User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Serum testosterone and estradiol levels associated with current asthma in women
possibly explaining in part the different prevalence of asthma in men and women, according to the findings of a large cross-sectional population based study.
Yueh-Ying Han, PhD, of the Children’s Hospital of Pittsburgh and colleagues investigated the role of free testosterone and estradiol levels and current asthma among adults. The impact of obesity on that association was also examined. The investigators analyzed data from 7,615 adults (3,953 men and 3,662 women) who participated in the 2013-2014 and 2015-2016 U.S. National Health and Nutrition Examination Survey. The data included health interviews, examination components, and laboratory tests on each patient. Serum samples were analyzed by the division of laboratory sciences of the Centers for Disease Control and Prevention. Logistic regression was used for the multivariable analysis of sex hormone levels (as quartiles) and current asthma, and the analysis was done separately on men and women. Pregnant women were excluded, in addition to individuals with incomplete data. The exclusions tended to be Hispanic, former smokers, lower income, and lacking private insurance. The overall prevalence of current asthma in the sample was 9% (6% in men and 13% in women).
Three models were generated based on serum levels in women and in men.
For model 1 (unadjusted for estradiol), women whose serum testosterone levels were in the second and fourth quartiles had 30%-45% significantly lower odds of having current asthma than those whose serum testosterone level was in the lowest quartile. Among men, those whose serum testosterone levels were in the second and fourth quartiles had 12%-13% lower odds for current asthma.
For model 2 (unadjusted for free testosterone), women whose serum estradiol levels were in the third quartile had 34% significantly lower odds of having current asthma than those whose estradiol levels were in the lowest quartile. The findings were similar for men, that is, those whose serum estradiol levels were in the third quartile had 30% lower odds for having asthma, compared with those with in the lowest quartile.
For model 3 (a multivariable model including serum levels of both estradiol and free testosterone), women whose serum testosterone levels were in the second and fourth quartiles had 30% and 44% lower odds of current asthma than those whose serum testosterone levels were in the lowest quartile. But in this multivariable model, the association between serum estradiol and current asthma was not significant. Among men (models 1-3), the magnitude of the estimated effect of serum testosterone and serum estradiol on current asthma was similar to that observed in female participants, but neither serum testosterone nor serum estradiol was significantly associated with current asthma.
The investigators then analyzed the impact of obesity on the relationship between serum hormone levels and obesity. Obesity was defined as body mass index equal to or greater than 30 kg/m2. A total of 1,370 men and 1,653 women were included in this analysis. In multivariable analyses of the obese participants, adjustment without (model 1) and with (model 3) serum estradiol, serum free-testosterone levels in the highest (fourth) quartile were significantly associated with reduced odds of asthma in obese women. In multivariable analyses without (model 2) and with (model 3), serum estradiol levels above the first quartile were significantly associated with reduced odds of current asthma in obese women.
In contrast to the results in obese women, neither serum free testosterone nor serum estradiol was significantly associated with current asthma in obese men or nonobese women.
Dr. Han and coauthors suggested a possible mechanism of the role of sex hormones in asthma. “Androgens such as testosterone may reduce innate and adaptive immune responses, while estrogen and progesterone may enhance T-helper cell type 2 allergic airway inflammation.”
They concluded: “We found that elevated serum levels of both free testosterone and estradiol were significantly associated with reduced odds of asthma in obese women, and that elevated levels of serum estradiol were significantly associated with reduced odds of asthma in nonobese men. Our findings further suggest that sex steroid hormones play a role in known sex differences in asthma among adults.”
One coauthor has received research materials from Merck and GlaxoSmithKline (inhaled steroids), as well as Pharmavite (vitamin D and placebo capsules), to provide medications free of cost to participants in National Institutes for Health–funded studies, unrelated to the current work. The other authors reported no conflicts of interest.
SOURCE: Han Y-Y et al. J Respir Crit Care Med. 2019 Sep 16. doi: 10.1164/rccm.201905-0996OC.
possibly explaining in part the different prevalence of asthma in men and women, according to the findings of a large cross-sectional population based study.
Yueh-Ying Han, PhD, of the Children’s Hospital of Pittsburgh and colleagues investigated the role of free testosterone and estradiol levels and current asthma among adults. The impact of obesity on that association was also examined. The investigators analyzed data from 7,615 adults (3,953 men and 3,662 women) who participated in the 2013-2014 and 2015-2016 U.S. National Health and Nutrition Examination Survey. The data included health interviews, examination components, and laboratory tests on each patient. Serum samples were analyzed by the division of laboratory sciences of the Centers for Disease Control and Prevention. Logistic regression was used for the multivariable analysis of sex hormone levels (as quartiles) and current asthma, and the analysis was done separately on men and women. Pregnant women were excluded, in addition to individuals with incomplete data. The exclusions tended to be Hispanic, former smokers, lower income, and lacking private insurance. The overall prevalence of current asthma in the sample was 9% (6% in men and 13% in women).
Three models were generated based on serum levels in women and in men.
For model 1 (unadjusted for estradiol), women whose serum testosterone levels were in the second and fourth quartiles had 30%-45% significantly lower odds of having current asthma than those whose serum testosterone level was in the lowest quartile. Among men, those whose serum testosterone levels were in the second and fourth quartiles had 12%-13% lower odds for current asthma.
For model 2 (unadjusted for free testosterone), women whose serum estradiol levels were in the third quartile had 34% significantly lower odds of having current asthma than those whose estradiol levels were in the lowest quartile. The findings were similar for men, that is, those whose serum estradiol levels were in the third quartile had 30% lower odds for having asthma, compared with those with in the lowest quartile.
For model 3 (a multivariable model including serum levels of both estradiol and free testosterone), women whose serum testosterone levels were in the second and fourth quartiles had 30% and 44% lower odds of current asthma than those whose serum testosterone levels were in the lowest quartile. But in this multivariable model, the association between serum estradiol and current asthma was not significant. Among men (models 1-3), the magnitude of the estimated effect of serum testosterone and serum estradiol on current asthma was similar to that observed in female participants, but neither serum testosterone nor serum estradiol was significantly associated with current asthma.
The investigators then analyzed the impact of obesity on the relationship between serum hormone levels and obesity. Obesity was defined as body mass index equal to or greater than 30 kg/m2. A total of 1,370 men and 1,653 women were included in this analysis. In multivariable analyses of the obese participants, adjustment without (model 1) and with (model 3) serum estradiol, serum free-testosterone levels in the highest (fourth) quartile were significantly associated with reduced odds of asthma in obese women. In multivariable analyses without (model 2) and with (model 3), serum estradiol levels above the first quartile were significantly associated with reduced odds of current asthma in obese women.
In contrast to the results in obese women, neither serum free testosterone nor serum estradiol was significantly associated with current asthma in obese men or nonobese women.
Dr. Han and coauthors suggested a possible mechanism of the role of sex hormones in asthma. “Androgens such as testosterone may reduce innate and adaptive immune responses, while estrogen and progesterone may enhance T-helper cell type 2 allergic airway inflammation.”
They concluded: “We found that elevated serum levels of both free testosterone and estradiol were significantly associated with reduced odds of asthma in obese women, and that elevated levels of serum estradiol were significantly associated with reduced odds of asthma in nonobese men. Our findings further suggest that sex steroid hormones play a role in known sex differences in asthma among adults.”
One coauthor has received research materials from Merck and GlaxoSmithKline (inhaled steroids), as well as Pharmavite (vitamin D and placebo capsules), to provide medications free of cost to participants in National Institutes for Health–funded studies, unrelated to the current work. The other authors reported no conflicts of interest.
SOURCE: Han Y-Y et al. J Respir Crit Care Med. 2019 Sep 16. doi: 10.1164/rccm.201905-0996OC.
possibly explaining in part the different prevalence of asthma in men and women, according to the findings of a large cross-sectional population based study.
Yueh-Ying Han, PhD, of the Children’s Hospital of Pittsburgh and colleagues investigated the role of free testosterone and estradiol levels and current asthma among adults. The impact of obesity on that association was also examined. The investigators analyzed data from 7,615 adults (3,953 men and 3,662 women) who participated in the 2013-2014 and 2015-2016 U.S. National Health and Nutrition Examination Survey. The data included health interviews, examination components, and laboratory tests on each patient. Serum samples were analyzed by the division of laboratory sciences of the Centers for Disease Control and Prevention. Logistic regression was used for the multivariable analysis of sex hormone levels (as quartiles) and current asthma, and the analysis was done separately on men and women. Pregnant women were excluded, in addition to individuals with incomplete data. The exclusions tended to be Hispanic, former smokers, lower income, and lacking private insurance. The overall prevalence of current asthma in the sample was 9% (6% in men and 13% in women).
Three models were generated based on serum levels in women and in men.
For model 1 (unadjusted for estradiol), women whose serum testosterone levels were in the second and fourth quartiles had 30%-45% significantly lower odds of having current asthma than those whose serum testosterone level was in the lowest quartile. Among men, those whose serum testosterone levels were in the second and fourth quartiles had 12%-13% lower odds for current asthma.
For model 2 (unadjusted for free testosterone), women whose serum estradiol levels were in the third quartile had 34% significantly lower odds of having current asthma than those whose estradiol levels were in the lowest quartile. The findings were similar for men, that is, those whose serum estradiol levels were in the third quartile had 30% lower odds for having asthma, compared with those with in the lowest quartile.
For model 3 (a multivariable model including serum levels of both estradiol and free testosterone), women whose serum testosterone levels were in the second and fourth quartiles had 30% and 44% lower odds of current asthma than those whose serum testosterone levels were in the lowest quartile. But in this multivariable model, the association between serum estradiol and current asthma was not significant. Among men (models 1-3), the magnitude of the estimated effect of serum testosterone and serum estradiol on current asthma was similar to that observed in female participants, but neither serum testosterone nor serum estradiol was significantly associated with current asthma.
The investigators then analyzed the impact of obesity on the relationship between serum hormone levels and obesity. Obesity was defined as body mass index equal to or greater than 30 kg/m2. A total of 1,370 men and 1,653 women were included in this analysis. In multivariable analyses of the obese participants, adjustment without (model 1) and with (model 3) serum estradiol, serum free-testosterone levels in the highest (fourth) quartile were significantly associated with reduced odds of asthma in obese women. In multivariable analyses without (model 2) and with (model 3), serum estradiol levels above the first quartile were significantly associated with reduced odds of current asthma in obese women.
In contrast to the results in obese women, neither serum free testosterone nor serum estradiol was significantly associated with current asthma in obese men or nonobese women.
Dr. Han and coauthors suggested a possible mechanism of the role of sex hormones in asthma. “Androgens such as testosterone may reduce innate and adaptive immune responses, while estrogen and progesterone may enhance T-helper cell type 2 allergic airway inflammation.”
They concluded: “We found that elevated serum levels of both free testosterone and estradiol were significantly associated with reduced odds of asthma in obese women, and that elevated levels of serum estradiol were significantly associated with reduced odds of asthma in nonobese men. Our findings further suggest that sex steroid hormones play a role in known sex differences in asthma among adults.”
One coauthor has received research materials from Merck and GlaxoSmithKline (inhaled steroids), as well as Pharmavite (vitamin D and placebo capsules), to provide medications free of cost to participants in National Institutes for Health–funded studies, unrelated to the current work. The other authors reported no conflicts of interest.
SOURCE: Han Y-Y et al. J Respir Crit Care Med. 2019 Sep 16. doi: 10.1164/rccm.201905-0996OC.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Step-up therapy with glucocorticoids benefits black children with asthma
based on data from 280 children aged 5-11 years with at least one grandparent identified as black.
Previous studies have suggested that long-acting beta2-agonists (LABAs) may be more effective for patients with poorly controlled asthma, but such step-up therapy has not been well studied in black patients, wrote Michael E. Wechsler, MD, of National Jewish Health, Denver, and colleagues.
In a study published in the New England Journal of Medicine, the researchers reported results of two parallel BARD (Best African American Response to Asthma Drugs) trials conducted at nine centers between January 2014 and March 2016 of individuals with poorly controlled asthma. One trial included 280 children aged 5-11 years (average age, 8.5 years); the second trial included adolescents aged 12 years and older and adults (average age, 37 years) who had family backgrounds that were similar to those of the children.
The researchers randomized the children to four groups to compare the following protocols: doubling the dose of a glucocorticoid (fluticasone propionate) to a dose of 100 mcg, twice daily (the double-fluticasone group); doubling the dose of fluticasone to 100 mcg and adding 50 mcg of the LABA salmeterol (the salmeterol/double-fluticasone group); quintupling the dose of fluticasone to 250 mcg (the quintuple-fluticasone group); or quintupling the dose of fluticasone to 250 mcg and adding 50 mcg of salmeterol (the salmeterol/quintuple-fluticasone group). The trial consisted of a four-way crossover design with each treatment period lasting 14 weeks.
The primary outcome was a composite measure including asthma exacerbations, asthma control days, and percentage of predicted forced expiratory volume in the first second at the end of each treatment.
Overall, a superior response occurred in 53% of the salmeterol/double-fluticasone group, 41% of the double-fluticasone group, 43% of the salmeterol/quintuple fluticasone group, and 47% of the quintuple-fluticasone group.
The superior response was 46% for both groups when the researchers compared a quintupled dose of fluticasone propionate (250 mcg) with a two step–up strategy of adding salmeterol at a dose of 50 mcg and increasing the dose of fluticasone to 100 mcg.
“In contrast to black adults and white persons of all ages, almost half the children who had at least one grandparent who identified as black and who had poorly controlled asthma had a superior response to an increased dose of an inhaled glucocorticoid over the addition of a LABA,” Dr. Wechsler and coauthors wrote. No more than 12% of the children in any treatment group did not have a superior response. No significant differences in reports of respiratory tract infections or pneumonia were seen between the groups. Children younger than 8 years showed a decrease in the ratio of urinary cortisol to creatinine with an increased dose of inhaled glucocorticoids.
In the adolescent and adult study using the same treatment protocols, 20%-25% of patients did not have a differential outcome between treatments. “In adolescents and adults, the addition of a LABA was more likely to produce superior responses than increasing the dose of an inhaled glucocorticoid,” Dr. Wechsler and coauthors wrote.
The study findings were limited by several factors, including the inability to assess long-term effects on growth and inability to detect biomarkers associated with responses to specific therapies, the researchers noted. However, the results suggest that black children with poorly controlled asthma can benefit from additional inhaled glucocorticoids, and larger studies are needed to identify the best treatment for this patient population.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Wechsler reported relationships with companies including AstraZeneca, Equillium, Genentech, GlaxoSmithKline, Mylan, Novartis, Regeneron, resTORbio, Sanofi, and others. Coauthors identified relationships with numerous pharmaceutical companies.
SOURCE: Wechsler ME et al. N Engl J Med. 2019 Sep 25. doi: 10.1056/NEJMoa1905560.
based on data from 280 children aged 5-11 years with at least one grandparent identified as black.
Previous studies have suggested that long-acting beta2-agonists (LABAs) may be more effective for patients with poorly controlled asthma, but such step-up therapy has not been well studied in black patients, wrote Michael E. Wechsler, MD, of National Jewish Health, Denver, and colleagues.
In a study published in the New England Journal of Medicine, the researchers reported results of two parallel BARD (Best African American Response to Asthma Drugs) trials conducted at nine centers between January 2014 and March 2016 of individuals with poorly controlled asthma. One trial included 280 children aged 5-11 years (average age, 8.5 years); the second trial included adolescents aged 12 years and older and adults (average age, 37 years) who had family backgrounds that were similar to those of the children.
