User login
Metastatic eccrine carcinoma with stomach and pericardial involvement
Skin adnexal tumors (SAT) are rare tumors that make up about 1%-2% of all cutaneous malignancies. They represent a various group of benign and malignant tumors that arise from skin adnexal epithelial structures: hair follicle, pilosebaceous unit, and apocrine or eccrine sweat glands. Although this derivation provides a practical basis for classification, some tumors may exhibit a mixed or more than one line of differentiation, rendering precise classification of those neoplasms difficult, and such cases should be categorized according to prevailing phenotype. In this report, we present a patient with metastatic eccrine carcinoma. Clinical experience for metastatic disease treatment is derived from a few reports, and there are no universal treatment guidelines. Given the few reported cases and the absence of randomized clinical trials for these patients, it is important to collect clinical experiences.
Case presentation and summary
A 56-year-old African man presented with a 5-week history of multiple nontender subcutaneous skin nodules all over his body except for his palms and soles, and associated with generalized itching. He had a mass in the sole of his right foot 35 years previously in another country. The mass had recurred 15 years later and was excised again. The exact etiology of the mass was unknown to the patient. He had no other medical problems. He was on no medications and did not smoke, drink, or use recreational drugs.
His vital signs on admission were normal. Examination was significant for innumerable superficial skin nodules in the scalp, back, torso, and abdomen. The largest was in the neck and measured 4 x 2 cm. A firm right inguinal mass of 7 x 4 cm was palpable. An abdominal exam revealed large ascites but no organomegaly.
The results of laboratory tests were significant for hyponatremia 126 mEq/L (normal, 135-145), hypercalcemia of 12.2 mg/dL (8.5-10.5), with normal phosphorous of 2.5 mg/dL (2.5-4.5), parathyroid of 11.5 pg/ml (6-65), and low vitamin D level of <7 ng/ml (30-100). Other test results were: carcinoembryonic antigen (CEA), 4.36 ng/ml (0.00-2.99); alpha fetoprotein, 2.39 IU/ml (0.00-9.0); calcium 11.6 mg/dL (8.5-10.2); lactate dehydrogenase, 325 U/L (85-210); aspartate aminotransferase, 59 U/L (0-40); alanine aminotransferase 43 U/L (5-35); alkaline phosphatase, 65 u/L (50-120); albumin, 2.7 g/dL (3.8-5.2); white blood cell count, 14.1 k/uL (4.4-10.6); h
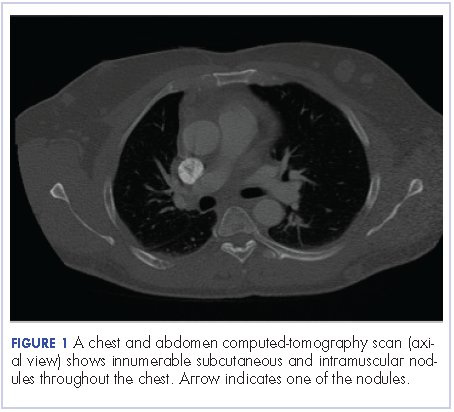
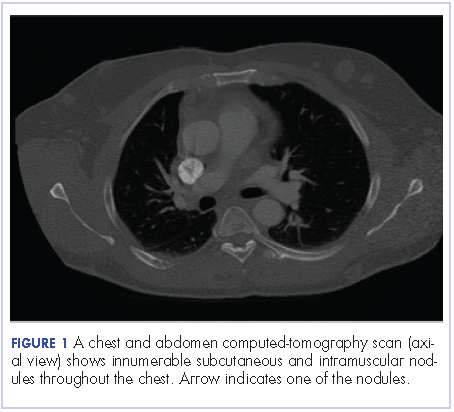
A chest and abdomen computed-tomography scan on presentation showed presence of innumerable subcutaneous and intramuscular nodules throughout the chest, abdomen, and pelvis (Figure 1).
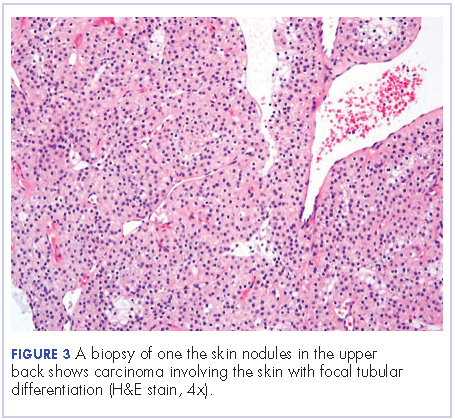
Extensive peritoneal carcinomatosis in addition to moderate ascites and perivascular lymphadenopathy were evident in the abdomen cuts. Remarkably, multiple lytic, osseous metastases were seen with subacute pathologic fracture of right fourth rib in addition to mediastinal lymphadenopathy with small pericardial effusion in the chest cuts. The right thigh mass was described as a large lobulated solid and cystic mass. Ascitic fluid analysis was negative for malignant cells. Biopsy of one the skin nodules in the upper back showed carcinoma involving the skin with focal tubular differentiation (Figure 2).
Immunohistochemical stains were positive for p63, epithelial membrane antigen, high molecular weight keratin, and p40. The lesional cells were negative for CEA, bcl-2, Ber-Ep4, CK7, and CK20. The profile was compatible with a skin adnexal carcinoma of sweat gland origin. The groin lymph node showed eccrine acrospiroma.
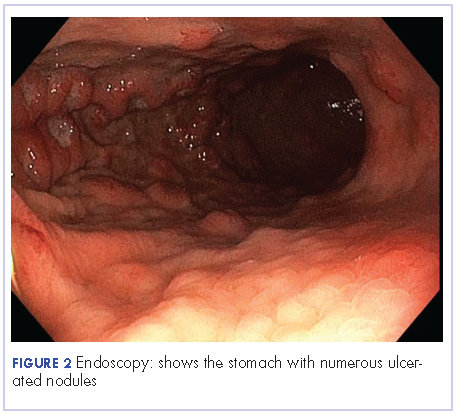
The patient underwent an upper endoscopy to assess for recurrent vomiting and it revealed diffuse areas of large erythematous ulcerated nodules noted in the cardia, fundus, and body of the stomach (Figure 3). A biopsy of the gastric nodules revealed gastric mucosa with metastatic carcinoma.
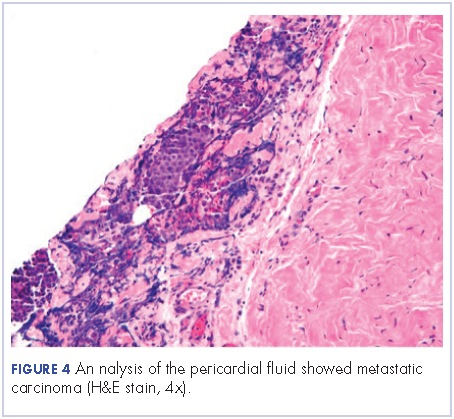
After a thorough review of the literature, he was started on palliative chemotherapy 13 days after initial presentation with docetaxel 75 mg/m2, carboplatin AUC 5 (470 mg), and 5-FU (5-fluorouracil, 750 mg/m2) over 24 hours on days 1 through 5. However, on day 2 of the chemotherapy, he became hypotensive and was found to have cardiac tamponade. He underwent an emergent pericardial window procedure. Analysis of the pericardial fluid was consistent with metastatic carcinoma (Figure 4). Chemotherapy was discontinued while he remained hypotensive requiring multiple vasopressors. His clinical condition did not improve and he passed away 27 days from initial presentation.
Discussion
Sweat gland carcinomas are very rare malignant tumors of the adnexal epithelial structures of the skin, sebaceous, hair follicle, apocrine or eccrine glands that were first described by Cornil in 1865.1 They occur primarily in adult patients, with a peak incidence in fifth and sixth decades of life.2,3 The etiology is unknown, but some cases have been reported to be a consequence of radiation therapy.4 They are almost always an incidental histologic diagnosis.2,5 The tumors usually appear as single nodule, and multinodularity usually associated with both local and metastatic disease.6 There are no characteristic findings to suggest that a particular nodule may represent sweat gland carcinoma, and even if sweat gland tumor is suspected, benign counterparts are more common.
Eccrine carcinoma is the most aggressive among skin adnexal tumors. They can arise on the lower limbs, trunk, head and neck, scalp and ears, upper extremities, abdomen, and genital sites.7
The cells of eccrine sweat glands express low molecular weight keratin, epithelial membrane antigen, carcinoembryonic antigen, as well as S100 protein, smooth muscle actin, p63, calponin, cytokeratin 14, and bcl-2.8 Skin tumors with eccrine differentiation may stain for estrogen and progesterone, which has important clinical implications because those patients can be treated with hormonal therapy.9 Positivity for estrogen receptors does not differentiate cutaneous eccrine tumors from cutaneous metastases of breast cancers.8,9 Androgen receptor evaluation in these cases can help distinguish between the two.10 Human epidermal growth factor receptor 2 (HER-2) is expressed in 3.5% of skin adnexal tumors.11
The molecular pathogenesis of malignant adnexal tumors is not clear, but overexpression of tumor suppressor protein p16 has been described as a common feature in eccrine carcinomas.12
Prognostic factors for sweat gland carcinoma are difficult to identify, because of the small number of reported cases. The likely prognostic factors include size, histological type, lymph node involvement, and presence of distant metastasis. Absent of lymph node involvement correlates with 10-year disease-free survival rate of 56%, which falls to 9% if nodes are involved.13
There are no uniform guidelines for the treatment sweat gland carcinomas, and the clinical experience described in the literature is the only source of available information.
The treatment of choice of all subtypes of localized sweat gland carcinomas is wide surgical excision with broad tumor margins, given the propensity for local recurrences along with regional lymph node dissection in the presence of clinically positive nodes. Prophylactic lymph node resection does not seem to improve survival or decrease recurrence rates.7 The use of adjuvant radiotherapy to prevent local recurrence also is not well established. One report suggested radiosensitivity of these tumors, and adjuvant radiation was therefore recommended in high-risk cases (ie, large tumors of 5 cm and positive surgical margins of 1 cm) and moderate to poorly differentiated tumors with lymphovascular invasion.14 Adjuvant radiation to the involved lymph node basin is suggested in the setting of extranodal extension or extensive involvement, that is, 4 lymph nodes.15 The role of lymphadenectomy has not been adequately addressed in the literature.
The role of chemotherapy in metastatic disease is not clear, but sweat gland carcinomas are considered chemoresistant (Table). Several combinations have been used with short-term responses. In one case treated with doxorubicin, mitomycin, vincristine, and 5-FU followed by maintenance therapy, the patient achieved a complete response that lasted for 16 months.16 In another report, the treatment response was 2 years with treatment consisted of anthracyclin, cyclophosphamide, vincristine, and bloemycin.17 Other combinations used in the literature include carboplatin and paclitaxel, which led to prolonged remission.14 Cisplatin and 5-FU, or cisplatin plus cetuximab have been reported but with discouraging results.18 Results to taxanes showed conflicting results.19,20
Hormonal therapy can be effective in cases in which estrogen and progesterone receptors are expressed, which can range from 19%-30% of eccrine sweat gland carcinomas.21,22 Two cases have reported complete regression of lymph nodes in patients with metastatic disease, and in 1 patient relief from pain caused by bone metastases with durable response of around 3 years.23,24 a
Experience with targeted therapy is very limited. Sunitinib has been reported to have some activity in metastatic adnexal tumors as a second-line therapy in 2 patients, with disease control for 8 and 10 months respectively.25 Trastuzumab has been reported as having activity in 1 patient with strong HER2 expression (IHC score of 3+, denoting HER2 positivity), with complete regression of metastatic tumor. Upon progression in the same patient, a combination of lapatinib and capecitabine also showed positive response.26
In conclusion, metastatic sweat gland tumors treatment has not been standardized because of a dearth of reports in the literatures. Its early identification and complete excision gives the best chance of a cure. Neither chemotherapy nor radiation therapy has been proven to be of clinical benefit in treating metastatic disease.
1. Gates O, Warren S, Warvi WN. Tumors of sweat glands. Am J Pathol. 1943;19(4):591-631.
2. Mitts DL, Smith MT, Russell L, Bannayan GA, Cruz AB. Sweat gland carcinoma: a clinico-pathological reappraisal. J Surg Oncol. 1976;8(1):23-29.
3. Panoussopoulos D, Darom A, Lazaris AC, Misthos P, Papadimitriou K, Androulakis G. Sweat gland carcinoma with multiple local recurrences: a case report. Adv Clin Path. 1999;3(3):63-68.
4. Marone U, Caracò C, Anniciello AM, et al. Metastatic eccrine porocarcinoma : report of a case and review of the literature. World J Surg Oncol. 2011;9:32.
5. Yildirim S, Aköz T, Akan M, Ege GA. De novo malignant eccrine spiradenoma with an interesting and unusual location. Dermatol Surg. 2001;27(4):417-420.
6. Shaw M, McKee PH, Lowe D, Black MM. Malignant eccrine poroma: a study of twenty-seven cases. Br J Dermatol. 1982;107(6):675-680.
7. De Iuliis F, Amoroso L, Taglieri L, et al. Chemotherapy of rare skin adnexal tumors: a review of literature. Anticancer Res. 2014;34(10):5263-5268.
8. Alsaad KO, Obaidat NA, Ghazarian D. Skin adnexal neoplasms – part 1: an approach to tumours of the pilosebaceous unit. J Clin Pathol. 2007;60(2):129-144.
9. Serhrouchni KI, Harmouch T, Chbani L, et al. Eccrine carcinoma : a rare cutaneous neoplasm. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3570399/. Published online February 4, 2013. Accessed October 11, 2017.
10. Shidham VB, Komorowski RA, Machhi JK. Androgen receptor expression in metastatic adenocarcinoma in females favors a breast primary. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1601970/. Published online October 4, 2006. Accessed October 11, 2017.
11. Hiatt KM, Pillow JL, Smoller BR. Her-2 expression in cutaneous eccrine and apocrine neoplasms. Mod Pathol. 2004;17(1):28-32.
12. Gu L-H, Ichiki Y, Kitajima Y. Aberrant expression of p16 and RB protein in eccrine porocarcinoma. J Cutan Pathol. 2002;29(8):473-479.
13. el-Domeiri AA, Brasfield RD, Huvos AG, Strong EW. Sweat gland carcinoma: a clinico-pathologic study of 83 patients. Ann Surg. 1971;173(2):270-274.
14. Tlemcani K, Levine D, Smith R V, et al. Metastatic apocrine carcinoma of the scalp: prolonged response to systemic chemotherapy. J Clin Oncol. 2010;28(24):e412-e414.
15. Chamberlain RS, Huber K, White JC, Travaglino-Parda R. Apocrine gland carcinoma of the axilla: review of the literature and recommendations for treatment. Am J Clin Oncol. 1999;22(2):131-135.
16. Gutermuth J, Audring H, Voit C, Trefzer U, Haas N. Antitumour activity of paclitaxel and interferon-alpha in a case of metastatic eccrine porocarcinoma. J Eur Acad Dermatol Venereol. 2004;18(4):477-479.
17. Mezger J, Remberger K, Schalhorn A, Wohlrab A, Wilmanns W. Treatment of metastatic sweat gland carcinoma by a four drug combination chemotherapy: response in two cases. Med Oncol Tumor Pharmacother. 1986;3(1):29-34.
18. Aaribi I, Mohtaram A, Ben Ameur El Youbi M, et al. Successful management of metastatic eccrine porocarcinoma. https://www.hindawi.com/journals/crionm/2013/282536/. Published 2013. Accessed October 10, 2017.
19. Shiohara J, Koga H, Uhara H, Takata M, Saida T. Eccrine porocarcinoma: clinical and pathological studies of 12 cases. J Dermatol. 2007;34(8):516-522.
20. Swanson PE, Mazoujian G, Mills SE, Campbell RJ, Wick MR. Immunoreactivity for estrogen receptor protein in sweat gland tumors. Am J Surg Pathol. 1991;15(9):835-841.
21. Busam KJ, Tan LK, Granter SR, et al. Epidermal growth factor, estrogen, and progesterone receptor expression in primary sweat gland carcinomas and primary and metastatic mammary carcinomas. Mod Pathol. 1999;12(8):786-793.
22. Sridhar KS, Benedetto P, Otrakji CL, Charyulu KK. Response of eccrine adenocarcinoma to tamoxifen. Cancer. 1989;64(2):366-370.
23. Daniel SJ, Nader R, Kost K, Hüttner I. Facial sweat gland carcinoma metastasizing to neck nodes: a diagnostic and therapeutic challenge. Arch Otolaryngol Head Neck Surg. 2001;127(12):1495-1498.
24. Battistella M, Mateus C, Lassau N, et al. Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol. 2010;24(2):199-203.
25. Hidaka T, Fujimura T, Watabe A, et al. Successful treatment of HER-2-positive metastatic apocrine carcinoma of the skin with lapatinib and capecitabine. Acta Derm Venereol. 2012;92(6):654-655.
26. Mandaliya H, Nordman I. Metastatic eccrine porocarcinoma: a rare case of successful treatment. Case Rep Oncol. 2016;9(2):454-456.
27. de Bree E, Volalakis E, Tsetis D, et al. Treatment of advanced malignant eccrine poroma with locoregional chemotherapy. Br J Dermatol. 2005;152(5):1051-1055.
28. Bahl A, Sharma DN, Julka PK, Das A, Rath GK. Sweat gland carcinoma with lung metastases. J Cancer Res Ther. 2(4):209-211.
29. Wang X-X, Wang H-Y, Zheng J-N, Sui J-C. Primary cutaneous sweat gland carcinoma. J Cancer Res Ther. 10(2):390-392.
Skin adnexal tumors (SAT) are rare tumors that make up about 1%-2% of all cutaneous malignancies. They represent a various group of benign and malignant tumors that arise from skin adnexal epithelial structures: hair follicle, pilosebaceous unit, and apocrine or eccrine sweat glands. Although this derivation provides a practical basis for classification, some tumors may exhibit a mixed or more than one line of differentiation, rendering precise classification of those neoplasms difficult, and such cases should be categorized according to prevailing phenotype. In this report, we present a patient with metastatic eccrine carcinoma. Clinical experience for metastatic disease treatment is derived from a few reports, and there are no universal treatment guidelines. Given the few reported cases and the absence of randomized clinical trials for these patients, it is important to collect clinical experiences.
Case presentation and summary
A 56-year-old African man presented with a 5-week history of multiple nontender subcutaneous skin nodules all over his body except for his palms and soles, and associated with generalized itching. He had a mass in the sole of his right foot 35 years previously in another country. The mass had recurred 15 years later and was excised again. The exact etiology of the mass was unknown to the patient. He had no other medical problems. He was on no medications and did not smoke, drink, or use recreational drugs.
His vital signs on admission were normal. Examination was significant for innumerable superficial skin nodules in the scalp, back, torso, and abdomen. The largest was in the neck and measured 4 x 2 cm. A firm right inguinal mass of 7 x 4 cm was palpable. An abdominal exam revealed large ascites but no organomegaly.
The results of laboratory tests were significant for hyponatremia 126 mEq/L (normal, 135-145), hypercalcemia of 12.2 mg/dL (8.5-10.5), with normal phosphorous of 2.5 mg/dL (2.5-4.5), parathyroid of 11.5 pg/ml (6-65), and low vitamin D level of <7 ng/ml (30-100). Other test results were: carcinoembryonic antigen (CEA), 4.36 ng/ml (0.00-2.99); alpha fetoprotein, 2.39 IU/ml (0.00-9.0); calcium 11.6 mg/dL (8.5-10.2); lactate dehydrogenase, 325 U/L (85-210); aspartate aminotransferase, 59 U/L (0-40); alanine aminotransferase 43 U/L (5-35); alkaline phosphatase, 65 u/L (50-120); albumin, 2.7 g/dL (3.8-5.2); white blood cell count, 14.1 k/uL (4.4-10.6); h
A chest and abdomen computed-tomography scan on presentation showed presence of innumerable subcutaneous and intramuscular nodules throughout the chest, abdomen, and pelvis (Figure 1).
Extensive peritoneal carcinomatosis in addition to moderate ascites and perivascular lymphadenopathy were evident in the abdomen cuts. Remarkably, multiple lytic, osseous metastases were seen with subacute pathologic fracture of right fourth rib in addition to mediastinal lymphadenopathy with small pericardial effusion in the chest cuts. The right thigh mass was described as a large lobulated solid and cystic mass. Ascitic fluid analysis was negative for malignant cells. Biopsy of one the skin nodules in the upper back showed carcinoma involving the skin with focal tubular differentiation (Figure 2).
Immunohistochemical stains were positive for p63, epithelial membrane antigen, high molecular weight keratin, and p40. The lesional cells were negative for CEA, bcl-2, Ber-Ep4, CK7, and CK20. The profile was compatible with a skin adnexal carcinoma of sweat gland origin. The groin lymph node showed eccrine acrospiroma.
The patient underwent an upper endoscopy to assess for recurrent vomiting and it revealed diffuse areas of large erythematous ulcerated nodules noted in the cardia, fundus, and body of the stomach (Figure 3). A biopsy of the gastric nodules revealed gastric mucosa with metastatic carcinoma.
After a thorough review of the literature, he was started on palliative chemotherapy 13 days after initial presentation with docetaxel 75 mg/m2, carboplatin AUC 5 (470 mg), and 5-FU (5-fluorouracil, 750 mg/m2) over 24 hours on days 1 through 5. However, on day 2 of the chemotherapy, he became hypotensive and was found to have cardiac tamponade. He underwent an emergent pericardial window procedure. Analysis of the pericardial fluid was consistent with metastatic carcinoma (Figure 4). Chemotherapy was discontinued while he remained hypotensive requiring multiple vasopressors. His clinical condition did not improve and he passed away 27 days from initial presentation.
