User login
Treatment of basal cell carcinoma with 1064-nm Nd:YAG laser promising
DALLAS – One year after patients underwent treatment of basal cell carcinoma (BCC) with the 1064-nm Nd:YAG laser, no recurrences have occurred, according to early results from a study being conducted at two centers.
“ Arisa E. Ortiz, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Clearance rates are comparable to or better than other topical modalities such as electrodesiccation and curettage and topical imiquimod. It’s a reasonable alternative for treatment patients with multiple tumors or those who are poor surgical candidates.”
In an ongoing study, she and Mathew M. Avram, MD, director of the Massachusetts General Hospital Dermatology Laser & Cosmetic Center, Boston, have treated 19 superficial, nodular, and pigmented BCC tumors in 11 patients 31-85 years of age. Tumor sizes ranged from 3 mm x 3 mm to 21 mm x 11 mm. Indications for laser treatment have included being a poor surgical candidate (one patient had a history of bleeding complications), having multiple tumors (one patient had Curry-Jones syndrome) – or simply wishing to not undergo surgery. “They didn’t want a surgical scar, or they didn’t want to limit their activity after surgery,” Dr. Ortiz said.
Patients underwent one 1064-nm Nd:YAG laser treatment. The anesthesia was 0.5% lidocaine with no epinephrine. Treatment settings were a 5-mm spot size delivered at a fluence of 140 J/cm2 in a pulse duration of 7-8 milliseconds. The number of pulses ranged from 14 to 36. The immediate endpoint was slight graying and slight contraction. “When you’re using the 1064-nm Nd:YAG for cosmetic purposes, you don’t want to see these endpoints, but we are treating skin cancer, so you do want to see some contraction and graying,” she said. The procedure was covered under insurance and billed as malignant destruction (CPT codes 17260-17266 and 17280-17283).
Dr. Ortiz reported that there have been no recurrences in the 11 patients at 1-year follow-up as determined by clinical observation. “There are many advantages to laser treatment of basal cell carcinoma,” she concluded. “There’s only one treatment visit, so you don’t have to come back for suture removal, and it’s a very quick treatment. There’s no significant downtime or limitation on activities, and there’s minimal wound care – just ointment and a Band-Aid – and relatively decreased risk for complications such as infection or bleeding, and minimal to no scar.”
“Laser surgery also provides precision that ordinary surgical techniques cannot match. Despite the obvious obstacles, there is no reason such surgical techniques cannot be expanded someday to other internal cutaneous tumors, including GI tumors. To some extent this is happening already. Vascular lasers are being used to treat a bleeding disorder of the colon known as angiodysplasia. Cautious exploration of laser- and light-based treatments should be further explored as a means of sparing tissue and surgical morbidity.”
Dr. Ortiz disclosed that she has received grant funding from Sienna and Revance, as well as equipment from BTL, Invasix, and Sciton. She has received consulting fees from Alastin, Merz, and Sciton; honoraria from Alastin, Cutera, Invasix, and Sciton; and she holds ownership interest with Allergan. She also has served on the advisory boards for Alastin, Allergan, Invasix, Rodan + Fields, Sciton, Sienna, and Merz.
Dr. Avram disclosed that he serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also a consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
DALLAS – One year after patients underwent treatment of basal cell carcinoma (BCC) with the 1064-nm Nd:YAG laser, no recurrences have occurred, according to early results from a study being conducted at two centers.
“ Arisa E. Ortiz, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Clearance rates are comparable to or better than other topical modalities such as electrodesiccation and curettage and topical imiquimod. It’s a reasonable alternative for treatment patients with multiple tumors or those who are poor surgical candidates.”
In an ongoing study, she and Mathew M. Avram, MD, director of the Massachusetts General Hospital Dermatology Laser & Cosmetic Center, Boston, have treated 19 superficial, nodular, and pigmented BCC tumors in 11 patients 31-85 years of age. Tumor sizes ranged from 3 mm x 3 mm to 21 mm x 11 mm. Indications for laser treatment have included being a poor surgical candidate (one patient had a history of bleeding complications), having multiple tumors (one patient had Curry-Jones syndrome) – or simply wishing to not undergo surgery. “They didn’t want a surgical scar, or they didn’t want to limit their activity after surgery,” Dr. Ortiz said.
Patients underwent one 1064-nm Nd:YAG laser treatment. The anesthesia was 0.5% lidocaine with no epinephrine. Treatment settings were a 5-mm spot size delivered at a fluence of 140 J/cm2 in a pulse duration of 7-8 milliseconds. The number of pulses ranged from 14 to 36. The immediate endpoint was slight graying and slight contraction. “When you’re using the 1064-nm Nd:YAG for cosmetic purposes, you don’t want to see these endpoints, but we are treating skin cancer, so you do want to see some contraction and graying,” she said. The procedure was covered under insurance and billed as malignant destruction (CPT codes 17260-17266 and 17280-17283).
Dr. Ortiz reported that there have been no recurrences in the 11 patients at 1-year follow-up as determined by clinical observation. “There are many advantages to laser treatment of basal cell carcinoma,” she concluded. “There’s only one treatment visit, so you don’t have to come back for suture removal, and it’s a very quick treatment. There’s no significant downtime or limitation on activities, and there’s minimal wound care – just ointment and a Band-Aid – and relatively decreased risk for complications such as infection or bleeding, and minimal to no scar.”
“Laser surgery also provides precision that ordinary surgical techniques cannot match. Despite the obvious obstacles, there is no reason such surgical techniques cannot be expanded someday to other internal cutaneous tumors, including GI tumors. To some extent this is happening already. Vascular lasers are being used to treat a bleeding disorder of the colon known as angiodysplasia. Cautious exploration of laser- and light-based treatments should be further explored as a means of sparing tissue and surgical morbidity.”
Dr. Ortiz disclosed that she has received grant funding from Sienna and Revance, as well as equipment from BTL, Invasix, and Sciton. She has received consulting fees from Alastin, Merz, and Sciton; honoraria from Alastin, Cutera, Invasix, and Sciton; and she holds ownership interest with Allergan. She also has served on the advisory boards for Alastin, Allergan, Invasix, Rodan + Fields, Sciton, Sienna, and Merz.
Dr. Avram disclosed that he serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also a consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
DALLAS – One year after patients underwent treatment of basal cell carcinoma (BCC) with the 1064-nm Nd:YAG laser, no recurrences have occurred, according to early results from a study being conducted at two centers.
“ Arisa E. Ortiz, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Clearance rates are comparable to or better than other topical modalities such as electrodesiccation and curettage and topical imiquimod. It’s a reasonable alternative for treatment patients with multiple tumors or those who are poor surgical candidates.”
In an ongoing study, she and Mathew M. Avram, MD, director of the Massachusetts General Hospital Dermatology Laser & Cosmetic Center, Boston, have treated 19 superficial, nodular, and pigmented BCC tumors in 11 patients 31-85 years of age. Tumor sizes ranged from 3 mm x 3 mm to 21 mm x 11 mm. Indications for laser treatment have included being a poor surgical candidate (one patient had a history of bleeding complications), having multiple tumors (one patient had Curry-Jones syndrome) – or simply wishing to not undergo surgery. “They didn’t want a surgical scar, or they didn’t want to limit their activity after surgery,” Dr. Ortiz said.
Patients underwent one 1064-nm Nd:YAG laser treatment. The anesthesia was 0.5% lidocaine with no epinephrine. Treatment settings were a 5-mm spot size delivered at a fluence of 140 J/cm2 in a pulse duration of 7-8 milliseconds. The number of pulses ranged from 14 to 36. The immediate endpoint was slight graying and slight contraction. “When you’re using the 1064-nm Nd:YAG for cosmetic purposes, you don’t want to see these endpoints, but we are treating skin cancer, so you do want to see some contraction and graying,” she said. The procedure was covered under insurance and billed as malignant destruction (CPT codes 17260-17266 and 17280-17283).
Dr. Ortiz reported that there have been no recurrences in the 11 patients at 1-year follow-up as determined by clinical observation. “There are many advantages to laser treatment of basal cell carcinoma,” she concluded. “There’s only one treatment visit, so you don’t have to come back for suture removal, and it’s a very quick treatment. There’s no significant downtime or limitation on activities, and there’s minimal wound care – just ointment and a Band-Aid – and relatively decreased risk for complications such as infection or bleeding, and minimal to no scar.”
“Laser surgery also provides precision that ordinary surgical techniques cannot match. Despite the obvious obstacles, there is no reason such surgical techniques cannot be expanded someday to other internal cutaneous tumors, including GI tumors. To some extent this is happening already. Vascular lasers are being used to treat a bleeding disorder of the colon known as angiodysplasia. Cautious exploration of laser- and light-based treatments should be further explored as a means of sparing tissue and surgical morbidity.”
Dr. Ortiz disclosed that she has received grant funding from Sienna and Revance, as well as equipment from BTL, Invasix, and Sciton. She has received consulting fees from Alastin, Merz, and Sciton; honoraria from Alastin, Cutera, Invasix, and Sciton; and she holds ownership interest with Allergan. She also has served on the advisory boards for Alastin, Allergan, Invasix, Rodan + Fields, Sciton, Sienna, and Merz.
Dr. Avram disclosed that he serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also a consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
REPORTING FROM ASLMS 2018
Key clinical point: Clinicians can noninvasively treat certain basal cell carcinoma tumor subtypes with the 1064-nm Nd:YAG laser.
Major finding: After 1 year of follow-up, no recurrences of basal cell carcinoma have occurred.
Study details: A 1-year follow-up study of 19 BCC tumors in 11 patients 31 to 85 years of age who were treated with the 1064-nm Nd:YAG laser.
Disclosures: Dr. Ortiz disclosed that she has received grant funding from Sienna and Revance, as well as equipment from BTL, Invasix, and Sciton. She has received consulting fees from Alastin, Merz, and Sciton; honoraria from Alastin, Cutera, Invasix, and Sciton; and she holds ownership interest with Allergan. She also has served on the advisory boards for Alastin, Allergan, Invasix, Rodan + Fields, Sciton, Sienna, and Merz.
Dr. Avram disclosed that he serves on the medical advisory board of Sciton and on the scientific advisory boards of Sienna Biopharmaceuticals, Cytrellis, and Allergan. He is also a consultant for Merz Aesthetics, Allergan, Soliton, Invasix, and Revance and has intellectual property with Cytrellis. He also holds stock options with Cytrellis, Invasix, and Zalea.
Enhanced Melanoma Diagnosis With Multispectral Digital Skin Lesion Analysis
Early detection of melanoma, which is known to improve survival rates, remains a challenge for dermatologists. Suspicious pigmented lesions typically are evaluated via clinical examination and dermoscopy; however, new technologies are being developed to provide additional objective information for clinicians to incorporate into their biopsy decisions.
Multispectral digital skin lesion analysis (MSDSLA) uses 10 bands of visible and near-infrared light (430–950 nm) to image and analyze pigmented skin lesions (PSLs) down to 2.5 mm below the skin surface and measures the distribution of melanin using 75 unique algorithms to determine the degree of the morphologic disorder. Using a logical regression model previously validated on a set of 1632 PSLs, the probability of melanoma and probability of being a melanoma/PSL of high-risk malignant potential are then provided to the clinician.1
In this study, we analyzed aggregate data from 7 prior studies2-8 to better determine how MSDSLA impacts the biopsy decisions of dermatologists and nondermatologists following clinical examination and dermoscopic evaluation of PSLs.
Methods
Results
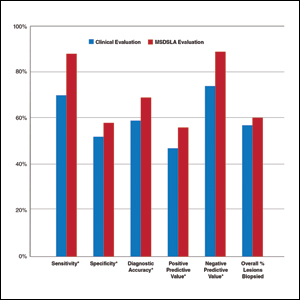
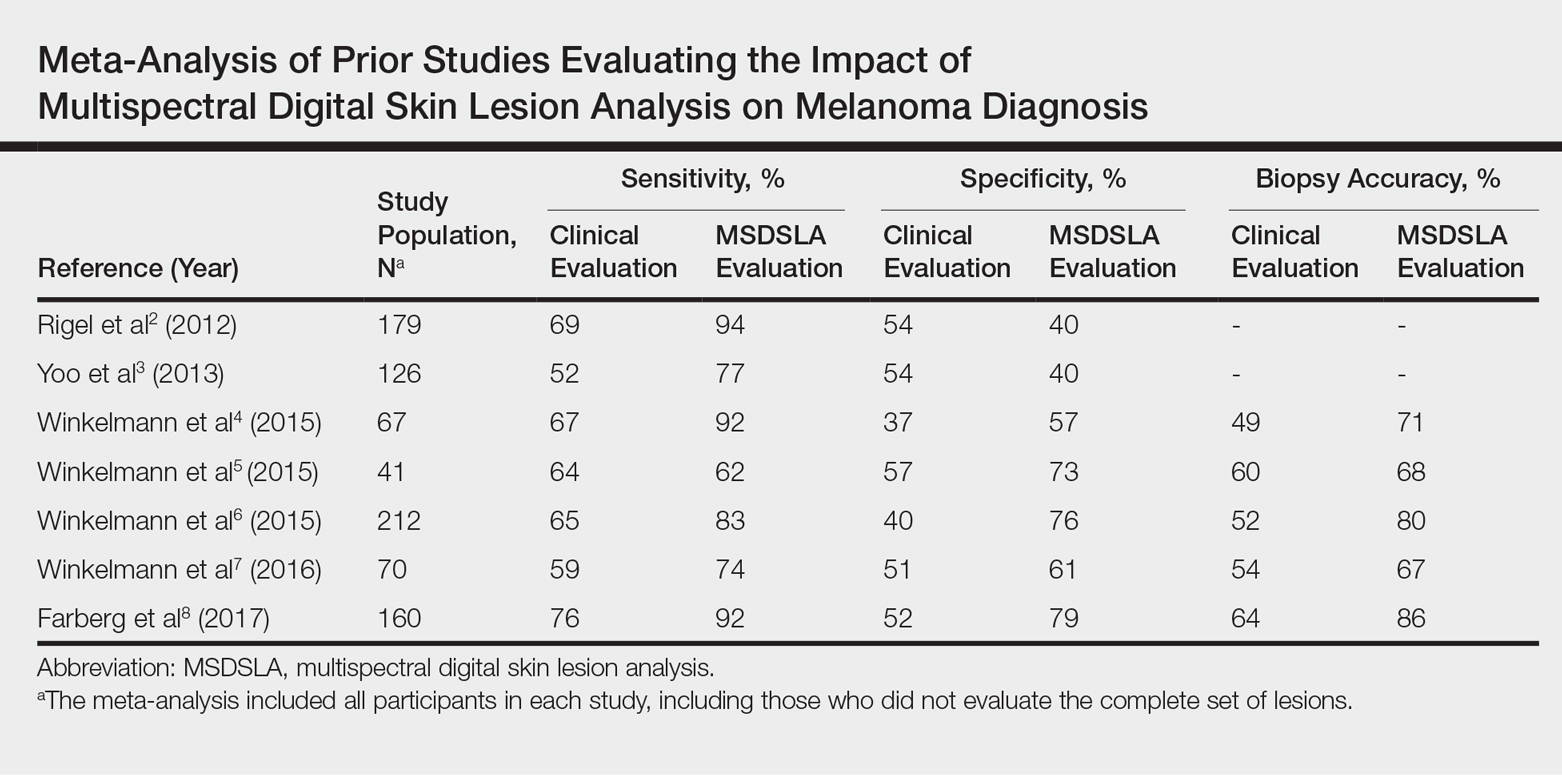
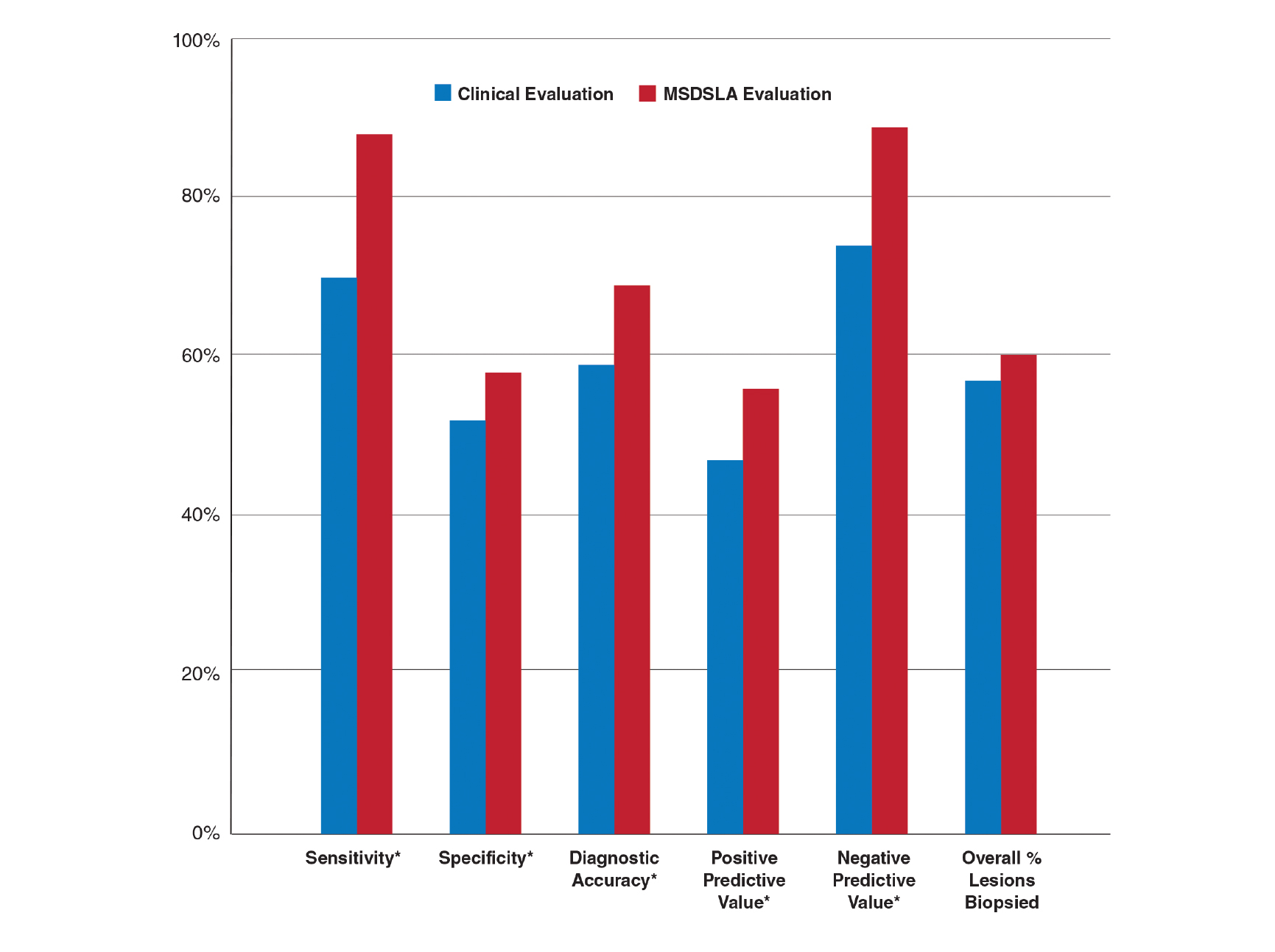
Overall sensitivity for the detection of melanoma or other high-grade PSLs improved from 70% on clinical and dermoscopic evaluation to 88% after MSDSLA information was provided (P<.0001), and specificity increased from 52% to 58% (P<.001). Diagnostic accuracy also improved from 59% on clinical evaluation to 69% after review of MSDSLA findings (P<.0001). The positive predictive value of biopsy decisions was 47% following clinical evaluation, which improved to 56% after evaluation of MSDSLA findings (P<.001), and the negative predictive value increased from 74% to 89% (P<.0001). The overall percentage of lesions selected for biopsy did not significantly change following MSDSLA data integration (57% vs 60%)(Figure). Given that similar numbers of lesions were biopsied with improved sensitivity and specificity, the integration of MSDSLA data into the biopsy decision led to an improved biopsy ratio (ratio of melanomas biopsied to total biopsies) and fewer unnecessary biopsies.
