User login
- Endoanal ultrasound is superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography, and 50% for clinical assessment.
- Pelvic floor rehabilitation with biofeedback/electrical stimulation is the mainstay nonsurgical treatment. Some centers report success rates as high as 70%.
- Consider surgery only for distinct anatomic defects, and only when nonsurgical interventions have not succeeded.
Thanks to improvements in our understanding of the causes of uncontrolled defecation and an increasing array of diagnostic and treatment options, physicians can now manage this condition more effectively.
Fecal incontinence is a relevant clinical issue for Ob/Gyns, and requires compassion and emotional support on the part of the physician and health-care team. This debilitating problem is underreported, but is thought to affect 2.2% of the general population. Women are 8 times more likely to have fecal incontinence than men, in age-controlled groups.1
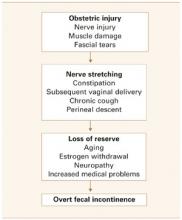
Although sphincter damage during vaginal delivery—even in deliveries over an intact perineum2,3—may be clinically silent in most women, these women are at risk of developing overt fecal incontinence with subsequent deliveries, and with aging and its decompensating factors (FIGURE 1).
A directed examination, optimal diagnostic testing, and a step-by-step treatment algorithm are the basis of effective management.
FIGURE 1 Common pathway leading to fecal incontinence
How ‘silent’ sphincter damage can lead to incontinence
Obstetric disruption of the anal sphincter structure or function is a common cause of fecal incontinence, although it is usually multifactorial, involving abnormalities of 1 or more continence mechanisms (FIGURE 2).
Weakening of the sphincter mechanism after vaginal delivery is attributed to both direct trauma to the muscle and indirect trauma to the pudendal nerve (unilateral or bilateral). Anatomic injury usually occurs in the anterior portion of the sphincter—a site amenable to direct repair, with good results.
Of the various investigational diagnostic modalities available, endoanal ultrasound has been superior, though not perfect. In 1 series, endoanal sonography detected anal sphincter tears in 35% of primiparous and 40% of multiparous women; of these, 13% and 23%, respectively, had symptoms of fecal incontinence or urgency.4
The pudendal nerve is susceptible to traction injury from childbirth and chronic straining. In a prospective study of the effect of vaginal deliveries on the pelvic floor, 80% of primigravidas sustained reversible pudendal nerve damage—which, in some women, persisted for up to 5 years.5
Obstetric risk factors include:
- infant weighing more than 4,000 g,
- use of forceps,
- prolonged second stage of labor,
- midline episiotomy,
- occipitoposterior position, and
- failed detection or suboptimal repair of anal sphincterinjuryat the time of vaginaldelivery.
Onset of menopause and subsequent pelvic floor weakness and perineal descent aggravates this occult neuropathy.
Minor incontinence is also common in patients with advanced age, internal sphincter defects, prolapsing hemorrhoids, rectal prolapse, and fecal impaction (the rectum chronically distends with feces, continuously inducing internal anal sphincter relaxation and resulting in leakage of stool).
Evaluation: Directed history, 4-point physical exam
A directed history helps indicate which diagnostic tests are most relevant. Establish severity and frequency of fecal incontinence, presence of fecal urgency, pad use, type of incontinence (solid, liquid, or gas), and presence of associated urinary incontinence and prolapse.
Obstetric history should identify:
- parity,
- duration of the second stage of labor,
- occipitoposterior presentation,
- use of forceps,
- extent of vaginal lacerations, and
- weight of the infant.
Dietary habits (especially fiber intake) and chronic laxative use should be noted.
Also note any history of abdominopelvic surgery, radiation, back injury, and neurologic disorders.
4-point physical exam
- Inspect the perineum/perianal area for evidence of dermatitis, infection, fistula, hemorrhoids, and deformities of the anus. Ask the patient to bear down as you look for evidence of pelvic floor weakness and rectal prolapse.
- Assess the anal wink (cutaneoanal contractile reflex) by stroking the skin surrounding the anus. Absence of a reflexive contraction of the external anal sphincter suggests nerve damage or spinal arc interruption.
- Perform a vaginal exam to identify pelvic prolapse and exclude rectovaginal fistula. At this time assess pelvic muscle strength and teach the patient proper pelvic floor exercises.
- Conduct a digital rectal examination to assess the length and orientation of the analcanal, bulk of the perianal tissues, and tone of the anal sphincters, as well as to rule out a mass or impaction. Repeat digital examination with the patient bearing down to assess the squeeze pressures, movement of the puborectalis muscle, and extent of pelvic floor descent.
These findings are predominantly subjective and depend on operator experience. In general, they correlate poorly with objective measurements of sphincter pressures.6
If a rectovaginal fistula is suspected, try retrograde filling of the rectum with 200 cc of dye-stained fluid (using a 50-cc catheter tip syringe), then observe vaginal extravasation.
Diagnostic strategy: Routine endoanal ultrasound, manometry
Our strategy is to routinely proceed with endoanal ultrasound and anal manometry to assess the anal sphincters. We may then recommend sigmoidoscopy or anoscopy to rule out mucosal lesions.
It is important to bear in mind that continence depends on multiple physiologic mechanisms and, therefore, no single diagnostic test yields positive results in all patients.
Endoanal ultrasound has significantly changed the evaluation of fecal incontinence over the last decade. In women with fecal incontinence in whom obstetric injury is suspected, 90% have sonographic evidence of injury to 1 or both sphincters.
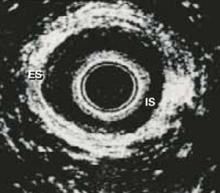
These ultrasound images show 2 discrete rings of tissue: The inner hypoechoic ring represents the internal anal sphincter and the outer hyperechoic ring represents the external anal sphincter (FIGURE 3). Disrupted continuity of these rings is consistent with structural damage to the sphincter.
Endoanal ultrasound is regarded as superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography (EMG), and 50% for clinical assessment.7,8 It is the simplest and least-invasive test to assess the structural integrity of the anal sphincters.
It can also serve as a screening tool to detect occult sphincter injury following vaginal delivery, thus identifying women at high risk for future incontinence. Women with transient stool or gas incontinence following vaginal delivery may be candidates for screening endoanal ultrasound for further evaluation.
- Where endoanal ultrasound is unavailable, transperineal ultrasound may be an alternative option.
Anorectal manometry can detect functional weakness of sphincters that are anatomically intact by measuring sensation and squeeze pressures. Patients with intact sphincters but abnormal anal manometry findings may benefit from physical therapy and biofeedback aimed at increasing muscular function by stimulating the neuromuscular feedback loop.
During manometry, the physician assesses maximum resting and squeeze pressures of the anal canal, the rectoanal inhibitory reflex, rectal sensation, and rectal compliance using either water-perfused catheters, solid-state microtransducers, or air-or water-filled balloons. Normal values vary widely among institutions and are poorly standardized.
Decreased resting pressure suggests isolated internal anal sphincter injury; decreased squeeze pressure suggests external anal sphincter injury. Of all parameters, maximum squeeze pressure has the greatest sensitivity and specificity at a cut-off of 60 mm Hg in women.9
Rectal sensory testing also is useful in identifying patients who may benefit from biofeedback therapy, and can detect afferent nerve injury as a contributing cause of incontinence. Biofeedback is unlikely to be useful in patients with a poor or absent sensation. In addition, for patients already involved in biofeedback, the test can assess rectal response to training and indicate rectal irritability or poor compliance.
