User login
No evidence of subclinical axial involvement seen in skin psoriasis
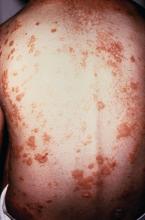
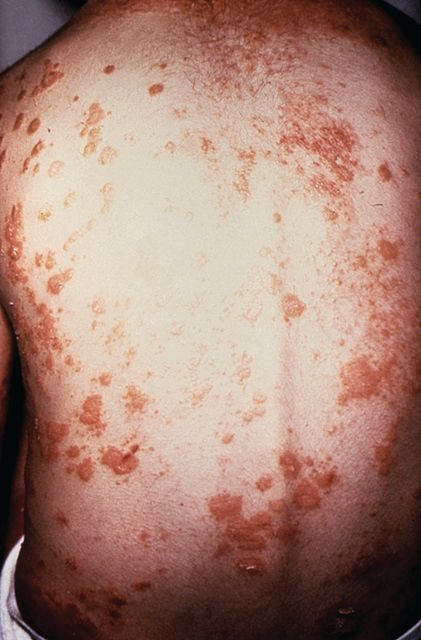
A study of individuals with longstanding skin psoriasis but no clinical arthritis or spondylitis has found no evidence of subclinical involvement of the sacroiliac joint or spine.
The prevalence of sacroiliac lesions on blinded MRI assessment was similar in 20 patients who had skin psoriasis for a median of 23 years and in 22 healthy controls, and no sacroiliac ankylosis was seen in either group. Similarly, there was no significant difference between the two groups in spinal lesions on MRI, nor in any of the five levels of lesion frequency, Vlad A. Bratu, MD, of the department of radiology at University Hospital Basel (Switzerland) and his coauthors reported in Arthritis Care & Research.
On blinded MRI assessment, five (25%) patients with skin psoriasis and two (9.1%) controls were classified as having inflammation of the sacroiliac joint. Three of these patients in the psoriasis group and one in the control group were older than 50, and the three with psoriasis had had the condition for 26-35 years.
Dr. Bratu and his colleagues said that subclinical peripheral joint inflammation on MRI had previously been a common finding in patients who had skin psoriasis but no clinical signs of psoriatic arthritis. But given the limited evidence of concomitant subclinical axial or spinal inflammation in their study, the authors argued there was no support for routine screening for potential subclinical axial inflammation in patients with longstanding skin psoriasis.
They noted that bone marrow edema lesions in at least two sacroiliac joint quadrants were seen in 35% of patients with psoriasis and 23% of healthy controls, a finding that reflected those seen in other studies in healthy individuals.
“If a specificity threshold for a given MRI lesion of at least 0.9 is applied for axial MRI to discriminate between axial SpA [spondyloarthritis] and background variation in healthy controls or in differential diagnostic conditions, no more than 10% of healthy controls in our study should meet this criterion by an individual level data analysis,” they wrote.
The authors also pointed out the impact of age on lesion frequency, which was more evident in spinal lesions.
“This observation supports the hypothesis that some spinal alterations in higher age may reflect degenerative rather than inflammatory changes,” they wrote. “However, there is a gap in knowledge with virtually no evidence about presence and pattern of degenerative versus inflammatory spinal lesions in subjects beyond 50 years of age.”
The study was supported by the Gottfried and Julia Bangerter-Rhyner Foundation. No conflicts of interest were declared.
SOURCE: Bratu V et al. Arthritis Care Res. 2018 Sep 22. doi: 10.1002/acr.23767.
A study of individuals with longstanding skin psoriasis but no clinical arthritis or spondylitis has found no evidence of subclinical involvement of the sacroiliac joint or spine.
The prevalence of sacroiliac lesions on blinded MRI assessment was similar in 20 patients who had skin psoriasis for a median of 23 years and in 22 healthy controls, and no sacroiliac ankylosis was seen in either group. Similarly, there was no significant difference between the two groups in spinal lesions on MRI, nor in any of the five levels of lesion frequency, Vlad A. Bratu, MD, of the department of radiology at University Hospital Basel (Switzerland) and his coauthors reported in Arthritis Care & Research.
On blinded MRI assessment, five (25%) patients with skin psoriasis and two (9.1%) controls were classified as having inflammation of the sacroiliac joint. Three of these patients in the psoriasis group and one in the control group were older than 50, and the three with psoriasis had had the condition for 26-35 years.
Dr. Bratu and his colleagues said that subclinical peripheral joint inflammation on MRI had previously been a common finding in patients who had skin psoriasis but no clinical signs of psoriatic arthritis. But given the limited evidence of concomitant subclinical axial or spinal inflammation in their study, the authors argued there was no support for routine screening for potential subclinical axial inflammation in patients with longstanding skin psoriasis.
They noted that bone marrow edema lesions in at least two sacroiliac joint quadrants were seen in 35% of patients with psoriasis and 23% of healthy controls, a finding that reflected those seen in other studies in healthy individuals.
“If a specificity threshold for a given MRI lesion of at least 0.9 is applied for axial MRI to discriminate between axial SpA [spondyloarthritis] and background variation in healthy controls or in differential diagnostic conditions, no more than 10% of healthy controls in our study should meet this criterion by an individual level data analysis,” they wrote.
The authors also pointed out the impact of age on lesion frequency, which was more evident in spinal lesions.
“This observation supports the hypothesis that some spinal alterations in higher age may reflect degenerative rather than inflammatory changes,” they wrote. “However, there is a gap in knowledge with virtually no evidence about presence and pattern of degenerative versus inflammatory spinal lesions in subjects beyond 50 years of age.”
The study was supported by the Gottfried and Julia Bangerter-Rhyner Foundation. No conflicts of interest were declared.
SOURCE: Bratu V et al. Arthritis Care Res. 2018 Sep 22. doi: 10.1002/acr.23767.
A study of individuals with longstanding skin psoriasis but no clinical arthritis or spondylitis has found no evidence of subclinical involvement of the sacroiliac joint or spine.
The prevalence of sacroiliac lesions on blinded MRI assessment was similar in 20 patients who had skin psoriasis for a median of 23 years and in 22 healthy controls, and no sacroiliac ankylosis was seen in either group. Similarly, there was no significant difference between the two groups in spinal lesions on MRI, nor in any of the five levels of lesion frequency, Vlad A. Bratu, MD, of the department of radiology at University Hospital Basel (Switzerland) and his coauthors reported in Arthritis Care & Research.
On blinded MRI assessment, five (25%) patients with skin psoriasis and two (9.1%) controls were classified as having inflammation of the sacroiliac joint. Three of these patients in the psoriasis group and one in the control group were older than 50, and the three with psoriasis had had the condition for 26-35 years.
Dr. Bratu and his colleagues said that subclinical peripheral joint inflammation on MRI had previously been a common finding in patients who had skin psoriasis but no clinical signs of psoriatic arthritis. But given the limited evidence of concomitant subclinical axial or spinal inflammation in their study, the authors argued there was no support for routine screening for potential subclinical axial inflammation in patients with longstanding skin psoriasis.
They noted that bone marrow edema lesions in at least two sacroiliac joint quadrants were seen in 35% of patients with psoriasis and 23% of healthy controls, a finding that reflected those seen in other studies in healthy individuals.
“If a specificity threshold for a given MRI lesion of at least 0.9 is applied for axial MRI to discriminate between axial SpA [spondyloarthritis] and background variation in healthy controls or in differential diagnostic conditions, no more than 10% of healthy controls in our study should meet this criterion by an individual level data analysis,” they wrote.
The authors also pointed out the impact of age on lesion frequency, which was more evident in spinal lesions.
“This observation supports the hypothesis that some spinal alterations in higher age may reflect degenerative rather than inflammatory changes,” they wrote. “However, there is a gap in knowledge with virtually no evidence about presence and pattern of degenerative versus inflammatory spinal lesions in subjects beyond 50 years of age.”
The study was supported by the Gottfried and Julia Bangerter-Rhyner Foundation. No conflicts of interest were declared.
SOURCE: Bratu V et al. Arthritis Care Res. 2018 Sep 22. doi: 10.1002/acr.23767.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point:
Major finding: The prevalence of sacroiliac bone marrow lesions was similar between patients with skin psoriasis and healthy controls.
Study details: Case-control study in 20 patients with skin psoriasis and 22 healthy controls.
Disclosures: The study was supported by the Gottfried and Julia Bangerter-Rhyner Foundation. No conflicts of interest were declared.
Source: Bratu V et al. Arthritis Care Res. 2018 Sep 22. doi: 10.1002/acr.23767.
Pregnancy registries are a valuable resource for dermatologists
Pregnancy registries are valuable sources of information. For many drugs, they are the primary source of the human pregnancy experience. However, although most of the registries use the word “pregnancy,” it is important to note that many also enroll women who took the target drug shortly before conception.
The strengths of these registries are their prospective nature (enrolled before the outcome is known) and enrollment over a wide geographical area. Typically, two types of pregnancy outcomes are obtained: those with birth defects and those without known birth defects (classified as live births, fetal deaths, and spontaneous abortions). Registries can identify early signals of teratogenicity, but they have several limitations: selection bias that results from voluntary reporting; target populations that are not representative; lost-to-follow-up pregnancies that may have had different outcomes than those with documented outcomes; elective terminations and fetal deaths without birth defects and spontaneous abortions, all of which may lack details; the lack of control groups (with some exceptions); and the publication of results that may be delayed or not be in a peer-reviewed journal. Because the total number of exposed pregnancies is unknown, the data cannot be used to calculate prevalences, but they can be used to estimate the proportion of birth defects. Some registries also collect data on retrospective reports (reported after outcome is known). Such reports are less representative of the target population because they can be biased toward the reporting of more unusual and severe outcomes. But they may be helpful in detecting unusual patterns of birth defects.
For the following drugs, web addresses can be obtained from the Food and Drug Administration website, List of Pregnancy Exposure Registries.
MotherToBaby
A large registry, the MotherToBaby Organization of Teratology Information Specialists (OTIS) (877-311-8972), involves patients in several different categories and the effects of the drugs on the embryo-fetus: autoimmune diseases (ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, psoriasis, Crohn’s disease, and multiple sclerosis); asthma at less than 20 weeks’ gestation; vaccines; and heterozygous or homozygous familial hypercholesterolemia.
For the autoimmune diseases, the drugs and trade names are abatacept (Orencia), adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), infliximab (Remicade), leflunomide (Arava), otezla (Apremilast), teriflunomide (Aubagio), tocilizumab (Actemra), tofacitinib (Xeljanz), and ustekinumab (Stelara).
For the asthma group, the drug being investigated is mepolizumab (Nucala).
Two vaccines – for tetanus, diphtheria, and pertussis (Tdap) and meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menveo) – are being studied.
The last category is heterozygous or homozygous familial hypercholesterolemia. The two agents in this category are alirocumab (Praluent) and evolocumab (Repatha).
Pregnancy registries are valuable sources of information. For many drugs, they are the primary source of the human pregnancy experience. However, although most of the registries use the word “pregnancy,” it is important to note that many also enroll women who took the target drug shortly before conception.
The strengths of these registries are their prospective nature (enrolled before the outcome is known) and enrollment over a wide geographical area. Typically, two types of pregnancy outcomes are obtained: those with birth defects and those without known birth defects (classified as live births, fetal deaths, and spontaneous abortions). Registries can identify early signals of teratogenicity, but they have several limitations: selection bias that results from voluntary reporting; target populations that are not representative; lost-to-follow-up pregnancies that may have had different outcomes than those with documented outcomes; elective terminations and fetal deaths without birth defects and spontaneous abortions, all of which may lack details; the lack of control groups (with some exceptions); and the publication of results that may be delayed or not be in a peer-reviewed journal. Because the total number of exposed pregnancies is unknown, the data cannot be used to calculate prevalences, but they can be used to estimate the proportion of birth defects. Some registries also collect data on retrospective reports (reported after outcome is known). Such reports are less representative of the target population because they can be biased toward the reporting of more unusual and severe outcomes. But they may be helpful in detecting unusual patterns of birth defects.
For the following drugs, web addresses can be obtained from the Food and Drug Administration website, List of Pregnancy Exposure Registries.
MotherToBaby
A large registry, the MotherToBaby Organization of Teratology Information Specialists (OTIS) (877-311-8972), involves patients in several different categories and the effects of the drugs on the embryo-fetus: autoimmune diseases (ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, psoriasis, Crohn’s disease, and multiple sclerosis); asthma at less than 20 weeks’ gestation; vaccines; and heterozygous or homozygous familial hypercholesterolemia.
For the autoimmune diseases, the drugs and trade names are abatacept (Orencia), adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), infliximab (Remicade), leflunomide (Arava), otezla (Apremilast), teriflunomide (Aubagio), tocilizumab (Actemra), tofacitinib (Xeljanz), and ustekinumab (Stelara).
For the asthma group, the drug being investigated is mepolizumab (Nucala).
Two vaccines – for tetanus, diphtheria, and pertussis (Tdap) and meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menveo) – are being studied.
