User login
Short, long-lasting bronchodilators similar for exacerbated COPD
HONOLULU – in safety and efficacy to a short-acting combination of albuterol and ipratropium.
The 2023 Gold Report on prevention, management, and diagnosis of COPD recommended switching to long-acting bronchodilators despite a lack of clinical evidence showing safety in patients hospitalized for COPD exacerbation, according to Rajiv Dhand, MD, who presented the new study at the annual meeting of the American College of Chest Physicians (CHEST).
“We wanted to establish the safety, because long-acting agents are approved only for use in nonhospitalized patients. We established that it was safe and that it was comparably effective, but you could give 30% lower doses. Patients don’t have to be woken up to get the medication, and there’s a better chance that all the doses will be administered to these patients. So I think that it provides convenience with similar efficacy and safety,” said Dr. Dhand, a pulmonologist and professor of medicine at the University of Tennessee, Knoxville.
The researchers randomized 60 patients to receive nebulized albuterol (2.5 mg) and ipratropium (0.5 mg) every 6 hours (short-acting group) or nebulized formoterol (20 mcg) every 12 hours and revefenacin (175 mcg) every 24 hours (long-acting group). The mean age was 63.2 years, 58.3% were male, and 65% were current smokers.
The median decrease between day 1 and day 3 in the Modified Borg Dyspnea score was 4.0 in the long-acting group (P < .001), and 2.0 in the short-acting group, though the latter was not statistically significant (P = .134). Both groups had a decrease in supplemental oxygen requirement, with no difference between the two groups. There was also no difference in the number of respiratory visits for rescue therapy.
Respiratory therapists in the audience welcomed the new evidence. “As a respiratory therapist, I feel that we should move away from giving good short acting [therapies] ... the new guidelines state that we should move away from them, but I think that physicians in general have not gone that way. The way that we’re working, giving short acting every four hours – I don’t see that it’s a benefit to our patients,” said Sharon Armstead, who attended the session and was asked to comment on the study. She is a respiratory therapist at Ascension Health and an instructor at Concordia University, Austin, Texas. Ms. Armstead has asthma, and has first-hand experience as a patient when respiratory therapists are unable to attend to the patient every 4 hours.
She suggested that continued use of short-acting therapies may be due to inertia. “It’s easier [for a physician] to click a button on [a computer screen] than to actually slow down and write the order. If we need a rescue, then we’ll call for a rescue,” Ms. Armstead said.
She anticipates that long-acting therapies will ultimately lead to better outcomes because they will increase the time that respiratory therapists can spend with patients. “That’s what we really want to do. We want to spend time with our patients and stay there and watch our patients. But if you’re just telling us to [administer a therapy] every 4 hours, it’s not really giving the patient what they need.”
Specifically, there were concerns about cardiovascular safety, but the researchers found no between-group differences.
Asked for comment, session co-moderator Brittany Duchene, MD remarked: “It’s super interesting, but I worry about the cost. From a practical perspective, it’s challenging to get those drugs placed on an outpatient basis. They are very expensive, and they’re newer [drugs], but I think overall it’s good to give less,” said Dr. Duchene, a pulmonary critical care physician at Northeastern Vermont Regional Hospital, St. Johnsbury.
A potential concern raised by one audience member is that some patients are used to frequent treatment and may grow anxious with less frequent therapy. “I think we just need some reeducation that this is like a long-acting medicine. It also decreases the burden on our respiratory therapists, which is very good,” said Dr. Duchene.
The study was funded by Mylan/Theravance Biopharma. Dr. Dhand has received research support from Theravance, Mylan, and Viatris. He has received honoraria from Teva and UpToDate. Ms. Armstead and Dr. Duchene have no relevant financial disclosures.
HONOLULU – in safety and efficacy to a short-acting combination of albuterol and ipratropium.
The 2023 Gold Report on prevention, management, and diagnosis of COPD recommended switching to long-acting bronchodilators despite a lack of clinical evidence showing safety in patients hospitalized for COPD exacerbation, according to Rajiv Dhand, MD, who presented the new study at the annual meeting of the American College of Chest Physicians (CHEST).
“We wanted to establish the safety, because long-acting agents are approved only for use in nonhospitalized patients. We established that it was safe and that it was comparably effective, but you could give 30% lower doses. Patients don’t have to be woken up to get the medication, and there’s a better chance that all the doses will be administered to these patients. So I think that it provides convenience with similar efficacy and safety,” said Dr. Dhand, a pulmonologist and professor of medicine at the University of Tennessee, Knoxville.
The researchers randomized 60 patients to receive nebulized albuterol (2.5 mg) and ipratropium (0.5 mg) every 6 hours (short-acting group) or nebulized formoterol (20 mcg) every 12 hours and revefenacin (175 mcg) every 24 hours (long-acting group). The mean age was 63.2 years, 58.3% were male, and 65% were current smokers.
The median decrease between day 1 and day 3 in the Modified Borg Dyspnea score was 4.0 in the long-acting group (P < .001), and 2.0 in the short-acting group, though the latter was not statistically significant (P = .134). Both groups had a decrease in supplemental oxygen requirement, with no difference between the two groups. There was also no difference in the number of respiratory visits for rescue therapy.
Respiratory therapists in the audience welcomed the new evidence. “As a respiratory therapist, I feel that we should move away from giving good short acting [therapies] ... the new guidelines state that we should move away from them, but I think that physicians in general have not gone that way. The way that we’re working, giving short acting every four hours – I don’t see that it’s a benefit to our patients,” said Sharon Armstead, who attended the session and was asked to comment on the study. She is a respiratory therapist at Ascension Health and an instructor at Concordia University, Austin, Texas. Ms. Armstead has asthma, and has first-hand experience as a patient when respiratory therapists are unable to attend to the patient every 4 hours.
She suggested that continued use of short-acting therapies may be due to inertia. “It’s easier [for a physician] to click a button on [a computer screen] than to actually slow down and write the order. If we need a rescue, then we’ll call for a rescue,” Ms. Armstead said.
She anticipates that long-acting therapies will ultimately lead to better outcomes because they will increase the time that respiratory therapists can spend with patients. “That’s what we really want to do. We want to spend time with our patients and stay there and watch our patients. But if you’re just telling us to [administer a therapy] every 4 hours, it’s not really giving the patient what they need.”
Specifically, there were concerns about cardiovascular safety, but the researchers found no between-group differences.
Asked for comment, session co-moderator Brittany Duchene, MD remarked: “It’s super interesting, but I worry about the cost. From a practical perspective, it’s challenging to get those drugs placed on an outpatient basis. They are very expensive, and they’re newer [drugs], but I think overall it’s good to give less,” said Dr. Duchene, a pulmonary critical care physician at Northeastern Vermont Regional Hospital, St. Johnsbury.
A potential concern raised by one audience member is that some patients are used to frequent treatment and may grow anxious with less frequent therapy. “I think we just need some reeducation that this is like a long-acting medicine. It also decreases the burden on our respiratory therapists, which is very good,” said Dr. Duchene.
The study was funded by Mylan/Theravance Biopharma. Dr. Dhand has received research support from Theravance, Mylan, and Viatris. He has received honoraria from Teva and UpToDate. Ms. Armstead and Dr. Duchene have no relevant financial disclosures.
HONOLULU – in safety and efficacy to a short-acting combination of albuterol and ipratropium.
The 2023 Gold Report on prevention, management, and diagnosis of COPD recommended switching to long-acting bronchodilators despite a lack of clinical evidence showing safety in patients hospitalized for COPD exacerbation, according to Rajiv Dhand, MD, who presented the new study at the annual meeting of the American College of Chest Physicians (CHEST).
“We wanted to establish the safety, because long-acting agents are approved only for use in nonhospitalized patients. We established that it was safe and that it was comparably effective, but you could give 30% lower doses. Patients don’t have to be woken up to get the medication, and there’s a better chance that all the doses will be administered to these patients. So I think that it provides convenience with similar efficacy and safety,” said Dr. Dhand, a pulmonologist and professor of medicine at the University of Tennessee, Knoxville.
The researchers randomized 60 patients to receive nebulized albuterol (2.5 mg) and ipratropium (0.5 mg) every 6 hours (short-acting group) or nebulized formoterol (20 mcg) every 12 hours and revefenacin (175 mcg) every 24 hours (long-acting group). The mean age was 63.2 years, 58.3% were male, and 65% were current smokers.
The median decrease between day 1 and day 3 in the Modified Borg Dyspnea score was 4.0 in the long-acting group (P < .001), and 2.0 in the short-acting group, though the latter was not statistically significant (P = .134). Both groups had a decrease in supplemental oxygen requirement, with no difference between the two groups. There was also no difference in the number of respiratory visits for rescue therapy.
Respiratory therapists in the audience welcomed the new evidence. “As a respiratory therapist, I feel that we should move away from giving good short acting [therapies] ... the new guidelines state that we should move away from them, but I think that physicians in general have not gone that way. The way that we’re working, giving short acting every four hours – I don’t see that it’s a benefit to our patients,” said Sharon Armstead, who attended the session and was asked to comment on the study. She is a respiratory therapist at Ascension Health and an instructor at Concordia University, Austin, Texas. Ms. Armstead has asthma, and has first-hand experience as a patient when respiratory therapists are unable to attend to the patient every 4 hours.
She suggested that continued use of short-acting therapies may be due to inertia. “It’s easier [for a physician] to click a button on [a computer screen] than to actually slow down and write the order. If we need a rescue, then we’ll call for a rescue,” Ms. Armstead said.
She anticipates that long-acting therapies will ultimately lead to better outcomes because they will increase the time that respiratory therapists can spend with patients. “That’s what we really want to do. We want to spend time with our patients and stay there and watch our patients. But if you’re just telling us to [administer a therapy] every 4 hours, it’s not really giving the patient what they need.”
Specifically, there were concerns about cardiovascular safety, but the researchers found no between-group differences.
Asked for comment, session co-moderator Brittany Duchene, MD remarked: “It’s super interesting, but I worry about the cost. From a practical perspective, it’s challenging to get those drugs placed on an outpatient basis. They are very expensive, and they’re newer [drugs], but I think overall it’s good to give less,” said Dr. Duchene, a pulmonary critical care physician at Northeastern Vermont Regional Hospital, St. Johnsbury.
A potential concern raised by one audience member is that some patients are used to frequent treatment and may grow anxious with less frequent therapy. “I think we just need some reeducation that this is like a long-acting medicine. It also decreases the burden on our respiratory therapists, which is very good,” said Dr. Duchene.
The study was funded by Mylan/Theravance Biopharma. Dr. Dhand has received research support from Theravance, Mylan, and Viatris. He has received honoraria from Teva and UpToDate. Ms. Armstead and Dr. Duchene have no relevant financial disclosures.
AT CHEST 2023
Respiratory infections, asthma rise before type 2 diabetes
HAMBURG, GERMANY – , shows a longitudinal study looking at comorbidities both 25 years before and 25 years after a type 2 diabetes diagnosis.
About 40% of people had respiratory tract infections at the time of diagnosis with type 2 diabetes, compared with 4% who were not diagnosed. Likewise, ear, nose, and throat infections were present in 20% of people at type 2 diabetes diagnosis, compared with around 2% who were not diagnosed. A similar pattern was seen with asthma.
Taken together, the data suggest that subacute inflammation manifesting in asthma as well as the onset of asthma or an acute infection may be a precursor to a type 2 diabetes diagnosis.
“We have also found that in the years prior to diagnosis, there are associations with infections and inflammatory disorders to a much greater degree than in those people who do not get a diabetes diagnosis but who have very similar demographics,” Adrian Heald, MD, study lead and diabetes consultant from Salford (England) Royal Hospital, said in an interview.
Five years prior to diagnosis, respiratory tract infections were documented in around 23% of patients who were later diagnosed with type 2 diabetes versus 2.5% in those not diagnosed, and a similar pattern was seen for ear, nose, and throat infections and asthma. The findings suggest that patients reporting infections, in addition to other known risk factors for type 2 diabetes, might benefit from diabetes tests and early interventions, if needed.
“These novel insights offer a fascinating and fresh perspective on the onset and natural progression to type 2 diabetes and beyond, suggesting an early phase of inflammation-related disease activity long before any clinical diagnosis of type 2 diabetes is made.”
Dr. Heald points out that clinicians may intervene to stave off progression to a type 2 diabetes diagnosis in at risk patients. “At this point, an intervention could relate to lifestyle changes and involve highlighting to the patient that the morbidity they have already accumulated is suggestive of diabetes risk,” he said, adding that, “they may have dyslipidemia, hypertension, and most often excess weight so annual checks of their HbA1c, weight management, and blood pressure would need checking,” he explained.
Moderator Coen Stehouwer, MD, professor of internal medicine at Maastricht University, the Netherlands, commented, “Before clinical diagnosis of type 2 diabetes there is often a lengthy period of undiagnosed disease and before that, prediabetes, because glucose can be abnormal up to 10 years prior to clinical diagnosis.”
But he added that, “It’s not entirely clear whether the rise seen before clinical diagnosis in this study correlates with undiagnosed diabetes or prediabetes or even if it precedes type 2 diabetes – it might be because inflammation is a common origin for type 2 diabetes and various comorbidities. This might explain how they go together.”
