User login
Health-Related Quality of Life in Skin Cancer Patients
As the most common form of cancer in the United States,1 dermatologists often focus on treating the physical aspects of skin cancer, but it is equally important to consider the consequences that this disease has on a patient’s quality of life (QOL). Health is a dynamic process, encompassing one’s physical, emotional, and psychosocial well-being. There are a number of ways to measure health outcomes including mortality, morbidity, health status, and QOL. In recent years, health-related QOL (HRQOL) outcomes in dermatology have become increasingly important to clinical practice and may become factors in quality measurement or reimbursement.
Understanding a patient’s HRQOL allows health care providers to better evaluate the burden of disease and disability associated with skin cancer and its treatment. Clinical severity is not always able to capture the extent to which a disease affects one’s life.2 Furthermore, physician estimation of disease severity is not always consistent with patient-reported outcomes.3 As such, clinical questionnaires may be invaluable tools capable of objectively reporting a patient’s perception of improvement in health, which may affect how a dermatologist approaches treatment, discussion, and maintenance.
Nonmelanoma Skin Cancer
Most nonmelanoma skin cancer (NMSC) occurs in readily visible areas, namely the head and neck. Surgical treatment minimizes recurrence and complication rates. Nonmelanoma skin cancer has a low mortality and a high cure rate if diagnosed early; therefore, it may be difficult to assess treatment efficacy on cure rates alone. The amalgamation of anxiety associated with the diagnosis, aesthetic and functional concerns regarding treatment, and long-term consequences including fear of future skin cancer may have a lasting effect on an individual’s psychosocial relationships and underscores the need for QOL studies.
Most generic QOL and dermatology-specific QOL instruments fail to accurately detect the concerns of patients with NMSC.4-6 Generic QOL measures used for skin cancer patients report scores of patients that were similar to population norms,4 suggesting that these tools may fail to appropriately assess unique QOL concerns among individuals with skin cancer. Furthermore, dermatology-specific instruments have been reported to be insensitive to specific appearance-related concerns of patients with NMSC, likely because skin cancer patients made up a small percentage of the initial population in their design.4,7 Nevertheless, dermatology-specific instruments may be suitable depending on the objectives of the study.8
Recently, skin cancer–specific QOL instruments have been developed to fill the paucity of appropriate tools for this population. These questionnaires include the Facial Skin Cancer Index, Skin Cancer Index, and the Skin Cancer Quality of Life Impact Tool.7 The Skin Cancer Index is a 15-item questionnaire validated in patients undergoing Mohs micrographic surgery and has been used to assess behavior modification and risk perceptions in NMSC patients. Importantly, it does ask the patient if he/she is worried about scarring. The Facial Skin Cancer Index and the Skin Cancer Quality of Life Impact Tool do not take into account detailed aesthetic concerns regarding facial disfigurement and scarring or expectations of reconstruction.7 It may be prudent to assess these areas with supplemental scales.
Melanoma
Melanoma, the third most common skin cancer, is highly aggressive and can affect young and middle-aged patients. Because the mortality associated with later-stage melanoma is greater, the QOL impact of melanoma differs from NMSC. There are also 3 distinct periods of melanoma HRQOL impact: diagnosis, treatment, and follow-up. Approximately 30% of patients diagnosed with melanoma report high levels of psychological distress.9 The psychosocial effects of a melanoma diagnosis are longitudinal, as there is a high survival rate in early disease but also an increased future risk for melanoma, affecting future behaviors and overall QOL. The diagnosis of melanoma also affects family members due to the increased risk among first-degree relatives. After removal of deeper melanoma, the patient remains at risk for disease progression, which can have a profound impact on his/her social and professional activities and overall lifestyle. There may be a role for longitudinal QOL assessments to monitor changes over time and direct ongoing therapy.
The proportion of patients with melanoma who report high levels of impairment in QOL is comparable to that seen in other malignancies.10 Generic QOL instruments have found that melanoma patients have medium to high levels of distress and substantial improvement in HRQOL has been achieved with cognitive-behavioral intervention.11 Quality-of-life studies also have shown levels of distress are highest at initial diagnosis and immediately following treatment.12 In a randomized surgical trial, patients with a larger excision margin had poorer mental and physical function scores on assessment.13 Skin-specific QOL instruments have been used in studies of patients with melanoma and found that postmelanoma surveillance did not impact QOL. Also, women experienced greater improvements in QOL over time after reporting lower scores immediately postsurgery.13
The FACT-melanoma (Functional Assessment of Cancer Therapy) is a melanoma-specific HRQOL assessment that has been used in patients undergoing clinical trials. It has been shown to distinguish between early and advanced-stage (stages III or IV) HRQOL issues.14 Patients with early-stage melanoma are more concerned with cosmetic outcome, and those with later-stage melanoma are more concerned with morbidity and mortality associated with treatment.
Comment
Choosing the best QOL instrument depends on the specific objectives of the study. Although generic QOL questionnaires have performed poorly in studies of specific skin diseases and even dermatology-specific tools have shown limited responsiveness in skin cancer, a combination of tools may be an effective approach. However, dermatologists must be cautious when administering these valuable tools to ensure that they do not become a burdensome task for the patient.15 Although no single skin cancer–specific QOL tool is perfect, it is likely that the current questionnaires still allow for aid with appropriate patient management and comparison of treatments.16
It behooves clinicians to recognize and appreciate the value of QOL instruments as an important adjunct to treatment. These tools have shown QOL to be an independent predictor of survival among many types of cancer patients, including melanoma.10 Currently, the psychological and emotional needs of skin cancer patients often go overlooked and undetected by conventional methods. Within one’s own practice, introducing QOL assessments can improve patient self-awareness and physician awareness of matters that may have a greater impact on patient health. On a larger scale, introducing patient-reported outcome measures can affect resource allocation by identifying patient populations that may be most impacted and can give a comprehensive method for physicians to gauge treatment efficacy, leading to improved outcomes.
1. Robinson JK. Sun exposure, sun protection, and vitamin D. JAMA. 2005;294:1541-1543.
2. Motley RJ, Finlay AY. Practical use of a disability index in the routine management of acne. Clin Exp Dermatol. 1992;17:1-3.
3. Chren MM, Lasek RJ, Quinn LM, et al. Skindex, a quality-of-life measure for patients with skin disease: reliability, validity, and responsiveness. J Invest Dermatol. 1996;107:707-713.
4. Gibbons EC, Comabella CI, Fitzpatrick R. A structured review of patient-reported outcome measures for patients with skin cancer, 2013. Br J Dermatol. 2013;168:1176-1186.
5. Burdon-Jones D, Thomas P, Baker R. Quality of life issues in nonmetastatic skin cancer. Br J Dermatol. 2010;162:147-151.
6. Lear W, Akeroyd JD, Mittmann N, et al. Measurement of utility in nonmelanoma skin cancer. J Cutan Med Surg. 2008;12:102-106.
7. Bates AS, Davis CR, Takwale A, et al. Patient-reported outcome measures in nonmelanoma skin cancer of the face: a systematic review. Br J Dermatol. 2013;168:1187-1194.
8. Lee EH, Klassen AF, Nehal KS, et al. A systematic review of patient-reported outcome instruments of nonmelanoma skin cancer in the dermatologic population. J Am Acad Dermatol. 2013;69:e59-e67.
9. Kasparian NA. Psychological stress and melanoma: are we meeting our patients’ psychological needs? Clin Dermatol. 2013;31:41-46.
10. Cormier JN, Cromwell KD, Ross MI. Health-related quality of life in patients with melanoma: overview of instruments and outcomes. Dermatol Clin. 2012;30:245-254.
11. Trask PC, Paterson AG, Griffith KA, et al. Cognitive-behavioral intervention for distress in patients with melanoma: comparison with standard medical care and impact on quality of life. Cancer. 2003;98:854-864.
12. Boyle DA. Psychological adjustment to the melanoma experience. Semin Oncol Nurs. 2003;191:70-77.
13. Newton-Bishop JA, Nolan C, Turner F, et al. A quality-of-life study in high-risk (thickness > = or 2 mm) cutaneous melanoma patients in a randomized trial of 1-cm versus 3-cm surgical excision margins. J Investig Dermatol Symp Proc. 2004;9:152-159.
14. Winstanley JB, Saw R, Boyle F, et al. The FACT-Melanoma quality-of-life instrument: comparison of a five-point and four-point response scale using the Rasch measurement model. Melanoma Res. 2013;23:61-69.
15. Swartz RJ, Baum GP, Askew RL, et al. Reducing patient burden to the FACT-Melanoma quality-of-life questionnaire. Melanoma Res. 2012;22:158-163.
16. Black N. Patient-reported outcome measures in skin cancer. Br J Dermatol. 2013;168:1151.
As the most common form of cancer in the United States,1 dermatologists often focus on treating the physical aspects of skin cancer, but it is equally important to consider the consequences that this disease has on a patient’s quality of life (QOL). Health is a dynamic process, encompassing one’s physical, emotional, and psychosocial well-being. There are a number of ways to measure health outcomes including mortality, morbidity, health status, and QOL. In recent years, health-related QOL (HRQOL) outcomes in dermatology have become increasingly important to clinical practice and may become factors in quality measurement or reimbursement.
Understanding a patient’s HRQOL allows health care providers to better evaluate the burden of disease and disability associated with skin cancer and its treatment. Clinical severity is not always able to capture the extent to which a disease affects one’s life.2 Furthermore, physician estimation of disease severity is not always consistent with patient-reported outcomes.3 As such, clinical questionnaires may be invaluable tools capable of objectively reporting a patient’s perception of improvement in health, which may affect how a dermatologist approaches treatment, discussion, and maintenance.
Nonmelanoma Skin Cancer
Most nonmelanoma skin cancer (NMSC) occurs in readily visible areas, namely the head and neck. Surgical treatment minimizes recurrence and complication rates. Nonmelanoma skin cancer has a low mortality and a high cure rate if diagnosed early; therefore, it may be difficult to assess treatment efficacy on cure rates alone. The amalgamation of anxiety associated with the diagnosis, aesthetic and functional concerns regarding treatment, and long-term consequences including fear of future skin cancer may have a lasting effect on an individual’s psychosocial relationships and underscores the need for QOL studies.
Most generic QOL and dermatology-specific QOL instruments fail to accurately detect the concerns of patients with NMSC.4-6 Generic QOL measures used for skin cancer patients report scores of patients that were similar to population norms,4 suggesting that these tools may fail to appropriately assess unique QOL concerns among individuals with skin cancer. Furthermore, dermatology-specific instruments have been reported to be insensitive to specific appearance-related concerns of patients with NMSC, likely because skin cancer patients made up a small percentage of the initial population in their design.4,7 Nevertheless, dermatology-specific instruments may be suitable depending on the objectives of the study.8
Recently, skin cancer–specific QOL instruments have been developed to fill the paucity of appropriate tools for this population. These questionnaires include the Facial Skin Cancer Index, Skin Cancer Index, and the Skin Cancer Quality of Life Impact Tool.7 The Skin Cancer Index is a 15-item questionnaire validated in patients undergoing Mohs micrographic surgery and has been used to assess behavior modification and risk perceptions in NMSC patients. Importantly, it does ask the patient if he/she is worried about scarring. The Facial Skin Cancer Index and the Skin Cancer Quality of Life Impact Tool do not take into account detailed aesthetic concerns regarding facial disfigurement and scarring or expectations of reconstruction.7 It may be prudent to assess these areas with supplemental scales.
Melanoma
Melanoma, the third most common skin cancer, is highly aggressive and can affect young and middle-aged patients. Because the mortality associated with later-stage melanoma is greater, the QOL impact of melanoma differs from NMSC. There are also 3 distinct periods of melanoma HRQOL impact: diagnosis, treatment, and follow-up. Approximately 30% of patients diagnosed with melanoma report high levels of psychological distress.9 The psychosocial effects of a melanoma diagnosis are longitudinal, as there is a high survival rate in early disease but also an increased future risk for melanoma, affecting future behaviors and overall QOL. The diagnosis of melanoma also affects family members due to the increased risk among first-degree relatives. After removal of deeper melanoma, the patient remains at risk for disease progression, which can have a profound impact on his/her social and professional activities and overall lifestyle. There may be a role for longitudinal QOL assessments to monitor changes over time and direct ongoing therapy.
The proportion of patients with melanoma who report high levels of impairment in QOL is comparable to that seen in other malignancies.10 Generic QOL instruments have found that melanoma patients have medium to high levels of distress and substantial improvement in HRQOL has been achieved with cognitive-behavioral intervention.11 Quality-of-life studies also have shown levels of distress are highest at initial diagnosis and immediately following treatment.12 In a randomized surgical trial, patients with a larger excision margin had poorer mental and physical function scores on assessment.13 Skin-specific QOL instruments have been used in studies of patients with melanoma and found that postmelanoma surveillance did not impact QOL. Also, women experienced greater improvements in QOL over time after reporting lower scores immediately postsurgery.13
The FACT-melanoma (Functional Assessment of Cancer Therapy) is a melanoma-specific HRQOL assessment that has been used in patients undergoing clinical trials. It has been shown to distinguish between early and advanced-stage (stages III or IV) HRQOL issues.14 Patients with early-stage melanoma are more concerned with cosmetic outcome, and those with later-stage melanoma are more concerned with morbidity and mortality associated with treatment.
Comment
Choosing the best QOL instrument depends on the specific objectives of the study. Although generic QOL questionnaires have performed poorly in studies of specific skin diseases and even dermatology-specific tools have shown limited responsiveness in skin cancer, a combination of tools may be an effective approach. However, dermatologists must be cautious when administering these valuable tools to ensure that they do not become a burdensome task for the patient.15 Although no single skin cancer–specific QOL tool is perfect, it is likely that the current questionnaires still allow for aid with appropriate patient management and comparison of treatments.16
It behooves clinicians to recognize and appreciate the value of QOL instruments as an important adjunct to treatment. These tools have shown QOL to be an independent predictor of survival among many types of cancer patients, including melanoma.10 Currently, the psychological and emotional needs of skin cancer patients often go overlooked and undetected by conventional methods. Within one’s own practice, introducing QOL assessments can improve patient self-awareness and physician awareness of matters that may have a greater impact on patient health. On a larger scale, introducing patient-reported outcome measures can affect resource allocation by identifying patient populations that may be most impacted and can give a comprehensive method for physicians to gauge treatment efficacy, leading to improved outcomes.
As the most common form of cancer in the United States,1 dermatologists often focus on treating the physical aspects of skin cancer, but it is equally important to consider the consequences that this disease has on a patient’s quality of life (QOL). Health is a dynamic process, encompassing one’s physical, emotional, and psychosocial well-being. There are a number of ways to measure health outcomes including mortality, morbidity, health status, and QOL. In recent years, health-related QOL (HRQOL) outcomes in dermatology have become increasingly important to clinical practice and may become factors in quality measurement or reimbursement.
Understanding a patient’s HRQOL allows health care providers to better evaluate the burden of disease and disability associated with skin cancer and its treatment. Clinical severity is not always able to capture the extent to which a disease affects one’s life.2 Furthermore, physician estimation of disease severity is not always consistent with patient-reported outcomes.3 As such, clinical questionnaires may be invaluable tools capable of objectively reporting a patient’s perception of improvement in health, which may affect how a dermatologist approaches treatment, discussion, and maintenance.
Nonmelanoma Skin Cancer
Most nonmelanoma skin cancer (NMSC) occurs in readily visible areas, namely the head and neck. Surgical treatment minimizes recurrence and complication rates. Nonmelanoma skin cancer has a low mortality and a high cure rate if diagnosed early; therefore, it may be difficult to assess treatment efficacy on cure rates alone. The amalgamation of anxiety associated with the diagnosis, aesthetic and functional concerns regarding treatment, and long-term consequences including fear of future skin cancer may have a lasting effect on an individual’s psychosocial relationships and underscores the need for QOL studies.
Most generic QOL and dermatology-specific QOL instruments fail to accurately detect the concerns of patients with NMSC.4-6 Generic QOL measures used for skin cancer patients report scores of patients that were similar to population norms,4 suggesting that these tools may fail to appropriately assess unique QOL concerns among individuals with skin cancer. Furthermore, dermatology-specific instruments have been reported to be insensitive to specific appearance-related concerns of patients with NMSC, likely because skin cancer patients made up a small percentage of the initial population in their design.4,7 Nevertheless, dermatology-specific instruments may be suitable depending on the objectives of the study.8
Recently, skin cancer–specific QOL instruments have been developed to fill the paucity of appropriate tools for this population. These questionnaires include the Facial Skin Cancer Index, Skin Cancer Index, and the Skin Cancer Quality of Life Impact Tool.7 The Skin Cancer Index is a 15-item questionnaire validated in patients undergoing Mohs micrographic surgery and has been used to assess behavior modification and risk perceptions in NMSC patients. Importantly, it does ask the patient if he/she is worried about scarring. The Facial Skin Cancer Index and the Skin Cancer Quality of Life Impact Tool do not take into account detailed aesthetic concerns regarding facial disfigurement and scarring or expectations of reconstruction.7 It may be prudent to assess these areas with supplemental scales.
Melanoma
Melanoma, the third most common skin cancer, is highly aggressive and can affect young and middle-aged patients. Because the mortality associated with later-stage melanoma is greater, the QOL impact of melanoma differs from NMSC. There are also 3 distinct periods of melanoma HRQOL impact: diagnosis, treatment, and follow-up. Approximately 30% of patients diagnosed with melanoma report high levels of psychological distress.9 The psychosocial effects of a melanoma diagnosis are longitudinal, as there is a high survival rate in early disease but also an increased future risk for melanoma, affecting future behaviors and overall QOL. The diagnosis of melanoma also affects family members due to the increased risk among first-degree relatives. After removal of deeper melanoma, the patient remains at risk for disease progression, which can have a profound impact on his/her social and professional activities and overall lifestyle. There may be a role for longitudinal QOL assessments to monitor changes over time and direct ongoing therapy.
The proportion of patients with melanoma who report high levels of impairment in QOL is comparable to that seen in other malignancies.10 Generic QOL instruments have found that melanoma patients have medium to high levels of distress and substantial improvement in HRQOL has been achieved with cognitive-behavioral intervention.11 Quality-of-life studies also have shown levels of distress are highest at initial diagnosis and immediately following treatment.12 In a randomized surgical trial, patients with a larger excision margin had poorer mental and physical function scores on assessment.13 Skin-specific QOL instruments have been used in studies of patients with melanoma and found that postmelanoma surveillance did not impact QOL. Also, women experienced greater improvements in QOL over time after reporting lower scores immediately postsurgery.13
The FACT-melanoma (Functional Assessment of Cancer Therapy) is a melanoma-specific HRQOL assessment that has been used in patients undergoing clinical trials. It has been shown to distinguish between early and advanced-stage (stages III or IV) HRQOL issues.14 Patients with early-stage melanoma are more concerned with cosmetic outcome, and those with later-stage melanoma are more concerned with morbidity and mortality associated with treatment.
Comment
Choosing the best QOL instrument depends on the specific objectives of the study. Although generic QOL questionnaires have performed poorly in studies of specific skin diseases and even dermatology-specific tools have shown limited responsiveness in skin cancer, a combination of tools may be an effective approach. However, dermatologists must be cautious when administering these valuable tools to ensure that they do not become a burdensome task for the patient.15 Although no single skin cancer–specific QOL tool is perfect, it is likely that the current questionnaires still allow for aid with appropriate patient management and comparison of treatments.16
It behooves clinicians to recognize and appreciate the value of QOL instruments as an important adjunct to treatment. These tools have shown QOL to be an independent predictor of survival among many types of cancer patients, including melanoma.10 Currently, the psychological and emotional needs of skin cancer patients often go overlooked and undetected by conventional methods. Within one’s own practice, introducing QOL assessments can improve patient self-awareness and physician awareness of matters that may have a greater impact on patient health. On a larger scale, introducing patient-reported outcome measures can affect resource allocation by identifying patient populations that may be most impacted and can give a comprehensive method for physicians to gauge treatment efficacy, leading to improved outcomes.
1. Robinson JK. Sun exposure, sun protection, and vitamin D. JAMA. 2005;294:1541-1543.
2. Motley RJ, Finlay AY. Practical use of a disability index in the routine management of acne. Clin Exp Dermatol. 1992;17:1-3.
3. Chren MM, Lasek RJ, Quinn LM, et al. Skindex, a quality-of-life measure for patients with skin disease: reliability, validity, and responsiveness. J Invest Dermatol. 1996;107:707-713.
4. Gibbons EC, Comabella CI, Fitzpatrick R. A structured review of patient-reported outcome measures for patients with skin cancer, 2013. Br J Dermatol. 2013;168:1176-1186.
5. Burdon-Jones D, Thomas P, Baker R. Quality of life issues in nonmetastatic skin cancer. Br J Dermatol. 2010;162:147-151.
6. Lear W, Akeroyd JD, Mittmann N, et al. Measurement of utility in nonmelanoma skin cancer. J Cutan Med Surg. 2008;12:102-106.
7. Bates AS, Davis CR, Takwale A, et al. Patient-reported outcome measures in nonmelanoma skin cancer of the face: a systematic review. Br J Dermatol. 2013;168:1187-1194.
8. Lee EH, Klassen AF, Nehal KS, et al. A systematic review of patient-reported outcome instruments of nonmelanoma skin cancer in the dermatologic population. J Am Acad Dermatol. 2013;69:e59-e67.
9. Kasparian NA. Psychological stress and melanoma: are we meeting our patients’ psychological needs? Clin Dermatol. 2013;31:41-46.
10. Cormier JN, Cromwell KD, Ross MI. Health-related quality of life in patients with melanoma: overview of instruments and outcomes. Dermatol Clin. 2012;30:245-254.
11. Trask PC, Paterson AG, Griffith KA, et al. Cognitive-behavioral intervention for distress in patients with melanoma: comparison with standard medical care and impact on quality of life. Cancer. 2003;98:854-864.
12. Boyle DA. Psychological adjustment to the melanoma experience. Semin Oncol Nurs. 2003;191:70-77.
13. Newton-Bishop JA, Nolan C, Turner F, et al. A quality-of-life study in high-risk (thickness > = or 2 mm) cutaneous melanoma patients in a randomized trial of 1-cm versus 3-cm surgical excision margins. J Investig Dermatol Symp Proc. 2004;9:152-159.
14. Winstanley JB, Saw R, Boyle F, et al. The FACT-Melanoma quality-of-life instrument: comparison of a five-point and four-point response scale using the Rasch measurement model. Melanoma Res. 2013;23:61-69.
15. Swartz RJ, Baum GP, Askew RL, et al. Reducing patient burden to the FACT-Melanoma quality-of-life questionnaire. Melanoma Res. 2012;22:158-163.
16. Black N. Patient-reported outcome measures in skin cancer. Br J Dermatol. 2013;168:1151.
1. Robinson JK. Sun exposure, sun protection, and vitamin D. JAMA. 2005;294:1541-1543.
2. Motley RJ, Finlay AY. Practical use of a disability index in the routine management of acne. Clin Exp Dermatol. 1992;17:1-3.
3. Chren MM, Lasek RJ, Quinn LM, et al. Skindex, a quality-of-life measure for patients with skin disease: reliability, validity, and responsiveness. J Invest Dermatol. 1996;107:707-713.
4. Gibbons EC, Comabella CI, Fitzpatrick R. A structured review of patient-reported outcome measures for patients with skin cancer, 2013. Br J Dermatol. 2013;168:1176-1186.
5. Burdon-Jones D, Thomas P, Baker R. Quality of life issues in nonmetastatic skin cancer. Br J Dermatol. 2010;162:147-151.
6. Lear W, Akeroyd JD, Mittmann N, et al. Measurement of utility in nonmelanoma skin cancer. J Cutan Med Surg. 2008;12:102-106.
7. Bates AS, Davis CR, Takwale A, et al. Patient-reported outcome measures in nonmelanoma skin cancer of the face: a systematic review. Br J Dermatol. 2013;168:1187-1194.
8. Lee EH, Klassen AF, Nehal KS, et al. A systematic review of patient-reported outcome instruments of nonmelanoma skin cancer in the dermatologic population. J Am Acad Dermatol. 2013;69:e59-e67.
9. Kasparian NA. Psychological stress and melanoma: are we meeting our patients’ psychological needs? Clin Dermatol. 2013;31:41-46.
10. Cormier JN, Cromwell KD, Ross MI. Health-related quality of life in patients with melanoma: overview of instruments and outcomes. Dermatol Clin. 2012;30:245-254.
11. Trask PC, Paterson AG, Griffith KA, et al. Cognitive-behavioral intervention for distress in patients with melanoma: comparison with standard medical care and impact on quality of life. Cancer. 2003;98:854-864.
12. Boyle DA. Psychological adjustment to the melanoma experience. Semin Oncol Nurs. 2003;191:70-77.
13. Newton-Bishop JA, Nolan C, Turner F, et al. A quality-of-life study in high-risk (thickness > = or 2 mm) cutaneous melanoma patients in a randomized trial of 1-cm versus 3-cm surgical excision margins. J Investig Dermatol Symp Proc. 2004;9:152-159.
14. Winstanley JB, Saw R, Boyle F, et al. The FACT-Melanoma quality-of-life instrument: comparison of a five-point and four-point response scale using the Rasch measurement model. Melanoma Res. 2013;23:61-69.
15. Swartz RJ, Baum GP, Askew RL, et al. Reducing patient burden to the FACT-Melanoma quality-of-life questionnaire. Melanoma Res. 2012;22:158-163.
16. Black N. Patient-reported outcome measures in skin cancer. Br J Dermatol. 2013;168:1151.
Combination Therapy for Perianal Squamous Cell Carcinoma In Situ With Imiquimod and Photodynamic Therapy
Perianal squamous cell carcinoma in situ (SCCIS) is an intraepidermal neoplasm with human papillomavirus implicated in its etiology.1 It can present as a raised, scaly, erythematous, fissured, ulcerated, or pigmented lesion; however, perianal SCCIS often is subclinical and therefore requires a high level of suspicion in individuals with risk factors (eg, history of genital or perianal human papillomavirus infection, other sexually transmitted diseases, or cervical dysplasia).2 Although relatively rare, perianal SCCIS is believed to be increasing in frequency and has the potential to progress to invasive squamous cell carcinoma.1,3 The rarity of this neoplasm and its uncertain natural history has made the development of a definitive, evidence-based management strategy difficult and controversial.1,4,5 We present the case of a 61-year-old woman with perianal SCCIS who was treated with a novel combination of 5-aminolevulinic acid–based photodynamic therapy (ALA-PDT) and topical imiquimod cream 5% following 2 unsuccessful surgical excisions. Using this treatment regimen, the neoplasm resolved completely with no evidence of recurrence at 2 years’ follow-up.
Case Report
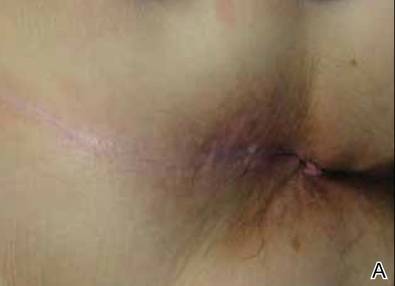
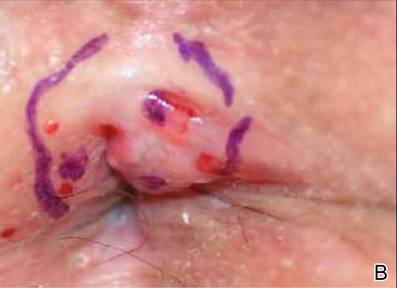
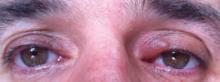
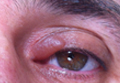
A 61-year-old woman was referred to our dermatology clinic for management of persistent SCCIS of the perianal region. A colorectal surgeon performed 2 unsuccessful excisions of the neoplasm at 6 months and 1 month prior to presentation. Biopsy results from the second excision demonstrated persistent perianal SCCIS with positive margins (Figure 1). The patient was referred to our clinic to discuss Mohs micrographic surgery versus nonsurgical treatment options. The patient’s medical history was remarkable for an abnormal Papanicolaou test 10 years prior, which resulted in cervical cryotherapy.
|
|
Physical examination revealed a 2×1-cm pink scar with peripheral scaling on the right anal verge (Figure 2A). Potential therapies discussed with the patient included Mohs surgery, ablative laser treatment, and nonsurgical treatment with ALA-PDT in combination with topical imiquimod cream 5%. To avoid further invasive treatments, we decided to administer a 3-week course of topical imiquimod cream 5% applied 3 times weekly to the entire affected area, followed 1 week later with a treatment of ALA-PDT. The incubation time was 6 hours. After completing 4 cycles of this therapeutic regimen, the erythema and scaling resolved (Figure 2B). The patient did not note any remarkable side effects associated with imiquimod but reported progressively resolving pain in the week following each ALA-PDT treatment, with the worst pain occurring during the first 48 hours after treatment. Three posttreatment scout biopsies revealed no evidence of residual perianal SCCIS. Two years after the negative biopsies, ongoing physical examinations demonstrated no evidence of clinical recurrence. Physicians in the dermatology and gynecology departments are following her closely.
Comment
Wide surgical excision is currently recommended as first-line therapy for perianal SCCIS.1,3,4 Unfortunately, surgical excision has been associated with difficulty in achieving disease-free margins, recurrence rates as high as 31%, and substantial morbidity.1,4 Other nonsurgical treatment modalities have been investigated for treating this neoplasm to simultaneously reduce recurrence rates while minimizing structural and functional damage to the treatment area, including radiotherapy, imiquimod, laser ablation, and ALA-PDT.1,3,4,5
In particular, imiquimod and ALA-PDT have shown promise as monotherapies for the treatment of perianal SCCIS, with several case reports describing complete resolution, low rates of recurrence, and preserved structure and function of the surrounding tissue after therapy1,3,4; however, recurrence of perianal SCCIS is still known to occur after monotherapy with either ALA-PDT or imiquimod.5,6
In contrast to noninvasive monotherapies, the literature is largely devoid of reports of noninvasive combination treatments used for perianal SCCIS. Our case represents successful use of a combination of imiquimod and ALA-PDT to treat persistent perianal SCCIS.
Conclusion
Although further research is necessary, the therapeutic success presented in our case suggests noninvasive combination therapy with imiquimod and ALA-PDT may represent a viable alternative to both surgical excision and noninvasive monotherapies for the treatment of perianal SCCIS.
Acknowledgement—The authors would like to thank Alejandro Gru, MD, Columbus, Ohio, for his review of the pathology slides for this case.
1. van Egmond S, Hoedemaker C, Sinclair R. Successful treatment of perianal Bowen’s disease with imiquimod. Int J Dermatol. 2007;46:318-319.
2. Abbasakoor F, Boulos PB. Anal intraepithelial neoplasia. Br J Surg. 2005;92:277-290.
3. Petrelli NJ, Cebollero JA, Rodriguez-Bigas M, et al. Photodynamic therapy in the management of neoplasms of the perianal skin. Arch Surg. 1992;127:1436-1438.
4. Wietfeldt E, Thiele J. Malignancies of the anal margin and perianal skin. Clin Colon Rectal Surg. 2009;22:127-135.
5. Pineda C, Welton M. Management of anal squamous intraepithelial lesions. Clin Colon Rectal Surg. 2009;22:94-101.
6. Runfola MA, Weber TK, Rodriguez-Bigas MA, et al. Photodynamic therapy for residual neoplasms of the perianal skin. Dis Colon Rectum. 2000;43:499-502.
Perianal squamous cell carcinoma in situ (SCCIS) is an intraepidermal neoplasm with human papillomavirus implicated in its etiology.1 It can present as a raised, scaly, erythematous, fissured, ulcerated, or pigmented lesion; however, perianal SCCIS often is subclinical and therefore requires a high level of suspicion in individuals with risk factors (eg, history of genital or perianal human papillomavirus infection, other sexually transmitted diseases, or cervical dysplasia).2 Although relatively rare, perianal SCCIS is believed to be increasing in frequency and has the potential to progress to invasive squamous cell carcinoma.1,3 The rarity of this neoplasm and its uncertain natural history has made the development of a definitive, evidence-based management strategy difficult and controversial.1,4,5 We present the case of a 61-year-old woman with perianal SCCIS who was treated with a novel combination of 5-aminolevulinic acid–based photodynamic therapy (ALA-PDT) and topical imiquimod cream 5% following 2 unsuccessful surgical excisions. Using this treatment regimen, the neoplasm resolved completely with no evidence of recurrence at 2 years’ follow-up.
Case Report
A 61-year-old woman was referred to our dermatology clinic for management of persistent SCCIS of the perianal region. A colorectal surgeon performed 2 unsuccessful excisions of the neoplasm at 6 months and 1 month prior to presentation. Biopsy results from the second excision demonstrated persistent perianal SCCIS with positive margins (Figure 1). The patient was referred to our clinic to discuss Mohs micrographic surgery versus nonsurgical treatment options. The patient’s medical history was remarkable for an abnormal Papanicolaou test 10 years prior, which resulted in cervical cryotherapy.