Comment
Our broad analysis further supported the findings of prior studies that decisions to biopsy clinically suspicious PSLs are more sensitive, specific, and accurate when practitioners are provided MSDSLA information following clinical examination.2-8
Given the evolution in health care economics, it is clear that greater emphasis will continue to be placed on superior, evidence-based, effective care. The reported diagnostic sensitivities and specificities of clinical evaluation and dermoscopy for melanoma detection vary widely throughout the literature, with sensitivities ranging from 58% to over 90% and specificities ranging from 77% to 99%.9-11
Our study had several limitations. For this analysis to be more representative of lesion biopsy selection in the clinical setting, biopsy sensitivity (correctly identifying lesions appropriate for biopsy) vs melanoma sensitivity (identifying a lesion as melanoma) was used.13 The overall sensitivity found was within the range of prior studies,2-8 but this approach may have potentially led to a lower specificity due to an increased number of lesions biopsied. Additionally, the melanomas selected for these studies were early (malignant melanoma in situ or mean thickness of invasive malignant melanoma of 0.3 mm), and the nonmelanomas (including low-grade dysplastic nevi) were not necessarily diagnostically straightforward. This may have led to the clinical and dermoscopic sensitivity and specificity noted being lower than in some prior studies.9-11
The risk of missing a melanoma with MSDSLA devices has led manufacturers to strive for a high sensitivity for their devices, leading to lower specificity as a consequence. For this reason and other ambiguous practical considerations (eg, device and patient costs, difficulty with insurance reimbursement), the adoption of this technology into routine clinical practice has remained relatively static; however, using enhanced diagnostic technologies such as MSDSLA may help with more accurate identification of high-risk PSLs, thereby leading to earlier detection and overall less expensive, more cost-effective treatment of melanoma.
- Monheit G, Cognetta AB, Ferris L, et al. The performance of MelaFind: a prospective multicenter study. Arch Dermatol. 2011;147:188-194.
- Rigel DS, Roy M, Yoo J, et al. Impact of guidance from a computer-aided multispectral digital skin lesion analysis device on decision to biopsy lesions clinically suggestive of melanoma. Arch Dermatol. 2012;148:541-543.
- Yoo J, Rigel DS, Roy M, et al. Impact of guidance from a multispectral digital skin lesion analysis device on dermatology residents decisions to biopsy lesions clinically suggestive of melanoma. J Am Acad Dermatol. 2013;68:AB152.
- Winkelmann RR, Yoo J, Tucker N, et al. Impact of guidance provided by a multispectral digital skin lesion analysis device following dermoscopy on decisions to biopsy atypical melanocytic lesions. J Clin Aesthet Dermatol. 2015;8:21-24.
- Winkelmann RR, Hauschild A, Tucker N, et al. The impact of multispectral digital skin lesion analysis on German dermatologist decisions to biopsy atypical pigmented lesions with clinical characteristics of melanoma. J Clin Aesthet Dermatol. 2015;8:27-29.
- Winkelmann RR, Tucker N, White R, et al. Pigmented skin lesion biopsies after computer-aided multispectral digital skin lesion analysis. J Am Osteopath Assoc. 2015;115:666-669.
- Winkelmann RR, Farberg AS, Tucker N, et al. Enhancement of international dermatologists’ pigmented skin lesion biopsy decisions following dermoscopy with subsequent integration of multispectral digital skin lesion analysis [published online July 1, 2016]. J Clin Aesthet Dermatol. 2016;9:53-55.
- Farberg AS, Winkelmann RR, Tucker N, et al. The impact of quantitative data provided by a multi-spectral digital skin lesion analysis device on dermatologists’ decisions to biopsy pigmented lesions [published online September 1, 2017]. J Clin Aesthet Dermatol. 2017;10:24-26.
- Wolf IH, Smolle J, Soyer HP, et al. Sensitivity in the clinical diagnosis of malignant melanoma. Melanoma Res. 1998;8:425-429.
- Kittler H, Pehamberger H, Wolff K, et al. Diagnostic accuracy of dermoscopy. Lancet Oncol. 2002;3:159-165.
- Ascierto PA, Palmieri G, Celentano E, et al. Sensitivity and specificity of epiluminescence microscopy: evaluation on a sample of 2731 excised cutaneous pigmented lesions: the Melanoma Cooperative Study. Br J Dermatol. 2000;142:893-898.
- Carli P, Nardini P, Crocetti E, et al. Frequency and characteristics of melanomas missed at a pigmented lesion clinic: a registry-based study. Melanoma Res. 2004;14:403-407.
- Friedman RJ, Gutkowicz-Krusin D, Farber MJ, et al. The diagnostic performance of expert dermoscopists vs a computer-vision system on small-diameter melanomas. Arch Dermatol. 2008;144:476-482.
Early detection of melanoma, which is known to improve survival rates, remains a challenge for dermatologists. Suspicious pigmented lesions typically are evaluated via clinical examination and dermoscopy; however, new technologies are being developed to provide additional objective information for clinicians to incorporate into their biopsy decisions.
Multispectral digital skin lesion analysis (MSDSLA) uses 10 bands of visible and near-infrared light (430–950 nm) to image and analyze pigmented skin lesions (PSLs) down to 2.5 mm below the skin surface and measures the distribution of melanin using 75 unique algorithms to determine the degree of the morphologic disorder. Using a logical regression model previously validated on a set of 1632 PSLs, the probability of melanoma and probability of being a melanoma/PSL of high-risk malignant potential are then provided to the clinician.1
In this study, we analyzed aggregate data from 7 prior studies2-8 to better determine how MSDSLA impacts the biopsy decisions of dermatologists and nondermatologists following clinical examination and dermoscopic evaluation of PSLs.
Methods

Results
Overall sensitivity for the detection of melanoma or other high-grade PSLs improved from 70% on clinical and dermoscopic evaluation to 88% after MSDSLA information was provided (P<.0001), and specificity increased from 52% to 58% (P<.001). Diagnostic accuracy also improved from 59% on clinical evaluation to 69% after review of MSDSLA findings (P<.0001). The positive predictive value of biopsy decisions was 47% following clinical evaluation, which improved to 56% after evaluation of MSDSLA findings (P<.001), and the negative predictive value increased from 74% to 89% (P<.0001). The overall percentage of lesions selected for biopsy did not significantly change following MSDSLA data integration (57% vs 60%)(Figure). Given that similar numbers of lesions were biopsied with improved sensitivity and specificity, the integration of MSDSLA data into the biopsy decision led to an improved biopsy ratio (ratio of melanomas biopsied to total biopsies) and fewer unnecessary biopsies.

Comment
Our broad analysis further supported the findings of prior studies that decisions to biopsy clinically suspicious PSLs are more sensitive, specific, and accurate when practitioners are provided MSDSLA information following clinical examination.2-8
Given the evolution in health care economics, it is clear that greater emphasis will continue to be placed on superior, evidence-based, effective care. The reported diagnostic sensitivities and specificities of clinical evaluation and dermoscopy for melanoma detection vary widely throughout the literature, with sensitivities ranging from 58% to over 90% and specificities ranging from 77% to 99%.9-11
Our study had several limitations. For this analysis to be more representative of lesion biopsy selection in the clinical setting, biopsy sensitivity (correctly identifying lesions appropriate for biopsy) vs melanoma sensitivity (identifying a lesion as melanoma) was used.13 The overall sensitivity found was within the range of prior studies,2-8 but this approach may have potentially led to a lower specificity due to an increased number of lesions biopsied. Additionally, the melanomas selected for these studies were early (malignant melanoma in situ or mean thickness of invasive malignant melanoma of 0.3 mm), and the nonmelanomas (including low-grade dysplastic nevi) were not necessarily diagnostically straightforward. This may have led to the clinical and dermoscopic sensitivity and specificity noted being lower than in some prior studies.9-11
The risk of missing a melanoma with MSDSLA devices has led manufacturers to strive for a high sensitivity for their devices, leading to lower specificity as a consequence. For this reason and other ambiguous practical considerations (eg, device and patient costs, difficulty with insurance reimbursement), the adoption of this technology into routine clinical practice has remained relatively static; however, using enhanced diagnostic technologies such as MSDSLA may help with more accurate identification of high-risk PSLs, thereby leading to earlier detection and overall less expensive, more cost-effective treatment of melanoma.
Early detection of melanoma, which is known to improve survival rates, remains a challenge for dermatologists. Suspicious pigmented lesions typically are evaluated via clinical examination and dermoscopy; however, new technologies are being developed to provide additional objective information for clinicians to incorporate into their biopsy decisions.
Multispectral digital skin lesion analysis (MSDSLA) uses 10 bands of visible and near-infrared light (430–950 nm) to image and analyze pigmented skin lesions (PSLs) down to 2.5 mm below the skin surface and measures the distribution of melanin using 75 unique algorithms to determine the degree of the morphologic disorder. Using a logical regression model previously validated on a set of 1632 PSLs, the probability of melanoma and probability of being a melanoma/PSL of high-risk malignant potential are then provided to the clinician.1
In this study, we analyzed aggregate data from 7 prior studies2-8 to better determine how MSDSLA impacts the biopsy decisions of dermatologists and nondermatologists following clinical examination and dermoscopic evaluation of PSLs.
Methods

Results
Overall sensitivity for the detection of melanoma or other high-grade PSLs improved from 70% on clinical and dermoscopic evaluation to 88% after MSDSLA information was provided (P<.0001), and specificity increased from 52% to 58% (P<.001). Diagnostic accuracy also improved from 59% on clinical evaluation to 69% after review of MSDSLA findings (P<.0001). The positive predictive value of biopsy decisions was 47% following clinical evaluation, which improved to 56% after evaluation of MSDSLA findings (P<.001), and the negative predictive value increased from 74% to 89% (P<.0001). The overall percentage of lesions selected for biopsy did not significantly change following MSDSLA data integration (57% vs 60%)(Figure). Given that similar numbers of lesions were biopsied with improved sensitivity and specificity, the integration of MSDSLA data into the biopsy decision led to an improved biopsy ratio (ratio of melanomas biopsied to total biopsies) and fewer unnecessary biopsies.

Comment
Our broad analysis further supported the findings of prior studies that decisions to biopsy clinically suspicious PSLs are more sensitive, specific, and accurate when practitioners are provided MSDSLA information following clinical examination.2-8
Given the evolution in health care economics, it is clear that greater emphasis will continue to be placed on superior, evidence-based, effective care. The reported diagnostic sensitivities and specificities of clinical evaluation and dermoscopy for melanoma detection vary widely throughout the literature, with sensitivities ranging from 58% to over 90% and specificities ranging from 77% to 99%.9-11
Our study had several limitations. For this analysis to be more representative of lesion biopsy selection in the clinical setting, biopsy sensitivity (correctly identifying lesions appropriate for biopsy) vs melanoma sensitivity (identifying a lesion as melanoma) was used.13 The overall sensitivity found was within the range of prior studies,2-8 but this approach may have potentially led to a lower specificity due to an increased number of lesions biopsied. Additionally, the melanomas selected for these studies were early (malignant melanoma in situ or mean thickness of invasive malignant melanoma of 0.3 mm), and the nonmelanomas (including low-grade dysplastic nevi) were not necessarily diagnostically straightforward. This may have led to the clinical and dermoscopic sensitivity and specificity noted being lower than in some prior studies.9-11
The risk of missing a melanoma with MSDSLA devices has led manufacturers to strive for a high sensitivity for their devices, leading to lower specificity as a consequence. For this reason and other ambiguous practical considerations (eg, device and patient costs, difficulty with insurance reimbursement), the adoption of this technology into routine clinical practice has remained relatively static; however, using enhanced diagnostic technologies such as MSDSLA may help with more accurate identification of high-risk PSLs, thereby leading to earlier detection and overall less expensive, more cost-effective treatment of melanoma.
- Monheit G, Cognetta AB, Ferris L, et al. The performance of MelaFind: a prospective multicenter study. Arch Dermatol. 2011;147:188-194.
- Rigel DS, Roy M, Yoo J, et al. Impact of guidance from a computer-aided multispectral digital skin lesion analysis device on decision to biopsy lesions clinically suggestive of melanoma. Arch Dermatol. 2012;148:541-543.
- Yoo J, Rigel DS, Roy M, et al. Impact of guidance from a multispectral digital skin lesion analysis device on dermatology residents decisions to biopsy lesions clinically suggestive of melanoma. J Am Acad Dermatol. 2013;68:AB152.
- Winkelmann RR, Yoo J, Tucker N, et al. Impact of guidance provided by a multispectral digital skin lesion analysis device following dermoscopy on decisions to biopsy atypical melanocytic lesions. J Clin Aesthet Dermatol. 2015;8:21-24.
- Winkelmann RR, Hauschild A, Tucker N, et al. The impact of multispectral digital skin lesion analysis on German dermatologist decisions to biopsy atypical pigmented lesions with clinical characteristics of melanoma. J Clin Aesthet Dermatol. 2015;8:27-29.
- Winkelmann RR, Tucker N, White R, et al. Pigmented skin lesion biopsies after computer-aided multispectral digital skin lesion analysis. J Am Osteopath Assoc. 2015;115:666-669.
- Winkelmann RR, Farberg AS, Tucker N, et al. Enhancement of international dermatologists’ pigmented skin lesion biopsy decisions following dermoscopy with subsequent integration of multispectral digital skin lesion analysis [published online July 1, 2016]. J Clin Aesthet Dermatol. 2016;9:53-55.
- Farberg AS, Winkelmann RR, Tucker N, et al. The impact of quantitative data provided by a multi-spectral digital skin lesion analysis device on dermatologists’ decisions to biopsy pigmented lesions [published online September 1, 2017]. J Clin Aesthet Dermatol. 2017;10:24-26.
- Wolf IH, Smolle J, Soyer HP, et al. Sensitivity in the clinical diagnosis of malignant melanoma. Melanoma Res. 1998;8:425-429.
- Kittler H, Pehamberger H, Wolff K, et al. Diagnostic accuracy of dermoscopy. Lancet Oncol. 2002;3:159-165.
- Ascierto PA, Palmieri G, Celentano E, et al. Sensitivity and specificity of epiluminescence microscopy: evaluation on a sample of 2731 excised cutaneous pigmented lesions: the Melanoma Cooperative Study. Br J Dermatol. 2000;142:893-898.
- Carli P, Nardini P, Crocetti E, et al. Frequency and characteristics of melanomas missed at a pigmented lesion clinic: a registry-based study. Melanoma Res. 2004;14:403-407.
- Friedman RJ, Gutkowicz-Krusin D, Farber MJ, et al. The diagnostic performance of expert dermoscopists vs a computer-vision system on small-diameter melanomas. Arch Dermatol. 2008;144:476-482.
- Monheit G, Cognetta AB, Ferris L, et al. The performance of MelaFind: a prospective multicenter study. Arch Dermatol. 2011;147:188-194.
- Rigel DS, Roy M, Yoo J, et al. Impact of guidance from a computer-aided multispectral digital skin lesion analysis device on decision to biopsy lesions clinically suggestive of melanoma. Arch Dermatol. 2012;148:541-543.
- Yoo J, Rigel DS, Roy M, et al. Impact of guidance from a multispectral digital skin lesion analysis device on dermatology residents decisions to biopsy lesions clinically suggestive of melanoma. J Am Acad Dermatol. 2013;68:AB152.
- Winkelmann RR, Yoo J, Tucker N, et al. Impact of guidance provided by a multispectral digital skin lesion analysis device following dermoscopy on decisions to biopsy atypical melanocytic lesions. J Clin Aesthet Dermatol. 2015;8:21-24.
