User login
Every year the Advisory Committee on Immunization Practices (ACIP) updates the recommended immunization schedules for children/adolescents and adults on the Web site of the Centers for Disease Control and Prevention (www.cdc.gov/vaccines/schedules/hcp/index.html). The schedules for 2020 reflect additions and changes adopted by ACIP in 2019 and are discussed in this Practice Alert.
Hepatitis A: New directives on homelessness, HIV, and vaccine catch-up
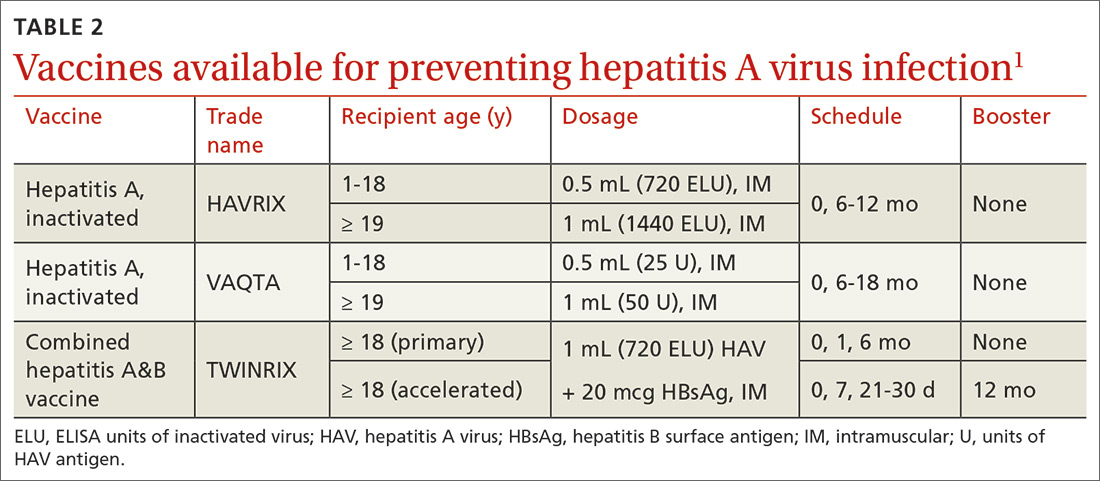
Hepatitis A (HepA) vaccination is recommended for children ages 12 to 23 months, and for those at increased risk for hepatitis A virus (HAV) infection or for complications from HAV infection (TABLE 1).1-3 Routine vaccination is either 2 doses of HepA given 6 months apart or a 3-dose schedule of combined hepatitis A and B vaccine (Twinrix). Vaccines licensed in the United States for the prevention of HAV infection are listed in TABLE 2.1

ACIP recently added homeless individuals to the list of those who should receive HepA vaccine.4 This step was taken in response to numerous outbreaks among those who are homeless or who use illicit drugs. These outbreaks have increased rates of HAV infection overall as well as rates of hospitalization (71%) and death (3%) among those infected.5 Concern about a homeless individual’s ability to complete a 2- or 3-dose series should not preclude initiating HepA vaccination; even 1 dose achieves protective immunity in 94% to 100% of those who have intact immune systems.2
At its June 2019 meeting, ACIP made 2 other additions to its recommendations regarding HepA vaccination.1 First, those infected with the human immunodeficiency virus (HIV) are now among the individuals who should receive HepA vaccine. Those who are HIV-positive and ≥ 1 year old were recommended for HepA vaccination because they often have one of the other risks for HAV infection and have higher rates of complications and prolonged infections if they contract HAV.1 Second, catch-up HepA vaccination is indicated for children and adolescents ages 2 through 18 years who have not been previously vaccinated.1Also at the June 2019 meeting, the safety of HepA vaccination during pregnancy was confirmed. ACIP recommends HepA vaccine for any pregnant woman not previously vaccinated who is at risk for HAV infection or for a severe outcome from HAV infection.1
Japanese encephalitis: Vaccination can be accelerated
Japanese encephalitis (JE) is a serious mosquito-borne vaccine-preventable infection endemic to most of Asia and parts of the western Pacific. Most travelers to countries with endemic JE are at low risk of infection. But risk increases with prolonged visits to these areas and particularly during the JE virus transmission season (summer/fall in temperate areas; year-round in tropical climates). Risk is also heightened by traveling to, or living in, rural Asian areas, by participating in extensive outdoor activities, and by staying in accommodations without air-conditioning, screens, or bed nets.6
The only JE vaccine licensed in the United States is JE-VC (Ixiaro), manufactured by Valneva Austria GmbH. It is approved for use in children ≥ 2 months and adults. It requires a 2-dose series with 28 days between doses, and a booster after 1 year. ACIP recently approved an accelerated schedule for adults ages 18 to 65 years that allows the second dose to be administered as early as 7 days after the first. A full description of the epidemiology of JE and ACIP recommendations regarding JE-VC were published in July 2019.6
Meningococcal B vaccine booster doses recommended
Meningococcal B (MenB) vaccine is recommended for individuals ≥ 10 years old who are at increased risk of meningococcal infection, including those with complement deficiency, complement inhibitor use, or asplenia; microbiologists; and individuals exposed during an outbreak.7 It is also recommended for those ages 16 to 23 years who desire vaccination after individual clinical decision making.8
Continue to: Two MenB vaccines...
