User login
Combo produces CR/CRis in FLT3-ITD AML

Photo by Bill Branson
A 2-drug combination has shown promise for treating patients with FLT3-ITD acute myeloid leukemia (AML), according to research published in Science Translational Medicine.
Researchers found that omacetaxine mepesuccinate (formerly known as homoharringtonine) exhibits preferential antileukemic activity against FLT3-ITD AML.
Subsequent preclinical experiments revealed that omacetaxine synergizes with sorafenib and other FLT3 inhibitors.
So researchers tested omacetaxine in combination with sorafenib in a phase 2 trial of patients with FLT3-ITD AML.
The combination produced complete responses (CRs) or CRs with incomplete hematologic recovery (CRis) in a majority of patients, and researchers said the treatment was well-tolerated.
Anskar Y. H. Leung, MD, PhD, of The University of Hong Kong, and his colleagues conducted this research.
The team first performed an in vitro screen on AML patient samples to determine their responses to various drugs.
One of the compounds tested, the protein translation inhibitor omacetaxine mepesuccinate, showed strong antileukemic effects against FLT3-ITD AML. In fact, omacetaxine preferentially inhibited the growth of FLT3-ITD cell lines.
The researchers then found that omacetaxine synergizes with sorafenib and other FLT3 inhibitors to suppress leukemia growth in FLT3-ITD AML cell lines.
Omacetaxine and sorafenib in combination also prolonged survival in mouse models of FLT3-ITD AML (mice transplanted with MV4-11 or MOLM-13 cells).
Phase 2 trial
The researchers went on to test omacetaxine and sorafenib in a phase 2 trial. The trial enrolled 24 patients with FLT3-ITD AML and a median age of 50 (range, 21-76).
Most of the patients had relapsed or refractory disease, but 2 were unsuitable for induction chemotherapy because of advanced age and comorbidities.
The patients received omacetaxine and sorafenib continuously until intolerance, disease progression, or allogeneic hematopoietic stem cell transplant (HSCT).
Twenty patients (83.3%) achieved a CR or CRi at a median of 22 days (range, 18-55). Three patients did not respond, and 1 patient experienced a near-CRi—a reduction of blasts without complete clearance.
Fifteen of the responders relapsed, but 3 of these patients received omacetaxine and sorafenib again and achieved a CRi. One patient received re-treatment and failed to achieve a response.
Seven patients proceeded to HSCT after receiving omacetaxine and sorafenib.
At a median follow-up of 7.1 months (range, 2.2 to 20.5), 4 patients were still in CR/CRi (3 patients after HSCT), and 1 patient who had relapsed was still alive.
The remaining 19 patients had died—14 due to relapse, 4 due to non-response (1 after re-treatment), and 1 due to HSCT.
The median leukemia-free survival was 88 days (range, 9-510), and the median overall survival was 228 days (range, 53 to 615).
Adverse events occurring after treatment with omacetaxine and sorafenib included fever (n=14), rash (n=8), hand-foot-skin reactions (n=6), pneumonia (n=2), neutropenic fever (n=1), and bacteremia (n=1).
The researchers said this study validated the principle and clinical relevance of in vitro drug testing and identified a drug combination that might improve the treatment of FLT3-ITD AML. ![]()

Photo by Bill Branson
A 2-drug combination has shown promise for treating patients with FLT3-ITD acute myeloid leukemia (AML), according to research published in Science Translational Medicine.
Researchers found that omacetaxine mepesuccinate (formerly known as homoharringtonine) exhibits preferential antileukemic activity against FLT3-ITD AML.
Subsequent preclinical experiments revealed that omacetaxine synergizes with sorafenib and other FLT3 inhibitors.
So researchers tested omacetaxine in combination with sorafenib in a phase 2 trial of patients with FLT3-ITD AML.
The combination produced complete responses (CRs) or CRs with incomplete hematologic recovery (CRis) in a majority of patients, and researchers said the treatment was well-tolerated.
Anskar Y. H. Leung, MD, PhD, of The University of Hong Kong, and his colleagues conducted this research.
The team first performed an in vitro screen on AML patient samples to determine their responses to various drugs.
One of the compounds tested, the protein translation inhibitor omacetaxine mepesuccinate, showed strong antileukemic effects against FLT3-ITD AML. In fact, omacetaxine preferentially inhibited the growth of FLT3-ITD cell lines.
The researchers then found that omacetaxine synergizes with sorafenib and other FLT3 inhibitors to suppress leukemia growth in FLT3-ITD AML cell lines.
Omacetaxine and sorafenib in combination also prolonged survival in mouse models of FLT3-ITD AML (mice transplanted with MV4-11 or MOLM-13 cells).
Phase 2 trial
The researchers went on to test omacetaxine and sorafenib in a phase 2 trial. The trial enrolled 24 patients with FLT3-ITD AML and a median age of 50 (range, 21-76).
Most of the patients had relapsed or refractory disease, but 2 were unsuitable for induction chemotherapy because of advanced age and comorbidities.
The patients received omacetaxine and sorafenib continuously until intolerance, disease progression, or allogeneic hematopoietic stem cell transplant (HSCT).
Twenty patients (83.3%) achieved a CR or CRi at a median of 22 days (range, 18-55). Three patients did not respond, and 1 patient experienced a near-CRi—a reduction of blasts without complete clearance.
Fifteen of the responders relapsed, but 3 of these patients received omacetaxine and sorafenib again and achieved a CRi. One patient received re-treatment and failed to achieve a response.
Seven patients proceeded to HSCT after receiving omacetaxine and sorafenib.
At a median follow-up of 7.1 months (range, 2.2 to 20.5), 4 patients were still in CR/CRi (3 patients after HSCT), and 1 patient who had relapsed was still alive.
The remaining 19 patients had died—14 due to relapse, 4 due to non-response (1 after re-treatment), and 1 due to HSCT.
The median leukemia-free survival was 88 days (range, 9-510), and the median overall survival was 228 days (range, 53 to 615).
Adverse events occurring after treatment with omacetaxine and sorafenib included fever (n=14), rash (n=8), hand-foot-skin reactions (n=6), pneumonia (n=2), neutropenic fever (n=1), and bacteremia (n=1).
The researchers said this study validated the principle and clinical relevance of in vitro drug testing and identified a drug combination that might improve the treatment of FLT3-ITD AML. ![]()

Photo by Bill Branson
A 2-drug combination has shown promise for treating patients with FLT3-ITD acute myeloid leukemia (AML), according to research published in Science Translational Medicine.
Researchers found that omacetaxine mepesuccinate (formerly known as homoharringtonine) exhibits preferential antileukemic activity against FLT3-ITD AML.
Subsequent preclinical experiments revealed that omacetaxine synergizes with sorafenib and other FLT3 inhibitors.
So researchers tested omacetaxine in combination with sorafenib in a phase 2 trial of patients with FLT3-ITD AML.
The combination produced complete responses (CRs) or CRs with incomplete hematologic recovery (CRis) in a majority of patients, and researchers said the treatment was well-tolerated.
Anskar Y. H. Leung, MD, PhD, of The University of Hong Kong, and his colleagues conducted this research.
The team first performed an in vitro screen on AML patient samples to determine their responses to various drugs.
One of the compounds tested, the protein translation inhibitor omacetaxine mepesuccinate, showed strong antileukemic effects against FLT3-ITD AML. In fact, omacetaxine preferentially inhibited the growth of FLT3-ITD cell lines.
The researchers then found that omacetaxine synergizes with sorafenib and other FLT3 inhibitors to suppress leukemia growth in FLT3-ITD AML cell lines.
Omacetaxine and sorafenib in combination also prolonged survival in mouse models of FLT3-ITD AML (mice transplanted with MV4-11 or MOLM-13 cells).
Phase 2 trial
The researchers went on to test omacetaxine and sorafenib in a phase 2 trial. The trial enrolled 24 patients with FLT3-ITD AML and a median age of 50 (range, 21-76).
Most of the patients had relapsed or refractory disease, but 2 were unsuitable for induction chemotherapy because of advanced age and comorbidities.
The patients received omacetaxine and sorafenib continuously until intolerance, disease progression, or allogeneic hematopoietic stem cell transplant (HSCT).
Twenty patients (83.3%) achieved a CR or CRi at a median of 22 days (range, 18-55). Three patients did not respond, and 1 patient experienced a near-CRi—a reduction of blasts without complete clearance.
Fifteen of the responders relapsed, but 3 of these patients received omacetaxine and sorafenib again and achieved a CRi. One patient received re-treatment and failed to achieve a response.
Seven patients proceeded to HSCT after receiving omacetaxine and sorafenib.
At a median follow-up of 7.1 months (range, 2.2 to 20.5), 4 patients were still in CR/CRi (3 patients after HSCT), and 1 patient who had relapsed was still alive.
The remaining 19 patients had died—14 due to relapse, 4 due to non-response (1 after re-treatment), and 1 due to HSCT.
The median leukemia-free survival was 88 days (range, 9-510), and the median overall survival was 228 days (range, 53 to 615).
Adverse events occurring after treatment with omacetaxine and sorafenib included fever (n=14), rash (n=8), hand-foot-skin reactions (n=6), pneumonia (n=2), neutropenic fever (n=1), and bacteremia (n=1).
The researchers said this study validated the principle and clinical relevance of in vitro drug testing and identified a drug combination that might improve the treatment of FLT3-ITD AML. ![]()
Gene therapy could treat hemophilia A