The researchers randomized the children to four groups to compare the following protocols: doubling the dose of a glucocorticoid (fluticasone propionate) to a dose of 100 mcg, twice daily (the double-fluticasone group); doubling the dose of fluticasone to 100 mcg and adding 50 mcg of the LABA salmeterol (the salmeterol/double-fluticasone group); quintupling the dose of fluticasone to 250 mcg (the quintuple-fluticasone group); or quintupling the dose of fluticasone to 250 mcg and adding 50 mcg of salmeterol (the salmeterol/quintuple-fluticasone group). The trial consisted of a four-way crossover design with each treatment period lasting 14 weeks.
The primary outcome was a composite measure including asthma exacerbations, asthma control days, and percentage of predicted forced expiratory volume in the first second at the end of each treatment.
Overall, a superior response occurred in 53% of the salmeterol/double-fluticasone group, 41% of the double-fluticasone group, 43% of the salmeterol/quintuple fluticasone group, and 47% of the quintuple-fluticasone group.
The superior response was 46% for both groups when the researchers compared a quintupled dose of fluticasone propionate (250 mcg) with a two step–up strategy of adding salmeterol at a dose of 50 mcg and increasing the dose of fluticasone to 100 mcg.
“In contrast to black adults and white persons of all ages, almost half the children who had at least one grandparent who identified as black and who had poorly controlled asthma had a superior response to an increased dose of an inhaled glucocorticoid over the addition of a LABA,” Dr. Wechsler and coauthors wrote. No more than 12% of the children in any treatment group did not have a superior response. No significant differences in reports of respiratory tract infections or pneumonia were seen between the groups. Children younger than 8 years showed a decrease in the ratio of urinary cortisol to creatinine with an increased dose of inhaled glucocorticoids.
In the adolescent and adult study using the same treatment protocols, 20%-25% of patients did not have a differential outcome between treatments. “In adolescents and adults, the addition of a LABA was more likely to produce superior responses than increasing the dose of an inhaled glucocorticoid,” Dr. Wechsler and coauthors wrote.
The study findings were limited by several factors, including the inability to assess long-term effects on growth and inability to detect biomarkers associated with responses to specific therapies, the researchers noted. However, the results suggest that black children with poorly controlled asthma can benefit from additional inhaled glucocorticoids, and larger studies are needed to identify the best treatment for this patient population.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Wechsler reported relationships with companies including AstraZeneca, Equillium, Genentech, GlaxoSmithKline, Mylan, Novartis, Regeneron, resTORbio, Sanofi, and others. Coauthors identified relationships with numerous pharmaceutical companies.
SOURCE: Wechsler ME et al. N Engl J Med. 2019 Sep 25. doi: 10.1056/NEJMoa1905560.
based on data from 280 children aged 5-11 years with at least one grandparent identified as black.
Previous studies have suggested that long-acting beta2-agonists (LABAs) may be more effective for patients with poorly controlled asthma, but such step-up therapy has not been well studied in black patients, wrote Michael E. Wechsler, MD, of National Jewish Health, Denver, and colleagues.
In a study published in the New England Journal of Medicine, the researchers reported results of two parallel BARD (Best African American Response to Asthma Drugs) trials conducted at nine centers between January 2014 and March 2016 of individuals with poorly controlled asthma. One trial included 280 children aged 5-11 years (average age, 8.5 years); the second trial included adolescents aged 12 years and older and adults (average age, 37 years) who had family backgrounds that were similar to those of the children.
The researchers randomized the children to four groups to compare the following protocols: doubling the dose of a glucocorticoid (fluticasone propionate) to a dose of 100 mcg, twice daily (the double-fluticasone group); doubling the dose of fluticasone to 100 mcg and adding 50 mcg of the LABA salmeterol (the salmeterol/double-fluticasone group); quintupling the dose of fluticasone to 250 mcg (the quintuple-fluticasone group); or quintupling the dose of fluticasone to 250 mcg and adding 50 mcg of salmeterol (the salmeterol/quintuple-fluticasone group). The trial consisted of a four-way crossover design with each treatment period lasting 14 weeks.
The primary outcome was a composite measure including asthma exacerbations, asthma control days, and percentage of predicted forced expiratory volume in the first second at the end of each treatment.
Overall, a superior response occurred in 53% of the salmeterol/double-fluticasone group, 41% of the double-fluticasone group, 43% of the salmeterol/quintuple fluticasone group, and 47% of the quintuple-fluticasone group.
The superior response was 46% for both groups when the researchers compared a quintupled dose of fluticasone propionate (250 mcg) with a two step–up strategy of adding salmeterol at a dose of 50 mcg and increasing the dose of fluticasone to 100 mcg.
“In contrast to black adults and white persons of all ages, almost half the children who had at least one grandparent who identified as black and who had poorly controlled asthma had a superior response to an increased dose of an inhaled glucocorticoid over the addition of a LABA,” Dr. Wechsler and coauthors wrote. No more than 12% of the children in any treatment group did not have a superior response. No significant differences in reports of respiratory tract infections or pneumonia were seen between the groups. Children younger than 8 years showed a decrease in the ratio of urinary cortisol to creatinine with an increased dose of inhaled glucocorticoids.
In the adolescent and adult study using the same treatment protocols, 20%-25% of patients did not have a differential outcome between treatments. “In adolescents and adults, the addition of a LABA was more likely to produce superior responses than increasing the dose of an inhaled glucocorticoid,” Dr. Wechsler and coauthors wrote.
The study findings were limited by several factors, including the inability to assess long-term effects on growth and inability to detect biomarkers associated with responses to specific therapies, the researchers noted. However, the results suggest that black children with poorly controlled asthma can benefit from additional inhaled glucocorticoids, and larger studies are needed to identify the best treatment for this patient population.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Wechsler reported relationships with companies including AstraZeneca, Equillium, Genentech, GlaxoSmithKline, Mylan, Novartis, Regeneron, resTORbio, Sanofi, and others. Coauthors identified relationships with numerous pharmaceutical companies.
SOURCE: Wechsler ME et al. N Engl J Med. 2019 Sep 25. doi: 10.1056/NEJMoa1905560.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Black children with poorly controlled asthma had a superior response to stepped increases in inhaled glucocorticoids, compared with stepped increases in long-acting beta2-agonists.
Major finding: A total of 46% of black children with poorly controlled asthma improved when inhaled glucocorticoids were increased.
Study details: The data come from the BARD trials, a pair of prospective, randomized, double-blind trials including 280 children aged 5-11 years with at least one grandparent identified as black and 294 adolescents and adults who had family backgrounds that were similar to the children.
Disclosures: The study was supported by the National Heart, Lung, and Blood Institute. Dr. Wechsler reported relationships with companies including AstraZeneca, Equillium, Genentech, GlaxoSmithKline, Mylan, Novartis, Regeneron, resTORbio, Sanofi, and others. Coauthors identified relationships with numerous pharmaceutical companies.
Source: Wechsler ME et al. N Engl J Med. 2019 Sep 25. doi: 10.1056/NEJMoa1905560.
Acting FDA commissioner issues remarks on ENDS, vaping illnesses
Norman E. Sharpless, MD, acting commissioner of the Food and Drug Administration, has issued remarks that were prepared for a testimony before a U.S. House Energy and Commerce subcommittee on FDA regulation of electronic nicotine delivery systems and investigation of vaping illnesses.
Dr. Sharpless’s statement focuses on two priorities: the continuing investigation into the cause of lung injury associated with the use of vaping products and the FDA’s ongoing efforts to address an epidemic of youth use of electronic nicotine delivery systems (ENDS). In regards to lung injuries associated with vaping, Dr. Sharpless said that the FDA and Centers for Disease Control and Prevention, in conjunction with state partners, have been investigating the outbreak and noted that, while most cases have involved tetrahydrocannabinol, there is as of yet no common cause or product linked to all cases.
The FDA is not currently pursuing any legal action against personal usage of vaping products, but “if we determine that someone is manufacturing or distributing illicit, adulterated vaping products that caused illness and death for personal profit, we would consider that to be a criminal act,” Dr. Sharpless said.
Research from the 2018 and 2019 National Youth Tobacco Surveys has indicated that ENDS usage among youth has risen dramatically in recent years, Dr. Sharpless continued in the statement. The FDA has pursued several courses of action, including the issue of a warning letter to Juul for marketing unauthorized modified-risk tobacco products to children.
However, he noted, youth e-cigarette use continues to rise, which is the reason for the FDA’s intention to finalize a compliance policy related to flavored ENDS, a policy supported by President TRump.
“FDA is not ‘banning’ flavors, as has been described in some news reports. Rather, FDA intends to enforce existing law that limits the marketing of such products,” Dr. Sharpless said. “This policy would not mean that flavored e-cigarettes could never be marketed. If a company can show through an application to FDA that a specific product meets the standard set forth by Congress, then the FDA would authorize that ENDS product for sale.”
Find the full statement on the FDA website.
Norman E. Sharpless, MD, acting commissioner of the Food and Drug Administration, has issued remarks that were prepared for a testimony before a U.S. House Energy and Commerce subcommittee on FDA regulation of electronic nicotine delivery systems and investigation of vaping illnesses.
Dr. Sharpless’s statement focuses on two priorities: the continuing investigation into the cause of lung injury associated with the use of vaping products and the FDA’s ongoing efforts to address an epidemic of youth use of electronic nicotine delivery systems (ENDS). In regards to lung injuries associated with vaping, Dr. Sharpless said that the FDA and Centers for Disease Control and Prevention, in conjunction with state partners, have been investigating the outbreak and noted that, while most cases have involved tetrahydrocannabinol, there is as of yet no common cause or product linked to all cases.
The FDA is not currently pursuing any legal action against personal usage of vaping products, but “if we determine that someone is manufacturing or distributing illicit, adulterated vaping products that caused illness and death for personal profit, we would consider that to be a criminal act,” Dr. Sharpless said.
Research from the 2018 and 2019 National Youth Tobacco Surveys has indicated that ENDS usage among youth has risen dramatically in recent years, Dr. Sharpless continued in the statement. The FDA has pursued several courses of action, including the issue of a warning letter to Juul for marketing unauthorized modified-risk tobacco products to children.
However, he noted, youth e-cigarette use continues to rise, which is the reason for the FDA’s intention to finalize a compliance policy related to flavored ENDS, a policy supported by President TRump.
“FDA is not ‘banning’ flavors, as has been described in some news reports. Rather, FDA intends to enforce existing law that limits the marketing of such products,” Dr. Sharpless said. “This policy would not mean that flavored e-cigarettes could never be marketed. If a company can show through an application to FDA that a specific product meets the standard set forth by Congress, then the FDA would authorize that ENDS product for sale.”
Find the full statement on the FDA website.
Norman E. Sharpless, MD, acting commissioner of the Food and Drug Administration, has issued remarks that were prepared for a testimony before a U.S. House Energy and Commerce subcommittee on FDA regulation of electronic nicotine delivery systems and investigation of vaping illnesses.
Dr. Sharpless’s statement focuses on two priorities: the continuing investigation into the cause of lung injury associated with the use of vaping products and the FDA’s ongoing efforts to address an epidemic of youth use of electronic nicotine delivery systems (ENDS). In regards to lung injuries associated with vaping, Dr. Sharpless said that the FDA and Centers for Disease Control and Prevention, in conjunction with state partners, have been investigating the outbreak and noted that, while most cases have involved tetrahydrocannabinol, there is as of yet no common cause or product linked to all cases.
The FDA is not currently pursuing any legal action against personal usage of vaping products, but “if we determine that someone is manufacturing or distributing illicit, adulterated vaping products that caused illness and death for personal profit, we would consider that to be a criminal act,” Dr. Sharpless said.
Research from the 2018 and 2019 National Youth Tobacco Surveys has indicated that ENDS usage among youth has risen dramatically in recent years, Dr. Sharpless continued in the statement. The FDA has pursued several courses of action, including the issue of a warning letter to Juul for marketing unauthorized modified-risk tobacco products to children.
However, he noted, youth e-cigarette use continues to rise, which is the reason for the FDA’s intention to finalize a compliance policy related to flavored ENDS, a policy supported by President TRump.
“FDA is not ‘banning’ flavors, as has been described in some news reports. Rather, FDA intends to enforce existing law that limits the marketing of such products,” Dr. Sharpless said. “This policy would not mean that flavored e-cigarettes could never be marketed. If a company can show through an application to FDA that a specific product meets the standard set forth by Congress, then the FDA would authorize that ENDS product for sale.”
Find the full statement on the FDA website.
PRAGMA-CF shows disease progression of cystic fibrosis in children
reported Nynke R. Bouma, BSc, and colleagues.
“Even though bronchiectasis is present in 60% to 80% of children with CF in school age, the extent and severity of bronchiectasis in preschool children are generally lower ... however, diffuse airway abnormalities such as airway wall thickening and mucus plugging are observed in many preschool children. It is hypothesized that these preschool airway changes reflect diffuse airway disease that eventually will result in bronchiectasis in school age,” they noted.
The PRAGMA-CF image scoring system can measure airway disease and can also be used to monitor disease progression, noted Ms. Bouma of Sophia Children’s Hospital, Rotterdam, and colleagues. The study was published in Pediatric Pulmonology. PRAGMA-CF is a composite score of airway wall thickening, mucus plugging, and bronchiectasis as percent disease (%disease). “In preschool children, %disease measured by PRAGMA-CF on chest CT allows quantification of early clinically relevant morphological features of CF airway disease and it is associated with later school-age bronchiectasis,” the team wrote. “These findings support the use of %disease as a clinically relevant outcome measure in early CF lung disease.”
The team conducted a prospective cohort study of 61 children (mean age 4 years) with cystic fibrosis, following them for a mean of 5 years. A total of 122 CT scans were available from this group, in addition to spirometry data and cystic fibrosis quality of life scores.
From preschool age to school age, the %disease on PRAGMA-CF increased significantly, from a mean of 0.7% to 1.73%. Scores on another composite measuring tool (%MUPAT, a composite score of airway wall thickening and mucus plugging) went from 0.46 to 0.58 – not a significant difference.
A multivariate analysis corrected for age in each school group and the type of scanner used to acquire the images. That analysis determined that each 1% increase in %disease at preschool age resulted in an increase of 1.18% of bronchiectasis at school age.
A cross-sectional analysis of the group at school age found significant associations between the %disease and percent of forced expiratory volume and the cystic fibrosis quality of life score.
At least one pulmonary exacerbation requiring intravenous antibiotics occurred in 19 of the patients. However, the investigators didn’t find any significant interactions between the %disease in preschool and these exacerbations..
“These findings are in line with previous studies in school‐aged children that showed that mucus plugging is associated with inflammation and airway wall thickening, and that these are thought to be risk factors for later bronchiectasis,” they concluded. “On the basis of our findings, we suggest that %disease and %MUPAT could be used as a clinically relevant outcome measure in clinical studies in preschool patients with cystic fibrosis, as these measures predict later bronchiectasis. Percent disease may be preferred as it captures all the principal features of CF airways disease including bronchiectasis.”
Ms. Bouma had no financial disclosures.
