User login
Cancer rate doubles in HCV patients
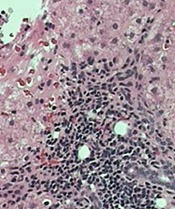
tissue with active HCV
Photo: Sutter Health
VIENNA, AUSTRIA—A 5-year retrospective study has shown that the cancer rate in patients with hepatitis C virus (HCV) is about double that for people without HCV, even when liver cancer is excluded.
And when liver cancer is included, the rate increases to 2.5 times higher in people with HCV.
Researchers presented these findings at the International Liver Congress 2015 as abstract 0058.
The team reviewed patient records from 2008 to 2012 at Kaiser Permanente Southern California, recording all cancer diagnoses in patients 18 years or older with or without HCV.
During the 5-year time period, there were 145,210 patient years in the HCV cohort and 13,948,826 patient years in the non-HCV cohort.
The mean age at cancer diagnosis was 61.8 in the HCV cohort and 63.5 in the non-HCV cohort.
Researchers recorded 2213 cancer diagnoses in the HCV cohort (1524/100,000). This number decreased to 1654 when they excluded liver cancer (1139/100,000).
In the non-HCV cohort, they recorded 84,419 cancer diagnoses (605/100,000), which decreased to 83,795 when liver cancer was excluded (601/100,000).
Cancer types known to be associated with HCV include non-Hodgkin lymphoma (NHL), renal and prostate cancers, and liver cancer.
NHL occurred 3.63 times more frequently in patients with HCV than in those without HCV, and myeloma occurred 2.93 times more frequently.
Renal cancer occurred 3.27 times and prostate cancer 1.98 times more frequently in patients with HCV than in those without.
“The results suggest that cancer rates are increased in the cohort of hepatitis C patients versus the non-hepatitis C patients, both including and excluding liver cancers,” said senior study author Lisa Nyberg, MD, MPH, of Kaiser Permanente.
“These findings certainly point to the suggestion that hepatitis C may be associated with an increased risk of cancer.”
However, she added that the findings “must be interpreted with caution, as the study also showed that confounding factors such as alcohol abuse, tobacco, obesity, and diabetes modified the results.” ![]()

tissue with active HCV
Photo: Sutter Health
VIENNA, AUSTRIA—A 5-year retrospective study has shown that the cancer rate in patients with hepatitis C virus (HCV) is about double that for people without HCV, even when liver cancer is excluded.
And when liver cancer is included, the rate increases to 2.5 times higher in people with HCV.
Researchers presented these findings at the International Liver Congress 2015 as abstract 0058.
The team reviewed patient records from 2008 to 2012 at Kaiser Permanente Southern California, recording all cancer diagnoses in patients 18 years or older with or without HCV.
During the 5-year time period, there were 145,210 patient years in the HCV cohort and 13,948,826 patient years in the non-HCV cohort.
The mean age at cancer diagnosis was 61.8 in the HCV cohort and 63.5 in the non-HCV cohort.
Researchers recorded 2213 cancer diagnoses in the HCV cohort (1524/100,000). This number decreased to 1654 when they excluded liver cancer (1139/100,000).
In the non-HCV cohort, they recorded 84,419 cancer diagnoses (605/100,000), which decreased to 83,795 when liver cancer was excluded (601/100,000).
Cancer types known to be associated with HCV include non-Hodgkin lymphoma (NHL), renal and prostate cancers, and liver cancer.
NHL occurred 3.63 times more frequently in patients with HCV than in those without HCV, and myeloma occurred 2.93 times more frequently.
Renal cancer occurred 3.27 times and prostate cancer 1.98 times more frequently in patients with HCV than in those without.
“The results suggest that cancer rates are increased in the cohort of hepatitis C patients versus the non-hepatitis C patients, both including and excluding liver cancers,” said senior study author Lisa Nyberg, MD, MPH, of Kaiser Permanente.
“These findings certainly point to the suggestion that hepatitis C may be associated with an increased risk of cancer.”
However, she added that the findings “must be interpreted with caution, as the study also showed that confounding factors such as alcohol abuse, tobacco, obesity, and diabetes modified the results.” ![]()

tissue with active HCV
Photo: Sutter Health
VIENNA, AUSTRIA—A 5-year retrospective study has shown that the cancer rate in patients with hepatitis C virus (HCV) is about double that for people without HCV, even when liver cancer is excluded.
And when liver cancer is included, the rate increases to 2.5 times higher in people with HCV.
Researchers presented these findings at the International Liver Congress 2015 as abstract 0058.
The team reviewed patient records from 2008 to 2012 at Kaiser Permanente Southern California, recording all cancer diagnoses in patients 18 years or older with or without HCV.
During the 5-year time period, there were 145,210 patient years in the HCV cohort and 13,948,826 patient years in the non-HCV cohort.
The mean age at cancer diagnosis was 61.8 in the HCV cohort and 63.5 in the non-HCV cohort.
Researchers recorded 2213 cancer diagnoses in the HCV cohort (1524/100,000). This number decreased to 1654 when they excluded liver cancer (1139/100,000).
In the non-HCV cohort, they recorded 84,419 cancer diagnoses (605/100,000), which decreased to 83,795 when liver cancer was excluded (601/100,000).
Cancer types known to be associated with HCV include non-Hodgkin lymphoma (NHL), renal and prostate cancers, and liver cancer.
NHL occurred 3.63 times more frequently in patients with HCV than in those without HCV, and myeloma occurred 2.93 times more frequently.
Renal cancer occurred 3.27 times and prostate cancer 1.98 times more frequently in patients with HCV than in those without.
“The results suggest that cancer rates are increased in the cohort of hepatitis C patients versus the non-hepatitis C patients, both including and excluding liver cancers,” said senior study author Lisa Nyberg, MD, MPH, of Kaiser Permanente.
“These findings certainly point to the suggestion that hepatitis C may be associated with an increased risk of cancer.”
However, she added that the findings “must be interpreted with caution, as the study also showed that confounding factors such as alcohol abuse, tobacco, obesity, and diabetes modified the results.” ![]()
First spray-dried fibrin sealant approved by FDA
Photo by Piotr Bodzek
The US Food and Drug Administration (FDA) has approved the first spray-dried fibrin sealant to help control bleeding during surgery.
The agency approved Raplixa (formerly known as Fibrocaps) for use in adults to control bleeding from small blood vessels when standard surgical techniques are ineffective or impractical.
The standard techniques include suture, ligature, or cautery.
Raplixa contains fibrinogen and thrombin, proteins found in human plasma. When the surgical team applies Raplixa to the bleeding site, the sealant dissolves in the blood, and the fibrinogen and thrombin proteins react, resulting in the formation of blood clots.
The protein components are individually purified using a manufacturing process that includes virus inactivation and removal steps to help reduce the risk for the transmission of blood-borne viruses. The fibrin sealant components are then spray-dried, blended, and packaged in a vial.
Raplixa can be applied directly from the original product vial or by spraying it with a delivery device. Raplixa is used in conjunction with an absorbable gelatin sponge.
“The spray-drying process used to manufacture Raplixa produces dried powders that can be combined into a single vial,” said Karen Midthun, MD, of the FDA’s Center for Biologics Evaluation and Research.
“This eliminates the need to combine the fibrinogen and thrombin before use and allows the product to be stored at room temperature.”
Raplixa was approved for use in the European Union in March, based on the recommendation of the European Medicines Agency’s Committee for Medicinal Products for Human Use.
Phase 3 trial
The FDA approved Raplixa based on data from the FINISH-3 trial, a randomized, single-blind, controlled, phase 3 study of 719 patients undergoing spinal surgery (n=183), hepatic resection (n=180), vascular surgery (n=175), or soft tissue dissection (n=181) in 4 countries over 11 months.
As reported in the Journal of the American College of Surgeons, adults with mild or moderate surgical bleeding were randomized to recieve Raplixa or the gelatin sponge. Researchers recorded the time to hemostasis over 5 minutes within each surgical indication.
Four hundred and eighty patients were treated with Raplixa, and 239 were treated with the gelatin sponge. Surgeons used the spray device in 53% of Raplixa procedures.
Raplixa used in conjunction with the gelatin sponge significantly reduced time to hemostasis compared to the gelatin sponge alone (P<0.001 for each of the 4 sugical indications).
Raplixa also significantly reduced median time to hemostasis for each indication (P<0.0001).
Adverse events were similar between the treatment arms. The most commonly reported adverse reactions were surgical pain, nausea, constipation, fever, and decreased blood pressure.
Two percent of Raplixa-treated patients developed non-neutralizing anti-thrombin antibodies, as did 3% of patients treated with the gelatin sponge.
Raplixa is manufactured by ProFibrix BV, a wholly owned subsidiary of The Medicines Company, based in Parsippany, New Jersey. ![]()

