User login
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
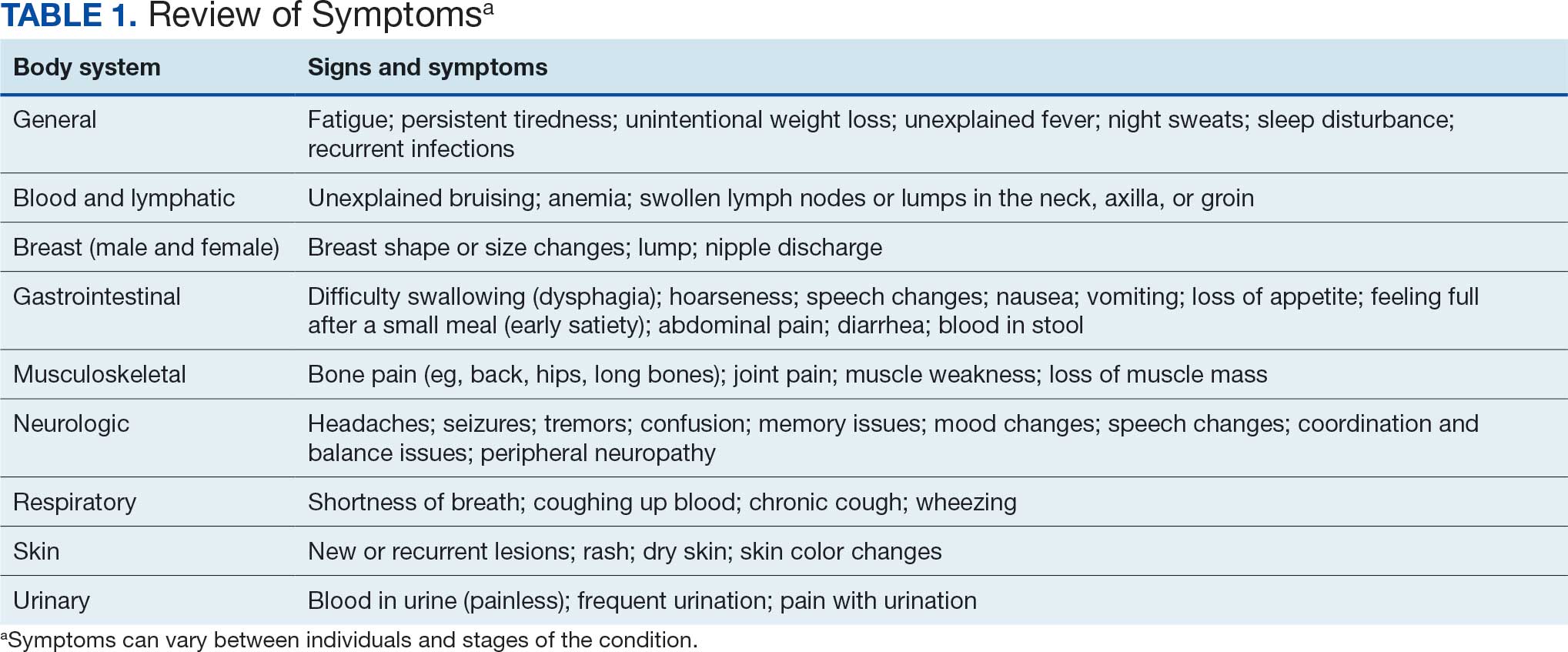
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).
Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
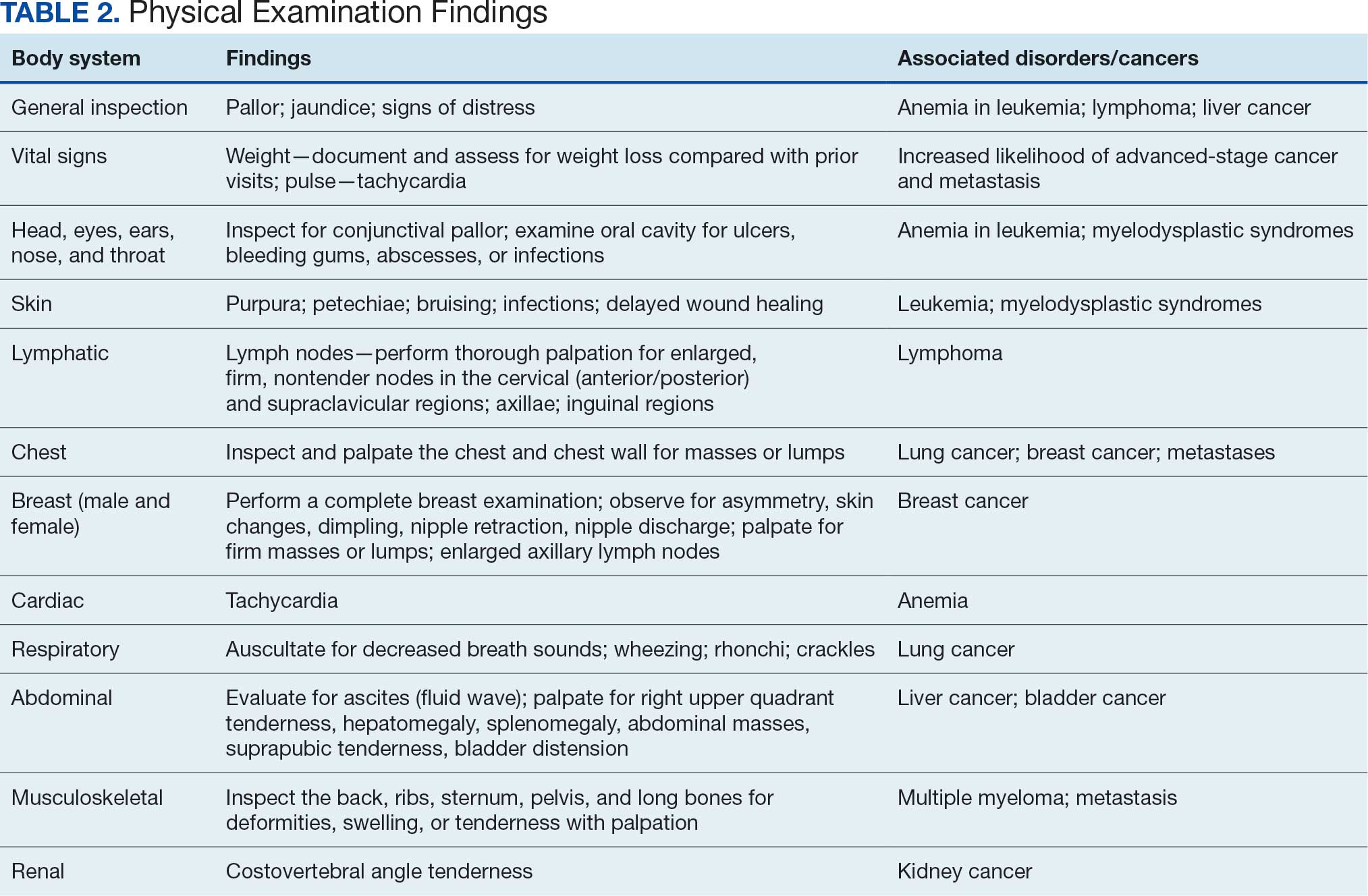
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.
Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).
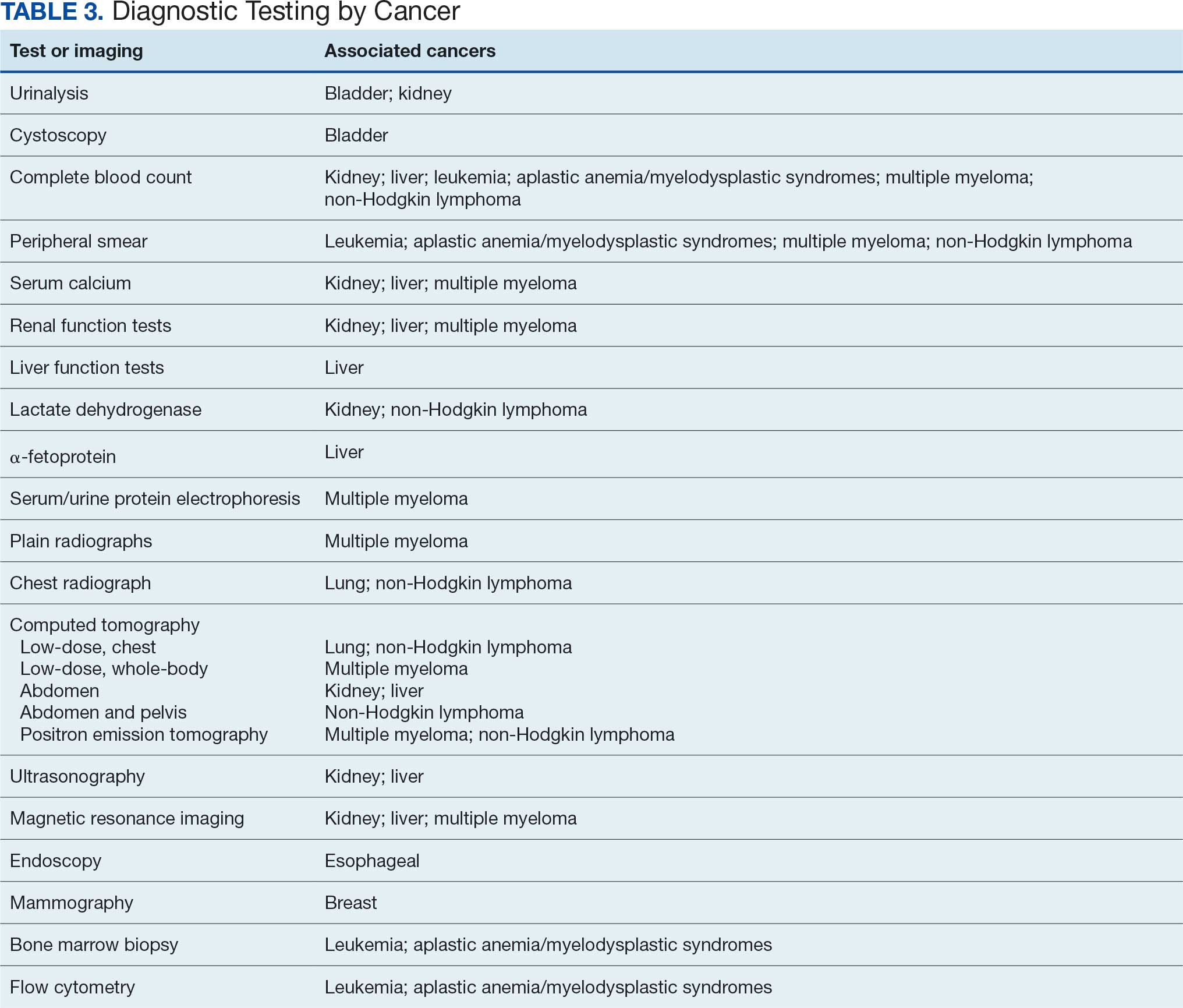
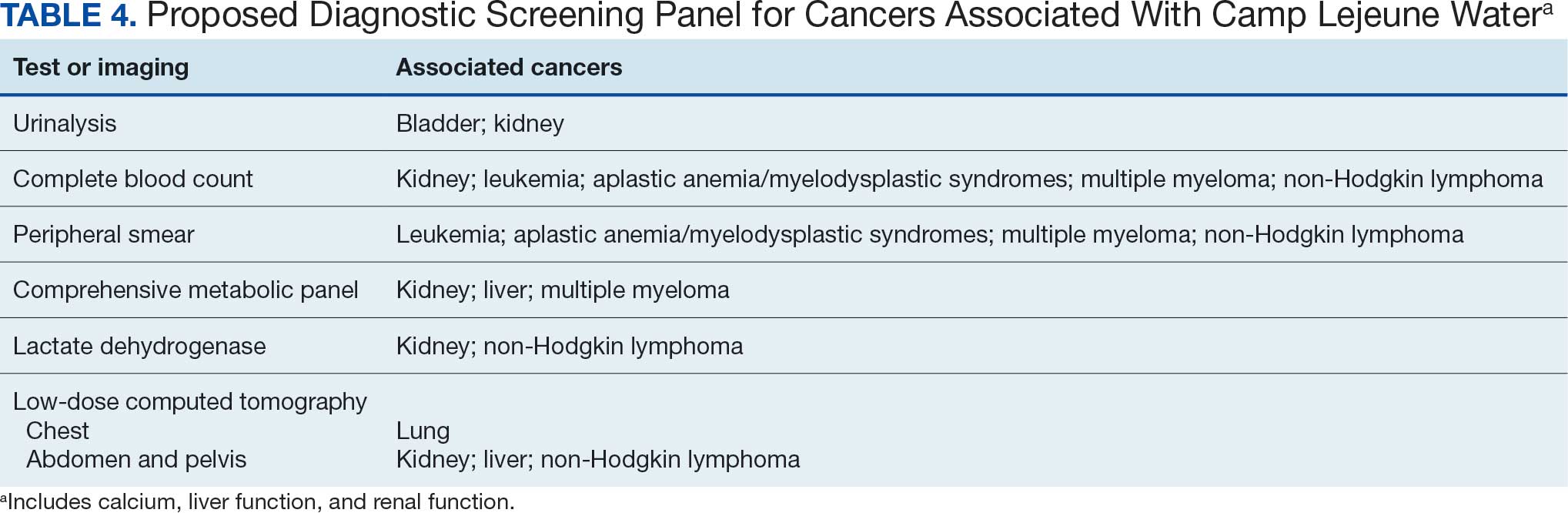
A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
VA Performs Its First ‘Bloodless’ Stem Cell Transplant
PHOENIX ‑ A US Department of Veterans Affairs (VA) hospital in Tennessee has performed the first “bloodless” autologous stem cell transplant within the Veterans Health Administration, treating a 61-year-old Jehovah’s Witness patient with multiple myeloma who traveled from California for the procedure.
The case, presented at the annual meeting of the Association of VA Hematology/Oncology, stated that “we should not withhold any therapies for patients who are Jehovah’s Witnesses out of fear of them bleeding out or having complications from anemia,” said Bhagirathbhai Dholaria, MBBS, an associate professor of medicine at Vanderbilt University Medical Center who worked with the VA Tennessee Valley Healthcare System in Nashville.
While Jehovah’s Witnesses accept medical treatment, their faith forbids blood transfusions, including of preoperative autologous blood, due to its interpretation of the Bible. The faith allows individuals to decide whether to accept stem cells collected from their blood or someone else’s “provided that blood components are not intentionally collected, stored, and reinfused along with the stem cells.”
There are an estimated 1.2 million Jehovah’s Witnesses in the US.
Traditional Stem Cell Transplants Require Blood Support
In conventional autologous stem cell transplants for multiple myeloma, high-dose chemotherapy temporarily wipes out the patient’s bone marrow for about 2 to 3 weeks, Dholaria explained. During this period, patients typically receive 2 units of packed red blood cells and platelet transfusions to prevent severe complications from anemia and low platelet counts.
“Because of this reason, Jehovah’s Witnesses have been traditionally denied these therapies,” Dholaria said.
However, bloodless autologous transplants have been performed for about 2 decades, and Vanderbilt University has been offering the procedures for about 3 years, according to Dholaria.
For the first bloodless procedure in the VA, the patient–who had an aggressive, newly diagnosed IgG kappa multiple myeloma–was evaluated.
“He had been treated by local doctors in California. Otherwise, he was actually in really good shape. Physically, he didn’t have any major issues,” Dholaria said. “So, he met the criteria for our bloodless protocol, and we decided to offer him the procedure.”
The team consulted ethics and legal departments and noted the patient’s blood product preferences in his electronic health record. The patient then underwent a preoptimization protocol that included erythropoiesis-stimulating agents, intravenous iron, and vitamin B12 supplementation to boost blood counts before the transplant, according to the case presentation.
