User login
Streamlining the Acute Care Pharmacy Consultation Process for Patients With Dysphagia or Enteral Feeding Tubes
Streamlining the Acute Care Pharmacy Consultation Process for Patients With Dysphagia or Enteral Feeding Tubes
Medication regimens may require adjustment in acute care settings due to dysphagia and/or enteral feeding tubes. When a patient has dysphagia and/or a feeding tube, the health care team must review the pharmacotherapy regimen to assess the appropriateness of medication formulations. Patient anatomy, the type of feeding tube in place, pharmacokinetic and pharmacodynamic properties of medications, risk of feeding tube obstruction, and potential for interactions between enteral nutrition and medications should be considered when clinicians administer medications through feeding tubes. The risk of feeding tube obstruction and clogging rises with increasing tube length and decreasing tube lumen. Incidence of obstructed percutaneous endoscopic gastrotomy tubes is reported to be 23% to 35%.1
A coordinated effort by all members of the health care team is essential to provide safe and effective care to patients with dysphagia and/or enteral feeding tubes. To decrease the risk of feeding tube obstruction, medications should be dissolved in water or administered in liquid form, saline fluids should be avoided, and the tube should be flushed with water before and after administering medications.
The pharmacokinetics of medications can be altered when tablets are crushed or capsules are opened. The bioavailability of dabigatran, for example, increases by 75% when the capsules are opened and pellets are taken orally.2 Medications may become intolerable after manipulation due to taste.3 Others may also increase the risk of feeding tube obstruction, such as omeprazole granules that increase the risk of small-bore feeding tube obstruction.4
Prior assessments of drug administration for patients with dysphagia and/or enteral feeding tubes has shown medication errors are prevalent.5-7 The Institute for Safe Medication Practices (ISMP) issued a Medication Safety Alert that provides a framework for preventing medication errors when preparing and administering medications via enteral feeding tubes.8 Other resources, such as monographs, are also available to guide pharmacotherapy decisions when oral medications require manipulation for administration to patients with dysphagia and/or enteral feeding tubes.9-11
In 2021, the Kansas City Veterans Affairs Medical Center (KCVAMC) was recognized as a Veterans Health Administration (VHA) Shark Tank finalist for improving the safety of medication administration for patients with enteral feeding tubes.12 This involved the addition of a Computerized Patient Record System (CPRS), clinical reminder order check (CROC), and a comprehensive medication review by a pharmacist. After implementing the CROC alert and pharmacy e-consultation workflow, the KCVAMC team reported that the number of inappropriate medications (ie, drugs on the ISMP do not crush list) was reduced from 41 to 6 in 1 year, resulting in an 85.4% reduction in potential medication errors.13
In 2014, the Richard L. Roudebush VAMC (RLRVAMC) created a pharmacy consultation process for patients with dysphagia and/or enteral feeding tubes. Any clinician could place a pharmacy consultation in CPRS. A pharmacist then reviewed patient charts, medication information resources, the VA formulary, and RLRVAMC pharmacy inventory. The pharmacist conferred with the patient’s care team to adjust pharmacotherapy, completed a consultation note, and updated medication order comments in Veterans Health Information Systems and Technology Architecture (VistA). These comments interfaced with the barcode medication administration software for the health care professional administering medications.
Despite the 2014 quality improvement (QI) process, medication errors involving the inappropriate ordering, preparation, and administration of medications for patients with dysphagia and/or enteral feeding tubes continued to be reported. Additionally, anonymous feedback revealed that only 3 of 10 responding pharmacists were satisfied with the existing medication use process for patients with dysphagia and/or enteral feeding tubes. Pharmacists expressed concerns that (1) clinicians were inappropriately crushing and/or manipulating new medications that were ordered after pharmacy consultations; (2) there was a lack of comprehensive documentation in CPRS; and (3) there were too many manual steps in the process. In response, RLRVAMC initiated a new QI initiative to improve the medication use process for patients with dysphagia and/or enteral feeding tubes in the acute care setting.
Quality Improvement Project
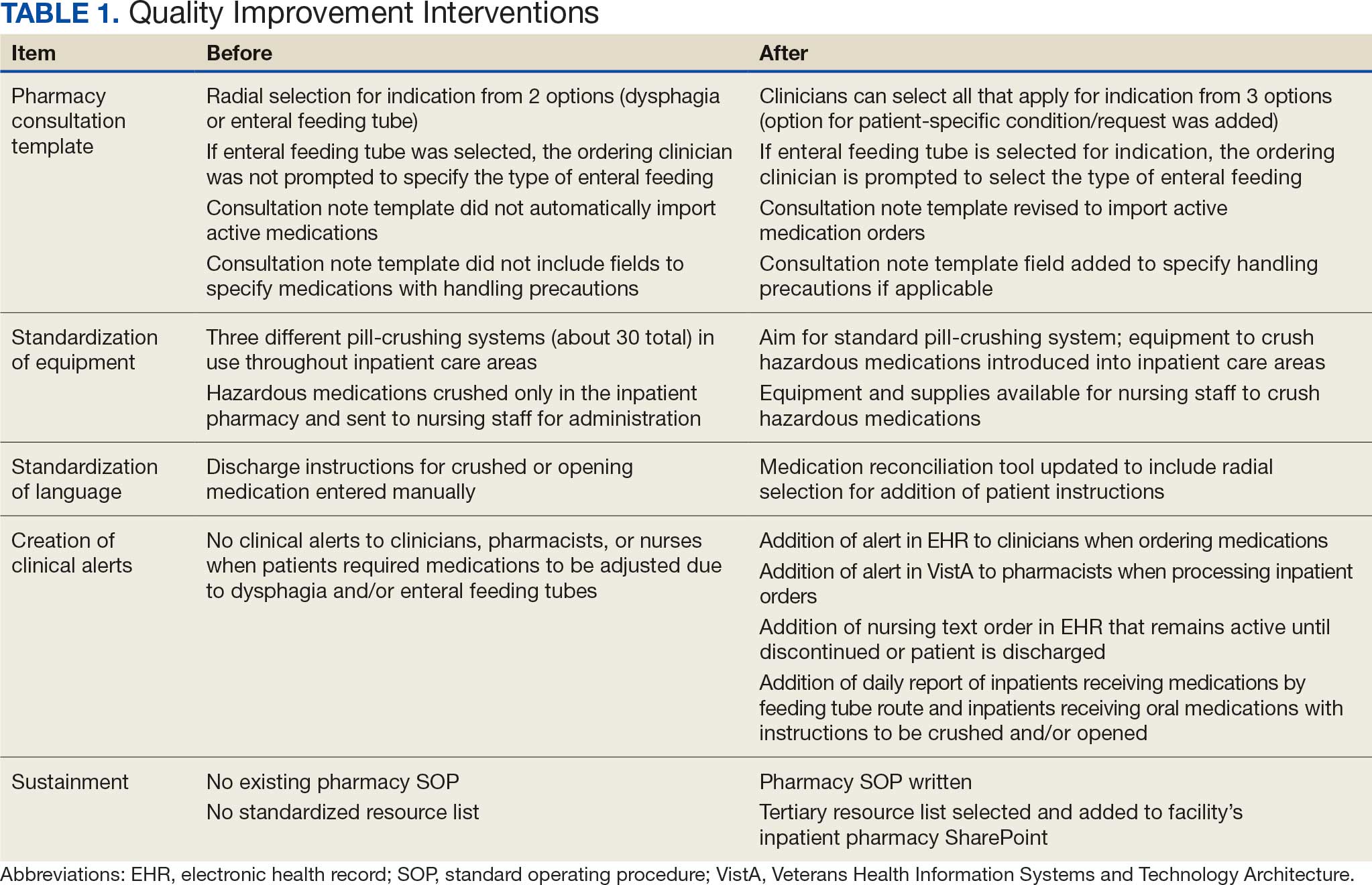
This multidisciplinary RLRVAMC QI project began November 2024 to improve pharmacotherapy care for patients with dysphagia and/or enteral feeding tubes in acute care. It was approved by the RLRVAMC Pharmacy Service. This intervention addressed the pharmacy consultation template, standardization of equipment, standardization of language, creation of clinical alerts, and sustainment (Table 1).
RLRVAMC has about 8600 annual inpatient admissions and 159 acute care beds.14 The project charter was drafted, and local stakeholders were identified including pharmacy technicians, pharmacists, nurses, speech language pathologists, and acute care clinicians. Pharmacy consultation workload was retrospectively reviewed to describe the scope of the existing state.
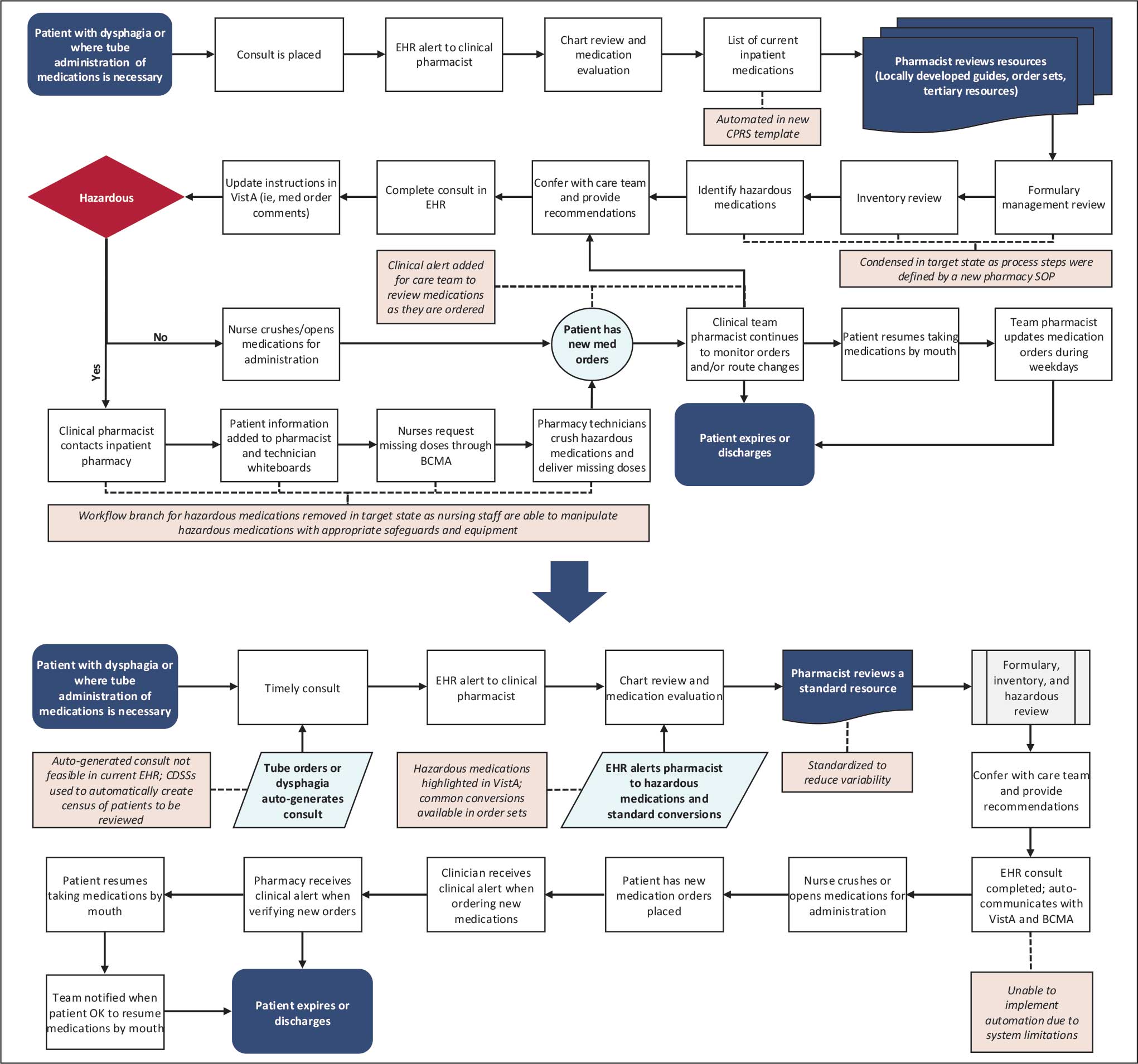
A workshop with 12 QI project stakeholders in December 2024 used A3 methodology to define the current process and the target state, barriers and solutions, prioritize interventions on an impact-effort matrix, perform a gap analysis, identify rapid plan-do-study-act (PDSA) experiments, and develop a completion plan (Figure). Five postworkshop PDSA experiments engaged additional stakeholders, clinical application coordinators, and medical supply representatives to ascertain the feasibility of the tools implemented.
Abbreviations: BCMA, barcode medication administration; CDSS, clinical decision support system; CPRS, Computerized Patient Record System;
EHR, electronic health record; SOP, standard operating procedure; VistA, Veterans Health Information Systems and Technology Architecture.
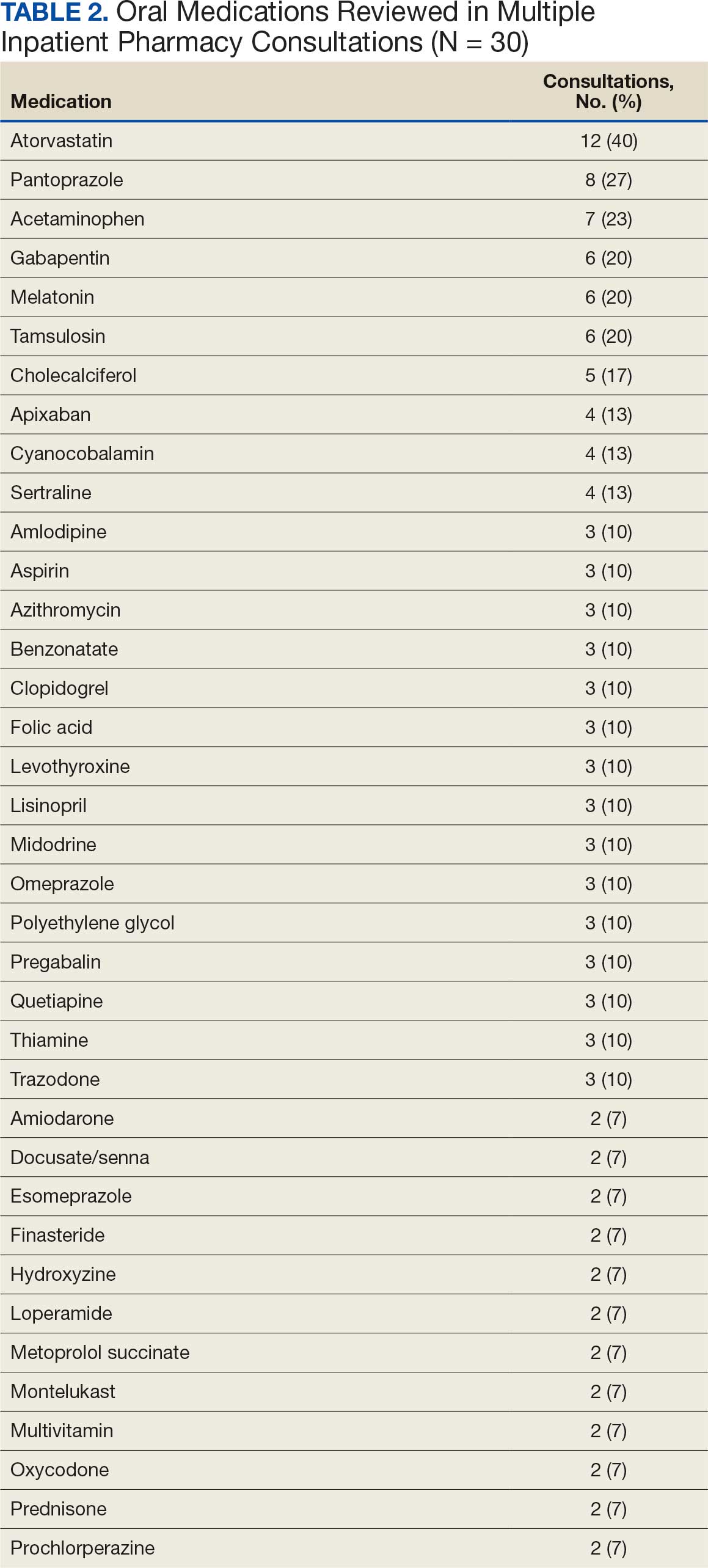
About 3% of RLRVAMC admissions involve a pharmacy consultation to review medications for dysphagia and/or enteral feeding tubes. Clinicians reviewed 30 preimplementation inpatient pharmacy consultations involving 200 oral medications. Pharmacists were more frequently consulted for inpatients with dysphagia (19 [63%]) than for patients with enteral feeding tubes (11 [37%]) (Table 2).
Pharmacy Consultation Template
The pharmacy consultation was updated in CPRS. Prior to this QI project, the ordering clinician was prompted to select 1 option for the indication: dysphagia or enteral feeding tube. The type of enteral feeding tube was not prompted by the consultation text nor required to be specified in the consultation. The ordering clinician could provide free-text comments. Of 11 preimplementation consultations, the type of enteral feeding tube was specified in 5 (45%). The consultation template entry was updated to include an option to check a box for the consultation indication from 3 options: dysphagia, enteral feeding, or other patient- specific condition/request. If enteral feeding tube is selected, then the clinician is prompted to select the type of enteral feeding tube. Since the completion of the project, there have been no patient safety reports concerning an erroneous or incomplete consultation entry (Supplemental Material).
The note template was updated to import the list of active inpatient medications and provide sections for the adjudicating pharmacist to document which medications can be crushed (or opened), which require adjustment, and which are hazardous and require special handling. Additionally, the revised template added a statement clarifying that the documented recommendations apply only to the medication regimen at the time of the consultation (Supplemental Material).
Standardizations
There are multiple pill-crushing devices used at RLRVAMC that vary in crushing mechanism, corresponding medication pouches, and degree of protection when manipulating hazardous medications. Prior to this QI project, RLRVAMC used 3 pill-crushing devices (about 30 total devices in inpatient care areas). Only 1 device with corresponding closed pouches for preparation of hazardous medications was available, which was stored in the RLRVAMC inpatient pharmacy. This workflow resulted in waste and posed potential risks for delays in care. This project incorporated a standard pill-crushing system with the corresponding medication pouches in all inpatient care areas, which provided safeguards for clinicians to prepare and administer hazardous medications (Supplemental Material).
Patients requiring medications to be crushed or opened on discharge should receive education, written instruction, and have care plans documented in CPRS. RLRVAMC patients receive education and a printed medication list. Prior to this QI project, the instructions for crushing or opening medications could only be entered by free text in the electronic medication reconciliation tool, allowing for the potential for inconsistent language or omissions.
This QI project included an update to the electronic medication reconciliation tool. An optional checkbox selection was added for patients requiring medications to be manipulated. When checked, a radial selection for individual medications is displayed, prompting the clinician and pharmacist to indicate either do not crush tablet or OK to crush tablet. These selections appear in clinical care notes and on the printed medication list provided to the patient (Supplemental Material).
Clinical Alerts
As part of the RLRVAMC QI initiative, a CROC alert was implemented, based on the KCVAMC intervention for patients with enteral feeding tubes.13 The RLRVAMC CROC alert also included patients with dysphagia. A nursing text order was made available in CPRS for patients requiring medications and remains active throughout the duration of the patient’s admission or until discontinued. It generates CROC alerts in CPRS and VistA when new medication orders are entered and reviewed by pharmacists.
Clinicians used clinical decision support systems to create daily lists of patients receiving medications by feeding tube and patients receiving crushed/opened medications due to dysphagia. This allows pharmacists to perform a census review of all inpatients to confirm appropriateness of medication orders. Clinical alerts for patients with enteral feeding tubes are advised by the ISMP and have data demonstrating a reduction in medication errors (Supplemental Material).14,15
Sustainment
During the sustainment phase, process owners were identified and a Pharmacy Service standard operating procedure (SOP) was written. The development of an institutional do not crush medication list was discussed; however, it was determined to be difficult to develop and maintain. An institutional tertiary resource list was selected in favor of a locally developed resource. These resources include the Handbook of Drug Administration via Enteral Feeding Tubes, Third Edition, the Pharmacist’s Letter list, “Meds that Should Not be Crushed,” and the Up- ToDate Lexidrug list, “Oral Medications That Should Not Be Crushed or Altered.”9-11 Links to the resources were added to the RLRVAMC pharmacy service SharePoint. In addition to defining the preferred tertiary resources, the SOP defined the process for reviewing inventory and the process for reviewing medication orders for hazard risk.
Discussion
Continued patient safety reports and low satisfaction rates among pharmacists prompted this QI project to improve safety for patients with dysphagia and/or enteral feeding tubes at RLRVAMC. The project engaged stakeholders and also identified and addressed gaps with potential for patient harm.
The tools implemented by this initiative drew from previous work by the KCVAMC and from framework provided by the ISMP.8,13 We expanded the QI intervention to include acute care patients with dysphagia.
RLRVAMC did not take steps to track the impact of the interventions on medication errors. However, no patient safety reports concerning an erroneous or incomplete pharmacy consultation entry have been reported. We also think that it is reasonable to assume that the adoption of the safety tools described here will have a positive impact on patient safety. RLRVAMC pharmacists have noted an increased appreciation for medication safety when processing medication orders for patients with dysphagia and/or enteral feeding tubes. While the workflow took time to adopt and integrate, clinical pharmacists perceived it as an improvement in patient safety. Our future focus is aimed at translating the process improvement into the Oracle/Cerner electronic health record, which is scheduled to be deployed at the RLRVAMC in August 2026.
Limitations
This QI project did not aim to quantify or compare medication errors before and after the intervention. An accurate number of unreported errors in the medication use process for patients with dysphagia and/or enteral feeding tubes would be challenging to quantify without direct observation. Multiple clinicians are engaged in the medication use process and individual steps may not be documented at all, or documented properly. In addition, medication errors are often underreported and may not reflect the total number of errors and/or potential for errors. That said, reported medication errors in the medication use process for patients with dysphagia and/or enteral feeding tubes are reviewed on a monthly basis by the RLRVAMC Multidisciplinary Medication Safety committee to continuously improve patient safety.
Another potential limitation is the extent to which the project can be adapted at other VHA sites. For example, RLRVAMC uses CPRS; the framework and tools to improve medication safety may not translate to sites using the Oracle/Cerner electronic health record. Furthermore, this QI project included a pharmacy consultation workflow that relied on pharmacists who are available at any hour. Other facilities may not have continuous consultation coverage to review medications for patients with dysphagia and/or enteral feeding tubes.
Conclusions
This QI project drew from ISMP recommendations, previous work within the VHA, local practice, and insight from multiple disciplines on the health care team to revise and create tools to improve medication safety for patients with dysphagia and/or enteral feeding tubes in the acute care setting. These tools included a revised pharmacy consultation workflow with improvements to the pharmacy consultation template, standardization of the pill-crushing devices and language used for patient medication lists, implementation of CROC alerts within the EHR, and development of an SOP.
The RLRVAMC Pharmacy Service intends to continue reviewing patient safety reports, assessing staff perspectives, and refining (and potentially adding) tools for medication safety. Future QI initiatives may focus on improving medication safety for outpatients with dysphagia and/or enteral feeding tubes. We also hope that these tools can be adapted at other VAMCs to promote medication safety for patients with dysphagia and/or enteral feeding tubes.
- Blumenstein I, Shastri YM, Stein J. Gastroenteric tube feeding: techniques, problems and solutions. World J Gastroenterol. 2014;20:8505-8524. doi:10.3748/wjg.v20.i26.8505
- Pradaxa (dabigatran etexilate). Prescribing information. Boehringer Ingelheim Pharmaceuticals, Inc; 2025. https:// pro.boehringer-ingelheim.com/us/products/pradaxa/bipdf /pradaxa-capsules-us-pi
- Lovell AG, Protus BM, Dickman JR, et al. Palatability of crushed over-the-counter medications. J Pain Symptom Manage. 2021;61:755-762. doi:10.1016/j.jpainsymman.2020.09.020
- Messaouik D, Sautou-Miranda V, Bagel-Boithias S, et al. Comparative study and optimisation of the administration mode of three proton pump inhibitors by nasogastric tube. Int J Pharm. 2005;299:65-72. doi:10.1016/j.ijpharm.2005.04.034
- Demirkan K, Bayraktar-Ekincioglu A, Gulhan-Halil M, et al. Assessment of drug administration via feeding tube and the knowledge of health-care professionals in a university hospital. Eur J Clin Nutr. 2017;71:164-168. doi:10.1038/ejcn.2016.147
- Fodil M, Nghiem D, Colas M, et al. Assessment of clinical practices for crushing medication in geriatric units. J Nutr Health Aging. 2017;21:904-908. doi:10.1007/s12603-017-0886-3
- Zhu LL, Xu LC, Wang HQ, et al. Appropriateness of administration of nasogastric medication and preliminary intervention. Ther Clin Risk Manag. 2012;8:393-401. doi:10.2147/TCRM.S37785
- Institute for Safe Medication Practices (ISMP). Preventing errors when preparing and administering medications via enteral feeding tubes. Acute Care ISMP Medication Safety Alert. November 17, 2022. Accessed March 17, 2026. https://nutritioncare.org/wp-content/uploads/2025/02 /ISMP-Safety-Alert_Medications-and-Enteral-Feeding -Tubes.pdf
- White R, Bradnam V. Handbook of Drug Administration via Enteral Feeding Tubes. 3rd ed. Pharmaceutical Press; 2015.
- Clinical resource, meds that should not be crushed. Pharmacist’s Letter/Pharmacy Technician’s Letter/Prescriber Insights. Updated April 2025. Accessed March 17, 2026. https://pharmacist.therapeuticresearch.com/en/Content /Segments/PRL/2014/Aug/Meds-That-Should-Not-Be -Crushed-7309
- Oral medications that should not be crushed or altered. In: Lexidrug. UpToDate, Inc. https://online.lexi.com/lco /action/doc/retrieve/docid/patch_f/4227
- Uttaro E, Zhao F, Schweighardt A. Filling the gaps on the Institute for Safe Medication Practices (ISMP) do not crush list for immediate-release products. Int J Pharm Compd. 2021;25:364-371.
- US Dept of Veterans Affairs. VA Diffusion Marketplace. Improved safety of enteral tube medication administration. Updated 2024. Accessed March 17, 2026. https:// marketplace.va.gov/innovations/improved-safety-of -enteral-tube-medication-administration
- US Dept of Veterans Affairs. About us. VA Indiana Healthcare System. Updated October 17, 2024. Accessed March 2, 2026. https://www.va.gov/indiana-health-care/about-us/
- Wasylewicz ATM, van Grinsven RJB, Bikker JMW, et al. Clinical decision support system-assisted pharmacy intervention reduces feeding tube-related medication errors in hospitalized patients: a focus on medication suitable for feeding-tube administration. JPEN J Parenter Enteral Nutr. 2021;45:625-632. doi:10.1002/jpen.1869
Medication regimens may require adjustment in acute care settings due to dysphagia and/or enteral feeding tubes. When a patient has dysphagia and/or a feeding tube, the health care team must review the pharmacotherapy regimen to assess the appropriateness of medication formulations. Patient anatomy, the type of feeding tube in place, pharmacokinetic and pharmacodynamic properties of medications, risk of feeding tube obstruction, and potential for interactions between enteral nutrition and medications should be considered when clinicians administer medications through feeding tubes. The risk of feeding tube obstruction and clogging rises with increasing tube length and decreasing tube lumen. Incidence of obstructed percutaneous endoscopic gastrotomy tubes is reported to be 23% to 35%.1
A coordinated effort by all members of the health care team is essential to provide safe and effective care to patients with dysphagia and/or enteral feeding tubes. To decrease the risk of feeding tube obstruction, medications should be dissolved in water or administered in liquid form, saline fluids should be avoided, and the tube should be flushed with water before and after administering medications.
The pharmacokinetics of medications can be altered when tablets are crushed or capsules are opened. The bioavailability of dabigatran, for example, increases by 75% when the capsules are opened and pellets are taken orally.2 Medications may become intolerable after manipulation due to taste.3 Others may also increase the risk of feeding tube obstruction, such as omeprazole granules that increase the risk of small-bore feeding tube obstruction.4
Prior assessments of drug administration for patients with dysphagia and/or enteral feeding tubes has shown medication errors are prevalent.5-7 The Institute for Safe Medication Practices (ISMP) issued a Medication Safety Alert that provides a framework for preventing medication errors when preparing and administering medications via enteral feeding tubes.8 Other resources, such as monographs, are also available to guide pharmacotherapy decisions when oral medications require manipulation for administration to patients with dysphagia and/or enteral feeding tubes.9-11
In 2021, the Kansas City Veterans Affairs Medical Center (KCVAMC) was recognized as a Veterans Health Administration (VHA) Shark Tank finalist for improving the safety of medication administration for patients with enteral feeding tubes.12 This involved the addition of a Computerized Patient Record System (CPRS), clinical reminder order check (CROC), and a comprehensive medication review by a pharmacist. After implementing the CROC alert and pharmacy e-consultation workflow, the KCVAMC team reported that the number of inappropriate medications (ie, drugs on the ISMP do not crush list) was reduced from 41 to 6 in 1 year, resulting in an 85.4% reduction in potential medication errors.13
In 2014, the Richard L. Roudebush VAMC (RLRVAMC) created a pharmacy consultation process for patients with dysphagia and/or enteral feeding tubes. Any clinician could place a pharmacy consultation in CPRS. A pharmacist then reviewed patient charts, medication information resources, the VA formulary, and RLRVAMC pharmacy inventory. The pharmacist conferred with the patient’s care team to adjust pharmacotherapy, completed a consultation note, and updated medication order comments in Veterans Health Information Systems and Technology Architecture (VistA). These comments interfaced with the barcode medication administration software for the health care professional administering medications.
Despite the 2014 quality improvement (QI) process, medication errors involving the inappropriate ordering, preparation, and administration of medications for patients with dysphagia and/or enteral feeding tubes continued to be reported. Additionally, anonymous feedback revealed that only 3 of 10 responding pharmacists were satisfied with the existing medication use process for patients with dysphagia and/or enteral feeding tubes. Pharmacists expressed concerns that (1) clinicians were inappropriately crushing and/or manipulating new medications that were ordered after pharmacy consultations; (2) there was a lack of comprehensive documentation in CPRS; and (3) there were too many manual steps in the process. In response, RLRVAMC initiated a new QI initiative to improve the medication use process for patients with dysphagia and/or enteral feeding tubes in the acute care setting.
Quality Improvement Project
This multidisciplinary RLRVAMC QI project began November 2024 to improve pharmacotherapy care for patients with dysphagia and/or enteral feeding tubes in acute care. It was approved by the RLRVAMC Pharmacy Service. This intervention addressed the pharmacy consultation template, standardization of equipment, standardization of language, creation of clinical alerts, and sustainment (Table 1).

