User login
- Antepartum recognition of fetal growth restriction is essential for proper surveillance and management.
- Because growth-restricted fetuses are at risk for adverse outcomes in utero, fetal surveillance is vital for timely recognition and intervention.
- Once growth restriction is identified, management should be individualized to ensure optimal gestational development and safe delivery.
Not only is fetal growth restriction (FGR) associated with perinatal mortality and morbidity, but it may be linked to adverse consequences in adulthood.1 Its many causes involve diverse pathological processes; thus, it should not be considered a single disease. Many aspects remain unclear, a substantial number of affected infants are not identified before birth, and effective antepartum prevention and treatment remain elusive.
Fortunately, managing FGR has hopeful aspects. Meaningful recent advances elucidate its etiologic and pathophysiologic mechanisms and help clarify diagnosis and management.
This article offers an up-to-date, evidence-based approach and includes guidelines on 3 keys to success: antepartum recognition, fetal surveillance, and antepartum and intrapartum management. These guidelines are not intended as a strict protocol, since the clinical course of FGR is highly variable, but as the starting point for individualized care.
An amorphous entity: FGR terminology
Fetal growth restriction implies failure to realize genetically determined growth potential. Terms include fetal growth restriction, intrauterine growth restriction, and small for gestational age (SGA). The pejorative term growth retardation is obsolete.
Traditionally, FGR refers to prenatally identified fetal growth deficiency, whereas SGA refers to suboptimal birth weight for the gestational age. However, some small fetuses are merely constitutionally small, not growth-restricted. Conversely, not all growth-restricted fetuses are small in size or weight for gestational age. Yet defining these groups is difficult, as the tools are imprecise and controversial.
This article uses these definitions:
Fetal growth restriction identified in the antepartum period refers to a fetus with sonographically measured fetal dimensions, particularly abdominal circumference or estimated weight, below an age-specific threshold, typically the 10th percentile.
Fetal growth restriction identified at birth is birth weight below the 10th percentile for gestational age, or SGA. Unfortunately, this definition may fail to identify some fetuses that are truly growth-restricted. Alternative, more sensitive definitions, such as the Ponderal index or birth weight ratio, are used primarily in research.
Consequences of FGR
Perinatal outcomes. Perinatal morbidities include prematurity, oligohydramnios, nonreassuring fetal heart rate patterns with a higher incidence of cesarean delivery, birth asphyxia, low Apgar score, neonatal hypoglycemia, hypocalcemia, polycythemia, hyperbilirubinemia, hypothermia, apnea, seizure disorders, and infection.
Fetal and neonatal mortality is significantly increased. Perinatal mortality is influenced by many factors, including severity of growth restriction, timing of onset, gestational age, and cause of growth restriction. The lower the birth-weight percentile for gestational age, the higher the mortality rate.
Effects in infancy. Although many SGA infants “catch up” growth in infancy, the pattern varies. Height and weight catch-up growth of preterm FGR infants lags behind that of preterm infants that are appropriate for gestational age (AGA) at birth.2 Those with early-onset or severe growth deficit continue to lag behind in postnatal growth. Although recent studies indicate that rapid postnatal growth in SGA infants may lead to increased risk of chronic diseases, including type 2 diabetes, others have found tangible short-term benefits of less frequent morbidity and mortality in infancy.3
FGR has been linked to a spectrum of neurodevelopmental risks, including subtle behavioral abnormalities, immature sleep patterns, decreased visual fixation, decreased general activity, altered early mother-infant interaction, altered motor skills, and hyperactivity.4,5 Infants born SGA at 32 to 42 weeks were 4 to 6 times more likely to have cerebral palsy, yet those whose birth weight was above the 97th percentile also had increased risk. It remains uncertain whether deviant growth is the cause or a consequence of this disability.6
Long-term effects. Lifetime sequelae of early nutritional deprivation have been demonstrated in animals.7 Moreover, epidemiological evidence suggesting an association between SGA at birth or infancy and increased risk of abnormal blood lipid values, diabetes, hypertension, and ischemic heart disease in adult life led Barker and associates to propose the fetal origins hypothesis.1
1. Antepartum recognitionDetermine gestational age
A reliable estimate of gestational age is central to identification of fetal growth compromise in utero or at birth. In pregnancies at risk for fetal growth restriction, gestational age should be established early, preferably in the first trimester.
The method of determining gestational age influences the observed frequency of FGR and SGA births. Estimates are more accurate when based on early ultrasound biometry than on menstrual history. The latter, if well documented, regular, and ovulatory, may be reliable if it differs by no more than 1 week from the sonographic gestational age. Otherwise, early-pregnancy, ultrasoundbased age is more accurate.
Screening for FGR
Screening can be done clinically or by special investigation. The following methods are used, some of which remain experimental:
- assessment of historical clinical risks
- clinical evaluation of fetal and uterine size
- ultrasound fetal biometry
- umbilical arterial and uterine arterial
Doppler ultrasound
Clinical risk assessment. Evaluate all gravidas for risk factors (TABLE 1). If a woman is determined to be at heightened risk, take appropriate steps to diagnose FGR as early as possible.
Clinical evaluation of fetal and uterine size. Clinical assessment of fetal growth includes estimating fetal size by traditional obstetrical manual examination and by measuring the uterine fundal height.
- Abdominal palpation is inadequate to identify the fetus at increased risk of growth restriction, missing 74% of cases.8
- Serial measurements of the uterine fundal height, however, may be more reliable.9 This method consists of measuring fundal height from the symphysis pubis using a nonstretchable tape measure and assessing the results against a gestational age-specific nomogram. Limitations include significant interobserver differences and varying effects of maternal weight, parity, and fetal sex. A randomized trial of the method did not demonstrate any benefits, and a subsequent Cochrane review of the same study considered the evidence insufficient for any recommendations.10,11
Nevertheless, clinical assessment of uterine and fetal size is an essential, inexpensive component of prenatal care and a simple screening tool for identifying mothers who would benefit from further, more definitive sonographic investigation.
Routine ultrasound biometry. The potential benefits of accurately determined gestational age, and recognition of fetal malformations and multiple gestation, via earlyor mid-pregnancy ultrasound are well recognized and justify widespread use.
Routine scanning increases detection of SGA infants.12 A population-based cohort study13 involving over 16,000 singleton pregnancies found that fetuses smaller than expected at mid-second trimester ultrasound (discrepancy exceeding 14 days) were at increased risk for adverse outcomes, including perinatal mortality and SGA.
Unfortunately, these findings have not led to improved outcomes. A high false-positive rate remains a major concern. A prospective observational study14 found that routine ultrasound did not identify most cases of FGR, but resulted in a fivefold increase in iatrogenic premature delivery and significantly increased neonatal intensive care admissions.
- The Routine Antenatal Diagnostic Imaging with Ultrasound (RADIUS) trial15 randomized 15,000 low-risk gravidas to routine ultrasound imaging (at 15 to 22 weeks and again at 31 to 35 weeks) or to ultrasound only when indicated. The groups had similar rates of adverse perinatal outcome, distribution of birth weights, and preterm delivery. The trial’s weaknesses include selection criteria for low risk that excluded most pregnancies, inappropriate perinatal-outcome parameters, and suboptimal ultrasonographer expertise.
- A recent Cochrane review16 of 7 trials involving more than 25,000 women failed to demonstrate any improvements in perinatal mortality and morbidity with routine ultrasound, or any difference in antenatal, obstetric, and neonatal interventions.
Estimated fetal weight may differ from actual weight by as much as 20%.
We also lack evidence regarding longterm outcomes such as neurodevelopment.
Umbilical arterial Doppler. A meta-analysis17 of 4 randomized trials in unselected or low-risk pregnancies with a total population of 11,375 women found no effect of screening Doppler umbilical artery velocimetry on perinatal death, stillbirth, antenatal hospitalization, obstetric outcome, or perinatal morbidity.
A subsequent meta-analysis18 of 5 trials of routine Doppler ultrasound in unselected and low-risk pregnancies with a total population of more than 14,000 women also found no benefit for mother or infant.
We lack evidence on long-term outcomes.
Uterine arterial Doppler screening. Increased pulsatility of the uterine arterial Doppler waveform, persistence of the notch, and a significant difference between right and left uterine arteries have been associated with FGR, pregnancy-induced hypertension, and adverse perinatal outcome. A review19 of 15 studies of routine uterine Doppler in unselected populations showed considerable heterogeneity, but indicated that increased impedance in the uterine arteries identifies about 20% of those who develop FGR, with a positive likelihood ratio of 3.5.
We lack exclusive randomized trials of routine uterine Doppler sonography in unselected and low-risk pregnancies. However, 2 studies done in conjunction with umbilical arterial Doppler found no impact on outcome, and a recent Cochrane review18 found insufficient evidence to support routine uterine Doppler for FGR screening.
TABLE 1
Risk factors
| MATERNAL FACTORS |
| Medical disease |
| Preeclampsia/hypertension |
| Renal disease |
| Antiphospholipid antibody syndrome |
| Inherited thrombophilia |
| Diabetes with vasculopathy |
| Cyanotic heart disease |
| Asthma |
| Hemoglobinopathy |
| Phenylketonuria |
| Life circumstance |
| Severe malnutrition |
| Smoking |
| Substance abuse (eg, alcohol, heroine, cocaine) |
| PLACENTAL FACTORS |
| Confined placental mosaic |
| Placenta previa |
| Abruptio placentae |
| Infarction |
| Circumvallate placenta |
| Placenta accreta |
| Hemangioma |
| FETAL FACTORS |
| Multiple gestation |
| Prematurity |
| Unexplained elevated alpha-fetoprotein |
| Infection (eg, rubella, cytomegalovirus, herpes, malaria, toxoplasmosis) |
| Malformations (eg, gastroschisis, omphalocele, diaphragmatic hernia, congenital heart defect) |
| Genetic disorders (eg, trisomy 13, 18, and 21; triploidy; some cases of Turner’s syndrome) |
Identifying FGR in utero
Antepartum diagnosis is based on sonographic measurement of various fetal dimensions.
Abdominal circumference and estimated fetal weight. A review20 of 60 studies found that abdominal circumference (AC) and sonographically estimated fetal weight (EFW) were the best predictors of birth weight below the 10th percentile in high-risk pregnancies. AC below the 10th percentile had sensitivities ranging from 72.9% to 94.5%, false-positive rates of 16.2% to 49.4%, and a common odds ratio of 18.4. An EFW below the 10th percentile had a common odds ratio of 39.1, which was the highest among all the biometric measurements. Its sensitivity ranged from 33.3% to 89.2%, and false-positive rates ranged from 9.1% to 46.3%.
The proportionality of fetal dimensions, such as the head/abdominal ratio, was not a good predictor, although it is routinely generated in ultrasound biometry.
Ultrasound estimation of fetal weight is based on a combination of 2 or 3 biometric measurements of the fetus, including the biparietal diameter (BPD) or the head circumference (HC), AC, and femur length (FL). Several formulae yield varying estimations of weight. Thus, a fetus identified as growth-restricted by 1 formula may not be so diagnosed by another. For this reason, it is prudent to be consistent in their use. EFW generated from measurements of the head (BPD or HC), AC, and FL is most reliable.21
The EFW also is expressed as the percentile for the gestational age.
Limitations of ultrasound estimation.
The optimal process of translating dimensional measurements into fetal body mass for both health and disease remains unknown. This leads to inaccurate assumptions and erroneous weight projections.
Inaccuracies also result from variations in measurement. In addition, the estimation is less accurate in the lower and upper ranges of fetal weight distribution and in the presence of oligohydramnios. As a result, estimated weight may differ from actual weight by as much as 20%, with greater margins of error at the lower and upper extremes.
