User login
Computer models simulate HSCT recovery

Photo by Darren Baker
New research indicates that computer models can simulate the recovery of the immune system in patients undergoing hematopoietic stem cell transplant (HSCT).
The study suggests the possibility of using DNA sequencing and computer modeling to predict which HSCT recipients might suffer complications such as graft-versus-host-disease.
The research was published in Biology of Blood and Marrow Transplantation.
The study builds upon prior research, which showed that the immune system may be modeled as a dynamical system. Dynamical systems are mathematical objects used to model physical phenomena that change over time. These systems can be used to predict future states via observations of past and present states.
Researchers say the ability to predict immune system recovery after HSCT could potentially allow doctors to refine donor selection and better personalize post-transplant care to improve outcomes.
With this in mind, the team sequenced the DNA of 34 HSCT donor-recipient pairs and used the resulting information in a computer model to simulate how the recipient’s T-cell repertoire will recover following transplant.
“This study is the first to simulate the growth of the T-cell repertoire following transplantation using variables that aren’t accounted for in typical HLA donor-recipient matching,” said study author Amir Ahmed Toor, MD, of Virginia Commonwealth University in Richmond.
“Using a larger cohort of patients than in previous studies, we were able to mathematically predict the interactions of these variables, which led to simulations that appear to be very similar to clinically observed post-transplantation T-cell repertoire development.”
Previous research by Dr Toor and his colleagues revealed large variations between donor-recipient minor histocompatibility antigens that could potentially contribute to transplant complications not accounted for by HLA testing.
The models used in the computer simulations were driven by population growth formulas developed from past studies by Dr Toor and his colleagues that revealed distinct patterns of lymphocyte recovery in HSCT recipients.
Using matrix mathematics to develop the simulations, the researchers observed competition among T cells as the T-cell repertoire develops.
This competition leads to certain families of T cells becoming dominant and more numerous, which crowds out weaker T-cell families, causing them to develop later and in fewer numbers.
“We are attempting to account for the many variables that could impact T-cell repertoire development and, in turn, patient outcomes,” Dr Toor said.
“In future studies, we hope to explore the impact of organ-specific antigen expression. The knowledge gained from this research could potentially allow more accurate predictions of which organs could be most affected by graft-versus-host-disease.” ![]()

Photo by Darren Baker
New research indicates that computer models can simulate the recovery of the immune system in patients undergoing hematopoietic stem cell transplant (HSCT).
The study suggests the possibility of using DNA sequencing and computer modeling to predict which HSCT recipients might suffer complications such as graft-versus-host-disease.
The research was published in Biology of Blood and Marrow Transplantation.
The study builds upon prior research, which showed that the immune system may be modeled as a dynamical system. Dynamical systems are mathematical objects used to model physical phenomena that change over time. These systems can be used to predict future states via observations of past and present states.
Researchers say the ability to predict immune system recovery after HSCT could potentially allow doctors to refine donor selection and better personalize post-transplant care to improve outcomes.
With this in mind, the team sequenced the DNA of 34 HSCT donor-recipient pairs and used the resulting information in a computer model to simulate how the recipient’s T-cell repertoire will recover following transplant.
“This study is the first to simulate the growth of the T-cell repertoire following transplantation using variables that aren’t accounted for in typical HLA donor-recipient matching,” said study author Amir Ahmed Toor, MD, of Virginia Commonwealth University in Richmond.
“Using a larger cohort of patients than in previous studies, we were able to mathematically predict the interactions of these variables, which led to simulations that appear to be very similar to clinically observed post-transplantation T-cell repertoire development.”
Previous research by Dr Toor and his colleagues revealed large variations between donor-recipient minor histocompatibility antigens that could potentially contribute to transplant complications not accounted for by HLA testing.
The models used in the computer simulations were driven by population growth formulas developed from past studies by Dr Toor and his colleagues that revealed distinct patterns of lymphocyte recovery in HSCT recipients.
Using matrix mathematics to develop the simulations, the researchers observed competition among T cells as the T-cell repertoire develops.
This competition leads to certain families of T cells becoming dominant and more numerous, which crowds out weaker T-cell families, causing them to develop later and in fewer numbers.
“We are attempting to account for the many variables that could impact T-cell repertoire development and, in turn, patient outcomes,” Dr Toor said.
“In future studies, we hope to explore the impact of organ-specific antigen expression. The knowledge gained from this research could potentially allow more accurate predictions of which organs could be most affected by graft-versus-host-disease.” ![]()

Photo by Darren Baker
New research indicates that computer models can simulate the recovery of the immune system in patients undergoing hematopoietic stem cell transplant (HSCT).
The study suggests the possibility of using DNA sequencing and computer modeling to predict which HSCT recipients might suffer complications such as graft-versus-host-disease.
The research was published in Biology of Blood and Marrow Transplantation.
The study builds upon prior research, which showed that the immune system may be modeled as a dynamical system. Dynamical systems are mathematical objects used to model physical phenomena that change over time. These systems can be used to predict future states via observations of past and present states.
Researchers say the ability to predict immune system recovery after HSCT could potentially allow doctors to refine donor selection and better personalize post-transplant care to improve outcomes.
With this in mind, the team sequenced the DNA of 34 HSCT donor-recipient pairs and used the resulting information in a computer model to simulate how the recipient’s T-cell repertoire will recover following transplant.
“This study is the first to simulate the growth of the T-cell repertoire following transplantation using variables that aren’t accounted for in typical HLA donor-recipient matching,” said study author Amir Ahmed Toor, MD, of Virginia Commonwealth University in Richmond.
“Using a larger cohort of patients than in previous studies, we were able to mathematically predict the interactions of these variables, which led to simulations that appear to be very similar to clinically observed post-transplantation T-cell repertoire development.”
Previous research by Dr Toor and his colleagues revealed large variations between donor-recipient minor histocompatibility antigens that could potentially contribute to transplant complications not accounted for by HLA testing.
The models used in the computer simulations were driven by population growth formulas developed from past studies by Dr Toor and his colleagues that revealed distinct patterns of lymphocyte recovery in HSCT recipients.
Using matrix mathematics to develop the simulations, the researchers observed competition among T cells as the T-cell repertoire develops.
This competition leads to certain families of T cells becoming dominant and more numerous, which crowds out weaker T-cell families, causing them to develop later and in fewer numbers.
“We are attempting to account for the many variables that could impact T-cell repertoire development and, in turn, patient outcomes,” Dr Toor said.
“In future studies, we hope to explore the impact of organ-specific antigen expression. The knowledge gained from this research could potentially allow more accurate predictions of which organs could be most affected by graft-versus-host-disease.” ![]()
Antibody recognizes human plasma cells
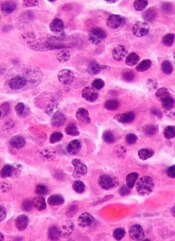
Researchers say they have generated a monoclonal antibody that could have diagnostic and therapeutic applications for multiple myeloma (MM) and other plasma cell disorders.
The team generated this antibody, VLRB MM3, from immunized lampreys, a type of jawless fish.
Experiments with VLRB MM3 showed that it can identify normal plasma cells in samples from healthy donors and malignant plasma cells in samples from patients with MM.
Götz Ehrhardt, PhD, of the University of Toronto in Ontario, Canada, and his colleagues described this work in JCI Insight.
The researchers noted that antibody-secreting plasma cells arise from B-cell precursors and are essential for adaptive immune responses against invading pathogens. Plasma cell dysfunction is associated with autoimmune and neoplastic disorders, including MM.
Surface markers that are specific to plasma cells have not been identified, and antibodies that only recognize these cells have been challenging to generate using conventional systems.
However, Dr Ehrhardt and his colleagues found they could generate a plasma-cell-specific antibody from immunized lampreys.
The researchers injected lamprey larvae with a bone marrow isolate from an MM patient and screened the resulting monoclonal antibodies for those that recognized both malignant and non-malignant plasma cells.
Further characterization of the antibody VLRB MM3 revealed that it is specific to plasma cells and does not recognize other B-cell populations or progenitors.
VLRB MM3 binding was shown to coincide with CD38 dimerization and correlate with and impede the NAD glycohydrolase activity of this glycoprotein.
Considering these findings together, the researchers concluded that VLRB MM3 represents a unique tool that might aid the treatment and diagnosis of plasma cell disorders. ![]()