The last category is heterozygous or homozygous familial hypercholesterolemia. The two agents in this category are alirocumab (Praluent) and evolocumab (Repatha).
Pregnancy registries are valuable sources of information. For many drugs, they are the primary source of the human pregnancy experience. However, although most of the registries use the word “pregnancy,” it is important to note that many also enroll women who took the target drug shortly before conception.
The strengths of these registries are their prospective nature (enrolled before the outcome is known) and enrollment over a wide geographical area. Typically, two types of pregnancy outcomes are obtained: those with birth defects and those without known birth defects (classified as live births, fetal deaths, and spontaneous abortions). Registries can identify early signals of teratogenicity, but they have several limitations: selection bias that results from voluntary reporting; target populations that are not representative; lost-to-follow-up pregnancies that may have had different outcomes than those with documented outcomes; elective terminations and fetal deaths without birth defects and spontaneous abortions, all of which may lack details; the lack of control groups (with some exceptions); and the publication of results that may be delayed or not be in a peer-reviewed journal. Because the total number of exposed pregnancies is unknown, the data cannot be used to calculate prevalences, but they can be used to estimate the proportion of birth defects. Some registries also collect data on retrospective reports (reported after outcome is known). Such reports are less representative of the target population because they can be biased toward the reporting of more unusual and severe outcomes. But they may be helpful in detecting unusual patterns of birth defects.
For the following drugs, web addresses can be obtained from the Food and Drug Administration website, List of Pregnancy Exposure Registries.
MotherToBaby
A large registry, the MotherToBaby Organization of Teratology Information Specialists (OTIS) (877-311-8972), involves patients in several different categories and the effects of the drugs on the embryo-fetus: autoimmune diseases (ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, psoriasis, Crohn’s disease, and multiple sclerosis); asthma at less than 20 weeks’ gestation; vaccines; and heterozygous or homozygous familial hypercholesterolemia.
For the autoimmune diseases, the drugs and trade names are abatacept (Orencia), adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), infliximab (Remicade), leflunomide (Arava), otezla (Apremilast), teriflunomide (Aubagio), tocilizumab (Actemra), tofacitinib (Xeljanz), and ustekinumab (Stelara).
For the asthma group, the drug being investigated is mepolizumab (Nucala).
Two vaccines – for tetanus, diphtheria, and pertussis (Tdap) and meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menveo) – are being studied.
The last category is heterozygous or homozygous familial hypercholesterolemia. The two agents in this category are alirocumab (Praluent) and evolocumab (Repatha).
Pregnancy registries are a valuable resource
Pregnancy registries are valuable sources of information. For many drugs, they are the primary source of the human pregnancy experience. However, although most of the registries use the word “pregnancy,” it is important to note that many also enroll women who took the target drug shortly before conception.
The strengths of these registries are their prospective nature (enrolled before the outcome is known) and enrollment over a wide geographical area. Typically, two types of pregnancy outcomes are obtained: those with birth defects and those without known birth defects (classified as live births, fetal deaths, and spontaneous abortions). Registries can identify early signals of teratogenicity, but they have several limitations: selection bias that results from voluntary reporting; target populations that are not representative; lost-to-follow-up pregnancies that may have had different outcomes than those with documented outcomes; elective terminations and fetal deaths without birth defects and spontaneous abortions, all of which may lack details; the lack of control groups (with some exceptions); and the publication of results that may be delayed or not be in a peer-reviewed journal. Because the total number of exposed pregnancies is unknown, the data cannot be used to calculate prevalences, but they can be used to estimate the proportion of birth defects. Some registries also collect data on retrospective reports (reported after outcome is known). Such reports are less representative of the target population because they can be biased toward the reporting of more unusual and severe outcomes. But they may be helpful in detecting unusual patterns of birth defects.
For the following drugs, web addresses can be obtained from the Food and Drug Administration website, List of Pregnancy Exposure Registries.
MotherToBaby
A large registry, the MotherToBaby Organization of Teratology Information Specialists (OTIS) (877-311-8972), involves patients in several different categories and the effects of the drugs on the embryo-fetus: autoimmune diseases (ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, psoriasis, Crohn’s disease, and multiple sclerosis); asthma at less than 20 weeks’ gestation; vaccines; and heterozygous or homozygous familial hypercholesterolemia.
For the autoimmune diseases, the drugs and trade names are abatacept (Orencia), adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), infliximab (Remicade), leflunomide (Arava), otezla (Apremilast), teriflunomide (Aubagio), tocilizumab (Actemra), tofacitinib (Xeljanz), and ustekinumab (Stelara).
For the asthma group, the drug being investigated is mepolizumab (Nucala).
Two vaccines – for tetanus, diphtheria, and pertussis (Tdap) and meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menveo) – are being studied.
The last category is heterozygous or homozygous familial hypercholesterolemia. The two agents in this category are alirocumab (Praluent) and evolocumab (Repatha).
Other registries
Breast cancer
The Mother Pregnancy Registry, INC Research (800-690-6720), is enrolling breast cancer patients who have been treated during pregnancy with ado-trastuzumab emtansine (Kadcyla), pertuzumab (Perjeta), or trastuzumab (Herceptin).
Epilepsy
The Antiepileptic Drug Pregnancy registry (888-233-2334) is studying eslicarbazepine (Aptiom) and pregabalin (Lyrica).
Fabry disease
The Fabry Registry, Genzyme Corp (617-591-5500) is studying the use in pregnancy of agalsidase beta (Fabrazyme) for Fabry disease.
Fibromyalgia
The Savella Pregnancy Registry (877-643-3010) is looking for patients with fibromyalgia who are being treated with milnacipran (Savella).
Hepatitis B
The Ribavirin Pregnancy Registry, INC Research (800-593-2214) is looking for subjects with hepatitis C who have been treated with ribavirin (Copegus).
Hypercholesterolemia
Lomitapide (Juxtapid) is being studied by the Global Lomitapide Pregnancy Exposure Registry managed by Aegerion (877-902-4099). The drug is used for homozygous familial hypercholesterolemia.
Mucopolysaccharidosis
The Mucopolysaccharidosis I (MPS I) registry, Genzyme (617-591-5500) is studying the use of laronidase (Aldurazyme) for Hurler syndrome, Scheie syndrome, and Hurler-Scheie syndrome.
The use of galsulfase (Naglazyme) for Maroteaux-Lamy syndrome during pregnancy is under study by the Mucopolysaccharidosis VI (MPS VI), clinical surveillance program (BioMarin Pharmaceutical) (415-506-6849 or 415-506-6703).
Multiple sclerosis
Novartis is conducting the Gilenya Pregnancy Registry (877-598-7237) for patients with multiple sclerosis who are taking fingolimod (Gilenya).
Alemtuzumab (Lemtrada), also indicated for multiple sclerosis, is the target agent for the LEMTRADA Pregnancy Exposure Registry (866-758-2990).
Narcolepsy and other sleep disorders
Armodafinil (Nuvigil), used for excessive sleepiness associated with narcolepsy and other sleep disorders, is being studied in the Nuvigil Pregnancy Registry (866-404-4106). A second drug with the same indication and telephone number, modafinil (Provigil), is in the Provigil Pregnancy Registry.
Osteoporosis
Amgen’s Pregnancy Surveillance Program (800-772-6436) is enrolling pregnant subjects with osteoporosis who are being treated with denosumab (Prolia).
Others
Two Merck pregnancy registries (800-986-8999) cover the following conditions: type 2 diabetes sitagliptin+metformin (Janumet) or sitagliptin (Januvia); and migraine headaches rizatriptan (Maxalt).
GlaxoSmithKline is conducting two registries: the Belimumab Pregnancy Registry for patients with systemic lupus erythematosus treated with belimumab (Benlysta) (877-681-6296); and Promacta Pregnancy Registry for women treated for thrombocytopenia with eltrombopag (Promacta) (888-825-5249).
Psychiatric Drugs
The National Pregnancy Registry for Atypical Antipsychotics (866-961-2388) is studying 10 drugs: aripiprazole (Abilify), asenapine (Saphris), clozapine (Clozaril), iloperidone (Fanapt), lurasidone (Latuda), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal), and ziprasidone (Geodon).
The National Pregnancy Registry for Antidepressants (844-405-6185) is studying amitriptyline (Elavil), amoxapine (Asendin), bupropion (Forfivo XL and Wellbutrin), citalopram (Celexa), clomipramine (Anafranil), desipramine (Norpramin), desvenlafaxine (Prisiq), doxepin (Sinequan), escitalopram (Lexapro), fluvoxamine (Luvox), fluoxetine (Prozac), imipramine (Tofranil), isocarboxazid (Marplan), levomilnacipran (Fetzima), maprotiline (Ludiomil), mirtazapine (Remeron), nefazodone (Serzone), nortriptyline (Pamelor), paroxetine (Paxil), phenelzine (Nardill), protriptyline (Vivactil), selegiline (Emsam), sertraline (Zoloft), tranylcypromine (Pamate), trazodone (Desyrel), trimipramine (Surmontil), venlafaxine (Effexor), and vilazodone (Viibryd).
The National Pregnancy Registry of Psychostimulants (866-961-2388) is studying amphetamine (Adderall), dextroamphetamine (Dexedrine and Focalin), lisdexamfetamine (Vyvanse), methylphenidate (Concerta, Daytrana, Desoxyn, Ritalin), and modafinil (Provigil).
The antidepressant duloxetine (Cymbalta) is being studied by the Cymbalta Pregnancy Registry (866-814-6975).
Transplant patients
Renal transplant patients exposed to mycophenolate (CellCept) can be enrolled in the Transplantation Pregnancy Registry International (877-955-6877) or the Mycophenolate Pregnancy Registry (800-617-8191). The Transplantation Pregnancy Registry International also is enrolling renal transplant patients exposed to belatacept (Nulojix).
Vaccines
A quadrivalent influenza vaccine (Afluria) is being studied by the Seqirus Influenza Vaccine Pregnancy Registry (855-358-8972). A second vaccine for meningococcal disease meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menactra) is under study by the Menactra Vaccine Pregnancy Registry (800-822-2463). The Bexsero Pregnancy Registry (877-413-4759) is open to patients who have received the meningococcal group B vaccine (Bexsero). The Hepatitis B Vaccine [Recombinant] Adjuvanted Pregnancy Registry, also listed as HEPLISAV-B, is enrolling patients who have received that vaccine (844-443-7734); it is supported by the Dynavax Technologies Corporation.
Because the strength of a registry is based on numbers, health care professionals are encouraged to enroll potential subjects or have their patients call to enroll themselves.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at [email protected].
Pregnancy registries are valuable sources of information. For many drugs, they are the primary source of the human pregnancy experience. However, although most of the registries use the word “pregnancy,” it is important to note that many also enroll women who took the target drug shortly before conception.
The strengths of these registries are their prospective nature (enrolled before the outcome is known) and enrollment over a wide geographical area. Typically, two types of pregnancy outcomes are obtained: those with birth defects and those without known birth defects (classified as live births, fetal deaths, and spontaneous abortions). Registries can identify early signals of teratogenicity, but they have several limitations: selection bias that results from voluntary reporting; target populations that are not representative; lost-to-follow-up pregnancies that may have had different outcomes than those with documented outcomes; elective terminations and fetal deaths without birth defects and spontaneous abortions, all of which may lack details; the lack of control groups (with some exceptions); and the publication of results that may be delayed or not be in a peer-reviewed journal. Because the total number of exposed pregnancies is unknown, the data cannot be used to calculate prevalences, but they can be used to estimate the proportion of birth defects. Some registries also collect data on retrospective reports (reported after outcome is known). Such reports are less representative of the target population because they can be biased toward the reporting of more unusual and severe outcomes. But they may be helpful in detecting unusual patterns of birth defects.
For the following drugs, web addresses can be obtained from the Food and Drug Administration website, List of Pregnancy Exposure Registries.
MotherToBaby
A large registry, the MotherToBaby Organization of Teratology Information Specialists (OTIS) (877-311-8972), involves patients in several different categories and the effects of the drugs on the embryo-fetus: autoimmune diseases (ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, psoriasis, Crohn’s disease, and multiple sclerosis); asthma at less than 20 weeks’ gestation; vaccines; and heterozygous or homozygous familial hypercholesterolemia.
For the autoimmune diseases, the drugs and trade names are abatacept (Orencia), adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), infliximab (Remicade), leflunomide (Arava), otezla (Apremilast), teriflunomide (Aubagio), tocilizumab (Actemra), tofacitinib (Xeljanz), and ustekinumab (Stelara).
For the asthma group, the drug being investigated is mepolizumab (Nucala).
Two vaccines – for tetanus, diphtheria, and pertussis (Tdap) and meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menveo) – are being studied.
The last category is heterozygous or homozygous familial hypercholesterolemia. The two agents in this category are alirocumab (Praluent) and evolocumab (Repatha).
