User login
Basal Cell Carcinoma: Analysis of Factors Associated With Incomplete Excision at a Referral Hospital in Southern Spain
Cutaneous Squamous Cell Carcinoma With Perineural Invasion: A Case Report and Review of the Literature
Case Report
A 74-year-old man with a history of squamous cell carcinoma (SCC) on the right side of the temple that was treated with Mohs micrographic surgery (MMS) 3 years prior presented with a burning and tingling sensation of 3 months’ duration in the medial border of the repair scar. The patient denied prior anesthesia or muscle weakness of the face as well as any loss or change in vision.
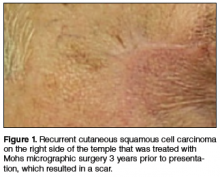
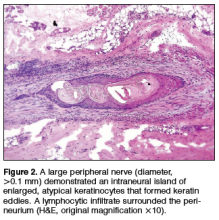
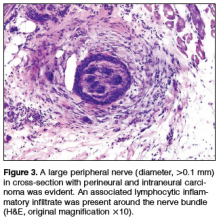
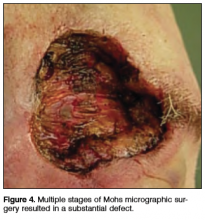
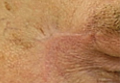
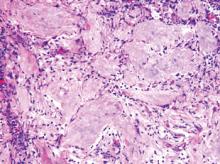
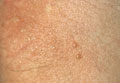
Physical examination revealed a well-healed advancement flap scar with induration at the medial border (Figure 1). Biopsy results were positive for recurrent SCC. Based on anatomic location, clinical symptoms, and tumor recurrence, treatment with MMS was initiated. Mohs sections demonstrated perineural invasion (PNI) (Figures 2 and 3). Multiple treatment stages were required for tumor clearance following the retrograde course of a nerve, which resulted in a substantial defect (Figure 4). The defect was allowed to heal by second intention followed by radiation therapy.
Comment
Incidence and Pathogenesis—Perineural invasion was first described by Cruveilhier1 in a report of invasion of the facial nerve in a patient with mammary carcinoma. Neumann2 reported the first case of a primary cutaneous lesion exhibiting PNI in a patient with a primary carcinoma of the lower lip with invasion and spread along the mental nerve. Perineural invasion is seen in approximately 5% of 200,000 total cases of cutaneous SCC reported annually in the United States.3,4 Other malignancies exhibit PNI more frequently, such as microcystic adnexal carcinoma of the skin, which has been reported to have an 80% rate of perineural growth.5
Perineural invasion can involve nerves of variable thickness, but invasion of larger nerves typically portends a poorer prognosis.6 Characteristics of cutaneous SCC that predispose the lesion to PNI include size greater than 2 cm, male gender, location on the face, and prior treatment of the lesion.6,7 In a study of cutaneous SCC, Leibovitch et al7 found PNI in 4.7% (36/772) of primary lesions and 6.9% (34/491) of recurrent lesions. In another study of 180 SCC tumors of the head and neck with PNI, Carter et al8 found that PNI was most commonly seen in tumors that were greater than 2.5 cm, suggesting that larger lesions have an increased predisposition for PNI.
The mechanism(s) by which PNI develops from these malignancies has not been fully elucidated, but some clues have been found. Vural et al9 showed a statistically significant difference (P<.01) in expression of neural cell adhesion molecules with 93% (38/41) of SCCs with PNI showing evidence of expression versus 36% (9/25) of SCCs without PNI. Chen-Tsai et al10 also suggested that levels of neural cell adhesion molecules may be a factor in determining the metastatic potential of cutaneous SCCs and that levels of neurotrophic tyrosine kinase receptor type 1 (TrkA) may predict PNI, but their study results lacked statistical power to form a firm conclusion.
Diagnosis and Prognosis—Perineural invasion can be diagnosed clinically, radiologically, or microscopically. On clinical examination, PNI is suggested by findings of neuropathy most frequently in cranial nerves V and/or VII, likely due to their extensive subcutaneous distribution.11 Common symptoms include pain, loss of motor skills, anesthesia, dysesthesia, and/or paresthesia (ie, tingling, burning, pricking, numbness).12,13 In a study of 72 cases, Goepfert et al14 found that only 40% (29/72) of patients with pathologically confirmed PNI presented with clinical symptoms and these patients had a poorer prognosis.
Radiologically, PNI can be identified via computed tomography or magnetic resonance imaging through findings of enlargement or abnormal enhancement of the nerve, obliteration of the normal fat plane surrounding the nerve, or erosion or enlargement of its related foramen.15 Magnetic resonance imaging is the preferred method for assessing enhancement of the nerve, while computed tomography is preferred to assess involvement of bone.16,17 Microscopically, there is some debate as to what defines PNI. Suggested findings include the presence of cells inside the epineurium, involvement of nerves outside the main bulk of the tumor, or presence of tumor cells surrounding a nerve.18
These definitions have prognostic significance. Mendenhall et al16 found that patients with radiologic evidence of PNI without clinical symptoms had a higher cure rate using surgery and postoperative irradiation compared to patients with clinical symptoms (80% vs 45%). Although prognosis generally is good in patients with cutaneous SCC without PNI, prognosis is notably poorer when PNI is present due to the association of this finding with increased tumor recurrence and both local and distant metastasis.13 Most frequently, cutaneous SCC with PNI spreads proximally, which can lead to invasion into the base of the brain, but also can extend distally, leading to increased local burden.12,19 In a study of 64 patients with mucosal SCC, Soo et al20 found that patients with lesions that exhibited PNI had a 5-year survival of 16% versus 44% in those without PNI. In their study of SCC of the head and neck, Goepfert et al14 reported that 46% (33/72) of patients with PNI had died or were alive with recurrence at 2 years’ follow-up versus 9.1% (41/448) of patients without PNI. In a systematic review of outcomes, Jambusaria-Pahlajani et al21 reported a disease-specific death rate of 16% for cutaneous SCC with PNI compared to 4% for SCC without PNI.
Perineural invasion can be further classified as clinical or microscopic (incidental) for prognostic purposes. A study by Garcia-Serra et al13 found that patients with clinical PNI had a notably poorer prognosis than those with microscopic (incidental) PNI. The clinical group achieved a local control rate of 55% at 5 years’ follow-up versus 87% in the microscopic group. McCord et al22 found a 5-year local control rate of 78% for microscopic (incidental) PNI versus 50% for clinical PNI; they also found that patients with radiologic evidence of PNI had a worse prognosis, noting that patients with radiologic evidence of PNI were nearly all clinically symptomatic.
Prognosis also is altered by the diameter of the nerve involved. In a study of 48 patients, Ross et al23 found that patients with cutaneous SCC involving small-caliber nerves (diameter, ≤0.1 mm) had a 0% disease-specific death rate versus 32% in those with large-caliber nerves (>0.1 mm). Perineural involvement of small-caliber nerves (<0.1 mm) was a positive prognostic indicator in that it was associated with smaller tumor diameter, more shallow invasion, and increased likelihood to be primary tumors.23 In a recent study, Jambusaria-Pahlajani et al24 investigated tumor staging for cutaneous SCC and reported that PNI is a statistically independent prognostic risk factor for nodal metastasis (subhazard ratio, 2.2 [95% confidence interval, 0.9-5.1]) and disease-specific death (subhazard ratio, 3.4 [95% confidence interval, 0.9-13.3]). Of interest, this increased risk applied only to PNI in nerves that were greater than 0.1 mm.24
Treatment Options—Management of confirmed cases of cutaneous SCC with PNI is difficult because of the nature of the lesions, including their increased propensity for metastasis, increased frequency of poorly differentiated cell types, highly aggressive nature, and the unique challenge of skip lesions.4,16 Skip lesions are found microscopically and show (or appear to show) neoplastic cells invading a nerve in a discontinuous fashion. This phenomenon has been suggested as one explanation for the relatively higher postsurgical recurrence rate of SCC with PNI compared to lesions without PNI.7 They are of particular interest when removing cutaneous SCC with PNI using MMS and attempting to define clear margins. Despite this limitation, MMS generally is accepted as the primary mode of excision of cutaneous SCCs with PNI, as it has the highest known cure rate.7 Cottel4 did not report any cases of local recurrence over 1 to 42 months in 17 patients who were treated with MMS, in contrast to Rowe et al25 who demonstrated that traditional surgical excision had a 47% (34/72) local recurrence rate; however, it bears noting that the varying follow-up periods in the Cottel4 study may underestimate recurrence rate. Leibovitch et al7 had similar findings in their prospective case series study of 70 patients, which revealed an 8% recurrence rate within 5 years in patients treated with MMS, a rate lower than other non-MMS modalities. In this same study, the authors noted that some researchers believe an additional level should be taken with MMS beyond the appearance of free margins in cases with PNI.7
Jambusaria-Pahlajani et al21 reported that PNI is one of the most common reasons cited for using adjuvant radiation therapy for cases of cutaneous SCC because of the known propensity of local recurrence; however, in 74 reviewed cases, there was no statistically significant difference in outcomes in cases of surgery alone versus surgery and adjuvant irradiation. Radiation therapy is a possible alternative primary treatment of cutaneous SCC with PNI, especially in cases of perineural involvement that is extensive or affects proximal portions of cranial nerves when surgery is a less viable option.17 Mendenhall et al16 suggested that patients with positive margins after excision who display extensive PNI should be treated with adjuvant irradiation locally and along the course of the involved nerve to the skull base.
Conclusion
Physicians should recognize the importance of early detection of PNI in cases of cutaneous SCC. A thorough history with good neurologic examination of the head and neck in patients with cutaneous SCC is imperative so patients can be treated earlier in the course of the lesion, increasing the likelihood of local control, minimizing the risk for future recurrence, and decreasing mortality.
1. Cruveilhier J. Maladies des nerfs. In: Cruveilhier J, ed. Anatomie Pathologique du Corps Humain. 2nd ed. Paris, France: JB Bailliere; 1835:1-3.
2. Neumann E. Secondare cancroid infiltration des nervus mentalis bei einem fall von lippincroid. Arch Pathol Anat. 1862;24:201-205.
3. Salasche S. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
4. Cottel WI. Perineural invasion by squamous-cell carcinoma. J Dermatol Surg Oncol. 1982;8:589-600.
5. Cooper PH, Mills SE, Leonard DD, et al. Sclerosing sweat duct (syringomatous) carcinoma. Am J Surg Pathol. 1985;9:422-433.
6. Ross AS, Whalen FM, Elenitsas R, et al. Diameter of involved nerves predicts outcomes in cutaneous squamous cell carcinoma with perineural invasion: an investigator-blinded retrospective cohort study. Dermatol Surg. 2009;35:1859-1866.
7. Leibovitch I, Huilgol SC, Selva D, et al. Cutaneous squamous cell carcinoma treated with Mohs micrographic surgery in Australia II. perineural invasion. J Am Acad Dermatol. 2005;53:261-266.
8. Carter RL, Foster CS, Dinsdale EA, et al. Perineural spread by squamous carcinomas of the head and neck: a morphological study using antiaxonal and antimyelin monoclonal antibodies. J Clin Pathol. 1983;36:269-275.