Photo by Piotr Bodzek
The US Food and Drug Administration (FDA) has approved the first spray-dried fibrin sealant to help control bleeding during surgery.
The agency approved Raplixa (formerly known as Fibrocaps) for use in adults to control bleeding from small blood vessels when standard surgical techniques are ineffective or impractical.
The standard techniques include suture, ligature, or cautery.
Raplixa contains fibrinogen and thrombin, proteins found in human plasma. When the surgical team applies Raplixa to the bleeding site, the sealant dissolves in the blood, and the fibrinogen and thrombin proteins react, resulting in the formation of blood clots.
The protein components are individually purified using a manufacturing process that includes virus inactivation and removal steps to help reduce the risk for the transmission of blood-borne viruses. The fibrin sealant components are then spray-dried, blended, and packaged in a vial.
Raplixa can be applied directly from the original product vial or by spraying it with a delivery device. Raplixa is used in conjunction with an absorbable gelatin sponge.
“The spray-drying process used to manufacture Raplixa produces dried powders that can be combined into a single vial,” said Karen Midthun, MD, of the FDA’s Center for Biologics Evaluation and Research.
“This eliminates the need to combine the fibrinogen and thrombin before use and allows the product to be stored at room temperature.”
Raplixa was approved for use in the European Union in March, based on the recommendation of the European Medicines Agency’s Committee for Medicinal Products for Human Use.
Phase 3 trial
The FDA approved Raplixa based on data from the FINISH-3 trial, a randomized, single-blind, controlled, phase 3 study of 719 patients undergoing spinal surgery (n=183), hepatic resection (n=180), vascular surgery (n=175), or soft tissue dissection (n=181) in 4 countries over 11 months.
As reported in the Journal of the American College of Surgeons, adults with mild or moderate surgical bleeding were randomized to recieve Raplixa or the gelatin sponge. Researchers recorded the time to hemostasis over 5 minutes within each surgical indication.
Four hundred and eighty patients were treated with Raplixa, and 239 were treated with the gelatin sponge. Surgeons used the spray device in 53% of Raplixa procedures.
Raplixa used in conjunction with the gelatin sponge significantly reduced time to hemostasis compared to the gelatin sponge alone (P<0.001 for each of the 4 sugical indications).
Raplixa also significantly reduced median time to hemostasis for each indication (P<0.0001).
Adverse events were similar between the treatment arms. The most commonly reported adverse reactions were surgical pain, nausea, constipation, fever, and decreased blood pressure.
Two percent of Raplixa-treated patients developed non-neutralizing anti-thrombin antibodies, as did 3% of patients treated with the gelatin sponge.
Raplixa is manufactured by ProFibrix BV, a wholly owned subsidiary of The Medicines Company, based in Parsippany, New Jersey. ![]()

Photo by Piotr Bodzek
The US Food and Drug Administration (FDA) has approved the first spray-dried fibrin sealant to help control bleeding during surgery.
The agency approved Raplixa (formerly known as Fibrocaps) for use in adults to control bleeding from small blood vessels when standard surgical techniques are ineffective or impractical.
The standard techniques include suture, ligature, or cautery.
Raplixa contains fibrinogen and thrombin, proteins found in human plasma. When the surgical team applies Raplixa to the bleeding site, the sealant dissolves in the blood, and the fibrinogen and thrombin proteins react, resulting in the formation of blood clots.
The protein components are individually purified using a manufacturing process that includes virus inactivation and removal steps to help reduce the risk for the transmission of blood-borne viruses. The fibrin sealant components are then spray-dried, blended, and packaged in a vial.
Raplixa can be applied directly from the original product vial or by spraying it with a delivery device. Raplixa is used in conjunction with an absorbable gelatin sponge.
“The spray-drying process used to manufacture Raplixa produces dried powders that can be combined into a single vial,” said Karen Midthun, MD, of the FDA’s Center for Biologics Evaluation and Research.
“This eliminates the need to combine the fibrinogen and thrombin before use and allows the product to be stored at room temperature.”
Raplixa was approved for use in the European Union in March, based on the recommendation of the European Medicines Agency’s Committee for Medicinal Products for Human Use.
Phase 3 trial
The FDA approved Raplixa based on data from the FINISH-3 trial, a randomized, single-blind, controlled, phase 3 study of 719 patients undergoing spinal surgery (n=183), hepatic resection (n=180), vascular surgery (n=175), or soft tissue dissection (n=181) in 4 countries over 11 months.
As reported in the Journal of the American College of Surgeons, adults with mild or moderate surgical bleeding were randomized to recieve Raplixa or the gelatin sponge. Researchers recorded the time to hemostasis over 5 minutes within each surgical indication.
Four hundred and eighty patients were treated with Raplixa, and 239 were treated with the gelatin sponge. Surgeons used the spray device in 53% of Raplixa procedures.
Raplixa used in conjunction with the gelatin sponge significantly reduced time to hemostasis compared to the gelatin sponge alone (P<0.001 for each of the 4 sugical indications).
Raplixa also significantly reduced median time to hemostasis for each indication (P<0.0001).
Adverse events were similar between the treatment arms. The most commonly reported adverse reactions were surgical pain, nausea, constipation, fever, and decreased blood pressure.
Two percent of Raplixa-treated patients developed non-neutralizing anti-thrombin antibodies, as did 3% of patients treated with the gelatin sponge.
Raplixa is manufactured by ProFibrix BV, a wholly owned subsidiary of The Medicines Company, based in Parsippany, New Jersey. ![]()
Targeting receptors to better treat AML

Photo courtesy of UT
Southwestern Medical Center
Preclinical research suggests that certain receptors containing the immunoreceptor tyrosine-based inhibition motif (ITIM) are important for the development of acute myeloid leukemia (AML).
“Although counterintuitive, this result is consistent with the generally immune-suppressive and, thus, tumor-promoting roles of inhibitory receptors in the immune system,” said Chengcheng Zhang, PhD, of UT Southwestern Medical Center in Dallas, Texas.
“These findings suggest that blocking ITIM-receptor signaling in combination with conventional therapies may represent a novel strategy for AML treatment.”
Dr Zhang and his colleagues reported their findings in Nature Cell Biology.
The team focused mainly on an ITIM-containing receptor called LAIR1. They found that deleting LAIR1 abolished leukemia in several different mouse models, without affecting normal hematopoiesis.
The investigators also identified a pathway that sustains the survival and self-renewal of AML cells, the mechanism by which LAIR1 supports AML development.
They said LAIR1 induces activation of SHP-1, which acts as a phosphatase-independent signaling adaptor to recruit CAMK1 for activation of downstream CREB in AML cells. And the LAIR1–SHP-1–CAMK1–CREB pathway sustains AML stem cells.
So the investigators believe that inhibiting the signaling initiated by LAIR1 and other ITIM-containing receptors could help us treat AML more effectively.
“Our study suggests that current treatment options, including chemotherapy, may not efficiently target cancer stem cells because these inhibitory receptors enable the leukemia stem cells to survive conventional therapies, eventually resulting in tumor relapse,” Dr Zhang said.
“The blockade of ITIM-receptor signaling may prove to be a novel, effective strategy for elimination of leukemia stem cells and lead to complete remission in patients.” ![]()

