User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Ways to make sure 2022 doesn’t stink for docs
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Gut bacteria linked with long COVID
While links have been found between the gut’s microbiome and COVID-19, as well as other diseases, this is the first published research to show a link specifically to COVID’s long-term effects, the investigators, based at the Chinese University of Hong Kong, wrote in Gut.
“To our knowledge, this is the first study to show that altered gut microbiome composition is strongly associated with persistent symptoms in patients with COVID-19 up to 6 months after clearance of SARS-CoV-2 virus,” said Siew Ng, MBBS, PhD, associate director at the university’s Center for Gut Microbiota Research.
At three hospitals, the researchers enrolled 106 patients with COVID-19 from February to August 2020 with stool samples at admission and at 1 month and 6 months after discharge, and compared them with people who did not have COVID, recruited in 2019. The severity of COVID in the enrolled patients was mostly mild to moderate.
At 3 months, 86 of the patients with COVID had post–acute COVID-19 syndrome (PACS) – defined as at least one persistent, otherwise unexplained symptom 4 weeks after clearance of the virus. And 81 patients had PACS at 6 months, most commonly fatigue, poor memory, hair loss, anxiety, and trouble sleeping.
Using stool samples for their analysis, the researchers found that, broadly, the diversity of the types of bacteria, and the abundance of these bacteria, were significantly lower at 6 months for those with PACS, compared with those without PACS and with controls (P < .05 and P < .0001, respectively). Among those with PACS, 28 bacteria species were diminished and 14 were enriched, both at baseline and follow-up. Those patients who had COVID but not PACS showed just 25 alterations of bacteria species at the time of hospital admission, and they all normalized by 6 months.
Having respiratory symptoms at 6 months was linked with higher levels of opportunistic pathogens such as Streptococcus anginosus and S. vestibularis. Neuropsychiatric symptoms and fatigue were associated with nosocomial pathogens that are linked to opportunistic infections, such as Clostridium innocuum and Actinomyces naeslundii (P < .05).
Bacteria known for producing butyrate, a beneficial fatty acid, were significantly depleted in those patients with hair loss. And certain of these bacteria, including Bifidobacterium pseudocatenulatum and Faecalibacterium prausnitzii, had the largest inverse correlations with PACS at 6 months (P < .05), the researchers found.
“Particular gut microbial profiles may indicate heightened susceptibility,” Dr. Ng said.
Although the findings were drawn from patients with earlier strains of the COVID-19 virus, the findings still apply to new variants, including Omicron, since these pose the same problem of persistent disruption of the immune system, Dr. Ng said.
Her group is conducting trials to look at how modulating the microbiome might prevent long COVID and boost antibodies after vaccination in high-risk people, she said.
“Gut microbiota influences the health of the host,” Dr. Ng said. “It provides crucial benefits in the form of immune system development, prevention of infections, nutrient acquisition, and brain and nervous system functionality. Considering the millions of people infected during the ongoing pandemic, our findings are a strong impetus for consideration of microbiota modulation to facilitate timely recovery and reduce the burden of post–acute COVID-19 syndrome.”
John Haran, MD, PhD, associate professor of microbiology and physiological systems and emergency medicine at the University of Massachusetts, Worcester, said the research adds to the evidence base on the gut microbiome’s links to COVID, but there was likely be no clinical impact yet. Still, he said the findings linking specific species to specific symptoms was particularly interesting.
“Very early on during hospitalization, [the researchers] saw these differences and correlated out with people who have longer symptoms, and especially different groups of people that have longer symptoms, too,” said Dr. Haran, who has done research on the topic. “It’s very different if you have different symptoms, for example, you keep coughing for months versus you have brain fog and fatigue, or other debilitating symptoms.”
Dr. Haran noted that the findings didn’t identify bacteria types especially linked to COVID, but rather species that have already been found to be associated with a “bad” microbiome. He also pointed out that the patients enrolled in the study were not vaccinated, because vaccines weren’t available at the time. Still, further study to see whether modulation of gut bacteria can be a therapy seems worthwhile.
“Microbiome modulation is pretty safe, and that’s really the next big step that needs to be taken in this,” he said.
For now, the findings don’t give the clinician much new ammunition for treatment.
“We’re not there yet,” he added. “It’s not as if clinicians are going to tell their COVID patients: ‘Go out and buy some kale.’ ”
Eugene Chang, MD, professor of medicine at the University of Chicago, who has studied the gut microbiome and gastrointestinal disease, said it’s “too preliminary” to say whether the findings could lead to a clinical impact. The measures used merely identify the microbes present, but not what they are doing.
“These measures are unlikely to perform well enough to be useful for risk assessment or predicting clinical outcomes,” he said. “That being said, advances in technology are being made where next generations of metrics could be developed and useful as stratifiers and predictors of risk.”
Seeing shifting patterns associated with certain symptoms, he said, is “notable because it suggests that the disturbances of the gut microbiota in PACS are significant.”
But he said it’s important to know whether these changes are a cause of PACS in some way or just an effect of it.
“If causative or contributory – this has to be proven – then ‘microbiota modulation’ would make sense and could be a priority for development,” he said. “If merely an effect, these metrics and better ones to come could be useful as predictors or measures of the patient’s general state of health.”
As seen in his group’s work and other work, he said, “the gut microbiota is highly sensitive to changes in their ecosystem, which is influenced by the health state of the patient.”
Dr. Ng, Dr. Haran, and Dr. Chang reported no relevant disclosures.
This article was updated Jan. 27, 2022.
While links have been found between the gut’s microbiome and COVID-19, as well as other diseases, this is the first published research to show a link specifically to COVID’s long-term effects, the investigators, based at the Chinese University of Hong Kong, wrote in Gut.
“To our knowledge, this is the first study to show that altered gut microbiome composition is strongly associated with persistent symptoms in patients with COVID-19 up to 6 months after clearance of SARS-CoV-2 virus,” said Siew Ng, MBBS, PhD, associate director at the university’s Center for Gut Microbiota Research.
At three hospitals, the researchers enrolled 106 patients with COVID-19 from February to August 2020 with stool samples at admission and at 1 month and 6 months after discharge, and compared them with people who did not have COVID, recruited in 2019. The severity of COVID in the enrolled patients was mostly mild to moderate.
At 3 months, 86 of the patients with COVID had post–acute COVID-19 syndrome (PACS) – defined as at least one persistent, otherwise unexplained symptom 4 weeks after clearance of the virus. And 81 patients had PACS at 6 months, most commonly fatigue, poor memory, hair loss, anxiety, and trouble sleeping.
Using stool samples for their analysis, the researchers found that, broadly, the diversity of the types of bacteria, and the abundance of these bacteria, were significantly lower at 6 months for those with PACS, compared with those without PACS and with controls (P < .05 and P < .0001, respectively). Among those with PACS, 28 bacteria species were diminished and 14 were enriched, both at baseline and follow-up. Those patients who had COVID but not PACS showed just 25 alterations of bacteria species at the time of hospital admission, and they all normalized by 6 months.
Having respiratory symptoms at 6 months was linked with higher levels of opportunistic pathogens such as Streptococcus anginosus and S. vestibularis. Neuropsychiatric symptoms and fatigue were associated with nosocomial pathogens that are linked to opportunistic infections, such as Clostridium innocuum and Actinomyces naeslundii (P < .05).
Bacteria known for producing butyrate, a beneficial fatty acid, were significantly depleted in those patients with hair loss. And certain of these bacteria, including Bifidobacterium pseudocatenulatum and Faecalibacterium prausnitzii, had the largest inverse correlations with PACS at 6 months (P < .05), the researchers found.
“Particular gut microbial profiles may indicate heightened susceptibility,” Dr. Ng said.
Although the findings were drawn from patients with earlier strains of the COVID-19 virus, the findings still apply to new variants, including Omicron, since these pose the same problem of persistent disruption of the immune system, Dr. Ng said.
Her group is conducting trials to look at how modulating the microbiome might prevent long COVID and boost antibodies after vaccination in high-risk people, she said.
“Gut microbiota influences the health of the host,” Dr. Ng said. “It provides crucial benefits in the form of immune system development, prevention of infections, nutrient acquisition, and brain and nervous system functionality. Considering the millions of people infected during the ongoing pandemic, our findings are a strong impetus for consideration of microbiota modulation to facilitate timely recovery and reduce the burden of post–acute COVID-19 syndrome.”
John Haran, MD, PhD, associate professor of microbiology and physiological systems and emergency medicine at the University of Massachusetts, Worcester, said the research adds to the evidence base on the gut microbiome’s links to COVID, but there was likely be no clinical impact yet. Still, he said the findings linking specific species to specific symptoms was particularly interesting.
“Very early on during hospitalization, [the researchers] saw these differences and correlated out with people who have longer symptoms, and especially different groups of people that have longer symptoms, too,” said Dr. Haran, who has done research on the topic. “It’s very different if you have different symptoms, for example, you keep coughing for months versus you have brain fog and fatigue, or other debilitating symptoms.”
Dr. Haran noted that the findings didn’t identify bacteria types especially linked to COVID, but rather species that have already been found to be associated with a “bad” microbiome. He also pointed out that the patients enrolled in the study were not vaccinated, because vaccines weren’t available at the time. Still, further study to see whether modulation of gut bacteria can be a therapy seems worthwhile.
“Microbiome modulation is pretty safe, and that’s really the next big step that needs to be taken in this,” he said.
For now, the findings don’t give the clinician much new ammunition for treatment.
“We’re not there yet,” he added. “It’s not as if clinicians are going to tell their COVID patients: ‘Go out and buy some kale.’ ”
Eugene Chang, MD, professor of medicine at the University of Chicago, who has studied the gut microbiome and gastrointestinal disease, said it’s “too preliminary” to say whether the findings could lead to a clinical impact. The measures used merely identify the microbes present, but not what they are doing.
“These measures are unlikely to perform well enough to be useful for risk assessment or predicting clinical outcomes,” he said. “That being said, advances in technology are being made where next generations of metrics could be developed and useful as stratifiers and predictors of risk.”
Seeing shifting patterns associated with certain symptoms, he said, is “notable because it suggests that the disturbances of the gut microbiota in PACS are significant.”
But he said it’s important to know whether these changes are a cause of PACS in some way or just an effect of it.
“If causative or contributory – this has to be proven – then ‘microbiota modulation’ would make sense and could be a priority for development,” he said. “If merely an effect, these metrics and better ones to come could be useful as predictors or measures of the patient’s general state of health.”
As seen in his group’s work and other work, he said, “the gut microbiota is highly sensitive to changes in their ecosystem, which is influenced by the health state of the patient.”
Dr. Ng, Dr. Haran, and Dr. Chang reported no relevant disclosures.
This article was updated Jan. 27, 2022.
While links have been found between the gut’s microbiome and COVID-19, as well as other diseases, this is the first published research to show a link specifically to COVID’s long-term effects, the investigators, based at the Chinese University of Hong Kong, wrote in Gut.
“To our knowledge, this is the first study to show that altered gut microbiome composition is strongly associated with persistent symptoms in patients with COVID-19 up to 6 months after clearance of SARS-CoV-2 virus,” said Siew Ng, MBBS, PhD, associate director at the university’s Center for Gut Microbiota Research.
At three hospitals, the researchers enrolled 106 patients with COVID-19 from February to August 2020 with stool samples at admission and at 1 month and 6 months after discharge, and compared them with people who did not have COVID, recruited in 2019. The severity of COVID in the enrolled patients was mostly mild to moderate.
At 3 months, 86 of the patients with COVID had post–acute COVID-19 syndrome (PACS) – defined as at least one persistent, otherwise unexplained symptom 4 weeks after clearance of the virus. And 81 patients had PACS at 6 months, most commonly fatigue, poor memory, hair loss, anxiety, and trouble sleeping.
Using stool samples for their analysis, the researchers found that, broadly, the diversity of the types of bacteria, and the abundance of these bacteria, were significantly lower at 6 months for those with PACS, compared with those without PACS and with controls (P < .05 and P < .0001, respectively). Among those with PACS, 28 bacteria species were diminished and 14 were enriched, both at baseline and follow-up. Those patients who had COVID but not PACS showed just 25 alterations of bacteria species at the time of hospital admission, and they all normalized by 6 months.
Having respiratory symptoms at 6 months was linked with higher levels of opportunistic pathogens such as Streptococcus anginosus and S. vestibularis. Neuropsychiatric symptoms and fatigue were associated with nosocomial pathogens that are linked to opportunistic infections, such as Clostridium innocuum and Actinomyces naeslundii (P < .05).
Bacteria known for producing butyrate, a beneficial fatty acid, were significantly depleted in those patients with hair loss. And certain of these bacteria, including Bifidobacterium pseudocatenulatum and Faecalibacterium prausnitzii, had the largest inverse correlations with PACS at 6 months (P < .05), the researchers found.
“Particular gut microbial profiles may indicate heightened susceptibility,” Dr. Ng said.
Although the findings were drawn from patients with earlier strains of the COVID-19 virus, the findings still apply to new variants, including Omicron, since these pose the same problem of persistent disruption of the immune system, Dr. Ng said.
Her group is conducting trials to look at how modulating the microbiome might prevent long COVID and boost antibodies after vaccination in high-risk people, she said.
“Gut microbiota influences the health of the host,” Dr. Ng said. “It provides crucial benefits in the form of immune system development, prevention of infections, nutrient acquisition, and brain and nervous system functionality. Considering the millions of people infected during the ongoing pandemic, our findings are a strong impetus for consideration of microbiota modulation to facilitate timely recovery and reduce the burden of post–acute COVID-19 syndrome.”
John Haran, MD, PhD, associate professor of microbiology and physiological systems and emergency medicine at the University of Massachusetts, Worcester, said the research adds to the evidence base on the gut microbiome’s links to COVID, but there was likely be no clinical impact yet. Still, he said the findings linking specific species to specific symptoms was particularly interesting.
“Very early on during hospitalization, [the researchers] saw these differences and correlated out with people who have longer symptoms, and especially different groups of people that have longer symptoms, too,” said Dr. Haran, who has done research on the topic. “It’s very different if you have different symptoms, for example, you keep coughing for months versus you have brain fog and fatigue, or other debilitating symptoms.”
Dr. Haran noted that the findings didn’t identify bacteria types especially linked to COVID, but rather species that have already been found to be associated with a “bad” microbiome. He also pointed out that the patients enrolled in the study were not vaccinated, because vaccines weren’t available at the time. Still, further study to see whether modulation of gut bacteria can be a therapy seems worthwhile.
“Microbiome modulation is pretty safe, and that’s really the next big step that needs to be taken in this,” he said.
For now, the findings don’t give the clinician much new ammunition for treatment.
“We’re not there yet,” he added. “It’s not as if clinicians are going to tell their COVID patients: ‘Go out and buy some kale.’ ”
Eugene Chang, MD, professor of medicine at the University of Chicago, who has studied the gut microbiome and gastrointestinal disease, said it’s “too preliminary” to say whether the findings could lead to a clinical impact. The measures used merely identify the microbes present, but not what they are doing.
“These measures are unlikely to perform well enough to be useful for risk assessment or predicting clinical outcomes,” he said. “That being said, advances in technology are being made where next generations of metrics could be developed and useful as stratifiers and predictors of risk.”
Seeing shifting patterns associated with certain symptoms, he said, is “notable because it suggests that the disturbances of the gut microbiota in PACS are significant.”
But he said it’s important to know whether these changes are a cause of PACS in some way or just an effect of it.
“If causative or contributory – this has to be proven – then ‘microbiota modulation’ would make sense and could be a priority for development,” he said. “If merely an effect, these metrics and better ones to come could be useful as predictors or measures of the patient’s general state of health.”
As seen in his group’s work and other work, he said, “the gut microbiota is highly sensitive to changes in their ecosystem, which is influenced by the health state of the patient.”
Dr. Ng, Dr. Haran, and Dr. Chang reported no relevant disclosures.
This article was updated Jan. 27, 2022.
FROM GUT
‘Post-truth era’ hurts COVID-19 response, trust in science
Can you tell which of the following statements are true and which are false?
COVID-19 is not a threat to younger people, and only those who have other medical conditions are dying from it.
The mRNA vaccines developed to prevent the coronavirus alter your genes, can make your body “magnetic,” and are killing more people than the virus itself.
President Joe Biden’s climate change plan calls for a ban on meat consumption to cut greenhouse gas emissions.
The 2020 presidential election was rigged and stolen.
If you guessed that all of these claims are false, you’re right – take a bow. Not a single one of these statements has any factual support, according to scientific research, legal rulings, and legitimate government authorities.
And yet public opinion surveys show millions of Americans, and others around the world, believe some of these falsehoods are true and can’t be convinced otherwise.
Social media, politicians and partisan websites, TV programs, and commentators have widely circulated these and other unfounded claims so frequently that many people say they simply can’t tell what’s objectively true and not anymore.
So much so,
The new study – The Rise and Fall of Rationality in Language, published in the Proceedings of the National Academy of Sciences – found that facts have become less important in public discourse.
As a result, unsupported beliefs have taken precedent over readily identifiable truths in discussions of health, science, and politics. The upshot: “Feelings trump facts” in social media, news reports, books, and other sources of information.
And here’s the kicker: The trend did not begin with the rise of former President Donald Trump, the COVID-19 pandemic, or the advent of social media; in fact, it has been growing for much longer than you might think.
“While the current ‘post-truth era’ has taken many by surprise, the study shows that over the past 40 years, public interest has undergone an accelerating shift from the collective to the individual, and from rationality towards emotion,” concluded the researchers from Indiana University and Wageningen University & Research in the Netherlands.
“Our work suggests that the societal balance between emotion and reason has shifted back to what it used to be around 150 years ago,” says lead researcher Marten Scheffer, PhD, a professor in the department of environmental sciences at WUR. “This implies that scientists, experts, and policymakers will have to think about the best way to respond to that social change.”
Researchers surprised by findings
The findings are based on a very detailed analysis of language from millions of books, newspaper articles, Google searches, TV reports, social media posts, and other sources dating back to 1850.
The researchers analyzed how often the 5,000 most used words appeared over the past 170 years and found that the use of those having to do with facts and reasoning, such as “determine” and “conclusion,” has fallen dramatically since 1980. Meanwhile, the use of words related to human emotion, such as “feel” and “believe,” have skyrocketed.
Dr. Scheffer notes rapid developments in science and technology from 1850 to 1980 had profound social and economic benefits that helped boost the status of the scientific approach. That shift in public attitudes had ripple effects on culture, society, education, politics, and religion – and “the role of spiritualism dwindled” in the modern world, he says.
But since 1980, that trend has seen a major reversal, with beliefs becoming more important than facts to many people, he says. At the same time, trust in science and scientists has fallen.
Dr. Scheffer says the researchers expected to find some evidence of a swing toward more belief-based sentiments during the Trump era but were surprised to discover how strong it is and that the trend has actually been a long time coming.
“The shift in interest from rational to intuitive/emotional is pretty obvious now in the post-truth political and social media discussion,” he says. “However, our work shows that it already started in the 1980s. For me personally, that went under the radar, except perhaps for the rise of alternative (to religion) forms of spirituality.
“We were especially struck by how strong the patterns are and how universal they appear across languages, nonfiction and fiction, and even in The New York Times.”
In the political world, the implications are significant enough – impacting policies and politicians on both sides of the aisle and across the globe. Just look at the deepening political divisions during the Trump presidency.
But for health and science, the spread of misinformation and falsehoods can be matters of life or death, as we have seen in the politically charged debates over how best to combat COVID-19 and global climate change.
“Our public debate seems increasingly driven by what people want to be true rather than what is actually true. As a scientist, that worries me,” says study co-author Johan Bollen, PhD, a professor of informatics at Indiana University.
“As a society, we are now faced with major collective problems that we need to approach from a pragmatic, rational, and objective perspective to be successful,” he says. “After all, global warming doesn’t care about whether you believe in it or not … but we will all suffer as a society if we fail to take adequate measures.”
For WUR co-researcher Ingrid van de Leemput, the trend isn’t merely academic; she’s seen it play out in her personal life.
“I do speak to people that, for instance, think the vaccines are poison,” she says. “I’m also on Twitter, and there, I’m every day surprised about how easily many people form their opinions, based on feelings, on what others say, or on some unfounded source.”
Public health experts say the embrace of personal beliefs over facts is one reason only 63% of Americans have been vaccinated against COVID-19. The result: millions of preventable infections among those who downplay the risks of the virus and reject the strong scientific evidence of vaccine safety and effectiveness.
“None of this really surprises me,” Johns Hopkins University social and behavioral scientist Rupali Limaye, PhD, says of the new study findings. Dr. Limaye coauthored a paper in 2016 in JAMA Pediatrics about how to talk to parents about vaccine hesitancy and the fact that we’re living in what they called “this post-truth era.”
Dr. Limaye says the trend has made it difficult for doctors, scientists, and health authorities to make fact-based arguments for COVID-19 vaccination, mask-wearing, social distancing, and other measures to control the virus.
“It’s been really hard being a scientist to hear people say, ‘Well, that’s not true’ when we say something very basic that I think all of us can agree on – like the grass is green,” she says. “To be honest, I worry that a lot of scientists are going to quit being in science because they’re exhausted.”
What’s driving the trend?
So, what’s behind the embrace of “alternative facts,” as former White House counselor Kellyanne Conway put it so brazenly in 2017, in defending the White House’s false claims that Trump’s inauguration crowd was the largest ever?
Dr. Scheffer and colleagues identified a handful of things that have encouraged the embrace of falsehoods over facts in recent years.
- The Internet: Its rise in the late 1980s, and its growing role as a primary source of news and information, has allowed more belief-based misinformation to flourish and spread like wildfire.
- Social media: The new study found the use of sentiment- and intuition-related words accelerated around 2007, along with a global surge in social media that catapulted Facebook, Twitter, and others into the mainstream, replacing more traditional fact-based media (i.e., newspapers and magazines).
- The 2007 financial crisis: The downturn in the global economy meant more people were dealing with job stress, investment losses, and other problems that fed the interest in belief-based, anti-establishment social media posts.
- Conspiracy theories: Falsehoods involving hidden political agendas, shadow “elites,” and wealthy people with dark motives tend to thrive during times of crisis and societal anxiety. “Conspiracy theories originate particularly in times of uncertainty and crisis and generally depict established institutions as hiding the truth and sustaining an unfair situation,” the researchers noted. “As a result, they may find fertile grounds on social media platforms promulgating a sense of unfairness, subsequently feeding anti-system sentiments.”
