User login
Mouse model replicates aggressive AML subtype

Researchers have developed a mouse model of an aggressive type of acute myeloid leukemia (AML) that, they believe, accurately replicates the human form of the disease.
The model replicates AML with co-occurring mutations in FLT3 and DNMT3A.
The researchers said they found that mice with Flt3-ITD and inducible deletion of Dnmt3a developed a rapidly lethal, completely penetrant, and transplantable AML of normal karyotype.
The team described this work in Cancer Discovery.
“Our goal was to create a model that was faithful to the human form of the disease that can be used for preclinical testing of potential cures,” said study author H. Leighton Grimes, PhD, of Cincinnati Children’s Hospital Medical Center in Ohio.
“Previous models were slow, difficult to analyze, and did not accurately represent the human disease. This model is rapid, fully penetrant, and completely spontaneous. We hope that it will open the way for other researchers to join us in attacking this particularly lethal AML subtype.”
Dr Grimes and his colleagues said they were able to look at the disease in a new way with the help of a powerful new core facility utilizing analytical tools related to single-cell RNA sequencing. The team used complementary single-cell analyses to identify the core leukemia-causing stem cells of the tumor.
“Before, researchers were comparing the gene expression patterns of one AML subtype to either normal cells or other AML subtypes,” said study author Sara Meyer, PhD, a fellow in the Grimes lab.
“That approach made it difficult to tease out the specific impact of Dnmt3a mutation. Instead, we isolated the variables and studied only human and murine AML with Flt3 mutation. Comparing Flt3-mutant AML with and without Dnmt3a mutation allowed us to more finely identify those patterns that were specific to the Dnmt3a mutation.”
With that more detailed understanding, the researchers gained new insights into the contributions of the Dnmt3a mutation to the disease.
First, their work confirms suspicions that low-level Dnmt3a activity is cancer-causing. Moreover, they discovered that reduced Dnmt3a function allows genes that are normally expressed only at early development stages of blood cell formation to continue expression at later stages, leading to the development of AML.
The researchers also found that, in mouse tumor cells, rescuing expression of Dnmt3a reversed the leukemia phenotypes and gene expression. But they said more research is warranted to determine if rescuing normal levels of DNMT3A function is a viable method for treating human AML.
The team also identified several potential treatment targets that are unique to this type of AML. In future studies, they plan to proceed with testing potential therapies. ![]()

Researchers have developed a mouse model of an aggressive type of acute myeloid leukemia (AML) that, they believe, accurately replicates the human form of the disease.
The model replicates AML with co-occurring mutations in FLT3 and DNMT3A.
The researchers said they found that mice with Flt3-ITD and inducible deletion of Dnmt3a developed a rapidly lethal, completely penetrant, and transplantable AML of normal karyotype.
The team described this work in Cancer Discovery.
“Our goal was to create a model that was faithful to the human form of the disease that can be used for preclinical testing of potential cures,” said study author H. Leighton Grimes, PhD, of Cincinnati Children’s Hospital Medical Center in Ohio.
“Previous models were slow, difficult to analyze, and did not accurately represent the human disease. This model is rapid, fully penetrant, and completely spontaneous. We hope that it will open the way for other researchers to join us in attacking this particularly lethal AML subtype.”
Dr Grimes and his colleagues said they were able to look at the disease in a new way with the help of a powerful new core facility utilizing analytical tools related to single-cell RNA sequencing. The team used complementary single-cell analyses to identify the core leukemia-causing stem cells of the tumor.
“Before, researchers were comparing the gene expression patterns of one AML subtype to either normal cells or other AML subtypes,” said study author Sara Meyer, PhD, a fellow in the Grimes lab.
“That approach made it difficult to tease out the specific impact of Dnmt3a mutation. Instead, we isolated the variables and studied only human and murine AML with Flt3 mutation. Comparing Flt3-mutant AML with and without Dnmt3a mutation allowed us to more finely identify those patterns that were specific to the Dnmt3a mutation.”
With that more detailed understanding, the researchers gained new insights into the contributions of the Dnmt3a mutation to the disease.
First, their work confirms suspicions that low-level Dnmt3a activity is cancer-causing. Moreover, they discovered that reduced Dnmt3a function allows genes that are normally expressed only at early development stages of blood cell formation to continue expression at later stages, leading to the development of AML.
The researchers also found that, in mouse tumor cells, rescuing expression of Dnmt3a reversed the leukemia phenotypes and gene expression. But they said more research is warranted to determine if rescuing normal levels of DNMT3A function is a viable method for treating human AML.
The team also identified several potential treatment targets that are unique to this type of AML. In future studies, they plan to proceed with testing potential therapies. ![]()