Photo courtesy of UT
Southwestern Medical Center
Preclinical research suggests that certain receptors containing the immunoreceptor tyrosine-based inhibition motif (ITIM) are important for the development of acute myeloid leukemia (AML).
“Although counterintuitive, this result is consistent with the generally immune-suppressive and, thus, tumor-promoting roles of inhibitory receptors in the immune system,” said Chengcheng Zhang, PhD, of UT Southwestern Medical Center in Dallas, Texas.
“These findings suggest that blocking ITIM-receptor signaling in combination with conventional therapies may represent a novel strategy for AML treatment.”
Dr Zhang and his colleagues reported their findings in Nature Cell Biology.
The team focused mainly on an ITIM-containing receptor called LAIR1. They found that deleting LAIR1 abolished leukemia in several different mouse models, without affecting normal hematopoiesis.
The investigators also identified a pathway that sustains the survival and self-renewal of AML cells, the mechanism by which LAIR1 supports AML development.
They said LAIR1 induces activation of SHP-1, which acts as a phosphatase-independent signaling adaptor to recruit CAMK1 for activation of downstream CREB in AML cells. And the LAIR1–SHP-1–CAMK1–CREB pathway sustains AML stem cells.
So the investigators believe that inhibiting the signaling initiated by LAIR1 and other ITIM-containing receptors could help us treat AML more effectively.
“Our study suggests that current treatment options, including chemotherapy, may not efficiently target cancer stem cells because these inhibitory receptors enable the leukemia stem cells to survive conventional therapies, eventually resulting in tumor relapse,” Dr Zhang said.
“The blockade of ITIM-receptor signaling may prove to be a novel, effective strategy for elimination of leukemia stem cells and lead to complete remission in patients.” ![]()

Photo courtesy of UT
Southwestern Medical Center
Preclinical research suggests that certain receptors containing the immunoreceptor tyrosine-based inhibition motif (ITIM) are important for the development of acute myeloid leukemia (AML).
“Although counterintuitive, this result is consistent with the generally immune-suppressive and, thus, tumor-promoting roles of inhibitory receptors in the immune system,” said Chengcheng Zhang, PhD, of UT Southwestern Medical Center in Dallas, Texas.
“These findings suggest that blocking ITIM-receptor signaling in combination with conventional therapies may represent a novel strategy for AML treatment.”
Dr Zhang and his colleagues reported their findings in Nature Cell Biology.
The team focused mainly on an ITIM-containing receptor called LAIR1. They found that deleting LAIR1 abolished leukemia in several different mouse models, without affecting normal hematopoiesis.
The investigators also identified a pathway that sustains the survival and self-renewal of AML cells, the mechanism by which LAIR1 supports AML development.
They said LAIR1 induces activation of SHP-1, which acts as a phosphatase-independent signaling adaptor to recruit CAMK1 for activation of downstream CREB in AML cells. And the LAIR1–SHP-1–CAMK1–CREB pathway sustains AML stem cells.
So the investigators believe that inhibiting the signaling initiated by LAIR1 and other ITIM-containing receptors could help us treat AML more effectively.
“Our study suggests that current treatment options, including chemotherapy, may not efficiently target cancer stem cells because these inhibitory receptors enable the leukemia stem cells to survive conventional therapies, eventually resulting in tumor relapse,” Dr Zhang said.
“The blockade of ITIM-receptor signaling may prove to be a novel, effective strategy for elimination of leukemia stem cells and lead to complete remission in patients.” ![]()
Vigorous physical activity may lower risk of NHL

Photo by Shannon E. Renfroe
People who regularly engage in vigorous physical activity throughout their lifetime may have a lower risk of developing non-Hodgkin lymphoma (NHL), according to research published in Cancer Epidemiology, Biomarkers & Prevention.
“We know that being physically active reduces the risk of colon cancer and breast cancer, and also leads to a range of other physical and mental health benefits,” said study author Terry Boyle, PhD, of the University of British Columbia in Vancouver, Canada.
“Our findings suggest that people who do vigorous physical activity may also have a lower risk for NHL.”
Dr Boyle and his colleagues used data from a case-control study conducted between 2000 and 2004 in British Columbia. The team analyzed 749 NHL patients and 818 control subjects matched for age, gender, and residential location.
Study subjects recorded information on demographics and various risk factors for NHL, including lifetime recreational physical activity, on a questionnaire. Participants were asked to record the average number of days per week and average number of hours per day they performed mild, moderate, or vigorous physical activity for each decade of life.
The researchers defined “mild” activities as those that increase heart and breathing rates above resting level, “moderate” activities as those that increase heart rate moderately, and “vigorous” activities as those that increase breathing and heart rates to a high level. Mild and moderate activity were ultimately combined into a single category.
The team assigned a metabolic-equivalent (MET) value to the different types of physical activity.
Then, to assess the association between lifetime physical activity and NHL risk, the researchers calculated the average MET-hours per week over a lifetime for total physical activity, moderate-intensity activity, and vigorous-intensity activity. Finally, they classified participants into quartiles.
Participants who engaged in the most vigorously intense physical activity throughout their lifetime were classified in the second, third, and fourth quartiles. These subjects had about a 25% to 30% lower risk for NHL when compared to participants in the lowest (first) quartile of vigorously intense physical activity.
The adjusted odds ratio was 0.69 for the second quartile, 0.68 for the third, and 0.75 for the fourth (PTrend=0.072).
There was an inverse association between lifetime vigorous-intensity physical activity and overall NHL risk in males and females, as well as for all NHL subtypes. Furthermore, vigorous physical activity did not confer a greater benefit for any specific age group.
The researchers found no association between total lifetime physical activity and NHL risk or lifetime moderate-intensity physical activity and NHL risk.
Despite these results, Dr Boyle said there isn’t enough research on this topic to confirm that being physically active reduces the risk of NHL.
“So we are planning to pool data from several studies to investigate this topic further,” he said. “We know that different types of NHL may have different risk factors, so we are also planning to investigate whether physical activity influences the risk for different types of NHL in different ways.” ![]()

Photo by Shannon E. Renfroe
People who regularly engage in vigorous physical activity throughout their lifetime may have a lower risk of developing non-Hodgkin lymphoma (NHL), according to research published in Cancer Epidemiology, Biomarkers & Prevention.
“We know that being physically active reduces the risk of colon cancer and breast cancer, and also leads to a range of other physical and mental health benefits,” said study author Terry Boyle, PhD, of the University of British Columbia in Vancouver, Canada.
“Our findings suggest that people who do vigorous physical activity may also have a lower risk for NHL.”
Dr Boyle and his colleagues used data from a case-control study conducted between 2000 and 2004 in British Columbia. The team analyzed 749 NHL patients and 818 control subjects matched for age, gender, and residential location.
Study subjects recorded information on demographics and various risk factors for NHL, including lifetime recreational physical activity, on a questionnaire. Participants were asked to record the average number of days per week and average number of hours per day they performed mild, moderate, or vigorous physical activity for each decade of life.
The researchers defined “mild” activities as those that increase heart and breathing rates above resting level, “moderate” activities as those that increase heart rate moderately, and “vigorous” activities as those that increase breathing and heart rates to a high level. Mild and moderate activity were ultimately combined into a single category.
The team assigned a metabolic-equivalent (MET) value to the different types of physical activity.
Then, to assess the association between lifetime physical activity and NHL risk, the researchers calculated the average MET-hours per week over a lifetime for total physical activity, moderate-intensity activity, and vigorous-intensity activity. Finally, they classified participants into quartiles.
Participants who engaged in the most vigorously intense physical activity throughout their lifetime were classified in the second, third, and fourth quartiles. These subjects had about a 25% to 30% lower risk for NHL when compared to participants in the lowest (first) quartile of vigorously intense physical activity.
The adjusted odds ratio was 0.69 for the second quartile, 0.68 for the third, and 0.75 for the fourth (PTrend=0.072).
There was an inverse association between lifetime vigorous-intensity physical activity and overall NHL risk in males and females, as well as for all NHL subtypes. Furthermore, vigorous physical activity did not confer a greater benefit for any specific age group.
The researchers found no association between total lifetime physical activity and NHL risk or lifetime moderate-intensity physical activity and NHL risk.
Despite these results, Dr Boyle said there isn’t enough research on this topic to confirm that being physically active reduces the risk of NHL.
“So we are planning to pool data from several studies to investigate this topic further,” he said. “We know that different types of NHL may have different risk factors, so we are also planning to investigate whether physical activity influences the risk for different types of NHL in different ways.” ![]()