Other registries
Breast cancer
The Mother Pregnancy Registry, INC Research (800-690-6720), is enrolling breast cancer patients who have been treated during pregnancy with ado-trastuzumab emtansine (Kadcyla), pertuzumab (Perjeta), or trastuzumab (Herceptin).
Epilepsy
The Antiepileptic Drug Pregnancy registry (888-233-2334) is studying eslicarbazepine (Aptiom) and pregabalin (Lyrica).
Fabry disease
The Fabry Registry, Genzyme Corp (617-591-5500) is studying the use in pregnancy of agalsidase beta (Fabrazyme) for Fabry disease.
Fibromyalgia
The Savella Pregnancy Registry (877-643-3010) is looking for patients with fibromyalgia who are being treated with milnacipran (Savella).
Hepatitis B
The Ribavirin Pregnancy Registry, INC Research (800-593-2214) is looking for subjects with hepatitis C who have been treated with ribavirin (Copegus).
Hypercholesterolemia
Lomitapide (Juxtapid) is being studied by the Global Lomitapide Pregnancy Exposure Registry managed by Aegerion (877-902-4099). The drug is used for homozygous familial hypercholesterolemia.
Mucopolysaccharidosis
The Mucopolysaccharidosis I (MPS I) registry, Genzyme (617-591-5500) is studying the use of laronidase (Aldurazyme) for Hurler syndrome, Scheie syndrome, and Hurler-Scheie syndrome.
The use of galsulfase (Naglazyme) for Maroteaux-Lamy syndrome during pregnancy is under study by the Mucopolysaccharidosis VI (MPS VI), clinical surveillance program (BioMarin Pharmaceutical) (415-506-6849 or 415-506-6703).
Multiple sclerosis
Novartis is conducting the Gilenya Pregnancy Registry (877-598-7237) for patients with multiple sclerosis who are taking fingolimod (Gilenya).
Alemtuzumab (Lemtrada), also indicated for multiple sclerosis, is the target agent for the LEMTRADA Pregnancy Exposure Registry (866-758-2990).
Narcolepsy and other sleep disorders
Armodafinil (Nuvigil), used for excessive sleepiness associated with narcolepsy and other sleep disorders, is being studied in the Nuvigil Pregnancy Registry (866-404-4106). A second drug with the same indication and telephone number, modafinil (Provigil), is in the Provigil Pregnancy Registry.
Osteoporosis
Amgen’s Pregnancy Surveillance Program (800-772-6436) is enrolling pregnant subjects with osteoporosis who are being treated with denosumab (Prolia).
Others
Two Merck pregnancy registries (800-986-8999) cover the following conditions: type 2 diabetes sitagliptin+metformin (Janumet) or sitagliptin (Januvia); and migraine headaches rizatriptan (Maxalt).
GlaxoSmithKline is conducting two registries: the Belimumab Pregnancy Registry for patients with systemic lupus erythematosus treated with belimumab (Benlysta) (877-681-6296); and Promacta Pregnancy Registry for women treated for thrombocytopenia with eltrombopag (Promacta) (888-825-5249).
Psychiatric Drugs
The National Pregnancy Registry for Atypical Antipsychotics (866-961-2388) is studying 10 drugs: aripiprazole (Abilify), asenapine (Saphris), clozapine (Clozaril), iloperidone (Fanapt), lurasidone (Latuda), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal), and ziprasidone (Geodon).
The National Pregnancy Registry for Antidepressants (844-405-6185) is studying amitriptyline (Elavil), amoxapine (Asendin), bupropion (Forfivo XL and Wellbutrin), citalopram (Celexa), clomipramine (Anafranil), desipramine (Norpramin), desvenlafaxine (Prisiq), doxepin (Sinequan), escitalopram (Lexapro), fluvoxamine (Luvox), fluoxetine (Prozac), imipramine (Tofranil), isocarboxazid (Marplan), levomilnacipran (Fetzima), maprotiline (Ludiomil), mirtazapine (Remeron), nefazodone (Serzone), nortriptyline (Pamelor), paroxetine (Paxil), phenelzine (Nardill), protriptyline (Vivactil), selegiline (Emsam), sertraline (Zoloft), tranylcypromine (Pamate), trazodone (Desyrel), trimipramine (Surmontil), venlafaxine (Effexor), and vilazodone (Viibryd).
The National Pregnancy Registry of Psychostimulants (866-961-2388) is studying amphetamine (Adderall), dextroamphetamine (Dexedrine and Focalin), lisdexamfetamine (Vyvanse), methylphenidate (Concerta, Daytrana, Desoxyn, Ritalin), and modafinil (Provigil).
The antidepressant duloxetine (Cymbalta) is being studied by the Cymbalta Pregnancy Registry (866-814-6975).
Transplant patients
Renal transplant patients exposed to mycophenolate (CellCept) can be enrolled in the Transplantation Pregnancy Registry International (877-955-6877) or the Mycophenolate Pregnancy Registry (800-617-8191). The Transplantation Pregnancy Registry International also is enrolling renal transplant patients exposed to belatacept (Nulojix).
Vaccines
A quadrivalent influenza vaccine (Afluria) is being studied by the Seqirus Influenza Vaccine Pregnancy Registry (855-358-8972). A second vaccine for meningococcal disease meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menactra) is under study by the Menactra Vaccine Pregnancy Registry (800-822-2463). The Bexsero Pregnancy Registry (877-413-4759) is open to patients who have received the meningococcal group B vaccine (Bexsero). The Hepatitis B Vaccine [Recombinant] Adjuvanted Pregnancy Registry, also listed as HEPLISAV-B, is enrolling patients who have received that vaccine (844-443-7734); it is supported by the Dynavax Technologies Corporation.
Because the strength of a registry is based on numbers, health care professionals are encouraged to enroll potential subjects or have their patients call to enroll themselves.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at [email protected].
Pregnancy registries are valuable sources of information. For many drugs, they are the primary source of the human pregnancy experience. However, although most of the registries use the word “pregnancy,” it is important to note that many also enroll women who took the target drug shortly before conception.
The strengths of these registries are their prospective nature (enrolled before the outcome is known) and enrollment over a wide geographical area. Typically, two types of pregnancy outcomes are obtained: those with birth defects and those without known birth defects (classified as live births, fetal deaths, and spontaneous abortions). Registries can identify early signals of teratogenicity, but they have several limitations: selection bias that results from voluntary reporting; target populations that are not representative; lost-to-follow-up pregnancies that may have had different outcomes than those with documented outcomes; elective terminations and fetal deaths without birth defects and spontaneous abortions, all of which may lack details; the lack of control groups (with some exceptions); and the publication of results that may be delayed or not be in a peer-reviewed journal. Because the total number of exposed pregnancies is unknown, the data cannot be used to calculate prevalences, but they can be used to estimate the proportion of birth defects. Some registries also collect data on retrospective reports (reported after outcome is known). Such reports are less representative of the target population because they can be biased toward the reporting of more unusual and severe outcomes. But they may be helpful in detecting unusual patterns of birth defects.
For the following drugs, web addresses can be obtained from the Food and Drug Administration website, List of Pregnancy Exposure Registries.
MotherToBaby
A large registry, the MotherToBaby Organization of Teratology Information Specialists (OTIS) (877-311-8972), involves patients in several different categories and the effects of the drugs on the embryo-fetus: autoimmune diseases (ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, psoriasis, Crohn’s disease, and multiple sclerosis); asthma at less than 20 weeks’ gestation; vaccines; and heterozygous or homozygous familial hypercholesterolemia.
For the autoimmune diseases, the drugs and trade names are abatacept (Orencia), adalimumab (Humira), certolizumab pegol (Cimzia), etanercept (Enbrel), infliximab (Remicade), leflunomide (Arava), otezla (Apremilast), teriflunomide (Aubagio), tocilizumab (Actemra), tofacitinib (Xeljanz), and ustekinumab (Stelara).
For the asthma group, the drug being investigated is mepolizumab (Nucala).
Two vaccines – for tetanus, diphtheria, and pertussis (Tdap) and meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menveo) – are being studied.
The last category is heterozygous or homozygous familial hypercholesterolemia. The two agents in this category are alirocumab (Praluent) and evolocumab (Repatha).
Other registries
Breast cancer
The Mother Pregnancy Registry, INC Research (800-690-6720), is enrolling breast cancer patients who have been treated during pregnancy with ado-trastuzumab emtansine (Kadcyla), pertuzumab (Perjeta), or trastuzumab (Herceptin).
Epilepsy
The Antiepileptic Drug Pregnancy registry (888-233-2334) is studying eslicarbazepine (Aptiom) and pregabalin (Lyrica).
Fabry disease
The Fabry Registry, Genzyme Corp (617-591-5500) is studying the use in pregnancy of agalsidase beta (Fabrazyme) for Fabry disease.
Fibromyalgia
The Savella Pregnancy Registry (877-643-3010) is looking for patients with fibromyalgia who are being treated with milnacipran (Savella).
Hepatitis B
The Ribavirin Pregnancy Registry, INC Research (800-593-2214) is looking for subjects with hepatitis C who have been treated with ribavirin (Copegus).
Hypercholesterolemia
Lomitapide (Juxtapid) is being studied by the Global Lomitapide Pregnancy Exposure Registry managed by Aegerion (877-902-4099). The drug is used for homozygous familial hypercholesterolemia.
Mucopolysaccharidosis
The Mucopolysaccharidosis I (MPS I) registry, Genzyme (617-591-5500) is studying the use of laronidase (Aldurazyme) for Hurler syndrome, Scheie syndrome, and Hurler-Scheie syndrome.
The use of galsulfase (Naglazyme) for Maroteaux-Lamy syndrome during pregnancy is under study by the Mucopolysaccharidosis VI (MPS VI), clinical surveillance program (BioMarin Pharmaceutical) (415-506-6849 or 415-506-6703).
Multiple sclerosis
Novartis is conducting the Gilenya Pregnancy Registry (877-598-7237) for patients with multiple sclerosis who are taking fingolimod (Gilenya).
Alemtuzumab (Lemtrada), also indicated for multiple sclerosis, is the target agent for the LEMTRADA Pregnancy Exposure Registry (866-758-2990).
Narcolepsy and other sleep disorders
Armodafinil (Nuvigil), used for excessive sleepiness associated with narcolepsy and other sleep disorders, is being studied in the Nuvigil Pregnancy Registry (866-404-4106). A second drug with the same indication and telephone number, modafinil (Provigil), is in the Provigil Pregnancy Registry.
Osteoporosis
Amgen’s Pregnancy Surveillance Program (800-772-6436) is enrolling pregnant subjects with osteoporosis who are being treated with denosumab (Prolia).
Others
Two Merck pregnancy registries (800-986-8999) cover the following conditions: type 2 diabetes sitagliptin+metformin (Janumet) or sitagliptin (Januvia); and migraine headaches rizatriptan (Maxalt).
GlaxoSmithKline is conducting two registries: the Belimumab Pregnancy Registry for patients with systemic lupus erythematosus treated with belimumab (Benlysta) (877-681-6296); and Promacta Pregnancy Registry for women treated for thrombocytopenia with eltrombopag (Promacta) (888-825-5249).
Psychiatric Drugs
The National Pregnancy Registry for Atypical Antipsychotics (866-961-2388) is studying 10 drugs: aripiprazole (Abilify), asenapine (Saphris), clozapine (Clozaril), iloperidone (Fanapt), lurasidone (Latuda), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal), and ziprasidone (Geodon).
The National Pregnancy Registry for Antidepressants (844-405-6185) is studying amitriptyline (Elavil), amoxapine (Asendin), bupropion (Forfivo XL and Wellbutrin), citalopram (Celexa), clomipramine (Anafranil), desipramine (Norpramin), desvenlafaxine (Prisiq), doxepin (Sinequan), escitalopram (Lexapro), fluvoxamine (Luvox), fluoxetine (Prozac), imipramine (Tofranil), isocarboxazid (Marplan), levomilnacipran (Fetzima), maprotiline (Ludiomil), mirtazapine (Remeron), nefazodone (Serzone), nortriptyline (Pamelor), paroxetine (Paxil), phenelzine (Nardill), protriptyline (Vivactil), selegiline (Emsam), sertraline (Zoloft), tranylcypromine (Pamate), trazodone (Desyrel), trimipramine (Surmontil), venlafaxine (Effexor), and vilazodone (Viibryd).
The National Pregnancy Registry of Psychostimulants (866-961-2388) is studying amphetamine (Adderall), dextroamphetamine (Dexedrine and Focalin), lisdexamfetamine (Vyvanse), methylphenidate (Concerta, Daytrana, Desoxyn, Ritalin), and modafinil (Provigil).
The antidepressant duloxetine (Cymbalta) is being studied by the Cymbalta Pregnancy Registry (866-814-6975).
Transplant patients
Renal transplant patients exposed to mycophenolate (CellCept) can be enrolled in the Transplantation Pregnancy Registry International (877-955-6877) or the Mycophenolate Pregnancy Registry (800-617-8191). The Transplantation Pregnancy Registry International also is enrolling renal transplant patients exposed to belatacept (Nulojix).
