User login
Team calls for change in warfarin dosing algorithms

Photo courtesy of NIGMS
Investigators have proposed the use of race-stratified algorithms to help clinicians better calculate the appropriate warfarin dose for a patient.
The team’s study, published in Blood, showed that clinical and genetic factors affecting warfarin dose requirements vary by race.
“As the outcomes of disease can vary by race, so can response to medications,” said Nita Limdi, PhD, PharmD, of the University of Alabama at Birmingham.
“Therefore, warfarin dosing equations that combine race groups for analysis (race-adjusted analysis) assume that the effect of variables—such as age and genetics—are the same across race groups, which may compromise dose prediction among patients of both races.”
To better understand how genetics and clinical factors influence warfarin dosing across race groups, Dr Limdi and her colleagues analyzed 1357 patients—595 African American and 762 European American—treated with warfarin.
The team calculated and compared dose recommendations according to both race-adjusted dosing models and race-stratified dosing models. They found that race-stratified analysis improved dose prediction in both racial groups, as compared to race- adjusted analysis.
Race-stratified analysis showed that European Americans with the CYP2C9*2 variant required less warfarin than European Americans with wild-type CYP2C9. But the same was not true for African Americans.
And although all participants who carried VKORC1 required lower doses, regardless of race, the proportional dose reduction was greater among European Americans.
The investigators therefore concluded that the influence of genetic and clinical factors on warfarin dose differs by race. So race-stratified algorithms, rather than race-adjusted algorithms, should be used to guide warfarin dosing.
“Our findings highlight the need for adequate racial representation in warfarin dosing studies to improve our understanding of how the factors that influence warfarin dose differ according to race,” Dr Limdi said. “This is the first step to developing race-specific algorithms to personalize therapy.” ![]()

Photo courtesy of NIGMS
Investigators have proposed the use of race-stratified algorithms to help clinicians better calculate the appropriate warfarin dose for a patient.
The team’s study, published in Blood, showed that clinical and genetic factors affecting warfarin dose requirements vary by race.
“As the outcomes of disease can vary by race, so can response to medications,” said Nita Limdi, PhD, PharmD, of the University of Alabama at Birmingham.
“Therefore, warfarin dosing equations that combine race groups for analysis (race-adjusted analysis) assume that the effect of variables—such as age and genetics—are the same across race groups, which may compromise dose prediction among patients of both races.”
To better understand how genetics and clinical factors influence warfarin dosing across race groups, Dr Limdi and her colleagues analyzed 1357 patients—595 African American and 762 European American—treated with warfarin.
The team calculated and compared dose recommendations according to both race-adjusted dosing models and race-stratified dosing models. They found that race-stratified analysis improved dose prediction in both racial groups, as compared to race- adjusted analysis.
Race-stratified analysis showed that European Americans with the CYP2C9*2 variant required less warfarin than European Americans with wild-type CYP2C9. But the same was not true for African Americans.
And although all participants who carried VKORC1 required lower doses, regardless of race, the proportional dose reduction was greater among European Americans.
The investigators therefore concluded that the influence of genetic and clinical factors on warfarin dose differs by race. So race-stratified algorithms, rather than race-adjusted algorithms, should be used to guide warfarin dosing.
“Our findings highlight the need for adequate racial representation in warfarin dosing studies to improve our understanding of how the factors that influence warfarin dose differ according to race,” Dr Limdi said. “This is the first step to developing race-specific algorithms to personalize therapy.” ![]()

Photo courtesy of NIGMS
Investigators have proposed the use of race-stratified algorithms to help clinicians better calculate the appropriate warfarin dose for a patient.
The team’s study, published in Blood, showed that clinical and genetic factors affecting warfarin dose requirements vary by race.
“As the outcomes of disease can vary by race, so can response to medications,” said Nita Limdi, PhD, PharmD, of the University of Alabama at Birmingham.
“Therefore, warfarin dosing equations that combine race groups for analysis (race-adjusted analysis) assume that the effect of variables—such as age and genetics—are the same across race groups, which may compromise dose prediction among patients of both races.”
To better understand how genetics and clinical factors influence warfarin dosing across race groups, Dr Limdi and her colleagues analyzed 1357 patients—595 African American and 762 European American—treated with warfarin.
The team calculated and compared dose recommendations according to both race-adjusted dosing models and race-stratified dosing models. They found that race-stratified analysis improved dose prediction in both racial groups, as compared to race- adjusted analysis.
Race-stratified analysis showed that European Americans with the CYP2C9*2 variant required less warfarin than European Americans with wild-type CYP2C9. But the same was not true for African Americans.
And although all participants who carried VKORC1 required lower doses, regardless of race, the proportional dose reduction was greater among European Americans.
The investigators therefore concluded that the influence of genetic and clinical factors on warfarin dose differs by race. So race-stratified algorithms, rather than race-adjusted algorithms, should be used to guide warfarin dosing.
“Our findings highlight the need for adequate racial representation in warfarin dosing studies to improve our understanding of how the factors that influence warfarin dose differ according to race,” Dr Limdi said. “This is the first step to developing race-specific algorithms to personalize therapy.” ![]()
Hodgkin lymphoma incidence on the decline worldwide

Photo courtesy of NIH
In trying to estimate the global cancer burden, researchers found that cases of Hodgkin lymphoma (HL) have decreased worldwide over the last 2 decades.
The team studied 28 cancer types in 188 countries, and HL was the only malignancy whose incidence decreased from 1990 to 2013.
And the number of HL deaths in 2013 was comparatively low. When the researchers ranked cancers according to the number of global deaths, HL was 26th on the list of 28.
The researchers disclosed these results in JAMA Oncology.
The team collected data from cancer registries, vital records, verbal autopsy reports, and other sources to estimate the global cancer burden.
The data suggested that, in 2013, there were 14.9 million new cancer cases and 8.2 million cancer deaths worldwide. The proportion of cancer deaths as part of all deaths increased from 12% in 1990 to 15% in 2013.
The most common malignancy in men was prostate cancer, with 1.4 million cases in 2013. For women, it was breast cancer, with 1.8 million cases in 2013.
Tracheal, bronchus, and lung cancers were the leading cause of cancer death in men and women, with 1.6 million deaths in 2013.
Hematologic malignancies
Globally, the age-standardized incidence of HL per 100,000 people decreased by 34% during the time period studied. Cases of HL fell from about 103,000 in 1990 to 93,000 in 2013.
When the researchers ranked cancer types according to the number of global deaths in 2013, HL came in 26th. There were about 24,000 HL deaths in 2013—14,000 among men and 10,000 among women.
Non-Hodgkin lymphoma (NHL) came in 11th for global cancer deaths in 2013. There were about 226,000 NHL deaths—133,000 among men and 92,000 among women.
In addition, the incidence of NHL more than doubled from 1990 to 2013, rising from about 227,000 to 465,000. According to 2013 data, 1 in 103 men and 1 in 151 women developed NHL between birth and 79 years of age.
The researchers observed an increase in cases of multiple myeloma (MM) as well, from about 63,000 in 1990 to 117,000 in 2013.
In 2013, there were about 79,000 MM deaths—42,000 among men and 37,000 among women. MM ranked 19th on the list of global cancer deaths in 2013.
Leukemia ranked 9th on the list. There were about 265,000 leukemia deaths in 2013—149,000 among men and 116,000 among women.
Cases of leukemia increased from 297,000 in 1990 to 414,000 in 2013. According to 2013 data, 1 in 127 men and 1 in 203 women developed leukemia between birth and 79 years of age.
This research shows that cancer remains a major threat to people’s health around the world, said study author Christina Fitzmaurice, MD, of the University of Washington in Seattle.
“Cancer prevention, screening, and treatment programs are costly,” she noted, “and it is very important for countries to know which cancers cause the highest disease burden in order to allocate scarce resources appropriately.” ![]()

