User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
AASM hypopnea definition best for detecting OSA cases, study finds
SAN DIEGO – The prevalence of obstructive sleep apnea (OSA) is substantially lower using the Centers for Medicare & Medicaid Services apnea-hypopnea index definition of OSA than using the one recommended by the American Academy of Sleep Medicine.
In addition,
The findings come from an analysis which set out to assess the relationship between OSA and hypertension using the AASM-recommended definition and the 2018 American Heart Association/American College of Cardiology blood pressure guidelines, and to determine if there is an association between hypertension and OSA among individuals who did not meet the CMS definition of OSA.
“Given the substantial morbidity associated with hypertension, these results suggest that universal adoption of the AASM AHI definition would be a reasonable step in ensuring appropriate diagnosis and treatment of OSA,” lead study author Stuart F. Quan, MD, said at the annual meeting of the Associated Professional Sleep Societies.
Dr. Quan, of the division of sleep and circadian disorders at Brigham and Women’s Hospital in Boston, noted that a number of studies have demonstrated that OSA is a risk factor for hypertension and a variety of other medical conditions. “Rightly or wrongly, the most important metric for determining whether OSA is present and determining its severity, is the apnea-hypopnea index,” he said. “It’s the most common metric used for determining OSA severity, and mostly importantly, Medicare and some other insurers use this metric to determine whether a person is eligible for treatment. If a person falls above the line, they can get continuous positive airway pressure, for example. If they’re below the line, that’s too bad; they don’t have OSA insofar as the insurance company is concerned.”
There is no controversy as to what constitutes apnea, he continued, but some disagreement exists on the definition of hypopnea. The AASM recommends using a 3% oxygen desaturation or an arousal, while Medicare uses a definition of hypopnea requiring only a 4% oxygen desaturation. Hypertension definitions have changed recently as well. Before 2018, the definition of hypertension was greater than 140/90 mm Hg for people younger than age 65 years and 150/80 mm Hg for people age 65 years and older. In 2018, the AHA and ACC changed the hypertension guidelines, defining normal as less than 120/80 mm Hg.
“Previous studies linking OSA and hypertension used older definitions, but to my knowledge there are no current studies examining the association between OSA and hypertension using new definitions,” Dr. Quan said.
He reported on results from an analysis of 6,307 participants in the Sleep Heart Health Study who underwent home polysomnography. Their AHI defined by a 3% oxygen desaturation or an arousal was classified into four categories of OSA severity: fewer than 5 events per hour (normal sleep), 5-14 events per hour (mild sleep apnea), 15-29 events per hour (moderate sleep apnea), and 30 or more events per hour (severe sleep apnea).
The researchers used three definitions of dichotomous BP elevation: elevated (greater than 120/80 mm Hg or use of hypertension medications [meds]), stage 1 (greater than 130/80 mm Hg or meds), or stage 2 (greater than 140/90 mm Hg or meds). They used logistic regression to assess the association between elevated BP and/or hypertension and OSA severity, controlling for demographics and body mass index. Additional analyses utilized multiple linear regression to determine the relationship between natural log AHI and systolic and diastolic BP, controlling for the same covariates.
For all definitions of elevated BP, increasing OSA severity was associated with greater likelihood of an elevated or hypertensive status in fully adjusted models. Specifically, the odds ratios among those with elevated BP was 1.30 (95% confidence interval, 1.10-1.54), 1.41 (95% CI, 1.15-1.72), and 1.69 (95% CI, 1.32-2.17) for mild, moderate, and severe sleep apnea, respectively. The ORs among those with stage 1 BP was 1.27 (95% CI, 1.09-1.49), 1.36 (95% CI, 1.13-1.63), 1.58 (95% CI, 1.27-1.97) for mild, moderate, and severe sleep apnea, while the OR among those with stage 2 BP was 1.07 (95% CI, 0.92-1.26), 1.22 (95% CI, 1.02-1.45), 1.38 (95% CI, 1.12-1.69) for mild, moderate, and severe sleep apnea. Linear regression found that AHI was associated with both systolic and diastolic BP in fully adjusted models.
“Using the AASM and CMS AHI definitions, increasing severity of AHI is associated with greater likelihood of having an elevated blood pressure or hypertension,” Dr. Quan concluded. “However, the prevalence of OSA was substantially lower using the CMS definition of OSA. In fact, 218 of these individuals had moderate to severe OSA when the AASM definition was applied.”
He characterized the study as “a practical analysis, a way to help identify patients who might benefit from treatment. This is not the issue of whether the science of 3% AHI is better than 4%.”
The Sleep Heart Health Study was supported by the National Heart, Lung, and Blood Institute. Dr. Quan reported that he helped draft the AASM AHI recommendations but had no other relevant disclosures.
SOURCE: Quan SF et al. SLEEP 2019, Abstract 0501.
SAN DIEGO – The prevalence of obstructive sleep apnea (OSA) is substantially lower using the Centers for Medicare & Medicaid Services apnea-hypopnea index definition of OSA than using the one recommended by the American Academy of Sleep Medicine.
In addition,
The findings come from an analysis which set out to assess the relationship between OSA and hypertension using the AASM-recommended definition and the 2018 American Heart Association/American College of Cardiology blood pressure guidelines, and to determine if there is an association between hypertension and OSA among individuals who did not meet the CMS definition of OSA.
“Given the substantial morbidity associated with hypertension, these results suggest that universal adoption of the AASM AHI definition would be a reasonable step in ensuring appropriate diagnosis and treatment of OSA,” lead study author Stuart F. Quan, MD, said at the annual meeting of the Associated Professional Sleep Societies.
Dr. Quan, of the division of sleep and circadian disorders at Brigham and Women’s Hospital in Boston, noted that a number of studies have demonstrated that OSA is a risk factor for hypertension and a variety of other medical conditions. “Rightly or wrongly, the most important metric for determining whether OSA is present and determining its severity, is the apnea-hypopnea index,” he said. “It’s the most common metric used for determining OSA severity, and mostly importantly, Medicare and some other insurers use this metric to determine whether a person is eligible for treatment. If a person falls above the line, they can get continuous positive airway pressure, for example. If they’re below the line, that’s too bad; they don’t have OSA insofar as the insurance company is concerned.”
There is no controversy as to what constitutes apnea, he continued, but some disagreement exists on the definition of hypopnea. The AASM recommends using a 3% oxygen desaturation or an arousal, while Medicare uses a definition of hypopnea requiring only a 4% oxygen desaturation. Hypertension definitions have changed recently as well. Before 2018, the definition of hypertension was greater than 140/90 mm Hg for people younger than age 65 years and 150/80 mm Hg for people age 65 years and older. In 2018, the AHA and ACC changed the hypertension guidelines, defining normal as less than 120/80 mm Hg.
“Previous studies linking OSA and hypertension used older definitions, but to my knowledge there are no current studies examining the association between OSA and hypertension using new definitions,” Dr. Quan said.
He reported on results from an analysis of 6,307 participants in the Sleep Heart Health Study who underwent home polysomnography. Their AHI defined by a 3% oxygen desaturation or an arousal was classified into four categories of OSA severity: fewer than 5 events per hour (normal sleep), 5-14 events per hour (mild sleep apnea), 15-29 events per hour (moderate sleep apnea), and 30 or more events per hour (severe sleep apnea).
The researchers used three definitions of dichotomous BP elevation: elevated (greater than 120/80 mm Hg or use of hypertension medications [meds]), stage 1 (greater than 130/80 mm Hg or meds), or stage 2 (greater than 140/90 mm Hg or meds). They used logistic regression to assess the association between elevated BP and/or hypertension and OSA severity, controlling for demographics and body mass index. Additional analyses utilized multiple linear regression to determine the relationship between natural log AHI and systolic and diastolic BP, controlling for the same covariates.
For all definitions of elevated BP, increasing OSA severity was associated with greater likelihood of an elevated or hypertensive status in fully adjusted models. Specifically, the odds ratios among those with elevated BP was 1.30 (95% confidence interval, 1.10-1.54), 1.41 (95% CI, 1.15-1.72), and 1.69 (95% CI, 1.32-2.17) for mild, moderate, and severe sleep apnea, respectively. The ORs among those with stage 1 BP was 1.27 (95% CI, 1.09-1.49), 1.36 (95% CI, 1.13-1.63), 1.58 (95% CI, 1.27-1.97) for mild, moderate, and severe sleep apnea, while the OR among those with stage 2 BP was 1.07 (95% CI, 0.92-1.26), 1.22 (95% CI, 1.02-1.45), 1.38 (95% CI, 1.12-1.69) for mild, moderate, and severe sleep apnea. Linear regression found that AHI was associated with both systolic and diastolic BP in fully adjusted models.
“Using the AASM and CMS AHI definitions, increasing severity of AHI is associated with greater likelihood of having an elevated blood pressure or hypertension,” Dr. Quan concluded. “However, the prevalence of OSA was substantially lower using the CMS definition of OSA. In fact, 218 of these individuals had moderate to severe OSA when the AASM definition was applied.”
He characterized the study as “a practical analysis, a way to help identify patients who might benefit from treatment. This is not the issue of whether the science of 3% AHI is better than 4%.”
The Sleep Heart Health Study was supported by the National Heart, Lung, and Blood Institute. Dr. Quan reported that he helped draft the AASM AHI recommendations but had no other relevant disclosures.
SOURCE: Quan SF et al. SLEEP 2019, Abstract 0501.
SAN DIEGO – The prevalence of obstructive sleep apnea (OSA) is substantially lower using the Centers for Medicare & Medicaid Services apnea-hypopnea index definition of OSA than using the one recommended by the American Academy of Sleep Medicine.
In addition,
The findings come from an analysis which set out to assess the relationship between OSA and hypertension using the AASM-recommended definition and the 2018 American Heart Association/American College of Cardiology blood pressure guidelines, and to determine if there is an association between hypertension and OSA among individuals who did not meet the CMS definition of OSA.
“Given the substantial morbidity associated with hypertension, these results suggest that universal adoption of the AASM AHI definition would be a reasonable step in ensuring appropriate diagnosis and treatment of OSA,” lead study author Stuart F. Quan, MD, said at the annual meeting of the Associated Professional Sleep Societies.
Dr. Quan, of the division of sleep and circadian disorders at Brigham and Women’s Hospital in Boston, noted that a number of studies have demonstrated that OSA is a risk factor for hypertension and a variety of other medical conditions. “Rightly or wrongly, the most important metric for determining whether OSA is present and determining its severity, is the apnea-hypopnea index,” he said. “It’s the most common metric used for determining OSA severity, and mostly importantly, Medicare and some other insurers use this metric to determine whether a person is eligible for treatment. If a person falls above the line, they can get continuous positive airway pressure, for example. If they’re below the line, that’s too bad; they don’t have OSA insofar as the insurance company is concerned.”
There is no controversy as to what constitutes apnea, he continued, but some disagreement exists on the definition of hypopnea. The AASM recommends using a 3% oxygen desaturation or an arousal, while Medicare uses a definition of hypopnea requiring only a 4% oxygen desaturation. Hypertension definitions have changed recently as well. Before 2018, the definition of hypertension was greater than 140/90 mm Hg for people younger than age 65 years and 150/80 mm Hg for people age 65 years and older. In 2018, the AHA and ACC changed the hypertension guidelines, defining normal as less than 120/80 mm Hg.
“Previous studies linking OSA and hypertension used older definitions, but to my knowledge there are no current studies examining the association between OSA and hypertension using new definitions,” Dr. Quan said.
He reported on results from an analysis of 6,307 participants in the Sleep Heart Health Study who underwent home polysomnography. Their AHI defined by a 3% oxygen desaturation or an arousal was classified into four categories of OSA severity: fewer than 5 events per hour (normal sleep), 5-14 events per hour (mild sleep apnea), 15-29 events per hour (moderate sleep apnea), and 30 or more events per hour (severe sleep apnea).
The researchers used three definitions of dichotomous BP elevation: elevated (greater than 120/80 mm Hg or use of hypertension medications [meds]), stage 1 (greater than 130/80 mm Hg or meds), or stage 2 (greater than 140/90 mm Hg or meds). They used logistic regression to assess the association between elevated BP and/or hypertension and OSA severity, controlling for demographics and body mass index. Additional analyses utilized multiple linear regression to determine the relationship between natural log AHI and systolic and diastolic BP, controlling for the same covariates.
For all definitions of elevated BP, increasing OSA severity was associated with greater likelihood of an elevated or hypertensive status in fully adjusted models. Specifically, the odds ratios among those with elevated BP was 1.30 (95% confidence interval, 1.10-1.54), 1.41 (95% CI, 1.15-1.72), and 1.69 (95% CI, 1.32-2.17) for mild, moderate, and severe sleep apnea, respectively. The ORs among those with stage 1 BP was 1.27 (95% CI, 1.09-1.49), 1.36 (95% CI, 1.13-1.63), 1.58 (95% CI, 1.27-1.97) for mild, moderate, and severe sleep apnea, while the OR among those with stage 2 BP was 1.07 (95% CI, 0.92-1.26), 1.22 (95% CI, 1.02-1.45), 1.38 (95% CI, 1.12-1.69) for mild, moderate, and severe sleep apnea. Linear regression found that AHI was associated with both systolic and diastolic BP in fully adjusted models.
“Using the AASM and CMS AHI definitions, increasing severity of AHI is associated with greater likelihood of having an elevated blood pressure or hypertension,” Dr. Quan concluded. “However, the prevalence of OSA was substantially lower using the CMS definition of OSA. In fact, 218 of these individuals had moderate to severe OSA when the AASM definition was applied.”
He characterized the study as “a practical analysis, a way to help identify patients who might benefit from treatment. This is not the issue of whether the science of 3% AHI is better than 4%.”
The Sleep Heart Health Study was supported by the National Heart, Lung, and Blood Institute. Dr. Quan reported that he helped draft the AASM AHI recommendations but had no other relevant disclosures.
SOURCE: Quan SF et al. SLEEP 2019, Abstract 0501.
REPORTING FROM SLEEP 2019
Sleepiest OSA patients have worse CV outcomes
SAN ANTONIO – Patients with obstructive sleep apnea who complain of feeling tired when they wake up, being sleepy during the day, and have a high score on the Epworth Sleepiness Scale face an increased risk for cardiovascular disease, results from a population-based analysis suggest.
“OSA is a highly heterogeneous disease, with multiple clinical presentations and consequences,” the study’s first author, Diego R. Mazzotti, PhD, said at the annual meeting of the Associated Professional Sleep Societies. “These patients also have diverse comorbidities, and there are arbitrary severity definitions and variable therapeutic responses. It’s difficult to lump these patients together.”
Symptom subtypes of OSA were originally described in the Icelandic Sleep Apnea Cohort, and defined as excessively sleepy, minimally symptomatic, and disturbed sleep (Eur Respir J. 2014; 44[6]:1600-7). These distinct clusters were identified based on symptom experiences and the existence of major comorbidities. “This concept is more popular today, trying to identify symptom clusters, or groups of individuals, that share similar polysomnographic data, and then compare differences in prevalence or incidence of cardiovascular disease,” said Dr. Mazzotti, a research associate at the University of Pennsylvania, Philadelphia. “That’s a concept that needs to be moving forward.”
Dr. Mazzotti and colleagues set out to determine if OSA symptom subtypes are present in the Sleep Heart Health Study, a multicenter, prospective, community-based cohort of individuals aged 40 years and older designed to assess the cardiovascular (CV) consequences of OSA. They also wanted to know if there is additional evidence of the relevance of OSA symptom subtypes, particularly with respect to cardiovascular disease .
Participant-reported symptoms, such as difficulty falling and staying asleep, snoring, fatigue, drowsy driving and daytime sleepiness, and responses to the Epworth Sleepiness Scale were used to determine the patient’s subtype. Assessments including questionnaires and in-home polysomnography were conducted at baseline (between 1995 and 1998) and follow-up (between 2001 and 2003), while CV outcomes were assessed until the end of follow-up (between 2008 and 2011).
In all, 1,207 patients from the Sleep Heart Health Study met criteria for moderate to severe OSA (apnea-hypopnea index, or AHI, of 15 or greater) and were included in the final analysis. They were followed for a mean of 12 years. Based on the clustering of symptoms, the researchers identified four OSA symptom subtypes: disturbed sleep (12%), minimally symptomatic (33%), excessively sleepy (17%), and moderately sleepy (38%) – proportions that were similar to those observed in prior studies.
The disturbed sleep subtype presented with increased prevalence of “insomnialike” symptoms, such as difficulty initiating or maintaining sleep, according to Dr. Mazzotti. “On the other hand, the excessively sleepy subtype presented with a very high prevalence of several symptoms related to excessive daytime sleepiness, while the moderately sleepy showed a moderately high prevalence of such symptoms, but not as much when compared to the excessively sleepy subtype,” he explained. “Finally, the minimally symptomatic subtype was found to have the lowest prevalence of all investigated symptoms, suggesting that these patients have low symptom burden. They do not complain as much, even though they have moderate-to-severe OSA.”
Next, Dr. Mazzotti and colleagues used Kaplan-Meier survival analysis and Cox proportional hazards models to evaluate whether subtypes were associated with incident coronary heart disease (CHD), heart failure, and CV disease, including CV mortality. Similar analyses were performed comparing each symptom subtype with 2,830 individuals without OSA (AHI less than 5).
Compared with other subtypes, the excessively sleepy group had a more than threefold increased odds of prevalent heart failure, after adjustment for other CV risk factors. They also had a 1.7- to 2.3-fold increased risk for incident CV disease (P less than .001), CHD (P = .015) and heart failure (P = 0.018), after adjustment for other CV risk factors.
“Compared to individuals without OSA, the excessively sleepy subtype is the only subtype with increased risk of incident CV disease and CHD,” Dr. Mazzotti said. “It is possible that excessively sleepy OSA patients are more likely to benefit from CPAP therapy in preventing CV disease.” These results were published online earlier this year (Am J Respir Crit Care Med. 2019 Feb 15. doi: 10.1164/rccm.201808-1509OC).
Dr. Mazzotti reported having no financial disclosures.
SOURCE: Mazzotti D et al. SLEEP 2019, Abstract 0586.
SAN ANTONIO – Patients with obstructive sleep apnea who complain of feeling tired when they wake up, being sleepy during the day, and have a high score on the Epworth Sleepiness Scale face an increased risk for cardiovascular disease, results from a population-based analysis suggest.
“OSA is a highly heterogeneous disease, with multiple clinical presentations and consequences,” the study’s first author, Diego R. Mazzotti, PhD, said at the annual meeting of the Associated Professional Sleep Societies. “These patients also have diverse comorbidities, and there are arbitrary severity definitions and variable therapeutic responses. It’s difficult to lump these patients together.”
Symptom subtypes of OSA were originally described in the Icelandic Sleep Apnea Cohort, and defined as excessively sleepy, minimally symptomatic, and disturbed sleep (Eur Respir J. 2014; 44[6]:1600-7). These distinct clusters were identified based on symptom experiences and the existence of major comorbidities. “This concept is more popular today, trying to identify symptom clusters, or groups of individuals, that share similar polysomnographic data, and then compare differences in prevalence or incidence of cardiovascular disease,” said Dr. Mazzotti, a research associate at the University of Pennsylvania, Philadelphia. “That’s a concept that needs to be moving forward.”
Dr. Mazzotti and colleagues set out to determine if OSA symptom subtypes are present in the Sleep Heart Health Study, a multicenter, prospective, community-based cohort of individuals aged 40 years and older designed to assess the cardiovascular (CV) consequences of OSA. They also wanted to know if there is additional evidence of the relevance of OSA symptom subtypes, particularly with respect to cardiovascular disease .
Participant-reported symptoms, such as difficulty falling and staying asleep, snoring, fatigue, drowsy driving and daytime sleepiness, and responses to the Epworth Sleepiness Scale were used to determine the patient’s subtype. Assessments including questionnaires and in-home polysomnography were conducted at baseline (between 1995 and 1998) and follow-up (between 2001 and 2003), while CV outcomes were assessed until the end of follow-up (between 2008 and 2011).
In all, 1,207 patients from the Sleep Heart Health Study met criteria for moderate to severe OSA (apnea-hypopnea index, or AHI, of 15 or greater) and were included in the final analysis. They were followed for a mean of 12 years. Based on the clustering of symptoms, the researchers identified four OSA symptom subtypes: disturbed sleep (12%), minimally symptomatic (33%), excessively sleepy (17%), and moderately sleepy (38%) – proportions that were similar to those observed in prior studies.
The disturbed sleep subtype presented with increased prevalence of “insomnialike” symptoms, such as difficulty initiating or maintaining sleep, according to Dr. Mazzotti. “On the other hand, the excessively sleepy subtype presented with a very high prevalence of several symptoms related to excessive daytime sleepiness, while the moderately sleepy showed a moderately high prevalence of such symptoms, but not as much when compared to the excessively sleepy subtype,” he explained. “Finally, the minimally symptomatic subtype was found to have the lowest prevalence of all investigated symptoms, suggesting that these patients have low symptom burden. They do not complain as much, even though they have moderate-to-severe OSA.”
Next, Dr. Mazzotti and colleagues used Kaplan-Meier survival analysis and Cox proportional hazards models to evaluate whether subtypes were associated with incident coronary heart disease (CHD), heart failure, and CV disease, including CV mortality. Similar analyses were performed comparing each symptom subtype with 2,830 individuals without OSA (AHI less than 5).
Compared with other subtypes, the excessively sleepy group had a more than threefold increased odds of prevalent heart failure, after adjustment for other CV risk factors. They also had a 1.7- to 2.3-fold increased risk for incident CV disease (P less than .001), CHD (P = .015) and heart failure (P = 0.018), after adjustment for other CV risk factors.
“Compared to individuals without OSA, the excessively sleepy subtype is the only subtype with increased risk of incident CV disease and CHD,” Dr. Mazzotti said. “It is possible that excessively sleepy OSA patients are more likely to benefit from CPAP therapy in preventing CV disease.” These results were published online earlier this year (Am J Respir Crit Care Med. 2019 Feb 15. doi: 10.1164/rccm.201808-1509OC).
Dr. Mazzotti reported having no financial disclosures.
SOURCE: Mazzotti D et al. SLEEP 2019, Abstract 0586.
SAN ANTONIO – Patients with obstructive sleep apnea who complain of feeling tired when they wake up, being sleepy during the day, and have a high score on the Epworth Sleepiness Scale face an increased risk for cardiovascular disease, results from a population-based analysis suggest.
“OSA is a highly heterogeneous disease, with multiple clinical presentations and consequences,” the study’s first author, Diego R. Mazzotti, PhD, said at the annual meeting of the Associated Professional Sleep Societies. “These patients also have diverse comorbidities, and there are arbitrary severity definitions and variable therapeutic responses. It’s difficult to lump these patients together.”
Symptom subtypes of OSA were originally described in the Icelandic Sleep Apnea Cohort, and defined as excessively sleepy, minimally symptomatic, and disturbed sleep (Eur Respir J. 2014; 44[6]:1600-7). These distinct clusters were identified based on symptom experiences and the existence of major comorbidities. “This concept is more popular today, trying to identify symptom clusters, or groups of individuals, that share similar polysomnographic data, and then compare differences in prevalence or incidence of cardiovascular disease,” said Dr. Mazzotti, a research associate at the University of Pennsylvania, Philadelphia. “That’s a concept that needs to be moving forward.”
Dr. Mazzotti and colleagues set out to determine if OSA symptom subtypes are present in the Sleep Heart Health Study, a multicenter, prospective, community-based cohort of individuals aged 40 years and older designed to assess the cardiovascular (CV) consequences of OSA. They also wanted to know if there is additional evidence of the relevance of OSA symptom subtypes, particularly with respect to cardiovascular disease .
Participant-reported symptoms, such as difficulty falling and staying asleep, snoring, fatigue, drowsy driving and daytime sleepiness, and responses to the Epworth Sleepiness Scale were used to determine the patient’s subtype. Assessments including questionnaires and in-home polysomnography were conducted at baseline (between 1995 and 1998) and follow-up (between 2001 and 2003), while CV outcomes were assessed until the end of follow-up (between 2008 and 2011).
In all, 1,207 patients from the Sleep Heart Health Study met criteria for moderate to severe OSA (apnea-hypopnea index, or AHI, of 15 or greater) and were included in the final analysis. They were followed for a mean of 12 years. Based on the clustering of symptoms, the researchers identified four OSA symptom subtypes: disturbed sleep (12%), minimally symptomatic (33%), excessively sleepy (17%), and moderately sleepy (38%) – proportions that were similar to those observed in prior studies.
The disturbed sleep subtype presented with increased prevalence of “insomnialike” symptoms, such as difficulty initiating or maintaining sleep, according to Dr. Mazzotti. “On the other hand, the excessively sleepy subtype presented with a very high prevalence of several symptoms related to excessive daytime sleepiness, while the moderately sleepy showed a moderately high prevalence of such symptoms, but not as much when compared to the excessively sleepy subtype,” he explained. “Finally, the minimally symptomatic subtype was found to have the lowest prevalence of all investigated symptoms, suggesting that these patients have low symptom burden. They do not complain as much, even though they have moderate-to-severe OSA.”
Next, Dr. Mazzotti and colleagues used Kaplan-Meier survival analysis and Cox proportional hazards models to evaluate whether subtypes were associated with incident coronary heart disease (CHD), heart failure, and CV disease, including CV mortality. Similar analyses were performed comparing each symptom subtype with 2,830 individuals without OSA (AHI less than 5).
Compared with other subtypes, the excessively sleepy group had a more than threefold increased odds of prevalent heart failure, after adjustment for other CV risk factors. They also had a 1.7- to 2.3-fold increased risk for incident CV disease (P less than .001), CHD (P = .015) and heart failure (P = 0.018), after adjustment for other CV risk factors.
“Compared to individuals without OSA, the excessively sleepy subtype is the only subtype with increased risk of incident CV disease and CHD,” Dr. Mazzotti said. “It is possible that excessively sleepy OSA patients are more likely to benefit from CPAP therapy in preventing CV disease.” These results were published online earlier this year (Am J Respir Crit Care Med. 2019 Feb 15. doi: 10.1164/rccm.201808-1509OC).
Dr. Mazzotti reported having no financial disclosures.
SOURCE: Mazzotti D et al. SLEEP 2019, Abstract 0586.
REPORTING FROM SLEEP 2019
Risk of cardiac events jumps after COPD exacerbation
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
FROM RESPIROLOGY
Evidence supports accuracy of COPD diagnosis tool
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
FROM JAMA
Data sharing to third parties prevalent in depression, smoking cessation apps
“Mechanisms that potentially enable a small number of dominant online service providers to link information about the use of mental health apps, without either user consent or awareness, appear to be prevalent,” Kit Huckvale, MB ChB, MSc, PhD, of Black Dog Institute at the University of New South Wales Sydney in Randwick, New South Wales, Australia, and colleagues wrote in their study. “Mismatches between declared privacy policies and observed behavior highlight the continuing need for innovation around trust and transparency for health apps.” The study was published in JAMA Network Open.
Dr. Huckvale and colleagues examined the top 36 depression and smoking cessation apps for Android and iOS in the United States accessed in January 2018; Of the apps downloaded, 15 apps were Android-only, 14 apps were iOS-only, and 7 apps were available on both platforms. The apps were assessed over a series of two sessions while network traffic was captured during use, which allowed researchers to determine what personal information was in each data transmission and where the information was going.
There were 25 apps with a privacy policy (69%), 22 of 25 apps (88%) described how that app primarily collected data, and only 16 of 25 apps (64%) provided information on secondary uses of data. Despite 23 of 25 apps (92%) addressing “the possibility of transmission of data to any third party,” 33 of 36 apps overall (92%) transmitted data to third parties. The two most common entities that received third-party data for marketing, advertising, or analytic purposes were Google and Facebook (29 of 36 apps; 81%). However, 12 of 28 apps (43%) that sent data to Google and 6 of 12 apps (50%) that sent data to Facebook disclosed that they would share data with those companies.