- Winkelmann RR, Hauschild A, Tucker N, et al. The impact of multispectral digital skin lesion analysis on German dermatologist decisions to biopsy atypical pigmented lesions with clinical characteristics of melanoma. J Clin Aesthet Dermatol. 2015;8:27-29.
- Winkelmann RR, Tucker N, White R, et al. Pigmented skin lesion biopsies after computer-aided multispectral digital skin lesion analysis. J Am Osteopath Assoc. 2015;115:666-669.
- Winkelmann RR, Farberg AS, Tucker N, et al. Enhancement of international dermatologists’ pigmented skin lesion biopsy decisions following dermoscopy with subsequent integration of multispectral digital skin lesion analysis [published online July 1, 2016]. J Clin Aesthet Dermatol. 2016;9:53-55.
- Farberg AS, Winkelmann RR, Tucker N, et al. The impact of quantitative data provided by a multi-spectral digital skin lesion analysis device on dermatologists’ decisions to biopsy pigmented lesions [published online September 1, 2017]. J Clin Aesthet Dermatol. 2017;10:24-26.
- Wolf IH, Smolle J, Soyer HP, et al. Sensitivity in the clinical diagnosis of malignant melanoma. Melanoma Res. 1998;8:425-429.
- Kittler H, Pehamberger H, Wolff K, et al. Diagnostic accuracy of dermoscopy. Lancet Oncol. 2002;3:159-165.
- Ascierto PA, Palmieri G, Celentano E, et al. Sensitivity and specificity of epiluminescence microscopy: evaluation on a sample of 2731 excised cutaneous pigmented lesions: the Melanoma Cooperative Study. Br J Dermatol. 2000;142:893-898.
- Carli P, Nardini P, Crocetti E, et al. Frequency and characteristics of melanomas missed at a pigmented lesion clinic: a registry-based study. Melanoma Res. 2004;14:403-407.
- Friedman RJ, Gutkowicz-Krusin D, Farber MJ, et al. The diagnostic performance of expert dermoscopists vs a computer-vision system on small-diameter melanomas. Arch Dermatol. 2008;144:476-482.
Practice Points
- Multispectral digital skin lesion analysis (MSDSLA) can be a valuable tool in the evaluation of pigmented skin lesions (PSLs).
- MSDSLA may help to better identify high-risk PSLs and improve cost of care.
Mohs Micrographic Surgery for Digital Melanoma and Nonmelanoma Skin Cancers
Mohs micrographic surgery (MMS) is a specialized surgical technique for the treatment of melanoma and nonmelanoma skin cancers (NMSCs).1-3 The procedure involves surgical excision, histopathologic examination, precise mapping of malignant tissue, and wound management. Indications for MMS in skin cancer patients include recurring lesions, lesions in high-risk anatomic locations, aggressive histologic subtypes (ie, morpheaform, micronodular, infiltrative, high-grade, poorly differentiated), perineural invasion, large lesion size (>2 cm in diameter), poorly defined lateral or vertical clinical borders, rapid growth of the lesion, immunocompromised status, and sites of positive margins on prior excision. The therapeutic advantages of MMS include tissue conservation and optimal margin control in cosmetically or functionally sensitive areas, such as acral sites (eg, hands, feet, digits).1,3
The intricacies of the nail apparatus complicate diagnostic biopsy and precise delineation of peripheral margins in digital skin cancers; thus, early diagnosis and intraoperative histologic examination of the margins are essential. Traditionally, the surgical approach to subungual cutaneous tumors such as melanoma has included digital amputation4; however, a study of the treatment of subungual melanoma revealed no difference in survival based on the level of amputation, therefore advocating for less radical treatment.4
Interestingly, MMS for cutaneous tumors localized to the digits is not frequently reviewed in the dermatologic literature. We present a retrospective case series evaluating the clinical outcomes of digital melanoma and NMSCs treated with MMS.
Methods
A retrospective chart review was performed at a private dermatology practice to identify patients who underwent MMS for melanoma or NMSC localized to the digits from January 2009 to December 2014. All patients were treated in the office by 1 Mohs surgeon (A.H.) and were evaluated before and after MMS. Data were collected from the electronic medical record of the practice, including patient demographics, histopathologic diagnosis, tumor status (primary or recurrent lesion), anatomic site of the tumor, preoperative and postoperative size of the lesion, number of MMS stages, surgical repair technique, postoperative complications, and follow-up period.
Results
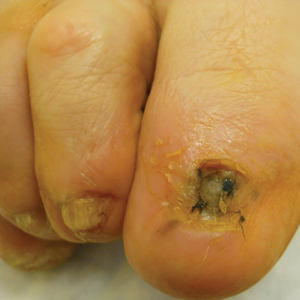
Twenty-seven patients (13 male, 14 female) with a total of 28 lesions (malignant melanoma or NMSC) localized to the digits were identified (Table). The mean age at the time of MMS was 64.07 years.
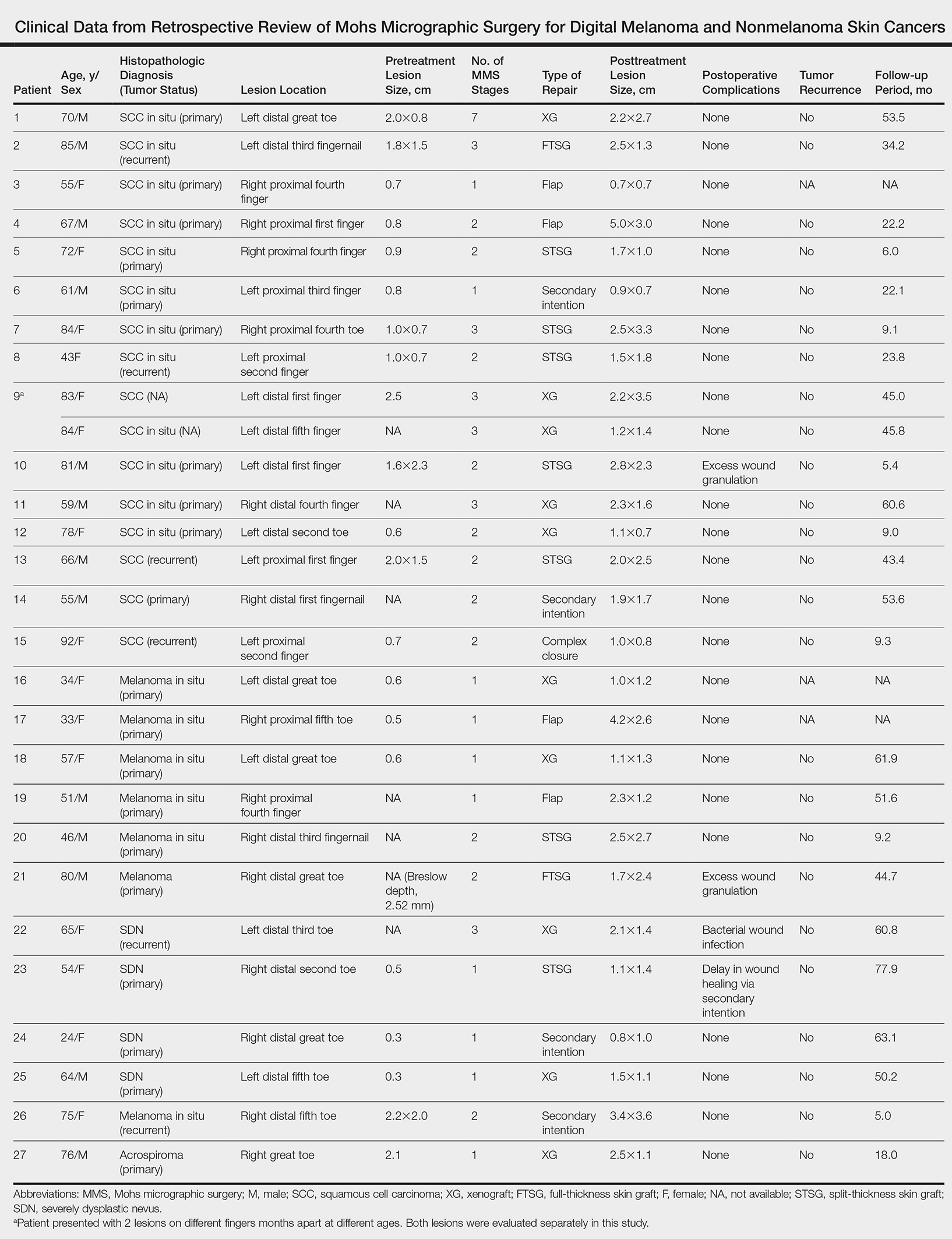
Surgical techniques used for repair following MMS included xenograft (10/28 [35.71%]); split-thickness skin graft (7/28 [25.0%]); secondary intention (4/28 [14.29%]); flap (4/28 [14.29%]); full-thickness skin graft (2/28 [7.14%]); and complex closure (1/28 [3.57%]). Clinical preoperative, operative, and postoperative photos from Patient 21 in this series are shown here (Figure). Two patients required bony phalanx resection due to invasion of the tumor into the periosteum: 1 had a malignant melanoma (Breslow depth, 2.52 mm); the other had an SCC. In addition, following removal of a severely dysplastic nevus, debulked tissue revealed melanoma in 1 patient.
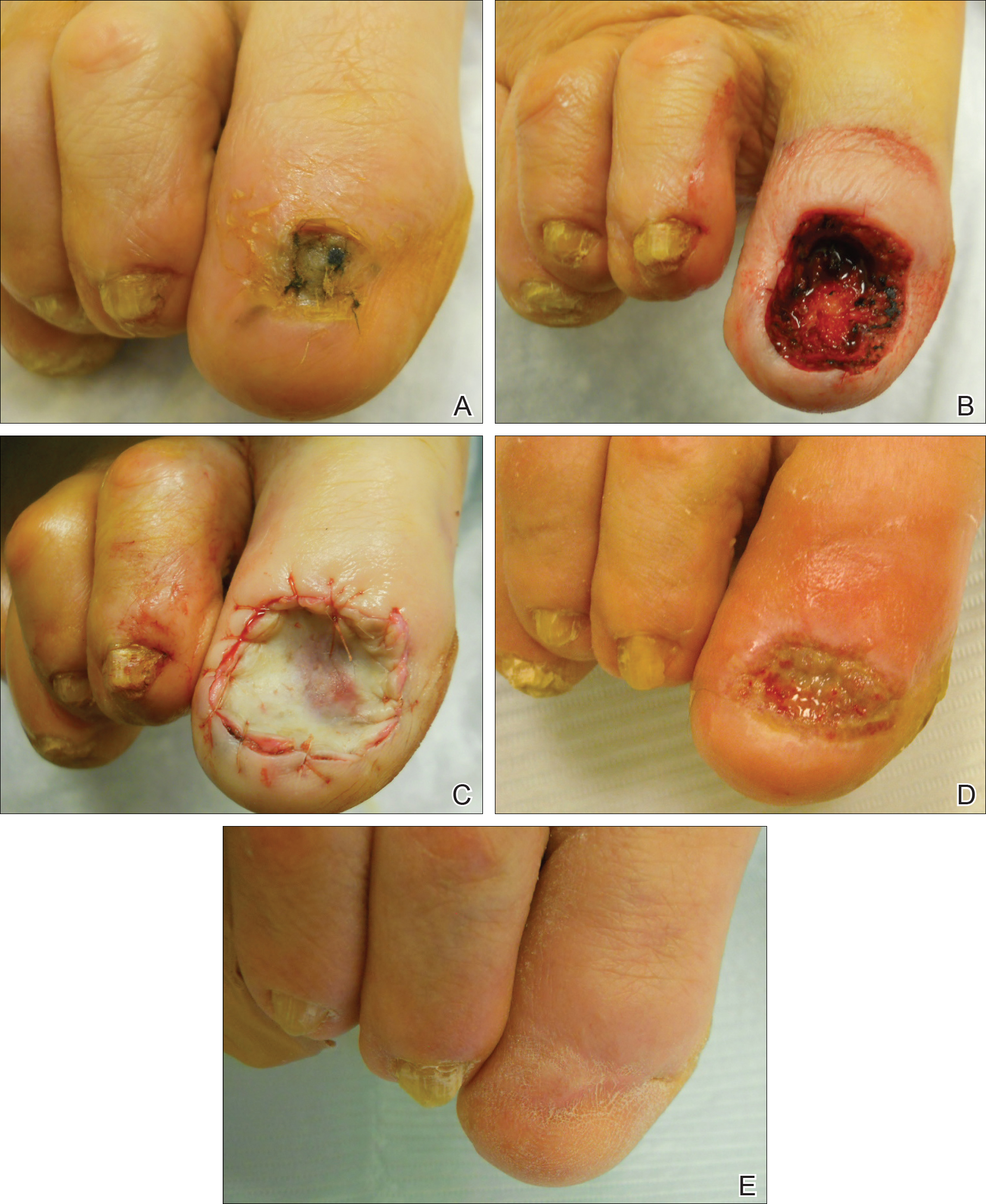
Postoperative complications were noted in 4 (14.29%) of 28 MMS procedures, including bacterial wound infection (3.57%), excess granulation tissue that required wound debridement (7.14%), and delay in wound healing (3.57%). Follow-up data were available for 25 of the 28 MMS procedures (mean follow-up, 35.4 months), during which no recurrences were observed.
Comment
Mohs micrographic surgery is a specialized technique used in the treatment of cutaneous tumors, including basal cell carcinoma, SCC, melanoma in situ, atypical fibroxanthoma, dermatofibrosarcoma protuberans, sebaceous carcinoma, microcystic adnexal carcinoma, and Merkel cell carcinoma, among other cutaneous tumors.1-3 Mohs micrographic surgery provides the advantage of tissue conservation as well as optimal margin control in cosmetically or functionally sensitive areas while providing a higher cure rate than surgical excision. During the procedure, the surgical margin is examined histologically, thus ensuring definitive removal of the tumor but minimal loss of surrounding normal tissue.1-3 Mohs micrographic surgery is particularly useful for treating lesions on acral sites (eg, hands, feet, and digits).3-5
The treatment of digital skin cancers has evolved over the past 50 years with advancements resulting in more precise, tissue-sparing methods, in contrast to previous treatments such as amputation and wide local excision.6 More specifically, traditional digital amputation for the treatment of subungual melanoma has been reevaluated in multiple studies, which did not demonstrate a statistically significant difference in survival based on the level of amputation, thereby favoring less radical treatment.4,6 Moehrle et al7 found no statistical difference in recurrence rate when comparing patients with digital melanomas treated with partial amputation and those treated with digit-sparing surgery with limited excision and histologic evaluation of margins. Additionally, in a study conducted by Lazar et al,8 no recurrence of 13 subungual malignancies treated with MMS that utilized a full-thickness graft was reported at 4-year follow-up. In a large retrospective series of digital melanomas treated with MMS, Terushkin et al5 reported that 96.5% (55/57) of patients with primary melanomas that were treated with MMS avoided amputation, and the 5- and 10-year melanoma-specific survival rates for all patients treated with MMS were 95.0% and 82.6%, respectively.
In our study, cutaneous malignancies were located most often on the fingers, and the most common skin cancer identified was SCC in situ. The literature has shown that SCC in situ and SCC are the most common cutaneous neoplasms of the digits and nail unit.9 The most common specific anatomic site of cutaneous malignancy in our study was the great toe, followed by the fourth finger. A study conducted by Tan et al9 revealed that the great toe was the most common location of melanoma of the nail bed and subungual region, followed by the thumb. In contrast, primary subungual SCCs occur most frequently on the finger, with rare cases involving the toes.10
The etiology of digital SCC may involve extensive sun exposure, chronic trauma and wounds, and viral infection.9,11 More specifically, the dermatologic literature provides evidence of human papillomavirus (HPV) type 16 involvement in the pathogenesis of digital and periungual SCC. A genital-digital mechanism of spread has been implicated.11,12 An increased recurrence rate of HPV-associated digital SCCs has been reported following MMS, likely secondary to residual postsurgical HPV infection.11,12
Maintaining function and cosmesis of the hands, feet, and digits following MMS can be challenging, sometimes requiring skin grafts and flaps to close the defect. In the 28 MMS procedures evaluated in our study, 19 (67.9%) surgical defects were repaired with a graft (ie, split-thickness skin graft, full-thickness skin graft, xenograft), 4 (14.3%) with a flap (advancement and rotation), 4 (14.3%) by secondary intention, and 1 (3.6%) with primary complex closure.
Surgical grafts can be categorized based on the origin of the graft.2,13 Autografts, derived from the patient’s skin, are the most frequently used dermatologic graft and can be further categorized as full-thickness skin grafts, which include the epidermis and the entire dermis, thus preserving adnexal structures, and split-thickness skin grafts, which include the epidermis and partial dermis.2,13
A cross-sectional survey of fellowship-trained Mohs surgeons revealed that more than two-thirds of repairs for cutaneous acral cancers were performed using a primary closure technique, and one-fourth of closures were performed using secondary intention.15 Of the less frequently utilized skin-graft repairs, more were for acral lesions on the legs than on the arms.14 The type of procedure and graft used is dependent on multiple variables, including the anatomic location of the lesion and final size of the defect following MMS.2 Similarly, the use of specific types of sutures depends on the anatomic location of the lesion, relative thickness of the skin, degree of tension, and desired cosmetic result.15 The expertise of a hand surgeon may be required, particularly in cases in which the extensor tendon of the distal interphalangeal joint is compromised, manifested by a droopy fingertip when the hand is held horizontally. Additionally, special attention should be paid to removing the entire nail matrix before skin grafting for subungual tumors to avoid nail growth under the skin graft.
Evaluation of debulked tissue from digital skin cancers proved to be important in our study. In Patient 21, debulked tissue revealed melanoma following removal of a severely dysplastic nevus. This finding emphasizes the importance of complete excision of such lesions, as remaining underlying portions of the lesion can reveal residual tumor of the same or different histopathology.