Guidelines for screening and diagnosis
Assess all pregnancies for risk factors and determine the gestational age in early pregnancy, especially in women at higher risk of FGR. If the patient has substantial clinical risks or there is suspicion of growth restriction, fetal ultrasound biometry is recommended. Diagnosis of FGR is based on fetal sonographic measurements, especially AC; estimated fetal weight derived from BPD or HC, AC and FL; and longitudinal progression of fetal growth.
Current evidence suggests the use of a 10th percentile diagnostic threshold for these measurements. Follow the biometric parameters longitudinally, repeating the measurement every 2 to 4 weeks. More frequent examination is unreliable.
2. Fetal surveillanceAntepartum monitoring
Fetuses identified as growth-restricted are at risk for adverse outcomes in utero. Thus, antepartum monitoring is indicated for timely recognition and intervention.
Tests commonly used include the nonstress test (NST), biophysical profile (BPP), sonographic estimation of amniotic fluid volume, and Doppler velocimetry of the umbilical artery or other vessels of interest.
With progressive fetal compromise, compensatory mechanisms may be insufficient to maintain fetal homeostasis. The sequence of loss of compensatory mechanisms is reflected in surveillance tests. Thus, changes in umbil-ical arterial Doppler indices usually precede nonreassuring NST or BPP.
Nonstress test: The standard of care. The NST is probably the most common fetal surveillance test in high-risk pregnancies, including those complicated by FGR. However, it is personnel- and time-intensive, and interpretation often is subjective.
A nonreactive NST is associated with adverse perinatal outcome, although it more commonly is related to the fetal sleep cycle.
In predicting a potentially preventable fetal death, the NST has a false-negative rate of 2 to 3 per 1,000, a negative predictive value of 99.8%, and a false-positive rate of 80%.
Evidence of its efficacy is lacking. Four randomized trials in the early 1980s, which involved more than 1,500 women, found no improvement in outcomes. (All 4 trials lacked sufficient power.)
- When to test. Despite these limitations, the NST has become an integral part of obstetri-cal practice and remains a standard of care for antepartum fetal surveillance in high-risk pregnancies. It should be used as a surveillance tool for the growth-restricted fetus. The test initially is performed weekly.
Depending on severity of growth restriction, testing may increase to twice weekly or even daily.
Amniotic fluid volume monitoring is warranted. Oligohydramnios—highly prevalent in FGR—is associated with adverse perina-tal outcomes.
Although the sonographically determined amniotic fluid index (AFI) is the most frequently used modality to evaluate amniotic fluid volume, its accuracy has been questioned. Nevertheless, a meta-analysis22 of 18 studies involving more than 10,000 patients showed that antepartum AFI equal to or below 5 cm was associated with an increased risk of cesarean section for fetal distress and an Apgar score below 7 at 5 minutes.
Even marginal AFI values (5–10 cm) have been associated with doubling of the risk of adverse perinatal outcome.23
- When to test. We lack randomized trials of amniotic fluid volume assessment in high-risk pregnancies, but observational studies suggest that weekly determination of AFI is helpful in sonographically identified growth-restricted fetuses.
A low or marginal AFI should be followed by more frequent examinations. Even a marginal AFI may be followed by twice-weekly surveillance.
Biophysical profile effectively predicts adverse perinatal outcome, with a false-negative rate of 0.8 per 1,000, negative predictive value of 99.9%, and a false-positive rate of 40% to 50%.24 A low BPP is associated with fetal hypoxia and acidosis, and a declining BPP reflects progressive worsening of the fetal condition.
In FGR, a significant association exists between abnormal Doppler indices and fetal acid-base compromise.
Although equal weight was originally accorded all the parameters of the profile, experience suggests that the independent risk of oligohydramnios warrants immediate reassessment of the management plan.
Like the nonstress test, the BPP has a low false-negative rate and a high false-positive rate. Four randomized trials of the test have been conducted, involving about 2,800 patients, with no confirmation of its effectiveness. A Cochrane systematic review25 concluded that this cumulative sample size is insufficient to draw definitive conclusions.
The test remains a standard, however, and is recommended to confirm fetal wellbeing or as part of a comprehensive test panel in significantly high-risk pregnancies.
- When to test. Initially perform the test once a week, increasing to twice weekly when the severity of growth restriction warrants.
Daily testing may be indicated in complications such as severe growth restriction and pregestational diabetes. In early preterm pregnancies (less than 32 weeks) with nonreassuring NST or umbilical arterial Doppler, BPP can help guide optimal management.
Umbilical arterial Doppler ultrasound is a powerful predictor of adverse perinatal outcomes in high-risk pregnancies.
Doppler indices include the pulsatility index, systolic/diastolic ratio, diastolic average ratio, and the resistance index. Of these, the last demonstrated the greatest ability to predict abnormal perinatal outcomes.
A prospective, blinded study26 in highrisk pregnancies demonstrated significant diagnostic efficacy with a sensitivity of 79%, specificity of 93%, positive predictive value of 83%, negative predictive value of 91%, and kappa index of 73%. The last value is consistent with a good to excellent diagnostic test.
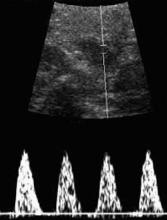
A significant association also exists between abnormal Doppler indices and fetal acid-base compromise in FGR. The absence of enddiastolic flow (FIGURE) is associated with markedly adverse perinatal outcome, particularly a high perinatal mortality rate, malformations, and aneuploidy. Nicolaides and coworkers27 found that, when the end-diastolic velocity was absent, 67% to 80% of the fetuses were hypoxic and 45% were acidotic. Reversal of end-diastolic flow is associated with an even worse prognosis. The adverse consequences of absent or reverse end-diastolic flow are listed in TABLE 2.
In contrast to the NST and BPP, the effectiveness of fetal surveillance with umbilical arterial Doppler ultrasound in improving perinatal outcome in high-risk pregnancies has been confirmed by extensive randomized trials and their meta-analyses.28,29 These studies provide compelling evidence that, when used in well-defined high-risk pregnancies, especially those involving FGR or preeclampsia, umbilical arterial Doppler sonography reduces perinatal death, cesarean delivery for fetal distress, elective deliveries, and antenatal admissions.
Doppler sonography of the fetal cerebral and venous circulation also helps identify progressive fetal compromise in high-risk pregnancies, including those with FGR. When the fetal condition is deteriorating, cerebral arteries dilate to compensate, and pulsations appear in the umbilical vein with reversal of flow in the ductus venous.
These signs of grave fetal status indicate immediate delivery.
FIGURE 1 Duplex pulsed Doppler
Sonogram showing absent end-diastolic flow in the umbilical artery of a growth-restricted fetus.TABLE 2
Perinatal outcomes for absent and reversed end-diastolic velocity in the umbilical artery
| PERINATAL OUTCOME | MEAN | RANGE |
|---|---|---|
| Mortality | 45% | 17–100% |
| Gestational age | 31.6 weeks | 29–33 weeks |
| Birth weight | 1,056 g | 910–1,481 g |
| SGA | 68% | 53–100% |
| Cesarean section for fetal distress | 73% | 24–100% |
| Apgar score <7 at 5 minutes | 26% | 7–69% |
| Admission to NICU | 84% | 77–97% |
| Congenital anomalies | 10% | 0–24% |
| Aneuploidy | 6.4% | 0–18% |
| SGA = small for gestational age; NICU = neonatal intensive care unit | ||
| Reprinted with permission from: Maulik D, ed. Doppler Ultrasound in Obstetrics and Gynecology. | ||
| New York: Springer-Verlag; 1997:364, Table 21.2. | ||
Assessing the fetus for malformation and aneuploidy
Because of the association between FGR and fetal malformations and aneuploidy, the fetus should be assessed for these complications, especially when growth restriction is severe, develops early, and is not associated with hypertensive disorders, oligohydramnios, or abnormal umbilical arterial Doppler indices.
Most etiologies are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother.
The current standard practice is aneuploidy screening in early pregnancy and fetal anatomical scanning at midgestation. If significant risks of aneuploidy are present, a fetal karyotype is recommended, along with appropriate counseling.
3. Individualized managementAntepartum treatment
The question of bed rest. At present, there are no grounds for recommending strict bed rest in FGR cases. A Cochrane review30 showed no improvements with hospitalized bed rest compared with ambulation. Prolonged rest increases risk of thromboembolism and can be costly and inconvenient.
Nutritional remedies. Of the options that have been tested, which include high protein supplementation, nutrient supplementation such as beef liver extract, and balanced energy/protein supplementation, only the last (with protein content comprising less than 25% of total energy content) has led to a significant reduction in SGA births.31-33
Other approaches. We lack reliable evidence that other interventions such as oxygen administration, abdominal decompression, and pharmacological agents, including calcium channel blockers, beta mimetics, and magnesium, are beneficial or effective in improving or preventing FGR.
A meta-analysis34 of 13 trials involving more than 13,000 women showed that early aspirin treatment reduced the risk of FGR but failed to improve outcome. A more recent meta-analysis35 of 38 trials of aspirin in highrisk pregnancies found no reduction in the incidence of FGR or perinatal death, although a reduction in risk of preterm births was noted.
Etiologic management
Most etiologic conditions are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother. An example is maternal hypertensive disease, in which the indicated treatment has no beneficial effect on fetal growth.
Treatment of poor lifestyle habits may be helpful. If the mother smokes, vigorous smoking-cessation education and counseling is urged. Also address alcohol consumption and other substance abuse, and offer remedial measures.
The diagnosis of fetal viral and parasitic infections is important for prognostication and neonatal management. Although few perinatal infections are treatable in utero, maternal therapy may prevent certain infections from spreading to the fetus. Examples include toxoplasmosis and malaria.
If lethal malformations or lethal aneuploidy are identified prenatally, avoid fetal surveillance and unnecessary intervention, which may expose the mother to unnecessary and unjustifiable risks.
Fetal surveillance strategy
If fetal biometry indicates fetal weight below the 10th percentile, begin fetal surveillance. The current standard, umbilical arterial Doppler sonography, is the primary test in sonographically documented growth restriction. Also assess amniotic fluid volume—as part of the BPP or independently. AFI is the most commonly used BPP tool.
In the United States, the nonstress test traditionally is the primary monitoring modality, with the BPP as backup, although the BPP—which includes the NST—has been recommended as the primary test.
Normal Doppler findings. When the Doppler index remains within normal limits or does not progressively rise, weekly testing should suffice, with the NST or BPP as backup or in conjunction with Doppler.
If fetal and maternal conditions remain reassuring, allow the pregnancy to continue to maturity and assess the patient for delivery. Postdate pregnancy is not advised in the presence of sonographically confirmed growth compromise.
A high or increasing Doppler index warrants more intensive fetal surveillance consisting of weekly umbilical arterial Doppler and once- or twice-weekly NST and BPP until fetal maturity.
If these tests indicate fetal compromise, or absent end-diastolic velocity develops, the likelihood of poor perinatal outcome is increased and an urgent clinical response is indicated. Hospitalize the patient and individualize management, depending on gestational age and fetal status.
Optimal timing of delivery
The optimal timing of delivery in a preterm pregnancy with FGR is unclear. A recent multicenter randomized controlled trial, the Growth Restriction Intervention Trial (GRIT),36 compared 2 strategies: delivery within 48 hours with steroid administration or delivery delayed as long as fetal status permits. The population consisted of high-risk gravidas between 24 and 36 weeks’ gestation. More than 90% of the women had pregnancies complicated by FGR. No significant differences were noted between the 2 groups in stillbirth rates.