A new discovery contradicts prevailing assumptions about hemophilia and could change the treatment of hemophilia A, according to researchers.
The team discovered that the protein furin, which is required in factor IX (FIX) replacement, is not required in factor VIII (FVIII) replacement.
In fact, furin impairs clotting in hemophilia A, so gene therapy that can avoid furin processing may provide more effective treatment of hemophilia A.
Experiments in mice and dogs with severe hemophilia A supported this idea. Researchers described the experiments in JCI Insight.
“The clotting factors involved in hemophilia A and hemophilia B are very different, and this has important implications in devising new treatments,” said study author Valder R. Arruda, MD, PhD, of The Children’s Hospital of Philadelphia in Pennsylvania.
Dr Arruda and his colleagues noted that biological differences between FVIII and FIX mean there are obstacles to effective gene therapy in hemophilia A that are not an issue in hemophilia B.
Specifically, the gene encoding FIX is much smaller than the gene for FVIII. So the gene encoding FIX is easier to fit into a vector designed to deliver the therapy to a patient.
“In gene therapy, size matters,” Dr Arruda said. “It’s important to reduce the gene package for FVIII to the smallest effective size.”
He added that, according to his group’s research, deleting the furin-recognition components both decreases the size of the gene therapy payload and strengthens its benefits for treating hemophilia A.
Dr Arruda and his colleagues bioengineered a new variant protein, FVIII-ΔF, which avoids interacting with furin. The team then used that variant in gene therapy experiments in animals with severe hemophilia A.
In mice, FVIII-ΔF gene therapy increased recombinant protein yields, enhanced clotting activity, and produced higher circulating FVIII levels when compared to B-domain-deleted FVIII.
In lab dogs with naturally occurring severe hemophilia A, FVIII-ΔF gene therapy decreased bleeding without triggering a higher level of unwanted immune reactions.
“While much work remains to be done to develop this research into clinical applications, our findings could have a promising translational impact, both for protein replacement and gene therapy,” Dr Arruda said.
“Because this variant provides more efficient bleeding control than currently available replacement drugs, while avoiding immune reactions, this could address the unmet needs of hemophilia A patients worldwide. It may also advance gene therapy for this disorder as well.” ![]()

A new discovery contradicts prevailing assumptions about hemophilia and could change the treatment of hemophilia A, according to researchers.
The team discovered that the protein furin, which is required in factor IX (FIX) replacement, is not required in factor VIII (FVIII) replacement.
In fact, furin impairs clotting in hemophilia A, so gene therapy that can avoid furin processing may provide more effective treatment of hemophilia A.
Experiments in mice and dogs with severe hemophilia A supported this idea. Researchers described the experiments in JCI Insight.
“The clotting factors involved in hemophilia A and hemophilia B are very different, and this has important implications in devising new treatments,” said study author Valder R. Arruda, MD, PhD, of The Children’s Hospital of Philadelphia in Pennsylvania.
Dr Arruda and his colleagues noted that biological differences between FVIII and FIX mean there are obstacles to effective gene therapy in hemophilia A that are not an issue in hemophilia B.
Specifically, the gene encoding FIX is much smaller than the gene for FVIII. So the gene encoding FIX is easier to fit into a vector designed to deliver the therapy to a patient.
“In gene therapy, size matters,” Dr Arruda said. “It’s important to reduce the gene package for FVIII to the smallest effective size.”
He added that, according to his group’s research, deleting the furin-recognition components both decreases the size of the gene therapy payload and strengthens its benefits for treating hemophilia A.
Dr Arruda and his colleagues bioengineered a new variant protein, FVIII-ΔF, which avoids interacting with furin. The team then used that variant in gene therapy experiments in animals with severe hemophilia A.
In mice, FVIII-ΔF gene therapy increased recombinant protein yields, enhanced clotting activity, and produced higher circulating FVIII levels when compared to B-domain-deleted FVIII.
In lab dogs with naturally occurring severe hemophilia A, FVIII-ΔF gene therapy decreased bleeding without triggering a higher level of unwanted immune reactions.
“While much work remains to be done to develop this research into clinical applications, our findings could have a promising translational impact, both for protein replacement and gene therapy,” Dr Arruda said.
“Because this variant provides more efficient bleeding control than currently available replacement drugs, while avoiding immune reactions, this could address the unmet needs of hemophilia A patients worldwide. It may also advance gene therapy for this disorder as well.” ![]()

A new discovery contradicts prevailing assumptions about hemophilia and could change the treatment of hemophilia A, according to researchers.
The team discovered that the protein furin, which is required in factor IX (FIX) replacement, is not required in factor VIII (FVIII) replacement.
In fact, furin impairs clotting in hemophilia A, so gene therapy that can avoid furin processing may provide more effective treatment of hemophilia A.
Experiments in mice and dogs with severe hemophilia A supported this idea. Researchers described the experiments in JCI Insight.
“The clotting factors involved in hemophilia A and hemophilia B are very different, and this has important implications in devising new treatments,” said study author Valder R. Arruda, MD, PhD, of The Children’s Hospital of Philadelphia in Pennsylvania.
Dr Arruda and his colleagues noted that biological differences between FVIII and FIX mean there are obstacles to effective gene therapy in hemophilia A that are not an issue in hemophilia B.
Specifically, the gene encoding FIX is much smaller than the gene for FVIII. So the gene encoding FIX is easier to fit into a vector designed to deliver the therapy to a patient.
“In gene therapy, size matters,” Dr Arruda said. “It’s important to reduce the gene package for FVIII to the smallest effective size.”
He added that, according to his group’s research, deleting the furin-recognition components both decreases the size of the gene therapy payload and strengthens its benefits for treating hemophilia A.
Dr Arruda and his colleagues bioengineered a new variant protein, FVIII-ΔF, which avoids interacting with furin. The team then used that variant in gene therapy experiments in animals with severe hemophilia A.
In mice, FVIII-ΔF gene therapy increased recombinant protein yields, enhanced clotting activity, and produced higher circulating FVIII levels when compared to B-domain-deleted FVIII.
In lab dogs with naturally occurring severe hemophilia A, FVIII-ΔF gene therapy decreased bleeding without triggering a higher level of unwanted immune reactions.
“While much work remains to be done to develop this research into clinical applications, our findings could have a promising translational impact, both for protein replacement and gene therapy,” Dr Arruda said.
“Because this variant provides more efficient bleeding control than currently available replacement drugs, while avoiding immune reactions, this could address the unmet needs of hemophilia A patients worldwide. It may also advance gene therapy for this disorder as well.” ![]()
Work reveals potential therapeutic targets in AML

By adapting CRISPR-Cas9 technology and using it to screen the leukemia genome, researchers have identified hundreds of potential therapeutic targets for acute myeloid leukemia (AML).
The group’s work revealed nearly 500 genes, many of which had not been identified previously, that might serve as targets for AML treatment.
Subsequent experiments showed that targeting one of the genes, KAT2A, can destroy AML cells without harming normal blood cells.
This research was published in Cell Reports.
For this study, the researchers used CRISPR-Cas9 gene-editing technology to screen leukemia cells for vulnerable points. The team said they refined the technology so they could disrupt all genes in the leukemia cell genome individually.
This allowed the researchers to identify those genes whose disruption was detrimental to the growth and survival of AML cells, particularly the AML cell lines MOLM-13, HL-60, OCI-AML2, OCI-AML3, and MV4-11.
“Previous studies showed proof of principle, but this is one of the first systematic attempts to identify the genetic vulnerabilities of AML,” said study author Kosuke Yusa, PhD, of Wellcome Trust Sanger Institute in Hinxton, Cambridge, UK.
“We have improved and applied CRISPR-Cas9 technology to look at what actually kills cells.”
In this way, the researchers identified 492 genes that are essential for AML cell survival, including 227 genes that are druggable.
The team noted that a handful of the genes they identified—including DOT1L, BCL2, and MEN1—are already established therapeutic targets, but most of them are not.
The researchers chose to perform additional experiments with one of the genes they identified, KAT2A, to demonstrate the validity of their findings.
KAT2A was one of 66 genes that were essential to 3 or more of the AML cell lines studied. KAT2A was essential for survival in MOLM-13, OCI-AML2, and OCI-AML3.
The team inhibited KAT2A in vitro using genetic and drug-based techniques. Results showed that disrupting KAT2A inhibited the growth and survival of AML cells but did not affect normal blood cells.
“This is an exciting finding, as KAT2A inhibition worked on a number of primary AML cells with diverse genotypes,” said study author Konstantinos Tzelepis, a PhD student at Wellcome Trust Sanger Institute.
“Whilst the gene needs to be studied in greater depth to understand its potential for use in the clinic, we show that targeting KAT2A destroyed AML cells in the laboratory while sparing healthy blood cells.”
The researchers also targeted KAT2A in transgenic mice. The team observed a significant reduction in AML cell expansion and a significant improvement in survival when KAT2A was disrupted.
“This research has led to the identification of many potential gene targets for future AML therapy, which we are making available to other researchers to explore,” said study author George Vassiliou, PhD, of Wellcome Trust Sanger Institute.
“Whilst KAT2A inhibition now needs to be investigated as a treatment strategy for acute myeloid leukemia, there are many more candidates to pursue by the leukemia research community. Our hope is that this work will lead to more effective treatments against AML that will improve both the survival and the quality of life of patients.” ![]()