Photo by Shannon E. Renfroe
People who regularly engage in vigorous physical activity throughout their lifetime may have a lower risk of developing non-Hodgkin lymphoma (NHL), according to research published in Cancer Epidemiology, Biomarkers & Prevention.
“We know that being physically active reduces the risk of colon cancer and breast cancer, and also leads to a range of other physical and mental health benefits,” said study author Terry Boyle, PhD, of the University of British Columbia in Vancouver, Canada.
“Our findings suggest that people who do vigorous physical activity may also have a lower risk for NHL.”
Dr Boyle and his colleagues used data from a case-control study conducted between 2000 and 2004 in British Columbia. The team analyzed 749 NHL patients and 818 control subjects matched for age, gender, and residential location.
Study subjects recorded information on demographics and various risk factors for NHL, including lifetime recreational physical activity, on a questionnaire. Participants were asked to record the average number of days per week and average number of hours per day they performed mild, moderate, or vigorous physical activity for each decade of life.
The researchers defined “mild” activities as those that increase heart and breathing rates above resting level, “moderate” activities as those that increase heart rate moderately, and “vigorous” activities as those that increase breathing and heart rates to a high level. Mild and moderate activity were ultimately combined into a single category.
The team assigned a metabolic-equivalent (MET) value to the different types of physical activity.
Then, to assess the association between lifetime physical activity and NHL risk, the researchers calculated the average MET-hours per week over a lifetime for total physical activity, moderate-intensity activity, and vigorous-intensity activity. Finally, they classified participants into quartiles.
Participants who engaged in the most vigorously intense physical activity throughout their lifetime were classified in the second, third, and fourth quartiles. These subjects had about a 25% to 30% lower risk for NHL when compared to participants in the lowest (first) quartile of vigorously intense physical activity.
The adjusted odds ratio was 0.69 for the second quartile, 0.68 for the third, and 0.75 for the fourth (PTrend=0.072).
There was an inverse association between lifetime vigorous-intensity physical activity and overall NHL risk in males and females, as well as for all NHL subtypes. Furthermore, vigorous physical activity did not confer a greater benefit for any specific age group.
The researchers found no association between total lifetime physical activity and NHL risk or lifetime moderate-intensity physical activity and NHL risk.
Despite these results, Dr Boyle said there isn’t enough research on this topic to confirm that being physically active reduces the risk of NHL.
“So we are planning to pool data from several studies to investigate this topic further,” he said. “We know that different types of NHL may have different risk factors, so we are also planning to investigate whether physical activity influences the risk for different types of NHL in different ways.” ![]()
Symptoms confer higher-than-expected risk of HL, NHL
Results from two new studies indicate that lymphadenopathy and head and neck masses are associated with a higher risk of lymphoma than we thought.
These two factors proved to be the strongest predictors of Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL).
So unless these symptoms can be explained, general practitioners should refer affected patients to specialists as quickly as possible, study investigators said.
Both studies were published in the British Journal of General Practice.
“Cancer guidelines are based on the most robust evidence, and, up to now, this has been missing,” said Willie Hamilton, MD, of the University of Exeter Medical School in the UK.
“Our research has revealed the importance of persistent, swollen lymph glands, particularly in the neck, as part of cancer. Of course, swollen glands are common with throat infections, but in cancer, they are usually larger and painless. It’s been known for a long time that this could represent cancer. This study shows that the risk is higher than previously thought.”
The first study was a large-scale assessment of symptoms that are markers of NHL. Researchers assessed 4362 NHL patients (≥ 40 years of age) and 19,468 controls.
The 5 symptoms associated with the highest risk of developing NHL were lymphadenopathy (odds ratio [OR]=263), head and neck mass not described as lymphadenopathy (OR=49), other mass (OR=12), weight loss (OR=3.2), and abdominal pain (OR=2.5).
In the second study, investigators assessed 283 HL patients (≥ 40 years of age) and 1237 control subjects.
The team found that 6 features were independently associated with HL—lymphadenopathy (OR=280), head and neck mass not described as lymphadenopathy (OR=260), other mass (OR=12), thrombocytosis (OR=6.0), raised inflammatory markers (OR=5.2), and low full blood count (OR=2.8).
Combining the results of both studies, the investigators found that, for subjects older than 60 years of age, lymphadenopathy had a positive-predictive value of 18.6% for either NHL or HL. The positive-predictive value was 4.6% for head and neck mass and 1.1% for a mass elsewhere.
Therefore, the team said patients in this age group who present with lymphadenopathy or a head and neck mass should be referred to a specialist, unless there is a clear alternative explanation.
Referral is particularly urgent if either symptom has been present for 6 weeks or more, according to the investigators. They said that no blood test or other symptoms change that.
“Early diagnosis is vital to reducing cancer deaths,” said Liz Shephard, PhD, of the University of Exeter Medical School. “We now hope that this research will feed into guidelines to help GPs refer earlier and potentially to save lives.” ![]()

Results from two new studies indicate that lymphadenopathy and head and neck masses are associated with a higher risk of lymphoma than we thought.
These two factors proved to be the strongest predictors of Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL).
So unless these symptoms can be explained, general practitioners should refer affected patients to specialists as quickly as possible, study investigators said.
Both studies were published in the British Journal of General Practice.
“Cancer guidelines are based on the most robust evidence, and, up to now, this has been missing,” said Willie Hamilton, MD, of the University of Exeter Medical School in the UK.
“Our research has revealed the importance of persistent, swollen lymph glands, particularly in the neck, as part of cancer. Of course, swollen glands are common with throat infections, but in cancer, they are usually larger and painless. It’s been known for a long time that this could represent cancer. This study shows that the risk is higher than previously thought.”
The first study was a large-scale assessment of symptoms that are markers of NHL. Researchers assessed 4362 NHL patients (≥ 40 years of age) and 19,468 controls.
The 5 symptoms associated with the highest risk of developing NHL were lymphadenopathy (odds ratio [OR]=263), head and neck mass not described as lymphadenopathy (OR=49), other mass (OR=12), weight loss (OR=3.2), and abdominal pain (OR=2.5).
In the second study, investigators assessed 283 HL patients (≥ 40 years of age) and 1237 control subjects.
The team found that 6 features were independently associated with HL—lymphadenopathy (OR=280), head and neck mass not described as lymphadenopathy (OR=260), other mass (OR=12), thrombocytosis (OR=6.0), raised inflammatory markers (OR=5.2), and low full blood count (OR=2.8).
Combining the results of both studies, the investigators found that, for subjects older than 60 years of age, lymphadenopathy had a positive-predictive value of 18.6% for either NHL or HL. The positive-predictive value was 4.6% for head and neck mass and 1.1% for a mass elsewhere.
Therefore, the team said patients in this age group who present with lymphadenopathy or a head and neck mass should be referred to a specialist, unless there is a clear alternative explanation.
Referral is particularly urgent if either symptom has been present for 6 weeks or more, according to the investigators. They said that no blood test or other symptoms change that.
“Early diagnosis is vital to reducing cancer deaths,” said Liz Shephard, PhD, of the University of Exeter Medical School. “We now hope that this research will feed into guidelines to help GPs refer earlier and potentially to save lives.” ![]()