Longitudinal study 25 years before and 25 years after type 2 diagnosis
Dr. Heald presented the findings at a session on inflammation in diabetes at the annual meeting of the European Association for the Study of Diabetes. The work was also published in Diabetes Therapy.
The researchers wanted to investigate the pattern of comorbidities in the years and decades prior to a diagnosis of type 2 diabetes as well as after: “With the database we used, called DARE [Diabetes Alliance for Research in England], we are able to explore phenomena longitudinally going right back to the beginning of their digital health records, looking at phenotypes over time.”
By mapping significant health issues in people who went on to develop type 2 diabetes alongside those that did not, Dr. Heald managed to develop a continuum spanning 25 years prior and 25 years after diagnosis of type 2 diabetes. The researchers also examined relationships between sociodemographic factors and longitudinal health outcomes of relevance to cardiac conditions and lower respiratory tract infections. His talk in Hamburg primarily addressed clinical phenotypes before the point of diagnosis.
Data were drawn from 1,932 people with (1,196) and without (736) type 2 diabetes. Participants in both groups were aged 66-67 years, 43%-46% were women, age at diagnosis was 50-52 years, and participants lived in Greater Manchester, United Kingdom.
In the years leading up to type 2 diagnosis, individuals consistently exhibited a considerable increase in several clinical phenotypes, reported Dr. Heald. Of note, he added, “immediately prior to type 2 diagnosis, there was a significantly greater proportion of hypertension at 35%, respiratory tract infection at 34%, heart disease at 17%, ear, nose, and throat infection at 19%, and asthma at 12%. And by comparison, the corresponding disease trajectory in matched controls was much less dramatic.”
“There is a huge difference in people who went on to receive a diagnosis of type 2 diabetes and those who did not, and not just what we’d expect – so hypertension for example or manifestations of renal disease, but importantly inflammatory disorders are more common,” he emphasized.
In addition, a larger signal for ischemic heart disease was seen just before type 2 diabetes diagnosis.
These data suggest that longitudinal clinical histories prior to a diagnosis of type 2 diabetes might offer new information, both genetic and nongenetic, about development of type 2 diabetes in relation to comorbidities.
After type 2 diabetes diagnosis, the proportion of people exhibiting coronary artery disease, hypertension, chronic kidney disease, retinopathy, and infections climbed rapidly before plateauing, reported Dr. Heald. “We also know that individuals with coronary artery disease are more highly represented in socially disadvantaged groups, and this is borne out in the data at 25 years prior and after type 2 diagnosis.”
Dr. Heald has received speaker fees or contributed to advisory boards from Lilly, AstraZeneca, Janssen, Bristol-Myers Squibb, Besins, Bayer, Sanofi, and Recordati. Research grants from Novo Nordisk, Pfizer, and Besins. Professor Stehouwer has declared no relevant conflicts.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – , shows a longitudinal study looking at comorbidities both 25 years before and 25 years after a type 2 diabetes diagnosis.
About 40% of people had respiratory tract infections at the time of diagnosis with type 2 diabetes, compared with 4% who were not diagnosed. Likewise, ear, nose, and throat infections were present in 20% of people at type 2 diabetes diagnosis, compared with around 2% who were not diagnosed. A similar pattern was seen with asthma.
Taken together, the data suggest that subacute inflammation manifesting in asthma as well as the onset of asthma or an acute infection may be a precursor to a type 2 diabetes diagnosis.
“We have also found that in the years prior to diagnosis, there are associations with infections and inflammatory disorders to a much greater degree than in those people who do not get a diabetes diagnosis but who have very similar demographics,” Adrian Heald, MD, study lead and diabetes consultant from Salford (England) Royal Hospital, said in an interview.
Five years prior to diagnosis, respiratory tract infections were documented in around 23% of patients who were later diagnosed with type 2 diabetes versus 2.5% in those not diagnosed, and a similar pattern was seen for ear, nose, and throat infections and asthma. The findings suggest that patients reporting infections, in addition to other known risk factors for type 2 diabetes, might benefit from diabetes tests and early interventions, if needed.
“These novel insights offer a fascinating and fresh perspective on the onset and natural progression to type 2 diabetes and beyond, suggesting an early phase of inflammation-related disease activity long before any clinical diagnosis of type 2 diabetes is made.”
Dr. Heald points out that clinicians may intervene to stave off progression to a type 2 diabetes diagnosis in at risk patients. “At this point, an intervention could relate to lifestyle changes and involve highlighting to the patient that the morbidity they have already accumulated is suggestive of diabetes risk,” he said, adding that, “they may have dyslipidemia, hypertension, and most often excess weight so annual checks of their HbA1c, weight management, and blood pressure would need checking,” he explained.
Moderator Coen Stehouwer, MD, professor of internal medicine at Maastricht University, the Netherlands, commented, “Before clinical diagnosis of type 2 diabetes there is often a lengthy period of undiagnosed disease and before that, prediabetes, because glucose can be abnormal up to 10 years prior to clinical diagnosis.”
But he added that, “It’s not entirely clear whether the rise seen before clinical diagnosis in this study correlates with undiagnosed diabetes or prediabetes or even if it precedes type 2 diabetes – it might be because inflammation is a common origin for type 2 diabetes and various comorbidities. This might explain how they go together.”
Longitudinal study 25 years before and 25 years after type 2 diagnosis
Dr. Heald presented the findings at a session on inflammation in diabetes at the annual meeting of the European Association for the Study of Diabetes. The work was also published in Diabetes Therapy.
The researchers wanted to investigate the pattern of comorbidities in the years and decades prior to a diagnosis of type 2 diabetes as well as after: “With the database we used, called DARE [Diabetes Alliance for Research in England], we are able to explore phenomena longitudinally going right back to the beginning of their digital health records, looking at phenotypes over time.”
By mapping significant health issues in people who went on to develop type 2 diabetes alongside those that did not, Dr. Heald managed to develop a continuum spanning 25 years prior and 25 years after diagnosis of type 2 diabetes. The researchers also examined relationships between sociodemographic factors and longitudinal health outcomes of relevance to cardiac conditions and lower respiratory tract infections. His talk in Hamburg primarily addressed clinical phenotypes before the point of diagnosis.
Data were drawn from 1,932 people with (1,196) and without (736) type 2 diabetes. Participants in both groups were aged 66-67 years, 43%-46% were women, age at diagnosis was 50-52 years, and participants lived in Greater Manchester, United Kingdom.
In the years leading up to type 2 diagnosis, individuals consistently exhibited a considerable increase in several clinical phenotypes, reported Dr. Heald. Of note, he added, “immediately prior to type 2 diagnosis, there was a significantly greater proportion of hypertension at 35%, respiratory tract infection at 34%, heart disease at 17%, ear, nose, and throat infection at 19%, and asthma at 12%. And by comparison, the corresponding disease trajectory in matched controls was much less dramatic.”
“There is a huge difference in people who went on to receive a diagnosis of type 2 diabetes and those who did not, and not just what we’d expect – so hypertension for example or manifestations of renal disease, but importantly inflammatory disorders are more common,” he emphasized.
In addition, a larger signal for ischemic heart disease was seen just before type 2 diabetes diagnosis.
These data suggest that longitudinal clinical histories prior to a diagnosis of type 2 diabetes might offer new information, both genetic and nongenetic, about development of type 2 diabetes in relation to comorbidities.
After type 2 diabetes diagnosis, the proportion of people exhibiting coronary artery disease, hypertension, chronic kidney disease, retinopathy, and infections climbed rapidly before plateauing, reported Dr. Heald. “We also know that individuals with coronary artery disease are more highly represented in socially disadvantaged groups, and this is borne out in the data at 25 years prior and after type 2 diagnosis.”
Dr. Heald has received speaker fees or contributed to advisory boards from Lilly, AstraZeneca, Janssen, Bristol-Myers Squibb, Besins, Bayer, Sanofi, and Recordati. Research grants from Novo Nordisk, Pfizer, and Besins. Professor Stehouwer has declared no relevant conflicts.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – , shows a longitudinal study looking at comorbidities both 25 years before and 25 years after a type 2 diabetes diagnosis.
About 40% of people had respiratory tract infections at the time of diagnosis with type 2 diabetes, compared with 4% who were not diagnosed. Likewise, ear, nose, and throat infections were present in 20% of people at type 2 diabetes diagnosis, compared with around 2% who were not diagnosed. A similar pattern was seen with asthma.
Taken together, the data suggest that subacute inflammation manifesting in asthma as well as the onset of asthma or an acute infection may be a precursor to a type 2 diabetes diagnosis.
“We have also found that in the years prior to diagnosis, there are associations with infections and inflammatory disorders to a much greater degree than in those people who do not get a diabetes diagnosis but who have very similar demographics,” Adrian Heald, MD, study lead and diabetes consultant from Salford (England) Royal Hospital, said in an interview.
Five years prior to diagnosis, respiratory tract infections were documented in around 23% of patients who were later diagnosed with type 2 diabetes versus 2.5% in those not diagnosed, and a similar pattern was seen for ear, nose, and throat infections and asthma. The findings suggest that patients reporting infections, in addition to other known risk factors for type 2 diabetes, might benefit from diabetes tests and early interventions, if needed.
“These novel insights offer a fascinating and fresh perspective on the onset and natural progression to type 2 diabetes and beyond, suggesting an early phase of inflammation-related disease activity long before any clinical diagnosis of type 2 diabetes is made.”
Dr. Heald points out that clinicians may intervene to stave off progression to a type 2 diabetes diagnosis in at risk patients. “At this point, an intervention could relate to lifestyle changes and involve highlighting to the patient that the morbidity they have already accumulated is suggestive of diabetes risk,” he said, adding that, “they may have dyslipidemia, hypertension, and most often excess weight so annual checks of their HbA1c, weight management, and blood pressure would need checking,” he explained.
Moderator Coen Stehouwer, MD, professor of internal medicine at Maastricht University, the Netherlands, commented, “Before clinical diagnosis of type 2 diabetes there is often a lengthy period of undiagnosed disease and before that, prediabetes, because glucose can be abnormal up to 10 years prior to clinical diagnosis.”
But he added that, “It’s not entirely clear whether the rise seen before clinical diagnosis in this study correlates with undiagnosed diabetes or prediabetes or even if it precedes type 2 diabetes – it might be because inflammation is a common origin for type 2 diabetes and various comorbidities. This might explain how they go together.”
Longitudinal study 25 years before and 25 years after type 2 diagnosis
Dr. Heald presented the findings at a session on inflammation in diabetes at the annual meeting of the European Association for the Study of Diabetes. The work was also published in Diabetes Therapy.
The researchers wanted to investigate the pattern of comorbidities in the years and decades prior to a diagnosis of type 2 diabetes as well as after: “With the database we used, called DARE [Diabetes Alliance for Research in England], we are able to explore phenomena longitudinally going right back to the beginning of their digital health records, looking at phenotypes over time.”
By mapping significant health issues in people who went on to develop type 2 diabetes alongside those that did not, Dr. Heald managed to develop a continuum spanning 25 years prior and 25 years after diagnosis of type 2 diabetes. The researchers also examined relationships between sociodemographic factors and longitudinal health outcomes of relevance to cardiac conditions and lower respiratory tract infections. His talk in Hamburg primarily addressed clinical phenotypes before the point of diagnosis.
Data were drawn from 1,932 people with (1,196) and without (736) type 2 diabetes. Participants in both groups were aged 66-67 years, 43%-46% were women, age at diagnosis was 50-52 years, and participants lived in Greater Manchester, United Kingdom.
In the years leading up to type 2 diagnosis, individuals consistently exhibited a considerable increase in several clinical phenotypes, reported Dr. Heald. Of note, he added, “immediately prior to type 2 diagnosis, there was a significantly greater proportion of hypertension at 35%, respiratory tract infection at 34%, heart disease at 17%, ear, nose, and throat infection at 19%, and asthma at 12%. And by comparison, the corresponding disease trajectory in matched controls was much less dramatic.”
“There is a huge difference in people who went on to receive a diagnosis of type 2 diabetes and those who did not, and not just what we’d expect – so hypertension for example or manifestations of renal disease, but importantly inflammatory disorders are more common,” he emphasized.
In addition, a larger signal for ischemic heart disease was seen just before type 2 diabetes diagnosis.
These data suggest that longitudinal clinical histories prior to a diagnosis of type 2 diabetes might offer new information, both genetic and nongenetic, about development of type 2 diabetes in relation to comorbidities.
After type 2 diabetes diagnosis, the proportion of people exhibiting coronary artery disease, hypertension, chronic kidney disease, retinopathy, and infections climbed rapidly before plateauing, reported Dr. Heald. “We also know that individuals with coronary artery disease are more highly represented in socially disadvantaged groups, and this is borne out in the data at 25 years prior and after type 2 diagnosis.”
Dr. Heald has received speaker fees or contributed to advisory boards from Lilly, AstraZeneca, Janssen, Bristol-Myers Squibb, Besins, Bayer, Sanofi, and Recordati. Research grants from Novo Nordisk, Pfizer, and Besins. Professor Stehouwer has declared no relevant conflicts.
A version of this article first appeared on Medscape.com.