Special Protocol Required in ‘Bloodless’ Procedures
After stem cell collection and chemotherapy, patients undergoing bloodless procedures receive aggressive growth factor support to minimize the duration and severity of cytopenia, Dholaria said. As part of the protocol, the care team uses pediatric tubes for blood draws to minimize blood loss and monitors patients closely on cardiac monitors, he added. In addition, blood draws are only performed every 3 days.
“We watch for any cardiac decompensation because these patients have severe anemia for a brief period of time. We make sure they don’t [have a] heart attack or arrhythmias,” Dholaria said. “Or if the platelets are too low, and they start oozing blood from the nose, gums, or gut, that needs to be dealt with accordingly.”
For bleeding complications, the team uses clotting factors and intravenous and oral medications to support remaining platelet function rather than platelet transfusions.
The patient in this case tolerated the transplant “exceptionally well with minimal complications,” according to the case presentation. He achieved full engraftment on day 14 after transplant and was discharged from inpatient care with continued monitoring through day 30.
“The patient was very compliant,” said Salyka Sengsayadeth, MD, medical director of the VA Tennessee Valley Healthcare System Stem Cell Transplant and Cellular Therapy Program and associate professor of medicine at Vanderbilt.
“He tolerated everything that we needed to do,” she said. “He called us when he needed to call us and did everything that we asked and recommended for him.”
The patient’s roughly 30-day hospital stay matched that of typical transplant patients, Sengsayadeth noted. His myeloma responded to treatment, and he returned to California, Dholaria said.
‘Bloodless’ Procedures Not for All Stem Cell Transplants
The case highlights the availability of stem cell transplants in the VA–they are only performed in Seattle and Nashville–and opportunities for patients who wish to avoid blood transfusions. Sengsayadeth said the bloodless protocol is available for patients without religious objections who simply prefer to avoid blood products.
Dholaria cautioned that bloodless protocol applies specifically to autologous transplants, where patients receive their own stem cells. The team does not plan to offer bloodless allogeneic transplants, which use donor stem cells for conditions like leukemia, due to higher risks. In addition, most Jehovah’s Witnesses decline allogeneic transplants because they do not accept stem cells from another person, Dholaria said.
Beyond multiple myeloma, the Tennessee Valley Healthcare System offers bloodless autologous transplants for various blood cancers, including non-Hodgkin lymphomas such as large B-cell lymphoma, follicular lymphoma, and mantle cell lymphoma, as well as lymphomas affecting the brain, Dholaria said.
Clinicians “should start thinking about this early on, as soon as the cancer diagnosis is made, to make the referral and get the patient on our radar,” Dholaria said.
Sengsayadeth said physicians within the VA typically know how to refer appropriate patients to her team. “They just send us an email or give us a call or a message to say ‘I have this patient. Do you think they’re someone I should send to you?’ We usually answer right back, and then we can proceed with the full evaluation if we think that’s a reasonable thing to do.”
‘Treated Like Family’
The patient, a Marine Corps veteran named Keith Cody, spoke about the procedure in a video interview. Cody said he was reluctant at first to undergo the procedure because he didn’t understand what it would accomplish.
“As I was doing the massive chemo every week, and then suffering with the side effects, I decided to ask again about this procedure and how it improves my quality of life,” he said.
At the time of the taping of the video, Cody was getting ready to go home to California. “They’ve told me that I’ll still need more time to get my energy back, but I do feel much better already,” he said.
He also praised the staff. “Everybody that we came across, I enjoyed the interactions. It’s actually sad to leave people behind that you really felt treated you like family.”
Dholaria discloses relationships with Janssen, Angiocrine, Pfizer, Poseida, MEI, Orcabio, Wugen, Allovir, Adicet, BMS, Molecular Templates, Atara, MJH, Arvinas, Janssen, ADC, Gilead, GSK, Caribou, F. Hoffmann-La Roche AG, Autolus, and Pierre Fabre.
Sengsayadeth has no disclosures.
PHOENIX ‑ A US Department of Veterans Affairs (VA) hospital in Tennessee has performed the first “bloodless” autologous stem cell transplant within the Veterans Health Administration, treating a 61-year-old Jehovah’s Witness patient with multiple myeloma who traveled from California for the procedure.
The case, presented at the annual meeting of the Association of VA Hematology/Oncology, stated that “we should not withhold any therapies for patients who are Jehovah’s Witnesses out of fear of them bleeding out or having complications from anemia,” said Bhagirathbhai Dholaria, MBBS, an associate professor of medicine at Vanderbilt University Medical Center who worked with the VA Tennessee Valley Healthcare System in Nashville.
While Jehovah’s Witnesses accept medical treatment, their faith forbids blood transfusions, including of preoperative autologous blood, due to its interpretation of the Bible. The faith allows individuals to decide whether to accept stem cells collected from their blood or someone else’s “provided that blood components are not intentionally collected, stored, and reinfused along with the stem cells.”
There are an estimated 1.2 million Jehovah’s Witnesses in the US.
Traditional Stem Cell Transplants Require Blood Support
In conventional autologous stem cell transplants for multiple myeloma, high-dose chemotherapy temporarily wipes out the patient’s bone marrow for about 2 to 3 weeks, Dholaria explained. During this period, patients typically receive 2 units of packed red blood cells and platelet transfusions to prevent severe complications from anemia and low platelet counts.
“Because of this reason, Jehovah’s Witnesses have been traditionally denied these therapies,” Dholaria said.
However, bloodless autologous transplants have been performed for about 2 decades, and Vanderbilt University has been offering the procedures for about 3 years, according to Dholaria.
For the first bloodless procedure in the VA, the patient–who had an aggressive, newly diagnosed IgG kappa multiple myeloma–was evaluated.
“He had been treated by local doctors in California. Otherwise, he was actually in really good shape. Physically, he didn’t have any major issues,” Dholaria said. “So, he met the criteria for our bloodless protocol, and we decided to offer him the procedure.”
The team consulted ethics and legal departments and noted the patient’s blood product preferences in his electronic health record. The patient then underwent a preoptimization protocol that included erythropoiesis-stimulating agents, intravenous iron, and vitamin B12 supplementation to boost blood counts before the transplant, according to the case presentation.
Special Protocol Required in ‘Bloodless’ Procedures
After stem cell collection and chemotherapy, patients undergoing bloodless procedures receive aggressive growth factor support to minimize the duration and severity of cytopenia, Dholaria said. As part of the protocol, the care team uses pediatric tubes for blood draws to minimize blood loss and monitors patients closely on cardiac monitors, he added. In addition, blood draws are only performed every 3 days.
“We watch for any cardiac decompensation because these patients have severe anemia for a brief period of time. We make sure they don’t [have a] heart attack or arrhythmias,” Dholaria said. “Or if the platelets are too low, and they start oozing blood from the nose, gums, or gut, that needs to be dealt with accordingly.”
For bleeding complications, the team uses clotting factors and intravenous and oral medications to support remaining platelet function rather than platelet transfusions.
The patient in this case tolerated the transplant “exceptionally well with minimal complications,” according to the case presentation. He achieved full engraftment on day 14 after transplant and was discharged from inpatient care with continued monitoring through day 30.
“The patient was very compliant,” said Salyka Sengsayadeth, MD, medical director of the VA Tennessee Valley Healthcare System Stem Cell Transplant and Cellular Therapy Program and associate professor of medicine at Vanderbilt.
“He tolerated everything that we needed to do,” she said. “He called us when he needed to call us and did everything that we asked and recommended for him.”
The patient’s roughly 30-day hospital stay matched that of typical transplant patients, Sengsayadeth noted. His myeloma responded to treatment, and he returned to California, Dholaria said.
‘Bloodless’ Procedures Not for All Stem Cell Transplants
The case highlights the availability of stem cell transplants in the VA–they are only performed in Seattle and Nashville–and opportunities for patients who wish to avoid blood transfusions. Sengsayadeth said the bloodless protocol is available for patients without religious objections who simply prefer to avoid blood products.
Dholaria cautioned that bloodless protocol applies specifically to autologous transplants, where patients receive their own stem cells. The team does not plan to offer bloodless allogeneic transplants, which use donor stem cells for conditions like leukemia, due to higher risks. In addition, most Jehovah’s Witnesses decline allogeneic transplants because they do not accept stem cells from another person, Dholaria said.
Beyond multiple myeloma, the Tennessee Valley Healthcare System offers bloodless autologous transplants for various blood cancers, including non-Hodgkin lymphomas such as large B-cell lymphoma, follicular lymphoma, and mantle cell lymphoma, as well as lymphomas affecting the brain, Dholaria said.
Clinicians “should start thinking about this early on, as soon as the cancer diagnosis is made, to make the referral and get the patient on our radar,” Dholaria said.
Sengsayadeth said physicians within the VA typically know how to refer appropriate patients to her team. “They just send us an email or give us a call or a message to say ‘I have this patient. Do you think they’re someone I should send to you?’ We usually answer right back, and then we can proceed with the full evaluation if we think that’s a reasonable thing to do.”
‘Treated Like Family’
The patient, a Marine Corps veteran named Keith Cody, spoke about the procedure in a video interview. Cody said he was reluctant at first to undergo the procedure because he didn’t understand what it would accomplish.
“As I was doing the massive chemo every week, and then suffering with the side effects, I decided to ask again about this procedure and how it improves my quality of life,” he said.
At the time of the taping of the video, Cody was getting ready to go home to California. “They’ve told me that I’ll still need more time to get my energy back, but I do feel much better already,” he said.
He also praised the staff. “Everybody that we came across, I enjoyed the interactions. It’s actually sad to leave people behind that you really felt treated you like family.”
Dholaria discloses relationships with Janssen, Angiocrine, Pfizer, Poseida, MEI, Orcabio, Wugen, Allovir, Adicet, BMS, Molecular Templates, Atara, MJH, Arvinas, Janssen, ADC, Gilead, GSK, Caribou, F. Hoffmann-La Roche AG, Autolus, and Pierre Fabre.
Sengsayadeth has no disclosures.
PHOENIX ‑ A US Department of Veterans Affairs (VA) hospital in Tennessee has performed the first “bloodless” autologous stem cell transplant within the Veterans Health Administration, treating a 61-year-old Jehovah’s Witness patient with multiple myeloma who traveled from California for the procedure.
The case, presented at the annual meeting of the Association of VA Hematology/Oncology, stated that “we should not withhold any therapies for patients who are Jehovah’s Witnesses out of fear of them bleeding out or having complications from anemia,” said Bhagirathbhai Dholaria, MBBS, an associate professor of medicine at Vanderbilt University Medical Center who worked with the VA Tennessee Valley Healthcare System in Nashville.
While Jehovah’s Witnesses accept medical treatment, their faith forbids blood transfusions, including of preoperative autologous blood, due to its interpretation of the Bible. The faith allows individuals to decide whether to accept stem cells collected from their blood or someone else’s “provided that blood components are not intentionally collected, stored, and reinfused along with the stem cells.”
There are an estimated 1.2 million Jehovah’s Witnesses in the US.
Traditional Stem Cell Transplants Require Blood Support
In conventional autologous stem cell transplants for multiple myeloma, high-dose chemotherapy temporarily wipes out the patient’s bone marrow for about 2 to 3 weeks, Dholaria explained. During this period, patients typically receive 2 units of packed red blood cells and platelet transfusions to prevent severe complications from anemia and low platelet counts.
“Because of this reason, Jehovah’s Witnesses have been traditionally denied these therapies,” Dholaria said.
However, bloodless autologous transplants have been performed for about 2 decades, and Vanderbilt University has been offering the procedures for about 3 years, according to Dholaria.
For the first bloodless procedure in the VA, the patient–who had an aggressive, newly diagnosed IgG kappa multiple myeloma–was evaluated.
“He had been treated by local doctors in California. Otherwise, he was actually in really good shape. Physically, he didn’t have any major issues,” Dholaria said. “So, he met the criteria for our bloodless protocol, and we decided to offer him the procedure.”
The team consulted ethics and legal departments and noted the patient’s blood product preferences in his electronic health record. The patient then underwent a preoptimization protocol that included erythropoiesis-stimulating agents, intravenous iron, and vitamin B12 supplementation to boost blood counts before the transplant, according to the case presentation.
Special Protocol Required in ‘Bloodless’ Procedures
After stem cell collection and chemotherapy, patients undergoing bloodless procedures receive aggressive growth factor support to minimize the duration and severity of cytopenia, Dholaria said. As part of the protocol, the care team uses pediatric tubes for blood draws to minimize blood loss and monitors patients closely on cardiac monitors, he added. In addition, blood draws are only performed every 3 days.
“We watch for any cardiac decompensation because these patients have severe anemia for a brief period of time. We make sure they don’t [have a] heart attack or arrhythmias,” Dholaria said. “Or if the platelets are too low, and they start oozing blood from the nose, gums, or gut, that needs to be dealt with accordingly.”
For bleeding complications, the team uses clotting factors and intravenous and oral medications to support remaining platelet function rather than platelet transfusions.
The patient in this case tolerated the transplant “exceptionally well with minimal complications,” according to the case presentation. He achieved full engraftment on day 14 after transplant and was discharged from inpatient care with continued monitoring through day 30.
“The patient was very compliant,” said Salyka Sengsayadeth, MD, medical director of the VA Tennessee Valley Healthcare System Stem Cell Transplant and Cellular Therapy Program and associate professor of medicine at Vanderbilt.
“He tolerated everything that we needed to do,” she said. “He called us when he needed to call us and did everything that we asked and recommended for him.”
The patient’s roughly 30-day hospital stay matched that of typical transplant patients, Sengsayadeth noted. His myeloma responded to treatment, and he returned to California, Dholaria said.
‘Bloodless’ Procedures Not for All Stem Cell Transplants
The case highlights the availability of stem cell transplants in the VA–they are only performed in Seattle and Nashville–and opportunities for patients who wish to avoid blood transfusions. Sengsayadeth said the bloodless protocol is available for patients without religious objections who simply prefer to avoid blood products.
Dholaria cautioned that bloodless protocol applies specifically to autologous transplants, where patients receive their own stem cells. The team does not plan to offer bloodless allogeneic transplants, which use donor stem cells for conditions like leukemia, due to higher risks. In addition, most Jehovah’s Witnesses decline allogeneic transplants because they do not accept stem cells from another person, Dholaria said.
Beyond multiple myeloma, the Tennessee Valley Healthcare System offers bloodless autologous transplants for various blood cancers, including non-Hodgkin lymphomas such as large B-cell lymphoma, follicular lymphoma, and mantle cell lymphoma, as well as lymphomas affecting the brain, Dholaria said.
Clinicians “should start thinking about this early on, as soon as the cancer diagnosis is made, to make the referral and get the patient on our radar,” Dholaria said.
Sengsayadeth said physicians within the VA typically know how to refer appropriate patients to her team. “They just send us an email or give us a call or a message to say ‘I have this patient. Do you think they’re someone I should send to you?’ We usually answer right back, and then we can proceed with the full evaluation if we think that’s a reasonable thing to do.”
‘Treated Like Family’
The patient, a Marine Corps veteran named Keith Cody, spoke about the procedure in a video interview. Cody said he was reluctant at first to undergo the procedure because he didn’t understand what it would accomplish.
“As I was doing the massive chemo every week, and then suffering with the side effects, I decided to ask again about this procedure and how it improves my quality of life,” he said.
At the time of the taping of the video, Cody was getting ready to go home to California. “They’ve told me that I’ll still need more time to get my energy back, but I do feel much better already,” he said.
He also praised the staff. “Everybody that we came across, I enjoyed the interactions. It’s actually sad to leave people behind that you really felt treated you like family.”
Dholaria discloses relationships with Janssen, Angiocrine, Pfizer, Poseida, MEI, Orcabio, Wugen, Allovir, Adicet, BMS, Molecular Templates, Atara, MJH, Arvinas, Janssen, ADC, Gilead, GSK, Caribou, F. Hoffmann-La Roche AG, Autolus, and Pierre Fabre.
Sengsayadeth has no disclosures.
