User login
In our current society, it is likely that the “skinny patient with COPD” who walks into your clinic is less and less your “traditional” patient with COPD. We are seeing in our health care systems more of the “blue bloaters” – patients with COPD and significant obesity. This phenotype is representing what we are seeing worldwide as a consequence of the rising obesity prevalence. In the United States, the prepandemic (2017-2020) estimated percentage of adults over the age of 40 with obesity, defined as a body mass index (BMI) of at least 30 kg/m2, was over 40%. Moreover, the estimated percentage of adults with morbid obesity (BMI at least 40 kg/m2) is close to 10% (Akinbami, LJ et al. Vital Health Stat. 2022:190:1-36) and trending up. These patients with the “triple overlap” of morbid obesity, COPD, and awake daytime hypercapnia are being seen in clinics and in-hospital settings with increasing frequency, often presenting with complicating comorbidities such as acute respiratory failure, acute heart failure, kidney disease, or pulmonary hypertension. We are now faced with managing these patients with complex disease.
The obesity paradox does not seem applicable in the triple overlap phenotype. Patients with COPD who are overweight, defined as “mild obesity,” have lower mortality when compared with normal weight and underweight patients with COPD; however, this effect diminishes when BMI increases beyond 32 kg/m2. With increasing obesity severity and aging, the risk of both obstructive sleep apnea (OSA) and hypoventilation increases. It is well documented that COPD-OSA overlap is linked to worse outcomes and that continuous positive airway pressure (CPAP) as first-line therapy decreases readmission rates and mortality. The pathophysiology of hypoventilation in obesity is complex and multifactorial, and, although significant overlaps likely exist with comorbid COPD, by current definitions, to establish a diagnosis of obesity hypoventilation syndrome (OHS), one must have excluded other causes of hypoventilation, such as COPD.
These patients with the triple overlap of morbid obesity, awake daytime hypercapnia, and COPD are the subset of patients that providers struggle to fit in a diagnosis or in clinical research trials.
The triple overlap is a distinct syndrome
Different labels have been used in the medical literature: hypercapnic OSA-COPD overlap, morbid obesity and OSA-COPD overlap, hypercapnic morbidly obese COPD and OHS-COPD overlap. A better characterization of this distinctive phenotype is much needed. Patients with OSA-COPD overlap, for example, have an increased propensity to develop hypercapnia at higher FEV1 when compared with COPD without OSA – but this is thought to be a consequence of prolonged and frequent apneas and hypopneas compounded with obesity-related central hypoventilation. We found that morbidly obese patients with OSA-COPD overlap have a higher hypoxia burden, more severe OSA, and are frequently prescribed noninvasive ventilation after a failed titration polysomnogram (Htun ZM, et al. Am J Respir Crit Care Med. 2019;199:A1382), perhaps signaling a distinctive phenotype with worse outcomes, but the study had the inherent limitations of a single-center, retrospective design lacking data on awake hypercapnia. On the other side, the term OHS-COPD is contradictory and confusing based on current OHS diagnostic criteria.
In standardizing diagnostic criteria for patients with this triple overlap syndrome, challenges remain: would the patient with a BMI of 70 kg/m2 and fixed chronic airflow obstruction with FEV1 72% fall under the category of hypercapnic COPD vs OHS? Do these patients have worse outcomes regardless of their predominant feature? Would outcomes change if the apnea hypopnea index (AHI) is 10/h vs 65/h? More importantly, do patients with the triple overlap of COPD, morbid obesity, and daytime hypercapnia have worse outcomes when compared with hypercapnic COPD, or OHS with/without OSA? These questions can be better addressed once we agree on a definition. The patients with triple overlap syndrome have been traditionally excluded from clinical trials: the patient with morbid obesity has been excluded from chronic hypercapnic COPD clinical trials, and the patient with COPD has been excluded from OHS trials.
