User login
Confirm Celiac Diagnosis by Biopsy Before Advising Gluten-Free Diet
SAN DIEGO – An intestinal biopsy is almost always necessary to confirm celiac disease and is a must before committing a patient to the only effective treatment – a lifelong gluten-free diet.
Sticking to such a restricted diet is difficult and expensive, Dr. Sheila Crowe said at the annual meeting of the American College of Physicians. "A lifelong gluten-free diet sounds simple, unless you’re the patient. ... Eating out is very difficult, especially for children and teens who face a lot of peer pressure. And eating gluten free at home is expensive. Studies in the United States, Canada, and the United Kingdom confirm that a lifelong diet of gluten-free foods costs about three times more than a normal diet," said Dr. Crowe, professor in the division of gastroenterology and hepatology at the University of Virginia, Charlottesville.
Because treating celiac disease requires this lifelong commitment, a positive serologic test isn’t enough to rule it in, she said. Nor are any of the available immunologic tests, including the most widely used – tissue transglutaminase IgA (tTG IgA) – specific enough to replace intestinal biopsy as the sole method for reliably diagnosing celiac disease. "A positive tTG test is not enough to place a person on this lifelong treatment without confirmation from an intestinal biopsy," she said. "This is especially important for children, because of the higher likelihood of false positives in that group."
tTG IgA has a very high sensitivity and specificity, but it isn’t perfect, Dr. Crowe said. "If you have a patient with clinical symptoms and the tTG comes back negative, there is still a 10% chance that’s a false negative. Another scenario could be a patient who has an autoimmune disease or a relative with celiac, and is experiencing celiac symptoms. If the tTG came back negative on that person, I would still do an endoscopy."
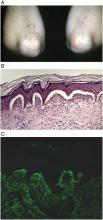
The only possible exception might be a patient with celiac symptoms who already has biopsy-proven dermatitis herpetiformis, with the classic immunofluorescent deposits at the dermal-epidermal junction. "If you biopsy these patients, the intestine will show the changes associated with celiac disease every time," Dr. Crowe said.
Celiac disease is no longer considered a disorder of childhood. "The disease is there lifelong. It appears you cannot suppress the immune response," she said.
The intestine rapidly responds to a gluten-free diet, "But the tendency to have an immunologic response to gluten is always there," Dr. Crowe said.
Relapses are common. Patients are most likely to "fall off" the diet when symptoms begin to abate, she said. They may simply feel "cured" and resume old eating patterns, or they may drop the diet because of changes that can occur as the intestine heals. "If a patient had diarrhea and malabsorption of nutrients, they might find themselves getting constipated and gaining weight on the gluten-free diet, fall off, and get ill again. Even if the intestine is healed, the vast majority of data tell us that patient will relapse," at some point after abandoning the dietary restriction, Dr. Crowe said.
This can lead to the development of refractory celiac disease, in which the intestine fails to recover despite a gluten-free diet. Patients with refractory celiac disease may be unable to fully absorb nutrients and need supplemental feeding methods.
The goal of celiac management is to promote intestinal healing, optimize nutrition, and avoid long-term damage, Dr. Crowe said. "It’s key to bring in a knowledgeable dietitian to help. And I mean knowledgeable – not someone who is going to hand your patient a diet sheet and that’s all."
Many celiac patients are already nutritionally compromised at the time of diagnosis. "This is the time to measure their nutritional parameters," Dr. Crowe said. "They may need supplements. Many are deficient in vitamin D, iron, folate, zinc, or other trace elements."
The risks of untreated disease "are not inconsequential." Patients can develop problems related to nutrient malabsorption, including osteopenia, infertility, miscarriage, and intrauterine growth restriction, and are four times more likely than the general population to develop a malignancy.
Guidelines for managing nutritional status exist, but there are no evidence-based treatment guidelines for celiac disease, Dr. Crowe said. "Aside from nutrition, you have to just advise them to follow good health practices. I have several patients who still smoke, and yet they’re morose about their gluten-free diet. I say, ‘Why are you worried about that? You need to worry about quitting smoking!’ "
Dr. Crowe reported that she receives royalties from a book written for patients with celiac disease.
SAN DIEGO – An intestinal biopsy is almost always necessary to confirm celiac disease and is a must before committing a patient to the only effective treatment – a lifelong gluten-free diet.
Sticking to such a restricted diet is difficult and expensive, Dr. Sheila Crowe said at the annual meeting of the American College of Physicians. "A lifelong gluten-free diet sounds simple, unless you’re the patient. ... Eating out is very difficult, especially for children and teens who face a lot of peer pressure. And eating gluten free at home is expensive. Studies in the United States, Canada, and the United Kingdom confirm that a lifelong diet of gluten-free foods costs about three times more than a normal diet," said Dr. Crowe, professor in the division of gastroenterology and hepatology at the University of Virginia, Charlottesville.
Because treating celiac disease requires this lifelong commitment, a positive serologic test isn’t enough to rule it in, she said. Nor are any of the available immunologic tests, including the most widely used – tissue transglutaminase IgA (tTG IgA) – specific enough to replace intestinal biopsy as the sole method for reliably diagnosing celiac disease. "A positive tTG test is not enough to place a person on this lifelong treatment without confirmation from an intestinal biopsy," she said. "This is especially important for children, because of the higher likelihood of false positives in that group."
tTG IgA has a very high sensitivity and specificity, but it isn’t perfect, Dr. Crowe said. "If you have a patient with clinical symptoms and the tTG comes back negative, there is still a 10% chance that’s a false negative. Another scenario could be a patient who has an autoimmune disease or a relative with celiac, and is experiencing celiac symptoms. If the tTG came back negative on that person, I would still do an endoscopy."
The only possible exception might be a patient with celiac symptoms who already has biopsy-proven dermatitis herpetiformis, with the classic immunofluorescent deposits at the dermal-epidermal junction. "If you biopsy these patients, the intestine will show the changes associated with celiac disease every time," Dr. Crowe said.
Celiac disease is no longer considered a disorder of childhood. "The disease is there lifelong. It appears you cannot suppress the immune response," she said.
The intestine rapidly responds to a gluten-free diet, "But the tendency to have an immunologic response to gluten is always there," Dr. Crowe said.
Relapses are common. Patients are most likely to "fall off" the diet when symptoms begin to abate, she said. They may simply feel "cured" and resume old eating patterns, or they may drop the diet because of changes that can occur as the intestine heals. "If a patient had diarrhea and malabsorption of nutrients, they might find themselves getting constipated and gaining weight on the gluten-free diet, fall off, and get ill again. Even if the intestine is healed, the vast majority of data tell us that patient will relapse," at some point after abandoning the dietary restriction, Dr. Crowe said.
This can lead to the development of refractory celiac disease, in which the intestine fails to recover despite a gluten-free diet. Patients with refractory celiac disease may be unable to fully absorb nutrients and need supplemental feeding methods.
The goal of celiac management is to promote intestinal healing, optimize nutrition, and avoid long-term damage, Dr. Crowe said. "It’s key to bring in a knowledgeable dietitian to help. And I mean knowledgeable – not someone who is going to hand your patient a diet sheet and that’s all."
Many celiac patients are already nutritionally compromised at the time of diagnosis. "This is the time to measure their nutritional parameters," Dr. Crowe said. "They may need supplements. Many are deficient in vitamin D, iron, folate, zinc, or other trace elements."
The risks of untreated disease "are not inconsequential." Patients can develop problems related to nutrient malabsorption, including osteopenia, infertility, miscarriage, and intrauterine growth restriction, and are four times more likely than the general population to develop a malignancy.
Guidelines for managing nutritional status exist, but there are no evidence-based treatment guidelines for celiac disease, Dr. Crowe said. "Aside from nutrition, you have to just advise them to follow good health practices. I have several patients who still smoke, and yet they’re morose about their gluten-free diet. I say, ‘Why are you worried about that? You need to worry about quitting smoking!’ "
Dr. Crowe reported that she receives royalties from a book written for patients with celiac disease.
SAN DIEGO – An intestinal biopsy is almost always necessary to confirm celiac disease and is a must before committing a patient to the only effective treatment – a lifelong gluten-free diet.
Sticking to such a restricted diet is difficult and expensive, Dr. Sheila Crowe said at the annual meeting of the American College of Physicians. "A lifelong gluten-free diet sounds simple, unless you’re the patient. ... Eating out is very difficult, especially for children and teens who face a lot of peer pressure. And eating gluten free at home is expensive. Studies in the United States, Canada, and the United Kingdom confirm that a lifelong diet of gluten-free foods costs about three times more than a normal diet," said Dr. Crowe, professor in the division of gastroenterology and hepatology at the University of Virginia, Charlottesville.
Because treating celiac disease requires this lifelong commitment, a positive serologic test isn’t enough to rule it in, she said. Nor are any of the available immunologic tests, including the most widely used – tissue transglutaminase IgA (tTG IgA) – specific enough to replace intestinal biopsy as the sole method for reliably diagnosing celiac disease. "A positive tTG test is not enough to place a person on this lifelong treatment without confirmation from an intestinal biopsy," she said. "This is especially important for children, because of the higher likelihood of false positives in that group."
tTG IgA has a very high sensitivity and specificity, but it isn’t perfect, Dr. Crowe said. "If you have a patient with clinical symptoms and the tTG comes back negative, there is still a 10% chance that’s a false negative. Another scenario could be a patient who has an autoimmune disease or a relative with celiac, and is experiencing celiac symptoms. If the tTG came back negative on that person, I would still do an endoscopy."
The only possible exception might be a patient with celiac symptoms who already has biopsy-proven dermatitis herpetiformis, with the classic immunofluorescent deposits at the dermal-epidermal junction. "If you biopsy these patients, the intestine will show the changes associated with celiac disease every time," Dr. Crowe said.
Celiac disease is no longer considered a disorder of childhood. "The disease is there lifelong. It appears you cannot suppress the immune response," she said.
The intestine rapidly responds to a gluten-free diet, "But the tendency to have an immunologic response to gluten is always there," Dr. Crowe said.
Relapses are common. Patients are most likely to "fall off" the diet when symptoms begin to abate, she said. They may simply feel "cured" and resume old eating patterns, or they may drop the diet because of changes that can occur as the intestine heals. "If a patient had diarrhea and malabsorption of nutrients, they might find themselves getting constipated and gaining weight on the gluten-free diet, fall off, and get ill again. Even if the intestine is healed, the vast majority of data tell us that patient will relapse," at some point after abandoning the dietary restriction, Dr. Crowe said.
This can lead to the development of refractory celiac disease, in which the intestine fails to recover despite a gluten-free diet. Patients with refractory celiac disease may be unable to fully absorb nutrients and need supplemental feeding methods.
The goal of celiac management is to promote intestinal healing, optimize nutrition, and avoid long-term damage, Dr. Crowe said. "It’s key to bring in a knowledgeable dietitian to help. And I mean knowledgeable – not someone who is going to hand your patient a diet sheet and that’s all."
Many celiac patients are already nutritionally compromised at the time of diagnosis. "This is the time to measure their nutritional parameters," Dr. Crowe said. "They may need supplements. Many are deficient in vitamin D, iron, folate, zinc, or other trace elements."
The risks of untreated disease "are not inconsequential." Patients can develop problems related to nutrient malabsorption, including osteopenia, infertility, miscarriage, and intrauterine growth restriction, and are four times more likely than the general population to develop a malignancy.
Guidelines for managing nutritional status exist, but there are no evidence-based treatment guidelines for celiac disease, Dr. Crowe said. "Aside from nutrition, you have to just advise them to follow good health practices. I have several patients who still smoke, and yet they’re morose about their gluten-free diet. I say, ‘Why are you worried about that? You need to worry about quitting smoking!’ "
Dr. Crowe reported that she receives royalties from a book written for patients with celiac disease.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF PHYSICIANS
Confirm Celiac Diagnosis by Biopsy Before Advising Gluten-Free Diet
SAN DIEGO – An intestinal biopsy is almost always necessary to confirm celiac disease and is a must before committing a patient to the only effective treatment – a lifelong gluten-free diet.
Sticking to such a restricted diet is difficult and expensive, Dr. Sheila Crowe said at the annual meeting of the American College of Physicians. "A lifelong gluten-free diet sounds simple, unless you’re the patient. ... Eating out is very difficult, especially for children and teens who face a lot of peer pressure. And eating gluten free at home is expensive. Studies in the United States, Canada, and the United Kingdom confirm that a lifelong diet of gluten-free foods costs about three times more than a normal diet," said Dr. Crowe, professor in the division of gastroenterology and hepatology at the University of Virginia, Charlottesville.
Because treating celiac disease requires this lifelong commitment, a positive serologic test isn’t enough to rule it in, she said. Nor are any of the available immunologic tests, including the most widely used – tissue transglutaminase IgA (tTG IgA) – specific enough to replace intestinal biopsy as the sole method for reliably diagnosing celiac disease. "A positive tTG test is not enough to place a person on this lifelong treatment without confirmation from an intestinal biopsy," she said. "This is especially important for children, because of the higher likelihood of false positives in that group."
tTG IgA has a very high sensitivity and specificity, but it isn’t perfect, Dr. Crowe said. "If you have a patient with clinical symptoms and the tTG comes back negative, there is still a 10% chance that’s a false negative. Another scenario could be a patient who has an autoimmune disease or a relative with celiac, and is experiencing celiac symptoms. If the tTG came back negative on that person, I would still do an endoscopy."
The only possible exception might be a patient with celiac symptoms who already has biopsy-proven dermatitis herpetiformis, with the classic immunofluorescent deposits at the dermal-epidermal junction. "If you biopsy these patients, the intestine will show the changes associated with celiac disease every time," Dr. Crowe said.
Celiac disease is no longer considered a disorder of childhood. "The disease is there lifelong. It appears you cannot suppress the immune response," she said.
The intestine rapidly responds to a gluten-free diet, "But the tendency to have an immunologic response to gluten is always there," Dr. Crowe said.
Relapses are common. Patients are most likely to "fall off" the diet when symptoms begin to abate, she said. They may simply feel "cured" and resume old eating patterns, or they may drop the diet because of changes that can occur as the intestine heals. "If a patient had diarrhea and malabsorption of nutrients, they might find themselves getting constipated and gaining weight on the gluten-free diet, fall off, and get ill again. Even if the intestine is healed, the vast majority of data tell us that patient will relapse," at some point after abandoning the dietary restriction, Dr. Crowe said.