Dr. Scheffer says that growing political divisions during the Trump era have widened the fact-vs.-fiction divide. The ex-president voiced many anti-science views on global climate change, for instance, and spread so many falsehoods about COVID-19 and the 2020 election that Facebook, Twitter, and YouTube suspended his accounts.
Yet Trump remains a popular figure among Republicans, with most saying in a December poll they believe his baseless claims that the 2020 election was “rigged” and “stolen,” despite all credible, easily accessible evidence that it was secure, according to a recent poll by the University of Massachusetts at Amherst.
More than 60 courts have rejected Trump’s lawsuits seeking to overturn the election results. All 50 states, the District of Columbia, and both branches of Congress have certified the election results, giving Biden the White House. Even Trump’s own Justice Department confirmed that the 2020 election was free and fair.
Nevertheless, the University of Massachusetts survey found that most Republicans believe one or more conspiracy theories floated by the former president and those pushing his “big lie” that Democrats rigged the election to elect Biden.
Ed Berliner, an Emmy Award-winning broadcast journalist and media consultant, suggests something else is driving the spread of misinformation: the pursuit of ratings by cable TV and media companies to boost ad and subscriber revenues.
As a former executive producer and syndicated cable TV show host, he says he has seen firsthand how facts are often lost in opinion-driven news programs, even on network programs claiming to offer “fair and balanced” journalism.
“Propaganda is the new currency in America, and those who do not fight back against it are doomed to be overrun by the misinformation,” says Mr. Berliner, host of The Man in the Arena and CEO of Entourage Media LLC.
“The broadcast news media has to stop this incessant ‘infotainment’ prattle, stop trying to nuzzle up to a soft side, and bear down on hard facts, exposing the lies and refusing to back down.”
Public health implications
Public health and media experts alike say the PNAS study findings are disheartening but underscore the need for doctors and scientists to do a better job of communicating about COVID-19 and other pressing issues.
Dr. Limaye, from Johns Hopkins, is particularly concerned about the rise in conspiracy theories that has led to COVID-19 vaccine hesitancy.
“When we speak to individuals about getting the COVID vaccine…the types of concerns that come up now are very different than they were 8 years ago,” she says. “The comments we used to hear were much more related to vaccine safety. [People] would say, ‘I’m worried about an ingredient in the vaccine’ or ‘I’m worried that my kiddo has to get three different shots within 6 months to have a series dose completed.’”
But now, a lot of comments they receive are about government and pharma conspiracies.
What that means is doctors and scientists must do more than simply say “here are the facts” and “trust me, I’m a doctor or a scientist,” she says. And these approaches don’t only apply to public health.
“It’s funny, because when we talk to climate change scientists, as vaccine [specialists], we’ll say we can’t believe that people think COVID is a hoax,” she says. “And they’re like, ‘Hold my beer, we’ve been dealing with this for 20 years. Hello, it’s just your guys’ turn to deal with this public denial of science.’”
Dr. Limaye is also concerned about the impacts on funding for scientific research.
“There’s always been a really strong bipartisan effort with regards to funding for science, when you look at Congress and when you look at appropriations,” she says. “But what ended up happening, especially with the Trump administration, was that there was a real shift in that. We’ve never really seen that before in past generations.”
So, what’s the big take-home message?
Dr. Limaye believes doctors and public health experts must show more empathy – and not be combative or arrogant – in communicating science in one-on-one conversations. This month, she’s launching a new course for parents, school administrators, and nurses on how to do precisely that.
“It’s really all about how to have hard conversations with people who might be anti-science,” she says. “It’s being empathetic and not being dismissive. But it’s hard work, and I think a lot of people are just not cut out for it and just don’t have the time for it…You can’t just say, ‘Well, this is science, and I’m a doctor’ – that doesn’t work anymore.”
Brendan Nyhan, PhD, a Dartmouth College political scientist, echoes those sentiments in a separate paper recently published in the Proceedings of the National Academy of Sciences. In fact, he suggests that providing accurate, fact-based information to counter false claims may actually backfire and reinforce some people’s unfounded beliefs.
“One response to the prevalence of mistaken beliefs is to try to set the record straight by providing accurate information – for instance, by providing evidence of the scientific consensus on climate change,” he writes. “The failures of this approach, which is sometimes referred to as the ‘deficit model’ in science communication, are well-known.”
Dr. Nyhan argues two things make some people more prone to believe falsehoods:
What scientists call “ingrouping,” a kind of tribal mentality that makes some people choose social identity or politics over truth-seeking and demonize others who don’t agree with their views
The rise of high-profile political figures, such as Trump, who encourage their followers to indulge in their desire for “identify-affirming misinformation”
Dr. Scheffer says the most important thing for doctors, health experts, and scientists to recognize is that it’s crucial to gain the trust of someone who may believe fictions over facts to make any persuasive argument on COVID-19 or any other issue.
He also has a standard response to those who present falsehoods to him as facts that he suggests anyone can use: “That is interesting. Would you mind helping me understand how you came to that opinion?”
A version of this article first appeared on WebMD.com.
Can you tell which of the following statements are true and which are false?
COVID-19 is not a threat to younger people, and only those who have other medical conditions are dying from it.
The mRNA vaccines developed to prevent the coronavirus alter your genes, can make your body “magnetic,” and are killing more people than the virus itself.
President Joe Biden’s climate change plan calls for a ban on meat consumption to cut greenhouse gas emissions.
The 2020 presidential election was rigged and stolen.
If you guessed that all of these claims are false, you’re right – take a bow. Not a single one of these statements has any factual support, according to scientific research, legal rulings, and legitimate government authorities.
And yet public opinion surveys show millions of Americans, and others around the world, believe some of these falsehoods are true and can’t be convinced otherwise.
Social media, politicians and partisan websites, TV programs, and commentators have widely circulated these and other unfounded claims so frequently that many people say they simply can’t tell what’s objectively true and not anymore.
So much so,
The new study – The Rise and Fall of Rationality in Language, published in the Proceedings of the National Academy of Sciences – found that facts have become less important in public discourse.
As a result, unsupported beliefs have taken precedent over readily identifiable truths in discussions of health, science, and politics. The upshot: “Feelings trump facts” in social media, news reports, books, and other sources of information.
And here’s the kicker: The trend did not begin with the rise of former President Donald Trump, the COVID-19 pandemic, or the advent of social media; in fact, it has been growing for much longer than you might think.
“While the current ‘post-truth era’ has taken many by surprise, the study shows that over the past 40 years, public interest has undergone an accelerating shift from the collective to the individual, and from rationality towards emotion,” concluded the researchers from Indiana University and Wageningen University & Research in the Netherlands.
“Our work suggests that the societal balance between emotion and reason has shifted back to what it used to be around 150 years ago,” says lead researcher Marten Scheffer, PhD, a professor in the department of environmental sciences at WUR. “This implies that scientists, experts, and policymakers will have to think about the best way to respond to that social change.”
Researchers surprised by findings
The findings are based on a very detailed analysis of language from millions of books, newspaper articles, Google searches, TV reports, social media posts, and other sources dating back to 1850.
The researchers analyzed how often the 5,000 most used words appeared over the past 170 years and found that the use of those having to do with facts and reasoning, such as “determine” and “conclusion,” has fallen dramatically since 1980. Meanwhile, the use of words related to human emotion, such as “feel” and “believe,” have skyrocketed.
Dr. Scheffer notes rapid developments in science and technology from 1850 to 1980 had profound social and economic benefits that helped boost the status of the scientific approach. That shift in public attitudes had ripple effects on culture, society, education, politics, and religion – and “the role of spiritualism dwindled” in the modern world, he says.
But since 1980, that trend has seen a major reversal, with beliefs becoming more important than facts to many people, he says. At the same time, trust in science and scientists has fallen.
Dr. Scheffer says the researchers expected to find some evidence of a swing toward more belief-based sentiments during the Trump era but were surprised to discover how strong it is and that the trend has actually been a long time coming.
“The shift in interest from rational to intuitive/emotional is pretty obvious now in the post-truth political and social media discussion,” he says. “However, our work shows that it already started in the 1980s. For me personally, that went under the radar, except perhaps for the rise of alternative (to religion) forms of spirituality.
“We were especially struck by how strong the patterns are and how universal they appear across languages, nonfiction and fiction, and even in The New York Times.”
In the political world, the implications are significant enough – impacting policies and politicians on both sides of the aisle and across the globe. Just look at the deepening political divisions during the Trump presidency.
But for health and science, the spread of misinformation and falsehoods can be matters of life or death, as we have seen in the politically charged debates over how best to combat COVID-19 and global climate change.
“Our public debate seems increasingly driven by what people want to be true rather than what is actually true. As a scientist, that worries me,” says study co-author Johan Bollen, PhD, a professor of informatics at Indiana University.
“As a society, we are now faced with major collective problems that we need to approach from a pragmatic, rational, and objective perspective to be successful,” he says. “After all, global warming doesn’t care about whether you believe in it or not … but we will all suffer as a society if we fail to take adequate measures.”
For WUR co-researcher Ingrid van de Leemput, the trend isn’t merely academic; she’s seen it play out in her personal life.
“I do speak to people that, for instance, think the vaccines are poison,” she says. “I’m also on Twitter, and there, I’m every day surprised about how easily many people form their opinions, based on feelings, on what others say, or on some unfounded source.”
Public health experts say the embrace of personal beliefs over facts is one reason only 63% of Americans have been vaccinated against COVID-19. The result: millions of preventable infections among those who downplay the risks of the virus and reject the strong scientific evidence of vaccine safety and effectiveness.
“None of this really surprises me,” Johns Hopkins University social and behavioral scientist Rupali Limaye, PhD, says of the new study findings. Dr. Limaye coauthored a paper in 2016 in JAMA Pediatrics about how to talk to parents about vaccine hesitancy and the fact that we’re living in what they called “this post-truth era.”
Dr. Limaye says the trend has made it difficult for doctors, scientists, and health authorities to make fact-based arguments for COVID-19 vaccination, mask-wearing, social distancing, and other measures to control the virus.
“It’s been really hard being a scientist to hear people say, ‘Well, that’s not true’ when we say something very basic that I think all of us can agree on – like the grass is green,” she says. “To be honest, I worry that a lot of scientists are going to quit being in science because they’re exhausted.”
What’s driving the trend?
So, what’s behind the embrace of “alternative facts,” as former White House counselor Kellyanne Conway put it so brazenly in 2017, in defending the White House’s false claims that Trump’s inauguration crowd was the largest ever?
Dr. Scheffer and colleagues identified a handful of things that have encouraged the embrace of falsehoods over facts in recent years.
- The Internet: Its rise in the late 1980s, and its growing role as a primary source of news and information, has allowed more belief-based misinformation to flourish and spread like wildfire.
- Social media: The new study found the use of sentiment- and intuition-related words accelerated around 2007, along with a global surge in social media that catapulted Facebook, Twitter, and others into the mainstream, replacing more traditional fact-based media (i.e., newspapers and magazines).
- The 2007 financial crisis: The downturn in the global economy meant more people were dealing with job stress, investment losses, and other problems that fed the interest in belief-based, anti-establishment social media posts.
- Conspiracy theories: Falsehoods involving hidden political agendas, shadow “elites,” and wealthy people with dark motives tend to thrive during times of crisis and societal anxiety. “Conspiracy theories originate particularly in times of uncertainty and crisis and generally depict established institutions as hiding the truth and sustaining an unfair situation,” the researchers noted. “As a result, they may find fertile grounds on social media platforms promulgating a sense of unfairness, subsequently feeding anti-system sentiments.”
Dr. Scheffer says that growing political divisions during the Trump era have widened the fact-vs.-fiction divide. The ex-president voiced many anti-science views on global climate change, for instance, and spread so many falsehoods about COVID-19 and the 2020 election that Facebook, Twitter, and YouTube suspended his accounts.
Yet Trump remains a popular figure among Republicans, with most saying in a December poll they believe his baseless claims that the 2020 election was “rigged” and “stolen,” despite all credible, easily accessible evidence that it was secure, according to a recent poll by the University of Massachusetts at Amherst.
More than 60 courts have rejected Trump’s lawsuits seeking to overturn the election results. All 50 states, the District of Columbia, and both branches of Congress have certified the election results, giving Biden the White House. Even Trump’s own Justice Department confirmed that the 2020 election was free and fair.
Nevertheless, the University of Massachusetts survey found that most Republicans believe one or more conspiracy theories floated by the former president and those pushing his “big lie” that Democrats rigged the election to elect Biden.
Ed Berliner, an Emmy Award-winning broadcast journalist and media consultant, suggests something else is driving the spread of misinformation: the pursuit of ratings by cable TV and media companies to boost ad and subscriber revenues.
As a former executive producer and syndicated cable TV show host, he says he has seen firsthand how facts are often lost in opinion-driven news programs, even on network programs claiming to offer “fair and balanced” journalism.
“Propaganda is the new currency in America, and those who do not fight back against it are doomed to be overrun by the misinformation,” says Mr. Berliner, host of The Man in the Arena and CEO of Entourage Media LLC.
“The broadcast news media has to stop this incessant ‘infotainment’ prattle, stop trying to nuzzle up to a soft side, and bear down on hard facts, exposing the lies and refusing to back down.”
Public health implications
Public health and media experts alike say the PNAS study findings are disheartening but underscore the need for doctors and scientists to do a better job of communicating about COVID-19 and other pressing issues.
Dr. Limaye, from Johns Hopkins, is particularly concerned about the rise in conspiracy theories that has led to COVID-19 vaccine hesitancy.
“When we speak to individuals about getting the COVID vaccine…the types of concerns that come up now are very different than they were 8 years ago,” she says. “The comments we used to hear were much more related to vaccine safety. [People] would say, ‘I’m worried about an ingredient in the vaccine’ or ‘I’m worried that my kiddo has to get three different shots within 6 months to have a series dose completed.’”
But now, a lot of comments they receive are about government and pharma conspiracies.
What that means is doctors and scientists must do more than simply say “here are the facts” and “trust me, I’m a doctor or a scientist,” she says. And these approaches don’t only apply to public health.
“It’s funny, because when we talk to climate change scientists, as vaccine [specialists], we’ll say we can’t believe that people think COVID is a hoax,” she says. “And they’re like, ‘Hold my beer, we’ve been dealing with this for 20 years. Hello, it’s just your guys’ turn to deal with this public denial of science.’”
Dr. Limaye is also concerned about the impacts on funding for scientific research.
“There’s always been a really strong bipartisan effort with regards to funding for science, when you look at Congress and when you look at appropriations,” she says. “But what ended up happening, especially with the Trump administration, was that there was a real shift in that. We’ve never really seen that before in past generations.”
So, what’s the big take-home message?
Dr. Limaye believes doctors and public health experts must show more empathy – and not be combative or arrogant – in communicating science in one-on-one conversations. This month, she’s launching a new course for parents, school administrators, and nurses on how to do precisely that.
“It’s really all about how to have hard conversations with people who might be anti-science,” she says. “It’s being empathetic and not being dismissive. But it’s hard work, and I think a lot of people are just not cut out for it and just don’t have the time for it…You can’t just say, ‘Well, this is science, and I’m a doctor’ – that doesn’t work anymore.”
Brendan Nyhan, PhD, a Dartmouth College political scientist, echoes those sentiments in a separate paper recently published in the Proceedings of the National Academy of Sciences. In fact, he suggests that providing accurate, fact-based information to counter false claims may actually backfire and reinforce some people’s unfounded beliefs.
“One response to the prevalence of mistaken beliefs is to try to set the record straight by providing accurate information – for instance, by providing evidence of the scientific consensus on climate change,” he writes. “The failures of this approach, which is sometimes referred to as the ‘deficit model’ in science communication, are well-known.”
Dr. Nyhan argues two things make some people more prone to believe falsehoods:
What scientists call “ingrouping,” a kind of tribal mentality that makes some people choose social identity or politics over truth-seeking and demonize others who don’t agree with their views
The rise of high-profile political figures, such as Trump, who encourage their followers to indulge in their desire for “identify-affirming misinformation”
Dr. Scheffer says the most important thing for doctors, health experts, and scientists to recognize is that it’s crucial to gain the trust of someone who may believe fictions over facts to make any persuasive argument on COVID-19 or any other issue.
He also has a standard response to those who present falsehoods to him as facts that he suggests anyone can use: “That is interesting. Would you mind helping me understand how you came to that opinion?”
A version of this article first appeared on WebMD.com.
Can you tell which of the following statements are true and which are false?
COVID-19 is not a threat to younger people, and only those who have other medical conditions are dying from it.
The mRNA vaccines developed to prevent the coronavirus alter your genes, can make your body “magnetic,” and are killing more people than the virus itself.
President Joe Biden’s climate change plan calls for a ban on meat consumption to cut greenhouse gas emissions.
The 2020 presidential election was rigged and stolen.
If you guessed that all of these claims are false, you’re right – take a bow. Not a single one of these statements has any factual support, according to scientific research, legal rulings, and legitimate government authorities.
And yet public opinion surveys show millions of Americans, and others around the world, believe some of these falsehoods are true and can’t be convinced otherwise.
Social media, politicians and partisan websites, TV programs, and commentators have widely circulated these and other unfounded claims so frequently that many people say they simply can’t tell what’s objectively true and not anymore.
So much so,
The new study – The Rise and Fall of Rationality in Language, published in the Proceedings of the National Academy of Sciences – found that facts have become less important in public discourse.
As a result, unsupported beliefs have taken precedent over readily identifiable truths in discussions of health, science, and politics. The upshot: “Feelings trump facts” in social media, news reports, books, and other sources of information.
And here’s the kicker: The trend did not begin with the rise of former President Donald Trump, the COVID-19 pandemic, or the advent of social media; in fact, it has been growing for much longer than you might think.
“While the current ‘post-truth era’ has taken many by surprise, the study shows that over the past 40 years, public interest has undergone an accelerating shift from the collective to the individual, and from rationality towards emotion,” concluded the researchers from Indiana University and Wageningen University & Research in the Netherlands.
“Our work suggests that the societal balance between emotion and reason has shifted back to what it used to be around 150 years ago,” says lead researcher Marten Scheffer, PhD, a professor in the department of environmental sciences at WUR. “This implies that scientists, experts, and policymakers will have to think about the best way to respond to that social change.”
Researchers surprised by findings
The findings are based on a very detailed analysis of language from millions of books, newspaper articles, Google searches, TV reports, social media posts, and other sources dating back to 1850.
The researchers analyzed how often the 5,000 most used words appeared over the past 170 years and found that the use of those having to do with facts and reasoning, such as “determine” and “conclusion,” has fallen dramatically since 1980. Meanwhile, the use of words related to human emotion, such as “feel” and “believe,” have skyrocketed.
Dr. Scheffer notes rapid developments in science and technology from 1850 to 1980 had profound social and economic benefits that helped boost the status of the scientific approach. That shift in public attitudes had ripple effects on culture, society, education, politics, and religion – and “the role of spiritualism dwindled” in the modern world, he says.
But since 1980, that trend has seen a major reversal, with beliefs becoming more important than facts to many people, he says. At the same time, trust in science and scientists has fallen.
Dr. Scheffer says the researchers expected to find some evidence of a swing toward more belief-based sentiments during the Trump era but were surprised to discover how strong it is and that the trend has actually been a long time coming.
“The shift in interest from rational to intuitive/emotional is pretty obvious now in the post-truth political and social media discussion,” he says. “However, our work shows that it already started in the 1980s. For me personally, that went under the radar, except perhaps for the rise of alternative (to religion) forms of spirituality.
“We were especially struck by how strong the patterns are and how universal they appear across languages, nonfiction and fiction, and even in The New York Times.”
In the political world, the implications are significant enough – impacting policies and politicians on both sides of the aisle and across the globe. Just look at the deepening political divisions during the Trump presidency.
But for health and science, the spread of misinformation and falsehoods can be matters of life or death, as we have seen in the politically charged debates over how best to combat COVID-19 and global climate change.
“Our public debate seems increasingly driven by what people want to be true rather than what is actually true. As a scientist, that worries me,” says study co-author Johan Bollen, PhD, a professor of informatics at Indiana University.
“As a society, we are now faced with major collective problems that we need to approach from a pragmatic, rational, and objective perspective to be successful,” he says. “After all, global warming doesn’t care about whether you believe in it or not … but we will all suffer as a society if we fail to take adequate measures.”
For WUR co-researcher Ingrid van de Leemput, the trend isn’t merely academic; she’s seen it play out in her personal life.
“I do speak to people that, for instance, think the vaccines are poison,” she says. “I’m also on Twitter, and there, I’m every day surprised about how easily many people form their opinions, based on feelings, on what others say, or on some unfounded source.”
Public health experts say the embrace of personal beliefs over facts is one reason only 63% of Americans have been vaccinated against COVID-19. The result: millions of preventable infections among those who downplay the risks of the virus and reject the strong scientific evidence of vaccine safety and effectiveness.
“None of this really surprises me,” Johns Hopkins University social and behavioral scientist Rupali Limaye, PhD, says of the new study findings. Dr. Limaye coauthored a paper in 2016 in JAMA Pediatrics about how to talk to parents about vaccine hesitancy and the fact that we’re living in what they called “this post-truth era.”
Dr. Limaye says the trend has made it difficult for doctors, scientists, and health authorities to make fact-based arguments for COVID-19 vaccination, mask-wearing, social distancing, and other measures to control the virus.
“It’s been really hard being a scientist to hear people say, ‘Well, that’s not true’ when we say something very basic that I think all of us can agree on – like the grass is green,” she says. “To be honest, I worry that a lot of scientists are going to quit being in science because they’re exhausted.”
What’s driving the trend?
So, what’s behind the embrace of “alternative facts,” as former White House counselor Kellyanne Conway put it so brazenly in 2017, in defending the White House’s false claims that Trump’s inauguration crowd was the largest ever?
Dr. Scheffer and colleagues identified a handful of things that have encouraged the embrace of falsehoods over facts in recent years.
- The Internet: Its rise in the late 1980s, and its growing role as a primary source of news and information, has allowed more belief-based misinformation to flourish and spread like wildfire.
- Social media: The new study found the use of sentiment- and intuition-related words accelerated around 2007, along with a global surge in social media that catapulted Facebook, Twitter, and others into the mainstream, replacing more traditional fact-based media (i.e., newspapers and magazines).
- The 2007 financial crisis: The downturn in the global economy meant more people were dealing with job stress, investment losses, and other problems that fed the interest in belief-based, anti-establishment social media posts.
- Conspiracy theories: Falsehoods involving hidden political agendas, shadow “elites,” and wealthy people with dark motives tend to thrive during times of crisis and societal anxiety. “Conspiracy theories originate particularly in times of uncertainty and crisis and generally depict established institutions as hiding the truth and sustaining an unfair situation,” the researchers noted. “As a result, they may find fertile grounds on social media platforms promulgating a sense of unfairness, subsequently feeding anti-system sentiments.”