|
|
Physical examination revealed a 2×1-cm pink scar with peripheral scaling on the right anal verge (Figure 2A). Potential therapies discussed with the patient included Mohs surgery, ablative laser treatment, and nonsurgical treatment with ALA-PDT in combination with topical imiquimod cream 5%. To avoid further invasive treatments, we decided to administer a 3-week course of topical imiquimod cream 5% applied 3 times weekly to the entire affected area, followed 1 week later with a treatment of ALA-PDT. The incubation time was 6 hours. After completing 4 cycles of this therapeutic regimen, the erythema and scaling resolved (Figure 2B). The patient did not note any remarkable side effects associated with imiquimod but reported progressively resolving pain in the week following each ALA-PDT treatment, with the worst pain occurring during the first 48 hours after treatment. Three posttreatment scout biopsies revealed no evidence of residual perianal SCCIS. Two years after the negative biopsies, ongoing physical examinations demonstrated no evidence of clinical recurrence. Physicians in the dermatology and gynecology departments are following her closely.
Comment
Wide surgical excision is currently recommended as first-line therapy for perianal SCCIS.1,3,4 Unfortunately, surgical excision has been associated with difficulty in achieving disease-free margins, recurrence rates as high as 31%, and substantial morbidity.1,4 Other nonsurgical treatment modalities have been investigated for treating this neoplasm to simultaneously reduce recurrence rates while minimizing structural and functional damage to the treatment area, including radiotherapy, imiquimod, laser ablation, and ALA-PDT.1,3,4,5
In particular, imiquimod and ALA-PDT have shown promise as monotherapies for the treatment of perianal SCCIS, with several case reports describing complete resolution, low rates of recurrence, and preserved structure and function of the surrounding tissue after therapy1,3,4; however, recurrence of perianal SCCIS is still known to occur after monotherapy with either ALA-PDT or imiquimod.5,6
In contrast to noninvasive monotherapies, the literature is largely devoid of reports of noninvasive combination treatments used for perianal SCCIS. Our case represents successful use of a combination of imiquimod and ALA-PDT to treat persistent perianal SCCIS.
Conclusion
Although further research is necessary, the therapeutic success presented in our case suggests noninvasive combination therapy with imiquimod and ALA-PDT may represent a viable alternative to both surgical excision and noninvasive monotherapies for the treatment of perianal SCCIS.
Acknowledgement—The authors would like to thank Alejandro Gru, MD, Columbus, Ohio, for his review of the pathology slides for this case.
Perianal squamous cell carcinoma in situ (SCCIS) is an intraepidermal neoplasm with human papillomavirus implicated in its etiology.1 It can present as a raised, scaly, erythematous, fissured, ulcerated, or pigmented lesion; however, perianal SCCIS often is subclinical and therefore requires a high level of suspicion in individuals with risk factors (eg, history of genital or perianal human papillomavirus infection, other sexually transmitted diseases, or cervical dysplasia).2 Although relatively rare, perianal SCCIS is believed to be increasing in frequency and has the potential to progress to invasive squamous cell carcinoma.1,3 The rarity of this neoplasm and its uncertain natural history has made the development of a definitive, evidence-based management strategy difficult and controversial.1,4,5 We present the case of a 61-year-old woman with perianal SCCIS who was treated with a novel combination of 5-aminolevulinic acid–based photodynamic therapy (ALA-PDT) and topical imiquimod cream 5% following 2 unsuccessful surgical excisions. Using this treatment regimen, the neoplasm resolved completely with no evidence of recurrence at 2 years’ follow-up.
Case Report
A 61-year-old woman was referred to our dermatology clinic for management of persistent SCCIS of the perianal region. A colorectal surgeon performed 2 unsuccessful excisions of the neoplasm at 6 months and 1 month prior to presentation. Biopsy results from the second excision demonstrated persistent perianal SCCIS with positive margins (Figure 1). The patient was referred to our clinic to discuss Mohs micrographic surgery versus nonsurgical treatment options. The patient’s medical history was remarkable for an abnormal Papanicolaou test 10 years prior, which resulted in cervical cryotherapy.