The type of data sent to Google and Facebook consisted of a strong identifier to the device or a username (9 of 33 apps; 27%), or a weak identifier in the form of an advertising identifier or a pseudonymous profile that can link users to their behavior on the app and on other products and platforms (26 of 33 apps; 79%).
“As smartphones continue to gain capabilities to collect new forms of personal, biometric, and health information, it is imperative for the health care community to respond with new methods and processes to review apps and ensure they remain safe and protect personal health information,” the researchers concluded.
One of the investigators, Mark E. Larsen, DPhil, reported receiving grants from National Health and Medical Research Council. The other authors reported no relevant conflicts of interest.
SOURCE: Huckvale K et al. JAMA Netw Open. 2019. doi: 10.1001/jamanetworkopen.2019.2542.
“Mechanisms that potentially enable a small number of dominant online service providers to link information about the use of mental health apps, without either user consent or awareness, appear to be prevalent,” Kit Huckvale, MB ChB, MSc, PhD, of Black Dog Institute at the University of New South Wales Sydney in Randwick, New South Wales, Australia, and colleagues wrote in their study. “Mismatches between declared privacy policies and observed behavior highlight the continuing need for innovation around trust and transparency for health apps.” The study was published in JAMA Network Open.
Dr. Huckvale and colleagues examined the top 36 depression and smoking cessation apps for Android and iOS in the United States accessed in January 2018; Of the apps downloaded, 15 apps were Android-only, 14 apps were iOS-only, and 7 apps were available on both platforms. The apps were assessed over a series of two sessions while network traffic was captured during use, which allowed researchers to determine what personal information was in each data transmission and where the information was going.
There were 25 apps with a privacy policy (69%), 22 of 25 apps (88%) described how that app primarily collected data, and only 16 of 25 apps (64%) provided information on secondary uses of data. Despite 23 of 25 apps (92%) addressing “the possibility of transmission of data to any third party,” 33 of 36 apps overall (92%) transmitted data to third parties. The two most common entities that received third-party data for marketing, advertising, or analytic purposes were Google and Facebook (29 of 36 apps; 81%). However, 12 of 28 apps (43%) that sent data to Google and 6 of 12 apps (50%) that sent data to Facebook disclosed that they would share data with those companies.
The type of data sent to Google and Facebook consisted of a strong identifier to the device or a username (9 of 33 apps; 27%), or a weak identifier in the form of an advertising identifier or a pseudonymous profile that can link users to their behavior on the app and on other products and platforms (26 of 33 apps; 79%).
“As smartphones continue to gain capabilities to collect new forms of personal, biometric, and health information, it is imperative for the health care community to respond with new methods and processes to review apps and ensure they remain safe and protect personal health information,” the researchers concluded.
One of the investigators, Mark E. Larsen, DPhil, reported receiving grants from National Health and Medical Research Council. The other authors reported no relevant conflicts of interest.
SOURCE: Huckvale K et al. JAMA Netw Open. 2019. doi: 10.1001/jamanetworkopen.2019.2542.
“Mechanisms that potentially enable a small number of dominant online service providers to link information about the use of mental health apps, without either user consent or awareness, appear to be prevalent,” Kit Huckvale, MB ChB, MSc, PhD, of Black Dog Institute at the University of New South Wales Sydney in Randwick, New South Wales, Australia, and colleagues wrote in their study. “Mismatches between declared privacy policies and observed behavior highlight the continuing need for innovation around trust and transparency for health apps.” The study was published in JAMA Network Open.
Dr. Huckvale and colleagues examined the top 36 depression and smoking cessation apps for Android and iOS in the United States accessed in January 2018; Of the apps downloaded, 15 apps were Android-only, 14 apps were iOS-only, and 7 apps were available on both platforms. The apps were assessed over a series of two sessions while network traffic was captured during use, which allowed researchers to determine what personal information was in each data transmission and where the information was going.
There were 25 apps with a privacy policy (69%), 22 of 25 apps (88%) described how that app primarily collected data, and only 16 of 25 apps (64%) provided information on secondary uses of data. Despite 23 of 25 apps (92%) addressing “the possibility of transmission of data to any third party,” 33 of 36 apps overall (92%) transmitted data to third parties. The two most common entities that received third-party data for marketing, advertising, or analytic purposes were Google and Facebook (29 of 36 apps; 81%). However, 12 of 28 apps (43%) that sent data to Google and 6 of 12 apps (50%) that sent data to Facebook disclosed that they would share data with those companies.
The type of data sent to Google and Facebook consisted of a strong identifier to the device or a username (9 of 33 apps; 27%), or a weak identifier in the form of an advertising identifier or a pseudonymous profile that can link users to their behavior on the app and on other products and platforms (26 of 33 apps; 79%).
“As smartphones continue to gain capabilities to collect new forms of personal, biometric, and health information, it is imperative for the health care community to respond with new methods and processes to review apps and ensure they remain safe and protect personal health information,” the researchers concluded.
One of the investigators, Mark E. Larsen, DPhil, reported receiving grants from National Health and Medical Research Council. The other authors reported no relevant conflicts of interest.
SOURCE: Huckvale K et al. JAMA Netw Open. 2019. doi: 10.1001/jamanetworkopen.2019.2542.
FROM JAMA NETWORK OPEN
Stable COPD: Initiating and Optimizing Therapy
Chronic obstructive pulmonary disease (COPD) is a systemic inflammatory disease characterized by irreversible obstructive ventilatory defects.1-4 It is a major cause of morbidity and mortality, affecting 5% of the population in the United States and ranking as the third leading cause of death in 2008.5,6 The goals in COPD management are to provide symptom relief, improve the quality of life, preserve lung function, and reduce the frequency of exacerbations and mortality. In this 3-part review, we discuss the management of stable COPD in the context of 3 common clinical scenarios: initiating and optimizing therapy, managing acute exacerbations, and managing advanced disease.
Case Presentation
A 65-year-old man with COPD underwent pulmonary function testing (PFT), which demonstrated an obstructive ventilatory defect: forced expiratory volume in 1 second/forced vital capacity ratio (FEV1/FVC), 0.45; FEV1, 2 L (65% of predicted); and diffusing capacity of the lung for carbon monoxide, 15 mL/min/mm Hg (65% of predicted). He has dyspnea with strenuous exercise but is comfortable at rest and with minimal exercise. He has had 1 exacerbation in the past year, and this was treated on an outpatient basis with steroids and antibiotics. His medication regimen includes inhaled tiotropium once daily and inhaled albuterol as needed that he uses roughly twice a week.
What determines the appropriate therapy for a given COPD patient?
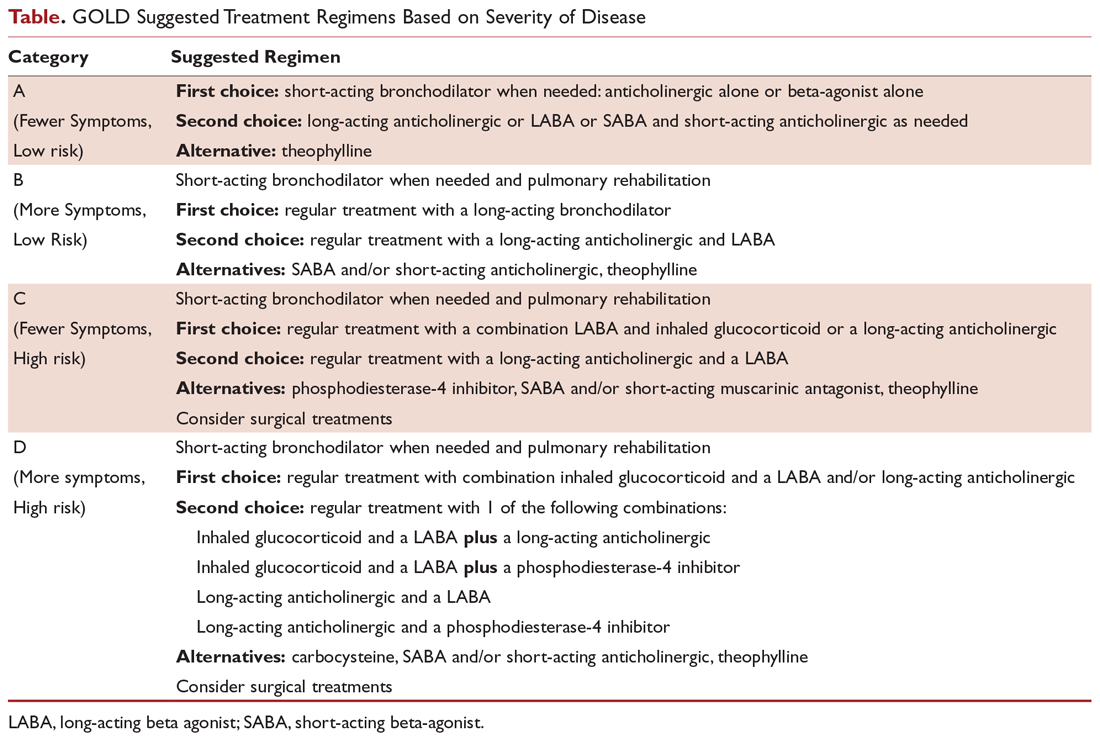
COPD management is guided by disease severity that is measured using a multimodal staging system developed by the Global Initiative for Chronic Obstructive Lung Disease (GOLD). The initial classification adopted by the GOLD 2011 report encompassed 4 categories based on symptoms, number of exacerbations, and degree of airflow limitation on PFT. However, in 2017 the GOLD ABCD classification was modified to consider only symptoms and risk of exacerbation in classifying patients, regardless of performance on spirometry and FEV1 (Figure 1).7,8 This approach was intended to make therapy more individualized based on the patient clinical profile. The Table provides a summary of the recommended treatments according to classification based on the GOLD 2017 report.
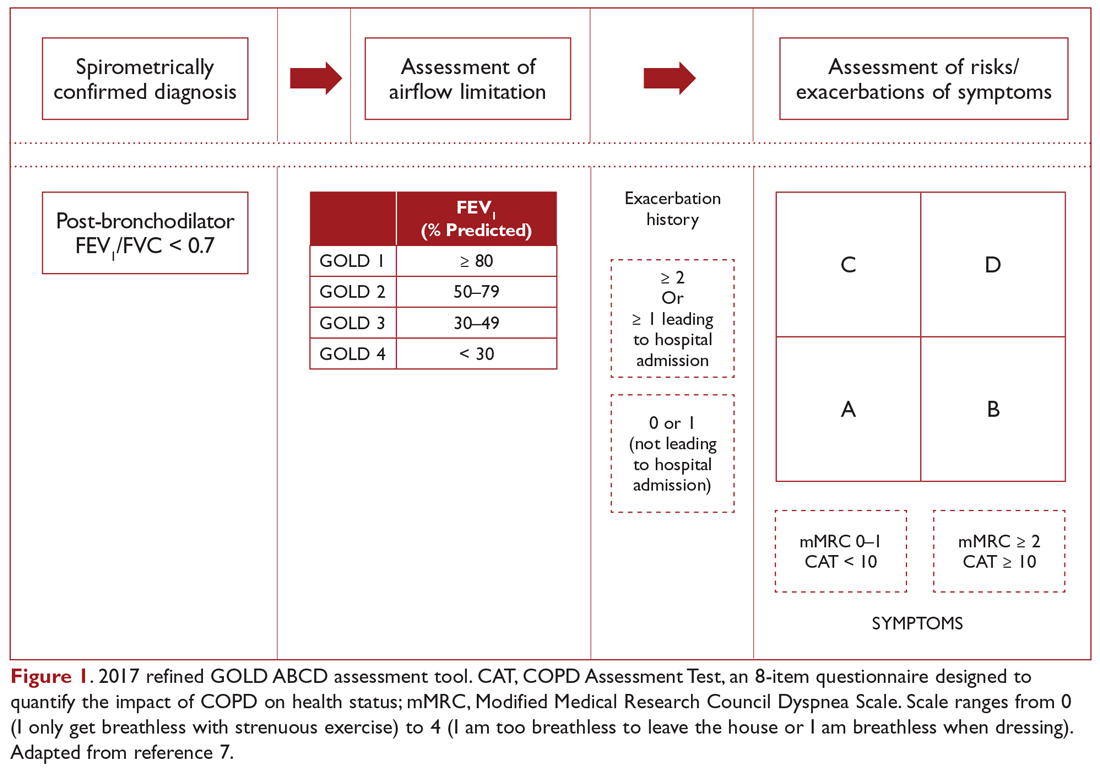
The patient in our clinical scenario can be classified as GOLD category B.
What is the approach to building a pharmacologic regimen for the patient with COPD?
The backbone of the pharmacologic regimen for COPD includes short- and long-acting bronchodilators. They are usually given in an inhaled form to maximize local effects on the lungs and minimize systemic side effects. There are 2 main classes of bronchodilators, beta-agonists and muscarinic antagonists, and each targets specific receptors on the surface of airway smooth muscle cells. Beta- agonists work by stimulating beta-2 receptors, resulting in bronchodilation, while muscarinic antagonists work by blocking the bronchoconstrictor action of M3 muscarinic receptors. Inhaled corticosteroids can be added to long-acting bronchodilator therapy but cannot be used as stand-alone therapy. Theophylline is an oral bronchodilator that is used infrequently due to its narrow therapeutic index, toxicity, and multiple drug interactions.
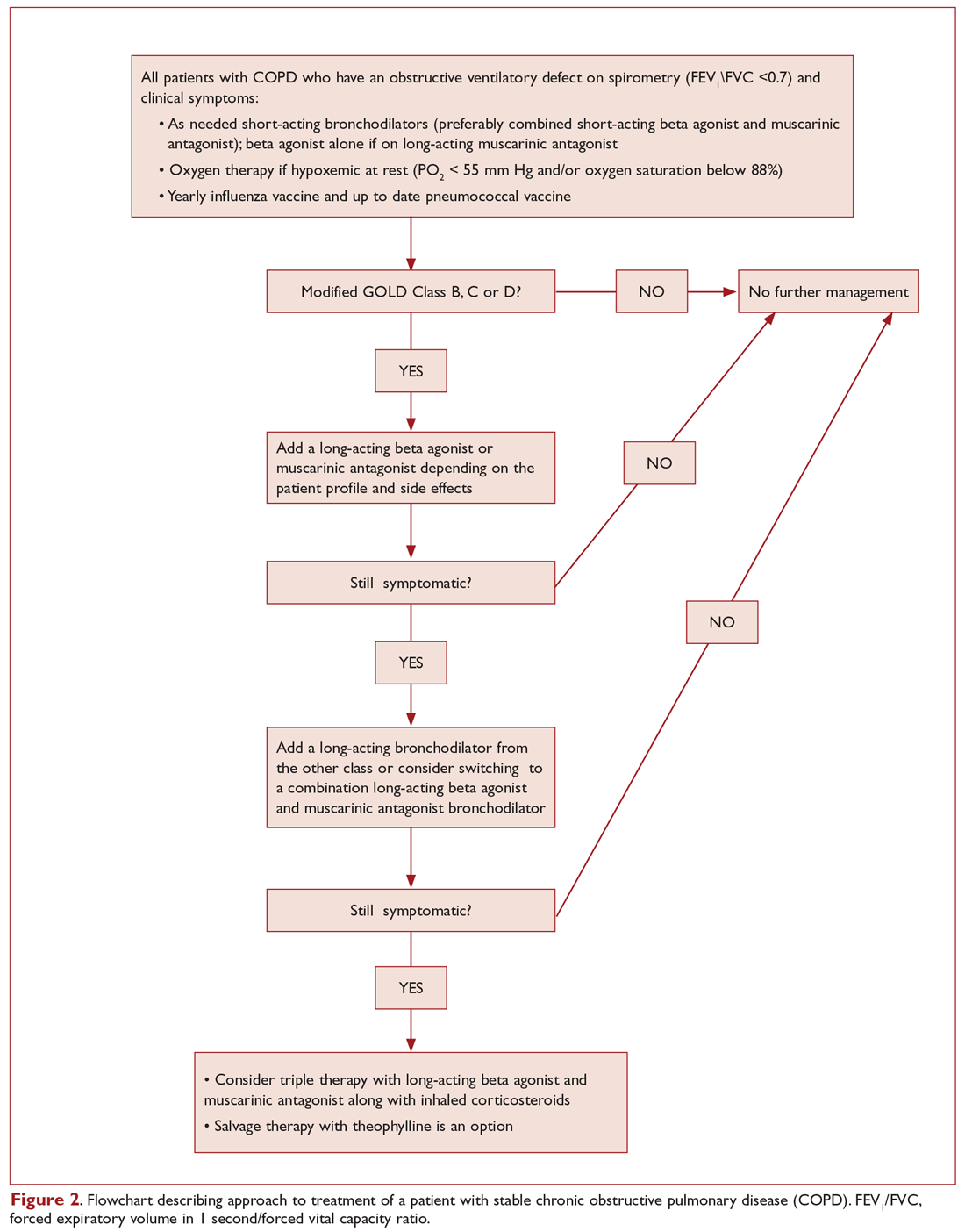
Figure 2 presents an approach to building a treatment plan for the patient with stable COPD.
Who should be on short-acting bronchodilators? What is the best agent? Should it be scheduled or used as needed?
All patients with COPD should be an on inhaled short-acting bronchodilator as needed for relief of symptoms.7 Both short-acting beta-agonists (albuterol and levalbuterol) and short-acting muscarinic antagonists (ipratropium) have been shown in clinical trials and meta-analyses to improve symptoms and lung function in patients with stable COPD9,10 and seem to have comparative efficacy when compared head-to-head in trials.11 However, the airway bronchodilator effect achieved by both classes seems to be additive when used in combination and is also associated with fewer exacerbations compared to albuterol alone.12 On the other hand, adding albuterol to ipratropium increased the bronchodilator response but did not reduce the exacerbation rate.11-13 Inhaled short-acting beta-agonists when used as needed rather than scheduled are associated with less medication use without any significant difference in symptoms or lung function.14
The side effects related to using recommended doses of a short-acting bronchodilator are minimal. In retrospective studies, short-acting beta-agonists increased the risk of severe cardiac arrhythmias.15 Levalbuterol, the active enantiomer of albuterol (R-albuterol) developed for the theoretical benefits of reduced tachycardia, increased tolerability, and better or equal efficacy compared to racemic albuterol, failed to show a clinically significant difference in inducing tachycardia.16 Beta-agonist overuse is associated with tremor and in severe cases hypokalemia, which happens mainly when patients try to achieve maximal bronchodilation; the clinically used doses of beta agonists are associated with fewer side effects but achieve less than maximal bronchodilation.17 Ipratropium can produce systemic anticholinergic side effects, urinary retention being the most clinically significant, especially when combined with long-acting anticholinergic agents.18
In light of the above discussion, a combination of a short-acting beta-agonist and a muscarinic antagonist is recommended in all patients with COPD, unless the patient is on a long-acting muscarinic antagonist (LAMA).7,18 In the latter case, a short-acting beta agonist used as a rescue inhaler is the best option. In our patient, albuterol was the choice for his short-acting bronchodilator, as he was using the LAMA tiotropium.
Are short-acting bronchodilators enough? What do we use for maintenance therapy?
All patients with COPD who are category B or higher according to the modified GOLD staging system should be on a long-acting bronchodilator:7,19 either a long-acting beta-agonist (LABA) or a LAMA. Long-acting bronchodilators work on the same receptors as their short-acting counterparts but have structural differences. Salmeterol is the prototype long-acting selective beta-2 agonist. It is structurally similar to albuterol but has an elongated side chain that allows it to bind firmly to the area of beta receptors and stimulate them repetitively, resulting in an extended-duration of action.20 Tiotropium on the other hand is a quaternary ammonium of ipratropium that is a nonselective muscarinic antagonist.21 Compared to ipratropium, tiotropium dissociates more quickly from M2 receptors, which is responsible for the undesired anticholinergic effects, while at the same time it binds M1 and M3 receptors for a prolonged time, resulting in an extended duration of action.21 Revefenacin is a new lung-selective LAMA that is under development and has shown promise among those with moderate to very severe COPD. Results are only limited to phase 3 trials, and clinical studies are still underway.22
The currently available LABAs include salmeterol, formoterol, arformoterol, olodaterol, and indacaterol. The last 2 have the advantage of once-daily dosing rather than twice daily.23,24 LABAs have been shown to improve lung function, exacerbation rate, and quality of life in multiple clinical trials.23,25 Vilanterol is another LABA that has a long duration of action and can be used once daily,26 but is only available in a combination with umeclidinium, a LAMA. Several LAMAs are approved for use in COPD, including the prototype tiotropium, in addition to aclidinium, umeclidinium, and glycopyrronium. These have been shown in clinical trials to improve lung function, symptoms, and exacerbation rate.27-30
Patients can be started on either a LAMA or LABA depending on the individual patient's needs and the agent's adverse effects.7 Both have comparable adverse effects and efficacy, as detailed below. Concerning adverse effects, there is conflicting data concerning an association of cardiovascular events with both classes of long-acting bronchodilators. While clinical trials failed to show an increased risk,25,31,32 several retrospective studies showed an increased risk of emergency room visits and hospitalizations due to tachyarrhythmias, heart failure, myocardial infarction, and stroke upon initiation of long-acting bronchodilators.33,34 There was no difference in risk for adverse cardiovascular events between LABA and LAMA in 1 Canadian study, and slightly more with LABA in a study using an American database.33,34 Wang et al reported that the risk of cardiovascular adverse effects, defined as hospitalizations and emergency room visits from heart failure, arrythmia, stroke, or ischemia, was 1.5 times the baseline risk in the first 30 days of starting a LABA or LAMA.35 The risk was subsequently the same as baseline or even lower after that period. Urinary retention is another possible complication of LAMA supported by evidence from meta-analyses and retrospective studies, but not clinical trials; the possibility of urinary retention should be discussed with patients upon initiation.36,37 Concerns about increased mortality with the soft mist formulation of tiotropium were put to rest by the Tiotropium Safety and Performance in Respimat (TIOSPIR) trial, which showed no increased mortality compared to Handihaler.38
As far as efficacy and benefits, tiotropium and salmeterol were compared head-to-head in a clinical trial, and tiotropium increased the time before developing first exacerbation and decreased the overall rate of exacerbations.39 No difference in hospitalization rate or mortality was noted in 1 meta-analysis, although tiotropium was more effective in reducing exacerbations.40 The choice of agent should be made based on patient comorbidities and side effects. For example, an elderly patient with severe benign prostatic hyperplasia and urinary retention should try a LABA, while a LAMA would be a better first agent for a patient with severe tachycardia induced by albuterol.
What is the role of inhaled corticosteroids in COPD?
Inhaled corticosteroids (ICS) are believed to work in COPD by reducing airway inflammation.41 ICS should not be used alone for COPD management and are always combined with a LABA.7 Several ICS formulations are approved for use in COPD, including budesonide and fluticasone. ICS has been shown to decrease symptoms and exacerbations, with modest effect on lung function and no change in mortality.42 Side effects include oral candidiasis, dysphonia, and skin bruising.43 There is also an increased risk of pneumonia.44 ICS are best reserved for patients with a component of asthma or asthma–COPD overlap syndrome (ACOS).45 ACOS is characterized by persistent airflow limitation with several features usually associated with asthma and several features usually associated with COPD.46
What if the patient is still symptomatic on a LABA or LAMA?
For patients whose symptoms are not controlled on one class of LABA, recommendations are to add a bronchodilator from the other class.7 There are also multiple combined LAMA-LABA inhalers that are approved in the United States and can possibly improve adherence to therapy. These include tiotropium-olodaterol, umeclidinium-vilanterol, glycopyrronium-indacaterol, and glycopyrrolate-formoterol. In a large systematic review and meta-analysis comparing LABA-LAMA combination to either agent alone, there was a modest improvement in post-bronchodilator FEV1 and quality of life, with no change in hospital admissions, mortality, or adverse effects.47 Interestingly, adding tiotropium to LABA reduced exacerbations, although adding LABA to tiotropium did not.47
Current guidelines recommend that patients in GOLD categories C and D who are not well controlled should receive a combination of LABA-ICS.7 However, a new randomized trial showed better reduction of exacerbations and decreased occurrence of pneumonia in patients receiving LAMA-LABA compared to LABA-ICS.48 In light of this new evidence, it is prudent to use a LAMA-LABA combination before adding ICS.
Triple therapy with LAMA, LABA, and ICS is a common approach for patients with severe uncontrolled disease and has been shown to decrease exacerbations and improve quality of life.7,49 Adding tiotropium to LABA-ICS decreased exacerbations and improved quality of life and airflow in the landmark UPLIFT trial.27 In another clinical trial, triple therapy with LAMA, LABA, and ICS compared to tiotropium alone decreased severe exacerbations, pre-bronchodilator FEV1, and morning symptoms.50 A combination of triple therapy with fluticasone furoate, umeclidinium, and vilanterol was recently noted to result in a lower rate of moderate or severe COPD exacerbations, preserve lung function, and maintain health-related quality of life, as compared with fluticasone furoate/vilanterol or umeclidinium/vilanterol combination therapy among those with symptomatic COPD with a history of exacerbations.51
Is there a role for theophylline? Other agents?
Theophylline
Theophylline is an oral adenosine diphosphate antagonist with indirect adrenergic activity, which is responsible for the bronchodilator therapeutic effect in patients with obstructive lung disease. It is also thought to work by an additional mechanism that decreases inflammation in the airways.52 Theophylline has a serious adverse-effect profile that includes ventricular arrhythmias, seizures, vomiting, and tremor.53 It is metabolized in the liver and has multiple drug interactions and a narrow therapeutic index. It has been shown to improve lung function, gas exchange and symptoms in meta-analysis and clinical trials.54,55
In light of the nature of the adverse effects and the wide array of safer and more effective pharmacologic agents available, theophylline should be avoided early on in the treatment of COPD. Its use can be justified as an add-on therapy in patients with refractory disease on triple therapy for symptomatic relief.53 If used, the therapeutic range of theophylline for COPD is 8 to 12 mcg/mL peak level measured 3 to 7 hours after morning dose, and this level is usually achieved using a daily dose of 10 mg per kilogram of body weight for nonobese patients.56
Systemic Steroids
Oral steroids are used in COPD exacerbations but should never be used chronically in COPD patients, regardless of disease severity, as they increase morbidity and mortality without improving symptoms or lung function.57,58 The dose of systemic steroids should be tapered and finally discontinued.
Mucolytics
Classes of mucolytics include thiol derivatives, inhaled dornase alfa, hypertonic saline, and iodine preparations. Thiol derivatives such as N-acetylcysteine are the most widely studied.59 There is no consistent evidence of beneficial role of mucolytics in COPD patients.7,59 The PANTHEON trial showed decreased exacerbations with N-acetylcysteine (1.16 exacerbations per patient-year compared to 1.49 exacerbations per patient-year in the placebo group; risk ratio, 0.78; 95% CI, 0.67-0.90; P = 0.001) but had methodologic issues including high drop-out rate, exclusion of patients on oxygen, and a large of proportion of nonsmokers.60
Long-Term Antibiotics
There is no role for long-term antibiotics in the management of COPD.7 Macrolides are an exception but are used for their anti-inflammatory effects rather than their antibiotic effects. They should be reserved for patients with frequent exacerbations on optimal therapy and will be discussed later in the review.61
What nonpharmacologic treatments are recommended for COPD patients?