AT EASD 2023
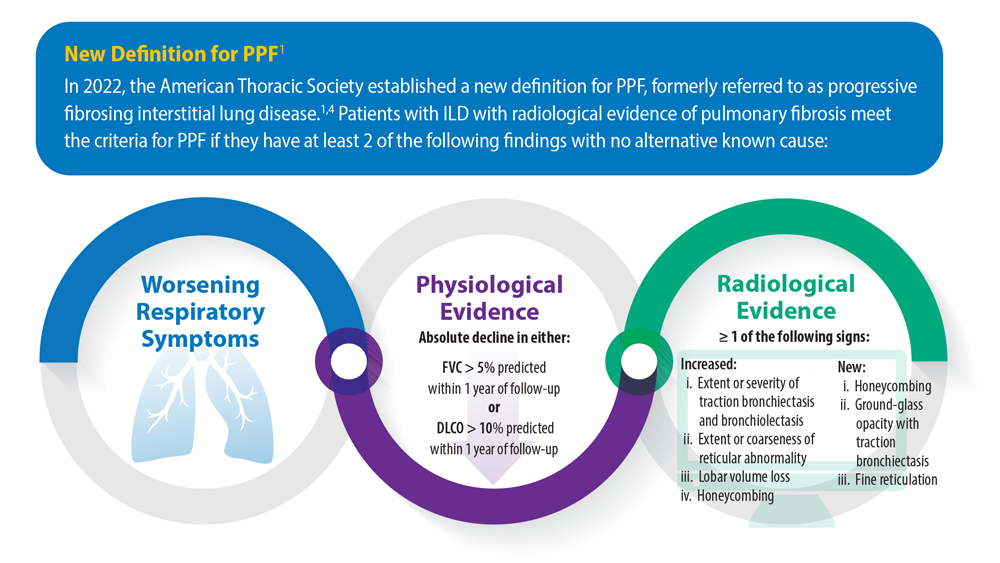
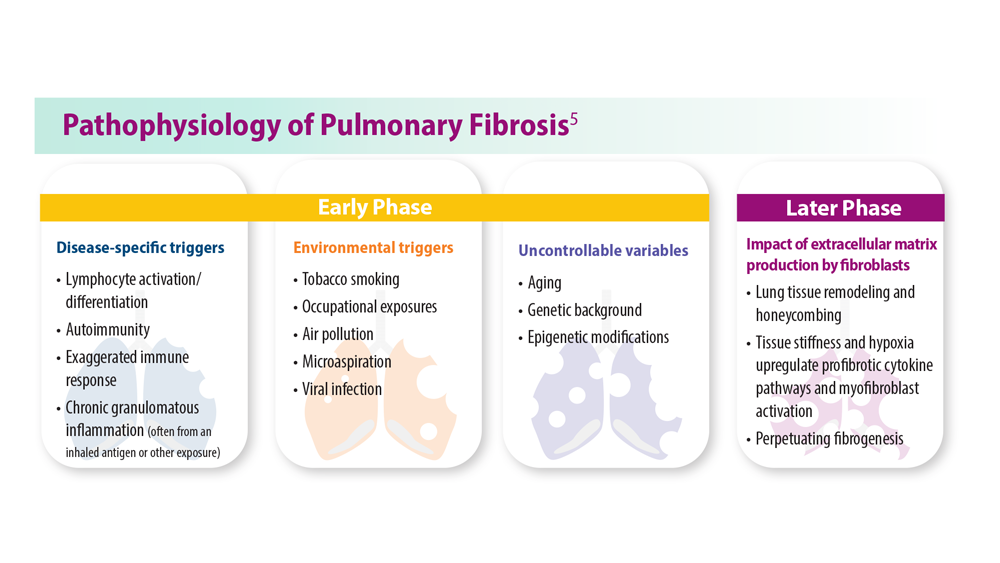
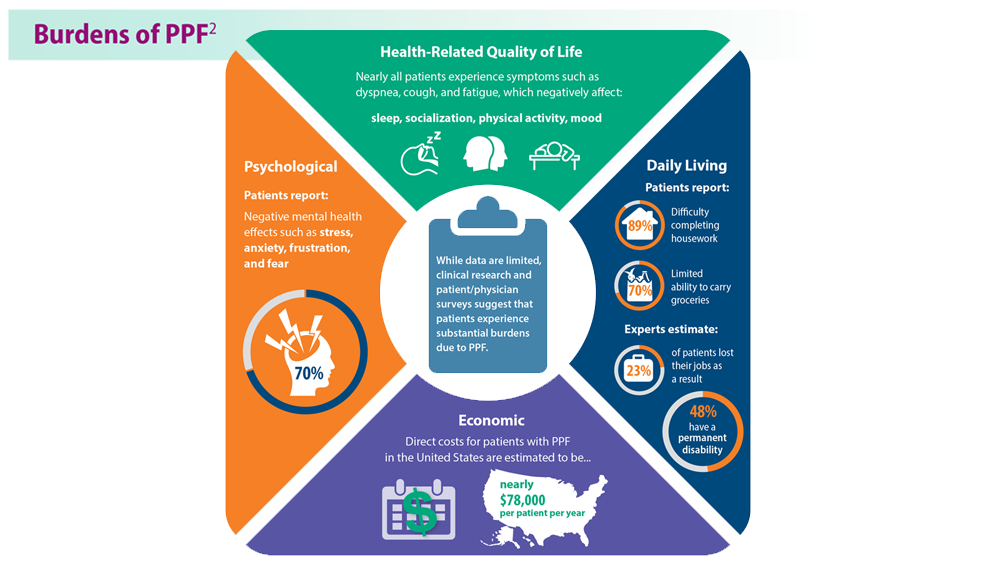
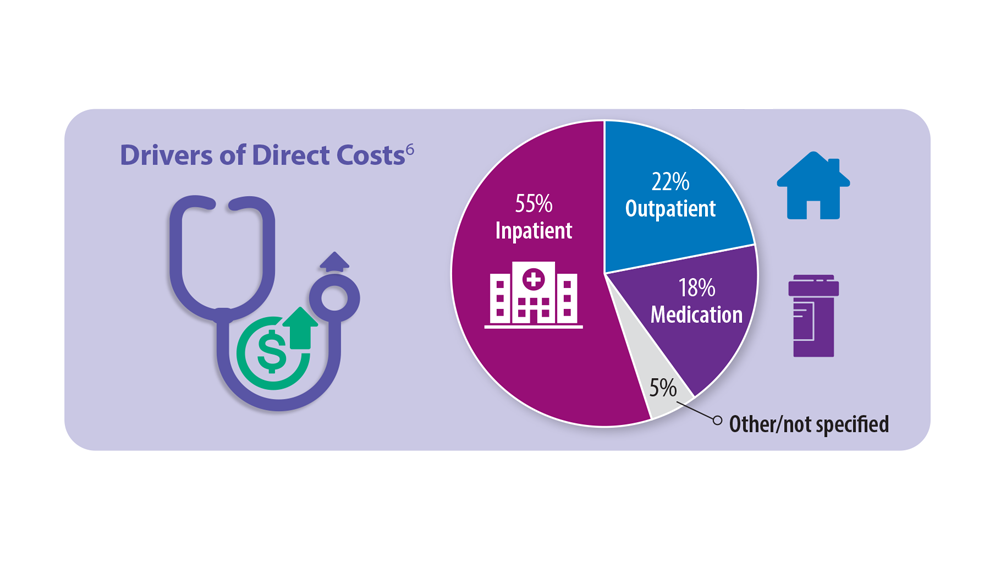
Progressive Pulmonary Fibrosis: Understanding Its Many Forms
- Raghu G et al. Am J Respir Crit Care Med. 2022;205(9):e18-e47. doi:10.1164/rccm.202202-0399ST
- Cottin V et al. Front Med (Lausanne). 2022;9:799912. doi:10.3389/fmed.2022.799912
- Molina-Molina M et al. Expert Rev Respir Med. 2022;16(7):765-774. doi:10.1080/17476348.2022.2107508
- Cottin V. Am J Respir Crit Care Med. 2023;207(1):11-13. doi:10.1164/rccm.202208-1639ED
- Wijsenbeek M, Cottin V. N Engl J Med. 2020;383(10):958-968. doi:10.1056/NEJMra2005230
- Chiu YH et al. Front Med (Lausanne). 2023;10:1106560. doi:10.3389/fmed.2023.1106560
- Wong AW et al. BMC Pulm Med. 2022;22(1):148. doi:10.1186/s12890-022-01922-2
- Raghu G et al. Am J Respir Crit Care Med. 2022;205(9):e18-e47. doi:10.1164/rccm.202202-0399ST
- Cottin V et al. Front Med (Lausanne). 2022;9:799912. doi:10.3389/fmed.2022.799912
- Molina-Molina M et al. Expert Rev Respir Med. 2022;16(7):765-774. doi:10.1080/17476348.2022.2107508
- Cottin V. Am J Respir Crit Care Med. 2023;207(1):11-13. doi:10.1164/rccm.202208-1639ED
- Wijsenbeek M, Cottin V. N Engl J Med. 2020;383(10):958-968. doi:10.1056/NEJMra2005230
- Chiu YH et al. Front Med (Lausanne). 2023;10:1106560. doi:10.3389/fmed.2023.1106560
- Wong AW et al. BMC Pulm Med. 2022;22(1):148. doi:10.1186/s12890-022-01922-2
- Raghu G et al. Am J Respir Crit Care Med. 2022;205(9):e18-e47. doi:10.1164/rccm.202202-0399ST
- Cottin V et al. Front Med (Lausanne). 2022;9:799912. doi:10.3389/fmed.2022.799912
- Molina-Molina M et al. Expert Rev Respir Med. 2022;16(7):765-774. doi:10.1080/17476348.2022.2107508
- Cottin V. Am J Respir Crit Care Med. 2023;207(1):11-13. doi:10.1164/rccm.202208-1639ED
- Wijsenbeek M, Cottin V. N Engl J Med. 2020;383(10):958-968. doi:10.1056/NEJMra2005230
- Chiu YH et al. Front Med (Lausanne). 2023;10:1106560. doi:10.3389/fmed.2023.1106560
- Wong AW et al. BMC Pulm Med. 2022;22(1):148. doi:10.1186/s12890-022-01922-2
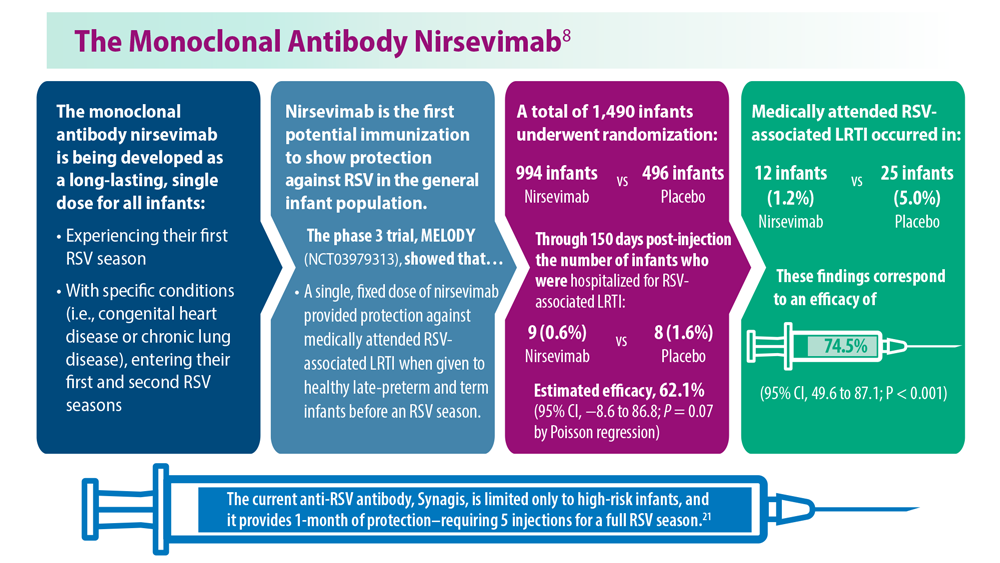
Long-Awaited RSV Vaccines Now Available for Older Adults and Pediatric Patients
- Jha A et al. Respiratory syncytial virus. In: Hui DS, Rossi GA, Johnston SL, eds. Respiratory Syncytial Virus. SARS, MERS and Other Viral Lung Infections. European Respiratory Society; 2016:chap 5. Accessed May 17, 2023.