Fortunately, thanks to recent advances in perinatal care, a management strategy can be recommended:
At or near term (34 weeks or beyond), the absence of end-diastolic flow in the umbilical artery should prompt consideration of immediate delivery.
Other ominous findings that prompt such consideration include:
- Cessation of fetal growth on successive ultrasound examinations
- Progression of umbilical arterial absent enddiastolic flow to reversed end-diastolic flow
- Nonreassuring heart rate patterns including nonreactive NST, poor fetal heart rate baseline variability, and persistent variable or late decelerations
- Oligohydramnios
- BPP score of 4 or below
Less than 34 weeks. When absent enddiastolic flow develops in a preterm pregnancy with a significant risk of fetal lung immaturity, seek further assurance of fetal well-being via daily surveillance with umbilical arterial Doppler sonography, nonstress test, and biophysical profile. Administer betamethasone to enhance fetal lung maturity. Delivery is indicated regardless of maturity when a single test or combination indicates imminent fetal danger and the fetal risk from a hostile intrauterine environment is judged to be greater than that from pulmonary immaturity.
Intrapartum management
Labor and delivery of growth-restricted infants often is associated with a higher risk of asphyxia. The frequency of nonreassuring fetal heart rate patterns is increased. In addition, because of the greater prevalence of oligohydramnios, cord compression is frequent and associated with variable decelerations. There also is an increased risk of neonatal morbidities, as discussed above. Because of these risks, the growth-restricted fetus should be delivered in a facility that can offer appropriate inhouse support services such as neonatology and anesthesia.
If the patient is allowed to labor, use continuous electronic fetal heart rate monitoring with vigilance and perform the appropriate secondary tests, such as fetal scalp sampling or scalp stimulation. The ability of a growth-restricted fetus to tolerate labor is likely very limited.
Any confirmed signs of fetal compromise indicate cesarean as the optimal mode of delivery.
The authors report no financial relationships relevant to this article.
1. Barker DJP, ed. Fetal and Infant Origins of Adult Disease. London, UK: BMJ Publishing; 1992.
2. Strauss RS, Dietz WH. Effects of intrauterine growth retardation in premature infants on early childhood growth. J Pediatr. 1997;13:95-102.
3. Victora CG, Barros FC, Horta BL, Martorell R. Short-term benefits of catch-up growth for small-for-gestational-age infants. Int J Epidemiol. 2001;30:1325-1330.
4. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on the fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
5. Low JA, Handley-Derry MH, Burke SO, et al. Association of intrauterine fetal growth retardation and learning deficits at age 9 to 11 years. Am J Obstet Gynecol. 1992;167:1499-1505.
6. Jarvis S, Glinianaia SV, Torrioli MG, et al. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003;362:1106-1111.
7. Lucas A. Programming by early nutrition: an experimental approach. J Nutr. 1998;128(suppl 2):401-406S.
8. Hepburn M, Rosenberg K. An audit of the detection and management of small-for-gestational age babies. Br J Obstet Gynaecol. 1986;93:212-216.
9. Belizan JM, Villar J, Nardin JC, Malamud J, Sainz de Vicuna L. Diagnosis of intrauterine growth retardation by a simple clinical method: measurement of uterine height. Am J Obstet Gynecol. 1978;131:643-646.
10. Lindhard A, Nielsen PV, Mouritsen LA, Zachariassen A, Sorensen HU, Roseno H. The implications of introducing the symphyseal-fundal height measurement. A prospective randomized controlled trial. Br J Obstet Gynaecol. 1990;97:675-680.
11. Neilson JP. Symphysis-fundal height measurement in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD000944.-
12. Hughey MJ. Routine ultrasound for detection and management of the small for-gestational-age fetus. Obstet Gynecol. 1984;64:101-107.
13. Nakling J, Backe B. Adverse obstetric outcome in fetuses that are smaller than expected at second trimester routine ultrasound examination. Acta Obstet Gynecol Scand. 2002;81:846-851.
14. Jahn A, Razum O, Berle P. Routine screening for intrauterine growth retardation in Germany: low sensitivity and questionable benefit for diagnosed cases. Acta Obstet Gynecol Scand. 2002;81:846-851.
15. Ewigman BG, Crane JP, Frigoletto FD, LeFevre ML, Bain RP, McNellis D. Effect of prenatal ultrasound screening on perinatal outcome. RADIUS Study Group. N Engl J Med. 1993; 16;329:821-827.
16. Bricker L, Neilson JP. Routine ultrasound in late pregnancy (after 24 weeks gestation). Cochrane Database Syst Rev. 2000;(2):CD001451.-
17. Goffinet F, Paris Llado J, Nisand I, Breart G. Umbilical artery Doppler velocimetry in unselected and low risk pregnancies: a review of randomized controlled trials. Br J Obstet Gynaecol. 1997;104:425-430.
18. Bricker L, Neilson JP. Routine Doppler ultrasound in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD001450.-
19. Papageorghiou AT, Yu CK, Cicero S, Bower S, Nicolaides KH. Second-trimester uterine artery Doppler screening in unselected populations: a review. J Matern Fetal Neonatal Med. 2002;12:78-88.
20. Chang TC, Robson SC, Boys RJ, Spencer JA. Prediction of the small for gestational age infant: which ultrasonic measurement is best? Obstet Gynecol. 1992;80:1030-1038.
21. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151:333-337.
22. Chauhan SP, Sanderson M, Hendrix NW, Magann EF, Devoe LD. Perinatal outcome and amniotic fluid index in the antepartum and intrapartum periods: a meta-analysis. Am J Obstet Gynecol. 1999;18:1473-1478.
23. Banks EH, Miller DA. Perinatal risks associated with borderline amniotic fluid index. Am J Obstet Gynecol. 1999;180:1461-1463.
24. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
25. Alfirevic Z, Neilson JP. Biophysical profile for fetal assessment in high risk pregnancies. Cochrane Database Syst Rev. 2003;(1).:The Cochrane Collaboration.
26. Maulik D, Yarlagadda P, Youngblood JP, et al. The diagnostic efficacy of the UA systolic/diastolic ratio as a screening tool a prospective blinded study. Am J Obstet Gynecol. 1990;162:1518-1523.
27. Nicolaides KH, Bilardo CM, Soothill PW, et al. Absence of end diastolic frequencies in umbilical artery: a sign of fetal hypoxia and acidosis. B Med J. 1988;297:1026-1027.
28. Alfirevic Z, Neilson JP. Doppler ultrasonography in high-risk pregnancies: systematic review with meta-analysis. Am J Obstet Gynecol. 1995;172:1379-1387.
29. Westergaard HB, Langhoff-Roos J, Lingman G, Marsal K, Kreiner S. A critical appraisal of the use of umbilical artery Doppler ultrasound in high-risk pregnancies: use of meta-analyses in evidence-based obstetrics. Ultrasound Obstet Gynecol. 2001;17:464-465.
30. Gulmezoglu AM, Hofmeyr GJ. Bed rest in hospital for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
31. Kramer MS. High protein supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
32. Kramer MS. Balanced protein/energy supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
33. Gulmezoglu AM, Hofmeyr GJ. Maternal nutrient supplementation for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
34. Leitich H, Egarter C, Husslein P, Kaider A, Schemper M. A meta-analysis of low dose aspirin for the prevention of intrauterine growth retardation. Br J Obstet Gynaecol. 1997;104:450-459.
35. Kozer E, Costei AM, Boskovic R, Nulman I, Nikfar S, Koren G. Effects of aspirin consumption during pregnancy on pregnancy outcomes: meta-analysis. Birth Defects Res Part B Dev Reprod Toxicol. 2003;68:70-84.
36. The GRIT Study Group. A randomized trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. BJOG. 2003;110:27-32.
- Antepartum recognition of fetal growth restriction is essential for proper surveillance and management.
- Because growth-restricted fetuses are at risk for adverse outcomes in utero, fetal surveillance is vital for timely recognition and intervention.
- Once growth restriction is identified, management should be individualized to ensure optimal gestational development and safe delivery.
Not only is fetal growth restriction (FGR) associated with perinatal mortality and morbidity, but it may be linked to adverse consequences in adulthood.1 Its many causes involve diverse pathological processes; thus, it should not be considered a single disease. Many aspects remain unclear, a substantial number of affected infants are not identified before birth, and effective antepartum prevention and treatment remain elusive.
Fortunately, managing FGR has hopeful aspects. Meaningful recent advances elucidate its etiologic and pathophysiologic mechanisms and help clarify diagnosis and management.
This article offers an up-to-date, evidence-based approach and includes guidelines on 3 keys to success: antepartum recognition, fetal surveillance, and antepartum and intrapartum management. These guidelines are not intended as a strict protocol, since the clinical course of FGR is highly variable, but as the starting point for individualized care.
An amorphous entity: FGR terminology
Fetal growth restriction implies failure to realize genetically determined growth potential. Terms include fetal growth restriction, intrauterine growth restriction, and small for gestational age (SGA). The pejorative term growth retardation is obsolete.
Traditionally, FGR refers to prenatally identified fetal growth deficiency, whereas SGA refers to suboptimal birth weight for the gestational age. However, some small fetuses are merely constitutionally small, not growth-restricted. Conversely, not all growth-restricted fetuses are small in size or weight for gestational age. Yet defining these groups is difficult, as the tools are imprecise and controversial.
This article uses these definitions:
Fetal growth restriction identified in the antepartum period refers to a fetus with sonographically measured fetal dimensions, particularly abdominal circumference or estimated weight, below an age-specific threshold, typically the 10th percentile.
Fetal growth restriction identified at birth is birth weight below the 10th percentile for gestational age, or SGA. Unfortunately, this definition may fail to identify some fetuses that are truly growth-restricted. Alternative, more sensitive definitions, such as the Ponderal index or birth weight ratio, are used primarily in research.
Consequences of FGR
Perinatal outcomes. Perinatal morbidities include prematurity, oligohydramnios, nonreassuring fetal heart rate patterns with a higher incidence of cesarean delivery, birth asphyxia, low Apgar score, neonatal hypoglycemia, hypocalcemia, polycythemia, hyperbilirubinemia, hypothermia, apnea, seizure disorders, and infection.
Fetal and neonatal mortality is significantly increased. Perinatal mortality is influenced by many factors, including severity of growth restriction, timing of onset, gestational age, and cause of growth restriction. The lower the birth-weight percentile for gestational age, the higher the mortality rate.
Effects in infancy. Although many SGA infants “catch up” growth in infancy, the pattern varies. Height and weight catch-up growth of preterm FGR infants lags behind that of preterm infants that are appropriate for gestational age (AGA) at birth.2 Those with early-onset or severe growth deficit continue to lag behind in postnatal growth. Although recent studies indicate that rapid postnatal growth in SGA infants may lead to increased risk of chronic diseases, including type 2 diabetes, others have found tangible short-term benefits of less frequent morbidity and mortality in infancy.3
FGR has been linked to a spectrum of neurodevelopmental risks, including subtle behavioral abnormalities, immature sleep patterns, decreased visual fixation, decreased general activity, altered early mother-infant interaction, altered motor skills, and hyperactivity.4,5 Infants born SGA at 32 to 42 weeks were 4 to 6 times more likely to have cerebral palsy, yet those whose birth weight was above the 97th percentile also had increased risk. It remains uncertain whether deviant growth is the cause or a consequence of this disability.6
Long-term effects. Lifetime sequelae of early nutritional deprivation have been demonstrated in animals.7 Moreover, epidemiological evidence suggesting an association between SGA at birth or infancy and increased risk of abnormal blood lipid values, diabetes, hypertension, and ischemic heart disease in adult life led Barker and associates to propose the fetal origins hypothesis.1
1. Antepartum recognitionDetermine gestational age
A reliable estimate of gestational age is central to identification of fetal growth compromise in utero or at birth. In pregnancies at risk for fetal growth restriction, gestational age should be established early, preferably in the first trimester.