Researchers say they have generated a monoclonal antibody that could have diagnostic and therapeutic applications for multiple myeloma (MM) and other plasma cell disorders.
The team generated this antibody, VLRB MM3, from immunized lampreys, a type of jawless fish.
Experiments with VLRB MM3 showed that it can identify normal plasma cells in samples from healthy donors and malignant plasma cells in samples from patients with MM.
Götz Ehrhardt, PhD, of the University of Toronto in Ontario, Canada, and his colleagues described this work in JCI Insight.
The researchers noted that antibody-secreting plasma cells arise from B-cell precursors and are essential for adaptive immune responses against invading pathogens. Plasma cell dysfunction is associated with autoimmune and neoplastic disorders, including MM.
Surface markers that are specific to plasma cells have not been identified, and antibodies that only recognize these cells have been challenging to generate using conventional systems.
However, Dr Ehrhardt and his colleagues found they could generate a plasma-cell-specific antibody from immunized lampreys.
The researchers injected lamprey larvae with a bone marrow isolate from an MM patient and screened the resulting monoclonal antibodies for those that recognized both malignant and non-malignant plasma cells.
Further characterization of the antibody VLRB MM3 revealed that it is specific to plasma cells and does not recognize other B-cell populations or progenitors.
VLRB MM3 binding was shown to coincide with CD38 dimerization and correlate with and impede the NAD glycohydrolase activity of this glycoprotein.
Considering these findings together, the researchers concluded that VLRB MM3 represents a unique tool that might aid the treatment and diagnosis of plasma cell disorders. ![]()

Researchers say they have generated a monoclonal antibody that could have diagnostic and therapeutic applications for multiple myeloma (MM) and other plasma cell disorders.
The team generated this antibody, VLRB MM3, from immunized lampreys, a type of jawless fish.
Experiments with VLRB MM3 showed that it can identify normal plasma cells in samples from healthy donors and malignant plasma cells in samples from patients with MM.
Götz Ehrhardt, PhD, of the University of Toronto in Ontario, Canada, and his colleagues described this work in JCI Insight.
The researchers noted that antibody-secreting plasma cells arise from B-cell precursors and are essential for adaptive immune responses against invading pathogens. Plasma cell dysfunction is associated with autoimmune and neoplastic disorders, including MM.
Surface markers that are specific to plasma cells have not been identified, and antibodies that only recognize these cells have been challenging to generate using conventional systems.
However, Dr Ehrhardt and his colleagues found they could generate a plasma-cell-specific antibody from immunized lampreys.
The researchers injected lamprey larvae with a bone marrow isolate from an MM patient and screened the resulting monoclonal antibodies for those that recognized both malignant and non-malignant plasma cells.
Further characterization of the antibody VLRB MM3 revealed that it is specific to plasma cells and does not recognize other B-cell populations or progenitors.
VLRB MM3 binding was shown to coincide with CD38 dimerization and correlate with and impede the NAD glycohydrolase activity of this glycoprotein.
Considering these findings together, the researchers concluded that VLRB MM3 represents a unique tool that might aid the treatment and diagnosis of plasma cell disorders. ![]()
FDA authorizes new test for Zika virus

Photo by Graham Colm
In response to a request from the US Centers for Disease Control and Prevention (CDC), the US Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) for the Trioplex Real-time RT-PCR Assay, a tool that can be used to detect Zika virus.
The assay allows doctors to tell if an individual is infected with chikungunya, dengue, or Zika virus using a single test, instead of having to perform 3 separate tests to identify the infection.
The Trioplex Real-time RT-PCR Assay can be used to detect virus RNA in serum, cerebrospinal fluid, urine, and amniotic fluid specimens.
The CDC hopes this EUA will allow the agency to more rapidly perform testing to detect acute Zika virus infection.
An EUA allows the use of unapproved medical products or unapproved uses of approved medical products in an emergency. The products must be used to diagnose, treat, or prevent serious or life-threatening conditions caused by chemical, biological, radiological, or nuclear threat agents, when there are no adequate alternatives.
The CDC said it will begin distributing the Trioplex Real-time RT-PCR Assay during the next 2 weeks to qualified laboratories in the Laboratory Response Network, an integrated network of domestic and international laboratories that respond to public health emergencies.
The test will not be available in US hospitals or other primary care settings.
Last month, the FDA issued an EUA for a different test used to detect the Zika virus, the Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA).
This test was distributed to qualified laboratories in the Laboratory Response Network but was not made available in US hospitals or other primary care settings. ![]()

Photo by Graham Colm
In response to a request from the US Centers for Disease Control and Prevention (CDC), the US Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) for the Trioplex Real-time RT-PCR Assay, a tool that can be used to detect Zika virus.
The assay allows doctors to tell if an individual is infected with chikungunya, dengue, or Zika virus using a single test, instead of having to perform 3 separate tests to identify the infection.
The Trioplex Real-time RT-PCR Assay can be used to detect virus RNA in serum, cerebrospinal fluid, urine, and amniotic fluid specimens.
The CDC hopes this EUA will allow the agency to more rapidly perform testing to detect acute Zika virus infection.
An EUA allows the use of unapproved medical products or unapproved uses of approved medical products in an emergency. The products must be used to diagnose, treat, or prevent serious or life-threatening conditions caused by chemical, biological, radiological, or nuclear threat agents, when there are no adequate alternatives.
The CDC said it will begin distributing the Trioplex Real-time RT-PCR Assay during the next 2 weeks to qualified laboratories in the Laboratory Response Network, an integrated network of domestic and international laboratories that respond to public health emergencies.
The test will not be available in US hospitals or other primary care settings.
Last month, the FDA issued an EUA for a different test used to detect the Zika virus, the Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA).
This test was distributed to qualified laboratories in the Laboratory Response Network but was not made available in US hospitals or other primary care settings. ![]()

Photo by Graham Colm
In response to a request from the US Centers for Disease Control and Prevention (CDC), the US Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) for the Trioplex Real-time RT-PCR Assay, a tool that can be used to detect Zika virus.
The assay allows doctors to tell if an individual is infected with chikungunya, dengue, or Zika virus using a single test, instead of having to perform 3 separate tests to identify the infection.
The Trioplex Real-time RT-PCR Assay can be used to detect virus RNA in serum, cerebrospinal fluid, urine, and amniotic fluid specimens.
The CDC hopes this EUA will allow the agency to more rapidly perform testing to detect acute Zika virus infection.
An EUA allows the use of unapproved medical products or unapproved uses of approved medical products in an emergency. The products must be used to diagnose, treat, or prevent serious or life-threatening conditions caused by chemical, biological, radiological, or nuclear threat agents, when there are no adequate alternatives.
The CDC said it will begin distributing the Trioplex Real-time RT-PCR Assay during the next 2 weeks to qualified laboratories in the Laboratory Response Network, an integrated network of domestic and international laboratories that respond to public health emergencies.
The test will not be available in US hospitals or other primary care settings.
Last month, the FDA issued an EUA for a different test used to detect the Zika virus, the Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA).
This test was distributed to qualified laboratories in the Laboratory Response Network but was not made available in US hospitals or other primary care settings. ![]()
FDA rejects ready-to-use bivalirudin formulation

Image by Kevin MacKenzie
The US Food and Drug Administration (FDA) has said it cannot, at present, approve a ready-to-use (RTU) formulation of bivalirudin (Kangio).
The product is a liquid, intravenous formulation of 5 mg/mL of bivalirudin in a 50-mL vial.
It does not require reconstitution before administration.
This RTU bivalirudin is intended for use as an anticoagulant in patients with unstable angina who are undergoing percutaneous transluminal coronary angioplasty.
The drug is also intended for patients undergoing percutaneous coronary intervention (PCI) with provisional use of a glycoprotein IIb/IIIa inhibitor and for patients undergoing PCI who have or are at risk of developing heparin-induced thrombocytopenia and thrombosis syndrome.
The RTU bivalirudin contains the same active ingredient and is designed to be given at the same dose and rate as Angiomax, a lyophilized powder form of bivalirudin that must be reconstituted. The RTU formulation does not require any reconstitution or initial dilution, which is intended to reduce work flow and the risk of dosing errors.
The FDA issued a complete response letter to the company developing the RTU formulation of bivalirudin, Eagle Pharmaceuticals.
The FDA issues complete response letters to communicate that the initial review of an application is complete, but the agency cannot approve the application in its present form and requests additional information.
In its letter to Eagle Pharmaceuticals, the FDA requested further characterization of bivalirudin-related substances in the drug product. The company said it will work directly with the FDA to determine an appropriate path forward to address the comments.
“We are evaluating the FDA’s response and will work closely with the agency to better understand and address their comments regarding Kangio,” said Scott Tarriff, president and chief executive officer of Eagle Pharmaceuticals.
“We remain committed to Kangio as an important new formulation of bivalirudin for intravenous use, offering multiple benefits for patients and caregivers.” ![]()

