User login
Flooring poses higher cancer risk than previously reported

while woman looks on
US government agencies have released a revised report on the health risks associated with formaldehyde in certain types of laminate flooring.
The new report corrects a previous error and reveals an increase in the estimated lifetime risk of cancers, including leukemias, for individuals who regularly breathe in formaldehyde from the flooring.
The report also suggests that irritation and breathing problems can result in anyone exposed to the flooring.
The report was compiled by the Agency for Toxic Substances and Disease Registry (ATSDR) and the Centers for Disease Control and Prevention’s National Center for Environmental Health (NCEH).
About the report
On March 1, 2015, the CBS news program 60 Minutes reported that an American company, Lumber Liquidators®, was selling laminate wood flooring produced in China that released elevated levels of formaldehyde.
Based on this allegation, the US Consumer Product Safety Commission (CPSC) tested laminate flooring samples manufactured in China from 2012 to 2014 that were sold at Lumber Liquidators® stores. The CPSC then requested that the NCEH and ATSDR evaluate the test results for possible health effects.
The NCEH and ATSDR published a report detailing the possible health effects on February 10, 2016. But the report was pulled on February 19, 2016, after the agencies were informed that the report’s indoor air model incorporated an incorrect value for ceiling height.
As a result, the health risks were calculated using airborne concentration estimates about 3 times lower than they should have been.
Since the discovery of the error, the NCEH and ATSDR revised the value in the model, conducted a review of the revised results, and re-evaluated the possible health implications.
In addition, the revised report has been reviewed by outside experts and experts from the CPSC, the US Environmental Protection Agency, and the US Department of Housing and Urban Development.
Results
The revised report concludes that irritation and breathing problems could occur in everyone exposed to formaldehyde in the tested flooring.
The previous report suggested such problems might only occur in sensitive groups (eg, children) and people with pre-existing health conditions (eg, asthma).
The new report also increased the estimated lifetime cancer risk from breathing the highest levels of formaldehyde from the affected flooring all day, every day for 2 years.
The previous estimate of lifetime cancer risk was 2 to 9 extra cases for every 100,000 people.
The new estimate is 6 to 30 extra cases per 100,000 people.
There is conflicting data regarding the types of cancers associated with formaldehyde exposure, but many studies have suggested that formaldehyde causes cancer of the nasopharynx, sinuses, and nasal cavity, as well as leukemia, particularly myeloid leukemia.
Recommendations
Although the revised report shows an increase in health risks associated with the flooring, the NCEH and ATSDR said their recommendations remain the same.
They recommend that people with the affected flooring:
- Reduce their exposure to formaldehyde in their homes by opening windows, running exhaust fans, avoiding use of other products containing formaldehyde, etc.
- See a doctor for ongoing health symptoms such as breathing problems or irritation of the eyes, nose, or throat
- Weigh the pros and cons of professional air testing
- Consult a professional before removing the flooring, as removing it may release more formaldehyde into the home.


while woman looks on
US government agencies have released a revised report on the health risks associated with formaldehyde in certain types of laminate flooring.
The new report corrects a previous error and reveals an increase in the estimated lifetime risk of cancers, including leukemias, for individuals who regularly breathe in formaldehyde from the flooring.
The report also suggests that irritation and breathing problems can result in anyone exposed to the flooring.
The report was compiled by the Agency for Toxic Substances and Disease Registry (ATSDR) and the Centers for Disease Control and Prevention’s National Center for Environmental Health (NCEH).
About the report
On March 1, 2015, the CBS news program 60 Minutes reported that an American company, Lumber Liquidators®, was selling laminate wood flooring produced in China that released elevated levels of formaldehyde.
Based on this allegation, the US Consumer Product Safety Commission (CPSC) tested laminate flooring samples manufactured in China from 2012 to 2014 that were sold at Lumber Liquidators® stores. The CPSC then requested that the NCEH and ATSDR evaluate the test results for possible health effects.
The NCEH and ATSDR published a report detailing the possible health effects on February 10, 2016. But the report was pulled on February 19, 2016, after the agencies were informed that the report’s indoor air model incorporated an incorrect value for ceiling height.
As a result, the health risks were calculated using airborne concentration estimates about 3 times lower than they should have been.
Since the discovery of the error, the NCEH and ATSDR revised the value in the model, conducted a review of the revised results, and re-evaluated the possible health implications.
In addition, the revised report has been reviewed by outside experts and experts from the CPSC, the US Environmental Protection Agency, and the US Department of Housing and Urban Development.
Results
The revised report concludes that irritation and breathing problems could occur in everyone exposed to formaldehyde in the tested flooring.
The previous report suggested such problems might only occur in sensitive groups (eg, children) and people with pre-existing health conditions (eg, asthma).
The new report also increased the estimated lifetime cancer risk from breathing the highest levels of formaldehyde from the affected flooring all day, every day for 2 years.
The previous estimate of lifetime cancer risk was 2 to 9 extra cases for every 100,000 people.
The new estimate is 6 to 30 extra cases per 100,000 people.
There is conflicting data regarding the types of cancers associated with formaldehyde exposure, but many studies have suggested that formaldehyde causes cancer of the nasopharynx, sinuses, and nasal cavity, as well as leukemia, particularly myeloid leukemia.
Recommendations
Although the revised report shows an increase in health risks associated with the flooring, the NCEH and ATSDR said their recommendations remain the same.
They recommend that people with the affected flooring:
- Reduce their exposure to formaldehyde in their homes by opening windows, running exhaust fans, avoiding use of other products containing formaldehyde, etc.
- See a doctor for ongoing health symptoms such as breathing problems or irritation of the eyes, nose, or throat
- Weigh the pros and cons of professional air testing
- Consult a professional before removing the flooring, as removing it may release more formaldehyde into the home.


while woman looks on
US government agencies have released a revised report on the health risks associated with formaldehyde in certain types of laminate flooring.
The new report corrects a previous error and reveals an increase in the estimated lifetime risk of cancers, including leukemias, for individuals who regularly breathe in formaldehyde from the flooring.
The report also suggests that irritation and breathing problems can result in anyone exposed to the flooring.
The report was compiled by the Agency for Toxic Substances and Disease Registry (ATSDR) and the Centers for Disease Control and Prevention’s National Center for Environmental Health (NCEH).
About the report
On March 1, 2015, the CBS news program 60 Minutes reported that an American company, Lumber Liquidators®, was selling laminate wood flooring produced in China that released elevated levels of formaldehyde.
Based on this allegation, the US Consumer Product Safety Commission (CPSC) tested laminate flooring samples manufactured in China from 2012 to 2014 that were sold at Lumber Liquidators® stores. The CPSC then requested that the NCEH and ATSDR evaluate the test results for possible health effects.
The NCEH and ATSDR published a report detailing the possible health effects on February 10, 2016. But the report was pulled on February 19, 2016, after the agencies were informed that the report’s indoor air model incorporated an incorrect value for ceiling height.
As a result, the health risks were calculated using airborne concentration estimates about 3 times lower than they should have been.
Since the discovery of the error, the NCEH and ATSDR revised the value in the model, conducted a review of the revised results, and re-evaluated the possible health implications.
In addition, the revised report has been reviewed by outside experts and experts from the CPSC, the US Environmental Protection Agency, and the US Department of Housing and Urban Development.
Results
The revised report concludes that irritation and breathing problems could occur in everyone exposed to formaldehyde in the tested flooring.
The previous report suggested such problems might only occur in sensitive groups (eg, children) and people with pre-existing health conditions (eg, asthma).
The new report also increased the estimated lifetime cancer risk from breathing the highest levels of formaldehyde from the affected flooring all day, every day for 2 years.
The previous estimate of lifetime cancer risk was 2 to 9 extra cases for every 100,000 people.
The new estimate is 6 to 30 extra cases per 100,000 people.
There is conflicting data regarding the types of cancers associated with formaldehyde exposure, but many studies have suggested that formaldehyde causes cancer of the nasopharynx, sinuses, and nasal cavity, as well as leukemia, particularly myeloid leukemia.
Recommendations
Although the revised report shows an increase in health risks associated with the flooring, the NCEH and ATSDR said their recommendations remain the same.
They recommend that people with the affected flooring:
- Reduce their exposure to formaldehyde in their homes by opening windows, running exhaust fans, avoiding use of other products containing formaldehyde, etc.
- See a doctor for ongoing health symptoms such as breathing problems or irritation of the eyes, nose, or throat
- Weigh the pros and cons of professional air testing
- Consult a professional before removing the flooring, as removing it may release more formaldehyde into the home.