|
|
Physical examination revealed a 2×1-cm pink scar with peripheral scaling on the right anal verge (Figure 2A). Potential therapies discussed with the patient included Mohs surgery, ablative laser treatment, and nonsurgical treatment with ALA-PDT in combination with topical imiquimod cream 5%. To avoid further invasive treatments, we decided to administer a 3-week course of topical imiquimod cream 5% applied 3 times weekly to the entire affected area, followed 1 week later with a treatment of ALA-PDT. The incubation time was 6 hours. After completing 4 cycles of this therapeutic regimen, the erythema and scaling resolved (Figure 2B). The patient did not note any remarkable side effects associated with imiquimod but reported progressively resolving pain in the week following each ALA-PDT treatment, with the worst pain occurring during the first 48 hours after treatment. Three posttreatment scout biopsies revealed no evidence of residual perianal SCCIS. Two years after the negative biopsies, ongoing physical examinations demonstrated no evidence of clinical recurrence. Physicians in the dermatology and gynecology departments are following her closely.
Comment
Wide surgical excision is currently recommended as first-line therapy for perianal SCCIS.1,3,4 Unfortunately, surgical excision has been associated with difficulty in achieving disease-free margins, recurrence rates as high as 31%, and substantial morbidity.1,4 Other nonsurgical treatment modalities have been investigated for treating this neoplasm to simultaneously reduce recurrence rates while minimizing structural and functional damage to the treatment area, including radiotherapy, imiquimod, laser ablation, and ALA-PDT.1,3,4,5
In particular, imiquimod and ALA-PDT have shown promise as monotherapies for the treatment of perianal SCCIS, with several case reports describing complete resolution, low rates of recurrence, and preserved structure and function of the surrounding tissue after therapy1,3,4; however, recurrence of perianal SCCIS is still known to occur after monotherapy with either ALA-PDT or imiquimod.5,6
In contrast to noninvasive monotherapies, the literature is largely devoid of reports of noninvasive combination treatments used for perianal SCCIS. Our case represents successful use of a combination of imiquimod and ALA-PDT to treat persistent perianal SCCIS.
Conclusion
Although further research is necessary, the therapeutic success presented in our case suggests noninvasive combination therapy with imiquimod and ALA-PDT may represent a viable alternative to both surgical excision and noninvasive monotherapies for the treatment of perianal SCCIS.
Acknowledgement—The authors would like to thank Alejandro Gru, MD, Columbus, Ohio, for his review of the pathology slides for this case.
1. van Egmond S, Hoedemaker C, Sinclair R. Successful treatment of perianal Bowen’s disease with imiquimod. Int J Dermatol. 2007;46:318-319.
2. Abbasakoor F, Boulos PB. Anal intraepithelial neoplasia. Br J Surg. 2005;92:277-290.
3. Petrelli NJ, Cebollero JA, Rodriguez-Bigas M, et al. Photodynamic therapy in the management of neoplasms of the perianal skin. Arch Surg. 1992;127:1436-1438.
4. Wietfeldt E, Thiele J. Malignancies of the anal margin and perianal skin. Clin Colon Rectal Surg. 2009;22:127-135.
5. Pineda C, Welton M. Management of anal squamous intraepithelial lesions. Clin Colon Rectal Surg. 2009;22:94-101.
6. Runfola MA, Weber TK, Rodriguez-Bigas MA, et al. Photodynamic therapy for residual neoplasms of the perianal skin. Dis Colon Rectum. 2000;43:499-502.
1. van Egmond S, Hoedemaker C, Sinclair R. Successful treatment of perianal Bowen’s disease with imiquimod. Int J Dermatol. 2007;46:318-319.
2. Abbasakoor F, Boulos PB. Anal intraepithelial neoplasia. Br J Surg. 2005;92:277-290.
3. Petrelli NJ, Cebollero JA, Rodriguez-Bigas M, et al. Photodynamic therapy in the management of neoplasms of the perianal skin. Arch Surg. 1992;127:1436-1438.
4. Wietfeldt E, Thiele J. Malignancies of the anal margin and perianal skin. Clin Colon Rectal Surg. 2009;22:127-135.
5. Pineda C, Welton M. Management of anal squamous intraepithelial lesions. Clin Colon Rectal Surg. 2009;22:94-101.
6. Runfola MA, Weber TK, Rodriguez-Bigas MA, et al. Photodynamic therapy for residual neoplasms of the perianal skin. Dis Colon Rectum. 2000;43:499-502.
Practice Points
- Perianal squamous cell carcinoma in situ (SCCIS) is an uncommon malignancy with most cases related to human papillomavirus infection.
- Treatment of perianal SCCIS is important to prevent progression to invasive squamous cell carcinoma.
- Treatment usually is surgical, but nonsurgical modalities such as combination therapy with imiquimod and 5-aminolevulinic acid–based photodynamic therapy should be considered in patients who are poor surgical candidates or in cases of recurrences after excisional surgery.
Cutaneous Adenosquamous Carcinoma: A Rare Neoplasm With Biphasic Differentiation
Case Report
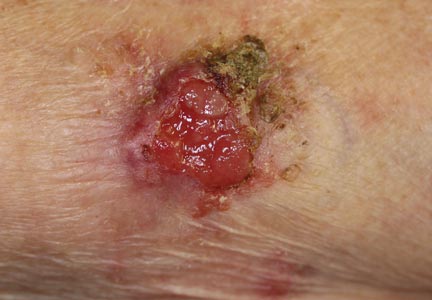
An 85-year-old woman presented with a painless red plaque on the right bicep of 5 years’ duration. The patient had not seen a physician in the last 63 years and had unsuccessfully attempted to treat the plaque by occlusion with an adhesive bandage. A review of systems was negative for pain, pruritus, bleeding, fever, unexplained weight loss, and night sweats. Physical examination revealed a raised, 2×4×1-cm, red, nontender, ulcerated plaque with slight exudate and gelatinous texture on the right bicep (Figure 1). Full-body skin examination revealed erythema and swelling of the right wrist and forearm consistent with cellulitis as well as tinea pedis and onychomycosis of the toenails of both feet.
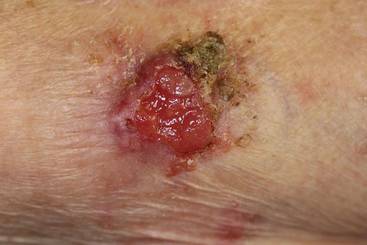
Figure 1. A raised, red, nontender, ulcerated plaque with slight exudate and gelatinous texture on the right bicep. 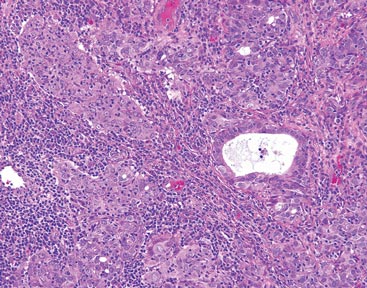
Figure 2. The tumor was comprised of gland-forming cells and exhibited crowding, pleomorphism, enlarged hyperchromatic nuclei, and mitotic division figures (H&E, original magnification ×100). 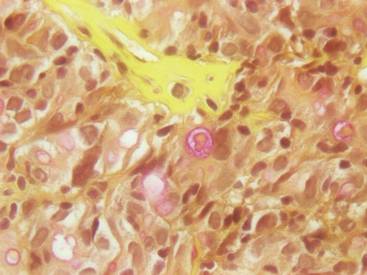
Figure 3. Mucicarmine staining highlighted sialomucin within the glandular component (original magnification ×400). |
Hematoxylin and eosin as well as mucicarmine staining of a shave biopsy from the lesion demonstrated an invasive epithelial neoplasm comprised of squamoid and gland-forming cells broadly attached to the epidermis, which suggested a primary cutaneous origin (Figures 2 and 3). The tumor cells were arranged in infiltrating cords and nests; they exhibited crowding, pleomorphism, enlarged hyperchromatic nuclei, and mitotic division figures. Epithelial mucin (sialomucin) within the glandular component was highlighted on mucicarmine staining. The gland-forming segment of the tumor was strongly positive for cytokeratin (CK) 7. Gastrointestinal tumors were excluded on negative CDX2 and CK20 staining, pulmonary and thyroid tumors were excluded on negative thyroid transcription factor 1 staining, and endometrial and ovarian tumors were excluded with negative estrogen receptor staining. On physical examination the breasts were soft, nontender, and without deformity. A chest radiograph demonstrated normal heart size and pulmonary vasculature with mild bibasilar atelectasis and no areas of consolidation. Given these clinical findings along with a negative history of cancer and negative estrogen receptor staining, breast cancer was excluded from the differential diagnosis, and the diagnosis of cASC was made. The tumor was excised using Mohs micrographic surgery and was free of recurrence at 6- and 12-month follow-up.
Comment
Primary cutaneous adenosquamous carcinoma (cASC) is an aggressive subtype of squamous cell carcinoma that was first described in 1985.1 It typically presents as an erythematous, indurated, keratotic papule or plaque with a predilection for the face, scalp, and upper extremities of immunocompromised individuals and elderly men.2,3 Biopsies generally demonstrate a malignant epithelial neoplasm arising from the epidermis and exhibiting squamous and glandular differentiation. The glandular segment usually is indistinguishable from adenocarcinoma and can be highlighted on CK7, carcinoembryonic antigen, mucicarmine, and periodic acid–Schiff staining. The squamous segment typically is indistinguishable from squamous cell carcinoma and shows aberrant keratinization and intercellular bridges. Tumors often are deeply invasive, poorly differentiated, and associated with a desmoplastic stromal reaction. Local recurrence rates are between 22% and 26%,4 but metastasis is rare. Surgical excision is the mainstay of therapy. When clear margins cannot be obtained using Mohs micrographic surgery, adjuvant external beam radiation therapy and epidermal growth factor receptor inhibitors can be used to treat locally recurrent cASCs.2
The differential diagnosis for cASC includes cutaneous mucoepidermoid carcinoma, cutaneous acantholytic squamous cell carcinoma, and cutaneous manifestations of metastatic visceral adenosquamous carcinoma. Mucoepidermoid carcinoma sometimes is used interchangeably with cASC in the literature, but it is a different cutaneous neoplasm that forms goblet cells, intermediate cells, and squamous cells. It is considered the cutaneous analogue of salivary gland mucoepidermoid carcinoma and does not exhibit the anaplasia, stromal desmoplasia, and aggressive course of cASC.5 The acantholytic subtype of squamous cell carcinoma forms glandlike spaces due to poor adhesion between keratinocytes, but the glandlike spaces do not form mucin or stain positive for CK7 or carcinoembryonic antigen. Adenosquamous carcinomas are well recognized in the lungs, breasts, genitourinary tract, pancreas, and gastroenteric system. Visceral tumor metastasis to the skin should be excluded by appropriate screening.
Conclusion
Although cASCs are not commonly encountered in clinical practice, accurate diagnosis of these lesions is important due to their potentially aggressive behavior. Misdiagnosis and improper treatment could be attributed to lack of awareness of this type of lesion.
1. Weidner N, Foucar E. Adenosquamous carcinoma of the skin. an aggressive mucin-and gland-forming squamous carcinoma. Arch Dermatol. 1985;121:775-779.
2. Fu JM, McCalmont T, Siegrid YS. Adenosquamous carcinoma of the skin: a case series. Arch Dermatol. 2009;145:1152-1158.
3. Ko JK, Leffel DJ, McNiff JM. Adenosquamous carcinoma: a report of 9 cases with p63 and cytokeratin 5/6 staining. J Cutan Pathol. 2009;36:448-452.
4. Banks ER, Cooper PH. Adenosquamous carcinoma of the skin: a report of 10 cases. J Cutan Pathol. 1991;18:227-234.
5. Riedlinger WF, Hurley MY, Dehner LP, et al. Muco-epidermoid carcinoma of the skin: a distinct entity from adenosquamous carcinoma: a case study with a review of the literature. Am J Surg Pathol. 2005;29:131-135.
Case Report
An 85-year-old woman presented with a painless red plaque on the right bicep of 5 years’ duration. The patient had not seen a physician in the last 63 years and had unsuccessfully attempted to treat the plaque by occlusion with an adhesive bandage. A review of systems was negative for pain, pruritus, bleeding, fever, unexplained weight loss, and night sweats. Physical examination revealed a raised, 2×4×1-cm, red, nontender, ulcerated plaque with slight exudate and gelatinous texture on the right bicep (Figure 1). Full-body skin examination revealed erythema and swelling of the right wrist and forearm consistent with cellulitis as well as tinea pedis and onychomycosis of the toenails of both feet.