The method of determining gestational age influences the observed frequency of FGR and SGA births. Estimates are more accurate when based on early ultrasound biometry than on menstrual history. The latter, if well documented, regular, and ovulatory, may be reliable if it differs by no more than 1 week from the sonographic gestational age. Otherwise, early-pregnancy, ultrasoundbased age is more accurate.
Screening for FGR
Screening can be done clinically or by special investigation. The following methods are used, some of which remain experimental:
- assessment of historical clinical risks
- clinical evaluation of fetal and uterine size
- ultrasound fetal biometry
- umbilical arterial and uterine arterial
Doppler ultrasound
Clinical risk assessment. Evaluate all gravidas for risk factors (TABLE 1). If a woman is determined to be at heightened risk, take appropriate steps to diagnose FGR as early as possible.
Clinical evaluation of fetal and uterine size. Clinical assessment of fetal growth includes estimating fetal size by traditional obstetrical manual examination and by measuring the uterine fundal height.
- Abdominal palpation is inadequate to identify the fetus at increased risk of growth restriction, missing 74% of cases.8
- Serial measurements of the uterine fundal height, however, may be more reliable.9 This method consists of measuring fundal height from the symphysis pubis using a nonstretchable tape measure and assessing the results against a gestational age-specific nomogram. Limitations include significant interobserver differences and varying effects of maternal weight, parity, and fetal sex. A randomized trial of the method did not demonstrate any benefits, and a subsequent Cochrane review of the same study considered the evidence insufficient for any recommendations.10,11
Nevertheless, clinical assessment of uterine and fetal size is an essential, inexpensive component of prenatal care and a simple screening tool for identifying mothers who would benefit from further, more definitive sonographic investigation.
Routine ultrasound biometry. The potential benefits of accurately determined gestational age, and recognition of fetal malformations and multiple gestation, via earlyor mid-pregnancy ultrasound are well recognized and justify widespread use.
Routine scanning increases detection of SGA infants.12 A population-based cohort study13 involving over 16,000 singleton pregnancies found that fetuses smaller than expected at mid-second trimester ultrasound (discrepancy exceeding 14 days) were at increased risk for adverse outcomes, including perinatal mortality and SGA.
Unfortunately, these findings have not led to improved outcomes. A high false-positive rate remains a major concern. A prospective observational study14 found that routine ultrasound did not identify most cases of FGR, but resulted in a fivefold increase in iatrogenic premature delivery and significantly increased neonatal intensive care admissions.
- The Routine Antenatal Diagnostic Imaging with Ultrasound (RADIUS) trial15 randomized 15,000 low-risk gravidas to routine ultrasound imaging (at 15 to 22 weeks and again at 31 to 35 weeks) or to ultrasound only when indicated. The groups had similar rates of adverse perinatal outcome, distribution of birth weights, and preterm delivery. The trial’s weaknesses include selection criteria for low risk that excluded most pregnancies, inappropriate perinatal-outcome parameters, and suboptimal ultrasonographer expertise.
- A recent Cochrane review16 of 7 trials involving more than 25,000 women failed to demonstrate any improvements in perinatal mortality and morbidity with routine ultrasound, or any difference in antenatal, obstetric, and neonatal interventions.
Estimated fetal weight may differ from actual weight by as much as 20%.
We also lack evidence regarding longterm outcomes such as neurodevelopment.
Umbilical arterial Doppler. A meta-analysis17 of 4 randomized trials in unselected or low-risk pregnancies with a total population of 11,375 women found no effect of screening Doppler umbilical artery velocimetry on perinatal death, stillbirth, antenatal hospitalization, obstetric outcome, or perinatal morbidity.
A subsequent meta-analysis18 of 5 trials of routine Doppler ultrasound in unselected and low-risk pregnancies with a total population of more than 14,000 women also found no benefit for mother or infant.
We lack evidence on long-term outcomes.
Uterine arterial Doppler screening. Increased pulsatility of the uterine arterial Doppler waveform, persistence of the notch, and a significant difference between right and left uterine arteries have been associated with FGR, pregnancy-induced hypertension, and adverse perinatal outcome. A review19 of 15 studies of routine uterine Doppler in unselected populations showed considerable heterogeneity, but indicated that increased impedance in the uterine arteries identifies about 20% of those who develop FGR, with a positive likelihood ratio of 3.5.
We lack exclusive randomized trials of routine uterine Doppler sonography in unselected and low-risk pregnancies. However, 2 studies done in conjunction with umbilical arterial Doppler found no impact on outcome, and a recent Cochrane review18 found insufficient evidence to support routine uterine Doppler for FGR screening.
TABLE 1
Risk factors
| MATERNAL FACTORS |
| Medical disease |
| Preeclampsia/hypertension |
| Renal disease |
| Antiphospholipid antibody syndrome |
| Inherited thrombophilia |
| Diabetes with vasculopathy |
| Cyanotic heart disease |
| Asthma |
| Hemoglobinopathy |
| Phenylketonuria |
| Life circumstance |
| Severe malnutrition |
| Smoking |
| Substance abuse (eg, alcohol, heroine, cocaine) |
| PLACENTAL FACTORS |
| Confined placental mosaic |
| Placenta previa |
| Abruptio placentae |
| Infarction |
| Circumvallate placenta |
| Placenta accreta |
| Hemangioma |
| FETAL FACTORS |
| Multiple gestation |
| Prematurity |
| Unexplained elevated alpha-fetoprotein |
| Infection (eg, rubella, cytomegalovirus, herpes, malaria, toxoplasmosis) |
| Malformations (eg, gastroschisis, omphalocele, diaphragmatic hernia, congenital heart defect) |
| Genetic disorders (eg, trisomy 13, 18, and 21; triploidy; some cases of Turner’s syndrome) |
Identifying FGR in utero
Antepartum diagnosis is based on sonographic measurement of various fetal dimensions.
Abdominal circumference and estimated fetal weight. A review20 of 60 studies found that abdominal circumference (AC) and sonographically estimated fetal weight (EFW) were the best predictors of birth weight below the 10th percentile in high-risk pregnancies. AC below the 10th percentile had sensitivities ranging from 72.9% to 94.5%, false-positive rates of 16.2% to 49.4%, and a common odds ratio of 18.4. An EFW below the 10th percentile had a common odds ratio of 39.1, which was the highest among all the biometric measurements. Its sensitivity ranged from 33.3% to 89.2%, and false-positive rates ranged from 9.1% to 46.3%.
The proportionality of fetal dimensions, such as the head/abdominal ratio, was not a good predictor, although it is routinely generated in ultrasound biometry.
Ultrasound estimation of fetal weight is based on a combination of 2 or 3 biometric measurements of the fetus, including the biparietal diameter (BPD) or the head circumference (HC), AC, and femur length (FL). Several formulae yield varying estimations of weight. Thus, a fetus identified as growth-restricted by 1 formula may not be so diagnosed by another. For this reason, it is prudent to be consistent in their use. EFW generated from measurements of the head (BPD or HC), AC, and FL is most reliable.21
The EFW also is expressed as the percentile for the gestational age.
Limitations of ultrasound estimation.
The optimal process of translating dimensional measurements into fetal body mass for both health and disease remains unknown. This leads to inaccurate assumptions and erroneous weight projections.
Inaccuracies also result from variations in measurement. In addition, the estimation is less accurate in the lower and upper ranges of fetal weight distribution and in the presence of oligohydramnios. As a result, estimated weight may differ from actual weight by as much as 20%, with greater margins of error at the lower and upper extremes.
Guidelines for screening and diagnosis
Assess all pregnancies for risk factors and determine the gestational age in early pregnancy, especially in women at higher risk of FGR. If the patient has substantial clinical risks or there is suspicion of growth restriction, fetal ultrasound biometry is recommended. Diagnosis of FGR is based on fetal sonographic measurements, especially AC; estimated fetal weight derived from BPD or HC, AC and FL; and longitudinal progression of fetal growth.
Current evidence suggests the use of a 10th percentile diagnostic threshold for these measurements. Follow the biometric parameters longitudinally, repeating the measurement every 2 to 4 weeks. More frequent examination is unreliable.
2. Fetal surveillanceAntepartum monitoring
Fetuses identified as growth-restricted are at risk for adverse outcomes in utero. Thus, antepartum monitoring is indicated for timely recognition and intervention.
Tests commonly used include the nonstress test (NST), biophysical profile (BPP), sonographic estimation of amniotic fluid volume, and Doppler velocimetry of the umbilical artery or other vessels of interest.
With progressive fetal compromise, compensatory mechanisms may be insufficient to maintain fetal homeostasis. The sequence of loss of compensatory mechanisms is reflected in surveillance tests. Thus, changes in umbil-ical arterial Doppler indices usually precede nonreassuring NST or BPP.
Nonstress test: The standard of care. The NST is probably the most common fetal surveillance test in high-risk pregnancies, including those complicated by FGR. However, it is personnel- and time-intensive, and interpretation often is subjective.
A nonreactive NST is associated with adverse perinatal outcome, although it more commonly is related to the fetal sleep cycle.
In predicting a potentially preventable fetal death, the NST has a false-negative rate of 2 to 3 per 1,000, a negative predictive value of 99.8%, and a false-positive rate of 80%.
Evidence of its efficacy is lacking. Four randomized trials in the early 1980s, which involved more than 1,500 women, found no improvement in outcomes. (All 4 trials lacked sufficient power.)
- When to test. Despite these limitations, the NST has become an integral part of obstetri-cal practice and remains a standard of care for antepartum fetal surveillance in high-risk pregnancies. It should be used as a surveillance tool for the growth-restricted fetus. The test initially is performed weekly.
Depending on severity of growth restriction, testing may increase to twice weekly or even daily.
Amniotic fluid volume monitoring is warranted. Oligohydramnios—highly prevalent in FGR—is associated with adverse perina-tal outcomes.
Although the sonographically determined amniotic fluid index (AFI) is the most frequently used modality to evaluate amniotic fluid volume, its accuracy has been questioned. Nevertheless, a meta-analysis22 of 18 studies involving more than 10,000 patients showed that antepartum AFI equal to or below 5 cm was associated with an increased risk of cesarean section for fetal distress and an Apgar score below 7 at 5 minutes.
Even marginal AFI values (5–10 cm) have been associated with doubling of the risk of adverse perinatal outcome.23
- When to test. We lack randomized trials of amniotic fluid volume assessment in high-risk pregnancies, but observational studies suggest that weekly determination of AFI is helpful in sonographically identified growth-restricted fetuses.
A low or marginal AFI should be followed by more frequent examinations. Even a marginal AFI may be followed by twice-weekly surveillance.
Biophysical profile effectively predicts adverse perinatal outcome, with a false-negative rate of 0.8 per 1,000, negative predictive value of 99.9%, and a false-positive rate of 40% to 50%.24 A low BPP is associated with fetal hypoxia and acidosis, and a declining BPP reflects progressive worsening of the fetal condition.
In FGR, a significant association exists between abnormal Doppler indices and fetal acid-base compromise.
Although equal weight was originally accorded all the parameters of the profile, experience suggests that the independent risk of oligohydramnios warrants immediate reassessment of the management plan.
Like the nonstress test, the BPP has a low false-negative rate and a high false-positive rate. Four randomized trials of the test have been conducted, involving about 2,800 patients, with no confirmation of its effectiveness. A Cochrane systematic review25 concluded that this cumulative sample size is insufficient to draw definitive conclusions.
The test remains a standard, however, and is recommended to confirm fetal wellbeing or as part of a comprehensive test panel in significantly high-risk pregnancies.