In a prospective study, MMS was shown to have a low rate (0.91%; 95% confidence interval, 0.38%-1.45%) of surgical site infection in the absence of prophylactic antibiotics.16 The highest rates of surgical site infection were closely associated with flap closure. In our study, most patients had an uncomplicated and successful postoperative recovery. Only 1 (3.57%) of the 28 MMS procedures (Patient 22) was complicated by a bacterial wound infection postoperatively. The lesion removed in this case was a severely dysplastic melanocytic nevus on the toe. Infection resolved after a course of oral antibiotics, but the underlying cause of the wound infection in the patient was unclear. Other postoperative complications in our study included delayed wound healing and excess granulation tissue requiring wound debridement.
There are limited data in the dermatologic literature regarding outcomes following MMS for the treatment of cutaneous malignancies localized to the digits.
Additional limitations of this case review include its single-center and retrospective design, the small sample size, and 1 Mohs surgeon having performed all surgeries.
Conclusion
This study provides further evidence of the benefit of MMS for the treatment of malignant melanoma and NMSCs of the digits. This procedure provides margin-controlled excision of these malignant neoplasms while preserving maximal normal tissue, thereby providing patients with improved postoperative function and cosmesis. Long-term follow-up data demonstrating a lack of tumor recurrence underscores the assertion that MMS is safe and effective for the treatment of skin cancer of the digits.
- Dim-Jamora KC, Perone JB. Management of cutaneous tumors with mohs micrographic surgery. Semin Plast Surg. 2008;22:247-256.
- McLeod MP, Choudhary S, Alqubaisy YA, et al. Indications for Mohs micrographic surgery. In: Nouri K, ed. Mohs Micrographic Surgery. New York, NY: Springer; 2012:5-13.
- Loosemore MP, Morales-Burgos A, Goldberg LH. Acral lentiginous melanoma of the toe treated using Mohs surgery with sparing of the digit and subsequent reconstruction using split-thickness skin graft. Dermatol Surg. 2013;39:136-138.
- Rayatt SS, Dancey AL, Davison PM. Thumb subungual melanoma: is amputation necessary? J Plast Reconstr Aesthet Surg. 2007;60:635-638.
- Terushkin V, Brodland DG, Sharon DJ, et al. Digit-sparing Mohs surgery for melanoma. Dermatol Surg. 2016;42:83-93.
- Viola KV, Jhaveri MB, Soulos PR, et al. Mohs micrographic surgery and surgical excision for nonmelanoma skin cancer treatment in the Medicare population. Arch Dermatol. 2012;148:473-477.
- Moehrle M, Metzger S, Schippert W. “Functional” surgery in subungual melanoma. Dermatol Surg. 2003;29:366-374.
- Lazar A, Abimelec P, Dumontier C, et al. Full thickness skin graft from nail unit reconstruction. J Hand Surg Br. 2005;30:194-198.
- Tan KB, Moncrieff M, Thompson JF, et al. Subungual melanoma: a study of 124 cases highlighting features of early lesions, potential for histologic reports. Am J Surg Pathol. 2007;31:1902-1912.
- Nasca MR, Innocenzi D, Micali G. Subungual squamous cell carcinoma of the toe: report on three cases. Dermatol Surg. 2004;30:345-348.
- Dika E, Piraccini BM, Balestri RR, et al. Mohs surgery for squamous cell carcinoma of the nail: report of 15 cases. our experience and a long-term follow-up. Br J Dermatol. 2012;167:1310-1314.
- Alam M, Caldwell JB, Eliezri YD. Human papillomavirus-associated digital squamous cell carcinoma: literature review and report of 21 new cases. J Am Acad Dermatol. 2003;48:385-393.
- Filho L, Anselmo J, Dadalti P, et al. Skin grafts in cutaneous oncology. Braz Ann Dermatol. 2006;81:465-472.
- Raimer DW, Group AR, Petitt MS, et al. Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17:1.
- Alam M, Helenowksi IB, Cohen JL, et al. Association between type of reconstruction after Mohs micrographic surgery and surgeon-, patient-, and tumor-specific features: a cross-sectional study. Dermatol Surg. 2013;39:51-55.
- Rogers HD, Desciak EB, Marcus RP, et al. Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol. 2010;63:842-851.
Mohs micrographic surgery (MMS) is a specialized surgical technique for the treatment of melanoma and nonmelanoma skin cancers (NMSCs).1-3 The procedure involves surgical excision, histopathologic examination, precise mapping of malignant tissue, and wound management. Indications for MMS in skin cancer patients include recurring lesions, lesions in high-risk anatomic locations, aggressive histologic subtypes (ie, morpheaform, micronodular, infiltrative, high-grade, poorly differentiated), perineural invasion, large lesion size (>2 cm in diameter), poorly defined lateral or vertical clinical borders, rapid growth of the lesion, immunocompromised status, and sites of positive margins on prior excision. The therapeutic advantages of MMS include tissue conservation and optimal margin control in cosmetically or functionally sensitive areas, such as acral sites (eg, hands, feet, digits).1,3
The intricacies of the nail apparatus complicate diagnostic biopsy and precise delineation of peripheral margins in digital skin cancers; thus, early diagnosis and intraoperative histologic examination of the margins are essential. Traditionally, the surgical approach to subungual cutaneous tumors such as melanoma has included digital amputation4; however, a study of the treatment of subungual melanoma revealed no difference in survival based on the level of amputation, therefore advocating for less radical treatment.4
Interestingly, MMS for cutaneous tumors localized to the digits is not frequently reviewed in the dermatologic literature. We present a retrospective case series evaluating the clinical outcomes of digital melanoma and NMSCs treated with MMS.
Methods
A retrospective chart review was performed at a private dermatology practice to identify patients who underwent MMS for melanoma or NMSC localized to the digits from January 2009 to December 2014. All patients were treated in the office by 1 Mohs surgeon (A.H.) and were evaluated before and after MMS. Data were collected from the electronic medical record of the practice, including patient demographics, histopathologic diagnosis, tumor status (primary or recurrent lesion), anatomic site of the tumor, preoperative and postoperative size of the lesion, number of MMS stages, surgical repair technique, postoperative complications, and follow-up period.
Results
Twenty-seven patients (13 male, 14 female) with a total of 28 lesions (malignant melanoma or NMSC) localized to the digits were identified (Table). The mean age at the time of MMS was 64.07 years.

Surgical techniques used for repair following MMS included xenograft (10/28 [35.71%]); split-thickness skin graft (7/28 [25.0%]); secondary intention (4/28 [14.29%]); flap (4/28 [14.29%]); full-thickness skin graft (2/28 [7.14%]); and complex closure (1/28 [3.57%]). Clinical preoperative, operative, and postoperative photos from Patient 21 in this series are shown here (Figure). Two patients required bony phalanx resection due to invasion of the tumor into the periosteum: 1 had a malignant melanoma (Breslow depth, 2.52 mm); the other had an SCC. In addition, following removal of a severely dysplastic nevus, debulked tissue revealed melanoma in 1 patient.

Postoperative complications were noted in 4 (14.29%) of 28 MMS procedures, including bacterial wound infection (3.57%), excess granulation tissue that required wound debridement (7.14%), and delay in wound healing (3.57%). Follow-up data were available for 25 of the 28 MMS procedures (mean follow-up, 35.4 months), during which no recurrences were observed.
Comment
Mohs micrographic surgery is a specialized technique used in the treatment of cutaneous tumors, including basal cell carcinoma, SCC, melanoma in situ, atypical fibroxanthoma, dermatofibrosarcoma protuberans, sebaceous carcinoma, microcystic adnexal carcinoma, and Merkel cell carcinoma, among other cutaneous tumors.1-3 Mohs micrographic surgery provides the advantage of tissue conservation as well as optimal margin control in cosmetically or functionally sensitive areas while providing a higher cure rate than surgical excision. During the procedure, the surgical margin is examined histologically, thus ensuring definitive removal of the tumor but minimal loss of surrounding normal tissue.1-3 Mohs micrographic surgery is particularly useful for treating lesions on acral sites (eg, hands, feet, and digits).3-5
The treatment of digital skin cancers has evolved over the past 50 years with advancements resulting in more precise, tissue-sparing methods, in contrast to previous treatments such as amputation and wide local excision.6 More specifically, traditional digital amputation for the treatment of subungual melanoma has been reevaluated in multiple studies, which did not demonstrate a statistically significant difference in survival based on the level of amputation, thereby favoring less radical treatment.4,6 Moehrle et al7 found no statistical difference in recurrence rate when comparing patients with digital melanomas treated with partial amputation and those treated with digit-sparing surgery with limited excision and histologic evaluation of margins. Additionally, in a study conducted by Lazar et al,8 no recurrence of 13 subungual malignancies treated with MMS that utilized a full-thickness graft was reported at 4-year follow-up. In a large retrospective series of digital melanomas treated with MMS, Terushkin et al5 reported that 96.5% (55/57) of patients with primary melanomas that were treated with MMS avoided amputation, and the 5- and 10-year melanoma-specific survival rates for all patients treated with MMS were 95.0% and 82.6%, respectively.
In our study, cutaneous malignancies were located most often on the fingers, and the most common skin cancer identified was SCC in situ. The literature has shown that SCC in situ and SCC are the most common cutaneous neoplasms of the digits and nail unit.9 The most common specific anatomic site of cutaneous malignancy in our study was the great toe, followed by the fourth finger. A study conducted by Tan et al9 revealed that the great toe was the most common location of melanoma of the nail bed and subungual region, followed by the thumb. In contrast, primary subungual SCCs occur most frequently on the finger, with rare cases involving the toes.10
The etiology of digital SCC may involve extensive sun exposure, chronic trauma and wounds, and viral infection.9,11 More specifically, the dermatologic literature provides evidence of human papillomavirus (HPV) type 16 involvement in the pathogenesis of digital and periungual SCC. A genital-digital mechanism of spread has been implicated.11,12 An increased recurrence rate of HPV-associated digital SCCs has been reported following MMS, likely secondary to residual postsurgical HPV infection.11,12
Maintaining function and cosmesis of the hands, feet, and digits following MMS can be challenging, sometimes requiring skin grafts and flaps to close the defect. In the 28 MMS procedures evaluated in our study, 19 (67.9%) surgical defects were repaired with a graft (ie, split-thickness skin graft, full-thickness skin graft, xenograft), 4 (14.3%) with a flap (advancement and rotation), 4 (14.3%) by secondary intention, and 1 (3.6%) with primary complex closure.
Surgical grafts can be categorized based on the origin of the graft.2,13 Autografts, derived from the patient’s skin, are the most frequently used dermatologic graft and can be further categorized as full-thickness skin grafts, which include the epidermis and the entire dermis, thus preserving adnexal structures, and split-thickness skin grafts, which include the epidermis and partial dermis.2,13
A cross-sectional survey of fellowship-trained Mohs surgeons revealed that more than two-thirds of repairs for cutaneous acral cancers were performed using a primary closure technique, and one-fourth of closures were performed using secondary intention.15 Of the less frequently utilized skin-graft repairs, more were for acral lesions on the legs than on the arms.14 The type of procedure and graft used is dependent on multiple variables, including the anatomic location of the lesion and final size of the defect following MMS.2 Similarly, the use of specific types of sutures depends on the anatomic location of the lesion, relative thickness of the skin, degree of tension, and desired cosmetic result.15 The expertise of a hand surgeon may be required, particularly in cases in which the extensor tendon of the distal interphalangeal joint is compromised, manifested by a droopy fingertip when the hand is held horizontally. Additionally, special attention should be paid to removing the entire nail matrix before skin grafting for subungual tumors to avoid nail growth under the skin graft.
Evaluation of debulked tissue from digital skin cancers proved to be important in our study. In Patient 21, debulked tissue revealed melanoma following removal of a severely dysplastic nevus. This finding emphasizes the importance of complete excision of such lesions, as remaining underlying portions of the lesion can reveal residual tumor of the same or different histopathology.
In a prospective study, MMS was shown to have a low rate (0.91%; 95% confidence interval, 0.38%-1.45%) of surgical site infection in the absence of prophylactic antibiotics.16 The highest rates of surgical site infection were closely associated with flap closure. In our study, most patients had an uncomplicated and successful postoperative recovery. Only 1 (3.57%) of the 28 MMS procedures (Patient 22) was complicated by a bacterial wound infection postoperatively. The lesion removed in this case was a severely dysplastic melanocytic nevus on the toe. Infection resolved after a course of oral antibiotics, but the underlying cause of the wound infection in the patient was unclear. Other postoperative complications in our study included delayed wound healing and excess granulation tissue requiring wound debridement.
There are limited data in the dermatologic literature regarding outcomes following MMS for the treatment of cutaneous malignancies localized to the digits.
Additional limitations of this case review include its single-center and retrospective design, the small sample size, and 1 Mohs surgeon having performed all surgeries.
Conclusion
This study provides further evidence of the benefit of MMS for the treatment of malignant melanoma and NMSCs of the digits. This procedure provides margin-controlled excision of these malignant neoplasms while preserving maximal normal tissue, thereby providing patients with improved postoperative function and cosmesis. Long-term follow-up data demonstrating a lack of tumor recurrence underscores the assertion that MMS is safe and effective for the treatment of skin cancer of the digits.
Mohs micrographic surgery (MMS) is a specialized surgical technique for the treatment of melanoma and nonmelanoma skin cancers (NMSCs).1-3 The procedure involves surgical excision, histopathologic examination, precise mapping of malignant tissue, and wound management. Indications for MMS in skin cancer patients include recurring lesions, lesions in high-risk anatomic locations, aggressive histologic subtypes (ie, morpheaform, micronodular, infiltrative, high-grade, poorly differentiated), perineural invasion, large lesion size (>2 cm in diameter), poorly defined lateral or vertical clinical borders, rapid growth of the lesion, immunocompromised status, and sites of positive margins on prior excision. The therapeutic advantages of MMS include tissue conservation and optimal margin control in cosmetically or functionally sensitive areas, such as acral sites (eg, hands, feet, digits).1,3
The intricacies of the nail apparatus complicate diagnostic biopsy and precise delineation of peripheral margins in digital skin cancers; thus, early diagnosis and intraoperative histologic examination of the margins are essential. Traditionally, the surgical approach to subungual cutaneous tumors such as melanoma has included digital amputation4; however, a study of the treatment of subungual melanoma revealed no difference in survival based on the level of amputation, therefore advocating for less radical treatment.4
Interestingly, MMS for cutaneous tumors localized to the digits is not frequently reviewed in the dermatologic literature. We present a retrospective case series evaluating the clinical outcomes of digital melanoma and NMSCs treated with MMS.
Methods
A retrospective chart review was performed at a private dermatology practice to identify patients who underwent MMS for melanoma or NMSC localized to the digits from January 2009 to December 2014. All patients were treated in the office by 1 Mohs surgeon (A.H.) and were evaluated before and after MMS. Data were collected from the electronic medical record of the practice, including patient demographics, histopathologic diagnosis, tumor status (primary or recurrent lesion), anatomic site of the tumor, preoperative and postoperative size of the lesion, number of MMS stages, surgical repair technique, postoperative complications, and follow-up period.
Results
Twenty-seven patients (13 male, 14 female) with a total of 28 lesions (malignant melanoma or NMSC) localized to the digits were identified (Table). The mean age at the time of MMS was 64.07 years.

Surgical techniques used for repair following MMS included xenograft (10/28 [35.71%]); split-thickness skin graft (7/28 [25.0%]); secondary intention (4/28 [14.29%]); flap (4/28 [14.29%]); full-thickness skin graft (2/28 [7.14%]); and complex closure (1/28 [3.57%]). Clinical preoperative, operative, and postoperative photos from Patient 21 in this series are shown here (Figure). Two patients required bony phalanx resection due to invasion of the tumor into the periosteum: 1 had a malignant melanoma (Breslow depth, 2.52 mm); the other had an SCC. In addition, following removal of a severely dysplastic nevus, debulked tissue revealed melanoma in 1 patient.

Postoperative complications were noted in 4 (14.29%) of 28 MMS procedures, including bacterial wound infection (3.57%), excess granulation tissue that required wound debridement (7.14%), and delay in wound healing (3.57%). Follow-up data were available for 25 of the 28 MMS procedures (mean follow-up, 35.4 months), during which no recurrences were observed.
Comment
Mohs micrographic surgery is a specialized technique used in the treatment of cutaneous tumors, including basal cell carcinoma, SCC, melanoma in situ, atypical fibroxanthoma, dermatofibrosarcoma protuberans, sebaceous carcinoma, microcystic adnexal carcinoma, and Merkel cell carcinoma, among other cutaneous tumors.1-3 Mohs micrographic surgery provides the advantage of tissue conservation as well as optimal margin control in cosmetically or functionally sensitive areas while providing a higher cure rate than surgical excision. During the procedure, the surgical margin is examined histologically, thus ensuring definitive removal of the tumor but minimal loss of surrounding normal tissue.1-3 Mohs micrographic surgery is particularly useful for treating lesions on acral sites (eg, hands, feet, and digits).3-5
The treatment of digital skin cancers has evolved over the past 50 years with advancements resulting in more precise, tissue-sparing methods, in contrast to previous treatments such as amputation and wide local excision.6 More specifically, traditional digital amputation for the treatment of subungual melanoma has been reevaluated in multiple studies, which did not demonstrate a statistically significant difference in survival based on the level of amputation, thereby favoring less radical treatment.4,6 Moehrle et al7 found no statistical difference in recurrence rate when comparing patients with digital melanomas treated with partial amputation and those treated with digit-sparing surgery with limited excision and histologic evaluation of margins. Additionally, in a study conducted by Lazar et al,8 no recurrence of 13 subungual malignancies treated with MMS that utilized a full-thickness graft was reported at 4-year follow-up. In a large retrospective series of digital melanomas treated with MMS, Terushkin et al5 reported that 96.5% (55/57) of patients with primary melanomas that were treated with MMS avoided amputation, and the 5- and 10-year melanoma-specific survival rates for all patients treated with MMS were 95.0% and 82.6%, respectively.