SOURCE: Bouma NR et al. Pediatr Pulmonol. 2019 Sep 9 doi: 10.1002/ppul.24498; Rosenow et al. Am J Respir Crit Care Med. 2015 May 15. doi: 10.1164/rccm.201501-0061OC.
reported Nynke R. Bouma, BSc, and colleagues.
“Even though bronchiectasis is present in 60% to 80% of children with CF in school age, the extent and severity of bronchiectasis in preschool children are generally lower ... however, diffuse airway abnormalities such as airway wall thickening and mucus plugging are observed in many preschool children. It is hypothesized that these preschool airway changes reflect diffuse airway disease that eventually will result in bronchiectasis in school age,” they noted.
The PRAGMA-CF image scoring system can measure airway disease and can also be used to monitor disease progression, noted Ms. Bouma of Sophia Children’s Hospital, Rotterdam, and colleagues. The study was published in Pediatric Pulmonology. PRAGMA-CF is a composite score of airway wall thickening, mucus plugging, and bronchiectasis as percent disease (%disease). “In preschool children, %disease measured by PRAGMA-CF on chest CT allows quantification of early clinically relevant morphological features of CF airway disease and it is associated with later school-age bronchiectasis,” the team wrote. “These findings support the use of %disease as a clinically relevant outcome measure in early CF lung disease.”
The team conducted a prospective cohort study of 61 children (mean age 4 years) with cystic fibrosis, following them for a mean of 5 years. A total of 122 CT scans were available from this group, in addition to spirometry data and cystic fibrosis quality of life scores.
From preschool age to school age, the %disease on PRAGMA-CF increased significantly, from a mean of 0.7% to 1.73%. Scores on another composite measuring tool (%MUPAT, a composite score of airway wall thickening and mucus plugging) went from 0.46 to 0.58 – not a significant difference.
A multivariate analysis corrected for age in each school group and the type of scanner used to acquire the images. That analysis determined that each 1% increase in %disease at preschool age resulted in an increase of 1.18% of bronchiectasis at school age.
A cross-sectional analysis of the group at school age found significant associations between the %disease and percent of forced expiratory volume and the cystic fibrosis quality of life score.
At least one pulmonary exacerbation requiring intravenous antibiotics occurred in 19 of the patients. However, the investigators didn’t find any significant interactions between the %disease in preschool and these exacerbations..
“These findings are in line with previous studies in school‐aged children that showed that mucus plugging is associated with inflammation and airway wall thickening, and that these are thought to be risk factors for later bronchiectasis,” they concluded. “On the basis of our findings, we suggest that %disease and %MUPAT could be used as a clinically relevant outcome measure in clinical studies in preschool patients with cystic fibrosis, as these measures predict later bronchiectasis. Percent disease may be preferred as it captures all the principal features of CF airways disease including bronchiectasis.”
Ms. Bouma had no financial disclosures.
SOURCE: Bouma NR et al. Pediatr Pulmonol. 2019 Sep 9 doi: 10.1002/ppul.24498; Rosenow et al. Am J Respir Crit Care Med. 2015 May 15. doi: 10.1164/rccm.201501-0061OC.
reported Nynke R. Bouma, BSc, and colleagues.
“Even though bronchiectasis is present in 60% to 80% of children with CF in school age, the extent and severity of bronchiectasis in preschool children are generally lower ... however, diffuse airway abnormalities such as airway wall thickening and mucus plugging are observed in many preschool children. It is hypothesized that these preschool airway changes reflect diffuse airway disease that eventually will result in bronchiectasis in school age,” they noted.
The PRAGMA-CF image scoring system can measure airway disease and can also be used to monitor disease progression, noted Ms. Bouma of Sophia Children’s Hospital, Rotterdam, and colleagues. The study was published in Pediatric Pulmonology. PRAGMA-CF is a composite score of airway wall thickening, mucus plugging, and bronchiectasis as percent disease (%disease). “In preschool children, %disease measured by PRAGMA-CF on chest CT allows quantification of early clinically relevant morphological features of CF airway disease and it is associated with later school-age bronchiectasis,” the team wrote. “These findings support the use of %disease as a clinically relevant outcome measure in early CF lung disease.”
The team conducted a prospective cohort study of 61 children (mean age 4 years) with cystic fibrosis, following them for a mean of 5 years. A total of 122 CT scans were available from this group, in addition to spirometry data and cystic fibrosis quality of life scores.
From preschool age to school age, the %disease on PRAGMA-CF increased significantly, from a mean of 0.7% to 1.73%. Scores on another composite measuring tool (%MUPAT, a composite score of airway wall thickening and mucus plugging) went from 0.46 to 0.58 – not a significant difference.
A multivariate analysis corrected for age in each school group and the type of scanner used to acquire the images. That analysis determined that each 1% increase in %disease at preschool age resulted in an increase of 1.18% of bronchiectasis at school age.
A cross-sectional analysis of the group at school age found significant associations between the %disease and percent of forced expiratory volume and the cystic fibrosis quality of life score.
At least one pulmonary exacerbation requiring intravenous antibiotics occurred in 19 of the patients. However, the investigators didn’t find any significant interactions between the %disease in preschool and these exacerbations..
“These findings are in line with previous studies in school‐aged children that showed that mucus plugging is associated with inflammation and airway wall thickening, and that these are thought to be risk factors for later bronchiectasis,” they concluded. “On the basis of our findings, we suggest that %disease and %MUPAT could be used as a clinically relevant outcome measure in clinical studies in preschool patients with cystic fibrosis, as these measures predict later bronchiectasis. Percent disease may be preferred as it captures all the principal features of CF airways disease including bronchiectasis.”
Ms. Bouma had no financial disclosures.
SOURCE: Bouma NR et al. Pediatr Pulmonol. 2019 Sep 9 doi: 10.1002/ppul.24498; Rosenow et al. Am J Respir Crit Care Med. 2015 May 15. doi: 10.1164/rccm.201501-0061OC.
FROM PEDIATRIC PULMONOLOGY
Many institutions exceed recommended radiation doses during lung cancer screening
according to a study published in JAMA Internal Medicine.
Various institutional characteristics, such as allowing any radiologist to establish CT scan protocols, are associated with a greater likelihood of using higher radiation doses. “Dose optimization practices may benefit from being tailored to specific practice types, as well as different organizational structures, to have a higher likelihood of meeting dose guidelines,” wrote Joshua Demb, PhD, MPH, a cancer epidemiologist at the University of California, San Diego, and colleagues.
Lung cancer screening benefits patients when low-dose CT is used, but not when high-dose CT is used, because radiation from higher doses may cause as many cancers as are detected by screening. The Centers for Medicare & Medicaid Services require institutions to use low-dose techniques and participate in a dose registry to be reimbursed for lung cancer screening. The American College of Radiology recommends that lung cancer screening scans have a volume CT dose index (CTDIvol) of 3 mGy or lower and an effective dose (ED) of 1 millisieverts (mSv) or lower.
A prospective study of registry data
Dr. Demb and colleagues conducted a study to describe CT radiation doses for lung cancer screening in current practice and to identify the factors that explain variation in doses between institutions. They prospectively collected lung cancer screening examination dose metrics from 2016 to 2017 at U.S. institutions participating in the University of California, San Francisco, International Dose Registry. Eligible institutions performed a minimum of 24 lung cancer screening scans during the study period. At baseline, the investigators surveyed institutions about their characteristics (for example, how they perform and oversee CT). Dr. Demb and colleagues estimated mixed-effects linear and logistic regression models using forward variable selection. They conducted their analysis between 2018 and 2019.
The researchers chose four outcome measures. The first was mean CTDIvol, reflecting the average radiation dose per slice. The second was mean ED, reflecting the total dose received and estimated future cancer risk. The third was the proportion of CT scans using radiation doses above ACR benchmarks. The fourth was the proportion of CT scans using radiation doses above the 75th percentile of registry doses (CTDIvol greater than 2.7 mGy and ED greater than 1.4 mSv).
Institutional characteristics associated with radiation dose
Dr. Demb and colleagues collected data from 72 institutions about 12,529 patients undergoing CT scans for lung cancer screening. Approximately 58% of patients were men, and the patients’ median age was 65 years. The mean CTDIvol, adjusted for patient size, was 2.4 mGy. The mean ED for lung cancer screening, adjusted for chest diameter, was 1.2 mSv.
A total of 15 institutions (21%) had a median adjusted CTDIvol value higher than the ACR guideline, and 47 (65%) had a median adjusted ED higher than the ACR guideline. Approximately 18% of CT scans had a CTDIvol higher than guidelines, and 50% had an ED higher than ACR guidelines.
Institutions that permitted any radiologist to establish CT protocols had 44% higher mean CTDIvol and 27% higher mean ED, compared with institutions that restricted who could establish protocols. Institutions that permitted any radiologist to establish protocols also had higher odds of conducting examinations that exceeded ACR CTDIvol guidelines (odds ratio, 12.0) and of being in the 75th percentile of the registry CTDIvol (OR, 19.0) or ED (OR, 8.5) values.
In contrast, having lead radiologists establish CT protocols resulted in lower odds of using doses that exceeded ACR ED guidelines (OR, 0.01). Employing external, rather than internal, medical physicists was associated with increased odds of exceeding ACR CTDIvol guidelines (OR, 6.1). Having medical physicists establish protocols was associated with decreased odds of exceeding the 75th percentile of the registry CTDIvol (OR, 0.09) values. Institutions that updated protocols as needed, rather than annually, had 27% higher mean CTDIvol.
“Although we cannot establish causality in this observational study, our results suggest that considering these factors (for example, allowing only lead radiologists to establish protocols) could have a meaningful impact on dose, and could be important areas to develop interventions to optimize doses of CT protocols” the investigators wrote.
The Patient Centered Outcomes Research Institute and the National Institutes of Health supported this research. The authors reported no conflicts of interest.
SOURCE: Demb J et al. JAMA Intern Med. 2019 Sep 23. doi: 10.1001/jamainternmed.2019.3893.
according to a study published in JAMA Internal Medicine.
Various institutional characteristics, such as allowing any radiologist to establish CT scan protocols, are associated with a greater likelihood of using higher radiation doses. “Dose optimization practices may benefit from being tailored to specific practice types, as well as different organizational structures, to have a higher likelihood of meeting dose guidelines,” wrote Joshua Demb, PhD, MPH, a cancer epidemiologist at the University of California, San Diego, and colleagues.
Lung cancer screening benefits patients when low-dose CT is used, but not when high-dose CT is used, because radiation from higher doses may cause as many cancers as are detected by screening. The Centers for Medicare & Medicaid Services require institutions to use low-dose techniques and participate in a dose registry to be reimbursed for lung cancer screening. The American College of Radiology recommends that lung cancer screening scans have a volume CT dose index (CTDIvol) of 3 mGy or lower and an effective dose (ED) of 1 millisieverts (mSv) or lower.
A prospective study of registry data
Dr. Demb and colleagues conducted a study to describe CT radiation doses for lung cancer screening in current practice and to identify the factors that explain variation in doses between institutions. They prospectively collected lung cancer screening examination dose metrics from 2016 to 2017 at U.S. institutions participating in the University of California, San Francisco, International Dose Registry. Eligible institutions performed a minimum of 24 lung cancer screening scans during the study period. At baseline, the investigators surveyed institutions about their characteristics (for example, how they perform and oversee CT). Dr. Demb and colleagues estimated mixed-effects linear and logistic regression models using forward variable selection. They conducted their analysis between 2018 and 2019.
The researchers chose four outcome measures. The first was mean CTDIvol, reflecting the average radiation dose per slice. The second was mean ED, reflecting the total dose received and estimated future cancer risk. The third was the proportion of CT scans using radiation doses above ACR benchmarks. The fourth was the proportion of CT scans using radiation doses above the 75th percentile of registry doses (CTDIvol greater than 2.7 mGy and ED greater than 1.4 mSv).
Institutional characteristics associated with radiation dose
Dr. Demb and colleagues collected data from 72 institutions about 12,529 patients undergoing CT scans for lung cancer screening. Approximately 58% of patients were men, and the patients’ median age was 65 years. The mean CTDIvol, adjusted for patient size, was 2.4 mGy. The mean ED for lung cancer screening, adjusted for chest diameter, was 1.2 mSv.
A total of 15 institutions (21%) had a median adjusted CTDIvol value higher than the ACR guideline, and 47 (65%) had a median adjusted ED higher than the ACR guideline. Approximately 18% of CT scans had a CTDIvol higher than guidelines, and 50% had an ED higher than ACR guidelines.
Institutions that permitted any radiologist to establish CT protocols had 44% higher mean CTDIvol and 27% higher mean ED, compared with institutions that restricted who could establish protocols. Institutions that permitted any radiologist to establish protocols also had higher odds of conducting examinations that exceeded ACR CTDIvol guidelines (odds ratio, 12.0) and of being in the 75th percentile of the registry CTDIvol (OR, 19.0) or ED (OR, 8.5) values.
In contrast, having lead radiologists establish CT protocols resulted in lower odds of using doses that exceeded ACR ED guidelines (OR, 0.01). Employing external, rather than internal, medical physicists was associated with increased odds of exceeding ACR CTDIvol guidelines (OR, 6.1). Having medical physicists establish protocols was associated with decreased odds of exceeding the 75th percentile of the registry CTDIvol (OR, 0.09) values. Institutions that updated protocols as needed, rather than annually, had 27% higher mean CTDIvol.
“Although we cannot establish causality in this observational study, our results suggest that considering these factors (for example, allowing only lead radiologists to establish protocols) could have a meaningful impact on dose, and could be important areas to develop interventions to optimize doses of CT protocols” the investigators wrote.
The Patient Centered Outcomes Research Institute and the National Institutes of Health supported this research. The authors reported no conflicts of interest.
SOURCE: Demb J et al. JAMA Intern Med. 2019 Sep 23. doi: 10.1001/jamainternmed.2019.3893.
according to a study published in JAMA Internal Medicine.
Various institutional characteristics, such as allowing any radiologist to establish CT scan protocols, are associated with a greater likelihood of using higher radiation doses. “Dose optimization practices may benefit from being tailored to specific practice types, as well as different organizational structures, to have a higher likelihood of meeting dose guidelines,” wrote Joshua Demb, PhD, MPH, a cancer epidemiologist at the University of California, San Diego, and colleagues.
Lung cancer screening benefits patients when low-dose CT is used, but not when high-dose CT is used, because radiation from higher doses may cause as many cancers as are detected by screening. The Centers for Medicare & Medicaid Services require institutions to use low-dose techniques and participate in a dose registry to be reimbursed for lung cancer screening. The American College of Radiology recommends that lung cancer screening scans have a volume CT dose index (CTDIvol) of 3 mGy or lower and an effective dose (ED) of 1 millisieverts (mSv) or lower.
A prospective study of registry data
Dr. Demb and colleagues conducted a study to describe CT radiation doses for lung cancer screening in current practice and to identify the factors that explain variation in doses between institutions. They prospectively collected lung cancer screening examination dose metrics from 2016 to 2017 at U.S. institutions participating in the University of California, San Francisco, International Dose Registry. Eligible institutions performed a minimum of 24 lung cancer screening scans during the study period. At baseline, the investigators surveyed institutions about their characteristics (for example, how they perform and oversee CT). Dr. Demb and colleagues estimated mixed-effects linear and logistic regression models using forward variable selection. They conducted their analysis between 2018 and 2019.