Smoking cessation, oxygen therapy for severe hypoxemia (resting O2 saturation ≤ 88% or PaO2 ≤ 55 mm Hg), vaccination for influenza and pneumococcus, and appropriate nutrition should be provided in all COPD patients. Pulmonary rehabilitation is indicated for patients in GOLD categories B, C, and D.7 It improves symptoms, quality of life, exercise tolerance, and health care utilization. Beneficial effects last for about 2 years.62,63
What other diagnoses should be considered in patients who continue to be symptomatic on optimal therapy?
Other diseases that share the same risk factors as COPD and can contribute to dyspnea, including coronary heart disease, heart failure, thromboembolic disease, and pulmonary hypertension, should be considered. In addition, all patients with refractory disease should have a careful assessment of their inhaler technique, continued smoking, need for oxygen therapy, and associated deconditioning.
1. Segreti A, Stirpe E, Rogliani P, Cazzola M. Defining phenotypes in COPD: an aid to personalized healthcare. Mol Diagn Ther. 2014;18:381-388.
2. Han MK, Agusti A, Calverley PM, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am J Respir Crit Care Med. 2010;182:598-604.
3. Aubier M, Marthan R, Berger P, et al. [COPD and inflammation: statement from a French expert group: inflammation and remodelling mechanisms]. Rev Mal Respir. 2010;27:1254-1266.
4. Wang ZL. Evolving role of systemic inflammation in comorbidities of chronic obstructive pulmonary disease. Chin Med J (Engl). 2010;123:3467-3478.
5. Buist AS, McBurnie MA, Vollmer WM, et al. International variation in the prevalence of COPD (the BOLD Study): a population-based prevalence study. Lancet. 2007;370:741-750.
6. Miniño AM, Murphy SL, Xu J, Kochanek KD. Deaths: final data for 2008. Natl Vital Stat Rep. 2011;59:1-126.
7. Global Initiative for Chronic Obstructive Lung Disease (GOLD): Global strategy for the diagnosis, management, and prevention of COPD 2017. www.goldcopd.org. Accessed July 9, 2019.
8. Jones PW, Harding G, Berry P, et al. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34:648-654.
9. Wadbo M, Löfdahl CG, Larsson K, et al. Effects of formoterol and ipratropium bromide in COPD: a 3-month placebo-controlled study. Eur Respir J. 2002;20:1138-1146.
10. Ram FS, Sestini P. Regular inhaled short acting beta2 agonists for the management of stable chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. Thorax. 2003;58:580-584.
11. Colice GL. Nebulized bronchodilators for outpatient management of stable chronic obstructive pulmonary disease. Am J Med. 1996;100(1A):11S-8S.
12. In chronic obstructive pulmonary disease, a combination of ipratropium and albuterol is more effective than either agent alone. An 85-day multicenter trial. COMBIVENT Inhalation Aerosol Study Group. Chest. 1994;105:1411-1419.
13. Friedman M, Serby CW, Menjoge SS, et al. Pharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPD. Chest. 1999;115:635-641.
14. Cook D, Guyatt G, Wong E, et al. Regular versus as-needed short-acting inhaled beta-agonist therapy for chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001;163:85-90.
15. Wilchesky M, Ernst P, Brophy JM, et al. Bronchodilator use and the risk of arrhythmia in COPD: part 2: reassessment in the larger Quebec cohort. Chest. 2012;142:305-311.
16. Scott VL, Frazee LA. Retrospective comparison of nebulized levalbuterol and albuterol for adverse events in patients with acute airflow obstruction. Am J Ther. 2003;10:341-347.
17. Wong CS, Pavord ID, Williams J, et al. Bronchodilator, cardiovascular, and hypokalaemic effects of fenoterol, salbutamol, and terbutaline in asthma. Lancet. 1990;336:1396-1399.
18. Cole JM, Sheehan AH, Jordan JK. Concomitant use of ipratropium and tiotropium in chronic obstructive pulmonary disease. Ann Pharmacother. 2012;46:1717-1721.
19. Qaseem A, Wilt TJ, Weinberger SE, et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155:179-191.
20. Pearlman DS, Chervinsky P, LaForce C, et al. A comparison of salmeterol with albuterol in the treatment of mild-to-moderate asthma. N Engl J Med. 1992;327:1420-1425.
21. Takahashi T, Belvisi MG, Patel H, et al. Effect of Ba 679 BR, a novel long-acting anticholinergic agent, on cholinergic neurotransmission in guinea pig and human airways. Am J Respir Crit Care Med. 1994;150(6 Pt 1):1640-1645.
22. Ferguson GT, Feldman G, Pudi KK, et al. improvements in lung function with nebulized revefenacin in the treatment of patients with moderate to very severe COPD: results from two replicate phase III clinical trials. Chronic Obstr Pulm Dis. 2019;6:154-165.
23. Donohue JF, Fogarty C, Lötvall J, et al. Once-daily bronchodilators for chronic obstructive pulmonary disease: indacaterol versus tiotropium. Am J Respir Crit Care Med. 2010;182:155-162.
24. Koch A, Pizzichini E, Hamilton A, et al. Lung function efficacy and symptomatic benefit of olodaterol once daily delivered via Respimat versus placebo and formoterol twice daily in patients with GOLD 2-4 COPD: results from two replicate 48-week studies. Int J Chron Obstruct Pulmon Dis. 2014;9:697-714.
25. Calverley PM, Anderson JA, Celli B, et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356:775-789.
26. Hanania NA, Feldman G, Zachgo W, et al. The efficacy and safety of the novel long-acting β2 agonist vilanterol in patients with COPD: a randomized placebo-controlled trial. Chest. 2012;142:119-127.
27. Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359:1543-1554.
28. Decramer ML, Chapman KR, Dahl R, et al. Once-daily indacaterol versus tiotropium for patients with severe chronic obstructive pulmonary disease (INVIGORATE): a randomised, blinded, parallel-group study. Lancet Respir Med. 2013;1:524-533.
29. Jones PW, Singh D, Bateman ED, et al. Efficacy and safety of twice-daily aclidinium bromide in COPD patients: the ATTAIN study. Eur Respir J. 2012;40:830-836.
30. D’Urzo A, Ferguson GT, van Noord JA, et al. Efficacy and safety of once-daily NVA237 in patients with moderate-to-severe COPD: the GLOW1 trial. Respir Res. 2011;12:156.
31. Antoniu SA. UPLIFT Study: the effects of long-term therapy with inhaled tiotropium in chronic obstructive pulmonary disease. Evaluation of: Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359:1543-1554. Expert Opin Pharmacother. 2009;10:719–22.
32. Nelson HS, Gross NJ, Levine B, et al. Cardiac safety profile of nebulized formoterol in adults with COPD: a 12-week, multicenter, randomized, double- blind, double-dummy, placebo- and active-controlled trial. Clin Ther. 2007;29:2167-2178.
33. Gershon A, Croxford R, Calzavara A, et al. Cardiovascular safety of inhaled long-acting bronchodilators in individuals with chronic obstructive pulmonary disease. JAMA Intern Med. 2013;173:1175-1185.
34. Aljaafareh A, Valle JR, Lin YL, et al. Risk of cardiovascular events after initiation of long-acting bronchodilators in patients with chronic obstructive lung disease: A population-based study. SAGE Open Med. 2016;4:2050312116671337.
35. Wang MT, Liou JT, Lin CW, et al. Association of cardiovascular risk with inhaled long-acting bronchodilators in patients with chronic obstructive pulmonary disease: a nested case-Control Study. JAMA Intern Med. 2018;178:229-238.
36. O’Connor AB. Tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2009;360:185-186.
37. Kesten S, Jara M, Wentworth C, Lanes S. Pooled clinical trial analysis of tiotropium safety. Chest. 2006;130:1695-1703.
38. Wise RA, Anzueto A, Cotton D, et al. Tiotropium Respimat inhaler and the risk of death in COPD. N Engl J Med. 2013;369:1491-1501.
39. Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364:1093-1103.
40. Chong J, Karner C, Poole P. Tiotropium versus long-acting beta-agonists for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012(9):CD009157.
41. Gan WQ, Man SF, Sin DD. Effects of inhaled corticosteroids on sputum cell counts in stable chronic obstructive pulmonary disease: a systematic review and a meta-analysis. BMC Pulm Med. 2005;5:3.
42. Yang IA, Clarke MS, Sim EH, Fong KM. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012(7):CD002991.
43. Roland NJ, Bhalla RK, Earis J. The local side effects of inhaled corticosteroids: current understanding and review of the literature. Chest. 2004;126:213-219.
44. Drummond MB, Dasenbrook EC, Pitz MW, et al. Inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease: a systematic review and meta-analysis. JAMA. 2008;300:2407-2416.
45. Lee SY, Park HY, Kim EK, et al. Combination therapy of inhaled steroids and long-acting beta2-agonists in asthma-COPD overlap syndrome. Int J Chron Obstruct Pulmon Dis. 2016;11:2797-2803.
46. Postma DS, Rabe KF. The asthma-COPD overlap syndrome. N Engl J Med. 2015;373:1241-1249.
47. Farne HA, Cates CJ. Long-acting beta2-agonist in addition to tiotropium versus either tiotropium or long-acting beta2-agonist alone for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015:CD008989.
48. Wedzicha JA, Banerji D, Chapman KR, et al. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. N Engl J Med. 2016;374:2222-2234.
49. Aaron SD, Vandemheen KL, Fergusson D, et al. Tiotropium in combination with placebo, salmeterol, or fluticasone-salmeterol for treatment of chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2007;146:545-555.
50. Welte T, Miravitlles M, Hernandez P, et al. Efficacy and tolerability of budesonide/formoterol added to tiotropium in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2009;180:741-750.
51. Lipson DA, Barnhart, Brealey N, et al; IMPACT Investigators. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378:1671-1680.
52. Gallelli L, Falcone D, Cannataro R, et al. Theophylline action on primary human bronchial epithelial cells under proinflammatory stimuli and steroidal drugs: a therapeutic rationale approach. Drug Des Devel Ther. 2017;11:265-272.
53. Paloucek FP, Rodvold KA. Evaluation of theophylline overdoses and toxicities. Ann Emerg Med. 1988;17:135-144.
54. Ram FS, Jones PW, Castro AA, et al. Oral theophylline for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2002(4):CD003902.
55. Murciano D, Auclair MH, Pariente R, Aubier M. A randomized, controlled trial of theophylline in patients with severe chronic obstructive pulmonary disease. N Engl J Med. 1989;320:1521-1525.
56. Devereux G, Cotton S, Barnes P, et al. Use of low-dose oral theophylline as an adjunct to inhaled corticosteroids in preventing exacerbations of chronic obstructive pulmonary disease: study protocol for a randomised controlled trial. Trials. 2015;16:267.
57. Walters JA, Walters EH, Wood-Baker R. Oral corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2005(3):CD005374.
58. Horita N, Miyazawa N, Morita S, et al. Evidence suggesting that oral corticosteroids increase mortality in stable chronic obstructive pulmonary disease. Respir Res. 2014;15:37.
59. Poole P, Chong J, Cates CJ. Mucolytic agents versus placebo for chronic bronchitis or chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015(7):CD001287.
60. Zheng JP, Wen FQ, Bai CX, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir Med. 2014;2:187-194.
61. Seemungal TA, Wilkinson TM, Hurst JR, et al. Long-term erythromycin therapy is associated with decreased chronic obstructive pulmonary disease exacerbations. Am J Respir Crit Care Med. 2008;178:1139-1147.
62. Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Ann Intern Med. 1995;122:823-832.
63. Güell R, Casan P, Belda J, et al. Long-term effects of outpatient rehabilitation of COPD: a randomized trial. Chest. 2000;117:976-983.
Chronic obstructive pulmonary disease (COPD) is a systemic inflammatory disease characterized by irreversible obstructive ventilatory defects.1-4 It is a major cause of morbidity and mortality, affecting 5% of the population in the United States and ranking as the third leading cause of death in 2008.5,6 The goals in COPD management are to provide symptom relief, improve the quality of life, preserve lung function, and reduce the frequency of exacerbations and mortality. In this 3-part review, we discuss the management of stable COPD in the context of 3 common clinical scenarios: initiating and optimizing therapy, managing acute exacerbations, and managing advanced disease.
Case Presentation
A 65-year-old man with COPD underwent pulmonary function testing (PFT), which demonstrated an obstructive ventilatory defect: forced expiratory volume in 1 second/forced vital capacity ratio (FEV1/FVC), 0.45; FEV1, 2 L (65% of predicted); and diffusing capacity of the lung for carbon monoxide, 15 mL/min/mm Hg (65% of predicted). He has dyspnea with strenuous exercise but is comfortable at rest and with minimal exercise. He has had 1 exacerbation in the past year, and this was treated on an outpatient basis with steroids and antibiotics. His medication regimen includes inhaled tiotropium once daily and inhaled albuterol as needed that he uses roughly twice a week.
What determines the appropriate therapy for a given COPD patient?
COPD management is guided by disease severity that is measured using a multimodal staging system developed by the Global Initiative for Chronic Obstructive Lung Disease (GOLD). The initial classification adopted by the GOLD 2011 report encompassed 4 categories based on symptoms, number of exacerbations, and degree of airflow limitation on PFT. However, in 2017 the GOLD ABCD classification was modified to consider only symptoms and risk of exacerbation in classifying patients, regardless of performance on spirometry and FEV1 (Figure 1).7,8 This approach was intended to make therapy more individualized based on the patient clinical profile. The Table provides a summary of the recommended treatments according to classification based on the GOLD 2017 report.

The patient in our clinical scenario can be classified as GOLD category B.

What is the approach to building a pharmacologic regimen for the patient with COPD?
The backbone of the pharmacologic regimen for COPD includes short- and long-acting bronchodilators. They are usually given in an inhaled form to maximize local effects on the lungs and minimize systemic side effects. There are 2 main classes of bronchodilators, beta-agonists and muscarinic antagonists, and each targets specific receptors on the surface of airway smooth muscle cells. Beta- agonists work by stimulating beta-2 receptors, resulting in bronchodilation, while muscarinic antagonists work by blocking the bronchoconstrictor action of M3 muscarinic receptors. Inhaled corticosteroids can be added to long-acting bronchodilator therapy but cannot be used as stand-alone therapy. Theophylline is an oral bronchodilator that is used infrequently due to its narrow therapeutic index, toxicity, and multiple drug interactions.
Figure 2 presents an approach to building a treatment plan for the patient with stable COPD.

Who should be on short-acting bronchodilators? What is the best agent? Should it be scheduled or used as needed?
All patients with COPD should be an on inhaled short-acting bronchodilator as needed for relief of symptoms.7 Both short-acting beta-agonists (albuterol and levalbuterol) and short-acting muscarinic antagonists (ipratropium) have been shown in clinical trials and meta-analyses to improve symptoms and lung function in patients with stable COPD9,10 and seem to have comparative efficacy when compared head-to-head in trials.11 However, the airway bronchodilator effect achieved by both classes seems to be additive when used in combination and is also associated with fewer exacerbations compared to albuterol alone.12 On the other hand, adding albuterol to ipratropium increased the bronchodilator response but did not reduce the exacerbation rate.11-13 Inhaled short-acting beta-agonists when used as needed rather than scheduled are associated with less medication use without any significant difference in symptoms or lung function.14
The side effects related to using recommended doses of a short-acting bronchodilator are minimal. In retrospective studies, short-acting beta-agonists increased the risk of severe cardiac arrhythmias.15 Levalbuterol, the active enantiomer of albuterol (R-albuterol) developed for the theoretical benefits of reduced tachycardia, increased tolerability, and better or equal efficacy compared to racemic albuterol, failed to show a clinically significant difference in inducing tachycardia.16 Beta-agonist overuse is associated with tremor and in severe cases hypokalemia, which happens mainly when patients try to achieve maximal bronchodilation; the clinically used doses of beta agonists are associated with fewer side effects but achieve less than maximal bronchodilation.17 Ipratropium can produce systemic anticholinergic side effects, urinary retention being the most clinically significant, especially when combined with long-acting anticholinergic agents.18
In light of the above discussion, a combination of a short-acting beta-agonist and a muscarinic antagonist is recommended in all patients with COPD, unless the patient is on a long-acting muscarinic antagonist (LAMA).7,18 In the latter case, a short-acting beta agonist used as a rescue inhaler is the best option. In our patient, albuterol was the choice for his short-acting bronchodilator, as he was using the LAMA tiotropium.
Are short-acting bronchodilators enough? What do we use for maintenance therapy?
All patients with COPD who are category B or higher according to the modified GOLD staging system should be on a long-acting bronchodilator:7,19 either a long-acting beta-agonist (LABA) or a LAMA. Long-acting bronchodilators work on the same receptors as their short-acting counterparts but have structural differences. Salmeterol is the prototype long-acting selective beta-2 agonist. It is structurally similar to albuterol but has an elongated side chain that allows it to bind firmly to the area of beta receptors and stimulate them repetitively, resulting in an extended-duration of action.20 Tiotropium on the other hand is a quaternary ammonium of ipratropium that is a nonselective muscarinic antagonist.21 Compared to ipratropium, tiotropium dissociates more quickly from M2 receptors, which is responsible for the undesired anticholinergic effects, while at the same time it binds M1 and M3 receptors for a prolonged time, resulting in an extended duration of action.21 Revefenacin is a new lung-selective LAMA that is under development and has shown promise among those with moderate to very severe COPD. Results are only limited to phase 3 trials, and clinical studies are still underway.22
The currently available LABAs include salmeterol, formoterol, arformoterol, olodaterol, and indacaterol. The last 2 have the advantage of once-daily dosing rather than twice daily.23,24 LABAs have been shown to improve lung function, exacerbation rate, and quality of life in multiple clinical trials.23,25 Vilanterol is another LABA that has a long duration of action and can be used once daily,26 but is only available in a combination with umeclidinium, a LAMA. Several LAMAs are approved for use in COPD, including the prototype tiotropium, in addition to aclidinium, umeclidinium, and glycopyrronium. These have been shown in clinical trials to improve lung function, symptoms, and exacerbation rate.27-30
Patients can be started on either a LAMA or LABA depending on the individual patient's needs and the agent's adverse effects.7 Both have comparable adverse effects and efficacy, as detailed below. Concerning adverse effects, there is conflicting data concerning an association of cardiovascular events with both classes of long-acting bronchodilators. While clinical trials failed to show an increased risk,25,31,32 several retrospective studies showed an increased risk of emergency room visits and hospitalizations due to tachyarrhythmias, heart failure, myocardial infarction, and stroke upon initiation of long-acting bronchodilators.33,34 There was no difference in risk for adverse cardiovascular events between LABA and LAMA in 1 Canadian study, and slightly more with LABA in a study using an American database.33,34 Wang et al reported that the risk of cardiovascular adverse effects, defined as hospitalizations and emergency room visits from heart failure, arrythmia, stroke, or ischemia, was 1.5 times the baseline risk in the first 30 days of starting a LABA or LAMA.35 The risk was subsequently the same as baseline or even lower after that period. Urinary retention is another possible complication of LAMA supported by evidence from meta-analyses and retrospective studies, but not clinical trials; the possibility of urinary retention should be discussed with patients upon initiation.36,37 Concerns about increased mortality with the soft mist formulation of tiotropium were put to rest by the Tiotropium Safety and Performance in Respimat (TIOSPIR) trial, which showed no increased mortality compared to Handihaler.38
As far as efficacy and benefits, tiotropium and salmeterol were compared head-to-head in a clinical trial, and tiotropium increased the time before developing first exacerbation and decreased the overall rate of exacerbations.39 No difference in hospitalization rate or mortality was noted in 1 meta-analysis, although tiotropium was more effective in reducing exacerbations.40 The choice of agent should be made based on patient comorbidities and side effects. For example, an elderly patient with severe benign prostatic hyperplasia and urinary retention should try a LABA, while a LAMA would be a better first agent for a patient with severe tachycardia induced by albuterol.
What is the role of inhaled corticosteroids in COPD?
Inhaled corticosteroids (ICS) are believed to work in COPD by reducing airway inflammation.41 ICS should not be used alone for COPD management and are always combined with a LABA.7 Several ICS formulations are approved for use in COPD, including budesonide and fluticasone. ICS has been shown to decrease symptoms and exacerbations, with modest effect on lung function and no change in mortality.42 Side effects include oral candidiasis, dysphonia, and skin bruising.43 There is also an increased risk of pneumonia.44 ICS are best reserved for patients with a component of asthma or asthma–COPD overlap syndrome (ACOS).45 ACOS is characterized by persistent airflow limitation with several features usually associated with asthma and several features usually associated with COPD.46
What if the patient is still symptomatic on a LABA or LAMA?
For patients whose symptoms are not controlled on one class of LABA, recommendations are to add a bronchodilator from the other class.7 There are also multiple combined LAMA-LABA inhalers that are approved in the United States and can possibly improve adherence to therapy. These include tiotropium-olodaterol, umeclidinium-vilanterol, glycopyrronium-indacaterol, and glycopyrrolate-formoterol. In a large systematic review and meta-analysis comparing LABA-LAMA combination to either agent alone, there was a modest improvement in post-bronchodilator FEV1 and quality of life, with no change in hospital admissions, mortality, or adverse effects.47 Interestingly, adding tiotropium to LABA reduced exacerbations, although adding LABA to tiotropium did not.47
Current guidelines recommend that patients in GOLD categories C and D who are not well controlled should receive a combination of LABA-ICS.7 However, a new randomized trial showed better reduction of exacerbations and decreased occurrence of pneumonia in patients receiving LAMA-LABA compared to LABA-ICS.48 In light of this new evidence, it is prudent to use a LAMA-LABA combination before adding ICS.
Triple therapy with LAMA, LABA, and ICS is a common approach for patients with severe uncontrolled disease and has been shown to decrease exacerbations and improve quality of life.7,49 Adding tiotropium to LABA-ICS decreased exacerbations and improved quality of life and airflow in the landmark UPLIFT trial.27 In another clinical trial, triple therapy with LAMA, LABA, and ICS compared to tiotropium alone decreased severe exacerbations, pre-bronchodilator FEV1, and morning symptoms.50 A combination of triple therapy with fluticasone furoate, umeclidinium, and vilanterol was recently noted to result in a lower rate of moderate or severe COPD exacerbations, preserve lung function, and maintain health-related quality of life, as compared with fluticasone furoate/vilanterol or umeclidinium/vilanterol combination therapy among those with symptomatic COPD with a history of exacerbations.51
Is there a role for theophylline? Other agents?
Theophylline
Theophylline is an oral adenosine diphosphate antagonist with indirect adrenergic activity, which is responsible for the bronchodilator therapeutic effect in patients with obstructive lung disease. It is also thought to work by an additional mechanism that decreases inflammation in the airways.52 Theophylline has a serious adverse-effect profile that includes ventricular arrhythmias, seizures, vomiting, and tremor.53 It is metabolized in the liver and has multiple drug interactions and a narrow therapeutic index. It has been shown to improve lung function, gas exchange and symptoms in meta-analysis and clinical trials.54,55
In light of the nature of the adverse effects and the wide array of safer and more effective pharmacologic agents available, theophylline should be avoided early on in the treatment of COPD. Its use can be justified as an add-on therapy in patients with refractory disease on triple therapy for symptomatic relief.53 If used, the therapeutic range of theophylline for COPD is 8 to 12 mcg/mL peak level measured 3 to 7 hours after morning dose, and this level is usually achieved using a daily dose of 10 mg per kilogram of body weight for nonobese patients.56
Systemic Steroids
Oral steroids are used in COPD exacerbations but should never be used chronically in COPD patients, regardless of disease severity, as they increase morbidity and mortality without improving symptoms or lung function.57,58 The dose of systemic steroids should be tapered and finally discontinued.
Mucolytics
Classes of mucolytics include thiol derivatives, inhaled dornase alfa, hypertonic saline, and iodine preparations. Thiol derivatives such as N-acetylcysteine are the most widely studied.59 There is no consistent evidence of beneficial role of mucolytics in COPD patients.7,59 The PANTHEON trial showed decreased exacerbations with N-acetylcysteine (1.16 exacerbations per patient-year compared to 1.49 exacerbations per patient-year in the placebo group; risk ratio, 0.78; 95% CI, 0.67-0.90; P = 0.001) but had methodologic issues including high drop-out rate, exclusion of patients on oxygen, and a large of proportion of nonsmokers.60
Long-Term Antibiotics
There is no role for long-term antibiotics in the management of COPD.7 Macrolides are an exception but are used for their anti-inflammatory effects rather than their antibiotic effects. They should be reserved for patients with frequent exacerbations on optimal therapy and will be discussed later in the review.61
What nonpharmacologic treatments are recommended for COPD patients?
Smoking cessation, oxygen therapy for severe hypoxemia (resting O2 saturation ≤ 88% or PaO2 ≤ 55 mm Hg), vaccination for influenza and pneumococcus, and appropriate nutrition should be provided in all COPD patients. Pulmonary rehabilitation is indicated for patients in GOLD categories B, C, and D.7 It improves symptoms, quality of life, exercise tolerance, and health care utilization. Beneficial effects last for about 2 years.62,63
What other diagnoses should be considered in patients who continue to be symptomatic on optimal therapy?
Other diseases that share the same risk factors as COPD and can contribute to dyspnea, including coronary heart disease, heart failure, thromboembolic disease, and pulmonary hypertension, should be considered. In addition, all patients with refractory disease should have a careful assessment of their inhaler technique, continued smoking, need for oxygen therapy, and associated deconditioning.
Chronic obstructive pulmonary disease (COPD) is a systemic inflammatory disease characterized by irreversible obstructive ventilatory defects.1-4 It is a major cause of morbidity and mortality, affecting 5% of the population in the United States and ranking as the third leading cause of death in 2008.5,6 The goals in COPD management are to provide symptom relief, improve the quality of life, preserve lung function, and reduce the frequency of exacerbations and mortality. In this 3-part review, we discuss the management of stable COPD in the context of 3 common clinical scenarios: initiating and optimizing therapy, managing acute exacerbations, and managing advanced disease.
Case Presentation
A 65-year-old man with COPD underwent pulmonary function testing (PFT), which demonstrated an obstructive ventilatory defect: forced expiratory volume in 1 second/forced vital capacity ratio (FEV1/FVC), 0.45; FEV1, 2 L (65% of predicted); and diffusing capacity of the lung for carbon monoxide, 15 mL/min/mm Hg (65% of predicted). He has dyspnea with strenuous exercise but is comfortable at rest and with minimal exercise. He has had 1 exacerbation in the past year, and this was treated on an outpatient basis with steroids and antibiotics. His medication regimen includes inhaled tiotropium once daily and inhaled albuterol as needed that he uses roughly twice a week.
What determines the appropriate therapy for a given COPD patient?
COPD management is guided by disease severity that is measured using a multimodal staging system developed by the Global Initiative for Chronic Obstructive Lung Disease (GOLD). The initial classification adopted by the GOLD 2011 report encompassed 4 categories based on symptoms, number of exacerbations, and degree of airflow limitation on PFT. However, in 2017 the GOLD ABCD classification was modified to consider only symptoms and risk of exacerbation in classifying patients, regardless of performance on spirometry and FEV1 (Figure 1).7,8 This approach was intended to make therapy more individualized based on the patient clinical profile. The Table provides a summary of the recommended treatments according to classification based on the GOLD 2017 report.

The patient in our clinical scenario can be classified as GOLD category B.

What is the approach to building a pharmacologic regimen for the patient with COPD?
The backbone of the pharmacologic regimen for COPD includes short- and long-acting bronchodilators. They are usually given in an inhaled form to maximize local effects on the lungs and minimize systemic side effects. There are 2 main classes of bronchodilators, beta-agonists and muscarinic antagonists, and each targets specific receptors on the surface of airway smooth muscle cells. Beta- agonists work by stimulating beta-2 receptors, resulting in bronchodilation, while muscarinic antagonists work by blocking the bronchoconstrictor action of M3 muscarinic receptors. Inhaled corticosteroids can be added to long-acting bronchodilator therapy but cannot be used as stand-alone therapy. Theophylline is an oral bronchodilator that is used infrequently due to its narrow therapeutic index, toxicity, and multiple drug interactions.