- When to test. Initially perform the test once a week, increasing to twice weekly when the severity of growth restriction warrants.
Daily testing may be indicated in complications such as severe growth restriction and pregestational diabetes. In early preterm pregnancies (less than 32 weeks) with nonreassuring NST or umbilical arterial Doppler, BPP can help guide optimal management.
Umbilical arterial Doppler ultrasound is a powerful predictor of adverse perinatal outcomes in high-risk pregnancies.
Doppler indices include the pulsatility index, systolic/diastolic ratio, diastolic average ratio, and the resistance index. Of these, the last demonstrated the greatest ability to predict abnormal perinatal outcomes.
A prospective, blinded study26 in highrisk pregnancies demonstrated significant diagnostic efficacy with a sensitivity of 79%, specificity of 93%, positive predictive value of 83%, negative predictive value of 91%, and kappa index of 73%. The last value is consistent with a good to excellent diagnostic test.
A significant association also exists between abnormal Doppler indices and fetal acid-base compromise in FGR. The absence of enddiastolic flow (FIGURE) is associated with markedly adverse perinatal outcome, particularly a high perinatal mortality rate, malformations, and aneuploidy. Nicolaides and coworkers27 found that, when the end-diastolic velocity was absent, 67% to 80% of the fetuses were hypoxic and 45% were acidotic. Reversal of end-diastolic flow is associated with an even worse prognosis. The adverse consequences of absent or reverse end-diastolic flow are listed in TABLE 2.
In contrast to the NST and BPP, the effectiveness of fetal surveillance with umbilical arterial Doppler ultrasound in improving perinatal outcome in high-risk pregnancies has been confirmed by extensive randomized trials and their meta-analyses.28,29 These studies provide compelling evidence that, when used in well-defined high-risk pregnancies, especially those involving FGR or preeclampsia, umbilical arterial Doppler sonography reduces perinatal death, cesarean delivery for fetal distress, elective deliveries, and antenatal admissions.
Doppler sonography of the fetal cerebral and venous circulation also helps identify progressive fetal compromise in high-risk pregnancies, including those with FGR. When the fetal condition is deteriorating, cerebral arteries dilate to compensate, and pulsations appear in the umbilical vein with reversal of flow in the ductus venous.
These signs of grave fetal status indicate immediate delivery.
FIGURE 1 Duplex pulsed Doppler
Sonogram showing absent end-diastolic flow in the umbilical artery of a growth-restricted fetus.TABLE 2
Perinatal outcomes for absent and reversed end-diastolic velocity in the umbilical artery
| PERINATAL OUTCOME | MEAN | RANGE |
|---|---|---|
| Mortality | 45% | 17–100% |
| Gestational age | 31.6 weeks | 29–33 weeks |
| Birth weight | 1,056 g | 910–1,481 g |
| SGA | 68% | 53–100% |
| Cesarean section for fetal distress | 73% | 24–100% |
| Apgar score <7 at 5 minutes | 26% | 7–69% |
| Admission to NICU | 84% | 77–97% |
| Congenital anomalies | 10% | 0–24% |
| Aneuploidy | 6.4% | 0–18% |
| SGA = small for gestational age; NICU = neonatal intensive care unit | ||
| Reprinted with permission from: Maulik D, ed. Doppler Ultrasound in Obstetrics and Gynecology. | ||
| New York: Springer-Verlag; 1997:364, Table 21.2. | ||
Assessing the fetus for malformation and aneuploidy
Because of the association between FGR and fetal malformations and aneuploidy, the fetus should be assessed for these complications, especially when growth restriction is severe, develops early, and is not associated with hypertensive disorders, oligohydramnios, or abnormal umbilical arterial Doppler indices.
Most etiologies are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother.
The current standard practice is aneuploidy screening in early pregnancy and fetal anatomical scanning at midgestation. If significant risks of aneuploidy are present, a fetal karyotype is recommended, along with appropriate counseling.
3. Individualized managementAntepartum treatment
The question of bed rest. At present, there are no grounds for recommending strict bed rest in FGR cases. A Cochrane review30 showed no improvements with hospitalized bed rest compared with ambulation. Prolonged rest increases risk of thromboembolism and can be costly and inconvenient.
Nutritional remedies. Of the options that have been tested, which include high protein supplementation, nutrient supplementation such as beef liver extract, and balanced energy/protein supplementation, only the last (with protein content comprising less than 25% of total energy content) has led to a significant reduction in SGA births.31-33
Other approaches. We lack reliable evidence that other interventions such as oxygen administration, abdominal decompression, and pharmacological agents, including calcium channel blockers, beta mimetics, and magnesium, are beneficial or effective in improving or preventing FGR.
A meta-analysis34 of 13 trials involving more than 13,000 women showed that early aspirin treatment reduced the risk of FGR but failed to improve outcome. A more recent meta-analysis35 of 38 trials of aspirin in highrisk pregnancies found no reduction in the incidence of FGR or perinatal death, although a reduction in risk of preterm births was noted.
Etiologic management
Most etiologic conditions are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother. An example is maternal hypertensive disease, in which the indicated treatment has no beneficial effect on fetal growth.
Treatment of poor lifestyle habits may be helpful. If the mother smokes, vigorous smoking-cessation education and counseling is urged. Also address alcohol consumption and other substance abuse, and offer remedial measures.
The diagnosis of fetal viral and parasitic infections is important for prognostication and neonatal management. Although few perinatal infections are treatable in utero, maternal therapy may prevent certain infections from spreading to the fetus. Examples include toxoplasmosis and malaria.
If lethal malformations or lethal aneuploidy are identified prenatally, avoid fetal surveillance and unnecessary intervention, which may expose the mother to unnecessary and unjustifiable risks.
Fetal surveillance strategy
If fetal biometry indicates fetal weight below the 10th percentile, begin fetal surveillance. The current standard, umbilical arterial Doppler sonography, is the primary test in sonographically documented growth restriction. Also assess amniotic fluid volume—as part of the BPP or independently. AFI is the most commonly used BPP tool.
In the United States, the nonstress test traditionally is the primary monitoring modality, with the BPP as backup, although the BPP—which includes the NST—has been recommended as the primary test.
Normal Doppler findings. When the Doppler index remains within normal limits or does not progressively rise, weekly testing should suffice, with the NST or BPP as backup or in conjunction with Doppler.
If fetal and maternal conditions remain reassuring, allow the pregnancy to continue to maturity and assess the patient for delivery. Postdate pregnancy is not advised in the presence of sonographically confirmed growth compromise.
A high or increasing Doppler index warrants more intensive fetal surveillance consisting of weekly umbilical arterial Doppler and once- or twice-weekly NST and BPP until fetal maturity.
If these tests indicate fetal compromise, or absent end-diastolic velocity develops, the likelihood of poor perinatal outcome is increased and an urgent clinical response is indicated. Hospitalize the patient and individualize management, depending on gestational age and fetal status.
Optimal timing of delivery
The optimal timing of delivery in a preterm pregnancy with FGR is unclear. A recent multicenter randomized controlled trial, the Growth Restriction Intervention Trial (GRIT),36 compared 2 strategies: delivery within 48 hours with steroid administration or delivery delayed as long as fetal status permits. The population consisted of high-risk gravidas between 24 and 36 weeks’ gestation. More than 90% of the women had pregnancies complicated by FGR. No significant differences were noted between the 2 groups in stillbirth rates.
Fortunately, thanks to recent advances in perinatal care, a management strategy can be recommended:
At or near term (34 weeks or beyond), the absence of end-diastolic flow in the umbilical artery should prompt consideration of immediate delivery.
Other ominous findings that prompt such consideration include:
- Cessation of fetal growth on successive ultrasound examinations
- Progression of umbilical arterial absent enddiastolic flow to reversed end-diastolic flow
- Nonreassuring heart rate patterns including nonreactive NST, poor fetal heart rate baseline variability, and persistent variable or late decelerations
- Oligohydramnios
- BPP score of 4 or below
Less than 34 weeks. When absent enddiastolic flow develops in a preterm pregnancy with a significant risk of fetal lung immaturity, seek further assurance of fetal well-being via daily surveillance with umbilical arterial Doppler sonography, nonstress test, and biophysical profile. Administer betamethasone to enhance fetal lung maturity. Delivery is indicated regardless of maturity when a single test or combination indicates imminent fetal danger and the fetal risk from a hostile intrauterine environment is judged to be greater than that from pulmonary immaturity.
Intrapartum management
Labor and delivery of growth-restricted infants often is associated with a higher risk of asphyxia. The frequency of nonreassuring fetal heart rate patterns is increased. In addition, because of the greater prevalence of oligohydramnios, cord compression is frequent and associated with variable decelerations. There also is an increased risk of neonatal morbidities, as discussed above. Because of these risks, the growth-restricted fetus should be delivered in a facility that can offer appropriate inhouse support services such as neonatology and anesthesia.
If the patient is allowed to labor, use continuous electronic fetal heart rate monitoring with vigilance and perform the appropriate secondary tests, such as fetal scalp sampling or scalp stimulation. The ability of a growth-restricted fetus to tolerate labor is likely very limited.
Any confirmed signs of fetal compromise indicate cesarean as the optimal mode of delivery.
The authors report no financial relationships relevant to this article.
- Antepartum recognition of fetal growth restriction is essential for proper surveillance and management.
- Because growth-restricted fetuses are at risk for adverse outcomes in utero, fetal surveillance is vital for timely recognition and intervention.
- Once growth restriction is identified, management should be individualized to ensure optimal gestational development and safe delivery.
Not only is fetal growth restriction (FGR) associated with perinatal mortality and morbidity, but it may be linked to adverse consequences in adulthood.1 Its many causes involve diverse pathological processes; thus, it should not be considered a single disease. Many aspects remain unclear, a substantial number of affected infants are not identified before birth, and effective antepartum prevention and treatment remain elusive.
Fortunately, managing FGR has hopeful aspects. Meaningful recent advances elucidate its etiologic and pathophysiologic mechanisms and help clarify diagnosis and management.
This article offers an up-to-date, evidence-based approach and includes guidelines on 3 keys to success: antepartum recognition, fetal surveillance, and antepartum and intrapartum management. These guidelines are not intended as a strict protocol, since the clinical course of FGR is highly variable, but as the starting point for individualized care.
An amorphous entity: FGR terminology
Fetal growth restriction implies failure to realize genetically determined growth potential. Terms include fetal growth restriction, intrauterine growth restriction, and small for gestational age (SGA). The pejorative term growth retardation is obsolete.
Traditionally, FGR refers to prenatally identified fetal growth deficiency, whereas SGA refers to suboptimal birth weight for the gestational age. However, some small fetuses are merely constitutionally small, not growth-restricted. Conversely, not all growth-restricted fetuses are small in size or weight for gestational age. Yet defining these groups is difficult, as the tools are imprecise and controversial.
This article uses these definitions:
Fetal growth restriction identified in the antepartum period refers to a fetus with sonographically measured fetal dimensions, particularly abdominal circumference or estimated weight, below an age-specific threshold, typically the 10th percentile.
Fetal growth restriction identified at birth is birth weight below the 10th percentile for gestational age, or SGA. Unfortunately, this definition may fail to identify some fetuses that are truly growth-restricted. Alternative, more sensitive definitions, such as the Ponderal index or birth weight ratio, are used primarily in research.
Consequences of FGR
Perinatal outcomes. Perinatal morbidities include prematurity, oligohydramnios, nonreassuring fetal heart rate patterns with a higher incidence of cesarean delivery, birth asphyxia, low Apgar score, neonatal hypoglycemia, hypocalcemia, polycythemia, hyperbilirubinemia, hypothermia, apnea, seizure disorders, and infection.