An Uncommon Presentation of Marginal Zone Lymphoma Involving the Sciatic Foramen
Background
Marginal zone lymphoma (MZL) is an indolent B-cell non-Hodgkin lymphoma most commonly arising in mucosal, nodal, or splenic tissues. While extranodal presentations are recognized, involvement of the sciatic foramen is exceedingly rare. We present a unique case of stage IV MZL with primary involvement of the left sciatic foramen, identified incidentally during urologic evaluation.
Case Presentation
A 74-year-old male patient was referred for hematologic evaluation after imaging revealed a left sciatic foraminal mass during work-up for elevated PSA. CT abdomen/pelvis revealed a 4.7 cm mass in the left sciatic foramen. Follow-up PET-CT confirmed hypermetabolic activity in the mass, with additional areas of uptake in the right ilium and pleural- pericardial regions. The patient was asymptomatic and denied B-symptoms. CT-guided biopsy of the sciatic mass revealed low-grade B-cell lymphoma. Flow cytometry showed a CD20-positive, CD5-negative, CD10-negative, lambda light chain–restricted population consistent with marginal zone lymphoma. Laboratory studies demonstrated iron deficiency anemia, with otherwise unremarkable counts and chemistries. He was started on monotherapy with rituximab for four cycles. He tolerated treatment well. Interval PET imaging in April 2025 showed stable disease in the sciatic foramen and mild improvement in pleural- pericardial uptake. He is planned to start obinutuzumab in the upcoming month.
Discussion
This case illustrates a rare anatomic presentation of MZL, likely representing primary sciatic foramen involvement. The presence of additional PETavid lesions complicates staging, raising consideration of stage I vs. III/IV disease. Biopsy was limited to the sciatic lesion, and no bone marrow sampling was performed. Given the patient’s excellent performance status, absence of symptoms, and low tumor burden, single-agent rituximab was chosen initially in accordance with NCCN guidelines.
Conclusions
Sciatic foramen involvement by MZL is an extremely rare occurrence and may mimic more common soft tissue or neurogenic tumors radiographically. This case underscores the importance of biopsy for diagnosis and the value of multidisciplinary care. In the veteran population, such incidental findings on imaging warrant comprehensive evaluation, particularly in atypical anatomical sites.
Background
Marginal zone lymphoma (MZL) is an indolent B-cell non-Hodgkin lymphoma most commonly arising in mucosal, nodal, or splenic tissues. While extranodal presentations are recognized, involvement of the sciatic foramen is exceedingly rare. We present a unique case of stage IV MZL with primary involvement of the left sciatic foramen, identified incidentally during urologic evaluation.
Case Presentation
A 74-year-old male patient was referred for hematologic evaluation after imaging revealed a left sciatic foraminal mass during work-up for elevated PSA. CT abdomen/pelvis revealed a 4.7 cm mass in the left sciatic foramen. Follow-up PET-CT confirmed hypermetabolic activity in the mass, with additional areas of uptake in the right ilium and pleural- pericardial regions. The patient was asymptomatic and denied B-symptoms. CT-guided biopsy of the sciatic mass revealed low-grade B-cell lymphoma. Flow cytometry showed a CD20-positive, CD5-negative, CD10-negative, lambda light chain–restricted population consistent with marginal zone lymphoma. Laboratory studies demonstrated iron deficiency anemia, with otherwise unremarkable counts and chemistries. He was started on monotherapy with rituximab for four cycles. He tolerated treatment well. Interval PET imaging in April 2025 showed stable disease in the sciatic foramen and mild improvement in pleural- pericardial uptake. He is planned to start obinutuzumab in the upcoming month.
Discussion
This case illustrates a rare anatomic presentation of MZL, likely representing primary sciatic foramen involvement. The presence of additional PETavid lesions complicates staging, raising consideration of stage I vs. III/IV disease. Biopsy was limited to the sciatic lesion, and no bone marrow sampling was performed. Given the patient’s excellent performance status, absence of symptoms, and low tumor burden, single-agent rituximab was chosen initially in accordance with NCCN guidelines.
Conclusions
Sciatic foramen involvement by MZL is an extremely rare occurrence and may mimic more common soft tissue or neurogenic tumors radiographically. This case underscores the importance of biopsy for diagnosis and the value of multidisciplinary care. In the veteran population, such incidental findings on imaging warrant comprehensive evaluation, particularly in atypical anatomical sites.
Background
Marginal zone lymphoma (MZL) is an indolent B-cell non-Hodgkin lymphoma most commonly arising in mucosal, nodal, or splenic tissues. While extranodal presentations are recognized, involvement of the sciatic foramen is exceedingly rare. We present a unique case of stage IV MZL with primary involvement of the left sciatic foramen, identified incidentally during urologic evaluation.
Case Presentation
A 74-year-old male patient was referred for hematologic evaluation after imaging revealed a left sciatic foraminal mass during work-up for elevated PSA. CT abdomen/pelvis revealed a 4.7 cm mass in the left sciatic foramen. Follow-up PET-CT confirmed hypermetabolic activity in the mass, with additional areas of uptake in the right ilium and pleural- pericardial regions. The patient was asymptomatic and denied B-symptoms. CT-guided biopsy of the sciatic mass revealed low-grade B-cell lymphoma. Flow cytometry showed a CD20-positive, CD5-negative, CD10-negative, lambda light chain–restricted population consistent with marginal zone lymphoma. Laboratory studies demonstrated iron deficiency anemia, with otherwise unremarkable counts and chemistries. He was started on monotherapy with rituximab for four cycles. He tolerated treatment well. Interval PET imaging in April 2025 showed stable disease in the sciatic foramen and mild improvement in pleural- pericardial uptake. He is planned to start obinutuzumab in the upcoming month.
Discussion
This case illustrates a rare anatomic presentation of MZL, likely representing primary sciatic foramen involvement. The presence of additional PETavid lesions complicates staging, raising consideration of stage I vs. III/IV disease. Biopsy was limited to the sciatic lesion, and no bone marrow sampling was performed. Given the patient’s excellent performance status, absence of symptoms, and low tumor burden, single-agent rituximab was chosen initially in accordance with NCCN guidelines.
Conclusions
Sciatic foramen involvement by MZL is an extremely rare occurrence and may mimic more common soft tissue or neurogenic tumors radiographically. This case underscores the importance of biopsy for diagnosis and the value of multidisciplinary care. In the veteran population, such incidental findings on imaging warrant comprehensive evaluation, particularly in atypical anatomical sites.
Study Investigates Non-Hodgkin Lymphoma in Air Force Missileers
Individuals working near intercontinental ballistic missiles (ICBMs), may be at higher risk of developing non-Hodgkin lymphoma (NHL) according to a preprint analysis conducted on missileers at Malmstrom Air Force Base in Montana. The study, which has not undergone peer review, found higher rates of NHL diagnosis at younger ages compared with the general population. The study also found a statistically significant increase in NHL diagnoses among older missileers, with such rates surpassing expected benchmarks.
The findings build on anecdotal and evidentiary data gathered in the last 50-plus years, including from the Torchlight Initiative, established in 2023 to collect self-reported cancer diagnoses and related fatalities from personnel and family members associated with the ICBM community.
The report shows patterns that “warranted a detailed statistical analysis,” leading to a granular examination of the registry and categorization of the data by cancer type, geographical location, and specific demographics. This narrowed the focus to 18 missileers who served at Malmstrom and were diagnosed with NHL.
In 2001, the Air Force Institute for Operational Health did a site evaluation and sampled for potential chemical and biological contaminants at Malmstrom following various reports of cancers from missileers, including 2 who died after being diagnosed with NHL. In a 2005 review, the Air Force said, “there is not sufficient evidence to consider the possibility of a cancer clustering to justify further investigation.”
In 2022, Lt. Col. Daniel Sebeck, a vice commander of Space Delta 8 in Colorado who served at Malmstrom and a close friend and fellow missileer were diagnosed with NHL. Sebeck discovered 36 cancer cases among missileers who had been stationed at Malmstrom. Ten developed NHL, 2 developed Hodgkin lymphoma, and 24 developed another form of cancer. The Air Force has acknowledged the concerns.
In 2023, US Air Force School of Aerospace Medicine (USAFSAM) approved the Missile Community Cancer Study (MCCS) to assess “specific cancer concerns raised by missile community members across related career fields and also examines the possibility of clusters of non-Hodgkin’s lymphoma at intercontinental ballistic missile bases.” The study compares 14 common cancers in the general population with that of missile-related career fields. The USAFSAM is reviewing records from former and current Missile Community members on active duty from 1976-2010, as well as state and national cancer data from multiple registries.
Early results from the MCCS suggested elevated rates of some cancers—mainly breast and prostate cancer—among missileers, maintainers, and other ICMB-related job positions, which aligns with other national cancer data.
At a June 2024 AFGSC town hall, officials announced that missileers would now have their information submitted to the Defense Occupational and Environmental Health Readiness System (DOEHRS), a Pentagon database for reporting occupational and exposure hazards.
"This info from DOEHRS flows into the recently developed Individual Longitudinal Exposure Record, a system that compiles occupational and environmental health data throughout a person's career," Lt. Col. John Severns, a spokesperson for Air Force Global Strike Command, said. DOEHRS, which has tracked Air Force records since 2010, allows US Department of Defense and US Department of Veterans Affairs clinical staff to access the information.
MCCS considers potential PCB exposures an occupational hazard. The Air Force says researchers are working with the System Program Offices and leadership to determine the timeframe of PCB removal from bases.
The lack of incontrovertible evidence connecting workplace toxins to NHL has often stymied patients and their family members from receiving appropriate benefits. An “informal talk” in April led by Rep. Mark Takano (D-CA) and Sen. Richard Blumenthal (D-CT) focused on exposures to hazardous materials at US military bases. Participants included various advocacy groups like the Torchlight Initiative, the Invisible Enemy, and Burn Pits 360.
More than a dozen veterans spoke about serving at military bases where they were exposed to a variety of harmful substances, and issues they faced in receiving coverage. David Crete, a veteran and chairman of The Invisible Enemy, said, “I am asking Congress to please allow us to get the benefits every other veteran earned. We are not asking to be special but to be treated equal.”
Rep. Takano called for greater focus on toxic exposures at US military bases: “We must push back against the idea that service members are only in harm’s way in war zones.”
Individuals working near intercontinental ballistic missiles (ICBMs), may be at higher risk of developing non-Hodgkin lymphoma (NHL) according to a preprint analysis conducted on missileers at Malmstrom Air Force Base in Montana. The study, which has not undergone peer review, found higher rates of NHL diagnosis at younger ages compared with the general population. The study also found a statistically significant increase in NHL diagnoses among older missileers, with such rates surpassing expected benchmarks.
The findings build on anecdotal and evidentiary data gathered in the last 50-plus years, including from the Torchlight Initiative, established in 2023 to collect self-reported cancer diagnoses and related fatalities from personnel and family members associated with the ICBM community.
The report shows patterns that “warranted a detailed statistical analysis,” leading to a granular examination of the registry and categorization of the data by cancer type, geographical location, and specific demographics. This narrowed the focus to 18 missileers who served at Malmstrom and were diagnosed with NHL.
In 2001, the Air Force Institute for Operational Health did a site evaluation and sampled for potential chemical and biological contaminants at Malmstrom following various reports of cancers from missileers, including 2 who died after being diagnosed with NHL. In a 2005 review, the Air Force said, “there is not sufficient evidence to consider the possibility of a cancer clustering to justify further investigation.”
In 2022, Lt. Col. Daniel Sebeck, a vice commander of Space Delta 8 in Colorado who served at Malmstrom and a close friend and fellow missileer were diagnosed with NHL. Sebeck discovered 36 cancer cases among missileers who had been stationed at Malmstrom. Ten developed NHL, 2 developed Hodgkin lymphoma, and 24 developed another form of cancer. The Air Force has acknowledged the concerns.
In 2023, US Air Force School of Aerospace Medicine (USAFSAM) approved the Missile Community Cancer Study (MCCS) to assess “specific cancer concerns raised by missile community members across related career fields and also examines the possibility of clusters of non-Hodgkin’s lymphoma at intercontinental ballistic missile bases.” The study compares 14 common cancers in the general population with that of missile-related career fields. The USAFSAM is reviewing records from former and current Missile Community members on active duty from 1976-2010, as well as state and national cancer data from multiple registries.
Early results from the MCCS suggested elevated rates of some cancers—mainly breast and prostate cancer—among missileers, maintainers, and other ICMB-related job positions, which aligns with other national cancer data.
At a June 2024 AFGSC town hall, officials announced that missileers would now have their information submitted to the Defense Occupational and Environmental Health Readiness System (DOEHRS), a Pentagon database for reporting occupational and exposure hazards.
"This info from DOEHRS flows into the recently developed Individual Longitudinal Exposure Record, a system that compiles occupational and environmental health data throughout a person's career," Lt. Col. John Severns, a spokesperson for Air Force Global Strike Command, said. DOEHRS, which has tracked Air Force records since 2010, allows US Department of Defense and US Department of Veterans Affairs clinical staff to access the information.
MCCS considers potential PCB exposures an occupational hazard. The Air Force says researchers are working with the System Program Offices and leadership to determine the timeframe of PCB removal from bases.
The lack of incontrovertible evidence connecting workplace toxins to NHL has often stymied patients and their family members from receiving appropriate benefits. An “informal talk” in April led by Rep. Mark Takano (D-CA) and Sen. Richard Blumenthal (D-CT) focused on exposures to hazardous materials at US military bases. Participants included various advocacy groups like the Torchlight Initiative, the Invisible Enemy, and Burn Pits 360.
More than a dozen veterans spoke about serving at military bases where they were exposed to a variety of harmful substances, and issues they faced in receiving coverage. David Crete, a veteran and chairman of The Invisible Enemy, said, “I am asking Congress to please allow us to get the benefits every other veteran earned. We are not asking to be special but to be treated equal.”
Rep. Takano called for greater focus on toxic exposures at US military bases: “We must push back against the idea that service members are only in harm’s way in war zones.”
Individuals working near intercontinental ballistic missiles (ICBMs), may be at higher risk of developing non-Hodgkin lymphoma (NHL) according to a preprint analysis conducted on missileers at Malmstrom Air Force Base in Montana. The study, which has not undergone peer review, found higher rates of NHL diagnosis at younger ages compared with the general population. The study also found a statistically significant increase in NHL diagnoses among older missileers, with such rates surpassing expected benchmarks.
The findings build on anecdotal and evidentiary data gathered in the last 50-plus years, including from the Torchlight Initiative, established in 2023 to collect self-reported cancer diagnoses and related fatalities from personnel and family members associated with the ICBM community.
The report shows patterns that “warranted a detailed statistical analysis,” leading to a granular examination of the registry and categorization of the data by cancer type, geographical location, and specific demographics. This narrowed the focus to 18 missileers who served at Malmstrom and were diagnosed with NHL.
In 2001, the Air Force Institute for Operational Health did a site evaluation and sampled for potential chemical and biological contaminants at Malmstrom following various reports of cancers from missileers, including 2 who died after being diagnosed with NHL. In a 2005 review, the Air Force said, “there is not sufficient evidence to consider the possibility of a cancer clustering to justify further investigation.”
In 2022, Lt. Col. Daniel Sebeck, a vice commander of Space Delta 8 in Colorado who served at Malmstrom and a close friend and fellow missileer were diagnosed with NHL. Sebeck discovered 36 cancer cases among missileers who had been stationed at Malmstrom. Ten developed NHL, 2 developed Hodgkin lymphoma, and 24 developed another form of cancer. The Air Force has acknowledged the concerns.
In 2023, US Air Force School of Aerospace Medicine (USAFSAM) approved the Missile Community Cancer Study (MCCS) to assess “specific cancer concerns raised by missile community members across related career fields and also examines the possibility of clusters of non-Hodgkin’s lymphoma at intercontinental ballistic missile bases.” The study compares 14 common cancers in the general population with that of missile-related career fields. The USAFSAM is reviewing records from former and current Missile Community members on active duty from 1976-2010, as well as state and national cancer data from multiple registries.
Early results from the MCCS suggested elevated rates of some cancers—mainly breast and prostate cancer—among missileers, maintainers, and other ICMB-related job positions, which aligns with other national cancer data.