Figure 1. A raised, red, nontender, ulcerated plaque with slight exudate and gelatinous texture on the right bicep. 
Figure 2. The tumor was comprised of gland-forming cells and exhibited crowding, pleomorphism, enlarged hyperchromatic nuclei, and mitotic division figures (H&E, original magnification ×100). 
Figure 3. Mucicarmine staining highlighted sialomucin within the glandular component (original magnification ×400). |
Hematoxylin and eosin as well as mucicarmine staining of a shave biopsy from the lesion demonstrated an invasive epithelial neoplasm comprised of squamoid and gland-forming cells broadly attached to the epidermis, which suggested a primary cutaneous origin (Figures 2 and 3). The tumor cells were arranged in infiltrating cords and nests; they exhibited crowding, pleomorphism, enlarged hyperchromatic nuclei, and mitotic division figures. Epithelial mucin (sialomucin) within the glandular component was highlighted on mucicarmine staining. The gland-forming segment of the tumor was strongly positive for cytokeratin (CK) 7. Gastrointestinal tumors were excluded on negative CDX2 and CK20 staining, pulmonary and thyroid tumors were excluded on negative thyroid transcription factor 1 staining, and endometrial and ovarian tumors were excluded with negative estrogen receptor staining. On physical examination the breasts were soft, nontender, and without deformity. A chest radiograph demonstrated normal heart size and pulmonary vasculature with mild bibasilar atelectasis and no areas of consolidation. Given these clinical findings along with a negative history of cancer and negative estrogen receptor staining, breast cancer was excluded from the differential diagnosis, and the diagnosis of cASC was made. The tumor was excised using Mohs micrographic surgery and was free of recurrence at 6- and 12-month follow-up.
Comment
Primary cutaneous adenosquamous carcinoma (cASC) is an aggressive subtype of squamous cell carcinoma that was first described in 1985.1 It typically presents as an erythematous, indurated, keratotic papule or plaque with a predilection for the face, scalp, and upper extremities of immunocompromised individuals and elderly men.2,3 Biopsies generally demonstrate a malignant epithelial neoplasm arising from the epidermis and exhibiting squamous and glandular differentiation. The glandular segment usually is indistinguishable from adenocarcinoma and can be highlighted on CK7, carcinoembryonic antigen, mucicarmine, and periodic acid–Schiff staining. The squamous segment typically is indistinguishable from squamous cell carcinoma and shows aberrant keratinization and intercellular bridges. Tumors often are deeply invasive, poorly differentiated, and associated with a desmoplastic stromal reaction. Local recurrence rates are between 22% and 26%,4 but metastasis is rare. Surgical excision is the mainstay of therapy. When clear margins cannot be obtained using Mohs micrographic surgery, adjuvant external beam radiation therapy and epidermal growth factor receptor inhibitors can be used to treat locally recurrent cASCs.2
The differential diagnosis for cASC includes cutaneous mucoepidermoid carcinoma, cutaneous acantholytic squamous cell carcinoma, and cutaneous manifestations of metastatic visceral adenosquamous carcinoma. Mucoepidermoid carcinoma sometimes is used interchangeably with cASC in the literature, but it is a different cutaneous neoplasm that forms goblet cells, intermediate cells, and squamous cells. It is considered the cutaneous analogue of salivary gland mucoepidermoid carcinoma and does not exhibit the anaplasia, stromal desmoplasia, and aggressive course of cASC.5 The acantholytic subtype of squamous cell carcinoma forms glandlike spaces due to poor adhesion between keratinocytes, but the glandlike spaces do not form mucin or stain positive for CK7 or carcinoembryonic antigen. Adenosquamous carcinomas are well recognized in the lungs, breasts, genitourinary tract, pancreas, and gastroenteric system. Visceral tumor metastasis to the skin should be excluded by appropriate screening.
Conclusion
Although cASCs are not commonly encountered in clinical practice, accurate diagnosis of these lesions is important due to their potentially aggressive behavior. Misdiagnosis and improper treatment could be attributed to lack of awareness of this type of lesion.
Case Report
An 85-year-old woman presented with a painless red plaque on the right bicep of 5 years’ duration. The patient had not seen a physician in the last 63 years and had unsuccessfully attempted to treat the plaque by occlusion with an adhesive bandage. A review of systems was negative for pain, pruritus, bleeding, fever, unexplained weight loss, and night sweats. Physical examination revealed a raised, 2×4×1-cm, red, nontender, ulcerated plaque with slight exudate and gelatinous texture on the right bicep (Figure 1). Full-body skin examination revealed erythema and swelling of the right wrist and forearm consistent with cellulitis as well as tinea pedis and onychomycosis of the toenails of both feet.

Figure 1. A raised, red, nontender, ulcerated plaque with slight exudate and gelatinous texture on the right bicep. 
Figure 2. The tumor was comprised of gland-forming cells and exhibited crowding, pleomorphism, enlarged hyperchromatic nuclei, and mitotic division figures (H&E, original magnification ×100). 
Figure 3. Mucicarmine staining highlighted sialomucin within the glandular component (original magnification ×400). |
Hematoxylin and eosin as well as mucicarmine staining of a shave biopsy from the lesion demonstrated an invasive epithelial neoplasm comprised of squamoid and gland-forming cells broadly attached to the epidermis, which suggested a primary cutaneous origin (Figures 2 and 3). The tumor cells were arranged in infiltrating cords and nests; they exhibited crowding, pleomorphism, enlarged hyperchromatic nuclei, and mitotic division figures. Epithelial mucin (sialomucin) within the glandular component was highlighted on mucicarmine staining. The gland-forming segment of the tumor was strongly positive for cytokeratin (CK) 7. Gastrointestinal tumors were excluded on negative CDX2 and CK20 staining, pulmonary and thyroid tumors were excluded on negative thyroid transcription factor 1 staining, and endometrial and ovarian tumors were excluded with negative estrogen receptor staining. On physical examination the breasts were soft, nontender, and without deformity. A chest radiograph demonstrated normal heart size and pulmonary vasculature with mild bibasilar atelectasis and no areas of consolidation. Given these clinical findings along with a negative history of cancer and negative estrogen receptor staining, breast cancer was excluded from the differential diagnosis, and the diagnosis of cASC was made. The tumor was excised using Mohs micrographic surgery and was free of recurrence at 6- and 12-month follow-up.
Comment
Primary cutaneous adenosquamous carcinoma (cASC) is an aggressive subtype of squamous cell carcinoma that was first described in 1985.1 It typically presents as an erythematous, indurated, keratotic papule or plaque with a predilection for the face, scalp, and upper extremities of immunocompromised individuals and elderly men.2,3 Biopsies generally demonstrate a malignant epithelial neoplasm arising from the epidermis and exhibiting squamous and glandular differentiation. The glandular segment usually is indistinguishable from adenocarcinoma and can be highlighted on CK7, carcinoembryonic antigen, mucicarmine, and periodic acid–Schiff staining. The squamous segment typically is indistinguishable from squamous cell carcinoma and shows aberrant keratinization and intercellular bridges. Tumors often are deeply invasive, poorly differentiated, and associated with a desmoplastic stromal reaction. Local recurrence rates are between 22% and 26%,4 but metastasis is rare. Surgical excision is the mainstay of therapy. When clear margins cannot be obtained using Mohs micrographic surgery, adjuvant external beam radiation therapy and epidermal growth factor receptor inhibitors can be used to treat locally recurrent cASCs.2
The differential diagnosis for cASC includes cutaneous mucoepidermoid carcinoma, cutaneous acantholytic squamous cell carcinoma, and cutaneous manifestations of metastatic visceral adenosquamous carcinoma. Mucoepidermoid carcinoma sometimes is used interchangeably with cASC in the literature, but it is a different cutaneous neoplasm that forms goblet cells, intermediate cells, and squamous cells. It is considered the cutaneous analogue of salivary gland mucoepidermoid carcinoma and does not exhibit the anaplasia, stromal desmoplasia, and aggressive course of cASC.5 The acantholytic subtype of squamous cell carcinoma forms glandlike spaces due to poor adhesion between keratinocytes, but the glandlike spaces do not form mucin or stain positive for CK7 or carcinoembryonic antigen. Adenosquamous carcinomas are well recognized in the lungs, breasts, genitourinary tract, pancreas, and gastroenteric system. Visceral tumor metastasis to the skin should be excluded by appropriate screening.
Conclusion
Although cASCs are not commonly encountered in clinical practice, accurate diagnosis of these lesions is important due to their potentially aggressive behavior. Misdiagnosis and improper treatment could be attributed to lack of awareness of this type of lesion.
1. Weidner N, Foucar E. Adenosquamous carcinoma of the skin. an aggressive mucin-and gland-forming squamous carcinoma. Arch Dermatol. 1985;121:775-779.
2. Fu JM, McCalmont T, Siegrid YS. Adenosquamous carcinoma of the skin: a case series. Arch Dermatol. 2009;145:1152-1158.
3. Ko JK, Leffel DJ, McNiff JM. Adenosquamous carcinoma: a report of 9 cases with p63 and cytokeratin 5/6 staining. J Cutan Pathol. 2009;36:448-452.
4. Banks ER, Cooper PH. Adenosquamous carcinoma of the skin: a report of 10 cases. J Cutan Pathol. 1991;18:227-234.
5. Riedlinger WF, Hurley MY, Dehner LP, et al. Muco-epidermoid carcinoma of the skin: a distinct entity from adenosquamous carcinoma: a case study with a review of the literature. Am J Surg Pathol. 2005;29:131-135.
1. Weidner N, Foucar E. Adenosquamous carcinoma of the skin. an aggressive mucin-and gland-forming squamous carcinoma. Arch Dermatol. 1985;121:775-779.
2. Fu JM, McCalmont T, Siegrid YS. Adenosquamous carcinoma of the skin: a case series. Arch Dermatol. 2009;145:1152-1158.
3. Ko JK, Leffel DJ, McNiff JM. Adenosquamous carcinoma: a report of 9 cases with p63 and cytokeratin 5/6 staining. J Cutan Pathol. 2009;36:448-452.
4. Banks ER, Cooper PH. Adenosquamous carcinoma of the skin: a report of 10 cases. J Cutan Pathol. 1991;18:227-234.
5. Riedlinger WF, Hurley MY, Dehner LP, et al. Muco-epidermoid carcinoma of the skin: a distinct entity from adenosquamous carcinoma: a case study with a review of the literature. Am J Surg Pathol. 2005;29:131-135.
Practice Points
- Cutaneous adenosquamous carcinoma (cASC) is an extremely rare malignant neoplasm with histologic similarities to both squamous cell carcinoma and adenocarcinoma.
- Mohs micrographic surgery for excision is recommended; however, adjuvant external beam radiation therapy and epidermal growth factor receptor inhibitors also have been used to treat locally recurrent cASCs.
Leser-Trélat Sign: A Paraneoplastic Process?
To the Editor:
Leser-Trélat sign is a rare skin condition characterized by the sudden appearance of seborrheic keratoses that rapidly increase in number and size within weeks to months. Co-occurrence has been reported with a large number of malignancies, particularly adenocarcinoma and lymphoma. We present a case of Leser-Trélat sign that was not associated with an underlying malignancy.
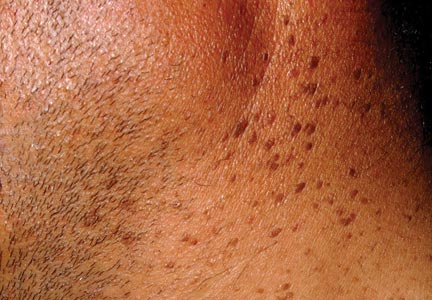
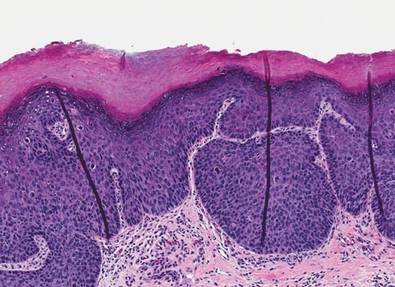
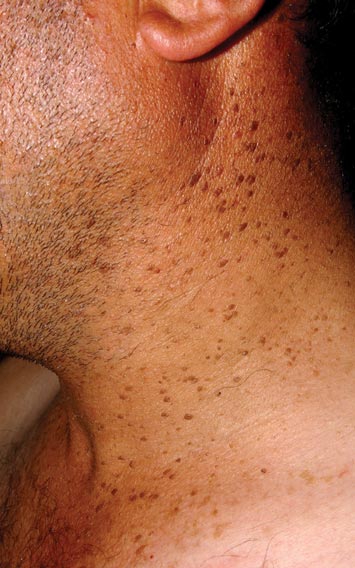
A 44-year-old man was admitted to our dermatology outpatient department with a serpigo on the neck that had grown rapidly in the last month. His medical history and family history were unremarkable. Dermatologic examination revealed numerous 3- to 4-mm brown and slightly verrucous papules on the neck (Figure 1). A punch biopsy of the lesion showed acanthosis of predominantly basaloid cells, papillomatosis, and hyperkeratosis, as well as the presence of characteristic horn cysts (Figure 2). He was tested for possible underlying internal malignancy. Liver and kidney function tests, electrolyte count, protein electrophoresis, and whole blood and urine tests were within reference range. Chest radiography and abdominal ultrasonography revealed no signs of pathology. The erythrocyte sedimentation rate was 20 mm/h (reference range, 0–20 mm/h) and tests for hepatitis, human immunodeficiency virus, and syphilis were negative. Abdominal, cranial, and thorax computed tomography revealed no abnormalities. Otolaryngologic examinations also were negative. Additional endoscopic analyses, esophagogastroduodenoscopy, and colonoscopy revealed no abnormalities. At 1-year follow-up, the seborrheic keratoses remained unchanged. He has remained in good health without specific signs or symptoms suggestive of an underlying malignancy.
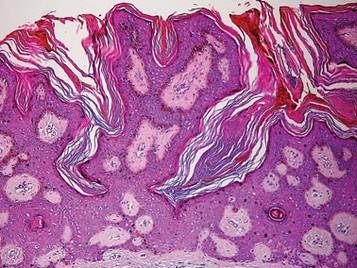
Paraneoplastic syndromes are associated with malignancy but progress without connection to a primary tumor or metastasis and form a group of clinical manifestations. The characteristic progress of paraneoplastic syndromes shows parallelism with the progression of the tumor. The mechanism underlying the development is not known, though the actions of bioactive substances that cause responses in the tumor, such as polypeptide hormones, hormonelike peptides, antibodies or immune complexes, and cytokines or growth factors, have been implicated.1
Although the term paraneoplastic syndrome commonly is used for Leser-Trélat sign, we do not believe it is accurate. As Fink et al2 and Schwengle et al3 indicated, the possibility of the co-occurrence being fortuitous is high. Showing a parallel progress of malignancy with paraneoplastic dermatosis requires that the paraneoplastic syndrome also diminish when the tumor is cured.4 It should then reappear with cancer recurrence or metastasis, which has not been exhibited in many case presentations in the literature.3 Disease regression was observed in only 1 of 3 seborrheic keratosis cases after primary cancer treatment.5
In patients with a malignancy, the sudden increase in seborrheic keratosis is based exclusively on the subjective evaluation of the patient, which may not be reliable. Schwengle et al3 stated that this sudden increase can be related to the awareness level of the patient who had a cancer diagnosis. Bräuer et al6 stated that no plausible definition distinguishes eruptive versus common seborrheic keratoses.
As a result, the results regarding the relationship between malignancy and Leser-Trélat sign are conflicting, and no strong evidence supports the presence of the sign. Only case reports have suggested that Leser-Trélat sign accompanies malignancy. Studies investigating its etiopathogenesis have not revealed a substance that has been released from or as a response to a tumor.
We believe that the presence of eruptive seborrheic keratosis does not necessitate screening for underlying internal malignancies.
- Cohen PR. Paraneoplastic dermatopathology: cutaneous paraneoplastic syndromes. Adv Dermatol. 1996;11:215-252.
- Fink AM, Filz D, Krajnik G, et al. Seborrhoeic keratoses in patients with internal malignancies: a case-control study with a prospective accrual of patients. J Eur Acad Dermatol Venereol. 2009;23:1316-1319.
- Schwengle LE, Rampen FH, Wobbes T. Seborrhoeic keratoses and internal malignancies. a case control study. Clin Exp Dermatol. 1988;13:177-179.
- Curth HO. Skin lesions and internal carcinoma. In: Andrade S, Gumport S, Popkin GL, et al, eds. Cancer of the Skin: Biology, Diagnosis, and Management. Vol 2. Philadelphia, PA: WB Saunders; 1976:1308-1341.
- Heaphy MR Jr, Millns JL, Schroeter AL. The sign of Leser-Trélat in a case of adenocarcinoma of the lung. J Am Acad Dermatol. 2000;43(2, pt 2):386-390.
- Bräuer J, Happle R, Gieler U. The sign of Leser-Trélat: fact or myth? J Eur Acad Dermatol Venereol. 1992;1:77-80.
To the Editor:
Leser-Trélat sign is a rare skin condition characterized by the sudden appearance of seborrheic keratoses that rapidly increase in number and size within weeks to months. Co-occurrence has been reported with a large number of malignancies, particularly adenocarcinoma and lymphoma. We present a case of Leser-Trélat sign that was not associated with an underlying malignancy.
A 44-year-old man was admitted to our dermatology outpatient department with a serpigo on the neck that had grown rapidly in the last month. His medical history and family history were unremarkable. Dermatologic examination revealed numerous 3- to 4-mm brown and slightly verrucous papules on the neck (Figure 1). A punch biopsy of the lesion showed acanthosis of predominantly basaloid cells, papillomatosis, and hyperkeratosis, as well as the presence of characteristic horn cysts (Figure 2). He was tested for possible underlying internal malignancy. Liver and kidney function tests, electrolyte count, protein electrophoresis, and whole blood and urine tests were within reference range. Chest radiography and abdominal ultrasonography revealed no signs of pathology. The erythrocyte sedimentation rate was 20 mm/h (reference range, 0–20 mm/h) and tests for hepatitis, human immunodeficiency virus, and syphilis were negative. Abdominal, cranial, and thorax computed tomography revealed no abnormalities. Otolaryngologic examinations also were negative. Additional endoscopic analyses, esophagogastroduodenoscopy, and colonoscopy revealed no abnormalities. At 1-year follow-up, the seborrheic keratoses remained unchanged. He has remained in good health without specific signs or symptoms suggestive of an underlying malignancy.