Figure 2 presents an approach to building a treatment plan for the patient with stable COPD.

Who should be on short-acting bronchodilators? What is the best agent? Should it be scheduled or used as needed?
All patients with COPD should be an on inhaled short-acting bronchodilator as needed for relief of symptoms.7 Both short-acting beta-agonists (albuterol and levalbuterol) and short-acting muscarinic antagonists (ipratropium) have been shown in clinical trials and meta-analyses to improve symptoms and lung function in patients with stable COPD9,10 and seem to have comparative efficacy when compared head-to-head in trials.11 However, the airway bronchodilator effect achieved by both classes seems to be additive when used in combination and is also associated with fewer exacerbations compared to albuterol alone.12 On the other hand, adding albuterol to ipratropium increased the bronchodilator response but did not reduce the exacerbation rate.11-13 Inhaled short-acting beta-agonists when used as needed rather than scheduled are associated with less medication use without any significant difference in symptoms or lung function.14
The side effects related to using recommended doses of a short-acting bronchodilator are minimal. In retrospective studies, short-acting beta-agonists increased the risk of severe cardiac arrhythmias.15 Levalbuterol, the active enantiomer of albuterol (R-albuterol) developed for the theoretical benefits of reduced tachycardia, increased tolerability, and better or equal efficacy compared to racemic albuterol, failed to show a clinically significant difference in inducing tachycardia.16 Beta-agonist overuse is associated with tremor and in severe cases hypokalemia, which happens mainly when patients try to achieve maximal bronchodilation; the clinically used doses of beta agonists are associated with fewer side effects but achieve less than maximal bronchodilation.17 Ipratropium can produce systemic anticholinergic side effects, urinary retention being the most clinically significant, especially when combined with long-acting anticholinergic agents.18
In light of the above discussion, a combination of a short-acting beta-agonist and a muscarinic antagonist is recommended in all patients with COPD, unless the patient is on a long-acting muscarinic antagonist (LAMA).7,18 In the latter case, a short-acting beta agonist used as a rescue inhaler is the best option. In our patient, albuterol was the choice for his short-acting bronchodilator, as he was using the LAMA tiotropium.
Are short-acting bronchodilators enough? What do we use for maintenance therapy?
All patients with COPD who are category B or higher according to the modified GOLD staging system should be on a long-acting bronchodilator:7,19 either a long-acting beta-agonist (LABA) or a LAMA. Long-acting bronchodilators work on the same receptors as their short-acting counterparts but have structural differences. Salmeterol is the prototype long-acting selective beta-2 agonist. It is structurally similar to albuterol but has an elongated side chain that allows it to bind firmly to the area of beta receptors and stimulate them repetitively, resulting in an extended-duration of action.20 Tiotropium on the other hand is a quaternary ammonium of ipratropium that is a nonselective muscarinic antagonist.21 Compared to ipratropium, tiotropium dissociates more quickly from M2 receptors, which is responsible for the undesired anticholinergic effects, while at the same time it binds M1 and M3 receptors for a prolonged time, resulting in an extended duration of action.21 Revefenacin is a new lung-selective LAMA that is under development and has shown promise among those with moderate to very severe COPD. Results are only limited to phase 3 trials, and clinical studies are still underway.22
The currently available LABAs include salmeterol, formoterol, arformoterol, olodaterol, and indacaterol. The last 2 have the advantage of once-daily dosing rather than twice daily.23,24 LABAs have been shown to improve lung function, exacerbation rate, and quality of life in multiple clinical trials.23,25 Vilanterol is another LABA that has a long duration of action and can be used once daily,26 but is only available in a combination with umeclidinium, a LAMA. Several LAMAs are approved for use in COPD, including the prototype tiotropium, in addition to aclidinium, umeclidinium, and glycopyrronium. These have been shown in clinical trials to improve lung function, symptoms, and exacerbation rate.27-30
Patients can be started on either a LAMA or LABA depending on the individual patient's needs and the agent's adverse effects.7 Both have comparable adverse effects and efficacy, as detailed below. Concerning adverse effects, there is conflicting data concerning an association of cardiovascular events with both classes of long-acting bronchodilators. While clinical trials failed to show an increased risk,25,31,32 several retrospective studies showed an increased risk of emergency room visits and hospitalizations due to tachyarrhythmias, heart failure, myocardial infarction, and stroke upon initiation of long-acting bronchodilators.33,34 There was no difference in risk for adverse cardiovascular events between LABA and LAMA in 1 Canadian study, and slightly more with LABA in a study using an American database.33,34 Wang et al reported that the risk of cardiovascular adverse effects, defined as hospitalizations and emergency room visits from heart failure, arrythmia, stroke, or ischemia, was 1.5 times the baseline risk in the first 30 days of starting a LABA or LAMA.35 The risk was subsequently the same as baseline or even lower after that period. Urinary retention is another possible complication of LAMA supported by evidence from meta-analyses and retrospective studies, but not clinical trials; the possibility of urinary retention should be discussed with patients upon initiation.36,37 Concerns about increased mortality with the soft mist formulation of tiotropium were put to rest by the Tiotropium Safety and Performance in Respimat (TIOSPIR) trial, which showed no increased mortality compared to Handihaler.38
As far as efficacy and benefits, tiotropium and salmeterol were compared head-to-head in a clinical trial, and tiotropium increased the time before developing first exacerbation and decreased the overall rate of exacerbations.39 No difference in hospitalization rate or mortality was noted in 1 meta-analysis, although tiotropium was more effective in reducing exacerbations.40 The choice of agent should be made based on patient comorbidities and side effects. For example, an elderly patient with severe benign prostatic hyperplasia and urinary retention should try a LABA, while a LAMA would be a better first agent for a patient with severe tachycardia induced by albuterol.
What is the role of inhaled corticosteroids in COPD?
Inhaled corticosteroids (ICS) are believed to work in COPD by reducing airway inflammation.41 ICS should not be used alone for COPD management and are always combined with a LABA.7 Several ICS formulations are approved for use in COPD, including budesonide and fluticasone. ICS has been shown to decrease symptoms and exacerbations, with modest effect on lung function and no change in mortality.42 Side effects include oral candidiasis, dysphonia, and skin bruising.43 There is also an increased risk of pneumonia.44 ICS are best reserved for patients with a component of asthma or asthma–COPD overlap syndrome (ACOS).45 ACOS is characterized by persistent airflow limitation with several features usually associated with asthma and several features usually associated with COPD.46
What if the patient is still symptomatic on a LABA or LAMA?
For patients whose symptoms are not controlled on one class of LABA, recommendations are to add a bronchodilator from the other class.7 There are also multiple combined LAMA-LABA inhalers that are approved in the United States and can possibly improve adherence to therapy. These include tiotropium-olodaterol, umeclidinium-vilanterol, glycopyrronium-indacaterol, and glycopyrrolate-formoterol. In a large systematic review and meta-analysis comparing LABA-LAMA combination to either agent alone, there was a modest improvement in post-bronchodilator FEV1 and quality of life, with no change in hospital admissions, mortality, or adverse effects.47 Interestingly, adding tiotropium to LABA reduced exacerbations, although adding LABA to tiotropium did not.47
Current guidelines recommend that patients in GOLD categories C and D who are not well controlled should receive a combination of LABA-ICS.7 However, a new randomized trial showed better reduction of exacerbations and decreased occurrence of pneumonia in patients receiving LAMA-LABA compared to LABA-ICS.48 In light of this new evidence, it is prudent to use a LAMA-LABA combination before adding ICS.
Triple therapy with LAMA, LABA, and ICS is a common approach for patients with severe uncontrolled disease and has been shown to decrease exacerbations and improve quality of life.7,49 Adding tiotropium to LABA-ICS decreased exacerbations and improved quality of life and airflow in the landmark UPLIFT trial.27 In another clinical trial, triple therapy with LAMA, LABA, and ICS compared to tiotropium alone decreased severe exacerbations, pre-bronchodilator FEV1, and morning symptoms.50 A combination of triple therapy with fluticasone furoate, umeclidinium, and vilanterol was recently noted to result in a lower rate of moderate or severe COPD exacerbations, preserve lung function, and maintain health-related quality of life, as compared with fluticasone furoate/vilanterol or umeclidinium/vilanterol combination therapy among those with symptomatic COPD with a history of exacerbations.51
Is there a role for theophylline? Other agents?
Theophylline
Theophylline is an oral adenosine diphosphate antagonist with indirect adrenergic activity, which is responsible for the bronchodilator therapeutic effect in patients with obstructive lung disease. It is also thought to work by an additional mechanism that decreases inflammation in the airways.52 Theophylline has a serious adverse-effect profile that includes ventricular arrhythmias, seizures, vomiting, and tremor.53 It is metabolized in the liver and has multiple drug interactions and a narrow therapeutic index. It has been shown to improve lung function, gas exchange and symptoms in meta-analysis and clinical trials.54,55
In light of the nature of the adverse effects and the wide array of safer and more effective pharmacologic agents available, theophylline should be avoided early on in the treatment of COPD. Its use can be justified as an add-on therapy in patients with refractory disease on triple therapy for symptomatic relief.53 If used, the therapeutic range of theophylline for COPD is 8 to 12 mcg/mL peak level measured 3 to 7 hours after morning dose, and this level is usually achieved using a daily dose of 10 mg per kilogram of body weight for nonobese patients.56
Systemic Steroids
Oral steroids are used in COPD exacerbations but should never be used chronically in COPD patients, regardless of disease severity, as they increase morbidity and mortality without improving symptoms or lung function.57,58 The dose of systemic steroids should be tapered and finally discontinued.
Mucolytics
Classes of mucolytics include thiol derivatives, inhaled dornase alfa, hypertonic saline, and iodine preparations. Thiol derivatives such as N-acetylcysteine are the most widely studied.59 There is no consistent evidence of beneficial role of mucolytics in COPD patients.7,59 The PANTHEON trial showed decreased exacerbations with N-acetylcysteine (1.16 exacerbations per patient-year compared to 1.49 exacerbations per patient-year in the placebo group; risk ratio, 0.78; 95% CI, 0.67-0.90; P = 0.001) but had methodologic issues including high drop-out rate, exclusion of patients on oxygen, and a large of proportion of nonsmokers.60
Long-Term Antibiotics
There is no role for long-term antibiotics in the management of COPD.7 Macrolides are an exception but are used for their anti-inflammatory effects rather than their antibiotic effects. They should be reserved for patients with frequent exacerbations on optimal therapy and will be discussed later in the review.61
What nonpharmacologic treatments are recommended for COPD patients?
Smoking cessation, oxygen therapy for severe hypoxemia (resting O2 saturation ≤ 88% or PaO2 ≤ 55 mm Hg), vaccination for influenza and pneumococcus, and appropriate nutrition should be provided in all COPD patients. Pulmonary rehabilitation is indicated for patients in GOLD categories B, C, and D.7 It improves symptoms, quality of life, exercise tolerance, and health care utilization. Beneficial effects last for about 2 years.62,63
What other diagnoses should be considered in patients who continue to be symptomatic on optimal therapy?
Other diseases that share the same risk factors as COPD and can contribute to dyspnea, including coronary heart disease, heart failure, thromboembolic disease, and pulmonary hypertension, should be considered. In addition, all patients with refractory disease should have a careful assessment of their inhaler technique, continued smoking, need for oxygen therapy, and associated deconditioning.
1. Segreti A, Stirpe E, Rogliani P, Cazzola M. Defining phenotypes in COPD: an aid to personalized healthcare. Mol Diagn Ther. 2014;18:381-388.
2. Han MK, Agusti A, Calverley PM, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am J Respir Crit Care Med. 2010;182:598-604.
3. Aubier M, Marthan R, Berger P, et al. [COPD and inflammation: statement from a French expert group: inflammation and remodelling mechanisms]. Rev Mal Respir. 2010;27:1254-1266.
4. Wang ZL. Evolving role of systemic inflammation in comorbidities of chronic obstructive pulmonary disease. Chin Med J (Engl). 2010;123:3467-3478.
5. Buist AS, McBurnie MA, Vollmer WM, et al. International variation in the prevalence of COPD (the BOLD Study): a population-based prevalence study. Lancet. 2007;370:741-750.
6. Miniño AM, Murphy SL, Xu J, Kochanek KD. Deaths: final data for 2008. Natl Vital Stat Rep. 2011;59:1-126.
7. Global Initiative for Chronic Obstructive Lung Disease (GOLD): Global strategy for the diagnosis, management, and prevention of COPD 2017. www.goldcopd.org. Accessed July 9, 2019.
8. Jones PW, Harding G, Berry P, et al. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34:648-654.
9. Wadbo M, Löfdahl CG, Larsson K, et al. Effects of formoterol and ipratropium bromide in COPD: a 3-month placebo-controlled study. Eur Respir J. 2002;20:1138-1146.
10. Ram FS, Sestini P. Regular inhaled short acting beta2 agonists for the management of stable chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. Thorax. 2003;58:580-584.
11. Colice GL. Nebulized bronchodilators for outpatient management of stable chronic obstructive pulmonary disease. Am J Med. 1996;100(1A):11S-8S.
12. In chronic obstructive pulmonary disease, a combination of ipratropium and albuterol is more effective than either agent alone. An 85-day multicenter trial. COMBIVENT Inhalation Aerosol Study Group. Chest. 1994;105:1411-1419.
13. Friedman M, Serby CW, Menjoge SS, et al. Pharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPD. Chest. 1999;115:635-641.
14. Cook D, Guyatt G, Wong E, et al. Regular versus as-needed short-acting inhaled beta-agonist therapy for chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001;163:85-90.
15. Wilchesky M, Ernst P, Brophy JM, et al. Bronchodilator use and the risk of arrhythmia in COPD: part 2: reassessment in the larger Quebec cohort. Chest. 2012;142:305-311.
16. Scott VL, Frazee LA. Retrospective comparison of nebulized levalbuterol and albuterol for adverse events in patients with acute airflow obstruction. Am J Ther. 2003;10:341-347.
17. Wong CS, Pavord ID, Williams J, et al. Bronchodilator, cardiovascular, and hypokalaemic effects of fenoterol, salbutamol, and terbutaline in asthma. Lancet. 1990;336:1396-1399.
18. Cole JM, Sheehan AH, Jordan JK. Concomitant use of ipratropium and tiotropium in chronic obstructive pulmonary disease. Ann Pharmacother. 2012;46:1717-1721.
19. Qaseem A, Wilt TJ, Weinberger SE, et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155:179-191.
20. Pearlman DS, Chervinsky P, LaForce C, et al. A comparison of salmeterol with albuterol in the treatment of mild-to-moderate asthma. N Engl J Med. 1992;327:1420-1425.
21. Takahashi T, Belvisi MG, Patel H, et al. Effect of Ba 679 BR, a novel long-acting anticholinergic agent, on cholinergic neurotransmission in guinea pig and human airways. Am J Respir Crit Care Med. 1994;150(6 Pt 1):1640-1645.
22. Ferguson GT, Feldman G, Pudi KK, et al. improvements in lung function with nebulized revefenacin in the treatment of patients with moderate to very severe COPD: results from two replicate phase III clinical trials. Chronic Obstr Pulm Dis. 2019;6:154-165.
23. Donohue JF, Fogarty C, Lötvall J, et al. Once-daily bronchodilators for chronic obstructive pulmonary disease: indacaterol versus tiotropium. Am J Respir Crit Care Med. 2010;182:155-162.
24. Koch A, Pizzichini E, Hamilton A, et al. Lung function efficacy and symptomatic benefit of olodaterol once daily delivered via Respimat versus placebo and formoterol twice daily in patients with GOLD 2-4 COPD: results from two replicate 48-week studies. Int J Chron Obstruct Pulmon Dis. 2014;9:697-714.
25. Calverley PM, Anderson JA, Celli B, et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356:775-789.
26. Hanania NA, Feldman G, Zachgo W, et al. The efficacy and safety of the novel long-acting β2 agonist vilanterol in patients with COPD: a randomized placebo-controlled trial. Chest. 2012;142:119-127.
27. Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359:1543-1554.
28. Decramer ML, Chapman KR, Dahl R, et al. Once-daily indacaterol versus tiotropium for patients with severe chronic obstructive pulmonary disease (INVIGORATE): a randomised, blinded, parallel-group study. Lancet Respir Med. 2013;1:524-533.
29. Jones PW, Singh D, Bateman ED, et al. Efficacy and safety of twice-daily aclidinium bromide in COPD patients: the ATTAIN study. Eur Respir J. 2012;40:830-836.
30. D’Urzo A, Ferguson GT, van Noord JA, et al. Efficacy and safety of once-daily NVA237 in patients with moderate-to-severe COPD: the GLOW1 trial. Respir Res. 2011;12:156.
31. Antoniu SA. UPLIFT Study: the effects of long-term therapy with inhaled tiotropium in chronic obstructive pulmonary disease. Evaluation of: Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359:1543-1554. Expert Opin Pharmacother. 2009;10:719–22.
32. Nelson HS, Gross NJ, Levine B, et al. Cardiac safety profile of nebulized formoterol in adults with COPD: a 12-week, multicenter, randomized, double- blind, double-dummy, placebo- and active-controlled trial. Clin Ther. 2007;29:2167-2178.
33. Gershon A, Croxford R, Calzavara A, et al. Cardiovascular safety of inhaled long-acting bronchodilators in individuals with chronic obstructive pulmonary disease. JAMA Intern Med. 2013;173:1175-1185.
34. Aljaafareh A, Valle JR, Lin YL, et al. Risk of cardiovascular events after initiation of long-acting bronchodilators in patients with chronic obstructive lung disease: A population-based study. SAGE Open Med. 2016;4:2050312116671337.
35. Wang MT, Liou JT, Lin CW, et al. Association of cardiovascular risk with inhaled long-acting bronchodilators in patients with chronic obstructive pulmonary disease: a nested case-Control Study. JAMA Intern Med. 2018;178:229-238.
36. O’Connor AB. Tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2009;360:185-186.
37. Kesten S, Jara M, Wentworth C, Lanes S. Pooled clinical trial analysis of tiotropium safety. Chest. 2006;130:1695-1703.
38. Wise RA, Anzueto A, Cotton D, et al. Tiotropium Respimat inhaler and the risk of death in COPD. N Engl J Med. 2013;369:1491-1501.
39. Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364:1093-1103.
40. Chong J, Karner C, Poole P. Tiotropium versus long-acting beta-agonists for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012(9):CD009157.
41. Gan WQ, Man SF, Sin DD. Effects of inhaled corticosteroids on sputum cell counts in stable chronic obstructive pulmonary disease: a systematic review and a meta-analysis. BMC Pulm Med. 2005;5:3.
42. Yang IA, Clarke MS, Sim EH, Fong KM. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012(7):CD002991.
43. Roland NJ, Bhalla RK, Earis J. The local side effects of inhaled corticosteroids: current understanding and review of the literature. Chest. 2004;126:213-219.
44. Drummond MB, Dasenbrook EC, Pitz MW, et al. Inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease: a systematic review and meta-analysis. JAMA. 2008;300:2407-2416.
45. Lee SY, Park HY, Kim EK, et al. Combination therapy of inhaled steroids and long-acting beta2-agonists in asthma-COPD overlap syndrome. Int J Chron Obstruct Pulmon Dis. 2016;11:2797-2803.
46. Postma DS, Rabe KF. The asthma-COPD overlap syndrome. N Engl J Med. 2015;373:1241-1249.
47. Farne HA, Cates CJ. Long-acting beta2-agonist in addition to tiotropium versus either tiotropium or long-acting beta2-agonist alone for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015:CD008989.
48. Wedzicha JA, Banerji D, Chapman KR, et al. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. N Engl J Med. 2016;374:2222-2234.
49. Aaron SD, Vandemheen KL, Fergusson D, et al. Tiotropium in combination with placebo, salmeterol, or fluticasone-salmeterol for treatment of chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2007;146:545-555.
50. Welte T, Miravitlles M, Hernandez P, et al. Efficacy and tolerability of budesonide/formoterol added to tiotropium in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2009;180:741-750.
51. Lipson DA, Barnhart, Brealey N, et al; IMPACT Investigators. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378:1671-1680.
52. Gallelli L, Falcone D, Cannataro R, et al. Theophylline action on primary human bronchial epithelial cells under proinflammatory stimuli and steroidal drugs: a therapeutic rationale approach. Drug Des Devel Ther. 2017;11:265-272.
53. Paloucek FP, Rodvold KA. Evaluation of theophylline overdoses and toxicities. Ann Emerg Med. 1988;17:135-144.
54. Ram FS, Jones PW, Castro AA, et al. Oral theophylline for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2002(4):CD003902.
55. Murciano D, Auclair MH, Pariente R, Aubier M. A randomized, controlled trial of theophylline in patients with severe chronic obstructive pulmonary disease. N Engl J Med. 1989;320:1521-1525.
56. Devereux G, Cotton S, Barnes P, et al. Use of low-dose oral theophylline as an adjunct to inhaled corticosteroids in preventing exacerbations of chronic obstructive pulmonary disease: study protocol for a randomised controlled trial. Trials. 2015;16:267.
57. Walters JA, Walters EH, Wood-Baker R. Oral corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2005(3):CD005374.
58. Horita N, Miyazawa N, Morita S, et al. Evidence suggesting that oral corticosteroids increase mortality in stable chronic obstructive pulmonary disease. Respir Res. 2014;15:37.
59. Poole P, Chong J, Cates CJ. Mucolytic agents versus placebo for chronic bronchitis or chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015(7):CD001287.
60. Zheng JP, Wen FQ, Bai CX, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir Med. 2014;2:187-194.
61. Seemungal TA, Wilkinson TM, Hurst JR, et al. Long-term erythromycin therapy is associated with decreased chronic obstructive pulmonary disease exacerbations. Am J Respir Crit Care Med. 2008;178:1139-1147.
62. Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Ann Intern Med. 1995;122:823-832.
63. Güell R, Casan P, Belda J, et al. Long-term effects of outpatient rehabilitation of COPD: a randomized trial. Chest. 2000;117:976-983.
1. Segreti A, Stirpe E, Rogliani P, Cazzola M. Defining phenotypes in COPD: an aid to personalized healthcare. Mol Diagn Ther. 2014;18:381-388.
2. Han MK, Agusti A, Calverley PM, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am J Respir Crit Care Med. 2010;182:598-604.
3. Aubier M, Marthan R, Berger P, et al. [COPD and inflammation: statement from a French expert group: inflammation and remodelling mechanisms]. Rev Mal Respir. 2010;27:1254-1266.
4. Wang ZL. Evolving role of systemic inflammation in comorbidities of chronic obstructive pulmonary disease. Chin Med J (Engl). 2010;123:3467-3478.
5. Buist AS, McBurnie MA, Vollmer WM, et al. International variation in the prevalence of COPD (the BOLD Study): a population-based prevalence study. Lancet. 2007;370:741-750.
6. Miniño AM, Murphy SL, Xu J, Kochanek KD. Deaths: final data for 2008. Natl Vital Stat Rep. 2011;59:1-126.
7. Global Initiative for Chronic Obstructive Lung Disease (GOLD): Global strategy for the diagnosis, management, and prevention of COPD 2017. www.goldcopd.org. Accessed July 9, 2019.
8. Jones PW, Harding G, Berry P, et al. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34:648-654.
9. Wadbo M, Löfdahl CG, Larsson K, et al. Effects of formoterol and ipratropium bromide in COPD: a 3-month placebo-controlled study. Eur Respir J. 2002;20:1138-1146.
10. Ram FS, Sestini P. Regular inhaled short acting beta2 agonists for the management of stable chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. Thorax. 2003;58:580-584.
11. Colice GL. Nebulized bronchodilators for outpatient management of stable chronic obstructive pulmonary disease. Am J Med. 1996;100(1A):11S-8S.
12. In chronic obstructive pulmonary disease, a combination of ipratropium and albuterol is more effective than either agent alone. An 85-day multicenter trial. COMBIVENT Inhalation Aerosol Study Group. Chest. 1994;105:1411-1419.
13. Friedman M, Serby CW, Menjoge SS, et al. Pharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPD. Chest. 1999;115:635-641.
14. Cook D, Guyatt G, Wong E, et al. Regular versus as-needed short-acting inhaled beta-agonist therapy for chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001;163:85-90.
15. Wilchesky M, Ernst P, Brophy JM, et al. Bronchodilator use and the risk of arrhythmia in COPD: part 2: reassessment in the larger Quebec cohort. Chest. 2012;142:305-311.
16. Scott VL, Frazee LA. Retrospective comparison of nebulized levalbuterol and albuterol for adverse events in patients with acute airflow obstruction. Am J Ther. 2003;10:341-347.
17. Wong CS, Pavord ID, Williams J, et al. Bronchodilator, cardiovascular, and hypokalaemic effects of fenoterol, salbutamol, and terbutaline in asthma. Lancet. 1990;336:1396-1399.
18. Cole JM, Sheehan AH, Jordan JK. Concomitant use of ipratropium and tiotropium in chronic obstructive pulmonary disease. Ann Pharmacother. 2012;46:1717-1721.
19. Qaseem A, Wilt TJ, Weinberger SE, et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155:179-191.
20. Pearlman DS, Chervinsky P, LaForce C, et al. A comparison of salmeterol with albuterol in the treatment of mild-to-moderate asthma. N Engl J Med. 1992;327:1420-1425.
21. Takahashi T, Belvisi MG, Patel H, et al. Effect of Ba 679 BR, a novel long-acting anticholinergic agent, on cholinergic neurotransmission in guinea pig and human airways. Am J Respir Crit Care Med. 1994;150(6 Pt 1):1640-1645.
22. Ferguson GT, Feldman G, Pudi KK, et al. improvements in lung function with nebulized revefenacin in the treatment of patients with moderate to very severe COPD: results from two replicate phase III clinical trials. Chronic Obstr Pulm Dis. 2019;6:154-165.
23. Donohue JF, Fogarty C, Lötvall J, et al. Once-daily bronchodilators for chronic obstructive pulmonary disease: indacaterol versus tiotropium. Am J Respir Crit Care Med. 2010;182:155-162.
24. Koch A, Pizzichini E, Hamilton A, et al. Lung function efficacy and symptomatic benefit of olodaterol once daily delivered via Respimat versus placebo and formoterol twice daily in patients with GOLD 2-4 COPD: results from two replicate 48-week studies. Int J Chron Obstruct Pulmon Dis. 2014;9:697-714.
25. Calverley PM, Anderson JA, Celli B, et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356:775-789.
26. Hanania NA, Feldman G, Zachgo W, et al. The efficacy and safety of the novel long-acting β2 agonist vilanterol in patients with COPD: a randomized placebo-controlled trial. Chest. 2012;142:119-127.
27. Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359:1543-1554.
28. Decramer ML, Chapman KR, Dahl R, et al. Once-daily indacaterol versus tiotropium for patients with severe chronic obstructive pulmonary disease (INVIGORATE): a randomised, blinded, parallel-group study. Lancet Respir Med. 2013;1:524-533.
29. Jones PW, Singh D, Bateman ED, et al. Efficacy and safety of twice-daily aclidinium bromide in COPD patients: the ATTAIN study. Eur Respir J. 2012;40:830-836.
30. D’Urzo A, Ferguson GT, van Noord JA, et al. Efficacy and safety of once-daily NVA237 in patients with moderate-to-severe COPD: the GLOW1 trial. Respir Res. 2011;12:156.
31. Antoniu SA. UPLIFT Study: the effects of long-term therapy with inhaled tiotropium in chronic obstructive pulmonary disease. Evaluation of: Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359:1543-1554. Expert Opin Pharmacother. 2009;10:719–22.