Two MenB vaccines are available in the United States: MenB-FHbp (Trumenba, Wyeth Pharmaceuticals, Inc.) and MenB-4C (Bexsero, GlaxoSmithKline). Either MenB vaccine can be used; however, they are not interchangeable and the same product must be used for all doses an individual receives. MenB-FHbp is licensed as a 3-dose series given at 0, 1-2, and 6 months, or as a 2-dose series given at 0 and 6 months. ACIP recommends the 3-dose schedule for individuals at increased risk for meningococcal disease or for use during community outbreaks of serogroup B meningococcal disease.9 For healthy adolescents who are not at increased risk for meningococcal disease, ACIP recommends using the 2-dose schedule of MenB-FHbp.9 MenB-4C is licensed as a 2-dose series, with doses administered at least 1 month apart.
At the June 2019 meeting, ACIP voted to recommend a MenB booster dose for those who are still at increased risk 1 year following completion of a MenB primary series, followed by booster doses every 2 to 3 years thereafter for as long as increased risk remains. This recommendation was made because of a rapid waning of immunity following the primary series and subsequent booster doses. A booster dose was not recommended for those who choose to be vaccinated after clinical decision making unless they are exposed during an outbreak and it has been at least a year since they received the primary series. An interval of 6 months for the booster can be considered, depending on the outbreak situation.10
A new DTaP product, and substituting Tdap for Td is approved
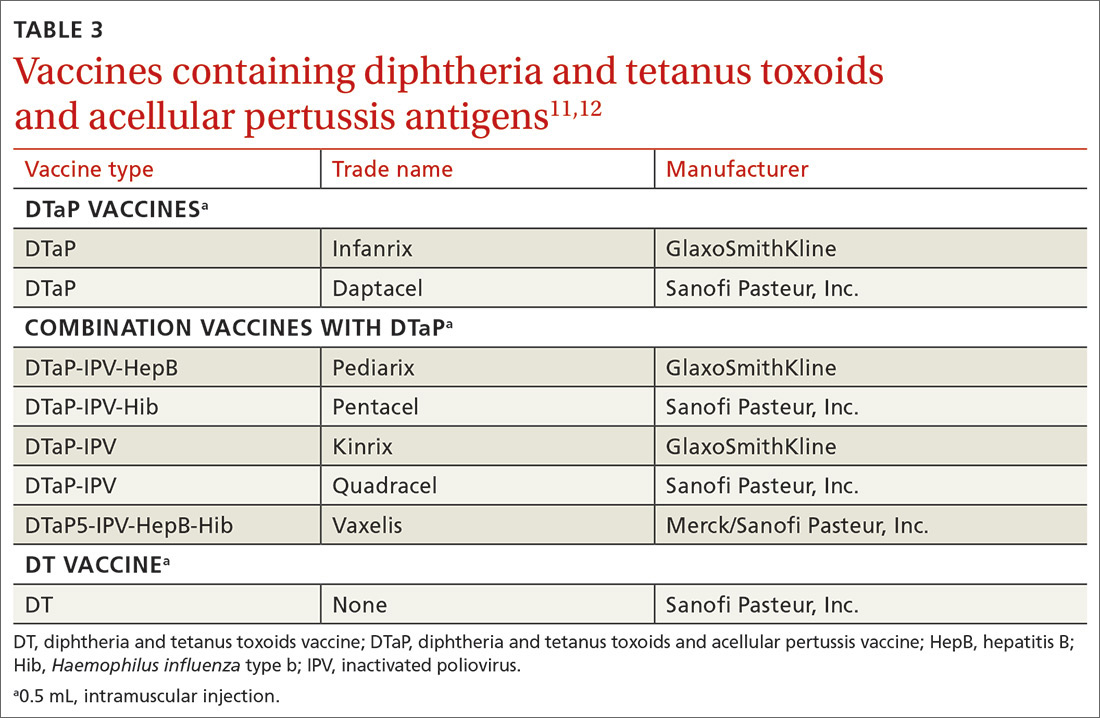
Diphtheria and tetanus toxoids and acellular pertussis vaccine (DTaP) is recommended for children as a 3-dose primary series (2, 4, 6 months) followed by 2 booster doses (at 15-18 months and at 4-6 years). These 3 antigens are available as DTaP products solely or as part of vaccines that combine other antigens with DTaP (TABLE 3).11,12 In addition, as a joint venture between Merck and Sanofi Pasteur, a new pediatric hexavalent vaccine containing DTaP5, polio, Haemophilus influenzae type b, and hepatitis B antigens is now available to be given at ages 2, 4, and 6 months.12
Tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccine is recommended for adolescents ages 11 to 12 years.11 It is also recommended once for adults who have not previously received it. The exception to the single Tdap dose for adults is during pregnancy; it is recommended as a single dose during each pregnancy regardless of the previous number of Tdap doses received.11
Td is recommended every 10 years after Tdap given at ages 11 to 12, for protection against tetanus and diphtheria. Tdap can be substituted for one of these decennial Td boosters. Tdap can also be substituted for Td for tetanus prophylaxis after a patient sustains a wound.11 The recommended single dose of Tdap for adolescents/adults also can be administered as part of a catch-up 3-dose Td series in previously unvaccinated adolescents and adults.
Continue to: It has become common...
