User login
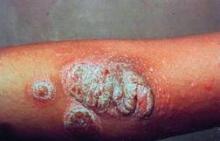
The new National Psoriasis Foundation consensus guidelines on the management of plaque psoriasis take a practical approach to helping physicians manage the disease beyond topical treatment.
The guidelines feature a table summarizing the latest consensus on eight systemic medications, and they hone in on specific forms of psoriasis in a way that is more quickly understandable than in the past, according to lead author Dr. Sylvia Hsu, chief of Ben Taub Dermatology Service and a dermatology professor at Baylor College of Medicine in Houston.
"The aim is to simplify and clarify when to use these drugs for patients whose psoriasis is beyond topical treatment," she said. "A lot of the drugs can be used first line, so it’s not like a treatment algorithm."
Dr. Hsu acknowledged that the guidelines are a bit bold because they directly state, for example, that acitretin doesn’t work well for plaque psoriasis, and that evidence is scant to support the use of alefacept to achieve full clearance. The latter position was devised well before Astellas announced in December 2011 that it was pulling alefacept from the market, she said in an interview.
Physicians may also be surprised that the guidelines, published in Archives of Dermatology (2012;148:95-102), recommend cyclosporine as a short-term solution for up to 12 weeks to control a psoriasis flare.
"We know that you can use cyclosporine for up to a year because it’s FDA-approved for up to 12 months, but the typical use is as a quick-fix to transition to other agents that are slower acting," she said. "It’s a practical table."
Established drugs such as etanercept (Enbrel) and adalimumab (Humira) are recommended as first-line therapies for plaque psoriasis, but so is the newer monoclonal antibody ustekinumab (Stelara), so avoid the misconception that dermatologists need to prescribe systemic medications in a particular order based upon time on the market, Dr. Hsu noted.
The National Psoriasis Foundation (NPF) guidelines, however, do not provide a formula for how to treat patients, said coauthor Dr. Steven R. Feldman, professor of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C.
"They don’t say if you have this patient, this must be done first and then if that doesn’t work, do this second, and that third," he said. "There is no guideline of that type for psoriasis because no one treatment is better than all the other treatments in every way and patients weigh the various variables differently."
The new guidelines were adapted from the Canadian Guidelines for the Management of Plaque Psoriasis to reflect United States practice patterns, and are very similar to those published on the subject by the American Academy of Dermatology (AAD), Dr. Feldman said.
"I think these guidelines are clearly consensus guidelines, getting the experts together and deciding what they feel is best, whereas the AAD made a special effort to create evidence-based guidelines based on the available data," he said. "In the end, I think the recommendations are basically the same thing. Neither one tells physicians exactly what to do in a given situation; it’s more about describing the reasonable options that a patient and physician could choose in any situation."
NPF and AAD guidelines coauthor Dr. Mark Lebwohl, professor and chair of the department dermatology at Mount Sinai School of Medicine in New York, said the AAD guidelines are superb, but that "having this second set of guidelines allows us to answer even more questions."
He noted that there is often information in one guideline that is missing from another because new psoriasis developments happen so quickly. For example, a section recommending that physicians consider withholding tumor necrosis factor antagonists in psoriasis patients undergoing elective surgery was borrowed from the Canadian guidelines, but does not appear in the AAD guidelines, he said. The section discusses the controversy over the potential for increased risk of postsurgical infection in patients on biologics, and provides guidance on when specific agents should be withheld.
Conversely, the NPF guidelines discuss the pros and cons of the excimer laser, but this discussion is missing from the Canadian guidelines because the laser is not approved there.
"In a year, the guidelines will all be out of date again; they are a work in progress," said Dr. Lebwohl, who added that the NPF is trying to find a mechanism to update the guidelines in real time as new developments arise.
What the NPF/Canadian guidelines offer physicians is a rigorous evaluation of the literature with a patient-focused approach to psoriasis treatment, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., who helped coauthor both sets of guidelines.
"We all appreciate that the art of medicine incorporates habit, consensus, experience, and hopefully sound data," he said. "Consequently, a second and perhaps equally important offering of the guidelines to physicians is an independent affirmation or an independent negation of one’s art in treating psoriasis."
Where the NPF guidelines may prove particularly helpful is in supporting that therapeutic choice with insurers. If a physician thought that a particular biologic agent was the best treatment option for a patient, that physician could point to the guidelines as a way to let insurers know that what is being recommended is within the standard of care, said Dr. Feldman.