- Ginsburg SA, Srikantiah P. Lancet Glob Health. 2021;9(12):e1644-e6145. doi:10.1016/S2214-109X(21)00455-1
- US Food and Drug Administration. FDA approves first respiratory syncytial virus (RSV) vaccine [press release]. Published May 3, 2023. Accessed May 17, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-respiratory-syncytial-virus-rsv-vaccine
- US Food and Drug Administration. FDA Approves New Drug to Prevent RSV in Babies and Toddlers [press release]. Published July 17, 2023. Accessed August 11, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-prevent-rsv-babies-and-toddlers
- US Food and Drug Administration. FDA Approves First Vaccine for Pregnant Individuals to Prevent RSV in Infants. Published August 21, 2023. Accessed August 22, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-vaccine-pregnant-individuals-prevent-rsv-infants
- Madhi SA et al. N Engl J Med. 2020;383(5):426-439. doi:10.1056/ NEJMoa1908380
- Centers for Disease Control. Advisory Committee on Immunization Practices (ACIP) Meeting recommendations, August 2023. https://www.cdc.gov/vaccines/acip/recommendations.html
- Hammit LL et al. N Engl J Med. 2022;386(9):837-846. doi:10.1056/ NEJMoa2110275
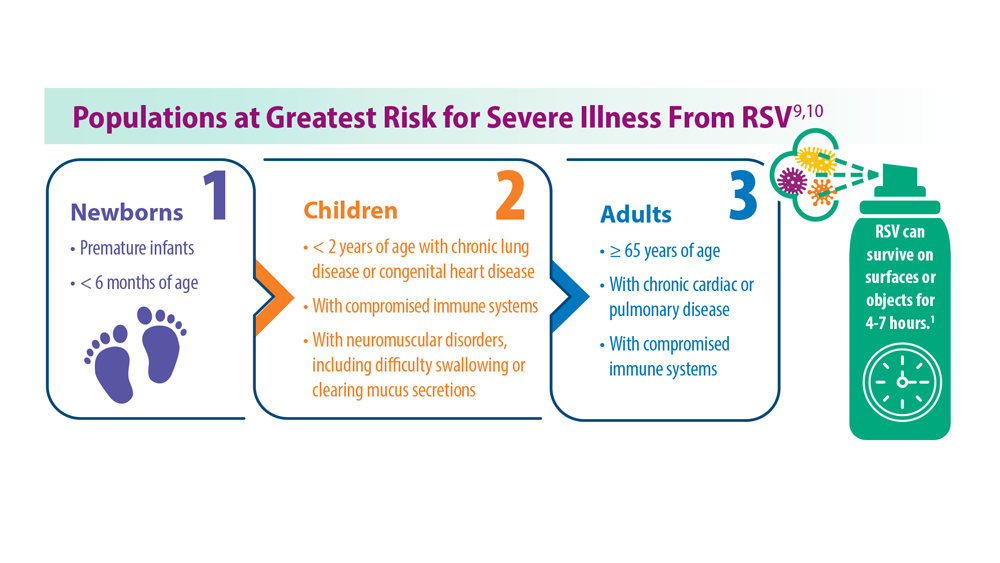
- Centers for Disease Control and Prevention. RSV in infants and young children. Updated October 28, 2022. Accessed May 30, 2023. https://www.cdc.gov/rsv/ high-risk/infants-young-children.html
- Centers for Disease Control and Prevention. RSV in older adults and adults with chronic medical conditions. Updated October 28, 2022. Accessed May 30, 2023. https://www.cdc.gov/rsv/high-risk/older-adults.html
- Widmer K et al. J Infect Dis. 2012;206(1):56-62. doi:10.1093/infdis/jis309
- Hall CB et al. N Engl J Med. 2009;360(6):588-598. doi:10.1056/NEJMoa0804877
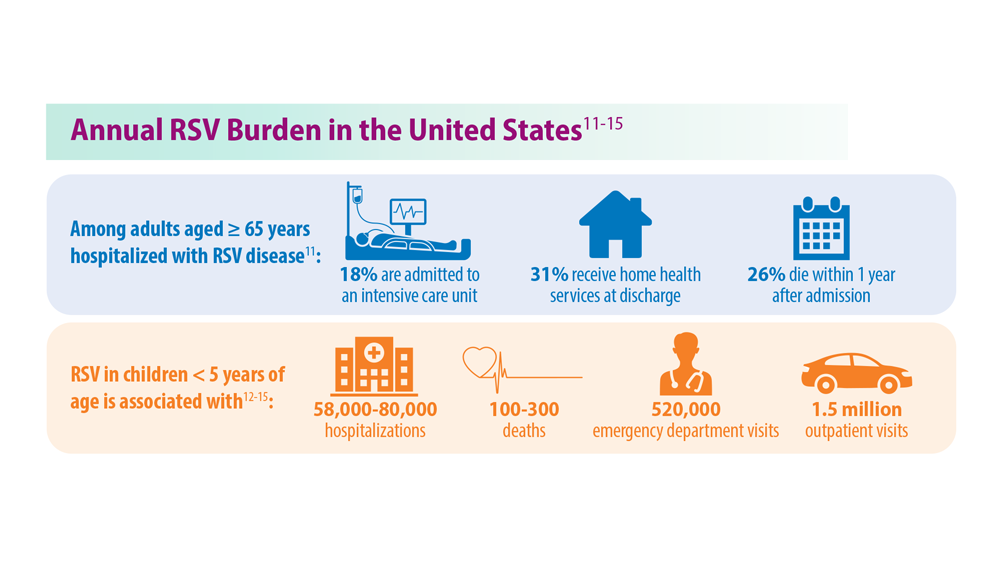
- McLaughlin JM et al. Open Forum Infect Dis. 2022;9(7):ofac300. doi:10.1093/ofid/ofac300
- Thompson et al. JAMA. 2003;289(2):179-186. doi:10.1001/jama.289.2.179
- Hansen CL et al. JAMA Netw Open. 2022;5(2):e220527. doi:10.1001/jamanetworkopen.2022.0527
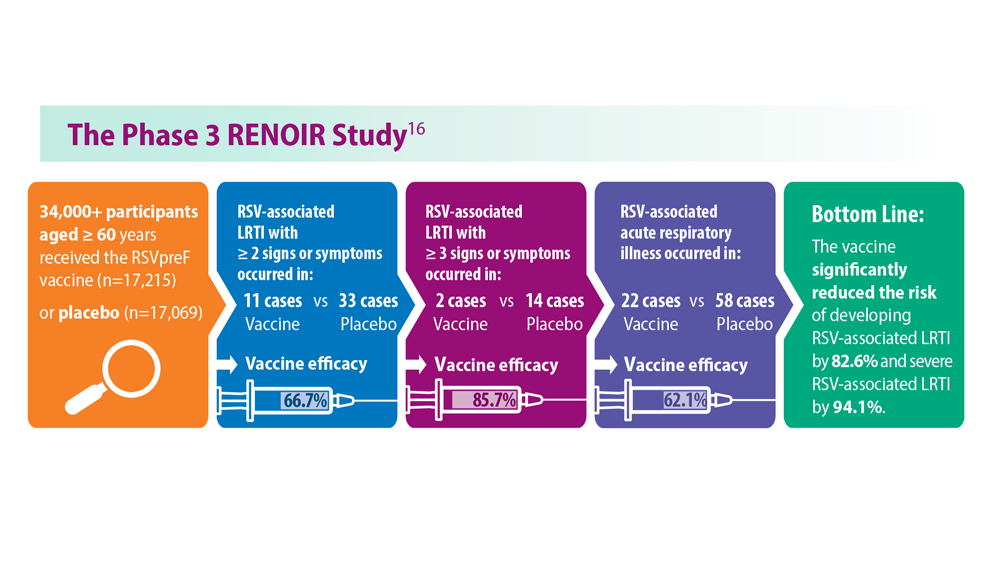
- Walsh EE et al; RENOIR Clinical Trial Group. N Engl J Med. 2023;388(16):1465-1477. doi:10.1056/NEJMoa2213836
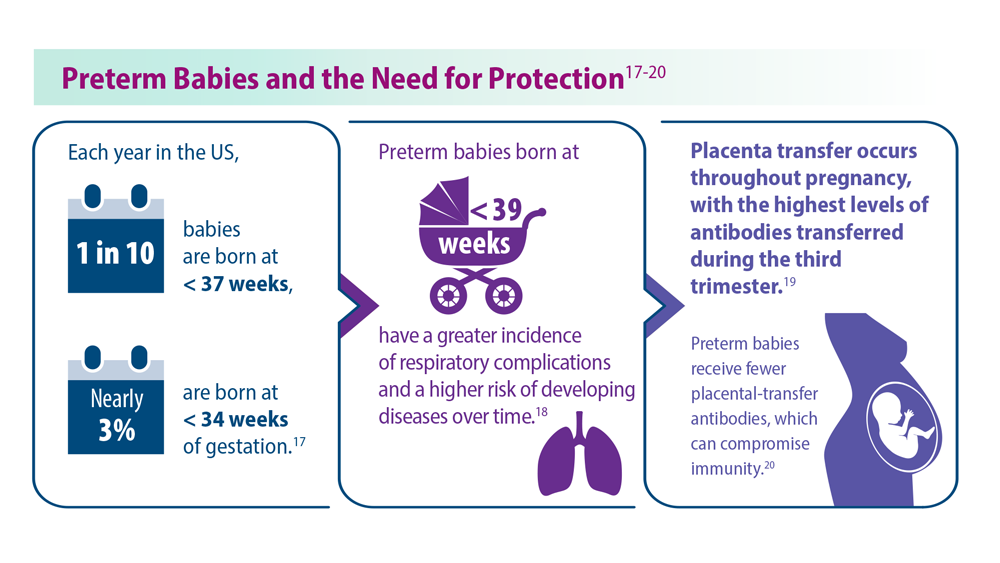
- Martin JA et al. Natl Vital Stat Rep. 2019;68(13):1-47. PMID:32501202
- Townsi N et al. Eur Clin Respir J. 2018;5(1):1487214. doi:10.1080/20018525.20 18.1487214
- Malek A et al. Am J Reprod Immunol. 1994;32(1):8-14. doi:10.1111/j.1600-0897.1994.tb00873.x
- Kampmann B et al; MATISSE Study Group. N Engl J Med. 2023;388(16):1451- 1464. doi:10.1056/NEJMoa2216480
- Synagis (palivizumab) injection prescribing information. Published June 2023. Accessed August 2023. https://www.synagis.com/synagis.pdf
- Jha A et al. Respiratory syncytial virus. In: Hui DS, Rossi GA, Johnston SL, eds. Respiratory Syncytial Virus. SARS, MERS and Other Viral Lung Infections. European Respiratory Society; 2016:chap 5. Accessed May 17, 2023.
- Ginsburg SA, Srikantiah P. Lancet Glob Health. 2021;9(12):e1644-e6145. doi:10.1016/S2214-109X(21)00455-1
- US Food and Drug Administration. FDA approves first respiratory syncytial virus (RSV) vaccine [press release]. Published May 3, 2023. Accessed May 17, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-respiratory-syncytial-virus-rsv-vaccine
- US Food and Drug Administration. FDA Approves New Drug to Prevent RSV in Babies and Toddlers [press release]. Published July 17, 2023. Accessed August 11, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-prevent-rsv-babies-and-toddlers
- US Food and Drug Administration. FDA Approves First Vaccine for Pregnant Individuals to Prevent RSV in Infants. Published August 21, 2023. Accessed August 22, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-vaccine-pregnant-individuals-prevent-rsv-infants
- Madhi SA et al. N Engl J Med. 2020;383(5):426-439. doi:10.1056/ NEJMoa1908380
- Centers for Disease Control. Advisory Committee on Immunization Practices (ACIP) Meeting recommendations, August 2023. https://www.cdc.gov/vaccines/acip/recommendations.html
- Hammit LL et al. N Engl J Med. 2022;386(9):837-846. doi:10.1056/ NEJMoa2110275
- Centers for Disease Control and Prevention. RSV in infants and young children. Updated October 28, 2022. Accessed May 30, 2023. https://www.cdc.gov/rsv/ high-risk/infants-young-children.html
- Centers for Disease Control and Prevention. RSV in older adults and adults with chronic medical conditions. Updated October 28, 2022. Accessed May 30, 2023. https://www.cdc.gov/rsv/high-risk/older-adults.html
- Widmer K et al. J Infect Dis. 2012;206(1):56-62. doi:10.1093/infdis/jis309
- Hall CB et al. N Engl J Med. 2009;360(6):588-598. doi:10.1056/NEJMoa0804877
- McLaughlin JM et al. Open Forum Infect Dis. 2022;9(7):ofac300. doi:10.1093/ofid/ofac300
- Thompson et al. JAMA. 2003;289(2):179-186. doi:10.1001/jama.289.2.179
- Hansen CL et al. JAMA Netw Open. 2022;5(2):e220527. doi:10.1001/jamanetworkopen.2022.0527
- Walsh EE et al; RENOIR Clinical Trial Group. N Engl J Med. 2023;388(16):1465-1477. doi:10.1056/NEJMoa2213836
- Martin JA et al. Natl Vital Stat Rep. 2019;68(13):1-47. PMID:32501202
- Townsi N et al. Eur Clin Respir J. 2018;5(1):1487214. doi:10.1080/20018525.20 18.1487214
- Malek A et al. Am J Reprod Immunol. 1994;32(1):8-14. doi:10.1111/j.1600-0897.1994.tb00873.x
- Kampmann B et al; MATISSE Study Group. N Engl J Med. 2023;388(16):1451- 1464. doi:10.1056/NEJMoa2216480
- Synagis (palivizumab) injection prescribing information. Published June 2023. Accessed August 2023. https://www.synagis.com/synagis.pdf
- Jha A et al. Respiratory syncytial virus. In: Hui DS, Rossi GA, Johnston SL, eds. Respiratory Syncytial Virus. SARS, MERS and Other Viral Lung Infections. European Respiratory Society; 2016:chap 5. Accessed May 17, 2023.
