User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
A third person living with HIV has been cured by transplant
In a first, If she remains off treatment without any hint of HIV, she would be only the third person in the world – after the Berlin Patient and the London Patient – to be cured through a transplant.
“Her own virus could not infect her cells,” said Yvonne Bryson, MD, chief of pediatric infectious diseases at the University of California, Los Angeles, who presented the study at the Conference on Retroviruses and Opportunistic Infections, which both presenters and the audience attended remotely.
The middle-aged New York woman of mixed race, who has asked that her specific race and age not be shared to protect her privacy, was diagnosed with HIV in 2013 when she was still in the very early stages of infection. She started treatment immediately and quickly achieved an undetectable viral load. An undetectable viral load not only prevents someone from transmitting HIV to others but also reduces or eliminates HIV replication, which means fewer variants and less time for the virus to infiltrate cells where it can hide.
But in 2017, she was diagnosed with leukemia. As a last resort to cure her of the cancer, she received a combination of adult stem cells from a relative’s blood that closely matched her own and umbilical cord blood obtained from a cord blood bank. That particular sample of cord blood was selected for its genetic mutation against the CCR5 receptor on immune cells, CD4 T cells. That mutation makes the immune system resistant to HIV.
The two previous HIV cures, of Berlin Patient Timothy Ray Brown and London Patient Adam Castillejo, also used stem cell transplantation with a CCR5 mutation, but theirs were bone marrow transplants. Bone marrow transplants are more arduous than cord blood transplants, which are commonly used in pediatric cancer treatment.
In this case, the physicians treating her used both.
“This allows the adult cells to accelerate and grow up until the cord blood takes over,” said Dr. Bryson. During her presentation, Dr. Bryson pointed to two types of data: First, she presented data showing the level of HIV in the patient’s blood. Soon after HIV diagnosis and treatment, her viral load dropped to undetectable levels. She had a spike of virus when she received the transplant, but then it went back to undetectable and has stayed that way ever since.
Meanwhile, following the transplant, her immune system started rebuilding itself using the new, HIV-resistant cells provided in the transplant. As her care team watched, no graft-versus-host (GVH) disease, a common side effect of stem cell transplants, emerged. In fact, the transplant went so well that she was discharged early from the hospital.
One hundred days after the transplant, the immune system contained within the cord blood had taken over. Her CD4 immune cells returned to normal levels a little more than a year after the transplant. By 27 months, she decided to stop all HIV treatment to see if the transplant had worked.
This was the real test. But as Dr. Bryson and colleagues continued to watch her HIV viral load and her CD4 counts and search for infectious virus, they didn’t find any. She tested negative for HIV by antibody test. Dr. Bryson grew 75 million of her cells in a lab to look for any HIV. None. Aside from one blip in detectable HIV DNA at 14 weeks, researchers never found HIV in the patient again.
“Her cells are resistant to HIV now – both her own strains and laboratory strains,” Dr. Bryson told this news organization. “It’s been 14 months since then. She has no rebound and no detectable virus.”
The presentation drew as raucous as praise gets in a virtual environment. The comments began pouring in.
“Impressive results,” wrote Jim Hoxie, MD, professor emeritus at the University of Pennsylvania, Philadelphia.
“Exciting case,” wrote Allison Agwu, MD, a professor of pediatrics at Johns Hopkins University, Baltimore.
And Dennis Copertino, a research specialist at Weill Cornell Medicine, New York, wrote: “Thank you so much for translating this important cure strategy to people of color.”
Most donors with CCR5 mutations are White, Dr. Bryson said, suggesting that this approach, in a mixed-race woman, could expand the pool of people living with HIV and cancer who are good candidates for the approach.
But other observers had questions, ones that may require more research to answer. Some asked why this woman’s virus, after transplantation, wasn’t just immune to viruses with CCR5 but also another variant, called CXCR4, that one wouldn’t expect. Luis Montaner, DVM, director of the Immunopathogenesis Laboratory at the Wistar Institute in Philadelphia, wondered whether it was more than the blood that had cleared HIV. Did it get into the tissue, too? That question has not yet been answered.
For Carl Dieffenbach, PhD, director of the division of AIDS at the National Institute of Allergy and Infectious Diseases, the lack of GVH disease was a powerful and hopeful finding.
“There’s been this ongoing hypothesis that maybe graft-versus-host disease was needed at some level to help clear out every last single CD4+ T cell that may or may not have been harboring replication-competent virus,” Dr. Dieffenbach said in an interview. “But there was no GVH disease. That’s incredible. It’s a wonderful thing.”
Now the challenge is to move from a single case to making cure available to other people living with HIV.
The case also got cure researchers thinking.
Dr. Montaner called the case “an encouraging roadmap supporting anti-CCR5 strategies by CRISPR Cas9,” studies that are now underway.
Steven Deeks, MD, called the case “perhaps a model for how we might do this using a person’s own cells. Because we were never really going to be transplanting cells from another person as a scalable cure.”
For people living with HIV, particularly women of color, the results raise hopes and questions. Nina Martinez knows something about being a “first.” In 2019, she was the first American woman of color living with HIV to donate a kidney to another person living with the virus. To her, the excitement over the first woman of color being cured of HIV just shines a light on how very White and male HIV cure studies have been until now.
“For me, I’m not looking for a cure in which the successful step forward is me getting cancer,” she said in an interview. “I’m looking at, what’s going to be sustainable? I want to know what’s going to work for a group of people.”
Gina Marie Brown, a social worker living with HIV in New Orleans, is also thinking of groups of people.
“Every time we get a breakthrough, it’s like the sun is taken from behind the clouds a little more,” said Ms. Brown. “I think about people in the South, who bear a huge burden of HIV. I think about trans women. I think about Black women, and gay, bisexual, and same-gender-loving men. This could really impact HIV – in the same way that PrEP [pre-exposure prophylaxis] has, the same way that one pill once a day has.”
When Ms. Brown was diagnosed with HIV 22 years ago, she started to plan her funeral.
“That’s how much I thought HIV was a death sentence,” she told this news organization. “Oh my goodness! Glad you stuck around, Gina.”
The study was funded by the National Institutes of Health. Dr. Bryson, Dr. Dieffenbach, Dr. Deeks, and Dr. Montaner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a first, If she remains off treatment without any hint of HIV, she would be only the third person in the world – after the Berlin Patient and the London Patient – to be cured through a transplant.
“Her own virus could not infect her cells,” said Yvonne Bryson, MD, chief of pediatric infectious diseases at the University of California, Los Angeles, who presented the study at the Conference on Retroviruses and Opportunistic Infections, which both presenters and the audience attended remotely.
The middle-aged New York woman of mixed race, who has asked that her specific race and age not be shared to protect her privacy, was diagnosed with HIV in 2013 when she was still in the very early stages of infection. She started treatment immediately and quickly achieved an undetectable viral load. An undetectable viral load not only prevents someone from transmitting HIV to others but also reduces or eliminates HIV replication, which means fewer variants and less time for the virus to infiltrate cells where it can hide.
But in 2017, she was diagnosed with leukemia. As a last resort to cure her of the cancer, she received a combination of adult stem cells from a relative’s blood that closely matched her own and umbilical cord blood obtained from a cord blood bank. That particular sample of cord blood was selected for its genetic mutation against the CCR5 receptor on immune cells, CD4 T cells. That mutation makes the immune system resistant to HIV.
The two previous HIV cures, of Berlin Patient Timothy Ray Brown and London Patient Adam Castillejo, also used stem cell transplantation with a CCR5 mutation, but theirs were bone marrow transplants. Bone marrow transplants are more arduous than cord blood transplants, which are commonly used in pediatric cancer treatment.
In this case, the physicians treating her used both.
“This allows the adult cells to accelerate and grow up until the cord blood takes over,” said Dr. Bryson. During her presentation, Dr. Bryson pointed to two types of data: First, she presented data showing the level of HIV in the patient’s blood. Soon after HIV diagnosis and treatment, her viral load dropped to undetectable levels. She had a spike of virus when she received the transplant, but then it went back to undetectable and has stayed that way ever since.
Meanwhile, following the transplant, her immune system started rebuilding itself using the new, HIV-resistant cells provided in the transplant. As her care team watched, no graft-versus-host (GVH) disease, a common side effect of stem cell transplants, emerged. In fact, the transplant went so well that she was discharged early from the hospital.
One hundred days after the transplant, the immune system contained within the cord blood had taken over. Her CD4 immune cells returned to normal levels a little more than a year after the transplant. By 27 months, she decided to stop all HIV treatment to see if the transplant had worked.
This was the real test. But as Dr. Bryson and colleagues continued to watch her HIV viral load and her CD4 counts and search for infectious virus, they didn’t find any. She tested negative for HIV by antibody test. Dr. Bryson grew 75 million of her cells in a lab to look for any HIV. None. Aside from one blip in detectable HIV DNA at 14 weeks, researchers never found HIV in the patient again.
“Her cells are resistant to HIV now – both her own strains and laboratory strains,” Dr. Bryson told this news organization. “It’s been 14 months since then. She has no rebound and no detectable virus.”
The presentation drew as raucous as praise gets in a virtual environment. The comments began pouring in.
“Impressive results,” wrote Jim Hoxie, MD, professor emeritus at the University of Pennsylvania, Philadelphia.
“Exciting case,” wrote Allison Agwu, MD, a professor of pediatrics at Johns Hopkins University, Baltimore.
And Dennis Copertino, a research specialist at Weill Cornell Medicine, New York, wrote: “Thank you so much for translating this important cure strategy to people of color.”
Most donors with CCR5 mutations are White, Dr. Bryson said, suggesting that this approach, in a mixed-race woman, could expand the pool of people living with HIV and cancer who are good candidates for the approach.
But other observers had questions, ones that may require more research to answer. Some asked why this woman’s virus, after transplantation, wasn’t just immune to viruses with CCR5 but also another variant, called CXCR4, that one wouldn’t expect. Luis Montaner, DVM, director of the Immunopathogenesis Laboratory at the Wistar Institute in Philadelphia, wondered whether it was more than the blood that had cleared HIV. Did it get into the tissue, too? That question has not yet been answered.
For Carl Dieffenbach, PhD, director of the division of AIDS at the National Institute of Allergy and Infectious Diseases, the lack of GVH disease was a powerful and hopeful finding.
“There’s been this ongoing hypothesis that maybe graft-versus-host disease was needed at some level to help clear out every last single CD4+ T cell that may or may not have been harboring replication-competent virus,” Dr. Dieffenbach said in an interview. “But there was no GVH disease. That’s incredible. It’s a wonderful thing.”
Now the challenge is to move from a single case to making cure available to other people living with HIV.
The case also got cure researchers thinking.
Dr. Montaner called the case “an encouraging roadmap supporting anti-CCR5 strategies by CRISPR Cas9,” studies that are now underway.
Steven Deeks, MD, called the case “perhaps a model for how we might do this using a person’s own cells. Because we were never really going to be transplanting cells from another person as a scalable cure.”
For people living with HIV, particularly women of color, the results raise hopes and questions. Nina Martinez knows something about being a “first.” In 2019, she was the first American woman of color living with HIV to donate a kidney to another person living with the virus. To her, the excitement over the first woman of color being cured of HIV just shines a light on how very White and male HIV cure studies have been until now.
“For me, I’m not looking for a cure in which the successful step forward is me getting cancer,” she said in an interview. “I’m looking at, what’s going to be sustainable? I want to know what’s going to work for a group of people.”
Gina Marie Brown, a social worker living with HIV in New Orleans, is also thinking of groups of people.
“Every time we get a breakthrough, it’s like the sun is taken from behind the clouds a little more,” said Ms. Brown. “I think about people in the South, who bear a huge burden of HIV. I think about trans women. I think about Black women, and gay, bisexual, and same-gender-loving men. This could really impact HIV – in the same way that PrEP [pre-exposure prophylaxis] has, the same way that one pill once a day has.”
When Ms. Brown was diagnosed with HIV 22 years ago, she started to plan her funeral.
“That’s how much I thought HIV was a death sentence,” she told this news organization. “Oh my goodness! Glad you stuck around, Gina.”
The study was funded by the National Institutes of Health. Dr. Bryson, Dr. Dieffenbach, Dr. Deeks, and Dr. Montaner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a first, If she remains off treatment without any hint of HIV, she would be only the third person in the world – after the Berlin Patient and the London Patient – to be cured through a transplant.
“Her own virus could not infect her cells,” said Yvonne Bryson, MD, chief of pediatric infectious diseases at the University of California, Los Angeles, who presented the study at the Conference on Retroviruses and Opportunistic Infections, which both presenters and the audience attended remotely.
The middle-aged New York woman of mixed race, who has asked that her specific race and age not be shared to protect her privacy, was diagnosed with HIV in 2013 when she was still in the very early stages of infection. She started treatment immediately and quickly achieved an undetectable viral load. An undetectable viral load not only prevents someone from transmitting HIV to others but also reduces or eliminates HIV replication, which means fewer variants and less time for the virus to infiltrate cells where it can hide.
But in 2017, she was diagnosed with leukemia. As a last resort to cure her of the cancer, she received a combination of adult stem cells from a relative’s blood that closely matched her own and umbilical cord blood obtained from a cord blood bank. That particular sample of cord blood was selected for its genetic mutation against the CCR5 receptor on immune cells, CD4 T cells. That mutation makes the immune system resistant to HIV.
The two previous HIV cures, of Berlin Patient Timothy Ray Brown and London Patient Adam Castillejo, also used stem cell transplantation with a CCR5 mutation, but theirs were bone marrow transplants. Bone marrow transplants are more arduous than cord blood transplants, which are commonly used in pediatric cancer treatment.
In this case, the physicians treating her used both.
“This allows the adult cells to accelerate and grow up until the cord blood takes over,” said Dr. Bryson. During her presentation, Dr. Bryson pointed to two types of data: First, she presented data showing the level of HIV in the patient’s blood. Soon after HIV diagnosis and treatment, her viral load dropped to undetectable levels. She had a spike of virus when she received the transplant, but then it went back to undetectable and has stayed that way ever since.
Meanwhile, following the transplant, her immune system started rebuilding itself using the new, HIV-resistant cells provided in the transplant. As her care team watched, no graft-versus-host (GVH) disease, a common side effect of stem cell transplants, emerged. In fact, the transplant went so well that she was discharged early from the hospital.
One hundred days after the transplant, the immune system contained within the cord blood had taken over. Her CD4 immune cells returned to normal levels a little more than a year after the transplant. By 27 months, she decided to stop all HIV treatment to see if the transplant had worked.
This was the real test. But as Dr. Bryson and colleagues continued to watch her HIV viral load and her CD4 counts and search for infectious virus, they didn’t find any. She tested negative for HIV by antibody test. Dr. Bryson grew 75 million of her cells in a lab to look for any HIV. None. Aside from one blip in detectable HIV DNA at 14 weeks, researchers never found HIV in the patient again.
“Her cells are resistant to HIV now – both her own strains and laboratory strains,” Dr. Bryson told this news organization. “It’s been 14 months since then. She has no rebound and no detectable virus.”
The presentation drew as raucous as praise gets in a virtual environment. The comments began pouring in.
“Impressive results,” wrote Jim Hoxie, MD, professor emeritus at the University of Pennsylvania, Philadelphia.
“Exciting case,” wrote Allison Agwu, MD, a professor of pediatrics at Johns Hopkins University, Baltimore.
And Dennis Copertino, a research specialist at Weill Cornell Medicine, New York, wrote: “Thank you so much for translating this important cure strategy to people of color.”
Most donors with CCR5 mutations are White, Dr. Bryson said, suggesting that this approach, in a mixed-race woman, could expand the pool of people living with HIV and cancer who are good candidates for the approach.
But other observers had questions, ones that may require more research to answer. Some asked why this woman’s virus, after transplantation, wasn’t just immune to viruses with CCR5 but also another variant, called CXCR4, that one wouldn’t expect. Luis Montaner, DVM, director of the Immunopathogenesis Laboratory at the Wistar Institute in Philadelphia, wondered whether it was more than the blood that had cleared HIV. Did it get into the tissue, too? That question has not yet been answered.
For Carl Dieffenbach, PhD, director of the division of AIDS at the National Institute of Allergy and Infectious Diseases, the lack of GVH disease was a powerful and hopeful finding.
“There’s been this ongoing hypothesis that maybe graft-versus-host disease was needed at some level to help clear out every last single CD4+ T cell that may or may not have been harboring replication-competent virus,” Dr. Dieffenbach said in an interview. “But there was no GVH disease. That’s incredible. It’s a wonderful thing.”
Now the challenge is to move from a single case to making cure available to other people living with HIV.
The case also got cure researchers thinking.
Dr. Montaner called the case “an encouraging roadmap supporting anti-CCR5 strategies by CRISPR Cas9,” studies that are now underway.