9. Vural E, Hutcheson J, Korourian S, et al. Correlation of neural cell adhesion molecules with perineural spread of squamous cell carcinoma of the head and neck. Otolaryngol Head Neck Surg. 2000;122:717-720.
10. Chen-Tsai CP, Colome-Grimmer M, Wagner RF Jr. Correlations among neural cell adhesion molecule, nerve growth factor, and its receptors, TrkA, TrkB, TrkC, and p75, in perineural invasion by basal cell and cutaneous squamous cell carcinomas. Dermatol Surg. 2004;30:1009-1016.
11. McCord M, Mendenhall WM, Parsons JT, et al. Skin cancer of the head and neck with clinical perineural invasion. Int J Radiat Oncol Biol Phys. 2000;47:89-93.
12. Ampil FL, Hardin JC, Peskind SP, et al. Perineural invasion in skin cancer of the head and neck: a review of nine cases. J Oral Maxillofac Surg. 1995;53:34-38.
13. Garcia-Serra A, Hinerman RW, Mendenhall WM, et al. Carcinoma of the skin with perineural invasion. Head Neck. 2003;25:1027-1033.
14. Goepfert H, Dichtel WJ, Medina JE, et al. Perineural invasion in squamous cell skin carcinoma of the head and neck. Am J Surg. 1984;148:542-547.
15. Galloway TJ, Morris CG, Mancuso AA, et al. Impact of radiographic findings on prognosis for skin carcinoma with clinical perineural invasion. Cancer. 2005;103:1254-1257.
16. Mendenhall WM, Amdur RJ, Williams LS, et al. Carcinoma of the skin of the head and neck with perineural invasion. Head Neck. 2002;24:78-83.
17. Williams LS, Mancuso AA, Mendenhall WM. Perineural spread of cutaneous squamous and basal cell carcinoma: CT and MR detection and its impact on patient management and prognosis. Int J Radiat Oncol Biol Phys. 2001;49:1061-1069.
18. Veness MJ, Biankin S. Perineural spread leading to orbital invasion from skin cancer. Australasian Radiol. 2000;44:296-302.
19. Feasel AM, Brown TJ, Bogle MA, et al. Perineural invasion of cutaneous malignancies. Dermatol Surg. 2001;27:531-542.
20. Soo K, Carter RL, O’Brien CJ, et al. Prognostic implications of perineural spread in squamous carcinomas of the head and neck. Laryngoscope. 1986;96:1145-1148.
21. Jambusaria-Pahlajani A, Miller CJ, Quon H, et al. Surgical monotherapy versus surgery plus adjuvant radiotherapy in high-risk cutaneous squamous cell carcinoma: a systematic review of outcomes. Dermatol Surg. 2009;35:574-584.
22. McCord MW, Mendenhall WM, Parsons JT, et al. Skin cancer of the head and neck with incidental microscopic perineural invasion. Int J Radiat Oncol Biol Phys. 1999;43:591-595.
23. Ross AS, Whalen FM, Elenitsas R, et al. Diameter of involved nerves predicts outcomes in cutaneous squamous cell carcinoma with perineural invasion: an investigator-blinded retrospective cohort study. Dermatol Surg. 2009;35:1859-1866.
24. Jambusaria-Pahlajani A, Kanetsky PA, Karia PS, et al. Evaluation of AJCC tumor staging for cutaneous squamous cell carcinoma and a proposed alternative tumor staging system. JAMA Dermatol. 2013;149:402-410.
25. Rowe DE, Carroll RJ, Day CL. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
Case Report
A 74-year-old man with a history of squamous cell carcinoma (SCC) on the right side of the temple that was treated with Mohs micrographic surgery (MMS) 3 years prior presented with a burning and tingling sensation of 3 months’ duration in the medial border of the repair scar. The patient denied prior anesthesia or muscle weakness of the face as well as any loss or change in vision.
Physical examination revealed a well-healed advancement flap scar with induration at the medial border (Figure 1). Biopsy results were positive for recurrent SCC. Based on anatomic location, clinical symptoms, and tumor recurrence, treatment with MMS was initiated. Mohs sections demonstrated perineural invasion (PNI) (Figures 2 and 3). Multiple treatment stages were required for tumor clearance following the retrograde course of a nerve, which resulted in a substantial defect (Figure 4). The defect was allowed to heal by second intention followed by radiation therapy.
Comment
Incidence and Pathogenesis—Perineural invasion was first described by Cruveilhier1 in a report of invasion of the facial nerve in a patient with mammary carcinoma. Neumann2 reported the first case of a primary cutaneous lesion exhibiting PNI in a patient with a primary carcinoma of the lower lip with invasion and spread along the mental nerve. Perineural invasion is seen in approximately 5% of 200,000 total cases of cutaneous SCC reported annually in the United States.3,4 Other malignancies exhibit PNI more frequently, such as microcystic adnexal carcinoma of the skin, which has been reported to have an 80% rate of perineural growth.5
Perineural invasion can involve nerves of variable thickness, but invasion of larger nerves typically portends a poorer prognosis.6 Characteristics of cutaneous SCC that predispose the lesion to PNI include size greater than 2 cm, male gender, location on the face, and prior treatment of the lesion.6,7 In a study of cutaneous SCC, Leibovitch et al7 found PNI in 4.7% (36/772) of primary lesions and 6.9% (34/491) of recurrent lesions. In another study of 180 SCC tumors of the head and neck with PNI, Carter et al8 found that PNI was most commonly seen in tumors that were greater than 2.5 cm, suggesting that larger lesions have an increased predisposition for PNI.
The mechanism(s) by which PNI develops from these malignancies has not been fully elucidated, but some clues have been found. Vural et al9 showed a statistically significant difference (P<.01) in expression of neural cell adhesion molecules with 93% (38/41) of SCCs with PNI showing evidence of expression versus 36% (9/25) of SCCs without PNI. Chen-Tsai et al10 also suggested that levels of neural cell adhesion molecules may be a factor in determining the metastatic potential of cutaneous SCCs and that levels of neurotrophic tyrosine kinase receptor type 1 (TrkA) may predict PNI, but their study results lacked statistical power to form a firm conclusion.
Diagnosis and Prognosis—Perineural invasion can be diagnosed clinically, radiologically, or microscopically. On clinical examination, PNI is suggested by findings of neuropathy most frequently in cranial nerves V and/or VII, likely due to their extensive subcutaneous distribution.11 Common symptoms include pain, loss of motor skills, anesthesia, dysesthesia, and/or paresthesia (ie, tingling, burning, pricking, numbness).12,13 In a study of 72 cases, Goepfert et al14 found that only 40% (29/72) of patients with pathologically confirmed PNI presented with clinical symptoms and these patients had a poorer prognosis.
Radiologically, PNI can be identified via computed tomography or magnetic resonance imaging through findings of enlargement or abnormal enhancement of the nerve, obliteration of the normal fat plane surrounding the nerve, or erosion or enlargement of its related foramen.15 Magnetic resonance imaging is the preferred method for assessing enhancement of the nerve, while computed tomography is preferred to assess involvement of bone.16,17 Microscopically, there is some debate as to what defines PNI. Suggested findings include the presence of cells inside the epineurium, involvement of nerves outside the main bulk of the tumor, or presence of tumor cells surrounding a nerve.18
These definitions have prognostic significance. Mendenhall et al16 found that patients with radiologic evidence of PNI without clinical symptoms had a higher cure rate using surgery and postoperative irradiation compared to patients with clinical symptoms (80% vs 45%). Although prognosis generally is good in patients with cutaneous SCC without PNI, prognosis is notably poorer when PNI is present due to the association of this finding with increased tumor recurrence and both local and distant metastasis.13 Most frequently, cutaneous SCC with PNI spreads proximally, which can lead to invasion into the base of the brain, but also can extend distally, leading to increased local burden.12,19 In a study of 64 patients with mucosal SCC, Soo et al20 found that patients with lesions that exhibited PNI had a 5-year survival of 16% versus 44% in those without PNI. In their study of SCC of the head and neck, Goepfert et al14 reported that 46% (33/72) of patients with PNI had died or were alive with recurrence at 2 years’ follow-up versus 9.1% (41/448) of patients without PNI. In a systematic review of outcomes, Jambusaria-Pahlajani et al21 reported a disease-specific death rate of 16% for cutaneous SCC with PNI compared to 4% for SCC without PNI.
Perineural invasion can be further classified as clinical or microscopic (incidental) for prognostic purposes. A study by Garcia-Serra et al13 found that patients with clinical PNI had a notably poorer prognosis than those with microscopic (incidental) PNI. The clinical group achieved a local control rate of 55% at 5 years’ follow-up versus 87% in the microscopic group. McCord et al22 found a 5-year local control rate of 78% for microscopic (incidental) PNI versus 50% for clinical PNI; they also found that patients with radiologic evidence of PNI had a worse prognosis, noting that patients with radiologic evidence of PNI were nearly all clinically symptomatic.
Prognosis also is altered by the diameter of the nerve involved. In a study of 48 patients, Ross et al23 found that patients with cutaneous SCC involving small-caliber nerves (diameter, ≤0.1 mm) had a 0% disease-specific death rate versus 32% in those with large-caliber nerves (>0.1 mm). Perineural involvement of small-caliber nerves (<0.1 mm) was a positive prognostic indicator in that it was associated with smaller tumor diameter, more shallow invasion, and increased likelihood to be primary tumors.23 In a recent study, Jambusaria-Pahlajani et al24 investigated tumor staging for cutaneous SCC and reported that PNI is a statistically independent prognostic risk factor for nodal metastasis (subhazard ratio, 2.2 [95% confidence interval, 0.9-5.1]) and disease-specific death (subhazard ratio, 3.4 [95% confidence interval, 0.9-13.3]). Of interest, this increased risk applied only to PNI in nerves that were greater than 0.1 mm.24
Treatment Options—Management of confirmed cases of cutaneous SCC with PNI is difficult because of the nature of the lesions, including their increased propensity for metastasis, increased frequency of poorly differentiated cell types, highly aggressive nature, and the unique challenge of skip lesions.4,16 Skip lesions are found microscopically and show (or appear to show) neoplastic cells invading a nerve in a discontinuous fashion. This phenomenon has been suggested as one explanation for the relatively higher postsurgical recurrence rate of SCC with PNI compared to lesions without PNI.7 They are of particular interest when removing cutaneous SCC with PNI using MMS and attempting to define clear margins. Despite this limitation, MMS generally is accepted as the primary mode of excision of cutaneous SCCs with PNI, as it has the highest known cure rate.7 Cottel4 did not report any cases of local recurrence over 1 to 42 months in 17 patients who were treated with MMS, in contrast to Rowe et al25 who demonstrated that traditional surgical excision had a 47% (34/72) local recurrence rate; however, it bears noting that the varying follow-up periods in the Cottel4 study may underestimate recurrence rate. Leibovitch et al7 had similar findings in their prospective case series study of 70 patients, which revealed an 8% recurrence rate within 5 years in patients treated with MMS, a rate lower than other non-MMS modalities. In this same study, the authors noted that some researchers believe an additional level should be taken with MMS beyond the appearance of free margins in cases with PNI.7
Jambusaria-Pahlajani et al21 reported that PNI is one of the most common reasons cited for using adjuvant radiation therapy for cases of cutaneous SCC because of the known propensity of local recurrence; however, in 74 reviewed cases, there was no statistically significant difference in outcomes in cases of surgery alone versus surgery and adjuvant irradiation. Radiation therapy is a possible alternative primary treatment of cutaneous SCC with PNI, especially in cases of perineural involvement that is extensive or affects proximal portions of cranial nerves when surgery is a less viable option.17 Mendenhall et al16 suggested that patients with positive margins after excision who display extensive PNI should be treated with adjuvant irradiation locally and along the course of the involved nerve to the skull base.