By adapting CRISPR-Cas9 technology and using it to screen the leukemia genome, researchers have identified hundreds of potential therapeutic targets for acute myeloid leukemia (AML).
The group’s work revealed nearly 500 genes, many of which had not been identified previously, that might serve as targets for AML treatment.
Subsequent experiments showed that targeting one of the genes, KAT2A, can destroy AML cells without harming normal blood cells.
This research was published in Cell Reports.
For this study, the researchers used CRISPR-Cas9 gene-editing technology to screen leukemia cells for vulnerable points. The team said they refined the technology so they could disrupt all genes in the leukemia cell genome individually.
This allowed the researchers to identify those genes whose disruption was detrimental to the growth and survival of AML cells, particularly the AML cell lines MOLM-13, HL-60, OCI-AML2, OCI-AML3, and MV4-11.
“Previous studies showed proof of principle, but this is one of the first systematic attempts to identify the genetic vulnerabilities of AML,” said study author Kosuke Yusa, PhD, of Wellcome Trust Sanger Institute in Hinxton, Cambridge, UK.
“We have improved and applied CRISPR-Cas9 technology to look at what actually kills cells.”
In this way, the researchers identified 492 genes that are essential for AML cell survival, including 227 genes that are druggable.
The team noted that a handful of the genes they identified—including DOT1L, BCL2, and MEN1—are already established therapeutic targets, but most of them are not.
The researchers chose to perform additional experiments with one of the genes they identified, KAT2A, to demonstrate the validity of their findings.
KAT2A was one of 66 genes that were essential to 3 or more of the AML cell lines studied. KAT2A was essential for survival in MOLM-13, OCI-AML2, and OCI-AML3.
The team inhibited KAT2A in vitro using genetic and drug-based techniques. Results showed that disrupting KAT2A inhibited the growth and survival of AML cells but did not affect normal blood cells.
“This is an exciting finding, as KAT2A inhibition worked on a number of primary AML cells with diverse genotypes,” said study author Konstantinos Tzelepis, a PhD student at Wellcome Trust Sanger Institute.
“Whilst the gene needs to be studied in greater depth to understand its potential for use in the clinic, we show that targeting KAT2A destroyed AML cells in the laboratory while sparing healthy blood cells.”
The researchers also targeted KAT2A in transgenic mice. The team observed a significant reduction in AML cell expansion and a significant improvement in survival when KAT2A was disrupted.
“This research has led to the identification of many potential gene targets for future AML therapy, which we are making available to other researchers to explore,” said study author George Vassiliou, PhD, of Wellcome Trust Sanger Institute.
“Whilst KAT2A inhibition now needs to be investigated as a treatment strategy for acute myeloid leukemia, there are many more candidates to pursue by the leukemia research community. Our hope is that this work will lead to more effective treatments against AML that will improve both the survival and the quality of life of patients.” ![]()

By adapting CRISPR-Cas9 technology and using it to screen the leukemia genome, researchers have identified hundreds of potential therapeutic targets for acute myeloid leukemia (AML).
The group’s work revealed nearly 500 genes, many of which had not been identified previously, that might serve as targets for AML treatment.
Subsequent experiments showed that targeting one of the genes, KAT2A, can destroy AML cells without harming normal blood cells.
This research was published in Cell Reports.
For this study, the researchers used CRISPR-Cas9 gene-editing technology to screen leukemia cells for vulnerable points. The team said they refined the technology so they could disrupt all genes in the leukemia cell genome individually.
This allowed the researchers to identify those genes whose disruption was detrimental to the growth and survival of AML cells, particularly the AML cell lines MOLM-13, HL-60, OCI-AML2, OCI-AML3, and MV4-11.
“Previous studies showed proof of principle, but this is one of the first systematic attempts to identify the genetic vulnerabilities of AML,” said study author Kosuke Yusa, PhD, of Wellcome Trust Sanger Institute in Hinxton, Cambridge, UK.
“We have improved and applied CRISPR-Cas9 technology to look at what actually kills cells.”
In this way, the researchers identified 492 genes that are essential for AML cell survival, including 227 genes that are druggable.
The team noted that a handful of the genes they identified—including DOT1L, BCL2, and MEN1—are already established therapeutic targets, but most of them are not.
The researchers chose to perform additional experiments with one of the genes they identified, KAT2A, to demonstrate the validity of their findings.
KAT2A was one of 66 genes that were essential to 3 or more of the AML cell lines studied. KAT2A was essential for survival in MOLM-13, OCI-AML2, and OCI-AML3.
The team inhibited KAT2A in vitro using genetic and drug-based techniques. Results showed that disrupting KAT2A inhibited the growth and survival of AML cells but did not affect normal blood cells.
“This is an exciting finding, as KAT2A inhibition worked on a number of primary AML cells with diverse genotypes,” said study author Konstantinos Tzelepis, a PhD student at Wellcome Trust Sanger Institute.
“Whilst the gene needs to be studied in greater depth to understand its potential for use in the clinic, we show that targeting KAT2A destroyed AML cells in the laboratory while sparing healthy blood cells.”
The researchers also targeted KAT2A in transgenic mice. The team observed a significant reduction in AML cell expansion and a significant improvement in survival when KAT2A was disrupted.
“This research has led to the identification of many potential gene targets for future AML therapy, which we are making available to other researchers to explore,” said study author George Vassiliou, PhD, of Wellcome Trust Sanger Institute.
“Whilst KAT2A inhibition now needs to be investigated as a treatment strategy for acute myeloid leukemia, there are many more candidates to pursue by the leukemia research community. Our hope is that this work will lead to more effective treatments against AML that will improve both the survival and the quality of life of patients.” ![]()
New nanoparticles may improve chemo delivery

Image from PNAS
A new type of nanoparticle can deliver chemotherapy directly and efficiently to individual cells, according to research published in the Journal of the American Chemical Society.
These nanoparticles, known as connectosomes, are equipped with gap junctions—a pathway that allows for the rapid movement of molecules between 2 cells.
The gap junctions allow the connectosomes to create a direct channel to deliver drugs to each individual cell.
“Gap junctions are the cells’ mechanism for sharing small molecules between neighboring cells,” said study author Jeanne Stachowiak, PhD, of The University of Texas at Austin.
“We believed that there must be a way to utilize them for better drug delivery. The big challenge was in making the materials efficiently and showing that the drugs are delivered through the gap junctions and not some other component.”
To create the connectosomes, the researchers used a chemical process to derive liposomes from donor cells that were engineered to over-produce gap junctions, which are made of proteins.
The team then loaded the connectosomes with the chemotherapy drug doxorubicin.
In in vitro tests with human cells, the researchers found that doxorubicin delivered through connectosomes was 10 times as efficient at killing cancer cells as freely delivered doxorubicin.
Connectosomes were also 100 to 100,000 times as efficient as conventional nanoparticles in delivering doxorubicin, because a drug can diffuse more efficiently through a gap junction than across the oily lipid membrane.
“Connectosomes could open doors for the improved utilization of nanoparticles to deliver other types of therapies,” said Avinash Gadok, a doctoral student at The University of Texas at Austin.
“A huge advantage of nanoparticles is that they can target cells, which helps protect off-target tissues.”
Now, the researchers are investigating whether connectosomes can biochemically target tumor cells and whether they could be useful in inhibiting the migration of tumor cells.
Gap junctions are known to suppress cell migration, creating the potential for connectosomes to help control the movement of tumor cells out of the tumor and into the bloodstream.
“We would like to see whether this approach could delay metastasis while treating the tumor,” Dr Stachowiak said.
“It would be nice to have a multi-pronged approach where you have a particle that slows down metastasis, rapidly delivers drugs, and turns off expression of genes that are promoting the migration of tumor cells.” ![]()

Image from PNAS
A new type of nanoparticle can deliver chemotherapy directly and efficiently to individual cells, according to research published in the Journal of the American Chemical Society.
These nanoparticles, known as connectosomes, are equipped with gap junctions—a pathway that allows for the rapid movement of molecules between 2 cells.
The gap junctions allow the connectosomes to create a direct channel to deliver drugs to each individual cell.
“Gap junctions are the cells’ mechanism for sharing small molecules between neighboring cells,” said study author Jeanne Stachowiak, PhD, of The University of Texas at Austin.
“We believed that there must be a way to utilize them for better drug delivery. The big challenge was in making the materials efficiently and showing that the drugs are delivered through the gap junctions and not some other component.”
To create the connectosomes, the researchers used a chemical process to derive liposomes from donor cells that were engineered to over-produce gap junctions, which are made of proteins.
The team then loaded the connectosomes with the chemotherapy drug doxorubicin.
In in vitro tests with human cells, the researchers found that doxorubicin delivered through connectosomes was 10 times as efficient at killing cancer cells as freely delivered doxorubicin.
Connectosomes were also 100 to 100,000 times as efficient as conventional nanoparticles in delivering doxorubicin, because a drug can diffuse more efficiently through a gap junction than across the oily lipid membrane.
“Connectosomes could open doors for the improved utilization of nanoparticles to deliver other types of therapies,” said Avinash Gadok, a doctoral student at The University of Texas at Austin.
“A huge advantage of nanoparticles is that they can target cells, which helps protect off-target tissues.”
Now, the researchers are investigating whether connectosomes can biochemically target tumor cells and whether they could be useful in inhibiting the migration of tumor cells.
Gap junctions are known to suppress cell migration, creating the potential for connectosomes to help control the movement of tumor cells out of the tumor and into the bloodstream.
“We would like to see whether this approach could delay metastasis while treating the tumor,” Dr Stachowiak said.
“It would be nice to have a multi-pronged approach where you have a particle that slows down metastasis, rapidly delivers drugs, and turns off expression of genes that are promoting the migration of tumor cells.” ![]()