It has become common practice throughout the country to substitute Tdap for Td when Td is indicated, even if Tdap has been received previously. ACIP looked at the safety of repeated doses of Tdap and found no safety concerns. For practicality, ACIP voted to recommend either Td or Tdap for these situations: the decennial booster, when tetanus prophylaxis is indicated in wound management, and when catch-up is needed in previously unvaccinated or inadequately vaccinated individuals who are 7 years of age and older. The resulting increase in the number of Tdap doses is not expected to have a major impact on the incidence of pertussis.13
Additional recommendations
Recommendations for preventing influenza in the 2019-2020 season are discussed in a previous Practice Alert.14
In 2019, ACIP also changed a previous recommendation on the routine use of 13-valent pneumococcal conjugate vaccine (PCV13) in adults ≥ 65 years. The new recommendation, covered in another Practice Alert, states that PCV13 should be used in immunocompetent adults ≥ 65 years only after individual clinical decision making.15
ACIP also changed its recommendations pertaining to human papillomavirus (HPV) vaccine. Catch-up vaccination is now recommended for all individuals through age 26 years. Previously catch up was recommended only for women and for men who have sex with men. And, even though use of HPV vaccine has been approved by the US Food and Drug Administration for adults ages 27 to 45 years, ACIP did not recommend its routine use in this age group but instead recommended it only after individual clinical decision making.16,17
1. Nelson N. Hepatitis A vaccine. Presentation to the ACIP; June 27, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-06/Hepatitis-2-Nelson-508.pdf. Accessed February 24, 2020.
2. Fiore AE, Wasley A, Bell BP; Advisory Committee on Immunization Practices (ACIP). Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2006;55(No. RR-7):1-23.
3. CDC. Prevention of hepatitis A through active or passive immunization. MMWR Wkly. 2006;55:1-23.
4. Doshani M, Weng M, Moore KL, et al. Recommendations of the Advisory Committee on Immunization Practices for use of hepatitis A vaccine for persons experiencing homelessness. MMWR Morb Mortal Wkly Rep. 2019;68:153-156.
5. Foster M, Ramachandran S, Myatt K, et al. Hepatitis A virus outbreaks associated with drug use and homelessness—California, Kentucky, Michigan, and Utah, 2017. MMWR Morb Mortal Wkly Rep. 2018;67:1208-1210.
6. Hills SL, Walter EB, Atmar RL, et al. Japanese encephalitis vaccine: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2019;68:1-33.
7. CDC. Meningococcal vaccination: what everyone should know. www.cdc.gov/vaccines/vpd/mening/public/index.html. Accessed February 24, 2020.
8. MacNeil JR, Rubin L, Folaranmi T, et al. Use of seroproup B meningococcal vaccine in adolescents and young adults: recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2015; 64:1171-1176.
9. Patton M, Stephens D, Moore K, et al. Updated recommendations for use of MenB-FHbp seropgroup B meningococcal vaccine—Advisory Committee on Immunization Practices, 2016. MMWR Morb Mortal Wkly Rep. 2017;66:509-513.