This can lead to the development of refractory celiac disease, in which the intestine fails to recover despite a gluten-free diet. Patients with refractory celiac disease may be unable to fully absorb nutrients and need supplemental feeding methods.
The goal of celiac management is to promote intestinal healing, optimize nutrition, and avoid long-term damage, Dr. Crowe said. "It’s key to bring in a knowledgeable dietitian to help. And I mean knowledgeable – not someone who is going to hand your patient a diet sheet and that’s all."
Many celiac patients are already nutritionally compromised at the time of diagnosis. "This is the time to measure their nutritional parameters," Dr. Crowe said. "They may need supplements. Many are deficient in vitamin D, iron, folate, zinc, or other trace elements."
The risks of untreated disease "are not inconsequential." Patients can develop problems related to nutrient malabsorption, including osteopenia, infertility, miscarriage, and intrauterine growth restriction, and are four times more likely than the general population to develop a malignancy.
Guidelines for managing nutritional status exist, but there are no evidence-based treatment guidelines for celiac disease, Dr. Crowe said. "Aside from nutrition, you have to just advise them to follow good health practices. I have several patients who still smoke, and yet they’re morose about their gluten-free diet. I say, ‘Why are you worried about that? You need to worry about quitting smoking!’ "
Dr. Crowe reported that she receives royalties from a book written for patients with celiac disease.
SAN DIEGO – An intestinal biopsy is almost always necessary to confirm celiac disease and is a must before committing a patient to the only effective treatment – a lifelong gluten-free diet.
Sticking to such a restricted diet is difficult and expensive, Dr. Sheila Crowe said at the annual meeting of the American College of Physicians. "A lifelong gluten-free diet sounds simple, unless you’re the patient. ... Eating out is very difficult, especially for children and teens who face a lot of peer pressure. And eating gluten free at home is expensive. Studies in the United States, Canada, and the United Kingdom confirm that a lifelong diet of gluten-free foods costs about three times more than a normal diet," said Dr. Crowe, professor in the division of gastroenterology and hepatology at the University of Virginia, Charlottesville.
Because treating celiac disease requires this lifelong commitment, a positive serologic test isn’t enough to rule it in, she said. Nor are any of the available immunologic tests, including the most widely used – tissue transglutaminase IgA (tTG IgA) – specific enough to replace intestinal biopsy as the sole method for reliably diagnosing celiac disease. "A positive tTG test is not enough to place a person on this lifelong treatment without confirmation from an intestinal biopsy," she said. "This is especially important for children, because of the higher likelihood of false positives in that group."
tTG IgA has a very high sensitivity and specificity, but it isn’t perfect, Dr. Crowe said. "If you have a patient with clinical symptoms and the tTG comes back negative, there is still a 10% chance that’s a false negative. Another scenario could be a patient who has an autoimmune disease or a relative with celiac, and is experiencing celiac symptoms. If the tTG came back negative on that person, I would still do an endoscopy."
The only possible exception might be a patient with celiac symptoms who already has biopsy-proven dermatitis herpetiformis, with the classic immunofluorescent deposits at the dermal-epidermal junction. "If you biopsy these patients, the intestine will show the changes associated with celiac disease every time," Dr. Crowe said.
Celiac disease is no longer considered a disorder of childhood. "The disease is there lifelong. It appears you cannot suppress the immune response," she said.
The intestine rapidly responds to a gluten-free diet, "But the tendency to have an immunologic response to gluten is always there," Dr. Crowe said.
Relapses are common. Patients are most likely to "fall off" the diet when symptoms begin to abate, she said. They may simply feel "cured" and resume old eating patterns, or they may drop the diet because of changes that can occur as the intestine heals. "If a patient had diarrhea and malabsorption of nutrients, they might find themselves getting constipated and gaining weight on the gluten-free diet, fall off, and get ill again. Even if the intestine is healed, the vast majority of data tell us that patient will relapse," at some point after abandoning the dietary restriction, Dr. Crowe said.
This can lead to the development of refractory celiac disease, in which the intestine fails to recover despite a gluten-free diet. Patients with refractory celiac disease may be unable to fully absorb nutrients and need supplemental feeding methods.
The goal of celiac management is to promote intestinal healing, optimize nutrition, and avoid long-term damage, Dr. Crowe said. "It’s key to bring in a knowledgeable dietitian to help. And I mean knowledgeable – not someone who is going to hand your patient a diet sheet and that’s all."
Many celiac patients are already nutritionally compromised at the time of diagnosis. "This is the time to measure their nutritional parameters," Dr. Crowe said. "They may need supplements. Many are deficient in vitamin D, iron, folate, zinc, or other trace elements."
The risks of untreated disease "are not inconsequential." Patients can develop problems related to nutrient malabsorption, including osteopenia, infertility, miscarriage, and intrauterine growth restriction, and are four times more likely than the general population to develop a malignancy.
Guidelines for managing nutritional status exist, but there are no evidence-based treatment guidelines for celiac disease, Dr. Crowe said. "Aside from nutrition, you have to just advise them to follow good health practices. I have several patients who still smoke, and yet they’re morose about their gluten-free diet. I say, ‘Why are you worried about that? You need to worry about quitting smoking!’ "
Dr. Crowe reported that she receives royalties from a book written for patients with celiac disease.
SAN DIEGO – An intestinal biopsy is almost always necessary to confirm celiac disease and is a must before committing a patient to the only effective treatment – a lifelong gluten-free diet.
Sticking to such a restricted diet is difficult and expensive, Dr. Sheila Crowe said at the annual meeting of the American College of Physicians. "A lifelong gluten-free diet sounds simple, unless you’re the patient. ... Eating out is very difficult, especially for children and teens who face a lot of peer pressure. And eating gluten free at home is expensive. Studies in the United States, Canada, and the United Kingdom confirm that a lifelong diet of gluten-free foods costs about three times more than a normal diet," said Dr. Crowe, professor in the division of gastroenterology and hepatology at the University of Virginia, Charlottesville.
Because treating celiac disease requires this lifelong commitment, a positive serologic test isn’t enough to rule it in, she said. Nor are any of the available immunologic tests, including the most widely used – tissue transglutaminase IgA (tTG IgA) – specific enough to replace intestinal biopsy as the sole method for reliably diagnosing celiac disease. "A positive tTG test is not enough to place a person on this lifelong treatment without confirmation from an intestinal biopsy," she said. "This is especially important for children, because of the higher likelihood of false positives in that group."
tTG IgA has a very high sensitivity and specificity, but it isn’t perfect, Dr. Crowe said. "If you have a patient with clinical symptoms and the tTG comes back negative, there is still a 10% chance that’s a false negative. Another scenario could be a patient who has an autoimmune disease or a relative with celiac, and is experiencing celiac symptoms. If the tTG came back negative on that person, I would still do an endoscopy."
The only possible exception might be a patient with celiac symptoms who already has biopsy-proven dermatitis herpetiformis, with the classic immunofluorescent deposits at the dermal-epidermal junction. "If you biopsy these patients, the intestine will show the changes associated with celiac disease every time," Dr. Crowe said.
Celiac disease is no longer considered a disorder of childhood. "The disease is there lifelong. It appears you cannot suppress the immune response," she said.
The intestine rapidly responds to a gluten-free diet, "But the tendency to have an immunologic response to gluten is always there," Dr. Crowe said.
Relapses are common. Patients are most likely to "fall off" the diet when symptoms begin to abate, she said. They may simply feel "cured" and resume old eating patterns, or they may drop the diet because of changes that can occur as the intestine heals. "If a patient had diarrhea and malabsorption of nutrients, they might find themselves getting constipated and gaining weight on the gluten-free diet, fall off, and get ill again. Even if the intestine is healed, the vast majority of data tell us that patient will relapse," at some point after abandoning the dietary restriction, Dr. Crowe said.
This can lead to the development of refractory celiac disease, in which the intestine fails to recover despite a gluten-free diet. Patients with refractory celiac disease may be unable to fully absorb nutrients and need supplemental feeding methods.
The goal of celiac management is to promote intestinal healing, optimize nutrition, and avoid long-term damage, Dr. Crowe said. "It’s key to bring in a knowledgeable dietitian to help. And I mean knowledgeable – not someone who is going to hand your patient a diet sheet and that’s all."
Many celiac patients are already nutritionally compromised at the time of diagnosis. "This is the time to measure their nutritional parameters," Dr. Crowe said. "They may need supplements. Many are deficient in vitamin D, iron, folate, zinc, or other trace elements."
The risks of untreated disease "are not inconsequential." Patients can develop problems related to nutrient malabsorption, including osteopenia, infertility, miscarriage, and intrauterine growth restriction, and are four times more likely than the general population to develop a malignancy.
Guidelines for managing nutritional status exist, but there are no evidence-based treatment guidelines for celiac disease, Dr. Crowe said. "Aside from nutrition, you have to just advise them to follow good health practices. I have several patients who still smoke, and yet they’re morose about their gluten-free diet. I say, ‘Why are you worried about that? You need to worry about quitting smoking!’ "
Dr. Crowe reported that she receives royalties from a book written for patients with celiac disease.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF PHYSICIANS
Delay in Mammography May Put Young Women at Risk
WASHINGTON – Younger women may suffer under new national mammography screening guidelines that recommend that the procedure become biennial and begin at age 50 years, according to investigators who conducted a retrospective study of breast cancer patients in the 40- to 49-year age group.
Breast tumors that arise in this group may not be discovered until they present clinically, at which time treatment will be more expensive and curative therapy perhaps impossible, lead author Dr. Paul Dale said at the annual meeting of the American Society of Breast Surgeons.
"Our study found that tumors identified through mammography generally had better outcomes after treatment [than did] those found through clinical exam," Dr. Dale said at a press briefing. "Breast cancer has a better prognosis when treated before tumors become palpable and identifiable" through a physician- or self-exam of the breast.
The 10-year retrospective study found that women aged 40-49 years who presented with a breast cancer through clinical symptoms or palpation had significantly larger tumor size, more nodal involvement, and lower 5-year survival rates than did a similarly aged group whose cancers were detected through mammography.
The study comprised 311 women aged 40-49 years who were treated for breast cancer at a single center in 2004-2008. Of these, 145 (47%) had undergone a screening mammography that detected the tumor, whereas 166 (53%) had a tumor that presented clinically, either by symptoms or by physician- or self-exam of the breast.
Tumors in the mammography group were significantly smaller than those among the clinically presenting group (median, 2 cm vs. 3 cm; P less than or equal to .001). Sentinel lymph node involvement occurred in significantly fewer of those in the mammography group (28 vs. 115; P less than or equal to .001)
The 5-year disease-free survival rate was significantly better in the mammographically detected group (94% vs. 71%); their 5-year overall survival was also significantly greater (97% vs. 78%).
These advantages occurred despite the fact that significantly more women in the mammographically detected group had a family history of breast cancer (25% vs. 15%; P = .034).
A multivariate analysis found that mammographic cancer detection, node negativity, and smaller tumor size were all significantly associated with an increase overall survival.
"In our institution, we find that 20% of the women diagnosed with breast cancer are younger than age 50," said Dr. Dale, chief of surgical oncology at the University of Missouri–Columbia. Both the findings of this study and his own clinical experience have convinced him that annual mammographic screening has "great value" to this younger set of women, despite the 2010 U.S. Preventive Services Task Force (USPSTF) recommendation that biennial screening mammograms begin at age 50.
The agency recommended this screening regimen for women aged 50-75 years, but said that for women aged 40-49 years the benefit of screening is small and is balanced by "moderate harms," including false positives that lead to unnecessary invasive interventions, anxiety, and the small impact of pain from biopsy and radiation exposure.
The statement was largely informed by a 2009 review of the SEER (Surveillance Epidemiology and End Results) database. That review concluded that among women aged 40-49, the number needed to treat to prevent one breast cancer death was 1,904, compared with 1,339 for women aged 50-59.
"Although the relative risk reduction is nearly identical (15% and 14%) for these two age groups, the risk for breast cancer increases steeply with age starting at age 40 years," the document stated. "Thus, the absolute risk reduction from screening ... is greater for women aged 50-59 years than for those aged 40-49 years."
However, the USPSTF document did not recommend against earlier screening, saying that the decision should be based on a woman’s family history of the disease and her individual desires, and only after a discussion about the relative risks and benefits.
In an interview, Dr. Dale debated this approach, saying that "when it’s your cancer, it matters a lot.
"I have been doing this for 20 years, and of all the women I have put through a breast biopsy because of something suspicious identified on a screening mammogram, I can tell you that 100% of those with a negative result were glad they did it. The woman’s level of comfort in hearing that is huge," he added.
A 2011 study supports the idea that screening more women will save more lives, Dr. Dale said, referring to another analysis of the same SEER data. Dr. Edward Hendrick of the University of Colorado at Denver and colleagues, concluded that annual screening for women aged 40-84 years would result in a 71% greater mortality reduction than the USPSTF recommendation of biennial screening in those aged 50-74 years. An annual screening for women aged 40-84 years would save almost 100,000 more lives, the authors argued (AJR 2011;196:W112-6).
Dr. Dale also suggested that resource allocation and federal funding concerns may have at least partially motivated the government study. "The government is paying the brunt of this, and annual screening runs into the billions," he said. "But if you really look at the economics of it, and the years of life it can save – the fact that these women are not undergoing the much more expensive therapies [of treating more advanced cancer], and the economic benefit their productive lives give our economy – the economic picture doesn’t look that bad."
Another 2011 study supports this conclusion, he said. Dr. Blake Cady of the Cambridge (Mass.) Breast Center and associates suggested that financial resources were a driving point of the recommendations.
The annual cost of an additional 25,000 mammograms could well be offset by an estimated $50,000-$100,000 per life saved, they said. "Why the USPSTF deliberately chose a less effective method of preventing mortality in the most frequent and feared cancer of women is a puzzle, especially as cost considerations may not be a major adverse factor, although resource allocation is increased," they concluded (Ann. Surg. Oncol. 2011;18:903-6).
The American Cancer Society, American College of Surgeons, and American College of Obstetricians and Gynecologists still recommend either annual or biennial screening for women, beginning at age 40.