Parasite competition influences drug resistance

infecting a red blood cell
Image courtesy of St. Jude
Children’s Research Hospital
Researchers say they have documented how competition among different malaria parasite strains in human hosts could influence the spread of drug resistance.
“We found that when hosts are co-infected with drug-resistant and drug-sensitive strains, both strains are competitively suppressed,” said Mary Bushman, of Emory University in Atlanta, Georgia.
“Antimalarial therapy, by clearing drug-sensitive parasites from mixed infections, may result in competitive release of resistant strains.”
Bushman and her colleagues described these findings in Proceedings of the Royal Society B.
The researchers focused on the parasite Plasmodium falciparum, which has developed resistance to former first-line therapies chloroquine and sulfadoxine-pyrimethamine.
“We’re now down to our last treatment, artemisinin combination therapy, or ACT, and resistance to that recently emerged in Southeast Asia,” Bushman said. “If ACT resistance continues to follow the same pattern, the world may soon be without reliable antimalarial drugs.”
In addition, people infected with P falciparum often have multiple strains of the parasite, especially in high-transmission areas such as sub-Saharan Africa, where infectious mosquito bites occur frequently. Many people have developed partial immunity, making asymptomatic infections common and further complicating control efforts.
With previous work in lab mice, the researchers found that competition between mixed strains of malaria parasites were a crucial determinant to the spread of resistance.
“In the mouse studies, we found that drug-sensitive parasites suppress resistant parasites,” said Jaap de Roode, PhD, of Emory University.
“We also found that by clearing these sensitive parasites with drugs, the resistant parasites had a big advantage, growing up to high numbers and transmitting to mosquitoes at high rates. Ever since doing that work, I have wanted to see if the same could apply to humans.”
To find out, Dr de Roode and his colleagues analyzed 1341 blood samples from untreated children with malaria living in Angola, Ghana, and Tanzania.
The researchers extracted the DNA of malaria parasites from the blood samples and used polymerase chain reaction technology to determine the densities of drug-resistant strains and drug-sensitive ones. About 15% of the samples had mixtures of both types.
Analyses showed that, in mixed-strain infections, densities of chloroquine-sensitive and chloroquine-resistant strains were reduced in the presence of competitors.
The results also showed that, in the absence of chloroquine, the resistant strains had lower densities than sensitive strains.
“The results were really clear cut, which rarely happens in human studies,” Bushman said. “We found almost complete consistency between the 3 data sets [divided by country].”
Bushman added that the tendency is to use a “one-size-fits-all” strategy for controlling malaria, but this research suggests that more tailored approaches are needed.
For example, a strategy of mass drug administration might be effective in a place with a low prevalence of malaria and less likelihood of mixed-strain infections. However, that same strategy might actually boost drug resistance without reducing the burden of disease in areas where most of the population is infected with multiple strains of malaria parasites.
“The epidemiology of malaria infection is different for different places and different conditions,” Bushman said. “We hope that our work will spur development of new strategies to minimize resistance while maximizing the benefits of control measures.”
However, more questions must be answered to guide the development of these new strategies.
“As a first step, we need to determine if the observed suppression of resistance in humans also results in reduced transmission to mosquitoes,” Dr de Roode said.
Another avenue to explore is resistance among patients who have received antimalarial treatment.
“We need to find out if drug treatment of people infected with malaria removes competition and gives resistance a boost, as we have found in mice before,” Dr de Roode noted. ![]()

infecting a red blood cell
Image courtesy of St. Jude
Children’s Research Hospital
Researchers say they have documented how competition among different malaria parasite strains in human hosts could influence the spread of drug resistance.
“We found that when hosts are co-infected with drug-resistant and drug-sensitive strains, both strains are competitively suppressed,” said Mary Bushman, of Emory University in Atlanta, Georgia.
“Antimalarial therapy, by clearing drug-sensitive parasites from mixed infections, may result in competitive release of resistant strains.”
Bushman and her colleagues described these findings in Proceedings of the Royal Society B.
The researchers focused on the parasite Plasmodium falciparum, which has developed resistance to former first-line therapies chloroquine and sulfadoxine-pyrimethamine.
“We’re now down to our last treatment, artemisinin combination therapy, or ACT, and resistance to that recently emerged in Southeast Asia,” Bushman said. “If ACT resistance continues to follow the same pattern, the world may soon be without reliable antimalarial drugs.”
In addition, people infected with P falciparum often have multiple strains of the parasite, especially in high-transmission areas such as sub-Saharan Africa, where infectious mosquito bites occur frequently. Many people have developed partial immunity, making asymptomatic infections common and further complicating control efforts.
With previous work in lab mice, the researchers found that competition between mixed strains of malaria parasites were a crucial determinant to the spread of resistance.
“In the mouse studies, we found that drug-sensitive parasites suppress resistant parasites,” said Jaap de Roode, PhD, of Emory University.
“We also found that by clearing these sensitive parasites with drugs, the resistant parasites had a big advantage, growing up to high numbers and transmitting to mosquitoes at high rates. Ever since doing that work, I have wanted to see if the same could apply to humans.”
To find out, Dr de Roode and his colleagues analyzed 1341 blood samples from untreated children with malaria living in Angola, Ghana, and Tanzania.
The researchers extracted the DNA of malaria parasites from the blood samples and used polymerase chain reaction technology to determine the densities of drug-resistant strains and drug-sensitive ones. About 15% of the samples had mixtures of both types.
Analyses showed that, in mixed-strain infections, densities of chloroquine-sensitive and chloroquine-resistant strains were reduced in the presence of competitors.
The results also showed that, in the absence of chloroquine, the resistant strains had lower densities than sensitive strains.
“The results were really clear cut, which rarely happens in human studies,” Bushman said. “We found almost complete consistency between the 3 data sets [divided by country].”
Bushman added that the tendency is to use a “one-size-fits-all” strategy for controlling malaria, but this research suggests that more tailored approaches are needed.
For example, a strategy of mass drug administration might be effective in a place with a low prevalence of malaria and less likelihood of mixed-strain infections. However, that same strategy might actually boost drug resistance without reducing the burden of disease in areas where most of the population is infected with multiple strains of malaria parasites.
“The epidemiology of malaria infection is different for different places and different conditions,” Bushman said. “We hope that our work will spur development of new strategies to minimize resistance while maximizing the benefits of control measures.”
However, more questions must be answered to guide the development of these new strategies.
“As a first step, we need to determine if the observed suppression of resistance in humans also results in reduced transmission to mosquitoes,” Dr de Roode said.
Another avenue to explore is resistance among patients who have received antimalarial treatment.
“We need to find out if drug treatment of people infected with malaria removes competition and gives resistance a boost, as we have found in mice before,” Dr de Roode noted. ![]()

infecting a red blood cell
Image courtesy of St. Jude
Children’s Research Hospital
Researchers say they have documented how competition among different malaria parasite strains in human hosts could influence the spread of drug resistance.
“We found that when hosts are co-infected with drug-resistant and drug-sensitive strains, both strains are competitively suppressed,” said Mary Bushman, of Emory University in Atlanta, Georgia.
“Antimalarial therapy, by clearing drug-sensitive parasites from mixed infections, may result in competitive release of resistant strains.”
Bushman and her colleagues described these findings in Proceedings of the Royal Society B.
The researchers focused on the parasite Plasmodium falciparum, which has developed resistance to former first-line therapies chloroquine and sulfadoxine-pyrimethamine.
“We’re now down to our last treatment, artemisinin combination therapy, or ACT, and resistance to that recently emerged in Southeast Asia,” Bushman said. “If ACT resistance continues to follow the same pattern, the world may soon be without reliable antimalarial drugs.”
In addition, people infected with P falciparum often have multiple strains of the parasite, especially in high-transmission areas such as sub-Saharan Africa, where infectious mosquito bites occur frequently. Many people have developed partial immunity, making asymptomatic infections common and further complicating control efforts.
With previous work in lab mice, the researchers found that competition between mixed strains of malaria parasites were a crucial determinant to the spread of resistance.
“In the mouse studies, we found that drug-sensitive parasites suppress resistant parasites,” said Jaap de Roode, PhD, of Emory University.
“We also found that by clearing these sensitive parasites with drugs, the resistant parasites had a big advantage, growing up to high numbers and transmitting to mosquitoes at high rates. Ever since doing that work, I have wanted to see if the same could apply to humans.”
To find out, Dr de Roode and his colleagues analyzed 1341 blood samples from untreated children with malaria living in Angola, Ghana, and Tanzania.
The researchers extracted the DNA of malaria parasites from the blood samples and used polymerase chain reaction technology to determine the densities of drug-resistant strains and drug-sensitive ones. About 15% of the samples had mixtures of both types.
Analyses showed that, in mixed-strain infections, densities of chloroquine-sensitive and chloroquine-resistant strains were reduced in the presence of competitors.
The results also showed that, in the absence of chloroquine, the resistant strains had lower densities than sensitive strains.
“The results were really clear cut, which rarely happens in human studies,” Bushman said. “We found almost complete consistency between the 3 data sets [divided by country].”
Bushman added that the tendency is to use a “one-size-fits-all” strategy for controlling malaria, but this research suggests that more tailored approaches are needed.
For example, a strategy of mass drug administration might be effective in a place with a low prevalence of malaria and less likelihood of mixed-strain infections. However, that same strategy might actually boost drug resistance without reducing the burden of disease in areas where most of the population is infected with multiple strains of malaria parasites.
“The epidemiology of malaria infection is different for different places and different conditions,” Bushman said. “We hope that our work will spur development of new strategies to minimize resistance while maximizing the benefits of control measures.”
However, more questions must be answered to guide the development of these new strategies.
“As a first step, we need to determine if the observed suppression of resistance in humans also results in reduced transmission to mosquitoes,” Dr de Roode said.
Another avenue to explore is resistance among patients who have received antimalarial treatment.
“We need to find out if drug treatment of people infected with malaria removes competition and gives resistance a boost, as we have found in mice before,” Dr de Roode noted. ![]()
Studies explain how mutations promote lymphoma