Vaccines
A quadrivalent influenza vaccine (Afluria) is being studied by the Seqirus Influenza Vaccine Pregnancy Registry (855-358-8972). A second vaccine for meningococcal disease meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135 (Menactra) is under study by the Menactra Vaccine Pregnancy Registry (800-822-2463). The Bexsero Pregnancy Registry (877-413-4759) is open to patients who have received the meningococcal group B vaccine (Bexsero). The Hepatitis B Vaccine [Recombinant] Adjuvanted Pregnancy Registry, also listed as HEPLISAV-B, is enrolling patients who have received that vaccine (844-443-7734); it is supported by the Dynavax Technologies Corporation.
Because the strength of a registry is based on numbers, health care professionals are encouraged to enroll potential subjects or have their patients call to enroll themselves.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at [email protected].
Novel oral agent shows unprecedented efficacy in psoriasis
PARIS – A novel in a phase 2 clinical trial including 267 adults with moderate to severe disease, James G. Krueger, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“I would say the clinical response here is almost dead-on as a copy for ustekinumab, which is an [injectable interleukin] IL-23/IL-12 blocker. And we’re only at 12 weeks here; some of the curves look like they’re on a trajectory to go up further in terms of improvement. So I’m getting a performance with an oral drug that is just so much better than the approved alternatives that we have,” said Dr. Krueger, head of the laboratory of investigative dermatology and professor in clinical investigation at Rockefeller University in New York.
Oral apremilast (Otezla), for example, can’t touch those PASI 75 response rates in patients with moderate to severe psoriasis. Indeed, many psoriasis experts favor reserving apremilast for patients with moderate disease.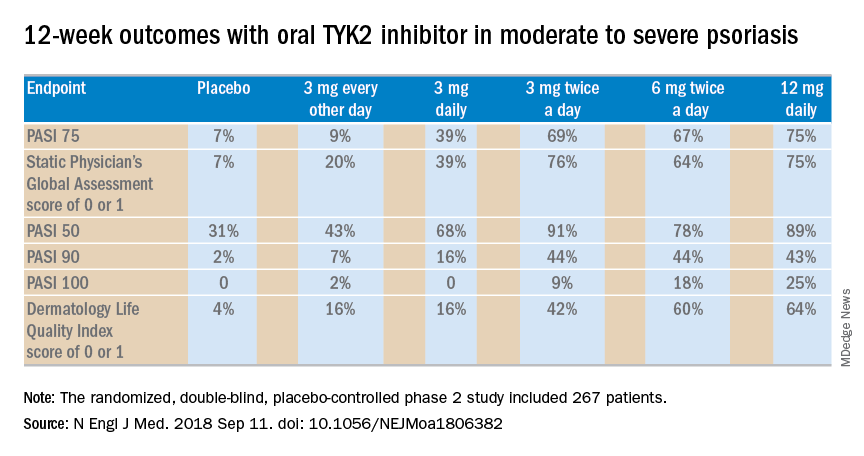
The 12-week, double-blind, placebo-controlled study was conducted at 82 sites in the United States and seven other countries. In this dose-ranging study, participants were randomized to the oral selective tyrosine kinase 2 (TYK2) inhibitor, known for the time being as BMS-986165, at 3 mg every other day, 3 mg daily, 3 mg twice a day, 6 mg twice a day, 12 mg daily, or to placebo.
The primary outcome was a 75% or greater reduction from baseline in Psoriasis Area and Severity Index score (PASI 75) at week 12. The TYK2 inhibitor outperformed placebo in dose-dependent fashion starting at the 3 mg/day dose. The PASI 75 rate was 7% with placebo, 9% with 3 mg of BMS-986165 every other day, 39% with 3 mg daily, 69% with 3 mg BID, 67% with 6 mg BID, and 75% with 12 mg/day. All secondary endpoints followed suit.
A striking finding in the phase 2 study was that when the drug was stopped for a month at the end of the 12-week treatment period, for the most part, the PASI 75 response and other clinical benefits were retained.
“I would contrast this to experiments that I have personally done with cyclosporine, where I have cleared people with cyclosporine, stopped it, and a month later every single patient has rip-roaring disease back. So I think this TYK2 inhibitor has some different performance features than just blocking a downstream T-cell transduction molecule,” observed the dermatologist, who is credited as the discoverer of the importance of the T cell in psoriasis pathogenesis.
The strong multidimensional evidence of clinical efficacy in the phase 2 study was supported mechanistically by analysis of skin biopsies obtained on study days 1, 15, and 85. The laboratory studies showed that the oral drug improved molecular, cellular, and clinical biomarkers associated with treatment efficacy. For example, at doses of 3 mg twice a day or higher, the TYK2 inhibitor reduced expression of IL-19 and IL-36A, which are key drivers of keratinocyte activation and epidermal hyperplasia. The drug also markedly decreased expression of genes in the Th17 pathway and essentially normalized expression of the proinflammatory genes beta defensin and S100A9.
In contrast to the Janus kinase (JAK) 1/3 and JAK 2 inhibitors in development for treatment of psoriasis, which paint with a much broader brush, the TYK2 inhibitor is highly selective for IL-23, IL-12, and interferon alpha.
“Previous studies have shown pan-JAK inhibition can be very effective in remitting psoriasis. The problem is that if one inhibits JAK1 and JAK3, one blocks the transduction of effector cytokines that are essentially there for protective immunity. That could lead to undesirable levels of immunosuppression,” Dr. Krueger explained.
The most important cytokine in the pathogenesis of psoriasis is clearly IL-23, he continued. In cell-based assays, the TYK2 inhibitor has been shown to be 100 times more selective in inhibiting IL-23 , IL-12, and interferon-alpha than JAK 1/3 inhibitors and 3,000 times more selective than JAK 2 inhibitors. This high degree of selectivity makes for fewer off-target effects and for a favorable safety profile.
“There were no major safety signals that would lead you to be concerned,” Dr. Krueger said. Indeed, based upon the encouraging safety and efficacy demonstrated this phase 2 study, a phase 3 program known as POETYK-PSO is underway (POETYK-PSO-1 and POETYK-PSO-2).
The phase 2 clinical trial results were published online in conjunction with the EADV congress.
The TYK2 inhibitor is being developed by Bristol-Myers Squibb. Dr. Krueger reported receiving personal fees as well as research grants paid directly to Rockefeller University from that pharmaceutical company and numerous others.
Source: Papp K et al. N Engl J Med. 2018 Sep 11. doi: 10.1056/NEJMoa1806382.
PARIS – A novel in a phase 2 clinical trial including 267 adults with moderate to severe disease, James G. Krueger, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“I would say the clinical response here is almost dead-on as a copy for ustekinumab, which is an [injectable interleukin] IL-23/IL-12 blocker. And we’re only at 12 weeks here; some of the curves look like they’re on a trajectory to go up further in terms of improvement. So I’m getting a performance with an oral drug that is just so much better than the approved alternatives that we have,” said Dr. Krueger, head of the laboratory of investigative dermatology and professor in clinical investigation at Rockefeller University in New York.
Oral apremilast (Otezla), for example, can’t touch those PASI 75 response rates in patients with moderate to severe psoriasis. Indeed, many psoriasis experts favor reserving apremilast for patients with moderate disease.
The 12-week, double-blind, placebo-controlled study was conducted at 82 sites in the United States and seven other countries. In this dose-ranging study, participants were randomized to the oral selective tyrosine kinase 2 (TYK2) inhibitor, known for the time being as BMS-986165, at 3 mg every other day, 3 mg daily, 3 mg twice a day, 6 mg twice a day, 12 mg daily, or to placebo.
The primary outcome was a 75% or greater reduction from baseline in Psoriasis Area and Severity Index score (PASI 75) at week 12. The TYK2 inhibitor outperformed placebo in dose-dependent fashion starting at the 3 mg/day dose. The PASI 75 rate was 7% with placebo, 9% with 3 mg of BMS-986165 every other day, 39% with 3 mg daily, 69% with 3 mg BID, 67% with 6 mg BID, and 75% with 12 mg/day. All secondary endpoints followed suit.
A striking finding in the phase 2 study was that when the drug was stopped for a month at the end of the 12-week treatment period, for the most part, the PASI 75 response and other clinical benefits were retained.
“I would contrast this to experiments that I have personally done with cyclosporine, where I have cleared people with cyclosporine, stopped it, and a month later every single patient has rip-roaring disease back. So I think this TYK2 inhibitor has some different performance features than just blocking a downstream T-cell transduction molecule,” observed the dermatologist, who is credited as the discoverer of the importance of the T cell in psoriasis pathogenesis.
The strong multidimensional evidence of clinical efficacy in the phase 2 study was supported mechanistically by analysis of skin biopsies obtained on study days 1, 15, and 85. The laboratory studies showed that the oral drug improved molecular, cellular, and clinical biomarkers associated with treatment efficacy. For example, at doses of 3 mg twice a day or higher, the TYK2 inhibitor reduced expression of IL-19 and IL-36A, which are key drivers of keratinocyte activation and epidermal hyperplasia. The drug also markedly decreased expression of genes in the Th17 pathway and essentially normalized expression of the proinflammatory genes beta defensin and S100A9.
In contrast to the Janus kinase (JAK) 1/3 and JAK 2 inhibitors in development for treatment of psoriasis, which paint with a much broader brush, the TYK2 inhibitor is highly selective for IL-23, IL-12, and interferon alpha.
“Previous studies have shown pan-JAK inhibition can be very effective in remitting psoriasis. The problem is that if one inhibits JAK1 and JAK3, one blocks the transduction of effector cytokines that are essentially there for protective immunity. That could lead to undesirable levels of immunosuppression,” Dr. Krueger explained.
The most important cytokine in the pathogenesis of psoriasis is clearly IL-23, he continued. In cell-based assays, the TYK2 inhibitor has been shown to be 100 times more selective in inhibiting IL-23 , IL-12, and interferon-alpha than JAK 1/3 inhibitors and 3,000 times more selective than JAK 2 inhibitors. This high degree of selectivity makes for fewer off-target effects and for a favorable safety profile.
“There were no major safety signals that would lead you to be concerned,” Dr. Krueger said. Indeed, based upon the encouraging safety and efficacy demonstrated this phase 2 study, a phase 3 program known as POETYK-PSO is underway (POETYK-PSO-1 and POETYK-PSO-2).
The phase 2 clinical trial results were published online in conjunction with the EADV congress.
The TYK2 inhibitor is being developed by Bristol-Myers Squibb. Dr. Krueger reported receiving personal fees as well as research grants paid directly to Rockefeller University from that pharmaceutical company and numerous others.
Source: Papp K et al. N Engl J Med. 2018 Sep 11. doi: 10.1056/NEJMoa1806382.
PARIS – A novel in a phase 2 clinical trial including 267 adults with moderate to severe disease, James G. Krueger, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“I would say the clinical response here is almost dead-on as a copy for ustekinumab, which is an [injectable interleukin] IL-23/IL-12 blocker. And we’re only at 12 weeks here; some of the curves look like they’re on a trajectory to go up further in terms of improvement. So I’m getting a performance with an oral drug that is just so much better than the approved alternatives that we have,” said Dr. Krueger, head of the laboratory of investigative dermatology and professor in clinical investigation at Rockefeller University in New York.
Oral apremilast (Otezla), for example, can’t touch those PASI 75 response rates in patients with moderate to severe psoriasis. Indeed, many psoriasis experts favor reserving apremilast for patients with moderate disease.
The 12-week, double-blind, placebo-controlled study was conducted at 82 sites in the United States and seven other countries. In this dose-ranging study, participants were randomized to the oral selective tyrosine kinase 2 (TYK2) inhibitor, known for the time being as BMS-986165, at 3 mg every other day, 3 mg daily, 3 mg twice a day, 6 mg twice a day, 12 mg daily, or to placebo.
The primary outcome was a 75% or greater reduction from baseline in Psoriasis Area and Severity Index score (PASI 75) at week 12. The TYK2 inhibitor outperformed placebo in dose-dependent fashion starting at the 3 mg/day dose. The PASI 75 rate was 7% with placebo, 9% with 3 mg of BMS-986165 every other day, 39% with 3 mg daily, 69% with 3 mg BID, 67% with 6 mg BID, and 75% with 12 mg/day. All secondary endpoints followed suit.
A striking finding in the phase 2 study was that when the drug was stopped for a month at the end of the 12-week treatment period, for the most part, the PASI 75 response and other clinical benefits were retained.
“I would contrast this to experiments that I have personally done with cyclosporine, where I have cleared people with cyclosporine, stopped it, and a month later every single patient has rip-roaring disease back. So I think this TYK2 inhibitor has some different performance features than just blocking a downstream T-cell transduction molecule,” observed the dermatologist, who is credited as the discoverer of the importance of the T cell in psoriasis pathogenesis.
