User login
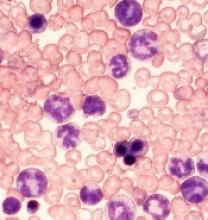
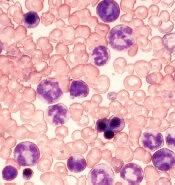
Researchers say they have identified 2 signaling proteins that enable resistance to tyrosine kinase inhibitors (TKIs) and could be therapeutic targets for acute lymphoblastic leukemia (ALL) and chronic myeloid leukemia (CML).
The team found that by deleting these proteins—c-Fos and Dusp1—they could eradicate BCR-ABL-induced B-cell ALL in mice.
And treatment combining c-Fos and Dusp1 inhibitors with the TKI imatinib was able to cure mice with BCR-ABL-driven CML.
The researchers reported these findings in Nature Medicine.
“We think that, within the next 5 years, our data will change the way people think about cancer development and targeted therapy,” said study author Mohammad Azam, PhD, of Cincinnati Children’s Hospital Medical Center in Ohio.
“This study identifies a potential Achilles’ heel of kinase-driven cancers, and what we propose is intended to be curative, not just treatment.”
The potential Achilles’ heel is a common point of passage in cells—a signaling node—that appears to be required to generate cancer cells. The node is formed by the signaling proteins c-Fos and Dusp1, according to the researchers.
The team identified c-Fos and Dusp1 by conducting global gene-expression analysis of mouse leukemia cells and human CML cells. Analysis of the human cells revealed extremely high levels of c-FOS and DUSP1 in BCR-ABL-positive, TKI-resistant cells.
Dr Azam and his colleagues found that signaling from tyrosine kinase and growth factor proteins that support cell expansion (such as IL-3 and IL-6) converge to elevate c-Fos and Dusp1 levels in leukemia cells.
Working together, these molecules maintain the survival of leukemia stem cells (LSCs), which translates to minimal residual disease (MRD) after treatment.
Dr Azam said Dusp1 and c-Fos support the survival of LSCs by increasing the toxic threshold needed to kill them. This means imatinib and other TKIs cannot eliminate the residual LSCs.
After describing the roles of c-Fos and Dusp1, Dr Azam and his colleagues put their ideas to the test in mouse models of CML.
The team tested several treatments in these mice, including:
- monotherapy with imatinib
- inhibitors of c-Fos and Dusp1
- treatment with imatinib and inhibitors of c-Fos and Dusp1.
As suspected, treatment with imatinib alone initially stopped CML progression, but mice ultimately relapsed.
Treatment with c-Fos and Dusp1 inhibitors significantly slowed CML progression and prolonged survival in a majority of mice, but this treatment wasn’t curative.
However, a month of treatment with c-Fos and Dusp1 inhibitors as well as imatinib cured about 90% of mice with CML, and there were no signs of MRD.
The researchers also found that simply deleting c-Fos and Dusp1 was sufficient to block the development of B-cell ALL in mice.
The team said they are following up this study by testing c-Fos and Dusp1 as treatment targets for different kinase-fueled cancers. ![]()
Researchers say they have identified 2 signaling proteins that enable resistance to tyrosine kinase inhibitors (TKIs) and could be therapeutic targets for acute lymphoblastic leukemia (ALL) and chronic myeloid leukemia (CML).
The team found that by deleting these proteins—c-Fos and Dusp1—they could eradicate BCR-ABL-induced B-cell ALL in mice.
And treatment combining c-Fos and Dusp1 inhibitors with the TKI imatinib was able to cure mice with BCR-ABL-driven CML.
The researchers reported these findings in Nature Medicine.
“We think that, within the next 5 years, our data will change the way people think about cancer development and targeted therapy,” said study author Mohammad Azam, PhD, of Cincinnati Children’s Hospital Medical Center in Ohio.
“This study identifies a potential Achilles’ heel of kinase-driven cancers, and what we propose is intended to be curative, not just treatment.”
The potential Achilles’ heel is a common point of passage in cells—a signaling node—that appears to be required to generate cancer cells. The node is formed by the signaling proteins c-Fos and Dusp1, according to the researchers.
The team identified c-Fos and Dusp1 by conducting global gene-expression analysis of mouse leukemia cells and human CML cells. Analysis of the human cells revealed extremely high levels of c-FOS and DUSP1 in BCR-ABL-positive, TKI-resistant cells.