32. Nelson HS, Gross NJ, Levine B, et al. Cardiac safety profile of nebulized formoterol in adults with COPD: a 12-week, multicenter, randomized, double- blind, double-dummy, placebo- and active-controlled trial. Clin Ther. 2007;29:2167-2178.
33. Gershon A, Croxford R, Calzavara A, et al. Cardiovascular safety of inhaled long-acting bronchodilators in individuals with chronic obstructive pulmonary disease. JAMA Intern Med. 2013;173:1175-1185.
34. Aljaafareh A, Valle JR, Lin YL, et al. Risk of cardiovascular events after initiation of long-acting bronchodilators in patients with chronic obstructive lung disease: A population-based study. SAGE Open Med. 2016;4:2050312116671337.
35. Wang MT, Liou JT, Lin CW, et al. Association of cardiovascular risk with inhaled long-acting bronchodilators in patients with chronic obstructive pulmonary disease: a nested case-Control Study. JAMA Intern Med. 2018;178:229-238.
36. O’Connor AB. Tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2009;360:185-186.
37. Kesten S, Jara M, Wentworth C, Lanes S. Pooled clinical trial analysis of tiotropium safety. Chest. 2006;130:1695-1703.
38. Wise RA, Anzueto A, Cotton D, et al. Tiotropium Respimat inhaler and the risk of death in COPD. N Engl J Med. 2013;369:1491-1501.
39. Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364:1093-1103.
40. Chong J, Karner C, Poole P. Tiotropium versus long-acting beta-agonists for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012(9):CD009157.
41. Gan WQ, Man SF, Sin DD. Effects of inhaled corticosteroids on sputum cell counts in stable chronic obstructive pulmonary disease: a systematic review and a meta-analysis. BMC Pulm Med. 2005;5:3.
42. Yang IA, Clarke MS, Sim EH, Fong KM. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012(7):CD002991.
43. Roland NJ, Bhalla RK, Earis J. The local side effects of inhaled corticosteroids: current understanding and review of the literature. Chest. 2004;126:213-219.
44. Drummond MB, Dasenbrook EC, Pitz MW, et al. Inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease: a systematic review and meta-analysis. JAMA. 2008;300:2407-2416.
45. Lee SY, Park HY, Kim EK, et al. Combination therapy of inhaled steroids and long-acting beta2-agonists in asthma-COPD overlap syndrome. Int J Chron Obstruct Pulmon Dis. 2016;11:2797-2803.
46. Postma DS, Rabe KF. The asthma-COPD overlap syndrome. N Engl J Med. 2015;373:1241-1249.
47. Farne HA, Cates CJ. Long-acting beta2-agonist in addition to tiotropium versus either tiotropium or long-acting beta2-agonist alone for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015:CD008989.
48. Wedzicha JA, Banerji D, Chapman KR, et al. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. N Engl J Med. 2016;374:2222-2234.
49. Aaron SD, Vandemheen KL, Fergusson D, et al. Tiotropium in combination with placebo, salmeterol, or fluticasone-salmeterol for treatment of chronic obstructive pulmonary disease: a randomized trial. Ann Intern Med. 2007;146:545-555.
50. Welte T, Miravitlles M, Hernandez P, et al. Efficacy and tolerability of budesonide/formoterol added to tiotropium in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2009;180:741-750.
51. Lipson DA, Barnhart, Brealey N, et al; IMPACT Investigators. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378:1671-1680.
52. Gallelli L, Falcone D, Cannataro R, et al. Theophylline action on primary human bronchial epithelial cells under proinflammatory stimuli and steroidal drugs: a therapeutic rationale approach. Drug Des Devel Ther. 2017;11:265-272.
53. Paloucek FP, Rodvold KA. Evaluation of theophylline overdoses and toxicities. Ann Emerg Med. 1988;17:135-144.
54. Ram FS, Jones PW, Castro AA, et al. Oral theophylline for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2002(4):CD003902.
55. Murciano D, Auclair MH, Pariente R, Aubier M. A randomized, controlled trial of theophylline in patients with severe chronic obstructive pulmonary disease. N Engl J Med. 1989;320:1521-1525.
56. Devereux G, Cotton S, Barnes P, et al. Use of low-dose oral theophylline as an adjunct to inhaled corticosteroids in preventing exacerbations of chronic obstructive pulmonary disease: study protocol for a randomised controlled trial. Trials. 2015;16:267.
57. Walters JA, Walters EH, Wood-Baker R. Oral corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2005(3):CD005374.
58. Horita N, Miyazawa N, Morita S, et al. Evidence suggesting that oral corticosteroids increase mortality in stable chronic obstructive pulmonary disease. Respir Res. 2014;15:37.
59. Poole P, Chong J, Cates CJ. Mucolytic agents versus placebo for chronic bronchitis or chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015(7):CD001287.
60. Zheng JP, Wen FQ, Bai CX, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir Med. 2014;2:187-194.
61. Seemungal TA, Wilkinson TM, Hurst JR, et al. Long-term erythromycin therapy is associated with decreased chronic obstructive pulmonary disease exacerbations. Am J Respir Crit Care Med. 2008;178:1139-1147.
62. Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Ann Intern Med. 1995;122:823-832.
63. Güell R, Casan P, Belda J, et al. Long-term effects of outpatient rehabilitation of COPD: a randomized trial. Chest. 2000;117:976-983.
Stable COPD: Managing Acute Exacerbations
Case Presentation
A 70-year-old man with severe chronic obstructive pulmonary disease (COPD) on oxygen therapy and obstructive sleep apnea treated with nocturnal continuous positive airway pressure was seen in the pulmonary clinic for evaluation of his dyspnea. He was symptomatic with minimal activity and had chronic cough with some sputum production. He had been hospitalized 3 times over the past 12 months and had been to the emergency department (ED) the same number of times for dyspnea. Pertinent medications included as-needed albuterol inhaler, inhaled steroids, and tiotropium 18 mcg inhaled daily. He demonstrated good inhaler technique. On examination, his vital signs were pulse 99 beats/min, oxygen saturation 94% on 2 L/min of oxygen by nasal cannula, blood pressure 126/72 mm Hg, respiratory rate 15 breaths/min, and body mass index 35 kg/m2. He appeared chronically ill but in no acute distress. No wheezing or rales were heard. He had no lower extremity edema. The remainder of the exam was within normal limits. His last pulmonary function test demonstrated moderate obstruction with significant bronchodilator response to 2 puffs of albuterol. The side effects of chronic steroid therapy were impressed upon the patient and 500 mg of roflumilast was started daily. Over the course of the next 3 months, he had no further exacerbations. Roflumilast was continued. He has not required any further hospitalizations, ED visits, or oral steroid use since the last clinic visit.
What is the significance of acute exacerbations of COPD?
Acute exacerbation of COPD (AECOPD) is a frequently observed complication for many patients with COPD.1,2 AECOPD is associated with accelerated disease progression, augmented decline in health status and quality of life, and increased mortality.3 Exacerbations account for most of the costs associated with COPD. Estimates suggest that the aggregate costs associated with the treatment of AECOPD are between $3.2 and $3.8 billion, and that annual health care costs are 10-fold greater for patients with COPD associated with acute exacerbations than for patients with COPD but without exacerbations.4 Hence, any intervention that could potentially minimize or prevent this complication will have far-reaching benefits to patients with COPD as well as provide significant cost saving.
How is AECOPD defined?
COPD exacerbation is defined as a baseline change of the patient’s dyspnea, cough, and/or sputum that is acute in onset, and may warrant a change in regular medication in a patient with underlying COPD.5 Exacerbation in clinical trials has been defined on the basis of whether an increase in the level of care beyond regular care is required primarily in the hospital or ED.6 Frequent exacerbations are defined as 3 symptom-defined exacerbations per year or 2 per year if defined by the need for therapy with corticosteroids, antibiotics, or both.7
What is the underlying pathophysiology?
AECOPD is associated with enhanced upper and lower airway and systemic inflammation. The bronchial mucosa of stable COPD patients have increased numbers of CD8+ lymphocytes and macrophages. In mild AECOPD, eosinophils are increased in the bronchial mucosa and modest elevation of neutrophils, T lymphocytes (CD3), and TNF-α positive cells has also been reported.2 With more severe AECOPD, airway neutrophils are increased. Oxidative stress is a key factor in the development of airway inflammation in COPD.1 Patients with severe exacerbations have augmented large airway interleukin-8 (IL-8) levels and increased oxidative stress as demonstrated by markers such as hydrogen peroxide and 8-isoprostane.6
How do acute exacerbations affect the course of the disease?
In general, as the severity of the underlying COPD increases, exacerbations become both more severe and more frequent. Patients with frequent exacerbations have a worse quality of life than patients with a history of less frequent exacerbations.8 Frequent exacerbations have also been linked to a decline in lung function, with studies suggesting that there might be a decline of 7 mL in forced expiratory volume in 1 second (FEV1) per lower respiratory tract infection per year,9,10 and approximately 8 mL per year in patients with frequent exacerbations as compared to those with sporadic exacerbations.11
What are the triggers for COPD exacerbation?
Respiratory infections are estimated to trigger approximately two-thirds of exacerbations.2 Viral and bacterial infections cause most exacerbations. The effect of the infective triggers is to increase inflammation, cause bronchoconstriction, edema, and mucus production, with a resultant increase in dynamic hyperinflation.12 Thus, any intervention that reduces inflammation in COPD reduces the number and severity of exacerbations, whereas bronchodilators have an impact on exacerbation by their effects on reducing dynamic hyperinflation. The triggers for the one-third of exacerbations not triggered by infection are postulated to be related to other medical conditions, including pulmonary embolism, aspiration, heart failure, and myocardial ischemia.6
What are the pharmacologic options available for prevention of AECOPD?
In recognition of the importance of preventing COPD exacerbations, the American College of Chest Physicians and Canadian Thoracic Society5 have published an evidence-informed clinical guideline specifically examining the prevention of AECOPD, with the goal of assisting clinicians in providing optimal management for COPD patients. The following pharmacologic agents have been recognized as being effective at reducing the frequency of acute exacerbations without any impact on the severity of COPD itself.
Roflumilast
Phosphodiesterase 4 (PDE4) inhibition appears to have inflammatory-modulating properties in the airways, although the exact mechanism of action is unclear. Some have proposed that it reduces inflammation by inhibiting the breakdown of intracellular cyclic adenosine monophosphate.13 In 2 large clinical trials,14,15 daily use of a PDE4 inhibitor (roflumilast) showed a significant (15%–18%) reduction in yearly AECOPD incidence (approximate number needed to treat: 4). This benefit was seen in patients with GOLD stage 3–4 disease (FEV1 < 50% predicted) with the chronic bronchitic phenotype and who had experienced at least 1 exacerbation in the previous year.
Importantly, these clinical trials specifically prohibited the use of inhaled corticosteroids (ICS) and long-acting muscarinic antagonists (LAMAs). Thus, it remains unclear if PDE4 inhibition should be used as an add-on to ICS/LAMA therapy in patients who continue to have frequent AECOPD or whether PDE4 inhibition could be used instead of these standard therapies in patients with well-controlled daily symptoms without ICS or LAMA therapy but who experience frequent exacerbations.
Of note, earlier trials with roflumilast included patients with ICS and LAMA use.14,16 These trials were focused on FEV1 improvement and found no benefit. It was only in post ad hoc analyses that a reduction in AECOPD in patients with frequent exacerbations was found among those taking roflumilast, regardless of ICS or LAMA use.17 While roflumilast has documented benefit in improving lung function and reducing the rate of exacerbations, it has not been reported to decrease hospitalizations.4 This indicates that although the drug reduces the total number of exacerbations, it may not be as useful in preventing episodes of severe exacerbations of COPD.
Although PDE4 inhibitors are easy to administer (a once-daily pill), they are associated with significant gastrointestinal side effects (diarrhea, nausea, reduced appetite), weight loss, headache, and sleep disturbance.18 Adverse effects tend to occur early during treatment, are reversible, and lessen over time with treatment.6 Studies reported an average unexplained weight loss of 2 kg, and monitoring weight during treatment is advised. In addition, it is important to avoid roflumilast in underweight patients. Roflumilast should also be used with caution in depressed patients.5
N-acetylcysteine
N-acetylcysteine (NAC) reduces the viscosity of respiratory secretions as a result of the cleavage of the disulfide bonds and has been studied as a mucolytic agent to aid in the elimination of respiratory secretions.19 Oral NAC is quickly absorbed and is rapidly present in an active form in lung tissue and respiratory secretions after ingestion. NAC is well-tolerated except for occasional patients with GI adverse effects. The role of NAC in preventing AECOPD has been studied for more than 3 decades,20-22 although the largest clinical trial to date was reported in 2014.23 Taken together, the combined data demonstrate a significant reduction in the rate of COPD exacerbations associated with the use of NAC when compared with placebo (odds ratio [OR], 0.61; 95% confidence interval [CI], 0.37-0.99). Clinical guidelines suggest that in patients with moderate to severe COPD (FEV1/forced vital capacity ratio < 0.7, and FEV1 < 80% predicted) receiving maintenance bronchodilator therapy combined with ICS and history of 2 more exacerbations in the previous 2 years, treatment with oral NAC can be administered to prevent AECOPD.
Macrolides
Continuous prophylactic use of antibiotics in older studies had no effect on the frequency of AECOPD.24,25 But it is known that macrolide antibiotics have several antimicrobial, anti-inflammatory and immunomodulating effects and have been used for many years in the management of other chronic airway disease, including diffuse pan-bronchiolitis and cystic fibrosis.5 One recent study showed that the use of once-daily generic azithromycin 5 days per week appeared to have an impact on AECOPD incidence.26 In this study, the rate of AECOPD was reduced from 1.83 to 1.48 exacerbations per patient-year (relative risk, 0.83; 95% CI, 0.72–0.95; P = 0.01). Azithromycin also prevented severe AECOPD. Greater benefit was obtained with milder forms of the disease and in the elderly. Azithromycin did not appear to provide any benefit in those who continued to smoke (hazard ratio, 0.99).27 Other studies have shown that azithromycin was associated with an increased incidence of bacterial resistance and impaired hearing.28 Overall data from the available clinical trials are robust and demonstrate that regular macrolide therapy definitely reduces the risk of AECOPD. Due to potential adverse effects, however, macrolide therapy is an option rather than a strong recommendation.5 The prescribing clinician also needs to consider potential of prolongation of the QT interval.26
Immunostimulants
Immunostimulants have also been reported to reduce frequency of AECOPD.29,30 Bacterial lysates, reconstituted mixtures of bacterial antigens present in the lower airways of COPD patients, act as immunostimulants through the induction of cellular maturation, stimulating lymphocyte chemotaxis and increasing opsonization when administered to individuals with COPD.6 Studies have demonstrated a reduction in the severe complications of exacerbations and hospital admissions in COPD patients with OM-85, a detoxified oral immunoactive bacterial extract.29,30 However, most of these trials were conducted prior to the routine use of long-acting bronchodilators and ICS in COPD. A study that evaluated the efficacy of ismigen, a bacterial lysate, in reducing AECOPD31 found no difference in the exacerbation rate between ismigen and placebo or the time to first exacerbation. Additional studies are needed to examine the long-term effects of this therapy in patients receiving currently recommended COPD maintenance therapy.6
β-Blockers
Observational studies of β-blocker use in preventing AECOPD have yielded encouraging results, with one study showing a reduction in AECOPD risk (incidence risk ratio, 0.73; 95% CI, 0.60–0.90) in patients receiving β-blockers versus those not on β-blockers.32 Based on these findings, a clinical trial investigating the impact of metoprolol on risk of AECOPD is ongoing.33
Proton Pump Inhibitors
Gastroesophageal reflux disease is an independent risk factor for exacerbations.34 Two small, single-center studies,35,36 have shown that use of lansoprazole decreases the risk and frequency of AECOPD. However, data from the Predicting Outcome using Systemic Markers in Severe Exacerbations of COPD (PROMISE-COPD) study,6 which was a multicenter prospective observational study, suggested that patients with stable COPD receiving a proton pump inhibitor were at high risk of frequent and severe exacerbations.37 Thus, at this stage, their definitive role needs to be defined, possibly with a randomized, placebo-controlled study.
1. Wedzicha JA, Singh R, Mackay AJ. Acute COPD exacerbations. Clin Chest Med. 2014;35:157-163.
2. Wedzicha JA, Seemungal TAR. COPD exacerbations: defining their cause and prevention. Lancet. 2007;370:786-796.
3. Spencer S, Calverley PMA, Burge PS, Jones PW. Impact of preventing exacerbations on deterioration of health status in COPD. Eur Respir J. 2004;23:698-702.
4. Blanchette CM, Gross NJ, Altman P. Rising costs of COPD and the potential for maintenance therapy to slow the trend. Am Health Drug Benef. 2014;7:98.
5. Criner GJ, Bourbeau J, Diekemper RL, et al. Prevention of acute exacerbations of COPD: American College of Chest Physicians and Canadian Thoracic Society Guideline. Chest. 2015;147:894-942.
6. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management and prevention of chronic obstructive lung disease 2017 report. Respirology. 2017;22:575-601.
7. Wedzicha JA, Brill SE, Allinson JP, Donaldson GC. Mechanisms and impact of the frequent exacerbator phenotype in chronic obstructive pulmonary disease. BMC Med. 2013;11:181.
8. Seemungal TAR, Donaldson GC, Paul EA, et al. Effect of exacerbation on quality of life in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;157:1418-1422.
9. Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Ann Intern Med. 1995;122:823-832.
10. Kanner RE, Anthonisen NR, Connett JE. Lower respiratory illnesses promote FEV1 decline in current smokers but not ex-smokers with mild chronic obstructive pulmonary disease: results from the lung health study. Am J Respir Crit Care Med. 2001;164:358-364.
11. Donaldson GC, Seemungal TAR, Bhowmik A, Wedzicha JA. Relationship between exacerbation frequency and lung function decline in chronic obstructive pulmonary disease. Thorax. 2002;57:847-852.
12. Papi A, Bellettato CM, Braccioni F, et al. Infections and airway inflammation in chronic obstructive pulmonary disease severe exacerbations. Am J Respir Crit Care Med. 2006;173:1114-1121.
13. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163:53-67.
14. Calverley PMA, Rabe KF, Goehring U-M, et al. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. Lancet. 2009;374:685-694.
15. Fabbri LM, Calverley PMA, Izquierdo-Alonso JL, et al. Roflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with long-acting bronchodilators: two randomised clinical trials. Lancet. 2009;374:695-703.
16. Lee S, Hui DSC, Mahayiddin AA, et al. Roflumilast in Asian patients with COPD: a randomized placebo-controlled trial. Respirology. 2011;16:1249-1257.
17. Calverley PM, Martinez FJ, Fabbri LM, et al. Does roflumilast decrease exacerbations in severe COPD patients not controlled by inhaled combination therapy? The REACT study protocol. Int J Chron Obstruct Pulmon Dis. 2012;7:375-382.
18. Chong J, Leung B, Poole P. Phosphodiesterase 4 inhibitors for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2013(11):CD002309.
19. Sheffner AL, Medler EM, Jacobs LW, Sarett HP. The in vitro reduction in viscosity of human tracheobronchial secretions by acetylcysteine. Am Rev Respir Dis. 1964;90:721-729.
20. Boman G, Bäcker U, Larsson S, et al. Oral acetylcysteine reduces exacerbation rate in chronic bronchitis: report of a trial organized by the Swedish Society for Pulmonary Diseases. Eur J Respir Dis. 1983;64:405-415.
21. Grassi C, Morandini GC. A controlled trial of intermittent oral acetylcysteine in the long-term treatment of chronic bronchitis. Eur J Clin Pharmacol. 1976;9:393-396.
22. Hansen NCG, Skriver A, Brorsen-Riis L, et al. Orally administered N-acetylcysteine may improve general well-being in patients with mild chronic bronchitis. Respir Med. 1994;88:531-535.
23. Zheng JP, Wen FQ, Bai CX, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir Med. 2014;2:187-194.
24. Francis RS, Spicer CC. Chemotherapy in chronic bronchitis: Influence of daily penicillin and tetracycline on exacerbations and their cost: A report to the research committee of the British Tuberculosis Association by Their Chronic Bronchitis Subcommittee. BMJ. 1960;1:297-303.
25. Francis RS, May JR, Spicer CC. Chemotherapy of bronchitis. BMJ. 1961;2:979.
26. Albert RK, Connett J, Bailey WC, et al. Azithromycin for prevention of exacerbations of COPD. N Engl J Med. 2011;365:689-698.
27. Han MK, Tayob N, Murray S, et al. Predictors of chronic obstructive pulmonary disease exacerbation reduction in response to daily azithromycin therapy. Am J Respir Crit Care Med. 2014;189:1503-1508.
28. Uzun S, Djamin RS, Kluytmans JAJW, et al. Azithromycin maintenance treatment in patients with frequent exacerbations of chronic obstructive pulmonary disease (COLUMBUS): a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2014;2:361-368.
29. Collet JP, Shapiro S, Ernst P, et al. Effects of an immunostimulating agent on acute exacerbations and hospitalizations in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1997;156:1719-1724.
30. Jing LI. Protective effect of a bacterial extract against acute exacerbation in patients with chronic bronchitis accompanied by chronic obstructive pulmonary. Age. 2004;67:828-834.
31. Braido F, Tarantini F, Ghiglione V, et al. Bacterial lysate in the prevention of acute exacerbation of COPD and in respiratory recurrent infections. Int J Chron Obstruct Pulmon Dis. 2007;2:335.
32. Bhatt SP, Wells JM, Kinney GL, et al. β-Blockers are associated with a reduction in COPD exacerbations. Thorax. 2016;71:8-14.
33. Bhatt SP, Connett JE, Voelker H, et al. β-Blockers for the prevention of acute exacerbations of chronic obstructive pulmonary disease (βLOCK COPD): a randomised controlled study protocol. BMJ Open. 2016;6:e012292.
34. Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363:1128-1138.
35. Sasaki T, Nakayama K, Yasuda H, et al. A randomized, single-blind study of lansoprazole for the prevention of exacerbations of chronic obstructive pulmonary disease in older patients. J Am Geriatr Soc. 2009;57:1453-1457.
36. Xiong W, Zhang Qs, Zhao W, et al. A 12-month follow-up study on the preventive effect of oral lansoprazole on acute exacerbation of chronic obstructive pulmonary disease. Int J Exper Pathol. 2016;97:107-113.
37. Baumeler L, Papakonstantinou E, Milenkovic B, et al. Therapy with proton-pump inhibitors for gastroesophageal reflux disease does not reduce the risk for severe exacerbations in COPD. Respirology. 2016;21:883-890.
Case Presentation
A 70-year-old man with severe chronic obstructive pulmonary disease (COPD) on oxygen therapy and obstructive sleep apnea treated with nocturnal continuous positive airway pressure was seen in the pulmonary clinic for evaluation of his dyspnea. He was symptomatic with minimal activity and had chronic cough with some sputum production. He had been hospitalized 3 times over the past 12 months and had been to the emergency department (ED) the same number of times for dyspnea. Pertinent medications included as-needed albuterol inhaler, inhaled steroids, and tiotropium 18 mcg inhaled daily. He demonstrated good inhaler technique. On examination, his vital signs were pulse 99 beats/min, oxygen saturation 94% on 2 L/min of oxygen by nasal cannula, blood pressure 126/72 mm Hg, respiratory rate 15 breaths/min, and body mass index 35 kg/m2. He appeared chronically ill but in no acute distress. No wheezing or rales were heard. He had no lower extremity edema. The remainder of the exam was within normal limits. His last pulmonary function test demonstrated moderate obstruction with significant bronchodilator response to 2 puffs of albuterol. The side effects of chronic steroid therapy were impressed upon the patient and 500 mg of roflumilast was started daily. Over the course of the next 3 months, he had no further exacerbations. Roflumilast was continued. He has not required any further hospitalizations, ED visits, or oral steroid use since the last clinic visit.
What is the significance of acute exacerbations of COPD?
Acute exacerbation of COPD (AECOPD) is a frequently observed complication for many patients with COPD.1,2 AECOPD is associated with accelerated disease progression, augmented decline in health status and quality of life, and increased mortality.3 Exacerbations account for most of the costs associated with COPD. Estimates suggest that the aggregate costs associated with the treatment of AECOPD are between $3.2 and $3.8 billion, and that annual health care costs are 10-fold greater for patients with COPD associated with acute exacerbations than for patients with COPD but without exacerbations.4 Hence, any intervention that could potentially minimize or prevent this complication will have far-reaching benefits to patients with COPD as well as provide significant cost saving.
How is AECOPD defined?
COPD exacerbation is defined as a baseline change of the patient’s dyspnea, cough, and/or sputum that is acute in onset, and may warrant a change in regular medication in a patient with underlying COPD.5 Exacerbation in clinical trials has been defined on the basis of whether an increase in the level of care beyond regular care is required primarily in the hospital or ED.6 Frequent exacerbations are defined as 3 symptom-defined exacerbations per year or 2 per year if defined by the need for therapy with corticosteroids, antibiotics, or both.7
What is the underlying pathophysiology?
AECOPD is associated with enhanced upper and lower airway and systemic inflammation. The bronchial mucosa of stable COPD patients have increased numbers of CD8+ lymphocytes and macrophages. In mild AECOPD, eosinophils are increased in the bronchial mucosa and modest elevation of neutrophils, T lymphocytes (CD3), and TNF-α positive cells has also been reported.2 With more severe AECOPD, airway neutrophils are increased. Oxidative stress is a key factor in the development of airway inflammation in COPD.1 Patients with severe exacerbations have augmented large airway interleukin-8 (IL-8) levels and increased oxidative stress as demonstrated by markers such as hydrogen peroxide and 8-isoprostane.6
How do acute exacerbations affect the course of the disease?
In general, as the severity of the underlying COPD increases, exacerbations become both more severe and more frequent. Patients with frequent exacerbations have a worse quality of life than patients with a history of less frequent exacerbations.8 Frequent exacerbations have also been linked to a decline in lung function, with studies suggesting that there might be a decline of 7 mL in forced expiratory volume in 1 second (FEV1) per lower respiratory tract infection per year,9,10 and approximately 8 mL per year in patients with frequent exacerbations as compared to those with sporadic exacerbations.11
What are the triggers for COPD exacerbation?
Respiratory infections are estimated to trigger approximately two-thirds of exacerbations.2 Viral and bacterial infections cause most exacerbations. The effect of the infective triggers is to increase inflammation, cause bronchoconstriction, edema, and mucus production, with a resultant increase in dynamic hyperinflation.12 Thus, any intervention that reduces inflammation in COPD reduces the number and severity of exacerbations, whereas bronchodilators have an impact on exacerbation by their effects on reducing dynamic hyperinflation. The triggers for the one-third of exacerbations not triggered by infection are postulated to be related to other medical conditions, including pulmonary embolism, aspiration, heart failure, and myocardial ischemia.6
What are the pharmacologic options available for prevention of AECOPD?
In recognition of the importance of preventing COPD exacerbations, the American College of Chest Physicians and Canadian Thoracic Society5 have published an evidence-informed clinical guideline specifically examining the prevention of AECOPD, with the goal of assisting clinicians in providing optimal management for COPD patients. The following pharmacologic agents have been recognized as being effective at reducing the frequency of acute exacerbations without any impact on the severity of COPD itself.
Roflumilast
Phosphodiesterase 4 (PDE4) inhibition appears to have inflammatory-modulating properties in the airways, although the exact mechanism of action is unclear. Some have proposed that it reduces inflammation by inhibiting the breakdown of intracellular cyclic adenosine monophosphate.13 In 2 large clinical trials,14,15 daily use of a PDE4 inhibitor (roflumilast) showed a significant (15%–18%) reduction in yearly AECOPD incidence (approximate number needed to treat: 4). This benefit was seen in patients with GOLD stage 3–4 disease (FEV1 < 50% predicted) with the chronic bronchitic phenotype and who had experienced at least 1 exacerbation in the previous year.