10. Mbaeyi S. Serogroup B Meningococcal vaccine booster doses. Presentation to ACIP; June 27, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-06/Meningococcal-2-Mbaeyi-508.pdf. Accessed February 24, 2020.
11. Liang JL, Tiwari T, Moro P, et al. Prevention of pertussis, tetanus, and diphtheria with vaccines in the United States: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2018;67(No. RR-2):1-44.
12. Lee A. Immunogenicity and safety of DTaP5-IPV-HepB-Hib (Vaxelis™), a pediatric hexavalent combination vaccine. Presentation to the Advisory Committee on Immunization Practices; February 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-02/Combo-vaccine-2-Lee-508.pdf. Accessed February 24, 2020.
13. Havers F. Tdap and Td: summary of work group considerations and proposed policy options. Presentation to ACIP; October 23, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-10/Pertussis-03-Havers-508.pdf. Accessed February 24, 2020.
14. Campos-Outcalt D. Influenza update. J Fam Pract. 2019;68:456-458.
15. Campos-Outcalt D. Pneumococcal conjugate vaccine update. J Fam Pract. 2019;68:564-566.
16. Meites E, Szilagyi PG, Chesson HW, et al. Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2019; 68:698–702.
17. Campos-Outcalt D. ACIP issues 2 new recs on HPV vaccine [audio]. J Fam Pract. September 2019. www.mdedge.com/familymedicine/article/205784/vaccines/acip-issues-2-new-recs-hpv-vaccination. Accessed February 24, 2020.
Every year the Advisory Committee on Immunization Practices (ACIP) updates the recommended immunization schedules for children/adolescents and adults on the Web site of the Centers for Disease Control and Prevention (www.cdc.gov/vaccines/schedules/hcp/index.html). The schedules for 2020 reflect additions and changes adopted by ACIP in 2019 and are discussed in this Practice Alert.
Hepatitis A: New directives on homelessness, HIV, and vaccine catch-up
Hepatitis A (HepA) vaccination is recommended for children ages 12 to 23 months, and for those at increased risk for hepatitis A virus (HAV) infection or for complications from HAV infection (TABLE 1).1-3 Routine vaccination is either 2 doses of HepA given 6 months apart or a 3-dose schedule of combined hepatitis A and B vaccine (Twinrix). Vaccines licensed in the United States for the prevention of HAV infection are listed in TABLE 2.1

ACIP recently added homeless individuals to the list of those who should receive HepA vaccine.4 This step was taken in response to numerous outbreaks among those who are homeless or who use illicit drugs. These outbreaks have increased rates of HAV infection overall as well as rates of hospitalization (71%) and death (3%) among those infected.5 Concern about a homeless individual’s ability to complete a 2- or 3-dose series should not preclude initiating HepA vaccination; even 1 dose achieves protective immunity in 94% to 100% of those who have intact immune systems.2

At its June 2019 meeting, ACIP made 2 other additions to its recommendations regarding HepA vaccination.1 First, those infected with the human immunodeficiency virus (HIV) are now among the individuals who should receive HepA vaccine. Those who are HIV-positive and ≥ 1 year old were recommended for HepA vaccination because they often have one of the other risks for HAV infection and have higher rates of complications and prolonged infections if they contract HAV.1 Second, catch-up HepA vaccination is indicated for children and adolescents ages 2 through 18 years who have not been previously vaccinated.1Also at the June 2019 meeting, the safety of HepA vaccination during pregnancy was confirmed. ACIP recommends HepA vaccine for any pregnant woman not previously vaccinated who is at risk for HAV infection or for a severe outcome from HAV infection.1
Japanese encephalitis: Vaccination can be accelerated
Japanese encephalitis (JE) is a serious mosquito-borne vaccine-preventable infection endemic to most of Asia and parts of the western Pacific. Most travelers to countries with endemic JE are at low risk of infection. But risk increases with prolonged visits to these areas and particularly during the JE virus transmission season (summer/fall in temperate areas; year-round in tropical climates). Risk is also heightened by traveling to, or living in, rural Asian areas, by participating in extensive outdoor activities, and by staying in accommodations without air-conditioning, screens, or bed nets.6
The only JE vaccine licensed in the United States is JE-VC (Ixiaro), manufactured by Valneva Austria GmbH. It is approved for use in children ≥ 2 months and adults. It requires a 2-dose series with 28 days between doses, and a booster after 1 year. ACIP recently approved an accelerated schedule for adults ages 18 to 65 years that allows the second dose to be administered as early as 7 days after the first. A full description of the epidemiology of JE and ACIP recommendations regarding JE-VC were published in July 2019.6
Meningococcal B vaccine booster doses recommended
Meningococcal B (MenB) vaccine is recommended for individuals ≥ 10 years old who are at increased risk of meningococcal infection, including those with complement deficiency, complement inhibitor use, or asplenia; microbiologists; and individuals exposed during an outbreak.7 It is also recommended for those ages 16 to 23 years who desire vaccination after individual clinical decision making.8
Continue to: Two MenB vaccines...