Photo courtesy of NIH
In trying to estimate the global cancer burden, researchers found that cases of Hodgkin lymphoma (HL) have decreased worldwide over the last 2 decades.
The team studied 28 cancer types in 188 countries, and HL was the only malignancy whose incidence decreased from 1990 to 2013.
And the number of HL deaths in 2013 was comparatively low. When the researchers ranked cancers according to the number of global deaths, HL was 26th on the list of 28.
The researchers disclosed these results in JAMA Oncology.
The team collected data from cancer registries, vital records, verbal autopsy reports, and other sources to estimate the global cancer burden.
The data suggested that, in 2013, there were 14.9 million new cancer cases and 8.2 million cancer deaths worldwide. The proportion of cancer deaths as part of all deaths increased from 12% in 1990 to 15% in 2013.
The most common malignancy in men was prostate cancer, with 1.4 million cases in 2013. For women, it was breast cancer, with 1.8 million cases in 2013.
Tracheal, bronchus, and lung cancers were the leading cause of cancer death in men and women, with 1.6 million deaths in 2013.
Hematologic malignancies
Globally, the age-standardized incidence of HL per 100,000 people decreased by 34% during the time period studied. Cases of HL fell from about 103,000 in 1990 to 93,000 in 2013.
When the researchers ranked cancer types according to the number of global deaths in 2013, HL came in 26th. There were about 24,000 HL deaths in 2013—14,000 among men and 10,000 among women.
Non-Hodgkin lymphoma (NHL) came in 11th for global cancer deaths in 2013. There were about 226,000 NHL deaths—133,000 among men and 92,000 among women.
In addition, the incidence of NHL more than doubled from 1990 to 2013, rising from about 227,000 to 465,000. According to 2013 data, 1 in 103 men and 1 in 151 women developed NHL between birth and 79 years of age.
The researchers observed an increase in cases of multiple myeloma (MM) as well, from about 63,000 in 1990 to 117,000 in 2013.
In 2013, there were about 79,000 MM deaths—42,000 among men and 37,000 among women. MM ranked 19th on the list of global cancer deaths in 2013.
Leukemia ranked 9th on the list. There were about 265,000 leukemia deaths in 2013—149,000 among men and 116,000 among women.
Cases of leukemia increased from 297,000 in 1990 to 414,000 in 2013. According to 2013 data, 1 in 127 men and 1 in 203 women developed leukemia between birth and 79 years of age.
This research shows that cancer remains a major threat to people’s health around the world, said study author Christina Fitzmaurice, MD, of the University of Washington in Seattle.
“Cancer prevention, screening, and treatment programs are costly,” she noted, “and it is very important for countries to know which cancers cause the highest disease burden in order to allocate scarce resources appropriately.” ![]()

Photo courtesy of NIH
In trying to estimate the global cancer burden, researchers found that cases of Hodgkin lymphoma (HL) have decreased worldwide over the last 2 decades.
The team studied 28 cancer types in 188 countries, and HL was the only malignancy whose incidence decreased from 1990 to 2013.
And the number of HL deaths in 2013 was comparatively low. When the researchers ranked cancers according to the number of global deaths, HL was 26th on the list of 28.
The researchers disclosed these results in JAMA Oncology.
The team collected data from cancer registries, vital records, verbal autopsy reports, and other sources to estimate the global cancer burden.
The data suggested that, in 2013, there were 14.9 million new cancer cases and 8.2 million cancer deaths worldwide. The proportion of cancer deaths as part of all deaths increased from 12% in 1990 to 15% in 2013.
The most common malignancy in men was prostate cancer, with 1.4 million cases in 2013. For women, it was breast cancer, with 1.8 million cases in 2013.
Tracheal, bronchus, and lung cancers were the leading cause of cancer death in men and women, with 1.6 million deaths in 2013.
Hematologic malignancies
Globally, the age-standardized incidence of HL per 100,000 people decreased by 34% during the time period studied. Cases of HL fell from about 103,000 in 1990 to 93,000 in 2013.
When the researchers ranked cancer types according to the number of global deaths in 2013, HL came in 26th. There were about 24,000 HL deaths in 2013—14,000 among men and 10,000 among women.
Non-Hodgkin lymphoma (NHL) came in 11th for global cancer deaths in 2013. There were about 226,000 NHL deaths—133,000 among men and 92,000 among women.
In addition, the incidence of NHL more than doubled from 1990 to 2013, rising from about 227,000 to 465,000. According to 2013 data, 1 in 103 men and 1 in 151 women developed NHL between birth and 79 years of age.
The researchers observed an increase in cases of multiple myeloma (MM) as well, from about 63,000 in 1990 to 117,000 in 2013.
In 2013, there were about 79,000 MM deaths—42,000 among men and 37,000 among women. MM ranked 19th on the list of global cancer deaths in 2013.
Leukemia ranked 9th on the list. There were about 265,000 leukemia deaths in 2013—149,000 among men and 116,000 among women.
Cases of leukemia increased from 297,000 in 1990 to 414,000 in 2013. According to 2013 data, 1 in 127 men and 1 in 203 women developed leukemia between birth and 79 years of age.
This research shows that cancer remains a major threat to people’s health around the world, said study author Christina Fitzmaurice, MD, of the University of Washington in Seattle.
“Cancer prevention, screening, and treatment programs are costly,” she noted, “and it is very important for countries to know which cancers cause the highest disease burden in order to allocate scarce resources appropriately.” ![]()
DART molecule proves active against AML

Photo by Sakurai Midori
An artificial antibody that redirects T cells to target cancer cells shows promise for treating acute myeloid leukemia (AML), according to preclinical research.
The antibody, MGD006, induced tumor regression in mouse models of AML and was largely well-tolerated in cynomolgus monkeys.
Investigators say these results support clinical testing of MGD006 in AML, which is currently underway.
MGD006 is a humanized, dual-affinity re-targeting (DART) molecule that combines a portion of an antibody recognizing CD3, an activating molecule expressed by T cells, with an arm that recognizes CD123.
MGD006 redirects T cells to kill cells expressing CD123, which is upregulated in AML and other hematologic diseases.
Gurunadh Chichili, PhD, of MacroGenics, Inc., in Rockville, Maryland, and his colleagues described their work with MGD006 in Science Translational Medicine. MacroGenics, the company developing MGD006, funded this research.
Because MGD006 is designed to be cleared rapidly, it requires continuous delivery. So in mice, the investigators administered the molecule continuously for up to a week via peritoneally implanted osmotic pumps.
The NSG/b2m−/− mice had been reconstituted with human peripheral blood mononuclear cells and grafted with KG-1a cells, an AML-M0 line. The mice received MGD006 after tumors were allowed to grow to an average size of about 100 mm3.
Treated mice experienced significant tumor regression at all doses of MGD006 (P<0.005), but there was no activity in mice treated with a control DART molecule. The investigators found that 500 ng/kg of MGD006 per day was sufficient to completely eliminate leukemic cells.
The team also tested MGD006 in macaques and found the molecule binds to human and cynomolgus monkey antigens with similar affinities and redirects T cells from either species to kill CD123-expressing target cells.
The monkeys received continuous infusions of MGD006, starting at 0.1 mg/kg per day and escalating weekly to up to 1 mg/kg per day for a 4-week period. The treatment depleted circulating CD123-positive cells beginning at 72 hours and continuing throughout the infusion period.
The monkeys experienced cytokine release, but it was transient and most significant after the first dose of MDG006. After the first dose, IL-6 concentration returned to baseline by 72 hours, and the magnitude of IL-6 response decreased with each successive MGD006 infusion, even when the dose was increased.
The animals experienced reversible reductions in hematocrit and red cell mass at the highest doses of MDG006 but no neutropenia or thrombocytopenia.
“This research paved the way for our initiation of a phase 1 clinical study of MGD006 in 2014,” said Scott Koenig, MD, PhD, President and CEO of MacroGenics.
“MGD006 has demonstrated great promise as a T-cell-redirected cancer immunotherapy in preclinical studies. We are hopeful that these studies will translate into clinical trial results indicative of clinical improvement for patients with AML, myelodysplastic syndrome, and several other forms of leukemia and lymphoma.” ![]()