The new guidelines could also push physicians to be more aggressive in using some of the available systemic agents. He pointed out that one of the "major enthusiasms" for creating the guidelines was a 2009 National Psoriasis Foundation survey finding that 57% of patients with severe psoriasis were receiving treatment insufficient to achieve adequate control (J. Am. Acad. Dermatol. 2007;57:957-62).
"There needed to be guidelines that would encourage physicians to provide patients treatment that met the severity of their illness," said Dr. Feldman, who suggested that safety concerns, dated training, and a perception that psoriasis isn’t such a bad disease, all played into the lack of aggressive treatment.
The NPF guidelines clearly detail the safety risks associated with each agent, and have a specific section on special populations, including pregnant women, patients with HIV, and those with hepatitis B or C.
For the purpose of the new guidelines, patients are considered to have moderate to severe psoriasis if they cannot achieve or would not be expected to achieve adequate control using topical agents, with adequacy defined by the patient’s own perception of the disease and its burdens. Cutoffs based on clinical metrics, such as the Psoriasis Area and Severity Index, percentage of body surface area affected, and the Dermatological Life Quality Index are necessary in clinical trials, but "have little value in daily practice," the authors noted.
Strict numeric cutoffs can also cause problems with insurers and reimbursement.
"This [document] provides the insurers and other regulators the opportunity to set more meaningful standards for their policies," Dr. Feldman said. "When they see guidelines published in the Archives and supported by the National Psoriasis Foundation, it lends a lot of weight to doing things the way doctors do things in the patient’s best interest."
The physicians interviewed for this story all disclosed consultancies and research arrangements with multiple companies that manufacture psoriasis treatments.
The new National Psoriasis Foundation consensus guidelines on the management of plaque psoriasis take a practical approach to helping physicians manage the disease beyond topical treatment.
The guidelines feature a table summarizing the latest consensus on eight systemic medications, and they hone in on specific forms of psoriasis in a way that is more quickly understandable than in the past, according to lead author Dr. Sylvia Hsu, chief of Ben Taub Dermatology Service and a dermatology professor at Baylor College of Medicine in Houston.
"The aim is to simplify and clarify when to use these drugs for patients whose psoriasis is beyond topical treatment," she said. "A lot of the drugs can be used first line, so it’s not like a treatment algorithm."
Dr. Hsu acknowledged that the guidelines are a bit bold because they directly state, for example, that acitretin doesn’t work well for plaque psoriasis, and that evidence is scant to support the use of alefacept to achieve full clearance. The latter position was devised well before Astellas announced in December 2011 that it was pulling alefacept from the market, she said in an interview.
Physicians may also be surprised that the guidelines, published in Archives of Dermatology (2012;148:95-102), recommend cyclosporine as a short-term solution for up to 12 weeks to control a psoriasis flare.
"We know that you can use cyclosporine for up to a year because it’s FDA-approved for up to 12 months, but the typical use is as a quick-fix to transition to other agents that are slower acting," she said. "It’s a practical table."
Established drugs such as etanercept (Enbrel) and adalimumab (Humira) are recommended as first-line therapies for plaque psoriasis, but so is the newer monoclonal antibody ustekinumab (Stelara), so avoid the misconception that dermatologists need to prescribe systemic medications in a particular order based upon time on the market, Dr. Hsu noted.
The National Psoriasis Foundation (NPF) guidelines, however, do not provide a formula for how to treat patients, said coauthor Dr. Steven R. Feldman, professor of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C.
"They don’t say if you have this patient, this must be done first and then if that doesn’t work, do this second, and that third," he said. "There is no guideline of that type for psoriasis because no one treatment is better than all the other treatments in every way and patients weigh the various variables differently."
The new guidelines were adapted from the Canadian Guidelines for the Management of Plaque Psoriasis to reflect United States practice patterns, and are very similar to those published on the subject by the American Academy of Dermatology (AAD), Dr. Feldman said.
"I think these guidelines are clearly consensus guidelines, getting the experts together and deciding what they feel is best, whereas the AAD made a special effort to create evidence-based guidelines based on the available data," he said. "In the end, I think the recommendations are basically the same thing. Neither one tells physicians exactly what to do in a given situation; it’s more about describing the reasonable options that a patient and physician could choose in any situation."
NPF and AAD guidelines coauthor Dr. Mark Lebwohl, professor and chair of the department dermatology at Mount Sinai School of Medicine in New York, said the AAD guidelines are superb, but that "having this second set of guidelines allows us to answer even more questions."