Discussion
Sweat gland carcinomas are very rare malignant tumors of the adnexal epithelial structures of the skin, sebaceous, hair follicle, apocrine or eccrine glands that were first described by Cornil in 1865.1 They occur primarily in adult patients, with a peak incidence in fifth and sixth decades of life.2,3 The etiology is unknown, but some cases have been reported to be a consequence of radiation therapy.4 They are almost always an incidental histologic diagnosis.2,5 The tumors usually appear as single nodule, and multinodularity usually associated with both local and metastatic disease.6 There are no characteristic findings to suggest that a particular nodule may represent sweat gland carcinoma, and even if sweat gland tumor is suspected, benign counterparts are more common.
Eccrine carcinoma is the most aggressive among skin adnexal tumors. They can arise on the lower limbs, trunk, head and neck, scalp and ears, upper extremities, abdomen, and genital sites.7
The cells of eccrine sweat glands express low molecular weight keratin, epithelial membrane antigen, carcinoembryonic antigen, as well as S100 protein, smooth muscle actin, p63, calponin, cytokeratin 14, and bcl-2.8 Skin tumors with eccrine differentiation may stain for estrogen and progesterone, which has important clinical implications because those patients can be treated with hormonal therapy.9 Positivity for estrogen receptors does not differentiate cutaneous eccrine tumors from cutaneous metastases of breast cancers.8,9 Androgen receptor evaluation in these cases can help distinguish between the two.10 Human epidermal growth factor receptor 2 (HER-2) is expressed in 3.5% of skin adnexal tumors.11
The molecular pathogenesis of malignant adnexal tumors is not clear, but overexpression of tumor suppressor protein p16 has been described as a common feature in eccrine carcinomas.12
Prognostic factors for sweat gland carcinoma are difficult to identify, because of the small number of reported cases. The likely prognostic factors include size, histological type, lymph node involvement, and presence of distant metastasis. Absent of lymph node involvement correlates with 10-year disease-free survival rate of 56%, which falls to 9% if nodes are involved.13
There are no uniform guidelines for the treatment sweat gland carcinomas, and the clinical experience described in the literature is the only source of available information.
The treatment of choice of all subtypes of localized sweat gland carcinomas is wide surgical excision with broad tumor margins, given the propensity for local recurrences along with regional lymph node dissection in the presence of clinically positive nodes. Prophylactic lymph node resection does not seem to improve survival or decrease recurrence rates.7 The use of adjuvant radiotherapy to prevent local recurrence also is not well established. One report suggested radiosensitivity of these tumors, and adjuvant radiation was therefore recommended in high-risk cases (ie, large tumors of 5 cm and positive surgical margins of 1 cm) and moderate to poorly differentiated tumors with lymphovascular invasion.14 Adjuvant radiation to the involved lymph node basin is suggested in the setting of extranodal extension or extensive involvement, that is, 4 lymph nodes.15 The role of lymphadenectomy has not been adequately addressed in the literature.
The role of chemotherapy in metastatic disease is not clear, but sweat gland carcinomas are considered chemoresistant (Table). Several combinations have been used with short-term responses. In one case treated with doxorubicin, mitomycin, vincristine, and 5-FU followed by maintenance therapy, the patient achieved a complete response that lasted for 16 months.16 In another report, the treatment response was 2 years with treatment consisted of anthracyclin, cyclophosphamide, vincristine, and bloemycin.17 Other combinations used in the literature include carboplatin and paclitaxel, which led to prolonged remission.14 Cisplatin and 5-FU, or cisplatin plus cetuximab have been reported but with discouraging results.18 Results to taxanes showed conflicting results.19,20
Hormonal therapy can be effective in cases in which estrogen and progesterone receptors are expressed, which can range from 19%-30% of eccrine sweat gland carcinomas.21,22 Two cases have reported complete regression of lymph nodes in patients with metastatic disease, and in 1 patient relief from pain caused by bone metastases with durable response of around 3 years.23,24 a
Experience with targeted therapy is very limited. Sunitinib has been reported to have some activity in metastatic adnexal tumors as a second-line therapy in 2 patients, with disease control for 8 and 10 months respectively.25 Trastuzumab has been reported as having activity in 1 patient with strong HER2 expression (IHC score of 3+, denoting HER2 positivity), with complete regression of metastatic tumor. Upon progression in the same patient, a combination of lapatinib and capecitabine also showed positive response.26
In conclusion, metastatic sweat gland tumors treatment has not been standardized because of a dearth of reports in the literatures. Its early identification and complete excision gives the best chance of a cure. Neither chemotherapy nor radiation therapy has been proven to be of clinical benefit in treating metastatic disease.
Skin adnexal tumors (SAT) are rare tumors that make up about 1%-2% of all cutaneous malignancies. They represent a various group of benign and malignant tumors that arise from skin adnexal epithelial structures: hair follicle, pilosebaceous unit, and apocrine or eccrine sweat glands. Although this derivation provides a practical basis for classification, some tumors may exhibit a mixed or more than one line of differentiation, rendering precise classification of those neoplasms difficult, and such cases should be categorized according to prevailing phenotype. In this report, we present a patient with metastatic eccrine carcinoma. Clinical experience for metastatic disease treatment is derived from a few reports, and there are no universal treatment guidelines. Given the few reported cases and the absence of randomized clinical trials for these patients, it is important to collect clinical experiences.
Case presentation and summary
A 56-year-old African man presented with a 5-week history of multiple nontender subcutaneous skin nodules all over his body except for his palms and soles, and associated with generalized itching. He had a mass in the sole of his right foot 35 years previously in another country. The mass had recurred 15 years later and was excised again. The exact etiology of the mass was unknown to the patient. He had no other medical problems. He was on no medications and did not smoke, drink, or use recreational drugs.
His vital signs on admission were normal. Examination was significant for innumerable superficial skin nodules in the scalp, back, torso, and abdomen. The largest was in the neck and measured 4 x 2 cm. A firm right inguinal mass of 7 x 4 cm was palpable. An abdominal exam revealed large ascites but no organomegaly.
The results of laboratory tests were significant for hyponatremia 126 mEq/L (normal, 135-145), hypercalcemia of 12.2 mg/dL (8.5-10.5), with normal phosphorous of 2.5 mg/dL (2.5-4.5), parathyroid of 11.5 pg/ml (6-65), and low vitamin D level of <7 ng/ml (30-100). Other test results were: carcinoembryonic antigen (CEA), 4.36 ng/ml (0.00-2.99); alpha fetoprotein, 2.39 IU/ml (0.00-9.0); calcium 11.6 mg/dL (8.5-10.2); lactate dehydrogenase, 325 U/L (85-210); aspartate aminotransferase, 59 U/L (0-40); alanine aminotransferase 43 U/L (5-35); alkaline phosphatase, 65 u/L (50-120); albumin, 2.7 g/dL (3.8-5.2); white blood cell count, 14.1 k/uL (4.4-10.6); h
A chest and abdomen computed-tomography scan on presentation showed presence of innumerable subcutaneous and intramuscular nodules throughout the chest, abdomen, and pelvis (Figure 1).
Extensive peritoneal carcinomatosis in addition to moderate ascites and perivascular lymphadenopathy were evident in the abdomen cuts. Remarkably, multiple lytic, osseous metastases were seen with subacute pathologic fracture of right fourth rib in addition to mediastinal lymphadenopathy with small pericardial effusion in the chest cuts. The right thigh mass was described as a large lobulated solid and cystic mass. Ascitic fluid analysis was negative for malignant cells. Biopsy of one the skin nodules in the upper back showed carcinoma involving the skin with focal tubular differentiation (Figure 2).
Immunohistochemical stains were positive for p63, epithelial membrane antigen, high molecular weight keratin, and p40. The lesional cells were negative for CEA, bcl-2, Ber-Ep4, CK7, and CK20. The profile was compatible with a skin adnexal carcinoma of sweat gland origin. The groin lymph node showed eccrine acrospiroma.
The patient underwent an upper endoscopy to assess for recurrent vomiting and it revealed diffuse areas of large erythematous ulcerated nodules noted in the cardia, fundus, and body of the stomach (Figure 3). A biopsy of the gastric nodules revealed gastric mucosa with metastatic carcinoma.
After a thorough review of the literature, he was started on palliative chemotherapy 13 days after initial presentation with docetaxel 75 mg/m2, carboplatin AUC 5 (470 mg), and 5-FU (5-fluorouracil, 750 mg/m2) over 24 hours on days 1 through 5. However, on day 2 of the chemotherapy, he became hypotensive and was found to have cardiac tamponade. He underwent an emergent pericardial window procedure. Analysis of the pericardial fluid was consistent with metastatic carcinoma (Figure 4). Chemotherapy was discontinued while he remained hypotensive requiring multiple vasopressors. His clinical condition did not improve and he passed away 27 days from initial presentation.
Discussion
Sweat gland carcinomas are very rare malignant tumors of the adnexal epithelial structures of the skin, sebaceous, hair follicle, apocrine or eccrine glands that were first described by Cornil in 1865.1 They occur primarily in adult patients, with a peak incidence in fifth and sixth decades of life.2,3 The etiology is unknown, but some cases have been reported to be a consequence of radiation therapy.4 They are almost always an incidental histologic diagnosis.2,5 The tumors usually appear as single nodule, and multinodularity usually associated with both local and metastatic disease.6 There are no characteristic findings to suggest that a particular nodule may represent sweat gland carcinoma, and even if sweat gland tumor is suspected, benign counterparts are more common.
Eccrine carcinoma is the most aggressive among skin adnexal tumors. They can arise on the lower limbs, trunk, head and neck, scalp and ears, upper extremities, abdomen, and genital sites.7
The cells of eccrine sweat glands express low molecular weight keratin, epithelial membrane antigen, carcinoembryonic antigen, as well as S100 protein, smooth muscle actin, p63, calponin, cytokeratin 14, and bcl-2.8 Skin tumors with eccrine differentiation may stain for estrogen and progesterone, which has important clinical implications because those patients can be treated with hormonal therapy.9 Positivity for estrogen receptors does not differentiate cutaneous eccrine tumors from cutaneous metastases of breast cancers.8,9 Androgen receptor evaluation in these cases can help distinguish between the two.10 Human epidermal growth factor receptor 2 (HER-2) is expressed in 3.5% of skin adnexal tumors.11
The molecular pathogenesis of malignant adnexal tumors is not clear, but overexpression of tumor suppressor protein p16 has been described as a common feature in eccrine carcinomas.12
Prognostic factors for sweat gland carcinoma are difficult to identify, because of the small number of reported cases. The likely prognostic factors include size, histological type, lymph node involvement, and presence of distant metastasis. Absent of lymph node involvement correlates with 10-year disease-free survival rate of 56%, which falls to 9% if nodes are involved.13
There are no uniform guidelines for the treatment sweat gland carcinomas, and the clinical experience described in the literature is the only source of available information.
The treatment of choice of all subtypes of localized sweat gland carcinomas is wide surgical excision with broad tumor margins, given the propensity for local recurrences along with regional lymph node dissection in the presence of clinically positive nodes. Prophylactic lymph node resection does not seem to improve survival or decrease recurrence rates.7 The use of adjuvant radiotherapy to prevent local recurrence also is not well established. One report suggested radiosensitivity of these tumors, and adjuvant radiation was therefore recommended in high-risk cases (ie, large tumors of 5 cm and positive surgical margins of 1 cm) and moderate to poorly differentiated tumors with lymphovascular invasion.14 Adjuvant radiation to the involved lymph node basin is suggested in the setting of extranodal extension or extensive involvement, that is, 4 lymph nodes.15 The role of lymphadenectomy has not been adequately addressed in the literature.
The role of chemotherapy in metastatic disease is not clear, but sweat gland carcinomas are considered chemoresistant (Table). Several combinations have been used with short-term responses. In one case treated with doxorubicin, mitomycin, vincristine, and 5-FU followed by maintenance therapy, the patient achieved a complete response that lasted for 16 months.16 In another report, the treatment response was 2 years with treatment consisted of anthracyclin, cyclophosphamide, vincristine, and bloemycin.17 Other combinations used in the literature include carboplatin and paclitaxel, which led to prolonged remission.14 Cisplatin and 5-FU, or cisplatin plus cetuximab have been reported but with discouraging results.18 Results to taxanes showed conflicting results.19,20
Hormonal therapy can be effective in cases in which estrogen and progesterone receptors are expressed, which can range from 19%-30% of eccrine sweat gland carcinomas.21,22 Two cases have reported complete regression of lymph nodes in patients with metastatic disease, and in 1 patient relief from pain caused by bone metastases with durable response of around 3 years.23,24 a
Experience with targeted therapy is very limited. Sunitinib has been reported to have some activity in metastatic adnexal tumors as a second-line therapy in 2 patients, with disease control for 8 and 10 months respectively.25 Trastuzumab has been reported as having activity in 1 patient with strong HER2 expression (IHC score of 3+, denoting HER2 positivity), with complete regression of metastatic tumor. Upon progression in the same patient, a combination of lapatinib and capecitabine also showed positive response.26
In conclusion, metastatic sweat gland tumors treatment has not been standardized because of a dearth of reports in the literatures. Its early identification and complete excision gives the best chance of a cure. Neither chemotherapy nor radiation therapy has been proven to be of clinical benefit in treating metastatic disease.
1. Gates O, Warren S, Warvi WN. Tumors of sweat glands. Am J Pathol. 1943;19(4):591-631.
2. Mitts DL, Smith MT, Russell L, Bannayan GA, Cruz AB. Sweat gland carcinoma: a clinico-pathological reappraisal. J Surg Oncol. 1976;8(1):23-29.
3. Panoussopoulos D, Darom A, Lazaris AC, Misthos P, Papadimitriou K, Androulakis G. Sweat gland carcinoma with multiple local recurrences: a case report. Adv Clin Path. 1999;3(3):63-68.
4. Marone U, Caracò C, Anniciello AM, et al. Metastatic eccrine porocarcinoma : report of a case and review of the literature. World J Surg Oncol. 2011;9:32.
5. Yildirim S, Aköz T, Akan M, Ege GA. De novo malignant eccrine spiradenoma with an interesting and unusual location. Dermatol Surg. 2001;27(4):417-420.
6. Shaw M, McKee PH, Lowe D, Black MM. Malignant eccrine poroma: a study of twenty-seven cases. Br J Dermatol. 1982;107(6):675-680.
7. De Iuliis F, Amoroso L, Taglieri L, et al. Chemotherapy of rare skin adnexal tumors: a review of literature. Anticancer Res. 2014;34(10):5263-5268.
8. Alsaad KO, Obaidat NA, Ghazarian D. Skin adnexal neoplasms – part 1: an approach to tumours of the pilosebaceous unit. J Clin Pathol. 2007;60(2):129-144.
9. Serhrouchni KI, Harmouch T, Chbani L, et al. Eccrine carcinoma : a rare cutaneous neoplasm. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3570399/. Published online February 4, 2013. Accessed October 11, 2017.
10. Shidham VB, Komorowski RA, Machhi JK. Androgen receptor expression in metastatic adenocarcinoma in females favors a breast primary. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1601970/. Published online October 4, 2006. Accessed October 11, 2017.
11. Hiatt KM, Pillow JL, Smoller BR. Her-2 expression in cutaneous eccrine and apocrine neoplasms. Mod Pathol. 2004;17(1):28-32.
12. Gu L-H, Ichiki Y, Kitajima Y. Aberrant expression of p16 and RB protein in eccrine porocarcinoma. J Cutan Pathol. 2002;29(8):473-479.
13. el-Domeiri AA, Brasfield RD, Huvos AG, Strong EW. Sweat gland carcinoma: a clinico-pathologic study of 83 patients. Ann Surg. 1971;173(2):270-274.
14. Tlemcani K, Levine D, Smith R V, et al. Metastatic apocrine carcinoma of the scalp: prolonged response to systemic chemotherapy. J Clin Oncol. 2010;28(24):e412-e414.
15. Chamberlain RS, Huber K, White JC, Travaglino-Parda R. Apocrine gland carcinoma of the axilla: review of the literature and recommendations for treatment. Am J Clin Oncol. 1999;22(2):131-135.
16. Gutermuth J, Audring H, Voit C, Trefzer U, Haas N. Antitumour activity of paclitaxel and interferon-alpha in a case of metastatic eccrine porocarcinoma. J Eur Acad Dermatol Venereol. 2004;18(4):477-479.
17. Mezger J, Remberger K, Schalhorn A, Wohlrab A, Wilmanns W. Treatment of metastatic sweat gland carcinoma by a four drug combination chemotherapy: response in two cases. Med Oncol Tumor Pharmacother. 1986;3(1):29-34.
18. Aaribi I, Mohtaram A, Ben Ameur El Youbi M, et al. Successful management of metastatic eccrine porocarcinoma. https://www.hindawi.com/journals/crionm/2013/282536/. Published 2013. Accessed October 10, 2017.
19. Shiohara J, Koga H, Uhara H, Takata M, Saida T. Eccrine porocarcinoma: clinical and pathological studies of 12 cases. J Dermatol. 2007;34(8):516-522.
20. Swanson PE, Mazoujian G, Mills SE, Campbell RJ, Wick MR. Immunoreactivity for estrogen receptor protein in sweat gland tumors. Am J Surg Pathol. 1991;15(9):835-841.
21. Busam KJ, Tan LK, Granter SR, et al. Epidermal growth factor, estrogen, and progesterone receptor expression in primary sweat gland carcinomas and primary and metastatic mammary carcinomas. Mod Pathol. 1999;12(8):786-793.
22. Sridhar KS, Benedetto P, Otrakji CL, Charyulu KK. Response of eccrine adenocarcinoma to tamoxifen. Cancer. 1989;64(2):366-370.
23. Daniel SJ, Nader R, Kost K, Hüttner I. Facial sweat gland carcinoma metastasizing to neck nodes: a diagnostic and therapeutic challenge. Arch Otolaryngol Head Neck Surg. 2001;127(12):1495-1498.
24. Battistella M, Mateus C, Lassau N, et al. Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol. 2010;24(2):199-203.
25. Hidaka T, Fujimura T, Watabe A, et al. Successful treatment of HER-2-positive metastatic apocrine carcinoma of the skin with lapatinib and capecitabine. Acta Derm Venereol. 2012;92(6):654-655.
26. Mandaliya H, Nordman I. Metastatic eccrine porocarcinoma: a rare case of successful treatment. Case Rep Oncol. 2016;9(2):454-456.
27. de Bree E, Volalakis E, Tsetis D, et al. Treatment of advanced malignant eccrine poroma with locoregional chemotherapy. Br J Dermatol. 2005;152(5):1051-1055.
28. Bahl A, Sharma DN, Julka PK, Das A, Rath GK. Sweat gland carcinoma with lung metastases. J Cancer Res Ther. 2(4):209-211.
29. Wang X-X, Wang H-Y, Zheng J-N, Sui J-C. Primary cutaneous sweat gland carcinoma. J Cancer Res Ther. 10(2):390-392.
1. Gates O, Warren S, Warvi WN. Tumors of sweat glands. Am J Pathol. 1943;19(4):591-631.
2. Mitts DL, Smith MT, Russell L, Bannayan GA, Cruz AB. Sweat gland carcinoma: a clinico-pathological reappraisal. J Surg Oncol. 1976;8(1):23-29.
3. Panoussopoulos D, Darom A, Lazaris AC, Misthos P, Papadimitriou K, Androulakis G. Sweat gland carcinoma with multiple local recurrences: a case report. Adv Clin Path. 1999;3(3):63-68.
4. Marone U, Caracò C, Anniciello AM, et al. Metastatic eccrine porocarcinoma : report of a case and review of the literature. World J Surg Oncol. 2011;9:32.
5. Yildirim S, Aköz T, Akan M, Ege GA. De novo malignant eccrine spiradenoma with an interesting and unusual location. Dermatol Surg. 2001;27(4):417-420.
6. Shaw M, McKee PH, Lowe D, Black MM. Malignant eccrine poroma: a study of twenty-seven cases. Br J Dermatol. 1982;107(6):675-680.
7. De Iuliis F, Amoroso L, Taglieri L, et al. Chemotherapy of rare skin adnexal tumors: a review of literature. Anticancer Res. 2014;34(10):5263-5268.
8. Alsaad KO, Obaidat NA, Ghazarian D. Skin adnexal neoplasms – part 1: an approach to tumours of the pilosebaceous unit. J Clin Pathol. 2007;60(2):129-144.
9. Serhrouchni KI, Harmouch T, Chbani L, et al. Eccrine carcinoma : a rare cutaneous neoplasm. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3570399/. Published online February 4, 2013. Accessed October 11, 2017.
10. Shidham VB, Komorowski RA, Machhi JK. Androgen receptor expression in metastatic adenocarcinoma in females favors a breast primary. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1601970/. Published online October 4, 2006. Accessed October 11, 2017.
11. Hiatt KM, Pillow JL, Smoller BR. Her-2 expression in cutaneous eccrine and apocrine neoplasms. Mod Pathol. 2004;17(1):28-32.
12. Gu L-H, Ichiki Y, Kitajima Y. Aberrant expression of p16 and RB protein in eccrine porocarcinoma. J Cutan Pathol. 2002;29(8):473-479.
13. el-Domeiri AA, Brasfield RD, Huvos AG, Strong EW. Sweat gland carcinoma: a clinico-pathologic study of 83 patients. Ann Surg. 1971;173(2):270-274.
14. Tlemcani K, Levine D, Smith R V, et al. Metastatic apocrine carcinoma of the scalp: prolonged response to systemic chemotherapy. J Clin Oncol. 2010;28(24):e412-e414.
15. Chamberlain RS, Huber K, White JC, Travaglino-Parda R. Apocrine gland carcinoma of the axilla: review of the literature and recommendations for treatment. Am J Clin Oncol. 1999;22(2):131-135.