New research suggests there are 4 different “locks” that keep the CARD11 protein in check.
Researchers say these locks, also known as “repressive elements,” work together to prevent unwarranted CARD11 signaling.
But mutations in CARD11 can perturb or bypass the action of the repressive elements, which leads to a level of hyperactive CARD11 signaling that supports lymphoma growth.
Joel Pomerantz, PhD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland, and his colleagues reported these findings in 2 articles published in The Journal of Biological Chemistry.1,2
The researchers noted that mutated CARD11 proteins are sometimes found in patients with lymphoma, particularly diffuse large B-cell lymphoma. But none of the mutations exist in the “lock region” of CARD11, known as the autoinhibitory domain.
To find out why, the team conducted experiments with T cells. They first genetically deleted CARD11’s autoinhibitory domain so that the protein was always “on” and signaling.
Then, to figure out which region of the autoinhibitory domain accounted for its function, the researchers systematically deleted 6 different segments of it one at a time, rather than deleting the whole thing.
They expected most of the deletions to keep CARD11 inactive and 1 or 2 to “unlock” it. However, none of the deletions unlocked it, which suggested there was more than 1 repressive element within the autoinhibitory domain.
To see if that was the case and to find out how many repressive elements there might be, the researchers deleted the full autoinhibitory domain again, then added back small regions of it.
In this way, they pieced together the presence of 4 different biochemical regions, or repressive elements, that are each capable of locking CARD11 on their own.
“Having 4 redundant repressive elements seems to explain why patients with lymphoma don’t have mutations in CARD11’s autoinhibitory domain,” Dr Pomerantz said. “A mutation in any one of the repressive elements would only unlock 1 of the 4 locks, keeping CARD11’s signaling under the control of the other 3.”
So where do the lymphoma-associated mutations occur, and how do they circumvent CARD11’s quadruple locks? Clinical data show that the mutations occur in 3 other regions of CARD11—the CARD, LATCH, and coiled-coil regions.
Dr Pomerantz and his colleagues conducted biochemical tests on T cells and found that those 3 regions clasp the autoinhibitory domain to keep CARD11’s activity on lockdown. This makes sense, Dr Pomerantz said, because CARD11 uses those same regions to connect with other signaling proteins to send its message.
“When they are interacting with the autoinhibitory domain, they can’t be interacting with other proteins,” he said.
According to Dr Pomerantz, single mutations in the CARD, LATCH, or coiled-coil regions appear to be sufficient to disable the 4 repressive elements. And the ultimate goal is to advance the development of therapies that rein in hyperactive CARD11 in patients with lymphoma.
“If we can understand how CARD11 is normally kept off, we might be able to mimic that with a drug,” he said. “We also hope to shed light on how different mutations in CARD11 affect its function and a patient’s prognosis.” ![]()

New research suggests there are 4 different “locks” that keep the CARD11 protein in check.
Researchers say these locks, also known as “repressive elements,” work together to prevent unwarranted CARD11 signaling.
But mutations in CARD11 can perturb or bypass the action of the repressive elements, which leads to a level of hyperactive CARD11 signaling that supports lymphoma growth.
Joel Pomerantz, PhD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland, and his colleagues reported these findings in 2 articles published in The Journal of Biological Chemistry.1,2
The researchers noted that mutated CARD11 proteins are sometimes found in patients with lymphoma, particularly diffuse large B-cell lymphoma. But none of the mutations exist in the “lock region” of CARD11, known as the autoinhibitory domain.
To find out why, the team conducted experiments with T cells. They first genetically deleted CARD11’s autoinhibitory domain so that the protein was always “on” and signaling.
Then, to figure out which region of the autoinhibitory domain accounted for its function, the researchers systematically deleted 6 different segments of it one at a time, rather than deleting the whole thing.
They expected most of the deletions to keep CARD11 inactive and 1 or 2 to “unlock” it. However, none of the deletions unlocked it, which suggested there was more than 1 repressive element within the autoinhibitory domain.
To see if that was the case and to find out how many repressive elements there might be, the researchers deleted the full autoinhibitory domain again, then added back small regions of it.
In this way, they pieced together the presence of 4 different biochemical regions, or repressive elements, that are each capable of locking CARD11 on their own.
“Having 4 redundant repressive elements seems to explain why patients with lymphoma don’t have mutations in CARD11’s autoinhibitory domain,” Dr Pomerantz said. “A mutation in any one of the repressive elements would only unlock 1 of the 4 locks, keeping CARD11’s signaling under the control of the other 3.”
So where do the lymphoma-associated mutations occur, and how do they circumvent CARD11’s quadruple locks? Clinical data show that the mutations occur in 3 other regions of CARD11—the CARD, LATCH, and coiled-coil regions.
Dr Pomerantz and his colleagues conducted biochemical tests on T cells and found that those 3 regions clasp the autoinhibitory domain to keep CARD11’s activity on lockdown. This makes sense, Dr Pomerantz said, because CARD11 uses those same regions to connect with other signaling proteins to send its message.
“When they are interacting with the autoinhibitory domain, they can’t be interacting with other proteins,” he said.
According to Dr Pomerantz, single mutations in the CARD, LATCH, or coiled-coil regions appear to be sufficient to disable the 4 repressive elements. And the ultimate goal is to advance the development of therapies that rein in hyperactive CARD11 in patients with lymphoma.
“If we can understand how CARD11 is normally kept off, we might be able to mimic that with a drug,” he said. “We also hope to shed light on how different mutations in CARD11 affect its function and a patient’s prognosis.” ![]()

New research suggests there are 4 different “locks” that keep the CARD11 protein in check.
Researchers say these locks, also known as “repressive elements,” work together to prevent unwarranted CARD11 signaling.
But mutations in CARD11 can perturb or bypass the action of the repressive elements, which leads to a level of hyperactive CARD11 signaling that supports lymphoma growth.
Joel Pomerantz, PhD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland, and his colleagues reported these findings in 2 articles published in The Journal of Biological Chemistry.1,2
The researchers noted that mutated CARD11 proteins are sometimes found in patients with lymphoma, particularly diffuse large B-cell lymphoma. But none of the mutations exist in the “lock region” of CARD11, known as the autoinhibitory domain.
To find out why, the team conducted experiments with T cells. They first genetically deleted CARD11’s autoinhibitory domain so that the protein was always “on” and signaling.
Then, to figure out which region of the autoinhibitory domain accounted for its function, the researchers systematically deleted 6 different segments of it one at a time, rather than deleting the whole thing.
They expected most of the deletions to keep CARD11 inactive and 1 or 2 to “unlock” it. However, none of the deletions unlocked it, which suggested there was more than 1 repressive element within the autoinhibitory domain.
To see if that was the case and to find out how many repressive elements there might be, the researchers deleted the full autoinhibitory domain again, then added back small regions of it.
In this way, they pieced together the presence of 4 different biochemical regions, or repressive elements, that are each capable of locking CARD11 on their own.
“Having 4 redundant repressive elements seems to explain why patients with lymphoma don’t have mutations in CARD11’s autoinhibitory domain,” Dr Pomerantz said. “A mutation in any one of the repressive elements would only unlock 1 of the 4 locks, keeping CARD11’s signaling under the control of the other 3.”
So where do the lymphoma-associated mutations occur, and how do they circumvent CARD11’s quadruple locks? Clinical data show that the mutations occur in 3 other regions of CARD11—the CARD, LATCH, and coiled-coil regions.
Dr Pomerantz and his colleagues conducted biochemical tests on T cells and found that those 3 regions clasp the autoinhibitory domain to keep CARD11’s activity on lockdown. This makes sense, Dr Pomerantz said, because CARD11 uses those same regions to connect with other signaling proteins to send its message.
“When they are interacting with the autoinhibitory domain, they can’t be interacting with other proteins,” he said.
According to Dr Pomerantz, single mutations in the CARD, LATCH, or coiled-coil regions appear to be sufficient to disable the 4 repressive elements. And the ultimate goal is to advance the development of therapies that rein in hyperactive CARD11 in patients with lymphoma.
“If we can understand how CARD11 is normally kept off, we might be able to mimic that with a drug,” he said. “We also hope to shed light on how different mutations in CARD11 affect its function and a patient’s prognosis.” ![]()
Survey reveals HSCT trends in Europe

Photo by Chad McNeeley
Results from the 2014 Transplant Activity Survey suggest the use of hematopoietic stem cell transplant (HSCT) is still on the rise in Europe.
The European Society for Blood and Marrow Transplantation (EBMT) introduced this survey in 1990 to assess the use of HSCT in Europe.
Every year, all EBMT members and affiliated teams report their number of transplant patients by indication, donor type, and stem cell source.
Results from the 2014 survey revealed that 40,829 HSCTs were conducted in 36,469 patients at 656 centers in 47 countries. Fifty-seven percent of these transplants were autologous (n=20,704), and 43% were allogeneic (n=15,765).
When compared to data from the 2013 survey, the total number of transplants increased by 4.1%—4.5% for allogeneic HSCT and 3.8% for autologous HSCT.
The greatest increases in allogeneic HSCT occurred in Romania, Russia, Turkey, Croatia, Lithuania, and Serbia. And the greatest increases for autologous HSCT occurred in Romania, Serbia, Russia, Turkey, and Iran.
The most common indication for HSCT in 2014 was lymphoid neoplasias (57%, n=20,802), followed by leukemias (33%, n=11,853), non-malignant disorders (6%, n=2203), and solid tumors (4%, n=1458).
Compared to data from 2013, there was an increase in allogeneic HSCT for a few indications. There was a 13% increase in allogeneic HSCT for acute myeloid leukemia in first complete remission, a 14% increase for myeloproliferative neoplasms, and a 12% increase for severe aplastic anemia.
For autologous HSCT, there was a 21% decrease for chronic lymphocytic leukemia, a 5% increase for myeloma, a 44% increase for amyloidosis, an 8% increase for Hodgkin lymphoma, and a 40% increase for autoimmune diseases.
For more details, see the full report in Bone Marrow Transplantation. ![]()