Conclusion
Physicians should recognize the importance of early detection of PNI in cases of cutaneous SCC. A thorough history with good neurologic examination of the head and neck in patients with cutaneous SCC is imperative so patients can be treated earlier in the course of the lesion, increasing the likelihood of local control, minimizing the risk for future recurrence, and decreasing mortality.
Case Report
A 74-year-old man with a history of squamous cell carcinoma (SCC) on the right side of the temple that was treated with Mohs micrographic surgery (MMS) 3 years prior presented with a burning and tingling sensation of 3 months’ duration in the medial border of the repair scar. The patient denied prior anesthesia or muscle weakness of the face as well as any loss or change in vision.
Physical examination revealed a well-healed advancement flap scar with induration at the medial border (Figure 1). Biopsy results were positive for recurrent SCC. Based on anatomic location, clinical symptoms, and tumor recurrence, treatment with MMS was initiated. Mohs sections demonstrated perineural invasion (PNI) (Figures 2 and 3). Multiple treatment stages were required for tumor clearance following the retrograde course of a nerve, which resulted in a substantial defect (Figure 4). The defect was allowed to heal by second intention followed by radiation therapy.
Comment
Incidence and Pathogenesis—Perineural invasion was first described by Cruveilhier1 in a report of invasion of the facial nerve in a patient with mammary carcinoma. Neumann2 reported the first case of a primary cutaneous lesion exhibiting PNI in a patient with a primary carcinoma of the lower lip with invasion and spread along the mental nerve. Perineural invasion is seen in approximately 5% of 200,000 total cases of cutaneous SCC reported annually in the United States.3,4 Other malignancies exhibit PNI more frequently, such as microcystic adnexal carcinoma of the skin, which has been reported to have an 80% rate of perineural growth.5
Perineural invasion can involve nerves of variable thickness, but invasion of larger nerves typically portends a poorer prognosis.6 Characteristics of cutaneous SCC that predispose the lesion to PNI include size greater than 2 cm, male gender, location on the face, and prior treatment of the lesion.6,7 In a study of cutaneous SCC, Leibovitch et al7 found PNI in 4.7% (36/772) of primary lesions and 6.9% (34/491) of recurrent lesions. In another study of 180 SCC tumors of the head and neck with PNI, Carter et al8 found that PNI was most commonly seen in tumors that were greater than 2.5 cm, suggesting that larger lesions have an increased predisposition for PNI.
The mechanism(s) by which PNI develops from these malignancies has not been fully elucidated, but some clues have been found. Vural et al9 showed a statistically significant difference (P<.01) in expression of neural cell adhesion molecules with 93% (38/41) of SCCs with PNI showing evidence of expression versus 36% (9/25) of SCCs without PNI. Chen-Tsai et al10 also suggested that levels of neural cell adhesion molecules may be a factor in determining the metastatic potential of cutaneous SCCs and that levels of neurotrophic tyrosine kinase receptor type 1 (TrkA) may predict PNI, but their study results lacked statistical power to form a firm conclusion.
Diagnosis and Prognosis—Perineural invasion can be diagnosed clinically, radiologically, or microscopically. On clinical examination, PNI is suggested by findings of neuropathy most frequently in cranial nerves V and/or VII, likely due to their extensive subcutaneous distribution.11 Common symptoms include pain, loss of motor skills, anesthesia, dysesthesia, and/or paresthesia (ie, tingling, burning, pricking, numbness).12,13 In a study of 72 cases, Goepfert et al14 found that only 40% (29/72) of patients with pathologically confirmed PNI presented with clinical symptoms and these patients had a poorer prognosis.
Radiologically, PNI can be identified via computed tomography or magnetic resonance imaging through findings of enlargement or abnormal enhancement of the nerve, obliteration of the normal fat plane surrounding the nerve, or erosion or enlargement of its related foramen.15 Magnetic resonance imaging is the preferred method for assessing enhancement of the nerve, while computed tomography is preferred to assess involvement of bone.16,17 Microscopically, there is some debate as to what defines PNI. Suggested findings include the presence of cells inside the epineurium, involvement of nerves outside the main bulk of the tumor, or presence of tumor cells surrounding a nerve.18
These definitions have prognostic significance. Mendenhall et al16 found that patients with radiologic evidence of PNI without clinical symptoms had a higher cure rate using surgery and postoperative irradiation compared to patients with clinical symptoms (80% vs 45%). Although prognosis generally is good in patients with cutaneous SCC without PNI, prognosis is notably poorer when PNI is present due to the association of this finding with increased tumor recurrence and both local and distant metastasis.13 Most frequently, cutaneous SCC with PNI spreads proximally, which can lead to invasion into the base of the brain, but also can extend distally, leading to increased local burden.12,19 In a study of 64 patients with mucosal SCC, Soo et al20 found that patients with lesions that exhibited PNI had a 5-year survival of 16% versus 44% in those without PNI. In their study of SCC of the head and neck, Goepfert et al14 reported that 46% (33/72) of patients with PNI had died or were alive with recurrence at 2 years’ follow-up versus 9.1% (41/448) of patients without PNI. In a systematic review of outcomes, Jambusaria-Pahlajani et al21 reported a disease-specific death rate of 16% for cutaneous SCC with PNI compared to 4% for SCC without PNI.
Perineural invasion can be further classified as clinical or microscopic (incidental) for prognostic purposes. A study by Garcia-Serra et al13 found that patients with clinical PNI had a notably poorer prognosis than those with microscopic (incidental) PNI. The clinical group achieved a local control rate of 55% at 5 years’ follow-up versus 87% in the microscopic group. McCord et al22 found a 5-year local control rate of 78% for microscopic (incidental) PNI versus 50% for clinical PNI; they also found that patients with radiologic evidence of PNI had a worse prognosis, noting that patients with radiologic evidence of PNI were nearly all clinically symptomatic.
Prognosis also is altered by the diameter of the nerve involved. In a study of 48 patients, Ross et al23 found that patients with cutaneous SCC involving small-caliber nerves (diameter, ≤0.1 mm) had a 0% disease-specific death rate versus 32% in those with large-caliber nerves (>0.1 mm). Perineural involvement of small-caliber nerves (<0.1 mm) was a positive prognostic indicator in that it was associated with smaller tumor diameter, more shallow invasion, and increased likelihood to be primary tumors.23 In a recent study, Jambusaria-Pahlajani et al24 investigated tumor staging for cutaneous SCC and reported that PNI is a statistically independent prognostic risk factor for nodal metastasis (subhazard ratio, 2.2 [95% confidence interval, 0.9-5.1]) and disease-specific death (subhazard ratio, 3.4 [95% confidence interval, 0.9-13.3]). Of interest, this increased risk applied only to PNI in nerves that were greater than 0.1 mm.24
Treatment Options—Management of confirmed cases of cutaneous SCC with PNI is difficult because of the nature of the lesions, including their increased propensity for metastasis, increased frequency of poorly differentiated cell types, highly aggressive nature, and the unique challenge of skip lesions.4,16 Skip lesions are found microscopically and show (or appear to show) neoplastic cells invading a nerve in a discontinuous fashion. This phenomenon has been suggested as one explanation for the relatively higher postsurgical recurrence rate of SCC with PNI compared to lesions without PNI.7 They are of particular interest when removing cutaneous SCC with PNI using MMS and attempting to define clear margins. Despite this limitation, MMS generally is accepted as the primary mode of excision of cutaneous SCCs with PNI, as it has the highest known cure rate.7 Cottel4 did not report any cases of local recurrence over 1 to 42 months in 17 patients who were treated with MMS, in contrast to Rowe et al25 who demonstrated that traditional surgical excision had a 47% (34/72) local recurrence rate; however, it bears noting that the varying follow-up periods in the Cottel4 study may underestimate recurrence rate. Leibovitch et al7 had similar findings in their prospective case series study of 70 patients, which revealed an 8% recurrence rate within 5 years in patients treated with MMS, a rate lower than other non-MMS modalities. In this same study, the authors noted that some researchers believe an additional level should be taken with MMS beyond the appearance of free margins in cases with PNI.7
Jambusaria-Pahlajani et al21 reported that PNI is one of the most common reasons cited for using adjuvant radiation therapy for cases of cutaneous SCC because of the known propensity of local recurrence; however, in 74 reviewed cases, there was no statistically significant difference in outcomes in cases of surgery alone versus surgery and adjuvant irradiation. Radiation therapy is a possible alternative primary treatment of cutaneous SCC with PNI, especially in cases of perineural involvement that is extensive or affects proximal portions of cranial nerves when surgery is a less viable option.17 Mendenhall et al16 suggested that patients with positive margins after excision who display extensive PNI should be treated with adjuvant irradiation locally and along the course of the involved nerve to the skull base.
Conclusion
Physicians should recognize the importance of early detection of PNI in cases of cutaneous SCC. A thorough history with good neurologic examination of the head and neck in patients with cutaneous SCC is imperative so patients can be treated earlier in the course of the lesion, increasing the likelihood of local control, minimizing the risk for future recurrence, and decreasing mortality.
1. Cruveilhier J. Maladies des nerfs. In: Cruveilhier J, ed. Anatomie Pathologique du Corps Humain. 2nd ed. Paris, France: JB Bailliere; 1835:1-3.
2. Neumann E. Secondare cancroid infiltration des nervus mentalis bei einem fall von lippincroid. Arch Pathol Anat. 1862;24:201-205.
3. Salasche S. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
4. Cottel WI. Perineural invasion by squamous-cell carcinoma. J Dermatol Surg Oncol. 1982;8:589-600.
5. Cooper PH, Mills SE, Leonard DD, et al. Sclerosing sweat duct (syringomatous) carcinoma. Am J Surg Pathol. 1985;9:422-433.
6. Ross AS, Whalen FM, Elenitsas R, et al. Diameter of involved nerves predicts outcomes in cutaneous squamous cell carcinoma with perineural invasion: an investigator-blinded retrospective cohort study. Dermatol Surg. 2009;35:1859-1866.
7. Leibovitch I, Huilgol SC, Selva D, et al. Cutaneous squamous cell carcinoma treated with Mohs micrographic surgery in Australia II. perineural invasion. J Am Acad Dermatol. 2005;53:261-266.
8. Carter RL, Foster CS, Dinsdale EA, et al. Perineural spread by squamous carcinomas of the head and neck: a morphological study using antiaxonal and antimyelin monoclonal antibodies. J Clin Pathol. 1983;36:269-275.
9. Vural E, Hutcheson J, Korourian S, et al. Correlation of neural cell adhesion molecules with perineural spread of squamous cell carcinoma of the head and neck. Otolaryngol Head Neck Surg. 2000;122:717-720.
10. Chen-Tsai CP, Colome-Grimmer M, Wagner RF Jr. Correlations among neural cell adhesion molecule, nerve growth factor, and its receptors, TrkA, TrkB, TrkC, and p75, in perineural invasion by basal cell and cutaneous squamous cell carcinomas. Dermatol Surg. 2004;30:1009-1016.