Image by Kevin MacKenzie
The US Food and Drug Administration (FDA) has said it cannot, at present, approve a ready-to-use (RTU) formulation of bivalirudin (Kangio).
The product is a liquid, intravenous formulation of 5 mg/mL of bivalirudin in a 50-mL vial.
It does not require reconstitution before administration.
This RTU bivalirudin is intended for use as an anticoagulant in patients with unstable angina who are undergoing percutaneous transluminal coronary angioplasty.
The drug is also intended for patients undergoing percutaneous coronary intervention (PCI) with provisional use of a glycoprotein IIb/IIIa inhibitor and for patients undergoing PCI who have or are at risk of developing heparin-induced thrombocytopenia and thrombosis syndrome.
The RTU bivalirudin contains the same active ingredient and is designed to be given at the same dose and rate as Angiomax, a lyophilized powder form of bivalirudin that must be reconstituted. The RTU formulation does not require any reconstitution or initial dilution, which is intended to reduce work flow and the risk of dosing errors.
The FDA issued a complete response letter to the company developing the RTU formulation of bivalirudin, Eagle Pharmaceuticals.
The FDA issues complete response letters to communicate that the initial review of an application is complete, but the agency cannot approve the application in its present form and requests additional information.
In its letter to Eagle Pharmaceuticals, the FDA requested further characterization of bivalirudin-related substances in the drug product. The company said it will work directly with the FDA to determine an appropriate path forward to address the comments.
“We are evaluating the FDA’s response and will work closely with the agency to better understand and address their comments regarding Kangio,” said Scott Tarriff, president and chief executive officer of Eagle Pharmaceuticals.
“We remain committed to Kangio as an important new formulation of bivalirudin for intravenous use, offering multiple benefits for patients and caregivers.” ![]()

Image by Kevin MacKenzie
The US Food and Drug Administration (FDA) has said it cannot, at present, approve a ready-to-use (RTU) formulation of bivalirudin (Kangio).
The product is a liquid, intravenous formulation of 5 mg/mL of bivalirudin in a 50-mL vial.
It does not require reconstitution before administration.
This RTU bivalirudin is intended for use as an anticoagulant in patients with unstable angina who are undergoing percutaneous transluminal coronary angioplasty.
The drug is also intended for patients undergoing percutaneous coronary intervention (PCI) with provisional use of a glycoprotein IIb/IIIa inhibitor and for patients undergoing PCI who have or are at risk of developing heparin-induced thrombocytopenia and thrombosis syndrome.
The RTU bivalirudin contains the same active ingredient and is designed to be given at the same dose and rate as Angiomax, a lyophilized powder form of bivalirudin that must be reconstituted. The RTU formulation does not require any reconstitution or initial dilution, which is intended to reduce work flow and the risk of dosing errors.
The FDA issued a complete response letter to the company developing the RTU formulation of bivalirudin, Eagle Pharmaceuticals.
The FDA issues complete response letters to communicate that the initial review of an application is complete, but the agency cannot approve the application in its present form and requests additional information.
In its letter to Eagle Pharmaceuticals, the FDA requested further characterization of bivalirudin-related substances in the drug product. The company said it will work directly with the FDA to determine an appropriate path forward to address the comments.
“We are evaluating the FDA’s response and will work closely with the agency to better understand and address their comments regarding Kangio,” said Scott Tarriff, president and chief executive officer of Eagle Pharmaceuticals.
“We remain committed to Kangio as an important new formulation of bivalirudin for intravenous use, offering multiple benefits for patients and caregivers.” ![]()
Drug may best BSC in some high-risk MDS patients

Results of a phase 3 trial suggest the small-molecule inhibitor rigosertib may improve overall survival (OS) in some patients with higher-risk myelodysplastic syndromes (HR-MDS).
Overall, researchers found no significant difference in OS between patients who received rigosertib and those who received best supportive care (BSC).
However, the data indicate that rigosertib can confer a survival benefit in certain subgroups of HR-MDS patients.
The results of this trial, known as ONTIME, were published in The Lancet Oncology. The trial was sponsored by Onconova Therapeutics, Inc., the company developing rigosertib.
The trial enrolled 299 HR-MDS patients. They had refractory anemia with excess blasts (RAEB)-1, RAEB-2, RAEB-t, or chronic myelomonocytic leukemia based on local site assessment. They had all failed treatment with a hypomethylating agent (HMA) in the past 2 years.
The patients were randomized (2:1) to receive rigosertib at 1800 mg per 24 hours via 72-hour continuous intravenous (IV) infusion, administered every other week (n=199), or BSC with or without low-dose cytarabine (n=100).
At a median follow-up of 19.5 months, there was no significant difference in OS between the treatment arms. The median OS was 8.2 months in the rigosertib arm and 5.9 months in the BSC arm. The hazard ratio (HR) was 0.87 (P=0.33).
However, the researchers said that subgroup analyses suggested rigosertib may provide a survival benefit over BSC in some HR-MDS patients. This includes:
- Patients younger than 75 years of age (HR=0.55, P=0.0010)
- Patients who received HMA therapy for 9 months or fewer (HR=0.54, P=0.0016)
- Patients with primary, rather than secondary, HMA failure (HR=0.72, P=0.060)
- Patients who were classified as “very high risk” according to the Revised International Prognostic Scoring System (HR=0.61, P=0.015)
- Patients with monosomy 7 (HR=0.26, P=0.0041)
- Patients with trisomy 8 (HR=0.28, P=0.0083).
The most common grade 3 or higher adverse events—in the rigosertib and BSC arms, respectively—were anemia (18% vs 8%), thrombocytopenia (19% vs 7%), neutropenia (17% vs 8%), febrile neutropenia (12% vs 11%), and pneumonia (12% vs 11%).
Twenty-two percent of patients in the rigosertib arm and 33% in the BSC arm died due to adverse events. Three deaths were attributed to rigosertib.
“Rigosertib was well-tolerated in patients with a high unmet medical need who have no approved therapeutic options,” said study author Guillermo Garcia-Manero, MD, of The University of Texas MD Anderson Cancer Center in Houston.
“We are impressed by the trend to notable efficacy in well-defined, well-balanced subgroups of HR-MDS patients with very poor prognosis. Based on these findings, we have designed the new phase 3 INSPIRE study with IV rigosertib, which is currently enrolling patients.”
INSPIRE is a randomized, controlled study designed to assess the efficacy and safety of IV rigosertib in HR-MDS patients under 80 years of age who had progressed on, failed to respond to, or relapsed after previous treatment with an HMA within the first 9 months of HMA treatment initiation.
The trial is expected to enroll approximately 225 patients, who will be randomized at a 2:1 ratio into 2 treatment arms: IV rigosertib plus BSC versus physician’s choice plus BSC. The primary endpoint is OS. Full details on the trial can be found on clinicaltrials.gov (NCT02562443). ![]()