- Ginsburg SA, Srikantiah P. Lancet Glob Health. 2021;9(12):e1644-e6145. doi:10.1016/S2214-109X(21)00455-1
- US Food and Drug Administration. FDA approves first respiratory syncytial virus (RSV) vaccine [press release]. Published May 3, 2023. Accessed May 17, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-respiratory-syncytial-virus-rsv-vaccine
- US Food and Drug Administration. FDA Approves New Drug to Prevent RSV in Babies and Toddlers [press release]. Published July 17, 2023. Accessed August 11, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-prevent-rsv-babies-and-toddlers
- US Food and Drug Administration. FDA Approves First Vaccine for Pregnant Individuals to Prevent RSV in Infants. Published August 21, 2023. Accessed August 22, 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-vaccine-pregnant-individuals-prevent-rsv-infants
- Madhi SA et al. N Engl J Med. 2020;383(5):426-439. doi:10.1056/ NEJMoa1908380
- Centers for Disease Control. Advisory Committee on Immunization Practices (ACIP) Meeting recommendations, August 2023. https://www.cdc.gov/vaccines/acip/recommendations.html
- Hammit LL et al. N Engl J Med. 2022;386(9):837-846. doi:10.1056/ NEJMoa2110275
- Centers for Disease Control and Prevention. RSV in infants and young children. Updated October 28, 2022. Accessed May 30, 2023. https://www.cdc.gov/rsv/ high-risk/infants-young-children.html
- Centers for Disease Control and Prevention. RSV in older adults and adults with chronic medical conditions. Updated October 28, 2022. Accessed May 30, 2023. https://www.cdc.gov/rsv/high-risk/older-adults.html
- Widmer K et al. J Infect Dis. 2012;206(1):56-62. doi:10.1093/infdis/jis309
- Hall CB et al. N Engl J Med. 2009;360(6):588-598. doi:10.1056/NEJMoa0804877
- McLaughlin JM et al. Open Forum Infect Dis. 2022;9(7):ofac300. doi:10.1093/ofid/ofac300
- Thompson et al. JAMA. 2003;289(2):179-186. doi:10.1001/jama.289.2.179
- Hansen CL et al. JAMA Netw Open. 2022;5(2):e220527. doi:10.1001/jamanetworkopen.2022.0527
- Walsh EE et al; RENOIR Clinical Trial Group. N Engl J Med. 2023;388(16):1465-1477. doi:10.1056/NEJMoa2213836
- Martin JA et al. Natl Vital Stat Rep. 2019;68(13):1-47. PMID:32501202
- Townsi N et al. Eur Clin Respir J. 2018;5(1):1487214. doi:10.1080/20018525.20 18.1487214
- Malek A et al. Am J Reprod Immunol. 1994;32(1):8-14. doi:10.1111/j.1600-0897.1994.tb00873.x
- Kampmann B et al; MATISSE Study Group. N Engl J Med. 2023;388(16):1451- 1464. doi:10.1056/NEJMoa2216480
- Synagis (palivizumab) injection prescribing information. Published June 2023. Accessed August 2023. https://www.synagis.com/synagis.pdf
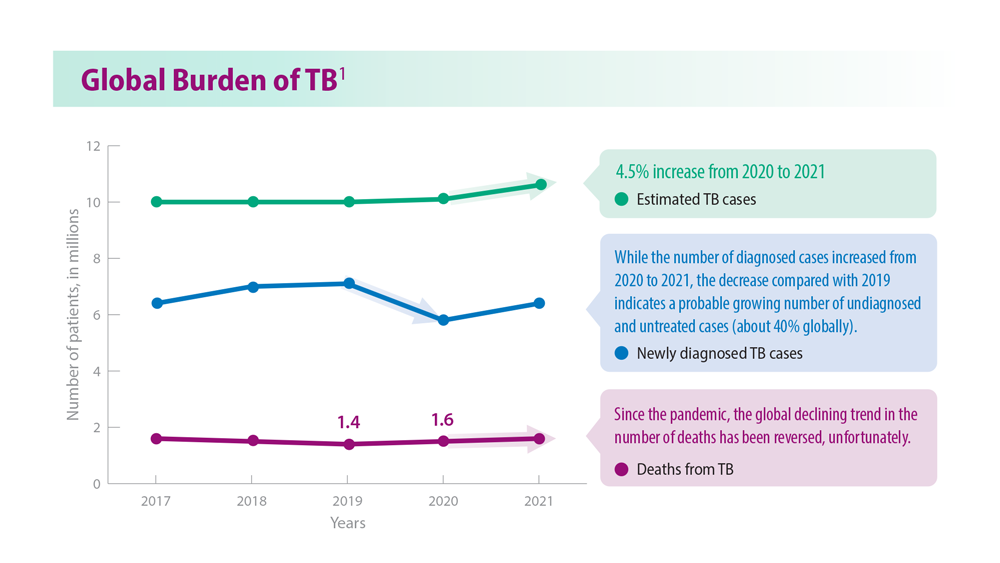
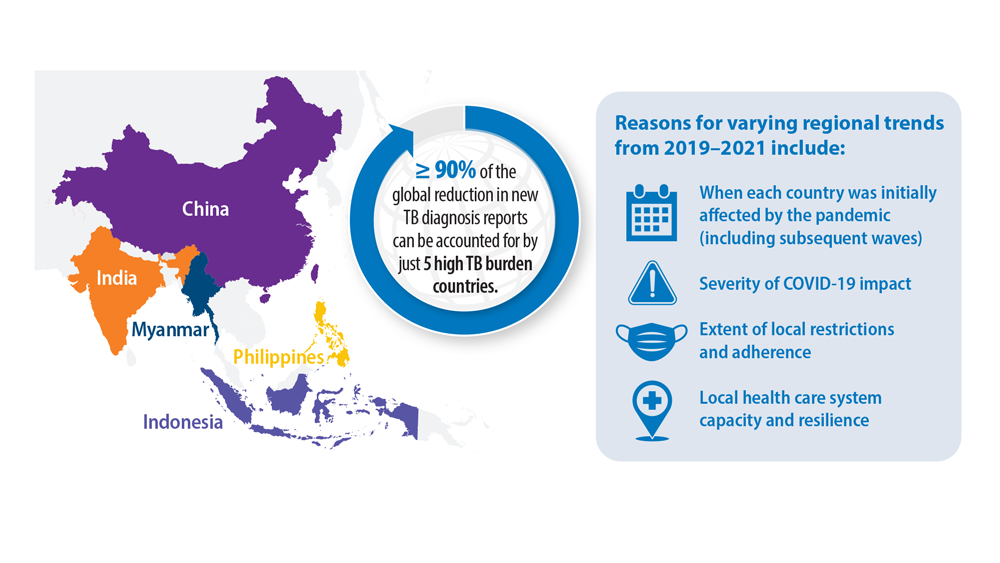
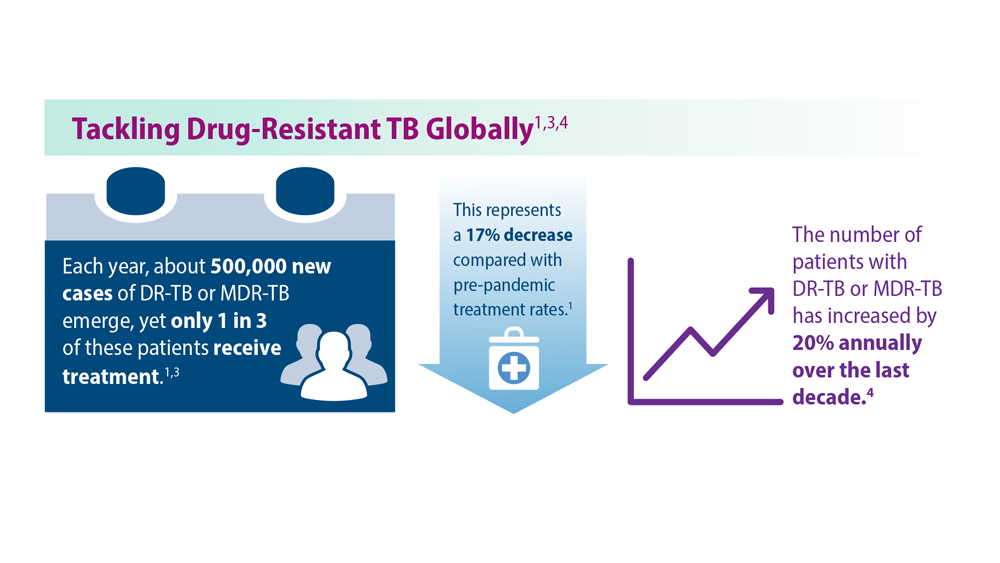
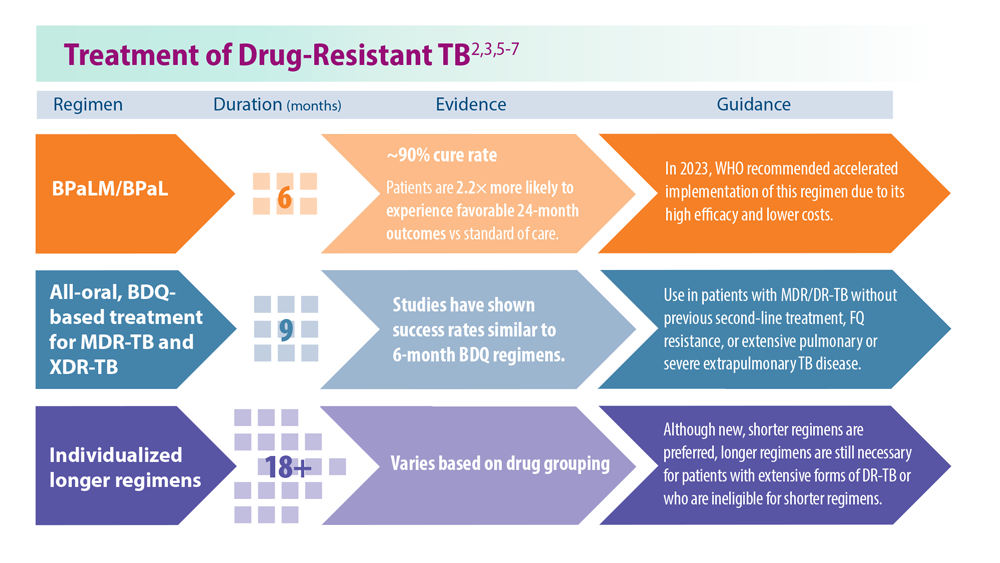
Tuberculosis Management: Returning to Pre-Pandemic Priorities
- Global tuberculosis report 2022. World Health Organization. Published October 27, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240061729
- WHO consolidated guidelines on tuberculosis. Module 4: treatment – drug-resistant tuberculosis treatment, 2022 update. World Health Organization. Published December 15, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240063129
- Migliori GB, Tiberi S. Int J Tuberc Lung Dis. 2022 ;26(7):590-591. doi:10.5588/ijtld.22.0263.
- Lange C et al. Am J Respir Crit Care Med. 2022;205(10):1142-1144. doi:10.1164/rccm.202202-0393ED
- Esmail A et al. Am J Respir Crit Care Med. 2022;205(10):1214-1227. doi:10.1164/rccm.202107-1779OC
- WHO BPaLM Accelerator Platform: to support the call to action for implementation of the shorter and more effective treatment for all people suffering from drug-resistant TB. World Health Organization. Published May 9, 2023. Accessed June 26, 2023. https://www.who.int/news-room/events/detail/2023/05/09/default-calendar/who-bpalm-accelerator-platform–to-support-the-call-to-action-for-implementation-of-the-shorter-and-moreeffective-
- Trevisi L et al. Am J Respir Crit Care Med. 2023;207(11):1525-1532. doi:10.1164/rccm.202211-2125OC
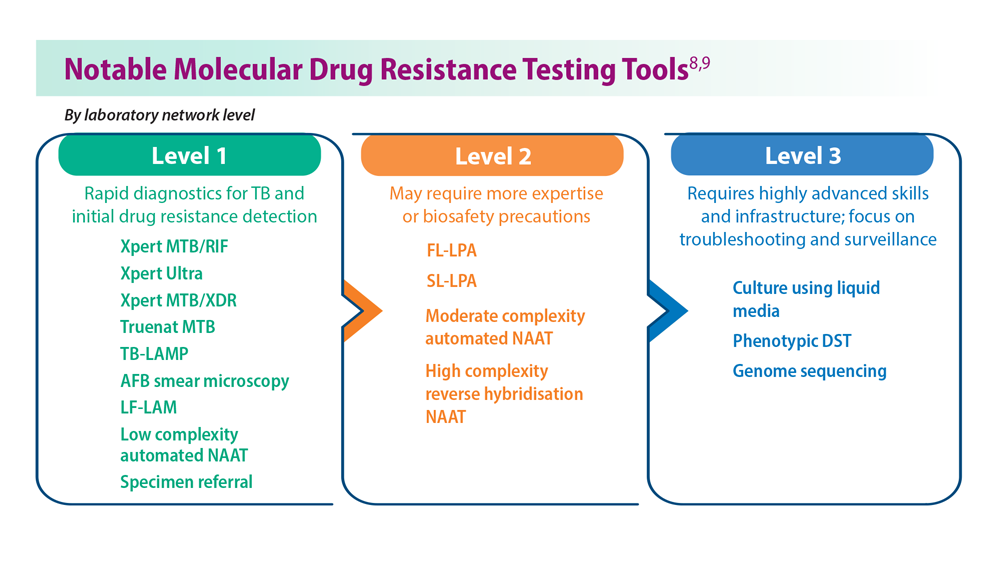
- Domínguez J et al; TBnet and RESIST-TB networks. Lancet Infect Dis. 2023;23(4):e122-e137. doi:10.1016/S1473-3099(22)00875-1
- WHO operational handbook on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 2021 update. World Health Organization. Published July 7, 2021. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240030589treatment-for-all-people-suffering-from-drug-resistant-tb
- Global tuberculosis report 2022. World Health Organization. Published October 27, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240061729
- WHO consolidated guidelines on tuberculosis. Module 4: treatment – drug-resistant tuberculosis treatment, 2022 update. World Health Organization. Published December 15, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240063129
- Migliori GB, Tiberi S. Int J Tuberc Lung Dis. 2022 ;26(7):590-591. doi:10.5588/ijtld.22.0263.