Dr. Scheffer says that growing political divisions during the Trump era have widened the fact-vs.-fiction divide. The ex-president voiced many anti-science views on global climate change, for instance, and spread so many falsehoods about COVID-19 and the 2020 election that Facebook, Twitter, and YouTube suspended his accounts.
Yet Trump remains a popular figure among Republicans, with most saying in a December poll they believe his baseless claims that the 2020 election was “rigged” and “stolen,” despite all credible, easily accessible evidence that it was secure, according to a recent poll by the University of Massachusetts at Amherst.
More than 60 courts have rejected Trump’s lawsuits seeking to overturn the election results. All 50 states, the District of Columbia, and both branches of Congress have certified the election results, giving Biden the White House. Even Trump’s own Justice Department confirmed that the 2020 election was free and fair.
Nevertheless, the University of Massachusetts survey found that most Republicans believe one or more conspiracy theories floated by the former president and those pushing his “big lie” that Democrats rigged the election to elect Biden.
Ed Berliner, an Emmy Award-winning broadcast journalist and media consultant, suggests something else is driving the spread of misinformation: the pursuit of ratings by cable TV and media companies to boost ad and subscriber revenues.
As a former executive producer and syndicated cable TV show host, he says he has seen firsthand how facts are often lost in opinion-driven news programs, even on network programs claiming to offer “fair and balanced” journalism.
“Propaganda is the new currency in America, and those who do not fight back against it are doomed to be overrun by the misinformation,” says Mr. Berliner, host of The Man in the Arena and CEO of Entourage Media LLC.
“The broadcast news media has to stop this incessant ‘infotainment’ prattle, stop trying to nuzzle up to a soft side, and bear down on hard facts, exposing the lies and refusing to back down.”
Public health implications
Public health and media experts alike say the PNAS study findings are disheartening but underscore the need for doctors and scientists to do a better job of communicating about COVID-19 and other pressing issues.
Dr. Limaye, from Johns Hopkins, is particularly concerned about the rise in conspiracy theories that has led to COVID-19 vaccine hesitancy.
“When we speak to individuals about getting the COVID vaccine…the types of concerns that come up now are very different than they were 8 years ago,” she says. “The comments we used to hear were much more related to vaccine safety. [People] would say, ‘I’m worried about an ingredient in the vaccine’ or ‘I’m worried that my kiddo has to get three different shots within 6 months to have a series dose completed.’”
But now, a lot of comments they receive are about government and pharma conspiracies.
What that means is doctors and scientists must do more than simply say “here are the facts” and “trust me, I’m a doctor or a scientist,” she says. And these approaches don’t only apply to public health.
“It’s funny, because when we talk to climate change scientists, as vaccine [specialists], we’ll say we can’t believe that people think COVID is a hoax,” she says. “And they’re like, ‘Hold my beer, we’ve been dealing with this for 20 years. Hello, it’s just your guys’ turn to deal with this public denial of science.’”
Dr. Limaye is also concerned about the impacts on funding for scientific research.
“There’s always been a really strong bipartisan effort with regards to funding for science, when you look at Congress and when you look at appropriations,” she says. “But what ended up happening, especially with the Trump administration, was that there was a real shift in that. We’ve never really seen that before in past generations.”
So, what’s the big take-home message?
Dr. Limaye believes doctors and public health experts must show more empathy – and not be combative or arrogant – in communicating science in one-on-one conversations. This month, she’s launching a new course for parents, school administrators, and nurses on how to do precisely that.
“It’s really all about how to have hard conversations with people who might be anti-science,” she says. “It’s being empathetic and not being dismissive. But it’s hard work, and I think a lot of people are just not cut out for it and just don’t have the time for it…You can’t just say, ‘Well, this is science, and I’m a doctor’ – that doesn’t work anymore.”
Brendan Nyhan, PhD, a Dartmouth College political scientist, echoes those sentiments in a separate paper recently published in the Proceedings of the National Academy of Sciences. In fact, he suggests that providing accurate, fact-based information to counter false claims may actually backfire and reinforce some people’s unfounded beliefs.
“One response to the prevalence of mistaken beliefs is to try to set the record straight by providing accurate information – for instance, by providing evidence of the scientific consensus on climate change,” he writes. “The failures of this approach, which is sometimes referred to as the ‘deficit model’ in science communication, are well-known.”
Dr. Nyhan argues two things make some people more prone to believe falsehoods:
What scientists call “ingrouping,” a kind of tribal mentality that makes some people choose social identity or politics over truth-seeking and demonize others who don’t agree with their views
The rise of high-profile political figures, such as Trump, who encourage their followers to indulge in their desire for “identify-affirming misinformation”
Dr. Scheffer says the most important thing for doctors, health experts, and scientists to recognize is that it’s crucial to gain the trust of someone who may believe fictions over facts to make any persuasive argument on COVID-19 or any other issue.
He also has a standard response to those who present falsehoods to him as facts that he suggests anyone can use: “That is interesting. Would you mind helping me understand how you came to that opinion?”
A version of this article first appeared on WebMD.com.
Alternative birthing practices tied to neonatal infection risk
Increasingly popular alternative peripartum practices such as water immersion and nonseverance of the umbilical cord may increase the risk of infections in newborns, a new clinical report from the American Academy of Pediatrics found.
Another perinatal measure potentially raising infection risk was placentophagy, according to a review led by Dawn Nolt, MD, MPH, a professor of pediatric infectious diseases at Oregon Health & Science University, Portland.
“Awareness of emerging alternative peripartum and neonatal practices helps pediatricians provide counseling to families before birth and to appropriately evaluate and treat neonates who have been exposed to these practices,” Dr. Nolt and colleagues wrote online in Pediatrics.
Amid growing inquiries made from women seeking a positive and meaningful birth experience through alternative approaches as well as reports of possibly related illness in newborns, Dr. Nolt’s group reviewed observational studies, case series, and medical society guidance on the risks associated with seven alternative birthing practices.
Based on their summation, it was not possible to quantify the actual risk associated with any one practice. “But of the seven we reviewed, as an infectious disease pediatrician I would say the most discernible immediate risk is likely attached to nonseverance of the cord,” Dr. Nolt said in an interview. “Left attached, the tissue can potentially necrote and transfer bacteria directly to the child.”
The authors made the following recommendations:
- Water immersion for labor and delivery. While this can increase the comfort of the mother in the first stages of labor, the water can become contaminated and increase the infant’s exposure to water-borne pathogens such as Legionella and Pseudomonas. It is not recommended after the second stage of labor and if offered, requires rigorous prophylactic and infection-control measures. This practice has also been linked to aspiration, drowning, hyponatremia, cord rupture, and death.
- Vaginal seeding. The skin, noses, and mouths of infants born by cesarean section are inoculated with swabs of vaginal fluid in order to expose them to vaginal bacteria that positively influence the infant’s microbiome. Of no known benefit, this measure can expose newborns to microbes such as group B Streptococcus and herpes simplex virus. Infants born by C-section receiving vaginal seeding should be evaluated the same way as those delivered vaginally.
- Umbilical cord nonseverance. Colloquially known as lotus birth, this is another practice with no evidence of advantage but with the potential to raise the risk of neonatal sepsis owing to the presence of necrotic umbilical or placental tissue. Some parents may view the placenta as a spiritual entity and fail to recognize it may be contaminated with harmful pathogens. Any placenta and umbilical cord attached to a febrile or ill-seeming neonate should be immediately removed.
- Placentophagy. Proponents believe placental consumption has antidepressive, analgesic, galactogogic, and nutritional properties. But eating raw, cooked, or dehydrated afterbirth tissue – viewed by some as a spiritual event – can expose a neonate to flora from the mother’s genitourinary tract and other sources encountered during preparation. Placentophagy has been associated with a case of recurrent late-onset group B streptococcal sepsis in a newborn. Strict food-handling practices at the level for raw meat should be maintained.
- HBV vaccine deferral. Viewed as “a critical safety net in preventing HBV infection,” the birth dose of the hepatitis B virus vaccine should not be postponed except for medical reasons. An estimated 1,000 new perinatally acquired HBV cases occurred annually in the United States from 2000 to 2009.
- Deferral of ocular prophylaxis. While ocular prophylaxis with topical erythromycin protects against gonococcal ophthalmia neonatorum, particularly in infants of high-risk mothers, it is not effective against other common pathogens. Parents and health care providers have recently questioned the need for its routine application, with concerns including its limited range of effectiveness as well as antibiotic resistance and shortages. With adequate prenatal testing, the risk of this neonatal conjunctivitis is significantly reduced, and deferral of prophylaxis may be considered in low-risk situations although it may be mandated by state legislation.
- Delayed bathing. The practice of delaying the infant’s first bath until several hours after birth may have several benefits. These include the initiation and exclusivity of breastfeeding, decreased mother/child separation time and risk of hypothermia, and protection of the neonatal skin microbiome. It should be discouraged, however, in neonates exposed to active herpes simplex virus lesions or whose mothers have a known history of HIV infection.
When women inquire about alternative practices, physicians need to strike a diplomatic balance between respecting women’s wishes and the benefits they hope to gain and at the same time informing them of potential risks, Dr. Nolt said. “The conversation we want to have with them should show compassion and sympathy but also tell them what the medical literature shows.” Patient and doctor should engage in shared decision-making about the safety of various alternative approaches.
“Over the last decade information on a variety of birth practices have become more widely available through social media and other Internet forums, which certainly has increased the variety of questions to health professionals, Amy C. Hermesch, MD, PhD, director of obstetric services at OHSC, said in an interview.
“We counsel about rare but serious risk, as noted in Dr. Nolt’s article,” said Dr. Hermesch, who was not involved in the AAP report. Most important is a discussion about appropriate pregnancy risk stratification. “For example, persons considering water immersion birth, probably the most common one I get inquiries about, should have an otherwise uncomplicated pregnancy with good mobility to get in and out of tub in the event of an emergency.”
While adverse events can happen during any birth, she sees these more often in mothers who underestimate the risk level of their situation or pregnancy when declining provider-recommended interventions. “I encourage pregnant persons to find a health care professional they trust who is knowledgeable about the benefits and the risk of all birth environments and interventions.”
Dr. Hermesch added that most alternative practices have little data to guide decisions, so she offers professional society recommendations, evidence review, and her own professional experiences. “The patient must weight the risk and benefits in the context of their value system and sometimes this means not following my advice or recommendations. My medical recommendation with the best of intentions does not remove patient autonomy.”
This report had no external funding. The authors had no potential conflicts of interest to disclose. Dr. Hermesch had no competing interests to declare.
Increasingly popular alternative peripartum practices such as water immersion and nonseverance of the umbilical cord may increase the risk of infections in newborns, a new clinical report from the American Academy of Pediatrics found.
Another perinatal measure potentially raising infection risk was placentophagy, according to a review led by Dawn Nolt, MD, MPH, a professor of pediatric infectious diseases at Oregon Health & Science University, Portland.
“Awareness of emerging alternative peripartum and neonatal practices helps pediatricians provide counseling to families before birth and to appropriately evaluate and treat neonates who have been exposed to these practices,” Dr. Nolt and colleagues wrote online in Pediatrics.
Amid growing inquiries made from women seeking a positive and meaningful birth experience through alternative approaches as well as reports of possibly related illness in newborns, Dr. Nolt’s group reviewed observational studies, case series, and medical society guidance on the risks associated with seven alternative birthing practices.
Based on their summation, it was not possible to quantify the actual risk associated with any one practice. “But of the seven we reviewed, as an infectious disease pediatrician I would say the most discernible immediate risk is likely attached to nonseverance of the cord,” Dr. Nolt said in an interview. “Left attached, the tissue can potentially necrote and transfer bacteria directly to the child.”
The authors made the following recommendations:
- Water immersion for labor and delivery. While this can increase the comfort of the mother in the first stages of labor, the water can become contaminated and increase the infant’s exposure to water-borne pathogens such as Legionella and Pseudomonas. It is not recommended after the second stage of labor and if offered, requires rigorous prophylactic and infection-control measures. This practice has also been linked to aspiration, drowning, hyponatremia, cord rupture, and death.
- Vaginal seeding. The skin, noses, and mouths of infants born by cesarean section are inoculated with swabs of vaginal fluid in order to expose them to vaginal bacteria that positively influence the infant’s microbiome. Of no known benefit, this measure can expose newborns to microbes such as group B Streptococcus and herpes simplex virus. Infants born by C-section receiving vaginal seeding should be evaluated the same way as those delivered vaginally.
- Umbilical cord nonseverance. Colloquially known as lotus birth, this is another practice with no evidence of advantage but with the potential to raise the risk of neonatal sepsis owing to the presence of necrotic umbilical or placental tissue. Some parents may view the placenta as a spiritual entity and fail to recognize it may be contaminated with harmful pathogens. Any placenta and umbilical cord attached to a febrile or ill-seeming neonate should be immediately removed.
- Placentophagy. Proponents believe placental consumption has antidepressive, analgesic, galactogogic, and nutritional properties. But eating raw, cooked, or dehydrated afterbirth tissue – viewed by some as a spiritual event – can expose a neonate to flora from the mother’s genitourinary tract and other sources encountered during preparation. Placentophagy has been associated with a case of recurrent late-onset group B streptococcal sepsis in a newborn. Strict food-handling practices at the level for raw meat should be maintained.
- HBV vaccine deferral. Viewed as “a critical safety net in preventing HBV infection,” the birth dose of the hepatitis B virus vaccine should not be postponed except for medical reasons. An estimated 1,000 new perinatally acquired HBV cases occurred annually in the United States from 2000 to 2009.
- Deferral of ocular prophylaxis. While ocular prophylaxis with topical erythromycin protects against gonococcal ophthalmia neonatorum, particularly in infants of high-risk mothers, it is not effective against other common pathogens. Parents and health care providers have recently questioned the need for its routine application, with concerns including its limited range of effectiveness as well as antibiotic resistance and shortages. With adequate prenatal testing, the risk of this neonatal conjunctivitis is significantly reduced, and deferral of prophylaxis may be considered in low-risk situations although it may be mandated by state legislation.
- Delayed bathing. The practice of delaying the infant’s first bath until several hours after birth may have several benefits. These include the initiation and exclusivity of breastfeeding, decreased mother/child separation time and risk of hypothermia, and protection of the neonatal skin microbiome. It should be discouraged, however, in neonates exposed to active herpes simplex virus lesions or whose mothers have a known history of HIV infection.
When women inquire about alternative practices, physicians need to strike a diplomatic balance between respecting women’s wishes and the benefits they hope to gain and at the same time informing them of potential risks, Dr. Nolt said. “The conversation we want to have with them should show compassion and sympathy but also tell them what the medical literature shows.” Patient and doctor should engage in shared decision-making about the safety of various alternative approaches.
“Over the last decade information on a variety of birth practices have become more widely available through social media and other Internet forums, which certainly has increased the variety of questions to health professionals, Amy C. Hermesch, MD, PhD, director of obstetric services at OHSC, said in an interview.
“We counsel about rare but serious risk, as noted in Dr. Nolt’s article,” said Dr. Hermesch, who was not involved in the AAP report. Most important is a discussion about appropriate pregnancy risk stratification. “For example, persons considering water immersion birth, probably the most common one I get inquiries about, should have an otherwise uncomplicated pregnancy with good mobility to get in and out of tub in the event of an emergency.”
While adverse events can happen during any birth, she sees these more often in mothers who underestimate the risk level of their situation or pregnancy when declining provider-recommended interventions. “I encourage pregnant persons to find a health care professional they trust who is knowledgeable about the benefits and the risk of all birth environments and interventions.”
Dr. Hermesch added that most alternative practices have little data to guide decisions, so she offers professional society recommendations, evidence review, and her own professional experiences. “The patient must weight the risk and benefits in the context of their value system and sometimes this means not following my advice or recommendations. My medical recommendation with the best of intentions does not remove patient autonomy.”
This report had no external funding. The authors had no potential conflicts of interest to disclose. Dr. Hermesch had no competing interests to declare.
Increasingly popular alternative peripartum practices such as water immersion and nonseverance of the umbilical cord may increase the risk of infections in newborns, a new clinical report from the American Academy of Pediatrics found.
Another perinatal measure potentially raising infection risk was placentophagy, according to a review led by Dawn Nolt, MD, MPH, a professor of pediatric infectious diseases at Oregon Health & Science University, Portland.
“Awareness of emerging alternative peripartum and neonatal practices helps pediatricians provide counseling to families before birth and to appropriately evaluate and treat neonates who have been exposed to these practices,” Dr. Nolt and colleagues wrote online in Pediatrics.
Amid growing inquiries made from women seeking a positive and meaningful birth experience through alternative approaches as well as reports of possibly related illness in newborns, Dr. Nolt’s group reviewed observational studies, case series, and medical society guidance on the risks associated with seven alternative birthing practices.
Based on their summation, it was not possible to quantify the actual risk associated with any one practice. “But of the seven we reviewed, as an infectious disease pediatrician I would say the most discernible immediate risk is likely attached to nonseverance of the cord,” Dr. Nolt said in an interview. “Left attached, the tissue can potentially necrote and transfer bacteria directly to the child.”
The authors made the following recommendations:
- Water immersion for labor and delivery. While this can increase the comfort of the mother in the first stages of labor, the water can become contaminated and increase the infant’s exposure to water-borne pathogens such as Legionella and Pseudomonas. It is not recommended after the second stage of labor and if offered, requires rigorous prophylactic and infection-control measures. This practice has also been linked to aspiration, drowning, hyponatremia, cord rupture, and death.
- Vaginal seeding. The skin, noses, and mouths of infants born by cesarean section are inoculated with swabs of vaginal fluid in order to expose them to vaginal bacteria that positively influence the infant’s microbiome. Of no known benefit, this measure can expose newborns to microbes such as group B Streptococcus and herpes simplex virus. Infants born by C-section receiving vaginal seeding should be evaluated the same way as those delivered vaginally.
- Umbilical cord nonseverance. Colloquially known as lotus birth, this is another practice with no evidence of advantage but with the potential to raise the risk of neonatal sepsis owing to the presence of necrotic umbilical or placental tissue. Some parents may view the placenta as a spiritual entity and fail to recognize it may be contaminated with harmful pathogens. Any placenta and umbilical cord attached to a febrile or ill-seeming neonate should be immediately removed.
- Placentophagy. Proponents believe placental consumption has antidepressive, analgesic, galactogogic, and nutritional properties. But eating raw, cooked, or dehydrated afterbirth tissue – viewed by some as a spiritual event – can expose a neonate to flora from the mother’s genitourinary tract and other sources encountered during preparation. Placentophagy has been associated with a case of recurrent late-onset group B streptococcal sepsis in a newborn. Strict food-handling practices at the level for raw meat should be maintained.
- HBV vaccine deferral. Viewed as “a critical safety net in preventing HBV infection,” the birth dose of the hepatitis B virus vaccine should not be postponed except for medical reasons. An estimated 1,000 new perinatally acquired HBV cases occurred annually in the United States from 2000 to 2009.
- Deferral of ocular prophylaxis. While ocular prophylaxis with topical erythromycin protects against gonococcal ophthalmia neonatorum, particularly in infants of high-risk mothers, it is not effective against other common pathogens. Parents and health care providers have recently questioned the need for its routine application, with concerns including its limited range of effectiveness as well as antibiotic resistance and shortages. With adequate prenatal testing, the risk of this neonatal conjunctivitis is significantly reduced, and deferral of prophylaxis may be considered in low-risk situations although it may be mandated by state legislation.
- Delayed bathing. The practice of delaying the infant’s first bath until several hours after birth may have several benefits. These include the initiation and exclusivity of breastfeeding, decreased mother/child separation time and risk of hypothermia, and protection of the neonatal skin microbiome. It should be discouraged, however, in neonates exposed to active herpes simplex virus lesions or whose mothers have a known history of HIV infection.
When women inquire about alternative practices, physicians need to strike a diplomatic balance between respecting women’s wishes and the benefits they hope to gain and at the same time informing them of potential risks, Dr. Nolt said. “The conversation we want to have with them should show compassion and sympathy but also tell them what the medical literature shows.” Patient and doctor should engage in shared decision-making about the safety of various alternative approaches.
“Over the last decade information on a variety of birth practices have become more widely available through social media and other Internet forums, which certainly has increased the variety of questions to health professionals, Amy C. Hermesch, MD, PhD, director of obstetric services at OHSC, said in an interview.
“We counsel about rare but serious risk, as noted in Dr. Nolt’s article,” said Dr. Hermesch, who was not involved in the AAP report. Most important is a discussion about appropriate pregnancy risk stratification. “For example, persons considering water immersion birth, probably the most common one I get inquiries about, should have an otherwise uncomplicated pregnancy with good mobility to get in and out of tub in the event of an emergency.”
While adverse events can happen during any birth, she sees these more often in mothers who underestimate the risk level of their situation or pregnancy when declining provider-recommended interventions. “I encourage pregnant persons to find a health care professional they trust who is knowledgeable about the benefits and the risk of all birth environments and interventions.”
Dr. Hermesch added that most alternative practices have little data to guide decisions, so she offers professional society recommendations, evidence review, and her own professional experiences. “The patient must weight the risk and benefits in the context of their value system and sometimes this means not following my advice or recommendations. My medical recommendation with the best of intentions does not remove patient autonomy.”
This report had no external funding. The authors had no potential conflicts of interest to disclose. Dr. Hermesch had no competing interests to declare.
FROM PEDIATRICS
Physician burnout, depression compounded by COVID: Survey
In 2020, it was hard to imagine that the situation could get worse for doctors.
But 2021 presented a new set of challenges. As quarantines lifted and physicians tried to get back to work, they were forced to deal with reduced staff, continuing COVID stress, and pandemic-related anxieties about family and loved ones.
Medscape’s National Burnout and Depression Report 2022 asked more than 13,000 physicians from 29 specialties to share details about their lives and struggles with burnout and depression in 2021. The results paint a picture of physicians trying to fulfill their mission to care for patients, but struggling to maintain their own well-being amid a global pandemic.
Burnout bump
In 2021’s report, 42% of physicians said they were burned out. In 2022, that number increased to 47%. Perhaps not surprisingly, burnout among emergency physicians took the biggest leap, increasing from 43% to 60%. Critical care (56%), ob.gyn. (53%), and infectious disease and family medicine (both at 51%) rounded out the top five specialties with doctors experiencing burnout in 2021.