Results of a phase 3 trial suggest the small-molecule inhibitor rigosertib may improve overall survival (OS) in some patients with higher-risk myelodysplastic syndromes (HR-MDS).
Overall, researchers found no significant difference in OS between patients who received rigosertib and those who received best supportive care (BSC).
However, the data indicate that rigosertib can confer a survival benefit in certain subgroups of HR-MDS patients.
The results of this trial, known as ONTIME, were published in The Lancet Oncology. The trial was sponsored by Onconova Therapeutics, Inc., the company developing rigosertib.
The trial enrolled 299 HR-MDS patients. They had refractory anemia with excess blasts (RAEB)-1, RAEB-2, RAEB-t, or chronic myelomonocytic leukemia based on local site assessment. They had all failed treatment with a hypomethylating agent (HMA) in the past 2 years.
The patients were randomized (2:1) to receive rigosertib at 1800 mg per 24 hours via 72-hour continuous intravenous (IV) infusion, administered every other week (n=199), or BSC with or without low-dose cytarabine (n=100).
At a median follow-up of 19.5 months, there was no significant difference in OS between the treatment arms. The median OS was 8.2 months in the rigosertib arm and 5.9 months in the BSC arm. The hazard ratio (HR) was 0.87 (P=0.33).
However, the researchers said that subgroup analyses suggested rigosertib may provide a survival benefit over BSC in some HR-MDS patients. This includes:
- Patients younger than 75 years of age (HR=0.55, P=0.0010)
- Patients who received HMA therapy for 9 months or fewer (HR=0.54, P=0.0016)
- Patients with primary, rather than secondary, HMA failure (HR=0.72, P=0.060)
- Patients who were classified as “very high risk” according to the Revised International Prognostic Scoring System (HR=0.61, P=0.015)
- Patients with monosomy 7 (HR=0.26, P=0.0041)
- Patients with trisomy 8 (HR=0.28, P=0.0083).
The most common grade 3 or higher adverse events—in the rigosertib and BSC arms, respectively—were anemia (18% vs 8%), thrombocytopenia (19% vs 7%), neutropenia (17% vs 8%), febrile neutropenia (12% vs 11%), and pneumonia (12% vs 11%).
Twenty-two percent of patients in the rigosertib arm and 33% in the BSC arm died due to adverse events. Three deaths were attributed to rigosertib.
“Rigosertib was well-tolerated in patients with a high unmet medical need who have no approved therapeutic options,” said study author Guillermo Garcia-Manero, MD, of The University of Texas MD Anderson Cancer Center in Houston.
“We are impressed by the trend to notable efficacy in well-defined, well-balanced subgroups of HR-MDS patients with very poor prognosis. Based on these findings, we have designed the new phase 3 INSPIRE study with IV rigosertib, which is currently enrolling patients.”
INSPIRE is a randomized, controlled study designed to assess the efficacy and safety of IV rigosertib in HR-MDS patients under 80 years of age who had progressed on, failed to respond to, or relapsed after previous treatment with an HMA within the first 9 months of HMA treatment initiation.
The trial is expected to enroll approximately 225 patients, who will be randomized at a 2:1 ratio into 2 treatment arms: IV rigosertib plus BSC versus physician’s choice plus BSC. The primary endpoint is OS. Full details on the trial can be found on clinicaltrials.gov (NCT02562443). ![]()

Results of a phase 3 trial suggest the small-molecule inhibitor rigosertib may improve overall survival (OS) in some patients with higher-risk myelodysplastic syndromes (HR-MDS).
Overall, researchers found no significant difference in OS between patients who received rigosertib and those who received best supportive care (BSC).
However, the data indicate that rigosertib can confer a survival benefit in certain subgroups of HR-MDS patients.
The results of this trial, known as ONTIME, were published in The Lancet Oncology. The trial was sponsored by Onconova Therapeutics, Inc., the company developing rigosertib.
The trial enrolled 299 HR-MDS patients. They had refractory anemia with excess blasts (RAEB)-1, RAEB-2, RAEB-t, or chronic myelomonocytic leukemia based on local site assessment. They had all failed treatment with a hypomethylating agent (HMA) in the past 2 years.
The patients were randomized (2:1) to receive rigosertib at 1800 mg per 24 hours via 72-hour continuous intravenous (IV) infusion, administered every other week (n=199), or BSC with or without low-dose cytarabine (n=100).
At a median follow-up of 19.5 months, there was no significant difference in OS between the treatment arms. The median OS was 8.2 months in the rigosertib arm and 5.9 months in the BSC arm. The hazard ratio (HR) was 0.87 (P=0.33).
However, the researchers said that subgroup analyses suggested rigosertib may provide a survival benefit over BSC in some HR-MDS patients. This includes:
- Patients younger than 75 years of age (HR=0.55, P=0.0010)
- Patients who received HMA therapy for 9 months or fewer (HR=0.54, P=0.0016)
- Patients with primary, rather than secondary, HMA failure (HR=0.72, P=0.060)
- Patients who were classified as “very high risk” according to the Revised International Prognostic Scoring System (HR=0.61, P=0.015)
- Patients with monosomy 7 (HR=0.26, P=0.0041)
- Patients with trisomy 8 (HR=0.28, P=0.0083).
The most common grade 3 or higher adverse events—in the rigosertib and BSC arms, respectively—were anemia (18% vs 8%), thrombocytopenia (19% vs 7%), neutropenia (17% vs 8%), febrile neutropenia (12% vs 11%), and pneumonia (12% vs 11%).
Twenty-two percent of patients in the rigosertib arm and 33% in the BSC arm died due to adverse events. Three deaths were attributed to rigosertib.
“Rigosertib was well-tolerated in patients with a high unmet medical need who have no approved therapeutic options,” said study author Guillermo Garcia-Manero, MD, of The University of Texas MD Anderson Cancer Center in Houston.
“We are impressed by the trend to notable efficacy in well-defined, well-balanced subgroups of HR-MDS patients with very poor prognosis. Based on these findings, we have designed the new phase 3 INSPIRE study with IV rigosertib, which is currently enrolling patients.”
INSPIRE is a randomized, controlled study designed to assess the efficacy and safety of IV rigosertib in HR-MDS patients under 80 years of age who had progressed on, failed to respond to, or relapsed after previous treatment with an HMA within the first 9 months of HMA treatment initiation.
The trial is expected to enroll approximately 225 patients, who will be randomized at a 2:1 ratio into 2 treatment arms: IV rigosertib plus BSC versus physician’s choice plus BSC. The primary endpoint is OS. Full details on the trial can be found on clinicaltrials.gov (NCT02562443). ![]()
FDA approves product for hemophilia A

Photo by Bill Branson
The US Food and Drug Administration (FDA) has approved the recombinant antihemophilic factor Kovaltry for the treatment of adults and children with hemophilia A.
Kovaltry is an unmodified, full-length factor VIII compound indicated for on-demand treatment and control of bleeding episodes, perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes.
Dosing of Kovaltry should be individualized based on each patient’s clinical response.
The recommended dosing for adults and adolescents is 20 to 40 IU per kg of body weight 2 or 3 times per week. The recommended dosing for children age 12 and younger is 25 to 50 IU per kg of body weight twice weekly, 3 times weekly, or every other day, according to individual requirements.
For more details, see the full prescribing information.
The FDA’s approval of Kovaltry is based on results from the LEOPOLD trials—3 multinational trials of patients with severe hemophilia A. The trials were supported by Bayer HealthCare AG, the company developing Kovaltry.
LEOPOLD I
LEOPOLD I is an open-label, cross-over, phase 3 study of males, ages 12 to 65, with severe hemophilia A. Sixty-two patients were assigned to either 2- or 3-times-weekly dosing with Kovaltry, based on each patient’s phenotype, prior bleeding history, and other factors.
The median annualized bleeding rate (ABR) was 1.0 for all the patients who received Kovaltry prophylaxis, 1.0 for patients who received twice-weekly prophylaxis, and 2.0 for patients who received thrice-weekly prophylaxis.
LEOPOLD II
LEOPOLD II is a randomized, cross-over, open-label trial conducted in males ages 12 to 65. In this phase 3 study, 80 subjects were randomized to receive Kovaltry as a low-dose prophylaxis regimen (n=28) twice per week, high-dose prophylaxis (n=31) 3 times a week, or on-demand treatment (n=21).
The median ABR was significantly lower in patients who received either prophylactic regimen than those who received on-demand treatment—2.0 and 60.0, respectively (P<0.0001). The median ABR was 4.0 for patients who received twice-weekly prophylaxis and 2.0 for patients who received thrice-weekly prophylaxis.
LEOPOLD Kids
LEOPOLD Kids is an open-label, non-randomized, phase 3 study designed to evaluate Kovaltry in children age 12 and younger. The study is divided into 2 parts. Part A enrolled only previously treated children, and part B, which is ongoing, includes only untreated children.
For part A, 51 children received Kovaltry twice a week, 3 times a week, or every other day (according to investigator decision) for at least 50 exposure days. The median ABR within 48 hours of prophylactic injection was 0, and the median ABR independent of the time of injection was 1.9.
Safety results
For all 3 trials, 193 patients were evaluable for safety. Adverse reactions were defined as treatment-emergent adverse events with at least a reasonable suspected causal relationship to Kovaltry.
The researchers said the frequency, type, and severity of adverse reactions in children were similar to those observed in adults and adolescents.
The adverse reactions included headache (7.3%), pyrexia (4.1%), pruritus (3.1%), injection site reactions (2.6%), insomnia (2.6%), rash (2.6%), abdominal pain (2.1%), dyspepsia (2.1%), abdominal discomfort (1.6%), lymphadenopathy (1%), dizziness (1%), allergic dermatitis (1%), heart palpitations (1%), sinus tachycardia (1%), chest discomfort (1%), hypersensitivity (0.5%), dysgeusia (0.5%), urticaria (0.5%), and flushing (0.5%).
None of the patients developed factor VIII inhibitors. ![]()

