User login
Gut microbes affect platelet function, thrombosis risk

Image by Andre E.X. Brown
New research indicates that gut microbes alter platelet function, which affects the risk of thrombosis and related events like heart attack and stroke.
When the nutrient choline, which is abundant in animal products like meat and egg yolk, is ingested, gut microbes play a role in breaking it down and producing the compound TMAO.
Recent studies have shown that blood TMAO levels are associated with a heightened risk of heart attack and stroke.
The new study, published in Cell, suggests that TMAO encourages hyper-reactive platelet function, thereby increasing the likelihood of thrombosis.
Researchers said this could be the mechanism by which TMAO increases the risk of heart attack and stroke. And these findings reveal a previously unrecognized mechanistic link between specific dietary nutrients, gut microbes, platelet function, and thrombosis risk.
“It is remarkable that gut microbes produce a compound that alters platelet function and thrombotic heart attack and stroke risk,” said study author Stanley Hazen, MD, PhD, of the Cleveland Clinic in Ohio.
“This new link helps explain how diet-induced TMAO generation is mechanistically linked to development of lethal adverse complications of heart disease.”
Dr Hazen and his colleagues first discovered a link between TMAO, gut microbes, and heart disease 5 years ago.
For the current study, the researchers analyzed blood levels of TMAO in more than 4000 patients and saw a significant correlation between higher TMAO and thrombosis potential. This led to the hypothesis that TMAO may directly impact platelet function.
Subsequent studies with both human platelets and animal models confirmed that TMAO makes platelets hyper-reactive, heightening thrombosis potential and accelerating clotting rates.
“We have shown that TMAO fundamentally alters calcium signaling within platelets,” Dr Hazen said. “When TMAO is elevated, platelet responsiveness to known triggers like thrombin, collagen, or ADP is heightened.”
“In general, there’s a broad range for how quickly different people will form clots. However, across the board, when TMAO is elevated, platelet responsiveness jumps to the hyper-reactive side of normal.”
Dr Hazen and his colleagues said these results suggest that lowering TMAO—via dietary manipulation, alteration in microbial community with a probiotic or prebiotic, or direct pharmacological inhibition of microbial enzymes involved in TMA production—may be a way to reduce the risk of thrombotic events.
They noted that, unlike current antiplatelet therapies, targeting TMAO would likely reduce platelet hyper-responsiveness to the normal range and not induce impairment in overall platelet function. So the intervention could attenuate pro-thrombotic tendencies without increasing the risk of bleeding complications. ![]()

Image by Andre E.X. Brown
New research indicates that gut microbes alter platelet function, which affects the risk of thrombosis and related events like heart attack and stroke.
When the nutrient choline, which is abundant in animal products like meat and egg yolk, is ingested, gut microbes play a role in breaking it down and producing the compound TMAO.
Recent studies have shown that blood TMAO levels are associated with a heightened risk of heart attack and stroke.
The new study, published in Cell, suggests that TMAO encourages hyper-reactive platelet function, thereby increasing the likelihood of thrombosis.
Researchers said this could be the mechanism by which TMAO increases the risk of heart attack and stroke. And these findings reveal a previously unrecognized mechanistic link between specific dietary nutrients, gut microbes, platelet function, and thrombosis risk.
“It is remarkable that gut microbes produce a compound that alters platelet function and thrombotic heart attack and stroke risk,” said study author Stanley Hazen, MD, PhD, of the Cleveland Clinic in Ohio.
“This new link helps explain how diet-induced TMAO generation is mechanistically linked to development of lethal adverse complications of heart disease.”
Dr Hazen and his colleagues first discovered a link between TMAO, gut microbes, and heart disease 5 years ago.
For the current study, the researchers analyzed blood levels of TMAO in more than 4000 patients and saw a significant correlation between higher TMAO and thrombosis potential. This led to the hypothesis that TMAO may directly impact platelet function.
Subsequent studies with both human platelets and animal models confirmed that TMAO makes platelets hyper-reactive, heightening thrombosis potential and accelerating clotting rates.
“We have shown that TMAO fundamentally alters calcium signaling within platelets,” Dr Hazen said. “When TMAO is elevated, platelet responsiveness to known triggers like thrombin, collagen, or ADP is heightened.”
“In general, there’s a broad range for how quickly different people will form clots. However, across the board, when TMAO is elevated, platelet responsiveness jumps to the hyper-reactive side of normal.”
Dr Hazen and his colleagues said these results suggest that lowering TMAO—via dietary manipulation, alteration in microbial community with a probiotic or prebiotic, or direct pharmacological inhibition of microbial enzymes involved in TMA production—may be a way to reduce the risk of thrombotic events.
They noted that, unlike current antiplatelet therapies, targeting TMAO would likely reduce platelet hyper-responsiveness to the normal range and not induce impairment in overall platelet function. So the intervention could attenuate pro-thrombotic tendencies without increasing the risk of bleeding complications. ![]()

