User login
Verrucous Carcinoma on the Lower Extremities
To the Editor:
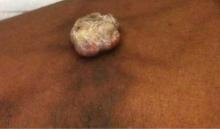
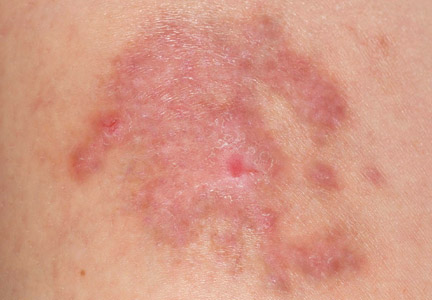
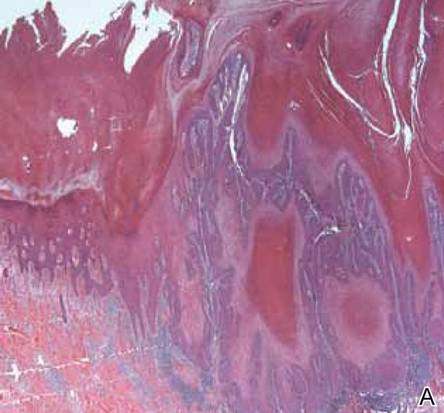
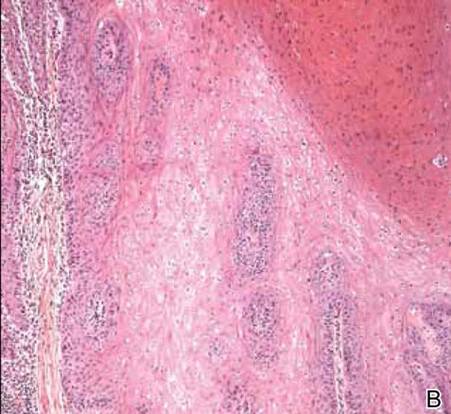
A 38-year-old black man presented with a slowly enlarging growth on the left thigh of 7 years’ duration. The lesion would occasionally scrape off but always recurred. He reported that the tumor developed in the area of a prior nevus. He reported no direct trauma to the area, chronic inflammation, or similar lesions elsewhere. His medical history included gastroesophageal reflux disease and inactive sarcoidosis. Physical examination revealed a 3×3×1-cm exophytic, hyperkeratotic, erythematous nodule with surrounding stellate and branching hyperpigmentation on the anterior aspect of the thigh (Figure 1). Pathologic examination demonstrated hyperkeratosis with an endophytic proliferation of mildly atypical keratinocytes with broad blunted rete ridges (Figure 2). Complete excision of the lesion was performed.
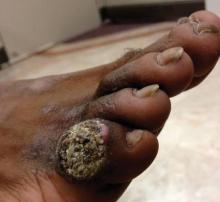
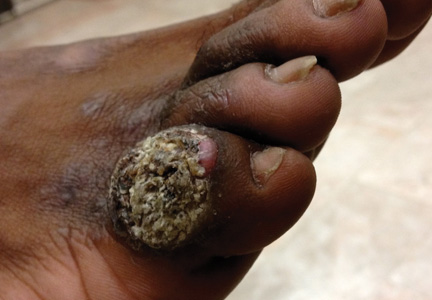
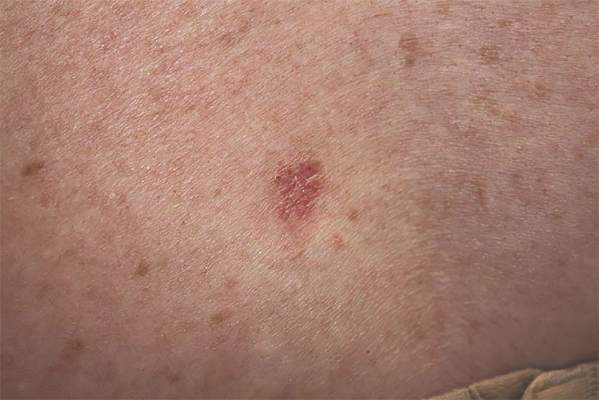
A 33-year-old black man presented with a rapidly growing lesion on the right fifth toe of 3 months’ duration. The patient originally believed the initial small papule was a corn, and after attempts to shave it down with a razor blade, the lesion grew rapidly into a large painful tumor. He reported no prior trauma to the area or history of a similar lesion. Physical examination revealed a 2×2×0.5-cm hyperkeratotic, papillated, hard nodule with a heaped-up border and no ulceration or drainage (Figure 3). A shave biopsy of the lesion was obtained. Microscopic examination revealed hyperkeratosis, parakeratosis, and papillomatosis with deep extension of mildly atypical keratinocytes into the dermis. Small toe amputation was performed by an orthopedic surgeon.
|
Verrucous carcinoma, first described by Ackerman1 in 1948, is an uncommon, low-grade, well-differentiated variant of squamous cell carcinoma. It presents as a slow-growing, bulky, exophytic tumor with a broad base. The tumor can ulcerate or present with surface sinus tracts that drain foul-smelling material. Typically, the tumor occurs in the fifth to sixth decades of life, with men outnumbering women by a ratio of 5.3 to 1.2 The prevalence of verrucous carcinoma in black individuals is unknown. A review of nonmelanoma skin cancers in skin of color identifies squamous cell carcinoma as the most common cutaneous carcinoma but does not report on the rare verrucous variant.3
Verrucous carcinoma is found in a variety of mucosal and skin surfaces. Verrucous carcinoma of the oral cavity, found most commonly on the buccal mucosa, is known as florid oral papillomatosis or Ackerman carcinoma. Cutaneous verrucous carcinoma is referred to as carcinoma cuniculatum or epithelioma cuniculatum and is predominantly located on the plantar surface of the foot. It is less commonly reported on the palm, scalp, face, extremities, and back. Verrucous carcinoma found in the anogenital area is referred to as the Buschke-Löwenstein tumor.4
Histologically, the lesion shows minimal cytologic atypia. Topped by an undulating keratinized mass, the deep margin of the tumor advances as a broad bulbous projection, compressing the underlying connective tissue in a bulldozing manner. Typically there also are keratin-filled sinuses and intraepidermal microabscesses.1
Human papillomavirus types 6, 11, 16, and 18 may be involved in the induction of the tumor. Human papillomavirus types 6 and 11 are frequently associated with the Buschke-Löwenstein tumor,2,4 while carcinoma cuniculatum is most commonly associated with human papillomavirus 16.5-7 In several cases of verrucous carcinoma, the tumor was reported to arise from preexisting lesions with chronic inflammation, such as a chronic ulcer, inflamed cyst, or burn scar.2 Ackerman carcinoma has been associated with the use of snuff, chewing tobacco, and betel nuts.
Morbidity and mortality from verrucous carcinoma arises from local invasion and infiltration into adjacent bone. The tumor rarely metastasizes, with regional lymph nodes being the only reported site of metastasis.8 The treatment of cutaneous verrucous carcinoma is complete surgical excision. Mohs micrographic surgery is preferred because it minimizes recurrence risk.4 Radiation therapy is contraindicated because it has been reported to cause the tumor to become more aggressive.9,10 Although local recurrence may occur, the prognosis is usually favorable.
1. Ackerman LV. Verrucous carcinoma of the oral cavity. Surgery. 1948;23:670-678.
2. Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin). a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
3. Jackson BA. Nonmelanoma skin cancer in persons of color. Semin Cutan Med Surg. 2009;28:93-95.
4. Schwartz RA. Verrucous carcinoma of the skin and mucosa. J Am Acad Dermatol. 1995;32:1-21; quiz 22-24.
5. Assaf C, Steinhoff M, Petrov I, et al. Verrucous carcinoma of the axilla: case report and review. J Cutan Pathol. 2004;31:199-204.
6. Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
7. Miyamoto T, Sasaoka R, Hagari Y, et al. Association of cutaneous verrucous carcinoma with human papillomavirus type 16. Br J Dermatol. 1999;140:168-169.
8. Walvekar RR, Chaukar DA, Deshpande MS, et al. Verrucous carcinoma of the oral cavity: a clinical and pathological study of 101 cases [published online ahead of print July 11, 2008]. Oral Oncol. 2009;45:47-51.
9. Perez CA, Krans FT, Evans JC, et al. Anaplastic transformation in verrucous carcinoma of the oral cavity after radiation therapy. Radiology. 1966;86:108-115.
10. Proffett SD, Spooner TR, Kosek JC. Origin of undifferentiated neoplasm from verrucous epidermal carcinoma of oral cavity following irradiation. Cancer. 1970;26:389-393.
To the Editor:
A 38-year-old black man presented with a slowly enlarging growth on the left thigh of 7 years’ duration. The lesion would occasionally scrape off but always recurred. He reported that the tumor developed in the area of a prior nevus. He reported no direct trauma to the area, chronic inflammation, or similar lesions elsewhere. His medical history included gastroesophageal reflux disease and inactive sarcoidosis. Physical examination revealed a 3×3×1-cm exophytic, hyperkeratotic, erythematous nodule with surrounding stellate and branching hyperpigmentation on the anterior aspect of the thigh (Figure 1). Pathologic examination demonstrated hyperkeratosis with an endophytic proliferation of mildly atypical keratinocytes with broad blunted rete ridges (Figure 2). Complete excision of the lesion was performed.
A 33-year-old black man presented with a rapidly growing lesion on the right fifth toe of 3 months’ duration. The patient originally believed the initial small papule was a corn, and after attempts to shave it down with a razor blade, the lesion grew rapidly into a large painful tumor. He reported no prior trauma to the area or history of a similar lesion. Physical examination revealed a 2×2×0.5-cm hyperkeratotic, papillated, hard nodule with a heaped-up border and no ulceration or drainage (Figure 3). A shave biopsy of the lesion was obtained. Microscopic examination revealed hyperkeratosis, parakeratosis, and papillomatosis with deep extension of mildly atypical keratinocytes into the dermis. Small toe amputation was performed by an orthopedic surgeon.


|
Verrucous carcinoma, first described by Ackerman1 in 1948, is an uncommon, low-grade, well-differentiated variant of squamous cell carcinoma. It presents as a slow-growing, bulky, exophytic tumor with a broad base. The tumor can ulcerate or present with surface sinus tracts that drain foul-smelling material. Typically, the tumor occurs in the fifth to sixth decades of life, with men outnumbering women by a ratio of 5.3 to 1.2 The prevalence of verrucous carcinoma in black individuals is unknown. A review of nonmelanoma skin cancers in skin of color identifies squamous cell carcinoma as the most common cutaneous carcinoma but does not report on the rare verrucous variant.3
Verrucous carcinoma is found in a variety of mucosal and skin surfaces. Verrucous carcinoma of the oral cavity, found most commonly on the buccal mucosa, is known as florid oral papillomatosis or Ackerman carcinoma. Cutaneous verrucous carcinoma is referred to as carcinoma cuniculatum or epithelioma cuniculatum and is predominantly located on the plantar surface of the foot. It is less commonly reported on the palm, scalp, face, extremities, and back. Verrucous carcinoma found in the anogenital area is referred to as the Buschke-Löwenstein tumor.4
Histologically, the lesion shows minimal cytologic atypia. Topped by an undulating keratinized mass, the deep margin of the tumor advances as a broad bulbous projection, compressing the underlying connective tissue in a bulldozing manner. Typically there also are keratin-filled sinuses and intraepidermal microabscesses.1
Human papillomavirus types 6, 11, 16, and 18 may be involved in the induction of the tumor. Human papillomavirus types 6 and 11 are frequently associated with the Buschke-Löwenstein tumor,2,4 while carcinoma cuniculatum is most commonly associated with human papillomavirus 16.5-7 In several cases of verrucous carcinoma, the tumor was reported to arise from preexisting lesions with chronic inflammation, such as a chronic ulcer, inflamed cyst, or burn scar.2 Ackerman carcinoma has been associated with the use of snuff, chewing tobacco, and betel nuts.
Morbidity and mortality from verrucous carcinoma arises from local invasion and infiltration into adjacent bone. The tumor rarely metastasizes, with regional lymph nodes being the only reported site of metastasis.8 The treatment of cutaneous verrucous carcinoma is complete surgical excision. Mohs micrographic surgery is preferred because it minimizes recurrence risk.4 Radiation therapy is contraindicated because it has been reported to cause the tumor to become more aggressive.9,10 Although local recurrence may occur, the prognosis is usually favorable.
To the Editor:
A 38-year-old black man presented with a slowly enlarging growth on the left thigh of 7 years’ duration. The lesion would occasionally scrape off but always recurred. He reported that the tumor developed in the area of a prior nevus. He reported no direct trauma to the area, chronic inflammation, or similar lesions elsewhere. His medical history included gastroesophageal reflux disease and inactive sarcoidosis. Physical examination revealed a 3×3×1-cm exophytic, hyperkeratotic, erythematous nodule with surrounding stellate and branching hyperpigmentation on the anterior aspect of the thigh (Figure 1). Pathologic examination demonstrated hyperkeratosis with an endophytic proliferation of mildly atypical keratinocytes with broad blunted rete ridges (Figure 2). Complete excision of the lesion was performed.
A 33-year-old black man presented with a rapidly growing lesion on the right fifth toe of 3 months’ duration. The patient originally believed the initial small papule was a corn, and after attempts to shave it down with a razor blade, the lesion grew rapidly into a large painful tumor. He reported no prior trauma to the area or history of a similar lesion. Physical examination revealed a 2×2×0.5-cm hyperkeratotic, papillated, hard nodule with a heaped-up border and no ulceration or drainage (Figure 3). A shave biopsy of the lesion was obtained. Microscopic examination revealed hyperkeratosis, parakeratosis, and papillomatosis with deep extension of mildly atypical keratinocytes into the dermis. Small toe amputation was performed by an orthopedic surgeon.


|
Verrucous carcinoma, first described by Ackerman1 in 1948, is an uncommon, low-grade, well-differentiated variant of squamous cell carcinoma. It presents as a slow-growing, bulky, exophytic tumor with a broad base. The tumor can ulcerate or present with surface sinus tracts that drain foul-smelling material. Typically, the tumor occurs in the fifth to sixth decades of life, with men outnumbering women by a ratio of 5.3 to 1.2 The prevalence of verrucous carcinoma in black individuals is unknown. A review of nonmelanoma skin cancers in skin of color identifies squamous cell carcinoma as the most common cutaneous carcinoma but does not report on the rare verrucous variant.3
Verrucous carcinoma is found in a variety of mucosal and skin surfaces. Verrucous carcinoma of the oral cavity, found most commonly on the buccal mucosa, is known as florid oral papillomatosis or Ackerman carcinoma. Cutaneous verrucous carcinoma is referred to as carcinoma cuniculatum or epithelioma cuniculatum and is predominantly located on the plantar surface of the foot. It is less commonly reported on the palm, scalp, face, extremities, and back. Verrucous carcinoma found in the anogenital area is referred to as the Buschke-Löwenstein tumor.4
Histologically, the lesion shows minimal cytologic atypia. Topped by an undulating keratinized mass, the deep margin of the tumor advances as a broad bulbous projection, compressing the underlying connective tissue in a bulldozing manner. Typically there also are keratin-filled sinuses and intraepidermal microabscesses.1
Human papillomavirus types 6, 11, 16, and 18 may be involved in the induction of the tumor. Human papillomavirus types 6 and 11 are frequently associated with the Buschke-Löwenstein tumor,2,4 while carcinoma cuniculatum is most commonly associated with human papillomavirus 16.5-7 In several cases of verrucous carcinoma, the tumor was reported to arise from preexisting lesions with chronic inflammation, such as a chronic ulcer, inflamed cyst, or burn scar.2 Ackerman carcinoma has been associated with the use of snuff, chewing tobacco, and betel nuts.
Morbidity and mortality from verrucous carcinoma arises from local invasion and infiltration into adjacent bone. The tumor rarely metastasizes, with regional lymph nodes being the only reported site of metastasis.8 The treatment of cutaneous verrucous carcinoma is complete surgical excision. Mohs micrographic surgery is preferred because it minimizes recurrence risk.4 Radiation therapy is contraindicated because it has been reported to cause the tumor to become more aggressive.9,10 Although local recurrence may occur, the prognosis is usually favorable.