Photo by Bill Branson
The US Food and Drug Administration (FDA) has approved the recombinant antihemophilic factor Kovaltry for the treatment of adults and children with hemophilia A.
Kovaltry is an unmodified, full-length factor VIII compound indicated for on-demand treatment and control of bleeding episodes, perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes.
Dosing of Kovaltry should be individualized based on each patient’s clinical response.
The recommended dosing for adults and adolescents is 20 to 40 IU per kg of body weight 2 or 3 times per week. The recommended dosing for children age 12 and younger is 25 to 50 IU per kg of body weight twice weekly, 3 times weekly, or every other day, according to individual requirements.
For more details, see the full prescribing information.
The FDA’s approval of Kovaltry is based on results from the LEOPOLD trials—3 multinational trials of patients with severe hemophilia A. The trials were supported by Bayer HealthCare AG, the company developing Kovaltry.
LEOPOLD I
LEOPOLD I is an open-label, cross-over, phase 3 study of males, ages 12 to 65, with severe hemophilia A. Sixty-two patients were assigned to either 2- or 3-times-weekly dosing with Kovaltry, based on each patient’s phenotype, prior bleeding history, and other factors.
The median annualized bleeding rate (ABR) was 1.0 for all the patients who received Kovaltry prophylaxis, 1.0 for patients who received twice-weekly prophylaxis, and 2.0 for patients who received thrice-weekly prophylaxis.
LEOPOLD II
LEOPOLD II is a randomized, cross-over, open-label trial conducted in males ages 12 to 65. In this phase 3 study, 80 subjects were randomized to receive Kovaltry as a low-dose prophylaxis regimen (n=28) twice per week, high-dose prophylaxis (n=31) 3 times a week, or on-demand treatment (n=21).
The median ABR was significantly lower in patients who received either prophylactic regimen than those who received on-demand treatment—2.0 and 60.0, respectively (P<0.0001). The median ABR was 4.0 for patients who received twice-weekly prophylaxis and 2.0 for patients who received thrice-weekly prophylaxis.
LEOPOLD Kids
LEOPOLD Kids is an open-label, non-randomized, phase 3 study designed to evaluate Kovaltry in children age 12 and younger. The study is divided into 2 parts. Part A enrolled only previously treated children, and part B, which is ongoing, includes only untreated children.
For part A, 51 children received Kovaltry twice a week, 3 times a week, or every other day (according to investigator decision) for at least 50 exposure days. The median ABR within 48 hours of prophylactic injection was 0, and the median ABR independent of the time of injection was 1.9.
Safety results
For all 3 trials, 193 patients were evaluable for safety. Adverse reactions were defined as treatment-emergent adverse events with at least a reasonable suspected causal relationship to Kovaltry.
The researchers said the frequency, type, and severity of adverse reactions in children were similar to those observed in adults and adolescents.
The adverse reactions included headache (7.3%), pyrexia (4.1%), pruritus (3.1%), injection site reactions (2.6%), insomnia (2.6%), rash (2.6%), abdominal pain (2.1%), dyspepsia (2.1%), abdominal discomfort (1.6%), lymphadenopathy (1%), dizziness (1%), allergic dermatitis (1%), heart palpitations (1%), sinus tachycardia (1%), chest discomfort (1%), hypersensitivity (0.5%), dysgeusia (0.5%), urticaria (0.5%), and flushing (0.5%).
None of the patients developed factor VIII inhibitors. ![]()

Photo by Bill Branson
The US Food and Drug Administration (FDA) has approved the recombinant antihemophilic factor Kovaltry for the treatment of adults and children with hemophilia A.
Kovaltry is an unmodified, full-length factor VIII compound indicated for on-demand treatment and control of bleeding episodes, perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes.
Dosing of Kovaltry should be individualized based on each patient’s clinical response.
The recommended dosing for adults and adolescents is 20 to 40 IU per kg of body weight 2 or 3 times per week. The recommended dosing for children age 12 and younger is 25 to 50 IU per kg of body weight twice weekly, 3 times weekly, or every other day, according to individual requirements.
For more details, see the full prescribing information.
The FDA’s approval of Kovaltry is based on results from the LEOPOLD trials—3 multinational trials of patients with severe hemophilia A. The trials were supported by Bayer HealthCare AG, the company developing Kovaltry.
LEOPOLD I
LEOPOLD I is an open-label, cross-over, phase 3 study of males, ages 12 to 65, with severe hemophilia A. Sixty-two patients were assigned to either 2- or 3-times-weekly dosing with Kovaltry, based on each patient’s phenotype, prior bleeding history, and other factors.
The median annualized bleeding rate (ABR) was 1.0 for all the patients who received Kovaltry prophylaxis, 1.0 for patients who received twice-weekly prophylaxis, and 2.0 for patients who received thrice-weekly prophylaxis.
LEOPOLD II
LEOPOLD II is a randomized, cross-over, open-label trial conducted in males ages 12 to 65. In this phase 3 study, 80 subjects were randomized to receive Kovaltry as a low-dose prophylaxis regimen (n=28) twice per week, high-dose prophylaxis (n=31) 3 times a week, or on-demand treatment (n=21).
The median ABR was significantly lower in patients who received either prophylactic regimen than those who received on-demand treatment—2.0 and 60.0, respectively (P<0.0001). The median ABR was 4.0 for patients who received twice-weekly prophylaxis and 2.0 for patients who received thrice-weekly prophylaxis.
LEOPOLD Kids
LEOPOLD Kids is an open-label, non-randomized, phase 3 study designed to evaluate Kovaltry in children age 12 and younger. The study is divided into 2 parts. Part A enrolled only previously treated children, and part B, which is ongoing, includes only untreated children.
For part A, 51 children received Kovaltry twice a week, 3 times a week, or every other day (according to investigator decision) for at least 50 exposure days. The median ABR within 48 hours of prophylactic injection was 0, and the median ABR independent of the time of injection was 1.9.
Safety results
For all 3 trials, 193 patients were evaluable for safety. Adverse reactions were defined as treatment-emergent adverse events with at least a reasonable suspected causal relationship to Kovaltry.
The researchers said the frequency, type, and severity of adverse reactions in children were similar to those observed in adults and adolescents.
The adverse reactions included headache (7.3%), pyrexia (4.1%), pruritus (3.1%), injection site reactions (2.6%), insomnia (2.6%), rash (2.6%), abdominal pain (2.1%), dyspepsia (2.1%), abdominal discomfort (1.6%), lymphadenopathy (1%), dizziness (1%), allergic dermatitis (1%), heart palpitations (1%), sinus tachycardia (1%), chest discomfort (1%), hypersensitivity (0.5%), dysgeusia (0.5%), urticaria (0.5%), and flushing (0.5%).
None of the patients developed factor VIII inhibitors. ![]()
Inhibitor exhibits activity against resistant AML

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models.
Bloodstream infection linked to antinausea drug
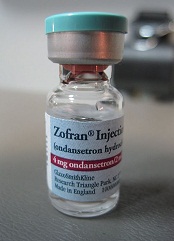
Researchers say they have discovered the source of a bloodstream infection observed in more than 50 South American cancer patients.
Using whole-genome sequence typing (WGST), the team was able to link infection with the fungus Sarocladium kiliense to a tainted antinausea medication, ondansetron, that was given to cancer patients in Chile and Colombia.
This work is described in Emerging Infectious Diseases.
“Contamination of medical products, particularly with environmental fungi, poses growing concern and a public health threat, especially in vulnerable populations such as cancer patients,” said study author David Engelthaler, PhD, of The Translational Genomics Research Institute in Flagstaff, Arizona.
“Increased vigilance and the use of advanced technologies are needed to rapidly identify the likely sources of infection to efficiently guide epidemiologic investigations and initiate appropriate control measures.”
The S kiliense bloodstream-infection outbreak, which occurred from June 2013 through January 2014, included a cluster of cases at 8 hospitals in Santiago, Chile.
All of the patients received the same 4 intravenous medications. But only the antinausea medication ondansetron was given exclusively to cancer patients.
All of the patients infected with S kiliense received ondansetron from the same source, a pharmaceutical company called Vitrofarma SA (specifically, Plant No. 8 in Bogotá, Colombia). The drug was imported by LabVitales Chile SA and distributed by Pharma Isa Ltda.
Two of 3 lots of unopened ondansetron, tested by the Chilean Ministry of Health, yielded vials contaminated with S kiliense, forcing a recall of all ondansetron in Chile that was made by Vitrofarma SA.
Subsequently, Colombian officials discovered 14 other cases in which patients who were given ondansetron from Vitrofarma SA were infected with S kiliense.
S kiliense has been implicated in healthcare-related infections before, but the lack of available typing methods has precluded the ability to substantiate sources.
“The use of WGST to investigate fungal outbreaks has become integral to epidemiologic investigations,” Dr Engelthaler said. “Our WGST analysis demonstrated that the patient isolates from Chile and Colombia were nearly genetically indistinguishable from those recovered from the unopened medication vials, indicating the likely presence of a single-source infection.”