He noted that there is often information in one guideline that is missing from another because new psoriasis developments happen so quickly. For example, a section recommending that physicians consider withholding tumor necrosis factor antagonists in psoriasis patients undergoing elective surgery was borrowed from the Canadian guidelines, but does not appear in the AAD guidelines, he said. The section discusses the controversy over the potential for increased risk of postsurgical infection in patients on biologics, and provides guidance on when specific agents should be withheld.
Conversely, the NPF guidelines discuss the pros and cons of the excimer laser, but this discussion is missing from the Canadian guidelines because the laser is not approved there.
"In a year, the guidelines will all be out of date again; they are a work in progress," said Dr. Lebwohl, who added that the NPF is trying to find a mechanism to update the guidelines in real time as new developments arise.
What the NPF/Canadian guidelines offer physicians is a rigorous evaluation of the literature with a patient-focused approach to psoriasis treatment, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., who helped coauthor both sets of guidelines.
"We all appreciate that the art of medicine incorporates habit, consensus, experience, and hopefully sound data," he said. "Consequently, a second and perhaps equally important offering of the guidelines to physicians is an independent affirmation or an independent negation of one’s art in treating psoriasis."
Where the NPF guidelines may prove particularly helpful is in supporting that therapeutic choice with insurers. If a physician thought that a particular biologic agent was the best treatment option for a patient, that physician could point to the guidelines as a way to let insurers know that what is being recommended is within the standard of care, said Dr. Feldman.
The new guidelines could also push physicians to be more aggressive in using some of the available systemic agents. He pointed out that one of the "major enthusiasms" for creating the guidelines was a 2009 National Psoriasis Foundation survey finding that 57% of patients with severe psoriasis were receiving treatment insufficient to achieve adequate control (J. Am. Acad. Dermatol. 2007;57:957-62).
"There needed to be guidelines that would encourage physicians to provide patients treatment that met the severity of their illness," said Dr. Feldman, who suggested that safety concerns, dated training, and a perception that psoriasis isn’t such a bad disease, all played into the lack of aggressive treatment.
The NPF guidelines clearly detail the safety risks associated with each agent, and have a specific section on special populations, including pregnant women, patients with HIV, and those with hepatitis B or C.
For the purpose of the new guidelines, patients are considered to have moderate to severe psoriasis if they cannot achieve or would not be expected to achieve adequate control using topical agents, with adequacy defined by the patient’s own perception of the disease and its burdens. Cutoffs based on clinical metrics, such as the Psoriasis Area and Severity Index, percentage of body surface area affected, and the Dermatological Life Quality Index are necessary in clinical trials, but "have little value in daily practice," the authors noted.
Strict numeric cutoffs can also cause problems with insurers and reimbursement.
"This [document] provides the insurers and other regulators the opportunity to set more meaningful standards for their policies," Dr. Feldman said. "When they see guidelines published in the Archives and supported by the National Psoriasis Foundation, it lends a lot of weight to doing things the way doctors do things in the patient’s best interest."
The physicians interviewed for this story all disclosed consultancies and research arrangements with multiple companies that manufacture psoriasis treatments.
The new National Psoriasis Foundation consensus guidelines on the management of plaque psoriasis take a practical approach to helping physicians manage the disease beyond topical treatment.
The guidelines feature a table summarizing the latest consensus on eight systemic medications, and they hone in on specific forms of psoriasis in a way that is more quickly understandable than in the past, according to lead author Dr. Sylvia Hsu, chief of Ben Taub Dermatology Service and a dermatology professor at Baylor College of Medicine in Houston.
"The aim is to simplify and clarify when to use these drugs for patients whose psoriasis is beyond topical treatment," she said. "A lot of the drugs can be used first line, so it’s not like a treatment algorithm."
Dr. Hsu acknowledged that the guidelines are a bit bold because they directly state, for example, that acitretin doesn’t work well for plaque psoriasis, and that evidence is scant to support the use of alefacept to achieve full clearance. The latter position was devised well before Astellas announced in December 2011 that it was pulling alefacept from the market, she said in an interview.
Physicians may also be surprised that the guidelines, published in Archives of Dermatology (2012;148:95-102), recommend cyclosporine as a short-term solution for up to 12 weeks to control a psoriasis flare.
"We know that you can use cyclosporine for up to a year because it’s FDA-approved for up to 12 months, but the typical use is as a quick-fix to transition to other agents that are slower acting," she said. "It’s a practical table."