11. McCord M, Mendenhall WM, Parsons JT, et al. Skin cancer of the head and neck with clinical perineural invasion. Int J Radiat Oncol Biol Phys. 2000;47:89-93.
12. Ampil FL, Hardin JC, Peskind SP, et al. Perineural invasion in skin cancer of the head and neck: a review of nine cases. J Oral Maxillofac Surg. 1995;53:34-38.
13. Garcia-Serra A, Hinerman RW, Mendenhall WM, et al. Carcinoma of the skin with perineural invasion. Head Neck. 2003;25:1027-1033.
14. Goepfert H, Dichtel WJ, Medina JE, et al. Perineural invasion in squamous cell skin carcinoma of the head and neck. Am J Surg. 1984;148:542-547.
15. Galloway TJ, Morris CG, Mancuso AA, et al. Impact of radiographic findings on prognosis for skin carcinoma with clinical perineural invasion. Cancer. 2005;103:1254-1257.
16. Mendenhall WM, Amdur RJ, Williams LS, et al. Carcinoma of the skin of the head and neck with perineural invasion. Head Neck. 2002;24:78-83.
17. Williams LS, Mancuso AA, Mendenhall WM. Perineural spread of cutaneous squamous and basal cell carcinoma: CT and MR detection and its impact on patient management and prognosis. Int J Radiat Oncol Biol Phys. 2001;49:1061-1069.
18. Veness MJ, Biankin S. Perineural spread leading to orbital invasion from skin cancer. Australasian Radiol. 2000;44:296-302.
19. Feasel AM, Brown TJ, Bogle MA, et al. Perineural invasion of cutaneous malignancies. Dermatol Surg. 2001;27:531-542.
20. Soo K, Carter RL, O’Brien CJ, et al. Prognostic implications of perineural spread in squamous carcinomas of the head and neck. Laryngoscope. 1986;96:1145-1148.
21. Jambusaria-Pahlajani A, Miller CJ, Quon H, et al. Surgical monotherapy versus surgery plus adjuvant radiotherapy in high-risk cutaneous squamous cell carcinoma: a systematic review of outcomes. Dermatol Surg. 2009;35:574-584.
22. McCord MW, Mendenhall WM, Parsons JT, et al. Skin cancer of the head and neck with incidental microscopic perineural invasion. Int J Radiat Oncol Biol Phys. 1999;43:591-595.
23. Ross AS, Whalen FM, Elenitsas R, et al. Diameter of involved nerves predicts outcomes in cutaneous squamous cell carcinoma with perineural invasion: an investigator-blinded retrospective cohort study. Dermatol Surg. 2009;35:1859-1866.
24. Jambusaria-Pahlajani A, Kanetsky PA, Karia PS, et al. Evaluation of AJCC tumor staging for cutaneous squamous cell carcinoma and a proposed alternative tumor staging system. JAMA Dermatol. 2013;149:402-410.
25. Rowe DE, Carroll RJ, Day CL. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
1. Cruveilhier J. Maladies des nerfs. In: Cruveilhier J, ed. Anatomie Pathologique du Corps Humain. 2nd ed. Paris, France: JB Bailliere; 1835:1-3.
2. Neumann E. Secondare cancroid infiltration des nervus mentalis bei einem fall von lippincroid. Arch Pathol Anat. 1862;24:201-205.
3. Salasche S. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
4. Cottel WI. Perineural invasion by squamous-cell carcinoma. J Dermatol Surg Oncol. 1982;8:589-600.
5. Cooper PH, Mills SE, Leonard DD, et al. Sclerosing sweat duct (syringomatous) carcinoma. Am J Surg Pathol. 1985;9:422-433.
6. Ross AS, Whalen FM, Elenitsas R, et al. Diameter of involved nerves predicts outcomes in cutaneous squamous cell carcinoma with perineural invasion: an investigator-blinded retrospective cohort study. Dermatol Surg. 2009;35:1859-1866.
7. Leibovitch I, Huilgol SC, Selva D, et al. Cutaneous squamous cell carcinoma treated with Mohs micrographic surgery in Australia II. perineural invasion. J Am Acad Dermatol. 2005;53:261-266.
8. Carter RL, Foster CS, Dinsdale EA, et al. Perineural spread by squamous carcinomas of the head and neck: a morphological study using antiaxonal and antimyelin monoclonal antibodies. J Clin Pathol. 1983;36:269-275.
9. Vural E, Hutcheson J, Korourian S, et al. Correlation of neural cell adhesion molecules with perineural spread of squamous cell carcinoma of the head and neck. Otolaryngol Head Neck Surg. 2000;122:717-720.
10. Chen-Tsai CP, Colome-Grimmer M, Wagner RF Jr. Correlations among neural cell adhesion molecule, nerve growth factor, and its receptors, TrkA, TrkB, TrkC, and p75, in perineural invasion by basal cell and cutaneous squamous cell carcinomas. Dermatol Surg. 2004;30:1009-1016.
11. McCord M, Mendenhall WM, Parsons JT, et al. Skin cancer of the head and neck with clinical perineural invasion. Int J Radiat Oncol Biol Phys. 2000;47:89-93.
12. Ampil FL, Hardin JC, Peskind SP, et al. Perineural invasion in skin cancer of the head and neck: a review of nine cases. J Oral Maxillofac Surg. 1995;53:34-38.
13. Garcia-Serra A, Hinerman RW, Mendenhall WM, et al. Carcinoma of the skin with perineural invasion. Head Neck. 2003;25:1027-1033.
14. Goepfert H, Dichtel WJ, Medina JE, et al. Perineural invasion in squamous cell skin carcinoma of the head and neck. Am J Surg. 1984;148:542-547.
15. Galloway TJ, Morris CG, Mancuso AA, et al. Impact of radiographic findings on prognosis for skin carcinoma with clinical perineural invasion. Cancer. 2005;103:1254-1257.
16. Mendenhall WM, Amdur RJ, Williams LS, et al. Carcinoma of the skin of the head and neck with perineural invasion. Head Neck. 2002;24:78-83.
17. Williams LS, Mancuso AA, Mendenhall WM. Perineural spread of cutaneous squamous and basal cell carcinoma: CT and MR detection and its impact on patient management and prognosis. Int J Radiat Oncol Biol Phys. 2001;49:1061-1069.
18. Veness MJ, Biankin S. Perineural spread leading to orbital invasion from skin cancer. Australasian Radiol. 2000;44:296-302.
19. Feasel AM, Brown TJ, Bogle MA, et al. Perineural invasion of cutaneous malignancies. Dermatol Surg. 2001;27:531-542.
20. Soo K, Carter RL, O’Brien CJ, et al. Prognostic implications of perineural spread in squamous carcinomas of the head and neck. Laryngoscope. 1986;96:1145-1148.
21. Jambusaria-Pahlajani A, Miller CJ, Quon H, et al. Surgical monotherapy versus surgery plus adjuvant radiotherapy in high-risk cutaneous squamous cell carcinoma: a systematic review of outcomes. Dermatol Surg. 2009;35:574-584.
22. McCord MW, Mendenhall WM, Parsons JT, et al. Skin cancer of the head and neck with incidental microscopic perineural invasion. Int J Radiat Oncol Biol Phys. 1999;43:591-595.
23. Ross AS, Whalen FM, Elenitsas R, et al. Diameter of involved nerves predicts outcomes in cutaneous squamous cell carcinoma with perineural invasion: an investigator-blinded retrospective cohort study. Dermatol Surg. 2009;35:1859-1866.
24. Jambusaria-Pahlajani A, Kanetsky PA, Karia PS, et al. Evaluation of AJCC tumor staging for cutaneous squamous cell carcinoma and a proposed alternative tumor staging system. JAMA Dermatol. 2013;149:402-410.
25. Rowe DE, Carroll RJ, Day CL. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J Am Acad Dermatol. 1992;26:976-990.
Practice Points
• Patients with suspected cutaneous squamous cell carcinoma should be asked about neurological symptoms including pain, loss of motor skills, anesthesia, dysesthesia, and/or paresthesia, which may indicate perineural invasion.
• Patients with perineural invasion carry a much higher risk for local and distant recurrence and may require more aggressive treatment including Mohs micrographic surgery and adjuvant radiation.
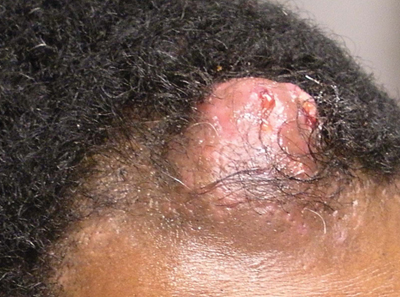
Solitary Nodular Lesion on the Scalp
The Diagnosis: Pilomatricoma
Pilomatricoma, first described by Malherbe and Chenantais1 in 1880, is a benign appendageal tumor derived from hair follicle matrix cells. It classically manifests as a solitary, asymptomatic, firm dermal nodule with a normal overlying epidermis. Less common morphologic variants include perforating, lymphagiectatic, keratoacanthomalike, pigmented, and anetodermalike surface changes.2 Inflammation and erosion through the skin surface are observed in the rare perforating variant, as seen in our patient. The average size is 1 cm, and it rarely exceeds 3 cm in diameter.3 The tumors predominantly occur on the head, neck, and upper extremities, with only 9.5% on the scalp.2 It may occur at any age, though it has a bimodal distribution with peaks in childhood and in adults older than 60 years. A slight preponderance in females has been observed with a female to male ratio of 1.5 to 1.2 Although our patient is black, most reported cases have occurred in individuals of European descent. Because cases of pilomatricoma are not systematically reported, it is uncertain if this finding represents a publication bias or if race is an actual risk factor. Multiple pilomatricomas and familial cases have been described in association with myotonic dystrophy, Turner syndrome, Gardner syndrome, Rubinstein-Taybi syndrome, polyfactorial coagulopathy, trisomy 9, xeroderma pigmentosum, and basal cell nevus syndrome.2,4
It has been shown that the proliferating cells of pilomatricomas stain with antibodies directed against Lef1 (lymphoid enhancer binding factor 1), a marker from hair matrix cells, providing biochemical evidence for the morphologic appearance of these neoplasms.5 Pilomatricomas have been associated with B-cell/chronic lymphocytic leukemia lymphoma 2 gene, BCL2, expression, a proto-oncogene that suppresses apoptosis in benign and malignant neoplasms, which may contribute to the pathogenesis of these tumors.6 Pilomatricomas also have been associated with β-catenin mutation, expression of Bmp2 (bone morphogenetic protein 2), and human hair keratin basic 1.7-9
Definitive diagnosis is obtained through biopsy, looking for characteristic histopathologic findings. The lesion usually is found in the lower dermis and subcutaneous fat. However, in the perforating variant, the lesion is more superficial, located in the papillary and mid dermis, as seen in our patient.10
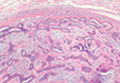
Pilomatricomas are sharply demarcated, often surrounded by a connective-tissue capsule. Histopathologic analysis reveals islands of epithelial cells comprised of 3 subtypes: basophilic cells with scant cytoplasm, shadow cells with a central pallor (Figure), and transitional cells between the former 2 cellular types.11 The number of basophilic and transitional cells is inversely related to the number of shadow cells. In older lesions, the shadow cells predominate, while the basophilic cells are few in number or absent. Calcium deposits are seen in 80% of lesions with von Kossa staining.12
Transformation into malignancy, known as pilomatrical carcinoma, is rare. These malignant neoplasms are characterized by aggressive biologic behavior such as recurrence, diffuse spread, or metastasis, or by cytologic abnormalities such as poor cellular organization, squamous differentiation, and conspicuous mitotic activity.13 The recent growth of the long-standing lesion in our patient might be interpreted as a sign of malignant transformation. However, this observation may be related to the intense inflammatory reaction supported by the histopathology.