Researchers say they have discovered the source of a bloodstream infection observed in more than 50 South American cancer patients.
Using whole-genome sequence typing (WGST), the team was able to link infection with the fungus Sarocladium kiliense to a tainted antinausea medication, ondansetron, that was given to cancer patients in Chile and Colombia.
This work is described in Emerging Infectious Diseases.
“Contamination of medical products, particularly with environmental fungi, poses growing concern and a public health threat, especially in vulnerable populations such as cancer patients,” said study author David Engelthaler, PhD, of The Translational Genomics Research Institute in Flagstaff, Arizona.
“Increased vigilance and the use of advanced technologies are needed to rapidly identify the likely sources of infection to efficiently guide epidemiologic investigations and initiate appropriate control measures.”
The S kiliense bloodstream-infection outbreak, which occurred from June 2013 through January 2014, included a cluster of cases at 8 hospitals in Santiago, Chile.
All of the patients received the same 4 intravenous medications. But only the antinausea medication ondansetron was given exclusively to cancer patients.
All of the patients infected with S kiliense received ondansetron from the same source, a pharmaceutical company called Vitrofarma SA (specifically, Plant No. 8 in Bogotá, Colombia). The drug was imported by LabVitales Chile SA and distributed by Pharma Isa Ltda.
Two of 3 lots of unopened ondansetron, tested by the Chilean Ministry of Health, yielded vials contaminated with S kiliense, forcing a recall of all ondansetron in Chile that was made by Vitrofarma SA.
Subsequently, Colombian officials discovered 14 other cases in which patients who were given ondansetron from Vitrofarma SA were infected with S kiliense.
S kiliense has been implicated in healthcare-related infections before, but the lack of available typing methods has precluded the ability to substantiate sources.
“The use of WGST to investigate fungal outbreaks has become integral to epidemiologic investigations,” Dr Engelthaler said. “Our WGST analysis demonstrated that the patient isolates from Chile and Colombia were nearly genetically indistinguishable from those recovered from the unopened medication vials, indicating the likely presence of a single-source infection.”

Researchers say they have discovered the source of a bloodstream infection observed in more than 50 South American cancer patients.
Using whole-genome sequence typing (WGST), the team was able to link infection with the fungus Sarocladium kiliense to a tainted antinausea medication, ondansetron, that was given to cancer patients in Chile and Colombia.
This work is described in Emerging Infectious Diseases.
“Contamination of medical products, particularly with environmental fungi, poses growing concern and a public health threat, especially in vulnerable populations such as cancer patients,” said study author David Engelthaler, PhD, of The Translational Genomics Research Institute in Flagstaff, Arizona.
“Increased vigilance and the use of advanced technologies are needed to rapidly identify the likely sources of infection to efficiently guide epidemiologic investigations and initiate appropriate control measures.”
The S kiliense bloodstream-infection outbreak, which occurred from June 2013 through January 2014, included a cluster of cases at 8 hospitals in Santiago, Chile.
All of the patients received the same 4 intravenous medications. But only the antinausea medication ondansetron was given exclusively to cancer patients.
All of the patients infected with S kiliense received ondansetron from the same source, a pharmaceutical company called Vitrofarma SA (specifically, Plant No. 8 in Bogotá, Colombia). The drug was imported by LabVitales Chile SA and distributed by Pharma Isa Ltda.
Two of 3 lots of unopened ondansetron, tested by the Chilean Ministry of Health, yielded vials contaminated with S kiliense, forcing a recall of all ondansetron in Chile that was made by Vitrofarma SA.
Subsequently, Colombian officials discovered 14 other cases in which patients who were given ondansetron from Vitrofarma SA were infected with S kiliense.
S kiliense has been implicated in healthcare-related infections before, but the lack of available typing methods has precluded the ability to substantiate sources.
“The use of WGST to investigate fungal outbreaks has become integral to epidemiologic investigations,” Dr Engelthaler said. “Our WGST analysis demonstrated that the patient isolates from Chile and Colombia were nearly genetically indistinguishable from those recovered from the unopened medication vials, indicating the likely presence of a single-source infection.”
FDA calls hospital-based Zika test ‘high risk’

Photo by Juan D. Alfonso
The US Food and Drug Administration (FDA) has deemed a hospital-based test for the Zika virus “high risk,” as the test has not been cleared by the FDA.
The test was developed by scientists at Texas Children’s Hospital and Houston Methodist Hospital. It has been available at both hospitals since last month.
The FDA has requested more information on the test but has not asked the hospitals to stop using it.
According to the hospitals, the test identifies virus-specific RNA sequences to detect the Zika virus. It can distinguish Zika infection from dengue, West Nile, or chikungunya infections. And it can be performed on blood, amniotic fluid, urine, or spinal fluid.
In a letter to the hospitals, the FDA said this test appears to meet the definition of a device, as defined in section 201(h) of the Federal Food Drug and Cosmetic Act. Yet the test has not been granted premarket clearance, approval, or Emergency Use Authorization review by the FDA.
Therefore, the FDA has asked for information on the test’s design, validation, and performance characteristics. The agency said the Centers for Disease Control and Prevention (CDC) and the Centers for Medicare & Medicaid Services asked the FDA to review the science behind the test.
The FDA has not asked the hospitals to stop using the test while the review is underway, according to a statement from Texas Children’s Hospital.
Nevertheless, the Association for Molecular Pathology (AMP) said it is “concerned and disappointed” to see the FDA taking enforcement action regarding this test. The AMP said these types of tests are critical for patient care and should be made available to patients in need.
In fact, the AMP said this is an example of how FDA regulation of laboratory developed procedures would hinder patient access to vital medical services. That’s because the FDA’s Emergency Use Authorization for antibody testing at the CDC or state public health labs does not provide results in the timely fashion needed for immediate patient care.
The FDA recently issued Emergency Use Authorization for the Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA), which was developed by the CDC.
The test was distributed to labs in the US and abroad, but it was not made available in US hospitals or other primary care settings.

Photo by Juan D. Alfonso
The US Food and Drug Administration (FDA) has deemed a hospital-based test for the Zika virus “high risk,” as the test has not been cleared by the FDA.
The test was developed by scientists at Texas Children’s Hospital and Houston Methodist Hospital. It has been available at both hospitals since last month.
The FDA has requested more information on the test but has not asked the hospitals to stop using it.
According to the hospitals, the test identifies virus-specific RNA sequences to detect the Zika virus. It can distinguish Zika infection from dengue, West Nile, or chikungunya infections. And it can be performed on blood, amniotic fluid, urine, or spinal fluid.
In a letter to the hospitals, the FDA said this test appears to meet the definition of a device, as defined in section 201(h) of the Federal Food Drug and Cosmetic Act. Yet the test has not been granted premarket clearance, approval, or Emergency Use Authorization review by the FDA.
Therefore, the FDA has asked for information on the test’s design, validation, and performance characteristics. The agency said the Centers for Disease Control and Prevention (CDC) and the Centers for Medicare & Medicaid Services asked the FDA to review the science behind the test.
The FDA has not asked the hospitals to stop using the test while the review is underway, according to a statement from Texas Children’s Hospital.
Nevertheless, the Association for Molecular Pathology (AMP) said it is “concerned and disappointed” to see the FDA taking enforcement action regarding this test. The AMP said these types of tests are critical for patient care and should be made available to patients in need.
In fact, the AMP said this is an example of how FDA regulation of laboratory developed procedures would hinder patient access to vital medical services. That’s because the FDA’s Emergency Use Authorization for antibody testing at the CDC or state public health labs does not provide results in the timely fashion needed for immediate patient care.
The FDA recently issued Emergency Use Authorization for the Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA), which was developed by the CDC.
The test was distributed to labs in the US and abroad, but it was not made available in US hospitals or other primary care settings.