The strong multidimensional evidence of clinical efficacy in the phase 2 study was supported mechanistically by analysis of skin biopsies obtained on study days 1, 15, and 85. The laboratory studies showed that the oral drug improved molecular, cellular, and clinical biomarkers associated with treatment efficacy. For example, at doses of 3 mg twice a day or higher, the TYK2 inhibitor reduced expression of IL-19 and IL-36A, which are key drivers of keratinocyte activation and epidermal hyperplasia. The drug also markedly decreased expression of genes in the Th17 pathway and essentially normalized expression of the proinflammatory genes beta defensin and S100A9.
In contrast to the Janus kinase (JAK) 1/3 and JAK 2 inhibitors in development for treatment of psoriasis, which paint with a much broader brush, the TYK2 inhibitor is highly selective for IL-23, IL-12, and interferon alpha.
“Previous studies have shown pan-JAK inhibition can be very effective in remitting psoriasis. The problem is that if one inhibits JAK1 and JAK3, one blocks the transduction of effector cytokines that are essentially there for protective immunity. That could lead to undesirable levels of immunosuppression,” Dr. Krueger explained.
The most important cytokine in the pathogenesis of psoriasis is clearly IL-23, he continued. In cell-based assays, the TYK2 inhibitor has been shown to be 100 times more selective in inhibiting IL-23 , IL-12, and interferon-alpha than JAK 1/3 inhibitors and 3,000 times more selective than JAK 2 inhibitors. This high degree of selectivity makes for fewer off-target effects and for a favorable safety profile.
“There were no major safety signals that would lead you to be concerned,” Dr. Krueger said. Indeed, based upon the encouraging safety and efficacy demonstrated this phase 2 study, a phase 3 program known as POETYK-PSO is underway (POETYK-PSO-1 and POETYK-PSO-2).
The phase 2 clinical trial results were published online in conjunction with the EADV congress.
The TYK2 inhibitor is being developed by Bristol-Myers Squibb. Dr. Krueger reported receiving personal fees as well as research grants paid directly to Rockefeller University from that pharmaceutical company and numerous others.
Source: Papp K et al. N Engl J Med. 2018 Sep 11. doi: 10.1056/NEJMoa1806382.
REPORTING FROM THE EADV CONGRESS
Key clinical point: A novel selective tyrosine kinase 2 inhibitor achieves response rates previously unheard of in oral therapy for moderate to severe psoriasis.
Major finding: At the top dose of oral BMS-986165 studied to date, the PASI 75 rate at 12 weeks was 75%.
Study details: This eight-country, randomized, double-blind, placebo-controlled phase 2 study included 267 patients with moderate to severe psoriasis.
Disclosures: The study was sponsored by Bristol-Myers Squibb. The presenter reported receiving personal fees and institutional research grants from that pharmaceutical company and numerous others.
Source: Papp K et al. N Engl J Med. 2018 Sep 11. doi: 10.1056/NEJMoa1806382.
Elevated type 2 diabetes risk seen in PsA patients
Patients with incident psoriatic arthritis are at a significantly increased risk of developing type 2 diabetes when compared against patients with psoriasis alone and with the general population, according to recent research published in Rheumatology.
Rachel Charlton, PhD, of the department of pharmacy and pharmacology at the University of Bath (England), and her colleagues performed an analysis of 6,783 incident cases of psoriatic arthritis (PsA) from the U.K. Clinical Practice Research Datalink who were diagnosed during 1998-2014. Patients were between 18 years and 89 years old with a median age of 49 years at PsA diagnosis.
In the study, the researchers randomly matched PsA cases at a 1:4 ratio to either a cohort of general population patients with no PsA, psoriasis, or inflammatory arthritis or a cohort of patients with psoriasis but no PsA or inflammatory arthritis. Patients were followed from match to the point where they either no longer met inclusion criteria for the cohort or received a diagnosis of type 2 diabetes, cerebrovascular disease (CVD), ischemic heart disease (IHD), or peripheral vascular disease (PVD) with a mean follow-up duration of approximately 5.5 years across all patient groups.
Patients in the PsA group had a significantly higher incidence of type 2 diabetes, compared with the general population (adjusted relative risk, 1.40; 95% confidence interval, 1.15-1.70; P = .0007) and psoriasis groups (adjusted RR, 1.53; 95% CI, 1.19-1.97; P = .0009). In the PsA group, risk of CVD (adjusted RR, 1.24; 95% CI, 0.99-1.56; P = .06), IHD (adjusted RR, 1.27; 95% CI, 1.05-1.54; P = .02), and PVD (adjusted RR, 1.40; 95% CI, 1.02-1.92; P = .04) were significantly higher than in the general population but not when compared with the psoriasis group. The overall risk of cardiovascular disease (including CVD, IHD, and PVD) for the PsA group was significantly higher (adjusted RR, 1.29; 95% CI, 1.12-1.48; P = .0005), compared with the general population.
“These results support the proposal in existing clinical guidelines that, in order to reduce cardiovascular risk in patients with PsA, it is important to treat inflammatory disease as well as to screen and treat traditional risk factors early in the disease course,” Ms. Charlton and her colleagues wrote in their study.
This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
SOURCE: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
Patients with incident psoriatic arthritis are at a significantly increased risk of developing type 2 diabetes when compared against patients with psoriasis alone and with the general population, according to recent research published in Rheumatology.
Rachel Charlton, PhD, of the department of pharmacy and pharmacology at the University of Bath (England), and her colleagues performed an analysis of 6,783 incident cases of psoriatic arthritis (PsA) from the U.K. Clinical Practice Research Datalink who were diagnosed during 1998-2014. Patients were between 18 years and 89 years old with a median age of 49 years at PsA diagnosis.
In the study, the researchers randomly matched PsA cases at a 1:4 ratio to either a cohort of general population patients with no PsA, psoriasis, or inflammatory arthritis or a cohort of patients with psoriasis but no PsA or inflammatory arthritis. Patients were followed from match to the point where they either no longer met inclusion criteria for the cohort or received a diagnosis of type 2 diabetes, cerebrovascular disease (CVD), ischemic heart disease (IHD), or peripheral vascular disease (PVD) with a mean follow-up duration of approximately 5.5 years across all patient groups.
Patients in the PsA group had a significantly higher incidence of type 2 diabetes, compared with the general population (adjusted relative risk, 1.40; 95% confidence interval, 1.15-1.70; P = .0007) and psoriasis groups (adjusted RR, 1.53; 95% CI, 1.19-1.97; P = .0009). In the PsA group, risk of CVD (adjusted RR, 1.24; 95% CI, 0.99-1.56; P = .06), IHD (adjusted RR, 1.27; 95% CI, 1.05-1.54; P = .02), and PVD (adjusted RR, 1.40; 95% CI, 1.02-1.92; P = .04) were significantly higher than in the general population but not when compared with the psoriasis group. The overall risk of cardiovascular disease (including CVD, IHD, and PVD) for the PsA group was significantly higher (adjusted RR, 1.29; 95% CI, 1.12-1.48; P = .0005), compared with the general population.
“These results support the proposal in existing clinical guidelines that, in order to reduce cardiovascular risk in patients with PsA, it is important to treat inflammatory disease as well as to screen and treat traditional risk factors early in the disease course,” Ms. Charlton and her colleagues wrote in their study.
This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
SOURCE: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
Patients with incident psoriatic arthritis are at a significantly increased risk of developing type 2 diabetes when compared against patients with psoriasis alone and with the general population, according to recent research published in Rheumatology.
Rachel Charlton, PhD, of the department of pharmacy and pharmacology at the University of Bath (England), and her colleagues performed an analysis of 6,783 incident cases of psoriatic arthritis (PsA) from the U.K. Clinical Practice Research Datalink who were diagnosed during 1998-2014. Patients were between 18 years and 89 years old with a median age of 49 years at PsA diagnosis.
In the study, the researchers randomly matched PsA cases at a 1:4 ratio to either a cohort of general population patients with no PsA, psoriasis, or inflammatory arthritis or a cohort of patients with psoriasis but no PsA or inflammatory arthritis. Patients were followed from match to the point where they either no longer met inclusion criteria for the cohort or received a diagnosis of type 2 diabetes, cerebrovascular disease (CVD), ischemic heart disease (IHD), or peripheral vascular disease (PVD) with a mean follow-up duration of approximately 5.5 years across all patient groups.
Patients in the PsA group had a significantly higher incidence of type 2 diabetes, compared with the general population (adjusted relative risk, 1.40; 95% confidence interval, 1.15-1.70; P = .0007) and psoriasis groups (adjusted RR, 1.53; 95% CI, 1.19-1.97; P = .0009). In the PsA group, risk of CVD (adjusted RR, 1.24; 95% CI, 0.99-1.56; P = .06), IHD (adjusted RR, 1.27; 95% CI, 1.05-1.54; P = .02), and PVD (adjusted RR, 1.40; 95% CI, 1.02-1.92; P = .04) were significantly higher than in the general population but not when compared with the psoriasis group. The overall risk of cardiovascular disease (including CVD, IHD, and PVD) for the PsA group was significantly higher (adjusted RR, 1.29; 95% CI, 1.12-1.48; P = .0005), compared with the general population.
“These results support the proposal in existing clinical guidelines that, in order to reduce cardiovascular risk in patients with PsA, it is important to treat inflammatory disease as well as to screen and treat traditional risk factors early in the disease course,” Ms. Charlton and her colleagues wrote in their study.
This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
SOURCE: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
FROM RHEUMATOLOGY
Key clinical point: It is important to treat inflammatory disease as well as to screen and treat traditional cardiovascular risk factors early in the course of PsA.
Major finding: (adjusted RR = 1.53) and a general population control group (adjusted RR = 1.40).
Study details: An analysis of 6,783 patients with psoriatic arthritis in the U.K. Clinical Practice Research Datalink who were diagnosed between 1998 and 2014.
Disclosures: This study was funded by a grant from the National Institute for Health Research in the United Kingdom. The authors reported no relevant conflicts of interest.
Source: Charlton RA et al. Rheumatology. 2018 Sep 6. doi: 10.1093/rheumatology/key286.
Psoriatic arthritis activity spikes briefly postpartum
Disease activity for women with psoriatic arthritis seeking pregnancy was relatively stable through 1 year after delivery, but there was a significant jump at 6 months postpartum follow-up, based on data from a prospective study of more than 100 patients.
Previous research has described rheumatoid arthritis remission during pregnancy, but the symptoms of psoriatic arthritis (PsA) before, during, and after pregnancy have not been well studied, wrote Kristin Ursin, MD, of Trondheim (Norway) University Hospital, and her colleagues.
In a study published in Arthritis Care & Research, the investigators reviewed data from 108 pregnancies in 103 women with PsA who were diagnosed between January 2006 and October 2017.
The participants were enrolled in a Norwegian nationwide registry that followed women with inflammatory diseases from preconception through 1 year after delivery. Disease activity was measured using the DAS28-CRP (28-Joint Disease Activity Score with C-reactive Protein). Participants were assessed at seven time points: before pregnancy, during each trimester, and at 6 weeks, 6 months, and 12 months after delivery.
Although approximately 75% of the women had stable disease activity throughout the study period, activity spiked at 6 months after delivery; DAS28-CRP scores at 6 months post partum were significantly higher than scores at 6 weeks post partum (2.71 vs. 2.45, respectively; P = .016).
The researchers conducted an additional analysis of the potential role of tumor necrosis factor inhibitor use and found that women taking a TNFi had significantly lower disease activity during pregnancy, compared with women not taking a TNFi; mean DAS28-CRP scores at 6 months post partum for these groups were 2.22 and 2.72, respectively (P = .043).
The study was limited by the use of DAS28-CRP as the main measure of disease activity; the index does not include potentially affected distal interphalangeal joints. In addition, not all the participants were assessed at each of the seven time points. However, the results suggest that most pregnant women with PsA experience low levels of disease activity, the researchers said. “Future research on pregnancy in women with PsA should include extended joint count (66/68 joints), and assessment of dactylitis, entheses, axial skeleton, and psoriasis,” they added.
The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
SOURCE: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
Disease activity for women with psoriatic arthritis seeking pregnancy was relatively stable through 1 year after delivery, but there was a significant jump at 6 months postpartum follow-up, based on data from a prospective study of more than 100 patients.
Previous research has described rheumatoid arthritis remission during pregnancy, but the symptoms of psoriatic arthritis (PsA) before, during, and after pregnancy have not been well studied, wrote Kristin Ursin, MD, of Trondheim (Norway) University Hospital, and her colleagues.
In a study published in Arthritis Care & Research, the investigators reviewed data from 108 pregnancies in 103 women with PsA who were diagnosed between January 2006 and October 2017.
The participants were enrolled in a Norwegian nationwide registry that followed women with inflammatory diseases from preconception through 1 year after delivery. Disease activity was measured using the DAS28-CRP (28-Joint Disease Activity Score with C-reactive Protein). Participants were assessed at seven time points: before pregnancy, during each trimester, and at 6 weeks, 6 months, and 12 months after delivery.