Image from PNAS
A new type of nanoparticle can deliver chemotherapy directly and efficiently to individual cells, according to research published in the Journal of the American Chemical Society.
These nanoparticles, known as connectosomes, are equipped with gap junctions—a pathway that allows for the rapid movement of molecules between 2 cells.
The gap junctions allow the connectosomes to create a direct channel to deliver drugs to each individual cell.
“Gap junctions are the cells’ mechanism for sharing small molecules between neighboring cells,” said study author Jeanne Stachowiak, PhD, of The University of Texas at Austin.
“We believed that there must be a way to utilize them for better drug delivery. The big challenge was in making the materials efficiently and showing that the drugs are delivered through the gap junctions and not some other component.”
To create the connectosomes, the researchers used a chemical process to derive liposomes from donor cells that were engineered to over-produce gap junctions, which are made of proteins.
The team then loaded the connectosomes with the chemotherapy drug doxorubicin.
In in vitro tests with human cells, the researchers found that doxorubicin delivered through connectosomes was 10 times as efficient at killing cancer cells as freely delivered doxorubicin.
Connectosomes were also 100 to 100,000 times as efficient as conventional nanoparticles in delivering doxorubicin, because a drug can diffuse more efficiently through a gap junction than across the oily lipid membrane.
“Connectosomes could open doors for the improved utilization of nanoparticles to deliver other types of therapies,” said Avinash Gadok, a doctoral student at The University of Texas at Austin.
“A huge advantage of nanoparticles is that they can target cells, which helps protect off-target tissues.”
Now, the researchers are investigating whether connectosomes can biochemically target tumor cells and whether they could be useful in inhibiting the migration of tumor cells.
Gap junctions are known to suppress cell migration, creating the potential for connectosomes to help control the movement of tumor cells out of the tumor and into the bloodstream.
“We would like to see whether this approach could delay metastasis while treating the tumor,” Dr Stachowiak said.
“It would be nice to have a multi-pronged approach where you have a particle that slows down metastasis, rapidly delivers drugs, and turns off expression of genes that are promoting the migration of tumor cells.” ![]()
Team explains how MM cells alter BM to thrive
New research helps explain how multiple myeloma (MM) cells manipulate the bone marrow (BM) environment to promote disease progression.
The researchers knew that mesenchymal stem cells (MSCs) are often altered in MM in a way that favors disease progression, but the mechanisms behind this phenomenon weren’t well understood.
So the team set out to determine how and when normal MSCs evolve into tumor-promoting MSCs.
Mahmoud Dabbah, of Meir Medical Center in Kfar Saba, Israel, and his colleagues conducted this research and reported the results in the Journal of Leukocyte Biology.
The researchers cultured the MM cell lines U266 and ARP1 with MSCs from healthy donors, looking for changes in the MSCs. The team observed changes in migration and protein translation initiation.
Specifically, they found that co-culturing MSCs with MM cell lines induced an elevation in translation initiation factors eIF4E and eIF4GI as well as their regulators and targets. But these changes were reversible.
MM-conditioned MSCs had significantly elevated levels of peIF4E, total eIF4E, peIF4GI, and total eIF4GI after 3 days of co-culture with both MM cell lines (all increased about 200%, P<0.05).
In fact, levels of peIF4GI and peIF4E were significantly elevated in the MSCs within 1.5 hours of co-culture (both increased more than 250%, P<0.05).
However, when the MSCs were removed from co-culture, the levels returned to baseline within 3 to 6 hours.
MM-conditioned MSCs also showed a significant increase in migration. When cultured for 16 hours, MSC migration increased more than 400% (P<0.05).
Accordingly, the MM-conditioned MSCs expressed lower levels of microRNAs with established roles in cell migration.
The MSCs showed decreased levels of MIR-125a-5p and MIR-199b-3p after 12 hours of co-culture (a decrease of 160% to 250%, P<0.05). And this effect was maintained as long as the co-culture continued.
The researchers said their findings suggest a dynamic interaction between MM cells and the BM niche that causes profound changes in non-malignant BM constituents. They hope that future studies will reveal clinically relevant means of blocking this crosstalk to improve MM therapy.
“Our research should help identify therapeutic targets that may be used to minimize the collateral damage,” Dabbah said. “The identification of the translation initiation phase as a dialogue platform affords a potential new therapeutic target to be explored.” ![]()

New research helps explain how multiple myeloma (MM) cells manipulate the bone marrow (BM) environment to promote disease progression.
The researchers knew that mesenchymal stem cells (MSCs) are often altered in MM in a way that favors disease progression, but the mechanisms behind this phenomenon weren’t well understood.
So the team set out to determine how and when normal MSCs evolve into tumor-promoting MSCs.
Mahmoud Dabbah, of Meir Medical Center in Kfar Saba, Israel, and his colleagues conducted this research and reported the results in the Journal of Leukocyte Biology.
The researchers cultured the MM cell lines U266 and ARP1 with MSCs from healthy donors, looking for changes in the MSCs. The team observed changes in migration and protein translation initiation.
Specifically, they found that co-culturing MSCs with MM cell lines induced an elevation in translation initiation factors eIF4E and eIF4GI as well as their regulators and targets. But these changes were reversible.
MM-conditioned MSCs had significantly elevated levels of peIF4E, total eIF4E, peIF4GI, and total eIF4GI after 3 days of co-culture with both MM cell lines (all increased about 200%, P<0.05).
In fact, levels of peIF4GI and peIF4E were significantly elevated in the MSCs within 1.5 hours of co-culture (both increased more than 250%, P<0.05).
However, when the MSCs were removed from co-culture, the levels returned to baseline within 3 to 6 hours.
MM-conditioned MSCs also showed a significant increase in migration. When cultured for 16 hours, MSC migration increased more than 400% (P<0.05).
Accordingly, the MM-conditioned MSCs expressed lower levels of microRNAs with established roles in cell migration.
The MSCs showed decreased levels of MIR-125a-5p and MIR-199b-3p after 12 hours of co-culture (a decrease of 160% to 250%, P<0.05). And this effect was maintained as long as the co-culture continued.
The researchers said their findings suggest a dynamic interaction between MM cells and the BM niche that causes profound changes in non-malignant BM constituents. They hope that future studies will reveal clinically relevant means of blocking this crosstalk to improve MM therapy.
“Our research should help identify therapeutic targets that may be used to minimize the collateral damage,” Dabbah said. “The identification of the translation initiation phase as a dialogue platform affords a potential new therapeutic target to be explored.” ![]()

New research helps explain how multiple myeloma (MM) cells manipulate the bone marrow (BM) environment to promote disease progression.
The researchers knew that mesenchymal stem cells (MSCs) are often altered in MM in a way that favors disease progression, but the mechanisms behind this phenomenon weren’t well understood.
So the team set out to determine how and when normal MSCs evolve into tumor-promoting MSCs.
Mahmoud Dabbah, of Meir Medical Center in Kfar Saba, Israel, and his colleagues conducted this research and reported the results in the Journal of Leukocyte Biology.
The researchers cultured the MM cell lines U266 and ARP1 with MSCs from healthy donors, looking for changes in the MSCs. The team observed changes in migration and protein translation initiation.
Specifically, they found that co-culturing MSCs with MM cell lines induced an elevation in translation initiation factors eIF4E and eIF4GI as well as their regulators and targets. But these changes were reversible.
MM-conditioned MSCs had significantly elevated levels of peIF4E, total eIF4E, peIF4GI, and total eIF4GI after 3 days of co-culture with both MM cell lines (all increased about 200%, P<0.05).
In fact, levels of peIF4GI and peIF4E were significantly elevated in the MSCs within 1.5 hours of co-culture (both increased more than 250%, P<0.05).
However, when the MSCs were removed from co-culture, the levels returned to baseline within 3 to 6 hours.
MM-conditioned MSCs also showed a significant increase in migration. When cultured for 16 hours, MSC migration increased more than 400% (P<0.05).
Accordingly, the MM-conditioned MSCs expressed lower levels of microRNAs with established roles in cell migration.
The MSCs showed decreased levels of MIR-125a-5p and MIR-199b-3p after 12 hours of co-culture (a decrease of 160% to 250%, P<0.05). And this effect was maintained as long as the co-culture continued.
The researchers said their findings suggest a dynamic interaction between MM cells and the BM niche that causes profound changes in non-malignant BM constituents. They hope that future studies will reveal clinically relevant means of blocking this crosstalk to improve MM therapy.
“Our research should help identify therapeutic targets that may be used to minimize the collateral damage,” Dabbah said. “The identification of the translation initiation phase as a dialogue platform affords a potential new therapeutic target to be explored.” ![]()
Trial of hemophilia therapy back on