Photo by Juan D. Alfonso
The US Food and Drug Administration (FDA) has deemed a hospital-based test for the Zika virus “high risk,” as the test has not been cleared by the FDA.
The test was developed by scientists at Texas Children’s Hospital and Houston Methodist Hospital. It has been available at both hospitals since last month.
The FDA has requested more information on the test but has not asked the hospitals to stop using it.
According to the hospitals, the test identifies virus-specific RNA sequences to detect the Zika virus. It can distinguish Zika infection from dengue, West Nile, or chikungunya infections. And it can be performed on blood, amniotic fluid, urine, or spinal fluid.
In a letter to the hospitals, the FDA said this test appears to meet the definition of a device, as defined in section 201(h) of the Federal Food Drug and Cosmetic Act. Yet the test has not been granted premarket clearance, approval, or Emergency Use Authorization review by the FDA.
Therefore, the FDA has asked for information on the test’s design, validation, and performance characteristics. The agency said the Centers for Disease Control and Prevention (CDC) and the Centers for Medicare & Medicaid Services asked the FDA to review the science behind the test.
The FDA has not asked the hospitals to stop using the test while the review is underway, according to a statement from Texas Children’s Hospital.
Nevertheless, the Association for Molecular Pathology (AMP) said it is “concerned and disappointed” to see the FDA taking enforcement action regarding this test. The AMP said these types of tests are critical for patient care and should be made available to patients in need.
In fact, the AMP said this is an example of how FDA regulation of laboratory developed procedures would hinder patient access to vital medical services. That’s because the FDA’s Emergency Use Authorization for antibody testing at the CDC or state public health labs does not provide results in the timely fashion needed for immediate patient care.
The FDA recently issued Emergency Use Authorization for the Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA), which was developed by the CDC.
The test was distributed to labs in the US and abroad, but it was not made available in US hospitals or other primary care settings.
Severe hemophilia still tough to manage, study shows

A large study suggests that, despite treatment advances, men with severe hemophilia have not seen great reductions in bleeding events.
Data on more than 7000 men with hemophilia revealed substantial differences in the care received by men born before 1958 and those born between 1983
and 1992.
However, the data also showed that frequent bleeding was common in patients with severe hemophilia, regardless of when they were born.
These data were published in Blood.
“Our analysis provides a snapshot of how improvements in care have translated into outcomes across different generations of men with hemophilia,” said study author Paul E. Monahan, MD, of The University of North Carolina-Chapel Hill.
“While there is reason to be pleased with the progress we’ve made, our data show some surprising deficits and suggest that efforts are needed to more consistently apply the integration of standard-of-care multidisciplinary services and preventive blood clotting factor treatments to further normalize the lives of men living with hemophilia.”
Dr Monahan and his colleagues analyzed data on 7486 men—4899 with severe hemophilia (65.4%), 2587 with mild hemophilia (34.6%), 6094 with hemophilia A (81.4%), and 1392 with hemophilia B (18.6%).
The data were collected prospectively by the US Centers for Disease Control and Prevention and 130 federally supported Hemophilia Treatment Centers (HTCs) between 1998 and 2011. This represents the largest database of men living with hemophilia.
The researchers grouped the men into 4 eras (birth cohorts) to evaluate how outcomes—access to care, physical and social functioning, complications, and mortality—have changed over the last 50 years.
The cohorts were as follows:
- Era A: born before 1958 (median age 58)
- Era B: born between 1958 and 1975 (median age 40)
- Era C: born between 1976 and 1982 (median age 28)
- Era D: born between 1983 and 1992 (median age 21).
Access to care
The researchers found that the proportion of men who started home infusions before age 6 was far greater in Era D than Era A—50.4% and 2%, respectively. And the proportion of patients reporting a first HTC visit before age 2 rose nearly 10-fold from Era A to Era D—8.8% and 100%, respectively.
In addition, the use of a continuous prophylactic regimen was nearly 3-fold greater in Era D than Era A—46.7% and 16%, respectively.
However, patients in the youngest 2 birth cohorts were more likely than their older counterparts to be uninsured. The proportion of uninsured patients was 16.4% in Era D, 20.5% in Era C, 11.1% in Era B, and 5.7% in Era A.
Bleeding events
The proportion of patients reporting frequent bleeds decreased from Era A to Era D. However, frequent bleeding was common in men with severe hemophilia regardless of when they were born.
Even men from Era D—who had access to effective, safe clotting therapies and multidisciplinary care throughout their lifetimes—reported frequent bleeds.
The proportion of patients with severe hemophilia reporting 5 or more joint bleeds in the last 6 months was 42.6% in Era A and 35.5% in Era D. The proportion of patients with a joint affected by recurrent bleeding was 32.6% and 24.9%, respectively.
Functioning
The researchers also discovered that patients with severe hemophilia were 3 times as likely to report limitations in their activities or to be disabled, when compared to patients with mild hemophilia, regardless of when they were born.
Still, men from Era A were more likely to report limitations in their overall activity level—68.8% of severe hemophilia patients and 21.1% of mild hemophilia patients—than men from Era D—14.9% of severe hemophilia patients and 4.3% of mild hemophilia patients.
Men with severe hemophilia were more likely than men with mild hemophilia to report missing more than 10 days of work or school during the previous year. In Era A, the proportions were 6.9% and 2.6%, respectively. In Era D, the proportions were 5.6% and 3%, respectively.
“Clear disparities remain in terms of frequent bleeding and disability between men with severe hemophilia and mild hemophilia across every decade of adult life,” Dr Monahan said.
“We thought the difference in functional outcomes would have narrowed over the years. That is, men with severe hemophilia should look more like those with
mild disorder, given improved therapeutics and access to care, but this wasn’t the case.”
“What needs examination is why, despite widespread availability of preventive and on-demand therapies for home use, we still see disparities. It speaks to the need for continued disease surveillance to monitor and inform hemophilia interventions and outcomes.”
Mortality
There were 551 deaths during the study period. The Era A and B cohorts accounted for 82% of the deaths in the severe hemophilia population and 96% of the deaths in the mild hemophilia population.
The researchers noted that liver failure has surpassed bleeding issues and HIV as the leading cause of death among US men with hemophilia.
Although there were no liver-related deaths in the 2 youngest cohorts, liver failure was the most commonly reported cause of death across all the cohorts, for both severe hemophilia (33% of deaths) and mild hemophilia (26% of deaths).
The researchers said this finding underscores the need to swiftly evaluate and treat HCV infections.
“Liver disease worsens bleeding, so eradicating hepatitis C infections needs to be a priority, especially as we now have remarkably effective therapies,” Dr Monahan said.
Across all the birth cohorts, hemophilia-related deaths accounted for 14.6% of deaths in patients with severe hemophilia and 10.7% of deaths in those with mild hemophilia.