RLRVAMC has about 8600 annual inpatient admissions and 159 acute care beds.14 The project charter was drafted, and local stakeholders were identified including pharmacy technicians, pharmacists, nurses, speech language pathologists, and acute care clinicians. Pharmacy consultation workload was retrospectively reviewed to describe the scope of the existing state.
A workshop with 12 QI project stakeholders in December 2024 used A3 methodology to define the current process and the target state, barriers and solutions, prioritize interventions on an impact-effort matrix, perform a gap analysis, identify rapid plan-do-study-act (PDSA) experiments, and develop a completion plan (Figure). Five postworkshop PDSA experiments engaged additional stakeholders, clinical application coordinators, and medical supply representatives to ascertain the feasibility of the tools implemented.

Abbreviations: BCMA, barcode medication administration; CDSS, clinical decision support system; CPRS, Computerized Patient Record System;
EHR, electronic health record; SOP, standard operating procedure; VistA, Veterans Health Information Systems and Technology Architecture.
About 3% of RLRVAMC admissions involve a pharmacy consultation to review medications for dysphagia and/or enteral feeding tubes. Clinicians reviewed 30 preimplementation inpatient pharmacy consultations involving 200 oral medications. Pharmacists were more frequently consulted for inpatients with dysphagia (19 [63%]) than for patients with enteral feeding tubes (11 [37%]) (Table 2).

Pharmacy Consultation Template
The pharmacy consultation was updated in CPRS. Prior to this QI project, the ordering clinician was prompted to select 1 option for the indication: dysphagia or enteral feeding tube. The type of enteral feeding tube was not prompted by the consultation text nor required to be specified in the consultation. The ordering clinician could provide free-text comments. Of 11 preimplementation consultations, the type of enteral feeding tube was specified in 5 (45%). The consultation template entry was updated to include an option to check a box for the consultation indication from 3 options: dysphagia, enteral feeding, or other patient- specific condition/request. If enteral feeding tube is selected, then the clinician is prompted to select the type of enteral feeding tube. Since the completion of the project, there have been no patient safety reports concerning an erroneous or incomplete consultation entry (Supplemental Material).
The note template was updated to import the list of active inpatient medications and provide sections for the adjudicating pharmacist to document which medications can be crushed (or opened), which require adjustment, and which are hazardous and require special handling. Additionally, the revised template added a statement clarifying that the documented recommendations apply only to the medication regimen at the time of the consultation (Supplemental Material).
Standardizations
There are multiple pill-crushing devices used at RLRVAMC that vary in crushing mechanism, corresponding medication pouches, and degree of protection when manipulating hazardous medications. Prior to this QI project, RLRVAMC used 3 pill-crushing devices (about 30 total devices in inpatient care areas). Only 1 device with corresponding closed pouches for preparation of hazardous medications was available, which was stored in the RLRVAMC inpatient pharmacy. This workflow resulted in waste and posed potential risks for delays in care. This project incorporated a standard pill-crushing system with the corresponding medication pouches in all inpatient care areas, which provided safeguards for clinicians to prepare and administer hazardous medications (Supplemental Material).
Patients requiring medications to be crushed or opened on discharge should receive education, written instruction, and have care plans documented in CPRS. RLRVAMC patients receive education and a printed medication list. Prior to this QI project, the instructions for crushing or opening medications could only be entered by free text in the electronic medication reconciliation tool, allowing for the potential for inconsistent language or omissions.
This QI project included an update to the electronic medication reconciliation tool. An optional checkbox selection was added for patients requiring medications to be manipulated. When checked, a radial selection for individual medications is displayed, prompting the clinician and pharmacist to indicate either do not crush tablet or OK to crush tablet. These selections appear in clinical care notes and on the printed medication list provided to the patient (Supplemental Material).
Clinical Alerts
As part of the RLRVAMC QI initiative, a CROC alert was implemented, based on the KCVAMC intervention for patients with enteral feeding tubes.13 The RLRVAMC CROC alert also included patients with dysphagia. A nursing text order was made available in CPRS for patients requiring medications and remains active throughout the duration of the patient’s admission or until discontinued. It generates CROC alerts in CPRS and VistA when new medication orders are entered and reviewed by pharmacists.
Clinicians used clinical decision support systems to create daily lists of patients receiving medications by feeding tube and patients receiving crushed/opened medications due to dysphagia. This allows pharmacists to perform a census review of all inpatients to confirm appropriateness of medication orders. Clinical alerts for patients with enteral feeding tubes are advised by the ISMP and have data demonstrating a reduction in medication errors (Supplemental Material).14,15
Sustainment
During the sustainment phase, process owners were identified and a Pharmacy Service standard operating procedure (SOP) was written. The development of an institutional do not crush medication list was discussed; however, it was determined to be difficult to develop and maintain. An institutional tertiary resource list was selected in favor of a locally developed resource. These resources include the Handbook of Drug Administration via Enteral Feeding Tubes, Third Edition, the Pharmacist’s Letter list, “Meds that Should Not be Crushed,” and the Up- ToDate Lexidrug list, “Oral Medications That Should Not Be Crushed or Altered.”9-11 Links to the resources were added to the RLRVAMC pharmacy service SharePoint. In addition to defining the preferred tertiary resources, the SOP defined the process for reviewing inventory and the process for reviewing medication orders for hazard risk.
Discussion
Continued patient safety reports and low satisfaction rates among pharmacists prompted this QI project to improve safety for patients with dysphagia and/or enteral feeding tubes at RLRVAMC. The project engaged stakeholders and also identified and addressed gaps with potential for patient harm.
The tools implemented by this initiative drew from previous work by the KCVAMC and from framework provided by the ISMP.8,13 We expanded the QI intervention to include acute care patients with dysphagia.
RLRVAMC did not take steps to track the impact of the interventions on medication errors. However, no patient safety reports concerning an erroneous or incomplete pharmacy consultation entry have been reported. We also think that it is reasonable to assume that the adoption of the safety tools described here will have a positive impact on patient safety. RLRVAMC pharmacists have noted an increased appreciation for medication safety when processing medication orders for patients with dysphagia and/or enteral feeding tubes. While the workflow took time to adopt and integrate, clinical pharmacists perceived it as an improvement in patient safety. Our future focus is aimed at translating the process improvement into the Oracle/Cerner electronic health record, which is scheduled to be deployed at the RLRVAMC in August 2026.
Limitations
This QI project did not aim to quantify or compare medication errors before and after the intervention. An accurate number of unreported errors in the medication use process for patients with dysphagia and/or enteral feeding tubes would be challenging to quantify without direct observation. Multiple clinicians are engaged in the medication use process and individual steps may not be documented at all, or documented properly. In addition, medication errors are often underreported and may not reflect the total number of errors and/or potential for errors. That said, reported medication errors in the medication use process for patients with dysphagia and/or enteral feeding tubes are reviewed on a monthly basis by the RLRVAMC Multidisciplinary Medication Safety committee to continuously improve patient safety.
Another potential limitation is the extent to which the project can be adapted at other VHA sites. For example, RLRVAMC uses CPRS; the framework and tools to improve medication safety may not translate to sites using the Oracle/Cerner electronic health record. Furthermore, this QI project included a pharmacy consultation workflow that relied on pharmacists who are available at any hour. Other facilities may not have continuous consultation coverage to review medications for patients with dysphagia and/or enteral feeding tubes.
Conclusions
This QI project drew from ISMP recommendations, previous work within the VHA, local practice, and insight from multiple disciplines on the health care team to revise and create tools to improve medication safety for patients with dysphagia and/or enteral feeding tubes in the acute care setting. These tools included a revised pharmacy consultation workflow with improvements to the pharmacy consultation template, standardization of the pill-crushing devices and language used for patient medication lists, implementation of CROC alerts within the EHR, and development of an SOP.
The RLRVAMC Pharmacy Service intends to continue reviewing patient safety reports, assessing staff perspectives, and refining (and potentially adding) tools for medication safety. Future QI initiatives may focus on improving medication safety for outpatients with dysphagia and/or enteral feeding tubes. We also hope that these tools can be adapted at other VAMCs to promote medication safety for patients with dysphagia and/or enteral feeding tubes.
Medication regimens may require adjustment in acute care settings due to dysphagia and/or enteral feeding tubes. When a patient has dysphagia and/or a feeding tube, the health care team must review the pharmacotherapy regimen to assess the appropriateness of medication formulations. Patient anatomy, the type of feeding tube in place, pharmacokinetic and pharmacodynamic properties of medications, risk of feeding tube obstruction, and potential for interactions between enteral nutrition and medications should be considered when clinicians administer medications through feeding tubes. The risk of feeding tube obstruction and clogging rises with increasing tube length and decreasing tube lumen. Incidence of obstructed percutaneous endoscopic gastrotomy tubes is reported to be 23% to 35%.1
A coordinated effort by all members of the health care team is essential to provide safe and effective care to patients with dysphagia and/or enteral feeding tubes. To decrease the risk of feeding tube obstruction, medications should be dissolved in water or administered in liquid form, saline fluids should be avoided, and the tube should be flushed with water before and after administering medications.
The pharmacokinetics of medications can be altered when tablets are crushed or capsules are opened. The bioavailability of dabigatran, for example, increases by 75% when the capsules are opened and pellets are taken orally.2 Medications may become intolerable after manipulation due to taste.3 Others may also increase the risk of feeding tube obstruction, such as omeprazole granules that increase the risk of small-bore feeding tube obstruction.4
Prior assessments of drug administration for patients with dysphagia and/or enteral feeding tubes has shown medication errors are prevalent.5-7 The Institute for Safe Medication Practices (ISMP) issued a Medication Safety Alert that provides a framework for preventing medication errors when preparing and administering medications via enteral feeding tubes.8 Other resources, such as monographs, are also available to guide pharmacotherapy decisions when oral medications require manipulation for administration to patients with dysphagia and/or enteral feeding tubes.9-11
In 2021, the Kansas City Veterans Affairs Medical Center (KCVAMC) was recognized as a Veterans Health Administration (VHA) Shark Tank finalist for improving the safety of medication administration for patients with enteral feeding tubes.12 This involved the addition of a Computerized Patient Record System (CPRS), clinical reminder order check (CROC), and a comprehensive medication review by a pharmacist. After implementing the CROC alert and pharmacy e-consultation workflow, the KCVAMC team reported that the number of inappropriate medications (ie, drugs on the ISMP do not crush list) was reduced from 41 to 6 in 1 year, resulting in an 85.4% reduction in potential medication errors.13
In 2014, the Richard L. Roudebush VAMC (RLRVAMC) created a pharmacy consultation process for patients with dysphagia and/or enteral feeding tubes. Any clinician could place a pharmacy consultation in CPRS. A pharmacist then reviewed patient charts, medication information resources, the VA formulary, and RLRVAMC pharmacy inventory. The pharmacist conferred with the patient’s care team to adjust pharmacotherapy, completed a consultation note, and updated medication order comments in Veterans Health Information Systems and Technology Architecture (VistA). These comments interfaced with the barcode medication administration software for the health care professional administering medications.
Despite the 2014 quality improvement (QI) process, medication errors involving the inappropriate ordering, preparation, and administration of medications for patients with dysphagia and/or enteral feeding tubes continued to be reported. Additionally, anonymous feedback revealed that only 3 of 10 responding pharmacists were satisfied with the existing medication use process for patients with dysphagia and/or enteral feeding tubes. Pharmacists expressed concerns that (1) clinicians were inappropriately crushing and/or manipulating new medications that were ordered after pharmacy consultations; (2) there was a lack of comprehensive documentation in CPRS; and (3) there were too many manual steps in the process. In response, RLRVAMC initiated a new QI initiative to improve the medication use process for patients with dysphagia and/or enteral feeding tubes in the acute care setting.
Quality Improvement Project
This multidisciplinary RLRVAMC QI project began November 2024 to improve pharmacotherapy care for patients with dysphagia and/or enteral feeding tubes in acute care. It was approved by the RLRVAMC Pharmacy Service. This intervention addressed the pharmacy consultation template, standardization of equipment, standardization of language, creation of clinical alerts, and sustainment (Table 1).

RLRVAMC has about 8600 annual inpatient admissions and 159 acute care beds.14 The project charter was drafted, and local stakeholders were identified including pharmacy technicians, pharmacists, nurses, speech language pathologists, and acute care clinicians. Pharmacy consultation workload was retrospectively reviewed to describe the scope of the existing state.
A workshop with 12 QI project stakeholders in December 2024 used A3 methodology to define the current process and the target state, barriers and solutions, prioritize interventions on an impact-effort matrix, perform a gap analysis, identify rapid plan-do-study-act (PDSA) experiments, and develop a completion plan (Figure). Five postworkshop PDSA experiments engaged additional stakeholders, clinical application coordinators, and medical supply representatives to ascertain the feasibility of the tools implemented.

Abbreviations: BCMA, barcode medication administration; CDSS, clinical decision support system; CPRS, Computerized Patient Record System;
EHR, electronic health record; SOP, standard operating procedure; VistA, Veterans Health Information Systems and Technology Architecture.
About 3% of RLRVAMC admissions involve a pharmacy consultation to review medications for dysphagia and/or enteral feeding tubes. Clinicians reviewed 30 preimplementation inpatient pharmacy consultations involving 200 oral medications. Pharmacists were more frequently consulted for inpatients with dysphagia (19 [63%]) than for patients with enteral feeding tubes (11 [37%]) (Table 2).

