User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
ACIP favors shared decision on pneumococcal vaccine for older adults
Pneumococcal vaccination with the 13-valent pneumococcal conjugate vaccine (PCV13) based on shared clinical decision making is recommended for immunocompetent adults aged 65 years and older who have not previously received PCV13, and all adults aged 65 years and older should continue to receive the pneumococcal polysaccharide vaccine (PPSV23), according to a vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The motion passed with an 11-1 vote after members voted down two other options to either discontinue or continue the current recommendation of PCV13 for all immunocompetent adults aged 65 years and older. The current recommendation for PCV13 for adults aged 65 years and older has been in place since 2014.
The pneumococcal work group assessed indirect effects of the pediatric PCV vaccination on older adults prior to 2014 and since 2014, and what additional benefits might be expected if routine vaccination of older adults continued.
“Indirect effects have been observed in all age groups” said Almea Matanock, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. Although there were no safety concerns, the public health impact of continued vaccination of adults was minimal.
Although PCV13 resulted in a 75% reduction in vaccine-type invasive pneumococcal disease and a 45% reduction in vaccine-type nonbacteremic pneumonia in 2014, the annual number needed to vaccinate to prevent a single case of outpatient pneumonia was 2,600, said Dr. Matanock.
Dr. Matanock presented key issues from the Evidence to Recommendations Framework for and against the recommendation for PCV13 in older adults. Work group comments in favor of continuing the recommendation for PCV13 in older adults included effective disease prevention and the potential negative impact on the importance of adult vaccines if the vaccine was no longer recommended. However, some work group members and committee members expressed concern about resource allocation and steering vaccines away from younger age groups in whom they have been more consistently effective.
Paul Hunter, MD, of the City of Milwaukee Health Department, voted against the shared clinical decision making, and instead favored discontinuing the recommendation for PCV13 for older adults. “I think clinicians need a clear message,” he said, adding that “the public health bang for the buck is with the kids.”
“I think there was a recognition that the population level benefit is minimal,” said work group chair Grace Lee, MD.
Although the work group recognized some benefit for older adults, the burden of disease for PCV-specific disease is low, compared with all-cause pneumonia, said Dr. Lee of Lucile Packard Children’s Hospital at Stanford, Calif. However, the recommendation for shared clinical decision making allows for potential insurance coverage of the vaccine for adults who decide after discussion with their health care provider that they would benefit.
“We are still unpacking this construct” of shared clinical decision making, which in this case applies to adults without immunocompromising conditions, and is more of a provider assessment than a risk assessment, she said.
The ACIP members had no financial conflicts to disclose.
Pneumococcal vaccination with the 13-valent pneumococcal conjugate vaccine (PCV13) based on shared clinical decision making is recommended for immunocompetent adults aged 65 years and older who have not previously received PCV13, and all adults aged 65 years and older should continue to receive the pneumococcal polysaccharide vaccine (PPSV23), according to a vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The motion passed with an 11-1 vote after members voted down two other options to either discontinue or continue the current recommendation of PCV13 for all immunocompetent adults aged 65 years and older. The current recommendation for PCV13 for adults aged 65 years and older has been in place since 2014.
The pneumococcal work group assessed indirect effects of the pediatric PCV vaccination on older adults prior to 2014 and since 2014, and what additional benefits might be expected if routine vaccination of older adults continued.
“Indirect effects have been observed in all age groups” said Almea Matanock, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. Although there were no safety concerns, the public health impact of continued vaccination of adults was minimal.
Although PCV13 resulted in a 75% reduction in vaccine-type invasive pneumococcal disease and a 45% reduction in vaccine-type nonbacteremic pneumonia in 2014, the annual number needed to vaccinate to prevent a single case of outpatient pneumonia was 2,600, said Dr. Matanock.
Dr. Matanock presented key issues from the Evidence to Recommendations Framework for and against the recommendation for PCV13 in older adults. Work group comments in favor of continuing the recommendation for PCV13 in older adults included effective disease prevention and the potential negative impact on the importance of adult vaccines if the vaccine was no longer recommended. However, some work group members and committee members expressed concern about resource allocation and steering vaccines away from younger age groups in whom they have been more consistently effective.
Paul Hunter, MD, of the City of Milwaukee Health Department, voted against the shared clinical decision making, and instead favored discontinuing the recommendation for PCV13 for older adults. “I think clinicians need a clear message,” he said, adding that “the public health bang for the buck is with the kids.”
“I think there was a recognition that the population level benefit is minimal,” said work group chair Grace Lee, MD.
Although the work group recognized some benefit for older adults, the burden of disease for PCV-specific disease is low, compared with all-cause pneumonia, said Dr. Lee of Lucile Packard Children’s Hospital at Stanford, Calif. However, the recommendation for shared clinical decision making allows for potential insurance coverage of the vaccine for adults who decide after discussion with their health care provider that they would benefit.
“We are still unpacking this construct” of shared clinical decision making, which in this case applies to adults without immunocompromising conditions, and is more of a provider assessment than a risk assessment, she said.
The ACIP members had no financial conflicts to disclose.
Pneumococcal vaccination with the 13-valent pneumococcal conjugate vaccine (PCV13) based on shared clinical decision making is recommended for immunocompetent adults aged 65 years and older who have not previously received PCV13, and all adults aged 65 years and older should continue to receive the pneumococcal polysaccharide vaccine (PPSV23), according to a vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The motion passed with an 11-1 vote after members voted down two other options to either discontinue or continue the current recommendation of PCV13 for all immunocompetent adults aged 65 years and older. The current recommendation for PCV13 for adults aged 65 years and older has been in place since 2014.
The pneumococcal work group assessed indirect effects of the pediatric PCV vaccination on older adults prior to 2014 and since 2014, and what additional benefits might be expected if routine vaccination of older adults continued.
“Indirect effects have been observed in all age groups” said Almea Matanock, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. Although there were no safety concerns, the public health impact of continued vaccination of adults was minimal.
Although PCV13 resulted in a 75% reduction in vaccine-type invasive pneumococcal disease and a 45% reduction in vaccine-type nonbacteremic pneumonia in 2014, the annual number needed to vaccinate to prevent a single case of outpatient pneumonia was 2,600, said Dr. Matanock.
Dr. Matanock presented key issues from the Evidence to Recommendations Framework for and against the recommendation for PCV13 in older adults. Work group comments in favor of continuing the recommendation for PCV13 in older adults included effective disease prevention and the potential negative impact on the importance of adult vaccines if the vaccine was no longer recommended. However, some work group members and committee members expressed concern about resource allocation and steering vaccines away from younger age groups in whom they have been more consistently effective.
Paul Hunter, MD, of the City of Milwaukee Health Department, voted against the shared clinical decision making, and instead favored discontinuing the recommendation for PCV13 for older adults. “I think clinicians need a clear message,” he said, adding that “the public health bang for the buck is with the kids.”
“I think there was a recognition that the population level benefit is minimal,” said work group chair Grace Lee, MD.
Although the work group recognized some benefit for older adults, the burden of disease for PCV-specific disease is low, compared with all-cause pneumonia, said Dr. Lee of Lucile Packard Children’s Hospital at Stanford, Calif. However, the recommendation for shared clinical decision making allows for potential insurance coverage of the vaccine for adults who decide after discussion with their health care provider that they would benefit.
“We are still unpacking this construct” of shared clinical decision making, which in this case applies to adults without immunocompromising conditions, and is more of a provider assessment than a risk assessment, she said.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
FDA approves dupilumab for chronic rhinosinusitis with nasal polyps
The Food and Drug Administration has approved dupilumab (Dupixent) for the treatment of chronic rhinosinusitis accompanied by nasal polyps in adults.
FDA approval is based on results from a pair of studies involving 724 patients aged 18 years or older with chronic rhinosinusitis with nasal polyps who were symptomatic despite undergoing treatment with intranasal corticosteroids and who received either dupilumab or a placebo. Compared with the placebo group, patients receiving dupilumab had statistically significant reductions in nasal polyp size and nasal congestion; they also had an increased ability to smell and required less nasal polyp surgery and oral steroids.
The most commonly reported adverse events were injection-site reactions and eye and eyelid inflammation, which included redness, swelling, and itching. The drug can cause severe allergic reactions and eye problems, such as conjunctivitis or keratitis; patients should also not receive live vaccines while taking dupilumab.
“Nasal polyps can lead to loss of smell, and often patients require surgery to remove the polyps. Dupixent provides an important treatment option for patients whose nasal polyps are not adequately controlled with intranasal steroids. It also reduces the need for nasal polyp surgery and oral steroids,” said Sally Seymour, MD, director of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Find the full press release on the FDA website.
The Food and Drug Administration has approved dupilumab (Dupixent) for the treatment of chronic rhinosinusitis accompanied by nasal polyps in adults.
FDA approval is based on results from a pair of studies involving 724 patients aged 18 years or older with chronic rhinosinusitis with nasal polyps who were symptomatic despite undergoing treatment with intranasal corticosteroids and who received either dupilumab or a placebo. Compared with the placebo group, patients receiving dupilumab had statistically significant reductions in nasal polyp size and nasal congestion; they also had an increased ability to smell and required less nasal polyp surgery and oral steroids.
The most commonly reported adverse events were injection-site reactions and eye and eyelid inflammation, which included redness, swelling, and itching. The drug can cause severe allergic reactions and eye problems, such as conjunctivitis or keratitis; patients should also not receive live vaccines while taking dupilumab.
“Nasal polyps can lead to loss of smell, and often patients require surgery to remove the polyps. Dupixent provides an important treatment option for patients whose nasal polyps are not adequately controlled with intranasal steroids. It also reduces the need for nasal polyp surgery and oral steroids,” said Sally Seymour, MD, director of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Find the full press release on the FDA website.
The Food and Drug Administration has approved dupilumab (Dupixent) for the treatment of chronic rhinosinusitis accompanied by nasal polyps in adults.
FDA approval is based on results from a pair of studies involving 724 patients aged 18 years or older with chronic rhinosinusitis with nasal polyps who were symptomatic despite undergoing treatment with intranasal corticosteroids and who received either dupilumab or a placebo. Compared with the placebo group, patients receiving dupilumab had statistically significant reductions in nasal polyp size and nasal congestion; they also had an increased ability to smell and required less nasal polyp surgery and oral steroids.
The most commonly reported adverse events were injection-site reactions and eye and eyelid inflammation, which included redness, swelling, and itching. The drug can cause severe allergic reactions and eye problems, such as conjunctivitis or keratitis; patients should also not receive live vaccines while taking dupilumab.
“Nasal polyps can lead to loss of smell, and often patients require surgery to remove the polyps. Dupixent provides an important treatment option for patients whose nasal polyps are not adequately controlled with intranasal steroids. It also reduces the need for nasal polyp surgery and oral steroids,” said Sally Seymour, MD, director of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Find the full press release on the FDA website.
Tocilizumab preserves lung function in systemic sclerosis
MADRID – , according to a secondary endpoint analysis of the phase 3, double-blind, randomized, controlled focuSSced trial.

After 48 weeks, a significantly lower proportion of patients treated with tocilizumab than placebo experienced any decline in lung function from baseline (50.5% versus 70.3% (P = .015), as defined by the percentage increase in predicted forced vital capacity (%pFVC). When only patients with interstitial lung disease (ILD) were considered, the respective percentages were 51.7% and 75.5% (P = .003).
In SSc-ILD patients, a clinically meaningful decline of 10% or more of the %pFVC in lung function was seen in 24.5% given placebo but in just 8.6% of those treated with tocilizumab.
“ILD is a major complication of scleroderma; it has high morbidity and mortality ... and it’s largely irreversible,” Dinesh Khanna, MD, said at the European Congress of Rheumatology.
“In this day and age, when we treat ILD, we wait for a patient to develop clinical ILD,” added Dr. Khanna, director of the scleroderma program at the University of Michigan, Ann Arbor. Clinical ILD can be defined by symptoms, abnormal pulmonary function tests, and marked abnormalities on high resolution computed tomography (HRCT) scans. He indicated that if improving ILD was not possible, then the next best thing would be to stabilize the disease and ensure there was no worsening in lung function.