Dr. Dale declared no financial conflicts with regard to his study.
WASHINGTON – Younger women may suffer under new national mammography screening guidelines that recommend that the procedure become biennial and begin at age 50 years, according to investigators who conducted a retrospective study of breast cancer patients in the 40- to 49-year age group.
Breast tumors that arise in this group may not be discovered until they present clinically, at which time treatment will be more expensive and curative therapy perhaps impossible, lead author Dr. Paul Dale said at the annual meeting of the American Society of Breast Surgeons.
"Our study found that tumors identified through mammography generally had better outcomes after treatment [than did] those found through clinical exam," Dr. Dale said at a press briefing. "Breast cancer has a better prognosis when treated before tumors become palpable and identifiable" through a physician- or self-exam of the breast.
The 10-year retrospective study found that women aged 40-49 years who presented with a breast cancer through clinical symptoms or palpation had significantly larger tumor size, more nodal involvement, and lower 5-year survival rates than did a similarly aged group whose cancers were detected through mammography.
The study comprised 311 women aged 40-49 years who were treated for breast cancer at a single center in 2004-2008. Of these, 145 (47%) had undergone a screening mammography that detected the tumor, whereas 166 (53%) had a tumor that presented clinically, either by symptoms or by physician- or self-exam of the breast.
Tumors in the mammography group were significantly smaller than those among the clinically presenting group (median, 2 cm vs. 3 cm; P less than or equal to .001). Sentinel lymph node involvement occurred in significantly fewer of those in the mammography group (28 vs. 115; P less than or equal to .001)
The 5-year disease-free survival rate was significantly better in the mammographically detected group (94% vs. 71%); their 5-year overall survival was also significantly greater (97% vs. 78%).
These advantages occurred despite the fact that significantly more women in the mammographically detected group had a family history of breast cancer (25% vs. 15%; P = .034).
A multivariate analysis found that mammographic cancer detection, node negativity, and smaller tumor size were all significantly associated with an increase overall survival.
"In our institution, we find that 20% of the women diagnosed with breast cancer are younger than age 50," said Dr. Dale, chief of surgical oncology at the University of Missouri–Columbia. Both the findings of this study and his own clinical experience have convinced him that annual mammographic screening has "great value" to this younger set of women, despite the 2010 U.S. Preventive Services Task Force (USPSTF) recommendation that biennial screening mammograms begin at age 50.
The agency recommended this screening regimen for women aged 50-75 years, but said that for women aged 40-49 years the benefit of screening is small and is balanced by "moderate harms," including false positives that lead to unnecessary invasive interventions, anxiety, and the small impact of pain from biopsy and radiation exposure.
The statement was largely informed by a 2009 review of the SEER (Surveillance Epidemiology and End Results) database. That review concluded that among women aged 40-49, the number needed to treat to prevent one breast cancer death was 1,904, compared with 1,339 for women aged 50-59.
"Although the relative risk reduction is nearly identical (15% and 14%) for these two age groups, the risk for breast cancer increases steeply with age starting at age 40 years," the document stated. "Thus, the absolute risk reduction from screening ... is greater for women aged 50-59 years than for those aged 40-49 years."
However, the USPSTF document did not recommend against earlier screening, saying that the decision should be based on a woman’s family history of the disease and her individual desires, and only after a discussion about the relative risks and benefits.
In an interview, Dr. Dale debated this approach, saying that "when it’s your cancer, it matters a lot.
"I have been doing this for 20 years, and of all the women I have put through a breast biopsy because of something suspicious identified on a screening mammogram, I can tell you that 100% of those with a negative result were glad they did it. The woman’s level of comfort in hearing that is huge," he added.
A 2011 study supports the idea that screening more women will save more lives, Dr. Dale said, referring to another analysis of the same SEER data. Dr. Edward Hendrick of the University of Colorado at Denver and colleagues, concluded that annual screening for women aged 40-84 years would result in a 71% greater mortality reduction than the USPSTF recommendation of biennial screening in those aged 50-74 years. An annual screening for women aged 40-84 years would save almost 100,000 more lives, the authors argued (AJR 2011;196:W112-6).
Dr. Dale also suggested that resource allocation and federal funding concerns may have at least partially motivated the government study. "The government is paying the brunt of this, and annual screening runs into the billions," he said. "But if you really look at the economics of it, and the years of life it can save – the fact that these women are not undergoing the much more expensive therapies [of treating more advanced cancer], and the economic benefit their productive lives give our economy – the economic picture doesn’t look that bad."
Another 2011 study supports this conclusion, he said. Dr. Blake Cady of the Cambridge (Mass.) Breast Center and associates suggested that financial resources were a driving point of the recommendations.
The annual cost of an additional 25,000 mammograms could well be offset by an estimated $50,000-$100,000 per life saved, they said. "Why the USPSTF deliberately chose a less effective method of preventing mortality in the most frequent and feared cancer of women is a puzzle, especially as cost considerations may not be a major adverse factor, although resource allocation is increased," they concluded (Ann. Surg. Oncol. 2011;18:903-6).
The American Cancer Society, American College of Surgeons, and American College of Obstetricians and Gynecologists still recommend either annual or biennial screening for women, beginning at age 40.
Dr. Dale declared no financial conflicts with regard to his study.
WASHINGTON – Younger women may suffer under new national mammography screening guidelines that recommend that the procedure become biennial and begin at age 50 years, according to investigators who conducted a retrospective study of breast cancer patients in the 40- to 49-year age group.
Breast tumors that arise in this group may not be discovered until they present clinically, at which time treatment will be more expensive and curative therapy perhaps impossible, lead author Dr. Paul Dale said at the annual meeting of the American Society of Breast Surgeons.
"Our study found that tumors identified through mammography generally had better outcomes after treatment [than did] those found through clinical exam," Dr. Dale said at a press briefing. "Breast cancer has a better prognosis when treated before tumors become palpable and identifiable" through a physician- or self-exam of the breast.
The 10-year retrospective study found that women aged 40-49 years who presented with a breast cancer through clinical symptoms or palpation had significantly larger tumor size, more nodal involvement, and lower 5-year survival rates than did a similarly aged group whose cancers were detected through mammography.
The study comprised 311 women aged 40-49 years who were treated for breast cancer at a single center in 2004-2008. Of these, 145 (47%) had undergone a screening mammography that detected the tumor, whereas 166 (53%) had a tumor that presented clinically, either by symptoms or by physician- or self-exam of the breast.
Tumors in the mammography group were significantly smaller than those among the clinically presenting group (median, 2 cm vs. 3 cm; P less than or equal to .001). Sentinel lymph node involvement occurred in significantly fewer of those in the mammography group (28 vs. 115; P less than or equal to .001)
The 5-year disease-free survival rate was significantly better in the mammographically detected group (94% vs. 71%); their 5-year overall survival was also significantly greater (97% vs. 78%).
These advantages occurred despite the fact that significantly more women in the mammographically detected group had a family history of breast cancer (25% vs. 15%; P = .034).
A multivariate analysis found that mammographic cancer detection, node negativity, and smaller tumor size were all significantly associated with an increase overall survival.
"In our institution, we find that 20% of the women diagnosed with breast cancer are younger than age 50," said Dr. Dale, chief of surgical oncology at the University of Missouri–Columbia. Both the findings of this study and his own clinical experience have convinced him that annual mammographic screening has "great value" to this younger set of women, despite the 2010 U.S. Preventive Services Task Force (USPSTF) recommendation that biennial screening mammograms begin at age 50.
The agency recommended this screening regimen for women aged 50-75 years, but said that for women aged 40-49 years the benefit of screening is small and is balanced by "moderate harms," including false positives that lead to unnecessary invasive interventions, anxiety, and the small impact of pain from biopsy and radiation exposure.
The statement was largely informed by a 2009 review of the SEER (Surveillance Epidemiology and End Results) database. That review concluded that among women aged 40-49, the number needed to treat to prevent one breast cancer death was 1,904, compared with 1,339 for women aged 50-59.
"Although the relative risk reduction is nearly identical (15% and 14%) for these two age groups, the risk for breast cancer increases steeply with age starting at age 40 years," the document stated. "Thus, the absolute risk reduction from screening ... is greater for women aged 50-59 years than for those aged 40-49 years."
However, the USPSTF document did not recommend against earlier screening, saying that the decision should be based on a woman’s family history of the disease and her individual desires, and only after a discussion about the relative risks and benefits.
In an interview, Dr. Dale debated this approach, saying that "when it’s your cancer, it matters a lot.
"I have been doing this for 20 years, and of all the women I have put through a breast biopsy because of something suspicious identified on a screening mammogram, I can tell you that 100% of those with a negative result were glad they did it. The woman’s level of comfort in hearing that is huge," he added.
A 2011 study supports the idea that screening more women will save more lives, Dr. Dale said, referring to another analysis of the same SEER data. Dr. Edward Hendrick of the University of Colorado at Denver and colleagues, concluded that annual screening for women aged 40-84 years would result in a 71% greater mortality reduction than the USPSTF recommendation of biennial screening in those aged 50-74 years. An annual screening for women aged 40-84 years would save almost 100,000 more lives, the authors argued (AJR 2011;196:W112-6).
Dr. Dale also suggested that resource allocation and federal funding concerns may have at least partially motivated the government study. "The government is paying the brunt of this, and annual screening runs into the billions," he said. "But if you really look at the economics of it, and the years of life it can save – the fact that these women are not undergoing the much more expensive therapies [of treating more advanced cancer], and the economic benefit their productive lives give our economy – the economic picture doesn’t look that bad."
Another 2011 study supports this conclusion, he said. Dr. Blake Cady of the Cambridge (Mass.) Breast Center and associates suggested that financial resources were a driving point of the recommendations.
The annual cost of an additional 25,000 mammograms could well be offset by an estimated $50,000-$100,000 per life saved, they said. "Why the USPSTF deliberately chose a less effective method of preventing mortality in the most frequent and feared cancer of women is a puzzle, especially as cost considerations may not be a major adverse factor, although resource allocation is increased," they concluded (Ann. Surg. Oncol. 2011;18:903-6).
The American Cancer Society, American College of Surgeons, and American College of Obstetricians and Gynecologists still recommend either annual or biennial screening for women, beginning at age 40.
Dr. Dale declared no financial conflicts with regard to his study.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF BREAST SURGEONS
Major Finding: The 5-year disease-free survival rate was significantly better when breast tumors were detected by mammography (94% vs. 71% when tumors presented clinically), as was 5-year overall survival (97% vs. 78%).
Data Source: A 10-year retrospective study of 311 women aged 40-49 years who were treated for breast cancer.
Disclosures: Dr. Dale declared no financial conflicts with regard to his study.
Delay in Mammography May Put Young Women at Risk
WASHINGTON – Younger women may suffer under new national mammography screening guidelines that recommend that the procedure become biennial and begin at age 50 years, according to investigators who conducted a retrospective study of breast cancer patients in the 40- to 49-year age group.
Breast tumors that arise in this group may not be discovered until they present clinically, at which time treatment will be more expensive and curative therapy perhaps impossible, lead author Dr. Paul Dale said at the annual meeting of the American Society of Breast Surgeons.
"Our study found that tumors identified through mammography generally had better outcomes after treatment [than did] those found through clinical exam," Dr. Dale said at a press briefing. "Breast cancer has a better prognosis when treated before tumors become palpable and identifiable" through a physician- or self-exam of the breast.
The 10-year retrospective study found that women aged 40-49 years who presented with a breast cancer through clinical symptoms or palpation had significantly larger tumor size, more nodal involvement, and lower 5-year survival rates than did a similarly aged group whose cancers were detected through mammography.
The study comprised 311 women aged 40-49 years who were treated for breast cancer at a single center in 2004-2008. Of these, 145 (47%) had undergone a screening mammography that detected the tumor, whereas 166 (53%) had a tumor that presented clinically, either by symptoms or by physician- or self-exam of the breast.
Tumors in the mammography group were significantly smaller than those among the clinically presenting group (median, 2 cm vs. 3 cm; P less than or equal to .001). Sentinel lymph node involvement occurred in significantly fewer of those in the mammography group (28 vs. 115; P less than or equal to .001)
The 5-year disease-free survival rate was significantly better in the mammographically detected group (94% vs. 71%); their 5-year overall survival was also significantly greater (97% vs. 78%).
These advantages occurred despite the fact that significantly more women in the mammographically detected group had a family history of breast cancer (25% vs. 15%; P = .034).
A multivariate analysis found that mammographic cancer detection, node negativity, and smaller tumor size were all significantly associated with an increase overall survival.
"In our institution, we find that 20% of the women diagnosed with breast cancer are younger than age 50," said Dr. Dale, chief of surgical oncology at the University of Missouri–Columbia. Both the findings of this study and his own clinical experience have convinced him that annual mammographic screening has "great value" to this younger set of women, despite the 2010 U.S. Preventive Services Task Force (USPSTF) recommendation that biennial screening mammograms begin at age 50.
The agency recommended this screening regimen for women aged 50-75 years, but said that for women aged 40-49 years the benefit of screening is small and is balanced by "moderate harms," including false positives that lead to unnecessary invasive interventions, anxiety, and the small impact of pain from biopsy and radiation exposure.
The statement was largely informed by a 2009 review of the SEER (Surveillance Epidemiology and End Results) database. That review concluded that among women aged 40-49, the number needed to treat to prevent one breast cancer death was 1,904, compared with 1,339 for women aged 50-59.
"Although the relative risk reduction is nearly identical (15% and 14%) for these two age groups, the risk for breast cancer increases steeply with age starting at age 40 years," the document stated. "Thus, the absolute risk reduction from screening ... is greater for women aged 50-59 years than for those aged 40-49 years."
However, the USPSTF document did not recommend against earlier screening, saying that the decision should be based on a woman’s family history of the disease and her individual desires, and only after a discussion about the relative risks and benefits.
In an interview, Dr. Dale debated this approach, saying that "when it’s your cancer, it matters a lot.
"I have been doing this for 20 years, and of all the women I have put through a breast biopsy because of something suspicious identified on a screening mammogram, I can tell you that 100% of those with a negative result were glad they did it. The woman’s level of comfort in hearing that is huge," he added.
