User login
Metabolic syndrome, type 2 diabetes mellitus (T2DM), and the “diabetes syndrome,” are interrelated, serious health conditions that share common risk factors and mechanisms. While they are each distinct conditions, a significant association exists between them, with metabolic syndrome often being considered a precursor to the development of typical T2DM.
Metabolic syndrome is a cluster of individual metabolic abnormalities that includes a combination of risk factors such as abdominal obesity, high blood pressure, elevated insulin levels, high triglyceride levels, and low levels of high-density lipoprotein (HDL) cholesterol related to genes and epigenetic changes associated with insulin resistance. These risk factors increase the likelihood of developing cardiovascular diseases, such as heart disease and stroke, and, when combined with significant damage to β -cell function and the influence of concordant environmental precipitants, result in hyperglycemia/overt diabetes—classically defined as T2DM.
It is estimated that there will be a staggering 3.1 billion people living with T2DM by 2050, according to a recent article in The Lancet. This devastating number will place a heavy burden on the health care system.
However, this typical pathophysiologic definition of T2DM is imprecise. Twenty percent of patients with T2DM have islet-cell antibodies that are typical of the immune destruction of β-cells in patients with type 1 diabetes mellitus (T1DM). Furthermore, approximately 40% of patients with T1DM have insulin resistance.
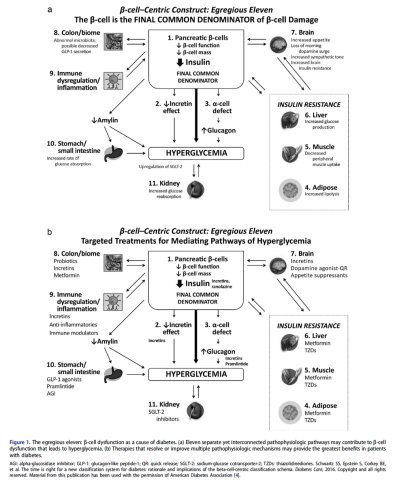
Thus, to better understand and distinguish the disease processes unique to each individual, we have defined a new beta cell classification for all forms of diabetes mellitus (DM). In this classification, there are 4 common pathophysiologic causes of all DM (including classic T2DM), with resultant damage to the β-cells (ie, genetic and epigenetic changes, inflammation, an abnormal environment, and insulin resistance), which results in 11 mechanisms of hyperglycemia, represented as “the egregious eleven” in Figure 1.
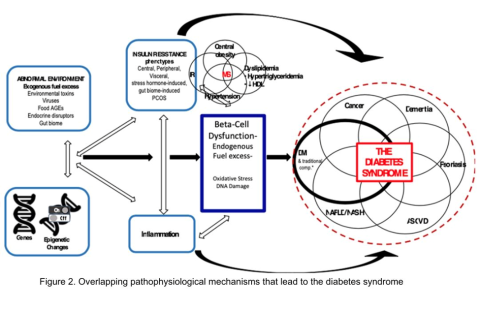
Additionally, Figure 2 illustrates the association between overlapping genes/epigenetic changes responsible for DM and the increased susceptibility to developing various microvascular complications commonly observed in all forms of DM, including classic T2DM. These complications, now recognized as components of the diabetes syndrome, encompass a range of conditions with shared interrelated pathophysiologic mechanisms, such as arteriosclerotic vascular disease (ASVD), dementia, some cancers, nonalcoholic fatty liver disease or nonalcoholic steatohepatitis (NAFLD/NASH), or psoriasis.
The likelihood of developing a specific type of DM, with classic complications or associated conditions, is contingent on an individual’s genes, epigenetic factors, inflammation, insulin resistance, and environmental exposures over time. It has now been postulated that these factors can be identified in a particular individual by a set of genomics, metabolomics, proteomics, and markers of these processes.
This more precise approach has the added benefit of giving rise to a more accurate individualization of therapy—precision medicine.