As yet, there are no disease-modifying treatments available to treat SSc but there are “ample data that interleukin-6 plays a very important role in the pathogenesis of scleroderma,” Dr. Khanna observed. Tocilizumab is a humanized monoclonal antibody against the interleukin-6 receptor.
Data from the phase 2 faSScinate trial showed initial promise for the drug in SSc where a numerical, but not statistically significant, improvement in skin thickening was seen, and the results had hinted at a possible benefit on lung function (Lancet. 2016 Jun 25;387:2630-40).
However, in the phase 3 focuSSced trial, there was no statistically significant difference in the change from baseline to week 48 modified Rodnan skin score (mRSS) between tocilizumab and placebo, which was the primary endpoint. The least square mean change in mRSS was –6.14 for tocilizumab and –4.41 for placebo (P = .0983).
A total of 205 patients with SSc were studied and randomized, 1:1 in a double-blind fashion, to receive either a once-weekly, subcutaneous dose of 162 mg tocilizumab or a weekly subcutaneous placebo injection for 48 weeks.
For inclusion in the study, patients had to have SSc that met American College of Rheumatology and European League Against Rheumatism (EULAR) criteria and be diagnosed less than 60 months previously. Patients had to have an mRSS of 10-35 units and active disease with one or more of the following: C-reactive protein of 6 mg/L or higher; erythrocyte sedimentation rate of 28 mm/h or higher; and platelet count of330 x 109 L.
“What was astonishing in the trial was that every patient had HRCT at baseline and at the end of the study,” Dr. Khanna reported. These scans showed that 64% of patients had evidence of ILD at baseline and that those treated with tocilizumab had less evidence of fibrosis at week 48 versus placebo, indicating a stabilization rather than worsening of disease.
A time to treatment failure analysis also favored tocilizumab over placebo, but there were no significant changes in patient-reported outcomes.
Dr. Khanna’s slides stated that “given that the primary endpoint for mRSS was not met, all other P values are presented for information purposes only and cannot be considered statistically significant despite the strength of the evidence.” During the Q&A after his presentation, he noted that it was unlikely that the study’s sponsors (Roche/Genentech) will now pursue a license for tocilizumab in SSc.
Nevertheless, Dr. Khanna concluded, “we have the opportunity, based on these data, to treat these patients early on, where you can preserve the lung function, which is a paradigm shift versus waiting for the lung function to decline, become clinically meaningful, significant, and then treat this patient population.”
Roche/Genentech sponsored the study. Dr. Khanna acts as a consultant to Roche/Genentech and eight other pharmaceutical companies. He owns stock in Eicos Sciences.
SOURCE: Khanna D et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):202-3. Abstract OP0245, doi: 10.1136/annrheumdis-2019-eular.2120
MADRID – , according to a secondary endpoint analysis of the phase 3, double-blind, randomized, controlled focuSSced trial.

After 48 weeks, a significantly lower proportion of patients treated with tocilizumab than placebo experienced any decline in lung function from baseline (50.5% versus 70.3% (P = .015), as defined by the percentage increase in predicted forced vital capacity (%pFVC). When only patients with interstitial lung disease (ILD) were considered, the respective percentages were 51.7% and 75.5% (P = .003).
In SSc-ILD patients, a clinically meaningful decline of 10% or more of the %pFVC in lung function was seen in 24.5% given placebo but in just 8.6% of those treated with tocilizumab.
“ILD is a major complication of scleroderma; it has high morbidity and mortality ... and it’s largely irreversible,” Dinesh Khanna, MD, said at the European Congress of Rheumatology.
“In this day and age, when we treat ILD, we wait for a patient to develop clinical ILD,” added Dr. Khanna, director of the scleroderma program at the University of Michigan, Ann Arbor. Clinical ILD can be defined by symptoms, abnormal pulmonary function tests, and marked abnormalities on high resolution computed tomography (HRCT) scans. He indicated that if improving ILD was not possible, then the next best thing would be to stabilize the disease and ensure there was no worsening in lung function.
As yet, there are no disease-modifying treatments available to treat SSc but there are “ample data that interleukin-6 plays a very important role in the pathogenesis of scleroderma,” Dr. Khanna observed. Tocilizumab is a humanized monoclonal antibody against the interleukin-6 receptor.
Data from the phase 2 faSScinate trial showed initial promise for the drug in SSc where a numerical, but not statistically significant, improvement in skin thickening was seen, and the results had hinted at a possible benefit on lung function (Lancet. 2016 Jun 25;387:2630-40).
However, in the phase 3 focuSSced trial, there was no statistically significant difference in the change from baseline to week 48 modified Rodnan skin score (mRSS) between tocilizumab and placebo, which was the primary endpoint. The least square mean change in mRSS was –6.14 for tocilizumab and –4.41 for placebo (P = .0983).
A total of 205 patients with SSc were studied and randomized, 1:1 in a double-blind fashion, to receive either a once-weekly, subcutaneous dose of 162 mg tocilizumab or a weekly subcutaneous placebo injection for 48 weeks.
For inclusion in the study, patients had to have SSc that met American College of Rheumatology and European League Against Rheumatism (EULAR) criteria and be diagnosed less than 60 months previously. Patients had to have an mRSS of 10-35 units and active disease with one or more of the following: C-reactive protein of 6 mg/L or higher; erythrocyte sedimentation rate of 28 mm/h or higher; and platelet count of330 x 109 L.
“What was astonishing in the trial was that every patient had HRCT at baseline and at the end of the study,” Dr. Khanna reported. These scans showed that 64% of patients had evidence of ILD at baseline and that those treated with tocilizumab had less evidence of fibrosis at week 48 versus placebo, indicating a stabilization rather than worsening of disease.
A time to treatment failure analysis also favored tocilizumab over placebo, but there were no significant changes in patient-reported outcomes.
Dr. Khanna’s slides stated that “given that the primary endpoint for mRSS was not met, all other P values are presented for information purposes only and cannot be considered statistically significant despite the strength of the evidence.” During the Q&A after his presentation, he noted that it was unlikely that the study’s sponsors (Roche/Genentech) will now pursue a license for tocilizumab in SSc.
Nevertheless, Dr. Khanna concluded, “we have the opportunity, based on these data, to treat these patients early on, where you can preserve the lung function, which is a paradigm shift versus waiting for the lung function to decline, become clinically meaningful, significant, and then treat this patient population.”
Roche/Genentech sponsored the study. Dr. Khanna acts as a consultant to Roche/Genentech and eight other pharmaceutical companies. He owns stock in Eicos Sciences.
SOURCE: Khanna D et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):202-3. Abstract OP0245, doi: 10.1136/annrheumdis-2019-eular.2120
MADRID – , according to a secondary endpoint analysis of the phase 3, double-blind, randomized, controlled focuSSced trial.

After 48 weeks, a significantly lower proportion of patients treated with tocilizumab than placebo experienced any decline in lung function from baseline (50.5% versus 70.3% (P = .015), as defined by the percentage increase in predicted forced vital capacity (%pFVC). When only patients with interstitial lung disease (ILD) were considered, the respective percentages were 51.7% and 75.5% (P = .003).
In SSc-ILD patients, a clinically meaningful decline of 10% or more of the %pFVC in lung function was seen in 24.5% given placebo but in just 8.6% of those treated with tocilizumab.
“ILD is a major complication of scleroderma; it has high morbidity and mortality ... and it’s largely irreversible,” Dinesh Khanna, MD, said at the European Congress of Rheumatology.
“In this day and age, when we treat ILD, we wait for a patient to develop clinical ILD,” added Dr. Khanna, director of the scleroderma program at the University of Michigan, Ann Arbor. Clinical ILD can be defined by symptoms, abnormal pulmonary function tests, and marked abnormalities on high resolution computed tomography (HRCT) scans. He indicated that if improving ILD was not possible, then the next best thing would be to stabilize the disease and ensure there was no worsening in lung function.
As yet, there are no disease-modifying treatments available to treat SSc but there are “ample data that interleukin-6 plays a very important role in the pathogenesis of scleroderma,” Dr. Khanna observed. Tocilizumab is a humanized monoclonal antibody against the interleukin-6 receptor.
Data from the phase 2 faSScinate trial showed initial promise for the drug in SSc where a numerical, but not statistically significant, improvement in skin thickening was seen, and the results had hinted at a possible benefit on lung function (Lancet. 2016 Jun 25;387:2630-40).
However, in the phase 3 focuSSced trial, there was no statistically significant difference in the change from baseline to week 48 modified Rodnan skin score (mRSS) between tocilizumab and placebo, which was the primary endpoint. The least square mean change in mRSS was –6.14 for tocilizumab and –4.41 for placebo (P = .0983).
A total of 205 patients with SSc were studied and randomized, 1:1 in a double-blind fashion, to receive either a once-weekly, subcutaneous dose of 162 mg tocilizumab or a weekly subcutaneous placebo injection for 48 weeks.
For inclusion in the study, patients had to have SSc that met American College of Rheumatology and European League Against Rheumatism (EULAR) criteria and be diagnosed less than 60 months previously. Patients had to have an mRSS of 10-35 units and active disease with one or more of the following: C-reactive protein of 6 mg/L or higher; erythrocyte sedimentation rate of 28 mm/h or higher; and platelet count of330 x 109 L.
“What was astonishing in the trial was that every patient had HRCT at baseline and at the end of the study,” Dr. Khanna reported. These scans showed that 64% of patients had evidence of ILD at baseline and that those treated with tocilizumab had less evidence of fibrosis at week 48 versus placebo, indicating a stabilization rather than worsening of disease.
A time to treatment failure analysis also favored tocilizumab over placebo, but there were no significant changes in patient-reported outcomes.
Dr. Khanna’s slides stated that “given that the primary endpoint for mRSS was not met, all other P values are presented for information purposes only and cannot be considered statistically significant despite the strength of the evidence.” During the Q&A after his presentation, he noted that it was unlikely that the study’s sponsors (Roche/Genentech) will now pursue a license for tocilizumab in SSc.
Nevertheless, Dr. Khanna concluded, “we have the opportunity, based on these data, to treat these patients early on, where you can preserve the lung function, which is a paradigm shift versus waiting for the lung function to decline, become clinically meaningful, significant, and then treat this patient population.”
Roche/Genentech sponsored the study. Dr. Khanna acts as a consultant to Roche/Genentech and eight other pharmaceutical companies. He owns stock in Eicos Sciences.
SOURCE: Khanna D et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):202-3. Abstract OP0245, doi: 10.1136/annrheumdis-2019-eular.2120
REPORTING FROM THE EULAR 2019 CONGRESS
Medicare may best Medicare Advantage at reducing readmissions
Although earlier research may suggest otherwise, traditional new research suggests.
Researchers used what they described as “a novel data linkage” comparing 30-day readmission rates after hospitalization for three major conditions in the Hospital Readmissions Reduction Program for patients using traditional Medicare versus Medicare Advantage. Those conditions included acute MI, heart failure, and pneumonia.
“Our results contrast with those of previous studies that have reported lower or statistically similar readmission rates for Medicare Advantage beneficiaries,” Orestis A. Panagiotou, MD, of Brown University, Providence, R.I., and colleagues wrote in a research report published in Annals of Internal Medicine.
In this retrospective cohort study, the researchers linked data from 2011 to 2014 from the Medicare Provider Analysis and Review (MedPAR) file to the Healthcare Effectiveness Data and Information Set (HEDIS).
The novel linkage found that HEDIS data underreported hospital admissions for acute MI, heart failure, and pneumonia, the researchers stated. “Plans incorrectly excluded hospitalizations that should have qualified for the readmission measure, and readmission rates were substantially higher among incorrectly excluded hospitalizations.”
Despite this, in analyses using the linkage of HEDIS and MedPAR, “Medicare Advantage beneficiaries had higher 30-day risk-adjusted readmission rates after [acute MI, heart failure, and pneumonia] than did traditional Medicare beneficiaries,” the investigators noted.