16. Gutermuth J, Audring H, Voit C, Trefzer U, Haas N. Antitumour activity of paclitaxel and interferon-alpha in a case of metastatic eccrine porocarcinoma. J Eur Acad Dermatol Venereol. 2004;18(4):477-479.
17. Mezger J, Remberger K, Schalhorn A, Wohlrab A, Wilmanns W. Treatment of metastatic sweat gland carcinoma by a four drug combination chemotherapy: response in two cases. Med Oncol Tumor Pharmacother. 1986;3(1):29-34.
18. Aaribi I, Mohtaram A, Ben Ameur El Youbi M, et al. Successful management of metastatic eccrine porocarcinoma. https://www.hindawi.com/journals/crionm/2013/282536/. Published 2013. Accessed October 10, 2017.
19. Shiohara J, Koga H, Uhara H, Takata M, Saida T. Eccrine porocarcinoma: clinical and pathological studies of 12 cases. J Dermatol. 2007;34(8):516-522.
20. Swanson PE, Mazoujian G, Mills SE, Campbell RJ, Wick MR. Immunoreactivity for estrogen receptor protein in sweat gland tumors. Am J Surg Pathol. 1991;15(9):835-841.
21. Busam KJ, Tan LK, Granter SR, et al. Epidermal growth factor, estrogen, and progesterone receptor expression in primary sweat gland carcinomas and primary and metastatic mammary carcinomas. Mod Pathol. 1999;12(8):786-793.
22. Sridhar KS, Benedetto P, Otrakji CL, Charyulu KK. Response of eccrine adenocarcinoma to tamoxifen. Cancer. 1989;64(2):366-370.
23. Daniel SJ, Nader R, Kost K, Hüttner I. Facial sweat gland carcinoma metastasizing to neck nodes: a diagnostic and therapeutic challenge. Arch Otolaryngol Head Neck Surg. 2001;127(12):1495-1498.
24. Battistella M, Mateus C, Lassau N, et al. Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol. 2010;24(2):199-203.
25. Hidaka T, Fujimura T, Watabe A, et al. Successful treatment of HER-2-positive metastatic apocrine carcinoma of the skin with lapatinib and capecitabine. Acta Derm Venereol. 2012;92(6):654-655.
26. Mandaliya H, Nordman I. Metastatic eccrine porocarcinoma: a rare case of successful treatment. Case Rep Oncol. 2016;9(2):454-456.
27. de Bree E, Volalakis E, Tsetis D, et al. Treatment of advanced malignant eccrine poroma with locoregional chemotherapy. Br J Dermatol. 2005;152(5):1051-1055.
28. Bahl A, Sharma DN, Julka PK, Das A, Rath GK. Sweat gland carcinoma with lung metastases. J Cancer Res Ther. 2(4):209-211.
29. Wang X-X, Wang H-Y, Zheng J-N, Sui J-C. Primary cutaneous sweat gland carcinoma. J Cancer Res Ther. 10(2):390-392.
Cold hemolytic anemia: a rare complication of influenza A
Autoimmune hemolytic anemia (AIHA) is characterized by the temperature at which the auto-antibody has the greatest avidity for the target red cell antigen, either warm or cold forms. It is detected by a positive direct antiglobulin test (DAT) also known as the direct Coombs test. DAT is used to determine if red cells have been coated in vivo with immunoglobulin, complement, or both.1 Some causes of a positive DAT include hemolytic transfusion reactions, hemolytic disease of the fetus and newborn, AIHA, and drug-induced immune hemolysis.
Case presentation and summary
A 58-year-old woman from Brazil with past medical history only significant for cholecystectomy and cesarean section had been visiting in United States for 2 months when she presented to an outside hospital with fever, shortness of breath, and syncope that had resulted in a foot injury. She reported she had been feeling short of breath and had a nonproductive cough and malaise for about 2 weeks before presentation with sick contacts at home. On admission it was noted that she had a hemoglobin level of 7.7 g/dL (normal, 12.0-15.5 g/dL; MCV, 94 fL), total bilirubin of 2.14 mg/dL (normal, 0.2-1.0 mg/dL), and lactate dehydrogenase of 523 U/L (normal, 81-234 U/L). There were no signs of bleeding on her examination. Her DAT was positive and moderate red blood cell agglutination was reported. During the first admission at the outside hospital she was diagnosed with influenza A and completed a full course of oseltamivir (75 mg po twice daily for 5 days). A chest X-ray was negative for infiltrates and showed that the patient’s lung fields were clear. She was transfused 2 units of packed red blood cells with response in hemoglobin up to 9.8 g/dL. The patient was treated with dexamethasone (4 mg IV Q8) as an inpatient and was discharged on a prednisone taper (40 mg, with taper by 10 mg every 3 days) with hemoglobin of 8.1 g/dL.
The patient continued to have nonproductive cough, dyspnea, fevers, chills, and generalized weakness, when she returned to the same outside hospital’s emergency department 2 days after her discharge. At that time, it was noted that she had leucocytosis (white blood cell count, 34.6 x 109 per L), a hemoglobin level of 6.8 g/dL, and her total bilirubin level was 6.9 mg/dL. Her hemodynamics were unstable and she was admitted to their intensive care unit. The results of a chest X-ray revealed right lung consolidation.
The day after this admission, her hemoglobin level fell to 4.7 g/dL, and she was transfused 2 units of packed red blood cells before being transferred to our hospital. A chest X-ray at our hospital confirmed a right lung infiltrate. Vancomycin (1,250 mg IV Q12), levaquin (750 mg IV Q24), and maxipime (1 g IV Q12) were initiated for pneumonia and the patient was transferred to our hospital’s intensive care unit. She was afebrile at 98.3°F, her pulse rate was 84 beats per minute, she was tachypneic with respiratory rate of 26 breaths per minute, her blood pressure was 98/51 mmHg, and she had an oxygen saturation of 99% on 2L oxygen via nasal cannula.
On physical examination she was noted to have scleral icterus and was in mild respiratory distress. A chest X-ray revealed a patchy opacity in the right mid to lower lung. Her initial complete blood panel revealed anemia, with hemoglobin, 6.3 g/dL; white blood cell count, 27 x 109 per L; and platelets, 533 x 109 per L. The patient was then transfused another 2 units of packed red blood cells. She was given intravenous hydration, acetaminophen, and albuterol nebulizer treatments as supportive care. She was provided with blankets to keep warm. In addition to her antibiotics, she was also given prednisone 70 mg for her respiratory symptoms.
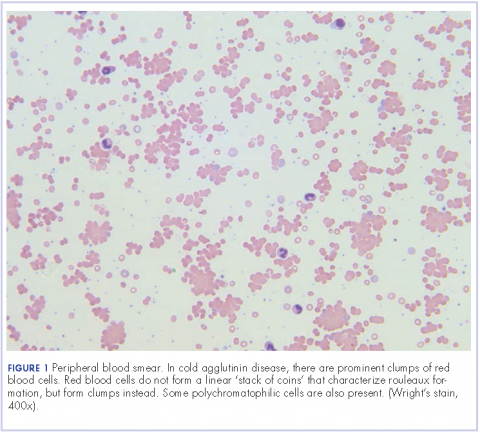
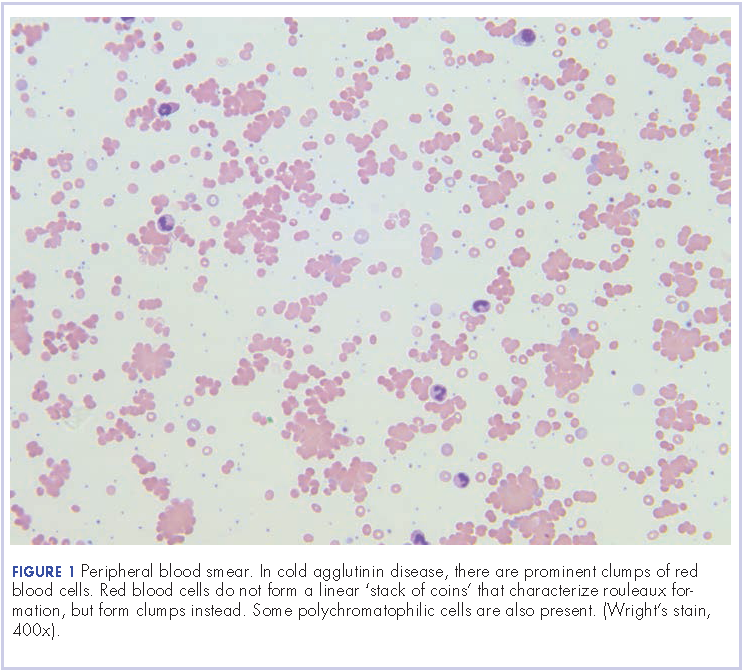
Further tests revealed haptoglobin, <30 mg/dL (normal, 36-195 mg/dL); lactate dehydrogenase, 371 U/L (normal, 98-192 U/L); and complements C3, 90 mg/dL (normal, 79-152 mg/dL) and C4, <8 mg/dL (normal, 18-55 mg/dL). Her DAT was positive, and agglutination was seen on peripheral smear (Figure 1). This was her second positive DAT as she had positive one at the outside hospital initially. Her tests for mycoplasma pneumonia, the PCR and IgM, were negative, as were the Monospot for mononucleosis and the ANA for autoimmune disorders. Her cold agglutinin titer was 1:256 (normal, no agglutination <1:64). The patient’s repeat respiratory viral panel was negative given recent full treatment for her influenza A at the previous hospital. Her blood and urine cultures were negative.
The patient was given antibiotics (vancomycin 1,250 mg IV Q12, cefepime 2 g IV Q8, and azithromycin 500 mg daily) for her pneumonia. Her respiratory status improved, and she was transferred to general medical floors after the first day of her admission. Her total bilirubin trended down to 1.9 mg/dL. She remained on prednisone 70 mg daily.
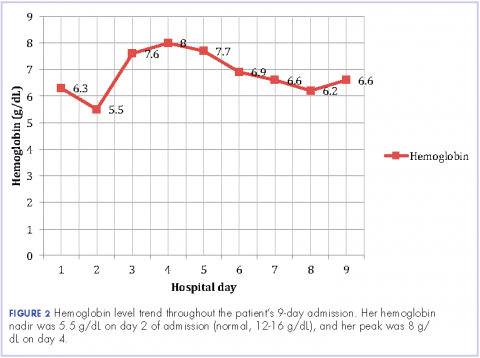
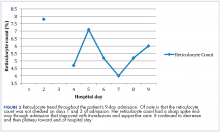
The patient remained in the hospital for an additional 6 days before being discharged home on prednisone. She wanted to return to her home country of Brazil as soon as she was able to and said she would seek outpatient follow-up there with a hematologist. At the time of her discharge, her hemoglobin was 6.6 g/dL and her reticulocyte count, 6.0%. Figures 2 and 3 illustrate her hemoglobin and reticulocyte trend during her admission at our hospital.
Discussion
The incidence of cold AIHA or cold agglutinin disease (CAD) occurs about 4 per 1 million people and commonly affects women more often than men.2 The cause of CAD can be subdivided into primary, idiopathic, or secondary causes, which can include infections, malignancies, or benign diseases.3,4 Primary CAD is a chronic disorder that is generally seen in older women. Secondary CAD can be associated with B-cell lymphoproliferative disorders, such as Waldenstrom macroglobulinemia or chronic lymphocytic leukemia, and infectious agents such as Mycoplasma pneumoniae and mononucleosis caused by Epstein-Barr virus.
Mild hemolysis or acrocyanosis may occur with exposure to cold. The blood smear in CAD demonstrates red blood cell agglutination or clumping, polychromasia, and an absence of spherocytosis. In general, most cases require no treatment, but cytotoxic agents or rituximab can be used to treat more severe cases. Appropriate treatment for infectious causes of CAD includes supportive care aimed at the underlying disease process. In addition, it is helpful to keep the patient warm. There is no role for steroid therapy in CAD unlike in warm AIHA. However, our patient was symptomatic from her pneumonia, so we added steroids to help with her pulmonary insult.
The patient had a cold agglutinin titer of 1:256. Titers of 1:32 or higher are considered elevated by this technique. Elevated titers are generally rarely seen except in primary atypical pneumonia due to either M. pneumoniae, influenza A, influenza B, parainfluenza, and adenovirus, and in certain hemolytic anemias. Low titers of cold agglutinins have been demonstrated in malaria, peripheral vascular disease, and common respiratory diseases.
Warm AIHA is caused by IgG antibody activities at body temperature or at 98.6°F. They may or may not bind complement and are removed from circulation by the spleen. Cold AIHA is due to IgM antibodies coating red cells at lower temperatures. They bind complement and lead to red blood cell destruction of agglutinated cells. If the antibody is active at temperatures approaching 98.6°F, clinically significant intravascular and sometimes extravascular complement-mediated hemolysis occur in the liver.5
The incidence of warm AIHA occurs about 10 per 1 million people and affects women twice often as men.2 It can be primary or idiopathic, or associated with various underlying conditions, including autoimmune disorders, immunodeficiency syndromes, lymphoproliferative disorders, other malignancies, and certain drugs. In more severe cases, jaundice and splenomegaly may occur. The blood smear in warm AIHA demonstrates variable spherocytosis, polychromasia, and rare erythrophagocytosis. Treatment usually includes steroids, cytotoxic agents, and splenectomy in severe cases.
There have been few case reports describing influenza as a cause of cold agglutinin hemolytic anemia. Chen and colleagues reported a case of influenza A infection in a 22-month-old boy.6Schoindre and colleagues reported the case of a 60-year-old woman infected with influenza A H1N1 virus who died from CAD.7 Shizuma reported the case of a 67-year-old man with alcoholic cirrhosis who developed a mixed hemolytic anemia and was positive for influenza A.8Our patient presented with influenza A, which had been diagnosed by respiratory virus panel at a different hospital, and she was anemic at the time of presentation to the outside hospital, with a positive DAT test. She was treated for influenza A with a full course of osltamivir and then returned with complaints of worsening fatigue and was again noted to be anemic with the development of patchy opacities on chest X-ray. The patient was subsequently transferred to our hospital and remained anemic during the course of her treatment. She received supportive care for her underlying influenza A and had symptomatic improvement. She ultimately decided the she would like to pursue further treatment in her native country and was discharged.
In conclusion, this case represents a rare complication of a common illness. Few cases of influenza causing hemolytic anemia have been reported in the literature. There have been reports of oseltamivir causing hemolytic anemia, but our patient presented with evidence of hemolytic anemia before initiation of the medication. In all the aforementioned cases, the patients died as a result of comorbid conditions. Our patient was stable enough to be discharged from the hospital after treatment of her comorbid conditions.
Acknowledgment
The authors thank David Henry, MD, at Pennsylvania Hospital, Philadelphia, for sharing this case and for his guidance during this patient’s treatment.
1. Roback JD, Grossman BJ, Harris T, Hillyer CD. Technical manual [17th ed]. Bethesda, MD; American Association of Blood Banks; 2011.
2. Jaffee ES, Harris NL, Vardiman JW, Campo E, Arber DA. Hematopathology. St. Louis, MO; Elsevier Saunders, 2011.
3. Feizi T. Monotypic cold agglutinins in infection by Mycoplasma pneumoniae. Nature. 1967;215(5100):540-542.
4. Horwitz CA, Moulds J, Henle W, et al. Cold agglutinins in infectious mononucleosis and heterophil-antibody-negative mononucleosis-like syndromes. Blood. 1977;50(2):195-202.
5. Hsi ED, editor. Hematopathology [3rd ed]. Philadelphia, PA; Elsevier Saunders; 2012.
6. Chen H, Jia XL, Gao HM, Qian SY. Comorbid presentation of severe novel influenza A (H1N1) and Evans syndrome: a case report. Chin Med J. 2011;124(11):1743-1746.
7. Schoindre Y, Bollée G, Dumont MD, Lesavre P, Servais A. Cold agglutinin syndrome associated with a 2009 influenza A H1N1 infection. http://www.amjmed.com/article/S0002-9343(10)00482-1/fulltext. Published February 2011. Accessed October 10, 2017.
8. [Article in Japanese] Shizuma T. [A case of autoimmune hemolytic anemia caused by type A influenza infection in a patient with alcoholic liver cirrhosis]. Kansenshogaku Zasshi. 2010;84(3):296-299.
Autoimmune hemolytic anemia (AIHA) is characterized by the temperature at which the auto-antibody has the greatest avidity for the target red cell antigen, either warm or cold forms. It is detected by a positive direct antiglobulin test (DAT) also known as the direct Coombs test. DAT is used to determine if red cells have been coated in vivo with immunoglobulin, complement, or both.1 Some causes of a positive DAT include hemolytic transfusion reactions, hemolytic disease of the fetus and newborn, AIHA, and drug-induced immune hemolysis.
Case presentation and summary
A 58-year-old woman from Brazil with past medical history only significant for cholecystectomy and cesarean section had been visiting in United States for 2 months when she presented to an outside hospital with fever, shortness of breath, and syncope that had resulted in a foot injury. She reported she had been feeling short of breath and had a nonproductive cough and malaise for about 2 weeks before presentation with sick contacts at home. On admission it was noted that she had a hemoglobin level of 7.7 g/dL (normal, 12.0-15.5 g/dL; MCV, 94 fL), total bilirubin of 2.14 mg/dL (normal, 0.2-1.0 mg/dL), and lactate dehydrogenase of 523 U/L (normal, 81-234 U/L). There were no signs of bleeding on her examination. Her DAT was positive and moderate red blood cell agglutination was reported. During the first admission at the outside hospital she was diagnosed with influenza A and completed a full course of oseltamivir (75 mg po twice daily for 5 days). A chest X-ray was negative for infiltrates and showed that the patient’s lung fields were clear. She was transfused 2 units of packed red blood cells with response in hemoglobin up to 9.8 g/dL. The patient was treated with dexamethasone (4 mg IV Q8) as an inpatient and was discharged on a prednisone taper (40 mg, with taper by 10 mg every 3 days) with hemoglobin of 8.1 g/dL.
The patient continued to have nonproductive cough, dyspnea, fevers, chills, and generalized weakness, when she returned to the same outside hospital’s emergency department 2 days after her discharge. At that time, it was noted that she had leucocytosis (white blood cell count, 34.6 x 109 per L), a hemoglobin level of 6.8 g/dL, and her total bilirubin level was 6.9 mg/dL. Her hemodynamics were unstable and she was admitted to their intensive care unit. The results of a chest X-ray revealed right lung consolidation.
The day after this admission, her hemoglobin level fell to 4.7 g/dL, and she was transfused 2 units of packed red blood cells before being transferred to our hospital. A chest X-ray at our hospital confirmed a right lung infiltrate. Vancomycin (1,250 mg IV Q12), levaquin (750 mg IV Q24), and maxipime (1 g IV Q12) were initiated for pneumonia and the patient was transferred to our hospital’s intensive care unit. She was afebrile at 98.3°F, her pulse rate was 84 beats per minute, she was tachypneic with respiratory rate of 26 breaths per minute, her blood pressure was 98/51 mmHg, and she had an oxygen saturation of 99% on 2L oxygen via nasal cannula.
On physical examination she was noted to have scleral icterus and was in mild respiratory distress. A chest X-ray revealed a patchy opacity in the right mid to lower lung. Her initial complete blood panel revealed anemia, with hemoglobin, 6.3 g/dL; white blood cell count, 27 x 109 per L; and platelets, 533 x 109 per L. The patient was then transfused another 2 units of packed red blood cells. She was given intravenous hydration, acetaminophen, and albuterol nebulizer treatments as supportive care. She was provided with blankets to keep warm. In addition to her antibiotics, she was also given prednisone 70 mg for her respiratory symptoms.
Further tests revealed haptoglobin, <30 mg/dL (normal, 36-195 mg/dL); lactate dehydrogenase, 371 U/L (normal, 98-192 U/L); and complements C3, 90 mg/dL (normal, 79-152 mg/dL) and C4, <8 mg/dL (normal, 18-55 mg/dL). Her DAT was positive, and agglutination was seen on peripheral smear (Figure 1). This was her second positive DAT as she had positive one at the outside hospital initially. Her tests for mycoplasma pneumonia, the PCR and IgM, were negative, as were the Monospot for mononucleosis and the ANA for autoimmune disorders. Her cold agglutinin titer was 1:256 (normal, no agglutination <1:64). The patient’s repeat respiratory viral panel was negative given recent full treatment for her influenza A at the previous hospital. Her blood and urine cultures were negative.
The patient was given antibiotics (vancomycin 1,250 mg IV Q12, cefepime 2 g IV Q8, and azithromycin 500 mg daily) for her pneumonia. Her respiratory status improved, and she was transferred to general medical floors after the first day of her admission. Her total bilirubin trended down to 1.9 mg/dL. She remained on prednisone 70 mg daily.
The patient remained in the hospital for an additional 6 days before being discharged home on prednisone. She wanted to return to her home country of Brazil as soon as she was able to and said she would seek outpatient follow-up there with a hematologist. At the time of her discharge, her hemoglobin was 6.6 g/dL and her reticulocyte count, 6.0%. Figures 2 and 3 illustrate her hemoglobin and reticulocyte trend during her admission at our hospital.
Discussion
The incidence of cold AIHA or cold agglutinin disease (CAD) occurs about 4 per 1 million people and commonly affects women more often than men.2 The cause of CAD can be subdivided into primary, idiopathic, or secondary causes, which can include infections, malignancies, or benign diseases.3,4 Primary CAD is a chronic disorder that is generally seen in older women. Secondary CAD can be associated with B-cell lymphoproliferative disorders, such as Waldenstrom macroglobulinemia or chronic lymphocytic leukemia, and infectious agents such as Mycoplasma pneumoniae and mononucleosis caused by Epstein-Barr virus.