Importantly, these clinical trials specifically prohibited the use of inhaled corticosteroids (ICS) and long-acting muscarinic antagonists (LAMAs). Thus, it remains unclear if PDE4 inhibition should be used as an add-on to ICS/LAMA therapy in patients who continue to have frequent AECOPD or whether PDE4 inhibition could be used instead of these standard therapies in patients with well-controlled daily symptoms without ICS or LAMA therapy but who experience frequent exacerbations.
Of note, earlier trials with roflumilast included patients with ICS and LAMA use.14,16 These trials were focused on FEV1 improvement and found no benefit. It was only in post ad hoc analyses that a reduction in AECOPD in patients with frequent exacerbations was found among those taking roflumilast, regardless of ICS or LAMA use.17 While roflumilast has documented benefit in improving lung function and reducing the rate of exacerbations, it has not been reported to decrease hospitalizations.4 This indicates that although the drug reduces the total number of exacerbations, it may not be as useful in preventing episodes of severe exacerbations of COPD.
Although PDE4 inhibitors are easy to administer (a once-daily pill), they are associated with significant gastrointestinal side effects (diarrhea, nausea, reduced appetite), weight loss, headache, and sleep disturbance.18 Adverse effects tend to occur early during treatment, are reversible, and lessen over time with treatment.6 Studies reported an average unexplained weight loss of 2 kg, and monitoring weight during treatment is advised. In addition, it is important to avoid roflumilast in underweight patients. Roflumilast should also be used with caution in depressed patients.5
N-acetylcysteine
N-acetylcysteine (NAC) reduces the viscosity of respiratory secretions as a result of the cleavage of the disulfide bonds and has been studied as a mucolytic agent to aid in the elimination of respiratory secretions.19 Oral NAC is quickly absorbed and is rapidly present in an active form in lung tissue and respiratory secretions after ingestion. NAC is well-tolerated except for occasional patients with GI adverse effects. The role of NAC in preventing AECOPD has been studied for more than 3 decades,20-22 although the largest clinical trial to date was reported in 2014.23 Taken together, the combined data demonstrate a significant reduction in the rate of COPD exacerbations associated with the use of NAC when compared with placebo (odds ratio [OR], 0.61; 95% confidence interval [CI], 0.37-0.99). Clinical guidelines suggest that in patients with moderate to severe COPD (FEV1/forced vital capacity ratio < 0.7, and FEV1 < 80% predicted) receiving maintenance bronchodilator therapy combined with ICS and history of 2 more exacerbations in the previous 2 years, treatment with oral NAC can be administered to prevent AECOPD.
Macrolides
Continuous prophylactic use of antibiotics in older studies had no effect on the frequency of AECOPD.24,25 But it is known that macrolide antibiotics have several antimicrobial, anti-inflammatory and immunomodulating effects and have been used for many years in the management of other chronic airway disease, including diffuse pan-bronchiolitis and cystic fibrosis.5 One recent study showed that the use of once-daily generic azithromycin 5 days per week appeared to have an impact on AECOPD incidence.26 In this study, the rate of AECOPD was reduced from 1.83 to 1.48 exacerbations per patient-year (relative risk, 0.83; 95% CI, 0.72–0.95; P = 0.01). Azithromycin also prevented severe AECOPD. Greater benefit was obtained with milder forms of the disease and in the elderly. Azithromycin did not appear to provide any benefit in those who continued to smoke (hazard ratio, 0.99).27 Other studies have shown that azithromycin was associated with an increased incidence of bacterial resistance and impaired hearing.28 Overall data from the available clinical trials are robust and demonstrate that regular macrolide therapy definitely reduces the risk of AECOPD. Due to potential adverse effects, however, macrolide therapy is an option rather than a strong recommendation.5 The prescribing clinician also needs to consider potential of prolongation of the QT interval.26
Immunostimulants
Immunostimulants have also been reported to reduce frequency of AECOPD.29,30 Bacterial lysates, reconstituted mixtures of bacterial antigens present in the lower airways of COPD patients, act as immunostimulants through the induction of cellular maturation, stimulating lymphocyte chemotaxis and increasing opsonization when administered to individuals with COPD.6 Studies have demonstrated a reduction in the severe complications of exacerbations and hospital admissions in COPD patients with OM-85, a detoxified oral immunoactive bacterial extract.29,30 However, most of these trials were conducted prior to the routine use of long-acting bronchodilators and ICS in COPD. A study that evaluated the efficacy of ismigen, a bacterial lysate, in reducing AECOPD31 found no difference in the exacerbation rate between ismigen and placebo or the time to first exacerbation. Additional studies are needed to examine the long-term effects of this therapy in patients receiving currently recommended COPD maintenance therapy.6
β-Blockers
Observational studies of β-blocker use in preventing AECOPD have yielded encouraging results, with one study showing a reduction in AECOPD risk (incidence risk ratio, 0.73; 95% CI, 0.60–0.90) in patients receiving β-blockers versus those not on β-blockers.32 Based on these findings, a clinical trial investigating the impact of metoprolol on risk of AECOPD is ongoing.33
Proton Pump Inhibitors
Gastroesophageal reflux disease is an independent risk factor for exacerbations.34 Two small, single-center studies,35,36 have shown that use of lansoprazole decreases the risk and frequency of AECOPD. However, data from the Predicting Outcome using Systemic Markers in Severe Exacerbations of COPD (PROMISE-COPD) study,6 which was a multicenter prospective observational study, suggested that patients with stable COPD receiving a proton pump inhibitor were at high risk of frequent and severe exacerbations.37 Thus, at this stage, their definitive role needs to be defined, possibly with a randomized, placebo-controlled study.
Case Presentation
A 70-year-old man with severe chronic obstructive pulmonary disease (COPD) on oxygen therapy and obstructive sleep apnea treated with nocturnal continuous positive airway pressure was seen in the pulmonary clinic for evaluation of his dyspnea. He was symptomatic with minimal activity and had chronic cough with some sputum production. He had been hospitalized 3 times over the past 12 months and had been to the emergency department (ED) the same number of times for dyspnea. Pertinent medications included as-needed albuterol inhaler, inhaled steroids, and tiotropium 18 mcg inhaled daily. He demonstrated good inhaler technique. On examination, his vital signs were pulse 99 beats/min, oxygen saturation 94% on 2 L/min of oxygen by nasal cannula, blood pressure 126/72 mm Hg, respiratory rate 15 breaths/min, and body mass index 35 kg/m2. He appeared chronically ill but in no acute distress. No wheezing or rales were heard. He had no lower extremity edema. The remainder of the exam was within normal limits. His last pulmonary function test demonstrated moderate obstruction with significant bronchodilator response to 2 puffs of albuterol. The side effects of chronic steroid therapy were impressed upon the patient and 500 mg of roflumilast was started daily. Over the course of the next 3 months, he had no further exacerbations. Roflumilast was continued. He has not required any further hospitalizations, ED visits, or oral steroid use since the last clinic visit.
What is the significance of acute exacerbations of COPD?
Acute exacerbation of COPD (AECOPD) is a frequently observed complication for many patients with COPD.1,2 AECOPD is associated with accelerated disease progression, augmented decline in health status and quality of life, and increased mortality.3 Exacerbations account for most of the costs associated with COPD. Estimates suggest that the aggregate costs associated with the treatment of AECOPD are between $3.2 and $3.8 billion, and that annual health care costs are 10-fold greater for patients with COPD associated with acute exacerbations than for patients with COPD but without exacerbations.4 Hence, any intervention that could potentially minimize or prevent this complication will have far-reaching benefits to patients with COPD as well as provide significant cost saving.
How is AECOPD defined?
COPD exacerbation is defined as a baseline change of the patient’s dyspnea, cough, and/or sputum that is acute in onset, and may warrant a change in regular medication in a patient with underlying COPD.5 Exacerbation in clinical trials has been defined on the basis of whether an increase in the level of care beyond regular care is required primarily in the hospital or ED.6 Frequent exacerbations are defined as 3 symptom-defined exacerbations per year or 2 per year if defined by the need for therapy with corticosteroids, antibiotics, or both.7
What is the underlying pathophysiology?
AECOPD is associated with enhanced upper and lower airway and systemic inflammation. The bronchial mucosa of stable COPD patients have increased numbers of CD8+ lymphocytes and macrophages. In mild AECOPD, eosinophils are increased in the bronchial mucosa and modest elevation of neutrophils, T lymphocytes (CD3), and TNF-α positive cells has also been reported.2 With more severe AECOPD, airway neutrophils are increased. Oxidative stress is a key factor in the development of airway inflammation in COPD.1 Patients with severe exacerbations have augmented large airway interleukin-8 (IL-8) levels and increased oxidative stress as demonstrated by markers such as hydrogen peroxide and 8-isoprostane.6
How do acute exacerbations affect the course of the disease?
In general, as the severity of the underlying COPD increases, exacerbations become both more severe and more frequent. Patients with frequent exacerbations have a worse quality of life than patients with a history of less frequent exacerbations.8 Frequent exacerbations have also been linked to a decline in lung function, with studies suggesting that there might be a decline of 7 mL in forced expiratory volume in 1 second (FEV1) per lower respiratory tract infection per year,9,10 and approximately 8 mL per year in patients with frequent exacerbations as compared to those with sporadic exacerbations.11
What are the triggers for COPD exacerbation?
Respiratory infections are estimated to trigger approximately two-thirds of exacerbations.2 Viral and bacterial infections cause most exacerbations. The effect of the infective triggers is to increase inflammation, cause bronchoconstriction, edema, and mucus production, with a resultant increase in dynamic hyperinflation.12 Thus, any intervention that reduces inflammation in COPD reduces the number and severity of exacerbations, whereas bronchodilators have an impact on exacerbation by their effects on reducing dynamic hyperinflation. The triggers for the one-third of exacerbations not triggered by infection are postulated to be related to other medical conditions, including pulmonary embolism, aspiration, heart failure, and myocardial ischemia.6
What are the pharmacologic options available for prevention of AECOPD?
In recognition of the importance of preventing COPD exacerbations, the American College of Chest Physicians and Canadian Thoracic Society5 have published an evidence-informed clinical guideline specifically examining the prevention of AECOPD, with the goal of assisting clinicians in providing optimal management for COPD patients. The following pharmacologic agents have been recognized as being effective at reducing the frequency of acute exacerbations without any impact on the severity of COPD itself.
Roflumilast
Phosphodiesterase 4 (PDE4) inhibition appears to have inflammatory-modulating properties in the airways, although the exact mechanism of action is unclear. Some have proposed that it reduces inflammation by inhibiting the breakdown of intracellular cyclic adenosine monophosphate.13 In 2 large clinical trials,14,15 daily use of a PDE4 inhibitor (roflumilast) showed a significant (15%–18%) reduction in yearly AECOPD incidence (approximate number needed to treat: 4). This benefit was seen in patients with GOLD stage 3–4 disease (FEV1 < 50% predicted) with the chronic bronchitic phenotype and who had experienced at least 1 exacerbation in the previous year.
Importantly, these clinical trials specifically prohibited the use of inhaled corticosteroids (ICS) and long-acting muscarinic antagonists (LAMAs). Thus, it remains unclear if PDE4 inhibition should be used as an add-on to ICS/LAMA therapy in patients who continue to have frequent AECOPD or whether PDE4 inhibition could be used instead of these standard therapies in patients with well-controlled daily symptoms without ICS or LAMA therapy but who experience frequent exacerbations.
Of note, earlier trials with roflumilast included patients with ICS and LAMA use.14,16 These trials were focused on FEV1 improvement and found no benefit. It was only in post ad hoc analyses that a reduction in AECOPD in patients with frequent exacerbations was found among those taking roflumilast, regardless of ICS or LAMA use.17 While roflumilast has documented benefit in improving lung function and reducing the rate of exacerbations, it has not been reported to decrease hospitalizations.4 This indicates that although the drug reduces the total number of exacerbations, it may not be as useful in preventing episodes of severe exacerbations of COPD.
Although PDE4 inhibitors are easy to administer (a once-daily pill), they are associated with significant gastrointestinal side effects (diarrhea, nausea, reduced appetite), weight loss, headache, and sleep disturbance.18 Adverse effects tend to occur early during treatment, are reversible, and lessen over time with treatment.6 Studies reported an average unexplained weight loss of 2 kg, and monitoring weight during treatment is advised. In addition, it is important to avoid roflumilast in underweight patients. Roflumilast should also be used with caution in depressed patients.5
N-acetylcysteine
N-acetylcysteine (NAC) reduces the viscosity of respiratory secretions as a result of the cleavage of the disulfide bonds and has been studied as a mucolytic agent to aid in the elimination of respiratory secretions.19 Oral NAC is quickly absorbed and is rapidly present in an active form in lung tissue and respiratory secretions after ingestion. NAC is well-tolerated except for occasional patients with GI adverse effects. The role of NAC in preventing AECOPD has been studied for more than 3 decades,20-22 although the largest clinical trial to date was reported in 2014.23 Taken together, the combined data demonstrate a significant reduction in the rate of COPD exacerbations associated with the use of NAC when compared with placebo (odds ratio [OR], 0.61; 95% confidence interval [CI], 0.37-0.99). Clinical guidelines suggest that in patients with moderate to severe COPD (FEV1/forced vital capacity ratio < 0.7, and FEV1 < 80% predicted) receiving maintenance bronchodilator therapy combined with ICS and history of 2 more exacerbations in the previous 2 years, treatment with oral NAC can be administered to prevent AECOPD.
Macrolides
Continuous prophylactic use of antibiotics in older studies had no effect on the frequency of AECOPD.24,25 But it is known that macrolide antibiotics have several antimicrobial, anti-inflammatory and immunomodulating effects and have been used for many years in the management of other chronic airway disease, including diffuse pan-bronchiolitis and cystic fibrosis.5 One recent study showed that the use of once-daily generic azithromycin 5 days per week appeared to have an impact on AECOPD incidence.26 In this study, the rate of AECOPD was reduced from 1.83 to 1.48 exacerbations per patient-year (relative risk, 0.83; 95% CI, 0.72–0.95; P = 0.01). Azithromycin also prevented severe AECOPD. Greater benefit was obtained with milder forms of the disease and in the elderly. Azithromycin did not appear to provide any benefit in those who continued to smoke (hazard ratio, 0.99).27 Other studies have shown that azithromycin was associated with an increased incidence of bacterial resistance and impaired hearing.28 Overall data from the available clinical trials are robust and demonstrate that regular macrolide therapy definitely reduces the risk of AECOPD. Due to potential adverse effects, however, macrolide therapy is an option rather than a strong recommendation.5 The prescribing clinician also needs to consider potential of prolongation of the QT interval.26
Immunostimulants
Immunostimulants have also been reported to reduce frequency of AECOPD.29,30 Bacterial lysates, reconstituted mixtures of bacterial antigens present in the lower airways of COPD patients, act as immunostimulants through the induction of cellular maturation, stimulating lymphocyte chemotaxis and increasing opsonization when administered to individuals with COPD.6 Studies have demonstrated a reduction in the severe complications of exacerbations and hospital admissions in COPD patients with OM-85, a detoxified oral immunoactive bacterial extract.29,30 However, most of these trials were conducted prior to the routine use of long-acting bronchodilators and ICS in COPD. A study that evaluated the efficacy of ismigen, a bacterial lysate, in reducing AECOPD31 found no difference in the exacerbation rate between ismigen and placebo or the time to first exacerbation. Additional studies are needed to examine the long-term effects of this therapy in patients receiving currently recommended COPD maintenance therapy.6
β-Blockers
Observational studies of β-blocker use in preventing AECOPD have yielded encouraging results, with one study showing a reduction in AECOPD risk (incidence risk ratio, 0.73; 95% CI, 0.60–0.90) in patients receiving β-blockers versus those not on β-blockers.32 Based on these findings, a clinical trial investigating the impact of metoprolol on risk of AECOPD is ongoing.33
Proton Pump Inhibitors
Gastroesophageal reflux disease is an independent risk factor for exacerbations.34 Two small, single-center studies,35,36 have shown that use of lansoprazole decreases the risk and frequency of AECOPD. However, data from the Predicting Outcome using Systemic Markers in Severe Exacerbations of COPD (PROMISE-COPD) study,6 which was a multicenter prospective observational study, suggested that patients with stable COPD receiving a proton pump inhibitor were at high risk of frequent and severe exacerbations.37 Thus, at this stage, their definitive role needs to be defined, possibly with a randomized, placebo-controlled study.
1. Wedzicha JA, Singh R, Mackay AJ. Acute COPD exacerbations. Clin Chest Med. 2014;35:157-163.
2. Wedzicha JA, Seemungal TAR. COPD exacerbations: defining their cause and prevention. Lancet. 2007;370:786-796.
3. Spencer S, Calverley PMA, Burge PS, Jones PW. Impact of preventing exacerbations on deterioration of health status in COPD. Eur Respir J. 2004;23:698-702.
4. Blanchette CM, Gross NJ, Altman P. Rising costs of COPD and the potential for maintenance therapy to slow the trend. Am Health Drug Benef. 2014;7:98.
5. Criner GJ, Bourbeau J, Diekemper RL, et al. Prevention of acute exacerbations of COPD: American College of Chest Physicians and Canadian Thoracic Society Guideline. Chest. 2015;147:894-942.
6. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management and prevention of chronic obstructive lung disease 2017 report. Respirology. 2017;22:575-601.
7. Wedzicha JA, Brill SE, Allinson JP, Donaldson GC. Mechanisms and impact of the frequent exacerbator phenotype in chronic obstructive pulmonary disease. BMC Med. 2013;11:181.
8. Seemungal TAR, Donaldson GC, Paul EA, et al. Effect of exacerbation on quality of life in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;157:1418-1422.
9. Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Ann Intern Med. 1995;122:823-832.
10. Kanner RE, Anthonisen NR, Connett JE. Lower respiratory illnesses promote FEV1 decline in current smokers but not ex-smokers with mild chronic obstructive pulmonary disease: results from the lung health study. Am J Respir Crit Care Med. 2001;164:358-364.
11. Donaldson GC, Seemungal TAR, Bhowmik A, Wedzicha JA. Relationship between exacerbation frequency and lung function decline in chronic obstructive pulmonary disease. Thorax. 2002;57:847-852.
12. Papi A, Bellettato CM, Braccioni F, et al. Infections and airway inflammation in chronic obstructive pulmonary disease severe exacerbations. Am J Respir Crit Care Med. 2006;173:1114-1121.
13. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163:53-67.
14. Calverley PMA, Rabe KF, Goehring U-M, et al. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. Lancet. 2009;374:685-694.
15. Fabbri LM, Calverley PMA, Izquierdo-Alonso JL, et al. Roflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with long-acting bronchodilators: two randomised clinical trials. Lancet. 2009;374:695-703.
16. Lee S, Hui DSC, Mahayiddin AA, et al. Roflumilast in Asian patients with COPD: a randomized placebo-controlled trial. Respirology. 2011;16:1249-1257.
17. Calverley PM, Martinez FJ, Fabbri LM, et al. Does roflumilast decrease exacerbations in severe COPD patients not controlled by inhaled combination therapy? The REACT study protocol. Int J Chron Obstruct Pulmon Dis. 2012;7:375-382.
18. Chong J, Leung B, Poole P. Phosphodiesterase 4 inhibitors for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2013(11):CD002309.
19. Sheffner AL, Medler EM, Jacobs LW, Sarett HP. The in vitro reduction in viscosity of human tracheobronchial secretions by acetylcysteine. Am Rev Respir Dis. 1964;90:721-729.
20. Boman G, Bäcker U, Larsson S, et al. Oral acetylcysteine reduces exacerbation rate in chronic bronchitis: report of a trial organized by the Swedish Society for Pulmonary Diseases. Eur J Respir Dis. 1983;64:405-415.
21. Grassi C, Morandini GC. A controlled trial of intermittent oral acetylcysteine in the long-term treatment of chronic bronchitis. Eur J Clin Pharmacol. 1976;9:393-396.
22. Hansen NCG, Skriver A, Brorsen-Riis L, et al. Orally administered N-acetylcysteine may improve general well-being in patients with mild chronic bronchitis. Respir Med. 1994;88:531-535.
23. Zheng JP, Wen FQ, Bai CX, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir Med. 2014;2:187-194.
24. Francis RS, Spicer CC. Chemotherapy in chronic bronchitis: Influence of daily penicillin and tetracycline on exacerbations and their cost: A report to the research committee of the British Tuberculosis Association by Their Chronic Bronchitis Subcommittee. BMJ. 1960;1:297-303.
25. Francis RS, May JR, Spicer CC. Chemotherapy of bronchitis. BMJ. 1961;2:979.
26. Albert RK, Connett J, Bailey WC, et al. Azithromycin for prevention of exacerbations of COPD. N Engl J Med. 2011;365:689-698.
27. Han MK, Tayob N, Murray S, et al. Predictors of chronic obstructive pulmonary disease exacerbation reduction in response to daily azithromycin therapy. Am J Respir Crit Care Med. 2014;189:1503-1508.
28. Uzun S, Djamin RS, Kluytmans JAJW, et al. Azithromycin maintenance treatment in patients with frequent exacerbations of chronic obstructive pulmonary disease (COLUMBUS): a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2014;2:361-368.
29. Collet JP, Shapiro S, Ernst P, et al. Effects of an immunostimulating agent on acute exacerbations and hospitalizations in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1997;156:1719-1724.
30. Jing LI. Protective effect of a bacterial extract against acute exacerbation in patients with chronic bronchitis accompanied by chronic obstructive pulmonary. Age. 2004;67:828-834.
31. Braido F, Tarantini F, Ghiglione V, et al. Bacterial lysate in the prevention of acute exacerbation of COPD and in respiratory recurrent infections. Int J Chron Obstruct Pulmon Dis. 2007;2:335.
32. Bhatt SP, Wells JM, Kinney GL, et al. β-Blockers are associated with a reduction in COPD exacerbations. Thorax. 2016;71:8-14.
33. Bhatt SP, Connett JE, Voelker H, et al. β-Blockers for the prevention of acute exacerbations of chronic obstructive pulmonary disease (βLOCK COPD): a randomised controlled study protocol. BMJ Open. 2016;6:e012292.
34. Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363:1128-1138.
35. Sasaki T, Nakayama K, Yasuda H, et al. A randomized, single-blind study of lansoprazole for the prevention of exacerbations of chronic obstructive pulmonary disease in older patients. J Am Geriatr Soc. 2009;57:1453-1457.
36. Xiong W, Zhang Qs, Zhao W, et al. A 12-month follow-up study on the preventive effect of oral lansoprazole on acute exacerbation of chronic obstructive pulmonary disease. Int J Exper Pathol. 2016;97:107-113.
37. Baumeler L, Papakonstantinou E, Milenkovic B, et al. Therapy with proton-pump inhibitors for gastroesophageal reflux disease does not reduce the risk for severe exacerbations in COPD. Respirology. 2016;21:883-890.
1. Wedzicha JA, Singh R, Mackay AJ. Acute COPD exacerbations. Clin Chest Med. 2014;35:157-163.
2. Wedzicha JA, Seemungal TAR. COPD exacerbations: defining their cause and prevention. Lancet. 2007;370:786-796.
3. Spencer S, Calverley PMA, Burge PS, Jones PW. Impact of preventing exacerbations on deterioration of health status in COPD. Eur Respir J. 2004;23:698-702.
4. Blanchette CM, Gross NJ, Altman P. Rising costs of COPD and the potential for maintenance therapy to slow the trend. Am Health Drug Benef. 2014;7:98.
5. Criner GJ, Bourbeau J, Diekemper RL, et al. Prevention of acute exacerbations of COPD: American College of Chest Physicians and Canadian Thoracic Society Guideline. Chest. 2015;147:894-942.
6. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management and prevention of chronic obstructive lung disease 2017 report. Respirology. 2017;22:575-601.
7. Wedzicha JA, Brill SE, Allinson JP, Donaldson GC. Mechanisms and impact of the frequent exacerbator phenotype in chronic obstructive pulmonary disease. BMC Med. 2013;11:181.
8. Seemungal TAR, Donaldson GC, Paul EA, et al. Effect of exacerbation on quality of life in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;157:1418-1422.
9. Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Ann Intern Med. 1995;122:823-832.
10. Kanner RE, Anthonisen NR, Connett JE. Lower respiratory illnesses promote FEV1 decline in current smokers but not ex-smokers with mild chronic obstructive pulmonary disease: results from the lung health study. Am J Respir Crit Care Med. 2001;164:358-364.
11. Donaldson GC, Seemungal TAR, Bhowmik A, Wedzicha JA. Relationship between exacerbation frequency and lung function decline in chronic obstructive pulmonary disease. Thorax. 2002;57:847-852.
12. Papi A, Bellettato CM, Braccioni F, et al. Infections and airway inflammation in chronic obstructive pulmonary disease severe exacerbations. Am J Respir Crit Care Med. 2006;173:1114-1121.
13. Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163:53-67.
14. Calverley PMA, Rabe KF, Goehring U-M, et al. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. Lancet. 2009;374:685-694.
15. Fabbri LM, Calverley PMA, Izquierdo-Alonso JL, et al. Roflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with long-acting bronchodilators: two randomised clinical trials. Lancet. 2009;374:695-703.
16. Lee S, Hui DSC, Mahayiddin AA, et al. Roflumilast in Asian patients with COPD: a randomized placebo-controlled trial. Respirology. 2011;16:1249-1257.
17. Calverley PM, Martinez FJ, Fabbri LM, et al. Does roflumilast decrease exacerbations in severe COPD patients not controlled by inhaled combination therapy? The REACT study protocol. Int J Chron Obstruct Pulmon Dis. 2012;7:375-382.
18. Chong J, Leung B, Poole P. Phosphodiesterase 4 inhibitors for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2013(11):CD002309.
19. Sheffner AL, Medler EM, Jacobs LW, Sarett HP. The in vitro reduction in viscosity of human tracheobronchial secretions by acetylcysteine. Am Rev Respir Dis. 1964;90:721-729.
20. Boman G, Bäcker U, Larsson S, et al. Oral acetylcysteine reduces exacerbation rate in chronic bronchitis: report of a trial organized by the Swedish Society for Pulmonary Diseases. Eur J Respir Dis. 1983;64:405-415.
21. Grassi C, Morandini GC. A controlled trial of intermittent oral acetylcysteine in the long-term treatment of chronic bronchitis. Eur J Clin Pharmacol. 1976;9:393-396.
22. Hansen NCG, Skriver A, Brorsen-Riis L, et al. Orally administered N-acetylcysteine may improve general well-being in patients with mild chronic bronchitis. Respir Med. 1994;88:531-535.
23. Zheng JP, Wen FQ, Bai CX, et al. Twice daily N-acetylcysteine 600 mg for exacerbations of chronic obstructive pulmonary disease (PANTHEON): a randomised, double-blind placebo-controlled trial. Lancet Respir Med. 2014;2:187-194.
24. Francis RS, Spicer CC. Chemotherapy in chronic bronchitis: Influence of daily penicillin and tetracycline on exacerbations and their cost: A report to the research committee of the British Tuberculosis Association by Their Chronic Bronchitis Subcommittee. BMJ. 1960;1:297-303.
25. Francis RS, May JR, Spicer CC. Chemotherapy of bronchitis. BMJ. 1961;2:979.
26. Albert RK, Connett J, Bailey WC, et al. Azithromycin for prevention of exacerbations of COPD. N Engl J Med. 2011;365:689-698.