Researchers have developed a mouse model of an aggressive type of acute myeloid leukemia (AML) that, they believe, accurately replicates the human form of the disease.
The model replicates AML with co-occurring mutations in FLT3 and DNMT3A.
The researchers said they found that mice with Flt3-ITD and inducible deletion of Dnmt3a developed a rapidly lethal, completely penetrant, and transplantable AML of normal karyotype.
The team described this work in Cancer Discovery.
“Our goal was to create a model that was faithful to the human form of the disease that can be used for preclinical testing of potential cures,” said study author H. Leighton Grimes, PhD, of Cincinnati Children’s Hospital Medical Center in Ohio.
“Previous models were slow, difficult to analyze, and did not accurately represent the human disease. This model is rapid, fully penetrant, and completely spontaneous. We hope that it will open the way for other researchers to join us in attacking this particularly lethal AML subtype.”
Dr Grimes and his colleagues said they were able to look at the disease in a new way with the help of a powerful new core facility utilizing analytical tools related to single-cell RNA sequencing. The team used complementary single-cell analyses to identify the core leukemia-causing stem cells of the tumor.
“Before, researchers were comparing the gene expression patterns of one AML subtype to either normal cells or other AML subtypes,” said study author Sara Meyer, PhD, a fellow in the Grimes lab.
“That approach made it difficult to tease out the specific impact of Dnmt3a mutation. Instead, we isolated the variables and studied only human and murine AML with Flt3 mutation. Comparing Flt3-mutant AML with and without Dnmt3a mutation allowed us to more finely identify those patterns that were specific to the Dnmt3a mutation.”
With that more detailed understanding, the researchers gained new insights into the contributions of the Dnmt3a mutation to the disease.
First, their work confirms suspicions that low-level Dnmt3a activity is cancer-causing. Moreover, they discovered that reduced Dnmt3a function allows genes that are normally expressed only at early development stages of blood cell formation to continue expression at later stages, leading to the development of AML.
The researchers also found that, in mouse tumor cells, rescuing expression of Dnmt3a reversed the leukemia phenotypes and gene expression. But they said more research is warranted to determine if rescuing normal levels of DNMT3A function is a viable method for treating human AML.
The team also identified several potential treatment targets that are unique to this type of AML. In future studies, they plan to proceed with testing potential therapies. ![]()
FDA OKs use of test to screen blood donations for Zika virus
The US Food and Drug Administration (FDA) is allowing the use of an investigational test to screen blood donations for Zika virus.
The test, known as the cobas® Zika test, has not been granted FDA clearance or approval, but it may be used under an investigational new drug application protocol for screening donated blood in areas with active, mosquito-borne transmission of Zika virus.
This means the test can be used by US blood screening laboratories, but the laboratories will need to be enrolled in and contracted into a clinical trial for the test, as specified and agreed with the FDA’s Center for Biologics Evaluation and Research.
By authorizing use of the cobas® Zika test, the FDA is allowing blood establishments in Puerto Rico—a US territory with local, mosquito-borne transmission of the Zika virus—to resume collecting donations of whole blood and blood components.
“The availability of an investigational test to screen donated blood for Zika virus is an important step forward in maintaining the safety of the nation’s blood supply, especially for those US territories already experiencing active transmission,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research.
“In the future, should Zika virus transmission occur in other areas, blood collection establishments will be able to continue to collect blood and use the investigational screening test, minimizing disruption to the blood supply.”
About the test
The cobas® Zika test is a qualitative in vitro nucleic acid screening test for the direct detection of Zika virus RNA in plasma specimens from individual human
blood donors.
The test is based on fully automated sample preparation (nucleic acid extraction and purification), followed by PCR amplification and detection.
The cobas® Zika test is manufactured by Roche and is intended for use with Roche’s cobas® 6800/8800 Systems.
The cobas® 6800/8800 Systems consist of the sample supply module, the transfer module, the processing module, and the analytic module. Automated data management is performed by the cobas® 6800/8800 software, which assigns test results for all tests as non-reactive, reactive, or invalid.
“The cobas® Zika test has been specifically designed utilizing the generic cobas® omni Utility Channel on the cobas® 6800/8800 Systems,” said Roland Diggelmann, chief operating officer of Roche Diagnostics.
“These fully automated, high-volume systems provide solutions for blood services to detect the virus and ensure that potentially infected blood units are not made available for transfusion.”
Test availability
Initially, the cobas® Zika test will be deployed to screen blood donations collected locally in Puerto Rico. It is expected that this testing will enable the
reinstatement of the blood services in Puerto Rico and reduce the reliance of blood importation from other areas in the US.
The second stage of deployment for the cobas® Zika test will be to prepare for screening of blood donations collected by blood services in the southern US.
In addition, Roche said it is working with regulators around the world to determine the path forward to implement the cobas® Zika test for blood screening.
Implications for Puerto Rico
On February 16, the FDA issued a guidance for US blood establishments to reduce the risk of transfusion-transmitted Zika virus. In the guidance, the FDA recommended that areas with active transmission of Zika virus obtain whole blood and blood components from areas without active transmission of the virus.
As a result, local blood collection in Puerto Rico was suspended. On March 7, the Department of Health and Human Services announced that it arranged for shipments of blood products from the continental US to Puerto Rico.
The FDA guidance also states that establishments in areas with active Zika transmission may collect locally if a licensed or investigational test for screening donated blood is available.
Once screening of blood donations for Zika virus using the cobas® Zika test begins, blood establishments in Puerto Rico may resume collecting donations of whole blood and blood components. However, the FDA’s recommendations for Zika blood donor deferrals remain in place. ![]()
The US Food and Drug Administration (FDA) is allowing the use of an investigational test to screen blood donations for Zika virus.
The test, known as the cobas® Zika test, has not been granted FDA clearance or approval, but it may be used under an investigational new drug application protocol for screening donated blood in areas with active, mosquito-borne transmission of Zika virus.
This means the test can be used by US blood screening laboratories, but the laboratories will need to be enrolled in and contracted into a clinical trial for the test, as specified and agreed with the FDA’s Center for Biologics Evaluation and Research.
By authorizing use of the cobas® Zika test, the FDA is allowing blood establishments in Puerto Rico—a US territory with local, mosquito-borne transmission of the Zika virus—to resume collecting donations of whole blood and blood components.
“The availability of an investigational test to screen donated blood for Zika virus is an important step forward in maintaining the safety of the nation’s blood supply, especially for those US territories already experiencing active transmission,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research.
“In the future, should Zika virus transmission occur in other areas, blood collection establishments will be able to continue to collect blood and use the investigational screening test, minimizing disruption to the blood supply.”
About the test
The cobas® Zika test is a qualitative in vitro nucleic acid screening test for the direct detection of Zika virus RNA in plasma specimens from individual human
blood donors.
The test is based on fully automated sample preparation (nucleic acid extraction and purification), followed by PCR amplification and detection.
The cobas® Zika test is manufactured by Roche and is intended for use with Roche’s cobas® 6800/8800 Systems.
The cobas® 6800/8800 Systems consist of the sample supply module, the transfer module, the processing module, and the analytic module. Automated data management is performed by the cobas® 6800/8800 software, which assigns test results for all tests as non-reactive, reactive, or invalid.
“The cobas® Zika test has been specifically designed utilizing the generic cobas® omni Utility Channel on the cobas® 6800/8800 Systems,” said Roland Diggelmann, chief operating officer of Roche Diagnostics.
“These fully automated, high-volume systems provide solutions for blood services to detect the virus and ensure that potentially infected blood units are not made available for transfusion.”
Test availability
Initially, the cobas® Zika test will be deployed to screen blood donations collected locally in Puerto Rico. It is expected that this testing will enable the
reinstatement of the blood services in Puerto Rico and reduce the reliance of blood importation from other areas in the US.
The second stage of deployment for the cobas® Zika test will be to prepare for screening of blood donations collected by blood services in the southern US.
In addition, Roche said it is working with regulators around the world to determine the path forward to implement the cobas® Zika test for blood screening.
Implications for Puerto Rico
On February 16, the FDA issued a guidance for US blood establishments to reduce the risk of transfusion-transmitted Zika virus. In the guidance, the FDA recommended that areas with active transmission of Zika virus obtain whole blood and blood components from areas without active transmission of the virus.
As a result, local blood collection in Puerto Rico was suspended. On March 7, the Department of Health and Human Services announced that it arranged for shipments of blood products from the continental US to Puerto Rico.
The FDA guidance also states that establishments in areas with active Zika transmission may collect locally if a licensed or investigational test for screening donated blood is available.
Once screening of blood donations for Zika virus using the cobas® Zika test begins, blood establishments in Puerto Rico may resume collecting donations of whole blood and blood components. However, the FDA’s recommendations for Zika blood donor deferrals remain in place. ![]()
The US Food and Drug Administration (FDA) is allowing the use of an investigational test to screen blood donations for Zika virus.
The test, known as the cobas® Zika test, has not been granted FDA clearance or approval, but it may be used under an investigational new drug application protocol for screening donated blood in areas with active, mosquito-borne transmission of Zika virus.
This means the test can be used by US blood screening laboratories, but the laboratories will need to be enrolled in and contracted into a clinical trial for the test, as specified and agreed with the FDA’s Center for Biologics Evaluation and Research.
By authorizing use of the cobas® Zika test, the FDA is allowing blood establishments in Puerto Rico—a US territory with local, mosquito-borne transmission of the Zika virus—to resume collecting donations of whole blood and blood components.
“The availability of an investigational test to screen donated blood for Zika virus is an important step forward in maintaining the safety of the nation’s blood supply, especially for those US territories already experiencing active transmission,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research.
“In the future, should Zika virus transmission occur in other areas, blood collection establishments will be able to continue to collect blood and use the investigational screening test, minimizing disruption to the blood supply.”
About the test
The cobas® Zika test is a qualitative in vitro nucleic acid screening test for the direct detection of Zika virus RNA in plasma specimens from individual human
blood donors.
The test is based on fully automated sample preparation (nucleic acid extraction and purification), followed by PCR amplification and detection.
The cobas® Zika test is manufactured by Roche and is intended for use with Roche’s cobas® 6800/8800 Systems.
The cobas® 6800/8800 Systems consist of the sample supply module, the transfer module, the processing module, and the analytic module. Automated data management is performed by the cobas® 6800/8800 software, which assigns test results for all tests as non-reactive, reactive, or invalid.
“The cobas® Zika test has been specifically designed utilizing the generic cobas® omni Utility Channel on the cobas® 6800/8800 Systems,” said Roland Diggelmann, chief operating officer of Roche Diagnostics.
“These fully automated, high-volume systems provide solutions for blood services to detect the virus and ensure that potentially infected blood units are not made available for transfusion.”
Test availability
Initially, the cobas® Zika test will be deployed to screen blood donations collected locally in Puerto Rico. It is expected that this testing will enable the
reinstatement of the blood services in Puerto Rico and reduce the reliance of blood importation from other areas in the US.
The second stage of deployment for the cobas® Zika test will be to prepare for screening of blood donations collected by blood services in the southern US.
In addition, Roche said it is working with regulators around the world to determine the path forward to implement the cobas® Zika test for blood screening.
Implications for Puerto Rico
On February 16, the FDA issued a guidance for US blood establishments to reduce the risk of transfusion-transmitted Zika virus. In the guidance, the FDA recommended that areas with active transmission of Zika virus obtain whole blood and blood components from areas without active transmission of the virus.
As a result, local blood collection in Puerto Rico was suspended. On March 7, the Department of Health and Human Services announced that it arranged for shipments of blood products from the continental US to Puerto Rico.
The FDA guidance also states that establishments in areas with active Zika transmission may collect locally if a licensed or investigational test for screening donated blood is available.
Once screening of blood donations for Zika virus using the cobas® Zika test begins, blood establishments in Puerto Rico may resume collecting donations of whole blood and blood components. However, the FDA’s recommendations for Zika blood donor deferrals remain in place. ![]()
Tool predicts risks of DAPT with ‘modest accuracy’
Researchers believe a new tool could help physicians predict the risks and benefits of extended dual antiplatelet therapy (DAPT) in patients who have undergone percutaneous coronary intervention (PCI).
The team said the tool, known as the DAPT Score, exhibited “modest accuracy” for determining which patients were at high risk for late ischemic events and would therefore benefit most from longer-term DAPT therapy.
The DAPT Score also proved somewhat accurate for identifying patients who were at high risk of late bleeding events and might be harmed by continuing DAPT for more than a year after PCI.
Still, the researchers said the scoring system requires further validation and prospective evaluation to assess its potential effects on patient care.
Robert W. Yeh, MD, of Beth Israel Deaconess Medical Center in Boston, Massachusetts, and his colleagues reported these results in JAMA.
“Dual antiplatelet therapy is standard for patients following coronary stent procedures, but we haven’t had good tools to help us determine how long we should be treating individual patients,” Dr Yeh said.
So he and his colleagues set out to identify factors that would predict whether the expected benefit of reduced ischemia would outweigh the expected increase in bleeding associated with continuing DAPT for more than a year after PCI.
The team used 11,648 patients treated on the DAPT study to create the DAPT Score. Patients in this trial had a drug-eluting stent placed, then received 12 months of open-label thienopyridine plus aspirin. After that, they were randomized to 18 months of continued thienopyridine plus aspirin or placebo plus aspirin.
The DAPT Score was designed to distinguish ischemic and bleeding risk 12 to 30 months after PCI. Patients are given a numerical score (-2 to 10) based on certain risk factors. They receive:
- 1 point each for myocardial infarction at presentation, prior myocardial infarction or PCI, diabetes, stent diameter less than 3 mm, smoking, and paclitaxel-eluting stent
- 2 points each for history of congestive heart failure/low ejection fraction and vein graft intervention
- −1 point for age 65 to younger than 75
- −2 points for age 75 or older.
The researchers validated the DAPT Score in 8136 patients from the PROTECT trial. In this trial, researchers assessed the effect of DAPT on the incidence of stent thrombosis at 3 years in patients randomized to receive the Endeavor zotarolimus-eluting stent or the Cypher sirolimus-eluting stent.
After stent placement, patients were assigned to receive aspirin indefinitely and clopidogrel/ticlopidine for at least 3 months and up to 12 months.
Results in DAPT cohort
In the DAPT cohort, ischemia occurred in 348 patients (3.0%), and bleeding occurred in 215 (1.8%).
The researchers said the derivation cohort models predicting ischemia and bleeding had moderate discrimination, with c statistics of 0.70 and 0.68, respectively. After bootstrap internal validation, optimism-corrected c statistics were 0.68 and 0.66, respectively.
The researchers also compared patients with high DAPT scores (≥2 points) to those with lower scores (<2). As expected, continued DAPT was associated with larger reductions in ischemia and smaller increases in bleeding in the high-score group (n=5917) than in the low-score group (n=5731).
In the high-score group, the incidence of ischemia was 2.7% for continued DAPT and 5.7% for placebo plus aspirin (P<0.001). In the low-score group, the incidence was 1.7% for continued DAPT and 2.3% for placebo plus aspirin (P=0.07; interaction P<0.001).
In the high-score group, the incidence of bleeding was 1.8% for continued DAPT and 1.4% for placebo plus aspirin (P=0.26). In the low-score group, the incidence was 3.0% for continued DAPT and 1.4% for placebo plus aspirin (P<0.001; interaction P=0.02).
Results in PROTECT cohort
In the PROTECT cohort, ischemia occurred in 79 patients (1.0%) and bleeding in 37 patients (0.5%). Again, the models predicting ischemia and bleeding had moderate discrimination, with c statistics of 0.64 for both outcomes.
The rate of ischemia from 12 through 30 months after PCI was greater among the high-score patients (n=2848) than the low-score patients (n=5288). The rates were 1.5% and 0.7%, respectively. The hazard ratio was 2.01 (P=0.002).
Rates of moderate or severe bleeding were not significantly different by DAPT Score. The rates were 0.4% in the high-score patients and 0.5% in the low-score patients. The hazard ratio was 0.69 (P=0.31).
The researchers said that, based on these results, use of the DAPT Score should be cautious pending further validation.
“We haven’t prospectively validated the use of the score, and it’s only applicable to patients similar to those who were randomized in the DAPT study, so we still need to be cautious,” Dr Yeh said. “Nevertheless, we think it represents a significant step forward in understanding benefits and risks of treatment.” ![]()
Researchers believe a new tool could help physicians predict the risks and benefits of extended dual antiplatelet therapy (DAPT) in patients who have undergone percutaneous coronary intervention (PCI).
The team said the tool, known as the DAPT Score, exhibited “modest accuracy” for determining which patients were at high risk for late ischemic events and would therefore benefit most from longer-term DAPT therapy.
The DAPT Score also proved somewhat accurate for identifying patients who were at high risk of late bleeding events and might be harmed by continuing DAPT for more than a year after PCI.
Still, the researchers said the scoring system requires further validation and prospective evaluation to assess its potential effects on patient care.
Robert W. Yeh, MD, of Beth Israel Deaconess Medical Center in Boston, Massachusetts, and his colleagues reported these results in JAMA.
“Dual antiplatelet therapy is standard for patients following coronary stent procedures, but we haven’t had good tools to help us determine how long we should be treating individual patients,” Dr Yeh said.
So he and his colleagues set out to identify factors that would predict whether the expected benefit of reduced ischemia would outweigh the expected increase in bleeding associated with continuing DAPT for more than a year after PCI.
The team used 11,648 patients treated on the DAPT study to create the DAPT Score. Patients in this trial had a drug-eluting stent placed, then received 12 months of open-label thienopyridine plus aspirin. After that, they were randomized to 18 months of continued thienopyridine plus aspirin or placebo plus aspirin.
The DAPT Score was designed to distinguish ischemic and bleeding risk 12 to 30 months after PCI. Patients are given a numerical score (-2 to 10) based on certain risk factors. They receive:
- 1 point each for myocardial infarction at presentation, prior myocardial infarction or PCI, diabetes, stent diameter less than 3 mm, smoking, and paclitaxel-eluting stent
- 2 points each for history of congestive heart failure/low ejection fraction and vein graft intervention
- −1 point for age 65 to younger than 75
- −2 points for age 75 or older.
The researchers validated the DAPT Score in 8136 patients from the PROTECT trial. In this trial, researchers assessed the effect of DAPT on the incidence of stent thrombosis at 3 years in patients randomized to receive the Endeavor zotarolimus-eluting stent or the Cypher sirolimus-eluting stent.
After stent placement, patients were assigned to receive aspirin indefinitely and clopidogrel/ticlopidine for at least 3 months and up to 12 months.
Results in DAPT cohort
In the DAPT cohort, ischemia occurred in 348 patients (3.0%), and bleeding occurred in 215 (1.8%).
The researchers said the derivation cohort models predicting ischemia and bleeding had moderate discrimination, with c statistics of 0.70 and 0.68, respectively. After bootstrap internal validation, optimism-corrected c statistics were 0.68 and 0.66, respectively.
The researchers also compared patients with high DAPT scores (≥2 points) to those with lower scores (<2). As expected, continued DAPT was associated with larger reductions in ischemia and smaller increases in bleeding in the high-score group (n=5917) than in the low-score group (n=5731).
In the high-score group, the incidence of ischemia was 2.7% for continued DAPT and 5.7% for placebo plus aspirin (P<0.001). In the low-score group, the incidence was 1.7% for continued DAPT and 2.3% for placebo plus aspirin (P=0.07; interaction P<0.001).
In the high-score group, the incidence of bleeding was 1.8% for continued DAPT and 1.4% for placebo plus aspirin (P=0.26). In the low-score group, the incidence was 3.0% for continued DAPT and 1.4% for placebo plus aspirin (P<0.001; interaction P=0.02).
Results in PROTECT cohort
In the PROTECT cohort, ischemia occurred in 79 patients (1.0%) and bleeding in 37 patients (0.5%). Again, the models predicting ischemia and bleeding had moderate discrimination, with c statistics of 0.64 for both outcomes.
The rate of ischemia from 12 through 30 months after PCI was greater among the high-score patients (n=2848) than the low-score patients (n=5288). The rates were 1.5% and 0.7%, respectively. The hazard ratio was 2.01 (P=0.002).
Rates of moderate or severe bleeding were not significantly different by DAPT Score. The rates were 0.4% in the high-score patients and 0.5% in the low-score patients. The hazard ratio was 0.69 (P=0.31).
The researchers said that, based on these results, use of the DAPT Score should be cautious pending further validation.
“We haven’t prospectively validated the use of the score, and it’s only applicable to patients similar to those who were randomized in the DAPT study, so we still need to be cautious,” Dr Yeh said. “Nevertheless, we think it represents a significant step forward in understanding benefits and risks of treatment.” ![]()
Researchers believe a new tool could help physicians predict the risks and benefits of extended dual antiplatelet therapy (DAPT) in patients who have undergone percutaneous coronary intervention (PCI).
The team said the tool, known as the DAPT Score, exhibited “modest accuracy” for determining which patients were at high risk for late ischemic events and would therefore benefit most from longer-term DAPT therapy.
The DAPT Score also proved somewhat accurate for identifying patients who were at high risk of late bleeding events and might be harmed by continuing DAPT for more than a year after PCI.
Still, the researchers said the scoring system requires further validation and prospective evaluation to assess its potential effects on patient care.
Robert W. Yeh, MD, of Beth Israel Deaconess Medical Center in Boston, Massachusetts, and his colleagues reported these results in JAMA.
“Dual antiplatelet therapy is standard for patients following coronary stent procedures, but we haven’t had good tools to help us determine how long we should be treating individual patients,” Dr Yeh said.
So he and his colleagues set out to identify factors that would predict whether the expected benefit of reduced ischemia would outweigh the expected increase in bleeding associated with continuing DAPT for more than a year after PCI.
The team used 11,648 patients treated on the DAPT study to create the DAPT Score. Patients in this trial had a drug-eluting stent placed, then received 12 months of open-label thienopyridine plus aspirin. After that, they were randomized to 18 months of continued thienopyridine plus aspirin or placebo plus aspirin.
The DAPT Score was designed to distinguish ischemic and bleeding risk 12 to 30 months after PCI. Patients are given a numerical score (-2 to 10) based on certain risk factors. They receive:
- 1 point each for myocardial infarction at presentation, prior myocardial infarction or PCI, diabetes, stent diameter less than 3 mm, smoking, and paclitaxel-eluting stent
- 2 points each for history of congestive heart failure/low ejection fraction and vein graft intervention
- −1 point for age 65 to younger than 75
- −2 points for age 75 or older.
The researchers validated the DAPT Score in 8136 patients from the PROTECT trial. In this trial, researchers assessed the effect of DAPT on the incidence of stent thrombosis at 3 years in patients randomized to receive the Endeavor zotarolimus-eluting stent or the Cypher sirolimus-eluting stent.
After stent placement, patients were assigned to receive aspirin indefinitely and clopidogrel/ticlopidine for at least 3 months and up to 12 months.
Results in DAPT cohort
In the DAPT cohort, ischemia occurred in 348 patients (3.0%), and bleeding occurred in 215 (1.8%).
The researchers said the derivation cohort models predicting ischemia and bleeding had moderate discrimination, with c statistics of 0.70 and 0.68, respectively. After bootstrap internal validation, optimism-corrected c statistics were 0.68 and 0.66, respectively.
The researchers also compared patients with high DAPT scores (≥2 points) to those with lower scores (<2). As expected, continued DAPT was associated with larger reductions in ischemia and smaller increases in bleeding in the high-score group (n=5917) than in the low-score group (n=5731).
In the high-score group, the incidence of ischemia was 2.7% for continued DAPT and 5.7% for placebo plus aspirin (P<0.001). In the low-score group, the incidence was 1.7% for continued DAPT and 2.3% for placebo plus aspirin (P=0.07; interaction P<0.001).
In the high-score group, the incidence of bleeding was 1.8% for continued DAPT and 1.4% for placebo plus aspirin (P=0.26). In the low-score group, the incidence was 3.0% for continued DAPT and 1.4% for placebo plus aspirin (P<0.001; interaction P=0.02).
Results in PROTECT cohort
In the PROTECT cohort, ischemia occurred in 79 patients (1.0%) and bleeding in 37 patients (0.5%). Again, the models predicting ischemia and bleeding had moderate discrimination, with c statistics of 0.64 for both outcomes.
The rate of ischemia from 12 through 30 months after PCI was greater among the high-score patients (n=2848) than the low-score patients (n=5288). The rates were 1.5% and 0.7%, respectively. The hazard ratio was 2.01 (P=0.002).
Rates of moderate or severe bleeding were not significantly different by DAPT Score. The rates were 0.4% in the high-score patients and 0.5% in the low-score patients. The hazard ratio was 0.69 (P=0.31).
The researchers said that, based on these results, use of the DAPT Score should be cautious pending further validation.
“We haven’t prospectively validated the use of the score, and it’s only applicable to patients similar to those who were randomized in the DAPT study, so we still need to be cautious,” Dr Yeh said. “Nevertheless, we think it represents a significant step forward in understanding benefits and risks of treatment.” ![]()
PET probe could aid treatment for leukemia

