User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
'The whitest specialty,' revisited
A recent STAT article by Usha Lee McFarling identified orthopedic surgery as “the whitest specialty.” That’s a problem many, perhaps most, orthopedic surgeons are aware of. But seeing it stated so bluntly is jolting. It’s disconcerting to think that the orthopedic community is making so little progress toward achieving the principal ideal articulated in our country’s fundamental declaration of moral values: that all people are created equal and that they have inalienable rights – in our case, that everyone, Black, brown, as well as White, has the right to the same high level of medical care.
Unfortunately, as study after study has shown, minorities do not enjoy the right to equitable care. Instead, they are subject to disparities in treatment and outcomes that speak to the prejudices that are built into the health care system and are present – sometimes consciously, but most often subconsciously – in the minds of physicians. One important contributing element to these disparities is the paucity of minority practitioners. Studies have also shown that Black patients, for example, respond better to Black physicians, who so often share a psychological and cultural sympathy unavailable to most White physicians. It’s for that reason that being identified as “the whitest specialty” is so immensely troubling.
In researching her STAT article, Ms. McFarling spoke with American Academy of Orthopaedic Surgeons leaders, practicing surgeons, residents, and med students about the dearth of minority and female orthopedic surgeons. What she heard was perplexity and frustration about why better progress hasn’t been made toward correcting the gross underrepresentation of everyone other than White men. The AAOS, she noted, was one of the first specialties to recognize the lack of diversity and over the years has put in great effort to address the problem, creating task forces, committees, and diversity awards and sponsoring conferences and discussions. Yet progress has been glacial, at best.
From her respondents, Ms. McFarling heard an array of reasons for this. Black, Hispanic, and Native American persons are underrepresented in medical schools, so the pool of potential applicants for orthopedic residencies is shallow to begin with. STEM studies are notoriously inadequate in poorer primary and secondary schools, in which so many minority students are educated. The MCAT and USMLE Step 1 test, which play a role in acceptance to residencies, have been shown to be biased. The specialty has few Black or brown role models and, consequently, few advocates and a lack of mentorship. Overt bias may be fairly rare (though microaggressions are still a common and ongoing problem), but most minority and female orthopedic surgeons feel strongly that implicit or subconscious bias is entrenched and works against acceptance to residencies, success in residencies, and advancement in the field.
One of this article’s authors (AW) saw all these factors at work as a resident, then as an admissions committee member at both Yale and Harvard. But the fact is that other medical specialties face exactly these problems and barriers, and yet have been substantially more successful in overcoming them.
What seems to be distinctive about orthopedics is that the mindset which perpetuated (and still perpetuates) In this regard, Kristy Weber, MD, the first female president of the AAOS, told Ms. McFarling that the critical first step to bringing in more women or people of color is changing the culture. There seems to be a consensus about that.
So, what does that mean, given that the AAOS has made serious efforts in that regard that have clearly been less than effective?
The answer, as we see it, is first – to not give in to frustration. The time frames involved in changing customary states of mind are typically elongated, and the deeper the habituation, the longer transformation takes. Deep changes always mean a long, hard slog. For transformations of this sort to take place, the requirements are a general agreement on the value of the transformation, exposure to the destructive consequences of the customary modus operandi, and persuasion for why change needs to happen.
In orthopedics, the first requirement has been met. The AAOS espouses diversity and inclusion as a high-level value. In terms of the second two requirements – exposure and persuasion – orthopedic surgeons have been witness to events, campaigns, conferences, et cetera. But these have not been enough, which means that efforts need to be focused, enlarged, sustained, determined, and innovative.
Does the orthopedic surgery community have the ability to do that?
The answer is: Yes, it does.
Currently the orthopedic surgeon community boasts a number of organizations, groups, and individuals pushing for change, in addition to the AAOS’s Diversity Advisory Board. The predominantly African American J. Robert Gladden Orthopaedic Society, the Ruth Jackson Orthopaedic Society of female orthopedic physicians, and the Association of Latino Orthopaedic Surgeons are all energetic advocates, as is Nth Dimensions, the Perry Initiative, and various ad hoc and individual endeavors.
These are all strong proponents for their own groups in their own way. But history has shown in so many cases that concerted rather than individual action empowers advocacy, and what orthopedic surgery needs in its current situation of gross underrepresentation of minorities and women is an enhanced campaign to raise awareness and redouble persuasion.
One of many examples of the power of collective action is the Association of Minority Health Professions Schools founded by Dr. Louis Sullivan in 1977.* Dr. Sullivan (later secretary of the Department of Health & Human Services) was at that time the founding dean of Morehouse School of Medicine. Morehouse had been launched on a shoestring and needed funding urgently. Other Black health schools, such as Meharry Medical College and Tuskegee College of Veterinary Medicine, were in even more pressing financial need. The coalition of schools that Dr. Sullivan organized became a powerful force in Congress and the National Institutes of Health, magnitudes more effective in raising funds from government and other sources than the best individual efforts of the separate institutions.
Dr. Sullivan’s association is only one of a multitude of historical examples of the effectiveness of unified action. AAOS currently has no single officer charged with bringing together the efforts of the change assets that already exist. It could, perhaps should, have someone in that position. AAOS could invest that same office with a mandate to survey the other medical specialties and bring to bear the most effective diversity, equity, and inclusion (DEI) practices in their arsenals.
Finally, despite the attention AAOS has brought to DEI needs, a look at the organization’s strategic goals, its core values, and its “key enablers” finds not a single mention of diversity or inclusion. Given the country’s current focus on the need for equality, given the poor performance of the orthopedic surgery specialty in terms of inclusion, the obvious question is: Should there not be an official declaration positing diversity as a primary AAOS desideratum?
There is recent precedent for this in the American College of Physicians/American Board of Internal Medicine’s Physician Charter on Professionalism, which includes “social justice” as a primary goal of medical practice. This highlights and reinforces the humanitarian strivings of the profession. In light of the paralysis illuminated by Ms. McFarling’s STAT article, a clear, concise declaration by the AAOS of the value and need for DEI as a central component of the organization’s values should be high on the AAOS order of business. A commitment in that form would serve as a powerful catalyst for bringing orthopedic surgery into step with its sister specialties, as well as affirming the core egalitarian principle that underlies all of medical care.
Dr. White is the Ellen and Melvin Gordon Distinguished Professor of Medical Education and Professor of Orthopedic Surgery at Harvard Medical School, Boston. Dr. Chanoff is a founding board member of the Augustus A. White III Institute for Healthcare Equity. Neither Dr. White nor Dr. Chanoff reported any conflicts of interest. A version of this article first appeared on Medscape.com.
Correction, 2/1/22: An earlier version of this article omitted the title of "Dr." before Dr. Louis Sullivan's name.
A recent STAT article by Usha Lee McFarling identified orthopedic surgery as “the whitest specialty.” That’s a problem many, perhaps most, orthopedic surgeons are aware of. But seeing it stated so bluntly is jolting. It’s disconcerting to think that the orthopedic community is making so little progress toward achieving the principal ideal articulated in our country’s fundamental declaration of moral values: that all people are created equal and that they have inalienable rights – in our case, that everyone, Black, brown, as well as White, has the right to the same high level of medical care.
Unfortunately, as study after study has shown, minorities do not enjoy the right to equitable care. Instead, they are subject to disparities in treatment and outcomes that speak to the prejudices that are built into the health care system and are present – sometimes consciously, but most often subconsciously – in the minds of physicians. One important contributing element to these disparities is the paucity of minority practitioners. Studies have also shown that Black patients, for example, respond better to Black physicians, who so often share a psychological and cultural sympathy unavailable to most White physicians. It’s for that reason that being identified as “the whitest specialty” is so immensely troubling.
In researching her STAT article, Ms. McFarling spoke with American Academy of Orthopaedic Surgeons leaders, practicing surgeons, residents, and med students about the dearth of minority and female orthopedic surgeons. What she heard was perplexity and frustration about why better progress hasn’t been made toward correcting the gross underrepresentation of everyone other than White men. The AAOS, she noted, was one of the first specialties to recognize the lack of diversity and over the years has put in great effort to address the problem, creating task forces, committees, and diversity awards and sponsoring conferences and discussions. Yet progress has been glacial, at best.
From her respondents, Ms. McFarling heard an array of reasons for this. Black, Hispanic, and Native American persons are underrepresented in medical schools, so the pool of potential applicants for orthopedic residencies is shallow to begin with. STEM studies are notoriously inadequate in poorer primary and secondary schools, in which so many minority students are educated. The MCAT and USMLE Step 1 test, which play a role in acceptance to residencies, have been shown to be biased. The specialty has few Black or brown role models and, consequently, few advocates and a lack of mentorship. Overt bias may be fairly rare (though microaggressions are still a common and ongoing problem), but most minority and female orthopedic surgeons feel strongly that implicit or subconscious bias is entrenched and works against acceptance to residencies, success in residencies, and advancement in the field.
One of this article’s authors (AW) saw all these factors at work as a resident, then as an admissions committee member at both Yale and Harvard. But the fact is that other medical specialties face exactly these problems and barriers, and yet have been substantially more successful in overcoming them.
What seems to be distinctive about orthopedics is that the mindset which perpetuated (and still perpetuates) In this regard, Kristy Weber, MD, the first female president of the AAOS, told Ms. McFarling that the critical first step to bringing in more women or people of color is changing the culture. There seems to be a consensus about that.
So, what does that mean, given that the AAOS has made serious efforts in that regard that have clearly been less than effective?
The answer, as we see it, is first – to not give in to frustration. The time frames involved in changing customary states of mind are typically elongated, and the deeper the habituation, the longer transformation takes. Deep changes always mean a long, hard slog. For transformations of this sort to take place, the requirements are a general agreement on the value of the transformation, exposure to the destructive consequences of the customary modus operandi, and persuasion for why change needs to happen.
In orthopedics, the first requirement has been met. The AAOS espouses diversity and inclusion as a high-level value. In terms of the second two requirements – exposure and persuasion – orthopedic surgeons have been witness to events, campaigns, conferences, et cetera. But these have not been enough, which means that efforts need to be focused, enlarged, sustained, determined, and innovative.
Does the orthopedic surgery community have the ability to do that?
The answer is: Yes, it does.
Currently the orthopedic surgeon community boasts a number of organizations, groups, and individuals pushing for change, in addition to the AAOS’s Diversity Advisory Board. The predominantly African American J. Robert Gladden Orthopaedic Society, the Ruth Jackson Orthopaedic Society of female orthopedic physicians, and the Association of Latino Orthopaedic Surgeons are all energetic advocates, as is Nth Dimensions, the Perry Initiative, and various ad hoc and individual endeavors.
These are all strong proponents for their own groups in their own way. But history has shown in so many cases that concerted rather than individual action empowers advocacy, and what orthopedic surgery needs in its current situation of gross underrepresentation of minorities and women is an enhanced campaign to raise awareness and redouble persuasion.
One of many examples of the power of collective action is the Association of Minority Health Professions Schools founded by Dr. Louis Sullivan in 1977.* Dr. Sullivan (later secretary of the Department of Health & Human Services) was at that time the founding dean of Morehouse School of Medicine. Morehouse had been launched on a shoestring and needed funding urgently. Other Black health schools, such as Meharry Medical College and Tuskegee College of Veterinary Medicine, were in even more pressing financial need. The coalition of schools that Dr. Sullivan organized became a powerful force in Congress and the National Institutes of Health, magnitudes more effective in raising funds from government and other sources than the best individual efforts of the separate institutions.
Dr. Sullivan’s association is only one of a multitude of historical examples of the effectiveness of unified action. AAOS currently has no single officer charged with bringing together the efforts of the change assets that already exist. It could, perhaps should, have someone in that position. AAOS could invest that same office with a mandate to survey the other medical specialties and bring to bear the most effective diversity, equity, and inclusion (DEI) practices in their arsenals.
Finally, despite the attention AAOS has brought to DEI needs, a look at the organization’s strategic goals, its core values, and its “key enablers” finds not a single mention of diversity or inclusion. Given the country’s current focus on the need for equality, given the poor performance of the orthopedic surgery specialty in terms of inclusion, the obvious question is: Should there not be an official declaration positing diversity as a primary AAOS desideratum?
There is recent precedent for this in the American College of Physicians/American Board of Internal Medicine’s Physician Charter on Professionalism, which includes “social justice” as a primary goal of medical practice. This highlights and reinforces the humanitarian strivings of the profession. In light of the paralysis illuminated by Ms. McFarling’s STAT article, a clear, concise declaration by the AAOS of the value and need for DEI as a central component of the organization’s values should be high on the AAOS order of business. A commitment in that form would serve as a powerful catalyst for bringing orthopedic surgery into step with its sister specialties, as well as affirming the core egalitarian principle that underlies all of medical care.
Dr. White is the Ellen and Melvin Gordon Distinguished Professor of Medical Education and Professor of Orthopedic Surgery at Harvard Medical School, Boston. Dr. Chanoff is a founding board member of the Augustus A. White III Institute for Healthcare Equity. Neither Dr. White nor Dr. Chanoff reported any conflicts of interest. A version of this article first appeared on Medscape.com.
Correction, 2/1/22: An earlier version of this article omitted the title of "Dr." before Dr. Louis Sullivan's name.
A recent STAT article by Usha Lee McFarling identified orthopedic surgery as “the whitest specialty.” That’s a problem many, perhaps most, orthopedic surgeons are aware of. But seeing it stated so bluntly is jolting. It’s disconcerting to think that the orthopedic community is making so little progress toward achieving the principal ideal articulated in our country’s fundamental declaration of moral values: that all people are created equal and that they have inalienable rights – in our case, that everyone, Black, brown, as well as White, has the right to the same high level of medical care.
Unfortunately, as study after study has shown, minorities do not enjoy the right to equitable care. Instead, they are subject to disparities in treatment and outcomes that speak to the prejudices that are built into the health care system and are present – sometimes consciously, but most often subconsciously – in the minds of physicians. One important contributing element to these disparities is the paucity of minority practitioners. Studies have also shown that Black patients, for example, respond better to Black physicians, who so often share a psychological and cultural sympathy unavailable to most White physicians. It’s for that reason that being identified as “the whitest specialty” is so immensely troubling.
In researching her STAT article, Ms. McFarling spoke with American Academy of Orthopaedic Surgeons leaders, practicing surgeons, residents, and med students about the dearth of minority and female orthopedic surgeons. What she heard was perplexity and frustration about why better progress hasn’t been made toward correcting the gross underrepresentation of everyone other than White men. The AAOS, she noted, was one of the first specialties to recognize the lack of diversity and over the years has put in great effort to address the problem, creating task forces, committees, and diversity awards and sponsoring conferences and discussions. Yet progress has been glacial, at best.
From her respondents, Ms. McFarling heard an array of reasons for this. Black, Hispanic, and Native American persons are underrepresented in medical schools, so the pool of potential applicants for orthopedic residencies is shallow to begin with. STEM studies are notoriously inadequate in poorer primary and secondary schools, in which so many minority students are educated. The MCAT and USMLE Step 1 test, which play a role in acceptance to residencies, have been shown to be biased. The specialty has few Black or brown role models and, consequently, few advocates and a lack of mentorship. Overt bias may be fairly rare (though microaggressions are still a common and ongoing problem), but most minority and female orthopedic surgeons feel strongly that implicit or subconscious bias is entrenched and works against acceptance to residencies, success in residencies, and advancement in the field.
One of this article’s authors (AW) saw all these factors at work as a resident, then as an admissions committee member at both Yale and Harvard. But the fact is that other medical specialties face exactly these problems and barriers, and yet have been substantially more successful in overcoming them.
What seems to be distinctive about orthopedics is that the mindset which perpetuated (and still perpetuates) In this regard, Kristy Weber, MD, the first female president of the AAOS, told Ms. McFarling that the critical first step to bringing in more women or people of color is changing the culture. There seems to be a consensus about that.
So, what does that mean, given that the AAOS has made serious efforts in that regard that have clearly been less than effective?
The answer, as we see it, is first – to not give in to frustration. The time frames involved in changing customary states of mind are typically elongated, and the deeper the habituation, the longer transformation takes. Deep changes always mean a long, hard slog. For transformations of this sort to take place, the requirements are a general agreement on the value of the transformation, exposure to the destructive consequences of the customary modus operandi, and persuasion for why change needs to happen.
In orthopedics, the first requirement has been met. The AAOS espouses diversity and inclusion as a high-level value. In terms of the second two requirements – exposure and persuasion – orthopedic surgeons have been witness to events, campaigns, conferences, et cetera. But these have not been enough, which means that efforts need to be focused, enlarged, sustained, determined, and innovative.
Does the orthopedic surgery community have the ability to do that?
The answer is: Yes, it does.
Currently the orthopedic surgeon community boasts a number of organizations, groups, and individuals pushing for change, in addition to the AAOS’s Diversity Advisory Board. The predominantly African American J. Robert Gladden Orthopaedic Society, the Ruth Jackson Orthopaedic Society of female orthopedic physicians, and the Association of Latino Orthopaedic Surgeons are all energetic advocates, as is Nth Dimensions, the Perry Initiative, and various ad hoc and individual endeavors.
These are all strong proponents for their own groups in their own way. But history has shown in so many cases that concerted rather than individual action empowers advocacy, and what orthopedic surgery needs in its current situation of gross underrepresentation of minorities and women is an enhanced campaign to raise awareness and redouble persuasion.
One of many examples of the power of collective action is the Association of Minority Health Professions Schools founded by Dr. Louis Sullivan in 1977.* Dr. Sullivan (later secretary of the Department of Health & Human Services) was at that time the founding dean of Morehouse School of Medicine. Morehouse had been launched on a shoestring and needed funding urgently. Other Black health schools, such as Meharry Medical College and Tuskegee College of Veterinary Medicine, were in even more pressing financial need. The coalition of schools that Dr. Sullivan organized became a powerful force in Congress and the National Institutes of Health, magnitudes more effective in raising funds from government and other sources than the best individual efforts of the separate institutions.