There are no specific clinical guidelines for this triple overlap phenotype. Positive airway pressure is the mainstay of treatment. CPAP is recommended as first-line therapy for patients with OSA-COPD overlap syndrome, while noninvasive ventilation (NIV) with bilevel positive airway pressure (BPAP) is recommended as first-line for the stable ambulatory hypercapnic patient with COPD. It is unclear if NIV is superior to CPAP in patients with triple overlap syndrome, although recently published data showed greater efficacy in reducing carbon dioxide (PaCO2) and improving quality of life in a small group of subjects (Zheng et al. J Clin Sleep Med. 2022;18[1]:99-107). To take a step further, the subtleties of NIV set up, such as rise time and minimum inspiratory time, are contradictory: the goal in ventilating patients with COPD is to shorten inspiratory time, prolonging expiratory time, therefore allowing a shortened inspiratory cycle. In obesity, ventilation strategies aim to prolong and sustain inspiratory time to improve ventilation and dependent atelectasis. Another area of uncertainty is device selection. Should we aim to provide a respiratory assist device (RAD): the traditional, rent to own bilevel PAP without auto-expiratory positive airway pressure (EPAP) capabilities and lower maximum inspiratory pressure delivery capacity, vs a home mechanical ventilator at a higher expense, life-time rental, and one-way only data monitoring, which limits remote prescription adjustments, but allow auto-EPAP settings for patients with comorbid OSA? More importantly, how do we get these patients, who do not fit in any of the specified insurance criteria for PAP therapy approved for treatment?
A uniform diagnostic definition and clear taxonomy allows for resource allocation, from government funded grants for clinical trials to a better-informed distribution of health care systems resources and support health care policy changes to improve patient-centric outcomes. Here, we propose that the morbidly obese patient (BMI >40 kg/m2) with chronic airflow obstruction and a forced expiratory ratio (FEV1/FVC) <0.7 with awake daytime hypercapnia (PaCO2 > 45 mm Hg) represents a different entity/phenotype and fits best under the triple overlap syndrome taxonomy.
We suspect that these patients have worse outcomes, including comorbidity burden, quality of life, exacerbation rates, longer hospital length-of-stay, and respiratory and all-cause mortality. Large, multicenter, controlled trials comparing the long-term effectiveness of NIV and CPAP: measurements of respiratory function, gas exchange, blood pressure, and health related quality of life are needed. This is a group of patients that may specifically benefit from volume-targeted pressure support mode ventilation with auto-EPAP capabilities upon discharge from the hospital after an acute exacerbation.
Inpatient (sleep medicine) and outpatient transitions
In patients hospitalized with the triple overlap syndrome, there are certain considerations that are of special interest. Given comorbid hypercapnia and limited data on NIV superiority over CPAP, a sleep study should not be needed for NIV qualification. In addition, the medical team may consider the following (Figure 1):
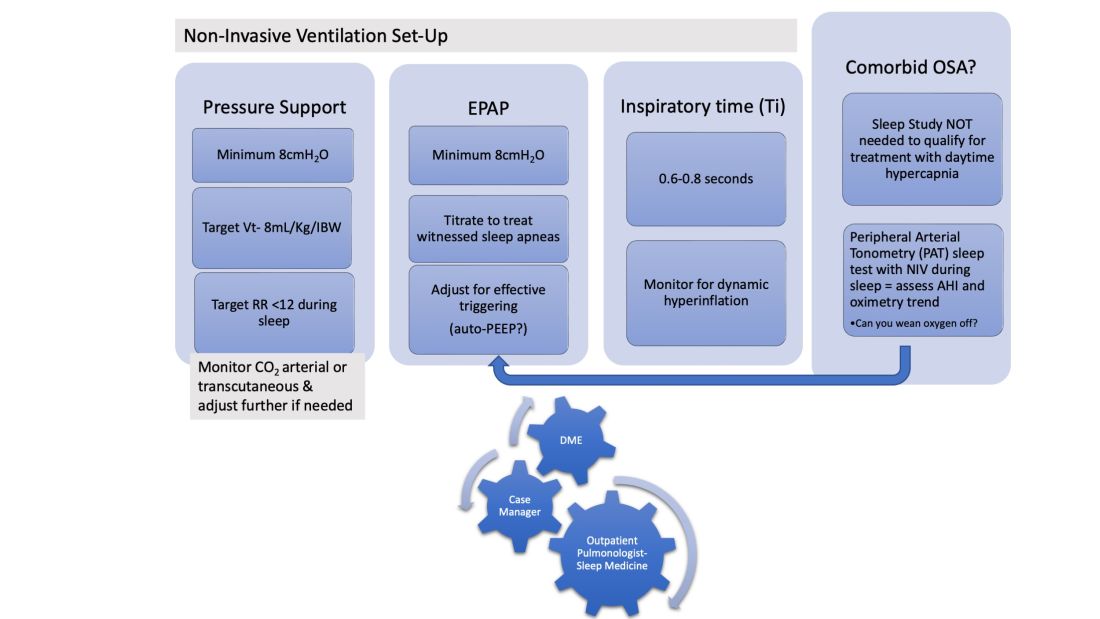
1. Noninvasive Ventilation:
a. Maintaining a high-pressure support differential between inspiratory positive airway pressure (IPAP) and EPAP. This can usually be achieved at 8-10 cm H2O, further adjusting to target a tidal volume (Vt) of 8 mL/kg of ideal body weight (IBW).
