User login
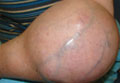
Eccrine Porocarcinoma: A Report of 2 Cases and Review of the Literature
Test your knowledge on eccrine porocarcinoma with MD-IQ: the medical intelligence quiz. Click here to answer 5 questions.
Large Monophasic Synovial Sarcoma: A Case Report and Review of the Literature
Beta-blockers and Melanoma

I have taken beta-blockers for nearly 30 years to combat a non–life-threatening but very disconcerting supraventricular arrhythmia. Also being fair skinned and having suffered through many a blistering sunburn in my youth, I was excited to read the most recent publication regarding the relationship between beta-blocker ingestion and survival from melanoma by De Giorgi et al (Mayo Clin Proc. 2013;88:1196-1203). The Italian investigators collected data from all melanoma patients diagnosed in the dermatology department at the University of Florence (1993-2009). After excluding patients who presented with metastatic disease, they then compared the courses of the remaining patients based on whether or not they had been prescribed beta-blockers for at least 1 year before or after their cutaneous melanoma diagnosis. Of 741 consecutive patients who fit the retrospective study criteria, 79 (11%) had been taking beta-blockers and the remaining 662 (89%) had not been taking beta-blockers.
An analysis based on the multivariate Cox model indicated that the beta-blocker group had improved overall survival after a median follow-up of 4.2 years (P=.005). Looked at in another way, significantly more patients in the untreated group (8%) than the beta-blocker group (3%) experienced disease progression or death (P<.001). The “protective” effect of beta-blockers was so striking that it could be quantified; for each year of beta-blocker use, the risk for death was reduced by 38%. More notable was the fact that the beneficial effect of beta-blocker administration was consistent, even among patients with inherently unfavorable prognostic factors, such as advanced age, thicker lesions, higher frequency of mitoses and ulceration, and nodular melanoma subtype. This large study confirms the results of a much smaller study (N=121) published by the same group in 2011 (Arch Intern Med. 2011;171:779-781).
What’s the issue?
Why might this phenomenon occur? Perhaps blocking sympathetic nervous system neurotransmitters might help avoid the suspected immunosuppression that accompanies stress. Sympathetic neurotransmitters also are known to interact with molecular pathways implicated in abnormal cellular replication, such as the p38/MAPK pathway. Thus, blunting the effects of epinephrine and norepinephrine by blocking some of their receptors might produce a salutatory benefit in the cancer (in this case, melanoma) patient.
Before we all rush out and give all our melanoma patients daily doses of metoprolol, however, De Giorgi et al noted in the most recent study that about two-thirds of the patients were already taking a beta-blocker when their melanoma was diagnosed. Although this study suggests that beta-blockers reduce the risk for recurrence and disease-specific mortality, these drugs clearly do not prevent melanoma in the first place. Furthermore, it should be noted that the number of melanoma patients receiving beta-blockers was small, and conclusions should always be tempered in a retrospective study. The authors duly admit that their investigation indicates the strong need for a truly randomized prospective clinical trial. Of course, administration of beta-blockers is not a totally benign endeavor, as serious hypotension and/or bradycardia may occur, leading to syncope or even worse problems. Finally, the evidence of beta-blocker benefit in melanoma patients is contradictory! A Dutch cohort of 709 melanoma patients recently was studied (Eur J Cancer. 2013;49:3863-3871); this set of investigators found that exposure to beta-blockers did not impact overall melanoma survival regardless of the timing, duration, or dosage of beta-blocker use. In summary, therefore, we still need to determine many things about the relationship between beta-blockers and melanoma survival. That is, if there is a relationship at all. What do you think?