Dr. Sullivan’s association is only one of a multitude of historical examples of the effectiveness of unified action. AAOS currently has no single officer charged with bringing together the efforts of the change assets that already exist. It could, perhaps should, have someone in that position. AAOS could invest that same office with a mandate to survey the other medical specialties and bring to bear the most effective diversity, equity, and inclusion (DEI) practices in their arsenals.
Finally, despite the attention AAOS has brought to DEI needs, a look at the organization’s strategic goals, its core values, and its “key enablers” finds not a single mention of diversity or inclusion. Given the country’s current focus on the need for equality, given the poor performance of the orthopedic surgery specialty in terms of inclusion, the obvious question is: Should there not be an official declaration positing diversity as a primary AAOS desideratum?
There is recent precedent for this in the American College of Physicians/American Board of Internal Medicine’s Physician Charter on Professionalism, which includes “social justice” as a primary goal of medical practice. This highlights and reinforces the humanitarian strivings of the profession. In light of the paralysis illuminated by Ms. McFarling’s STAT article, a clear, concise declaration by the AAOS of the value and need for DEI as a central component of the organization’s values should be high on the AAOS order of business. A commitment in that form would serve as a powerful catalyst for bringing orthopedic surgery into step with its sister specialties, as well as affirming the core egalitarian principle that underlies all of medical care.
Dr. White is the Ellen and Melvin Gordon Distinguished Professor of Medical Education and Professor of Orthopedic Surgery at Harvard Medical School, Boston. Dr. Chanoff is a founding board member of the Augustus A. White III Institute for Healthcare Equity. Neither Dr. White nor Dr. Chanoff reported any conflicts of interest. A version of this article first appeared on Medscape.com.
Correction, 2/1/22: An earlier version of this article omitted the title of "Dr." before Dr. Louis Sullivan's name.
We’re dying to tell you about fatigability
Are you tired? Or are you death tired?
When we’re feeling that burnout monster creep in we sometimes say that we’re being worked to death or that we’re dead tired, but what if that feeling could predict when it’s your actual time to go?
In a recent study published in the Journals of Gerontology: Series A, epidemiologists from the University of Pittsburgh were able to associate a level of “physical fatigability” with mortality.
The researchers administered the Pittsburgh Fatigability Scale to almost 3,000 participants aged ≥ 60 years, who ranked from 0 to 5 on how tired they thought they would be after doing activities like light housework or a leisurely 30-minute walk. After accounting for factors such as preexisting conditions and mental health, the researchers found that people who scored 25 or more points were 2.3 times more likely to die in the next 2.7 years, compared with those who scored under 25.
So what does that tell us about the importance of being continuously active? It’s pretty important.
“Previous research indicates that getting more physical activity can reduce a person’s fatigability. Our study is the first to link more severe physical fatigability to an earlier death,” lead author Nancy W. Glynn, PhD, said in a separate statement. The best way to keep physically active, she suggested, is to set manageable goals and a routine.
A nice walk around the neighborhood during golden hour or a little bit of yoga before breakfast could be a great way to keep the body moving, because you know what they say: Use it or lose it.
This work is NFT protected: Do not screenshot
If you’ve been following the nonmedical news, you’ve likely heard the term “NFT” explode in the past few months. Standing for nonfungible token, NFTs are, at least theoretically, a proof of ownership for digital creations that prevents anyone other than the buyer from reselling the artwork. Sounds like a great idea: It protects artists and buyers alike.
Much like its cousin cryptocurrency, however, the NFT world is rife with speculation, scams, misunderstanding, and drawings of bored monkeys. It’s the Wild West out there in the digital art universe: One poor unfortunate accidentally sold a $300k NFT image for $3,000, a group of investors spent $3 million buying an NFT for a rare version of Dune believing it gave them the copyright (it did not), and an Indonesian engineering student’s 5-year series of expressionless selfies is now worth a million dollars.
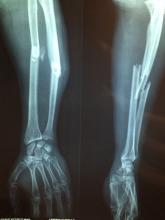
This is a column detailing weird medical news, however, so with our setup complete (though our understanding of NFTs is very much not), we move to France and meet our hero (?), Emmanuel Masmejean, an orthopedic surgeon who apparently wasn’t making enough money in his lucrative medical career.
In a move of apocalyptic madness, he threw ethics out the window, delved into his archive, and found an x-ray of a young woman with a bullet lodged in her arm. The woman was a survivor of the Bataclan mass shooting and bombing in 2015, and don’t you worry, our intrepid entrepreneur made sure to identify her as such when he tried selling the x-ray as an NFT on an online art website for $2,776. Yes, this is very much a violation of doctor-patient confidentiality, and no, that’s not a lot of money to risk your medical career on.
Naturally, the woman was horrified and shocked to learn that the image was being sold, her lawyer told the Guardian. When the doctor called her, he merely attempted to justify his action, rather than apologizing or showing any remorse. Dr. Masmejean is now facing legal action and a disciplinary charge for his attempted entry into the NFT world for publishing the image without permission, and the NFT has been removed from the website. Should have stuck with the bored monkeys.
Avatars could be the future
Zoom, FaceTime, and Skype are great when people can’t be together in the same room, state, or country. Not the same as being somewhere in person, but a pretty good replacement during a global pandemic. But what if you had a robot that could be present for you?
Seven-year-old Joshua Martinangeli of Berlin has a severe lung disease and needs to wear a tube in his neck, so he cannot attend school. A robot avatar, donated to Joshua through a private initiative, sits in his seat in the classroom and is able to interact with the students and teacher, according to Reuters. A light on the avatar blinks when Joshua wants to speak and the children can talk with him too. Joshua and his classmates agree that it’s not the same as him really being there to talk and learn, but it’s a great way to keep him included.
“We are the only district in Berlin that has bought four avatars for its schools. The impetus was COVID-19, but I think this will be the future well beyond the pandemic,” Torsten Kuehne, district education councilor, told Reuters.
So where do we get an avatar to go out and run errands? Can we send it to the office instead of Zooming the next meeting? Or maybe our avatar could go to the gym for us. But how do we get the results to show up on our bodies? C’mon science, figure this out.
Futility, thy name is Kiribati
Before we get to the rest of our regularly scheduled hilarity, a brief geography lesson is in order: Kiribati is an island nation – actually 32 atolls and one coral island – in the central Pacific Ocean. Those atolls are spread out across 1.4 million square miles around the intersection of the equator and the International Date Line, so Kiribati is the only country in the world located in all four hemispheres.
Now, back to the news.
Kiribati closed its borders early in the COVID-19 pandemic and recorded only two cases in almost 2 years. Things were going so well that the authorities recently decided to reopen the country to international travelers. Silly authorities.
The first plane was set to arrive on Jan. 14 from Fiji. This being the age of COVID, plans were made and precautions were taken. All 54 passengers quarantined for 2 weeks before the flight and underwent regular testing, the Guardian noted, and “they were only allowed on the flight after returning negative tests.”
You guessed it. Two-thirds of those 54 people tested positive for COVID-19 after landing in Kiribati.
All of the passengers were quarantined, but since then a security guard at the quarantine center has tested positive, as has someone who was not involved in the quarantine. According to NPR, the government said that “there is now an assumption that COVID-19 is now spreading in the community on South Tarawa and Betio.”
Moral of the story? You can’t beat COVID, so never try.
[EDITOR: Is that really the message we want to send to our readers?]
If you can’t beat them, join them.
[EDITOR: Nope. Try again.]
Resistance is futile?
[EDITOR: Sigh. Close enough.]
Are you tired? Or are you death tired?
When we’re feeling that burnout monster creep in we sometimes say that we’re being worked to death or that we’re dead tired, but what if that feeling could predict when it’s your actual time to go?
In a recent study published in the Journals of Gerontology: Series A, epidemiologists from the University of Pittsburgh were able to associate a level of “physical fatigability” with mortality.
The researchers administered the Pittsburgh Fatigability Scale to almost 3,000 participants aged ≥ 60 years, who ranked from 0 to 5 on how tired they thought they would be after doing activities like light housework or a leisurely 30-minute walk. After accounting for factors such as preexisting conditions and mental health, the researchers found that people who scored 25 or more points were 2.3 times more likely to die in the next 2.7 years, compared with those who scored under 25.
So what does that tell us about the importance of being continuously active? It’s pretty important.
“Previous research indicates that getting more physical activity can reduce a person’s fatigability. Our study is the first to link more severe physical fatigability to an earlier death,” lead author Nancy W. Glynn, PhD, said in a separate statement. The best way to keep physically active, she suggested, is to set manageable goals and a routine.
A nice walk around the neighborhood during golden hour or a little bit of yoga before breakfast could be a great way to keep the body moving, because you know what they say: Use it or lose it.
This work is NFT protected: Do not screenshot
If you’ve been following the nonmedical news, you’ve likely heard the term “NFT” explode in the past few months. Standing for nonfungible token, NFTs are, at least theoretically, a proof of ownership for digital creations that prevents anyone other than the buyer from reselling the artwork. Sounds like a great idea: It protects artists and buyers alike.
Much like its cousin cryptocurrency, however, the NFT world is rife with speculation, scams, misunderstanding, and drawings of bored monkeys. It’s the Wild West out there in the digital art universe: One poor unfortunate accidentally sold a $300k NFT image for $3,000, a group of investors spent $3 million buying an NFT for a rare version of Dune believing it gave them the copyright (it did not), and an Indonesian engineering student’s 5-year series of expressionless selfies is now worth a million dollars.
This is a column detailing weird medical news, however, so with our setup complete (though our understanding of NFTs is very much not), we move to France and meet our hero (?), Emmanuel Masmejean, an orthopedic surgeon who apparently wasn’t making enough money in his lucrative medical career.
In a move of apocalyptic madness, he threw ethics out the window, delved into his archive, and found an x-ray of a young woman with a bullet lodged in her arm. The woman was a survivor of the Bataclan mass shooting and bombing in 2015, and don’t you worry, our intrepid entrepreneur made sure to identify her as such when he tried selling the x-ray as an NFT on an online art website for $2,776. Yes, this is very much a violation of doctor-patient confidentiality, and no, that’s not a lot of money to risk your medical career on.
Naturally, the woman was horrified and shocked to learn that the image was being sold, her lawyer told the Guardian. When the doctor called her, he merely attempted to justify his action, rather than apologizing or showing any remorse. Dr. Masmejean is now facing legal action and a disciplinary charge for his attempted entry into the NFT world for publishing the image without permission, and the NFT has been removed from the website. Should have stuck with the bored monkeys.
Avatars could be the future
Zoom, FaceTime, and Skype are great when people can’t be together in the same room, state, or country. Not the same as being somewhere in person, but a pretty good replacement during a global pandemic. But what if you had a robot that could be present for you?
Seven-year-old Joshua Martinangeli of Berlin has a severe lung disease and needs to wear a tube in his neck, so he cannot attend school. A robot avatar, donated to Joshua through a private initiative, sits in his seat in the classroom and is able to interact with the students and teacher, according to Reuters. A light on the avatar blinks when Joshua wants to speak and the children can talk with him too. Joshua and his classmates agree that it’s not the same as him really being there to talk and learn, but it’s a great way to keep him included.
“We are the only district in Berlin that has bought four avatars for its schools. The impetus was COVID-19, but I think this will be the future well beyond the pandemic,” Torsten Kuehne, district education councilor, told Reuters.
So where do we get an avatar to go out and run errands? Can we send it to the office instead of Zooming the next meeting? Or maybe our avatar could go to the gym for us. But how do we get the results to show up on our bodies? C’mon science, figure this out.
Futility, thy name is Kiribati
Before we get to the rest of our regularly scheduled hilarity, a brief geography lesson is in order: Kiribati is an island nation – actually 32 atolls and one coral island – in the central Pacific Ocean. Those atolls are spread out across 1.4 million square miles around the intersection of the equator and the International Date Line, so Kiribati is the only country in the world located in all four hemispheres.
Now, back to the news.
Kiribati closed its borders early in the COVID-19 pandemic and recorded only two cases in almost 2 years. Things were going so well that the authorities recently decided to reopen the country to international travelers. Silly authorities.
The first plane was set to arrive on Jan. 14 from Fiji. This being the age of COVID, plans were made and precautions were taken. All 54 passengers quarantined for 2 weeks before the flight and underwent regular testing, the Guardian noted, and “they were only allowed on the flight after returning negative tests.”
You guessed it. Two-thirds of those 54 people tested positive for COVID-19 after landing in Kiribati.
All of the passengers were quarantined, but since then a security guard at the quarantine center has tested positive, as has someone who was not involved in the quarantine. According to NPR, the government said that “there is now an assumption that COVID-19 is now spreading in the community on South Tarawa and Betio.”
Moral of the story? You can’t beat COVID, so never try.
[EDITOR: Is that really the message we want to send to our readers?]
If you can’t beat them, join them.
[EDITOR: Nope. Try again.]
Resistance is futile?
[EDITOR: Sigh. Close enough.]
Are you tired? Or are you death tired?
When we’re feeling that burnout monster creep in we sometimes say that we’re being worked to death or that we’re dead tired, but what if that feeling could predict when it’s your actual time to go?
In a recent study published in the Journals of Gerontology: Series A, epidemiologists from the University of Pittsburgh were able to associate a level of “physical fatigability” with mortality.
The researchers administered the Pittsburgh Fatigability Scale to almost 3,000 participants aged ≥ 60 years, who ranked from 0 to 5 on how tired they thought they would be after doing activities like light housework or a leisurely 30-minute walk. After accounting for factors such as preexisting conditions and mental health, the researchers found that people who scored 25 or more points were 2.3 times more likely to die in the next 2.7 years, compared with those who scored under 25.
So what does that tell us about the importance of being continuously active? It’s pretty important.
“Previous research indicates that getting more physical activity can reduce a person’s fatigability. Our study is the first to link more severe physical fatigability to an earlier death,” lead author Nancy W. Glynn, PhD, said in a separate statement. The best way to keep physically active, she suggested, is to set manageable goals and a routine.
A nice walk around the neighborhood during golden hour or a little bit of yoga before breakfast could be a great way to keep the body moving, because you know what they say: Use it or lose it.
This work is NFT protected: Do not screenshot
If you’ve been following the nonmedical news, you’ve likely heard the term “NFT” explode in the past few months. Standing for nonfungible token, NFTs are, at least theoretically, a proof of ownership for digital creations that prevents anyone other than the buyer from reselling the artwork. Sounds like a great idea: It protects artists and buyers alike.
Much like its cousin cryptocurrency, however, the NFT world is rife with speculation, scams, misunderstanding, and drawings of bored monkeys. It’s the Wild West out there in the digital art universe: One poor unfortunate accidentally sold a $300k NFT image for $3,000, a group of investors spent $3 million buying an NFT for a rare version of Dune believing it gave them the copyright (it did not), and an Indonesian engineering student’s 5-year series of expressionless selfies is now worth a million dollars.
This is a column detailing weird medical news, however, so with our setup complete (though our understanding of NFTs is very much not), we move to France and meet our hero (?), Emmanuel Masmejean, an orthopedic surgeon who apparently wasn’t making enough money in his lucrative medical career.
In a move of apocalyptic madness, he threw ethics out the window, delved into his archive, and found an x-ray of a young woman with a bullet lodged in her arm. The woman was a survivor of the Bataclan mass shooting and bombing in 2015, and don’t you worry, our intrepid entrepreneur made sure to identify her as such when he tried selling the x-ray as an NFT on an online art website for $2,776. Yes, this is very much a violation of doctor-patient confidentiality, and no, that’s not a lot of money to risk your medical career on.
Naturally, the woman was horrified and shocked to learn that the image was being sold, her lawyer told the Guardian. When the doctor called her, he merely attempted to justify his action, rather than apologizing or showing any remorse. Dr. Masmejean is now facing legal action and a disciplinary charge for his attempted entry into the NFT world for publishing the image without permission, and the NFT has been removed from the website. Should have stuck with the bored monkeys.
Avatars could be the future
Zoom, FaceTime, and Skype are great when people can’t be together in the same room, state, or country. Not the same as being somewhere in person, but a pretty good replacement during a global pandemic. But what if you had a robot that could be present for you?
Seven-year-old Joshua Martinangeli of Berlin has a severe lung disease and needs to wear a tube in his neck, so he cannot attend school. A robot avatar, donated to Joshua through a private initiative, sits in his seat in the classroom and is able to interact with the students and teacher, according to Reuters. A light on the avatar blinks when Joshua wants to speak and the children can talk with him too. Joshua and his classmates agree that it’s not the same as him really being there to talk and learn, but it’s a great way to keep him included.
“We are the only district in Berlin that has bought four avatars for its schools. The impetus was COVID-19, but I think this will be the future well beyond the pandemic,” Torsten Kuehne, district education councilor, told Reuters.
So where do we get an avatar to go out and run errands? Can we send it to the office instead of Zooming the next meeting? Or maybe our avatar could go to the gym for us. But how do we get the results to show up on our bodies? C’mon science, figure this out.
Futility, thy name is Kiribati
Before we get to the rest of our regularly scheduled hilarity, a brief geography lesson is in order: Kiribati is an island nation – actually 32 atolls and one coral island – in the central Pacific Ocean. Those atolls are spread out across 1.4 million square miles around the intersection of the equator and the International Date Line, so Kiribati is the only country in the world located in all four hemispheres.
Now, back to the news.
Kiribati closed its borders early in the COVID-19 pandemic and recorded only two cases in almost 2 years. Things were going so well that the authorities recently decided to reopen the country to international travelers. Silly authorities.