Fetal and neonatal mortality is significantly increased. Perinatal mortality is influenced by many factors, including severity of growth restriction, timing of onset, gestational age, and cause of growth restriction. The lower the birth-weight percentile for gestational age, the higher the mortality rate.
Effects in infancy. Although many SGA infants “catch up” growth in infancy, the pattern varies. Height and weight catch-up growth of preterm FGR infants lags behind that of preterm infants that are appropriate for gestational age (AGA) at birth.2 Those with early-onset or severe growth deficit continue to lag behind in postnatal growth. Although recent studies indicate that rapid postnatal growth in SGA infants may lead to increased risk of chronic diseases, including type 2 diabetes, others have found tangible short-term benefits of less frequent morbidity and mortality in infancy.3
FGR has been linked to a spectrum of neurodevelopmental risks, including subtle behavioral abnormalities, immature sleep patterns, decreased visual fixation, decreased general activity, altered early mother-infant interaction, altered motor skills, and hyperactivity.4,5 Infants born SGA at 32 to 42 weeks were 4 to 6 times more likely to have cerebral palsy, yet those whose birth weight was above the 97th percentile also had increased risk. It remains uncertain whether deviant growth is the cause or a consequence of this disability.6
Long-term effects. Lifetime sequelae of early nutritional deprivation have been demonstrated in animals.7 Moreover, epidemiological evidence suggesting an association between SGA at birth or infancy and increased risk of abnormal blood lipid values, diabetes, hypertension, and ischemic heart disease in adult life led Barker and associates to propose the fetal origins hypothesis.1
1. Antepartum recognitionDetermine gestational age
A reliable estimate of gestational age is central to identification of fetal growth compromise in utero or at birth. In pregnancies at risk for fetal growth restriction, gestational age should be established early, preferably in the first trimester.
The method of determining gestational age influences the observed frequency of FGR and SGA births. Estimates are more accurate when based on early ultrasound biometry than on menstrual history. The latter, if well documented, regular, and ovulatory, may be reliable if it differs by no more than 1 week from the sonographic gestational age. Otherwise, early-pregnancy, ultrasoundbased age is more accurate.
Screening for FGR
Screening can be done clinically or by special investigation. The following methods are used, some of which remain experimental:
- assessment of historical clinical risks
- clinical evaluation of fetal and uterine size
- ultrasound fetal biometry
- umbilical arterial and uterine arterial
Doppler ultrasound
Clinical risk assessment. Evaluate all gravidas for risk factors (TABLE 1). If a woman is determined to be at heightened risk, take appropriate steps to diagnose FGR as early as possible.
Clinical evaluation of fetal and uterine size. Clinical assessment of fetal growth includes estimating fetal size by traditional obstetrical manual examination and by measuring the uterine fundal height.
- Abdominal palpation is inadequate to identify the fetus at increased risk of growth restriction, missing 74% of cases.8
- Serial measurements of the uterine fundal height, however, may be more reliable.9 This method consists of measuring fundal height from the symphysis pubis using a nonstretchable tape measure and assessing the results against a gestational age-specific nomogram. Limitations include significant interobserver differences and varying effects of maternal weight, parity, and fetal sex. A randomized trial of the method did not demonstrate any benefits, and a subsequent Cochrane review of the same study considered the evidence insufficient for any recommendations.10,11
Nevertheless, clinical assessment of uterine and fetal size is an essential, inexpensive component of prenatal care and a simple screening tool for identifying mothers who would benefit from further, more definitive sonographic investigation.
Routine ultrasound biometry. The potential benefits of accurately determined gestational age, and recognition of fetal malformations and multiple gestation, via earlyor mid-pregnancy ultrasound are well recognized and justify widespread use.
Routine scanning increases detection of SGA infants.12 A population-based cohort study13 involving over 16,000 singleton pregnancies found that fetuses smaller than expected at mid-second trimester ultrasound (discrepancy exceeding 14 days) were at increased risk for adverse outcomes, including perinatal mortality and SGA.
Unfortunately, these findings have not led to improved outcomes. A high false-positive rate remains a major concern. A prospective observational study14 found that routine ultrasound did not identify most cases of FGR, but resulted in a fivefold increase in iatrogenic premature delivery and significantly increased neonatal intensive care admissions.
- The Routine Antenatal Diagnostic Imaging with Ultrasound (RADIUS) trial15 randomized 15,000 low-risk gravidas to routine ultrasound imaging (at 15 to 22 weeks and again at 31 to 35 weeks) or to ultrasound only when indicated. The groups had similar rates of adverse perinatal outcome, distribution of birth weights, and preterm delivery. The trial’s weaknesses include selection criteria for low risk that excluded most pregnancies, inappropriate perinatal-outcome parameters, and suboptimal ultrasonographer expertise.
- A recent Cochrane review16 of 7 trials involving more than 25,000 women failed to demonstrate any improvements in perinatal mortality and morbidity with routine ultrasound, or any difference in antenatal, obstetric, and neonatal interventions.
Estimated fetal weight may differ from actual weight by as much as 20%.
We also lack evidence regarding longterm outcomes such as neurodevelopment.
Umbilical arterial Doppler. A meta-analysis17 of 4 randomized trials in unselected or low-risk pregnancies with a total population of 11,375 women found no effect of screening Doppler umbilical artery velocimetry on perinatal death, stillbirth, antenatal hospitalization, obstetric outcome, or perinatal morbidity.
A subsequent meta-analysis18 of 5 trials of routine Doppler ultrasound in unselected and low-risk pregnancies with a total population of more than 14,000 women also found no benefit for mother or infant.
We lack evidence on long-term outcomes.
Uterine arterial Doppler screening. Increased pulsatility of the uterine arterial Doppler waveform, persistence of the notch, and a significant difference between right and left uterine arteries have been associated with FGR, pregnancy-induced hypertension, and adverse perinatal outcome. A review19 of 15 studies of routine uterine Doppler in unselected populations showed considerable heterogeneity, but indicated that increased impedance in the uterine arteries identifies about 20% of those who develop FGR, with a positive likelihood ratio of 3.5.
We lack exclusive randomized trials of routine uterine Doppler sonography in unselected and low-risk pregnancies. However, 2 studies done in conjunction with umbilical arterial Doppler found no impact on outcome, and a recent Cochrane review18 found insufficient evidence to support routine uterine Doppler for FGR screening.
TABLE 1
Risk factors
| MATERNAL FACTORS |
| Medical disease |
| Preeclampsia/hypertension |
| Renal disease |
| Antiphospholipid antibody syndrome |
| Inherited thrombophilia |
| Diabetes with vasculopathy |
| Cyanotic heart disease |
| Asthma |
| Hemoglobinopathy |
| Phenylketonuria |
| Life circumstance |
| Severe malnutrition |
| Smoking |
| Substance abuse (eg, alcohol, heroine, cocaine) |
| PLACENTAL FACTORS |
| Confined placental mosaic |
| Placenta previa |
| Abruptio placentae |
| Infarction |
| Circumvallate placenta |
| Placenta accreta |
| Hemangioma |
| FETAL FACTORS |
| Multiple gestation |
| Prematurity |
| Unexplained elevated alpha-fetoprotein |
| Infection (eg, rubella, cytomegalovirus, herpes, malaria, toxoplasmosis) |
| Malformations (eg, gastroschisis, omphalocele, diaphragmatic hernia, congenital heart defect) |
| Genetic disorders (eg, trisomy 13, 18, and 21; triploidy; some cases of Turner’s syndrome) |
Identifying FGR in utero
Antepartum diagnosis is based on sonographic measurement of various fetal dimensions.
Abdominal circumference and estimated fetal weight. A review20 of 60 studies found that abdominal circumference (AC) and sonographically estimated fetal weight (EFW) were the best predictors of birth weight below the 10th percentile in high-risk pregnancies. AC below the 10th percentile had sensitivities ranging from 72.9% to 94.5%, false-positive rates of 16.2% to 49.4%, and a common odds ratio of 18.4. An EFW below the 10th percentile had a common odds ratio of 39.1, which was the highest among all the biometric measurements. Its sensitivity ranged from 33.3% to 89.2%, and false-positive rates ranged from 9.1% to 46.3%.
The proportionality of fetal dimensions, such as the head/abdominal ratio, was not a good predictor, although it is routinely generated in ultrasound biometry.
Ultrasound estimation of fetal weight is based on a combination of 2 or 3 biometric measurements of the fetus, including the biparietal diameter (BPD) or the head circumference (HC), AC, and femur length (FL). Several formulae yield varying estimations of weight. Thus, a fetus identified as growth-restricted by 1 formula may not be so diagnosed by another. For this reason, it is prudent to be consistent in their use. EFW generated from measurements of the head (BPD or HC), AC, and FL is most reliable.21
The EFW also is expressed as the percentile for the gestational age.
Limitations of ultrasound estimation.
The optimal process of translating dimensional measurements into fetal body mass for both health and disease remains unknown. This leads to inaccurate assumptions and erroneous weight projections.
Inaccuracies also result from variations in measurement. In addition, the estimation is less accurate in the lower and upper ranges of fetal weight distribution and in the presence of oligohydramnios. As a result, estimated weight may differ from actual weight by as much as 20%, with greater margins of error at the lower and upper extremes.
Guidelines for screening and diagnosis
Assess all pregnancies for risk factors and determine the gestational age in early pregnancy, especially in women at higher risk of FGR. If the patient has substantial clinical risks or there is suspicion of growth restriction, fetal ultrasound biometry is recommended. Diagnosis of FGR is based on fetal sonographic measurements, especially AC; estimated fetal weight derived from BPD or HC, AC and FL; and longitudinal progression of fetal growth.
Current evidence suggests the use of a 10th percentile diagnostic threshold for these measurements. Follow the biometric parameters longitudinally, repeating the measurement every 2 to 4 weeks. More frequent examination is unreliable.
2. Fetal surveillanceAntepartum monitoring
Fetuses identified as growth-restricted are at risk for adverse outcomes in utero. Thus, antepartum monitoring is indicated for timely recognition and intervention.
Tests commonly used include the nonstress test (NST), biophysical profile (BPP), sonographic estimation of amniotic fluid volume, and Doppler velocimetry of the umbilical artery or other vessels of interest.
With progressive fetal compromise, compensatory mechanisms may be insufficient to maintain fetal homeostasis. The sequence of loss of compensatory mechanisms is reflected in surveillance tests. Thus, changes in umbil-ical arterial Doppler indices usually precede nonreassuring NST or BPP.
Nonstress test: The standard of care. The NST is probably the most common fetal surveillance test in high-risk pregnancies, including those complicated by FGR. However, it is personnel- and time-intensive, and interpretation often is subjective.
A nonreactive NST is associated with adverse perinatal outcome, although it more commonly is related to the fetal sleep cycle.
In predicting a potentially preventable fetal death, the NST has a false-negative rate of 2 to 3 per 1,000, a negative predictive value of 99.8%, and a false-positive rate of 80%.
Evidence of its efficacy is lacking. Four randomized trials in the early 1980s, which involved more than 1,500 women, found no improvement in outcomes. (All 4 trials lacked sufficient power.)
- When to test. Despite these limitations, the NST has become an integral part of obstetri-cal practice and remains a standard of care for antepartum fetal surveillance in high-risk pregnancies. It should be used as a surveillance tool for the growth-restricted fetus. The test initially is performed weekly.
Depending on severity of growth restriction, testing may increase to twice weekly or even daily.
Amniotic fluid volume monitoring is warranted. Oligohydramnios—highly prevalent in FGR—is associated with adverse perina-tal outcomes.