Burnout has typically been a greater problem for women than men physicians, and the pandemic hasn’t changed that. “There’s no question that women have reported far more role strain during the pandemic than men,” says Carol A. Bernstein, MD, psychiatrist at Montefiore Health System and professor and vice chair for faculty development and well-being at the Albert Einstein College of Medicine, both in New York. And indeed, 56% of women and 41% of men reported burnout in the 2022 survey.
The causes, however, weren’t especially pandemic related – or at least not directly. As in previous surveys, the major contributing factor to burnout was too much paperwork (60%), such as charting and other bureaucratic tasks. Treating COVID-19 patients was cited as the major source of stress by 10% of respondents. About 34% said too many hours at work was the biggest contributing factor to burnout.
The nature of the beast
What is burnout like for these doctors? One described the conditions that lead to burnout like this: “I barely spend enough time with most patients, just running from one to the next; and then after work, I spend hours documenting, charting, dealing with reports. I feel like an overpaid clerk.” Another said: “Where’s the relationships with patients that used to make this worthwhile?” Others fingered staffing shortages at work or an overwhelming home life: “Staff calls in sick; we’re all running around trying to find things and get things done. It never ends.”
Of those who do experience burnout, the problem reaches beyond the workplace, with 54% saying that their burnout has a strong/severe impact on life and 68% reporting that burnout affects their relationships. One respondent said: “I’m always tired; I have trouble concentrating, no time for the children, more arguments with my hubby.” Another put it this way: “Home is just as busy and chaotic as work. I can never relax.”
It doesn’t help matters that physicians are likely to think they’re the only professionals experiencing job burnout. For example, only 36% of respondents believe teachers experience comparable burnout, yet more than 41% of teachers leave the profession within 5 years of starting – often because of burnout.
When it comes to methods for coping with burnout, exercise is the clear favorite, with 63% of respondents saying exercise helps maintain their mental health. About 41% talk with family members or close friends. However, less healthy coping mechanisms were cited as well, such as isolating themselves from others (45%), sleeping (41%), and eating junk food (35%) or drinking alcohol (24%).
When it comes to trying to alleviate burnout, 29% have tried meditation or similar stress-reduction techniques, while others have reduced their work hours (29%) or changed their work settings (19%).
‘Now I feel like there’s no hope’
About a fifth of physicians (21%) said they suffered from clinical depression, and 64% reported feeling “blue, down, or sad.” One physician characterized their depression this way: “I used to think my life would be great. Now I feel like there’s no hope, this will never get better, I’ll never be happy.”
Of doctors reporting depression, 53% said their illness did not affect their interactions with patients, while 34% said depression caused them to be more easily exasperated by patients.
When asked about seeking help for depression, about half (49%) said they believed they could deal with emotional stress on their own. Unfortunately, fear of medical boards finding out keeps 43% of physicians from reaching out for help, according to the survey.
A version of this article first appeared on Medscape.com.
In 2020, it was hard to imagine that the situation could get worse for doctors.
But 2021 presented a new set of challenges. As quarantines lifted and physicians tried to get back to work, they were forced to deal with reduced staff, continuing COVID stress, and pandemic-related anxieties about family and loved ones.
Medscape’s National Burnout and Depression Report 2022 asked more than 13,000 physicians from 29 specialties to share details about their lives and struggles with burnout and depression in 2021. The results paint a picture of physicians trying to fulfill their mission to care for patients, but struggling to maintain their own well-being amid a global pandemic.
Burnout bump
In 2021’s report, 42% of physicians said they were burned out. In 2022, that number increased to 47%. Perhaps not surprisingly, burnout among emergency physicians took the biggest leap, increasing from 43% to 60%. Critical care (56%), ob.gyn. (53%), and infectious disease and family medicine (both at 51%) rounded out the top five specialties with doctors experiencing burnout in 2021.
Burnout has typically been a greater problem for women than men physicians, and the pandemic hasn’t changed that. “There’s no question that women have reported far more role strain during the pandemic than men,” says Carol A. Bernstein, MD, psychiatrist at Montefiore Health System and professor and vice chair for faculty development and well-being at the Albert Einstein College of Medicine, both in New York. And indeed, 56% of women and 41% of men reported burnout in the 2022 survey.
The causes, however, weren’t especially pandemic related – or at least not directly. As in previous surveys, the major contributing factor to burnout was too much paperwork (60%), such as charting and other bureaucratic tasks. Treating COVID-19 patients was cited as the major source of stress by 10% of respondents. About 34% said too many hours at work was the biggest contributing factor to burnout.
The nature of the beast
What is burnout like for these doctors? One described the conditions that lead to burnout like this: “I barely spend enough time with most patients, just running from one to the next; and then after work, I spend hours documenting, charting, dealing with reports. I feel like an overpaid clerk.” Another said: “Where’s the relationships with patients that used to make this worthwhile?” Others fingered staffing shortages at work or an overwhelming home life: “Staff calls in sick; we’re all running around trying to find things and get things done. It never ends.”
Of those who do experience burnout, the problem reaches beyond the workplace, with 54% saying that their burnout has a strong/severe impact on life and 68% reporting that burnout affects their relationships. One respondent said: “I’m always tired; I have trouble concentrating, no time for the children, more arguments with my hubby.” Another put it this way: “Home is just as busy and chaotic as work. I can never relax.”
It doesn’t help matters that physicians are likely to think they’re the only professionals experiencing job burnout. For example, only 36% of respondents believe teachers experience comparable burnout, yet more than 41% of teachers leave the profession within 5 years of starting – often because of burnout.
When it comes to methods for coping with burnout, exercise is the clear favorite, with 63% of respondents saying exercise helps maintain their mental health. About 41% talk with family members or close friends. However, less healthy coping mechanisms were cited as well, such as isolating themselves from others (45%), sleeping (41%), and eating junk food (35%) or drinking alcohol (24%).
When it comes to trying to alleviate burnout, 29% have tried meditation or similar stress-reduction techniques, while others have reduced their work hours (29%) or changed their work settings (19%).
‘Now I feel like there’s no hope’
About a fifth of physicians (21%) said they suffered from clinical depression, and 64% reported feeling “blue, down, or sad.” One physician characterized their depression this way: “I used to think my life would be great. Now I feel like there’s no hope, this will never get better, I’ll never be happy.”
Of doctors reporting depression, 53% said their illness did not affect their interactions with patients, while 34% said depression caused them to be more easily exasperated by patients.
When asked about seeking help for depression, about half (49%) said they believed they could deal with emotional stress on their own. Unfortunately, fear of medical boards finding out keeps 43% of physicians from reaching out for help, according to the survey.
A version of this article first appeared on Medscape.com.
In 2020, it was hard to imagine that the situation could get worse for doctors.
But 2021 presented a new set of challenges. As quarantines lifted and physicians tried to get back to work, they were forced to deal with reduced staff, continuing COVID stress, and pandemic-related anxieties about family and loved ones.
Medscape’s National Burnout and Depression Report 2022 asked more than 13,000 physicians from 29 specialties to share details about their lives and struggles with burnout and depression in 2021. The results paint a picture of physicians trying to fulfill their mission to care for patients, but struggling to maintain their own well-being amid a global pandemic.
Burnout bump
In 2021’s report, 42% of physicians said they were burned out. In 2022, that number increased to 47%. Perhaps not surprisingly, burnout among emergency physicians took the biggest leap, increasing from 43% to 60%. Critical care (56%), ob.gyn. (53%), and infectious disease and family medicine (both at 51%) rounded out the top five specialties with doctors experiencing burnout in 2021.
Burnout has typically been a greater problem for women than men physicians, and the pandemic hasn’t changed that. “There’s no question that women have reported far more role strain during the pandemic than men,” says Carol A. Bernstein, MD, psychiatrist at Montefiore Health System and professor and vice chair for faculty development and well-being at the Albert Einstein College of Medicine, both in New York. And indeed, 56% of women and 41% of men reported burnout in the 2022 survey.
The causes, however, weren’t especially pandemic related – or at least not directly. As in previous surveys, the major contributing factor to burnout was too much paperwork (60%), such as charting and other bureaucratic tasks. Treating COVID-19 patients was cited as the major source of stress by 10% of respondents. About 34% said too many hours at work was the biggest contributing factor to burnout.
The nature of the beast
What is burnout like for these doctors? One described the conditions that lead to burnout like this: “I barely spend enough time with most patients, just running from one to the next; and then after work, I spend hours documenting, charting, dealing with reports. I feel like an overpaid clerk.” Another said: “Where’s the relationships with patients that used to make this worthwhile?” Others fingered staffing shortages at work or an overwhelming home life: “Staff calls in sick; we’re all running around trying to find things and get things done. It never ends.”
Of those who do experience burnout, the problem reaches beyond the workplace, with 54% saying that their burnout has a strong/severe impact on life and 68% reporting that burnout affects their relationships. One respondent said: “I’m always tired; I have trouble concentrating, no time for the children, more arguments with my hubby.” Another put it this way: “Home is just as busy and chaotic as work. I can never relax.”
It doesn’t help matters that physicians are likely to think they’re the only professionals experiencing job burnout. For example, only 36% of respondents believe teachers experience comparable burnout, yet more than 41% of teachers leave the profession within 5 years of starting – often because of burnout.
When it comes to methods for coping with burnout, exercise is the clear favorite, with 63% of respondents saying exercise helps maintain their mental health. About 41% talk with family members or close friends. However, less healthy coping mechanisms were cited as well, such as isolating themselves from others (45%), sleeping (41%), and eating junk food (35%) or drinking alcohol (24%).
When it comes to trying to alleviate burnout, 29% have tried meditation or similar stress-reduction techniques, while others have reduced their work hours (29%) or changed their work settings (19%).
‘Now I feel like there’s no hope’
About a fifth of physicians (21%) said they suffered from clinical depression, and 64% reported feeling “blue, down, or sad.” One physician characterized their depression this way: “I used to think my life would be great. Now I feel like there’s no hope, this will never get better, I’ll never be happy.”
Of doctors reporting depression, 53% said their illness did not affect their interactions with patients, while 34% said depression caused them to be more easily exasperated by patients.
When asked about seeking help for depression, about half (49%) said they believed they could deal with emotional stress on their own. Unfortunately, fear of medical boards finding out keeps 43% of physicians from reaching out for help, according to the survey.
A version of this article first appeared on Medscape.com.
Peanut oral immunotherapy is safe and effective in toddlers in large placebo-controlled trial
In a large, blinded study of peanut-allergic toddlers published in The Lancet, 71% of treated participants could safely consume 5,000 mg of peanut protein – equivalent to nearly 17 peanuts – after 2½ years on oral immunotherapy. Even after stopping maintenance dosing for the next 6 months, more than 1 in 5 maintained that level of protection, and nearly 3 in 5 still met the 600-mg benchmark (about 2 peanuts) set by the phase 3 PALISADE trial of the FDA-approved peanut-flour product, Palforzia.
About 2% of children in the United States are allergic to peanuts, and most will not outgrow this allergy. In addition, other research suggests that the immune system is more malleable during early childhood.
Consistent with this idea, prior research showed that toddlers can succeed with peanut oral immunotherapy (OIT) – a regimen that builds tolerance through small amounts of the allergen consumed daily for months. However, that trial (DEVIL) was small, was conducted at a single site, and had no placebo group.
In contrast, the Peanut Oral Immunotherapy in Children Trial (IMPACT) enrolled 146 children aged 1-3 years at five academic medical centers in the United States – the first placebo-controlled study of OIT in this younger age group.
“This is a well done study,” Jaclyn Bjelac, MD, associate director of the Food Allergy Center of Excellence at the Cleveland Clinic, told this news organization. “We have seen improved outcomes in OIT, both in our own experience and other published studies, so while this is no surprise, the outcomes and large number of participants contribute to this being a really exciting publication.”
The trial was long and demanding for families. Toddlers who reacted to 500 mg or less of peanut protein in an entry food challenge were randomized in a 2:1 ratio to receive daily peanut flour or oat flour placebo. After initial dose escalation (from 0.1 mg to 6 mg) and biweekly buildup to a 2,000-mg target dose by week 30, participants continued with 20,00-mg daily maintenance dosing through week 134 – at which point they underwent a food challenge. They then went off treatment for 26 weeks and had another food challenge (week 160). In addition, participants came in for skin-prick and blood tests at baseline and at weeks 30, 82, 134, and 160.
In the placebo group, only 23 of 50 participants (46%) completed the study. “If you did 2½ years of this and then bombed the food challenge, you probably can guess that you were not on the real thing. And they were still asked to come back in 6 months and do it again. So, sure enough, a big chunk of those people chose not to continue, and you can’t blame them,” said Lancet co-author Edwin Kim, MD, in an interview. Dr. Kim directs the UNC Food Allergy Initiative at the University of North Carolina School of Medicine, Chapel Hill.
There was attrition in the treatment group as well. Among 96 children initially assigned to this arm, 68 (71%) passed the 5,000-mg peanut challenge at week 134 – but 11 withdrew in the study’s off-treatment phase. “It was a very tough decision. How much do you give toward science?” said Dr. Kim. “When push came to shove, some of the families couldn’t pull the trigger to potentially give up what they worked so hard for.”
In the intention-to-treat analysis, 20 of 96 treated participants (21%) could still tolerate 5,000 mg of peanut protein after going off therapy for 6 months. That translates to a 29% remission rate in the per-protocol subset (n = 70) who completed the study. Forty (57%) of these completers safely consumed at least 1,755 mg of peanut (cumulative dose). By comparison, the PALISADE trial of Palforzia used a 10,430-mg cumulative peanut dose to measure treatment efficacy.
On safety, 98% of treated participants – but also 80% of the placebo group – reported reactions, of which 35 were treated with epinephrine in 21 children receiving peanut OIT.
While some have noted that epinephrine use seemed high, Dr. Kim said, “we’re actually OK with that, because we’d much rather they overtreat and make sure that 1-year-old is safe than take any chances.” Overall, the safety profile looks similar to prior OIT studies of older children. “I think it suggests that, yeah, side effects will happen, [but] they’re all manageable, and people are not anaphylaxing left and right.”
On remission and immunologic parameters, benefits seemed stronger in the youngest subset (12 to 24 months), particularly those with lower peanut-specific IgE at baseline. These trends require further analyses, though, given the limited number of participants under 24 months.
Another noteworthy observation from longitudinal peanut-specific IgE trends in the placebo group: “Avoidance may not be benign,” Dr. Kim said. “If you look at their labs, they don’t stay flat. They actually go up.” The results jibe with the long-held idea of an early window of opportunity while a child’s immune system is maturing. “If you can grab this kid when his IgE is 10, versus next year when it might be 50, maybe you’ll get a different treatment effect,” Dr. Kim said. “We don’t know that for sure, but the placebo labs kind of point toward that.”
Beyond the science, there are practical advantages to starting OIT early. “Trying to convince a 9-year-old who’s been petrified of peanuts for their whole life to start doing this every day is not an easy task,” whereas with a 1- or 2-year-old, “you build it into their routine,” Dr. Kim said.
Plus, some say there’s no need for families to wait for regulatory approval of additional commercial products for very young children. Though some have advocated against the use of “grocery store” products, most peanut OIT research “has used the same 12% light roast defatted peanut flour used in IMPACT,” noted Marcus S. Shaker, MD, professor of pediatrics and of medicine at the Dartmouth Geisel School of Medicine and a physician at the Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire. The commercial product (Palforzia) and grocery-store products “come from the exact same source in the U.S.,” he said in an interview. “Both are an option for parents to consider, but a commercial product is not, nor has [it] ever been, a necessity.”
Dr. Bjelac reports no relevant financial relationships. Dr. Kim reports consultancy with Aimmune Therapeutics, Allako, AllerGenis, Belhaven Pharma, DBV Technologies, Duke Clinical Research Institute, and Nutricia; advisory board membership with ALK, DBV Technologies, Kenota Health, and Ukko; and grant support from the NIH’s National Institute of Allergy and Infectious Diseases, National Center for Complementary and Integrative Health and Immune Tolerance Network, Food Allergy Research and Education, and the Wallace Research Foundation. Dr. Shaker has participated in research funded by DBV, is cochair of the AAAAI/ACAAI Joint Task Force on Practice Parameters, is an associate editor at the Annals of Allergy, Asthma, and Immunology, and is an editorial board member of the Journal of Allergy and Clinical Immunology in Practice.
A version of this article first appeared on Medscape.com.
In a large, blinded study of peanut-allergic toddlers published in The Lancet, 71% of treated participants could safely consume 5,000 mg of peanut protein – equivalent to nearly 17 peanuts – after 2½ years on oral immunotherapy. Even after stopping maintenance dosing for the next 6 months, more than 1 in 5 maintained that level of protection, and nearly 3 in 5 still met the 600-mg benchmark (about 2 peanuts) set by the phase 3 PALISADE trial of the FDA-approved peanut-flour product, Palforzia.
About 2% of children in the United States are allergic to peanuts, and most will not outgrow this allergy. In addition, other research suggests that the immune system is more malleable during early childhood.
Consistent with this idea, prior research showed that toddlers can succeed with peanut oral immunotherapy (OIT) – a regimen that builds tolerance through small amounts of the allergen consumed daily for months. However, that trial (DEVIL) was small, was conducted at a single site, and had no placebo group.
In contrast, the Peanut Oral Immunotherapy in Children Trial (IMPACT) enrolled 146 children aged 1-3 years at five academic medical centers in the United States – the first placebo-controlled study of OIT in this younger age group.
“This is a well done study,” Jaclyn Bjelac, MD, associate director of the Food Allergy Center of Excellence at the Cleveland Clinic, told this news organization. “We have seen improved outcomes in OIT, both in our own experience and other published studies, so while this is no surprise, the outcomes and large number of participants contribute to this being a really exciting publication.”
The trial was long and demanding for families. Toddlers who reacted to 500 mg or less of peanut protein in an entry food challenge were randomized in a 2:1 ratio to receive daily peanut flour or oat flour placebo. After initial dose escalation (from 0.1 mg to 6 mg) and biweekly buildup to a 2,000-mg target dose by week 30, participants continued with 20,00-mg daily maintenance dosing through week 134 – at which point they underwent a food challenge. They then went off treatment for 26 weeks and had another food challenge (week 160). In addition, participants came in for skin-prick and blood tests at baseline and at weeks 30, 82, 134, and 160.
In the placebo group, only 23 of 50 participants (46%) completed the study. “If you did 2½ years of this and then bombed the food challenge, you probably can guess that you were not on the real thing. And they were still asked to come back in 6 months and do it again. So, sure enough, a big chunk of those people chose not to continue, and you can’t blame them,” said Lancet co-author Edwin Kim, MD, in an interview. Dr. Kim directs the UNC Food Allergy Initiative at the University of North Carolina School of Medicine, Chapel Hill.
There was attrition in the treatment group as well. Among 96 children initially assigned to this arm, 68 (71%) passed the 5,000-mg peanut challenge at week 134 – but 11 withdrew in the study’s off-treatment phase. “It was a very tough decision. How much do you give toward science?” said Dr. Kim. “When push came to shove, some of the families couldn’t pull the trigger to potentially give up what they worked so hard for.”
In the intention-to-treat analysis, 20 of 96 treated participants (21%) could still tolerate 5,000 mg of peanut protein after going off therapy for 6 months. That translates to a 29% remission rate in the per-protocol subset (n = 70) who completed the study. Forty (57%) of these completers safely consumed at least 1,755 mg of peanut (cumulative dose). By comparison, the PALISADE trial of Palforzia used a 10,430-mg cumulative peanut dose to measure treatment efficacy.
On safety, 98% of treated participants – but also 80% of the placebo group – reported reactions, of which 35 were treated with epinephrine in 21 children receiving peanut OIT.
While some have noted that epinephrine use seemed high, Dr. Kim said, “we’re actually OK with that, because we’d much rather they overtreat and make sure that 1-year-old is safe than take any chances.” Overall, the safety profile looks similar to prior OIT studies of older children. “I think it suggests that, yeah, side effects will happen, [but] they’re all manageable, and people are not anaphylaxing left and right.”
On remission and immunologic parameters, benefits seemed stronger in the youngest subset (12 to 24 months), particularly those with lower peanut-specific IgE at baseline. These trends require further analyses, though, given the limited number of participants under 24 months.
Another noteworthy observation from longitudinal peanut-specific IgE trends in the placebo group: “Avoidance may not be benign,” Dr. Kim said. “If you look at their labs, they don’t stay flat. They actually go up.” The results jibe with the long-held idea of an early window of opportunity while a child’s immune system is maturing. “If you can grab this kid when his IgE is 10, versus next year when it might be 50, maybe you’ll get a different treatment effect,” Dr. Kim said. “We don’t know that for sure, but the placebo labs kind of point toward that.”
Beyond the science, there are practical advantages to starting OIT early. “Trying to convince a 9-year-old who’s been petrified of peanuts for their whole life to start doing this every day is not an easy task,” whereas with a 1- or 2-year-old, “you build it into their routine,” Dr. Kim said.
Plus, some say there’s no need for families to wait for regulatory approval of additional commercial products for very young children. Though some have advocated against the use of “grocery store” products, most peanut OIT research “has used the same 12% light roast defatted peanut flour used in IMPACT,” noted Marcus S. Shaker, MD, professor of pediatrics and of medicine at the Dartmouth Geisel School of Medicine and a physician at the Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire. The commercial product (Palforzia) and grocery-store products “come from the exact same source in the U.S.,” he said in an interview. “Both are an option for parents to consider, but a commercial product is not, nor has [it] ever been, a necessity.”
Dr. Bjelac reports no relevant financial relationships. Dr. Kim reports consultancy with Aimmune Therapeutics, Allako, AllerGenis, Belhaven Pharma, DBV Technologies, Duke Clinical Research Institute, and Nutricia; advisory board membership with ALK, DBV Technologies, Kenota Health, and Ukko; and grant support from the NIH’s National Institute of Allergy and Infectious Diseases, National Center for Complementary and Integrative Health and Immune Tolerance Network, Food Allergy Research and Education, and the Wallace Research Foundation. Dr. Shaker has participated in research funded by DBV, is cochair of the AAAAI/ACAAI Joint Task Force on Practice Parameters, is an associate editor at the Annals of Allergy, Asthma, and Immunology, and is an editorial board member of the Journal of Allergy and Clinical Immunology in Practice.
A version of this article first appeared on Medscape.com.
In a large, blinded study of peanut-allergic toddlers published in The Lancet, 71% of treated participants could safely consume 5,000 mg of peanut protein – equivalent to nearly 17 peanuts – after 2½ years on oral immunotherapy. Even after stopping maintenance dosing for the next 6 months, more than 1 in 5 maintained that level of protection, and nearly 3 in 5 still met the 600-mg benchmark (about 2 peanuts) set by the phase 3 PALISADE trial of the FDA-approved peanut-flour product, Palforzia.