A 2011 study supports the idea that screening more women will save more lives, Dr. Dale said, referring to another analysis of the same SEER data. Dr. Edward Hendrick of the University of Colorado at Denver and colleagues, concluded that annual screening for women aged 40-84 years would result in a 71% greater mortality reduction than the USPSTF recommendation of biennial screening in those aged 50-74 years. An annual screening for women aged 40-84 years would save almost 100,000 more lives, the authors argued (AJR 2011;196:W112-6).
Dr. Dale also suggested that resource allocation and federal funding concerns may have at least partially motivated the government study. "The government is paying the brunt of this, and annual screening runs into the billions," he said. "But if you really look at the economics of it, and the years of life it can save – the fact that these women are not undergoing the much more expensive therapies [of treating more advanced cancer], and the economic benefit their productive lives give our economy – the economic picture doesn’t look that bad."
Another 2011 study supports this conclusion, he said. Dr. Blake Cady of the Cambridge (Mass.) Breast Center and associates suggested that financial resources were a driving point of the recommendations.
The annual cost of an additional 25,000 mammograms could well be offset by an estimated $50,000-$100,000 per life saved, they said. "Why the USPSTF deliberately chose a less effective method of preventing mortality in the most frequent and feared cancer of women is a puzzle, especially as cost considerations may not be a major adverse factor, although resource allocation is increased," they concluded (Ann. Surg. Oncol. 2011;18:903-6).
The American Cancer Society, American College of Surgeons, and American College of Obstetricians and Gynecologists still recommend either annual or biennial screening for women, beginning at age 40.
Dr. Dale declared no financial conflicts with regard to his study.
WASHINGTON – Younger women may suffer under new national mammography screening guidelines that recommend that the procedure become biennial and begin at age 50 years, according to investigators who conducted a retrospective study of breast cancer patients in the 40- to 49-year age group.
Breast tumors that arise in this group may not be discovered until they present clinically, at which time treatment will be more expensive and curative therapy perhaps impossible, lead author Dr. Paul Dale said at the annual meeting of the American Society of Breast Surgeons.
"Our study found that tumors identified through mammography generally had better outcomes after treatment [than did] those found through clinical exam," Dr. Dale said at a press briefing. "Breast cancer has a better prognosis when treated before tumors become palpable and identifiable" through a physician- or self-exam of the breast.
The 10-year retrospective study found that women aged 40-49 years who presented with a breast cancer through clinical symptoms or palpation had significantly larger tumor size, more nodal involvement, and lower 5-year survival rates than did a similarly aged group whose cancers were detected through mammography.
The study comprised 311 women aged 40-49 years who were treated for breast cancer at a single center in 2004-2008. Of these, 145 (47%) had undergone a screening mammography that detected the tumor, whereas 166 (53%) had a tumor that presented clinically, either by symptoms or by physician- or self-exam of the breast.
Tumors in the mammography group were significantly smaller than those among the clinically presenting group (median, 2 cm vs. 3 cm; P less than or equal to .001). Sentinel lymph node involvement occurred in significantly fewer of those in the mammography group (28 vs. 115; P less than or equal to .001)
The 5-year disease-free survival rate was significantly better in the mammographically detected group (94% vs. 71%); their 5-year overall survival was also significantly greater (97% vs. 78%).
These advantages occurred despite the fact that significantly more women in the mammographically detected group had a family history of breast cancer (25% vs. 15%; P = .034).
A multivariate analysis found that mammographic cancer detection, node negativity, and smaller tumor size were all significantly associated with an increase overall survival.
"In our institution, we find that 20% of the women diagnosed with breast cancer are younger than age 50," said Dr. Dale, chief of surgical oncology at the University of Missouri–Columbia. Both the findings of this study and his own clinical experience have convinced him that annual mammographic screening has "great value" to this younger set of women, despite the 2010 U.S. Preventive Services Task Force (USPSTF) recommendation that biennial screening mammograms begin at age 50.
The agency recommended this screening regimen for women aged 50-75 years, but said that for women aged 40-49 years the benefit of screening is small and is balanced by "moderate harms," including false positives that lead to unnecessary invasive interventions, anxiety, and the small impact of pain from biopsy and radiation exposure.
The statement was largely informed by a 2009 review of the SEER (Surveillance Epidemiology and End Results) database. That review concluded that among women aged 40-49, the number needed to treat to prevent one breast cancer death was 1,904, compared with 1,339 for women aged 50-59.
"Although the relative risk reduction is nearly identical (15% and 14%) for these two age groups, the risk for breast cancer increases steeply with age starting at age 40 years," the document stated. "Thus, the absolute risk reduction from screening ... is greater for women aged 50-59 years than for those aged 40-49 years."
However, the USPSTF document did not recommend against earlier screening, saying that the decision should be based on a woman’s family history of the disease and her individual desires, and only after a discussion about the relative risks and benefits.
In an interview, Dr. Dale debated this approach, saying that "when it’s your cancer, it matters a lot.
"I have been doing this for 20 years, and of all the women I have put through a breast biopsy because of something suspicious identified on a screening mammogram, I can tell you that 100% of those with a negative result were glad they did it. The woman’s level of comfort in hearing that is huge," he added.
A 2011 study supports the idea that screening more women will save more lives, Dr. Dale said, referring to another analysis of the same SEER data. Dr. Edward Hendrick of the University of Colorado at Denver and colleagues, concluded that annual screening for women aged 40-84 years would result in a 71% greater mortality reduction than the USPSTF recommendation of biennial screening in those aged 50-74 years. An annual screening for women aged 40-84 years would save almost 100,000 more lives, the authors argued (AJR 2011;196:W112-6).
Dr. Dale also suggested that resource allocation and federal funding concerns may have at least partially motivated the government study. "The government is paying the brunt of this, and annual screening runs into the billions," he said. "But if you really look at the economics of it, and the years of life it can save – the fact that these women are not undergoing the much more expensive therapies [of treating more advanced cancer], and the economic benefit their productive lives give our economy – the economic picture doesn’t look that bad."
Another 2011 study supports this conclusion, he said. Dr. Blake Cady of the Cambridge (Mass.) Breast Center and associates suggested that financial resources were a driving point of the recommendations.
The annual cost of an additional 25,000 mammograms could well be offset by an estimated $50,000-$100,000 per life saved, they said. "Why the USPSTF deliberately chose a less effective method of preventing mortality in the most frequent and feared cancer of women is a puzzle, especially as cost considerations may not be a major adverse factor, although resource allocation is increased," they concluded (Ann. Surg. Oncol. 2011;18:903-6).
The American Cancer Society, American College of Surgeons, and American College of Obstetricians and Gynecologists still recommend either annual or biennial screening for women, beginning at age 40.
Dr. Dale declared no financial conflicts with regard to his study.
WASHINGTON – Younger women may suffer under new national mammography screening guidelines that recommend that the procedure become biennial and begin at age 50 years, according to investigators who conducted a retrospective study of breast cancer patients in the 40- to 49-year age group.
Breast tumors that arise in this group may not be discovered until they present clinically, at which time treatment will be more expensive and curative therapy perhaps impossible, lead author Dr. Paul Dale said at the annual meeting of the American Society of Breast Surgeons.
"Our study found that tumors identified through mammography generally had better outcomes after treatment [than did] those found through clinical exam," Dr. Dale said at a press briefing. "Breast cancer has a better prognosis when treated before tumors become palpable and identifiable" through a physician- or self-exam of the breast.
The 10-year retrospective study found that women aged 40-49 years who presented with a breast cancer through clinical symptoms or palpation had significantly larger tumor size, more nodal involvement, and lower 5-year survival rates than did a similarly aged group whose cancers were detected through mammography.
The study comprised 311 women aged 40-49 years who were treated for breast cancer at a single center in 2004-2008. Of these, 145 (47%) had undergone a screening mammography that detected the tumor, whereas 166 (53%) had a tumor that presented clinically, either by symptoms or by physician- or self-exam of the breast.
Tumors in the mammography group were significantly smaller than those among the clinically presenting group (median, 2 cm vs. 3 cm; P less than or equal to .001). Sentinel lymph node involvement occurred in significantly fewer of those in the mammography group (28 vs. 115; P less than or equal to .001)
The 5-year disease-free survival rate was significantly better in the mammographically detected group (94% vs. 71%); their 5-year overall survival was also significantly greater (97% vs. 78%).
These advantages occurred despite the fact that significantly more women in the mammographically detected group had a family history of breast cancer (25% vs. 15%; P = .034).
A multivariate analysis found that mammographic cancer detection, node negativity, and smaller tumor size were all significantly associated with an increase overall survival.
"In our institution, we find that 20% of the women diagnosed with breast cancer are younger than age 50," said Dr. Dale, chief of surgical oncology at the University of Missouri–Columbia. Both the findings of this study and his own clinical experience have convinced him that annual mammographic screening has "great value" to this younger set of women, despite the 2010 U.S. Preventive Services Task Force (USPSTF) recommendation that biennial screening mammograms begin at age 50.
The agency recommended this screening regimen for women aged 50-75 years, but said that for women aged 40-49 years the benefit of screening is small and is balanced by "moderate harms," including false positives that lead to unnecessary invasive interventions, anxiety, and the small impact of pain from biopsy and radiation exposure.
The statement was largely informed by a 2009 review of the SEER (Surveillance Epidemiology and End Results) database. That review concluded that among women aged 40-49, the number needed to treat to prevent one breast cancer death was 1,904, compared with 1,339 for women aged 50-59.
"Although the relative risk reduction is nearly identical (15% and 14%) for these two age groups, the risk for breast cancer increases steeply with age starting at age 40 years," the document stated. "Thus, the absolute risk reduction from screening ... is greater for women aged 50-59 years than for those aged 40-49 years."
However, the USPSTF document did not recommend against earlier screening, saying that the decision should be based on a woman’s family history of the disease and her individual desires, and only after a discussion about the relative risks and benefits.
In an interview, Dr. Dale debated this approach, saying that "when it’s your cancer, it matters a lot.
"I have been doing this for 20 years, and of all the women I have put through a breast biopsy because of something suspicious identified on a screening mammogram, I can tell you that 100% of those with a negative result were glad they did it. The woman’s level of comfort in hearing that is huge," he added.
A 2011 study supports the idea that screening more women will save more lives, Dr. Dale said, referring to another analysis of the same SEER data. Dr. Edward Hendrick of the University of Colorado at Denver and colleagues, concluded that annual screening for women aged 40-84 years would result in a 71% greater mortality reduction than the USPSTF recommendation of biennial screening in those aged 50-74 years. An annual screening for women aged 40-84 years would save almost 100,000 more lives, the authors argued (AJR 2011;196:W112-6).
Dr. Dale also suggested that resource allocation and federal funding concerns may have at least partially motivated the government study. "The government is paying the brunt of this, and annual screening runs into the billions," he said. "But if you really look at the economics of it, and the years of life it can save – the fact that these women are not undergoing the much more expensive therapies [of treating more advanced cancer], and the economic benefit their productive lives give our economy – the economic picture doesn’t look that bad."
Another 2011 study supports this conclusion, he said. Dr. Blake Cady of the Cambridge (Mass.) Breast Center and associates suggested that financial resources were a driving point of the recommendations.
The annual cost of an additional 25,000 mammograms could well be offset by an estimated $50,000-$100,000 per life saved, they said. "Why the USPSTF deliberately chose a less effective method of preventing mortality in the most frequent and feared cancer of women is a puzzle, especially as cost considerations may not be a major adverse factor, although resource allocation is increased," they concluded (Ann. Surg. Oncol. 2011;18:903-6).
The American Cancer Society, American College of Surgeons, and American College of Obstetricians and Gynecologists still recommend either annual or biennial screening for women, beginning at age 40.
Dr. Dale declared no financial conflicts with regard to his study.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF BREAST SURGEONS
Major Finding: The 5-year disease-free survival rate was significantly better when breast tumors were detected by mammography (94% vs. 71% when tumors presented clinically), as was 5-year overall survival (97% vs. 78%).
Data Source: A 10-year retrospective study of 311 women aged 40-49 years who were treated for breast cancer.
Disclosures: Dr. Dale declared no financial conflicts with regard to his study.
Change in Mammography Guidelines May Adversely Affect Young Minority Women
WASHINGTON – The revised national screening mammography guidelines may especially impact the health of younger minority women for whom annual screening is no longer recommended, investigators suggested.
A retrospective study derived from a large state cancer registry found that Hispanic, Asian, and black women aged 40-49 years were up to 60% more likely to be diagnosed with ductal cancer in situ (DCIS) and up to 80% more like to have small invasive breast tumors (T1N0) than were their white counterparts.
These women were significantly more likely to have tumors that respond best to very early therapy, Dr. Sharon Lum said at the annual meeting of the American Society of Breast Surgeons. But if their cancers are not detected through mammography, women in these groups might not receive such therapy.
"We already know that breast cancer occurs at a younger age in minorities, and that minority women present with later-stage breast tumors and they have poorer survival, "said Dr. Lum of Loma Linda (Calif.) University. "Yet under the new guidelines, the diagnosis of patients such as these would be delayed until they developed larger tumors evident though manual breast exams. Now, through our study, we know that minority women fall into these categories in a higher percentage" than do white women.
Dr. Lum presented an analysis of the California Cancer Registry, focusing on 46,691 women aged 40-74 years who were diagnosed with DCIS or T1N0 tumors in 2004-2008. She and her colleagues divided the women into two age groups: 40-49 years (23%), for whom annual screening mammograms are no longer recommended by the U.S. Preventive Services Task Force (USPSTF), and 50-74 years (77%), for whom the annual screening recommendation has not changed.
The patients were further subdivided into four race/ethnicity groups: white (65%), Hispanic (15%), Asian/Pacific Islander (13%) and black (5%). Ethnicity was not specified for the remainder of the study group.
Overall, there were 16,067 cases of DCIS and 30,624 T1N0 tumors in the group. Compared with white women, Hispanic women were significantly more likely to have DCIS (odds ratio, 1.62) and T1N0 tumors (OR, 1.82). Women of Asian/Pacific Island descent had a 50% increased risk of DCIS and a 66% increased risk of T1N0 disease, compared with white women. Black women were significantly more likely than whites to have T1N0 cancers (OR, 1.44), but not more likely to have DCIS (OR, 0.0.91).