Rectal sensation is assessed by inflating a balloon in the rectum and recording the smallest volume of rectal distention for first detectable sensation (rectal sensory threshold), sensory urgency, and pain (maximum tolerable volume).
Ancillary tests are of limited benefit as screening tools, but may have utility in special circumstances.
- Electromyography of the external anal sphincter and the pelvic floor muscles—traditionally performed using needle electrodes or surface electrodes—is helpful in delineating areas of sphincter injury by mapping the sphincter. However, much of this information is now obtained accurately by endoanal ultrasound, which has replaced needle EMG for most clinicians. Surface EMG is still useful for evaluating sphincter function and in biofeedback training.
- Defecography involves imaging the rectum after filling it with contrast material, and then observing the process, rate, and completeness of rectal evacuation using fluoroscopy. Its usefulness in the evaluation of fecal incontinence is limited to cases of rectal prolapse and for the diagnosis of rectocele and enterocele.
- Investigational: Pudendal nerve testing. This test uses an electrode to measure pudendal nerve conduction time, known as pudendal nerve terminal motor latency (PNTML)—thus allowing further investigation for nerve injury.
- Normal PNTML value is 2.2 milliseconds.
- A value between 2.2 and 2.6 milliseconds indicates probable nerve damage.
- A value of 2.6 milliseconds or greater confirms nerve damage.
Clinical use of PNTML is controversial; it helps diagnose nerve injury, but is currently reserved for investigational purposes.10
FIGURE 3 Endoanal ultrasound
Disruption of continuity of the inner hypoechoic ring (IS, internal sphincter) and the outer hyperechoic ring (ES, external sphincter) on endoanal ultrasound is consistent with structural damage to the sphincter. This image shows normal anatomy.
Mainstays: Nonsurgical treatments
The general principles of treatment are to identify the underlying cause of incontinence and direct therapy based on these findings. Since much of the disability in these patients is psychological, a supportive medical team is essential. Patient educational books, advice on the use of continent aides, and perianal skin care also are crucial.
The physician and patient must formulate a treatment plan based on the severity of the incontinence and degree of social impairment. In general, a trial of nonsurgical therapy is recommended before proceeding to surgery. A standardized treatment algorithm (FIGURE 4) should be adapted to each patient.
The aim of nonsurgical management is:
- to increase anorectal sensation, and
- to increase the strength of the external sphincter and pelvic floor.
First: Diet and medical therapy. For patients with fecal incontinence, nonsurgical intervention remains the first line of treatment given the high rate of underlying neuromuscular damage.
The aims of medical therapy are to decrease stool frequency and improve stool consistency. The patient’s diet should include adequate fiber and fluid intake to promote regular, soft, bulky stools. High-fiber foods including whole-wheat grains, fresh vegetables, and beans should be advised. Bulking agents like methylcellulose (eg, Citrucel), or psyllium (eg, Metamucil) can be helpful.
Stool frequency can be reduced with antidiarrheal drugs like loperamide (2-4 mg 2 or 3 times daily) or diphenoxalate (eg, Lomotil). Loperamide is usually preferred, since it also increases internal anal sphincter tone. Antiflatulants are also recommended.
Amitryptiline is useful in some women with idiopathic fecal incontinence.11,12
Biofeedback therapy. Pelvic floor rehabilitation with biofeedback and/or electrical stimulation—which focuses on the common neuromuscular etiology of fecal incontinence—is the mainstay of nonsurgical management. Some centers report a success rate as high as 70% with this therapy.13
Biofeedback is a painless, minimally invasive method of retraining pelvic floor muscles with the help of sensors placed in either the vagina or the rectum. This technique improves rectal sensation and muscular tone. It is especially useful in incontinence patients with anatomically intact anal sphincters.
Biofeedback typically involves anal manometry as well as electromyographic sensors. It is generally conducted in an outpatient treatment program that includes regular evaluation of sensation and motor tone. However, home-based units are available.
Investigational: Neuromodulation. Although approved by the US Food and Drug Administration for treatment of urge incontinence and nonobstructive voiding dysfunction, neuromodulation is still investigational for treatment of fecal incontinence—but results are encouraging.14
In this technique, electrodes attached to a portable stimulator intermittently apply low-level electrical impulses to the anal canal via the pelvic nerve supply. Newer methods involve implanting electrodes percutaneously in the S3 or S4 foramina under fluoroscopic guidance, then stimulating the sacral nerves.
Neuromodulation aims to increase maximum squeeze pressure and decrease rectal sensitivity to distention. In early trials, significant improvement of incontinence and urgency symptoms was noted.15,16 Larger and longer-duration trials are needed to elucidate long-term effectiveness.
When to consider surgery
Operative therapy should be considered only for cases in which nonsurgical interventions have failed or in which distinct anatomic defects are present.
Note that most surgical candidates still require physical therapy with biofeedback before or after the procedure.
Multiple surgical procedures have been described; some are technically challenging and should be performed only by skilled surgeons after careful preoperative counseling.
Anterior overlapping external sphincteroplasty is done in patients with obstetric or iatrogenic disruption of the external anal sphincter. In this procedure, the surgeon divides the scar and reapproximates viable sphincter muscle to produce a ring of functional muscle. Few good data exist comparing this approach with end-to-end repairs; anterior sphincteroplasty is our preferred method.
- Controversy exists over the need to plicate the internal anal sphincter. Studies have shown that more extensive repairs offer no additional advantage with regard to out-comes.17,18 Most pelvic reconstructive surgeons prefer to perform colporrhaphy and perineorrhaphy at the time of sphincter repair, since defects in the posterior compartment often coexist in these patients.
The success rate of sphincteroplasty ranges from 40% to 75% in appropriately selected patients.19,20
Patients with satisfactory results demonstrate an increase in squeeze pressure postoperatively. Failure to elicit this response correlated accurately with a persistent sphincter defect.21
Total pelvic floor repair may help patients with neurogenic or idiopathic fecal incontinence. The principle of this procedure is to restore the anorectal angle, lengthen the anal canal, and create a circumferential buttress around the anorectum.
In this rather extensive surgery, the surgeon plicates the puborectalis, ischiococcygeus, and iliococcygeus muscles to the rectum; the levator muscles and the external anal sphincter are plicated anteriorly. Success rates vary from 14% to 55%.22,23
Skeletal muscle transposition surgery, with creation of a neosphincter from the gracilis muscle, is performed in patients with pelvic floor muscle weakness so profound that the above techniques offer no improvement. It should be viewed as a salvage operation.
The gracilis muscle is detached from its insertion, wrapped around the anus, and reattached to the contralateral ischial tuberosity. The success of this surgery may be improved by electrically stimulating the transposed muscle with an implanted stimulator.
This procedure is associated with significant postoperative complications; many patients require multiple reoperations. Thus, this intervention is useful only in patients with refractory incontinence.
Artificial anal sphincter procedure involves placing a cuff around the anal canal and a pressure-regulating balloon and pump in the labia majora. The cuff remains inflated, thereby occluding the anal canal until the patient experiences a desire to defecate, at which time it is deflated.
Results are encouraging, with initial success rates from 60% to 75%.24,25 However, local infection can be problematic.
Less common procedures include anal encirclement (Thiersch procedure), perianal fat injection, antegrade continence enema, and fecal diversion.