Image by Jens Langner
A PET probe known as [18F]CFA could be used to aid the treatment of leukemias and other cancers, according to research published in PNAS.
Investigators say [18F]CFA can detect the activity of deoxycytidine kinase (dCK) in humans more effectively than existing probes.
dCK is a rate-limiting enzyme in the cytosolic deoxyribonucleoside salvage pathway and is considered an important therapeutic and PET-imaging target in certain cancers.
Research has shown that dCK is highly expressed in acute leukemia cells and activated lymphocytes. And the enzyme plays an integral role in allowing drugs such as clofarabine, cytarabine, and fludarabine to treat certain leukemias.
“This enzyme is essential for the therapeutic activity of an entire class of anticancer drugs and even for some antiviral drugs,” said study author Caius Radu, MD, of the University of California, Los Angeles.
“It can take an inactive drug and activate it. If you trick a cancer cell or virus to activate the drug, it would be toxic for the cancer cell or viral genome.”
Until recently, PET technology was only able to clearly detect dCK in mice due to the metabolic instability of the available probes and cross-reactivity with a dCK-related enzyme in humans.
However, Dr Radu and his colleagues showed that [18F]CFA can clearly detect dCK in humans.
The team found that [18F]CFA accumulation in leukemia cells correlated with dCK expression, and they were able to inhibit [18F]CFA accumulation with a dCK inhibitor.
Experiments with [18F]CFA PET/CT in humans showed probe accumulation in tissues with high dCK expression, such as hematopoietic bone marrow and secondary lymphoid organs.
“We are able to clearly see tissues, including tumor tissues, with high dCK activity that we haven’t seen before in humans using any of the other probes previously developed for this enzyme,” Dr Radu said.
He added that, since activated immune cells increase their expression of dCK, [18F]CFA could also be used to monitor the effectiveness of immunotherapeutic interventions.
The investigators hope to begin clinical trials with [18F]CFA in the near future.
Dr Radu and his team invented [18F]CFA and its analogs, which were patented by the University of California and have been licensed to Sofie Biosciences, a company founded by Dr Radu and his team. The University of California also patented additional intellectual property for small-molecule dCK inhibitors. ![]()