- Lange C et al. Am J Respir Crit Care Med. 2022;205(10):1142-1144. doi:10.1164/rccm.202202-0393ED
- Esmail A et al. Am J Respir Crit Care Med. 2022;205(10):1214-1227. doi:10.1164/rccm.202107-1779OC
- WHO BPaLM Accelerator Platform: to support the call to action for implementation of the shorter and more effective treatment for all people suffering from drug-resistant TB. World Health Organization. Published May 9, 2023. Accessed June 26, 2023. https://www.who.int/news-room/events/detail/2023/05/09/default-calendar/who-bpalm-accelerator-platform–to-support-the-call-to-action-for-implementation-of-the-shorter-and-moreeffective-
- Trevisi L et al. Am J Respir Crit Care Med. 2023;207(11):1525-1532. doi:10.1164/rccm.202211-2125OC
- Domínguez J et al; TBnet and RESIST-TB networks. Lancet Infect Dis. 2023;23(4):e122-e137. doi:10.1016/S1473-3099(22)00875-1
- WHO operational handbook on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 2021 update. World Health Organization. Published July 7, 2021. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240030589treatment-for-all-people-suffering-from-drug-resistant-tb
- Global tuberculosis report 2022. World Health Organization. Published October 27, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240061729
- WHO consolidated guidelines on tuberculosis. Module 4: treatment – drug-resistant tuberculosis treatment, 2022 update. World Health Organization. Published December 15, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240063129
- Migliori GB, Tiberi S. Int J Tuberc Lung Dis. 2022 ;26(7):590-591. doi:10.5588/ijtld.22.0263.
- Lange C et al. Am J Respir Crit Care Med. 2022;205(10):1142-1144. doi:10.1164/rccm.202202-0393ED
- Esmail A et al. Am J Respir Crit Care Med. 2022;205(10):1214-1227. doi:10.1164/rccm.202107-1779OC
- WHO BPaLM Accelerator Platform: to support the call to action for implementation of the shorter and more effective treatment for all people suffering from drug-resistant TB. World Health Organization. Published May 9, 2023. Accessed June 26, 2023. https://www.who.int/news-room/events/detail/2023/05/09/default-calendar/who-bpalm-accelerator-platform–to-support-the-call-to-action-for-implementation-of-the-shorter-and-moreeffective-
- Trevisi L et al. Am J Respir Crit Care Med. 2023;207(11):1525-1532. doi:10.1164/rccm.202211-2125OC
- Domínguez J et al; TBnet and RESIST-TB networks. Lancet Infect Dis. 2023;23(4):e122-e137. doi:10.1016/S1473-3099(22)00875-1
- WHO operational handbook on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 2021 update. World Health Organization. Published July 7, 2021. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240030589treatment-for-all-people-suffering-from-drug-resistant-tb
Severity score predicts mortality in pulmonary tuberculosis
, based on data from approximately 400 individuals.
Although a mortality risk-prediction score could improve treatment for pulmonary tuberculosis patients, such a score has not been previously reported, wrote Takeshi Osawa, MD, of Fukujuji Hospital, Tokyo, and colleagues.
In a study published in the journal CHEST, the researchers used 252 patients from a previous perspective study of newly diagnosed pulmonary tuberculosis as the development cohort, and recruited 165 additional patients between March 2021 and September 2022.
The primary endpoint was all-cause in-hospital mortality. Based on data from the development group, the researchers found that age 65 years and older and age 80 years and older, hypoxemia, activities of daily living, bilateral pulmonary lesions, lymphocyte count of less than 720 microliters, serum albumin less than 2.86 mg/dL, C-reactive protein (CRP) 3.97 mg/dL or higher, and procalcitonin (PCT) 0.130 ng/mL or higher were predictors of all-cause in hospital mortality.
The researchers used this information to create the disease severity score, known as the AHL score. The AHL included three clinical parameters: activity in daily living (semi-dependent, 1 point; totally dependent, 2 points); hypoxemia (1 point) and lymphocytes (< 720 /mcL, 1 point).
The scoring systems for the three parameters were, respectively, 1 point for semi-dependent and 2 points totally dependent (for activity in daily living), 1 point for presence of hypoxemia, and 1 point for lymphocytes less than 720 per microliter. The researchers stratified the scores into levels of low, intermediate, and high risk, with scores of 0, 1-2, and 3-4, respectively.
All-cause in hospital mortality occurred in 39 (15.5%) and 17 (10.3%) of patients in the developmental and validation cohorts, respectively.
The AHL score effectively predicted mortality, dividing patients into three groups of 1.3% low-risk, 8.9% intermediate risk, and 39.3% high-risk in the validation cohort, with a Harrell’s c-statistic of 0.842.
The corresponding numbers for the development cohort were 0, 13.5%, and 55.8%, with a c-statistic of 0.902.
The findings were limited by several factors, including the lack of data from “smear-negative” patients who were treated as outpatients, and more research is needed to determine the applicability of the AHL score in an outpatient population, the researchers noted. Other limitations included the lack of data on long-term mortality in surviving patients who were discharged, and the reliance on assessments that can be performed only in clinical settings in developed countries, they said.
However, the results support the feasibility of the AHL score in clinical settings to accurately predict mortality in patients with pulmonary TB, and may help optimize treatments for this population, they concluded.
The study received no outside funding. All authors disclosed nonfinancial support in the form of measuring reagents from Fujifilm Wako Pure Chemical Corporation during the study but had no relevant financial conflicts to disclose.
, based on data from approximately 400 individuals.
Although a mortality risk-prediction score could improve treatment for pulmonary tuberculosis patients, such a score has not been previously reported, wrote Takeshi Osawa, MD, of Fukujuji Hospital, Tokyo, and colleagues.
In a study published in the journal CHEST, the researchers used 252 patients from a previous perspective study of newly diagnosed pulmonary tuberculosis as the development cohort, and recruited 165 additional patients between March 2021 and September 2022.
The primary endpoint was all-cause in-hospital mortality. Based on data from the development group, the researchers found that age 65 years and older and age 80 years and older, hypoxemia, activities of daily living, bilateral pulmonary lesions, lymphocyte count of less than 720 microliters, serum albumin less than 2.86 mg/dL, C-reactive protein (CRP) 3.97 mg/dL or higher, and procalcitonin (PCT) 0.130 ng/mL or higher were predictors of all-cause in hospital mortality.
The researchers used this information to create the disease severity score, known as the AHL score. The AHL included three clinical parameters: activity in daily living (semi-dependent, 1 point; totally dependent, 2 points); hypoxemia (1 point) and lymphocytes (< 720 /mcL, 1 point).
The scoring systems for the three parameters were, respectively, 1 point for semi-dependent and 2 points totally dependent (for activity in daily living), 1 point for presence of hypoxemia, and 1 point for lymphocytes less than 720 per microliter. The researchers stratified the scores into levels of low, intermediate, and high risk, with scores of 0, 1-2, and 3-4, respectively.
All-cause in hospital mortality occurred in 39 (15.5%) and 17 (10.3%) of patients in the developmental and validation cohorts, respectively.
The AHL score effectively predicted mortality, dividing patients into three groups of 1.3% low-risk, 8.9% intermediate risk, and 39.3% high-risk in the validation cohort, with a Harrell’s c-statistic of 0.842.
The corresponding numbers for the development cohort were 0, 13.5%, and 55.8%, with a c-statistic of 0.902.
The findings were limited by several factors, including the lack of data from “smear-negative” patients who were treated as outpatients, and more research is needed to determine the applicability of the AHL score in an outpatient population, the researchers noted. Other limitations included the lack of data on long-term mortality in surviving patients who were discharged, and the reliance on assessments that can be performed only in clinical settings in developed countries, they said.
However, the results support the feasibility of the AHL score in clinical settings to accurately predict mortality in patients with pulmonary TB, and may help optimize treatments for this population, they concluded.
The study received no outside funding. All authors disclosed nonfinancial support in the form of measuring reagents from Fujifilm Wako Pure Chemical Corporation during the study but had no relevant financial conflicts to disclose.
, based on data from approximately 400 individuals.
Although a mortality risk-prediction score could improve treatment for pulmonary tuberculosis patients, such a score has not been previously reported, wrote Takeshi Osawa, MD, of Fukujuji Hospital, Tokyo, and colleagues.
In a study published in the journal CHEST, the researchers used 252 patients from a previous perspective study of newly diagnosed pulmonary tuberculosis as the development cohort, and recruited 165 additional patients between March 2021 and September 2022.
The primary endpoint was all-cause in-hospital mortality. Based on data from the development group, the researchers found that age 65 years and older and age 80 years and older, hypoxemia, activities of daily living, bilateral pulmonary lesions, lymphocyte count of less than 720 microliters, serum albumin less than 2.86 mg/dL, C-reactive protein (CRP) 3.97 mg/dL or higher, and procalcitonin (PCT) 0.130 ng/mL or higher were predictors of all-cause in hospital mortality.
The researchers used this information to create the disease severity score, known as the AHL score. The AHL included three clinical parameters: activity in daily living (semi-dependent, 1 point; totally dependent, 2 points); hypoxemia (1 point) and lymphocytes (< 720 /mcL, 1 point).
The scoring systems for the three parameters were, respectively, 1 point for semi-dependent and 2 points totally dependent (for activity in daily living), 1 point for presence of hypoxemia, and 1 point for lymphocytes less than 720 per microliter. The researchers stratified the scores into levels of low, intermediate, and high risk, with scores of 0, 1-2, and 3-4, respectively.
All-cause in hospital mortality occurred in 39 (15.5%) and 17 (10.3%) of patients in the developmental and validation cohorts, respectively.
The AHL score effectively predicted mortality, dividing patients into three groups of 1.3% low-risk, 8.9% intermediate risk, and 39.3% high-risk in the validation cohort, with a Harrell’s c-statistic of 0.842.
The corresponding numbers for the development cohort were 0, 13.5%, and 55.8%, with a c-statistic of 0.902.
The findings were limited by several factors, including the lack of data from “smear-negative” patients who were treated as outpatients, and more research is needed to determine the applicability of the AHL score in an outpatient population, the researchers noted. Other limitations included the lack of data on long-term mortality in surviving patients who were discharged, and the reliance on assessments that can be performed only in clinical settings in developed countries, they said.
However, the results support the feasibility of the AHL score in clinical settings to accurately predict mortality in patients with pulmonary TB, and may help optimize treatments for this population, they concluded.
The study received no outside funding. All authors disclosed nonfinancial support in the form of measuring reagents from Fujifilm Wako Pure Chemical Corporation during the study but had no relevant financial conflicts to disclose.
FROM THE JOURNAL CHEST
Pulmonary aspergillosis predicts poor outcomes in critically ill flu patients
Critically ill influenza patients with associated pulmonary aspergillosis were more than twice as likely to die in intensive care than those without the added infection, based on data from a meta-analysis of more than 1,700 individuals.
Reports of influenza-associated pulmonary aspergillosis (IAPA) are rising in critically ill patients, but data on risk factors, clinical features, and outcomes are limited, Lawrence Y. Lu, MD, of The Prince Charles Hospital, Brisbane, Australia, and colleagues wrote. In addition, diagnosis of IAPA can be challenging, and many clinicians report low awareness of the condition.
In a study published in the journal Chest, the researchers reviewed data from 10 observational studies including 1,720 critically ill influenza patients aged 16 years and older; of these, 331 had IAPA, for a prevalence of 19.2%. The primary outcomes were all-cause mortality in the hospital and in the ICU. Secondary outcomes included ICU length of stay, hospital length of stay, and the need for supportive care (invasive and noninvasive mechanical ventilation, renal replacement therapy, pressor support, and extracorporeal membranous oxygenation).
Overall, mortality among flu patients in the ICU was significantly higher for those with IAPA than those without IAPA (45.0% vs. 23.8%, respectively), as was all-cause mortality (46.4% vs. 26.2%, respectively; odds ratio, 2.6 and P < .001 for both ICU and all-cause mortality).