At a June 2024 AFGSC town hall, officials announced that missileers would now have their information submitted to the Defense Occupational and Environmental Health Readiness System (DOEHRS), a Pentagon database for reporting occupational and exposure hazards.
"This info from DOEHRS flows into the recently developed Individual Longitudinal Exposure Record, a system that compiles occupational and environmental health data throughout a person's career," Lt. Col. John Severns, a spokesperson for Air Force Global Strike Command, said. DOEHRS, which has tracked Air Force records since 2010, allows US Department of Defense and US Department of Veterans Affairs clinical staff to access the information.
MCCS considers potential PCB exposures an occupational hazard. The Air Force says researchers are working with the System Program Offices and leadership to determine the timeframe of PCB removal from bases.
The lack of incontrovertible evidence connecting workplace toxins to NHL has often stymied patients and their family members from receiving appropriate benefits. An “informal talk” in April led by Rep. Mark Takano (D-CA) and Sen. Richard Blumenthal (D-CT) focused on exposures to hazardous materials at US military bases. Participants included various advocacy groups like the Torchlight Initiative, the Invisible Enemy, and Burn Pits 360.
More than a dozen veterans spoke about serving at military bases where they were exposed to a variety of harmful substances, and issues they faced in receiving coverage. David Crete, a veteran and chairman of The Invisible Enemy, said, “I am asking Congress to please allow us to get the benefits every other veteran earned. We are not asking to be special but to be treated equal.”
Rep. Takano called for greater focus on toxic exposures at US military bases: “We must push back against the idea that service members are only in harm’s way in war zones.”
FL: Tafasitamab Plus Combo Boosts Outcomes
“This study is the first to validate combining two monoclonal antibodies (anti-CD19 with anti-CD20) in the treatment of lymphoma,” said first author Laurie H. Sehn, MD, MPH, a clinical professor with the BC Cancer Centre for Lymphoid Cancer and the University of British Columbia in Vancouver, Canada. She presented the findings at the American Society of Hematology 2024 Annual Meeting.
“Tafasitamab plus lenalidomide and rituximab can be administered in community as well as academic settings and represents a potential new standard-of-care option for patients with R/R follicular lymphoma,” she noted.
The results are from the international, phase 3 inMIND multicenter trial involving 548 patients with R/R FL, with a median age of 64. Of the patients, 45% had been treated with two or more prior lines of therapy.
At a median follow up of 14.1 months, those randomized to treatment with tafasitamab and len+R (n = 273) had a significantly lower risk for progression, relapse, or death than those receiving the double therapy with placebo (n = 275), with a median progression-free survival of 22.4 months vs 13.9 months, respectively (hazard ratio [HR], 0.43; P < .0001).
Improved progression-free survival was observed across all prespecified subgroups, including patients with disease progression within 24 months, those who were refractory to prior anti-CD20 monoclonal antibodies, and who received multiple prior lines of therapy.
“Although this study was not powered for individual subgroups, it is clear that there’s a significant benefit of tafasitamab in all subgroups regardless of status of [disease progression within 24 months] and regardless of refractoriness to prior anti-CD20 monoclonal antibodies,” Sehn noted.
FL, the most common, indolent form of B-cell non-Hodgkin lymphoma (NHL), is commonly treated with frontline therapy of chemoimmunotherapy; however, response durations begin to dwindle after successive lines of treatment.
Lenalidomide and rituximab are approved and commonly used in the treatment of FL after more than one prior line of treatment.
With tafasitamab, which is administered intravenously, already having been approved for use in combination with lenalidomide in the treatment of R/R diffuse large B-cell lymphoma, based on results of the previous L-MIND study, Sehn and colleagues investigated its benefits in FL or marginal zone NHL.
Of the patients included in the trial, 55% were men and 79% had intermediate or high-risk FLIPI scores, referring to the FL International Prognostic Index, a scoring system used for predicting prognoses of patients with FL.
Treatment in the study consisted of 12 mg/kg intravenous tafasitamab or placebo on days 1, 8, 15, and 22 of cycles 1-3 and days 1 and 15 of cycles 4-12, with standard dosing of len+R for up to 12 cycles of 28 days each.
In terms of the data cutoff, patients in the tafasitamab arm received a median of 12 cycles of treatment vs 11 cycles for placebo, and 19% and 15% were still on treatment, respectively, at the cut-off.
Of the patients, 81% and 84%, respectively, had discontinued treatment primarily because of treatment completion (54% and 43%) or disease progression (11% and 31%).
In addition to the progression-free survival primary endpoint, the tafasitamab arm also had a higher rate of complete response (CR) on PET (49.4% vs 39.8%; P = .029) and higher overall response rate (83.5% vs 72.4%; P = .0014) than the placebo arm.
The duration of response was also higher with tafasitamab (median, 21.2 months vs 13.6 months; HR, 0.47; P < .0001).
Overall survival data, though immature, also favored tafasitamab (HR, 0.59).
The two arms had similar rates of treatment-emergent adverse events, with similar toxicity profiles and no new significant safety signals related to tafasitamab. The rates of discontinuations and dose reductions were similar, with a median dose intensity of 86% with tafasitamab vs 87%.
The most common grade 3 or 4 adverse events were similar between the tafasitamab and placebo groups, including neutropenia (40% vs 38%, respectively), pneumonia (8% vs 5%), thrombocytopenia (6% vs 7%), decreased neutrophils (6% vs 7%), COVID-19 (6% vs 2%), and COVID-19 pneumonia (5% vs 1%).
“Importantly, the addition of tafasitamab did not impede the delivery of lenalidomide and rituximab, with similar observed dose discontinuations or interruptions in both cohorts,” Sehn said.
“The inMIND phase 3 study met its primary endpoint of improved progression-free survival with the addition of tafasitamab to lenalidomide and rituximab in patients with R/R follicular lymphoma, representing a 57% reduction in the risk of progression, relapse, or death,” Sehn said.
Commenting on the study, Juan Pablo Alderuccio, MD, an associate professor of medicine, Division of Hematology in the Sylvester Comprehensive Cancer Center, University of Miami Miller School of Medicine in Florida, said the findings are notable in that “this is the first time two monoclonal antibodies have been tested in follicular lymphoma.”
“The study demonstrates that simultaneously targeting CD19 and CD20 improves outcomes,” he said in an interview.
Alderuccio noted the key caveats include that “PET/CT complete response correlates well with survival in follicular lymphoma. In this study, the PET/CT CR rate was 49.4%, underscoring the need for longer follow-ups to better assess those responses’ durability.”
“Another caveat of this regimen is the treatment schedule, which requires weekly tafasitamab infusions during cycles 1-3 and every 2 weeks during cycles 4-12. This is associated with rituximab and lenalidomide administration,” he said.
Ultimately, “the results underscore the potential of tafasitamab in combination with lenalidomide and rituximab to become a new treatment option in the second-line or later follicular lymphoma.”
“However, I would like to see more follow-up data before considering it a new standard of care,” he cautioned.
The study was funded by Incyte. Sehn reported ties with AbbVie, Amgen, AstraZeneca, BeiGene, BMS/Celgene, Genmab, Kite/Gilead, Incyte, Janssen, and Merck. Alderuccio disclosed relationships with Genmab, ADC Therapeutics, BeiGene, AbbVie, Genentech, Novartis, Regeneron, and Lilly.
A version of this article appeared on Medscape.com.
“This study is the first to validate combining two monoclonal antibodies (anti-CD19 with anti-CD20) in the treatment of lymphoma,” said first author Laurie H. Sehn, MD, MPH, a clinical professor with the BC Cancer Centre for Lymphoid Cancer and the University of British Columbia in Vancouver, Canada. She presented the findings at the American Society of Hematology 2024 Annual Meeting.
“Tafasitamab plus lenalidomide and rituximab can be administered in community as well as academic settings and represents a potential new standard-of-care option for patients with R/R follicular lymphoma,” she noted.
The results are from the international, phase 3 inMIND multicenter trial involving 548 patients with R/R FL, with a median age of 64. Of the patients, 45% had been treated with two or more prior lines of therapy.
At a median follow up of 14.1 months, those randomized to treatment with tafasitamab and len+R (n = 273) had a significantly lower risk for progression, relapse, or death than those receiving the double therapy with placebo (n = 275), with a median progression-free survival of 22.4 months vs 13.9 months, respectively (hazard ratio [HR], 0.43; P < .0001).
Improved progression-free survival was observed across all prespecified subgroups, including patients with disease progression within 24 months, those who were refractory to prior anti-CD20 monoclonal antibodies, and who received multiple prior lines of therapy.
“Although this study was not powered for individual subgroups, it is clear that there’s a significant benefit of tafasitamab in all subgroups regardless of status of [disease progression within 24 months] and regardless of refractoriness to prior anti-CD20 monoclonal antibodies,” Sehn noted.
FL, the most common, indolent form of B-cell non-Hodgkin lymphoma (NHL), is commonly treated with frontline therapy of chemoimmunotherapy; however, response durations begin to dwindle after successive lines of treatment.
Lenalidomide and rituximab are approved and commonly used in the treatment of FL after more than one prior line of treatment.
With tafasitamab, which is administered intravenously, already having been approved for use in combination with lenalidomide in the treatment of R/R diffuse large B-cell lymphoma, based on results of the previous L-MIND study, Sehn and colleagues investigated its benefits in FL or marginal zone NHL.
Of the patients included in the trial, 55% were men and 79% had intermediate or high-risk FLIPI scores, referring to the FL International Prognostic Index, a scoring system used for predicting prognoses of patients with FL.
Treatment in the study consisted of 12 mg/kg intravenous tafasitamab or placebo on days 1, 8, 15, and 22 of cycles 1-3 and days 1 and 15 of cycles 4-12, with standard dosing of len+R for up to 12 cycles of 28 days each.
In terms of the data cutoff, patients in the tafasitamab arm received a median of 12 cycles of treatment vs 11 cycles for placebo, and 19% and 15% were still on treatment, respectively, at the cut-off.
Of the patients, 81% and 84%, respectively, had discontinued treatment primarily because of treatment completion (54% and 43%) or disease progression (11% and 31%).
In addition to the progression-free survival primary endpoint, the tafasitamab arm also had a higher rate of complete response (CR) on PET (49.4% vs 39.8%; P = .029) and higher overall response rate (83.5% vs 72.4%; P = .0014) than the placebo arm.
The duration of response was also higher with tafasitamab (median, 21.2 months vs 13.6 months; HR, 0.47; P < .0001).
Overall survival data, though immature, also favored tafasitamab (HR, 0.59).
The two arms had similar rates of treatment-emergent adverse events, with similar toxicity profiles and no new significant safety signals related to tafasitamab. The rates of discontinuations and dose reductions were similar, with a median dose intensity of 86% with tafasitamab vs 87%.
The most common grade 3 or 4 adverse events were similar between the tafasitamab and placebo groups, including neutropenia (40% vs 38%, respectively), pneumonia (8% vs 5%), thrombocytopenia (6% vs 7%), decreased neutrophils (6% vs 7%), COVID-19 (6% vs 2%), and COVID-19 pneumonia (5% vs 1%).
“Importantly, the addition of tafasitamab did not impede the delivery of lenalidomide and rituximab, with similar observed dose discontinuations or interruptions in both cohorts,” Sehn said.
“The inMIND phase 3 study met its primary endpoint of improved progression-free survival with the addition of tafasitamab to lenalidomide and rituximab in patients with R/R follicular lymphoma, representing a 57% reduction in the risk of progression, relapse, or death,” Sehn said.
Commenting on the study, Juan Pablo Alderuccio, MD, an associate professor of medicine, Division of Hematology in the Sylvester Comprehensive Cancer Center, University of Miami Miller School of Medicine in Florida, said the findings are notable in that “this is the first time two monoclonal antibodies have been tested in follicular lymphoma.”
“The study demonstrates that simultaneously targeting CD19 and CD20 improves outcomes,” he said in an interview.
Alderuccio noted the key caveats include that “PET/CT complete response correlates well with survival in follicular lymphoma. In this study, the PET/CT CR rate was 49.4%, underscoring the need for longer follow-ups to better assess those responses’ durability.”
“Another caveat of this regimen is the treatment schedule, which requires weekly tafasitamab infusions during cycles 1-3 and every 2 weeks during cycles 4-12. This is associated with rituximab and lenalidomide administration,” he said.
Ultimately, “the results underscore the potential of tafasitamab in combination with lenalidomide and rituximab to become a new treatment option in the second-line or later follicular lymphoma.”
“However, I would like to see more follow-up data before considering it a new standard of care,” he cautioned.
The study was funded by Incyte. Sehn reported ties with AbbVie, Amgen, AstraZeneca, BeiGene, BMS/Celgene, Genmab, Kite/Gilead, Incyte, Janssen, and Merck. Alderuccio disclosed relationships with Genmab, ADC Therapeutics, BeiGene, AbbVie, Genentech, Novartis, Regeneron, and Lilly.
A version of this article appeared on Medscape.com.
“This study is the first to validate combining two monoclonal antibodies (anti-CD19 with anti-CD20) in the treatment of lymphoma,” said first author Laurie H. Sehn, MD, MPH, a clinical professor with the BC Cancer Centre for Lymphoid Cancer and the University of British Columbia in Vancouver, Canada. She presented the findings at the American Society of Hematology 2024 Annual Meeting.
“Tafasitamab plus lenalidomide and rituximab can be administered in community as well as academic settings and represents a potential new standard-of-care option for patients with R/R follicular lymphoma,” she noted.
The results are from the international, phase 3 inMIND multicenter trial involving 548 patients with R/R FL, with a median age of 64. Of the patients, 45% had been treated with two or more prior lines of therapy.
At a median follow up of 14.1 months, those randomized to treatment with tafasitamab and len+R (n = 273) had a significantly lower risk for progression, relapse, or death than those receiving the double therapy with placebo (n = 275), with a median progression-free survival of 22.4 months vs 13.9 months, respectively (hazard ratio [HR], 0.43; P < .0001).
Improved progression-free survival was observed across all prespecified subgroups, including patients with disease progression within 24 months, those who were refractory to prior anti-CD20 monoclonal antibodies, and who received multiple prior lines of therapy.
“Although this study was not powered for individual subgroups, it is clear that there’s a significant benefit of tafasitamab in all subgroups regardless of status of [disease progression within 24 months] and regardless of refractoriness to prior anti-CD20 monoclonal antibodies,” Sehn noted.
FL, the most common, indolent form of B-cell non-Hodgkin lymphoma (NHL), is commonly treated with frontline therapy of chemoimmunotherapy; however, response durations begin to dwindle after successive lines of treatment.
Lenalidomide and rituximab are approved and commonly used in the treatment of FL after more than one prior line of treatment.
With tafasitamab, which is administered intravenously, already having been approved for use in combination with lenalidomide in the treatment of R/R diffuse large B-cell lymphoma, based on results of the previous L-MIND study, Sehn and colleagues investigated its benefits in FL or marginal zone NHL.
Of the patients included in the trial, 55% were men and 79% had intermediate or high-risk FLIPI scores, referring to the FL International Prognostic Index, a scoring system used for predicting prognoses of patients with FL.
Treatment in the study consisted of 12 mg/kg intravenous tafasitamab or placebo on days 1, 8, 15, and 22 of cycles 1-3 and days 1 and 15 of cycles 4-12, with standard dosing of len+R for up to 12 cycles of 28 days each.
In terms of the data cutoff, patients in the tafasitamab arm received a median of 12 cycles of treatment vs 11 cycles for placebo, and 19% and 15% were still on treatment, respectively, at the cut-off.
Of the patients, 81% and 84%, respectively, had discontinued treatment primarily because of treatment completion (54% and 43%) or disease progression (11% and 31%).
In addition to the progression-free survival primary endpoint, the tafasitamab arm also had a higher rate of complete response (CR) on PET (49.4% vs 39.8%; P = .029) and higher overall response rate (83.5% vs 72.4%; P = .0014) than the placebo arm.
The duration of response was also higher with tafasitamab (median, 21.2 months vs 13.6 months; HR, 0.47; P < .0001).