Image courtesy of NIGMS
A UK regulatory agency has lifted the hold placed on a phase 1/2 study of the gene therapy BMN 270 in patients with hemophilia A.
Dosing was suspended in this trial after the first 9 patients were enrolled, but the Medicines and Healthcare Products Regulatory Agency (MHRA) has decided the study can continue.
BioMarin Pharmaceutical Inc., the company developing BMN 270, said the trial should resume enrollment by the end of this year.
BMN 270 is a recombinant adeno-associated virus vector coding for human coagulation factor VIII (FVIII).
The phase 1/2 study of BMN 270 was designed to evaluate the safety and efficacy of the therapy in up to 15 patients with severe hemophilia A.
Results in 7 patients on this study were recently presented at the World Federation of Hemophilia 2016 World Congress.
However, after the study had enrolled 9 patients, dosing of BMN 270 was suspended due to increases in alanine aminotransferase levels that exceeded a pre-specified threshold.
Following the suspension, BioMarin reviewed safety and efficacy data on the 9 patients with the MHRA. Based on this review, the MHRA approved resumption of the study.
The agency also approved the company’s proposed amendments to the study, which included eliminating the requirement for prophylactic corticosteroids and increasing potential additional enrollment from up to 3 additional patients to up to 6 additional patients.
BioMarin said it intends to resume enrollment in the study before the end of 2016. Based on protocol amendments, 3 patients will be enrolled at a dose of 4 x 1013 vg/kg, and an additional 3 patients may be enrolled at this dose or the previously tested high dose of 6 x 1013 vg/kg.
In the up to 6 additional patients, the requirement for prophylactic corticosteroids has been removed, and the threshold for starting therapeutic corticosteroids has been increased.
BioMarin said safety and efficacy data from these patients will inform the phase 2b study expected to begin in the second half of 2017.
“We are pleased that MHRA has approved the resumption of enrollment of the BMN 270 study, as well as the study amendments,” said Hank Fuchs, MD, chief medical officer at BioMarin.
“We believe that the amendments will allow us to optimize the design of a robust phase 2b clinical trial, which potentially could support an accelerated approval by health authorities. We are grateful to the patients who are participating in this current study and are encouraged by the results so far for this phase 1/2 trial.” ![]()

Image courtesy of NIGMS
A UK regulatory agency has lifted the hold placed on a phase 1/2 study of the gene therapy BMN 270 in patients with hemophilia A.
Dosing was suspended in this trial after the first 9 patients were enrolled, but the Medicines and Healthcare Products Regulatory Agency (MHRA) has decided the study can continue.
BioMarin Pharmaceutical Inc., the company developing BMN 270, said the trial should resume enrollment by the end of this year.
BMN 270 is a recombinant adeno-associated virus vector coding for human coagulation factor VIII (FVIII).
The phase 1/2 study of BMN 270 was designed to evaluate the safety and efficacy of the therapy in up to 15 patients with severe hemophilia A.
Results in 7 patients on this study were recently presented at the World Federation of Hemophilia 2016 World Congress.
However, after the study had enrolled 9 patients, dosing of BMN 270 was suspended due to increases in alanine aminotransferase levels that exceeded a pre-specified threshold.
Following the suspension, BioMarin reviewed safety and efficacy data on the 9 patients with the MHRA. Based on this review, the MHRA approved resumption of the study.
The agency also approved the company’s proposed amendments to the study, which included eliminating the requirement for prophylactic corticosteroids and increasing potential additional enrollment from up to 3 additional patients to up to 6 additional patients.
BioMarin said it intends to resume enrollment in the study before the end of 2016. Based on protocol amendments, 3 patients will be enrolled at a dose of 4 x 1013 vg/kg, and an additional 3 patients may be enrolled at this dose or the previously tested high dose of 6 x 1013 vg/kg.
In the up to 6 additional patients, the requirement for prophylactic corticosteroids has been removed, and the threshold for starting therapeutic corticosteroids has been increased.
BioMarin said safety and efficacy data from these patients will inform the phase 2b study expected to begin in the second half of 2017.
“We are pleased that MHRA has approved the resumption of enrollment of the BMN 270 study, as well as the study amendments,” said Hank Fuchs, MD, chief medical officer at BioMarin.
“We believe that the amendments will allow us to optimize the design of a robust phase 2b clinical trial, which potentially could support an accelerated approval by health authorities. We are grateful to the patients who are participating in this current study and are encouraged by the results so far for this phase 1/2 trial.” ![]()

Image courtesy of NIGMS
A UK regulatory agency has lifted the hold placed on a phase 1/2 study of the gene therapy BMN 270 in patients with hemophilia A.
Dosing was suspended in this trial after the first 9 patients were enrolled, but the Medicines and Healthcare Products Regulatory Agency (MHRA) has decided the study can continue.
BioMarin Pharmaceutical Inc., the company developing BMN 270, said the trial should resume enrollment by the end of this year.
BMN 270 is a recombinant adeno-associated virus vector coding for human coagulation factor VIII (FVIII).
The phase 1/2 study of BMN 270 was designed to evaluate the safety and efficacy of the therapy in up to 15 patients with severe hemophilia A.
Results in 7 patients on this study were recently presented at the World Federation of Hemophilia 2016 World Congress.
However, after the study had enrolled 9 patients, dosing of BMN 270 was suspended due to increases in alanine aminotransferase levels that exceeded a pre-specified threshold.
Following the suspension, BioMarin reviewed safety and efficacy data on the 9 patients with the MHRA. Based on this review, the MHRA approved resumption of the study.
The agency also approved the company’s proposed amendments to the study, which included eliminating the requirement for prophylactic corticosteroids and increasing potential additional enrollment from up to 3 additional patients to up to 6 additional patients.
BioMarin said it intends to resume enrollment in the study before the end of 2016. Based on protocol amendments, 3 patients will be enrolled at a dose of 4 x 1013 vg/kg, and an additional 3 patients may be enrolled at this dose or the previously tested high dose of 6 x 1013 vg/kg.
In the up to 6 additional patients, the requirement for prophylactic corticosteroids has been removed, and the threshold for starting therapeutic corticosteroids has been increased.
BioMarin said safety and efficacy data from these patients will inform the phase 2b study expected to begin in the second half of 2017.
“We are pleased that MHRA has approved the resumption of enrollment of the BMN 270 study, as well as the study amendments,” said Hank Fuchs, MD, chief medical officer at BioMarin.
“We believe that the amendments will allow us to optimize the design of a robust phase 2b clinical trial, which potentially could support an accelerated approval by health authorities. We are grateful to the patients who are participating in this current study and are encouraged by the results so far for this phase 1/2 trial.” ![]()
COMP recommends orphan designation for CMV-CTLs