Patients in Medicare Advantage had lower unadjusted readmission rates compared with those in traditional Medicare (16.6% vs. 17.1% for acute MI; 21.4% vs. 21.7% for heart failure; and 16.3% vs. 16.4% for pneumonia). After standardization, Medicare Advantage patients had higher readmission rates, compared with those in traditional Medicare (17.2% vs. 16.9% for acute MI; 21.7% vs. 21.4% for heart failure; and 16.5% vs. 16.0% for pneumonia).
The study authors added that, while unadjusted readmission rates were higher for traditional Medicare beneficiaries, “the direction of the difference reversed after standardization. This occurred because Medicare Advantage beneficiaries have, on average, a lower expected readmission risk [that is, they are ‘healthier’].” Prior studies have documented that Medicare Advantage plans enroll beneficiaries with fewer comorbid conditions and that high-cost beneficiaries switch out of Medicare Advantage and into traditional Medicare.
The researchers suggested four reasons for the differences between the results in this study versus others that compared patients using Medicare with those using Medicare Advantage. These were that the new study included a more comprehensive data set, analyses with comorbid conditions “from a well-validated model applied by CMS [Centers for Medicare & Medicaid Services],” national data focused on three conditions included in the Hospital Readmissions Reduction Program, and patients discharged to places other than skilled nursing facilities and inpatient rehabilitation facilities.
Authors of an accompanying editorial called for caution to be used in interpreting Medicare Advantage enrollment as causing an increased readmission risk.
“[The] results are sensitive to adjustment for case mix,” wrote Peter Huckfeldt, PhD, of the University of Minnesota, Minneapolis, and Neeraj Sood, PhD, of the University of Southern California, Los Angeles, in the editorial published in Annals of Internal Medicine (2019 June 25. doi:10.7326/M19-1599) “Using diagnosis codes on hospital claims for case-mix adjustments may be increasingly perilous. ... To our knowledge, there is no recent evidence comparing the intensity of diagnostic coding between clinically similar [traditional Medicare] and [Medicare Advantage] hospital admissions, but if [traditional Medicare] enrollees were coded more intensively than [Medicare Advantage] enrollees, this could lead to [traditional Medicare] enrollees having lower risk-adjusted readmission rares due to coding practices.”
The editorialists added that using a cross-sectional comparison of Medicare Advantage and traditional Medicare patients is concerning because a “key challenge in estimating the effect of [Medicare Advantage] is that enrollment is voluntary,” which can lead to a number of analytical concerns.
The researchers concluded that their findings “are concerning because CMS uses HEDIS performance to construct composite quality ratings and assign payment bonuses to Medicare Advantage plans.
“Our study suggests a need for improved monitoring of the accuracy of HEDIS data,” they noted.
The National Institute on Aging provided the primary funding for this study. A number of the authors received grants from the National Institutes of Health during the conduct of the study. No other relevant disclosures were reported.
SOURCE: Panagiotou OA et al. Ann Intern Med. 2019 Jun 25. doi: 10.7326/M18-1795.
Although earlier research may suggest otherwise, traditional new research suggests.
Researchers used what they described as “a novel data linkage” comparing 30-day readmission rates after hospitalization for three major conditions in the Hospital Readmissions Reduction Program for patients using traditional Medicare versus Medicare Advantage. Those conditions included acute MI, heart failure, and pneumonia.
“Our results contrast with those of previous studies that have reported lower or statistically similar readmission rates for Medicare Advantage beneficiaries,” Orestis A. Panagiotou, MD, of Brown University, Providence, R.I., and colleagues wrote in a research report published in Annals of Internal Medicine.
In this retrospective cohort study, the researchers linked data from 2011 to 2014 from the Medicare Provider Analysis and Review (MedPAR) file to the Healthcare Effectiveness Data and Information Set (HEDIS).
The novel linkage found that HEDIS data underreported hospital admissions for acute MI, heart failure, and pneumonia, the researchers stated. “Plans incorrectly excluded hospitalizations that should have qualified for the readmission measure, and readmission rates were substantially higher among incorrectly excluded hospitalizations.”
Despite this, in analyses using the linkage of HEDIS and MedPAR, “Medicare Advantage beneficiaries had higher 30-day risk-adjusted readmission rates after [acute MI, heart failure, and pneumonia] than did traditional Medicare beneficiaries,” the investigators noted.
Patients in Medicare Advantage had lower unadjusted readmission rates compared with those in traditional Medicare (16.6% vs. 17.1% for acute MI; 21.4% vs. 21.7% for heart failure; and 16.3% vs. 16.4% for pneumonia). After standardization, Medicare Advantage patients had higher readmission rates, compared with those in traditional Medicare (17.2% vs. 16.9% for acute MI; 21.7% vs. 21.4% for heart failure; and 16.5% vs. 16.0% for pneumonia).
The study authors added that, while unadjusted readmission rates were higher for traditional Medicare beneficiaries, “the direction of the difference reversed after standardization. This occurred because Medicare Advantage beneficiaries have, on average, a lower expected readmission risk [that is, they are ‘healthier’].” Prior studies have documented that Medicare Advantage plans enroll beneficiaries with fewer comorbid conditions and that high-cost beneficiaries switch out of Medicare Advantage and into traditional Medicare.
The researchers suggested four reasons for the differences between the results in this study versus others that compared patients using Medicare with those using Medicare Advantage. These were that the new study included a more comprehensive data set, analyses with comorbid conditions “from a well-validated model applied by CMS [Centers for Medicare & Medicaid Services],” national data focused on three conditions included in the Hospital Readmissions Reduction Program, and patients discharged to places other than skilled nursing facilities and inpatient rehabilitation facilities.
Authors of an accompanying editorial called for caution to be used in interpreting Medicare Advantage enrollment as causing an increased readmission risk.
“[The] results are sensitive to adjustment for case mix,” wrote Peter Huckfeldt, PhD, of the University of Minnesota, Minneapolis, and Neeraj Sood, PhD, of the University of Southern California, Los Angeles, in the editorial published in Annals of Internal Medicine (2019 June 25. doi:10.7326/M19-1599) “Using diagnosis codes on hospital claims for case-mix adjustments may be increasingly perilous. ... To our knowledge, there is no recent evidence comparing the intensity of diagnostic coding between clinically similar [traditional Medicare] and [Medicare Advantage] hospital admissions, but if [traditional Medicare] enrollees were coded more intensively than [Medicare Advantage] enrollees, this could lead to [traditional Medicare] enrollees having lower risk-adjusted readmission rares due to coding practices.”
The editorialists added that using a cross-sectional comparison of Medicare Advantage and traditional Medicare patients is concerning because a “key challenge in estimating the effect of [Medicare Advantage] is that enrollment is voluntary,” which can lead to a number of analytical concerns.
The researchers concluded that their findings “are concerning because CMS uses HEDIS performance to construct composite quality ratings and assign payment bonuses to Medicare Advantage plans.
“Our study suggests a need for improved monitoring of the accuracy of HEDIS data,” they noted.
The National Institute on Aging provided the primary funding for this study. A number of the authors received grants from the National Institutes of Health during the conduct of the study. No other relevant disclosures were reported.
SOURCE: Panagiotou OA et al. Ann Intern Med. 2019 Jun 25. doi: 10.7326/M18-1795.
Although earlier research may suggest otherwise, traditional new research suggests.
Researchers used what they described as “a novel data linkage” comparing 30-day readmission rates after hospitalization for three major conditions in the Hospital Readmissions Reduction Program for patients using traditional Medicare versus Medicare Advantage. Those conditions included acute MI, heart failure, and pneumonia.
“Our results contrast with those of previous studies that have reported lower or statistically similar readmission rates for Medicare Advantage beneficiaries,” Orestis A. Panagiotou, MD, of Brown University, Providence, R.I., and colleagues wrote in a research report published in Annals of Internal Medicine.
In this retrospective cohort study, the researchers linked data from 2011 to 2014 from the Medicare Provider Analysis and Review (MedPAR) file to the Healthcare Effectiveness Data and Information Set (HEDIS).
The novel linkage found that HEDIS data underreported hospital admissions for acute MI, heart failure, and pneumonia, the researchers stated. “Plans incorrectly excluded hospitalizations that should have qualified for the readmission measure, and readmission rates were substantially higher among incorrectly excluded hospitalizations.”
Despite this, in analyses using the linkage of HEDIS and MedPAR, “Medicare Advantage beneficiaries had higher 30-day risk-adjusted readmission rates after [acute MI, heart failure, and pneumonia] than did traditional Medicare beneficiaries,” the investigators noted.
Patients in Medicare Advantage had lower unadjusted readmission rates compared with those in traditional Medicare (16.6% vs. 17.1% for acute MI; 21.4% vs. 21.7% for heart failure; and 16.3% vs. 16.4% for pneumonia). After standardization, Medicare Advantage patients had higher readmission rates, compared with those in traditional Medicare (17.2% vs. 16.9% for acute MI; 21.7% vs. 21.4% for heart failure; and 16.5% vs. 16.0% for pneumonia).
The study authors added that, while unadjusted readmission rates were higher for traditional Medicare beneficiaries, “the direction of the difference reversed after standardization. This occurred because Medicare Advantage beneficiaries have, on average, a lower expected readmission risk [that is, they are ‘healthier’].” Prior studies have documented that Medicare Advantage plans enroll beneficiaries with fewer comorbid conditions and that high-cost beneficiaries switch out of Medicare Advantage and into traditional Medicare.
The researchers suggested four reasons for the differences between the results in this study versus others that compared patients using Medicare with those using Medicare Advantage. These were that the new study included a more comprehensive data set, analyses with comorbid conditions “from a well-validated model applied by CMS [Centers for Medicare & Medicaid Services],” national data focused on three conditions included in the Hospital Readmissions Reduction Program, and patients discharged to places other than skilled nursing facilities and inpatient rehabilitation facilities.
Authors of an accompanying editorial called for caution to be used in interpreting Medicare Advantage enrollment as causing an increased readmission risk.
“[The] results are sensitive to adjustment for case mix,” wrote Peter Huckfeldt, PhD, of the University of Minnesota, Minneapolis, and Neeraj Sood, PhD, of the University of Southern California, Los Angeles, in the editorial published in Annals of Internal Medicine (2019 June 25. doi:10.7326/M19-1599) “Using diagnosis codes on hospital claims for case-mix adjustments may be increasingly perilous. ... To our knowledge, there is no recent evidence comparing the intensity of diagnostic coding between clinically similar [traditional Medicare] and [Medicare Advantage] hospital admissions, but if [traditional Medicare] enrollees were coded more intensively than [Medicare Advantage] enrollees, this could lead to [traditional Medicare] enrollees having lower risk-adjusted readmission rares due to coding practices.”
The editorialists added that using a cross-sectional comparison of Medicare Advantage and traditional Medicare patients is concerning because a “key challenge in estimating the effect of [Medicare Advantage] is that enrollment is voluntary,” which can lead to a number of analytical concerns.
The researchers concluded that their findings “are concerning because CMS uses HEDIS performance to construct composite quality ratings and assign payment bonuses to Medicare Advantage plans.
“Our study suggests a need for improved monitoring of the accuracy of HEDIS data,” they noted.
The National Institute on Aging provided the primary funding for this study. A number of the authors received grants from the National Institutes of Health during the conduct of the study. No other relevant disclosures were reported.
SOURCE: Panagiotou OA et al. Ann Intern Med. 2019 Jun 25. doi: 10.7326/M18-1795.
FROM ANNALS OF INTERNAL MEDICINE
Penicillin-susceptible Streptococcus pneumoniae most common cause of bacteremic CAP
A study found that only 2% of children hospitalized with community-acquired pneumonia (CAP) actually had any causative pathogen in their blood culture results, despite national guidelines that recommend blood cultures for all children hospitalized with moderate to severe CAP.