Photo by Chad McNeeley
Results from the 2014 Transplant Activity Survey suggest the use of hematopoietic stem cell transplant (HSCT) is still on the rise in Europe.
The European Society for Blood and Marrow Transplantation (EBMT) introduced this survey in 1990 to assess the use of HSCT in Europe.
Every year, all EBMT members and affiliated teams report their number of transplant patients by indication, donor type, and stem cell source.
Results from the 2014 survey revealed that 40,829 HSCTs were conducted in 36,469 patients at 656 centers in 47 countries. Fifty-seven percent of these transplants were autologous (n=20,704), and 43% were allogeneic (n=15,765).
When compared to data from the 2013 survey, the total number of transplants increased by 4.1%—4.5% for allogeneic HSCT and 3.8% for autologous HSCT.
The greatest increases in allogeneic HSCT occurred in Romania, Russia, Turkey, Croatia, Lithuania, and Serbia. And the greatest increases for autologous HSCT occurred in Romania, Serbia, Russia, Turkey, and Iran.
The most common indication for HSCT in 2014 was lymphoid neoplasias (57%, n=20,802), followed by leukemias (33%, n=11,853), non-malignant disorders (6%, n=2203), and solid tumors (4%, n=1458).
Compared to data from 2013, there was an increase in allogeneic HSCT for a few indications. There was a 13% increase in allogeneic HSCT for acute myeloid leukemia in first complete remission, a 14% increase for myeloproliferative neoplasms, and a 12% increase for severe aplastic anemia.
For autologous HSCT, there was a 21% decrease for chronic lymphocytic leukemia, a 5% increase for myeloma, a 44% increase for amyloidosis, an 8% increase for Hodgkin lymphoma, and a 40% increase for autoimmune diseases.
For more details, see the full report in Bone Marrow Transplantation. ![]()

Photo by Chad McNeeley
Results from the 2014 Transplant Activity Survey suggest the use of hematopoietic stem cell transplant (HSCT) is still on the rise in Europe.
The European Society for Blood and Marrow Transplantation (EBMT) introduced this survey in 1990 to assess the use of HSCT in Europe.
Every year, all EBMT members and affiliated teams report their number of transplant patients by indication, donor type, and stem cell source.
Results from the 2014 survey revealed that 40,829 HSCTs were conducted in 36,469 patients at 656 centers in 47 countries. Fifty-seven percent of these transplants were autologous (n=20,704), and 43% were allogeneic (n=15,765).
When compared to data from the 2013 survey, the total number of transplants increased by 4.1%—4.5% for allogeneic HSCT and 3.8% for autologous HSCT.
The greatest increases in allogeneic HSCT occurred in Romania, Russia, Turkey, Croatia, Lithuania, and Serbia. And the greatest increases for autologous HSCT occurred in Romania, Serbia, Russia, Turkey, and Iran.
The most common indication for HSCT in 2014 was lymphoid neoplasias (57%, n=20,802), followed by leukemias (33%, n=11,853), non-malignant disorders (6%, n=2203), and solid tumors (4%, n=1458).
Compared to data from 2013, there was an increase in allogeneic HSCT for a few indications. There was a 13% increase in allogeneic HSCT for acute myeloid leukemia in first complete remission, a 14% increase for myeloproliferative neoplasms, and a 12% increase for severe aplastic anemia.
For autologous HSCT, there was a 21% decrease for chronic lymphocytic leukemia, a 5% increase for myeloma, a 44% increase for amyloidosis, an 8% increase for Hodgkin lymphoma, and a 40% increase for autoimmune diseases.
For more details, see the full report in Bone Marrow Transplantation. ![]()
Use of dangerous drug combos on the rise

Photo by Rhoda Baer
A new study suggests that older adults in the US may be using potentially deadly combinations of prescription drugs, over-the-counter medications, and dietary supplements.
Investigators observed a significant increase in the proportion of adults ages 62 to 85 who were at risk of major drug-drug interactions—from 8% in 2005-2006 to 15% in 2010-2011.
Most of the combinations consisted of preventative cardiovascular medications such as antiplatelet drugs, statins, and omega-3 fish oil supplements.
Dima Mazen Qato, PharmD, of the University of Illinois at Chicago, and her colleagues reported these results in JAMA Internal Medicine.
The investigators examined changes in medication use in a nationally representative sample of adults between the ages of 62 and 85. The team conducted in-home interviews to accurately identify medications. They interviewed 2351 subjects in 2005-2006 and 2206 subjects in 2010-2011.
The results showed a significant increase in the proportion of subjects taking at least 1 prescription medication—from 84.1% in 2005-2006 to 87.7% in 2010-2011 (P=0.003).
The proportion of subjects taking at least 5 prescription medications rose significantly as well—from 30.6% to 35.8% (P=0.02).
The investigators said factors that may account for these increases include the implementation of Medicare Part D, changes in treatment guidelines, and the increased availability of generics for many commonly used drugs.
As an example, use of the statin simvastatin (Zocor)—which became available as a generic in 2006—rose from 10.3% in 2005-2006 to 22.5% in 2010-2011.
The use of over-the-counter medications declined from 44.4% in 2005-2006 to 37.9% in 2010-2011 (P<0.001), while the use of dietary supplements increased from 51.8% to 63.7% (P<0.001).
Drug-drug interactions
The investigators identified 15 potentially life-threatening combinations of the most commonly used medications and supplements in the study.
They found that 15.1% of subjects were at risk for a potential major drug-drug interaction in 2010-2011, compared with an estimated 8.4% in 2005-2006 (P<0.001).
Dr Qato noted that more than half of the potential interactions involved a nonprescription medication or dietary supplement.
Preventative cardiovascular medications such as statins (particularly, simvastatin), antiplatelet drugs (such as clopidogrel and aspirin), and supplements (specifically, omega-3 fish oil) accounted for the vast majority of these combinations.
“Many older patients seeking to improve their cardiovascular health are also regularly using interacting drug combinations that may worsen cardiovascular risk,” Dr Qato said.
“For example, the use of clopidogrel in combination with the proton-pump inhibitor omeprazole, aspirin, or naproxen—all over-the-counter medications—is associated with an increased risk of heart attacks, bleeding complications, or death. However, about 1.8%—or 1 million—older adults regularly use clopidogrel in interacting combinations.”
Dr Qato added that healthcare professionals should consider the adverse effects of commonly used prescription and nonprescription medication combinations when treating older adults and counsel patients about the risks.
“Improving safety in the use of interacting medication combinations has the potential to reduce preventable, potentially fatal, adverse drug events,” she said. ![]()

Photo by Rhoda Baer
A new study suggests that older adults in the US may be using potentially deadly combinations of prescription drugs, over-the-counter medications, and dietary supplements.
Investigators observed a significant increase in the proportion of adults ages 62 to 85 who were at risk of major drug-drug interactions—from 8% in 2005-2006 to 15% in 2010-2011.
Most of the combinations consisted of preventative cardiovascular medications such as antiplatelet drugs, statins, and omega-3 fish oil supplements.
Dima Mazen Qato, PharmD, of the University of Illinois at Chicago, and her colleagues reported these results in JAMA Internal Medicine.
The investigators examined changes in medication use in a nationally representative sample of adults between the ages of 62 and 85. The team conducted in-home interviews to accurately identify medications. They interviewed 2351 subjects in 2005-2006 and 2206 subjects in 2010-2011.
The results showed a significant increase in the proportion of subjects taking at least 1 prescription medication—from 84.1% in 2005-2006 to 87.7% in 2010-2011 (P=0.003).
The proportion of subjects taking at least 5 prescription medications rose significantly as well—from 30.6% to 35.8% (P=0.02).
The investigators said factors that may account for these increases include the implementation of Medicare Part D, changes in treatment guidelines, and the increased availability of generics for many commonly used drugs.
As an example, use of the statin simvastatin (Zocor)—which became available as a generic in 2006—rose from 10.3% in 2005-2006 to 22.5% in 2010-2011.
The use of over-the-counter medications declined from 44.4% in 2005-2006 to 37.9% in 2010-2011 (P<0.001), while the use of dietary supplements increased from 51.8% to 63.7% (P<0.001).
Drug-drug interactions
The investigators identified 15 potentially life-threatening combinations of the most commonly used medications and supplements in the study.
They found that 15.1% of subjects were at risk for a potential major drug-drug interaction in 2010-2011, compared with an estimated 8.4% in 2005-2006 (P<0.001).
Dr Qato noted that more than half of the potential interactions involved a nonprescription medication or dietary supplement.
Preventative cardiovascular medications such as statins (particularly, simvastatin), antiplatelet drugs (such as clopidogrel and aspirin), and supplements (specifically, omega-3 fish oil) accounted for the vast majority of these combinations.
“Many older patients seeking to improve their cardiovascular health are also regularly using interacting drug combinations that may worsen cardiovascular risk,” Dr Qato said.
“For example, the use of clopidogrel in combination with the proton-pump inhibitor omeprazole, aspirin, or naproxen—all over-the-counter medications—is associated with an increased risk of heart attacks, bleeding complications, or death. However, about 1.8%—or 1 million—older adults regularly use clopidogrel in interacting combinations.”
Dr Qato added that healthcare professionals should consider the adverse effects of commonly used prescription and nonprescription medication combinations when treating older adults and counsel patients about the risks.
“Improving safety in the use of interacting medication combinations has the potential to reduce preventable, potentially fatal, adverse drug events,” she said. ![]()