A large study suggests that, despite treatment advances, men with severe hemophilia have not seen great reductions in bleeding events.
Data on more than 7000 men with hemophilia revealed substantial differences in the care received by men born before 1958 and those born between 1983
and 1992.
However, the data also showed that frequent bleeding was common in patients with severe hemophilia, regardless of when they were born.
These data were published in Blood.
“Our analysis provides a snapshot of how improvements in care have translated into outcomes across different generations of men with hemophilia,” said study author Paul E. Monahan, MD, of The University of North Carolina-Chapel Hill.
“While there is reason to be pleased with the progress we’ve made, our data show some surprising deficits and suggest that efforts are needed to more consistently apply the integration of standard-of-care multidisciplinary services and preventive blood clotting factor treatments to further normalize the lives of men living with hemophilia.”
Dr Monahan and his colleagues analyzed data on 7486 men—4899 with severe hemophilia (65.4%), 2587 with mild hemophilia (34.6%), 6094 with hemophilia A (81.4%), and 1392 with hemophilia B (18.6%).
The data were collected prospectively by the US Centers for Disease Control and Prevention and 130 federally supported Hemophilia Treatment Centers (HTCs) between 1998 and 2011. This represents the largest database of men living with hemophilia.
The researchers grouped the men into 4 eras (birth cohorts) to evaluate how outcomes—access to care, physical and social functioning, complications, and mortality—have changed over the last 50 years.
The cohorts were as follows:
- Era A: born before 1958 (median age 58)
- Era B: born between 1958 and 1975 (median age 40)
- Era C: born between 1976 and 1982 (median age 28)
- Era D: born between 1983 and 1992 (median age 21).
Access to care
The researchers found that the proportion of men who started home infusions before age 6 was far greater in Era D than Era A—50.4% and 2%, respectively. And the proportion of patients reporting a first HTC visit before age 2 rose nearly 10-fold from Era A to Era D—8.8% and 100%, respectively.
In addition, the use of a continuous prophylactic regimen was nearly 3-fold greater in Era D than Era A—46.7% and 16%, respectively.
However, patients in the youngest 2 birth cohorts were more likely than their older counterparts to be uninsured. The proportion of uninsured patients was 16.4% in Era D, 20.5% in Era C, 11.1% in Era B, and 5.7% in Era A.
Bleeding events
The proportion of patients reporting frequent bleeds decreased from Era A to Era D. However, frequent bleeding was common in men with severe hemophilia regardless of when they were born.
Even men from Era D—who had access to effective, safe clotting therapies and multidisciplinary care throughout their lifetimes—reported frequent bleeds.
The proportion of patients with severe hemophilia reporting 5 or more joint bleeds in the last 6 months was 42.6% in Era A and 35.5% in Era D. The proportion of patients with a joint affected by recurrent bleeding was 32.6% and 24.9%, respectively.
Functioning
The researchers also discovered that patients with severe hemophilia were 3 times as likely to report limitations in their activities or to be disabled, when compared to patients with mild hemophilia, regardless of when they were born.
Still, men from Era A were more likely to report limitations in their overall activity level—68.8% of severe hemophilia patients and 21.1% of mild hemophilia patients—than men from Era D—14.9% of severe hemophilia patients and 4.3% of mild hemophilia patients.
Men with severe hemophilia were more likely than men with mild hemophilia to report missing more than 10 days of work or school during the previous year. In Era A, the proportions were 6.9% and 2.6%, respectively. In Era D, the proportions were 5.6% and 3%, respectively.
“Clear disparities remain in terms of frequent bleeding and disability between men with severe hemophilia and mild hemophilia across every decade of adult life,” Dr Monahan said.
“We thought the difference in functional outcomes would have narrowed over the years. That is, men with severe hemophilia should look more like those with
mild disorder, given improved therapeutics and access to care, but this wasn’t the case.”
“What needs examination is why, despite widespread availability of preventive and on-demand therapies for home use, we still see disparities. It speaks to the need for continued disease surveillance to monitor and inform hemophilia interventions and outcomes.”
Mortality
There were 551 deaths during the study period. The Era A and B cohorts accounted for 82% of the deaths in the severe hemophilia population and 96% of the deaths in the mild hemophilia population.
The researchers noted that liver failure has surpassed bleeding issues and HIV as the leading cause of death among US men with hemophilia.
Although there were no liver-related deaths in the 2 youngest cohorts, liver failure was the most commonly reported cause of death across all the cohorts, for both severe hemophilia (33% of deaths) and mild hemophilia (26% of deaths).
The researchers said this finding underscores the need to swiftly evaluate and treat HCV infections.
“Liver disease worsens bleeding, so eradicating hepatitis C infections needs to be a priority, especially as we now have remarkably effective therapies,” Dr Monahan said.
Across all the birth cohorts, hemophilia-related deaths accounted for 14.6% of deaths in patients with severe hemophilia and 10.7% of deaths in those with mild hemophilia.

A large study suggests that, despite treatment advances, men with severe hemophilia have not seen great reductions in bleeding events.
Data on more than 7000 men with hemophilia revealed substantial differences in the care received by men born before 1958 and those born between 1983
and 1992.
However, the data also showed that frequent bleeding was common in patients with severe hemophilia, regardless of when they were born.
These data were published in Blood.
“Our analysis provides a snapshot of how improvements in care have translated into outcomes across different generations of men with hemophilia,” said study author Paul E. Monahan, MD, of The University of North Carolina-Chapel Hill.
“While there is reason to be pleased with the progress we’ve made, our data show some surprising deficits and suggest that efforts are needed to more consistently apply the integration of standard-of-care multidisciplinary services and preventive blood clotting factor treatments to further normalize the lives of men living with hemophilia.”
Dr Monahan and his colleagues analyzed data on 7486 men—4899 with severe hemophilia (65.4%), 2587 with mild hemophilia (34.6%), 6094 with hemophilia A (81.4%), and 1392 with hemophilia B (18.6%).
The data were collected prospectively by the US Centers for Disease Control and Prevention and 130 federally supported Hemophilia Treatment Centers (HTCs) between 1998 and 2011. This represents the largest database of men living with hemophilia.
The researchers grouped the men into 4 eras (birth cohorts) to evaluate how outcomes—access to care, physical and social functioning, complications, and mortality—have changed over the last 50 years.
The cohorts were as follows:
- Era A: born before 1958 (median age 58)
- Era B: born between 1958 and 1975 (median age 40)
- Era C: born between 1976 and 1982 (median age 28)
- Era D: born between 1983 and 1992 (median age 21).
Access to care
The researchers found that the proportion of men who started home infusions before age 6 was far greater in Era D than Era A—50.4% and 2%, respectively. And the proportion of patients reporting a first HTC visit before age 2 rose nearly 10-fold from Era A to Era D—8.8% and 100%, respectively.
In addition, the use of a continuous prophylactic regimen was nearly 3-fold greater in Era D than Era A—46.7% and 16%, respectively.
However, patients in the youngest 2 birth cohorts were more likely than their older counterparts to be uninsured. The proportion of uninsured patients was 16.4% in Era D, 20.5% in Era C, 11.1% in Era B, and 5.7% in Era A.
Bleeding events
The proportion of patients reporting frequent bleeds decreased from Era A to Era D. However, frequent bleeding was common in men with severe hemophilia regardless of when they were born.
Even men from Era D—who had access to effective, safe clotting therapies and multidisciplinary care throughout their lifetimes—reported frequent bleeds.
The proportion of patients with severe hemophilia reporting 5 or more joint bleeds in the last 6 months was 42.6% in Era A and 35.5% in Era D. The proportion of patients with a joint affected by recurrent bleeding was 32.6% and 24.9%, respectively.
Functioning
The researchers also discovered that patients with severe hemophilia were 3 times as likely to report limitations in their activities or to be disabled, when compared to patients with mild hemophilia, regardless of when they were born.
Still, men from Era A were more likely to report limitations in their overall activity level—68.8% of severe hemophilia patients and 21.1% of mild hemophilia patients—than men from Era D—14.9% of severe hemophilia patients and 4.3% of mild hemophilia patients.
Men with severe hemophilia were more likely than men with mild hemophilia to report missing more than 10 days of work or school during the previous year. In Era A, the proportions were 6.9% and 2.6%, respectively. In Era D, the proportions were 5.6% and 3%, respectively.
“Clear disparities remain in terms of frequent bleeding and disability between men with severe hemophilia and mild hemophilia across every decade of adult life,” Dr Monahan said.
“We thought the difference in functional outcomes would have narrowed over the years. That is, men with severe hemophilia should look more like those with
mild disorder, given improved therapeutics and access to care, but this wasn’t the case.”
“What needs examination is why, despite widespread availability of preventive and on-demand therapies for home use, we still see disparities. It speaks to the need for continued disease surveillance to monitor and inform hemophilia interventions and outcomes.”
Mortality
There were 551 deaths during the study period. The Era A and B cohorts accounted for 82% of the deaths in the severe hemophilia population and 96% of the deaths in the mild hemophilia population.
The researchers noted that liver failure has surpassed bleeding issues and HIV as the leading cause of death among US men with hemophilia.
Although there were no liver-related deaths in the 2 youngest cohorts, liver failure was the most commonly reported cause of death across all the cohorts, for both severe hemophilia (33% of deaths) and mild hemophilia (26% of deaths).
The researchers said this finding underscores the need to swiftly evaluate and treat HCV infections.
“Liver disease worsens bleeding, so eradicating hepatitis C infections needs to be a priority, especially as we now have remarkably effective therapies,” Dr Monahan said.
Across all the birth cohorts, hemophilia-related deaths accounted for 14.6% of deaths in patients with severe hemophilia and 10.7% of deaths in those with mild hemophilia.