Established drugs such as etanercept (Enbrel) and adalimumab (Humira) are recommended as first-line therapies for plaque psoriasis, but so is the newer monoclonal antibody ustekinumab (Stelara), so avoid the misconception that dermatologists need to prescribe systemic medications in a particular order based upon time on the market, Dr. Hsu noted.
The National Psoriasis Foundation (NPF) guidelines, however, do not provide a formula for how to treat patients, said coauthor Dr. Steven R. Feldman, professor of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C.
"They don’t say if you have this patient, this must be done first and then if that doesn’t work, do this second, and that third," he said. "There is no guideline of that type for psoriasis because no one treatment is better than all the other treatments in every way and patients weigh the various variables differently."
The new guidelines were adapted from the Canadian Guidelines for the Management of Plaque Psoriasis to reflect United States practice patterns, and are very similar to those published on the subject by the American Academy of Dermatology (AAD), Dr. Feldman said.
"I think these guidelines are clearly consensus guidelines, getting the experts together and deciding what they feel is best, whereas the AAD made a special effort to create evidence-based guidelines based on the available data," he said. "In the end, I think the recommendations are basically the same thing. Neither one tells physicians exactly what to do in a given situation; it’s more about describing the reasonable options that a patient and physician could choose in any situation."
NPF and AAD guidelines coauthor Dr. Mark Lebwohl, professor and chair of the department dermatology at Mount Sinai School of Medicine in New York, said the AAD guidelines are superb, but that "having this second set of guidelines allows us to answer even more questions."
He noted that there is often information in one guideline that is missing from another because new psoriasis developments happen so quickly. For example, a section recommending that physicians consider withholding tumor necrosis factor antagonists in psoriasis patients undergoing elective surgery was borrowed from the Canadian guidelines, but does not appear in the AAD guidelines, he said. The section discusses the controversy over the potential for increased risk of postsurgical infection in patients on biologics, and provides guidance on when specific agents should be withheld.
Conversely, the NPF guidelines discuss the pros and cons of the excimer laser, but this discussion is missing from the Canadian guidelines because the laser is not approved there.
"In a year, the guidelines will all be out of date again; they are a work in progress," said Dr. Lebwohl, who added that the NPF is trying to find a mechanism to update the guidelines in real time as new developments arise.
What the NPF/Canadian guidelines offer physicians is a rigorous evaluation of the literature with a patient-focused approach to psoriasis treatment, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., who helped coauthor both sets of guidelines.
"We all appreciate that the art of medicine incorporates habit, consensus, experience, and hopefully sound data," he said. "Consequently, a second and perhaps equally important offering of the guidelines to physicians is an independent affirmation or an independent negation of one’s art in treating psoriasis."
Where the NPF guidelines may prove particularly helpful is in supporting that therapeutic choice with insurers. If a physician thought that a particular biologic agent was the best treatment option for a patient, that physician could point to the guidelines as a way to let insurers know that what is being recommended is within the standard of care, said Dr. Feldman.
The new guidelines could also push physicians to be more aggressive in using some of the available systemic agents. He pointed out that one of the "major enthusiasms" for creating the guidelines was a 2009 National Psoriasis Foundation survey finding that 57% of patients with severe psoriasis were receiving treatment insufficient to achieve adequate control (J. Am. Acad. Dermatol. 2007;57:957-62).
"There needed to be guidelines that would encourage physicians to provide patients treatment that met the severity of their illness," said Dr. Feldman, who suggested that safety concerns, dated training, and a perception that psoriasis isn’t such a bad disease, all played into the lack of aggressive treatment.
The NPF guidelines clearly detail the safety risks associated with each agent, and have a specific section on special populations, including pregnant women, patients with HIV, and those with hepatitis B or C.
For the purpose of the new guidelines, patients are considered to have moderate to severe psoriasis if they cannot achieve or would not be expected to achieve adequate control using topical agents, with adequacy defined by the patient’s own perception of the disease and its burdens. Cutoffs based on clinical metrics, such as the Psoriasis Area and Severity Index, percentage of body surface area affected, and the Dermatological Life Quality Index are necessary in clinical trials, but "have little value in daily practice," the authors noted.
Strict numeric cutoffs can also cause problems with insurers and reimbursement.
"This [document] provides the insurers and other regulators the opportunity to set more meaningful standards for their policies," Dr. Feldman said. "When they see guidelines published in the Archives and supported by the National Psoriasis Foundation, it lends a lot of weight to doing things the way doctors do things in the patient’s best interest."
The physicians interviewed for this story all disclosed consultancies and research arrangements with multiple companies that manufacture psoriasis treatments.