About 2% of children in the United States are allergic to peanuts, and most will not outgrow this allergy. In addition, other research suggests that the immune system is more malleable during early childhood.
Consistent with this idea, prior research showed that toddlers can succeed with peanut oral immunotherapy (OIT) – a regimen that builds tolerance through small amounts of the allergen consumed daily for months. However, that trial (DEVIL) was small, was conducted at a single site, and had no placebo group.
In contrast, the Peanut Oral Immunotherapy in Children Trial (IMPACT) enrolled 146 children aged 1-3 years at five academic medical centers in the United States – the first placebo-controlled study of OIT in this younger age group.
“This is a well done study,” Jaclyn Bjelac, MD, associate director of the Food Allergy Center of Excellence at the Cleveland Clinic, told this news organization. “We have seen improved outcomes in OIT, both in our own experience and other published studies, so while this is no surprise, the outcomes and large number of participants contribute to this being a really exciting publication.”
The trial was long and demanding for families. Toddlers who reacted to 500 mg or less of peanut protein in an entry food challenge were randomized in a 2:1 ratio to receive daily peanut flour or oat flour placebo. After initial dose escalation (from 0.1 mg to 6 mg) and biweekly buildup to a 2,000-mg target dose by week 30, participants continued with 20,00-mg daily maintenance dosing through week 134 – at which point they underwent a food challenge. They then went off treatment for 26 weeks and had another food challenge (week 160). In addition, participants came in for skin-prick and blood tests at baseline and at weeks 30, 82, 134, and 160.
In the placebo group, only 23 of 50 participants (46%) completed the study. “If you did 2½ years of this and then bombed the food challenge, you probably can guess that you were not on the real thing. And they were still asked to come back in 6 months and do it again. So, sure enough, a big chunk of those people chose not to continue, and you can’t blame them,” said Lancet co-author Edwin Kim, MD, in an interview. Dr. Kim directs the UNC Food Allergy Initiative at the University of North Carolina School of Medicine, Chapel Hill.
There was attrition in the treatment group as well. Among 96 children initially assigned to this arm, 68 (71%) passed the 5,000-mg peanut challenge at week 134 – but 11 withdrew in the study’s off-treatment phase. “It was a very tough decision. How much do you give toward science?” said Dr. Kim. “When push came to shove, some of the families couldn’t pull the trigger to potentially give up what they worked so hard for.”
In the intention-to-treat analysis, 20 of 96 treated participants (21%) could still tolerate 5,000 mg of peanut protein after going off therapy for 6 months. That translates to a 29% remission rate in the per-protocol subset (n = 70) who completed the study. Forty (57%) of these completers safely consumed at least 1,755 mg of peanut (cumulative dose). By comparison, the PALISADE trial of Palforzia used a 10,430-mg cumulative peanut dose to measure treatment efficacy.
On safety, 98% of treated participants – but also 80% of the placebo group – reported reactions, of which 35 were treated with epinephrine in 21 children receiving peanut OIT.
While some have noted that epinephrine use seemed high, Dr. Kim said, “we’re actually OK with that, because we’d much rather they overtreat and make sure that 1-year-old is safe than take any chances.” Overall, the safety profile looks similar to prior OIT studies of older children. “I think it suggests that, yeah, side effects will happen, [but] they’re all manageable, and people are not anaphylaxing left and right.”
On remission and immunologic parameters, benefits seemed stronger in the youngest subset (12 to 24 months), particularly those with lower peanut-specific IgE at baseline. These trends require further analyses, though, given the limited number of participants under 24 months.
Another noteworthy observation from longitudinal peanut-specific IgE trends in the placebo group: “Avoidance may not be benign,” Dr. Kim said. “If you look at their labs, they don’t stay flat. They actually go up.” The results jibe with the long-held idea of an early window of opportunity while a child’s immune system is maturing. “If you can grab this kid when his IgE is 10, versus next year when it might be 50, maybe you’ll get a different treatment effect,” Dr. Kim said. “We don’t know that for sure, but the placebo labs kind of point toward that.”
Beyond the science, there are practical advantages to starting OIT early. “Trying to convince a 9-year-old who’s been petrified of peanuts for their whole life to start doing this every day is not an easy task,” whereas with a 1- or 2-year-old, “you build it into their routine,” Dr. Kim said.
Plus, some say there’s no need for families to wait for regulatory approval of additional commercial products for very young children. Though some have advocated against the use of “grocery store” products, most peanut OIT research “has used the same 12% light roast defatted peanut flour used in IMPACT,” noted Marcus S. Shaker, MD, professor of pediatrics and of medicine at the Dartmouth Geisel School of Medicine and a physician at the Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire. The commercial product (Palforzia) and grocery-store products “come from the exact same source in the U.S.,” he said in an interview. “Both are an option for parents to consider, but a commercial product is not, nor has [it] ever been, a necessity.”
Dr. Bjelac reports no relevant financial relationships. Dr. Kim reports consultancy with Aimmune Therapeutics, Allako, AllerGenis, Belhaven Pharma, DBV Technologies, Duke Clinical Research Institute, and Nutricia; advisory board membership with ALK, DBV Technologies, Kenota Health, and Ukko; and grant support from the NIH’s National Institute of Allergy and Infectious Diseases, National Center for Complementary and Integrative Health and Immune Tolerance Network, Food Allergy Research and Education, and the Wallace Research Foundation. Dr. Shaker has participated in research funded by DBV, is cochair of the AAAAI/ACAAI Joint Task Force on Practice Parameters, is an associate editor at the Annals of Allergy, Asthma, and Immunology, and is an editorial board member of the Journal of Allergy and Clinical Immunology in Practice.
A version of this article first appeared on Medscape.com.
Rituximab and COVID-19 vaccines: Studies begin to answer key questions
Rituximab has presented something of a conundrum for patients taking the monoclonal antibody during the COVID-19 pandemic.
Used to manage a variety of autoimmune diseases and cancers, rituximab acts against CD20 proteins expressed on the surface of B cells, causing B-cell depletion. However, it is this B-cell depletion that may put these patients at greater risk of COVID-19 development, progression to more severe disease, and in-hospital mortality. Evidence for this appears to be mixed, with studies showing both that patients using rituximab to manage various diseases are and are not at increased risk for SARS-CoV-2 infection, COVID-19 progression, and mortality.
As COVID-19 vaccine rollouts take place across the world, more questions have been raised about the relationship between B-cell depletion from anti-CD20 therapies and COVID-19 vaccines. Do rituximab and other anti-CD20 therapies affect a patient’s response to COVID-19 vaccines? If this is the case, does the timing of anti-CD20 treatment matter to maximize B-cell levels and improve the vaccine’s effectiveness? And how do COVID-19 vaccine booster doses factor into the equation?
Humoral and cell-mediated responses following COVID-19 vaccination
First, the bad news: The vaccine is unquestionably safe to administer in patients taking rituximab, but one thing that has been well established is that antibody response to COVID-19 vaccination in these individuals does is reduced. This isn’t entirely unprecedented, as previous studies have shown a weakened immune response to pneumococcal polysaccharide and keyhole limpet hemocyanin vaccines among patients taking rituximab.
“Compromised immunogenicity to the SARS-CoV-2 vaccines has been demonstrated in rituximab-treated patients, which is of particular concern given the observation that B-cell–depleting therapies may be associated with worse COVID outcomes,” Robert F. Spiera, MD, director of the Scleroderma, Vasculitis, and Myositis Center at the Hospital for Special Surgery in New York, said in an interview.
For example, in a recent study from the Medical University of Vienna, 29 (39%) of 74 patients receiving rituximab (43% as monotherapy, 57% with conventional-synthetic disease-modifying antirheumatic drugs) who were vaccinated with either the Comirnaty (Pfizer-BioNTech) or Spikevax (Moderna) COVID-19 vaccine achieved seroconversion, compared with 100% of patients in a healthy control group, and all but 1 patient without detectable CD19+ peripheral B cells did not develop anti–SARS-CoV-2 receptor-binding domain antibodies.
“There is an increasing number of studies in this field, and they confirm that patients treated with rituximab and other anti-CD20 agents have severely reduced serological responses to COVID-19 vaccines,” Ingrid Jyssum, MD, of the division of rheumatology and research at Diakonhjemmet Hospital in Oslo, said in an interview.
One silver lining is that patients treated with anti-CD20 therapies appear to have a cell-mediated response following vaccination even if they don’t develop SARS-CoV-2 antibodies. “Studies that also investigate T-cell responses are starting to emerge, and so far, they show that, even if the patients do not have antibodies, they may have T-cell responses,” Dr. Jyssum said.
One study of 24 patients with autoimmune diseases taking rituximab that evaluated humoral and T-cell responses following vaccination with the Comirnaty vaccine found that none had a humoral response to the vaccine, but the T-cell response from that group did not significantly differ from 35 patients receiving other immunosuppressants and 26 patients in a healthy control group. In another study of rituximab- or ocrelizumab-treated patients who received mRNA-based COVID-19 vaccines, 69.4% developed SARS-CoV-2–specific antibodies, compared with a control group, but 96.2% of patients taking ocrelizumab and 81.8% of patients taking rituximab mounted a spike-specific CD8+ T-cell response, compared with 66.7% in the control group, and there were comparable rates (85%-90%) of spike-specific CD4+ T cells in all groups. In the study from the Medical University of Vienna, T-cell response was detected in rituximab-treated patients who both did and did not mount an antibody response.
The clinical relevance of how a blunted humoral immune response but a respectable T-cell response to COVID-19 vaccines affects patients treated with anti-CD20 therapies isn’t currently known, Dr. Jyssum said.
While these data are reassuring, they’re also incomplete, Dr. Spiera noted. “The ultimate outcome of relevance to assess vaccine efficacy is protection from COVID and from severe outcomes of COVID infection (i.e., hospitalization, mechanical ventilation, death). That data will require assessment of very large numbers of rituximab-treated vaccinated patients to be compared with rituximab-treated unvaccinated patients, and is unlikely to be forthcoming in the very near future.
“In the meantime, however, achieving serologic positivity, meaning having evidence of serologic as well as cellular immunity following vaccination, is a desired outcome, and likely implies more robust immunity.”
Does treatment timing impact COVID-19 vaccine response?
Given enough time, B-cell reconstitution will occur in patients taking rituximab. With that in mind, is it beneficial to wait a certain amount of time after a patient has stopped rituximab therapy or time since their last dose before giving them a COVID-19 vaccine? In their guidance on COVID-19 vaccines for patients with rheumatic and musculoskeletal diseases, the American College of Rheumatology said there is moderate evidence to consider “optimal timing of dosing and vaccination with the rheumatology provider before proceeding.”
“Guidelines and preliminary studies of serologic response to COVID vaccine in rituximab-treated patients have suggested that longer time from last rituximab exposure is associated with a greater likelihood of a serologic response,” Dr. Spiera said.
In a brief report published in Arthritis & Rheumatology, Dr. Spiera and colleagues performed a retrospective chart review of 56 patients with varying levels of last exposure to rituximab who received a COVID-19 vaccine. Their results showed that, when patients were vaccinated 6-12 months after the last rituximab dose, 55% were seronegative, and when this was more than 12 months, only 13% were seronegative, compared with seronegativity in 86% who were vaccinated less than 6 months after their last rituximab dose.
The RituxiVac trial, conducted by researchers in Switzerland, also examined vaccine responses of 96 rituximab-treated patients who received Comirnaty or Spikevax; results recently published in The Lancet Rheumatology showed findings similar to other studies, with reduced humoral and cell-mediated responses. In the RituxiVac trial, the median time to last anti-CD20 treatment was 1.07 years.
“The typical interval between rituximab doses [for treatment of rheumatoid arthritis, as well as for remission maintenance in antineutrophil cytoplasmic antibody–associated vasculitis] is typically 6 months, and this has become widely used as the interval from last rituximab to time of COVID vaccination, with a recommendation to wait 4 weeks (if possible) from time of vaccination until the next rituximab administration,” Dr. Spiera explained. However, this window seems to vary depending on the study.
Recent research published in Arthritis & Rheumatology indicates B-cell levels could be a relevant indicator for humoral and cell-mediated response in patients with rheumatic diseases treated with rituximab, with a level of 10 B cells/mcL (0.4% of lymphocytes) identified as one potential marker for likely seroconversion following COVID-19 vaccination.
“In some smaller case series, it has been further recognized that rituximab-treated patients who were beginning to reconstitute peripheral B cells were most likely to respond serologically. Our present study confirmed those findings, demonstrating that the presence of detectable B cells was strongly associated with vaccine responsiveness, and affords complementary information to time from last [rituximab dose] in informing the likelihood of a vaccine response,” Dr. Spiera said.
However, the literature is limited in this area, and an exact cutoff for B-cell counts in these patients isn’t currently known, Dr. Jyssum said. A better metric is time away from anti-CD20 therapies, with CD19 cell count being highly correlated with last infusion.
Dr. Spiera agreed that there is no consistent B-cell percentage that works as a cutoff. “In our study, we looked at it as a binary variable, although we did find that a higher percentage of B cells in the peripheral lymphocyte population was associated with a higher likelihood of seroconversion. We did not, however, identify a ‘threshold’ for vaccine serologic responsiveness.”
Should clinicians measure antibodies?
The Food and Drug Administration and the Centers for Disease Control and Prevention have recommended that health care providers and the public not use COVID-19 antibody tests as a way to gauge immunity after exposure to SARS-CoV-2 and after receiving a COVID-19 vaccination. The ACR’s guidance on COVID-19 vaccination for patients with rheumatic and musculoskeletal diseases strongly recommends against ordering antibody tests for patients with autoimmune inflammatory rheumatic diseases as a way to measure immunity.
“Generally, such measurements are not recommended as the clinical correlate of various antibody levels are not known,” Dr. Jyssum said. “With regular infusions of rituximab or other anti-CD20 agents, one cannot expect that these patients will develop significant levels of antibodies.”
However, she said there might be situations where it’s useful to know whether a patient has developed antibodies at all. “Assessing the significance of specific antibody levels is difficult, and the subject of scientific studies. Patients lacking a humoral vaccine response are left to rely on their T-cell responses and on infectious control measures to prevent disease.”
Dr. Spiera said he disagreed with guidelines recommending against checking antibody levels after vaccination, “particularly in patients treated with immunosuppressive medications that might be expected to blunt their serologic response to the vaccines.
“Although we cannot be sure what level of measurable antibodies offer what level of protection, most clinicians would agree that patients who demonstrate no detectable antibodies (which is a common finding in rituximab-treated patients) should be considered at higher risk,” he said. “Indeed, recommendations regarding booster vaccine administration in general was initially based on the observation of declining antibody levels with longer time from vaccination.”
Do COVID-19 vaccine boosters help patients on anti-CD20 therapy?
As of January 2022, the FDA and CDC have recommended a third primary series shot of COVID-19 vaccines for some moderately to severely immunocompromised patients as young as 5 years old (for Comirnaty vaccine) or a booster shot of either Comirnaty or Spikevax for everyone aged 12 years and older, including immunocompromised people, while the ACR goes into more detail and recommends clinicians time a patient’s booster shot with temporary treatment interruption.
In The Lancet Rheumatology, Dr. Jyssum and colleagues recently published results from the prospective Nor-vaC study examining the humoral and cell-mediated immune responses of 87 patients with RA being treated with rituximab who received the Comirnaty, Spikevax, or Vaxzevria (AstraZeneca) COVID-19 vaccines; of these, 49 patients received a booster dose at a median of 70 days after completing their primary series. The results showed 19 patients (28.1%) had a serologic response after their primary series, while 8 of 49 patients (16.3%) who received their booster dose had a serologic response.
All patients who received a third dose in the study had a T-cell response, Dr. Jyssum said. “This is reassuring for patients and clinicians. T cells have been found to be important in countering COVID-19 disease, but whether we can rely on the T-cell response alone in the absence of antibodies to protect patients from infection or from serious COVID disease is still not determined,” she said.
When asked if she would recommend COVID-19 vaccine booster doses for patients on rituximab, Dr. Jyssum replied: “Absolutely.”
Another study, recently published in Annals of the Rheumatic Diseases, examined heterologous and homologous booster doses for 60 patients receiving rituximab without seroconversion after their COVID-19 vaccine primary series. The results showed no significant difference in new seroconversion at 4 weeks based on whether the patient received a vector or mRNA vaccine (22% vs. 32%), but all patients who received a booster dose with a vector vaccine had specific T-cell responses, compared with 81% of patients who received an mRNA vaccine booster. There was a new humoral and/or cellular response in 9 of 11 patients (82%), and most patients with peripheral B cells (12 of 18 patients; 67%) achieved seroconversion.
“Our data show that a cellular and/or humoral immune response can be achieved on a third COVID-19 vaccination in most of the patients who initially developed neither a humoral nor a cellular immune response,” the researchers concluded. “The efficacy data together with the safety data seen in our trial provide a favorable risk/benefit ratio and support the implementation of a third vaccination for nonseroconverted high-risk autoimmune disease patients treated with B-cell–depleting agents.”
Dr. Spiera said booster doses are an important part of the equation, and “it is important to consider factors that would be associated with a greater likelihood of achieving a serologic response, particularly in those patients who did not demonstrate a serologic response to the initial vaccines series.
“Preliminary data shows that the beginnings of B-cell reconstitution is also associated with a positive serologic response following a booster of the COVID-19 vaccine,” he said.
The authors of the cited studies reported numerous relevant financial disclosures. Dr. Spiera and Dr. Jyssum reported no relevant financial disclosures.
Rituximab has presented something of a conundrum for patients taking the monoclonal antibody during the COVID-19 pandemic.
Used to manage a variety of autoimmune diseases and cancers, rituximab acts against CD20 proteins expressed on the surface of B cells, causing B-cell depletion. However, it is this B-cell depletion that may put these patients at greater risk of COVID-19 development, progression to more severe disease, and in-hospital mortality. Evidence for this appears to be mixed, with studies showing both that patients using rituximab to manage various diseases are and are not at increased risk for SARS-CoV-2 infection, COVID-19 progression, and mortality.
As COVID-19 vaccine rollouts take place across the world, more questions have been raised about the relationship between B-cell depletion from anti-CD20 therapies and COVID-19 vaccines. Do rituximab and other anti-CD20 therapies affect a patient’s response to COVID-19 vaccines? If this is the case, does the timing of anti-CD20 treatment matter to maximize B-cell levels and improve the vaccine’s effectiveness? And how do COVID-19 vaccine booster doses factor into the equation?
Humoral and cell-mediated responses following COVID-19 vaccination
First, the bad news: The vaccine is unquestionably safe to administer in patients taking rituximab, but one thing that has been well established is that antibody response to COVID-19 vaccination in these individuals does is reduced. This isn’t entirely unprecedented, as previous studies have shown a weakened immune response to pneumococcal polysaccharide and keyhole limpet hemocyanin vaccines among patients taking rituximab.
“Compromised immunogenicity to the SARS-CoV-2 vaccines has been demonstrated in rituximab-treated patients, which is of particular concern given the observation that B-cell–depleting therapies may be associated with worse COVID outcomes,” Robert F. Spiera, MD, director of the Scleroderma, Vasculitis, and Myositis Center at the Hospital for Special Surgery in New York, said in an interview.
For example, in a recent study from the Medical University of Vienna, 29 (39%) of 74 patients receiving rituximab (43% as monotherapy, 57% with conventional-synthetic disease-modifying antirheumatic drugs) who were vaccinated with either the Comirnaty (Pfizer-BioNTech) or Spikevax (Moderna) COVID-19 vaccine achieved seroconversion, compared with 100% of patients in a healthy control group, and all but 1 patient without detectable CD19+ peripheral B cells did not develop anti–SARS-CoV-2 receptor-binding domain antibodies.
“There is an increasing number of studies in this field, and they confirm that patients treated with rituximab and other anti-CD20 agents have severely reduced serological responses to COVID-19 vaccines,” Ingrid Jyssum, MD, of the division of rheumatology and research at Diakonhjemmet Hospital in Oslo, said in an interview.
One silver lining is that patients treated with anti-CD20 therapies appear to have a cell-mediated response following vaccination even if they don’t develop SARS-CoV-2 antibodies. “Studies that also investigate T-cell responses are starting to emerge, and so far, they show that, even if the patients do not have antibodies, they may have T-cell responses,” Dr. Jyssum said.
One study of 24 patients with autoimmune diseases taking rituximab that evaluated humoral and T-cell responses following vaccination with the Comirnaty vaccine found that none had a humoral response to the vaccine, but the T-cell response from that group did not significantly differ from 35 patients receiving other immunosuppressants and 26 patients in a healthy control group. In another study of rituximab- or ocrelizumab-treated patients who received mRNA-based COVID-19 vaccines, 69.4% developed SARS-CoV-2–specific antibodies, compared with a control group, but 96.2% of patients taking ocrelizumab and 81.8% of patients taking rituximab mounted a spike-specific CD8+ T-cell response, compared with 66.7% in the control group, and there were comparable rates (85%-90%) of spike-specific CD4+ T cells in all groups. In the study from the Medical University of Vienna, T-cell response was detected in rituximab-treated patients who both did and did not mount an antibody response.
The clinical relevance of how a blunted humoral immune response but a respectable T-cell response to COVID-19 vaccines affects patients treated with anti-CD20 therapies isn’t currently known, Dr. Jyssum said.
While these data are reassuring, they’re also incomplete, Dr. Spiera noted. “The ultimate outcome of relevance to assess vaccine efficacy is protection from COVID and from severe outcomes of COVID infection (i.e., hospitalization, mechanical ventilation, death). That data will require assessment of very large numbers of rituximab-treated vaccinated patients to be compared with rituximab-treated unvaccinated patients, and is unlikely to be forthcoming in the very near future.
“In the meantime, however, achieving serologic positivity, meaning having evidence of serologic as well as cellular immunity following vaccination, is a desired outcome, and likely implies more robust immunity.”
Does treatment timing impact COVID-19 vaccine response?
Given enough time, B-cell reconstitution will occur in patients taking rituximab. With that in mind, is it beneficial to wait a certain amount of time after a patient has stopped rituximab therapy or time since their last dose before giving them a COVID-19 vaccine? In their guidance on COVID-19 vaccines for patients with rheumatic and musculoskeletal diseases, the American College of Rheumatology said there is moderate evidence to consider “optimal timing of dosing and vaccination with the rheumatology provider before proceeding.”
“Guidelines and preliminary studies of serologic response to COVID vaccine in rituximab-treated patients have suggested that longer time from last rituximab exposure is associated with a greater likelihood of a serologic response,” Dr. Spiera said.