Dr Azam and his colleagues found that signaling from tyrosine kinase and growth factor proteins that support cell expansion (such as IL-3 and IL-6) converge to elevate c-Fos and Dusp1 levels in leukemia cells.
Working together, these molecules maintain the survival of leukemia stem cells (LSCs), which translates to minimal residual disease (MRD) after treatment.
Dr Azam said Dusp1 and c-Fos support the survival of LSCs by increasing the toxic threshold needed to kill them. This means imatinib and other TKIs cannot eliminate the residual LSCs.
After describing the roles of c-Fos and Dusp1, Dr Azam and his colleagues put their ideas to the test in mouse models of CML.
The team tested several treatments in these mice, including:
- monotherapy with imatinib
- inhibitors of c-Fos and Dusp1
- treatment with imatinib and inhibitors of c-Fos and Dusp1.
As suspected, treatment with imatinib alone initially stopped CML progression, but mice ultimately relapsed.
Treatment with c-Fos and Dusp1 inhibitors significantly slowed CML progression and prolonged survival in a majority of mice, but this treatment wasn’t curative.
However, a month of treatment with c-Fos and Dusp1 inhibitors as well as imatinib cured about 90% of mice with CML, and there were no signs of MRD.
The researchers also found that simply deleting c-Fos and Dusp1 was sufficient to block the development of B-cell ALL in mice.
The team said they are following up this study by testing c-Fos and Dusp1 as treatment targets for different kinase-fueled cancers. ![]()
Researchers say they have identified 2 signaling proteins that enable resistance to tyrosine kinase inhibitors (TKIs) and could be therapeutic targets for acute lymphoblastic leukemia (ALL) and chronic myeloid leukemia (CML).
The team found that by deleting these proteins—c-Fos and Dusp1—they could eradicate BCR-ABL-induced B-cell ALL in mice.
And treatment combining c-Fos and Dusp1 inhibitors with the TKI imatinib was able to cure mice with BCR-ABL-driven CML.
The researchers reported these findings in Nature Medicine.
“We think that, within the next 5 years, our data will change the way people think about cancer development and targeted therapy,” said study author Mohammad Azam, PhD, of Cincinnati Children’s Hospital Medical Center in Ohio.
“This study identifies a potential Achilles’ heel of kinase-driven cancers, and what we propose is intended to be curative, not just treatment.”
The potential Achilles’ heel is a common point of passage in cells—a signaling node—that appears to be required to generate cancer cells. The node is formed by the signaling proteins c-Fos and Dusp1, according to the researchers.
The team identified c-Fos and Dusp1 by conducting global gene-expression analysis of mouse leukemia cells and human CML cells. Analysis of the human cells revealed extremely high levels of c-FOS and DUSP1 in BCR-ABL-positive, TKI-resistant cells.
Dr Azam and his colleagues found that signaling from tyrosine kinase and growth factor proteins that support cell expansion (such as IL-3 and IL-6) converge to elevate c-Fos and Dusp1 levels in leukemia cells.
Working together, these molecules maintain the survival of leukemia stem cells (LSCs), which translates to minimal residual disease (MRD) after treatment.
Dr Azam said Dusp1 and c-Fos support the survival of LSCs by increasing the toxic threshold needed to kill them. This means imatinib and other TKIs cannot eliminate the residual LSCs.
After describing the roles of c-Fos and Dusp1, Dr Azam and his colleagues put their ideas to the test in mouse models of CML.
The team tested several treatments in these mice, including:
- monotherapy with imatinib
- inhibitors of c-Fos and Dusp1
- treatment with imatinib and inhibitors of c-Fos and Dusp1.
As suspected, treatment with imatinib alone initially stopped CML progression, but mice ultimately relapsed.
Treatment with c-Fos and Dusp1 inhibitors significantly slowed CML progression and prolonged survival in a majority of mice, but this treatment wasn’t curative.
However, a month of treatment with c-Fos and Dusp1 inhibitors as well as imatinib cured about 90% of mice with CML, and there were no signs of MRD.
The researchers also found that simply deleting c-Fos and Dusp1 was sufficient to block the development of B-cell ALL in mice.
The team said they are following up this study by testing c-Fos and Dusp1 as treatment targets for different kinase-fueled cancers. ![]()