Mild hemolysis or acrocyanosis may occur with exposure to cold. The blood smear in CAD demonstrates red blood cell agglutination or clumping, polychromasia, and an absence of spherocytosis. In general, most cases require no treatment, but cytotoxic agents or rituximab can be used to treat more severe cases. Appropriate treatment for infectious causes of CAD includes supportive care aimed at the underlying disease process. In addition, it is helpful to keep the patient warm. There is no role for steroid therapy in CAD unlike in warm AIHA. However, our patient was symptomatic from her pneumonia, so we added steroids to help with her pulmonary insult.
The patient had a cold agglutinin titer of 1:256. Titers of 1:32 or higher are considered elevated by this technique. Elevated titers are generally rarely seen except in primary atypical pneumonia due to either M. pneumoniae, influenza A, influenza B, parainfluenza, and adenovirus, and in certain hemolytic anemias. Low titers of cold agglutinins have been demonstrated in malaria, peripheral vascular disease, and common respiratory diseases.
Warm AIHA is caused by IgG antibody activities at body temperature or at 98.6°F. They may or may not bind complement and are removed from circulation by the spleen. Cold AIHA is due to IgM antibodies coating red cells at lower temperatures. They bind complement and lead to red blood cell destruction of agglutinated cells. If the antibody is active at temperatures approaching 98.6°F, clinically significant intravascular and sometimes extravascular complement-mediated hemolysis occur in the liver.5
The incidence of warm AIHA occurs about 10 per 1 million people and affects women twice often as men.2 It can be primary or idiopathic, or associated with various underlying conditions, including autoimmune disorders, immunodeficiency syndromes, lymphoproliferative disorders, other malignancies, and certain drugs. In more severe cases, jaundice and splenomegaly may occur. The blood smear in warm AIHA demonstrates variable spherocytosis, polychromasia, and rare erythrophagocytosis. Treatment usually includes steroids, cytotoxic agents, and splenectomy in severe cases.
There have been few case reports describing influenza as a cause of cold agglutinin hemolytic anemia. Chen and colleagues reported a case of influenza A infection in a 22-month-old boy.6Schoindre and colleagues reported the case of a 60-year-old woman infected with influenza A H1N1 virus who died from CAD.7 Shizuma reported the case of a 67-year-old man with alcoholic cirrhosis who developed a mixed hemolytic anemia and was positive for influenza A.8Our patient presented with influenza A, which had been diagnosed by respiratory virus panel at a different hospital, and she was anemic at the time of presentation to the outside hospital, with a positive DAT test. She was treated for influenza A with a full course of osltamivir and then returned with complaints of worsening fatigue and was again noted to be anemic with the development of patchy opacities on chest X-ray. The patient was subsequently transferred to our hospital and remained anemic during the course of her treatment. She received supportive care for her underlying influenza A and had symptomatic improvement. She ultimately decided the she would like to pursue further treatment in her native country and was discharged.
In conclusion, this case represents a rare complication of a common illness. Few cases of influenza causing hemolytic anemia have been reported in the literature. There have been reports of oseltamivir causing hemolytic anemia, but our patient presented with evidence of hemolytic anemia before initiation of the medication. In all the aforementioned cases, the patients died as a result of comorbid conditions. Our patient was stable enough to be discharged from the hospital after treatment of her comorbid conditions.
Acknowledgment
The authors thank David Henry, MD, at Pennsylvania Hospital, Philadelphia, for sharing this case and for his guidance during this patient’s treatment.
Autoimmune hemolytic anemia (AIHA) is characterized by the temperature at which the auto-antibody has the greatest avidity for the target red cell antigen, either warm or cold forms. It is detected by a positive direct antiglobulin test (DAT) also known as the direct Coombs test. DAT is used to determine if red cells have been coated in vivo with immunoglobulin, complement, or both.1 Some causes of a positive DAT include hemolytic transfusion reactions, hemolytic disease of the fetus and newborn, AIHA, and drug-induced immune hemolysis.
Case presentation and summary
A 58-year-old woman from Brazil with past medical history only significant for cholecystectomy and cesarean section had been visiting in United States for 2 months when she presented to an outside hospital with fever, shortness of breath, and syncope that had resulted in a foot injury. She reported she had been feeling short of breath and had a nonproductive cough and malaise for about 2 weeks before presentation with sick contacts at home. On admission it was noted that she had a hemoglobin level of 7.7 g/dL (normal, 12.0-15.5 g/dL; MCV, 94 fL), total bilirubin of 2.14 mg/dL (normal, 0.2-1.0 mg/dL), and lactate dehydrogenase of 523 U/L (normal, 81-234 U/L). There were no signs of bleeding on her examination. Her DAT was positive and moderate red blood cell agglutination was reported. During the first admission at the outside hospital she was diagnosed with influenza A and completed a full course of oseltamivir (75 mg po twice daily for 5 days). A chest X-ray was negative for infiltrates and showed that the patient’s lung fields were clear. She was transfused 2 units of packed red blood cells with response in hemoglobin up to 9.8 g/dL. The patient was treated with dexamethasone (4 mg IV Q8) as an inpatient and was discharged on a prednisone taper (40 mg, with taper by 10 mg every 3 days) with hemoglobin of 8.1 g/dL.
The patient continued to have nonproductive cough, dyspnea, fevers, chills, and generalized weakness, when she returned to the same outside hospital’s emergency department 2 days after her discharge. At that time, it was noted that she had leucocytosis (white blood cell count, 34.6 x 109 per L), a hemoglobin level of 6.8 g/dL, and her total bilirubin level was 6.9 mg/dL. Her hemodynamics were unstable and she was admitted to their intensive care unit. The results of a chest X-ray revealed right lung consolidation.
The day after this admission, her hemoglobin level fell to 4.7 g/dL, and she was transfused 2 units of packed red blood cells before being transferred to our hospital. A chest X-ray at our hospital confirmed a right lung infiltrate. Vancomycin (1,250 mg IV Q12), levaquin (750 mg IV Q24), and maxipime (1 g IV Q12) were initiated for pneumonia and the patient was transferred to our hospital’s intensive care unit. She was afebrile at 98.3°F, her pulse rate was 84 beats per minute, she was tachypneic with respiratory rate of 26 breaths per minute, her blood pressure was 98/51 mmHg, and she had an oxygen saturation of 99% on 2L oxygen via nasal cannula.
On physical examination she was noted to have scleral icterus and was in mild respiratory distress. A chest X-ray revealed a patchy opacity in the right mid to lower lung. Her initial complete blood panel revealed anemia, with hemoglobin, 6.3 g/dL; white blood cell count, 27 x 109 per L; and platelets, 533 x 109 per L. The patient was then transfused another 2 units of packed red blood cells. She was given intravenous hydration, acetaminophen, and albuterol nebulizer treatments as supportive care. She was provided with blankets to keep warm. In addition to her antibiotics, she was also given prednisone 70 mg for her respiratory symptoms.
Further tests revealed haptoglobin, <30 mg/dL (normal, 36-195 mg/dL); lactate dehydrogenase, 371 U/L (normal, 98-192 U/L); and complements C3, 90 mg/dL (normal, 79-152 mg/dL) and C4, <8 mg/dL (normal, 18-55 mg/dL). Her DAT was positive, and agglutination was seen on peripheral smear (Figure 1). This was her second positive DAT as she had positive one at the outside hospital initially. Her tests for mycoplasma pneumonia, the PCR and IgM, were negative, as were the Monospot for mononucleosis and the ANA for autoimmune disorders. Her cold agglutinin titer was 1:256 (normal, no agglutination <1:64). The patient’s repeat respiratory viral panel was negative given recent full treatment for her influenza A at the previous hospital. Her blood and urine cultures were negative.
The patient was given antibiotics (vancomycin 1,250 mg IV Q12, cefepime 2 g IV Q8, and azithromycin 500 mg daily) for her pneumonia. Her respiratory status improved, and she was transferred to general medical floors after the first day of her admission. Her total bilirubin trended down to 1.9 mg/dL. She remained on prednisone 70 mg daily.
The patient remained in the hospital for an additional 6 days before being discharged home on prednisone. She wanted to return to her home country of Brazil as soon as she was able to and said she would seek outpatient follow-up there with a hematologist. At the time of her discharge, her hemoglobin was 6.6 g/dL and her reticulocyte count, 6.0%. Figures 2 and 3 illustrate her hemoglobin and reticulocyte trend during her admission at our hospital.
Discussion
The incidence of cold AIHA or cold agglutinin disease (CAD) occurs about 4 per 1 million people and commonly affects women more often than men.2 The cause of CAD can be subdivided into primary, idiopathic, or secondary causes, which can include infections, malignancies, or benign diseases.3,4 Primary CAD is a chronic disorder that is generally seen in older women. Secondary CAD can be associated with B-cell lymphoproliferative disorders, such as Waldenstrom macroglobulinemia or chronic lymphocytic leukemia, and infectious agents such as Mycoplasma pneumoniae and mononucleosis caused by Epstein-Barr virus.
Mild hemolysis or acrocyanosis may occur with exposure to cold. The blood smear in CAD demonstrates red blood cell agglutination or clumping, polychromasia, and an absence of spherocytosis. In general, most cases require no treatment, but cytotoxic agents or rituximab can be used to treat more severe cases. Appropriate treatment for infectious causes of CAD includes supportive care aimed at the underlying disease process. In addition, it is helpful to keep the patient warm. There is no role for steroid therapy in CAD unlike in warm AIHA. However, our patient was symptomatic from her pneumonia, so we added steroids to help with her pulmonary insult.
The patient had a cold agglutinin titer of 1:256. Titers of 1:32 or higher are considered elevated by this technique. Elevated titers are generally rarely seen except in primary atypical pneumonia due to either M. pneumoniae, influenza A, influenza B, parainfluenza, and adenovirus, and in certain hemolytic anemias. Low titers of cold agglutinins have been demonstrated in malaria, peripheral vascular disease, and common respiratory diseases.
Warm AIHA is caused by IgG antibody activities at body temperature or at 98.6°F. They may or may not bind complement and are removed from circulation by the spleen. Cold AIHA is due to IgM antibodies coating red cells at lower temperatures. They bind complement and lead to red blood cell destruction of agglutinated cells. If the antibody is active at temperatures approaching 98.6°F, clinically significant intravascular and sometimes extravascular complement-mediated hemolysis occur in the liver.5
The incidence of warm AIHA occurs about 10 per 1 million people and affects women twice often as men.2 It can be primary or idiopathic, or associated with various underlying conditions, including autoimmune disorders, immunodeficiency syndromes, lymphoproliferative disorders, other malignancies, and certain drugs. In more severe cases, jaundice and splenomegaly may occur. The blood smear in warm AIHA demonstrates variable spherocytosis, polychromasia, and rare erythrophagocytosis. Treatment usually includes steroids, cytotoxic agents, and splenectomy in severe cases.
There have been few case reports describing influenza as a cause of cold agglutinin hemolytic anemia. Chen and colleagues reported a case of influenza A infection in a 22-month-old boy.6Schoindre and colleagues reported the case of a 60-year-old woman infected with influenza A H1N1 virus who died from CAD.7 Shizuma reported the case of a 67-year-old man with alcoholic cirrhosis who developed a mixed hemolytic anemia and was positive for influenza A.8Our patient presented with influenza A, which had been diagnosed by respiratory virus panel at a different hospital, and she was anemic at the time of presentation to the outside hospital, with a positive DAT test. She was treated for influenza A with a full course of osltamivir and then returned with complaints of worsening fatigue and was again noted to be anemic with the development of patchy opacities on chest X-ray. The patient was subsequently transferred to our hospital and remained anemic during the course of her treatment. She received supportive care for her underlying influenza A and had symptomatic improvement. She ultimately decided the she would like to pursue further treatment in her native country and was discharged.
In conclusion, this case represents a rare complication of a common illness. Few cases of influenza causing hemolytic anemia have been reported in the literature. There have been reports of oseltamivir causing hemolytic anemia, but our patient presented with evidence of hemolytic anemia before initiation of the medication. In all the aforementioned cases, the patients died as a result of comorbid conditions. Our patient was stable enough to be discharged from the hospital after treatment of her comorbid conditions.
Acknowledgment
The authors thank David Henry, MD, at Pennsylvania Hospital, Philadelphia, for sharing this case and for his guidance during this patient’s treatment.
1. Roback JD, Grossman BJ, Harris T, Hillyer CD. Technical manual [17th ed]. Bethesda, MD; American Association of Blood Banks; 2011.
2. Jaffee ES, Harris NL, Vardiman JW, Campo E, Arber DA. Hematopathology. St. Louis, MO; Elsevier Saunders, 2011.
3. Feizi T. Monotypic cold agglutinins in infection by Mycoplasma pneumoniae. Nature. 1967;215(5100):540-542.
4. Horwitz CA, Moulds J, Henle W, et al. Cold agglutinins in infectious mononucleosis and heterophil-antibody-negative mononucleosis-like syndromes. Blood. 1977;50(2):195-202.
5. Hsi ED, editor. Hematopathology [3rd ed]. Philadelphia, PA; Elsevier Saunders; 2012.
6. Chen H, Jia XL, Gao HM, Qian SY. Comorbid presentation of severe novel influenza A (H1N1) and Evans syndrome: a case report. Chin Med J. 2011;124(11):1743-1746.
7. Schoindre Y, Bollée G, Dumont MD, Lesavre P, Servais A. Cold agglutinin syndrome associated with a 2009 influenza A H1N1 infection. http://www.amjmed.com/article/S0002-9343(10)00482-1/fulltext. Published February 2011. Accessed October 10, 2017.
8. [Article in Japanese] Shizuma T. [A case of autoimmune hemolytic anemia caused by type A influenza infection in a patient with alcoholic liver cirrhosis]. Kansenshogaku Zasshi. 2010;84(3):296-299.
1. Roback JD, Grossman BJ, Harris T, Hillyer CD. Technical manual [17th ed]. Bethesda, MD; American Association of Blood Banks; 2011.
2. Jaffee ES, Harris NL, Vardiman JW, Campo E, Arber DA. Hematopathology. St. Louis, MO; Elsevier Saunders, 2011.
3. Feizi T. Monotypic cold agglutinins in infection by Mycoplasma pneumoniae. Nature. 1967;215(5100):540-542.
4. Horwitz CA, Moulds J, Henle W, et al. Cold agglutinins in infectious mononucleosis and heterophil-antibody-negative mononucleosis-like syndromes. Blood. 1977;50(2):195-202.
5. Hsi ED, editor. Hematopathology [3rd ed]. Philadelphia, PA; Elsevier Saunders; 2012.
6. Chen H, Jia XL, Gao HM, Qian SY. Comorbid presentation of severe novel influenza A (H1N1) and Evans syndrome: a case report. Chin Med J. 2011;124(11):1743-1746.
7. Schoindre Y, Bollée G, Dumont MD, Lesavre P, Servais A. Cold agglutinin syndrome associated with a 2009 influenza A H1N1 infection. http://www.amjmed.com/article/S0002-9343(10)00482-1/fulltext. Published February 2011. Accessed October 10, 2017.
8. [Article in Japanese] Shizuma T. [A case of autoimmune hemolytic anemia caused by type A influenza infection in a patient with alcoholic liver cirrhosis]. Kansenshogaku Zasshi. 2010;84(3):296-299.
Physician-assisted suicide – an update
Question: Choose the best answer regarding physician-assisted suicide in the United States:
A. It is now legal in most states.
B. Under California law, assisting or causing one to commit suicide, including physician-assisted suicide, still remains a felony.
C. Both the U.S. Supreme Court and the New York Court of Appeals have held there is no constitutional right to physician-assisted suicide.
D. The American Medical Association is neutral on the issue.
E. Pain relief is the overriding reason for patients who request physician-assisted suicide.
Answer: C. We reviewed this topic in one of our regular columns in 2013.1 At that time, efforts to legalize physician-assisted suicide (PAS) appeared to be gathering momentum across the country, with four jurisdictions having legalized the practice, beginning with Oregon in 1994. The other states were Washington, Vermont, and Montana, whose Supreme Court held that there was no public interest reason against the practice.2
Since that time, California, Colorado, and the District of Columbia have joined the group. Currently, PAS – but not euthanasia – is legally available in these jurisdictions and in Switzerland, but both can be legally practiced in Belgium, Canada, Colombia, Luxembourg, and the Netherlands.
All state statutes permitting PAS provide similar provisions and safeguards. Only competent individuals who are terminally ill, i.e., death expected within 6 months, can make a request for a lethal dose of medication to carry out the suicidal act. The request to the doctor is first made verbally, then in writing, and a second opinion must be obtained to confirm the patient’s intent, understanding, and free choice. There is also a waiting period.
Public support for euthanasia and PAS in the United States is said to have plateaued since the 1990s. But a significant number of Americans, 67%, still favor PAS, up from 56% a decade ago.3 However, not many patients resort to PAS – usually those with terminal cancers or neuromuscular conditions – and only a minority of physicians are participants.
For example, 61 physicians in Oregon wrote a total of 115 prescriptions in 2012; there were 77 known Death With Dignity Act deaths in Oregon that year.4 In Oregon and Washington State, less than 1% of licensed physicians write prescriptions for physician-assisted suicide each year. In contrast, about half or more of physicians in the Netherlands and Belgium reported ever having received a request, and 60% of Dutch physicians have granted such requests.
The California Department of Public Health reported that 111 terminally ill patients availed themselves of California’s End of Life Option Act in the 7 months after it became effective on June 9, 2016.
In a recent review on euthanasia and PAS for the period 1947-2016, Ezekiel Emanuel, MD, and colleagues noted that typical patients were older, white, and well educated, and pain was mostly not reported as the primary motivation.5 A large portion of patients receiving PAS in Oregon and Washington were enrolled in hospice or palliative care. Abuses have not been apparent.
In the vast majority of jurisdictions, assisting or causing one to commit suicide, including PAS, still remains a crime; for example, it is considered manslaughter under Hawaii state law §707-702.
In distinguishing between assisting suicide and withdrawing life-sustaining treatment, the U.S. Supreme Court’s landmark 1997 Vacco v. Quill decision emphasized issues of causation and intent.6 On causation, the court reasoned that when a patient refuses life-sustaining treatment, he dies from an underlying fatal disease; but if a patient ingests a lethal medication, he is killed by that medication. As to intent, a physician who honors a patient’s refusal of treatment purposefully intends only to respect his patient’s wishes and to cease doing futile or degrading things. On the other hand, a doctor who assists a suicide “must, necessarily and indubitably, intend primarily that the patient be made dead.”
In its companion case Washington v. Glucksberg, the Supreme Court held that the asserted “right” to assistance in committing suicide is not a fundamental liberty interest protected by the due process clause.7
State supreme courts in Florida, New Mexico, and elsewhere have likewise rebuffed claims of any constitutional right to PAS. The latest court to so rule is in New York, which has a long history of criminalizing assisted suicide.8 The New York Court of Appeals recently addressed claims brought by three terminally ill individuals, several medical providers, and a nonprofit entity seeking a declaration that New York’s “assisted suicide” statutes exclude physicians from prescribing a lethal dose of drugs to terminally ill, competent patients.
The court unequivocally rejected such claims and affirmed that a physician who assists a suicide by prescribing lethal doses of drugs is subject to criminal prosecution for second-degree manslaughter. It refused to regard PAS as being different from assisted suicide in general, and it rejected the constitutional claim to assisted suicide by a terminally ill person. The state appeals court reiterated the U.S. Supreme Court’s distinction between refusing life-sustaining treatment and assisted suicide, the former being “at least partially rooted in notions of bodily integrity, as the right to refuse treatment is a consequence of a person’s right to resist unwanted bodily invasions.” The New York Court of Appeals also noted that the state has a legitimate purpose and a rational basis for guarding against the risks of mistake and abuse.
These developments may signal a shift away from the legalization of PAS, as recently suggested in a Washington Post article.9 According to the end-of-life advocacy organization Compassion and Choices, none of the 27 states where such measures were introduced in 2017 passed them into law, including states such as Connecticut, Hawaii, and Rhode Island. In Central and Eastern Europe, support is decreasing, whereas the opposite is true in Western Europe.
U.S. federal lawmakers also appear to be pushing back. On July 13, 2017, the U.S. House Committee on Appropriations voted to block implementation of a “death with dignity” statute passed by the District of Columbia. Further, 11 House members – including 6 Democrats – have introduced a resolution asserting that PAS undermines a key safeguard that protects our nation’s most vulnerable citizens, including the elderly, people with disabilities, and people experiencing psychiatric diagnoses.10
The American Medical Association is steadfast in its opposition to PAS and euthanasia. In its latest Code of Ethics, the AMA reaffirmed its long-held position that “allowing physicians to engage in assisted suicide would cause more harm than good. Physician-assisted suicide is fundamentally incompatible with the physician’s role as healer, would be difficult or impossible to control, and would pose serious societal risks. … Instead of participating in assisted suicide, physicians must aggressively respond to the needs of patients at the end of life.”11
References
1. “Physician-assisted suicide,” Internal Medicine News, Oct. 14, 2013.
2. Baxter v. State of Montana, 224 P. 3d 1211 (2010).
3. “Majority of Americans Remain Supportive of Euthanasia,” Gallup News, June 12, 2017.
4. Statistics available at public.health.oregon.gov under Oregon Death with Dignity Act.
5. JAMA. 2016 Jul 5;316(1):79-90.
6. Vacco v. Quill, 117 S. Ct. 2293 (1997).
7. Washington v. Glucksberg, 521 U.S. 702 (1997).
8. Myers v. Schneiderman, New York Court of Appeals, 2017.
9. “Legalizing assisted suicide has stalled at every level,” Washington Post, Oct. 24, 2017.