Results from two new studies indicate that lymphadenopathy and head and neck masses are associated with a higher risk of lymphoma than we thought.
These two factors proved to be the strongest predictors of Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL).
So unless these symptoms can be explained, general practitioners should refer affected patients to specialists as quickly as possible, study investigators said.
Both studies were published in the British Journal of General Practice.
“Cancer guidelines are based on the most robust evidence, and, up to now, this has been missing,” said Willie Hamilton, MD, of the University of Exeter Medical School in the UK.
“Our research has revealed the importance of persistent, swollen lymph glands, particularly in the neck, as part of cancer. Of course, swollen glands are common with throat infections, but in cancer, they are usually larger and painless. It’s been known for a long time that this could represent cancer. This study shows that the risk is higher than previously thought.”
The first study was a large-scale assessment of symptoms that are markers of NHL. Researchers assessed 4362 NHL patients (≥ 40 years of age) and 19,468 controls.
The 5 symptoms associated with the highest risk of developing NHL were lymphadenopathy (odds ratio [OR]=263), head and neck mass not described as lymphadenopathy (OR=49), other mass (OR=12), weight loss (OR=3.2), and abdominal pain (OR=2.5).
In the second study, investigators assessed 283 HL patients (≥ 40 years of age) and 1237 control subjects.
The team found that 6 features were independently associated with HL—lymphadenopathy (OR=280), head and neck mass not described as lymphadenopathy (OR=260), other mass (OR=12), thrombocytosis (OR=6.0), raised inflammatory markers (OR=5.2), and low full blood count (OR=2.8).
Combining the results of both studies, the investigators found that, for subjects older than 60 years of age, lymphadenopathy had a positive-predictive value of 18.6% for either NHL or HL. The positive-predictive value was 4.6% for head and neck mass and 1.1% for a mass elsewhere.
Therefore, the team said patients in this age group who present with lymphadenopathy or a head and neck mass should be referred to a specialist, unless there is a clear alternative explanation.
Referral is particularly urgent if either symptom has been present for 6 weeks or more, according to the investigators. They said that no blood test or other symptoms change that.
“Early diagnosis is vital to reducing cancer deaths,” said Liz Shephard, PhD, of the University of Exeter Medical School. “We now hope that this research will feed into guidelines to help GPs refer earlier and potentially to save lives.” ![]()
Artificial blood vessels give way to the real thing

biocompatible polymer
Photo courtesy of Vienna
University of Technology
Scientists have created implantable artificial blood vessels using a newly developed polymer that is biodegradable.
In experiments with rats, these thin-walled vascular grafts were replaced by endogenous material, ultimately leaving natural, fully functional blood vessels in their place.
Furthermore, 6 months after the artificial vessels were implanted, none of the animals had experienced thromboses, inflammation, or aneurysms.
Helga Bergmeister, MD, DVM, PhD, of the Medical University of Vienna in Austria, and her colleagues conducted this research and described the results in Acta Biomaterialia.
To create artificial blood vessels that are compatible with body tissue, the researchers developed a new polymer—thermoplastic polyurethane.
“By selecting very specific molecular building blocks, we have succeeded in synthesizing a polymer with the desired properties,” said Robert Liska, PhD, of the Vienna University of Technology.
To produce the grafts, the researchers spun polymer solutions in an electrical field to form very fine threads and wound these threads onto a spool.
“The wall of these artificial blood vessels is very similar to that of natural ones,” said Heinrich Schima, PhD, of the Medical University of Vienna.
The polymer fabric is slightly porous. So, initially, it allows a small amount of blood to seep through, which enriches the wall with growth factors. And this encourages the migration of endogenous cells.
The researchers implanted these artificial blood vessels in rats and found them to be safe and functional long-term.
“The rats’ blood vessels were examined 6 months after insertion of the vascular prostheses,” Dr Bergmeister said.
“We did not find any aneurysms, thrombosis, or inflammation. Endogenous cells had colonized the vascular prostheses and turned the artificial constructs into natural body tissue.”
In fact, natural body tissue regrew much faster than expected.
The researchers said their thin-walled grafts “offer a new and desirable form of biodegradable vascular implant.” ![]()

biocompatible polymer
Photo courtesy of Vienna
University of Technology
Scientists have created implantable artificial blood vessels using a newly developed polymer that is biodegradable.
In experiments with rats, these thin-walled vascular grafts were replaced by endogenous material, ultimately leaving natural, fully functional blood vessels in their place.
Furthermore, 6 months after the artificial vessels were implanted, none of the animals had experienced thromboses, inflammation, or aneurysms.
Helga Bergmeister, MD, DVM, PhD, of the Medical University of Vienna in Austria, and her colleagues conducted this research and described the results in Acta Biomaterialia.
To create artificial blood vessels that are compatible with body tissue, the researchers developed a new polymer—thermoplastic polyurethane.
“By selecting very specific molecular building blocks, we have succeeded in synthesizing a polymer with the desired properties,” said Robert Liska, PhD, of the Vienna University of Technology.
To produce the grafts, the researchers spun polymer solutions in an electrical field to form very fine threads and wound these threads onto a spool.
“The wall of these artificial blood vessels is very similar to that of natural ones,” said Heinrich Schima, PhD, of the Medical University of Vienna.
The polymer fabric is slightly porous. So, initially, it allows a small amount of blood to seep through, which enriches the wall with growth factors. And this encourages the migration of endogenous cells.
The researchers implanted these artificial blood vessels in rats and found them to be safe and functional long-term.
“The rats’ blood vessels were examined 6 months after insertion of the vascular prostheses,” Dr Bergmeister said.
“We did not find any aneurysms, thrombosis, or inflammation. Endogenous cells had colonized the vascular prostheses and turned the artificial constructs into natural body tissue.”
In fact, natural body tissue regrew much faster than expected.
The researchers said their thin-walled grafts “offer a new and desirable form of biodegradable vascular implant.” ![]()

biocompatible polymer
Photo courtesy of Vienna
University of Technology
Scientists have created implantable artificial blood vessels using a newly developed polymer that is biodegradable.
In experiments with rats, these thin-walled vascular grafts were replaced by endogenous material, ultimately leaving natural, fully functional blood vessels in their place.
Furthermore, 6 months after the artificial vessels were implanted, none of the animals had experienced thromboses, inflammation, or aneurysms.
Helga Bergmeister, MD, DVM, PhD, of the Medical University of Vienna in Austria, and her colleagues conducted this research and described the results in Acta Biomaterialia.
To create artificial blood vessels that are compatible with body tissue, the researchers developed a new polymer—thermoplastic polyurethane.
“By selecting very specific molecular building blocks, we have succeeded in synthesizing a polymer with the desired properties,” said Robert Liska, PhD, of the Vienna University of Technology.
To produce the grafts, the researchers spun polymer solutions in an electrical field to form very fine threads and wound these threads onto a spool.
“The wall of these artificial blood vessels is very similar to that of natural ones,” said Heinrich Schima, PhD, of the Medical University of Vienna.
The polymer fabric is slightly porous. So, initially, it allows a small amount of blood to seep through, which enriches the wall with growth factors. And this encourages the migration of endogenous cells.
The researchers implanted these artificial blood vessels in rats and found them to be safe and functional long-term.
“The rats’ blood vessels were examined 6 months after insertion of the vascular prostheses,” Dr Bergmeister said.
“We did not find any aneurysms, thrombosis, or inflammation. Endogenous cells had colonized the vascular prostheses and turned the artificial constructs into natural body tissue.”
In fact, natural body tissue regrew much faster than expected.
The researchers said their thin-walled grafts “offer a new and desirable form of biodegradable vascular implant.” ![]()
Enzyme could enable creation of universal blood type

Photo by Elise Amendola
Chemists have generated an enzyme that shows the potential for converting type A or B blood into a universal blood type.
The enzyme works by snipping off the antigens found in blood types A and B, making these blood types more like O, which can be given to patients of all blood types.
The enzyme was able to remove most of the antigens in type A and B blood. Before it can be used in clinical settings, however, all of the antigens would need to be removed.
David Kwan, PhD, of the University of British Columbia in Vancouver, Canada, and his colleagues described their work with this enzyme in the Journal of the American Chemical Society.
“We produced a mutant enzyme that is very efficient at cutting off the sugars in A and B blood and is much more proficient at removing the subtypes of the A antigen that the parent enzyme struggles with,” Dr Kwan said.
To create the enzyme, Dr Kwan and his colleagues used a technology called directed evolution. It involves inserting mutations into the gene that codes for the enzyme and selecting mutants that are more effective at cutting the antigens.
The team started with the family 98 glycoside hydrolase from Streptococcus pneumoniae SP3-BS71 (Sp3GH98), which cleaves the entire terminal trisaccharide antigenic determinants of both A and B antigens from some of the linkages on red blood cell surface glycans.
Through directed evolution, the researchers developed variants of Sp3GH98 that showed improved activity toward some of the linkages that are resistant to cleavage by the wild-type enzyme.
In 5 generations, the enzyme became 170 times more effective. This Sp3GH98 variant could remove the majority of the antigens in type A and B blood.
The researchers said the enzyme must be able to remove all of the antigens before it can be used in the clinic. The immune system is highly sensitive to blood groups, and even small amounts of residual antigens could trigger an immune response.
The concept of using an enzyme to change blood types is not new, said study author Steve Withers, PhD, also from the University of British Columbia.
“But, until now, we needed so much of the enzyme to make it work that it was impractical,” he said. “Now, I’m confident that we can take this a whole lot further.” ![]()