The guidelines are the 2011 guidelines for managing CAP published by the Pediatric Infectious Diseases Society (PIDS) and the Infectious Diseases Society of America (IDSA) (Clin Infect Dis. 2011 Oct;53[7]:617-30).
Cristin O. Fritz, MD, of the Children’s Hospital of Colorado, Aurora, and associates conducted a data analysis of the EPIC (Etiology of Pneumonia in the Community) study to estimate prevalence, risk factors, and clinical outcomes in children hospitalized with bacteremic CAP and to evaluate the relationship between positive blood culture results, empirical antibiotics, and changes in antibiotic treatment regimens.
Data were collected at two Tennessee hospitals and one Utah hospital during Jan. 1, 2010–June 30, 2012. Of the 2,358 children with CAP enrolled in the study, 2,143 (91%) with blood cultures were included in Dr. Fritz’s analysis. Of the 53 patients presenting with positive blood culture results, 46 (2%; 95% confidence interval: 1.6%-2.9%) were identified as having bacteremia. Half of all cases observed were caused by Streptococcus pneumoniae, with Staphylococcus aureus and Streptococcus pyogenes noted less frequently, according to the study published in Pediatrics.
A previous meta-analysis of smaller studies also found that children with CAP rarely had positive blood culture results, a pooled prevalence of 5% (Pediatr Infect Dis J. 2013;32[7]:736-40). Although it is believed that positive blood culture results are key to narrowing the choice of antibiotic and predicting treatment outcomes, the literature – to date – reveals a paucity of data supporting this assumption.
Overall, children in the study presenting with bacteremia experienced more severe clinical outcomes, including longer length of stay, greater likelihood of ICU admission, and invasive mechanical ventilation and/or shock. The authors also observed that bacteremia was less likely to be detected in children given antibiotics after admission but before cultures were obtained (0.8% vs 3%; P = .021). Pleural effusion detected with chest radiograph also consistently indicated bacteremic pneumonia, an observation made within this and other similar studies.
Also of note in detection is the biomarker procalcitonin, which is typically present with bacterial disease. Dr. Fritz and colleagues stressed that because the procalcitonin rate was higher in patients presenting with bacteremia, “this information could influence decisions around culturing if results are rapidly available.” Risk-stratification tools also might serve a valuable purpose in ferreting out those patients presenting with moderate to severe pneumonia most at increased risk for bacterial CAP.
Compared with other studies reporting prevalence ranges of 1%-7%, the prevalence of bacteremia in this study is lower at 2%. The authors attributed the difference to a possible potential limitation with the other studies, for which culture data was only available for a median 47% of enrollees. Dr. Fritz and her colleagues caution that “because cultures were obtained at the discretion of the treating clinician in a majority of studies, blood cultures were likely obtained more often in those with more severe illness or who had not already received antibiotics.” In this scenario, the likelihood that prevalence of bacteremia was overestimated is noteworthy.
The authors observed that penicillin-susceptible S. pneumonia was the most common cause of bacteremic CAP. They further acknowledged that their study and findings by Neuman et al. in 2017 give credence to the joint 2011 PIDS/IDSA guideline recommending narrow-spectrum aminopenicillins specifically to treat children hospitalized due to suspected bacterial CAP.
Despite its small sample size, the results of this study clearly demonstrate that children with bacteremia because of S. pyogenes or S. aureus experience increased morbidity, compared with children with S. pneumoniae, they said
While this is acknowledged to be one of the largest studies of its kind to date, a key limitation was the small number of observable patients with bacteremia, which prevented the researchers from conducting a more in-depth analysis of risk factors and pathogen-specific differences. That one-fourth of patients received in-patient antibiotics before cultures could be collected also likely led to an underestimation of risk factors and misclassification bias. Lastly, the use of blood culture instead of whole-blood polymerase chain reaction, which is known to be more sensitive, also may have led to underestimation of overall bacteremia prevalence.
“In an era with widespread pneumococcal vaccination and low prevalence of bacteremia in the United States, noted Dr. Fritz and associates.
Dr. Fritz had no conflicts of interest to report. Some coauthors cited multiple sources of potential conflict of interest related to consulting fees, grant support, and research support from various pharmaceutical companies and agencies. The study was funded by the National Institutes of Health and in part by a grant from the National Institute of Allergy and Infectious Diseases.
SOURCE: Fritz C et al. Pediatrics. 2019;144(1):e20183090.
A study found that only 2% of children hospitalized with community-acquired pneumonia (CAP) actually had any causative pathogen in their blood culture results, despite national guidelines that recommend blood cultures for all children hospitalized with moderate to severe CAP.

The guidelines are the 2011 guidelines for managing CAP published by the Pediatric Infectious Diseases Society (PIDS) and the Infectious Diseases Society of America (IDSA) (Clin Infect Dis. 2011 Oct;53[7]:617-30).
Cristin O. Fritz, MD, of the Children’s Hospital of Colorado, Aurora, and associates conducted a data analysis of the EPIC (Etiology of Pneumonia in the Community) study to estimate prevalence, risk factors, and clinical outcomes in children hospitalized with bacteremic CAP and to evaluate the relationship between positive blood culture results, empirical antibiotics, and changes in antibiotic treatment regimens.
Data were collected at two Tennessee hospitals and one Utah hospital during Jan. 1, 2010–June 30, 2012. Of the 2,358 children with CAP enrolled in the study, 2,143 (91%) with blood cultures were included in Dr. Fritz’s analysis. Of the 53 patients presenting with positive blood culture results, 46 (2%; 95% confidence interval: 1.6%-2.9%) were identified as having bacteremia. Half of all cases observed were caused by Streptococcus pneumoniae, with Staphylococcus aureus and Streptococcus pyogenes noted less frequently, according to the study published in Pediatrics.
A previous meta-analysis of smaller studies also found that children with CAP rarely had positive blood culture results, a pooled prevalence of 5% (Pediatr Infect Dis J. 2013;32[7]:736-40). Although it is believed that positive blood culture results are key to narrowing the choice of antibiotic and predicting treatment outcomes, the literature – to date – reveals a paucity of data supporting this assumption.
Overall, children in the study presenting with bacteremia experienced more severe clinical outcomes, including longer length of stay, greater likelihood of ICU admission, and invasive mechanical ventilation and/or shock. The authors also observed that bacteremia was less likely to be detected in children given antibiotics after admission but before cultures were obtained (0.8% vs 3%; P = .021). Pleural effusion detected with chest radiograph also consistently indicated bacteremic pneumonia, an observation made within this and other similar studies.
Also of note in detection is the biomarker procalcitonin, which is typically present with bacterial disease. Dr. Fritz and colleagues stressed that because the procalcitonin rate was higher in patients presenting with bacteremia, “this information could influence decisions around culturing if results are rapidly available.” Risk-stratification tools also might serve a valuable purpose in ferreting out those patients presenting with moderate to severe pneumonia most at increased risk for bacterial CAP.
Compared with other studies reporting prevalence ranges of 1%-7%, the prevalence of bacteremia in this study is lower at 2%. The authors attributed the difference to a possible potential limitation with the other studies, for which culture data was only available for a median 47% of enrollees. Dr. Fritz and her colleagues caution that “because cultures were obtained at the discretion of the treating clinician in a majority of studies, blood cultures were likely obtained more often in those with more severe illness or who had not already received antibiotics.” In this scenario, the likelihood that prevalence of bacteremia was overestimated is noteworthy.
The authors observed that penicillin-susceptible S. pneumonia was the most common cause of bacteremic CAP. They further acknowledged that their study and findings by Neuman et al. in 2017 give credence to the joint 2011 PIDS/IDSA guideline recommending narrow-spectrum aminopenicillins specifically to treat children hospitalized due to suspected bacterial CAP.
Despite its small sample size, the results of this study clearly demonstrate that children with bacteremia because of S. pyogenes or S. aureus experience increased morbidity, compared with children with S. pneumoniae, they said
While this is acknowledged to be one of the largest studies of its kind to date, a key limitation was the small number of observable patients with bacteremia, which prevented the researchers from conducting a more in-depth analysis of risk factors and pathogen-specific differences. That one-fourth of patients received in-patient antibiotics before cultures could be collected also likely led to an underestimation of risk factors and misclassification bias. Lastly, the use of blood culture instead of whole-blood polymerase chain reaction, which is known to be more sensitive, also may have led to underestimation of overall bacteremia prevalence.
“In an era with widespread pneumococcal vaccination and low prevalence of bacteremia in the United States, noted Dr. Fritz and associates.
Dr. Fritz had no conflicts of interest to report. Some coauthors cited multiple sources of potential conflict of interest related to consulting fees, grant support, and research support from various pharmaceutical companies and agencies. The study was funded by the National Institutes of Health and in part by a grant from the National Institute of Allergy and Infectious Diseases.
SOURCE: Fritz C et al. Pediatrics. 2019;144(1):e20183090.
A study found that only 2% of children hospitalized with community-acquired pneumonia (CAP) actually had any causative pathogen in their blood culture results, despite national guidelines that recommend blood cultures for all children hospitalized with moderate to severe CAP.

The guidelines are the 2011 guidelines for managing CAP published by the Pediatric Infectious Diseases Society (PIDS) and the Infectious Diseases Society of America (IDSA) (Clin Infect Dis. 2011 Oct;53[7]:617-30).
Cristin O. Fritz, MD, of the Children’s Hospital of Colorado, Aurora, and associates conducted a data analysis of the EPIC (Etiology of Pneumonia in the Community) study to estimate prevalence, risk factors, and clinical outcomes in children hospitalized with bacteremic CAP and to evaluate the relationship between positive blood culture results, empirical antibiotics, and changes in antibiotic treatment regimens.
Data were collected at two Tennessee hospitals and one Utah hospital during Jan. 1, 2010–June 30, 2012. Of the 2,358 children with CAP enrolled in the study, 2,143 (91%) with blood cultures were included in Dr. Fritz’s analysis. Of the 53 patients presenting with positive blood culture results, 46 (2%; 95% confidence interval: 1.6%-2.9%) were identified as having bacteremia. Half of all cases observed were caused by Streptococcus pneumoniae, with Staphylococcus aureus and Streptococcus pyogenes noted less frequently, according to the study published in Pediatrics.
A previous meta-analysis of smaller studies also found that children with CAP rarely had positive blood culture results, a pooled prevalence of 5% (Pediatr Infect Dis J. 2013;32[7]:736-40). Although it is believed that positive blood culture results are key to narrowing the choice of antibiotic and predicting treatment outcomes, the literature – to date – reveals a paucity of data supporting this assumption.
Overall, children in the study presenting with bacteremia experienced more severe clinical outcomes, including longer length of stay, greater likelihood of ICU admission, and invasive mechanical ventilation and/or shock. The authors also observed that bacteremia was less likely to be detected in children given antibiotics after admission but before cultures were obtained (0.8% vs 3%; P = .021). Pleural effusion detected with chest radiograph also consistently indicated bacteremic pneumonia, an observation made within this and other similar studies.
Also of note in detection is the biomarker procalcitonin, which is typically present with bacterial disease. Dr. Fritz and colleagues stressed that because the procalcitonin rate was higher in patients presenting with bacteremia, “this information could influence decisions around culturing if results are rapidly available.” Risk-stratification tools also might serve a valuable purpose in ferreting out those patients presenting with moderate to severe pneumonia most at increased risk for bacterial CAP.
Compared with other studies reporting prevalence ranges of 1%-7%, the prevalence of bacteremia in this study is lower at 2%. The authors attributed the difference to a possible potential limitation with the other studies, for which culture data was only available for a median 47% of enrollees. Dr. Fritz and her colleagues caution that “because cultures were obtained at the discretion of the treating clinician in a majority of studies, blood cultures were likely obtained more often in those with more severe illness or who had not already received antibiotics.” In this scenario, the likelihood that prevalence of bacteremia was overestimated is noteworthy.
The authors observed that penicillin-susceptible S. pneumonia was the most common cause of bacteremic CAP. They further acknowledged that their study and findings by Neuman et al. in 2017 give credence to the joint 2011 PIDS/IDSA guideline recommending narrow-spectrum aminopenicillins specifically to treat children hospitalized due to suspected bacterial CAP.