Although the sonographically determined amniotic fluid index (AFI) is the most frequently used modality to evaluate amniotic fluid volume, its accuracy has been questioned. Nevertheless, a meta-analysis22 of 18 studies involving more than 10,000 patients showed that antepartum AFI equal to or below 5 cm was associated with an increased risk of cesarean section for fetal distress and an Apgar score below 7 at 5 minutes.
Even marginal AFI values (5–10 cm) have been associated with doubling of the risk of adverse perinatal outcome.23
- When to test. We lack randomized trials of amniotic fluid volume assessment in high-risk pregnancies, but observational studies suggest that weekly determination of AFI is helpful in sonographically identified growth-restricted fetuses.
A low or marginal AFI should be followed by more frequent examinations. Even a marginal AFI may be followed by twice-weekly surveillance.
Biophysical profile effectively predicts adverse perinatal outcome, with a false-negative rate of 0.8 per 1,000, negative predictive value of 99.9%, and a false-positive rate of 40% to 50%.24 A low BPP is associated with fetal hypoxia and acidosis, and a declining BPP reflects progressive worsening of the fetal condition.
In FGR, a significant association exists between abnormal Doppler indices and fetal acid-base compromise.
Although equal weight was originally accorded all the parameters of the profile, experience suggests that the independent risk of oligohydramnios warrants immediate reassessment of the management plan.
Like the nonstress test, the BPP has a low false-negative rate and a high false-positive rate. Four randomized trials of the test have been conducted, involving about 2,800 patients, with no confirmation of its effectiveness. A Cochrane systematic review25 concluded that this cumulative sample size is insufficient to draw definitive conclusions.
The test remains a standard, however, and is recommended to confirm fetal wellbeing or as part of a comprehensive test panel in significantly high-risk pregnancies.
- When to test. Initially perform the test once a week, increasing to twice weekly when the severity of growth restriction warrants.
Daily testing may be indicated in complications such as severe growth restriction and pregestational diabetes. In early preterm pregnancies (less than 32 weeks) with nonreassuring NST or umbilical arterial Doppler, BPP can help guide optimal management.
Umbilical arterial Doppler ultrasound is a powerful predictor of adverse perinatal outcomes in high-risk pregnancies.
Doppler indices include the pulsatility index, systolic/diastolic ratio, diastolic average ratio, and the resistance index. Of these, the last demonstrated the greatest ability to predict abnormal perinatal outcomes.
A prospective, blinded study26 in highrisk pregnancies demonstrated significant diagnostic efficacy with a sensitivity of 79%, specificity of 93%, positive predictive value of 83%, negative predictive value of 91%, and kappa index of 73%. The last value is consistent with a good to excellent diagnostic test.
A significant association also exists between abnormal Doppler indices and fetal acid-base compromise in FGR. The absence of enddiastolic flow (FIGURE) is associated with markedly adverse perinatal outcome, particularly a high perinatal mortality rate, malformations, and aneuploidy. Nicolaides and coworkers27 found that, when the end-diastolic velocity was absent, 67% to 80% of the fetuses were hypoxic and 45% were acidotic. Reversal of end-diastolic flow is associated with an even worse prognosis. The adverse consequences of absent or reverse end-diastolic flow are listed in TABLE 2.
In contrast to the NST and BPP, the effectiveness of fetal surveillance with umbilical arterial Doppler ultrasound in improving perinatal outcome in high-risk pregnancies has been confirmed by extensive randomized trials and their meta-analyses.28,29 These studies provide compelling evidence that, when used in well-defined high-risk pregnancies, especially those involving FGR or preeclampsia, umbilical arterial Doppler sonography reduces perinatal death, cesarean delivery for fetal distress, elective deliveries, and antenatal admissions.
Doppler sonography of the fetal cerebral and venous circulation also helps identify progressive fetal compromise in high-risk pregnancies, including those with FGR. When the fetal condition is deteriorating, cerebral arteries dilate to compensate, and pulsations appear in the umbilical vein with reversal of flow in the ductus venous.
These signs of grave fetal status indicate immediate delivery.
FIGURE 1 Duplex pulsed Doppler
Sonogram showing absent end-diastolic flow in the umbilical artery of a growth-restricted fetus.TABLE 2
Perinatal outcomes for absent and reversed end-diastolic velocity in the umbilical artery
| PERINATAL OUTCOME | MEAN | RANGE |
|---|---|---|
| Mortality | 45% | 17–100% |
| Gestational age | 31.6 weeks | 29–33 weeks |
| Birth weight | 1,056 g | 910–1,481 g |
| SGA | 68% | 53–100% |
| Cesarean section for fetal distress | 73% | 24–100% |
| Apgar score <7 at 5 minutes | 26% | 7–69% |
| Admission to NICU | 84% | 77–97% |
| Congenital anomalies | 10% | 0–24% |
| Aneuploidy | 6.4% | 0–18% |
| SGA = small for gestational age; NICU = neonatal intensive care unit | ||
| Reprinted with permission from: Maulik D, ed. Doppler Ultrasound in Obstetrics and Gynecology. | ||
| New York: Springer-Verlag; 1997:364, Table 21.2. | ||
Assessing the fetus for malformation and aneuploidy
Because of the association between FGR and fetal malformations and aneuploidy, the fetus should be assessed for these complications, especially when growth restriction is severe, develops early, and is not associated with hypertensive disorders, oligohydramnios, or abnormal umbilical arterial Doppler indices.
Most etiologies are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother.
The current standard practice is aneuploidy screening in early pregnancy and fetal anatomical scanning at midgestation. If significant risks of aneuploidy are present, a fetal karyotype is recommended, along with appropriate counseling.
3. Individualized managementAntepartum treatment
The question of bed rest. At present, there are no grounds for recommending strict bed rest in FGR cases. A Cochrane review30 showed no improvements with hospitalized bed rest compared with ambulation. Prolonged rest increases risk of thromboembolism and can be costly and inconvenient.
Nutritional remedies. Of the options that have been tested, which include high protein supplementation, nutrient supplementation such as beef liver extract, and balanced energy/protein supplementation, only the last (with protein content comprising less than 25% of total energy content) has led to a significant reduction in SGA births.31-33
Other approaches. We lack reliable evidence that other interventions such as oxygen administration, abdominal decompression, and pharmacological agents, including calcium channel blockers, beta mimetics, and magnesium, are beneficial or effective in improving or preventing FGR.
A meta-analysis34 of 13 trials involving more than 13,000 women showed that early aspirin treatment reduced the risk of FGR but failed to improve outcome. A more recent meta-analysis35 of 38 trials of aspirin in highrisk pregnancies found no reduction in the incidence of FGR or perinatal death, although a reduction in risk of preterm births was noted.
Etiologic management
Most etiologic conditions are either not amenable to therapy or fetal growth is not improved by treatments that benefit the mother. An example is maternal hypertensive disease, in which the indicated treatment has no beneficial effect on fetal growth.
Treatment of poor lifestyle habits may be helpful. If the mother smokes, vigorous smoking-cessation education and counseling is urged. Also address alcohol consumption and other substance abuse, and offer remedial measures.
The diagnosis of fetal viral and parasitic infections is important for prognostication and neonatal management. Although few perinatal infections are treatable in utero, maternal therapy may prevent certain infections from spreading to the fetus. Examples include toxoplasmosis and malaria.
If lethal malformations or lethal aneuploidy are identified prenatally, avoid fetal surveillance and unnecessary intervention, which may expose the mother to unnecessary and unjustifiable risks.
Fetal surveillance strategy
If fetal biometry indicates fetal weight below the 10th percentile, begin fetal surveillance. The current standard, umbilical arterial Doppler sonography, is the primary test in sonographically documented growth restriction. Also assess amniotic fluid volume—as part of the BPP or independently. AFI is the most commonly used BPP tool.
In the United States, the nonstress test traditionally is the primary monitoring modality, with the BPP as backup, although the BPP—which includes the NST—has been recommended as the primary test.
Normal Doppler findings. When the Doppler index remains within normal limits or does not progressively rise, weekly testing should suffice, with the NST or BPP as backup or in conjunction with Doppler.
If fetal and maternal conditions remain reassuring, allow the pregnancy to continue to maturity and assess the patient for delivery. Postdate pregnancy is not advised in the presence of sonographically confirmed growth compromise.
A high or increasing Doppler index warrants more intensive fetal surveillance consisting of weekly umbilical arterial Doppler and once- or twice-weekly NST and BPP until fetal maturity.
If these tests indicate fetal compromise, or absent end-diastolic velocity develops, the likelihood of poor perinatal outcome is increased and an urgent clinical response is indicated. Hospitalize the patient and individualize management, depending on gestational age and fetal status.
Optimal timing of delivery
The optimal timing of delivery in a preterm pregnancy with FGR is unclear. A recent multicenter randomized controlled trial, the Growth Restriction Intervention Trial (GRIT),36 compared 2 strategies: delivery within 48 hours with steroid administration or delivery delayed as long as fetal status permits. The population consisted of high-risk gravidas between 24 and 36 weeks’ gestation. More than 90% of the women had pregnancies complicated by FGR. No significant differences were noted between the 2 groups in stillbirth rates.
Fortunately, thanks to recent advances in perinatal care, a management strategy can be recommended:
At or near term (34 weeks or beyond), the absence of end-diastolic flow in the umbilical artery should prompt consideration of immediate delivery.
Other ominous findings that prompt such consideration include:
- Cessation of fetal growth on successive ultrasound examinations
- Progression of umbilical arterial absent enddiastolic flow to reversed end-diastolic flow
- Nonreassuring heart rate patterns including nonreactive NST, poor fetal heart rate baseline variability, and persistent variable or late decelerations
- Oligohydramnios
- BPP score of 4 or below
Less than 34 weeks. When absent enddiastolic flow develops in a preterm pregnancy with a significant risk of fetal lung immaturity, seek further assurance of fetal well-being via daily surveillance with umbilical arterial Doppler sonography, nonstress test, and biophysical profile. Administer betamethasone to enhance fetal lung maturity. Delivery is indicated regardless of maturity when a single test or combination indicates imminent fetal danger and the fetal risk from a hostile intrauterine environment is judged to be greater than that from pulmonary immaturity.
Intrapartum management
Labor and delivery of growth-restricted infants often is associated with a higher risk of asphyxia. The frequency of nonreassuring fetal heart rate patterns is increased. In addition, because of the greater prevalence of oligohydramnios, cord compression is frequent and associated with variable decelerations. There also is an increased risk of neonatal morbidities, as discussed above. Because of these risks, the growth-restricted fetus should be delivered in a facility that can offer appropriate inhouse support services such as neonatology and anesthesia.
If the patient is allowed to labor, use continuous electronic fetal heart rate monitoring with vigilance and perform the appropriate secondary tests, such as fetal scalp sampling or scalp stimulation. The ability of a growth-restricted fetus to tolerate labor is likely very limited.
Any confirmed signs of fetal compromise indicate cesarean as the optimal mode of delivery.
The authors report no financial relationships relevant to this article.
1. Barker DJP, ed. Fetal and Infant Origins of Adult Disease. London, UK: BMJ Publishing; 1992.
2. Strauss RS, Dietz WH. Effects of intrauterine growth retardation in premature infants on early childhood growth. J Pediatr. 1997;13:95-102.
3. Victora CG, Barros FC, Horta BL, Martorell R. Short-term benefits of catch-up growth for small-for-gestational-age infants. Int J Epidemiol. 2001;30:1325-1330.
4. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on the fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
5. Low JA, Handley-Derry MH, Burke SO, et al. Association of intrauterine fetal growth retardation and learning deficits at age 9 to 11 years. Am J Obstet Gynecol. 1992;167:1499-1505.