Pilomatricomas are not associated with mortality. Pilomatrical carcinomas are uncommon but are locally invasive and can cause visceral metastases and death. Spontaneous regression has never been observed and medical treatment is ineffective. The treatment of choice is incision and curettage or surgical excision.14 Although recurrence has only been reported in 2.6% of cases from a large case series (N=228), patients should be monitored after surgical excision.12
1. Malherbe A, Chenantais J. Note sur l'epithelioma calcifie des glandes sebacees. Prog Med. 1880;8:826-828.
2. Julian CG, Bowers PW. A clinical review of 209 pilomatricomas. J Am Acad Dermatol. 1998;39(2, pt 1):191-195.
3. Lozzi GP, Soyer HP, Fruehauf J, et al. Giant pilomatricoma. Am J Dermatopathol. 2007;29:286-289.
4. Hubbard VG, Whittaker SJ. Multiple familial pilomatricomas: an unusual case. J Cutan Pathol. 2004;31:281-283.
5. Kizawa K, Toyoda M, Ito M, et al. Aberrantly differentiated cells in benign pilomatrixoma reflect the normal hair follicle: immunohistochemical analysis of Ca-binding S100A2, S100A3 and S100A6 proteins. Br J Dermatol. 2005;152:314-320.
6. Farrier S, Morgan M. bcl-2 expression in pilomatricoma. Am J Dermatopathol. 1997;19:254-257.
7. Park SW, Suh KS, Wang HY, et al. Beta-catenin expression in the transitional cell zone of pilomatricoma. Br J Dermatol. 2001;145:624-629.
8. Kurokawa I, Kusumoto K, Bessho K, et al. Immunohistochemical expression of bone morphogenetic protein-2 in pilomatricoma. Br J Dermatol. 2000;143:754-758.
9. Cribier B, Asch PH, Regnier C, et al. Expression of human hair keratin basic 1 in pilomatrixoma: a study of 128 cases. Br J Dermatol. 1999;140:600-604.
10. Bayle P, Bazex J, Lamant L, et al. Multiple perforating and non perforating pilomatricomas in a patient with Churg-Strauss syndrome and Rubinstein-Taybi syndrome. J Eur Acad Dermatol Venereol. 2004;18:607-610.
11. Elder D, Elenitsas R, Ragsdale BD. Pilomatricoma. In: Elder D, Elenitsas R, Jaworsky C, et al, eds. Histopathology of the Skin. 8th ed. Philadelphia, PA: Lippincott-Raven; 1997:757-759.
12. Forbis R Jr, Helwig EB. Pilomatrixoma (calcifying epithelioma). Arch Dermatol. 1961;83:606-618.
13. Wood MG, Parhizgar B, Beerman H. Malignant pilomatricoma. Arch Dermatol. 1984;120:770-773.
14. Thomas RW, Perkins JA, Ruegemer JL, et al. Surgical excision of pilomatrixoma of the head and neck: a retrospective review of 26 cases. Ear Nose Throat J. 1999;78:541, 544-546, 548.
The Diagnosis: Pilomatricoma
Pilomatricoma, first described by Malherbe and Chenantais1 in 1880, is a benign appendageal tumor derived from hair follicle matrix cells. It classically manifests as a solitary, asymptomatic, firm dermal nodule with a normal overlying epidermis. Less common morphologic variants include perforating, lymphagiectatic, keratoacanthomalike, pigmented, and anetodermalike surface changes.2 Inflammation and erosion through the skin surface are observed in the rare perforating variant, as seen in our patient. The average size is 1 cm, and it rarely exceeds 3 cm in diameter.3 The tumors predominantly occur on the head, neck, and upper extremities, with only 9.5% on the scalp.2 It may occur at any age, though it has a bimodal distribution with peaks in childhood and in adults older than 60 years. A slight preponderance in females has been observed with a female to male ratio of 1.5 to 1.2 Although our patient is black, most reported cases have occurred in individuals of European descent. Because cases of pilomatricoma are not systematically reported, it is uncertain if this finding represents a publication bias or if race is an actual risk factor. Multiple pilomatricomas and familial cases have been described in association with myotonic dystrophy, Turner syndrome, Gardner syndrome, Rubinstein-Taybi syndrome, polyfactorial coagulopathy, trisomy 9, xeroderma pigmentosum, and basal cell nevus syndrome.2,4
It has been shown that the proliferating cells of pilomatricomas stain with antibodies directed against Lef1 (lymphoid enhancer binding factor 1), a marker from hair matrix cells, providing biochemical evidence for the morphologic appearance of these neoplasms.5 Pilomatricomas have been associated with B-cell/chronic lymphocytic leukemia lymphoma 2 gene, BCL2, expression, a proto-oncogene that suppresses apoptosis in benign and malignant neoplasms, which may contribute to the pathogenesis of these tumors.6 Pilomatricomas also have been associated with β-catenin mutation, expression of Bmp2 (bone morphogenetic protein 2), and human hair keratin basic 1.7-9
Definitive diagnosis is obtained through biopsy, looking for characteristic histopathologic findings. The lesion usually is found in the lower dermis and subcutaneous fat. However, in the perforating variant, the lesion is more superficial, located in the papillary and mid dermis, as seen in our patient.10
Pilomatricomas are sharply demarcated, often surrounded by a connective-tissue capsule. Histopathologic analysis reveals islands of epithelial cells comprised of 3 subtypes: basophilic cells with scant cytoplasm, shadow cells with a central pallor (Figure), and transitional cells between the former 2 cellular types.11 The number of basophilic and transitional cells is inversely related to the number of shadow cells. In older lesions, the shadow cells predominate, while the basophilic cells are few in number or absent. Calcium deposits are seen in 80% of lesions with von Kossa staining.12
Transformation into malignancy, known as pilomatrical carcinoma, is rare. These malignant neoplasms are characterized by aggressive biologic behavior such as recurrence, diffuse spread, or metastasis, or by cytologic abnormalities such as poor cellular organization, squamous differentiation, and conspicuous mitotic activity.13 The recent growth of the long-standing lesion in our patient might be interpreted as a sign of malignant transformation. However, this observation may be related to the intense inflammatory reaction supported by the histopathology.
Pilomatricomas are not associated with mortality. Pilomatrical carcinomas are uncommon but are locally invasive and can cause visceral metastases and death. Spontaneous regression has never been observed and medical treatment is ineffective. The treatment of choice is incision and curettage or surgical excision.14 Although recurrence has only been reported in 2.6% of cases from a large case series (N=228), patients should be monitored after surgical excision.12
The Diagnosis: Pilomatricoma
Pilomatricoma, first described by Malherbe and Chenantais1 in 1880, is a benign appendageal tumor derived from hair follicle matrix cells. It classically manifests as a solitary, asymptomatic, firm dermal nodule with a normal overlying epidermis. Less common morphologic variants include perforating, lymphagiectatic, keratoacanthomalike, pigmented, and anetodermalike surface changes.2 Inflammation and erosion through the skin surface are observed in the rare perforating variant, as seen in our patient. The average size is 1 cm, and it rarely exceeds 3 cm in diameter.3 The tumors predominantly occur on the head, neck, and upper extremities, with only 9.5% on the scalp.2 It may occur at any age, though it has a bimodal distribution with peaks in childhood and in adults older than 60 years. A slight preponderance in females has been observed with a female to male ratio of 1.5 to 1.2 Although our patient is black, most reported cases have occurred in individuals of European descent. Because cases of pilomatricoma are not systematically reported, it is uncertain if this finding represents a publication bias or if race is an actual risk factor. Multiple pilomatricomas and familial cases have been described in association with myotonic dystrophy, Turner syndrome, Gardner syndrome, Rubinstein-Taybi syndrome, polyfactorial coagulopathy, trisomy 9, xeroderma pigmentosum, and basal cell nevus syndrome.2,4
It has been shown that the proliferating cells of pilomatricomas stain with antibodies directed against Lef1 (lymphoid enhancer binding factor 1), a marker from hair matrix cells, providing biochemical evidence for the morphologic appearance of these neoplasms.5 Pilomatricomas have been associated with B-cell/chronic lymphocytic leukemia lymphoma 2 gene, BCL2, expression, a proto-oncogene that suppresses apoptosis in benign and malignant neoplasms, which may contribute to the pathogenesis of these tumors.6 Pilomatricomas also have been associated with β-catenin mutation, expression of Bmp2 (bone morphogenetic protein 2), and human hair keratin basic 1.7-9
Definitive diagnosis is obtained through biopsy, looking for characteristic histopathologic findings. The lesion usually is found in the lower dermis and subcutaneous fat. However, in the perforating variant, the lesion is more superficial, located in the papillary and mid dermis, as seen in our patient.10
Pilomatricomas are sharply demarcated, often surrounded by a connective-tissue capsule. Histopathologic analysis reveals islands of epithelial cells comprised of 3 subtypes: basophilic cells with scant cytoplasm, shadow cells with a central pallor (Figure), and transitional cells between the former 2 cellular types.11 The number of basophilic and transitional cells is inversely related to the number of shadow cells. In older lesions, the shadow cells predominate, while the basophilic cells are few in number or absent. Calcium deposits are seen in 80% of lesions with von Kossa staining.12
Transformation into malignancy, known as pilomatrical carcinoma, is rare. These malignant neoplasms are characterized by aggressive biologic behavior such as recurrence, diffuse spread, or metastasis, or by cytologic abnormalities such as poor cellular organization, squamous differentiation, and conspicuous mitotic activity.13 The recent growth of the long-standing lesion in our patient might be interpreted as a sign of malignant transformation. However, this observation may be related to the intense inflammatory reaction supported by the histopathology.
Pilomatricomas are not associated with mortality. Pilomatrical carcinomas are uncommon but are locally invasive and can cause visceral metastases and death. Spontaneous regression has never been observed and medical treatment is ineffective. The treatment of choice is incision and curettage or surgical excision.14 Although recurrence has only been reported in 2.6% of cases from a large case series (N=228), patients should be monitored after surgical excision.12
1. Malherbe A, Chenantais J. Note sur l'epithelioma calcifie des glandes sebacees. Prog Med. 1880;8:826-828.
2. Julian CG, Bowers PW. A clinical review of 209 pilomatricomas. J Am Acad Dermatol. 1998;39(2, pt 1):191-195.
3. Lozzi GP, Soyer HP, Fruehauf J, et al. Giant pilomatricoma. Am J Dermatopathol. 2007;29:286-289.
4. Hubbard VG, Whittaker SJ. Multiple familial pilomatricomas: an unusual case. J Cutan Pathol. 2004;31:281-283.
5. Kizawa K, Toyoda M, Ito M, et al. Aberrantly differentiated cells in benign pilomatrixoma reflect the normal hair follicle: immunohistochemical analysis of Ca-binding S100A2, S100A3 and S100A6 proteins. Br J Dermatol. 2005;152:314-320.