Steven Deeks, MD, called the case “perhaps a model for how we might do this using a person’s own cells. Because we were never really going to be transplanting cells from another person as a scalable cure.”
For people living with HIV, particularly women of color, the results raise hopes and questions. Nina Martinez knows something about being a “first.” In 2019, she was the first American woman of color living with HIV to donate a kidney to another person living with the virus. To her, the excitement over the first woman of color being cured of HIV just shines a light on how very White and male HIV cure studies have been until now.
“For me, I’m not looking for a cure in which the successful step forward is me getting cancer,” she said in an interview. “I’m looking at, what’s going to be sustainable? I want to know what’s going to work for a group of people.”
Gina Marie Brown, a social worker living with HIV in New Orleans, is also thinking of groups of people.
“Every time we get a breakthrough, it’s like the sun is taken from behind the clouds a little more,” said Ms. Brown. “I think about people in the South, who bear a huge burden of HIV. I think about trans women. I think about Black women, and gay, bisexual, and same-gender-loving men. This could really impact HIV – in the same way that PrEP [pre-exposure prophylaxis] has, the same way that one pill once a day has.”
When Ms. Brown was diagnosed with HIV 22 years ago, she started to plan her funeral.
“That’s how much I thought HIV was a death sentence,” she told this news organization. “Oh my goodness! Glad you stuck around, Gina.”
The study was funded by the National Institutes of Health. Dr. Bryson, Dr. Dieffenbach, Dr. Deeks, and Dr. Montaner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CROI 2022
Tips for connecting with your patients
It is a tough time to be a doctor. With the stresses of the pandemic, the continued unfettered rise of insurance company BS, and so many medical groups being bought up that we often don’t even know who makes the decisions, the patient can sometimes be hidden in the equation.
Be curious
When physicians are curious about why patients have symptoms, how those symptoms will affect their lives, and how worried the patient is about them, patients feel cared about.
Ascertaining how concerned patients are about their symptoms will help you make decisions on whether symptoms you are not concerned about actually need to be treated.
Limit use of EHRs when possible
Use of the electronic health record during visits is essential, but focusing on it too much can put a barrier between the physician and the patient.
Marmor and colleagues found there is an inverse relationship between time spent on the EHR by a patient’s physician and the patient’s satisfaction.1
Eye contact with the patient is important, especially when patients are sharing concerns they are scared about and upsetting experiences. There can be awkward pauses when looking things up on the EHR. Fill those pauses by explaining to the patient what you are doing, or chatting with the patient.
Consider teaching medical students
When a medical student works with you, it doubles the time the patient gets with a concerned listener. Students also can do a great job with timely follow-up and checking in with worried patients.
By having the student present in the clinic room, with the patient present, the patient can really feel heard. The student shares all the details the patient shared, and now their physician is hearing an organized, thoughtful report of the patients concerns.
In fact, I was involved in a study that showed that patients preferred in room presentations, and that they were more satisfied when students presented in the room.2
Use healing words
Some words carry loaded emotions. The word chronic, for example, has negative connotations, whereas the term persisting does not.
I will often ask patients how long they have been suffering from a symptom to imply my concern for what they are going through. The term “chief complaint” is outdated, and upsets patients when they see it in their medical record.
As a patient of mine once said to me: “I never complained about that problem, I just brought it to your attention.” No one wants to be seen as a complainer. Substituting the word concern for complaint works well.
Explain as you examine
People love to hear the term normal. When you are examining a patient, let them know when findings are normal.
I also find it helpful to explain to patients why I am doing certain physical exam maneuvers. This helps them assess how thorough we are in our thought process.
When patients feel their physicians are thorough, they have more confidence in them.
In summary
- Be curious.
- Do not overly focus on the EHR.
- Consider teaching a medical student.
- Be careful of word choice.
- “Overexplain” the physical exam.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Marmor RA et al. Appl Clin Inform. 2018 Jan;9(1):11-4.
2. Rogers HD et al. Acad Med. 2003 Sep;78(9):945-9.
It is a tough time to be a doctor. With the stresses of the pandemic, the continued unfettered rise of insurance company BS, and so many medical groups being bought up that we often don’t even know who makes the decisions, the patient can sometimes be hidden in the equation.
Be curious
When physicians are curious about why patients have symptoms, how those symptoms will affect their lives, and how worried the patient is about them, patients feel cared about.
Ascertaining how concerned patients are about their symptoms will help you make decisions on whether symptoms you are not concerned about actually need to be treated.
Limit use of EHRs when possible
Use of the electronic health record during visits is essential, but focusing on it too much can put a barrier between the physician and the patient.
Marmor and colleagues found there is an inverse relationship between time spent on the EHR by a patient’s physician and the patient’s satisfaction.1
Eye contact with the patient is important, especially when patients are sharing concerns they are scared about and upsetting experiences. There can be awkward pauses when looking things up on the EHR. Fill those pauses by explaining to the patient what you are doing, or chatting with the patient.
Consider teaching medical students
When a medical student works with you, it doubles the time the patient gets with a concerned listener. Students also can do a great job with timely follow-up and checking in with worried patients.
By having the student present in the clinic room, with the patient present, the patient can really feel heard. The student shares all the details the patient shared, and now their physician is hearing an organized, thoughtful report of the patients concerns.
In fact, I was involved in a study that showed that patients preferred in room presentations, and that they were more satisfied when students presented in the room.2
Use healing words
Some words carry loaded emotions. The word chronic, for example, has negative connotations, whereas the term persisting does not.
I will often ask patients how long they have been suffering from a symptom to imply my concern for what they are going through. The term “chief complaint” is outdated, and upsets patients when they see it in their medical record.
As a patient of mine once said to me: “I never complained about that problem, I just brought it to your attention.” No one wants to be seen as a complainer. Substituting the word concern for complaint works well.
Explain as you examine
People love to hear the term normal. When you are examining a patient, let them know when findings are normal.
I also find it helpful to explain to patients why I am doing certain physical exam maneuvers. This helps them assess how thorough we are in our thought process.
When patients feel their physicians are thorough, they have more confidence in them.
In summary
- Be curious.
- Do not overly focus on the EHR.
- Consider teaching a medical student.
- Be careful of word choice.
- “Overexplain” the physical exam.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Marmor RA et al. Appl Clin Inform. 2018 Jan;9(1):11-4.
2. Rogers HD et al. Acad Med. 2003 Sep;78(9):945-9.
It is a tough time to be a doctor. With the stresses of the pandemic, the continued unfettered rise of insurance company BS, and so many medical groups being bought up that we often don’t even know who makes the decisions, the patient can sometimes be hidden in the equation.
Be curious
When physicians are curious about why patients have symptoms, how those symptoms will affect their lives, and how worried the patient is about them, patients feel cared about.
Ascertaining how concerned patients are about their symptoms will help you make decisions on whether symptoms you are not concerned about actually need to be treated.
Limit use of EHRs when possible
Use of the electronic health record during visits is essential, but focusing on it too much can put a barrier between the physician and the patient.
Marmor and colleagues found there is an inverse relationship between time spent on the EHR by a patient’s physician and the patient’s satisfaction.1
Eye contact with the patient is important, especially when patients are sharing concerns they are scared about and upsetting experiences. There can be awkward pauses when looking things up on the EHR. Fill those pauses by explaining to the patient what you are doing, or chatting with the patient.
Consider teaching medical students
When a medical student works with you, it doubles the time the patient gets with a concerned listener. Students also can do a great job with timely follow-up and checking in with worried patients.
By having the student present in the clinic room, with the patient present, the patient can really feel heard. The student shares all the details the patient shared, and now their physician is hearing an organized, thoughtful report of the patients concerns.
In fact, I was involved in a study that showed that patients preferred in room presentations, and that they were more satisfied when students presented in the room.2
Use healing words
Some words carry loaded emotions. The word chronic, for example, has negative connotations, whereas the term persisting does not.
I will often ask patients how long they have been suffering from a symptom to imply my concern for what they are going through. The term “chief complaint” is outdated, and upsets patients when they see it in their medical record.
As a patient of mine once said to me: “I never complained about that problem, I just brought it to your attention.” No one wants to be seen as a complainer. Substituting the word concern for complaint works well.
Explain as you examine
People love to hear the term normal. When you are examining a patient, let them know when findings are normal.
I also find it helpful to explain to patients why I am doing certain physical exam maneuvers. This helps them assess how thorough we are in our thought process.
When patients feel their physicians are thorough, they have more confidence in them.
In summary
- Be curious.
- Do not overly focus on the EHR.
- Consider teaching a medical student.
- Be careful of word choice.
- “Overexplain” the physical exam.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Marmor RA et al. Appl Clin Inform. 2018 Jan;9(1):11-4.
2. Rogers HD et al. Acad Med. 2003 Sep;78(9):945-9.
Children and COVID: Weekly cases down by more than half
A third consecutive week of declines in new COVID-19 cases among children has brought the weekly count down by 74% since the Omicron surge peaked in mid-January, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
and by 74% from the peak of 1.15 million cases recorded for the week of Jan. 14-20, the AAP and CHA said in their weekly COVID report. They also noted that the weekly tally was still higher than anything seen during the Delta surge.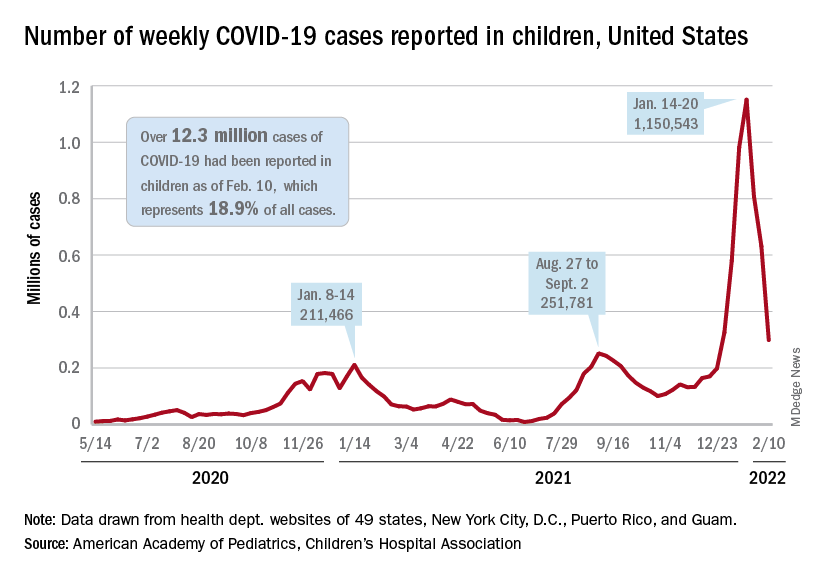
The total number of pediatric cases was over 12.3 million as of Feb. 10, with children representing 18.9% of cases in all ages, according to the AAP/CHA report. The Centers for Disease Control and Prevention puts the two measures at 10.4 million and 17.3% on its COVID Data Tracker, based on availability of age data for 59.6 million total cases as of Feb. 14. The CDC also reported that 1,282 children have died from COVID-19 so far, which is about 0.17% of all deaths with age data available.
The AAP and CHA have been collecting data from state and territorial health departments, which have not always been consistently available over the course of the pandemic. Also, the CDC defines children as those under age 18 years, but that upper boundary varies from 14 to 20 among the states.
The decline of the Omicron variant also can be seen in new admissions of children with confirmed COVID-19, which continued to drop. The 7-day average of 435 admissions per day for the week of Feb. 6-12 was less than half of the peak seen in mid-January, when it reached 914 per day. The daily admission rate on Feb. 12 was 0.60 per 100,000 children aged 0-17 years – again, less than half the peak rate of 1.25 reported on Jan. 16, CDC data show.
The fading threat of Omicron also seems to be reflected in recent vaccination trends. Both initial doses and completions declined for the fourth consecutive week (Feb. 3-9) among children aged 5-11 years, while initiations held steady for 12- to 17-year-olds but completions declined for the third straight week, the AAP said in its separate vaccination report, which is based on data from the CDC.
As of Feb. 14, almost 32% of children aged 5-11 – that’s almost 9.2 million individuals – had received at least one dose of the COVID-19 vaccine and just over 24% (6.9 million) were fully vaccinated, the CDC reported. For children aged 12-17, the corresponding figures are 67% (16.9 million) and 57% (14.4 million). Newly available data from the CDC also indicate that 19.5% (2.8 million) of children aged 12-17 have received a booster dose.
A third consecutive week of declines in new COVID-19 cases among children has brought the weekly count down by 74% since the Omicron surge peaked in mid-January, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
and by 74% from the peak of 1.15 million cases recorded for the week of Jan. 14-20, the AAP and CHA said in their weekly COVID report. They also noted that the weekly tally was still higher than anything seen during the Delta surge.
The total number of pediatric cases was over 12.3 million as of Feb. 10, with children representing 18.9% of cases in all ages, according to the AAP/CHA report. The Centers for Disease Control and Prevention puts the two measures at 10.4 million and 17.3% on its COVID Data Tracker, based on availability of age data for 59.6 million total cases as of Feb. 14. The CDC also reported that 1,282 children have died from COVID-19 so far, which is about 0.17% of all deaths with age data available.
The AAP and CHA have been collecting data from state and territorial health departments, which have not always been consistently available over the course of the pandemic. Also, the CDC defines children as those under age 18 years, but that upper boundary varies from 14 to 20 among the states.
The decline of the Omicron variant also can be seen in new admissions of children with confirmed COVID-19, which continued to drop. The 7-day average of 435 admissions per day for the week of Feb. 6-12 was less than half of the peak seen in mid-January, when it reached 914 per day. The daily admission rate on Feb. 12 was 0.60 per 100,000 children aged 0-17 years – again, less than half the peak rate of 1.25 reported on Jan. 16, CDC data show.
The fading threat of Omicron also seems to be reflected in recent vaccination trends. Both initial doses and completions declined for the fourth consecutive week (Feb. 3-9) among children aged 5-11 years, while initiations held steady for 12- to 17-year-olds but completions declined for the third straight week, the AAP said in its separate vaccination report, which is based on data from the CDC.
As of Feb. 14, almost 32% of children aged 5-11 – that’s almost 9.2 million individuals – had received at least one dose of the COVID-19 vaccine and just over 24% (6.9 million) were fully vaccinated, the CDC reported. For children aged 12-17, the corresponding figures are 67% (16.9 million) and 57% (14.4 million). Newly available data from the CDC also indicate that 19.5% (2.8 million) of children aged 12-17 have received a booster dose.
A third consecutive week of declines in new COVID-19 cases among children has brought the weekly count down by 74% since the Omicron surge peaked in mid-January, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
and by 74% from the peak of 1.15 million cases recorded for the week of Jan. 14-20, the AAP and CHA said in their weekly COVID report. They also noted that the weekly tally was still higher than anything seen during the Delta surge.
The total number of pediatric cases was over 12.3 million as of Feb. 10, with children representing 18.9% of cases in all ages, according to the AAP/CHA report. The Centers for Disease Control and Prevention puts the two measures at 10.4 million and 17.3% on its COVID Data Tracker, based on availability of age data for 59.6 million total cases as of Feb. 14. The CDC also reported that 1,282 children have died from COVID-19 so far, which is about 0.17% of all deaths with age data available.
The AAP and CHA have been collecting data from state and territorial health departments, which have not always been consistently available over the course of the pandemic. Also, the CDC defines children as those under age 18 years, but that upper boundary varies from 14 to 20 among the states.
The decline of the Omicron variant also can be seen in new admissions of children with confirmed COVID-19, which continued to drop. The 7-day average of 435 admissions per day for the week of Feb. 6-12 was less than half of the peak seen in mid-January, when it reached 914 per day. The daily admission rate on Feb. 12 was 0.60 per 100,000 children aged 0-17 years – again, less than half the peak rate of 1.25 reported on Jan. 16, CDC data show.
The fading threat of Omicron also seems to be reflected in recent vaccination trends. Both initial doses and completions declined for the fourth consecutive week (Feb. 3-9) among children aged 5-11 years, while initiations held steady for 12- to 17-year-olds but completions declined for the third straight week, the AAP said in its separate vaccination report, which is based on data from the CDC.
As of Feb. 14, almost 32% of children aged 5-11 – that’s almost 9.2 million individuals – had received at least one dose of the COVID-19 vaccine and just over 24% (6.9 million) were fully vaccinated, the CDC reported. For children aged 12-17, the corresponding figures are 67% (16.9 million) and 57% (14.4 million). Newly available data from the CDC also indicate that 19.5% (2.8 million) of children aged 12-17 have received a booster dose.
Long COVID symptoms linked to effects on vagus nerve
Several long COVID symptoms could be linked to the effects of the coronavirus on a vital central nerve, according to new research being released in the spring.