In our study, cutaneous malignancies were located most often on the fingers, and the most common skin cancer identified was SCC in situ. The literature has shown that SCC in situ and SCC are the most common cutaneous neoplasms of the digits and nail unit.9 The most common specific anatomic site of cutaneous malignancy in our study was the great toe, followed by the fourth finger. A study conducted by Tan et al9 revealed that the great toe was the most common location of melanoma of the nail bed and subungual region, followed by the thumb. In contrast, primary subungual SCCs occur most frequently on the finger, with rare cases involving the toes.10
The etiology of digital SCC may involve extensive sun exposure, chronic trauma and wounds, and viral infection.9,11 More specifically, the dermatologic literature provides evidence of human papillomavirus (HPV) type 16 involvement in the pathogenesis of digital and periungual SCC. A genital-digital mechanism of spread has been implicated.11,12 An increased recurrence rate of HPV-associated digital SCCs has been reported following MMS, likely secondary to residual postsurgical HPV infection.11,12
Maintaining function and cosmesis of the hands, feet, and digits following MMS can be challenging, sometimes requiring skin grafts and flaps to close the defect. In the 28 MMS procedures evaluated in our study, 19 (67.9%) surgical defects were repaired with a graft (ie, split-thickness skin graft, full-thickness skin graft, xenograft), 4 (14.3%) with a flap (advancement and rotation), 4 (14.3%) by secondary intention, and 1 (3.6%) with primary complex closure.
Surgical grafts can be categorized based on the origin of the graft.2,13 Autografts, derived from the patient’s skin, are the most frequently used dermatologic graft and can be further categorized as full-thickness skin grafts, which include the epidermis and the entire dermis, thus preserving adnexal structures, and split-thickness skin grafts, which include the epidermis and partial dermis.2,13
A cross-sectional survey of fellowship-trained Mohs surgeons revealed that more than two-thirds of repairs for cutaneous acral cancers were performed using a primary closure technique, and one-fourth of closures were performed using secondary intention.15 Of the less frequently utilized skin-graft repairs, more were for acral lesions on the legs than on the arms.14 The type of procedure and graft used is dependent on multiple variables, including the anatomic location of the lesion and final size of the defect following MMS.2 Similarly, the use of specific types of sutures depends on the anatomic location of the lesion, relative thickness of the skin, degree of tension, and desired cosmetic result.15 The expertise of a hand surgeon may be required, particularly in cases in which the extensor tendon of the distal interphalangeal joint is compromised, manifested by a droopy fingertip when the hand is held horizontally. Additionally, special attention should be paid to removing the entire nail matrix before skin grafting for subungual tumors to avoid nail growth under the skin graft.
Evaluation of debulked tissue from digital skin cancers proved to be important in our study. In Patient 21, debulked tissue revealed melanoma following removal of a severely dysplastic nevus. This finding emphasizes the importance of complete excision of such lesions, as remaining underlying portions of the lesion can reveal residual tumor of the same or different histopathology.
In a prospective study, MMS was shown to have a low rate (0.91%; 95% confidence interval, 0.38%-1.45%) of surgical site infection in the absence of prophylactic antibiotics.16 The highest rates of surgical site infection were closely associated with flap closure. In our study, most patients had an uncomplicated and successful postoperative recovery. Only 1 (3.57%) of the 28 MMS procedures (Patient 22) was complicated by a bacterial wound infection postoperatively. The lesion removed in this case was a severely dysplastic melanocytic nevus on the toe. Infection resolved after a course of oral antibiotics, but the underlying cause of the wound infection in the patient was unclear. Other postoperative complications in our study included delayed wound healing and excess granulation tissue requiring wound debridement.
There are limited data in the dermatologic literature regarding outcomes following MMS for the treatment of cutaneous malignancies localized to the digits.
Additional limitations of this case review include its single-center and retrospective design, the small sample size, and 1 Mohs surgeon having performed all surgeries.
Conclusion
This study provides further evidence of the benefit of MMS for the treatment of malignant melanoma and NMSCs of the digits. This procedure provides margin-controlled excision of these malignant neoplasms while preserving maximal normal tissue, thereby providing patients with improved postoperative function and cosmesis. Long-term follow-up data demonstrating a lack of tumor recurrence underscores the assertion that MMS is safe and effective for the treatment of skin cancer of the digits.
- Dim-Jamora KC, Perone JB. Management of cutaneous tumors with mohs micrographic surgery. Semin Plast Surg. 2008;22:247-256.
- McLeod MP, Choudhary S, Alqubaisy YA, et al. Indications for Mohs micrographic surgery. In: Nouri K, ed. Mohs Micrographic Surgery. New York, NY: Springer; 2012:5-13.
- Loosemore MP, Morales-Burgos A, Goldberg LH. Acral lentiginous melanoma of the toe treated using Mohs surgery with sparing of the digit and subsequent reconstruction using split-thickness skin graft. Dermatol Surg. 2013;39:136-138.
- Rayatt SS, Dancey AL, Davison PM. Thumb subungual melanoma: is amputation necessary? J Plast Reconstr Aesthet Surg. 2007;60:635-638.
- Terushkin V, Brodland DG, Sharon DJ, et al. Digit-sparing Mohs surgery for melanoma. Dermatol Surg. 2016;42:83-93.
- Viola KV, Jhaveri MB, Soulos PR, et al. Mohs micrographic surgery and surgical excision for nonmelanoma skin cancer treatment in the Medicare population. Arch Dermatol. 2012;148:473-477.
- Moehrle M, Metzger S, Schippert W. “Functional” surgery in subungual melanoma. Dermatol Surg. 2003;29:366-374.
- Lazar A, Abimelec P, Dumontier C, et al. Full thickness skin graft from nail unit reconstruction. J Hand Surg Br. 2005;30:194-198.
- Tan KB, Moncrieff M, Thompson JF, et al. Subungual melanoma: a study of 124 cases highlighting features of early lesions, potential for histologic reports. Am J Surg Pathol. 2007;31:1902-1912.
- Nasca MR, Innocenzi D, Micali G. Subungual squamous cell carcinoma of the toe: report on three cases. Dermatol Surg. 2004;30:345-348.
- Dika E, Piraccini BM, Balestri RR, et al. Mohs surgery for squamous cell carcinoma of the nail: report of 15 cases. our experience and a long-term follow-up. Br J Dermatol. 2012;167:1310-1314.
- Alam M, Caldwell JB, Eliezri YD. Human papillomavirus-associated digital squamous cell carcinoma: literature review and report of 21 new cases. J Am Acad Dermatol. 2003;48:385-393.
- Filho L, Anselmo J, Dadalti P, et al. Skin grafts in cutaneous oncology. Braz Ann Dermatol. 2006;81:465-472.
- Raimer DW, Group AR, Petitt MS, et al. Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17:1.
- Alam M, Helenowksi IB, Cohen JL, et al. Association between type of reconstruction after Mohs micrographic surgery and surgeon-, patient-, and tumor-specific features: a cross-sectional study. Dermatol Surg. 2013;39:51-55.
- Rogers HD, Desciak EB, Marcus RP, et al. Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol. 2010;63:842-851.
- Dim-Jamora KC, Perone JB. Management of cutaneous tumors with mohs micrographic surgery. Semin Plast Surg. 2008;22:247-256.
- McLeod MP, Choudhary S, Alqubaisy YA, et al. Indications for Mohs micrographic surgery. In: Nouri K, ed. Mohs Micrographic Surgery. New York, NY: Springer; 2012:5-13.
- Loosemore MP, Morales-Burgos A, Goldberg LH. Acral lentiginous melanoma of the toe treated using Mohs surgery with sparing of the digit and subsequent reconstruction using split-thickness skin graft. Dermatol Surg. 2013;39:136-138.
- Rayatt SS, Dancey AL, Davison PM. Thumb subungual melanoma: is amputation necessary? J Plast Reconstr Aesthet Surg. 2007;60:635-638.
- Terushkin V, Brodland DG, Sharon DJ, et al. Digit-sparing Mohs surgery for melanoma. Dermatol Surg. 2016;42:83-93.
- Viola KV, Jhaveri MB, Soulos PR, et al. Mohs micrographic surgery and surgical excision for nonmelanoma skin cancer treatment in the Medicare population. Arch Dermatol. 2012;148:473-477.
- Moehrle M, Metzger S, Schippert W. “Functional” surgery in subungual melanoma. Dermatol Surg. 2003;29:366-374.
- Lazar A, Abimelec P, Dumontier C, et al. Full thickness skin graft from nail unit reconstruction. J Hand Surg Br. 2005;30:194-198.
- Tan KB, Moncrieff M, Thompson JF, et al. Subungual melanoma: a study of 124 cases highlighting features of early lesions, potential for histologic reports. Am J Surg Pathol. 2007;31:1902-1912.
- Nasca MR, Innocenzi D, Micali G. Subungual squamous cell carcinoma of the toe: report on three cases. Dermatol Surg. 2004;30:345-348.
- Dika E, Piraccini BM, Balestri RR, et al. Mohs surgery for squamous cell carcinoma of the nail: report of 15 cases. our experience and a long-term follow-up. Br J Dermatol. 2012;167:1310-1314.
- Alam M, Caldwell JB, Eliezri YD. Human papillomavirus-associated digital squamous cell carcinoma: literature review and report of 21 new cases. J Am Acad Dermatol. 2003;48:385-393.
- Filho L, Anselmo J, Dadalti P, et al. Skin grafts in cutaneous oncology. Braz Ann Dermatol. 2006;81:465-472.
- Raimer DW, Group AR, Petitt MS, et al. Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17:1.
- Alam M, Helenowksi IB, Cohen JL, et al. Association between type of reconstruction after Mohs micrographic surgery and surgeon-, patient-, and tumor-specific features: a cross-sectional study. Dermatol Surg. 2013;39:51-55.
- Rogers HD, Desciak EB, Marcus RP, et al. Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol. 2010;63:842-851.
Practice Points
- Melanoma and nonmelanoma skin cancers of the digits traditionally have been treated with wide local surgical excision and even amputation.
- Conservative tissue sparing techniques such as Mohs micrographic surgery can be used to treat digital skin cancers with high cure rates and improved functional and cosmetic results.
Novel, noninvasive skin cancer detection device shows promise
DALLAS – An investigational device that couples laser spectroscopy with a machine-learning algorithm demonstrated a high sensitivity and specificity for discriminating skin cancers from benign lesions in real time, results from a single-center study showed.
“More than 5.4 million cases of nonmelanoma skin cancer were treated in 2012, but the accuracy of skin cancer screening prior to biopsy is pretty low, about 70%, and is individual dependent,” lead study author Sung Hyun Pyun, PhD, said at the annual conference of the American Society for Laser Medicine and Surgery. “There have been several in vivo skin cancer screening devices based on noninvasive techniques such as multispectral imaging, Raman spectroscopy, and electrical impedance spectroscopy, but their diagnostic accuracies were not sufficient for clinical use and could not be applied in real time.”
For the single-site study, carried out in Australia, the researchers collected 502 emission spectra from skin cancers confirmed with biopsy results. They also collected 1,429 emission spectra from benign lesions. They achieved a sensitivity of 92% and a specificity of 90% out of 1,931 spectral data sets. No adverse events occurred and no microscopic damage of the irradiated skin was observed.
“Pathologic diagnosis-based cancer detection is considered to be time- and labor-consuming, and can sometimes be individual dependent,” Dr. Pyun said. “Our real-time, noninvasive, in vivo skin cancer detection device demonstrated a high sensitivity and specificity for discriminating skin cancers from benign lesions.” He added that the device could be helpful in office-based cancer screening and real-time, on-site cancer detection during skin cancer surgeries.
Larger, multicenter studies of the device are being planned. Dr. Pyun holds ownership interests with Speclipse, and is an employee of the company.
DALLAS – An investigational device that couples laser spectroscopy with a machine-learning algorithm demonstrated a high sensitivity and specificity for discriminating skin cancers from benign lesions in real time, results from a single-center study showed.
“More than 5.4 million cases of nonmelanoma skin cancer were treated in 2012, but the accuracy of skin cancer screening prior to biopsy is pretty low, about 70%, and is individual dependent,” lead study author Sung Hyun Pyun, PhD, said at the annual conference of the American Society for Laser Medicine and Surgery. “There have been several in vivo skin cancer screening devices based on noninvasive techniques such as multispectral imaging, Raman spectroscopy, and electrical impedance spectroscopy, but their diagnostic accuracies were not sufficient for clinical use and could not be applied in real time.”
For the single-site study, carried out in Australia, the researchers collected 502 emission spectra from skin cancers confirmed with biopsy results. They also collected 1,429 emission spectra from benign lesions. They achieved a sensitivity of 92% and a specificity of 90% out of 1,931 spectral data sets. No adverse events occurred and no microscopic damage of the irradiated skin was observed.
“Pathologic diagnosis-based cancer detection is considered to be time- and labor-consuming, and can sometimes be individual dependent,” Dr. Pyun said. “Our real-time, noninvasive, in vivo skin cancer detection device demonstrated a high sensitivity and specificity for discriminating skin cancers from benign lesions.” He added that the device could be helpful in office-based cancer screening and real-time, on-site cancer detection during skin cancer surgeries.
Larger, multicenter studies of the device are being planned. Dr. Pyun holds ownership interests with Speclipse, and is an employee of the company.
DALLAS – An investigational device that couples laser spectroscopy with a machine-learning algorithm demonstrated a high sensitivity and specificity for discriminating skin cancers from benign lesions in real time, results from a single-center study showed.
“More than 5.4 million cases of nonmelanoma skin cancer were treated in 2012, but the accuracy of skin cancer screening prior to biopsy is pretty low, about 70%, and is individual dependent,” lead study author Sung Hyun Pyun, PhD, said at the annual conference of the American Society for Laser Medicine and Surgery. “There have been several in vivo skin cancer screening devices based on noninvasive techniques such as multispectral imaging, Raman spectroscopy, and electrical impedance spectroscopy, but their diagnostic accuracies were not sufficient for clinical use and could not be applied in real time.”
For the single-site study, carried out in Australia, the researchers collected 502 emission spectra from skin cancers confirmed with biopsy results. They also collected 1,429 emission spectra from benign lesions. They achieved a sensitivity of 92% and a specificity of 90% out of 1,931 spectral data sets. No adverse events occurred and no microscopic damage of the irradiated skin was observed.
“Pathologic diagnosis-based cancer detection is considered to be time- and labor-consuming, and can sometimes be individual dependent,” Dr. Pyun said. “Our real-time, noninvasive, in vivo skin cancer detection device demonstrated a high sensitivity and specificity for discriminating skin cancers from benign lesions.” He added that the device could be helpful in office-based cancer screening and real-time, on-site cancer detection during skin cancer surgeries.
Larger, multicenter studies of the device are being planned. Dr. Pyun holds ownership interests with Speclipse, and is an employee of the company.
REPORTING FROM ASLMS 2018
Key clinical point: A novel device that uses spectroscopy and machine-learning algorithms was found to be a promising tool for the detection of skin cancer.
Major finding: Out of 1,931 spectral data sets, the device achieved a sensitivity of 92% and a specificity of 90%.
Study details: A single-center analysis of 502 emission spectra from skin cancers confirmed with biopsy results.
Disclosures: Dr. Pyun holds ownership interests with Speclipse and is an employee of the company.
New Guidelines of Care for the Management of Nonmelanoma Skin Cancer
In January 2018, the American Academy of Dermatology (AAD) released its first guidelines of care for the management of nonmelanoma skin cancer (NMSC), which established official recommendations for the treatment of basal cell carcinoma (BCC)1 and cutaneous squamous cell carcinoma (cSCC).2 The guidelines will help dermatologists address the growing health concern of skin cancer, which remains the most common of any type of cancer in the United States.3 Affecting more than 3 million Americans every year, NMSC is the most common type of skin cancer, and its incidence has continued to increase every year over the past few decades.3,4 During the past 30 years, the incidence of both BCC and cSCC has more than doubled.5
Commonly used guidelines for the management of NMSC are available from the National Comprehensive Cancer Network (NCCN).6,7 Although the NCCN aimed to develop multidisciplinary guidelines, the new AAD guidelines were established primarily by dermatologists for dermatologists. The NCCN guidelines frequently are referenced throughout the new AAD guidelines, which also recognize the importance of multidisciplinary care. The authors of the AAD guidelines noted that, although many of the NCCN recommendations reiterated prevailing knowledge or current practice, some recommendations highlighted alternative tenets that were not as widely considered or were supported by insufficient evidence.
The AAD guidelines address the complete management of NMSC, which includes biopsy technique, staging, treatment, follow-up, metastatic disease, and prevention.1,2 Also included are evidence tables evaluating the current literature and available recommendations.