b. Higher EPAP: To overcome dependent atelectasis, improve ventilation-perfusion (VQ) matching, and better treat upper airway resistance both during wakefulness and sleep. Also, adjustments of EPAP at bedside should be considered to counteract auto-PEEP-related ineffective triggering if observed.
c. OSA screening and EPAP adjustment: for high residual obstructive apneas or hypopneas if data are available on the NIV device, or with the use of peripheral arterial tonometry sleep testing devices with NIV on overnight before discharge.
d. Does the patient meet criteria for oxygen supplementation at home? Wean oxygen off, if possible.
2. Case-managers can help establish services with a durable medical equipment provider with expertise in advanced PAP devices.3. Obesity management, Consider referral to an obesity management program for lifestyle/dietary modifications along with pharmacotherapy or bariatric surgery interventions.
4. Close follow-up, track exacerbations. Device download data are crucial to monitor adherence/tolerance and treatment effectiveness with particular interest in AHI, oximetry, and CO2 trends monitoring. Some patients may need dedicated titration polysomnograms to adjust ventilation settings, for optimization of residual OSA or for oxygen addition or discontinuation.
Conclusion
Patients with the triple overlap phenotype have not been systematically defined, studied, or included in clinical trials. We anticipate that these patients have worse outcomes: quality of life, symptom and comorbidity burden, exacerbation rates, in-hospital mortality, longer hospital stay and ICU stay, and respiratory and all-cause mortality. This is a group of patients that may specifically benefit from domiciliary NIV set-up upon discharge from the hospital with close follow-up. Properly identifying these patients will help pulmonologists and health care systems direct resources to optimally manage this complex group of patients. Funding of research trials to support clinical guidelines development should be prioritized. Triple overlap syndrome is different from COPD-OSA overlap, OHS with moderate to severe OSA, or OHS without significant OSA.
In our current society, it is likely that the “skinny patient with COPD” who walks into your clinic is less and less your “traditional” patient with COPD. We are seeing in our health care systems more of the “blue bloaters” – patients with COPD and significant obesity. This phenotype is representing what we are seeing worldwide as a consequence of the rising obesity prevalence. In the United States, the prepandemic (2017-2020) estimated percentage of adults over the age of 40 with obesity, defined as a body mass index (BMI) of at least 30 kg/m2, was over 40%. Moreover, the estimated percentage of adults with morbid obesity (BMI at least 40 kg/m2) is close to 10% (Akinbami, LJ et al. Vital Health Stat. 2022:190:1-36) and trending up. These patients with the “triple overlap” of morbid obesity, COPD, and awake daytime hypercapnia are being seen in clinics and in-hospital settings with increasing frequency, often presenting with complicating comorbidities such as acute respiratory failure, acute heart failure, kidney disease, or pulmonary hypertension. We are now faced with managing these patients with complex disease.