6. Farrier S, Morgan M. bcl-2 expression in pilomatricoma. Am J Dermatopathol. 1997;19:254-257.
7. Park SW, Suh KS, Wang HY, et al. Beta-catenin expression in the transitional cell zone of pilomatricoma. Br J Dermatol. 2001;145:624-629.
8. Kurokawa I, Kusumoto K, Bessho K, et al. Immunohistochemical expression of bone morphogenetic protein-2 in pilomatricoma. Br J Dermatol. 2000;143:754-758.
9. Cribier B, Asch PH, Regnier C, et al. Expression of human hair keratin basic 1 in pilomatrixoma: a study of 128 cases. Br J Dermatol. 1999;140:600-604.
10. Bayle P, Bazex J, Lamant L, et al. Multiple perforating and non perforating pilomatricomas in a patient with Churg-Strauss syndrome and Rubinstein-Taybi syndrome. J Eur Acad Dermatol Venereol. 2004;18:607-610.
11. Elder D, Elenitsas R, Ragsdale BD. Pilomatricoma. In: Elder D, Elenitsas R, Jaworsky C, et al, eds. Histopathology of the Skin. 8th ed. Philadelphia, PA: Lippincott-Raven; 1997:757-759.
12. Forbis R Jr, Helwig EB. Pilomatrixoma (calcifying epithelioma). Arch Dermatol. 1961;83:606-618.
13. Wood MG, Parhizgar B, Beerman H. Malignant pilomatricoma. Arch Dermatol. 1984;120:770-773.
14. Thomas RW, Perkins JA, Ruegemer JL, et al. Surgical excision of pilomatrixoma of the head and neck: a retrospective review of 26 cases. Ear Nose Throat J. 1999;78:541, 544-546, 548.
1. Malherbe A, Chenantais J. Note sur l'epithelioma calcifie des glandes sebacees. Prog Med. 1880;8:826-828.
2. Julian CG, Bowers PW. A clinical review of 209 pilomatricomas. J Am Acad Dermatol. 1998;39(2, pt 1):191-195.
3. Lozzi GP, Soyer HP, Fruehauf J, et al. Giant pilomatricoma. Am J Dermatopathol. 2007;29:286-289.
4. Hubbard VG, Whittaker SJ. Multiple familial pilomatricomas: an unusual case. J Cutan Pathol. 2004;31:281-283.
5. Kizawa K, Toyoda M, Ito M, et al. Aberrantly differentiated cells in benign pilomatrixoma reflect the normal hair follicle: immunohistochemical analysis of Ca-binding S100A2, S100A3 and S100A6 proteins. Br J Dermatol. 2005;152:314-320.
6. Farrier S, Morgan M. bcl-2 expression in pilomatricoma. Am J Dermatopathol. 1997;19:254-257.
7. Park SW, Suh KS, Wang HY, et al. Beta-catenin expression in the transitional cell zone of pilomatricoma. Br J Dermatol. 2001;145:624-629.
8. Kurokawa I, Kusumoto K, Bessho K, et al. Immunohistochemical expression of bone morphogenetic protein-2 in pilomatricoma. Br J Dermatol. 2000;143:754-758.
9. Cribier B, Asch PH, Regnier C, et al. Expression of human hair keratin basic 1 in pilomatrixoma: a study of 128 cases. Br J Dermatol. 1999;140:600-604.
10. Bayle P, Bazex J, Lamant L, et al. Multiple perforating and non perforating pilomatricomas in a patient with Churg-Strauss syndrome and Rubinstein-Taybi syndrome. J Eur Acad Dermatol Venereol. 2004;18:607-610.
11. Elder D, Elenitsas R, Ragsdale BD. Pilomatricoma. In: Elder D, Elenitsas R, Jaworsky C, et al, eds. Histopathology of the Skin. 8th ed. Philadelphia, PA: Lippincott-Raven; 1997:757-759.
12. Forbis R Jr, Helwig EB. Pilomatrixoma (calcifying epithelioma). Arch Dermatol. 1961;83:606-618.
13. Wood MG, Parhizgar B, Beerman H. Malignant pilomatricoma. Arch Dermatol. 1984;120:770-773.
14. Thomas RW, Perkins JA, Ruegemer JL, et al. Surgical excision of pilomatrixoma of the head and neck: a retrospective review of 26 cases. Ear Nose Throat J. 1999;78:541, 544-546, 548.
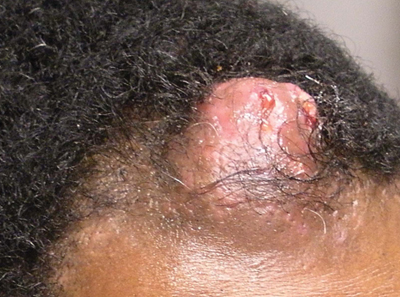
An otherwise healthy 40-year-old man presented for examination of a solitary nodular lesion on the frontal aspect of the scalp of 1 year’s duration. The lesion had rapidly increased in size in the 2 weeks prior to presentation. He presented to the emergency department after he noted pain and drainage from the lesion. Biopsy of the lesion revealed islands of pale eosinophilic shadow cells with an intense dermal infiltrate consisting of lymphocytes, histiocytes, plasma cells, and neutrophils.
Double-Positive CD4+CD8+ Sézary Syndrome: An Unusual Phenotype With an Aggressive Clinical Course
Aggressive secondary squamous carcinoma appeared during BRAF inhibitor targeted therapy
A woman undergoing BRAF inhibitor targeted therapy for advanced melanoma has presented with invasive spindle cell squamous carcinoma masquerading as a secondary cutaneous squamous cell carcinoma, highlighting the importance of histologic evaluation of these lesions.
"Secondary cutaneous squamous cell carcinomas (cSCCs) are adverse effects of BRAF inhibitor targeted therapy for advanced melanoma," wrote Dr. Daniel N. Cohen and his associates online Feb. 26 in JAMA Dermatology.
The most commonly seen histologic type of secondary cutaneous squamous cell carcinomas is keratoacanthoma-like cSCC (cSCC-KA), which is thought to have a low risk of metastasis or recurrence, said lead author Dr. Cohen of the Vanderbilt University Medical Center, Nashville, Tenn.
In this case report, however, a woman in her 50s with BRAF-mutant metastatic melanoma developed more than 100 new cutaneous squamous proliferations across her face, trunk, and extremities within 4 weeks of starting treatment with the BRAF inhibitor dabrafenib as part of a clinical trial, with some lesions appearing to be a more aggressive type upon analysis.
The lesions began as acrochordons on her face and extremities, as well as new nevi on her torso and axilla. She also developed fever, chills, and fatigue and had enlarging, tender, and bleeding lesions on her trunk and extremities.
Seven large, tender, and indurated lesions were removed using a deep scoop shave biopsy, revealing a biphasic malignant growth pattern (JAMA Dermatology 2014 Feb 26 [doi:10.1001/jamadermatol.2013.7784]).
"The superficial portion demonstrated conventional cSCC-KA features of hyperkeratosis, epidermal acanthosis, and central core of glassy eosinophilic keratin with pseudopapillomatosis and a base with focal invasive lobules of cytologically atypical keratinocytes, consistent with previously reported cSCC-KA" the study authors reported.
However, they added, "in stark contrast to prior reports, the deep aspects of 6 of 7 lesions showed invasive spindled and epithelioid cells with monomorphic elongated nuclei with condensed chromatin and mitoses consistent with spindle cell squamous carcinoma, an aggressive subtype of squamous cell carcinoma."
The cells were strongly immunoreactive for cytokeratin CK5/6 and CK903, and both the squamous and spindle components were vimentin reactive and showed increased proliferation index. In contrast to the usual pattern of a spindle cell melanoma, the spindle tumor cells were also MART-1 and S100 negative.
The authors said this was the first known report of invasive spindle cell squamous carcinoma that mimicked keratoacanthoma-like secondary SCC appearing during BRAF inhibitor therapy and suggested that the discovery has implications for management of secondary squamous cell carcinoma.
"Because the clinical appearance of cSCC-KA and the spindle cell squamous carcinomas in our patient are indistinguishable, histologic evaluation of the entire lesion (via saucerization biopsy or incisional biopsy) is vital to prevent inadequate treatment of a deeply invasive process with a probable higher malignant potential," Dr. Cohen and his associates wrote.
The patient stopped BRAF inhibitor therapy and had no recurrence or new development of cutaneous secondary SCCs. A single later recurrence of melanoma was resected but she remains disease free.
One author declared a consultancy with Bristol-Myers Squibb and Genentech, as well as grant support from Genentech. There were no other financial disclosures reported.
A woman undergoing BRAF inhibitor targeted therapy for advanced melanoma has presented with invasive spindle cell squamous carcinoma masquerading as a secondary cutaneous squamous cell carcinoma, highlighting the importance of histologic evaluation of these lesions.
"Secondary cutaneous squamous cell carcinomas (cSCCs) are adverse effects of BRAF inhibitor targeted therapy for advanced melanoma," wrote Dr. Daniel N. Cohen and his associates online Feb. 26 in JAMA Dermatology.
The most commonly seen histologic type of secondary cutaneous squamous cell carcinomas is keratoacanthoma-like cSCC (cSCC-KA), which is thought to have a low risk of metastasis or recurrence, said lead author Dr. Cohen of the Vanderbilt University Medical Center, Nashville, Tenn.
In this case report, however, a woman in her 50s with BRAF-mutant metastatic melanoma developed more than 100 new cutaneous squamous proliferations across her face, trunk, and extremities within 4 weeks of starting treatment with the BRAF inhibitor dabrafenib as part of a clinical trial, with some lesions appearing to be a more aggressive type upon analysis.
The lesions began as acrochordons on her face and extremities, as well as new nevi on her torso and axilla. She also developed fever, chills, and fatigue and had enlarging, tender, and bleeding lesions on her trunk and extremities.
Seven large, tender, and indurated lesions were removed using a deep scoop shave biopsy, revealing a biphasic malignant growth pattern (JAMA Dermatology 2014 Feb 26 [doi:10.1001/jamadermatol.2013.7784]).
"The superficial portion demonstrated conventional cSCC-KA features of hyperkeratosis, epidermal acanthosis, and central core of glassy eosinophilic keratin with pseudopapillomatosis and a base with focal invasive lobules of cytologically atypical keratinocytes, consistent with previously reported cSCC-KA" the study authors reported.
However, they added, "in stark contrast to prior reports, the deep aspects of 6 of 7 lesions showed invasive spindled and epithelioid cells with monomorphic elongated nuclei with condensed chromatin and mitoses consistent with spindle cell squamous carcinoma, an aggressive subtype of squamous cell carcinoma."
The cells were strongly immunoreactive for cytokeratin CK5/6 and CK903, and both the squamous and spindle components were vimentin reactive and showed increased proliferation index. In contrast to the usual pattern of a spindle cell melanoma, the spindle tumor cells were also MART-1 and S100 negative.