The vagus nerve, which runs from the brain into the body, connects to the heart, lungs, intestines, and several muscles involved with swallowing. It plays a role in several body functions that control heart rate, speech, the gag reflex, sweating, and digestion.
Those with long COVID and vagus nerve problems could face long-term issues with their voice, a hard time swallowing, dizziness, a high heart rate, low blood pressure, and diarrhea, the study authors found.
Their findings will be presented at the 2022 European Congress of Clinical Microbiology and Infectious Diseases in late April.
“Most long COVID subjects with vagus nerve dysfunction symptoms had a range of significant, clinically relevant, structural and/or functional alterations in their vagus nerve, including nerve thickening, trouble swallowing, and symptoms of impaired breathing,” the study authors wrote. “Our findings so far thus point at vagus nerve dysfunction as a central pathophysiological feature of long COVID.”
Researchers from the University Hospital Germans Trias i Pujol in Barcelona performed a study to look at vagus nerve functioning in long COVID patients. Among 348 patients, about 66% had at least one symptom that suggested vagus nerve dysfunction. The researchers did a broad evaluation with imaging and functional tests for 22 patients in the university’s Long COVID Clinic from March to June 2021.
Of the 22 patients, 20 were women, and the median age was 44. The most frequent symptoms related to vagus nerve dysfunction were diarrhea (73%), high heart rates (59%), dizziness (45%), swallowing problems (45%), voice problems (45%), and low blood pressure (14%).
Almost all (19 of 22 patients) had three or more symptoms related to vagus nerve dysfunction. The average length of symptoms was 14 months.
Of 22 patients, 6 had a change in the vagus nerve in the neck, which the researchers observed by ultrasound. They had a thickening of the vagus nerve and increased “echogenicity,” which suggests inflammation.
What’s more, 10 of 22 patients had flattened “diaphragmatic curves” during a thoracic ultrasound, which means the diaphragm doesn’t move as well as it should during breathing, and abnormal breathing. In another assessment, 10 of 16 patients had lower maximum inspiration pressures, suggesting a weakness in breathing muscles.
Eating and digestion were also impaired in some patients, with 13 reporting trouble with swallowing. During a gastric and bowel function assessment, eight patients couldn’t move food from the esophagus to the stomach as well as they should, while nine patients had acid reflux. Three patients had a hiatal hernia, which happens when the upper part of the stomach bulges through the diaphragm into the chest cavity.
The voices of some patients changed as well. Eight patients had an abnormal voice handicap index 30 test, which is a standard way to measure voice function. Among those, seven patients had dysphonia, or persistent voice problems.
The study is ongoing, and the research team is continuing to recruit patients to study the links between long COVID and the vagus nerve. The full paper isn’t yet available, and the research hasn’t yet been peer reviewed.
“The study appears to add to a growing collection of data suggesting at least some of the symptoms of long COVID is mediated through a direct impact on the nervous system,” David Strain, MD, a clinical senior lecturer at the University of Exeter (England), told the Science Media Centre.
“Establishing vagal nerve damage is useful information, as there are recognized, albeit not perfect, treatments for other causes of vagal nerve dysfunction that may be extrapolated to be beneficial for people with this type of long COVID,” he said.
A version of this article first appeared on WebMD.com.
Several long COVID symptoms could be linked to the effects of the coronavirus on a vital central nerve, according to new research being released in the spring.
The vagus nerve, which runs from the brain into the body, connects to the heart, lungs, intestines, and several muscles involved with swallowing. It plays a role in several body functions that control heart rate, speech, the gag reflex, sweating, and digestion.
Those with long COVID and vagus nerve problems could face long-term issues with their voice, a hard time swallowing, dizziness, a high heart rate, low blood pressure, and diarrhea, the study authors found.
Their findings will be presented at the 2022 European Congress of Clinical Microbiology and Infectious Diseases in late April.
“Most long COVID subjects with vagus nerve dysfunction symptoms had a range of significant, clinically relevant, structural and/or functional alterations in their vagus nerve, including nerve thickening, trouble swallowing, and symptoms of impaired breathing,” the study authors wrote. “Our findings so far thus point at vagus nerve dysfunction as a central pathophysiological feature of long COVID.”
Researchers from the University Hospital Germans Trias i Pujol in Barcelona performed a study to look at vagus nerve functioning in long COVID patients. Among 348 patients, about 66% had at least one symptom that suggested vagus nerve dysfunction. The researchers did a broad evaluation with imaging and functional tests for 22 patients in the university’s Long COVID Clinic from March to June 2021.
Of the 22 patients, 20 were women, and the median age was 44. The most frequent symptoms related to vagus nerve dysfunction were diarrhea (73%), high heart rates (59%), dizziness (45%), swallowing problems (45%), voice problems (45%), and low blood pressure (14%).
Almost all (19 of 22 patients) had three or more symptoms related to vagus nerve dysfunction. The average length of symptoms was 14 months.
Of 22 patients, 6 had a change in the vagus nerve in the neck, which the researchers observed by ultrasound. They had a thickening of the vagus nerve and increased “echogenicity,” which suggests inflammation.
What’s more, 10 of 22 patients had flattened “diaphragmatic curves” during a thoracic ultrasound, which means the diaphragm doesn’t move as well as it should during breathing, and abnormal breathing. In another assessment, 10 of 16 patients had lower maximum inspiration pressures, suggesting a weakness in breathing muscles.
Eating and digestion were also impaired in some patients, with 13 reporting trouble with swallowing. During a gastric and bowel function assessment, eight patients couldn’t move food from the esophagus to the stomach as well as they should, while nine patients had acid reflux. Three patients had a hiatal hernia, which happens when the upper part of the stomach bulges through the diaphragm into the chest cavity.
The voices of some patients changed as well. Eight patients had an abnormal voice handicap index 30 test, which is a standard way to measure voice function. Among those, seven patients had dysphonia, or persistent voice problems.
The study is ongoing, and the research team is continuing to recruit patients to study the links between long COVID and the vagus nerve. The full paper isn’t yet available, and the research hasn’t yet been peer reviewed.
“The study appears to add to a growing collection of data suggesting at least some of the symptoms of long COVID is mediated through a direct impact on the nervous system,” David Strain, MD, a clinical senior lecturer at the University of Exeter (England), told the Science Media Centre.
“Establishing vagal nerve damage is useful information, as there are recognized, albeit not perfect, treatments for other causes of vagal nerve dysfunction that may be extrapolated to be beneficial for people with this type of long COVID,” he said.
A version of this article first appeared on WebMD.com.
Several long COVID symptoms could be linked to the effects of the coronavirus on a vital central nerve, according to new research being released in the spring.
The vagus nerve, which runs from the brain into the body, connects to the heart, lungs, intestines, and several muscles involved with swallowing. It plays a role in several body functions that control heart rate, speech, the gag reflex, sweating, and digestion.
Those with long COVID and vagus nerve problems could face long-term issues with their voice, a hard time swallowing, dizziness, a high heart rate, low blood pressure, and diarrhea, the study authors found.
Their findings will be presented at the 2022 European Congress of Clinical Microbiology and Infectious Diseases in late April.
“Most long COVID subjects with vagus nerve dysfunction symptoms had a range of significant, clinically relevant, structural and/or functional alterations in their vagus nerve, including nerve thickening, trouble swallowing, and symptoms of impaired breathing,” the study authors wrote. “Our findings so far thus point at vagus nerve dysfunction as a central pathophysiological feature of long COVID.”
Researchers from the University Hospital Germans Trias i Pujol in Barcelona performed a study to look at vagus nerve functioning in long COVID patients. Among 348 patients, about 66% had at least one symptom that suggested vagus nerve dysfunction. The researchers did a broad evaluation with imaging and functional tests for 22 patients in the university’s Long COVID Clinic from March to June 2021.
Of the 22 patients, 20 were women, and the median age was 44. The most frequent symptoms related to vagus nerve dysfunction were diarrhea (73%), high heart rates (59%), dizziness (45%), swallowing problems (45%), voice problems (45%), and low blood pressure (14%).
Almost all (19 of 22 patients) had three or more symptoms related to vagus nerve dysfunction. The average length of symptoms was 14 months.
Of 22 patients, 6 had a change in the vagus nerve in the neck, which the researchers observed by ultrasound. They had a thickening of the vagus nerve and increased “echogenicity,” which suggests inflammation.
What’s more, 10 of 22 patients had flattened “diaphragmatic curves” during a thoracic ultrasound, which means the diaphragm doesn’t move as well as it should during breathing, and abnormal breathing. In another assessment, 10 of 16 patients had lower maximum inspiration pressures, suggesting a weakness in breathing muscles.
Eating and digestion were also impaired in some patients, with 13 reporting trouble with swallowing. During a gastric and bowel function assessment, eight patients couldn’t move food from the esophagus to the stomach as well as they should, while nine patients had acid reflux. Three patients had a hiatal hernia, which happens when the upper part of the stomach bulges through the diaphragm into the chest cavity.
The voices of some patients changed as well. Eight patients had an abnormal voice handicap index 30 test, which is a standard way to measure voice function. Among those, seven patients had dysphonia, or persistent voice problems.
The study is ongoing, and the research team is continuing to recruit patients to study the links between long COVID and the vagus nerve. The full paper isn’t yet available, and the research hasn’t yet been peer reviewed.
“The study appears to add to a growing collection of data suggesting at least some of the symptoms of long COVID is mediated through a direct impact on the nervous system,” David Strain, MD, a clinical senior lecturer at the University of Exeter (England), told the Science Media Centre.
“Establishing vagal nerve damage is useful information, as there are recognized, albeit not perfect, treatments for other causes of vagal nerve dysfunction that may be extrapolated to be beneficial for people with this type of long COVID,” he said.
A version of this article first appeared on WebMD.com.
Ear tubes not recommended for recurrent AOM without effusion, ENTs maintain
A practice guideline update from the ENT community on tympanostomy tubes in children reaffirms that tube insertion should not be considered in cases of otitis media with effusion (OME) lasting less than 3 months, or in children with recurrent acute otitis media (AOM) without middle ear effusion at the time of assessment for the procedure.
New in the update from the American Academy of Otolaryngology–Head and Neck Surgery Foundation (AAO-HNSF) is a strong recommendation for timely follow-up after surgery and recommendations against both routine use of prophylactic antibiotic ear drops after surgery and the initial use of long-term tubes except when there are specific reasons for doing so.
The update also expands the list of risk factors that place children with OME at increased risk of developmental difficulties – and often in need of timely ear tube placement – to include intellectual disability, learning disorder, and attention-deficit/hyperactivity disorder.
“Most of what we said in the 2013 [original] guideline was good and still valid ... and [important for] pediatricians, who are the key players” in managing otitis media, Jesse Hackell, MD, one of two general pediatricians who served on the Academy’s guideline update committee, said in an interview.
OME spontaneously clears up to 90% of the time within 3 months, said Dr. Hackell, of Pomona (New York) Pediatrics, and chair of the American Academy of Pediatrics (AAP) Committee on Practice and Ambulatory Medicine.
The updated guideline, for children 6 months to 12 years, reaffirms a recommendation that tube insertion be offered to children with “bilateral OME for 3 months or longer AND documented hearing difficulties.”
It also reaffirms “options” (a lesser quality of evidence) that in the absence of hearing difficulties, surgery may be performed for children with chronic OME (3 months or longer) in one or both ears if 1) they are at increased risk of developmental difficulties from OME or 2) effusion is likely contributing to balance problems, poor school performance, behavioral problems, ear discomfort, or reduced quality of life.
Children with chronic OME who do not undergo surgery should be reevaluated at 3- to 6-month intervals and monitored until effusion is no longer present, significant hearing loss is detected, or structural abnormalities of the tympanic membrane or middle ear are detected, the update again recommends.
Tympanostomy tube placement is the most common ambulatory surgery performed on children in the United States, the guideline authors say. In 2014, about 9% of children had undergone the surgery, they wrote, noting also that “tubes were placed in 25%-30% of children with frequent ear infections.”
Recurrent AOM
The AAO-HNSF guidance regarding tympanostomy tubes for OME is similar overall to management guidance issued by the AAP in its clinical practice guideline on OME.
The organizations differ, however, on their guidance for tube insertion for recurrent AOM. In its 2013 clinical practice guideline on AOM, the AAP recommends that clinicians may offer tube insertion for recurrent AOM, with no mention of the presence or absence of persistent fluid as a consideration.
According to the AAO-HNSF update, grade A evidence, including some research published since its original 2013 guideline, has shown little benefit to tube insertion in reducing the incidence of AOM in otherwise healthy children who don’t have middle ear effusion.
One study published in 2019 assessed outcomes after watchful waiting and found that only one-third of 123 children eventually went on to tympanostomy tube placement, noted Richard M. Rosenfeld, MD, distinguished professor and chairman of otolaryngology at SUNY Downstate Health Sciences University in Brooklyn, N.Y., and lead author of the original and updated guidelines.
In practice, “the real question [for the ENT] is the future. If the ears are perfectly clear, will tubes really reduce the frequency of infections going forward?” Dr. Rosenfeld said in an interview. “All the evidence seems to say no, it doesn’t make much of a difference.”
Dr. Hackell said he’s confident that the question “is settled enough.” While there “could be stronger research and higher quality studies, the evidence is still pretty good to suggest you gain little to no benefit with tubes when you’re dealing with recurrent AOM without effusion,” he said.
Asked to comment on the ENT update and its guidance on tympanostomy tubes for children with recurrent AOM, an AAP spokesperson said the “issue is under review” and that the AAP did not currently have a statement.
At-risk children
The AAO-HNSF update renews a recommendation to evaluate children with either recurrent AOM or OME of any duration for increased risk for speech, language, or learning problems from OME because of baseline factors (sensory, physical, cognitive, or behavioral).
When OME becomes chronic – or when a tympanogram gives a flat-line reading – OME is likely to persist, and families of at-risk children especially should be encouraged to pursue tube placement, Dr. Rosenfeld said.
Despite prior guidance to this effect, he said, ear tubes are being underutilized in at-risk children, with effusion being missed in primary care and with ENTs not expediting tube placement upon referral.
“These children have learning issues, cognitive issues, developmental issues,” he said in the interview. “It’s a population that does very poorly with ears full of fluid ... and despite guidance suggesting these children should be prioritized with tubes, it doesn’t seem to be happening enough.”
Formulating guidelines for at-risk children is challenging because they are often excluded from trials, Dr. Rosenfeld said, which limits evidence about the benefits of tubes and limits the strength of recommendations.
The addition of attention-deficit/hyperactivity disorder, intellectual disability, and learning disorder to the list of risk factors is notable, Dr. Hackell said. (The list includes autism spectrum disorder, developmental delay, and suspected or confirmed speech and language delay or disorder.)
“We know that kids with ADHD take in and process information a little differently ... it may be harder to get their attention with auditory stimulation,” he said. “So anything that would impact the taking in of information even for a short period of time increases their risk.”
Surgical practice
ENTs are advised in the new guidance to use long-term tubes and perioperative antibiotic ear drops more judiciously. “Long-term tubes have a role, but there are some doctors who routinely use them, even for a first-time surgery,” said Dr. Rosenfeld.
Overuse of long-term tubes results in a higher incidence of tympanic membrane perforation, chronic drainage, and other complications, as well as greater need for long-term follow-up. “There needs to be a reason – something to justify the need for prolonged ventilation,” he said.
Perioperative antibiotic ear drops are often administered during surgery and then prescribed routinely for all children afterward, but research has shown that saline irrigation during surgery and a single application of antibiotic/steroid drops is similarly efficacious in preventing otorrhea, the guideline says. Antibiotic ear drops are also “expensive,” noted Dr. Hackell. “There’s not enough benefit to justify it.”
The update also more explicitly advises selective use of adenoidectomy. A new option says that clinicians may perform the procedure as an adjunct to tube insertion for children 4 years or older to potentially reduce the future incidence of recurrent OME or the need for repeat surgery.
However, in younger children, it should not be offered unless there are symptoms directly related to adenoid infection or nasal obstruction. “Under 4 years, there’s no primary benefit for the ears,” said Dr. Rosenfeld.
Follow-up with the surgeon after tympanostomy tube insertion should occur within 3 months to assess outcomes and educate the family, the update strongly recommends.
And pediatricians should know, Dr. Hackell notes, that clinical evidence continues to show that earplugs and other water precautions are not routinely needed for children who have tubes in place. A good approach, the guideline says, is to “first avoid water precautions and instead reserve them for children with recurrent or persistent tympanostomy tube otorrhea.”
Asked to comment on the guideline update, Tim Joos, MD, MPH, who practices combined internal medicine/pediatrics in Seattle and is an editorial advisory board member of Pediatric News, noted the inclusion of patient information sheets with frequently asked questions – resources that can be useful for guiding parents through what’s often a shared decision-making process.