A word on prevention
During childbirth. Strategies that may reduce pelvic floor trauma:
- Favor mediolateral over midline episiotomy,
- avoid a prolonged second stage of labor and forceps delivery, and
- repair anal sphincter lacerations with an overlapping technique.
Also, discuss regular pelvic floor exercises, constipation prevention, weight loss, and smoking cessation with all obstetric patients—especially those with fecal incontinence risk factors.
Debate continues about elective cesarean section in women with occult sphincter injury on postpartum endoanal ultrasound, transient symptoms of fecal incontinence with prior delivery, or overt gas incontinence.26 This practice may have clinical utility, but data are insufficient to recommend it routinely.
The authors report no financial relationships relevant to this article.
1. Furner S, Cautley E, Norton N, Nelson R. Community-based prevalence of anal incontinence. JAMA. 1995;274:559.-
2. Sultan AH, Kamm MA, Hudson CN, et al. Anal-sphincter disruption during vaginal delivery. N Engl J Med. 1993;329:1905.-
3. Abramowitz L, Sobhani I, Ganansia R, et al. Are sphincter defects the cause of anal incontinence after vaginal delivery? Results of a prospective study. Dis Colon Rectum. May 2000;43:590-596; discussion:596–598.
4. Fynes M, Donnelly VS, O’Connell PR, O’Herlihy C. Cesarean delivery and anal sphincter injury. Obstet Gynecol. 1998;92:496-500.
5. Haadem K, Dahlstrom JA, Lingman G. Anal sphincter function after delivery: a prospective study in women with sphincter rupture and controls. Eur J Obstet Gynecol Reprod Biol. 1990;35:7.-
6. Coller JA. Clinical application of anorectal manometry. Gastroenterol Clin North Am. 1987;16:17.-
7. Sultan AH, Kamm MA, Talbot IC, et al. Anal endosonography for identifying external sphincter defects confirmed histologically. Br J Surg. 1994;81:463.-
8. Tjandra JJ, Milsom JW, Schroeder T, Fazio VW. Endoluminal ultrasound is preferable to electromyography in mapping anal sphincteric defects. Dis Colon Rectum. 1993;36:689-692.
9. Felt-Bersma RJF, Klinkenberg-Knol EC, Meuwissen SGM. Anorectal function investigations in incontinent and continent patients. Dis Colon Rectum. 1990;33:479.-
10. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
11. Santoro GA, Eitan BZ, Pryde A, Bartolo DC. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis Colon Rectum. 2000;43:1676.-
12. Read M, Read NW, Barber DC, Duthie HL. Effects of loperamide on anal sphincter function in patients complaining of chronic diarrhea with fecal incontinence and urgency. Dig Dis Sci. 1982;27:807.-
13. Ryn AK, Morren GL, Hallbook O, Sjodahl R. Long-term results of electromyo-graphic biofeedback training for fecal incontinence. Dis Colon Rectum. 2000;43:1262-1266
14. Matzel KE, Stadelmaier U, Hohenfellner M, Gall FP. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet. 1995;346:1124.-
15. Matzel KE, Stadelmaier U, Hohenfellner M, Hohenberger W. Chronic sacral spinal nerve stimulation for fecal incontinence: long-term results with foramen and cuff electrodes. Dis Colon Rectum. 2001;44:59.-
16. Rosen HR, Urbarz C, Holzer B, et al. Sacral nerve stimulation as a treatment for fecal incontinence. Gastroenterology. 2001;121:536.-
17. Briel JW, de Boer LM, Hop WC, Schouten WR. Clinical outcome of anterior overlapping external anal sphincter repair with internal anal sphincter imbrication. Dis Colon Rectum. 1998;41:209-214.
18. Laurberg S, Swash M, Henry MM. Effect of postanal repair on progress of neurogenic damage to the pelvic floor. Br J Surg. 1990;77:519-522.
19. Kamm MA. Faecal incontinence. BMJ. 1998;316:528.-
20. Malouf AJ, Norton CS, Engel AF, et al. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355:260.-
21. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
22. Orrom WJ, Miller R, Cornes H, et al. Comparison of anterior sphincteroplasty and postanal repair in the treatment of idiopathic fecal incontinence. Dis Colon Rectum. 1991;34:305-310.
23. Schiller, Lawrence R. Fecal incontinence. In: Feldman M, et al, eds. Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. Philadelphia, Pa: WB Saunders Co; 1997.
24. Giamundo P, Welber A, Weiss EG, et al. The Procon incontinence device: a new nonsurgical approach to preventing episodes of fecal incontinence. Am J Gastroenterol. 2002;97:232.-
25. Wong WD, Congliosi SM, Spencer MP, et al. The safety and efficacy of the artificial bowel sphincter for fecal incontinence: results from a multicenter cohort study. Dis Colon Rectum. 2002;45:1139.-
26. McKenna DS, Ester JB, Fischer JR. Elective cesarean delivery for women with a previous anal sphincter rupture. Am J Obstet Gynecol. 2003;189:1251.-
- Endoanal ultrasound is superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography, and 50% for clinical assessment.
- Pelvic floor rehabilitation with biofeedback/electrical stimulation is the mainstay nonsurgical treatment. Some centers report success rates as high as 70%.
- Consider surgery only for distinct anatomic defects, and only when nonsurgical interventions have not succeeded.
Thanks to improvements in our understanding of the causes of uncontrolled defecation and an increasing array of diagnostic and treatment options, physicians can now manage this condition more effectively.
Fecal incontinence is a relevant clinical issue for Ob/Gyns, and requires compassion and emotional support on the part of the physician and health-care team. This debilitating problem is underreported, but is thought to affect 2.2% of the general population. Women are 8 times more likely to have fecal incontinence than men, in age-controlled groups.1
Although sphincter damage during vaginal delivery—even in deliveries over an intact perineum2,3—may be clinically silent in most women, these women are at risk of developing overt fecal incontinence with subsequent deliveries, and with aging and its decompensating factors (FIGURE 1).
A directed examination, optimal diagnostic testing, and a step-by-step treatment algorithm are the basis of effective management.
FIGURE 1 Common pathway leading to fecal incontinence
How ‘silent’ sphincter damage can lead to incontinence
Obstetric disruption of the anal sphincter structure or function is a common cause of fecal incontinence, although it is usually multifactorial, involving abnormalities of 1 or more continence mechanisms (FIGURE 2).
Weakening of the sphincter mechanism after vaginal delivery is attributed to both direct trauma to the muscle and indirect trauma to the pudendal nerve (unilateral or bilateral). Anatomic injury usually occurs in the anterior portion of the sphincter—a site amenable to direct repair, with good results.
Of the various investigational diagnostic modalities available, endoanal ultrasound has been superior, though not perfect. In 1 series, endoanal sonography detected anal sphincter tears in 35% of primiparous and 40% of multiparous women; of these, 13% and 23%, respectively, had symptoms of fecal incontinence or urgency.4
The pudendal nerve is susceptible to traction injury from childbirth and chronic straining. In a prospective study of the effect of vaginal deliveries on the pelvic floor, 80% of primigravidas sustained reversible pudendal nerve damage—which, in some women, persisted for up to 5 years.5
Obstetric risk factors include:
- infant weighing more than 4,000 g,
- use of forceps,
- prolonged second stage of labor,
- midline episiotomy,
- occipitoposterior position, and
- failed detection or suboptimal repair of anal sphincterinjuryat the time of vaginaldelivery.