The researchers chose four outcome measures. The first was mean CTDIvol, reflecting the average radiation dose per slice. The second was mean ED, reflecting the total dose received and estimated future cancer risk. The third was the proportion of CT scans using radiation doses above ACR benchmarks. The fourth was the proportion of CT scans using radiation doses above the 75th percentile of registry doses (CTDIvol greater than 2.7 mGy and ED greater than 1.4 mSv).
Institutional characteristics associated with radiation dose
Dr. Demb and colleagues collected data from 72 institutions about 12,529 patients undergoing CT scans for lung cancer screening. Approximately 58% of patients were men, and the patients’ median age was 65 years. The mean CTDIvol, adjusted for patient size, was 2.4 mGy. The mean ED for lung cancer screening, adjusted for chest diameter, was 1.2 mSv.
A total of 15 institutions (21%) had a median adjusted CTDIvol value higher than the ACR guideline, and 47 (65%) had a median adjusted ED higher than the ACR guideline. Approximately 18% of CT scans had a CTDIvol higher than guidelines, and 50% had an ED higher than ACR guidelines.
Institutions that permitted any radiologist to establish CT protocols had 44% higher mean CTDIvol and 27% higher mean ED, compared with institutions that restricted who could establish protocols. Institutions that permitted any radiologist to establish protocols also had higher odds of conducting examinations that exceeded ACR CTDIvol guidelines (odds ratio, 12.0) and of being in the 75th percentile of the registry CTDIvol (OR, 19.0) or ED (OR, 8.5) values.
In contrast, having lead radiologists establish CT protocols resulted in lower odds of using doses that exceeded ACR ED guidelines (OR, 0.01). Employing external, rather than internal, medical physicists was associated with increased odds of exceeding ACR CTDIvol guidelines (OR, 6.1). Having medical physicists establish protocols was associated with decreased odds of exceeding the 75th percentile of the registry CTDIvol (OR, 0.09) values. Institutions that updated protocols as needed, rather than annually, had 27% higher mean CTDIvol.
“Although we cannot establish causality in this observational study, our results suggest that considering these factors (for example, allowing only lead radiologists to establish protocols) could have a meaningful impact on dose, and could be important areas to develop interventions to optimize doses of CT protocols” the investigators wrote.
The Patient Centered Outcomes Research Institute and the National Institutes of Health supported this research. The authors reported no conflicts of interest.
SOURCE: Demb J et al. JAMA Intern Med. 2019 Sep 23. doi: 10.1001/jamainternmed.2019.3893.
FROM JAMA INTERNAL MEDICINE
Key clinical point: A significant proportion of institutions exceed guideline-recommended dose levels for CT screening for lung cancer.
Major finding: About 21% of institutions have median volume CT dose index above American College of Radiology guidelines, and 65% have median effective dose above ACR guidelines.
Study details: A prospective study of data for 12,529 patients undergoing screening at 72 institutions.
Disclosures: The Patient Centered Outcomes Research Institute and the National Institutes of Health supported this research. The authors reported no conflicts of interest.
Source: Demb J et al. JAMA Intern Med. 2019 Sep 23. doi: 10.1001/jamainternmed.2019.3893.
New guideline conditionally recommends long-term home NIV for COPD patients
from a European Respiratory Society task force.
“Our recommendations, based on the best available evidence, can guide the management of chronic hypercapnic respiratory failure in COPD patients aimed at improving patient outcomes,” wrote Begum Ergan, MD, of Dokuz Eylul University, Izmir, Turkey, and coauthors. The guideline was published in the European Respiratory Journal.
To provide insight into the clinical application of LTH-NIV, the European Respiratory Society convened a task force of 20 clinicians, methodologists, and experts. Their four recommendations were developed based on the GRADE (Grading, Recommendation, Assessment, Development and Evaluation) methodology.
The first recommendation was to use LTH-NIV for patients with chronic stable hypercapnic COPD. Though an analysis of randomized, controlled trials showed little effect on mortality or hospitalizations, pooled analyses showed that NIV may decrease dyspnea scores (standardized mean difference, –0.51; 95% confidence interval, –0.06 to –0.95) and increase health-related quality of life (SMD, 0.49; 95% CI, –0.01 to 0.98).
The second was to use LTH-NIV in patients with COPD following a life-threatening episode of acute hypercapnic respiratory failure requiring acute NIV, if hypercapnia persists. Though it was not associated with a reduction in mortality (risk ratio, 0.92; 95% CI, 0.67-1.25), it was found to potentially reduce exacerbations (SMD, 0.19; 95% CI, –0.40 to 0.01) and hospitalizations (RR, 0.61; 95% CI, 0.30-1.24).
The third was to titrate LTH-NIV to normalize or reduce PaCO2 levels in patients with COPD. While this recommendation was issued with a very low certainty of evidence, it was driven by the “minimal potential harms of targeted PaCO2 reduction.”
The fourth was to use fixed pressure support mode as first-choice ventilator mode in patients with COPD using LTH-NIV. The six trials on this subject did not provide insight into long-term outcomes, nor were there significant improvements seen in health-related quality of life, sleep quality, or exercise tolerance. As such, it was also issued with a very low certainty of evidence.
The authors acknowledged all four recommendations as weak and conditional, “due to limitations in the certainty of the available evidence.” As such, they noted that their recommendations “require consideration of individual preferences, resource considerations, technical expertise, and clinical circumstances prior to implementation in clinical practice.”
The authors reported numerous disclosures, including receiving grants and personal fees from various medical supply companies.
SOURCE: Ergan B et al. Eur Respir J. 2019 Aug 29. doi: 10.1183/13993003.01003-2019.
from a European Respiratory Society task force.
“Our recommendations, based on the best available evidence, can guide the management of chronic hypercapnic respiratory failure in COPD patients aimed at improving patient outcomes,” wrote Begum Ergan, MD, of Dokuz Eylul University, Izmir, Turkey, and coauthors. The guideline was published in the European Respiratory Journal.
To provide insight into the clinical application of LTH-NIV, the European Respiratory Society convened a task force of 20 clinicians, methodologists, and experts. Their four recommendations were developed based on the GRADE (Grading, Recommendation, Assessment, Development and Evaluation) methodology.
The first recommendation was to use LTH-NIV for patients with chronic stable hypercapnic COPD. Though an analysis of randomized, controlled trials showed little effect on mortality or hospitalizations, pooled analyses showed that NIV may decrease dyspnea scores (standardized mean difference, –0.51; 95% confidence interval, –0.06 to –0.95) and increase health-related quality of life (SMD, 0.49; 95% CI, –0.01 to 0.98).
The second was to use LTH-NIV in patients with COPD following a life-threatening episode of acute hypercapnic respiratory failure requiring acute NIV, if hypercapnia persists. Though it was not associated with a reduction in mortality (risk ratio, 0.92; 95% CI, 0.67-1.25), it was found to potentially reduce exacerbations (SMD, 0.19; 95% CI, –0.40 to 0.01) and hospitalizations (RR, 0.61; 95% CI, 0.30-1.24).
The third was to titrate LTH-NIV to normalize or reduce PaCO2 levels in patients with COPD. While this recommendation was issued with a very low certainty of evidence, it was driven by the “minimal potential harms of targeted PaCO2 reduction.”
The fourth was to use fixed pressure support mode as first-choice ventilator mode in patients with COPD using LTH-NIV. The six trials on this subject did not provide insight into long-term outcomes, nor were there significant improvements seen in health-related quality of life, sleep quality, or exercise tolerance. As such, it was also issued with a very low certainty of evidence.
The authors acknowledged all four recommendations as weak and conditional, “due to limitations in the certainty of the available evidence.” As such, they noted that their recommendations “require consideration of individual preferences, resource considerations, technical expertise, and clinical circumstances prior to implementation in clinical practice.”
The authors reported numerous disclosures, including receiving grants and personal fees from various medical supply companies.
SOURCE: Ergan B et al. Eur Respir J. 2019 Aug 29. doi: 10.1183/13993003.01003-2019.
from a European Respiratory Society task force.
“Our recommendations, based on the best available evidence, can guide the management of chronic hypercapnic respiratory failure in COPD patients aimed at improving patient outcomes,” wrote Begum Ergan, MD, of Dokuz Eylul University, Izmir, Turkey, and coauthors. The guideline was published in the European Respiratory Journal.
To provide insight into the clinical application of LTH-NIV, the European Respiratory Society convened a task force of 20 clinicians, methodologists, and experts. Their four recommendations were developed based on the GRADE (Grading, Recommendation, Assessment, Development and Evaluation) methodology.
The first recommendation was to use LTH-NIV for patients with chronic stable hypercapnic COPD. Though an analysis of randomized, controlled trials showed little effect on mortality or hospitalizations, pooled analyses showed that NIV may decrease dyspnea scores (standardized mean difference, –0.51; 95% confidence interval, –0.06 to –0.95) and increase health-related quality of life (SMD, 0.49; 95% CI, –0.01 to 0.98).
The second was to use LTH-NIV in patients with COPD following a life-threatening episode of acute hypercapnic respiratory failure requiring acute NIV, if hypercapnia persists. Though it was not associated with a reduction in mortality (risk ratio, 0.92; 95% CI, 0.67-1.25), it was found to potentially reduce exacerbations (SMD, 0.19; 95% CI, –0.40 to 0.01) and hospitalizations (RR, 0.61; 95% CI, 0.30-1.24).
The third was to titrate LTH-NIV to normalize or reduce PaCO2 levels in patients with COPD. While this recommendation was issued with a very low certainty of evidence, it was driven by the “minimal potential harms of targeted PaCO2 reduction.”
The fourth was to use fixed pressure support mode as first-choice ventilator mode in patients with COPD using LTH-NIV. The six trials on this subject did not provide insight into long-term outcomes, nor were there significant improvements seen in health-related quality of life, sleep quality, or exercise tolerance. As such, it was also issued with a very low certainty of evidence.
The authors acknowledged all four recommendations as weak and conditional, “due to limitations in the certainty of the available evidence.” As such, they noted that their recommendations “require consideration of individual preferences, resource considerations, technical expertise, and clinical circumstances prior to implementation in clinical practice.”
The authors reported numerous disclosures, including receiving grants and personal fees from various medical supply companies.
SOURCE: Ergan B et al. Eur Respir J. 2019 Aug 29. doi: 10.1183/13993003.01003-2019.
FROM THE EUROPEAN RESPIRATORY JOURNAL
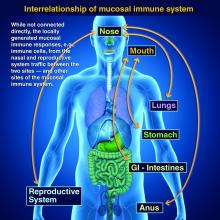
Taking vaccines to the next level via mucosal immunity
Vaccines are marvelous, and there are many well documented success stories, including rotavirus (RV) vaccines, where a live vaccine is administered to the gastrointestinal mucosa via oral drops. Antigens presented at the mucosal/epithelial surface not only induce systemic serum IgG – as do injectable vaccines – but also induce secretory IgA (sIgA), which is most helpful in diseases that directly affect the mucosa.

Mucosal vs. systemic immunity
Antibody being present on mucosal surfaces (point of initial pathogen contact) has a chance to neutralize the pathogen before it gains a foothold. Pathogen-specific mucosal lymphoid elements (e.g. in Peyer’s patches in the gut) also appear critical for optimal protection.1 The presence of both mucosal immune elements means that infection is severely limited or at times entirely prevented. So virus entering the GI tract causes minimal to no gut lining injury. Hence, there is no or mostly reduced vomiting/diarrhea. A downside of mucosally-administered live vaccines is that preexisting antibody to the vaccine antigens can reduce or block vaccine virus replication in the vaccinee, blunting or preventing protection. Note: Preexisting antibody also affects injectable live vaccines, such as the measles vaccine, similarly.
Classic injectable live or nonlive vaccines provide their most potent protection via systemic cellular responses antibody and/or antibodies in serum and extracellular fluid (ECF) where IgG and IgM are in highest concentrations. So even successful injectable vaccines still allow mucosal infection to start but then intercept further spread and prevent most of the downstream damage (think pertussis) or neutralize an infection-generated toxin (pertussis or tetanus). It usually is only after infection-induced damage occurs that systemic IgG and IgM gain better access to respiratory epithelial surfaces, but still only at a fraction of circulating concentrations. Indeed, pertussis vaccine–induced systemic immunity allows the pathogen to attack and replicate in/on host surface cells, causing toxin release and variable amounts of local mucosal injury/inflammation before vaccine-induced systemic immunity gains adequate access to the pathogen and/or to its toxin which may enter systemic circulation.
Live attenuated influenza vaccine (LAIV) induces mucosal immunity
Another “standard” vaccine that induces mucosal immunity – LAIV – was developed to improve on protection afforded by injectable influenza vaccines (IIVs), but LAIV has had hiccups in the United States. One example is several years of negligible protection against H1N1 disease. As long as LAIV’s vaccine strain had reasonably matched the circulating strains, LAIV worked at least as well as injectable influenza vaccine, and even offered some cross-protection against mildly mismatched strains. But after a number of years of LAIV use, vaccine effectiveness in the United States vs. H1N1 strains appeared to fade due to previously undetected but significant changes in the circulating H1N1 strain. The lesson is that mucosal immunity’s advantages are lost if too much change occurs in the pathogen target for sIgA and mucosally-associated lymphoid tissue cells (MALT)).
Other vaccines likely need to induce mucosal immunity
Protection at the mucosal level will likely be needed for success against norovirus, parainfluenza, respiratory syncytial virus (RSV), Neisseria gonorrhea, and chlamydia. Another helpful aspect of mucosal immunity is that immune cells and sIgA not only reside on the mucosa where the antigen was originally presented, but there is also a reasonable chance that these components will traffic to other mucosal surfaces.2
So intranasal vaccine could be expected to protect distant mucosal surfaces (urogenital, GI, and respiratory), leading to vaccine-induced systemic antibody plus mucosal immunity (sIGA and MALT responses) at each site.
Let’s look at a novel “two-site” chlamydia vaccine
Recently a phase 1 chlamydia vaccine that used a novel two-pronged administration site/schedule was successful at inducing both mucosal and systemic immunity in a proof-of-concept study – achieving the best of both worlds.3 This may be a template for vaccines in years to come. British investigators studied 50 healthy women aged 19-45 years in a double-blind, parallel, randomized, placebo-controlled trial that used a recombinant chlamydia protein subunit antigen (CTH522). The vaccine schedule involved three injectable priming doses followed soon thereafter by two intranasal boosting doses. There were three groups:
1. CTH522 adjuvanted with CAF01 liposomes (CTH522:CAF01).
2. CTH522 adjuvanted with aluminum hydroxide (CTH522:AH).
3. Placebo (saline).
The intramuscular (IM) priming schedule was 0, 1, and 4 months. The intranasal vaccine booster doses or placebo were given at 4.5 and 5 months. No related serious adverse reactions occurred. For injectable dosing, the most frequent adverse event was mild local injection-site reactions in all subjects in both vaccine groups vs. in 60% of placebo recipients (P = .053). The adjuvants were the likely cause for local reactions. Intranasal doses had local reactions in 47% of both vaccine groups and 60% of placebo recipients; P = 1.000).
Both vaccines produced systemic IgG seroconversion (including neutralizing antibody) plus small amounts of IgG in the nasal cavity and genital tract in all vaccine recipients; no placebo recipient seroconverted. Interestingly, liposomally-adjuvanted vaccine produced a more rapid systemic IgG response and higher serum titers than the alum-adjuvanted vaccine. Likewise, the IM liposomal vaccine also induced higher but still small mucosal IgG antibody responses (P = .0091). Intranasal IM-induced IgG titers were not boosted by later intranasal vaccine dosing.