Paraneoplastic syndromes are associated with malignancy but progress without connection to a primary tumor or metastasis and form a group of clinical manifestations. The characteristic progress of paraneoplastic syndromes shows parallelism with the progression of the tumor. The mechanism underlying the development is not known, though the actions of bioactive substances that cause responses in the tumor, such as polypeptide hormones, hormonelike peptides, antibodies or immune complexes, and cytokines or growth factors, have been implicated.1
Although the term paraneoplastic syndrome commonly is used for Leser-Trélat sign, we do not believe it is accurate. As Fink et al2 and Schwengle et al3 indicated, the possibility of the co-occurrence being fortuitous is high. Showing a parallel progress of malignancy with paraneoplastic dermatosis requires that the paraneoplastic syndrome also diminish when the tumor is cured.4 It should then reappear with cancer recurrence or metastasis, which has not been exhibited in many case presentations in the literature.3 Disease regression was observed in only 1 of 3 seborrheic keratosis cases after primary cancer treatment.5
In patients with a malignancy, the sudden increase in seborrheic keratosis is based exclusively on the subjective evaluation of the patient, which may not be reliable. Schwengle et al3 stated that this sudden increase can be related to the awareness level of the patient who had a cancer diagnosis. Bräuer et al6 stated that no plausible definition distinguishes eruptive versus common seborrheic keratoses.
As a result, the results regarding the relationship between malignancy and Leser-Trélat sign are conflicting, and no strong evidence supports the presence of the sign. Only case reports have suggested that Leser-Trélat sign accompanies malignancy. Studies investigating its etiopathogenesis have not revealed a substance that has been released from or as a response to a tumor.
We believe that the presence of eruptive seborrheic keratosis does not necessitate screening for underlying internal malignancies.
To the Editor:
Leser-Trélat sign is a rare skin condition characterized by the sudden appearance of seborrheic keratoses that rapidly increase in number and size within weeks to months. Co-occurrence has been reported with a large number of malignancies, particularly adenocarcinoma and lymphoma. We present a case of Leser-Trélat sign that was not associated with an underlying malignancy.
A 44-year-old man was admitted to our dermatology outpatient department with a serpigo on the neck that had grown rapidly in the last month. His medical history and family history were unremarkable. Dermatologic examination revealed numerous 3- to 4-mm brown and slightly verrucous papules on the neck (Figure 1). A punch biopsy of the lesion showed acanthosis of predominantly basaloid cells, papillomatosis, and hyperkeratosis, as well as the presence of characteristic horn cysts (Figure 2). He was tested for possible underlying internal malignancy. Liver and kidney function tests, electrolyte count, protein electrophoresis, and whole blood and urine tests were within reference range. Chest radiography and abdominal ultrasonography revealed no signs of pathology. The erythrocyte sedimentation rate was 20 mm/h (reference range, 0–20 mm/h) and tests for hepatitis, human immunodeficiency virus, and syphilis were negative. Abdominal, cranial, and thorax computed tomography revealed no abnormalities. Otolaryngologic examinations also were negative. Additional endoscopic analyses, esophagogastroduodenoscopy, and colonoscopy revealed no abnormalities. At 1-year follow-up, the seborrheic keratoses remained unchanged. He has remained in good health without specific signs or symptoms suggestive of an underlying malignancy.