Onset of menopause and subsequent pelvic floor weakness and perineal descent aggravates this occult neuropathy.
Minor incontinence is also common in patients with advanced age, internal sphincter defects, prolapsing hemorrhoids, rectal prolapse, and fecal impaction (the rectum chronically distends with feces, continuously inducing internal anal sphincter relaxation and resulting in leakage of stool).
Evaluation: Directed history, 4-point physical exam
A directed history helps indicate which diagnostic tests are most relevant. Establish severity and frequency of fecal incontinence, presence of fecal urgency, pad use, type of incontinence (solid, liquid, or gas), and presence of associated urinary incontinence and prolapse.
Obstetric history should identify:
- parity,
- duration of the second stage of labor,
- occipitoposterior presentation,
- use of forceps,
- extent of vaginal lacerations, and
- weight of the infant.
Dietary habits (especially fiber intake) and chronic laxative use should be noted.
Also note any history of abdominopelvic surgery, radiation, back injury, and neurologic disorders.
4-point physical exam
- Inspect the perineum/perianal area for evidence of dermatitis, infection, fistula, hemorrhoids, and deformities of the anus. Ask the patient to bear down as you look for evidence of pelvic floor weakness and rectal prolapse.
- Assess the anal wink (cutaneoanal contractile reflex) by stroking the skin surrounding the anus. Absence of a reflexive contraction of the external anal sphincter suggests nerve damage or spinal arc interruption.
- Perform a vaginal exam to identify pelvic prolapse and exclude rectovaginal fistula. At this time assess pelvic muscle strength and teach the patient proper pelvic floor exercises.
- Conduct a digital rectal examination to assess the length and orientation of the analcanal, bulk of the perianal tissues, and tone of the anal sphincters, as well as to rule out a mass or impaction. Repeat digital examination with the patient bearing down to assess the squeeze pressures, movement of the puborectalis muscle, and extent of pelvic floor descent.
These findings are predominantly subjective and depend on operator experience. In general, they correlate poorly with objective measurements of sphincter pressures.6
If a rectovaginal fistula is suspected, try retrograde filling of the rectum with 200 cc of dye-stained fluid (using a 50-cc catheter tip syringe), then observe vaginal extravasation.
Diagnostic strategy: Routine endoanal ultrasound, manometry
Our strategy is to routinely proceed with endoanal ultrasound and anal manometry to assess the anal sphincters. We may then recommend sigmoidoscopy or anoscopy to rule out mucosal lesions.
It is important to bear in mind that continence depends on multiple physiologic mechanisms and, therefore, no single diagnostic test yields positive results in all patients.
Endoanal ultrasound has significantly changed the evaluation of fecal incontinence over the last decade. In women with fecal incontinence in whom obstetric injury is suspected, 90% have sonographic evidence of injury to 1 or both sphincters.
These ultrasound images show 2 discrete rings of tissue: The inner hypoechoic ring represents the internal anal sphincter and the outer hyperechoic ring represents the external anal sphincter (FIGURE 3). Disrupted continuity of these rings is consistent with structural damage to the sphincter.
Endoanal ultrasound is regarded as superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography (EMG), and 50% for clinical assessment.7,8 It is the simplest and least-invasive test to assess the structural integrity of the anal sphincters.
It can also serve as a screening tool to detect occult sphincter injury following vaginal delivery, thus identifying women at high risk for future incontinence. Women with transient stool or gas incontinence following vaginal delivery may be candidates for screening endoanal ultrasound for further evaluation.
- Where endoanal ultrasound is unavailable, transperineal ultrasound may be an alternative option.
Anorectal manometry can detect functional weakness of sphincters that are anatomically intact by measuring sensation and squeeze pressures. Patients with intact sphincters but abnormal anal manometry findings may benefit from physical therapy and biofeedback aimed at increasing muscular function by stimulating the neuromuscular feedback loop.
During manometry, the physician assesses maximum resting and squeeze pressures of the anal canal, the rectoanal inhibitory reflex, rectal sensation, and rectal compliance using either water-perfused catheters, solid-state microtransducers, or air-or water-filled balloons. Normal values vary widely among institutions and are poorly standardized.
Decreased resting pressure suggests isolated internal anal sphincter injury; decreased squeeze pressure suggests external anal sphincter injury. Of all parameters, maximum squeeze pressure has the greatest sensitivity and specificity at a cut-off of 60 mm Hg in women.9
Rectal sensory testing also is useful in identifying patients who may benefit from biofeedback therapy, and can detect afferent nerve injury as a contributing cause of incontinence. Biofeedback is unlikely to be useful in patients with a poor or absent sensation. In addition, for patients already involved in biofeedback, the test can assess rectal response to training and indicate rectal irritability or poor compliance.
Rectal sensation is assessed by inflating a balloon in the rectum and recording the smallest volume of rectal distention for first detectable sensation (rectal sensory threshold), sensory urgency, and pain (maximum tolerable volume).
Ancillary tests are of limited benefit as screening tools, but may have utility in special circumstances.
- Electromyography of the external anal sphincter and the pelvic floor muscles—traditionally performed using needle electrodes or surface electrodes—is helpful in delineating areas of sphincter injury by mapping the sphincter. However, much of this information is now obtained accurately by endoanal ultrasound, which has replaced needle EMG for most clinicians. Surface EMG is still useful for evaluating sphincter function and in biofeedback training.
- Defecography involves imaging the rectum after filling it with contrast material, and then observing the process, rate, and completeness of rectal evacuation using fluoroscopy. Its usefulness in the evaluation of fecal incontinence is limited to cases of rectal prolapse and for the diagnosis of rectocele and enterocele.
- Investigational: Pudendal nerve testing. This test uses an electrode to measure pudendal nerve conduction time, known as pudendal nerve terminal motor latency (PNTML)—thus allowing further investigation for nerve injury.
- Normal PNTML value is 2.2 milliseconds.
- A value between 2.2 and 2.6 milliseconds indicates probable nerve damage.
- A value of 2.6 milliseconds or greater confirms nerve damage.
Clinical use of PNTML is controversial; it helps diagnose nerve injury, but is currently reserved for investigational purposes.10
FIGURE 3 Endoanal ultrasound
Disruption of continuity of the inner hypoechoic ring (IS, internal sphincter) and the outer hyperechoic ring (ES, external sphincter) on endoanal ultrasound is consistent with structural damage to the sphincter. This image shows normal anatomy.
Mainstays: Nonsurgical treatments
The general principles of treatment are to identify the underlying cause of incontinence and direct therapy based on these findings. Since much of the disability in these patients is psychological, a supportive medical team is essential. Patient educational books, advice on the use of continent aides, and perianal skin care also are crucial.
The physician and patient must formulate a treatment plan based on the severity of the incontinence and degree of social impairment. In general, a trial of nonsurgical therapy is recommended before proceeding to surgery. A standardized treatment algorithm (FIGURE 4) should be adapted to each patient.
The aim of nonsurgical management is:
- to increase anorectal sensation, and
- to increase the strength of the external sphincter and pelvic floor.
First: Diet and medical therapy. For patients with fecal incontinence, nonsurgical intervention remains the first line of treatment given the high rate of underlying neuromuscular damage.