Photo by Rhoda Baer
A new study suggests that older adults in the US may be using potentially deadly combinations of prescription drugs, over-the-counter medications, and dietary supplements.
Investigators observed a significant increase in the proportion of adults ages 62 to 85 who were at risk of major drug-drug interactions—from 8% in 2005-2006 to 15% in 2010-2011.
Most of the combinations consisted of preventative cardiovascular medications such as antiplatelet drugs, statins, and omega-3 fish oil supplements.
Dima Mazen Qato, PharmD, of the University of Illinois at Chicago, and her colleagues reported these results in JAMA Internal Medicine.
The investigators examined changes in medication use in a nationally representative sample of adults between the ages of 62 and 85. The team conducted in-home interviews to accurately identify medications. They interviewed 2351 subjects in 2005-2006 and 2206 subjects in 2010-2011.
The results showed a significant increase in the proportion of subjects taking at least 1 prescription medication—from 84.1% in 2005-2006 to 87.7% in 2010-2011 (P=0.003).
The proportion of subjects taking at least 5 prescription medications rose significantly as well—from 30.6% to 35.8% (P=0.02).
The investigators said factors that may account for these increases include the implementation of Medicare Part D, changes in treatment guidelines, and the increased availability of generics for many commonly used drugs.
As an example, use of the statin simvastatin (Zocor)—which became available as a generic in 2006—rose from 10.3% in 2005-2006 to 22.5% in 2010-2011.
The use of over-the-counter medications declined from 44.4% in 2005-2006 to 37.9% in 2010-2011 (P<0.001), while the use of dietary supplements increased from 51.8% to 63.7% (P<0.001).
Drug-drug interactions
The investigators identified 15 potentially life-threatening combinations of the most commonly used medications and supplements in the study.
They found that 15.1% of subjects were at risk for a potential major drug-drug interaction in 2010-2011, compared with an estimated 8.4% in 2005-2006 (P<0.001).
Dr Qato noted that more than half of the potential interactions involved a nonprescription medication or dietary supplement.
Preventative cardiovascular medications such as statins (particularly, simvastatin), antiplatelet drugs (such as clopidogrel and aspirin), and supplements (specifically, omega-3 fish oil) accounted for the vast majority of these combinations.
“Many older patients seeking to improve their cardiovascular health are also regularly using interacting drug combinations that may worsen cardiovascular risk,” Dr Qato said.
“For example, the use of clopidogrel in combination with the proton-pump inhibitor omeprazole, aspirin, or naproxen—all over-the-counter medications—is associated with an increased risk of heart attacks, bleeding complications, or death. However, about 1.8%—or 1 million—older adults regularly use clopidogrel in interacting combinations.”
Dr Qato added that healthcare professionals should consider the adverse effects of commonly used prescription and nonprescription medication combinations when treating older adults and counsel patients about the risks.
“Improving safety in the use of interacting medication combinations has the potential to reduce preventable, potentially fatal, adverse drug events,” she said. ![]()
ROCK2 inhibitor treats chronic GVHD in mice

Preclinical data suggest that blocking the Rho-associated coiled-coil kinase 2 (ROCK2) signaling pathway with the ROCK2 inhibitor KD025 can ameliorate chronic graft-versus-host disease (cGVHD).
Previous studies have shown that IL-21 and IL-17, 2 pro-inflammatory cytokines regulated by the ROCK2 signaling pathway, play a key role in cGVHD pathogenesis.
In the new study, targeted ROCK2 inhibition with KD025 had a therapeutic effect on cGVHD cytokines and signaling pathways involved in the pathogenesis of cGVHD.
Bruce R. Blazar, MD, of the University of Minnesota in Minneapolis, and his colleagues reported these findings in Blood.
The research was supported by Kadmon Corporation, LLC, the company developing KD025, as well as other sources.
The researchers found that KD025 reversed the clinical and immunological symptoms of cGVHD in 2 murine models: a full major histocompatibility complex (MHC)-mismatch model of multi-organ system cGVHD with bronchiolitis obliterans syndrome and a minor MHC-mismatch model of sclerodermatous GVHD.
Mice treated with KD025 showed improvements in pulmonary function and had a significant decrease in cGVHD pathology in the lung, liver, and spleen, when compared to vehicle-treated animals.
In addition, KD025 treatment did not affect the graft-versus-leukemia effect or immune response to viral pathogens.
In both animal models, KD025 treatment downregulated phosphorylation of STAT3, a pro-inflammatory signaling pathway that plays a key role in the development of cGVHD.
In human cells, KD025 inhibited the production of IL-21, IL-17, and IFN-γ. The drug also decreased STAT3 activity.
In both murine and human cells, KD025 downregulated STAT3 phosphorylation and simultaneously upregulated STAT5 phosphorylation, thereby reducing cGVHD progression.
“These data demonstrate that ROCK2 inhibition decreases cGVHD manifestation in murine and human models, which correlates with a demonstrated effect on molecular signaling pathways responsible for cGVHD pathogenesis,” Dr Blazar said.
Based on these results, Kadmon is planning to initiate a phase 2 study of KD025 for the treatment of cGVHD in mid-2016. The drug has already been tested in phase 1 studies of healthy volunteers. ![]()

Preclinical data suggest that blocking the Rho-associated coiled-coil kinase 2 (ROCK2) signaling pathway with the ROCK2 inhibitor KD025 can ameliorate chronic graft-versus-host disease (cGVHD).
Previous studies have shown that IL-21 and IL-17, 2 pro-inflammatory cytokines regulated by the ROCK2 signaling pathway, play a key role in cGVHD pathogenesis.
In the new study, targeted ROCK2 inhibition with KD025 had a therapeutic effect on cGVHD cytokines and signaling pathways involved in the pathogenesis of cGVHD.
Bruce R. Blazar, MD, of the University of Minnesota in Minneapolis, and his colleagues reported these findings in Blood.
The research was supported by Kadmon Corporation, LLC, the company developing KD025, as well as other sources.
The researchers found that KD025 reversed the clinical and immunological symptoms of cGVHD in 2 murine models: a full major histocompatibility complex (MHC)-mismatch model of multi-organ system cGVHD with bronchiolitis obliterans syndrome and a minor MHC-mismatch model of sclerodermatous GVHD.
Mice treated with KD025 showed improvements in pulmonary function and had a significant decrease in cGVHD pathology in the lung, liver, and spleen, when compared to vehicle-treated animals.
In addition, KD025 treatment did not affect the graft-versus-leukemia effect or immune response to viral pathogens.
In both animal models, KD025 treatment downregulated phosphorylation of STAT3, a pro-inflammatory signaling pathway that plays a key role in the development of cGVHD.
In human cells, KD025 inhibited the production of IL-21, IL-17, and IFN-γ. The drug also decreased STAT3 activity.
In both murine and human cells, KD025 downregulated STAT3 phosphorylation and simultaneously upregulated STAT5 phosphorylation, thereby reducing cGVHD progression.
“These data demonstrate that ROCK2 inhibition decreases cGVHD manifestation in murine and human models, which correlates with a demonstrated effect on molecular signaling pathways responsible for cGVHD pathogenesis,” Dr Blazar said.
Based on these results, Kadmon is planning to initiate a phase 2 study of KD025 for the treatment of cGVHD in mid-2016. The drug has already been tested in phase 1 studies of healthy volunteers. ![]()