Photo by Sakurai Midori
An artificial antibody that redirects T cells to target cancer cells shows promise for treating acute myeloid leukemia (AML), according to preclinical research.
The antibody, MGD006, induced tumor regression in mouse models of AML and was largely well-tolerated in cynomolgus monkeys.
Investigators say these results support clinical testing of MGD006 in AML, which is currently underway.
MGD006 is a humanized, dual-affinity re-targeting (DART) molecule that combines a portion of an antibody recognizing CD3, an activating molecule expressed by T cells, with an arm that recognizes CD123.
MGD006 redirects T cells to kill cells expressing CD123, which is upregulated in AML and other hematologic diseases.
Gurunadh Chichili, PhD, of MacroGenics, Inc., in Rockville, Maryland, and his colleagues described their work with MGD006 in Science Translational Medicine. MacroGenics, the company developing MGD006, funded this research.
Because MGD006 is designed to be cleared rapidly, it requires continuous delivery. So in mice, the investigators administered the molecule continuously for up to a week via peritoneally implanted osmotic pumps.
The NSG/b2m−/− mice had been reconstituted with human peripheral blood mononuclear cells and grafted with KG-1a cells, an AML-M0 line. The mice received MGD006 after tumors were allowed to grow to an average size of about 100 mm3.
Treated mice experienced significant tumor regression at all doses of MGD006 (P<0.005), but there was no activity in mice treated with a control DART molecule. The investigators found that 500 ng/kg of MGD006 per day was sufficient to completely eliminate leukemic cells.
The team also tested MGD006 in macaques and found the molecule binds to human and cynomolgus monkey antigens with similar affinities and redirects T cells from either species to kill CD123-expressing target cells.
The monkeys received continuous infusions of MGD006, starting at 0.1 mg/kg per day and escalating weekly to up to 1 mg/kg per day for a 4-week period. The treatment depleted circulating CD123-positive cells beginning at 72 hours and continuing throughout the infusion period.
The monkeys experienced cytokine release, but it was transient and most significant after the first dose of MDG006. After the first dose, IL-6 concentration returned to baseline by 72 hours, and the magnitude of IL-6 response decreased with each successive MGD006 infusion, even when the dose was increased.
The animals experienced reversible reductions in hematocrit and red cell mass at the highest doses of MDG006 but no neutropenia or thrombocytopenia.
“This research paved the way for our initiation of a phase 1 clinical study of MGD006 in 2014,” said Scott Koenig, MD, PhD, President and CEO of MacroGenics.
“MGD006 has demonstrated great promise as a T-cell-redirected cancer immunotherapy in preclinical studies. We are hopeful that these studies will translate into clinical trial results indicative of clinical improvement for patients with AML, myelodysplastic syndrome, and several other forms of leukemia and lymphoma.” ![]()

Photo by Sakurai Midori
An artificial antibody that redirects T cells to target cancer cells shows promise for treating acute myeloid leukemia (AML), according to preclinical research.
The antibody, MGD006, induced tumor regression in mouse models of AML and was largely well-tolerated in cynomolgus monkeys.
Investigators say these results support clinical testing of MGD006 in AML, which is currently underway.
MGD006 is a humanized, dual-affinity re-targeting (DART) molecule that combines a portion of an antibody recognizing CD3, an activating molecule expressed by T cells, with an arm that recognizes CD123.
MGD006 redirects T cells to kill cells expressing CD123, which is upregulated in AML and other hematologic diseases.
Gurunadh Chichili, PhD, of MacroGenics, Inc., in Rockville, Maryland, and his colleagues described their work with MGD006 in Science Translational Medicine. MacroGenics, the company developing MGD006, funded this research.
Because MGD006 is designed to be cleared rapidly, it requires continuous delivery. So in mice, the investigators administered the molecule continuously for up to a week via peritoneally implanted osmotic pumps.
The NSG/b2m−/− mice had been reconstituted with human peripheral blood mononuclear cells and grafted with KG-1a cells, an AML-M0 line. The mice received MGD006 after tumors were allowed to grow to an average size of about 100 mm3.
Treated mice experienced significant tumor regression at all doses of MGD006 (P<0.005), but there was no activity in mice treated with a control DART molecule. The investigators found that 500 ng/kg of MGD006 per day was sufficient to completely eliminate leukemic cells.
The team also tested MGD006 in macaques and found the molecule binds to human and cynomolgus monkey antigens with similar affinities and redirects T cells from either species to kill CD123-expressing target cells.
The monkeys received continuous infusions of MGD006, starting at 0.1 mg/kg per day and escalating weekly to up to 1 mg/kg per day for a 4-week period. The treatment depleted circulating CD123-positive cells beginning at 72 hours and continuing throughout the infusion period.
The monkeys experienced cytokine release, but it was transient and most significant after the first dose of MDG006. After the first dose, IL-6 concentration returned to baseline by 72 hours, and the magnitude of IL-6 response decreased with each successive MGD006 infusion, even when the dose was increased.
The animals experienced reversible reductions in hematocrit and red cell mass at the highest doses of MDG006 but no neutropenia or thrombocytopenia.
“This research paved the way for our initiation of a phase 1 clinical study of MGD006 in 2014,” said Scott Koenig, MD, PhD, President and CEO of MacroGenics.
“MGD006 has demonstrated great promise as a T-cell-redirected cancer immunotherapy in preclinical studies. We are hopeful that these studies will translate into clinical trial results indicative of clinical improvement for patients with AML, myelodysplastic syndrome, and several other forms of leukemia and lymphoma.” ![]()
Urine test could reduce need for blood samples

Photo by Juan D. Alfonso
A new approach to urine testing could make the tests more versatile and therefore decrease the need for blood tests, according to researchers.
They believe the method could also reduce costs, produce results faster than current tests, and lower the volume of urine needed for a sample.
R. Kenneth Marcus, PhD, of Clemson University in South Carolina, and his colleagues described this method in Proteomics-Clinical Applications.
Dr Marcus noted that the trouble with testing urine is that it’s awash in salt, so it can be tricky to isolate the proteins that act as biomarkers.
To overcome this problem, he and his colleagues used a string made of capillary-channeled polymer fibers. The team packed the fibers into plastic tubes and then passed urine samples through the tubes by spinning them in a centrifuge for 30 seconds.
Then, the researchers ran de-ionized water through the tubes for a minute to wash off salt and other contaminants.
As proteins are hydrophobic, they remained stuck to the fibers. The team extracted the proteins by running a solvent through the tubes in the centrifuge for 30 seconds.
When this process was complete, the researchers were left with purified proteins that could be stored in a plastic vial and refrigerated until testing time.
The team was able to extract 12 samples in about 5 minutes, limited only by centrifuge capacity.
In addition to being faster and cheaper than current urine tests, the new testing method should also make it easier to test urine samples from infants, Dr Marcus said.
One of the challenges now is getting a large enough sample, but the new method requires only a few microliters of urine. ![]()