Precision medicine is an approach to healthcare that considers an individual's specific characteristics, such as genetic makeup, lifestyle, and environmental factors, to tailor medical treatments and interventions. In the context of this discussion on T2DM, precision medicine’s goal is to provide targeted therapies and interventions based on an individual's unique -omic profile to improve treatment outcomes and minimize side effects. An additional benefit of precision medicine use in diabetes syndrome is giving the diabetes specialist the opportunity to treat the whole patient, looking for complications and associated conditions earlier via defining the presence or absence of various markers of their individual pathophysiology. Additionally, we have come to recognize that many of the medications for treating T2DM (eg, glucagon-like peptide 1 receptor agonists [GLP-1 RA], dipeptidyl peptidase 4 inhibitors [DPP-4 inhibitors], sodium-glucose cotransporter-2 inhibitors [SGLT-2 inhibitors], metformin, Cycloset [bromocriptine mesylate]) can offer other benefits for the patient—treating not only multiple mechanisms of hyperglycemia (the egregious eleven: use the fewest number of agents in combination to treat the most number of mechanisms of hyperglycemia) but also recognize that they can prevent and treat the complications and associated conditions of the diabetes syndrome: cardiovascular, renal, liver, some cancers, psoriasis, and dementia.
The classic link between metabolic syndrome and T2DM is important to consider when applying precision medicine approaches to the management of T2DM. Here are some examples of how precision medicine is being applied in the management of T2DM:
Genetic testing: Genetic testing can help identify specific genetic variants or mutations that may influence an individual's risk of developing T2DM or their response to certain medications. By understanding a person's genetic predisposition, clinicians can make more informed decisions about treatment options and develop personalized strategies for their patients.
Pharmacogenomics: Certain genetic variations can impact how a person metabolizes and responds to specific diabetes medications. By analyzing an individual's genetic profile, medications that are more likely to be effective and have fewer adverse effects for that patient may be selected.
Continuous glucose monitoring (CGM): CGM devices provide real-time information about an individual’s blood glucose levels, allowing for more precise management of diabetes. By continuously monitoring glucose levels, patterns can be identified, allowing for adjustments to medication dosages, dietary recommendations, and lifestyle modifications on an individualized basis.
Lifestyle interventions: Precision medicine also recognizes that lifestyle factors play a crucial role in the development and management of T2DM. Lifestyle interventions, such as diet and exercise plans, based on an individual's preferences, metabolic profile, and response to different interventions can be personalized (ie, some individuals may benefit more from a low-carbohydrate diet, while others may respond better to a Mediterranean-style diet).
Predictive modeling and risk stratification: Precision medicine leverages data analytics and predictive modeling to assess an individual's risk of developing complications associated with T2DM. By analyzing various factors such as medical history, genetics, lifestyle, and biomarkers, individuals who are at a higher risk of developing complications can be identified, and their treatment plans can be tailored accordingly. Precision medicine enables early identification of individuals who are at a higher risk of developing T2DM based on their metabolic syndrome status.
In summary, precision medicine for T2DM considers the link between metabolic syndrome and diabetes syndrome to develop personalized approaches for prevention, early intervention, and treatment. By understanding an individual's metabolic and genetic profile, targeted strategies to optimize management and improve outcomes for patients with metabolic syndrome and those at risk of developing diabetes can be implemented.
It is important to note that while precision medicine holds promise in improving diabetes management, it is still an evolving field, and its widespread implementation is not yet fully realized. Collaboration between clinicians, researchers, and technological advancements will continue to drive the progress of precision medicine in T2DM management.
Metabolic syndrome, type 2 diabetes mellitus (T2DM), and the “diabetes syndrome,” are interrelated, serious health conditions that share common risk factors and mechanisms. While they are each distinct conditions, a significant association exists between them, with metabolic syndrome often being considered a precursor to the development of typical T2DM.