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) is recommending orphan designation for a cytomegalovirus-specific cytotoxic T-lymphocyte product (CMV-CTLs) intended to treat CMV infection in patients with impaired cell-mediated immunity.
The CMV-CTLs are designed to find and kill cells expressing CMV.
To create CMV-CTLs, T cells are collected from the blood of third-party donors and then exposed to CMV antigens.
The resulting activated T cells are then expanded, characterized, and stored for future use in a partially HLA-matched patient.
The CMV-CTLs are being developed by Atara Biotherapeutics, Inc.
The cells are currently under investigation in a pair of phase 2 trials (NCT01646645 and NCT02136797).
Results of a phase 1 trial (published in Biology of Blood and Marrow Transplantation in 2015) suggested CMV-CTLs are safe and can clear CMV infection in patients who have undergone allogeneic hematopoietic stem cell transplant.
The trial included 17 transplant recipients with CMV viremia or clinical infection that persisted despite prolonged treatment with antiviral drugs. Fourteen of the patients had received T-cell-depleted transplants without graft-versus-host disease (GVHD) prophylaxis.
Sixteen of the patients received CMV-CTLs created using cells derived from their transplant donor, and 1 patient received cells from a third-party donor.
Fifteen patients achieved clearance of CMV viremia, including 3 of the 5 patients with overt disease and the patient who received cells from a third-party donor.
In addition, the researchers said CMV-CTLs were well-tolerated. None of the patients experienced fever, alterations in vital signs, or other toxicities during the first 48 hours of observation.
None of the patients developed manifestations of de novo acute GVHD, and GHVD did not worsen in either of the 2 patients who had GVHD prior to infusion.
About orphan designation
The COMP adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the European Commission for a final decision.
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) is recommending orphan designation for a cytomegalovirus-specific cytotoxic T-lymphocyte product (CMV-CTLs) intended to treat CMV infection in patients with impaired cell-mediated immunity.
The CMV-CTLs are designed to find and kill cells expressing CMV.
To create CMV-CTLs, T cells are collected from the blood of third-party donors and then exposed to CMV antigens.
The resulting activated T cells are then expanded, characterized, and stored for future use in a partially HLA-matched patient.
The CMV-CTLs are being developed by Atara Biotherapeutics, Inc.
The cells are currently under investigation in a pair of phase 2 trials (NCT01646645 and NCT02136797).
Results of a phase 1 trial (published in Biology of Blood and Marrow Transplantation in 2015) suggested CMV-CTLs are safe and can clear CMV infection in patients who have undergone allogeneic hematopoietic stem cell transplant.
The trial included 17 transplant recipients with CMV viremia or clinical infection that persisted despite prolonged treatment with antiviral drugs. Fourteen of the patients had received T-cell-depleted transplants without graft-versus-host disease (GVHD) prophylaxis.
Sixteen of the patients received CMV-CTLs created using cells derived from their transplant donor, and 1 patient received cells from a third-party donor.
Fifteen patients achieved clearance of CMV viremia, including 3 of the 5 patients with overt disease and the patient who received cells from a third-party donor.
In addition, the researchers said CMV-CTLs were well-tolerated. None of the patients experienced fever, alterations in vital signs, or other toxicities during the first 48 hours of observation.
None of the patients developed manifestations of de novo acute GVHD, and GHVD did not worsen in either of the 2 patients who had GVHD prior to infusion.
About orphan designation
The COMP adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the European Commission for a final decision.
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) is recommending orphan designation for a cytomegalovirus-specific cytotoxic T-lymphocyte product (CMV-CTLs) intended to treat CMV infection in patients with impaired cell-mediated immunity.
The CMV-CTLs are designed to find and kill cells expressing CMV.
To create CMV-CTLs, T cells are collected from the blood of third-party donors and then exposed to CMV antigens.
The resulting activated T cells are then expanded, characterized, and stored for future use in a partially HLA-matched patient.
The CMV-CTLs are being developed by Atara Biotherapeutics, Inc.
The cells are currently under investigation in a pair of phase 2 trials (NCT01646645 and NCT02136797).
Results of a phase 1 trial (published in Biology of Blood and Marrow Transplantation in 2015) suggested CMV-CTLs are safe and can clear CMV infection in patients who have undergone allogeneic hematopoietic stem cell transplant.
The trial included 17 transplant recipients with CMV viremia or clinical infection that persisted despite prolonged treatment with antiviral drugs. Fourteen of the patients had received T-cell-depleted transplants without graft-versus-host disease (GVHD) prophylaxis.
Sixteen of the patients received CMV-CTLs created using cells derived from their transplant donor, and 1 patient received cells from a third-party donor.
Fifteen patients achieved clearance of CMV viremia, including 3 of the 5 patients with overt disease and the patient who received cells from a third-party donor.
In addition, the researchers said CMV-CTLs were well-tolerated. None of the patients experienced fever, alterations in vital signs, or other toxicities during the first 48 hours of observation.
None of the patients developed manifestations of de novo acute GVHD, and GHVD did not worsen in either of the 2 patients who had GVHD prior to infusion.
About orphan designation
The COMP adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the European Commission for a final decision.
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
Findings could aid treatment of resistant T-ALL
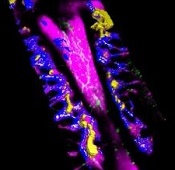
(blue and purple) invaded
by leukemia cells (yellow).
Image courtesy of Edwin
Hawkins, Delfim Duarte,
and Imperial College London
Preclinical research has shed light on how certain leukemia cells survive treatment and could pave the way for better therapeutic targeting of these resistant cells.
Researchers have speculated that some leukemia cells survive treatment by hiding out in specific niches in the bone marrow.
Results of the new research, conducted in mouse models and human samples of T-cell acute lymphoblastic leukemia (T-ALL), contradict that theory.
The experiments showed that resistant T-ALL cells move rapidly through the bone marrow before, during, and after treatment, interacting with—and sometimes killing—healthy cells.
“We expected the cells that survived treatment to be sat in particular niches, but, instead, they are very active throughout the bone marrow,” said Cristina Lo Celso, PhD, of Imperial College London in the UK.
“We now know that it would be ineffective to target particular niches in the bone marrow to tackle treatment-resistant leukemia. Now that we know that the cells don’t hide, we can explore why that is and how their movement helps them to survive. Ultimately, we want to find out whether we can stop the movement and whether this could kill the treatment-resistant cells.”
Dr Lo Celso and her colleagues described these findings in a letter to Nature.
The researchers used intravital microscopy to track the movement of T-ALL cells in mice—before, during, and after treatment. Treatment consisted of dexamethasone alone, vincristine alone, or combination dexamethasone, vincristine, and L-asparaginase.
The team found that T-ALL cells moved around rapidly, not showing any preference for bone marrow subcompartments. The cells’ behavior was consistent over time—from the earliest bone marrow seeding through to treatment response and resistance.
However, the researchers noted that surviving T-ALL cells were “highly migratory” and travelled at significantly faster speeds than early infiltrating cells. In addition, resistant T-ALL cells were still capable of undergoing division at times when other T-ALL cells were dying.
The team suggested that the act of moving may help T-ALL cells to survive, possibly through short-lived interactions with other cells.
This theory was supported by the discovery that T-ALL cells actively attack osteoblasts. The researchers noted that osteoblasts are associated with hematopoietic fitness. So the loss of osteoblasts may contribute to the loss of healthy hematopoiesis observed in leukemia patients.
The team believes this insight could aid the development of treatments to safeguard the production of healthy blood cells in T-ALL patients.
“Our study supports the idea that, at least in this leukemia, new therapies should target the cancer cells themselves instead of the surrounding normal stromal cells to better eradicate the disease,” said study author Delfim Duarte, MD, a PhD student at Imperial College London.
“Our work also suggests that protecting normal stromal bone cells from the attack of leukemia cells can have wide implications in the support of healthy blood cell production,” said Edwin Hawkins, PhD, of the Walter and Eliza Hall Institute of Medical Research in Melbourne, Victoria, Australia.
“Keeping blood cell levels up would prevent anemia, infection, and bleeding.”

(blue and purple) invaded
by leukemia cells (yellow).
Image courtesy of Edwin
Hawkins, Delfim Duarte,
and Imperial College London
Preclinical research has shed light on how certain leukemia cells survive treatment and could pave the way for better therapeutic targeting of these resistant cells.
Researchers have speculated that some leukemia cells survive treatment by hiding out in specific niches in the bone marrow.
Results of the new research, conducted in mouse models and human samples of T-cell acute lymphoblastic leukemia (T-ALL), contradict that theory.
The experiments showed that resistant T-ALL cells move rapidly through the bone marrow before, during, and after treatment, interacting with—and sometimes killing—healthy cells.
“We expected the cells that survived treatment to be sat in particular niches, but, instead, they are very active throughout the bone marrow,” said Cristina Lo Celso, PhD, of Imperial College London in the UK.
“We now know that it would be ineffective to target particular niches in the bone marrow to tackle treatment-resistant leukemia. Now that we know that the cells don’t hide, we can explore why that is and how their movement helps them to survive. Ultimately, we want to find out whether we can stop the movement and whether this could kill the treatment-resistant cells.”
Dr Lo Celso and her colleagues described these findings in a letter to Nature.
The researchers used intravital microscopy to track the movement of T-ALL cells in mice—before, during, and after treatment. Treatment consisted of dexamethasone alone, vincristine alone, or combination dexamethasone, vincristine, and L-asparaginase.
The team found that T-ALL cells moved around rapidly, not showing any preference for bone marrow subcompartments. The cells’ behavior was consistent over time—from the earliest bone marrow seeding through to treatment response and resistance.
However, the researchers noted that surviving T-ALL cells were “highly migratory” and travelled at significantly faster speeds than early infiltrating cells. In addition, resistant T-ALL cells were still capable of undergoing division at times when other T-ALL cells were dying.
The team suggested that the act of moving may help T-ALL cells to survive, possibly through short-lived interactions with other cells.
This theory was supported by the discovery that T-ALL cells actively attack osteoblasts. The researchers noted that osteoblasts are associated with hematopoietic fitness. So the loss of osteoblasts may contribute to the loss of healthy hematopoiesis observed in leukemia patients.
The team believes this insight could aid the development of treatments to safeguard the production of healthy blood cells in T-ALL patients.
“Our study supports the idea that, at least in this leukemia, new therapies should target the cancer cells themselves instead of the surrounding normal stromal cells to better eradicate the disease,” said study author Delfim Duarte, MD, a PhD student at Imperial College London.
“Our work also suggests that protecting normal stromal bone cells from the attack of leukemia cells can have wide implications in the support of healthy blood cell production,” said Edwin Hawkins, PhD, of the Walter and Eliza Hall Institute of Medical Research in Melbourne, Victoria, Australia.
“Keeping blood cell levels up would prevent anemia, infection, and bleeding.”