Photo by Juan D. Alfonso
A new approach to urine testing could make the tests more versatile and therefore decrease the need for blood tests, according to researchers.
They believe the method could also reduce costs, produce results faster than current tests, and lower the volume of urine needed for a sample.
R. Kenneth Marcus, PhD, of Clemson University in South Carolina, and his colleagues described this method in Proteomics-Clinical Applications.
Dr Marcus noted that the trouble with testing urine is that it’s awash in salt, so it can be tricky to isolate the proteins that act as biomarkers.
To overcome this problem, he and his colleagues used a string made of capillary-channeled polymer fibers. The team packed the fibers into plastic tubes and then passed urine samples through the tubes by spinning them in a centrifuge for 30 seconds.
Then, the researchers ran de-ionized water through the tubes for a minute to wash off salt and other contaminants.
As proteins are hydrophobic, they remained stuck to the fibers. The team extracted the proteins by running a solvent through the tubes in the centrifuge for 30 seconds.
When this process was complete, the researchers were left with purified proteins that could be stored in a plastic vial and refrigerated until testing time.
The team was able to extract 12 samples in about 5 minutes, limited only by centrifuge capacity.
In addition to being faster and cheaper than current urine tests, the new testing method should also make it easier to test urine samples from infants, Dr Marcus said.
One of the challenges now is getting a large enough sample, but the new method requires only a few microliters of urine. ![]()

Photo by Juan D. Alfonso
A new approach to urine testing could make the tests more versatile and therefore decrease the need for blood tests, according to researchers.
They believe the method could also reduce costs, produce results faster than current tests, and lower the volume of urine needed for a sample.
R. Kenneth Marcus, PhD, of Clemson University in South Carolina, and his colleagues described this method in Proteomics-Clinical Applications.
Dr Marcus noted that the trouble with testing urine is that it’s awash in salt, so it can be tricky to isolate the proteins that act as biomarkers.
To overcome this problem, he and his colleagues used a string made of capillary-channeled polymer fibers. The team packed the fibers into plastic tubes and then passed urine samples through the tubes by spinning them in a centrifuge for 30 seconds.
Then, the researchers ran de-ionized water through the tubes for a minute to wash off salt and other contaminants.
As proteins are hydrophobic, they remained stuck to the fibers. The team extracted the proteins by running a solvent through the tubes in the centrifuge for 30 seconds.
When this process was complete, the researchers were left with purified proteins that could be stored in a plastic vial and refrigerated until testing time.
The team was able to extract 12 samples in about 5 minutes, limited only by centrifuge capacity.
In addition to being faster and cheaper than current urine tests, the new testing method should also make it easier to test urine samples from infants, Dr Marcus said.
One of the challenges now is getting a large enough sample, but the new method requires only a few microliters of urine. ![]()
Understanding taste dysfunction in cancer
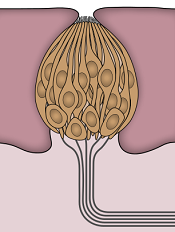
Image by Jonas Töle
Investigators have identified a molecular pathway that aids the renewal of taste buds, and they believe this discovery may have implications for cancer patients who suffer from an altered sense of taste during treatment.
“Taste dysfunction can . . . result from an alteration of the renewal capacities of taste buds and is often associated with psychological distress and malnutrition,” said Dany Gaillard, PhD, of the University of Colorado Anschutz Medical Campus in Aurora.
He and his colleagues decided to investigate this dysfunction using mouse models, and the group reported their findings in PLOS Genetics.
The investigators discovered that a protein in the Wnt pathway, ß-catenin, controls the renewal of taste cells by regulating separate stages of taste-cell turnover.
Previous research showed that Wnt/β-catenin signaling is crucial in developing taste buds in embryos and regulating the renewal of epithelial tissue in adults, including skin, hair follicles, the intestine, and the mouth.
“We show that activating this pathway directs the newly born cells to become, primarily, a specific taste- cell type whose role is to support the other taste cells and help them work efficiently,” said Linda Barlow, PhD, also of the University of Colorado Anschutz Medical Campus.
As chemotherapy destroys dividing precursor cells, including those that produce taste cells, the investigators believe that activating Wnt signaling may be a way to renew taste buds after chemotherapy.
New small-molecule drugs that specifically block the Wnt pathway are under development, and Drs Gaillard and Barlow predict these drugs could also cause taste dysfunction.
Dr Barlow said more research is needed to understand how taste is altered at the cellular level, but this research holds promise for developing new ways to improve cancer patients’ quality of life. ![]()

Image by Jonas Töle
Investigators have identified a molecular pathway that aids the renewal of taste buds, and they believe this discovery may have implications for cancer patients who suffer from an altered sense of taste during treatment.
“Taste dysfunction can . . . result from an alteration of the renewal capacities of taste buds and is often associated with psychological distress and malnutrition,” said Dany Gaillard, PhD, of the University of Colorado Anschutz Medical Campus in Aurora.
He and his colleagues decided to investigate this dysfunction using mouse models, and the group reported their findings in PLOS Genetics.
The investigators discovered that a protein in the Wnt pathway, ß-catenin, controls the renewal of taste cells by regulating separate stages of taste-cell turnover.
Previous research showed that Wnt/β-catenin signaling is crucial in developing taste buds in embryos and regulating the renewal of epithelial tissue in adults, including skin, hair follicles, the intestine, and the mouth.
“We show that activating this pathway directs the newly born cells to become, primarily, a specific taste- cell type whose role is to support the other taste cells and help them work efficiently,” said Linda Barlow, PhD, also of the University of Colorado Anschutz Medical Campus.
As chemotherapy destroys dividing precursor cells, including those that produce taste cells, the investigators believe that activating Wnt signaling may be a way to renew taste buds after chemotherapy.
New small-molecule drugs that specifically block the Wnt pathway are under development, and Drs Gaillard and Barlow predict these drugs could also cause taste dysfunction.
Dr Barlow said more research is needed to understand how taste is altered at the cellular level, but this research holds promise for developing new ways to improve cancer patients’ quality of life. ![]()

Image by Jonas Töle
Investigators have identified a molecular pathway that aids the renewal of taste buds, and they believe this discovery may have implications for cancer patients who suffer from an altered sense of taste during treatment.
“Taste dysfunction can . . . result from an alteration of the renewal capacities of taste buds and is often associated with psychological distress and malnutrition,” said Dany Gaillard, PhD, of the University of Colorado Anschutz Medical Campus in Aurora.
He and his colleagues decided to investigate this dysfunction using mouse models, and the group reported their findings in PLOS Genetics.
The investigators discovered that a protein in the Wnt pathway, ß-catenin, controls the renewal of taste cells by regulating separate stages of taste-cell turnover.
Previous research showed that Wnt/β-catenin signaling is crucial in developing taste buds in embryos and regulating the renewal of epithelial tissue in adults, including skin, hair follicles, the intestine, and the mouth.
“We show that activating this pathway directs the newly born cells to become, primarily, a specific taste- cell type whose role is to support the other taste cells and help them work efficiently,” said Linda Barlow, PhD, also of the University of Colorado Anschutz Medical Campus.
As chemotherapy destroys dividing precursor cells, including those that produce taste cells, the investigators believe that activating Wnt signaling may be a way to renew taste buds after chemotherapy.
New small-molecule drugs that specifically block the Wnt pathway are under development, and Drs Gaillard and Barlow predict these drugs could also cause taste dysfunction.
Dr Barlow said more research is needed to understand how taste is altered at the cellular level, but this research holds promise for developing new ways to improve cancer patients’ quality of life. ![]()
Bridge therapy may be detrimental for some VTE patients