Neither Dr. Rosenfeld nor Dr. Hackell reported any disclosures. Other members of the guideline update committee reported various book royalties, consulting fees, and other disclosures. Dr. Joos reported he has no connections to the guideline authors.
A practice guideline update from the ENT community on tympanostomy tubes in children reaffirms that tube insertion should not be considered in cases of otitis media with effusion (OME) lasting less than 3 months, or in children with recurrent acute otitis media (AOM) without middle ear effusion at the time of assessment for the procedure.
New in the update from the American Academy of Otolaryngology–Head and Neck Surgery Foundation (AAO-HNSF) is a strong recommendation for timely follow-up after surgery and recommendations against both routine use of prophylactic antibiotic ear drops after surgery and the initial use of long-term tubes except when there are specific reasons for doing so.
The update also expands the list of risk factors that place children with OME at increased risk of developmental difficulties – and often in need of timely ear tube placement – to include intellectual disability, learning disorder, and attention-deficit/hyperactivity disorder.
“Most of what we said in the 2013 [original] guideline was good and still valid ... and [important for] pediatricians, who are the key players” in managing otitis media, Jesse Hackell, MD, one of two general pediatricians who served on the Academy’s guideline update committee, said in an interview.
OME spontaneously clears up to 90% of the time within 3 months, said Dr. Hackell, of Pomona (New York) Pediatrics, and chair of the American Academy of Pediatrics (AAP) Committee on Practice and Ambulatory Medicine.
The updated guideline, for children 6 months to 12 years, reaffirms a recommendation that tube insertion be offered to children with “bilateral OME for 3 months or longer AND documented hearing difficulties.”
It also reaffirms “options” (a lesser quality of evidence) that in the absence of hearing difficulties, surgery may be performed for children with chronic OME (3 months or longer) in one or both ears if 1) they are at increased risk of developmental difficulties from OME or 2) effusion is likely contributing to balance problems, poor school performance, behavioral problems, ear discomfort, or reduced quality of life.
Children with chronic OME who do not undergo surgery should be reevaluated at 3- to 6-month intervals and monitored until effusion is no longer present, significant hearing loss is detected, or structural abnormalities of the tympanic membrane or middle ear are detected, the update again recommends.
Tympanostomy tube placement is the most common ambulatory surgery performed on children in the United States, the guideline authors say. In 2014, about 9% of children had undergone the surgery, they wrote, noting also that “tubes were placed in 25%-30% of children with frequent ear infections.”
Recurrent AOM
The AAO-HNSF guidance regarding tympanostomy tubes for OME is similar overall to management guidance issued by the AAP in its clinical practice guideline on OME.
The organizations differ, however, on their guidance for tube insertion for recurrent AOM. In its 2013 clinical practice guideline on AOM, the AAP recommends that clinicians may offer tube insertion for recurrent AOM, with no mention of the presence or absence of persistent fluid as a consideration.
According to the AAO-HNSF update, grade A evidence, including some research published since its original 2013 guideline, has shown little benefit to tube insertion in reducing the incidence of AOM in otherwise healthy children who don’t have middle ear effusion.
One study published in 2019 assessed outcomes after watchful waiting and found that only one-third of 123 children eventually went on to tympanostomy tube placement, noted Richard M. Rosenfeld, MD, distinguished professor and chairman of otolaryngology at SUNY Downstate Health Sciences University in Brooklyn, N.Y., and lead author of the original and updated guidelines.
In practice, “the real question [for the ENT] is the future. If the ears are perfectly clear, will tubes really reduce the frequency of infections going forward?” Dr. Rosenfeld said in an interview. “All the evidence seems to say no, it doesn’t make much of a difference.”
Dr. Hackell said he’s confident that the question “is settled enough.” While there “could be stronger research and higher quality studies, the evidence is still pretty good to suggest you gain little to no benefit with tubes when you’re dealing with recurrent AOM without effusion,” he said.
Asked to comment on the ENT update and its guidance on tympanostomy tubes for children with recurrent AOM, an AAP spokesperson said the “issue is under review” and that the AAP did not currently have a statement.
At-risk children
The AAO-HNSF update renews a recommendation to evaluate children with either recurrent AOM or OME of any duration for increased risk for speech, language, or learning problems from OME because of baseline factors (sensory, physical, cognitive, or behavioral).
When OME becomes chronic – or when a tympanogram gives a flat-line reading – OME is likely to persist, and families of at-risk children especially should be encouraged to pursue tube placement, Dr. Rosenfeld said.
Despite prior guidance to this effect, he said, ear tubes are being underutilized in at-risk children, with effusion being missed in primary care and with ENTs not expediting tube placement upon referral.
“These children have learning issues, cognitive issues, developmental issues,” he said in the interview. “It’s a population that does very poorly with ears full of fluid ... and despite guidance suggesting these children should be prioritized with tubes, it doesn’t seem to be happening enough.”
Formulating guidelines for at-risk children is challenging because they are often excluded from trials, Dr. Rosenfeld said, which limits evidence about the benefits of tubes and limits the strength of recommendations.
The addition of attention-deficit/hyperactivity disorder, intellectual disability, and learning disorder to the list of risk factors is notable, Dr. Hackell said. (The list includes autism spectrum disorder, developmental delay, and suspected or confirmed speech and language delay or disorder.)
“We know that kids with ADHD take in and process information a little differently ... it may be harder to get their attention with auditory stimulation,” he said. “So anything that would impact the taking in of information even for a short period of time increases their risk.”
Surgical practice
ENTs are advised in the new guidance to use long-term tubes and perioperative antibiotic ear drops more judiciously. “Long-term tubes have a role, but there are some doctors who routinely use them, even for a first-time surgery,” said Dr. Rosenfeld.
Overuse of long-term tubes results in a higher incidence of tympanic membrane perforation, chronic drainage, and other complications, as well as greater need for long-term follow-up. “There needs to be a reason – something to justify the need for prolonged ventilation,” he said.
Perioperative antibiotic ear drops are often administered during surgery and then prescribed routinely for all children afterward, but research has shown that saline irrigation during surgery and a single application of antibiotic/steroid drops is similarly efficacious in preventing otorrhea, the guideline says. Antibiotic ear drops are also “expensive,” noted Dr. Hackell. “There’s not enough benefit to justify it.”
The update also more explicitly advises selective use of adenoidectomy. A new option says that clinicians may perform the procedure as an adjunct to tube insertion for children 4 years or older to potentially reduce the future incidence of recurrent OME or the need for repeat surgery.
However, in younger children, it should not be offered unless there are symptoms directly related to adenoid infection or nasal obstruction. “Under 4 years, there’s no primary benefit for the ears,” said Dr. Rosenfeld.
Follow-up with the surgeon after tympanostomy tube insertion should occur within 3 months to assess outcomes and educate the family, the update strongly recommends.
And pediatricians should know, Dr. Hackell notes, that clinical evidence continues to show that earplugs and other water precautions are not routinely needed for children who have tubes in place. A good approach, the guideline says, is to “first avoid water precautions and instead reserve them for children with recurrent or persistent tympanostomy tube otorrhea.”
Asked to comment on the guideline update, Tim Joos, MD, MPH, who practices combined internal medicine/pediatrics in Seattle and is an editorial advisory board member of Pediatric News, noted the inclusion of patient information sheets with frequently asked questions – resources that can be useful for guiding parents through what’s often a shared decision-making process.
Neither Dr. Rosenfeld nor Dr. Hackell reported any disclosures. Other members of the guideline update committee reported various book royalties, consulting fees, and other disclosures. Dr. Joos reported he has no connections to the guideline authors.
A practice guideline update from the ENT community on tympanostomy tubes in children reaffirms that tube insertion should not be considered in cases of otitis media with effusion (OME) lasting less than 3 months, or in children with recurrent acute otitis media (AOM) without middle ear effusion at the time of assessment for the procedure.
New in the update from the American Academy of Otolaryngology–Head and Neck Surgery Foundation (AAO-HNSF) is a strong recommendation for timely follow-up after surgery and recommendations against both routine use of prophylactic antibiotic ear drops after surgery and the initial use of long-term tubes except when there are specific reasons for doing so.
The update also expands the list of risk factors that place children with OME at increased risk of developmental difficulties – and often in need of timely ear tube placement – to include intellectual disability, learning disorder, and attention-deficit/hyperactivity disorder.
“Most of what we said in the 2013 [original] guideline was good and still valid ... and [important for] pediatricians, who are the key players” in managing otitis media, Jesse Hackell, MD, one of two general pediatricians who served on the Academy’s guideline update committee, said in an interview.
OME spontaneously clears up to 90% of the time within 3 months, said Dr. Hackell, of Pomona (New York) Pediatrics, and chair of the American Academy of Pediatrics (AAP) Committee on Practice and Ambulatory Medicine.
The updated guideline, for children 6 months to 12 years, reaffirms a recommendation that tube insertion be offered to children with “bilateral OME for 3 months or longer AND documented hearing difficulties.”
It also reaffirms “options” (a lesser quality of evidence) that in the absence of hearing difficulties, surgery may be performed for children with chronic OME (3 months or longer) in one or both ears if 1) they are at increased risk of developmental difficulties from OME or 2) effusion is likely contributing to balance problems, poor school performance, behavioral problems, ear discomfort, or reduced quality of life.
Children with chronic OME who do not undergo surgery should be reevaluated at 3- to 6-month intervals and monitored until effusion is no longer present, significant hearing loss is detected, or structural abnormalities of the tympanic membrane or middle ear are detected, the update again recommends.
Tympanostomy tube placement is the most common ambulatory surgery performed on children in the United States, the guideline authors say. In 2014, about 9% of children had undergone the surgery, they wrote, noting also that “tubes were placed in 25%-30% of children with frequent ear infections.”
Recurrent AOM
The AAO-HNSF guidance regarding tympanostomy tubes for OME is similar overall to management guidance issued by the AAP in its clinical practice guideline on OME.
The organizations differ, however, on their guidance for tube insertion for recurrent AOM. In its 2013 clinical practice guideline on AOM, the AAP recommends that clinicians may offer tube insertion for recurrent AOM, with no mention of the presence or absence of persistent fluid as a consideration.
According to the AAO-HNSF update, grade A evidence, including some research published since its original 2013 guideline, has shown little benefit to tube insertion in reducing the incidence of AOM in otherwise healthy children who don’t have middle ear effusion.
One study published in 2019 assessed outcomes after watchful waiting and found that only one-third of 123 children eventually went on to tympanostomy tube placement, noted Richard M. Rosenfeld, MD, distinguished professor and chairman of otolaryngology at SUNY Downstate Health Sciences University in Brooklyn, N.Y., and lead author of the original and updated guidelines.
In practice, “the real question [for the ENT] is the future. If the ears are perfectly clear, will tubes really reduce the frequency of infections going forward?” Dr. Rosenfeld said in an interview. “All the evidence seems to say no, it doesn’t make much of a difference.”
Dr. Hackell said he’s confident that the question “is settled enough.” While there “could be stronger research and higher quality studies, the evidence is still pretty good to suggest you gain little to no benefit with tubes when you’re dealing with recurrent AOM without effusion,” he said.
Asked to comment on the ENT update and its guidance on tympanostomy tubes for children with recurrent AOM, an AAP spokesperson said the “issue is under review” and that the AAP did not currently have a statement.
At-risk children
The AAO-HNSF update renews a recommendation to evaluate children with either recurrent AOM or OME of any duration for increased risk for speech, language, or learning problems from OME because of baseline factors (sensory, physical, cognitive, or behavioral).
When OME becomes chronic – or when a tympanogram gives a flat-line reading – OME is likely to persist, and families of at-risk children especially should be encouraged to pursue tube placement, Dr. Rosenfeld said.
Despite prior guidance to this effect, he said, ear tubes are being underutilized in at-risk children, with effusion being missed in primary care and with ENTs not expediting tube placement upon referral.
“These children have learning issues, cognitive issues, developmental issues,” he said in the interview. “It’s a population that does very poorly with ears full of fluid ... and despite guidance suggesting these children should be prioritized with tubes, it doesn’t seem to be happening enough.”
Formulating guidelines for at-risk children is challenging because they are often excluded from trials, Dr. Rosenfeld said, which limits evidence about the benefits of tubes and limits the strength of recommendations.
The addition of attention-deficit/hyperactivity disorder, intellectual disability, and learning disorder to the list of risk factors is notable, Dr. Hackell said. (The list includes autism spectrum disorder, developmental delay, and suspected or confirmed speech and language delay or disorder.)
“We know that kids with ADHD take in and process information a little differently ... it may be harder to get their attention with auditory stimulation,” he said. “So anything that would impact the taking in of information even for a short period of time increases their risk.”
Surgical practice
ENTs are advised in the new guidance to use long-term tubes and perioperative antibiotic ear drops more judiciously. “Long-term tubes have a role, but there are some doctors who routinely use them, even for a first-time surgery,” said Dr. Rosenfeld.
Overuse of long-term tubes results in a higher incidence of tympanic membrane perforation, chronic drainage, and other complications, as well as greater need for long-term follow-up. “There needs to be a reason – something to justify the need for prolonged ventilation,” he said.
Perioperative antibiotic ear drops are often administered during surgery and then prescribed routinely for all children afterward, but research has shown that saline irrigation during surgery and a single application of antibiotic/steroid drops is similarly efficacious in preventing otorrhea, the guideline says. Antibiotic ear drops are also “expensive,” noted Dr. Hackell. “There’s not enough benefit to justify it.”
The update also more explicitly advises selective use of adenoidectomy. A new option says that clinicians may perform the procedure as an adjunct to tube insertion for children 4 years or older to potentially reduce the future incidence of recurrent OME or the need for repeat surgery.
However, in younger children, it should not be offered unless there are symptoms directly related to adenoid infection or nasal obstruction. “Under 4 years, there’s no primary benefit for the ears,” said Dr. Rosenfeld.
Follow-up with the surgeon after tympanostomy tube insertion should occur within 3 months to assess outcomes and educate the family, the update strongly recommends.
And pediatricians should know, Dr. Hackell notes, that clinical evidence continues to show that earplugs and other water precautions are not routinely needed for children who have tubes in place. A good approach, the guideline says, is to “first avoid water precautions and instead reserve them for children with recurrent or persistent tympanostomy tube otorrhea.”
Asked to comment on the guideline update, Tim Joos, MD, MPH, who practices combined internal medicine/pediatrics in Seattle and is an editorial advisory board member of Pediatric News, noted the inclusion of patient information sheets with frequently asked questions – resources that can be useful for guiding parents through what’s often a shared decision-making process.
Neither Dr. Rosenfeld nor Dr. Hackell reported any disclosures. Other members of the guideline update committee reported various book royalties, consulting fees, and other disclosures. Dr. Joos reported he has no connections to the guideline authors.
FROM OTOLARYNGOLOGY HEAD AND NECK SURGERY
Treatment duration for acute otitis media – so many choices
Twenty years ago, the dilemma in treating acute otitis media (AOM) was which among 10-plus antibiotics to prescribe. A recent column discussed the evolving pathogen distribution in AOM and its effects on antibiotic choices.1 But here we consider treatment duration. Until the past decade, AOM treatment (except azithromycin) involved 10-day courses. But lately, 10-day antibiotic regimens for uncomplicated infections are disappearing. Shorter-course recommendations are the new norm because of the evolving clinical data showing that an appropriately chosen antibiotic (in partnership with host defenses and source control) resolves infection faster than was previously thought. Shorter courses make sense because of fewer adverse effects, less distortion of normal flora, and less likely induction of pathogen resistance. Table 4.12 in the newest 2021-2024 SOID Redbook lists three antibiotic durations for AOM, and actually there are more than that.
Why so many duration options? Clinical data show that not all AOM is alike and short courses work for subsets of AOM because, besides antibiotics, key elements in AOM resolution are host anatomy and immunity. Bacterial AOM results from a combination of refluxed pathogens in the middle ear being trapped when the eustachian tube malfunctions (infection occurs when middle ear plumbing gets stopped up). If the eustachian tube spontaneously drains and the host immune response slows/stops pathogen growth, no antibiotics are needed. Indeed, a sizable proportion of mild/moderate AOM episodes spontaneously resolve, particularly in children over 2 years old. So a high likelihood of spontaneous remission allows an initial 0-days duration option (watchful waiting) or delayed antibiotics (rescue prescriptions) for older children.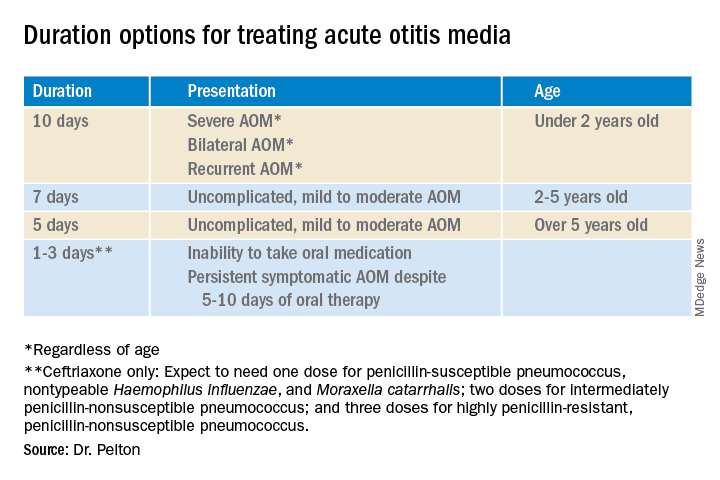
That said, when one chooses to initially prescribe antibiotics for AOM, different durations are recommended. Table 1 has my suggestions.