Image by Jens Langner
A PET probe known as [18F]CFA could be used to aid the treatment of leukemias and other cancers, according to research published in PNAS.
Investigators say [18F]CFA can detect the activity of deoxycytidine kinase (dCK) in humans more effectively than existing probes.
dCK is a rate-limiting enzyme in the cytosolic deoxyribonucleoside salvage pathway and is considered an important therapeutic and PET-imaging target in certain cancers.
Research has shown that dCK is highly expressed in acute leukemia cells and activated lymphocytes. And the enzyme plays an integral role in allowing drugs such as clofarabine, cytarabine, and fludarabine to treat certain leukemias.
“This enzyme is essential for the therapeutic activity of an entire class of anticancer drugs and even for some antiviral drugs,” said study author Caius Radu, MD, of the University of California, Los Angeles.
“It can take an inactive drug and activate it. If you trick a cancer cell or virus to activate the drug, it would be toxic for the cancer cell or viral genome.”
Until recently, PET technology was only able to clearly detect dCK in mice due to the metabolic instability of the available probes and cross-reactivity with a dCK-related enzyme in humans.
However, Dr Radu and his colleagues showed that [18F]CFA can clearly detect dCK in humans.
The team found that [18F]CFA accumulation in leukemia cells correlated with dCK expression, and they were able to inhibit [18F]CFA accumulation with a dCK inhibitor.
Experiments with [18F]CFA PET/CT in humans showed probe accumulation in tissues with high dCK expression, such as hematopoietic bone marrow and secondary lymphoid organs.
“We are able to clearly see tissues, including tumor tissues, with high dCK activity that we haven’t seen before in humans using any of the other probes previously developed for this enzyme,” Dr Radu said.
He added that, since activated immune cells increase their expression of dCK, [18F]CFA could also be used to monitor the effectiveness of immunotherapeutic interventions.
The investigators hope to begin clinical trials with [18F]CFA in the near future.
Dr Radu and his team invented [18F]CFA and its analogs, which were patented by the University of California and have been licensed to Sofie Biosciences, a company founded by Dr Radu and his team. The University of California also patented additional intellectual property for small-molecule dCK inhibitors. ![]()

Image by Jens Langner
A PET probe known as [18F]CFA could be used to aid the treatment of leukemias and other cancers, according to research published in PNAS.
Investigators say [18F]CFA can detect the activity of deoxycytidine kinase (dCK) in humans more effectively than existing probes.
dCK is a rate-limiting enzyme in the cytosolic deoxyribonucleoside salvage pathway and is considered an important therapeutic and PET-imaging target in certain cancers.
Research has shown that dCK is highly expressed in acute leukemia cells and activated lymphocytes. And the enzyme plays an integral role in allowing drugs such as clofarabine, cytarabine, and fludarabine to treat certain leukemias.
“This enzyme is essential for the therapeutic activity of an entire class of anticancer drugs and even for some antiviral drugs,” said study author Caius Radu, MD, of the University of California, Los Angeles.
“It can take an inactive drug and activate it. If you trick a cancer cell or virus to activate the drug, it would be toxic for the cancer cell or viral genome.”
Until recently, PET technology was only able to clearly detect dCK in mice due to the metabolic instability of the available probes and cross-reactivity with a dCK-related enzyme in humans.
However, Dr Radu and his colleagues showed that [18F]CFA can clearly detect dCK in humans.
The team found that [18F]CFA accumulation in leukemia cells correlated with dCK expression, and they were able to inhibit [18F]CFA accumulation with a dCK inhibitor.
Experiments with [18F]CFA PET/CT in humans showed probe accumulation in tissues with high dCK expression, such as hematopoietic bone marrow and secondary lymphoid organs.
“We are able to clearly see tissues, including tumor tissues, with high dCK activity that we haven’t seen before in humans using any of the other probes previously developed for this enzyme,” Dr Radu said.
He added that, since activated immune cells increase their expression of dCK, [18F]CFA could also be used to monitor the effectiveness of immunotherapeutic interventions.
The investigators hope to begin clinical trials with [18F]CFA in the near future.
Dr Radu and his team invented [18F]CFA and its analogs, which were patented by the University of California and have been licensed to Sofie Biosciences, a company founded by Dr Radu and his team. The University of California also patented additional intellectual property for small-molecule dCK inhibitors. ![]()
Blood culture panel cleared by FDA

Staphylococcus infection
Photo by Bill Branson
The US Food and Drug Administration (FDA) has granted 510(k) clearance for a blood culture panel that detects sepsis caused by methicillin-resistant Staphylococcus aureus (MRSA) and other Staphylococcus species.
The Staph ID/R Blood Culture Panel is a product of Great Basin Scientific, Inc.
It is an automated, DNA multiplex assay used to identify Staphylococcus species directly from positive blood cultures in about 2 hours.
The panel also detects the mecA gene, a drug-resistance marker that confers resistance to methicillin and other beta-lactams and creates MRSA.
In addition, the Staph ID/R Blood Culture Panel identifies coagulase-negative staphylococci.
According to the US Centers for Disease Control and Prevention, 20% to 50% of all positive blood cultures are likely false positives due to contamination caused by coagulase-negative staphylococci, many of which are part of the normal flora of human skin and are not dangerous.
The Staph ID/R Blood Culture Panel is run on the Great Basin Analyzer. The company says the assay requires less than a minute of hands-on time and no results interpretation due to electronic results reporting. ![]()