Subjects getting liposomal vaccine (but not alum vaccine or placebo) boosters had detectable sIgA titers in both nasal and genital tract secretions. Liposomal vaccine recipients also had fivefold to sixfold higher median titers than alum vaccine recipients after the priming dose, and these higher titers persisted to the end of the study. All liposomal vaccine recipients developed antichlamydial cell-mediated responses vs. 57% alum-adjuvanted vaccine recipients. (P = .01). So both use of two-site dosing and the liposomal adjuvant appeared critical to better responses.
In summary
While this candidate vaccine has hurdles to overcome before coming into routine use, the proof-of-principle that a combination injectable-intranasal vaccine schedule can induce robust systemic and mucosal immunity when given with an appropriate adjuvant is very promising. Adding more vaccines to the schedule then becomes an issue, but that is one of those “good” problems we can deal with later.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital-Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines, receives funding from GlaxoSmithKline for studies on pneumococcal and rotavirus vaccines, and from Pfizer for a study on pneumococcal vaccine on which Dr. Harrison is a sub-investigator. The hospital also receives Centers for Disease Control and Prevention funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus, and also for rotavirus. Email Dr. Harrison at [email protected].
References
1. PLOS Biology. 2012 Sep 1. doi: 10.1371/journal.pbio.1001397.
2. Mucosal Immunity in the Human Female Reproductive Tract in “Mucosal Immunology,” 4th ed., Volume 2 (Cambridge, MA: Academic Press, 2015, pp. 2097-124).
3. Lancet Infect Dis. 2019. doi: 10.1016/S1473-3099(19)30279-8.
Vaccines are marvelous, and there are many well documented success stories, including rotavirus (RV) vaccines, where a live vaccine is administered to the gastrointestinal mucosa via oral drops. Antigens presented at the mucosal/epithelial surface not only induce systemic serum IgG – as do injectable vaccines – but also induce secretory IgA (sIgA), which is most helpful in diseases that directly affect the mucosa.

Mucosal vs. systemic immunity
Antibody being present on mucosal surfaces (point of initial pathogen contact) has a chance to neutralize the pathogen before it gains a foothold. Pathogen-specific mucosal lymphoid elements (e.g. in Peyer’s patches in the gut) also appear critical for optimal protection.1 The presence of both mucosal immune elements means that infection is severely limited or at times entirely prevented. So virus entering the GI tract causes minimal to no gut lining injury. Hence, there is no or mostly reduced vomiting/diarrhea. A downside of mucosally-administered live vaccines is that preexisting antibody to the vaccine antigens can reduce or block vaccine virus replication in the vaccinee, blunting or preventing protection. Note: Preexisting antibody also affects injectable live vaccines, such as the measles vaccine, similarly.
Classic injectable live or nonlive vaccines provide their most potent protection via systemic cellular responses antibody and/or antibodies in serum and extracellular fluid (ECF) where IgG and IgM are in highest concentrations. So even successful injectable vaccines still allow mucosal infection to start but then intercept further spread and prevent most of the downstream damage (think pertussis) or neutralize an infection-generated toxin (pertussis or tetanus). It usually is only after infection-induced damage occurs that systemic IgG and IgM gain better access to respiratory epithelial surfaces, but still only at a fraction of circulating concentrations. Indeed, pertussis vaccine–induced systemic immunity allows the pathogen to attack and replicate in/on host surface cells, causing toxin release and variable amounts of local mucosal injury/inflammation before vaccine-induced systemic immunity gains adequate access to the pathogen and/or to its toxin which may enter systemic circulation.
Live attenuated influenza vaccine (LAIV) induces mucosal immunity
Another “standard” vaccine that induces mucosal immunity – LAIV – was developed to improve on protection afforded by injectable influenza vaccines (IIVs), but LAIV has had hiccups in the United States. One example is several years of negligible protection against H1N1 disease. As long as LAIV’s vaccine strain had reasonably matched the circulating strains, LAIV worked at least as well as injectable influenza vaccine, and even offered some cross-protection against mildly mismatched strains. But after a number of years of LAIV use, vaccine effectiveness in the United States vs. H1N1 strains appeared to fade due to previously undetected but significant changes in the circulating H1N1 strain. The lesson is that mucosal immunity’s advantages are lost if too much change occurs in the pathogen target for sIgA and mucosally-associated lymphoid tissue cells (MALT)).
Other vaccines likely need to induce mucosal immunity
Protection at the mucosal level will likely be needed for success against norovirus, parainfluenza, respiratory syncytial virus (RSV), Neisseria gonorrhea, and chlamydia. Another helpful aspect of mucosal immunity is that immune cells and sIgA not only reside on the mucosa where the antigen was originally presented, but there is also a reasonable chance that these components will traffic to other mucosal surfaces.2
So intranasal vaccine could be expected to protect distant mucosal surfaces (urogenital, GI, and respiratory), leading to vaccine-induced systemic antibody plus mucosal immunity (sIGA and MALT responses) at each site.
Let’s look at a novel “two-site” chlamydia vaccine
Recently a phase 1 chlamydia vaccine that used a novel two-pronged administration site/schedule was successful at inducing both mucosal and systemic immunity in a proof-of-concept study – achieving the best of both worlds.3 This may be a template for vaccines in years to come. British investigators studied 50 healthy women aged 19-45 years in a double-blind, parallel, randomized, placebo-controlled trial that used a recombinant chlamydia protein subunit antigen (CTH522). The vaccine schedule involved three injectable priming doses followed soon thereafter by two intranasal boosting doses. There were three groups:
1. CTH522 adjuvanted with CAF01 liposomes (CTH522:CAF01).
2. CTH522 adjuvanted with aluminum hydroxide (CTH522:AH).
3. Placebo (saline).
The intramuscular (IM) priming schedule was 0, 1, and 4 months. The intranasal vaccine booster doses or placebo were given at 4.5 and 5 months. No related serious adverse reactions occurred. For injectable dosing, the most frequent adverse event was mild local injection-site reactions in all subjects in both vaccine groups vs. in 60% of placebo recipients (P = .053). The adjuvants were the likely cause for local reactions. Intranasal doses had local reactions in 47% of both vaccine groups and 60% of placebo recipients; P = 1.000).
Both vaccines produced systemic IgG seroconversion (including neutralizing antibody) plus small amounts of IgG in the nasal cavity and genital tract in all vaccine recipients; no placebo recipient seroconverted. Interestingly, liposomally-adjuvanted vaccine produced a more rapid systemic IgG response and higher serum titers than the alum-adjuvanted vaccine. Likewise, the IM liposomal vaccine also induced higher but still small mucosal IgG antibody responses (P = .0091). Intranasal IM-induced IgG titers were not boosted by later intranasal vaccine dosing.
Subjects getting liposomal vaccine (but not alum vaccine or placebo) boosters had detectable sIgA titers in both nasal and genital tract secretions. Liposomal vaccine recipients also had fivefold to sixfold higher median titers than alum vaccine recipients after the priming dose, and these higher titers persisted to the end of the study. All liposomal vaccine recipients developed antichlamydial cell-mediated responses vs. 57% alum-adjuvanted vaccine recipients. (P = .01). So both use of two-site dosing and the liposomal adjuvant appeared critical to better responses.
In summary
While this candidate vaccine has hurdles to overcome before coming into routine use, the proof-of-principle that a combination injectable-intranasal vaccine schedule can induce robust systemic and mucosal immunity when given with an appropriate adjuvant is very promising. Adding more vaccines to the schedule then becomes an issue, but that is one of those “good” problems we can deal with later.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital-Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines, receives funding from GlaxoSmithKline for studies on pneumococcal and rotavirus vaccines, and from Pfizer for a study on pneumococcal vaccine on which Dr. Harrison is a sub-investigator. The hospital also receives Centers for Disease Control and Prevention funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus, and also for rotavirus. Email Dr. Harrison at [email protected].
References
1. PLOS Biology. 2012 Sep 1. doi: 10.1371/journal.pbio.1001397.
2. Mucosal Immunity in the Human Female Reproductive Tract in “Mucosal Immunology,” 4th ed., Volume 2 (Cambridge, MA: Academic Press, 2015, pp. 2097-124).
3. Lancet Infect Dis. 2019. doi: 10.1016/S1473-3099(19)30279-8.
Vaccines are marvelous, and there are many well documented success stories, including rotavirus (RV) vaccines, where a live vaccine is administered to the gastrointestinal mucosa via oral drops. Antigens presented at the mucosal/epithelial surface not only induce systemic serum IgG – as do injectable vaccines – but also induce secretory IgA (sIgA), which is most helpful in diseases that directly affect the mucosa.

Mucosal vs. systemic immunity
Antibody being present on mucosal surfaces (point of initial pathogen contact) has a chance to neutralize the pathogen before it gains a foothold. Pathogen-specific mucosal lymphoid elements (e.g. in Peyer’s patches in the gut) also appear critical for optimal protection.1 The presence of both mucosal immune elements means that infection is severely limited or at times entirely prevented. So virus entering the GI tract causes minimal to no gut lining injury. Hence, there is no or mostly reduced vomiting/diarrhea. A downside of mucosally-administered live vaccines is that preexisting antibody to the vaccine antigens can reduce or block vaccine virus replication in the vaccinee, blunting or preventing protection. Note: Preexisting antibody also affects injectable live vaccines, such as the measles vaccine, similarly.
Classic injectable live or nonlive vaccines provide their most potent protection via systemic cellular responses antibody and/or antibodies in serum and extracellular fluid (ECF) where IgG and IgM are in highest concentrations. So even successful injectable vaccines still allow mucosal infection to start but then intercept further spread and prevent most of the downstream damage (think pertussis) or neutralize an infection-generated toxin (pertussis or tetanus). It usually is only after infection-induced damage occurs that systemic IgG and IgM gain better access to respiratory epithelial surfaces, but still only at a fraction of circulating concentrations. Indeed, pertussis vaccine–induced systemic immunity allows the pathogen to attack and replicate in/on host surface cells, causing toxin release and variable amounts of local mucosal injury/inflammation before vaccine-induced systemic immunity gains adequate access to the pathogen and/or to its toxin which may enter systemic circulation.
Live attenuated influenza vaccine (LAIV) induces mucosal immunity
Another “standard” vaccine that induces mucosal immunity – LAIV – was developed to improve on protection afforded by injectable influenza vaccines (IIVs), but LAIV has had hiccups in the United States. One example is several years of negligible protection against H1N1 disease. As long as LAIV’s vaccine strain had reasonably matched the circulating strains, LAIV worked at least as well as injectable influenza vaccine, and even offered some cross-protection against mildly mismatched strains. But after a number of years of LAIV use, vaccine effectiveness in the United States vs. H1N1 strains appeared to fade due to previously undetected but significant changes in the circulating H1N1 strain. The lesson is that mucosal immunity’s advantages are lost if too much change occurs in the pathogen target for sIgA and mucosally-associated lymphoid tissue cells (MALT)).
Other vaccines likely need to induce mucosal immunity
Protection at the mucosal level will likely be needed for success against norovirus, parainfluenza, respiratory syncytial virus (RSV), Neisseria gonorrhea, and chlamydia. Another helpful aspect of mucosal immunity is that immune cells and sIgA not only reside on the mucosa where the antigen was originally presented, but there is also a reasonable chance that these components will traffic to other mucosal surfaces.2
So intranasal vaccine could be expected to protect distant mucosal surfaces (urogenital, GI, and respiratory), leading to vaccine-induced systemic antibody plus mucosal immunity (sIGA and MALT responses) at each site.
Let’s look at a novel “two-site” chlamydia vaccine
Recently a phase 1 chlamydia vaccine that used a novel two-pronged administration site/schedule was successful at inducing both mucosal and systemic immunity in a proof-of-concept study – achieving the best of both worlds.3 This may be a template for vaccines in years to come. British investigators studied 50 healthy women aged 19-45 years in a double-blind, parallel, randomized, placebo-controlled trial that used a recombinant chlamydia protein subunit antigen (CTH522). The vaccine schedule involved three injectable priming doses followed soon thereafter by two intranasal boosting doses. There were three groups:
1. CTH522 adjuvanted with CAF01 liposomes (CTH522:CAF01).
2. CTH522 adjuvanted with aluminum hydroxide (CTH522:AH).
3. Placebo (saline).
The intramuscular (IM) priming schedule was 0, 1, and 4 months. The intranasal vaccine booster doses or placebo were given at 4.5 and 5 months. No related serious adverse reactions occurred. For injectable dosing, the most frequent adverse event was mild local injection-site reactions in all subjects in both vaccine groups vs. in 60% of placebo recipients (P = .053). The adjuvants were the likely cause for local reactions. Intranasal doses had local reactions in 47% of both vaccine groups and 60% of placebo recipients; P = 1.000).
Both vaccines produced systemic IgG seroconversion (including neutralizing antibody) plus small amounts of IgG in the nasal cavity and genital tract in all vaccine recipients; no placebo recipient seroconverted. Interestingly, liposomally-adjuvanted vaccine produced a more rapid systemic IgG response and higher serum titers than the alum-adjuvanted vaccine. Likewise, the IM liposomal vaccine also induced higher but still small mucosal IgG antibody responses (P = .0091). Intranasal IM-induced IgG titers were not boosted by later intranasal vaccine dosing.
Subjects getting liposomal vaccine (but not alum vaccine or placebo) boosters had detectable sIgA titers in both nasal and genital tract secretions. Liposomal vaccine recipients also had fivefold to sixfold higher median titers than alum vaccine recipients after the priming dose, and these higher titers persisted to the end of the study. All liposomal vaccine recipients developed antichlamydial cell-mediated responses vs. 57% alum-adjuvanted vaccine recipients. (P = .01). So both use of two-site dosing and the liposomal adjuvant appeared critical to better responses.
In summary
While this candidate vaccine has hurdles to overcome before coming into routine use, the proof-of-principle that a combination injectable-intranasal vaccine schedule can induce robust systemic and mucosal immunity when given with an appropriate adjuvant is very promising. Adding more vaccines to the schedule then becomes an issue, but that is one of those “good” problems we can deal with later.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital-Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines, receives funding from GlaxoSmithKline for studies on pneumococcal and rotavirus vaccines, and from Pfizer for a study on pneumococcal vaccine on which Dr. Harrison is a sub-investigator. The hospital also receives Centers for Disease Control and Prevention funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus, and also for rotavirus. Email Dr. Harrison at [email protected].
References
1. PLOS Biology. 2012 Sep 1. doi: 10.1371/journal.pbio.1001397.
2. Mucosal Immunity in the Human Female Reproductive Tract in “Mucosal Immunology,” 4th ed., Volume 2 (Cambridge, MA: Academic Press, 2015, pp. 2097-124).
3. Lancet Infect Dis. 2019. doi: 10.1016/S1473-3099(19)30279-8.
Growing vaping habit may lead to nicotine addiction in adolescents
and in 2019 almost 12% of high school seniors reported that they were vaping every day, according to data from the Monitoring the Future surveys.