Data that gave me better microbiological understanding of why oral AOM trials less than 10 days were successful involved purulent AOM drainage from children who had pressure-equalizing (PE) tubes.2 The authors randomized children to either standard-dose amoxicillin-clavulanate or placebo. Of note, 95% of pathogens were susceptible to the antibiotic; 5% were pneumococcus intermediately resistant to penicillin. The authors sampled ear drainage daily for 7 days. Figure 1 shows that cultures remained positive in only around 5% of children by day 3-5 of antibiotics, but viable bacteria persisted through 7 days in over half of placebo recipients. Remember, both groups benefited from a form of source control (drainage of the middle ear via PE tubes). So, if antibiotics can do the job in 3-5 days, why continue antibiotics beyond 5 days?
Anatomy and severity. In children over 5 years old (reasonably mature eustachian tube anatomy) with nonrecurrent (no AOM in past month), nonsevere (no otalgia or high fever) AOM, 5 days is enough. But 2- to 5-year-olds (less mature anatomy) need 7 days and those <2 years old (least mature plumbing) need 10 days. Likewise, severe AOM usually warrants 10 days. Some experts recommend 10 days for bilateral AOM as well.
These age/severity differences make sense because failures are more frequent with:
1. Younger age.3 While not proven, my hypothesis is that “natural” source control (spontaneous internal draining the middle ear into the nasopharynx [NP]) is less frequent in younger children because they have less mature eustachian tube systems. Further, reflux of persisting NP organisms could restart a new AOM episode even if the original pathogen was eliminated by a short 5-day course.
2. Severe AOM. A rationale for longer courses in severe AOM (ear pain, high fever) is that high middle-ear pressures (indicated by degree of tympanic membrane bulging and ear pain) could impede antibiotic penetration, or that high initial bacterial loads (perhaps indicated by systemic fever) require more antibiotic. And finally, return to baseline eustachian tube function may take longer if severe AOM caused enhanced inflammation.
3. Recurrent AOM. (AOM within 1 prior month) – With recurrent AOM, the second “hit” to the eustachian tube may lead to more dysfunction, so a longer antibiotic course may be required to allow more complete source control and more time for more complete functional recovery after a repeated inflammatory injury.
4. Bilateral AOM. Two independent but infected sites mean twice the chance for failure. So, a longer course could allow more time for both sites to undergo “natural” source control.4
More bacteria – more antibiotic? So, is more antibiotic really needed for a higher bacterial load? In vitro this is known as the “inoculum effect,” particularly for beta-lactam drugs, for example, amoxicillin and cephalosporins. Laboratory susceptibility testing is performed with a specifically defined quantity of bacteria (105 bacteria/mL) and the minimum inhibitory concentration (MIC) is the lowest antibiotic concentration that stops bacterial growth. We know that drugs will likely fail if the MIC exceeds the achievable antibiotic concentration at the infection site. But is it as simple as just exceeding the MIC at the infection site? No, pharmacodynamics tell us that overall antibiotic exposure is also important. For example, to be successful, beta-lactam concentrations need to be above the MIC for 40%-50% of the day.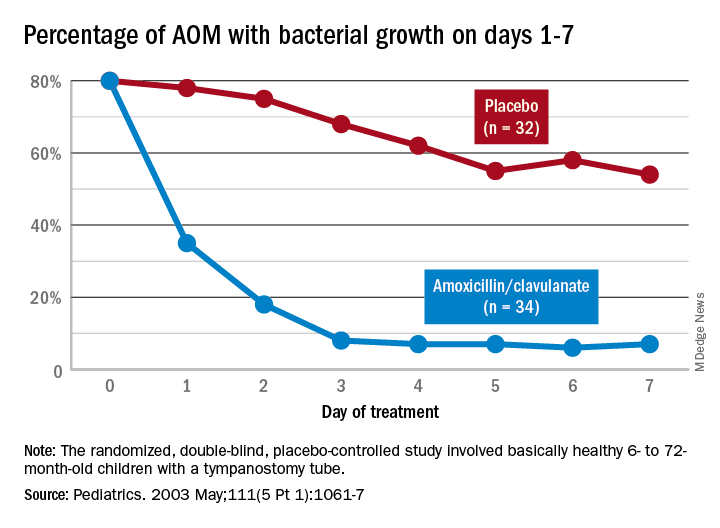
Higher MIC with higher bacterial load. Particularly for beta-lactams, testing with a quantity of bacteria >105/mL produces a higher MIC in vitro. This suggests that clinical failure could occur, even when our in vivo dosing leads to 40%-50% above the “standard” MIC that was obtained from testing the lab standard of 105/mL bacteria, when the infected site’s (middle ear) bacterial load is >105/mL (such higher bacterial loads occur in up to 30% of AOM).5 One way to negate inoculum effect is source control (drain the abscess or debridement), which reduces the bacterial load as well as allowing better antibiotic penetration– both favoring infection resolution. But with suboptimal source control, for example, the middle ear is not drained externally or internally, longer courses (more antibiotic exposure) could aid resolution. Whether the exposure can be administered as higher doses in fewer days or standard doses for more days is debatable but consider that a single parenteral dose of ceftriaxone successfully resolves AOM not attributable to penicillin-nonsusceptible pneumococcus.6Bottom line: Even though the number of potential antibiotics has contracted in the past 20 years, the need to individualize AOM treatment remains important and duration choices are more complex. Indeed, AOM comes in different flavors with patient age, clinical presentation, and episode frequency dictating the choice of duration.
Dr. Christopher J. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. Email him at [email protected].
References
1. Pichichero ME. MDedge. 2022 Jan 11.
2. Ruohola A et al. Pediatrics. 2003;111(5):1061-7.
3. Hoberman A et al. N Engl J Med. 2016;375(25):2446-56.
4. Pichichero ME et al. Otolaryngol Head Neck Surg. 2001;124(4):381-7.
5. Harrison CJ et al. Pediatr Infect Dis. 1985;4(6):641-6.
6. Leibovitz E et al. Pediatr Infect Dis. 2000;19(11):1040-5.
Twenty years ago, the dilemma in treating acute otitis media (AOM) was which among 10-plus antibiotics to prescribe. A recent column discussed the evolving pathogen distribution in AOM and its effects on antibiotic choices.1 But here we consider treatment duration. Until the past decade, AOM treatment (except azithromycin) involved 10-day courses. But lately, 10-day antibiotic regimens for uncomplicated infections are disappearing. Shorter-course recommendations are the new norm because of the evolving clinical data showing that an appropriately chosen antibiotic (in partnership with host defenses and source control) resolves infection faster than was previously thought. Shorter courses make sense because of fewer adverse effects, less distortion of normal flora, and less likely induction of pathogen resistance. Table 4.12 in the newest 2021-2024 SOID Redbook lists three antibiotic durations for AOM, and actually there are more than that.
Why so many duration options? Clinical data show that not all AOM is alike and short courses work for subsets of AOM because, besides antibiotics, key elements in AOM resolution are host anatomy and immunity. Bacterial AOM results from a combination of refluxed pathogens in the middle ear being trapped when the eustachian tube malfunctions (infection occurs when middle ear plumbing gets stopped up). If the eustachian tube spontaneously drains and the host immune response slows/stops pathogen growth, no antibiotics are needed. Indeed, a sizable proportion of mild/moderate AOM episodes spontaneously resolve, particularly in children over 2 years old. So a high likelihood of spontaneous remission allows an initial 0-days duration option (watchful waiting) or delayed antibiotics (rescue prescriptions) for older children.
That said, when one chooses to initially prescribe antibiotics for AOM, different durations are recommended. Table 1 has my suggestions.
Data that gave me better microbiological understanding of why oral AOM trials less than 10 days were successful involved purulent AOM drainage from children who had pressure-equalizing (PE) tubes.2 The authors randomized children to either standard-dose amoxicillin-clavulanate or placebo. Of note, 95% of pathogens were susceptible to the antibiotic; 5% were pneumococcus intermediately resistant to penicillin. The authors sampled ear drainage daily for 7 days. Figure 1 shows that cultures remained positive in only around 5% of children by day 3-5 of antibiotics, but viable bacteria persisted through 7 days in over half of placebo recipients. Remember, both groups benefited from a form of source control (drainage of the middle ear via PE tubes). So, if antibiotics can do the job in 3-5 days, why continue antibiotics beyond 5 days?
Anatomy and severity. In children over 5 years old (reasonably mature eustachian tube anatomy) with nonrecurrent (no AOM in past month), nonsevere (no otalgia or high fever) AOM, 5 days is enough. But 2- to 5-year-olds (less mature anatomy) need 7 days and those <2 years old (least mature plumbing) need 10 days. Likewise, severe AOM usually warrants 10 days. Some experts recommend 10 days for bilateral AOM as well.
These age/severity differences make sense because failures are more frequent with:
1. Younger age.3 While not proven, my hypothesis is that “natural” source control (spontaneous internal draining the middle ear into the nasopharynx [NP]) is less frequent in younger children because they have less mature eustachian tube systems. Further, reflux of persisting NP organisms could restart a new AOM episode even if the original pathogen was eliminated by a short 5-day course.
2. Severe AOM. A rationale for longer courses in severe AOM (ear pain, high fever) is that high middle-ear pressures (indicated by degree of tympanic membrane bulging and ear pain) could impede antibiotic penetration, or that high initial bacterial loads (perhaps indicated by systemic fever) require more antibiotic. And finally, return to baseline eustachian tube function may take longer if severe AOM caused enhanced inflammation.
3. Recurrent AOM. (AOM within 1 prior month) – With recurrent AOM, the second “hit” to the eustachian tube may lead to more dysfunction, so a longer antibiotic course may be required to allow more complete source control and more time for more complete functional recovery after a repeated inflammatory injury.
4. Bilateral AOM. Two independent but infected sites mean twice the chance for failure. So, a longer course could allow more time for both sites to undergo “natural” source control.4
More bacteria – more antibiotic? So, is more antibiotic really needed for a higher bacterial load? In vitro this is known as the “inoculum effect,” particularly for beta-lactam drugs, for example, amoxicillin and cephalosporins. Laboratory susceptibility testing is performed with a specifically defined quantity of bacteria (105 bacteria/mL) and the minimum inhibitory concentration (MIC) is the lowest antibiotic concentration that stops bacterial growth. We know that drugs will likely fail if the MIC exceeds the achievable antibiotic concentration at the infection site. But is it as simple as just exceeding the MIC at the infection site? No, pharmacodynamics tell us that overall antibiotic exposure is also important. For example, to be successful, beta-lactam concentrations need to be above the MIC for 40%-50% of the day.
Higher MIC with higher bacterial load. Particularly for beta-lactams, testing with a quantity of bacteria >105/mL produces a higher MIC in vitro. This suggests that clinical failure could occur, even when our in vivo dosing leads to 40%-50% above the “standard” MIC that was obtained from testing the lab standard of 105/mL bacteria, when the infected site’s (middle ear) bacterial load is >105/mL (such higher bacterial loads occur in up to 30% of AOM).5 One way to negate inoculum effect is source control (drain the abscess or debridement), which reduces the bacterial load as well as allowing better antibiotic penetration– both favoring infection resolution. But with suboptimal source control, for example, the middle ear is not drained externally or internally, longer courses (more antibiotic exposure) could aid resolution. Whether the exposure can be administered as higher doses in fewer days or standard doses for more days is debatable but consider that a single parenteral dose of ceftriaxone successfully resolves AOM not attributable to penicillin-nonsusceptible pneumococcus.6Bottom line: Even though the number of potential antibiotics has contracted in the past 20 years, the need to individualize AOM treatment remains important and duration choices are more complex. Indeed, AOM comes in different flavors with patient age, clinical presentation, and episode frequency dictating the choice of duration.
Dr. Christopher J. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. Email him at [email protected].
References
1. Pichichero ME. MDedge. 2022 Jan 11.
2. Ruohola A et al. Pediatrics. 2003;111(5):1061-7.
3. Hoberman A et al. N Engl J Med. 2016;375(25):2446-56.
4. Pichichero ME et al. Otolaryngol Head Neck Surg. 2001;124(4):381-7.
5. Harrison CJ et al. Pediatr Infect Dis. 1985;4(6):641-6.
6. Leibovitz E et al. Pediatr Infect Dis. 2000;19(11):1040-5.
Twenty years ago, the dilemma in treating acute otitis media (AOM) was which among 10-plus antibiotics to prescribe. A recent column discussed the evolving pathogen distribution in AOM and its effects on antibiotic choices.1 But here we consider treatment duration. Until the past decade, AOM treatment (except azithromycin) involved 10-day courses. But lately, 10-day antibiotic regimens for uncomplicated infections are disappearing. Shorter-course recommendations are the new norm because of the evolving clinical data showing that an appropriately chosen antibiotic (in partnership with host defenses and source control) resolves infection faster than was previously thought. Shorter courses make sense because of fewer adverse effects, less distortion of normal flora, and less likely induction of pathogen resistance. Table 4.12 in the newest 2021-2024 SOID Redbook lists three antibiotic durations for AOM, and actually there are more than that.
Why so many duration options? Clinical data show that not all AOM is alike and short courses work for subsets of AOM because, besides antibiotics, key elements in AOM resolution are host anatomy and immunity. Bacterial AOM results from a combination of refluxed pathogens in the middle ear being trapped when the eustachian tube malfunctions (infection occurs when middle ear plumbing gets stopped up). If the eustachian tube spontaneously drains and the host immune response slows/stops pathogen growth, no antibiotics are needed. Indeed, a sizable proportion of mild/moderate AOM episodes spontaneously resolve, particularly in children over 2 years old. So a high likelihood of spontaneous remission allows an initial 0-days duration option (watchful waiting) or delayed antibiotics (rescue prescriptions) for older children.
That said, when one chooses to initially prescribe antibiotics for AOM, different durations are recommended. Table 1 has my suggestions.
Data that gave me better microbiological understanding of why oral AOM trials less than 10 days were successful involved purulent AOM drainage from children who had pressure-equalizing (PE) tubes.2 The authors randomized children to either standard-dose amoxicillin-clavulanate or placebo. Of note, 95% of pathogens were susceptible to the antibiotic; 5% were pneumococcus intermediately resistant to penicillin. The authors sampled ear drainage daily for 7 days. Figure 1 shows that cultures remained positive in only around 5% of children by day 3-5 of antibiotics, but viable bacteria persisted through 7 days in over half of placebo recipients. Remember, both groups benefited from a form of source control (drainage of the middle ear via PE tubes). So, if antibiotics can do the job in 3-5 days, why continue antibiotics beyond 5 days?
Anatomy and severity. In children over 5 years old (reasonably mature eustachian tube anatomy) with nonrecurrent (no AOM in past month), nonsevere (no otalgia or high fever) AOM, 5 days is enough. But 2- to 5-year-olds (less mature anatomy) need 7 days and those <2 years old (least mature plumbing) need 10 days. Likewise, severe AOM usually warrants 10 days. Some experts recommend 10 days for bilateral AOM as well.
These age/severity differences make sense because failures are more frequent with:
1. Younger age.3 While not proven, my hypothesis is that “natural” source control (spontaneous internal draining the middle ear into the nasopharynx [NP]) is less frequent in younger children because they have less mature eustachian tube systems. Further, reflux of persisting NP organisms could restart a new AOM episode even if the original pathogen was eliminated by a short 5-day course.
2. Severe AOM. A rationale for longer courses in severe AOM (ear pain, high fever) is that high middle-ear pressures (indicated by degree of tympanic membrane bulging and ear pain) could impede antibiotic penetration, or that high initial bacterial loads (perhaps indicated by systemic fever) require more antibiotic. And finally, return to baseline eustachian tube function may take longer if severe AOM caused enhanced inflammation.
3. Recurrent AOM. (AOM within 1 prior month) – With recurrent AOM, the second “hit” to the eustachian tube may lead to more dysfunction, so a longer antibiotic course may be required to allow more complete source control and more time for more complete functional recovery after a repeated inflammatory injury.