Pharmacy Consultation Template
The pharmacy consultation was updated in CPRS. Prior to this QI project, the ordering clinician was prompted to select 1 option for the indication: dysphagia or enteral feeding tube. The type of enteral feeding tube was not prompted by the consultation text nor required to be specified in the consultation. The ordering clinician could provide free-text comments. Of 11 preimplementation consultations, the type of enteral feeding tube was specified in 5 (45%). The consultation template entry was updated to include an option to check a box for the consultation indication from 3 options: dysphagia, enteral feeding, or other patient- specific condition/request. If enteral feeding tube is selected, then the clinician is prompted to select the type of enteral feeding tube. Since the completion of the project, there have been no patient safety reports concerning an erroneous or incomplete consultation entry (Supplemental Material).
The note template was updated to import the list of active inpatient medications and provide sections for the adjudicating pharmacist to document which medications can be crushed (or opened), which require adjustment, and which are hazardous and require special handling. Additionally, the revised template added a statement clarifying that the documented recommendations apply only to the medication regimen at the time of the consultation (Supplemental Material).
Standardizations
There are multiple pill-crushing devices used at RLRVAMC that vary in crushing mechanism, corresponding medication pouches, and degree of protection when manipulating hazardous medications. Prior to this QI project, RLRVAMC used 3 pill-crushing devices (about 30 total devices in inpatient care areas). Only 1 device with corresponding closed pouches for preparation of hazardous medications was available, which was stored in the RLRVAMC inpatient pharmacy. This workflow resulted in waste and posed potential risks for delays in care. This project incorporated a standard pill-crushing system with the corresponding medication pouches in all inpatient care areas, which provided safeguards for clinicians to prepare and administer hazardous medications (Supplemental Material).
Patients requiring medications to be crushed or opened on discharge should receive education, written instruction, and have care plans documented in CPRS. RLRVAMC patients receive education and a printed medication list. Prior to this QI project, the instructions for crushing or opening medications could only be entered by free text in the electronic medication reconciliation tool, allowing for the potential for inconsistent language or omissions.
This QI project included an update to the electronic medication reconciliation tool. An optional checkbox selection was added for patients requiring medications to be manipulated. When checked, a radial selection for individual medications is displayed, prompting the clinician and pharmacist to indicate either do not crush tablet or OK to crush tablet. These selections appear in clinical care notes and on the printed medication list provided to the patient (Supplemental Material).
Clinical Alerts
As part of the RLRVAMC QI initiative, a CROC alert was implemented, based on the KCVAMC intervention for patients with enteral feeding tubes.13 The RLRVAMC CROC alert also included patients with dysphagia. A nursing text order was made available in CPRS for patients requiring medications and remains active throughout the duration of the patient’s admission or until discontinued. It generates CROC alerts in CPRS and VistA when new medication orders are entered and reviewed by pharmacists.
Clinicians used clinical decision support systems to create daily lists of patients receiving medications by feeding tube and patients receiving crushed/opened medications due to dysphagia. This allows pharmacists to perform a census review of all inpatients to confirm appropriateness of medication orders. Clinical alerts for patients with enteral feeding tubes are advised by the ISMP and have data demonstrating a reduction in medication errors (Supplemental Material).14,15
Sustainment
During the sustainment phase, process owners were identified and a Pharmacy Service standard operating procedure (SOP) was written. The development of an institutional do not crush medication list was discussed; however, it was determined to be difficult to develop and maintain. An institutional tertiary resource list was selected in favor of a locally developed resource. These resources include the Handbook of Drug Administration via Enteral Feeding Tubes, Third Edition, the Pharmacist’s Letter list, “Meds that Should Not be Crushed,” and the Up- ToDate Lexidrug list, “Oral Medications That Should Not Be Crushed or Altered.”9-11 Links to the resources were added to the RLRVAMC pharmacy service SharePoint. In addition to defining the preferred tertiary resources, the SOP defined the process for reviewing inventory and the process for reviewing medication orders for hazard risk.
Discussion
Continued patient safety reports and low satisfaction rates among pharmacists prompted this QI project to improve safety for patients with dysphagia and/or enteral feeding tubes at RLRVAMC. The project engaged stakeholders and also identified and addressed gaps with potential for patient harm.
The tools implemented by this initiative drew from previous work by the KCVAMC and from framework provided by the ISMP.8,13 We expanded the QI intervention to include acute care patients with dysphagia.
RLRVAMC did not take steps to track the impact of the interventions on medication errors. However, no patient safety reports concerning an erroneous or incomplete pharmacy consultation entry have been reported. We also think that it is reasonable to assume that the adoption of the safety tools described here will have a positive impact on patient safety. RLRVAMC pharmacists have noted an increased appreciation for medication safety when processing medication orders for patients with dysphagia and/or enteral feeding tubes. While the workflow took time to adopt and integrate, clinical pharmacists perceived it as an improvement in patient safety. Our future focus is aimed at translating the process improvement into the Oracle/Cerner electronic health record, which is scheduled to be deployed at the RLRVAMC in August 2026.
Limitations
This QI project did not aim to quantify or compare medication errors before and after the intervention. An accurate number of unreported errors in the medication use process for patients with dysphagia and/or enteral feeding tubes would be challenging to quantify without direct observation. Multiple clinicians are engaged in the medication use process and individual steps may not be documented at all, or documented properly. In addition, medication errors are often underreported and may not reflect the total number of errors and/or potential for errors. That said, reported medication errors in the medication use process for patients with dysphagia and/or enteral feeding tubes are reviewed on a monthly basis by the RLRVAMC Multidisciplinary Medication Safety committee to continuously improve patient safety.
Another potential limitation is the extent to which the project can be adapted at other VHA sites. For example, RLRVAMC uses CPRS; the framework and tools to improve medication safety may not translate to sites using the Oracle/Cerner electronic health record. Furthermore, this QI project included a pharmacy consultation workflow that relied on pharmacists who are available at any hour. Other facilities may not have continuous consultation coverage to review medications for patients with dysphagia and/or enteral feeding tubes.
Conclusions
This QI project drew from ISMP recommendations, previous work within the VHA, local practice, and insight from multiple disciplines on the health care team to revise and create tools to improve medication safety for patients with dysphagia and/or enteral feeding tubes in the acute care setting. These tools included a revised pharmacy consultation workflow with improvements to the pharmacy consultation template, standardization of the pill-crushing devices and language used for patient medication lists, implementation of CROC alerts within the EHR, and development of an SOP.
The RLRVAMC Pharmacy Service intends to continue reviewing patient safety reports, assessing staff perspectives, and refining (and potentially adding) tools for medication safety. Future QI initiatives may focus on improving medication safety for outpatients with dysphagia and/or enteral feeding tubes. We also hope that these tools can be adapted at other VAMCs to promote medication safety for patients with dysphagia and/or enteral feeding tubes.
- Blumenstein I, Shastri YM, Stein J. Gastroenteric tube feeding: techniques, problems and solutions. World J Gastroenterol. 2014;20:8505-8524. doi:10.3748/wjg.v20.i26.8505
- Pradaxa (dabigatran etexilate). Prescribing information. Boehringer Ingelheim Pharmaceuticals, Inc; 2025. https:// pro.boehringer-ingelheim.com/us/products/pradaxa/bipdf /pradaxa-capsules-us-pi
- Lovell AG, Protus BM, Dickman JR, et al. Palatability of crushed over-the-counter medications. J Pain Symptom Manage. 2021;61:755-762. doi:10.1016/j.jpainsymman.2020.09.020
- Messaouik D, Sautou-Miranda V, Bagel-Boithias S, et al. Comparative study and optimisation of the administration mode of three proton pump inhibitors by nasogastric tube. Int J Pharm. 2005;299:65-72. doi:10.1016/j.ijpharm.2005.04.034
- Demirkan K, Bayraktar-Ekincioglu A, Gulhan-Halil M, et al. Assessment of drug administration via feeding tube and the knowledge of health-care professionals in a university hospital. Eur J Clin Nutr. 2017;71:164-168. doi:10.1038/ejcn.2016.147
- Fodil M, Nghiem D, Colas M, et al. Assessment of clinical practices for crushing medication in geriatric units. J Nutr Health Aging. 2017;21:904-908. doi:10.1007/s12603-017-0886-3
- Zhu LL, Xu LC, Wang HQ, et al. Appropriateness of administration of nasogastric medication and preliminary intervention. Ther Clin Risk Manag. 2012;8:393-401. doi:10.2147/TCRM.S37785
- Institute for Safe Medication Practices (ISMP). Preventing errors when preparing and administering medications via enteral feeding tubes. Acute Care ISMP Medication Safety Alert. November 17, 2022. Accessed March 17, 2026. https://nutritioncare.org/wp-content/uploads/2025/02 /ISMP-Safety-Alert_Medications-and-Enteral-Feeding -Tubes.pdf
- White R, Bradnam V. Handbook of Drug Administration via Enteral Feeding Tubes. 3rd ed. Pharmaceutical Press; 2015.
- Clinical resource, meds that should not be crushed. Pharmacist’s Letter/Pharmacy Technician’s Letter/Prescriber Insights. Updated April 2025. Accessed March 17, 2026. https://pharmacist.therapeuticresearch.com/en/Content /Segments/PRL/2014/Aug/Meds-That-Should-Not-Be -Crushed-7309
- Oral medications that should not be crushed or altered. In: Lexidrug. UpToDate, Inc. https://online.lexi.com/lco /action/doc/retrieve/docid/patch_f/4227
- Uttaro E, Zhao F, Schweighardt A. Filling the gaps on the Institute for Safe Medication Practices (ISMP) do not crush list for immediate-release products. Int J Pharm Compd. 2021;25:364-371.
- US Dept of Veterans Affairs. VA Diffusion Marketplace. Improved safety of enteral tube medication administration. Updated 2024. Accessed March 17, 2026. https:// marketplace.va.gov/innovations/improved-safety-of -enteral-tube-medication-administration
- US Dept of Veterans Affairs. About us. VA Indiana Healthcare System. Updated October 17, 2024. Accessed March 2, 2026. https://www.va.gov/indiana-health-care/about-us/
- Wasylewicz ATM, van Grinsven RJB, Bikker JMW, et al. Clinical decision support system-assisted pharmacy intervention reduces feeding tube-related medication errors in hospitalized patients: a focus on medication suitable for feeding-tube administration. JPEN J Parenter Enteral Nutr. 2021;45:625-632. doi:10.1002/jpen.1869
- Blumenstein I, Shastri YM, Stein J. Gastroenteric tube feeding: techniques, problems and solutions. World J Gastroenterol. 2014;20:8505-8524. doi:10.3748/wjg.v20.i26.8505
- Pradaxa (dabigatran etexilate). Prescribing information. Boehringer Ingelheim Pharmaceuticals, Inc; 2025. https:// pro.boehringer-ingelheim.com/us/products/pradaxa/bipdf /pradaxa-capsules-us-pi
- Lovell AG, Protus BM, Dickman JR, et al. Palatability of crushed over-the-counter medications. J Pain Symptom Manage. 2021;61:755-762. doi:10.1016/j.jpainsymman.2020.09.020
- Messaouik D, Sautou-Miranda V, Bagel-Boithias S, et al. Comparative study and optimisation of the administration mode of three proton pump inhibitors by nasogastric tube. Int J Pharm. 2005;299:65-72. doi:10.1016/j.ijpharm.2005.04.034
- Demirkan K, Bayraktar-Ekincioglu A, Gulhan-Halil M, et al. Assessment of drug administration via feeding tube and the knowledge of health-care professionals in a university hospital. Eur J Clin Nutr. 2017;71:164-168. doi:10.1038/ejcn.2016.147
- Fodil M, Nghiem D, Colas M, et al. Assessment of clinical practices for crushing medication in geriatric units. J Nutr Health Aging. 2017;21:904-908. doi:10.1007/s12603-017-0886-3
- Zhu LL, Xu LC, Wang HQ, et al. Appropriateness of administration of nasogastric medication and preliminary intervention. Ther Clin Risk Manag. 2012;8:393-401. doi:10.2147/TCRM.S37785
- Institute for Safe Medication Practices (ISMP). Preventing errors when preparing and administering medications via enteral feeding tubes. Acute Care ISMP Medication Safety Alert. November 17, 2022. Accessed March 17, 2026. https://nutritioncare.org/wp-content/uploads/2025/02 /ISMP-Safety-Alert_Medications-and-Enteral-Feeding -Tubes.pdf
- White R, Bradnam V. Handbook of Drug Administration via Enteral Feeding Tubes. 3rd ed. Pharmaceutical Press; 2015.
- Clinical resource, meds that should not be crushed. Pharmacist’s Letter/Pharmacy Technician’s Letter/Prescriber Insights. Updated April 2025. Accessed March 17, 2026. https://pharmacist.therapeuticresearch.com/en/Content /Segments/PRL/2014/Aug/Meds-That-Should-Not-Be -Crushed-7309
- Oral medications that should not be crushed or altered. In: Lexidrug. UpToDate, Inc. https://online.lexi.com/lco /action/doc/retrieve/docid/patch_f/4227
- Uttaro E, Zhao F, Schweighardt A. Filling the gaps on the Institute for Safe Medication Practices (ISMP) do not crush list for immediate-release products. Int J Pharm Compd. 2021;25:364-371.
- US Dept of Veterans Affairs. VA Diffusion Marketplace. Improved safety of enteral tube medication administration. Updated 2024. Accessed March 17, 2026. https:// marketplace.va.gov/innovations/improved-safety-of -enteral-tube-medication-administration
- US Dept of Veterans Affairs. About us. VA Indiana Healthcare System. Updated October 17, 2024. Accessed March 2, 2026. https://www.va.gov/indiana-health-care/about-us/
- Wasylewicz ATM, van Grinsven RJB, Bikker JMW, et al. Clinical decision support system-assisted pharmacy intervention reduces feeding tube-related medication errors in hospitalized patients: a focus on medication suitable for feeding-tube administration. JPEN J Parenter Enteral Nutr. 2021;45:625-632. doi:10.1002/jpen.1869
Streamlining the Acute Care Pharmacy Consultation Process for Patients With Dysphagia or Enteral Feeding Tubes
Streamlining the Acute Care Pharmacy Consultation Process for Patients With Dysphagia or Enteral Feeding Tubes
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).
Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
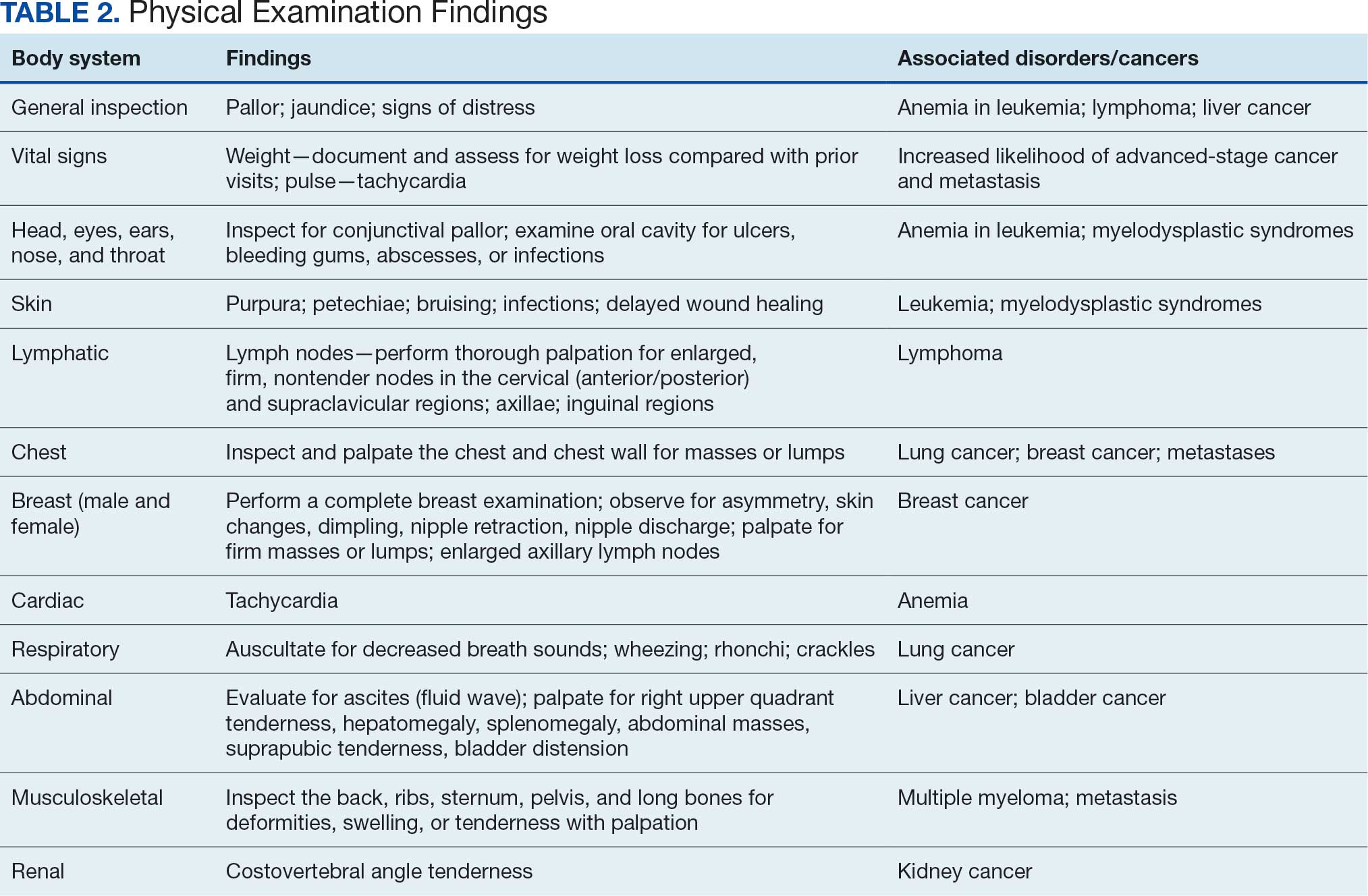
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.
Proposed Diagnostic Screening Panel
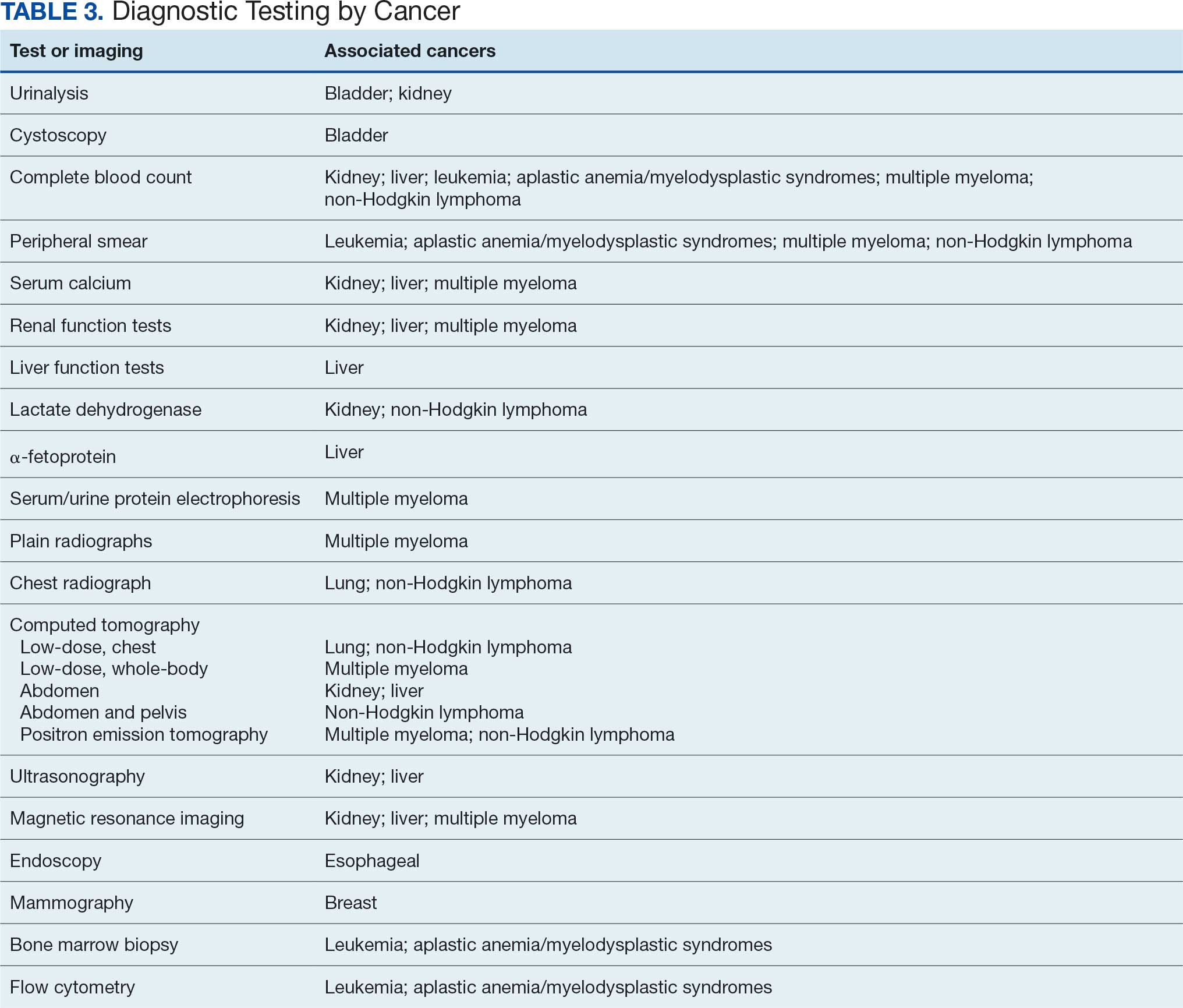
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).
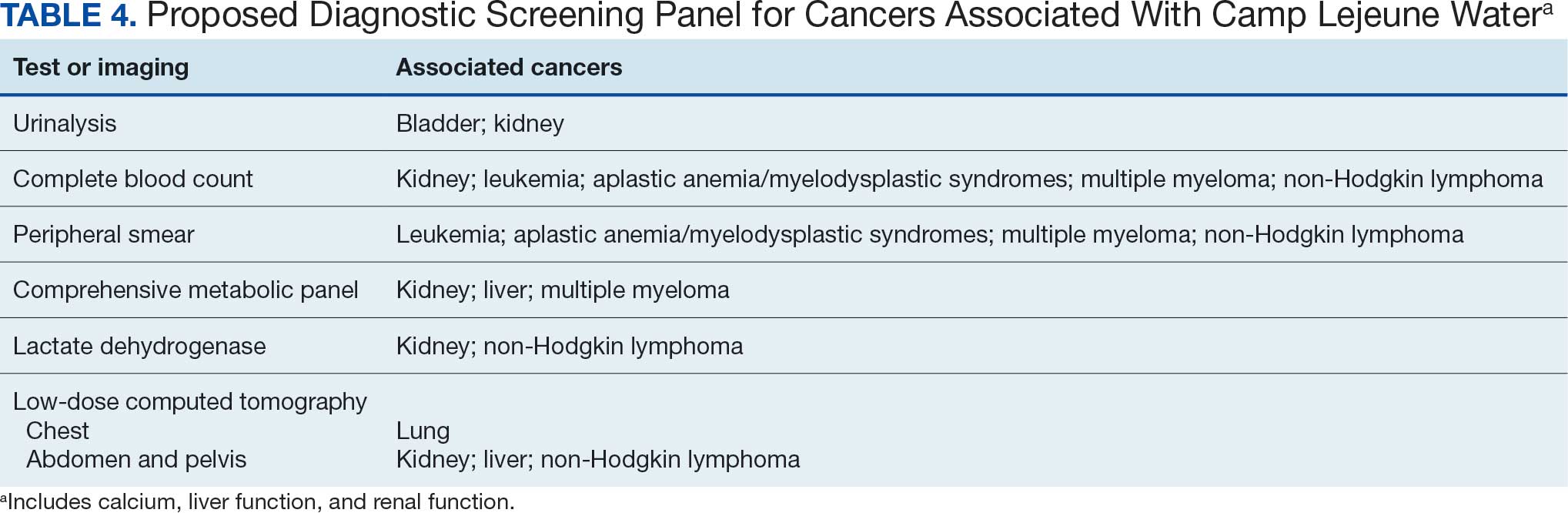
A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).
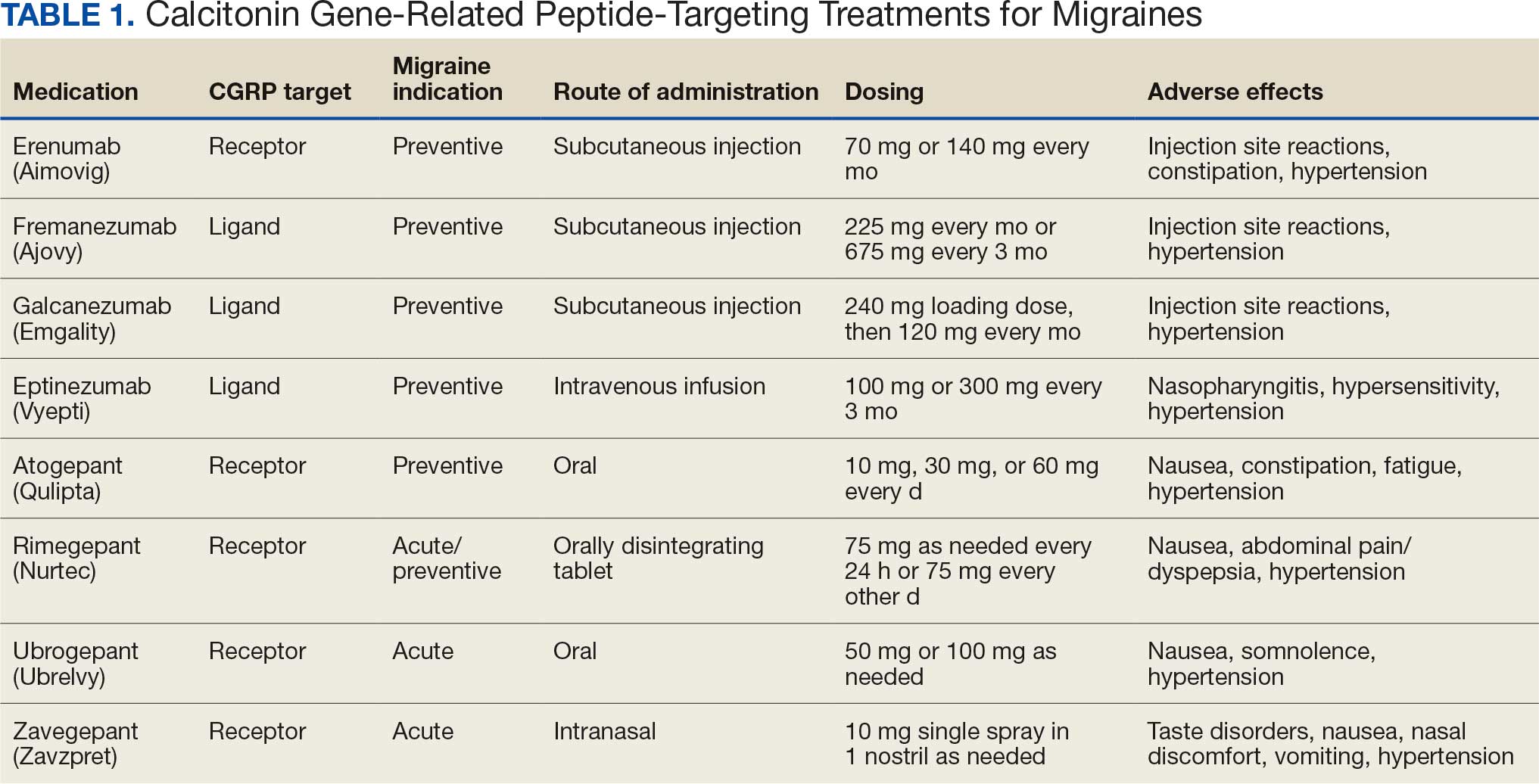
CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).
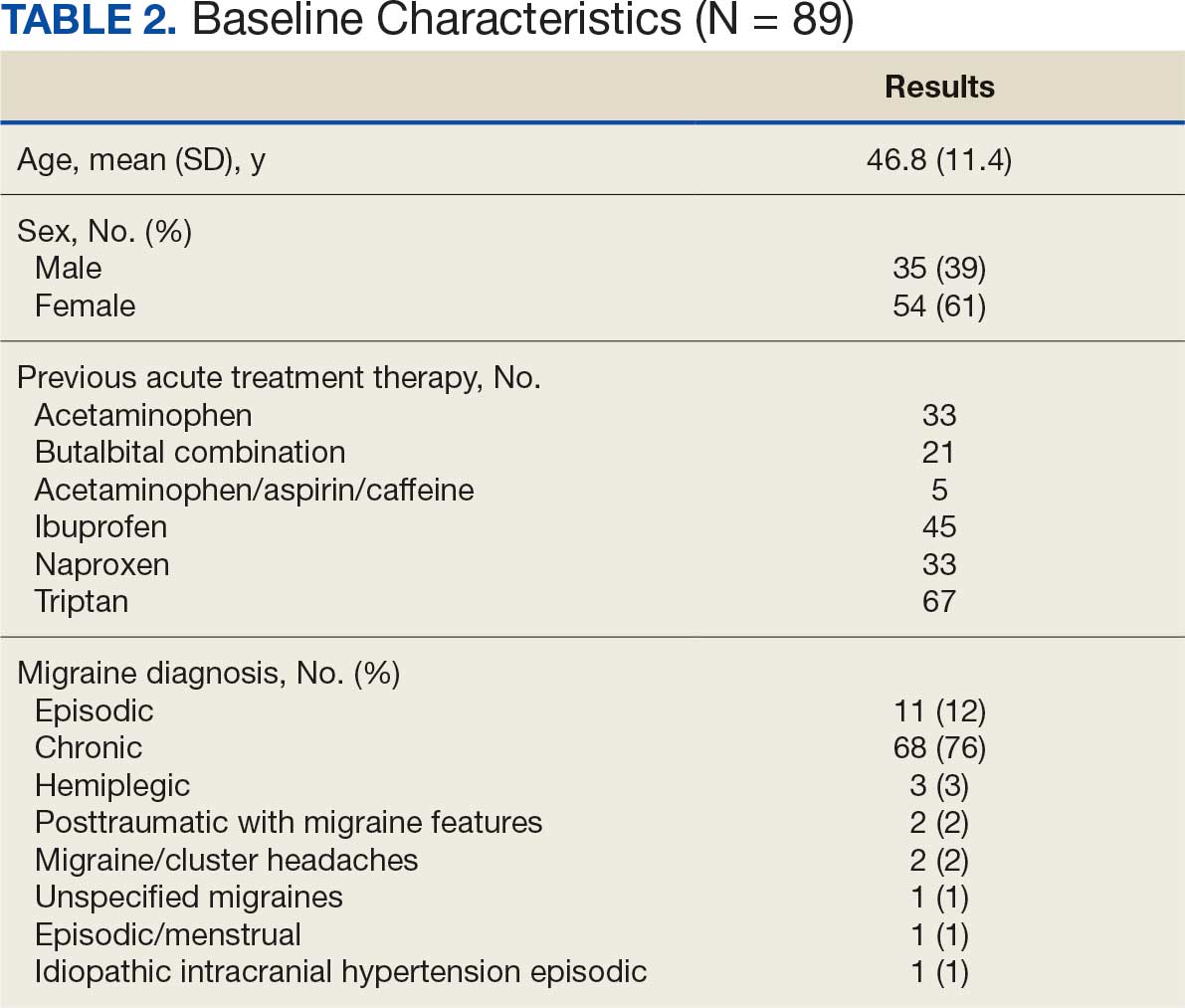
Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.
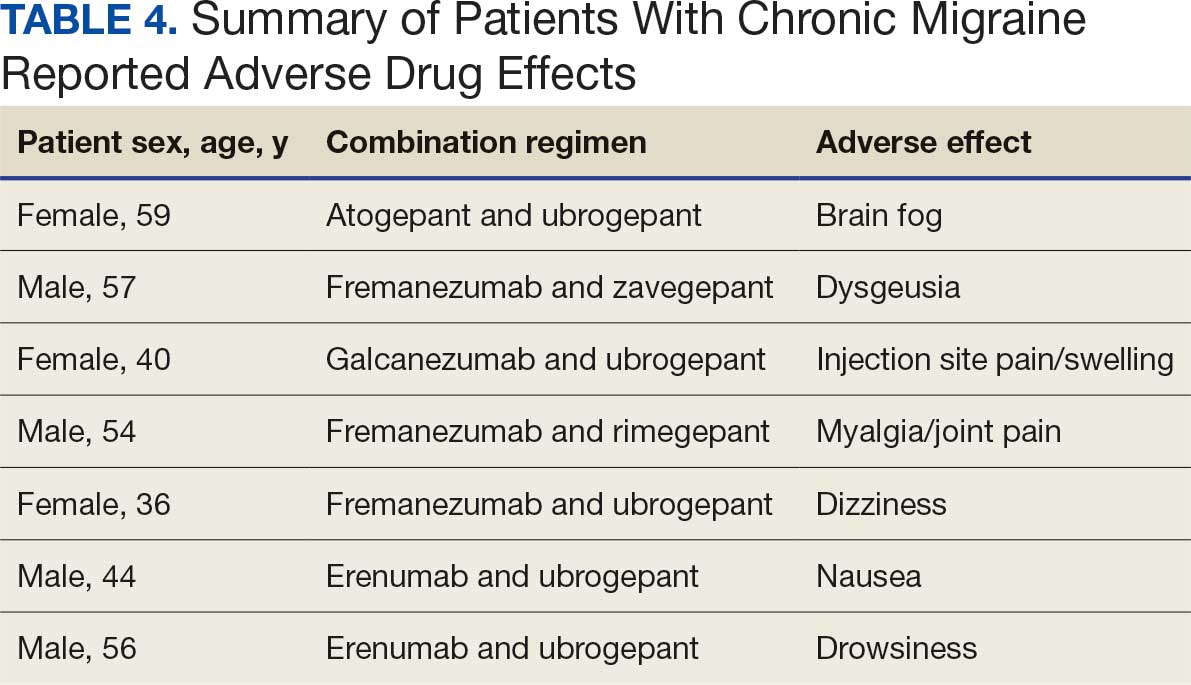
Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).
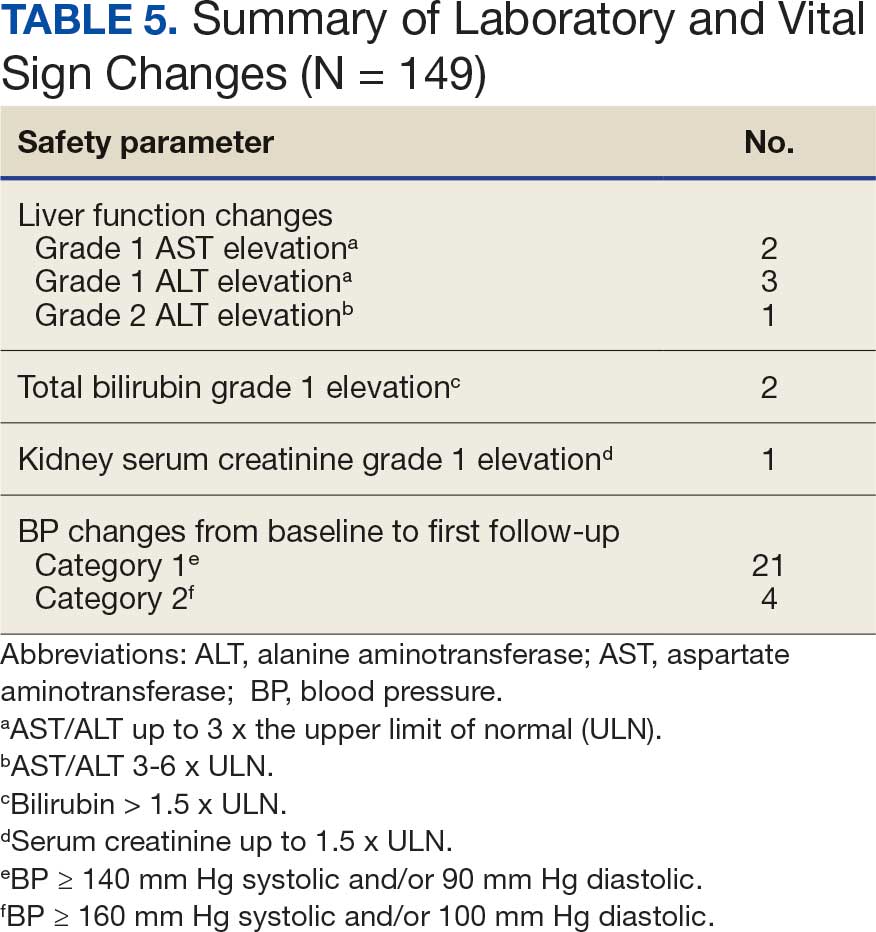
Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
Heart Failure Diagnostic Alerts to Prompt Pharmacist Evaluation and Medication Optimization
Heart Failure Diagnostic Alerts to Prompt Pharmacist Evaluation and Medication Optimization
Heart failure (HF) is a prevalent disease in the United States affecting > 6.5 million adults and contributing to significant morbidity and mortality.1 The disease course associated with HF includes potential symptom improvement with intermittent periods of decompensation and possible clinical deterioration. Multiple therapies have been developed to improve outcomes in people with HF—to palliate HF symptoms, prevent hospitalizations, and reduce mortality.2 However, the risks of decompensation and hospitalization remain. HF decompensation development may precede clear actionable symptoms such as worsening dyspnea, noticeable edema, or weight gain. Tools to identify patient deterioration and trigger interventions to prevent HF admissions are clinically attractive compared with reliance on subjective factors alone.
Cardiac resynchronization therapy (CRT) and implantable cardioverter-defibrillator (ICD) devices made by Boston Scientific include the HeartLogic monitoring feature. Five main sensors produce an index risk score; an index score > 16 warns clinicians that the patient is at an increased risk for a HF event.3 The 5 sensors are thoracic impedance, first (S1) and third heart sounds (S3), night heart rate (NHR), respiratory rate (RR), and activity. Each sensor can draw attention to the primary driver of the alert and guide health care practitioners (HCPs) to the appropriate interventions.3 A HeartLogic alert example is shown in Figure 1.
The S3 occurs during the early diastolic phase when blood moves into the ventricles. As HF worsens, with a combination of elevated filling pressures and reduced cardiac muscle compliance, S3 can become more pronounced.4 The S1 is correlated with the contractility of the left ventricle and will be reduced in patients at risk for HF events.5 Physical activity is a long-term prognostic marker in patients with HF; reduced activity is associated with mortality and increased risk of an HF event.6 Thoracic impedance is a sensor used to identify pulmonary congestion, pocket infections, pleural/pericardial effusion, and respiratory infections. The accumulation of intrathoracic fluid during pulmonary congestion increases conductance, causing a decrease in impedance.7 RR will increase as patients experience dyspnea with a more rapid, shallow breath and may trigger alerts closer to the actual HF event than other sensors. Nearly 90% of patients hospitalized for HF experience shortness of breath.8,9 NHR is used as a surrogate for resting heart rate (HR). A high resting HR is correlated with the progression of coronary atherosclerosis, harmful effects on left ventricular function, and increased risk of myocardial ischemia and ventricular arrhythmias.10
One of the challenges with preventing hospitalizations may be the lack of patient reported symptoms leading up to the event. The purpose of the sensors and HeartLogic index is to identify patients a median of 34 days before an HF event (HF admission or unscheduled intervention with intravenous treatment) with a sensitivity rate of 70%.3 According to real-word experience data, alerts have been found to precede HF symptoms by a median of 12 days and HF events such as hospitalizations by a median of 38 days, with an overall 67% reduction in HF hospitalizations when integrated into clinical care.11,12
MANAGE-HF evaluated 191 patients with HF with reduced ejection fraction (HFrEF) (< 35%), New York Heart Association class II-III symptoms, and who had an implanted CRT and/or ICD to develop an alert management guide to optimize medical treatment.12 It aimed to adjust patient regimen within 6 days of an elevated Heart- Logic index by either initiation, escalation, or maintenance of HF treatment depending on the index trend after the initial alert. This trial found that by focusing on such optimization, HF treatment was augmented during 74% of the 585 alert cases and during 54% of 3290 weekly alerts.
Initiation and uptitration of the 4 primary components of guideline-directed medical therapy (GDMT) are recommended by the 2022 Heart Failure Guidelines to reduce mortality and morbidity in patients with HFrEF.2 The 4 pillars of GDMT consist of -blockers (BB), sodium-glucose cotransporter type 2 inhibitors (SGLT2i), mineralocorticoid receptor antagonists (MRA), and renin-angiotensin-system inhibitors (RASi) including angiotensin II receptor blocker/neprilysin inhibitors (ARNi), angiotensin-converting enzyme inhibitors (ACEi) and angiotensin II receptor blockers (ARB) (Appendix 1). Obtaining and titrating to target doses wherever possible is recommended, as those were the doses that established safety and efficacy in patients with HFrEF in clinical trials.2 Pharmacists are adequately equipped to optimize HF GDMT and appropriately monitor drug response.

Through the use of HeartLogic in clinical practice, patients with HF have been shown to have improved clinical outcomes and are more likely to receive effective care; 80% of alerts were shown to provide new information to clinicians.13 This project sought to quantify the total number and types of pharmacist interventions driven by integration of HeartLogic index monitoring into practice.
Methods
The West Palm Beach Veterans Affairs Medical Center (WPBVAMC) Research Program Office approved this project and determined it was exempt from institutional review board oversight. Patients were screened retrospectively and prospectively from May 26, 2022, through December 31, 2022, by a cardiology clinical pharmacist practitioner (CPP) and a cardiology pharmacy resident using the local monitoring suite for the HeartLogic-compatible device, LATITUDE NXT. Read-only access to the local monitoring suit was granted by the National Cardiac Device Surveillance Program. Training for HeartLogic was completed through continuing education courses provided by Boston Scientific. Additional information was provided by Boston Scientific representative presentations and collaboration with WPBVAMC pacemaker clinic HCPs.
Individuals included were patients with HeartLogic-capable ICDs. A HeartLogic alert had to be present at initial patient contact. Patients were also contacted as part of routine clinical practice, but no formal number or frequency of calls to patients was required. The initial contact must be with a pharmacist for the patient to be included, but subsequent contact by other HCPs was included. Patients in the cardiology clinic are required to meet with a cardiologist at least annually; however, interim visits can be completed by advanced practice registered nurse practitioners, physicians assistants, or CPPs.
Patients in alert status were contacted by telephone and appropriate modifications of HF therapy were made by the CPP based on score metrics, medical record review, and patient interview. Information surrounding the initial alert, baseline patient data, medication and monitoring interventions made, and clinical outcomes such as hospitalization, symptom improvement, follow-up, and mortality were collected. Information for each encounter was collected until 42 days from the initial date of pharmacist contact.
Clinically successful tolerability of intervention implementation was defined as tolerability, adherence, and lack of adverse effects (AEs) per patient report at follow-up or within 42 days from initial alert (Appendix 2). A decrease in dose was not counted as intolerance. A single patient may have been counted as multiple encounters if the original intervention resulted in treatment intolerance and the patient remained in alert or if an additional alert occurred after 42 days of the initial alert. There were no specific time criteria for follow-up, which occurred at the CPP’s discretion.