Staphylococcus infection
Photo by Bill Branson
The US Food and Drug Administration (FDA) has granted 510(k) clearance for a blood culture panel that detects sepsis caused by methicillin-resistant Staphylococcus aureus (MRSA) and other Staphylococcus species.
The Staph ID/R Blood Culture Panel is a product of Great Basin Scientific, Inc.
It is an automated, DNA multiplex assay used to identify Staphylococcus species directly from positive blood cultures in about 2 hours.
The panel also detects the mecA gene, a drug-resistance marker that confers resistance to methicillin and other beta-lactams and creates MRSA.
In addition, the Staph ID/R Blood Culture Panel identifies coagulase-negative staphylococci.
According to the US Centers for Disease Control and Prevention, 20% to 50% of all positive blood cultures are likely false positives due to contamination caused by coagulase-negative staphylococci, many of which are part of the normal flora of human skin and are not dangerous.
The Staph ID/R Blood Culture Panel is run on the Great Basin Analyzer. The company says the assay requires less than a minute of hands-on time and no results interpretation due to electronic results reporting. ![]()

Staphylococcus infection
Photo by Bill Branson
The US Food and Drug Administration (FDA) has granted 510(k) clearance for a blood culture panel that detects sepsis caused by methicillin-resistant Staphylococcus aureus (MRSA) and other Staphylococcus species.
The Staph ID/R Blood Culture Panel is a product of Great Basin Scientific, Inc.
It is an automated, DNA multiplex assay used to identify Staphylococcus species directly from positive blood cultures in about 2 hours.
The panel also detects the mecA gene, a drug-resistance marker that confers resistance to methicillin and other beta-lactams and creates MRSA.
In addition, the Staph ID/R Blood Culture Panel identifies coagulase-negative staphylococci.
According to the US Centers for Disease Control and Prevention, 20% to 50% of all positive blood cultures are likely false positives due to contamination caused by coagulase-negative staphylococci, many of which are part of the normal flora of human skin and are not dangerous.
The Staph ID/R Blood Culture Panel is run on the Great Basin Analyzer. The company says the assay requires less than a minute of hands-on time and no results interpretation due to electronic results reporting. ![]()
Accuracy of blood test results varies

Photo by Graham Colm
A comparison of commercially available blood tests has revealed more variability than expected, according to researchers.
The group compared basic blood tests run by commercial laboratories and found the testing service, type of test, and time of collection all influenced the accuracy of results.
Given that these tests can be used for disease diagnosis or to determine whether a patient’s medication is working, the researchers said this study highlights the importance of knowing the accuracy and variability of blood test results.
“While most of the variability we found was within clinically accepted ranges, there were several cases where inaccurate results would have led to incorrect medical decisions,” said Joel Dudley, PhD, of the Icahn School of Medicine at Mount Sinai in New York, New York.
“We hope this study will inspire the biomedical community to take a critical look at all testing variables to ensure that lab results are as robust and reproducible as possible.”
Dr Dudley and his colleagues described this study in the Journal of Clinical Investigation.
The researchers collected peripheral blood samples from 60 healthy adults at 4 separate time points within a 6.5-hour window. The samples were collected in Phoenix, Arizona, at an ambulatory clinic and at retail outlets with point-of-care services.
The team collected 14 samples per subject and used those samples to compare 22 common clinical lab tests conducted at 3 commercial labs. One lab, Theranos, offered blood tests obtained from a finger prick, and the other 2, Quest and LabCorp, required standard venipuncture draws.
More than half of the test results showed significant differences between test providers. Of the 22 tests, 15 (68%) showed significant variability between labs (P<0.002).
Triglyceride levels and red blood cell counts were among the most consistent results, while white blood cell counts and overall cholesterol levels were among the most variable.
Test results from Theranos were flagged by Theranos as abnormal 1.6 times more often than tests from LabCorp or Quest (P<0.0001). The percentages for measurements outside their normal range were 8.3% for LabCorp, 7.5% for Quest, and 12.2% for Theranos.
In addition, the researchers noted that, although they controlled subjects’ eating and physical activity, data from blood samples collected earlier in the day were sometimes significantly different from samples taken from the same subjects later in the day.
There were significant difference between measurements collected at time points 1 and 2 vs time points 3 and 4 for 13 of the 22 tests (P<0.002).
“These testing disparities occurred despite rigorous laboratory certification and proficiency standards designed to ensure consistency,” said study author Eric Schadt, PhD, of Mount Sinai.
“Our results suggest the need for greater transparency in lab technologies and procedures, as well as a much more thorough investigation of biological mechanisms that may contribute to more dynamic levels than we currently understand.” ![]()

Photo by Graham Colm
A comparison of commercially available blood tests has revealed more variability than expected, according to researchers.
The group compared basic blood tests run by commercial laboratories and found the testing service, type of test, and time of collection all influenced the accuracy of results.
Given that these tests can be used for disease diagnosis or to determine whether a patient’s medication is working, the researchers said this study highlights the importance of knowing the accuracy and variability of blood test results.
“While most of the variability we found was within clinically accepted ranges, there were several cases where inaccurate results would have led to incorrect medical decisions,” said Joel Dudley, PhD, of the Icahn School of Medicine at Mount Sinai in New York, New York.
“We hope this study will inspire the biomedical community to take a critical look at all testing variables to ensure that lab results are as robust and reproducible as possible.”
Dr Dudley and his colleagues described this study in the Journal of Clinical Investigation.
The researchers collected peripheral blood samples from 60 healthy adults at 4 separate time points within a 6.5-hour window. The samples were collected in Phoenix, Arizona, at an ambulatory clinic and at retail outlets with point-of-care services.
The team collected 14 samples per subject and used those samples to compare 22 common clinical lab tests conducted at 3 commercial labs. One lab, Theranos, offered blood tests obtained from a finger prick, and the other 2, Quest and LabCorp, required standard venipuncture draws.
More than half of the test results showed significant differences between test providers. Of the 22 tests, 15 (68%) showed significant variability between labs (P<0.002).
Triglyceride levels and red blood cell counts were among the most consistent results, while white blood cell counts and overall cholesterol levels were among the most variable.
Test results from Theranos were flagged by Theranos as abnormal 1.6 times more often than tests from LabCorp or Quest (P<0.0001). The percentages for measurements outside their normal range were 8.3% for LabCorp, 7.5% for Quest, and 12.2% for Theranos.
In addition, the researchers noted that, although they controlled subjects’ eating and physical activity, data from blood samples collected earlier in the day were sometimes significantly different from samples taken from the same subjects later in the day.
There were significant difference between measurements collected at time points 1 and 2 vs time points 3 and 4 for 13 of the 22 tests (P<0.002).
“These testing disparities occurred despite rigorous laboratory certification and proficiency standards designed to ensure consistency,” said study author Eric Schadt, PhD, of Mount Sinai.
“Our results suggest the need for greater transparency in lab technologies and procedures, as well as a much more thorough investigation of biological mechanisms that may contribute to more dynamic levels than we currently understand.” ![]()

Photo by Graham Colm
A comparison of commercially available blood tests has revealed more variability than expected, according to researchers.
The group compared basic blood tests run by commercial laboratories and found the testing service, type of test, and time of collection all influenced the accuracy of results.
Given that these tests can be used for disease diagnosis or to determine whether a patient’s medication is working, the researchers said this study highlights the importance of knowing the accuracy and variability of blood test results.
“While most of the variability we found was within clinically accepted ranges, there were several cases where inaccurate results would have led to incorrect medical decisions,” said Joel Dudley, PhD, of the Icahn School of Medicine at Mount Sinai in New York, New York.
“We hope this study will inspire the biomedical community to take a critical look at all testing variables to ensure that lab results are as robust and reproducible as possible.”
Dr Dudley and his colleagues described this study in the Journal of Clinical Investigation.
The researchers collected peripheral blood samples from 60 healthy adults at 4 separate time points within a 6.5-hour window. The samples were collected in Phoenix, Arizona, at an ambulatory clinic and at retail outlets with point-of-care services.
The team collected 14 samples per subject and used those samples to compare 22 common clinical lab tests conducted at 3 commercial labs. One lab, Theranos, offered blood tests obtained from a finger prick, and the other 2, Quest and LabCorp, required standard venipuncture draws.
More than half of the test results showed significant differences between test providers. Of the 22 tests, 15 (68%) showed significant variability between labs (P<0.002).
Triglyceride levels and red blood cell counts were among the most consistent results, while white blood cell counts and overall cholesterol levels were among the most variable.
Test results from Theranos were flagged by Theranos as abnormal 1.6 times more often than tests from LabCorp or Quest (P<0.0001). The percentages for measurements outside their normal range were 8.3% for LabCorp, 7.5% for Quest, and 12.2% for Theranos.
In addition, the researchers noted that, although they controlled subjects’ eating and physical activity, data from blood samples collected earlier in the day were sometimes significantly different from samples taken from the same subjects later in the day.
There were significant difference between measurements collected at time points 1 and 2 vs time points 3 and 4 for 13 of the 22 tests (P<0.002).
“These testing disparities occurred despite rigorous laboratory certification and proficiency standards designed to ensure consistency,” said study author Eric Schadt, PhD, of Mount Sinai.
“Our results suggest the need for greater transparency in lab technologies and procedures, as well as a much more thorough investigation of biological mechanisms that may contribute to more dynamic levels than we currently understand.” ![]()
High costs limit CML patients’ access to TKIs