Preclinical data suggest that blocking the Rho-associated coiled-coil kinase 2 (ROCK2) signaling pathway with the ROCK2 inhibitor KD025 can ameliorate chronic graft-versus-host disease (cGVHD).
Previous studies have shown that IL-21 and IL-17, 2 pro-inflammatory cytokines regulated by the ROCK2 signaling pathway, play a key role in cGVHD pathogenesis.
In the new study, targeted ROCK2 inhibition with KD025 had a therapeutic effect on cGVHD cytokines and signaling pathways involved in the pathogenesis of cGVHD.
Bruce R. Blazar, MD, of the University of Minnesota in Minneapolis, and his colleagues reported these findings in Blood.
The research was supported by Kadmon Corporation, LLC, the company developing KD025, as well as other sources.
The researchers found that KD025 reversed the clinical and immunological symptoms of cGVHD in 2 murine models: a full major histocompatibility complex (MHC)-mismatch model of multi-organ system cGVHD with bronchiolitis obliterans syndrome and a minor MHC-mismatch model of sclerodermatous GVHD.
Mice treated with KD025 showed improvements in pulmonary function and had a significant decrease in cGVHD pathology in the lung, liver, and spleen, when compared to vehicle-treated animals.
In addition, KD025 treatment did not affect the graft-versus-leukemia effect or immune response to viral pathogens.
In both animal models, KD025 treatment downregulated phosphorylation of STAT3, a pro-inflammatory signaling pathway that plays a key role in the development of cGVHD.
In human cells, KD025 inhibited the production of IL-21, IL-17, and IFN-γ. The drug also decreased STAT3 activity.
In both murine and human cells, KD025 downregulated STAT3 phosphorylation and simultaneously upregulated STAT5 phosphorylation, thereby reducing cGVHD progression.
“These data demonstrate that ROCK2 inhibition decreases cGVHD manifestation in murine and human models, which correlates with a demonstrated effect on molecular signaling pathways responsible for cGVHD pathogenesis,” Dr Blazar said.
Based on these results, Kadmon is planning to initiate a phase 2 study of KD025 for the treatment of cGVHD in mid-2016. The drug has already been tested in phase 1 studies of healthy volunteers. ![]()
Increasing metabolite levels may prevent GVHD

Photo by Aaron Logan
Increasing levels of a gut microbiome-derived metabolite could help prevent graft-versus-host disease (GVHD) after hematopoietic stem cell transplant (HSCT), according to preclinical research published in Nature Immunology.
For this study, researchers searched for alterations in the gut microbiome to see whether metabolites could impact outcomes after HSCT.
They found that levels of a metabolite called butyrate were significantly reduced in the intestinal tract of mice that received a transplant.
When the researchers increased butyrate levels in these mice, they saw a decrease in the incidence and severity of GVHD.
“Our findings suggest we can prevent graft-vs-host disease by bolstering the amount of the microbiome-derived metabolite butyrate,” said study author Pavan Reddy, MD, of the University of Michigan Comprehensive Cancer Center in Ann Arbor.
He and his colleagues found they could mitigate GVHD by administering butyrate to mice or by altering the composition of indigenous gastrointestinal microbiota so they produced high levels of butyrate.
The team noted that diet is another way to control butyrate production by the gut microbiome—specifically, through resistant starch such as that found in potatoes.
The researchers hope to begin a clinical trial later this year in which they will combine resistant starch with current methods of preventing GVHD to assess whether they can increase butyrate and reduce the incidence and severity of GVHD in HSCT recipients.
“This is a whole new approach,” Dr Reddy said. “The idea is to make the host cells stronger, to be able to withstand the assault of the donor immune cells while reducing the risk of infection or [disease] relapse.” ![]()

Photo by Aaron Logan
Increasing levels of a gut microbiome-derived metabolite could help prevent graft-versus-host disease (GVHD) after hematopoietic stem cell transplant (HSCT), according to preclinical research published in Nature Immunology.
For this study, researchers searched for alterations in the gut microbiome to see whether metabolites could impact outcomes after HSCT.
They found that levels of a metabolite called butyrate were significantly reduced in the intestinal tract of mice that received a transplant.
When the researchers increased butyrate levels in these mice, they saw a decrease in the incidence and severity of GVHD.
“Our findings suggest we can prevent graft-vs-host disease by bolstering the amount of the microbiome-derived metabolite butyrate,” said study author Pavan Reddy, MD, of the University of Michigan Comprehensive Cancer Center in Ann Arbor.
He and his colleagues found they could mitigate GVHD by administering butyrate to mice or by altering the composition of indigenous gastrointestinal microbiota so they produced high levels of butyrate.
The team noted that diet is another way to control butyrate production by the gut microbiome—specifically, through resistant starch such as that found in potatoes.
The researchers hope to begin a clinical trial later this year in which they will combine resistant starch with current methods of preventing GVHD to assess whether they can increase butyrate and reduce the incidence and severity of GVHD in HSCT recipients.
“This is a whole new approach,” Dr Reddy said. “The idea is to make the host cells stronger, to be able to withstand the assault of the donor immune cells while reducing the risk of infection or [disease] relapse.” ![]()

Photo by Aaron Logan
Increasing levels of a gut microbiome-derived metabolite could help prevent graft-versus-host disease (GVHD) after hematopoietic stem cell transplant (HSCT), according to preclinical research published in Nature Immunology.
For this study, researchers searched for alterations in the gut microbiome to see whether metabolites could impact outcomes after HSCT.
They found that levels of a metabolite called butyrate were significantly reduced in the intestinal tract of mice that received a transplant.
When the researchers increased butyrate levels in these mice, they saw a decrease in the incidence and severity of GVHD.
“Our findings suggest we can prevent graft-vs-host disease by bolstering the amount of the microbiome-derived metabolite butyrate,” said study author Pavan Reddy, MD, of the University of Michigan Comprehensive Cancer Center in Ann Arbor.
He and his colleagues found they could mitigate GVHD by administering butyrate to mice or by altering the composition of indigenous gastrointestinal microbiota so they produced high levels of butyrate.
The team noted that diet is another way to control butyrate production by the gut microbiome—specifically, through resistant starch such as that found in potatoes.
The researchers hope to begin a clinical trial later this year in which they will combine resistant starch with current methods of preventing GVHD to assess whether they can increase butyrate and reduce the incidence and severity of GVHD in HSCT recipients.
“This is a whole new approach,” Dr Reddy said. “The idea is to make the host cells stronger, to be able to withstand the assault of the donor immune cells while reducing the risk of infection or [disease] relapse.”
EMA recommends safety measures for idelalisib

Photo courtesy of
Gilead Sciences, Inc.
In light of recently reported safety concerns, the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) has issued provisional advice for doctors and patients using idelalisib (Zydelig).
The EMA recently began reviewing the safety of idelalisib because of serious adverse events, most of which were infection-related, that were reported in 3 clinical trials investigating idelalisib in combination with other drugs.
These trials have since been stopped.
Now, the PRAC is making provisional recommendations to ensure idelalisib is used as safely as possible outside of clinical trials.
These recommendations will be forwarded to the European Commission, which will issue a provisional legally binding decision applicable in all European Union (EU) member states.
In the EU, idelalisib is approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
Idelalisib is also approved for use in combination with rituximab to treat adults with chronic lymphocytic leukemia (CLL) who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients considered unsuitable for chemo-immunotherapy.
Recommendations
The PRAC is recommending that previously untreated CLL patients with 17p deletion or TP53 mutation do not begin treatment with idelalisib. In addition, doctors should re-evaluate each patient taking idelalisib as first-line treatment for CLL and only continue treatment if the benefits outweigh the risks.
Idelalisib can continue to be used in combination, only with rituximab, in CLL patients who have received at least 1 prior therapy and as monotherapy in patients with follicular lymphoma that is refractory to 2 lines of treatment.
Patients with any evidence of ongoing systemic infection should not be started on treatment with idelalisib.
And patients should be informed about the risk of serious infections with idelalisib.
All patients who do receive idelalisib should take antibiotics to prevent Pneumocystis jirovecii pneumonia, and they should be monitored for respiratory signs and symptoms. Regular clinical and laboratory monitoring for cytomegalovirus infection is also recommended.
Patients should have regular blood tests to detect neutropenia. In case a patient has moderate or severe neutropenia, treatment with idelalisib may have to be interrupted, in line with the updated summary of product characteristics.
The EMA said further details on these provisional measures will be provided in writing to healthcare professionals, and the product information will be updated accordingly.
Likewise, further information on the review of idelalisib will be provided as necessary and once the review is finished.
About the review
The EMA’s review of idelalisib started after a higher rate of serious adverse events, including deaths, was seen in 3 clinical trials evaluating the addition of idelalisib to standard therapy in first-line CLL and relapsed indolent non-Hodgkin lymphoma (NHL).
Most of the deaths were related to infections such as Pneumocystis jirovecii pneumonia and cytomegalovirus infection. Other excess deaths were related mainly to respiratory events.
The NHL studies (NCT01732926 and NCT01732913) included patients with disease characteristics different from those covered by the currently approved indication for idelalisib and investigated combinations of drugs that are not currently approved in the EU—idelalisib plus rituximab for NHL and idelalisib plus bendamustine and rituximab for NHL.
The CLL trial (NCT01980888) involved patients who had not received previous treatment, some of whom had the 17p deletion or TP53 mutation. However, the trial also investigated a combination of drugs not currently approved in the EU—idelalisib plus bendamustine and rituximab.
At present, the PRAC is conducting the review. Once the PRAC is finished, its recommendations will be forwarded to the EMA’s Committee for Medicinal Products for Human Use, which will adopt a final opinion.
The European Commission will take this opinion into account and adopt a legally binding decision that is applicable in all EU member states.