The aims of medical therapy are to decrease stool frequency and improve stool consistency. The patient’s diet should include adequate fiber and fluid intake to promote regular, soft, bulky stools. High-fiber foods including whole-wheat grains, fresh vegetables, and beans should be advised. Bulking agents like methylcellulose (eg, Citrucel), or psyllium (eg, Metamucil) can be helpful.
Stool frequency can be reduced with antidiarrheal drugs like loperamide (2-4 mg 2 or 3 times daily) or diphenoxalate (eg, Lomotil). Loperamide is usually preferred, since it also increases internal anal sphincter tone. Antiflatulants are also recommended.
Amitryptiline is useful in some women with idiopathic fecal incontinence.11,12
Biofeedback therapy. Pelvic floor rehabilitation with biofeedback and/or electrical stimulation—which focuses on the common neuromuscular etiology of fecal incontinence—is the mainstay of nonsurgical management. Some centers report a success rate as high as 70% with this therapy.13
Biofeedback is a painless, minimally invasive method of retraining pelvic floor muscles with the help of sensors placed in either the vagina or the rectum. This technique improves rectal sensation and muscular tone. It is especially useful in incontinence patients with anatomically intact anal sphincters.
Biofeedback typically involves anal manometry as well as electromyographic sensors. It is generally conducted in an outpatient treatment program that includes regular evaluation of sensation and motor tone. However, home-based units are available.
Investigational: Neuromodulation. Although approved by the US Food and Drug Administration for treatment of urge incontinence and nonobstructive voiding dysfunction, neuromodulation is still investigational for treatment of fecal incontinence—but results are encouraging.14
In this technique, electrodes attached to a portable stimulator intermittently apply low-level electrical impulses to the anal canal via the pelvic nerve supply. Newer methods involve implanting electrodes percutaneously in the S3 or S4 foramina under fluoroscopic guidance, then stimulating the sacral nerves.
Neuromodulation aims to increase maximum squeeze pressure and decrease rectal sensitivity to distention. In early trials, significant improvement of incontinence and urgency symptoms was noted.15,16 Larger and longer-duration trials are needed to elucidate long-term effectiveness.
When to consider surgery
Operative therapy should be considered only for cases in which nonsurgical interventions have failed or in which distinct anatomic defects are present.
Note that most surgical candidates still require physical therapy with biofeedback before or after the procedure.
Multiple surgical procedures have been described; some are technically challenging and should be performed only by skilled surgeons after careful preoperative counseling.
Anterior overlapping external sphincteroplasty is done in patients with obstetric or iatrogenic disruption of the external anal sphincter. In this procedure, the surgeon divides the scar and reapproximates viable sphincter muscle to produce a ring of functional muscle. Few good data exist comparing this approach with end-to-end repairs; anterior sphincteroplasty is our preferred method.
- Controversy exists over the need to plicate the internal anal sphincter. Studies have shown that more extensive repairs offer no additional advantage with regard to out-comes.17,18 Most pelvic reconstructive surgeons prefer to perform colporrhaphy and perineorrhaphy at the time of sphincter repair, since defects in the posterior compartment often coexist in these patients.
The success rate of sphincteroplasty ranges from 40% to 75% in appropriately selected patients.19,20
Patients with satisfactory results demonstrate an increase in squeeze pressure postoperatively. Failure to elicit this response correlated accurately with a persistent sphincter defect.21
Total pelvic floor repair may help patients with neurogenic or idiopathic fecal incontinence. The principle of this procedure is to restore the anorectal angle, lengthen the anal canal, and create a circumferential buttress around the anorectum.
In this rather extensive surgery, the surgeon plicates the puborectalis, ischiococcygeus, and iliococcygeus muscles to the rectum; the levator muscles and the external anal sphincter are plicated anteriorly. Success rates vary from 14% to 55%.22,23
Skeletal muscle transposition surgery, with creation of a neosphincter from the gracilis muscle, is performed in patients with pelvic floor muscle weakness so profound that the above techniques offer no improvement. It should be viewed as a salvage operation.
The gracilis muscle is detached from its insertion, wrapped around the anus, and reattached to the contralateral ischial tuberosity. The success of this surgery may be improved by electrically stimulating the transposed muscle with an implanted stimulator.
This procedure is associated with significant postoperative complications; many patients require multiple reoperations. Thus, this intervention is useful only in patients with refractory incontinence.
Artificial anal sphincter procedure involves placing a cuff around the anal canal and a pressure-regulating balloon and pump in the labia majora. The cuff remains inflated, thereby occluding the anal canal until the patient experiences a desire to defecate, at which time it is deflated.
Results are encouraging, with initial success rates from 60% to 75%.24,25 However, local infection can be problematic.
Less common procedures include anal encirclement (Thiersch procedure), perianal fat injection, antegrade continence enema, and fecal diversion.
A word on prevention
During childbirth. Strategies that may reduce pelvic floor trauma:
- Favor mediolateral over midline episiotomy,
- avoid a prolonged second stage of labor and forceps delivery, and
- repair anal sphincter lacerations with an overlapping technique.
Also, discuss regular pelvic floor exercises, constipation prevention, weight loss, and smoking cessation with all obstetric patients—especially those with fecal incontinence risk factors.
Debate continues about elective cesarean section in women with occult sphincter injury on postpartum endoanal ultrasound, transient symptoms of fecal incontinence with prior delivery, or overt gas incontinence.26 This practice may have clinical utility, but data are insufficient to recommend it routinely.
The authors report no financial relationships relevant to this article.
- Endoanal ultrasound is superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography, and 50% for clinical assessment.
- Pelvic floor rehabilitation with biofeedback/electrical stimulation is the mainstay nonsurgical treatment. Some centers report success rates as high as 70%.
- Consider surgery only for distinct anatomic defects, and only when nonsurgical interventions have not succeeded.
Thanks to improvements in our understanding of the causes of uncontrolled defecation and an increasing array of diagnostic and treatment options, physicians can now manage this condition more effectively.
Fecal incontinence is a relevant clinical issue for Ob/Gyns, and requires compassion and emotional support on the part of the physician and health-care team. This debilitating problem is underreported, but is thought to affect 2.2% of the general population. Women are 8 times more likely to have fecal incontinence than men, in age-controlled groups.1
Although sphincter damage during vaginal delivery—even in deliveries over an intact perineum2,3—may be clinically silent in most women, these women are at risk of developing overt fecal incontinence with subsequent deliveries, and with aging and its decompensating factors (FIGURE 1).
A directed examination, optimal diagnostic testing, and a step-by-step treatment algorithm are the basis of effective management.
FIGURE 1 Common pathway leading to fecal incontinence
How ‘silent’ sphincter damage can lead to incontinence
Obstetric disruption of the anal sphincter structure or function is a common cause of fecal incontinence, although it is usually multifactorial, involving abnormalities of 1 or more continence mechanisms (FIGURE 2).
Weakening of the sphincter mechanism after vaginal delivery is attributed to both direct trauma to the muscle and indirect trauma to the pudendal nerve (unilateral or bilateral). Anatomic injury usually occurs in the anterior portion of the sphincter—a site amenable to direct repair, with good results.