Although approximately 75% of the women had stable disease activity throughout the study period, activity spiked at 6 months after delivery; DAS28-CRP scores at 6 months post partum were significantly higher than scores at 6 weeks post partum (2.71 vs. 2.45, respectively; P = .016).
The researchers conducted an additional analysis of the potential role of tumor necrosis factor inhibitor use and found that women taking a TNFi had significantly lower disease activity during pregnancy, compared with women not taking a TNFi; mean DAS28-CRP scores at 6 months post partum for these groups were 2.22 and 2.72, respectively (P = .043).
The study was limited by the use of DAS28-CRP as the main measure of disease activity; the index does not include potentially affected distal interphalangeal joints. In addition, not all the participants were assessed at each of the seven time points. However, the results suggest that most pregnant women with PsA experience low levels of disease activity, the researchers said. “Future research on pregnancy in women with PsA should include extended joint count (66/68 joints), and assessment of dactylitis, entheses, axial skeleton, and psoriasis,” they added.
The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
SOURCE: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
Disease activity for women with psoriatic arthritis seeking pregnancy was relatively stable through 1 year after delivery, but there was a significant jump at 6 months postpartum follow-up, based on data from a prospective study of more than 100 patients.
Previous research has described rheumatoid arthritis remission during pregnancy, but the symptoms of psoriatic arthritis (PsA) before, during, and after pregnancy have not been well studied, wrote Kristin Ursin, MD, of Trondheim (Norway) University Hospital, and her colleagues.
In a study published in Arthritis Care & Research, the investigators reviewed data from 108 pregnancies in 103 women with PsA who were diagnosed between January 2006 and October 2017.
The participants were enrolled in a Norwegian nationwide registry that followed women with inflammatory diseases from preconception through 1 year after delivery. Disease activity was measured using the DAS28-CRP (28-Joint Disease Activity Score with C-reactive Protein). Participants were assessed at seven time points: before pregnancy, during each trimester, and at 6 weeks, 6 months, and 12 months after delivery.
Although approximately 75% of the women had stable disease activity throughout the study period, activity spiked at 6 months after delivery; DAS28-CRP scores at 6 months post partum were significantly higher than scores at 6 weeks post partum (2.71 vs. 2.45, respectively; P = .016).
The researchers conducted an additional analysis of the potential role of tumor necrosis factor inhibitor use and found that women taking a TNFi had significantly lower disease activity during pregnancy, compared with women not taking a TNFi; mean DAS28-CRP scores at 6 months post partum for these groups were 2.22 and 2.72, respectively (P = .043).
The study was limited by the use of DAS28-CRP as the main measure of disease activity; the index does not include potentially affected distal interphalangeal joints. In addition, not all the participants were assessed at each of the seven time points. However, the results suggest that most pregnant women with PsA experience low levels of disease activity, the researchers said. “Future research on pregnancy in women with PsA should include extended joint count (66/68 joints), and assessment of dactylitis, entheses, axial skeleton, and psoriasis,” they added.
The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
SOURCE: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Disease activity for pregnant women with PsA decreased during pregnancy, increased significantly by 6 months post partum, and returned to baseline by 12 months post partum.
Major finding: The average DAS28-CRP was 2.71 at 6 months vs. 2.45 at 6 weeks (P = .016).
Study details: The data come from 108 women with 103 pregnancies who were part of a national registry in Norway.
Disclosures: The researchers had no financial conflicts to disclose. The study was funded by the department of rheumatology at Trondheim University Hospital and the Research Fund of the Norwegian organization for people with rheumatic disease.
Source: Ursin K et al. Arthritis Care Res. 2018 Sep 7. doi: 10.1002/acr.23747.
Infliximab biosimilar only moderately less expensive in Medicare Part D
The infliximab-dyyb biosimilar was only moderately less expensive than the originator infliximab product Remicade in the United States in 2017 under Medicare Part D, an analysis shows.
Infliximab-dyyb (Inflectra) cost 18% less than infliximab, with an annual cost exceeding $14,000 in an analysis published online Sept. 4 in JAMA by Jinoos Yazdany, MD, of the division of rheumatology at the University of California, San Francisco, and her coauthors.
However, “without biosimilar gap discounts in 2017, beneficiaries would have paid more than $5,100 for infliximab-dyyb, or nearly $1,700 more in projected out-of-pocket costs than infliximab,” Dr. Yazdany and her coauthors wrote.
Biologics represent only 2% of U.S. prescriptions but made up 38% of drug spending in 2015 and accounted for 70% of growth in drug spending from 2010 to 2015, according to Dr. Yazdany and her colleagues.
Biologics for rheumatoid arthritis (RA) cost more than $14,000 per year, and in 2015, 3 were among the top 15 drugs in terms of Medicare expenditures, they added.
While biosimilars are supposed to increase competition and lower prices, it’s an open question whether they actually reduce out-of-pocket expenditures for the 43 million individuals with drug benefits under Medicare Part D.
That uncertainty is due in part to the complex cost-sharing design of Part D, which includes an initial deductible, a coverage phase, a coverage gap, and catastrophic coverage.
In 2017, the plan included an initial $400 deductible, followed by the coverage phase, in which the patient paid 25% of drug costs. In the coverage gap, which started at $3,700 in total drug costs, the patient’s share of drug costs increased to 40% for biologics, and 51% for biosimilars. In the catastrophic coverage phase, triggered when out-of-pocket costs exceeded $4,950, the patient was responsible for 5% of drug costs.
“Currently, beneficiaries receive a 50% manufacturer discount during the gap for brand-name drugs and biologics, but not for biosimilars,” Dr. Yazdany and her coauthors said in the report.
To evaluate cost-sharing for infliximab-dyyb, which in 2016 became the first available RA biosimilar, the authors analyzed data for all Part D plans in the June 2017 Medicare Prescription Drug Plan Formulary, Pharmacy Network, and Pricing Information Files.
Out of 2,547 plans, only 10% covered the biosimilar, while 96% covered infliximab, the authors found.
The mean total cost of infliximab-dyyb was “modestly lower,” they reported. Eight-week prescription costs were $2,185 for infliximab-dyyb versus $2,667 for infliximab, while annual costs were $14,202 for the biosimilar and $17,335 for infliximab.
However, all plans required coinsurance cost-sharing for the biosimilar, they said. The mean coinsurance rate was 26.6% of the total drug cost for the biosimilar and 28.4% for infliximab.
For beneficiaries, projected annual out-of-pocket costs without the gap discount were $5,118 for infliximab-dyyb and $3,432 for infliximab, the researchers said.
Biosimilar gap discounts are set to start in 2019, according to the authors. However, they said those discounts may not substantially reduce out-of-pocket costs for Part D beneficiaries because of the high price of infliximab-dyyb and a coinsurance cost-sharing rate similar to that of infliximab.
“Further policies are needed to address affordability and access to specialty drugs,” Dr. Yazdany and her coauthors concluded.
The study was funded in part by grants from the Agency for Healthcare Research and Quality, the Robert L. Kroc Endowed Chair in Rheumatic and Connective Tissue Diseases, and other sources. Dr. Yazdany reported receiving an independent investigator award from Pfizer. Her coauthors reported no conflict of interest disclosures.
The infliximab-dyyb biosimilar was only moderately less expensive than the originator infliximab product Remicade in the United States in 2017 under Medicare Part D, an analysis shows.
Infliximab-dyyb (Inflectra) cost 18% less than infliximab, with an annual cost exceeding $14,000 in an analysis published online Sept. 4 in JAMA by Jinoos Yazdany, MD, of the division of rheumatology at the University of California, San Francisco, and her coauthors.
However, “without biosimilar gap discounts in 2017, beneficiaries would have paid more than $5,100 for infliximab-dyyb, or nearly $1,700 more in projected out-of-pocket costs than infliximab,” Dr. Yazdany and her coauthors wrote.
Biologics represent only 2% of U.S. prescriptions but made up 38% of drug spending in 2015 and accounted for 70% of growth in drug spending from 2010 to 2015, according to Dr. Yazdany and her colleagues.
Biologics for rheumatoid arthritis (RA) cost more than $14,000 per year, and in 2015, 3 were among the top 15 drugs in terms of Medicare expenditures, they added.
While biosimilars are supposed to increase competition and lower prices, it’s an open question whether they actually reduce out-of-pocket expenditures for the 43 million individuals with drug benefits under Medicare Part D.
That uncertainty is due in part to the complex cost-sharing design of Part D, which includes an initial deductible, a coverage phase, a coverage gap, and catastrophic coverage.
In 2017, the plan included an initial $400 deductible, followed by the coverage phase, in which the patient paid 25% of drug costs. In the coverage gap, which started at $3,700 in total drug costs, the patient’s share of drug costs increased to 40% for biologics, and 51% for biosimilars. In the catastrophic coverage phase, triggered when out-of-pocket costs exceeded $4,950, the patient was responsible for 5% of drug costs.
“Currently, beneficiaries receive a 50% manufacturer discount during the gap for brand-name drugs and biologics, but not for biosimilars,” Dr. Yazdany and her coauthors said in the report.
To evaluate cost-sharing for infliximab-dyyb, which in 2016 became the first available RA biosimilar, the authors analyzed data for all Part D plans in the June 2017 Medicare Prescription Drug Plan Formulary, Pharmacy Network, and Pricing Information Files.
Out of 2,547 plans, only 10% covered the biosimilar, while 96% covered infliximab, the authors found.
The mean total cost of infliximab-dyyb was “modestly lower,” they reported. Eight-week prescription costs were $2,185 for infliximab-dyyb versus $2,667 for infliximab, while annual costs were $14,202 for the biosimilar and $17,335 for infliximab.
However, all plans required coinsurance cost-sharing for the biosimilar, they said. The mean coinsurance rate was 26.6% of the total drug cost for the biosimilar and 28.4% for infliximab.
For beneficiaries, projected annual out-of-pocket costs without the gap discount were $5,118 for infliximab-dyyb and $3,432 for infliximab, the researchers said.
Biosimilar gap discounts are set to start in 2019, according to the authors. However, they said those discounts may not substantially reduce out-of-pocket costs for Part D beneficiaries because of the high price of infliximab-dyyb and a coinsurance cost-sharing rate similar to that of infliximab.
“Further policies are needed to address affordability and access to specialty drugs,” Dr. Yazdany and her coauthors concluded.
The study was funded in part by grants from the Agency for Healthcare Research and Quality, the Robert L. Kroc Endowed Chair in Rheumatic and Connective Tissue Diseases, and other sources. Dr. Yazdany reported receiving an independent investigator award from Pfizer. Her coauthors reported no conflict of interest disclosures.
The infliximab-dyyb biosimilar was only moderately less expensive than the originator infliximab product Remicade in the United States in 2017 under Medicare Part D, an analysis shows.
Infliximab-dyyb (Inflectra) cost 18% less than infliximab, with an annual cost exceeding $14,000 in an analysis published online Sept. 4 in JAMA by Jinoos Yazdany, MD, of the division of rheumatology at the University of California, San Francisco, and her coauthors.
However, “without biosimilar gap discounts in 2017, beneficiaries would have paid more than $5,100 for infliximab-dyyb, or nearly $1,700 more in projected out-of-pocket costs than infliximab,” Dr. Yazdany and her coauthors wrote.
Biologics represent only 2% of U.S. prescriptions but made up 38% of drug spending in 2015 and accounted for 70% of growth in drug spending from 2010 to 2015, according to Dr. Yazdany and her colleagues.
Biologics for rheumatoid arthritis (RA) cost more than $14,000 per year, and in 2015, 3 were among the top 15 drugs in terms of Medicare expenditures, they added.
While biosimilars are supposed to increase competition and lower prices, it’s an open question whether they actually reduce out-of-pocket expenditures for the 43 million individuals with drug benefits under Medicare Part D.
That uncertainty is due in part to the complex cost-sharing design of Part D, which includes an initial deductible, a coverage phase, a coverage gap, and catastrophic coverage.
In 2017, the plan included an initial $400 deductible, followed by the coverage phase, in which the patient paid 25% of drug costs. In the coverage gap, which started at $3,700 in total drug costs, the patient’s share of drug costs increased to 40% for biologics, and 51% for biosimilars. In the catastrophic coverage phase, triggered when out-of-pocket costs exceeded $4,950, the patient was responsible for 5% of drug costs.
“Currently, beneficiaries receive a 50% manufacturer discount during the gap for brand-name drugs and biologics, but not for biosimilars,” Dr. Yazdany and her coauthors said in the report.
To evaluate cost-sharing for infliximab-dyyb, which in 2016 became the first available RA biosimilar, the authors analyzed data for all Part D plans in the June 2017 Medicare Prescription Drug Plan Formulary, Pharmacy Network, and Pricing Information Files.
Out of 2,547 plans, only 10% covered the biosimilar, while 96% covered infliximab, the authors found.
The mean total cost of infliximab-dyyb was “modestly lower,” they reported. Eight-week prescription costs were $2,185 for infliximab-dyyb versus $2,667 for infliximab, while annual costs were $14,202 for the biosimilar and $17,335 for infliximab.