In a brief report published in Arthritis & Rheumatology, Dr. Spiera and colleagues performed a retrospective chart review of 56 patients with varying levels of last exposure to rituximab who received a COVID-19 vaccine. Their results showed that, when patients were vaccinated 6-12 months after the last rituximab dose, 55% were seronegative, and when this was more than 12 months, only 13% were seronegative, compared with seronegativity in 86% who were vaccinated less than 6 months after their last rituximab dose.
The RituxiVac trial, conducted by researchers in Switzerland, also examined vaccine responses of 96 rituximab-treated patients who received Comirnaty or Spikevax; results recently published in The Lancet Rheumatology showed findings similar to other studies, with reduced humoral and cell-mediated responses. In the RituxiVac trial, the median time to last anti-CD20 treatment was 1.07 years.
“The typical interval between rituximab doses [for treatment of rheumatoid arthritis, as well as for remission maintenance in antineutrophil cytoplasmic antibody–associated vasculitis] is typically 6 months, and this has become widely used as the interval from last rituximab to time of COVID vaccination, with a recommendation to wait 4 weeks (if possible) from time of vaccination until the next rituximab administration,” Dr. Spiera explained. However, this window seems to vary depending on the study.
Recent research published in Arthritis & Rheumatology indicates B-cell levels could be a relevant indicator for humoral and cell-mediated response in patients with rheumatic diseases treated with rituximab, with a level of 10 B cells/mcL (0.4% of lymphocytes) identified as one potential marker for likely seroconversion following COVID-19 vaccination.
“In some smaller case series, it has been further recognized that rituximab-treated patients who were beginning to reconstitute peripheral B cells were most likely to respond serologically. Our present study confirmed those findings, demonstrating that the presence of detectable B cells was strongly associated with vaccine responsiveness, and affords complementary information to time from last [rituximab dose] in informing the likelihood of a vaccine response,” Dr. Spiera said.
However, the literature is limited in this area, and an exact cutoff for B-cell counts in these patients isn’t currently known, Dr. Jyssum said. A better metric is time away from anti-CD20 therapies, with CD19 cell count being highly correlated with last infusion.
Dr. Spiera agreed that there is no consistent B-cell percentage that works as a cutoff. “In our study, we looked at it as a binary variable, although we did find that a higher percentage of B cells in the peripheral lymphocyte population was associated with a higher likelihood of seroconversion. We did not, however, identify a ‘threshold’ for vaccine serologic responsiveness.”
Should clinicians measure antibodies?
The Food and Drug Administration and the Centers for Disease Control and Prevention have recommended that health care providers and the public not use COVID-19 antibody tests as a way to gauge immunity after exposure to SARS-CoV-2 and after receiving a COVID-19 vaccination. The ACR’s guidance on COVID-19 vaccination for patients with rheumatic and musculoskeletal diseases strongly recommends against ordering antibody tests for patients with autoimmune inflammatory rheumatic diseases as a way to measure immunity.
“Generally, such measurements are not recommended as the clinical correlate of various antibody levels are not known,” Dr. Jyssum said. “With regular infusions of rituximab or other anti-CD20 agents, one cannot expect that these patients will develop significant levels of antibodies.”
However, she said there might be situations where it’s useful to know whether a patient has developed antibodies at all. “Assessing the significance of specific antibody levels is difficult, and the subject of scientific studies. Patients lacking a humoral vaccine response are left to rely on their T-cell responses and on infectious control measures to prevent disease.”
Dr. Spiera said he disagreed with guidelines recommending against checking antibody levels after vaccination, “particularly in patients treated with immunosuppressive medications that might be expected to blunt their serologic response to the vaccines.
“Although we cannot be sure what level of measurable antibodies offer what level of protection, most clinicians would agree that patients who demonstrate no detectable antibodies (which is a common finding in rituximab-treated patients) should be considered at higher risk,” he said. “Indeed, recommendations regarding booster vaccine administration in general was initially based on the observation of declining antibody levels with longer time from vaccination.”
Do COVID-19 vaccine boosters help patients on anti-CD20 therapy?
As of January 2022, the FDA and CDC have recommended a third primary series shot of COVID-19 vaccines for some moderately to severely immunocompromised patients as young as 5 years old (for Comirnaty vaccine) or a booster shot of either Comirnaty or Spikevax for everyone aged 12 years and older, including immunocompromised people, while the ACR goes into more detail and recommends clinicians time a patient’s booster shot with temporary treatment interruption.
In The Lancet Rheumatology, Dr. Jyssum and colleagues recently published results from the prospective Nor-vaC study examining the humoral and cell-mediated immune responses of 87 patients with RA being treated with rituximab who received the Comirnaty, Spikevax, or Vaxzevria (AstraZeneca) COVID-19 vaccines; of these, 49 patients received a booster dose at a median of 70 days after completing their primary series. The results showed 19 patients (28.1%) had a serologic response after their primary series, while 8 of 49 patients (16.3%) who received their booster dose had a serologic response.
All patients who received a third dose in the study had a T-cell response, Dr. Jyssum said. “This is reassuring for patients and clinicians. T cells have been found to be important in countering COVID-19 disease, but whether we can rely on the T-cell response alone in the absence of antibodies to protect patients from infection or from serious COVID disease is still not determined,” she said.
When asked if she would recommend COVID-19 vaccine booster doses for patients on rituximab, Dr. Jyssum replied: “Absolutely.”
Another study, recently published in Annals of the Rheumatic Diseases, examined heterologous and homologous booster doses for 60 patients receiving rituximab without seroconversion after their COVID-19 vaccine primary series. The results showed no significant difference in new seroconversion at 4 weeks based on whether the patient received a vector or mRNA vaccine (22% vs. 32%), but all patients who received a booster dose with a vector vaccine had specific T-cell responses, compared with 81% of patients who received an mRNA vaccine booster. There was a new humoral and/or cellular response in 9 of 11 patients (82%), and most patients with peripheral B cells (12 of 18 patients; 67%) achieved seroconversion.
“Our data show that a cellular and/or humoral immune response can be achieved on a third COVID-19 vaccination in most of the patients who initially developed neither a humoral nor a cellular immune response,” the researchers concluded. “The efficacy data together with the safety data seen in our trial provide a favorable risk/benefit ratio and support the implementation of a third vaccination for nonseroconverted high-risk autoimmune disease patients treated with B-cell–depleting agents.”
Dr. Spiera said booster doses are an important part of the equation, and “it is important to consider factors that would be associated with a greater likelihood of achieving a serologic response, particularly in those patients who did not demonstrate a serologic response to the initial vaccines series.
“Preliminary data shows that the beginnings of B-cell reconstitution is also associated with a positive serologic response following a booster of the COVID-19 vaccine,” he said.
The authors of the cited studies reported numerous relevant financial disclosures. Dr. Spiera and Dr. Jyssum reported no relevant financial disclosures.
Rituximab has presented something of a conundrum for patients taking the monoclonal antibody during the COVID-19 pandemic.
Used to manage a variety of autoimmune diseases and cancers, rituximab acts against CD20 proteins expressed on the surface of B cells, causing B-cell depletion. However, it is this B-cell depletion that may put these patients at greater risk of COVID-19 development, progression to more severe disease, and in-hospital mortality. Evidence for this appears to be mixed, with studies showing both that patients using rituximab to manage various diseases are and are not at increased risk for SARS-CoV-2 infection, COVID-19 progression, and mortality.
As COVID-19 vaccine rollouts take place across the world, more questions have been raised about the relationship between B-cell depletion from anti-CD20 therapies and COVID-19 vaccines. Do rituximab and other anti-CD20 therapies affect a patient’s response to COVID-19 vaccines? If this is the case, does the timing of anti-CD20 treatment matter to maximize B-cell levels and improve the vaccine’s effectiveness? And how do COVID-19 vaccine booster doses factor into the equation?
Humoral and cell-mediated responses following COVID-19 vaccination
First, the bad news: The vaccine is unquestionably safe to administer in patients taking rituximab, but one thing that has been well established is that antibody response to COVID-19 vaccination in these individuals does is reduced. This isn’t entirely unprecedented, as previous studies have shown a weakened immune response to pneumococcal polysaccharide and keyhole limpet hemocyanin vaccines among patients taking rituximab.
“Compromised immunogenicity to the SARS-CoV-2 vaccines has been demonstrated in rituximab-treated patients, which is of particular concern given the observation that B-cell–depleting therapies may be associated with worse COVID outcomes,” Robert F. Spiera, MD, director of the Scleroderma, Vasculitis, and Myositis Center at the Hospital for Special Surgery in New York, said in an interview.
For example, in a recent study from the Medical University of Vienna, 29 (39%) of 74 patients receiving rituximab (43% as monotherapy, 57% with conventional-synthetic disease-modifying antirheumatic drugs) who were vaccinated with either the Comirnaty (Pfizer-BioNTech) or Spikevax (Moderna) COVID-19 vaccine achieved seroconversion, compared with 100% of patients in a healthy control group, and all but 1 patient without detectable CD19+ peripheral B cells did not develop anti–SARS-CoV-2 receptor-binding domain antibodies.
“There is an increasing number of studies in this field, and they confirm that patients treated with rituximab and other anti-CD20 agents have severely reduced serological responses to COVID-19 vaccines,” Ingrid Jyssum, MD, of the division of rheumatology and research at Diakonhjemmet Hospital in Oslo, said in an interview.
One silver lining is that patients treated with anti-CD20 therapies appear to have a cell-mediated response following vaccination even if they don’t develop SARS-CoV-2 antibodies. “Studies that also investigate T-cell responses are starting to emerge, and so far, they show that, even if the patients do not have antibodies, they may have T-cell responses,” Dr. Jyssum said.
One study of 24 patients with autoimmune diseases taking rituximab that evaluated humoral and T-cell responses following vaccination with the Comirnaty vaccine found that none had a humoral response to the vaccine, but the T-cell response from that group did not significantly differ from 35 patients receiving other immunosuppressants and 26 patients in a healthy control group. In another study of rituximab- or ocrelizumab-treated patients who received mRNA-based COVID-19 vaccines, 69.4% developed SARS-CoV-2–specific antibodies, compared with a control group, but 96.2% of patients taking ocrelizumab and 81.8% of patients taking rituximab mounted a spike-specific CD8+ T-cell response, compared with 66.7% in the control group, and there were comparable rates (85%-90%) of spike-specific CD4+ T cells in all groups. In the study from the Medical University of Vienna, T-cell response was detected in rituximab-treated patients who both did and did not mount an antibody response.
The clinical relevance of how a blunted humoral immune response but a respectable T-cell response to COVID-19 vaccines affects patients treated with anti-CD20 therapies isn’t currently known, Dr. Jyssum said.
While these data are reassuring, they’re also incomplete, Dr. Spiera noted. “The ultimate outcome of relevance to assess vaccine efficacy is protection from COVID and from severe outcomes of COVID infection (i.e., hospitalization, mechanical ventilation, death). That data will require assessment of very large numbers of rituximab-treated vaccinated patients to be compared with rituximab-treated unvaccinated patients, and is unlikely to be forthcoming in the very near future.
“In the meantime, however, achieving serologic positivity, meaning having evidence of serologic as well as cellular immunity following vaccination, is a desired outcome, and likely implies more robust immunity.”
Does treatment timing impact COVID-19 vaccine response?
Given enough time, B-cell reconstitution will occur in patients taking rituximab. With that in mind, is it beneficial to wait a certain amount of time after a patient has stopped rituximab therapy or time since their last dose before giving them a COVID-19 vaccine? In their guidance on COVID-19 vaccines for patients with rheumatic and musculoskeletal diseases, the American College of Rheumatology said there is moderate evidence to consider “optimal timing of dosing and vaccination with the rheumatology provider before proceeding.”
“Guidelines and preliminary studies of serologic response to COVID vaccine in rituximab-treated patients have suggested that longer time from last rituximab exposure is associated with a greater likelihood of a serologic response,” Dr. Spiera said.
In a brief report published in Arthritis & Rheumatology, Dr. Spiera and colleagues performed a retrospective chart review of 56 patients with varying levels of last exposure to rituximab who received a COVID-19 vaccine. Their results showed that, when patients were vaccinated 6-12 months after the last rituximab dose, 55% were seronegative, and when this was more than 12 months, only 13% were seronegative, compared with seronegativity in 86% who were vaccinated less than 6 months after their last rituximab dose.
The RituxiVac trial, conducted by researchers in Switzerland, also examined vaccine responses of 96 rituximab-treated patients who received Comirnaty or Spikevax; results recently published in The Lancet Rheumatology showed findings similar to other studies, with reduced humoral and cell-mediated responses. In the RituxiVac trial, the median time to last anti-CD20 treatment was 1.07 years.
“The typical interval between rituximab doses [for treatment of rheumatoid arthritis, as well as for remission maintenance in antineutrophil cytoplasmic antibody–associated vasculitis] is typically 6 months, and this has become widely used as the interval from last rituximab to time of COVID vaccination, with a recommendation to wait 4 weeks (if possible) from time of vaccination until the next rituximab administration,” Dr. Spiera explained. However, this window seems to vary depending on the study.
Recent research published in Arthritis & Rheumatology indicates B-cell levels could be a relevant indicator for humoral and cell-mediated response in patients with rheumatic diseases treated with rituximab, with a level of 10 B cells/mcL (0.4% of lymphocytes) identified as one potential marker for likely seroconversion following COVID-19 vaccination.
“In some smaller case series, it has been further recognized that rituximab-treated patients who were beginning to reconstitute peripheral B cells were most likely to respond serologically. Our present study confirmed those findings, demonstrating that the presence of detectable B cells was strongly associated with vaccine responsiveness, and affords complementary information to time from last [rituximab dose] in informing the likelihood of a vaccine response,” Dr. Spiera said.
However, the literature is limited in this area, and an exact cutoff for B-cell counts in these patients isn’t currently known, Dr. Jyssum said. A better metric is time away from anti-CD20 therapies, with CD19 cell count being highly correlated with last infusion.
Dr. Spiera agreed that there is no consistent B-cell percentage that works as a cutoff. “In our study, we looked at it as a binary variable, although we did find that a higher percentage of B cells in the peripheral lymphocyte population was associated with a higher likelihood of seroconversion. We did not, however, identify a ‘threshold’ for vaccine serologic responsiveness.”
Should clinicians measure antibodies?
The Food and Drug Administration and the Centers for Disease Control and Prevention have recommended that health care providers and the public not use COVID-19 antibody tests as a way to gauge immunity after exposure to SARS-CoV-2 and after receiving a COVID-19 vaccination. The ACR’s guidance on COVID-19 vaccination for patients with rheumatic and musculoskeletal diseases strongly recommends against ordering antibody tests for patients with autoimmune inflammatory rheumatic diseases as a way to measure immunity.
“Generally, such measurements are not recommended as the clinical correlate of various antibody levels are not known,” Dr. Jyssum said. “With regular infusions of rituximab or other anti-CD20 agents, one cannot expect that these patients will develop significant levels of antibodies.”
However, she said there might be situations where it’s useful to know whether a patient has developed antibodies at all. “Assessing the significance of specific antibody levels is difficult, and the subject of scientific studies. Patients lacking a humoral vaccine response are left to rely on their T-cell responses and on infectious control measures to prevent disease.”
Dr. Spiera said he disagreed with guidelines recommending against checking antibody levels after vaccination, “particularly in patients treated with immunosuppressive medications that might be expected to blunt their serologic response to the vaccines.
“Although we cannot be sure what level of measurable antibodies offer what level of protection, most clinicians would agree that patients who demonstrate no detectable antibodies (which is a common finding in rituximab-treated patients) should be considered at higher risk,” he said. “Indeed, recommendations regarding booster vaccine administration in general was initially based on the observation of declining antibody levels with longer time from vaccination.”
Do COVID-19 vaccine boosters help patients on anti-CD20 therapy?
As of January 2022, the FDA and CDC have recommended a third primary series shot of COVID-19 vaccines for some moderately to severely immunocompromised patients as young as 5 years old (for Comirnaty vaccine) or a booster shot of either Comirnaty or Spikevax for everyone aged 12 years and older, including immunocompromised people, while the ACR goes into more detail and recommends clinicians time a patient’s booster shot with temporary treatment interruption.
In The Lancet Rheumatology, Dr. Jyssum and colleagues recently published results from the prospective Nor-vaC study examining the humoral and cell-mediated immune responses of 87 patients with RA being treated with rituximab who received the Comirnaty, Spikevax, or Vaxzevria (AstraZeneca) COVID-19 vaccines; of these, 49 patients received a booster dose at a median of 70 days after completing their primary series. The results showed 19 patients (28.1%) had a serologic response after their primary series, while 8 of 49 patients (16.3%) who received their booster dose had a serologic response.
All patients who received a third dose in the study had a T-cell response, Dr. Jyssum said. “This is reassuring for patients and clinicians. T cells have been found to be important in countering COVID-19 disease, but whether we can rely on the T-cell response alone in the absence of antibodies to protect patients from infection or from serious COVID disease is still not determined,” she said.
When asked if she would recommend COVID-19 vaccine booster doses for patients on rituximab, Dr. Jyssum replied: “Absolutely.”
Another study, recently published in Annals of the Rheumatic Diseases, examined heterologous and homologous booster doses for 60 patients receiving rituximab without seroconversion after their COVID-19 vaccine primary series. The results showed no significant difference in new seroconversion at 4 weeks based on whether the patient received a vector or mRNA vaccine (22% vs. 32%), but all patients who received a booster dose with a vector vaccine had specific T-cell responses, compared with 81% of patients who received an mRNA vaccine booster. There was a new humoral and/or cellular response in 9 of 11 patients (82%), and most patients with peripheral B cells (12 of 18 patients; 67%) achieved seroconversion.
“Our data show that a cellular and/or humoral immune response can be achieved on a third COVID-19 vaccination in most of the patients who initially developed neither a humoral nor a cellular immune response,” the researchers concluded. “The efficacy data together with the safety data seen in our trial provide a favorable risk/benefit ratio and support the implementation of a third vaccination for nonseroconverted high-risk autoimmune disease patients treated with B-cell–depleting agents.”
Dr. Spiera said booster doses are an important part of the equation, and “it is important to consider factors that would be associated with a greater likelihood of achieving a serologic response, particularly in those patients who did not demonstrate a serologic response to the initial vaccines series.
“Preliminary data shows that the beginnings of B-cell reconstitution is also associated with a positive serologic response following a booster of the COVID-19 vaccine,” he said.
The authors of the cited studies reported numerous relevant financial disclosures. Dr. Spiera and Dr. Jyssum reported no relevant financial disclosures.
Antimicrobial resistance linked to 1.2 million global deaths in 2019
More than HIV, more than malaria.
In terms of preventable deaths, 1.27 million people could have been saved if drug-resistant infections were replaced with infections susceptible to current antibiotics. Furthermore, 4.95 million fewer people would have died if drug-resistant infections were replaced by no infections, researchers estimated.
Although the COVID-19 pandemic took some focus off the AMR burden worldwide over the past 2 years, the urgency to address risk to public health did not ebb. In fact, based on the findings, the researchers noted that AMR is now a leading cause of death worldwide.
“If left unchecked, the spread of AMR could make many bacterial pathogens much more lethal in the future than they are today,” the researchers noted in the study, published online Jan. 20, 2022, in The Lancet.
“These findings are a warning signal that antibiotic resistance is placing pressure on health care systems and leading to significant health loss,” study author Kevin Ikuta, MD, MPH, told this news organization.
“We need to continue to adhere to and support infection prevention and control programs, be thoughtful about our antibiotic use, and advocate for increased funding to vaccine discovery and the antibiotic development pipeline,” added Dr. Ikuta, health sciences assistant clinical professor of medicine at the University of California, Los Angeles.
Although many investigators have studied AMR, this study is the largest in scope, covering 204 countries and territories and incorporating data on a comprehensive range of pathogens and pathogen-drug combinations.
Dr. Ikuta, lead author Christopher J.L. Murray, DPhil, and colleagues estimated the global burden of AMR using the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019. They specifically looked at rates of death directly attributed to and separately those associated with resistance.
Regional differences
Broken down by 21 regions, Australasia had 6.5 deaths per 100,000 people attributable to AMR, the lowest rate reported. This region also had 28 deaths per 100,000 associated with AMR.
Researchers found the highest rates in western sub-Saharan Africa. Deaths attributable to AMR were 27.3 per 100,000 and associated death rate was 114.8 per 100,000.
Lower- and middle-income regions had the highest AMR death rates, although resistance remains a high-priority issue for high-income countries as well.
“It’s important to take a global perspective on resistant infections because we can learn about regions and countries that are experiencing the greatest burden, information that was previously unknown,” Dr. Ikuta said. “With these estimates policy makers can prioritize regions that are hotspots and would most benefit from additional interventions.”
Furthermore, the study emphasized the global nature of AMR. “We’ve seen over the last 2 years with COVID-19 that this sort of problem doesn’t respect country borders, and high rates of resistance in one location can spread across a region or spread globally pretty quickly,” Dr. Ikuta said.
Leading resistant infections
Lower respiratory and thorax infections, bloodstream infections, and intra-abdominal infections together accounted for almost 79% of such deaths linked to AMR.
The six leading pathogens are likely household names among infectious disease specialists. The researchers found Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa, each responsible for more than 250,000 AMR-associated deaths.
The study also revealed that resistance to several first-line antibiotic agents often used empirically to treat infections accounted for more than 70% of the AMR-attributable deaths. These included fluoroquinolones and beta-lactam antibiotics such as carbapenems, cephalosporins, and penicillins.