Despite its small sample size, the results of this study clearly demonstrate that children with bacteremia because of S. pyogenes or S. aureus experience increased morbidity, compared with children with S. pneumoniae, they said
While this is acknowledged to be one of the largest studies of its kind to date, a key limitation was the small number of observable patients with bacteremia, which prevented the researchers from conducting a more in-depth analysis of risk factors and pathogen-specific differences. That one-fourth of patients received in-patient antibiotics before cultures could be collected also likely led to an underestimation of risk factors and misclassification bias. Lastly, the use of blood culture instead of whole-blood polymerase chain reaction, which is known to be more sensitive, also may have led to underestimation of overall bacteremia prevalence.
“In an era with widespread pneumococcal vaccination and low prevalence of bacteremia in the United States, noted Dr. Fritz and associates.
Dr. Fritz had no conflicts of interest to report. Some coauthors cited multiple sources of potential conflict of interest related to consulting fees, grant support, and research support from various pharmaceutical companies and agencies. The study was funded by the National Institutes of Health and in part by a grant from the National Institute of Allergy and Infectious Diseases.
SOURCE: Fritz C et al. Pediatrics. 2019;144(1):e20183090.
FROM PEDIATRICS
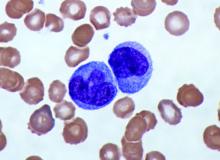
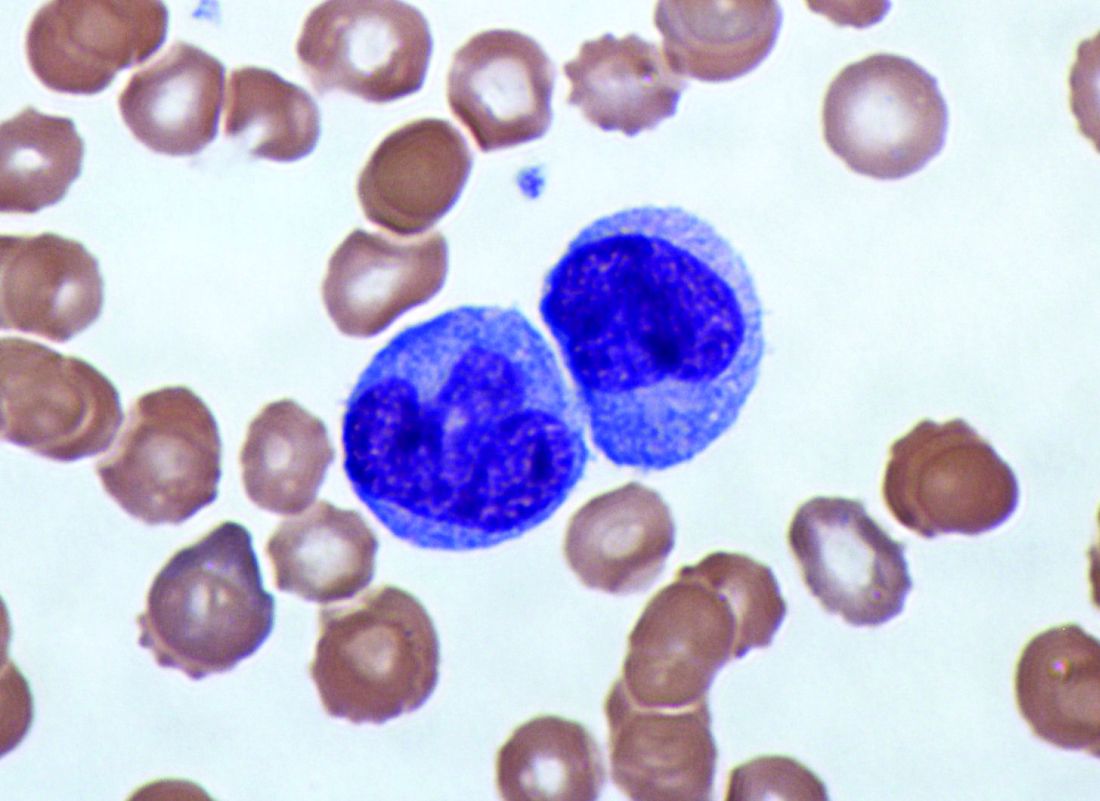
Elevated monocyte count predicts poor outcomes in idiopathic pulmonary fibrosis
, including hypertrophic cardiomyopathy, systemic sclerosis, and myelofibrosis, according to research published in The Lancet Respiratory Medicine.
The data indicate that “a single threshold value of absolute monocyte counts of 0.95 K/mcL could be used to identify high-risk patients with a fibrotic disease,” said Madeleine K. D. Scott, a researcher at Stanford (Calif.) University, and coauthors. The results “suggest that monocyte count should be incorporated into the clinical assessment” and may “enable more conscientious allocation of scarce resources, including lung transplantations,” they said.
While other published biomarkers – including gene panels and multicytokine signatures – may be expensive and not readily available, “absolute monocyte count is routinely measured as part of a complete blood count, an inexpensive test used in clinical practice worldwide,” the authors said.
Further study of monocytes’ mechanistic role in fibrosis ultimately could point to new treatment approaches.
A retrospective multicenter cohort study
To assess whether immune cells may identify patients with idiopathic pulmonary fibrosis at greater risk of poor outcomes, Ms. Scott and her collaborators conducted a retrospective multicenter cohort study.
They first analyzed transcriptome data from 120 peripheral blood mononuclear cell samples of patients with idiopathic pulmonary fibrosis, which they obtained from the Gene Expression Omnibus at the National Center for Biotechnology Information. They used statistical deconvolution to estimate percentages of 13 immune cell types and examined their associations with transplant-free survival. Their discovery analysis found that estimated CD14+ classical monocyte percentages above the mean correlated with shorter transplant-free survival times (hazard ratio, 1.82), but percentages of T cells and B cells did not.
The researchers then validated these results using samples from patients with idiopathic pulmonary fibrosis in two independent cohorts. In the COMET validation cohort, which included 45 patients with idiopathic pulmonary fibrosis whose monocyte counts were measured using flow cytometry, higher monocyte counts were significantly associated with greater risk of disease progression. In the Yale cohort, which included 15 patients with idiopathic pulmonary fibrosis, the 6 patients who were classified as high risk on the basis of a 52-gene signature had more CD14+ monocytes than the 9 low-risk patients did.
In addition, Ms. Scott and her collaborators looked at complete blood count values in the electronic health records of 45,068 patients with idiopathic pulmonary fibrosis, systemic sclerosis, hypertrophic cardiomyopathy, or myelofibrosis in Stanford, Northwestern, Vanderbilt, and Optum Clinformatics Data Mart cohorts.
Among patients in the COMET, Stanford, and Northwestern datasets, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (HR, 2.47) and the gender, age, and physiology index (HR, 2.06). Data from 7,459 patients with idiopathic pulmonary fibrosis “showed that patients with monocyte counts of 0.95 K/mcL or greater were at increased risk of mortality with lung transplantation as a censoring event, after adjusting for age at diagnosis and sex” in the Stanford (HR, 2.30), Vanderbilt (HR, 1.52), and Optum (HR, 1.74) cohorts. “Likewise, higher absolute monocyte count was associated with shortened survival in patients with hypertrophic cardiomyopathy across all three cohorts, and in patients with systemic sclerosis or myelofibrosis in two of the three cohorts,” the researchers said.
The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
SOURCE: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
The study by Scott et al. provides evidence that monocyte count may be a “novel, simple, and inexpensive prognostic biomarker in idiopathic pulmonary fibrosis,” according to an accompanying editorial.
Progress has been made in the treatment of idiopathic pulmonary fibrosis, but patient prognosis remains “challenging to predict,” wrote Michael Kreuter, MD, of University of Heidelberg, Germany, and Toby M. Maher, MB, MSc, PhD, of Royal Brompton Hospital in London and Imperial College London. “One lesson that can be learned from other respiratory disorders is that routinely measured cellular biomarkers, such as blood eosinophil counts in chronic obstructive pulmonary disease (COPD), can predict treatment responses” (Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600[19]30050-5).
Increased blood monocyte counts in idiopathic pulmonary fibrosis may reflect disease activity, which “could explain the outcome differences,” said Dr. Kreuter and Dr. Maher. “As highlighted by the investigators themselves, before introducing assessment of monocyte counts as part of routine clinical care for individuals with idiopathic pulmonary fibrosis, the limitations of this research should be taken into account. These include uncertainty around diagnosis and disease severity in a substantial subset of the patients, and the unknown effect of medical therapies (including corticosteroids and immunosuppressant and antifibrotic drugs) on monocyte counts and prognosis.” Researchers should validate the clinical value of blood monocyte counts in existing and future cohorts and evaluate the biomarker in clinical trials.
The editorialists have received compensation and funding from various pharmaceutical companies.
The study by Scott et al. provides evidence that monocyte count may be a “novel, simple, and inexpensive prognostic biomarker in idiopathic pulmonary fibrosis,” according to an accompanying editorial.
Progress has been made in the treatment of idiopathic pulmonary fibrosis, but patient prognosis remains “challenging to predict,” wrote Michael Kreuter, MD, of University of Heidelberg, Germany, and Toby M. Maher, MB, MSc, PhD, of Royal Brompton Hospital in London and Imperial College London. “One lesson that can be learned from other respiratory disorders is that routinely measured cellular biomarkers, such as blood eosinophil counts in chronic obstructive pulmonary disease (COPD), can predict treatment responses” (Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600[19]30050-5).
Increased blood monocyte counts in idiopathic pulmonary fibrosis may reflect disease activity, which “could explain the outcome differences,” said Dr. Kreuter and Dr. Maher. “As highlighted by the investigators themselves, before introducing assessment of monocyte counts as part of routine clinical care for individuals with idiopathic pulmonary fibrosis, the limitations of this research should be taken into account. These include uncertainty around diagnosis and disease severity in a substantial subset of the patients, and the unknown effect of medical therapies (including corticosteroids and immunosuppressant and antifibrotic drugs) on monocyte counts and prognosis.” Researchers should validate the clinical value of blood monocyte counts in existing and future cohorts and evaluate the biomarker in clinical trials.
The editorialists have received compensation and funding from various pharmaceutical companies.
The study by Scott et al. provides evidence that monocyte count may be a “novel, simple, and inexpensive prognostic biomarker in idiopathic pulmonary fibrosis,” according to an accompanying editorial.
Progress has been made in the treatment of idiopathic pulmonary fibrosis, but patient prognosis remains “challenging to predict,” wrote Michael Kreuter, MD, of University of Heidelberg, Germany, and Toby M. Maher, MB, MSc, PhD, of Royal Brompton Hospital in London and Imperial College London. “One lesson that can be learned from other respiratory disorders is that routinely measured cellular biomarkers, such as blood eosinophil counts in chronic obstructive pulmonary disease (COPD), can predict treatment responses” (Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600[19]30050-5).
Increased blood monocyte counts in idiopathic pulmonary fibrosis may reflect disease activity, which “could explain the outcome differences,” said Dr. Kreuter and Dr. Maher. “As highlighted by the investigators themselves, before introducing assessment of monocyte counts as part of routine clinical care for individuals with idiopathic pulmonary fibrosis, the limitations of this research should be taken into account. These include uncertainty around diagnosis and disease severity in a substantial subset of the patients, and the unknown effect of medical therapies (including corticosteroids and immunosuppressant and antifibrotic drugs) on monocyte counts and prognosis.” Researchers should validate the clinical value of blood monocyte counts in existing and future cohorts and evaluate the biomarker in clinical trials.
The editorialists have received compensation and funding from various pharmaceutical companies.
, including hypertrophic cardiomyopathy, systemic sclerosis, and myelofibrosis, according to research published in The Lancet Respiratory Medicine.