27. Han MK, Tayob N, Murray S, et al. Predictors of chronic obstructive pulmonary disease exacerbation reduction in response to daily azithromycin therapy. Am J Respir Crit Care Med. 2014;189:1503-1508.
28. Uzun S, Djamin RS, Kluytmans JAJW, et al. Azithromycin maintenance treatment in patients with frequent exacerbations of chronic obstructive pulmonary disease (COLUMBUS): a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2014;2:361-368.
29. Collet JP, Shapiro S, Ernst P, et al. Effects of an immunostimulating agent on acute exacerbations and hospitalizations in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1997;156:1719-1724.
30. Jing LI. Protective effect of a bacterial extract against acute exacerbation in patients with chronic bronchitis accompanied by chronic obstructive pulmonary. Age. 2004;67:828-834.
31. Braido F, Tarantini F, Ghiglione V, et al. Bacterial lysate in the prevention of acute exacerbation of COPD and in respiratory recurrent infections. Int J Chron Obstruct Pulmon Dis. 2007;2:335.
32. Bhatt SP, Wells JM, Kinney GL, et al. β-Blockers are associated with a reduction in COPD exacerbations. Thorax. 2016;71:8-14.
33. Bhatt SP, Connett JE, Voelker H, et al. β-Blockers for the prevention of acute exacerbations of chronic obstructive pulmonary disease (βLOCK COPD): a randomised controlled study protocol. BMJ Open. 2016;6:e012292.
34. Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363:1128-1138.
35. Sasaki T, Nakayama K, Yasuda H, et al. A randomized, single-blind study of lansoprazole for the prevention of exacerbations of chronic obstructive pulmonary disease in older patients. J Am Geriatr Soc. 2009;57:1453-1457.
36. Xiong W, Zhang Qs, Zhao W, et al. A 12-month follow-up study on the preventive effect of oral lansoprazole on acute exacerbation of chronic obstructive pulmonary disease. Int J Exper Pathol. 2016;97:107-113.
37. Baumeler L, Papakonstantinou E, Milenkovic B, et al. Therapy with proton-pump inhibitors for gastroesophageal reflux disease does not reduce the risk for severe exacerbations in COPD. Respirology. 2016;21:883-890.
Stable COPD: Managing Advanced Disease
Case Presentation
A 65-year-old man with severe chronic obstructive disease (COPD; forced expiratory volume in 1 second/forced vital capacity ratio [FEV1/FVC], 27; FEV1 25% of predicted; residual volume 170% of predicted for his age and height) was seen in the pulmonary clinic. His medications include a long-acting beta agonist (LABA)/long-acting muscarinic antagonist (LAMA) combination that he uses twice daily as advised. He uses his rescue albuterol inhaler roughly once a week. The patient complains of severe disabling shortness of breath with exertion and severe limitation of his quality of life because of his inability to lead a normal active life. He is on 2 L/min of oxygen at all times. He has received pulmonary rehabilitation in hopes of improving his quality of life but can only climb a flight of stairs before he must stop to rest. He asks about options but does not want to consider lung transplantation today. His most recent chest computed tomography (CT) scan demonstrates upper lobe predominant emphysematous changes with no masses or nodules.
What are the patient's options at this time?
Lung volume reduction surgery (LVRS) attempts to reduce space-occupying severely diseased, hyperexpanded lung, thus allowing the relatively normal adjoining lung parenchyma to expand into the vacated space and function effectively.1 Hence, such therapies are suitable for patients with emphysematous lungs and not those with bronchitic-predominant COPD. LVRS offers a greater chance of improvement in exercise capacity, lung function, quality of life, and dyspnea in the correctly chosen patient population, as compared with pharmacologic management alone.2 However, the procedure is associated with risks, including higher short-term morbidity and mortality.2 Patients with predominantly upper-lobe emphysema and a low maximal workload after rehabilitation were noted to have lower mortality, a greater probability of improvement in exercise capacity, and a greater probability of improvement in symptoms if they underwent surgery compared to medical therapy alone.2 On the contrary, patients with predominantly non–upper-lobe emphysema and a high maximal workload after rehabilitation had higher mortality if they underwent surgery compared to receiving medical therapy alone.2 Thus, a subgroup of patients with homogeneous emphysema symmetrically affecting the upper and lower lobes are considered to be unlikely to benefit from this surgery.2,3
Valves and other methods of lung volume reduction such as coils, sealants, intrapulmonary vents, and thermal vapor in the bronchi or subsegmental airways have emerged as new techniques for nonsurgical lung volume reduction.4-9 Endobronchial-valve therapy is associated with improvement in lung function and with clinical benefits that are greatest in the presence of heterogeneous lung involvement. This works by the same principle as LVRS, by reduction of the most severely diseased lung units, expansion of the more viable, less emphysematous lung results in substantial improvements in lung mechanics.10,11 The most important complications of this procedure include pneumonia, pneumothorax, hemoptysis, and increased frequency of COPD exacerbation in the following 30 days. The fact that a high-heterogeneity subgroup had greater improvements in both the FEV1 and distance on the 6-minute walk test than did patients with lower heterogeneity supports the use of quantitative high-resolution computed tomography (HRCT) in selecting patients for endobronchial-valve therapy.12 The HRCT scans also help in identifying those with complete fissures, a marker of lack of collateral ventilation (CV+) between different lobes. Presence of CV+ state predicts failure of endobronchial valve and all forms of endoscopic LVRS.13 Bronchoscopic thermal vapor ablation (BTVA) therapy can potentially work on a subsegmental level and be successful for treatment of emphysema with lack of intact fissures on CT scans. Other methods that have the potential to be effective in those with collateral ventilation would be endoscopic coil therapy and polymeric lung volume reduction.11,14 Unfortunately, there are no randomized controlled trial data demonstrating clinically meaningful improvement following coil therapy or polymeric lung volume reduction in this CV+ patient population. Vapor therapy is perhaps the only technique that has been found to be effective in upper lobe predominant emphysema even with CV+ status.13
Our patient has evidence of air trapping and emphysema based on a high residual volume. A CT scan of the chest can determine the nature of the emphysema (heterogeneous versus homogenous) and based on these findings, further determination of the best strategy for lung volume reduction can be made.
Is there a role for long-term oxygen therapy?
Long-term oxygen therapy (LTOT) used for more than 15 hours a day is thought to reduce mortality among patients with COPD and severe resting hypoxemia.15-18 More recent studies have failed to show similar beneficial effects of LTOT. A recent study examined the effects of LTOT in randomized fashion and determined that supplemental oxygen for patients with stable COPD and resting or exercise-induced moderate desaturation did not affect the time to death or first hospitalization, time to first COPD exacerbation, time to first hospitalization for a COPD exacerbation, the rate of all hospitalizations, the rate of all COPD exacerbations, or changes in measures of quality of life, depression, anxiety, or functional status.19
Our patient is currently on long-term oxygen therapy and in spite of some uncertainty as to its benefit, it is prudent to order oxygen therapy until further clarification is available.
What is the role of pulmonary rehabilitation?
Pulmonary rehabilitation is an established treatment for patients with chronic lung disease.20 Benefits include improvement in exercise tolerance, symptoms, and quality of life, with a reduction in the use of health care resources.21 A Spanish population-based cohort study that looked at the influence of regular physical activity on COPD showed that patients who reported low, moderate, or high physical activity had a lower risk of COPD admissions and all-cause mortality than patients with very low physical activity after adjusting for all confounders.22
As previously mentioned, patients in GOLD categories B, C, and D should be offered pulmonary rehabilitation as part of their treatment.23 The ideal patient is one who is not too sick to undergo rehabilitation and is motivated to improve his or her quality of life.
What is the current scope of lung transplantation in the management of severe COPD?
There is an indisputable role for lung transplantation in end-stage COPD. However, lung transplantation does not benefit all COPD patients. There is a subset of patients for whom the treatment provides a survival benefit. It has been reported that 79% of patients with an FEV1 < 16% predicted will survive at least 1 additional year after transplant, but only 11% of patients with an FEV1 > 25% will do so.24 The pre-transplant BODE (body mass index, airflow obstruction/FEV1, dyspnea, and exercise capacity) index score is used to identify patients who will benefit from lung transplantation.25,26 International guidelines for the selection of lung transplant candidates identify the following patient characteristics:27
- The disease is progressive, despite maximal treatment including medication, pulmonary rehabilitation, and oxygen therapy;
- The patient is not a candidate for endoscopic or surgical LVRS;
- BODE index is 5 to 6;
- The PCO2 is greater than 50 mm Hg (6.6 kPa) and/or PO2 is less than 60 mm Hg (8 kPa);
- FEV1 is 25% predicted.
The perioperative mortality of lung transplantation surgery has been reduced to less than 10%. Risk of complications from surgery in the perioperative period, such as bronchial dehiscence, infectious complications, and acute rejection, have also been reduced but do occur. Chronic allograft dysfunction and the risk of lung cancer in cases of single lung transplant should be discussed with the patient before surgery.28
How can we incorporate palliative care into the management plan for patients with COPD?
Among patients with end-stage COPD on home oxygen therapy who have required mechanical ventilation for an exacerbation, only 55% are alive at 1 year.29 COPD patients at high risk of death within the next year of life as well as patients with refractory symptoms and unmet needs are candidates for early palliative care. Palliative care and palliative care specialists can aid in reducing symptom burden and improving quality of life among these patients and their family members, and palliative care is recommended by multiple international societies for patients with advanced COPD.30,31 In spite of these recommendations, the utilization of palliative care resources has been dismally low.32,33 Improving physician-patient communication regarding palliative services and patients’ unmet care needs will help ensure that COPD patients receive adequate palliative care services at the appropriate time.
Conclusion
COPD is a leading cause of morbidity and mortality in the United States and represents a significant economic burden for both individuals and society. The goals in COPD management are to provide symptom relief, improve the quality of life, preserve lung function, and reduce the frequency of exacerbations and mortality. COPD management is guided by disease severity that is measured using the GOLD multimodal staging system and requires a multidisciplinary approach. Several classes of medication are available for treatment, and a step-wise approach should be applied in building an effective pharmacologic regimen. In addition to pharmacologic therapies, nonpharmacologic therapies, including smoking cessation, vaccinations, proper nutrition, and maintaining physical activity, are an important part of long-term management. Those who continue to be symptomatic despite appropriate maximal therapy may be candidates for lung volume reduction. Palliative care services for COPD patients, which can aid in reducing symptom burden and improving quality of life, should not be overlooked.
1. Sabanathan A, Sabanathan S, Shah R, Richardson J. Lung volume reduction surgery for emphysema: a review. J Cardiovasc Surg. 1998;39:237.
2. Group NETTR. Patients at high risk of death after lung-volume–reduction surgery. N Engl J Med. 2001;345:1075-1083.
3. Group NETTR. A randomized trial comparing lung-volume–reduction surgery with medical therapy for severe emphysema. N Engl J Med. 2003;348:2059-2073.
4. Decker MR, Leverson GE, Jaoude WA, Maloney JD. Lung volume reduction surgery since the National Emphysema Treatment Trial: study of Society of Thoracic Surgeons database. J Thorac Cardiovasc Surg. 2014;148:2651-2658.
5. Deslée G, Mal H, Dutau H, et al. Lung volume reduction coil treatment vs usual care in patients with severe emphysema: the REVOLENS randomized clinical trial. JAMA. 2016;315:175-184.
6. Hartman JE, Klooster K, Gortzak K, et al. Long-term follow-up after bronchoscopic lung volume reduction treatment with coils in patients with severe emphysema. Respirology. 2015;20:319-326.
7. Snell GI, Hopkins P, Westall G, et al. A feasibility and safety study of bronchoscopic thermal vapor ablation: a novel emphysema therapy. Ann Thorac Surg. 2009;88:1993-1998.
8. Ingenito EP, Berger RL, Henderson AC, et al. Bronchoscopic lung volume reduction using tissue engineering principles. Am J Respir Crit Care Med. 2003;167:771-778.
9. Ingenito EP, Loring SH, Moy ML, et al. Comparison of physiological and radiological screening for lung volume reduction surgery. Am J Respir Crit Care Med. 2001;163:1068-1073.
10. Shah P, Slebos D, Cardoso P, et al. Bronchoscopic lung-volume reduction with Exhale airway stents for emphysema (EASE trial): randomised, sham-controlled, multicentre trial. Lancet. 2011;378:997-1005.
11. Sciurba FC, Ernst A, Herth FJ, et al. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med. 2010;363:1233-1244.
12. Wan IY, Toma TP, Geddes DM, et al. Bronchoscopic lung volume reduction for end-stage emphysema: report on the first 98 patients. Chest. 2006;129:518-526.
13. Gompelmann D, Eberhardt R, Schuhmann M, et al. Lung volume reduction with vapor ablation in the presence of incomplete fissures: 12-month results from the STEP-UP randomized controlled study. Respiration. 2016;92:397-403.
14. Come CE, Kramer MR, Dransfield MT, et al. A randomised trial of lung sealant versus medical therapy for advanced emphysema. Eur Respir J. 2015;46:651-662.
15. Group NOTT. Continuous or nocturnal oxygen therapy in hypoxemic chronic obstructive lung disease: a clinical trial. Ann Intern Med. 1980;93:391-398.
16. Council M. Long term domiciliary oxygen therapy in chronic hypoxic cor pulmonale complicating chronic bronchitis and emphysema: Report of the Medical Research Council Working Party. Lancet. 1981;1:681-686.
17. Qaseem A, Wilt TJ, Weinberger SE, et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155:179-191.
18. Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187:347-365.
19. Group L-TOTTR. A randomized trial of long-term oxygen for COPD with moderate desaturation. N Engl J Med. 2016;375:1617-1627.
20. McCarthy B, Casey D, Devane D, et al. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015(2):CD003793.
21. Griffiths TL, Burr ML, Campbell IA, et al. Results at 1 year of outpatient multidisciplinary pulmonary rehabilitation: a randomised controlled trial. Lancet. 2000;355:362-368.
22. Garcia-Aymerich J, Lange P, Benet M, et al. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population based cohort study. Thorax. 2006;61:772-778.
23. Global Initiative for Chronic Obstructive Lung Disease (GOLD): Global strategy for the diagnosis, management, and prevention of COPD 2017. www.goldcopd.org. Accessed July 9, 2019.
24. Thabut G, Ravaud P, Christie JD, et al. Determinants of the survival benefit of lung transplantation in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2008;177:1156-1163.
25. Lahzami S, Bridevaux PO, Soccal PM, et al. Survival impact of lung transplantation for COPD. Eur Respir J. 2010;36:74-80.
26. Cerón Navarro J, de Aguiar Quevedo K, Ansótegui Barrera E, et al. Functional outcomes after lung transplant in chronic obstructive pulmonary disease. Arch Bronconeumol. 2015;51:109-114.
27. Weill D, Benden C, Corris PA, et al. A consensus document for the selection of lung transplant candidates: 2014--an update from the Pulmonary Transplantation Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2015;34:1-15.
28. Minai OA, Shah S, Mazzone P, et al. Bronchogenic carcinoma after lung transplantation: characteristics and outcomes. J Thorac Oncol. 2008;3:1404-1409.
29. Hajizadeh N, Goldfeld K, Crothers K. What happens to patients with COPD with long-term oxygen treatment who receive mechanical ventilation for COPD exacerbation? A 1-year retrospective follow- up study. Thorax. 2015;70:294-296.
30. Siouta N, van Beek K, Preston N, et al. Towards integration of palliative care in patients with chronic heart failure and chronic obstructive pulmonary disease: a systematic literature review of European guidelines and pathways. BMC Palliat Care. 2016;15:18.
31. Celli BR, MacNee W; ATS/ERS Task Force. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur Respir J. 2004;23:932-946.
32. Szekendi MK, Vaughn J, Lal A, et al. The prevalence of inpatients at thirty-three U.S. hospitals appropriate for and receiving referral to palliative care. J Palliat Med. 2016;19:360-372.
33. Rush B, Hertz P, Bond A, et al. Use of palliative care in patients with end-stage COPD and receiving home oxygen: national trends and barriers to care in the United States. Chest. 2017;151:41-46.
Case Presentation
A 65-year-old man with severe chronic obstructive disease (COPD; forced expiratory volume in 1 second/forced vital capacity ratio [FEV1/FVC], 27; FEV1 25% of predicted; residual volume 170% of predicted for his age and height) was seen in the pulmonary clinic. His medications include a long-acting beta agonist (LABA)/long-acting muscarinic antagonist (LAMA) combination that he uses twice daily as advised. He uses his rescue albuterol inhaler roughly once a week. The patient complains of severe disabling shortness of breath with exertion and severe limitation of his quality of life because of his inability to lead a normal active life. He is on 2 L/min of oxygen at all times. He has received pulmonary rehabilitation in hopes of improving his quality of life but can only climb a flight of stairs before he must stop to rest. He asks about options but does not want to consider lung transplantation today. His most recent chest computed tomography (CT) scan demonstrates upper lobe predominant emphysematous changes with no masses or nodules.
What are the patient's options at this time?
Lung volume reduction surgery (LVRS) attempts to reduce space-occupying severely diseased, hyperexpanded lung, thus allowing the relatively normal adjoining lung parenchyma to expand into the vacated space and function effectively.1 Hence, such therapies are suitable for patients with emphysematous lungs and not those with bronchitic-predominant COPD. LVRS offers a greater chance of improvement in exercise capacity, lung function, quality of life, and dyspnea in the correctly chosen patient population, as compared with pharmacologic management alone.2 However, the procedure is associated with risks, including higher short-term morbidity and mortality.2 Patients with predominantly upper-lobe emphysema and a low maximal workload after rehabilitation were noted to have lower mortality, a greater probability of improvement in exercise capacity, and a greater probability of improvement in symptoms if they underwent surgery compared to medical therapy alone.2 On the contrary, patients with predominantly non–upper-lobe emphysema and a high maximal workload after rehabilitation had higher mortality if they underwent surgery compared to receiving medical therapy alone.2 Thus, a subgroup of patients with homogeneous emphysema symmetrically affecting the upper and lower lobes are considered to be unlikely to benefit from this surgery.2,3
Valves and other methods of lung volume reduction such as coils, sealants, intrapulmonary vents, and thermal vapor in the bronchi or subsegmental airways have emerged as new techniques for nonsurgical lung volume reduction.4-9 Endobronchial-valve therapy is associated with improvement in lung function and with clinical benefits that are greatest in the presence of heterogeneous lung involvement. This works by the same principle as LVRS, by reduction of the most severely diseased lung units, expansion of the more viable, less emphysematous lung results in substantial improvements in lung mechanics.10,11 The most important complications of this procedure include pneumonia, pneumothorax, hemoptysis, and increased frequency of COPD exacerbation in the following 30 days. The fact that a high-heterogeneity subgroup had greater improvements in both the FEV1 and distance on the 6-minute walk test than did patients with lower heterogeneity supports the use of quantitative high-resolution computed tomography (HRCT) in selecting patients for endobronchial-valve therapy.12 The HRCT scans also help in identifying those with complete fissures, a marker of lack of collateral ventilation (CV+) between different lobes. Presence of CV+ state predicts failure of endobronchial valve and all forms of endoscopic LVRS.13 Bronchoscopic thermal vapor ablation (BTVA) therapy can potentially work on a subsegmental level and be successful for treatment of emphysema with lack of intact fissures on CT scans. Other methods that have the potential to be effective in those with collateral ventilation would be endoscopic coil therapy and polymeric lung volume reduction.11,14 Unfortunately, there are no randomized controlled trial data demonstrating clinically meaningful improvement following coil therapy or polymeric lung volume reduction in this CV+ patient population. Vapor therapy is perhaps the only technique that has been found to be effective in upper lobe predominant emphysema even with CV+ status.13
Our patient has evidence of air trapping and emphysema based on a high residual volume. A CT scan of the chest can determine the nature of the emphysema (heterogeneous versus homogenous) and based on these findings, further determination of the best strategy for lung volume reduction can be made.
Is there a role for long-term oxygen therapy?
Long-term oxygen therapy (LTOT) used for more than 15 hours a day is thought to reduce mortality among patients with COPD and severe resting hypoxemia.15-18 More recent studies have failed to show similar beneficial effects of LTOT. A recent study examined the effects of LTOT in randomized fashion and determined that supplemental oxygen for patients with stable COPD and resting or exercise-induced moderate desaturation did not affect the time to death or first hospitalization, time to first COPD exacerbation, time to first hospitalization for a COPD exacerbation, the rate of all hospitalizations, the rate of all COPD exacerbations, or changes in measures of quality of life, depression, anxiety, or functional status.19
Our patient is currently on long-term oxygen therapy and in spite of some uncertainty as to its benefit, it is prudent to order oxygen therapy until further clarification is available.
What is the role of pulmonary rehabilitation?
Pulmonary rehabilitation is an established treatment for patients with chronic lung disease.20 Benefits include improvement in exercise tolerance, symptoms, and quality of life, with a reduction in the use of health care resources.21 A Spanish population-based cohort study that looked at the influence of regular physical activity on COPD showed that patients who reported low, moderate, or high physical activity had a lower risk of COPD admissions and all-cause mortality than patients with very low physical activity after adjusting for all confounders.22
As previously mentioned, patients in GOLD categories B, C, and D should be offered pulmonary rehabilitation as part of their treatment.23 The ideal patient is one who is not too sick to undergo rehabilitation and is motivated to improve his or her quality of life.
What is the current scope of lung transplantation in the management of severe COPD?
There is an indisputable role for lung transplantation in end-stage COPD. However, lung transplantation does not benefit all COPD patients. There is a subset of patients for whom the treatment provides a survival benefit. It has been reported that 79% of patients with an FEV1 < 16% predicted will survive at least 1 additional year after transplant, but only 11% of patients with an FEV1 > 25% will do so.24 The pre-transplant BODE (body mass index, airflow obstruction/FEV1, dyspnea, and exercise capacity) index score is used to identify patients who will benefit from lung transplantation.25,26 International guidelines for the selection of lung transplant candidates identify the following patient characteristics:27
- The disease is progressive, despite maximal treatment including medication, pulmonary rehabilitation, and oxygen therapy;
- The patient is not a candidate for endoscopic or surgical LVRS;
- BODE index is 5 to 6;
- The PCO2 is greater than 50 mm Hg (6.6 kPa) and/or PO2 is less than 60 mm Hg (8 kPa);
- FEV1 is 25% predicted.
The perioperative mortality of lung transplantation surgery has been reduced to less than 10%. Risk of complications from surgery in the perioperative period, such as bronchial dehiscence, infectious complications, and acute rejection, have also been reduced but do occur. Chronic allograft dysfunction and the risk of lung cancer in cases of single lung transplant should be discussed with the patient before surgery.28
How can we incorporate palliative care into the management plan for patients with COPD?
Among patients with end-stage COPD on home oxygen therapy who have required mechanical ventilation for an exacerbation, only 55% are alive at 1 year.29 COPD patients at high risk of death within the next year of life as well as patients with refractory symptoms and unmet needs are candidates for early palliative care. Palliative care and palliative care specialists can aid in reducing symptom burden and improving quality of life among these patients and their family members, and palliative care is recommended by multiple international societies for patients with advanced COPD.30,31 In spite of these recommendations, the utilization of palliative care resources has been dismally low.32,33 Improving physician-patient communication regarding palliative services and patients’ unmet care needs will help ensure that COPD patients receive adequate palliative care services at the appropriate time.
Conclusion
COPD is a leading cause of morbidity and mortality in the United States and represents a significant economic burden for both individuals and society. The goals in COPD management are to provide symptom relief, improve the quality of life, preserve lung function, and reduce the frequency of exacerbations and mortality. COPD management is guided by disease severity that is measured using the GOLD multimodal staging system and requires a multidisciplinary approach. Several classes of medication are available for treatment, and a step-wise approach should be applied in building an effective pharmacologic regimen. In addition to pharmacologic therapies, nonpharmacologic therapies, including smoking cessation, vaccinations, proper nutrition, and maintaining physical activity, are an important part of long-term management. Those who continue to be symptomatic despite appropriate maximal therapy may be candidates for lung volume reduction. Palliative care services for COPD patients, which can aid in reducing symptom burden and improving quality of life, should not be overlooked.
Case Presentation
A 65-year-old man with severe chronic obstructive disease (COPD; forced expiratory volume in 1 second/forced vital capacity ratio [FEV1/FVC], 27; FEV1 25% of predicted; residual volume 170% of predicted for his age and height) was seen in the pulmonary clinic. His medications include a long-acting beta agonist (LABA)/long-acting muscarinic antagonist (LAMA) combination that he uses twice daily as advised. He uses his rescue albuterol inhaler roughly once a week. The patient complains of severe disabling shortness of breath with exertion and severe limitation of his quality of life because of his inability to lead a normal active life. He is on 2 L/min of oxygen at all times. He has received pulmonary rehabilitation in hopes of improving his quality of life but can only climb a flight of stairs before he must stop to rest. He asks about options but does not want to consider lung transplantation today. His most recent chest computed tomography (CT) scan demonstrates upper lobe predominant emphysematous changes with no masses or nodules.
What are the patient's options at this time?
Lung volume reduction surgery (LVRS) attempts to reduce space-occupying severely diseased, hyperexpanded lung, thus allowing the relatively normal adjoining lung parenchyma to expand into the vacated space and function effectively.1 Hence, such therapies are suitable for patients with emphysematous lungs and not those with bronchitic-predominant COPD. LVRS offers a greater chance of improvement in exercise capacity, lung function, quality of life, and dyspnea in the correctly chosen patient population, as compared with pharmacologic management alone.2 However, the procedure is associated with risks, including higher short-term morbidity and mortality.2 Patients with predominantly upper-lobe emphysema and a low maximal workload after rehabilitation were noted to have lower mortality, a greater probability of improvement in exercise capacity, and a greater probability of improvement in symptoms if they underwent surgery compared to medical therapy alone.2 On the contrary, patients with predominantly non–upper-lobe emphysema and a high maximal workload after rehabilitation had higher mortality if they underwent surgery compared to receiving medical therapy alone.2 Thus, a subgroup of patients with homogeneous emphysema symmetrically affecting the upper and lower lobes are considered to be unlikely to benefit from this surgery.2,3
Valves and other methods of lung volume reduction such as coils, sealants, intrapulmonary vents, and thermal vapor in the bronchi or subsegmental airways have emerged as new techniques for nonsurgical lung volume reduction.4-9 Endobronchial-valve therapy is associated with improvement in lung function and with clinical benefits that are greatest in the presence of heterogeneous lung involvement. This works by the same principle as LVRS, by reduction of the most severely diseased lung units, expansion of the more viable, less emphysematous lung results in substantial improvements in lung mechanics.10,11 The most important complications of this procedure include pneumonia, pneumothorax, hemoptysis, and increased frequency of COPD exacerbation in the following 30 days. The fact that a high-heterogeneity subgroup had greater improvements in both the FEV1 and distance on the 6-minute walk test than did patients with lower heterogeneity supports the use of quantitative high-resolution computed tomography (HRCT) in selecting patients for endobronchial-valve therapy.12 The HRCT scans also help in identifying those with complete fissures, a marker of lack of collateral ventilation (CV+) between different lobes. Presence of CV+ state predicts failure of endobronchial valve and all forms of endoscopic LVRS.13 Bronchoscopic thermal vapor ablation (BTVA) therapy can potentially work on a subsegmental level and be successful for treatment of emphysema with lack of intact fissures on CT scans. Other methods that have the potential to be effective in those with collateral ventilation would be endoscopic coil therapy and polymeric lung volume reduction.11,14 Unfortunately, there are no randomized controlled trial data demonstrating clinically meaningful improvement following coil therapy or polymeric lung volume reduction in this CV+ patient population. Vapor therapy is perhaps the only technique that has been found to be effective in upper lobe predominant emphysema even with CV+ status.13
Our patient has evidence of air trapping and emphysema based on a high residual volume. A CT scan of the chest can determine the nature of the emphysema (heterogeneous versus homogenous) and based on these findings, further determination of the best strategy for lung volume reduction can be made.