There was no mandated algorithm used to alter medications based on the Heart- Logic score, nor were there required minimum or maximum numbers of interventions after an alert. Patient contact by telephone initiated an encounter. The types of interventions included medication increases, decreases, initiation, discontinuation, or no medication change. Each medication change and rationale, if applicable, was recorded for the encounter ≤ 42 days after the initial contact date. If a medication with required monitoring parameters was augmented, the pharmacist was responsible for ordering laboratory testing and follow-up. Most interventions were completed by telephone; however, some patients had in-person visits in the HF CPP clinic.
Outcomes
The primary outcome was the number of pharmacist interventions made to optimize GDMT, defined as either an initiation or dose increase. Key intervention analysis included the use and dosing of the 4 primary components of HF GDMT: BB, SGLT2i, MRA, and ARNi/ARB/ACEi. In addition to the 4 primary components of GDMT, loop diuretic changes were also recorded and analyzed. Secondary endpoints were the number of HF hospitalizations ≤ 42 days after the initial alert, and the effect of medication interventions on device metrics, patient symptoms, and tolerability. Successful tolerability was defined as continued use of augmented GDMT without intolerance or discontinuation. The primary analysis was analyzed through descriptive statistics. Median changes in HeartLogic scores and metrics from baseline were analyzed using a paired, 2-sided t test with an α of .05 to detect significance.
Results
There were 39 WPBVAMC patients with a HeartLogic-capable device. Twenty-one alert encounters were analyzed in 16 patients (41%) over 31 weeks of data collection. The 16 patients at baseline had a mean age of 74 years, all were male, and 12 (75%) were White. Eight patients (50%) had a recent ejection fraction (EF) between 30% and 40%. Three patients had an EF ≥ 40%. At the time of alert, 15 patients used BB (94%), 10 used loop diuretics (63%), and 9 used ARNi (56%) (Table 1).
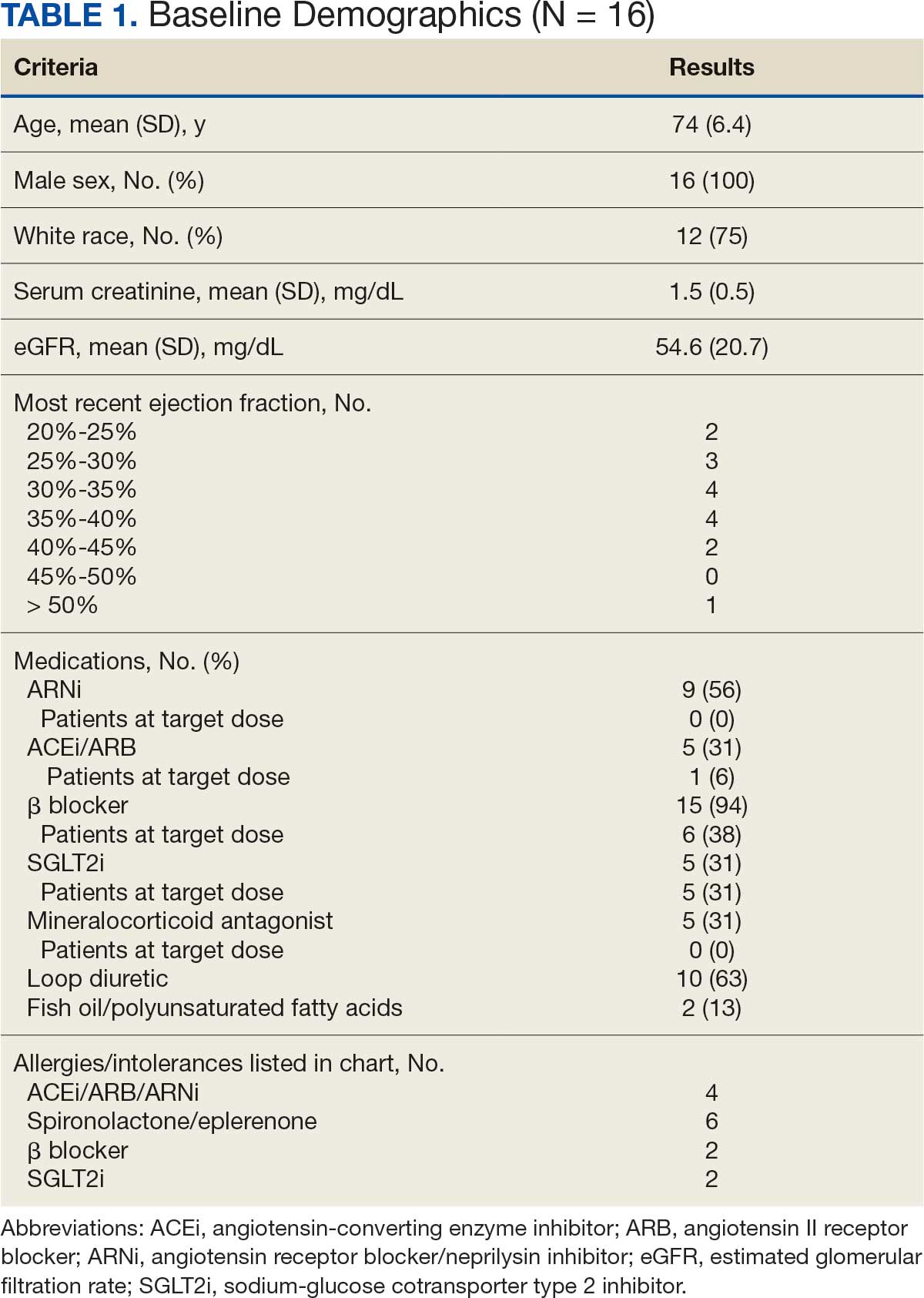
There were 23 medication changes made during initial contact. The most common change was starting an SGLT2i (30%; n = 7), followed by starting an MRA (22%; n = 5), and increasing the ARNi dose (22%; n = 5). At the initial contact, ≥ 1 medication optimization occurred in 95% (n = 20) of encounters. The CPP contacted patients a mean of 4.8 days after the initial alert.
Patients were taking a mean of 2.6 primary GDMT medications at baseline and 3.0 at 42 days. CPP encounters led to a mean of 1.8 medication changes over the 6-week period (range, 0-5). Seventeen medications were started, 13 medications were increased, 3 medications were decreased, and 4 medications were stopped (Table 2). One ACEi and 1 ARB were switched as a therapy escalation to an ARNi. One patient was on 1 of 4 primary GDMTs at baseline, which increased to 4 GDMT agents at 42 days.
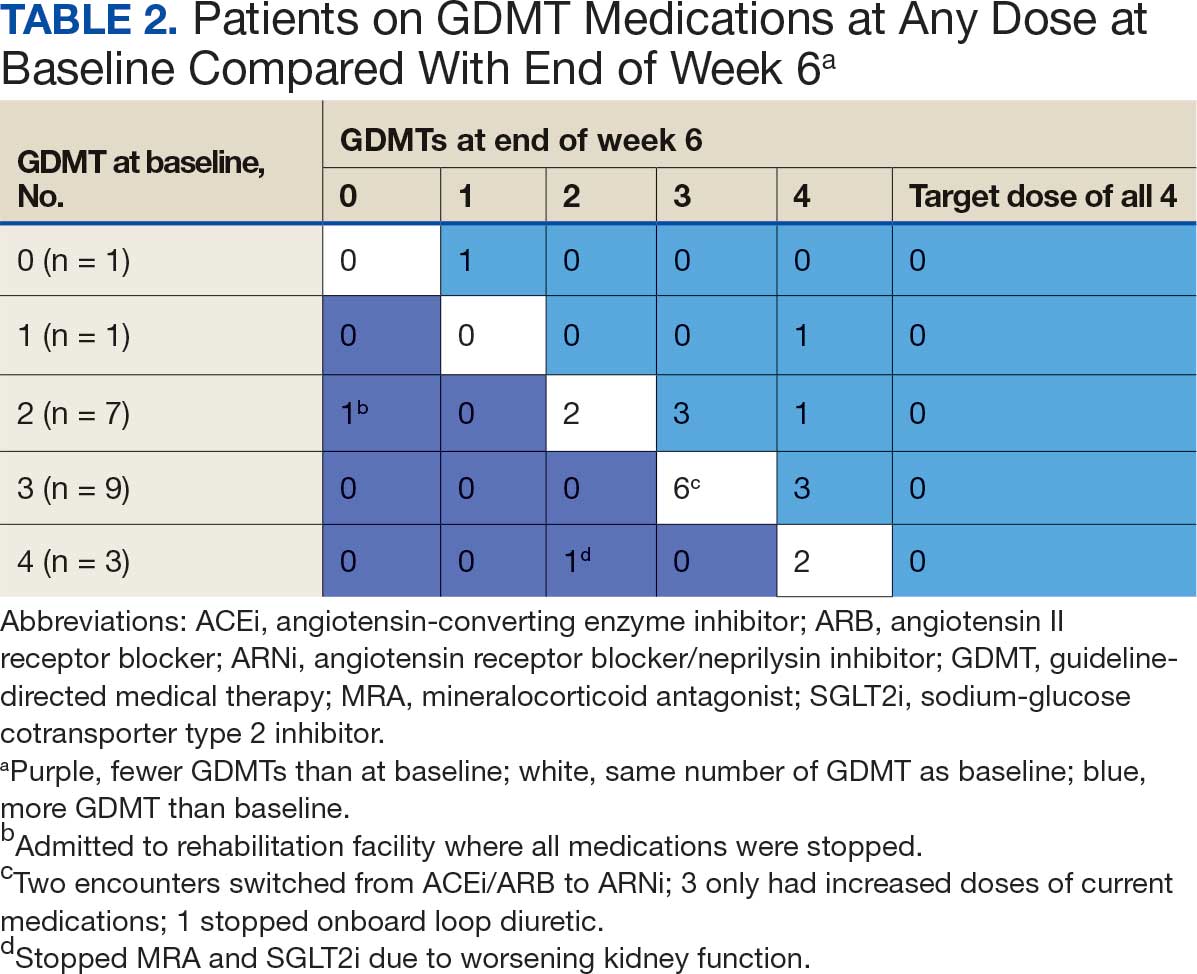
SGLT2 inhibitors were added most often at initial contact (54%) and throughout the 42-day period (41%). The most common successfully tolerated optimizations were RASi, followed by MRA, SGLT2 inhibitors, BB, and loop diuretics with 11, 6, 5, 3, and 2 patients, respectively. Interventions were tolerated by 90% of patients, and no HF hospitalization occurred during follow-up. All possible rationales for patients with the same or reduced number of GDMT at 42 days compared with baseline are shown in Appendix 2.
Device Metrics
During initial contact, the most common HeartLogic metric category that was predominantly worsening were heart sounds (S1, S3, and S3/S1 ratio), followed by compensatory mechanism sensors (NHR and RR) and congestion (impedance) at rates of 61.9%, 23.8%, and 14.3%, respectively (Figure 2).
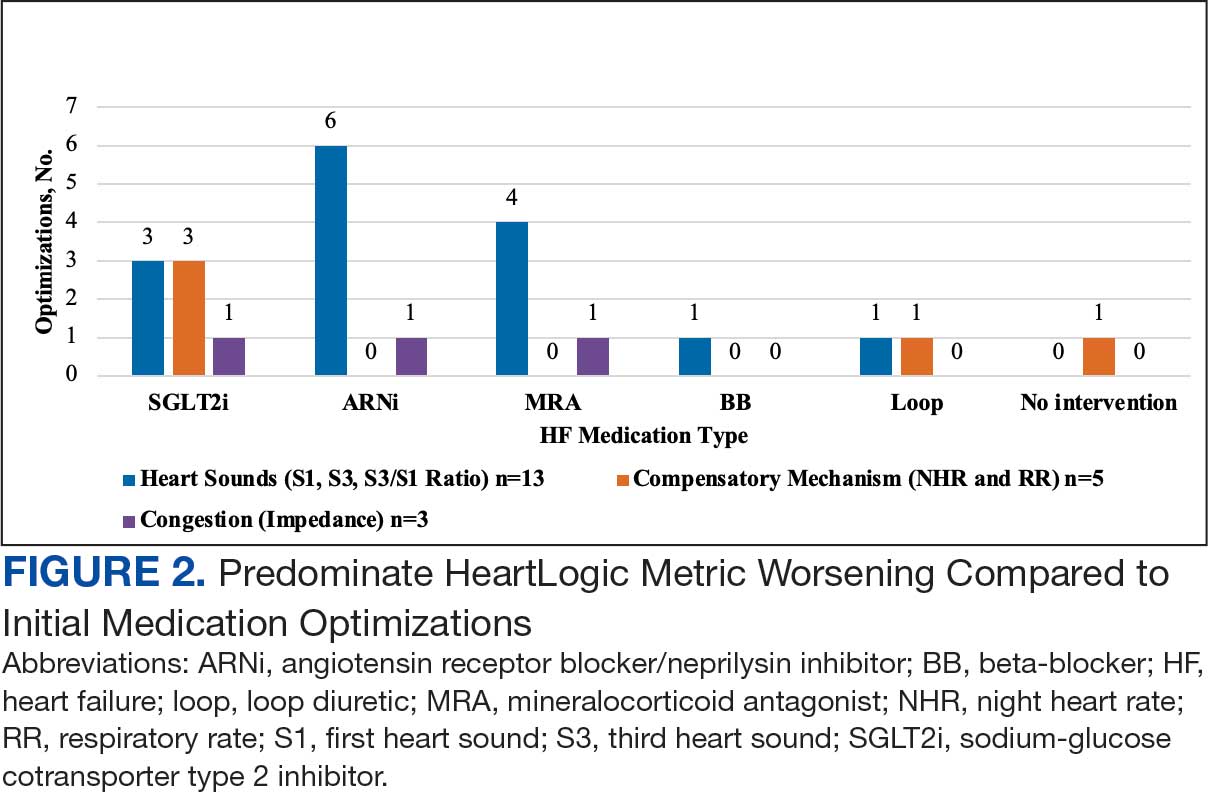
The median HeartLogic index score was 18 at baseline and 5 at the end of the follow-up period (P < .001). The changes in score and metrics were compared with the type of successfully tolerated GDMT optimization made (Table 3). The GDMT optimization analysis included SGLT2i, RASi, MRA, BB, and loop diuretics. All interventions reduced the overall HeartLogic index score, ranging from a 9.5-point reduction (loop diuretics) to a 16-point reduction (SGLT2i and BB). Optimization of SGLT2i, RASi, and loop diuretics had a positive impact on S1 score. For S3 score, SGLT2i, MRA, and BB had a positive impact. All medications, except for SGLT2i therapy, reduced the NHR score. Optimization of MRA, SGLT2i, and BB had positive impacts on the impedance score. All medications reduced RR from baseline. Only SGLT2i and loop diuretics had positive impacts on the activity score.
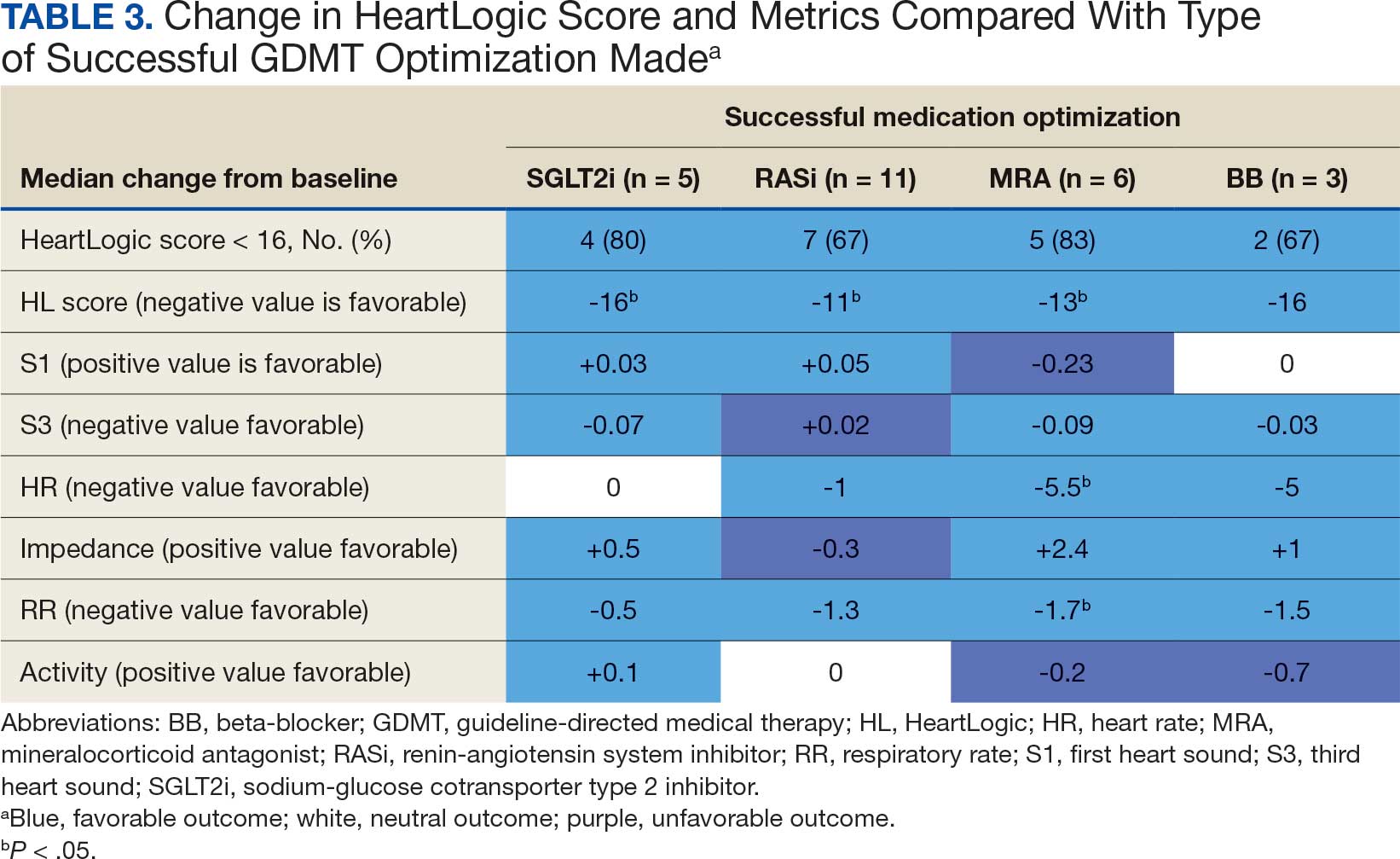
Clinical Outcomes and Adverse Effects
Within 42 days of contact, 17 encounters (81%) had ≥ 1 follow-up appointment with a CPP and all 21 patients had ≥ 1 follow-up health care team member. One patient had a HF-related hospitalization within 42 days of contact; however, that individual refused the recommended medication intervention. There were 13 encounters (62%) with reported symptoms at the time of the initial alert and 10 (77%) had subjective symptom improvement at 42 days (Appendix 3).
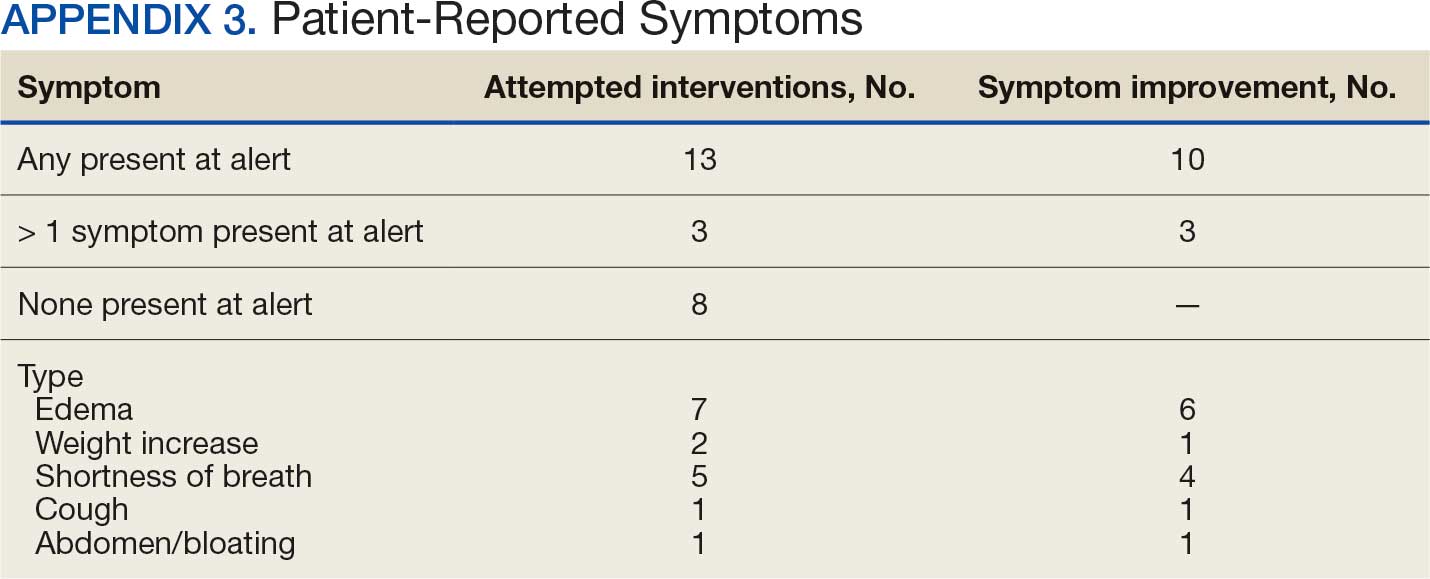
Of 30 medication optimizations, 27 primary GDMT medications were tolerated. Two medication intolerances led to discontinuation (1 SGLT2i and 1 loop diuretic) and 1 patient never started the SGLT2i (Table 4). There was only 1 known patient who did not follow the directions to adjust their medications. That individual was included because the patient agreed to the change during the CPP visit but later reported that he had never started the SGLT2i.
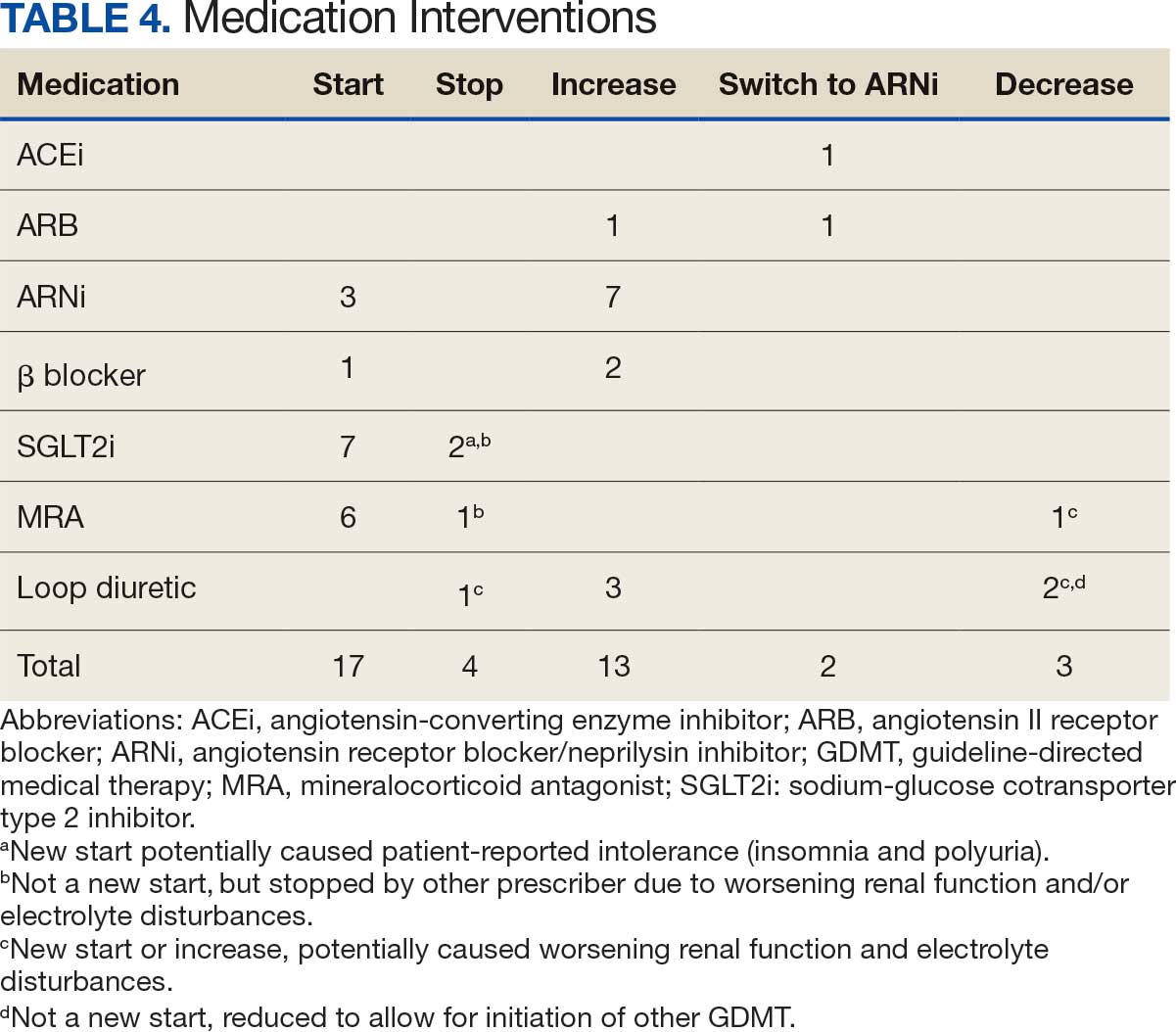
Discussion
The HeartLogic tool created a bridge for patients with HF to work with CPPs as soon as possible to optimize medication therapy to reduce HF events. This study highlights an additional area of expertise and service that CPPs may offer to their specialty HF clinic team. Over 31 weeks, 21 encounters and 30 medication optimizations were completed. These interventions led to significant reductions in HeartLogic scores, improvements in symptoms, and optimization of HF care, most of which were well tolerated.
Additional hemodynamic monitoring devices are available. Similar to HeartLogic, OptiVol is a tool embedded in select Medtronic implantable devices that monitors fluid status. 14 CardioMEMS is an implantable pulmonary artery pressure sensor used as a presymptomatic data point to alert clinicians when HF is worsening. In the CHAMPION trial, the use of CardioMEMS showed a 28% reduction in HF-related hospitalization at 6 months.15 Conversely, in the GUIDE-HF trial, monitoring with CardioMEMS did not significantly reduce the composite endpoint of mortality and total HF events.16 Therefore, remote hemodynamic monitoring has variable results and the use of these tools remains uncertain per the clinical guidelines.2
The MANAGE-HF study that contributed to the validation of the HeartLogic tool may provide a comparison with this smaller single-center project. The time to follow-up within 7 days of alert was noted in only 54% of the patients in MANAGE-HF.12 In this study, 86% of patients received follow-up within 7 days, with a mean of 4.8 days. The quick turnaround from the time of alert to intervention portrays pharmacists as readily available HCPs.
In MANAGE-HF, 89% of medication augmentation involved loop diuretics or thiazides; in our project, loop diuretics were the least frequently changed medication. Most optimizations in this project included ARNi, SGLT2i, BB, and MRA, which have been shown to reduce morbidity and mortality.2 Our project included use of SGLT2i therapy to affect HeartLogic metrics, which has not been evaluated previously. SGLT2i were the most commonly initiated medication after an alert. Of the 5 tolerated SGLT2i optimization encounters, 4 were out of alert at 42 days.
SGLT2i resulted in a significant decrease in HeartLogic index score from baseline and were the only class of medication that did not produce a negative change in any metric. In this study, CPPs utilizing and acting on HeartLogic alerts led to 1 (4.8%) hospitalization with HF as the primary reason for admission and no hospitalizations as a secondary cause in 42 days, compared to 37% and 7.9% in the MANAGE-HF in 1 year, respectively. An additional screening 1 year after the initial alert found that 2 (12.5%) of 12 patients had been admitted with 1 HF hospitalization each.
A strength of this study was the ability to use HeartLogic to identify high-risk patients, provide a source of patient contact and monitoring, interpret 5 cardiac sensors, and optimize all HF GDMT, not just volume management. By focusing efforts on making patient contact and pharmacotherapy interventions with morbidity and mortality benefit, remote hemodynamic monitoring may show a clear clinical benefit and become a vital part of HF care.
Limitations
Checking for adherence and tolerance to medications were mainly patient reported if there was a CPP follow-up within 42 days, or potentially through refill history when unclear. However, this limitation is reflective of current practice where patients may have multiple clinicians working to optimize HF care and where there is reliance on patients in order to guide continued therapy. Although unable to explicitly show a reduction in HF events given lack of comparator group, the interventions made are associated with improved outcomes and thus would be expected to improve patient outcomes. Changes in vital signs were not tracked as part of this project, however the main rationale for changes made were to optimize GDMT therapy, not specifically to impact vital sign measures.
HeartLogic alerts prompted identification of high-risk patients with HF, pharmacist evaluation and outreach, patient-focused pharmacotherapy care, and beneficial patient outcomes. With only 2 cardiology CPPs checking alerts once weekly, future studies may be needed with larger samples to create algorithms and protocols to increase the clinical utility of this tool on a greater scale.
Conclusions
Cardiology CPP-led HF interventions triggered by HeartLogic alerts lead to effective patient identification, increased access to care, reductions in HeartLogic scores, improvements in symptoms, and optimization of HF care. This project demonstrates the practical utility of the HeartLogic suite in conjunction with CPP care to prioritize treatment for highrisk patients with HF in an efficient manner. The data highlight the potential value of the HeartLogic tool and a CPP in HF care to facilitate initiation and optimization of GDMT to ultimately improve the morbidity and mortality in patients with HF.
- Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2023 update: a report from the American Heart Association. Circulation. 2023;147:e93-e621. doi:10.1161/CIR.0000000000001123
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/ American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895-e1032. doi:10.1161/CIR.0000000000001063
- Boehmer JP, Hariharan R, Devecchi FG, et al. A multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. J Am Coll Cardiol HF. 2017;5:216-225. doi:10.1016/j.jchf.2016.12.011
- Cao M, Gardner RS, Hariharan R, et al. Ambulatory monitoring of heart sounds via an implanted device is superior to auscultation for prediction of heart failure events. J Card Fail. 2020;26:151-159. doi:10.1016/j.cardfail.2019.10.006
- Calò L, Capucci A, Santini L, et al. ICD-measured heart sounds and their correlation with echocardiographic indexes of systolic and diastolic function. J Interv Card Electrophysiol. 2020;58:95-101. doi:10.1007/s10840-019-00668
- Del Buono MG, Arena R, Borlaug BA, et al. Exercise intolerance in patients with heart failure: JACC state-of-the- art review. J Am Coll Cardiol. 2019;73:2209-2225. doi:10.1016/j.jacc.2019.01.072
- Yu CM, Wang L, Chau E, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112:841-848. doi:10.1161/CIRCULATIONAHA.104.492207
- Rials S, Aktas M, An Q, et al. Continuous respiratory rate is superior to routine outpatient dyspnea assessment for predicting heart failure events. J Card Fail. 2018;24:S45.
- Fonarow GC, ADHERE Scientific Advisory Committee. The Acute Decompensated Heart Failure National Registry (ADHERE): opportunities to improve care of patients hospitalized with acute decompensated heart failure. Rev Cardiovasc Med. 2003;4(suppl 7):S21-S30. doi:10.1016/j.cardfail.2018.07.130
- Fox K, Borer JS, Camm AJ, et al. Resting heart rate in cardiovascular disease. J Am Coll Cardiol. 2007;50:823-830. doi:10.1016/j.jacc.2007.04.079
- De Ruvo E, Capucci A, Ammirati F, et al. Preliminary experience of remote management of heart failure patients with a multisensor ICD alert [abstract P1536]. Eur J Heart Fail. 2019;21(suppl S1):370.
- Hernandez AF, Albert NM, Allen LA, et al. Multiple cardiac sensors for management of heart failure (MANAGE- HF) - phase I evaluation of the integration and safety of the HeartLogic multisensor algorithm in patients with heart failure. J Card Fail. 2022;28:1245-1254. doi:10.1016/j.cardfail.2022.03.349
- Santini L, D’Onofrio A, Dello Russo A, et al. Prospective evaluation of the multisensor HeartLogic algorithm for heart failure monitoring. Clin Cardiol. 2020;43:691-697. doi:10.1002/clc.23366
- Yu CM, Wang L, Chau E, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112:841-848. doi:10.1161/CIRCULATIONAHA.104.492207
- Adamson PB, Abraham WT, Stevenson LW, et al. Pulmonary artery pressure-guided heart failure management reduces 30-day readmissions. Circ Heart Fail. 2016;9:e002600. doi:10.1161/CIRCHEARTFAILURE.115.002600
- Lindenfeld J, Zile MR, Desai AS, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomised controlled trial. Lancet. 2021;398:991-1001. doi:10.1016/S0140-6736(21)01754-2
Heart failure (HF) is a prevalent disease in the United States affecting > 6.5 million adults and contributing to significant morbidity and mortality.1 The disease course associated with HF includes potential symptom improvement with intermittent periods of decompensation and possible clinical deterioration. Multiple therapies have been developed to improve outcomes in people with HF—to palliate HF symptoms, prevent hospitalizations, and reduce mortality.2 However, the risks of decompensation and hospitalization remain. HF decompensation development may precede clear actionable symptoms such as worsening dyspnea, noticeable edema, or weight gain. Tools to identify patient deterioration and trigger interventions to prevent HF admissions are clinically attractive compared with reliance on subjective factors alone.
Cardiac resynchronization therapy (CRT) and implantable cardioverter-defibrillator (ICD) devices made by Boston Scientific include the HeartLogic monitoring feature. Five main sensors produce an index risk score; an index score > 16 warns clinicians that the patient is at an increased risk for a HF event.3 The 5 sensors are thoracic impedance, first (S1) and third heart sounds (S3), night heart rate (NHR), respiratory rate (RR), and activity. Each sensor can draw attention to the primary driver of the alert and guide health care practitioners (HCPs) to the appropriate interventions.3 A HeartLogic alert example is shown in Figure 1.

The S3 occurs during the early diastolic phase when blood moves into the ventricles. As HF worsens, with a combination of elevated filling pressures and reduced cardiac muscle compliance, S3 can become more pronounced.4 The S1 is correlated with the contractility of the left ventricle and will be reduced in patients at risk for HF events.5 Physical activity is a long-term prognostic marker in patients with HF; reduced activity is associated with mortality and increased risk of an HF event.6 Thoracic impedance is a sensor used to identify pulmonary congestion, pocket infections, pleural/pericardial effusion, and respiratory infections. The accumulation of intrathoracic fluid during pulmonary congestion increases conductance, causing a decrease in impedance.7 RR will increase as patients experience dyspnea with a more rapid, shallow breath and may trigger alerts closer to the actual HF event than other sensors. Nearly 90% of patients hospitalized for HF experience shortness of breath.8,9 NHR is used as a surrogate for resting heart rate (HR). A high resting HR is correlated with the progression of coronary atherosclerosis, harmful effects on left ventricular function, and increased risk of myocardial ischemia and ventricular arrhythmias.10
One of the challenges with preventing hospitalizations may be the lack of patient reported symptoms leading up to the event. The purpose of the sensors and HeartLogic index is to identify patients a median of 34 days before an HF event (HF admission or unscheduled intervention with intravenous treatment) with a sensitivity rate of 70%.3 According to real-word experience data, alerts have been found to precede HF symptoms by a median of 12 days and HF events such as hospitalizations by a median of 38 days, with an overall 67% reduction in HF hospitalizations when integrated into clinical care.11,12
MANAGE-HF evaluated 191 patients with HF with reduced ejection fraction (HFrEF) (< 35%), New York Heart Association class II-III symptoms, and who had an implanted CRT and/or ICD to develop an alert management guide to optimize medical treatment.12 It aimed to adjust patient regimen within 6 days of an elevated Heart- Logic index by either initiation, escalation, or maintenance of HF treatment depending on the index trend after the initial alert. This trial found that by focusing on such optimization, HF treatment was augmented during 74% of the 585 alert cases and during 54% of 3290 weekly alerts.
Initiation and uptitration of the 4 primary components of guideline-directed medical therapy (GDMT) are recommended by the 2022 Heart Failure Guidelines to reduce mortality and morbidity in patients with HFrEF.2 The 4 pillars of GDMT consist of -blockers (BB), sodium-glucose cotransporter type 2 inhibitors (SGLT2i), mineralocorticoid receptor antagonists (MRA), and renin-angiotensin-system inhibitors (RASi) including angiotensin II receptor blocker/neprilysin inhibitors (ARNi), angiotensin-converting enzyme inhibitors (ACEi) and angiotensin II receptor blockers (ARB) (Appendix 1). Obtaining and titrating to target doses wherever possible is recommended, as those were the doses that established safety and efficacy in patients with HFrEF in clinical trials.2 Pharmacists are adequately equipped to optimize HF GDMT and appropriately monitor drug response.