The obesity paradox does not seem applicable in the triple overlap phenotype. Patients with COPD who are overweight, defined as “mild obesity,” have lower mortality when compared with normal weight and underweight patients with COPD; however, this effect diminishes when BMI increases beyond 32 kg/m2. With increasing obesity severity and aging, the risk of both obstructive sleep apnea (OSA) and hypoventilation increases. It is well documented that COPD-OSA overlap is linked to worse outcomes and that continuous positive airway pressure (CPAP) as first-line therapy decreases readmission rates and mortality. The pathophysiology of hypoventilation in obesity is complex and multifactorial, and, although significant overlaps likely exist with comorbid COPD, by current definitions, to establish a diagnosis of obesity hypoventilation syndrome (OHS), one must have excluded other causes of hypoventilation, such as COPD.
These patients with the triple overlap of morbid obesity, awake daytime hypercapnia, and COPD are the subset of patients that providers struggle to fit in a diagnosis or in clinical research trials.
The triple overlap is a distinct syndrome
Different labels have been used in the medical literature: hypercapnic OSA-COPD overlap, morbid obesity and OSA-COPD overlap, hypercapnic morbidly obese COPD and OHS-COPD overlap. A better characterization of this distinctive phenotype is much needed. Patients with OSA-COPD overlap, for example, have an increased propensity to develop hypercapnia at higher FEV1 when compared with COPD without OSA – but this is thought to be a consequence of prolonged and frequent apneas and hypopneas compounded with obesity-related central hypoventilation. We found that morbidly obese patients with OSA-COPD overlap have a higher hypoxia burden, more severe OSA, and are frequently prescribed noninvasive ventilation after a failed titration polysomnogram (Htun ZM, et al. Am J Respir Crit Care Med. 2019;199:A1382), perhaps signaling a distinctive phenotype with worse outcomes, but the study had the inherent limitations of a single-center, retrospective design lacking data on awake hypercapnia. On the other side, the term OHS-COPD is contradictory and confusing based on current OHS diagnostic criteria.
In standardizing diagnostic criteria for patients with this triple overlap syndrome, challenges remain: would the patient with a BMI of 70 kg/m2 and fixed chronic airflow obstruction with FEV1 72% fall under the category of hypercapnic COPD vs OHS? Do these patients have worse outcomes regardless of their predominant feature? Would outcomes change if the apnea hypopnea index (AHI) is 10/h vs 65/h? More importantly, do patients with the triple overlap of COPD, morbid obesity, and daytime hypercapnia have worse outcomes when compared with hypercapnic COPD, or OHS with/without OSA? These questions can be better addressed once we agree on a definition. The patients with triple overlap syndrome have been traditionally excluded from clinical trials: the patient with morbid obesity has been excluded from chronic hypercapnic COPD clinical trials, and the patient with COPD has been excluded from OHS trials.
There are no specific clinical guidelines for this triple overlap phenotype. Positive airway pressure is the mainstay of treatment. CPAP is recommended as first-line therapy for patients with OSA-COPD overlap syndrome, while noninvasive ventilation (NIV) with bilevel positive airway pressure (BPAP) is recommended as first-line for the stable ambulatory hypercapnic patient with COPD. It is unclear if NIV is superior to CPAP in patients with triple overlap syndrome, although recently published data showed greater efficacy in reducing carbon dioxide (PaCO2) and improving quality of life in a small group of subjects (Zheng et al. J Clin Sleep Med. 2022;18[1]:99-107). To take a step further, the subtleties of NIV set up, such as rise time and minimum inspiratory time, are contradictory: the goal in ventilating patients with COPD is to shorten inspiratory time, prolonging expiratory time, therefore allowing a shortened inspiratory cycle. In obesity, ventilation strategies aim to prolong and sustain inspiratory time to improve ventilation and dependent atelectasis. Another area of uncertainty is device selection. Should we aim to provide a respiratory assist device (RAD): the traditional, rent to own bilevel PAP without auto-expiratory positive airway pressure (EPAP) capabilities and lower maximum inspiratory pressure delivery capacity, vs a home mechanical ventilator at a higher expense, life-time rental, and one-way only data monitoring, which limits remote prescription adjustments, but allow auto-EPAP settings for patients with comorbid OSA? More importantly, how do we get these patients, who do not fit in any of the specified insurance criteria for PAP therapy approved for treatment?