Of the various investigational diagnostic modalities available, endoanal ultrasound has been superior, though not perfect. In 1 series, endoanal sonography detected anal sphincter tears in 35% of primiparous and 40% of multiparous women; of these, 13% and 23%, respectively, had symptoms of fecal incontinence or urgency.4
The pudendal nerve is susceptible to traction injury from childbirth and chronic straining. In a prospective study of the effect of vaginal deliveries on the pelvic floor, 80% of primigravidas sustained reversible pudendal nerve damage—which, in some women, persisted for up to 5 years.5
Obstetric risk factors include:
- infant weighing more than 4,000 g,
- use of forceps,
- prolonged second stage of labor,
- midline episiotomy,
- occipitoposterior position, and
- failed detection or suboptimal repair of anal sphincterinjuryat the time of vaginaldelivery.
Onset of menopause and subsequent pelvic floor weakness and perineal descent aggravates this occult neuropathy.
Minor incontinence is also common in patients with advanced age, internal sphincter defects, prolapsing hemorrhoids, rectal prolapse, and fecal impaction (the rectum chronically distends with feces, continuously inducing internal anal sphincter relaxation and resulting in leakage of stool).
Evaluation: Directed history, 4-point physical exam
A directed history helps indicate which diagnostic tests are most relevant. Establish severity and frequency of fecal incontinence, presence of fecal urgency, pad use, type of incontinence (solid, liquid, or gas), and presence of associated urinary incontinence and prolapse.
Obstetric history should identify:
- parity,
- duration of the second stage of labor,
- occipitoposterior presentation,
- use of forceps,
- extent of vaginal lacerations, and
- weight of the infant.
Dietary habits (especially fiber intake) and chronic laxative use should be noted.
Also note any history of abdominopelvic surgery, radiation, back injury, and neurologic disorders.
4-point physical exam
- Inspect the perineum/perianal area for evidence of dermatitis, infection, fistula, hemorrhoids, and deformities of the anus. Ask the patient to bear down as you look for evidence of pelvic floor weakness and rectal prolapse.
- Assess the anal wink (cutaneoanal contractile reflex) by stroking the skin surrounding the anus. Absence of a reflexive contraction of the external anal sphincter suggests nerve damage or spinal arc interruption.
- Perform a vaginal exam to identify pelvic prolapse and exclude rectovaginal fistula. At this time assess pelvic muscle strength and teach the patient proper pelvic floor exercises.
- Conduct a digital rectal examination to assess the length and orientation of the analcanal, bulk of the perianal tissues, and tone of the anal sphincters, as well as to rule out a mass or impaction. Repeat digital examination with the patient bearing down to assess the squeeze pressures, movement of the puborectalis muscle, and extent of pelvic floor descent.
These findings are predominantly subjective and depend on operator experience. In general, they correlate poorly with objective measurements of sphincter pressures.6
If a rectovaginal fistula is suspected, try retrograde filling of the rectum with 200 cc of dye-stained fluid (using a 50-cc catheter tip syringe), then observe vaginal extravasation.
Diagnostic strategy: Routine endoanal ultrasound, manometry
Our strategy is to routinely proceed with endoanal ultrasound and anal manometry to assess the anal sphincters. We may then recommend sigmoidoscopy or anoscopy to rule out mucosal lesions.
It is important to bear in mind that continence depends on multiple physiologic mechanisms and, therefore, no single diagnostic test yields positive results in all patients.
Endoanal ultrasound has significantly changed the evaluation of fecal incontinence over the last decade. In women with fecal incontinence in whom obstetric injury is suspected, 90% have sonographic evidence of injury to 1 or both sphincters.
These ultrasound images show 2 discrete rings of tissue: The inner hypoechoic ring represents the internal anal sphincter and the outer hyperechoic ring represents the external anal sphincter (FIGURE 3). Disrupted continuity of these rings is consistent with structural damage to the sphincter.
Endoanal ultrasound is regarded as superior to other diagnostic methods, with an accuracy of over 90% in detecting sphincter injuries, compared to 75% for manometry, 75% for electromyography (EMG), and 50% for clinical assessment.7,8 It is the simplest and least-invasive test to assess the structural integrity of the anal sphincters.
It can also serve as a screening tool to detect occult sphincter injury following vaginal delivery, thus identifying women at high risk for future incontinence. Women with transient stool or gas incontinence following vaginal delivery may be candidates for screening endoanal ultrasound for further evaluation.
- Where endoanal ultrasound is unavailable, transperineal ultrasound may be an alternative option.
Anorectal manometry can detect functional weakness of sphincters that are anatomically intact by measuring sensation and squeeze pressures. Patients with intact sphincters but abnormal anal manometry findings may benefit from physical therapy and biofeedback aimed at increasing muscular function by stimulating the neuromuscular feedback loop.
During manometry, the physician assesses maximum resting and squeeze pressures of the anal canal, the rectoanal inhibitory reflex, rectal sensation, and rectal compliance using either water-perfused catheters, solid-state microtransducers, or air-or water-filled balloons. Normal values vary widely among institutions and are poorly standardized.
Decreased resting pressure suggests isolated internal anal sphincter injury; decreased squeeze pressure suggests external anal sphincter injury. Of all parameters, maximum squeeze pressure has the greatest sensitivity and specificity at a cut-off of 60 mm Hg in women.9
Rectal sensory testing also is useful in identifying patients who may benefit from biofeedback therapy, and can detect afferent nerve injury as a contributing cause of incontinence. Biofeedback is unlikely to be useful in patients with a poor or absent sensation. In addition, for patients already involved in biofeedback, the test can assess rectal response to training and indicate rectal irritability or poor compliance.
Rectal sensation is assessed by inflating a balloon in the rectum and recording the smallest volume of rectal distention for first detectable sensation (rectal sensory threshold), sensory urgency, and pain (maximum tolerable volume).
Ancillary tests are of limited benefit as screening tools, but may have utility in special circumstances.
- Electromyography of the external anal sphincter and the pelvic floor muscles—traditionally performed using needle electrodes or surface electrodes—is helpful in delineating areas of sphincter injury by mapping the sphincter. However, much of this information is now obtained accurately by endoanal ultrasound, which has replaced needle EMG for most clinicians. Surface EMG is still useful for evaluating sphincter function and in biofeedback training.
- Defecography involves imaging the rectum after filling it with contrast material, and then observing the process, rate, and completeness of rectal evacuation using fluoroscopy. Its usefulness in the evaluation of fecal incontinence is limited to cases of rectal prolapse and for the diagnosis of rectocele and enterocele.
- Investigational: Pudendal nerve testing. This test uses an electrode to measure pudendal nerve conduction time, known as pudendal nerve terminal motor latency (PNTML)—thus allowing further investigation for nerve injury.
- Normal PNTML value is 2.2 milliseconds.
- A value between 2.2 and 2.6 milliseconds indicates probable nerve damage.
- A value of 2.6 milliseconds or greater confirms nerve damage.
Clinical use of PNTML is controversial; it helps diagnose nerve injury, but is currently reserved for investigational purposes.10
FIGURE 3 Endoanal ultrasound
Disruption of continuity of the inner hypoechoic ring (IS, internal sphincter) and the outer hyperechoic ring (ES, external sphincter) on endoanal ultrasound is consistent with structural damage to the sphincter. This image shows normal anatomy.
Mainstays: Nonsurgical treatments
The general principles of treatment are to identify the underlying cause of incontinence and direct therapy based on these findings. Since much of the disability in these patients is psychological, a supportive medical team is essential. Patient educational books, advice on the use of continent aides, and perianal skin care also are crucial.