Photo courtesy of
Gilead Sciences, Inc.
In light of recently reported safety concerns, the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) has issued provisional advice for doctors and patients using idelalisib (Zydelig).
The EMA recently began reviewing the safety of idelalisib because of serious adverse events, most of which were infection-related, that were reported in 3 clinical trials investigating idelalisib in combination with other drugs.
These trials have since been stopped.
Now, the PRAC is making provisional recommendations to ensure idelalisib is used as safely as possible outside of clinical trials.
These recommendations will be forwarded to the European Commission, which will issue a provisional legally binding decision applicable in all European Union (EU) member states.
In the EU, idelalisib is approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
Idelalisib is also approved for use in combination with rituximab to treat adults with chronic lymphocytic leukemia (CLL) who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients considered unsuitable for chemo-immunotherapy.
Recommendations
The PRAC is recommending that previously untreated CLL patients with 17p deletion or TP53 mutation do not begin treatment with idelalisib. In addition, doctors should re-evaluate each patient taking idelalisib as first-line treatment for CLL and only continue treatment if the benefits outweigh the risks.
Idelalisib can continue to be used in combination, only with rituximab, in CLL patients who have received at least 1 prior therapy and as monotherapy in patients with follicular lymphoma that is refractory to 2 lines of treatment.
Patients with any evidence of ongoing systemic infection should not be started on treatment with idelalisib.
And patients should be informed about the risk of serious infections with idelalisib.
All patients who do receive idelalisib should take antibiotics to prevent Pneumocystis jirovecii pneumonia, and they should be monitored for respiratory signs and symptoms. Regular clinical and laboratory monitoring for cytomegalovirus infection is also recommended.
Patients should have regular blood tests to detect neutropenia. In case a patient has moderate or severe neutropenia, treatment with idelalisib may have to be interrupted, in line with the updated summary of product characteristics.
The EMA said further details on these provisional measures will be provided in writing to healthcare professionals, and the product information will be updated accordingly.
Likewise, further information on the review of idelalisib will be provided as necessary and once the review is finished.
About the review
The EMA’s review of idelalisib started after a higher rate of serious adverse events, including deaths, was seen in 3 clinical trials evaluating the addition of idelalisib to standard therapy in first-line CLL and relapsed indolent non-Hodgkin lymphoma (NHL).
Most of the deaths were related to infections such as Pneumocystis jirovecii pneumonia and cytomegalovirus infection. Other excess deaths were related mainly to respiratory events.
The NHL studies (NCT01732926 and NCT01732913) included patients with disease characteristics different from those covered by the currently approved indication for idelalisib and investigated combinations of drugs that are not currently approved in the EU—idelalisib plus rituximab for NHL and idelalisib plus bendamustine and rituximab for NHL.
The CLL trial (NCT01980888) involved patients who had not received previous treatment, some of whom had the 17p deletion or TP53 mutation. However, the trial also investigated a combination of drugs not currently approved in the EU—idelalisib plus bendamustine and rituximab.
At present, the PRAC is conducting the review. Once the PRAC is finished, its recommendations will be forwarded to the EMA’s Committee for Medicinal Products for Human Use, which will adopt a final opinion.
The European Commission will take this opinion into account and adopt a legally binding decision that is applicable in all EU member states.

Photo courtesy of
Gilead Sciences, Inc.
In light of recently reported safety concerns, the European Medicines Agency’s (EMA) Pharmacovigilance Risk Assessment Committee (PRAC) has issued provisional advice for doctors and patients using idelalisib (Zydelig).
The EMA recently began reviewing the safety of idelalisib because of serious adverse events, most of which were infection-related, that were reported in 3 clinical trials investigating idelalisib in combination with other drugs.
These trials have since been stopped.
Now, the PRAC is making provisional recommendations to ensure idelalisib is used as safely as possible outside of clinical trials.
These recommendations will be forwarded to the European Commission, which will issue a provisional legally binding decision applicable in all European Union (EU) member states.
In the EU, idelalisib is approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
Idelalisib is also approved for use in combination with rituximab to treat adults with chronic lymphocytic leukemia (CLL) who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients considered unsuitable for chemo-immunotherapy.
Recommendations
The PRAC is recommending that previously untreated CLL patients with 17p deletion or TP53 mutation do not begin treatment with idelalisib. In addition, doctors should re-evaluate each patient taking idelalisib as first-line treatment for CLL and only continue treatment if the benefits outweigh the risks.
Idelalisib can continue to be used in combination, only with rituximab, in CLL patients who have received at least 1 prior therapy and as monotherapy in patients with follicular lymphoma that is refractory to 2 lines of treatment.
Patients with any evidence of ongoing systemic infection should not be started on treatment with idelalisib.
And patients should be informed about the risk of serious infections with idelalisib.
All patients who do receive idelalisib should take antibiotics to prevent Pneumocystis jirovecii pneumonia, and they should be monitored for respiratory signs and symptoms. Regular clinical and laboratory monitoring for cytomegalovirus infection is also recommended.
Patients should have regular blood tests to detect neutropenia. In case a patient has moderate or severe neutropenia, treatment with idelalisib may have to be interrupted, in line with the updated summary of product characteristics.
The EMA said further details on these provisional measures will be provided in writing to healthcare professionals, and the product information will be updated accordingly.
Likewise, further information on the review of idelalisib will be provided as necessary and once the review is finished.
About the review
The EMA’s review of idelalisib started after a higher rate of serious adverse events, including deaths, was seen in 3 clinical trials evaluating the addition of idelalisib to standard therapy in first-line CLL and relapsed indolent non-Hodgkin lymphoma (NHL).
Most of the deaths were related to infections such as Pneumocystis jirovecii pneumonia and cytomegalovirus infection. Other excess deaths were related mainly to respiratory events.
The NHL studies (NCT01732926 and NCT01732913) included patients with disease characteristics different from those covered by the currently approved indication for idelalisib and investigated combinations of drugs that are not currently approved in the EU—idelalisib plus rituximab for NHL and idelalisib plus bendamustine and rituximab for NHL.
The CLL trial (NCT01980888) involved patients who had not received previous treatment, some of whom had the 17p deletion or TP53 mutation. However, the trial also investigated a combination of drugs not currently approved in the EU—idelalisib plus bendamustine and rituximab.
At present, the PRAC is conducting the review. Once the PRAC is finished, its recommendations will be forwarded to the EMA’s Committee for Medicinal Products for Human Use, which will adopt a final opinion.
The European Commission will take this opinion into account and adopt a legally binding decision that is applicable in all EU member states.
FDA proposes ban on most powdered medical gloves
Photo by Chonion Antoine
The US Food and Drug Administration (FDA) is proposing a ban on most powdered gloves in the US.
The proposed ban applies to powdered surgeon’s gloves, powdered patient examination gloves, and absorbable powder for lubricating a surgeon’s glove.
The FDA said these gloves pose an unreasonable and substantial risk of illness or injury to healthcare providers, patients, and other individuals who are exposed to them, which cannot be corrected through new or updated labeling.
“This ban is about protecting patients and healthcare professionals from a danger they might not even be aware of,” said Jeffrey Shuren, MD, director of FDA’s Center for Devices and Radiological Health.
“We take bans very seriously and only take this action when we feel it’s necessary to protect the public health.”
The FDA said powdered gloves are dangerous for a variety of reasons. In particular, aerosolized glove powder on natural rubber latex gloves, but not on synthetic powdered gloves, can carry proteins that may cause respiratory allergic reactions.
Although powdered synthetic gloves do not present the risk of allergic reactions, these gloves are associated with an extensive list of potentially serious adverse events, including severe airway inflammation, wound inflammation, and post-surgical adhesions. These side effects have been attributed to the use of glove powder with all types of gloves.
As these risks cannot be corrected through new or updated labeling, the FDA is moving forward with the proposal to ban these products, which—if finalized—would ultimately remove them from the marketplace completely.
In making the determination that these products are dangerous and present an unreasonable and substantial risk, the FDA considered all available evidence, which included a thorough review of the scientific literature and comments received on a February 2011 Federal Register Notice.
The FDA also conducted an economic analysis that showed a powdered glove ban would not cause a glove shortage, and the economic impact of a ban would not be significant. The ban is also not likely to impact medical practice, because many non-powdered protective glove options are currently available.
The FDA has determined that the banning standard would not apply to powdered radiographic protection gloves. The agency is not aware of any powdered radiographic protection gloves that are currently on the market.
Non-powdered surgeon gloves and non-powdered patient examination gloves will not be included in the ban and will remain Class I medical devices. Therefore, the FDA is also proposing amendments to their classification regulations to clarify that they apply only to non-powdered gloves.
The proposed rule is available online at www.regulations.gov for public comment for 90 days.

Photo by Chonion Antoine
The US Food and Drug Administration (FDA) is proposing a ban on most powdered gloves in the US.
The proposed ban applies to powdered surgeon’s gloves, powdered patient examination gloves, and absorbable powder for lubricating a surgeon’s glove.
The FDA said these gloves pose an unreasonable and substantial risk of illness or injury to healthcare providers, patients, and other individuals who are exposed to them, which cannot be corrected through new or updated labeling.
“This ban is about protecting patients and healthcare professionals from a danger they might not even be aware of,” said Jeffrey Shuren, MD, director of FDA’s Center for Devices and Radiological Health.
“We take bans very seriously and only take this action when we feel it’s necessary to protect the public health.”
The FDA said powdered gloves are dangerous for a variety of reasons. In particular, aerosolized glove powder on natural rubber latex gloves, but not on synthetic powdered gloves, can carry proteins that may cause respiratory allergic reactions.
Although powdered synthetic gloves do not present the risk of allergic reactions, these gloves are associated with an extensive list of potentially serious adverse events, including severe airway inflammation, wound inflammation, and post-surgical adhesions. These side effects have been attributed to the use of glove powder with all types of gloves.
As these risks cannot be corrected through new or updated labeling, the FDA is moving forward with the proposal to ban these products, which—if finalized—would ultimately remove them from the marketplace completely.
In making the determination that these products are dangerous and present an unreasonable and substantial risk, the FDA considered all available evidence, which included a thorough review of the scientific literature and comments received on a February 2011 Federal Register Notice.
The FDA also conducted an economic analysis that showed a powdered glove ban would not cause a glove shortage, and the economic impact of a ban would not be significant. The ban is also not likely to impact medical practice, because many non-powdered protective glove options are currently available.
The FDA has determined that the banning standard would not apply to powdered radiographic protection gloves. The agency is not aware of any powdered radiographic protection gloves that are currently on the market.
Non-powdered surgeon gloves and non-powdered patient examination gloves will not be included in the ban and will remain Class I medical devices. Therefore, the FDA is also proposing amendments to their classification regulations to clarify that they apply only to non-powdered gloves.
The proposed rule is available online at www.regulations.gov for public comment for 90 days.