6. Jarvis S, Glinianaia SV, Torrioli MG, et al. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003;362:1106-1111.
7. Lucas A. Programming by early nutrition: an experimental approach. J Nutr. 1998;128(suppl 2):401-406S.
8. Hepburn M, Rosenberg K. An audit of the detection and management of small-for-gestational age babies. Br J Obstet Gynaecol. 1986;93:212-216.
9. Belizan JM, Villar J, Nardin JC, Malamud J, Sainz de Vicuna L. Diagnosis of intrauterine growth retardation by a simple clinical method: measurement of uterine height. Am J Obstet Gynecol. 1978;131:643-646.
10. Lindhard A, Nielsen PV, Mouritsen LA, Zachariassen A, Sorensen HU, Roseno H. The implications of introducing the symphyseal-fundal height measurement. A prospective randomized controlled trial. Br J Obstet Gynaecol. 1990;97:675-680.
11. Neilson JP. Symphysis-fundal height measurement in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD000944.-
12. Hughey MJ. Routine ultrasound for detection and management of the small for-gestational-age fetus. Obstet Gynecol. 1984;64:101-107.
13. Nakling J, Backe B. Adverse obstetric outcome in fetuses that are smaller than expected at second trimester routine ultrasound examination. Acta Obstet Gynecol Scand. 2002;81:846-851.
14. Jahn A, Razum O, Berle P. Routine screening for intrauterine growth retardation in Germany: low sensitivity and questionable benefit for diagnosed cases. Acta Obstet Gynecol Scand. 2002;81:846-851.
15. Ewigman BG, Crane JP, Frigoletto FD, LeFevre ML, Bain RP, McNellis D. Effect of prenatal ultrasound screening on perinatal outcome. RADIUS Study Group. N Engl J Med. 1993; 16;329:821-827.
16. Bricker L, Neilson JP. Routine ultrasound in late pregnancy (after 24 weeks gestation). Cochrane Database Syst Rev. 2000;(2):CD001451.-
17. Goffinet F, Paris Llado J, Nisand I, Breart G. Umbilical artery Doppler velocimetry in unselected and low risk pregnancies: a review of randomized controlled trials. Br J Obstet Gynaecol. 1997;104:425-430.
18. Bricker L, Neilson JP. Routine Doppler ultrasound in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD001450.-
19. Papageorghiou AT, Yu CK, Cicero S, Bower S, Nicolaides KH. Second-trimester uterine artery Doppler screening in unselected populations: a review. J Matern Fetal Neonatal Med. 2002;12:78-88.
20. Chang TC, Robson SC, Boys RJ, Spencer JA. Prediction of the small for gestational age infant: which ultrasonic measurement is best? Obstet Gynecol. 1992;80:1030-1038.
21. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151:333-337.
22. Chauhan SP, Sanderson M, Hendrix NW, Magann EF, Devoe LD. Perinatal outcome and amniotic fluid index in the antepartum and intrapartum periods: a meta-analysis. Am J Obstet Gynecol. 1999;18:1473-1478.
23. Banks EH, Miller DA. Perinatal risks associated with borderline amniotic fluid index. Am J Obstet Gynecol. 1999;180:1461-1463.
24. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
25. Alfirevic Z, Neilson JP. Biophysical profile for fetal assessment in high risk pregnancies. Cochrane Database Syst Rev. 2003;(1).:The Cochrane Collaboration.
26. Maulik D, Yarlagadda P, Youngblood JP, et al. The diagnostic efficacy of the UA systolic/diastolic ratio as a screening tool a prospective blinded study. Am J Obstet Gynecol. 1990;162:1518-1523.
27. Nicolaides KH, Bilardo CM, Soothill PW, et al. Absence of end diastolic frequencies in umbilical artery: a sign of fetal hypoxia and acidosis. B Med J. 1988;297:1026-1027.
28. Alfirevic Z, Neilson JP. Doppler ultrasonography in high-risk pregnancies: systematic review with meta-analysis. Am J Obstet Gynecol. 1995;172:1379-1387.
29. Westergaard HB, Langhoff-Roos J, Lingman G, Marsal K, Kreiner S. A critical appraisal of the use of umbilical artery Doppler ultrasound in high-risk pregnancies: use of meta-analyses in evidence-based obstetrics. Ultrasound Obstet Gynecol. 2001;17:464-465.
30. Gulmezoglu AM, Hofmeyr GJ. Bed rest in hospital for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
31. Kramer MS. High protein supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
32. Kramer MS. Balanced protein/energy supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
33. Gulmezoglu AM, Hofmeyr GJ. Maternal nutrient supplementation for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
34. Leitich H, Egarter C, Husslein P, Kaider A, Schemper M. A meta-analysis of low dose aspirin for the prevention of intrauterine growth retardation. Br J Obstet Gynaecol. 1997;104:450-459.
35. Kozer E, Costei AM, Boskovic R, Nulman I, Nikfar S, Koren G. Effects of aspirin consumption during pregnancy on pregnancy outcomes: meta-analysis. Birth Defects Res Part B Dev Reprod Toxicol. 2003;68:70-84.
36. The GRIT Study Group. A randomized trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. BJOG. 2003;110:27-32.
1. Barker DJP, ed. Fetal and Infant Origins of Adult Disease. London, UK: BMJ Publishing; 1992.
2. Strauss RS, Dietz WH. Effects of intrauterine growth retardation in premature infants on early childhood growth. J Pediatr. 1997;13:95-102.
3. Victora CG, Barros FC, Horta BL, Martorell R. Short-term benefits of catch-up growth for small-for-gestational-age infants. Int J Epidemiol. 2001;30:1325-1330.
4. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on the fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
5. Low JA, Handley-Derry MH, Burke SO, et al. Association of intrauterine fetal growth retardation and learning deficits at age 9 to 11 years. Am J Obstet Gynecol. 1992;167:1499-1505.
6. Jarvis S, Glinianaia SV, Torrioli MG, et al. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003;362:1106-1111.
7. Lucas A. Programming by early nutrition: an experimental approach. J Nutr. 1998;128(suppl 2):401-406S.
8. Hepburn M, Rosenberg K. An audit of the detection and management of small-for-gestational age babies. Br J Obstet Gynaecol. 1986;93:212-216.
9. Belizan JM, Villar J, Nardin JC, Malamud J, Sainz de Vicuna L. Diagnosis of intrauterine growth retardation by a simple clinical method: measurement of uterine height. Am J Obstet Gynecol. 1978;131:643-646.
10. Lindhard A, Nielsen PV, Mouritsen LA, Zachariassen A, Sorensen HU, Roseno H. The implications of introducing the symphyseal-fundal height measurement. A prospective randomized controlled trial. Br J Obstet Gynaecol. 1990;97:675-680.
11. Neilson JP. Symphysis-fundal height measurement in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD000944.-
12. Hughey MJ. Routine ultrasound for detection and management of the small for-gestational-age fetus. Obstet Gynecol. 1984;64:101-107.
13. Nakling J, Backe B. Adverse obstetric outcome in fetuses that are smaller than expected at second trimester routine ultrasound examination. Acta Obstet Gynecol Scand. 2002;81:846-851.
14. Jahn A, Razum O, Berle P. Routine screening for intrauterine growth retardation in Germany: low sensitivity and questionable benefit for diagnosed cases. Acta Obstet Gynecol Scand. 2002;81:846-851.
15. Ewigman BG, Crane JP, Frigoletto FD, LeFevre ML, Bain RP, McNellis D. Effect of prenatal ultrasound screening on perinatal outcome. RADIUS Study Group. N Engl J Med. 1993; 16;329:821-827.
16. Bricker L, Neilson JP. Routine ultrasound in late pregnancy (after 24 weeks gestation). Cochrane Database Syst Rev. 2000;(2):CD001451.-
17. Goffinet F, Paris Llado J, Nisand I, Breart G. Umbilical artery Doppler velocimetry in unselected and low risk pregnancies: a review of randomized controlled trials. Br J Obstet Gynaecol. 1997;104:425-430.
18. Bricker L, Neilson JP. Routine Doppler ultrasound in pregnancy. Cochrane Database Syst Rev. 2000;(2):CD001450.-
19. Papageorghiou AT, Yu CK, Cicero S, Bower S, Nicolaides KH. Second-trimester uterine artery Doppler screening in unselected populations: a review. J Matern Fetal Neonatal Med. 2002;12:78-88.
20. Chang TC, Robson SC, Boys RJ, Spencer JA. Prediction of the small for gestational age infant: which ultrasonic measurement is best? Obstet Gynecol. 1992;80:1030-1038.
21. Hadlock FP, Harrist RB, Sharman RS, Deter RL, Park SK. Estimation of fetal weight with the use of head, body, and femur measurements—a prospective study. Am J Obstet Gynecol. 1985;151:333-337.
22. Chauhan SP, Sanderson M, Hendrix NW, Magann EF, Devoe LD. Perinatal outcome and amniotic fluid index in the antepartum and intrapartum periods: a meta-analysis. Am J Obstet Gynecol. 1999;18:1473-1478.
23. Banks EH, Miller DA. Perinatal risks associated with borderline amniotic fluid index. Am J Obstet Gynecol. 1999;180:1461-1463.
24. Manning FA, Morrison I, Harman CR, Lange IR, Menticoglou S. Fetal assessment based on fetal biophysical profile scoring: experience in 19,221 referred high-risk pregnancies. II. An analysis of false-negative fetal deaths. Am J Obstet Gynecol. 1987;157:880-884.
25. Alfirevic Z, Neilson JP. Biophysical profile for fetal assessment in high risk pregnancies. Cochrane Database Syst Rev. 2003;(1).:The Cochrane Collaboration.
26. Maulik D, Yarlagadda P, Youngblood JP, et al. The diagnostic efficacy of the UA systolic/diastolic ratio as a screening tool a prospective blinded study. Am J Obstet Gynecol. 1990;162:1518-1523.
27. Nicolaides KH, Bilardo CM, Soothill PW, et al. Absence of end diastolic frequencies in umbilical artery: a sign of fetal hypoxia and acidosis. B Med J. 1988;297:1026-1027.
28. Alfirevic Z, Neilson JP. Doppler ultrasonography in high-risk pregnancies: systematic review with meta-analysis. Am J Obstet Gynecol. 1995;172:1379-1387.
29. Westergaard HB, Langhoff-Roos J, Lingman G, Marsal K, Kreiner S. A critical appraisal of the use of umbilical artery Doppler ultrasound in high-risk pregnancies: use of meta-analyses in evidence-based obstetrics. Ultrasound Obstet Gynecol. 2001;17:464-465.
30. Gulmezoglu AM, Hofmeyr GJ. Bed rest in hospital for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
31. Kramer MS. High protein supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
32. Kramer MS. Balanced protein/energy supplementation in pregnancy. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
33. Gulmezoglu AM, Hofmeyr GJ. Maternal nutrient supplementation for suspected impaired fetal growth. Cochrane Database Syst Rev. 2002;(4).-Oxford Update Software.
34. Leitich H, Egarter C, Husslein P, Kaider A, Schemper M. A meta-analysis of low dose aspirin for the prevention of intrauterine growth retardation. Br J Obstet Gynaecol. 1997;104:450-459.
35. Kozer E, Costei AM, Boskovic R, Nulman I, Nikfar S, Koren G. Effects of aspirin consumption during pregnancy on pregnancy outcomes: meta-analysis. Birth Defects Res Part B Dev Reprod Toxicol. 2003;68:70-84.
36. The GRIT Study Group. A randomized trial of timed delivery for the compromised preterm fetus: short term outcomes and Bayesian interpretation. BJOG. 2003;110:27-32.