The data indicate that “a single threshold value of absolute monocyte counts of 0.95 K/mcL could be used to identify high-risk patients with a fibrotic disease,” said Madeleine K. D. Scott, a researcher at Stanford (Calif.) University, and coauthors. The results “suggest that monocyte count should be incorporated into the clinical assessment” and may “enable more conscientious allocation of scarce resources, including lung transplantations,” they said.
While other published biomarkers – including gene panels and multicytokine signatures – may be expensive and not readily available, “absolute monocyte count is routinely measured as part of a complete blood count, an inexpensive test used in clinical practice worldwide,” the authors said.
Further study of monocytes’ mechanistic role in fibrosis ultimately could point to new treatment approaches.
A retrospective multicenter cohort study
To assess whether immune cells may identify patients with idiopathic pulmonary fibrosis at greater risk of poor outcomes, Ms. Scott and her collaborators conducted a retrospective multicenter cohort study.
They first analyzed transcriptome data from 120 peripheral blood mononuclear cell samples of patients with idiopathic pulmonary fibrosis, which they obtained from the Gene Expression Omnibus at the National Center for Biotechnology Information. They used statistical deconvolution to estimate percentages of 13 immune cell types and examined their associations with transplant-free survival. Their discovery analysis found that estimated CD14+ classical monocyte percentages above the mean correlated with shorter transplant-free survival times (hazard ratio, 1.82), but percentages of T cells and B cells did not.
The researchers then validated these results using samples from patients with idiopathic pulmonary fibrosis in two independent cohorts. In the COMET validation cohort, which included 45 patients with idiopathic pulmonary fibrosis whose monocyte counts were measured using flow cytometry, higher monocyte counts were significantly associated with greater risk of disease progression. In the Yale cohort, which included 15 patients with idiopathic pulmonary fibrosis, the 6 patients who were classified as high risk on the basis of a 52-gene signature had more CD14+ monocytes than the 9 low-risk patients did.
In addition, Ms. Scott and her collaborators looked at complete blood count values in the electronic health records of 45,068 patients with idiopathic pulmonary fibrosis, systemic sclerosis, hypertrophic cardiomyopathy, or myelofibrosis in Stanford, Northwestern, Vanderbilt, and Optum Clinformatics Data Mart cohorts.
Among patients in the COMET, Stanford, and Northwestern datasets, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (HR, 2.47) and the gender, age, and physiology index (HR, 2.06). Data from 7,459 patients with idiopathic pulmonary fibrosis “showed that patients with monocyte counts of 0.95 K/mcL or greater were at increased risk of mortality with lung transplantation as a censoring event, after adjusting for age at diagnosis and sex” in the Stanford (HR, 2.30), Vanderbilt (HR, 1.52), and Optum (HR, 1.74) cohorts. “Likewise, higher absolute monocyte count was associated with shortened survival in patients with hypertrophic cardiomyopathy across all three cohorts, and in patients with systemic sclerosis or myelofibrosis in two of the three cohorts,” the researchers said.
The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
SOURCE: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
, including hypertrophic cardiomyopathy, systemic sclerosis, and myelofibrosis, according to research published in The Lancet Respiratory Medicine.
The data indicate that “a single threshold value of absolute monocyte counts of 0.95 K/mcL could be used to identify high-risk patients with a fibrotic disease,” said Madeleine K. D. Scott, a researcher at Stanford (Calif.) University, and coauthors. The results “suggest that monocyte count should be incorporated into the clinical assessment” and may “enable more conscientious allocation of scarce resources, including lung transplantations,” they said.
While other published biomarkers – including gene panels and multicytokine signatures – may be expensive and not readily available, “absolute monocyte count is routinely measured as part of a complete blood count, an inexpensive test used in clinical practice worldwide,” the authors said.
Further study of monocytes’ mechanistic role in fibrosis ultimately could point to new treatment approaches.
A retrospective multicenter cohort study
To assess whether immune cells may identify patients with idiopathic pulmonary fibrosis at greater risk of poor outcomes, Ms. Scott and her collaborators conducted a retrospective multicenter cohort study.
They first analyzed transcriptome data from 120 peripheral blood mononuclear cell samples of patients with idiopathic pulmonary fibrosis, which they obtained from the Gene Expression Omnibus at the National Center for Biotechnology Information. They used statistical deconvolution to estimate percentages of 13 immune cell types and examined their associations with transplant-free survival. Their discovery analysis found that estimated CD14+ classical monocyte percentages above the mean correlated with shorter transplant-free survival times (hazard ratio, 1.82), but percentages of T cells and B cells did not.
The researchers then validated these results using samples from patients with idiopathic pulmonary fibrosis in two independent cohorts. In the COMET validation cohort, which included 45 patients with idiopathic pulmonary fibrosis whose monocyte counts were measured using flow cytometry, higher monocyte counts were significantly associated with greater risk of disease progression. In the Yale cohort, which included 15 patients with idiopathic pulmonary fibrosis, the 6 patients who were classified as high risk on the basis of a 52-gene signature had more CD14+ monocytes than the 9 low-risk patients did.
In addition, Ms. Scott and her collaborators looked at complete blood count values in the electronic health records of 45,068 patients with idiopathic pulmonary fibrosis, systemic sclerosis, hypertrophic cardiomyopathy, or myelofibrosis in Stanford, Northwestern, Vanderbilt, and Optum Clinformatics Data Mart cohorts.
Among patients in the COMET, Stanford, and Northwestern datasets, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (HR, 2.47) and the gender, age, and physiology index (HR, 2.06). Data from 7,459 patients with idiopathic pulmonary fibrosis “showed that patients with monocyte counts of 0.95 K/mcL or greater were at increased risk of mortality with lung transplantation as a censoring event, after adjusting for age at diagnosis and sex” in the Stanford (HR, 2.30), Vanderbilt (HR, 1.52), and Optum (HR, 1.74) cohorts. “Likewise, higher absolute monocyte count was associated with shortened survival in patients with hypertrophic cardiomyopathy across all three cohorts, and in patients with systemic sclerosis or myelofibrosis in two of the three cohorts,” the researchers said.
The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
SOURCE: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
FROM THE LANCET RESPIRATORY MEDICINE
Key clinical point: An increased monocyte count predicts poor outcomes among patients with idiopathic pulmonary fibrosis and other fibrotic diseases.
Major finding: Among patients in three cohorts, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (hazard ratio, 2.47) and the gender, age, and physiology index (HR, 2.06).
Study details: A retrospective analysis of data from 7,000 patients with idiopathic pulmonary fibrosis from five independent cohorts.
Disclosures: The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
Source: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
Cannabis vaping among teens tied to tobacco use
and that practice is associated with cigars, waterpipe and e-cigarette use, findings from a survey of nearly 3,000 adolescents have shown.
“Although the prevalence of e-cigarette use among youth has increased dramatically in the past decade, little epidemiologic data exist on the prevalence of using e-cigarette devices or other specialised devices to vaporise (‘vape’) cannabis in the form of hash oil, tetrahydrocannabinol (THC) wax or oil, or dried cannabis buds or leaves,” wrote Sarah D. Kowitt, PhD, of the University of North Carolina, Chapel Hill, and colleagues. “This is surprising given that (1) cannabis (also referred to as marijuana) and e-cigarettes are the most commonly used substances by adolescents in the USA, (2) evidence exists that adolescents dual use both tobacco e-cigarettes and cannabis, and (3) longitudinal research suggests that use of e-cigarettes is associated with progression to use of cannabis.”
In a study published in BMJ Open, the researchers used data from the 2017 North Carolina Youth Tobacco Survey, a school-based survey of students in grades 6-12. The study population included 2,835 adolescents in grades 9-12.
Overall, 9.6% of students reported ever vaping cannabis. In multivariate analysis, cannabis vaping was significantly more likely among adolescents who reported using e-cigarettes (adjusted odds ratio 3.18), cigars (aOR 3.76), or water pipes (aOR 2.32) in the past 30 days, compared with peers who didn’t use tobacco.
The researchers found no significant association between smokeless tobacco use or traditional cigarette use in the past 30 days and vaping cannabis.
In a bivariate analysis, vaping cannabis was significantly more common among males vs. females (11% vs. 8.2%) and among non-Hispanic white students (11.3%), Hispanic students (10.5%), and other non-Hispanic students (11.8%) compared with non-Hispanic black students (5.0%).
In addition, prevalence of cannabis vaping increased with grade level, from 4.7% of 9th graders to 15.5% of 12th graders.
The health impacts of vaping cannabis are not well researched, but the researchers note that among the potential safety issues are earlier initiation of tobacco or cannabis use, concomitant tobacco and cannabis use, increased frequency of use or misuse of tobacco or cannabis, or increased potency of cannabis.
The results of the study were limited by several factors including the use of data only from the state of North Carolina, the lack of data on frequency or current vaping cannabis behavior, lack of data on specific products, and lack of data on whether teens used specialized devices or e-cigarettes for cannabis vaping. However, the findings are consistent with studies on prevalence of cannabis vaping in other states such as Connecticut and California. “No studies to our knowledge have examined how adolescents who vape cannabis use other specific tobacco products (i.e., cigarettes, cigars, waterpipe, smokeless tobacco),” the researchers wrote.
The findings confirm that a large number of adolescents who use tobacco products have vaped cannabis as well, and this growing public health issue “is likely to affect and be affected by tobacco control and cannabis policies in states and at the federal level in the USA,” the researchers concluded.
“Increased research investigating how youth use e-cigarette devices for other purposes beyond vaping nicotine, like the current study, is needed,” they added.
The study was supported in part by the National Cancer Institute and the Food and Drug Administration’s Center for Tobacco Products. The researchers had no financial conflicts to disclose.
SOURCE: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
and that practice is associated with cigars, waterpipe and e-cigarette use, findings from a survey of nearly 3,000 adolescents have shown.
“Although the prevalence of e-cigarette use among youth has increased dramatically in the past decade, little epidemiologic data exist on the prevalence of using e-cigarette devices or other specialised devices to vaporise (‘vape’) cannabis in the form of hash oil, tetrahydrocannabinol (THC) wax or oil, or dried cannabis buds or leaves,” wrote Sarah D. Kowitt, PhD, of the University of North Carolina, Chapel Hill, and colleagues. “This is surprising given that (1) cannabis (also referred to as marijuana) and e-cigarettes are the most commonly used substances by adolescents in the USA, (2) evidence exists that adolescents dual use both tobacco e-cigarettes and cannabis, and (3) longitudinal research suggests that use of e-cigarettes is associated with progression to use of cannabis.”
In a study published in BMJ Open, the researchers used data from the 2017 North Carolina Youth Tobacco Survey, a school-based survey of students in grades 6-12. The study population included 2,835 adolescents in grades 9-12.
Overall, 9.6% of students reported ever vaping cannabis. In multivariate analysis, cannabis vaping was significantly more likely among adolescents who reported using e-cigarettes (adjusted odds ratio 3.18), cigars (aOR 3.76), or water pipes (aOR 2.32) in the past 30 days, compared with peers who didn’t use tobacco.
The researchers found no significant association between smokeless tobacco use or traditional cigarette use in the past 30 days and vaping cannabis.
In a bivariate analysis, vaping cannabis was significantly more common among males vs. females (11% vs. 8.2%) and among non-Hispanic white students (11.3%), Hispanic students (10.5%), and other non-Hispanic students (11.8%) compared with non-Hispanic black students (5.0%).
In addition, prevalence of cannabis vaping increased with grade level, from 4.7% of 9th graders to 15.5% of 12th graders.
The health impacts of vaping cannabis are not well researched, but the researchers note that among the potential safety issues are earlier initiation of tobacco or cannabis use, concomitant tobacco and cannabis use, increased frequency of use or misuse of tobacco or cannabis, or increased potency of cannabis.
The results of the study were limited by several factors including the use of data only from the state of North Carolina, the lack of data on frequency or current vaping cannabis behavior, lack of data on specific products, and lack of data on whether teens used specialized devices or e-cigarettes for cannabis vaping. However, the findings are consistent with studies on prevalence of cannabis vaping in other states such as Connecticut and California. “No studies to our knowledge have examined how adolescents who vape cannabis use other specific tobacco products (i.e., cigarettes, cigars, waterpipe, smokeless tobacco),” the researchers wrote.