The physician and patient must formulate a treatment plan based on the severity of the incontinence and degree of social impairment. In general, a trial of nonsurgical therapy is recommended before proceeding to surgery. A standardized treatment algorithm (FIGURE 4) should be adapted to each patient.
The aim of nonsurgical management is:
- to increase anorectal sensation, and
- to increase the strength of the external sphincter and pelvic floor.
First: Diet and medical therapy. For patients with fecal incontinence, nonsurgical intervention remains the first line of treatment given the high rate of underlying neuromuscular damage.
The aims of medical therapy are to decrease stool frequency and improve stool consistency. The patient’s diet should include adequate fiber and fluid intake to promote regular, soft, bulky stools. High-fiber foods including whole-wheat grains, fresh vegetables, and beans should be advised. Bulking agents like methylcellulose (eg, Citrucel), or psyllium (eg, Metamucil) can be helpful.
Stool frequency can be reduced with antidiarrheal drugs like loperamide (2-4 mg 2 or 3 times daily) or diphenoxalate (eg, Lomotil). Loperamide is usually preferred, since it also increases internal anal sphincter tone. Antiflatulants are also recommended.
Amitryptiline is useful in some women with idiopathic fecal incontinence.11,12
Biofeedback therapy. Pelvic floor rehabilitation with biofeedback and/or electrical stimulation—which focuses on the common neuromuscular etiology of fecal incontinence—is the mainstay of nonsurgical management. Some centers report a success rate as high as 70% with this therapy.13
Biofeedback is a painless, minimally invasive method of retraining pelvic floor muscles with the help of sensors placed in either the vagina or the rectum. This technique improves rectal sensation and muscular tone. It is especially useful in incontinence patients with anatomically intact anal sphincters.
Biofeedback typically involves anal manometry as well as electromyographic sensors. It is generally conducted in an outpatient treatment program that includes regular evaluation of sensation and motor tone. However, home-based units are available.
Investigational: Neuromodulation. Although approved by the US Food and Drug Administration for treatment of urge incontinence and nonobstructive voiding dysfunction, neuromodulation is still investigational for treatment of fecal incontinence—but results are encouraging.14
In this technique, electrodes attached to a portable stimulator intermittently apply low-level electrical impulses to the anal canal via the pelvic nerve supply. Newer methods involve implanting electrodes percutaneously in the S3 or S4 foramina under fluoroscopic guidance, then stimulating the sacral nerves.
Neuromodulation aims to increase maximum squeeze pressure and decrease rectal sensitivity to distention. In early trials, significant improvement of incontinence and urgency symptoms was noted.15,16 Larger and longer-duration trials are needed to elucidate long-term effectiveness.
When to consider surgery
Operative therapy should be considered only for cases in which nonsurgical interventions have failed or in which distinct anatomic defects are present.
Note that most surgical candidates still require physical therapy with biofeedback before or after the procedure.
Multiple surgical procedures have been described; some are technically challenging and should be performed only by skilled surgeons after careful preoperative counseling.
Anterior overlapping external sphincteroplasty is done in patients with obstetric or iatrogenic disruption of the external anal sphincter. In this procedure, the surgeon divides the scar and reapproximates viable sphincter muscle to produce a ring of functional muscle. Few good data exist comparing this approach with end-to-end repairs; anterior sphincteroplasty is our preferred method.
- Controversy exists over the need to plicate the internal anal sphincter. Studies have shown that more extensive repairs offer no additional advantage with regard to out-comes.17,18 Most pelvic reconstructive surgeons prefer to perform colporrhaphy and perineorrhaphy at the time of sphincter repair, since defects in the posterior compartment often coexist in these patients.
The success rate of sphincteroplasty ranges from 40% to 75% in appropriately selected patients.19,20
Patients with satisfactory results demonstrate an increase in squeeze pressure postoperatively. Failure to elicit this response correlated accurately with a persistent sphincter defect.21
Total pelvic floor repair may help patients with neurogenic or idiopathic fecal incontinence. The principle of this procedure is to restore the anorectal angle, lengthen the anal canal, and create a circumferential buttress around the anorectum.
In this rather extensive surgery, the surgeon plicates the puborectalis, ischiococcygeus, and iliococcygeus muscles to the rectum; the levator muscles and the external anal sphincter are plicated anteriorly. Success rates vary from 14% to 55%.22,23
Skeletal muscle transposition surgery, with creation of a neosphincter from the gracilis muscle, is performed in patients with pelvic floor muscle weakness so profound that the above techniques offer no improvement. It should be viewed as a salvage operation.
The gracilis muscle is detached from its insertion, wrapped around the anus, and reattached to the contralateral ischial tuberosity. The success of this surgery may be improved by electrically stimulating the transposed muscle with an implanted stimulator.
This procedure is associated with significant postoperative complications; many patients require multiple reoperations. Thus, this intervention is useful only in patients with refractory incontinence.
Artificial anal sphincter procedure involves placing a cuff around the anal canal and a pressure-regulating balloon and pump in the labia majora. The cuff remains inflated, thereby occluding the anal canal until the patient experiences a desire to defecate, at which time it is deflated.
Results are encouraging, with initial success rates from 60% to 75%.24,25 However, local infection can be problematic.
Less common procedures include anal encirclement (Thiersch procedure), perianal fat injection, antegrade continence enema, and fecal diversion.
A word on prevention
During childbirth. Strategies that may reduce pelvic floor trauma:
- Favor mediolateral over midline episiotomy,
- avoid a prolonged second stage of labor and forceps delivery, and
- repair anal sphincter lacerations with an overlapping technique.
Also, discuss regular pelvic floor exercises, constipation prevention, weight loss, and smoking cessation with all obstetric patients—especially those with fecal incontinence risk factors.
Debate continues about elective cesarean section in women with occult sphincter injury on postpartum endoanal ultrasound, transient symptoms of fecal incontinence with prior delivery, or overt gas incontinence.26 This practice may have clinical utility, but data are insufficient to recommend it routinely.
The authors report no financial relationships relevant to this article.
1. Furner S, Cautley E, Norton N, Nelson R. Community-based prevalence of anal incontinence. JAMA. 1995;274:559.-
2. Sultan AH, Kamm MA, Hudson CN, et al. Anal-sphincter disruption during vaginal delivery. N Engl J Med. 1993;329:1905.-
3. Abramowitz L, Sobhani I, Ganansia R, et al. Are sphincter defects the cause of anal incontinence after vaginal delivery? Results of a prospective study. Dis Colon Rectum. May 2000;43:590-596; discussion:596–598.
4. Fynes M, Donnelly VS, O’Connell PR, O’Herlihy C. Cesarean delivery and anal sphincter injury. Obstet Gynecol. 1998;92:496-500.
5. Haadem K, Dahlstrom JA, Lingman G. Anal sphincter function after delivery: a prospective study in women with sphincter rupture and controls. Eur J Obstet Gynecol Reprod Biol. 1990;35:7.-
6. Coller JA. Clinical application of anorectal manometry. Gastroenterol Clin North Am. 1987;16:17.-
7. Sultan AH, Kamm MA, Talbot IC, et al. Anal endosonography for identifying external sphincter defects confirmed histologically. Br J Surg. 1994;81:463.-
8. Tjandra JJ, Milsom JW, Schroeder T, Fazio VW. Endoluminal ultrasound is preferable to electromyography in mapping anal sphincteric defects. Dis Colon Rectum. 1993;36:689-692.