Photo by Chonion Antoine
The US Food and Drug Administration (FDA) is proposing a ban on most powdered gloves in the US.
The proposed ban applies to powdered surgeon’s gloves, powdered patient examination gloves, and absorbable powder for lubricating a surgeon’s glove.
The FDA said these gloves pose an unreasonable and substantial risk of illness or injury to healthcare providers, patients, and other individuals who are exposed to them, which cannot be corrected through new or updated labeling.
“This ban is about protecting patients and healthcare professionals from a danger they might not even be aware of,” said Jeffrey Shuren, MD, director of FDA’s Center for Devices and Radiological Health.
“We take bans very seriously and only take this action when we feel it’s necessary to protect the public health.”
The FDA said powdered gloves are dangerous for a variety of reasons. In particular, aerosolized glove powder on natural rubber latex gloves, but not on synthetic powdered gloves, can carry proteins that may cause respiratory allergic reactions.
Although powdered synthetic gloves do not present the risk of allergic reactions, these gloves are associated with an extensive list of potentially serious adverse events, including severe airway inflammation, wound inflammation, and post-surgical adhesions. These side effects have been attributed to the use of glove powder with all types of gloves.
As these risks cannot be corrected through new or updated labeling, the FDA is moving forward with the proposal to ban these products, which—if finalized—would ultimately remove them from the marketplace completely.
In making the determination that these products are dangerous and present an unreasonable and substantial risk, the FDA considered all available evidence, which included a thorough review of the scientific literature and comments received on a February 2011 Federal Register Notice.
The FDA also conducted an economic analysis that showed a powdered glove ban would not cause a glove shortage, and the economic impact of a ban would not be significant. The ban is also not likely to impact medical practice, because many non-powdered protective glove options are currently available.
The FDA has determined that the banning standard would not apply to powdered radiographic protection gloves. The agency is not aware of any powdered radiographic protection gloves that are currently on the market.
Non-powdered surgeon gloves and non-powdered patient examination gloves will not be included in the ban and will remain Class I medical devices. Therefore, the FDA is also proposing amendments to their classification regulations to clarify that they apply only to non-powdered gloves.
The proposed rule is available online at www.regulations.gov for public comment for 90 days.
Four genes control growth of HSCs, team says
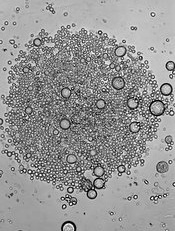
Image by John Perry
Four genes govern the growth and multiplication of hematopoietic stem cells (HSCs), according to a study published in Cell Reports.
Investigators conducted a genome-wide RNA interference screen in cells derived from human cord blood, looking for genes whose knockdown maintained the HSC phenotype during culture.
This revealed the 4 genes—STAG2, RAD21, STAG1, and SMC3—which are all members of the cohesin complex.
“The discovery showed that this protein complex is crucial and has an overarching function in the growth of the blood stem cells,” said study author Jonas Larsson, MD, PhD, of Lund University in Sweden.
Dr Larsson and his colleagues screened 15,000 genes for this study and found 20 candidates with a strong capacity to affect the balance of growth in HSCs.
Four of the genes—STAG2, RAD21, STAG1, and SMC3—were physically connected through cooperation in the cohesin complex. This complex forms a sort of brace that holds different parts of the DNA strand together in the cell.
The researchers believe this allows the cohesin complex to control access to the “on/off switches” in the DNA and to change the impulses that HSCs receive from various genes, thereby affecting whether an HSC multiplies or matures.
“Clarifying what regulates the balance between multiplication and maturation of blood stem cells could provide the right keys to expanding them outside the body,” said Roman Galeev, a doctoral student at Lund University.
“In addition, it would enable the identification of new points of attack for the treatment of blood cancer, which is precisely a disruption of the balance between multiplication and maturation.”
Galeev noted that research by other groups has revealed mutations in the cohesin complex genes in patients with hematologic malignancies (Mazumdar C, Cell Stem Cell 2015; Mullenders J, J Exp Med 2015; and Viny AD, J Exp Med 2015).
“This is incredibly exciting,” Galeev said. “Together with the results from our study, this indicates that the cohesin genes are directly and crucially significant in the development of blood cancer.”
“Our findings entail a new understanding of how the expansion of blood stem cells is controlled. Eventually, this can lead to new ways of affecting the process, either to prevent the development of cancer or to expand the stem cells for transplant.”

Image by John Perry
Four genes govern the growth and multiplication of hematopoietic stem cells (HSCs), according to a study published in Cell Reports.
Investigators conducted a genome-wide RNA interference screen in cells derived from human cord blood, looking for genes whose knockdown maintained the HSC phenotype during culture.
This revealed the 4 genes—STAG2, RAD21, STAG1, and SMC3—which are all members of the cohesin complex.
“The discovery showed that this protein complex is crucial and has an overarching function in the growth of the blood stem cells,” said study author Jonas Larsson, MD, PhD, of Lund University in Sweden.
Dr Larsson and his colleagues screened 15,000 genes for this study and found 20 candidates with a strong capacity to affect the balance of growth in HSCs.
Four of the genes—STAG2, RAD21, STAG1, and SMC3—were physically connected through cooperation in the cohesin complex. This complex forms a sort of brace that holds different parts of the DNA strand together in the cell.
The researchers believe this allows the cohesin complex to control access to the “on/off switches” in the DNA and to change the impulses that HSCs receive from various genes, thereby affecting whether an HSC multiplies or matures.
“Clarifying what regulates the balance between multiplication and maturation of blood stem cells could provide the right keys to expanding them outside the body,” said Roman Galeev, a doctoral student at Lund University.
“In addition, it would enable the identification of new points of attack for the treatment of blood cancer, which is precisely a disruption of the balance between multiplication and maturation.”
Galeev noted that research by other groups has revealed mutations in the cohesin complex genes in patients with hematologic malignancies (Mazumdar C, Cell Stem Cell 2015; Mullenders J, J Exp Med 2015; and Viny AD, J Exp Med 2015).
“This is incredibly exciting,” Galeev said. “Together with the results from our study, this indicates that the cohesin genes are directly and crucially significant in the development of blood cancer.”
“Our findings entail a new understanding of how the expansion of blood stem cells is controlled. Eventually, this can lead to new ways of affecting the process, either to prevent the development of cancer or to expand the stem cells for transplant.”

Image by John Perry
Four genes govern the growth and multiplication of hematopoietic stem cells (HSCs), according to a study published in Cell Reports.
Investigators conducted a genome-wide RNA interference screen in cells derived from human cord blood, looking for genes whose knockdown maintained the HSC phenotype during culture.
This revealed the 4 genes—STAG2, RAD21, STAG1, and SMC3—which are all members of the cohesin complex.
“The discovery showed that this protein complex is crucial and has an overarching function in the growth of the blood stem cells,” said study author Jonas Larsson, MD, PhD, of Lund University in Sweden.
Dr Larsson and his colleagues screened 15,000 genes for this study and found 20 candidates with a strong capacity to affect the balance of growth in HSCs.
Four of the genes—STAG2, RAD21, STAG1, and SMC3—were physically connected through cooperation in the cohesin complex. This complex forms a sort of brace that holds different parts of the DNA strand together in the cell.
The researchers believe this allows the cohesin complex to control access to the “on/off switches” in the DNA and to change the impulses that HSCs receive from various genes, thereby affecting whether an HSC multiplies or matures.
“Clarifying what regulates the balance between multiplication and maturation of blood stem cells could provide the right keys to expanding them outside the body,” said Roman Galeev, a doctoral student at Lund University.
“In addition, it would enable the identification of new points of attack for the treatment of blood cancer, which is precisely a disruption of the balance between multiplication and maturation.”
Galeev noted that research by other groups has revealed mutations in the cohesin complex genes in patients with hematologic malignancies (Mazumdar C, Cell Stem Cell 2015; Mullenders J, J Exp Med 2015; and Viny AD, J Exp Med 2015).
“This is incredibly exciting,” Galeev said. “Together with the results from our study, this indicates that the cohesin genes are directly and crucially significant in the development of blood cancer.”
“Our findings entail a new understanding of how the expansion of blood stem cells is controlled. Eventually, this can lead to new ways of affecting the process, either to prevent the development of cancer or to expand the stem cells for transplant.”