User login
Combination Guselkumab and Secukinumab for Plaque Psoriasis
To the Editor:
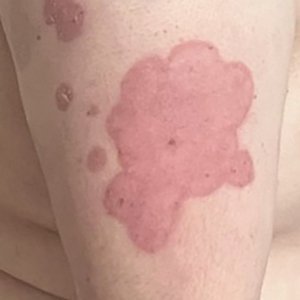
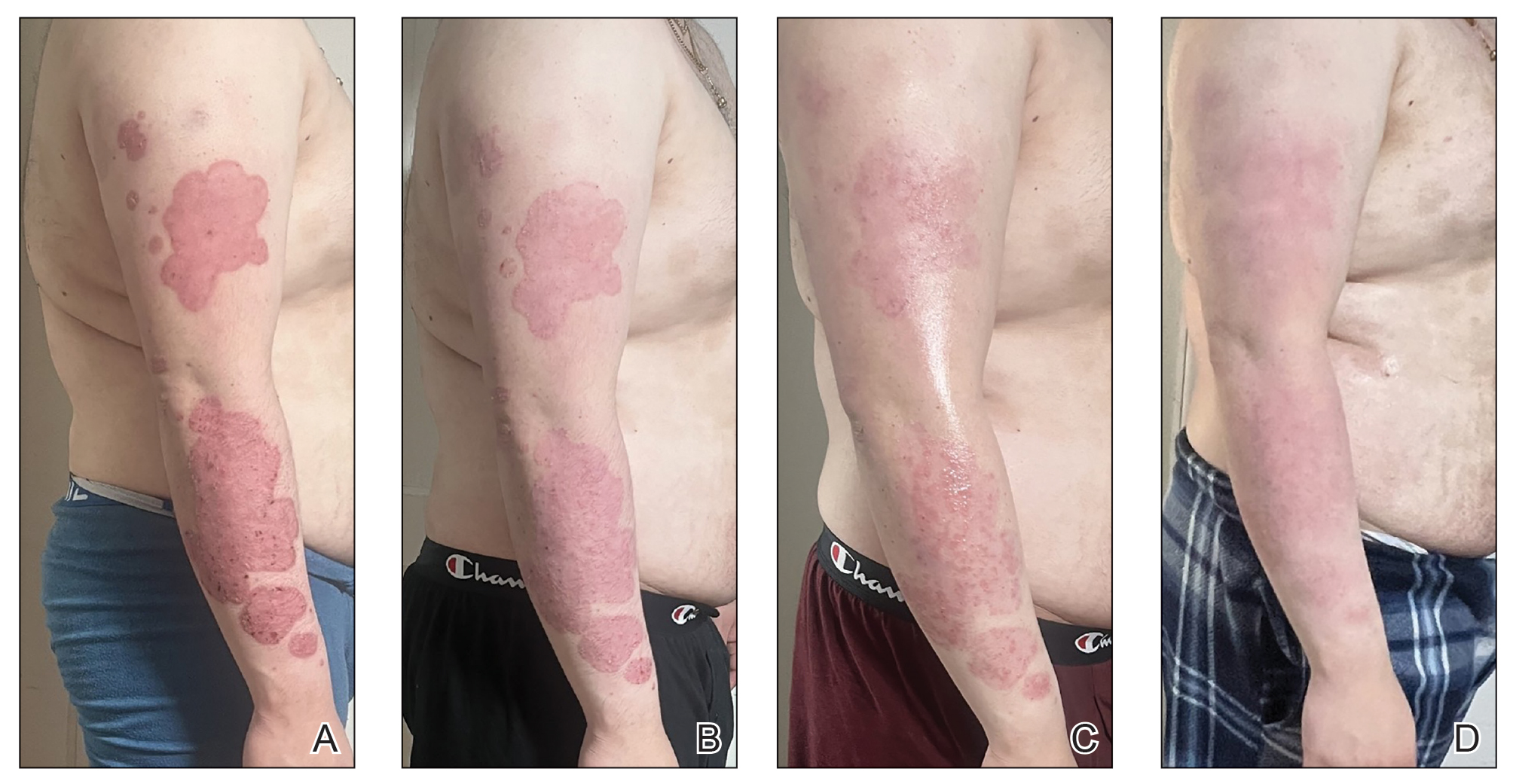
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.
Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
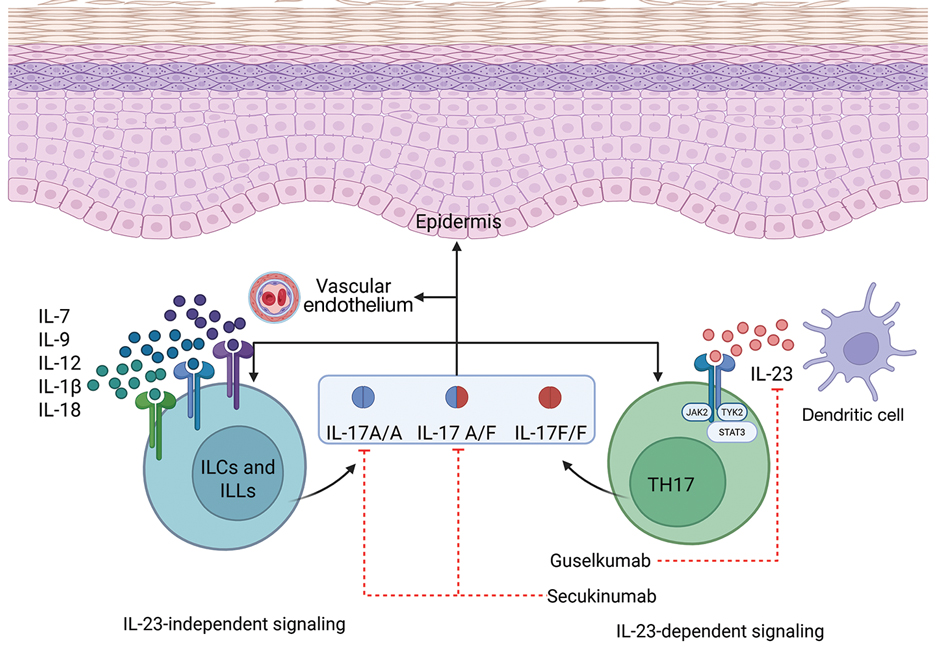
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.
The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Practice Points
- Combination therapy with 2 complementary biologics for psoriasis may have synergistic effects.
- Washout periods likely are unnecessary when switching between guselkumab and secukinumab.