1. Ackerman LV. Verrucous carcinoma of the oral cavity. Surgery. 1948;23:670-678.
2. Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin). a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
3. Jackson BA. Nonmelanoma skin cancer in persons of color. Semin Cutan Med Surg. 2009;28:93-95.
4. Schwartz RA. Verrucous carcinoma of the skin and mucosa. J Am Acad Dermatol. 1995;32:1-21; quiz 22-24.
5. Assaf C, Steinhoff M, Petrov I, et al. Verrucous carcinoma of the axilla: case report and review. J Cutan Pathol. 2004;31:199-204.
6. Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
7. Miyamoto T, Sasaoka R, Hagari Y, et al. Association of cutaneous verrucous carcinoma with human papillomavirus type 16. Br J Dermatol. 1999;140:168-169.
8. Walvekar RR, Chaukar DA, Deshpande MS, et al. Verrucous carcinoma of the oral cavity: a clinical and pathological study of 101 cases [published online ahead of print July 11, 2008]. Oral Oncol. 2009;45:47-51.
9. Perez CA, Krans FT, Evans JC, et al. Anaplastic transformation in verrucous carcinoma of the oral cavity after radiation therapy. Radiology. 1966;86:108-115.
10. Proffett SD, Spooner TR, Kosek JC. Origin of undifferentiated neoplasm from verrucous epidermal carcinoma of oral cavity following irradiation. Cancer. 1970;26:389-393.
1. Ackerman LV. Verrucous carcinoma of the oral cavity. Surgery. 1948;23:670-678.
2. Kao GF, Graham JH, Helwig EB. Carcinoma cuniculatum (verrucous carcinoma of the skin). a clinicopathologic study of 46 cases with ultrastructural observations. Cancer. 1982;49:2395-2403.
3. Jackson BA. Nonmelanoma skin cancer in persons of color. Semin Cutan Med Surg. 2009;28:93-95.
4. Schwartz RA. Verrucous carcinoma of the skin and mucosa. J Am Acad Dermatol. 1995;32:1-21; quiz 22-24.
5. Assaf C, Steinhoff M, Petrov I, et al. Verrucous carcinoma of the axilla: case report and review. J Cutan Pathol. 2004;31:199-204.
6. Schell BJ, Rosen T, Rády P, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol. 2001;45:49-55.
7. Miyamoto T, Sasaoka R, Hagari Y, et al. Association of cutaneous verrucous carcinoma with human papillomavirus type 16. Br J Dermatol. 1999;140:168-169.
8. Walvekar RR, Chaukar DA, Deshpande MS, et al. Verrucous carcinoma of the oral cavity: a clinical and pathological study of 101 cases [published online ahead of print July 11, 2008]. Oral Oncol. 2009;45:47-51.
9. Perez CA, Krans FT, Evans JC, et al. Anaplastic transformation in verrucous carcinoma of the oral cavity after radiation therapy. Radiology. 1966;86:108-115.
10. Proffett SD, Spooner TR, Kosek JC. Origin of undifferentiated neoplasm from verrucous epidermal carcinoma of oral cavity following irradiation. Cancer. 1970;26:389-393.
Round Purple Erythematous Tumors
The Diagnosis: Skin Metastases of Vulvar Carcinoma
A Tzanck cytodiagnosis was performed and a punch biopsy specimen was obtained for histologic examination. The patient’s Tzanck cytodiagnosis was initially interpreted as a viral cytopathic effect due to the presence of multinucleated atypical cells; hence, she was treated with intravenous acyclovir. The biopsy specimen showed a preserved epidermis and a dense infiltrate in the mid and deep dermis (Figure 1) formed by atypical squamous cells with enlarged nuclei and anisokaryosis, evident nucleoli, atypical mitoses, and multinucleated cells (Figure 2). The histopathologic diagnosis was skin metastases of moderately differentiated squamous cell carcinoma. The subsequent clinical course was unfavorableand the patient died during hospitalization from septic shock.
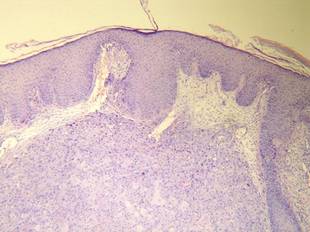
Figure 1. Histology revealed a preserved epidermis with a dense infiltrate in the mid and deep dermis (H&E, original magnification ×40). | 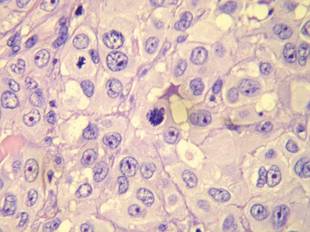
Figure 2. Atypical squamous cells with enlarged nuclei and anisokaryosis, evident nucleoli, atypical mitoses, and multinucleated cells were evident (H&E, original magnification ×400). |
Cutaneous metastases occur in 0.7% to 9% of solid tumors in advanced stages of disease progression or occasionally as an initial manifestation; they are a predictor of poor prognosis and breast cancer is the most frequent cause in women.1 Vulvar carcinoma comprises 5% of all malignant neoplasms of the female genital tract and 95% are squamous cell carcinoma; metastases appear most frequently in the inguinal and pelvic lymph nodes, followed by the lungs, liver, and bones.2 Skin metastases are extremely rare, with few cases documented.2,3
Clinically, skin metastases present most frequently as nodules, either solitary or multiple, that are sometimes ulcerated. However, a wide spectrum of metastases has been described, including erysipeloid, sclerodermiform (en cuirasse), telangiectatic papulovesicles, purpuric plaques mimicking vasculitis, alopecia areata–like scalp lesions, and others. The zosteriform pattern has been described in few cases, including vesiculobullous herpetiform lesions or nodular metastases with metameric distribution.4 In more than half of cases, the lesions were initially interpreted as herpes zoster and hence treated with acyclovir.5
Regarding cutaneous metastases of vulvar carcinoma, a case of metastases mimicking primary varicella-zoster virus infection has been reported,3 but our case represents a zosteriform pattern, which is unique.
Multiple theories have been proposed to explain the pathogenic mechanism of zosteriform spread, though none have been adequately proven.4-7 Some of the patients described in the literature had a history of viral infection by serology or polymerase chain reaction for herpes simplex virus types 1 and 2 and varicella-zoster virus in the same dermatome where metastases were observed. It has been suggested that neural damage caused by the herpesvirus results in an immune function impairment of the overlying skin, which consequently may be more receptive to the inflow of metastatic cells. In such cases, the zosteriform pattern could be due to a Wolf or Köbner phenomenon in an area of vulnerable skin.4-9
Other possible explanations for the zosteriform spread are the invasion of perineural lymphatic vessels or of the dorsal root ganglion, direct invasion from deeper structures, or the surgical implantation of neoplastic cells,4-7 though the latter 2 mechanisms do not correspond to true metastases.
1. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33(2, pt 1):161-182.
2. Wang AR, O’Brien M, Ross R, et al. Epidermotropic metastasis from vulvar squamous cell carcinoma: a rare cutaneous manifestation. J Am Acad Dermatol. 2010;63:1088-1091.
3. Mailhot J, O’Donnell P, Han R, et al. Metastatic squamous cell carcinoma of the vulva mimicking primary varicella-zoster virus infection. J Am Acad Dermatol. 2011;65:e63-e64.
4. Savoia P, Fava P, Deboli T, et al. Zosteriform cutaneous metastases: a literature meta-analysis and a clinical report of three melanoma cases. Dermatol Surg. 2009;35:1355-1363.
5. Niiyama S, Satoh K, Kaneko S, et al. Zosteriform skin involvement of nodal T-cell lymphoma: a review of the published work of cutaneous malignancies mimicking herpes zoster. J Dermatol. 2007;34:68-73.
6. Zalaudek I, Leinweber B, Richtig E, et al. Cutaneous zosteriform melanoma metastases arising after herpes zoster infection: a case report and review of the literature. Melanoma Res. 2003;13:635-639.
7. LeSueur BW, Abraham LJ, DiCaudo DJ, et al. Zosteriform skin metastases. Int J Dermatol. 2004;43:126-128.
8. Ruocco V, Ruocco E, Brunetti G, et al. Opportunistic localization of skin lesions on vulnerable areas. Clin Dermatol. 2011;29:483-488.
9. Wolf R, Wolf D, Ruocco E, et al. Wolf’s isotopic response. Clin Dermatol. 2011;29:237-240.
The Diagnosis: Skin Metastases of Vulvar Carcinoma
A Tzanck cytodiagnosis was performed and a punch biopsy specimen was obtained for histologic examination. The patient’s Tzanck cytodiagnosis was initially interpreted as a viral cytopathic effect due to the presence of multinucleated atypical cells; hence, she was treated with intravenous acyclovir. The biopsy specimen showed a preserved epidermis and a dense infiltrate in the mid and deep dermis (Figure 1) formed by atypical squamous cells with enlarged nuclei and anisokaryosis, evident nucleoli, atypical mitoses, and multinucleated cells (Figure 2). The histopathologic diagnosis was skin metastases of moderately differentiated squamous cell carcinoma. The subsequent clinical course was unfavorableand the patient died during hospitalization from septic shock.

Figure 1. Histology revealed a preserved epidermis with a dense infiltrate in the mid and deep dermis (H&E, original magnification ×40). | 
Figure 2. Atypical squamous cells with enlarged nuclei and anisokaryosis, evident nucleoli, atypical mitoses, and multinucleated cells were evident (H&E, original magnification ×400). |
Cutaneous metastases occur in 0.7% to 9% of solid tumors in advanced stages of disease progression or occasionally as an initial manifestation; they are a predictor of poor prognosis and breast cancer is the most frequent cause in women.1 Vulvar carcinoma comprises 5% of all malignant neoplasms of the female genital tract and 95% are squamous cell carcinoma; metastases appear most frequently in the inguinal and pelvic lymph nodes, followed by the lungs, liver, and bones.2 Skin metastases are extremely rare, with few cases documented.2,3
Clinically, skin metastases present most frequently as nodules, either solitary or multiple, that are sometimes ulcerated. However, a wide spectrum of metastases has been described, including erysipeloid, sclerodermiform (en cuirasse), telangiectatic papulovesicles, purpuric plaques mimicking vasculitis, alopecia areata–like scalp lesions, and others. The zosteriform pattern has been described in few cases, including vesiculobullous herpetiform lesions or nodular metastases with metameric distribution.4 In more than half of cases, the lesions were initially interpreted as herpes zoster and hence treated with acyclovir.5
Regarding cutaneous metastases of vulvar carcinoma, a case of metastases mimicking primary varicella-zoster virus infection has been reported,3 but our case represents a zosteriform pattern, which is unique.
Multiple theories have been proposed to explain the pathogenic mechanism of zosteriform spread, though none have been adequately proven.4-7 Some of the patients described in the literature had a history of viral infection by serology or polymerase chain reaction for herpes simplex virus types 1 and 2 and varicella-zoster virus in the same dermatome where metastases were observed. It has been suggested that neural damage caused by the herpesvirus results in an immune function impairment of the overlying skin, which consequently may be more receptive to the inflow of metastatic cells. In such cases, the zosteriform pattern could be due to a Wolf or Köbner phenomenon in an area of vulnerable skin.4-9
Other possible explanations for the zosteriform spread are the invasion of perineural lymphatic vessels or of the dorsal root ganglion, direct invasion from deeper structures, or the surgical implantation of neoplastic cells,4-7 though the latter 2 mechanisms do not correspond to true metastases.
The Diagnosis: Skin Metastases of Vulvar Carcinoma
A Tzanck cytodiagnosis was performed and a punch biopsy specimen was obtained for histologic examination. The patient’s Tzanck cytodiagnosis was initially interpreted as a viral cytopathic effect due to the presence of multinucleated atypical cells; hence, she was treated with intravenous acyclovir. The biopsy specimen showed a preserved epidermis and a dense infiltrate in the mid and deep dermis (Figure 1) formed by atypical squamous cells with enlarged nuclei and anisokaryosis, evident nucleoli, atypical mitoses, and multinucleated cells (Figure 2). The histopathologic diagnosis was skin metastases of moderately differentiated squamous cell carcinoma. The subsequent clinical course was unfavorableand the patient died during hospitalization from septic shock.

Figure 1. Histology revealed a preserved epidermis with a dense infiltrate in the mid and deep dermis (H&E, original magnification ×40). | 
Figure 2. Atypical squamous cells with enlarged nuclei and anisokaryosis, evident nucleoli, atypical mitoses, and multinucleated cells were evident (H&E, original magnification ×400). |
Cutaneous metastases occur in 0.7% to 9% of solid tumors in advanced stages of disease progression or occasionally as an initial manifestation; they are a predictor of poor prognosis and breast cancer is the most frequent cause in women.1 Vulvar carcinoma comprises 5% of all malignant neoplasms of the female genital tract and 95% are squamous cell carcinoma; metastases appear most frequently in the inguinal and pelvic lymph nodes, followed by the lungs, liver, and bones.2 Skin metastases are extremely rare, with few cases documented.2,3
Clinically, skin metastases present most frequently as nodules, either solitary or multiple, that are sometimes ulcerated. However, a wide spectrum of metastases has been described, including erysipeloid, sclerodermiform (en cuirasse), telangiectatic papulovesicles, purpuric plaques mimicking vasculitis, alopecia areata–like scalp lesions, and others. The zosteriform pattern has been described in few cases, including vesiculobullous herpetiform lesions or nodular metastases with metameric distribution.4 In more than half of cases, the lesions were initially interpreted as herpes zoster and hence treated with acyclovir.5
Regarding cutaneous metastases of vulvar carcinoma, a case of metastases mimicking primary varicella-zoster virus infection has been reported,3 but our case represents a zosteriform pattern, which is unique.
Multiple theories have been proposed to explain the pathogenic mechanism of zosteriform spread, though none have been adequately proven.4-7 Some of the patients described in the literature had a history of viral infection by serology or polymerase chain reaction for herpes simplex virus types 1 and 2 and varicella-zoster virus in the same dermatome where metastases were observed. It has been suggested that neural damage caused by the herpesvirus results in an immune function impairment of the overlying skin, which consequently may be more receptive to the inflow of metastatic cells. In such cases, the zosteriform pattern could be due to a Wolf or Köbner phenomenon in an area of vulnerable skin.4-9
Other possible explanations for the zosteriform spread are the invasion of perineural lymphatic vessels or of the dorsal root ganglion, direct invasion from deeper structures, or the surgical implantation of neoplastic cells,4-7 though the latter 2 mechanisms do not correspond to true metastases.
1. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33(2, pt 1):161-182.
2. Wang AR, O’Brien M, Ross R, et al. Epidermotropic metastasis from vulvar squamous cell carcinoma: a rare cutaneous manifestation. J Am Acad Dermatol. 2010;63:1088-1091.
3. Mailhot J, O’Donnell P, Han R, et al. Metastatic squamous cell carcinoma of the vulva mimicking primary varicella-zoster virus infection. J Am Acad Dermatol. 2011;65:e63-e64.
4. Savoia P, Fava P, Deboli T, et al. Zosteriform cutaneous metastases: a literature meta-analysis and a clinical report of three melanoma cases. Dermatol Surg. 2009;35:1355-1363.
5. Niiyama S, Satoh K, Kaneko S, et al. Zosteriform skin involvement of nodal T-cell lymphoma: a review of the published work of cutaneous malignancies mimicking herpes zoster. J Dermatol. 2007;34:68-73.
6. Zalaudek I, Leinweber B, Richtig E, et al. Cutaneous zosteriform melanoma metastases arising after herpes zoster infection: a case report and review of the literature. Melanoma Res. 2003;13:635-639.
7. LeSueur BW, Abraham LJ, DiCaudo DJ, et al. Zosteriform skin metastases. Int J Dermatol. 2004;43:126-128.
8. Ruocco V, Ruocco E, Brunetti G, et al. Opportunistic localization of skin lesions on vulnerable areas. Clin Dermatol. 2011;29:483-488.
9. Wolf R, Wolf D, Ruocco E, et al. Wolf’s isotopic response. Clin Dermatol. 2011;29:237-240.
1. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33(2, pt 1):161-182.
2. Wang AR, O’Brien M, Ross R, et al. Epidermotropic metastasis from vulvar squamous cell carcinoma: a rare cutaneous manifestation. J Am Acad Dermatol. 2010;63:1088-1091.
3. Mailhot J, O’Donnell P, Han R, et al. Metastatic squamous cell carcinoma of the vulva mimicking primary varicella-zoster virus infection. J Am Acad Dermatol. 2011;65:e63-e64.
4. Savoia P, Fava P, Deboli T, et al. Zosteriform cutaneous metastases: a literature meta-analysis and a clinical report of three melanoma cases. Dermatol Surg. 2009;35:1355-1363.
5. Niiyama S, Satoh K, Kaneko S, et al. Zosteriform skin involvement of nodal T-cell lymphoma: a review of the published work of cutaneous malignancies mimicking herpes zoster. J Dermatol. 2007;34:68-73.
6. Zalaudek I, Leinweber B, Richtig E, et al. Cutaneous zosteriform melanoma metastases arising after herpes zoster infection: a case report and review of the literature. Melanoma Res. 2003;13:635-639.
7. LeSueur BW, Abraham LJ, DiCaudo DJ, et al. Zosteriform skin metastases. Int J Dermatol. 2004;43:126-128.
8. Ruocco V, Ruocco E, Brunetti G, et al. Opportunistic localization of skin lesions on vulnerable areas. Clin Dermatol. 2011;29:483-488.
9. Wolf R, Wolf D, Ruocco E, et al. Wolf’s isotopic response. Clin Dermatol. 2011;29:237-240.
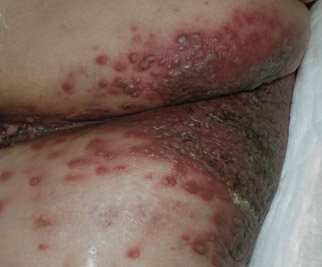
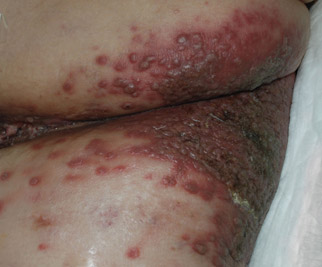
A 48-year-old woman presented with a dermatosis on the left thigh. She had a history of hypertension, obesity, and a stage IIIC vulvar carcinoma treated with radical vulvectomy and bilateral inguinal lymphadenectomy. Two months after surgery she was hospitalized because of a left popliteal deep vein thrombosis and an abscess involving the site of the lymphadenectomy. One month later she presented with a painful dermatosis on the left thigh. Physical examination revealed multiple round-shaped, purple, erythematous tumors with a smooth surface on the upper third of the left thigh extending to the lower abdomen. The tumors measured 0.5 cm in diameter and were grouped to form an indurated plaque on an erythematous base; some tumors were arranged as satellite lesions in the periphery. The dermatosis had a distinct zosteriform appearance.
FDA approves hedgehog pathway inhibitor for locally advanced BCC
Sonidegib, a hedgehog pathway inhibitor, has been approved for treating patients with locally advanced basal cell carcinoma (BCC) that has recurred following surgery or radiation therapy, or those who are not candidates for surgery or radiation therapy, the Food and Drug Administration announced on July 24. The drug is taken once a day, at a recommended dose of 200 mg on an empty stomach and will be marketed as Odomzo by Novartis Pharmaceuticals. Approval was based on a study that found the objective response rate to be 58% among patients treated with the 200-mg dose; an effect that lasted for about 2-19 months, according to the FDA.
“Our increasing understanding of molecular pathways involved in cancer has led to approvals of many oncology drugs in difficult-to-treat diseases for which few therapeutic options previously existed,” Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement. “Thanks to a better understanding of the hedgehog pathway, the FDA has now approved two drugs for the treatment of basal cell carcinoma just in the last 3 years,” he added.
The first drug to treat locally advanced and metastatic basal cell carcinoma was vismodegib, marketed as Erivedge by Genentech in 2012.
The most common adverse events associated with the 200-mg dose include muscle spasms, alopecia, dysgeusia, fatigue, nausea, musculoskeletal pain, diarrhea, decreased weight, decreased appetite, myalgia, abdominal pain, headache, pain, vomiting, and pruritus (itching). It has also been associated “with rare reports” of rhabdomyolysis, muscle spasms, and myalgia, according to the FDA.
The prescribing information includes a boxed warning about the risk of embryo-fetal toxicity, and includes recommendations to verify that women of reproductive potential are not pregnant before starting treatment and to use effective contraception during treatment and for at least 20 months after the last dose. Men on this treatment should also be advised about “the potential risk of exposure through semen and to use condoms with a pregnant partner or a female partner of reproductive potential during treatment,” and for at least 8 months after stopping treatment.
More information on the approval is available on the FDA website at www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm455865.htm.
Exposed pregnancies should be reported to Novartis at 888-669-6682. Serious adverse events associated with sonidegib should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/Safety/MedWatch.
Sonidegib, a hedgehog pathway inhibitor, has been approved for treating patients with locally advanced basal cell carcinoma (BCC) that has recurred following surgery or radiation therapy, or those who are not candidates for surgery or radiation therapy, the Food and Drug Administration announced on July 24. The drug is taken once a day, at a recommended dose of 200 mg on an empty stomach and will be marketed as Odomzo by Novartis Pharmaceuticals. Approval was based on a study that found the objective response rate to be 58% among patients treated with the 200-mg dose; an effect that lasted for about 2-19 months, according to the FDA.
“Our increasing understanding of molecular pathways involved in cancer has led to approvals of many oncology drugs in difficult-to-treat diseases for which few therapeutic options previously existed,” Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement. “Thanks to a better understanding of the hedgehog pathway, the FDA has now approved two drugs for the treatment of basal cell carcinoma just in the last 3 years,” he added.
The first drug to treat locally advanced and metastatic basal cell carcinoma was vismodegib, marketed as Erivedge by Genentech in 2012.
The most common adverse events associated with the 200-mg dose include muscle spasms, alopecia, dysgeusia, fatigue, nausea, musculoskeletal pain, diarrhea, decreased weight, decreased appetite, myalgia, abdominal pain, headache, pain, vomiting, and pruritus (itching). It has also been associated “with rare reports” of rhabdomyolysis, muscle spasms, and myalgia, according to the FDA.
The prescribing information includes a boxed warning about the risk of embryo-fetal toxicity, and includes recommendations to verify that women of reproductive potential are not pregnant before starting treatment and to use effective contraception during treatment and for at least 20 months after the last dose. Men on this treatment should also be advised about “the potential risk of exposure through semen and to use condoms with a pregnant partner or a female partner of reproductive potential during treatment,” and for at least 8 months after stopping treatment.
More information on the approval is available on the FDA website at www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm455865.htm.
Exposed pregnancies should be reported to Novartis at 888-669-6682. Serious adverse events associated with sonidegib should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/Safety/MedWatch.
Sonidegib, a hedgehog pathway inhibitor, has been approved for treating patients with locally advanced basal cell carcinoma (BCC) that has recurred following surgery or radiation therapy, or those who are not candidates for surgery or radiation therapy, the Food and Drug Administration announced on July 24. The drug is taken once a day, at a recommended dose of 200 mg on an empty stomach and will be marketed as Odomzo by Novartis Pharmaceuticals. Approval was based on a study that found the objective response rate to be 58% among patients treated with the 200-mg dose; an effect that lasted for about 2-19 months, according to the FDA.
“Our increasing understanding of molecular pathways involved in cancer has led to approvals of many oncology drugs in difficult-to-treat diseases for which few therapeutic options previously existed,” Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement. “Thanks to a better understanding of the hedgehog pathway, the FDA has now approved two drugs for the treatment of basal cell carcinoma just in the last 3 years,” he added.
The first drug to treat locally advanced and metastatic basal cell carcinoma was vismodegib, marketed as Erivedge by Genentech in 2012.
The most common adverse events associated with the 200-mg dose include muscle spasms, alopecia, dysgeusia, fatigue, nausea, musculoskeletal pain, diarrhea, decreased weight, decreased appetite, myalgia, abdominal pain, headache, pain, vomiting, and pruritus (itching). It has also been associated “with rare reports” of rhabdomyolysis, muscle spasms, and myalgia, according to the FDA.
The prescribing information includes a boxed warning about the risk of embryo-fetal toxicity, and includes recommendations to verify that women of reproductive potential are not pregnant before starting treatment and to use effective contraception during treatment and for at least 20 months after the last dose. Men on this treatment should also be advised about “the potential risk of exposure through semen and to use condoms with a pregnant partner or a female partner of reproductive potential during treatment,” and for at least 8 months after stopping treatment.
More information on the approval is available on the FDA website at www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm455865.htm.
Exposed pregnancies should be reported to Novartis at 888-669-6682. Serious adverse events associated with sonidegib should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/Safety/MedWatch.
Amlodipine reduced vismodegib-induced muscle cramps
The calcium channel blocker amlodipine besylate was effective in reducing the number of muscle cramps caused by vismodegib, a basal cell carcinoma drug, according to a research letter from Dr. Mina Ally and her associates.
Patients who took amlodipine had the number of muscle cramps halved after 2 weeks of treatment, and this level was maintained for the 8-week medication regimen. No significant change in cramp severity, duration, or frequency of nighttime awakenings was seen. Side effects only appeared in too patients, with one reporting mild intermittent dizziness and another reporting grade 1 peripheral edema.
The control group saw a nonsignificant increase in cramp frequency, compared with the significant decrease in the amlodipine group. No change was seen in cramp severity, duration, or number of nighttime awakenings in the control group.
“Amlodipine may be effective in vismodegib-induced muscle cramps because it blocks voltage-gated calcium channels and inhibits the transport of extracellular calcium into muscle that is required for contraction,” the investigators noted.
Find the full research letter in JAMA Dermatology (doi:10.1001/jamadermatol.2015.1937).
The calcium channel blocker amlodipine besylate was effective in reducing the number of muscle cramps caused by vismodegib, a basal cell carcinoma drug, according to a research letter from Dr. Mina Ally and her associates.
Patients who took amlodipine had the number of muscle cramps halved after 2 weeks of treatment, and this level was maintained for the 8-week medication regimen. No significant change in cramp severity, duration, or frequency of nighttime awakenings was seen. Side effects only appeared in too patients, with one reporting mild intermittent dizziness and another reporting grade 1 peripheral edema.
The control group saw a nonsignificant increase in cramp frequency, compared with the significant decrease in the amlodipine group. No change was seen in cramp severity, duration, or number of nighttime awakenings in the control group.
“Amlodipine may be effective in vismodegib-induced muscle cramps because it blocks voltage-gated calcium channels and inhibits the transport of extracellular calcium into muscle that is required for contraction,” the investigators noted.
Find the full research letter in JAMA Dermatology (doi:10.1001/jamadermatol.2015.1937).
The calcium channel blocker amlodipine besylate was effective in reducing the number of muscle cramps caused by vismodegib, a basal cell carcinoma drug, according to a research letter from Dr. Mina Ally and her associates.
Patients who took amlodipine had the number of muscle cramps halved after 2 weeks of treatment, and this level was maintained for the 8-week medication regimen. No significant change in cramp severity, duration, or frequency of nighttime awakenings was seen. Side effects only appeared in too patients, with one reporting mild intermittent dizziness and another reporting grade 1 peripheral edema.
The control group saw a nonsignificant increase in cramp frequency, compared with the significant decrease in the amlodipine group. No change was seen in cramp severity, duration, or number of nighttime awakenings in the control group.
“Amlodipine may be effective in vismodegib-induced muscle cramps because it blocks voltage-gated calcium channels and inhibits the transport of extracellular calcium into muscle that is required for contraction,” the investigators noted.
Find the full research letter in JAMA Dermatology (doi:10.1001/jamadermatol.2015.1937).
Patient Compliance With Photoprotection
What does your patient need to know at the first visit?
Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history of melanoma and nonmelanoma skin cancer; skin examination for photodamage and photoaging as well as number and type of pigmented lesions; and lifestyle history, which should include location of residence as well as occupation and recreational pursuits. This discussion should, as usual, include questions about general health, systemic and skin disease, and medication usage, with particular focus on photoaggravated diseases such as lupus and melasma as well as ongoing use of topical agents and systemic photosensitizers. These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer and other specific conditions that alter the advice you would give.
What are your go-to treatments? Is your recommendation anecdotal or evidence based? What are the side effects?
I always recommend that my patients use a product that they like, which may sound simplistic. But if the patient doesn’t like the feel and look of the sunscreen, he/she won’t use it. Patients routinely should use a sunscreen with a sun protection factor (SPF) of 30 or higher that also carries a “broad spectrum” label. At the beach or during sweaty sports, patients should use one with a water-resistant SPF.
I prefer spray sunscreens for application on the back if the patient is alone without someone to help apply sunscreen to hard-to-reach areas and for male scalps. But you never know how much spray to use, so use a lot!
If patients are at the beach, playing sports, or watching sports outside, then they should reapply sunscreen every 2 hours. If patients work indoors and use a facial sunscreen in the morning, that’s sufficient.
Although there is no evidence that sunscreens are harmful for children older than 6 months of age and pregnant women, if patients in these special populations have concerns, I recommend using agents with inorganic compounds (physical blockers) such as titanium dioxide and zinc oxide only. Children are best protected with clothing and hats.
The evidence supports this approach. Patients really don’t need SPF 30 protection, but no one uses the amount of product that will result in the SPF listed on the bottle. So if patients use an SPF 30 or greater, they will get at least an SPF 15, which is sufficient everywhere but at the equator. Using SPF 30 the way we all apply it will give SPF 15–level protection.
There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma.
The biggest side effect is not using the sunscreen. Others include irritation and allergy. Irritation is common, but finding a product to use without irritation should be easy. Allergy is rarer, and when it occurs, it is usually due to the preservative or fragrance, not the active ingredients. If allergy does occur, patch testing by a dermatologist is necessary to determine the allergen.
Although it is still controversial, wearing sunscreens religiously can lead to vitamin D insufficiency or deficiency, which is particularly true for individuals with skin of color—Fitzpatrick skin types IV, V, and VI—and those cancer patients who adhere to rigorous photoprotection. These patients should be encouraged to take supplemental vitamin D3 and I suggest 2000 IU; this recommendation is my opinion and is not evidence based.
As to the literature in the laypress about hormonal changes from benzophenone, cancer from retinoids, and nanoparticle toxicity: There is no evidence to support those claims.
How do you keep patients compliant with treatment?
Keep telling them, and then tell them again.
What do you do if they refuse treatment?
Tell them to see someone else.
What resources do you recommend to patients for more information?
Consult the American Academy of Dermatology Web site (www.aad.org) and the Skin Cancer Foundation (www.skincancer.org).
Editorial Note
Practical Pearls From the Cutis® Board is a new feature that will appear in print and online (www.cutis.com). Each month a member of the Cutis Editorial Board will provide pearls relating to the practice needs of dermatologists. Future topics will include:
- Electronic Medical Record Implementation
- Injection Technique With Fillers
- Psoriasis Treatment in Pregnancy
- Technology to Aid in Melanoma Diagnosis
- Plus more
Looking for pearls on a specific topic? The Editorial Board welcomes your feedback on potential topics. Send an e-mail to the Editorial Office ([email protected]) with your suggestions.
What does your patient need to know at the first visit?
Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history of melanoma and nonmelanoma skin cancer; skin examination for photodamage and photoaging as well as number and type of pigmented lesions; and lifestyle history, which should include location of residence as well as occupation and recreational pursuits. This discussion should, as usual, include questions about general health, systemic and skin disease, and medication usage, with particular focus on photoaggravated diseases such as lupus and melasma as well as ongoing use of topical agents and systemic photosensitizers. These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer and other specific conditions that alter the advice you would give.
What are your go-to treatments? Is your recommendation anecdotal or evidence based? What are the side effects?
I always recommend that my patients use a product that they like, which may sound simplistic. But if the patient doesn’t like the feel and look of the sunscreen, he/she won’t use it. Patients routinely should use a sunscreen with a sun protection factor (SPF) of 30 or higher that also carries a “broad spectrum” label. At the beach or during sweaty sports, patients should use one with a water-resistant SPF.
I prefer spray sunscreens for application on the back if the patient is alone without someone to help apply sunscreen to hard-to-reach areas and for male scalps. But you never know how much spray to use, so use a lot!
If patients are at the beach, playing sports, or watching sports outside, then they should reapply sunscreen every 2 hours. If patients work indoors and use a facial sunscreen in the morning, that’s sufficient.
Although there is no evidence that sunscreens are harmful for children older than 6 months of age and pregnant women, if patients in these special populations have concerns, I recommend using agents with inorganic compounds (physical blockers) such as titanium dioxide and zinc oxide only. Children are best protected with clothing and hats.
The evidence supports this approach. Patients really don’t need SPF 30 protection, but no one uses the amount of product that will result in the SPF listed on the bottle. So if patients use an SPF 30 or greater, they will get at least an SPF 15, which is sufficient everywhere but at the equator. Using SPF 30 the way we all apply it will give SPF 15–level protection.
There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma.
The biggest side effect is not using the sunscreen. Others include irritation and allergy. Irritation is common, but finding a product to use without irritation should be easy. Allergy is rarer, and when it occurs, it is usually due to the preservative or fragrance, not the active ingredients. If allergy does occur, patch testing by a dermatologist is necessary to determine the allergen.
Although it is still controversial, wearing sunscreens religiously can lead to vitamin D insufficiency or deficiency, which is particularly true for individuals with skin of color—Fitzpatrick skin types IV, V, and VI—and those cancer patients who adhere to rigorous photoprotection. These patients should be encouraged to take supplemental vitamin D3 and I suggest 2000 IU; this recommendation is my opinion and is not evidence based.
As to the literature in the laypress about hormonal changes from benzophenone, cancer from retinoids, and nanoparticle toxicity: There is no evidence to support those claims.
How do you keep patients compliant with treatment?
Keep telling them, and then tell them again.
What do you do if they refuse treatment?
Tell them to see someone else.
What resources do you recommend to patients for more information?
Consult the American Academy of Dermatology Web site (www.aad.org) and the Skin Cancer Foundation (www.skincancer.org).
Editorial Note
Practical Pearls From the Cutis® Board is a new feature that will appear in print and online (www.cutis.com). Each month a member of the Cutis Editorial Board will provide pearls relating to the practice needs of dermatologists. Future topics will include:
- Electronic Medical Record Implementation
- Injection Technique With Fillers
- Psoriasis Treatment in Pregnancy
- Technology to Aid in Melanoma Diagnosis
- Plus more
Looking for pearls on a specific topic? The Editorial Board welcomes your feedback on potential topics. Send an e-mail to the Editorial Office ([email protected]) with your suggestions.
What does your patient need to know at the first visit?
Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history of melanoma and nonmelanoma skin cancer; skin examination for photodamage and photoaging as well as number and type of pigmented lesions; and lifestyle history, which should include location of residence as well as occupation and recreational pursuits. This discussion should, as usual, include questions about general health, systemic and skin disease, and medication usage, with particular focus on photoaggravated diseases such as lupus and melasma as well as ongoing use of topical agents and systemic photosensitizers. These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer and other specific conditions that alter the advice you would give.
What are your go-to treatments? Is your recommendation anecdotal or evidence based? What are the side effects?
I always recommend that my patients use a product that they like, which may sound simplistic. But if the patient doesn’t like the feel and look of the sunscreen, he/she won’t use it. Patients routinely should use a sunscreen with a sun protection factor (SPF) of 30 or higher that also carries a “broad spectrum” label. At the beach or during sweaty sports, patients should use one with a water-resistant SPF.
I prefer spray sunscreens for application on the back if the patient is alone without someone to help apply sunscreen to hard-to-reach areas and for male scalps. But you never know how much spray to use, so use a lot!
If patients are at the beach, playing sports, or watching sports outside, then they should reapply sunscreen every 2 hours. If patients work indoors and use a facial sunscreen in the morning, that’s sufficient.
Although there is no evidence that sunscreens are harmful for children older than 6 months of age and pregnant women, if patients in these special populations have concerns, I recommend using agents with inorganic compounds (physical blockers) such as titanium dioxide and zinc oxide only. Children are best protected with clothing and hats.
The evidence supports this approach. Patients really don’t need SPF 30 protection, but no one uses the amount of product that will result in the SPF listed on the bottle. So if patients use an SPF 30 or greater, they will get at least an SPF 15, which is sufficient everywhere but at the equator. Using SPF 30 the way we all apply it will give SPF 15–level protection.
There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma.
The biggest side effect is not using the sunscreen. Others include irritation and allergy. Irritation is common, but finding a product to use without irritation should be easy. Allergy is rarer, and when it occurs, it is usually due to the preservative or fragrance, not the active ingredients. If allergy does occur, patch testing by a dermatologist is necessary to determine the allergen.
Although it is still controversial, wearing sunscreens religiously can lead to vitamin D insufficiency or deficiency, which is particularly true for individuals with skin of color—Fitzpatrick skin types IV, V, and VI—and those cancer patients who adhere to rigorous photoprotection. These patients should be encouraged to take supplemental vitamin D3 and I suggest 2000 IU; this recommendation is my opinion and is not evidence based.
As to the literature in the laypress about hormonal changes from benzophenone, cancer from retinoids, and nanoparticle toxicity: There is no evidence to support those claims.
How do you keep patients compliant with treatment?
Keep telling them, and then tell them again.
What do you do if they refuse treatment?
Tell them to see someone else.
What resources do you recommend to patients for more information?
Consult the American Academy of Dermatology Web site (www.aad.org) and the Skin Cancer Foundation (www.skincancer.org).
Editorial Note
Practical Pearls From the Cutis® Board is a new feature that will appear in print and online (www.cutis.com). Each month a member of the Cutis Editorial Board will provide pearls relating to the practice needs of dermatologists. Future topics will include:
- Electronic Medical Record Implementation
- Injection Technique With Fillers
- Psoriasis Treatment in Pregnancy
- Technology to Aid in Melanoma Diagnosis
- Plus more
Looking for pearls on a specific topic? The Editorial Board welcomes your feedback on potential topics. Send an e-mail to the Editorial Office ([email protected]) with your suggestions.
Three Cheers for B3?
At the recent American Society of Clinical Oncology Annual Meeting, Martin et al presented data from the Australian Oral Nicotinamide to Reduce Actinic Cancer (ONTRAC) study. This prospective double-blind, randomized, controlled trial examined 386 immunocompetent patients with 2 or more nonmelanoma skin cancers (NMSCs) in the last 5 years (average, 8). The patients were randomized to receive oral nicotinamide 500 mg twice daily or placebo for 1 year, resulting in significant reduction of new NMSCs (average rate 1.77 vs 2.42; relative rate reduction, 23%; P=.02), with similar results for basal and squamous cell carcinomas. Actinic keratosis counts were reduced throughout the year by up to 20%, peaking at 9 months. No differences in adverse events were noted between the treatment and placebo groups.
What’s the issue?
High-risk NMSC patients present a challenge to dermatologists, as their need for constant surveillance, field therapy for actinic keratoses, and revolving visits between skin examinations and procedural modalities such as Mohs micrographic surgery can be staggering. Chemopreventive strategies pose difficulties, especially for elderly patients, due to tolerability and adherence, skin irritation and cosmetic limitations of topical therapies such as 5-fluorouracil, and inadequacy or financial inaccessibility of oral therapies such as acitretin.
Nicotinamide is a confusing supplement, as it is also called niacinamide. One of the 2 forms of vitamin B3, nicotinic acid (or niacin) is the other form and can be converted to nicotinamide in the body. It has cholesterol and vasodilatory/flushing effects that nicotinamide itself does not. Therefore, these supplement subtypes are not generally interchangeable.
Nicotinamide is postulated to enhance DNA repair and reverse UV immunosuppression in NMSC patients and is a well-tolerated and inexpensive supplement (approximately $10 a month for the dosage in this study). Although the decrease in skin cancer number per year seems modest in this study, my patients would likely welcome at least 1 fewer surgery per year and much less cryotherapy or 5-fluorouracil cream, especially if it is as simple as buying the supplement at the grocery store as they do for their fish oil capsules and probiotics. Does vitamin B3 hold promise for your high-risk NMSC patients?
At the recent American Society of Clinical Oncology Annual Meeting, Martin et al presented data from the Australian Oral Nicotinamide to Reduce Actinic Cancer (ONTRAC) study. This prospective double-blind, randomized, controlled trial examined 386 immunocompetent patients with 2 or more nonmelanoma skin cancers (NMSCs) in the last 5 years (average, 8). The patients were randomized to receive oral nicotinamide 500 mg twice daily or placebo for 1 year, resulting in significant reduction of new NMSCs (average rate 1.77 vs 2.42; relative rate reduction, 23%; P=.02), with similar results for basal and squamous cell carcinomas. Actinic keratosis counts were reduced throughout the year by up to 20%, peaking at 9 months. No differences in adverse events were noted between the treatment and placebo groups.
What’s the issue?
High-risk NMSC patients present a challenge to dermatologists, as their need for constant surveillance, field therapy for actinic keratoses, and revolving visits between skin examinations and procedural modalities such as Mohs micrographic surgery can be staggering. Chemopreventive strategies pose difficulties, especially for elderly patients, due to tolerability and adherence, skin irritation and cosmetic limitations of topical therapies such as 5-fluorouracil, and inadequacy or financial inaccessibility of oral therapies such as acitretin.
Nicotinamide is a confusing supplement, as it is also called niacinamide. One of the 2 forms of vitamin B3, nicotinic acid (or niacin) is the other form and can be converted to nicotinamide in the body. It has cholesterol and vasodilatory/flushing effects that nicotinamide itself does not. Therefore, these supplement subtypes are not generally interchangeable.
Nicotinamide is postulated to enhance DNA repair and reverse UV immunosuppression in NMSC patients and is a well-tolerated and inexpensive supplement (approximately $10 a month for the dosage in this study). Although the decrease in skin cancer number per year seems modest in this study, my patients would likely welcome at least 1 fewer surgery per year and much less cryotherapy or 5-fluorouracil cream, especially if it is as simple as buying the supplement at the grocery store as they do for their fish oil capsules and probiotics. Does vitamin B3 hold promise for your high-risk NMSC patients?
At the recent American Society of Clinical Oncology Annual Meeting, Martin et al presented data from the Australian Oral Nicotinamide to Reduce Actinic Cancer (ONTRAC) study. This prospective double-blind, randomized, controlled trial examined 386 immunocompetent patients with 2 or more nonmelanoma skin cancers (NMSCs) in the last 5 years (average, 8). The patients were randomized to receive oral nicotinamide 500 mg twice daily or placebo for 1 year, resulting in significant reduction of new NMSCs (average rate 1.77 vs 2.42; relative rate reduction, 23%; P=.02), with similar results for basal and squamous cell carcinomas. Actinic keratosis counts were reduced throughout the year by up to 20%, peaking at 9 months. No differences in adverse events were noted between the treatment and placebo groups.
What’s the issue?
High-risk NMSC patients present a challenge to dermatologists, as their need for constant surveillance, field therapy for actinic keratoses, and revolving visits between skin examinations and procedural modalities such as Mohs micrographic surgery can be staggering. Chemopreventive strategies pose difficulties, especially for elderly patients, due to tolerability and adherence, skin irritation and cosmetic limitations of topical therapies such as 5-fluorouracil, and inadequacy or financial inaccessibility of oral therapies such as acitretin.
Nicotinamide is a confusing supplement, as it is also called niacinamide. One of the 2 forms of vitamin B3, nicotinic acid (or niacin) is the other form and can be converted to nicotinamide in the body. It has cholesterol and vasodilatory/flushing effects that nicotinamide itself does not. Therefore, these supplement subtypes are not generally interchangeable.
Nicotinamide is postulated to enhance DNA repair and reverse UV immunosuppression in NMSC patients and is a well-tolerated and inexpensive supplement (approximately $10 a month for the dosage in this study). Although the decrease in skin cancer number per year seems modest in this study, my patients would likely welcome at least 1 fewer surgery per year and much less cryotherapy or 5-fluorouracil cream, especially if it is as simple as buying the supplement at the grocery store as they do for their fish oil capsules and probiotics. Does vitamin B3 hold promise for your high-risk NMSC patients?
Fake Melanoma and Nonmelanoma Skin Cancer “Cures”
Skin cancer patients should beware of products available online that fraudulently claim to prevent and cure cancer, including melanoma and nonmelanoma skin cancers, according to the US Food and Drug Administration (FDA). These products often are marketed as natural treatments or dietary supplements. They have not gained FDA approval and therefore are not proven to be safe or effective. Rather, they can cause more harm to patients and delay the effects of conventional cancer treatments.
Firms that illegally market fraudulent cancer treatments often use exaggerated unsubstantiated claims to promote their products. The FDA has provided consumer health information with several phrases that consumers should recognize as warning signs for fraudulent cancer treatments:
- “Scientific breakthrough”
- “Miraculous cure”
- “Ancient remedy”
- “Treats all forms of cancer”
- “Skin cancers disappear”
- “Shrinks malignant tumors”
- “Nontoxic”
- “Doesn’t make you sick”
- “Avoid painful surgery, radiotherapy, chemotherapy, or other conventional treatments”
- “Treat nonmelanoma skin cancers easily and safely”
- “Target cancer cells while leaving healthy cells alone”
Undocumented case histories or personal testimonials from patients or physicians claiming amazing results; suggestions that a product can treat serious or incurable diseases; and promises of no-risk, money-back guarantees also are signs of health fraud.
The FDA has cited black salves as one of the fake cancer remedies that have proven to be harmful. In a June 2015 Cutis article “Black Salve and Bloodroot Extract in Dermatologic Conditions,” Hou and Brewer reported an increased popularity of self-treatment with black salves in curing skin cancers and healing other skin conditions due to extensive advertising of its effectiveness. According to the FDA, black salves are sold with false promises that they will cure melanoma and nonmelanoma skin cancers by “drawing out” the disease from beneath the skin. However, Hou and Brewer warned that some black salves contain escharotics such as zinc chloride and bloodroot, which could cause damage to healthy tissue.
“Despite the information and testimonials that are widely available on the Internet, black salve use has not been validated by rigorous studies,” the authors reported. “[It] is not regulated by the US Food and Drug Administration, resulting in poor quality control and inconsistent user instructions.”
Dermatologists should be aware that skin cancer patients may be attracted to alternative treatments such as black salves. Health care professionals should educate patients about fraudulent cancer treatments versus investigational treatments.
For a complete list of fake cancer cures consumers should avoid, consult the FDA.
Skin cancer patients should beware of products available online that fraudulently claim to prevent and cure cancer, including melanoma and nonmelanoma skin cancers, according to the US Food and Drug Administration (FDA). These products often are marketed as natural treatments or dietary supplements. They have not gained FDA approval and therefore are not proven to be safe or effective. Rather, they can cause more harm to patients and delay the effects of conventional cancer treatments.
Firms that illegally market fraudulent cancer treatments often use exaggerated unsubstantiated claims to promote their products. The FDA has provided consumer health information with several phrases that consumers should recognize as warning signs for fraudulent cancer treatments:
- “Scientific breakthrough”
- “Miraculous cure”
- “Ancient remedy”
- “Treats all forms of cancer”
- “Skin cancers disappear”
- “Shrinks malignant tumors”
- “Nontoxic”
- “Doesn’t make you sick”
- “Avoid painful surgery, radiotherapy, chemotherapy, or other conventional treatments”
- “Treat nonmelanoma skin cancers easily and safely”
- “Target cancer cells while leaving healthy cells alone”
Undocumented case histories or personal testimonials from patients or physicians claiming amazing results; suggestions that a product can treat serious or incurable diseases; and promises of no-risk, money-back guarantees also are signs of health fraud.
The FDA has cited black salves as one of the fake cancer remedies that have proven to be harmful. In a June 2015 Cutis article “Black Salve and Bloodroot Extract in Dermatologic Conditions,” Hou and Brewer reported an increased popularity of self-treatment with black salves in curing skin cancers and healing other skin conditions due to extensive advertising of its effectiveness. According to the FDA, black salves are sold with false promises that they will cure melanoma and nonmelanoma skin cancers by “drawing out” the disease from beneath the skin. However, Hou and Brewer warned that some black salves contain escharotics such as zinc chloride and bloodroot, which could cause damage to healthy tissue.
“Despite the information and testimonials that are widely available on the Internet, black salve use has not been validated by rigorous studies,” the authors reported. “[It] is not regulated by the US Food and Drug Administration, resulting in poor quality control and inconsistent user instructions.”
Dermatologists should be aware that skin cancer patients may be attracted to alternative treatments such as black salves. Health care professionals should educate patients about fraudulent cancer treatments versus investigational treatments.
For a complete list of fake cancer cures consumers should avoid, consult the FDA.
Skin cancer patients should beware of products available online that fraudulently claim to prevent and cure cancer, including melanoma and nonmelanoma skin cancers, according to the US Food and Drug Administration (FDA). These products often are marketed as natural treatments or dietary supplements. They have not gained FDA approval and therefore are not proven to be safe or effective. Rather, they can cause more harm to patients and delay the effects of conventional cancer treatments.
Firms that illegally market fraudulent cancer treatments often use exaggerated unsubstantiated claims to promote their products. The FDA has provided consumer health information with several phrases that consumers should recognize as warning signs for fraudulent cancer treatments:
- “Scientific breakthrough”
- “Miraculous cure”
- “Ancient remedy”
- “Treats all forms of cancer”
- “Skin cancers disappear”
- “Shrinks malignant tumors”
- “Nontoxic”
- “Doesn’t make you sick”
- “Avoid painful surgery, radiotherapy, chemotherapy, or other conventional treatments”
- “Treat nonmelanoma skin cancers easily and safely”
- “Target cancer cells while leaving healthy cells alone”
Undocumented case histories or personal testimonials from patients or physicians claiming amazing results; suggestions that a product can treat serious or incurable diseases; and promises of no-risk, money-back guarantees also are signs of health fraud.
The FDA has cited black salves as one of the fake cancer remedies that have proven to be harmful. In a June 2015 Cutis article “Black Salve and Bloodroot Extract in Dermatologic Conditions,” Hou and Brewer reported an increased popularity of self-treatment with black salves in curing skin cancers and healing other skin conditions due to extensive advertising of its effectiveness. According to the FDA, black salves are sold with false promises that they will cure melanoma and nonmelanoma skin cancers by “drawing out” the disease from beneath the skin. However, Hou and Brewer warned that some black salves contain escharotics such as zinc chloride and bloodroot, which could cause damage to healthy tissue.
“Despite the information and testimonials that are widely available on the Internet, black salve use has not been validated by rigorous studies,” the authors reported. “[It] is not regulated by the US Food and Drug Administration, resulting in poor quality control and inconsistent user instructions.”
Dermatologists should be aware that skin cancer patients may be attracted to alternative treatments such as black salves. Health care professionals should educate patients about fraudulent cancer treatments versus investigational treatments.
For a complete list of fake cancer cures consumers should avoid, consult the FDA.
Prevalence and Impact of Health-Related Internet and Smartphone Use Among Dermatology Patients
Patients increasingly use the Internet and/or smartphone applications (apps) to seek health information and track personal health data,1,2 typically in the spirit of being a more educated consumer. However, many patients use the Internet in an attempt to self-diagnose and independently find treatment options, thus avoiding (in their opinion) the need to seek in-person medical care. Additionally, electronic access to health information has expanded beyond computers to smartphones with apps that can provide users with a simple interface to personalize the health information they seek and receive.
Prior studies have shown that seeking online health information and health-related social media is more common among women, younger patients, those with a college education, and those with a higher income.3,4 However, the prevalence of health-related Internet and smartphone use among dermatology patients as well as how patients ultimately use this information is not well studied. This information about patient behavior is important because of the potential harm that may come from patient self-diagnosis, which may delay or prevent treatment, as well as the benefits of patient self-education, which may expedite diagnosis and treatment.5 We surveyed a heterogeneous patient population at 2 dermatology offices in a major academic medical center to assess the prevalence and predictors of Internet and smartphone use to obtain both general medical and dermatologic information among dermatology patients. We also evaluated the impact that health information obtained from online sources has on a patient’s degree of concern about cutaneous disease and the likelihood of seeing a dermatologist for a skin problem.
Methods
Survey and Participants
This study was approved by the institutional review board at the University of Pittsburgh, Pennsylvania. All patients aged 18 years or older who presented to the department of dermatology at 2 offices of the University of Pittsburgh Medical Center from September 2013 through July 2014 were invited to participate in an anonymous 33-question survey regarding their use of the Internet and smartphone apps to obtain health information and make health care decisions. Patients were asked to complete the survey prior to seeing a health care provider and return it to a locked box by the front desk before leaving the office. Survey questions were designed by physicians with content expertise (J.A.W. and L.K.F.) and were reviewed by a statistician with survey expertise (D.G.W.). The survey included questions about patient demographics, Internet and smartphone use (both general and health related), and specific sources accessed. The survey also inquired about the impact of health information obtained via the Internet and smartphone apps on respondents’ degree of worry about a hypothetical skin condition or lesion using a 5-point Likert scale (1=no worry; 5=very worried). Respondents also were asked which skin conditions they previously researched online and whether their findings impacted their decision to see a dermatologist. Additionally, respondents were asked to list the smartphone apps and other online health resources they had used within the last 3 months. Prior to distribution, the survey was piloted with 10 participants and no issues with comprehensibility were noted.
Statistical Analysis
We described demographic traits (eg, age, sex, race/ethnicity, level of education, income) and factors associated with access to health care (eg, specialist co-pay, travel time from dermatology office) of respondents using proportions. We evaluated respondents’ access to and use of Internet- and smartphone-based health information using proportions and used χ² tests to quantify differences by sex and age (<50 years and ≥50 years).
We analyzed the impact of Internet and smartphone-based health information on patient worry about skin conditions by obtaining median worry on a 5-point Likert scale. Due to the nonparametric nature of the data, we used the Mann-Whitney U test to quantify differences by sex and age (<50 and ≥50 years). We used multiple logistic regression to identify factors associated with 3 outcomes: (1) using the Internet to self-diagnose a dermatologic disease, (2) using the Internet to obtain dermatology-related information within the last 3 months, (3) and previously refraining from visiting a dermatologist based on reassurance from online resources. Predictors included the aforementioned demographic and health-care access–related traits. We also categorized smartphone apps used by respondents (ie, fitness/nutrition, reference, self-help, health monitoring, diagnostic aids, electronic medical record) and calculated the proportion of respondents with 1 or more of each type of app on their smartphones. Analyses were conducted in Stata 13.1 and IBM SPSS 22.0.
Results
Of 1000 patients who were invited to participate in the study, a total of 775 respondents completed the survey, yielding a response rate of 77.5%. The majority of respondents were aged 30 to 60 years (mean age [standard deviation], 44.5 [17.2] years; median age [interquartile range], 44 [29–59] years), female (66.7%), and non-Hispanic white (83.3%)(Table 1). The majority of respondents (88.8%) had completed at least some college. Nearly all respondents had medical insurance (97.8%), but annual household income and insurance co-pay varied considerably. Only 10.8% of respondents traveled more than an hour to our offices.
The majority of respondents had access to home Internet and owned a smartphone (Table 2). Use of the Internet to obtain health-related information in the 3 months prior to presentation was more common among females (77.9% vs 70.1%; P=.03) and respondents younger than 50 years (83.4% vs 62.5%; P<.001); the same was true for dermatology-related infor-mation (females: 43.2% vs 31.0%; P=.003; aged <50 years, 51.6% vs 22.2%; P<.001). The majority of respondents indicated that they use the Internet to obtain health-related information both before and after they see their doctor. Most respondents indicated that they sometimes discuss health-related information found on the Internet with a physician. Smartphone use to obtain health-related information was more common among respondents younger than 50 years versus those who were 50 years or older (55.5% vs 24.1%; P<.001), as was smartphone use to diagnose skin problems (20.0% vs 6.3%; P<.001).
In multivariable analysis, use of the Internet or a smartphone to obtain health-related information was associated with younger age (<50 years) and a higher level of education (both P<.001). Use of the Internet to obtain dermatology-related information (P<.001) and use of a smartphone to help diagnose a skin problem (P=.001) was associated with younger age (<50 years) only. Income, sex, co-pay to see a dermatologist, and travel time to the dermatology office were not associated with use of online resources for general or dermatology-specific health-related information or assistance with diagnosing a skin problem.
Of 204 respondents who indicated that they previously attempted to self-diagnose a skin condition using the Internet, the most commonly researched condition was skin cancer/moles/unknown spots (64.7%), followed by rashes (40.7%), acne (20.6%), cosmetic issues (16.2%), psoriasis (12.7%), dermatitis (3.4%), warts (1.5%), tick bites (1.0%), and lupus (1.0%)(some respondents selected more than one condition). Only 7.0% of respondents indicated that they previously had refrained from visiting a dermatologist based on reassurance from online resources. Compared to the rest of the surveyed population, these respondents were younger (P=.001), but there were no significant differences in sex, highest level of education, household income, or travel time to the dermatology office. The most commonly researched condition among these respondents was acne (12 respondents), and 11 respondents indicated that they had attempted to self-diagnose a mole or potential cancer using online sources.
Of 557 respondents who owned a smartphone, 31.8% reported using at least 1 health-related app (mean number of health apps per respondent, 1.5). Of the apps that respondents used, 45.9% focused on fitness/nutrition, 28.7% provided reference information, 13.4% were a patient portal for receiving information from their electronic medical record, 8.6% provided a health monitoring function, 1.9% served as a diagnostic aid, and 1.5% provided coping assistance and emotional support for individuals with cognitive or emotional conditions; only 1 respondent reported using an app related to dermatology.
All respondents were asked to rate their anticipated degree of worry if the Internet or a smartphone app suggested that a skin lesion was benign versus dangerous on a 5-point scale. Overall, the median worry rating increased from 3 to 5 when information accessed via the Internet or a smartphone app suggested a lesion was dangerous rather than benign. A change in worry of 2 or more points was seen in 36.1% of females and 49.1% of males (P=.002) when information obtained via the Internet indicated a lesion was dangerous and in 47.5% of females and 58.8% of males (P=.006) when a smartphone app indicated that a lesion was dangerous. When information obtained via the Internet indicated a lesion was dangerous, a change in worry of 2 or more points was seen in 41.8% of respondents who were younger than 50 years and in 41.1% of those who were 50 years or older (P=.93). When a smartphone app indicated a lesion was dangerous, a change in worry of 2 or more points was seen in 50.2% of respondents who were younger than 50 years and in 52.2% of those who were 50 years or older (P=.61).
Discussion
In this cross-sectional study, we found that health-related Internet and smartphone use among dermatology patients is common and may impact both patients’ degree of concern about a skin lesion as well as the likelihood of seeking in-person medical care if they are reassured by the results of their online findings. Age and level of education were associated with Internet and smartphone use to obtain dermatology-related health information but not factors related to health care access. More patients used the Internet or a smartphone to obtain general medical information versus dermatology-related information. Respondents who indicated that they used the Internet to obtain health-related information tended to do so before visiting their physician.
Our finding that a patient’s level of worry about a hypothetical skin condition or lesion is influenced by health information obtained via the Internet or a smartphone app is concerning. One study found that participants who used a popular search engine to look for information about vaccine safety and dangers were directed to Web sites with inaccurate information more than 50% of the time, and 65% of the information they obtained from these sites was false.6 In our study, approximately 25% of respondents had previously consulted online resources to attempt toself-diagnose a skin condition. Online sources about dermatologic conditions were consulted most frequently for information about potential skin cancers, moles, and unknown spots. A prior study showed that smartphone apps that claim to aid patients in determining whether a skin lesion is low or high risk for melanoma often are inaccurate and are associated with a high rate of missed melanomas.5 Even though we surveyed patients who did end up seeing a dermatologist, some respondents had previously opted out of seeing a dermatologist based on information they had found online. Because our study was conducted among patients who chose to seek care at a dermatology office, the problem is likely greater than estimated from our findings because we had no way of reaching individuals who decided to completely forgo a visit with a dermatologist.
Although use of the Internet to obtain health-related information was common among older adults in our population, it was nearly universal in younger adults. Health-related smartphone use was more than twice as common in younger versus older adults, which could be due to an increased comfort with technology and its integration into daily life. The fact that age and education were associated with Internet use for dermatology-related health information but not household income or travel time to the dermatology office suggests that information seeking is not due to lack of resources limiting access to dermatologic care but rather to the greater role that rapid access to online information plays in patients’ lives. Our findings are similar to another study that examined the use of online sources for general health information.7
This study has several limitations. First, there may have been some selection bias. We specifically aimed to understand the health-related Internet and smartphone use among dermatology patients, thus restricting our sample to this population. By doing so, we were unable to assess the use of such resources by the general population, particularly those individuals who chose not to see a dermatologist at all based on their own online research. Our findings may not apply to other practices and regions of the country, as we implemented our study in one geographic location and in offices of an academic practice. Although our sample size and diversity with regard to income, education, and age suggest that our results are likely generalizable to many settings, it is important to note that nearly all respondents in this study had health insurance and our findings are thus not necessarily applicable to those individuals who are uninsured.
Conclusion
Our findings suggest that the availability of online health information regarding dermatologic conditions provides dermatologists with both opportunities and challenges. Many patients consult online resources for health information, and the popularity of this practice is likely to increase with time, particularly as newer smartphones with features designed to allow users to monitor their health are developed with health-conscious consumers in mind. Most large health care systems provide patients with resources to view laboratory results and communicate with physicians online. It is important for dermatologists to be involved in the development of high-quality online content that educates the public while also emphasizing the need to seek in-person medical care, particularly in potential cases of skin cancer. It also is important for patients to be involved in the content development process to ensure that the messages they take away from online resources are the ones physicians wish to convey. Ideally, online forms of education will increase patients’ sense of self-efficacy while encouraging appropriate consultation for potentially harmful skin conditions.
1. Atkinson NL, Saperstein SL, Pleis J. Using the Internet for health-related activities: findings from a national probability sample. J Med Internet Res. 2009;11:e4.
2. Ybarra M, Suman M. Reasons, assessments and actions taken: sex and age differences in uses of Internet health information. Health Educ Res. 2008;23:512-521.
3. Bhandari N, Shi Y, Jung K. Seeking health information online: does limited healthcare access matter? J Am Med Inform Assoc. 2014;21:1113-1117.
4. Thackeray R, Crookston BT, West JH. Correlates of health-related social media use among adults. J Med Internet Res. 2013;15:e21.
5. Wolf JA, Moreau JF, Akilov O, et al. Diagnostic inaccuracy of smartphone applications for melanoma detection. JAMA Dermatol. 2013;149:422-426.
6. Kortum P, Edwards C, Richards-Kortum R. The impact of inaccurate Internet health information in a secondary school learning environment. J Med Internet Res. 2008;10:e17.
7. Mead N, Varnam R, Rogers A, et al. What predicts patients’ interest in the internet as a health resource in primary care in England? J Health Serv Res Policy. 2003;8:33-39.
Patients increasingly use the Internet and/or smartphone applications (apps) to seek health information and track personal health data,1,2 typically in the spirit of being a more educated consumer. However, many patients use the Internet in an attempt to self-diagnose and independently find treatment options, thus avoiding (in their opinion) the need to seek in-person medical care. Additionally, electronic access to health information has expanded beyond computers to smartphones with apps that can provide users with a simple interface to personalize the health information they seek and receive.
Prior studies have shown that seeking online health information and health-related social media is more common among women, younger patients, those with a college education, and those with a higher income.3,4 However, the prevalence of health-related Internet and smartphone use among dermatology patients as well as how patients ultimately use this information is not well studied. This information about patient behavior is important because of the potential harm that may come from patient self-diagnosis, which may delay or prevent treatment, as well as the benefits of patient self-education, which may expedite diagnosis and treatment.5 We surveyed a heterogeneous patient population at 2 dermatology offices in a major academic medical center to assess the prevalence and predictors of Internet and smartphone use to obtain both general medical and dermatologic information among dermatology patients. We also evaluated the impact that health information obtained from online sources has on a patient’s degree of concern about cutaneous disease and the likelihood of seeing a dermatologist for a skin problem.
Methods
Survey and Participants
This study was approved by the institutional review board at the University of Pittsburgh, Pennsylvania. All patients aged 18 years or older who presented to the department of dermatology at 2 offices of the University of Pittsburgh Medical Center from September 2013 through July 2014 were invited to participate in an anonymous 33-question survey regarding their use of the Internet and smartphone apps to obtain health information and make health care decisions. Patients were asked to complete the survey prior to seeing a health care provider and return it to a locked box by the front desk before leaving the office. Survey questions were designed by physicians with content expertise (J.A.W. and L.K.F.) and were reviewed by a statistician with survey expertise (D.G.W.). The survey included questions about patient demographics, Internet and smartphone use (both general and health related), and specific sources accessed. The survey also inquired about the impact of health information obtained via the Internet and smartphone apps on respondents’ degree of worry about a hypothetical skin condition or lesion using a 5-point Likert scale (1=no worry; 5=very worried). Respondents also were asked which skin conditions they previously researched online and whether their findings impacted their decision to see a dermatologist. Additionally, respondents were asked to list the smartphone apps and other online health resources they had used within the last 3 months. Prior to distribution, the survey was piloted with 10 participants and no issues with comprehensibility were noted.
Statistical Analysis
We described demographic traits (eg, age, sex, race/ethnicity, level of education, income) and factors associated with access to health care (eg, specialist co-pay, travel time from dermatology office) of respondents using proportions. We evaluated respondents’ access to and use of Internet- and smartphone-based health information using proportions and used χ² tests to quantify differences by sex and age (<50 years and ≥50 years).
We analyzed the impact of Internet and smartphone-based health information on patient worry about skin conditions by obtaining median worry on a 5-point Likert scale. Due to the nonparametric nature of the data, we used the Mann-Whitney U test to quantify differences by sex and age (<50 and ≥50 years). We used multiple logistic regression to identify factors associated with 3 outcomes: (1) using the Internet to self-diagnose a dermatologic disease, (2) using the Internet to obtain dermatology-related information within the last 3 months, (3) and previously refraining from visiting a dermatologist based on reassurance from online resources. Predictors included the aforementioned demographic and health-care access–related traits. We also categorized smartphone apps used by respondents (ie, fitness/nutrition, reference, self-help, health monitoring, diagnostic aids, electronic medical record) and calculated the proportion of respondents with 1 or more of each type of app on their smartphones. Analyses were conducted in Stata 13.1 and IBM SPSS 22.0.
Results
Of 1000 patients who were invited to participate in the study, a total of 775 respondents completed the survey, yielding a response rate of 77.5%. The majority of respondents were aged 30 to 60 years (mean age [standard deviation], 44.5 [17.2] years; median age [interquartile range], 44 [29–59] years), female (66.7%), and non-Hispanic white (83.3%)(Table 1). The majority of respondents (88.8%) had completed at least some college. Nearly all respondents had medical insurance (97.8%), but annual household income and insurance co-pay varied considerably. Only 10.8% of respondents traveled more than an hour to our offices.
The majority of respondents had access to home Internet and owned a smartphone (Table 2). Use of the Internet to obtain health-related information in the 3 months prior to presentation was more common among females (77.9% vs 70.1%; P=.03) and respondents younger than 50 years (83.4% vs 62.5%; P<.001); the same was true for dermatology-related infor-mation (females: 43.2% vs 31.0%; P=.003; aged <50 years, 51.6% vs 22.2%; P<.001). The majority of respondents indicated that they use the Internet to obtain health-related information both before and after they see their doctor. Most respondents indicated that they sometimes discuss health-related information found on the Internet with a physician. Smartphone use to obtain health-related information was more common among respondents younger than 50 years versus those who were 50 years or older (55.5% vs 24.1%; P<.001), as was smartphone use to diagnose skin problems (20.0% vs 6.3%; P<.001).
In multivariable analysis, use of the Internet or a smartphone to obtain health-related information was associated with younger age (<50 years) and a higher level of education (both P<.001). Use of the Internet to obtain dermatology-related information (P<.001) and use of a smartphone to help diagnose a skin problem (P=.001) was associated with younger age (<50 years) only. Income, sex, co-pay to see a dermatologist, and travel time to the dermatology office were not associated with use of online resources for general or dermatology-specific health-related information or assistance with diagnosing a skin problem.
Of 204 respondents who indicated that they previously attempted to self-diagnose a skin condition using the Internet, the most commonly researched condition was skin cancer/moles/unknown spots (64.7%), followed by rashes (40.7%), acne (20.6%), cosmetic issues (16.2%), psoriasis (12.7%), dermatitis (3.4%), warts (1.5%), tick bites (1.0%), and lupus (1.0%)(some respondents selected more than one condition). Only 7.0% of respondents indicated that they previously had refrained from visiting a dermatologist based on reassurance from online resources. Compared to the rest of the surveyed population, these respondents were younger (P=.001), but there were no significant differences in sex, highest level of education, household income, or travel time to the dermatology office. The most commonly researched condition among these respondents was acne (12 respondents), and 11 respondents indicated that they had attempted to self-diagnose a mole or potential cancer using online sources.
Of 557 respondents who owned a smartphone, 31.8% reported using at least 1 health-related app (mean number of health apps per respondent, 1.5). Of the apps that respondents used, 45.9% focused on fitness/nutrition, 28.7% provided reference information, 13.4% were a patient portal for receiving information from their electronic medical record, 8.6% provided a health monitoring function, 1.9% served as a diagnostic aid, and 1.5% provided coping assistance and emotional support for individuals with cognitive or emotional conditions; only 1 respondent reported using an app related to dermatology.
All respondents were asked to rate their anticipated degree of worry if the Internet or a smartphone app suggested that a skin lesion was benign versus dangerous on a 5-point scale. Overall, the median worry rating increased from 3 to 5 when information accessed via the Internet or a smartphone app suggested a lesion was dangerous rather than benign. A change in worry of 2 or more points was seen in 36.1% of females and 49.1% of males (P=.002) when information obtained via the Internet indicated a lesion was dangerous and in 47.5% of females and 58.8% of males (P=.006) when a smartphone app indicated that a lesion was dangerous. When information obtained via the Internet indicated a lesion was dangerous, a change in worry of 2 or more points was seen in 41.8% of respondents who were younger than 50 years and in 41.1% of those who were 50 years or older (P=.93). When a smartphone app indicated a lesion was dangerous, a change in worry of 2 or more points was seen in 50.2% of respondents who were younger than 50 years and in 52.2% of those who were 50 years or older (P=.61).
Discussion
In this cross-sectional study, we found that health-related Internet and smartphone use among dermatology patients is common and may impact both patients’ degree of concern about a skin lesion as well as the likelihood of seeking in-person medical care if they are reassured by the results of their online findings. Age and level of education were associated with Internet and smartphone use to obtain dermatology-related health information but not factors related to health care access. More patients used the Internet or a smartphone to obtain general medical information versus dermatology-related information. Respondents who indicated that they used the Internet to obtain health-related information tended to do so before visiting their physician.
Our finding that a patient’s level of worry about a hypothetical skin condition or lesion is influenced by health information obtained via the Internet or a smartphone app is concerning. One study found that participants who used a popular search engine to look for information about vaccine safety and dangers were directed to Web sites with inaccurate information more than 50% of the time, and 65% of the information they obtained from these sites was false.6 In our study, approximately 25% of respondents had previously consulted online resources to attempt toself-diagnose a skin condition. Online sources about dermatologic conditions were consulted most frequently for information about potential skin cancers, moles, and unknown spots. A prior study showed that smartphone apps that claim to aid patients in determining whether a skin lesion is low or high risk for melanoma often are inaccurate and are associated with a high rate of missed melanomas.5 Even though we surveyed patients who did end up seeing a dermatologist, some respondents had previously opted out of seeing a dermatologist based on information they had found online. Because our study was conducted among patients who chose to seek care at a dermatology office, the problem is likely greater than estimated from our findings because we had no way of reaching individuals who decided to completely forgo a visit with a dermatologist.
Although use of the Internet to obtain health-related information was common among older adults in our population, it was nearly universal in younger adults. Health-related smartphone use was more than twice as common in younger versus older adults, which could be due to an increased comfort with technology and its integration into daily life. The fact that age and education were associated with Internet use for dermatology-related health information but not household income or travel time to the dermatology office suggests that information seeking is not due to lack of resources limiting access to dermatologic care but rather to the greater role that rapid access to online information plays in patients’ lives. Our findings are similar to another study that examined the use of online sources for general health information.7
This study has several limitations. First, there may have been some selection bias. We specifically aimed to understand the health-related Internet and smartphone use among dermatology patients, thus restricting our sample to this population. By doing so, we were unable to assess the use of such resources by the general population, particularly those individuals who chose not to see a dermatologist at all based on their own online research. Our findings may not apply to other practices and regions of the country, as we implemented our study in one geographic location and in offices of an academic practice. Although our sample size and diversity with regard to income, education, and age suggest that our results are likely generalizable to many settings, it is important to note that nearly all respondents in this study had health insurance and our findings are thus not necessarily applicable to those individuals who are uninsured.
Conclusion
Our findings suggest that the availability of online health information regarding dermatologic conditions provides dermatologists with both opportunities and challenges. Many patients consult online resources for health information, and the popularity of this practice is likely to increase with time, particularly as newer smartphones with features designed to allow users to monitor their health are developed with health-conscious consumers in mind. Most large health care systems provide patients with resources to view laboratory results and communicate with physicians online. It is important for dermatologists to be involved in the development of high-quality online content that educates the public while also emphasizing the need to seek in-person medical care, particularly in potential cases of skin cancer. It also is important for patients to be involved in the content development process to ensure that the messages they take away from online resources are the ones physicians wish to convey. Ideally, online forms of education will increase patients’ sense of self-efficacy while encouraging appropriate consultation for potentially harmful skin conditions.
Patients increasingly use the Internet and/or smartphone applications (apps) to seek health information and track personal health data,1,2 typically in the spirit of being a more educated consumer. However, many patients use the Internet in an attempt to self-diagnose and independently find treatment options, thus avoiding (in their opinion) the need to seek in-person medical care. Additionally, electronic access to health information has expanded beyond computers to smartphones with apps that can provide users with a simple interface to personalize the health information they seek and receive.
Prior studies have shown that seeking online health information and health-related social media is more common among women, younger patients, those with a college education, and those with a higher income.3,4 However, the prevalence of health-related Internet and smartphone use among dermatology patients as well as how patients ultimately use this information is not well studied. This information about patient behavior is important because of the potential harm that may come from patient self-diagnosis, which may delay or prevent treatment, as well as the benefits of patient self-education, which may expedite diagnosis and treatment.5 We surveyed a heterogeneous patient population at 2 dermatology offices in a major academic medical center to assess the prevalence and predictors of Internet and smartphone use to obtain both general medical and dermatologic information among dermatology patients. We also evaluated the impact that health information obtained from online sources has on a patient’s degree of concern about cutaneous disease and the likelihood of seeing a dermatologist for a skin problem.
Methods
Survey and Participants
This study was approved by the institutional review board at the University of Pittsburgh, Pennsylvania. All patients aged 18 years or older who presented to the department of dermatology at 2 offices of the University of Pittsburgh Medical Center from September 2013 through July 2014 were invited to participate in an anonymous 33-question survey regarding their use of the Internet and smartphone apps to obtain health information and make health care decisions. Patients were asked to complete the survey prior to seeing a health care provider and return it to a locked box by the front desk before leaving the office. Survey questions were designed by physicians with content expertise (J.A.W. and L.K.F.) and were reviewed by a statistician with survey expertise (D.G.W.). The survey included questions about patient demographics, Internet and smartphone use (both general and health related), and specific sources accessed. The survey also inquired about the impact of health information obtained via the Internet and smartphone apps on respondents’ degree of worry about a hypothetical skin condition or lesion using a 5-point Likert scale (1=no worry; 5=very worried). Respondents also were asked which skin conditions they previously researched online and whether their findings impacted their decision to see a dermatologist. Additionally, respondents were asked to list the smartphone apps and other online health resources they had used within the last 3 months. Prior to distribution, the survey was piloted with 10 participants and no issues with comprehensibility were noted.
Statistical Analysis
We described demographic traits (eg, age, sex, race/ethnicity, level of education, income) and factors associated with access to health care (eg, specialist co-pay, travel time from dermatology office) of respondents using proportions. We evaluated respondents’ access to and use of Internet- and smartphone-based health information using proportions and used χ² tests to quantify differences by sex and age (<50 years and ≥50 years).
We analyzed the impact of Internet and smartphone-based health information on patient worry about skin conditions by obtaining median worry on a 5-point Likert scale. Due to the nonparametric nature of the data, we used the Mann-Whitney U test to quantify differences by sex and age (<50 and ≥50 years). We used multiple logistic regression to identify factors associated with 3 outcomes: (1) using the Internet to self-diagnose a dermatologic disease, (2) using the Internet to obtain dermatology-related information within the last 3 months, (3) and previously refraining from visiting a dermatologist based on reassurance from online resources. Predictors included the aforementioned demographic and health-care access–related traits. We also categorized smartphone apps used by respondents (ie, fitness/nutrition, reference, self-help, health monitoring, diagnostic aids, electronic medical record) and calculated the proportion of respondents with 1 or more of each type of app on their smartphones. Analyses were conducted in Stata 13.1 and IBM SPSS 22.0.
Results
Of 1000 patients who were invited to participate in the study, a total of 775 respondents completed the survey, yielding a response rate of 77.5%. The majority of respondents were aged 30 to 60 years (mean age [standard deviation], 44.5 [17.2] years; median age [interquartile range], 44 [29–59] years), female (66.7%), and non-Hispanic white (83.3%)(Table 1). The majority of respondents (88.8%) had completed at least some college. Nearly all respondents had medical insurance (97.8%), but annual household income and insurance co-pay varied considerably. Only 10.8% of respondents traveled more than an hour to our offices.
The majority of respondents had access to home Internet and owned a smartphone (Table 2). Use of the Internet to obtain health-related information in the 3 months prior to presentation was more common among females (77.9% vs 70.1%; P=.03) and respondents younger than 50 years (83.4% vs 62.5%; P<.001); the same was true for dermatology-related infor-mation (females: 43.2% vs 31.0%; P=.003; aged <50 years, 51.6% vs 22.2%; P<.001). The majority of respondents indicated that they use the Internet to obtain health-related information both before and after they see their doctor. Most respondents indicated that they sometimes discuss health-related information found on the Internet with a physician. Smartphone use to obtain health-related information was more common among respondents younger than 50 years versus those who were 50 years or older (55.5% vs 24.1%; P<.001), as was smartphone use to diagnose skin problems (20.0% vs 6.3%; P<.001).
In multivariable analysis, use of the Internet or a smartphone to obtain health-related information was associated with younger age (<50 years) and a higher level of education (both P<.001). Use of the Internet to obtain dermatology-related information (P<.001) and use of a smartphone to help diagnose a skin problem (P=.001) was associated with younger age (<50 years) only. Income, sex, co-pay to see a dermatologist, and travel time to the dermatology office were not associated with use of online resources for general or dermatology-specific health-related information or assistance with diagnosing a skin problem.
Of 204 respondents who indicated that they previously attempted to self-diagnose a skin condition using the Internet, the most commonly researched condition was skin cancer/moles/unknown spots (64.7%), followed by rashes (40.7%), acne (20.6%), cosmetic issues (16.2%), psoriasis (12.7%), dermatitis (3.4%), warts (1.5%), tick bites (1.0%), and lupus (1.0%)(some respondents selected more than one condition). Only 7.0% of respondents indicated that they previously had refrained from visiting a dermatologist based on reassurance from online resources. Compared to the rest of the surveyed population, these respondents were younger (P=.001), but there were no significant differences in sex, highest level of education, household income, or travel time to the dermatology office. The most commonly researched condition among these respondents was acne (12 respondents), and 11 respondents indicated that they had attempted to self-diagnose a mole or potential cancer using online sources.
Of 557 respondents who owned a smartphone, 31.8% reported using at least 1 health-related app (mean number of health apps per respondent, 1.5). Of the apps that respondents used, 45.9% focused on fitness/nutrition, 28.7% provided reference information, 13.4% were a patient portal for receiving information from their electronic medical record, 8.6% provided a health monitoring function, 1.9% served as a diagnostic aid, and 1.5% provided coping assistance and emotional support for individuals with cognitive or emotional conditions; only 1 respondent reported using an app related to dermatology.
All respondents were asked to rate their anticipated degree of worry if the Internet or a smartphone app suggested that a skin lesion was benign versus dangerous on a 5-point scale. Overall, the median worry rating increased from 3 to 5 when information accessed via the Internet or a smartphone app suggested a lesion was dangerous rather than benign. A change in worry of 2 or more points was seen in 36.1% of females and 49.1% of males (P=.002) when information obtained via the Internet indicated a lesion was dangerous and in 47.5% of females and 58.8% of males (P=.006) when a smartphone app indicated that a lesion was dangerous. When information obtained via the Internet indicated a lesion was dangerous, a change in worry of 2 or more points was seen in 41.8% of respondents who were younger than 50 years and in 41.1% of those who were 50 years or older (P=.93). When a smartphone app indicated a lesion was dangerous, a change in worry of 2 or more points was seen in 50.2% of respondents who were younger than 50 years and in 52.2% of those who were 50 years or older (P=.61).
Discussion
In this cross-sectional study, we found that health-related Internet and smartphone use among dermatology patients is common and may impact both patients’ degree of concern about a skin lesion as well as the likelihood of seeking in-person medical care if they are reassured by the results of their online findings. Age and level of education were associated with Internet and smartphone use to obtain dermatology-related health information but not factors related to health care access. More patients used the Internet or a smartphone to obtain general medical information versus dermatology-related information. Respondents who indicated that they used the Internet to obtain health-related information tended to do so before visiting their physician.
Our finding that a patient’s level of worry about a hypothetical skin condition or lesion is influenced by health information obtained via the Internet or a smartphone app is concerning. One study found that participants who used a popular search engine to look for information about vaccine safety and dangers were directed to Web sites with inaccurate information more than 50% of the time, and 65% of the information they obtained from these sites was false.6 In our study, approximately 25% of respondents had previously consulted online resources to attempt toself-diagnose a skin condition. Online sources about dermatologic conditions were consulted most frequently for information about potential skin cancers, moles, and unknown spots. A prior study showed that smartphone apps that claim to aid patients in determining whether a skin lesion is low or high risk for melanoma often are inaccurate and are associated with a high rate of missed melanomas.5 Even though we surveyed patients who did end up seeing a dermatologist, some respondents had previously opted out of seeing a dermatologist based on information they had found online. Because our study was conducted among patients who chose to seek care at a dermatology office, the problem is likely greater than estimated from our findings because we had no way of reaching individuals who decided to completely forgo a visit with a dermatologist.
Although use of the Internet to obtain health-related information was common among older adults in our population, it was nearly universal in younger adults. Health-related smartphone use was more than twice as common in younger versus older adults, which could be due to an increased comfort with technology and its integration into daily life. The fact that age and education were associated with Internet use for dermatology-related health information but not household income or travel time to the dermatology office suggests that information seeking is not due to lack of resources limiting access to dermatologic care but rather to the greater role that rapid access to online information plays in patients’ lives. Our findings are similar to another study that examined the use of online sources for general health information.7
This study has several limitations. First, there may have been some selection bias. We specifically aimed to understand the health-related Internet and smartphone use among dermatology patients, thus restricting our sample to this population. By doing so, we were unable to assess the use of such resources by the general population, particularly those individuals who chose not to see a dermatologist at all based on their own online research. Our findings may not apply to other practices and regions of the country, as we implemented our study in one geographic location and in offices of an academic practice. Although our sample size and diversity with regard to income, education, and age suggest that our results are likely generalizable to many settings, it is important to note that nearly all respondents in this study had health insurance and our findings are thus not necessarily applicable to those individuals who are uninsured.
Conclusion
Our findings suggest that the availability of online health information regarding dermatologic conditions provides dermatologists with both opportunities and challenges. Many patients consult online resources for health information, and the popularity of this practice is likely to increase with time, particularly as newer smartphones with features designed to allow users to monitor their health are developed with health-conscious consumers in mind. Most large health care systems provide patients with resources to view laboratory results and communicate with physicians online. It is important for dermatologists to be involved in the development of high-quality online content that educates the public while also emphasizing the need to seek in-person medical care, particularly in potential cases of skin cancer. It also is important for patients to be involved in the content development process to ensure that the messages they take away from online resources are the ones physicians wish to convey. Ideally, online forms of education will increase patients’ sense of self-efficacy while encouraging appropriate consultation for potentially harmful skin conditions.
1. Atkinson NL, Saperstein SL, Pleis J. Using the Internet for health-related activities: findings from a national probability sample. J Med Internet Res. 2009;11:e4.
2. Ybarra M, Suman M. Reasons, assessments and actions taken: sex and age differences in uses of Internet health information. Health Educ Res. 2008;23:512-521.
3. Bhandari N, Shi Y, Jung K. Seeking health information online: does limited healthcare access matter? J Am Med Inform Assoc. 2014;21:1113-1117.
4. Thackeray R, Crookston BT, West JH. Correlates of health-related social media use among adults. J Med Internet Res. 2013;15:e21.
5. Wolf JA, Moreau JF, Akilov O, et al. Diagnostic inaccuracy of smartphone applications for melanoma detection. JAMA Dermatol. 2013;149:422-426.
6. Kortum P, Edwards C, Richards-Kortum R. The impact of inaccurate Internet health information in a secondary school learning environment. J Med Internet Res. 2008;10:e17.
7. Mead N, Varnam R, Rogers A, et al. What predicts patients’ interest in the internet as a health resource in primary care in England? J Health Serv Res Policy. 2003;8:33-39.
1. Atkinson NL, Saperstein SL, Pleis J. Using the Internet for health-related activities: findings from a national probability sample. J Med Internet Res. 2009;11:e4.
2. Ybarra M, Suman M. Reasons, assessments and actions taken: sex and age differences in uses of Internet health information. Health Educ Res. 2008;23:512-521.
3. Bhandari N, Shi Y, Jung K. Seeking health information online: does limited healthcare access matter? J Am Med Inform Assoc. 2014;21:1113-1117.
4. Thackeray R, Crookston BT, West JH. Correlates of health-related social media use among adults. J Med Internet Res. 2013;15:e21.
5. Wolf JA, Moreau JF, Akilov O, et al. Diagnostic inaccuracy of smartphone applications for melanoma detection. JAMA Dermatol. 2013;149:422-426.
6. Kortum P, Edwards C, Richards-Kortum R. The impact of inaccurate Internet health information in a secondary school learning environment. J Med Internet Res. 2008;10:e17.
7. Mead N, Varnam R, Rogers A, et al. What predicts patients’ interest in the internet as a health resource in primary care in England? J Health Serv Res Policy. 2003;8:33-39.
Black Salve and Bloodroot Extract in Dermatologic Conditions
Black salve is composed of various ingredients, many of which are inert; however, some black salves contain escharotics, the 2 most common are zinc chloride and bloodroot (Sanguinaria canadensis) extract. In high doses, such as those contained in most black salve products, these corrosive agents can indiscriminately damage both healthy and diseased tissue.1 Nevertheless, many black salve products currently are advertised as safe and natural methods for curing skin cancer2-4 or treating a variety of other skin conditions (eg, moles, warts, skin tags, boils, abscesses, bee stings, other minor wounds)1,5 and even nondermatologic conditions such as a sore throat.6 Despite the information and testimonials that are widely available on the Internet, black salve use has not been validated by rigorous studies. Black salve is not regulated by the US Food and Drug Administration, resulting in poor quality control and inconsistent user instructions. We report the case of application of black salve to a biopsy site of a compound nevus with moderate atypia that resulted in the formation of a dermatitis plaque with subsequent scarring and basal layer pigmentation.
Case Report
A 35-year-old woman with a family history of melanoma presented for follow-up of a compound nevus with moderate atypia on the right anterior thigh that had been biopsied 6 months prior. Complete excision of the lesion was recommended at the initial presentation but was not performed due to scheduling conflicts. The patient reported applying black salve to the biopsy site and also to the left thigh 3 months later. There was no reaction on the left thigh after one 24-hour application of black salve, but an area around the biopsy site on the right thigh became thickened and irritated with superficial erosion of the skin following 2 applications of black salve, each of 24 hours’ duration. Physical examination revealed a granulomatous plaque at the biopsy site that was approximately 5 cm in diameter (Figure 1A). One year later the lesion had completely healed (Figure 1B) and a biopsy revealed scarring with basal layer pigmentation (Figure 2).
| 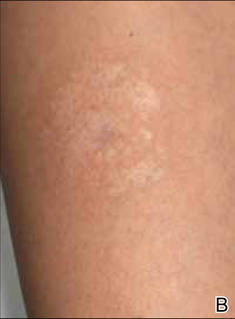
| 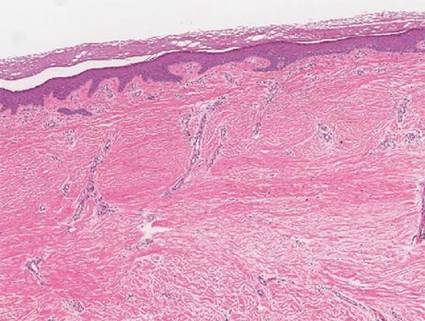
| |||
| Figure 1. A 5-cm granulomatous reaction surrounding a biopsy site on the right anterior thigh 3 months after application of black salve (A). One year later, the lesion had completely healed (B). | Figure 2. A biopsy one year following application of black salve demonstrated scarring with basal layer pigmentation (H&E, original magnification ×4). | ||||
Comment
A Web search using the term black salve yields a large number of products labeled as skin cancer salves, many showing glowing reviews and some being sold by major US retailers. The ingredients in black salves often vary in the innocuous substances they contain, but most products include the escharotics zinc chloride and bloodroot extract, which is derived from the plant S canadensis.1,3 For example, the ingredients of one popular black salve product include zinc chloride, chaparral (active ingredient is nordihydroguaiaretic acid), graviola leaf extract, oleander leaf extract, bloodroot extract, and glycerine,7 while another product includes bloodroot extract, zinc chloride, chaparral, cayenne pepper, red clover, birch bark, dimethyl sulfoxide, and burdock root.4
Bloodroot extract’s antimicrobial, anti-inflammatory, antioxidant, and immunomodulatory effects derive from its benzylisoquinoline alkaloids including sanguinarine, allocryptopine, berberine, coptisine, protopine, and stylopine.3,8 Bloodroot extract possesses some degree of tumoricidal potency, with one study finding that it selectively targets cancer cells.9 However, this differential response is seen only at low doses and not at the high concentrations contained in most black salve products.1 According to fluorometric assays, sanguinarine is not selective for tumor cells and therefore damages healthy tissue in addition to the unwanted lesions.6,10,11 The US Food and Drug Administration includes black salve products on its list of fake cancer cures that consumers should avoid.12 Reports of extensive damage from black salve use include skin ulceration2,10 and complete loss of a naris1 and nasal ala.5 Our case suggests the possible association between black salve use and an irritant reaction and erosion of the skin.
Furthermore, reliance on black salve alone in the treatment of skin cancer poses the threat of recurrence or metastasis of cancer because there is no way to know if the salve completely removed the cancer without a biopsy. Self-treatment can delay more effective therapy and may require further treatments.
Black salve should be subject to standarddrug regulations and its use discouraged by dermatologists due to the associated harmful effects and the availability of safer treatments. To better treat and inform their patients, dermatologists should be aware that patients may be attracted to alternative treatments such as black salves.
1. Eastman KL, McFarland LV, Raugi GJ. A review of topical corrosive black salve. J Altern Complement Med. 2014;20:284-289.
2. Eastman KL, McFarland LV, Raugi GJ. Buyer beware: a black salve caution. J Am Acad Dermatol. 2011;65:e154-e155.
3. Sivyer GW, Rosendahl C. Application of black salve to a thin melanoma that subsequently progressed to metastatic melanoma: a case study. Dermatol Pract Concept. 2014;4:77-80.
4. McDaniel S, Goldman GD. Consequences of using escharotic agents as primary treatment for nonmelanoma skin cancer. Arch Dermatol. 2002;138:1593-1596.
5. Payne CE. ‘Black Salve’ and melanomas [published online ahead of print August 11, 2010]. J Plast Reconstr Aesthet Surg. 2011;64:422.
6. Cienki JJ, Zaret L. An Internet misadventure: bloodroot salve toxicity. J Altern Complement Med. 2010;16:1125-1127.
7. Cansema and escharotics. Alpha Omega Labs Web site. http://www.altcancer.com/faqcan.htm. Accessed May 6, 2015.
8. Vlachojannis C, Magora F, Chrubasik S. Rise and fall of oral health products with Canadian bloodroot extract. Phytother Res. 2012;26:1423-1426.
9. Ahmad N, Gupta S, Husain MM, et al. Differential antiproliferative and apoptotic response of sanguinarine for cancer cells versus normal cells. Clin Cancer Res. 2000;6:1524-1528.
10. Saltzberg F, Barron G, Fenske N. Deforming self-treatment with herbal “black salve.” Dermatol Surg. 2009;35:1152-1154.
11. Debiton E, Madelmont JC, Legault J, et al. Sanguinarine-induced apoptosis is associated with an early and severe cellular glutathione depletion. Cancer Chemother Pharmacol. 2003;51:474-482.
12. 187 fake cancer “cures” consumers should avoid. US Food and Drug Administration Web site. http://www.fda.gov/Drugs/GuidanceCompliance RegulatoryInformation/EnforcementActivitiesbyFDA/ucm171057.htm. Updated July 9, 2009. Accessed May 6, 2015.
Black salve is composed of various ingredients, many of which are inert; however, some black salves contain escharotics, the 2 most common are zinc chloride and bloodroot (Sanguinaria canadensis) extract. In high doses, such as those contained in most black salve products, these corrosive agents can indiscriminately damage both healthy and diseased tissue.1 Nevertheless, many black salve products currently are advertised as safe and natural methods for curing skin cancer2-4 or treating a variety of other skin conditions (eg, moles, warts, skin tags, boils, abscesses, bee stings, other minor wounds)1,5 and even nondermatologic conditions such as a sore throat.6 Despite the information and testimonials that are widely available on the Internet, black salve use has not been validated by rigorous studies. Black salve is not regulated by the US Food and Drug Administration, resulting in poor quality control and inconsistent user instructions. We report the case of application of black salve to a biopsy site of a compound nevus with moderate atypia that resulted in the formation of a dermatitis plaque with subsequent scarring and basal layer pigmentation.
Case Report
A 35-year-old woman with a family history of melanoma presented for follow-up of a compound nevus with moderate atypia on the right anterior thigh that had been biopsied 6 months prior. Complete excision of the lesion was recommended at the initial presentation but was not performed due to scheduling conflicts. The patient reported applying black salve to the biopsy site and also to the left thigh 3 months later. There was no reaction on the left thigh after one 24-hour application of black salve, but an area around the biopsy site on the right thigh became thickened and irritated with superficial erosion of the skin following 2 applications of black salve, each of 24 hours’ duration. Physical examination revealed a granulomatous plaque at the biopsy site that was approximately 5 cm in diameter (Figure 1A). One year later the lesion had completely healed (Figure 1B) and a biopsy revealed scarring with basal layer pigmentation (Figure 2).

| 
| 
| |||
| Figure 1. A 5-cm granulomatous reaction surrounding a biopsy site on the right anterior thigh 3 months after application of black salve (A). One year later, the lesion had completely healed (B). | Figure 2. A biopsy one year following application of black salve demonstrated scarring with basal layer pigmentation (H&E, original magnification ×4). | ||||
Comment
A Web search using the term black salve yields a large number of products labeled as skin cancer salves, many showing glowing reviews and some being sold by major US retailers. The ingredients in black salves often vary in the innocuous substances they contain, but most products include the escharotics zinc chloride and bloodroot extract, which is derived from the plant S canadensis.1,3 For example, the ingredients of one popular black salve product include zinc chloride, chaparral (active ingredient is nordihydroguaiaretic acid), graviola leaf extract, oleander leaf extract, bloodroot extract, and glycerine,7 while another product includes bloodroot extract, zinc chloride, chaparral, cayenne pepper, red clover, birch bark, dimethyl sulfoxide, and burdock root.4
Bloodroot extract’s antimicrobial, anti-inflammatory, antioxidant, and immunomodulatory effects derive from its benzylisoquinoline alkaloids including sanguinarine, allocryptopine, berberine, coptisine, protopine, and stylopine.3,8 Bloodroot extract possesses some degree of tumoricidal potency, with one study finding that it selectively targets cancer cells.9 However, this differential response is seen only at low doses and not at the high concentrations contained in most black salve products.1 According to fluorometric assays, sanguinarine is not selective for tumor cells and therefore damages healthy tissue in addition to the unwanted lesions.6,10,11 The US Food and Drug Administration includes black salve products on its list of fake cancer cures that consumers should avoid.12 Reports of extensive damage from black salve use include skin ulceration2,10 and complete loss of a naris1 and nasal ala.5 Our case suggests the possible association between black salve use and an irritant reaction and erosion of the skin.