Two MenB vaccines are available in the United States: MenB-FHbp (Trumenba, Wyeth Pharmaceuticals, Inc.) and MenB-4C (Bexsero, GlaxoSmithKline). Either MenB vaccine can be used; however, they are not interchangeable and the same product must be used for all doses an individual receives. MenB-FHbp is licensed as a 3-dose series given at 0, 1-2, and 6 months, or as a 2-dose series given at 0 and 6 months. ACIP recommends the 3-dose schedule for individuals at increased risk for meningococcal disease or for use during community outbreaks of serogroup B meningococcal disease.9 For healthy adolescents who are not at increased risk for meningococcal disease, ACIP recommends using the 2-dose schedule of MenB-FHbp.9 MenB-4C is licensed as a 2-dose series, with doses administered at least 1 month apart.
At the June 2019 meeting, ACIP voted to recommend a MenB booster dose for those who are still at increased risk 1 year following completion of a MenB primary series, followed by booster doses every 2 to 3 years thereafter for as long as increased risk remains. This recommendation was made because of a rapid waning of immunity following the primary series and subsequent booster doses. A booster dose was not recommended for those who choose to be vaccinated after clinical decision making unless they are exposed during an outbreak and it has been at least a year since they received the primary series. An interval of 6 months for the booster can be considered, depending on the outbreak situation.10
A new DTaP product, and substituting Tdap for Td is approved
Diphtheria and tetanus toxoids and acellular pertussis vaccine (DTaP) is recommended for children as a 3-dose primary series (2, 4, 6 months) followed by 2 booster doses (at 15-18 months and at 4-6 years). These 3 antigens are available as DTaP products solely or as part of vaccines that combine other antigens with DTaP (TABLE 3).11,12 In addition, as a joint venture between Merck and Sanofi Pasteur, a new pediatric hexavalent vaccine containing DTaP5, polio, Haemophilus influenzae type b, and hepatitis B antigens is now available to be given at ages 2, 4, and 6 months.12

Tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccine is recommended for adolescents ages 11 to 12 years.11 It is also recommended once for adults who have not previously received it. The exception to the single Tdap dose for adults is during pregnancy; it is recommended as a single dose during each pregnancy regardless of the previous number of Tdap doses received.11
Td is recommended every 10 years after Tdap given at ages 11 to 12, for protection against tetanus and diphtheria. Tdap can be substituted for one of these decennial Td boosters. Tdap can also be substituted for Td for tetanus prophylaxis after a patient sustains a wound.11 The recommended single dose of Tdap for adolescents/adults also can be administered as part of a catch-up 3-dose Td series in previously unvaccinated adolescents and adults.
Continue to: It has become common...
It has become common practice throughout the country to substitute Tdap for Td when Td is indicated, even if Tdap has been received previously. ACIP looked at the safety of repeated doses of Tdap and found no safety concerns. For practicality, ACIP voted to recommend either Td or Tdap for these situations: the decennial booster, when tetanus prophylaxis is indicated in wound management, and when catch-up is needed in previously unvaccinated or inadequately vaccinated individuals who are 7 years of age and older. The resulting increase in the number of Tdap doses is not expected to have a major impact on the incidence of pertussis.13
Additional recommendations
Recommendations for preventing influenza in the 2019-2020 season are discussed in a previous Practice Alert.14
In 2019, ACIP also changed a previous recommendation on the routine use of 13-valent pneumococcal conjugate vaccine (PCV13) in adults ≥ 65 years. The new recommendation, covered in another Practice Alert, states that PCV13 should be used in immunocompetent adults ≥ 65 years only after individual clinical decision making.15
ACIP also changed its recommendations pertaining to human papillomavirus (HPV) vaccine. Catch-up vaccination is now recommended for all individuals through age 26 years. Previously catch up was recommended only for women and for men who have sex with men. And, even though use of HPV vaccine has been approved by the US Food and Drug Administration for adults ages 27 to 45 years, ACIP did not recommend its routine use in this age group but instead recommended it only after individual clinical decision making.16,17
Every year the Advisory Committee on Immunization Practices (ACIP) updates the recommended immunization schedules for children/adolescents and adults on the Web site of the Centers for Disease Control and Prevention (www.cdc.gov/vaccines/schedules/hcp/index.html). The schedules for 2020 reflect additions and changes adopted by ACIP in 2019 and are discussed in this Practice Alert.