Anticoagulant bridge therapy may be harmful for patients with a low to moderate risk of venous thromboembolism (VTE), according to research published in JAMA Internal Medicine.
The study included more than 1000 patients with VTE who had to stop receiving warfarin prior to a procedure.
Investigators compared outcomes in patients who received bridge therapy—a short-acting anticoagulant during the periprocedural period—and patients who did not.
Nathan Clark, PharmD, of Kaiser Permanente Colorado in Aurora, and his colleagues conducted this research.
They examined the electronic medical records of 1178 patients with VTE who underwent 1812 invasive diagnostic or surgical procedures between January 2006 and March 2012 that required the interruption of warfarin.
The investigators divided patients into 3 groups based on their annual risk of VTE recurrence without anticoagulant therapy. Seventy-nine percent of patients were categorized as low-risk, 17.9% as moderate-risk, and 3.1% as high-risk.
The team then divided patients according to the use of bridge therapy. Of the 1812 procedures, 555 included bridge therapy, and 1257 did not.
Patients in the bridged group had a significantly higher incidence of clinically relevant bleeding at 30 days than patients in the non-bridged group—2.7% and 0.2%, respectively (P=0.01).
When the investigators assessed patients according to VTE risk, they found the increased incidence of bleeding in the bridged group was significant among low-risk patients (2.0% vs 0.1%, P<0.001) and moderate-risk patients (4.6% vs 0%, P=0.004) but not high-risk patients (5.6% vs 4.8%, P=0.90).
On the other hand, there was no significant difference in VTE recurrence between the bridged and non-bridged groups—for all risk categories (0 vs 3 cases, P=0.56), low-risk patients (0 vs 2, P=0.37), moderate-risk patients (0 vs 1, P=0.48), or high-risk patients (0 vs 0, P>0.99)
The investigators said further research is needed to identify patient and procedure-related characteristics associated with the highest risk for perioperative VTE recurrence where targeted bridge therapy may be beneficial. ![]()

Anticoagulant bridge therapy may be harmful for patients with a low to moderate risk of venous thromboembolism (VTE), according to research published in JAMA Internal Medicine.
The study included more than 1000 patients with VTE who had to stop receiving warfarin prior to a procedure.
Investigators compared outcomes in patients who received bridge therapy—a short-acting anticoagulant during the periprocedural period—and patients who did not.
Nathan Clark, PharmD, of Kaiser Permanente Colorado in Aurora, and his colleagues conducted this research.
They examined the electronic medical records of 1178 patients with VTE who underwent 1812 invasive diagnostic or surgical procedures between January 2006 and March 2012 that required the interruption of warfarin.
The investigators divided patients into 3 groups based on their annual risk of VTE recurrence without anticoagulant therapy. Seventy-nine percent of patients were categorized as low-risk, 17.9% as moderate-risk, and 3.1% as high-risk.
The team then divided patients according to the use of bridge therapy. Of the 1812 procedures, 555 included bridge therapy, and 1257 did not.
Patients in the bridged group had a significantly higher incidence of clinically relevant bleeding at 30 days than patients in the non-bridged group—2.7% and 0.2%, respectively (P=0.01).
When the investigators assessed patients according to VTE risk, they found the increased incidence of bleeding in the bridged group was significant among low-risk patients (2.0% vs 0.1%, P<0.001) and moderate-risk patients (4.6% vs 0%, P=0.004) but not high-risk patients (5.6% vs 4.8%, P=0.90).
On the other hand, there was no significant difference in VTE recurrence between the bridged and non-bridged groups—for all risk categories (0 vs 3 cases, P=0.56), low-risk patients (0 vs 2, P=0.37), moderate-risk patients (0 vs 1, P=0.48), or high-risk patients (0 vs 0, P>0.99)
The investigators said further research is needed to identify patient and procedure-related characteristics associated with the highest risk for perioperative VTE recurrence where targeted bridge therapy may be beneficial. ![]()

Anticoagulant bridge therapy may be harmful for patients with a low to moderate risk of venous thromboembolism (VTE), according to research published in JAMA Internal Medicine.
The study included more than 1000 patients with VTE who had to stop receiving warfarin prior to a procedure.
Investigators compared outcomes in patients who received bridge therapy—a short-acting anticoagulant during the periprocedural period—and patients who did not.
Nathan Clark, PharmD, of Kaiser Permanente Colorado in Aurora, and his colleagues conducted this research.
They examined the electronic medical records of 1178 patients with VTE who underwent 1812 invasive diagnostic or surgical procedures between January 2006 and March 2012 that required the interruption of warfarin.
The investigators divided patients into 3 groups based on their annual risk of VTE recurrence without anticoagulant therapy. Seventy-nine percent of patients were categorized as low-risk, 17.9% as moderate-risk, and 3.1% as high-risk.
The team then divided patients according to the use of bridge therapy. Of the 1812 procedures, 555 included bridge therapy, and 1257 did not.
Patients in the bridged group had a significantly higher incidence of clinically relevant bleeding at 30 days than patients in the non-bridged group—2.7% and 0.2%, respectively (P=0.01).
When the investigators assessed patients according to VTE risk, they found the increased incidence of bleeding in the bridged group was significant among low-risk patients (2.0% vs 0.1%, P<0.001) and moderate-risk patients (4.6% vs 0%, P=0.004) but not high-risk patients (5.6% vs 4.8%, P=0.90).
On the other hand, there was no significant difference in VTE recurrence between the bridged and non-bridged groups—for all risk categories (0 vs 3 cases, P=0.56), low-risk patients (0 vs 2, P=0.37), moderate-risk patients (0 vs 1, P=0.48), or high-risk patients (0 vs 0, P>0.99)
The investigators said further research is needed to identify patient and procedure-related characteristics associated with the highest risk for perioperative VTE recurrence where targeted bridge therapy may be beneficial. ![]()
Reducing distress in caregivers of HSCT recipients

Photo by Chad McNeeley
Research has shown that caring for cancer patients after hematopoietic stem cell transplant (HSCT) can have negative psychological effects on the caregiver, but results of a new study suggest a psychosocial intervention could change that.
The trial showed that counseling sessions focused on stress management could significantly reduce stress, anxiety, depression, and mood disturbance among these caregivers.
“The first 100 days after a stem cell transplant is a critical period for patients, in which caregivers are called upon to deliver around-the-clock care, providing support for patients’ everyday needs and also patients’ emotional health, but who takes care of the caregivers?” asked Mark Laudenslager, PhD, of the University of Colorado Denver.
To address this problem, Dr Laudenslager and his colleagues studied 148 caregivers of patients who underwent allogeneic HSCT. The team described this research in Bone Marrow Transplantation.
The caregivers were randomized to a group that was offered a psychosocial intervention (n=74) and a group that received standard treatment, in which mental health support services were available but not required (n=74).
In the experimental group, caregivers attended 8 sessions on stress management. These one-on-one sessions focused on understanding stress and its physical consequences, changing roles as caregivers, cognitive behavioral stress management, pacing respiration, and identifying social support. The researchers call this intervention PsychoEducation, Paced Respiration and Relaxation (PEPRR).
After a patient underwent HSCT, Dr Laudenslager and his colleagues used several questionnaires to follow the trajectory of caregiver distress over time. The questionnaires were used to measure stress, depression, anxiety, mood disturbance, sleep quality, and other mental health outcomes.
There was no significant difference in stress or other mental health measures between the 2 treatment groups at baseline.
However, at 3 months after transplant, caregivers in the PEPRR group saw some significant improvements over caregivers in the standard treatment group.
The PEPRR group had less stress according to the Perceived Stress Scale (P=0.039), less depression according to the Center for Epidemiologic Studies Depression test (P=0.016), less anxiety according to the State-Trait Anxiety Inventory-State questionnaire (P=0.0009), and less mood disturbance according to the Profile of Mood States-Total Mood Disturbance test (P=0.039).
Overall caregiver distress (composite scores from the questionnaires) was significantly lower in the PEPRR group than the standard treatment group (P=0.019).
However, there was no significant difference in caregiver well-being (composite scores) or scores on the Caregiver Reaction Assessment, Pittsburgh Sleep Quality Index, Short Form 36 Health Survey, or Impact of Events scale.
Still, the other improvements caregivers experienced suggest PEPRR is a promising intervention, Dr Laudenslager said.
He and his colleagues are now recruiting subjects for a follow-up study (NCT02037568) focused on evaluating quality of life in allogeneic HSCT recipients whose caregivers participate in programs similar to the PEPRR intervention. ![]()