Photo by Elise Amendola
Chemists have generated an enzyme that shows the potential for converting type A or B blood into a universal blood type.
The enzyme works by snipping off the antigens found in blood types A and B, making these blood types more like O, which can be given to patients of all blood types.
The enzyme was able to remove most of the antigens in type A and B blood. Before it can be used in clinical settings, however, all of the antigens would need to be removed.
David Kwan, PhD, of the University of British Columbia in Vancouver, Canada, and his colleagues described their work with this enzyme in the Journal of the American Chemical Society.
“We produced a mutant enzyme that is very efficient at cutting off the sugars in A and B blood and is much more proficient at removing the subtypes of the A antigen that the parent enzyme struggles with,” Dr Kwan said.
To create the enzyme, Dr Kwan and his colleagues used a technology called directed evolution. It involves inserting mutations into the gene that codes for the enzyme and selecting mutants that are more effective at cutting the antigens.
The team started with the family 98 glycoside hydrolase from Streptococcus pneumoniae SP3-BS71 (Sp3GH98), which cleaves the entire terminal trisaccharide antigenic determinants of both A and B antigens from some of the linkages on red blood cell surface glycans.
Through directed evolution, the researchers developed variants of Sp3GH98 that showed improved activity toward some of the linkages that are resistant to cleavage by the wild-type enzyme.
In 5 generations, the enzyme became 170 times more effective. This Sp3GH98 variant could remove the majority of the antigens in type A and B blood.
The researchers said the enzyme must be able to remove all of the antigens before it can be used in the clinic. The immune system is highly sensitive to blood groups, and even small amounts of residual antigens could trigger an immune response.
The concept of using an enzyme to change blood types is not new, said study author Steve Withers, PhD, also from the University of British Columbia.
“But, until now, we needed so much of the enzyme to make it work that it was impractical,” he said. “Now, I’m confident that we can take this a whole lot further.” ![]()

Photo by Elise Amendola
Chemists have generated an enzyme that shows the potential for converting type A or B blood into a universal blood type.
The enzyme works by snipping off the antigens found in blood types A and B, making these blood types more like O, which can be given to patients of all blood types.
The enzyme was able to remove most of the antigens in type A and B blood. Before it can be used in clinical settings, however, all of the antigens would need to be removed.
David Kwan, PhD, of the University of British Columbia in Vancouver, Canada, and his colleagues described their work with this enzyme in the Journal of the American Chemical Society.
“We produced a mutant enzyme that is very efficient at cutting off the sugars in A and B blood and is much more proficient at removing the subtypes of the A antigen that the parent enzyme struggles with,” Dr Kwan said.
To create the enzyme, Dr Kwan and his colleagues used a technology called directed evolution. It involves inserting mutations into the gene that codes for the enzyme and selecting mutants that are more effective at cutting the antigens.
The team started with the family 98 glycoside hydrolase from Streptococcus pneumoniae SP3-BS71 (Sp3GH98), which cleaves the entire terminal trisaccharide antigenic determinants of both A and B antigens from some of the linkages on red blood cell surface glycans.
Through directed evolution, the researchers developed variants of Sp3GH98 that showed improved activity toward some of the linkages that are resistant to cleavage by the wild-type enzyme.
In 5 generations, the enzyme became 170 times more effective. This Sp3GH98 variant could remove the majority of the antigens in type A and B blood.
The researchers said the enzyme must be able to remove all of the antigens before it can be used in the clinic. The immune system is highly sensitive to blood groups, and even small amounts of residual antigens could trigger an immune response.
The concept of using an enzyme to change blood types is not new, said study author Steve Withers, PhD, also from the University of British Columbia.
“But, until now, we needed so much of the enzyme to make it work that it was impractical,” he said. “Now, I’m confident that we can take this a whole lot further.”
Identifying artemisinin resistance not so straightforward

Photo by Juan D. Alfonso
The current method used to identify resistance to the antimalarial drug artemisinin is not entirely accurate, according to research published in PLOS Medicine.
Artemisinin rapidly clears malaria parasites from the blood of infected patients. When parasites develop resistance, clearance takes longer.
The best measure of parasite clearance is the parasite half-life in a patient’s blood, and a common cutoff used to denote artemisinin resistance is 5 hours.
Study author Lisa White, of Mahidol University in Bangkok, Thailand, and her colleagues found that parasite half-life predicts the likelihood of an artemisinin-resistant infection for individual patients. But the half-life is influenced by how common resistance is in the particular area.
The critical half-life varied between 3.5 hours in areas where resistance is rare to 5.5 hours in areas where resistance is common. This means there is no universal cutoff value in parasite half-life that can determine whether a particular infection is “sensitive” or “resistant” to artemisinin-based combination (ACT) therapy.
Because measuring the parasite half-life requires frequent blood sampling that is difficult to do in resource-limited settings, the World Health Organization (WHO) uses the following working definition for surveillance. Artemisinin resistance in a population is suspected if more than 10% of patients are still carrying parasites 3 days after the start of ACT.
Arguing that the cutoff used in the WHO’s working definition is based on limited data, the researchers examined how well the definition matches actual data from patients in areas with artemisinin-resistant parasites.
Applying a model specifically developed for this purpose, the team found that the WHO’s day-3 cutoff value of 10% is useful, but it would be more informative if the parasite load at the start of ACT was taken into account.
The researchers concluded that the WHO definition alone cannot be used to accurately predict the real proportion of artemisinin-resistant parasites, so a more detailed assessment is needed.

Photo by Juan D. Alfonso
The current method used to identify resistance to the antimalarial drug artemisinin is not entirely accurate, according to research published in PLOS Medicine.
Artemisinin rapidly clears malaria parasites from the blood of infected patients. When parasites develop resistance, clearance takes longer.
The best measure of parasite clearance is the parasite half-life in a patient’s blood, and a common cutoff used to denote artemisinin resistance is 5 hours.
Study author Lisa White, of Mahidol University in Bangkok, Thailand, and her colleagues found that parasite half-life predicts the likelihood of an artemisinin-resistant infection for individual patients. But the half-life is influenced by how common resistance is in the particular area.
The critical half-life varied between 3.5 hours in areas where resistance is rare to 5.5 hours in areas where resistance is common. This means there is no universal cutoff value in parasite half-life that can determine whether a particular infection is “sensitive” or “resistant” to artemisinin-based combination (ACT) therapy.
Because measuring the parasite half-life requires frequent blood sampling that is difficult to do in resource-limited settings, the World Health Organization (WHO) uses the following working definition for surveillance. Artemisinin resistance in a population is suspected if more than 10% of patients are still carrying parasites 3 days after the start of ACT.
Arguing that the cutoff used in the WHO’s working definition is based on limited data, the researchers examined how well the definition matches actual data from patients in areas with artemisinin-resistant parasites.
Applying a model specifically developed for this purpose, the team found that the WHO’s day-3 cutoff value of 10% is useful, but it would be more informative if the parasite load at the start of ACT was taken into account.
The researchers concluded that the WHO definition alone cannot be used to accurately predict the real proportion of artemisinin-resistant parasites, so a more detailed assessment is needed.