Hepatitis A: New directives on homelessness, HIV, and vaccine catch-up
Hepatitis A (HepA) vaccination is recommended for children ages 12 to 23 months, and for those at increased risk for hepatitis A virus (HAV) infection or for complications from HAV infection (TABLE 1).1-3 Routine vaccination is either 2 doses of HepA given 6 months apart or a 3-dose schedule of combined hepatitis A and B vaccine (Twinrix). Vaccines licensed in the United States for the prevention of HAV infection are listed in TABLE 2.1

ACIP recently added homeless individuals to the list of those who should receive HepA vaccine.4 This step was taken in response to numerous outbreaks among those who are homeless or who use illicit drugs. These outbreaks have increased rates of HAV infection overall as well as rates of hospitalization (71%) and death (3%) among those infected.5 Concern about a homeless individual’s ability to complete a 2- or 3-dose series should not preclude initiating HepA vaccination; even 1 dose achieves protective immunity in 94% to 100% of those who have intact immune systems.2

At its June 2019 meeting, ACIP made 2 other additions to its recommendations regarding HepA vaccination.1 First, those infected with the human immunodeficiency virus (HIV) are now among the individuals who should receive HepA vaccine. Those who are HIV-positive and ≥ 1 year old were recommended for HepA vaccination because they often have one of the other risks for HAV infection and have higher rates of complications and prolonged infections if they contract HAV.1 Second, catch-up HepA vaccination is indicated for children and adolescents ages 2 through 18 years who have not been previously vaccinated.1Also at the June 2019 meeting, the safety of HepA vaccination during pregnancy was confirmed. ACIP recommends HepA vaccine for any pregnant woman not previously vaccinated who is at risk for HAV infection or for a severe outcome from HAV infection.1
Japanese encephalitis: Vaccination can be accelerated
Japanese encephalitis (JE) is a serious mosquito-borne vaccine-preventable infection endemic to most of Asia and parts of the western Pacific. Most travelers to countries with endemic JE are at low risk of infection. But risk increases with prolonged visits to these areas and particularly during the JE virus transmission season (summer/fall in temperate areas; year-round in tropical climates). Risk is also heightened by traveling to, or living in, rural Asian areas, by participating in extensive outdoor activities, and by staying in accommodations without air-conditioning, screens, or bed nets.6
The only JE vaccine licensed in the United States is JE-VC (Ixiaro), manufactured by Valneva Austria GmbH. It is approved for use in children ≥ 2 months and adults. It requires a 2-dose series with 28 days between doses, and a booster after 1 year. ACIP recently approved an accelerated schedule for adults ages 18 to 65 years that allows the second dose to be administered as early as 7 days after the first. A full description of the epidemiology of JE and ACIP recommendations regarding JE-VC were published in July 2019.6
Meningococcal B vaccine booster doses recommended
Meningococcal B (MenB) vaccine is recommended for individuals ≥ 10 years old who are at increased risk of meningococcal infection, including those with complement deficiency, complement inhibitor use, or asplenia; microbiologists; and individuals exposed during an outbreak.7 It is also recommended for those ages 16 to 23 years who desire vaccination after individual clinical decision making.8
Continue to: Two MenB vaccines...
Two MenB vaccines are available in the United States: MenB-FHbp (Trumenba, Wyeth Pharmaceuticals, Inc.) and MenB-4C (Bexsero, GlaxoSmithKline). Either MenB vaccine can be used; however, they are not interchangeable and the same product must be used for all doses an individual receives. MenB-FHbp is licensed as a 3-dose series given at 0, 1-2, and 6 months, or as a 2-dose series given at 0 and 6 months. ACIP recommends the 3-dose schedule for individuals at increased risk for meningococcal disease or for use during community outbreaks of serogroup B meningococcal disease.9 For healthy adolescents who are not at increased risk for meningococcal disease, ACIP recommends using the 2-dose schedule of MenB-FHbp.9 MenB-4C is licensed as a 2-dose series, with doses administered at least 1 month apart.