The findings confirm that a large number of adolescents who use tobacco products have vaped cannabis as well, and this growing public health issue “is likely to affect and be affected by tobacco control and cannabis policies in states and at the federal level in the USA,” the researchers concluded.
“Increased research investigating how youth use e-cigarette devices for other purposes beyond vaping nicotine, like the current study, is needed,” they added.
The study was supported in part by the National Cancer Institute and the Food and Drug Administration’s Center for Tobacco Products. The researchers had no financial conflicts to disclose.
SOURCE: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
and that practice is associated with cigars, waterpipe and e-cigarette use, findings from a survey of nearly 3,000 adolescents have shown.
“Although the prevalence of e-cigarette use among youth has increased dramatically in the past decade, little epidemiologic data exist on the prevalence of using e-cigarette devices or other specialised devices to vaporise (‘vape’) cannabis in the form of hash oil, tetrahydrocannabinol (THC) wax or oil, or dried cannabis buds or leaves,” wrote Sarah D. Kowitt, PhD, of the University of North Carolina, Chapel Hill, and colleagues. “This is surprising given that (1) cannabis (also referred to as marijuana) and e-cigarettes are the most commonly used substances by adolescents in the USA, (2) evidence exists that adolescents dual use both tobacco e-cigarettes and cannabis, and (3) longitudinal research suggests that use of e-cigarettes is associated with progression to use of cannabis.”
In a study published in BMJ Open, the researchers used data from the 2017 North Carolina Youth Tobacco Survey, a school-based survey of students in grades 6-12. The study population included 2,835 adolescents in grades 9-12.
Overall, 9.6% of students reported ever vaping cannabis. In multivariate analysis, cannabis vaping was significantly more likely among adolescents who reported using e-cigarettes (adjusted odds ratio 3.18), cigars (aOR 3.76), or water pipes (aOR 2.32) in the past 30 days, compared with peers who didn’t use tobacco.
The researchers found no significant association between smokeless tobacco use or traditional cigarette use in the past 30 days and vaping cannabis.
In a bivariate analysis, vaping cannabis was significantly more common among males vs. females (11% vs. 8.2%) and among non-Hispanic white students (11.3%), Hispanic students (10.5%), and other non-Hispanic students (11.8%) compared with non-Hispanic black students (5.0%).
In addition, prevalence of cannabis vaping increased with grade level, from 4.7% of 9th graders to 15.5% of 12th graders.
The health impacts of vaping cannabis are not well researched, but the researchers note that among the potential safety issues are earlier initiation of tobacco or cannabis use, concomitant tobacco and cannabis use, increased frequency of use or misuse of tobacco or cannabis, or increased potency of cannabis.
The results of the study were limited by several factors including the use of data only from the state of North Carolina, the lack of data on frequency or current vaping cannabis behavior, lack of data on specific products, and lack of data on whether teens used specialized devices or e-cigarettes for cannabis vaping. However, the findings are consistent with studies on prevalence of cannabis vaping in other states such as Connecticut and California. “No studies to our knowledge have examined how adolescents who vape cannabis use other specific tobacco products (i.e., cigarettes, cigars, waterpipe, smokeless tobacco),” the researchers wrote.
The findings confirm that a large number of adolescents who use tobacco products have vaped cannabis as well, and this growing public health issue “is likely to affect and be affected by tobacco control and cannabis policies in states and at the federal level in the USA,” the researchers concluded.
“Increased research investigating how youth use e-cigarette devices for other purposes beyond vaping nicotine, like the current study, is needed,” they added.
The study was supported in part by the National Cancer Institute and the Food and Drug Administration’s Center for Tobacco Products. The researchers had no financial conflicts to disclose.
SOURCE: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
FROM BMJ OPEN
Key clinical point: Use of tobacco products was significantly associated with cannabis vaping in teens.
Major finding: Approximately 10% of adolescents reported vaping cannabis.
Study details: The data come from a survey of 2,835 adolescents in North Carolina.
Disclosures: The study was supported in part by the National Cancer Institute and the FDA Center for Tobacco Products. The researchers had no financial conflicts to disclose.
Source: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
Inhaler technique not to blame for uncontrolled asthma in inner-city study
, a study has found.
“Incorrect inhaler technique cannot explain the poor disease control in our patient population,” wrote Patrick K. Gleeson, MD, of the University of Pennsylvania, Philadelphia, and coinvestigators. Their report is in the Journal of Allergy and Clinical Immunology: In Practice. “In individuals with poorly controlled asthma, other factors contributing to disease mortality must be considered.”
The 586 patients in the study were observed using their inhalers, and their technique was scored by way of a checklist developed for the study. Inhaler technique – widely regarded as a risk factor for poor disease control – was “better than expected,” the investigators reported, with 56% of patients using metered dose inhalers and 64% of those using dry powder inhalers not making any errors.
“The seeming disassociation between subjects’ asthma control and inhaler technique is counterintuitive, and may be explained by important baseline characteristics in our patients,” they wrote. For instance, participants had suboptimal living conditions in lower income Philadelphia neighborhoods. Almost a quarter – 23% – were current smokers, and almost half were Medicaid recipients. In addition, their mean body mass index was 35.1 kg/m2.
The investigators hypothesized that patients with lower health literacy would have poorer technique but found instead that technique did not vary by reading comprehension or numeracy levels.
More than half of the adults in the study had uncontrolled asthma as defined by prednisone use, an emergency department visit, or a hospitalization for asthma in the past 12 months. A subset had moderate to severe disease per a physician’s diagnosis, forced expiratory volume in 1 second less than 80% predicted, and improvement with a bronchodilator. All patients, however, were considered to have uncontrolled asthma.
There is “uncertainty” in the field about how to measure inhaler technique, and the technique checklist used in the study “may have omitted potentially important errors,” the investigators noted. Still, “good technique predominated among our [population of vulnerable patients].”
The project was supported through awards from the National Institutes of Health/National Heart, Lung, and Blood Institute and the Patient-Centered Outcomes Research Institute.
Coinvestigator Andrea J. Apter, MD, reported that she consults for UpToDate and is an associate editor for the journal. Coinvestigator Knashawn H. Morales, ScD, reported owning stock in Altria Group, British American Tobacco, and Philip Morris International. The other authors reported having no conflicts of interest.
SOURCE: Gleeson PK. J Allergy Clin Immunol Pract. 2019 Jun 5. doi: 10.1016/j.jaip.2019.05.048.
, a study has found.
“Incorrect inhaler technique cannot explain the poor disease control in our patient population,” wrote Patrick K. Gleeson, MD, of the University of Pennsylvania, Philadelphia, and coinvestigators. Their report is in the Journal of Allergy and Clinical Immunology: In Practice. “In individuals with poorly controlled asthma, other factors contributing to disease mortality must be considered.”
The 586 patients in the study were observed using their inhalers, and their technique was scored by way of a checklist developed for the study. Inhaler technique – widely regarded as a risk factor for poor disease control – was “better than expected,” the investigators reported, with 56% of patients using metered dose inhalers and 64% of those using dry powder inhalers not making any errors.
“The seeming disassociation between subjects’ asthma control and inhaler technique is counterintuitive, and may be explained by important baseline characteristics in our patients,” they wrote. For instance, participants had suboptimal living conditions in lower income Philadelphia neighborhoods. Almost a quarter – 23% – were current smokers, and almost half were Medicaid recipients. In addition, their mean body mass index was 35.1 kg/m2.
The investigators hypothesized that patients with lower health literacy would have poorer technique but found instead that technique did not vary by reading comprehension or numeracy levels.
More than half of the adults in the study had uncontrolled asthma as defined by prednisone use, an emergency department visit, or a hospitalization for asthma in the past 12 months. A subset had moderate to severe disease per a physician’s diagnosis, forced expiratory volume in 1 second less than 80% predicted, and improvement with a bronchodilator. All patients, however, were considered to have uncontrolled asthma.
There is “uncertainty” in the field about how to measure inhaler technique, and the technique checklist used in the study “may have omitted potentially important errors,” the investigators noted. Still, “good technique predominated among our [population of vulnerable patients].”
The project was supported through awards from the National Institutes of Health/National Heart, Lung, and Blood Institute and the Patient-Centered Outcomes Research Institute.
Coinvestigator Andrea J. Apter, MD, reported that she consults for UpToDate and is an associate editor for the journal. Coinvestigator Knashawn H. Morales, ScD, reported owning stock in Altria Group, British American Tobacco, and Philip Morris International. The other authors reported having no conflicts of interest.
SOURCE: Gleeson PK. J Allergy Clin Immunol Pract. 2019 Jun 5. doi: 10.1016/j.jaip.2019.05.048.
, a study has found.
“Incorrect inhaler technique cannot explain the poor disease control in our patient population,” wrote Patrick K. Gleeson, MD, of the University of Pennsylvania, Philadelphia, and coinvestigators. Their report is in the Journal of Allergy and Clinical Immunology: In Practice. “In individuals with poorly controlled asthma, other factors contributing to disease mortality must be considered.”
The 586 patients in the study were observed using their inhalers, and their technique was scored by way of a checklist developed for the study. Inhaler technique – widely regarded as a risk factor for poor disease control – was “better than expected,” the investigators reported, with 56% of patients using metered dose inhalers and 64% of those using dry powder inhalers not making any errors.
“The seeming disassociation between subjects’ asthma control and inhaler technique is counterintuitive, and may be explained by important baseline characteristics in our patients,” they wrote. For instance, participants had suboptimal living conditions in lower income Philadelphia neighborhoods. Almost a quarter – 23% – were current smokers, and almost half were Medicaid recipients. In addition, their mean body mass index was 35.1 kg/m2.
The investigators hypothesized that patients with lower health literacy would have poorer technique but found instead that technique did not vary by reading comprehension or numeracy levels.
More than half of the adults in the study had uncontrolled asthma as defined by prednisone use, an emergency department visit, or a hospitalization for asthma in the past 12 months. A subset had moderate to severe disease per a physician’s diagnosis, forced expiratory volume in 1 second less than 80% predicted, and improvement with a bronchodilator. All patients, however, were considered to have uncontrolled asthma.
There is “uncertainty” in the field about how to measure inhaler technique, and the technique checklist used in the study “may have omitted potentially important errors,” the investigators noted. Still, “good technique predominated among our [population of vulnerable patients].”
The project was supported through awards from the National Institutes of Health/National Heart, Lung, and Blood Institute and the Patient-Centered Outcomes Research Institute.
Coinvestigator Andrea J. Apter, MD, reported that she consults for UpToDate and is an associate editor for the journal. Coinvestigator Knashawn H. Morales, ScD, reported owning stock in Altria Group, British American Tobacco, and Philip Morris International. The other authors reported having no conflicts of interest.
SOURCE: Gleeson PK. J Allergy Clin Immunol Pract. 2019 Jun 5. doi: 10.1016/j.jaip.2019.05.048.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY: IN PRACTICE
Key clinical point: Factors other than inhaler technique should be considered to explain uncontrolled asthma in a low-income, inner-city population.
Major finding: In the study, 56% of patients using metered dose inhalers and 64% of those using dry powder inhalers were using their devices correctly.
Study details: In all, 586 patients were observed using their inhalers, and their technique was scored by way of a checklist developed for the study.
Disclosures: The National Institutes of Health/National Heart, Lung, and Blood Institute and the Patient-Centered Outcomes Research Institute supported the study. Coinvestigator Andrea J. Apter, MD, consults for UpToDate and is an associate editor for the journal. Coinvestigator Knashawn H. Morales, ScD, reported owning stock in Altria Group, British American Tobacco, and Philip Morris International. The other authors reported having no conflicts of interest.
Source: Gleeson PK. J Allergy Clin Immunol Pract. 2019 Jun 5. doi: 10.1016/j.jaip.2019.05.048.