BCC Guidelines
For suspected BCCs, the recommended biopsy techniques are punch biopsy, shave biopsy, and excisional biopsy, all of which can detect the most aggressive histology subtypes.1 Rebiopsy is recommended if the initial specimen is inadequate. The pathology report should include histologic subtype, invasion beyond the reticular dermis, and perineural involvement. The AAD guidelines do not include a formal staging system for risk stratification but rather refer to the NCCN guidelines, which take both clinical and pathologic parameters into account. The AAD treatment recommendations are based on this stratification.1
Treatment of BCC includes a broad range of therapeutic modalities. Recurrence rate, preservation of function, patient expectations, and potential adverse effects should be considered in the treatment plan.1 Curettage and electrodessication may be considered for low-risk tumors in nonterminal hair-bearing locations. Surgical excision with 4-mm margins is recommended for low-risk primary tumors. For high-risk BCC, Mohs micrographic surgery is recommended, although standard excision along with attention to margin control may also be considered. Nonsurgical treatments also may be considered when more effective surgical therapies are contraindicated or impractical. If surgical therapy is not feasible or preferred, other treatment options for low-risk BCCs include cryotherapy, topical
Multidisciplinary consultation is recommended in patients with metastatic BCCs along with first-line treatment with a smoothened inhibitor.1 Alternative treatment options include platinum-based chemotherapy and/or supportive care. For locally advanced disease, surgery and radiation therapy remain the initial treatments, but smoothened inhibitors and supportive care are suitable alternative treatments.1
The AAD guidelines also offer recommendations for follow-up and reducing future risk of skin cancer. After the first diagnosis of BCC, a skin cancer screening should be performed at least annually, and patients should be counseled about self-examinations and sun protection.1 Topical and oral retinoids are not recommended for the prevention of additional skin cancers, nor is dietary supplementation with selenium or beta-carotene. There also is insufficient evidence regarding the use of oral nicotinamide, celecoxib, or α-difluoromethylornithine for chemoprevention of disease.1
cSCC Guidelines
For suspected cSCCs, no single optimal biopsy technique is recommended, but repeat biopsy may be considered if the initial biopsy is insufficient for diagnosis.2 The guidelines further recommend an extensive list of elements to be included in the final pathology report (eg, lesion size, immunosuppression, depth of invasion, degree of differentiation). There is no universally recognized stratification for localized cSCC; therefore, the AAD guidelines refer to the framework provided by the NCCN. Also mentioned is the recent release of the American Joint Committee on Cancer’s staging manual,8 which includes the management of cSCC in conjunction with all SCCs of the head and neck. The Brigham and Women’s system9 was considered as an alternative classification system; however, the NCCN guidelines were chosen because they primarily provide clinical guidance for treatment of cSCC rather than provide accurate prognostication or outcome assessment.
Considerations for surgical treatment of cSCC are similar to those for BCC.2 In low-risk tumors, surgical excision with 4- to 6-mm margins to the midsubcutaneous fat or curettage with electrodessication may be considered. Mohs micrographic surgery or standard excision with attention to margin control may be considered for high-risk tumors. Nonsurgical therapies generally are not recommended as a first-line treatment, particularly in cSCC, due to possible recurrence and metastasis. When nonsurgical therapies are preferred, options may include cryosurgery or radiation therapy, with the understanding that cure rates may be lower than with surgical options. Topical therapy with imiquimod or 5-fluorouracil as well as photodynamic or laser therapy are not recommended for cSCCs.2
For patients with metastatic cSCC or locally advanced disease, multidisciplinary consultation is recommended.2 In cSCCs with regional lymph node metastases, the recommended approach includes surgical resection with possible adjuvant radiation therapy and/or systemic therapy. For inoperable disease, combination chemoradiation may be considered. Epidermal growth factor inhibitors and cisplatin may be considered in metastatic disease, although there are limited data to support their efficacy. As with BCC, all patients with cSCCs should receive supportive and palliative care to optimize quality of life.2
Recommendations for follow-up after the first diagnosis of cSCC are the same as those for BCC.2 Additionally, acitretin is the only therapy that may be beneficial in the reduction of recurrent skin cancer in patients who are solid-organ transplant recipients.
Final Thoughts
A comprehensive understanding of the management of NMSC and the evidence on which recommendations are based is critically important for optimal patient care. These guidelines are an efficient way for dermatologists and their colleagues to understand the latest evidence and recommendations. The AAD guidelines provide support for clinical decision making with standardized approaches to the diagnosis, care, and prevention of NMSC that are consistent with established practice patterns.
With few exceptions, surgical therapy is the most effective approach for the treatment of BCC and cSCC; however, the AAD guidelines include an important review on nonsurgical management options.1,2 The AAD guidelines help to highlight where data on evidence-based outcomes exist and reveal where data remain insufficient. This is illustrated by the guideline recommendations for providing additional histopathologic characteristics in the pathology reports, which will likely produce future data to enhance the prognosis and eventual treatment of patients with NMSC.1,2 Future guidelines also may include newer technologies (eg, gene expression profiling).
The guidelines do not cover the management of premalignant and in situ lesions, nor do they provide details on the management of metastatic or locally advanced disease. These topics certainly will require a similar critical review and may be addressed separately. The guidelines are identifying unanswered questions about patient care and are concurrently establishing the collection of appropriate data to answer these questions in the future.
Official guidelines often become the primary source for the measured standard of both treatment and outcomes in patient care; therefore, it is critical that dermatologists and the AAD take the lead in creating these guidelines so that we can provide our patients with the best evidenced-based comprehensive care.
The AAD guidelines emphasize the importance of considering the patient perspective in determining how to treat BCCs and cSCCs.1,2 It is important for patients to understand the available treatment options and participate in their own medical care. The AAD work group for these guidelines included patient advocates to ensure that the guidelines would promote further dialogue between physicians and their patients.
The AAD guidelines for the management of NMSC were developed by board-certified dermatologists and other experts in the field. They allow dermatologists to work with patients diagnosed with NMSC to determine the treatment option that is best for each individual patient.
- Bichakjian C, Armstrong A, Baum C, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
- Burden of skin disease. American Academy of Dermatology website. https://www.aad.org/about/burden-of-skin-disease. Accessed April 17, 2018.
- Rogers HW, Weinstock MA, Feldman SR, et al. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population. JAMA Dermatol. 2015;151:1081-1086.
- Muzic JG, Schmitt AR, Wright AC, et al. Incidence and trends of basal cell carcinoma and cutaneous squamous cell carcinoma: a population-based study in Olmstead County, Minnnesota, 2000-2010. Mayo Clin Proc. 2017;92:890-898.
- Bichakjian CK, Olencki T, Aasi SZ, et al. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Basal Cell Skin Cancer. National Comprehensive Cancer Network website. https://www.nccn.org/professionals/physician_gls/pdf/nmsc.pdf. Published September 18, 2017. Accessed April 17, 2018.
- Bichakjian CK, Olencki T, Aasi SZ, et al. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Squamous Cell Skin Cancer. National Comprehensive Cancer Network website. Published October 5, 2017. Accessed April 17, 2018.
- Amin MB, Edge SB, Greene FL, et al. AJCC Cancer Staging Manual. 8th ed. New York, NY: Springer International Publishing; 2016.
- Jambusaria-Pahlajani A, Kanetsky PA, Karia PS, et al. Evaluation of AJCC tumor staging for cutaneous squamous cell carcinoma and a proposed alternative tumor staging system. JAMA Dermatol. 2013;149:402-410.
In January 2018, the American Academy of Dermatology (AAD) released its first guidelines of care for the management of nonmelanoma skin cancer (NMSC), which established official recommendations for the treatment of basal cell carcinoma (BCC)1 and cutaneous squamous cell carcinoma (cSCC).2 The guidelines will help dermatologists address the growing health concern of skin cancer, which remains the most common of any type of cancer in the United States.3 Affecting more than 3 million Americans every year, NMSC is the most common type of skin cancer, and its incidence has continued to increase every year over the past few decades.3,4 During the past 30 years, the incidence of both BCC and cSCC has more than doubled.5
Commonly used guidelines for the management of NMSC are available from the National Comprehensive Cancer Network (NCCN).6,7 Although the NCCN aimed to develop multidisciplinary guidelines, the new AAD guidelines were established primarily by dermatologists for dermatologists. The NCCN guidelines frequently are referenced throughout the new AAD guidelines, which also recognize the importance of multidisciplinary care. The authors of the AAD guidelines noted that, although many of the NCCN recommendations reiterated prevailing knowledge or current practice, some recommendations highlighted alternative tenets that were not as widely considered or were supported by insufficient evidence.
The AAD guidelines address the complete management of NMSC, which includes biopsy technique, staging, treatment, follow-up, metastatic disease, and prevention.1,2 Also included are evidence tables evaluating the current literature and available recommendations.
BCC Guidelines
For suspected BCCs, the recommended biopsy techniques are punch biopsy, shave biopsy, and excisional biopsy, all of which can detect the most aggressive histology subtypes.1 Rebiopsy is recommended if the initial specimen is inadequate. The pathology report should include histologic subtype, invasion beyond the reticular dermis, and perineural involvement. The AAD guidelines do not include a formal staging system for risk stratification but rather refer to the NCCN guidelines, which take both clinical and pathologic parameters into account. The AAD treatment recommendations are based on this stratification.1
Treatment of BCC includes a broad range of therapeutic modalities. Recurrence rate, preservation of function, patient expectations, and potential adverse effects should be considered in the treatment plan.1 Curettage and electrodessication may be considered for low-risk tumors in nonterminal hair-bearing locations. Surgical excision with 4-mm margins is recommended for low-risk primary tumors. For high-risk BCC, Mohs micrographic surgery is recommended, although standard excision along with attention to margin control may also be considered. Nonsurgical treatments also may be considered when more effective surgical therapies are contraindicated or impractical. If surgical therapy is not feasible or preferred, other treatment options for low-risk BCCs include cryotherapy, topical
Multidisciplinary consultation is recommended in patients with metastatic BCCs along with first-line treatment with a smoothened inhibitor.1 Alternative treatment options include platinum-based chemotherapy and/or supportive care. For locally advanced disease, surgery and radiation therapy remain the initial treatments, but smoothened inhibitors and supportive care are suitable alternative treatments.1
The AAD guidelines also offer recommendations for follow-up and reducing future risk of skin cancer. After the first diagnosis of BCC, a skin cancer screening should be performed at least annually, and patients should be counseled about self-examinations and sun protection.1 Topical and oral retinoids are not recommended for the prevention of additional skin cancers, nor is dietary supplementation with selenium or beta-carotene. There also is insufficient evidence regarding the use of oral nicotinamide, celecoxib, or α-difluoromethylornithine for chemoprevention of disease.1
cSCC Guidelines
For suspected cSCCs, no single optimal biopsy technique is recommended, but repeat biopsy may be considered if the initial biopsy is insufficient for diagnosis.2 The guidelines further recommend an extensive list of elements to be included in the final pathology report (eg, lesion size, immunosuppression, depth of invasion, degree of differentiation). There is no universally recognized stratification for localized cSCC; therefore, the AAD guidelines refer to the framework provided by the NCCN. Also mentioned is the recent release of the American Joint Committee on Cancer’s staging manual,8 which includes the management of cSCC in conjunction with all SCCs of the head and neck. The Brigham and Women’s system9 was considered as an alternative classification system; however, the NCCN guidelines were chosen because they primarily provide clinical guidance for treatment of cSCC rather than provide accurate prognostication or outcome assessment.
Considerations for surgical treatment of cSCC are similar to those for BCC.2 In low-risk tumors, surgical excision with 4- to 6-mm margins to the midsubcutaneous fat or curettage with electrodessication may be considered. Mohs micrographic surgery or standard excision with attention to margin control may be considered for high-risk tumors. Nonsurgical therapies generally are not recommended as a first-line treatment, particularly in cSCC, due to possible recurrence and metastasis. When nonsurgical therapies are preferred, options may include cryosurgery or radiation therapy, with the understanding that cure rates may be lower than with surgical options. Topical therapy with imiquimod or 5-fluorouracil as well as photodynamic or laser therapy are not recommended for cSCCs.2
For patients with metastatic cSCC or locally advanced disease, multidisciplinary consultation is recommended.2 In cSCCs with regional lymph node metastases, the recommended approach includes surgical resection with possible adjuvant radiation therapy and/or systemic therapy. For inoperable disease, combination chemoradiation may be considered. Epidermal growth factor inhibitors and cisplatin may be considered in metastatic disease, although there are limited data to support their efficacy. As with BCC, all patients with cSCCs should receive supportive and palliative care to optimize quality of life.2
Recommendations for follow-up after the first diagnosis of cSCC are the same as those for BCC.2 Additionally, acitretin is the only therapy that may be beneficial in the reduction of recurrent skin cancer in patients who are solid-organ transplant recipients.
Final Thoughts
A comprehensive understanding of the management of NMSC and the evidence on which recommendations are based is critically important for optimal patient care. These guidelines are an efficient way for dermatologists and their colleagues to understand the latest evidence and recommendations. The AAD guidelines provide support for clinical decision making with standardized approaches to the diagnosis, care, and prevention of NMSC that are consistent with established practice patterns.
With few exceptions, surgical therapy is the most effective approach for the treatment of BCC and cSCC; however, the AAD guidelines include an important review on nonsurgical management options.1,2 The AAD guidelines help to highlight where data on evidence-based outcomes exist and reveal where data remain insufficient. This is illustrated by the guideline recommendations for providing additional histopathologic characteristics in the pathology reports, which will likely produce future data to enhance the prognosis and eventual treatment of patients with NMSC.1,2 Future guidelines also may include newer technologies (eg, gene expression profiling).
The guidelines do not cover the management of premalignant and in situ lesions, nor do they provide details on the management of metastatic or locally advanced disease. These topics certainly will require a similar critical review and may be addressed separately. The guidelines are identifying unanswered questions about patient care and are concurrently establishing the collection of appropriate data to answer these questions in the future.
Official guidelines often become the primary source for the measured standard of both treatment and outcomes in patient care; therefore, it is critical that dermatologists and the AAD take the lead in creating these guidelines so that we can provide our patients with the best evidenced-based comprehensive care.
The AAD guidelines emphasize the importance of considering the patient perspective in determining how to treat BCCs and cSCCs.1,2 It is important for patients to understand the available treatment options and participate in their own medical care. The AAD work group for these guidelines included patient advocates to ensure that the guidelines would promote further dialogue between physicians and their patients.
The AAD guidelines for the management of NMSC were developed by board-certified dermatologists and other experts in the field. They allow dermatologists to work with patients diagnosed with NMSC to determine the treatment option that is best for each individual patient.
In January 2018, the American Academy of Dermatology (AAD) released its first guidelines of care for the management of nonmelanoma skin cancer (NMSC), which established official recommendations for the treatment of basal cell carcinoma (BCC)1 and cutaneous squamous cell carcinoma (cSCC).2 The guidelines will help dermatologists address the growing health concern of skin cancer, which remains the most common of any type of cancer in the United States.3 Affecting more than 3 million Americans every year, NMSC is the most common type of skin cancer, and its incidence has continued to increase every year over the past few decades.3,4 During the past 30 years, the incidence of both BCC and cSCC has more than doubled.5
Commonly used guidelines for the management of NMSC are available from the National Comprehensive Cancer Network (NCCN).6,7 Although the NCCN aimed to develop multidisciplinary guidelines, the new AAD guidelines were established primarily by dermatologists for dermatologists. The NCCN guidelines frequently are referenced throughout the new AAD guidelines, which also recognize the importance of multidisciplinary care. The authors of the AAD guidelines noted that, although many of the NCCN recommendations reiterated prevailing knowledge or current practice, some recommendations highlighted alternative tenets that were not as widely considered or were supported by insufficient evidence.
The AAD guidelines address the complete management of NMSC, which includes biopsy technique, staging, treatment, follow-up, metastatic disease, and prevention.1,2 Also included are evidence tables evaluating the current literature and available recommendations.
BCC Guidelines
For suspected BCCs, the recommended biopsy techniques are punch biopsy, shave biopsy, and excisional biopsy, all of which can detect the most aggressive histology subtypes.1 Rebiopsy is recommended if the initial specimen is inadequate. The pathology report should include histologic subtype, invasion beyond the reticular dermis, and perineural involvement. The AAD guidelines do not include a formal staging system for risk stratification but rather refer to the NCCN guidelines, which take both clinical and pathologic parameters into account. The AAD treatment recommendations are based on this stratification.1
Treatment of BCC includes a broad range of therapeutic modalities. Recurrence rate, preservation of function, patient expectations, and potential adverse effects should be considered in the treatment plan.1 Curettage and electrodessication may be considered for low-risk tumors in nonterminal hair-bearing locations. Surgical excision with 4-mm margins is recommended for low-risk primary tumors. For high-risk BCC, Mohs micrographic surgery is recommended, although standard excision along with attention to margin control may also be considered. Nonsurgical treatments also may be considered when more effective surgical therapies are contraindicated or impractical. If surgical therapy is not feasible or preferred, other treatment options for low-risk BCCs include cryotherapy, topical
Multidisciplinary consultation is recommended in patients with metastatic BCCs along with first-line treatment with a smoothened inhibitor.1 Alternative treatment options include platinum-based chemotherapy and/or supportive care. For locally advanced disease, surgery and radiation therapy remain the initial treatments, but smoothened inhibitors and supportive care are suitable alternative treatments.1
The AAD guidelines also offer recommendations for follow-up and reducing future risk of skin cancer. After the first diagnosis of BCC, a skin cancer screening should be performed at least annually, and patients should be counseled about self-examinations and sun protection.1 Topical and oral retinoids are not recommended for the prevention of additional skin cancers, nor is dietary supplementation with selenium or beta-carotene. There also is insufficient evidence regarding the use of oral nicotinamide, celecoxib, or α-difluoromethylornithine for chemoprevention of disease.1
cSCC Guidelines
For suspected cSCCs, no single optimal biopsy technique is recommended, but repeat biopsy may be considered if the initial biopsy is insufficient for diagnosis.2 The guidelines further recommend an extensive list of elements to be included in the final pathology report (eg, lesion size, immunosuppression, depth of invasion, degree of differentiation). There is no universally recognized stratification for localized cSCC; therefore, the AAD guidelines refer to the framework provided by the NCCN. Also mentioned is the recent release of the American Joint Committee on Cancer’s staging manual,8 which includes the management of cSCC in conjunction with all SCCs of the head and neck. The Brigham and Women’s system9 was considered as an alternative classification system; however, the NCCN guidelines were chosen because they primarily provide clinical guidance for treatment of cSCC rather than provide accurate prognostication or outcome assessment.