A uniform diagnostic definition and clear taxonomy allows for resource allocation, from government funded grants for clinical trials to a better-informed distribution of health care systems resources and support health care policy changes to improve patient-centric outcomes. Here, we propose that the morbidly obese patient (BMI >40 kg/m2) with chronic airflow obstruction and a forced expiratory ratio (FEV1/FVC) <0.7 with awake daytime hypercapnia (PaCO2 > 45 mm Hg) represents a different entity/phenotype and fits best under the triple overlap syndrome taxonomy.
We suspect that these patients have worse outcomes, including comorbidity burden, quality of life, exacerbation rates, longer hospital length-of-stay, and respiratory and all-cause mortality. Large, multicenter, controlled trials comparing the long-term effectiveness of NIV and CPAP: measurements of respiratory function, gas exchange, blood pressure, and health related quality of life are needed. This is a group of patients that may specifically benefit from volume-targeted pressure support mode ventilation with auto-EPAP capabilities upon discharge from the hospital after an acute exacerbation.
Inpatient (sleep medicine) and outpatient transitions
In patients hospitalized with the triple overlap syndrome, there are certain considerations that are of special interest. Given comorbid hypercapnia and limited data on NIV superiority over CPAP, a sleep study should not be needed for NIV qualification. In addition, the medical team may consider the following (Figure 1):

1. Noninvasive Ventilation:
a. Maintaining a high-pressure support differential between inspiratory positive airway pressure (IPAP) and EPAP. This can usually be achieved at 8-10 cm H2O, further adjusting to target a tidal volume (Vt) of 8 mL/kg of ideal body weight (IBW).
b. Higher EPAP: To overcome dependent atelectasis, improve ventilation-perfusion (VQ) matching, and better treat upper airway resistance both during wakefulness and sleep. Also, adjustments of EPAP at bedside should be considered to counteract auto-PEEP-related ineffective triggering if observed.
c. OSA screening and EPAP adjustment: for high residual obstructive apneas or hypopneas if data are available on the NIV device, or with the use of peripheral arterial tonometry sleep testing devices with NIV on overnight before discharge.
d. Does the patient meet criteria for oxygen supplementation at home? Wean oxygen off, if possible.
2. Case-managers can help establish services with a durable medical equipment provider with expertise in advanced PAP devices.3. Obesity management, Consider referral to an obesity management program for lifestyle/dietary modifications along with pharmacotherapy or bariatric surgery interventions.
4. Close follow-up, track exacerbations. Device download data are crucial to monitor adherence/tolerance and treatment effectiveness with particular interest in AHI, oximetry, and CO2 trends monitoring. Some patients may need dedicated titration polysomnograms to adjust ventilation settings, for optimization of residual OSA or for oxygen addition or discontinuation.
Conclusion
Patients with the triple overlap phenotype have not been systematically defined, studied, or included in clinical trials. We anticipate that these patients have worse outcomes: quality of life, symptom and comorbidity burden, exacerbation rates, in-hospital mortality, longer hospital stay and ICU stay, and respiratory and all-cause mortality. This is a group of patients that may specifically benefit from domiciliary NIV set-up upon discharge from the hospital with close follow-up. Properly identifying these patients will help pulmonologists and health care systems direct resources to optimally manage this complex group of patients. Funding of research trials to support clinical guidelines development should be prioritized. Triple overlap syndrome is different from COPD-OSA overlap, OHS with moderate to severe OSA, or OHS without significant OSA.
In our current society, it is likely that the “skinny patient with COPD” who walks into your clinic is less and less your “traditional” patient with COPD. We are seeing in our health care systems more of the “blue bloaters” – patients with COPD and significant obesity. This phenotype is representing what we are seeing worldwide as a consequence of the rising obesity prevalence. In the United States, the prepandemic (2017-2020) estimated percentage of adults over the age of 40 with obesity, defined as a body mass index (BMI) of at least 30 kg/m2, was over 40%. Moreover, the estimated percentage of adults with morbid obesity (BMI at least 40 kg/m2) is close to 10% (Akinbami, LJ et al. Vital Health Stat. 2022:190:1-36) and trending up. These patients with the “triple overlap” of morbid obesity, COPD, and awake daytime hypercapnia are being seen in clinics and in-hospital settings with increasing frequency, often presenting with complicating comorbidities such as acute respiratory failure, acute heart failure, kidney disease, or pulmonary hypertension. We are now faced with managing these patients with complex disease.