Image by Andre E.X. Brown
New research indicates that gut microbes alter platelet function, which affects the risk of thrombosis and related events like heart attack and stroke.
When the nutrient choline, which is abundant in animal products like meat and egg yolk, is ingested, gut microbes play a role in breaking it down and producing the compound TMAO.
Recent studies have shown that blood TMAO levels are associated with a heightened risk of heart attack and stroke.
The new study, published in Cell, suggests that TMAO encourages hyper-reactive platelet function, thereby increasing the likelihood of thrombosis.
Researchers said this could be the mechanism by which TMAO increases the risk of heart attack and stroke. And these findings reveal a previously unrecognized mechanistic link between specific dietary nutrients, gut microbes, platelet function, and thrombosis risk.
“It is remarkable that gut microbes produce a compound that alters platelet function and thrombotic heart attack and stroke risk,” said study author Stanley Hazen, MD, PhD, of the Cleveland Clinic in Ohio.
“This new link helps explain how diet-induced TMAO generation is mechanistically linked to development of lethal adverse complications of heart disease.”
Dr Hazen and his colleagues first discovered a link between TMAO, gut microbes, and heart disease 5 years ago.
For the current study, the researchers analyzed blood levels of TMAO in more than 4000 patients and saw a significant correlation between higher TMAO and thrombosis potential. This led to the hypothesis that TMAO may directly impact platelet function.
Subsequent studies with both human platelets and animal models confirmed that TMAO makes platelets hyper-reactive, heightening thrombosis potential and accelerating clotting rates.
“We have shown that TMAO fundamentally alters calcium signaling within platelets,” Dr Hazen said. “When TMAO is elevated, platelet responsiveness to known triggers like thrombin, collagen, or ADP is heightened.”
“In general, there’s a broad range for how quickly different people will form clots. However, across the board, when TMAO is elevated, platelet responsiveness jumps to the hyper-reactive side of normal.”
Dr Hazen and his colleagues said these results suggest that lowering TMAO—via dietary manipulation, alteration in microbial community with a probiotic or prebiotic, or direct pharmacological inhibition of microbial enzymes involved in TMA production—may be a way to reduce the risk of thrombotic events.
They noted that, unlike current antiplatelet therapies, targeting TMAO would likely reduce platelet hyper-responsiveness to the normal range and not induce impairment in overall platelet function. So the intervention could attenuate pro-thrombotic tendencies without increasing the risk of bleeding complications. ![]()
Negative cancer trials have long-term impact

for a clinical trial
Photo by Esther Dyson
Cancer trials with negative results don’t make an immediate splash in the scientific literature, but they do have a long-term impact on research, according to a study published in JAMA Oncology.
Researchers found that first reports of positive phase 3 cancer trials were twice as likely as first reports of negative phase 3 cancer trials
to be cited in scientific journals.
But over time, when all articles associated with the trials were considered, the scientific impact of negative trials and positive trials was about the same.
“Negative trials aren’t scientific failures,” said study author Joseph Unger, PhD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
“We found that they have a positive, lasting impact on cancer research.”
Dr Unger and his colleagues analyzed every randomized, phase 3 cancer trial completed by the cooperative group SWOG from 1984 to 2014. This amounted to 94 studies involving 46,424 patients.
Of those 94 studies, 26 were positive, meaning that the treatment tested performed measurably better than the standard treatment at the time.
Analyses revealed that primary manuscripts first announcing these encouraging results were published in journals with higher impact factors and were cited twice as often as primary manuscripts of negative trials.
The mean 2-year impact factor of the journals was 28 for positive trials and 18 for negative trials (P=0.007). And the mean number of citations per year was 43 for positive trials and 21 for negative trials (P=0.03).
However, when the researchers looked at the number of citations from all primary and secondary manuscripts, they did not see a significant difference between positive and negative trials. The mean number of citations per year was 55 and 45, respectively (P=0.53).
“Negative trials matter because they tell us what doesn’t work, which can be as important as what does,” said study author Dawn Hershman, MD, of Columbia University Medical Center in New York, New York.
“Negative trials are also critical for secondary research, which mines existing trial data to answer new questions in cancer care and prevention. Negative trials are used frequently in secondary research and add great value to the scientific community.” ![]()