I have taken beta-blockers for nearly 30 years to combat a non–life-threatening but very disconcerting supraventricular arrhythmia. Also being fair skinned and having suffered through many a blistering sunburn in my youth, I was excited to read the most recent publication regarding the relationship between beta-blocker ingestion and survival from melanoma by De Giorgi et al (Mayo Clin Proc. 2013;88:1196-1203). The Italian investigators collected data from all melanoma patients diagnosed in the dermatology department at the University of Florence (1993-2009). After excluding patients who presented with metastatic disease, they then compared the courses of the remaining patients based on whether or not they had been prescribed beta-blockers for at least 1 year before or after their cutaneous melanoma diagnosis. Of 741 consecutive patients who fit the retrospective study criteria, 79 (11%) had been taking beta-blockers and the remaining 662 (89%) had not been taking beta-blockers.
An analysis based on the multivariate Cox model indicated that the beta-blocker group had improved overall survival after a median follow-up of 4.2 years (P=.005). Looked at in another way, significantly more patients in the untreated group (8%) than the beta-blocker group (3%) experienced disease progression or death (P<.001). The “protective” effect of beta-blockers was so striking that it could be quantified; for each year of beta-blocker use, the risk for death was reduced by 38%. More notable was the fact that the beneficial effect of beta-blocker administration was consistent, even among patients with inherently unfavorable prognostic factors, such as advanced age, thicker lesions, higher frequency of mitoses and ulceration, and nodular melanoma subtype. This large study confirms the results of a much smaller study (N=121) published by the same group in 2011 (Arch Intern Med. 2011;171:779-781).
What’s the issue?
Why might this phenomenon occur? Perhaps blocking sympathetic nervous system neurotransmitters might help avoid the suspected immunosuppression that accompanies stress. Sympathetic neurotransmitters also are known to interact with molecular pathways implicated in abnormal cellular replication, such as the p38/MAPK pathway. Thus, blunting the effects of epinephrine and norepinephrine by blocking some of their receptors might produce a salutatory benefit in the cancer (in this case, melanoma) patient.
Before we all rush out and give all our melanoma patients daily doses of metoprolol, however, De Giorgi et al noted in the most recent study that about two-thirds of the patients were already taking a beta-blocker when their melanoma was diagnosed. Although this study suggests that beta-blockers reduce the risk for recurrence and disease-specific mortality, these drugs clearly do not prevent melanoma in the first place. Furthermore, it should be noted that the number of melanoma patients receiving beta-blockers was small, and conclusions should always be tempered in a retrospective study. The authors duly admit that their investigation indicates the strong need for a truly randomized prospective clinical trial. Of course, administration of beta-blockers is not a totally benign endeavor, as serious hypotension and/or bradycardia may occur, leading to syncope or even worse problems. Finally, the evidence of beta-blocker benefit in melanoma patients is contradictory! A Dutch cohort of 709 melanoma patients recently was studied (Eur J Cancer. 2013;49:3863-3871); this set of investigators found that exposure to beta-blockers did not impact overall melanoma survival regardless of the timing, duration, or dosage of beta-blocker use. In summary, therefore, we still need to determine many things about the relationship between beta-blockers and melanoma survival. That is, if there is a relationship at all. What do you think?

I have taken beta-blockers for nearly 30 years to combat a non–life-threatening but very disconcerting supraventricular arrhythmia. Also being fair skinned and having suffered through many a blistering sunburn in my youth, I was excited to read the most recent publication regarding the relationship between beta-blocker ingestion and survival from melanoma by De Giorgi et al (Mayo Clin Proc. 2013;88:1196-1203). The Italian investigators collected data from all melanoma patients diagnosed in the dermatology department at the University of Florence (1993-2009). After excluding patients who presented with metastatic disease, they then compared the courses of the remaining patients based on whether or not they had been prescribed beta-blockers for at least 1 year before or after their cutaneous melanoma diagnosis. Of 741 consecutive patients who fit the retrospective study criteria, 79 (11%) had been taking beta-blockers and the remaining 662 (89%) had not been taking beta-blockers.
An analysis based on the multivariate Cox model indicated that the beta-blocker group had improved overall survival after a median follow-up of 4.2 years (P=.005). Looked at in another way, significantly more patients in the untreated group (8%) than the beta-blocker group (3%) experienced disease progression or death (P<.001). The “protective” effect of beta-blockers was so striking that it could be quantified; for each year of beta-blocker use, the risk for death was reduced by 38%. More notable was the fact that the beneficial effect of beta-blocker administration was consistent, even among patients with inherently unfavorable prognostic factors, such as advanced age, thicker lesions, higher frequency of mitoses and ulceration, and nodular melanoma subtype. This large study confirms the results of a much smaller study (N=121) published by the same group in 2011 (Arch Intern Med. 2011;171:779-781).
What’s the issue?
Why might this phenomenon occur? Perhaps blocking sympathetic nervous system neurotransmitters might help avoid the suspected immunosuppression that accompanies stress. Sympathetic neurotransmitters also are known to interact with molecular pathways implicated in abnormal cellular replication, such as the p38/MAPK pathway. Thus, blunting the effects of epinephrine and norepinephrine by blocking some of their receptors might produce a salutatory benefit in the cancer (in this case, melanoma) patient.
Before we all rush out and give all our melanoma patients daily doses of metoprolol, however, De Giorgi et al noted in the most recent study that about two-thirds of the patients were already taking a beta-blocker when their melanoma was diagnosed. Although this study suggests that beta-blockers reduce the risk for recurrence and disease-specific mortality, these drugs clearly do not prevent melanoma in the first place. Furthermore, it should be noted that the number of melanoma patients receiving beta-blockers was small, and conclusions should always be tempered in a retrospective study. The authors duly admit that their investigation indicates the strong need for a truly randomized prospective clinical trial. Of course, administration of beta-blockers is not a totally benign endeavor, as serious hypotension and/or bradycardia may occur, leading to syncope or even worse problems. Finally, the evidence of beta-blocker benefit in melanoma patients is contradictory! A Dutch cohort of 709 melanoma patients recently was studied (Eur J Cancer. 2013;49:3863-3871); this set of investigators found that exposure to beta-blockers did not impact overall melanoma survival regardless of the timing, duration, or dosage of beta-blocker use. In summary, therefore, we still need to determine many things about the relationship between beta-blockers and melanoma survival. That is, if there is a relationship at all. What do you think?
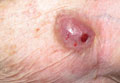
What Is Your Diagnosis? Spiradenocarcinoma
Test your knowledge on diagnosing cancer with MD-IQ: the medical intelligence quiz. Click here to answer 5 questions.
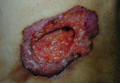
Cutaneous SCC in a Renal Transplant Patient Derived From Donor Kidney Tumor Cells: Should Donor Transplant Organs Undergo Genetic Profiling for Cancer-Associated Mutations?

Squamous cell carcinoma (SCC) on sun-exposed sites following prolonged immunosuppression is one of the main long-term complications of allogeneic transplantations. In an article published in the Journal of Clinical Investigation (2013;123:3797-3801), the investigators demonstrated that the SCC tumor cells from a renal transplant patient had the donor genotype and harbored a TP53 (tumor-suppressing p53) mutation in codon 175; in addition, the same TP53 mutation had previously been documented 7 years earlier in p53+ cells in the renal tubules from a kidney graft biopsy. The observations in this patient provide evidence that the kidney donor can contribute to subsequent SCCs in renal transplant recipients.
What’s the issue?
Donor contribution to the malignant epithelium of cutaneous cancer in organ transplant recipients has several important implications. First, this observation provides additional insight into the initiation and progression of tumor carcinogenesis. Second, because there is longer survival of patients following renal transplant and therefore a prolonged duration of immunosuppression in these individuals, the development of long-term complications such as SCC of sun-exposed skin may be greater. Third, should genetic profiling of kidneys from potential donors to screen for cancer-associated mutations be performed? And fourth, is this phenomenon restricted only to patients receiving kidneys or might it be a possible complication in the recipients of other organs such as hearts, lungs, or even faces?

Squamous cell carcinoma (SCC) on sun-exposed sites following prolonged immunosuppression is one of the main long-term complications of allogeneic transplantations. In an article published in the Journal of Clinical Investigation (2013;123:3797-3801), the investigators demonstrated that the SCC tumor cells from a renal transplant patient had the donor genotype and harbored a TP53 (tumor-suppressing p53) mutation in codon 175; in addition, the same TP53 mutation had previously been documented 7 years earlier in p53+ cells in the renal tubules from a kidney graft biopsy. The observations in this patient provide evidence that the kidney donor can contribute to subsequent SCCs in renal transplant recipients.
What’s the issue?
Donor contribution to the malignant epithelium of cutaneous cancer in organ transplant recipients has several important implications. First, this observation provides additional insight into the initiation and progression of tumor carcinogenesis. Second, because there is longer survival of patients following renal transplant and therefore a prolonged duration of immunosuppression in these individuals, the development of long-term complications such as SCC of sun-exposed skin may be greater. Third, should genetic profiling of kidneys from potential donors to screen for cancer-associated mutations be performed? And fourth, is this phenomenon restricted only to patients receiving kidneys or might it be a possible complication in the recipients of other organs such as hearts, lungs, or even faces?

Squamous cell carcinoma (SCC) on sun-exposed sites following prolonged immunosuppression is one of the main long-term complications of allogeneic transplantations. In an article published in the Journal of Clinical Investigation (2013;123:3797-3801), the investigators demonstrated that the SCC tumor cells from a renal transplant patient had the donor genotype and harbored a TP53 (tumor-suppressing p53) mutation in codon 175; in addition, the same TP53 mutation had previously been documented 7 years earlier in p53+ cells in the renal tubules from a kidney graft biopsy. The observations in this patient provide evidence that the kidney donor can contribute to subsequent SCCs in renal transplant recipients.
What’s the issue?
Donor contribution to the malignant epithelium of cutaneous cancer in organ transplant recipients has several important implications. First, this observation provides additional insight into the initiation and progression of tumor carcinogenesis. Second, because there is longer survival of patients following renal transplant and therefore a prolonged duration of immunosuppression in these individuals, the development of long-term complications such as SCC of sun-exposed skin may be greater. Third, should genetic profiling of kidneys from potential donors to screen for cancer-associated mutations be performed? And fourth, is this phenomenon restricted only to patients receiving kidneys or might it be a possible complication in the recipients of other organs such as hearts, lungs, or even faces?