(blue and purple) invaded
by leukemia cells (yellow).
Image courtesy of Edwin
Hawkins, Delfim Duarte,
and Imperial College London
Preclinical research has shed light on how certain leukemia cells survive treatment and could pave the way for better therapeutic targeting of these resistant cells.
Researchers have speculated that some leukemia cells survive treatment by hiding out in specific niches in the bone marrow.
Results of the new research, conducted in mouse models and human samples of T-cell acute lymphoblastic leukemia (T-ALL), contradict that theory.
The experiments showed that resistant T-ALL cells move rapidly through the bone marrow before, during, and after treatment, interacting with—and sometimes killing—healthy cells.
“We expected the cells that survived treatment to be sat in particular niches, but, instead, they are very active throughout the bone marrow,” said Cristina Lo Celso, PhD, of Imperial College London in the UK.
“We now know that it would be ineffective to target particular niches in the bone marrow to tackle treatment-resistant leukemia. Now that we know that the cells don’t hide, we can explore why that is and how their movement helps them to survive. Ultimately, we want to find out whether we can stop the movement and whether this could kill the treatment-resistant cells.”
Dr Lo Celso and her colleagues described these findings in a letter to Nature.
The researchers used intravital microscopy to track the movement of T-ALL cells in mice—before, during, and after treatment. Treatment consisted of dexamethasone alone, vincristine alone, or combination dexamethasone, vincristine, and L-asparaginase.
The team found that T-ALL cells moved around rapidly, not showing any preference for bone marrow subcompartments. The cells’ behavior was consistent over time—from the earliest bone marrow seeding through to treatment response and resistance.
However, the researchers noted that surviving T-ALL cells were “highly migratory” and travelled at significantly faster speeds than early infiltrating cells. In addition, resistant T-ALL cells were still capable of undergoing division at times when other T-ALL cells were dying.
The team suggested that the act of moving may help T-ALL cells to survive, possibly through short-lived interactions with other cells.
This theory was supported by the discovery that T-ALL cells actively attack osteoblasts. The researchers noted that osteoblasts are associated with hematopoietic fitness. So the loss of osteoblasts may contribute to the loss of healthy hematopoiesis observed in leukemia patients.
The team believes this insight could aid the development of treatments to safeguard the production of healthy blood cells in T-ALL patients.
“Our study supports the idea that, at least in this leukemia, new therapies should target the cancer cells themselves instead of the surrounding normal stromal cells to better eradicate the disease,” said study author Delfim Duarte, MD, a PhD student at Imperial College London.
“Our work also suggests that protecting normal stromal bone cells from the attack of leukemia cells can have wide implications in the support of healthy blood cell production,” said Edwin Hawkins, PhD, of the Walter and Eliza Hall Institute of Medical Research in Melbourne, Victoria, Australia.
“Keeping blood cell levels up would prevent anemia, infection, and bleeding.”
Proteins may be therapeutic targets for AML subtype

Image by Eric Smith
Preclinical research suggests a pair of histone-modifying proteins may be promising therapeutic targets for NPM1-mutated acute myeloid leukemia (AML).
Investigators found that these proteins—MLL and DOT1L—play key roles in NPM1-mutated AML.
Pharmacologic inhibition of either protein alone produced anti-leukemic activity in vitro and in vivo, but inhibiting both proteins together had a more profound effect.
Michael Kühn, MD, of the Mainz University Medical Center in Mainz, Germany, and his colleagues reported these findings in Cancer Discovery.
The investigators noted that nearly all NPM1-mutated AMLs are characterized by aberrant HOX expression, and FLT3 is concomitantly mutated in roughly 60% of these cases. However, it hasn’t been clear how mutant NPM1 cells maintain aberrant gene expression.
With this study, Dr Kühn and his colleagues showed that MLL1 and DOT1L control HOX and FLT3 expression and differentiation in NPM1-mutated AML.
The investigators were able to demonstrate that survival of NPM1-mutated AML cells depends on these 2 proteins. And NPM1-mutated AML is “exceptionally dependent” on the menin binding site in MLL1.
The team tested MI-503, a menin–MLL1 inhibitor, and the DOT1L inhibitor EPZ4777 in human and murine models of NPM1-mutated AML.
Each of the drugs reduced the activity of HOX genes in NPM1-mutated AML cells, but combining the drugs resulted in near-complete inactivation of HOX genes.
When given alone, EPZ4777 and MI-503 each reduced the proliferation and colony-forming potential of NPM1-mutated AML cells in vitro. And each of the drugs prolonged survival in mouse models of NPM1-mutated AML.
However, EPZ4777 and MI-503 given in combination significantly delayed the onset of leukemia and significantly prolonged survival when compared to either drug given alone.
The investigators said this suggests that inhibiting both DOT1L and menin–MLL1 affects leukemia-initiating cells, and this approach represents the first molecularly targeted treatment of NPM1-mutated AML that works by reversing a key mechanism of leukemogenesis.
They added that this research paves the way for trials assessing EPZ4777 and MI-503 in patients with NPM1-mutated AML.

Image by Eric Smith
Preclinical research suggests a pair of histone-modifying proteins may be promising therapeutic targets for NPM1-mutated acute myeloid leukemia (AML).
Investigators found that these proteins—MLL and DOT1L—play key roles in NPM1-mutated AML.
Pharmacologic inhibition of either protein alone produced anti-leukemic activity in vitro and in vivo, but inhibiting both proteins together had a more profound effect.
Michael Kühn, MD, of the Mainz University Medical Center in Mainz, Germany, and his colleagues reported these findings in Cancer Discovery.
The investigators noted that nearly all NPM1-mutated AMLs are characterized by aberrant HOX expression, and FLT3 is concomitantly mutated in roughly 60% of these cases. However, it hasn’t been clear how mutant NPM1 cells maintain aberrant gene expression.
With this study, Dr Kühn and his colleagues showed that MLL1 and DOT1L control HOX and FLT3 expression and differentiation in NPM1-mutated AML.
The investigators were able to demonstrate that survival of NPM1-mutated AML cells depends on these 2 proteins. And NPM1-mutated AML is “exceptionally dependent” on the menin binding site in MLL1.
The team tested MI-503, a menin–MLL1 inhibitor, and the DOT1L inhibitor EPZ4777 in human and murine models of NPM1-mutated AML.
Each of the drugs reduced the activity of HOX genes in NPM1-mutated AML cells, but combining the drugs resulted in near-complete inactivation of HOX genes.
When given alone, EPZ4777 and MI-503 each reduced the proliferation and colony-forming potential of NPM1-mutated AML cells in vitro. And each of the drugs prolonged survival in mouse models of NPM1-mutated AML.
However, EPZ4777 and MI-503 given in combination significantly delayed the onset of leukemia and significantly prolonged survival when compared to either drug given alone.
The investigators said this suggests that inhibiting both DOT1L and menin–MLL1 affects leukemia-initiating cells, and this approach represents the first molecularly targeted treatment of NPM1-mutated AML that works by reversing a key mechanism of leukemogenesis.
They added that this research paves the way for trials assessing EPZ4777 and MI-503 in patients with NPM1-mutated AML.

Image by Eric Smith
Preclinical research suggests a pair of histone-modifying proteins may be promising therapeutic targets for NPM1-mutated acute myeloid leukemia (AML).
Investigators found that these proteins—MLL and DOT1L—play key roles in NPM1-mutated AML.
Pharmacologic inhibition of either protein alone produced anti-leukemic activity in vitro and in vivo, but inhibiting both proteins together had a more profound effect.
Michael Kühn, MD, of the Mainz University Medical Center in Mainz, Germany, and his colleagues reported these findings in Cancer Discovery.
The investigators noted that nearly all NPM1-mutated AMLs are characterized by aberrant HOX expression, and FLT3 is concomitantly mutated in roughly 60% of these cases. However, it hasn’t been clear how mutant NPM1 cells maintain aberrant gene expression.
With this study, Dr Kühn and his colleagues showed that MLL1 and DOT1L control HOX and FLT3 expression and differentiation in NPM1-mutated AML.
The investigators were able to demonstrate that survival of NPM1-mutated AML cells depends on these 2 proteins. And NPM1-mutated AML is “exceptionally dependent” on the menin binding site in MLL1.
The team tested MI-503, a menin–MLL1 inhibitor, and the DOT1L inhibitor EPZ4777 in human and murine models of NPM1-mutated AML.
Each of the drugs reduced the activity of HOX genes in NPM1-mutated AML cells, but combining the drugs resulted in near-complete inactivation of HOX genes.
When given alone, EPZ4777 and MI-503 each reduced the proliferation and colony-forming potential of NPM1-mutated AML cells in vitro. And each of the drugs prolonged survival in mouse models of NPM1-mutated AML.
However, EPZ4777 and MI-503 given in combination significantly delayed the onset of leukemia and significantly prolonged survival when compared to either drug given alone.
The investigators said this suggests that inhibiting both DOT1L and menin–MLL1 affects leukemia-initiating cells, and this approach represents the first molecularly targeted treatment of NPM1-mutated AML that works by reversing a key mechanism of leukemogenesis.
They added that this research paves the way for trials assessing EPZ4777 and MI-503 in patients with NPM1-mutated AML.
CHMP recommends conditional approval of drug for CLL