Metabolic syndrome is a cluster of individual metabolic abnormalities that includes a combination of risk factors such as abdominal obesity, high blood pressure, elevated insulin levels, high triglyceride levels, and low levels of high-density lipoprotein (HDL) cholesterol related to genes and epigenetic changes associated with insulin resistance. These risk factors increase the likelihood of developing cardiovascular diseases, such as heart disease and stroke, and, when combined with significant damage to β -cell function and the influence of concordant environmental precipitants, result in hyperglycemia/overt diabetes—classically defined as T2DM.
It is estimated that there will be a staggering 3.1 billion people living with T2DM by 2050, according to a recent article in The Lancet. This devastating number will place a heavy burden on the health care system.
However, this typical pathophysiologic definition of T2DM is imprecise. Twenty percent of patients with T2DM have islet-cell antibodies that are typical of the immune destruction of β-cells in patients with type 1 diabetes mellitus (T1DM). Furthermore, approximately 40% of patients with T1DM have insulin resistance.
Thus, to better understand and distinguish the disease processes unique to each individual, we have defined a new beta cell classification for all forms of diabetes mellitus (DM). In this classification, there are 4 common pathophysiologic causes of all DM (including classic T2DM), with resultant damage to the β-cells (ie, genetic and epigenetic changes, inflammation, an abnormal environment, and insulin resistance), which results in 11 mechanisms of hyperglycemia, represented as “the egregious eleven” in Figure 1.
Additionally, Figure 2 illustrates the association between overlapping genes/epigenetic changes responsible for DM and the increased susceptibility to developing various microvascular complications commonly observed in all forms of DM, including classic T2DM. These complications, now recognized as components of the diabetes syndrome, encompass a range of conditions with shared interrelated pathophysiologic mechanisms, such as arteriosclerotic vascular disease (ASVD), dementia, some cancers, nonalcoholic fatty liver disease or nonalcoholic steatohepatitis (NAFLD/NASH), or psoriasis.
The likelihood of developing a specific type of DM, with classic complications or associated conditions, is contingent on an individual’s genes, epigenetic factors, inflammation, insulin resistance, and environmental exposures over time. It has now been postulated that these factors can be identified in a particular individual by a set of genomics, metabolomics, proteomics, and markers of these processes.
This more precise approach has the added benefit of giving rise to a more accurate individualization of therapy—precision medicine.
Precision medicine is an approach to healthcare that considers an individual's specific characteristics, such as genetic makeup, lifestyle, and environmental factors, to tailor medical treatments and interventions. In the context of this discussion on T2DM, precision medicine’s goal is to provide targeted therapies and interventions based on an individual's unique -omic profile to improve treatment outcomes and minimize side effects. An additional benefit of precision medicine use in diabetes syndrome is giving the diabetes specialist the opportunity to treat the whole patient, looking for complications and associated conditions earlier via defining the presence or absence of various markers of their individual pathophysiology. Additionally, we have come to recognize that many of the medications for treating T2DM (eg, glucagon-like peptide 1 receptor agonists [GLP-1 RA], dipeptidyl peptidase 4 inhibitors [DPP-4 inhibitors], sodium-glucose cotransporter-2 inhibitors [SGLT-2 inhibitors], metformin, Cycloset [bromocriptine mesylate]) can offer other benefits for the patient—treating not only multiple mechanisms of hyperglycemia (the egregious eleven: use the fewest number of agents in combination to treat the most number of mechanisms of hyperglycemia) but also recognize that they can prevent and treat the complications and associated conditions of the diabetes syndrome: cardiovascular, renal, liver, some cancers, psoriasis, and dementia.
The classic link between metabolic syndrome and T2DM is important to consider when applying precision medicine approaches to the management of T2DM. Here are some examples of how precision medicine is being applied in the management of T2DM:
Genetic testing: Genetic testing can help identify specific genetic variants or mutations that may influence an individual's risk of developing T2DM or their response to certain medications. By understanding a person's genetic predisposition, clinicians can make more informed decisions about treatment options and develop personalized strategies for their patients.
Pharmacogenomics: Certain genetic variations can impact how a person metabolizes and responds to specific diabetes medications. By analyzing an individual's genetic profile, medications that are more likely to be effective and have fewer adverse effects for that patient may be selected.