The authors said this was the first known report of invasive spindle cell squamous carcinoma that mimicked keratoacanthoma-like secondary SCC appearing during BRAF inhibitor therapy and suggested that the discovery has implications for management of secondary squamous cell carcinoma.
"Because the clinical appearance of cSCC-KA and the spindle cell squamous carcinomas in our patient are indistinguishable, histologic evaluation of the entire lesion (via saucerization biopsy or incisional biopsy) is vital to prevent inadequate treatment of a deeply invasive process with a probable higher malignant potential," Dr. Cohen and his associates wrote.
The patient stopped BRAF inhibitor therapy and had no recurrence or new development of cutaneous secondary SCCs. A single later recurrence of melanoma was resected but she remains disease free.
One author declared a consultancy with Bristol-Myers Squibb and Genentech, as well as grant support from Genentech. There were no other financial disclosures reported.
A woman undergoing BRAF inhibitor targeted therapy for advanced melanoma has presented with invasive spindle cell squamous carcinoma masquerading as a secondary cutaneous squamous cell carcinoma, highlighting the importance of histologic evaluation of these lesions.
"Secondary cutaneous squamous cell carcinomas (cSCCs) are adverse effects of BRAF inhibitor targeted therapy for advanced melanoma," wrote Dr. Daniel N. Cohen and his associates online Feb. 26 in JAMA Dermatology.
The most commonly seen histologic type of secondary cutaneous squamous cell carcinomas is keratoacanthoma-like cSCC (cSCC-KA), which is thought to have a low risk of metastasis or recurrence, said lead author Dr. Cohen of the Vanderbilt University Medical Center, Nashville, Tenn.
In this case report, however, a woman in her 50s with BRAF-mutant metastatic melanoma developed more than 100 new cutaneous squamous proliferations across her face, trunk, and extremities within 4 weeks of starting treatment with the BRAF inhibitor dabrafenib as part of a clinical trial, with some lesions appearing to be a more aggressive type upon analysis.
The lesions began as acrochordons on her face and extremities, as well as new nevi on her torso and axilla. She also developed fever, chills, and fatigue and had enlarging, tender, and bleeding lesions on her trunk and extremities.
Seven large, tender, and indurated lesions were removed using a deep scoop shave biopsy, revealing a biphasic malignant growth pattern (JAMA Dermatology 2014 Feb 26 [doi:10.1001/jamadermatol.2013.7784]).
"The superficial portion demonstrated conventional cSCC-KA features of hyperkeratosis, epidermal acanthosis, and central core of glassy eosinophilic keratin with pseudopapillomatosis and a base with focal invasive lobules of cytologically atypical keratinocytes, consistent with previously reported cSCC-KA" the study authors reported.
However, they added, "in stark contrast to prior reports, the deep aspects of 6 of 7 lesions showed invasive spindled and epithelioid cells with monomorphic elongated nuclei with condensed chromatin and mitoses consistent with spindle cell squamous carcinoma, an aggressive subtype of squamous cell carcinoma."
The cells were strongly immunoreactive for cytokeratin CK5/6 and CK903, and both the squamous and spindle components were vimentin reactive and showed increased proliferation index. In contrast to the usual pattern of a spindle cell melanoma, the spindle tumor cells were also MART-1 and S100 negative.
The authors said this was the first known report of invasive spindle cell squamous carcinoma that mimicked keratoacanthoma-like secondary SCC appearing during BRAF inhibitor therapy and suggested that the discovery has implications for management of secondary squamous cell carcinoma.
"Because the clinical appearance of cSCC-KA and the spindle cell squamous carcinomas in our patient are indistinguishable, histologic evaluation of the entire lesion (via saucerization biopsy or incisional biopsy) is vital to prevent inadequate treatment of a deeply invasive process with a probable higher malignant potential," Dr. Cohen and his associates wrote.
The patient stopped BRAF inhibitor therapy and had no recurrence or new development of cutaneous secondary SCCs. A single later recurrence of melanoma was resected but she remains disease free.
One author declared a consultancy with Bristol-Myers Squibb and Genentech, as well as grant support from Genentech. There were no other financial disclosures reported.
FROM JAMA DERMATOLOGY
Major finding: A woman undergoing BRAF inhibitor targeted therapy for advanced melanoma has presented with invasive spindle cell squamous carcinoma masquerading as a secondary cutaneous squamous cell carcinoma.
Data source: Case report.
Disclosures: One author declared a consultancy with Bristol-Myers Squibb and Genentech, as well as grant support from Genentech. There were no other financial disclosures reported.
Chondroid Syringoma
Immunosuppressive therapy ups risk for skin cancers
HOLLYWOOD, FLA. – Current and previous use of thiopurines, biologics, and combination therapies are all independent risk factors for skin cancer, according to expert analysis given at a conference on inflammatory bowel diseases.
Although population-based cohort studies have shown that the baseline risk for nonmelanoma skin cancer in IBD has risen more than a third since the preimmunomodulator era, regardless of the mode of treatment, "Cutaneous side effects of immunomodulators and biologics are a rising concern in clinical practice," said Dr. Jean-Frederic Colombel of the Icahn School of Medicine at Mount Sinai in New York. "Patients with Crohn’s disease in particular have shown a twofold increased risk for nonmelanoma skin cancer, outside of any kind of immunomodulator or biologic therapy."
Thiopurine use has been associated with a twofold increased risk of nonmelanoma skin cancer that persists even after withdrawal from the medication, although there is not an increased risk for melanoma, according to Dr. Colombel.
In a comparative analysis of studies published primarily since 2011, he noted an epidemiologic study with an A level of evidence for nearly 10,000 IBD patients undergoing thiopurine treatment that had an increased risk for NMSC with an odds ratio of approximately 2.2 (95% CI, 1.24-3.81).
"What is very important to note is that the risk of nonmelanoma skin cancer persists even after the antimetabolite has been stopped," said Dr. Colombel, referring to data from the CESAME study that indicated, regardless of age, past and current thiopurine use was associated with higher incidence rates of NMSC.
To date, other immunomodulators have not been associated with NMSC, said Dr. Colombel. "What we are observing is quite specific to azathioprine."
Thiopurines are not considered a risk factor for melanoma, said Dr. Colombel. He cited a study of records from a health care claims database for the period between 1997 and 2009. In the study, 209 melanoma cases were matched with 823 controls. Exposure to thiopurines was associated with an OR of 1.1 for melanoma. In that same study, patients exposed to biologics were found to have an increased risk of melanoma, although the risk was higher in patients with Crohn’s disease than those with ulcerative colitis (OR, 1.94 vs. 1.73).
Biologics may also increase the risk of NMSC, although currently the data are "controversial" said Dr. Colombel. "The results are more difficult to interpret."
The risk for NMSC in IBD patients exposed to biologics was evaluated in three studies published between 2010 and 2013. One study indicated no significant increased risk for either past or current biologic use (OR 1.14, 95% CI 0.95-1.36).
Another study indicated a twofold increased risk for patients who’d withdrawn from biologic therapy (OR 2.07, 95% CI 1.28-3.33) and who had persistent use (OR 2.18, 95% CI, 1.07-4.46).
A third study showed a 2.3 increased NMSC risk (95% CI, 1.44-3.47).
In a meta-analysis published in 2013, the pooled relative risk for melanoma in IBD patients exposed to biologics was not shown to be significant (RR, 1.10).
Combination therapy has been found to increase the risk of NMSC nearly fourfold, said Dr. Colombel.
Even though more data are needed, Dr. Colombel said that all patients who are scheduled to start immunosuppression should be informed of the potential for dermatologic complications. "Personally, I am now sending all my patients to a dermatologist for a baseline evaluation," he said.
The conference was sponsored by the Crohn’s & Colitis Foundation of America. Dr. Colombel reported many disclosures, including Abbott Laboratories, Bristol-Myers Squibb, Genentech, Inc., Pfizer Inc. and sanofi-aventis, among others.
HOLLYWOOD, FLA. – Current and previous use of thiopurines, biologics, and combination therapies are all independent risk factors for skin cancer, according to expert analysis given at a conference on inflammatory bowel diseases.
Although population-based cohort studies have shown that the baseline risk for nonmelanoma skin cancer in IBD has risen more than a third since the preimmunomodulator era, regardless of the mode of treatment, "Cutaneous side effects of immunomodulators and biologics are a rising concern in clinical practice," said Dr. Jean-Frederic Colombel of the Icahn School of Medicine at Mount Sinai in New York. "Patients with Crohn’s disease in particular have shown a twofold increased risk for nonmelanoma skin cancer, outside of any kind of immunomodulator or biologic therapy."
Thiopurine use has been associated with a twofold increased risk of nonmelanoma skin cancer that persists even after withdrawal from the medication, although there is not an increased risk for melanoma, according to Dr. Colombel.
In a comparative analysis of studies published primarily since 2011, he noted an epidemiologic study with an A level of evidence for nearly 10,000 IBD patients undergoing thiopurine treatment that had an increased risk for NMSC with an odds ratio of approximately 2.2 (95% CI, 1.24-3.81).
"What is very important to note is that the risk of nonmelanoma skin cancer persists even after the antimetabolite has been stopped," said Dr. Colombel, referring to data from the CESAME study that indicated, regardless of age, past and current thiopurine use was associated with higher incidence rates of NMSC.
To date, other immunomodulators have not been associated with NMSC, said Dr. Colombel. "What we are observing is quite specific to azathioprine."
Thiopurines are not considered a risk factor for melanoma, said Dr. Colombel. He cited a study of records from a health care claims database for the period between 1997 and 2009. In the study, 209 melanoma cases were matched with 823 controls. Exposure to thiopurines was associated with an OR of 1.1 for melanoma. In that same study, patients exposed to biologics were found to have an increased risk of melanoma, although the risk was higher in patients with Crohn’s disease than those with ulcerative colitis (OR, 1.94 vs. 1.73).
Biologics may also increase the risk of NMSC, although currently the data are "controversial" said Dr. Colombel. "The results are more difficult to interpret."
The risk for NMSC in IBD patients exposed to biologics was evaluated in three studies published between 2010 and 2013. One study indicated no significant increased risk for either past or current biologic use (OR 1.14, 95% CI 0.95-1.36).
Another study indicated a twofold increased risk for patients who’d withdrawn from biologic therapy (OR 2.07, 95% CI 1.28-3.33) and who had persistent use (OR 2.18, 95% CI, 1.07-4.46).
A third study showed a 2.3 increased NMSC risk (95% CI, 1.44-3.47).
In a meta-analysis published in 2013, the pooled relative risk for melanoma in IBD patients exposed to biologics was not shown to be significant (RR, 1.10).
Combination therapy has been found to increase the risk of NMSC nearly fourfold, said Dr. Colombel.
Even though more data are needed, Dr. Colombel said that all patients who are scheduled to start immunosuppression should be informed of the potential for dermatologic complications. "Personally, I am now sending all my patients to a dermatologist for a baseline evaluation," he said.
The conference was sponsored by the Crohn’s & Colitis Foundation of America. Dr. Colombel reported many disclosures, including Abbott Laboratories, Bristol-Myers Squibb, Genentech, Inc., Pfizer Inc. and sanofi-aventis, among others.
HOLLYWOOD, FLA. – Current and previous use of thiopurines, biologics, and combination therapies are all independent risk factors for skin cancer, according to expert analysis given at a conference on inflammatory bowel diseases.