Photo courtesy of the CDC
A new study suggests that cost-sharing policies in the US create a barrier to the treatment of chronic myeloid leukemia (CML).
Researchers examined Medicare claims data and found that “Part D” (prescription drug plan) co-insurance policies for “specialty drugs” seem to be reducing or delaying the use of tyrosine kinase inhibitors (TKIs) in patients with CML.
The team reported these findings in the American Journal of Managed Care.
“High out-of-pocket costs for specialty drugs appear to pose a very real barrier to treatment,” said study author Jalpa A. Doshi, PhD, of the Perelman School of Medicine at the University of Pennsylvania in Philadelphia.
While there is no standard definition for specialty drugs, the term typically refers to medications requiring special handling, administration, or monitoring. Most are aimed at treating chronic or life-threatening diseases.
Although specialty drugs typically tend to offer significant medical advances over non-specialty drugs, they are correspondingly more expensive. In 2014, such drugs accounted for less than 1% of prescriptions in the US but nearly a third of total prescription spending.
While insurers have been imposing higher cost-sharing requirements as part of their efforts to manage specialty drug spending, there has been limited information about the corresponding impact on patients.
“[I]t was particularly important to examine the extent to which the aggressive cost-sharing policies for specialty drugs seen under Medicare Part D, which are increasingly making their way into the private insurance market, adversely impact access to these treatments even for a condition like cancer,” Dr Doshi said.
So she and her colleagues examined the impact of specialty drug cost-sharing under the Medicare Part D prescription drug benefit on patients with CML. The team analyzed Medicare data on patients who were newly diagnosed with CML to examine whether and how quickly they initiated TKI treatment.
The researchers compared patients who were eligible for low-income subsidies and therefore faced nominal out-of-pocket costs to patients who faced average out-of-pocket costs of $2600 or more for their first 30-day TKI prescription fill.
Results showed that patients in the high-cost group were significantly less likely than the low-cost group to have a Part D claim for a TKI prescription within 6 months of their CML diagnosis. The rates were 45.3% and 66.9%, respectively (P<0.001).
Patients in the high cost-sharing group also took twice as long, on average, to initiate TKI treatment. The mean time to fill a TKI prescription was 50.9 days in the high-cost group and 23.7 days in the low-cost group (P<0.001).
“Medicare Part D was created to increase access to prescription drug treatment among beneficiaries, but our data suggest that current policies are interfering with that goal when it comes to specialty drugs,” Dr Doshi said.
She added that making Part D out-of-pocket costs more consistent and limiting them to more reasonable sums would help mitigate this negative impact.
Dr Doshi and her colleagues are now pursuing further studies of the impact of Part D cost-sharing policies in different disease areas. They hope to gain a better understanding of changes in drug access and of the long-range clinical outcomes and costs associated with any delays or interruptions in treatment.
“We need to know if the current aggressive cost-sharing arrangements have adverse long-term impacts on health and perhaps, paradoxically, increase overall spending due to complications of poorly controlled disease,” Dr Doshi said. ![]()

Photo courtesy of the CDC
A new study suggests that cost-sharing policies in the US create a barrier to the treatment of chronic myeloid leukemia (CML).
Researchers examined Medicare claims data and found that “Part D” (prescription drug plan) co-insurance policies for “specialty drugs” seem to be reducing or delaying the use of tyrosine kinase inhibitors (TKIs) in patients with CML.
The team reported these findings in the American Journal of Managed Care.
“High out-of-pocket costs for specialty drugs appear to pose a very real barrier to treatment,” said study author Jalpa A. Doshi, PhD, of the Perelman School of Medicine at the University of Pennsylvania in Philadelphia.
While there is no standard definition for specialty drugs, the term typically refers to medications requiring special handling, administration, or monitoring. Most are aimed at treating chronic or life-threatening diseases.
Although specialty drugs typically tend to offer significant medical advances over non-specialty drugs, they are correspondingly more expensive. In 2014, such drugs accounted for less than 1% of prescriptions in the US but nearly a third of total prescription spending.
While insurers have been imposing higher cost-sharing requirements as part of their efforts to manage specialty drug spending, there has been limited information about the corresponding impact on patients.
“[I]t was particularly important to examine the extent to which the aggressive cost-sharing policies for specialty drugs seen under Medicare Part D, which are increasingly making their way into the private insurance market, adversely impact access to these treatments even for a condition like cancer,” Dr Doshi said.
So she and her colleagues examined the impact of specialty drug cost-sharing under the Medicare Part D prescription drug benefit on patients with CML. The team analyzed Medicare data on patients who were newly diagnosed with CML to examine whether and how quickly they initiated TKI treatment.
The researchers compared patients who were eligible for low-income subsidies and therefore faced nominal out-of-pocket costs to patients who faced average out-of-pocket costs of $2600 or more for their first 30-day TKI prescription fill.
Results showed that patients in the high-cost group were significantly less likely than the low-cost group to have a Part D claim for a TKI prescription within 6 months of their CML diagnosis. The rates were 45.3% and 66.9%, respectively (P<0.001).
Patients in the high cost-sharing group also took twice as long, on average, to initiate TKI treatment. The mean time to fill a TKI prescription was 50.9 days in the high-cost group and 23.7 days in the low-cost group (P<0.001).
“Medicare Part D was created to increase access to prescription drug treatment among beneficiaries, but our data suggest that current policies are interfering with that goal when it comes to specialty drugs,” Dr Doshi said.
She added that making Part D out-of-pocket costs more consistent and limiting them to more reasonable sums would help mitigate this negative impact.
Dr Doshi and her colleagues are now pursuing further studies of the impact of Part D cost-sharing policies in different disease areas. They hope to gain a better understanding of changes in drug access and of the long-range clinical outcomes and costs associated with any delays or interruptions in treatment.
“We need to know if the current aggressive cost-sharing arrangements have adverse long-term impacts on health and perhaps, paradoxically, increase overall spending due to complications of poorly controlled disease,” Dr Doshi said. ![]()

Photo courtesy of the CDC
A new study suggests that cost-sharing policies in the US create a barrier to the treatment of chronic myeloid leukemia (CML).
Researchers examined Medicare claims data and found that “Part D” (prescription drug plan) co-insurance policies for “specialty drugs” seem to be reducing or delaying the use of tyrosine kinase inhibitors (TKIs) in patients with CML.
The team reported these findings in the American Journal of Managed Care.
“High out-of-pocket costs for specialty drugs appear to pose a very real barrier to treatment,” said study author Jalpa A. Doshi, PhD, of the Perelman School of Medicine at the University of Pennsylvania in Philadelphia.
While there is no standard definition for specialty drugs, the term typically refers to medications requiring special handling, administration, or monitoring. Most are aimed at treating chronic or life-threatening diseases.
Although specialty drugs typically tend to offer significant medical advances over non-specialty drugs, they are correspondingly more expensive. In 2014, such drugs accounted for less than 1% of prescriptions in the US but nearly a third of total prescription spending.
While insurers have been imposing higher cost-sharing requirements as part of their efforts to manage specialty drug spending, there has been limited information about the corresponding impact on patients.
“[I]t was particularly important to examine the extent to which the aggressive cost-sharing policies for specialty drugs seen under Medicare Part D, which are increasingly making their way into the private insurance market, adversely impact access to these treatments even for a condition like cancer,” Dr Doshi said.
So she and her colleagues examined the impact of specialty drug cost-sharing under the Medicare Part D prescription drug benefit on patients with CML. The team analyzed Medicare data on patients who were newly diagnosed with CML to examine whether and how quickly they initiated TKI treatment.
The researchers compared patients who were eligible for low-income subsidies and therefore faced nominal out-of-pocket costs to patients who faced average out-of-pocket costs of $2600 or more for their first 30-day TKI prescription fill.
Results showed that patients in the high-cost group were significantly less likely than the low-cost group to have a Part D claim for a TKI prescription within 6 months of their CML diagnosis. The rates were 45.3% and 66.9%, respectively (P<0.001).
Patients in the high cost-sharing group also took twice as long, on average, to initiate TKI treatment. The mean time to fill a TKI prescription was 50.9 days in the high-cost group and 23.7 days in the low-cost group (P<0.001).
“Medicare Part D was created to increase access to prescription drug treatment among beneficiaries, but our data suggest that current policies are interfering with that goal when it comes to specialty drugs,” Dr Doshi said.
She added that making Part D out-of-pocket costs more consistent and limiting them to more reasonable sums would help mitigate this negative impact.
Dr Doshi and her colleagues are now pursuing further studies of the impact of Part D cost-sharing policies in different disease areas. They hope to gain a better understanding of changes in drug access and of the long-range clinical outcomes and costs associated with any delays or interruptions in treatment.
“We need to know if the current aggressive cost-sharing arrangements have adverse long-term impacts on health and perhaps, paradoxically, increase overall spending due to complications of poorly controlled disease,” Dr Doshi said.
iPSCs can differentiate into functional lymphocytes

Image from the Salk Institute
Researchers say they have generated induced pluripotent stem cells (iPSCs) that can differentiate into multiple lineages of functional lymphocytes.
The team noted that lymphohematopoietic stem cells (L-HSCs) generated from self-somatic cell-derived iPSCs could potentially be used to treat hematologic disorders, but no one has generated “truly functional” L-HSCs from iPSCs.
So the researchers set out to determine whether iPSCs have the inherent potential to generate multiple lineages of functional, terminally differentiated lymphocytes.
They described this work in Stem Cells and Development.
The researchers said they used tetraploid embryo complementation to provide a normal environment for the differentiation of L-HSCs from iPSCs and embryonic stem cells (ESCs). The team then compared lymphocytes derived from iPSCs, ESCs, and naïve isogenic C57BL/6 mice.
The researchers found that iPSC-derived lymphocytes expressed normal levels of major histocompatibility complex-I. Levels were comparable in iPSC-derived lymphocytes, ESC-derived lymphocytes, and lymphocytes from the control mice.
In addition, iPSC-derived lymphocytes were able to differentiate into multiple cell types—CD4+ T cells, CD8+ T cells, regulatory T cells, B cells, and natural killer cells.
Lymphocytes generated from iPSCs and lymphocytes generated from ESCs had the same capacity as lymphocytes from the control mice to proliferate and secrete chemical signals, such as cytokines.
All 3 types of lymphocytes proliferated under allogeneic stimulation but not under syngeneic stimulation. And the researchers found similar levels of IL-2, IL-4, IL-6, IL-10, IL-17, TNF, and IFN-γ in iPSC, ESC, and C57BL/6 lymphocyte culture supernatants.
The team also found that lymphocytes generated by iPSC-derived bone marrow cells could repopulate the hematopoietic systems of lethally irradiated recipient mice.
The iPSC bone marrow cells proved as effective as ESC-derived bone marrow cells and wild-type bone marrow cells. All 3 types of cells negated lymphocyte storage exhaustion in the spleen and peripheral blood.
In addition, there were no major phenotypic or behavioral abnormalities in any of the mice more than 1 month after cell transplantation.
The researchers said this work shows that truly functional lymphocytes can be generated from iPSCs, and it supports the clinical application of iPSC technology to develop treatments for hematologic disorders.