Photo by Chad McNeeley
Research has shown that caring for cancer patients after hematopoietic stem cell transplant (HSCT) can have negative psychological effects on the caregiver, but results of a new study suggest a psychosocial intervention could change that.
The trial showed that counseling sessions focused on stress management could significantly reduce stress, anxiety, depression, and mood disturbance among these caregivers.
“The first 100 days after a stem cell transplant is a critical period for patients, in which caregivers are called upon to deliver around-the-clock care, providing support for patients’ everyday needs and also patients’ emotional health, but who takes care of the caregivers?” asked Mark Laudenslager, PhD, of the University of Colorado Denver.
To address this problem, Dr Laudenslager and his colleagues studied 148 caregivers of patients who underwent allogeneic HSCT. The team described this research in Bone Marrow Transplantation.
The caregivers were randomized to a group that was offered a psychosocial intervention (n=74) and a group that received standard treatment, in which mental health support services were available but not required (n=74).
In the experimental group, caregivers attended 8 sessions on stress management. These one-on-one sessions focused on understanding stress and its physical consequences, changing roles as caregivers, cognitive behavioral stress management, pacing respiration, and identifying social support. The researchers call this intervention PsychoEducation, Paced Respiration and Relaxation (PEPRR).
After a patient underwent HSCT, Dr Laudenslager and his colleagues used several questionnaires to follow the trajectory of caregiver distress over time. The questionnaires were used to measure stress, depression, anxiety, mood disturbance, sleep quality, and other mental health outcomes.
There was no significant difference in stress or other mental health measures between the 2 treatment groups at baseline.
However, at 3 months after transplant, caregivers in the PEPRR group saw some significant improvements over caregivers in the standard treatment group.
The PEPRR group had less stress according to the Perceived Stress Scale (P=0.039), less depression according to the Center for Epidemiologic Studies Depression test (P=0.016), less anxiety according to the State-Trait Anxiety Inventory-State questionnaire (P=0.0009), and less mood disturbance according to the Profile of Mood States-Total Mood Disturbance test (P=0.039).
Overall caregiver distress (composite scores from the questionnaires) was significantly lower in the PEPRR group than the standard treatment group (P=0.019).
However, there was no significant difference in caregiver well-being (composite scores) or scores on the Caregiver Reaction Assessment, Pittsburgh Sleep Quality Index, Short Form 36 Health Survey, or Impact of Events scale.
Still, the other improvements caregivers experienced suggest PEPRR is a promising intervention, Dr Laudenslager said.
He and his colleagues are now recruiting subjects for a follow-up study (NCT02037568) focused on evaluating quality of life in allogeneic HSCT recipients whose caregivers participate in programs similar to the PEPRR intervention. ![]()

Photo by Chad McNeeley
Research has shown that caring for cancer patients after hematopoietic stem cell transplant (HSCT) can have negative psychological effects on the caregiver, but results of a new study suggest a psychosocial intervention could change that.
The trial showed that counseling sessions focused on stress management could significantly reduce stress, anxiety, depression, and mood disturbance among these caregivers.
“The first 100 days after a stem cell transplant is a critical period for patients, in which caregivers are called upon to deliver around-the-clock care, providing support for patients’ everyday needs and also patients’ emotional health, but who takes care of the caregivers?” asked Mark Laudenslager, PhD, of the University of Colorado Denver.
To address this problem, Dr Laudenslager and his colleagues studied 148 caregivers of patients who underwent allogeneic HSCT. The team described this research in Bone Marrow Transplantation.
The caregivers were randomized to a group that was offered a psychosocial intervention (n=74) and a group that received standard treatment, in which mental health support services were available but not required (n=74).
In the experimental group, caregivers attended 8 sessions on stress management. These one-on-one sessions focused on understanding stress and its physical consequences, changing roles as caregivers, cognitive behavioral stress management, pacing respiration, and identifying social support. The researchers call this intervention PsychoEducation, Paced Respiration and Relaxation (PEPRR).
After a patient underwent HSCT, Dr Laudenslager and his colleagues used several questionnaires to follow the trajectory of caregiver distress over time. The questionnaires were used to measure stress, depression, anxiety, mood disturbance, sleep quality, and other mental health outcomes.
There was no significant difference in stress or other mental health measures between the 2 treatment groups at baseline.
However, at 3 months after transplant, caregivers in the PEPRR group saw some significant improvements over caregivers in the standard treatment group.
The PEPRR group had less stress according to the Perceived Stress Scale (P=0.039), less depression according to the Center for Epidemiologic Studies Depression test (P=0.016), less anxiety according to the State-Trait Anxiety Inventory-State questionnaire (P=0.0009), and less mood disturbance according to the Profile of Mood States-Total Mood Disturbance test (P=0.039).
Overall caregiver distress (composite scores from the questionnaires) was significantly lower in the PEPRR group than the standard treatment group (P=0.019).
However, there was no significant difference in caregiver well-being (composite scores) or scores on the Caregiver Reaction Assessment, Pittsburgh Sleep Quality Index, Short Form 36 Health Survey, or Impact of Events scale.
Still, the other improvements caregivers experienced suggest PEPRR is a promising intervention, Dr Laudenslager said.
He and his colleagues are now recruiting subjects for a follow-up study (NCT02037568) focused on evaluating quality of life in allogeneic HSCT recipients whose caregivers participate in programs similar to the PEPRR intervention.
CAR T-cell therapy appears feasible in HL

LONDON—Results of a small, phase 1 trial suggest CD30-directed chimeric antigen receptor (CAR) T-cell therapy is feasible in patients with aggressive Hodgkin lymphoma (HL).
The trial included 7 patients with relapsed or refractory HL.
Five of the patients achieved stable disease or better after infusions of CAR T cells, and the researchers said treatment-related adverse events were manageable.
William (Wei) Cao, PhD, of Cellular Biomedicine Group, presented these results at the 10th Annual World Stem Cells & Regenerative Medicine Congress.
The research was funded by Cellular Biomedicine Group, the company developing the CAR T-cell therapy (known as CBM-C30.1), as well as by grants from the National Natural Science Foundation of China and the National Basic Science and Development Program of China.
The trial included 7 patients with progressive HL. Two patients had stage III disease, and 5 had stage IV. The patients had a median of 16 prior treatments (range, 8-24) and limited prognosis (several months to less than 2-year survival) with currently available therapies.
The patients received escalating doses of autologous T cells transduced with a CD30-directed CAR moiety for 3 to 5 days, following a conditioning regimen. The researchers measured the level of CAR transgenes in peripheral blood and biopsied tumor tissues by quantitative PCR.
Two patients achieved a partial response to CAR T-cell therapy, and 3 attained stable disease. So the therapy resulted in an overall disease control rate of 71.4% (5/7) and an objective response rate of 28.6% (2/7).
Stable disease lasted 2 months in 2 of the patients and more than 3.5 months in the third patient. Partial response lasted more than 2 months in 1 patient and more than 3.5 months in the other.
Dr Cao said adverse events consisted largely of fever and were manageable with medical intervention. One patient experienced 5-day self-limiting arthralgia, myalgia, and dual knee swelling 2 weeks after cell infusion. There were no delayed or severe adverse events.
“We are very encouraged by the efficacy and toxicity profile of our CAR-T CD30 technology,” Dr Cao said, “given that the [patients] were diagnosed with stage III and IV Hodgkin’s lymphoma.”