Through the use of HeartLogic in clinical practice, patients with HF have been shown to have improved clinical outcomes and are more likely to receive effective care; 80% of alerts were shown to provide new information to clinicians.13 This project sought to quantify the total number and types of pharmacist interventions driven by integration of HeartLogic index monitoring into practice.
Methods
The West Palm Beach Veterans Affairs Medical Center (WPBVAMC) Research Program Office approved this project and determined it was exempt from institutional review board oversight. Patients were screened retrospectively and prospectively from May 26, 2022, through December 31, 2022, by a cardiology clinical pharmacist practitioner (CPP) and a cardiology pharmacy resident using the local monitoring suite for the HeartLogic-compatible device, LATITUDE NXT. Read-only access to the local monitoring suit was granted by the National Cardiac Device Surveillance Program. Training for HeartLogic was completed through continuing education courses provided by Boston Scientific. Additional information was provided by Boston Scientific representative presentations and collaboration with WPBVAMC pacemaker clinic HCPs.
Individuals included were patients with HeartLogic-capable ICDs. A HeartLogic alert had to be present at initial patient contact. Patients were also contacted as part of routine clinical practice, but no formal number or frequency of calls to patients was required. The initial contact must be with a pharmacist for the patient to be included, but subsequent contact by other HCPs was included. Patients in the cardiology clinic are required to meet with a cardiologist at least annually; however, interim visits can be completed by advanced practice registered nurse practitioners, physicians assistants, or CPPs.
Patients in alert status were contacted by telephone and appropriate modifications of HF therapy were made by the CPP based on score metrics, medical record review, and patient interview. Information surrounding the initial alert, baseline patient data, medication and monitoring interventions made, and clinical outcomes such as hospitalization, symptom improvement, follow-up, and mortality were collected. Information for each encounter was collected until 42 days from the initial date of pharmacist contact.
Clinically successful tolerability of intervention implementation was defined as tolerability, adherence, and lack of adverse effects (AEs) per patient report at follow-up or within 42 days from initial alert (Appendix 2). A decrease in dose was not counted as intolerance. A single patient may have been counted as multiple encounters if the original intervention resulted in treatment intolerance and the patient remained in alert or if an additional alert occurred after 42 days of the initial alert. There were no specific time criteria for follow-up, which occurred at the CPP’s discretion.

There was no mandated algorithm used to alter medications based on the Heart- Logic score, nor were there required minimum or maximum numbers of interventions after an alert. Patient contact by telephone initiated an encounter. The types of interventions included medication increases, decreases, initiation, discontinuation, or no medication change. Each medication change and rationale, if applicable, was recorded for the encounter ≤ 42 days after the initial contact date. If a medication with required monitoring parameters was augmented, the pharmacist was responsible for ordering laboratory testing and follow-up. Most interventions were completed by telephone; however, some patients had in-person visits in the HF CPP clinic.
Outcomes
The primary outcome was the number of pharmacist interventions made to optimize GDMT, defined as either an initiation or dose increase. Key intervention analysis included the use and dosing of the 4 primary components of HF GDMT: BB, SGLT2i, MRA, and ARNi/ARB/ACEi. In addition to the 4 primary components of GDMT, loop diuretic changes were also recorded and analyzed. Secondary endpoints were the number of HF hospitalizations ≤ 42 days after the initial alert, and the effect of medication interventions on device metrics, patient symptoms, and tolerability. Successful tolerability was defined as continued use of augmented GDMT without intolerance or discontinuation. The primary analysis was analyzed through descriptive statistics. Median changes in HeartLogic scores and metrics from baseline were analyzed using a paired, 2-sided t test with an α of .05 to detect significance.
Results
There were 39 WPBVAMC patients with a HeartLogic-capable device. Twenty-one alert encounters were analyzed in 16 patients (41%) over 31 weeks of data collection. The 16 patients at baseline had a mean age of 74 years, all were male, and 12 (75%) were White. Eight patients (50%) had a recent ejection fraction (EF) between 30% and 40%. Three patients had an EF ≥ 40%. At the time of alert, 15 patients used BB (94%), 10 used loop diuretics (63%), and 9 used ARNi (56%) (Table 1).

There were 23 medication changes made during initial contact. The most common change was starting an SGLT2i (30%; n = 7), followed by starting an MRA (22%; n = 5), and increasing the ARNi dose (22%; n = 5). At the initial contact, ≥ 1 medication optimization occurred in 95% (n = 20) of encounters. The CPP contacted patients a mean of 4.8 days after the initial alert.
Patients were taking a mean of 2.6 primary GDMT medications at baseline and 3.0 at 42 days. CPP encounters led to a mean of 1.8 medication changes over the 6-week period (range, 0-5). Seventeen medications were started, 13 medications were increased, 3 medications were decreased, and 4 medications were stopped (Table 2). One ACEi and 1 ARB were switched as a therapy escalation to an ARNi. One patient was on 1 of 4 primary GDMTs at baseline, which increased to 4 GDMT agents at 42 days.

SGLT2 inhibitors were added most often at initial contact (54%) and throughout the 42-day period (41%). The most common successfully tolerated optimizations were RASi, followed by MRA, SGLT2 inhibitors, BB, and loop diuretics with 11, 6, 5, 3, and 2 patients, respectively. Interventions were tolerated by 90% of patients, and no HF hospitalization occurred during follow-up. All possible rationales for patients with the same or reduced number of GDMT at 42 days compared with baseline are shown in Appendix 2.
Device Metrics
During initial contact, the most common HeartLogic metric category that was predominantly worsening were heart sounds (S1, S3, and S3/S1 ratio), followed by compensatory mechanism sensors (NHR and RR) and congestion (impedance) at rates of 61.9%, 23.8%, and 14.3%, respectively (Figure 2).

The median HeartLogic index score was 18 at baseline and 5 at the end of the follow-up period (P < .001). The changes in score and metrics were compared with the type of successfully tolerated GDMT optimization made (Table 3). The GDMT optimization analysis included SGLT2i, RASi, MRA, BB, and loop diuretics. All interventions reduced the overall HeartLogic index score, ranging from a 9.5-point reduction (loop diuretics) to a 16-point reduction (SGLT2i and BB). Optimization of SGLT2i, RASi, and loop diuretics had a positive impact on S1 score. For S3 score, SGLT2i, MRA, and BB had a positive impact. All medications, except for SGLT2i therapy, reduced the NHR score. Optimization of MRA, SGLT2i, and BB had positive impacts on the impedance score. All medications reduced RR from baseline. Only SGLT2i and loop diuretics had positive impacts on the activity score.

Clinical Outcomes and Adverse Effects
Within 42 days of contact, 17 encounters (81%) had ≥ 1 follow-up appointment with a CPP and all 21 patients had ≥ 1 follow-up health care team member. One patient had a HF-related hospitalization within 42 days of contact; however, that individual refused the recommended medication intervention. There were 13 encounters (62%) with reported symptoms at the time of the initial alert and 10 (77%) had subjective symptom improvement at 42 days (Appendix 3).

Of 30 medication optimizations, 27 primary GDMT medications were tolerated. Two medication intolerances led to discontinuation (1 SGLT2i and 1 loop diuretic) and 1 patient never started the SGLT2i (Table 4). There was only 1 known patient who did not follow the directions to adjust their medications. That individual was included because the patient agreed to the change during the CPP visit but later reported that he had never started the SGLT2i.

Discussion
The HeartLogic tool created a bridge for patients with HF to work with CPPs as soon as possible to optimize medication therapy to reduce HF events. This study highlights an additional area of expertise and service that CPPs may offer to their specialty HF clinic team. Over 31 weeks, 21 encounters and 30 medication optimizations were completed. These interventions led to significant reductions in HeartLogic scores, improvements in symptoms, and optimization of HF care, most of which were well tolerated.
Additional hemodynamic monitoring devices are available. Similar to HeartLogic, OptiVol is a tool embedded in select Medtronic implantable devices that monitors fluid status. 14 CardioMEMS is an implantable pulmonary artery pressure sensor used as a presymptomatic data point to alert clinicians when HF is worsening. In the CHAMPION trial, the use of CardioMEMS showed a 28% reduction in HF-related hospitalization at 6 months.15 Conversely, in the GUIDE-HF trial, monitoring with CardioMEMS did not significantly reduce the composite endpoint of mortality and total HF events.16 Therefore, remote hemodynamic monitoring has variable results and the use of these tools remains uncertain per the clinical guidelines.2
The MANAGE-HF study that contributed to the validation of the HeartLogic tool may provide a comparison with this smaller single-center project. The time to follow-up within 7 days of alert was noted in only 54% of the patients in MANAGE-HF.12 In this study, 86% of patients received follow-up within 7 days, with a mean of 4.8 days. The quick turnaround from the time of alert to intervention portrays pharmacists as readily available HCPs.
In MANAGE-HF, 89% of medication augmentation involved loop diuretics or thiazides; in our project, loop diuretics were the least frequently changed medication. Most optimizations in this project included ARNi, SGLT2i, BB, and MRA, which have been shown to reduce morbidity and mortality.2 Our project included use of SGLT2i therapy to affect HeartLogic metrics, which has not been evaluated previously. SGLT2i were the most commonly initiated medication after an alert. Of the 5 tolerated SGLT2i optimization encounters, 4 were out of alert at 42 days.
SGLT2i resulted in a significant decrease in HeartLogic index score from baseline and were the only class of medication that did not produce a negative change in any metric. In this study, CPPs utilizing and acting on HeartLogic alerts led to 1 (4.8%) hospitalization with HF as the primary reason for admission and no hospitalizations as a secondary cause in 42 days, compared to 37% and 7.9% in the MANAGE-HF in 1 year, respectively. An additional screening 1 year after the initial alert found that 2 (12.5%) of 12 patients had been admitted with 1 HF hospitalization each.
A strength of this study was the ability to use HeartLogic to identify high-risk patients, provide a source of patient contact and monitoring, interpret 5 cardiac sensors, and optimize all HF GDMT, not just volume management. By focusing efforts on making patient contact and pharmacotherapy interventions with morbidity and mortality benefit, remote hemodynamic monitoring may show a clear clinical benefit and become a vital part of HF care.
Limitations
Checking for adherence and tolerance to medications were mainly patient reported if there was a CPP follow-up within 42 days, or potentially through refill history when unclear. However, this limitation is reflective of current practice where patients may have multiple clinicians working to optimize HF care and where there is reliance on patients in order to guide continued therapy. Although unable to explicitly show a reduction in HF events given lack of comparator group, the interventions made are associated with improved outcomes and thus would be expected to improve patient outcomes. Changes in vital signs were not tracked as part of this project, however the main rationale for changes made were to optimize GDMT therapy, not specifically to impact vital sign measures.
HeartLogic alerts prompted identification of high-risk patients with HF, pharmacist evaluation and outreach, patient-focused pharmacotherapy care, and beneficial patient outcomes. With only 2 cardiology CPPs checking alerts once weekly, future studies may be needed with larger samples to create algorithms and protocols to increase the clinical utility of this tool on a greater scale.
Conclusions
Cardiology CPP-led HF interventions triggered by HeartLogic alerts lead to effective patient identification, increased access to care, reductions in HeartLogic scores, improvements in symptoms, and optimization of HF care. This project demonstrates the practical utility of the HeartLogic suite in conjunction with CPP care to prioritize treatment for highrisk patients with HF in an efficient manner. The data highlight the potential value of the HeartLogic tool and a CPP in HF care to facilitate initiation and optimization of GDMT to ultimately improve the morbidity and mortality in patients with HF.
Heart failure (HF) is a prevalent disease in the United States affecting > 6.5 million adults and contributing to significant morbidity and mortality.1 The disease course associated with HF includes potential symptom improvement with intermittent periods of decompensation and possible clinical deterioration. Multiple therapies have been developed to improve outcomes in people with HF—to palliate HF symptoms, prevent hospitalizations, and reduce mortality.2 However, the risks of decompensation and hospitalization remain. HF decompensation development may precede clear actionable symptoms such as worsening dyspnea, noticeable edema, or weight gain. Tools to identify patient deterioration and trigger interventions to prevent HF admissions are clinically attractive compared with reliance on subjective factors alone.
Cardiac resynchronization therapy (CRT) and implantable cardioverter-defibrillator (ICD) devices made by Boston Scientific include the HeartLogic monitoring feature. Five main sensors produce an index risk score; an index score > 16 warns clinicians that the patient is at an increased risk for a HF event.3 The 5 sensors are thoracic impedance, first (S1) and third heart sounds (S3), night heart rate (NHR), respiratory rate (RR), and activity. Each sensor can draw attention to the primary driver of the alert and guide health care practitioners (HCPs) to the appropriate interventions.3 A HeartLogic alert example is shown in Figure 1.

The S3 occurs during the early diastolic phase when blood moves into the ventricles. As HF worsens, with a combination of elevated filling pressures and reduced cardiac muscle compliance, S3 can become more pronounced.4 The S1 is correlated with the contractility of the left ventricle and will be reduced in patients at risk for HF events.5 Physical activity is a long-term prognostic marker in patients with HF; reduced activity is associated with mortality and increased risk of an HF event.6 Thoracic impedance is a sensor used to identify pulmonary congestion, pocket infections, pleural/pericardial effusion, and respiratory infections. The accumulation of intrathoracic fluid during pulmonary congestion increases conductance, causing a decrease in impedance.7 RR will increase as patients experience dyspnea with a more rapid, shallow breath and may trigger alerts closer to the actual HF event than other sensors. Nearly 90% of patients hospitalized for HF experience shortness of breath.8,9 NHR is used as a surrogate for resting heart rate (HR). A high resting HR is correlated with the progression of coronary atherosclerosis, harmful effects on left ventricular function, and increased risk of myocardial ischemia and ventricular arrhythmias.10
One of the challenges with preventing hospitalizations may be the lack of patient reported symptoms leading up to the event. The purpose of the sensors and HeartLogic index is to identify patients a median of 34 days before an HF event (HF admission or unscheduled intervention with intravenous treatment) with a sensitivity rate of 70%.3 According to real-word experience data, alerts have been found to precede HF symptoms by a median of 12 days and HF events such as hospitalizations by a median of 38 days, with an overall 67% reduction in HF hospitalizations when integrated into clinical care.11,12
MANAGE-HF evaluated 191 patients with HF with reduced ejection fraction (HFrEF) (< 35%), New York Heart Association class II-III symptoms, and who had an implanted CRT and/or ICD to develop an alert management guide to optimize medical treatment.12 It aimed to adjust patient regimen within 6 days of an elevated Heart- Logic index by either initiation, escalation, or maintenance of HF treatment depending on the index trend after the initial alert. This trial found that by focusing on such optimization, HF treatment was augmented during 74% of the 585 alert cases and during 54% of 3290 weekly alerts.
Initiation and uptitration of the 4 primary components of guideline-directed medical therapy (GDMT) are recommended by the 2022 Heart Failure Guidelines to reduce mortality and morbidity in patients with HFrEF.2 The 4 pillars of GDMT consist of -blockers (BB), sodium-glucose cotransporter type 2 inhibitors (SGLT2i), mineralocorticoid receptor antagonists (MRA), and renin-angiotensin-system inhibitors (RASi) including angiotensin II receptor blocker/neprilysin inhibitors (ARNi), angiotensin-converting enzyme inhibitors (ACEi) and angiotensin II receptor blockers (ARB) (Appendix 1). Obtaining and titrating to target doses wherever possible is recommended, as those were the doses that established safety and efficacy in patients with HFrEF in clinical trials.2 Pharmacists are adequately equipped to optimize HF GDMT and appropriately monitor drug response.

Through the use of HeartLogic in clinical practice, patients with HF have been shown to have improved clinical outcomes and are more likely to receive effective care; 80% of alerts were shown to provide new information to clinicians.13 This project sought to quantify the total number and types of pharmacist interventions driven by integration of HeartLogic index monitoring into practice.
Methods
The West Palm Beach Veterans Affairs Medical Center (WPBVAMC) Research Program Office approved this project and determined it was exempt from institutional review board oversight. Patients were screened retrospectively and prospectively from May 26, 2022, through December 31, 2022, by a cardiology clinical pharmacist practitioner (CPP) and a cardiology pharmacy resident using the local monitoring suite for the HeartLogic-compatible device, LATITUDE NXT. Read-only access to the local monitoring suit was granted by the National Cardiac Device Surveillance Program. Training for HeartLogic was completed through continuing education courses provided by Boston Scientific. Additional information was provided by Boston Scientific representative presentations and collaboration with WPBVAMC pacemaker clinic HCPs.
Individuals included were patients with HeartLogic-capable ICDs. A HeartLogic alert had to be present at initial patient contact. Patients were also contacted as part of routine clinical practice, but no formal number or frequency of calls to patients was required. The initial contact must be with a pharmacist for the patient to be included, but subsequent contact by other HCPs was included. Patients in the cardiology clinic are required to meet with a cardiologist at least annually; however, interim visits can be completed by advanced practice registered nurse practitioners, physicians assistants, or CPPs.
Patients in alert status were contacted by telephone and appropriate modifications of HF therapy were made by the CPP based on score metrics, medical record review, and patient interview. Information surrounding the initial alert, baseline patient data, medication and monitoring interventions made, and clinical outcomes such as hospitalization, symptom improvement, follow-up, and mortality were collected. Information for each encounter was collected until 42 days from the initial date of pharmacist contact.
Clinically successful tolerability of intervention implementation was defined as tolerability, adherence, and lack of adverse effects (AEs) per patient report at follow-up or within 42 days from initial alert (Appendix 2). A decrease in dose was not counted as intolerance. A single patient may have been counted as multiple encounters if the original intervention resulted in treatment intolerance and the patient remained in alert or if an additional alert occurred after 42 days of the initial alert. There were no specific time criteria for follow-up, which occurred at the CPP’s discretion.

There was no mandated algorithm used to alter medications based on the Heart- Logic score, nor were there required minimum or maximum numbers of interventions after an alert. Patient contact by telephone initiated an encounter. The types of interventions included medication increases, decreases, initiation, discontinuation, or no medication change. Each medication change and rationale, if applicable, was recorded for the encounter ≤ 42 days after the initial contact date. If a medication with required monitoring parameters was augmented, the pharmacist was responsible for ordering laboratory testing and follow-up. Most interventions were completed by telephone; however, some patients had in-person visits in the HF CPP clinic.
Outcomes
The primary outcome was the number of pharmacist interventions made to optimize GDMT, defined as either an initiation or dose increase. Key intervention analysis included the use and dosing of the 4 primary components of HF GDMT: BB, SGLT2i, MRA, and ARNi/ARB/ACEi. In addition to the 4 primary components of GDMT, loop diuretic changes were also recorded and analyzed. Secondary endpoints were the number of HF hospitalizations ≤ 42 days after the initial alert, and the effect of medication interventions on device metrics, patient symptoms, and tolerability. Successful tolerability was defined as continued use of augmented GDMT without intolerance or discontinuation. The primary analysis was analyzed through descriptive statistics. Median changes in HeartLogic scores and metrics from baseline were analyzed using a paired, 2-sided t test with an α of .05 to detect significance.
Results
There were 39 WPBVAMC patients with a HeartLogic-capable device. Twenty-one alert encounters were analyzed in 16 patients (41%) over 31 weeks of data collection. The 16 patients at baseline had a mean age of 74 years, all were male, and 12 (75%) were White. Eight patients (50%) had a recent ejection fraction (EF) between 30% and 40%. Three patients had an EF ≥ 40%. At the time of alert, 15 patients used BB (94%), 10 used loop diuretics (63%), and 9 used ARNi (56%) (Table 1).

There were 23 medication changes made during initial contact. The most common change was starting an SGLT2i (30%; n = 7), followed by starting an MRA (22%; n = 5), and increasing the ARNi dose (22%; n = 5). At the initial contact, ≥ 1 medication optimization occurred in 95% (n = 20) of encounters. The CPP contacted patients a mean of 4.8 days after the initial alert.
Patients were taking a mean of 2.6 primary GDMT medications at baseline and 3.0 at 42 days. CPP encounters led to a mean of 1.8 medication changes over the 6-week period (range, 0-5). Seventeen medications were started, 13 medications were increased, 3 medications were decreased, and 4 medications were stopped (Table 2). One ACEi and 1 ARB were switched as a therapy escalation to an ARNi. One patient was on 1 of 4 primary GDMTs at baseline, which increased to 4 GDMT agents at 42 days.

SGLT2 inhibitors were added most often at initial contact (54%) and throughout the 42-day period (41%). The most common successfully tolerated optimizations were RASi, followed by MRA, SGLT2 inhibitors, BB, and loop diuretics with 11, 6, 5, 3, and 2 patients, respectively. Interventions were tolerated by 90% of patients, and no HF hospitalization occurred during follow-up. All possible rationales for patients with the same or reduced number of GDMT at 42 days compared with baseline are shown in Appendix 2.
Device Metrics
During initial contact, the most common HeartLogic metric category that was predominantly worsening were heart sounds (S1, S3, and S3/S1 ratio), followed by compensatory mechanism sensors (NHR and RR) and congestion (impedance) at rates of 61.9%, 23.8%, and 14.3%, respectively (Figure 2).

The median HeartLogic index score was 18 at baseline and 5 at the end of the follow-up period (P < .001). The changes in score and metrics were compared with the type of successfully tolerated GDMT optimization made (Table 3). The GDMT optimization analysis included SGLT2i, RASi, MRA, BB, and loop diuretics. All interventions reduced the overall HeartLogic index score, ranging from a 9.5-point reduction (loop diuretics) to a 16-point reduction (SGLT2i and BB). Optimization of SGLT2i, RASi, and loop diuretics had a positive impact on S1 score. For S3 score, SGLT2i, MRA, and BB had a positive impact. All medications, except for SGLT2i therapy, reduced the NHR score. Optimization of MRA, SGLT2i, and BB had positive impacts on the impedance score. All medications reduced RR from baseline. Only SGLT2i and loop diuretics had positive impacts on the activity score.

Clinical Outcomes and Adverse Effects
Within 42 days of contact, 17 encounters (81%) had ≥ 1 follow-up appointment with a CPP and all 21 patients had ≥ 1 follow-up health care team member. One patient had a HF-related hospitalization within 42 days of contact; however, that individual refused the recommended medication intervention. There were 13 encounters (62%) with reported symptoms at the time of the initial alert and 10 (77%) had subjective symptom improvement at 42 days (Appendix 3).

Of 30 medication optimizations, 27 primary GDMT medications were tolerated. Two medication intolerances led to discontinuation (1 SGLT2i and 1 loop diuretic) and 1 patient never started the SGLT2i (Table 4). There was only 1 known patient who did not follow the directions to adjust their medications. That individual was included because the patient agreed to the change during the CPP visit but later reported that he had never started the SGLT2i.

Discussion
The HeartLogic tool created a bridge for patients with HF to work with CPPs as soon as possible to optimize medication therapy to reduce HF events. This study highlights an additional area of expertise and service that CPPs may offer to their specialty HF clinic team. Over 31 weeks, 21 encounters and 30 medication optimizations were completed. These interventions led to significant reductions in HeartLogic scores, improvements in symptoms, and optimization of HF care, most of which were well tolerated.
Additional hemodynamic monitoring devices are available. Similar to HeartLogic, OptiVol is a tool embedded in select Medtronic implantable devices that monitors fluid status. 14 CardioMEMS is an implantable pulmonary artery pressure sensor used as a presymptomatic data point to alert clinicians when HF is worsening. In the CHAMPION trial, the use of CardioMEMS showed a 28% reduction in HF-related hospitalization at 6 months.15 Conversely, in the GUIDE-HF trial, monitoring with CardioMEMS did not significantly reduce the composite endpoint of mortality and total HF events.16 Therefore, remote hemodynamic monitoring has variable results and the use of these tools remains uncertain per the clinical guidelines.2
The MANAGE-HF study that contributed to the validation of the HeartLogic tool may provide a comparison with this smaller single-center project. The time to follow-up within 7 days of alert was noted in only 54% of the patients in MANAGE-HF.12 In this study, 86% of patients received follow-up within 7 days, with a mean of 4.8 days. The quick turnaround from the time of alert to intervention portrays pharmacists as readily available HCPs.
In MANAGE-HF, 89% of medication augmentation involved loop diuretics or thiazides; in our project, loop diuretics were the least frequently changed medication. Most optimizations in this project included ARNi, SGLT2i, BB, and MRA, which have been shown to reduce morbidity and mortality.2 Our project included use of SGLT2i therapy to affect HeartLogic metrics, which has not been evaluated previously. SGLT2i were the most commonly initiated medication after an alert. Of the 5 tolerated SGLT2i optimization encounters, 4 were out of alert at 42 days.
SGLT2i resulted in a significant decrease in HeartLogic index score from baseline and were the only class of medication that did not produce a negative change in any metric. In this study, CPPs utilizing and acting on HeartLogic alerts led to 1 (4.8%) hospitalization with HF as the primary reason for admission and no hospitalizations as a secondary cause in 42 days, compared to 37% and 7.9% in the MANAGE-HF in 1 year, respectively. An additional screening 1 year after the initial alert found that 2 (12.5%) of 12 patients had been admitted with 1 HF hospitalization each.
A strength of this study was the ability to use HeartLogic to identify high-risk patients, provide a source of patient contact and monitoring, interpret 5 cardiac sensors, and optimize all HF GDMT, not just volume management. By focusing efforts on making patient contact and pharmacotherapy interventions with morbidity and mortality benefit, remote hemodynamic monitoring may show a clear clinical benefit and become a vital part of HF care.
Limitations
Checking for adherence and tolerance to medications were mainly patient reported if there was a CPP follow-up within 42 days, or potentially through refill history when unclear. However, this limitation is reflective of current practice where patients may have multiple clinicians working to optimize HF care and where there is reliance on patients in order to guide continued therapy. Although unable to explicitly show a reduction in HF events given lack of comparator group, the interventions made are associated with improved outcomes and thus would be expected to improve patient outcomes. Changes in vital signs were not tracked as part of this project, however the main rationale for changes made were to optimize GDMT therapy, not specifically to impact vital sign measures.
HeartLogic alerts prompted identification of high-risk patients with HF, pharmacist evaluation and outreach, patient-focused pharmacotherapy care, and beneficial patient outcomes. With only 2 cardiology CPPs checking alerts once weekly, future studies may be needed with larger samples to create algorithms and protocols to increase the clinical utility of this tool on a greater scale.
Conclusions
Cardiology CPP-led HF interventions triggered by HeartLogic alerts lead to effective patient identification, increased access to care, reductions in HeartLogic scores, improvements in symptoms, and optimization of HF care. This project demonstrates the practical utility of the HeartLogic suite in conjunction with CPP care to prioritize treatment for highrisk patients with HF in an efficient manner. The data highlight the potential value of the HeartLogic tool and a CPP in HF care to facilitate initiation and optimization of GDMT to ultimately improve the morbidity and mortality in patients with HF.
- Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2023 update: a report from the American Heart Association. Circulation. 2023;147:e93-e621. doi:10.1161/CIR.0000000000001123
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/ American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895-e1032. doi:10.1161/CIR.0000000000001063
- Boehmer JP, Hariharan R, Devecchi FG, et al. A multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. J Am Coll Cardiol HF. 2017;5:216-225. doi:10.1016/j.jchf.2016.12.011
- Cao M, Gardner RS, Hariharan R, et al. Ambulatory monitoring of heart sounds via an implanted device is superior to auscultation for prediction of heart failure events. J Card Fail. 2020;26:151-159. doi:10.1016/j.cardfail.2019.10.006
- Calò L, Capucci A, Santini L, et al. ICD-measured heart sounds and their correlation with echocardiographic indexes of systolic and diastolic function. J Interv Card Electrophysiol. 2020;58:95-101. doi:10.1007/s10840-019-00668
- Del Buono MG, Arena R, Borlaug BA, et al. Exercise intolerance in patients with heart failure: JACC state-of-the- art review. J Am Coll Cardiol. 2019;73:2209-2225. doi:10.1016/j.jacc.2019.01.072
- Yu CM, Wang L, Chau E, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112:841-848. doi:10.1161/CIRCULATIONAHA.104.492207
- Rials S, Aktas M, An Q, et al. Continuous respiratory rate is superior to routine outpatient dyspnea assessment for predicting heart failure events. J Card Fail. 2018;24:S45.
- Fonarow GC, ADHERE Scientific Advisory Committee. The Acute Decompensated Heart Failure National Registry (ADHERE): opportunities to improve care of patients hospitalized with acute decompensated heart failure. Rev Cardiovasc Med. 2003;4(suppl 7):S21-S30. doi:10.1016/j.cardfail.2018.07.130
- Fox K, Borer JS, Camm AJ, et al. Resting heart rate in cardiovascular disease. J Am Coll Cardiol. 2007;50:823-830. doi:10.1016/j.jacc.2007.04.079
- De Ruvo E, Capucci A, Ammirati F, et al. Preliminary experience of remote management of heart failure patients with a multisensor ICD alert [abstract P1536]. Eur J Heart Fail. 2019;21(suppl S1):370.
- Hernandez AF, Albert NM, Allen LA, et al. Multiple cardiac sensors for management of heart failure (MANAGE- HF) - phase I evaluation of the integration and safety of the HeartLogic multisensor algorithm in patients with heart failure. J Card Fail. 2022;28:1245-1254. doi:10.1016/j.cardfail.2022.03.349
- Santini L, D’Onofrio A, Dello Russo A, et al. Prospective evaluation of the multisensor HeartLogic algorithm for heart failure monitoring. Clin Cardiol. 2020;43:691-697. doi:10.1002/clc.23366
- Yu CM, Wang L, Chau E, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112:841-848. doi:10.1161/CIRCULATIONAHA.104.492207
- Adamson PB, Abraham WT, Stevenson LW, et al. Pulmonary artery pressure-guided heart failure management reduces 30-day readmissions. Circ Heart Fail. 2016;9:e002600. doi:10.1161/CIRCHEARTFAILURE.115.002600
- Lindenfeld J, Zile MR, Desai AS, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomised controlled trial. Lancet. 2021;398:991-1001. doi:10.1016/S0140-6736(21)01754-2
- Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2023 update: a report from the American Heart Association. Circulation. 2023;147:e93-e621. doi:10.1161/CIR.0000000000001123
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/ American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895-e1032. doi:10.1161/CIR.0000000000001063
- Boehmer JP, Hariharan R, Devecchi FG, et al. A multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. J Am Coll Cardiol HF. 2017;5:216-225. doi:10.1016/j.jchf.2016.12.011
- Cao M, Gardner RS, Hariharan R, et al. Ambulatory monitoring of heart sounds via an implanted device is superior to auscultation for prediction of heart failure events. J Card Fail. 2020;26:151-159. doi:10.1016/j.cardfail.2019.10.006
- Calò L, Capucci A, Santini L, et al. ICD-measured heart sounds and their correlation with echocardiographic indexes of systolic and diastolic function. J Interv Card Electrophysiol. 2020;58:95-101. doi:10.1007/s10840-019-00668
- Del Buono MG, Arena R, Borlaug BA, et al. Exercise intolerance in patients with heart failure: JACC state-of-the- art review. J Am Coll Cardiol. 2019;73:2209-2225. doi:10.1016/j.jacc.2019.01.072
- Yu CM, Wang L, Chau E, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112:841-848. doi:10.1161/CIRCULATIONAHA.104.492207
- Rials S, Aktas M, An Q, et al. Continuous respiratory rate is superior to routine outpatient dyspnea assessment for predicting heart failure events. J Card Fail. 2018;24:S45.
- Fonarow GC, ADHERE Scientific Advisory Committee. The Acute Decompensated Heart Failure National Registry (ADHERE): opportunities to improve care of patients hospitalized with acute decompensated heart failure. Rev Cardiovasc Med. 2003;4(suppl 7):S21-S30. doi:10.1016/j.cardfail.2018.07.130
- Fox K, Borer JS, Camm AJ, et al. Resting heart rate in cardiovascular disease. J Am Coll Cardiol. 2007;50:823-830. doi:10.1016/j.jacc.2007.04.079
- De Ruvo E, Capucci A, Ammirati F, et al. Preliminary experience of remote management of heart failure patients with a multisensor ICD alert [abstract P1536]. Eur J Heart Fail. 2019;21(suppl S1):370.
- Hernandez AF, Albert NM, Allen LA, et al. Multiple cardiac sensors for management of heart failure (MANAGE- HF) - phase I evaluation of the integration and safety of the HeartLogic multisensor algorithm in patients with heart failure. J Card Fail. 2022;28:1245-1254. doi:10.1016/j.cardfail.2022.03.349
- Santini L, D’Onofrio A, Dello Russo A, et al. Prospective evaluation of the multisensor HeartLogic algorithm for heart failure monitoring. Clin Cardiol. 2020;43:691-697. doi:10.1002/clc.23366
- Yu CM, Wang L, Chau E, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112:841-848. doi:10.1161/CIRCULATIONAHA.104.492207
- Adamson PB, Abraham WT, Stevenson LW, et al. Pulmonary artery pressure-guided heart failure management reduces 30-day readmissions. Circ Heart Fail. 2016;9:e002600. doi:10.1161/CIRCHEARTFAILURE.115.002600
- Lindenfeld J, Zile MR, Desai AS, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomised controlled trial. Lancet. 2021;398:991-1001. doi:10.1016/S0140-6736(21)01754-2
Heart Failure Diagnostic Alerts to Prompt Pharmacist Evaluation and Medication Optimization
Heart Failure Diagnostic Alerts to Prompt Pharmacist Evaluation and Medication Optimization
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).
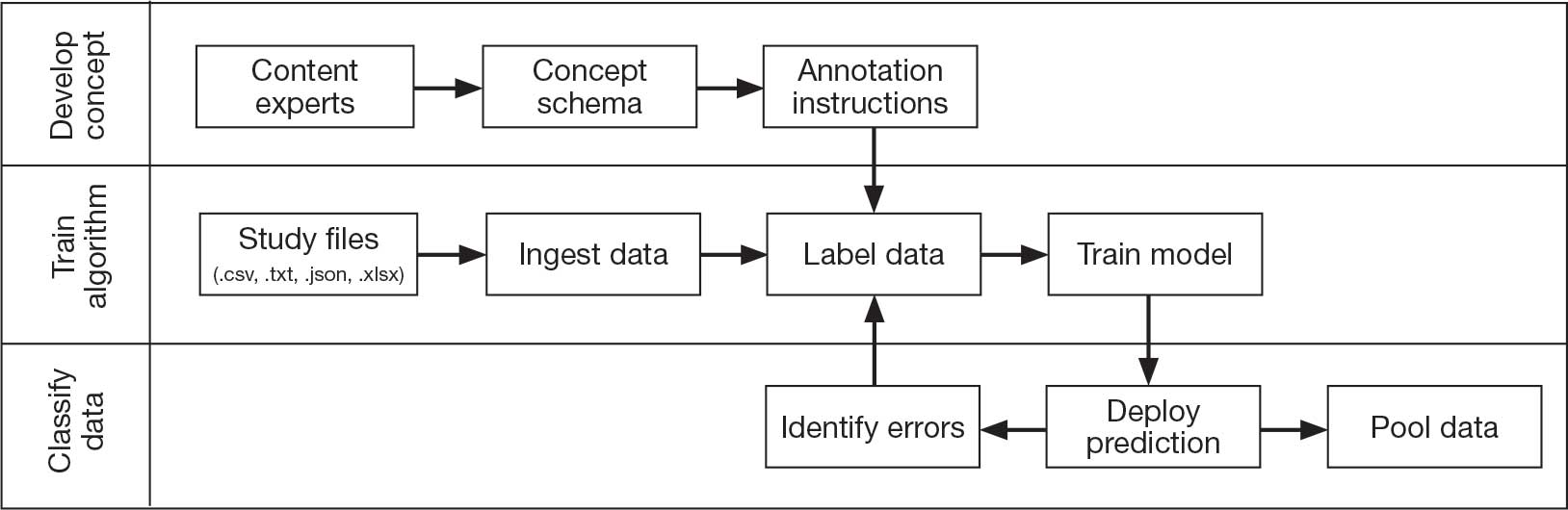
Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.
Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).

Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.

Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).

Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.

Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Codes, Contracts, and Commitments: Who Defines What is a Profession?
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Assessment of Automated vs Conventional Blood Pressure Measurements in a Veterans Affairs Clinical Practice Setting
Assessment of Automated vs Conventional Blood Pressure Measurements in a Veterans Affairs Clinical Practice Setting
Hypertension remains one of the most important modifiable risk factors for the prevention of cardiovascular (CV) events. According to a population-based study, 25% of CV events (CV death, heart disease, coronary revascularization, stroke, or heart failure) are attributable to hypertension.1 Recent guidelines have emphasized the importance of accurate blood pressure (BP) measurement in facilitating appropriate hypertension diagnosis and management.2-4
Currently, there are different BP measurement methods endorsed by practice guidelines. These include conventional in-office measurement, 24-hour ambulatory BP monitoring (ABPM), home BP monitoring (HBPM), and automated office BP (AOBP) measurement.2-4 AOBP device protocols vary but generally involve devices automatically taking multiple BP measurements while the patient is unattended. These measurements are then presented as a single averaged reading, with individual BP values available for review by the clinician.
Researchers have found that AOBP measurements have a greater association with ABPM values and can mitigate the white coat effect observed in a substantial proportion of patients during in-clinic BP measurement.5 A meta-analysis found that the use of AOBP was associated with a 10.5 mm Hg reduction in systolic BP (SBP) compared with traditional office-based BP assessments.5 Similarly, a separate meta-analysis found that AOBP SBP measures were on average 14.5 mm Hg lower than routine office or research setting values.6 In addition, CV risk outcomes data support the use of AOBP to screen and manage patients with hypertension. The Cardiovascular Health Awareness Program (CHAP) study used AOBP values to determine the risk for CV events (myocardial infarction, congestive heart failure, and stroke) in community-based patients aged ≥ 65 years.7 The study showed a significantly higher risk of CV events in patients with an SBP of 135 to 144 mm Hg and a diastolic BP (DBP) of 80 to 89 mm Hg. Therefore, the CHAP study researchers suggested an AOBP target of < 135/85 mm Hg to decrease the risk of CV events.7The landmark SPRINT trial, which was a major contributor to the development of BP target recommendations in guidelines, utilized AOBP to classify hypertension and guide management.2-4,8 SPRINT ultimately showed that intensive BP-lowering treatment (to SBP < 120 mm Hg) was associated with a 25% reduction in major CV events and a 27% reduction in all-cause mortality.8 Other evaluations found a close association between AOBP values and left ventricular mass index and carotid artery wall thickness as surrogate markers for end-organ damage.9,10 These data show AOBP as a reliable method to guide antihypertensive therapy interventions in the clinical setting.
Considering these proposed advantages, the 2017 Canadian guidelines for hypertension management recommend AOBP as the preferred method for clinic-based BP measurement, and the 2018 European Society of Cardiology/European Society of Hypertension blood pressure guidelines recommend the use of AOBP when feasible.3,4 The 2017 American College of Cardiology/American Heart Association Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults also discusses AOBP as a method to minimize potential confounders in BP values.2
This study evaluated the difference between AOBP and conventional in-office BP measurements obtained during cardiology clinic visits at the West Palm Beach Veterans Affairs Medical Center (WPBVAMC).
METHODS
A retrospective review of AOBP measurements was performed at the WPBVAMC cardiology clinic between May 26, 2017, and February 19, 2019. These AOBP measurements were taken at the discretion of a nurse or other clinician after initial, conventional BP measurements had been taken as part of clinic check-in procedures. No formal protocols dictated the use or timing of AOBP measurements. Similarly, the AOBP results were factored into clinical care decisions.
Clinicians at the cardiology clinic used AOBP averages that were derived using the BpTRU BPM-100 (BpTRU Medical Devices) meter, which averaged 5 BP readings taken at 1-minute intervals. Clinicians selected cuff size based on manufacturer recommendations. The testing was done with the patient seated alone in either a nursing triage area or a clinic office.
Data collected during the retrospective review included the clinician associated with the visit, the patient’s physical location and accompaniment status during AOBP measurement, conventionally measured BP and heart rates, and AOBP-derived BP and heart rate averages. Differences in BP values were compared with the paired t test, while binary comparisons were conducted through the McNemar test. Data collection and analysis were performed using Microsoft Excel.
During data collection, all information was stored in a secure drive accessible only to the investigators. The project was approved by the West Palm Beach Veterans Affairs Healthcare System Research and Development Committee as a nonresearch activity in accordance with Veterans Health Administration Handbook 1058.05; thus, institutional review board approval was not required.
RESULTS
Ninety-five nonconsecutive patients were included in the analysis. AOBP measurements were taken with the patient sitting alone in either a clinic office (n = 83) or nursing triage area (n = 12). Most patients were coming in for follow-up appointments; 13 patients (14%) had appointments related to a 24-hour ABPM session.
The mean SBP and DBP values were lower for the AOBP measurements vs the conventional BP measurements (mean SBP difference, 14.6 mm Hg; P < .001; mean DBP difference, 3.5 mm Hg; P = .0002) (Table). There were no appreciable differences in heart rates. The white coat effect was suggested based on an SBP reduction of > 20 mm Hg from conventional to AOBP measurements in 22 patients (23%), a DBP reduction of > 10 mm Hg in 21 patients (22%), and a reduction in both values in 8 patients (8%).
A controlled BP (< 130/80 mm Hg) was more common in the AOBP group than in the conventional group (22% vs 7%, respectively; P =.001).2 Review of conventional BP measurements indicated that 11 patients had systolic readings ≥ 180 mm Hg, 2 had diastolic readings ≥ 110 mm Hg, and 1 had a reading that was ≥ 180/110 mm Hg. AOBP measurements indicated that these 14 patients had SBP readings < 180 mm Hg and DBP readings < 110 mm Hg. The use of AOBP measurements may have mitigated unnecessary emergency room visits for these patients.
On review of clinic notes and actions associated with episodes of AOBP testing during routine follow-up clinic appointments, AOBP was determined to be useful with regard to clinical decision-making for 65 (79%) patients. Impacts of AOBP inclusion vs conventional BP assessments included clinician notation of AOBP, support for making changes that would have been considered based on conventional BP assessment. AOBP results gave support to forgoing a therapeutic intervention (ie, therapy addition or intensification) that may have been pursued based on conventional BP measurements in 25 of 82 patients (30%). These data suggest that AOBP readings can be useful and actionable by clinicians.
DISCUSSION
The findings of this study add to the growing evidence regarding AOBP use, application, and advantages in clinical practice. In this evaluation, the mean difference in SBP and DBP was 14.6 mm Hg and 3.5 mm Hg, respectively, from the conventional office measurements to the AOBP measurements. This difference is similar to that reported by the CAMBO trial and other evaluations, where the use of AOBP measurements corresponded to a reduction in SBP of between 10 and 20 mm Hg vs conventional measures.5,11-18
These findings showed a significantly higher percentage of controlled BP values (< 130/80 mm Hg) with AOBP values compared with conventional office measurements. The data supported the decision to defer antihypertensive therapy intervention in 30% of patients. Without AOBP data, patients may have been classified as uncontrolled, prompting therapy addition or intensification that could increase the risk of adverse events. Additionally, 14 patients would have met the criteria for hypertensive urgency under the guidelines at that time.2 With the use of AOBP readings, none of these patients were identified as having a hypertensive urgency, and they avoided an acute care referral or urgent intervention.
The discrepancy between AOBP and conventional office BP measurements suggested a white coat effect based on SBP and DBP readings in 22 (23%) and 21 (22%) patients, respectively. Practice guidelines recommend ABPM to mitigate a potential white coat effect.2-4 However, ABPM can be inconvenient for patients, as they need to travel to and from the clinic for fitting and removal (assuming that a facility has the device available for patient use). In addition, some patients may find it uncomfortable. Based on the correlation between AOBP and awake ABPM values, AOBP represents a feasible way to identify a white coat effect.
AOBP monitoring does not appear to be affected by the type of practice setting, as it has been evaluated in a variety of locations, including community-based pharmacies, primary care offices, and waiting rooms.12,19-22 However, potential AOBP implementation challenges may include office space constraints, clinician perception that it will delay workflow, and device cost. Costs associated with an AOBP meter vary widely based on device and procurement source, but have been estimated to range from $650 to > $2000.23 Published reports have described how to overcome AOBP implementation barriers.24,25
Limitations
The results of this evaluation should be interpreted cautiously due to several limitations. First, the retrospective study was conducted at a single clinic that may not be representative of other Veterans Health Administration or community-based populations. In addition, patient data such as age, sex, and body mass index were not available. AOBP measurements were obtained at the discretion of the clinician and not according to a prespecified protocol.
Conclusions
This analysis showed AOBP measurement leads to a greater percentage of controlled BP values compared with conventional office BP measurement, positioning it as a way to reduce BP misclassification, prevent potentially unnecessary therapeutic interventions, and mitigate the white coat effect.
- Cheng S, Claggett B, Correia AW, et al. Temporal Trends in the Population Attributable Risk for Cardiovascular Disease: The Atherosclerosis Risk in Communities Study. Circulation. 2014;130:820-828. doi.org/10.1161/CIRCULATIONAHA.113.008506
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):1269-1324. doi:10.1161/HYP.0000000000000066
- Leung AA, Daskalopoulou SS, Dasgupta K, et al. Hypertension Canada’s 2017 guidelines for diagnosis, risk assessment, prevention, and treatment of hypertension in adults. Can J Cardiol. 2017;33(5):557-576. doi:10.1016/j.cjca.2017.03.005
- Williams B, Mancia G, Spiering W, et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J. 2018;39(33):3021-3104. doi:10.1093/eurheartj/ehy339
- Pappaccogli M, Di Monaco S, Perlo E, et al. Comparison of automated office blood pressure with office and out-off-office measurement techniques. Hypertension. 2019;73(2):481-490. doi:10.1161/HYPERTENSIONAHA.118.12079
- Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension - a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362. doi:10.1001/jamainternmed.2018.6551
- Kaczorowski J, Chambers LW, Karwalajtys T, et al. Cardiovascular Health Awareness Program (CHAP): a community cluster-randomised trial among elderly Canadians. Prev Med. 2008;46(6):537-544. doi:10.1016/j.ypmed.2008.02.005
- SPRINT Research Group. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103-2116. doi:10.1056/NEJMoa1511939
- Andreadis EA, Agaliotis GD, Angelopoulos ET, et al. Automated office blood pressure and 24-h ambulatory measurements are equally associated with left ventricular mass index. Am J Hypertens. 2011;24(6):661-666. doi:10.1038/ajh.2011.38
- Campbell NRC, McKay DW, Conradson H, et al. Automated oscillometric blood pressure versus auscultatory blood pressure as a predictor of carotid intima-medial thickness in male firefighters. J Hum Hypertens. 2007;21(7):588-590. doi:10.1038/sj.jhh.1002190
- Myers MG, Godwin M, Dawes M et al. Conventional versus automated measurement of blood pressure in primary care patients with systolic hypertension: randomised parallel design controlled trial. BMJ. 2011;342:d286. doi:10.1136/bmj.d286
- Beckett L, Godwin M. The BpTRU automatic blood pressure monitor compared to 24 hour ambulatory blood pressure monitoring in the assessment of blood pressure in patients with hypertension. BMC Cardiovasc Disord. 2005;5(1):18. doi:10.1186/1471-2261-5-18
- Myers MG, Valdivieso M, Kiss A. Use of automated office blood pressure measurement to reduce the white coat response. J Hypertens. 2009;27(2):280-286. doi:10.1097/HJH.0b013e32831b9e6b
- Myers MG, Valdivieso M, Kiss A. Consistent relationship between automated office blood pressure recorded in different settings. Blood Press Monit. 2009;14(3):108-111. doi:10.1097/MBP.0b013e32832c5167
- Myers MG, Valdivieso M, Kiss A. Optimum frequency of office blood pressure measurement using an automated sphygmomanometer. Blood Press Monit. 2008;13(6):333-338. doi:10.1097/MBP.0b013e3283104247
- Myers MG. A proposed algorithm for diagnosing hypertension using automated office blood pressure measurement. J Hypertens. 2010;28(4):703-708. doi:10.1097/HJH.0b013e328335d091
- Godwin M, Birtwhistle R, Delva D, et al. Manual and automated office measurements in relation to awake ambulatory blood pressure monitoring. Fam Pract. 2011;28(1):110-117. doi:10.1093/fampra/cmq067
- Myers MG, Valdivieso M, Chessman M, Kiss A. Can sphygmomanometers designed for self-measurement of blood pressure in the home be used in office practice? Blood Press Monit. 2010;15(6):300-304. doi:10.1097/MBP.0b013e328340d128
- Leung AA, Nerenberg K, Daskalopoulou SS, et al. Hypertension Canada’s 2016 Canadian hypertension education program guidelines for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension. Can J Cardiol. 2016;32(5):569-588. doi:10.1016/j.cjca.2016.02.066
- Myers MG. A short history of automated office blood pressure - 15 years to SPRINT. J Clin Hypertens (Greenwich). 2016;18(8):721-724. doi:10.1111/jch.12820
- Myers MG, Kaczorowski J, Dawes M, Godwin M. Automated office blood pressure measurement in primary care. Can Fam Physician. 2014;60(2):127-132.
- Armstrong D, Matangi M, Brouillard D, Myers MG. Automated office blood pressure - being alone and not location is what matters most. Blood Press Monit. 2015;20(4):204-208. doi:10.1097/MBP.0000000000000133
- Yarows SA. What is the Cost of Measuring a Blood Pressure? Ann Clin Hypertens. 2018;2:59-66. doi:10.29328/journal.ach.1001012
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA. 1999;282(15):1458-1465. doi:10.1001/jama.282.15.1458
- Doane J, Buu J, Penrod MJ, et al. Measuring and managing blood pressure in a primary care setting: a pragmatic implementation study. J Am Board Fam Med. 2018;31(3):375-388. doi:10.3122/jabfm.2018.03.170450
Hypertension remains one of the most important modifiable risk factors for the prevention of cardiovascular (CV) events. According to a population-based study, 25% of CV events (CV death, heart disease, coronary revascularization, stroke, or heart failure) are attributable to hypertension.1 Recent guidelines have emphasized the importance of accurate blood pressure (BP) measurement in facilitating appropriate hypertension diagnosis and management.2-4
Currently, there are different BP measurement methods endorsed by practice guidelines. These include conventional in-office measurement, 24-hour ambulatory BP monitoring (ABPM), home BP monitoring (HBPM), and automated office BP (AOBP) measurement.2-4 AOBP device protocols vary but generally involve devices automatically taking multiple BP measurements while the patient is unattended. These measurements are then presented as a single averaged reading, with individual BP values available for review by the clinician.
Researchers have found that AOBP measurements have a greater association with ABPM values and can mitigate the white coat effect observed in a substantial proportion of patients during in-clinic BP measurement.5 A meta-analysis found that the use of AOBP was associated with a 10.5 mm Hg reduction in systolic BP (SBP) compared with traditional office-based BP assessments.5 Similarly, a separate meta-analysis found that AOBP SBP measures were on average 14.5 mm Hg lower than routine office or research setting values.6 In addition, CV risk outcomes data support the use of AOBP to screen and manage patients with hypertension. The Cardiovascular Health Awareness Program (CHAP) study used AOBP values to determine the risk for CV events (myocardial infarction, congestive heart failure, and stroke) in community-based patients aged ≥ 65 years.7 The study showed a significantly higher risk of CV events in patients with an SBP of 135 to 144 mm Hg and a diastolic BP (DBP) of 80 to 89 mm Hg. Therefore, the CHAP study researchers suggested an AOBP target of < 135/85 mm Hg to decrease the risk of CV events.7The landmark SPRINT trial, which was a major contributor to the development of BP target recommendations in guidelines, utilized AOBP to classify hypertension and guide management.2-4,8 SPRINT ultimately showed that intensive BP-lowering treatment (to SBP < 120 mm Hg) was associated with a 25% reduction in major CV events and a 27% reduction in all-cause mortality.8 Other evaluations found a close association between AOBP values and left ventricular mass index and carotid artery wall thickness as surrogate markers for end-organ damage.9,10 These data show AOBP as a reliable method to guide antihypertensive therapy interventions in the clinical setting.
Considering these proposed advantages, the 2017 Canadian guidelines for hypertension management recommend AOBP as the preferred method for clinic-based BP measurement, and the 2018 European Society of Cardiology/European Society of Hypertension blood pressure guidelines recommend the use of AOBP when feasible.3,4 The 2017 American College of Cardiology/American Heart Association Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults also discusses AOBP as a method to minimize potential confounders in BP values.2
This study evaluated the difference between AOBP and conventional in-office BP measurements obtained during cardiology clinic visits at the West Palm Beach Veterans Affairs Medical Center (WPBVAMC).
METHODS
A retrospective review of AOBP measurements was performed at the WPBVAMC cardiology clinic between May 26, 2017, and February 19, 2019. These AOBP measurements were taken at the discretion of a nurse or other clinician after initial, conventional BP measurements had been taken as part of clinic check-in procedures. No formal protocols dictated the use or timing of AOBP measurements. Similarly, the AOBP results were factored into clinical care decisions.
Clinicians at the cardiology clinic used AOBP averages that were derived using the BpTRU BPM-100 (BpTRU Medical Devices) meter, which averaged 5 BP readings taken at 1-minute intervals. Clinicians selected cuff size based on manufacturer recommendations. The testing was done with the patient seated alone in either a nursing triage area or a clinic office.
Data collected during the retrospective review included the clinician associated with the visit, the patient’s physical location and accompaniment status during AOBP measurement, conventionally measured BP and heart rates, and AOBP-derived BP and heart rate averages. Differences in BP values were compared with the paired t test, while binary comparisons were conducted through the McNemar test. Data collection and analysis were performed using Microsoft Excel.
During data collection, all information was stored in a secure drive accessible only to the investigators. The project was approved by the West Palm Beach Veterans Affairs Healthcare System Research and Development Committee as a nonresearch activity in accordance with Veterans Health Administration Handbook 1058.05; thus, institutional review board approval was not required.
RESULTS
Ninety-five nonconsecutive patients were included in the analysis. AOBP measurements were taken with the patient sitting alone in either a clinic office (n = 83) or nursing triage area (n = 12). Most patients were coming in for follow-up appointments; 13 patients (14%) had appointments related to a 24-hour ABPM session.
The mean SBP and DBP values were lower for the AOBP measurements vs the conventional BP measurements (mean SBP difference, 14.6 mm Hg; P < .001; mean DBP difference, 3.5 mm Hg; P = .0002) (Table). There were no appreciable differences in heart rates. The white coat effect was suggested based on an SBP reduction of > 20 mm Hg from conventional to AOBP measurements in 22 patients (23%), a DBP reduction of > 10 mm Hg in 21 patients (22%), and a reduction in both values in 8 patients (8%).

A controlled BP (< 130/80 mm Hg) was more common in the AOBP group than in the conventional group (22% vs 7%, respectively; P =.001).2 Review of conventional BP measurements indicated that 11 patients had systolic readings ≥ 180 mm Hg, 2 had diastolic readings ≥ 110 mm Hg, and 1 had a reading that was ≥ 180/110 mm Hg. AOBP measurements indicated that these 14 patients had SBP readings < 180 mm Hg and DBP readings < 110 mm Hg. The use of AOBP measurements may have mitigated unnecessary emergency room visits for these patients.
On review of clinic notes and actions associated with episodes of AOBP testing during routine follow-up clinic appointments, AOBP was determined to be useful with regard to clinical decision-making for 65 (79%) patients. Impacts of AOBP inclusion vs conventional BP assessments included clinician notation of AOBP, support for making changes that would have been considered based on conventional BP assessment. AOBP results gave support to forgoing a therapeutic intervention (ie, therapy addition or intensification) that may have been pursued based on conventional BP measurements in 25 of 82 patients (30%). These data suggest that AOBP readings can be useful and actionable by clinicians.
DISCUSSION
The findings of this study add to the growing evidence regarding AOBP use, application, and advantages in clinical practice. In this evaluation, the mean difference in SBP and DBP was 14.6 mm Hg and 3.5 mm Hg, respectively, from the conventional office measurements to the AOBP measurements. This difference is similar to that reported by the CAMBO trial and other evaluations, where the use of AOBP measurements corresponded to a reduction in SBP of between 10 and 20 mm Hg vs conventional measures.5,11-18
These findings showed a significantly higher percentage of controlled BP values (< 130/80 mm Hg) with AOBP values compared with conventional office measurements. The data supported the decision to defer antihypertensive therapy intervention in 30% of patients. Without AOBP data, patients may have been classified as uncontrolled, prompting therapy addition or intensification that could increase the risk of adverse events. Additionally, 14 patients would have met the criteria for hypertensive urgency under the guidelines at that time.2 With the use of AOBP readings, none of these patients were identified as having a hypertensive urgency, and they avoided an acute care referral or urgent intervention.
The discrepancy between AOBP and conventional office BP measurements suggested a white coat effect based on SBP and DBP readings in 22 (23%) and 21 (22%) patients, respectively. Practice guidelines recommend ABPM to mitigate a potential white coat effect.2-4 However, ABPM can be inconvenient for patients, as they need to travel to and from the clinic for fitting and removal (assuming that a facility has the device available for patient use). In addition, some patients may find it uncomfortable. Based on the correlation between AOBP and awake ABPM values, AOBP represents a feasible way to identify a white coat effect.
AOBP monitoring does not appear to be affected by the type of practice setting, as it has been evaluated in a variety of locations, including community-based pharmacies, primary care offices, and waiting rooms.12,19-22 However, potential AOBP implementation challenges may include office space constraints, clinician perception that it will delay workflow, and device cost. Costs associated with an AOBP meter vary widely based on device and procurement source, but have been estimated to range from $650 to > $2000.23 Published reports have described how to overcome AOBP implementation barriers.24,25
Limitations
The results of this evaluation should be interpreted cautiously due to several limitations. First, the retrospective study was conducted at a single clinic that may not be representative of other Veterans Health Administration or community-based populations. In addition, patient data such as age, sex, and body mass index were not available. AOBP measurements were obtained at the discretion of the clinician and not according to a prespecified protocol.
Conclusions
This analysis showed AOBP measurement leads to a greater percentage of controlled BP values compared with conventional office BP measurement, positioning it as a way to reduce BP misclassification, prevent potentially unnecessary therapeutic interventions, and mitigate the white coat effect.
Hypertension remains one of the most important modifiable risk factors for the prevention of cardiovascular (CV) events. According to a population-based study, 25% of CV events (CV death, heart disease, coronary revascularization, stroke, or heart failure) are attributable to hypertension.1 Recent guidelines have emphasized the importance of accurate blood pressure (BP) measurement in facilitating appropriate hypertension diagnosis and management.2-4
Currently, there are different BP measurement methods endorsed by practice guidelines. These include conventional in-office measurement, 24-hour ambulatory BP monitoring (ABPM), home BP monitoring (HBPM), and automated office BP (AOBP) measurement.2-4 AOBP device protocols vary but generally involve devices automatically taking multiple BP measurements while the patient is unattended. These measurements are then presented as a single averaged reading, with individual BP values available for review by the clinician.
Researchers have found that AOBP measurements have a greater association with ABPM values and can mitigate the white coat effect observed in a substantial proportion of patients during in-clinic BP measurement.5 A meta-analysis found that the use of AOBP was associated with a 10.5 mm Hg reduction in systolic BP (SBP) compared with traditional office-based BP assessments.5 Similarly, a separate meta-analysis found that AOBP SBP measures were on average 14.5 mm Hg lower than routine office or research setting values.6 In addition, CV risk outcomes data support the use of AOBP to screen and manage patients with hypertension. The Cardiovascular Health Awareness Program (CHAP) study used AOBP values to determine the risk for CV events (myocardial infarction, congestive heart failure, and stroke) in community-based patients aged ≥ 65 years.7 The study showed a significantly higher risk of CV events in patients with an SBP of 135 to 144 mm Hg and a diastolic BP (DBP) of 80 to 89 mm Hg. Therefore, the CHAP study researchers suggested an AOBP target of < 135/85 mm Hg to decrease the risk of CV events.7The landmark SPRINT trial, which was a major contributor to the development of BP target recommendations in guidelines, utilized AOBP to classify hypertension and guide management.2-4,8 SPRINT ultimately showed that intensive BP-lowering treatment (to SBP < 120 mm Hg) was associated with a 25% reduction in major CV events and a 27% reduction in all-cause mortality.8 Other evaluations found a close association between AOBP values and left ventricular mass index and carotid artery wall thickness as surrogate markers for end-organ damage.9,10 These data show AOBP as a reliable method to guide antihypertensive therapy interventions in the clinical setting.
Considering these proposed advantages, the 2017 Canadian guidelines for hypertension management recommend AOBP as the preferred method for clinic-based BP measurement, and the 2018 European Society of Cardiology/European Society of Hypertension blood pressure guidelines recommend the use of AOBP when feasible.3,4 The 2017 American College of Cardiology/American Heart Association Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults also discusses AOBP as a method to minimize potential confounders in BP values.2
This study evaluated the difference between AOBP and conventional in-office BP measurements obtained during cardiology clinic visits at the West Palm Beach Veterans Affairs Medical Center (WPBVAMC).
METHODS
A retrospective review of AOBP measurements was performed at the WPBVAMC cardiology clinic between May 26, 2017, and February 19, 2019. These AOBP measurements were taken at the discretion of a nurse or other clinician after initial, conventional BP measurements had been taken as part of clinic check-in procedures. No formal protocols dictated the use or timing of AOBP measurements. Similarly, the AOBP results were factored into clinical care decisions.
Clinicians at the cardiology clinic used AOBP averages that were derived using the BpTRU BPM-100 (BpTRU Medical Devices) meter, which averaged 5 BP readings taken at 1-minute intervals. Clinicians selected cuff size based on manufacturer recommendations. The testing was done with the patient seated alone in either a nursing triage area or a clinic office.
Data collected during the retrospective review included the clinician associated with the visit, the patient’s physical location and accompaniment status during AOBP measurement, conventionally measured BP and heart rates, and AOBP-derived BP and heart rate averages. Differences in BP values were compared with the paired t test, while binary comparisons were conducted through the McNemar test. Data collection and analysis were performed using Microsoft Excel.
During data collection, all information was stored in a secure drive accessible only to the investigators. The project was approved by the West Palm Beach Veterans Affairs Healthcare System Research and Development Committee as a nonresearch activity in accordance with Veterans Health Administration Handbook 1058.05; thus, institutional review board approval was not required.
RESULTS
Ninety-five nonconsecutive patients were included in the analysis. AOBP measurements were taken with the patient sitting alone in either a clinic office (n = 83) or nursing triage area (n = 12). Most patients were coming in for follow-up appointments; 13 patients (14%) had appointments related to a 24-hour ABPM session.
The mean SBP and DBP values were lower for the AOBP measurements vs the conventional BP measurements (mean SBP difference, 14.6 mm Hg; P < .001; mean DBP difference, 3.5 mm Hg; P = .0002) (Table). There were no appreciable differences in heart rates. The white coat effect was suggested based on an SBP reduction of > 20 mm Hg from conventional to AOBP measurements in 22 patients (23%), a DBP reduction of > 10 mm Hg in 21 patients (22%), and a reduction in both values in 8 patients (8%).