Overall survival data, though immature, also favored tafasitamab (HR, 0.59).
The two arms had similar rates of treatment-emergent adverse events, with similar toxicity profiles and no new significant safety signals related to tafasitamab. The rates of discontinuations and dose reductions were similar, with a median dose intensity of 86% with tafasitamab vs 87%.
The most common grade 3 or 4 adverse events were similar between the tafasitamab and placebo groups, including neutropenia (40% vs 38%, respectively), pneumonia (8% vs 5%), thrombocytopenia (6% vs 7%), decreased neutrophils (6% vs 7%), COVID-19 (6% vs 2%), and COVID-19 pneumonia (5% vs 1%).
“Importantly, the addition of tafasitamab did not impede the delivery of lenalidomide and rituximab, with similar observed dose discontinuations or interruptions in both cohorts,” Sehn said.
“The inMIND phase 3 study met its primary endpoint of improved progression-free survival with the addition of tafasitamab to lenalidomide and rituximab in patients with R/R follicular lymphoma, representing a 57% reduction in the risk of progression, relapse, or death,” Sehn said.
Commenting on the study, Juan Pablo Alderuccio, MD, an associate professor of medicine, Division of Hematology in the Sylvester Comprehensive Cancer Center, University of Miami Miller School of Medicine in Florida, said the findings are notable in that “this is the first time two monoclonal antibodies have been tested in follicular lymphoma.”
“The study demonstrates that simultaneously targeting CD19 and CD20 improves outcomes,” he said in an interview.
Alderuccio noted the key caveats include that “PET/CT complete response correlates well with survival in follicular lymphoma. In this study, the PET/CT CR rate was 49.4%, underscoring the need for longer follow-ups to better assess those responses’ durability.”
“Another caveat of this regimen is the treatment schedule, which requires weekly tafasitamab infusions during cycles 1-3 and every 2 weeks during cycles 4-12. This is associated with rituximab and lenalidomide administration,” he said.
Ultimately, “the results underscore the potential of tafasitamab in combination with lenalidomide and rituximab to become a new treatment option in the second-line or later follicular lymphoma.”
“However, I would like to see more follow-up data before considering it a new standard of care,” he cautioned.
The study was funded by Incyte. Sehn reported ties with AbbVie, Amgen, AstraZeneca, BeiGene, BMS/Celgene, Genmab, Kite/Gilead, Incyte, Janssen, and Merck. Alderuccio disclosed relationships with Genmab, ADC Therapeutics, BeiGene, AbbVie, Genentech, Novartis, Regeneron, and Lilly.
A version of this article appeared on Medscape.com.
FROM ASH 2024
New Cancer Drugs: Do Patients Prefer Faster Access or Clinical Benefit?
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
FROM THE LANCET ONCOLOGY
POLARIX: Extended Results Confirm Standard of Care for DLBCL
These findings “confirm pola-R-CHP as the standard of care for patients with previously untreated intermediate- or high-risk DLBCL,” said lead investigator Gilles Salles, MD, PhD, who presented the extended results at the American Society of Hematology (ASH) 2024 Annual Meeting.
Pola-R-CHP is a modified version of the R-CHOP regimen (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) in which vincristine is replaced with polatuzumab vedotin. The modified regimen was approved by the Food and Drug Administration in April 2023 on the basis of 2-year results from the same study.
In POLARIX, researchers randomized 879 previously untreated adult patients with CD20-positive DLBCL (median age, 66 years) to six to eight 21-day cycles of treatment with either pola-R-CHP or R-CHOP. The primary endpoint was progression-free survival, and secondary endpoints included complete remission and overall survival.
The initial 2-year results — progression-free survival of 76.7% for pola-R-CHP vs 70.2% for R-CHOP — “were the first in 20 years” showing benefits for patients with DLBCL, said Salles, from Memorial Sloan Kettering Cancer Center, New York City.
In the current analysis, there were 879 patients in the global intention-to-treat population — 440 who received pola-R-CHP and 439 who received R-CHOP — and 873 in the safety analysis.
At the 5-year follow-up, Salles and colleagues observed a sustained and significant progression-free survival benefit in patients treated with pola-R-CHP, compared with those who received R-CHOP (64.9% vs 59.1%; hazard ratio [HR], 0.77).
Patients receiving the modified regimen also demonstrated significantly higher rates of complete remission at 5 years (71.8% vs 66.5%; HR, 0.75). These data tell us that for patients who reach complete remission at the end of treatment, the vast majority are still in remission at 5 years, said Salles.
For patients who did progress, those treated with pola-R-CHP had fewer subsequent therapies than those treated with R-CHOP. This included radiotherapy (9.5% vs 14.1%), systemic therapy (20.0% vs 28.2%), platinum-based therapy (9.8% vs 15.5%), stem cell transplant (5.0% vs 8.4%), chimeric antigen receptor T-cell therapy (2.3% vs 4.1%), and bispecifics (1.4% vs 2.1%).
The 5-year overall survival, a secondary endpoint, was numerically better but not significantly so among patients treated with pola-R-CHP (82.3% vs 79.5%; HR, 0.85).
In a competing risk analysis, the cumulative incidence of lymphoma-related deaths at 5 years was 9.1% with pola-R-CHP vs 12.2% with R-CHOP. The probability of non-lymphoma–related deaths, including death due to study treatment, was similar between the two groups at 5 years — 8.56% with pola-R-CHP vs 8.93% with R-CHOP.
An exploratory analysis of progression-free survival and overall survival in subgroups of the patient population, including those with high-risk disease, showed a trend in favor of pola-R-CHP in almost all cases, but Salles warned that the analysis was underpowered.
“I think that interpreting this data to decide the care of the patient should be done very cautiously,” he said.
The researchers did not observe any new safety signals, with pola-R-CHP continuing to show a favorable benefit-risk profile. Looking at adverse events of all grades, Salles noted slightly less peripheral neuropathy with pola-R-CHP (50.3% vs 52.4%), slightly more infection (47.9% vs 44.0%), as well as a favorable trend for pola-R-CHP regarding cardiac arrhythmia (3.6% vs 5.2%) and carcinogenicity (1.0% vs 2.4%), and “very slight and tiny differences” in neutropenia (48.5% vs 45.8%), anemia (33.3% vs 30.1%), and thrombocytopenia (18.0% vs 17.3%).
While the researchers concluded that the study findings confirm pola-R-CHP as the standard of care for patients with previously untreated intermediate- or high-risk DLBCL, Ajay Major, MD, had some reservations.
The results confirm “a small but significant benefit” in progression-free survival and no significant difference in overall survival, but there is some question as to whether pola-R-CHP is appropriate for all-comers with intermediate- or high-risk DLBCL, noted Major, a lymphoma specialist and assistant professor of medicine at the University of Colorado School of Medicine, Aurora, who was not involved in this analysis.
“The subgroup analysis in POLARIX for efficacy of pola-R-CHP based on cell of origin is not adequately powered, and further dedicated on this question studies are needed,” Major said in an interview.
One study presented at the meeting by Major’s group concluded that cell of origin is a strong predictor of the activity of pola-containing therapy in patients with relapsed/refractory large B-cell lymphoma, while another analysis of POLARIX showed progression-free survival and overall survival benefits of pola-R-CHP over R-CHOP in patients with the activated B-cell subtype of DLBCL but not those with the germinal center B-cell subtype, Major noted.
“I think many oncologists will extrapolate from these other data sources and preferentially use pola-R-CHP in patients with activated B-cell or non-germinal center DLBCL,” he predicted.
The study was funded by F. Hoffmann–La Roche/Genentech. Salles reported disclosures with the following companies: BeiGene (consultancy), AbbVie (consultancy, research funding), Genentech/Roche (consultancy, research funding), Incyte (consultancy), and BMS/Celgene (consultancy), among others. Major reported conflicts of interest with Roche/Genentech (consultancy), GSK (research funding), and Incyte (research funding).
A version of this article appeared on Medscape.com.
These findings “confirm pola-R-CHP as the standard of care for patients with previously untreated intermediate- or high-risk DLBCL,” said lead investigator Gilles Salles, MD, PhD, who presented the extended results at the American Society of Hematology (ASH) 2024 Annual Meeting.
Pola-R-CHP is a modified version of the R-CHOP regimen (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) in which vincristine is replaced with polatuzumab vedotin. The modified regimen was approved by the Food and Drug Administration in April 2023 on the basis of 2-year results from the same study.
In POLARIX, researchers randomized 879 previously untreated adult patients with CD20-positive DLBCL (median age, 66 years) to six to eight 21-day cycles of treatment with either pola-R-CHP or R-CHOP. The primary endpoint was progression-free survival, and secondary endpoints included complete remission and overall survival.
The initial 2-year results — progression-free survival of 76.7% for pola-R-CHP vs 70.2% for R-CHOP — “were the first in 20 years” showing benefits for patients with DLBCL, said Salles, from Memorial Sloan Kettering Cancer Center, New York City.
In the current analysis, there were 879 patients in the global intention-to-treat population — 440 who received pola-R-CHP and 439 who received R-CHOP — and 873 in the safety analysis.
At the 5-year follow-up, Salles and colleagues observed a sustained and significant progression-free survival benefit in patients treated with pola-R-CHP, compared with those who received R-CHOP (64.9% vs 59.1%; hazard ratio [HR], 0.77).
Patients receiving the modified regimen also demonstrated significantly higher rates of complete remission at 5 years (71.8% vs 66.5%; HR, 0.75). These data tell us that for patients who reach complete remission at the end of treatment, the vast majority are still in remission at 5 years, said Salles.
For patients who did progress, those treated with pola-R-CHP had fewer subsequent therapies than those treated with R-CHOP. This included radiotherapy (9.5% vs 14.1%), systemic therapy (20.0% vs 28.2%), platinum-based therapy (9.8% vs 15.5%), stem cell transplant (5.0% vs 8.4%), chimeric antigen receptor T-cell therapy (2.3% vs 4.1%), and bispecifics (1.4% vs 2.1%).
The 5-year overall survival, a secondary endpoint, was numerically better but not significantly so among patients treated with pola-R-CHP (82.3% vs 79.5%; HR, 0.85).
In a competing risk analysis, the cumulative incidence of lymphoma-related deaths at 5 years was 9.1% with pola-R-CHP vs 12.2% with R-CHOP. The probability of non-lymphoma–related deaths, including death due to study treatment, was similar between the two groups at 5 years — 8.56% with pola-R-CHP vs 8.93% with R-CHOP.
An exploratory analysis of progression-free survival and overall survival in subgroups of the patient population, including those with high-risk disease, showed a trend in favor of pola-R-CHP in almost all cases, but Salles warned that the analysis was underpowered.
“I think that interpreting this data to decide the care of the patient should be done very cautiously,” he said.
The researchers did not observe any new safety signals, with pola-R-CHP continuing to show a favorable benefit-risk profile. Looking at adverse events of all grades, Salles noted slightly less peripheral neuropathy with pola-R-CHP (50.3% vs 52.4%), slightly more infection (47.9% vs 44.0%), as well as a favorable trend for pola-R-CHP regarding cardiac arrhythmia (3.6% vs 5.2%) and carcinogenicity (1.0% vs 2.4%), and “very slight and tiny differences” in neutropenia (48.5% vs 45.8%), anemia (33.3% vs 30.1%), and thrombocytopenia (18.0% vs 17.3%).
While the researchers concluded that the study findings confirm pola-R-CHP as the standard of care for patients with previously untreated intermediate- or high-risk DLBCL, Ajay Major, MD, had some reservations.
The results confirm “a small but significant benefit” in progression-free survival and no significant difference in overall survival, but there is some question as to whether pola-R-CHP is appropriate for all-comers with intermediate- or high-risk DLBCL, noted Major, a lymphoma specialist and assistant professor of medicine at the University of Colorado School of Medicine, Aurora, who was not involved in this analysis.
“The subgroup analysis in POLARIX for efficacy of pola-R-CHP based on cell of origin is not adequately powered, and further dedicated on this question studies are needed,” Major said in an interview.
One study presented at the meeting by Major’s group concluded that cell of origin is a strong predictor of the activity of pola-containing therapy in patients with relapsed/refractory large B-cell lymphoma, while another analysis of POLARIX showed progression-free survival and overall survival benefits of pola-R-CHP over R-CHOP in patients with the activated B-cell subtype of DLBCL but not those with the germinal center B-cell subtype, Major noted.
“I think many oncologists will extrapolate from these other data sources and preferentially use pola-R-CHP in patients with activated B-cell or non-germinal center DLBCL,” he predicted.
The study was funded by F. Hoffmann–La Roche/Genentech. Salles reported disclosures with the following companies: BeiGene (consultancy), AbbVie (consultancy, research funding), Genentech/Roche (consultancy, research funding), Incyte (consultancy), and BMS/Celgene (consultancy), among others. Major reported conflicts of interest with Roche/Genentech (consultancy), GSK (research funding), and Incyte (research funding).
A version of this article appeared on Medscape.com.
These findings “confirm pola-R-CHP as the standard of care for patients with previously untreated intermediate- or high-risk DLBCL,” said lead investigator Gilles Salles, MD, PhD, who presented the extended results at the American Society of Hematology (ASH) 2024 Annual Meeting.
Pola-R-CHP is a modified version of the R-CHOP regimen (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) in which vincristine is replaced with polatuzumab vedotin. The modified regimen was approved by the Food and Drug Administration in April 2023 on the basis of 2-year results from the same study.
In POLARIX, researchers randomized 879 previously untreated adult patients with CD20-positive DLBCL (median age, 66 years) to six to eight 21-day cycles of treatment with either pola-R-CHP or R-CHOP. The primary endpoint was progression-free survival, and secondary endpoints included complete remission and overall survival.
The initial 2-year results — progression-free survival of 76.7% for pola-R-CHP vs 70.2% for R-CHOP — “were the first in 20 years” showing benefits for patients with DLBCL, said Salles, from Memorial Sloan Kettering Cancer Center, New York City.
In the current analysis, there were 879 patients in the global intention-to-treat population — 440 who received pola-R-CHP and 439 who received R-CHOP — and 873 in the safety analysis.
At the 5-year follow-up, Salles and colleagues observed a sustained and significant progression-free survival benefit in patients treated with pola-R-CHP, compared with those who received R-CHOP (64.9% vs 59.1%; hazard ratio [HR], 0.77).
Patients receiving the modified regimen also demonstrated significantly higher rates of complete remission at 5 years (71.8% vs 66.5%; HR, 0.75). These data tell us that for patients who reach complete remission at the end of treatment, the vast majority are still in remission at 5 years, said Salles.
For patients who did progress, those treated with pola-R-CHP had fewer subsequent therapies than those treated with R-CHOP. This included radiotherapy (9.5% vs 14.1%), systemic therapy (20.0% vs 28.2%), platinum-based therapy (9.8% vs 15.5%), stem cell transplant (5.0% vs 8.4%), chimeric antigen receptor T-cell therapy (2.3% vs 4.1%), and bispecifics (1.4% vs 2.1%).
The 5-year overall survival, a secondary endpoint, was numerically better but not significantly so among patients treated with pola-R-CHP (82.3% vs 79.5%; HR, 0.85).
In a competing risk analysis, the cumulative incidence of lymphoma-related deaths at 5 years was 9.1% with pola-R-CHP vs 12.2% with R-CHOP. The probability of non-lymphoma–related deaths, including death due to study treatment, was similar between the two groups at 5 years — 8.56% with pola-R-CHP vs 8.93% with R-CHOP.
An exploratory analysis of progression-free survival and overall survival in subgroups of the patient population, including those with high-risk disease, showed a trend in favor of pola-R-CHP in almost all cases, but Salles warned that the analysis was underpowered.
“I think that interpreting this data to decide the care of the patient should be done very cautiously,” he said.
The researchers did not observe any new safety signals, with pola-R-CHP continuing to show a favorable benefit-risk profile. Looking at adverse events of all grades, Salles noted slightly less peripheral neuropathy with pola-R-CHP (50.3% vs 52.4%), slightly more infection (47.9% vs 44.0%), as well as a favorable trend for pola-R-CHP regarding cardiac arrhythmia (3.6% vs 5.2%) and carcinogenicity (1.0% vs 2.4%), and “very slight and tiny differences” in neutropenia (48.5% vs 45.8%), anemia (33.3% vs 30.1%), and thrombocytopenia (18.0% vs 17.3%).