Image from the Salk Institute
Researchers say they have generated induced pluripotent stem cells (iPSCs) that can differentiate into multiple lineages of functional lymphocytes.
The team noted that lymphohematopoietic stem cells (L-HSCs) generated from self-somatic cell-derived iPSCs could potentially be used to treat hematologic disorders, but no one has generated “truly functional” L-HSCs from iPSCs.
So the researchers set out to determine whether iPSCs have the inherent potential to generate multiple lineages of functional, terminally differentiated lymphocytes.
They described this work in Stem Cells and Development.
The researchers said they used tetraploid embryo complementation to provide a normal environment for the differentiation of L-HSCs from iPSCs and embryonic stem cells (ESCs). The team then compared lymphocytes derived from iPSCs, ESCs, and naïve isogenic C57BL/6 mice.
The researchers found that iPSC-derived lymphocytes expressed normal levels of major histocompatibility complex-I. Levels were comparable in iPSC-derived lymphocytes, ESC-derived lymphocytes, and lymphocytes from the control mice.
In addition, iPSC-derived lymphocytes were able to differentiate into multiple cell types—CD4+ T cells, CD8+ T cells, regulatory T cells, B cells, and natural killer cells.
Lymphocytes generated from iPSCs and lymphocytes generated from ESCs had the same capacity as lymphocytes from the control mice to proliferate and secrete chemical signals, such as cytokines.
All 3 types of lymphocytes proliferated under allogeneic stimulation but not under syngeneic stimulation. And the researchers found similar levels of IL-2, IL-4, IL-6, IL-10, IL-17, TNF, and IFN-γ in iPSC, ESC, and C57BL/6 lymphocyte culture supernatants.
The team also found that lymphocytes generated by iPSC-derived bone marrow cells could repopulate the hematopoietic systems of lethally irradiated recipient mice.
The iPSC bone marrow cells proved as effective as ESC-derived bone marrow cells and wild-type bone marrow cells. All 3 types of cells negated lymphocyte storage exhaustion in the spleen and peripheral blood.
In addition, there were no major phenotypic or behavioral abnormalities in any of the mice more than 1 month after cell transplantation.
The researchers said this work shows that truly functional lymphocytes can be generated from iPSCs, and it supports the clinical application of iPSC technology to develop treatments for hematologic disorders.

Image from the Salk Institute
Researchers say they have generated induced pluripotent stem cells (iPSCs) that can differentiate into multiple lineages of functional lymphocytes.
The team noted that lymphohematopoietic stem cells (L-HSCs) generated from self-somatic cell-derived iPSCs could potentially be used to treat hematologic disorders, but no one has generated “truly functional” L-HSCs from iPSCs.
So the researchers set out to determine whether iPSCs have the inherent potential to generate multiple lineages of functional, terminally differentiated lymphocytes.
They described this work in Stem Cells and Development.
The researchers said they used tetraploid embryo complementation to provide a normal environment for the differentiation of L-HSCs from iPSCs and embryonic stem cells (ESCs). The team then compared lymphocytes derived from iPSCs, ESCs, and naïve isogenic C57BL/6 mice.
The researchers found that iPSC-derived lymphocytes expressed normal levels of major histocompatibility complex-I. Levels were comparable in iPSC-derived lymphocytes, ESC-derived lymphocytes, and lymphocytes from the control mice.
In addition, iPSC-derived lymphocytes were able to differentiate into multiple cell types—CD4+ T cells, CD8+ T cells, regulatory T cells, B cells, and natural killer cells.
Lymphocytes generated from iPSCs and lymphocytes generated from ESCs had the same capacity as lymphocytes from the control mice to proliferate and secrete chemical signals, such as cytokines.
All 3 types of lymphocytes proliferated under allogeneic stimulation but not under syngeneic stimulation. And the researchers found similar levels of IL-2, IL-4, IL-6, IL-10, IL-17, TNF, and IFN-γ in iPSC, ESC, and C57BL/6 lymphocyte culture supernatants.
The team also found that lymphocytes generated by iPSC-derived bone marrow cells could repopulate the hematopoietic systems of lethally irradiated recipient mice.
The iPSC bone marrow cells proved as effective as ESC-derived bone marrow cells and wild-type bone marrow cells. All 3 types of cells negated lymphocyte storage exhaustion in the spleen and peripheral blood.
In addition, there were no major phenotypic or behavioral abnormalities in any of the mice more than 1 month after cell transplantation.
The researchers said this work shows that truly functional lymphocytes can be generated from iPSCs, and it supports the clinical application of iPSC technology to develop treatments for hematologic disorders.
A better method for detecting amyloidosis?
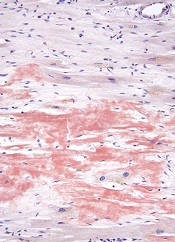
A novel molecular probe can detect amyloidosis at least as well as—and perhaps even better than—traditional methods, according to research published in Amyloid: The Journal of Protein Folding Disorders.
Investigators found that a luminescent conjugated oligothiophene, h-FTAA, allowed them to correctly identify amyloidosis in every sample tested.
But results also suggested h-FTAA may be more sensitive than traditional methods used to diagnose amyloidosis, as h-FTAA detected small amyloid deposits in samples that were previously determined to be amyloid-free.
The investigators said this suggests h-FTAA could be used to detect amyloidosis before symptoms present, leading to faster treatment.
“Given the sensitivity of the probe, we think this would make an excellent complement to traditional methods and could eventually be a replacement,” said study author Per Hammarström, PhD, of Linköping University in Sweden.
Dr Hammarström and his colleagues screened amyloid-containing tissues from 107 patients who had their amyloidosis verified by Congo red staining and/or immunohistochemistry, as well as tissues from 32 negative control cases.
The results showed that h-FTAA could detect amyloidosis with 100% sensitivity, identifying amyloid deposits in all 107 patients.
However, h-FTAA also detected microdeposits of amyloid-like protein aggregates in 5 of the control samples that were negative according to Congo red.
The investigators said they don’t know the clinical significance of these “false-positive” lesions. However, because h-FTAA fluorescence is 1 magnitude brighter than Congo red and because the staining is performed 4 magnitudes lower than the concentration of dye, the team believes these 5 cases may have been beyond detection by Congo red and h-FTAA may be a more sensitive technique.
They therefore concluded that h-FTAA could potentially be used as a complementary technique for accurate detection of amyloid in routine surgical pathology settings, for the detection of prodromal amyloidosis, and for the discovery of new amyloid-like protein aggregates.

A novel molecular probe can detect amyloidosis at least as well as—and perhaps even better than—traditional methods, according to research published in Amyloid: The Journal of Protein Folding Disorders.
Investigators found that a luminescent conjugated oligothiophene, h-FTAA, allowed them to correctly identify amyloidosis in every sample tested.
But results also suggested h-FTAA may be more sensitive than traditional methods used to diagnose amyloidosis, as h-FTAA detected small amyloid deposits in samples that were previously determined to be amyloid-free.
The investigators said this suggests h-FTAA could be used to detect amyloidosis before symptoms present, leading to faster treatment.
“Given the sensitivity of the probe, we think this would make an excellent complement to traditional methods and could eventually be a replacement,” said study author Per Hammarström, PhD, of Linköping University in Sweden.
Dr Hammarström and his colleagues screened amyloid-containing tissues from 107 patients who had their amyloidosis verified by Congo red staining and/or immunohistochemistry, as well as tissues from 32 negative control cases.
The results showed that h-FTAA could detect amyloidosis with 100% sensitivity, identifying amyloid deposits in all 107 patients.
However, h-FTAA also detected microdeposits of amyloid-like protein aggregates in 5 of the control samples that were negative according to Congo red.
The investigators said they don’t know the clinical significance of these “false-positive” lesions. However, because h-FTAA fluorescence is 1 magnitude brighter than Congo red and because the staining is performed 4 magnitudes lower than the concentration of dye, the team believes these 5 cases may have been beyond detection by Congo red and h-FTAA may be a more sensitive technique.
They therefore concluded that h-FTAA could potentially be used as a complementary technique for accurate detection of amyloid in routine surgical pathology settings, for the detection of prodromal amyloidosis, and for the discovery of new amyloid-like protein aggregates.