LONDON—Results of a small, phase 1 trial suggest CD30-directed chimeric antigen receptor (CAR) T-cell therapy is feasible in patients with aggressive Hodgkin lymphoma (HL).
The trial included 7 patients with relapsed or refractory HL.
Five of the patients achieved stable disease or better after infusions of CAR T cells, and the researchers said treatment-related adverse events were manageable.
William (Wei) Cao, PhD, of Cellular Biomedicine Group, presented these results at the 10th Annual World Stem Cells & Regenerative Medicine Congress.
The research was funded by Cellular Biomedicine Group, the company developing the CAR T-cell therapy (known as CBM-C30.1), as well as by grants from the National Natural Science Foundation of China and the National Basic Science and Development Program of China.
The trial included 7 patients with progressive HL. Two patients had stage III disease, and 5 had stage IV. The patients had a median of 16 prior treatments (range, 8-24) and limited prognosis (several months to less than 2-year survival) with currently available therapies.
The patients received escalating doses of autologous T cells transduced with a CD30-directed CAR moiety for 3 to 5 days, following a conditioning regimen. The researchers measured the level of CAR transgenes in peripheral blood and biopsied tumor tissues by quantitative PCR.
Two patients achieved a partial response to CAR T-cell therapy, and 3 attained stable disease. So the therapy resulted in an overall disease control rate of 71.4% (5/7) and an objective response rate of 28.6% (2/7).
Stable disease lasted 2 months in 2 of the patients and more than 3.5 months in the third patient. Partial response lasted more than 2 months in 1 patient and more than 3.5 months in the other.
Dr Cao said adverse events consisted largely of fever and were manageable with medical intervention. One patient experienced 5-day self-limiting arthralgia, myalgia, and dual knee swelling 2 weeks after cell infusion. There were no delayed or severe adverse events.
“We are very encouraged by the efficacy and toxicity profile of our CAR-T CD30 technology,” Dr Cao said, “given that the [patients] were diagnosed with stage III and IV Hodgkin’s lymphoma.”

LONDON—Results of a small, phase 1 trial suggest CD30-directed chimeric antigen receptor (CAR) T-cell therapy is feasible in patients with aggressive Hodgkin lymphoma (HL).
The trial included 7 patients with relapsed or refractory HL.
Five of the patients achieved stable disease or better after infusions of CAR T cells, and the researchers said treatment-related adverse events were manageable.
William (Wei) Cao, PhD, of Cellular Biomedicine Group, presented these results at the 10th Annual World Stem Cells & Regenerative Medicine Congress.
The research was funded by Cellular Biomedicine Group, the company developing the CAR T-cell therapy (known as CBM-C30.1), as well as by grants from the National Natural Science Foundation of China and the National Basic Science and Development Program of China.
The trial included 7 patients with progressive HL. Two patients had stage III disease, and 5 had stage IV. The patients had a median of 16 prior treatments (range, 8-24) and limited prognosis (several months to less than 2-year survival) with currently available therapies.
The patients received escalating doses of autologous T cells transduced with a CD30-directed CAR moiety for 3 to 5 days, following a conditioning regimen. The researchers measured the level of CAR transgenes in peripheral blood and biopsied tumor tissues by quantitative PCR.
Two patients achieved a partial response to CAR T-cell therapy, and 3 attained stable disease. So the therapy resulted in an overall disease control rate of 71.4% (5/7) and an objective response rate of 28.6% (2/7).
Stable disease lasted 2 months in 2 of the patients and more than 3.5 months in the third patient. Partial response lasted more than 2 months in 1 patient and more than 3.5 months in the other.
Dr Cao said adverse events consisted largely of fever and were manageable with medical intervention. One patient experienced 5-day self-limiting arthralgia, myalgia, and dual knee swelling 2 weeks after cell infusion. There were no delayed or severe adverse events.
“We are very encouraged by the efficacy and toxicity profile of our CAR-T CD30 technology,” Dr Cao said, “given that the [patients] were diagnosed with stage III and IV Hodgkin’s lymphoma.”
PBM program improves outcomes, study shows

Photo by Elise Amendola
A patient blood management (PBM) program can reduce transfusion use, cut costs, and improve outcomes in cardiac surgery patients, according to a single-center study.
A PBM program instituted at Eastern Maine Medical Center (EMMC) in Bangor substantially decreased the use of blood products, the loss of red blood cells, the length of hospital stays, the incidence of acute kidney injury, and direct costs.
Irwin Gross, MD, of EMMC, and his colleagues reported these results in Transfusion.
The team compared clinical and transfusion data from cardiac surgery patients treated at the center before the PBM program began (July 2006-March 2007) and after (April 2007-September 2012).
EMMC’s PBM initiative involved pre- and post-operative anemia management, a more restrictive transfusion threshold, the use of single-unit transfusions when necessary, and other measures.
The researchers analyzed data on 2662 patients, 387 treated before the PBM program began and 2275 treated after.
As expected, the rate of transfusions decreased after the PBM program began. The rate of red blood cell transfusion decreased from 39.3% to 20.8% (P<0.001), the rate of fresh-frozen plasma transfusion decreased from 18.3% to 6.5% (P<0.001), and the rate of platelet transfusion decreased from 17.8% to 9.8% (P<0.001).
Red blood cell loss decreased from a median of 721 mL to 552 mL (P<0.001), and pre-transfusion hemoglobin decreased from a mean of 7.2 ± 1.4 g/dL to 6.6 ± 1.2 g/dL (P<0.001).
Patients saw a decrease in the incidence of post-operative kidney injury from 7.6% to 5.0% (P=0.039) and a decrease in the median length of hospital stay from 10 days to 8 days (P<0.001).
Total adjusted direct costs decreased after the program began as well, falling from a median of $39,709 to $36,906 (P< 0.001).
There was no significant difference in the rate of hospital mortality or the incidence of cerebral vascular accident before and after the PBM program began.

Photo by Elise Amendola
A patient blood management (PBM) program can reduce transfusion use, cut costs, and improve outcomes in cardiac surgery patients, according to a single-center study.
A PBM program instituted at Eastern Maine Medical Center (EMMC) in Bangor substantially decreased the use of blood products, the loss of red blood cells, the length of hospital stays, the incidence of acute kidney injury, and direct costs.
Irwin Gross, MD, of EMMC, and his colleagues reported these results in Transfusion.
The team compared clinical and transfusion data from cardiac surgery patients treated at the center before the PBM program began (July 2006-March 2007) and after (April 2007-September 2012).
EMMC’s PBM initiative involved pre- and post-operative anemia management, a more restrictive transfusion threshold, the use of single-unit transfusions when necessary, and other measures.
The researchers analyzed data on 2662 patients, 387 treated before the PBM program began and 2275 treated after.
As expected, the rate of transfusions decreased after the PBM program began. The rate of red blood cell transfusion decreased from 39.3% to 20.8% (P<0.001), the rate of fresh-frozen plasma transfusion decreased from 18.3% to 6.5% (P<0.001), and the rate of platelet transfusion decreased from 17.8% to 9.8% (P<0.001).
Red blood cell loss decreased from a median of 721 mL to 552 mL (P<0.001), and pre-transfusion hemoglobin decreased from a mean of 7.2 ± 1.4 g/dL to 6.6 ± 1.2 g/dL (P<0.001).
Patients saw a decrease in the incidence of post-operative kidney injury from 7.6% to 5.0% (P=0.039) and a decrease in the median length of hospital stay from 10 days to 8 days (P<0.001).
Total adjusted direct costs decreased after the program began as well, falling from a median of $39,709 to $36,906 (P< 0.001).
There was no significant difference in the rate of hospital mortality or the incidence of cerebral vascular accident before and after the PBM program began.