Age also made a difference, Dr. Lum found. Compared with older women, the younger women were significantly more likely to have hormone receptor–positive DCIS (OR, 1.85) and T1N0 disease (OR, 1.43). Younger women were also more likely to have HER2-positive T1N0 tumors (OR, 1.46), and were 67% more likely to have the difficult-to-treat triple-negative tumors.
The molecular findings argue strongly in favor of regular screening mammograms for younger women – especially minority women, Dr. Lum said. Hormone receptor–positive tumors and HER2-positive tumors have highly effective, targeted therapies. And although triple-negative tumors are a therapeutic challenge, the best curative chance lies with early treatment, she said.
"By excluding these younger women from mammographic screening, you may be relatively diminishing the benefits of these targeted therapies," she said. "And while younger women do have lower cancer rates than older women, under these USPSTF guidelines, we could miss tremendous opportunities to improve outcomes in specific racial and disease groups."
Dr. Lum had no financial declarations with regard to her study.
breast cancer, Hispanic, Asian, black, ductal cancer in situ, DCIS, breast tumors, Dr. Sharon Lum, American Society of Breast Surgeons,
WASHINGTON – The revised national screening mammography guidelines may especially impact the health of younger minority women for whom annual screening is no longer recommended, investigators suggested.
A retrospective study derived from a large state cancer registry found that Hispanic, Asian, and black women aged 40-49 years were up to 60% more likely to be diagnosed with ductal cancer in situ (DCIS) and up to 80% more like to have small invasive breast tumors (T1N0) than were their white counterparts.
These women were significantly more likely to have tumors that respond best to very early therapy, Dr. Sharon Lum said at the annual meeting of the American Society of Breast Surgeons. But if their cancers are not detected through mammography, women in these groups might not receive such therapy.
"We already know that breast cancer occurs at a younger age in minorities, and that minority women present with later-stage breast tumors and they have poorer survival, "said Dr. Lum of Loma Linda (Calif.) University. "Yet under the new guidelines, the diagnosis of patients such as these would be delayed until they developed larger tumors evident though manual breast exams. Now, through our study, we know that minority women fall into these categories in a higher percentage" than do white women.
Dr. Lum presented an analysis of the California Cancer Registry, focusing on 46,691 women aged 40-74 years who were diagnosed with DCIS or T1N0 tumors in 2004-2008. She and her colleagues divided the women into two age groups: 40-49 years (23%), for whom annual screening mammograms are no longer recommended by the U.S. Preventive Services Task Force (USPSTF), and 50-74 years (77%), for whom the annual screening recommendation has not changed.
The patients were further subdivided into four race/ethnicity groups: white (65%), Hispanic (15%), Asian/Pacific Islander (13%) and black (5%). Ethnicity was not specified for the remainder of the study group.
Overall, there were 16,067 cases of DCIS and 30,624 T1N0 tumors in the group. Compared with white women, Hispanic women were significantly more likely to have DCIS (odds ratio, 1.62) and T1N0 tumors (OR, 1.82). Women of Asian/Pacific Island descent had a 50% increased risk of DCIS and a 66% increased risk of T1N0 disease, compared with white women. Black women were significantly more likely than whites to have T1N0 cancers (OR, 1.44), but not more likely to have DCIS (OR, 0.0.91).
Age also made a difference, Dr. Lum found. Compared with older women, the younger women were significantly more likely to have hormone receptor–positive DCIS (OR, 1.85) and T1N0 disease (OR, 1.43). Younger women were also more likely to have HER2-positive T1N0 tumors (OR, 1.46), and were 67% more likely to have the difficult-to-treat triple-negative tumors.
The molecular findings argue strongly in favor of regular screening mammograms for younger women – especially minority women, Dr. Lum said. Hormone receptor–positive tumors and HER2-positive tumors have highly effective, targeted therapies. And although triple-negative tumors are a therapeutic challenge, the best curative chance lies with early treatment, she said.
"By excluding these younger women from mammographic screening, you may be relatively diminishing the benefits of these targeted therapies," she said. "And while younger women do have lower cancer rates than older women, under these USPSTF guidelines, we could miss tremendous opportunities to improve outcomes in specific racial and disease groups."
Dr. Lum had no financial declarations with regard to her study.
WASHINGTON – The revised national screening mammography guidelines may especially impact the health of younger minority women for whom annual screening is no longer recommended, investigators suggested.
A retrospective study derived from a large state cancer registry found that Hispanic, Asian, and black women aged 40-49 years were up to 60% more likely to be diagnosed with ductal cancer in situ (DCIS) and up to 80% more like to have small invasive breast tumors (T1N0) than were their white counterparts.
These women were significantly more likely to have tumors that respond best to very early therapy, Dr. Sharon Lum said at the annual meeting of the American Society of Breast Surgeons. But if their cancers are not detected through mammography, women in these groups might not receive such therapy.
"We already know that breast cancer occurs at a younger age in minorities, and that minority women present with later-stage breast tumors and they have poorer survival, "said Dr. Lum of Loma Linda (Calif.) University. "Yet under the new guidelines, the diagnosis of patients such as these would be delayed until they developed larger tumors evident though manual breast exams. Now, through our study, we know that minority women fall into these categories in a higher percentage" than do white women.
Dr. Lum presented an analysis of the California Cancer Registry, focusing on 46,691 women aged 40-74 years who were diagnosed with DCIS or T1N0 tumors in 2004-2008. She and her colleagues divided the women into two age groups: 40-49 years (23%), for whom annual screening mammograms are no longer recommended by the U.S. Preventive Services Task Force (USPSTF), and 50-74 years (77%), for whom the annual screening recommendation has not changed.
The patients were further subdivided into four race/ethnicity groups: white (65%), Hispanic (15%), Asian/Pacific Islander (13%) and black (5%). Ethnicity was not specified for the remainder of the study group.
Overall, there were 16,067 cases of DCIS and 30,624 T1N0 tumors in the group. Compared with white women, Hispanic women were significantly more likely to have DCIS (odds ratio, 1.62) and T1N0 tumors (OR, 1.82). Women of Asian/Pacific Island descent had a 50% increased risk of DCIS and a 66% increased risk of T1N0 disease, compared with white women. Black women were significantly more likely than whites to have T1N0 cancers (OR, 1.44), but not more likely to have DCIS (OR, 0.0.91).
Age also made a difference, Dr. Lum found. Compared with older women, the younger women were significantly more likely to have hormone receptor–positive DCIS (OR, 1.85) and T1N0 disease (OR, 1.43). Younger women were also more likely to have HER2-positive T1N0 tumors (OR, 1.46), and were 67% more likely to have the difficult-to-treat triple-negative tumors.
The molecular findings argue strongly in favor of regular screening mammograms for younger women – especially minority women, Dr. Lum said. Hormone receptor–positive tumors and HER2-positive tumors have highly effective, targeted therapies. And although triple-negative tumors are a therapeutic challenge, the best curative chance lies with early treatment, she said.
"By excluding these younger women from mammographic screening, you may be relatively diminishing the benefits of these targeted therapies," she said. "And while younger women do have lower cancer rates than older women, under these USPSTF guidelines, we could miss tremendous opportunities to improve outcomes in specific racial and disease groups."
Dr. Lum had no financial declarations with regard to her study.
breast cancer, Hispanic, Asian, black, ductal cancer in situ, DCIS, breast tumors, Dr. Sharon Lum, American Society of Breast Surgeons,
breast cancer, Hispanic, Asian, black, ductal cancer in situ, DCIS, breast tumors, Dr. Sharon Lum, American Society of Breast Surgeons,
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF BREAST SURGEONS
Major Finding: Compared with white women, Hispanic women were significantly more likely to have DCIS (OR, 1.62) and T1N0 tumors (OR, 1.82).
Data Source: A retrospective review of almost 47,000 cancers among women aged 40-74 included in a California cancer database.
Disclosures: Dr. Lum declared no financial conflicts.
Change in Mammography Guidelines May Adversely Affect Young Minority Women
WASHINGTON – The revised national screening mammography guidelines may especially impact the health of younger minority women for whom annual screening is no longer recommended, investigators suggested.
A retrospective study derived from a large state cancer registry found that Hispanic, Asian, and black women aged 40-49 years were up to 60% more likely to be diagnosed with ductal cancer in situ (DCIS) and up to 80% more like to have small invasive breast tumors (T1N0) than were their white counterparts.
These women were significantly more likely to have tumors that respond best to very early therapy, Dr. Sharon Lum said at the annual meeting of the American Society of Breast Surgeons. But if their cancers are not detected through mammography, women in these groups might not receive such therapy.
"We already know that breast cancer occurs at a younger age in minorities, and that minority women present with later-stage breast tumors and they have poorer survival, "said Dr. Lum of Loma Linda (Calif.) University. "Yet under the new guidelines, the diagnosis of patients such as these would be delayed until they developed larger tumors evident though manual breast exams. Now, through our study, we know that minority women fall into these categories in a higher percentage" than do white women.
Dr. Lum presented an analysis of the California Cancer Registry, focusing on 46,691 women aged 40-74 years who were diagnosed with DCIS or T1N0 tumors in 2004-2008. She and her colleagues divided the women into two age groups: 40-49 years (23%), for whom annual screening mammograms are no longer recommended by the U.S. Preventive Services Task Force (USPSTF), and 50-74 years (77%), for whom the annual screening recommendation has not changed.
The patients were further subdivided into four race/ethnicity groups: white (65%), Hispanic (15%), Asian/Pacific Islander (13%) and black (5%). Ethnicity was not specified for the remainder of the study group.
Overall, there were 16,067 cases of DCIS and 30,624 T1N0 tumors in the group. Compared with white women, Hispanic women were significantly more likely to have DCIS (odds ratio, 1.62) and T1N0 tumors (OR, 1.82). Women of Asian/Pacific Island descent had a 50% increased risk of DCIS and a 66% increased risk of T1N0 disease, compared with white women. Black women were significantly more likely than whites to have T1N0 cancers (OR, 1.44), but not more likely to have DCIS (OR, 0.0.91).
Age also made a difference, Dr. Lum found. Compared with older women, the younger women were significantly more likely to have hormone receptor–positive DCIS (OR, 1.85) and T1N0 disease (OR, 1.43). Younger women were also more likely to have HER2-positive T1N0 tumors (OR, 1.46), and were 67% more likely to have the difficult-to-treat triple-negative tumors.
The molecular findings argue strongly in favor of regular screening mammograms for younger women – especially minority women, Dr. Lum said. Hormone receptor–positive tumors and HER2-positive tumors have highly effective, targeted therapies. And although triple-negative tumors are a therapeutic challenge, the best curative chance lies with early treatment, she said.
"By excluding these younger women from mammographic screening, you may be relatively diminishing the benefits of these targeted therapies," she said. "And while younger women do have lower cancer rates than older women, under these USPSTF guidelines, we could miss tremendous opportunities to improve outcomes in specific racial and disease groups."
Dr. Lum had no financial declarations with regard to her study.
breast cancer, Hispanic, Asian, black, ductal cancer in situ, DCIS, breast tumors, Dr. Sharon Lum, American Society of Breast Surgeons,
WASHINGTON – The revised national screening mammography guidelines may especially impact the health of younger minority women for whom annual screening is no longer recommended, investigators suggested.
A retrospective study derived from a large state cancer registry found that Hispanic, Asian, and black women aged 40-49 years were up to 60% more likely to be diagnosed with ductal cancer in situ (DCIS) and up to 80% more like to have small invasive breast tumors (T1N0) than were their white counterparts.
These women were significantly more likely to have tumors that respond best to very early therapy, Dr. Sharon Lum said at the annual meeting of the American Society of Breast Surgeons. But if their cancers are not detected through mammography, women in these groups might not receive such therapy.
"We already know that breast cancer occurs at a younger age in minorities, and that minority women present with later-stage breast tumors and they have poorer survival, "said Dr. Lum of Loma Linda (Calif.) University. "Yet under the new guidelines, the diagnosis of patients such as these would be delayed until they developed larger tumors evident though manual breast exams. Now, through our study, we know that minority women fall into these categories in a higher percentage" than do white women.
Dr. Lum presented an analysis of the California Cancer Registry, focusing on 46,691 women aged 40-74 years who were diagnosed with DCIS or T1N0 tumors in 2004-2008. She and her colleagues divided the women into two age groups: 40-49 years (23%), for whom annual screening mammograms are no longer recommended by the U.S. Preventive Services Task Force (USPSTF), and 50-74 years (77%), for whom the annual screening recommendation has not changed.
The patients were further subdivided into four race/ethnicity groups: white (65%), Hispanic (15%), Asian/Pacific Islander (13%) and black (5%). Ethnicity was not specified for the remainder of the study group.
Overall, there were 16,067 cases of DCIS and 30,624 T1N0 tumors in the group. Compared with white women, Hispanic women were significantly more likely to have DCIS (odds ratio, 1.62) and T1N0 tumors (OR, 1.82). Women of Asian/Pacific Island descent had a 50% increased risk of DCIS and a 66% increased risk of T1N0 disease, compared with white women. Black women were significantly more likely than whites to have T1N0 cancers (OR, 1.44), but not more likely to have DCIS (OR, 0.0.91).
Age also made a difference, Dr. Lum found. Compared with older women, the younger women were significantly more likely to have hormone receptor–positive DCIS (OR, 1.85) and T1N0 disease (OR, 1.43). Younger women were also more likely to have HER2-positive T1N0 tumors (OR, 1.46), and were 67% more likely to have the difficult-to-treat triple-negative tumors.
The molecular findings argue strongly in favor of regular screening mammograms for younger women – especially minority women, Dr. Lum said. Hormone receptor–positive tumors and HER2-positive tumors have highly effective, targeted therapies. And although triple-negative tumors are a therapeutic challenge, the best curative chance lies with early treatment, she said.
"By excluding these younger women from mammographic screening, you may be relatively diminishing the benefits of these targeted therapies," she said. "And while younger women do have lower cancer rates than older women, under these USPSTF guidelines, we could miss tremendous opportunities to improve outcomes in specific racial and disease groups."
Dr. Lum had no financial declarations with regard to her study.
WASHINGTON – The revised national screening mammography guidelines may especially impact the health of younger minority women for whom annual screening is no longer recommended, investigators suggested.