At the June 2019 meeting, ACIP voted to recommend a MenB booster dose for those who are still at increased risk 1 year following completion of a MenB primary series, followed by booster doses every 2 to 3 years thereafter for as long as increased risk remains. This recommendation was made because of a rapid waning of immunity following the primary series and subsequent booster doses. A booster dose was not recommended for those who choose to be vaccinated after clinical decision making unless they are exposed during an outbreak and it has been at least a year since they received the primary series. An interval of 6 months for the booster can be considered, depending on the outbreak situation.10
A new DTaP product, and substituting Tdap for Td is approved
Diphtheria and tetanus toxoids and acellular pertussis vaccine (DTaP) is recommended for children as a 3-dose primary series (2, 4, 6 months) followed by 2 booster doses (at 15-18 months and at 4-6 years). These 3 antigens are available as DTaP products solely or as part of vaccines that combine other antigens with DTaP (TABLE 3).11,12 In addition, as a joint venture between Merck and Sanofi Pasteur, a new pediatric hexavalent vaccine containing DTaP5, polio, Haemophilus influenzae type b, and hepatitis B antigens is now available to be given at ages 2, 4, and 6 months.12

Tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccine is recommended for adolescents ages 11 to 12 years.11 It is also recommended once for adults who have not previously received it. The exception to the single Tdap dose for adults is during pregnancy; it is recommended as a single dose during each pregnancy regardless of the previous number of Tdap doses received.11
Td is recommended every 10 years after Tdap given at ages 11 to 12, for protection against tetanus and diphtheria. Tdap can be substituted for one of these decennial Td boosters. Tdap can also be substituted for Td for tetanus prophylaxis after a patient sustains a wound.11 The recommended single dose of Tdap for adolescents/adults also can be administered as part of a catch-up 3-dose Td series in previously unvaccinated adolescents and adults.
Continue to: It has become common...
It has become common practice throughout the country to substitute Tdap for Td when Td is indicated, even if Tdap has been received previously. ACIP looked at the safety of repeated doses of Tdap and found no safety concerns. For practicality, ACIP voted to recommend either Td or Tdap for these situations: the decennial booster, when tetanus prophylaxis is indicated in wound management, and when catch-up is needed in previously unvaccinated or inadequately vaccinated individuals who are 7 years of age and older. The resulting increase in the number of Tdap doses is not expected to have a major impact on the incidence of pertussis.13
Additional recommendations
Recommendations for preventing influenza in the 2019-2020 season are discussed in a previous Practice Alert.14
In 2019, ACIP also changed a previous recommendation on the routine use of 13-valent pneumococcal conjugate vaccine (PCV13) in adults ≥ 65 years. The new recommendation, covered in another Practice Alert, states that PCV13 should be used in immunocompetent adults ≥ 65 years only after individual clinical decision making.15
ACIP also changed its recommendations pertaining to human papillomavirus (HPV) vaccine. Catch-up vaccination is now recommended for all individuals through age 26 years. Previously catch up was recommended only for women and for men who have sex with men. And, even though use of HPV vaccine has been approved by the US Food and Drug Administration for adults ages 27 to 45 years, ACIP did not recommend its routine use in this age group but instead recommended it only after individual clinical decision making.16,17
1. Nelson N. Hepatitis A vaccine. Presentation to the ACIP; June 27, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-06/Hepatitis-2-Nelson-508.pdf. Accessed February 24, 2020.
2. Fiore AE, Wasley A, Bell BP; Advisory Committee on Immunization Practices (ACIP). Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2006;55(No. RR-7):1-23.
3. CDC. Prevention of hepatitis A through active or passive immunization. MMWR Wkly. 2006;55:1-23.
4. Doshani M, Weng M, Moore KL, et al. Recommendations of the Advisory Committee on Immunization Practices for use of hepatitis A vaccine for persons experiencing homelessness. MMWR Morb Mortal Wkly Rep. 2019;68:153-156.
5. Foster M, Ramachandran S, Myatt K, et al. Hepatitis A virus outbreaks associated with drug use and homelessness—California, Kentucky, Michigan, and Utah, 2017. MMWR Morb Mortal Wkly Rep. 2018;67:1208-1210.
6. Hills SL, Walter EB, Atmar RL, et al. Japanese encephalitis vaccine: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2019;68:1-33.
7. CDC. Meningococcal vaccination: what everyone should know. www.cdc.gov/vaccines/vpd/mening/public/index.html. Accessed February 24, 2020.
8. MacNeil JR, Rubin L, Folaranmi T, et al. Use of seroproup B meningococcal vaccine in adolescents and young adults: recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2015; 64:1171-1176.
9. Patton M, Stephens D, Moore K, et al. Updated recommendations for use of MenB-FHbp seropgroup B meningococcal vaccine—Advisory Committee on Immunization Practices, 2016. MMWR Morb Mortal Wkly Rep. 2017;66:509-513.
10. Mbaeyi S. Serogroup B Meningococcal vaccine booster doses. Presentation to ACIP; June 27, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-06/Meningococcal-2-Mbaeyi-508.pdf. Accessed February 24, 2020.