Factors significantly associated with an increased risk for IAPA included organ transplant (OR, 4.8), hematogenous malignancy (OR, 2.5), being immunocompromised in some way (OR, 2.2), and prolonged corticosteroid use prior to hospital admission (OR, 2.4).
IAPA also was associated with more severe disease, a higher rate of complications, longer ICU stays, and a greater need for organ supports, the researchers noted. Clinical features not significantly more common in patients with IAPA included fever, hemoptysis, and acute respiratory distress syndrome.
The findings were limited by several factors including the retrospective design of the included studies and inability to control for all potential confounders, the researchers noted. Other limitations included the variations in study design, variability of practice patterns across locations, and inclusion of data mainly from countries of high socioeconomic status.
“Given the apparent waning of the COVID-19 pandemic and re-emergence of influenza, our analysis also revealed other gaps in the current literature, including the need to validate newer diagnostic methods and to develop a system to measure severity of IAPA,” the researchers added.
However, the current study results reflect IAPA prevalence from previous studies, and support the need to have a lower threshold for IAPA testing and initiation of antifungal treatment, even with limited data for clinical guidance, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Critically ill influenza patients with associated pulmonary aspergillosis were more than twice as likely to die in intensive care than those without the added infection, based on data from a meta-analysis of more than 1,700 individuals.
Reports of influenza-associated pulmonary aspergillosis (IAPA) are rising in critically ill patients, but data on risk factors, clinical features, and outcomes are limited, Lawrence Y. Lu, MD, of The Prince Charles Hospital, Brisbane, Australia, and colleagues wrote. In addition, diagnosis of IAPA can be challenging, and many clinicians report low awareness of the condition.
In a study published in the journal Chest, the researchers reviewed data from 10 observational studies including 1,720 critically ill influenza patients aged 16 years and older; of these, 331 had IAPA, for a prevalence of 19.2%. The primary outcomes were all-cause mortality in the hospital and in the ICU. Secondary outcomes included ICU length of stay, hospital length of stay, and the need for supportive care (invasive and noninvasive mechanical ventilation, renal replacement therapy, pressor support, and extracorporeal membranous oxygenation).
Overall, mortality among flu patients in the ICU was significantly higher for those with IAPA than those without IAPA (45.0% vs. 23.8%, respectively), as was all-cause mortality (46.4% vs. 26.2%, respectively; odds ratio, 2.6 and P < .001 for both ICU and all-cause mortality).
Factors significantly associated with an increased risk for IAPA included organ transplant (OR, 4.8), hematogenous malignancy (OR, 2.5), being immunocompromised in some way (OR, 2.2), and prolonged corticosteroid use prior to hospital admission (OR, 2.4).
IAPA also was associated with more severe disease, a higher rate of complications, longer ICU stays, and a greater need for organ supports, the researchers noted. Clinical features not significantly more common in patients with IAPA included fever, hemoptysis, and acute respiratory distress syndrome.
The findings were limited by several factors including the retrospective design of the included studies and inability to control for all potential confounders, the researchers noted. Other limitations included the variations in study design, variability of practice patterns across locations, and inclusion of data mainly from countries of high socioeconomic status.
“Given the apparent waning of the COVID-19 pandemic and re-emergence of influenza, our analysis also revealed other gaps in the current literature, including the need to validate newer diagnostic methods and to develop a system to measure severity of IAPA,” the researchers added.
However, the current study results reflect IAPA prevalence from previous studies, and support the need to have a lower threshold for IAPA testing and initiation of antifungal treatment, even with limited data for clinical guidance, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Critically ill influenza patients with associated pulmonary aspergillosis were more than twice as likely to die in intensive care than those without the added infection, based on data from a meta-analysis of more than 1,700 individuals.
Reports of influenza-associated pulmonary aspergillosis (IAPA) are rising in critically ill patients, but data on risk factors, clinical features, and outcomes are limited, Lawrence Y. Lu, MD, of The Prince Charles Hospital, Brisbane, Australia, and colleagues wrote. In addition, diagnosis of IAPA can be challenging, and many clinicians report low awareness of the condition.
In a study published in the journal Chest, the researchers reviewed data from 10 observational studies including 1,720 critically ill influenza patients aged 16 years and older; of these, 331 had IAPA, for a prevalence of 19.2%. The primary outcomes were all-cause mortality in the hospital and in the ICU. Secondary outcomes included ICU length of stay, hospital length of stay, and the need for supportive care (invasive and noninvasive mechanical ventilation, renal replacement therapy, pressor support, and extracorporeal membranous oxygenation).
Overall, mortality among flu patients in the ICU was significantly higher for those with IAPA than those without IAPA (45.0% vs. 23.8%, respectively), as was all-cause mortality (46.4% vs. 26.2%, respectively; odds ratio, 2.6 and P < .001 for both ICU and all-cause mortality).
Factors significantly associated with an increased risk for IAPA included organ transplant (OR, 4.8), hematogenous malignancy (OR, 2.5), being immunocompromised in some way (OR, 2.2), and prolonged corticosteroid use prior to hospital admission (OR, 2.4).
IAPA also was associated with more severe disease, a higher rate of complications, longer ICU stays, and a greater need for organ supports, the researchers noted. Clinical features not significantly more common in patients with IAPA included fever, hemoptysis, and acute respiratory distress syndrome.
The findings were limited by several factors including the retrospective design of the included studies and inability to control for all potential confounders, the researchers noted. Other limitations included the variations in study design, variability of practice patterns across locations, and inclusion of data mainly from countries of high socioeconomic status.
“Given the apparent waning of the COVID-19 pandemic and re-emergence of influenza, our analysis also revealed other gaps in the current literature, including the need to validate newer diagnostic methods and to develop a system to measure severity of IAPA,” the researchers added.
However, the current study results reflect IAPA prevalence from previous studies, and support the need to have a lower threshold for IAPA testing and initiation of antifungal treatment, even with limited data for clinical guidance, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL CHEST
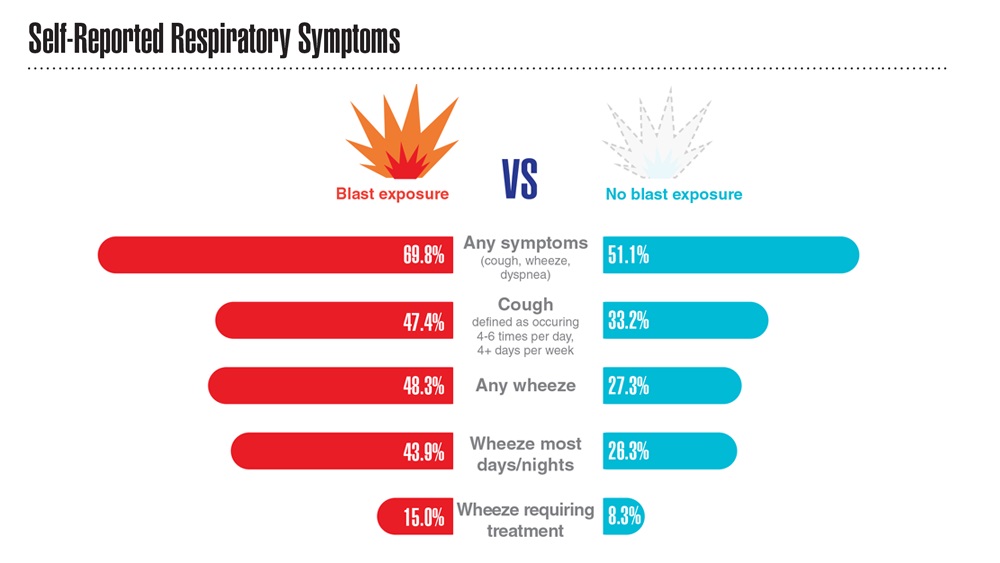
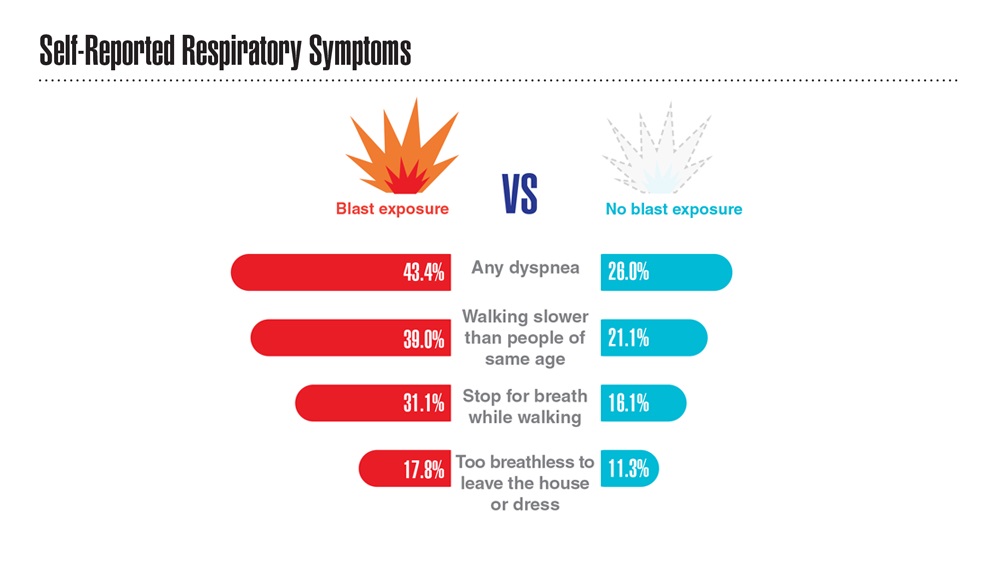
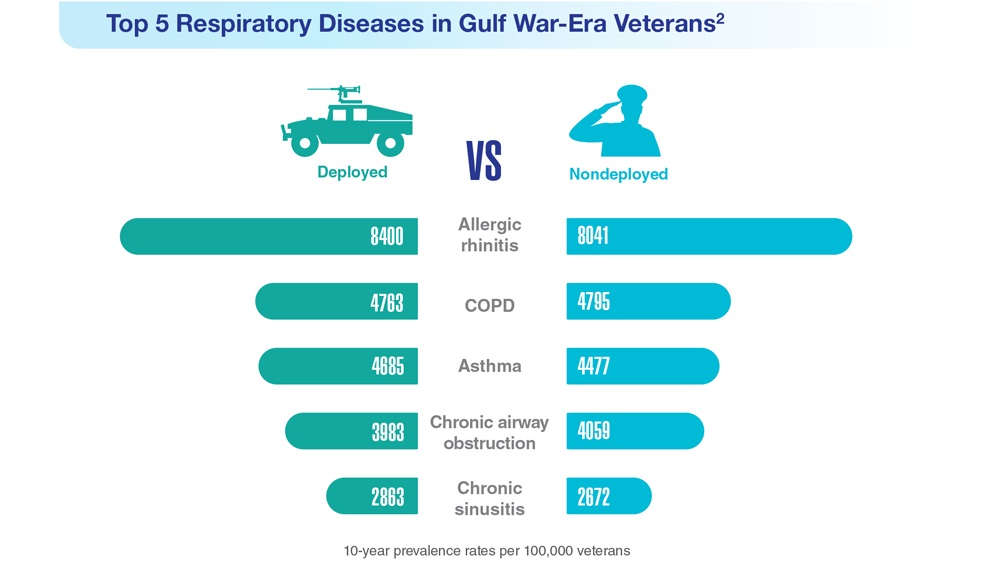
Data Trends 2023: Respiratory Illnesses
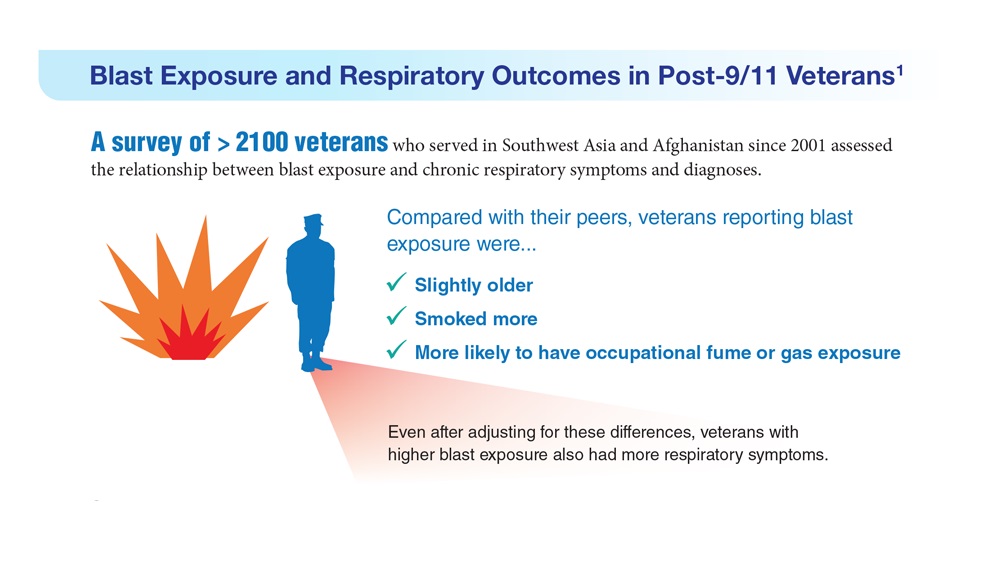
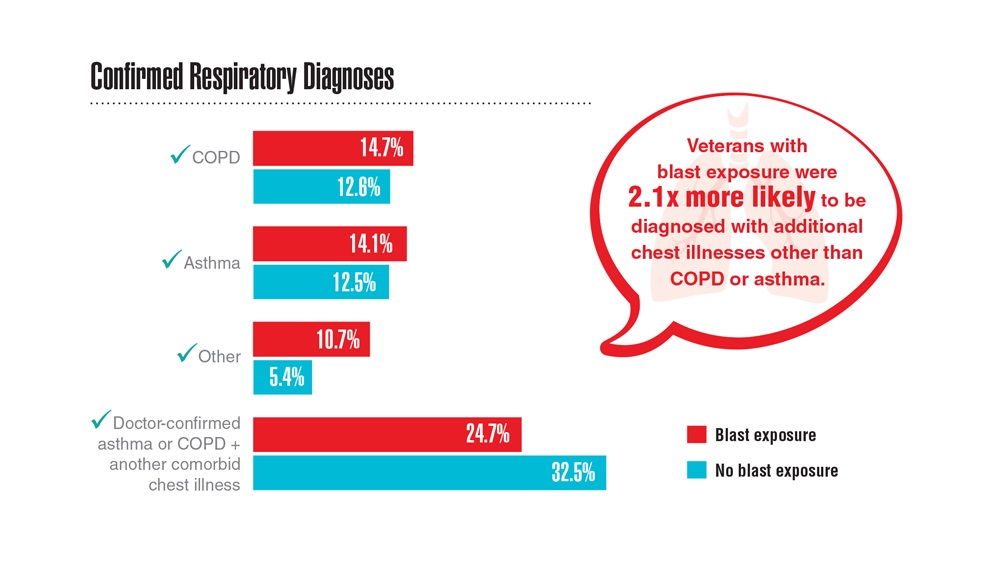
- Hines SE et al. Respir Med. 2022;202:106963. doi:10.1016/j.rmed.2022.106963
- Dursa EK et al. Am J Ind Med. 2020;63(11):980-987. doi:10.1002/ajim.23172
- Bamonti PM et al. Int J Chron Obstruct Pulmon Dis. 2022;17:1269-1283. doi:10.2147/COPD.S339323
- Hines SE et al. Respir Med. 2022;202:106963. doi:10.1016/j.rmed.2022.106963
- Dursa EK et al. Am J Ind Med. 2020;63(11):980-987. doi:10.1002/ajim.23172
- Bamonti PM et al. Int J Chron Obstruct Pulmon Dis. 2022;17:1269-1283. doi:10.2147/COPD.S339323
- Hines SE et al. Respir Med. 2022;202:106963. doi:10.1016/j.rmed.2022.106963
- Dursa EK et al. Am J Ind Med. 2020;63(11):980-987. doi:10.1002/ajim.23172
- Bamonti PM et al. Int J Chron Obstruct Pulmon Dis. 2022;17:1269-1283. doi:10.2147/COPD.S339323
Federal Health Care Data Trends 2023
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner, highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Limb Loss and Prostheses
- Neurology
- Cardiology
- Mental Health
- Diabetes
- Rheumatoid Arthritis
- Respiratory illnesses
- Women's Health
- HPV and Related Cancers
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner, highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Limb Loss and Prostheses
- Neurology
- Cardiology
- Mental Health
- Diabetes
- Rheumatoid Arthritis
- Respiratory illnesses
- Women's Health
- HPV and Related Cancers
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner, highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Limb Loss and Prostheses
- Neurology
- Cardiology
- Mental Health
- Diabetes
- Rheumatoid Arthritis
- Respiratory illnesses
- Women's Health
- HPV and Related Cancers
Three antibiotic regimens show similar effectiveness for CAP
Adults with nonsevere community-acquired pneumonia (CAP) responded nearly equally to three first-line and alternative antibiotic regimens, based on data from more than 23,000 individuals.
Current recommendations for the treatment of CAP vary across guidelines, wrote Anthony D. Bai, MD, of Queen’s University, Kingston, Ont., and colleagues. However, most guidelines were based on studies that were not powered to examine the effect of treatments on mortality, they said.
“Large observational studies could fill this gap by comparing multiple treatment arms, including patients not well represented in trials, and having a large sample size powered to detect a difference in mortality,” they noted.