Paraneoplastic syndromes are associated with malignancy but progress without connection to a primary tumor or metastasis and form a group of clinical manifestations. The characteristic progress of paraneoplastic syndromes shows parallelism with the progression of the tumor. The mechanism underlying the development is not known, though the actions of bioactive substances that cause responses in the tumor, such as polypeptide hormones, hormonelike peptides, antibodies or immune complexes, and cytokines or growth factors, have been implicated.1
Although the term paraneoplastic syndrome commonly is used for Leser-Trélat sign, we do not believe it is accurate. As Fink et al2 and Schwengle et al3 indicated, the possibility of the co-occurrence being fortuitous is high. Showing a parallel progress of malignancy with paraneoplastic dermatosis requires that the paraneoplastic syndrome also diminish when the tumor is cured.4 It should then reappear with cancer recurrence or metastasis, which has not been exhibited in many case presentations in the literature.3 Disease regression was observed in only 1 of 3 seborrheic keratosis cases after primary cancer treatment.5
In patients with a malignancy, the sudden increase in seborrheic keratosis is based exclusively on the subjective evaluation of the patient, which may not be reliable. Schwengle et al3 stated that this sudden increase can be related to the awareness level of the patient who had a cancer diagnosis. Bräuer et al6 stated that no plausible definition distinguishes eruptive versus common seborrheic keratoses.
As a result, the results regarding the relationship between malignancy and Leser-Trélat sign are conflicting, and no strong evidence supports the presence of the sign. Only case reports have suggested that Leser-Trélat sign accompanies malignancy. Studies investigating its etiopathogenesis have not revealed a substance that has been released from or as a response to a tumor.
We believe that the presence of eruptive seborrheic keratosis does not necessitate screening for underlying internal malignancies.
- Cohen PR. Paraneoplastic dermatopathology: cutaneous paraneoplastic syndromes. Adv Dermatol. 1996;11:215-252.
- Fink AM, Filz D, Krajnik G, et al. Seborrhoeic keratoses in patients with internal malignancies: a case-control study with a prospective accrual of patients. J Eur Acad Dermatol Venereol. 2009;23:1316-1319.
- Schwengle LE, Rampen FH, Wobbes T. Seborrhoeic keratoses and internal malignancies. a case control study. Clin Exp Dermatol. 1988;13:177-179.
- Curth HO. Skin lesions and internal carcinoma. In: Andrade S, Gumport S, Popkin GL, et al, eds. Cancer of the Skin: Biology, Diagnosis, and Management. Vol 2. Philadelphia, PA: WB Saunders; 1976:1308-1341.
- Heaphy MR Jr, Millns JL, Schroeter AL. The sign of Leser-Trélat in a case of adenocarcinoma of the lung. J Am Acad Dermatol. 2000;43(2, pt 2):386-390.
- Bräuer J, Happle R, Gieler U. The sign of Leser-Trélat: fact or myth? J Eur Acad Dermatol Venereol. 1992;1:77-80.
- Cohen PR. Paraneoplastic dermatopathology: cutaneous paraneoplastic syndromes. Adv Dermatol. 1996;11:215-252.
- Fink AM, Filz D, Krajnik G, et al. Seborrhoeic keratoses in patients with internal malignancies: a case-control study with a prospective accrual of patients. J Eur Acad Dermatol Venereol. 2009;23:1316-1319.
- Schwengle LE, Rampen FH, Wobbes T. Seborrhoeic keratoses and internal malignancies. a case control study. Clin Exp Dermatol. 1988;13:177-179.
- Curth HO. Skin lesions and internal carcinoma. In: Andrade S, Gumport S, Popkin GL, et al, eds. Cancer of the Skin: Biology, Diagnosis, and Management. Vol 2. Philadelphia, PA: WB Saunders; 1976:1308-1341.
- Heaphy MR Jr, Millns JL, Schroeter AL. The sign of Leser-Trélat in a case of adenocarcinoma of the lung. J Am Acad Dermatol. 2000;43(2, pt 2):386-390.
- Bräuer J, Happle R, Gieler U. The sign of Leser-Trélat: fact or myth? J Eur Acad Dermatol Venereol. 1992;1:77-80.
Drink more coffee to prevent a second BCC
AMSTERDAM – Liberal coffee consumption was independently associated with a reduced risk of developing a second basal cell carcinoma in individuals who’ve already had a first in an analysis from the Rotterdam Study.
“This might seem surprising, but several recent studies done in both humans and mice have shown this protective effect as well. The mechanism behind it is still unclear, but we think that caffeine might help to prevent UV-induced carcinogenesis,” Dr. Joris Verkouteren said in presenting the Rotterdam Study findings at the annual congress of the European Academy of Dermatology and Venereology.
Drinking more coffee was the only protective factor identified in the study. In a multivariate regression analysis, it was associated with a 30% reduction in the relative risk of developing at least a second BCC.
In contrast, two major predictors of increased likelihood of a second BCC emerged. The strongest was having more than one primary BCC at initial presentation, which carried an associated 2.5-fold increased risk. The other risk factor was older age at diagnosis of the first BCC, associated with a 60% increase in risk, reported Dr. Verkouteren of Ermasus University, Rotterdam.
Basal cell carcinoma is the most common of all malignancies. Roughly one-third of patients who present with a first one will develop a second BCC, most often within the next couple years. This makes BCC a logical target for secondary prevention measures. The purpose of this analysis of the Rotterdam Study was to identify factors to help physicians differentiate between patients who are likely to have just one primary BCC and those who will go on to develop multiple BCCs. This would aid clinicians in selecting patients for closer monitoring and in identifying good candidates for aggressive secondary prevention interventions, such as photodynamic therapy to address the field cancerization which results from lifetime exposure to UV, he explained.
The Rotterdam Study is an ongoing prospective population-based cohort study started in 1989. This analysis included 14,976 Rotterdam residents, all at least age 45 years at entry, who have been evaluated by physican examination and detailed questionnaires every 3-4 years. Through linkage to the Dutch national histopathologic database, it was possible to identify all study participants diagnosed with BCC.
The 784 study participants who developed a first primary BCC were prospectively followed for a median of more than 4 years, during which 293 developed at least a second BCC.
Individuals who developed a second BCC at least 6 months after their first drank an average of 2 cups of joe daily, while those who did not have another BCC quaffed an average of 5 cups per day.
The median age at the time of diagnosis of a first BCC was 67.6 years in individuals who didn’t develop a second one, compared to 80.7 years in those who did.
A particularly noteworthy study finding, in Dr. Verkouteren’s view, was that many of the well-established risk factors for a first BCC were not significant predictors of a second one. A tendency to sunburn easily, light hair and eye color, male gender, smoking, a history of outdoor work -- none of these factors proved helpful in predicting which patients with a first BCC would develop another.
Dr. Tamar Nijsten, senior coinvestigator in the study, said that he is skeptical of Dr. Verkouteren’s assertion that the observed association between greater coffee consumption and reduced risk of a second BCC is probably due to a some intrinsic anti-carcinogenic effect of caffeine.
“There are people – and I am one of them – who believe that coffee consumption is associated with health status. So people who drink more coffee have, on average, a more healthy lifestyle,” said Dr. Nijsten, professor and chair of the department of dermatology at Erasmus University Medical Center, Rotterdam.
Dr. Verkouteren and Dr. Nijsten reported having no financial conflicts regarding the Rotterdam Study.
AMSTERDAM – Liberal coffee consumption was independently associated with a reduced risk of developing a second basal cell carcinoma in individuals who’ve already had a first in an analysis from the Rotterdam Study.
“This might seem surprising, but several recent studies done in both humans and mice have shown this protective effect as well. The mechanism behind it is still unclear, but we think that caffeine might help to prevent UV-induced carcinogenesis,” Dr. Joris Verkouteren said in presenting the Rotterdam Study findings at the annual congress of the European Academy of Dermatology and Venereology.
Drinking more coffee was the only protective factor identified in the study. In a multivariate regression analysis, it was associated with a 30% reduction in the relative risk of developing at least a second BCC.
In contrast, two major predictors of increased likelihood of a second BCC emerged. The strongest was having more than one primary BCC at initial presentation, which carried an associated 2.5-fold increased risk. The other risk factor was older age at diagnosis of the first BCC, associated with a 60% increase in risk, reported Dr. Verkouteren of Ermasus University, Rotterdam.
Basal cell carcinoma is the most common of all malignancies. Roughly one-third of patients who present with a first one will develop a second BCC, most often within the next couple years. This makes BCC a logical target for secondary prevention measures. The purpose of this analysis of the Rotterdam Study was to identify factors to help physicians differentiate between patients who are likely to have just one primary BCC and those who will go on to develop multiple BCCs. This would aid clinicians in selecting patients for closer monitoring and in identifying good candidates for aggressive secondary prevention interventions, such as photodynamic therapy to address the field cancerization which results from lifetime exposure to UV, he explained.
The Rotterdam Study is an ongoing prospective population-based cohort study started in 1989. This analysis included 14,976 Rotterdam residents, all at least age 45 years at entry, who have been evaluated by physican examination and detailed questionnaires every 3-4 years. Through linkage to the Dutch national histopathologic database, it was possible to identify all study participants diagnosed with BCC.
The 784 study participants who developed a first primary BCC were prospectively followed for a median of more than 4 years, during which 293 developed at least a second BCC.
Individuals who developed a second BCC at least 6 months after their first drank an average of 2 cups of joe daily, while those who did not have another BCC quaffed an average of 5 cups per day.
The median age at the time of diagnosis of a first BCC was 67.6 years in individuals who didn’t develop a second one, compared to 80.7 years in those who did.
A particularly noteworthy study finding, in Dr. Verkouteren’s view, was that many of the well-established risk factors for a first BCC were not significant predictors of a second one. A tendency to sunburn easily, light hair and eye color, male gender, smoking, a history of outdoor work -- none of these factors proved helpful in predicting which patients with a first BCC would develop another.
Dr. Tamar Nijsten, senior coinvestigator in the study, said that he is skeptical of Dr. Verkouteren’s assertion that the observed association between greater coffee consumption and reduced risk of a second BCC is probably due to a some intrinsic anti-carcinogenic effect of caffeine.
“There are people – and I am one of them – who believe that coffee consumption is associated with health status. So people who drink more coffee have, on average, a more healthy lifestyle,” said Dr. Nijsten, professor and chair of the department of dermatology at Erasmus University Medical Center, Rotterdam.
Dr. Verkouteren and Dr. Nijsten reported having no financial conflicts regarding the Rotterdam Study.
AMSTERDAM – Liberal coffee consumption was independently associated with a reduced risk of developing a second basal cell carcinoma in individuals who’ve already had a first in an analysis from the Rotterdam Study.
“This might seem surprising, but several recent studies done in both humans and mice have shown this protective effect as well. The mechanism behind it is still unclear, but we think that caffeine might help to prevent UV-induced carcinogenesis,” Dr. Joris Verkouteren said in presenting the Rotterdam Study findings at the annual congress of the European Academy of Dermatology and Venereology.
Drinking more coffee was the only protective factor identified in the study. In a multivariate regression analysis, it was associated with a 30% reduction in the relative risk of developing at least a second BCC.
In contrast, two major predictors of increased likelihood of a second BCC emerged. The strongest was having more than one primary BCC at initial presentation, which carried an associated 2.5-fold increased risk. The other risk factor was older age at diagnosis of the first BCC, associated with a 60% increase in risk, reported Dr. Verkouteren of Ermasus University, Rotterdam.
Basal cell carcinoma is the most common of all malignancies. Roughly one-third of patients who present with a first one will develop a second BCC, most often within the next couple years. This makes BCC a logical target for secondary prevention measures. The purpose of this analysis of the Rotterdam Study was to identify factors to help physicians differentiate between patients who are likely to have just one primary BCC and those who will go on to develop multiple BCCs. This would aid clinicians in selecting patients for closer monitoring and in identifying good candidates for aggressive secondary prevention interventions, such as photodynamic therapy to address the field cancerization which results from lifetime exposure to UV, he explained.
The Rotterdam Study is an ongoing prospective population-based cohort study started in 1989. This analysis included 14,976 Rotterdam residents, all at least age 45 years at entry, who have been evaluated by physican examination and detailed questionnaires every 3-4 years. Through linkage to the Dutch national histopathologic database, it was possible to identify all study participants diagnosed with BCC.
The 784 study participants who developed a first primary BCC were prospectively followed for a median of more than 4 years, during which 293 developed at least a second BCC.
Individuals who developed a second BCC at least 6 months after their first drank an average of 2 cups of joe daily, while those who did not have another BCC quaffed an average of 5 cups per day.
The median age at the time of diagnosis of a first BCC was 67.6 years in individuals who didn’t develop a second one, compared to 80.7 years in those who did.
A particularly noteworthy study finding, in Dr. Verkouteren’s view, was that many of the well-established risk factors for a first BCC were not significant predictors of a second one. A tendency to sunburn easily, light hair and eye color, male gender, smoking, a history of outdoor work -- none of these factors proved helpful in predicting which patients with a first BCC would develop another.
Dr. Tamar Nijsten, senior coinvestigator in the study, said that he is skeptical of Dr. Verkouteren’s assertion that the observed association between greater coffee consumption and reduced risk of a second BCC is probably due to a some intrinsic anti-carcinogenic effect of caffeine.
“There are people – and I am one of them – who believe that coffee consumption is associated with health status. So people who drink more coffee have, on average, a more healthy lifestyle,” said Dr. Nijsten, professor and chair of the department of dermatology at Erasmus University Medical Center, Rotterdam.
Dr. Verkouteren and Dr. Nijsten reported having no financial conflicts regarding the Rotterdam Study.
AT THE EADV CONGRESS
Key clinical point: The more coffee you drink, the less likely you will develop a second basal cell carcinoma after being diagnosed with a first one.
Major finding: Increased coffee consumption was associated with a 30% relative risk reduction for diagnosis of a second BCC.
Data source: Analysis involving more than 14,000 participants in the Rotterdam Study, an ongoing prospective population-based cohort study.
Disclosures: The presenter reported having no financial conflicts of interest.
Flurbiprofen-Induced Unilateral Eyelid Angioedema
To the Editor:
Flurbiprofen, a member of the phenylalkanoic acid derivative group of nonsteroidal anti-inflammatory drugs (NSAIDs), are commonly used to treat fever, inflammation, and pain of arthritis.1 The exact prevalence of allergic reactions to NSAIDs in the general population is not known. Rhinoconjunctivitis, bronchospasm, urticaria, angioedema, and anaphylaxis can occur as an allergic reaction to NSAIDs. Isolated angioedema following NSAID ingestion typically involves the face, particularly the periorbital skin, lips, and mouth.2 These patients may develop urticaria and/or angioedema only after NSAID ingestion, but they do not have underlying chronic urticaria. We report a rare case of isolated unilateral eyelid angioedema with flurbiprofen.
A 39-year-old man presented with the onset of unilateral angioedema of the left upper eyelid that had developed approximately 30 minutes after taking flurbiprofen (100 mg). He reported frequent use of flurbiprofen for headaches. The patient also had a history of taking aspirin, ibuprofen, diclofenac, etodolac, and naproxen sodium as needed for migraines with no prior angioedema. He had no history of chronic urticaria or allergic disease. The patient was treated with oral pheniramine hydrogen maleate and angioedema resolved after 12 hours. Three days later, the patient used flurbiprofen again for a headache. He was readmitted to our clinic with unilateral angioedema of the left upper eyelid (Figure). The symptoms started approximately 30 minutes after taking flurbiprofen. Angioedema resolved within 1 day with oral pheniramine.
Nonsteroidal anti-inflammatory drugs are the most commonly prescribed class of drugs in the world and are the most common cause of all adverse drug reactions.3 Urticaria, angioedema, and anaphylaxis are common adverse reactions to NSAIDs. The prevalence of urticaria and angioedema to NSAIDs has been reported to be 0.1% to 3% worldwide.4
Angioedema is an abrupt localized swelling of the skin and mucous membranes of the face, lips, mouth, throat, larynx, extremities, and genitalia. Angioedema generally develops over minutes to hours and resolves in 24 to 48 hours.5 Angioedema without urticaria is the clinical syndrome that can be caused by an adverse drug reaction. In an Italian review of 2137 reactions, NSAIDs were causative agents in 33.6% of patients with drug-induced angioedema.6 In another study, Leeyaphan et al5 reported that 50% of patients with drug-induced angioedema resulted from NSAIDs, commonly with ibuprofen and diclofenac. Although angioedema is due to inhibition of cyclooxygenase 1, overproduction of leukotrienes, and possibly IgE-mediated reactions to single drugs,7 localized unilateral eyelid angioedema with NSAIDs is rare. The exact mechanism of localized eyelid edema is not known.8 We believe that the unilateral eyelid angioedema in our patient was caused by flurbiprofen use because the reaction recurred when the drug was used again.
1. Roszkowski MT, Swift JQ, Hargreaves KM. Effect of NSAID administration on tissue levels of immunoreactive prostaglandin E2, leukotriene B4, and (S)-flurbiprofen following extraction of impacted third molars. Pain. 1997;73:339-345.
2. Asero R. Multiple sensitivity to NSAID. Allergy. 2000;55:893-894.
3. Nettis E, Colanardi MC, Ferrannini A, et al. Update on sensitivity to nonsteroidal anti-inflammatory drugs. Curr Drug Targets Immune Endocr Metabol Disord. 2001;1:233-240.
4. Kulthanan K, Jiamton S, Boochangkool K, et al. Angioedema: clinical and etiological aspects. Clin Dev Immunol. 2007;2007:26438.
5. Leeyaphan C, Kulthanan K, Jongiarearnprasert K, et al. Drug-induced angioedema without urticaria: prevalence and clinical features [published online ahead of print November 17, 2009]. J Eur Acad Dermatol Venereol. 2010;24:685-691.
6. Cutaneous reactions to analgesic-antipyretics and nonsteroidal anti-inflammatory drugs. analysis of reports to the spontaneous reporting system of the Gruppo Italiano Studi Epidemiologici in Dermatologia. Dermatology. 1993;186:164-169.
7. Stevenson OE, Finch TM. Allergic contact dermatitis from rectified camphor oil in Earex ear drops. Contact Dermatitis. 2003;49:51.
8. Tsuruta D, Oshimo T, Sowa J, et al. Unilateral eyelid angioedema with congestion of the right bulbar conjunctiva due to loxoprofen sodium. Cutis. 2011;87:41-43.
To the Editor:
Flurbiprofen, a member of the phenylalkanoic acid derivative group of nonsteroidal anti-inflammatory drugs (NSAIDs), are commonly used to treat fever, inflammation, and pain of arthritis.1 The exact prevalence of allergic reactions to NSAIDs in the general population is not known. Rhinoconjunctivitis, bronchospasm, urticaria, angioedema, and anaphylaxis can occur as an allergic reaction to NSAIDs. Isolated angioedema following NSAID ingestion typically involves the face, particularly the periorbital skin, lips, and mouth.2 These patients may develop urticaria and/or angioedema only after NSAID ingestion, but they do not have underlying chronic urticaria. We report a rare case of isolated unilateral eyelid angioedema with flurbiprofen.
A 39-year-old man presented with the onset of unilateral angioedema of the left upper eyelid that had developed approximately 30 minutes after taking flurbiprofen (100 mg). He reported frequent use of flurbiprofen for headaches. The patient also had a history of taking aspirin, ibuprofen, diclofenac, etodolac, and naproxen sodium as needed for migraines with no prior angioedema. He had no history of chronic urticaria or allergic disease. The patient was treated with oral pheniramine hydrogen maleate and angioedema resolved after 12 hours. Three days later, the patient used flurbiprofen again for a headache. He was readmitted to our clinic with unilateral angioedema of the left upper eyelid (Figure). The symptoms started approximately 30 minutes after taking flurbiprofen. Angioedema resolved within 1 day with oral pheniramine.
Nonsteroidal anti-inflammatory drugs are the most commonly prescribed class of drugs in the world and are the most common cause of all adverse drug reactions.3 Urticaria, angioedema, and anaphylaxis are common adverse reactions to NSAIDs. The prevalence of urticaria and angioedema to NSAIDs has been reported to be 0.1% to 3% worldwide.4
Angioedema is an abrupt localized swelling of the skin and mucous membranes of the face, lips, mouth, throat, larynx, extremities, and genitalia. Angioedema generally develops over minutes to hours and resolves in 24 to 48 hours.5 Angioedema without urticaria is the clinical syndrome that can be caused by an adverse drug reaction. In an Italian review of 2137 reactions, NSAIDs were causative agents in 33.6% of patients with drug-induced angioedema.6 In another study, Leeyaphan et al5 reported that 50% of patients with drug-induced angioedema resulted from NSAIDs, commonly with ibuprofen and diclofenac. Although angioedema is due to inhibition of cyclooxygenase 1, overproduction of leukotrienes, and possibly IgE-mediated reactions to single drugs,7 localized unilateral eyelid angioedema with NSAIDs is rare. The exact mechanism of localized eyelid edema is not known.8 We believe that the unilateral eyelid angioedema in our patient was caused by flurbiprofen use because the reaction recurred when the drug was used again.
To the Editor:
Flurbiprofen, a member of the phenylalkanoic acid derivative group of nonsteroidal anti-inflammatory drugs (NSAIDs), are commonly used to treat fever, inflammation, and pain of arthritis.1 The exact prevalence of allergic reactions to NSAIDs in the general population is not known. Rhinoconjunctivitis, bronchospasm, urticaria, angioedema, and anaphylaxis can occur as an allergic reaction to NSAIDs. Isolated angioedema following NSAID ingestion typically involves the face, particularly the periorbital skin, lips, and mouth.2 These patients may develop urticaria and/or angioedema only after NSAID ingestion, but they do not have underlying chronic urticaria. We report a rare case of isolated unilateral eyelid angioedema with flurbiprofen.
A 39-year-old man presented with the onset of unilateral angioedema of the left upper eyelid that had developed approximately 30 minutes after taking flurbiprofen (100 mg). He reported frequent use of flurbiprofen for headaches. The patient also had a history of taking aspirin, ibuprofen, diclofenac, etodolac, and naproxen sodium as needed for migraines with no prior angioedema. He had no history of chronic urticaria or allergic disease. The patient was treated with oral pheniramine hydrogen maleate and angioedema resolved after 12 hours. Three days later, the patient used flurbiprofen again for a headache. He was readmitted to our clinic with unilateral angioedema of the left upper eyelid (Figure). The symptoms started approximately 30 minutes after taking flurbiprofen. Angioedema resolved within 1 day with oral pheniramine.
Nonsteroidal anti-inflammatory drugs are the most commonly prescribed class of drugs in the world and are the most common cause of all adverse drug reactions.3 Urticaria, angioedema, and anaphylaxis are common adverse reactions to NSAIDs. The prevalence of urticaria and angioedema to NSAIDs has been reported to be 0.1% to 3% worldwide.4
Angioedema is an abrupt localized swelling of the skin and mucous membranes of the face, lips, mouth, throat, larynx, extremities, and genitalia. Angioedema generally develops over minutes to hours and resolves in 24 to 48 hours.5 Angioedema without urticaria is the clinical syndrome that can be caused by an adverse drug reaction. In an Italian review of 2137 reactions, NSAIDs were causative agents in 33.6% of patients with drug-induced angioedema.6 In another study, Leeyaphan et al5 reported that 50% of patients with drug-induced angioedema resulted from NSAIDs, commonly with ibuprofen and diclofenac. Although angioedema is due to inhibition of cyclooxygenase 1, overproduction of leukotrienes, and possibly IgE-mediated reactions to single drugs,7 localized unilateral eyelid angioedema with NSAIDs is rare. The exact mechanism of localized eyelid edema is not known.8 We believe that the unilateral eyelid angioedema in our patient was caused by flurbiprofen use because the reaction recurred when the drug was used again.
1. Roszkowski MT, Swift JQ, Hargreaves KM. Effect of NSAID administration on tissue levels of immunoreactive prostaglandin E2, leukotriene B4, and (S)-flurbiprofen following extraction of impacted third molars. Pain. 1997;73:339-345.
2. Asero R. Multiple sensitivity to NSAID. Allergy. 2000;55:893-894.
3. Nettis E, Colanardi MC, Ferrannini A, et al. Update on sensitivity to nonsteroidal anti-inflammatory drugs. Curr Drug Targets Immune Endocr Metabol Disord. 2001;1:233-240.
4. Kulthanan K, Jiamton S, Boochangkool K, et al. Angioedema: clinical and etiological aspects. Clin Dev Immunol. 2007;2007:26438.
5. Leeyaphan C, Kulthanan K, Jongiarearnprasert K, et al. Drug-induced angioedema without urticaria: prevalence and clinical features [published online ahead of print November 17, 2009]. J Eur Acad Dermatol Venereol. 2010;24:685-691.
6. Cutaneous reactions to analgesic-antipyretics and nonsteroidal anti-inflammatory drugs. analysis of reports to the spontaneous reporting system of the Gruppo Italiano Studi Epidemiologici in Dermatologia. Dermatology. 1993;186:164-169.
7. Stevenson OE, Finch TM. Allergic contact dermatitis from rectified camphor oil in Earex ear drops. Contact Dermatitis. 2003;49:51.
8. Tsuruta D, Oshimo T, Sowa J, et al. Unilateral eyelid angioedema with congestion of the right bulbar conjunctiva due to loxoprofen sodium. Cutis. 2011;87:41-43.
1. Roszkowski MT, Swift JQ, Hargreaves KM. Effect of NSAID administration on tissue levels of immunoreactive prostaglandin E2, leukotriene B4, and (S)-flurbiprofen following extraction of impacted third molars. Pain. 1997;73:339-345.
2. Asero R. Multiple sensitivity to NSAID. Allergy. 2000;55:893-894.
3. Nettis E, Colanardi MC, Ferrannini A, et al. Update on sensitivity to nonsteroidal anti-inflammatory drugs. Curr Drug Targets Immune Endocr Metabol Disord. 2001;1:233-240.
4. Kulthanan K, Jiamton S, Boochangkool K, et al. Angioedema: clinical and etiological aspects. Clin Dev Immunol. 2007;2007:26438.
5. Leeyaphan C, Kulthanan K, Jongiarearnprasert K, et al. Drug-induced angioedema without urticaria: prevalence and clinical features [published online ahead of print November 17, 2009]. J Eur Acad Dermatol Venereol. 2010;24:685-691.
6. Cutaneous reactions to analgesic-antipyretics and nonsteroidal anti-inflammatory drugs. analysis of reports to the spontaneous reporting system of the Gruppo Italiano Studi Epidemiologici in Dermatologia. Dermatology. 1993;186:164-169.
7. Stevenson OE, Finch TM. Allergic contact dermatitis from rectified camphor oil in Earex ear drops. Contact Dermatitis. 2003;49:51.
8. Tsuruta D, Oshimo T, Sowa J, et al. Unilateral eyelid angioedema with congestion of the right bulbar conjunctiva due to loxoprofen sodium. Cutis. 2011;87:41-43.
Anal high-grade squamous intraepithelial lesions cleared in 42% of gay men
MELBOURNE – High-grade squamous intraepithelial lesions are prevalent among HIV-positive gay men and a significant number of these anal lesions resolve spontaneously, according to data from a longitudinal observational cohort study.
The Study of the Prevention of Anal Cancer (SPANC) is a 3-year prospective study of the natural history of anal HPV infection, which has so far enrolled 350 homosexual men over 35 years old. Dr. Andrew Grulich from the Kirby Institute, University of New South Wales, Sydney, presented trial data at the 20th International AIDS Conference that showed a 42% clearance rate for high-grade squamous intraepithelial lesions (HSIL).
Anal lesions that result from human papillomavirus infection are difficult to treat, and there is no evidence for the effectiveness of treatment, Dr. Grulich said. Further, anal lesions are far less likely than cervical lesions to progress to cancer. However, questions remain about which men are more likely to have persistent high-grade disease and therefore are at a higher risk of progression to cancer.
Interim data from the SPANC study found a 46% prevalence of HSIL among HIV-positive gay men and a 34% prevalence among HIV-negative gay men. The higher rate among HIV-positive individuals was largely driven by a higher prevalence of more advanced anal intraepithelial neoplasia.
Men with persistent infections due to human papillomavirus (HPV) 16 – the subtype most commonly associated with anal cancer – were much less likely to clear the high-grade lesions (hazard ratio = 0.22, 95% confidence interval, 0.11-0.46), as were men with multiple subtypes of HPV.
"What we’ve been able to show is that high-grade disease is very dynamic, with one in six [gay] men getting it and of those who get it about 40% clearing it each year," Dr. Grulich told the conference.
"Not all high-grade disease requires treatment," he said. "These data suggest that treatment can be targeted at those with persistent high-grade disease because much high-grade disease diagnosed on a single occasion will simply go away."
Treatment practices for HSIL vary with some choosing a ‘watch and wait’ approach and others choosing ablative treatment. Dr. Grulich suggested treatment based on "red flags" that suggest a higher risk of progression to cancer.
"If [the patient] is HPV 16–positive, that’s a definite red flag because 90% of anal cancer is caused by that one subtype," Dr. Grulich said. Also, high-grade disease that doesn’t clear is another red flag.
"I think it’s perfectly reasonable, given the state of the science, if you’re doing high-resolution anoscopy and you diagnose (HSIL), to explain to the patient that it’s highly likely to go away but it may not, and therefore get the patient back in about a year," he said.
The study is funded by the National Health and Medical Research Council of Australia, and the Cancer Council NSW. Some authors declared financial ties to pharmaceutical companies including a manufacturer of HPV vaccines.
MELBOURNE – High-grade squamous intraepithelial lesions are prevalent among HIV-positive gay men and a significant number of these anal lesions resolve spontaneously, according to data from a longitudinal observational cohort study.
The Study of the Prevention of Anal Cancer (SPANC) is a 3-year prospective study of the natural history of anal HPV infection, which has so far enrolled 350 homosexual men over 35 years old. Dr. Andrew Grulich from the Kirby Institute, University of New South Wales, Sydney, presented trial data at the 20th International AIDS Conference that showed a 42% clearance rate for high-grade squamous intraepithelial lesions (HSIL).
Anal lesions that result from human papillomavirus infection are difficult to treat, and there is no evidence for the effectiveness of treatment, Dr. Grulich said. Further, anal lesions are far less likely than cervical lesions to progress to cancer. However, questions remain about which men are more likely to have persistent high-grade disease and therefore are at a higher risk of progression to cancer.
Interim data from the SPANC study found a 46% prevalence of HSIL among HIV-positive gay men and a 34% prevalence among HIV-negative gay men. The higher rate among HIV-positive individuals was largely driven by a higher prevalence of more advanced anal intraepithelial neoplasia.
Men with persistent infections due to human papillomavirus (HPV) 16 – the subtype most commonly associated with anal cancer – were much less likely to clear the high-grade lesions (hazard ratio = 0.22, 95% confidence interval, 0.11-0.46), as were men with multiple subtypes of HPV.
"What we’ve been able to show is that high-grade disease is very dynamic, with one in six [gay] men getting it and of those who get it about 40% clearing it each year," Dr. Grulich told the conference.
"Not all high-grade disease requires treatment," he said. "These data suggest that treatment can be targeted at those with persistent high-grade disease because much high-grade disease diagnosed on a single occasion will simply go away."
Treatment practices for HSIL vary with some choosing a ‘watch and wait’ approach and others choosing ablative treatment. Dr. Grulich suggested treatment based on "red flags" that suggest a higher risk of progression to cancer.
"If [the patient] is HPV 16–positive, that’s a definite red flag because 90% of anal cancer is caused by that one subtype," Dr. Grulich said. Also, high-grade disease that doesn’t clear is another red flag.
"I think it’s perfectly reasonable, given the state of the science, if you’re doing high-resolution anoscopy and you diagnose (HSIL), to explain to the patient that it’s highly likely to go away but it may not, and therefore get the patient back in about a year," he said.
The study is funded by the National Health and Medical Research Council of Australia, and the Cancer Council NSW. Some authors declared financial ties to pharmaceutical companies including a manufacturer of HPV vaccines.
MELBOURNE – High-grade squamous intraepithelial lesions are prevalent among HIV-positive gay men and a significant number of these anal lesions resolve spontaneously, according to data from a longitudinal observational cohort study.
The Study of the Prevention of Anal Cancer (SPANC) is a 3-year prospective study of the natural history of anal HPV infection, which has so far enrolled 350 homosexual men over 35 years old. Dr. Andrew Grulich from the Kirby Institute, University of New South Wales, Sydney, presented trial data at the 20th International AIDS Conference that showed a 42% clearance rate for high-grade squamous intraepithelial lesions (HSIL).
Anal lesions that result from human papillomavirus infection are difficult to treat, and there is no evidence for the effectiveness of treatment, Dr. Grulich said. Further, anal lesions are far less likely than cervical lesions to progress to cancer. However, questions remain about which men are more likely to have persistent high-grade disease and therefore are at a higher risk of progression to cancer.
Interim data from the SPANC study found a 46% prevalence of HSIL among HIV-positive gay men and a 34% prevalence among HIV-negative gay men. The higher rate among HIV-positive individuals was largely driven by a higher prevalence of more advanced anal intraepithelial neoplasia.
Men with persistent infections due to human papillomavirus (HPV) 16 – the subtype most commonly associated with anal cancer – were much less likely to clear the high-grade lesions (hazard ratio = 0.22, 95% confidence interval, 0.11-0.46), as were men with multiple subtypes of HPV.
"What we’ve been able to show is that high-grade disease is very dynamic, with one in six [gay] men getting it and of those who get it about 40% clearing it each year," Dr. Grulich told the conference.
"Not all high-grade disease requires treatment," he said. "These data suggest that treatment can be targeted at those with persistent high-grade disease because much high-grade disease diagnosed on a single occasion will simply go away."
Treatment practices for HSIL vary with some choosing a ‘watch and wait’ approach and others choosing ablative treatment. Dr. Grulich suggested treatment based on "red flags" that suggest a higher risk of progression to cancer.
"If [the patient] is HPV 16–positive, that’s a definite red flag because 90% of anal cancer is caused by that one subtype," Dr. Grulich said. Also, high-grade disease that doesn’t clear is another red flag.
"I think it’s perfectly reasonable, given the state of the science, if you’re doing high-resolution anoscopy and you diagnose (HSIL), to explain to the patient that it’s highly likely to go away but it may not, and therefore get the patient back in about a year," he said.
The study is funded by the National Health and Medical Research Council of Australia, and the Cancer Council NSW. Some authors declared financial ties to pharmaceutical companies including a manufacturer of HPV vaccines.
AT AIDS 2014
Key clinical point: Watchful waiting may be an option for gay men with anal high-grade squamous intraepithelial lesions if the lesion is not HPV 16 positive and has not been present for more than a year.
Major finding: Over 40% of anal high-grade squamous intraepithelial lesions clear spontaneously without treatment, with similar clearance rates for HIV-positive and HIV-negative gay men.
Data source: Interim data on 350 gay men over 35 years old enrolled in SPANC, a 3-year prospective study of the natural history of anal HPV infection.
Disclosures: The study is funded by the National Health and Medical Research Council of Australia and the Cancer Council NSW. Some authors declared financial ties to pharmaceutical companies including a manufacturer of HPV vaccines.
UV Radiation Transmittance: Regular Clothing Versus Sun-Protective Clothing
Dermatologists frequently encounter patients who inquire about the need to buy special clothing and hats that claim to block UV light rather than using their regular clothing and hats. A patient may argue that he/she has never gotten sunburned through his/her favorite T-shirt while fishing, so why does he/she need to buy special clothing? The answer to this question is not straightforward. The dermatologist could easily say yes and advise patients to buy special sun-protective clothing, which could be especially tempting if a practitioner actually sells these items in the office. However, when considering evidence-based medicine, one needs to look at the data to appropriately answer the question.
Although it is still evolving, a standard has been set for UV protection factor (UPF) in the United States as well as other countries.1 Clothing with the maximum UPF rating of 50 blocks 98% of UVA/UVB radiation. Although there are data published in the literature regarding sun-protective clothing, there are scant data in the clinical dermatologic literature.2-5 To give patients an educated answer to this question, we measured and compared UVA/UVB radiation transmittance through regular (ie, non–UPF rated) clothing versus sun-protective clothing with a UPF rating.
Materials and Methods
A digital handheld UVA/UVB meter with an absorption spectrum of 280 to 400 nm was used to measure UV energy transmitted through sample clothing articles. The meter measured UVA/UVB light with a maximum reading of 40 mW/cm2. Clothing articles were selected of varied material/color and intended use.
Regular clothing articles included a straw golf hat (Figure 1A), an off-white and blue baseball hat (70% wool)(Figure 1B), a black baseball hat (100% wool)(Figure 1C), a white athletic tank shirt (100% cotton), a white T-shirt/undershirt (100% cotton), a thin-weave blue T-shirt (100% cotton), and a conventional-weave blue T-shirt (100% cotton)(Figure 1D). The regular clothing items, with the exception of the hats, had been laundered in conventional (ie, non–UV blocking) laundry detergent and no chemicals were applied to enhance UVA/UVB blocking properties. The exact number of times the items were laundered was unknown.