COPD rates reflect current smoking prevalence
, according to a Centers for Disease Control and Prevention analysis of respondents to a behavioral risk factor survey.
“Population-based strategies for smoking prevention and control have the potential to decrease the prevalence of COPD in the United States,” wrote Anne G. Wheaton, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and coauthors. The study was published in the Morbidity and Mortality Weekly Report.
Dr. Wheaton and her fellow researchers analyzed data from 418,378 adult respondents to the 2017 Behavioral Risk Factor Surveillance System survey. Responses came from all 50 states and Washington, D.C.; respondents who had smoked less than 100 lifetime cigarettes were categorized as “never smoked,” while those who had smoked at least 100 cigarettes but no longer smoked were categorized as “former smokers.” Anyone who had smoked at least 100 cigarettes and currently smoked was categorized as a “current smoker.”
The age-adjusted prevalence of COPD among U.S. adults was 6.2% (95% confidence interval, 6.0%-6.3%) in 2017. Current cigarette smokers had a prevalence of 15.2% (95% CI, 14.7%-15.7%); this dipped to 7.6% (95% CI, 7.3%-8.0%) among former smokers and 2.8% (95% CI, 2.7%-2.9%) among adults who had never smoked. Patterns were visible within states: Current smokers had a state-level prevalence of COPD that was strongly correlated with state-level current smoking prevalence (Pearson correlation coefficient, 0.69; P less than .001). State-level COPD prevalence among former smokers (Pearson correlation coefficient, 0.71; P less than .001) and those who never smoked (Pearson correlation coefficient, 0.64; P less than .001) were also strongly correlated with the current smoking prevalence, indicating secondhand smoke as a risk factor for COPD.
The coauthors acknowledged the study’s limitations, including relying on self-reporting for both COPD and smoking status. They also noted that there was no way to measure exposure to secondhand smoke, other indoor or outdoor air pollutants, or respiratory infection history, “all of which might contribute to COPD risk.”
No conflicts of interest were reported.
SOURCE: Wheaton AG et al. MMWR Morb Mortal Wkly Rep. 2019 Jun 21;68(24):533-8.
, according to a Centers for Disease Control and Prevention analysis of respondents to a behavioral risk factor survey.
“Population-based strategies for smoking prevention and control have the potential to decrease the prevalence of COPD in the United States,” wrote Anne G. Wheaton, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and coauthors. The study was published in the Morbidity and Mortality Weekly Report.
Dr. Wheaton and her fellow researchers analyzed data from 418,378 adult respondents to the 2017 Behavioral Risk Factor Surveillance System survey. Responses came from all 50 states and Washington, D.C.; respondents who had smoked less than 100 lifetime cigarettes were categorized as “never smoked,” while those who had smoked at least 100 cigarettes but no longer smoked were categorized as “former smokers.” Anyone who had smoked at least 100 cigarettes and currently smoked was categorized as a “current smoker.”
The age-adjusted prevalence of COPD among U.S. adults was 6.2% (95% confidence interval, 6.0%-6.3%) in 2017. Current cigarette smokers had a prevalence of 15.2% (95% CI, 14.7%-15.7%); this dipped to 7.6% (95% CI, 7.3%-8.0%) among former smokers and 2.8% (95% CI, 2.7%-2.9%) among adults who had never smoked. Patterns were visible within states: Current smokers had a state-level prevalence of COPD that was strongly correlated with state-level current smoking prevalence (Pearson correlation coefficient, 0.69; P less than .001). State-level COPD prevalence among former smokers (Pearson correlation coefficient, 0.71; P less than .001) and those who never smoked (Pearson correlation coefficient, 0.64; P less than .001) were also strongly correlated with the current smoking prevalence, indicating secondhand smoke as a risk factor for COPD.
The coauthors acknowledged the study’s limitations, including relying on self-reporting for both COPD and smoking status. They also noted that there was no way to measure exposure to secondhand smoke, other indoor or outdoor air pollutants, or respiratory infection history, “all of which might contribute to COPD risk.”
No conflicts of interest were reported.
SOURCE: Wheaton AG et al. MMWR Morb Mortal Wkly Rep. 2019 Jun 21;68(24):533-8.
, according to a Centers for Disease Control and Prevention analysis of respondents to a behavioral risk factor survey.
“Population-based strategies for smoking prevention and control have the potential to decrease the prevalence of COPD in the United States,” wrote Anne G. Wheaton, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and coauthors. The study was published in the Morbidity and Mortality Weekly Report.
Dr. Wheaton and her fellow researchers analyzed data from 418,378 adult respondents to the 2017 Behavioral Risk Factor Surveillance System survey. Responses came from all 50 states and Washington, D.C.; respondents who had smoked less than 100 lifetime cigarettes were categorized as “never smoked,” while those who had smoked at least 100 cigarettes but no longer smoked were categorized as “former smokers.” Anyone who had smoked at least 100 cigarettes and currently smoked was categorized as a “current smoker.”
The age-adjusted prevalence of COPD among U.S. adults was 6.2% (95% confidence interval, 6.0%-6.3%) in 2017. Current cigarette smokers had a prevalence of 15.2% (95% CI, 14.7%-15.7%); this dipped to 7.6% (95% CI, 7.3%-8.0%) among former smokers and 2.8% (95% CI, 2.7%-2.9%) among adults who had never smoked. Patterns were visible within states: Current smokers had a state-level prevalence of COPD that was strongly correlated with state-level current smoking prevalence (Pearson correlation coefficient, 0.69; P less than .001). State-level COPD prevalence among former smokers (Pearson correlation coefficient, 0.71; P less than .001) and those who never smoked (Pearson correlation coefficient, 0.64; P less than .001) were also strongly correlated with the current smoking prevalence, indicating secondhand smoke as a risk factor for COPD.
The coauthors acknowledged the study’s limitations, including relying on self-reporting for both COPD and smoking status. They also noted that there was no way to measure exposure to secondhand smoke, other indoor or outdoor air pollutants, or respiratory infection history, “all of which might contribute to COPD risk.”
No conflicts of interest were reported.
SOURCE: Wheaton AG et al. MMWR Morb Mortal Wkly Rep. 2019 Jun 21;68(24):533-8.
FROM MMWR
Vaping among teens increased significantly from 2017 to 2018
according to data from national cross-sectional surveys.
The prevalence of vaping in the past 30 days rose from 11% to 16% in the United States and from 8% to 14.6% in Canada, while use in England showed an nonsignificant increase of 8.7% to 8.9%, David Hammond, PhD, of the University of Waterloo (Canada) and associates said in the BMJ.
Embedded in those U.S. and Canadian increases is the recent evolution of the vaping market brought about by “the growth of JUUL e-cigarettes and similar products [that use] benzoic acid and nicotine salt technology to deliver higher concentrations of nicotine than conventional e-cigarettes,” they explained.
In England, the JUUL system is limited to less than half the nicotine concentration, at 20 mg/mL, compared with more than 50 mg/mL in the United States and Canada, and it was not available at all types of retail outlets at the time of the surveys. That situation changed in March 2019, when the company expanded to convenience stores, the investigators noted.
In the United States, JUUL was the second-most popular product among past–30-day vapers who had a usual brand in 2017, with 9% reporting use. In 2018, JUUL was the most popular brand and use was up to 28%. In Canada, the brand was not among the top five in 2017, but was third in 2018 at 10% in those who reported vaping in the past 30 days. The leading Canadian brand in 2018 was Smok, which released a nicotine-salt version in March of 2018, Dr. Hammond and associates reported.
“Before 2018, there was relatively little evidence of regular vaping among adolescents that might be indicative of nicotine addiction; however, the emergence of JUUL and nicotine salt–based products might signal a change,” they wrote.
The International Tobacco Control Policy Evaluation Project’s Youth Tobacco and Vaping Survey was conducted online in each country in two waves – July to August 2017 and August to September 2018 – with a sample size of approximately 12,000 for each.
The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
SOURCE: Hammond D et al. BMJ 2019 Jun 19. doi: 10.1136/bmj.l2219.
according to data from national cross-sectional surveys.

The prevalence of vaping in the past 30 days rose from 11% to 16% in the United States and from 8% to 14.6% in Canada, while use in England showed an nonsignificant increase of 8.7% to 8.9%, David Hammond, PhD, of the University of Waterloo (Canada) and associates said in the BMJ.
Embedded in those U.S. and Canadian increases is the recent evolution of the vaping market brought about by “the growth of JUUL e-cigarettes and similar products [that use] benzoic acid and nicotine salt technology to deliver higher concentrations of nicotine than conventional e-cigarettes,” they explained.
In England, the JUUL system is limited to less than half the nicotine concentration, at 20 mg/mL, compared with more than 50 mg/mL in the United States and Canada, and it was not available at all types of retail outlets at the time of the surveys. That situation changed in March 2019, when the company expanded to convenience stores, the investigators noted.
In the United States, JUUL was the second-most popular product among past–30-day vapers who had a usual brand in 2017, with 9% reporting use. In 2018, JUUL was the most popular brand and use was up to 28%. In Canada, the brand was not among the top five in 2017, but was third in 2018 at 10% in those who reported vaping in the past 30 days. The leading Canadian brand in 2018 was Smok, which released a nicotine-salt version in March of 2018, Dr. Hammond and associates reported.
“Before 2018, there was relatively little evidence of regular vaping among adolescents that might be indicative of nicotine addiction; however, the emergence of JUUL and nicotine salt–based products might signal a change,” they wrote.
The International Tobacco Control Policy Evaluation Project’s Youth Tobacco and Vaping Survey was conducted online in each country in two waves – July to August 2017 and August to September 2018 – with a sample size of approximately 12,000 for each.
The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
SOURCE: Hammond D et al. BMJ 2019 Jun 19. doi: 10.1136/bmj.l2219.
according to data from national cross-sectional surveys.

The prevalence of vaping in the past 30 days rose from 11% to 16% in the United States and from 8% to 14.6% in Canada, while use in England showed an nonsignificant increase of 8.7% to 8.9%, David Hammond, PhD, of the University of Waterloo (Canada) and associates said in the BMJ.
Embedded in those U.S. and Canadian increases is the recent evolution of the vaping market brought about by “the growth of JUUL e-cigarettes and similar products [that use] benzoic acid and nicotine salt technology to deliver higher concentrations of nicotine than conventional e-cigarettes,” they explained.
In England, the JUUL system is limited to less than half the nicotine concentration, at 20 mg/mL, compared with more than 50 mg/mL in the United States and Canada, and it was not available at all types of retail outlets at the time of the surveys. That situation changed in March 2019, when the company expanded to convenience stores, the investigators noted.
In the United States, JUUL was the second-most popular product among past–30-day vapers who had a usual brand in 2017, with 9% reporting use. In 2018, JUUL was the most popular brand and use was up to 28%. In Canada, the brand was not among the top five in 2017, but was third in 2018 at 10% in those who reported vaping in the past 30 days. The leading Canadian brand in 2018 was Smok, which released a nicotine-salt version in March of 2018, Dr. Hammond and associates reported.
“Before 2018, there was relatively little evidence of regular vaping among adolescents that might be indicative of nicotine addiction; however, the emergence of JUUL and nicotine salt–based products might signal a change,” they wrote.
The International Tobacco Control Policy Evaluation Project’s Youth Tobacco and Vaping Survey was conducted online in each country in two waves – July to August 2017 and August to September 2018 – with a sample size of approximately 12,000 for each.
The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
SOURCE: Hammond D et al. BMJ 2019 Jun 19. doi: 10.1136/bmj.l2219.
FROM THE BMJ
Key clinical point: Recent increases in vaping prevalence among teens “might be indicative of nicotine addiction.”
Major finding: Vaping prevalence increased from 11% to 16% in the United States and from 8% to 14.6% in Canada.
Study details: Two waves of a national, cross-sectional survey that included approximately 12,000 respondents each.
Disclosures: The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
Source: Hammond D et al. BMJ. 2019 Jun 19. doi: 10.1136/bmj.l2219.