10. H. Con. Res. 80, 115th Congress (2017-2018).
11. AMA Code of Medical Ethics §5.7 (2017).
Question: Choose the best answer regarding physician-assisted suicide in the United States:
A. It is now legal in most states.
B. Under California law, assisting or causing one to commit suicide, including physician-assisted suicide, still remains a felony.
C. Both the U.S. Supreme Court and the New York Court of Appeals have held there is no constitutional right to physician-assisted suicide.
D. The American Medical Association is neutral on the issue.
E. Pain relief is the overriding reason for patients who request physician-assisted suicide.
Answer: C. We reviewed this topic in one of our regular columns in 2013.1 At that time, efforts to legalize physician-assisted suicide (PAS) appeared to be gathering momentum across the country, with four jurisdictions having legalized the practice, beginning with Oregon in 1994. The other states were Washington, Vermont, and Montana, whose Supreme Court held that there was no public interest reason against the practice.2
Since that time, California, Colorado, and the District of Columbia have joined the group. Currently, PAS – but not euthanasia – is legally available in these jurisdictions and in Switzerland, but both can be legally practiced in Belgium, Canada, Colombia, Luxembourg, and the Netherlands.
All state statutes permitting PAS provide similar provisions and safeguards. Only competent individuals who are terminally ill, i.e., death expected within 6 months, can make a request for a lethal dose of medication to carry out the suicidal act. The request to the doctor is first made verbally, then in writing, and a second opinion must be obtained to confirm the patient’s intent, understanding, and free choice. There is also a waiting period.
Public support for euthanasia and PAS in the United States is said to have plateaued since the 1990s. But a significant number of Americans, 67%, still favor PAS, up from 56% a decade ago.3 However, not many patients resort to PAS – usually those with terminal cancers or neuromuscular conditions – and only a minority of physicians are participants.
For example, 61 physicians in Oregon wrote a total of 115 prescriptions in 2012; there were 77 known Death With Dignity Act deaths in Oregon that year.4 In Oregon and Washington State, less than 1% of licensed physicians write prescriptions for physician-assisted suicide each year. In contrast, about half or more of physicians in the Netherlands and Belgium reported ever having received a request, and 60% of Dutch physicians have granted such requests.
The California Department of Public Health reported that 111 terminally ill patients availed themselves of California’s End of Life Option Act in the 7 months after it became effective on June 9, 2016.
In a recent review on euthanasia and PAS for the period 1947-2016, Ezekiel Emanuel, MD, and colleagues noted that typical patients were older, white, and well educated, and pain was mostly not reported as the primary motivation.5 A large portion of patients receiving PAS in Oregon and Washington were enrolled in hospice or palliative care. Abuses have not been apparent.
In the vast majority of jurisdictions, assisting or causing one to commit suicide, including PAS, still remains a crime; for example, it is considered manslaughter under Hawaii state law §707-702.
In distinguishing between assisting suicide and withdrawing life-sustaining treatment, the U.S. Supreme Court’s landmark 1997 Vacco v. Quill decision emphasized issues of causation and intent.6 On causation, the court reasoned that when a patient refuses life-sustaining treatment, he dies from an underlying fatal disease; but if a patient ingests a lethal medication, he is killed by that medication. As to intent, a physician who honors a patient’s refusal of treatment purposefully intends only to respect his patient’s wishes and to cease doing futile or degrading things. On the other hand, a doctor who assists a suicide “must, necessarily and indubitably, intend primarily that the patient be made dead.”
In its companion case Washington v. Glucksberg, the Supreme Court held that the asserted “right” to assistance in committing suicide is not a fundamental liberty interest protected by the due process clause.7
State supreme courts in Florida, New Mexico, and elsewhere have likewise rebuffed claims of any constitutional right to PAS. The latest court to so rule is in New York, which has a long history of criminalizing assisted suicide.8 The New York Court of Appeals recently addressed claims brought by three terminally ill individuals, several medical providers, and a nonprofit entity seeking a declaration that New York’s “assisted suicide” statutes exclude physicians from prescribing a lethal dose of drugs to terminally ill, competent patients.
The court unequivocally rejected such claims and affirmed that a physician who assists a suicide by prescribing lethal doses of drugs is subject to criminal prosecution for second-degree manslaughter. It refused to regard PAS as being different from assisted suicide in general, and it rejected the constitutional claim to assisted suicide by a terminally ill person. The state appeals court reiterated the U.S. Supreme Court’s distinction between refusing life-sustaining treatment and assisted suicide, the former being “at least partially rooted in notions of bodily integrity, as the right to refuse treatment is a consequence of a person’s right to resist unwanted bodily invasions.” The New York Court of Appeals also noted that the state has a legitimate purpose and a rational basis for guarding against the risks of mistake and abuse.
These developments may signal a shift away from the legalization of PAS, as recently suggested in a Washington Post article.9 According to the end-of-life advocacy organization Compassion and Choices, none of the 27 states where such measures were introduced in 2017 passed them into law, including states such as Connecticut, Hawaii, and Rhode Island. In Central and Eastern Europe, support is decreasing, whereas the opposite is true in Western Europe.
U.S. federal lawmakers also appear to be pushing back. On July 13, 2017, the U.S. House Committee on Appropriations voted to block implementation of a “death with dignity” statute passed by the District of Columbia. Further, 11 House members – including 6 Democrats – have introduced a resolution asserting that PAS undermines a key safeguard that protects our nation’s most vulnerable citizens, including the elderly, people with disabilities, and people experiencing psychiatric diagnoses.10
The American Medical Association is steadfast in its opposition to PAS and euthanasia. In its latest Code of Ethics, the AMA reaffirmed its long-held position that “allowing physicians to engage in assisted suicide would cause more harm than good. Physician-assisted suicide is fundamentally incompatible with the physician’s role as healer, would be difficult or impossible to control, and would pose serious societal risks. … Instead of participating in assisted suicide, physicians must aggressively respond to the needs of patients at the end of life.”11
References
1. “Physician-assisted suicide,” Internal Medicine News, Oct. 14, 2013.
2. Baxter v. State of Montana, 224 P. 3d 1211 (2010).
3. “Majority of Americans Remain Supportive of Euthanasia,” Gallup News, June 12, 2017.
4. Statistics available at public.health.oregon.gov under Oregon Death with Dignity Act.
5. JAMA. 2016 Jul 5;316(1):79-90.
6. Vacco v. Quill, 117 S. Ct. 2293 (1997).
7. Washington v. Glucksberg, 521 U.S. 702 (1997).
8. Myers v. Schneiderman, New York Court of Appeals, 2017.
9. “Legalizing assisted suicide has stalled at every level,” Washington Post, Oct. 24, 2017.
10. H. Con. Res. 80, 115th Congress (2017-2018).
11. AMA Code of Medical Ethics §5.7 (2017).
Question: Choose the best answer regarding physician-assisted suicide in the United States:
A. It is now legal in most states.
B. Under California law, assisting or causing one to commit suicide, including physician-assisted suicide, still remains a felony.
C. Both the U.S. Supreme Court and the New York Court of Appeals have held there is no constitutional right to physician-assisted suicide.
D. The American Medical Association is neutral on the issue.
E. Pain relief is the overriding reason for patients who request physician-assisted suicide.
Answer: C. We reviewed this topic in one of our regular columns in 2013.1 At that time, efforts to legalize physician-assisted suicide (PAS) appeared to be gathering momentum across the country, with four jurisdictions having legalized the practice, beginning with Oregon in 1994. The other states were Washington, Vermont, and Montana, whose Supreme Court held that there was no public interest reason against the practice.2
Since that time, California, Colorado, and the District of Columbia have joined the group. Currently, PAS – but not euthanasia – is legally available in these jurisdictions and in Switzerland, but both can be legally practiced in Belgium, Canada, Colombia, Luxembourg, and the Netherlands.
All state statutes permitting PAS provide similar provisions and safeguards. Only competent individuals who are terminally ill, i.e., death expected within 6 months, can make a request for a lethal dose of medication to carry out the suicidal act. The request to the doctor is first made verbally, then in writing, and a second opinion must be obtained to confirm the patient’s intent, understanding, and free choice. There is also a waiting period.
Public support for euthanasia and PAS in the United States is said to have plateaued since the 1990s. But a significant number of Americans, 67%, still favor PAS, up from 56% a decade ago.3 However, not many patients resort to PAS – usually those with terminal cancers or neuromuscular conditions – and only a minority of physicians are participants.
For example, 61 physicians in Oregon wrote a total of 115 prescriptions in 2012; there were 77 known Death With Dignity Act deaths in Oregon that year.4 In Oregon and Washington State, less than 1% of licensed physicians write prescriptions for physician-assisted suicide each year. In contrast, about half or more of physicians in the Netherlands and Belgium reported ever having received a request, and 60% of Dutch physicians have granted such requests.
The California Department of Public Health reported that 111 terminally ill patients availed themselves of California’s End of Life Option Act in the 7 months after it became effective on June 9, 2016.
In a recent review on euthanasia and PAS for the period 1947-2016, Ezekiel Emanuel, MD, and colleagues noted that typical patients were older, white, and well educated, and pain was mostly not reported as the primary motivation.5 A large portion of patients receiving PAS in Oregon and Washington were enrolled in hospice or palliative care. Abuses have not been apparent.
In the vast majority of jurisdictions, assisting or causing one to commit suicide, including PAS, still remains a crime; for example, it is considered manslaughter under Hawaii state law §707-702.
In distinguishing between assisting suicide and withdrawing life-sustaining treatment, the U.S. Supreme Court’s landmark 1997 Vacco v. Quill decision emphasized issues of causation and intent.6 On causation, the court reasoned that when a patient refuses life-sustaining treatment, he dies from an underlying fatal disease; but if a patient ingests a lethal medication, he is killed by that medication. As to intent, a physician who honors a patient’s refusal of treatment purposefully intends only to respect his patient’s wishes and to cease doing futile or degrading things. On the other hand, a doctor who assists a suicide “must, necessarily and indubitably, intend primarily that the patient be made dead.”
In its companion case Washington v. Glucksberg, the Supreme Court held that the asserted “right” to assistance in committing suicide is not a fundamental liberty interest protected by the due process clause.7
State supreme courts in Florida, New Mexico, and elsewhere have likewise rebuffed claims of any constitutional right to PAS. The latest court to so rule is in New York, which has a long history of criminalizing assisted suicide.8 The New York Court of Appeals recently addressed claims brought by three terminally ill individuals, several medical providers, and a nonprofit entity seeking a declaration that New York’s “assisted suicide” statutes exclude physicians from prescribing a lethal dose of drugs to terminally ill, competent patients.
The court unequivocally rejected such claims and affirmed that a physician who assists a suicide by prescribing lethal doses of drugs is subject to criminal prosecution for second-degree manslaughter. It refused to regard PAS as being different from assisted suicide in general, and it rejected the constitutional claim to assisted suicide by a terminally ill person. The state appeals court reiterated the U.S. Supreme Court’s distinction between refusing life-sustaining treatment and assisted suicide, the former being “at least partially rooted in notions of bodily integrity, as the right to refuse treatment is a consequence of a person’s right to resist unwanted bodily invasions.” The New York Court of Appeals also noted that the state has a legitimate purpose and a rational basis for guarding against the risks of mistake and abuse.
These developments may signal a shift away from the legalization of PAS, as recently suggested in a Washington Post article.9 According to the end-of-life advocacy organization Compassion and Choices, none of the 27 states where such measures were introduced in 2017 passed them into law, including states such as Connecticut, Hawaii, and Rhode Island. In Central and Eastern Europe, support is decreasing, whereas the opposite is true in Western Europe.
U.S. federal lawmakers also appear to be pushing back. On July 13, 2017, the U.S. House Committee on Appropriations voted to block implementation of a “death with dignity” statute passed by the District of Columbia. Further, 11 House members – including 6 Democrats – have introduced a resolution asserting that PAS undermines a key safeguard that protects our nation’s most vulnerable citizens, including the elderly, people with disabilities, and people experiencing psychiatric diagnoses.10
The American Medical Association is steadfast in its opposition to PAS and euthanasia. In its latest Code of Ethics, the AMA reaffirmed its long-held position that “allowing physicians to engage in assisted suicide would cause more harm than good. Physician-assisted suicide is fundamentally incompatible with the physician’s role as healer, would be difficult or impossible to control, and would pose serious societal risks. … Instead of participating in assisted suicide, physicians must aggressively respond to the needs of patients at the end of life.”11
References
1. “Physician-assisted suicide,” Internal Medicine News, Oct. 14, 2013.
2. Baxter v. State of Montana, 224 P. 3d 1211 (2010).
3. “Majority of Americans Remain Supportive of Euthanasia,” Gallup News, June 12, 2017.
4. Statistics available at public.health.oregon.gov under Oregon Death with Dignity Act.
5. JAMA. 2016 Jul 5;316(1):79-90.
6. Vacco v. Quill, 117 S. Ct. 2293 (1997).
7. Washington v. Glucksberg, 521 U.S. 702 (1997).
8. Myers v. Schneiderman, New York Court of Appeals, 2017.
9. “Legalizing assisted suicide has stalled at every level,” Washington Post, Oct. 24, 2017.
10. H. Con. Res. 80, 115th Congress (2017-2018).
11. AMA Code of Medical Ethics §5.7 (2017).
Edoxaban noninferior to dalteparin for cancer-associated VTE
ATLANTA – Twelve months of daily treatment with the novel oral factor Xa inhibitor edoxaban was noninferior to standard subcutaneous therapy with dalteparin for treatment of venous thromboembolism in patients with cancer, according to late-breaking results from a randomized, open-label, blinded-outcomes trial.
Throughout follow-up, trial arms had nearly identical rates of survival free from recurrent VTE or major bleeding, Gary E. Raskob, PhD, reported during a late-breaking oral presentation at the annual meeting of the American Society of Hematology. “Edoxaban was associated with a lower rate of recurrent VTE, which was offset by a similar increase in risk of major bleeding,” he said. “Therefore, oral edoxaban was noninferior to subcutaneous dalteparin for the [combined] primary outcome.”
Venous thromboembolism affects about one in five patients with active cancer and is difficult to treat because patients face increased risks of recurrence and bleeding. The struggle to balance these risks fuels morbidity and mortality and can hamper cancer treatment, said Dr. Raskob of the University of Oklahoma, Oklahoma City.
Pharmacy and medical oncology societies recommend long-term low-molecular-weight heparin for cancer patients with VTE, but the daily burden of subcutaneous injections leads many to stop after about 2-4 months of treatment, Dr. Raskob said. “Direct oral anticoagulants may be an attractive alternative.”
For the trial, 1,446 adults with cancer and lower limb VTE from 114 clinics in North America, Europe, Australia, and New Zealand received either edoxaban (60 mg daily) or dalteparin (200 IU/kg for 30 days, followed by 150 IU/kg) for up to 12 months. Nearly all patients had active cancer. Tumor types reflected what’s most common in practice, such as malignancies of the lung, colon, and breast. About 50 patients had primary or metastatic brain cancers. Approximately two-thirds had pulmonary embolism with or without deep-vein thrombosis, while the rest had isolated deep-vein thrombosis.
After 12 months of follow-up, 12.8% of edoxaban patients had at least one recurrence of VTE or a major bleed, compared with 13.5% of dalteparin patients (hazard ratio, 0.97; 95% confidence interval, 0.70-1.36; P = .006 for noninferiority). Edoxaban also was noninferior to dalteparin after the first 6 months of treatment and in the per-protocol analysis (HRs, 1.0; P = .02 for noninferiority in each analysis). Thus, differences in efficacy did not only reflect better compliance to oral therapy, Dr. Raskob said.
He also reported on individual outcomes. In all, 10.3% of dalteparin recipients had a VTE recurrence, as did 6.5% of edoxaban recipients, for a risk difference of 3.8% (95% CI, 7.1%-0.4%). More than half of recurrences in each group were symptomatic, and none were fatal. Bleeding caused no deaths in either study arm, and each therapy conferred an identical chance of a grade 3-4 major bleed (2.3%).
Edoxaban was associated, however, with a greater frequency of major bleeds (33 events; 6.3%) than was dalteparin (17 events; 3.2%; risk difference, 3.1%; 95% CI, 0.5%-5.7%). In particular, patients who received edoxaban had a slightly higher rate of upper gastrointestinal bleeds. Most had gastric cancer.
Future studies should evaluate whether these patients should receive a lower dose of edoxaban, said Dr. Raskob. “We don’t yet fully know the minimum effective dose [of edoxaban] in cancer patients.”
He also addressed the idea that heparin has antineoplastic activity, calling it “one we should probably abandon. The concept originates from older trials in which researchers probably did not recognize that heparin was preventing fatal pulmonary embolism, he said.
The investigators soon will begin deeper analyses that should inform patient selection, he said. For now, he recommends discussing these findings with patients to help them make an informed choice between oral anticoagulation, with its ease of use but slightly higher rate of major bleeds, and subcutaneous heparin, with its lower bleeding rate and treatment burden.
Daiichi Sankyo provided funding. Dr. Raskob disclosed consulting relationships and honoraria from Daiichi Sankyo, Eli Lilly, Janssen, and several other pharmaceutical companies.
SOURCE: Raskob G et al. ASH Abstract LBA-6.
ATLANTA – Twelve months of daily treatment with the novel oral factor Xa inhibitor edoxaban was noninferior to standard subcutaneous therapy with dalteparin for treatment of venous thromboembolism in patients with cancer, according to late-breaking results from a randomized, open-label, blinded-outcomes trial.
Throughout follow-up, trial arms had nearly identical rates of survival free from recurrent VTE or major bleeding, Gary E. Raskob, PhD, reported during a late-breaking oral presentation at the annual meeting of the American Society of Hematology. “Edoxaban was associated with a lower rate of recurrent VTE, which was offset by a similar increase in risk of major bleeding,” he said. “Therefore, oral edoxaban was noninferior to subcutaneous dalteparin for the [combined] primary outcome.”
Venous thromboembolism affects about one in five patients with active cancer and is difficult to treat because patients face increased risks of recurrence and bleeding. The struggle to balance these risks fuels morbidity and mortality and can hamper cancer treatment, said Dr. Raskob of the University of Oklahoma, Oklahoma City.
Pharmacy and medical oncology societies recommend long-term low-molecular-weight heparin for cancer patients with VTE, but the daily burden of subcutaneous injections leads many to stop after about 2-4 months of treatment, Dr. Raskob said. “Direct oral anticoagulants may be an attractive alternative.”
For the trial, 1,446 adults with cancer and lower limb VTE from 114 clinics in North America, Europe, Australia, and New Zealand received either edoxaban (60 mg daily) or dalteparin (200 IU/kg for 30 days, followed by 150 IU/kg) for up to 12 months. Nearly all patients had active cancer. Tumor types reflected what’s most common in practice, such as malignancies of the lung, colon, and breast. About 50 patients had primary or metastatic brain cancers. Approximately two-thirds had pulmonary embolism with or without deep-vein thrombosis, while the rest had isolated deep-vein thrombosis.
After 12 months of follow-up, 12.8% of edoxaban patients had at least one recurrence of VTE or a major bleed, compared with 13.5% of dalteparin patients (hazard ratio, 0.97; 95% confidence interval, 0.70-1.36; P = .006 for noninferiority). Edoxaban also was noninferior to dalteparin after the first 6 months of treatment and in the per-protocol analysis (HRs, 1.0; P = .02 for noninferiority in each analysis). Thus, differences in efficacy did not only reflect better compliance to oral therapy, Dr. Raskob said.
He also reported on individual outcomes. In all, 10.3% of dalteparin recipients had a VTE recurrence, as did 6.5% of edoxaban recipients, for a risk difference of 3.8% (95% CI, 7.1%-0.4%). More than half of recurrences in each group were symptomatic, and none were fatal. Bleeding caused no deaths in either study arm, and each therapy conferred an identical chance of a grade 3-4 major bleed (2.3%).
Edoxaban was associated, however, with a greater frequency of major bleeds (33 events; 6.3%) than was dalteparin (17 events; 3.2%; risk difference, 3.1%; 95% CI, 0.5%-5.7%). In particular, patients who received edoxaban had a slightly higher rate of upper gastrointestinal bleeds. Most had gastric cancer.
Future studies should evaluate whether these patients should receive a lower dose of edoxaban, said Dr. Raskob. “We don’t yet fully know the minimum effective dose [of edoxaban] in cancer patients.”
He also addressed the idea that heparin has antineoplastic activity, calling it “one we should probably abandon. The concept originates from older trials in which researchers probably did not recognize that heparin was preventing fatal pulmonary embolism, he said.
The investigators soon will begin deeper analyses that should inform patient selection, he said. For now, he recommends discussing these findings with patients to help them make an informed choice between oral anticoagulation, with its ease of use but slightly higher rate of major bleeds, and subcutaneous heparin, with its lower bleeding rate and treatment burden.
Daiichi Sankyo provided funding. Dr. Raskob disclosed consulting relationships and honoraria from Daiichi Sankyo, Eli Lilly, Janssen, and several other pharmaceutical companies.
SOURCE: Raskob G et al. ASH Abstract LBA-6.
ATLANTA – Twelve months of daily treatment with the novel oral factor Xa inhibitor edoxaban was noninferior to standard subcutaneous therapy with dalteparin for treatment of venous thromboembolism in patients with cancer, according to late-breaking results from a randomized, open-label, blinded-outcomes trial.
Throughout follow-up, trial arms had nearly identical rates of survival free from recurrent VTE or major bleeding, Gary E. Raskob, PhD, reported during a late-breaking oral presentation at the annual meeting of the American Society of Hematology. “Edoxaban was associated with a lower rate of recurrent VTE, which was offset by a similar increase in risk of major bleeding,” he said. “Therefore, oral edoxaban was noninferior to subcutaneous dalteparin for the [combined] primary outcome.”