for a clinical trial
Photo by Esther Dyson
Cancer trials with negative results don’t make an immediate splash in the scientific literature, but they do have a long-term impact on research, according to a study published in JAMA Oncology.
Researchers found that first reports of positive phase 3 cancer trials were twice as likely as first reports of negative phase 3 cancer trials
to be cited in scientific journals.
But over time, when all articles associated with the trials were considered, the scientific impact of negative trials and positive trials was about the same.
“Negative trials aren’t scientific failures,” said study author Joseph Unger, PhD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
“We found that they have a positive, lasting impact on cancer research.”
Dr Unger and his colleagues analyzed every randomized, phase 3 cancer trial completed by the cooperative group SWOG from 1984 to 2014. This amounted to 94 studies involving 46,424 patients.
Of those 94 studies, 26 were positive, meaning that the treatment tested performed measurably better than the standard treatment at the time.
Analyses revealed that primary manuscripts first announcing these encouraging results were published in journals with higher impact factors and were cited twice as often as primary manuscripts of negative trials.
The mean 2-year impact factor of the journals was 28 for positive trials and 18 for negative trials (P=0.007). And the mean number of citations per year was 43 for positive trials and 21 for negative trials (P=0.03).
However, when the researchers looked at the number of citations from all primary and secondary manuscripts, they did not see a significant difference between positive and negative trials. The mean number of citations per year was 55 and 45, respectively (P=0.53).
“Negative trials matter because they tell us what doesn’t work, which can be as important as what does,” said study author Dawn Hershman, MD, of Columbia University Medical Center in New York, New York.
“Negative trials are also critical for secondary research, which mines existing trial data to answer new questions in cancer care and prevention. Negative trials are used frequently in secondary research and add great value to the scientific community.” ![]()

for a clinical trial
Photo by Esther Dyson
Cancer trials with negative results don’t make an immediate splash in the scientific literature, but they do have a long-term impact on research, according to a study published in JAMA Oncology.
Researchers found that first reports of positive phase 3 cancer trials were twice as likely as first reports of negative phase 3 cancer trials
to be cited in scientific journals.
But over time, when all articles associated with the trials were considered, the scientific impact of negative trials and positive trials was about the same.
“Negative trials aren’t scientific failures,” said study author Joseph Unger, PhD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
“We found that they have a positive, lasting impact on cancer research.”
Dr Unger and his colleagues analyzed every randomized, phase 3 cancer trial completed by the cooperative group SWOG from 1984 to 2014. This amounted to 94 studies involving 46,424 patients.
Of those 94 studies, 26 were positive, meaning that the treatment tested performed measurably better than the standard treatment at the time.
Analyses revealed that primary manuscripts first announcing these encouraging results were published in journals with higher impact factors and were cited twice as often as primary manuscripts of negative trials.
The mean 2-year impact factor of the journals was 28 for positive trials and 18 for negative trials (P=0.007). And the mean number of citations per year was 43 for positive trials and 21 for negative trials (P=0.03).
However, when the researchers looked at the number of citations from all primary and secondary manuscripts, they did not see a significant difference between positive and negative trials. The mean number of citations per year was 55 and 45, respectively (P=0.53).
“Negative trials matter because they tell us what doesn’t work, which can be as important as what does,” said study author Dawn Hershman, MD, of Columbia University Medical Center in New York, New York.
“Negative trials are also critical for secondary research, which mines existing trial data to answer new questions in cancer care and prevention. Negative trials are used frequently in secondary research and add great value to the scientific community.” ![]()
AEs prompt EMA review of idelalisib