A controlled BP (< 130/80 mm Hg) was more common in the AOBP group than in the conventional group (22% vs 7%, respectively; P =.001).2 Review of conventional BP measurements indicated that 11 patients had systolic readings ≥ 180 mm Hg, 2 had diastolic readings ≥ 110 mm Hg, and 1 had a reading that was ≥ 180/110 mm Hg. AOBP measurements indicated that these 14 patients had SBP readings < 180 mm Hg and DBP readings < 110 mm Hg. The use of AOBP measurements may have mitigated unnecessary emergency room visits for these patients.
On review of clinic notes and actions associated with episodes of AOBP testing during routine follow-up clinic appointments, AOBP was determined to be useful with regard to clinical decision-making for 65 (79%) patients. Impacts of AOBP inclusion vs conventional BP assessments included clinician notation of AOBP, support for making changes that would have been considered based on conventional BP assessment. AOBP results gave support to forgoing a therapeutic intervention (ie, therapy addition or intensification) that may have been pursued based on conventional BP measurements in 25 of 82 patients (30%). These data suggest that AOBP readings can be useful and actionable by clinicians.
DISCUSSION
The findings of this study add to the growing evidence regarding AOBP use, application, and advantages in clinical practice. In this evaluation, the mean difference in SBP and DBP was 14.6 mm Hg and 3.5 mm Hg, respectively, from the conventional office measurements to the AOBP measurements. This difference is similar to that reported by the CAMBO trial and other evaluations, where the use of AOBP measurements corresponded to a reduction in SBP of between 10 and 20 mm Hg vs conventional measures.5,11-18
These findings showed a significantly higher percentage of controlled BP values (< 130/80 mm Hg) with AOBP values compared with conventional office measurements. The data supported the decision to defer antihypertensive therapy intervention in 30% of patients. Without AOBP data, patients may have been classified as uncontrolled, prompting therapy addition or intensification that could increase the risk of adverse events. Additionally, 14 patients would have met the criteria for hypertensive urgency under the guidelines at that time.2 With the use of AOBP readings, none of these patients were identified as having a hypertensive urgency, and they avoided an acute care referral or urgent intervention.
The discrepancy between AOBP and conventional office BP measurements suggested a white coat effect based on SBP and DBP readings in 22 (23%) and 21 (22%) patients, respectively. Practice guidelines recommend ABPM to mitigate a potential white coat effect.2-4 However, ABPM can be inconvenient for patients, as they need to travel to and from the clinic for fitting and removal (assuming that a facility has the device available for patient use). In addition, some patients may find it uncomfortable. Based on the correlation between AOBP and awake ABPM values, AOBP represents a feasible way to identify a white coat effect.
AOBP monitoring does not appear to be affected by the type of practice setting, as it has been evaluated in a variety of locations, including community-based pharmacies, primary care offices, and waiting rooms.12,19-22 However, potential AOBP implementation challenges may include office space constraints, clinician perception that it will delay workflow, and device cost. Costs associated with an AOBP meter vary widely based on device and procurement source, but have been estimated to range from $650 to > $2000.23 Published reports have described how to overcome AOBP implementation barriers.24,25
Limitations
The results of this evaluation should be interpreted cautiously due to several limitations. First, the retrospective study was conducted at a single clinic that may not be representative of other Veterans Health Administration or community-based populations. In addition, patient data such as age, sex, and body mass index were not available. AOBP measurements were obtained at the discretion of the clinician and not according to a prespecified protocol.
Conclusions
This analysis showed AOBP measurement leads to a greater percentage of controlled BP values compared with conventional office BP measurement, positioning it as a way to reduce BP misclassification, prevent potentially unnecessary therapeutic interventions, and mitigate the white coat effect.
- Cheng S, Claggett B, Correia AW, et al. Temporal Trends in the Population Attributable Risk for Cardiovascular Disease: The Atherosclerosis Risk in Communities Study. Circulation. 2014;130:820-828. doi.org/10.1161/CIRCULATIONAHA.113.008506
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):1269-1324. doi:10.1161/HYP.0000000000000066
- Leung AA, Daskalopoulou SS, Dasgupta K, et al. Hypertension Canada’s 2017 guidelines for diagnosis, risk assessment, prevention, and treatment of hypertension in adults. Can J Cardiol. 2017;33(5):557-576. doi:10.1016/j.cjca.2017.03.005
- Williams B, Mancia G, Spiering W, et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J. 2018;39(33):3021-3104. doi:10.1093/eurheartj/ehy339
- Pappaccogli M, Di Monaco S, Perlo E, et al. Comparison of automated office blood pressure with office and out-off-office measurement techniques. Hypertension. 2019;73(2):481-490. doi:10.1161/HYPERTENSIONAHA.118.12079
- Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension - a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362. doi:10.1001/jamainternmed.2018.6551
- Kaczorowski J, Chambers LW, Karwalajtys T, et al. Cardiovascular Health Awareness Program (CHAP): a community cluster-randomised trial among elderly Canadians. Prev Med. 2008;46(6):537-544. doi:10.1016/j.ypmed.2008.02.005
- SPRINT Research Group. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103-2116. doi:10.1056/NEJMoa1511939
- Andreadis EA, Agaliotis GD, Angelopoulos ET, et al. Automated office blood pressure and 24-h ambulatory measurements are equally associated with left ventricular mass index. Am J Hypertens. 2011;24(6):661-666. doi:10.1038/ajh.2011.38
- Campbell NRC, McKay DW, Conradson H, et al. Automated oscillometric blood pressure versus auscultatory blood pressure as a predictor of carotid intima-medial thickness in male firefighters. J Hum Hypertens. 2007;21(7):588-590. doi:10.1038/sj.jhh.1002190
- Myers MG, Godwin M, Dawes M et al. Conventional versus automated measurement of blood pressure in primary care patients with systolic hypertension: randomised parallel design controlled trial. BMJ. 2011;342:d286. doi:10.1136/bmj.d286
- Beckett L, Godwin M. The BpTRU automatic blood pressure monitor compared to 24 hour ambulatory blood pressure monitoring in the assessment of blood pressure in patients with hypertension. BMC Cardiovasc Disord. 2005;5(1):18. doi:10.1186/1471-2261-5-18
- Myers MG, Valdivieso M, Kiss A. Use of automated office blood pressure measurement to reduce the white coat response. J Hypertens. 2009;27(2):280-286. doi:10.1097/HJH.0b013e32831b9e6b
- Myers MG, Valdivieso M, Kiss A. Consistent relationship between automated office blood pressure recorded in different settings. Blood Press Monit. 2009;14(3):108-111. doi:10.1097/MBP.0b013e32832c5167
- Myers MG, Valdivieso M, Kiss A. Optimum frequency of office blood pressure measurement using an automated sphygmomanometer. Blood Press Monit. 2008;13(6):333-338. doi:10.1097/MBP.0b013e3283104247
- Myers MG. A proposed algorithm for diagnosing hypertension using automated office blood pressure measurement. J Hypertens. 2010;28(4):703-708. doi:10.1097/HJH.0b013e328335d091
- Godwin M, Birtwhistle R, Delva D, et al. Manual and automated office measurements in relation to awake ambulatory blood pressure monitoring. Fam Pract. 2011;28(1):110-117. doi:10.1093/fampra/cmq067
- Myers MG, Valdivieso M, Chessman M, Kiss A. Can sphygmomanometers designed for self-measurement of blood pressure in the home be used in office practice? Blood Press Monit. 2010;15(6):300-304. doi:10.1097/MBP.0b013e328340d128
- Leung AA, Nerenberg K, Daskalopoulou SS, et al. Hypertension Canada’s 2016 Canadian hypertension education program guidelines for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension. Can J Cardiol. 2016;32(5):569-588. doi:10.1016/j.cjca.2016.02.066
- Myers MG. A short history of automated office blood pressure - 15 years to SPRINT. J Clin Hypertens (Greenwich). 2016;18(8):721-724. doi:10.1111/jch.12820
- Myers MG, Kaczorowski J, Dawes M, Godwin M. Automated office blood pressure measurement in primary care. Can Fam Physician. 2014;60(2):127-132.
- Armstrong D, Matangi M, Brouillard D, Myers MG. Automated office blood pressure - being alone and not location is what matters most. Blood Press Monit. 2015;20(4):204-208. doi:10.1097/MBP.0000000000000133
- Yarows SA. What is the Cost of Measuring a Blood Pressure? Ann Clin Hypertens. 2018;2:59-66. doi:10.29328/journal.ach.1001012
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA. 1999;282(15):1458-1465. doi:10.1001/jama.282.15.1458
- Doane J, Buu J, Penrod MJ, et al. Measuring and managing blood pressure in a primary care setting: a pragmatic implementation study. J Am Board Fam Med. 2018;31(3):375-388. doi:10.3122/jabfm.2018.03.170450
- Cheng S, Claggett B, Correia AW, et al. Temporal Trends in the Population Attributable Risk for Cardiovascular Disease: The Atherosclerosis Risk in Communities Study. Circulation. 2014;130:820-828. doi.org/10.1161/CIRCULATIONAHA.113.008506
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):1269-1324. doi:10.1161/HYP.0000000000000066
- Leung AA, Daskalopoulou SS, Dasgupta K, et al. Hypertension Canada’s 2017 guidelines for diagnosis, risk assessment, prevention, and treatment of hypertension in adults. Can J Cardiol. 2017;33(5):557-576. doi:10.1016/j.cjca.2017.03.005
- Williams B, Mancia G, Spiering W, et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J. 2018;39(33):3021-3104. doi:10.1093/eurheartj/ehy339
- Pappaccogli M, Di Monaco S, Perlo E, et al. Comparison of automated office blood pressure with office and out-off-office measurement techniques. Hypertension. 2019;73(2):481-490. doi:10.1161/HYPERTENSIONAHA.118.12079
- Roerecke M, Kaczorowski J, Myers MG. Comparing automated office blood pressure readings with other methods of blood pressure measurement for identifying patients with possible hypertension - a systematic review and meta-analysis. JAMA Intern Med. 2019;179:351-362. doi:10.1001/jamainternmed.2018.6551
- Kaczorowski J, Chambers LW, Karwalajtys T, et al. Cardiovascular Health Awareness Program (CHAP): a community cluster-randomised trial among elderly Canadians. Prev Med. 2008;46(6):537-544. doi:10.1016/j.ypmed.2008.02.005
- SPRINT Research Group. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103-2116. doi:10.1056/NEJMoa1511939
- Andreadis EA, Agaliotis GD, Angelopoulos ET, et al. Automated office blood pressure and 24-h ambulatory measurements are equally associated with left ventricular mass index. Am J Hypertens. 2011;24(6):661-666. doi:10.1038/ajh.2011.38
- Campbell NRC, McKay DW, Conradson H, et al. Automated oscillometric blood pressure versus auscultatory blood pressure as a predictor of carotid intima-medial thickness in male firefighters. J Hum Hypertens. 2007;21(7):588-590. doi:10.1038/sj.jhh.1002190
- Myers MG, Godwin M, Dawes M et al. Conventional versus automated measurement of blood pressure in primary care patients with systolic hypertension: randomised parallel design controlled trial. BMJ. 2011;342:d286. doi:10.1136/bmj.d286
- Beckett L, Godwin M. The BpTRU automatic blood pressure monitor compared to 24 hour ambulatory blood pressure monitoring in the assessment of blood pressure in patients with hypertension. BMC Cardiovasc Disord. 2005;5(1):18. doi:10.1186/1471-2261-5-18
- Myers MG, Valdivieso M, Kiss A. Use of automated office blood pressure measurement to reduce the white coat response. J Hypertens. 2009;27(2):280-286. doi:10.1097/HJH.0b013e32831b9e6b
- Myers MG, Valdivieso M, Kiss A. Consistent relationship between automated office blood pressure recorded in different settings. Blood Press Monit. 2009;14(3):108-111. doi:10.1097/MBP.0b013e32832c5167
- Myers MG, Valdivieso M, Kiss A. Optimum frequency of office blood pressure measurement using an automated sphygmomanometer. Blood Press Monit. 2008;13(6):333-338. doi:10.1097/MBP.0b013e3283104247
- Myers MG. A proposed algorithm for diagnosing hypertension using automated office blood pressure measurement. J Hypertens. 2010;28(4):703-708. doi:10.1097/HJH.0b013e328335d091
- Godwin M, Birtwhistle R, Delva D, et al. Manual and automated office measurements in relation to awake ambulatory blood pressure monitoring. Fam Pract. 2011;28(1):110-117. doi:10.1093/fampra/cmq067
- Myers MG, Valdivieso M, Chessman M, Kiss A. Can sphygmomanometers designed for self-measurement of blood pressure in the home be used in office practice? Blood Press Monit. 2010;15(6):300-304. doi:10.1097/MBP.0b013e328340d128
- Leung AA, Nerenberg K, Daskalopoulou SS, et al. Hypertension Canada’s 2016 Canadian hypertension education program guidelines for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension. Can J Cardiol. 2016;32(5):569-588. doi:10.1016/j.cjca.2016.02.066
- Myers MG. A short history of automated office blood pressure - 15 years to SPRINT. J Clin Hypertens (Greenwich). 2016;18(8):721-724. doi:10.1111/jch.12820
- Myers MG, Kaczorowski J, Dawes M, Godwin M. Automated office blood pressure measurement in primary care. Can Fam Physician. 2014;60(2):127-132.
- Armstrong D, Matangi M, Brouillard D, Myers MG. Automated office blood pressure - being alone and not location is what matters most. Blood Press Monit. 2015;20(4):204-208. doi:10.1097/MBP.0000000000000133
- Yarows SA. What is the Cost of Measuring a Blood Pressure? Ann Clin Hypertens. 2018;2:59-66. doi:10.29328/journal.ach.1001012
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA. 1999;282(15):1458-1465. doi:10.1001/jama.282.15.1458
- Doane J, Buu J, Penrod MJ, et al. Measuring and managing blood pressure in a primary care setting: a pragmatic implementation study. J Am Board Fam Med. 2018;31(3):375-388. doi:10.3122/jabfm.2018.03.170450
Assessment of Automated vs Conventional Blood Pressure Measurements in a Veterans Affairs Clinical Practice Setting
Assessment of Automated vs Conventional Blood Pressure Measurements in a Veterans Affairs Clinical Practice Setting
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.
Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.
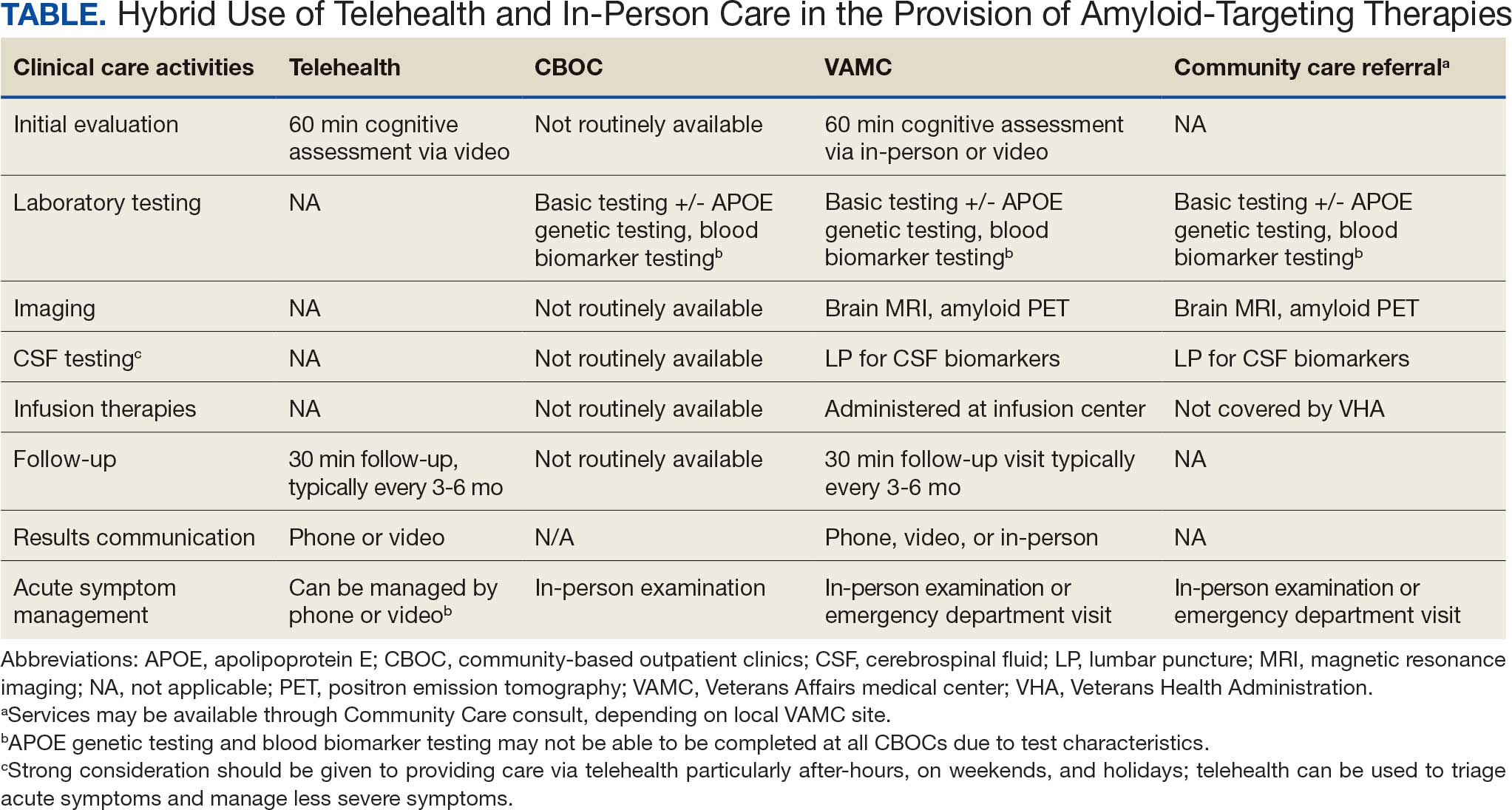
The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Due to the underlying mechanism of atrial fibrillation (Afib), clots can form within the left atrial appendage. Clots that become dislodged may lead to ischemic stroke and possibly death. The 2023 guidelines for atrial fibrillation from the American College of Cardiology and American Heart Association recommend anticoagulation therapy for patients with an Afib diagnosis and a CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke/vascular disease, age 65 to 74 years, and female sex) score pertinent for ≥ 1 non–sex-related factor (score ≥ 2 for women; ≥ 1 for men) to prevent stroke-related complications. The CHA2DS2-VASc score is a 9-point scoring tool based on comorbidities and conditions that increase risk of stroke in patients with Afib. Each value correlates to an annualized stroke risk percentage that increases as the score increases.
In clinical practice, patients meeting these thresholds are indicated for anticoagulation and are considered for indefinite use unless ≥ 1 of the following conditions are present: bleeding risk outweighs the stroke prevention benefit, Afib is episodic (< 48 hours) or a nonpharmacologic intervention, such as a left atrial appendage occlusion (LAAO) device is present.1
In patients with a diagnosed venous thromboembolism (VTE), such as deep vein thrombosis or pulmonary embolism, anticoagulation is used to treat the current thrombosis and prevent embolization that can ultimately lead to death. The 2021 guideline for VTE from the American College of Chest Physicians identifies certain risk factors that increase risk for VTE and categorizes them as transient or persistent. Transient risk factors include hospitalization > 3 days, major trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel > 8 hours. Persistent risk factors include malignancy, thrombophilia, and certain medications.
The guideline recommends therapy durations based on event frequency, the presence and classification of provoking risk factors, and bleeding risk. As the risk of recurrent thrombosis and other potential complications is greatest in the first 3 to 6 months after a diagnosed event, at least 3 months anticoagulation therapy is recommended following VTE diagnosis. At the 3-month mark, all regimens are suggested to be re-evaluated and considered for extended treatment duration if the event was unprovoked, recurrent, secondary to a persistent risk factor, or low bleed risk.2Anticoagulation is an important guideline-recommended pharmacologic intervention for various disease states, although its use is not without risks. The Institute for Safe Medication Practices has classified oral anticoagulants as high-alert medications. This designation was made because anticoagulant medications have the potential to cause harm when used or omitted in error and lead to life-threatening bleed or thrombotic complications.3Anticoagulation stewardship ensures that anticoagulation therapy is appropriately initiated, maintained, and discontinued when indicated. Because of the potential for harm, anticoagulation stewardship is an important part of Afib and VTE management. Pharmacists can help verify and evaluate anticoagulation therapies. Research suggests that pharmacist-led anticoagulation stewardship efforts may play a role in ensuring safer patient outcomes.4The purpose of this quality improvement (QI) study was to implement pharmacist-led anticoagulation stewardship practices at Veterans Affairs Phoenix Health Care System (VAPHCS) to identify veterans with Afib not currently on anticoagulation, as well as to identify veterans with a history of VTE events who have completed a sufficient treatment duration.
Methods
Anticoagulation stewardship efforts were implemented in 2 cohorts of patients: those with Afib who may be indicated to initiate anticoagulation, and those with a history of VTE events who may be indicated to consider anticoagulation discontinuation. Patient records were reviewed using a standardized note template, and recommendations to either initiate or discontinue anticoagulation therapy were documented. The VAPHCS Research Service reviewed this study and determined that it was not research and was exempt from institutional review board review.
Atrial Fibrillation Cohort
A population health dashboard created by the Stroke Prevention in Atrial Fibrillation/Flutter Targeting the uNTreated: a focus on health care disparities (SPAFF-TNT-D) national VA study team was used to identify veterans at VAPHCS with a diagnosis of Afib without an active VA prescription for an anticoagulant. The dashboard filtered and produced data points from the medical record that correlated to the components of the CHA2DS2-VASc score. All veterans identified by the dashboard with scores of 7 or 8 were included. No patients had a score of 9. Comprehensive chart reviews of available VA and non–VA-provided care records were conducted by the investigators, and a standardized note template designed by the SPAFF-TNT-D team (eAppendix 1) was used to document findings within the electronic health record (EHR). If anticoagulation was deemed to be indicated, the assigned primary care practitioner (PCP) as listed in the EHR was alerted to the note by the investigators for further evaluation and consideration of prescribing anticoagulation.
Venous Thromboembolism Cohort
VAPHCS pharmacy informatics pulled data that included veterans with documented VTE and an active VA anticoagulant prescription between November 2022 and November 2023. Veterans were reviewed in chronological order based on when the anticoagulant prescription was written. All veterans were included until an equal number of charts were reviewed in both the Afib and VTE cohorts. Comprehensive chart review of available VA- and non–VA-provided care records was conducted by the investigators, and a standardized note template as designed by the investigators (eAppendix 2) was used to document findings within the EHR. If the duration of anticoagulation therapy was deemed sufficient, the assigned anticoagulation clinical pharmacist practitioner (CPP) was alerted to the note by the investigators for further evaluation and consideration of discontinuing anticoagulation.
EHR reviews were conducted in October and November 2023 and lasted about 10 to 20 minutes per patient. To evaluate completeness and accuracy of the documented findings within the EHR, both investigators reviewed and cosigned the completed note template and verified the correct PCP was alerted to the recommendation for appropriate continuity of care. Results were reviewed in March 2024.
Outcomes
Atrial fibrillation cohort. The primary outcome was the number of veterans with Afib who were recommended to start anticoagulation therapy. Additional outcomes evaluated included the number of interventions completed, action taken by PCPs in response to the provided recommendation, and reasons provided by the investigators for not recommending initiation of anticoagulation therapy in specific veteran cases.
Venous thromboembolism cohort. The primary outcome was the number of veterans with a history of VTE events recommended to discontinue anticoagulation therapy. Additional outcomes included number of interventions completed, action taken by the anticoagulation CPP in response to the provided recommendation, and reasons provided by the investigators for not recommending discontinuation of anticoagulation therapy in specific veteran cases.
Analysis
Sample size was determined by the inclusion criteria and was not designed to attain statistical power. Data embedded in the Afib cohort standardized note template, also known as health factors, were later used for data analysis. Recommendations in the VTE cohort were manually tracked and recorded by the investigators. Results for this study were analyzed using descriptive statistics.
Results
A total of 114 veterans were reviewed and included in this study: 57 in each cohort. Seven recommendations were made regarding anticoagulation initiation for patients with Afib and 7 were made for anticoagulation discontinuation for patients with VTE (Table 1).
In the Afib cohort, 1 veteran was successfully initiated on anticoagulation therapy and 1 veteran was deemed appropriate for initiation of anticoagulation but was not reachable. Of the 5 recommendations with no action taken, 4 PCPs acknowledged the alert with no further documentation, and 1 PCP deferred the decision to cardiology with no further documentation. In the VTE cohort, 3 veterans successfully discontinued anticoagulation therapy and 2 veterans were further evaluated by the anticoagulation CPP and deemed appropriate to continue therapy based on potential for malignancy. Of the 2 recommendations with no action taken, 1 anticoagulation CPP acknowledged the alert with no further documentation and 1 anticoagulation CPP suggested further evaluation by PCP with no further documentation.
In the Afib cohort, a nonpharmacologic approach was defined as documentation of a LAAO device. An inaccurate diagnosis was defined as an Afib diagnosis being used in a previous visit, although there was no further confirmation of diagnosis via chart review. Veterans classified as already being on anticoagulation had documentation of non–VA-written anticoagulant prescriptions or receiving a supply of anticoagulants from a facility such as a nursing home. Anticoagulation was defined as unfavorable if a documented risk/benefit conversation was found via EHR review. Anticoagulation was defined as not indicated if the Afib was documented as transient, episodic, or historical (Table 2).
In the VTE cohort, no recommendations for discontinuation were made for veterans indicated to continue anticoagulation due to a concurrent Afib diagnosis. Chronic or recurrent events were defined as documentation of multiple VTE events and associated dates in the EHR. Persistent risk factors included malignancy or medications contributing to hypercoagulable states. Thrombophilia was defined as having documentation of a diagnosis in the EHR. An unprovoked event was defined as VTE without any documented transient risk factors (eg, hospitalization, trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel). Anticoagulation had already been discontinued in 1 veteran after the data were collected but before chart review occurred (Table 3).