|

|

|

|
| Figure 1. Regular clothing articles included a straw golf hat (A), an off-white and blue baseball hat (B), a black baseball hat (C), and white and blue T-shirts (D). |
Sun-protective clothing articles included a polyester floral splash bucket hat and a polyester ruffled swim romper (Figure 2), both with a UPF rating of 50+. These items were purchased from a manufacturer who regularly promotes sun-protective clothing to both dermatologists and the general public. The company “guarantees” a UPF rating of 50 and advertises that these clothing articles block 98% of harmful UV rays. These items were not laundered prior to the study, and no chemicals were applied to enhance UVA/UVB blocking properties.
The UVA/UVB meter was calibrated on a clear cloudless July day in Frankfort, Illinois. An initial reading was taken without any obstruction to the sunlight. The regular and sun-protective clothing articles then were placed over the meter to measure the amount of UVA/UVB transmitted through each item. Measurements were taken for each article of clothing after the meter was covered by the respective material for 10 seconds. Care was taken to cover the meter with only 1 layer of material for each article, which was intended to mimic the degree of UVA/UVB blocking and transmittance during normal wear.
Results
The full results from the study are outlined in the Table. The unobstructed sunlight exposure exceeded the maximum measure of 40 mW/cm2, indicating there was a sufficient amount of sunlight to conduct testing.
The data show that both regular and sun-protective clothing blocked UVA/UVB rays in the 280- to 400-nm range. The Table outlines the level of UVA/UVB transmittance for each article of clothing; a lower number indicates less UVA/UVB transmittance occurred and more radiation was blocked.
Several of the regular clothing blocked more UV radiation than the sun-protective clothing; specifically, the data indicate that the baseball hats or the straw golf hat provided better protection than the sun-protective bucket hat. The black baseball hat provided the best UV protection. However, the straw golf hat provided adequate protection and better coverage, making it the best recommendation for patients.
Comment
Several of the regular items included in the study allowed less UVA/UVB transmission than the sun-protective clothing. Although our small study tested a limited number and type of articles, we assert that similar regular clothing would have similar transmittance.
There are various factors that affect UVA/UVB transmittance. Fabric construction, weight, thickness, composition, and color will affect the degree of UVA/UVB transmittance.1 In our study, the thickness, weave, and color of the fabric of the regular hats may have contributed to the superior results compared with the sun-protective hat. It could be postulated that cotton is inherently a superior fabric to the polyester sun-protective clothing fabric. With regard to the regular T-shirts, thickness, weave, and color also may have played a role in blocking UVA/UVB transmittance.
Patients may be assured of a sufficient amount of UVA/UVB blocking with sun-protective clothing. However, our study indicated that the regular clothing articles we tested provided similar, if not better, protection against UV radiation compared with the sun-protective clothing articles.
Conclusion
Based on the data, we would advise patients that they do not need to buy special sun-protective clothing that claims to block UV radiation, as regular clothing will provide equivalent protection against UVA/UVB radiation. However, these findings do not suggest that the claims for sun-protective clothing are inaccurate. Nevertheless, similar UVA/UVB blocking may be achieved with clothing already owned by patients.
1. Gies P. Photoprotection by clothing. Photodermatol Photoimmunol Photomed. 2007;23:264-274.
2. Wilson CA, Bevin NK, Laing RM, et al. Solar protection—effect of selected fabric and use characteristics on ultraviolet transmission. Textile Research Journal. 2008;78:95-104.
3. Ghazi S, Couteau C, Coiffard LJ. How to guarantee sun protection for a young sportsperson. J Dtsch Dermatol Ges. 2011;9:470-474.
4. Ghazi S, Couteau C, Coiffard LJ. What level of protection can be obtained using sun protective clothing? determining effectiveness using an in vitro method. Int J Pharm. 2010;397:144-146.
5. Morison WL. Photoprotection by clothing. Dermatol Ther. 2003;16:16-22.
Dermatologists frequently encounter patients who inquire about the need to buy special clothing and hats that claim to block UV light rather than using their regular clothing and hats. A patient may argue that he/she has never gotten sunburned through his/her favorite T-shirt while fishing, so why does he/she need to buy special clothing? The answer to this question is not straightforward. The dermatologist could easily say yes and advise patients to buy special sun-protective clothing, which could be especially tempting if a practitioner actually sells these items in the office. However, when considering evidence-based medicine, one needs to look at the data to appropriately answer the question.
Although it is still evolving, a standard has been set for UV protection factor (UPF) in the United States as well as other countries.1 Clothing with the maximum UPF rating of 50 blocks 98% of UVA/UVB radiation. Although there are data published in the literature regarding sun-protective clothing, there are scant data in the clinical dermatologic literature.2-5 To give patients an educated answer to this question, we measured and compared UVA/UVB radiation transmittance through regular (ie, non–UPF rated) clothing versus sun-protective clothing with a UPF rating.
Materials and Methods
A digital handheld UVA/UVB meter with an absorption spectrum of 280 to 400 nm was used to measure UV energy transmitted through sample clothing articles. The meter measured UVA/UVB light with a maximum reading of 40 mW/cm2. Clothing articles were selected of varied material/color and intended use.
Regular clothing articles included a straw golf hat (Figure 1A), an off-white and blue baseball hat (70% wool)(Figure 1B), a black baseball hat (100% wool)(Figure 1C), a white athletic tank shirt (100% cotton), a white T-shirt/undershirt (100% cotton), a thin-weave blue T-shirt (100% cotton), and a conventional-weave blue T-shirt (100% cotton)(Figure 1D). The regular clothing items, with the exception of the hats, had been laundered in conventional (ie, non–UV blocking) laundry detergent and no chemicals were applied to enhance UVA/UVB blocking properties. The exact number of times the items were laundered was unknown.

|

|

|

|
| Figure 1. Regular clothing articles included a straw golf hat (A), an off-white and blue baseball hat (B), a black baseball hat (C), and white and blue T-shirts (D). |
Sun-protective clothing articles included a polyester floral splash bucket hat and a polyester ruffled swim romper (Figure 2), both with a UPF rating of 50+. These items were purchased from a manufacturer who regularly promotes sun-protective clothing to both dermatologists and the general public. The company “guarantees” a UPF rating of 50 and advertises that these clothing articles block 98% of harmful UV rays. These items were not laundered prior to the study, and no chemicals were applied to enhance UVA/UVB blocking properties.
The UVA/UVB meter was calibrated on a clear cloudless July day in Frankfort, Illinois. An initial reading was taken without any obstruction to the sunlight. The regular and sun-protective clothing articles then were placed over the meter to measure the amount of UVA/UVB transmitted through each item. Measurements were taken for each article of clothing after the meter was covered by the respective material for 10 seconds. Care was taken to cover the meter with only 1 layer of material for each article, which was intended to mimic the degree of UVA/UVB blocking and transmittance during normal wear.
Results
The full results from the study are outlined in the Table. The unobstructed sunlight exposure exceeded the maximum measure of 40 mW/cm2, indicating there was a sufficient amount of sunlight to conduct testing.
The data show that both regular and sun-protective clothing blocked UVA/UVB rays in the 280- to 400-nm range. The Table outlines the level of UVA/UVB transmittance for each article of clothing; a lower number indicates less UVA/UVB transmittance occurred and more radiation was blocked.
Several of the regular clothing blocked more UV radiation than the sun-protective clothing; specifically, the data indicate that the baseball hats or the straw golf hat provided better protection than the sun-protective bucket hat. The black baseball hat provided the best UV protection. However, the straw golf hat provided adequate protection and better coverage, making it the best recommendation for patients.
Comment
Several of the regular items included in the study allowed less UVA/UVB transmission than the sun-protective clothing. Although our small study tested a limited number and type of articles, we assert that similar regular clothing would have similar transmittance.
There are various factors that affect UVA/UVB transmittance. Fabric construction, weight, thickness, composition, and color will affect the degree of UVA/UVB transmittance.1 In our study, the thickness, weave, and color of the fabric of the regular hats may have contributed to the superior results compared with the sun-protective hat. It could be postulated that cotton is inherently a superior fabric to the polyester sun-protective clothing fabric. With regard to the regular T-shirts, thickness, weave, and color also may have played a role in blocking UVA/UVB transmittance.
Patients may be assured of a sufficient amount of UVA/UVB blocking with sun-protective clothing. However, our study indicated that the regular clothing articles we tested provided similar, if not better, protection against UV radiation compared with the sun-protective clothing articles.
Conclusion
Based on the data, we would advise patients that they do not need to buy special sun-protective clothing that claims to block UV radiation, as regular clothing will provide equivalent protection against UVA/UVB radiation. However, these findings do not suggest that the claims for sun-protective clothing are inaccurate. Nevertheless, similar UVA/UVB blocking may be achieved with clothing already owned by patients.
Dermatologists frequently encounter patients who inquire about the need to buy special clothing and hats that claim to block UV light rather than using their regular clothing and hats. A patient may argue that he/she has never gotten sunburned through his/her favorite T-shirt while fishing, so why does he/she need to buy special clothing? The answer to this question is not straightforward. The dermatologist could easily say yes and advise patients to buy special sun-protective clothing, which could be especially tempting if a practitioner actually sells these items in the office. However, when considering evidence-based medicine, one needs to look at the data to appropriately answer the question.
Although it is still evolving, a standard has been set for UV protection factor (UPF) in the United States as well as other countries.1 Clothing with the maximum UPF rating of 50 blocks 98% of UVA/UVB radiation. Although there are data published in the literature regarding sun-protective clothing, there are scant data in the clinical dermatologic literature.2-5 To give patients an educated answer to this question, we measured and compared UVA/UVB radiation transmittance through regular (ie, non–UPF rated) clothing versus sun-protective clothing with a UPF rating.
Materials and Methods
A digital handheld UVA/UVB meter with an absorption spectrum of 280 to 400 nm was used to measure UV energy transmitted through sample clothing articles. The meter measured UVA/UVB light with a maximum reading of 40 mW/cm2. Clothing articles were selected of varied material/color and intended use.
Regular clothing articles included a straw golf hat (Figure 1A), an off-white and blue baseball hat (70% wool)(Figure 1B), a black baseball hat (100% wool)(Figure 1C), a white athletic tank shirt (100% cotton), a white T-shirt/undershirt (100% cotton), a thin-weave blue T-shirt (100% cotton), and a conventional-weave blue T-shirt (100% cotton)(Figure 1D). The regular clothing items, with the exception of the hats, had been laundered in conventional (ie, non–UV blocking) laundry detergent and no chemicals were applied to enhance UVA/UVB blocking properties. The exact number of times the items were laundered was unknown.