Continuous glucose monitoring (CGM): CGM devices provide real-time information about an individual’s blood glucose levels, allowing for more precise management of diabetes. By continuously monitoring glucose levels, patterns can be identified, allowing for adjustments to medication dosages, dietary recommendations, and lifestyle modifications on an individualized basis.
Lifestyle interventions: Precision medicine also recognizes that lifestyle factors play a crucial role in the development and management of T2DM. Lifestyle interventions, such as diet and exercise plans, based on an individual's preferences, metabolic profile, and response to different interventions can be personalized (ie, some individuals may benefit more from a low-carbohydrate diet, while others may respond better to a Mediterranean-style diet).
Predictive modeling and risk stratification: Precision medicine leverages data analytics and predictive modeling to assess an individual's risk of developing complications associated with T2DM. By analyzing various factors such as medical history, genetics, lifestyle, and biomarkers, individuals who are at a higher risk of developing complications can be identified, and their treatment plans can be tailored accordingly. Precision medicine enables early identification of individuals who are at a higher risk of developing T2DM based on their metabolic syndrome status.
In summary, precision medicine for T2DM considers the link between metabolic syndrome and diabetes syndrome to develop personalized approaches for prevention, early intervention, and treatment. By understanding an individual's metabolic and genetic profile, targeted strategies to optimize management and improve outcomes for patients with metabolic syndrome and those at risk of developing diabetes can be implemented.
It is important to note that while precision medicine holds promise in improving diabetes management, it is still an evolving field, and its widespread implementation is not yet fully realized. Collaboration between clinicians, researchers, and technological advancements will continue to drive the progress of precision medicine in T2DM management.
Metabolic syndrome, type 2 diabetes mellitus (T2DM), and the “diabetes syndrome,” are interrelated, serious health conditions that share common risk factors and mechanisms. While they are each distinct conditions, a significant association exists between them, with metabolic syndrome often being considered a precursor to the development of typical T2DM.
Metabolic syndrome is a cluster of individual metabolic abnormalities that includes a combination of risk factors such as abdominal obesity, high blood pressure, elevated insulin levels, high triglyceride levels, and low levels of high-density lipoprotein (HDL) cholesterol related to genes and epigenetic changes associated with insulin resistance. These risk factors increase the likelihood of developing cardiovascular diseases, such as heart disease and stroke, and, when combined with significant damage to β -cell function and the influence of concordant environmental precipitants, result in hyperglycemia/overt diabetes—classically defined as T2DM.
It is estimated that there will be a staggering 3.1 billion people living with T2DM by 2050, according to a recent article in The Lancet. This devastating number will place a heavy burden on the health care system.
However, this typical pathophysiologic definition of T2DM is imprecise. Twenty percent of patients with T2DM have islet-cell antibodies that are typical of the immune destruction of β-cells in patients with type 1 diabetes mellitus (T1DM). Furthermore, approximately 40% of patients with T1DM have insulin resistance.
Thus, to better understand and distinguish the disease processes unique to each individual, we have defined a new beta cell classification for all forms of diabetes mellitus (DM). In this classification, there are 4 common pathophysiologic causes of all DM (including classic T2DM), with resultant damage to the β-cells (ie, genetic and epigenetic changes, inflammation, an abnormal environment, and insulin resistance), which results in 11 mechanisms of hyperglycemia, represented as “the egregious eleven” in Figure 1.
Additionally, Figure 2 illustrates the association between overlapping genes/epigenetic changes responsible for DM and the increased susceptibility to developing various microvascular complications commonly observed in all forms of DM, including classic T2DM. These complications, now recognized as components of the diabetes syndrome, encompass a range of conditions with shared interrelated pathophysiologic mechanisms, such as arteriosclerotic vascular disease (ASVD), dementia, some cancers, nonalcoholic fatty liver disease or nonalcoholic steatohepatitis (NAFLD/NASH), or psoriasis.