The obesity paradox does not seem applicable in the triple overlap phenotype. Patients with COPD who are overweight, defined as “mild obesity,” have lower mortality when compared with normal weight and underweight patients with COPD; however, this effect diminishes when BMI increases beyond 32 kg/m2. With increasing obesity severity and aging, the risk of both obstructive sleep apnea (OSA) and hypoventilation increases. It is well documented that COPD-OSA overlap is linked to worse outcomes and that continuous positive airway pressure (CPAP) as first-line therapy decreases readmission rates and mortality. The pathophysiology of hypoventilation in obesity is complex and multifactorial, and, although significant overlaps likely exist with comorbid COPD, by current definitions, to establish a diagnosis of obesity hypoventilation syndrome (OHS), one must have excluded other causes of hypoventilation, such as COPD.
These patients with the triple overlap of morbid obesity, awake daytime hypercapnia, and COPD are the subset of patients that providers struggle to fit in a diagnosis or in clinical research trials.
The triple overlap is a distinct syndrome
Different labels have been used in the medical literature: hypercapnic OSA-COPD overlap, morbid obesity and OSA-COPD overlap, hypercapnic morbidly obese COPD and OHS-COPD overlap. A better characterization of this distinctive phenotype is much needed. Patients with OSA-COPD overlap, for example, have an increased propensity to develop hypercapnia at higher FEV1 when compared with COPD without OSA – but this is thought to be a consequence of prolonged and frequent apneas and hypopneas compounded with obesity-related central hypoventilation. We found that morbidly obese patients with OSA-COPD overlap have a higher hypoxia burden, more severe OSA, and are frequently prescribed noninvasive ventilation after a failed titration polysomnogram (Htun ZM, et al. Am J Respir Crit Care Med. 2019;199:A1382), perhaps signaling a distinctive phenotype with worse outcomes, but the study had the inherent limitations of a single-center, retrospective design lacking data on awake hypercapnia. On the other side, the term OHS-COPD is contradictory and confusing based on current OHS diagnostic criteria.
In standardizing diagnostic criteria for patients with this triple overlap syndrome, challenges remain: would the patient with a BMI of 70 kg/m2 and fixed chronic airflow obstruction with FEV1 72% fall under the category of hypercapnic COPD vs OHS? Do these patients have worse outcomes regardless of their predominant feature? Would outcomes change if the apnea hypopnea index (AHI) is 10/h vs 65/h? More importantly, do patients with the triple overlap of COPD, morbid obesity, and daytime hypercapnia have worse outcomes when compared with hypercapnic COPD, or OHS with/without OSA? These questions can be better addressed once we agree on a definition. The patients with triple overlap syndrome have been traditionally excluded from clinical trials: the patient with morbid obesity has been excluded from chronic hypercapnic COPD clinical trials, and the patient with COPD has been excluded from OHS trials.
There are no specific clinical guidelines for this triple overlap phenotype. Positive airway pressure is the mainstay of treatment. CPAP is recommended as first-line therapy for patients with OSA-COPD overlap syndrome, while noninvasive ventilation (NIV) with bilevel positive airway pressure (BPAP) is recommended as first-line for the stable ambulatory hypercapnic patient with COPD. It is unclear if NIV is superior to CPAP in patients with triple overlap syndrome, although recently published data showed greater efficacy in reducing carbon dioxide (PaCO2) and improving quality of life in a small group of subjects (Zheng et al. J Clin Sleep Med. 2022;18[1]:99-107). To take a step further, the subtleties of NIV set up, such as rise time and minimum inspiratory time, are contradictory: the goal in ventilating patients with COPD is to shorten inspiratory time, prolonging expiratory time, therefore allowing a shortened inspiratory cycle. In obesity, ventilation strategies aim to prolong and sustain inspiratory time to improve ventilation and dependent atelectasis. Another area of uncertainty is device selection. Should we aim to provide a respiratory assist device (RAD): the traditional, rent to own bilevel PAP without auto-expiratory positive airway pressure (EPAP) capabilities and lower maximum inspiratory pressure delivery capacity, vs a home mechanical ventilator at a higher expense, life-time rental, and one-way only data monitoring, which limits remote prescription adjustments, but allow auto-EPAP settings for patients with comorbid OSA? More importantly, how do we get these patients, who do not fit in any of the specified insurance criteria for PAP therapy approved for treatment?