Photo by Juan D. Alfonso
The current method used to identify resistance to the antimalarial drug artemisinin is not entirely accurate, according to research published in PLOS Medicine.
Artemisinin rapidly clears malaria parasites from the blood of infected patients. When parasites develop resistance, clearance takes longer.
The best measure of parasite clearance is the parasite half-life in a patient’s blood, and a common cutoff used to denote artemisinin resistance is 5 hours.
Study author Lisa White, of Mahidol University in Bangkok, Thailand, and her colleagues found that parasite half-life predicts the likelihood of an artemisinin-resistant infection for individual patients. But the half-life is influenced by how common resistance is in the particular area.
The critical half-life varied between 3.5 hours in areas where resistance is rare to 5.5 hours in areas where resistance is common. This means there is no universal cutoff value in parasite half-life that can determine whether a particular infection is “sensitive” or “resistant” to artemisinin-based combination (ACT) therapy.
Because measuring the parasite half-life requires frequent blood sampling that is difficult to do in resource-limited settings, the World Health Organization (WHO) uses the following working definition for surveillance. Artemisinin resistance in a population is suspected if more than 10% of patients are still carrying parasites 3 days after the start of ACT.
Arguing that the cutoff used in the WHO’s working definition is based on limited data, the researchers examined how well the definition matches actual data from patients in areas with artemisinin-resistant parasites.
Applying a model specifically developed for this purpose, the team found that the WHO’s day-3 cutoff value of 10% is useful, but it would be more informative if the parasite load at the start of ACT was taken into account.
The researchers concluded that the WHO definition alone cannot be used to accurately predict the real proportion of artemisinin-resistant parasites, so a more detailed assessment is needed.
Vena cava filter doesn’t reduce risk of recurrent PE
Photo from Business Wire
Retrievable inferior vena cava filters do not reduce the risk of recurrent pulmonary embolism (PE) or death in patients with PE, according to a study
published in JAMA.
The study showed no significant difference in outcomes between patients who received anticoagulation in conjunction with an inferior vena cava
filter and patients who received anticoagulant therapy alone.
Studies have shown an increase in the placement of inferior vena cava filters over the past few decades. However, a lack of reliable data on the use of these filters has meant the benefit-risk ratio of using them in patients at risk of recurrent venous thromboembolism (VTE) is uncertain.
To gain more insight, Patrick Mismetti, MD, PhD, of the Centre Hospitalier Universitaire de Saint-Etienne in France, and his colleagues conducted a prospective study.
They enrolled hospitalized patients with acute, symptomatic PE associated with lower-limb vein thrombosis who had at least 1 criterion for severity. Patients were randomized to receive anticoagulation and a retrievable inferior vena cava filter (n=200) or anticoagulation alone (n=199).
The follow-up period was 6 months. Patients received full-dose anticoagulation for at least 6 months, and filter retrieval was planned at 3 months from placement.
Filters were successfully inserted in 193 patients. Filter retrieval was successful in 153 of the 164 patients in whom retrieval was attempted.
In the anticoagulant-only group, 6 patients ultimately received a filter, 4 because they were undergoing surgery and needed to stop anticoagulation, and 2 because of bleeding complications.
By 3 months, 6 patients (3.0%) had recurrent PE in the filter group, as did 3 patients (1.5%) in the anticoagulant-only group (P=0.50). All episodes in the filter group and 2 in the anticoagulant group were fatal.
The researchers observed 1 additional PE recurrence in each treatment group between 3 and 6 months (P=0.54). And 3 patients developed filter thrombosis.
At 3 months, there was no significant difference between the 2 treatment groups with regard to recurrent deep vein thrombosis (P>0.99), recurrent VTE (P=0.36), major bleeding (P=0.63), or death from any cause (P=0.55).
At 6 months, there was no significant difference between the treatment groups with regard to recurrent deep vein thrombosis (P>0.99), recurrent VTE (P=0.59), major bleeding (P=0.69), or death from any cause (P=0.29).
The main cause of death in both treatment groups was cancer.
The researchers noted that the availability of retrievable inferior vena cava filters has probably contributed to their increased use for managing acute VTE, including their use in addition to full-dose anticoagulant therapy in patients with PE, a large clot burden, a poor cardiopulmonary reserve, or a suspected increased risk for recurrence, as advocated by several guidelines.
However, the team said the results of this study do not support the use of these filters in patients who can be treated with anticoagulation.

Photo from Business Wire
Retrievable inferior vena cava filters do not reduce the risk of recurrent pulmonary embolism (PE) or death in patients with PE, according to a study
published in JAMA.
The study showed no significant difference in outcomes between patients who received anticoagulation in conjunction with an inferior vena cava
filter and patients who received anticoagulant therapy alone.
Studies have shown an increase in the placement of inferior vena cava filters over the past few decades. However, a lack of reliable data on the use of these filters has meant the benefit-risk ratio of using them in patients at risk of recurrent venous thromboembolism (VTE) is uncertain.
To gain more insight, Patrick Mismetti, MD, PhD, of the Centre Hospitalier Universitaire de Saint-Etienne in France, and his colleagues conducted a prospective study.
They enrolled hospitalized patients with acute, symptomatic PE associated with lower-limb vein thrombosis who had at least 1 criterion for severity. Patients were randomized to receive anticoagulation and a retrievable inferior vena cava filter (n=200) or anticoagulation alone (n=199).
The follow-up period was 6 months. Patients received full-dose anticoagulation for at least 6 months, and filter retrieval was planned at 3 months from placement.
Filters were successfully inserted in 193 patients. Filter retrieval was successful in 153 of the 164 patients in whom retrieval was attempted.
In the anticoagulant-only group, 6 patients ultimately received a filter, 4 because they were undergoing surgery and needed to stop anticoagulation, and 2 because of bleeding complications.
By 3 months, 6 patients (3.0%) had recurrent PE in the filter group, as did 3 patients (1.5%) in the anticoagulant-only group (P=0.50). All episodes in the filter group and 2 in the anticoagulant group were fatal.
The researchers observed 1 additional PE recurrence in each treatment group between 3 and 6 months (P=0.54). And 3 patients developed filter thrombosis.
At 3 months, there was no significant difference between the 2 treatment groups with regard to recurrent deep vein thrombosis (P>0.99), recurrent VTE (P=0.36), major bleeding (P=0.63), or death from any cause (P=0.55).
At 6 months, there was no significant difference between the treatment groups with regard to recurrent deep vein thrombosis (P>0.99), recurrent VTE (P=0.59), major bleeding (P=0.69), or death from any cause (P=0.29).
The main cause of death in both treatment groups was cancer.
The researchers noted that the availability of retrievable inferior vena cava filters has probably contributed to their increased use for managing acute VTE, including their use in addition to full-dose anticoagulant therapy in patients with PE, a large clot burden, a poor cardiopulmonary reserve, or a suspected increased risk for recurrence, as advocated by several guidelines.
However, the team said the results of this study do not support the use of these filters in patients who can be treated with anticoagulation.

Photo from Business Wire
Retrievable inferior vena cava filters do not reduce the risk of recurrent pulmonary embolism (PE) or death in patients with PE, according to a study
published in JAMA.
The study showed no significant difference in outcomes between patients who received anticoagulation in conjunction with an inferior vena cava
filter and patients who received anticoagulant therapy alone.
Studies have shown an increase in the placement of inferior vena cava filters over the past few decades. However, a lack of reliable data on the use of these filters has meant the benefit-risk ratio of using them in patients at risk of recurrent venous thromboembolism (VTE) is uncertain.
To gain more insight, Patrick Mismetti, MD, PhD, of the Centre Hospitalier Universitaire de Saint-Etienne in France, and his colleagues conducted a prospective study.
They enrolled hospitalized patients with acute, symptomatic PE associated with lower-limb vein thrombosis who had at least 1 criterion for severity. Patients were randomized to receive anticoagulation and a retrievable inferior vena cava filter (n=200) or anticoagulation alone (n=199).
The follow-up period was 6 months. Patients received full-dose anticoagulation for at least 6 months, and filter retrieval was planned at 3 months from placement.
Filters were successfully inserted in 193 patients. Filter retrieval was successful in 153 of the 164 patients in whom retrieval was attempted.
In the anticoagulant-only group, 6 patients ultimately received a filter, 4 because they were undergoing surgery and needed to stop anticoagulation, and 2 because of bleeding complications.
By 3 months, 6 patients (3.0%) had recurrent PE in the filter group, as did 3 patients (1.5%) in the anticoagulant-only group (P=0.50). All episodes in the filter group and 2 in the anticoagulant group were fatal.
The researchers observed 1 additional PE recurrence in each treatment group between 3 and 6 months (P=0.54). And 3 patients developed filter thrombosis.
At 3 months, there was no significant difference between the 2 treatment groups with regard to recurrent deep vein thrombosis (P>0.99), recurrent VTE (P=0.36), major bleeding (P=0.63), or death from any cause (P=0.55).
At 6 months, there was no significant difference between the treatment groups with regard to recurrent deep vein thrombosis (P>0.99), recurrent VTE (P=0.59), major bleeding (P=0.69), or death from any cause (P=0.29).
The main cause of death in both treatment groups was cancer.
The researchers noted that the availability of retrievable inferior vena cava filters has probably contributed to their increased use for managing acute VTE, including their use in addition to full-dose anticoagulant therapy in patients with PE, a large clot burden, a poor cardiopulmonary reserve, or a suspected increased risk for recurrence, as advocated by several guidelines.
However, the team said the results of this study do not support the use of these filters in patients who can be treated with anticoagulation.
Team discovers mechanism behind malaria progression