A retrospective study derived from a large state cancer registry found that Hispanic, Asian, and black women aged 40-49 years were up to 60% more likely to be diagnosed with ductal cancer in situ (DCIS) and up to 80% more like to have small invasive breast tumors (T1N0) than were their white counterparts.
These women were significantly more likely to have tumors that respond best to very early therapy, Dr. Sharon Lum said at the annual meeting of the American Society of Breast Surgeons. But if their cancers are not detected through mammography, women in these groups might not receive such therapy.
"We already know that breast cancer occurs at a younger age in minorities, and that minority women present with later-stage breast tumors and they have poorer survival, "said Dr. Lum of Loma Linda (Calif.) University. "Yet under the new guidelines, the diagnosis of patients such as these would be delayed until they developed larger tumors evident though manual breast exams. Now, through our study, we know that minority women fall into these categories in a higher percentage" than do white women.
Dr. Lum presented an analysis of the California Cancer Registry, focusing on 46,691 women aged 40-74 years who were diagnosed with DCIS or T1N0 tumors in 2004-2008. She and her colleagues divided the women into two age groups: 40-49 years (23%), for whom annual screening mammograms are no longer recommended by the U.S. Preventive Services Task Force (USPSTF), and 50-74 years (77%), for whom the annual screening recommendation has not changed.
The patients were further subdivided into four race/ethnicity groups: white (65%), Hispanic (15%), Asian/Pacific Islander (13%) and black (5%). Ethnicity was not specified for the remainder of the study group.
Overall, there were 16,067 cases of DCIS and 30,624 T1N0 tumors in the group. Compared with white women, Hispanic women were significantly more likely to have DCIS (odds ratio, 1.62) and T1N0 tumors (OR, 1.82). Women of Asian/Pacific Island descent had a 50% increased risk of DCIS and a 66% increased risk of T1N0 disease, compared with white women. Black women were significantly more likely than whites to have T1N0 cancers (OR, 1.44), but not more likely to have DCIS (OR, 0.0.91).
Age also made a difference, Dr. Lum found. Compared with older women, the younger women were significantly more likely to have hormone receptor–positive DCIS (OR, 1.85) and T1N0 disease (OR, 1.43). Younger women were also more likely to have HER2-positive T1N0 tumors (OR, 1.46), and were 67% more likely to have the difficult-to-treat triple-negative tumors.
The molecular findings argue strongly in favor of regular screening mammograms for younger women – especially minority women, Dr. Lum said. Hormone receptor–positive tumors and HER2-positive tumors have highly effective, targeted therapies. And although triple-negative tumors are a therapeutic challenge, the best curative chance lies with early treatment, she said.
"By excluding these younger women from mammographic screening, you may be relatively diminishing the benefits of these targeted therapies," she said. "And while younger women do have lower cancer rates than older women, under these USPSTF guidelines, we could miss tremendous opportunities to improve outcomes in specific racial and disease groups."
Dr. Lum had no financial declarations with regard to her study.
breast cancer, Hispanic, Asian, black, ductal cancer in situ, DCIS, breast tumors, Dr. Sharon Lum, American Society of Breast Surgeons,
breast cancer, Hispanic, Asian, black, ductal cancer in situ, DCIS, breast tumors, Dr. Sharon Lum, American Society of Breast Surgeons,
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF BREAST SURGEONS
Major Finding: Compared with white women, Hispanic women were significantly more likely to have DCIS (OR, 1.62) and T1N0 tumors (OR, 1.82).
Data Source: A retrospective review of almost 47,000 cancers among women aged 40-74 included in a California cancer database.
Disclosures: Dr. Lum declared no financial conflicts.
Allopurinol Still Deemed First Line for Gout
SAN DIEGO – Allopurinol should remain the first-line agent for gout prophylaxis, despite some competition from a newer drug, febuxostat, according to Dr. John Pendleton.
Allopurinol is generally very effective, can be safely titrated for those with renal impairment, and is the most cost-effective option, he said at the annual meeting of the American College of Physicians.
"I still recommend allopurinol as the initial treatment, because you don’t need a 24-hour urine collection to give it; it’s effective in both overproducers and underexcretors [of uric acid]; it can be taken just once a day; and it’s safe and effective for those with mild renal insufficiency when the dose is adjusted," said Dr. Pendleton, an internist in Roanoke, Va.
Price is also an important factor, he noted. "Generic allopurinol costs about $15 per month. The brand name costs about $43 per month. But the price for febuxostat comes in at about $156 per month."
With either drug, the treatment goal should be to lower uric acid levels to be 6 mg/dL, said Dr. Pendleton, referring to a retrospective study of 276 patients with recurrent gout attacks. "This study noted that among the 81 patients with a uric acid of less than 6 mg/dL, 88% had no recurrent attacks during the 3-year observational period" (Arthritis Rheum. 2004;51:321-5).
Allopurinol has been the "standby drug" for gouty arthritis for 60 years, and still performs admirably, Dr. Pendleton said. Although most initial doses range from 50 to 300 mg/day, "some recent studies suggest that only 25% of patients will reach the uric acid target on that regimen. For many patients, we need to increase the dose to get that level down."
The dose should be incrementally increased every 3-4 weeks to reach the uric acid target level; doses of up to 800 mg/day are approved for this indication. "But if you’re not able to achieve this desired level by pushing the dose close to 800 mg, I would consider trying febuxostat."
Febuxostat, a xanthine oxidase inhibitor, is more selective and potent than allopurinol. "It’s metabolized in the liver and very little of the active drug is excreted renally, raising the possibility that it might be safer in patients with mild to moderate renal insufficiency," Dr. Pendleton said.
It’s not easy to fully compare the two, because all three of the studies on the basis of which febuxostat was approved used a fixed-dose allopurinol regimen. "None of them allowed the total upward titration of allopurinol for a fair comparison," Dr. Pendleton pointed out.
The studies concluded that 40 mg of febuxostat was as effective as 300 mg of allopurinol. "The higher dose [of febuxostat 80 mg] seemed to be more effective than 400 mg allopurinol, but again, the studies did not allow for an upward titration" of the comparator, he said.
Although none of the patients in those trials had a creatinine level of more than 2.5 mg/dL, "a short-term study suggests that febuxostat dosing would not need to be adjusted even with a very low creatinine clearance [of 10-29 mL/min]. But just the same I would be very careful in that setting," Dr. Pendleton said (Am. J. Ther. 2005;12:22-34).
Febuxostat also appears to be safe for patients with mild hepatic dysfunction, with an adverse event profile similar to that of allopurinol. However, febuxostat is contraindicated in patients who are taking azathioprine, mercaptopurine, and theophylline.
Probenecid could be another option for some patients, especially those who underexcrete uric acid. Although probenecid has a somewhat better adverse event profile than allopurinol, it requires a 24-hour urine collection to rule out overproduction of uric acid and it seems to increase the risk of kidney stones. The drug is not as effective in patients with a low creatinine clearance (less than 60 mL/min), and "seems to have no effect at all at a creatinine level of 30 mL/min or lower," Dr. Pendleton said. Probenecid also must be taken three times a day – another drawback, in his opinion.
Dr. Pendleton said that he had no relevant financial disclosures.
SAN DIEGO – Allopurinol should remain the first-line agent for gout prophylaxis, despite some competition from a newer drug, febuxostat, according to Dr. John Pendleton.
Allopurinol is generally very effective, can be safely titrated for those with renal impairment, and is the most cost-effective option, he said at the annual meeting of the American College of Physicians.
"I still recommend allopurinol as the initial treatment, because you don’t need a 24-hour urine collection to give it; it’s effective in both overproducers and underexcretors [of uric acid]; it can be taken just once a day; and it’s safe and effective for those with mild renal insufficiency when the dose is adjusted," said Dr. Pendleton, an internist in Roanoke, Va.
Price is also an important factor, he noted. "Generic allopurinol costs about $15 per month. The brand name costs about $43 per month. But the price for febuxostat comes in at about $156 per month."
With either drug, the treatment goal should be to lower uric acid levels to be 6 mg/dL, said Dr. Pendleton, referring to a retrospective study of 276 patients with recurrent gout attacks. "This study noted that among the 81 patients with a uric acid of less than 6 mg/dL, 88% had no recurrent attacks during the 3-year observational period" (Arthritis Rheum. 2004;51:321-5).
Allopurinol has been the "standby drug" for gouty arthritis for 60 years, and still performs admirably, Dr. Pendleton said. Although most initial doses range from 50 to 300 mg/day, "some recent studies suggest that only 25% of patients will reach the uric acid target on that regimen. For many patients, we need to increase the dose to get that level down."
The dose should be incrementally increased every 3-4 weeks to reach the uric acid target level; doses of up to 800 mg/day are approved for this indication. "But if you’re not able to achieve this desired level by pushing the dose close to 800 mg, I would consider trying febuxostat."
Febuxostat, a xanthine oxidase inhibitor, is more selective and potent than allopurinol. "It’s metabolized in the liver and very little of the active drug is excreted renally, raising the possibility that it might be safer in patients with mild to moderate renal insufficiency," Dr. Pendleton said.
It’s not easy to fully compare the two, because all three of the studies on the basis of which febuxostat was approved used a fixed-dose allopurinol regimen. "None of them allowed the total upward titration of allopurinol for a fair comparison," Dr. Pendleton pointed out.
The studies concluded that 40 mg of febuxostat was as effective as 300 mg of allopurinol. "The higher dose [of febuxostat 80 mg] seemed to be more effective than 400 mg allopurinol, but again, the studies did not allow for an upward titration" of the comparator, he said.
Although none of the patients in those trials had a creatinine level of more than 2.5 mg/dL, "a short-term study suggests that febuxostat dosing would not need to be adjusted even with a very low creatinine clearance [of 10-29 mL/min]. But just the same I would be very careful in that setting," Dr. Pendleton said (Am. J. Ther. 2005;12:22-34).
Febuxostat also appears to be safe for patients with mild hepatic dysfunction, with an adverse event profile similar to that of allopurinol. However, febuxostat is contraindicated in patients who are taking azathioprine, mercaptopurine, and theophylline.
Probenecid could be another option for some patients, especially those who underexcrete uric acid. Although probenecid has a somewhat better adverse event profile than allopurinol, it requires a 24-hour urine collection to rule out overproduction of uric acid and it seems to increase the risk of kidney stones. The drug is not as effective in patients with a low creatinine clearance (less than 60 mL/min), and "seems to have no effect at all at a creatinine level of 30 mL/min or lower," Dr. Pendleton said. Probenecid also must be taken three times a day – another drawback, in his opinion.
Dr. Pendleton said that he had no relevant financial disclosures.
SAN DIEGO – Allopurinol should remain the first-line agent for gout prophylaxis, despite some competition from a newer drug, febuxostat, according to Dr. John Pendleton.
Allopurinol is generally very effective, can be safely titrated for those with renal impairment, and is the most cost-effective option, he said at the annual meeting of the American College of Physicians.
"I still recommend allopurinol as the initial treatment, because you don’t need a 24-hour urine collection to give it; it’s effective in both overproducers and underexcretors [of uric acid]; it can be taken just once a day; and it’s safe and effective for those with mild renal insufficiency when the dose is adjusted," said Dr. Pendleton, an internist in Roanoke, Va.
Price is also an important factor, he noted. "Generic allopurinol costs about $15 per month. The brand name costs about $43 per month. But the price for febuxostat comes in at about $156 per month."
With either drug, the treatment goal should be to lower uric acid levels to be 6 mg/dL, said Dr. Pendleton, referring to a retrospective study of 276 patients with recurrent gout attacks. "This study noted that among the 81 patients with a uric acid of less than 6 mg/dL, 88% had no recurrent attacks during the 3-year observational period" (Arthritis Rheum. 2004;51:321-5).
Allopurinol has been the "standby drug" for gouty arthritis for 60 years, and still performs admirably, Dr. Pendleton said. Although most initial doses range from 50 to 300 mg/day, "some recent studies suggest that only 25% of patients will reach the uric acid target on that regimen. For many patients, we need to increase the dose to get that level down."
The dose should be incrementally increased every 3-4 weeks to reach the uric acid target level; doses of up to 800 mg/day are approved for this indication. "But if you’re not able to achieve this desired level by pushing the dose close to 800 mg, I would consider trying febuxostat."
Febuxostat, a xanthine oxidase inhibitor, is more selective and potent than allopurinol. "It’s metabolized in the liver and very little of the active drug is excreted renally, raising the possibility that it might be safer in patients with mild to moderate renal insufficiency," Dr. Pendleton said.
It’s not easy to fully compare the two, because all three of the studies on the basis of which febuxostat was approved used a fixed-dose allopurinol regimen. "None of them allowed the total upward titration of allopurinol for a fair comparison," Dr. Pendleton pointed out.
The studies concluded that 40 mg of febuxostat was as effective as 300 mg of allopurinol. "The higher dose [of febuxostat 80 mg] seemed to be more effective than 400 mg allopurinol, but again, the studies did not allow for an upward titration" of the comparator, he said.
Although none of the patients in those trials had a creatinine level of more than 2.5 mg/dL, "a short-term study suggests that febuxostat dosing would not need to be adjusted even with a very low creatinine clearance [of 10-29 mL/min]. But just the same I would be very careful in that setting," Dr. Pendleton said (Am. J. Ther. 2005;12:22-34).
Febuxostat also appears to be safe for patients with mild hepatic dysfunction, with an adverse event profile similar to that of allopurinol. However, febuxostat is contraindicated in patients who are taking azathioprine, mercaptopurine, and theophylline.
Probenecid could be another option for some patients, especially those who underexcrete uric acid. Although probenecid has a somewhat better adverse event profile than allopurinol, it requires a 24-hour urine collection to rule out overproduction of uric acid and it seems to increase the risk of kidney stones. The drug is not as effective in patients with a low creatinine clearance (less than 60 mL/min), and "seems to have no effect at all at a creatinine level of 30 mL/min or lower," Dr. Pendleton said. Probenecid also must be taken three times a day – another drawback, in his opinion.
Dr. Pendleton said that he had no relevant financial disclosures.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF PHYSICIANS
Allopurinol Still Deemed First Line for Gout
SAN DIEGO – Allopurinol should remain the first-line agent for gout prophylaxis, despite some competition from a newer drug, febuxostat, according to Dr. John Pendleton.
Allopurinol is generally very effective, can be safely titrated for those with renal impairment, and is the most cost-effective option, he said at the annual meeting of the American College of Physicians.