4. Bilateral AOM. Two independent but infected sites mean twice the chance for failure. So, a longer course could allow more time for both sites to undergo “natural” source control.4
More bacteria – more antibiotic? So, is more antibiotic really needed for a higher bacterial load? In vitro this is known as the “inoculum effect,” particularly for beta-lactam drugs, for example, amoxicillin and cephalosporins. Laboratory susceptibility testing is performed with a specifically defined quantity of bacteria (105 bacteria/mL) and the minimum inhibitory concentration (MIC) is the lowest antibiotic concentration that stops bacterial growth. We know that drugs will likely fail if the MIC exceeds the achievable antibiotic concentration at the infection site. But is it as simple as just exceeding the MIC at the infection site? No, pharmacodynamics tell us that overall antibiotic exposure is also important. For example, to be successful, beta-lactam concentrations need to be above the MIC for 40%-50% of the day.
Higher MIC with higher bacterial load. Particularly for beta-lactams, testing with a quantity of bacteria >105/mL produces a higher MIC in vitro. This suggests that clinical failure could occur, even when our in vivo dosing leads to 40%-50% above the “standard” MIC that was obtained from testing the lab standard of 105/mL bacteria, when the infected site’s (middle ear) bacterial load is >105/mL (such higher bacterial loads occur in up to 30% of AOM).5 One way to negate inoculum effect is source control (drain the abscess or debridement), which reduces the bacterial load as well as allowing better antibiotic penetration– both favoring infection resolution. But with suboptimal source control, for example, the middle ear is not drained externally or internally, longer courses (more antibiotic exposure) could aid resolution. Whether the exposure can be administered as higher doses in fewer days or standard doses for more days is debatable but consider that a single parenteral dose of ceftriaxone successfully resolves AOM not attributable to penicillin-nonsusceptible pneumococcus.6Bottom line: Even though the number of potential antibiotics has contracted in the past 20 years, the need to individualize AOM treatment remains important and duration choices are more complex. Indeed, AOM comes in different flavors with patient age, clinical presentation, and episode frequency dictating the choice of duration.
Dr. Christopher J. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. Email him at [email protected].
References
1. Pichichero ME. MDedge. 2022 Jan 11.
2. Ruohola A et al. Pediatrics. 2003;111(5):1061-7.
3. Hoberman A et al. N Engl J Med. 2016;375(25):2446-56.
4. Pichichero ME et al. Otolaryngol Head Neck Surg. 2001;124(4):381-7.
5. Harrison CJ et al. Pediatr Infect Dis. 1985;4(6):641-6.
6. Leibovitz E et al. Pediatr Infect Dis. 2000;19(11):1040-5.
Sepsis common cause of ICU admissions in patients with MS
Sepsis is an alarmingly common cause behind ICU admissions in patients with multiple sclerosis (MS), a retrospective, population-based cohort study indicates.
Furthermore, it contributes to a disproportionately high percentage of the short-term mortality risk among patients with MS admitted to the ICU, findings also show. Short-term mortality risk was defined in the study as a combination of in-hospital death or discharge to hospice.
“We found that the risk of short-term mortality in critically ill patients with MS is four times higher among those with sepsis ... so sepsis appears to be comparatively more lethal among patients with MS than in the general population,” Lavi Oud, MD, professor of medicine, Texas Tech University HSC at the Permian Basin, Odessa, said in an email.
“[Although] the specific mechanisms underlying the markedly higher risk of sepsis among patients with MS compared to the general population remain to be fully elucidated ... it’s thought that the risk may stem from the dysfunction of the immune system in these patients related to MS itself and to the potentially adverse effect of the immunomodulating therapy we use in these patients,” he added.
The study was published online Jan. 11 in the Journal of Critical Care.
Sepsis rates
The Texas Inpatient Public Use Data File was used to identify adults with a diagnosis of MS admitted to the hospital between 2010 and 2017. Among the 19,837 patients with MS admitted to the ICU during the study interval, almost one-third (31.5%) had sepsis, investigators report. “The rate of sepsis among ICU admissions increased with age, ranging from 20.8% among those aged 18-44 to 39.4% among those aged 65 years or older,” investigators note.
The most common site of infection among MS patients admitted to the ICU were urinary in nature (65.2%), followed by respiratory (36.1%). A smaller proportion of infections (7.6%) involved the skin and soft tissues, researchers note. A full one-quarter of patients developed septic shock in response to their infection while the length of stay among patients with sepsis (mean of 10.9 days) was substantially longer than it was for those without sepsis (mean of 5.6 days), they observe.
At a mean total hospital cost of $121,797 for each ICU patient with sepsis, the cost of caring for each patient was nearly twofold higher than the mean total cost of taking care of ICU patients without sepsis (mean total cost, $65,179). On adjusted analysis, sepsis was associated with a 42.7% (95% confidence interval, 38.9-46.5; P < .0001) longer length of hospital stay and a 26.2% (95% CI, 23.1-29.1; P < .0001) higher total hospital cost compared with patients without sepsis, the authors point out.
Indeed, ICU admissions with sepsis accounted for 47.3% of all hospital days and for 46.1% of the aggregate hospital charges among all MS patients admitted to the ICU.
“The adjusted probability of short-term mortality was 13.4% (95% CI, 13.0-13.7) among ICU admissions with sepsis and 3.3% (95% CI, 3.2-3.4) among ICU admissions without sepsis,” the authors report.
This translated into a 44% higher risk of short-term mortality at an adjusted odds ratio of 1.44 (95% CI, 1.23-1.69; P < .0001) for those with sepsis, compared with those without, they add. Among all ICU admissions, sepsis was reported in over two-thirds of documented short-term mortality events. The risk of short-term mortality was also almost threefold higher among patients with sepsis who were age 65 years and older compared with patients aged 18-44.
As Dr. Oud noted, there is no specific test for sepsis, and it can initially present in an atypical manner, especially in older, frailer, chronically ill patients as well as in patients with immune dysfunction. “Thus, considering sepsis as a possible cause of new deterioration in a patient’s condition is essential, along with the timely start of sepsis-related care,” Dr. Oud observed.
A limitation of the study was that the dataset did not include information on the type of MS a patient had, the duration of their illness, the treatment received, the level of disease activity, or the level of disability.
The study had no specific funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sepsis is an alarmingly common cause behind ICU admissions in patients with multiple sclerosis (MS), a retrospective, population-based cohort study indicates.
Furthermore, it contributes to a disproportionately high percentage of the short-term mortality risk among patients with MS admitted to the ICU, findings also show. Short-term mortality risk was defined in the study as a combination of in-hospital death or discharge to hospice.
“We found that the risk of short-term mortality in critically ill patients with MS is four times higher among those with sepsis ... so sepsis appears to be comparatively more lethal among patients with MS than in the general population,” Lavi Oud, MD, professor of medicine, Texas Tech University HSC at the Permian Basin, Odessa, said in an email.
“[Although] the specific mechanisms underlying the markedly higher risk of sepsis among patients with MS compared to the general population remain to be fully elucidated ... it’s thought that the risk may stem from the dysfunction of the immune system in these patients related to MS itself and to the potentially adverse effect of the immunomodulating therapy we use in these patients,” he added.
The study was published online Jan. 11 in the Journal of Critical Care.
Sepsis rates
The Texas Inpatient Public Use Data File was used to identify adults with a diagnosis of MS admitted to the hospital between 2010 and 2017. Among the 19,837 patients with MS admitted to the ICU during the study interval, almost one-third (31.5%) had sepsis, investigators report. “The rate of sepsis among ICU admissions increased with age, ranging from 20.8% among those aged 18-44 to 39.4% among those aged 65 years or older,” investigators note.
The most common site of infection among MS patients admitted to the ICU were urinary in nature (65.2%), followed by respiratory (36.1%). A smaller proportion of infections (7.6%) involved the skin and soft tissues, researchers note. A full one-quarter of patients developed septic shock in response to their infection while the length of stay among patients with sepsis (mean of 10.9 days) was substantially longer than it was for those without sepsis (mean of 5.6 days), they observe.
At a mean total hospital cost of $121,797 for each ICU patient with sepsis, the cost of caring for each patient was nearly twofold higher than the mean total cost of taking care of ICU patients without sepsis (mean total cost, $65,179). On adjusted analysis, sepsis was associated with a 42.7% (95% confidence interval, 38.9-46.5; P < .0001) longer length of hospital stay and a 26.2% (95% CI, 23.1-29.1; P < .0001) higher total hospital cost compared with patients without sepsis, the authors point out.
Indeed, ICU admissions with sepsis accounted for 47.3% of all hospital days and for 46.1% of the aggregate hospital charges among all MS patients admitted to the ICU.
“The adjusted probability of short-term mortality was 13.4% (95% CI, 13.0-13.7) among ICU admissions with sepsis and 3.3% (95% CI, 3.2-3.4) among ICU admissions without sepsis,” the authors report.
This translated into a 44% higher risk of short-term mortality at an adjusted odds ratio of 1.44 (95% CI, 1.23-1.69; P < .0001) for those with sepsis, compared with those without, they add. Among all ICU admissions, sepsis was reported in over two-thirds of documented short-term mortality events. The risk of short-term mortality was also almost threefold higher among patients with sepsis who were age 65 years and older compared with patients aged 18-44.
As Dr. Oud noted, there is no specific test for sepsis, and it can initially present in an atypical manner, especially in older, frailer, chronically ill patients as well as in patients with immune dysfunction. “Thus, considering sepsis as a possible cause of new deterioration in a patient’s condition is essential, along with the timely start of sepsis-related care,” Dr. Oud observed.
A limitation of the study was that the dataset did not include information on the type of MS a patient had, the duration of their illness, the treatment received, the level of disease activity, or the level of disability.
The study had no specific funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sepsis is an alarmingly common cause behind ICU admissions in patients with multiple sclerosis (MS), a retrospective, population-based cohort study indicates.
Furthermore, it contributes to a disproportionately high percentage of the short-term mortality risk among patients with MS admitted to the ICU, findings also show. Short-term mortality risk was defined in the study as a combination of in-hospital death or discharge to hospice.
“We found that the risk of short-term mortality in critically ill patients with MS is four times higher among those with sepsis ... so sepsis appears to be comparatively more lethal among patients with MS than in the general population,” Lavi Oud, MD, professor of medicine, Texas Tech University HSC at the Permian Basin, Odessa, said in an email.
“[Although] the specific mechanisms underlying the markedly higher risk of sepsis among patients with MS compared to the general population remain to be fully elucidated ... it’s thought that the risk may stem from the dysfunction of the immune system in these patients related to MS itself and to the potentially adverse effect of the immunomodulating therapy we use in these patients,” he added.
The study was published online Jan. 11 in the Journal of Critical Care.
Sepsis rates
The Texas Inpatient Public Use Data File was used to identify adults with a diagnosis of MS admitted to the hospital between 2010 and 2017. Among the 19,837 patients with MS admitted to the ICU during the study interval, almost one-third (31.5%) had sepsis, investigators report. “The rate of sepsis among ICU admissions increased with age, ranging from 20.8% among those aged 18-44 to 39.4% among those aged 65 years or older,” investigators note.
The most common site of infection among MS patients admitted to the ICU were urinary in nature (65.2%), followed by respiratory (36.1%). A smaller proportion of infections (7.6%) involved the skin and soft tissues, researchers note. A full one-quarter of patients developed septic shock in response to their infection while the length of stay among patients with sepsis (mean of 10.9 days) was substantially longer than it was for those without sepsis (mean of 5.6 days), they observe.
At a mean total hospital cost of $121,797 for each ICU patient with sepsis, the cost of caring for each patient was nearly twofold higher than the mean total cost of taking care of ICU patients without sepsis (mean total cost, $65,179). On adjusted analysis, sepsis was associated with a 42.7% (95% confidence interval, 38.9-46.5; P < .0001) longer length of hospital stay and a 26.2% (95% CI, 23.1-29.1; P < .0001) higher total hospital cost compared with patients without sepsis, the authors point out.
Indeed, ICU admissions with sepsis accounted for 47.3% of all hospital days and for 46.1% of the aggregate hospital charges among all MS patients admitted to the ICU.
“The adjusted probability of short-term mortality was 13.4% (95% CI, 13.0-13.7) among ICU admissions with sepsis and 3.3% (95% CI, 3.2-3.4) among ICU admissions without sepsis,” the authors report.
This translated into a 44% higher risk of short-term mortality at an adjusted odds ratio of 1.44 (95% CI, 1.23-1.69; P < .0001) for those with sepsis, compared with those without, they add. Among all ICU admissions, sepsis was reported in over two-thirds of documented short-term mortality events. The risk of short-term mortality was also almost threefold higher among patients with sepsis who were age 65 years and older compared with patients aged 18-44.
As Dr. Oud noted, there is no specific test for sepsis, and it can initially present in an atypical manner, especially in older, frailer, chronically ill patients as well as in patients with immune dysfunction. “Thus, considering sepsis as a possible cause of new deterioration in a patient’s condition is essential, along with the timely start of sepsis-related care,” Dr. Oud observed.
A limitation of the study was that the dataset did not include information on the type of MS a patient had, the duration of their illness, the treatment received, the level of disease activity, or the level of disability.
The study had no specific funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CRITICAL CARE
Strep infection and tics in children: new data
Group A streptococcus (GAS) infection is not associated with new-onset tic disorders in at-risk children, findings from a large prospective study show.
The results mean that if preteens present with a new-onset tic condition, “they’re unlikely to have it as a result of a group A streptococcal throat infection,” study author Anette Eleonore Schrag, MD, PhD, professor, department of clinical neuroscience, Institute of Neurology, University College London, told this news organization.
Therefore, clinicians should not automatically prescribe antibiotics for children with tics, which sometimes occurs, said Dr. Schrag.
The study was published online Feb. 2 in Neurology.
Ongoing controversy
Research shows that genetic and environmental factors contribute to chronic tic disorders (CTDs) and Tourette syndrome (TS). Prenatal exposure to maternal smoking and central nervous system (CNS) stimulants, as well as psychosocial stress, may play a role.
There has been an ongoing controversy regarding the possible role of GAS in tics, with some studies showing an association and others not showing a link. However, previous studies have been retrospective, registry based, or had limited sample size.
This new prospective study is the first in children without a tic disorder but who were at relatively high risk of developing one. The children were followed to assess development of streptococcal infections and tics, said Dr. Schrag.
The study included 259 children aged 3-10 years (mean baseline age, 6.8 years; over half female) who had a first-degree relative such as a parent or sibling with TS or CTD.
The average age at TS onset is 7 years, peaking in prevalence and severity at about 9-12 years. GAS throat infections are common in this age group.
Although study participants did not have tics themselves, they represented “an enriched group,” said Dr. Schrag. “Because they had family history, we knew they were at increased risk for developing tics.”
Participants were evaluated every 2 months, alternating between scheduled hospital visits and telephone interviews. Parents kept a weekly diary and were instructed to bring their child in for assessment if they showed any signs of tics.
The average follow-up period was 1.6 years, but some of the children were followed for up to 48 months. During the study, there were a total of 1,944 assessments, including 939 telephone interviews and 1,005 clinical visits.
More common in boys
Investigators defined tic onset as the first occurrence of any sudden, rapid, recurrent, nonrhythmic involuntary movement and/or vocalization on at least three separate days within a period of 3 weeks.
The investigators assessed GAS exposure using parameters from throat swabs, serum anti-streptolysin O titers, and anti-DNAse B titers.
They used multiple definitions and combinations of GAS exposures “to make sure we weren’t missing any association because we didn’t use the right definition,” said Dr. Schrag. She explained a definitive strep infection is not always clear-cut.
At baseline, 17.0% participants tested positive for GAS, and 78.8% tested negative. No throat swab was available from 4.2% of participants.
During follow-up, the number of confirmed positive GAS exposures was 59, 102, 125, and 138, depending on the definition.
Researchers identified 61 tic cases during the study period. There was no evidence of an association of tic onset with GAS exposure after adjusting for age, sex, and parental education level.
However, there was a strong association between tic onset and sex, with girls being 60% less likely to develop tics than boys (hazard ratio, 0.4; 95% CI, 0.2-0.7; P < .01).
This result wasn’t particularly surprising, as it’s known that more boys develop tics than girls. “We just confirmed that in a prospective way,” said Dr. Schrag.
Results from sensitivity analyses confirmed the results. This was also the case with analyses that excluded visits with missing data on GAS exposure and that further adjusted for clinical site and psychotropic medication use.
Other pathogens?
Although the results showed no association between strep and tics in this population, it does not “close the door completely” on a potential relationship, said Dr. Schrag.
“By and large, the development of tics in children is not associated with group A strep, but differences in small subgroups can never be excluded by a study like this.”