a red blood cell
Photo courtesy of St. Jude
Children’s Research Hospital
Researchers say they have pinpointed one of the mechanisms responsible for the progression of malaria.
Computer modeling showed that the nanoscale knobs that form at the membrane of infected red blood cells cause the cell stiffening that is, in part, responsible for the reduced blood flow that can turn malaria deadly.
Subra Suresh, ScD, of Carnegie Mellon University in Pittsburgh, Pennsylvania, and his colleagues reported this finding in PNAS.
“Many of malaria’s symptoms are the result of impeded blood flow, which is directly tied to structural changes in infected red blood cells,” Dr Suresh said.
When a red blood cell is infected with malaria, the parasite releases proteins that interact with the cell membrane. The cell membrane undergoes a series of changes that result in stiffness and stickiness.
While researchers are fairly certain that the stickiness is caused by nanoscale knobs that protrude from the cell membrane, they were uncertain as to what caused the stiffness.
They hypothesized that the parasite protein/cell membrane interaction caused spectrin, a cytoskeletal protein that provides a scaffold for the cell membrane, to rearrange its networked structure to be more rigid.
However, the complexity of the cell membrane made it difficult for researchers to study and prove this hypothesis experimentally.
To visualize what happens at the cell membrane during malarial infection, Dr Suresh and his colleagues turned to a computer simulation technique called coarse-grained molecular dynamics (CGMD). CGMD has proven valuable for studying what happens at the cell membrane because it represents the membrane’s complex proteins and lipids with larger, simplified components rather than atom by atom.
Doing this requires less computing time and power than standard atomistic models, which allows scientists to run simulations for longer periods of time while still accurately recreating the behavior of the cell membrane.
Typically, researchers introduce different variables into the simulation and observe how the membrane reacts. In the current study, the researchers seeded the model membrane with proteins released by the malaria parasite Plasmodium falciparum.
From their simulation, the researchers found that the stiffening of the red blood cell membrane had little to do with the remodeling of spectrin.
Instead, the nanoscale knobs that cause the red blood cells to stick to the vein’s walls also cause the membrane to stiffen through a number of different mechanisms, including composite strengthening, strain hardening, and density-dependent vertical coupling effects.
According to the researchers, the discovery of this mechanism could provide a promising target for new antimalarial therapies.

a red blood cell
Photo courtesy of St. Jude
Children’s Research Hospital
Researchers say they have pinpointed one of the mechanisms responsible for the progression of malaria.
Computer modeling showed that the nanoscale knobs that form at the membrane of infected red blood cells cause the cell stiffening that is, in part, responsible for the reduced blood flow that can turn malaria deadly.
Subra Suresh, ScD, of Carnegie Mellon University in Pittsburgh, Pennsylvania, and his colleagues reported this finding in PNAS.
“Many of malaria’s symptoms are the result of impeded blood flow, which is directly tied to structural changes in infected red blood cells,” Dr Suresh said.
When a red blood cell is infected with malaria, the parasite releases proteins that interact with the cell membrane. The cell membrane undergoes a series of changes that result in stiffness and stickiness.
While researchers are fairly certain that the stickiness is caused by nanoscale knobs that protrude from the cell membrane, they were uncertain as to what caused the stiffness.
They hypothesized that the parasite protein/cell membrane interaction caused spectrin, a cytoskeletal protein that provides a scaffold for the cell membrane, to rearrange its networked structure to be more rigid.
However, the complexity of the cell membrane made it difficult for researchers to study and prove this hypothesis experimentally.
To visualize what happens at the cell membrane during malarial infection, Dr Suresh and his colleagues turned to a computer simulation technique called coarse-grained molecular dynamics (CGMD). CGMD has proven valuable for studying what happens at the cell membrane because it represents the membrane’s complex proteins and lipids with larger, simplified components rather than atom by atom.
Doing this requires less computing time and power than standard atomistic models, which allows scientists to run simulations for longer periods of time while still accurately recreating the behavior of the cell membrane.
Typically, researchers introduce different variables into the simulation and observe how the membrane reacts. In the current study, the researchers seeded the model membrane with proteins released by the malaria parasite Plasmodium falciparum.
From their simulation, the researchers found that the stiffening of the red blood cell membrane had little to do with the remodeling of spectrin.
Instead, the nanoscale knobs that cause the red blood cells to stick to the vein’s walls also cause the membrane to stiffen through a number of different mechanisms, including composite strengthening, strain hardening, and density-dependent vertical coupling effects.
According to the researchers, the discovery of this mechanism could provide a promising target for new antimalarial therapies.

a red blood cell
Photo courtesy of St. Jude
Children’s Research Hospital
Researchers say they have pinpointed one of the mechanisms responsible for the progression of malaria.
Computer modeling showed that the nanoscale knobs that form at the membrane of infected red blood cells cause the cell stiffening that is, in part, responsible for the reduced blood flow that can turn malaria deadly.
Subra Suresh, ScD, of Carnegie Mellon University in Pittsburgh, Pennsylvania, and his colleagues reported this finding in PNAS.
“Many of malaria’s symptoms are the result of impeded blood flow, which is directly tied to structural changes in infected red blood cells,” Dr Suresh said.
When a red blood cell is infected with malaria, the parasite releases proteins that interact with the cell membrane. The cell membrane undergoes a series of changes that result in stiffness and stickiness.
While researchers are fairly certain that the stickiness is caused by nanoscale knobs that protrude from the cell membrane, they were uncertain as to what caused the stiffness.
They hypothesized that the parasite protein/cell membrane interaction caused spectrin, a cytoskeletal protein that provides a scaffold for the cell membrane, to rearrange its networked structure to be more rigid.
However, the complexity of the cell membrane made it difficult for researchers to study and prove this hypothesis experimentally.
To visualize what happens at the cell membrane during malarial infection, Dr Suresh and his colleagues turned to a computer simulation technique called coarse-grained molecular dynamics (CGMD). CGMD has proven valuable for studying what happens at the cell membrane because it represents the membrane’s complex proteins and lipids with larger, simplified components rather than atom by atom.
Doing this requires less computing time and power than standard atomistic models, which allows scientists to run simulations for longer periods of time while still accurately recreating the behavior of the cell membrane.
Typically, researchers introduce different variables into the simulation and observe how the membrane reacts. In the current study, the researchers seeded the model membrane with proteins released by the malaria parasite Plasmodium falciparum.
From their simulation, the researchers found that the stiffening of the red blood cell membrane had little to do with the remodeling of spectrin.
Instead, the nanoscale knobs that cause the red blood cells to stick to the vein’s walls also cause the membrane to stiffen through a number of different mechanisms, including composite strengthening, strain hardening, and density-dependent vertical coupling effects.
According to the researchers, the discovery of this mechanism could provide a promising target for new antimalarial therapies.