While the researchers concluded that the study findings confirm pola-R-CHP as the standard of care for patients with previously untreated intermediate- or high-risk DLBCL, Ajay Major, MD, had some reservations.
The results confirm “a small but significant benefit” in progression-free survival and no significant difference in overall survival, but there is some question as to whether pola-R-CHP is appropriate for all-comers with intermediate- or high-risk DLBCL, noted Major, a lymphoma specialist and assistant professor of medicine at the University of Colorado School of Medicine, Aurora, who was not involved in this analysis.
“The subgroup analysis in POLARIX for efficacy of pola-R-CHP based on cell of origin is not adequately powered, and further dedicated on this question studies are needed,” Major said in an interview.
One study presented at the meeting by Major’s group concluded that cell of origin is a strong predictor of the activity of pola-containing therapy in patients with relapsed/refractory large B-cell lymphoma, while another analysis of POLARIX showed progression-free survival and overall survival benefits of pola-R-CHP over R-CHOP in patients with the activated B-cell subtype of DLBCL but not those with the germinal center B-cell subtype, Major noted.
“I think many oncologists will extrapolate from these other data sources and preferentially use pola-R-CHP in patients with activated B-cell or non-germinal center DLBCL,” he predicted.
The study was funded by F. Hoffmann–La Roche/Genentech. Salles reported disclosures with the following companies: BeiGene (consultancy), AbbVie (consultancy, research funding), Genentech/Roche (consultancy, research funding), Incyte (consultancy), and BMS/Celgene (consultancy), among others. Major reported conflicts of interest with Roche/Genentech (consultancy), GSK (research funding), and Incyte (research funding).
A version of this article appeared on Medscape.com.
FROM ASH 2024
Epcore NHL-1 Update: Treatment Effective Before CAR T
These findings show that epcoritamab — a bispecific antibody therapy available off the shelf and shown in the overall R/R LBCL population in Epcore NHL-1 to be effective following CAR T–cell therapy — can also be administered safely and effectively before CAR T in patients with R/R LBCL.
This is of particular note for patients who may be ineligible for CAR T or whose access to CAR T is limited, first author Yasmin H. Karimi, MD, said in an interview during a poster presentation at the annual American Society of Hematology (ASH) conference.
With over 3 years of follow-up, epcoritamab monotherapy was associated with an overall response rate and complete response rate of 61% and 45%, respectively, in 96 CAR T-naive patients, and side effects were manageable, said Karimi, of the University of Michigan, Ann Arbor, Michigan.
In Epcore NHL-1, epcoritamab — a CD3xCD20 T-cell-engaging bispecific antibody — led to deep and durable responses with manageable toxicity in the patients with R/R LBCL including both CAR T–treated and CAR T–naive patients. The agent was subsequently approved for the treatment of adults with different types of R/R LBCL and follicular lymphoma after at least two prior lines of therapy. Among those who had received CAR T–cell therapy, the ORR/CR rates were 54% and 34%, according to results reported at ASH 2022.
Patients included in the trial were adults with R/R CD20 LBCL, including diffuse LBCL, high-grade B-cell lymphoma, primary mediastinal large B-cell lymphoma, and grade 3B follicular lymphoma with at least two prior lines of systemic antilymphoma therapy. They were treated with two step-up doses of epcoritamab followed by 48-mg full doses in 28-day cycles, including weekly treatment for cycles 1-3, biweekly treatment for cycles 4-9, and treatment every 4 weeks for cycle 10 and any subsequent cycles, until disease progression or unacceptable toxicity.
In the current subgroup analysis of 96 CAR T–naive patients, median duration of response was 18.4 months, median duration of complete response was 28.6 months, and an estimated 46% of patients remained in complete response at 36 months, Karimi said.
Median progression-free survival duration was 4.3 months overall and 33.3 months among complete responders. Median overall survival was 15.4 months and was not reached in complete responders.
At 3 years, an estimated 40% of patients overall and 83% of complete responders had not initiated any other antilymphoma therapy, and of the 35 who received subsequent antilymphoma therapy, 10 (29%) received CAR T. Six of the 10 were alive at data cutoff, and 33 of 74 patients evaluable for minimal residual disease (MRD) were MRD-negative.
The most common treatment-emergent adverse events were cytokine release syndrome (CRS) in 60% of patients, diarrhea in 24%, pyrexia in 23%, fatigue in 22%, neutropenia in 22%, and injection-site reaction in 21%.
Fatal reactions occurred in 18 patients. Of these, nine were related to COVID-19 infection. CRS events were mostly of grade 1 or 2; 4% were of grade 3. CRS occurred most often following cycle 1 and resolved in all but two patients. ICANS occurred in seven patients, clinical tumor lysis syndrome occurred in two patients, and immunoglobulin G levels decreased by a median of about 20% after the story of treatment and remained stable over time, Karimi noted.
“These findings show that epcoritamab can be administered safely and effectively in CAR T–naive or CAR T–exposed patients with R/R LBCL,” she said.
Karimi reported relationships with AstraZeneca, Lily/Loxo, Merck, AbbVie, ADC Therapeutics, Xencor, and Roche/Genentech.
A version of this article first appeared on Medscape.com.
These findings show that epcoritamab — a bispecific antibody therapy available off the shelf and shown in the overall R/R LBCL population in Epcore NHL-1 to be effective following CAR T–cell therapy — can also be administered safely and effectively before CAR T in patients with R/R LBCL.
This is of particular note for patients who may be ineligible for CAR T or whose access to CAR T is limited, first author Yasmin H. Karimi, MD, said in an interview during a poster presentation at the annual American Society of Hematology (ASH) conference.
With over 3 years of follow-up, epcoritamab monotherapy was associated with an overall response rate and complete response rate of 61% and 45%, respectively, in 96 CAR T-naive patients, and side effects were manageable, said Karimi, of the University of Michigan, Ann Arbor, Michigan.
In Epcore NHL-1, epcoritamab — a CD3xCD20 T-cell-engaging bispecific antibody — led to deep and durable responses with manageable toxicity in the patients with R/R LBCL including both CAR T–treated and CAR T–naive patients. The agent was subsequently approved for the treatment of adults with different types of R/R LBCL and follicular lymphoma after at least two prior lines of therapy. Among those who had received CAR T–cell therapy, the ORR/CR rates were 54% and 34%, according to results reported at ASH 2022.
Patients included in the trial were adults with R/R CD20 LBCL, including diffuse LBCL, high-grade B-cell lymphoma, primary mediastinal large B-cell lymphoma, and grade 3B follicular lymphoma with at least two prior lines of systemic antilymphoma therapy. They were treated with two step-up doses of epcoritamab followed by 48-mg full doses in 28-day cycles, including weekly treatment for cycles 1-3, biweekly treatment for cycles 4-9, and treatment every 4 weeks for cycle 10 and any subsequent cycles, until disease progression or unacceptable toxicity.
In the current subgroup analysis of 96 CAR T–naive patients, median duration of response was 18.4 months, median duration of complete response was 28.6 months, and an estimated 46% of patients remained in complete response at 36 months, Karimi said.
Median progression-free survival duration was 4.3 months overall and 33.3 months among complete responders. Median overall survival was 15.4 months and was not reached in complete responders.
At 3 years, an estimated 40% of patients overall and 83% of complete responders had not initiated any other antilymphoma therapy, and of the 35 who received subsequent antilymphoma therapy, 10 (29%) received CAR T. Six of the 10 were alive at data cutoff, and 33 of 74 patients evaluable for minimal residual disease (MRD) were MRD-negative.
The most common treatment-emergent adverse events were cytokine release syndrome (CRS) in 60% of patients, diarrhea in 24%, pyrexia in 23%, fatigue in 22%, neutropenia in 22%, and injection-site reaction in 21%.
Fatal reactions occurred in 18 patients. Of these, nine were related to COVID-19 infection. CRS events were mostly of grade 1 or 2; 4% were of grade 3. CRS occurred most often following cycle 1 and resolved in all but two patients. ICANS occurred in seven patients, clinical tumor lysis syndrome occurred in two patients, and immunoglobulin G levels decreased by a median of about 20% after the story of treatment and remained stable over time, Karimi noted.
“These findings show that epcoritamab can be administered safely and effectively in CAR T–naive or CAR T–exposed patients with R/R LBCL,” she said.
Karimi reported relationships with AstraZeneca, Lily/Loxo, Merck, AbbVie, ADC Therapeutics, Xencor, and Roche/Genentech.
A version of this article first appeared on Medscape.com.
These findings show that epcoritamab — a bispecific antibody therapy available off the shelf and shown in the overall R/R LBCL population in Epcore NHL-1 to be effective following CAR T–cell therapy — can also be administered safely and effectively before CAR T in patients with R/R LBCL.
This is of particular note for patients who may be ineligible for CAR T or whose access to CAR T is limited, first author Yasmin H. Karimi, MD, said in an interview during a poster presentation at the annual American Society of Hematology (ASH) conference.
With over 3 years of follow-up, epcoritamab monotherapy was associated with an overall response rate and complete response rate of 61% and 45%, respectively, in 96 CAR T-naive patients, and side effects were manageable, said Karimi, of the University of Michigan, Ann Arbor, Michigan.
In Epcore NHL-1, epcoritamab — a CD3xCD20 T-cell-engaging bispecific antibody — led to deep and durable responses with manageable toxicity in the patients with R/R LBCL including both CAR T–treated and CAR T–naive patients. The agent was subsequently approved for the treatment of adults with different types of R/R LBCL and follicular lymphoma after at least two prior lines of therapy. Among those who had received CAR T–cell therapy, the ORR/CR rates were 54% and 34%, according to results reported at ASH 2022.
Patients included in the trial were adults with R/R CD20 LBCL, including diffuse LBCL, high-grade B-cell lymphoma, primary mediastinal large B-cell lymphoma, and grade 3B follicular lymphoma with at least two prior lines of systemic antilymphoma therapy. They were treated with two step-up doses of epcoritamab followed by 48-mg full doses in 28-day cycles, including weekly treatment for cycles 1-3, biweekly treatment for cycles 4-9, and treatment every 4 weeks for cycle 10 and any subsequent cycles, until disease progression or unacceptable toxicity.
In the current subgroup analysis of 96 CAR T–naive patients, median duration of response was 18.4 months, median duration of complete response was 28.6 months, and an estimated 46% of patients remained in complete response at 36 months, Karimi said.
Median progression-free survival duration was 4.3 months overall and 33.3 months among complete responders. Median overall survival was 15.4 months and was not reached in complete responders.
At 3 years, an estimated 40% of patients overall and 83% of complete responders had not initiated any other antilymphoma therapy, and of the 35 who received subsequent antilymphoma therapy, 10 (29%) received CAR T. Six of the 10 were alive at data cutoff, and 33 of 74 patients evaluable for minimal residual disease (MRD) were MRD-negative.
The most common treatment-emergent adverse events were cytokine release syndrome (CRS) in 60% of patients, diarrhea in 24%, pyrexia in 23%, fatigue in 22%, neutropenia in 22%, and injection-site reaction in 21%.
Fatal reactions occurred in 18 patients. Of these, nine were related to COVID-19 infection. CRS events were mostly of grade 1 or 2; 4% were of grade 3. CRS occurred most often following cycle 1 and resolved in all but two patients. ICANS occurred in seven patients, clinical tumor lysis syndrome occurred in two patients, and immunoglobulin G levels decreased by a median of about 20% after the story of treatment and remained stable over time, Karimi noted.
“These findings show that epcoritamab can be administered safely and effectively in CAR T–naive or CAR T–exposed patients with R/R LBCL,” she said.
Karimi reported relationships with AstraZeneca, Lily/Loxo, Merck, AbbVie, ADC Therapeutics, Xencor, and Roche/Genentech.
A version of this article first appeared on Medscape.com.
FROM ASH 2024
High-Fiber Diet Linked to Improved Stem Cell Transplant, GvHD Outcomes
Importantly, the findings suggest standard recommendations for patients of a low-fiber diet following allo-HCT may run counter to the potential benefits.
“Significant decrease of fiber intake during transplantation is detrimental. It’s a lost opportunity to promote a healthy gut microbiome, recover from treatment-related microbiota injury, and protect against GVHD,” first author Jenny Paredes, PhD, a staff scientist at City of Hope National Medical Center in Duarte, California, said in a press statement for the study presented at the American Society of Hematology (ASH) 2024 Annual Meeting.
Although the health benefits of dietary fiber on the gut microbiome are well-documented, the effects have recently been shown to extend to outcomes after allo-HCT in general, with researchers finding increased overall survival when there is higher diversity in the gut microbiome, including a higher abundance of butyrate producers and lower abundance of enterococcus, explained Paredes when presenting the findings.
Acute GvHD, a common and potentially life-threatening complication of allo-HCT, can have symptoms that mimic irritable bowel disease (IBD), including abdominal pain or cramps, nausea, vomiting, and diarrhea. The low-fiber diet recommendations, including avoidance of raw vegetables and fruits before and after the allo-HCT procedure, are designed to counter those effects, as well as reduce exposure to bacteria.
However, with data suggesting the potential benefits of dietary fiber could extend to the prevention of GvHD, Paredes and colleagues further investigated.
For the observational study, they evaluated all dietary data on 173 allo-HCT recipients at Memorial Sloan Kettering Cancer Center (MSKCC) from 10 days prior to transplantation to 30 days post-transplantation, representing 3837 patient-days in total.
Data collected from the patients also included rRNA sequencing of fecal samples and fecal short-chain fatty acid concentration.
Participants had a median age of 60, and 45% were female. The most common diseases being treated were leukemia (50%), myelodysplastic syndrome (25%), and non-Hodgkin’s lymphoma (8.7%).
After stratifying patients based on high- or low-fiber intake, those with high-fiber intake were found to have significantly higher rates of microbial α-diversity (P = .009), a higher abundance of butyrate producers (P = .03), and a higher concentration of butyrate (P = .02), a short-chain fatty acid that plays a key role in gut health.
Furthermore, the high-fiber group had significantly higher overall survival in an analysis extending to 24 months relative to day 12 of the study (P = .04).
Focusing on GvHD outcomes, the authors further evaluated data on 101 non-T-cell–depleted patients, and identified 29 patients without GvHD and 24 who developed lower gastrointestinal (GI) GvHD.
Patients with lower GI GvHD had significantly lower fecal concentrations of butyrate (P = .03) and acetate (P = .02).
However, patients among those in the high-fiber intake group had a significantly lower cumulative incidence of developing GvHD at day 100 (P = .034) and a lower incidence of lower GI GvHD (P = .04).
A separate preclinical analysis of a mouse model with GvHD further showed that a fiber-rich diet (12% cellulose) significantly increased the expression of genes associated with reduced GvHD, including IDO1 and CEACAM1, and those associated with enrichment of the bile acid pathway.
The findings suggest an opportunity to improve outcomes with relatively small dietary changes, Paredes said.
“Strategies to increase the fiber concentration in these diets paired with the safety that these patients need is what makes this study exciting,” she said in an interview.
“Increasing the fiber intake by 10 to 20 grams/day could potentially increase the microbiome diversity and abundance of butyrate producers, which have been correlated with higher overall survival rates post allo-HCT,” she continued.
“[For instance], that could be an avocado per day, or it could be a small salad per day, or a small vegetable soup per day,” she added. “I would encourage institutions to re-evaluate their menu planning and see how to include more fiber into the meals in a safe way.”
Ultimately, “I think that a dietary intervention outweighs the risks of a pharmacological intervention,” Paredes added.
The necessary duration of a high-fiber diet to produce the beneficial effects on allo-HCT outcomes would likely be over the course of the pre- and post-transplant periods, Paredes added.
“With the survival analysis extending from 5 days before transplantation to 12 days post, we are looking at an intervention that potentially could be around 20 days,” she said.