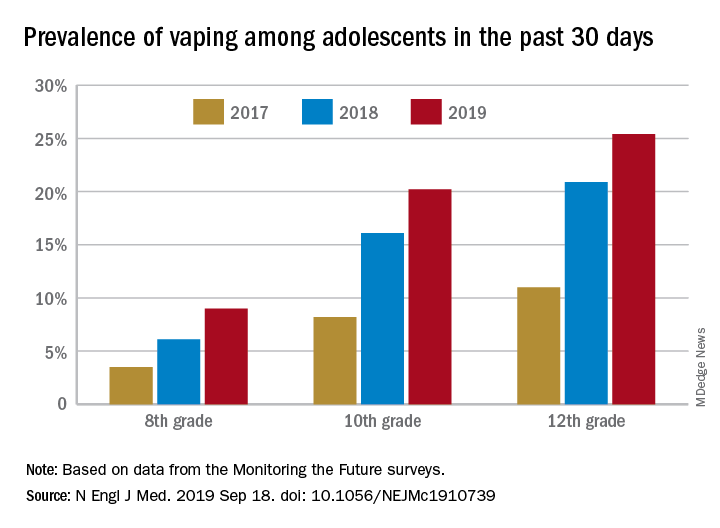
Daily use – defined as vaping on 20 or more of the previous 30 days – was reported by 6.9% of 10th-grade and 1.9% of 8th-grade respondents in the 2019 survey, which was the first time use in these age groups was assessed. “The substantial levels of daily vaping suggest the development of nicotine addiction,” Richard Miech, PhD, and associates said Sept. 18 in the New England Journal of Medicine.
From 2017 to 2019, e-cigarette use over the previous 30 days increased from 11.0% to 25.4% among 12th graders, from 8.2% to 20.2% in 10th graders, and from 3.5% to 9.0% of 8th graders, suggesting that “current efforts by the vaping industry, government agencies, and schools have thus far proved insufficient to stop the rapid spread of nicotine vaping among adolescents,” the investigators wrote.
By 2019, over 40% of 12th-grade students reported ever using e-cigarettes, along with more than 36% of 10th graders and almost 21% of 8th graders. Corresponding figures for past 12-month use were 35.1%, 31.1%, and 16.1%, they reported.
“New efforts are needed to protect youth from using nicotine during adolescence, when the developing brain is particularly susceptible to permanent changes from nicotine use and when almost all nicotine addiction is established,” the investigators wrote.
The analysis was funded by a grant from the National Institute on Drug Abuse to Dr. Miech.
SOURCE: Miech R et al. N Engl J Med. 2019 Sep 18. doi: 10.1056/NEJMc1910739.
and in 2019 almost 12% of high school seniors reported that they were vaping every day, according to data from the Monitoring the Future surveys.

Daily use – defined as vaping on 20 or more of the previous 30 days – was reported by 6.9% of 10th-grade and 1.9% of 8th-grade respondents in the 2019 survey, which was the first time use in these age groups was assessed. “The substantial levels of daily vaping suggest the development of nicotine addiction,” Richard Miech, PhD, and associates said Sept. 18 in the New England Journal of Medicine.
From 2017 to 2019, e-cigarette use over the previous 30 days increased from 11.0% to 25.4% among 12th graders, from 8.2% to 20.2% in 10th graders, and from 3.5% to 9.0% of 8th graders, suggesting that “current efforts by the vaping industry, government agencies, and schools have thus far proved insufficient to stop the rapid spread of nicotine vaping among adolescents,” the investigators wrote.
By 2019, over 40% of 12th-grade students reported ever using e-cigarettes, along with more than 36% of 10th graders and almost 21% of 8th graders. Corresponding figures for past 12-month use were 35.1%, 31.1%, and 16.1%, they reported.
“New efforts are needed to protect youth from using nicotine during adolescence, when the developing brain is particularly susceptible to permanent changes from nicotine use and when almost all nicotine addiction is established,” the investigators wrote.
The analysis was funded by a grant from the National Institute on Drug Abuse to Dr. Miech.
SOURCE: Miech R et al. N Engl J Med. 2019 Sep 18. doi: 10.1056/NEJMc1910739.
and in 2019 almost 12% of high school seniors reported that they were vaping every day, according to data from the Monitoring the Future surveys.

Daily use – defined as vaping on 20 or more of the previous 30 days – was reported by 6.9% of 10th-grade and 1.9% of 8th-grade respondents in the 2019 survey, which was the first time use in these age groups was assessed. “The substantial levels of daily vaping suggest the development of nicotine addiction,” Richard Miech, PhD, and associates said Sept. 18 in the New England Journal of Medicine.
From 2017 to 2019, e-cigarette use over the previous 30 days increased from 11.0% to 25.4% among 12th graders, from 8.2% to 20.2% in 10th graders, and from 3.5% to 9.0% of 8th graders, suggesting that “current efforts by the vaping industry, government agencies, and schools have thus far proved insufficient to stop the rapid spread of nicotine vaping among adolescents,” the investigators wrote.
By 2019, over 40% of 12th-grade students reported ever using e-cigarettes, along with more than 36% of 10th graders and almost 21% of 8th graders. Corresponding figures for past 12-month use were 35.1%, 31.1%, and 16.1%, they reported.
“New efforts are needed to protect youth from using nicotine during adolescence, when the developing brain is particularly susceptible to permanent changes from nicotine use and when almost all nicotine addiction is established,” the investigators wrote.
The analysis was funded by a grant from the National Institute on Drug Abuse to Dr. Miech.
SOURCE: Miech R et al. N Engl J Med. 2019 Sep 18. doi: 10.1056/NEJMc1910739.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Adolescents who use e-cigarettes every day may be developing nicotine addiction.
Major finding: In 2019, almost 12% of high school seniors were vaping every day.
Study details: Monitoring the Future surveys nationally representative samples of 8th-, 10th-, and 12th-grade students each year.
Disclosures: The analysis was funded by a grant from the National Institute on Drug Abuse to Dr. Miech.
Source: Miech R et al. N Engl J Med. 2019 Sep 18. doi: 10.1056/NEJMc1910739.
Wildfire smoke has acute cardiorespiratory impact, but long-term effects still under study
The 2019 wildfire season is underway in many locales across the United States, exposing millions of individuals to smoky conditions that will have health consequences ranging from stinging eyes to scratchy throats to a trip to the ED for asthma or chronic obstructive pulmonary disease (COPD) exacerbation. Questions about long-term health impacts are on the minds of many, including physicians and their patients who live with cardiorespiratory conditions.

John R. Balmes, MD, a pulmonologist at the University of California, San Francisco, and an expert on the respiratory and cardiovascular effects of air pollutants, suggested that the best available published literature points to “pretty strong evidence for acute effects of wildfire smoke on respiratory health, meaning people with preexisting asthma and COPD are at risk for exacerbations, and probably for respiratory tract infections as well.” He said, “It’s a little less clear, but there’s good biological plausibility for increased risk of respiratory tract infections because when your alveolar macrophages are overloaded with carbon particles that are toxic to those cells, they don’t function as well as a first line of defense against bacterial infection, for example.”
The new normal of wildfires
Warmer, drier summers in recent years in the western United States and many other regions, attributed by climate experts to global climate change, have produced catastrophic wildfires (PNAS;2016 Oct 18;113[42]11770-5; Science 2006 Aug 18;313:940-3). The Camp Fire in Northern California broke out in November 2018, took the lives of at least 85 people, and cost more than $16 billion in damage. Smoke from that blaze reached hazardous levels in San Francisco, Sacramento, Fresno, and many other smaller towns. Other forest fires in that year caused heavy smoke conditions in Portland, Seattle, Vancouver, and Anchorage. Such events are expected to be repeated often in the coming years (Int J Environ Res Public Health. 2019 Jul 6;16[13]).

Wildfire smoke can contain a wide range of substances, chemicals, and gases with known and unknown cardiorespiratory implications. “Smoke is composed primarily of carbon dioxide, water vapor, carbon monoxide, particulate matter, hydrocarbons and other organic chemicals, nitrogen oxides, trace minerals and several thousand other compounds,” according to the U.S. Environmental Protection Agency (Wildfire smoke: A guide for public health officials 2019. Washington, D.C.: EPA, 2019). The EPA report noted, “Particles with diameters less than 10 mcm (particulate matter, or PM10) can be inhaled into the lungs and affect the lungs, heart, and blood vessels. The smallest particles, those less than 2.5 mcm in diameter (PM2.5), are the greatest risk to public health because they can reach deep into the lungs and may even make it into the bloodstream.”
Research on health impact
In early June of 2008, Wayne Cascio, MD, awoke in his Greenville, N.C., home to the stench of smoke emanating from a large peat fire burning some 65 miles away. By the time he reached the parking lot at East Carolina University in Greenville to begin his workday as chief of cardiology, the haze of smoke had thickened to the point where he could only see a few feet in front of him.
Over the next several weeks, the fire scorched 41,000 acres and produced haze and air pollution that far exceeded National Ambient Air Quality Standards for particulate matter and blanketed rural communities in the state’s eastern region. The price tag for management of the blaze reached $20 million. Because of his interest in the health effects of wildfire smoke and because of his relationship with investigators at the EPA, Dr. Cascio initiated an epidemiology study to investigate the effects of exposure on cardiorespiratory outcomes in the population affected by the fire (Environ Health Perspect. 2011 Oct;119[10]:1415-20).
By combining satellite data with syndromic surveillance drawn from hospital records in 41 counties contained in the North Carolina Disease Event Tracking and Epidemiologic Collection Tool, he and his colleagues found that exposure to the peat wildfire smoke led to increases in the cumulative risk ratio for asthma (relative risk, 1.65), chronic obstructive pulmonary disease (RR, 1.73), and pneumonia and acute bronchitis (RR, 1.59). ED visits related to cardiopulmonary symptoms and heart failure also were significantly increased (RR, 1.23 and 1.37, respectively). “That was really the first study to strongly identify a cardiac endpoint related to wildfire smoke exposure,” said Dr. Cascio, who now directs the EPA’s National Health and Environmental Effects Research Laboratory. “It really pointed out how little we knew about the health effects of wildfire up until that time.”
Those early findings have been replicated in subsequent research about the acute health effects of exposure to wildfire smoke, which contains PM2.5 and other toxic substances from structures, electronic devices, and automobiles destroyed in the path of flames, including heavy metals and asbestos. Most of the work has focused on smoke-related cardiovascular and respiratory ED visits and hospitalizations.
A study of the 2008 California wildfire impact on ED visits accounted for ozone levels in addition to PM2.5 in the smoke. During the active fire periods, PM2.5 was significantly associated with exacerbations of asthma and COPD and these effects remained after controlling for ozone levels. PM2.5 inhalation during the wildfires was associated with increased risk of an ED visit for asthma (RR, 1.112; 95% confidence interval, 1.087-1.138) for a 10 mcg/m3 increase in PM2.5 and COPD (RR, 1.05; 95% CI, 1.019-1.0825), as well as for combined respiratory visits (RR, 1.035; 95% CI, 1.023-1.046) (Environ Int. 2109 Aug;129:291-8).
Researchers who evaluated the health impacts of wildfires in California during the 2015 fire season found an increase in all-cause cardiovascular and respiratory ED visits, especially among those aged 65 years and older during smoke days. The population-based study included 1,196,233 ED visits during May 1–Sept. 30 that year. PM2.5 concentrations were categorized as light, medium, or dense. Relative risk rose with the amount of smoke in the air. Rates of all-cause cardiovascular ED visits were elevated across levels of smoke density, with the greatest increase on dense smoke days and among those aged 65 years or older (RR,1.15; 95% CI, 1.09-1.22). All-cause cerebrovascular visits were associated with dense smoke days, especially among those aged 65 years and older (RR, 1.22; 95% CI, 1.00-1.49). Respiratory conditions also were increased on dense smoke days (RR, 1.18; 95% CI, 1.08-1.28) (J Am Heart Assoc. 2018 Apr 11;7:e007492. doi: 10.1161/JAHA.117.007492).
Long-term effects unknown
When it comes to the long-term effects of wildfire smoke on human health outcomes, much less is known. In a recent literature review, Colleen E. Reid, PhD, and Melissa May Maestas, PhD, found only one study that investigated long-term respiratory health impacts of wildfire smoke, and only a few studies that have estimated future health impacts of wildfires under likely climate change scenarios (Curr Opin Pulm Med. 2019 Mar;25:179-87).
“We know that there are immediate respiratory health effects from wildfire smoke,” said Dr. Reid of the department of geography at the University of Colorado Boulder. “What’s less known is everything else. That’s challenging, because people want to know about the long-term health effects.”
Evidence from the scientific literature suggests that exposure to air pollution adversely affects cardiovascular health, but whether exposure to wildfire smoke confers a similar risk is less clear. “Until just a few years ago we haven’t been able to study wildfire exposure measures on a large scale,” said EPA scientist Ana G. Rappold, PhD, a statistician there in the environmental public health division of the National Health and Environmental Effects Research Laboratory. “It’s also hard to predict wildfires, so it’s hard to plan for an epidemiologic study if you don’t know where they’re going to occur.”
Dr. Rappold and colleagues examined cardiopulmonary hospitalizations among adults aged 65 years and older in 692 U.S. counties within 200 km of 123 large wildfires during 2008-2010 (Environ Health Perspect. 2019;127[3]:37006. doi: 10.1289/EHP3860). They observed that an increased risk of PM2.5-related cardiopulmonary hospitalizations was similar on smoke and nonsmoke days across multiple lags and exposure metrics, while risk for asthma-related hospitalizations was higher during smoke days. “One hypothesis is that this was an older study population, so naturally if you’re inhaling smoke, the first organ that’s impacted in an older population is the lungs,” Dr. Rappold said. “If you go to the hospital for asthma, wheezing, or bronchitis, you are taken out of the risk pool for cardiovascular and other diseases. That could explain why in other studies we don’t see a clear cardiovascular signal as we have for air pollution studies in general. Another aspect to this study is, the exposure metric was PM2.5, but smoke contains many other components, particularly gases, which are respiratory irritants. It could be that this triggers a higher risk for respiratory [effects] than regular episodes of high PM2.5 exposure, just because of the additional gases that people are exposed to.”
Another complicating factor is the paucity of data about solutions to long-term exposure to wildfire smoke. “If you’re impacted by high-exposure levels for 60 days, that is not something we have experienced before,” Dr. Rappold noted. “What are the solutions for that community? What works? Can we show that by implementing community-level resilience plans with HEPA [high-efficiency particulate air] filters or other interventions, do the overall outcomes improve? Doctors are the first ones to talk with their patients about their symptoms and about how to take care of their conditions. They can clearly make a difference in emphasizing reducing exposures in a way that fits their patients individually, either reducing the amount of time spent outside, the duration of exposure, and the level of exposure. Maybe change activities based on the intensity of exposure. Don’t go for a run outside when it’s smoky, because your ventilation rate is higher and you will breathe in more smoke. Become aware of those things.”
Advising vulnerable patients
While research in this field advances, the unforgiving wildfire season looms, assuring more destruction of property and threats to cardiorespiratory health. “There are a lot of questions that research will have an opportunity to address as we go forward, including the utility and the benefit of N95 masks, the utility of HEPA filters used in the house, and even with HVAC [heating, ventilation, and air conditioning] systems,” Dr. Cascio said. “Can we really clean up the indoor air well enough to protect us from wildfire smoke?”