Considerations for surgical treatment of cSCC are similar to those for BCC.2 In low-risk tumors, surgical excision with 4- to 6-mm margins to the midsubcutaneous fat or curettage with electrodessication may be considered. Mohs micrographic surgery or standard excision with attention to margin control may be considered for high-risk tumors. Nonsurgical therapies generally are not recommended as a first-line treatment, particularly in cSCC, due to possible recurrence and metastasis. When nonsurgical therapies are preferred, options may include cryosurgery or radiation therapy, with the understanding that cure rates may be lower than with surgical options. Topical therapy with imiquimod or 5-fluorouracil as well as photodynamic or laser therapy are not recommended for cSCCs.2
For patients with metastatic cSCC or locally advanced disease, multidisciplinary consultation is recommended.2 In cSCCs with regional lymph node metastases, the recommended approach includes surgical resection with possible adjuvant radiation therapy and/or systemic therapy. For inoperable disease, combination chemoradiation may be considered. Epidermal growth factor inhibitors and cisplatin may be considered in metastatic disease, although there are limited data to support their efficacy. As with BCC, all patients with cSCCs should receive supportive and palliative care to optimize quality of life.2
Recommendations for follow-up after the first diagnosis of cSCC are the same as those for BCC.2 Additionally, acitretin is the only therapy that may be beneficial in the reduction of recurrent skin cancer in patients who are solid-organ transplant recipients.
Final Thoughts
A comprehensive understanding of the management of NMSC and the evidence on which recommendations are based is critically important for optimal patient care. These guidelines are an efficient way for dermatologists and their colleagues to understand the latest evidence and recommendations. The AAD guidelines provide support for clinical decision making with standardized approaches to the diagnosis, care, and prevention of NMSC that are consistent with established practice patterns.
With few exceptions, surgical therapy is the most effective approach for the treatment of BCC and cSCC; however, the AAD guidelines include an important review on nonsurgical management options.1,2 The AAD guidelines help to highlight where data on evidence-based outcomes exist and reveal where data remain insufficient. This is illustrated by the guideline recommendations for providing additional histopathologic characteristics in the pathology reports, which will likely produce future data to enhance the prognosis and eventual treatment of patients with NMSC.1,2 Future guidelines also may include newer technologies (eg, gene expression profiling).
The guidelines do not cover the management of premalignant and in situ lesions, nor do they provide details on the management of metastatic or locally advanced disease. These topics certainly will require a similar critical review and may be addressed separately. The guidelines are identifying unanswered questions about patient care and are concurrently establishing the collection of appropriate data to answer these questions in the future.
Official guidelines often become the primary source for the measured standard of both treatment and outcomes in patient care; therefore, it is critical that dermatologists and the AAD take the lead in creating these guidelines so that we can provide our patients with the best evidenced-based comprehensive care.
The AAD guidelines emphasize the importance of considering the patient perspective in determining how to treat BCCs and cSCCs.1,2 It is important for patients to understand the available treatment options and participate in their own medical care. The AAD work group for these guidelines included patient advocates to ensure that the guidelines would promote further dialogue between physicians and their patients.
The AAD guidelines for the management of NMSC were developed by board-certified dermatologists and other experts in the field. They allow dermatologists to work with patients diagnosed with NMSC to determine the treatment option that is best for each individual patient.
- Bichakjian C, Armstrong A, Baum C, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
- Burden of skin disease. American Academy of Dermatology website. https://www.aad.org/about/burden-of-skin-disease. Accessed April 17, 2018.
- Rogers HW, Weinstock MA, Feldman SR, et al. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population. JAMA Dermatol. 2015;151:1081-1086.
- Muzic JG, Schmitt AR, Wright AC, et al. Incidence and trends of basal cell carcinoma and cutaneous squamous cell carcinoma: a population-based study in Olmstead County, Minnnesota, 2000-2010. Mayo Clin Proc. 2017;92:890-898.
- Bichakjian CK, Olencki T, Aasi SZ, et al. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Basal Cell Skin Cancer. National Comprehensive Cancer Network website. https://www.nccn.org/professionals/physician_gls/pdf/nmsc.pdf. Published September 18, 2017. Accessed April 17, 2018.
- Bichakjian CK, Olencki T, Aasi SZ, et al. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Squamous Cell Skin Cancer. National Comprehensive Cancer Network website. Published October 5, 2017. Accessed April 17, 2018.
- Amin MB, Edge SB, Greene FL, et al. AJCC Cancer Staging Manual. 8th ed. New York, NY: Springer International Publishing; 2016.
- Jambusaria-Pahlajani A, Kanetsky PA, Karia PS, et al. Evaluation of AJCC tumor staging for cutaneous squamous cell carcinoma and a proposed alternative tumor staging system. JAMA Dermatol. 2013;149:402-410.
- Bichakjian C, Armstrong A, Baum C, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
- Burden of skin disease. American Academy of Dermatology website. https://www.aad.org/about/burden-of-skin-disease. Accessed April 17, 2018.
- Rogers HW, Weinstock MA, Feldman SR, et al. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population. JAMA Dermatol. 2015;151:1081-1086.
- Muzic JG, Schmitt AR, Wright AC, et al. Incidence and trends of basal cell carcinoma and cutaneous squamous cell carcinoma: a population-based study in Olmstead County, Minnnesota, 2000-2010. Mayo Clin Proc. 2017;92:890-898.
- Bichakjian CK, Olencki T, Aasi SZ, et al. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Basal Cell Skin Cancer. National Comprehensive Cancer Network website. https://www.nccn.org/professionals/physician_gls/pdf/nmsc.pdf. Published September 18, 2017. Accessed April 17, 2018.
- Bichakjian CK, Olencki T, Aasi SZ, et al. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Squamous Cell Skin Cancer. National Comprehensive Cancer Network website. Published October 5, 2017. Accessed April 17, 2018.
- Amin MB, Edge SB, Greene FL, et al. AJCC Cancer Staging Manual. 8th ed. New York, NY: Springer International Publishing; 2016.
- Jambusaria-Pahlajani A, Kanetsky PA, Karia PS, et al. Evaluation of AJCC tumor staging for cutaneous squamous cell carcinoma and a proposed alternative tumor staging system. JAMA Dermatol. 2013;149:402-410.
FDA begins priority review of cemiplimab for advanced cutaneous squamous cell carcinoma
The Food and Drug Administration will conduct a priority review of cemiplimab for the treatment of locally advanced and metastatic cutaneous squamous cell carcinoma (SCC), the companies developing the treatment announced on April 30.
Cemiplimab, a human monoclonal antibody being developed by Regeneron Pharmaceuticals and Sanofi, targets the checkpoint inhibitor programmed cell death protein-1 (PD-1). The drug was previously granted Breakthrough Therapy status by the FDA in September 2017.
No safety and efficacy data are available for cemiplimab at this time.
Find the full press release on the Regeneron website.
The Food and Drug Administration will conduct a priority review of cemiplimab for the treatment of locally advanced and metastatic cutaneous squamous cell carcinoma (SCC), the companies developing the treatment announced on April 30.
Cemiplimab, a human monoclonal antibody being developed by Regeneron Pharmaceuticals and Sanofi, targets the checkpoint inhibitor programmed cell death protein-1 (PD-1). The drug was previously granted Breakthrough Therapy status by the FDA in September 2017.
No safety and efficacy data are available for cemiplimab at this time.
Find the full press release on the Regeneron website.
The Food and Drug Administration will conduct a priority review of cemiplimab for the treatment of locally advanced and metastatic cutaneous squamous cell carcinoma (SCC), the companies developing the treatment announced on April 30.
Cemiplimab, a human monoclonal antibody being developed by Regeneron Pharmaceuticals and Sanofi, targets the checkpoint inhibitor programmed cell death protein-1 (PD-1). The drug was previously granted Breakthrough Therapy status by the FDA in September 2017.
No safety and efficacy data are available for cemiplimab at this time.
Find the full press release on the Regeneron website.
HIV infection linked to higher risk of non-melanoma skin cancer
A Danish cohort study provides more evidence of a significant link between HIV infection and two types of skin cancer.
, with incidence rate ratios (IRRs) of 1.79 and 5.40, respectively, when compared with a background population, who were HIV-negative.
“The risk of SCC seemed to increase with increasing level of immunosuppression while the increased risk of BCC was restricted to patients reporting MSM [men who have sex with men] as route of infection,” wrote the authors, led by Silje Haukali Omland, MD, PhD, of the department of dermato-venereology, Copenhagen University Hospital.
The Danish nationwide cohort study, which matched each HIV patient with 5 age- and sex-matched individuals from the background population, was published online March 26 in the Journal of the American Academy of Dermatology.
The results are similar to those published elsewhere, as is the finding that HIV-positive patients do not face a higher risk of malignant melanoma. “The results here confirm prior studies and support heightened vigilance for skin conditions, such as SCC and BCC in HIV patients,” said Michael J. Silverberg, PhD, of Kaiser Permanente Division of Research, in an interview after reviewing the study findings. He was not a study author.
Researchers have long noted a connection between various types of cancer and HIV infection. But, as noted in a 2013 study led by Dr. Silverberg, research into links between HIV and non-melanoma skin cancers has been sparse and inconclusive. That study of white adults found higher adjusted rate ratios for SCC (2.6) and BCC (2.1) among those who were HIV-positive compared with those who were HIV-negative (J Natl Cancer Inst. 2013 Mar 6;105[5]:350-60).
In the Danish study, researchers tracked sex-and age-matched cohorts of HIV-infected (4,280) and non-HIV-infected patients (21,399) aged 16 years or older from study inclusion through as late as 2014. All the HIV-positive subjects had taken antiretroviral medications. The researchers also compared the HIV-positive patients to their non-HIV-infected siblings.
Overall, those who were HIV-positive were more likely to develop BCC (IRR, 1.79, 95% CI, 1.43-2.22), and males who reported sex with men had an even higher risk (IRR, 2.30, 95% CI, 1.76-3.02).
As for SCC, the IRR was 5.40 (95% CI, 3.07-9.52) among those who were HIV-positive, compared with the background population, and the researchers found evidence that risk increased with level of immunosuppression. Those who indicated heterosexual and male homosexual transmission had similar rates of SCC.
The rates of BCC or SCC were not higher among siblings of HIV-positive patients.
In addition, the risk of melanoma was not increased among those who were HIV-positive subjects or their siblings, when compared with the background group. However, the researchers noted that the study turned up a low number of HIV-positive subjects with melanoma, potentially throwing off the results.
The researchers noted that the inclusion of siblings in the study suggests that sun exposure in childhood was not a confounding factor. Presumably, they wrote, the siblings had similar levels of exposure as children, although exposure to sun bed tanning could differ between siblings.
“Study methods appear very strong and consistent with other work done in the area,” Dr. Silverberg said in the interview. As for possible causes of the disparities, he noted that exposure to the sun or to tanning beds could explain the greater risk of BCC among men who have sex with men. “For SCC, there may be a biological link, as studies have suggested a link with human papillomavirus for that particular cancer,” he added.
No study funding was reported. The study authors reported disclosures that included grants, research grants, speaker fees, and/or advisory board honoraria from several drug manufacturers. Dr. Silverberg has no relevant disclosures.
A Danish cohort study provides more evidence of a significant link between HIV infection and two types of skin cancer.
, with incidence rate ratios (IRRs) of 1.79 and 5.40, respectively, when compared with a background population, who were HIV-negative.
“The risk of SCC seemed to increase with increasing level of immunosuppression while the increased risk of BCC was restricted to patients reporting MSM [men who have sex with men] as route of infection,” wrote the authors, led by Silje Haukali Omland, MD, PhD, of the department of dermato-venereology, Copenhagen University Hospital.
The Danish nationwide cohort study, which matched each HIV patient with 5 age- and sex-matched individuals from the background population, was published online March 26 in the Journal of the American Academy of Dermatology.
The results are similar to those published elsewhere, as is the finding that HIV-positive patients do not face a higher risk of malignant melanoma. “The results here confirm prior studies and support heightened vigilance for skin conditions, such as SCC and BCC in HIV patients,” said Michael J. Silverberg, PhD, of Kaiser Permanente Division of Research, in an interview after reviewing the study findings. He was not a study author.
Researchers have long noted a connection between various types of cancer and HIV infection. But, as noted in a 2013 study led by Dr. Silverberg, research into links between HIV and non-melanoma skin cancers has been sparse and inconclusive. That study of white adults found higher adjusted rate ratios for SCC (2.6) and BCC (2.1) among those who were HIV-positive compared with those who were HIV-negative (J Natl Cancer Inst. 2013 Mar 6;105[5]:350-60).
In the Danish study, researchers tracked sex-and age-matched cohorts of HIV-infected (4,280) and non-HIV-infected patients (21,399) aged 16 years or older from study inclusion through as late as 2014. All the HIV-positive subjects had taken antiretroviral medications. The researchers also compared the HIV-positive patients to their non-HIV-infected siblings.
Overall, those who were HIV-positive were more likely to develop BCC (IRR, 1.79, 95% CI, 1.43-2.22), and males who reported sex with men had an even higher risk (IRR, 2.30, 95% CI, 1.76-3.02).
As for SCC, the IRR was 5.40 (95% CI, 3.07-9.52) among those who were HIV-positive, compared with the background population, and the researchers found evidence that risk increased with level of immunosuppression. Those who indicated heterosexual and male homosexual transmission had similar rates of SCC.
The rates of BCC or SCC were not higher among siblings of HIV-positive patients.
In addition, the risk of melanoma was not increased among those who were HIV-positive subjects or their siblings, when compared with the background group. However, the researchers noted that the study turned up a low number of HIV-positive subjects with melanoma, potentially throwing off the results.
The researchers noted that the inclusion of siblings in the study suggests that sun exposure in childhood was not a confounding factor. Presumably, they wrote, the siblings had similar levels of exposure as children, although exposure to sun bed tanning could differ between siblings.
“Study methods appear very strong and consistent with other work done in the area,” Dr. Silverberg said in the interview. As for possible causes of the disparities, he noted that exposure to the sun or to tanning beds could explain the greater risk of BCC among men who have sex with men. “For SCC, there may be a biological link, as studies have suggested a link with human papillomavirus for that particular cancer,” he added.
No study funding was reported. The study authors reported disclosures that included grants, research grants, speaker fees, and/or advisory board honoraria from several drug manufacturers. Dr. Silverberg has no relevant disclosures.
A Danish cohort study provides more evidence of a significant link between HIV infection and two types of skin cancer.
, with incidence rate ratios (IRRs) of 1.79 and 5.40, respectively, when compared with a background population, who were HIV-negative.
“The risk of SCC seemed to increase with increasing level of immunosuppression while the increased risk of BCC was restricted to patients reporting MSM [men who have sex with men] as route of infection,” wrote the authors, led by Silje Haukali Omland, MD, PhD, of the department of dermato-venereology, Copenhagen University Hospital.
The Danish nationwide cohort study, which matched each HIV patient with 5 age- and sex-matched individuals from the background population, was published online March 26 in the Journal of the American Academy of Dermatology.
The results are similar to those published elsewhere, as is the finding that HIV-positive patients do not face a higher risk of malignant melanoma. “The results here confirm prior studies and support heightened vigilance for skin conditions, such as SCC and BCC in HIV patients,” said Michael J. Silverberg, PhD, of Kaiser Permanente Division of Research, in an interview after reviewing the study findings. He was not a study author.
Researchers have long noted a connection between various types of cancer and HIV infection. But, as noted in a 2013 study led by Dr. Silverberg, research into links between HIV and non-melanoma skin cancers has been sparse and inconclusive. That study of white adults found higher adjusted rate ratios for SCC (2.6) and BCC (2.1) among those who were HIV-positive compared with those who were HIV-negative (J Natl Cancer Inst. 2013 Mar 6;105[5]:350-60).
In the Danish study, researchers tracked sex-and age-matched cohorts of HIV-infected (4,280) and non-HIV-infected patients (21,399) aged 16 years or older from study inclusion through as late as 2014. All the HIV-positive subjects had taken antiretroviral medications. The researchers also compared the HIV-positive patients to their non-HIV-infected siblings.
Overall, those who were HIV-positive were more likely to develop BCC (IRR, 1.79, 95% CI, 1.43-2.22), and males who reported sex with men had an even higher risk (IRR, 2.30, 95% CI, 1.76-3.02).
As for SCC, the IRR was 5.40 (95% CI, 3.07-9.52) among those who were HIV-positive, compared with the background population, and the researchers found evidence that risk increased with level of immunosuppression. Those who indicated heterosexual and male homosexual transmission had similar rates of SCC.
The rates of BCC or SCC were not higher among siblings of HIV-positive patients.
In addition, the risk of melanoma was not increased among those who were HIV-positive subjects or their siblings, when compared with the background group. However, the researchers noted that the study turned up a low number of HIV-positive subjects with melanoma, potentially throwing off the results.
The researchers noted that the inclusion of siblings in the study suggests that sun exposure in childhood was not a confounding factor. Presumably, they wrote, the siblings had similar levels of exposure as children, although exposure to sun bed tanning could differ between siblings.
“Study methods appear very strong and consistent with other work done in the area,” Dr. Silverberg said in the interview. As for possible causes of the disparities, he noted that exposure to the sun or to tanning beds could explain the greater risk of BCC among men who have sex with men. “For SCC, there may be a biological link, as studies have suggested a link with human papillomavirus for that particular cancer,” he added.
No study funding was reported. The study authors reported disclosures that included grants, research grants, speaker fees, and/or advisory board honoraria from several drug manufacturers. Dr. Silverberg has no relevant disclosures.
FROM JAAD
Key clinical point: HIV-positive patients are at an increased risk for basal cell carcinoma (BCC) and squamous cell carcinoma (SCC).
Major finding: Among HIV-infected patients, the risk of BCC was increased by almost twofold and the risk of BCC was increased by more than fivefold.
Study details: A Danish population-based cohort study of 4,280 HIV-infected patients and 21,399 age-and sex-matched subjects.