Photo courtesy of
Gilead Sciences, Inc.
The European Medicines Agency (EMA) is reviewing the safety of idelalisib (Zydelig), a drug approved to treat chronic lymphocytic leukemia (CLL) and follicular lymphoma in the European Union (EU).
The European Commission (EC) requested the review because of serious adverse events (AEs), including deaths, reported in 3 clinical trials investigating idelalisib in combination with other drugs.
The AEs were mostly infection-related.
The EMA is reviewing data from these studies to assess whether the findings have any consequences for the authorized uses of idelalisib.
In the meantime, the EMA advises that patients starting or already on treatment with idelalisib be carefully monitored for signs of infections. If the drug is well tolerated, treatment should not be stopped.
The EMA is considering whether any other immediate measures are necessary during the review period. The agency said it will communicate further and keep doctors and patients informed as appropriate.
About idelalisib
In the EU, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients unsuitable for chemo-immunotherapy.
Idelalisib is also approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
About the trials
The trials in which patients have experienced serious AEs involve patients with CLL and indolent non-Hodgkin lymphoma (NHL).
In one trial (NCT01732926), researchers are evaluating idelalisib in combination with bendamustine and rituximab for previously treated indolent NHL.
In another (NCT01732913), researchers are testing idelalisib in combination with rituximab for previously treated indolent NHL.
And in the third (NCT01980888), researchers are evaluating idelalisib in combination with bendamustine and rituximab in patients with previously untreated CLL.
The EMA noted that these studies are investigating combinations of drugs that are currently not approved in the EU and include patients with disease characteristics different from those covered by the approved indications for idelalisib.
About the review
The EMA has begun the review of idelalisib at the request of the EC, under Article 20 of Directive 2001/83/EC.
The review is being carried out by the EMA’s Pharmacovigilance Risk Assessment Committee, the committee responsible for the evaluation of safety issues for human medicines, which will make a set of recommendations.
Those recommendations will then be forwarded to the Committee for Medicinal Products for Human Use, which is responsible for questions concerning medicines for human use and will adopt a final opinion on the safety of idelalisib.
The final stage of the review procedure is the EC’s adoption of a legally binding decision that is applicable in all EU member states. ![]()

Photo courtesy of
Gilead Sciences, Inc.
The European Medicines Agency (EMA) is reviewing the safety of idelalisib (Zydelig), a drug approved to treat chronic lymphocytic leukemia (CLL) and follicular lymphoma in the European Union (EU).
The European Commission (EC) requested the review because of serious adverse events (AEs), including deaths, reported in 3 clinical trials investigating idelalisib in combination with other drugs.
The AEs were mostly infection-related.
The EMA is reviewing data from these studies to assess whether the findings have any consequences for the authorized uses of idelalisib.
In the meantime, the EMA advises that patients starting or already on treatment with idelalisib be carefully monitored for signs of infections. If the drug is well tolerated, treatment should not be stopped.
The EMA is considering whether any other immediate measures are necessary during the review period. The agency said it will communicate further and keep doctors and patients informed as appropriate.
About idelalisib
In the EU, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients unsuitable for chemo-immunotherapy.
Idelalisib is also approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
About the trials
The trials in which patients have experienced serious AEs involve patients with CLL and indolent non-Hodgkin lymphoma (NHL).
In one trial (NCT01732926), researchers are evaluating idelalisib in combination with bendamustine and rituximab for previously treated indolent NHL.
In another (NCT01732913), researchers are testing idelalisib in combination with rituximab for previously treated indolent NHL.
And in the third (NCT01980888), researchers are evaluating idelalisib in combination with bendamustine and rituximab in patients with previously untreated CLL.
The EMA noted that these studies are investigating combinations of drugs that are currently not approved in the EU and include patients with disease characteristics different from those covered by the approved indications for idelalisib.
About the review
The EMA has begun the review of idelalisib at the request of the EC, under Article 20 of Directive 2001/83/EC.
The review is being carried out by the EMA’s Pharmacovigilance Risk Assessment Committee, the committee responsible for the evaluation of safety issues for human medicines, which will make a set of recommendations.
Those recommendations will then be forwarded to the Committee for Medicinal Products for Human Use, which is responsible for questions concerning medicines for human use and will adopt a final opinion on the safety of idelalisib.
The final stage of the review procedure is the EC’s adoption of a legally binding decision that is applicable in all EU member states. ![]()