Is there a role for long-term oxygen therapy?
Long-term oxygen therapy (LTOT) used for more than 15 hours a day is thought to reduce mortality among patients with COPD and severe resting hypoxemia.15-18 More recent studies have failed to show similar beneficial effects of LTOT. A recent study examined the effects of LTOT in randomized fashion and determined that supplemental oxygen for patients with stable COPD and resting or exercise-induced moderate desaturation did not affect the time to death or first hospitalization, time to first COPD exacerbation, time to first hospitalization for a COPD exacerbation, the rate of all hospitalizations, the rate of all COPD exacerbations, or changes in measures of quality of life, depression, anxiety, or functional status.19
Our patient is currently on long-term oxygen therapy and in spite of some uncertainty as to its benefit, it is prudent to order oxygen therapy until further clarification is available.
What is the role of pulmonary rehabilitation?
Pulmonary rehabilitation is an established treatment for patients with chronic lung disease.20 Benefits include improvement in exercise tolerance, symptoms, and quality of life, with a reduction in the use of health care resources.21 A Spanish population-based cohort study that looked at the influence of regular physical activity on COPD showed that patients who reported low, moderate, or high physical activity had a lower risk of COPD admissions and all-cause mortality than patients with very low physical activity after adjusting for all confounders.22
As previously mentioned, patients in GOLD categories B, C, and D should be offered pulmonary rehabilitation as part of their treatment.23 The ideal patient is one who is not too sick to undergo rehabilitation and is motivated to improve his or her quality of life.
What is the current scope of lung transplantation in the management of severe COPD?
There is an indisputable role for lung transplantation in end-stage COPD. However, lung transplantation does not benefit all COPD patients. There is a subset of patients for whom the treatment provides a survival benefit. It has been reported that 79% of patients with an FEV1 < 16% predicted will survive at least 1 additional year after transplant, but only 11% of patients with an FEV1 > 25% will do so.24 The pre-transplant BODE (body mass index, airflow obstruction/FEV1, dyspnea, and exercise capacity) index score is used to identify patients who will benefit from lung transplantation.25,26 International guidelines for the selection of lung transplant candidates identify the following patient characteristics:27
- The disease is progressive, despite maximal treatment including medication, pulmonary rehabilitation, and oxygen therapy;
- The patient is not a candidate for endoscopic or surgical LVRS;
- BODE index is 5 to 6;
- The PCO2 is greater than 50 mm Hg (6.6 kPa) and/or PO2 is less than 60 mm Hg (8 kPa);
- FEV1 is 25% predicted.
The perioperative mortality of lung transplantation surgery has been reduced to less than 10%. Risk of complications from surgery in the perioperative period, such as bronchial dehiscence, infectious complications, and acute rejection, have also been reduced but do occur. Chronic allograft dysfunction and the risk of lung cancer in cases of single lung transplant should be discussed with the patient before surgery.28
How can we incorporate palliative care into the management plan for patients with COPD?
Among patients with end-stage COPD on home oxygen therapy who have required mechanical ventilation for an exacerbation, only 55% are alive at 1 year.29 COPD patients at high risk of death within the next year of life as well as patients with refractory symptoms and unmet needs are candidates for early palliative care. Palliative care and palliative care specialists can aid in reducing symptom burden and improving quality of life among these patients and their family members, and palliative care is recommended by multiple international societies for patients with advanced COPD.30,31 In spite of these recommendations, the utilization of palliative care resources has been dismally low.32,33 Improving physician-patient communication regarding palliative services and patients’ unmet care needs will help ensure that COPD patients receive adequate palliative care services at the appropriate time.
Conclusion
COPD is a leading cause of morbidity and mortality in the United States and represents a significant economic burden for both individuals and society. The goals in COPD management are to provide symptom relief, improve the quality of life, preserve lung function, and reduce the frequency of exacerbations and mortality. COPD management is guided by disease severity that is measured using the GOLD multimodal staging system and requires a multidisciplinary approach. Several classes of medication are available for treatment, and a step-wise approach should be applied in building an effective pharmacologic regimen. In addition to pharmacologic therapies, nonpharmacologic therapies, including smoking cessation, vaccinations, proper nutrition, and maintaining physical activity, are an important part of long-term management. Those who continue to be symptomatic despite appropriate maximal therapy may be candidates for lung volume reduction. Palliative care services for COPD patients, which can aid in reducing symptom burden and improving quality of life, should not be overlooked.
1. Sabanathan A, Sabanathan S, Shah R, Richardson J. Lung volume reduction surgery for emphysema: a review. J Cardiovasc Surg. 1998;39:237.
2. Group NETTR. Patients at high risk of death after lung-volume–reduction surgery. N Engl J Med. 2001;345:1075-1083.
3. Group NETTR. A randomized trial comparing lung-volume–reduction surgery with medical therapy for severe emphysema. N Engl J Med. 2003;348:2059-2073.
4. Decker MR, Leverson GE, Jaoude WA, Maloney JD. Lung volume reduction surgery since the National Emphysema Treatment Trial: study of Society of Thoracic Surgeons database. J Thorac Cardiovasc Surg. 2014;148:2651-2658.
5. Deslée G, Mal H, Dutau H, et al. Lung volume reduction coil treatment vs usual care in patients with severe emphysema: the REVOLENS randomized clinical trial. JAMA. 2016;315:175-184.
6. Hartman JE, Klooster K, Gortzak K, et al. Long-term follow-up after bronchoscopic lung volume reduction treatment with coils in patients with severe emphysema. Respirology. 2015;20:319-326.
7. Snell GI, Hopkins P, Westall G, et al. A feasibility and safety study of bronchoscopic thermal vapor ablation: a novel emphysema therapy. Ann Thorac Surg. 2009;88:1993-1998.
8. Ingenito EP, Berger RL, Henderson AC, et al. Bronchoscopic lung volume reduction using tissue engineering principles. Am J Respir Crit Care Med. 2003;167:771-778.
9. Ingenito EP, Loring SH, Moy ML, et al. Comparison of physiological and radiological screening for lung volume reduction surgery. Am J Respir Crit Care Med. 2001;163:1068-1073.
10. Shah P, Slebos D, Cardoso P, et al. Bronchoscopic lung-volume reduction with Exhale airway stents for emphysema (EASE trial): randomised, sham-controlled, multicentre trial. Lancet. 2011;378:997-1005.
11. Sciurba FC, Ernst A, Herth FJ, et al. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med. 2010;363:1233-1244.
12. Wan IY, Toma TP, Geddes DM, et al. Bronchoscopic lung volume reduction for end-stage emphysema: report on the first 98 patients. Chest. 2006;129:518-526.
13. Gompelmann D, Eberhardt R, Schuhmann M, et al. Lung volume reduction with vapor ablation in the presence of incomplete fissures: 12-month results from the STEP-UP randomized controlled study. Respiration. 2016;92:397-403.
14. Come CE, Kramer MR, Dransfield MT, et al. A randomised trial of lung sealant versus medical therapy for advanced emphysema. Eur Respir J. 2015;46:651-662.
15. Group NOTT. Continuous or nocturnal oxygen therapy in hypoxemic chronic obstructive lung disease: a clinical trial. Ann Intern Med. 1980;93:391-398.
16. Council M. Long term domiciliary oxygen therapy in chronic hypoxic cor pulmonale complicating chronic bronchitis and emphysema: Report of the Medical Research Council Working Party. Lancet. 1981;1:681-686.
17. Qaseem A, Wilt TJ, Weinberger SE, et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155:179-191.
18. Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187:347-365.
19. Group L-TOTTR. A randomized trial of long-term oxygen for COPD with moderate desaturation. N Engl J Med. 2016;375:1617-1627.
20. McCarthy B, Casey D, Devane D, et al. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015(2):CD003793.
21. Griffiths TL, Burr ML, Campbell IA, et al. Results at 1 year of outpatient multidisciplinary pulmonary rehabilitation: a randomised controlled trial. Lancet. 2000;355:362-368.
22. Garcia-Aymerich J, Lange P, Benet M, et al. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population based cohort study. Thorax. 2006;61:772-778.
23. Global Initiative for Chronic Obstructive Lung Disease (GOLD): Global strategy for the diagnosis, management, and prevention of COPD 2017. www.goldcopd.org. Accessed July 9, 2019.
24. Thabut G, Ravaud P, Christie JD, et al. Determinants of the survival benefit of lung transplantation in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2008;177:1156-1163.
25. Lahzami S, Bridevaux PO, Soccal PM, et al. Survival impact of lung transplantation for COPD. Eur Respir J. 2010;36:74-80.
26. Cerón Navarro J, de Aguiar Quevedo K, Ansótegui Barrera E, et al. Functional outcomes after lung transplant in chronic obstructive pulmonary disease. Arch Bronconeumol. 2015;51:109-114.
27. Weill D, Benden C, Corris PA, et al. A consensus document for the selection of lung transplant candidates: 2014--an update from the Pulmonary Transplantation Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2015;34:1-15.
28. Minai OA, Shah S, Mazzone P, et al. Bronchogenic carcinoma after lung transplantation: characteristics and outcomes. J Thorac Oncol. 2008;3:1404-1409.
29. Hajizadeh N, Goldfeld K, Crothers K. What happens to patients with COPD with long-term oxygen treatment who receive mechanical ventilation for COPD exacerbation? A 1-year retrospective follow- up study. Thorax. 2015;70:294-296.
30. Siouta N, van Beek K, Preston N, et al. Towards integration of palliative care in patients with chronic heart failure and chronic obstructive pulmonary disease: a systematic literature review of European guidelines and pathways. BMC Palliat Care. 2016;15:18.
31. Celli BR, MacNee W; ATS/ERS Task Force. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur Respir J. 2004;23:932-946.
32. Szekendi MK, Vaughn J, Lal A, et al. The prevalence of inpatients at thirty-three U.S. hospitals appropriate for and receiving referral to palliative care. J Palliat Med. 2016;19:360-372.
33. Rush B, Hertz P, Bond A, et al. Use of palliative care in patients with end-stage COPD and receiving home oxygen: national trends and barriers to care in the United States. Chest. 2017;151:41-46.
1. Sabanathan A, Sabanathan S, Shah R, Richardson J. Lung volume reduction surgery for emphysema: a review. J Cardiovasc Surg. 1998;39:237.
2. Group NETTR. Patients at high risk of death after lung-volume–reduction surgery. N Engl J Med. 2001;345:1075-1083.
3. Group NETTR. A randomized trial comparing lung-volume–reduction surgery with medical therapy for severe emphysema. N Engl J Med. 2003;348:2059-2073.
4. Decker MR, Leverson GE, Jaoude WA, Maloney JD. Lung volume reduction surgery since the National Emphysema Treatment Trial: study of Society of Thoracic Surgeons database. J Thorac Cardiovasc Surg. 2014;148:2651-2658.
5. Deslée G, Mal H, Dutau H, et al. Lung volume reduction coil treatment vs usual care in patients with severe emphysema: the REVOLENS randomized clinical trial. JAMA. 2016;315:175-184.
6. Hartman JE, Klooster K, Gortzak K, et al. Long-term follow-up after bronchoscopic lung volume reduction treatment with coils in patients with severe emphysema. Respirology. 2015;20:319-326.
7. Snell GI, Hopkins P, Westall G, et al. A feasibility and safety study of bronchoscopic thermal vapor ablation: a novel emphysema therapy. Ann Thorac Surg. 2009;88:1993-1998.
8. Ingenito EP, Berger RL, Henderson AC, et al. Bronchoscopic lung volume reduction using tissue engineering principles. Am J Respir Crit Care Med. 2003;167:771-778.
9. Ingenito EP, Loring SH, Moy ML, et al. Comparison of physiological and radiological screening for lung volume reduction surgery. Am J Respir Crit Care Med. 2001;163:1068-1073.
10. Shah P, Slebos D, Cardoso P, et al. Bronchoscopic lung-volume reduction with Exhale airway stents for emphysema (EASE trial): randomised, sham-controlled, multicentre trial. Lancet. 2011;378:997-1005.
11. Sciurba FC, Ernst A, Herth FJ, et al. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med. 2010;363:1233-1244.
12. Wan IY, Toma TP, Geddes DM, et al. Bronchoscopic lung volume reduction for end-stage emphysema: report on the first 98 patients. Chest. 2006;129:518-526.
13. Gompelmann D, Eberhardt R, Schuhmann M, et al. Lung volume reduction with vapor ablation in the presence of incomplete fissures: 12-month results from the STEP-UP randomized controlled study. Respiration. 2016;92:397-403.
14. Come CE, Kramer MR, Dransfield MT, et al. A randomised trial of lung sealant versus medical therapy for advanced emphysema. Eur Respir J. 2015;46:651-662.
15. Group NOTT. Continuous or nocturnal oxygen therapy in hypoxemic chronic obstructive lung disease: a clinical trial. Ann Intern Med. 1980;93:391-398.
16. Council M. Long term domiciliary oxygen therapy in chronic hypoxic cor pulmonale complicating chronic bronchitis and emphysema: Report of the Medical Research Council Working Party. Lancet. 1981;1:681-686.
17. Qaseem A, Wilt TJ, Weinberger SE, et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline update from the American College of Physicians, American College of Chest Physicians, American Thoracic Society, and European Respiratory Society. Ann Intern Med. 2011;155:179-191.
18. Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187:347-365.
19. Group L-TOTTR. A randomized trial of long-term oxygen for COPD with moderate desaturation. N Engl J Med. 2016;375:1617-1627.
20. McCarthy B, Casey D, Devane D, et al. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2015(2):CD003793.
21. Griffiths TL, Burr ML, Campbell IA, et al. Results at 1 year of outpatient multidisciplinary pulmonary rehabilitation: a randomised controlled trial. Lancet. 2000;355:362-368.
22. Garcia-Aymerich J, Lange P, Benet M, et al. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population based cohort study. Thorax. 2006;61:772-778.
23. Global Initiative for Chronic Obstructive Lung Disease (GOLD): Global strategy for the diagnosis, management, and prevention of COPD 2017. www.goldcopd.org. Accessed July 9, 2019.
24. Thabut G, Ravaud P, Christie JD, et al. Determinants of the survival benefit of lung transplantation in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2008;177:1156-1163.
25. Lahzami S, Bridevaux PO, Soccal PM, et al. Survival impact of lung transplantation for COPD. Eur Respir J. 2010;36:74-80.
26. Cerón Navarro J, de Aguiar Quevedo K, Ansótegui Barrera E, et al. Functional outcomes after lung transplant in chronic obstructive pulmonary disease. Arch Bronconeumol. 2015;51:109-114.
27. Weill D, Benden C, Corris PA, et al. A consensus document for the selection of lung transplant candidates: 2014--an update from the Pulmonary Transplantation Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2015;34:1-15.
28. Minai OA, Shah S, Mazzone P, et al. Bronchogenic carcinoma after lung transplantation: characteristics and outcomes. J Thorac Oncol. 2008;3:1404-1409.
29. Hajizadeh N, Goldfeld K, Crothers K. What happens to patients with COPD with long-term oxygen treatment who receive mechanical ventilation for COPD exacerbation? A 1-year retrospective follow- up study. Thorax. 2015;70:294-296.
30. Siouta N, van Beek K, Preston N, et al. Towards integration of palliative care in patients with chronic heart failure and chronic obstructive pulmonary disease: a systematic literature review of European guidelines and pathways. BMC Palliat Care. 2016;15:18.
31. Celli BR, MacNee W; ATS/ERS Task Force. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur Respir J. 2004;23:932-946.
32. Szekendi MK, Vaughn J, Lal A, et al. The prevalence of inpatients at thirty-three U.S. hospitals appropriate for and receiving referral to palliative care. J Palliat Med. 2016;19:360-372.
33. Rush B, Hertz P, Bond A, et al. Use of palliative care in patients with end-stage COPD and receiving home oxygen: national trends and barriers to care in the United States. Chest. 2017;151:41-46.
Study explores link between inhaled corticosteroid for COPD and reduced lung cancer risk
recent research from the European Respiratory Journal has shown.
“The appropriate use of [inhaled corticosteroids] ICS in [chronic obstructive pulmonary disease] COPD patients is often debated and not all patients might benefit from the use of ICS. The clinical benefits and risk of use in an individual patient must be weighed by the physician,” wrote Adam J.N. Raymakers, MSc, PhD, of the University of British Columbia’s Collaboration for Outcomes Research and Evaluation (CORE), Vancouver, B.C., and colleagues.
“This study, however, indicates that potential benefits may accrue from ICS use in COPD patients in terms of reduced lung cancer risk, and that sustained use may be associated with reduced risk of lung cancer.”
Dr. Raymakers and colleagues did an analysis of 39,676 patients with COPD (mean age, 70.7 years; 53% female) who received ICS between 1997 and 2007 and linked those patients to a registry of cancer patients in British Columbia. The linked databases included the Medical Services Plan (MSP) payment information file, Discharge Abstract Database (DAD), PharmaNet data file, and the British Columbia Cancer Registry (BCCR). The researchers determined a patient had COPD if he or she received three or more prescriptions related to COPD, while ICS exposure was analyzed in the context of a patient’s ICS exposure, cumulative duration, cumulative dose, and weighted cumulative duration and dose.
The analysis revealed 372,075 prescriptions for ICS were dispensed and 71.2% of the patients were “distinct users” of ICS, with patients filling a median of eight prescriptions at mean 5.2 years of follow-up. Fluticasone propionate was the most common ICS prescribed at a dose of 0.64 mg per day, and patients had median 60 days of ICS supplied per person.
Overall, there were 994 cases of lung cancer (2.5%), and exposure to ICS was linked to a 30% reduction in lung cancer risk (hazard ratio, 0.70; 95% confidence interval, 0.61-0.80), while recency-weighted duration of ICS exposure was linked to a 26% reduction in lung cancer risk (HR, 0.74; 95% CI, 0.66-0.87). There was a 43% reduced risk of lung cancer per gram of ICS when the data were measured by recency-weighted cumulative dosage.
In a multivariate analysis, ICS use was associated with a 30% reduction in risk of non–small cell lung cancer (HR, 0.70; 95% CI, 0.60-0.82), which the researchers said suggests ICS provides a protective effect for patients against lung cancer. “These results highlight the importance of properly identifying which patients might be at the highest risk of lung cancer, to enhance the therapeutic benefits of ICS in these COPD patients,” they wrote.
This study received funding from the Canadian Institutes of Health Research. The authors report no conflicts of interest.
SOURCE: Raymakers A, et al. Eur Respir J. 2019. doi: 10.1183/13993003.01257-2018.
recent research from the European Respiratory Journal has shown.
“The appropriate use of [inhaled corticosteroids] ICS in [chronic obstructive pulmonary disease] COPD patients is often debated and not all patients might benefit from the use of ICS. The clinical benefits and risk of use in an individual patient must be weighed by the physician,” wrote Adam J.N. Raymakers, MSc, PhD, of the University of British Columbia’s Collaboration for Outcomes Research and Evaluation (CORE), Vancouver, B.C., and colleagues.
“This study, however, indicates that potential benefits may accrue from ICS use in COPD patients in terms of reduced lung cancer risk, and that sustained use may be associated with reduced risk of lung cancer.”
Dr. Raymakers and colleagues did an analysis of 39,676 patients with COPD (mean age, 70.7 years; 53% female) who received ICS between 1997 and 2007 and linked those patients to a registry of cancer patients in British Columbia. The linked databases included the Medical Services Plan (MSP) payment information file, Discharge Abstract Database (DAD), PharmaNet data file, and the British Columbia Cancer Registry (BCCR). The researchers determined a patient had COPD if he or she received three or more prescriptions related to COPD, while ICS exposure was analyzed in the context of a patient’s ICS exposure, cumulative duration, cumulative dose, and weighted cumulative duration and dose.
The analysis revealed 372,075 prescriptions for ICS were dispensed and 71.2% of the patients were “distinct users” of ICS, with patients filling a median of eight prescriptions at mean 5.2 years of follow-up. Fluticasone propionate was the most common ICS prescribed at a dose of 0.64 mg per day, and patients had median 60 days of ICS supplied per person.
Overall, there were 994 cases of lung cancer (2.5%), and exposure to ICS was linked to a 30% reduction in lung cancer risk (hazard ratio, 0.70; 95% confidence interval, 0.61-0.80), while recency-weighted duration of ICS exposure was linked to a 26% reduction in lung cancer risk (HR, 0.74; 95% CI, 0.66-0.87). There was a 43% reduced risk of lung cancer per gram of ICS when the data were measured by recency-weighted cumulative dosage.
In a multivariate analysis, ICS use was associated with a 30% reduction in risk of non–small cell lung cancer (HR, 0.70; 95% CI, 0.60-0.82), which the researchers said suggests ICS provides a protective effect for patients against lung cancer. “These results highlight the importance of properly identifying which patients might be at the highest risk of lung cancer, to enhance the therapeutic benefits of ICS in these COPD patients,” they wrote.
This study received funding from the Canadian Institutes of Health Research. The authors report no conflicts of interest.
SOURCE: Raymakers A, et al. Eur Respir J. 2019. doi: 10.1183/13993003.01257-2018.
recent research from the European Respiratory Journal has shown.
“The appropriate use of [inhaled corticosteroids] ICS in [chronic obstructive pulmonary disease] COPD patients is often debated and not all patients might benefit from the use of ICS. The clinical benefits and risk of use in an individual patient must be weighed by the physician,” wrote Adam J.N. Raymakers, MSc, PhD, of the University of British Columbia’s Collaboration for Outcomes Research and Evaluation (CORE), Vancouver, B.C., and colleagues.
“This study, however, indicates that potential benefits may accrue from ICS use in COPD patients in terms of reduced lung cancer risk, and that sustained use may be associated with reduced risk of lung cancer.”
Dr. Raymakers and colleagues did an analysis of 39,676 patients with COPD (mean age, 70.7 years; 53% female) who received ICS between 1997 and 2007 and linked those patients to a registry of cancer patients in British Columbia. The linked databases included the Medical Services Plan (MSP) payment information file, Discharge Abstract Database (DAD), PharmaNet data file, and the British Columbia Cancer Registry (BCCR). The researchers determined a patient had COPD if he or she received three or more prescriptions related to COPD, while ICS exposure was analyzed in the context of a patient’s ICS exposure, cumulative duration, cumulative dose, and weighted cumulative duration and dose.
The analysis revealed 372,075 prescriptions for ICS were dispensed and 71.2% of the patients were “distinct users” of ICS, with patients filling a median of eight prescriptions at mean 5.2 years of follow-up. Fluticasone propionate was the most common ICS prescribed at a dose of 0.64 mg per day, and patients had median 60 days of ICS supplied per person.
Overall, there were 994 cases of lung cancer (2.5%), and exposure to ICS was linked to a 30% reduction in lung cancer risk (hazard ratio, 0.70; 95% confidence interval, 0.61-0.80), while recency-weighted duration of ICS exposure was linked to a 26% reduction in lung cancer risk (HR, 0.74; 95% CI, 0.66-0.87). There was a 43% reduced risk of lung cancer per gram of ICS when the data were measured by recency-weighted cumulative dosage.
In a multivariate analysis, ICS use was associated with a 30% reduction in risk of non–small cell lung cancer (HR, 0.70; 95% CI, 0.60-0.82), which the researchers said suggests ICS provides a protective effect for patients against lung cancer. “These results highlight the importance of properly identifying which patients might be at the highest risk of lung cancer, to enhance the therapeutic benefits of ICS in these COPD patients,” they wrote.
This study received funding from the Canadian Institutes of Health Research. The authors report no conflicts of interest.
SOURCE: Raymakers A, et al. Eur Respir J. 2019. doi: 10.1183/13993003.01257-2018.
FROM THE EUROPEAN RESPIRATORY JOURNAL
ACIP adds hexavalent vaccine to VFC program
The pediatric hexavalent vaccine (DTaP-[inactivated poliovirus] IPV-[hepatitis B] HepB-[Haemophilis influenzae type b] Hib) should be included as an option in the Vaccines for Children (VFC) program for the infant series at ages 2, 4, and 6 months, according to unanimous votes at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The addition of the vaccine to the VFC program required no motions on the part of the committee, but involved separate votes on each component of the vaccine.
Combination vaccination has been associated with increased coverage and more likely completion of the full infant vaccine series, said Sara Oliver, MD, of the CDC’s National Center for Immunization and Respiratory Diseases.
The new vaccine is being developed jointly by Sanofi and Merck, and has been approved by the Food and Drug Administration for use in children through age 4 years.
Dr. Oliver presented evidence that the safety profile of the combination vaccine is consistent with that of the component vaccines. In addition, “use of combination vaccines can reduce the number of injections patient receive and alleviate concern associated with the number of injections,” she said. However, “considerations should include provider assessment, patient preference, and the potential for adverse events.”
although it will not be available until 2021 in order to ensure sufficient supply, Dr. Oliver noted.
The combination vaccination work group considered whether the new vaccine should be preferentially recommended for American Indian and Alaskan Native populations, but they concluded that post–dose one immunogenicity data are needed before such a preferential recommendation can be made.
The ACIP members had no financial conflicts to disclose.
The pediatric hexavalent vaccine (DTaP-[inactivated poliovirus] IPV-[hepatitis B] HepB-[Haemophilis influenzae type b] Hib) should be included as an option in the Vaccines for Children (VFC) program for the infant series at ages 2, 4, and 6 months, according to unanimous votes at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The addition of the vaccine to the VFC program required no motions on the part of the committee, but involved separate votes on each component of the vaccine.
Combination vaccination has been associated with increased coverage and more likely completion of the full infant vaccine series, said Sara Oliver, MD, of the CDC’s National Center for Immunization and Respiratory Diseases.
The new vaccine is being developed jointly by Sanofi and Merck, and has been approved by the Food and Drug Administration for use in children through age 4 years.
Dr. Oliver presented evidence that the safety profile of the combination vaccine is consistent with that of the component vaccines. In addition, “use of combination vaccines can reduce the number of injections patient receive and alleviate concern associated with the number of injections,” she said. However, “considerations should include provider assessment, patient preference, and the potential for adverse events.”
although it will not be available until 2021 in order to ensure sufficient supply, Dr. Oliver noted.
The combination vaccination work group considered whether the new vaccine should be preferentially recommended for American Indian and Alaskan Native populations, but they concluded that post–dose one immunogenicity data are needed before such a preferential recommendation can be made.
The ACIP members had no financial conflicts to disclose.
The pediatric hexavalent vaccine (DTaP-[inactivated poliovirus] IPV-[hepatitis B] HepB-[Haemophilis influenzae type b] Hib) should be included as an option in the Vaccines for Children (VFC) program for the infant series at ages 2, 4, and 6 months, according to unanimous votes at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The addition of the vaccine to the VFC program required no motions on the part of the committee, but involved separate votes on each component of the vaccine.
Combination vaccination has been associated with increased coverage and more likely completion of the full infant vaccine series, said Sara Oliver, MD, of the CDC’s National Center for Immunization and Respiratory Diseases.
The new vaccine is being developed jointly by Sanofi and Merck, and has been approved by the Food and Drug Administration for use in children through age 4 years.
Dr. Oliver presented evidence that the safety profile of the combination vaccine is consistent with that of the component vaccines. In addition, “use of combination vaccines can reduce the number of injections patient receive and alleviate concern associated with the number of injections,” she said. However, “considerations should include provider assessment, patient preference, and the potential for adverse events.”
although it will not be available until 2021 in order to ensure sufficient supply, Dr. Oliver noted.
The combination vaccination work group considered whether the new vaccine should be preferentially recommended for American Indian and Alaskan Native populations, but they concluded that post–dose one immunogenicity data are needed before such a preferential recommendation can be made.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING