User login
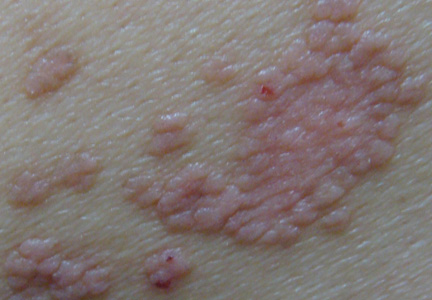
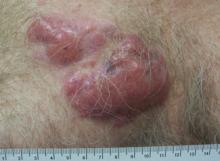
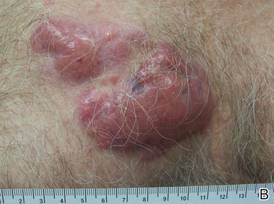
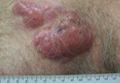
Multiple Firm Pink Papules and Nodules
The Diagnosis: Myeloid Leukemia Cutis
Leukemia cutis represents the infiltration of leukemic cells into the skin. It has been described in the setting of both myeloid and lymphoid leukemia. In the setting of acute myeloid leukemia, it has been reported to occur in 2% to 13% of patients overall,1,2 but it may occur in 31% of patients with the acute myelomonocytic or acute monocytic leukemia subtypes.3 Leukemia cutis is less common, with chronic myeloid leukemia occurring in 2.7% of patients in one study.4 In another study, 65% of patients with myeloid leukemia cutis had an acute myeloid leukemia.5
Myeloid leukemia cutis has been reported in patients aged 22 days to 90 years, with a median age of 62 years. There is a male predominance (1.4:1 ratio).5,6 The diagnosis of leukemia cutis is made concurrently with the diagnosis of leukemia in approximately 30% of cases, subsequent to the diagnosis of leukemia in approximately 60% of cases, and prior to the diagnosis of leukemia in approximately 10% of cases.5
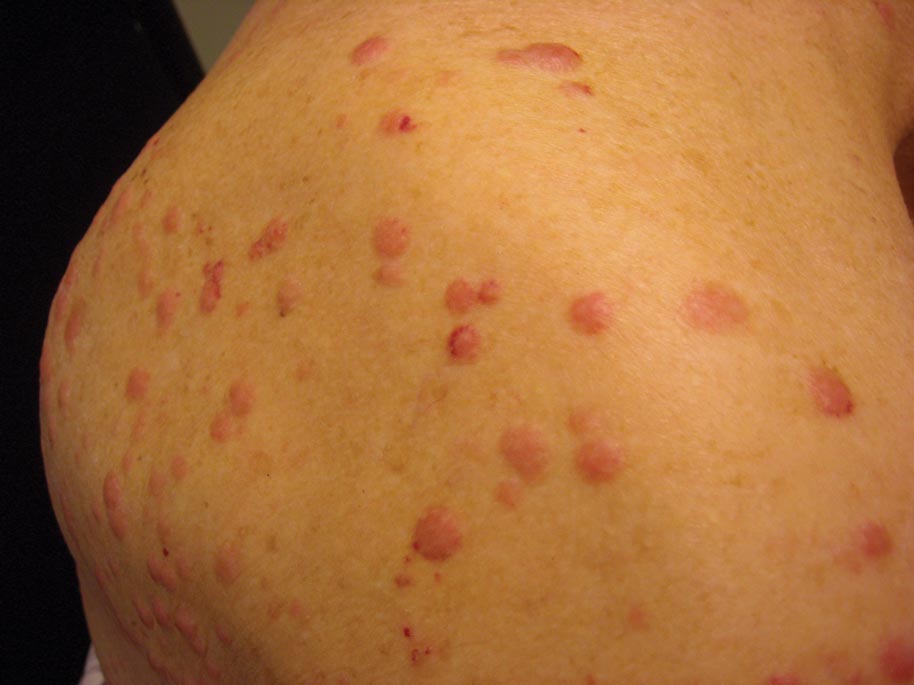
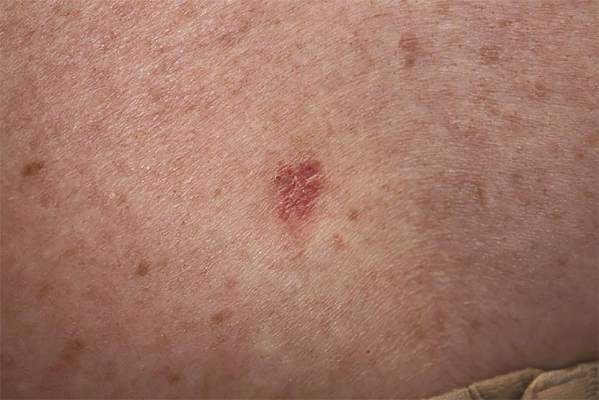
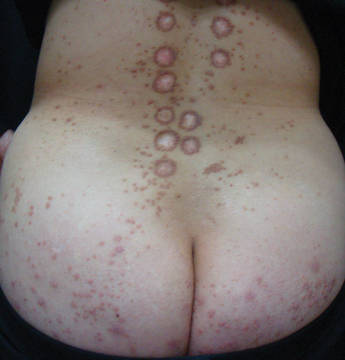
Clinically, myeloid leukemia cutis presents as an asymptomatic solitary lesion in 23% of cases or as multiple lesions in 77% of cases. Lesions consist of pink to red to violaceous papules, nodules, and macules that are occasionally purpuric and involve any cutaneous surface.5
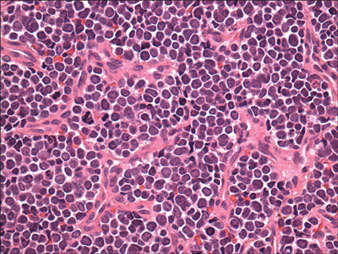
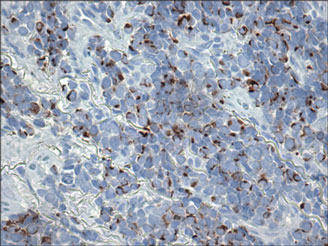
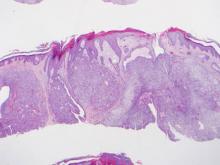
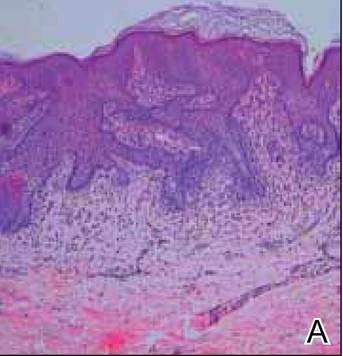
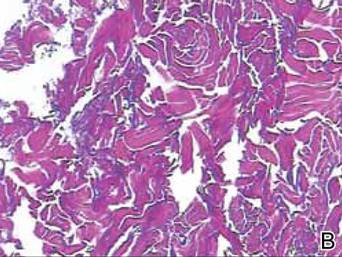
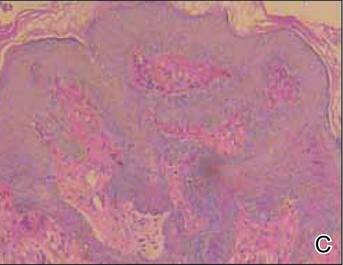
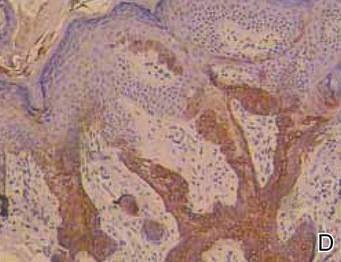
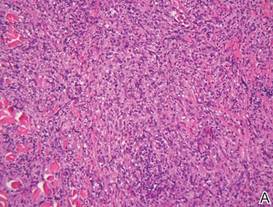
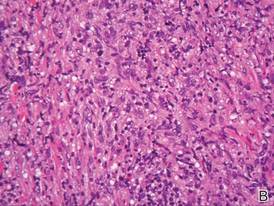
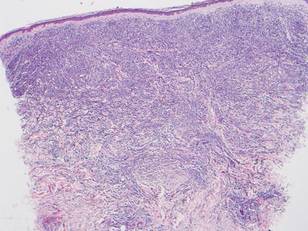
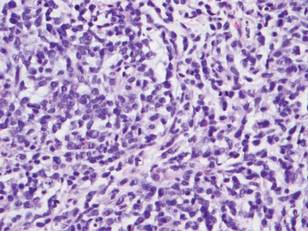
Histologically, the epidermis is unremarkable. Beneath a grenz zone within the dermis and usually extending into the subcutis there is a diffuse or nodular proliferation of neoplastic cells, often with perivascular and periadnexal accentuation and sometimes single filing of cells between collagen bundles (Figure 1). The cells are immature myeloid cells with irregular nuclear contours that may be indented or reniform (Figure 2). Nuclei contain finely dispersed chromatin with variably prominent nucleoli.5,6 Immunohistochemically, CD68 is positive in approximately 97% of cases, myeloperoxidase in 62%, and lysozyme in 85%. CD168, CD14, CD4, CD33, CD117, CD34, CD56, CD123, and CD303 are variably positive. CD3 and CD20, markers of lymphoid leukemia, are negative.5-8
Leukemia cutis in the setting of a myeloid leukemia portends a grave prognosis. In a series of 18 patients, 16 had additional extramedullary leukemia, including meningeal leukemia in 6 patients.2 Most patients with myeloid leukemia cutis die within an average of 1 to 8 months of diagnosis.9
- Boggs DR, Wintrobe MM, Cartwright GE. The acute leukemias. analysis of 322 cases and review of the literature. Medicine (Baltimore). 1962;41:163-225.
- Baer MR, Barcos M, Farrell H, et al. Acute myelogenous leukemia with leukemia cutis. eighteen cases seen between 1969 and 1986. Cancer. 1989;63:2192-2200.
- Straus DJ, Mertelsmann R, Koziner B, et al. The acute monocytic leukemias: multidisciplinary studies in 45 patients. Medicine (Baltimore). 1980;59:409-425.
- Rosenthal S, Canellos GP, DeVita VT Jr, et al. Characteristics of blast crisis in chronic granulocytic leukemia. Blood. 1977;49:705-714.
- Bénet C, Gomez A, Aguilar C, et al. Histologic and immunohistologic characterization of skin localization of myeloid disorders: a study of 173 cases. Am J Clin Pathol. 2011;135:278-290.
- Cronin DM, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009;132:101-110.
- Cho-Vega JH, Medeiros LJ, Prieto VG, et al. Leukemia cutis. Am J Clin Pathol. 2008;129:130-142.
- Kaddu S, Zenahlik P, Beham-Schmid C, et al. Specific cutaneous infiltrates in patients with myelogenous leukemia: a clinicopathologic study of 26 patients with assessment of diagnostic criteria. J Am Acad Dermatol. 1999;40:966-978.
- Su WP, Buechner SA, Li CY. Clinicopathologic correlations in leukemia cutis. J Am Acad Dermatol. 1984;11:121-128.
The Diagnosis: Myeloid Leukemia Cutis
Leukemia cutis represents the infiltration of leukemic cells into the skin. It has been described in the setting of both myeloid and lymphoid leukemia. In the setting of acute myeloid leukemia, it has been reported to occur in 2% to 13% of patients overall,1,2 but it may occur in 31% of patients with the acute myelomonocytic or acute monocytic leukemia subtypes.3 Leukemia cutis is less common, with chronic myeloid leukemia occurring in 2.7% of patients in one study.4 In another study, 65% of patients with myeloid leukemia cutis had an acute myeloid leukemia.5
Myeloid leukemia cutis has been reported in patients aged 22 days to 90 years, with a median age of 62 years. There is a male predominance (1.4:1 ratio).5,6 The diagnosis of leukemia cutis is made concurrently with the diagnosis of leukemia in approximately 30% of cases, subsequent to the diagnosis of leukemia in approximately 60% of cases, and prior to the diagnosis of leukemia in approximately 10% of cases.5
Clinically, myeloid leukemia cutis presents as an asymptomatic solitary lesion in 23% of cases or as multiple lesions in 77% of cases. Lesions consist of pink to red to violaceous papules, nodules, and macules that are occasionally purpuric and involve any cutaneous surface.5
Histologically, the epidermis is unremarkable. Beneath a grenz zone within the dermis and usually extending into the subcutis there is a diffuse or nodular proliferation of neoplastic cells, often with perivascular and periadnexal accentuation and sometimes single filing of cells between collagen bundles (Figure 1). The cells are immature myeloid cells with irregular nuclear contours that may be indented or reniform (Figure 2). Nuclei contain finely dispersed chromatin with variably prominent nucleoli.5,6 Immunohistochemically, CD68 is positive in approximately 97% of cases, myeloperoxidase in 62%, and lysozyme in 85%. CD168, CD14, CD4, CD33, CD117, CD34, CD56, CD123, and CD303 are variably positive. CD3 and CD20, markers of lymphoid leukemia, are negative.5-8


Leukemia cutis in the setting of a myeloid leukemia portends a grave prognosis. In a series of 18 patients, 16 had additional extramedullary leukemia, including meningeal leukemia in 6 patients.2 Most patients with myeloid leukemia cutis die within an average of 1 to 8 months of diagnosis.9
The Diagnosis: Myeloid Leukemia Cutis
Leukemia cutis represents the infiltration of leukemic cells into the skin. It has been described in the setting of both myeloid and lymphoid leukemia. In the setting of acute myeloid leukemia, it has been reported to occur in 2% to 13% of patients overall,1,2 but it may occur in 31% of patients with the acute myelomonocytic or acute monocytic leukemia subtypes.3 Leukemia cutis is less common, with chronic myeloid leukemia occurring in 2.7% of patients in one study.4 In another study, 65% of patients with myeloid leukemia cutis had an acute myeloid leukemia.5
Myeloid leukemia cutis has been reported in patients aged 22 days to 90 years, with a median age of 62 years. There is a male predominance (1.4:1 ratio).5,6 The diagnosis of leukemia cutis is made concurrently with the diagnosis of leukemia in approximately 30% of cases, subsequent to the diagnosis of leukemia in approximately 60% of cases, and prior to the diagnosis of leukemia in approximately 10% of cases.5
Clinically, myeloid leukemia cutis presents as an asymptomatic solitary lesion in 23% of cases or as multiple lesions in 77% of cases. Lesions consist of pink to red to violaceous papules, nodules, and macules that are occasionally purpuric and involve any cutaneous surface.5
Histologically, the epidermis is unremarkable. Beneath a grenz zone within the dermis and usually extending into the subcutis there is a diffuse or nodular proliferation of neoplastic cells, often with perivascular and periadnexal accentuation and sometimes single filing of cells between collagen bundles (Figure 1). The cells are immature myeloid cells with irregular nuclear contours that may be indented or reniform (Figure 2). Nuclei contain finely dispersed chromatin with variably prominent nucleoli.5,6 Immunohistochemically, CD68 is positive in approximately 97% of cases, myeloperoxidase in 62%, and lysozyme in 85%. CD168, CD14, CD4, CD33, CD117, CD34, CD56, CD123, and CD303 are variably positive. CD3 and CD20, markers of lymphoid leukemia, are negative.5-8


Leukemia cutis in the setting of a myeloid leukemia portends a grave prognosis. In a series of 18 patients, 16 had additional extramedullary leukemia, including meningeal leukemia in 6 patients.2 Most patients with myeloid leukemia cutis die within an average of 1 to 8 months of diagnosis.9
- Boggs DR, Wintrobe MM, Cartwright GE. The acute leukemias. analysis of 322 cases and review of the literature. Medicine (Baltimore). 1962;41:163-225.
- Baer MR, Barcos M, Farrell H, et al. Acute myelogenous leukemia with leukemia cutis. eighteen cases seen between 1969 and 1986. Cancer. 1989;63:2192-2200.
- Straus DJ, Mertelsmann R, Koziner B, et al. The acute monocytic leukemias: multidisciplinary studies in 45 patients. Medicine (Baltimore). 1980;59:409-425.
- Rosenthal S, Canellos GP, DeVita VT Jr, et al. Characteristics of blast crisis in chronic granulocytic leukemia. Blood. 1977;49:705-714.
- Bénet C, Gomez A, Aguilar C, et al. Histologic and immunohistologic characterization of skin localization of myeloid disorders: a study of 173 cases. Am J Clin Pathol. 2011;135:278-290.
- Cronin DM, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009;132:101-110.
- Cho-Vega JH, Medeiros LJ, Prieto VG, et al. Leukemia cutis. Am J Clin Pathol. 2008;129:130-142.
- Kaddu S, Zenahlik P, Beham-Schmid C, et al. Specific cutaneous infiltrates in patients with myelogenous leukemia: a clinicopathologic study of 26 patients with assessment of diagnostic criteria. J Am Acad Dermatol. 1999;40:966-978.
- Su WP, Buechner SA, Li CY. Clinicopathologic correlations in leukemia cutis. J Am Acad Dermatol. 1984;11:121-128.
- Boggs DR, Wintrobe MM, Cartwright GE. The acute leukemias. analysis of 322 cases and review of the literature. Medicine (Baltimore). 1962;41:163-225.
- Baer MR, Barcos M, Farrell H, et al. Acute myelogenous leukemia with leukemia cutis. eighteen cases seen between 1969 and 1986. Cancer. 1989;63:2192-2200.
- Straus DJ, Mertelsmann R, Koziner B, et al. The acute monocytic leukemias: multidisciplinary studies in 45 patients. Medicine (Baltimore). 1980;59:409-425.
- Rosenthal S, Canellos GP, DeVita VT Jr, et al. Characteristics of blast crisis in chronic granulocytic leukemia. Blood. 1977;49:705-714.
- Bénet C, Gomez A, Aguilar C, et al. Histologic and immunohistologic characterization of skin localization of myeloid disorders: a study of 173 cases. Am J Clin Pathol. 2011;135:278-290.
- Cronin DM, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009;132:101-110.
- Cho-Vega JH, Medeiros LJ, Prieto VG, et al. Leukemia cutis. Am J Clin Pathol. 2008;129:130-142.
- Kaddu S, Zenahlik P, Beham-Schmid C, et al. Specific cutaneous infiltrates in patients with myelogenous leukemia: a clinicopathologic study of 26 patients with assessment of diagnostic criteria. J Am Acad Dermatol. 1999;40:966-978.
- Su WP, Buechner SA, Li CY. Clinicopathologic correlations in leukemia cutis. J Am Acad Dermatol. 1984;11:121-128.
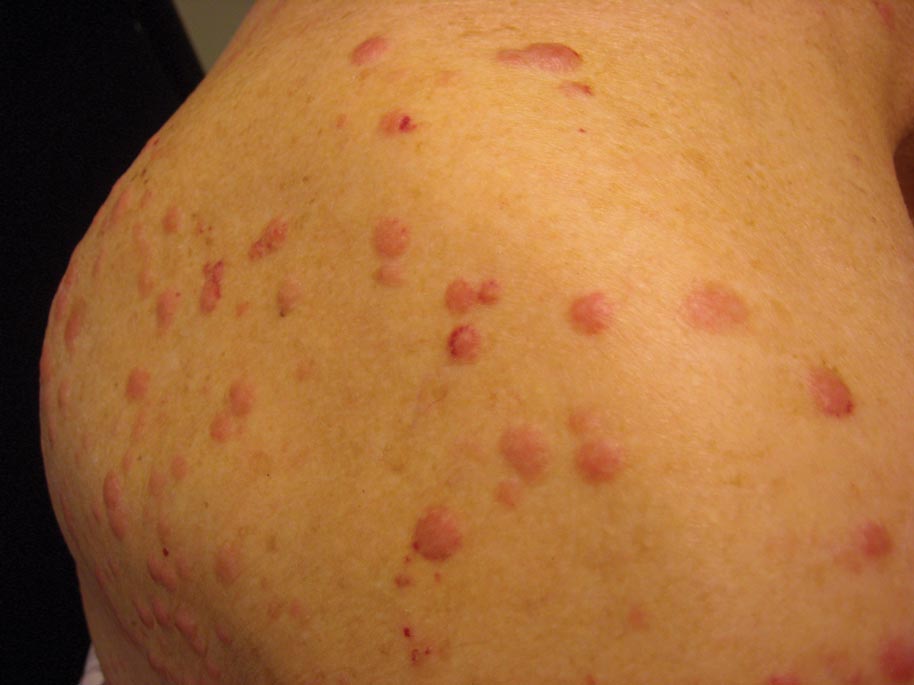
A 91-year-old man presented with numerous, scattered, asymptomatic, 3- to 9-mm, smooth, firm, pink papules and nodules involving the neck, trunk, and arms and legs of 1 week’s duration.
Erythematous Nodular Plaque Encircling the Lower Leg
The Diagnosis: Merkel Cell Carcinoma
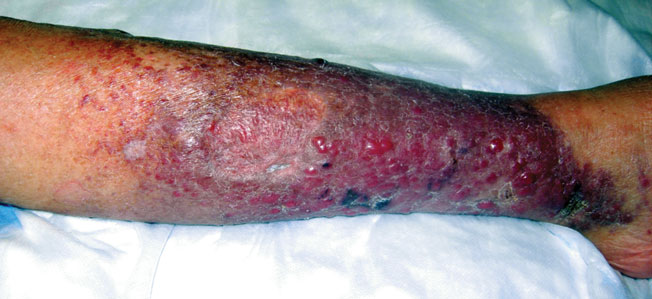
Histopathology showed small round cells (Figure 1) that stained positive for cytokeratin 20 in a paranuclear dotlike pattern (Figure 2). The tumor stained negative for lymphoma (CD45) marker. Fluorodeoxyglucose positron emission tomography showed focally increased activity at cutaneous sites corresponding to the nodules, but lymph nodes and visceral sites did not show areas of increased metabolic activity. She underwent an above-knee amputation. She was started on a chemotherapy regimen of etoposide and carboplatin given that the pathology of the excised limb demonstrated vascular and lymphatic invasion by the tumor cells in the proximal skin margin. After 4 months she presented with gangrenous changes of the amputated limb and evidence of metastasis to the region of the skin flap. Similar tumors presented on the ipsilateral hip. Given her general poor condition and aggressive nature of the tumor, the patient decided to pursue hospice care 6 months after her diagnosis of Merkel cell carcinoma (MCC).
Figure 1. The tumor was composed of sheets of cells with a high nuclear-cytoplasmic ratio and fine chromatin without nucleoli (H&E, original magnification ×40). |
Figure 2. Immunohistochemical staining for cytokera-tin 20 showed positivity in a paranuclear dotlike pattern (original magnification ×40). |
Merkel cell carcinoma usually presents as firm, red to purple papules on sun-exposed skin in older patients with light skin. Factors strongly associated with the development of MCC are age (>65 years), lighter skin types, history of extensive sun exposure, and chronic immune suppression (eg, kidney or heart transplantation, human immunodeficiency virus).1 The rate of MCC has increased 3-fold between 1986 and 2001; the rate of MCC was 0.15 cases per 100,000 individuals in 1986, climbing up to 0.44 cases per 100,000 individuals in 2001.2 Our patient had been on immunosuppressants—prednisone, cyclosporine, and sirolimus—for nearly a decade following kidney transplants, which had been discontinued 2 years prior to presentation.
Heath et al3 defined an acronym AEIOU (asymptomatic/lack of tenderness, expanding rapidly, immune suppression, older than 50 years, UV-exposed site on a person with fair skin) for MCC features derived from 195 patients. They advised that a biopsy is warranted if the patient presents with more than 3 of these features.3
The 1991 MCC staging system was revised in 1999 and 2005 based on experience at Memorial Sloan Kettering Cancer Center (New York, New York).4 In 2010 the American Joint Committee on Cancer staging was introduced for MCC, which follows other skin malignancies.5 Using this TNM staging system for primary tumors, regional lymph nodes, and distant metastasis, our patient at the time of presentation was stage IIB (T3N0M0), with tumor size greater than 5 cm, nodes negative by clinical examination, and no distant metastasis. In a span of 3 months, she had metastasis to the skin, subcutaneous tissue, and distant lymph nodes, which resulted in classification as stage IV, proving the aggressive nature of the tumor.
The newly discovered Merkel cell polyomavirus (MCPyV) is found integrating into the Merkel cell genome. Merkel cell polyomavirus is present in 80% of cancers and is expressed in a clonal pattern, while 90% of MCC patients are seropositive for the same. Unlike antibodies to MCPyV VP1 protein, antibodies to the T antigen for MCPyV track disease burden and may be a useful biomarker for MCC in the future.6
A study of 251 patients in 1970-2002 showed that pathologic nodal staging identifies a group of patients with excellent long-term survival.1 Our patient preferred to undergo positron emission tomography rather than a sentinel lymph node biopsy prior to surgery. Also, after margin-negative excision and pathologic nodal staging, local and nodal recurrence rates were low. Adjuvant chemotherapy for stage III patients showed a trend (P=.08) to decreased survival compared with stage II patients who did not receive chemotherapy.7 A multidisciplinary approach to treatment including surgery, radiation,8 and chemotherapy needs to be created for each individual patient. Merkel cell carcinoma is the cause of death in 35% of patients within 3 years of diagnosis.9
Merkel cell carcinoma is a rare orphan tumor with rapidly increasing incidence in an era of immunosuppression. It has a grave prognosis, as demonstrated in our case, if not detected early. People at increased risk for MCC must have regular skin checks. Unfortunately, our patient was a nursing home resident and had not had a skin check for 2 years prior to presentation.
1. Allen PJ, Bowne WB, Jaques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309.
2. Hodgson NC. Merkel cell carcinoma: changing incidence trends. J Surg Oncol. 2005;89:1-4.
3. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381.
4. Edge S, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. New York, NY: Springer; 2010.
5. Assouline A, Tai P, Joseph K, et al. Merkel cell carcinoma of skin-current controversies and recommendations.Rare Tumors. 2011;4:e23.
6. Paulson KG, Carter JJ, Johnson LG, et al. Antibodies to Merkel cell polyomavirus T-antigen oncoproteins reflect tumor burden in Merkel cell carcinoma patients. Cancer Res. 2010;70:8388-8397.
7. Poulsen M, Rischin D, Walpole E, et al; Trans-Tasman Radiation Oncology Group. High-risk Merkel cell carcinoma of the skin treated with synchronous carboplatin/etoposide and radiation: a Trans-Tasman Radiation Oncology Group Study—TROG 96:07. J Clin Oncol. 2003;21:4371-4376.
8. Lewis KG, Weinstock MA, Weaver AL, et al. Adjuvant local irradiation for Merkel cell carcinoma. Arch Dermatol. 2006;142:693-700.
9. Agelli M, Clegg LX. Epidemiology of primary Merkel cell carcinoma in the United States. J Am Acad Dermatol. 2003;49:832-841.
The Diagnosis: Merkel Cell Carcinoma
Histopathology showed small round cells (Figure 1) that stained positive for cytokeratin 20 in a paranuclear dotlike pattern (Figure 2). The tumor stained negative for lymphoma (CD45) marker. Fluorodeoxyglucose positron emission tomography showed focally increased activity at cutaneous sites corresponding to the nodules, but lymph nodes and visceral sites did not show areas of increased metabolic activity. She underwent an above-knee amputation. She was started on a chemotherapy regimen of etoposide and carboplatin given that the pathology of the excised limb demonstrated vascular and lymphatic invasion by the tumor cells in the proximal skin margin. After 4 months she presented with gangrenous changes of the amputated limb and evidence of metastasis to the region of the skin flap. Similar tumors presented on the ipsilateral hip. Given her general poor condition and aggressive nature of the tumor, the patient decided to pursue hospice care 6 months after her diagnosis of Merkel cell carcinoma (MCC).
Figure 1. The tumor was composed of sheets of cells with a high nuclear-cytoplasmic ratio and fine chromatin without nucleoli (H&E, original magnification ×40). |
Figure 2. Immunohistochemical staining for cytokera-tin 20 showed positivity in a paranuclear dotlike pattern (original magnification ×40). |
Merkel cell carcinoma usually presents as firm, red to purple papules on sun-exposed skin in older patients with light skin. Factors strongly associated with the development of MCC are age (>65 years), lighter skin types, history of extensive sun exposure, and chronic immune suppression (eg, kidney or heart transplantation, human immunodeficiency virus).1 The rate of MCC has increased 3-fold between 1986 and 2001; the rate of MCC was 0.15 cases per 100,000 individuals in 1986, climbing up to 0.44 cases per 100,000 individuals in 2001.2 Our patient had been on immunosuppressants—prednisone, cyclosporine, and sirolimus—for nearly a decade following kidney transplants, which had been discontinued 2 years prior to presentation.
Heath et al3 defined an acronym AEIOU (asymptomatic/lack of tenderness, expanding rapidly, immune suppression, older than 50 years, UV-exposed site on a person with fair skin) for MCC features derived from 195 patients. They advised that a biopsy is warranted if the patient presents with more than 3 of these features.3
The 1991 MCC staging system was revised in 1999 and 2005 based on experience at Memorial Sloan Kettering Cancer Center (New York, New York).4 In 2010 the American Joint Committee on Cancer staging was introduced for MCC, which follows other skin malignancies.5 Using this TNM staging system for primary tumors, regional lymph nodes, and distant metastasis, our patient at the time of presentation was stage IIB (T3N0M0), with tumor size greater than 5 cm, nodes negative by clinical examination, and no distant metastasis. In a span of 3 months, she had metastasis to the skin, subcutaneous tissue, and distant lymph nodes, which resulted in classification as stage IV, proving the aggressive nature of the tumor.
The newly discovered Merkel cell polyomavirus (MCPyV) is found integrating into the Merkel cell genome. Merkel cell polyomavirus is present in 80% of cancers and is expressed in a clonal pattern, while 90% of MCC patients are seropositive for the same. Unlike antibodies to MCPyV VP1 protein, antibodies to the T antigen for MCPyV track disease burden and may be a useful biomarker for MCC in the future.6
A study of 251 patients in 1970-2002 showed that pathologic nodal staging identifies a group of patients with excellent long-term survival.1 Our patient preferred to undergo positron emission tomography rather than a sentinel lymph node biopsy prior to surgery. Also, after margin-negative excision and pathologic nodal staging, local and nodal recurrence rates were low. Adjuvant chemotherapy for stage III patients showed a trend (P=.08) to decreased survival compared with stage II patients who did not receive chemotherapy.7 A multidisciplinary approach to treatment including surgery, radiation,8 and chemotherapy needs to be created for each individual patient. Merkel cell carcinoma is the cause of death in 35% of patients within 3 years of diagnosis.9
Merkel cell carcinoma is a rare orphan tumor with rapidly increasing incidence in an era of immunosuppression. It has a grave prognosis, as demonstrated in our case, if not detected early. People at increased risk for MCC must have regular skin checks. Unfortunately, our patient was a nursing home resident and had not had a skin check for 2 years prior to presentation.
The Diagnosis: Merkel Cell Carcinoma
Histopathology showed small round cells (Figure 1) that stained positive for cytokeratin 20 in a paranuclear dotlike pattern (Figure 2). The tumor stained negative for lymphoma (CD45) marker. Fluorodeoxyglucose positron emission tomography showed focally increased activity at cutaneous sites corresponding to the nodules, but lymph nodes and visceral sites did not show areas of increased metabolic activity. She underwent an above-knee amputation. She was started on a chemotherapy regimen of etoposide and carboplatin given that the pathology of the excised limb demonstrated vascular and lymphatic invasion by the tumor cells in the proximal skin margin. After 4 months she presented with gangrenous changes of the amputated limb and evidence of metastasis to the region of the skin flap. Similar tumors presented on the ipsilateral hip. Given her general poor condition and aggressive nature of the tumor, the patient decided to pursue hospice care 6 months after her diagnosis of Merkel cell carcinoma (MCC).
Figure 1. The tumor was composed of sheets of cells with a high nuclear-cytoplasmic ratio and fine chromatin without nucleoli (H&E, original magnification ×40). |
Figure 2. Immunohistochemical staining for cytokera-tin 20 showed positivity in a paranuclear dotlike pattern (original magnification ×40). |
Merkel cell carcinoma usually presents as firm, red to purple papules on sun-exposed skin in older patients with light skin. Factors strongly associated with the development of MCC are age (>65 years), lighter skin types, history of extensive sun exposure, and chronic immune suppression (eg, kidney or heart transplantation, human immunodeficiency virus).1 The rate of MCC has increased 3-fold between 1986 and 2001; the rate of MCC was 0.15 cases per 100,000 individuals in 1986, climbing up to 0.44 cases per 100,000 individuals in 2001.2 Our patient had been on immunosuppressants—prednisone, cyclosporine, and sirolimus—for nearly a decade following kidney transplants, which had been discontinued 2 years prior to presentation.
Heath et al3 defined an acronym AEIOU (asymptomatic/lack of tenderness, expanding rapidly, immune suppression, older than 50 years, UV-exposed site on a person with fair skin) for MCC features derived from 195 patients. They advised that a biopsy is warranted if the patient presents with more than 3 of these features.3
The 1991 MCC staging system was revised in 1999 and 2005 based on experience at Memorial Sloan Kettering Cancer Center (New York, New York).4 In 2010 the American Joint Committee on Cancer staging was introduced for MCC, which follows other skin malignancies.5 Using this TNM staging system for primary tumors, regional lymph nodes, and distant metastasis, our patient at the time of presentation was stage IIB (T3N0M0), with tumor size greater than 5 cm, nodes negative by clinical examination, and no distant metastasis. In a span of 3 months, she had metastasis to the skin, subcutaneous tissue, and distant lymph nodes, which resulted in classification as stage IV, proving the aggressive nature of the tumor.
The newly discovered Merkel cell polyomavirus (MCPyV) is found integrating into the Merkel cell genome. Merkel cell polyomavirus is present in 80% of cancers and is expressed in a clonal pattern, while 90% of MCC patients are seropositive for the same. Unlike antibodies to MCPyV VP1 protein, antibodies to the T antigen for MCPyV track disease burden and may be a useful biomarker for MCC in the future.6
A study of 251 patients in 1970-2002 showed that pathologic nodal staging identifies a group of patients with excellent long-term survival.1 Our patient preferred to undergo positron emission tomography rather than a sentinel lymph node biopsy prior to surgery. Also, after margin-negative excision and pathologic nodal staging, local and nodal recurrence rates were low. Adjuvant chemotherapy for stage III patients showed a trend (P=.08) to decreased survival compared with stage II patients who did not receive chemotherapy.7 A multidisciplinary approach to treatment including surgery, radiation,8 and chemotherapy needs to be created for each individual patient. Merkel cell carcinoma is the cause of death in 35% of patients within 3 years of diagnosis.9
Merkel cell carcinoma is a rare orphan tumor with rapidly increasing incidence in an era of immunosuppression. It has a grave prognosis, as demonstrated in our case, if not detected early. People at increased risk for MCC must have regular skin checks. Unfortunately, our patient was a nursing home resident and had not had a skin check for 2 years prior to presentation.
1. Allen PJ, Bowne WB, Jaques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309.
2. Hodgson NC. Merkel cell carcinoma: changing incidence trends. J Surg Oncol. 2005;89:1-4.
3. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381.
4. Edge S, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. New York, NY: Springer; 2010.
5. Assouline A, Tai P, Joseph K, et al. Merkel cell carcinoma of skin-current controversies and recommendations.Rare Tumors. 2011;4:e23.
6. Paulson KG, Carter JJ, Johnson LG, et al. Antibodies to Merkel cell polyomavirus T-antigen oncoproteins reflect tumor burden in Merkel cell carcinoma patients. Cancer Res. 2010;70:8388-8397.
7. Poulsen M, Rischin D, Walpole E, et al; Trans-Tasman Radiation Oncology Group. High-risk Merkel cell carcinoma of the skin treated with synchronous carboplatin/etoposide and radiation: a Trans-Tasman Radiation Oncology Group Study—TROG 96:07. J Clin Oncol. 2003;21:4371-4376.
8. Lewis KG, Weinstock MA, Weaver AL, et al. Adjuvant local irradiation for Merkel cell carcinoma. Arch Dermatol. 2006;142:693-700.
9. Agelli M, Clegg LX. Epidemiology of primary Merkel cell carcinoma in the United States. J Am Acad Dermatol. 2003;49:832-841.
1. Allen PJ, Bowne WB, Jaques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309.
2. Hodgson NC. Merkel cell carcinoma: changing incidence trends. J Surg Oncol. 2005;89:1-4.
3. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381.
4. Edge S, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. New York, NY: Springer; 2010.
5. Assouline A, Tai P, Joseph K, et al. Merkel cell carcinoma of skin-current controversies and recommendations.Rare Tumors. 2011;4:e23.
6. Paulson KG, Carter JJ, Johnson LG, et al. Antibodies to Merkel cell polyomavirus T-antigen oncoproteins reflect tumor burden in Merkel cell carcinoma patients. Cancer Res. 2010;70:8388-8397.
7. Poulsen M, Rischin D, Walpole E, et al; Trans-Tasman Radiation Oncology Group. High-risk Merkel cell carcinoma of the skin treated with synchronous carboplatin/etoposide and radiation: a Trans-Tasman Radiation Oncology Group Study—TROG 96:07. J Clin Oncol. 2003;21:4371-4376.
8. Lewis KG, Weinstock MA, Weaver AL, et al. Adjuvant local irradiation for Merkel cell carcinoma. Arch Dermatol. 2006;142:693-700.
9. Agelli M, Clegg LX. Epidemiology of primary Merkel cell carcinoma in the United States. J Am Acad Dermatol. 2003;49:832-841.
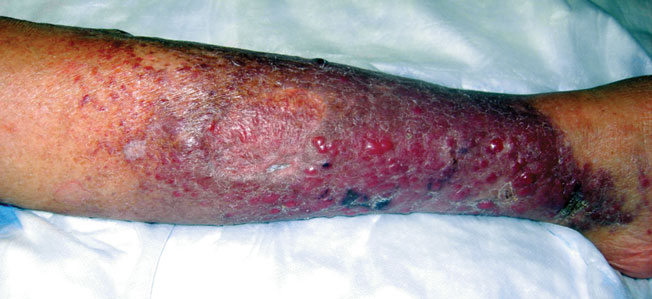
A 66-year-old woman presented with red to violaceous, rapidly growing nodules on the skin. Her medical history was remarkable for diabetes mellitus, hypertension, dyslipidemia, and renal failure. She had 2 rejected kidney transplants and was on hemodialysis at the time of presentation. She noticed asymptomatic nodules present on the left lower leg that progressively coalesced, finally encroaching the whole girth of the limb, spreading from the foot to the knee in a short duration of 3 months. The regional lymph nodes were not clinically palpable.
Ionizing radiation linked to BCC
SAN DIEGO – Patients treated with ionizing radiation were 2.65 to 3.78 times more likely to develop basal cell carcinoma than were controls, and exposure at younger ages or relatively high doses further increased that risk, according to a pooled analysis presented at the annual meeting of the American Society for Dermatologic Surgery.
“Concomitant exposure to ultraviolet radiation may potentiate this effect,” said Dr. Min Deng, a dermatology resident at the University of Chicago. “With newer treatment protocols and improved shielding, it would be interesting to see if the effect of ionizing radiation has changed, or whether we as dermatologists should more actively screen this at-risk population.”
Basal cell carcinoma (BCC) is the most common skin cancer worldwide, but relatively few dermatology papers have assessed the effects of ionizing radiation on the incidence of BCC, said Dr. Deng and coauthor Dr. Diana Bolotin, also of the University of Chicago.
To better understand the link, the researchers searched PubMed for controlled studies on the topic by using the terms “radiation,” “risk,” and “basal cell carcinoma.” They excluded case reports, animal studies, studies published in languages other than English, and trials of radiation as a treatment of BCC, they said. They also excluded studies of atomic bomb survivors, because exposure was uncontrolled and methods to estimate exposure in this group have changed over time, they noted.
In all, six studies published between 1991 and 2012 met the inclusion criteria, and all six showed a statistically significant relative risk or odds of BCC with ionizing radiation exposure, the investigators reported. Three analyses calculated the relative risk of BCC in patients who received radiation for tinea capitis or who underwent total-body irradiation before hematopoietic cell transplantation, they said. Two studies evaluated the odds of BCC in patients with a past history of radiation exposure, and one study assessed time to subsequent BCCs in patients with a history of BCC.
For the three studies that calculated relative risk, the researchers calculated a pooled RR of BCC after ionizing radiation treatment of 2.65 (95% confidence interval, 1.22-5.72) compared with controls. The study of total-body irradiation (TBI) did not report or control for the primary disease for which patients were treated, “which may have confounded the results,” they said. When they excluded that study from their calculation, the overall RR rose to 3.78 (95% CI, 2.62 to 5.44).
Notably, in the two studies of patients with tinea capitis, the relative risk of BCC fell by 12% to 16% for every additional year of age at which patients received radiation treatment, the researchers said. Similarly, risk of BCC dropped by 10.9% for every 1-year increase in age at total-body irradiation prior to cell transplantation.
The combined odds ratio for the next two studies was 4.28 (1.45-12.63). “Likewise, both studies found an elevated odds ratio with younger age at radiation exposure,” the researchers added. One study that stratified patients by type of medical condition detected a “markedly elevated” 8.7 odds of BCC after radiation treatment for acne (2.0 to 38.0), they noted.
The sixth study was a nested case-control analysis that found a statistically significant increase in the odds of BCC starting at a 1-Gy dose of ionizing radiation, and rising linearly up to doses of 35-63.3 Gy. “The risk for developing multiple BCCs also appears to be elevated in patients with a history of radiation therapy,” the researchers said. Patients who had been exposed to ionizing ratio were 2.3 times more likely to develop new BCCs compared with unexposed patients (1.7 to 3.1), they said.
The investigators reported no funding sources or conflicts of interest.
SAN DIEGO – Patients treated with ionizing radiation were 2.65 to 3.78 times more likely to develop basal cell carcinoma than were controls, and exposure at younger ages or relatively high doses further increased that risk, according to a pooled analysis presented at the annual meeting of the American Society for Dermatologic Surgery.
“Concomitant exposure to ultraviolet radiation may potentiate this effect,” said Dr. Min Deng, a dermatology resident at the University of Chicago. “With newer treatment protocols and improved shielding, it would be interesting to see if the effect of ionizing radiation has changed, or whether we as dermatologists should more actively screen this at-risk population.”
Basal cell carcinoma (BCC) is the most common skin cancer worldwide, but relatively few dermatology papers have assessed the effects of ionizing radiation on the incidence of BCC, said Dr. Deng and coauthor Dr. Diana Bolotin, also of the University of Chicago.
To better understand the link, the researchers searched PubMed for controlled studies on the topic by using the terms “radiation,” “risk,” and “basal cell carcinoma.” They excluded case reports, animal studies, studies published in languages other than English, and trials of radiation as a treatment of BCC, they said. They also excluded studies of atomic bomb survivors, because exposure was uncontrolled and methods to estimate exposure in this group have changed over time, they noted.
In all, six studies published between 1991 and 2012 met the inclusion criteria, and all six showed a statistically significant relative risk or odds of BCC with ionizing radiation exposure, the investigators reported. Three analyses calculated the relative risk of BCC in patients who received radiation for tinea capitis or who underwent total-body irradiation before hematopoietic cell transplantation, they said. Two studies evaluated the odds of BCC in patients with a past history of radiation exposure, and one study assessed time to subsequent BCCs in patients with a history of BCC.
For the three studies that calculated relative risk, the researchers calculated a pooled RR of BCC after ionizing radiation treatment of 2.65 (95% confidence interval, 1.22-5.72) compared with controls. The study of total-body irradiation (TBI) did not report or control for the primary disease for which patients were treated, “which may have confounded the results,” they said. When they excluded that study from their calculation, the overall RR rose to 3.78 (95% CI, 2.62 to 5.44).
Notably, in the two studies of patients with tinea capitis, the relative risk of BCC fell by 12% to 16% for every additional year of age at which patients received radiation treatment, the researchers said. Similarly, risk of BCC dropped by 10.9% for every 1-year increase in age at total-body irradiation prior to cell transplantation.
The combined odds ratio for the next two studies was 4.28 (1.45-12.63). “Likewise, both studies found an elevated odds ratio with younger age at radiation exposure,” the researchers added. One study that stratified patients by type of medical condition detected a “markedly elevated” 8.7 odds of BCC after radiation treatment for acne (2.0 to 38.0), they noted.
The sixth study was a nested case-control analysis that found a statistically significant increase in the odds of BCC starting at a 1-Gy dose of ionizing radiation, and rising linearly up to doses of 35-63.3 Gy. “The risk for developing multiple BCCs also appears to be elevated in patients with a history of radiation therapy,” the researchers said. Patients who had been exposed to ionizing ratio were 2.3 times more likely to develop new BCCs compared with unexposed patients (1.7 to 3.1), they said.
The investigators reported no funding sources or conflicts of interest.
SAN DIEGO – Patients treated with ionizing radiation were 2.65 to 3.78 times more likely to develop basal cell carcinoma than were controls, and exposure at younger ages or relatively high doses further increased that risk, according to a pooled analysis presented at the annual meeting of the American Society for Dermatologic Surgery.
“Concomitant exposure to ultraviolet radiation may potentiate this effect,” said Dr. Min Deng, a dermatology resident at the University of Chicago. “With newer treatment protocols and improved shielding, it would be interesting to see if the effect of ionizing radiation has changed, or whether we as dermatologists should more actively screen this at-risk population.”
Basal cell carcinoma (BCC) is the most common skin cancer worldwide, but relatively few dermatology papers have assessed the effects of ionizing radiation on the incidence of BCC, said Dr. Deng and coauthor Dr. Diana Bolotin, also of the University of Chicago.
To better understand the link, the researchers searched PubMed for controlled studies on the topic by using the terms “radiation,” “risk,” and “basal cell carcinoma.” They excluded case reports, animal studies, studies published in languages other than English, and trials of radiation as a treatment of BCC, they said. They also excluded studies of atomic bomb survivors, because exposure was uncontrolled and methods to estimate exposure in this group have changed over time, they noted.
In all, six studies published between 1991 and 2012 met the inclusion criteria, and all six showed a statistically significant relative risk or odds of BCC with ionizing radiation exposure, the investigators reported. Three analyses calculated the relative risk of BCC in patients who received radiation for tinea capitis or who underwent total-body irradiation before hematopoietic cell transplantation, they said. Two studies evaluated the odds of BCC in patients with a past history of radiation exposure, and one study assessed time to subsequent BCCs in patients with a history of BCC.
For the three studies that calculated relative risk, the researchers calculated a pooled RR of BCC after ionizing radiation treatment of 2.65 (95% confidence interval, 1.22-5.72) compared with controls. The study of total-body irradiation (TBI) did not report or control for the primary disease for which patients were treated, “which may have confounded the results,” they said. When they excluded that study from their calculation, the overall RR rose to 3.78 (95% CI, 2.62 to 5.44).
Notably, in the two studies of patients with tinea capitis, the relative risk of BCC fell by 12% to 16% for every additional year of age at which patients received radiation treatment, the researchers said. Similarly, risk of BCC dropped by 10.9% for every 1-year increase in age at total-body irradiation prior to cell transplantation.
The combined odds ratio for the next two studies was 4.28 (1.45-12.63). “Likewise, both studies found an elevated odds ratio with younger age at radiation exposure,” the researchers added. One study that stratified patients by type of medical condition detected a “markedly elevated” 8.7 odds of BCC after radiation treatment for acne (2.0 to 38.0), they noted.
The sixth study was a nested case-control analysis that found a statistically significant increase in the odds of BCC starting at a 1-Gy dose of ionizing radiation, and rising linearly up to doses of 35-63.3 Gy. “The risk for developing multiple BCCs also appears to be elevated in patients with a history of radiation therapy,” the researchers said. Patients who had been exposed to ionizing ratio were 2.3 times more likely to develop new BCCs compared with unexposed patients (1.7 to 3.1), they said.
The investigators reported no funding sources or conflicts of interest.
Key clinical point: Ionizing radiation therapy significantly increased risk of basal cell carcinoma, especially when patients were younger or were treated at relatively high doses.
Major finding: The pooled relative risk of BCC after ionizing radiation treatment was 2.65 (95% confidence interval, 1.22-5.72) compared with controls.
Data source: Pooled analysis of six studies of ionizing radiation exposure and risk or odds of basal cell carcinoma.
Disclosures: The researchers reported no funding sources or conflicts of interest.
What Is Your Diagnosis? Clear Cell Hidradenoma
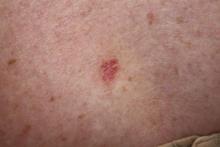
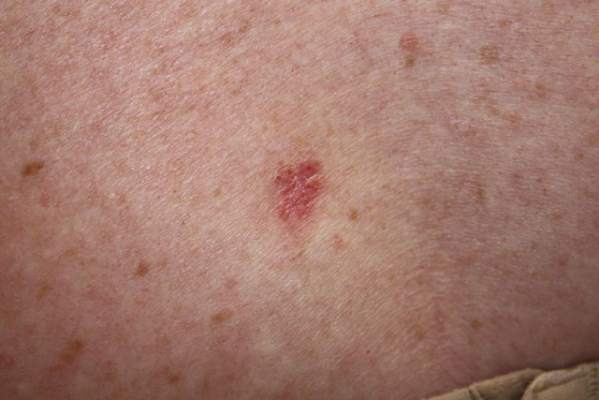
A 70-year-old woman presented to our dermatology clinic with an enlarging lesion on the left anterior aspect of the scalp of 4 years’ duration. She had a history of breast carcinoma in the left breast with positive lymph nodes 2 years prior. Physical examination revealed a 2.5-cm pink, pearly, exophytic plaque on the left anterior aspect of the scalp. The lesion was removed with clear margins by excisional surgery.
The Diagnosis: Clear Cell Hidradenoma
Clear cell hidradenoma (CCH) is a variant of nodular hidradenoma that may contain varying quantities of solid and cystic components and comprises approximately one-third of hidradenomas.1 Clear cell hidradenomas are slow-growing and fairly uncommon adnexal neoplasms derived from either eccrine sweat glands or apocrine glands. Some researchers have regarded hidradenomas as apocrine tumors due to evidence of apocrine decapitation secretion, whereas others note the lack of apocrine and ultrastructural features of immature eccrine glands.2 Clear cell hidradenomas typically develop between the fourth and eighth decades of life, usually peaking during the sixth decade.3 Clear cell hidradenomas usually range in size from 5 to 30 mm and frequently present on the scalp, head, chest, and abdomen; rarely, CCHs present on the joint spaces of the shoulders and knees.3-5 This neoplasm is more common in women than men3 and generally has a flesh-colored, erythematous, red-brown or blue appearance with a tendency to ulcerate and exude a serous discharge (Figure 1).5 The clinical differential diagnosis includes metastatic cancer (eg, renal cell carcinoma, keratoacanthoma, trichoblastoma, trichilemmoma) or other benign adnexal neoplasms.
Histopathologic examination of a CCH generally reveals an unencapsulated and circumscribed neoplasm in the mid or upper dermis with occasional extensions into the subcutaneous fat (Figure 2). The tumor typically presents with 2 types of cells: (1) round, fusiform, or polygonal cells with vesicular nuclei and eosinophilic cytoplasm, and (2) cells with clear cytoplasm and basophilic, often eccentrically located nuclei.6 Ducts are scattered within the neoplasm and are lined by a layer of cuboidal cells that can be highlighted on carcinoembryonic antigen and epithelial membrane antigen immunostaining.6 The tumor cells themselves are highlighted on cytokeratin AE1/AE3 staining.
Malignant transformation rarely is associated with CCH, with de novo clear cell hidradenocarcinoma being more common. Only approximately 6.7% of CCHs have been shown to be malignant, and the malignant tumors feature nuclear atypia, abnormal mitotic figures, necrosis, and infiltration.1,7 Although CCH is a benign adnexal neoplasm, it has a high recurrence rate (approximately 10%) following excision.7 The treatment of choice is complete surgical excision, though Mohs micrographic surgery is advocated, as it promotes thorough examination of the tumor margin to ensure complete tumor removal.8 Our case illustrates the importance of a broad differential diagnosis when treating patients with CCH as well as keeping in mind nonmalignant lesions are far more common than malignant lesions.
1. Volmar K, Cummings TJ, Wang WH, et al. Clear cell hidradenoma: a mimic of metastatic clear cell tumors. Arch Pathol Lab Med. 2005;129:E113-E116.
2. Goh SG, Carr R, Dayrit JF, et al. Mucinous hidradenoma: a report of three cases. J Cutan Pathol. 2007;34:497-502.
3. Gonul M, Cakmak SK, Gul U, et al. A skin tumor in a young girl. diagnosis: clear cell hidradenoma. Indian J Dermatol Venereol Leprol. 2010;76:445-446.
4. Singhal V, Sharma SC, Anil J, et al. Giant benign nodular hidradenoma of the shoulder: a rare tumor of orthopedic practice. Int J Shoulder Surg. 2010;4:93-96.
5. Yu G, Goodloe S Jr, D’Angelis CA, et al. Giant clear cell hidradenoma of the knee. J Cutan Pathol. 2010;37:E37-E41.
6. McKee PH, Calonje E, Granter SR. Tumors of the sweat glands. In: McKee PH, Calonje E, Granter SR. Pathology of the Skin With Clinical Correlations. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2005:1632-1635.
7. Ozawa T, Fujiwara M, Nose K, et al. Clear-cell hidradenoma of the forearm in a young boy. Pediatr Dermatol. 2005;22:450-452.
8. Yavel R, Hinshaw M, Rao V, et al. Hidradenoma and a hidradenocarcinoma of the scalp managed by Mohs micrographic surgery and a multidisciplinary approach: case reports and review of the literature. Dermatol Surg. 2009;35:273-281.
A 70-year-old woman presented to our dermatology clinic with an enlarging lesion on the left anterior aspect of the scalp of 4 years’ duration. She had a history of breast carcinoma in the left breast with positive lymph nodes 2 years prior. Physical examination revealed a 2.5-cm pink, pearly, exophytic plaque on the left anterior aspect of the scalp. The lesion was removed with clear margins by excisional surgery.
The Diagnosis: Clear Cell Hidradenoma
Clear cell hidradenoma (CCH) is a variant of nodular hidradenoma that may contain varying quantities of solid and cystic components and comprises approximately one-third of hidradenomas.1 Clear cell hidradenomas are slow-growing and fairly uncommon adnexal neoplasms derived from either eccrine sweat glands or apocrine glands. Some researchers have regarded hidradenomas as apocrine tumors due to evidence of apocrine decapitation secretion, whereas others note the lack of apocrine and ultrastructural features of immature eccrine glands.2 Clear cell hidradenomas typically develop between the fourth and eighth decades of life, usually peaking during the sixth decade.3 Clear cell hidradenomas usually range in size from 5 to 30 mm and frequently present on the scalp, head, chest, and abdomen; rarely, CCHs present on the joint spaces of the shoulders and knees.3-5 This neoplasm is more common in women than men3 and generally has a flesh-colored, erythematous, red-brown or blue appearance with a tendency to ulcerate and exude a serous discharge (Figure 1).5 The clinical differential diagnosis includes metastatic cancer (eg, renal cell carcinoma, keratoacanthoma, trichoblastoma, trichilemmoma) or other benign adnexal neoplasms.
Histopathologic examination of a CCH generally reveals an unencapsulated and circumscribed neoplasm in the mid or upper dermis with occasional extensions into the subcutaneous fat (Figure 2). The tumor typically presents with 2 types of cells: (1) round, fusiform, or polygonal cells with vesicular nuclei and eosinophilic cytoplasm, and (2) cells with clear cytoplasm and basophilic, often eccentrically located nuclei.6 Ducts are scattered within the neoplasm and are lined by a layer of cuboidal cells that can be highlighted on carcinoembryonic antigen and epithelial membrane antigen immunostaining.6 The tumor cells themselves are highlighted on cytokeratin AE1/AE3 staining.
Malignant transformation rarely is associated with CCH, with de novo clear cell hidradenocarcinoma being more common. Only approximately 6.7% of CCHs have been shown to be malignant, and the malignant tumors feature nuclear atypia, abnormal mitotic figures, necrosis, and infiltration.1,7 Although CCH is a benign adnexal neoplasm, it has a high recurrence rate (approximately 10%) following excision.7 The treatment of choice is complete surgical excision, though Mohs micrographic surgery is advocated, as it promotes thorough examination of the tumor margin to ensure complete tumor removal.8 Our case illustrates the importance of a broad differential diagnosis when treating patients with CCH as well as keeping in mind nonmalignant lesions are far more common than malignant lesions.
A 70-year-old woman presented to our dermatology clinic with an enlarging lesion on the left anterior aspect of the scalp of 4 years’ duration. She had a history of breast carcinoma in the left breast with positive lymph nodes 2 years prior. Physical examination revealed a 2.5-cm pink, pearly, exophytic plaque on the left anterior aspect of the scalp. The lesion was removed with clear margins by excisional surgery.
The Diagnosis: Clear Cell Hidradenoma
Clear cell hidradenoma (CCH) is a variant of nodular hidradenoma that may contain varying quantities of solid and cystic components and comprises approximately one-third of hidradenomas.1 Clear cell hidradenomas are slow-growing and fairly uncommon adnexal neoplasms derived from either eccrine sweat glands or apocrine glands. Some researchers have regarded hidradenomas as apocrine tumors due to evidence of apocrine decapitation secretion, whereas others note the lack of apocrine and ultrastructural features of immature eccrine glands.2 Clear cell hidradenomas typically develop between the fourth and eighth decades of life, usually peaking during the sixth decade.3 Clear cell hidradenomas usually range in size from 5 to 30 mm and frequently present on the scalp, head, chest, and abdomen; rarely, CCHs present on the joint spaces of the shoulders and knees.3-5 This neoplasm is more common in women than men3 and generally has a flesh-colored, erythematous, red-brown or blue appearance with a tendency to ulcerate and exude a serous discharge (Figure 1).5 The clinical differential diagnosis includes metastatic cancer (eg, renal cell carcinoma, keratoacanthoma, trichoblastoma, trichilemmoma) or other benign adnexal neoplasms.
Histopathologic examination of a CCH generally reveals an unencapsulated and circumscribed neoplasm in the mid or upper dermis with occasional extensions into the subcutaneous fat (Figure 2). The tumor typically presents with 2 types of cells: (1) round, fusiform, or polygonal cells with vesicular nuclei and eosinophilic cytoplasm, and (2) cells with clear cytoplasm and basophilic, often eccentrically located nuclei.6 Ducts are scattered within the neoplasm and are lined by a layer of cuboidal cells that can be highlighted on carcinoembryonic antigen and epithelial membrane antigen immunostaining.6 The tumor cells themselves are highlighted on cytokeratin AE1/AE3 staining.
Malignant transformation rarely is associated with CCH, with de novo clear cell hidradenocarcinoma being more common. Only approximately 6.7% of CCHs have been shown to be malignant, and the malignant tumors feature nuclear atypia, abnormal mitotic figures, necrosis, and infiltration.1,7 Although CCH is a benign adnexal neoplasm, it has a high recurrence rate (approximately 10%) following excision.7 The treatment of choice is complete surgical excision, though Mohs micrographic surgery is advocated, as it promotes thorough examination of the tumor margin to ensure complete tumor removal.8 Our case illustrates the importance of a broad differential diagnosis when treating patients with CCH as well as keeping in mind nonmalignant lesions are far more common than malignant lesions.
1. Volmar K, Cummings TJ, Wang WH, et al. Clear cell hidradenoma: a mimic of metastatic clear cell tumors. Arch Pathol Lab Med. 2005;129:E113-E116.
2. Goh SG, Carr R, Dayrit JF, et al. Mucinous hidradenoma: a report of three cases. J Cutan Pathol. 2007;34:497-502.
3. Gonul M, Cakmak SK, Gul U, et al. A skin tumor in a young girl. diagnosis: clear cell hidradenoma. Indian J Dermatol Venereol Leprol. 2010;76:445-446.
4. Singhal V, Sharma SC, Anil J, et al. Giant benign nodular hidradenoma of the shoulder: a rare tumor of orthopedic practice. Int J Shoulder Surg. 2010;4:93-96.
5. Yu G, Goodloe S Jr, D’Angelis CA, et al. Giant clear cell hidradenoma of the knee. J Cutan Pathol. 2010;37:E37-E41.
6. McKee PH, Calonje E, Granter SR. Tumors of the sweat glands. In: McKee PH, Calonje E, Granter SR. Pathology of the Skin With Clinical Correlations. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2005:1632-1635.
7. Ozawa T, Fujiwara M, Nose K, et al. Clear-cell hidradenoma of the forearm in a young boy. Pediatr Dermatol. 2005;22:450-452.
8. Yavel R, Hinshaw M, Rao V, et al. Hidradenoma and a hidradenocarcinoma of the scalp managed by Mohs micrographic surgery and a multidisciplinary approach: case reports and review of the literature. Dermatol Surg. 2009;35:273-281.
1. Volmar K, Cummings TJ, Wang WH, et al. Clear cell hidradenoma: a mimic of metastatic clear cell tumors. Arch Pathol Lab Med. 2005;129:E113-E116.
2. Goh SG, Carr R, Dayrit JF, et al. Mucinous hidradenoma: a report of three cases. J Cutan Pathol. 2007;34:497-502.
3. Gonul M, Cakmak SK, Gul U, et al. A skin tumor in a young girl. diagnosis: clear cell hidradenoma. Indian J Dermatol Venereol Leprol. 2010;76:445-446.
4. Singhal V, Sharma SC, Anil J, et al. Giant benign nodular hidradenoma of the shoulder: a rare tumor of orthopedic practice. Int J Shoulder Surg. 2010;4:93-96.
5. Yu G, Goodloe S Jr, D’Angelis CA, et al. Giant clear cell hidradenoma of the knee. J Cutan Pathol. 2010;37:E37-E41.
6. McKee PH, Calonje E, Granter SR. Tumors of the sweat glands. In: McKee PH, Calonje E, Granter SR. Pathology of the Skin With Clinical Correlations. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2005:1632-1635.
7. Ozawa T, Fujiwara M, Nose K, et al. Clear-cell hidradenoma of the forearm in a young boy. Pediatr Dermatol. 2005;22:450-452.
8. Yavel R, Hinshaw M, Rao V, et al. Hidradenoma and a hidradenocarcinoma of the scalp managed by Mohs micrographic surgery and a multidisciplinary approach: case reports and review of the literature. Dermatol Surg. 2009;35:273-281.
Multiple Tumors of the Follicular Infundibulum: A Cutaneous Reaction Pattern?
Tumor of the follicular infundibulum (TFI) was first described by Mehregan and Butler1 in a patient with multiple papules. It typically presents as a solitary lesion that mainly affects the face, neck, and upper trunk, and it generally occurs in elderly patients with a female predilection and occasional vulvar involvement. Sometimes TFI may coexist with an unusual trichilemmal tumor or basal cell carcinoma (BCC) with sebaceous or sweat duct differentiation. Other features of TFI include an eosinophilic cuticle, ductal differentiation, coronoid lamellae, and desmoplasia. Interestingly, approximately one-fourth of reported TFIs were found to be associated with other cutaneous lesions, including BCC, actinic keratosis, desmoplastic malignant melanoma, junctional melanocytic nevus, trichilemmoma, and epidermal inclusion cyst.2 Eruptive forms of TFI are rare, presenting as macules, smooth or slightly keratotic papules, or depressed lesions with a flesh-colored erythematous or hypopigmented appearance. We report the case of a 49-year-old woman with multiple TFIs and some new features.
Case Report
A 49-year-old woman presented with multiple lesions on the arms, shoulders, trunk, buttocks, and legs of more than 3 years’ duration. According to the patient, multiple small, reddish papules gradually appeared on the arms and legs approximately 3 years prior to presentation and were accompanied by considerable pruritus. She sought medical assistance several times at local clinics. A diagnosis of eczema and prurigo was made and oral antihistamines and topical glucocorticoids were administered for more than 2 years. The pruritus was controlled to some degree but recurred after discontinuation of treatment. Five months later, the lesions flared up suddenly and rapidly to the back and buttocks, just around the skin areas treated with cupping glass, a traditional Chinese therapy, for heat syncope. She reported constant itching that kept her awake at night.
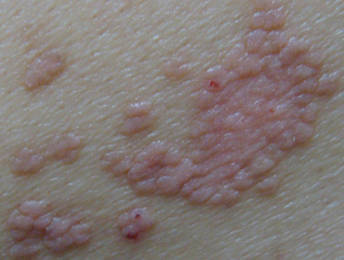
Physical examination revealed hundreds of erythematous maculopapules scattered over the arms, shoulders, trunk, buttocks, and legs. The individual lesions were minimally elevated, irregularly shaped, and slightly scaly, and they were distributed in a relatively symmetrical manner. Some of the lesions coalesced to form small plaques measuring approximately 0.5 to 3 cm in diameter (Figure 1). Interestingly, several annulated lesions with a hypopigmented center and hyperpigmented periphery could be observed at areas treated with cupping glass, showing a typical presentation of the Köbner phenomenon (Figure 2). No obvious association with sun exposure or hair loss was detected. The patient’s medical history was unremarkable and there was no known family history of similar skin lesions.
|
The differential diagnosis included lichen planus, prurigo, and adnexal tumors. Histopathologic examination of biopsies from lesions on the back and around the cupping glass areas revealed a benign platelike proliferation of pale-staining epithelial cells in the papillary dermis connected to the overlying epidermis with a moderate lymphatic cell infiltration (Figure 3A). Weigert staining revealed a network of elastic fibers surrounding the base of the tumor (Figure 3B). The pale epithelial cells were positive on periodic acid–Schiff staining (Figure 3C). Immunohistochemical staining revealed that the proliferation was positive for keratin 17 (Figure 3D).
These striking clinical and histological findings suggested the lesions were multiple TFIs. Treatment with tretinoin ointment 0.1% twice daily for 2 months was ineffective. The patient refused treatment with either oral acitretin or dapsone or invasive techniques such as lasers and cryotherapy. She has been on careful follow-up for the last 2 years with new lesions appearing intermittently.
Comment
The etiology of TFI still is unknown but may be related to environmental factors or genetic changes. Tumors of the follicular infundibulum generally are classified as solitary, eruptive, associated with other lesions of Cowden disease, associated with a single tumor, or TFI-like epidermal changes.3 The eruptive form has seldom been reported to date. Eruptive TFIs are nonspecific, ranging in number from several to more than 100. Clinical characteristics and distribution of the lesions can vary from patient to patient, but the lesions tend to be quite monomorphous, even in cases with more than 100 lesions.4 Histopathologic features usually are typical and distinctive, allowing its definite diagnosis. To confirm the diagnosis, the immunohistochemical profile may be useful to characterize its follicular origin, which is positive for keratin 17. Tumors of the follicular infundibulum usually are benign; however, in one patient with more than 100 lesions, there was documented transformation into BCC.5 Due to the potential for malignant transformation as well as its occurrence within the spectrum of lesions associated with Cowden disease,6,7 long-term supervision of TFI is strongly recommended, as was the case with our patient.
A remarkable feature in our case was the patient’s severe pruritus, as most reported cases have been asymptomatic. The increased association with other cutaneous lesions,2 the Köbner phenomenon, and the underlying inflammatory cell infiltration of the tumors in our case strongly suggested that eruptive TFI may represent a kind of cutaneous reaction.
|
Conclusion
The clinical findings of severe pruritus and the Köbner phenomenon in our patient further expand the constellation of the clinical presentation of the eruptive variant of TFI.
1. Mehregan AH, Butler JD. A tumor of follicular infundibulum. report of a case. Arch Dermatol. 1961;83:924-927.
2. Abbas O, Mahalingam M. Tumor of the follicular infundibulum: an epidermal reaction pattern? Am J Dermatopathol. 2009;31:626633.
3. Cribier B, Grosshans E. Tumor of the follicular infundibulum: a clinicopathologic study. J Am Acad Dermatol. 1995;33:979-984.
4. Sartorelli AC, Leite FE, Friedman IV, et al. Vitiligoid hypopigmented macules and tumor of the follicular infundibulum [in Portuguese]. An Bras Dermatol. 2009;84:68-70.
5. Schnitzler L, Civatte J, Robin F, et al. Multiple tumors of the follicular infundibulum with basocellular degeneration. apropos of a case [in French]. Ann Dermatol Venereol. 1987;114:551-556.
6. Starink TM, Meijer CJ, Brownstein MH. The cutaneous pathology of Cowden’s disease: new findings. J Cutan Pathol. 1985;12:83-93.
7. Weyers W, Hörster S, Diaz-Cascajo C. Tumor of follicular infundibulum is basal cell carcinoma. Am J Dermatopathol. 2009;31:634-641.
Tumor of the follicular infundibulum (TFI) was first described by Mehregan and Butler1 in a patient with multiple papules. It typically presents as a solitary lesion that mainly affects the face, neck, and upper trunk, and it generally occurs in elderly patients with a female predilection and occasional vulvar involvement. Sometimes TFI may coexist with an unusual trichilemmal tumor or basal cell carcinoma (BCC) with sebaceous or sweat duct differentiation. Other features of TFI include an eosinophilic cuticle, ductal differentiation, coronoid lamellae, and desmoplasia. Interestingly, approximately one-fourth of reported TFIs were found to be associated with other cutaneous lesions, including BCC, actinic keratosis, desmoplastic malignant melanoma, junctional melanocytic nevus, trichilemmoma, and epidermal inclusion cyst.2 Eruptive forms of TFI are rare, presenting as macules, smooth or slightly keratotic papules, or depressed lesions with a flesh-colored erythematous or hypopigmented appearance. We report the case of a 49-year-old woman with multiple TFIs and some new features.
Case Report
A 49-year-old woman presented with multiple lesions on the arms, shoulders, trunk, buttocks, and legs of more than 3 years’ duration. According to the patient, multiple small, reddish papules gradually appeared on the arms and legs approximately 3 years prior to presentation and were accompanied by considerable pruritus. She sought medical assistance several times at local clinics. A diagnosis of eczema and prurigo was made and oral antihistamines and topical glucocorticoids were administered for more than 2 years. The pruritus was controlled to some degree but recurred after discontinuation of treatment. Five months later, the lesions flared up suddenly and rapidly to the back and buttocks, just around the skin areas treated with cupping glass, a traditional Chinese therapy, for heat syncope. She reported constant itching that kept her awake at night.
Physical examination revealed hundreds of erythematous maculopapules scattered over the arms, shoulders, trunk, buttocks, and legs. The individual lesions were minimally elevated, irregularly shaped, and slightly scaly, and they were distributed in a relatively symmetrical manner. Some of the lesions coalesced to form small plaques measuring approximately 0.5 to 3 cm in diameter (Figure 1). Interestingly, several annulated lesions with a hypopigmented center and hyperpigmented periphery could be observed at areas treated with cupping glass, showing a typical presentation of the Köbner phenomenon (Figure 2). No obvious association with sun exposure or hair loss was detected. The patient’s medical history was unremarkable and there was no known family history of similar skin lesions.
|
The differential diagnosis included lichen planus, prurigo, and adnexal tumors. Histopathologic examination of biopsies from lesions on the back and around the cupping glass areas revealed a benign platelike proliferation of pale-staining epithelial cells in the papillary dermis connected to the overlying epidermis with a moderate lymphatic cell infiltration (Figure 3A). Weigert staining revealed a network of elastic fibers surrounding the base of the tumor (Figure 3B). The pale epithelial cells were positive on periodic acid–Schiff staining (Figure 3C). Immunohistochemical staining revealed that the proliferation was positive for keratin 17 (Figure 3D).
These striking clinical and histological findings suggested the lesions were multiple TFIs. Treatment with tretinoin ointment 0.1% twice daily for 2 months was ineffective. The patient refused treatment with either oral acitretin or dapsone or invasive techniques such as lasers and cryotherapy. She has been on careful follow-up for the last 2 years with new lesions appearing intermittently.
Comment
The etiology of TFI still is unknown but may be related to environmental factors or genetic changes. Tumors of the follicular infundibulum generally are classified as solitary, eruptive, associated with other lesions of Cowden disease, associated with a single tumor, or TFI-like epidermal changes.3 The eruptive form has seldom been reported to date. Eruptive TFIs are nonspecific, ranging in number from several to more than 100. Clinical characteristics and distribution of the lesions can vary from patient to patient, but the lesions tend to be quite monomorphous, even in cases with more than 100 lesions.4 Histopathologic features usually are typical and distinctive, allowing its definite diagnosis. To confirm the diagnosis, the immunohistochemical profile may be useful to characterize its follicular origin, which is positive for keratin 17. Tumors of the follicular infundibulum usually are benign; however, in one patient with more than 100 lesions, there was documented transformation into BCC.5 Due to the potential for malignant transformation as well as its occurrence within the spectrum of lesions associated with Cowden disease,6,7 long-term supervision of TFI is strongly recommended, as was the case with our patient.
A remarkable feature in our case was the patient’s severe pruritus, as most reported cases have been asymptomatic. The increased association with other cutaneous lesions,2 the Köbner phenomenon, and the underlying inflammatory cell infiltration of the tumors in our case strongly suggested that eruptive TFI may represent a kind of cutaneous reaction.
|
Conclusion
The clinical findings of severe pruritus and the Köbner phenomenon in our patient further expand the constellation of the clinical presentation of the eruptive variant of TFI.
Tumor of the follicular infundibulum (TFI) was first described by Mehregan and Butler1 in a patient with multiple papules. It typically presents as a solitary lesion that mainly affects the face, neck, and upper trunk, and it generally occurs in elderly patients with a female predilection and occasional vulvar involvement. Sometimes TFI may coexist with an unusual trichilemmal tumor or basal cell carcinoma (BCC) with sebaceous or sweat duct differentiation. Other features of TFI include an eosinophilic cuticle, ductal differentiation, coronoid lamellae, and desmoplasia. Interestingly, approximately one-fourth of reported TFIs were found to be associated with other cutaneous lesions, including BCC, actinic keratosis, desmoplastic malignant melanoma, junctional melanocytic nevus, trichilemmoma, and epidermal inclusion cyst.2 Eruptive forms of TFI are rare, presenting as macules, smooth or slightly keratotic papules, or depressed lesions with a flesh-colored erythematous or hypopigmented appearance. We report the case of a 49-year-old woman with multiple TFIs and some new features.
Case Report
A 49-year-old woman presented with multiple lesions on the arms, shoulders, trunk, buttocks, and legs of more than 3 years’ duration. According to the patient, multiple small, reddish papules gradually appeared on the arms and legs approximately 3 years prior to presentation and were accompanied by considerable pruritus. She sought medical assistance several times at local clinics. A diagnosis of eczema and prurigo was made and oral antihistamines and topical glucocorticoids were administered for more than 2 years. The pruritus was controlled to some degree but recurred after discontinuation of treatment. Five months later, the lesions flared up suddenly and rapidly to the back and buttocks, just around the skin areas treated with cupping glass, a traditional Chinese therapy, for heat syncope. She reported constant itching that kept her awake at night.
Physical examination revealed hundreds of erythematous maculopapules scattered over the arms, shoulders, trunk, buttocks, and legs. The individual lesions were minimally elevated, irregularly shaped, and slightly scaly, and they were distributed in a relatively symmetrical manner. Some of the lesions coalesced to form small plaques measuring approximately 0.5 to 3 cm in diameter (Figure 1). Interestingly, several annulated lesions with a hypopigmented center and hyperpigmented periphery could be observed at areas treated with cupping glass, showing a typical presentation of the Köbner phenomenon (Figure 2). No obvious association with sun exposure or hair loss was detected. The patient’s medical history was unremarkable and there was no known family history of similar skin lesions.
|
The differential diagnosis included lichen planus, prurigo, and adnexal tumors. Histopathologic examination of biopsies from lesions on the back and around the cupping glass areas revealed a benign platelike proliferation of pale-staining epithelial cells in the papillary dermis connected to the overlying epidermis with a moderate lymphatic cell infiltration (Figure 3A). Weigert staining revealed a network of elastic fibers surrounding the base of the tumor (Figure 3B). The pale epithelial cells were positive on periodic acid–Schiff staining (Figure 3C). Immunohistochemical staining revealed that the proliferation was positive for keratin 17 (Figure 3D).
These striking clinical and histological findings suggested the lesions were multiple TFIs. Treatment with tretinoin ointment 0.1% twice daily for 2 months was ineffective. The patient refused treatment with either oral acitretin or dapsone or invasive techniques such as lasers and cryotherapy. She has been on careful follow-up for the last 2 years with new lesions appearing intermittently.
Comment
The etiology of TFI still is unknown but may be related to environmental factors or genetic changes. Tumors of the follicular infundibulum generally are classified as solitary, eruptive, associated with other lesions of Cowden disease, associated with a single tumor, or TFI-like epidermal changes.3 The eruptive form has seldom been reported to date. Eruptive TFIs are nonspecific, ranging in number from several to more than 100. Clinical characteristics and distribution of the lesions can vary from patient to patient, but the lesions tend to be quite monomorphous, even in cases with more than 100 lesions.4 Histopathologic features usually are typical and distinctive, allowing its definite diagnosis. To confirm the diagnosis, the immunohistochemical profile may be useful to characterize its follicular origin, which is positive for keratin 17. Tumors of the follicular infundibulum usually are benign; however, in one patient with more than 100 lesions, there was documented transformation into BCC.5 Due to the potential for malignant transformation as well as its occurrence within the spectrum of lesions associated with Cowden disease,6,7 long-term supervision of TFI is strongly recommended, as was the case with our patient.
A remarkable feature in our case was the patient’s severe pruritus, as most reported cases have been asymptomatic. The increased association with other cutaneous lesions,2 the Köbner phenomenon, and the underlying inflammatory cell infiltration of the tumors in our case strongly suggested that eruptive TFI may represent a kind of cutaneous reaction.
|
Conclusion
The clinical findings of severe pruritus and the Köbner phenomenon in our patient further expand the constellation of the clinical presentation of the eruptive variant of TFI.
1. Mehregan AH, Butler JD. A tumor of follicular infundibulum. report of a case. Arch Dermatol. 1961;83:924-927.
2. Abbas O, Mahalingam M. Tumor of the follicular infundibulum: an epidermal reaction pattern? Am J Dermatopathol. 2009;31:626633.
3. Cribier B, Grosshans E. Tumor of the follicular infundibulum: a clinicopathologic study. J Am Acad Dermatol. 1995;33:979-984.
4. Sartorelli AC, Leite FE, Friedman IV, et al. Vitiligoid hypopigmented macules and tumor of the follicular infundibulum [in Portuguese]. An Bras Dermatol. 2009;84:68-70.
5. Schnitzler L, Civatte J, Robin F, et al. Multiple tumors of the follicular infundibulum with basocellular degeneration. apropos of a case [in French]. Ann Dermatol Venereol. 1987;114:551-556.
6. Starink TM, Meijer CJ, Brownstein MH. The cutaneous pathology of Cowden’s disease: new findings. J Cutan Pathol. 1985;12:83-93.
7. Weyers W, Hörster S, Diaz-Cascajo C. Tumor of follicular infundibulum is basal cell carcinoma. Am J Dermatopathol. 2009;31:634-641.
1. Mehregan AH, Butler JD. A tumor of follicular infundibulum. report of a case. Arch Dermatol. 1961;83:924-927.
2. Abbas O, Mahalingam M. Tumor of the follicular infundibulum: an epidermal reaction pattern? Am J Dermatopathol. 2009;31:626633.
3. Cribier B, Grosshans E. Tumor of the follicular infundibulum: a clinicopathologic study. J Am Acad Dermatol. 1995;33:979-984.
4. Sartorelli AC, Leite FE, Friedman IV, et al. Vitiligoid hypopigmented macules and tumor of the follicular infundibulum [in Portuguese]. An Bras Dermatol. 2009;84:68-70.
5. Schnitzler L, Civatte J, Robin F, et al. Multiple tumors of the follicular infundibulum with basocellular degeneration. apropos of a case [in French]. Ann Dermatol Venereol. 1987;114:551-556.
6. Starink TM, Meijer CJ, Brownstein MH. The cutaneous pathology of Cowden’s disease: new findings. J Cutan Pathol. 1985;12:83-93.
7. Weyers W, Hörster S, Diaz-Cascajo C. Tumor of follicular infundibulum is basal cell carcinoma. Am J Dermatopathol. 2009;31:634-641.
Practice Points
- Multiple tumors of the follicular infundibulum (TFIs) sometimes may have the potential for malignant transformation; therefore, long-term supervision of TFI is strongly recommended.
- Eruptive forms of TFI are rare. In our patient, severe pruritus, the Köbner phenomenon, and the underlying inflammatory cell infiltration of the tumors strongly suggested that eruptive TFI may represent a kind of cutaneous reaction.
Most Common Dermatologic Conditions Encountered by Dermatologists and Nondermatologists
Skin diseases are highly prevalent in the United States, affecting an estimated 1 in 3 Americans at any given time.1,2 In 2009 the direct medical costs associated with skin-related diseases, including health services and prescriptions, was approximately $22 billion; the annual total economic burden was estimated to be closer to $96 billion when factoring in the cost of lost productivity and pay for symptom relief.3,4 Effective and efficient management of skin disease is essential to minimizing cost and morbidity. Nondermatologists traditionally have diagnosed the majority of skin diseases.5,6 In particular, primary care physicians commonly manage dermatologic conditions and often are the first health care providers to encounter patients presenting with skin problems. A predicted shortage of dermatologists will likely contribute to an increase in this trend.7,8 Therefore, it is important to adequately prepare nondermatologists to evaluate and treat the skin conditions that they are most likely to encounter in their scope of practice.
Residents, particularly in primary care specialties, often have opportunities to spend 2 to 4 weeks with a dermatologist to learn about skin diseases; however, the skin conditions most often encountered by dermatologists may differ from those most often encountered by physicians in other specialties. For instance, one study demonstrated a disparity between the most common skin problems seen by dermatologists and internists.9 These dissimilarities should be recognized and addressed in curriculum content. The purpose of this study was to identify and compare the 20 most common dermatologic conditions reported by dermatologists versus those reported by nondermatologists (ie, internists, pediatricians, family physicians, emergency medicine physicians, general surgeons, otolaryngologists) from 2001 to 2010. Data also were analyzed to determine the top 20 conditions referred to dermatologists by nondermatologists as a potential indicator for areas of further improvement within medical education. With this knowledge, we hope educational curricula and self-study can be modified to reflect the current epidemiology of cutaneous diseases, thereby improving patient care.
Methods
Data from 2001 to 2010 were extracted from the National Ambulatory Medical Care Survey (NAMCS), which is an ongoing survey conducted by the National Center for Health Statistics. The NAMCS collects descriptive data regarding ambulatory visits to nonfederal office-based physicians in the United States. Participating physicians are instructed to record information about patient visits for a 1-week period, including patient demographics, insurance status, reason for visit, diagnoses, procedures, therapeutics, and referrals made at that time. Data collected for the NAMCS are entered into a multistage probability sample to produce national estimates. Within dermatology, an average of 118 dermatologists are sampled each year, and over the last 10 years, participation rates have ranged from 47% to 77%.
International Classification of Diseases, Ninth Revision, Clinical Modification codes were identified to determine the diagnoses that could be classified as dermatologic conditions. Select infectious and neoplastic disorders of the skin and mucous membrane conditions were included as well as the codes for skin diseases. Nondermatologic diagnoses and V codes were not included in the study. Data for all providers were studied to identify outpatient visits associated with the primary diagnosis of a dermatologic condition. Minor diagnoses that were considered to be subsets of major diagnoses were combined to allow better analysis of the data. For example, all tinea infections (ie, dermatophytosis of various sites, dermatomycosis unspecified) were combined into 1 diagnosis referred to as tinea because the recognition and treatment of this disease does not vary tremendously by anatomic location. Visits to dermatologists that listed nonspecific diagnoses and codes (eg, other postsurgical status [V45.89], neoplasm of uncertain behavior site unspecified [238.9]) were assumed to be for dermatologic problems.
Sampling weights were applied to obtain estimates for the number of each diagnosis made nationally. All data analyses were performed using SAS software and linear regression models were generated using SAS PROC SURVEYREG.
Data were analyzed to determine the dermatologic conditions most commonly encountered by dermatologists and nondermatologists in emergency medicine, family medicine, general surgery, internal medicine, otolaryngology, and pediatrics; these specialties include physicians who are known to commonly diagnose and treat skin diseases.10 Data also were analyzed to determine the most common conditions referred to dermatologists for treatment by nondermatologists from the selected specialties. Permission to conduct this study was obtained from the Wake Forest University institutional review board (Winston-Salem, North Carolina).
Results
From 2001 to 2010, more than 700 million outpatient visits for skin-related problems were identified, with 676.3 million visits to dermatologists, emergency medicine physicians, family practitioners, general surgeons, internists, otolaryngologists, and pediatricians. More than half (52.9%) of all skin-related visits were addressed by nondermatologists during this time. Among nondermatologists, family practitioners encountered the greatest number of skin diseases (20.5%), followed by pediatricians (11.3%), internists (9.2%), general surgeons (3.4%), otolaryngologists (1.0%), and emergency medicine physicians (0.2%)(Table 1).
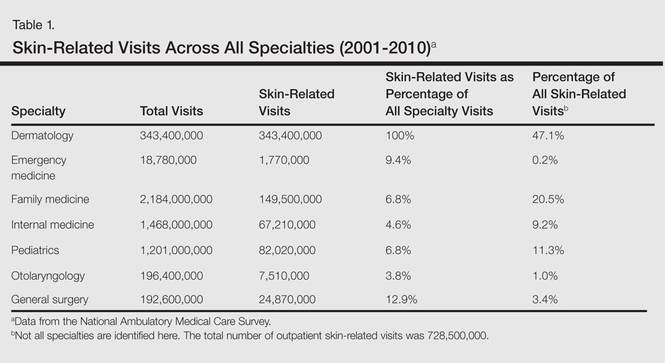
Benign tumors and acne were the most common cutaneous conditions referred to dermatologists by nondermatologists (10.6% and 10.1% of all dermatology referrals, respectively), followed by nonmelanoma skin cancers (9.7%), contact dermatitis (8.8%), and actinic keratosis (7.8%)(Table 2). The top 20 conditions referred to dermatologists accounted for 83.7% of all outpatient referrals to dermatologists.
Among the diseases most frequently reported by nondermatologists, contact dermatitis was the most common (12.0%), with twice the number of visits to nondermatologists for contact dermatitis than to dermatologists (51.6 million vs 25.3 million). In terms of disease categories, infectious skin diseases (ie, bacterial [cellulitis/abscess], viral [warts, herpesvirus], fungal [tinea] and yeast [candida] etiologies) were the most common dermatologic conditions reported by nondermatologists (Table 2).
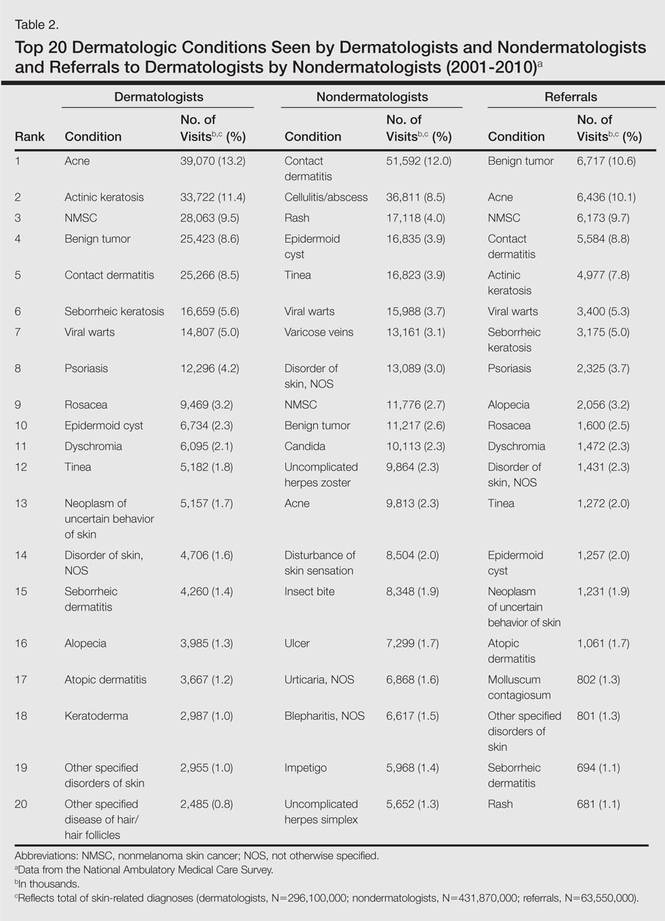
The top 20 dermatologic conditions reported by dermatologists accounted for 85.4% of all diagnoses made by dermatologists. Diseases that were among the top 20 conditions encountered by dermatologists but were not among the top 20 for nondermatologists included actinic keratosis, seborrheic keratosis, atopic dermatitis, psoriasis, alopecia, rosacea, dyschromia, seborrheic dermatitis, follicular disease, and neoplasm of uncertain behavior of skin. Additionally, 5 of the top 20 conditions encountered by dermatologists also were among the top 20 for only 1 individual nondermatologic specialty; these included atopic dermatitis (pediatrics), seborrheic dermatitis (pediatrics), psoriasis (internal medicine), rosacea (otolaryngology), and keratoderma (general surgery). Seborrheic dermatitis, psoriasis, and rosacea also were among the top 20 conditions most commonly referred to dermatologists for treatment by nondermatologists. Table 3 shows the top 20 dermatologic conditions encountered by nondermatologists by comparison.
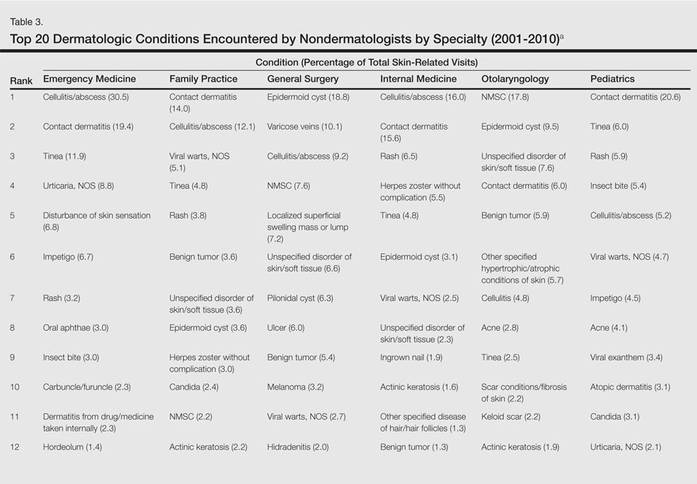
Comment
According to NAMCS data from 2001 to 2010, visits to nondermatologists accounted for more than half of total outpatient visits for cutaneous diseases in the United States, whereas visits to dermatologists accounted for 47.1%. These findings are consistent with historical data indicating that 30% to 40% of skin-related visits are to dermatologists, and the majority of patients with skin disease are diagnosed by nondermatologists.5,6
Past data indicate that most visits to dermatologists were for evaluation of acne, infections, psoriasis, and neoplasms, whereas most visits to nondermatologists were for evaluation of epidermoid cysts, impetigo, plant dermatitis, cellulitis, and diaper rash.9 Over the last 10 years, acne has been more commonly encountered by nondermatologists, especially pediatricians. Additionally, infectious etiologies have been seen in larger volume by nondermatologists.9 Together, infectious cutaneous conditions make up nearly one-fourth of dermatologic encounters by emergency medicine physicians, internists, and family practitioners but are not within the top 20 diagnoses referred to dermatologists, which suggests that uncomplicated cases of cellulitis, herpes zoster, and other skin-related infections are largely managed by nondermatologists.5,6 Contact dermatitis, often caused by specific allergens such as detergents, solvents, and topical products, was one of the most common reported dermatologic encounters among dermatologists and nondermatologists and also was the fourth most common condition referred to dermatologists by nondermatologists for treatment; however, there may be an element of overuse of the International Classification of Diseases, Ninth Revision code, as any presumed contact dermatitis of unspecified cause can be reported under 692.9 defined as contact dermatitis and other eczema, unspecified cause. The high rate of referrals to dermatologists by nondermatologists may be for patch testing and further management. Additionally, there are no specific codes for allergic or irritant dermatitis, thus these diseases may be lumped together.
Although nearly half of all dermatologic encounters were seen by nondermatologists, dermatologists see a much larger proportion of patients with skin disease than nondermatologists and nondermatologists often have limited exposure to the field of dermatology during residency training. Studies have demonstrated differences in the abilities of dermatologists and nondermatologists to correctly diagnose common cutaneous diseases, which unsurprisingly revealed greater diagnostic accuracy demonstrated by dermatologists.11-16 The increase in acne and skin-related infections reported by nondermatologists is consistent with possible efforts to increase formal training in frequently encountered skin diseases. In one study evaluating the impact of a formal 3-week dermatology curriculum on an internal medicine department, internists demonstrated 100% accuracy in the diagnosis of acne and herpes zoster in contrast to 29% for tinea and 12% for lichen planus.5,6
The current Accreditation Council for Graduate Medical Education guidelines place little emphasis on exposure to dermatology training during residency for internists and pediatricians, as this training is not a required component of these programs.17 Two core problems with current training regarding the evaluation and management of cutaneous disease are minimal exposure to dermatologic conditions in medical school and residency and lack of consensus on the core topics that should be taught to nondermatologists.18 Exposure to dermatologic conditions through rotations in medical school has been shown to increase residents’ self-reported confidence in diagnosing and treating alopecia, cutaneous drug eruptions, warts, acne, rosacea, nonmelanoma skin cancers, sun damage, psoriasis, seborrhea, atopic dermatitis, and contact dermatitis; however, the majority of primary care residents surveyed still felt that this exposure in medical school was inadequate.19
In creating a core curriculum for dermatology training for nondermatologists, it is important to consider the dermatologic conditions that are most frequently encountered by these specialties. Our study revealed that the most commonly encountered dermatologic conditions differ among dermatologists and nondermatologists, with a fair degree of variation even among individual specialties. Failure to recognize these discrepancies has likely contributed to the challenges faced by nondermatologists in the diagnosis and management of dermatologic disease. In this study, contact dermatitis, epidermoid cysts, and skin infections were the most common dermatologic conditions encountered by nondermatologists and also were among the top skin diseases referred to dermatologists by nondermatologists. This finding suggests that nondermatologists are able to identify these conditions but have a tendency to refer approximately 10% of these patients to dermatology for further management. Clinical evaluation and medical management of these cutaneous diseases may be an important area of focus for medical school curricula, as the treatment of these diseases is within the capabilities of the nondermatologist. For example, initial management of dermatitis requires determination of the type of dermatitis (ie, essential, contact, atopic, seborrheic, stasis) and selection of an appropriate topical steroid, with referral to a dermatologist needed for questionable or refractory cases. Although a curriculum cannot be built solely on a list of the top 20 diagnoses provided here, these data may serve as a preliminary platform for medical school dermatology curriculum design. The curriculum also should include serious skin diseases, such as melanoma and severe drug eruptions. Although these conditions are less commonly encountered by nondermatologists, missed diagnosis and/or improper management can be life threatening.
The use of NAMCS data presents a few limitations. For instance, these data only represent outpatient management of skin disease. There is the potential for misdiagnosis and coding errors by the reporting physicians. The volume of data (ie, billions of office visits) prevents verification of diagnostic accuracy. The coding system requires physicians to give a diagnosis but does not provide any means by which to determine the physician’s confidence in that diagnosis. There is no code for “uncertain” or “diagnosis not determined.” Additionally, an “unspecified” diagnosis may reflect uncertainty or may simply imply that no other code accurately described the condition. Despite these limitations, the NAMCS database is a large, nationally representative survey of actual patient visits and represents some of the best data available for a study such as ours.
Conclusion
This study provides an important analysis of the most common outpatient dermatologic conditions encountered by dermatologists and nondermatologists of various specialties and offers a foundation from which to construct curricula for dermatology training tailored to individual specialties based on their needs. In the future, identification of the most common inpatient dermatologic conditions managed by each specialty also may benefit curriculum design.
- Thorpe KE, Florence CS, Joski P. Which medical conditions account for the rise in health care spending? Health Aff (Millwood). 2004;(suppl web exclusives):W4-437-445.
- Johnson ML. Defining the burden of skin disease in the United States—a historical perspective. J Investig Dermatol Symp Proc. 2004;9:108-110.
- Agency for Healthcare Research and Quality. Medical expenditure panel survey. US Department of Health & Human Services Web site. http://meps.ahrq.gov. Accessed November 17, 2014.
- Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55:490-500.
- Johnson ML. On teaching dermatology to nondermatologists. Arch Dermatol. 1994;130:850-852.
- Ramsay DL, Weary PE. Primary care in dermatology: whose role should it be? J Am Acad Dermatol. 1996;35:1005-1008.
- Kimball AB, Resneck JS Jr. The US dermatology workforce: a specialty remains in shortage. J Am Acad Dermatol. 2008;59:741-745.
- Resneck JS Jr, Kimball AB. Who else is providing care in dermatology practices? trends in the use of nonphysician clinicians. J Am Acad Dermatol. 2008;58:211-216.
- Feldman SR, Fleischer AB Jr, McConnell RC. Most common dermatologic problems identified by internists, 1990-1994. Arch Intern Med. 1998;158:726-730.
- Ahn CS, Davis SA, Debade TS, et al. Noncosmetic skin-related procedures performed in the United States: an analysis of national ambulatory medical care survey data from 1995 to 2010. Dermatol Surg. 2013;39:1912-1921.
- Antic M, Conen D, Itin PH. Teaching effects of dermatological consultations on nondermatologists in the field of internal medicine. a study of 1290 inpatients. Dermatology. 2004;208:32-37.
- Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. a review of the literature. Arch Fam Med. 1999;8:170-172.
- Fleischer AB Jr, Herbert CR, Feldman SR, et al. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000;6:1149-1156.
- Kirsner RS, Federman DG. Lack of correlation between internists’ ability in dermatology and their patterns of treating patients with skin disease. Arch Dermatol. 1996;132:1043-1046.
- McCarthy GM, Lamb GC, Russell TJ, et al. Primary care-based dermatology practice: internists need more training. J Gen Intern Med. 1991;6:52-56.
- Sellheyer K, Bergfeld WF. A retrospective biopsy study of the clinical diagnostic accuracy of common skin diseases by different specialties compared with dermatology. J Am Acad Dermatol. 2005;52:823-830.
- Medical specialties. Accreditation Council for Graduate Medical Education Web site. http://www.acgme.org/acgmeweb/tabid/368ProgramandInstitutionalGuidelines/MedicalAccreditation.aspx. Accessed November 17, 2014.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
Skin diseases are highly prevalent in the United States, affecting an estimated 1 in 3 Americans at any given time.1,2 In 2009 the direct medical costs associated with skin-related diseases, including health services and prescriptions, was approximately $22 billion; the annual total economic burden was estimated to be closer to $96 billion when factoring in the cost of lost productivity and pay for symptom relief.3,4 Effective and efficient management of skin disease is essential to minimizing cost and morbidity. Nondermatologists traditionally have diagnosed the majority of skin diseases.5,6 In particular, primary care physicians commonly manage dermatologic conditions and often are the first health care providers to encounter patients presenting with skin problems. A predicted shortage of dermatologists will likely contribute to an increase in this trend.7,8 Therefore, it is important to adequately prepare nondermatologists to evaluate and treat the skin conditions that they are most likely to encounter in their scope of practice.
Residents, particularly in primary care specialties, often have opportunities to spend 2 to 4 weeks with a dermatologist to learn about skin diseases; however, the skin conditions most often encountered by dermatologists may differ from those most often encountered by physicians in other specialties. For instance, one study demonstrated a disparity between the most common skin problems seen by dermatologists and internists.9 These dissimilarities should be recognized and addressed in curriculum content. The purpose of this study was to identify and compare the 20 most common dermatologic conditions reported by dermatologists versus those reported by nondermatologists (ie, internists, pediatricians, family physicians, emergency medicine physicians, general surgeons, otolaryngologists) from 2001 to 2010. Data also were analyzed to determine the top 20 conditions referred to dermatologists by nondermatologists as a potential indicator for areas of further improvement within medical education. With this knowledge, we hope educational curricula and self-study can be modified to reflect the current epidemiology of cutaneous diseases, thereby improving patient care.
Methods
Data from 2001 to 2010 were extracted from the National Ambulatory Medical Care Survey (NAMCS), which is an ongoing survey conducted by the National Center for Health Statistics. The NAMCS collects descriptive data regarding ambulatory visits to nonfederal office-based physicians in the United States. Participating physicians are instructed to record information about patient visits for a 1-week period, including patient demographics, insurance status, reason for visit, diagnoses, procedures, therapeutics, and referrals made at that time. Data collected for the NAMCS are entered into a multistage probability sample to produce national estimates. Within dermatology, an average of 118 dermatologists are sampled each year, and over the last 10 years, participation rates have ranged from 47% to 77%.
International Classification of Diseases, Ninth Revision, Clinical Modification codes were identified to determine the diagnoses that could be classified as dermatologic conditions. Select infectious and neoplastic disorders of the skin and mucous membrane conditions were included as well as the codes for skin diseases. Nondermatologic diagnoses and V codes were not included in the study. Data for all providers were studied to identify outpatient visits associated with the primary diagnosis of a dermatologic condition. Minor diagnoses that were considered to be subsets of major diagnoses were combined to allow better analysis of the data. For example, all tinea infections (ie, dermatophytosis of various sites, dermatomycosis unspecified) were combined into 1 diagnosis referred to as tinea because the recognition and treatment of this disease does not vary tremendously by anatomic location. Visits to dermatologists that listed nonspecific diagnoses and codes (eg, other postsurgical status [V45.89], neoplasm of uncertain behavior site unspecified [238.9]) were assumed to be for dermatologic problems.
Sampling weights were applied to obtain estimates for the number of each diagnosis made nationally. All data analyses were performed using SAS software and linear regression models were generated using SAS PROC SURVEYREG.
Data were analyzed to determine the dermatologic conditions most commonly encountered by dermatologists and nondermatologists in emergency medicine, family medicine, general surgery, internal medicine, otolaryngology, and pediatrics; these specialties include physicians who are known to commonly diagnose and treat skin diseases.10 Data also were analyzed to determine the most common conditions referred to dermatologists for treatment by nondermatologists from the selected specialties. Permission to conduct this study was obtained from the Wake Forest University institutional review board (Winston-Salem, North Carolina).
Results
From 2001 to 2010, more than 700 million outpatient visits for skin-related problems were identified, with 676.3 million visits to dermatologists, emergency medicine physicians, family practitioners, general surgeons, internists, otolaryngologists, and pediatricians. More than half (52.9%) of all skin-related visits were addressed by nondermatologists during this time. Among nondermatologists, family practitioners encountered the greatest number of skin diseases (20.5%), followed by pediatricians (11.3%), internists (9.2%), general surgeons (3.4%), otolaryngologists (1.0%), and emergency medicine physicians (0.2%)(Table 1).

Benign tumors and acne were the most common cutaneous conditions referred to dermatologists by nondermatologists (10.6% and 10.1% of all dermatology referrals, respectively), followed by nonmelanoma skin cancers (9.7%), contact dermatitis (8.8%), and actinic keratosis (7.8%)(Table 2). The top 20 conditions referred to dermatologists accounted for 83.7% of all outpatient referrals to dermatologists.
Among the diseases most frequently reported by nondermatologists, contact dermatitis was the most common (12.0%), with twice the number of visits to nondermatologists for contact dermatitis than to dermatologists (51.6 million vs 25.3 million). In terms of disease categories, infectious skin diseases (ie, bacterial [cellulitis/abscess], viral [warts, herpesvirus], fungal [tinea] and yeast [candida] etiologies) were the most common dermatologic conditions reported by nondermatologists (Table 2).

The top 20 dermatologic conditions reported by dermatologists accounted for 85.4% of all diagnoses made by dermatologists. Diseases that were among the top 20 conditions encountered by dermatologists but were not among the top 20 for nondermatologists included actinic keratosis, seborrheic keratosis, atopic dermatitis, psoriasis, alopecia, rosacea, dyschromia, seborrheic dermatitis, follicular disease, and neoplasm of uncertain behavior of skin. Additionally, 5 of the top 20 conditions encountered by dermatologists also were among the top 20 for only 1 individual nondermatologic specialty; these included atopic dermatitis (pediatrics), seborrheic dermatitis (pediatrics), psoriasis (internal medicine), rosacea (otolaryngology), and keratoderma (general surgery). Seborrheic dermatitis, psoriasis, and rosacea also were among the top 20 conditions most commonly referred to dermatologists for treatment by nondermatologists. Table 3 shows the top 20 dermatologic conditions encountered by nondermatologists by comparison.


Comment
According to NAMCS data from 2001 to 2010, visits to nondermatologists accounted for more than half of total outpatient visits for cutaneous diseases in the United States, whereas visits to dermatologists accounted for 47.1%. These findings are consistent with historical data indicating that 30% to 40% of skin-related visits are to dermatologists, and the majority of patients with skin disease are diagnosed by nondermatologists.5,6
Past data indicate that most visits to dermatologists were for evaluation of acne, infections, psoriasis, and neoplasms, whereas most visits to nondermatologists were for evaluation of epidermoid cysts, impetigo, plant dermatitis, cellulitis, and diaper rash.9 Over the last 10 years, acne has been more commonly encountered by nondermatologists, especially pediatricians. Additionally, infectious etiologies have been seen in larger volume by nondermatologists.9 Together, infectious cutaneous conditions make up nearly one-fourth of dermatologic encounters by emergency medicine physicians, internists, and family practitioners but are not within the top 20 diagnoses referred to dermatologists, which suggests that uncomplicated cases of cellulitis, herpes zoster, and other skin-related infections are largely managed by nondermatologists.5,6 Contact dermatitis, often caused by specific allergens such as detergents, solvents, and topical products, was one of the most common reported dermatologic encounters among dermatologists and nondermatologists and also was the fourth most common condition referred to dermatologists by nondermatologists for treatment; however, there may be an element of overuse of the International Classification of Diseases, Ninth Revision code, as any presumed contact dermatitis of unspecified cause can be reported under 692.9 defined as contact dermatitis and other eczema, unspecified cause. The high rate of referrals to dermatologists by nondermatologists may be for patch testing and further management. Additionally, there are no specific codes for allergic or irritant dermatitis, thus these diseases may be lumped together.
Although nearly half of all dermatologic encounters were seen by nondermatologists, dermatologists see a much larger proportion of patients with skin disease than nondermatologists and nondermatologists often have limited exposure to the field of dermatology during residency training. Studies have demonstrated differences in the abilities of dermatologists and nondermatologists to correctly diagnose common cutaneous diseases, which unsurprisingly revealed greater diagnostic accuracy demonstrated by dermatologists.11-16 The increase in acne and skin-related infections reported by nondermatologists is consistent with possible efforts to increase formal training in frequently encountered skin diseases. In one study evaluating the impact of a formal 3-week dermatology curriculum on an internal medicine department, internists demonstrated 100% accuracy in the diagnosis of acne and herpes zoster in contrast to 29% for tinea and 12% for lichen planus.5,6
The current Accreditation Council for Graduate Medical Education guidelines place little emphasis on exposure to dermatology training during residency for internists and pediatricians, as this training is not a required component of these programs.17 Two core problems with current training regarding the evaluation and management of cutaneous disease are minimal exposure to dermatologic conditions in medical school and residency and lack of consensus on the core topics that should be taught to nondermatologists.18 Exposure to dermatologic conditions through rotations in medical school has been shown to increase residents’ self-reported confidence in diagnosing and treating alopecia, cutaneous drug eruptions, warts, acne, rosacea, nonmelanoma skin cancers, sun damage, psoriasis, seborrhea, atopic dermatitis, and contact dermatitis; however, the majority of primary care residents surveyed still felt that this exposure in medical school was inadequate.19
In creating a core curriculum for dermatology training for nondermatologists, it is important to consider the dermatologic conditions that are most frequently encountered by these specialties. Our study revealed that the most commonly encountered dermatologic conditions differ among dermatologists and nondermatologists, with a fair degree of variation even among individual specialties. Failure to recognize these discrepancies has likely contributed to the challenges faced by nondermatologists in the diagnosis and management of dermatologic disease. In this study, contact dermatitis, epidermoid cysts, and skin infections were the most common dermatologic conditions encountered by nondermatologists and also were among the top skin diseases referred to dermatologists by nondermatologists. This finding suggests that nondermatologists are able to identify these conditions but have a tendency to refer approximately 10% of these patients to dermatology for further management. Clinical evaluation and medical management of these cutaneous diseases may be an important area of focus for medical school curricula, as the treatment of these diseases is within the capabilities of the nondermatologist. For example, initial management of dermatitis requires determination of the type of dermatitis (ie, essential, contact, atopic, seborrheic, stasis) and selection of an appropriate topical steroid, with referral to a dermatologist needed for questionable or refractory cases. Although a curriculum cannot be built solely on a list of the top 20 diagnoses provided here, these data may serve as a preliminary platform for medical school dermatology curriculum design. The curriculum also should include serious skin diseases, such as melanoma and severe drug eruptions. Although these conditions are less commonly encountered by nondermatologists, missed diagnosis and/or improper management can be life threatening.
The use of NAMCS data presents a few limitations. For instance, these data only represent outpatient management of skin disease. There is the potential for misdiagnosis and coding errors by the reporting physicians. The volume of data (ie, billions of office visits) prevents verification of diagnostic accuracy. The coding system requires physicians to give a diagnosis but does not provide any means by which to determine the physician’s confidence in that diagnosis. There is no code for “uncertain” or “diagnosis not determined.” Additionally, an “unspecified” diagnosis may reflect uncertainty or may simply imply that no other code accurately described the condition. Despite these limitations, the NAMCS database is a large, nationally representative survey of actual patient visits and represents some of the best data available for a study such as ours.
Conclusion
This study provides an important analysis of the most common outpatient dermatologic conditions encountered by dermatologists and nondermatologists of various specialties and offers a foundation from which to construct curricula for dermatology training tailored to individual specialties based on their needs. In the future, identification of the most common inpatient dermatologic conditions managed by each specialty also may benefit curriculum design.
Skin diseases are highly prevalent in the United States, affecting an estimated 1 in 3 Americans at any given time.1,2 In 2009 the direct medical costs associated with skin-related diseases, including health services and prescriptions, was approximately $22 billion; the annual total economic burden was estimated to be closer to $96 billion when factoring in the cost of lost productivity and pay for symptom relief.3,4 Effective and efficient management of skin disease is essential to minimizing cost and morbidity. Nondermatologists traditionally have diagnosed the majority of skin diseases.5,6 In particular, primary care physicians commonly manage dermatologic conditions and often are the first health care providers to encounter patients presenting with skin problems. A predicted shortage of dermatologists will likely contribute to an increase in this trend.7,8 Therefore, it is important to adequately prepare nondermatologists to evaluate and treat the skin conditions that they are most likely to encounter in their scope of practice.
Residents, particularly in primary care specialties, often have opportunities to spend 2 to 4 weeks with a dermatologist to learn about skin diseases; however, the skin conditions most often encountered by dermatologists may differ from those most often encountered by physicians in other specialties. For instance, one study demonstrated a disparity between the most common skin problems seen by dermatologists and internists.9 These dissimilarities should be recognized and addressed in curriculum content. The purpose of this study was to identify and compare the 20 most common dermatologic conditions reported by dermatologists versus those reported by nondermatologists (ie, internists, pediatricians, family physicians, emergency medicine physicians, general surgeons, otolaryngologists) from 2001 to 2010. Data also were analyzed to determine the top 20 conditions referred to dermatologists by nondermatologists as a potential indicator for areas of further improvement within medical education. With this knowledge, we hope educational curricula and self-study can be modified to reflect the current epidemiology of cutaneous diseases, thereby improving patient care.
Methods
Data from 2001 to 2010 were extracted from the National Ambulatory Medical Care Survey (NAMCS), which is an ongoing survey conducted by the National Center for Health Statistics. The NAMCS collects descriptive data regarding ambulatory visits to nonfederal office-based physicians in the United States. Participating physicians are instructed to record information about patient visits for a 1-week period, including patient demographics, insurance status, reason for visit, diagnoses, procedures, therapeutics, and referrals made at that time. Data collected for the NAMCS are entered into a multistage probability sample to produce national estimates. Within dermatology, an average of 118 dermatologists are sampled each year, and over the last 10 years, participation rates have ranged from 47% to 77%.
International Classification of Diseases, Ninth Revision, Clinical Modification codes were identified to determine the diagnoses that could be classified as dermatologic conditions. Select infectious and neoplastic disorders of the skin and mucous membrane conditions were included as well as the codes for skin diseases. Nondermatologic diagnoses and V codes were not included in the study. Data for all providers were studied to identify outpatient visits associated with the primary diagnosis of a dermatologic condition. Minor diagnoses that were considered to be subsets of major diagnoses were combined to allow better analysis of the data. For example, all tinea infections (ie, dermatophytosis of various sites, dermatomycosis unspecified) were combined into 1 diagnosis referred to as tinea because the recognition and treatment of this disease does not vary tremendously by anatomic location. Visits to dermatologists that listed nonspecific diagnoses and codes (eg, other postsurgical status [V45.89], neoplasm of uncertain behavior site unspecified [238.9]) were assumed to be for dermatologic problems.
Sampling weights were applied to obtain estimates for the number of each diagnosis made nationally. All data analyses were performed using SAS software and linear regression models were generated using SAS PROC SURVEYREG.
Data were analyzed to determine the dermatologic conditions most commonly encountered by dermatologists and nondermatologists in emergency medicine, family medicine, general surgery, internal medicine, otolaryngology, and pediatrics; these specialties include physicians who are known to commonly diagnose and treat skin diseases.10 Data also were analyzed to determine the most common conditions referred to dermatologists for treatment by nondermatologists from the selected specialties. Permission to conduct this study was obtained from the Wake Forest University institutional review board (Winston-Salem, North Carolina).
Results
From 2001 to 2010, more than 700 million outpatient visits for skin-related problems were identified, with 676.3 million visits to dermatologists, emergency medicine physicians, family practitioners, general surgeons, internists, otolaryngologists, and pediatricians. More than half (52.9%) of all skin-related visits were addressed by nondermatologists during this time. Among nondermatologists, family practitioners encountered the greatest number of skin diseases (20.5%), followed by pediatricians (11.3%), internists (9.2%), general surgeons (3.4%), otolaryngologists (1.0%), and emergency medicine physicians (0.2%)(Table 1).

Benign tumors and acne were the most common cutaneous conditions referred to dermatologists by nondermatologists (10.6% and 10.1% of all dermatology referrals, respectively), followed by nonmelanoma skin cancers (9.7%), contact dermatitis (8.8%), and actinic keratosis (7.8%)(Table 2). The top 20 conditions referred to dermatologists accounted for 83.7% of all outpatient referrals to dermatologists.
Among the diseases most frequently reported by nondermatologists, contact dermatitis was the most common (12.0%), with twice the number of visits to nondermatologists for contact dermatitis than to dermatologists (51.6 million vs 25.3 million). In terms of disease categories, infectious skin diseases (ie, bacterial [cellulitis/abscess], viral [warts, herpesvirus], fungal [tinea] and yeast [candida] etiologies) were the most common dermatologic conditions reported by nondermatologists (Table 2).

The top 20 dermatologic conditions reported by dermatologists accounted for 85.4% of all diagnoses made by dermatologists. Diseases that were among the top 20 conditions encountered by dermatologists but were not among the top 20 for nondermatologists included actinic keratosis, seborrheic keratosis, atopic dermatitis, psoriasis, alopecia, rosacea, dyschromia, seborrheic dermatitis, follicular disease, and neoplasm of uncertain behavior of skin. Additionally, 5 of the top 20 conditions encountered by dermatologists also were among the top 20 for only 1 individual nondermatologic specialty; these included atopic dermatitis (pediatrics), seborrheic dermatitis (pediatrics), psoriasis (internal medicine), rosacea (otolaryngology), and keratoderma (general surgery). Seborrheic dermatitis, psoriasis, and rosacea also were among the top 20 conditions most commonly referred to dermatologists for treatment by nondermatologists. Table 3 shows the top 20 dermatologic conditions encountered by nondermatologists by comparison.


Comment
According to NAMCS data from 2001 to 2010, visits to nondermatologists accounted for more than half of total outpatient visits for cutaneous diseases in the United States, whereas visits to dermatologists accounted for 47.1%. These findings are consistent with historical data indicating that 30% to 40% of skin-related visits are to dermatologists, and the majority of patients with skin disease are diagnosed by nondermatologists.5,6
Past data indicate that most visits to dermatologists were for evaluation of acne, infections, psoriasis, and neoplasms, whereas most visits to nondermatologists were for evaluation of epidermoid cysts, impetigo, plant dermatitis, cellulitis, and diaper rash.9 Over the last 10 years, acne has been more commonly encountered by nondermatologists, especially pediatricians. Additionally, infectious etiologies have been seen in larger volume by nondermatologists.9 Together, infectious cutaneous conditions make up nearly one-fourth of dermatologic encounters by emergency medicine physicians, internists, and family practitioners but are not within the top 20 diagnoses referred to dermatologists, which suggests that uncomplicated cases of cellulitis, herpes zoster, and other skin-related infections are largely managed by nondermatologists.5,6 Contact dermatitis, often caused by specific allergens such as detergents, solvents, and topical products, was one of the most common reported dermatologic encounters among dermatologists and nondermatologists and also was the fourth most common condition referred to dermatologists by nondermatologists for treatment; however, there may be an element of overuse of the International Classification of Diseases, Ninth Revision code, as any presumed contact dermatitis of unspecified cause can be reported under 692.9 defined as contact dermatitis and other eczema, unspecified cause. The high rate of referrals to dermatologists by nondermatologists may be for patch testing and further management. Additionally, there are no specific codes for allergic or irritant dermatitis, thus these diseases may be lumped together.
Although nearly half of all dermatologic encounters were seen by nondermatologists, dermatologists see a much larger proportion of patients with skin disease than nondermatologists and nondermatologists often have limited exposure to the field of dermatology during residency training. Studies have demonstrated differences in the abilities of dermatologists and nondermatologists to correctly diagnose common cutaneous diseases, which unsurprisingly revealed greater diagnostic accuracy demonstrated by dermatologists.11-16 The increase in acne and skin-related infections reported by nondermatologists is consistent with possible efforts to increase formal training in frequently encountered skin diseases. In one study evaluating the impact of a formal 3-week dermatology curriculum on an internal medicine department, internists demonstrated 100% accuracy in the diagnosis of acne and herpes zoster in contrast to 29% for tinea and 12% for lichen planus.5,6
The current Accreditation Council for Graduate Medical Education guidelines place little emphasis on exposure to dermatology training during residency for internists and pediatricians, as this training is not a required component of these programs.17 Two core problems with current training regarding the evaluation and management of cutaneous disease are minimal exposure to dermatologic conditions in medical school and residency and lack of consensus on the core topics that should be taught to nondermatologists.18 Exposure to dermatologic conditions through rotations in medical school has been shown to increase residents’ self-reported confidence in diagnosing and treating alopecia, cutaneous drug eruptions, warts, acne, rosacea, nonmelanoma skin cancers, sun damage, psoriasis, seborrhea, atopic dermatitis, and contact dermatitis; however, the majority of primary care residents surveyed still felt that this exposure in medical school was inadequate.19
In creating a core curriculum for dermatology training for nondermatologists, it is important to consider the dermatologic conditions that are most frequently encountered by these specialties. Our study revealed that the most commonly encountered dermatologic conditions differ among dermatologists and nondermatologists, with a fair degree of variation even among individual specialties. Failure to recognize these discrepancies has likely contributed to the challenges faced by nondermatologists in the diagnosis and management of dermatologic disease. In this study, contact dermatitis, epidermoid cysts, and skin infections were the most common dermatologic conditions encountered by nondermatologists and also were among the top skin diseases referred to dermatologists by nondermatologists. This finding suggests that nondermatologists are able to identify these conditions but have a tendency to refer approximately 10% of these patients to dermatology for further management. Clinical evaluation and medical management of these cutaneous diseases may be an important area of focus for medical school curricula, as the treatment of these diseases is within the capabilities of the nondermatologist. For example, initial management of dermatitis requires determination of the type of dermatitis (ie, essential, contact, atopic, seborrheic, stasis) and selection of an appropriate topical steroid, with referral to a dermatologist needed for questionable or refractory cases. Although a curriculum cannot be built solely on a list of the top 20 diagnoses provided here, these data may serve as a preliminary platform for medical school dermatology curriculum design. The curriculum also should include serious skin diseases, such as melanoma and severe drug eruptions. Although these conditions are less commonly encountered by nondermatologists, missed diagnosis and/or improper management can be life threatening.
The use of NAMCS data presents a few limitations. For instance, these data only represent outpatient management of skin disease. There is the potential for misdiagnosis and coding errors by the reporting physicians. The volume of data (ie, billions of office visits) prevents verification of diagnostic accuracy. The coding system requires physicians to give a diagnosis but does not provide any means by which to determine the physician’s confidence in that diagnosis. There is no code for “uncertain” or “diagnosis not determined.” Additionally, an “unspecified” diagnosis may reflect uncertainty or may simply imply that no other code accurately described the condition. Despite these limitations, the NAMCS database is a large, nationally representative survey of actual patient visits and represents some of the best data available for a study such as ours.
Conclusion
This study provides an important analysis of the most common outpatient dermatologic conditions encountered by dermatologists and nondermatologists of various specialties and offers a foundation from which to construct curricula for dermatology training tailored to individual specialties based on their needs. In the future, identification of the most common inpatient dermatologic conditions managed by each specialty also may benefit curriculum design.
- Thorpe KE, Florence CS, Joski P. Which medical conditions account for the rise in health care spending? Health Aff (Millwood). 2004;(suppl web exclusives):W4-437-445.
- Johnson ML. Defining the burden of skin disease in the United States—a historical perspective. J Investig Dermatol Symp Proc. 2004;9:108-110.
- Agency for Healthcare Research and Quality. Medical expenditure panel survey. US Department of Health & Human Services Web site. http://meps.ahrq.gov. Accessed November 17, 2014.
- Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55:490-500.
- Johnson ML. On teaching dermatology to nondermatologists. Arch Dermatol. 1994;130:850-852.
- Ramsay DL, Weary PE. Primary care in dermatology: whose role should it be? J Am Acad Dermatol. 1996;35:1005-1008.
- Kimball AB, Resneck JS Jr. The US dermatology workforce: a specialty remains in shortage. J Am Acad Dermatol. 2008;59:741-745.
- Resneck JS Jr, Kimball AB. Who else is providing care in dermatology practices? trends in the use of nonphysician clinicians. J Am Acad Dermatol. 2008;58:211-216.
- Feldman SR, Fleischer AB Jr, McConnell RC. Most common dermatologic problems identified by internists, 1990-1994. Arch Intern Med. 1998;158:726-730.
- Ahn CS, Davis SA, Debade TS, et al. Noncosmetic skin-related procedures performed in the United States: an analysis of national ambulatory medical care survey data from 1995 to 2010. Dermatol Surg. 2013;39:1912-1921.
- Antic M, Conen D, Itin PH. Teaching effects of dermatological consultations on nondermatologists in the field of internal medicine. a study of 1290 inpatients. Dermatology. 2004;208:32-37.
- Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. a review of the literature. Arch Fam Med. 1999;8:170-172.
- Fleischer AB Jr, Herbert CR, Feldman SR, et al. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000;6:1149-1156.
- Kirsner RS, Federman DG. Lack of correlation between internists’ ability in dermatology and their patterns of treating patients with skin disease. Arch Dermatol. 1996;132:1043-1046.
- McCarthy GM, Lamb GC, Russell TJ, et al. Primary care-based dermatology practice: internists need more training. J Gen Intern Med. 1991;6:52-56.
- Sellheyer K, Bergfeld WF. A retrospective biopsy study of the clinical diagnostic accuracy of common skin diseases by different specialties compared with dermatology. J Am Acad Dermatol. 2005;52:823-830.
- Medical specialties. Accreditation Council for Graduate Medical Education Web site. http://www.acgme.org/acgmeweb/tabid/368ProgramandInstitutionalGuidelines/MedicalAccreditation.aspx. Accessed November 17, 2014.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
- Thorpe KE, Florence CS, Joski P. Which medical conditions account for the rise in health care spending? Health Aff (Millwood). 2004;(suppl web exclusives):W4-437-445.
- Johnson ML. Defining the burden of skin disease in the United States—a historical perspective. J Investig Dermatol Symp Proc. 2004;9:108-110.
- Agency for Healthcare Research and Quality. Medical expenditure panel survey. US Department of Health & Human Services Web site. http://meps.ahrq.gov. Accessed November 17, 2014.
- Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55:490-500.
- Johnson ML. On teaching dermatology to nondermatologists. Arch Dermatol. 1994;130:850-852.
- Ramsay DL, Weary PE. Primary care in dermatology: whose role should it be? J Am Acad Dermatol. 1996;35:1005-1008.
- Kimball AB, Resneck JS Jr. The US dermatology workforce: a specialty remains in shortage. J Am Acad Dermatol. 2008;59:741-745.
- Resneck JS Jr, Kimball AB. Who else is providing care in dermatology practices? trends in the use of nonphysician clinicians. J Am Acad Dermatol. 2008;58:211-216.
- Feldman SR, Fleischer AB Jr, McConnell RC. Most common dermatologic problems identified by internists, 1990-1994. Arch Intern Med. 1998;158:726-730.
- Ahn CS, Davis SA, Debade TS, et al. Noncosmetic skin-related procedures performed in the United States: an analysis of national ambulatory medical care survey data from 1995 to 2010. Dermatol Surg. 2013;39:1912-1921.
- Antic M, Conen D, Itin PH. Teaching effects of dermatological consultations on nondermatologists in the field of internal medicine. a study of 1290 inpatients. Dermatology. 2004;208:32-37.
- Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. a review of the literature. Arch Fam Med. 1999;8:170-172.
- Fleischer AB Jr, Herbert CR, Feldman SR, et al. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000;6:1149-1156.
- Kirsner RS, Federman DG. Lack of correlation between internists’ ability in dermatology and their patterns of treating patients with skin disease. Arch Dermatol. 1996;132:1043-1046.
- McCarthy GM, Lamb GC, Russell TJ, et al. Primary care-based dermatology practice: internists need more training. J Gen Intern Med. 1991;6:52-56.
- Sellheyer K, Bergfeld WF. A retrospective biopsy study of the clinical diagnostic accuracy of common skin diseases by different specialties compared with dermatology. J Am Acad Dermatol. 2005;52:823-830.
- Medical specialties. Accreditation Council for Graduate Medical Education Web site. http://www.acgme.org/acgmeweb/tabid/368ProgramandInstitutionalGuidelines/MedicalAccreditation.aspx. Accessed November 17, 2014.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
Practice Points
- Approximately half of skin-related visits are to nondermatologists, such as family medicine physicians, pediatricians, and internists.
- Skin conditions that most frequently present to nondermatologists are different from those seen by dermatologists.
- Education efforts in nondermatology specialties should be targeted toward the common skin diseases that present to these specialties to maximize the yield of medical education and improve diagnostic accuracy and patient outcomes.
Biopsy can underestimate diversity, aggressiveness of basal cell carcinomas
SAN DIEGO – Histology of basal cell carcinomas removed by Mohs micrographic surgery showed that presurgical biopsies had not revealed all tumor subtypes in 64% of cases, and had underestimated the aggressiveness of the tumors 24% of the time, according to data from a large, multicenter, retrospective study.
“Unfortunately, while cheap and cost-effective, biopsies are a subsample of the full malignancy,” said Dr. Murad Alam, professor of dermatology, otolaryngology, and surgery at Northwestern University in Chicago. “Skin biopsy of basal cell carcinoma [BCC] may fail to detect all BCC subtypes, and as such may underestimate the aggressiveness of an individual BCC tumor.”
Basal cell carcinoma is the most common skin cancer worldwide, and can broadly be grouped into aggressive and indolent types, Dr. Alam said at the annual meeting of the American Society for Dermatologic Surgery. But tumors often show mixed histology, and cancer treatment needs to target the most aggressive subtype present in the tumor, he added. Results of past studies suggested that biopsies of BCCs could miss tumor subtypes, but the current research is the first large, multicenter study to confirm these findings, he and his associates said.
For the study, the investigators compared biopsy reports and microscopic slides of Mohs micrographic surgery (MMS) specimens from 871 consecutive cases of BCC treated at three hospitals in Illinois from 2013 to 2014. Patients first underwent biopsies, followed by complete excision of their tumors during MMS. Almost 59% of patients were male, and tumors were most commonly removed from the nose or cheek. In all, 78% of biopsies were obtained by the shave technique, but punch and excisional biopsies also were performed, the researchers noted.
Using standard definitions of BCC subtypes, the investigators compared levels of concordance between biopsy and MMS histology findings, Dr. Alam said. They also grouped tumor specimens as high risk (that is, infiltrative, morpheic, micronodular, basosquamous) or low risk (superficial or nodular), and determined whether tumor biopsy and MMS histology yielded the same or discordant risk assessments, he added.
Biopsies identified only 18% of tumors as being of mixed histology, compared with 57% of MMS specimens, said Dr. Alam. Biopsy results matched MMS histologies in only 31% of cases, while in 64% of cases, the MMS specimen yielded more tumor subtypes than the biopsy specimen. The researchers noted that biopsy yielded more subtypes than did MMS in 4% of cases, and that MMS and biopsy subtypes were fully discordant in only four cases.
Dr. Alam and his associates declared no external funding sources or conflicts of interest.
SAN DIEGO – Histology of basal cell carcinomas removed by Mohs micrographic surgery showed that presurgical biopsies had not revealed all tumor subtypes in 64% of cases, and had underestimated the aggressiveness of the tumors 24% of the time, according to data from a large, multicenter, retrospective study.
“Unfortunately, while cheap and cost-effective, biopsies are a subsample of the full malignancy,” said Dr. Murad Alam, professor of dermatology, otolaryngology, and surgery at Northwestern University in Chicago. “Skin biopsy of basal cell carcinoma [BCC] may fail to detect all BCC subtypes, and as such may underestimate the aggressiveness of an individual BCC tumor.”
Basal cell carcinoma is the most common skin cancer worldwide, and can broadly be grouped into aggressive and indolent types, Dr. Alam said at the annual meeting of the American Society for Dermatologic Surgery. But tumors often show mixed histology, and cancer treatment needs to target the most aggressive subtype present in the tumor, he added. Results of past studies suggested that biopsies of BCCs could miss tumor subtypes, but the current research is the first large, multicenter study to confirm these findings, he and his associates said.
For the study, the investigators compared biopsy reports and microscopic slides of Mohs micrographic surgery (MMS) specimens from 871 consecutive cases of BCC treated at three hospitals in Illinois from 2013 to 2014. Patients first underwent biopsies, followed by complete excision of their tumors during MMS. Almost 59% of patients were male, and tumors were most commonly removed from the nose or cheek. In all, 78% of biopsies were obtained by the shave technique, but punch and excisional biopsies also were performed, the researchers noted.
Using standard definitions of BCC subtypes, the investigators compared levels of concordance between biopsy and MMS histology findings, Dr. Alam said. They also grouped tumor specimens as high risk (that is, infiltrative, morpheic, micronodular, basosquamous) or low risk (superficial or nodular), and determined whether tumor biopsy and MMS histology yielded the same or discordant risk assessments, he added.
Biopsies identified only 18% of tumors as being of mixed histology, compared with 57% of MMS specimens, said Dr. Alam. Biopsy results matched MMS histologies in only 31% of cases, while in 64% of cases, the MMS specimen yielded more tumor subtypes than the biopsy specimen. The researchers noted that biopsy yielded more subtypes than did MMS in 4% of cases, and that MMS and biopsy subtypes were fully discordant in only four cases.
Dr. Alam and his associates declared no external funding sources or conflicts of interest.
SAN DIEGO – Histology of basal cell carcinomas removed by Mohs micrographic surgery showed that presurgical biopsies had not revealed all tumor subtypes in 64% of cases, and had underestimated the aggressiveness of the tumors 24% of the time, according to data from a large, multicenter, retrospective study.
“Unfortunately, while cheap and cost-effective, biopsies are a subsample of the full malignancy,” said Dr. Murad Alam, professor of dermatology, otolaryngology, and surgery at Northwestern University in Chicago. “Skin biopsy of basal cell carcinoma [BCC] may fail to detect all BCC subtypes, and as such may underestimate the aggressiveness of an individual BCC tumor.”
Basal cell carcinoma is the most common skin cancer worldwide, and can broadly be grouped into aggressive and indolent types, Dr. Alam said at the annual meeting of the American Society for Dermatologic Surgery. But tumors often show mixed histology, and cancer treatment needs to target the most aggressive subtype present in the tumor, he added. Results of past studies suggested that biopsies of BCCs could miss tumor subtypes, but the current research is the first large, multicenter study to confirm these findings, he and his associates said.
For the study, the investigators compared biopsy reports and microscopic slides of Mohs micrographic surgery (MMS) specimens from 871 consecutive cases of BCC treated at three hospitals in Illinois from 2013 to 2014. Patients first underwent biopsies, followed by complete excision of their tumors during MMS. Almost 59% of patients were male, and tumors were most commonly removed from the nose or cheek. In all, 78% of biopsies were obtained by the shave technique, but punch and excisional biopsies also were performed, the researchers noted.
Using standard definitions of BCC subtypes, the investigators compared levels of concordance between biopsy and MMS histology findings, Dr. Alam said. They also grouped tumor specimens as high risk (that is, infiltrative, morpheic, micronodular, basosquamous) or low risk (superficial or nodular), and determined whether tumor biopsy and MMS histology yielded the same or discordant risk assessments, he added.
Biopsies identified only 18% of tumors as being of mixed histology, compared with 57% of MMS specimens, said Dr. Alam. Biopsy results matched MMS histologies in only 31% of cases, while in 64% of cases, the MMS specimen yielded more tumor subtypes than the biopsy specimen. The researchers noted that biopsy yielded more subtypes than did MMS in 4% of cases, and that MMS and biopsy subtypes were fully discordant in only four cases.
Dr. Alam and his associates declared no external funding sources or conflicts of interest.
Key clinical point: Definitive excision by Mohs micrographic surgery reveals more information about basal cell carcinoma subtypes and tumor behavior than does biopsy.
Major finding: Compared with Mohs specimens, biopsy underestimated the diversity of tumor subtypes in 64% of cases, and underestimated tumor aggressiveness in 24% of cases.
Data source: Multicenter retrospective study of 871 basal cell carcinomas that were biopsied and then removed by Mohs micrographic surgery.
Disclosures: The investigators declared no external funding sources or conflicts of interest.
Vismodegib offers promise for basal cell carcinoma, with caveats
SAN DIEGO – Patients treated with vismodegib for locally advanced or metastatic basal cell carcinoma went a median of 15 months before their disease progressed or they stopped treatment because of side effects, according to a 30-month update of the pivotal ERIVANCE basal cell carcinoma study.
Median progression-free survival on the first-in-class oral hedgehog-pathway inhibitor was 9 months, reported Dr. Seaver Soon at the annual meeting of the American Society for Dermatologic Surgery.
Data from two other trials of vismodegib resemble results from ERIVANCE, added Dr. Soon, a dermatologist in private practice in La Jolla, Calif. An expanded access study (J. Am. Acad. Dermatol 2014;70:60-9) of 119 patients with advanced basal cell carcinoma (BCC) reported comparable objective response rates (46.4% for patients with locally advanced BCC and 30.8% for patients with metastatic disease), and an interim analysis of data from the STEVIE trial had findings that were “very similar” to ERIVANCE, he said.
Thus far, vismodegib “offers a hope in treating otherwise difficult to manage, unresectable basal cell carcinoma tumors,” said Dr. Iren Kossintseva, a dermatologist in Vancouver, B.C. But the drug “may not be as tissue sparing as promised, she added. In a patient with chronic lymphocytic leukemia who had a large BCC on his lower eyelid and cheek, 7.5 months of vismodegib reduced the exophyticity and erosiveness of the tumor, but “likely did not substantially reduce the overall extent of necessary reconstruction,” she reported.
Vismodegib can cause potentially severe side effects. All seven patients who Dr. Kossintseva treated with 150 mg vismodegib per day during 2013-2014 developed “notable” adverse effects – including polycyclic rash, sensory and motor problems within the tumor area, bilateral edema of the lower limbs, congestive heart failure, and renal failure that has been slow to improve after stopping vismodegib, she said. “These are unique patients, and it’s often an uphill battle with these patients,” she added.
Tumors also can exhibit primary and secondary resistance to vismodegib, Dr. Soon noted. Studies have shown primary resistance characterized by tumor progression after as little as 2 months of treatment (Mol. Oncol. 2014; S1574-7891:00216-6) while secondary (or acquired) resistance occurs after an initial response to treatment and is linked to a mutation that interferes with drug binding, he said. Acquired resistance typically occurs when patients have been on vismodegib for about a year, Dr. Soon added. “Concurrent treatment with an alternative smoothened inhibitor, such as itraconazole, and downstream target inhibitors may overcome resistance,” he said. Dr. Kossintseva declared no conflicts of interest. Dr. Soon reported receiving honoraria and research grants from Genentech, the maker of vismodegib.
SAN DIEGO – Patients treated with vismodegib for locally advanced or metastatic basal cell carcinoma went a median of 15 months before their disease progressed or they stopped treatment because of side effects, according to a 30-month update of the pivotal ERIVANCE basal cell carcinoma study.
Median progression-free survival on the first-in-class oral hedgehog-pathway inhibitor was 9 months, reported Dr. Seaver Soon at the annual meeting of the American Society for Dermatologic Surgery.
Data from two other trials of vismodegib resemble results from ERIVANCE, added Dr. Soon, a dermatologist in private practice in La Jolla, Calif. An expanded access study (J. Am. Acad. Dermatol 2014;70:60-9) of 119 patients with advanced basal cell carcinoma (BCC) reported comparable objective response rates (46.4% for patients with locally advanced BCC and 30.8% for patients with metastatic disease), and an interim analysis of data from the STEVIE trial had findings that were “very similar” to ERIVANCE, he said.
Thus far, vismodegib “offers a hope in treating otherwise difficult to manage, unresectable basal cell carcinoma tumors,” said Dr. Iren Kossintseva, a dermatologist in Vancouver, B.C. But the drug “may not be as tissue sparing as promised, she added. In a patient with chronic lymphocytic leukemia who had a large BCC on his lower eyelid and cheek, 7.5 months of vismodegib reduced the exophyticity and erosiveness of the tumor, but “likely did not substantially reduce the overall extent of necessary reconstruction,” she reported.
Vismodegib can cause potentially severe side effects. All seven patients who Dr. Kossintseva treated with 150 mg vismodegib per day during 2013-2014 developed “notable” adverse effects – including polycyclic rash, sensory and motor problems within the tumor area, bilateral edema of the lower limbs, congestive heart failure, and renal failure that has been slow to improve after stopping vismodegib, she said. “These are unique patients, and it’s often an uphill battle with these patients,” she added.
Tumors also can exhibit primary and secondary resistance to vismodegib, Dr. Soon noted. Studies have shown primary resistance characterized by tumor progression after as little as 2 months of treatment (Mol. Oncol. 2014; S1574-7891:00216-6) while secondary (or acquired) resistance occurs after an initial response to treatment and is linked to a mutation that interferes with drug binding, he said. Acquired resistance typically occurs when patients have been on vismodegib for about a year, Dr. Soon added. “Concurrent treatment with an alternative smoothened inhibitor, such as itraconazole, and downstream target inhibitors may overcome resistance,” he said. Dr. Kossintseva declared no conflicts of interest. Dr. Soon reported receiving honoraria and research grants from Genentech, the maker of vismodegib.
SAN DIEGO – Patients treated with vismodegib for locally advanced or metastatic basal cell carcinoma went a median of 15 months before their disease progressed or they stopped treatment because of side effects, according to a 30-month update of the pivotal ERIVANCE basal cell carcinoma study.
Median progression-free survival on the first-in-class oral hedgehog-pathway inhibitor was 9 months, reported Dr. Seaver Soon at the annual meeting of the American Society for Dermatologic Surgery.
Data from two other trials of vismodegib resemble results from ERIVANCE, added Dr. Soon, a dermatologist in private practice in La Jolla, Calif. An expanded access study (J. Am. Acad. Dermatol 2014;70:60-9) of 119 patients with advanced basal cell carcinoma (BCC) reported comparable objective response rates (46.4% for patients with locally advanced BCC and 30.8% for patients with metastatic disease), and an interim analysis of data from the STEVIE trial had findings that were “very similar” to ERIVANCE, he said.
Thus far, vismodegib “offers a hope in treating otherwise difficult to manage, unresectable basal cell carcinoma tumors,” said Dr. Iren Kossintseva, a dermatologist in Vancouver, B.C. But the drug “may not be as tissue sparing as promised, she added. In a patient with chronic lymphocytic leukemia who had a large BCC on his lower eyelid and cheek, 7.5 months of vismodegib reduced the exophyticity and erosiveness of the tumor, but “likely did not substantially reduce the overall extent of necessary reconstruction,” she reported.
Vismodegib can cause potentially severe side effects. All seven patients who Dr. Kossintseva treated with 150 mg vismodegib per day during 2013-2014 developed “notable” adverse effects – including polycyclic rash, sensory and motor problems within the tumor area, bilateral edema of the lower limbs, congestive heart failure, and renal failure that has been slow to improve after stopping vismodegib, she said. “These are unique patients, and it’s often an uphill battle with these patients,” she added.
Tumors also can exhibit primary and secondary resistance to vismodegib, Dr. Soon noted. Studies have shown primary resistance characterized by tumor progression after as little as 2 months of treatment (Mol. Oncol. 2014; S1574-7891:00216-6) while secondary (or acquired) resistance occurs after an initial response to treatment and is linked to a mutation that interferes with drug binding, he said. Acquired resistance typically occurs when patients have been on vismodegib for about a year, Dr. Soon added. “Concurrent treatment with an alternative smoothened inhibitor, such as itraconazole, and downstream target inhibitors may overcome resistance,” he said. Dr. Kossintseva declared no conflicts of interest. Dr. Soon reported receiving honoraria and research grants from Genentech, the maker of vismodegib.
Old Concept, New Drug: Topical Application of Systemic Antineoplastic Agent to Treat Skin Cancer

In a June 29 article published online in Molecular Carcinogenesis, Fenton et al demonstrated that topical dasatinib treatment of UVB-exposed SKH1 hairless mice reduced the total tumor burden (ie, benign tumors, atypical benign tumors, squamous cell carcinomas) per mouse.
Dasatinib is a tyrosine kinase inhibitor currently used to treat imatinib-resistant chronic myeloid leukemia and Philadelphia chromosome positive acute lymphoblastic leukemia. Its mechanism of action is to block the activity of tyrosine kinases by attaching to their adenosine triphosphate–binding site. It can inhibit the activity of the following tyrosine kinases: Src family kinases (SFK), break point cluster region-Abelson (Bcr-Abl), Ephrin type-A receptor 2 (EphA2), platelet-derived growth factor receptor, and mast/stem cell factor receptor (also called CD117 or c-Kit).
Src family kinases are associated with transformation of cells and progression of cancer. Elevated Src family kinases activity is present in the majority of human carcinomas.
Fyn, a nonreceptor tyrosine kinase, is a member of the Src family kinases. Cell growth, cell migration, and protein kinase B (Akt)–mediated inhibition of apoptosis are influenced by Fyn activity. In addition, Fyn activity is overexpressed in cutaneous squamous cell carcinoma.
In conclusion, dasatinib—an Src family kinases inhibitor—was able to inhibit Fyn activity and thereby reduce UV-induced skin carcinogenesis.
What’s the issue?
In 1962, Falkson and Schulz (Br J Dermatol. 1962;74:229-236) noted not only inflammation and subsequent resolution of actinic keratoses after exposure to sunlight in a woman with colon cancer being treated with systemic 5-fluorouracil but also several other patients whose keratoses were seen to disappear during therapy without preceding erythema. Omura and Torre (JAMA. 1969;208:150-151) confirmed these observations in a woman with breast cancer whose actinic keratoses became inflamed after receiving intravenous 5-fluorouracil and subsequently faded. The route of drug administration was modified and 5-fluorouracil was applied topically. Today topical 5-fluorouracil is still used for the treatment of actinic keratoses.
Topical application of nitrogen mustard is used in the treatment of cutaneous T-cell lymphoma. In addition, intralesional administration of systemic antineoplastic agents has been used to treat cutaneous neoplasms: methotrexate for keratoacanthomas and rituximab for primary cutaneous B-cell lymphomas.
The Fenton et al study suggests that topical dasatinib may be a potential therapeutic intervention for the treatment of cutaneous squamous cell carcinoma. The investigators not only demonstrate laboratory data from mouse studies but also provide a potential molecular mechanism for drug-associated tumor suppression. Indeed, dasatinib solution, dasatinib cream, or both may be the next innovative therapy for the suppression of cutaneous squamous cell carcinoma in organ transplant and immunocompromised patients and for the potential management of this skin cancer in immunocompetent individuals. What do you think?

In a June 29 article published online in Molecular Carcinogenesis, Fenton et al demonstrated that topical dasatinib treatment of UVB-exposed SKH1 hairless mice reduced the total tumor burden (ie, benign tumors, atypical benign tumors, squamous cell carcinomas) per mouse.
Dasatinib is a tyrosine kinase inhibitor currently used to treat imatinib-resistant chronic myeloid leukemia and Philadelphia chromosome positive acute lymphoblastic leukemia. Its mechanism of action is to block the activity of tyrosine kinases by attaching to their adenosine triphosphate–binding site. It can inhibit the activity of the following tyrosine kinases: Src family kinases (SFK), break point cluster region-Abelson (Bcr-Abl), Ephrin type-A receptor 2 (EphA2), platelet-derived growth factor receptor, and mast/stem cell factor receptor (also called CD117 or c-Kit).
Src family kinases are associated with transformation of cells and progression of cancer. Elevated Src family kinases activity is present in the majority of human carcinomas.
Fyn, a nonreceptor tyrosine kinase, is a member of the Src family kinases. Cell growth, cell migration, and protein kinase B (Akt)–mediated inhibition of apoptosis are influenced by Fyn activity. In addition, Fyn activity is overexpressed in cutaneous squamous cell carcinoma.
In conclusion, dasatinib—an Src family kinases inhibitor—was able to inhibit Fyn activity and thereby reduce UV-induced skin carcinogenesis.
What’s the issue?
In 1962, Falkson and Schulz (Br J Dermatol. 1962;74:229-236) noted not only inflammation and subsequent resolution of actinic keratoses after exposure to sunlight in a woman with colon cancer being treated with systemic 5-fluorouracil but also several other patients whose keratoses were seen to disappear during therapy without preceding erythema. Omura and Torre (JAMA. 1969;208:150-151) confirmed these observations in a woman with breast cancer whose actinic keratoses became inflamed after receiving intravenous 5-fluorouracil and subsequently faded. The route of drug administration was modified and 5-fluorouracil was applied topically. Today topical 5-fluorouracil is still used for the treatment of actinic keratoses.
Topical application of nitrogen mustard is used in the treatment of cutaneous T-cell lymphoma. In addition, intralesional administration of systemic antineoplastic agents has been used to treat cutaneous neoplasms: methotrexate for keratoacanthomas and rituximab for primary cutaneous B-cell lymphomas.
The Fenton et al study suggests that topical dasatinib may be a potential therapeutic intervention for the treatment of cutaneous squamous cell carcinoma. The investigators not only demonstrate laboratory data from mouse studies but also provide a potential molecular mechanism for drug-associated tumor suppression. Indeed, dasatinib solution, dasatinib cream, or both may be the next innovative therapy for the suppression of cutaneous squamous cell carcinoma in organ transplant and immunocompromised patients and for the potential management of this skin cancer in immunocompetent individuals. What do you think?

In a June 29 article published online in Molecular Carcinogenesis, Fenton et al demonstrated that topical dasatinib treatment of UVB-exposed SKH1 hairless mice reduced the total tumor burden (ie, benign tumors, atypical benign tumors, squamous cell carcinomas) per mouse.
Dasatinib is a tyrosine kinase inhibitor currently used to treat imatinib-resistant chronic myeloid leukemia and Philadelphia chromosome positive acute lymphoblastic leukemia. Its mechanism of action is to block the activity of tyrosine kinases by attaching to their adenosine triphosphate–binding site. It can inhibit the activity of the following tyrosine kinases: Src family kinases (SFK), break point cluster region-Abelson (Bcr-Abl), Ephrin type-A receptor 2 (EphA2), platelet-derived growth factor receptor, and mast/stem cell factor receptor (also called CD117 or c-Kit).
Src family kinases are associated with transformation of cells and progression of cancer. Elevated Src family kinases activity is present in the majority of human carcinomas.
Fyn, a nonreceptor tyrosine kinase, is a member of the Src family kinases. Cell growth, cell migration, and protein kinase B (Akt)–mediated inhibition of apoptosis are influenced by Fyn activity. In addition, Fyn activity is overexpressed in cutaneous squamous cell carcinoma.
In conclusion, dasatinib—an Src family kinases inhibitor—was able to inhibit Fyn activity and thereby reduce UV-induced skin carcinogenesis.
What’s the issue?
In 1962, Falkson and Schulz (Br J Dermatol. 1962;74:229-236) noted not only inflammation and subsequent resolution of actinic keratoses after exposure to sunlight in a woman with colon cancer being treated with systemic 5-fluorouracil but also several other patients whose keratoses were seen to disappear during therapy without preceding erythema. Omura and Torre (JAMA. 1969;208:150-151) confirmed these observations in a woman with breast cancer whose actinic keratoses became inflamed after receiving intravenous 5-fluorouracil and subsequently faded. The route of drug administration was modified and 5-fluorouracil was applied topically. Today topical 5-fluorouracil is still used for the treatment of actinic keratoses.
Topical application of nitrogen mustard is used in the treatment of cutaneous T-cell lymphoma. In addition, intralesional administration of systemic antineoplastic agents has been used to treat cutaneous neoplasms: methotrexate for keratoacanthomas and rituximab for primary cutaneous B-cell lymphomas.
The Fenton et al study suggests that topical dasatinib may be a potential therapeutic intervention for the treatment of cutaneous squamous cell carcinoma. The investigators not only demonstrate laboratory data from mouse studies but also provide a potential molecular mechanism for drug-associated tumor suppression. Indeed, dasatinib solution, dasatinib cream, or both may be the next innovative therapy for the suppression of cutaneous squamous cell carcinoma in organ transplant and immunocompromised patients and for the potential management of this skin cancer in immunocompetent individuals. What do you think?
What Is Your Diagnosis? Cutaneous B-cell Lymphoma
A 59-year-old white man presented with 2 large erythematous lesions on the right side of the chest wall that had gradually progressed over the last 1.5 years. The patient denied any fever, night sweats, fatigue, unintentional weight loss, or loss of appetite. Physical examination revealed 2 large, well-circumscribed, nearly contiguous, firm, erythematous tumors. One tumor measured 7.5×4.5 cm and the other measured 4×3.5 cm.
The Diagnosis: Cutaneous B-cell Lymphoma
Biopsies from the right side of the chest wall (Figure 1) revealed an atypical dense and diffuse lymphocytic infiltrate throughout the dermis. There was extensive crush artifact throughout the specimen. However, the findings were consistent with cutaneous B-cell lymphoma (CBCL), and the diffuse large B-cell type was favored (Figure 2). Atypical lymphocytes stained positively for antibodies against CD20 (Figure 3), CD79a, and BCL-6, and stained negatively for antibodies against MUM-1 and BCL-2. Although flow cytometry revealed no definitive immunophenotypic lymphoma population, polymerase chain reaction analysis revealed a monoclonal immunoglobulin heavy chain gene rearrangement. Computed tomography (CT) scans of the chest, abdomen, and pelvis were unremarkable. A preliminary diagnosis of primary CBCL (PCBCL) was formulated. Diffuse large B-cell lymphoma (DLBCL) and follicle center lymphoma subtypes were each considered, which triggered further workup to rule out systemic involvement.
|
|
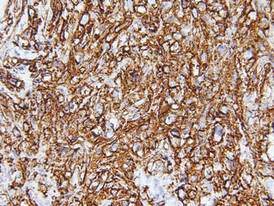
|
|
A bone marrow biopsy from the posterior iliac crest revealed normocellular bone marrow with normal trilineage hematopoiesis. However, whole-body staging with positron emission tomography (PET)–CT scanning revealed osseous disease in the left proximal humerus (Figure 4) as well as a slightly hypermetabolic right axillary lymph node. Magnetic resonance imaging of the brain showed no evidence of intracranial disease. Because of the apparent systemic involvement, stage IV non-Hodgkin lymphoma (DLBCL) became the new suspected diagnosis. The patient was started on the first of 6 cycles of chemotherapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), and the skin lesions quickly dissipated and flattened. A faint pink discoloration remained over a slightly indented area. A repeat PET-CT scan following 4 cycles of R-CHOP chemotherapy also confirmed a complete response to therapy.
In general, CBCL tends to affect adults and presents as relatively firm and plum-colored papules, nodules, tumors, or plaques, which can be either fast or slow growing. Cutaneous B-cell lymphoma may be primary or secondary to systemic involvement. Primary CBCL refers to a group of non-Hodgkin lymphomas that initially present in the skin with no evidence of extracutaneous involvement at the time of diagnosis.1,2 Secondary CBCL (SCBCL) refers to cutaneous disease that occurs secondary to systemic B-cell lymphoma. Detecting systemic involvement and distinguishing between PCBCL and SCBCL is valuable in determining prognosis and therapeutic options, as subtypes of PCBCL often have an improved prognosis and may be treated with local irradiation.
The initial staging techniques that are preferred for cutaneous lymphomas have been debated.3-5 For cutaneous lymphomas, except mycosis fungoides and Sézary syndrome, the International Society for Cutaneous Lymphomas and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer recommends obtaining a complete blood cell count with differential; complete metabolic studies including lactate dehydrogenase; and imaging studies of the chest, abdomen, and pelvis. Bone marrow biopsies and imaging studies of the neck or whole-body PET-CT scanning also may be useful depending on the clinical scenario.3 Although a more limited workup may be sufficient for PCBCLs such as primary cutaneous marginal zone lymphoma,5 a bone marrow biopsy is recommended for cases of primary cutaneous DLBCL (leg type).3 Senff et al5 supported the use of a bone marrow biopsy in the evaluation of follicle center lymphomas first presenting in the skin, though this method is controversial. In our patient, the laboratory results; bone marrow biopsy; and CT scan of the chest, abdomen, and pelvis failed to suggest extracutaneous disease, while the PET-CT scan revealed systemic involvement.
The differential diagnosis of CBCL includes cutaneous lymphoid hyperplasia (pseudolymphoma), which may be the result of insults such as arthropod bites, stings, vaccinations, or trauma. The clinical presentation, histology, and results of molecular studies and immunohistochemistry are essential in differentiating benign versus malignant processes.6 Lymphomas are expected to be larger and more persistent than benign processes, demonstrating an atypical lymphocytic infiltrate and monoclonality; immunohistochemistry will aid in the distinction between B-cell and T-cell processes and can delineate the type of B-cell lymphoma. Histology for CBCL typically reveals an atypical lymphocytic infiltrate showing a CD20+ and CD79a+ immunophenotype. Staining for antibodies against BCL-2, BCL-6, CD10, and MUM-1 also plays an important role in the diagnosis of cutaneous lymphoma and determining where the lesion(s) falls within the classification schemes. For example, to differentiate between primary cutaneous lymphoma subtypes, BCL-2 negativity and BCL-6 positivity in the context of a CD20+ and CD79a+ immunophenotype supports a follicle center lymphoma or a DLBCL (non–leg type). By contrast, CD20, CD79a, BCL-2, and MUM-1 positivity would favor a DLBCL (leg type).7
The natural history and therapeutic options differ greatly between subtypes of CBCL. For example, the prognosis of primary cutaneous follicle center lymphoma is generally favorable with a 5-year disease-specific survival rate of roughly 95%, and radiation therapy is recommended as a first-line therapy for localized disease.2,8 Conversely, primary cutaneous DLBCL (leg type) frequently spreads to extracutaneous sites8 and carries a much lower estimated 5-year disease-specific survival rate of 55%.2 Chemotherapy with R-CHOP is typically included in initial therapy for primary cutaneous DLBCL (leg type).8 The prognosis of systemic B-cell lymphomas also is highly variable and may depend on the type of B-cell lymphoma, the stage of disease at diagnosis, histologic and immunologic characteristics, and the therapy received. Wright et al9 reported that patients with systemic germinal center B cell–like DLBCL had a 5-year survival rate of 62%, whereas patients with activated B cell–like variants of DLBCL had a 5-year survival rate of 26%. Expression of CD40 may be a favorable prognostic factor following treatment with systemic chemotherapy in patients with DLBCL,10 whereas FOXP1 protein overexpression is correlated with poor disease-specific survival in certain DLBCL phenotypes.11
Although it is uncertain whether the cutaneous lesions preceded systemic disease in our patient, the cutaneous lesions could be arbitrarily classified as secondary because extracutaneous disease was discovered within 6 months of the initial diagnosis.1 However, classifying the CBCL as primary or secondary did not alter the course of treatment in our patient, as the presumed systemic disease necessitated treatment with systemic chemotherapy; both PCBCLs that develop systemic involvement and SCBCLs (primary extracutaneous disease) usually are treated with systemic chemotherapy. Our case highlights the importance of whole-body staging, as PET-CT scanning changed the course of care by detecting osseous involvement, necessitating systemic therapy as opposed to local radiation therapy alone. A multidisciplinary team with a focus on the diagnosis and management of cutaneous lymphomas helped streamline our patient’s laboratory testing and imaging studies, diagnostic and therapeutic decision making, and treatment implementation. Open channels and frequent opportunities for communication among dermatologists, dermatopathologists, medical oncologists, hematopathologists, radiologists, and radiation oncologists are needed to optimize and coordinate care for patients with cutaneous lymphoma who require transdisciplinary care.
Acknowledgement—The authors would like to thank Henry Koon, MD (hematology/oncology), for his input and expertise.
1. Willemze R, Kerl H, Sterry W, et al. EORTC classification for primary cutaneous lymphomas: a proposal from the cutaneous lymphoma study group of the European organization for research and treatment of cancer. Blood. 1997;90:354-371.
2. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
3. Kim YH, Willemze R, Pimpinelli N, et al. TNM classification system for primary cutaneous lymphomas other than mycosis fungoides and Sézary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110:479-484.
4. Quereux G, Frot AS, Brocard A, et al. Routine bone marrow biopsy in the initial evaluation of primary cutaneous B-cell lymphoma does not appear justified. Eur J Dermatol. 2009;19:216-220.
5. Senff NJ, Kluin-Nelemans HC, Willemze R. Results of bone marrow examination in 275 patients with histological features that suggest an indolent type of cutaneous B-cell lymphoma. Br J Haematol. 2008;142:52-56.
6. Gilliam AC, Wood GS. Cutaneous lymphoid hyperplasias. Semin Cutan Med Surg. 2000;19:133-141.
7. Burg G, Kempf W, Cozzio A, et al. WHO/EORTC classification of cutaneous lymphomas 2005: histological and molecular aspects. J Cutan Pathol. 2005;32:647-674.
8. Senff NJ, Noordijk EM, Kim YH, et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood. 2008;112:1600-1609.
9. Wright G, Tan B, Rosenwald A, et al. A gene expression-based method to diagnose clinically distinct subgroups of diffuse large B cell lymphoma. Proc Natl Acad Sci U S A. 2003;100:9991-9996.
10. Rydström K, Linderoth J, Nyman H, et al. CD40 is a potential marker of favorable prognosis in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Leuk Lymphoma. 2010;51:1643-1648.
11. Hoeller S, Schneider A, Haralambieva E, et al. FOXP1 protein overexpression is associated with inferior outcome in nodal diffuse large B-cell lymphomas with non-gerzminal centre phenotype, independent of gains and structural aberrations at 3p14.1. Histopathology. 2010;57:73-80.
A 59-year-old white man presented with 2 large erythematous lesions on the right side of the chest wall that had gradually progressed over the last 1.5 years. The patient denied any fever, night sweats, fatigue, unintentional weight loss, or loss of appetite. Physical examination revealed 2 large, well-circumscribed, nearly contiguous, firm, erythematous tumors. One tumor measured 7.5×4.5 cm and the other measured 4×3.5 cm.
The Diagnosis: Cutaneous B-cell Lymphoma
Biopsies from the right side of the chest wall (Figure 1) revealed an atypical dense and diffuse lymphocytic infiltrate throughout the dermis. There was extensive crush artifact throughout the specimen. However, the findings were consistent with cutaneous B-cell lymphoma (CBCL), and the diffuse large B-cell type was favored (Figure 2). Atypical lymphocytes stained positively for antibodies against CD20 (Figure 3), CD79a, and BCL-6, and stained negatively for antibodies against MUM-1 and BCL-2. Although flow cytometry revealed no definitive immunophenotypic lymphoma population, polymerase chain reaction analysis revealed a monoclonal immunoglobulin heavy chain gene rearrangement. Computed tomography (CT) scans of the chest, abdomen, and pelvis were unremarkable. A preliminary diagnosis of primary CBCL (PCBCL) was formulated. Diffuse large B-cell lymphoma (DLBCL) and follicle center lymphoma subtypes were each considered, which triggered further workup to rule out systemic involvement.
|
|

|

|
A bone marrow biopsy from the posterior iliac crest revealed normocellular bone marrow with normal trilineage hematopoiesis. However, whole-body staging with positron emission tomography (PET)–CT scanning revealed osseous disease in the left proximal humerus (Figure 4) as well as a slightly hypermetabolic right axillary lymph node. Magnetic resonance imaging of the brain showed no evidence of intracranial disease. Because of the apparent systemic involvement, stage IV non-Hodgkin lymphoma (DLBCL) became the new suspected diagnosis. The patient was started on the first of 6 cycles of chemotherapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), and the skin lesions quickly dissipated and flattened. A faint pink discoloration remained over a slightly indented area. A repeat PET-CT scan following 4 cycles of R-CHOP chemotherapy also confirmed a complete response to therapy.
In general, CBCL tends to affect adults and presents as relatively firm and plum-colored papules, nodules, tumors, or plaques, which can be either fast or slow growing. Cutaneous B-cell lymphoma may be primary or secondary to systemic involvement. Primary CBCL refers to a group of non-Hodgkin lymphomas that initially present in the skin with no evidence of extracutaneous involvement at the time of diagnosis.1,2 Secondary CBCL (SCBCL) refers to cutaneous disease that occurs secondary to systemic B-cell lymphoma. Detecting systemic involvement and distinguishing between PCBCL and SCBCL is valuable in determining prognosis and therapeutic options, as subtypes of PCBCL often have an improved prognosis and may be treated with local irradiation.
The initial staging techniques that are preferred for cutaneous lymphomas have been debated.3-5 For cutaneous lymphomas, except mycosis fungoides and Sézary syndrome, the International Society for Cutaneous Lymphomas and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer recommends obtaining a complete blood cell count with differential; complete metabolic studies including lactate dehydrogenase; and imaging studies of the chest, abdomen, and pelvis. Bone marrow biopsies and imaging studies of the neck or whole-body PET-CT scanning also may be useful depending on the clinical scenario.3 Although a more limited workup may be sufficient for PCBCLs such as primary cutaneous marginal zone lymphoma,5 a bone marrow biopsy is recommended for cases of primary cutaneous DLBCL (leg type).3 Senff et al5 supported the use of a bone marrow biopsy in the evaluation of follicle center lymphomas first presenting in the skin, though this method is controversial. In our patient, the laboratory results; bone marrow biopsy; and CT scan of the chest, abdomen, and pelvis failed to suggest extracutaneous disease, while the PET-CT scan revealed systemic involvement.
The differential diagnosis of CBCL includes cutaneous lymphoid hyperplasia (pseudolymphoma), which may be the result of insults such as arthropod bites, stings, vaccinations, or trauma. The clinical presentation, histology, and results of molecular studies and immunohistochemistry are essential in differentiating benign versus malignant processes.6 Lymphomas are expected to be larger and more persistent than benign processes, demonstrating an atypical lymphocytic infiltrate and monoclonality; immunohistochemistry will aid in the distinction between B-cell and T-cell processes and can delineate the type of B-cell lymphoma. Histology for CBCL typically reveals an atypical lymphocytic infiltrate showing a CD20+ and CD79a+ immunophenotype. Staining for antibodies against BCL-2, BCL-6, CD10, and MUM-1 also plays an important role in the diagnosis of cutaneous lymphoma and determining where the lesion(s) falls within the classification schemes. For example, to differentiate between primary cutaneous lymphoma subtypes, BCL-2 negativity and BCL-6 positivity in the context of a CD20+ and CD79a+ immunophenotype supports a follicle center lymphoma or a DLBCL (non–leg type). By contrast, CD20, CD79a, BCL-2, and MUM-1 positivity would favor a DLBCL (leg type).7
The natural history and therapeutic options differ greatly between subtypes of CBCL. For example, the prognosis of primary cutaneous follicle center lymphoma is generally favorable with a 5-year disease-specific survival rate of roughly 95%, and radiation therapy is recommended as a first-line therapy for localized disease.2,8 Conversely, primary cutaneous DLBCL (leg type) frequently spreads to extracutaneous sites8 and carries a much lower estimated 5-year disease-specific survival rate of 55%.2 Chemotherapy with R-CHOP is typically included in initial therapy for primary cutaneous DLBCL (leg type).8 The prognosis of systemic B-cell lymphomas also is highly variable and may depend on the type of B-cell lymphoma, the stage of disease at diagnosis, histologic and immunologic characteristics, and the therapy received. Wright et al9 reported that patients with systemic germinal center B cell–like DLBCL had a 5-year survival rate of 62%, whereas patients with activated B cell–like variants of DLBCL had a 5-year survival rate of 26%. Expression of CD40 may be a favorable prognostic factor following treatment with systemic chemotherapy in patients with DLBCL,10 whereas FOXP1 protein overexpression is correlated with poor disease-specific survival in certain DLBCL phenotypes.11
Although it is uncertain whether the cutaneous lesions preceded systemic disease in our patient, the cutaneous lesions could be arbitrarily classified as secondary because extracutaneous disease was discovered within 6 months of the initial diagnosis.1 However, classifying the CBCL as primary or secondary did not alter the course of treatment in our patient, as the presumed systemic disease necessitated treatment with systemic chemotherapy; both PCBCLs that develop systemic involvement and SCBCLs (primary extracutaneous disease) usually are treated with systemic chemotherapy. Our case highlights the importance of whole-body staging, as PET-CT scanning changed the course of care by detecting osseous involvement, necessitating systemic therapy as opposed to local radiation therapy alone. A multidisciplinary team with a focus on the diagnosis and management of cutaneous lymphomas helped streamline our patient’s laboratory testing and imaging studies, diagnostic and therapeutic decision making, and treatment implementation. Open channels and frequent opportunities for communication among dermatologists, dermatopathologists, medical oncologists, hematopathologists, radiologists, and radiation oncologists are needed to optimize and coordinate care for patients with cutaneous lymphoma who require transdisciplinary care.
Acknowledgement—The authors would like to thank Henry Koon, MD (hematology/oncology), for his input and expertise.
A 59-year-old white man presented with 2 large erythematous lesions on the right side of the chest wall that had gradually progressed over the last 1.5 years. The patient denied any fever, night sweats, fatigue, unintentional weight loss, or loss of appetite. Physical examination revealed 2 large, well-circumscribed, nearly contiguous, firm, erythematous tumors. One tumor measured 7.5×4.5 cm and the other measured 4×3.5 cm.
The Diagnosis: Cutaneous B-cell Lymphoma
Biopsies from the right side of the chest wall (Figure 1) revealed an atypical dense and diffuse lymphocytic infiltrate throughout the dermis. There was extensive crush artifact throughout the specimen. However, the findings were consistent with cutaneous B-cell lymphoma (CBCL), and the diffuse large B-cell type was favored (Figure 2). Atypical lymphocytes stained positively for antibodies against CD20 (Figure 3), CD79a, and BCL-6, and stained negatively for antibodies against MUM-1 and BCL-2. Although flow cytometry revealed no definitive immunophenotypic lymphoma population, polymerase chain reaction analysis revealed a monoclonal immunoglobulin heavy chain gene rearrangement. Computed tomography (CT) scans of the chest, abdomen, and pelvis were unremarkable. A preliminary diagnosis of primary CBCL (PCBCL) was formulated. Diffuse large B-cell lymphoma (DLBCL) and follicle center lymphoma subtypes were each considered, which triggered further workup to rule out systemic involvement.
|
|

|

|
A bone marrow biopsy from the posterior iliac crest revealed normocellular bone marrow with normal trilineage hematopoiesis. However, whole-body staging with positron emission tomography (PET)–CT scanning revealed osseous disease in the left proximal humerus (Figure 4) as well as a slightly hypermetabolic right axillary lymph node. Magnetic resonance imaging of the brain showed no evidence of intracranial disease. Because of the apparent systemic involvement, stage IV non-Hodgkin lymphoma (DLBCL) became the new suspected diagnosis. The patient was started on the first of 6 cycles of chemotherapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), and the skin lesions quickly dissipated and flattened. A faint pink discoloration remained over a slightly indented area. A repeat PET-CT scan following 4 cycles of R-CHOP chemotherapy also confirmed a complete response to therapy.
In general, CBCL tends to affect adults and presents as relatively firm and plum-colored papules, nodules, tumors, or plaques, which can be either fast or slow growing. Cutaneous B-cell lymphoma may be primary or secondary to systemic involvement. Primary CBCL refers to a group of non-Hodgkin lymphomas that initially present in the skin with no evidence of extracutaneous involvement at the time of diagnosis.1,2 Secondary CBCL (SCBCL) refers to cutaneous disease that occurs secondary to systemic B-cell lymphoma. Detecting systemic involvement and distinguishing between PCBCL and SCBCL is valuable in determining prognosis and therapeutic options, as subtypes of PCBCL often have an improved prognosis and may be treated with local irradiation.
The initial staging techniques that are preferred for cutaneous lymphomas have been debated.3-5 For cutaneous lymphomas, except mycosis fungoides and Sézary syndrome, the International Society for Cutaneous Lymphomas and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer recommends obtaining a complete blood cell count with differential; complete metabolic studies including lactate dehydrogenase; and imaging studies of the chest, abdomen, and pelvis. Bone marrow biopsies and imaging studies of the neck or whole-body PET-CT scanning also may be useful depending on the clinical scenario.3 Although a more limited workup may be sufficient for PCBCLs such as primary cutaneous marginal zone lymphoma,5 a bone marrow biopsy is recommended for cases of primary cutaneous DLBCL (leg type).3 Senff et al5 supported the use of a bone marrow biopsy in the evaluation of follicle center lymphomas first presenting in the skin, though this method is controversial. In our patient, the laboratory results; bone marrow biopsy; and CT scan of the chest, abdomen, and pelvis failed to suggest extracutaneous disease, while the PET-CT scan revealed systemic involvement.
The differential diagnosis of CBCL includes cutaneous lymphoid hyperplasia (pseudolymphoma), which may be the result of insults such as arthropod bites, stings, vaccinations, or trauma. The clinical presentation, histology, and results of molecular studies and immunohistochemistry are essential in differentiating benign versus malignant processes.6 Lymphomas are expected to be larger and more persistent than benign processes, demonstrating an atypical lymphocytic infiltrate and monoclonality; immunohistochemistry will aid in the distinction between B-cell and T-cell processes and can delineate the type of B-cell lymphoma. Histology for CBCL typically reveals an atypical lymphocytic infiltrate showing a CD20+ and CD79a+ immunophenotype. Staining for antibodies against BCL-2, BCL-6, CD10, and MUM-1 also plays an important role in the diagnosis of cutaneous lymphoma and determining where the lesion(s) falls within the classification schemes. For example, to differentiate between primary cutaneous lymphoma subtypes, BCL-2 negativity and BCL-6 positivity in the context of a CD20+ and CD79a+ immunophenotype supports a follicle center lymphoma or a DLBCL (non–leg type). By contrast, CD20, CD79a, BCL-2, and MUM-1 positivity would favor a DLBCL (leg type).7
The natural history and therapeutic options differ greatly between subtypes of CBCL. For example, the prognosis of primary cutaneous follicle center lymphoma is generally favorable with a 5-year disease-specific survival rate of roughly 95%, and radiation therapy is recommended as a first-line therapy for localized disease.2,8 Conversely, primary cutaneous DLBCL (leg type) frequently spreads to extracutaneous sites8 and carries a much lower estimated 5-year disease-specific survival rate of 55%.2 Chemotherapy with R-CHOP is typically included in initial therapy for primary cutaneous DLBCL (leg type).8 The prognosis of systemic B-cell lymphomas also is highly variable and may depend on the type of B-cell lymphoma, the stage of disease at diagnosis, histologic and immunologic characteristics, and the therapy received. Wright et al9 reported that patients with systemic germinal center B cell–like DLBCL had a 5-year survival rate of 62%, whereas patients with activated B cell–like variants of DLBCL had a 5-year survival rate of 26%. Expression of CD40 may be a favorable prognostic factor following treatment with systemic chemotherapy in patients with DLBCL,10 whereas FOXP1 protein overexpression is correlated with poor disease-specific survival in certain DLBCL phenotypes.11
Although it is uncertain whether the cutaneous lesions preceded systemic disease in our patient, the cutaneous lesions could be arbitrarily classified as secondary because extracutaneous disease was discovered within 6 months of the initial diagnosis.1 However, classifying the CBCL as primary or secondary did not alter the course of treatment in our patient, as the presumed systemic disease necessitated treatment with systemic chemotherapy; both PCBCLs that develop systemic involvement and SCBCLs (primary extracutaneous disease) usually are treated with systemic chemotherapy. Our case highlights the importance of whole-body staging, as PET-CT scanning changed the course of care by detecting osseous involvement, necessitating systemic therapy as opposed to local radiation therapy alone. A multidisciplinary team with a focus on the diagnosis and management of cutaneous lymphomas helped streamline our patient’s laboratory testing and imaging studies, diagnostic and therapeutic decision making, and treatment implementation. Open channels and frequent opportunities for communication among dermatologists, dermatopathologists, medical oncologists, hematopathologists, radiologists, and radiation oncologists are needed to optimize and coordinate care for patients with cutaneous lymphoma who require transdisciplinary care.
Acknowledgement—The authors would like to thank Henry Koon, MD (hematology/oncology), for his input and expertise.
1. Willemze R, Kerl H, Sterry W, et al. EORTC classification for primary cutaneous lymphomas: a proposal from the cutaneous lymphoma study group of the European organization for research and treatment of cancer. Blood. 1997;90:354-371.
2. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
3. Kim YH, Willemze R, Pimpinelli N, et al. TNM classification system for primary cutaneous lymphomas other than mycosis fungoides and Sézary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110:479-484.
4. Quereux G, Frot AS, Brocard A, et al. Routine bone marrow biopsy in the initial evaluation of primary cutaneous B-cell lymphoma does not appear justified. Eur J Dermatol. 2009;19:216-220.
5. Senff NJ, Kluin-Nelemans HC, Willemze R. Results of bone marrow examination in 275 patients with histological features that suggest an indolent type of cutaneous B-cell lymphoma. Br J Haematol. 2008;142:52-56.
6. Gilliam AC, Wood GS. Cutaneous lymphoid hyperplasias. Semin Cutan Med Surg. 2000;19:133-141.
7. Burg G, Kempf W, Cozzio A, et al. WHO/EORTC classification of cutaneous lymphomas 2005: histological and molecular aspects. J Cutan Pathol. 2005;32:647-674.
8. Senff NJ, Noordijk EM, Kim YH, et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood. 2008;112:1600-1609.
9. Wright G, Tan B, Rosenwald A, et al. A gene expression-based method to diagnose clinically distinct subgroups of diffuse large B cell lymphoma. Proc Natl Acad Sci U S A. 2003;100:9991-9996.
10. Rydström K, Linderoth J, Nyman H, et al. CD40 is a potential marker of favorable prognosis in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Leuk Lymphoma. 2010;51:1643-1648.
11. Hoeller S, Schneider A, Haralambieva E, et al. FOXP1 protein overexpression is associated with inferior outcome in nodal diffuse large B-cell lymphomas with non-gerzminal centre phenotype, independent of gains and structural aberrations at 3p14.1. Histopathology. 2010;57:73-80.
1. Willemze R, Kerl H, Sterry W, et al. EORTC classification for primary cutaneous lymphomas: a proposal from the cutaneous lymphoma study group of the European organization for research and treatment of cancer. Blood. 1997;90:354-371.
2. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
3. Kim YH, Willemze R, Pimpinelli N, et al. TNM classification system for primary cutaneous lymphomas other than mycosis fungoides and Sézary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the Cutaneous Lymphoma Task Force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110:479-484.
4. Quereux G, Frot AS, Brocard A, et al. Routine bone marrow biopsy in the initial evaluation of primary cutaneous B-cell lymphoma does not appear justified. Eur J Dermatol. 2009;19:216-220.
5. Senff NJ, Kluin-Nelemans HC, Willemze R. Results of bone marrow examination in 275 patients with histological features that suggest an indolent type of cutaneous B-cell lymphoma. Br J Haematol. 2008;142:52-56.
6. Gilliam AC, Wood GS. Cutaneous lymphoid hyperplasias. Semin Cutan Med Surg. 2000;19:133-141.
7. Burg G, Kempf W, Cozzio A, et al. WHO/EORTC classification of cutaneous lymphomas 2005: histological and molecular aspects. J Cutan Pathol. 2005;32:647-674.
8. Senff NJ, Noordijk EM, Kim YH, et al. European Organization for Research and Treatment of Cancer and International Society for Cutaneous Lymphoma consensus recommendations for the management of cutaneous B-cell lymphomas. Blood. 2008;112:1600-1609.
9. Wright G, Tan B, Rosenwald A, et al. A gene expression-based method to diagnose clinically distinct subgroups of diffuse large B cell lymphoma. Proc Natl Acad Sci U S A. 2003;100:9991-9996.
10. Rydström K, Linderoth J, Nyman H, et al. CD40 is a potential marker of favorable prognosis in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Leuk Lymphoma. 2010;51:1643-1648.
11. Hoeller S, Schneider A, Haralambieva E, et al. FOXP1 protein overexpression is associated with inferior outcome in nodal diffuse large B-cell lymphomas with non-gerzminal centre phenotype, independent of gains and structural aberrations at 3p14.1. Histopathology. 2010;57:73-80.