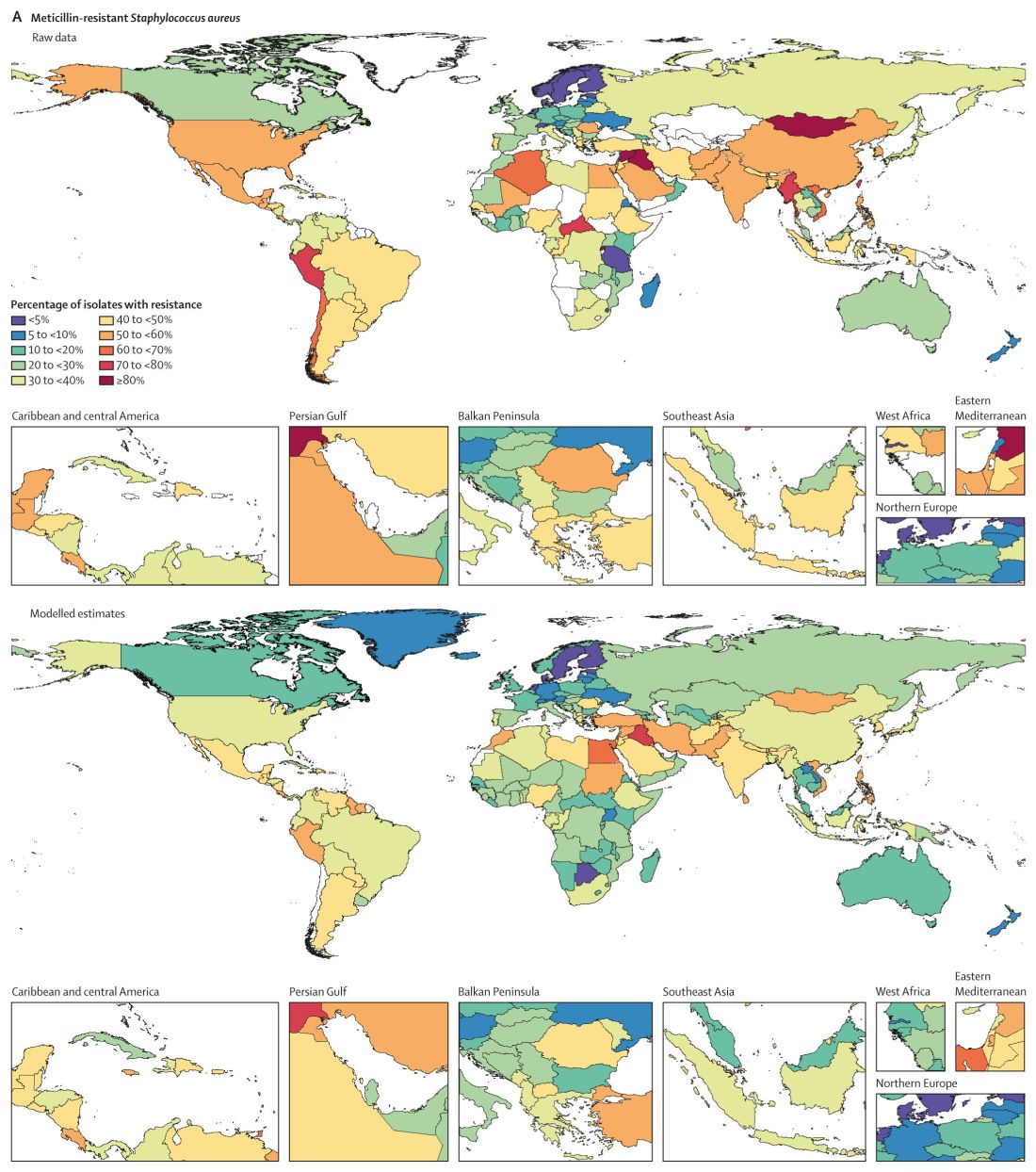
Consistent with previous studies, MRSA stood out as a major cause of mortality. Of 88 different pathogen-drug combinations evaluated, MRSA was responsible for the most mortality: more than 100,000 deaths and 3·5 million disability-adjusted life-years.
The current study findings on MRSA “being a particularly nasty culprit” in AMR infections validates previous work that reported similar results, Vance Fowler, MD, told this news organization when asked to comment on the research. “That is reassuring.”
Potential solutions offered
Dr. Murray and colleagues outlined five strategies to address the challenge of bacterial AMR:
- Infection prevention and control remain paramount in minimizing infections in general and AMR infections in particular.
- More vaccines are needed to reduce the need for antibiotics. “Vaccines are available for only one of the six leading pathogens (S. pneumoniae), although new vaccine programs are underway for S. aureus, E. coli, and others,” the researchers wrote.
- Reduce antibiotic use unrelated to treatment of human disease.
- Avoid using antibiotics for viral infections and other unnecessary indications.
- Invest in new antibiotic development and ensure access to second-line agents in areas without widespread access.
“Identifying strategies that can work to reduce the burden of bacterial AMR – either across a wide range of settings or those that are specifically tailored to the resources available and leading pathogen-drug combinations in a particular setting – is an urgent priority,” the researchers noted.
Admirable AMR research
The results of the study are “startling, but not surprising,” said Dr. Fowler, professor of medicine at Duke University, Durham, N.C.
The authors did a “nice job” of addressing both deaths attributable and associated with AMR, Dr. Fowler added. “Those two categories unlock applications, not just in terms of how you interpret it but also what you do about it.”
The deaths attributable to AMR show that there is more work to be done regarding infection control and prevention, Dr. Fowler said, including in areas of the world like lower- and middle-income countries where infection resistance is most pronounced.
The deaths associated with AMR can be more challenging to calculate – people with infections can die for multiple reasons. However, Dr. Fowler applauded the researchers for doing “as good a job as you can” in estimating the extent of associated mortality.
‘The overlooked pandemic of antimicrobial resistance’
In an accompanying editorial in The Lancet, Ramanan Laxminarayan, PhD, MPH, wrote: “As COVID-19 rages on, the pandemic of antimicrobial resistance continues in the shadows. The toll taken by AMR on patients and their families is largely invisible but is reflected in prolonged bacterial infections that extend hospital stays and cause needless deaths.”
Dr. Laxminarayan pointed out an irony with AMR in different regions. Some of the AMR burden in sub-Saharan Africa is “probably due to inadequate access to antibiotics and high infection levels, albeit at low levels of resistance, whereas in south Asia and Latin America, it is because of high resistance even with good access to antibiotics.”
More funding to address AMR is needed, Dr. Laxminarayan noted. “Even the lower end of 911,000 deaths estimated by Murray and colleagues is higher than the number of deaths from HIV, which attracts close to U.S. $50 billion each year. However, global spending on addressing AMR is probably much lower than that.” Dr. Laxminarayan is an economist and epidemiologist affiliated with the Center for Disease Dynamics, Economics & Policy in Washington, D.C., and the Global Antibiotic Research and Development Partnership in Geneva.
An overlap with COVID-19
The Lancet report is likely “to bring more attention to AMR, especially since so many people have been distracted by COVID, and rightly so,” Dr. Fowler predicted. “The world has had its hands full with COVID.”
The two infections interact in direct ways, Dr. Fowler added. For example, some people hospitalized for COVID-19 for an extended time could develop progressively drug-resistant bacteria – leading to a superinfection.
The overlap could be illustrated by a Venn diagram, he said. A yellow circle could illustrate people with COVID-19 who are asymptomatic or who remain outpatients. Next to that would be a blue circle showing people who develop AMR infections. Where the two circles overlap would be green for those hospitalized who – because of receiving steroids, being on a ventilator, or getting a central line – develop a superinfection.
Official guidance continues
The study comes in the context of recent guidance and federal action on AMR. For example, the Infectious Diseases Society of America released new guidelines for AMR in November 2021 as part of ongoing advice on prevention and treatment of this “ongoing crisis.”
This most recent IDSA guidance addresses three pathogens in particular: AmpC beta-lactamase–producing Enterobacterales, carbapenem-resistant A. baumannii, and Stenotrophomonas maltophilia.
Also in November, the World Health Organization released an updated fact sheet on antimicrobial resistance. The WHO declared AMR one of the world’s top 10 global public health threats. The agency emphasized that misuse and overuse of antimicrobials are the main drivers in the development of drug-resistant pathogens. The WHO also pointed out that lack of clean water and sanitation in many areas of the world contribute to spread of microbes, including those resistant to current treatment options.
In September 2021, the Biden administration acknowledged the threat of AMR with allocation of more than $2 billion of the American Rescue Plan money for prevention and treatment of these infections.
Asked if there are any reasons for hope or optimism at this point, Dr. Ikuta said: “Definitely. We know what needs to be done to combat the spread of resistance. COVID-19 has demonstrated the importance of global commitment to infection control measures, such as hand washing and surveillance, and rapid investments in treatments, which can all be applied to antimicrobial resistance.”
The Bill & Melinda Gates Foundation, the Wellcome Trust, and the U.K. Department of Health and Social Care using U.K. aid funding managed by the Fleming Fund and other organizations provided funding for the study. Dr. Ikuta and Dr. Laxminarayan have disclosed no relevant financial relationships. Dr. Fowler reported receiving grants or honoraria, as well as serving as a consultant, for numerous sources. He also reported a patent pending in sepsis diagnostics and serving as chair of the V710 Scientific Advisory Committee (Merck).
A version of this article first appeared on Medscape.com.
More than HIV, more than malaria.
In terms of preventable deaths, 1.27 million people could have been saved if drug-resistant infections were replaced with infections susceptible to current antibiotics. Furthermore, 4.95 million fewer people would have died if drug-resistant infections were replaced by no infections, researchers estimated.
Although the COVID-19 pandemic took some focus off the AMR burden worldwide over the past 2 years, the urgency to address risk to public health did not ebb. In fact, based on the findings, the researchers noted that AMR is now a leading cause of death worldwide.
“If left unchecked, the spread of AMR could make many bacterial pathogens much more lethal in the future than they are today,” the researchers noted in the study, published online Jan. 20, 2022, in The Lancet.
“These findings are a warning signal that antibiotic resistance is placing pressure on health care systems and leading to significant health loss,” study author Kevin Ikuta, MD, MPH, told this news organization.
“We need to continue to adhere to and support infection prevention and control programs, be thoughtful about our antibiotic use, and advocate for increased funding to vaccine discovery and the antibiotic development pipeline,” added Dr. Ikuta, health sciences assistant clinical professor of medicine at the University of California, Los Angeles.
Although many investigators have studied AMR, this study is the largest in scope, covering 204 countries and territories and incorporating data on a comprehensive range of pathogens and pathogen-drug combinations.
Dr. Ikuta, lead author Christopher J.L. Murray, DPhil, and colleagues estimated the global burden of AMR using the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019. They specifically looked at rates of death directly attributed to and separately those associated with resistance.
Regional differences
Broken down by 21 regions, Australasia had 6.5 deaths per 100,000 people attributable to AMR, the lowest rate reported. This region also had 28 deaths per 100,000 associated with AMR.
Researchers found the highest rates in western sub-Saharan Africa. Deaths attributable to AMR were 27.3 per 100,000 and associated death rate was 114.8 per 100,000.
Lower- and middle-income regions had the highest AMR death rates, although resistance remains a high-priority issue for high-income countries as well.
“It’s important to take a global perspective on resistant infections because we can learn about regions and countries that are experiencing the greatest burden, information that was previously unknown,” Dr. Ikuta said. “With these estimates policy makers can prioritize regions that are hotspots and would most benefit from additional interventions.”
Furthermore, the study emphasized the global nature of AMR. “We’ve seen over the last 2 years with COVID-19 that this sort of problem doesn’t respect country borders, and high rates of resistance in one location can spread across a region or spread globally pretty quickly,” Dr. Ikuta said.
Leading resistant infections
Lower respiratory and thorax infections, bloodstream infections, and intra-abdominal infections together accounted for almost 79% of such deaths linked to AMR.
The six leading pathogens are likely household names among infectious disease specialists. The researchers found Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa, each responsible for more than 250,000 AMR-associated deaths.
The study also revealed that resistance to several first-line antibiotic agents often used empirically to treat infections accounted for more than 70% of the AMR-attributable deaths. These included fluoroquinolones and beta-lactam antibiotics such as carbapenems, cephalosporins, and penicillins.

Consistent with previous studies, MRSA stood out as a major cause of mortality. Of 88 different pathogen-drug combinations evaluated, MRSA was responsible for the most mortality: more than 100,000 deaths and 3·5 million disability-adjusted life-years.
The current study findings on MRSA “being a particularly nasty culprit” in AMR infections validates previous work that reported similar results, Vance Fowler, MD, told this news organization when asked to comment on the research. “That is reassuring.”
Potential solutions offered
Dr. Murray and colleagues outlined five strategies to address the challenge of bacterial AMR:
- Infection prevention and control remain paramount in minimizing infections in general and AMR infections in particular.
- More vaccines are needed to reduce the need for antibiotics. “Vaccines are available for only one of the six leading pathogens (S. pneumoniae), although new vaccine programs are underway for S. aureus, E. coli, and others,” the researchers wrote.
- Reduce antibiotic use unrelated to treatment of human disease.
- Avoid using antibiotics for viral infections and other unnecessary indications.
- Invest in new antibiotic development and ensure access to second-line agents in areas without widespread access.
“Identifying strategies that can work to reduce the burden of bacterial AMR – either across a wide range of settings or those that are specifically tailored to the resources available and leading pathogen-drug combinations in a particular setting – is an urgent priority,” the researchers noted.
Admirable AMR research
The results of the study are “startling, but not surprising,” said Dr. Fowler, professor of medicine at Duke University, Durham, N.C.
The authors did a “nice job” of addressing both deaths attributable and associated with AMR, Dr. Fowler added. “Those two categories unlock applications, not just in terms of how you interpret it but also what you do about it.”
The deaths attributable to AMR show that there is more work to be done regarding infection control and prevention, Dr. Fowler said, including in areas of the world like lower- and middle-income countries where infection resistance is most pronounced.
The deaths associated with AMR can be more challenging to calculate – people with infections can die for multiple reasons. However, Dr. Fowler applauded the researchers for doing “as good a job as you can” in estimating the extent of associated mortality.
‘The overlooked pandemic of antimicrobial resistance’
In an accompanying editorial in The Lancet, Ramanan Laxminarayan, PhD, MPH, wrote: “As COVID-19 rages on, the pandemic of antimicrobial resistance continues in the shadows. The toll taken by AMR on patients and their families is largely invisible but is reflected in prolonged bacterial infections that extend hospital stays and cause needless deaths.”
Dr. Laxminarayan pointed out an irony with AMR in different regions. Some of the AMR burden in sub-Saharan Africa is “probably due to inadequate access to antibiotics and high infection levels, albeit at low levels of resistance, whereas in south Asia and Latin America, it is because of high resistance even with good access to antibiotics.”
More funding to address AMR is needed, Dr. Laxminarayan noted. “Even the lower end of 911,000 deaths estimated by Murray and colleagues is higher than the number of deaths from HIV, which attracts close to U.S. $50 billion each year. However, global spending on addressing AMR is probably much lower than that.” Dr. Laxminarayan is an economist and epidemiologist affiliated with the Center for Disease Dynamics, Economics & Policy in Washington, D.C., and the Global Antibiotic Research and Development Partnership in Geneva.
An overlap with COVID-19
The Lancet report is likely “to bring more attention to AMR, especially since so many people have been distracted by COVID, and rightly so,” Dr. Fowler predicted. “The world has had its hands full with COVID.”
The two infections interact in direct ways, Dr. Fowler added. For example, some people hospitalized for COVID-19 for an extended time could develop progressively drug-resistant bacteria – leading to a superinfection.
The overlap could be illustrated by a Venn diagram, he said. A yellow circle could illustrate people with COVID-19 who are asymptomatic or who remain outpatients. Next to that would be a blue circle showing people who develop AMR infections. Where the two circles overlap would be green for those hospitalized who – because of receiving steroids, being on a ventilator, or getting a central line – develop a superinfection.
Official guidance continues
The study comes in the context of recent guidance and federal action on AMR. For example, the Infectious Diseases Society of America released new guidelines for AMR in November 2021 as part of ongoing advice on prevention and treatment of this “ongoing crisis.”
This most recent IDSA guidance addresses three pathogens in particular: AmpC beta-lactamase–producing Enterobacterales, carbapenem-resistant A. baumannii, and Stenotrophomonas maltophilia.
Also in November, the World Health Organization released an updated fact sheet on antimicrobial resistance. The WHO declared AMR one of the world’s top 10 global public health threats. The agency emphasized that misuse and overuse of antimicrobials are the main drivers in the development of drug-resistant pathogens. The WHO also pointed out that lack of clean water and sanitation in many areas of the world contribute to spread of microbes, including those resistant to current treatment options.
In September 2021, the Biden administration acknowledged the threat of AMR with allocation of more than $2 billion of the American Rescue Plan money for prevention and treatment of these infections.
Asked if there are any reasons for hope or optimism at this point, Dr. Ikuta said: “Definitely. We know what needs to be done to combat the spread of resistance. COVID-19 has demonstrated the importance of global commitment to infection control measures, such as hand washing and surveillance, and rapid investments in treatments, which can all be applied to antimicrobial resistance.”
The Bill & Melinda Gates Foundation, the Wellcome Trust, and the U.K. Department of Health and Social Care using U.K. aid funding managed by the Fleming Fund and other organizations provided funding for the study. Dr. Ikuta and Dr. Laxminarayan have disclosed no relevant financial relationships. Dr. Fowler reported receiving grants or honoraria, as well as serving as a consultant, for numerous sources. He also reported a patent pending in sepsis diagnostics and serving as chair of the V710 Scientific Advisory Committee (Merck).
A version of this article first appeared on Medscape.com.
More than HIV, more than malaria.
In terms of preventable deaths, 1.27 million people could have been saved if drug-resistant infections were replaced with infections susceptible to current antibiotics. Furthermore, 4.95 million fewer people would have died if drug-resistant infections were replaced by no infections, researchers estimated.
Although the COVID-19 pandemic took some focus off the AMR burden worldwide over the past 2 years, the urgency to address risk to public health did not ebb. In fact, based on the findings, the researchers noted that AMR is now a leading cause of death worldwide.
“If left unchecked, the spread of AMR could make many bacterial pathogens much more lethal in the future than they are today,” the researchers noted in the study, published online Jan. 20, 2022, in The Lancet.
“These findings are a warning signal that antibiotic resistance is placing pressure on health care systems and leading to significant health loss,” study author Kevin Ikuta, MD, MPH, told this news organization.
“We need to continue to adhere to and support infection prevention and control programs, be thoughtful about our antibiotic use, and advocate for increased funding to vaccine discovery and the antibiotic development pipeline,” added Dr. Ikuta, health sciences assistant clinical professor of medicine at the University of California, Los Angeles.
Although many investigators have studied AMR, this study is the largest in scope, covering 204 countries and territories and incorporating data on a comprehensive range of pathogens and pathogen-drug combinations.
Dr. Ikuta, lead author Christopher J.L. Murray, DPhil, and colleagues estimated the global burden of AMR using the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2019. They specifically looked at rates of death directly attributed to and separately those associated with resistance.
Regional differences
Broken down by 21 regions, Australasia had 6.5 deaths per 100,000 people attributable to AMR, the lowest rate reported. This region also had 28 deaths per 100,000 associated with AMR.
Researchers found the highest rates in western sub-Saharan Africa. Deaths attributable to AMR were 27.3 per 100,000 and associated death rate was 114.8 per 100,000.
Lower- and middle-income regions had the highest AMR death rates, although resistance remains a high-priority issue for high-income countries as well.
“It’s important to take a global perspective on resistant infections because we can learn about regions and countries that are experiencing the greatest burden, information that was previously unknown,” Dr. Ikuta said. “With these estimates policy makers can prioritize regions that are hotspots and would most benefit from additional interventions.”
Furthermore, the study emphasized the global nature of AMR. “We’ve seen over the last 2 years with COVID-19 that this sort of problem doesn’t respect country borders, and high rates of resistance in one location can spread across a region or spread globally pretty quickly,” Dr. Ikuta said.
Leading resistant infections
Lower respiratory and thorax infections, bloodstream infections, and intra-abdominal infections together accounted for almost 79% of such deaths linked to AMR.
The six leading pathogens are likely household names among infectious disease specialists. The researchers found Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa, each responsible for more than 250,000 AMR-associated deaths.
The study also revealed that resistance to several first-line antibiotic agents often used empirically to treat infections accounted for more than 70% of the AMR-attributable deaths. These included fluoroquinolones and beta-lactam antibiotics such as carbapenems, cephalosporins, and penicillins.

Consistent with previous studies, MRSA stood out as a major cause of mortality. Of 88 different pathogen-drug combinations evaluated, MRSA was responsible for the most mortality: more than 100,000 deaths and 3·5 million disability-adjusted life-years.
The current study findings on MRSA “being a particularly nasty culprit” in AMR infections validates previous work that reported similar results, Vance Fowler, MD, told this news organization when asked to comment on the research. “That is reassuring.”
Potential solutions offered
Dr. Murray and colleagues outlined five strategies to address the challenge of bacterial AMR:
- Infection prevention and control remain paramount in minimizing infections in general and AMR infections in particular.
- More vaccines are needed to reduce the need for antibiotics. “Vaccines are available for only one of the six leading pathogens (S. pneumoniae), although new vaccine programs are underway for S. aureus, E. coli, and others,” the researchers wrote.
- Reduce antibiotic use unrelated to treatment of human disease.
- Avoid using antibiotics for viral infections and other unnecessary indications.
- Invest in new antibiotic development and ensure access to second-line agents in areas without widespread access.
“Identifying strategies that can work to reduce the burden of bacterial AMR – either across a wide range of settings or those that are specifically tailored to the resources available and leading pathogen-drug combinations in a particular setting – is an urgent priority,” the researchers noted.
Admirable AMR research
The results of the study are “startling, but not surprising,” said Dr. Fowler, professor of medicine at Duke University, Durham, N.C.
The authors did a “nice job” of addressing both deaths attributable and associated with AMR, Dr. Fowler added. “Those two categories unlock applications, not just in terms of how you interpret it but also what you do about it.”
The deaths attributable to AMR show that there is more work to be done regarding infection control and prevention, Dr. Fowler said, including in areas of the world like lower- and middle-income countries where infection resistance is most pronounced.
The deaths associated with AMR can be more challenging to calculate – people with infections can die for multiple reasons. However, Dr. Fowler applauded the researchers for doing “as good a job as you can” in estimating the extent of associated mortality.
‘The overlooked pandemic of antimicrobial resistance’
In an accompanying editorial in The Lancet, Ramanan Laxminarayan, PhD, MPH, wrote: “As COVID-19 rages on, the pandemic of antimicrobial resistance continues in the shadows. The toll taken by AMR on patients and their families is largely invisible but is reflected in prolonged bacterial infections that extend hospital stays and cause needless deaths.”
Dr. Laxminarayan pointed out an irony with AMR in different regions. Some of the AMR burden in sub-Saharan Africa is “probably due to inadequate access to antibiotics and high infection levels, albeit at low levels of resistance, whereas in south Asia and Latin America, it is because of high resistance even with good access to antibiotics.”
More funding to address AMR is needed, Dr. Laxminarayan noted. “Even the lower end of 911,000 deaths estimated by Murray and colleagues is higher than the number of deaths from HIV, which attracts close to U.S. $50 billion each year. However, global spending on addressing AMR is probably much lower than that.” Dr. Laxminarayan is an economist and epidemiologist affiliated with the Center for Disease Dynamics, Economics & Policy in Washington, D.C., and the Global Antibiotic Research and Development Partnership in Geneva.