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion of venetoclax (Venclyxto™).
The CHMP is recommending that venetoclax receive conditional marketing authorization to treat adults with chronic lymphocytic leukemia (CLL) who have 17p deletion or TP53 mutation and are unsuitable for or have failed treatment with an inhibitor of the B-cell receptor pathway.
The CHMP is also recommending the conditional authorization of venetoclax as a treatment for adults with CLL who do not have 17p deletion or TP53 mutation but have failed both chemo immunotherapy and treatment with an inhibitor of the B-cell receptor pathway.
The European Commission (EC) will review the CHMP’s opinion and is expected to make a final decision about venetoclax in late 2016.
If the EC follows the CHMP’s recommendations, venetoclax will become the first BCL-2 inhibitor approved for use in Europe. The authorization will be valid in all member states of the European Union, as well as Iceland, Liechtenstein, and Norway.
Conditional marketing authorization represents an expedited path for approval. The EC grants conditional marketing authorization to products whose benefits are thought to outweigh their risks, products that address unmet needs, and products that are expected to provide a significant public health benefit.
Conditional marketing authorization is granted before pivotal registration studies of a product are completed, but the company developing the product is required to complete post-marketing studies showing that the product provides a clinical benefit.
Venetoclax is being developed by AbbVie and Genentech, a member of the Roche Group. The drug is jointly commercialized by the companies in the US and by AbbVie outside of the US.
Venetoclax is currently approved for use in Argentina, Canada, Puerto Rico, and the US. The drug is being evaluated in phase 3 trials for the treatment of relapsed, refractory, and previously untreated CLL.
Phase 2 trial
Results from a phase 2 trial of venetoclax in CLL (M13-982, NCT01889186) were published in The Lancet Oncology in June. The trial enrolled 107 patients with relapsed or refractory CLL and 17p deletion.
Patients received venetoclax at 400 mg once daily following a weekly ramp-up schedule for the first 5 weeks. The primary endpoint was overall response rate, as determined by an independent review committee.
At a median follow-up of 12.1 months, 85 patients had responded to treatment, for an overall response rate of 79%.
Eight patients (8%) achieved a complete response or complete response with incomplete count recovery, 3 (3%) had a near-partial response, and 74 (69%) had a partial response. Twenty-two patients (21%) did not respond.
At the time of analysis, the median duration of response had not been reached. The same was true for progression-free survival and overall survival. The progression-free survival estimate for 12 months was 72%, and the overall survival estimate was 87%.
The incidence of treatment-emergent adverse events was 96%. The most frequent grade 3/4 adverse events were neutropenia (40%), infection (20%), anemia (18%), and thrombocytopenia (15%).
The incidence of serious adverse events was 55%. The most common of these were pyrexia (7%), autoimmune hemolytic anemia (7%), pneumonia (6%), and febrile neutropenia (5%).
Grade 3 laboratory tumor lysis syndrome (TLS) was reported in 5 patients during the ramp-up period only. Three of these patients continued on venetoclax, but 2 patients required a dose interruption of 1 day each.
In the past, TLS has caused deaths in patients receiving venetoclax. In response, AbbVie stopped dose-escalation in patients receiving the drug and suspended enrollment in phase 1 trials.
However, researchers subsequently found that a modified dosing schedule, prophylaxis, and patient monitoring can reduce the risk of TLS.

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion of venetoclax (Venclyxto™).
The CHMP is recommending that venetoclax receive conditional marketing authorization to treat adults with chronic lymphocytic leukemia (CLL) who have 17p deletion or TP53 mutation and are unsuitable for or have failed treatment with an inhibitor of the B-cell receptor pathway.
The CHMP is also recommending the conditional authorization of venetoclax as a treatment for adults with CLL who do not have 17p deletion or TP53 mutation but have failed both chemo immunotherapy and treatment with an inhibitor of the B-cell receptor pathway.
The European Commission (EC) will review the CHMP’s opinion and is expected to make a final decision about venetoclax in late 2016.
If the EC follows the CHMP’s recommendations, venetoclax will become the first BCL-2 inhibitor approved for use in Europe. The authorization will be valid in all member states of the European Union, as well as Iceland, Liechtenstein, and Norway.
Conditional marketing authorization represents an expedited path for approval. The EC grants conditional marketing authorization to products whose benefits are thought to outweigh their risks, products that address unmet needs, and products that are expected to provide a significant public health benefit.
Conditional marketing authorization is granted before pivotal registration studies of a product are completed, but the company developing the product is required to complete post-marketing studies showing that the product provides a clinical benefit.
Venetoclax is being developed by AbbVie and Genentech, a member of the Roche Group. The drug is jointly commercialized by the companies in the US and by AbbVie outside of the US.
Venetoclax is currently approved for use in Argentina, Canada, Puerto Rico, and the US. The drug is being evaluated in phase 3 trials for the treatment of relapsed, refractory, and previously untreated CLL.
Phase 2 trial
Results from a phase 2 trial of venetoclax in CLL (M13-982, NCT01889186) were published in The Lancet Oncology in June. The trial enrolled 107 patients with relapsed or refractory CLL and 17p deletion.
Patients received venetoclax at 400 mg once daily following a weekly ramp-up schedule for the first 5 weeks. The primary endpoint was overall response rate, as determined by an independent review committee.
At a median follow-up of 12.1 months, 85 patients had responded to treatment, for an overall response rate of 79%.
Eight patients (8%) achieved a complete response or complete response with incomplete count recovery, 3 (3%) had a near-partial response, and 74 (69%) had a partial response. Twenty-two patients (21%) did not respond.
At the time of analysis, the median duration of response had not been reached. The same was true for progression-free survival and overall survival. The progression-free survival estimate for 12 months was 72%, and the overall survival estimate was 87%.
The incidence of treatment-emergent adverse events was 96%. The most frequent grade 3/4 adverse events were neutropenia (40%), infection (20%), anemia (18%), and thrombocytopenia (15%).
The incidence of serious adverse events was 55%. The most common of these were pyrexia (7%), autoimmune hemolytic anemia (7%), pneumonia (6%), and febrile neutropenia (5%).
Grade 3 laboratory tumor lysis syndrome (TLS) was reported in 5 patients during the ramp-up period only. Three of these patients continued on venetoclax, but 2 patients required a dose interruption of 1 day each.
In the past, TLS has caused deaths in patients receiving venetoclax. In response, AbbVie stopped dose-escalation in patients receiving the drug and suspended enrollment in phase 1 trials.
However, researchers subsequently found that a modified dosing schedule, prophylaxis, and patient monitoring can reduce the risk of TLS.

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion of venetoclax (Venclyxto™).
The CHMP is recommending that venetoclax receive conditional marketing authorization to treat adults with chronic lymphocytic leukemia (CLL) who have 17p deletion or TP53 mutation and are unsuitable for or have failed treatment with an inhibitor of the B-cell receptor pathway.
The CHMP is also recommending the conditional authorization of venetoclax as a treatment for adults with CLL who do not have 17p deletion or TP53 mutation but have failed both chemo immunotherapy and treatment with an inhibitor of the B-cell receptor pathway.
The European Commission (EC) will review the CHMP’s opinion and is expected to make a final decision about venetoclax in late 2016.
If the EC follows the CHMP’s recommendations, venetoclax will become the first BCL-2 inhibitor approved for use in Europe. The authorization will be valid in all member states of the European Union, as well as Iceland, Liechtenstein, and Norway.
Conditional marketing authorization represents an expedited path for approval. The EC grants conditional marketing authorization to products whose benefits are thought to outweigh their risks, products that address unmet needs, and products that are expected to provide a significant public health benefit.
Conditional marketing authorization is granted before pivotal registration studies of a product are completed, but the company developing the product is required to complete post-marketing studies showing that the product provides a clinical benefit.
Venetoclax is being developed by AbbVie and Genentech, a member of the Roche Group. The drug is jointly commercialized by the companies in the US and by AbbVie outside of the US.
Venetoclax is currently approved for use in Argentina, Canada, Puerto Rico, and the US. The drug is being evaluated in phase 3 trials for the treatment of relapsed, refractory, and previously untreated CLL.
Phase 2 trial
Results from a phase 2 trial of venetoclax in CLL (M13-982, NCT01889186) were published in The Lancet Oncology in June. The trial enrolled 107 patients with relapsed or refractory CLL and 17p deletion.
Patients received venetoclax at 400 mg once daily following a weekly ramp-up schedule for the first 5 weeks. The primary endpoint was overall response rate, as determined by an independent review committee.
At a median follow-up of 12.1 months, 85 patients had responded to treatment, for an overall response rate of 79%.
Eight patients (8%) achieved a complete response or complete response with incomplete count recovery, 3 (3%) had a near-partial response, and 74 (69%) had a partial response. Twenty-two patients (21%) did not respond.
At the time of analysis, the median duration of response had not been reached. The same was true for progression-free survival and overall survival. The progression-free survival estimate for 12 months was 72%, and the overall survival estimate was 87%.
The incidence of treatment-emergent adverse events was 96%. The most frequent grade 3/4 adverse events were neutropenia (40%), infection (20%), anemia (18%), and thrombocytopenia (15%).
The incidence of serious adverse events was 55%. The most common of these were pyrexia (7%), autoimmune hemolytic anemia (7%), pneumonia (6%), and febrile neutropenia (5%).
Grade 3 laboratory tumor lysis syndrome (TLS) was reported in 5 patients during the ramp-up period only. Three of these patients continued on venetoclax, but 2 patients required a dose interruption of 1 day each.
In the past, TLS has caused deaths in patients receiving venetoclax. In response, AbbVie stopped dose-escalation in patients receiving the drug and suspended enrollment in phase 1 trials.
However, researchers subsequently found that a modified dosing schedule, prophylaxis, and patient monitoring can reduce the risk of TLS.