Venous thromboembolism affects about one in five patients with active cancer and is difficult to treat because patients face increased risks of recurrence and bleeding. The struggle to balance these risks fuels morbidity and mortality and can hamper cancer treatment, said Dr. Raskob of the University of Oklahoma, Oklahoma City.
Pharmacy and medical oncology societies recommend long-term low-molecular-weight heparin for cancer patients with VTE, but the daily burden of subcutaneous injections leads many to stop after about 2-4 months of treatment, Dr. Raskob said. “Direct oral anticoagulants may be an attractive alternative.”
For the trial, 1,446 adults with cancer and lower limb VTE from 114 clinics in North America, Europe, Australia, and New Zealand received either edoxaban (60 mg daily) or dalteparin (200 IU/kg for 30 days, followed by 150 IU/kg) for up to 12 months. Nearly all patients had active cancer. Tumor types reflected what’s most common in practice, such as malignancies of the lung, colon, and breast. About 50 patients had primary or metastatic brain cancers. Approximately two-thirds had pulmonary embolism with or without deep-vein thrombosis, while the rest had isolated deep-vein thrombosis.
After 12 months of follow-up, 12.8% of edoxaban patients had at least one recurrence of VTE or a major bleed, compared with 13.5% of dalteparin patients (hazard ratio, 0.97; 95% confidence interval, 0.70-1.36; P = .006 for noninferiority). Edoxaban also was noninferior to dalteparin after the first 6 months of treatment and in the per-protocol analysis (HRs, 1.0; P = .02 for noninferiority in each analysis). Thus, differences in efficacy did not only reflect better compliance to oral therapy, Dr. Raskob said.
He also reported on individual outcomes. In all, 10.3% of dalteparin recipients had a VTE recurrence, as did 6.5% of edoxaban recipients, for a risk difference of 3.8% (95% CI, 7.1%-0.4%). More than half of recurrences in each group were symptomatic, and none were fatal. Bleeding caused no deaths in either study arm, and each therapy conferred an identical chance of a grade 3-4 major bleed (2.3%).
Edoxaban was associated, however, with a greater frequency of major bleeds (33 events; 6.3%) than was dalteparin (17 events; 3.2%; risk difference, 3.1%; 95% CI, 0.5%-5.7%). In particular, patients who received edoxaban had a slightly higher rate of upper gastrointestinal bleeds. Most had gastric cancer.
Future studies should evaluate whether these patients should receive a lower dose of edoxaban, said Dr. Raskob. “We don’t yet fully know the minimum effective dose [of edoxaban] in cancer patients.”
He also addressed the idea that heparin has antineoplastic activity, calling it “one we should probably abandon. The concept originates from older trials in which researchers probably did not recognize that heparin was preventing fatal pulmonary embolism, he said.
The investigators soon will begin deeper analyses that should inform patient selection, he said. For now, he recommends discussing these findings with patients to help them make an informed choice between oral anticoagulation, with its ease of use but slightly higher rate of major bleeds, and subcutaneous heparin, with its lower bleeding rate and treatment burden.
Daiichi Sankyo provided funding. Dr. Raskob disclosed consulting relationships and honoraria from Daiichi Sankyo, Eli Lilly, Janssen, and several other pharmaceutical companies.
SOURCE: Raskob G et al. ASH Abstract LBA-6.
REPORTING FROM ASH 2017
Key clinical point: Oral anticoagulation with edoxaban is easier, but has a slightly higher rate of major bleeds than does subcutaneous heparin.
Major finding: .
Data source: A randomized, multicenter, open-label trial of 1,046 adults with cancer and VTE.
Disclosures: Daiichi Sankyo provided funding. Dr. Raskob disclosed consulting relationships and honoraria from Daiichi Sankyo, Eli Lilly, Janssen, and several other pharmaceutical companies.
Source: Raskob G et al. ASH Abstract LBA-6.
Extra years of adjuvant bisphosphonate not needed in early breast cancer
SAN ANTONIO – When it comes to adjuvant bisphosphonate therapy following adjuvant chemotherapy for high-risk early breast cancer, more is not better than less, phase 3 data from the randomized SUCCESS A study suggest.
Among 3,421 patients randomized to adjuvant bisphosphonate therapy following chemotherapy, there was barely a speck of difference in either disease-free survival (DFS) or overall survival (OS) between patients randomized to either 2 years or 5 years of adjuvant bisphosphonate therapy with zoledronate, reported Wolfgang Janni, MD, from University Hospital Ulm (Germany).
“We conclude 5 years of adjuvant zoledronate treatment should not be considered currently in these patients in the absence of decreased bone density,” he said at the San Antonio Breast Cancer Symposium.
Adjuvant bisphosphonate therapy in patients with early breast cancer is associated with improved breast cancer–specific survival and reduced rates of breast cancer recurrence in bone, especially for postmenopausal patients, as shown in a meta-analysis from the Early Breast Cancer Trialists’ Collaborative Group, Dr. Janni noted.
German breast cancer guidelines state that postmenopausal women should be offered bisphosphonates as part of their adjuvant systemic therapy, but the optimal duration of therapy is uncertain, prompting the investigators to examine the issue in a randomized trial.
SUCCESS A was a multicenter, phase 3, randomized trial with a multifactorial 2 x 2 design, in patients with high-risk node-negative and node-positive disease. Patients were randomized to FEC100 chemotherapy followed by docetaxel with or without gemcitabine. Chemotherapy was followed by endocrine therapy with 2 years of tamoxifen followed by 3 years of anastrozole (Arimidex). At the start of endocrine therapy, patients were further randomized to receive either 2 or 5 years of adjuvant zoledronate, 4 mg intravenously every 3 months for 2 years, or the same schedule over 2 years, followed by 4 mg every 6 months for 3 years.
A total of 2,987 of the 3,421 patients randomized to a zoledronate schedule were available for inclusion in the analysis.
As noted, adapted DFS and OS, measured starting from 2 years after the start of zoledronate with a maximum observation time of 48 months, were virtually identical between the two treatment groups, with respective P values of .827 and .713. Similarly, in a multivariate regression analysis model adjusted for age, body mass index, menopausal status, tumor size, nodal stage, histological grade and type, hormone receptor status, HER2 status, surgery type, and chemotherapy regimen, the hazard ratio for 5 vs. 2 years was 0.97 for DFS and 0.98 for OS. Neither endpoint was significantly different between the groups.
Similarly, there was no significant differences in the number of bone recurrences as first distant recurrences or in premenopausal vs. postmenopausal women.
Adverse events of any grade were significantly higher with 5 years of bisphosphonate therapy (46.2% vs. 27.2%, P less than .001), including significantly higher grade 3 or greater adverse events (7.6% vs. 5.1%, P = .006).
Following presentation of the data in an oral session, moderator Sibylle Loibl, MD, PhD, of the German Breast Group in Neu-Isenburg, Germany, questioned whether the follow-up was long enough to detect a clinically meaningful difference.
“The negative result of this study might be due to the small observation time,” Dr. Janni conceded.”We have a quite intensive drug regimen for the first 2 years, so this might also be a contributing factor [as to why] we did not see any difference.”
The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
SOURCE: Janni et al. SABCS 2017 Abstract GS1-06
SAN ANTONIO – When it comes to adjuvant bisphosphonate therapy following adjuvant chemotherapy for high-risk early breast cancer, more is not better than less, phase 3 data from the randomized SUCCESS A study suggest.
Among 3,421 patients randomized to adjuvant bisphosphonate therapy following chemotherapy, there was barely a speck of difference in either disease-free survival (DFS) or overall survival (OS) between patients randomized to either 2 years or 5 years of adjuvant bisphosphonate therapy with zoledronate, reported Wolfgang Janni, MD, from University Hospital Ulm (Germany).
“We conclude 5 years of adjuvant zoledronate treatment should not be considered currently in these patients in the absence of decreased bone density,” he said at the San Antonio Breast Cancer Symposium.
Adjuvant bisphosphonate therapy in patients with early breast cancer is associated with improved breast cancer–specific survival and reduced rates of breast cancer recurrence in bone, especially for postmenopausal patients, as shown in a meta-analysis from the Early Breast Cancer Trialists’ Collaborative Group, Dr. Janni noted.
German breast cancer guidelines state that postmenopausal women should be offered bisphosphonates as part of their adjuvant systemic therapy, but the optimal duration of therapy is uncertain, prompting the investigators to examine the issue in a randomized trial.
SUCCESS A was a multicenter, phase 3, randomized trial with a multifactorial 2 x 2 design, in patients with high-risk node-negative and node-positive disease. Patients were randomized to FEC100 chemotherapy followed by docetaxel with or without gemcitabine. Chemotherapy was followed by endocrine therapy with 2 years of tamoxifen followed by 3 years of anastrozole (Arimidex). At the start of endocrine therapy, patients were further randomized to receive either 2 or 5 years of adjuvant zoledronate, 4 mg intravenously every 3 months for 2 years, or the same schedule over 2 years, followed by 4 mg every 6 months for 3 years.
A total of 2,987 of the 3,421 patients randomized to a zoledronate schedule were available for inclusion in the analysis.
As noted, adapted DFS and OS, measured starting from 2 years after the start of zoledronate with a maximum observation time of 48 months, were virtually identical between the two treatment groups, with respective P values of .827 and .713. Similarly, in a multivariate regression analysis model adjusted for age, body mass index, menopausal status, tumor size, nodal stage, histological grade and type, hormone receptor status, HER2 status, surgery type, and chemotherapy regimen, the hazard ratio for 5 vs. 2 years was 0.97 for DFS and 0.98 for OS. Neither endpoint was significantly different between the groups.
Similarly, there was no significant differences in the number of bone recurrences as first distant recurrences or in premenopausal vs. postmenopausal women.
Adverse events of any grade were significantly higher with 5 years of bisphosphonate therapy (46.2% vs. 27.2%, P less than .001), including significantly higher grade 3 or greater adverse events (7.6% vs. 5.1%, P = .006).
Following presentation of the data in an oral session, moderator Sibylle Loibl, MD, PhD, of the German Breast Group in Neu-Isenburg, Germany, questioned whether the follow-up was long enough to detect a clinically meaningful difference.
“The negative result of this study might be due to the small observation time,” Dr. Janni conceded.”We have a quite intensive drug regimen for the first 2 years, so this might also be a contributing factor [as to why] we did not see any difference.”
The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
SOURCE: Janni et al. SABCS 2017 Abstract GS1-06
SAN ANTONIO – When it comes to adjuvant bisphosphonate therapy following adjuvant chemotherapy for high-risk early breast cancer, more is not better than less, phase 3 data from the randomized SUCCESS A study suggest.
Among 3,421 patients randomized to adjuvant bisphosphonate therapy following chemotherapy, there was barely a speck of difference in either disease-free survival (DFS) or overall survival (OS) between patients randomized to either 2 years or 5 years of adjuvant bisphosphonate therapy with zoledronate, reported Wolfgang Janni, MD, from University Hospital Ulm (Germany).
“We conclude 5 years of adjuvant zoledronate treatment should not be considered currently in these patients in the absence of decreased bone density,” he said at the San Antonio Breast Cancer Symposium.
Adjuvant bisphosphonate therapy in patients with early breast cancer is associated with improved breast cancer–specific survival and reduced rates of breast cancer recurrence in bone, especially for postmenopausal patients, as shown in a meta-analysis from the Early Breast Cancer Trialists’ Collaborative Group, Dr. Janni noted.
German breast cancer guidelines state that postmenopausal women should be offered bisphosphonates as part of their adjuvant systemic therapy, but the optimal duration of therapy is uncertain, prompting the investigators to examine the issue in a randomized trial.
SUCCESS A was a multicenter, phase 3, randomized trial with a multifactorial 2 x 2 design, in patients with high-risk node-negative and node-positive disease. Patients were randomized to FEC100 chemotherapy followed by docetaxel with or without gemcitabine. Chemotherapy was followed by endocrine therapy with 2 years of tamoxifen followed by 3 years of anastrozole (Arimidex). At the start of endocrine therapy, patients were further randomized to receive either 2 or 5 years of adjuvant zoledronate, 4 mg intravenously every 3 months for 2 years, or the same schedule over 2 years, followed by 4 mg every 6 months for 3 years.
A total of 2,987 of the 3,421 patients randomized to a zoledronate schedule were available for inclusion in the analysis.
As noted, adapted DFS and OS, measured starting from 2 years after the start of zoledronate with a maximum observation time of 48 months, were virtually identical between the two treatment groups, with respective P values of .827 and .713. Similarly, in a multivariate regression analysis model adjusted for age, body mass index, menopausal status, tumor size, nodal stage, histological grade and type, hormone receptor status, HER2 status, surgery type, and chemotherapy regimen, the hazard ratio for 5 vs. 2 years was 0.97 for DFS and 0.98 for OS. Neither endpoint was significantly different between the groups.
Similarly, there was no significant differences in the number of bone recurrences as first distant recurrences or in premenopausal vs. postmenopausal women.
Adverse events of any grade were significantly higher with 5 years of bisphosphonate therapy (46.2% vs. 27.2%, P less than .001), including significantly higher grade 3 or greater adverse events (7.6% vs. 5.1%, P = .006).
Following presentation of the data in an oral session, moderator Sibylle Loibl, MD, PhD, of the German Breast Group in Neu-Isenburg, Germany, questioned whether the follow-up was long enough to detect a clinically meaningful difference.
“The negative result of this study might be due to the small observation time,” Dr. Janni conceded.”We have a quite intensive drug regimen for the first 2 years, so this might also be a contributing factor [as to why] we did not see any difference.”
The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
SOURCE: Janni et al. SABCS 2017 Abstract GS1-06
REPORTING FROM SABCS 2017
Key clinical point: Five years of adjuvant bisphosphonate therapy offered no survival advantages over 2 years of therapy for women with early breast cancers.
Major finding: Neither adapted disease-free survival nor overall survival were significantly better with 3 extra years of zoledronate therapy.
Data source: Randomized phase 3 trial.
Disclosures: The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
Source: Janni et al., SABCS 2017 abstract GS1-06
Ovarian suppression during breast cancer chemo helps stave off early menopause, preserve fertility
SAN ANTONIO – Premenopausal women with early breast cancer should be offered temporary ovarian suppression during chemotherapy if they wish to remain fertile or avoid early menopause, suggests a meta-analysis of five randomized controlled trials among 873 women reported at the San Antonio Breast Cancer Symposium.
“Oocyte and embryo cryopreservation are standard strategies for fertility preservation in these patients, meaning increasing the chance of pregnancy after the end of treatment,” said lead author Matteo Lambertini, MD, medical oncologist and ESMO fellow at the Institut Jules Bordet in Brussels. “However, they do not prevent the risk of developing chemotherapy-induced premature ovarian insufficiency, and so patients are still at risk of developing early menopause.”
Data from individual trials of ovarian suppression have been mixed, and its use remains controversial, he further noted. As a result, guidelines from ASCO and ESMO for fertility preservation in cancer patients still consider ovarian suppression to be investigational.
Results of the new meta-analysis, reported in a press briefing and session, showed that, compared with control peers, premenopausal women given a gonadotropin-releasing hormone analog (GnRHa) to suppress ovarian function during breast cancer chemotherapy had a more than one-half reduction in odds of premature ovarian insufficiency and were almost twice as likely to become pregnant after completing their treatment.
“We believe that this strategy should now be considered a standard option to reduce the likelihood of chemotherapy-induced premature ovarian insufficiency and potentially improve future fertility in premenopausal early breast cancer patients who undergo adjuvant or neoadjuvant chemotherapy,” Dr. Lambertini maintained. The analysis and trial participants “remind us that there is a life after cancer and to cure the disease should not be considered enough any more.”
These new data are sufficient to put the controversy to rest, for several reasons, he contended. “First, this is an individual-patient meta-analysis of the five major randomized controlled trials in this setting, so it’s kind of the highest level of evidence that we can reach. Second, it’s very unlikely that we will have new randomized, controlled trials in this setting, and I would say also that, based on the results, it would probably be unethical to run randomized, controlled trials in this setting.”
Press briefing moderator Carlos L. Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, Dallas, wondered how trials were selected for inclusion in the meta-analysis.
Thirteen trials have been conducted on ovarian suppression in premenopausal patients with breast cancer, and the investigators included the five for which they were able to obtain patient-level data, a subset that contained both positive and negative trials, Dr. Lambertini replied. But findings would likely be the same had all trials been included, given that a 2015 analysis using trial-level data from the 12 trials completed at that time showed very similar results (Ann Oncol. 2015 Dec;26:2408-19).
“Societally, this is a hugely significant issue, but the difference you are showing I have to admit is rather modest,” commented Dr. Arteaga, who is also SABCS codirector and AACR past president. “So what kind of conversation do you have with the patient? Who are the ones who would be the best candidates for this approach?”
“The main message is that giving a GnRHa does not avoid the risk of early menopause in all patients, but still, it decreases significantly the number of patients who have this side effect,” Dr. Lambertini replied.
Two groups are optimal candidates for ovarian suppression, he proposed. “First, the patients who are concerned about developing early menopause and its related side effects, who are not interested per se in having a baby after the end of treatment, but may be preserving ovarian function. [Second], for patients interested in having a baby, so interested in fertility preservation, this strategy can be used after cryopreservation procedures or in patients who have no access, for different reasons, to cryopreservation strategies.”
Another point of view
In the session, attendee Kutluk Oktay, MD, PhD, professor of obstetrics & gynecology and reproductive sciences at Yale University, New Haven, and cochair of ASCO’s guideline committee on fertility preservation said, “I cannot agree with your conclusions based on what you presented to us.”
In particular, he took issue with the exclusion of additional trials in breast cancer as well as trials among patients with other types of cancers. “I’m wondering what the rationale is to limit this to breast cancer because chemotherapy is chemotherapy and ovary is ovary, so underlying disease should not matter. By limiting it to breast cancer, you are leaving out three important studies, all in hematological cancer, with better designs … three negative studies,” he commented.
From a clinical point of view, patients with lymphoma and patients with breast cancer differ greatly, Dr. Lambertini countered: The former are about 20 years younger, on average, and often receive less-granulotoxic chemotherapy. “For these reasons, I don’t believe that mixing these two populations would have been [appropriate] for analysis,” he said. “From a methodological point of view, the studies you have mentioned include overall [fewer] than 150 patients, so it’s a very small proportion in comparison to the data we have in breast cancer.”
Study details
For their meta-analysis, Dr. Lambertini and coinvestigators pooled individual patient data from five trials (PROMISE-GIM6, POEMS/SWOG S0230, Anglo-Celtic Group OPTION, GBG-37 ZORO, and a trial led by the Moffitt Cancer Center) that randomized premenopausal women with early breast cancer to adjuvant or neoadjuvant chemotherapy either with or without concurrent GnRHa therapy.
Two of the trials restricted enrollment to women with estrogen receptor (ER)-negative disease. The GnRHa agents used were triptorelin (Trelstar, Triptodur)and goserelin(Zoladex).
Main results showed that the rate of premature ovarian insufficiency, defined differently across trials, was 14.1% among women given a GnRHa and 30.9% among control women (adjusted odds ratio, 0.38; P less than .001), Dr. Lambertini reported. Findings were similar in subgroups stratified by age, ER status, and type and duration of chemotherapy.
The rate of amenorrhea, used as a standardized definition of premature ovarian insufficiency, was similar in the GnRHa and control groups at 1 year (36.8% and 40.4%) but sharply lower in the former at 2 years (18.2% vs. 30.0%; adjusted odds ratio, 0.51; P = .009).
Overall, 10.3% of women in the GnRHa group and 5.5% in the control group had at least one pregnancy after completing their breast cancer treatment (incidence rate ratio, 1.83; P = .030). “All of the randomized trials except for the POEMS study actually did not have fertility outcomes as a preplanned endpoint, and so the patients’ wish to have a pregnancy was not collected,” he noted; therefore, it was not possible to calculate pregnancy rates in the subset who actually wanted to conceive.
All pregnancies occurred among women aged 40 years or younger, and 86% occurred among women who had had ER-negative disease, likely reflecting use of adjuvant endocrine therapy in patients with ER-positive disease, he said. Of the 57 total pregnancies, 50 resulted in live births, and none of the infants had malformations; the other pregnancies ended in spontaneous or induced abortion.
With a median follow-up of 5 years, the groups did not differ significantly on rates of disease-free survival and overall survival, suggesting that ovarian suppression was safe, according to Dr. Lambertini. Findings were similar when patients were stratified by ER status.
“What I think researchers should do in the next year is to better understand how this strategy [of ovarian suppression] works because this is probably the main controversy right now, because it’s still not very clear how this strategy actually works,” he concluded.
SOURCE: Lambertini M et al., SABCS 2017 Abstract GS4-01.
SAN ANTONIO – Premenopausal women with early breast cancer should be offered temporary ovarian suppression during chemotherapy if they wish to remain fertile or avoid early menopause, suggests a meta-analysis of five randomized controlled trials among 873 women reported at the San Antonio Breast Cancer Symposium.
“Oocyte and embryo cryopreservation are standard strategies for fertility preservation in these patients, meaning increasing the chance of pregnancy after the end of treatment,” said lead author Matteo Lambertini, MD, medical oncologist and ESMO fellow at the Institut Jules Bordet in Brussels. “However, they do not prevent the risk of developing chemotherapy-induced premature ovarian insufficiency, and so patients are still at risk of developing early menopause.”
Data from individual trials of ovarian suppression have been mixed, and its use remains controversial, he further noted. As a result, guidelines from ASCO and ESMO for fertility preservation in cancer patients still consider ovarian suppression to be investigational.
Results of the new meta-analysis, reported in a press briefing and session, showed that, compared with control peers, premenopausal women given a gonadotropin-releasing hormone analog (GnRHa) to suppress ovarian function during breast cancer chemotherapy had a more than one-half reduction in odds of premature ovarian insufficiency and were almost twice as likely to become pregnant after completing their treatment.