“We would love to take advantage of the pretransplantation window, in particular, and we can see that just increasing the fiber intake by about 20 grams during this window was shown to improve overall survival after 24 months,” Paredes added.
Importantly, however, some patients may not be appropriate for high-fiber dietary changes, Paredes cautioned.
“Patients that have developed IBD-like symptoms and severe GvHD patients, for example, or with lower GI-GvHD grades 3 and 4 would be not appropriate candidates for a high-fiber diet,” she said.
High-Fiber Diet Slows MM Disease Progression?
The potential important benefits of a high-fiber diet in blood diseases were further demonstrated in a separate study also by MSKCC researchers presented at the meeting, which showed encouraging signs that a plant-based diet rich in fiber could potentially slow disease progression in multiple myeloma (MM).
NUTRIVENTION included 20 patients with the two precancerous MM conditions, monoclonal gammopathy of undetermined significance (MGUS) and smoldering multiple myeloma (SMM), which can last for years without progressing to MM and which researchers have speculated could be a potential opportunity to intervene to prevent progression to cancer.
Patients were provided with a 12-week controlled diet plus health coaching for another 3 months; no meals or coaching were provided for the rest of the 1-year study period. Participants had a median age of 62 and, with being overweight/obesity a risk factor for MM, had a body mass index (BMI) of 25 kg/m2 or higher.
The trial met its endpoint of feasibility, with 91% adherence in the first 3 months. The rate of consumption of unprocessed plant foods increased from 20% at baseline to 92% on the intervention. Overall adherence was 58%. Insulin and anti-inflammatory markers also improved and, despite no calorie restriction, there was a 7% sustained reduction in BMI.
Notably, two patients in the study had stabilization of disease progression.
“We saw improvements in all spheres, including metabolism, microbiome, and immune system markers, and we also saw that two patients with progressive disease had the progression stabilize and slow down on the intervention,” principal investigator Urvi A. Shah, MD, said in a press statement.
“Even though it’s just two cases, to our knowledge, it has not been shown before in an intervention setting that you can improve diet and lifestyle and actually slow or change the trajectory of the disease,” she noted.
The researchers caution that findings in mice do not necessarily translate to humans but note another experiment in mice with SMM that showed animals fed a normal diet had progression to MM after a median of 12 weeks, compared with a median of 30 weeks among those fed a high-fiber diet.
Notably, all mice in the normal-diet group progressed to MM, whereas 40% of mice in the high-fiber group did not.
“We found that a high-fiber plant-based diet can improve BMI, improve insulin resistance [and] the microbiome through diversity and butyrate producers, and with the production of short-chain fatty acids, can have effects on inflammation, immunity, innate and adaptive antitumor immunity, and tumor cells or plasma cells,” Shah said during her presentation.
The study was supported by funding from the National Cancer Institute and private foundations. Paredes has reported no relevant financial relationships. Shah has reported relationships with Sanofi, Bristol Myers Squibb, and Janssen.
A version of this article first appeared on Medscape.com.
Importantly, the findings suggest standard recommendations for patients of a low-fiber diet following allo-HCT may run counter to the potential benefits.
“Significant decrease of fiber intake during transplantation is detrimental. It’s a lost opportunity to promote a healthy gut microbiome, recover from treatment-related microbiota injury, and protect against GVHD,” first author Jenny Paredes, PhD, a staff scientist at City of Hope National Medical Center in Duarte, California, said in a press statement for the study presented at the American Society of Hematology (ASH) 2024 Annual Meeting.
Although the health benefits of dietary fiber on the gut microbiome are well-documented, the effects have recently been shown to extend to outcomes after allo-HCT in general, with researchers finding increased overall survival when there is higher diversity in the gut microbiome, including a higher abundance of butyrate producers and lower abundance of enterococcus, explained Paredes when presenting the findings.
Acute GvHD, a common and potentially life-threatening complication of allo-HCT, can have symptoms that mimic irritable bowel disease (IBD), including abdominal pain or cramps, nausea, vomiting, and diarrhea. The low-fiber diet recommendations, including avoidance of raw vegetables and fruits before and after the allo-HCT procedure, are designed to counter those effects, as well as reduce exposure to bacteria.
However, with data suggesting the potential benefits of dietary fiber could extend to the prevention of GvHD, Paredes and colleagues further investigated.
For the observational study, they evaluated all dietary data on 173 allo-HCT recipients at Memorial Sloan Kettering Cancer Center (MSKCC) from 10 days prior to transplantation to 30 days post-transplantation, representing 3837 patient-days in total.
Data collected from the patients also included rRNA sequencing of fecal samples and fecal short-chain fatty acid concentration.
Participants had a median age of 60, and 45% were female. The most common diseases being treated were leukemia (50%), myelodysplastic syndrome (25%), and non-Hodgkin’s lymphoma (8.7%).
After stratifying patients based on high- or low-fiber intake, those with high-fiber intake were found to have significantly higher rates of microbial α-diversity (P = .009), a higher abundance of butyrate producers (P = .03), and a higher concentration of butyrate (P = .02), a short-chain fatty acid that plays a key role in gut health.
Furthermore, the high-fiber group had significantly higher overall survival in an analysis extending to 24 months relative to day 12 of the study (P = .04).
Focusing on GvHD outcomes, the authors further evaluated data on 101 non-T-cell–depleted patients, and identified 29 patients without GvHD and 24 who developed lower gastrointestinal (GI) GvHD.
Patients with lower GI GvHD had significantly lower fecal concentrations of butyrate (P = .03) and acetate (P = .02).
However, patients among those in the high-fiber intake group had a significantly lower cumulative incidence of developing GvHD at day 100 (P = .034) and a lower incidence of lower GI GvHD (P = .04).
A separate preclinical analysis of a mouse model with GvHD further showed that a fiber-rich diet (12% cellulose) significantly increased the expression of genes associated with reduced GvHD, including IDO1 and CEACAM1, and those associated with enrichment of the bile acid pathway.
The findings suggest an opportunity to improve outcomes with relatively small dietary changes, Paredes said.
“Strategies to increase the fiber concentration in these diets paired with the safety that these patients need is what makes this study exciting,” she said in an interview.
“Increasing the fiber intake by 10 to 20 grams/day could potentially increase the microbiome diversity and abundance of butyrate producers, which have been correlated with higher overall survival rates post allo-HCT,” she continued.
“[For instance], that could be an avocado per day, or it could be a small salad per day, or a small vegetable soup per day,” she added. “I would encourage institutions to re-evaluate their menu planning and see how to include more fiber into the meals in a safe way.”
Ultimately, “I think that a dietary intervention outweighs the risks of a pharmacological intervention,” Paredes added.
The necessary duration of a high-fiber diet to produce the beneficial effects on allo-HCT outcomes would likely be over the course of the pre- and post-transplant periods, Paredes added.
“With the survival analysis extending from 5 days before transplantation to 12 days post, we are looking at an intervention that potentially could be around 20 days,” she said.
“We would love to take advantage of the pretransplantation window, in particular, and we can see that just increasing the fiber intake by about 20 grams during this window was shown to improve overall survival after 24 months,” Paredes added.
Importantly, however, some patients may not be appropriate for high-fiber dietary changes, Paredes cautioned.
“Patients that have developed IBD-like symptoms and severe GvHD patients, for example, or with lower GI-GvHD grades 3 and 4 would be not appropriate candidates for a high-fiber diet,” she said.
High-Fiber Diet Slows MM Disease Progression?
The potential important benefits of a high-fiber diet in blood diseases were further demonstrated in a separate study also by MSKCC researchers presented at the meeting, which showed encouraging signs that a plant-based diet rich in fiber could potentially slow disease progression in multiple myeloma (MM).
NUTRIVENTION included 20 patients with the two precancerous MM conditions, monoclonal gammopathy of undetermined significance (MGUS) and smoldering multiple myeloma (SMM), which can last for years without progressing to MM and which researchers have speculated could be a potential opportunity to intervene to prevent progression to cancer.
Patients were provided with a 12-week controlled diet plus health coaching for another 3 months; no meals or coaching were provided for the rest of the 1-year study period. Participants had a median age of 62 and, with being overweight/obesity a risk factor for MM, had a body mass index (BMI) of 25 kg/m2 or higher.
The trial met its endpoint of feasibility, with 91% adherence in the first 3 months. The rate of consumption of unprocessed plant foods increased from 20% at baseline to 92% on the intervention. Overall adherence was 58%. Insulin and anti-inflammatory markers also improved and, despite no calorie restriction, there was a 7% sustained reduction in BMI.
Notably, two patients in the study had stabilization of disease progression.
“We saw improvements in all spheres, including metabolism, microbiome, and immune system markers, and we also saw that two patients with progressive disease had the progression stabilize and slow down on the intervention,” principal investigator Urvi A. Shah, MD, said in a press statement.
“Even though it’s just two cases, to our knowledge, it has not been shown before in an intervention setting that you can improve diet and lifestyle and actually slow or change the trajectory of the disease,” she noted.
The researchers caution that findings in mice do not necessarily translate to humans but note another experiment in mice with SMM that showed animals fed a normal diet had progression to MM after a median of 12 weeks, compared with a median of 30 weeks among those fed a high-fiber diet.
Notably, all mice in the normal-diet group progressed to MM, whereas 40% of mice in the high-fiber group did not.
“We found that a high-fiber plant-based diet can improve BMI, improve insulin resistance [and] the microbiome through diversity and butyrate producers, and with the production of short-chain fatty acids, can have effects on inflammation, immunity, innate and adaptive antitumor immunity, and tumor cells or plasma cells,” Shah said during her presentation.
The study was supported by funding from the National Cancer Institute and private foundations. Paredes has reported no relevant financial relationships. Shah has reported relationships with Sanofi, Bristol Myers Squibb, and Janssen.
A version of this article first appeared on Medscape.com.
Importantly, the findings suggest standard recommendations for patients of a low-fiber diet following allo-HCT may run counter to the potential benefits.
“Significant decrease of fiber intake during transplantation is detrimental. It’s a lost opportunity to promote a healthy gut microbiome, recover from treatment-related microbiota injury, and protect against GVHD,” first author Jenny Paredes, PhD, a staff scientist at City of Hope National Medical Center in Duarte, California, said in a press statement for the study presented at the American Society of Hematology (ASH) 2024 Annual Meeting.
Although the health benefits of dietary fiber on the gut microbiome are well-documented, the effects have recently been shown to extend to outcomes after allo-HCT in general, with researchers finding increased overall survival when there is higher diversity in the gut microbiome, including a higher abundance of butyrate producers and lower abundance of enterococcus, explained Paredes when presenting the findings.
Acute GvHD, a common and potentially life-threatening complication of allo-HCT, can have symptoms that mimic irritable bowel disease (IBD), including abdominal pain or cramps, nausea, vomiting, and diarrhea. The low-fiber diet recommendations, including avoidance of raw vegetables and fruits before and after the allo-HCT procedure, are designed to counter those effects, as well as reduce exposure to bacteria.
However, with data suggesting the potential benefits of dietary fiber could extend to the prevention of GvHD, Paredes and colleagues further investigated.
For the observational study, they evaluated all dietary data on 173 allo-HCT recipients at Memorial Sloan Kettering Cancer Center (MSKCC) from 10 days prior to transplantation to 30 days post-transplantation, representing 3837 patient-days in total.
Data collected from the patients also included rRNA sequencing of fecal samples and fecal short-chain fatty acid concentration.
Participants had a median age of 60, and 45% were female. The most common diseases being treated were leukemia (50%), myelodysplastic syndrome (25%), and non-Hodgkin’s lymphoma (8.7%).
After stratifying patients based on high- or low-fiber intake, those with high-fiber intake were found to have significantly higher rates of microbial α-diversity (P = .009), a higher abundance of butyrate producers (P = .03), and a higher concentration of butyrate (P = .02), a short-chain fatty acid that plays a key role in gut health.
Furthermore, the high-fiber group had significantly higher overall survival in an analysis extending to 24 months relative to day 12 of the study (P = .04).
Focusing on GvHD outcomes, the authors further evaluated data on 101 non-T-cell–depleted patients, and identified 29 patients without GvHD and 24 who developed lower gastrointestinal (GI) GvHD.
Patients with lower GI GvHD had significantly lower fecal concentrations of butyrate (P = .03) and acetate (P = .02).
However, patients among those in the high-fiber intake group had a significantly lower cumulative incidence of developing GvHD at day 100 (P = .034) and a lower incidence of lower GI GvHD (P = .04).
A separate preclinical analysis of a mouse model with GvHD further showed that a fiber-rich diet (12% cellulose) significantly increased the expression of genes associated with reduced GvHD, including IDO1 and CEACAM1, and those associated with enrichment of the bile acid pathway.
The findings suggest an opportunity to improve outcomes with relatively small dietary changes, Paredes said.
“Strategies to increase the fiber concentration in these diets paired with the safety that these patients need is what makes this study exciting,” she said in an interview.
“Increasing the fiber intake by 10 to 20 grams/day could potentially increase the microbiome diversity and abundance of butyrate producers, which have been correlated with higher overall survival rates post allo-HCT,” she continued.
“[For instance], that could be an avocado per day, or it could be a small salad per day, or a small vegetable soup per day,” she added. “I would encourage institutions to re-evaluate their menu planning and see how to include more fiber into the meals in a safe way.”
Ultimately, “I think that a dietary intervention outweighs the risks of a pharmacological intervention,” Paredes added.
The necessary duration of a high-fiber diet to produce the beneficial effects on allo-HCT outcomes would likely be over the course of the pre- and post-transplant periods, Paredes added.
“With the survival analysis extending from 5 days before transplantation to 12 days post, we are looking at an intervention that potentially could be around 20 days,” she said.
“We would love to take advantage of the pretransplantation window, in particular, and we can see that just increasing the fiber intake by about 20 grams during this window was shown to improve overall survival after 24 months,” Paredes added.
Importantly, however, some patients may not be appropriate for high-fiber dietary changes, Paredes cautioned.
“Patients that have developed IBD-like symptoms and severe GvHD patients, for example, or with lower GI-GvHD grades 3 and 4 would be not appropriate candidates for a high-fiber diet,” she said.
High-Fiber Diet Slows MM Disease Progression?
The potential important benefits of a high-fiber diet in blood diseases were further demonstrated in a separate study also by MSKCC researchers presented at the meeting, which showed encouraging signs that a plant-based diet rich in fiber could potentially slow disease progression in multiple myeloma (MM).
NUTRIVENTION included 20 patients with the two precancerous MM conditions, monoclonal gammopathy of undetermined significance (MGUS) and smoldering multiple myeloma (SMM), which can last for years without progressing to MM and which researchers have speculated could be a potential opportunity to intervene to prevent progression to cancer.
Patients were provided with a 12-week controlled diet plus health coaching for another 3 months; no meals or coaching were provided for the rest of the 1-year study period. Participants had a median age of 62 and, with being overweight/obesity a risk factor for MM, had a body mass index (BMI) of 25 kg/m2 or higher.
The trial met its endpoint of feasibility, with 91% adherence in the first 3 months. The rate of consumption of unprocessed plant foods increased from 20% at baseline to 92% on the intervention. Overall adherence was 58%. Insulin and anti-inflammatory markers also improved and, despite no calorie restriction, there was a 7% sustained reduction in BMI.
Notably, two patients in the study had stabilization of disease progression.
“We saw improvements in all spheres, including metabolism, microbiome, and immune system markers, and we also saw that two patients with progressive disease had the progression stabilize and slow down on the intervention,” principal investigator Urvi A. Shah, MD, said in a press statement.
“Even though it’s just two cases, to our knowledge, it has not been shown before in an intervention setting that you can improve diet and lifestyle and actually slow or change the trajectory of the disease,” she noted.
The researchers caution that findings in mice do not necessarily translate to humans but note another experiment in mice with SMM that showed animals fed a normal diet had progression to MM after a median of 12 weeks, compared with a median of 30 weeks among those fed a high-fiber diet.
Notably, all mice in the normal-diet group progressed to MM, whereas 40% of mice in the high-fiber group did not.