Although population-based cohort studies have shown that the baseline risk for nonmelanoma skin cancer in IBD has risen more than a third since the preimmunomodulator era, regardless of the mode of treatment, "Cutaneous side effects of immunomodulators and biologics are a rising concern in clinical practice," said Dr. Jean-Frederic Colombel of the Icahn School of Medicine at Mount Sinai in New York. "Patients with Crohn’s disease in particular have shown a twofold increased risk for nonmelanoma skin cancer, outside of any kind of immunomodulator or biologic therapy."
Thiopurine use has been associated with a twofold increased risk of nonmelanoma skin cancer that persists even after withdrawal from the medication, although there is not an increased risk for melanoma, according to Dr. Colombel.
In a comparative analysis of studies published primarily since 2011, he noted an epidemiologic study with an A level of evidence for nearly 10,000 IBD patients undergoing thiopurine treatment that had an increased risk for NMSC with an odds ratio of approximately 2.2 (95% CI, 1.24-3.81).
"What is very important to note is that the risk of nonmelanoma skin cancer persists even after the antimetabolite has been stopped," said Dr. Colombel, referring to data from the CESAME study that indicated, regardless of age, past and current thiopurine use was associated with higher incidence rates of NMSC.
To date, other immunomodulators have not been associated with NMSC, said Dr. Colombel. "What we are observing is quite specific to azathioprine."
Thiopurines are not considered a risk factor for melanoma, said Dr. Colombel. He cited a study of records from a health care claims database for the period between 1997 and 2009. In the study, 209 melanoma cases were matched with 823 controls. Exposure to thiopurines was associated with an OR of 1.1 for melanoma. In that same study, patients exposed to biologics were found to have an increased risk of melanoma, although the risk was higher in patients with Crohn’s disease than those with ulcerative colitis (OR, 1.94 vs. 1.73).
Biologics may also increase the risk of NMSC, although currently the data are "controversial" said Dr. Colombel. "The results are more difficult to interpret."
The risk for NMSC in IBD patients exposed to biologics was evaluated in three studies published between 2010 and 2013. One study indicated no significant increased risk for either past or current biologic use (OR 1.14, 95% CI 0.95-1.36).
Another study indicated a twofold increased risk for patients who’d withdrawn from biologic therapy (OR 2.07, 95% CI 1.28-3.33) and who had persistent use (OR 2.18, 95% CI, 1.07-4.46).
A third study showed a 2.3 increased NMSC risk (95% CI, 1.44-3.47).
In a meta-analysis published in 2013, the pooled relative risk for melanoma in IBD patients exposed to biologics was not shown to be significant (RR, 1.10).
Combination therapy has been found to increase the risk of NMSC nearly fourfold, said Dr. Colombel.
Even though more data are needed, Dr. Colombel said that all patients who are scheduled to start immunosuppression should be informed of the potential for dermatologic complications. "Personally, I am now sending all my patients to a dermatologist for a baseline evaluation," he said.
The conference was sponsored by the Crohn’s & Colitis Foundation of America. Dr. Colombel reported many disclosures, including Abbott Laboratories, Bristol-Myers Squibb, Genentech, Inc., Pfizer Inc. and sanofi-aventis, among others.
EXPERT ANALYSIS FROM 2013 ADVANCES IN IBD
Multiple Familial Trichodiscomas
Growing Ears, Noses, and Skin: The New Frontier in Dermatology and Dermatologic Surgery?

In an article published online in Maclean’s magazine on October 15, 2013, the concept of developing artificial body parts is discussed. The technology now exists for scientists to grow tissue organs, such as ears, noses, and fingers. Several groups of investigators, including Anthony Atala, MD (Wake Forest Institute for Regenerative Medicine, Winston-Salem, North Carolina) and Alexander Seifalian, PhD (University College London, England), are busy making ears by creating a biodegradable scaffold onto which cells—either stem cells from the patient or cells that have been harvested from the patient’s original organ—are layered and permitted to multiply. The cell-layered scaffold is then placed in a bioreactor for a couple of weeks. Once ready, the new ear is transplanted onto the patient; subsequently, the scaffold melts away.
Dr. Atala’s laboratory also is busy growing fingers. Meanwhile, Dr. Seifalian grew a nose (inside the patient’s own arm) in only 3 months after the patient lost his nose to skin cancer. He made a mold based on the patient’s original nose, created a scaffold composed of nanocomposite material, added the patient’s stem cells to the scaffold, and put the nose in a bioreactor to mature. Meanwhile, he placed a tissue expander in the patient’s arm and subsequently inserted the nose into the arm so that it could become vascularized and covered by skin. The nose was transplanted to the patient’s face; additional surgery is planned to open the nostrils, followed by seeding them with stem cells to return his sense of smell.
Marc Jeschke, MD, PhD (Ross Tilley Burn Centre, Sunnybrook Health Sciences Centre, Toronto, Ontario) is developing a bioprinter to create skin. The bioprinter dispenses different types of cells (grown from the patient’s own cells) into a hydrogel matrix in 3 dimensions. Currently, Dr. Jeschke is working with mice; however, in a few years, he anticipates treating human patients. Other investigators, such as Michael C. McAlpine (Princeton University, New Jersey), recently used a commercial 3-dimensional printer to make an ear; the “inks” included calf cells and a silver nanoparticle paste that formed a coiled antenna inside the cartilage that was capable of receiving electromagnetic signals and transmitting them to the brain.
What’s the issue?
The ability to grow tissue organs is going to revolutionize the surgical management of patients who need solid organ replacement; kidneys, lungs, pancreases, spleens, and tracheas have already been successfully grown. Indeed several investigators are making ears and noses. How long will it be before flaps and grafts to repair wound defects following extensive and deforming skin cancer surgery are replaced by ears and noses that are grown from the patient’s own tissue cells or stem cells? Although it seems like science fiction today, how soon will it be before a cutaneous 3-dimensional printer becomes a standard piece of equipment in the dermatologist and dermatologic surgeon’s office for use to create skin to cover postoperative sites that cannot be closed by directly bringing the wound edges together?

In an article published online in Maclean’s magazine on October 15, 2013, the concept of developing artificial body parts is discussed. The technology now exists for scientists to grow tissue organs, such as ears, noses, and fingers. Several groups of investigators, including Anthony Atala, MD (Wake Forest Institute for Regenerative Medicine, Winston-Salem, North Carolina) and Alexander Seifalian, PhD (University College London, England), are busy making ears by creating a biodegradable scaffold onto which cells—either stem cells from the patient or cells that have been harvested from the patient’s original organ—are layered and permitted to multiply. The cell-layered scaffold is then placed in a bioreactor for a couple of weeks. Once ready, the new ear is transplanted onto the patient; subsequently, the scaffold melts away.
Dr. Atala’s laboratory also is busy growing fingers. Meanwhile, Dr. Seifalian grew a nose (inside the patient’s own arm) in only 3 months after the patient lost his nose to skin cancer. He made a mold based on the patient’s original nose, created a scaffold composed of nanocomposite material, added the patient’s stem cells to the scaffold, and put the nose in a bioreactor to mature. Meanwhile, he placed a tissue expander in the patient’s arm and subsequently inserted the nose into the arm so that it could become vascularized and covered by skin. The nose was transplanted to the patient’s face; additional surgery is planned to open the nostrils, followed by seeding them with stem cells to return his sense of smell.
Marc Jeschke, MD, PhD (Ross Tilley Burn Centre, Sunnybrook Health Sciences Centre, Toronto, Ontario) is developing a bioprinter to create skin. The bioprinter dispenses different types of cells (grown from the patient’s own cells) into a hydrogel matrix in 3 dimensions. Currently, Dr. Jeschke is working with mice; however, in a few years, he anticipates treating human patients. Other investigators, such as Michael C. McAlpine (Princeton University, New Jersey), recently used a commercial 3-dimensional printer to make an ear; the “inks” included calf cells and a silver nanoparticle paste that formed a coiled antenna inside the cartilage that was capable of receiving electromagnetic signals and transmitting them to the brain.
What’s the issue?
The ability to grow tissue organs is going to revolutionize the surgical management of patients who need solid organ replacement; kidneys, lungs, pancreases, spleens, and tracheas have already been successfully grown. Indeed several investigators are making ears and noses. How long will it be before flaps and grafts to repair wound defects following extensive and deforming skin cancer surgery are replaced by ears and noses that are grown from the patient’s own tissue cells or stem cells? Although it seems like science fiction today, how soon will it be before a cutaneous 3-dimensional printer becomes a standard piece of equipment in the dermatologist and dermatologic surgeon’s office for use to create skin to cover postoperative sites that cannot be closed by directly bringing the wound edges together?

In an article published online in Maclean’s magazine on October 15, 2013, the concept of developing artificial body parts is discussed. The technology now exists for scientists to grow tissue organs, such as ears, noses, and fingers. Several groups of investigators, including Anthony Atala, MD (Wake Forest Institute for Regenerative Medicine, Winston-Salem, North Carolina) and Alexander Seifalian, PhD (University College London, England), are busy making ears by creating a biodegradable scaffold onto which cells—either stem cells from the patient or cells that have been harvested from the patient’s original organ—are layered and permitted to multiply. The cell-layered scaffold is then placed in a bioreactor for a couple of weeks. Once ready, the new ear is transplanted onto the patient; subsequently, the scaffold melts away.
Dr. Atala’s laboratory also is busy growing fingers. Meanwhile, Dr. Seifalian grew a nose (inside the patient’s own arm) in only 3 months after the patient lost his nose to skin cancer. He made a mold based on the patient’s original nose, created a scaffold composed of nanocomposite material, added the patient’s stem cells to the scaffold, and put the nose in a bioreactor to mature. Meanwhile, he placed a tissue expander in the patient’s arm and subsequently inserted the nose into the arm so that it could become vascularized and covered by skin. The nose was transplanted to the patient’s face; additional surgery is planned to open the nostrils, followed by seeding them with stem cells to return his sense of smell.
Marc Jeschke, MD, PhD (Ross Tilley Burn Centre, Sunnybrook Health Sciences Centre, Toronto, Ontario) is developing a bioprinter to create skin. The bioprinter dispenses different types of cells (grown from the patient’s own cells) into a hydrogel matrix in 3 dimensions. Currently, Dr. Jeschke is working with mice; however, in a few years, he anticipates treating human patients. Other investigators, such as Michael C. McAlpine (Princeton University, New Jersey), recently used a commercial 3-dimensional printer to make an ear; the “inks” included calf cells and a silver nanoparticle paste that formed a coiled antenna inside the cartilage that was capable of receiving electromagnetic signals and transmitting them to the brain.
What’s the issue?
The ability to grow tissue organs is going to revolutionize the surgical management of patients who need solid organ replacement; kidneys, lungs, pancreases, spleens, and tracheas have already been successfully grown. Indeed several investigators are making ears and noses. How long will it be before flaps and grafts to repair wound defects following extensive and deforming skin cancer surgery are replaced by ears and noses that are grown from the patient’s own tissue cells or stem cells? Although it seems like science fiction today, how soon will it be before a cutaneous 3-dimensional printer becomes a standard piece of equipment in the dermatologist and dermatologic surgeon’s office for use to create skin to cover postoperative sites that cannot be closed by directly bringing the wound edges together?