The way he sees it, the time is ripe for clinicians and officials in public and private practice settings to refine how they distribute information to people living in areas affected by wildfire smoke. “We can’t force people do anything, but at least if they’re informed, then they understand they can make an informed decision about how they might want to affect what they do that would limit their exposure,” he said. “As a patient, my health care system sends text and email messages to me. So, why couldn’t the hospital send out a text message or an email to all of the patients with COPD, coronary disease, and heart failure when an area is impacted by smoke, saying, ‘Check your air quality and take action if air quality is poor?’ Physicians don’t have time to do this kind of education in the office for all of their patients. I know that from experience. But if one were to only focus on those at highest risk, and encourage them to follow our guidelines, which might include doing HEPA filter treatment in the home, we probably would reduce the number of clinical events in a cost-effective way.”
The 2019 wildfire season is underway in many locales across the United States, exposing millions of individuals to smoky conditions that will have health consequences ranging from stinging eyes to scratchy throats to a trip to the ED for asthma or chronic obstructive pulmonary disease (COPD) exacerbation. Questions about long-term health impacts are on the minds of many, including physicians and their patients who live with cardiorespiratory conditions.

John R. Balmes, MD, a pulmonologist at the University of California, San Francisco, and an expert on the respiratory and cardiovascular effects of air pollutants, suggested that the best available published literature points to “pretty strong evidence for acute effects of wildfire smoke on respiratory health, meaning people with preexisting asthma and COPD are at risk for exacerbations, and probably for respiratory tract infections as well.” He said, “It’s a little less clear, but there’s good biological plausibility for increased risk of respiratory tract infections because when your alveolar macrophages are overloaded with carbon particles that are toxic to those cells, they don’t function as well as a first line of defense against bacterial infection, for example.”
The new normal of wildfires
Warmer, drier summers in recent years in the western United States and many other regions, attributed by climate experts to global climate change, have produced catastrophic wildfires (PNAS;2016 Oct 18;113[42]11770-5; Science 2006 Aug 18;313:940-3). The Camp Fire in Northern California broke out in November 2018, took the lives of at least 85 people, and cost more than $16 billion in damage. Smoke from that blaze reached hazardous levels in San Francisco, Sacramento, Fresno, and many other smaller towns. Other forest fires in that year caused heavy smoke conditions in Portland, Seattle, Vancouver, and Anchorage. Such events are expected to be repeated often in the coming years (Int J Environ Res Public Health. 2019 Jul 6;16[13]).

Wildfire smoke can contain a wide range of substances, chemicals, and gases with known and unknown cardiorespiratory implications. “Smoke is composed primarily of carbon dioxide, water vapor, carbon monoxide, particulate matter, hydrocarbons and other organic chemicals, nitrogen oxides, trace minerals and several thousand other compounds,” according to the U.S. Environmental Protection Agency (Wildfire smoke: A guide for public health officials 2019. Washington, D.C.: EPA, 2019). The EPA report noted, “Particles with diameters less than 10 mcm (particulate matter, or PM10) can be inhaled into the lungs and affect the lungs, heart, and blood vessels. The smallest particles, those less than 2.5 mcm in diameter (PM2.5), are the greatest risk to public health because they can reach deep into the lungs and may even make it into the bloodstream.”
Research on health impact
In early June of 2008, Wayne Cascio, MD, awoke in his Greenville, N.C., home to the stench of smoke emanating from a large peat fire burning some 65 miles away. By the time he reached the parking lot at East Carolina University in Greenville to begin his workday as chief of cardiology, the haze of smoke had thickened to the point where he could only see a few feet in front of him.
Over the next several weeks, the fire scorched 41,000 acres and produced haze and air pollution that far exceeded National Ambient Air Quality Standards for particulate matter and blanketed rural communities in the state’s eastern region. The price tag for management of the blaze reached $20 million. Because of his interest in the health effects of wildfire smoke and because of his relationship with investigators at the EPA, Dr. Cascio initiated an epidemiology study to investigate the effects of exposure on cardiorespiratory outcomes in the population affected by the fire (Environ Health Perspect. 2011 Oct;119[10]:1415-20).
By combining satellite data with syndromic surveillance drawn from hospital records in 41 counties contained in the North Carolina Disease Event Tracking and Epidemiologic Collection Tool, he and his colleagues found that exposure to the peat wildfire smoke led to increases in the cumulative risk ratio for asthma (relative risk, 1.65), chronic obstructive pulmonary disease (RR, 1.73), and pneumonia and acute bronchitis (RR, 1.59). ED visits related to cardiopulmonary symptoms and heart failure also were significantly increased (RR, 1.23 and 1.37, respectively). “That was really the first study to strongly identify a cardiac endpoint related to wildfire smoke exposure,” said Dr. Cascio, who now directs the EPA’s National Health and Environmental Effects Research Laboratory. “It really pointed out how little we knew about the health effects of wildfire up until that time.”
Those early findings have been replicated in subsequent research about the acute health effects of exposure to wildfire smoke, which contains PM2.5 and other toxic substances from structures, electronic devices, and automobiles destroyed in the path of flames, including heavy metals and asbestos. Most of the work has focused on smoke-related cardiovascular and respiratory ED visits and hospitalizations.
A study of the 2008 California wildfire impact on ED visits accounted for ozone levels in addition to PM2.5 in the smoke. During the active fire periods, PM2.5 was significantly associated with exacerbations of asthma and COPD and these effects remained after controlling for ozone levels. PM2.5 inhalation during the wildfires was associated with increased risk of an ED visit for asthma (RR, 1.112; 95% confidence interval, 1.087-1.138) for a 10 mcg/m3 increase in PM2.5 and COPD (RR, 1.05; 95% CI, 1.019-1.0825), as well as for combined respiratory visits (RR, 1.035; 95% CI, 1.023-1.046) (Environ Int. 2109 Aug;129:291-8).
Researchers who evaluated the health impacts of wildfires in California during the 2015 fire season found an increase in all-cause cardiovascular and respiratory ED visits, especially among those aged 65 years and older during smoke days. The population-based study included 1,196,233 ED visits during May 1–Sept. 30 that year. PM2.5 concentrations were categorized as light, medium, or dense. Relative risk rose with the amount of smoke in the air. Rates of all-cause cardiovascular ED visits were elevated across levels of smoke density, with the greatest increase on dense smoke days and among those aged 65 years or older (RR,1.15; 95% CI, 1.09-1.22). All-cause cerebrovascular visits were associated with dense smoke days, especially among those aged 65 years and older (RR, 1.22; 95% CI, 1.00-1.49). Respiratory conditions also were increased on dense smoke days (RR, 1.18; 95% CI, 1.08-1.28) (J Am Heart Assoc. 2018 Apr 11;7:e007492. doi: 10.1161/JAHA.117.007492).
Long-term effects unknown
When it comes to the long-term effects of wildfire smoke on human health outcomes, much less is known. In a recent literature review, Colleen E. Reid, PhD, and Melissa May Maestas, PhD, found only one study that investigated long-term respiratory health impacts of wildfire smoke, and only a few studies that have estimated future health impacts of wildfires under likely climate change scenarios (Curr Opin Pulm Med. 2019 Mar;25:179-87).
“We know that there are immediate respiratory health effects from wildfire smoke,” said Dr. Reid of the department of geography at the University of Colorado Boulder. “What’s less known is everything else. That’s challenging, because people want to know about the long-term health effects.”
Evidence from the scientific literature suggests that exposure to air pollution adversely affects cardiovascular health, but whether exposure to wildfire smoke confers a similar risk is less clear. “Until just a few years ago we haven’t been able to study wildfire exposure measures on a large scale,” said EPA scientist Ana G. Rappold, PhD, a statistician there in the environmental public health division of the National Health and Environmental Effects Research Laboratory. “It’s also hard to predict wildfires, so it’s hard to plan for an epidemiologic study if you don’t know where they’re going to occur.”
Dr. Rappold and colleagues examined cardiopulmonary hospitalizations among adults aged 65 years and older in 692 U.S. counties within 200 km of 123 large wildfires during 2008-2010 (Environ Health Perspect. 2019;127[3]:37006. doi: 10.1289/EHP3860). They observed that an increased risk of PM2.5-related cardiopulmonary hospitalizations was similar on smoke and nonsmoke days across multiple lags and exposure metrics, while risk for asthma-related hospitalizations was higher during smoke days. “One hypothesis is that this was an older study population, so naturally if you’re inhaling smoke, the first organ that’s impacted in an older population is the lungs,” Dr. Rappold said. “If you go to the hospital for asthma, wheezing, or bronchitis, you are taken out of the risk pool for cardiovascular and other diseases. That could explain why in other studies we don’t see a clear cardiovascular signal as we have for air pollution studies in general. Another aspect to this study is, the exposure metric was PM2.5, but smoke contains many other components, particularly gases, which are respiratory irritants. It could be that this triggers a higher risk for respiratory [effects] than regular episodes of high PM2.5 exposure, just because of the additional gases that people are exposed to.”
Another complicating factor is the paucity of data about solutions to long-term exposure to wildfire smoke. “If you’re impacted by high-exposure levels for 60 days, that is not something we have experienced before,” Dr. Rappold noted. “What are the solutions for that community? What works? Can we show that by implementing community-level resilience plans with HEPA [high-efficiency particulate air] filters or other interventions, do the overall outcomes improve? Doctors are the first ones to talk with their patients about their symptoms and about how to take care of their conditions. They can clearly make a difference in emphasizing reducing exposures in a way that fits their patients individually, either reducing the amount of time spent outside, the duration of exposure, and the level of exposure. Maybe change activities based on the intensity of exposure. Don’t go for a run outside when it’s smoky, because your ventilation rate is higher and you will breathe in more smoke. Become aware of those things.”
Advising vulnerable patients
While research in this field advances, the unforgiving wildfire season looms, assuring more destruction of property and threats to cardiorespiratory health. “There are a lot of questions that research will have an opportunity to address as we go forward, including the utility and the benefit of N95 masks, the utility of HEPA filters used in the house, and even with HVAC [heating, ventilation, and air conditioning] systems,” Dr. Cascio said. “Can we really clean up the indoor air well enough to protect us from wildfire smoke?”
The way he sees it, the time is ripe for clinicians and officials in public and private practice settings to refine how they distribute information to people living in areas affected by wildfire smoke. “We can’t force people do anything, but at least if they’re informed, then they understand they can make an informed decision about how they might want to affect what they do that would limit their exposure,” he said. “As a patient, my health care system sends text and email messages to me. So, why couldn’t the hospital send out a text message or an email to all of the patients with COPD, coronary disease, and heart failure when an area is impacted by smoke, saying, ‘Check your air quality and take action if air quality is poor?’ Physicians don’t have time to do this kind of education in the office for all of their patients. I know that from experience. But if one were to only focus on those at highest risk, and encourage them to follow our guidelines, which might include doing HEPA filter treatment in the home, we probably would reduce the number of clinical events in a cost-effective way.”
The 2019 wildfire season is underway in many locales across the United States, exposing millions of individuals to smoky conditions that will have health consequences ranging from stinging eyes to scratchy throats to a trip to the ED for asthma or chronic obstructive pulmonary disease (COPD) exacerbation. Questions about long-term health impacts are on the minds of many, including physicians and their patients who live with cardiorespiratory conditions.

John R. Balmes, MD, a pulmonologist at the University of California, San Francisco, and an expert on the respiratory and cardiovascular effects of air pollutants, suggested that the best available published literature points to “pretty strong evidence for acute effects of wildfire smoke on respiratory health, meaning people with preexisting asthma and COPD are at risk for exacerbations, and probably for respiratory tract infections as well.” He said, “It’s a little less clear, but there’s good biological plausibility for increased risk of respiratory tract infections because when your alveolar macrophages are overloaded with carbon particles that are toxic to those cells, they don’t function as well as a first line of defense against bacterial infection, for example.”
The new normal of wildfires
Warmer, drier summers in recent years in the western United States and many other regions, attributed by climate experts to global climate change, have produced catastrophic wildfires (PNAS;2016 Oct 18;113[42]11770-5; Science 2006 Aug 18;313:940-3). The Camp Fire in Northern California broke out in November 2018, took the lives of at least 85 people, and cost more than $16 billion in damage. Smoke from that blaze reached hazardous levels in San Francisco, Sacramento, Fresno, and many other smaller towns. Other forest fires in that year caused heavy smoke conditions in Portland, Seattle, Vancouver, and Anchorage. Such events are expected to be repeated often in the coming years (Int J Environ Res Public Health. 2019 Jul 6;16[13]).

Wildfire smoke can contain a wide range of substances, chemicals, and gases with known and unknown cardiorespiratory implications. “Smoke is composed primarily of carbon dioxide, water vapor, carbon monoxide, particulate matter, hydrocarbons and other organic chemicals, nitrogen oxides, trace minerals and several thousand other compounds,” according to the U.S. Environmental Protection Agency (Wildfire smoke: A guide for public health officials 2019. Washington, D.C.: EPA, 2019). The EPA report noted, “Particles with diameters less than 10 mcm (particulate matter, or PM10) can be inhaled into the lungs and affect the lungs, heart, and blood vessels. The smallest particles, those less than 2.5 mcm in diameter (PM2.5), are the greatest risk to public health because they can reach deep into the lungs and may even make it into the bloodstream.”
Research on health impact
In early June of 2008, Wayne Cascio, MD, awoke in his Greenville, N.C., home to the stench of smoke emanating from a large peat fire burning some 65 miles away. By the time he reached the parking lot at East Carolina University in Greenville to begin his workday as chief of cardiology, the haze of smoke had thickened to the point where he could only see a few feet in front of him.
Over the next several weeks, the fire scorched 41,000 acres and produced haze and air pollution that far exceeded National Ambient Air Quality Standards for particulate matter and blanketed rural communities in the state’s eastern region. The price tag for management of the blaze reached $20 million. Because of his interest in the health effects of wildfire smoke and because of his relationship with investigators at the EPA, Dr. Cascio initiated an epidemiology study to investigate the effects of exposure on cardiorespiratory outcomes in the population affected by the fire (Environ Health Perspect. 2011 Oct;119[10]:1415-20).
By combining satellite data with syndromic surveillance drawn from hospital records in 41 counties contained in the North Carolina Disease Event Tracking and Epidemiologic Collection Tool, he and his colleagues found that exposure to the peat wildfire smoke led to increases in the cumulative risk ratio for asthma (relative risk, 1.65), chronic obstructive pulmonary disease (RR, 1.73), and pneumonia and acute bronchitis (RR, 1.59). ED visits related to cardiopulmonary symptoms and heart failure also were significantly increased (RR, 1.23 and 1.37, respectively). “That was really the first study to strongly identify a cardiac endpoint related to wildfire smoke exposure,” said Dr. Cascio, who now directs the EPA’s National Health and Environmental Effects Research Laboratory. “It really pointed out how little we knew about the health effects of wildfire up until that time.”
Those early findings have been replicated in subsequent research about the acute health effects of exposure to wildfire smoke, which contains PM2.5 and other toxic substances from structures, electronic devices, and automobiles destroyed in the path of flames, including heavy metals and asbestos. Most of the work has focused on smoke-related cardiovascular and respiratory ED visits and hospitalizations.
A study of the 2008 California wildfire impact on ED visits accounted for ozone levels in addition to PM2.5 in the smoke. During the active fire periods, PM2.5 was significantly associated with exacerbations of asthma and COPD and these effects remained after controlling for ozone levels. PM2.5 inhalation during the wildfires was associated with increased risk of an ED visit for asthma (RR, 1.112; 95% confidence interval, 1.087-1.138) for a 10 mcg/m3 increase in PM2.5 and COPD (RR, 1.05; 95% CI, 1.019-1.0825), as well as for combined respiratory visits (RR, 1.035; 95% CI, 1.023-1.046) (Environ Int. 2109 Aug;129:291-8).