Photo by Elise Amendola
A patient blood management (PBM) program can reduce transfusion use, cut costs, and improve outcomes in cardiac surgery patients, according to a single-center study.
A PBM program instituted at Eastern Maine Medical Center (EMMC) in Bangor substantially decreased the use of blood products, the loss of red blood cells, the length of hospital stays, the incidence of acute kidney injury, and direct costs.
Irwin Gross, MD, of EMMC, and his colleagues reported these results in Transfusion.
The team compared clinical and transfusion data from cardiac surgery patients treated at the center before the PBM program began (July 2006-March 2007) and after (April 2007-September 2012).
EMMC’s PBM initiative involved pre- and post-operative anemia management, a more restrictive transfusion threshold, the use of single-unit transfusions when necessary, and other measures.
The researchers analyzed data on 2662 patients, 387 treated before the PBM program began and 2275 treated after.
As expected, the rate of transfusions decreased after the PBM program began. The rate of red blood cell transfusion decreased from 39.3% to 20.8% (P<0.001), the rate of fresh-frozen plasma transfusion decreased from 18.3% to 6.5% (P<0.001), and the rate of platelet transfusion decreased from 17.8% to 9.8% (P<0.001).
Red blood cell loss decreased from a median of 721 mL to 552 mL (P<0.001), and pre-transfusion hemoglobin decreased from a mean of 7.2 ± 1.4 g/dL to 6.6 ± 1.2 g/dL (P<0.001).
Patients saw a decrease in the incidence of post-operative kidney injury from 7.6% to 5.0% (P=0.039) and a decrease in the median length of hospital stay from 10 days to 8 days (P<0.001).
Total adjusted direct costs decreased after the program began as well, falling from a median of $39,709 to $36,906 (P< 0.001).
There was no significant difference in the rate of hospital mortality or the incidence of cerebral vascular accident before and after the PBM program began.
Study quantifies VTE risk with different birth control pills

Results of a large, retrospective study support the association between newer contraceptive pills and a higher risk of venous thromboembolism (VTE).
The research showed that pills containing one of the newer types of progestogen—drospirenone, desogestrel, gestodene, and cyproterone—are associated with a nearly 2-fold higher risk of VTE than pills containing older progestogens—levonorgestrel, norethisterone, and norgestimate.
The researchers said this study has sufficient power to provide reliable comparative findings for different formulations of combined oral contraceptives. However, because it is an observational study, no definitive conclusions can be drawn about cause and effect.
The team described this research in BMJ alongside a related editorial.
Although the increased risk of VTE associated with combined oral contraceptives has been suggested previously, prior studies have used different methods to examine this link. So the relative risks associated with different combinations remain inconclusive.
Yana Vinogradova, of the University of Nottingham in the UK, and her colleagues tried to address these differences to help explain the range of results.
The team used prescription data from 2 large UK general practice databases to measure the associations between the use of combined oral contraceptives and the risk of VTE in women aged 15 to 49, adjusting for other known VTE risk factors.
The researchers matched 10,562 women with VTE to 42,034 control subjects and found that women who used any combined oral contraceptive within the past year had an increased risk of VTE compared with non-users of similar age and health status. The adjusted odds ratio was 2.97.
The risk of VTE was significantly higher for women who used the newer oral contraceptives than the older pills (P<0.001). The adjusted odds ratios were 4.28 for desogestrel, 4.27 for cyproterone, 4.12 for drospirenone, and 3.64 for gestodene, compared to 2.38 for levonorgestrel, 2.53 for norgestimate, and 2.56 for norethisterone.
The number of extra VTE cases per year per 10,000 treated women was lowest for levonorgestrel and norgestimate (6 cases for both) and highest for desogestrel and cyproterone (14 cases for both).
The researchers said that, although this is an observational study, it has produced the most reliable possible VTE risk estimates using currently available UK prescription data.

Results of a large, retrospective study support the association between newer contraceptive pills and a higher risk of venous thromboembolism (VTE).
The research showed that pills containing one of the newer types of progestogen—drospirenone, desogestrel, gestodene, and cyproterone—are associated with a nearly 2-fold higher risk of VTE than pills containing older progestogens—levonorgestrel, norethisterone, and norgestimate.
The researchers said this study has sufficient power to provide reliable comparative findings for different formulations of combined oral contraceptives. However, because it is an observational study, no definitive conclusions can be drawn about cause and effect.
The team described this research in BMJ alongside a related editorial.
Although the increased risk of VTE associated with combined oral contraceptives has been suggested previously, prior studies have used different methods to examine this link. So the relative risks associated with different combinations remain inconclusive.
Yana Vinogradova, of the University of Nottingham in the UK, and her colleagues tried to address these differences to help explain the range of results.
The team used prescription data from 2 large UK general practice databases to measure the associations between the use of combined oral contraceptives and the risk of VTE in women aged 15 to 49, adjusting for other known VTE risk factors.
The researchers matched 10,562 women with VTE to 42,034 control subjects and found that women who used any combined oral contraceptive within the past year had an increased risk of VTE compared with non-users of similar age and health status. The adjusted odds ratio was 2.97.
The risk of VTE was significantly higher for women who used the newer oral contraceptives than the older pills (P<0.001). The adjusted odds ratios were 4.28 for desogestrel, 4.27 for cyproterone, 4.12 for drospirenone, and 3.64 for gestodene, compared to 2.38 for levonorgestrel, 2.53 for norgestimate, and 2.56 for norethisterone.
The number of extra VTE cases per year per 10,000 treated women was lowest for levonorgestrel and norgestimate (6 cases for both) and highest for desogestrel and cyproterone (14 cases for both).
The researchers said that, although this is an observational study, it has produced the most reliable possible VTE risk estimates using currently available UK prescription data.

Results of a large, retrospective study support the association between newer contraceptive pills and a higher risk of venous thromboembolism (VTE).
The research showed that pills containing one of the newer types of progestogen—drospirenone, desogestrel, gestodene, and cyproterone—are associated with a nearly 2-fold higher risk of VTE than pills containing older progestogens—levonorgestrel, norethisterone, and norgestimate.
The researchers said this study has sufficient power to provide reliable comparative findings for different formulations of combined oral contraceptives. However, because it is an observational study, no definitive conclusions can be drawn about cause and effect.
The team described this research in BMJ alongside a related editorial.
Although the increased risk of VTE associated with combined oral contraceptives has been suggested previously, prior studies have used different methods to examine this link. So the relative risks associated with different combinations remain inconclusive.
Yana Vinogradova, of the University of Nottingham in the UK, and her colleagues tried to address these differences to help explain the range of results.
The team used prescription data from 2 large UK general practice databases to measure the associations between the use of combined oral contraceptives and the risk of VTE in women aged 15 to 49, adjusting for other known VTE risk factors.
The researchers matched 10,562 women with VTE to 42,034 control subjects and found that women who used any combined oral contraceptive within the past year had an increased risk of VTE compared with non-users of similar age and health status. The adjusted odds ratio was 2.97.
The risk of VTE was significantly higher for women who used the newer oral contraceptives than the older pills (P<0.001). The adjusted odds ratios were 4.28 for desogestrel, 4.27 for cyproterone, 4.12 for drospirenone, and 3.64 for gestodene, compared to 2.38 for levonorgestrel, 2.53 for norgestimate, and 2.56 for norethisterone.
The number of extra VTE cases per year per 10,000 treated women was lowest for levonorgestrel and norgestimate (6 cases for both) and highest for desogestrel and cyproterone (14 cases for both).
The researchers said that, although this is an observational study, it has produced the most reliable possible VTE risk estimates using currently available UK prescription data.