An overlap with COVID-19
The Lancet report is likely “to bring more attention to AMR, especially since so many people have been distracted by COVID, and rightly so,” Dr. Fowler predicted. “The world has had its hands full with COVID.”
The two infections interact in direct ways, Dr. Fowler added. For example, some people hospitalized for COVID-19 for an extended time could develop progressively drug-resistant bacteria – leading to a superinfection.
The overlap could be illustrated by a Venn diagram, he said. A yellow circle could illustrate people with COVID-19 who are asymptomatic or who remain outpatients. Next to that would be a blue circle showing people who develop AMR infections. Where the two circles overlap would be green for those hospitalized who – because of receiving steroids, being on a ventilator, or getting a central line – develop a superinfection.
Official guidance continues
The study comes in the context of recent guidance and federal action on AMR. For example, the Infectious Diseases Society of America released new guidelines for AMR in November 2021 as part of ongoing advice on prevention and treatment of this “ongoing crisis.”
This most recent IDSA guidance addresses three pathogens in particular: AmpC beta-lactamase–producing Enterobacterales, carbapenem-resistant A. baumannii, and Stenotrophomonas maltophilia.
Also in November, the World Health Organization released an updated fact sheet on antimicrobial resistance. The WHO declared AMR one of the world’s top 10 global public health threats. The agency emphasized that misuse and overuse of antimicrobials are the main drivers in the development of drug-resistant pathogens. The WHO also pointed out that lack of clean water and sanitation in many areas of the world contribute to spread of microbes, including those resistant to current treatment options.
In September 2021, the Biden administration acknowledged the threat of AMR with allocation of more than $2 billion of the American Rescue Plan money for prevention and treatment of these infections.
Asked if there are any reasons for hope or optimism at this point, Dr. Ikuta said: “Definitely. We know what needs to be done to combat the spread of resistance. COVID-19 has demonstrated the importance of global commitment to infection control measures, such as hand washing and surveillance, and rapid investments in treatments, which can all be applied to antimicrobial resistance.”
The Bill & Melinda Gates Foundation, the Wellcome Trust, and the U.K. Department of Health and Social Care using U.K. aid funding managed by the Fleming Fund and other organizations provided funding for the study. Dr. Ikuta and Dr. Laxminarayan have disclosed no relevant financial relationships. Dr. Fowler reported receiving grants or honoraria, as well as serving as a consultant, for numerous sources. He also reported a patent pending in sepsis diagnostics and serving as chair of the V710 Scientific Advisory Committee (Merck).
A version of this article first appeared on Medscape.com.
Five things you should know about ‘free’ at-home COVID tests
Americans keep hearing that it is important to test frequently for COVID-19 at home. But just try to find an “at-home” rapid COVID test in a store and at a price that makes frequent tests affordable.
Testing, as well as mask-wearing, is an important measure if the country ever hopes to beat COVID, restore normal routines and get the economy running efficiently. To get Americans cheaper tests, the federal government now plans to have insurance companies pay for them.
You can either get one without any out-of-pocket expense from retail pharmacies that are part of an insurance company’s network or buy it at any store and get reimbursed by the insurer.
Congress said private insurers must cover all COVID testing and any associated medical services when it passed the Families First Coronavirus Response Act and the Coronavirus Aid, Relief and Economic Security, or CARES, Act. The have-insurance-pay-for-it solution has been used frequently through the pandemic. Insurance companies have been told to pay for polymerase chain reaction tests, COVID treatments and the administration of vaccines. (Taxpayers are paying for the cost of the vaccines themselves.) It appears to be an elegant solution for a politician because it looks free and isn’t using taxpayer money.
1. Are the tests really free?
Well, no. As many an economist will tell you, there ain’t no such thing as a free lunch. Someone has to pick up the tab. Initially, the insurance companies bear the cost. Cynthia Cox, a vice president at KFF who studies the Affordable Care Act and private insurers, said the total bill could amount to billions of dollars. Exactly how much depends on “how easy it is to get them, and how many will be reimbursed,” she said.
2. Will the insurance company just swallow those imposed costs?
If companies draw from the time-tested insurance giants’ playbook, they’ll pass along those costs to customers. “This will put upward pressure on premiums,” said Emily Gee, vice president and coordinator for health policy at the Center for American Progress.
Major insurance companies like Cigna, Anthem, UnitedHealthcare, and Aetna did not respond to requests to discuss this issue.
3. If that’s the case, why haven’t I been hit with higher premiums already?
Insurance companies had the chance last year to raise premiums but, mostly, they did not.
Why? Perhaps because insurers have so far made so much money during the pandemic they didn’t need to. For example, the industry’s profits in 2020 increased 41% to $31 billion from $22 billion, according to the National Association of Insurance Commissioners. The NAIC said the industry has continued its “tremendous growth trend” that started before COVID emerged. Companies will be reporting 2021 results soon.
The reason behind these profits is clear. You were paying premiums based on projections your insurance company made about how much health care consumers would use that year. Because people stayed home, had fewer accidents, postponed surgeries and often avoided going to visit the doctor or the hospital, insurers paid out less. They rebated some of their earnings back to customers, but they pocketed a lot more.
As the companies’ actuaries work on predicting 2023 expenditures, premiums could go up if they foresee more claims and expenses. Paying for millions of rapid tests is something they would include in their calculations.
4. Regardless of my premiums, will the tests cost me money directly?
It’s quite possible. If your insurance company doesn’t have an arrangement with a retailer where you can simply pick up your allotted tests, you’ll have to pay for them – at whatever price the store sets. If that’s the case, you’ll need to fill out a form to request a reimbursement from the insurance company. How many times have you lost receipts or just plain neglected to mail in for rebates on something you bought? A lot, right?
Here’s another thing: The reimbursement is set at $12 per test. If you pay $30 for a test – and that is not unheard of – your insurer is only on the hook for $12. You eat the $18.
And by the way, people on Medicare will have to pay for their tests themselves. People who get their health care covered by Medicaid can obtain free test kits at community centers.
A few free tests are supposed to arrive at every American home via the U.S. Postal Service. And the Biden administration has activated a website where Americans can order free tests from a cache of a billion the federal government ordered.
5. Will this help bring down the costs of at-home tests and make them easier to find?
The free COVID tests are unlikely to have much immediate impact on general cost and availability. You will still need to search for them. The federal measures likely will stimulate the demand for tests, which in the short term may make them harder to find.
But the demand, and some government guarantees to manufacturers, may induce test makers to make more of them faster. The increased competition and supply theoretically could bring down the price. There is certainly room for prices to decline since the wholesale cost of the test is between $5 and $7, analysts estimate. “It’s a big step in the right direction,” Ms. Gee said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Americans keep hearing that it is important to test frequently for COVID-19 at home. But just try to find an “at-home” rapid COVID test in a store and at a price that makes frequent tests affordable.
Testing, as well as mask-wearing, is an important measure if the country ever hopes to beat COVID, restore normal routines and get the economy running efficiently. To get Americans cheaper tests, the federal government now plans to have insurance companies pay for them.
You can either get one without any out-of-pocket expense from retail pharmacies that are part of an insurance company’s network or buy it at any store and get reimbursed by the insurer.
Congress said private insurers must cover all COVID testing and any associated medical services when it passed the Families First Coronavirus Response Act and the Coronavirus Aid, Relief and Economic Security, or CARES, Act. The have-insurance-pay-for-it solution has been used frequently through the pandemic. Insurance companies have been told to pay for polymerase chain reaction tests, COVID treatments and the administration of vaccines. (Taxpayers are paying for the cost of the vaccines themselves.) It appears to be an elegant solution for a politician because it looks free and isn’t using taxpayer money.
1. Are the tests really free?
Well, no. As many an economist will tell you, there ain’t no such thing as a free lunch. Someone has to pick up the tab. Initially, the insurance companies bear the cost. Cynthia Cox, a vice president at KFF who studies the Affordable Care Act and private insurers, said the total bill could amount to billions of dollars. Exactly how much depends on “how easy it is to get them, and how many will be reimbursed,” she said.
2. Will the insurance company just swallow those imposed costs?
If companies draw from the time-tested insurance giants’ playbook, they’ll pass along those costs to customers. “This will put upward pressure on premiums,” said Emily Gee, vice president and coordinator for health policy at the Center for American Progress.
Major insurance companies like Cigna, Anthem, UnitedHealthcare, and Aetna did not respond to requests to discuss this issue.
3. If that’s the case, why haven’t I been hit with higher premiums already?
Insurance companies had the chance last year to raise premiums but, mostly, they did not.
Why? Perhaps because insurers have so far made so much money during the pandemic they didn’t need to. For example, the industry’s profits in 2020 increased 41% to $31 billion from $22 billion, according to the National Association of Insurance Commissioners. The NAIC said the industry has continued its “tremendous growth trend” that started before COVID emerged. Companies will be reporting 2021 results soon.
The reason behind these profits is clear. You were paying premiums based on projections your insurance company made about how much health care consumers would use that year. Because people stayed home, had fewer accidents, postponed surgeries and often avoided going to visit the doctor or the hospital, insurers paid out less. They rebated some of their earnings back to customers, but they pocketed a lot more.
As the companies’ actuaries work on predicting 2023 expenditures, premiums could go up if they foresee more claims and expenses. Paying for millions of rapid tests is something they would include in their calculations.
4. Regardless of my premiums, will the tests cost me money directly?
It’s quite possible. If your insurance company doesn’t have an arrangement with a retailer where you can simply pick up your allotted tests, you’ll have to pay for them – at whatever price the store sets. If that’s the case, you’ll need to fill out a form to request a reimbursement from the insurance company. How many times have you lost receipts or just plain neglected to mail in for rebates on something you bought? A lot, right?
Here’s another thing: The reimbursement is set at $12 per test. If you pay $30 for a test – and that is not unheard of – your insurer is only on the hook for $12. You eat the $18.
And by the way, people on Medicare will have to pay for their tests themselves. People who get their health care covered by Medicaid can obtain free test kits at community centers.
A few free tests are supposed to arrive at every American home via the U.S. Postal Service. And the Biden administration has activated a website where Americans can order free tests from a cache of a billion the federal government ordered.
5. Will this help bring down the costs of at-home tests and make them easier to find?
The free COVID tests are unlikely to have much immediate impact on general cost and availability. You will still need to search for them. The federal measures likely will stimulate the demand for tests, which in the short term may make them harder to find.
But the demand, and some government guarantees to manufacturers, may induce test makers to make more of them faster. The increased competition and supply theoretically could bring down the price. There is certainly room for prices to decline since the wholesale cost of the test is between $5 and $7, analysts estimate. “It’s a big step in the right direction,” Ms. Gee said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Americans keep hearing that it is important to test frequently for COVID-19 at home. But just try to find an “at-home” rapid COVID test in a store and at a price that makes frequent tests affordable.
Testing, as well as mask-wearing, is an important measure if the country ever hopes to beat COVID, restore normal routines and get the economy running efficiently. To get Americans cheaper tests, the federal government now plans to have insurance companies pay for them.
You can either get one without any out-of-pocket expense from retail pharmacies that are part of an insurance company’s network or buy it at any store and get reimbursed by the insurer.
Congress said private insurers must cover all COVID testing and any associated medical services when it passed the Families First Coronavirus Response Act and the Coronavirus Aid, Relief and Economic Security, or CARES, Act. The have-insurance-pay-for-it solution has been used frequently through the pandemic. Insurance companies have been told to pay for polymerase chain reaction tests, COVID treatments and the administration of vaccines. (Taxpayers are paying for the cost of the vaccines themselves.) It appears to be an elegant solution for a politician because it looks free and isn’t using taxpayer money.
1. Are the tests really free?
Well, no. As many an economist will tell you, there ain’t no such thing as a free lunch. Someone has to pick up the tab. Initially, the insurance companies bear the cost. Cynthia Cox, a vice president at KFF who studies the Affordable Care Act and private insurers, said the total bill could amount to billions of dollars. Exactly how much depends on “how easy it is to get them, and how many will be reimbursed,” she said.
2. Will the insurance company just swallow those imposed costs?
If companies draw from the time-tested insurance giants’ playbook, they’ll pass along those costs to customers. “This will put upward pressure on premiums,” said Emily Gee, vice president and coordinator for health policy at the Center for American Progress.
Major insurance companies like Cigna, Anthem, UnitedHealthcare, and Aetna did not respond to requests to discuss this issue.
3. If that’s the case, why haven’t I been hit with higher premiums already?
Insurance companies had the chance last year to raise premiums but, mostly, they did not.
Why? Perhaps because insurers have so far made so much money during the pandemic they didn’t need to. For example, the industry’s profits in 2020 increased 41% to $31 billion from $22 billion, according to the National Association of Insurance Commissioners. The NAIC said the industry has continued its “tremendous growth trend” that started before COVID emerged. Companies will be reporting 2021 results soon.
The reason behind these profits is clear. You were paying premiums based on projections your insurance company made about how much health care consumers would use that year. Because people stayed home, had fewer accidents, postponed surgeries and often avoided going to visit the doctor or the hospital, insurers paid out less. They rebated some of their earnings back to customers, but they pocketed a lot more.
As the companies’ actuaries work on predicting 2023 expenditures, premiums could go up if they foresee more claims and expenses. Paying for millions of rapid tests is something they would include in their calculations.
4. Regardless of my premiums, will the tests cost me money directly?
It’s quite possible. If your insurance company doesn’t have an arrangement with a retailer where you can simply pick up your allotted tests, you’ll have to pay for them – at whatever price the store sets. If that’s the case, you’ll need to fill out a form to request a reimbursement from the insurance company. How many times have you lost receipts or just plain neglected to mail in for rebates on something you bought? A lot, right?
Here’s another thing: The reimbursement is set at $12 per test. If you pay $30 for a test – and that is not unheard of – your insurer is only on the hook for $12. You eat the $18.
And by the way, people on Medicare will have to pay for their tests themselves. People who get their health care covered by Medicaid can obtain free test kits at community centers.
A few free tests are supposed to arrive at every American home via the U.S. Postal Service. And the Biden administration has activated a website where Americans can order free tests from a cache of a billion the federal government ordered.
5. Will this help bring down the costs of at-home tests and make them easier to find?
The free COVID tests are unlikely to have much immediate impact on general cost and availability. You will still need to search for them. The federal measures likely will stimulate the demand for tests, which in the short term may make them harder to find.
But the demand, and some government guarantees to manufacturers, may induce test makers to make more of them faster. The increased competition and supply theoretically could bring down the price. There is certainly room for prices to decline since the wholesale cost of the test is between $5 and $7, analysts estimate. “It’s a big step in the right direction,” Ms. Gee said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Two studies detail the dangers of COVID in pregnancy
Two new studies show how COVID-19 threatens the health of pregnant people and their newborn infants.
A study conducted in Scotland showed that unvaccinated pregnant people who got COVID were much more likely to have a stillborn infant or one that dies in the first 28 days. The study also found that pregnant women infected with COVID died or needed hospitalization at a much higher rate than vaccinated women who got pregnant.
The University of Edinburgh and Public Health Scotland studied national data in 88,000 pregnancies between Dec. 2020 and Oct. 2021, according to the study published in Nature Medicine.
Overall, 77.4% of infections, 90.9% of COVID-related hospitalizations, and 98% of critical care cases occurred in the unvaccinated people, as did all newborn deaths.
The study said 2,364 babies were born to women infected with COVID, with 2,353 live births. Eleven babies were stillborn and eight live-born babies died within 28 days. Of the live births, 241 were premature.
The problems were more likely if the infection occurred 28 days or less before the delivery date, the researchers said.
The authors said the low vaccination rate among pregnant people was a problem. Only 32% of people giving birth in Oct. 2021 were fully vaccinated, while 77% of the Scottish female population aged 18-44 was fully vaccinated.
“Vaccine hesitancy in pregnancy thus requires addressing, especially in light of new recommendations for booster vaccination administration 3 months after the initial vaccination course to help protect against new variants such as Omicron,” the authors wrote. “Addressing low vaccine uptake rates in pregnant women is imperative to protect the health of women and babies in the ongoing pandemic.”
Vaccinated women who were pregnant had complication rates that were about the same for all pregnant women, the study shows.
The second study, published in The Lancet, found that women who got COVID while pregnant in five Western U.S. states were more likely to have premature births, low birth weights, and stillbirths, even when the COVID cases are mild.
The Institute for Systems Biology researchers in Seattle studied data for women who gave birth in Alaska, California, Montana, Oregon, or Washington from March 5, 2020, to July 4, 2021. About 18,000 of them were tested for COVID, with 882 testing positive. Of the positive tests, 85 came in the first trimester, 226 in the second trimester, and 571 in the third semester. None of the pregnant women had been vaccinated at the time they were infected.
Most of the birth problems occurred with first and second trimester infections, the study noted, and problems occurred even if the pregnant person didn’t have respiratory complications, a major COVID symptom.
“Pregnant people are at an increased risk of adverse outcomes following SARS-CoV-2 infection, even when maternal COVID-19 is less severe, and they may benefit from increased monitoring following infection,” Jennifer Hadlock, MD, an author of the paper, said in a news release.
The study also pointed out continuing inequities in health care, with most of the positive cases occurring among young, non-White people with Medicaid and high body mass index.
A version of this article first appeared on WebMD.com.
Two new studies show how COVID-19 threatens the health of pregnant people and their newborn infants.
A study conducted in Scotland showed that unvaccinated pregnant people who got COVID were much more likely to have a stillborn infant or one that dies in the first 28 days. The study also found that pregnant women infected with COVID died or needed hospitalization at a much higher rate than vaccinated women who got pregnant.
The University of Edinburgh and Public Health Scotland studied national data in 88,000 pregnancies between Dec. 2020 and Oct. 2021, according to the study published in Nature Medicine.
Overall, 77.4% of infections, 90.9% of COVID-related hospitalizations, and 98% of critical care cases occurred in the unvaccinated people, as did all newborn deaths.
The study said 2,364 babies were born to women infected with COVID, with 2,353 live births. Eleven babies were stillborn and eight live-born babies died within 28 days. Of the live births, 241 were premature.
The problems were more likely if the infection occurred 28 days or less before the delivery date, the researchers said.
The authors said the low vaccination rate among pregnant people was a problem. Only 32% of people giving birth in Oct. 2021 were fully vaccinated, while 77% of the Scottish female population aged 18-44 was fully vaccinated.
“Vaccine hesitancy in pregnancy thus requires addressing, especially in light of new recommendations for booster vaccination administration 3 months after the initial vaccination course to help protect against new variants such as Omicron,” the authors wrote. “Addressing low vaccine uptake rates in pregnant women is imperative to protect the health of women and babies in the ongoing pandemic.”
Vaccinated women who were pregnant had complication rates that were about the same for all pregnant women, the study shows.
The second study, published in The Lancet, found that women who got COVID while pregnant in five Western U.S. states were more likely to have premature births, low birth weights, and stillbirths, even when the COVID cases are mild.
The Institute for Systems Biology researchers in Seattle studied data for women who gave birth in Alaska, California, Montana, Oregon, or Washington from March 5, 2020, to July 4, 2021. About 18,000 of them were tested for COVID, with 882 testing positive. Of the positive tests, 85 came in the first trimester, 226 in the second trimester, and 571 in the third semester. None of the pregnant women had been vaccinated at the time they were infected.
Most of the birth problems occurred with first and second trimester infections, the study noted, and problems occurred even if the pregnant person didn’t have respiratory complications, a major COVID symptom.
“Pregnant people are at an increased risk of adverse outcomes following SARS-CoV-2 infection, even when maternal COVID-19 is less severe, and they may benefit from increased monitoring following infection,” Jennifer Hadlock, MD, an author of the paper, said in a news release.
The study also pointed out continuing inequities in health care, with most of the positive cases occurring among young, non-White people with Medicaid and high body mass index.
A version of this article first appeared on WebMD.com.
Two new studies show how COVID-19 threatens the health of pregnant people and their newborn infants.
A study conducted in Scotland showed that unvaccinated pregnant people who got COVID were much more likely to have a stillborn infant or one that dies in the first 28 days. The study also found that pregnant women infected with COVID died or needed hospitalization at a much higher rate than vaccinated women who got pregnant.
The University of Edinburgh and Public Health Scotland studied national data in 88,000 pregnancies between Dec. 2020 and Oct. 2021, according to the study published in Nature Medicine.
Overall, 77.4% of infections, 90.9% of COVID-related hospitalizations, and 98% of critical care cases occurred in the unvaccinated people, as did all newborn deaths.
The study said 2,364 babies were born to women infected with COVID, with 2,353 live births. Eleven babies were stillborn and eight live-born babies died within 28 days. Of the live births, 241 were premature.
The problems were more likely if the infection occurred 28 days or less before the delivery date, the researchers said.
The authors said the low vaccination rate among pregnant people was a problem. Only 32% of people giving birth in Oct. 2021 were fully vaccinated, while 77% of the Scottish female population aged 18-44 was fully vaccinated.
“Vaccine hesitancy in pregnancy thus requires addressing, especially in light of new recommendations for booster vaccination administration 3 months after the initial vaccination course to help protect against new variants such as Omicron,” the authors wrote. “Addressing low vaccine uptake rates in pregnant women is imperative to protect the health of women and babies in the ongoing pandemic.”
Vaccinated women who were pregnant had complication rates that were about the same for all pregnant women, the study shows.
The second study, published in The Lancet, found that women who got COVID while pregnant in five Western U.S. states were more likely to have premature births, low birth weights, and stillbirths, even when the COVID cases are mild.
The Institute for Systems Biology researchers in Seattle studied data for women who gave birth in Alaska, California, Montana, Oregon, or Washington from March 5, 2020, to July 4, 2021. About 18,000 of them were tested for COVID, with 882 testing positive. Of the positive tests, 85 came in the first trimester, 226 in the second trimester, and 571 in the third semester. None of the pregnant women had been vaccinated at the time they were infected.
Most of the birth problems occurred with first and second trimester infections, the study noted, and problems occurred even if the pregnant person didn’t have respiratory complications, a major COVID symptom.
“Pregnant people are at an increased risk of adverse outcomes following SARS-CoV-2 infection, even when maternal COVID-19 is less severe, and they may benefit from increased monitoring following infection,” Jennifer Hadlock, MD, an author of the paper, said in a news release.
The study also pointed out continuing inequities in health care, with most of the positive cases occurring among young, non-White people with Medicaid and high body mass index.
A version of this article first appeared on WebMD.com.