Discussion
Pharmacy-led indication reviews resulted in appropriate recommendations for anticoagulation use in veterans with Afib and a history of VTE events. Overall, 12.3% of chart reviews in each cohort resulted in a recommendation being made, which was similar to the rate found by Koolian et al.5 In that study, 10% of recommendations were related to initiation or interruption of anticoagulation. This recommendation category consisted of several subcategories, including “suggesting therapeutic anticoagulation when none is currently ordered” and “suggesting anticoagulation cessation if no longer indicated,” but specific numerical prevalence was not provided.5
Online dashboard use allowed for greater population health management and identification of veterans with Afib who were not on active anticoagulation, providing opportunities to prevent stroke-related complications. Wang et al completed a similarly designed study that included a population health tool to identify patients with Afib who were not on anticoagulation and implemented pharmacist-led chart review and facilitation of recommendations to the responsible clinician. This study reviewed 1727 patients and recommended initiation of anticoagulation therapy for 75 (4.3%).6 The current study had a higher percentage of patients with recommendations for changes despite its smaller size.
Evaluating the duration of therapy for anticoagulation in veterans with a history of VTE events provided an opportunity to reduce unnecessary exposure to anticoagulation and minimize bleeding risks. Using a chart review process and standardized note template enabled the documentation of pertinent information that could be readily reviewed by the PCP. This process is a step toward ensuring VAPHCS PCPs provide guideline-recommended care and actively prevent stroke and bleeding complications. Adoption of this process into the current VAPHCS Anticoagulation Clinic workflow for review of veterans with either Afib or VTE could lead to more EHRs being reviewed and recommendations made, ultimately improving patient outcomes.
Therapeutic interventions based on the recommendations were completed for 1 of 7 veterans (14%) and 3 of 7 veterans (43%) in the Afib and VTE cohorts, respectively. The prevalence of completed interventions in this anticoagulation stewardship study was higher than those in Wang et al, who found only 9% of their recommendations resulted in PCPs considering action related to anticoagulation, and only 4% were successfully initiated.6
In the Afib cohort, veterans identified by the dashboard with a CHA2DS2-VASc of 7 or 8 were prioritized for review. Reviewing these veterans ensured that patients with the highest stroke risk were sufficiently evaluated and started on anticoagulation as needed to reduce stroke-related complications. In contrast, because these veterans had higher CHA2DS2-VASc scores, they may have already been evaluated for anticoagulation in the past and had a documented rationale for not being placed on anticoagulation (LAAO device placement was the most common rationale). Focusing on veterans with a lower CHA2DS2-VASc score such as 1 for men or 2 for women could potentially include more opportunities for recommendations. Although stroke risk may be lower in this population compared with those with higher CHA2DS2-VASc scores, guideline-recommended anticoagulation use may be missed for these patients.
In the VTE cohort, veterans with an anticoagulant prescription written 12 months before data collection were prioritized for review. Reviewing these veterans ensured that anticoagulation therapy met guideline recommendations of at least 3 months, with potential for extended duration upon further evaluation by a provider at that time. Based on collected results, most veterans were already reevaluated and had documented reasons why anticoagulation was still indicated; concurrent Afib was most common followed by chronic or recurrent VTE. Reviewing veterans with more recent prescriptions just over the recommended 3-month duration could potentially include more opportunities for recommendations to be made. It is more likely that by 3 months another PCP had not already weighed in on the duration of therapy, and the anticoagulation CPP could ensure a thorough review is conducted with guideline-based recommendations.
Most published literature on anticoagulation stewardship efforts is focused on inpatient management and policy changes, or concentrate on attributes of therapy such as appropriate dosing and drug interactions. This study highlighted that gaps in care related to anticoagulation use and discontinuation are present in the VAPHCS population and can be appropriately addressed via pharmacist-led indication reviews. Future studies designed to focus on initiating anticoagulation where appropriate, and discontinuing where a sufficient treatment period has been completed, are warranted to minimize this gap in care and allow health systems to work toward process changes to ensure safe and optimized care is provided for the patients they serve.
Limitations
In the Afib cohort, 5 of 7 recommendations (71%) had no further action taken by the PCP, which may represent a barrier to care. In contrast, 2 of 7 recommendations (29%) had no further action in the VTE cohort. It is possible that the difference can be attributed to the anticoagulation CPP receiving VTE alerts and PCPs receiving Afib alerts. The anticoagulation CPP was familiar with this QI study and may have better understood the purpose of the chart review and the need to provide a timely response. PCPs may have been less likely to take action because they were unfamiliar with the anticoagulation stewardship initiative and standardized note template or overwhelmed by too many EHR alerts.
The lack of PCP response to a virtual alert or message also was observed by Wang et al, whereas Koolian et al reported higher intervention completion rates, with verbal recommendations being made to the responsible clinicians. To further ensure these pertinent recommendations for anticoagulation initiation in veterans with Afib are properly reviewed and evaluated, future research could include intentional follow-up with the PCP regarding the alert, PCP-specific education about the anticoagulation stewardship initiative and the role of the standardized note template, and collaboration with PCPs to identify alternative ways to relay recommendations in a way that would ensure the completion of appropriate and timely review.
Conclusions
This study identified gaps in care related to anticoagulation needs in the VAPHCS veteran population. Utilizing a standardized indication review process allows pharmacists to evaluate anticoagulant use for both appropriate indication and duration of therapy. Providing recommendations via chart review notes and alerting respective PCPs and CPPs results in veterans receiving safe and optimized care regarding their anticoagulation needs.
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149:e1-e156. doi:10.1161/CIR.0000000000001193
- Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160:e545-e608. doi:10.1016/j.chest.2021.07.055
- Institute for Safe Medication Practices (ISMP). List of high-alert medications in community/ambulatory care settings. ISMP. September 30, 2021. Accessed September 11, 2025. https://home.ecri.org/blogs/ismp-resources/high-alert-medications-in-community-ambulatory-care-settings
- Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6:e12757. doi:10.1002/rth2.12757
- Koolian M, Wiseman D, Mantzanis H, et al. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6:e12758. doi:10.1002/rth2.12758
- Wang SV, Rogers JR, Jin Y, et al. Stepped-wedge randomised trial to evaluate population health intervention designed to increase appropriate anticoagulation in patients with atrial fibrillation. BMJ Qual Saf. 2019;28:835-842. doi:10.1136/bmjqs-2019-009367
Due to the underlying mechanism of atrial fibrillation (Afib), clots can form within the left atrial appendage. Clots that become dislodged may lead to ischemic stroke and possibly death. The 2023 guidelines for atrial fibrillation from the American College of Cardiology and American Heart Association recommend anticoagulation therapy for patients with an Afib diagnosis and a CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke/vascular disease, age 65 to 74 years, and female sex) score pertinent for ≥ 1 non–sex-related factor (score ≥ 2 for women; ≥ 1 for men) to prevent stroke-related complications. The CHA2DS2-VASc score is a 9-point scoring tool based on comorbidities and conditions that increase risk of stroke in patients with Afib. Each value correlates to an annualized stroke risk percentage that increases as the score increases.
In clinical practice, patients meeting these thresholds are indicated for anticoagulation and are considered for indefinite use unless ≥ 1 of the following conditions are present: bleeding risk outweighs the stroke prevention benefit, Afib is episodic (< 48 hours) or a nonpharmacologic intervention, such as a left atrial appendage occlusion (LAAO) device is present.1
In patients with a diagnosed venous thromboembolism (VTE), such as deep vein thrombosis or pulmonary embolism, anticoagulation is used to treat the current thrombosis and prevent embolization that can ultimately lead to death. The 2021 guideline for VTE from the American College of Chest Physicians identifies certain risk factors that increase risk for VTE and categorizes them as transient or persistent. Transient risk factors include hospitalization > 3 days, major trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel > 8 hours. Persistent risk factors include malignancy, thrombophilia, and certain medications.
The guideline recommends therapy durations based on event frequency, the presence and classification of provoking risk factors, and bleeding risk. As the risk of recurrent thrombosis and other potential complications is greatest in the first 3 to 6 months after a diagnosed event, at least 3 months anticoagulation therapy is recommended following VTE diagnosis. At the 3-month mark, all regimens are suggested to be re-evaluated and considered for extended treatment duration if the event was unprovoked, recurrent, secondary to a persistent risk factor, or low bleed risk.2Anticoagulation is an important guideline-recommended pharmacologic intervention for various disease states, although its use is not without risks. The Institute for Safe Medication Practices has classified oral anticoagulants as high-alert medications. This designation was made because anticoagulant medications have the potential to cause harm when used or omitted in error and lead to life-threatening bleed or thrombotic complications.3Anticoagulation stewardship ensures that anticoagulation therapy is appropriately initiated, maintained, and discontinued when indicated. Because of the potential for harm, anticoagulation stewardship is an important part of Afib and VTE management. Pharmacists can help verify and evaluate anticoagulation therapies. Research suggests that pharmacist-led anticoagulation stewardship efforts may play a role in ensuring safer patient outcomes.4The purpose of this quality improvement (QI) study was to implement pharmacist-led anticoagulation stewardship practices at Veterans Affairs Phoenix Health Care System (VAPHCS) to identify veterans with Afib not currently on anticoagulation, as well as to identify veterans with a history of VTE events who have completed a sufficient treatment duration.
Methods
Anticoagulation stewardship efforts were implemented in 2 cohorts of patients: those with Afib who may be indicated to initiate anticoagulation, and those with a history of VTE events who may be indicated to consider anticoagulation discontinuation. Patient records were reviewed using a standardized note template, and recommendations to either initiate or discontinue anticoagulation therapy were documented. The VAPHCS Research Service reviewed this study and determined that it was not research and was exempt from institutional review board review.
Atrial Fibrillation Cohort
A population health dashboard created by the Stroke Prevention in Atrial Fibrillation/Flutter Targeting the uNTreated: a focus on health care disparities (SPAFF-TNT-D) national VA study team was used to identify veterans at VAPHCS with a diagnosis of Afib without an active VA prescription for an anticoagulant. The dashboard filtered and produced data points from the medical record that correlated to the components of the CHA2DS2-VASc score. All veterans identified by the dashboard with scores of 7 or 8 were included. No patients had a score of 9. Comprehensive chart reviews of available VA and non–VA-provided care records were conducted by the investigators, and a standardized note template designed by the SPAFF-TNT-D team (eAppendix 1) was used to document findings within the electronic health record (EHR). If anticoagulation was deemed to be indicated, the assigned primary care practitioner (PCP) as listed in the EHR was alerted to the note by the investigators for further evaluation and consideration of prescribing anticoagulation.
Venous Thromboembolism Cohort
VAPHCS pharmacy informatics pulled data that included veterans with documented VTE and an active VA anticoagulant prescription between November 2022 and November 2023. Veterans were reviewed in chronological order based on when the anticoagulant prescription was written. All veterans were included until an equal number of charts were reviewed in both the Afib and VTE cohorts. Comprehensive chart review of available VA- and non–VA-provided care records was conducted by the investigators, and a standardized note template as designed by the investigators (eAppendix 2) was used to document findings within the EHR. If the duration of anticoagulation therapy was deemed sufficient, the assigned anticoagulation clinical pharmacist practitioner (CPP) was alerted to the note by the investigators for further evaluation and consideration of discontinuing anticoagulation.
EHR reviews were conducted in October and November 2023 and lasted about 10 to 20 minutes per patient. To evaluate completeness and accuracy of the documented findings within the EHR, both investigators reviewed and cosigned the completed note template and verified the correct PCP was alerted to the recommendation for appropriate continuity of care. Results were reviewed in March 2024.
Outcomes
Atrial fibrillation cohort. The primary outcome was the number of veterans with Afib who were recommended to start anticoagulation therapy. Additional outcomes evaluated included the number of interventions completed, action taken by PCPs in response to the provided recommendation, and reasons provided by the investigators for not recommending initiation of anticoagulation therapy in specific veteran cases.
Venous thromboembolism cohort. The primary outcome was the number of veterans with a history of VTE events recommended to discontinue anticoagulation therapy. Additional outcomes included number of interventions completed, action taken by the anticoagulation CPP in response to the provided recommendation, and reasons provided by the investigators for not recommending discontinuation of anticoagulation therapy in specific veteran cases.
Analysis
Sample size was determined by the inclusion criteria and was not designed to attain statistical power. Data embedded in the Afib cohort standardized note template, also known as health factors, were later used for data analysis. Recommendations in the VTE cohort were manually tracked and recorded by the investigators. Results for this study were analyzed using descriptive statistics.
Results
A total of 114 veterans were reviewed and included in this study: 57 in each cohort. Seven recommendations were made regarding anticoagulation initiation for patients with Afib and 7 were made for anticoagulation discontinuation for patients with VTE (Table 1).

In the Afib cohort, 1 veteran was successfully initiated on anticoagulation therapy and 1 veteran was deemed appropriate for initiation of anticoagulation but was not reachable. Of the 5 recommendations with no action taken, 4 PCPs acknowledged the alert with no further documentation, and 1 PCP deferred the decision to cardiology with no further documentation. In the VTE cohort, 3 veterans successfully discontinued anticoagulation therapy and 2 veterans were further evaluated by the anticoagulation CPP and deemed appropriate to continue therapy based on potential for malignancy. Of the 2 recommendations with no action taken, 1 anticoagulation CPP acknowledged the alert with no further documentation and 1 anticoagulation CPP suggested further evaluation by PCP with no further documentation.
In the Afib cohort, a nonpharmacologic approach was defined as documentation of a LAAO device. An inaccurate diagnosis was defined as an Afib diagnosis being used in a previous visit, although there was no further confirmation of diagnosis via chart review. Veterans classified as already being on anticoagulation had documentation of non–VA-written anticoagulant prescriptions or receiving a supply of anticoagulants from a facility such as a nursing home. Anticoagulation was defined as unfavorable if a documented risk/benefit conversation was found via EHR review. Anticoagulation was defined as not indicated if the Afib was documented as transient, episodic, or historical (Table 2).

In the VTE cohort, no recommendations for discontinuation were made for veterans indicated to continue anticoagulation due to a concurrent Afib diagnosis. Chronic or recurrent events were defined as documentation of multiple VTE events and associated dates in the EHR. Persistent risk factors included malignancy or medications contributing to hypercoagulable states. Thrombophilia was defined as having documentation of a diagnosis in the EHR. An unprovoked event was defined as VTE without any documented transient risk factors (eg, hospitalization, trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel). Anticoagulation had already been discontinued in 1 veteran after the data were collected but before chart review occurred (Table 3).

Discussion
Pharmacy-led indication reviews resulted in appropriate recommendations for anticoagulation use in veterans with Afib and a history of VTE events. Overall, 12.3% of chart reviews in each cohort resulted in a recommendation being made, which was similar to the rate found by Koolian et al.5 In that study, 10% of recommendations were related to initiation or interruption of anticoagulation. This recommendation category consisted of several subcategories, including “suggesting therapeutic anticoagulation when none is currently ordered” and “suggesting anticoagulation cessation if no longer indicated,” but specific numerical prevalence was not provided.5
Online dashboard use allowed for greater population health management and identification of veterans with Afib who were not on active anticoagulation, providing opportunities to prevent stroke-related complications. Wang et al completed a similarly designed study that included a population health tool to identify patients with Afib who were not on anticoagulation and implemented pharmacist-led chart review and facilitation of recommendations to the responsible clinician. This study reviewed 1727 patients and recommended initiation of anticoagulation therapy for 75 (4.3%).6 The current study had a higher percentage of patients with recommendations for changes despite its smaller size.
Evaluating the duration of therapy for anticoagulation in veterans with a history of VTE events provided an opportunity to reduce unnecessary exposure to anticoagulation and minimize bleeding risks. Using a chart review process and standardized note template enabled the documentation of pertinent information that could be readily reviewed by the PCP. This process is a step toward ensuring VAPHCS PCPs provide guideline-recommended care and actively prevent stroke and bleeding complications. Adoption of this process into the current VAPHCS Anticoagulation Clinic workflow for review of veterans with either Afib or VTE could lead to more EHRs being reviewed and recommendations made, ultimately improving patient outcomes.
Therapeutic interventions based on the recommendations were completed for 1 of 7 veterans (14%) and 3 of 7 veterans (43%) in the Afib and VTE cohorts, respectively. The prevalence of completed interventions in this anticoagulation stewardship study was higher than those in Wang et al, who found only 9% of their recommendations resulted in PCPs considering action related to anticoagulation, and only 4% were successfully initiated.6
In the Afib cohort, veterans identified by the dashboard with a CHA2DS2-VASc of 7 or 8 were prioritized for review. Reviewing these veterans ensured that patients with the highest stroke risk were sufficiently evaluated and started on anticoagulation as needed to reduce stroke-related complications. In contrast, because these veterans had higher CHA2DS2-VASc scores, they may have already been evaluated for anticoagulation in the past and had a documented rationale for not being placed on anticoagulation (LAAO device placement was the most common rationale). Focusing on veterans with a lower CHA2DS2-VASc score such as 1 for men or 2 for women could potentially include more opportunities for recommendations. Although stroke risk may be lower in this population compared with those with higher CHA2DS2-VASc scores, guideline-recommended anticoagulation use may be missed for these patients.
In the VTE cohort, veterans with an anticoagulant prescription written 12 months before data collection were prioritized for review. Reviewing these veterans ensured that anticoagulation therapy met guideline recommendations of at least 3 months, with potential for extended duration upon further evaluation by a provider at that time. Based on collected results, most veterans were already reevaluated and had documented reasons why anticoagulation was still indicated; concurrent Afib was most common followed by chronic or recurrent VTE. Reviewing veterans with more recent prescriptions just over the recommended 3-month duration could potentially include more opportunities for recommendations to be made. It is more likely that by 3 months another PCP had not already weighed in on the duration of therapy, and the anticoagulation CPP could ensure a thorough review is conducted with guideline-based recommendations.
Most published literature on anticoagulation stewardship efforts is focused on inpatient management and policy changes, or concentrate on attributes of therapy such as appropriate dosing and drug interactions. This study highlighted that gaps in care related to anticoagulation use and discontinuation are present in the VAPHCS population and can be appropriately addressed via pharmacist-led indication reviews. Future studies designed to focus on initiating anticoagulation where appropriate, and discontinuing where a sufficient treatment period has been completed, are warranted to minimize this gap in care and allow health systems to work toward process changes to ensure safe and optimized care is provided for the patients they serve.
Limitations
In the Afib cohort, 5 of 7 recommendations (71%) had no further action taken by the PCP, which may represent a barrier to care. In contrast, 2 of 7 recommendations (29%) had no further action in the VTE cohort. It is possible that the difference can be attributed to the anticoagulation CPP receiving VTE alerts and PCPs receiving Afib alerts. The anticoagulation CPP was familiar with this QI study and may have better understood the purpose of the chart review and the need to provide a timely response. PCPs may have been less likely to take action because they were unfamiliar with the anticoagulation stewardship initiative and standardized note template or overwhelmed by too many EHR alerts.
The lack of PCP response to a virtual alert or message also was observed by Wang et al, whereas Koolian et al reported higher intervention completion rates, with verbal recommendations being made to the responsible clinicians. To further ensure these pertinent recommendations for anticoagulation initiation in veterans with Afib are properly reviewed and evaluated, future research could include intentional follow-up with the PCP regarding the alert, PCP-specific education about the anticoagulation stewardship initiative and the role of the standardized note template, and collaboration with PCPs to identify alternative ways to relay recommendations in a way that would ensure the completion of appropriate and timely review.
Conclusions
This study identified gaps in care related to anticoagulation needs in the VAPHCS veteran population. Utilizing a standardized indication review process allows pharmacists to evaluate anticoagulant use for both appropriate indication and duration of therapy. Providing recommendations via chart review notes and alerting respective PCPs and CPPs results in veterans receiving safe and optimized care regarding their anticoagulation needs.
Due to the underlying mechanism of atrial fibrillation (Afib), clots can form within the left atrial appendage. Clots that become dislodged may lead to ischemic stroke and possibly death. The 2023 guidelines for atrial fibrillation from the American College of Cardiology and American Heart Association recommend anticoagulation therapy for patients with an Afib diagnosis and a CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke/vascular disease, age 65 to 74 years, and female sex) score pertinent for ≥ 1 non–sex-related factor (score ≥ 2 for women; ≥ 1 for men) to prevent stroke-related complications. The CHA2DS2-VASc score is a 9-point scoring tool based on comorbidities and conditions that increase risk of stroke in patients with Afib. Each value correlates to an annualized stroke risk percentage that increases as the score increases.
In clinical practice, patients meeting these thresholds are indicated for anticoagulation and are considered for indefinite use unless ≥ 1 of the following conditions are present: bleeding risk outweighs the stroke prevention benefit, Afib is episodic (< 48 hours) or a nonpharmacologic intervention, such as a left atrial appendage occlusion (LAAO) device is present.1
In patients with a diagnosed venous thromboembolism (VTE), such as deep vein thrombosis or pulmonary embolism, anticoagulation is used to treat the current thrombosis and prevent embolization that can ultimately lead to death. The 2021 guideline for VTE from the American College of Chest Physicians identifies certain risk factors that increase risk for VTE and categorizes them as transient or persistent. Transient risk factors include hospitalization > 3 days, major trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel > 8 hours. Persistent risk factors include malignancy, thrombophilia, and certain medications.
The guideline recommends therapy durations based on event frequency, the presence and classification of provoking risk factors, and bleeding risk. As the risk of recurrent thrombosis and other potential complications is greatest in the first 3 to 6 months after a diagnosed event, at least 3 months anticoagulation therapy is recommended following VTE diagnosis. At the 3-month mark, all regimens are suggested to be re-evaluated and considered for extended treatment duration if the event was unprovoked, recurrent, secondary to a persistent risk factor, or low bleed risk.2Anticoagulation is an important guideline-recommended pharmacologic intervention for various disease states, although its use is not without risks. The Institute for Safe Medication Practices has classified oral anticoagulants as high-alert medications. This designation was made because anticoagulant medications have the potential to cause harm when used or omitted in error and lead to life-threatening bleed or thrombotic complications.3Anticoagulation stewardship ensures that anticoagulation therapy is appropriately initiated, maintained, and discontinued when indicated. Because of the potential for harm, anticoagulation stewardship is an important part of Afib and VTE management. Pharmacists can help verify and evaluate anticoagulation therapies. Research suggests that pharmacist-led anticoagulation stewardship efforts may play a role in ensuring safer patient outcomes.4The purpose of this quality improvement (QI) study was to implement pharmacist-led anticoagulation stewardship practices at Veterans Affairs Phoenix Health Care System (VAPHCS) to identify veterans with Afib not currently on anticoagulation, as well as to identify veterans with a history of VTE events who have completed a sufficient treatment duration.
Methods
Anticoagulation stewardship efforts were implemented in 2 cohorts of patients: those with Afib who may be indicated to initiate anticoagulation, and those with a history of VTE events who may be indicated to consider anticoagulation discontinuation. Patient records were reviewed using a standardized note template, and recommendations to either initiate or discontinue anticoagulation therapy were documented. The VAPHCS Research Service reviewed this study and determined that it was not research and was exempt from institutional review board review.
Atrial Fibrillation Cohort
A population health dashboard created by the Stroke Prevention in Atrial Fibrillation/Flutter Targeting the uNTreated: a focus on health care disparities (SPAFF-TNT-D) national VA study team was used to identify veterans at VAPHCS with a diagnosis of Afib without an active VA prescription for an anticoagulant. The dashboard filtered and produced data points from the medical record that correlated to the components of the CHA2DS2-VASc score. All veterans identified by the dashboard with scores of 7 or 8 were included. No patients had a score of 9. Comprehensive chart reviews of available VA and non–VA-provided care records were conducted by the investigators, and a standardized note template designed by the SPAFF-TNT-D team (eAppendix 1) was used to document findings within the electronic health record (EHR). If anticoagulation was deemed to be indicated, the assigned primary care practitioner (PCP) as listed in the EHR was alerted to the note by the investigators for further evaluation and consideration of prescribing anticoagulation.
Venous Thromboembolism Cohort
VAPHCS pharmacy informatics pulled data that included veterans with documented VTE and an active VA anticoagulant prescription between November 2022 and November 2023. Veterans were reviewed in chronological order based on when the anticoagulant prescription was written. All veterans were included until an equal number of charts were reviewed in both the Afib and VTE cohorts. Comprehensive chart review of available VA- and non–VA-provided care records was conducted by the investigators, and a standardized note template as designed by the investigators (eAppendix 2) was used to document findings within the EHR. If the duration of anticoagulation therapy was deemed sufficient, the assigned anticoagulation clinical pharmacist practitioner (CPP) was alerted to the note by the investigators for further evaluation and consideration of discontinuing anticoagulation.
EHR reviews were conducted in October and November 2023 and lasted about 10 to 20 minutes per patient. To evaluate completeness and accuracy of the documented findings within the EHR, both investigators reviewed and cosigned the completed note template and verified the correct PCP was alerted to the recommendation for appropriate continuity of care. Results were reviewed in March 2024.
Outcomes
Atrial fibrillation cohort. The primary outcome was the number of veterans with Afib who were recommended to start anticoagulation therapy. Additional outcomes evaluated included the number of interventions completed, action taken by PCPs in response to the provided recommendation, and reasons provided by the investigators for not recommending initiation of anticoagulation therapy in specific veteran cases.
Venous thromboembolism cohort. The primary outcome was the number of veterans with a history of VTE events recommended to discontinue anticoagulation therapy. Additional outcomes included number of interventions completed, action taken by the anticoagulation CPP in response to the provided recommendation, and reasons provided by the investigators for not recommending discontinuation of anticoagulation therapy in specific veteran cases.
Analysis
Sample size was determined by the inclusion criteria and was not designed to attain statistical power. Data embedded in the Afib cohort standardized note template, also known as health factors, were later used for data analysis. Recommendations in the VTE cohort were manually tracked and recorded by the investigators. Results for this study were analyzed using descriptive statistics.
Results
A total of 114 veterans were reviewed and included in this study: 57 in each cohort. Seven recommendations were made regarding anticoagulation initiation for patients with Afib and 7 were made for anticoagulation discontinuation for patients with VTE (Table 1).

In the Afib cohort, 1 veteran was successfully initiated on anticoagulation therapy and 1 veteran was deemed appropriate for initiation of anticoagulation but was not reachable. Of the 5 recommendations with no action taken, 4 PCPs acknowledged the alert with no further documentation, and 1 PCP deferred the decision to cardiology with no further documentation. In the VTE cohort, 3 veterans successfully discontinued anticoagulation therapy and 2 veterans were further evaluated by the anticoagulation CPP and deemed appropriate to continue therapy based on potential for malignancy. Of the 2 recommendations with no action taken, 1 anticoagulation CPP acknowledged the alert with no further documentation and 1 anticoagulation CPP suggested further evaluation by PCP with no further documentation.
In the Afib cohort, a nonpharmacologic approach was defined as documentation of a LAAO device. An inaccurate diagnosis was defined as an Afib diagnosis being used in a previous visit, although there was no further confirmation of diagnosis via chart review. Veterans classified as already being on anticoagulation had documentation of non–VA-written anticoagulant prescriptions or receiving a supply of anticoagulants from a facility such as a nursing home. Anticoagulation was defined as unfavorable if a documented risk/benefit conversation was found via EHR review. Anticoagulation was defined as not indicated if the Afib was documented as transient, episodic, or historical (Table 2).

In the VTE cohort, no recommendations for discontinuation were made for veterans indicated to continue anticoagulation due to a concurrent Afib diagnosis. Chronic or recurrent events were defined as documentation of multiple VTE events and associated dates in the EHR. Persistent risk factors included malignancy or medications contributing to hypercoagulable states. Thrombophilia was defined as having documentation of a diagnosis in the EHR. An unprovoked event was defined as VTE without any documented transient risk factors (eg, hospitalization, trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel). Anticoagulation had already been discontinued in 1 veteran after the data were collected but before chart review occurred (Table 3).

Discussion
Pharmacy-led indication reviews resulted in appropriate recommendations for anticoagulation use in veterans with Afib and a history of VTE events. Overall, 12.3% of chart reviews in each cohort resulted in a recommendation being made, which was similar to the rate found by Koolian et al.5 In that study, 10% of recommendations were related to initiation or interruption of anticoagulation. This recommendation category consisted of several subcategories, including “suggesting therapeutic anticoagulation when none is currently ordered” and “suggesting anticoagulation cessation if no longer indicated,” but specific numerical prevalence was not provided.5
Online dashboard use allowed for greater population health management and identification of veterans with Afib who were not on active anticoagulation, providing opportunities to prevent stroke-related complications. Wang et al completed a similarly designed study that included a population health tool to identify patients with Afib who were not on anticoagulation and implemented pharmacist-led chart review and facilitation of recommendations to the responsible clinician. This study reviewed 1727 patients and recommended initiation of anticoagulation therapy for 75 (4.3%).6 The current study had a higher percentage of patients with recommendations for changes despite its smaller size.
Evaluating the duration of therapy for anticoagulation in veterans with a history of VTE events provided an opportunity to reduce unnecessary exposure to anticoagulation and minimize bleeding risks. Using a chart review process and standardized note template enabled the documentation of pertinent information that could be readily reviewed by the PCP. This process is a step toward ensuring VAPHCS PCPs provide guideline-recommended care and actively prevent stroke and bleeding complications. Adoption of this process into the current VAPHCS Anticoagulation Clinic workflow for review of veterans with either Afib or VTE could lead to more EHRs being reviewed and recommendations made, ultimately improving patient outcomes.
Therapeutic interventions based on the recommendations were completed for 1 of 7 veterans (14%) and 3 of 7 veterans (43%) in the Afib and VTE cohorts, respectively. The prevalence of completed interventions in this anticoagulation stewardship study was higher than those in Wang et al, who found only 9% of their recommendations resulted in PCPs considering action related to anticoagulation, and only 4% were successfully initiated.6
In the Afib cohort, veterans identified by the dashboard with a CHA2DS2-VASc of 7 or 8 were prioritized for review. Reviewing these veterans ensured that patients with the highest stroke risk were sufficiently evaluated and started on anticoagulation as needed to reduce stroke-related complications. In contrast, because these veterans had higher CHA2DS2-VASc scores, they may have already been evaluated for anticoagulation in the past and had a documented rationale for not being placed on anticoagulation (LAAO device placement was the most common rationale). Focusing on veterans with a lower CHA2DS2-VASc score such as 1 for men or 2 for women could potentially include more opportunities for recommendations. Although stroke risk may be lower in this population compared with those with higher CHA2DS2-VASc scores, guideline-recommended anticoagulation use may be missed for these patients.
In the VTE cohort, veterans with an anticoagulant prescription written 12 months before data collection were prioritized for review. Reviewing these veterans ensured that anticoagulation therapy met guideline recommendations of at least 3 months, with potential for extended duration upon further evaluation by a provider at that time. Based on collected results, most veterans were already reevaluated and had documented reasons why anticoagulation was still indicated; concurrent Afib was most common followed by chronic or recurrent VTE. Reviewing veterans with more recent prescriptions just over the recommended 3-month duration could potentially include more opportunities for recommendations to be made. It is more likely that by 3 months another PCP had not already weighed in on the duration of therapy, and the anticoagulation CPP could ensure a thorough review is conducted with guideline-based recommendations.
Most published literature on anticoagulation stewardship efforts is focused on inpatient management and policy changes, or concentrate on attributes of therapy such as appropriate dosing and drug interactions. This study highlighted that gaps in care related to anticoagulation use and discontinuation are present in the VAPHCS population and can be appropriately addressed via pharmacist-led indication reviews. Future studies designed to focus on initiating anticoagulation where appropriate, and discontinuing where a sufficient treatment period has been completed, are warranted to minimize this gap in care and allow health systems to work toward process changes to ensure safe and optimized care is provided for the patients they serve.
Limitations
In the Afib cohort, 5 of 7 recommendations (71%) had no further action taken by the PCP, which may represent a barrier to care. In contrast, 2 of 7 recommendations (29%) had no further action in the VTE cohort. It is possible that the difference can be attributed to the anticoagulation CPP receiving VTE alerts and PCPs receiving Afib alerts. The anticoagulation CPP was familiar with this QI study and may have better understood the purpose of the chart review and the need to provide a timely response. PCPs may have been less likely to take action because they were unfamiliar with the anticoagulation stewardship initiative and standardized note template or overwhelmed by too many EHR alerts.
The lack of PCP response to a virtual alert or message also was observed by Wang et al, whereas Koolian et al reported higher intervention completion rates, with verbal recommendations being made to the responsible clinicians. To further ensure these pertinent recommendations for anticoagulation initiation in veterans with Afib are properly reviewed and evaluated, future research could include intentional follow-up with the PCP regarding the alert, PCP-specific education about the anticoagulation stewardship initiative and the role of the standardized note template, and collaboration with PCPs to identify alternative ways to relay recommendations in a way that would ensure the completion of appropriate and timely review.
Conclusions
This study identified gaps in care related to anticoagulation needs in the VAPHCS veteran population. Utilizing a standardized indication review process allows pharmacists to evaluate anticoagulant use for both appropriate indication and duration of therapy. Providing recommendations via chart review notes and alerting respective PCPs and CPPs results in veterans receiving safe and optimized care regarding their anticoagulation needs.
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149:e1-e156. doi:10.1161/CIR.0000000000001193
- Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160:e545-e608. doi:10.1016/j.chest.2021.07.055
- Institute for Safe Medication Practices (ISMP). List of high-alert medications in community/ambulatory care settings. ISMP. September 30, 2021. Accessed September 11, 2025. https://home.ecri.org/blogs/ismp-resources/high-alert-medications-in-community-ambulatory-care-settings
- Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6:e12757. doi:10.1002/rth2.12757
- Koolian M, Wiseman D, Mantzanis H, et al. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6:e12758. doi:10.1002/rth2.12758
- Wang SV, Rogers JR, Jin Y, et al. Stepped-wedge randomised trial to evaluate population health intervention designed to increase appropriate anticoagulation in patients with atrial fibrillation. BMJ Qual Saf. 2019;28:835-842. doi:10.1136/bmjqs-2019-009367
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149:e1-e156. doi:10.1161/CIR.0000000000001193
- Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160:e545-e608. doi:10.1016/j.chest.2021.07.055
- Institute for Safe Medication Practices (ISMP). List of high-alert medications in community/ambulatory care settings. ISMP. September 30, 2021. Accessed September 11, 2025. https://home.ecri.org/blogs/ismp-resources/high-alert-medications-in-community-ambulatory-care-settings
- Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6:e12757. doi:10.1002/rth2.12757
- Koolian M, Wiseman D, Mantzanis H, et al. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6:e12758. doi:10.1002/rth2.12758
- Wang SV, Rogers JR, Jin Y, et al. Stepped-wedge randomised trial to evaluate population health intervention designed to increase appropriate anticoagulation in patients with atrial fibrillation. BMJ Qual Saf. 2019;28:835-842. doi:10.1136/bmjqs-2019-009367
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System