|

|

|

|
| Figure 1. Regular clothing articles included a straw golf hat (A), an off-white and blue baseball hat (B), a black baseball hat (C), and white and blue T-shirts (D). |
Sun-protective clothing articles included a polyester floral splash bucket hat and a polyester ruffled swim romper (Figure 2), both with a UPF rating of 50+. These items were purchased from a manufacturer who regularly promotes sun-protective clothing to both dermatologists and the general public. The company “guarantees” a UPF rating of 50 and advertises that these clothing articles block 98% of harmful UV rays. These items were not laundered prior to the study, and no chemicals were applied to enhance UVA/UVB blocking properties.
The UVA/UVB meter was calibrated on a clear cloudless July day in Frankfort, Illinois. An initial reading was taken without any obstruction to the sunlight. The regular and sun-protective clothing articles then were placed over the meter to measure the amount of UVA/UVB transmitted through each item. Measurements were taken for each article of clothing after the meter was covered by the respective material for 10 seconds. Care was taken to cover the meter with only 1 layer of material for each article, which was intended to mimic the degree of UVA/UVB blocking and transmittance during normal wear.
Results
The full results from the study are outlined in the Table. The unobstructed sunlight exposure exceeded the maximum measure of 40 mW/cm2, indicating there was a sufficient amount of sunlight to conduct testing.
The data show that both regular and sun-protective clothing blocked UVA/UVB rays in the 280- to 400-nm range. The Table outlines the level of UVA/UVB transmittance for each article of clothing; a lower number indicates less UVA/UVB transmittance occurred and more radiation was blocked.
Several of the regular clothing blocked more UV radiation than the sun-protective clothing; specifically, the data indicate that the baseball hats or the straw golf hat provided better protection than the sun-protective bucket hat. The black baseball hat provided the best UV protection. However, the straw golf hat provided adequate protection and better coverage, making it the best recommendation for patients.
Comment
Several of the regular items included in the study allowed less UVA/UVB transmission than the sun-protective clothing. Although our small study tested a limited number and type of articles, we assert that similar regular clothing would have similar transmittance.
There are various factors that affect UVA/UVB transmittance. Fabric construction, weight, thickness, composition, and color will affect the degree of UVA/UVB transmittance.1 In our study, the thickness, weave, and color of the fabric of the regular hats may have contributed to the superior results compared with the sun-protective hat. It could be postulated that cotton is inherently a superior fabric to the polyester sun-protective clothing fabric. With regard to the regular T-shirts, thickness, weave, and color also may have played a role in blocking UVA/UVB transmittance.
Patients may be assured of a sufficient amount of UVA/UVB blocking with sun-protective clothing. However, our study indicated that the regular clothing articles we tested provided similar, if not better, protection against UV radiation compared with the sun-protective clothing articles.
Conclusion
Based on the data, we would advise patients that they do not need to buy special sun-protective clothing that claims to block UV radiation, as regular clothing will provide equivalent protection against UVA/UVB radiation. However, these findings do not suggest that the claims for sun-protective clothing are inaccurate. Nevertheless, similar UVA/UVB blocking may be achieved with clothing already owned by patients.
1. Gies P. Photoprotection by clothing. Photodermatol Photoimmunol Photomed. 2007;23:264-274.
2. Wilson CA, Bevin NK, Laing RM, et al. Solar protection—effect of selected fabric and use characteristics on ultraviolet transmission. Textile Research Journal. 2008;78:95-104.
3. Ghazi S, Couteau C, Coiffard LJ. How to guarantee sun protection for a young sportsperson. J Dtsch Dermatol Ges. 2011;9:470-474.
4. Ghazi S, Couteau C, Coiffard LJ. What level of protection can be obtained using sun protective clothing? determining effectiveness using an in vitro method. Int J Pharm. 2010;397:144-146.
5. Morison WL. Photoprotection by clothing. Dermatol Ther. 2003;16:16-22.
1. Gies P. Photoprotection by clothing. Photodermatol Photoimmunol Photomed. 2007;23:264-274.
2. Wilson CA, Bevin NK, Laing RM, et al. Solar protection—effect of selected fabric and use characteristics on ultraviolet transmission. Textile Research Journal. 2008;78:95-104.
3. Ghazi S, Couteau C, Coiffard LJ. How to guarantee sun protection for a young sportsperson. J Dtsch Dermatol Ges. 2011;9:470-474.
4. Ghazi S, Couteau C, Coiffard LJ. What level of protection can be obtained using sun protective clothing? determining effectiveness using an in vitro method. Int J Pharm. 2010;397:144-146.
5. Morison WL. Photoprotection by clothing. Dermatol Ther. 2003;16:16-22.
- Dermatologists routinely advise patients that clothing is a method of UVA/UVB protection.
- Regular clothing items provide similar, if not superior, UVA/UVB protection compared to sun-protective clothing.
- Physicians may confidently inform patients that regular clothing items will provide adequate UVA/UVB protection.
VIDEO: German screening initiative catches skin cancer sooner
EDINBURGH – Is population screening for skin cancer worthwhile?
Yes, Dr. Eckhard Breitbart said in a debate at the 15th World Congress on Cancers of the Skin. "Screening is not a diagnostic procedure," he noted. But the potential benefits of screening, including a significant reduction in medical costs, outweigh the potential harms related to false negative or false positive findings, added Dr. Breitbart, a dermatologist in Hamburg, Germany.
In an interview at the meeting, Dr. Breitbart reviewed the findings from his study of the impact of a population-based skin cancer screening program in Germany, and he discussed what research is needed to support such screening elsewhere.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EDINBURGH – Is population screening for skin cancer worthwhile?
Yes, Dr. Eckhard Breitbart said in a debate at the 15th World Congress on Cancers of the Skin. "Screening is not a diagnostic procedure," he noted. But the potential benefits of screening, including a significant reduction in medical costs, outweigh the potential harms related to false negative or false positive findings, added Dr. Breitbart, a dermatologist in Hamburg, Germany.
In an interview at the meeting, Dr. Breitbart reviewed the findings from his study of the impact of a population-based skin cancer screening program in Germany, and he discussed what research is needed to support such screening elsewhere.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EDINBURGH – Is population screening for skin cancer worthwhile?
Yes, Dr. Eckhard Breitbart said in a debate at the 15th World Congress on Cancers of the Skin. "Screening is not a diagnostic procedure," he noted. But the potential benefits of screening, including a significant reduction in medical costs, outweigh the potential harms related to false negative or false positive findings, added Dr. Breitbart, a dermatologist in Hamburg, Germany.
In an interview at the meeting, Dr. Breitbart reviewed the findings from his study of the impact of a population-based skin cancer screening program in Germany, and he discussed what research is needed to support such screening elsewhere.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
FROM THE 2014 WORLD CONGRESS ON CANCERS OF THE SKINS
Don’t overlook anus, genitalia during total body skin exam
CHICAGO – The anus and genitalia are often overlooked during total body skin examinations, leaving mucosal diseases to go unchecked, especially in women, according to Dr. Bethanee Schlosser.
She acknowledged that there is no literature to quantify the issue but said that her experience suggests mucocutaneous exams may be getting short shrift.
When Dr. Schlosser queried some 300 dermatologists assembled earlier this year, almost all said that they examine the oral cavity during total body skin exams; three-fourths responded that they routinely examine male patients’ genitalia. When asked whether they do the same for their female patients, less than 20 hands went up in the crowd.
"I think there are a couple of reasons for it," Dr. Schlosser of Northwestern University, Chicago, said at the American Academy of Dermatology summer meeting.
First, dermatologists don’t often look at the genitalia, so they may not know what the normal variations are.
Second, many patients don’t expect a dermatologist to examine genitalia. "Patients may be like, ‘You want to look where? I just have a mole on my chest.’ So it’s a matter of patient education," she said.
Third is the added time involved, and finally, some dermatologists are hesitant because they may not be comfortable managing mucosal disease should they find it.
Most dermatologists assume that gynecologists are evaluating their patients’ vulvar skin, but some gynecologists view the vulva more as "the doorway to the cervix. They may simply walk through it, not looking at what is around them, and to that effect, I don’t think it’s necessarily their fault, but their training," Dr. Schlosser said. "While vulvar disease is on the ob.gyn. board exam, the senior ob.gyn. residents rotate through our clinic with me and they routinely say they don’t get that education anywhere else."
Dr. Schlosser offered pearls for managing a number of mucosal diseases, including vulvar lichen sclerosis (VLS).
VLS affects about 1 in 600 women and can carry significant morbidity, including complete obliteration of the clitoral hood and labia minora, narrowing of the vaginal introitus, sexual dysfunction, and potential urinary obstruction, she said.
The risk of developing squamous cell carcinoma (SCC) in patients with VLS is 300-fold higher than in the general patient population. The specific risk factors for vulvar SCC are not fully elucidated in VLS, but include localized hyperkeratosis and age over 75 years.
"It’s important to realize that these older patients ... often don’t see their gynecologists because they’re told the pelvic examination is not indicated anymore," Dr. Schlosser said. "It really behooves us as dermatologists to be doing a genital exam as part of our total body skin exam."
Researchers think, but don’t have the evidence to suggest, that treating VLS changes the risk of SCC, "which is one of the hardest things to discuss with our patients," she added.
Suspicion of SCC should be raised if the patient has hyperkeratotic lesions; ulceration, even pinpoint in size, that doesn’t improve with standard therapy; erythematous, indurated plaques; or if the patient reports a change in symptom quality – previously itchy and now painful, for example – or a change in symptom distribution, such as previously all over and now localized to one spot.
"That can really herald that something bad has occurred," Dr. Schlosser cautioned.
Superpotent topical corticosteroids such as clobetasol propionate are first-line therapy for VLS, with no rationale to support the old-school treatment of topical testosterone. Maintenance therapy is common, as up to 85% of women will relapse.
Dr. Schlosser also advised physicians to educate women on how much medication to use, to send them home with a diagram of the affected area, and to use a hand mirror during the exam.
"Patients don’t want to look, but I tell them you’re not going to get better if you don’t know where to put your medication," she said.
Dr. Schlosser reported no relevant conflicts of interest.
CHICAGO – The anus and genitalia are often overlooked during total body skin examinations, leaving mucosal diseases to go unchecked, especially in women, according to Dr. Bethanee Schlosser.
She acknowledged that there is no literature to quantify the issue but said that her experience suggests mucocutaneous exams may be getting short shrift.
When Dr. Schlosser queried some 300 dermatologists assembled earlier this year, almost all said that they examine the oral cavity during total body skin exams; three-fourths responded that they routinely examine male patients’ genitalia. When asked whether they do the same for their female patients, less than 20 hands went up in the crowd.
"I think there are a couple of reasons for it," Dr. Schlosser of Northwestern University, Chicago, said at the American Academy of Dermatology summer meeting.
First, dermatologists don’t often look at the genitalia, so they may not know what the normal variations are.
Second, many patients don’t expect a dermatologist to examine genitalia. "Patients may be like, ‘You want to look where? I just have a mole on my chest.’ So it’s a matter of patient education," she said.
Third is the added time involved, and finally, some dermatologists are hesitant because they may not be comfortable managing mucosal disease should they find it.
Most dermatologists assume that gynecologists are evaluating their patients’ vulvar skin, but some gynecologists view the vulva more as "the doorway to the cervix. They may simply walk through it, not looking at what is around them, and to that effect, I don’t think it’s necessarily their fault, but their training," Dr. Schlosser said. "While vulvar disease is on the ob.gyn. board exam, the senior ob.gyn. residents rotate through our clinic with me and they routinely say they don’t get that education anywhere else."
Dr. Schlosser offered pearls for managing a number of mucosal diseases, including vulvar lichen sclerosis (VLS).
VLS affects about 1 in 600 women and can carry significant morbidity, including complete obliteration of the clitoral hood and labia minora, narrowing of the vaginal introitus, sexual dysfunction, and potential urinary obstruction, she said.
The risk of developing squamous cell carcinoma (SCC) in patients with VLS is 300-fold higher than in the general patient population. The specific risk factors for vulvar SCC are not fully elucidated in VLS, but include localized hyperkeratosis and age over 75 years.
"It’s important to realize that these older patients ... often don’t see their gynecologists because they’re told the pelvic examination is not indicated anymore," Dr. Schlosser said. "It really behooves us as dermatologists to be doing a genital exam as part of our total body skin exam."
Researchers think, but don’t have the evidence to suggest, that treating VLS changes the risk of SCC, "which is one of the hardest things to discuss with our patients," she added.
Suspicion of SCC should be raised if the patient has hyperkeratotic lesions; ulceration, even pinpoint in size, that doesn’t improve with standard therapy; erythematous, indurated plaques; or if the patient reports a change in symptom quality – previously itchy and now painful, for example – or a change in symptom distribution, such as previously all over and now localized to one spot.
"That can really herald that something bad has occurred," Dr. Schlosser cautioned.
Superpotent topical corticosteroids such as clobetasol propionate are first-line therapy for VLS, with no rationale to support the old-school treatment of topical testosterone. Maintenance therapy is common, as up to 85% of women will relapse.
Dr. Schlosser also advised physicians to educate women on how much medication to use, to send them home with a diagram of the affected area, and to use a hand mirror during the exam.
"Patients don’t want to look, but I tell them you’re not going to get better if you don’t know where to put your medication," she said.
Dr. Schlosser reported no relevant conflicts of interest.
CHICAGO – The anus and genitalia are often overlooked during total body skin examinations, leaving mucosal diseases to go unchecked, especially in women, according to Dr. Bethanee Schlosser.
She acknowledged that there is no literature to quantify the issue but said that her experience suggests mucocutaneous exams may be getting short shrift.
When Dr. Schlosser queried some 300 dermatologists assembled earlier this year, almost all said that they examine the oral cavity during total body skin exams; three-fourths responded that they routinely examine male patients’ genitalia. When asked whether they do the same for their female patients, less than 20 hands went up in the crowd.
"I think there are a couple of reasons for it," Dr. Schlosser of Northwestern University, Chicago, said at the American Academy of Dermatology summer meeting.
First, dermatologists don’t often look at the genitalia, so they may not know what the normal variations are.
Second, many patients don’t expect a dermatologist to examine genitalia. "Patients may be like, ‘You want to look where? I just have a mole on my chest.’ So it’s a matter of patient education," she said.
Third is the added time involved, and finally, some dermatologists are hesitant because they may not be comfortable managing mucosal disease should they find it.
Most dermatologists assume that gynecologists are evaluating their patients’ vulvar skin, but some gynecologists view the vulva more as "the doorway to the cervix. They may simply walk through it, not looking at what is around them, and to that effect, I don’t think it’s necessarily their fault, but their training," Dr. Schlosser said. "While vulvar disease is on the ob.gyn. board exam, the senior ob.gyn. residents rotate through our clinic with me and they routinely say they don’t get that education anywhere else."
Dr. Schlosser offered pearls for managing a number of mucosal diseases, including vulvar lichen sclerosis (VLS).
VLS affects about 1 in 600 women and can carry significant morbidity, including complete obliteration of the clitoral hood and labia minora, narrowing of the vaginal introitus, sexual dysfunction, and potential urinary obstruction, she said.
The risk of developing squamous cell carcinoma (SCC) in patients with VLS is 300-fold higher than in the general patient population. The specific risk factors for vulvar SCC are not fully elucidated in VLS, but include localized hyperkeratosis and age over 75 years.
"It’s important to realize that these older patients ... often don’t see their gynecologists because they’re told the pelvic examination is not indicated anymore," Dr. Schlosser said. "It really behooves us as dermatologists to be doing a genital exam as part of our total body skin exam."
Researchers think, but don’t have the evidence to suggest, that treating VLS changes the risk of SCC, "which is one of the hardest things to discuss with our patients," she added.
Suspicion of SCC should be raised if the patient has hyperkeratotic lesions; ulceration, even pinpoint in size, that doesn’t improve with standard therapy; erythematous, indurated plaques; or if the patient reports a change in symptom quality – previously itchy and now painful, for example – or a change in symptom distribution, such as previously all over and now localized to one spot.
"That can really herald that something bad has occurred," Dr. Schlosser cautioned.
Superpotent topical corticosteroids such as clobetasol propionate are first-line therapy for VLS, with no rationale to support the old-school treatment of topical testosterone. Maintenance therapy is common, as up to 85% of women will relapse.
Dr. Schlosser also advised physicians to educate women on how much medication to use, to send them home with a diagram of the affected area, and to use a hand mirror during the exam.
"Patients don’t want to look, but I tell them you’re not going to get better if you don’t know where to put your medication," she said.
Dr. Schlosser reported no relevant conflicts of interest.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2014