“We believe that this strategy should now be considered a standard option to reduce the likelihood of chemotherapy-induced premature ovarian insufficiency and potentially improve future fertility in premenopausal early breast cancer patients who undergo adjuvant or neoadjuvant chemotherapy,” Dr. Lambertini maintained. The analysis and trial participants “remind us that there is a life after cancer and to cure the disease should not be considered enough any more.”
These new data are sufficient to put the controversy to rest, for several reasons, he contended. “First, this is an individual-patient meta-analysis of the five major randomized controlled trials in this setting, so it’s kind of the highest level of evidence that we can reach. Second, it’s very unlikely that we will have new randomized, controlled trials in this setting, and I would say also that, based on the results, it would probably be unethical to run randomized, controlled trials in this setting.”
Press briefing moderator Carlos L. Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, Dallas, wondered how trials were selected for inclusion in the meta-analysis.
Thirteen trials have been conducted on ovarian suppression in premenopausal patients with breast cancer, and the investigators included the five for which they were able to obtain patient-level data, a subset that contained both positive and negative trials, Dr. Lambertini replied. But findings would likely be the same had all trials been included, given that a 2015 analysis using trial-level data from the 12 trials completed at that time showed very similar results (Ann Oncol. 2015 Dec;26:2408-19).
“Societally, this is a hugely significant issue, but the difference you are showing I have to admit is rather modest,” commented Dr. Arteaga, who is also SABCS codirector and AACR past president. “So what kind of conversation do you have with the patient? Who are the ones who would be the best candidates for this approach?”
“The main message is that giving a GnRHa does not avoid the risk of early menopause in all patients, but still, it decreases significantly the number of patients who have this side effect,” Dr. Lambertini replied.
Two groups are optimal candidates for ovarian suppression, he proposed. “First, the patients who are concerned about developing early menopause and its related side effects, who are not interested per se in having a baby after the end of treatment, but may be preserving ovarian function. [Second], for patients interested in having a baby, so interested in fertility preservation, this strategy can be used after cryopreservation procedures or in patients who have no access, for different reasons, to cryopreservation strategies.”
Another point of view
In the session, attendee Kutluk Oktay, MD, PhD, professor of obstetrics & gynecology and reproductive sciences at Yale University, New Haven, and cochair of ASCO’s guideline committee on fertility preservation said, “I cannot agree with your conclusions based on what you presented to us.”
In particular, he took issue with the exclusion of additional trials in breast cancer as well as trials among patients with other types of cancers. “I’m wondering what the rationale is to limit this to breast cancer because chemotherapy is chemotherapy and ovary is ovary, so underlying disease should not matter. By limiting it to breast cancer, you are leaving out three important studies, all in hematological cancer, with better designs … three negative studies,” he commented.
From a clinical point of view, patients with lymphoma and patients with breast cancer differ greatly, Dr. Lambertini countered: The former are about 20 years younger, on average, and often receive less-granulotoxic chemotherapy. “For these reasons, I don’t believe that mixing these two populations would have been [appropriate] for analysis,” he said. “From a methodological point of view, the studies you have mentioned include overall [fewer] than 150 patients, so it’s a very small proportion in comparison to the data we have in breast cancer.”
Study details
For their meta-analysis, Dr. Lambertini and coinvestigators pooled individual patient data from five trials (PROMISE-GIM6, POEMS/SWOG S0230, Anglo-Celtic Group OPTION, GBG-37 ZORO, and a trial led by the Moffitt Cancer Center) that randomized premenopausal women with early breast cancer to adjuvant or neoadjuvant chemotherapy either with or without concurrent GnRHa therapy.
Two of the trials restricted enrollment to women with estrogen receptor (ER)-negative disease. The GnRHa agents used were triptorelin (Trelstar, Triptodur)and goserelin(Zoladex).
Main results showed that the rate of premature ovarian insufficiency, defined differently across trials, was 14.1% among women given a GnRHa and 30.9% among control women (adjusted odds ratio, 0.38; P less than .001), Dr. Lambertini reported. Findings were similar in subgroups stratified by age, ER status, and type and duration of chemotherapy.
The rate of amenorrhea, used as a standardized definition of premature ovarian insufficiency, was similar in the GnRHa and control groups at 1 year (36.8% and 40.4%) but sharply lower in the former at 2 years (18.2% vs. 30.0%; adjusted odds ratio, 0.51; P = .009).
Overall, 10.3% of women in the GnRHa group and 5.5% in the control group had at least one pregnancy after completing their breast cancer treatment (incidence rate ratio, 1.83; P = .030). “All of the randomized trials except for the POEMS study actually did not have fertility outcomes as a preplanned endpoint, and so the patients’ wish to have a pregnancy was not collected,” he noted; therefore, it was not possible to calculate pregnancy rates in the subset who actually wanted to conceive.
All pregnancies occurred among women aged 40 years or younger, and 86% occurred among women who had had ER-negative disease, likely reflecting use of adjuvant endocrine therapy in patients with ER-positive disease, he said. Of the 57 total pregnancies, 50 resulted in live births, and none of the infants had malformations; the other pregnancies ended in spontaneous or induced abortion.
With a median follow-up of 5 years, the groups did not differ significantly on rates of disease-free survival and overall survival, suggesting that ovarian suppression was safe, according to Dr. Lambertini. Findings were similar when patients were stratified by ER status.
“What I think researchers should do in the next year is to better understand how this strategy [of ovarian suppression] works because this is probably the main controversy right now, because it’s still not very clear how this strategy actually works,” he concluded.
SOURCE: Lambertini M et al., SABCS 2017 Abstract GS4-01.
SAN ANTONIO – Premenopausal women with early breast cancer should be offered temporary ovarian suppression during chemotherapy if they wish to remain fertile or avoid early menopause, suggests a meta-analysis of five randomized controlled trials among 873 women reported at the San Antonio Breast Cancer Symposium.
“Oocyte and embryo cryopreservation are standard strategies for fertility preservation in these patients, meaning increasing the chance of pregnancy after the end of treatment,” said lead author Matteo Lambertini, MD, medical oncologist and ESMO fellow at the Institut Jules Bordet in Brussels. “However, they do not prevent the risk of developing chemotherapy-induced premature ovarian insufficiency, and so patients are still at risk of developing early menopause.”
Data from individual trials of ovarian suppression have been mixed, and its use remains controversial, he further noted. As a result, guidelines from ASCO and ESMO for fertility preservation in cancer patients still consider ovarian suppression to be investigational.
Results of the new meta-analysis, reported in a press briefing and session, showed that, compared with control peers, premenopausal women given a gonadotropin-releasing hormone analog (GnRHa) to suppress ovarian function during breast cancer chemotherapy had a more than one-half reduction in odds of premature ovarian insufficiency and were almost twice as likely to become pregnant after completing their treatment.
“We believe that this strategy should now be considered a standard option to reduce the likelihood of chemotherapy-induced premature ovarian insufficiency and potentially improve future fertility in premenopausal early breast cancer patients who undergo adjuvant or neoadjuvant chemotherapy,” Dr. Lambertini maintained. The analysis and trial participants “remind us that there is a life after cancer and to cure the disease should not be considered enough any more.”
These new data are sufficient to put the controversy to rest, for several reasons, he contended. “First, this is an individual-patient meta-analysis of the five major randomized controlled trials in this setting, so it’s kind of the highest level of evidence that we can reach. Second, it’s very unlikely that we will have new randomized, controlled trials in this setting, and I would say also that, based on the results, it would probably be unethical to run randomized, controlled trials in this setting.”
Press briefing moderator Carlos L. Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, Dallas, wondered how trials were selected for inclusion in the meta-analysis.
Thirteen trials have been conducted on ovarian suppression in premenopausal patients with breast cancer, and the investigators included the five for which they were able to obtain patient-level data, a subset that contained both positive and negative trials, Dr. Lambertini replied. But findings would likely be the same had all trials been included, given that a 2015 analysis using trial-level data from the 12 trials completed at that time showed very similar results (Ann Oncol. 2015 Dec;26:2408-19).
“Societally, this is a hugely significant issue, but the difference you are showing I have to admit is rather modest,” commented Dr. Arteaga, who is also SABCS codirector and AACR past president. “So what kind of conversation do you have with the patient? Who are the ones who would be the best candidates for this approach?”
“The main message is that giving a GnRHa does not avoid the risk of early menopause in all patients, but still, it decreases significantly the number of patients who have this side effect,” Dr. Lambertini replied.
Two groups are optimal candidates for ovarian suppression, he proposed. “First, the patients who are concerned about developing early menopause and its related side effects, who are not interested per se in having a baby after the end of treatment, but may be preserving ovarian function. [Second], for patients interested in having a baby, so interested in fertility preservation, this strategy can be used after cryopreservation procedures or in patients who have no access, for different reasons, to cryopreservation strategies.”
Another point of view
In the session, attendee Kutluk Oktay, MD, PhD, professor of obstetrics & gynecology and reproductive sciences at Yale University, New Haven, and cochair of ASCO’s guideline committee on fertility preservation said, “I cannot agree with your conclusions based on what you presented to us.”
In particular, he took issue with the exclusion of additional trials in breast cancer as well as trials among patients with other types of cancers. “I’m wondering what the rationale is to limit this to breast cancer because chemotherapy is chemotherapy and ovary is ovary, so underlying disease should not matter. By limiting it to breast cancer, you are leaving out three important studies, all in hematological cancer, with better designs … three negative studies,” he commented.
From a clinical point of view, patients with lymphoma and patients with breast cancer differ greatly, Dr. Lambertini countered: The former are about 20 years younger, on average, and often receive less-granulotoxic chemotherapy. “For these reasons, I don’t believe that mixing these two populations would have been [appropriate] for analysis,” he said. “From a methodological point of view, the studies you have mentioned include overall [fewer] than 150 patients, so it’s a very small proportion in comparison to the data we have in breast cancer.”
Study details
For their meta-analysis, Dr. Lambertini and coinvestigators pooled individual patient data from five trials (PROMISE-GIM6, POEMS/SWOG S0230, Anglo-Celtic Group OPTION, GBG-37 ZORO, and a trial led by the Moffitt Cancer Center) that randomized premenopausal women with early breast cancer to adjuvant or neoadjuvant chemotherapy either with or without concurrent GnRHa therapy.
Two of the trials restricted enrollment to women with estrogen receptor (ER)-negative disease. The GnRHa agents used were triptorelin (Trelstar, Triptodur)and goserelin(Zoladex).
Main results showed that the rate of premature ovarian insufficiency, defined differently across trials, was 14.1% among women given a GnRHa and 30.9% among control women (adjusted odds ratio, 0.38; P less than .001), Dr. Lambertini reported. Findings were similar in subgroups stratified by age, ER status, and type and duration of chemotherapy.
The rate of amenorrhea, used as a standardized definition of premature ovarian insufficiency, was similar in the GnRHa and control groups at 1 year (36.8% and 40.4%) but sharply lower in the former at 2 years (18.2% vs. 30.0%; adjusted odds ratio, 0.51; P = .009).
Overall, 10.3% of women in the GnRHa group and 5.5% in the control group had at least one pregnancy after completing their breast cancer treatment (incidence rate ratio, 1.83; P = .030). “All of the randomized trials except for the POEMS study actually did not have fertility outcomes as a preplanned endpoint, and so the patients’ wish to have a pregnancy was not collected,” he noted; therefore, it was not possible to calculate pregnancy rates in the subset who actually wanted to conceive.
All pregnancies occurred among women aged 40 years or younger, and 86% occurred among women who had had ER-negative disease, likely reflecting use of adjuvant endocrine therapy in patients with ER-positive disease, he said. Of the 57 total pregnancies, 50 resulted in live births, and none of the infants had malformations; the other pregnancies ended in spontaneous or induced abortion.
With a median follow-up of 5 years, the groups did not differ significantly on rates of disease-free survival and overall survival, suggesting that ovarian suppression was safe, according to Dr. Lambertini. Findings were similar when patients were stratified by ER status.
“What I think researchers should do in the next year is to better understand how this strategy [of ovarian suppression] works because this is probably the main controversy right now, because it’s still not very clear how this strategy actually works,” he concluded.
SOURCE: Lambertini M et al., SABCS 2017 Abstract GS4-01.
REPORTING FROM SABCS 2017
Key clinical point:
Major finding: Compared with controls, women given a GnRHa during chemotherapy were less likely to develop premature ovarian insufficiency (adjusted OR, 0.38; P less than .001) and more likely to become pregnant after treatment (IRR, 1.83; P = .030).
Data source: A meta-analysis of individual patient data from five trials among 873 premenopausal women with early breast cancer.
Disclosures: Dr. Lambertini disclosed that he had no relevant conflicts of interest.
Source: Lambertini M et al., SABCS 2017 Abstract GS4-01.
VIDEO: Weight loss cut risk of breast cancer
SAN ANTONIO – A new analysis from the Women’s Health Initiative Observational Study gives postmenopausal women yet another reason to mind their weight. Results showed that women losing at least 5% of their body weight had a significant 12% reduction in adjusted breast cancer risk relative to peers who maintained a stable weight, reported lead author Rowan Chlebowski, MD, PhD, research professor in the department of medical oncology and therapeutics research at City of Hope in Duarte, Calif. Findings were much the same regardless of whether women were of normal weight, overweight, or obese at baseline. Dr. Chlebowski discussed the implications for patient counseling and insurance coverage of weight loss interventions, as well as planned research that will assess the physiologic mechanisms at play in a video interview.
SAN ANTONIO – A new analysis from the Women’s Health Initiative Observational Study gives postmenopausal women yet another reason to mind their weight. Results showed that women losing at least 5% of their body weight had a significant 12% reduction in adjusted breast cancer risk relative to peers who maintained a stable weight, reported lead author Rowan Chlebowski, MD, PhD, research professor in the department of medical oncology and therapeutics research at City of Hope in Duarte, Calif. Findings were much the same regardless of whether women were of normal weight, overweight, or obese at baseline. Dr. Chlebowski discussed the implications for patient counseling and insurance coverage of weight loss interventions, as well as planned research that will assess the physiologic mechanisms at play in a video interview.
SAN ANTONIO – A new analysis from the Women’s Health Initiative Observational Study gives postmenopausal women yet another reason to mind their weight. Results showed that women losing at least 5% of their body weight had a significant 12% reduction in adjusted breast cancer risk relative to peers who maintained a stable weight, reported lead author Rowan Chlebowski, MD, PhD, research professor in the department of medical oncology and therapeutics research at City of Hope in Duarte, Calif. Findings were much the same regardless of whether women were of normal weight, overweight, or obese at baseline. Dr. Chlebowski discussed the implications for patient counseling and insurance coverage of weight loss interventions, as well as planned research that will assess the physiologic mechanisms at play in a video interview.
REPORTING FROM SABCS 2017
2 = 5 for additional AI therapy for postmenopausal HR+ breast cancer
SAN ANTONIO – Five years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer, Austrian investigators reported.
In fact, the only thing that the additional years of the aromatase inhibitor (AI) anastrozole seemed to add was increased risk for fracture, said Michael Gnant, MD, from the Medical University of Vienna, on behalf of colleagues in the ABCSG-16 trial.
Previous trials have convincingly demonstrated the benefit of giving patients an AI for 5 years after 5 years of tamoxifen, but the optimal duration of extended adjuvant AI therapy is not known, Dr. Gnant said.
The ABCSG trialists recruited 3,484 postmenopausal women from with HR+, stage T1-3, node-negative or -positive, nonmetastatic breast cancer who had completed 4-6 years of endocrine therapy with either tamoxifen, an AI, or tamoxifen followed by an AI. The patients were randomly assigned at the end of initial endocrine therapy to either 2 years or 5 years of anastrozole.
As noted before, disease-free survival (DFS), the primary endpoint, was virtually identical between the treatment arms. The DFS rate at a median of 8.75 years after randomization – that is, approximately 14 years after diagnosis – was 71.1% among patients treated for 2 additional years, vs. 70.3% for patients treated for 5 extra years, translating into a hazard ratio of 1.007 and making the contest a statistical dead heat.
Similarly, there was no difference by anastrozole duration in the secondary endpoint of overall survival at 10 years, with respective rates of 85.3% vs. 84.9%, with a hazard ratio identical to that in the DFS analysis.
Where the 5-year schedule surpassed the 2-year schedule, however, was in apparent risk for fractures, which was 6.3% after 5 years of additional therapy, compared with 4.7% at 5 years among patients who received just 2 additional years of anastrozole. The hazard ratio associated with the difference was 1.353, but because the lower end of the 95% confidence interval was 1.00, the finding was of borderline significance (P = .053), Dr. Gnant acknowledged.
There are several ongoing translational studies that may help to identify specific molecular characteristics that could predict benefit from prolonged extended therapy in a given patient, “but for now we can conclude that 7 years are good enough for almost every patient with luminal breast cancer,” Dr. Gnant said at a briefing prior to his presentation of the study in an oral session.
“I do believe that for us as clinical scientists a negative trial is always disappointing, but I think the clinical take-home message can actually help to avoid unnecessary side effects for many, many women,” he added.
Asked at the briefing whether, given the identical survival curves between the two trial arms, additional therapy beyond 5 years was needed, Dr. Gnant replied “that was addressed by other trials. I think that the trials after tamoxifen are very clear: We have hazard ratios around 0.6 after tamoxifen, so some type of extension for adding aromatase inhibitors should be the standard of care.”
He noted that the optimal duration of additional therapy with an AI has not been known, because the trial that could have answered that question, the MA-17 trial, was halted and unblinded after just 2.5 years when an interim analysis showed superior survival with letrozole (Femara), compared with placebo.
More than 60% of patients in the placebo group in that trial were crossed over to letrozole, further muddying long-term follow-up results.
Carlos Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center in Dallas, who moderated the briefing, agreed with Dr. Gnant that this ostensibly negative trial had good results for patients.
“I hope that we continue to see more de-escalation studies. I hope that as we combine AIs with CDK4/6 inhibitors, we may make therapy even shorter. I think we should do better than just extending and extending and extending. We have to come up with better ideas,” he said in an interview.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
SOURCE: Gnant et al. SABCS 2017 Abstract GS3-01
SAN ANTONIO – Five years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer, Austrian investigators reported.
In fact, the only thing that the additional years of the aromatase inhibitor (AI) anastrozole seemed to add was increased risk for fracture, said Michael Gnant, MD, from the Medical University of Vienna, on behalf of colleagues in the ABCSG-16 trial.
Previous trials have convincingly demonstrated the benefit of giving patients an AI for 5 years after 5 years of tamoxifen, but the optimal duration of extended adjuvant AI therapy is not known, Dr. Gnant said.
The ABCSG trialists recruited 3,484 postmenopausal women from with HR+, stage T1-3, node-negative or -positive, nonmetastatic breast cancer who had completed 4-6 years of endocrine therapy with either tamoxifen, an AI, or tamoxifen followed by an AI. The patients were randomly assigned at the end of initial endocrine therapy to either 2 years or 5 years of anastrozole.
As noted before, disease-free survival (DFS), the primary endpoint, was virtually identical between the treatment arms. The DFS rate at a median of 8.75 years after randomization – that is, approximately 14 years after diagnosis – was 71.1% among patients treated for 2 additional years, vs. 70.3% for patients treated for 5 extra years, translating into a hazard ratio of 1.007 and making the contest a statistical dead heat.
Similarly, there was no difference by anastrozole duration in the secondary endpoint of overall survival at 10 years, with respective rates of 85.3% vs. 84.9%, with a hazard ratio identical to that in the DFS analysis.
Where the 5-year schedule surpassed the 2-year schedule, however, was in apparent risk for fractures, which was 6.3% after 5 years of additional therapy, compared with 4.7% at 5 years among patients who received just 2 additional years of anastrozole. The hazard ratio associated with the difference was 1.353, but because the lower end of the 95% confidence interval was 1.00, the finding was of borderline significance (P = .053), Dr. Gnant acknowledged.
There are several ongoing translational studies that may help to identify specific molecular characteristics that could predict benefit from prolonged extended therapy in a given patient, “but for now we can conclude that 7 years are good enough for almost every patient with luminal breast cancer,” Dr. Gnant said at a briefing prior to his presentation of the study in an oral session.
“I do believe that for us as clinical scientists a negative trial is always disappointing, but I think the clinical take-home message can actually help to avoid unnecessary side effects for many, many women,” he added.
Asked at the briefing whether, given the identical survival curves between the two trial arms, additional therapy beyond 5 years was needed, Dr. Gnant replied “that was addressed by other trials. I think that the trials after tamoxifen are very clear: We have hazard ratios around 0.6 after tamoxifen, so some type of extension for adding aromatase inhibitors should be the standard of care.”
He noted that the optimal duration of additional therapy with an AI has not been known, because the trial that could have answered that question, the MA-17 trial, was halted and unblinded after just 2.5 years when an interim analysis showed superior survival with letrozole (Femara), compared with placebo.
More than 60% of patients in the placebo group in that trial were crossed over to letrozole, further muddying long-term follow-up results.
Carlos Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center in Dallas, who moderated the briefing, agreed with Dr. Gnant that this ostensibly negative trial had good results for patients.
“I hope that we continue to see more de-escalation studies. I hope that as we combine AIs with CDK4/6 inhibitors, we may make therapy even shorter. I think we should do better than just extending and extending and extending. We have to come up with better ideas,” he said in an interview.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
SOURCE: Gnant et al. SABCS 2017 Abstract GS3-01
SAN ANTONIO – Five years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer, Austrian investigators reported.
In fact, the only thing that the additional years of the aromatase inhibitor (AI) anastrozole seemed to add was increased risk for fracture, said Michael Gnant, MD, from the Medical University of Vienna, on behalf of colleagues in the ABCSG-16 trial.
Previous trials have convincingly demonstrated the benefit of giving patients an AI for 5 years after 5 years of tamoxifen, but the optimal duration of extended adjuvant AI therapy is not known, Dr. Gnant said.