Participants in this study were part of the European Multicentre Tics in Children Studies (EMTICS), a prospective cohort study exploring the role of environmental and genetic factors in pediatric CTD. That project is also looking at immune system factors, “which might play a role in the development of chronic tic disorder and associated conditions,” said Dr. Schrag.
It’s still possible, she added, that other pathogens could play a role in tic development. “That’s going to be the subject of further analysis and future studies,” she said.
Tamara Pringsheim, MD, professor of clinical neurosciences, psychiatry, pediatrics, and community health sciences, University of Calgary (Alta.), praised the research.
“This was a well-designed study, with a large sample of 260 children followed for up to 4 years, using a standardized protocol to assess for group A streptococcal infection and new onset of tics.”
The study, which did not uncover an association between GAS exposure and tic onset, “provides high level evidence that group A streptococcal exposure is not an important risk factor for the new onset of tics in children with a family history of tic disorders.”
The study received funding from the European Union Seventh Framework Program for research technological development and demonstration. Dr. Schrag reports receiving consultancy or advisory board honoraria from Biogen, Abbvie, Bial, and Neurotechnology; research support from the National Institute of Health Research, Parkinsons UK, and the Economic and Social Research Council and the European Commission; and Royalties from Oxford University Press. Dr. Pringsheim reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Group A streptococcus (GAS) infection is not associated with new-onset tic disorders in at-risk children, findings from a large prospective study show.
The results mean that if preteens present with a new-onset tic condition, “they’re unlikely to have it as a result of a group A streptococcal throat infection,” study author Anette Eleonore Schrag, MD, PhD, professor, department of clinical neuroscience, Institute of Neurology, University College London, told this news organization.
Therefore, clinicians should not automatically prescribe antibiotics for children with tics, which sometimes occurs, said Dr. Schrag.
The study was published online Feb. 2 in Neurology.
Ongoing controversy
Research shows that genetic and environmental factors contribute to chronic tic disorders (CTDs) and Tourette syndrome (TS). Prenatal exposure to maternal smoking and central nervous system (CNS) stimulants, as well as psychosocial stress, may play a role.
There has been an ongoing controversy regarding the possible role of GAS in tics, with some studies showing an association and others not showing a link. However, previous studies have been retrospective, registry based, or had limited sample size.
This new prospective study is the first in children without a tic disorder but who were at relatively high risk of developing one. The children were followed to assess development of streptococcal infections and tics, said Dr. Schrag.
The study included 259 children aged 3-10 years (mean baseline age, 6.8 years; over half female) who had a first-degree relative such as a parent or sibling with TS or CTD.
The average age at TS onset is 7 years, peaking in prevalence and severity at about 9-12 years. GAS throat infections are common in this age group.
Although study participants did not have tics themselves, they represented “an enriched group,” said Dr. Schrag. “Because they had family history, we knew they were at increased risk for developing tics.”
Participants were evaluated every 2 months, alternating between scheduled hospital visits and telephone interviews. Parents kept a weekly diary and were instructed to bring their child in for assessment if they showed any signs of tics.
The average follow-up period was 1.6 years, but some of the children were followed for up to 48 months. During the study, there were a total of 1,944 assessments, including 939 telephone interviews and 1,005 clinical visits.
More common in boys
Investigators defined tic onset as the first occurrence of any sudden, rapid, recurrent, nonrhythmic involuntary movement and/or vocalization on at least three separate days within a period of 3 weeks.
The investigators assessed GAS exposure using parameters from throat swabs, serum anti-streptolysin O titers, and anti-DNAse B titers.
They used multiple definitions and combinations of GAS exposures “to make sure we weren’t missing any association because we didn’t use the right definition,” said Dr. Schrag. She explained a definitive strep infection is not always clear-cut.
At baseline, 17.0% participants tested positive for GAS, and 78.8% tested negative. No throat swab was available from 4.2% of participants.
During follow-up, the number of confirmed positive GAS exposures was 59, 102, 125, and 138, depending on the definition.
Researchers identified 61 tic cases during the study period. There was no evidence of an association of tic onset with GAS exposure after adjusting for age, sex, and parental education level.
However, there was a strong association between tic onset and sex, with girls being 60% less likely to develop tics than boys (hazard ratio, 0.4; 95% CI, 0.2-0.7; P < .01).
This result wasn’t particularly surprising, as it’s known that more boys develop tics than girls. “We just confirmed that in a prospective way,” said Dr. Schrag.
Results from sensitivity analyses confirmed the results. This was also the case with analyses that excluded visits with missing data on GAS exposure and that further adjusted for clinical site and psychotropic medication use.
Other pathogens?
Although the results showed no association between strep and tics in this population, it does not “close the door completely” on a potential relationship, said Dr. Schrag.
“By and large, the development of tics in children is not associated with group A strep, but differences in small subgroups can never be excluded by a study like this.”
Participants in this study were part of the European Multicentre Tics in Children Studies (EMTICS), a prospective cohort study exploring the role of environmental and genetic factors in pediatric CTD. That project is also looking at immune system factors, “which might play a role in the development of chronic tic disorder and associated conditions,” said Dr. Schrag.
It’s still possible, she added, that other pathogens could play a role in tic development. “That’s going to be the subject of further analysis and future studies,” she said.
Tamara Pringsheim, MD, professor of clinical neurosciences, psychiatry, pediatrics, and community health sciences, University of Calgary (Alta.), praised the research.
“This was a well-designed study, with a large sample of 260 children followed for up to 4 years, using a standardized protocol to assess for group A streptococcal infection and new onset of tics.”
The study, which did not uncover an association between GAS exposure and tic onset, “provides high level evidence that group A streptococcal exposure is not an important risk factor for the new onset of tics in children with a family history of tic disorders.”
The study received funding from the European Union Seventh Framework Program for research technological development and demonstration. Dr. Schrag reports receiving consultancy or advisory board honoraria from Biogen, Abbvie, Bial, and Neurotechnology; research support from the National Institute of Health Research, Parkinsons UK, and the Economic and Social Research Council and the European Commission; and Royalties from Oxford University Press. Dr. Pringsheim reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Group A streptococcus (GAS) infection is not associated with new-onset tic disorders in at-risk children, findings from a large prospective study show.
The results mean that if preteens present with a new-onset tic condition, “they’re unlikely to have it as a result of a group A streptococcal throat infection,” study author Anette Eleonore Schrag, MD, PhD, professor, department of clinical neuroscience, Institute of Neurology, University College London, told this news organization.
Therefore, clinicians should not automatically prescribe antibiotics for children with tics, which sometimes occurs, said Dr. Schrag.
The study was published online Feb. 2 in Neurology.
Ongoing controversy
Research shows that genetic and environmental factors contribute to chronic tic disorders (CTDs) and Tourette syndrome (TS). Prenatal exposure to maternal smoking and central nervous system (CNS) stimulants, as well as psychosocial stress, may play a role.
There has been an ongoing controversy regarding the possible role of GAS in tics, with some studies showing an association and others not showing a link. However, previous studies have been retrospective, registry based, or had limited sample size.
This new prospective study is the first in children without a tic disorder but who were at relatively high risk of developing one. The children were followed to assess development of streptococcal infections and tics, said Dr. Schrag.
The study included 259 children aged 3-10 years (mean baseline age, 6.8 years; over half female) who had a first-degree relative such as a parent or sibling with TS or CTD.
The average age at TS onset is 7 years, peaking in prevalence and severity at about 9-12 years. GAS throat infections are common in this age group.
Although study participants did not have tics themselves, they represented “an enriched group,” said Dr. Schrag. “Because they had family history, we knew they were at increased risk for developing tics.”
Participants were evaluated every 2 months, alternating between scheduled hospital visits and telephone interviews. Parents kept a weekly diary and were instructed to bring their child in for assessment if they showed any signs of tics.
The average follow-up period was 1.6 years, but some of the children were followed for up to 48 months. During the study, there were a total of 1,944 assessments, including 939 telephone interviews and 1,005 clinical visits.
More common in boys
Investigators defined tic onset as the first occurrence of any sudden, rapid, recurrent, nonrhythmic involuntary movement and/or vocalization on at least three separate days within a period of 3 weeks.
The investigators assessed GAS exposure using parameters from throat swabs, serum anti-streptolysin O titers, and anti-DNAse B titers.
They used multiple definitions and combinations of GAS exposures “to make sure we weren’t missing any association because we didn’t use the right definition,” said Dr. Schrag. She explained a definitive strep infection is not always clear-cut.
At baseline, 17.0% participants tested positive for GAS, and 78.8% tested negative. No throat swab was available from 4.2% of participants.
During follow-up, the number of confirmed positive GAS exposures was 59, 102, 125, and 138, depending on the definition.
Researchers identified 61 tic cases during the study period. There was no evidence of an association of tic onset with GAS exposure after adjusting for age, sex, and parental education level.
However, there was a strong association between tic onset and sex, with girls being 60% less likely to develop tics than boys (hazard ratio, 0.4; 95% CI, 0.2-0.7; P < .01).
This result wasn’t particularly surprising, as it’s known that more boys develop tics than girls. “We just confirmed that in a prospective way,” said Dr. Schrag.
Results from sensitivity analyses confirmed the results. This was also the case with analyses that excluded visits with missing data on GAS exposure and that further adjusted for clinical site and psychotropic medication use.
Other pathogens?
Although the results showed no association between strep and tics in this population, it does not “close the door completely” on a potential relationship, said Dr. Schrag.
“By and large, the development of tics in children is not associated with group A strep, but differences in small subgroups can never be excluded by a study like this.”
Participants in this study were part of the European Multicentre Tics in Children Studies (EMTICS), a prospective cohort study exploring the role of environmental and genetic factors in pediatric CTD. That project is also looking at immune system factors, “which might play a role in the development of chronic tic disorder and associated conditions,” said Dr. Schrag.
It’s still possible, she added, that other pathogens could play a role in tic development. “That’s going to be the subject of further analysis and future studies,” she said.
Tamara Pringsheim, MD, professor of clinical neurosciences, psychiatry, pediatrics, and community health sciences, University of Calgary (Alta.), praised the research.
“This was a well-designed study, with a large sample of 260 children followed for up to 4 years, using a standardized protocol to assess for group A streptococcal infection and new onset of tics.”
The study, which did not uncover an association between GAS exposure and tic onset, “provides high level evidence that group A streptococcal exposure is not an important risk factor for the new onset of tics in children with a family history of tic disorders.”
The study received funding from the European Union Seventh Framework Program for research technological development and demonstration. Dr. Schrag reports receiving consultancy or advisory board honoraria from Biogen, Abbvie, Bial, and Neurotechnology; research support from the National Institute of Health Research, Parkinsons UK, and the Economic and Social Research Council and the European Commission; and Royalties from Oxford University Press. Dr. Pringsheim reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID vaccines open rifts between parents, children
The picture of rebellious teenagers sneaking “shots” has widened beyond breaking into Mom and Dad’s liquor cabinet. For some teens now, it means getting a COVID-19 vaccination without their parents’ consent – and, unlike the cabinet raids for the booze, they have adults willing to endorse the practice.
Since the U.S. Food and Drug Administration first granted emergency use authorization to Pfizer’s COVID-19 vaccine for teenagers in mid-2021, health officials have had to deal with a small subset of vaccine hesitancy where minors want the shot over the objections of their reluctant parents. The split has buoyed groups that were formed initially to convince teenagers to get vaccinated against other diseases.
When 14-year-old Arin Parsa of San Jose, California founded Teens for Vaccines in 2019 after a measles outbreak among unvaccinated children, “hardly anyone was interested,” he said. “Many teens were into climate change and other causes. Then, when the pandemic hit, so many were suddenly aware.”
Heavy toll on teens
Mr. Parsa’s parents fully supported Teens for Vaccines, he said, but he quickly found out how “politicized” COVID shots had become.
“We find people who are sad, angry, and frustrated at this stage of the pandemic,” he told this news organization. “The anti-vax lobby is riding the coat-tails of other movements. It has a very severe effect on their mental health. They can’t go out with their friends and socialize.”
In the pandemic’s initial stages, children were less likely to fall sick with COVID, but the Omicron variant led to a dramatic increase in illnesses among young people. The American Academy of Pediatrics has found that 3.5 million of the 11.4 million pediatric cases of the virus in the United States were reported in January 2022 alone. Meanwhile, vaccination rates for children aged 12-17, which were only 34% in June 2021 and lagged through the fall, are now at about 61% thanks to a sharp uptick during the Omicron surge, according to polling by the Kaiser Family Foundation.
No statistics are available on how many minors have received a COVID vaccine against their parents’ wishes.
“It’s not like there’s a big movement,” said Arthur Caplan, PhD, who heads the Division of Medical Ethics at the NYU Grossman School of Medicine. He said he noticed a divide around the HPV and hepatitis B vaccines. “They were tied up with sexual behavior,” he said, but “there were also some kids whose parents were really antivaxxers.”
Mr. Parsa said his and similar teen-oriented groups, such as VaxTeen, seek to educate their teen cohort, convince family members of the vaccines’ benefits, and to connect them with resources to get a shot. They also strive to change laws to make it easier for teenagers to receive the vaccine.
Consent laws vary from state to state (and within states), and proposed changes are afoot – some to loosen the laws and some to tighten them. Currently a 14-year-old in Alabama may get a COVID shot without parental permission, according to VaxTeen. In California, minors may receive the HPV shot without parental consent but not a COVID vaccine, although groups like Teens for Vaccines are pushing to change that. A bill now before the state legislature, the Teens Choose Vaccines Act (Senate Bill 866), would allow adolescents aged 12 and older to be able receive any FDA-approved vaccine – including COVID vaccines – without parental consent.
A second bill in California, the Keep Schools Open and Safe Act, would add the COVID-19 vaccines to the required list of immunizations needed to attend school in the state as well as eliminate the “personal belief” exemption against immunization.
California Sen. Richard Pan, MD (D-6th District), cowrote both bills with fellow Democrat Sen. Scott Wiener (D-11th District) and teen advocates from Teens for Vaccines and Generation Up, who helped draft the language in consultation with the lawmakers.
“As a pediatrician, I have seen all manner of situations where the requirement for a signed form has prevented teens from being able to get a vaccine that otherwise they and their guardians approved of them getting,” Dr. Pan told this news organization. “As a father, I don’t want to see my kids or any teen that wishes to protect themselves from deadly diseases unable to do so, particularly as we continue to fight off the dangers of the COVID-19 pandemic. I always encourage parents or teens that have questions about vaccines to speak directly with their pediatrician.”
Lawmakers in Philadelphia passed a provision last year to allow anyone age 11 or over to get the COVID vaccine without parental permission, keeping it in line with other vaccinations like hepatitis or HPV. “People from surrounding counties have come into the city, but it hasn’t been a huge rush,” says James Garrow, MPH, a spokesman for the city’s Department of Health.
Strive for collaboration, but listen to the children
Experts say the best solution is to for a doctor to meet with minors and their reluctant parents to get them on board for a COVID shot.
“Physicians are still the trusted messengers,” said Emma Olivera, MD, a pediatrician in suburban Chicago who advises groups that combat COVID misinformation.
Dr. Olivera said she often finds that internet-savvy teenagers have access to more information than older people, including their parents.
Thanks to COVID policies, office meetings are “difficult to do,” NYU’s Dr. Caplan added. In such a meeting, Dr. Caplan said he would try to convince the parents that the shots are needed for their children to stay in school or play sports. In the end, he said minors should get the shot but would also notify the parents before that happens: “My duty is to them.”
If parents take opposite stances, the pro-vaccine side is likely to prevail, even in California, said Patrick Baghdaserians, JD, a family law attorney in Pasadena. Mr. Baghdaserians said he is now representing a father who wants his teenager to get vaccinated but the mother doesn’t. “The court will fall on our side,” he predicted.
A version of this article first appeared on Medscape.com.
The picture of rebellious teenagers sneaking “shots” has widened beyond breaking into Mom and Dad’s liquor cabinet. For some teens now, it means getting a COVID-19 vaccination without their parents’ consent – and, unlike the cabinet raids for the booze, they have adults willing to endorse the practice.
Since the U.S. Food and Drug Administration first granted emergency use authorization to Pfizer’s COVID-19 vaccine for teenagers in mid-2021, health officials have had to deal with a small subset of vaccine hesitancy where minors want the shot over the objections of their reluctant parents. The split has buoyed groups that were formed initially to convince teenagers to get vaccinated against other diseases.
When 14-year-old Arin Parsa of San Jose, California founded Teens for Vaccines in 2019 after a measles outbreak among unvaccinated children, “hardly anyone was interested,” he said. “Many teens were into climate change and other causes. Then, when the pandemic hit, so many were suddenly aware.”
Heavy toll on teens
Mr. Parsa’s parents fully supported Teens for Vaccines, he said, but he quickly found out how “politicized” COVID shots had become.