A uniform diagnostic definition and clear taxonomy allows for resource allocation, from government funded grants for clinical trials to a better-informed distribution of health care systems resources and support health care policy changes to improve patient-centric outcomes. Here, we propose that the morbidly obese patient (BMI >40 kg/m2) with chronic airflow obstruction and a forced expiratory ratio (FEV1/FVC) <0.7 with awake daytime hypercapnia (PaCO2 > 45 mm Hg) represents a different entity/phenotype and fits best under the triple overlap syndrome taxonomy.
We suspect that these patients have worse outcomes, including comorbidity burden, quality of life, exacerbation rates, longer hospital length-of-stay, and respiratory and all-cause mortality. Large, multicenter, controlled trials comparing the long-term effectiveness of NIV and CPAP: measurements of respiratory function, gas exchange, blood pressure, and health related quality of life are needed. This is a group of patients that may specifically benefit from volume-targeted pressure support mode ventilation with auto-EPAP capabilities upon discharge from the hospital after an acute exacerbation.
Inpatient (sleep medicine) and outpatient transitions
In patients hospitalized with the triple overlap syndrome, there are certain considerations that are of special interest. Given comorbid hypercapnia and limited data on NIV superiority over CPAP, a sleep study should not be needed for NIV qualification. In addition, the medical team may consider the following (Figure 1):

1. Noninvasive Ventilation:
a. Maintaining a high-pressure support differential between inspiratory positive airway pressure (IPAP) and EPAP. This can usually be achieved at 8-10 cm H2O, further adjusting to target a tidal volume (Vt) of 8 mL/kg of ideal body weight (IBW).
b. Higher EPAP: To overcome dependent atelectasis, improve ventilation-perfusion (VQ) matching, and better treat upper airway resistance both during wakefulness and sleep. Also, adjustments of EPAP at bedside should be considered to counteract auto-PEEP-related ineffective triggering if observed.
c. OSA screening and EPAP adjustment: for high residual obstructive apneas or hypopneas if data are available on the NIV device, or with the use of peripheral arterial tonometry sleep testing devices with NIV on overnight before discharge.
d. Does the patient meet criteria for oxygen supplementation at home? Wean oxygen off, if possible.
2. Case-managers can help establish services with a durable medical equipment provider with expertise in advanced PAP devices.3. Obesity management, Consider referral to an obesity management program for lifestyle/dietary modifications along with pharmacotherapy or bariatric surgery interventions.
4. Close follow-up, track exacerbations. Device download data are crucial to monitor adherence/tolerance and treatment effectiveness with particular interest in AHI, oximetry, and CO2 trends monitoring. Some patients may need dedicated titration polysomnograms to adjust ventilation settings, for optimization of residual OSA or for oxygen addition or discontinuation.
Conclusion
Patients with the triple overlap phenotype have not been systematically defined, studied, or included in clinical trials. We anticipate that these patients have worse outcomes: quality of life, symptom and comorbidity burden, exacerbation rates, in-hospital mortality, longer hospital stay and ICU stay, and respiratory and all-cause mortality. This is a group of patients that may specifically benefit from domiciliary NIV set-up upon discharge from the hospital with close follow-up. Properly identifying these patients will help pulmonologists and health care systems direct resources to optimally manage this complex group of patients. Funding of research trials to support clinical guidelines development should be prioritized. Triple overlap syndrome is different from COPD-OSA overlap, OHS with moderate to severe OSA, or OHS without significant OSA.