Disclosures: No study funding was reported. The authors reported disclosures that included research grants, speaker fees, and/or advisory board honoraria from several drug manufacturers.
Source: Omland S et al. J Am Acad Dermatol. 2018 Mar 24. pii: S0190-9622(18)30475-4. doi: 10.1016/j.jaad.2018.03.024.
New cases of Merkel cell carcinoma increased 95% between 2000 and 2013
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
The number of new cases per year of Merkel cell carcinoma (MCC) increased by 95% during 2000-2013, according to a review of Surveillance, Epidemiology, and End Results (SEER) data.
There were 652 cases of MCC in the SEER-18 registry in 2013, up from the 334 cases captured by the database in 2000.
This increase exceeded the 56.5% increase seen with melanoma over the same time period, the investigators wrote in the Journal of the American Academy of Dermatology.
The total number of incident MCC cases in the United States in 2013 was calculated as 2,488 cases/year by using SEER-derived incidence rates combined with U.S. Census population data. The MCC incidence rate rose precipitously with age, increasing 10-fold between ages 40-44 years (0.1 cases/100,000 person-years) and ages 60-64 years (0.9 cases/100,000 person-years).
Given the aging of the population and an assumption that the incidence rates within any given age group will remain stable, the annual incidence of Merkel cell carcinoma in the United States will increase to 3,284 cases/year in 2025, Kelly G. Paulson, MD, PhD, of the Fred Hutchinson Cancer Research Center, Seattle, and her colleagues projected.
“The incidence of MCC is increasing and will likely continue to rise as the Baby Boomer population enters the higher-risk age groups for MCC,” Dr. Paulson and colleagues said. ”Because of its high propensity for spread, the need for adjuvant radiation in many cases, and the clear role for early immunotherapy in the metastatic setting, both early detection and optimal management will be critical for improved outcomes,” they concluded.
SOURCE: Paulson KG et al. J Am Acad Derm. 2018 Mar;78(3):457-463.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
The number of new cases per year of Merkel cell carcinoma (MCC) increased by 95% during 2000-2013, according to a review of Surveillance, Epidemiology, and End Results (SEER) data.
There were 652 cases of MCC in the SEER-18 registry in 2013, up from the 334 cases captured by the database in 2000.
This increase exceeded the 56.5% increase seen with melanoma over the same time period, the investigators wrote in the Journal of the American Academy of Dermatology.
The total number of incident MCC cases in the United States in 2013 was calculated as 2,488 cases/year by using SEER-derived incidence rates combined with U.S. Census population data. The MCC incidence rate rose precipitously with age, increasing 10-fold between ages 40-44 years (0.1 cases/100,000 person-years) and ages 60-64 years (0.9 cases/100,000 person-years).
Given the aging of the population and an assumption that the incidence rates within any given age group will remain stable, the annual incidence of Merkel cell carcinoma in the United States will increase to 3,284 cases/year in 2025, Kelly G. Paulson, MD, PhD, of the Fred Hutchinson Cancer Research Center, Seattle, and her colleagues projected.
“The incidence of MCC is increasing and will likely continue to rise as the Baby Boomer population enters the higher-risk age groups for MCC,” Dr. Paulson and colleagues said. ”Because of its high propensity for spread, the need for adjuvant radiation in many cases, and the clear role for early immunotherapy in the metastatic setting, both early detection and optimal management will be critical for improved outcomes,” they concluded.
SOURCE: Paulson KG et al. J Am Acad Derm. 2018 Mar;78(3):457-463.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
The number of new cases per year of Merkel cell carcinoma (MCC) increased by 95% during 2000-2013, according to a review of Surveillance, Epidemiology, and End Results (SEER) data.
There were 652 cases of MCC in the SEER-18 registry in 2013, up from the 334 cases captured by the database in 2000.
This increase exceeded the 56.5% increase seen with melanoma over the same time period, the investigators wrote in the Journal of the American Academy of Dermatology.
The total number of incident MCC cases in the United States in 2013 was calculated as 2,488 cases/year by using SEER-derived incidence rates combined with U.S. Census population data. The MCC incidence rate rose precipitously with age, increasing 10-fold between ages 40-44 years (0.1 cases/100,000 person-years) and ages 60-64 years (0.9 cases/100,000 person-years).
Given the aging of the population and an assumption that the incidence rates within any given age group will remain stable, the annual incidence of Merkel cell carcinoma in the United States will increase to 3,284 cases/year in 2025, Kelly G. Paulson, MD, PhD, of the Fred Hutchinson Cancer Research Center, Seattle, and her colleagues projected.
“The incidence of MCC is increasing and will likely continue to rise as the Baby Boomer population enters the higher-risk age groups for MCC,” Dr. Paulson and colleagues said. ”Because of its high propensity for spread, the need for adjuvant radiation in many cases, and the clear role for early immunotherapy in the metastatic setting, both early detection and optimal management will be critical for improved outcomes,” they concluded.
SOURCE: Paulson KG et al. J Am Acad Derm. 2018 Mar;78(3):457-463.
Key clinical point: The incidence of Merkel cell carcinoma (MCC) is expected to continue at a brisk increase.
Major finding: During 2000-2013, the rate of new U.S. MCC cases increased by 95% to 2,488 diagnoses/year.
Study details: Incidence and future projections were calculated by combining registry data from the SEER-18 Database and U.S. Census data.
Disclosures: The study was funded by grants from the National Institutes of Health, the Prostate Cancer Foundation, the University of Washington MCC Patient Gift Fund, and the Bloom endowment at University of Washington in Seattle. One coauthor disclosed support from EMD Serono, Pfizer, and Bristol-Meyers Squibb. All other authors had no conflicts of interest.
Source: Paulson KG et al. J Am Acad Derm. 2018 Mar;78(3): 457-63.
CO2 laser guided by confocal microscopy effectively treated superficial BCC
DALLAS – The use of CO2 laser ablation guided by reflectance confocal microscopy is an effective, minimally invasive treatment for superficial and early nodular basal cell carcinoma (BCC), according to results from an ongoing study.
“While surgery is the gold standard for many basal cell carcinomas, nonsurgical therapies may be a good option for the superficial and early nodular subtypes,” lead study author Anthony M. Rossi, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Laser ablation was used many years ago, so this is not a novel concept, but we’re bringing it back and we’re trying to use confocal microscopy to hone in on the basal cell and selectively target the tumor.”
For the current analysis, he and his associates used with a mean age of 55 years. Of the 20 lesions, 18 were located on the limbs and trunk, while two were on the head and neck. The median lesion diameter was 7 mm. Prior to laser ablation, the researchers performed reflectance confocal microscopy to define lateral and deep margins and define the laser parameters.
The median number of laser passes was three, and ranged from two to eight, delivered at a fluence of 7.5 J/cm2. Reflectance confocal microscopy was repeated immediately after the laser treatment to the skin wound margins and deep margins, and it was performed every 3-6 months thereafter. “If you do confocal microscopy too early, you’ll see mainly inflammation and you may see residual tumor that hasn’t been fully resolved yet,” Dr. Rossi said.
As for future directions, he and his colleagues are developing contrast agents to enhance the ability to detect BCC tumors in vivo, to highlight tumor islands, and to differentiate sebaceous glands and hair follicles. Dr. Rossi reported having no relevant disclosures.
DALLAS – The use of CO2 laser ablation guided by reflectance confocal microscopy is an effective, minimally invasive treatment for superficial and early nodular basal cell carcinoma (BCC), according to results from an ongoing study.
“While surgery is the gold standard for many basal cell carcinomas, nonsurgical therapies may be a good option for the superficial and early nodular subtypes,” lead study author Anthony M. Rossi, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Laser ablation was used many years ago, so this is not a novel concept, but we’re bringing it back and we’re trying to use confocal microscopy to hone in on the basal cell and selectively target the tumor.”
For the current analysis, he and his associates used with a mean age of 55 years. Of the 20 lesions, 18 were located on the limbs and trunk, while two were on the head and neck. The median lesion diameter was 7 mm. Prior to laser ablation, the researchers performed reflectance confocal microscopy to define lateral and deep margins and define the laser parameters.
The median number of laser passes was three, and ranged from two to eight, delivered at a fluence of 7.5 J/cm2. Reflectance confocal microscopy was repeated immediately after the laser treatment to the skin wound margins and deep margins, and it was performed every 3-6 months thereafter. “If you do confocal microscopy too early, you’ll see mainly inflammation and you may see residual tumor that hasn’t been fully resolved yet,” Dr. Rossi said.
As for future directions, he and his colleagues are developing contrast agents to enhance the ability to detect BCC tumors in vivo, to highlight tumor islands, and to differentiate sebaceous glands and hair follicles. Dr. Rossi reported having no relevant disclosures.
DALLAS – The use of CO2 laser ablation guided by reflectance confocal microscopy is an effective, minimally invasive treatment for superficial and early nodular basal cell carcinoma (BCC), according to results from an ongoing study.
“While surgery is the gold standard for many basal cell carcinomas, nonsurgical therapies may be a good option for the superficial and early nodular subtypes,” lead study author Anthony M. Rossi, MD, said at the annual conference of the American Society for Laser Medicine and Surgery. “Laser ablation was used many years ago, so this is not a novel concept, but we’re bringing it back and we’re trying to use confocal microscopy to hone in on the basal cell and selectively target the tumor.”
For the current analysis, he and his associates used with a mean age of 55 years. Of the 20 lesions, 18 were located on the limbs and trunk, while two were on the head and neck. The median lesion diameter was 7 mm. Prior to laser ablation, the researchers performed reflectance confocal microscopy to define lateral and deep margins and define the laser parameters.
The median number of laser passes was three, and ranged from two to eight, delivered at a fluence of 7.5 J/cm2. Reflectance confocal microscopy was repeated immediately after the laser treatment to the skin wound margins and deep margins, and it was performed every 3-6 months thereafter. “If you do confocal microscopy too early, you’ll see mainly inflammation and you may see residual tumor that hasn’t been fully resolved yet,” Dr. Rossi said.
As for future directions, he and his colleagues are developing contrast agents to enhance the ability to detect BCC tumors in vivo, to highlight tumor islands, and to differentiate sebaceous glands and hair follicles. Dr. Rossi reported having no relevant disclosures.
REPORTING FROM ASLMS 2018
Key clinical point: Reflectance confocal microscopy-guided CO2 laser ablation of basal cell carcinoma (BCC) was found to be effective.
Major finding: After an average follow-up of 17 months, no recurrence of BCC has been detected clinically, dermoscopically, or by reflectance confocal microscopy.
Study details: A clinical analysis of seven adults with superficial BCC who were treated with a CO2 laser guided by confocal microscopy.
Disclosures: Dr. Rossi reported having no financial disclosures.
Tanning is the new tobacco
I was driving to work the other day, perched up in my pickup truck (somehow you knew that) and noticed a fancy race car in front of me with a vanity tag. It read HRTATTK 4. Well, I thought after four heart attacks maybe I would splurge on a special car too (more likely a newer truck). Then I noticed smoke coming out of the driver’s window, and I could see this guy in his side view mirror, presumably Mr. “Heart Attack 4,” puffing away on a cigarette. Wow.
Then I got to work and saw my secretary, who works with her oxygen on, out back puffing a cigarette. Wow.
It turns out that cigarette smoke contains substances that act as a monoamine oxidase (MAO) A inhibitor, prolonging the dopamine high in the brain (Proc Natl Acad Sci U S A. 1996 Nov 26;93[24]:14065-9). Makes sense and may explain the above smoking behavior. I truly believe cigarettes are as or more addictive than any other dopamine enhancing drug.
More than 50 years ago, a national campaign against smoking was launched after the 1964 Surgeon General’s report concluded that smoking was a major health hazard. (Looking back, one of the few losses of not having to pull journal articles from the stacks in the library, is that medical students and residents can’t shake their heads in wonder at the cigarette ads in old medical journals.) The impact of the national antismoking campaign has been dramatic, but smoking remains the leading preventable cause of death in the United States and globally, according to the Centers for Disease Control and Prevention.
in 2006, from 1992 (Arch Dermatol. 2010;146[3]:283-7), dermatologists had good footing on which to start a major prevention campaign. The American Cancer Society got on board, and in 2014, acting surgeon general Boris Lushniak, MD, issued a call to action to prevent skin cancer along with Howard Koh, MD, the assistant secretary of health, in “The Surgeon General’s Call to Action to Prevent Skin Cancer” in 2014, and the campaign was on.
Well, I am delighted to pass on a report from Leonard Lichtenfeld, MD, deputy chief medical officer for the American Cancer Society, who recently described in his March 2018 blog what may the first signs of the effectiveness of efforts to promote protection from ultraviolet ray exposure (JAMA Dermatol. 2018;154[3]:361-2). He writes: “In young white women ages 15 to 24, the incidence of melanoma has declined an average of 5.5% per year from January 2005 through December 2014. Not 5.5% over those ten years but 5.5 % PER YEAR. That’s remarkable, to say the least.”
As for the reasons behind these trends, he says, “no one can say for certain,” but he refers to national data indicating that indoor tanning has decreased in the past few years, especially among adolescents and young adults.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
I was driving to work the other day, perched up in my pickup truck (somehow you knew that) and noticed a fancy race car in front of me with a vanity tag. It read HRTATTK 4. Well, I thought after four heart attacks maybe I would splurge on a special car too (more likely a newer truck). Then I noticed smoke coming out of the driver’s window, and I could see this guy in his side view mirror, presumably Mr. “Heart Attack 4,” puffing away on a cigarette. Wow.
Then I got to work and saw my secretary, who works with her oxygen on, out back puffing a cigarette. Wow.
It turns out that cigarette smoke contains substances that act as a monoamine oxidase (MAO) A inhibitor, prolonging the dopamine high in the brain (Proc Natl Acad Sci U S A. 1996 Nov 26;93[24]:14065-9). Makes sense and may explain the above smoking behavior. I truly believe cigarettes are as or more addictive than any other dopamine enhancing drug.
More than 50 years ago, a national campaign against smoking was launched after the 1964 Surgeon General’s report concluded that smoking was a major health hazard. (Looking back, one of the few losses of not having to pull journal articles from the stacks in the library, is that medical students and residents can’t shake their heads in wonder at the cigarette ads in old medical journals.) The impact of the national antismoking campaign has been dramatic, but smoking remains the leading preventable cause of death in the United States and globally, according to the Centers for Disease Control and Prevention.
in 2006, from 1992 (Arch Dermatol. 2010;146[3]:283-7), dermatologists had good footing on which to start a major prevention campaign. The American Cancer Society got on board, and in 2014, acting surgeon general Boris Lushniak, MD, issued a call to action to prevent skin cancer along with Howard Koh, MD, the assistant secretary of health, in “The Surgeon General’s Call to Action to Prevent Skin Cancer” in 2014, and the campaign was on.
Well, I am delighted to pass on a report from Leonard Lichtenfeld, MD, deputy chief medical officer for the American Cancer Society, who recently described in his March 2018 blog what may the first signs of the effectiveness of efforts to promote protection from ultraviolet ray exposure (JAMA Dermatol. 2018;154[3]:361-2). He writes: “In young white women ages 15 to 24, the incidence of melanoma has declined an average of 5.5% per year from January 2005 through December 2014. Not 5.5% over those ten years but 5.5 % PER YEAR. That’s remarkable, to say the least.”
As for the reasons behind these trends, he says, “no one can say for certain,” but he refers to national data indicating that indoor tanning has decreased in the past few years, especially among adolescents and young adults.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
I was driving to work the other day, perched up in my pickup truck (somehow you knew that) and noticed a fancy race car in front of me with a vanity tag. It read HRTATTK 4. Well, I thought after four heart attacks maybe I would splurge on a special car too (more likely a newer truck). Then I noticed smoke coming out of the driver’s window, and I could see this guy in his side view mirror, presumably Mr. “Heart Attack 4,” puffing away on a cigarette. Wow.
Then I got to work and saw my secretary, who works with her oxygen on, out back puffing a cigarette. Wow.
It turns out that cigarette smoke contains substances that act as a monoamine oxidase (MAO) A inhibitor, prolonging the dopamine high in the brain (Proc Natl Acad Sci U S A. 1996 Nov 26;93[24]:14065-9). Makes sense and may explain the above smoking behavior. I truly believe cigarettes are as or more addictive than any other dopamine enhancing drug.
More than 50 years ago, a national campaign against smoking was launched after the 1964 Surgeon General’s report concluded that smoking was a major health hazard. (Looking back, one of the few losses of not having to pull journal articles from the stacks in the library, is that medical students and residents can’t shake their heads in wonder at the cigarette ads in old medical journals.) The impact of the national antismoking campaign has been dramatic, but smoking remains the leading preventable cause of death in the United States and globally, according to the Centers for Disease Control and Prevention.
in 2006, from 1992 (Arch Dermatol. 2010;146[3]:283-7), dermatologists had good footing on which to start a major prevention campaign. The American Cancer Society got on board, and in 2014, acting surgeon general Boris Lushniak, MD, issued a call to action to prevent skin cancer along with Howard Koh, MD, the assistant secretary of health, in “The Surgeon General’s Call to Action to Prevent Skin Cancer” in 2014, and the campaign was on.
Well, I am delighted to pass on a report from Leonard Lichtenfeld, MD, deputy chief medical officer for the American Cancer Society, who recently described in his March 2018 blog what may the first signs of the effectiveness of efforts to promote protection from ultraviolet ray exposure (JAMA Dermatol. 2018;154[3]:361-2). He writes: “In young white women ages 15 to 24, the incidence of melanoma has declined an average of 5.5% per year from January 2005 through December 2014. Not 5.5% over those ten years but 5.5 % PER YEAR. That’s remarkable, to say the least.”
As for the reasons behind these trends, he says, “no one can say for certain,” but he refers to national data indicating that indoor tanning has decreased in the past few years, especially among adolescents and young adults.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].