"I still recommend allopurinol as the initial treatment, because you don’t need a 24-hour urine collection to give it; it’s effective in both overproducers and underexcretors [of uric acid]; it can be taken just once a day; and it’s safe and effective for those with mild renal insufficiency when the dose is adjusted," said Dr. Pendleton, an internist in Roanoke, Va.
Price is also an important factor, he noted. "Generic allopurinol costs about $15 per month. The brand name costs about $43 per month. But the price for febuxostat comes in at about $156 per month."
With either drug, the treatment goal should be to lower uric acid levels to be 6 mg/dL, said Dr. Pendleton, referring to a retrospective study of 276 patients with recurrent gout attacks. "This study noted that among the 81 patients with a uric acid of less than 6 mg/dL, 88% had no recurrent attacks during the 3-year observational period" (Arthritis Rheum. 2004;51:321-5).
Allopurinol has been the "standby drug" for gouty arthritis for 60 years, and still performs admirably, Dr. Pendleton said. Although most initial doses range from 50 to 300 mg/day, "some recent studies suggest that only 25% of patients will reach the uric acid target on that regimen. For many patients, we need to increase the dose to get that level down."
The dose should be incrementally increased every 3-4 weeks to reach the uric acid target level; doses of up to 800 mg/day are approved for this indication. "But if you’re not able to achieve this desired level by pushing the dose close to 800 mg, I would consider trying febuxostat."
Febuxostat, a xanthine oxidase inhibitor, is more selective and potent than allopurinol. "It’s metabolized in the liver and very little of the active drug is excreted renally, raising the possibility that it might be safer in patients with mild to moderate renal insufficiency," Dr. Pendleton said.
It’s not easy to fully compare the two, because all three of the studies on the basis of which febuxostat was approved used a fixed-dose allopurinol regimen. "None of them allowed the total upward titration of allopurinol for a fair comparison," Dr. Pendleton pointed out.
The studies concluded that 40 mg of febuxostat was as effective as 300 mg of allopurinol. "The higher dose [of febuxostat 80 mg] seemed to be more effective than 400 mg allopurinol, but again, the studies did not allow for an upward titration" of the comparator, he said.
Although none of the patients in those trials had a creatinine level of more than 2.5 mg/dL, "a short-term study suggests that febuxostat dosing would not need to be adjusted even with a very low creatinine clearance [of 10-29 mL/min]. But just the same I would be very careful in that setting," Dr. Pendleton said (Am. J. Ther. 2005;12:22-34).
Febuxostat also appears to be safe for patients with mild hepatic dysfunction, with an adverse event profile similar to that of allopurinol. However, febuxostat is contraindicated in patients who are taking azathioprine, mercaptopurine, and theophylline.
Probenecid could be another option for some patients, especially those who underexcrete uric acid. Although probenecid has a somewhat better adverse event profile than allopurinol, it requires a 24-hour urine collection to rule out overproduction of uric acid and it seems to increase the risk of kidney stones. The drug is not as effective in patients with a low creatinine clearance (less than 60 mL/min), and "seems to have no effect at all at a creatinine level of 30 mL/min or lower," Dr. Pendleton said. Probenecid also must be taken three times a day – another drawback, in his opinion.
Dr. Pendleton said that he had no relevant financial disclosures.
SAN DIEGO – Allopurinol should remain the first-line agent for gout prophylaxis, despite some competition from a newer drug, febuxostat, according to Dr. John Pendleton.
Allopurinol is generally very effective, can be safely titrated for those with renal impairment, and is the most cost-effective option, he said at the annual meeting of the American College of Physicians.
"I still recommend allopurinol as the initial treatment, because you don’t need a 24-hour urine collection to give it; it’s effective in both overproducers and underexcretors [of uric acid]; it can be taken just once a day; and it’s safe and effective for those with mild renal insufficiency when the dose is adjusted," said Dr. Pendleton, an internist in Roanoke, Va.
Price is also an important factor, he noted. "Generic allopurinol costs about $15 per month. The brand name costs about $43 per month. But the price for febuxostat comes in at about $156 per month."
With either drug, the treatment goal should be to lower uric acid levels to be 6 mg/dL, said Dr. Pendleton, referring to a retrospective study of 276 patients with recurrent gout attacks. "This study noted that among the 81 patients with a uric acid of less than 6 mg/dL, 88% had no recurrent attacks during the 3-year observational period" (Arthritis Rheum. 2004;51:321-5).
Allopurinol has been the "standby drug" for gouty arthritis for 60 years, and still performs admirably, Dr. Pendleton said. Although most initial doses range from 50 to 300 mg/day, "some recent studies suggest that only 25% of patients will reach the uric acid target on that regimen. For many patients, we need to increase the dose to get that level down."
The dose should be incrementally increased every 3-4 weeks to reach the uric acid target level; doses of up to 800 mg/day are approved for this indication. "But if you’re not able to achieve this desired level by pushing the dose close to 800 mg, I would consider trying febuxostat."
Febuxostat, a xanthine oxidase inhibitor, is more selective and potent than allopurinol. "It’s metabolized in the liver and very little of the active drug is excreted renally, raising the possibility that it might be safer in patients with mild to moderate renal insufficiency," Dr. Pendleton said.
It’s not easy to fully compare the two, because all three of the studies on the basis of which febuxostat was approved used a fixed-dose allopurinol regimen. "None of them allowed the total upward titration of allopurinol for a fair comparison," Dr. Pendleton pointed out.
The studies concluded that 40 mg of febuxostat was as effective as 300 mg of allopurinol. "The higher dose [of febuxostat 80 mg] seemed to be more effective than 400 mg allopurinol, but again, the studies did not allow for an upward titration" of the comparator, he said.
Although none of the patients in those trials had a creatinine level of more than 2.5 mg/dL, "a short-term study suggests that febuxostat dosing would not need to be adjusted even with a very low creatinine clearance [of 10-29 mL/min]. But just the same I would be very careful in that setting," Dr. Pendleton said (Am. J. Ther. 2005;12:22-34).
Febuxostat also appears to be safe for patients with mild hepatic dysfunction, with an adverse event profile similar to that of allopurinol. However, febuxostat is contraindicated in patients who are taking azathioprine, mercaptopurine, and theophylline.
Probenecid could be another option for some patients, especially those who underexcrete uric acid. Although probenecid has a somewhat better adverse event profile than allopurinol, it requires a 24-hour urine collection to rule out overproduction of uric acid and it seems to increase the risk of kidney stones. The drug is not as effective in patients with a low creatinine clearance (less than 60 mL/min), and "seems to have no effect at all at a creatinine level of 30 mL/min or lower," Dr. Pendleton said. Probenecid also must be taken three times a day – another drawback, in his opinion.
Dr. Pendleton said that he had no relevant financial disclosures.
SAN DIEGO – Allopurinol should remain the first-line agent for gout prophylaxis, despite some competition from a newer drug, febuxostat, according to Dr. John Pendleton.
Allopurinol is generally very effective, can be safely titrated for those with renal impairment, and is the most cost-effective option, he said at the annual meeting of the American College of Physicians.
"I still recommend allopurinol as the initial treatment, because you don’t need a 24-hour urine collection to give it; it’s effective in both overproducers and underexcretors [of uric acid]; it can be taken just once a day; and it’s safe and effective for those with mild renal insufficiency when the dose is adjusted," said Dr. Pendleton, an internist in Roanoke, Va.
Price is also an important factor, he noted. "Generic allopurinol costs about $15 per month. The brand name costs about $43 per month. But the price for febuxostat comes in at about $156 per month."
With either drug, the treatment goal should be to lower uric acid levels to be 6 mg/dL, said Dr. Pendleton, referring to a retrospective study of 276 patients with recurrent gout attacks. "This study noted that among the 81 patients with a uric acid of less than 6 mg/dL, 88% had no recurrent attacks during the 3-year observational period" (Arthritis Rheum. 2004;51:321-5).
Allopurinol has been the "standby drug" for gouty arthritis for 60 years, and still performs admirably, Dr. Pendleton said. Although most initial doses range from 50 to 300 mg/day, "some recent studies suggest that only 25% of patients will reach the uric acid target on that regimen. For many patients, we need to increase the dose to get that level down."
The dose should be incrementally increased every 3-4 weeks to reach the uric acid target level; doses of up to 800 mg/day are approved for this indication. "But if you’re not able to achieve this desired level by pushing the dose close to 800 mg, I would consider trying febuxostat."
Febuxostat, a xanthine oxidase inhibitor, is more selective and potent than allopurinol. "It’s metabolized in the liver and very little of the active drug is excreted renally, raising the possibility that it might be safer in patients with mild to moderate renal insufficiency," Dr. Pendleton said.
It’s not easy to fully compare the two, because all three of the studies on the basis of which febuxostat was approved used a fixed-dose allopurinol regimen. "None of them allowed the total upward titration of allopurinol for a fair comparison," Dr. Pendleton pointed out.
The studies concluded that 40 mg of febuxostat was as effective as 300 mg of allopurinol. "The higher dose [of febuxostat 80 mg] seemed to be more effective than 400 mg allopurinol, but again, the studies did not allow for an upward titration" of the comparator, he said.
Although none of the patients in those trials had a creatinine level of more than 2.5 mg/dL, "a short-term study suggests that febuxostat dosing would not need to be adjusted even with a very low creatinine clearance [of 10-29 mL/min]. But just the same I would be very careful in that setting," Dr. Pendleton said (Am. J. Ther. 2005;12:22-34).
Febuxostat also appears to be safe for patients with mild hepatic dysfunction, with an adverse event profile similar to that of allopurinol. However, febuxostat is contraindicated in patients who are taking azathioprine, mercaptopurine, and theophylline.
Probenecid could be another option for some patients, especially those who underexcrete uric acid. Although probenecid has a somewhat better adverse event profile than allopurinol, it requires a 24-hour urine collection to rule out overproduction of uric acid and it seems to increase the risk of kidney stones. The drug is not as effective in patients with a low creatinine clearance (less than 60 mL/min), and "seems to have no effect at all at a creatinine level of 30 mL/min or lower," Dr. Pendleton said. Probenecid also must be taken three times a day – another drawback, in his opinion.
Dr. Pendleton said that he had no relevant financial disclosures.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF PHYSICIANS
Treating Mood Disorders: No Easy Decisions
MADRID – There are no easy choices for women who present with mood disorders during pregnancy – or for the physicians who treat them.
The decision to treat prenatal depression or anxiety is not to be taken lightly, but often must be considered, despite a relative dearth of data supporting or refuting drug safety during pregnancy, Dr. Shari I. Lusskin said at the conference.
“There are data, but not great data,” said Dr. Lusskin, director of reproductive psychiatry at the NYU Langone Medical Center, New York. “We will never have 10,000 patients taking medication compared to 10,000 not taking it, compared to a control group that is not depressed or anxious, nor will we have years of follow-up data on their children.”
The data that do exist are mostly comprised of case series, which are small and lack controls. “When you see a negative study [about the effect of treating mental disorders in a pregnant woman], keep in mind that there is often a lack of information about the mother's diagnosis. Reports of smoking and substance abuse are notoriously unreliable,” and can't be accounted for in the results, she said. “We also know nothing about the mother's body mass index, which is associated with many fetal complications that have nothing to do with drug exposure. And different studies use different comparison scales – you can't compare apples and oranges.”
The biggest exception is sodium valproate, an anticonvulsant also used to treat mood disorders, Dr. Lusskin said. The ongoing Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study has found in utero exposure to be associated with an increase in birth defects, and with cognitive problems in 3-year-olds whose mothers took it during pregnancy.
The uncertainty of treatment leaves pregnant women and their physicians to weigh the risks of psychotropic medications against the risks of untreated mood disorders. “There is no such thing as nonexposure,” Dr. Lusskin said. If the mother is not treated, “the fetus is going to be exposed to the mother's illness, which can create phenotypic changes with lifelong effects. If the mother is treated inadequately, the fetus will be exposed to both the effects of the illness and the medication. And if the mother is treated to remission, with the medication titrated to her response, medication exposure can be at least limited,” while the new mother becomes healthy enough to give her baby the best possible care.
The biggest barrier to treating mental illness during pregnancy is fear of fetal harm: teratogenicity, neonatal complications from drug exposure, and long-term neurodevelopmental effects. But these risks must be considered in light of real world experience – not just based on numbers from a study, Dr. Lusskin said. “Any risk of impairment has to be compared to the background risk of a birth defect, which is is 2%–4%. If a drug increases this risk in a clinically meaningful way, it has to be over and above this background rate.”
Even a substantial increase in a rare birth defect can be misleading. For instance, Ebstein's anomaly is a heart defect that occurs in 1 in 20,000 births. “Lithium is said to increase this rate to about 1 in 2,000 births. But this is still way below the expected background rate of birth defects. Risks are relevant in terms of their relativity – the absolute risk is just quoting a figure,” she said.
But untreated mental illness poses its own risk. Babies exposed in utero to the chronic stress hormones associated with anxiety and depressive disorders might become phenotypically programmed, prone themselves to early childhood behavioral problems and, in later life, to mental illness. “The fetal programming hypothesis is the interaction between the baby's genetics and the environmental exposure in utero, and it's mediated by the sex of the fetus and the timing of exposure,” Dr. Lusskin said.
Dr. Lusskin said she consults for the nonprofit drug safety database www.reprotox.org
MADRID – There are no easy choices for women who present with mood disorders during pregnancy – or for the physicians who treat them.
The decision to treat prenatal depression or anxiety is not to be taken lightly, but often must be considered, despite a relative dearth of data supporting or refuting drug safety during pregnancy, Dr. Shari I. Lusskin said at the conference.
“There are data, but not great data,” said Dr. Lusskin, director of reproductive psychiatry at the NYU Langone Medical Center, New York. “We will never have 10,000 patients taking medication compared to 10,000 not taking it, compared to a control group that is not depressed or anxious, nor will we have years of follow-up data on their children.”
The data that do exist are mostly comprised of case series, which are small and lack controls. “When you see a negative study [about the effect of treating mental disorders in a pregnant woman], keep in mind that there is often a lack of information about the mother's diagnosis. Reports of smoking and substance abuse are notoriously unreliable,” and can't be accounted for in the results, she said. “We also know nothing about the mother's body mass index, which is associated with many fetal complications that have nothing to do with drug exposure. And different studies use different comparison scales – you can't compare apples and oranges.”