However, all plans required coinsurance cost-sharing for the biosimilar, they said. The mean coinsurance rate was 26.6% of the total drug cost for the biosimilar and 28.4% for infliximab.
For beneficiaries, projected annual out-of-pocket costs without the gap discount were $5,118 for infliximab-dyyb and $3,432 for infliximab, the researchers said.
Biosimilar gap discounts are set to start in 2019, according to the authors. However, they said those discounts may not substantially reduce out-of-pocket costs for Part D beneficiaries because of the high price of infliximab-dyyb and a coinsurance cost-sharing rate similar to that of infliximab.
“Further policies are needed to address affordability and access to specialty drugs,” Dr. Yazdany and her coauthors concluded.
The study was funded in part by grants from the Agency for Healthcare Research and Quality, the Robert L. Kroc Endowed Chair in Rheumatic and Connective Tissue Diseases, and other sources. Dr. Yazdany reported receiving an independent investigator award from Pfizer. Her coauthors reported no conflict of interest disclosures.
FROM JAMA
Key clinical point:
Major finding: Infliximab-dyyb was 18% less costly than infliximab, with an annual cost exceeding $14,000.
Study details: Analysis of data for 2,547 Part D plans in the June 2017 Medicare Prescription Drug Plan Formulary, Pharmacy Network, and Pricing Information Files.
Disclosures: The study was funded in part by grants from the Agency for Healthcare Research and Quality, the Robert L. Kroc Endowed Chair in Rheumatic and Connective Tissue Diseases, and other sources. One author reported receiving an independent investigator award from Pfizer.
Source: Yazdany J et al. JAMA. 2018;320(9):931-3.
Sustained minimal disease activity in PsA reduced CV risk factors
Achieving sustained minimal disease activity in psoriatic arthritis could slow or prevent the progression of carotid atherosclerosis and arterial stiffness, new research suggests.
A prospective cohort study, published online Aug. 25 in Arthritis & Rheumatology, involved 90 patients with psoriatic arthritis (PsA) who were followed for at least 24 months. High-resolution carotid ultrasound and arterial stiffness markers were assessed annually.
Overall, 57 patients (63%) achieved minimal disease activity (MDA) – defined as meeting five or more cutoffs for seven domains of disease activity – at 1 year of follow-up, 69% achieved it by 2 years, and 46% sustained MDA from the 1-year follow-up to the 2-year follow-up.
The 41 patients who sustained MDA over the follow-up points showed significantly lower odds of progression of atherosclerotic carotid plaques (odds ratio = 0.273; P = .024). They were also significantly less likely to show increases in total carotid plaque area, increased intima-media thickness, or increased measures of arterial stiffness. The researchers also noted a trend toward improvements in pulse wave velocity in this group.
Patients who achieved sustained MDA had lower disease activity at baseline, and a higher proportion of them were already in a state of MDA at baseline, compared with the group of patients who did not achieved sustained MDA.
Sustained MDA appeared to be key to reducing atherosclerosis, as researchers did not see any significant differences in measures of arterial stiffness between those who achieved MDA – sustained or otherwise – at 2 years and those who did not, after adjusting for baseline differences and use of disease-modifying antirheumatic drugs (DMARDs).
“The most important finding in this study is that long-term control of inflammation is important in preventing progression of subclinical atherosclerosis and arterial stiffness, independent of traditional CV risk factors,” wrote Isaac T. Cheng of Prince of Wales Hospital at The Chinese University of Hong Kong, and his coauthors. “These data suggest that low stable disease activity over a prolonged period of time may have a significant protective effect against CVD in PsA compared with those patients with intermittent flare-ups.”
The study also looked at treatment effect. More patients treated with biologic DMARDs achieved MDA, but the researchers saw no association between the use of conventional synthetic DMARDs and the likelihood of achieving MDA.
They noted that the protective effects of achieving sustained MDA seemed to be independent of biologic DMARDs, “suggesting that controlling disease activity using various combinations of conventional and biological DMARD may be useful in improving CV risk in these patients.”
However, the investigators noted that the study was conducted in Hong Kong, where biologics are not reimbursed by the government. Patients in the study had to pay for these medications themselves, and some could not afford them.
They raised the possibility that early initiation with biologic DMARDs might further improve cardiovascular outcomes, but said this needed to be addressed in future studies.
“These data support the EULAR recommendation that disease activity should be controlled optimally in order to lower CVD risk in patients with PsA,” the authors wrote. “Data from the current study confirm that using a treat-to-target strategy, MDA is indeed an achievable target even in a health care system with resource constrain.”
The Health and Medical Research Fund supported the study. No conflicts of interest were declared.
SOURCE: Cheng I et al. Arthritis Rheumatol. 2018 Aug 25. doi: 10.1002/art.40695.
Achieving sustained minimal disease activity in psoriatic arthritis could slow or prevent the progression of carotid atherosclerosis and arterial stiffness, new research suggests.
A prospective cohort study, published online Aug. 25 in Arthritis & Rheumatology, involved 90 patients with psoriatic arthritis (PsA) who were followed for at least 24 months. High-resolution carotid ultrasound and arterial stiffness markers were assessed annually.
Overall, 57 patients (63%) achieved minimal disease activity (MDA) – defined as meeting five or more cutoffs for seven domains of disease activity – at 1 year of follow-up, 69% achieved it by 2 years, and 46% sustained MDA from the 1-year follow-up to the 2-year follow-up.
The 41 patients who sustained MDA over the follow-up points showed significantly lower odds of progression of atherosclerotic carotid plaques (odds ratio = 0.273; P = .024). They were also significantly less likely to show increases in total carotid plaque area, increased intima-media thickness, or increased measures of arterial stiffness. The researchers also noted a trend toward improvements in pulse wave velocity in this group.
Patients who achieved sustained MDA had lower disease activity at baseline, and a higher proportion of them were already in a state of MDA at baseline, compared with the group of patients who did not achieved sustained MDA.
Sustained MDA appeared to be key to reducing atherosclerosis, as researchers did not see any significant differences in measures of arterial stiffness between those who achieved MDA – sustained or otherwise – at 2 years and those who did not, after adjusting for baseline differences and use of disease-modifying antirheumatic drugs (DMARDs).
“The most important finding in this study is that long-term control of inflammation is important in preventing progression of subclinical atherosclerosis and arterial stiffness, independent of traditional CV risk factors,” wrote Isaac T. Cheng of Prince of Wales Hospital at The Chinese University of Hong Kong, and his coauthors. “These data suggest that low stable disease activity over a prolonged period of time may have a significant protective effect against CVD in PsA compared with those patients with intermittent flare-ups.”
The study also looked at treatment effect. More patients treated with biologic DMARDs achieved MDA, but the researchers saw no association between the use of conventional synthetic DMARDs and the likelihood of achieving MDA.
They noted that the protective effects of achieving sustained MDA seemed to be independent of biologic DMARDs, “suggesting that controlling disease activity using various combinations of conventional and biological DMARD may be useful in improving CV risk in these patients.”
However, the investigators noted that the study was conducted in Hong Kong, where biologics are not reimbursed by the government. Patients in the study had to pay for these medications themselves, and some could not afford them.
They raised the possibility that early initiation with biologic DMARDs might further improve cardiovascular outcomes, but said this needed to be addressed in future studies.
“These data support the EULAR recommendation that disease activity should be controlled optimally in order to lower CVD risk in patients with PsA,” the authors wrote. “Data from the current study confirm that using a treat-to-target strategy, MDA is indeed an achievable target even in a health care system with resource constrain.”
The Health and Medical Research Fund supported the study. No conflicts of interest were declared.
SOURCE: Cheng I et al. Arthritis Rheumatol. 2018 Aug 25. doi: 10.1002/art.40695.
Achieving sustained minimal disease activity in psoriatic arthritis could slow or prevent the progression of carotid atherosclerosis and arterial stiffness, new research suggests.
A prospective cohort study, published online Aug. 25 in Arthritis & Rheumatology, involved 90 patients with psoriatic arthritis (PsA) who were followed for at least 24 months. High-resolution carotid ultrasound and arterial stiffness markers were assessed annually.
Overall, 57 patients (63%) achieved minimal disease activity (MDA) – defined as meeting five or more cutoffs for seven domains of disease activity – at 1 year of follow-up, 69% achieved it by 2 years, and 46% sustained MDA from the 1-year follow-up to the 2-year follow-up.
The 41 patients who sustained MDA over the follow-up points showed significantly lower odds of progression of atherosclerotic carotid plaques (odds ratio = 0.273; P = .024). They were also significantly less likely to show increases in total carotid plaque area, increased intima-media thickness, or increased measures of arterial stiffness. The researchers also noted a trend toward improvements in pulse wave velocity in this group.
Patients who achieved sustained MDA had lower disease activity at baseline, and a higher proportion of them were already in a state of MDA at baseline, compared with the group of patients who did not achieved sustained MDA.
Sustained MDA appeared to be key to reducing atherosclerosis, as researchers did not see any significant differences in measures of arterial stiffness between those who achieved MDA – sustained or otherwise – at 2 years and those who did not, after adjusting for baseline differences and use of disease-modifying antirheumatic drugs (DMARDs).
“The most important finding in this study is that long-term control of inflammation is important in preventing progression of subclinical atherosclerosis and arterial stiffness, independent of traditional CV risk factors,” wrote Isaac T. Cheng of Prince of Wales Hospital at The Chinese University of Hong Kong, and his coauthors. “These data suggest that low stable disease activity over a prolonged period of time may have a significant protective effect against CVD in PsA compared with those patients with intermittent flare-ups.”
The study also looked at treatment effect. More patients treated with biologic DMARDs achieved MDA, but the researchers saw no association between the use of conventional synthetic DMARDs and the likelihood of achieving MDA.
They noted that the protective effects of achieving sustained MDA seemed to be independent of biologic DMARDs, “suggesting that controlling disease activity using various combinations of conventional and biological DMARD may be useful in improving CV risk in these patients.”
However, the investigators noted that the study was conducted in Hong Kong, where biologics are not reimbursed by the government. Patients in the study had to pay for these medications themselves, and some could not afford them.
They raised the possibility that early initiation with biologic DMARDs might further improve cardiovascular outcomes, but said this needed to be addressed in future studies.
“These data support the EULAR recommendation that disease activity should be controlled optimally in order to lower CVD risk in patients with PsA,” the authors wrote. “Data from the current study confirm that using a treat-to-target strategy, MDA is indeed an achievable target even in a health care system with resource constrain.”
The Health and Medical Research Fund supported the study. No conflicts of interest were declared.
SOURCE: Cheng I et al. Arthritis Rheumatol. 2018 Aug 25. doi: 10.1002/art.40695.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point:
Major finding: Patients who achieved sustained minimal disease activity had lower odds of progression of atherosclerotic carotid plaques.
Study details: Prospective cohort study of 90 patients with psoriatic arthritis.
Disclosures: The Health and Medical Research Fund supported the study. No conflicts of interest were declared.
Source: Cheng I et al. Arthritis Rheumatol. 2018 Aug 25. doi: 10.1002/art.40695.
Medication app boosts psoriasis patients’ short-term adherence
compared with those who did not use the app.
Treatment adherence remains a challenge in psoriasis, and although the field of electronic health interventions is growing, data on the effectiveness of such interventions are limited, wrote Mathias T. Svendsen, MD, of Odense University Hospital in Denmark, and his colleagues.
In a study published in the British Journal of Dermatology, 134 adults with psoriasis were randomized to use a smartphone app (68) or not (66) that provided daily medication reminders and daily information about the amount of treatment and number of product applications.
The primary outcome measure of treatment adherence was defined as once-daily application of topical medication – calcipotriol/betamethasone dipropionate cutaneous foam – for at least 80% of the days during the treatment period. A computer chip on the medication dispenser tracked patient use of the product and sent usage information to the patient’s smartphone via Bluetooth.
At 4 weeks, 65% of patients who used the app were adherent to treatment, versus 38% of those who didn’t use the app (P = .004).
In addition, patients who used the app showed significant improvement in disease severity, based on the secondary outcome measure of the Lattice System Physician’s Global Assessment (LS-PGA) at 4 weeks, with a mean change in score from baseline of 1.86 in the app group and 1.46 in the non-app group (P = .047). The LS-PGA and the Dermatology Life Quality Index (DLQI) were measured at all visits. No significant differences on the DLQI appeared between the groups.
During a 22-week follow-up completed by 122 patients, the effects were similar, but the differences were not statistically significant at weeks 8 and 26.
The study findings were limited by several factors, including the lack of knowledge of the correct amount of medication needed for the full benefit of the topical treatment and a lack of data on patient satisfaction with the app, the researchers noted. However, the results suggest that a medication reminder app improved disease severity as well as patient adherence rates in the short term, and that “there is potential for implementing patient-supporting apps in the dermatology clinic.”