The likelihood of developing a specific type of DM, with classic complications or associated conditions, is contingent on an individual’s genes, epigenetic factors, inflammation, insulin resistance, and environmental exposures over time. It has now been postulated that these factors can be identified in a particular individual by a set of genomics, metabolomics, proteomics, and markers of these processes.
This more precise approach has the added benefit of giving rise to a more accurate individualization of therapy—precision medicine.
Precision medicine is an approach to healthcare that considers an individual's specific characteristics, such as genetic makeup, lifestyle, and environmental factors, to tailor medical treatments and interventions. In the context of this discussion on T2DM, precision medicine’s goal is to provide targeted therapies and interventions based on an individual's unique -omic profile to improve treatment outcomes and minimize side effects. An additional benefit of precision medicine use in diabetes syndrome is giving the diabetes specialist the opportunity to treat the whole patient, looking for complications and associated conditions earlier via defining the presence or absence of various markers of their individual pathophysiology. Additionally, we have come to recognize that many of the medications for treating T2DM (eg, glucagon-like peptide 1 receptor agonists [GLP-1 RA], dipeptidyl peptidase 4 inhibitors [DPP-4 inhibitors], sodium-glucose cotransporter-2 inhibitors [SGLT-2 inhibitors], metformin, Cycloset [bromocriptine mesylate]) can offer other benefits for the patient—treating not only multiple mechanisms of hyperglycemia (the egregious eleven: use the fewest number of agents in combination to treat the most number of mechanisms of hyperglycemia) but also recognize that they can prevent and treat the complications and associated conditions of the diabetes syndrome: cardiovascular, renal, liver, some cancers, psoriasis, and dementia.
The classic link between metabolic syndrome and T2DM is important to consider when applying precision medicine approaches to the management of T2DM. Here are some examples of how precision medicine is being applied in the management of T2DM:
Genetic testing: Genetic testing can help identify specific genetic variants or mutations that may influence an individual's risk of developing T2DM or their response to certain medications. By understanding a person's genetic predisposition, clinicians can make more informed decisions about treatment options and develop personalized strategies for their patients.
Pharmacogenomics: Certain genetic variations can impact how a person metabolizes and responds to specific diabetes medications. By analyzing an individual's genetic profile, medications that are more likely to be effective and have fewer adverse effects for that patient may be selected.
Continuous glucose monitoring (CGM): CGM devices provide real-time information about an individual’s blood glucose levels, allowing for more precise management of diabetes. By continuously monitoring glucose levels, patterns can be identified, allowing for adjustments to medication dosages, dietary recommendations, and lifestyle modifications on an individualized basis.
Lifestyle interventions: Precision medicine also recognizes that lifestyle factors play a crucial role in the development and management of T2DM. Lifestyle interventions, such as diet and exercise plans, based on an individual's preferences, metabolic profile, and response to different interventions can be personalized (ie, some individuals may benefit more from a low-carbohydrate diet, while others may respond better to a Mediterranean-style diet).
Predictive modeling and risk stratification: Precision medicine leverages data analytics and predictive modeling to assess an individual's risk of developing complications associated with T2DM. By analyzing various factors such as medical history, genetics, lifestyle, and biomarkers, individuals who are at a higher risk of developing complications can be identified, and their treatment plans can be tailored accordingly. Precision medicine enables early identification of individuals who are at a higher risk of developing T2DM based on their metabolic syndrome status.
In summary, precision medicine for T2DM considers the link between metabolic syndrome and diabetes syndrome to develop personalized approaches for prevention, early intervention, and treatment. By understanding an individual's metabolic and genetic profile, targeted strategies to optimize management and improve outcomes for patients with metabolic syndrome and those at risk of developing diabetes can be implemented.
It is important to note that while precision medicine holds promise in improving diabetes management, it is still an evolving field, and its widespread implementation is not yet fully realized. Collaboration between clinicians, researchers, and technological advancements will continue to drive the progress of precision medicine in T2DM management.