The biggest exception is sodium valproate, an anticonvulsant also used to treat mood disorders, Dr. Lusskin said. The ongoing Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study has found in utero exposure to be associated with an increase in birth defects, and with cognitive problems in 3-year-olds whose mothers took it during pregnancy.
The uncertainty of treatment leaves pregnant women and their physicians to weigh the risks of psychotropic medications against the risks of untreated mood disorders. “There is no such thing as nonexposure,” Dr. Lusskin said. If the mother is not treated, “the fetus is going to be exposed to the mother's illness, which can create phenotypic changes with lifelong effects. If the mother is treated inadequately, the fetus will be exposed to both the effects of the illness and the medication. And if the mother is treated to remission, with the medication titrated to her response, medication exposure can be at least limited,” while the new mother becomes healthy enough to give her baby the best possible care.
The biggest barrier to treating mental illness during pregnancy is fear of fetal harm: teratogenicity, neonatal complications from drug exposure, and long-term neurodevelopmental effects. But these risks must be considered in light of real world experience – not just based on numbers from a study, Dr. Lusskin said. “Any risk of impairment has to be compared to the background risk of a birth defect, which is is 2%–4%. If a drug increases this risk in a clinically meaningful way, it has to be over and above this background rate.”
Even a substantial increase in a rare birth defect can be misleading. For instance, Ebstein's anomaly is a heart defect that occurs in 1 in 20,000 births. “Lithium is said to increase this rate to about 1 in 2,000 births. But this is still way below the expected background rate of birth defects. Risks are relevant in terms of their relativity – the absolute risk is just quoting a figure,” she said.
But untreated mental illness poses its own risk. Babies exposed in utero to the chronic stress hormones associated with anxiety and depressive disorders might become phenotypically programmed, prone themselves to early childhood behavioral problems and, in later life, to mental illness. “The fetal programming hypothesis is the interaction between the baby's genetics and the environmental exposure in utero, and it's mediated by the sex of the fetus and the timing of exposure,” Dr. Lusskin said.
Dr. Lusskin said she consults for the nonprofit drug safety database www.reprotox.org
MADRID – There are no easy choices for women who present with mood disorders during pregnancy – or for the physicians who treat them.
The decision to treat prenatal depression or anxiety is not to be taken lightly, but often must be considered, despite a relative dearth of data supporting or refuting drug safety during pregnancy, Dr. Shari I. Lusskin said at the conference.
“There are data, but not great data,” said Dr. Lusskin, director of reproductive psychiatry at the NYU Langone Medical Center, New York. “We will never have 10,000 patients taking medication compared to 10,000 not taking it, compared to a control group that is not depressed or anxious, nor will we have years of follow-up data on their children.”
The data that do exist are mostly comprised of case series, which are small and lack controls. “When you see a negative study [about the effect of treating mental disorders in a pregnant woman], keep in mind that there is often a lack of information about the mother's diagnosis. Reports of smoking and substance abuse are notoriously unreliable,” and can't be accounted for in the results, she said. “We also know nothing about the mother's body mass index, which is associated with many fetal complications that have nothing to do with drug exposure. And different studies use different comparison scales – you can't compare apples and oranges.”
The biggest exception is sodium valproate, an anticonvulsant also used to treat mood disorders, Dr. Lusskin said. The ongoing Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study has found in utero exposure to be associated with an increase in birth defects, and with cognitive problems in 3-year-olds whose mothers took it during pregnancy.
The uncertainty of treatment leaves pregnant women and their physicians to weigh the risks of psychotropic medications against the risks of untreated mood disorders. “There is no such thing as nonexposure,” Dr. Lusskin said. If the mother is not treated, “the fetus is going to be exposed to the mother's illness, which can create phenotypic changes with lifelong effects. If the mother is treated inadequately, the fetus will be exposed to both the effects of the illness and the medication. And if the mother is treated to remission, with the medication titrated to her response, medication exposure can be at least limited,” while the new mother becomes healthy enough to give her baby the best possible care.
The biggest barrier to treating mental illness during pregnancy is fear of fetal harm: teratogenicity, neonatal complications from drug exposure, and long-term neurodevelopmental effects. But these risks must be considered in light of real world experience – not just based on numbers from a study, Dr. Lusskin said. “Any risk of impairment has to be compared to the background risk of a birth defect, which is is 2%–4%. If a drug increases this risk in a clinically meaningful way, it has to be over and above this background rate.”
Even a substantial increase in a rare birth defect can be misleading. For instance, Ebstein's anomaly is a heart defect that occurs in 1 in 20,000 births. “Lithium is said to increase this rate to about 1 in 2,000 births. But this is still way below the expected background rate of birth defects. Risks are relevant in terms of their relativity – the absolute risk is just quoting a figure,” she said.
But untreated mental illness poses its own risk. Babies exposed in utero to the chronic stress hormones associated with anxiety and depressive disorders might become phenotypically programmed, prone themselves to early childhood behavioral problems and, in later life, to mental illness. “The fetal programming hypothesis is the interaction between the baby's genetics and the environmental exposure in utero, and it's mediated by the sex of the fetus and the timing of exposure,” Dr. Lusskin said.
Dr. Lusskin said she consults for the nonprofit drug safety database www.reprotox.org
Expert Analysis from the World Conference on Women's Mental Health
Poor Sleep May Drive Antenatal Depression
Major Finding: Pregnant women with severe physical discomfort in the first trimester might experience poor sleep, which could predispose them to antenatal depression.
Data Source: A prospective study of 257 pregnant women who were surveyed about physical symptoms, sleep quality, and mood in early and late pregnancy.
Disclosures: The Australian Research Council supported the study. Dr. Skouteris said she had no relevant financial disclosures.
MADRID – Poor sleep in early pregnancy might be one significant driver of depression in later pregnancy, an Australian prospective study has found.
“This finding suggests the need for clinicians to not just screen for depressive symptoms during pregnancy (which is the trend now), but to consider other potentially important problems, like the quality of sleep and severe physical symptoms during early pregnancy,” Helen Skouteris, Ph.D., said at the conference “These issues may very well contribute to the development of – or maintain – depression throughout pregnancy.”
Dr. Skouteris, an expert in developmental psychology at Deakin University in Burwood, Victoria, presented the results of a prospective study of 257 pregnant women in Australia who were recruited at 15–23 weeks' gestation. The study consisted of two self-reported questionnaires that assessed physical symptoms of pregnancy, sleep quality, and mood during early pregnancy and during the last trimester.
At recruitment, the women's mean age was 32 years. Almost half (48%) were first-time mothers and most (77%) were married. Most of the women (73%) also had at least some college education, and 57% had an annual household income of more than 75,000 Australian dollars ($77,500). “This is typical of the women who volunteer for our research programs,” Dr. Skouteris noted.
At the time of the first survey, the women had been pregnant for a mean of 18 weeks; the mean gestation at the time of the second survey was 34 weeks.
Both packets, which were mailed to the subjects, contained the self-administered Beck Depression Inventory, the Pittsburgh Sleep Quality Index, and a physical symptoms questionnaire that listed 29 common symptoms of pregnancy. Women were asked to rate these symptoms on a 0–3 scale (with 0 as “not at all” and 3 as “very much”) regarding discomfort from the symptoms, their frequency, and their general effect on life. Sleep was reported for the prior 4 weeks, physical symptoms for the prior 8 weeks, and mood at the time of the interview.
The current unpublished data, based on three regression analyses, show that poor sleep is the missing link between early physical symptoms and late depression. Fatigue was the most commonly reported physical symptom at both the early and later interviews (94% and 92%, respectively).
“The path seems to travel from severe physical symptoms early to poor sleep throughout, and then to depression later in pregnancy. Clearly, these results show that physical symptoms early in pregnancy might be a key risk factor for depression later on in pregnancy. Now we must consider other moderators in this relationship; there might be other things that contribute to this equation.”
She also mentioned that 15% of the women in the study rated very high on the anxiety measure.
“These women were very anxious about the delivery and what it might mean to their education, career, and general way of life,” Dr. Skouteris said. “We're now exploring these subjective ways a woman feels about her pregnancy, and their possible relationship between stress, anxiety, and depression throughout pregnancy and into the postnatal period.” This study will provide more data about these links, since the women will be assessed monthly instead of twice.
Major Finding: Pregnant women with severe physical discomfort in the first trimester might experience poor sleep, which could predispose them to antenatal depression.
Data Source: A prospective study of 257 pregnant women who were surveyed about physical symptoms, sleep quality, and mood in early and late pregnancy.
Disclosures: The Australian Research Council supported the study. Dr. Skouteris said she had no relevant financial disclosures.
MADRID – Poor sleep in early pregnancy might be one significant driver of depression in later pregnancy, an Australian prospective study has found.
“This finding suggests the need for clinicians to not just screen for depressive symptoms during pregnancy (which is the trend now), but to consider other potentially important problems, like the quality of sleep and severe physical symptoms during early pregnancy,” Helen Skouteris, Ph.D., said at the conference “These issues may very well contribute to the development of – or maintain – depression throughout pregnancy.”
Dr. Skouteris, an expert in developmental psychology at Deakin University in Burwood, Victoria, presented the results of a prospective study of 257 pregnant women in Australia who were recruited at 15–23 weeks' gestation. The study consisted of two self-reported questionnaires that assessed physical symptoms of pregnancy, sleep quality, and mood during early pregnancy and during the last trimester.
At recruitment, the women's mean age was 32 years. Almost half (48%) were first-time mothers and most (77%) were married. Most of the women (73%) also had at least some college education, and 57% had an annual household income of more than 75,000 Australian dollars ($77,500). “This is typical of the women who volunteer for our research programs,” Dr. Skouteris noted.
At the time of the first survey, the women had been pregnant for a mean of 18 weeks; the mean gestation at the time of the second survey was 34 weeks.
Both packets, which were mailed to the subjects, contained the self-administered Beck Depression Inventory, the Pittsburgh Sleep Quality Index, and a physical symptoms questionnaire that listed 29 common symptoms of pregnancy. Women were asked to rate these symptoms on a 0–3 scale (with 0 as “not at all” and 3 as “very much”) regarding discomfort from the symptoms, their frequency, and their general effect on life. Sleep was reported for the prior 4 weeks, physical symptoms for the prior 8 weeks, and mood at the time of the interview.
The current unpublished data, based on three regression analyses, show that poor sleep is the missing link between early physical symptoms and late depression. Fatigue was the most commonly reported physical symptom at both the early and later interviews (94% and 92%, respectively).
“The path seems to travel from severe physical symptoms early to poor sleep throughout, and then to depression later in pregnancy. Clearly, these results show that physical symptoms early in pregnancy might be a key risk factor for depression later on in pregnancy. Now we must consider other moderators in this relationship; there might be other things that contribute to this equation.”
She also mentioned that 15% of the women in the study rated very high on the anxiety measure.
“These women were very anxious about the delivery and what it might mean to their education, career, and general way of life,” Dr. Skouteris said. “We're now exploring these subjective ways a woman feels about her pregnancy, and their possible relationship between stress, anxiety, and depression throughout pregnancy and into the postnatal period.” This study will provide more data about these links, since the women will be assessed monthly instead of twice.
Major Finding: Pregnant women with severe physical discomfort in the first trimester might experience poor sleep, which could predispose them to antenatal depression.
Data Source: A prospective study of 257 pregnant women who were surveyed about physical symptoms, sleep quality, and mood in early and late pregnancy.
Disclosures: The Australian Research Council supported the study. Dr. Skouteris said she had no relevant financial disclosures.
MADRID – Poor sleep in early pregnancy might be one significant driver of depression in later pregnancy, an Australian prospective study has found.
“This finding suggests the need for clinicians to not just screen for depressive symptoms during pregnancy (which is the trend now), but to consider other potentially important problems, like the quality of sleep and severe physical symptoms during early pregnancy,” Helen Skouteris, Ph.D., said at the conference “These issues may very well contribute to the development of – or maintain – depression throughout pregnancy.”
Dr. Skouteris, an expert in developmental psychology at Deakin University in Burwood, Victoria, presented the results of a prospective study of 257 pregnant women in Australia who were recruited at 15–23 weeks' gestation. The study consisted of two self-reported questionnaires that assessed physical symptoms of pregnancy, sleep quality, and mood during early pregnancy and during the last trimester.
At recruitment, the women's mean age was 32 years. Almost half (48%) were first-time mothers and most (77%) were married. Most of the women (73%) also had at least some college education, and 57% had an annual household income of more than 75,000 Australian dollars ($77,500). “This is typical of the women who volunteer for our research programs,” Dr. Skouteris noted.
At the time of the first survey, the women had been pregnant for a mean of 18 weeks; the mean gestation at the time of the second survey was 34 weeks.
Both packets, which were mailed to the subjects, contained the self-administered Beck Depression Inventory, the Pittsburgh Sleep Quality Index, and a physical symptoms questionnaire that listed 29 common symptoms of pregnancy. Women were asked to rate these symptoms on a 0–3 scale (with 0 as “not at all” and 3 as “very much”) regarding discomfort from the symptoms, their frequency, and their general effect on life. Sleep was reported for the prior 4 weeks, physical symptoms for the prior 8 weeks, and mood at the time of the interview.
The current unpublished data, based on three regression analyses, show that poor sleep is the missing link between early physical symptoms and late depression. Fatigue was the most commonly reported physical symptom at both the early and later interviews (94% and 92%, respectively).
“The path seems to travel from severe physical symptoms early to poor sleep throughout, and then to depression later in pregnancy. Clearly, these results show that physical symptoms early in pregnancy might be a key risk factor for depression later on in pregnancy. Now we must consider other moderators in this relationship; there might be other things that contribute to this equation.”
She also mentioned that 15% of the women in the study rated very high on the anxiety measure.
“These women were very anxious about the delivery and what it might mean to their education, career, and general way of life,” Dr. Skouteris said. “We're now exploring these subjective ways a woman feels about her pregnancy, and their possible relationship between stress, anxiety, and depression throughout pregnancy and into the postnatal period.” This study will provide more data about these links, since the women will be assessed monthly instead of twice.
From the World Conference on Women's Mental Health