Furthermore, reliance on black salve alone in the treatment of skin cancer poses the threat of recurrence or metastasis of cancer because there is no way to know if the salve completely removed the cancer without a biopsy. Self-treatment can delay more effective therapy and may require further treatments.
Black salve should be subject to standarddrug regulations and its use discouraged by dermatologists due to the associated harmful effects and the availability of safer treatments. To better treat and inform their patients, dermatologists should be aware that patients may be attracted to alternative treatments such as black salves.
Black salve is composed of various ingredients, many of which are inert; however, some black salves contain escharotics, the 2 most common are zinc chloride and bloodroot (Sanguinaria canadensis) extract. In high doses, such as those contained in most black salve products, these corrosive agents can indiscriminately damage both healthy and diseased tissue.1 Nevertheless, many black salve products currently are advertised as safe and natural methods for curing skin cancer2-4 or treating a variety of other skin conditions (eg, moles, warts, skin tags, boils, abscesses, bee stings, other minor wounds)1,5 and even nondermatologic conditions such as a sore throat.6 Despite the information and testimonials that are widely available on the Internet, black salve use has not been validated by rigorous studies. Black salve is not regulated by the US Food and Drug Administration, resulting in poor quality control and inconsistent user instructions. We report the case of application of black salve to a biopsy site of a compound nevus with moderate atypia that resulted in the formation of a dermatitis plaque with subsequent scarring and basal layer pigmentation.
Case Report
A 35-year-old woman with a family history of melanoma presented for follow-up of a compound nevus with moderate atypia on the right anterior thigh that had been biopsied 6 months prior. Complete excision of the lesion was recommended at the initial presentation but was not performed due to scheduling conflicts. The patient reported applying black salve to the biopsy site and also to the left thigh 3 months later. There was no reaction on the left thigh after one 24-hour application of black salve, but an area around the biopsy site on the right thigh became thickened and irritated with superficial erosion of the skin following 2 applications of black salve, each of 24 hours’ duration. Physical examination revealed a granulomatous plaque at the biopsy site that was approximately 5 cm in diameter (Figure 1A). One year later the lesion had completely healed (Figure 1B) and a biopsy revealed scarring with basal layer pigmentation (Figure 2).

| 
| 
| |||
| Figure 1. A 5-cm granulomatous reaction surrounding a biopsy site on the right anterior thigh 3 months after application of black salve (A). One year later, the lesion had completely healed (B). | Figure 2. A biopsy one year following application of black salve demonstrated scarring with basal layer pigmentation (H&E, original magnification ×4). | ||||
Comment
A Web search using the term black salve yields a large number of products labeled as skin cancer salves, many showing glowing reviews and some being sold by major US retailers. The ingredients in black salves often vary in the innocuous substances they contain, but most products include the escharotics zinc chloride and bloodroot extract, which is derived from the plant S canadensis.1,3 For example, the ingredients of one popular black salve product include zinc chloride, chaparral (active ingredient is nordihydroguaiaretic acid), graviola leaf extract, oleander leaf extract, bloodroot extract, and glycerine,7 while another product includes bloodroot extract, zinc chloride, chaparral, cayenne pepper, red clover, birch bark, dimethyl sulfoxide, and burdock root.4
Bloodroot extract’s antimicrobial, anti-inflammatory, antioxidant, and immunomodulatory effects derive from its benzylisoquinoline alkaloids including sanguinarine, allocryptopine, berberine, coptisine, protopine, and stylopine.3,8 Bloodroot extract possesses some degree of tumoricidal potency, with one study finding that it selectively targets cancer cells.9 However, this differential response is seen only at low doses and not at the high concentrations contained in most black salve products.1 According to fluorometric assays, sanguinarine is not selective for tumor cells and therefore damages healthy tissue in addition to the unwanted lesions.6,10,11 The US Food and Drug Administration includes black salve products on its list of fake cancer cures that consumers should avoid.12 Reports of extensive damage from black salve use include skin ulceration2,10 and complete loss of a naris1 and nasal ala.5 Our case suggests the possible association between black salve use and an irritant reaction and erosion of the skin.
Furthermore, reliance on black salve alone in the treatment of skin cancer poses the threat of recurrence or metastasis of cancer because there is no way to know if the salve completely removed the cancer without a biopsy. Self-treatment can delay more effective therapy and may require further treatments.
Black salve should be subject to standarddrug regulations and its use discouraged by dermatologists due to the associated harmful effects and the availability of safer treatments. To better treat and inform their patients, dermatologists should be aware that patients may be attracted to alternative treatments such as black salves.
1. Eastman KL, McFarland LV, Raugi GJ. A review of topical corrosive black salve. J Altern Complement Med. 2014;20:284-289.
2. Eastman KL, McFarland LV, Raugi GJ. Buyer beware: a black salve caution. J Am Acad Dermatol. 2011;65:e154-e155.
3. Sivyer GW, Rosendahl C. Application of black salve to a thin melanoma that subsequently progressed to metastatic melanoma: a case study. Dermatol Pract Concept. 2014;4:77-80.
4. McDaniel S, Goldman GD. Consequences of using escharotic agents as primary treatment for nonmelanoma skin cancer. Arch Dermatol. 2002;138:1593-1596.
5. Payne CE. ‘Black Salve’ and melanomas [published online ahead of print August 11, 2010]. J Plast Reconstr Aesthet Surg. 2011;64:422.
6. Cienki JJ, Zaret L. An Internet misadventure: bloodroot salve toxicity. J Altern Complement Med. 2010;16:1125-1127.
7. Cansema and escharotics. Alpha Omega Labs Web site. http://www.altcancer.com/faqcan.htm. Accessed May 6, 2015.
8. Vlachojannis C, Magora F, Chrubasik S. Rise and fall of oral health products with Canadian bloodroot extract. Phytother Res. 2012;26:1423-1426.
9. Ahmad N, Gupta S, Husain MM, et al. Differential antiproliferative and apoptotic response of sanguinarine for cancer cells versus normal cells. Clin Cancer Res. 2000;6:1524-1528.
10. Saltzberg F, Barron G, Fenske N. Deforming self-treatment with herbal “black salve.” Dermatol Surg. 2009;35:1152-1154.
11. Debiton E, Madelmont JC, Legault J, et al. Sanguinarine-induced apoptosis is associated with an early and severe cellular glutathione depletion. Cancer Chemother Pharmacol. 2003;51:474-482.
12. 187 fake cancer “cures” consumers should avoid. US Food and Drug Administration Web site. http://www.fda.gov/Drugs/GuidanceCompliance RegulatoryInformation/EnforcementActivitiesbyFDA/ucm171057.htm. Updated July 9, 2009. Accessed May 6, 2015.
1. Eastman KL, McFarland LV, Raugi GJ. A review of topical corrosive black salve. J Altern Complement Med. 2014;20:284-289.
2. Eastman KL, McFarland LV, Raugi GJ. Buyer beware: a black salve caution. J Am Acad Dermatol. 2011;65:e154-e155.
3. Sivyer GW, Rosendahl C. Application of black salve to a thin melanoma that subsequently progressed to metastatic melanoma: a case study. Dermatol Pract Concept. 2014;4:77-80.
4. McDaniel S, Goldman GD. Consequences of using escharotic agents as primary treatment for nonmelanoma skin cancer. Arch Dermatol. 2002;138:1593-1596.
5. Payne CE. ‘Black Salve’ and melanomas [published online ahead of print August 11, 2010]. J Plast Reconstr Aesthet Surg. 2011;64:422.
6. Cienki JJ, Zaret L. An Internet misadventure: bloodroot salve toxicity. J Altern Complement Med. 2010;16:1125-1127.
7. Cansema and escharotics. Alpha Omega Labs Web site. http://www.altcancer.com/faqcan.htm. Accessed May 6, 2015.
8. Vlachojannis C, Magora F, Chrubasik S. Rise and fall of oral health products with Canadian bloodroot extract. Phytother Res. 2012;26:1423-1426.
9. Ahmad N, Gupta S, Husain MM, et al. Differential antiproliferative and apoptotic response of sanguinarine for cancer cells versus normal cells. Clin Cancer Res. 2000;6:1524-1528.
10. Saltzberg F, Barron G, Fenske N. Deforming self-treatment with herbal “black salve.” Dermatol Surg. 2009;35:1152-1154.
11. Debiton E, Madelmont JC, Legault J, et al. Sanguinarine-induced apoptosis is associated with an early and severe cellular glutathione depletion. Cancer Chemother Pharmacol. 2003;51:474-482.
12. 187 fake cancer “cures” consumers should avoid. US Food and Drug Administration Web site. http://www.fda.gov/Drugs/GuidanceCompliance RegulatoryInformation/EnforcementActivitiesbyFDA/ucm171057.htm. Updated July 9, 2009. Accessed May 6, 2015.
Practice Points
- Clinicians should be aware that black salve containing bloodroot extract is a popular alternative treatment used to cure a variety of skin ailments.
- Black salve containing bloodroot extract is not selective for tumor cells. Various case reports have shown that black salve can result in extensive tissue damage and recurrence or metastasis of skin cancer.
- Damage to healthy tissue can occur with as few as 2 applications of black salve.
Annual U.S. incidence of BCC pegged at 2 million
Approximately 2 million basal cell carcinomas occur in the United States each year, a number that has increased only modestly during the past 15 years, according to a report published online June 3 in JAMA Dermatology.
The epidemiology of BCCs has been difficult to pin down because these tumors are excluded from cancer registries and national cancer surveillance programs, and because when they are tracked they are usually lumped together with squamous cell carcinomas. The most recent National Cancer Institute–funded survey of BCC was done more than 30 years ago.
So the current incidence of BCC “is not well characterized.” Nevertheless. some researchers have posited that the incidence has risen 80%-200% during the past 10-20 years, said Dr. Maryam M. Asgari of Kaiser Permanente Northern California, Oakland, and the department of dermatology at the University of California, San Francisco.
To estimate incidence more accurately, Dr. Asgari and her associates analyzed data from every electronic pathology report in a registry covering the 3.2 million participants in the HMO from 1998 through 2012. They identified 221,624 cases of BCC in patients aged 2-105 years. The annual incidence increased from 513 to 600 cases per 100,000 population during the study period – a 17% rise that was deemed “not remarkable.”
“In extrapolating our data to the United States, we estimate that approximately 2 million individuals develop at least one BCC in the U.S. in a given year,” Dr. Asgari and her associates wrote (JAMA Dermatol. 2015 June 3 [doi:10.1001/jamadermatol.2015.1188]).
This total is higher than that cited on the National Institutes of Health website, which estimates the annual incidence of all nonmelanoma skin cancers, not just BCC, at 2 million per year.
Males were at higher risk than were females for developing BCC (incidence rate ratio, 1.65), and risk increased with increasing patient age. As expected, whites were at 8- to 70-fold greater risk than were Hispanics, Asians, or blacks. Contrary to one previous report, the incidence of BCC did not increase among Hispanics during the study period.
The National Cancer Institute and the National Institutes of Health supported the study. Dr. Asgari reported receiving research grants from Kaiser Permanente, Pfizer, and Valeant Pharmaceuticals, but not for this project.
Approximately 2 million basal cell carcinomas occur in the United States each year, a number that has increased only modestly during the past 15 years, according to a report published online June 3 in JAMA Dermatology.
The epidemiology of BCCs has been difficult to pin down because these tumors are excluded from cancer registries and national cancer surveillance programs, and because when they are tracked they are usually lumped together with squamous cell carcinomas. The most recent National Cancer Institute–funded survey of BCC was done more than 30 years ago.
So the current incidence of BCC “is not well characterized.” Nevertheless. some researchers have posited that the incidence has risen 80%-200% during the past 10-20 years, said Dr. Maryam M. Asgari of Kaiser Permanente Northern California, Oakland, and the department of dermatology at the University of California, San Francisco.
To estimate incidence more accurately, Dr. Asgari and her associates analyzed data from every electronic pathology report in a registry covering the 3.2 million participants in the HMO from 1998 through 2012. They identified 221,624 cases of BCC in patients aged 2-105 years. The annual incidence increased from 513 to 600 cases per 100,000 population during the study period – a 17% rise that was deemed “not remarkable.”
“In extrapolating our data to the United States, we estimate that approximately 2 million individuals develop at least one BCC in the U.S. in a given year,” Dr. Asgari and her associates wrote (JAMA Dermatol. 2015 June 3 [doi:10.1001/jamadermatol.2015.1188]).
This total is higher than that cited on the National Institutes of Health website, which estimates the annual incidence of all nonmelanoma skin cancers, not just BCC, at 2 million per year.
Males were at higher risk than were females for developing BCC (incidence rate ratio, 1.65), and risk increased with increasing patient age. As expected, whites were at 8- to 70-fold greater risk than were Hispanics, Asians, or blacks. Contrary to one previous report, the incidence of BCC did not increase among Hispanics during the study period.
The National Cancer Institute and the National Institutes of Health supported the study. Dr. Asgari reported receiving research grants from Kaiser Permanente, Pfizer, and Valeant Pharmaceuticals, but not for this project.
Approximately 2 million basal cell carcinomas occur in the United States each year, a number that has increased only modestly during the past 15 years, according to a report published online June 3 in JAMA Dermatology.
The epidemiology of BCCs has been difficult to pin down because these tumors are excluded from cancer registries and national cancer surveillance programs, and because when they are tracked they are usually lumped together with squamous cell carcinomas. The most recent National Cancer Institute–funded survey of BCC was done more than 30 years ago.
So the current incidence of BCC “is not well characterized.” Nevertheless. some researchers have posited that the incidence has risen 80%-200% during the past 10-20 years, said Dr. Maryam M. Asgari of Kaiser Permanente Northern California, Oakland, and the department of dermatology at the University of California, San Francisco.
To estimate incidence more accurately, Dr. Asgari and her associates analyzed data from every electronic pathology report in a registry covering the 3.2 million participants in the HMO from 1998 through 2012. They identified 221,624 cases of BCC in patients aged 2-105 years. The annual incidence increased from 513 to 600 cases per 100,000 population during the study period – a 17% rise that was deemed “not remarkable.”
“In extrapolating our data to the United States, we estimate that approximately 2 million individuals develop at least one BCC in the U.S. in a given year,” Dr. Asgari and her associates wrote (JAMA Dermatol. 2015 June 3 [doi:10.1001/jamadermatol.2015.1188]).
This total is higher than that cited on the National Institutes of Health website, which estimates the annual incidence of all nonmelanoma skin cancers, not just BCC, at 2 million per year.
Males were at higher risk than were females for developing BCC (incidence rate ratio, 1.65), and risk increased with increasing patient age. As expected, whites were at 8- to 70-fold greater risk than were Hispanics, Asians, or blacks. Contrary to one previous report, the incidence of BCC did not increase among Hispanics during the study period.
The National Cancer Institute and the National Institutes of Health supported the study. Dr. Asgari reported receiving research grants from Kaiser Permanente, Pfizer, and Valeant Pharmaceuticals, but not for this project.
Key clinical point: The annual incidence of basal cell carcinoma is estimated to be 2 million in the United States.
Major finding: The annual incidence of BCC increased from 513 to 600 cases per 100,000 population during the study period – a 17% rise that was deemed “not remarkable.”
Data source: A retrospective cohort study involving 147,093 patients with BCC treated at a northern California HMO during 1998-2012.
Disclosures: The National Cancer Institute and the National Institutes of Health supported the study. Dr. Asgari reported receiving research grants from Kaiser Permanente, Pfizer, and Valeant Pharmaceuticals, but not for this project.