“We find people who are sad, angry, and frustrated at this stage of the pandemic,” he told this news organization. “The anti-vax lobby is riding the coat-tails of other movements. It has a very severe effect on their mental health. They can’t go out with their friends and socialize.”
In the pandemic’s initial stages, children were less likely to fall sick with COVID, but the Omicron variant led to a dramatic increase in illnesses among young people. The American Academy of Pediatrics has found that 3.5 million of the 11.4 million pediatric cases of the virus in the United States were reported in January 2022 alone. Meanwhile, vaccination rates for children aged 12-17, which were only 34% in June 2021 and lagged through the fall, are now at about 61% thanks to a sharp uptick during the Omicron surge, according to polling by the Kaiser Family Foundation.
No statistics are available on how many minors have received a COVID vaccine against their parents’ wishes.
“It’s not like there’s a big movement,” said Arthur Caplan, PhD, who heads the Division of Medical Ethics at the NYU Grossman School of Medicine. He said he noticed a divide around the HPV and hepatitis B vaccines. “They were tied up with sexual behavior,” he said, but “there were also some kids whose parents were really antivaxxers.”
Mr. Parsa said his and similar teen-oriented groups, such as VaxTeen, seek to educate their teen cohort, convince family members of the vaccines’ benefits, and to connect them with resources to get a shot. They also strive to change laws to make it easier for teenagers to receive the vaccine.
Consent laws vary from state to state (and within states), and proposed changes are afoot – some to loosen the laws and some to tighten them. Currently a 14-year-old in Alabama may get a COVID shot without parental permission, according to VaxTeen. In California, minors may receive the HPV shot without parental consent but not a COVID vaccine, although groups like Teens for Vaccines are pushing to change that. A bill now before the state legislature, the Teens Choose Vaccines Act (Senate Bill 866), would allow adolescents aged 12 and older to be able receive any FDA-approved vaccine – including COVID vaccines – without parental consent.
A second bill in California, the Keep Schools Open and Safe Act, would add the COVID-19 vaccines to the required list of immunizations needed to attend school in the state as well as eliminate the “personal belief” exemption against immunization.
California Sen. Richard Pan, MD (D-6th District), cowrote both bills with fellow Democrat Sen. Scott Wiener (D-11th District) and teen advocates from Teens for Vaccines and Generation Up, who helped draft the language in consultation with the lawmakers.
“As a pediatrician, I have seen all manner of situations where the requirement for a signed form has prevented teens from being able to get a vaccine that otherwise they and their guardians approved of them getting,” Dr. Pan told this news organization. “As a father, I don’t want to see my kids or any teen that wishes to protect themselves from deadly diseases unable to do so, particularly as we continue to fight off the dangers of the COVID-19 pandemic. I always encourage parents or teens that have questions about vaccines to speak directly with their pediatrician.”
Lawmakers in Philadelphia passed a provision last year to allow anyone age 11 or over to get the COVID vaccine without parental permission, keeping it in line with other vaccinations like hepatitis or HPV. “People from surrounding counties have come into the city, but it hasn’t been a huge rush,” says James Garrow, MPH, a spokesman for the city’s Department of Health.
Strive for collaboration, but listen to the children
Experts say the best solution is to for a doctor to meet with minors and their reluctant parents to get them on board for a COVID shot.
“Physicians are still the trusted messengers,” said Emma Olivera, MD, a pediatrician in suburban Chicago who advises groups that combat COVID misinformation.
Dr. Olivera said she often finds that internet-savvy teenagers have access to more information than older people, including their parents.
Thanks to COVID policies, office meetings are “difficult to do,” NYU’s Dr. Caplan added. In such a meeting, Dr. Caplan said he would try to convince the parents that the shots are needed for their children to stay in school or play sports. In the end, he said minors should get the shot but would also notify the parents before that happens: “My duty is to them.”
If parents take opposite stances, the pro-vaccine side is likely to prevail, even in California, said Patrick Baghdaserians, JD, a family law attorney in Pasadena. Mr. Baghdaserians said he is now representing a father who wants his teenager to get vaccinated but the mother doesn’t. “The court will fall on our side,” he predicted.
A version of this article first appeared on Medscape.com.
The picture of rebellious teenagers sneaking “shots” has widened beyond breaking into Mom and Dad’s liquor cabinet. For some teens now, it means getting a COVID-19 vaccination without their parents’ consent – and, unlike the cabinet raids for the booze, they have adults willing to endorse the practice.
Since the U.S. Food and Drug Administration first granted emergency use authorization to Pfizer’s COVID-19 vaccine for teenagers in mid-2021, health officials have had to deal with a small subset of vaccine hesitancy where minors want the shot over the objections of their reluctant parents. The split has buoyed groups that were formed initially to convince teenagers to get vaccinated against other diseases.
When 14-year-old Arin Parsa of San Jose, California founded Teens for Vaccines in 2019 after a measles outbreak among unvaccinated children, “hardly anyone was interested,” he said. “Many teens were into climate change and other causes. Then, when the pandemic hit, so many were suddenly aware.”
Heavy toll on teens
Mr. Parsa’s parents fully supported Teens for Vaccines, he said, but he quickly found out how “politicized” COVID shots had become.
“We find people who are sad, angry, and frustrated at this stage of the pandemic,” he told this news organization. “The anti-vax lobby is riding the coat-tails of other movements. It has a very severe effect on their mental health. They can’t go out with their friends and socialize.”
In the pandemic’s initial stages, children were less likely to fall sick with COVID, but the Omicron variant led to a dramatic increase in illnesses among young people. The American Academy of Pediatrics has found that 3.5 million of the 11.4 million pediatric cases of the virus in the United States were reported in January 2022 alone. Meanwhile, vaccination rates for children aged 12-17, which were only 34% in June 2021 and lagged through the fall, are now at about 61% thanks to a sharp uptick during the Omicron surge, according to polling by the Kaiser Family Foundation.
No statistics are available on how many minors have received a COVID vaccine against their parents’ wishes.
“It’s not like there’s a big movement,” said Arthur Caplan, PhD, who heads the Division of Medical Ethics at the NYU Grossman School of Medicine. He said he noticed a divide around the HPV and hepatitis B vaccines. “They were tied up with sexual behavior,” he said, but “there were also some kids whose parents were really antivaxxers.”
Mr. Parsa said his and similar teen-oriented groups, such as VaxTeen, seek to educate their teen cohort, convince family members of the vaccines’ benefits, and to connect them with resources to get a shot. They also strive to change laws to make it easier for teenagers to receive the vaccine.
Consent laws vary from state to state (and within states), and proposed changes are afoot – some to loosen the laws and some to tighten them. Currently a 14-year-old in Alabama may get a COVID shot without parental permission, according to VaxTeen. In California, minors may receive the HPV shot without parental consent but not a COVID vaccine, although groups like Teens for Vaccines are pushing to change that. A bill now before the state legislature, the Teens Choose Vaccines Act (Senate Bill 866), would allow adolescents aged 12 and older to be able receive any FDA-approved vaccine – including COVID vaccines – without parental consent.
A second bill in California, the Keep Schools Open and Safe Act, would add the COVID-19 vaccines to the required list of immunizations needed to attend school in the state as well as eliminate the “personal belief” exemption against immunization.
California Sen. Richard Pan, MD (D-6th District), cowrote both bills with fellow Democrat Sen. Scott Wiener (D-11th District) and teen advocates from Teens for Vaccines and Generation Up, who helped draft the language in consultation with the lawmakers.
“As a pediatrician, I have seen all manner of situations where the requirement for a signed form has prevented teens from being able to get a vaccine that otherwise they and their guardians approved of them getting,” Dr. Pan told this news organization. “As a father, I don’t want to see my kids or any teen that wishes to protect themselves from deadly diseases unable to do so, particularly as we continue to fight off the dangers of the COVID-19 pandemic. I always encourage parents or teens that have questions about vaccines to speak directly with their pediatrician.”
Lawmakers in Philadelphia passed a provision last year to allow anyone age 11 or over to get the COVID vaccine without parental permission, keeping it in line with other vaccinations like hepatitis or HPV. “People from surrounding counties have come into the city, but it hasn’t been a huge rush,” says James Garrow, MPH, a spokesman for the city’s Department of Health.
Strive for collaboration, but listen to the children
Experts say the best solution is to for a doctor to meet with minors and their reluctant parents to get them on board for a COVID shot.
“Physicians are still the trusted messengers,” said Emma Olivera, MD, a pediatrician in suburban Chicago who advises groups that combat COVID misinformation.
Dr. Olivera said she often finds that internet-savvy teenagers have access to more information than older people, including their parents.
Thanks to COVID policies, office meetings are “difficult to do,” NYU’s Dr. Caplan added. In such a meeting, Dr. Caplan said he would try to convince the parents that the shots are needed for their children to stay in school or play sports. In the end, he said minors should get the shot but would also notify the parents before that happens: “My duty is to them.”
If parents take opposite stances, the pro-vaccine side is likely to prevail, even in California, said Patrick Baghdaserians, JD, a family law attorney in Pasadena. Mr. Baghdaserians said he is now representing a father who wants his teenager to get vaccinated but the mother doesn’t. “The court will fall on our side,” he predicted.
A version of this article first appeared on Medscape.com.
Omicron death rate higher than during Delta surge
With the Omicron variant now accounting for almost 100% of COVID-19 cases in the United States, the Washington Post reported.
That’s higher than the approximately 2,000 daily deaths in fall 2021 during the Delta surge, but less than the 3,000 daily deaths in January 2021, when COVID vaccines were not widely available, the Post’s data analysis said.
The Omicron variant generally causes less severe disease than other strains of COVID, but because it is so transmissible, Omicron is infecting higher raw numbers of people that previous strains.
“Even if on a per-case basis fewer people develop severe illness and die, when you apply a small percentage to a very large number, you get a substantial number,” Jennifer Nuzzo, DrPH, an epidemiologist at the Johns Hopkins University, Baltimore, told the Post.
The unvaccinated, people over 75, and people with underlying medical conditions are the groups most endangered by Omicron, the Post said. About half of the deaths in January 2022 were among people over 75, compared with about a third in September 2021 during the Delta surge.
The age trend is seen in Florida, said Jason Salemi, PhD, an epidemiologist at the University of South Florida, Tampa. He told the Post that seniors accounted for about 85% of deaths in the winter of 2020-2021, about 60% during the Delta surge, and about 80% now during the Omicron surge.
The uptick in senior deaths may have occurred because seniors who got vaccinated in early 2021 didn’t get boosted ahead of the Omicron surge, he said.
“Omicron may be less severe for younger people, but it will still find vulnerable seniors in our community,” Dr. Salemi said. “That vaccination back in February isn’t as effective now if you aren’t boosted.”
CDC data shows that 95% of people in the United States over 65 have gotten at least one dose of vaccine, 88.5% are fully vaccinated, but only 62.5% have gotten a booster dose.
The COVID death rate is highest in the Midwest. During the last 2 months, Chicago reported more than 1,000 COVID deaths, almost as much as the December 2020 peak, The Post said. Minorities have been hit hard. About third of the city’s population is Black but about half the COVID victims are Black, the Post said.
“It’s been challenging because it goes up against the national narrative that omicron is nothing dangerous,” said Allison Arwady, commissioner of the Chicago Department of Public Health.
In a Feb. 9 news briefing at the White House, CDC Director Rochelle Walensky, MD, provided slightly different statistics on COVID-related deaths. She said that the 7-day average of daily deaths was about 2,400, up 3% from the previous week.
The 7-day daily average of cases is about 247,300 cases per day, down 44% from the previous week, she said. Hospital admissions are about 13,000 daily, down 25% from the previous week.
Dr. Walensky said the Omicron variant now accounts for almost 100% of COVID viruses circulating in the United States.
A version of this article first appeared on WebMD.com.
With the Omicron variant now accounting for almost 100% of COVID-19 cases in the United States, the Washington Post reported.
That’s higher than the approximately 2,000 daily deaths in fall 2021 during the Delta surge, but less than the 3,000 daily deaths in January 2021, when COVID vaccines were not widely available, the Post’s data analysis said.
The Omicron variant generally causes less severe disease than other strains of COVID, but because it is so transmissible, Omicron is infecting higher raw numbers of people that previous strains.
“Even if on a per-case basis fewer people develop severe illness and die, when you apply a small percentage to a very large number, you get a substantial number,” Jennifer Nuzzo, DrPH, an epidemiologist at the Johns Hopkins University, Baltimore, told the Post.
The unvaccinated, people over 75, and people with underlying medical conditions are the groups most endangered by Omicron, the Post said. About half of the deaths in January 2022 were among people over 75, compared with about a third in September 2021 during the Delta surge.
The age trend is seen in Florida, said Jason Salemi, PhD, an epidemiologist at the University of South Florida, Tampa. He told the Post that seniors accounted for about 85% of deaths in the winter of 2020-2021, about 60% during the Delta surge, and about 80% now during the Omicron surge.
The uptick in senior deaths may have occurred because seniors who got vaccinated in early 2021 didn’t get boosted ahead of the Omicron surge, he said.
“Omicron may be less severe for younger people, but it will still find vulnerable seniors in our community,” Dr. Salemi said. “That vaccination back in February isn’t as effective now if you aren’t boosted.”
CDC data shows that 95% of people in the United States over 65 have gotten at least one dose of vaccine, 88.5% are fully vaccinated, but only 62.5% have gotten a booster dose.
The COVID death rate is highest in the Midwest. During the last 2 months, Chicago reported more than 1,000 COVID deaths, almost as much as the December 2020 peak, The Post said. Minorities have been hit hard. About third of the city’s population is Black but about half the COVID victims are Black, the Post said.
“It’s been challenging because it goes up against the national narrative that omicron is nothing dangerous,” said Allison Arwady, commissioner of the Chicago Department of Public Health.
In a Feb. 9 news briefing at the White House, CDC Director Rochelle Walensky, MD, provided slightly different statistics on COVID-related deaths. She said that the 7-day average of daily deaths was about 2,400, up 3% from the previous week.
The 7-day daily average of cases is about 247,300 cases per day, down 44% from the previous week, she said. Hospital admissions are about 13,000 daily, down 25% from the previous week.
Dr. Walensky said the Omicron variant now accounts for almost 100% of COVID viruses circulating in the United States.
A version of this article first appeared on WebMD.com.
With the Omicron variant now accounting for almost 100% of COVID-19 cases in the United States, the Washington Post reported.
That’s higher than the approximately 2,000 daily deaths in fall 2021 during the Delta surge, but less than the 3,000 daily deaths in January 2021, when COVID vaccines were not widely available, the Post’s data analysis said.
The Omicron variant generally causes less severe disease than other strains of COVID, but because it is so transmissible, Omicron is infecting higher raw numbers of people that previous strains.
“Even if on a per-case basis fewer people develop severe illness and die, when you apply a small percentage to a very large number, you get a substantial number,” Jennifer Nuzzo, DrPH, an epidemiologist at the Johns Hopkins University, Baltimore, told the Post.
The unvaccinated, people over 75, and people with underlying medical conditions are the groups most endangered by Omicron, the Post said. About half of the deaths in January 2022 were among people over 75, compared with about a third in September 2021 during the Delta surge.
The age trend is seen in Florida, said Jason Salemi, PhD, an epidemiologist at the University of South Florida, Tampa. He told the Post that seniors accounted for about 85% of deaths in the winter of 2020-2021, about 60% during the Delta surge, and about 80% now during the Omicron surge.
The uptick in senior deaths may have occurred because seniors who got vaccinated in early 2021 didn’t get boosted ahead of the Omicron surge, he said.
“Omicron may be less severe for younger people, but it will still find vulnerable seniors in our community,” Dr. Salemi said. “That vaccination back in February isn’t as effective now if you aren’t boosted.”
CDC data shows that 95% of people in the United States over 65 have gotten at least one dose of vaccine, 88.5% are fully vaccinated, but only 62.5% have gotten a booster dose.
The COVID death rate is highest in the Midwest. During the last 2 months, Chicago reported more than 1,000 COVID deaths, almost as much as the December 2020 peak, The Post said. Minorities have been hit hard. About third of the city’s population is Black but about half the COVID victims are Black, the Post said.
“It’s been challenging because it goes up against the national narrative that omicron is nothing dangerous,” said Allison Arwady, commissioner of the Chicago Department of Public Health.
In a Feb. 9 news briefing at the White House, CDC Director Rochelle Walensky, MD, provided slightly different statistics on COVID-related deaths. She said that the 7-day average of daily deaths was about 2,400, up 3% from the previous week.
The 7-day daily average of cases is about 247,300 cases per day, down 44% from the previous week, she said. Hospital admissions are about 13,000 daily, down 25% from the previous week.
Dr. Walensky said the Omicron variant now accounts for almost 100% of COVID viruses circulating in the United States.
A version of this article first appeared on WebMD.com.