“We found that a high-fiber plant-based diet can improve BMI, improve insulin resistance [and] the microbiome through diversity and butyrate producers, and with the production of short-chain fatty acids, can have effects on inflammation, immunity, innate and adaptive antitumor immunity, and tumor cells or plasma cells,” Shah said during her presentation.
The study was supported by funding from the National Cancer Institute and private foundations. Paredes has reported no relevant financial relationships. Shah has reported relationships with Sanofi, Bristol Myers Squibb, and Janssen.
A version of this article first appeared on Medscape.com.
FROM ASH 2024
LBCL: Bispecific Antibodies Fare Less Well in Real-World Analysis
In a presentation at the American Society of Hematology (ASH) 2024 Annual Meeting, researchers reported that of 172 patients treated with the drugs who had evaluable responses over a median follow-up of 5 months, median progression-free survival was 2.7 months (95% CI, 2.0-3.9) and median overall survival was 7.2 months (95% CI, 6.1–not reached).
It’s important to consider the real-world nature of the study’s patient population, said first author Taylor R. Brooks, MD, of Cleveland Clinic, Ohio, in an interview. “Compared to pivotal trials, our cohort was enriched for patients with high-risk features, with almost three quarters having some comorbidity that would’ve excluded them from one of the [earlier] studies.”
He added that “though individuals eligible to receive these medicines may be more sick with high-risk disease, a sizable fraction will respond, and some will maintain remissions.’”
According to Brooks, about one third of patients with diffuse LBCL relapse after standard front-line R-CHOP therapy. “The prognosis is poor for patients who are not candidates for aggressive salvage chemotherapy and for those who relapse after two or more lines,” Brooks said. “T cell–engaging bispecific antibodies have emerged as a promising option for patients with relapsed or refractory large B-cell lymphoma, given their favorable rates and duration of responses as well as their manageable rates of toxicities.”
The Food and Drug Administration (FDA) granted accelerated approval for epcoritamab and glofitamab in 2023.
“With increasing uptake into clinical practice following the FDA approvals, there is increasing interest in assessing the efficacy and safety of these drugs in real-world, nontrial settings,” Brooks said. “The goal of our study was to investigate outcomes and identify clinical factors associated with outcomes.”
The multicenter, retrospective, observational REALBiTE study tracked 209 patients with relapsed/refractory diffuse LBCL at 19 US centers (epcoritamab, n = 139; glofitamab, n = 70; median age at start of treatment, 67 years [58-76]; 62.2% male; 74.2% diffuse LBCL). The median number of lines of therapy was three (range, 1-12).
“Patients who received epcoritamab tended to be slightly older, were more likely to have a history of indolent non-Hodgkin lymphoma prior to their diagnosis of aggressive B-cell lymphoma and were more likely to have an elevated International Prognostic Index score at the start of bispecific therapy, suggesting that these patients may have been slightly older with higher-risk disease compared to those who received glofitamab,” Brooks said.
In total, 172 patients were response-evaluable. The overall response rate was 50.6% (complete response, 23.8%; partial response, 26.7%; stable disease, 5.8%; progressive disease, 43.6%).
The overall and complete response rates were “somewhat lower that what has been published in the pivotal trials of these medicines,” Brooks said. The low progression-free and overall survival rates “highlight the difficulty in managing this group of patients.”
Cytokine release syndrome (CRS) of any grade occurred in 39.2% of patients: 51% in the epcoritamab group and 28.6% in the glofitamab group. Grade ≥3 CRS occurred in 4.3% of patients, who were all taking epcoritamab.
“For epcoritamab, CRS was almost entirely of low grade, and most CRS events occurred around administration of the first full dose of the drug on day 15,” Brooks said. “Similarly, the CRS events for glofitamab were mostly of low grade, though events were observed to occur throughout the step-up dosing. Tocilizumab was administered in about one fifth of the patients.”
In addition, Brooks said, “we found that, among the 19 individuals with paired biopsy samples before and after bispecific therapy, nearly all — 89% — were found to have lost CD20 expression. We expected some patients to experience loss of this important target, but the rate at which we found this to be the case was surprisingly high.”
Brooks added that “clinicians should be acquainted with CRS, ICANS [immune effector cell-associated neurotoxicity syndrome], and mitigation strategies if they are prescribing these medicines. Appropriate and timely management using tocilizumab, steroids, and other adjunctive measures can effectively manage these complications and hopefully allow for the continued delivery of therapy.”
In an interview, Matthew Lunning, DO, associate professor at the University of Nebraska Medical Center/Fred & Pamela Buffett Cancer Center, Omaha, who didn’t take part in the new study, said the findings aren’t bad news. Instead, they’re “practical news,” because they offer insight into how the drugs work.
“The big lesson from this and other trials is the importance of assessing for CD20 expression prior to taking a bispecific off the shelf, “ he said. “These are learnings that often come after approval.”
He added that it’s clear that, “in more heavily pretreated patients, more disease led to less optimal results and higher risk for toxicities.”
Lunning also noted that both epcoritamab and glofitamab “entered into a crowded and chaotic relapsed/refractory LBCL space based high complete response rates with the opportunity for durability in those complete responses.”
Academic institutions were especially interested, as they can manage CRS and ICANS, but “significantly less enthusiasm has been seen in community practices that expect CRS/ICANS to be in the rear-view mirror if they are going to deliver any bispecific,” he said. “It is not that they don’t have the clinical acumen to manage CRS/ICANS. I believe it is the perception of the lack of supportive infrastructure necessary to manage these toxicities.”
There was no study funding. Brooks has reported no disclosures. Other authors have reported various disclosures including relationships with Novartis, AbbVie, Genentech, Genmab, Biogen, Amgen, and others. Lunning has disclosed ties with AbbVie, Genmab, Kite, Bristol-Myers Squibb, Regeneron, and ADC Therapeutics.
A version of this article first appeared on Medscape.com.
In a presentation at the American Society of Hematology (ASH) 2024 Annual Meeting, researchers reported that of 172 patients treated with the drugs who had evaluable responses over a median follow-up of 5 months, median progression-free survival was 2.7 months (95% CI, 2.0-3.9) and median overall survival was 7.2 months (95% CI, 6.1–not reached).
It’s important to consider the real-world nature of the study’s patient population, said first author Taylor R. Brooks, MD, of Cleveland Clinic, Ohio, in an interview. “Compared to pivotal trials, our cohort was enriched for patients with high-risk features, with almost three quarters having some comorbidity that would’ve excluded them from one of the [earlier] studies.”
He added that “though individuals eligible to receive these medicines may be more sick with high-risk disease, a sizable fraction will respond, and some will maintain remissions.’”
According to Brooks, about one third of patients with diffuse LBCL relapse after standard front-line R-CHOP therapy. “The prognosis is poor for patients who are not candidates for aggressive salvage chemotherapy and for those who relapse after two or more lines,” Brooks said. “T cell–engaging bispecific antibodies have emerged as a promising option for patients with relapsed or refractory large B-cell lymphoma, given their favorable rates and duration of responses as well as their manageable rates of toxicities.”
The Food and Drug Administration (FDA) granted accelerated approval for epcoritamab and glofitamab in 2023.
“With increasing uptake into clinical practice following the FDA approvals, there is increasing interest in assessing the efficacy and safety of these drugs in real-world, nontrial settings,” Brooks said. “The goal of our study was to investigate outcomes and identify clinical factors associated with outcomes.”
The multicenter, retrospective, observational REALBiTE study tracked 209 patients with relapsed/refractory diffuse LBCL at 19 US centers (epcoritamab, n = 139; glofitamab, n = 70; median age at start of treatment, 67 years [58-76]; 62.2% male; 74.2% diffuse LBCL). The median number of lines of therapy was three (range, 1-12).
“Patients who received epcoritamab tended to be slightly older, were more likely to have a history of indolent non-Hodgkin lymphoma prior to their diagnosis of aggressive B-cell lymphoma and were more likely to have an elevated International Prognostic Index score at the start of bispecific therapy, suggesting that these patients may have been slightly older with higher-risk disease compared to those who received glofitamab,” Brooks said.
In total, 172 patients were response-evaluable. The overall response rate was 50.6% (complete response, 23.8%; partial response, 26.7%; stable disease, 5.8%; progressive disease, 43.6%).
The overall and complete response rates were “somewhat lower that what has been published in the pivotal trials of these medicines,” Brooks said. The low progression-free and overall survival rates “highlight the difficulty in managing this group of patients.”
Cytokine release syndrome (CRS) of any grade occurred in 39.2% of patients: 51% in the epcoritamab group and 28.6% in the glofitamab group. Grade ≥3 CRS occurred in 4.3% of patients, who were all taking epcoritamab.
“For epcoritamab, CRS was almost entirely of low grade, and most CRS events occurred around administration of the first full dose of the drug on day 15,” Brooks said. “Similarly, the CRS events for glofitamab were mostly of low grade, though events were observed to occur throughout the step-up dosing. Tocilizumab was administered in about one fifth of the patients.”
In addition, Brooks said, “we found that, among the 19 individuals with paired biopsy samples before and after bispecific therapy, nearly all — 89% — were found to have lost CD20 expression. We expected some patients to experience loss of this important target, but the rate at which we found this to be the case was surprisingly high.”
Brooks added that “clinicians should be acquainted with CRS, ICANS [immune effector cell-associated neurotoxicity syndrome], and mitigation strategies if they are prescribing these medicines. Appropriate and timely management using tocilizumab, steroids, and other adjunctive measures can effectively manage these complications and hopefully allow for the continued delivery of therapy.”
In an interview, Matthew Lunning, DO, associate professor at the University of Nebraska Medical Center/Fred & Pamela Buffett Cancer Center, Omaha, who didn’t take part in the new study, said the findings aren’t bad news. Instead, they’re “practical news,” because they offer insight into how the drugs work.
“The big lesson from this and other trials is the importance of assessing for CD20 expression prior to taking a bispecific off the shelf, “ he said. “These are learnings that often come after approval.”
He added that it’s clear that, “in more heavily pretreated patients, more disease led to less optimal results and higher risk for toxicities.”
Lunning also noted that both epcoritamab and glofitamab “entered into a crowded and chaotic relapsed/refractory LBCL space based high complete response rates with the opportunity for durability in those complete responses.”
Academic institutions were especially interested, as they can manage CRS and ICANS, but “significantly less enthusiasm has been seen in community practices that expect CRS/ICANS to be in the rear-view mirror if they are going to deliver any bispecific,” he said. “It is not that they don’t have the clinical acumen to manage CRS/ICANS. I believe it is the perception of the lack of supportive infrastructure necessary to manage these toxicities.”
There was no study funding. Brooks has reported no disclosures. Other authors have reported various disclosures including relationships with Novartis, AbbVie, Genentech, Genmab, Biogen, Amgen, and others. Lunning has disclosed ties with AbbVie, Genmab, Kite, Bristol-Myers Squibb, Regeneron, and ADC Therapeutics.
A version of this article first appeared on Medscape.com.
In a presentation at the American Society of Hematology (ASH) 2024 Annual Meeting, researchers reported that of 172 patients treated with the drugs who had evaluable responses over a median follow-up of 5 months, median progression-free survival was 2.7 months (95% CI, 2.0-3.9) and median overall survival was 7.2 months (95% CI, 6.1–not reached).
It’s important to consider the real-world nature of the study’s patient population, said first author Taylor R. Brooks, MD, of Cleveland Clinic, Ohio, in an interview. “Compared to pivotal trials, our cohort was enriched for patients with high-risk features, with almost three quarters having some comorbidity that would’ve excluded them from one of the [earlier] studies.”
He added that “though individuals eligible to receive these medicines may be more sick with high-risk disease, a sizable fraction will respond, and some will maintain remissions.’”
According to Brooks, about one third of patients with diffuse LBCL relapse after standard front-line R-CHOP therapy. “The prognosis is poor for patients who are not candidates for aggressive salvage chemotherapy and for those who relapse after two or more lines,” Brooks said. “T cell–engaging bispecific antibodies have emerged as a promising option for patients with relapsed or refractory large B-cell lymphoma, given their favorable rates and duration of responses as well as their manageable rates of toxicities.”
The Food and Drug Administration (FDA) granted accelerated approval for epcoritamab and glofitamab in 2023.
“With increasing uptake into clinical practice following the FDA approvals, there is increasing interest in assessing the efficacy and safety of these drugs in real-world, nontrial settings,” Brooks said. “The goal of our study was to investigate outcomes and identify clinical factors associated with outcomes.”
The multicenter, retrospective, observational REALBiTE study tracked 209 patients with relapsed/refractory diffuse LBCL at 19 US centers (epcoritamab, n = 139; glofitamab, n = 70; median age at start of treatment, 67 years [58-76]; 62.2% male; 74.2% diffuse LBCL). The median number of lines of therapy was three (range, 1-12).
“Patients who received epcoritamab tended to be slightly older, were more likely to have a history of indolent non-Hodgkin lymphoma prior to their diagnosis of aggressive B-cell lymphoma and were more likely to have an elevated International Prognostic Index score at the start of bispecific therapy, suggesting that these patients may have been slightly older with higher-risk disease compared to those who received glofitamab,” Brooks said.
In total, 172 patients were response-evaluable. The overall response rate was 50.6% (complete response, 23.8%; partial response, 26.7%; stable disease, 5.8%; progressive disease, 43.6%).
The overall and complete response rates were “somewhat lower that what has been published in the pivotal trials of these medicines,” Brooks said. The low progression-free and overall survival rates “highlight the difficulty in managing this group of patients.”
Cytokine release syndrome (CRS) of any grade occurred in 39.2% of patients: 51% in the epcoritamab group and 28.6% in the glofitamab group. Grade ≥3 CRS occurred in 4.3% of patients, who were all taking epcoritamab.
“For epcoritamab, CRS was almost entirely of low grade, and most CRS events occurred around administration of the first full dose of the drug on day 15,” Brooks said. “Similarly, the CRS events for glofitamab were mostly of low grade, though events were observed to occur throughout the step-up dosing. Tocilizumab was administered in about one fifth of the patients.”
In addition, Brooks said, “we found that, among the 19 individuals with paired biopsy samples before and after bispecific therapy, nearly all — 89% — were found to have lost CD20 expression. We expected some patients to experience loss of this important target, but the rate at which we found this to be the case was surprisingly high.”
Brooks added that “clinicians should be acquainted with CRS, ICANS [immune effector cell-associated neurotoxicity syndrome], and mitigation strategies if they are prescribing these medicines. Appropriate and timely management using tocilizumab, steroids, and other adjunctive measures can effectively manage these complications and hopefully allow for the continued delivery of therapy.”
In an interview, Matthew Lunning, DO, associate professor at the University of Nebraska Medical Center/Fred & Pamela Buffett Cancer Center, Omaha, who didn’t take part in the new study, said the findings aren’t bad news. Instead, they’re “practical news,” because they offer insight into how the drugs work.
“The big lesson from this and other trials is the importance of assessing for CD20 expression prior to taking a bispecific off the shelf, “ he said. “These are learnings that often come after approval.”
He added that it’s clear that, “in more heavily pretreated patients, more disease led to less optimal results and higher risk for toxicities.”
Lunning also noted that both epcoritamab and glofitamab “entered into a crowded and chaotic relapsed/refractory LBCL space based high complete response rates with the opportunity for durability in those complete responses.”
Academic institutions were especially interested, as they can manage CRS and ICANS, but “significantly less enthusiasm has been seen in community practices that expect CRS/ICANS to be in the rear-view mirror if they are going to deliver any bispecific,” he said. “It is not that they don’t have the clinical acumen to manage CRS/ICANS. I believe it is the perception of the lack of supportive infrastructure necessary to manage these toxicities.”
There was no study funding. Brooks has reported no disclosures. Other authors have reported various disclosures including relationships with Novartis, AbbVie, Genentech, Genmab, Biogen, Amgen, and others. Lunning has disclosed ties with AbbVie, Genmab, Kite, Bristol-Myers Squibb, Regeneron, and ADC Therapeutics.
A version of this article first appeared on Medscape.com.
FROM ASH 2024