The first plane was set to arrive on Jan. 14 from Fiji. This being the age of COVID, plans were made and precautions were taken. All 54 passengers quarantined for 2 weeks before the flight and underwent regular testing, the Guardian noted, and “they were only allowed on the flight after returning negative tests.”
You guessed it. Two-thirds of those 54 people tested positive for COVID-19 after landing in Kiribati.
All of the passengers were quarantined, but since then a security guard at the quarantine center has tested positive, as has someone who was not involved in the quarantine. According to NPR, the government said that “there is now an assumption that COVID-19 is now spreading in the community on South Tarawa and Betio.”
Moral of the story? You can’t beat COVID, so never try.
[EDITOR: Is that really the message we want to send to our readers?]
If you can’t beat them, join them.
[EDITOR: Nope. Try again.]
Resistance is futile?
[EDITOR: Sigh. Close enough.]
35% of employers to proceed with vaccine mandate, poll shows
despite a recent U.S. Supreme Court ruling that blocked the Biden administration’s vaccine-or-test rule for big businesses.
But the poll by Gartner Inc. showed no consensus among employers. About 4% of polled executives said they’re dropping their vaccine mandate, 29% are in a wait-and-see position, and 12% are less likely to impose a mandate now, Bloomberg reported.
Executives were divided on how a vaccine mandate would affect absenteeism and employee morale. Almost 40% of polled employers said they thought a mandate would attract workers, but about 25% said it would do the opposite, Bloomberg said.
“What is more attractive -- to have a mandate or not?” Brian Kropp, PhD, Gartner’s chief of human resources research, said in an interview with Bloomberg. “Most are not exactly sure what to do.”
Big companies have reacted differently since the court’s ruling.
Starbucks announced it was dropping its vaccine-or-test rule for the company’s approximately 228,000 employees. General Electric dropped its mandate after the ruling, but Honeywell International Inc. announced it was staying with its vaccination policy, Bloomberg said.
The Supreme Court ruled Jan. 13 against the Biden administration’s mandate for businesses. The Occupational Safety and Health Administration had proposed that every company with more than 100 employees would be required to ensure workers were either vaccinated or tested weekly for COVID-19.
State governments and business groups immediately appealed, and the court ruled 6-3 against the mandate. The Biden administration officially dropped its rule on Wednesday.
A version of this article first appeared on WebMD.com.
despite a recent U.S. Supreme Court ruling that blocked the Biden administration’s vaccine-or-test rule for big businesses.
But the poll by Gartner Inc. showed no consensus among employers. About 4% of polled executives said they’re dropping their vaccine mandate, 29% are in a wait-and-see position, and 12% are less likely to impose a mandate now, Bloomberg reported.
Executives were divided on how a vaccine mandate would affect absenteeism and employee morale. Almost 40% of polled employers said they thought a mandate would attract workers, but about 25% said it would do the opposite, Bloomberg said.
“What is more attractive -- to have a mandate or not?” Brian Kropp, PhD, Gartner’s chief of human resources research, said in an interview with Bloomberg. “Most are not exactly sure what to do.”
Big companies have reacted differently since the court’s ruling.
Starbucks announced it was dropping its vaccine-or-test rule for the company’s approximately 228,000 employees. General Electric dropped its mandate after the ruling, but Honeywell International Inc. announced it was staying with its vaccination policy, Bloomberg said.
The Supreme Court ruled Jan. 13 against the Biden administration’s mandate for businesses. The Occupational Safety and Health Administration had proposed that every company with more than 100 employees would be required to ensure workers were either vaccinated or tested weekly for COVID-19.
State governments and business groups immediately appealed, and the court ruled 6-3 against the mandate. The Biden administration officially dropped its rule on Wednesday.
A version of this article first appeared on WebMD.com.
despite a recent U.S. Supreme Court ruling that blocked the Biden administration’s vaccine-or-test rule for big businesses.
But the poll by Gartner Inc. showed no consensus among employers. About 4% of polled executives said they’re dropping their vaccine mandate, 29% are in a wait-and-see position, and 12% are less likely to impose a mandate now, Bloomberg reported.
Executives were divided on how a vaccine mandate would affect absenteeism and employee morale. Almost 40% of polled employers said they thought a mandate would attract workers, but about 25% said it would do the opposite, Bloomberg said.
“What is more attractive -- to have a mandate or not?” Brian Kropp, PhD, Gartner’s chief of human resources research, said in an interview with Bloomberg. “Most are not exactly sure what to do.”
Big companies have reacted differently since the court’s ruling.
Starbucks announced it was dropping its vaccine-or-test rule for the company’s approximately 228,000 employees. General Electric dropped its mandate after the ruling, but Honeywell International Inc. announced it was staying with its vaccination policy, Bloomberg said.
The Supreme Court ruled Jan. 13 against the Biden administration’s mandate for businesses. The Occupational Safety and Health Administration had proposed that every company with more than 100 employees would be required to ensure workers were either vaccinated or tested weekly for COVID-19.
State governments and business groups immediately appealed, and the court ruled 6-3 against the mandate. The Biden administration officially dropped its rule on Wednesday.
A version of this article first appeared on WebMD.com.
Omicron survives longer on plastic, skin than other COVID variants
, one possible explanation for why Omicron has spread so rapidly around the world.
In a lab experiment, samples of different variants were applied to pieces of plastic and human skin collected from autopsies, researchers from Kyoto Prefectural University of Medicine wrote in bioRxiv. A variant “survived” until it could no longer be detected on the surface.
“This study showed that the Omicron variant also has the highest environmental stability among VOCs (variants of concern), which suggests that this high stability might also be one of the factors that have allowed the Omicron variant to replace the Delta variant and spread rapidly,” the researchers wrote.
On plastic, the Omicron variant samples survived an average of 193.5 hours, a little more than 8 days. By comparison, the other survival times on plastic were 56 hours for the original COVID strain, 191.3 hours for Alpha, 156.6 hours for Beta, 59.3 hours for Gamma, and 114 hours for Delta.
On skin samples, the Omicron samples survived an average of 21.1 hours. The other variants had these average survival times on skin: 8.6 hours for the original version, 19.6 hours for Alpha, 19.1 hours for Beta, 11 hours for Gamma, and 16.8 hours for Delta.
The study found that the variants had more resistance to ethanol than the original strain of COVID. That said, all COVID samples were inactivated after being exposed to alcohol-based hand sanitizers for 15 seconds.
“Therefore, it is highly recommended that current infection control (hand hygiene) practices use disinfectants ... as proposed by the World Health Organization,” the researchers said.
The study has not been peer-reviewed.
A version of this article first appeared on WebMD.com.
, one possible explanation for why Omicron has spread so rapidly around the world.
In a lab experiment, samples of different variants were applied to pieces of plastic and human skin collected from autopsies, researchers from Kyoto Prefectural University of Medicine wrote in bioRxiv. A variant “survived” until it could no longer be detected on the surface.
“This study showed that the Omicron variant also has the highest environmental stability among VOCs (variants of concern), which suggests that this high stability might also be one of the factors that have allowed the Omicron variant to replace the Delta variant and spread rapidly,” the researchers wrote.
On plastic, the Omicron variant samples survived an average of 193.5 hours, a little more than 8 days. By comparison, the other survival times on plastic were 56 hours for the original COVID strain, 191.3 hours for Alpha, 156.6 hours for Beta, 59.3 hours for Gamma, and 114 hours for Delta.
On skin samples, the Omicron samples survived an average of 21.1 hours. The other variants had these average survival times on skin: 8.6 hours for the original version, 19.6 hours for Alpha, 19.1 hours for Beta, 11 hours for Gamma, and 16.8 hours for Delta.
The study found that the variants had more resistance to ethanol than the original strain of COVID. That said, all COVID samples were inactivated after being exposed to alcohol-based hand sanitizers for 15 seconds.
“Therefore, it is highly recommended that current infection control (hand hygiene) practices use disinfectants ... as proposed by the World Health Organization,” the researchers said.
The study has not been peer-reviewed.
A version of this article first appeared on WebMD.com.
, one possible explanation for why Omicron has spread so rapidly around the world.
In a lab experiment, samples of different variants were applied to pieces of plastic and human skin collected from autopsies, researchers from Kyoto Prefectural University of Medicine wrote in bioRxiv. A variant “survived” until it could no longer be detected on the surface.
“This study showed that the Omicron variant also has the highest environmental stability among VOCs (variants of concern), which suggests that this high stability might also be one of the factors that have allowed the Omicron variant to replace the Delta variant and spread rapidly,” the researchers wrote.
On plastic, the Omicron variant samples survived an average of 193.5 hours, a little more than 8 days. By comparison, the other survival times on plastic were 56 hours for the original COVID strain, 191.3 hours for Alpha, 156.6 hours for Beta, 59.3 hours for Gamma, and 114 hours for Delta.
On skin samples, the Omicron samples survived an average of 21.1 hours. The other variants had these average survival times on skin: 8.6 hours for the original version, 19.6 hours for Alpha, 19.1 hours for Beta, 11 hours for Gamma, and 16.8 hours for Delta.
The study found that the variants had more resistance to ethanol than the original strain of COVID. That said, all COVID samples were inactivated after being exposed to alcohol-based hand sanitizers for 15 seconds.
“Therefore, it is highly recommended that current infection control (hand hygiene) practices use disinfectants ... as proposed by the World Health Organization,” the researchers said.
The study has not been peer-reviewed.
A version of this article first appeared on WebMD.com.
Presence of autoantibodies most predictive of long COVID in study
Other significant early predictors of prolonged COVID symptoms – which the researchers called postacute sequelae – were having type 2 diabetes, SARS-CoV-2 RNAemia, and Epstein-Barr virus (EBV) viremia, Yapeng Su, PhD, of the Institute for Systems Biology (ISB) in Seattle, and colleagues wrote in Cell.
Having EBV viremia suggested that latent EBV has been reactivated, the authors noted.
“The most important postacute sequelae [that is conditions that are consequences of a disease] of COVID is the presence of autoantibodies,” James R. Heath, PhD, president of ISB and a bioengineering professor at the University of Washington, Seattle, said in an interview. “It’s about two times more important than the others.”
Dr. Heath and coauthors said early detection of this and other variables could prompt earlier aggressive treatment in patients susceptible to long COVID and ward off lingering symptoms.
“These predictive measures of long COVID can also help to better inform patients of their possible disease course,” study coauthor Daniel G. Chen, an undergraduate researcher at ISB, said in an interview. “We were also able to partially resolve the immunological underpinnings of some postacute sequelae of COVID in a way that suggested potential therapies, and the timing of those therapies.”
For example, he continued, the use of antivirals very early in the infectious course may mitigate the later development of long COVID. “This will, of course, have to be explored in an appropriately designed clinical trial.
“We also identified biomarkers of certain types of long COVID, such as neurological sequelae. Those biomarkers can help define the condition, which is a first step towards developing treatments.”
Study findings
With COVID patients monitored for 2 or 3 months, the study findings of the international “multiomic profiling” analysis include:
- Subclinical patient autoantibodies that reduce anti–SARS-CoV-2 antibodies suggest there is immune dysregulation during COVID-19 infection.
- Reactivation of latent other viruses during initial infection may be contributing to long COVID.
- Gastrointestinal postacute sequelae of COVID presents with a unique postacute expansion of cytotoxic T cells.
- SARS-CoV-2–specific and cytomegalovirus-specific CD8+ T cells displayed unique dynamics during recovery from infection.
According to the authors, as many as 69% of COVID-19 patients suffer from long COVID – a range of new, recurrent, or ongoing problems 4 or more weeks following initial SARS-CoV-2 infection. These may include memory loss, gastrointestinal distress, fatigue, anosmia, and shortness of breath.
Long COVID has been associated with acute disease severity, and is suspected to be related to autoimmune factors and unresolved viral fragments, according to the paper.
Research methods
The international study did a deep and detailed dive into multiple molecular markers of long COVID. It enrolled 209 COVID-19 patients with varying degrees of disease severity and matched them to 457 healthy controls. The researchers’ goal was to identify discrete and quantifiable long COVID factors and guide possible preemptive treatment.
Patients were assessed at three time points: at initial diagnosis, during the acute disease phase about a week later, and again 2 to 3 months post onset of symptoms after recovery from the acute phase of COVID. At the third assessment, some patients had lingering symptoms such as fatigue (52% ), cough (25%), and loss of taste or sense of smell (18%).
Blood draws were analyzed for autoantibodies and SARS-CoV-2–specific antibodies, global plasma proteomic and metabolomic profiles, and single-cell multiomic characterizations of peripheral blood mononuclear cells.
Each blood draw was paired with nasal-swab and plasma measurements of SARS-CoV-2 viral load and the data sets were integrated with electronic health records and self-reported patient symptoms to guide the interpretation of the molecular signatures of long COVID.
Author conclusions
The authors found an association between T2 hyperinflammation and long COVID–anticipating autoantibodies. This association further implies that hyperinflammation-controlling therapies in the acute stage of COVID may influence whether a patient experiences long COVID. “However, the detailed timing and context of these therapies matter, and, thus, future well-controlled studies will be needed to test these and other therapeutic implications,” Dr. Su and colleagues wrote.
Moreover, the negative correlations between anti–SARS-CoV-2 IgG and certain autoantibodies may suggest that patients with elevated autoantibody levels are more susceptible to breakthrough infections, the authors said.
“Many patients with high autoantibodies simultaneously have low protective antibodies that neutralize SARS-CoV-2, and that’s going to make them more susceptible to breakthrough infections,” Mr. Chen explained.*
“Detectability of most [long COVID-19 factors] at COVID diagnosis emphasizes the importance of early disease measurements for understanding emergent chronic conditions and suggests [long COVID] treatment strategies,” they wrote.
According to Mr. Chen, there are clear similarities in underlying immunobiology between patients with COVID autoantibodies and patients with systemic lupus erythematosus.
“These findings are also helping us frame our thinking around other chronic autoimmune conditions, such as postacute Lyme syndrome, for example,” said Dr. Heath.
The bottom line, said Mr. Chen, is that measuring early long COVID indicators may result in preventive treatments. “An example is the cortisol deficiency we see in certain long COVID patients. There are known treatments such as cortisol replacement therapy that should be explored for this group.”
Outside expert’s take on findings
Commenting on the study, Sherry Hsiang-Yi Chou, MD, who was not involved in the research, called the study a very important first step in understanding the path of this complex phenomenon and perhaps other conditions with long-term side effects.
“The researchers have done huge amount of innovative scientific work. They’ve shown the DNA signature of how our bodies respond to this disease,” said Dr. Chou, who is chief of the division of neurocritical care at Northwestern Medicine in Chicago.
“This type of research will help us scientifically understand and differentiate the various syndromes within long COVID. It will help identify who’s at risk for different aspects of this syndrome and lead to following them for longer periods in clinical trials,” she added.
The authors acknowledged that lengthier studies in larger cohorts were needed to see which patients will develop long-term chronic postacute sequelae of COVID.
This research was supported by the Wilke Family Foundation, the Parker Institute for Cancer Immunotherapy, Merck, and the Biomedical Advanced Research and Development Authority. Other support came from the National Institutes of Health, the Bill and Melinda Gates Foundation, Saint John’s Cancer Center, Fred Hutchinson Cancer Research Center, and the European Union’s Horizon 2020 research and innovation program. Dr. Heath is a cofounder of Pact Pharma. He and several coauthors disclosed various ties to multiple private-sector companies. Mr. Chen and Dr. Chou had no competing interests.
*Correction, 1/28: An earlier version of this story misidentified Daniel G. Chen, an undergraduate researcher at ISB.
Other significant early predictors of prolonged COVID symptoms – which the researchers called postacute sequelae – were having type 2 diabetes, SARS-CoV-2 RNAemia, and Epstein-Barr virus (EBV) viremia, Yapeng Su, PhD, of the Institute for Systems Biology (ISB) in Seattle, and colleagues wrote in Cell.
Having EBV viremia suggested that latent EBV has been reactivated, the authors noted.
“The most important postacute sequelae [that is conditions that are consequences of a disease] of COVID is the presence of autoantibodies,” James R. Heath, PhD, president of ISB and a bioengineering professor at the University of Washington, Seattle, said in an interview. “It’s about two times more important than the others.”
Dr. Heath and coauthors said early detection of this and other variables could prompt earlier aggressive treatment in patients susceptible to long COVID and ward off lingering symptoms.
“These predictive measures of long COVID can also help to better inform patients of their possible disease course,” study coauthor Daniel G. Chen, an undergraduate researcher at ISB, said in an interview. “We were also able to partially resolve the immunological underpinnings of some postacute sequelae of COVID in a way that suggested potential therapies, and the timing of those therapies.”
For example, he continued, the use of antivirals very early in the infectious course may mitigate the later development of long COVID. “This will, of course, have to be explored in an appropriately designed clinical trial.
“We also identified biomarkers of certain types of long COVID, such as neurological sequelae. Those biomarkers can help define the condition, which is a first step towards developing treatments.”
Study findings
With COVID patients monitored for 2 or 3 months, the study findings of the international “multiomic profiling” analysis include:
- Subclinical patient autoantibodies that reduce anti–SARS-CoV-2 antibodies suggest there is immune dysregulation during COVID-19 infection.
- Reactivation of latent other viruses during initial infection may be contributing to long COVID.
- Gastrointestinal postacute sequelae of COVID presents with a unique postacute expansion of cytotoxic T cells.
- SARS-CoV-2–specific and cytomegalovirus-specific CD8+ T cells displayed unique dynamics during recovery from infection.
According to the authors, as many as 69% of COVID-19 patients suffer from long COVID – a range of new, recurrent, or ongoing problems 4 or more weeks following initial SARS-CoV-2 infection. These may include memory loss, gastrointestinal distress, fatigue, anosmia, and shortness of breath.
Long COVID has been associated with acute disease severity, and is suspected to be related to autoimmune factors and unresolved viral fragments, according to the paper.
Research methods
The international study did a deep and detailed dive into multiple molecular markers of long COVID. It enrolled 209 COVID-19 patients with varying degrees of disease severity and matched them to 457 healthy controls. The researchers’ goal was to identify discrete and quantifiable long COVID factors and guide possible preemptive treatment.
Patients were assessed at three time points: at initial diagnosis, during the acute disease phase about a week later, and again 2 to 3 months post onset of symptoms after recovery from the acute phase of COVID. At the third assessment, some patients had lingering symptoms such as fatigue (52% ), cough (25%), and loss of taste or sense of smell (18%).
Blood draws were analyzed for autoantibodies and SARS-CoV-2–specific antibodies, global plasma proteomic and metabolomic profiles, and single-cell multiomic characterizations of peripheral blood mononuclear cells.
Each blood draw was paired with nasal-swab and plasma measurements of SARS-CoV-2 viral load and the data sets were integrated with electronic health records and self-reported patient symptoms to guide the interpretation of the molecular signatures of long COVID.
Author conclusions
The authors found an association between T2 hyperinflammation and long COVID–anticipating autoantibodies. This association further implies that hyperinflammation-controlling therapies in the acute stage of COVID may influence whether a patient experiences long COVID. “However, the detailed timing and context of these therapies matter, and, thus, future well-controlled studies will be needed to test these and other therapeutic implications,” Dr. Su and colleagues wrote.
Moreover, the negative correlations between anti–SARS-CoV-2 IgG and certain autoantibodies may suggest that patients with elevated autoantibody levels are more susceptible to breakthrough infections, the authors said.
“Many patients with high autoantibodies simultaneously have low protective antibodies that neutralize SARS-CoV-2, and that’s going to make them more susceptible to breakthrough infections,” Mr. Chen explained.*
“Detectability of most [long COVID-19 factors] at COVID diagnosis emphasizes the importance of early disease measurements for understanding emergent chronic conditions and suggests [long COVID] treatment strategies,” they wrote.
According to Mr. Chen, there are clear similarities in underlying immunobiology between patients with COVID autoantibodies and patients with systemic lupus erythematosus.
“These findings are also helping us frame our thinking around other chronic autoimmune conditions, such as postacute Lyme syndrome, for example,” said Dr. Heath.
The bottom line, said Mr. Chen, is that measuring early long COVID indicators may result in preventive treatments. “An example is the cortisol deficiency we see in certain long COVID patients. There are known treatments such as cortisol replacement therapy that should be explored for this group.”
Outside expert’s take on findings
Commenting on the study, Sherry Hsiang-Yi Chou, MD, who was not involved in the research, called the study a very important first step in understanding the path of this complex phenomenon and perhaps other conditions with long-term side effects.
“The researchers have done huge amount of innovative scientific work. They’ve shown the DNA signature of how our bodies respond to this disease,” said Dr. Chou, who is chief of the division of neurocritical care at Northwestern Medicine in Chicago.
“This type of research will help us scientifically understand and differentiate the various syndromes within long COVID. It will help identify who’s at risk for different aspects of this syndrome and lead to following them for longer periods in clinical trials,” she added.
The authors acknowledged that lengthier studies in larger cohorts were needed to see which patients will develop long-term chronic postacute sequelae of COVID.
This research was supported by the Wilke Family Foundation, the Parker Institute for Cancer Immunotherapy, Merck, and the Biomedical Advanced Research and Development Authority. Other support came from the National Institutes of Health, the Bill and Melinda Gates Foundation, Saint John’s Cancer Center, Fred Hutchinson Cancer Research Center, and the European Union’s Horizon 2020 research and innovation program. Dr. Heath is a cofounder of Pact Pharma. He and several coauthors disclosed various ties to multiple private-sector companies. Mr. Chen and Dr. Chou had no competing interests.
*Correction, 1/28: An earlier version of this story misidentified Daniel G. Chen, an undergraduate researcher at ISB.
Other significant early predictors of prolonged COVID symptoms – which the researchers called postacute sequelae – were having type 2 diabetes, SARS-CoV-2 RNAemia, and Epstein-Barr virus (EBV) viremia, Yapeng Su, PhD, of the Institute for Systems Biology (ISB) in Seattle, and colleagues wrote in Cell.
Having EBV viremia suggested that latent EBV has been reactivated, the authors noted.
“The most important postacute sequelae [that is conditions that are consequences of a disease] of COVID is the presence of autoantibodies,” James R. Heath, PhD, president of ISB and a bioengineering professor at the University of Washington, Seattle, said in an interview. “It’s about two times more important than the others.”
Dr. Heath and coauthors said early detection of this and other variables could prompt earlier aggressive treatment in patients susceptible to long COVID and ward off lingering symptoms.
“These predictive measures of long COVID can also help to better inform patients of their possible disease course,” study coauthor Daniel G. Chen, an undergraduate researcher at ISB, said in an interview. “We were also able to partially resolve the immunological underpinnings of some postacute sequelae of COVID in a way that suggested potential therapies, and the timing of those therapies.”
For example, he continued, the use of antivirals very early in the infectious course may mitigate the later development of long COVID. “This will, of course, have to be explored in an appropriately designed clinical trial.
“We also identified biomarkers of certain types of long COVID, such as neurological sequelae. Those biomarkers can help define the condition, which is a first step towards developing treatments.”
Study findings
With COVID patients monitored for 2 or 3 months, the study findings of the international “multiomic profiling” analysis include:
- Subclinical patient autoantibodies that reduce anti–SARS-CoV-2 antibodies suggest there is immune dysregulation during COVID-19 infection.
- Reactivation of latent other viruses during initial infection may be contributing to long COVID.
- Gastrointestinal postacute sequelae of COVID presents with a unique postacute expansion of cytotoxic T cells.
- SARS-CoV-2–specific and cytomegalovirus-specific CD8+ T cells displayed unique dynamics during recovery from infection.
According to the authors, as many as 69% of COVID-19 patients suffer from long COVID – a range of new, recurrent, or ongoing problems 4 or more weeks following initial SARS-CoV-2 infection. These may include memory loss, gastrointestinal distress, fatigue, anosmia, and shortness of breath.
Long COVID has been associated with acute disease severity, and is suspected to be related to autoimmune factors and unresolved viral fragments, according to the paper.
Research methods
The international study did a deep and detailed dive into multiple molecular markers of long COVID. It enrolled 209 COVID-19 patients with varying degrees of disease severity and matched them to 457 healthy controls. The researchers’ goal was to identify discrete and quantifiable long COVID factors and guide possible preemptive treatment.
Patients were assessed at three time points: at initial diagnosis, during the acute disease phase about a week later, and again 2 to 3 months post onset of symptoms after recovery from the acute phase of COVID. At the third assessment, some patients had lingering symptoms such as fatigue (52% ), cough (25%), and loss of taste or sense of smell (18%).
Blood draws were analyzed for autoantibodies and SARS-CoV-2–specific antibodies, global plasma proteomic and metabolomic profiles, and single-cell multiomic characterizations of peripheral blood mononuclear cells.
Each blood draw was paired with nasal-swab and plasma measurements of SARS-CoV-2 viral load and the data sets were integrated with electronic health records and self-reported patient symptoms to guide the interpretation of the molecular signatures of long COVID.
Author conclusions
The authors found an association between T2 hyperinflammation and long COVID–anticipating autoantibodies. This association further implies that hyperinflammation-controlling therapies in the acute stage of COVID may influence whether a patient experiences long COVID. “However, the detailed timing and context of these therapies matter, and, thus, future well-controlled studies will be needed to test these and other therapeutic implications,” Dr. Su and colleagues wrote.
Moreover, the negative correlations between anti–SARS-CoV-2 IgG and certain autoantibodies may suggest that patients with elevated autoantibody levels are more susceptible to breakthrough infections, the authors said.
“Many patients with high autoantibodies simultaneously have low protective antibodies that neutralize SARS-CoV-2, and that’s going to make them more susceptible to breakthrough infections,” Mr. Chen explained.*
“Detectability of most [long COVID-19 factors] at COVID diagnosis emphasizes the importance of early disease measurements for understanding emergent chronic conditions and suggests [long COVID] treatment strategies,” they wrote.
According to Mr. Chen, there are clear similarities in underlying immunobiology between patients with COVID autoantibodies and patients with systemic lupus erythematosus.
“These findings are also helping us frame our thinking around other chronic autoimmune conditions, such as postacute Lyme syndrome, for example,” said Dr. Heath.
The bottom line, said Mr. Chen, is that measuring early long COVID indicators may result in preventive treatments. “An example is the cortisol deficiency we see in certain long COVID patients. There are known treatments such as cortisol replacement therapy that should be explored for this group.”
Outside expert’s take on findings
Commenting on the study, Sherry Hsiang-Yi Chou, MD, who was not involved in the research, called the study a very important first step in understanding the path of this complex phenomenon and perhaps other conditions with long-term side effects.
“The researchers have done huge amount of innovative scientific work. They’ve shown the DNA signature of how our bodies respond to this disease,” said Dr. Chou, who is chief of the division of neurocritical care at Northwestern Medicine in Chicago.
“This type of research will help us scientifically understand and differentiate the various syndromes within long COVID. It will help identify who’s at risk for different aspects of this syndrome and lead to following them for longer periods in clinical trials,” she added.
The authors acknowledged that lengthier studies in larger cohorts were needed to see which patients will develop long-term chronic postacute sequelae of COVID.
This research was supported by the Wilke Family Foundation, the Parker Institute for Cancer Immunotherapy, Merck, and the Biomedical Advanced Research and Development Authority. Other support came from the National Institutes of Health, the Bill and Melinda Gates Foundation, Saint John’s Cancer Center, Fred Hutchinson Cancer Research Center, and the European Union’s Horizon 2020 research and innovation program. Dr. Heath is a cofounder of Pact Pharma. He and several coauthors disclosed various ties to multiple private-sector companies. Mr. Chen and Dr. Chou had no competing interests.
*Correction, 1/28: An earlier version of this story misidentified Daniel G. Chen, an undergraduate researcher at ISB.
FROM CELL
Dapivirine vaginal ring for HIV prevention no longer under consideration by the FDA
Tosha Rogers, MD, is a one-woman HIV prevention evangelist. For nearly a decade now, the Atlanta-based ob/gyn has been on a mission to increase her gynecological colleagues’ awareness and prescribing of the oral HIV prevention pill. At the same time, she’s been tracking the development of a flexible vaginal ring loaded with a month’s worth of the HIV prevention medication dapivirine. That, she thought, would fit easily into women’s lives and into the toolbox of methods women already use to prevent pregnancy.
But now she’s not sure when – or if – the ring will find its way to her patients. In December, the ring’s maker, the International Partnership for Microbicides (IPM), pulled its application for FDA approval for the pre-exposure prophylaxis (PrEP) ring. Now, one year after the World Health Organization recommended the ring for member nations, there appears to be no path forward in the United States for either the dapivirine-only ring or an approach Dr. Rogers said would change the game: a vaginal ring that supplies both contraception and HIV prevention.
“It would take things to a whole other level,” she said. “It sucks that this happened, and I do think it was not anything medical. I think it was everything political.”
That leaves cisgender women – especially the Black and Latinx women who make up the vast majority of women who acquire HIV every year – with two HIV prevention options. One is the daily pill, first approved in 2012. It’s now generic but previously sold as Truvada by Gilead Sciences. The other is monthly injectable cabotegravir long-acting (Apretude). Another HIV prevention pill, tenofovir alafenamide/emtricitabine (Descovy), is approved for gay men and transgender women but not cisgender women.
Vagina-specific protection from HIV
The WHO recommendation for the vaginal ring was followed last July by a positive opinion from the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) for women in low- and middle-income countries outside the European Union.
The flexible silicone ring, similar to the hormonal NuvaRing contraceptive, works by slowly releasing the antiretroviral dapivirine directly into the vaginal canal, thereby protecting women who might be exposed to the virus through vaginal sex only. Because the medicine stays where it’s delivered and doesn’t circulate through the body, it has been found to be extremely safe with few adverse events.
However, in initial studies, the ring was found to be just 27% effective overall. Later studies, where scientists divided women by how much drug was missing from the ring – a proxy for use – found that higher use was associated with higher protection (as much as 54%). By comparison, Truvada has been found to be up to 99% effective when used daily, though it can take up to 21 days to be available in the vagina in high enough concentrations to protect women from vaginal exposure. And the HIV prevention shot was found to be 90% more effective than that in a recent trial of the two methods conducted by the HIV Prevention Trials Network.
This, and an orientation away from topical HIV prevention drugs and toward systemic options, led the National Institute of Allergy and Infectious Diseases (NIAID) to discontinue funding for such projects under its Microbicide Trials Network.
“Clearly you want to counsel women to use the highest efficacy method, and that is part of our label,” Zeda Rosenberg, ScD, IPM’s founder and chief executive officer, told this news organization. “Women should not choose the ring if they can and will use oral PrEP, and I would argue it should be the same thing for [cabotegravir shots]. But if they can’t or don’t want to – and we know that especially many young women don’t want to use systemic methods – then the dapivirine ring is a great option.”
Still, Dr. Rosenberg said that the gap in efficacy, the relatively small number of women affected by HIV in the U.S. compared with gay and bisexual men, and the emergence of products like the HIV prevention shot cabotegravir, made it “very unlikely” that FDA regulators would approve the ring. And rather than be “distracted” by the FDA process, Dr. Rosenberg said IPM chose to concentrate on the countries where the ring has already been approved or where women make up the vast majority of people affected by HIV.
Zimbabwe publicly announced it has approved the ring, and three other countries may have approved it, according to Dr. Rosenberg. She declined to name them, saying they had requested silence while they formulate their new HIV prevention guidelines. Aside from Zimbabwe, the other countries where women participated in the ring clinical trials were South Africa, Malawi, and Uganda.
“The U.S. population ... has widespread access to oral PrEP, which is unlike countries in Africa, and which would have widespread access to injectable cabotegravir,” she said. “The U.S. FDA may not see choice in the same way that African women and African activists and advocates see the need for choice.”
But women’s rates of accessing HIV prevention medications in the U.S. continues to be frustratingly low. At the end of 2018, just 7% of women who could benefit from HIV prevention drugs were taking them, according to Centers for Disease Control and Prevention data.
New CDC guidelines recommend clinicians talk to every sexually active adult and adolescent about HIV prevention medications at least once and prescribe it to anyone who asks for it, whether or not they understand their patients’ HIV risks. However, research continues to show that clinicians struggle with willingness to prescribe PrEP to Black women, and the American College of Obstetrics and Gynecology’s committee opinion on managing women using HIV prevention drugs has not been updated to reflect the new guidelines. And while the HIV prevention shot is approved for women and its maker ViiV Healthcare is already initiating postmarket studies of the ring in key populations including women, there are lots of things that need to line up in order for clinicians to be willing to stock it and prescribe it to women.
From where Dázon Dixon Diallo, executive director of the nonprofit SisterLove, sits, the decision to withdraw the ring from FDA consideration and the FDA’s seeming argument that the epidemiology in the U.S. doesn’t warrant the ring’s approval is a slap in the face to the Black women who have led the movement to end HIV in the U.S. for decades.
“No matter how you slice it, we’re talking about Black women, and then we’re talking about brown women,” said Ms. Diallo. “The value [they place on us] from a government standpoint, from a political standpoint, from a public health standpoint is just woeful. It’s woeful and it’s disrespectful and it’s insulting and I’m sick of it.”
‘America sneezes and Africa catches a cold’
When she first heard the decision to pull the ring from FDA consideration, Yvette Raphael, the South Africa-based executive director of Advocates for the Prevention of HIV in Africa, started asking, “What can we do to help our sisters in America get this ring?” And then she started worrying about other women in her own country and those nearby.
“The FDA plays a big role,” she said. “You know, America sneezes and Africa catches a cold.”
She worries that IPM’s decision to withdraw the ring from FDA consideration will signal to regulators in other countries either (a) that they should not approve it or (b) in countries where it’s already been approved but guidelines have not been issued, that they won’t invest money in rolling it out to women in those countries – especially now with the U.S. approval of the prevention shot. In much of Africa, ministries of health prefer to provide injectable contraception, often giving women few or no other options. But women, she said, think about more than administration of the drug. They look at if it’s an easier option for them to manage.
“This is a long journey, an emotional one too, for women in South Africa, because the idea of a microbicide is one of the ideas that came directly from women in South Africa,” she said. “[The jab] can be seen as a solution to all. We can just give jabs to all the women. And after all, we know that women don’t adhere, so we can just grab them.”
Dr. Rosenberg pointed to the positive opinion from the EMA as another “rigorous review” process that she said ought to equally influence ministries of health in countries where women tested the ring. And she pointed to the WHO statement released last month, the same day as IPM’s announcement that it was withdrawing the ring from FDA considerations, recommitting the ring as a good option in sub-Saharan Africa: “The U.S. FDA decision is not based on any new or additional data on efficacy and safety,” it stated. “WHO will continue to support countries as they consider whether to include the [dapivirine vaginal ring]. WHO recognizes that country decisionmaking will vary based on their context and that women’s voices remain central to discussions about their prevention choices.”
Dual action ring on the horizon, but not in U.S.
What this means, though, is that the next step in the ring’s development – the combination dapivirine ring with contraceptive levonorgestrel (used in the Mirena intrauterine device) – may not come to the U.S., at least for a long while.
“It’s not out of the question,” Dr. Rosenberg said of conducting HIV/pregnancy prevention ring trials in the U.S. “But without the approval of the dapivirine-only ring by FDA, I imagine they would want to see new efficacy data on dapivirine. That is a very difficult hill to climb. There would have to be an active control group [using oral PrEP or injectable cabotegravir], and it would be very difficult for the dapivirine ring to be able to go head-to-head for either noninferiority and certainly for superiority.”
The study would need to be quite large to get enough results to prove anything, and IPM is a research organization, not a large pharmaceutical company with deep enough pockets to fund that, she said. Raising those funds “would be difficult.”
In addition to NIAID discontinuing its funding for the Microbicides Trials Network, a new 5-year, $85 million research collaboration through USAID hasn’t slated any money to fund trials of the combination HIV prevention and contraceptive ring, according to Dr. Rosenberg.
But that doesn’t mean avenues for its development are closed. NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) is currently funding a phase 1/2 trial of the combination ring, and IPM continues to receive funding from research agencies in Germany, the Netherlands, Denmark, and Ireland. And this means, she said, that the E.U. – not the U.S. – is where they would seek approval for a combination ring first.
That leaves Ms. Rafael and Ms. Diallo debating how to work together to push the FDA – and maybe IPM – to reconsider the ring. For instance, Ms. Diallo suggested that instead of seeking an indication for all women, the FDA might consider the ring for women with very high risk of HIV, such as sex workers or women with HIV positive partners not on treatment. And she said that this has to be bigger than HIV prevention. It has to be about the ways in which women’s health issues in general lag at the FDA. For instance, she pointed to the movement to get contraceptive pills available over the counter, fights against FDA rulings on hormone replacement therapy, and fights for emergency contraception.
In the meantime, ob/gyn Dr. Rogers is expecting access to the ring to follow a similar path as the copper IUD, which migrated to the U.S. from Europe, where it has been among the most popular contraceptive methods for women.
“Contrary to what we may think, we are not innovators, especially for something like this,” she said. “Once we see it is working and doing a good job – that women in Europe love it – then someone here is going to pick it up and make it as if it’s the greatest thing. But for now, I think we’re going to have to take a back seat to Europe.”
Ms. Diallo reports receiving fees from Johnson & Johnson, ViiV Healthcare, and Gilead Sciences. Dr. Rosenberg and Dr. Rogers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tosha Rogers, MD, is a one-woman HIV prevention evangelist. For nearly a decade now, the Atlanta-based ob/gyn has been on a mission to increase her gynecological colleagues’ awareness and prescribing of the oral HIV prevention pill. At the same time, she’s been tracking the development of a flexible vaginal ring loaded with a month’s worth of the HIV prevention medication dapivirine. That, she thought, would fit easily into women’s lives and into the toolbox of methods women already use to prevent pregnancy.
But now she’s not sure when – or if – the ring will find its way to her patients. In December, the ring’s maker, the International Partnership for Microbicides (IPM), pulled its application for FDA approval for the pre-exposure prophylaxis (PrEP) ring. Now, one year after the World Health Organization recommended the ring for member nations, there appears to be no path forward in the United States for either the dapivirine-only ring or an approach Dr. Rogers said would change the game: a vaginal ring that supplies both contraception and HIV prevention.
“It would take things to a whole other level,” she said. “It sucks that this happened, and I do think it was not anything medical. I think it was everything political.”
That leaves cisgender women – especially the Black and Latinx women who make up the vast majority of women who acquire HIV every year – with two HIV prevention options. One is the daily pill, first approved in 2012. It’s now generic but previously sold as Truvada by Gilead Sciences. The other is monthly injectable cabotegravir long-acting (Apretude). Another HIV prevention pill, tenofovir alafenamide/emtricitabine (Descovy), is approved for gay men and transgender women but not cisgender women.
Vagina-specific protection from HIV
The WHO recommendation for the vaginal ring was followed last July by a positive opinion from the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) for women in low- and middle-income countries outside the European Union.
The flexible silicone ring, similar to the hormonal NuvaRing contraceptive, works by slowly releasing the antiretroviral dapivirine directly into the vaginal canal, thereby protecting women who might be exposed to the virus through vaginal sex only. Because the medicine stays where it’s delivered and doesn’t circulate through the body, it has been found to be extremely safe with few adverse events.
However, in initial studies, the ring was found to be just 27% effective overall. Later studies, where scientists divided women by how much drug was missing from the ring – a proxy for use – found that higher use was associated with higher protection (as much as 54%). By comparison, Truvada has been found to be up to 99% effective when used daily, though it can take up to 21 days to be available in the vagina in high enough concentrations to protect women from vaginal exposure. And the HIV prevention shot was found to be 90% more effective than that in a recent trial of the two methods conducted by the HIV Prevention Trials Network.
This, and an orientation away from topical HIV prevention drugs and toward systemic options, led the National Institute of Allergy and Infectious Diseases (NIAID) to discontinue funding for such projects under its Microbicide Trials Network.
“Clearly you want to counsel women to use the highest efficacy method, and that is part of our label,” Zeda Rosenberg, ScD, IPM’s founder and chief executive officer, told this news organization. “Women should not choose the ring if they can and will use oral PrEP, and I would argue it should be the same thing for [cabotegravir shots]. But if they can’t or don’t want to – and we know that especially many young women don’t want to use systemic methods – then the dapivirine ring is a great option.”
Still, Dr. Rosenberg said that the gap in efficacy, the relatively small number of women affected by HIV in the U.S. compared with gay and bisexual men, and the emergence of products like the HIV prevention shot cabotegravir, made it “very unlikely” that FDA regulators would approve the ring. And rather than be “distracted” by the FDA process, Dr. Rosenberg said IPM chose to concentrate on the countries where the ring has already been approved or where women make up the vast majority of people affected by HIV.
Zimbabwe publicly announced it has approved the ring, and three other countries may have approved it, according to Dr. Rosenberg. She declined to name them, saying they had requested silence while they formulate their new HIV prevention guidelines. Aside from Zimbabwe, the other countries where women participated in the ring clinical trials were South Africa, Malawi, and Uganda.
“The U.S. population ... has widespread access to oral PrEP, which is unlike countries in Africa, and which would have widespread access to injectable cabotegravir,” she said. “The U.S. FDA may not see choice in the same way that African women and African activists and advocates see the need for choice.”
But women’s rates of accessing HIV prevention medications in the U.S. continues to be frustratingly low. At the end of 2018, just 7% of women who could benefit from HIV prevention drugs were taking them, according to Centers for Disease Control and Prevention data.
New CDC guidelines recommend clinicians talk to every sexually active adult and adolescent about HIV prevention medications at least once and prescribe it to anyone who asks for it, whether or not they understand their patients’ HIV risks. However, research continues to show that clinicians struggle with willingness to prescribe PrEP to Black women, and the American College of Obstetrics and Gynecology’s committee opinion on managing women using HIV prevention drugs has not been updated to reflect the new guidelines. And while the HIV prevention shot is approved for women and its maker ViiV Healthcare is already initiating postmarket studies of the ring in key populations including women, there are lots of things that need to line up in order for clinicians to be willing to stock it and prescribe it to women.
From where Dázon Dixon Diallo, executive director of the nonprofit SisterLove, sits, the decision to withdraw the ring from FDA consideration and the FDA’s seeming argument that the epidemiology in the U.S. doesn’t warrant the ring’s approval is a slap in the face to the Black women who have led the movement to end HIV in the U.S. for decades.
“No matter how you slice it, we’re talking about Black women, and then we’re talking about brown women,” said Ms. Diallo. “The value [they place on us] from a government standpoint, from a political standpoint, from a public health standpoint is just woeful. It’s woeful and it’s disrespectful and it’s insulting and I’m sick of it.”
‘America sneezes and Africa catches a cold’
When she first heard the decision to pull the ring from FDA consideration, Yvette Raphael, the South Africa-based executive director of Advocates for the Prevention of HIV in Africa, started asking, “What can we do to help our sisters in America get this ring?” And then she started worrying about other women in her own country and those nearby.
“The FDA plays a big role,” she said. “You know, America sneezes and Africa catches a cold.”
She worries that IPM’s decision to withdraw the ring from FDA consideration will signal to regulators in other countries either (a) that they should not approve it or (b) in countries where it’s already been approved but guidelines have not been issued, that they won’t invest money in rolling it out to women in those countries – especially now with the U.S. approval of the prevention shot. In much of Africa, ministries of health prefer to provide injectable contraception, often giving women few or no other options. But women, she said, think about more than administration of the drug. They look at if it’s an easier option for them to manage.
“This is a long journey, an emotional one too, for women in South Africa, because the idea of a microbicide is one of the ideas that came directly from women in South Africa,” she said. “[The jab] can be seen as a solution to all. We can just give jabs to all the women. And after all, we know that women don’t adhere, so we can just grab them.”
Dr. Rosenberg pointed to the positive opinion from the EMA as another “rigorous review” process that she said ought to equally influence ministries of health in countries where women tested the ring. And she pointed to the WHO statement released last month, the same day as IPM’s announcement that it was withdrawing the ring from FDA considerations, recommitting the ring as a good option in sub-Saharan Africa: “The U.S. FDA decision is not based on any new or additional data on efficacy and safety,” it stated. “WHO will continue to support countries as they consider whether to include the [dapivirine vaginal ring]. WHO recognizes that country decisionmaking will vary based on their context and that women’s voices remain central to discussions about their prevention choices.”
Dual action ring on the horizon, but not in U.S.
What this means, though, is that the next step in the ring’s development – the combination dapivirine ring with contraceptive levonorgestrel (used in the Mirena intrauterine device) – may not come to the U.S., at least for a long while.
“It’s not out of the question,” Dr. Rosenberg said of conducting HIV/pregnancy prevention ring trials in the U.S. “But without the approval of the dapivirine-only ring by FDA, I imagine they would want to see new efficacy data on dapivirine. That is a very difficult hill to climb. There would have to be an active control group [using oral PrEP or injectable cabotegravir], and it would be very difficult for the dapivirine ring to be able to go head-to-head for either noninferiority and certainly for superiority.”
The study would need to be quite large to get enough results to prove anything, and IPM is a research organization, not a large pharmaceutical company with deep enough pockets to fund that, she said. Raising those funds “would be difficult.”
In addition to NIAID discontinuing its funding for the Microbicides Trials Network, a new 5-year, $85 million research collaboration through USAID hasn’t slated any money to fund trials of the combination HIV prevention and contraceptive ring, according to Dr. Rosenberg.
But that doesn’t mean avenues for its development are closed. NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) is currently funding a phase 1/2 trial of the combination ring, and IPM continues to receive funding from research agencies in Germany, the Netherlands, Denmark, and Ireland. And this means, she said, that the E.U. – not the U.S. – is where they would seek approval for a combination ring first.
That leaves Ms. Rafael and Ms. Diallo debating how to work together to push the FDA – and maybe IPM – to reconsider the ring. For instance, Ms. Diallo suggested that instead of seeking an indication for all women, the FDA might consider the ring for women with very high risk of HIV, such as sex workers or women with HIV positive partners not on treatment. And she said that this has to be bigger than HIV prevention. It has to be about the ways in which women’s health issues in general lag at the FDA. For instance, she pointed to the movement to get contraceptive pills available over the counter, fights against FDA rulings on hormone replacement therapy, and fights for emergency contraception.
In the meantime, ob/gyn Dr. Rogers is expecting access to the ring to follow a similar path as the copper IUD, which migrated to the U.S. from Europe, where it has been among the most popular contraceptive methods for women.
“Contrary to what we may think, we are not innovators, especially for something like this,” she said. “Once we see it is working and doing a good job – that women in Europe love it – then someone here is going to pick it up and make it as if it’s the greatest thing. But for now, I think we’re going to have to take a back seat to Europe.”
Ms. Diallo reports receiving fees from Johnson & Johnson, ViiV Healthcare, and Gilead Sciences. Dr. Rosenberg and Dr. Rogers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tosha Rogers, MD, is a one-woman HIV prevention evangelist. For nearly a decade now, the Atlanta-based ob/gyn has been on a mission to increase her gynecological colleagues’ awareness and prescribing of the oral HIV prevention pill. At the same time, she’s been tracking the development of a flexible vaginal ring loaded with a month’s worth of the HIV prevention medication dapivirine. That, she thought, would fit easily into women’s lives and into the toolbox of methods women already use to prevent pregnancy.
But now she’s not sure when – or if – the ring will find its way to her patients. In December, the ring’s maker, the International Partnership for Microbicides (IPM), pulled its application for FDA approval for the pre-exposure prophylaxis (PrEP) ring. Now, one year after the World Health Organization recommended the ring for member nations, there appears to be no path forward in the United States for either the dapivirine-only ring or an approach Dr. Rogers said would change the game: a vaginal ring that supplies both contraception and HIV prevention.
“It would take things to a whole other level,” she said. “It sucks that this happened, and I do think it was not anything medical. I think it was everything political.”
That leaves cisgender women – especially the Black and Latinx women who make up the vast majority of women who acquire HIV every year – with two HIV prevention options. One is the daily pill, first approved in 2012. It’s now generic but previously sold as Truvada by Gilead Sciences. The other is monthly injectable cabotegravir long-acting (Apretude). Another HIV prevention pill, tenofovir alafenamide/emtricitabine (Descovy), is approved for gay men and transgender women but not cisgender women.
Vagina-specific protection from HIV
The WHO recommendation for the vaginal ring was followed last July by a positive opinion from the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) for women in low- and middle-income countries outside the European Union.
The flexible silicone ring, similar to the hormonal NuvaRing contraceptive, works by slowly releasing the antiretroviral dapivirine directly into the vaginal canal, thereby protecting women who might be exposed to the virus through vaginal sex only. Because the medicine stays where it’s delivered and doesn’t circulate through the body, it has been found to be extremely safe with few adverse events.
However, in initial studies, the ring was found to be just 27% effective overall. Later studies, where scientists divided women by how much drug was missing from the ring – a proxy for use – found that higher use was associated with higher protection (as much as 54%). By comparison, Truvada has been found to be up to 99% effective when used daily, though it can take up to 21 days to be available in the vagina in high enough concentrations to protect women from vaginal exposure. And the HIV prevention shot was found to be 90% more effective than that in a recent trial of the two methods conducted by the HIV Prevention Trials Network.
This, and an orientation away from topical HIV prevention drugs and toward systemic options, led the National Institute of Allergy and Infectious Diseases (NIAID) to discontinue funding for such projects under its Microbicide Trials Network.
“Clearly you want to counsel women to use the highest efficacy method, and that is part of our label,” Zeda Rosenberg, ScD, IPM’s founder and chief executive officer, told this news organization. “Women should not choose the ring if they can and will use oral PrEP, and I would argue it should be the same thing for [cabotegravir shots]. But if they can’t or don’t want to – and we know that especially many young women don’t want to use systemic methods – then the dapivirine ring is a great option.”
Still, Dr. Rosenberg said that the gap in efficacy, the relatively small number of women affected by HIV in the U.S. compared with gay and bisexual men, and the emergence of products like the HIV prevention shot cabotegravir, made it “very unlikely” that FDA regulators would approve the ring. And rather than be “distracted” by the FDA process, Dr. Rosenberg said IPM chose to concentrate on the countries where the ring has already been approved or where women make up the vast majority of people affected by HIV.
Zimbabwe publicly announced it has approved the ring, and three other countries may have approved it, according to Dr. Rosenberg. She declined to name them, saying they had requested silence while they formulate their new HIV prevention guidelines. Aside from Zimbabwe, the other countries where women participated in the ring clinical trials were South Africa, Malawi, and Uganda.
“The U.S. population ... has widespread access to oral PrEP, which is unlike countries in Africa, and which would have widespread access to injectable cabotegravir,” she said. “The U.S. FDA may not see choice in the same way that African women and African activists and advocates see the need for choice.”
But women’s rates of accessing HIV prevention medications in the U.S. continues to be frustratingly low. At the end of 2018, just 7% of women who could benefit from HIV prevention drugs were taking them, according to Centers for Disease Control and Prevention data.
New CDC guidelines recommend clinicians talk to every sexually active adult and adolescent about HIV prevention medications at least once and prescribe it to anyone who asks for it, whether or not they understand their patients’ HIV risks. However, research continues to show that clinicians struggle with willingness to prescribe PrEP to Black women, and the American College of Obstetrics and Gynecology’s committee opinion on managing women using HIV prevention drugs has not been updated to reflect the new guidelines. And while the HIV prevention shot is approved for women and its maker ViiV Healthcare is already initiating postmarket studies of the ring in key populations including women, there are lots of things that need to line up in order for clinicians to be willing to stock it and prescribe it to women.
From where Dázon Dixon Diallo, executive director of the nonprofit SisterLove, sits, the decision to withdraw the ring from FDA consideration and the FDA’s seeming argument that the epidemiology in the U.S. doesn’t warrant the ring’s approval is a slap in the face to the Black women who have led the movement to end HIV in the U.S. for decades.
“No matter how you slice it, we’re talking about Black women, and then we’re talking about brown women,” said Ms. Diallo. “The value [they place on us] from a government standpoint, from a political standpoint, from a public health standpoint is just woeful. It’s woeful and it’s disrespectful and it’s insulting and I’m sick of it.”
‘America sneezes and Africa catches a cold’
When she first heard the decision to pull the ring from FDA consideration, Yvette Raphael, the South Africa-based executive director of Advocates for the Prevention of HIV in Africa, started asking, “What can we do to help our sisters in America get this ring?” And then she started worrying about other women in her own country and those nearby.
“The FDA plays a big role,” she said. “You know, America sneezes and Africa catches a cold.”
She worries that IPM’s decision to withdraw the ring from FDA consideration will signal to regulators in other countries either (a) that they should not approve it or (b) in countries where it’s already been approved but guidelines have not been issued, that they won’t invest money in rolling it out to women in those countries – especially now with the U.S. approval of the prevention shot. In much of Africa, ministries of health prefer to provide injectable contraception, often giving women few or no other options. But women, she said, think about more than administration of the drug. They look at if it’s an easier option for them to manage.
“This is a long journey, an emotional one too, for women in South Africa, because the idea of a microbicide is one of the ideas that came directly from women in South Africa,” she said. “[The jab] can be seen as a solution to all. We can just give jabs to all the women. And after all, we know that women don’t adhere, so we can just grab them.”
Dr. Rosenberg pointed to the positive opinion from the EMA as another “rigorous review” process that she said ought to equally influence ministries of health in countries where women tested the ring. And she pointed to the WHO statement released last month, the same day as IPM’s announcement that it was withdrawing the ring from FDA considerations, recommitting the ring as a good option in sub-Saharan Africa: “The U.S. FDA decision is not based on any new or additional data on efficacy and safety,” it stated. “WHO will continue to support countries as they consider whether to include the [dapivirine vaginal ring]. WHO recognizes that country decisionmaking will vary based on their context and that women’s voices remain central to discussions about their prevention choices.”
Dual action ring on the horizon, but not in U.S.
What this means, though, is that the next step in the ring’s development – the combination dapivirine ring with contraceptive levonorgestrel (used in the Mirena intrauterine device) – may not come to the U.S., at least for a long while.
“It’s not out of the question,” Dr. Rosenberg said of conducting HIV/pregnancy prevention ring trials in the U.S. “But without the approval of the dapivirine-only ring by FDA, I imagine they would want to see new efficacy data on dapivirine. That is a very difficult hill to climb. There would have to be an active control group [using oral PrEP or injectable cabotegravir], and it would be very difficult for the dapivirine ring to be able to go head-to-head for either noninferiority and certainly for superiority.”
The study would need to be quite large to get enough results to prove anything, and IPM is a research organization, not a large pharmaceutical company with deep enough pockets to fund that, she said. Raising those funds “would be difficult.”
In addition to NIAID discontinuing its funding for the Microbicides Trials Network, a new 5-year, $85 million research collaboration through USAID hasn’t slated any money to fund trials of the combination HIV prevention and contraceptive ring, according to Dr. Rosenberg.
But that doesn’t mean avenues for its development are closed. NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) is currently funding a phase 1/2 trial of the combination ring, and IPM continues to receive funding from research agencies in Germany, the Netherlands, Denmark, and Ireland. And this means, she said, that the E.U. – not the U.S. – is where they would seek approval for a combination ring first.
That leaves Ms. Rafael and Ms. Diallo debating how to work together to push the FDA – and maybe IPM – to reconsider the ring. For instance, Ms. Diallo suggested that instead of seeking an indication for all women, the FDA might consider the ring for women with very high risk of HIV, such as sex workers or women with HIV positive partners not on treatment. And she said that this has to be bigger than HIV prevention. It has to be about the ways in which women’s health issues in general lag at the FDA. For instance, she pointed to the movement to get contraceptive pills available over the counter, fights against FDA rulings on hormone replacement therapy, and fights for emergency contraception.
In the meantime, ob/gyn Dr. Rogers is expecting access to the ring to follow a similar path as the copper IUD, which migrated to the U.S. from Europe, where it has been among the most popular contraceptive methods for women.
“Contrary to what we may think, we are not innovators, especially for something like this,” she said. “Once we see it is working and doing a good job – that women in Europe love it – then someone here is going to pick it up and make it as if it’s the greatest thing. But for now, I think we’re going to have to take a back seat to Europe.”
Ms. Diallo reports receiving fees from Johnson & Johnson, ViiV Healthcare, and Gilead Sciences. Dr. Rosenberg and Dr. Rogers have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Children and COVID: United States passes 10 million total cases
Weekly COVID-19 cases in children topped 1 million for the first time as the cumulative count surpassed 10 million since the start of the pandemic, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
the AAP and CHA said in their weekly COVID report. Those 10.6 million child cases represent 18.4% of all cases, and the latest 1.15 million represented 25.5% of all cases for the week.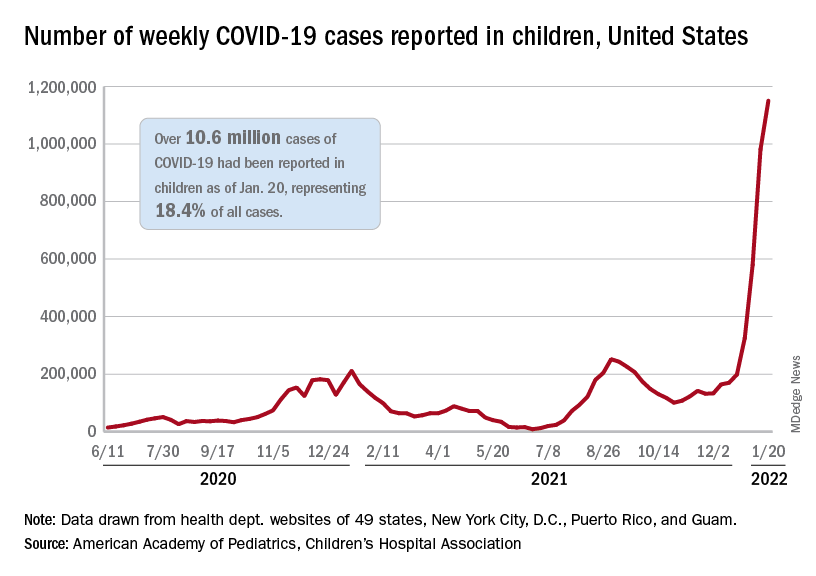
Regionally, the South had the most cases with over 380,000 for the week of Jan. 14-20, while the West was next with close to 350,000, followed by the Midwest and then the East. Among the states, the largest percent increases – on the order of 30% – came in New England (Massachusetts, Rhode Island, and Vermont), as well as Virginia and California, the AAP and CHA said.
Examining all those cases by vaccination status shows an obvious difference between the Omicron and Delta variants: The fully vaccinated have been hit much harder than before. For the week ending Dec. 25, 2021, the incidence of COVID-19 in children aged 12-17 years was 704 per 100,000 among those were unvaccinated and 384 per 100,000 in those who were fully vaccinated. During the Delta surge in the summer of 2021, the peak rates were 938 (unvaccinated) and 79 (vaccinated), the Centers for Disease Control and Prevention said.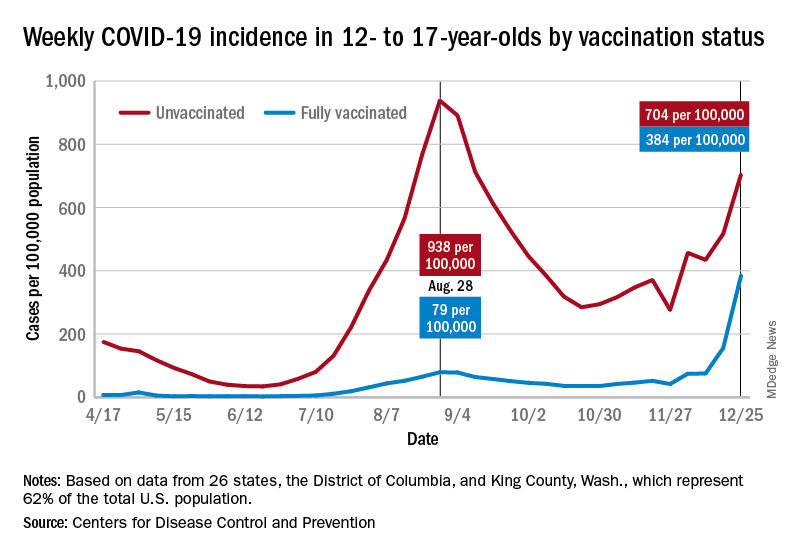
Hospitalizations are also at record levels, but two separate CDC databases seem to show a decline in child admissions over the last available week or so of data, which follows the trend among all ages. The peak among children aged 0-17 years came on Jan. 15, when the rate of new admissions reached 1.25 per 100,000, based on reporting to the CDC from 5,265 hospitals nationwide.
The second database, the COVID-19–Associated Hospitalization Surveillance Network (COVID-NET), indicates that children aged 0-4 years had the highest admission rate, 14.5 per 100,000, for the week ending Jan. 8, compared with 5.5 per 100,000 for 12- to 17-year-olds and 2.3 per 100,000 for those aged 5-11 years. COVID-NET covers almost 100 counties in 10 states, along with 4 entire states, and represents about 10% of the U.S. population.
Vaccinations rose briefly in late December and into January to meet the Omicron surge, but the numbers for the latest week show a return to their earlier levels. In children aged 5-11 years, new vaccinations went from 381,000 for the week of Dec. 20-26 to 524,000 for Jan. 3-9, but fell to just 260,000 during Jan. 17-23. The response was a little later for those aged 12-17, with the big week coming Jan. 10-16, but there was still a 38% drop for Jan. 17-23, according to the CDC’s COVID Data Tracker.
Currently, 29.3% of all 5- to 11-year-olds have received at least one dose of the COVID vaccine, and an even 20.0% are fully vaccinated. For children aged 12-17, the corresponding figures are 65.8% and 55.1%, the CDC said.
Statewide vaccination rates vary from Vermont’s high of 61% for those aged 5-11 to 12% for Alabama, Louisiana, and Mississippi, while Hawaii has the highest rate for 12- to 17-year-olds at 92% and Wyoming has the lowest at 39%, the AAP reported.
Weekly COVID-19 cases in children topped 1 million for the first time as the cumulative count surpassed 10 million since the start of the pandemic, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
the AAP and CHA said in their weekly COVID report. Those 10.6 million child cases represent 18.4% of all cases, and the latest 1.15 million represented 25.5% of all cases for the week.
Regionally, the South had the most cases with over 380,000 for the week of Jan. 14-20, while the West was next with close to 350,000, followed by the Midwest and then the East. Among the states, the largest percent increases – on the order of 30% – came in New England (Massachusetts, Rhode Island, and Vermont), as well as Virginia and California, the AAP and CHA said.
Examining all those cases by vaccination status shows an obvious difference between the Omicron and Delta variants: The fully vaccinated have been hit much harder than before. For the week ending Dec. 25, 2021, the incidence of COVID-19 in children aged 12-17 years was 704 per 100,000 among those were unvaccinated and 384 per 100,000 in those who were fully vaccinated. During the Delta surge in the summer of 2021, the peak rates were 938 (unvaccinated) and 79 (vaccinated), the Centers for Disease Control and Prevention said.
Hospitalizations are also at record levels, but two separate CDC databases seem to show a decline in child admissions over the last available week or so of data, which follows the trend among all ages. The peak among children aged 0-17 years came on Jan. 15, when the rate of new admissions reached 1.25 per 100,000, based on reporting to the CDC from 5,265 hospitals nationwide.
The second database, the COVID-19–Associated Hospitalization Surveillance Network (COVID-NET), indicates that children aged 0-4 years had the highest admission rate, 14.5 per 100,000, for the week ending Jan. 8, compared with 5.5 per 100,000 for 12- to 17-year-olds and 2.3 per 100,000 for those aged 5-11 years. COVID-NET covers almost 100 counties in 10 states, along with 4 entire states, and represents about 10% of the U.S. population.
Vaccinations rose briefly in late December and into January to meet the Omicron surge, but the numbers for the latest week show a return to their earlier levels. In children aged 5-11 years, new vaccinations went from 381,000 for the week of Dec. 20-26 to 524,000 for Jan. 3-9, but fell to just 260,000 during Jan. 17-23. The response was a little later for those aged 12-17, with the big week coming Jan. 10-16, but there was still a 38% drop for Jan. 17-23, according to the CDC’s COVID Data Tracker.
Currently, 29.3% of all 5- to 11-year-olds have received at least one dose of the COVID vaccine, and an even 20.0% are fully vaccinated. For children aged 12-17, the corresponding figures are 65.8% and 55.1%, the CDC said.
Statewide vaccination rates vary from Vermont’s high of 61% for those aged 5-11 to 12% for Alabama, Louisiana, and Mississippi, while Hawaii has the highest rate for 12- to 17-year-olds at 92% and Wyoming has the lowest at 39%, the AAP reported.
Weekly COVID-19 cases in children topped 1 million for the first time as the cumulative count surpassed 10 million since the start of the pandemic, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
the AAP and CHA said in their weekly COVID report. Those 10.6 million child cases represent 18.4% of all cases, and the latest 1.15 million represented 25.5% of all cases for the week.
Regionally, the South had the most cases with over 380,000 for the week of Jan. 14-20, while the West was next with close to 350,000, followed by the Midwest and then the East. Among the states, the largest percent increases – on the order of 30% – came in New England (Massachusetts, Rhode Island, and Vermont), as well as Virginia and California, the AAP and CHA said.
Examining all those cases by vaccination status shows an obvious difference between the Omicron and Delta variants: The fully vaccinated have been hit much harder than before. For the week ending Dec. 25, 2021, the incidence of COVID-19 in children aged 12-17 years was 704 per 100,000 among those were unvaccinated and 384 per 100,000 in those who were fully vaccinated. During the Delta surge in the summer of 2021, the peak rates were 938 (unvaccinated) and 79 (vaccinated), the Centers for Disease Control and Prevention said.
Hospitalizations are also at record levels, but two separate CDC databases seem to show a decline in child admissions over the last available week or so of data, which follows the trend among all ages. The peak among children aged 0-17 years came on Jan. 15, when the rate of new admissions reached 1.25 per 100,000, based on reporting to the CDC from 5,265 hospitals nationwide.
The second database, the COVID-19–Associated Hospitalization Surveillance Network (COVID-NET), indicates that children aged 0-4 years had the highest admission rate, 14.5 per 100,000, for the week ending Jan. 8, compared with 5.5 per 100,000 for 12- to 17-year-olds and 2.3 per 100,000 for those aged 5-11 years. COVID-NET covers almost 100 counties in 10 states, along with 4 entire states, and represents about 10% of the U.S. population.
Vaccinations rose briefly in late December and into January to meet the Omicron surge, but the numbers for the latest week show a return to their earlier levels. In children aged 5-11 years, new vaccinations went from 381,000 for the week of Dec. 20-26 to 524,000 for Jan. 3-9, but fell to just 260,000 during Jan. 17-23. The response was a little later for those aged 12-17, with the big week coming Jan. 10-16, but there was still a 38% drop for Jan. 17-23, according to the CDC’s COVID Data Tracker.
Currently, 29.3% of all 5- to 11-year-olds have received at least one dose of the COVID vaccine, and an even 20.0% are fully vaccinated. For children aged 12-17, the corresponding figures are 65.8% and 55.1%, the CDC said.
Statewide vaccination rates vary from Vermont’s high of 61% for those aged 5-11 to 12% for Alabama, Louisiana, and Mississippi, while Hawaii has the highest rate for 12- to 17-year-olds at 92% and Wyoming has the lowest at 39%, the AAP reported.
Watch, but don’t worry yet, about new Omicron subvariant
In the meantime, it’s worth watching BA.2, the World Health Organization said. The subvariant has been identified across at least 40 countries, including three cases reported in Houston and several in Washington state.
BA.2 accounts for only a small minority of reported cases so far, including 5% in India, 4% of those in the United Kingdom, and 2% each of cases in Sweden and Singapore.
The one exception is Denmark, a country with robust genetic sequencing abilities, where estimates range from 50% to 81% of cases.
The news throws a little more uncertainty into an already uncertain situation, including how close the world might be to a less life-altering infectious disease.
For example, the world is at an ideal point for a new variant to emerge, WHO Director General Tedros Adhanom Ghebreyesus, PhD, said during a Jan. 24 meeting of the WHO executive board. He also said it’s too early to call an “end game” to the pandemic.
Similarly, Anthony S. Fauci, MD, said on Jan. 19 that it remained “an open question” whether the Omicron variant could hasten endemic COVID-19, a situation where the virus still circulates but is much less disruptive to everyday life.
No Pi for you
This could be the first time a coronavirus subvariant rises to the level of a household name, or – if previous variants of the moment have shown us – it could recede from the spotlight.
For example, a lot of focus on the potential of the Mu variant to wreak havoc fizzled out a few weeks after the WHO listed it as a variant of interest on Aug. 30.
Subvariants can feature mutations and other small differences but are not distinct enough from an existing strain to be called a variant on their own and be named after the next letter in the Greek alphabet. That’s why BA.2 is not called the “Pi variant.”
Predicting what’s next for the coronavirus has puzzled many experts throughout the pandemic. That is why many public health officials wait for the WHO to officially designate a strain as a variant of interest or variant of concern before taking action.
At the moment with BA.2, it seems close monitoring is warranted.
Because it’s too early to call, expert predictions about BA.2 vary widely, from worry to cautious optimism.
For example, early data indicates that BA.2 could be more worrisome than original Omicron, Eric Feigl-Ding, ScD, an epidemiologist and health economist, said on Twitter.
Information from Denmark seems to show BA.2 either has “much faster transmission or it evades immunity even more,” he said.
The same day, Jan. 23, Dr. Feigl-Ding tweeted that other data shows the subvariant can spread twice as fast as Omicron, which was already much more contagious than previous versions of the virus.
At the same time, other experts appear less concerned. Robert Garry, PhD, a virologist at Tulane University, New Orleans, told the Washington Post that there is no reason to think BA.2 will be any worse than the original Omicron strain.
So which expert predictions will come closer to BA.2’s potential? For now, it’s just a watch-and-see situation.
For updated information, the website outbreak.info tracks BA.2’s average daily and cumulative prevalence in the United States and in other locations.
Also, if and when WHO experts decide to elevate BA.2 to a variant of interest or a variant of concern, it will be noted on its coronavirus variant tracking website.
A version of this article first appeared on WebMD.com.
In the meantime, it’s worth watching BA.2, the World Health Organization said. The subvariant has been identified across at least 40 countries, including three cases reported in Houston and several in Washington state.
BA.2 accounts for only a small minority of reported cases so far, including 5% in India, 4% of those in the United Kingdom, and 2% each of cases in Sweden and Singapore.
The one exception is Denmark, a country with robust genetic sequencing abilities, where estimates range from 50% to 81% of cases.
The news throws a little more uncertainty into an already uncertain situation, including how close the world might be to a less life-altering infectious disease.
For example, the world is at an ideal point for a new variant to emerge, WHO Director General Tedros Adhanom Ghebreyesus, PhD, said during a Jan. 24 meeting of the WHO executive board. He also said it’s too early to call an “end game” to the pandemic.
Similarly, Anthony S. Fauci, MD, said on Jan. 19 that it remained “an open question” whether the Omicron variant could hasten endemic COVID-19, a situation where the virus still circulates but is much less disruptive to everyday life.
No Pi for you
This could be the first time a coronavirus subvariant rises to the level of a household name, or – if previous variants of the moment have shown us – it could recede from the spotlight.
For example, a lot of focus on the potential of the Mu variant to wreak havoc fizzled out a few weeks after the WHO listed it as a variant of interest on Aug. 30.
Subvariants can feature mutations and other small differences but are not distinct enough from an existing strain to be called a variant on their own and be named after the next letter in the Greek alphabet. That’s why BA.2 is not called the “Pi variant.”
Predicting what’s next for the coronavirus has puzzled many experts throughout the pandemic. That is why many public health officials wait for the WHO to officially designate a strain as a variant of interest or variant of concern before taking action.
At the moment with BA.2, it seems close monitoring is warranted.
Because it’s too early to call, expert predictions about BA.2 vary widely, from worry to cautious optimism.
For example, early data indicates that BA.2 could be more worrisome than original Omicron, Eric Feigl-Ding, ScD, an epidemiologist and health economist, said on Twitter.
Information from Denmark seems to show BA.2 either has “much faster transmission or it evades immunity even more,” he said.
The same day, Jan. 23, Dr. Feigl-Ding tweeted that other data shows the subvariant can spread twice as fast as Omicron, which was already much more contagious than previous versions of the virus.
At the same time, other experts appear less concerned. Robert Garry, PhD, a virologist at Tulane University, New Orleans, told the Washington Post that there is no reason to think BA.2 will be any worse than the original Omicron strain.
So which expert predictions will come closer to BA.2’s potential? For now, it’s just a watch-and-see situation.
For updated information, the website outbreak.info tracks BA.2’s average daily and cumulative prevalence in the United States and in other locations.
Also, if and when WHO experts decide to elevate BA.2 to a variant of interest or a variant of concern, it will be noted on its coronavirus variant tracking website.
A version of this article first appeared on WebMD.com.
In the meantime, it’s worth watching BA.2, the World Health Organization said. The subvariant has been identified across at least 40 countries, including three cases reported in Houston and several in Washington state.
BA.2 accounts for only a small minority of reported cases so far, including 5% in India, 4% of those in the United Kingdom, and 2% each of cases in Sweden and Singapore.
The one exception is Denmark, a country with robust genetic sequencing abilities, where estimates range from 50% to 81% of cases.
The news throws a little more uncertainty into an already uncertain situation, including how close the world might be to a less life-altering infectious disease.
For example, the world is at an ideal point for a new variant to emerge, WHO Director General Tedros Adhanom Ghebreyesus, PhD, said during a Jan. 24 meeting of the WHO executive board. He also said it’s too early to call an “end game” to the pandemic.
Similarly, Anthony S. Fauci, MD, said on Jan. 19 that it remained “an open question” whether the Omicron variant could hasten endemic COVID-19, a situation where the virus still circulates but is much less disruptive to everyday life.
No Pi for you
This could be the first time a coronavirus subvariant rises to the level of a household name, or – if previous variants of the moment have shown us – it could recede from the spotlight.
For example, a lot of focus on the potential of the Mu variant to wreak havoc fizzled out a few weeks after the WHO listed it as a variant of interest on Aug. 30.
Subvariants can feature mutations and other small differences but are not distinct enough from an existing strain to be called a variant on their own and be named after the next letter in the Greek alphabet. That’s why BA.2 is not called the “Pi variant.”
Predicting what’s next for the coronavirus has puzzled many experts throughout the pandemic. That is why many public health officials wait for the WHO to officially designate a strain as a variant of interest or variant of concern before taking action.
At the moment with BA.2, it seems close monitoring is warranted.
Because it’s too early to call, expert predictions about BA.2 vary widely, from worry to cautious optimism.
For example, early data indicates that BA.2 could be more worrisome than original Omicron, Eric Feigl-Ding, ScD, an epidemiologist and health economist, said on Twitter.
Information from Denmark seems to show BA.2 either has “much faster transmission or it evades immunity even more,” he said.
The same day, Jan. 23, Dr. Feigl-Ding tweeted that other data shows the subvariant can spread twice as fast as Omicron, which was already much more contagious than previous versions of the virus.
At the same time, other experts appear less concerned. Robert Garry, PhD, a virologist at Tulane University, New Orleans, told the Washington Post that there is no reason to think BA.2 will be any worse than the original Omicron strain.
So which expert predictions will come closer to BA.2’s potential? For now, it’s just a watch-and-see situation.
For updated information, the website outbreak.info tracks BA.2’s average daily and cumulative prevalence in the United States and in other locations.
Also, if and when WHO experts decide to elevate BA.2 to a variant of interest or a variant of concern, it will be noted on its coronavirus variant tracking website.
A version of this article first appeared on WebMD.com.
This doc still supports NP/PA-led care ... with caveats
Two years ago, I argued that independent care from nurse practitioners (NPs) and physician assistants (PAs) would not have ill effects on health outcomes. To the surprise of no one, NPs and PAs embraced the argument; physicians clobbered it.
My case had three pegs: One was that medicine isn’t rocket science and clinicians control a lot less than we think we do. The second peg was that technology levels the playing field of clinical care. High-sensitivity troponin assays, for instance, make missing MI a lot less likely. The third peg was empirical: Studies have found little difference in MD versus non–MD-led care. Looking back, I now see empiricism as the weakest part of the argument because the studies had so many limitations.
I update this viewpoint now because health care is increasingly delivered by NPs and PAs. And there are two concerning trends regarding NP education and experience. First is that nurses are turning to advanced practitioner training earlier in their careers – without gathering much bedside experience. And these training programs are increasingly likely to be online, with minimal hands-on clinical tutoring.
Education and experience pop in my head often. Not every day, but many days I think back to my lucky 7 years in Indiana learning under the supervision of master clinicians – at a time when trainees were allowed the leeway to make decisions ... and mistakes. Then, when I joined private practice, I continued to learn from experienced practitioners.
It would be foolish to argue that training and experience aren’t important.
But here’s the thing:
I will make three points: First, I will bolster two of my old arguments as to why we shouldn’t be worried about non-MD clinicians, then I will propose some ideas to increase confidence in NP and PA care.
Health care does not equal health
On the matter of how much clinicians affect outcomes, a recently published randomized controlled trial performed in India found that subsidizing insurance care led to increased utilization of hospital services but had no significant effect on health outcomes. This follows the RAND and Oregon Health Insurance studies in the United States, which largely reported similar results.
We should also not dismiss the fact that – despite the massive technology gains over the past half-century in digital health and artificial intelligence and increased use of quality measures, new drugs and procedures, and mega-medical centers – the average lifespan of Americans is flat to declining (in most ethnic and racial groups). Worse than no gains in longevity, perhaps, is that death from diseases like dementia and Parkinson’s disease are on the rise.
A neutral Martian would look down and wonder why all this health care hasn’t translated to longer and better lives. The causes of this paradox remain speculative, and are for another column, but the point remains that – on average – more health care is clearly not delivering more health. And if that is true, one may deduce that much of U.S. health care is marginal when it comes to affecting major outcomes.
It’s about the delta
Logos trumps pathos. Sure, my physician colleagues can tell scary anecdotes of bad outcomes caused by an inexperienced NP or PA. I would counter that by saying I have sat on our hospital’s peer review committee for 2 decades, including the era before NPs or PAs were practicing, and I have plenty of stories of physician errors. These include, of course, my own errors.
Logos: We must consider the difference between non–MD-led care and MD-led care.
My arguments from 2020 remain relevant today. Most medical problems are not engineering puzzles. Many, perhaps most, patients fall into an easy protocol – say, chest pain, dyspnea, or atrial fibrillation. With basic training, a motivated serious person quickly gains skill in recognizing and treating everyday problems.
And just 2 years on, technology further levels the playing field. Consider radiology in 2022 – it’s easy to take for granted the speed of the CT scan, the fidelity of the MRI, and the easy access to both in the U.S. hospital system. Less experienced clinicians have never had more tools to assist with diagnostics and therapeutics.
The expansion of team-based care has also mitigated the effects of inexperience. It took Americans longer than Canadians to figure out how helpful pharmacists could be. Pharmacists in my hospital now help us dose complicated medicines and protect us against prescribing errors.
Then there is the immediate access to online information. Gone are the days when you had to memorize long-QT syndromes. Book knowledge – that I spent years acquiring – now comes in seconds. The other day an NP corrected me. I asked, Are you sure? Boom, she took out her phone and showed me the evidence.
In sum, if it were even possible to measure the clinical competence of care from NP and PA versus physicians, there would be two bell-shaped curves with a tremendous amount of overlap. And that overlap would steadily increase as a given NP or PA gathered experience. (The NP in our electrophysiology division has more than 25 years’ experience in heart rhythm care, and it is common for colleagues to call her before one of us docs. Rightly so.)
Three basic proposals regarding NP and PA care
To ensure quality of care, I have three proposals.
It has always seemed strange to me that an NP or PA can flip from one field to another without a period of training. I can’t just change practice from electrophysiology to dermatology without doing a residency. But NPs and PAs can.
My first proposal would be that NPs and PAs spend a substantial period of training in a field before practice – a legit apprenticeship. The duration of this period is a matter of debate, but it ought to be standardized.
My second proposal is that, if physicians are required to pass certification exams, so should NPs. (PAs have an exam every 10 years.) The exam should be the same as (or very similar to) the physician exam, and it should be specific to their field of practice.
While I have argued (and still feel) that the American Board of Internal Medicine brand of certification is dubious, the fact remains that physicians must maintain proficiency in their field. Requiring NPs and PAs to do the same would help foster specialization. And while I can’t cite empirical evidence, specialization seems super-important. We have NPs at my hospital who have been in the same area for years, and they exude clinical competence.
Finally, I have come to believe that the best way for nearly any clinician to practice medicine is as part of a team. (The exception being primary care in rural areas where there are clinician shortages.)
On the matter of team care, I’ve practiced for a long time, but nearly every day I run situations by a colleague; often this person is an NP. The economist Friedrich Hayek proposed that dispersed knowledge always outpaces the wisdom of any individual. That notion pertains well to the increasing complexities and specialization of modern medical practice.
A person who commits to learning one area of medicine, enjoys helping people, asks often for help, and has the support of colleagues is set up to be a successful clinician – whether the letters after their name are APRN, PA, DO, or MD.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky. He did not report any relevant financial disclosures. A version of this article first appeared on Medscape.com.
Two years ago, I argued that independent care from nurse practitioners (NPs) and physician assistants (PAs) would not have ill effects on health outcomes. To the surprise of no one, NPs and PAs embraced the argument; physicians clobbered it.
My case had three pegs: One was that medicine isn’t rocket science and clinicians control a lot less than we think we do. The second peg was that technology levels the playing field of clinical care. High-sensitivity troponin assays, for instance, make missing MI a lot less likely. The third peg was empirical: Studies have found little difference in MD versus non–MD-led care. Looking back, I now see empiricism as the weakest part of the argument because the studies had so many limitations.
I update this viewpoint now because health care is increasingly delivered by NPs and PAs. And there are two concerning trends regarding NP education and experience. First is that nurses are turning to advanced practitioner training earlier in their careers – without gathering much bedside experience. And these training programs are increasingly likely to be online, with minimal hands-on clinical tutoring.
Education and experience pop in my head often. Not every day, but many days I think back to my lucky 7 years in Indiana learning under the supervision of master clinicians – at a time when trainees were allowed the leeway to make decisions ... and mistakes. Then, when I joined private practice, I continued to learn from experienced practitioners.
It would be foolish to argue that training and experience aren’t important.
But here’s the thing:
I will make three points: First, I will bolster two of my old arguments as to why we shouldn’t be worried about non-MD clinicians, then I will propose some ideas to increase confidence in NP and PA care.
Health care does not equal health
On the matter of how much clinicians affect outcomes, a recently published randomized controlled trial performed in India found that subsidizing insurance care led to increased utilization of hospital services but had no significant effect on health outcomes. This follows the RAND and Oregon Health Insurance studies in the United States, which largely reported similar results.
We should also not dismiss the fact that – despite the massive technology gains over the past half-century in digital health and artificial intelligence and increased use of quality measures, new drugs and procedures, and mega-medical centers – the average lifespan of Americans is flat to declining (in most ethnic and racial groups). Worse than no gains in longevity, perhaps, is that death from diseases like dementia and Parkinson’s disease are on the rise.
A neutral Martian would look down and wonder why all this health care hasn’t translated to longer and better lives. The causes of this paradox remain speculative, and are for another column, but the point remains that – on average – more health care is clearly not delivering more health. And if that is true, one may deduce that much of U.S. health care is marginal when it comes to affecting major outcomes.
It’s about the delta
Logos trumps pathos. Sure, my physician colleagues can tell scary anecdotes of bad outcomes caused by an inexperienced NP or PA. I would counter that by saying I have sat on our hospital’s peer review committee for 2 decades, including the era before NPs or PAs were practicing, and I have plenty of stories of physician errors. These include, of course, my own errors.
Logos: We must consider the difference between non–MD-led care and MD-led care.
My arguments from 2020 remain relevant today. Most medical problems are not engineering puzzles. Many, perhaps most, patients fall into an easy protocol – say, chest pain, dyspnea, or atrial fibrillation. With basic training, a motivated serious person quickly gains skill in recognizing and treating everyday problems.
And just 2 years on, technology further levels the playing field. Consider radiology in 2022 – it’s easy to take for granted the speed of the CT scan, the fidelity of the MRI, and the easy access to both in the U.S. hospital system. Less experienced clinicians have never had more tools to assist with diagnostics and therapeutics.
The expansion of team-based care has also mitigated the effects of inexperience. It took Americans longer than Canadians to figure out how helpful pharmacists could be. Pharmacists in my hospital now help us dose complicated medicines and protect us against prescribing errors.
Then there is the immediate access to online information. Gone are the days when you had to memorize long-QT syndromes. Book knowledge – that I spent years acquiring – now comes in seconds. The other day an NP corrected me. I asked, Are you sure? Boom, she took out her phone and showed me the evidence.
In sum, if it were even possible to measure the clinical competence of care from NP and PA versus physicians, there would be two bell-shaped curves with a tremendous amount of overlap. And that overlap would steadily increase as a given NP or PA gathered experience. (The NP in our electrophysiology division has more than 25 years’ experience in heart rhythm care, and it is common for colleagues to call her before one of us docs. Rightly so.)
Three basic proposals regarding NP and PA care
To ensure quality of care, I have three proposals.
It has always seemed strange to me that an NP or PA can flip from one field to another without a period of training. I can’t just change practice from electrophysiology to dermatology without doing a residency. But NPs and PAs can.
My first proposal would be that NPs and PAs spend a substantial period of training in a field before practice – a legit apprenticeship. The duration of this period is a matter of debate, but it ought to be standardized.
My second proposal is that, if physicians are required to pass certification exams, so should NPs. (PAs have an exam every 10 years.) The exam should be the same as (or very similar to) the physician exam, and it should be specific to their field of practice.
While I have argued (and still feel) that the American Board of Internal Medicine brand of certification is dubious, the fact remains that physicians must maintain proficiency in their field. Requiring NPs and PAs to do the same would help foster specialization. And while I can’t cite empirical evidence, specialization seems super-important. We have NPs at my hospital who have been in the same area for years, and they exude clinical competence.
Finally, I have come to believe that the best way for nearly any clinician to practice medicine is as part of a team. (The exception being primary care in rural areas where there are clinician shortages.)
On the matter of team care, I’ve practiced for a long time, but nearly every day I run situations by a colleague; often this person is an NP. The economist Friedrich Hayek proposed that dispersed knowledge always outpaces the wisdom of any individual. That notion pertains well to the increasing complexities and specialization of modern medical practice.
A person who commits to learning one area of medicine, enjoys helping people, asks often for help, and has the support of colleagues is set up to be a successful clinician – whether the letters after their name are APRN, PA, DO, or MD.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky. He did not report any relevant financial disclosures. A version of this article first appeared on Medscape.com.
Two years ago, I argued that independent care from nurse practitioners (NPs) and physician assistants (PAs) would not have ill effects on health outcomes. To the surprise of no one, NPs and PAs embraced the argument; physicians clobbered it.
My case had three pegs: One was that medicine isn’t rocket science and clinicians control a lot less than we think we do. The second peg was that technology levels the playing field of clinical care. High-sensitivity troponin assays, for instance, make missing MI a lot less likely. The third peg was empirical: Studies have found little difference in MD versus non–MD-led care. Looking back, I now see empiricism as the weakest part of the argument because the studies had so many limitations.
I update this viewpoint now because health care is increasingly delivered by NPs and PAs. And there are two concerning trends regarding NP education and experience. First is that nurses are turning to advanced practitioner training earlier in their careers – without gathering much bedside experience. And these training programs are increasingly likely to be online, with minimal hands-on clinical tutoring.
Education and experience pop in my head often. Not every day, but many days I think back to my lucky 7 years in Indiana learning under the supervision of master clinicians – at a time when trainees were allowed the leeway to make decisions ... and mistakes. Then, when I joined private practice, I continued to learn from experienced practitioners.
It would be foolish to argue that training and experience aren’t important.
But here’s the thing:
I will make three points: First, I will bolster two of my old arguments as to why we shouldn’t be worried about non-MD clinicians, then I will propose some ideas to increase confidence in NP and PA care.
Health care does not equal health
On the matter of how much clinicians affect outcomes, a recently published randomized controlled trial performed in India found that subsidizing insurance care led to increased utilization of hospital services but had no significant effect on health outcomes. This follows the RAND and Oregon Health Insurance studies in the United States, which largely reported similar results.
We should also not dismiss the fact that – despite the massive technology gains over the past half-century in digital health and artificial intelligence and increased use of quality measures, new drugs and procedures, and mega-medical centers – the average lifespan of Americans is flat to declining (in most ethnic and racial groups). Worse than no gains in longevity, perhaps, is that death from diseases like dementia and Parkinson’s disease are on the rise.
A neutral Martian would look down and wonder why all this health care hasn’t translated to longer and better lives. The causes of this paradox remain speculative, and are for another column, but the point remains that – on average – more health care is clearly not delivering more health. And if that is true, one may deduce that much of U.S. health care is marginal when it comes to affecting major outcomes.
It’s about the delta
Logos trumps pathos. Sure, my physician colleagues can tell scary anecdotes of bad outcomes caused by an inexperienced NP or PA. I would counter that by saying I have sat on our hospital’s peer review committee for 2 decades, including the era before NPs or PAs were practicing, and I have plenty of stories of physician errors. These include, of course, my own errors.
Logos: We must consider the difference between non–MD-led care and MD-led care.
My arguments from 2020 remain relevant today. Most medical problems are not engineering puzzles. Many, perhaps most, patients fall into an easy protocol – say, chest pain, dyspnea, or atrial fibrillation. With basic training, a motivated serious person quickly gains skill in recognizing and treating everyday problems.
And just 2 years on, technology further levels the playing field. Consider radiology in 2022 – it’s easy to take for granted the speed of the CT scan, the fidelity of the MRI, and the easy access to both in the U.S. hospital system. Less experienced clinicians have never had more tools to assist with diagnostics and therapeutics.
The expansion of team-based care has also mitigated the effects of inexperience. It took Americans longer than Canadians to figure out how helpful pharmacists could be. Pharmacists in my hospital now help us dose complicated medicines and protect us against prescribing errors.
Then there is the immediate access to online information. Gone are the days when you had to memorize long-QT syndromes. Book knowledge – that I spent years acquiring – now comes in seconds. The other day an NP corrected me. I asked, Are you sure? Boom, she took out her phone and showed me the evidence.
In sum, if it were even possible to measure the clinical competence of care from NP and PA versus physicians, there would be two bell-shaped curves with a tremendous amount of overlap. And that overlap would steadily increase as a given NP or PA gathered experience. (The NP in our electrophysiology division has more than 25 years’ experience in heart rhythm care, and it is common for colleagues to call her before one of us docs. Rightly so.)
Three basic proposals regarding NP and PA care
To ensure quality of care, I have three proposals.
It has always seemed strange to me that an NP or PA can flip from one field to another without a period of training. I can’t just change practice from electrophysiology to dermatology without doing a residency. But NPs and PAs can.
My first proposal would be that NPs and PAs spend a substantial period of training in a field before practice – a legit apprenticeship. The duration of this period is a matter of debate, but it ought to be standardized.
My second proposal is that, if physicians are required to pass certification exams, so should NPs. (PAs have an exam every 10 years.) The exam should be the same as (or very similar to) the physician exam, and it should be specific to their field of practice.
While I have argued (and still feel) that the American Board of Internal Medicine brand of certification is dubious, the fact remains that physicians must maintain proficiency in their field. Requiring NPs and PAs to do the same would help foster specialization. And while I can’t cite empirical evidence, specialization seems super-important. We have NPs at my hospital who have been in the same area for years, and they exude clinical competence.
Finally, I have come to believe that the best way for nearly any clinician to practice medicine is as part of a team. (The exception being primary care in rural areas where there are clinician shortages.)
On the matter of team care, I’ve practiced for a long time, but nearly every day I run situations by a colleague; often this person is an NP. The economist Friedrich Hayek proposed that dispersed knowledge always outpaces the wisdom of any individual. That notion pertains well to the increasing complexities and specialization of modern medical practice.
A person who commits to learning one area of medicine, enjoys helping people, asks often for help, and has the support of colleagues is set up to be a successful clinician – whether the letters after their name are APRN, PA, DO, or MD.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky. He did not report any relevant financial disclosures. A version of this article first appeared on Medscape.com.
Seven ways doctors could get better payment from insurers
, say experts in physician-payer contracts.
Many doctors sign long-term agreements and then forget about them, says Marcia Brauchler, president and founder of Physicians’ Ally, Littleton, Colorado, a health care consulting company. “The average doctor is trying to run a practice on 2010 rates because they haven’t touched their insurance contracts for 10 years,” she says.
Payers also make a lot of money by adopting dozens of unilateral policy and procedure changes every year that they know physicians are too busy to read. They are counting on the fact that few doctors will understand what the policy changes are and that even fewer will contest them, says Greg Brodek, JD, chair of the health law practice group and head of the managed care litigation practice at Duane Morris, who represents doctors in disputes with payers.
These experts say doctors can push back on one-sided payer contracts and negotiate changes. Mr. Brodek says some practices have more leverage than others to influence payers – if they are larger, in a specialty that the payer needs in its network, or located in a remote area where the payer has limited options.
Here are seven key areas to pay attention to:
1. Long-term contracts. Most doctors sign multiyear “evergreen” contracts that renew automatically every year. This allows insurers to continue to pay doctors the same rate for years.
To avoid this, doctors should negotiate new rates when their agreements renew or, if they prefer, ask that a cost-of-living adjustment be included in the multiyear contract that applies to subsequent years, says Ms. Brauchler.
2. Fee schedules. Payers will “whitewash” what they’re paying you by saying it’s 100% of the payer fee schedule. When it comes to Medicare, they may be paying you a lot less, says Ms. Brauchler.
“My biggest takeaway is to compare the CPT codes of the payer’s fee schedule against what Medicare allows. For example, for CPT code 99213, a 15-minute established office visit, if Medicare pays you $100 and Aetna pays you $75.00, you’re getting 75% of Medicare,” says Ms. Brauchler. To avoid this, doctors should ask that the contract state that reimbursement be made according to Medicare’s medical policies rather than the payer’s.
3. Audits. Commercial payers will claim they have a contractual right to conduct pre- and post-payment audits of physicians’ claims that can result in reduced payments. The contract only states that if doctors correctly submit claims, they will get paid, not that they will have to go through extra steps, which is a breach of their agreement, says Mr. Brodek.
In his experience, 90% of payers back down when asked to provide the contractual basis to conduct these audits. “Or, they take the position that it’s not in the contract but that they have a policy.”
4. Contract amendments versus policies and procedures. This is a huge area that needs to be clarified in contracts and monitored by providers throughout their relationships with payers. Contracts have three elements: the parties, the services provided, and the payment. Changing any one of those terms requires an amendment and advance written notice that has to be delivered to the other party in a certain way, such as by overnight delivery, says Mr. Brodek.
In addition, both parties have to sign that they agree to an amendment. “But, that’s too cumbersome and complicated for payers who have decided to adopt policies instead. These are unilateral changes made with no advance notice given, since the payer typically posts the change on its website,” says Mr. Brodek.
5. Recoupment efforts. Payers will review claims after they’re paid and contact the doctor saying they found a mistake, such as inappropriate coding. They will claim that the doctor now owes them a large sum of money based on a percentage of claims reviewed. “They typically send the doctor a letter that ends with, ‘If you do not pay this amount within 30 days, we will offset the amount due against future payments that we would otherwise make to you,’” says Mr. Brodek.
He recommends that contracts include the doctors’ right to contest an audit so the “payer doesn’t have the unilateral right to disregard the initial coding that the doctor appropriately assigned to the claim and recoup the money anyway,” says Mr. Brodek.
6. Medical network rentals and products. Most contracts say that payers can rent out their medical networks to other health plans, such as HMOs, and that the clinicians agree to comply with all of their policies and procedures. The agreement may also cover the products of other plans.
“The problem is that physicians are not given information about the other plans, including their terms and conditions for getting paid,” says Mr. Brodek. If a problem with payment arises, they have no written agreement with that plan, which makes it harder to enforce.
“That’s why we recommend that doctors negotiate agreements that only cover the main payer. Most of the time, the payer is amenable to putting that language in the contract,” he says.
7. Payer products. In the past several years, a typical contract has included appendices that list the payer’s products, such as Medicare, workers compensation, auto insurance liability, or health care exchange products. Many clinicians don’t realize they can pick the plans they want to participate in by accepting or opting out, says Mr. Brodek.
“We advise clients to limit the contract to what you want covered and to make informed decisions, because some products have low fees set by the states, such as workers compensation and health care exchanges,” says Mr. Brodek.
A version of this article first appeared on Medscape.com.
, say experts in physician-payer contracts.
Many doctors sign long-term agreements and then forget about them, says Marcia Brauchler, president and founder of Physicians’ Ally, Littleton, Colorado, a health care consulting company. “The average doctor is trying to run a practice on 2010 rates because they haven’t touched their insurance contracts for 10 years,” she says.
Payers also make a lot of money by adopting dozens of unilateral policy and procedure changes every year that they know physicians are too busy to read. They are counting on the fact that few doctors will understand what the policy changes are and that even fewer will contest them, says Greg Brodek, JD, chair of the health law practice group and head of the managed care litigation practice at Duane Morris, who represents doctors in disputes with payers.
These experts say doctors can push back on one-sided payer contracts and negotiate changes. Mr. Brodek says some practices have more leverage than others to influence payers – if they are larger, in a specialty that the payer needs in its network, or located in a remote area where the payer has limited options.
Here are seven key areas to pay attention to:
1. Long-term contracts. Most doctors sign multiyear “evergreen” contracts that renew automatically every year. This allows insurers to continue to pay doctors the same rate for years.
To avoid this, doctors should negotiate new rates when their agreements renew or, if they prefer, ask that a cost-of-living adjustment be included in the multiyear contract that applies to subsequent years, says Ms. Brauchler.
2. Fee schedules. Payers will “whitewash” what they’re paying you by saying it’s 100% of the payer fee schedule. When it comes to Medicare, they may be paying you a lot less, says Ms. Brauchler.
“My biggest takeaway is to compare the CPT codes of the payer’s fee schedule against what Medicare allows. For example, for CPT code 99213, a 15-minute established office visit, if Medicare pays you $100 and Aetna pays you $75.00, you’re getting 75% of Medicare,” says Ms. Brauchler. To avoid this, doctors should ask that the contract state that reimbursement be made according to Medicare’s medical policies rather than the payer’s.
3. Audits. Commercial payers will claim they have a contractual right to conduct pre- and post-payment audits of physicians’ claims that can result in reduced payments. The contract only states that if doctors correctly submit claims, they will get paid, not that they will have to go through extra steps, which is a breach of their agreement, says Mr. Brodek.
In his experience, 90% of payers back down when asked to provide the contractual basis to conduct these audits. “Or, they take the position that it’s not in the contract but that they have a policy.”
4. Contract amendments versus policies and procedures. This is a huge area that needs to be clarified in contracts and monitored by providers throughout their relationships with payers. Contracts have three elements: the parties, the services provided, and the payment. Changing any one of those terms requires an amendment and advance written notice that has to be delivered to the other party in a certain way, such as by overnight delivery, says Mr. Brodek.
In addition, both parties have to sign that they agree to an amendment. “But, that’s too cumbersome and complicated for payers who have decided to adopt policies instead. These are unilateral changes made with no advance notice given, since the payer typically posts the change on its website,” says Mr. Brodek.
5. Recoupment efforts. Payers will review claims after they’re paid and contact the doctor saying they found a mistake, such as inappropriate coding. They will claim that the doctor now owes them a large sum of money based on a percentage of claims reviewed. “They typically send the doctor a letter that ends with, ‘If you do not pay this amount within 30 days, we will offset the amount due against future payments that we would otherwise make to you,’” says Mr. Brodek.
He recommends that contracts include the doctors’ right to contest an audit so the “payer doesn’t have the unilateral right to disregard the initial coding that the doctor appropriately assigned to the claim and recoup the money anyway,” says Mr. Brodek.
6. Medical network rentals and products. Most contracts say that payers can rent out their medical networks to other health plans, such as HMOs, and that the clinicians agree to comply with all of their policies and procedures. The agreement may also cover the products of other plans.
“The problem is that physicians are not given information about the other plans, including their terms and conditions for getting paid,” says Mr. Brodek. If a problem with payment arises, they have no written agreement with that plan, which makes it harder to enforce.
“That’s why we recommend that doctors negotiate agreements that only cover the main payer. Most of the time, the payer is amenable to putting that language in the contract,” he says.
7. Payer products. In the past several years, a typical contract has included appendices that list the payer’s products, such as Medicare, workers compensation, auto insurance liability, or health care exchange products. Many clinicians don’t realize they can pick the plans they want to participate in by accepting or opting out, says Mr. Brodek.
“We advise clients to limit the contract to what you want covered and to make informed decisions, because some products have low fees set by the states, such as workers compensation and health care exchanges,” says Mr. Brodek.
A version of this article first appeared on Medscape.com.
, say experts in physician-payer contracts.
Many doctors sign long-term agreements and then forget about them, says Marcia Brauchler, president and founder of Physicians’ Ally, Littleton, Colorado, a health care consulting company. “The average doctor is trying to run a practice on 2010 rates because they haven’t touched their insurance contracts for 10 years,” she says.
Payers also make a lot of money by adopting dozens of unilateral policy and procedure changes every year that they know physicians are too busy to read. They are counting on the fact that few doctors will understand what the policy changes are and that even fewer will contest them, says Greg Brodek, JD, chair of the health law practice group and head of the managed care litigation practice at Duane Morris, who represents doctors in disputes with payers.
These experts say doctors can push back on one-sided payer contracts and negotiate changes. Mr. Brodek says some practices have more leverage than others to influence payers – if they are larger, in a specialty that the payer needs in its network, or located in a remote area where the payer has limited options.
Here are seven key areas to pay attention to:
1. Long-term contracts. Most doctors sign multiyear “evergreen” contracts that renew automatically every year. This allows insurers to continue to pay doctors the same rate for years.
To avoid this, doctors should negotiate new rates when their agreements renew or, if they prefer, ask that a cost-of-living adjustment be included in the multiyear contract that applies to subsequent years, says Ms. Brauchler.
2. Fee schedules. Payers will “whitewash” what they’re paying you by saying it’s 100% of the payer fee schedule. When it comes to Medicare, they may be paying you a lot less, says Ms. Brauchler.
“My biggest takeaway is to compare the CPT codes of the payer’s fee schedule against what Medicare allows. For example, for CPT code 99213, a 15-minute established office visit, if Medicare pays you $100 and Aetna pays you $75.00, you’re getting 75% of Medicare,” says Ms. Brauchler. To avoid this, doctors should ask that the contract state that reimbursement be made according to Medicare’s medical policies rather than the payer’s.
3. Audits. Commercial payers will claim they have a contractual right to conduct pre- and post-payment audits of physicians’ claims that can result in reduced payments. The contract only states that if doctors correctly submit claims, they will get paid, not that they will have to go through extra steps, which is a breach of their agreement, says Mr. Brodek.
In his experience, 90% of payers back down when asked to provide the contractual basis to conduct these audits. “Or, they take the position that it’s not in the contract but that they have a policy.”
4. Contract amendments versus policies and procedures. This is a huge area that needs to be clarified in contracts and monitored by providers throughout their relationships with payers. Contracts have three elements: the parties, the services provided, and the payment. Changing any one of those terms requires an amendment and advance written notice that has to be delivered to the other party in a certain way, such as by overnight delivery, says Mr. Brodek.
In addition, both parties have to sign that they agree to an amendment. “But, that’s too cumbersome and complicated for payers who have decided to adopt policies instead. These are unilateral changes made with no advance notice given, since the payer typically posts the change on its website,” says Mr. Brodek.
5. Recoupment efforts. Payers will review claims after they’re paid and contact the doctor saying they found a mistake, such as inappropriate coding. They will claim that the doctor now owes them a large sum of money based on a percentage of claims reviewed. “They typically send the doctor a letter that ends with, ‘If you do not pay this amount within 30 days, we will offset the amount due against future payments that we would otherwise make to you,’” says Mr. Brodek.
He recommends that contracts include the doctors’ right to contest an audit so the “payer doesn’t have the unilateral right to disregard the initial coding that the doctor appropriately assigned to the claim and recoup the money anyway,” says Mr. Brodek.
6. Medical network rentals and products. Most contracts say that payers can rent out their medical networks to other health plans, such as HMOs, and that the clinicians agree to comply with all of their policies and procedures. The agreement may also cover the products of other plans.
“The problem is that physicians are not given information about the other plans, including their terms and conditions for getting paid,” says Mr. Brodek. If a problem with payment arises, they have no written agreement with that plan, which makes it harder to enforce.
“That’s why we recommend that doctors negotiate agreements that only cover the main payer. Most of the time, the payer is amenable to putting that language in the contract,” he says.
7. Payer products. In the past several years, a typical contract has included appendices that list the payer’s products, such as Medicare, workers compensation, auto insurance liability, or health care exchange products. Many clinicians don’t realize they can pick the plans they want to participate in by accepting or opting out, says Mr. Brodek.
“We advise clients to limit the contract to what you want covered and to make informed decisions, because some products have low fees set by the states, such as workers compensation and health care exchanges,” says Mr. Brodek.
A version of this article first appeared on Medscape.com.