The study was supported by LEO Pharma and by the Kirsten and Volmer Rask Nielsen’s Foundation; part of Dr. Svendsen’s salary during the study was paid by LEO Pharma, and several coauthors reported relationships with LEO Pharma.
SOURCE: Svendsen MT et al. Br J Dermatol. 2018 Apr 14. doi: 10.1111/bjd.16667.
compared with those who did not use the app.
Treatment adherence remains a challenge in psoriasis, and although the field of electronic health interventions is growing, data on the effectiveness of such interventions are limited, wrote Mathias T. Svendsen, MD, of Odense University Hospital in Denmark, and his colleagues.
In a study published in the British Journal of Dermatology, 134 adults with psoriasis were randomized to use a smartphone app (68) or not (66) that provided daily medication reminders and daily information about the amount of treatment and number of product applications.
The primary outcome measure of treatment adherence was defined as once-daily application of topical medication – calcipotriol/betamethasone dipropionate cutaneous foam – for at least 80% of the days during the treatment period. A computer chip on the medication dispenser tracked patient use of the product and sent usage information to the patient’s smartphone via Bluetooth.
At 4 weeks, 65% of patients who used the app were adherent to treatment, versus 38% of those who didn’t use the app (P = .004).
In addition, patients who used the app showed significant improvement in disease severity, based on the secondary outcome measure of the Lattice System Physician’s Global Assessment (LS-PGA) at 4 weeks, with a mean change in score from baseline of 1.86 in the app group and 1.46 in the non-app group (P = .047). The LS-PGA and the Dermatology Life Quality Index (DLQI) were measured at all visits. No significant differences on the DLQI appeared between the groups.
During a 22-week follow-up completed by 122 patients, the effects were similar, but the differences were not statistically significant at weeks 8 and 26.
The study findings were limited by several factors, including the lack of knowledge of the correct amount of medication needed for the full benefit of the topical treatment and a lack of data on patient satisfaction with the app, the researchers noted. However, the results suggest that a medication reminder app improved disease severity as well as patient adherence rates in the short term, and that “there is potential for implementing patient-supporting apps in the dermatology clinic.”
The study was supported by LEO Pharma and by the Kirsten and Volmer Rask Nielsen’s Foundation; part of Dr. Svendsen’s salary during the study was paid by LEO Pharma, and several coauthors reported relationships with LEO Pharma.
SOURCE: Svendsen MT et al. Br J Dermatol. 2018 Apr 14. doi: 10.1111/bjd.16667.
compared with those who did not use the app.
Treatment adherence remains a challenge in psoriasis, and although the field of electronic health interventions is growing, data on the effectiveness of such interventions are limited, wrote Mathias T. Svendsen, MD, of Odense University Hospital in Denmark, and his colleagues.
In a study published in the British Journal of Dermatology, 134 adults with psoriasis were randomized to use a smartphone app (68) or not (66) that provided daily medication reminders and daily information about the amount of treatment and number of product applications.
The primary outcome measure of treatment adherence was defined as once-daily application of topical medication – calcipotriol/betamethasone dipropionate cutaneous foam – for at least 80% of the days during the treatment period. A computer chip on the medication dispenser tracked patient use of the product and sent usage information to the patient’s smartphone via Bluetooth.
At 4 weeks, 65% of patients who used the app were adherent to treatment, versus 38% of those who didn’t use the app (P = .004).
In addition, patients who used the app showed significant improvement in disease severity, based on the secondary outcome measure of the Lattice System Physician’s Global Assessment (LS-PGA) at 4 weeks, with a mean change in score from baseline of 1.86 in the app group and 1.46 in the non-app group (P = .047). The LS-PGA and the Dermatology Life Quality Index (DLQI) were measured at all visits. No significant differences on the DLQI appeared between the groups.
During a 22-week follow-up completed by 122 patients, the effects were similar, but the differences were not statistically significant at weeks 8 and 26.
The study findings were limited by several factors, including the lack of knowledge of the correct amount of medication needed for the full benefit of the topical treatment and a lack of data on patient satisfaction with the app, the researchers noted. However, the results suggest that a medication reminder app improved disease severity as well as patient adherence rates in the short term, and that “there is potential for implementing patient-supporting apps in the dermatology clinic.”
The study was supported by LEO Pharma and by the Kirsten and Volmer Rask Nielsen’s Foundation; part of Dr. Svendsen’s salary during the study was paid by LEO Pharma, and several coauthors reported relationships with LEO Pharma.
SOURCE: Svendsen MT et al. Br J Dermatol. 2018 Apr 14. doi: 10.1111/bjd.16667.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: Using a smartphone app helped patients with psoriasis significantly improve their treatment adherence.
Major finding: Significantly more patients who used the app followed their topical treatment plan, compared with the no-app controls (65% vs. 38%).
Study details: The data come from 134 adults with psoriasis who were randomized to use an app or no app for 28 days.
Disclosures: The study was supported by LEO Pharma and by the Kirsten and Volmer Rask Nielsen’s Foundation; part of Dr. Svendsen’s salary during the study was paid by LEO Pharma, and several coauthors reported relationships with LEO Pharma.
Source: Svendsen MT et al. Br J Dermatol. 2018 Apr 14. doi: 10.1111/bjd.16667.
Risankizumab proves more effective in psoriasis than ustekinumab
, according to results of a pair of head-to-head trials published in the Lancet.
The replicate phase 3, randomized, double-blind, placebo- and active comparator–controlled trials, UltIMMa-1 (NCT02684370) and UltIMMa-2 (NCT02684375) altogether randomized 997 patients to risankizumab, ustekinumab, or placebo. The coprimary endpoints were the proportions of patients achieving 90% reduction in Psoriasis Area and Severity Index (PASI 90) at 16 weeks and a static Physician Global Assessment (sPGA) score of 0 or 1, and the 15 ranked secondary endpoints included proportions of those achieving PASI 100 or sPGA 0, both of which demonstrate total clearance of psoriasis, as well as measures of quality of life improvement.
Compared with those receiving either ustekinumab or placebo, a significantly higher proportion of patients receiving risankizumab achieved the coprimary endpoints, and all secondary endpoints were met. In UltIMMA-1, 75.3% of risankizumab patients achieved PASI 90, compared with 4.9% of placebo patients and 42% of ustekinumab patients (P less than .0001 when comparing it with both placebo and ustekinumab); sPGA of 0 or 1 was achieved by 87.8% of risankizumab patients and only 7.8% of placebo patients and 63% of ustekinumab patients (P less than .0001 when comparing it with both placebo and ustekinumab). Results were similar in UltIMMA-2: 74.8% of risankizumab patients achieved PASI 90, and 83.7% of them achieved sPGA 0 or 1 (P less than .0001 when comparing them with placebo and ustekinumab). According to results of the secondary endpoints, both studies also showed greater rates of clearance and improvements in quality of life among patients receiving risankizumab than among those receiving either placebo or ustekinumab.
The safety profiles across treatment groups were similar in both studies, with the most common adverse events including upper respiratory tract infection, headache, and diarrhea.
Risankizumab is a humanized IgG1 monoclonal antibody that targets the p19 subunit of only interleukin-23, unlike the studies’ active comparator, ustekinumab, which targets both interleukin-23 and interleukin-12. “Selectively blocking interleukin 23 with a p19 inhibitor appears to be one of the best ways to treat psoriasis,” commented Abigail Cline, MD, and Steven R. Feldman, MD, PhD, both of Wake Forest University, Winston-Salem, N.C., in an accompanying editorial (Lancet. 2018 Aug 7;392:616-71.).
The authors of the study reported relationships with various industry entities, including AbbVie, which sponsored the studies and developed risankizumab, and Boehringer Ingelheim, which collaborated in the studies. The authors of the editorial also disclosed relationships with entities, including AbbVie.
SOURCE: Gordon KB et al. Lancet. 2018 Aug 7;392:650-61.
, according to results of a pair of head-to-head trials published in the Lancet.
The replicate phase 3, randomized, double-blind, placebo- and active comparator–controlled trials, UltIMMa-1 (NCT02684370) and UltIMMa-2 (NCT02684375) altogether randomized 997 patients to risankizumab, ustekinumab, or placebo. The coprimary endpoints were the proportions of patients achieving 90% reduction in Psoriasis Area and Severity Index (PASI 90) at 16 weeks and a static Physician Global Assessment (sPGA) score of 0 or 1, and the 15 ranked secondary endpoints included proportions of those achieving PASI 100 or sPGA 0, both of which demonstrate total clearance of psoriasis, as well as measures of quality of life improvement.
Compared with those receiving either ustekinumab or placebo, a significantly higher proportion of patients receiving risankizumab achieved the coprimary endpoints, and all secondary endpoints were met. In UltIMMA-1, 75.3% of risankizumab patients achieved PASI 90, compared with 4.9% of placebo patients and 42% of ustekinumab patients (P less than .0001 when comparing it with both placebo and ustekinumab); sPGA of 0 or 1 was achieved by 87.8% of risankizumab patients and only 7.8% of placebo patients and 63% of ustekinumab patients (P less than .0001 when comparing it with both placebo and ustekinumab). Results were similar in UltIMMA-2: 74.8% of risankizumab patients achieved PASI 90, and 83.7% of them achieved sPGA 0 or 1 (P less than .0001 when comparing them with placebo and ustekinumab). According to results of the secondary endpoints, both studies also showed greater rates of clearance and improvements in quality of life among patients receiving risankizumab than among those receiving either placebo or ustekinumab.
The safety profiles across treatment groups were similar in both studies, with the most common adverse events including upper respiratory tract infection, headache, and diarrhea.
Risankizumab is a humanized IgG1 monoclonal antibody that targets the p19 subunit of only interleukin-23, unlike the studies’ active comparator, ustekinumab, which targets both interleukin-23 and interleukin-12. “Selectively blocking interleukin 23 with a p19 inhibitor appears to be one of the best ways to treat psoriasis,” commented Abigail Cline, MD, and Steven R. Feldman, MD, PhD, both of Wake Forest University, Winston-Salem, N.C., in an accompanying editorial (Lancet. 2018 Aug 7;392:616-71.).
The authors of the study reported relationships with various industry entities, including AbbVie, which sponsored the studies and developed risankizumab, and Boehringer Ingelheim, which collaborated in the studies. The authors of the editorial also disclosed relationships with entities, including AbbVie.
SOURCE: Gordon KB et al. Lancet. 2018 Aug 7;392:650-61.
, according to results of a pair of head-to-head trials published in the Lancet.
The replicate phase 3, randomized, double-blind, placebo- and active comparator–controlled trials, UltIMMa-1 (NCT02684370) and UltIMMa-2 (NCT02684375) altogether randomized 997 patients to risankizumab, ustekinumab, or placebo. The coprimary endpoints were the proportions of patients achieving 90% reduction in Psoriasis Area and Severity Index (PASI 90) at 16 weeks and a static Physician Global Assessment (sPGA) score of 0 or 1, and the 15 ranked secondary endpoints included proportions of those achieving PASI 100 or sPGA 0, both of which demonstrate total clearance of psoriasis, as well as measures of quality of life improvement.
Compared with those receiving either ustekinumab or placebo, a significantly higher proportion of patients receiving risankizumab achieved the coprimary endpoints, and all secondary endpoints were met. In UltIMMA-1, 75.3% of risankizumab patients achieved PASI 90, compared with 4.9% of placebo patients and 42% of ustekinumab patients (P less than .0001 when comparing it with both placebo and ustekinumab); sPGA of 0 or 1 was achieved by 87.8% of risankizumab patients and only 7.8% of placebo patients and 63% of ustekinumab patients (P less than .0001 when comparing it with both placebo and ustekinumab). Results were similar in UltIMMA-2: 74.8% of risankizumab patients achieved PASI 90, and 83.7% of them achieved sPGA 0 or 1 (P less than .0001 when comparing them with placebo and ustekinumab). According to results of the secondary endpoints, both studies also showed greater rates of clearance and improvements in quality of life among patients receiving risankizumab than among those receiving either placebo or ustekinumab.
The safety profiles across treatment groups were similar in both studies, with the most common adverse events including upper respiratory tract infection, headache, and diarrhea.
Risankizumab is a humanized IgG1 monoclonal antibody that targets the p19 subunit of only interleukin-23, unlike the studies’ active comparator, ustekinumab, which targets both interleukin-23 and interleukin-12. “Selectively blocking interleukin 23 with a p19 inhibitor appears to be one of the best ways to treat psoriasis,” commented Abigail Cline, MD, and Steven R. Feldman, MD, PhD, both of Wake Forest University, Winston-Salem, N.C., in an accompanying editorial (Lancet. 2018 Aug 7;392:616-71.).
The authors of the study reported relationships with various industry entities, including AbbVie, which sponsored the studies and developed risankizumab, and Boehringer Ingelheim, which collaborated in the studies. The authors of the editorial also disclosed relationships with entities, including AbbVie.
SOURCE: Gordon KB et al. Lancet. 2018 Aug 7;392:650-61.
FROM THE LANCET