9. Felt-Bersma RJF, Klinkenberg-Knol EC, Meuwissen SGM. Anorectal function investigations in incontinent and continent patients. Dis Colon Rectum. 1990;33:479.-
10. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
11. Santoro GA, Eitan BZ, Pryde A, Bartolo DC. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis Colon Rectum. 2000;43:1676.-
12. Read M, Read NW, Barber DC, Duthie HL. Effects of loperamide on anal sphincter function in patients complaining of chronic diarrhea with fecal incontinence and urgency. Dig Dis Sci. 1982;27:807.-
13. Ryn AK, Morren GL, Hallbook O, Sjodahl R. Long-term results of electromyo-graphic biofeedback training for fecal incontinence. Dis Colon Rectum. 2000;43:1262-1266
14. Matzel KE, Stadelmaier U, Hohenfellner M, Gall FP. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet. 1995;346:1124.-
15. Matzel KE, Stadelmaier U, Hohenfellner M, Hohenberger W. Chronic sacral spinal nerve stimulation for fecal incontinence: long-term results with foramen and cuff electrodes. Dis Colon Rectum. 2001;44:59.-
16. Rosen HR, Urbarz C, Holzer B, et al. Sacral nerve stimulation as a treatment for fecal incontinence. Gastroenterology. 2001;121:536.-
17. Briel JW, de Boer LM, Hop WC, Schouten WR. Clinical outcome of anterior overlapping external anal sphincter repair with internal anal sphincter imbrication. Dis Colon Rectum. 1998;41:209-214.
18. Laurberg S, Swash M, Henry MM. Effect of postanal repair on progress of neurogenic damage to the pelvic floor. Br J Surg. 1990;77:519-522.
19. Kamm MA. Faecal incontinence. BMJ. 1998;316:528.-
20. Malouf AJ, Norton CS, Engel AF, et al. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355:260.-
21. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
22. Orrom WJ, Miller R, Cornes H, et al. Comparison of anterior sphincteroplasty and postanal repair in the treatment of idiopathic fecal incontinence. Dis Colon Rectum. 1991;34:305-310.
23. Schiller, Lawrence R. Fecal incontinence. In: Feldman M, et al, eds. Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. Philadelphia, Pa: WB Saunders Co; 1997.
24. Giamundo P, Welber A, Weiss EG, et al. The Procon incontinence device: a new nonsurgical approach to preventing episodes of fecal incontinence. Am J Gastroenterol. 2002;97:232.-
25. Wong WD, Congliosi SM, Spencer MP, et al. The safety and efficacy of the artificial bowel sphincter for fecal incontinence: results from a multicenter cohort study. Dis Colon Rectum. 2002;45:1139.-
26. McKenna DS, Ester JB, Fischer JR. Elective cesarean delivery for women with a previous anal sphincter rupture. Am J Obstet Gynecol. 2003;189:1251.-
1. Furner S, Cautley E, Norton N, Nelson R. Community-based prevalence of anal incontinence. JAMA. 1995;274:559.-
2. Sultan AH, Kamm MA, Hudson CN, et al. Anal-sphincter disruption during vaginal delivery. N Engl J Med. 1993;329:1905.-
3. Abramowitz L, Sobhani I, Ganansia R, et al. Are sphincter defects the cause of anal incontinence after vaginal delivery? Results of a prospective study. Dis Colon Rectum. May 2000;43:590-596; discussion:596–598.
4. Fynes M, Donnelly VS, O’Connell PR, O’Herlihy C. Cesarean delivery and anal sphincter injury. Obstet Gynecol. 1998;92:496-500.
5. Haadem K, Dahlstrom JA, Lingman G. Anal sphincter function after delivery: a prospective study in women with sphincter rupture and controls. Eur J Obstet Gynecol Reprod Biol. 1990;35:7.-
6. Coller JA. Clinical application of anorectal manometry. Gastroenterol Clin North Am. 1987;16:17.-
7. Sultan AH, Kamm MA, Talbot IC, et al. Anal endosonography for identifying external sphincter defects confirmed histologically. Br J Surg. 1994;81:463.-
8. Tjandra JJ, Milsom JW, Schroeder T, Fazio VW. Endoluminal ultrasound is preferable to electromyography in mapping anal sphincteric defects. Dis Colon Rectum. 1993;36:689-692.
9. Felt-Bersma RJF, Klinkenberg-Knol EC, Meuwissen SGM. Anorectal function investigations in incontinent and continent patients. Dis Colon Rectum. 1990;33:479.-
10. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
11. Santoro GA, Eitan BZ, Pryde A, Bartolo DC. Open study of low-dose amitriptyline in the treatment of patients with idiopathic fecal incontinence. Dis Colon Rectum. 2000;43:1676.-
12. Read M, Read NW, Barber DC, Duthie HL. Effects of loperamide on anal sphincter function in patients complaining of chronic diarrhea with fecal incontinence and urgency. Dig Dis Sci. 1982;27:807.-
13. Ryn AK, Morren GL, Hallbook O, Sjodahl R. Long-term results of electromyo-graphic biofeedback training for fecal incontinence. Dis Colon Rectum. 2000;43:1262-1266
14. Matzel KE, Stadelmaier U, Hohenfellner M, Gall FP. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet. 1995;346:1124.-
15. Matzel KE, Stadelmaier U, Hohenfellner M, Hohenberger W. Chronic sacral spinal nerve stimulation for fecal incontinence: long-term results with foramen and cuff electrodes. Dis Colon Rectum. 2001;44:59.-
16. Rosen HR, Urbarz C, Holzer B, et al. Sacral nerve stimulation as a treatment for fecal incontinence. Gastroenterology. 2001;121:536.-
17. Briel JW, de Boer LM, Hop WC, Schouten WR. Clinical outcome of anterior overlapping external anal sphincter repair with internal anal sphincter imbrication. Dis Colon Rectum. 1998;41:209-214.
18. Laurberg S, Swash M, Henry MM. Effect of postanal repair on progress of neurogenic damage to the pelvic floor. Br J Surg. 1990;77:519-522.
19. Kamm MA. Faecal incontinence. BMJ. 1998;316:528.-
20. Malouf AJ, Norton CS, Engel AF, et al. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet. 2000;355:260.-
21. Barnett JL, Hasler WL, Camilleri M. American Gastroenterological Association medical position statement on anorectal testing techniques. American Gastroenterological Association. Gastroenterology. 1999;116:732.-
22. Orrom WJ, Miller R, Cornes H, et al. Comparison of anterior sphincteroplasty and postanal repair in the treatment of idiopathic fecal incontinence. Dis Colon Rectum. 1991;34:305-310.
23. Schiller, Lawrence R. Fecal incontinence. In: Feldman M, et al, eds. Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. Philadelphia, Pa: WB Saunders Co; 1997.
24. Giamundo P, Welber A, Weiss EG, et al. The Procon incontinence device: a new nonsurgical approach to preventing episodes of fecal incontinence. Am J Gastroenterol. 2002;97:232.-
25. Wong WD, Congliosi SM, Spencer MP, et al. The safety and efficacy of the artificial bowel sphincter for fecal incontinence: results from a multicenter cohort study. Dis Colon Rectum. 2002;45:1139.-
26. McKenna DS, Ester JB, Fischer JR. Elective cesarean delivery for women with a previous anal sphincter rupture. Am J Obstet Gynecol. 2003;189:1251.-