The ABCSG trialists recruited 3,484 postmenopausal women from with HR+, stage T1-3, node-negative or -positive, nonmetastatic breast cancer who had completed 4-6 years of endocrine therapy with either tamoxifen, an AI, or tamoxifen followed by an AI. The patients were randomly assigned at the end of initial endocrine therapy to either 2 years or 5 years of anastrozole.
As noted before, disease-free survival (DFS), the primary endpoint, was virtually identical between the treatment arms. The DFS rate at a median of 8.75 years after randomization – that is, approximately 14 years after diagnosis – was 71.1% among patients treated for 2 additional years, vs. 70.3% for patients treated for 5 extra years, translating into a hazard ratio of 1.007 and making the contest a statistical dead heat.
Similarly, there was no difference by anastrozole duration in the secondary endpoint of overall survival at 10 years, with respective rates of 85.3% vs. 84.9%, with a hazard ratio identical to that in the DFS analysis.
Where the 5-year schedule surpassed the 2-year schedule, however, was in apparent risk for fractures, which was 6.3% after 5 years of additional therapy, compared with 4.7% at 5 years among patients who received just 2 additional years of anastrozole. The hazard ratio associated with the difference was 1.353, but because the lower end of the 95% confidence interval was 1.00, the finding was of borderline significance (P = .053), Dr. Gnant acknowledged.
There are several ongoing translational studies that may help to identify specific molecular characteristics that could predict benefit from prolonged extended therapy in a given patient, “but for now we can conclude that 7 years are good enough for almost every patient with luminal breast cancer,” Dr. Gnant said at a briefing prior to his presentation of the study in an oral session.
“I do believe that for us as clinical scientists a negative trial is always disappointing, but I think the clinical take-home message can actually help to avoid unnecessary side effects for many, many women,” he added.
Asked at the briefing whether, given the identical survival curves between the two trial arms, additional therapy beyond 5 years was needed, Dr. Gnant replied “that was addressed by other trials. I think that the trials after tamoxifen are very clear: We have hazard ratios around 0.6 after tamoxifen, so some type of extension for adding aromatase inhibitors should be the standard of care.”
He noted that the optimal duration of additional therapy with an AI has not been known, because the trial that could have answered that question, the MA-17 trial, was halted and unblinded after just 2.5 years when an interim analysis showed superior survival with letrozole (Femara), compared with placebo.
More than 60% of patients in the placebo group in that trial were crossed over to letrozole, further muddying long-term follow-up results.
Carlos Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center in Dallas, who moderated the briefing, agreed with Dr. Gnant that this ostensibly negative trial had good results for patients.
“I hope that we continue to see more de-escalation studies. I hope that as we combine AIs with CDK4/6 inhibitors, we may make therapy even shorter. I think we should do better than just extending and extending and extending. We have to come up with better ideas,” he said in an interview.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
SOURCE: Gnant et al. SABCS 2017 Abstract GS3-01
REPORTING FROM SABCS 2017
Key clinical point: Disease-free and overall survival were no different for women treated with 2 or 5 additional years of aromatase inhibitor therapy following 4-6 years of initial endocrine therapy.
Major finding: The hazard ratio for both DFS and OS with 5 additional years of anastrozole, compared with 2 years, was 1.007 and was not statistically significant.
Data source: Randomized phase 3 trial in 3,484 postmenopausal women with hormone receptor–positive breast cancer.
Disclosures: The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
Source: Gnant et al. SABCS 2017 Abstract GS3-01
Acupuncture significantly reduces AI-associated arthralgias
SAN ANTONIO – Acupuncture significantly reduced joint pain that was associated with the use of aromatase inhibitors (AIs) in women with early breast cancer, according to new findings reported at the San Antonio Breast Cancer Symposium.
The randomized, phase 3 SWOG S1200 clinical trial found that, compared with sham acupuncture and a control group receiving no therapy, women receiving acupuncture reported significantly lower scores on the Brief Pain Inventory–Short Form (BPI).
“We have shown consistently, with multiple measures assessing pain and stiffness, that true acupuncture generated better outcomes than either control group in a large multicenter trial,” said lead author Dawn L. Hershman, MD, leader of the Breast Cancer Program at the Herbert Irving Comprehensive Cancer Center at NewYork-Presbyterian/Columbia University Medical Center. “Acupuncture provides a nonpharmacologic option that can improve symptoms and possibly increase AI adherence and subsequent breast cancer outcomes.”
AIs can reduce both early breast cancer recurrence and mortality. Dr. Hershman noted that these agents are effective in the adjuvant setting and for prevention “but we know that it doesn’t work if you don’t take it. Noncompliance is a major problem among women taking hormonal therapy.”
Noncompliance is multifactorial and one of the main reasons women discontinue their therapy early is because of arthralgias or joint discomfort. “We were interested in a nonpharmacologic intervention, to assess whether or not we could control these symptoms.”
Dr. Hershman pointed out acupuncture provides a safe and effective alternative for patients reluctant to take a prescription medication that can result in other side effects. “Identification of nonopioid options for pain control is a public health priority,” she said.
Acupuncture is a popular nonpharmacologic modality and widely used for a number of indications. Several single-institution studies have suggested that it may be useful for controlling AI-associated arthralgias, while other studies have not demonstrated a benefit.
In this trial, the authors evaluated the efficacy of acupuncture, compared with sham acupuncture or waitlist control, in the treatment of AI associated arthralgia in a large population of patients. The study was conducted at 11 centers.
The cohort comprised 226 postmenopausal women diagnosed with early-stage, hormone receptor–positive breast cancer who were receiving treatment with AIs. The primary endpoint was the decline in joint pain as measured by BPI-SF at 6 weeks, and to assess the duration of the effect, the women were followed for an additional 12 weeks.
Within this group, 110 were randomized to true acupuncture; 59 to sham acupuncture, and 57 to waitlist control (no treatment). Patients receiving true or sham acupuncture had sessions three times a week for 6 weeks followed by one session per week for 6 more weeks. Pain status was reported at baseline, during treatment, and then afterwards, using a variety of measurement tools including the BPI-SF, which is a self-administered 14-item questionnaire that evaluates pain severity on a 0-10 scale, and the impact of pain on activities of daily living.
At 6 weeks, the true acupuncture treatment arm reported significantly lower BPI worst pain scores than those in the sham acupuncture and the waitlist control arms. The mean BPI worst pain for the true acupuncture arm was 0.92 points lower than the sham acupuncture arm (P = .01) and 0.96 points lower than the waitlist control arm (P = .01). The proportion of patients experiencing a large reduction in BPI worst pain (greater than 2) was significantly greater in the true acupuncture arm, compared with the other groups: 58% versus 33% percent and 31%, respectively. The differences continued to remain statistically significant at 24 weeks, even though the treatment only continued for 12 weeks.
Associated adverse effects were minimal with true and sham acupuncture and limited to grade 1 bruising.
The cost of the 12-week intervention was about $1,250 or $65-$75 a session. “We feel that there is now sufficient evidence to support insurance coverage of acupuncture of AI arthralgia.”
In a discussion of the paper, Dr. Anne Partridge, from the Dana Farber Cancer Center, noted that it is imperative to seek new ways to improve outcomes in breast cancer, and AIs are contributing to that. However, she echoed the concern that nonadherence to treatment is a “tremendous problem” and hampers the clinical effectiveness of AI therapy.
The rate of discontinuation during the first year of therapy is 20% within the first year and up to 40% of patients do not take them daily. Both early discontinuation and nonadherence contribute to mortality.
Based on these results from the largest randomized controlled trial looking at acupuncture in this setting, should physicians be recommending acupuncture to patients prescribed AI therapy?
“The short answer is, why not?” said Dr. Partridge, “And that we should be recommending it for some of our patients.”
However, there are a number of issues that need to be addressed, she added. The duration of treatment is not known, and the need for follow-up treatment or the frequency of it is not known. The generalizability of it is also unclear when looking at a larger population, and acupuncture is highly operator dependent.
“There are cost and access issues, and insurance right now offers very limited coverage,” she said.
Importantly, Dr. Partridge emphasized, “We know that it will help symptoms, but will it improve adherence to AI?”
It may improve adherence for some patients, but “side effects are only one factor,” she said. “Adherence behavior is complicated. We need to figure out how to optimize these therapies in our patients.”
This study was supported by the National Institutes of Health National Center for Complementary and Integrative Health and the Office of Research on Women’s Health, and grants from the NIH/National Cancer Institute Division of Cancer Prevention. Dr. Hershman declared no conflicts of interest. Dr. Partridge had no disclosures.
SOURCE: Hershman et al. Abstract GS4-04
SAN ANTONIO – Acupuncture significantly reduced joint pain that was associated with the use of aromatase inhibitors (AIs) in women with early breast cancer, according to new findings reported at the San Antonio Breast Cancer Symposium.
The randomized, phase 3 SWOG S1200 clinical trial found that, compared with sham acupuncture and a control group receiving no therapy, women receiving acupuncture reported significantly lower scores on the Brief Pain Inventory–Short Form (BPI).
“We have shown consistently, with multiple measures assessing pain and stiffness, that true acupuncture generated better outcomes than either control group in a large multicenter trial,” said lead author Dawn L. Hershman, MD, leader of the Breast Cancer Program at the Herbert Irving Comprehensive Cancer Center at NewYork-Presbyterian/Columbia University Medical Center. “Acupuncture provides a nonpharmacologic option that can improve symptoms and possibly increase AI adherence and subsequent breast cancer outcomes.”
AIs can reduce both early breast cancer recurrence and mortality. Dr. Hershman noted that these agents are effective in the adjuvant setting and for prevention “but we know that it doesn’t work if you don’t take it. Noncompliance is a major problem among women taking hormonal therapy.”
Noncompliance is multifactorial and one of the main reasons women discontinue their therapy early is because of arthralgias or joint discomfort. “We were interested in a nonpharmacologic intervention, to assess whether or not we could control these symptoms.”
Dr. Hershman pointed out acupuncture provides a safe and effective alternative for patients reluctant to take a prescription medication that can result in other side effects. “Identification of nonopioid options for pain control is a public health priority,” she said.
Acupuncture is a popular nonpharmacologic modality and widely used for a number of indications. Several single-institution studies have suggested that it may be useful for controlling AI-associated arthralgias, while other studies have not demonstrated a benefit.
In this trial, the authors evaluated the efficacy of acupuncture, compared with sham acupuncture or waitlist control, in the treatment of AI associated arthralgia in a large population of patients. The study was conducted at 11 centers.
The cohort comprised 226 postmenopausal women diagnosed with early-stage, hormone receptor–positive breast cancer who were receiving treatment with AIs. The primary endpoint was the decline in joint pain as measured by BPI-SF at 6 weeks, and to assess the duration of the effect, the women were followed for an additional 12 weeks.
Within this group, 110 were randomized to true acupuncture; 59 to sham acupuncture, and 57 to waitlist control (no treatment). Patients receiving true or sham acupuncture had sessions three times a week for 6 weeks followed by one session per week for 6 more weeks. Pain status was reported at baseline, during treatment, and then afterwards, using a variety of measurement tools including the BPI-SF, which is a self-administered 14-item questionnaire that evaluates pain severity on a 0-10 scale, and the impact of pain on activities of daily living.
At 6 weeks, the true acupuncture treatment arm reported significantly lower BPI worst pain scores than those in the sham acupuncture and the waitlist control arms. The mean BPI worst pain for the true acupuncture arm was 0.92 points lower than the sham acupuncture arm (P = .01) and 0.96 points lower than the waitlist control arm (P = .01). The proportion of patients experiencing a large reduction in BPI worst pain (greater than 2) was significantly greater in the true acupuncture arm, compared with the other groups: 58% versus 33% percent and 31%, respectively. The differences continued to remain statistically significant at 24 weeks, even though the treatment only continued for 12 weeks.
Associated adverse effects were minimal with true and sham acupuncture and limited to grade 1 bruising.
The cost of the 12-week intervention was about $1,250 or $65-$75 a session. “We feel that there is now sufficient evidence to support insurance coverage of acupuncture of AI arthralgia.”
In a discussion of the paper, Dr. Anne Partridge, from the Dana Farber Cancer Center, noted that it is imperative to seek new ways to improve outcomes in breast cancer, and AIs are contributing to that. However, she echoed the concern that nonadherence to treatment is a “tremendous problem” and hampers the clinical effectiveness of AI therapy.
The rate of discontinuation during the first year of therapy is 20% within the first year and up to 40% of patients do not take them daily. Both early discontinuation and nonadherence contribute to mortality.
Based on these results from the largest randomized controlled trial looking at acupuncture in this setting, should physicians be recommending acupuncture to patients prescribed AI therapy?
“The short answer is, why not?” said Dr. Partridge, “And that we should be recommending it for some of our patients.”
However, there are a number of issues that need to be addressed, she added. The duration of treatment is not known, and the need for follow-up treatment or the frequency of it is not known. The generalizability of it is also unclear when looking at a larger population, and acupuncture is highly operator dependent.
“There are cost and access issues, and insurance right now offers very limited coverage,” she said.
Importantly, Dr. Partridge emphasized, “We know that it will help symptoms, but will it improve adherence to AI?”
It may improve adherence for some patients, but “side effects are only one factor,” she said. “Adherence behavior is complicated. We need to figure out how to optimize these therapies in our patients.”
This study was supported by the National Institutes of Health National Center for Complementary and Integrative Health and the Office of Research on Women’s Health, and grants from the NIH/National Cancer Institute Division of Cancer Prevention. Dr. Hershman declared no conflicts of interest. Dr. Partridge had no disclosures.
SOURCE: Hershman et al. Abstract GS4-04
SAN ANTONIO – Acupuncture significantly reduced joint pain that was associated with the use of aromatase inhibitors (AIs) in women with early breast cancer, according to new findings reported at the San Antonio Breast Cancer Symposium.
The randomized, phase 3 SWOG S1200 clinical trial found that, compared with sham acupuncture and a control group receiving no therapy, women receiving acupuncture reported significantly lower scores on the Brief Pain Inventory–Short Form (BPI).
“We have shown consistently, with multiple measures assessing pain and stiffness, that true acupuncture generated better outcomes than either control group in a large multicenter trial,” said lead author Dawn L. Hershman, MD, leader of the Breast Cancer Program at the Herbert Irving Comprehensive Cancer Center at NewYork-Presbyterian/Columbia University Medical Center. “Acupuncture provides a nonpharmacologic option that can improve symptoms and possibly increase AI adherence and subsequent breast cancer outcomes.”
AIs can reduce both early breast cancer recurrence and mortality. Dr. Hershman noted that these agents are effective in the adjuvant setting and for prevention “but we know that it doesn’t work if you don’t take it. Noncompliance is a major problem among women taking hormonal therapy.”
Noncompliance is multifactorial and one of the main reasons women discontinue their therapy early is because of arthralgias or joint discomfort. “We were interested in a nonpharmacologic intervention, to assess whether or not we could control these symptoms.”
Dr. Hershman pointed out acupuncture provides a safe and effective alternative for patients reluctant to take a prescription medication that can result in other side effects. “Identification of nonopioid options for pain control is a public health priority,” she said.
Acupuncture is a popular nonpharmacologic modality and widely used for a number of indications. Several single-institution studies have suggested that it may be useful for controlling AI-associated arthralgias, while other studies have not demonstrated a benefit.
In this trial, the authors evaluated the efficacy of acupuncture, compared with sham acupuncture or waitlist control, in the treatment of AI associated arthralgia in a large population of patients. The study was conducted at 11 centers.
The cohort comprised 226 postmenopausal women diagnosed with early-stage, hormone receptor–positive breast cancer who were receiving treatment with AIs. The primary endpoint was the decline in joint pain as measured by BPI-SF at 6 weeks, and to assess the duration of the effect, the women were followed for an additional 12 weeks.
Within this group, 110 were randomized to true acupuncture; 59 to sham acupuncture, and 57 to waitlist control (no treatment). Patients receiving true or sham acupuncture had sessions three times a week for 6 weeks followed by one session per week for 6 more weeks. Pain status was reported at baseline, during treatment, and then afterwards, using a variety of measurement tools including the BPI-SF, which is a self-administered 14-item questionnaire that evaluates pain severity on a 0-10 scale, and the impact of pain on activities of daily living.
At 6 weeks, the true acupuncture treatment arm reported significantly lower BPI worst pain scores than those in the sham acupuncture and the waitlist control arms. The mean BPI worst pain for the true acupuncture arm was 0.92 points lower than the sham acupuncture arm (P = .01) and 0.96 points lower than the waitlist control arm (P = .01). The proportion of patients experiencing a large reduction in BPI worst pain (greater than 2) was significantly greater in the true acupuncture arm, compared with the other groups: 58% versus 33% percent and 31%, respectively. The differences continued to remain statistically significant at 24 weeks, even though the treatment only continued for 12 weeks.
Associated adverse effects were minimal with true and sham acupuncture and limited to grade 1 bruising.
The cost of the 12-week intervention was about $1,250 or $65-$75 a session. “We feel that there is now sufficient evidence to support insurance coverage of acupuncture of AI arthralgia.”
In a discussion of the paper, Dr. Anne Partridge, from the Dana Farber Cancer Center, noted that it is imperative to seek new ways to improve outcomes in breast cancer, and AIs are contributing to that. However, she echoed the concern that nonadherence to treatment is a “tremendous problem” and hampers the clinical effectiveness of AI therapy.
The rate of discontinuation during the first year of therapy is 20% within the first year and up to 40% of patients do not take them daily. Both early discontinuation and nonadherence contribute to mortality.
Based on these results from the largest randomized controlled trial looking at acupuncture in this setting, should physicians be recommending acupuncture to patients prescribed AI therapy?
“The short answer is, why not?” said Dr. Partridge, “And that we should be recommending it for some of our patients.”
However, there are a number of issues that need to be addressed, she added. The duration of treatment is not known, and the need for follow-up treatment or the frequency of it is not known. The generalizability of it is also unclear when looking at a larger population, and acupuncture is highly operator dependent.
“There are cost and access issues, and insurance right now offers very limited coverage,” she said.
Importantly, Dr. Partridge emphasized, “We know that it will help symptoms, but will it improve adherence to AI?”
It may improve adherence for some patients, but “side effects are only one factor,” she said. “Adherence behavior is complicated. We need to figure out how to optimize these therapies in our patients.”
This study was supported by the National Institutes of Health National Center for Complementary and Integrative Health and the Office of Research on Women’s Health, and grants from the NIH/National Cancer Institute Division of Cancer Prevention. Dr. Hershman declared no conflicts of interest. Dr. Partridge had no disclosures.
SOURCE: Hershman et al. Abstract GS4-04
REPORTING FROM SABCS 2017
Key clinical point: Acupuncture significantly reduced joint pain associated with the use of aromatase inhibitors, compared with sham acupuncture and untreated controls.
Major finding: The proportion of patients who experienced a large reduction in BPI worst pain (less than 2) was significantly greater in the true acupuncture arm, compared with the other groups: 58% versus 33% percent and 31%, respectively.
Data source: Three-arm randomized phase 3 trial that included 226 patients with early-stage hormone receptor–positive breast cancer who were receiving treatment with AIs.
Disclosures: This study was supported by the National Institutes of Health National Center for Complementary and Integrative Health and the Office of Research on Women’s Health, and grants from the NIH/National Cancer Institute Division of Cancer Prevention. Dr. Hershman declared no conflicts of interest. Dr. Partridge had no disclosures.
Source: Hershman et al. Abstract GS4-04
VIDEO: 5 years of additional AI no better than 2 in HR+ breast cancer
SAN ANTONIO – Clinical trials have shown a clear benefit for preventing breast cancer recurrence with aromatase inhibitor (AI) therapy following 5 years of tamoxifen. Yet the optimal duration for additional AI therapy following 5 years of endocrine therapy with tamoxifen, an AI, or sequential therapies is not known, according to Michael Gnant, MD, from the Medical University of Vienna.
In the ABCSG-16 trial, Dr. Gnant and his colleagues reported that 5 years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer.
In this video interview at the San Antonio Breast Cancer Symposium, Dr. Gnant notes that, although some patients may still benefit from 5 years of additional therapy, the trial results suggest that most patients can be spared from such adverse events as risk for fractures associated with three additional and evidently unnecessary years of therapy.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from the company and others.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Clinical trials have shown a clear benefit for preventing breast cancer recurrence with aromatase inhibitor (AI) therapy following 5 years of tamoxifen. Yet the optimal duration for additional AI therapy following 5 years of endocrine therapy with tamoxifen, an AI, or sequential therapies is not known, according to Michael Gnant, MD, from the Medical University of Vienna.
In the ABCSG-16 trial, Dr. Gnant and his colleagues reported that 5 years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer.
In this video interview at the San Antonio Breast Cancer Symposium, Dr. Gnant notes that, although some patients may still benefit from 5 years of additional therapy, the trial results suggest that most patients can be spared from such adverse events as risk for fractures associated with three additional and evidently unnecessary years of therapy.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from the company and others.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Clinical trials have shown a clear benefit for preventing breast cancer recurrence with aromatase inhibitor (AI) therapy following 5 years of tamoxifen. Yet the optimal duration for additional AI therapy following 5 years of endocrine therapy with tamoxifen, an AI, or sequential therapies is not known, according to Michael Gnant, MD, from the Medical University of Vienna.
In the ABCSG-16 trial, Dr. Gnant and his colleagues reported that 5 years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer.
In this video interview at the San Antonio Breast Cancer Symposium, Dr. Gnant notes that, although some patients may still benefit from 5 years of additional therapy, the trial results suggest that most patients can be spared from such adverse events as risk for fractures associated with three additional and evidently unnecessary years of therapy.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from the company and others.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
REPORTING FROM SABCS 2017