11. Liang JL, Tiwari T, Moro P, et al. Prevention of pertussis, tetanus, and diphtheria with vaccines in the United States: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2018;67(No. RR-2):1-44.
12. Lee A. Immunogenicity and safety of DTaP5-IPV-HepB-Hib (Vaxelis™), a pediatric hexavalent combination vaccine. Presentation to the Advisory Committee on Immunization Practices; February 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-02/Combo-vaccine-2-Lee-508.pdf. Accessed February 24, 2020.
13. Havers F. Tdap and Td: summary of work group considerations and proposed policy options. Presentation to ACIP; October 23, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-10/Pertussis-03-Havers-508.pdf. Accessed February 24, 2020.
14. Campos-Outcalt D. Influenza update. J Fam Pract. 2019;68:456-458.
15. Campos-Outcalt D. Pneumococcal conjugate vaccine update. J Fam Pract. 2019;68:564-566.
16. Meites E, Szilagyi PG, Chesson HW, et al. Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2019; 68:698–702.
17. Campos-Outcalt D. ACIP issues 2 new recs on HPV vaccine [audio]. J Fam Pract. September 2019. www.mdedge.com/familymedicine/article/205784/vaccines/acip-issues-2-new-recs-hpv-vaccination. Accessed February 24, 2020.
1. Nelson N. Hepatitis A vaccine. Presentation to the ACIP; June 27, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-06/Hepatitis-2-Nelson-508.pdf. Accessed February 24, 2020.
2. Fiore AE, Wasley A, Bell BP; Advisory Committee on Immunization Practices (ACIP). Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2006;55(No. RR-7):1-23.
3. CDC. Prevention of hepatitis A through active or passive immunization. MMWR Wkly. 2006;55:1-23.
4. Doshani M, Weng M, Moore KL, et al. Recommendations of the Advisory Committee on Immunization Practices for use of hepatitis A vaccine for persons experiencing homelessness. MMWR Morb Mortal Wkly Rep. 2019;68:153-156.
5. Foster M, Ramachandran S, Myatt K, et al. Hepatitis A virus outbreaks associated with drug use and homelessness—California, Kentucky, Michigan, and Utah, 2017. MMWR Morb Mortal Wkly Rep. 2018;67:1208-1210.
6. Hills SL, Walter EB, Atmar RL, et al. Japanese encephalitis vaccine: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2019;68:1-33.
7. CDC. Meningococcal vaccination: what everyone should know. www.cdc.gov/vaccines/vpd/mening/public/index.html. Accessed February 24, 2020.
8. MacNeil JR, Rubin L, Folaranmi T, et al. Use of seroproup B meningococcal vaccine in adolescents and young adults: recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2015; 64:1171-1176.
9. Patton M, Stephens D, Moore K, et al. Updated recommendations for use of MenB-FHbp seropgroup B meningococcal vaccine—Advisory Committee on Immunization Practices, 2016. MMWR Morb Mortal Wkly Rep. 2017;66:509-513.
10. Mbaeyi S. Serogroup B Meningococcal vaccine booster doses. Presentation to ACIP; June 27, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-06/Meningococcal-2-Mbaeyi-508.pdf. Accessed February 24, 2020.
11. Liang JL, Tiwari T, Moro P, et al. Prevention of pertussis, tetanus, and diphtheria with vaccines in the United States: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2018;67(No. RR-2):1-44.
12. Lee A. Immunogenicity and safety of DTaP5-IPV-HepB-Hib (Vaxelis™), a pediatric hexavalent combination vaccine. Presentation to the Advisory Committee on Immunization Practices; February 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-02/Combo-vaccine-2-Lee-508.pdf. Accessed February 24, 2020.
13. Havers F. Tdap and Td: summary of work group considerations and proposed policy options. Presentation to ACIP; October 23, 2019. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2019-10/Pertussis-03-Havers-508.pdf. Accessed February 24, 2020.
14. Campos-Outcalt D. Influenza update. J Fam Pract. 2019;68:456-458.
15. Campos-Outcalt D. Pneumococcal conjugate vaccine update. J Fam Pract. 2019;68:564-566.
16. Meites E, Szilagyi PG, Chesson HW, et al. Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2019; 68:698–702.
17. Campos-Outcalt D. ACIP issues 2 new recs on HPV vaccine [audio]. J Fam Pract. September 2019. www.mdedge.com/familymedicine/article/205784/vaccines/acip-issues-2-new-recs-hpv-vaccination. Accessed February 24, 2020.