after allogeneic hematopoietic transplantation from unrelated donors in adults: improving outcomes over time. Bone Marrow Transplant 2015;50:274–81.
38. Orasch C, Weisser M, Mertz D, et al. Comparison of infectious complications during induction/consolidation chemotherapy versus allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:521–6.
39. Aguilar-Guisado M, Jimenez-Jambrina M, Espigado I, et al. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 2011;25:E629–38.
40. Palacios G, Hornig M, Cisterna D, et al. Streptococcus pneumoniae coinfection is correlated with the severity of H1N1 pandemic influenza. PLoS One 2009;4:e8540.
41. Hynicka LM, Ensor CR. Prophylaxis and treatment of respiratory syncytial virus in adult immunocompromised patients. Ann Pharmacother 2012;46:558–66.
42. Shah JN, Chemaly RF. Management of RSV infections in adult recipients of hematopoietic stem cell transplantation. Blood 2011;2755–63.
43. Marr KA, Bowden RA. Fungal infections in patients undergoing blood and marrow transplantation. Transpl Infect Dis 1999;1:237–46.
44. Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J Infect Dis 1997;175:1459–66.
45. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7–14.
46. Fisher CE, Stevens AM, Leisenring W, et al. Independent contribution of bronchoalveolar lavage and serum galactomannan in the diagnosis of invasive pulmonary aspergillosis. Transpl Infect Dis 2014;16:505–10.
47. Kojima R, Tateishi U, Kami M, et al. Chest computed tomography of late invasive aspergillosis after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:506–11.
48. Salmeron G, Porcher R, Bergeron A, et al. Persistent poor long-term prognosis of allogeneic hematopoietic stem cell transplant recipients surviving invasive aspergillosis. Haematologica 2012;97:1357–63.
49. McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982;92(10 Pt 1):1140.
50. Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012;54 Suppl 1:S55–60.
51. Klingspor L, Saaedi B, Ljungman P, Szakos A. Epidemiology and outcomes of patients with invasive mould infections: a retrospective observational study from a single centre (2005-2009). Mycoses 2015;58:470–7.
52. Danion F, Aguilar C, Catherinot E, et al. Mucormycosis: new developments in a persistently devastating infection. Semin Respir Crit Care Med 2015;36:692–70.
53. Rano A, Agusti C, Jimenez P, et al. Pulmonary infiltrates in non-HIV immunocompromised patients: a diagnostic approach using non-invasive and bronchoscopic procedures. Thorax 2001;56:379–87.
54. Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med 2008;36:100–7.
55. Jain P, Sandur S, Meli Y, et al. Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 2004;125:712–22.
56. Rano A, Agusti C, Benito N, et al. Prognostic factors of non-HIV immunocompromised patients with pulmonary infiltrates. Chest 2002;122:253–61.
57. Shannon VR, Andersson BS, Lei X, et al. Utility of early versus late fiberoptic bronchoscopy in the evaluation of new pulmonary infiltrates following hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:647–55.
58. Patel NR, Lee PS, Kim JH, et al. The influence of diagnostic bronchoscopy on clinical outcomes comparing adult autologous and allogeneic bone marrow transplant patients. Chest 2005;127:1388–96.
59. Chellapandian D, Lehrnbecher T, Phillips B, et al. Bronchoalveolar lavage and lung biopsy in patients with cancer and hematopoietic stem-cell transplantation recipients: a systematic review and meta-analysis. J Clin Oncol 2015;33:501–9.
60. Carr IM, Koegelenberg CF, von Groote-Bidlingmaier F, et al. Blood loss during flexible bronchoscopy: a prospective observational study. Respiration 2012;84:312–8.
61. Miyamoto M, Onizuka M, Machida S, et al. ACE deletion polymorphism is associated with a high risk of non-infectious pulmonary complications after stem cell transplantation. Int J Hematol 2014;99:175–83.
62. Capizzi SA, Kumar S, Huneke NE, et al. Peri-engraftment respiratory distress syndrome during autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1299–303.
63. Spitzer TR. Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:893–8.
64. Wanko SO, Broadwater G, Folz RJ, Chao NJ. Diffuse alveolar hemorrhage: retrospective review of clinical outcome in allogeneic transplant recipients treated with aminocaproic acid. Biol Blood Marrow Transplant 2006;12:949–53.
65. Metcalf JP, Rennard SI, Reed EC, et al. Corticosteroids as adjunctive therapy for diffuse alveolar hemorrhage associated with bone marrow transplantation. University of Nebraska Medical Center Bone Marrow Transplant Group. Am J Med 1994;96:327–34.
66. Rathi NK, Tanner AR, Dinh A, et al. Low-, medium- and high-dose steroids with or without aminocaproic acid in adult hematopoietic SCT patients with diffuse alveolar hemorrhage. Bone Marrow Transplant 2015;50:420–6.
67. Afessa B, Tefferi A, Litzow MR, Peters SG. Outcome of diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med 2002;166:1364–8.
68. Panoskaltsis-Mortari A, Griese M, Madtes DK, et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am J Respir Crit Care Med 2011;183:1262–79.
69. Clark JG, Hansen JA, Hertz MI, Pet al. NHLBI workshop summary. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Resp Dis 1993;147:1601–6.
70. Vande Vusse LK, Madtes DK. Early onset noninfectious pulmonary syndromes after hematopoietic cell transplantation. Clin Chest Med 2017;38:233–48.
71. Fukuda T, Hackman RC, Guthrie KA, et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003;102:2777–85.
72. Englund JA, Boeckh M, Kuypers J, et al. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med 2006;144:344–9.
73. Seo S, Renaud C, Kuypers JM, et al. Idiopathic pneumonia syndrome after hematopoietic cell transplantation: evidence of occult infectious etiologies. Blood 2015;125:3789–97.
74. Nakane T, Nakamae H, Kamoi H, et al. Prognostic value of serum surfactant protein D level prior to transplant for the development of bronchiolitis obliterans syndrome and idiopathic pneumonia syndrome following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;42:43–9.
75. Gilbert CR, Lerner A, Baram M, Awsare BK. Utility of flexible bronchoscopy in the evaluation of pulmonary infiltrates in the hematopoietic stem cell transplant population—a single center fourteen year experience. Arch Bronconeumol 2013;49:189–95.
76. Yanik GA, Horowitz MM, Weisdorf DJ, et al. Randomized, double-blind, placebo-controlled trial of soluble tumor necrosis factor receptor: enbrel (etanercept) for the treatment of idiopathic pneumonia syndrome after allogeneic stem cell transplantation: blood and marrow transplant clinical trials network protocol. Biol Blood Marrow Transplant 2014;20:858–64.
77. Levine JE, Paczesny S, Mineishi S, et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008;111:2470–5.
78. Yanik GA, Grupp SA, Pulsipher MA, et al. TNF-receptor inhibitor therapy for the treatment of children with idiopathic pneumonia syndrome. A joint Pediatric Blood and Marrow Transplant Consortium and Children’s Oncology Group Study (ASCT0521). Biol Blood Marrow Transplant 2015;21:67–73.
79. Thompson J, Yin Z, D’Souza A, et al. Etanercept and corticosteroid therapy for the treatment of late-onset idiopathic pneumonia syndrome. Biol Blood Marrow Transplant J 2017; 23:1955–60.
A Primary Care Provider’s Guide to Cataract Surgery in the Very Elderly
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.