In a study published in Chest, the researchers reviewed data from 23,512 consecutive patients admitted to 19 hospitals in Canada for CAP between 2015 and 2021. Patients were treated with one of four initial antibiotic regimens: beta-lactam plus macrolide (BL+M), beta-lactam alone (BL), respiratory fluoroquinolone (FQ), or beta-lactam plus doxycycline (BL+D). Of these, BL+M is generally considered the first-line regimen, the researchers noted.
Patients were divided into four groups according to their initial antibiotic treatment within 48 hours of admission; 9,340 patients received BL+M, 9,146 received BL, 4,510 received FQ, and 516 received BL+D. The duration of any antibiotic that was active against CAP was at least 4 days, or until hospital discharge or death.
The primary outcome was all-cause in-hospital mortality, which was 7.5%, 9.7%, 6.7%, and 6.0% for patients in each of the four treatment groups, respectively. Relative to the first-line therapy of BL+M, the adjusted risk differences for BL, FQ, and BL+D were 1.5%, –0.9%, and –1.9%, respectively.
The adjusted in-hospital mortality was not significantly different between BL+M and either FQ or BL+D, but the difference of 1.5% seen with BL alone suggested a “small but clinically important difference,” the researchers noted.
Key secondary outcomes were the length of hospital stay and being discharged alive. The median length of stay was 4.6 days for BL+M, 5.2 days for BL, 4.6 days for FQ, and 6.0 days for BL+D. Patients treated with BL also had a longer time to hospital discharge, which suggests that BL may not be as effective as the other regimens, the researchers said. In addition, patients in the BL group had a subdistribution hazard ratio of 0.90 for being discharged alive, compared with the BL+M group after adjustment with propensity scores and overlap weighting.
Overall, the results support dropping BL as a first-line regimen in the current ATS/IDSA guidelines, and support the recommendation of BL+M, FQ, and BL+D as similarly effective options as listed in other guidelines, applied according to other patient characteristics. For example, “Doxycycline may be preferred over a macrolide in many cases such as macrolide allergy, prolonged QT, or high [Clostridioides] difficile risk,” the researchers said.
The findings were limited by several factors including the lack of follow-up data after hospital discharge.
However, the results were strengthened by the large sample size and use of a comprehensive database that allowed adjustment for many variables, as well as the availability of complete follow-up data for the time spent in the hospital. Based on this study, clinicians may choose a respiratory fluoroquinolone, a beta-lactam plus macrolide, or a beta-lactam plus doxycycline for equally effective antibiotic treatment of CAP, based on the best fit for each individual patient, the researchers concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Adults with nonsevere community-acquired pneumonia (CAP) responded nearly equally to three first-line and alternative antibiotic regimens, based on data from more than 23,000 individuals.
Current recommendations for the treatment of CAP vary across guidelines, wrote Anthony D. Bai, MD, of Queen’s University, Kingston, Ont., and colleagues. However, most guidelines were based on studies that were not powered to examine the effect of treatments on mortality, they said.
“Large observational studies could fill this gap by comparing multiple treatment arms, including patients not well represented in trials, and having a large sample size powered to detect a difference in mortality,” they noted.
In a study published in Chest, the researchers reviewed data from 23,512 consecutive patients admitted to 19 hospitals in Canada for CAP between 2015 and 2021. Patients were treated with one of four initial antibiotic regimens: beta-lactam plus macrolide (BL+M), beta-lactam alone (BL), respiratory fluoroquinolone (FQ), or beta-lactam plus doxycycline (BL+D). Of these, BL+M is generally considered the first-line regimen, the researchers noted.
Patients were divided into four groups according to their initial antibiotic treatment within 48 hours of admission; 9,340 patients received BL+M, 9,146 received BL, 4,510 received FQ, and 516 received BL+D. The duration of any antibiotic that was active against CAP was at least 4 days, or until hospital discharge or death.
The primary outcome was all-cause in-hospital mortality, which was 7.5%, 9.7%, 6.7%, and 6.0% for patients in each of the four treatment groups, respectively. Relative to the first-line therapy of BL+M, the adjusted risk differences for BL, FQ, and BL+D were 1.5%, –0.9%, and –1.9%, respectively.
The adjusted in-hospital mortality was not significantly different between BL+M and either FQ or BL+D, but the difference of 1.5% seen with BL alone suggested a “small but clinically important difference,” the researchers noted.
Key secondary outcomes were the length of hospital stay and being discharged alive. The median length of stay was 4.6 days for BL+M, 5.2 days for BL, 4.6 days for FQ, and 6.0 days for BL+D. Patients treated with BL also had a longer time to hospital discharge, which suggests that BL may not be as effective as the other regimens, the researchers said. In addition, patients in the BL group had a subdistribution hazard ratio of 0.90 for being discharged alive, compared with the BL+M group after adjustment with propensity scores and overlap weighting.
Overall, the results support dropping BL as a first-line regimen in the current ATS/IDSA guidelines, and support the recommendation of BL+M, FQ, and BL+D as similarly effective options as listed in other guidelines, applied according to other patient characteristics. For example, “Doxycycline may be preferred over a macrolide in many cases such as macrolide allergy, prolonged QT, or high [Clostridioides] difficile risk,” the researchers said.
The findings were limited by several factors including the lack of follow-up data after hospital discharge.
However, the results were strengthened by the large sample size and use of a comprehensive database that allowed adjustment for many variables, as well as the availability of complete follow-up data for the time spent in the hospital. Based on this study, clinicians may choose a respiratory fluoroquinolone, a beta-lactam plus macrolide, or a beta-lactam plus doxycycline for equally effective antibiotic treatment of CAP, based on the best fit for each individual patient, the researchers concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Adults with nonsevere community-acquired pneumonia (CAP) responded nearly equally to three first-line and alternative antibiotic regimens, based on data from more than 23,000 individuals.
Current recommendations for the treatment of CAP vary across guidelines, wrote Anthony D. Bai, MD, of Queen’s University, Kingston, Ont., and colleagues. However, most guidelines were based on studies that were not powered to examine the effect of treatments on mortality, they said.
“Large observational studies could fill this gap by comparing multiple treatment arms, including patients not well represented in trials, and having a large sample size powered to detect a difference in mortality,” they noted.
In a study published in Chest, the researchers reviewed data from 23,512 consecutive patients admitted to 19 hospitals in Canada for CAP between 2015 and 2021. Patients were treated with one of four initial antibiotic regimens: beta-lactam plus macrolide (BL+M), beta-lactam alone (BL), respiratory fluoroquinolone (FQ), or beta-lactam plus doxycycline (BL+D). Of these, BL+M is generally considered the first-line regimen, the researchers noted.
Patients were divided into four groups according to their initial antibiotic treatment within 48 hours of admission; 9,340 patients received BL+M, 9,146 received BL, 4,510 received FQ, and 516 received BL+D. The duration of any antibiotic that was active against CAP was at least 4 days, or until hospital discharge or death.
The primary outcome was all-cause in-hospital mortality, which was 7.5%, 9.7%, 6.7%, and 6.0% for patients in each of the four treatment groups, respectively. Relative to the first-line therapy of BL+M, the adjusted risk differences for BL, FQ, and BL+D were 1.5%, –0.9%, and –1.9%, respectively.
The adjusted in-hospital mortality was not significantly different between BL+M and either FQ or BL+D, but the difference of 1.5% seen with BL alone suggested a “small but clinically important difference,” the researchers noted.
Key secondary outcomes were the length of hospital stay and being discharged alive. The median length of stay was 4.6 days for BL+M, 5.2 days for BL, 4.6 days for FQ, and 6.0 days for BL+D. Patients treated with BL also had a longer time to hospital discharge, which suggests that BL may not be as effective as the other regimens, the researchers said. In addition, patients in the BL group had a subdistribution hazard ratio of 0.90 for being discharged alive, compared with the BL+M group after adjustment with propensity scores and overlap weighting.
Overall, the results support dropping BL as a first-line regimen in the current ATS/IDSA guidelines, and support the recommendation of BL+M, FQ, and BL+D as similarly effective options as listed in other guidelines, applied according to other patient characteristics. For example, “Doxycycline may be preferred over a macrolide in many cases such as macrolide allergy, prolonged QT, or high [Clostridioides] difficile risk,” the researchers said.
The findings were limited by several factors including the lack of follow-up data after hospital discharge.
However, the results were strengthened by the large sample size and use of a comprehensive database that allowed adjustment for many variables, as well as the availability of complete follow-up data for the time spent in the hospital. Based on this study, clinicians may choose a respiratory fluoroquinolone, a beta-lactam plus macrolide, or a beta-lactam plus doxycycline for equally effective antibiotic treatment of CAP, based on the best fit for each individual patient, the researchers concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM CHEST