Researchers who evaluated the health impacts of wildfires in California during the 2015 fire season found an increase in all-cause cardiovascular and respiratory ED visits, especially among those aged 65 years and older during smoke days. The population-based study included 1,196,233 ED visits during May 1–Sept. 30 that year. PM2.5 concentrations were categorized as light, medium, or dense. Relative risk rose with the amount of smoke in the air. Rates of all-cause cardiovascular ED visits were elevated across levels of smoke density, with the greatest increase on dense smoke days and among those aged 65 years or older (RR,1.15; 95% CI, 1.09-1.22). All-cause cerebrovascular visits were associated with dense smoke days, especially among those aged 65 years and older (RR, 1.22; 95% CI, 1.00-1.49). Respiratory conditions also were increased on dense smoke days (RR, 1.18; 95% CI, 1.08-1.28) (J Am Heart Assoc. 2018 Apr 11;7:e007492. doi: 10.1161/JAHA.117.007492).
Long-term effects unknown
When it comes to the long-term effects of wildfire smoke on human health outcomes, much less is known. In a recent literature review, Colleen E. Reid, PhD, and Melissa May Maestas, PhD, found only one study that investigated long-term respiratory health impacts of wildfire smoke, and only a few studies that have estimated future health impacts of wildfires under likely climate change scenarios (Curr Opin Pulm Med. 2019 Mar;25:179-87).
“We know that there are immediate respiratory health effects from wildfire smoke,” said Dr. Reid of the department of geography at the University of Colorado Boulder. “What’s less known is everything else. That’s challenging, because people want to know about the long-term health effects.”
Evidence from the scientific literature suggests that exposure to air pollution adversely affects cardiovascular health, but whether exposure to wildfire smoke confers a similar risk is less clear. “Until just a few years ago we haven’t been able to study wildfire exposure measures on a large scale,” said EPA scientist Ana G. Rappold, PhD, a statistician there in the environmental public health division of the National Health and Environmental Effects Research Laboratory. “It’s also hard to predict wildfires, so it’s hard to plan for an epidemiologic study if you don’t know where they’re going to occur.”
Dr. Rappold and colleagues examined cardiopulmonary hospitalizations among adults aged 65 years and older in 692 U.S. counties within 200 km of 123 large wildfires during 2008-2010 (Environ Health Perspect. 2019;127[3]:37006. doi: 10.1289/EHP3860). They observed that an increased risk of PM2.5-related cardiopulmonary hospitalizations was similar on smoke and nonsmoke days across multiple lags and exposure metrics, while risk for asthma-related hospitalizations was higher during smoke days. “One hypothesis is that this was an older study population, so naturally if you’re inhaling smoke, the first organ that’s impacted in an older population is the lungs,” Dr. Rappold said. “If you go to the hospital for asthma, wheezing, or bronchitis, you are taken out of the risk pool for cardiovascular and other diseases. That could explain why in other studies we don’t see a clear cardiovascular signal as we have for air pollution studies in general. Another aspect to this study is, the exposure metric was PM2.5, but smoke contains many other components, particularly gases, which are respiratory irritants. It could be that this triggers a higher risk for respiratory [effects] than regular episodes of high PM2.5 exposure, just because of the additional gases that people are exposed to.”
Another complicating factor is the paucity of data about solutions to long-term exposure to wildfire smoke. “If you’re impacted by high-exposure levels for 60 days, that is not something we have experienced before,” Dr. Rappold noted. “What are the solutions for that community? What works? Can we show that by implementing community-level resilience plans with HEPA [high-efficiency particulate air] filters or other interventions, do the overall outcomes improve? Doctors are the first ones to talk with their patients about their symptoms and about how to take care of their conditions. They can clearly make a difference in emphasizing reducing exposures in a way that fits their patients individually, either reducing the amount of time spent outside, the duration of exposure, and the level of exposure. Maybe change activities based on the intensity of exposure. Don’t go for a run outside when it’s smoky, because your ventilation rate is higher and you will breathe in more smoke. Become aware of those things.”
Advising vulnerable patients
While research in this field advances, the unforgiving wildfire season looms, assuring more destruction of property and threats to cardiorespiratory health. “There are a lot of questions that research will have an opportunity to address as we go forward, including the utility and the benefit of N95 masks, the utility of HEPA filters used in the house, and even with HVAC [heating, ventilation, and air conditioning] systems,” Dr. Cascio said. “Can we really clean up the indoor air well enough to protect us from wildfire smoke?”
The way he sees it, the time is ripe for clinicians and officials in public and private practice settings to refine how they distribute information to people living in areas affected by wildfire smoke. “We can’t force people do anything, but at least if they’re informed, then they understand they can make an informed decision about how they might want to affect what they do that would limit their exposure,” he said. “As a patient, my health care system sends text and email messages to me. So, why couldn’t the hospital send out a text message or an email to all of the patients with COPD, coronary disease, and heart failure when an area is impacted by smoke, saying, ‘Check your air quality and take action if air quality is poor?’ Physicians don’t have time to do this kind of education in the office for all of their patients. I know that from experience. But if one were to only focus on those at highest risk, and encourage them to follow our guidelines, which might include doing HEPA filter treatment in the home, we probably would reduce the number of clinical events in a cost-effective way.”
Benefits of peanut desensitization may not last
based on data from a phase 2 randomized trial of individuals with confirmed peanut allergies.
Previous studies have shown that desensitization to peanuts can be successful, but sustained response to oral immunotherapy after treatment reduction or discontinuation has not been well studied, wrote R. Sharon Chinthrajah, MD, of Stanford (Calif.)University, and colleagues.
“We found that OIT with peanut was able to desensitise people with peanut allergy to 4,000 mg of peanut protein, but that discontinuation of peanut, or even a reduction to 300 mg daily, increased the likelihood of regaining clinical reactivity to peanut,” they wrote. “With peanut allergy therapies in varying stages of clinical development, and some nearing [Food and Drug Administration] approval, vital questions remain regarding the durability of treatment effects and the appropriate maintenance doses.”
In the Peanut Oral Immunotherapy Study: Safety Efficacy and Discovery (POISED), published in The Lancet, the researchers randomized 120 participants to three groups:
• 60 patients built up to a maintenance dose of 4,000 mg of peanut protein for 104 weeks followed by total discontinuation (peanut-0).
• 35 patients built up to a maintenance dose of 4,000 mg of peanut protein for 104 weeks followed by a 300-mg maintenance dose of peanut protein in the form of peanut flour (peanut-300).
• 25 patients to an oat flour placebo.
All participants were trained on how and when to use epinephrine autoinjector devices to treat allergic symptoms such as respiratory problems (cough, shortness of breath, or change in voice), widespread hives or erythema, repetitive vomiting, persistent abdominal pain, angioedema of the face, or feeling faint.
The primary outcome was passing a double-blind, placebo-controlled, food challenge (DBPCFC) to 4,000 mg of peanut protein, which was measured at baseline and at weeks 104, 117, 130, 143, and 156.
Overall, 35% of the peanut-0 group passed the challenge at 104 and 117 weeks, compared with 4% of the placebo group. At week 156 after discontinuing OIT, 13% of the peanut-0 group met the DBPCFC challenge, compared with 4% of the placebo group. However, 37% of participants randomized to a reduced peanut protein dose of 300 mg passed the challenge at 156 weeks, suggesting that more data are needed on optimal maintenance dosing strategies.
Baseline demographics were similar across all groups. The median age at study enrollment was 11 years and the median allergy duration was 9 years. The most common adverse events were mild gastrointestinal and respiratory problems. Adverse events decreased over time in all three groups.
“Higher levels of peanut-specific IgE to total IgE ratio, peanut sIgE, Ara h 1, Ara h 2, and Ara h 1 IgE to peanut-specific IgE ratio at baseline in participants were associated with increased frequencies of adverse events during active peanut OIT,” the researchers noted.
The study findings were limited by several factors including the ability of participants to tolerate 4,000 mg of peanut protein after achieving a maintenance dose but conducting serial testing only for those who passed the challenge. In addition, the results may be limited to peanut and not generalizable to other food allergies, the researchers said.
However, the results suggest that OIT remains a promising treatment for peanut allergies, and the association of biomarkers with clinical outcomes “might help the practitioner in identifying good candidates for OIT and those individuals who warrant increased vigilance against allergic reactions during OIT,” they said.
The National Institutes of Health supported the study. The researchers had no financial conflicts to disclose.
SOURCE: Chinthrajah RS et al. Lancet. 2019 Sep 12. doi: 10.1016/S0140-6736(19)31793-3.
based on data from a phase 2 randomized trial of individuals with confirmed peanut allergies.
Previous studies have shown that desensitization to peanuts can be successful, but sustained response to oral immunotherapy after treatment reduction or discontinuation has not been well studied, wrote R. Sharon Chinthrajah, MD, of Stanford (Calif.)University, and colleagues.
“We found that OIT with peanut was able to desensitise people with peanut allergy to 4,000 mg of peanut protein, but that discontinuation of peanut, or even a reduction to 300 mg daily, increased the likelihood of regaining clinical reactivity to peanut,” they wrote. “With peanut allergy therapies in varying stages of clinical development, and some nearing [Food and Drug Administration] approval, vital questions remain regarding the durability of treatment effects and the appropriate maintenance doses.”
In the Peanut Oral Immunotherapy Study: Safety Efficacy and Discovery (POISED), published in The Lancet, the researchers randomized 120 participants to three groups:
• 60 patients built up to a maintenance dose of 4,000 mg of peanut protein for 104 weeks followed by total discontinuation (peanut-0).
• 35 patients built up to a maintenance dose of 4,000 mg of peanut protein for 104 weeks followed by a 300-mg maintenance dose of peanut protein in the form of peanut flour (peanut-300).
• 25 patients to an oat flour placebo.
All participants were trained on how and when to use epinephrine autoinjector devices to treat allergic symptoms such as respiratory problems (cough, shortness of breath, or change in voice), widespread hives or erythema, repetitive vomiting, persistent abdominal pain, angioedema of the face, or feeling faint.
The primary outcome was passing a double-blind, placebo-controlled, food challenge (DBPCFC) to 4,000 mg of peanut protein, which was measured at baseline and at weeks 104, 117, 130, 143, and 156.
Overall, 35% of the peanut-0 group passed the challenge at 104 and 117 weeks, compared with 4% of the placebo group. At week 156 after discontinuing OIT, 13% of the peanut-0 group met the DBPCFC challenge, compared with 4% of the placebo group. However, 37% of participants randomized to a reduced peanut protein dose of 300 mg passed the challenge at 156 weeks, suggesting that more data are needed on optimal maintenance dosing strategies.
Baseline demographics were similar across all groups. The median age at study enrollment was 11 years and the median allergy duration was 9 years. The most common adverse events were mild gastrointestinal and respiratory problems. Adverse events decreased over time in all three groups.
“Higher levels of peanut-specific IgE to total IgE ratio, peanut sIgE, Ara h 1, Ara h 2, and Ara h 1 IgE to peanut-specific IgE ratio at baseline in participants were associated with increased frequencies of adverse events during active peanut OIT,” the researchers noted.
The study findings were limited by several factors including the ability of participants to tolerate 4,000 mg of peanut protein after achieving a maintenance dose but conducting serial testing only for those who passed the challenge. In addition, the results may be limited to peanut and not generalizable to other food allergies, the researchers said.
However, the results suggest that OIT remains a promising treatment for peanut allergies, and the association of biomarkers with clinical outcomes “might help the practitioner in identifying good candidates for OIT and those individuals who warrant increased vigilance against allergic reactions during OIT,” they said.
The National Institutes of Health supported the study. The researchers had no financial conflicts to disclose.
SOURCE: Chinthrajah RS et al. Lancet. 2019 Sep 12. doi: 10.1016/S0140-6736(19)31793-3.
based on data from a phase 2 randomized trial of individuals with confirmed peanut allergies.
Previous studies have shown that desensitization to peanuts can be successful, but sustained response to oral immunotherapy after treatment reduction or discontinuation has not been well studied, wrote R. Sharon Chinthrajah, MD, of Stanford (Calif.)University, and colleagues.
“We found that OIT with peanut was able to desensitise people with peanut allergy to 4,000 mg of peanut protein, but that discontinuation of peanut, or even a reduction to 300 mg daily, increased the likelihood of regaining clinical reactivity to peanut,” they wrote. “With peanut allergy therapies in varying stages of clinical development, and some nearing [Food and Drug Administration] approval, vital questions remain regarding the durability of treatment effects and the appropriate maintenance doses.”
In the Peanut Oral Immunotherapy Study: Safety Efficacy and Discovery (POISED), published in The Lancet, the researchers randomized 120 participants to three groups:
• 60 patients built up to a maintenance dose of 4,000 mg of peanut protein for 104 weeks followed by total discontinuation (peanut-0).
• 35 patients built up to a maintenance dose of 4,000 mg of peanut protein for 104 weeks followed by a 300-mg maintenance dose of peanut protein in the form of peanut flour (peanut-300).
• 25 patients to an oat flour placebo.
All participants were trained on how and when to use epinephrine autoinjector devices to treat allergic symptoms such as respiratory problems (cough, shortness of breath, or change in voice), widespread hives or erythema, repetitive vomiting, persistent abdominal pain, angioedema of the face, or feeling faint.
The primary outcome was passing a double-blind, placebo-controlled, food challenge (DBPCFC) to 4,000 mg of peanut protein, which was measured at baseline and at weeks 104, 117, 130, 143, and 156.
Overall, 35% of the peanut-0 group passed the challenge at 104 and 117 weeks, compared with 4% of the placebo group. At week 156 after discontinuing OIT, 13% of the peanut-0 group met the DBPCFC challenge, compared with 4% of the placebo group. However, 37% of participants randomized to a reduced peanut protein dose of 300 mg passed the challenge at 156 weeks, suggesting that more data are needed on optimal maintenance dosing strategies.
Baseline demographics were similar across all groups. The median age at study enrollment was 11 years and the median allergy duration was 9 years. The most common adverse events were mild gastrointestinal and respiratory problems. Adverse events decreased over time in all three groups.
“Higher levels of peanut-specific IgE to total IgE ratio, peanut sIgE, Ara h 1, Ara h 2, and Ara h 1 IgE to peanut-specific IgE ratio at baseline in participants were associated with increased frequencies of adverse events during active peanut OIT,” the researchers noted.
The study findings were limited by several factors including the ability of participants to tolerate 4,000 mg of peanut protein after achieving a maintenance dose but conducting serial testing only for those who passed the challenge. In addition, the results may be limited to peanut and not generalizable to other food allergies, the researchers said.
However, the results suggest that OIT remains a promising treatment for peanut allergies, and the association of biomarkers with clinical outcomes “might help the practitioner in identifying good candidates for OIT and those individuals who warrant increased vigilance against allergic reactions during OIT,” they said.
The National Institutes of Health supported the study. The researchers had no financial conflicts to disclose.
SOURCE: Chinthrajah RS et al. Lancet. 2019 Sep 12. doi: 10.1016/S0140-6736(19)31793-3.
FROM THE LANCET