A novel molecular probe can detect amyloidosis at least as well as—and perhaps even better than—traditional methods, according to research published in Amyloid: The Journal of Protein Folding Disorders.
Investigators found that a luminescent conjugated oligothiophene, h-FTAA, allowed them to correctly identify amyloidosis in every sample tested.
But results also suggested h-FTAA may be more sensitive than traditional methods used to diagnose amyloidosis, as h-FTAA detected small amyloid deposits in samples that were previously determined to be amyloid-free.
The investigators said this suggests h-FTAA could be used to detect amyloidosis before symptoms present, leading to faster treatment.
“Given the sensitivity of the probe, we think this would make an excellent complement to traditional methods and could eventually be a replacement,” said study author Per Hammarström, PhD, of Linköping University in Sweden.
Dr Hammarström and his colleagues screened amyloid-containing tissues from 107 patients who had their amyloidosis verified by Congo red staining and/or immunohistochemistry, as well as tissues from 32 negative control cases.
The results showed that h-FTAA could detect amyloidosis with 100% sensitivity, identifying amyloid deposits in all 107 patients.
However, h-FTAA also detected microdeposits of amyloid-like protein aggregates in 5 of the control samples that were negative according to Congo red.
The investigators said they don’t know the clinical significance of these “false-positive” lesions. However, because h-FTAA fluorescence is 1 magnitude brighter than Congo red and because the staining is performed 4 magnitudes lower than the concentration of dye, the team believes these 5 cases may have been beyond detection by Congo red and h-FTAA may be a more sensitive technique.
They therefore concluded that h-FTAA could potentially be used as a complementary technique for accurate detection of amyloid in routine surgical pathology settings, for the detection of prodromal amyloidosis, and for the discovery of new amyloid-like protein aggregates.
Drug ‘not powerful enough’ to treat CTCL

mycosis fungoides
Results of a phase 2 trial suggest the drug APO866 is not suitable for the treatment of relapsed or refractory cutaneous T-cell lymphoma (CTCL).
Researchers said APO866 had “a reasonable toxic effect,” but it was “not powerful enough,” so the trial was stopped early.
Two of the 14 patients studied achieved a partial response during the trial, but there were no complete responses, and most patients withdrew from the study early.
The researchers described these results in a letter to JAMA Dermatology. The study was initially sponsored by Apoxis SA and later by TopoTarget A/S, which is now known as Onxeo after merging with BioAlliance Pharma.
According to Onxeo, APO866 is an injectable molecule that induces apoptosis by inhibiting the biosynthesis of NAD+ from niacinamide, which is essential for the cellular metabolism, protein modification, and calcium-dependent messenger synthesis.
In the phase 2 trial, researchers tested APO866 in 14 patients with relapsed or refractory CTCL. The patients were 19 to 83 years of age, and half were female.
Eight patients had mycosis fungoides, 3 had Sézary syndrome, 1 had CD30+ anaplastic large-cell lymphoma, 1 had poikilodermic mycosis fungoides, and 1 had CD30- nonepidermotropic CTCL. One patient had stage IB disease, 2 had stage IIA, 3 had stage IIB, and 8 had stage IVA.
The patients received a continuous intravenous infusion, via pump, of APO866 at 0.126 mg/m2/h over the course of 96 hours. Patients received this treatment every 28 days for a total of 3 cycles.
Five patients completed all 3 treatment cycles and had no major protocol violations. Nine patients discontinued treatment early due to consent withdrawal (n=2), early disease progression (n=5), or adverse events (AEs, n=2).
At week 8, 1 patient had achieved a partial response to treatment, 4 patients had stable disease, 5 had progressed, and 4 patients were not evaluable because they had withdrawn from the study.
At week 16, 1 patient had a partial response (not the same patient as at week 8), 4 patients had stable disease, and 9 patients were not evaluable due to withdrawal.
There were a total of 141 AEs, and 77 of these were considered related to APO866. Most patients (n=12) had mild to moderate AEs, but there were 7 serious AEs thought to be treatment-related. These included pyrexia, lymphopenia (n=2), spondylitis, Staphylococcal sepsis, rhabdomyolysis, and thrombocytopenia.
There were 4 deaths, but they were not considered drug-related.
The researchers said these results suggest APO866 should not be pursued as a treatment for CTCL. However, as the drug induces immunosuppression and has insulin-mimicking effects, it might prove useful for treating other conditions.

mycosis fungoides
Results of a phase 2 trial suggest the drug APO866 is not suitable for the treatment of relapsed or refractory cutaneous T-cell lymphoma (CTCL).
Researchers said APO866 had “a reasonable toxic effect,” but it was “not powerful enough,” so the trial was stopped early.
Two of the 14 patients studied achieved a partial response during the trial, but there were no complete responses, and most patients withdrew from the study early.
The researchers described these results in a letter to JAMA Dermatology. The study was initially sponsored by Apoxis SA and later by TopoTarget A/S, which is now known as Onxeo after merging with BioAlliance Pharma.
According to Onxeo, APO866 is an injectable molecule that induces apoptosis by inhibiting the biosynthesis of NAD+ from niacinamide, which is essential for the cellular metabolism, protein modification, and calcium-dependent messenger synthesis.
In the phase 2 trial, researchers tested APO866 in 14 patients with relapsed or refractory CTCL. The patients were 19 to 83 years of age, and half were female.
Eight patients had mycosis fungoides, 3 had Sézary syndrome, 1 had CD30+ anaplastic large-cell lymphoma, 1 had poikilodermic mycosis fungoides, and 1 had CD30- nonepidermotropic CTCL. One patient had stage IB disease, 2 had stage IIA, 3 had stage IIB, and 8 had stage IVA.
The patients received a continuous intravenous infusion, via pump, of APO866 at 0.126 mg/m2/h over the course of 96 hours. Patients received this treatment every 28 days for a total of 3 cycles.
Five patients completed all 3 treatment cycles and had no major protocol violations. Nine patients discontinued treatment early due to consent withdrawal (n=2), early disease progression (n=5), or adverse events (AEs, n=2).
At week 8, 1 patient had achieved a partial response to treatment, 4 patients had stable disease, 5 had progressed, and 4 patients were not evaluable because they had withdrawn from the study.
At week 16, 1 patient had a partial response (not the same patient as at week 8), 4 patients had stable disease, and 9 patients were not evaluable due to withdrawal.
There were a total of 141 AEs, and 77 of these were considered related to APO866. Most patients (n=12) had mild to moderate AEs, but there were 7 serious AEs thought to be treatment-related. These included pyrexia, lymphopenia (n=2), spondylitis, Staphylococcal sepsis, rhabdomyolysis, and thrombocytopenia.
There were 4 deaths, but they were not considered drug-related.
The researchers said these results suggest APO866 should not be pursued as a treatment for CTCL. However, as the drug induces immunosuppression and has insulin-mimicking effects, it might prove useful for treating other conditions.

mycosis fungoides
Results of a phase 2 trial suggest the drug APO866 is not suitable for the treatment of relapsed or refractory cutaneous T-cell lymphoma (CTCL).
Researchers said APO866 had “a reasonable toxic effect,” but it was “not powerful enough,” so the trial was stopped early.
Two of the 14 patients studied achieved a partial response during the trial, but there were no complete responses, and most patients withdrew from the study early.
The researchers described these results in a letter to JAMA Dermatology. The study was initially sponsored by Apoxis SA and later by TopoTarget A/S, which is now known as Onxeo after merging with BioAlliance Pharma.
According to Onxeo, APO866 is an injectable molecule that induces apoptosis by inhibiting the biosynthesis of NAD+ from niacinamide, which is essential for the cellular metabolism, protein modification, and calcium-dependent messenger synthesis.
In the phase 2 trial, researchers tested APO866 in 14 patients with relapsed or refractory CTCL. The patients were 19 to 83 years of age, and half were female.
Eight patients had mycosis fungoides, 3 had Sézary syndrome, 1 had CD30+ anaplastic large-cell lymphoma, 1 had poikilodermic mycosis fungoides, and 1 had CD30- nonepidermotropic CTCL. One patient had stage IB disease, 2 had stage IIA, 3 had stage IIB, and 8 had stage IVA.
The patients received a continuous intravenous infusion, via pump, of APO866 at 0.126 mg/m2/h over the course of 96 hours. Patients received this treatment every 28 days for a total of 3 cycles.
Five patients completed all 3 treatment cycles and had no major protocol violations. Nine patients discontinued treatment early due to consent withdrawal (n=2), early disease progression (n=5), or adverse events (AEs, n=2).
At week 8, 1 patient had achieved a partial response to treatment, 4 patients had stable disease, 5 had progressed, and 4 patients were not evaluable because they had withdrawn from the study.
At week 16, 1 patient had a partial response (not the same patient as at week 8), 4 patients had stable disease, and 9 patients were not evaluable due to withdrawal.
There were a total of 141 AEs, and 77 of these were considered related to APO866. Most patients (n=12) had mild to moderate AEs, but there were 7 serious AEs thought to be treatment-related. These included pyrexia, lymphopenia (n=2), spondylitis, Staphylococcal sepsis, rhabdomyolysis, and thrombocytopenia.
There were 4 deaths, but they were not considered drug-related.
The researchers said these results suggest APO866 should not be pursued as a treatment for CTCL. However, as the drug induces immunosuppression and has insulin-mimicking effects, it might prove useful for treating other conditions.