Photo courtesy of
Gilead Sciences, Inc.
The European Medicines Agency (EMA) is reviewing the safety of idelalisib (Zydelig), a drug approved to treat chronic lymphocytic leukemia (CLL) and follicular lymphoma in the European Union (EU).
The European Commission (EC) requested the review because of serious adverse events (AEs), including deaths, reported in 3 clinical trials investigating idelalisib in combination with other drugs.
The AEs were mostly infection-related.
The EMA is reviewing data from these studies to assess whether the findings have any consequences for the authorized uses of idelalisib.
In the meantime, the EMA advises that patients starting or already on treatment with idelalisib be carefully monitored for signs of infections. If the drug is well tolerated, treatment should not be stopped.
The EMA is considering whether any other immediate measures are necessary during the review period. The agency said it will communicate further and keep doctors and patients informed as appropriate.
About idelalisib
In the EU, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients unsuitable for chemo-immunotherapy.
Idelalisib is also approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
About the trials
The trials in which patients have experienced serious AEs involve patients with CLL and indolent non-Hodgkin lymphoma (NHL).
In one trial (NCT01732926), researchers are evaluating idelalisib in combination with bendamustine and rituximab for previously treated indolent NHL.
In another (NCT01732913), researchers are testing idelalisib in combination with rituximab for previously treated indolent NHL.
And in the third (NCT01980888), researchers are evaluating idelalisib in combination with bendamustine and rituximab in patients with previously untreated CLL.
The EMA noted that these studies are investigating combinations of drugs that are currently not approved in the EU and include patients with disease characteristics different from those covered by the approved indications for idelalisib.
About the review
The EMA has begun the review of idelalisib at the request of the EC, under Article 20 of Directive 2001/83/EC.
The review is being carried out by the EMA’s Pharmacovigilance Risk Assessment Committee, the committee responsible for the evaluation of safety issues for human medicines, which will make a set of recommendations.
Those recommendations will then be forwarded to the Committee for Medicinal Products for Human Use, which is responsible for questions concerning medicines for human use and will adopt a final opinion on the safety of idelalisib.
The final stage of the review procedure is the EC’s adoption of a legally binding decision that is applicable in all EU member states. ![]()
Class of drugs could treat B-cell malignancies
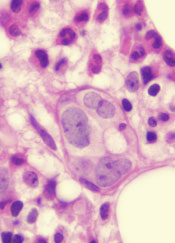
A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()

A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()

A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()
Allele associated with poor outcome in CLL

A form of the CYP3A7 gene is associated with poor outcomes in chronic lymphocytic leukemia (CLL) and other cancers, according to a study published in Cancer Research.
Among patients with CLL, breast cancer, or lung cancer, those with the CYP3A7*1C allele were more likely than those without it to experience disease progression or death.
Researchers believe this may be related to how patients metabolize treatment.
“The CYP3A7 gene encodes an enzyme that breaks down all sorts of naturally occurring substances—such as sex steroids like estrogen and testosterone—as well as a wide range of drugs that are used in the treatment of cancer,” said Olivia Fletcher, PhD, of The Institute of Cancer Research in London, UK.
“The CYP3A7 gene is normally turned on in an embryo and then turned off shortly after a baby is born, but individuals who have 1 or more copies of the CYP3A7*1C form of the gene turn on their CYP3A7 gene in adult life.”
“We found that individuals with breast cancer, lung cancer, or CLL who carry 1 or more copies of the CYP3A7*1C allele tend to have worse outcomes. One possibility is that these patients break down the drugs that they are given to treat their cancer too fast. However, further independent studies that replicate our findings in larger numbers of patients and rule out biases are needed before we could recommend any changes to the treatment that cancer patients with the CYP3A7*1C allele receive.”
To assess the impact of the CYP3A7*1C allele on patient outcomes, Dr Fletcher and her colleagues analyzed DNA samples from 1008 breast cancer patients, 1142 patients with lung cancer, and 356 patients with CLL.
The team looked for the presence of the single nucleotide polymorphism (SNP) rs45446698. Dr Fletcher explained that rs45446698 is 1 of 7 SNPs that cluster together to form the CYP3A7*1C allele.
The researchers found that, among CLL patients, rs45446698 (and, therefore, the CYP3A7*1C allele) was associated with a 62% increased risk of disease progression (P=0.03).
Among breast cancer patients, rs45446698 was associated with a 74% increased risk of breast cancer mortality (P=0.03). And among the lung cancer patients, the SNP was associated with a 43% increased risk of death from any cause (P=0.009).
The researchers also found borderline evidence of a statistical interaction between the CYP3A7*1C allele, treatment of patients with a cytotoxic agent that is a CYP3A substrate, and clinical outcome (P=0.06).
“Even though we did not see a statistically significant difference when stratifying patients by treatment with a CYP3A7 substrate, the fact that we see the same effect in 3 very different cancer types suggests to me that it is more likely to be something to do with treatment than the disease itself,” Dr Fletcher said.
“However, we are looking at ways of replicating these results in additional cohorts of patients and types of cancer, as well as overcoming the limitations of this study.”
Dr Fletcher explained that the main limitation of this study is that the researchers used samples and clinical information collected for other studies. So they did not have the same clinical information for each patient, and the samples were collected at different time points and for patients treated with various drugs.
She also noted that the researchers were not able to determine how quickly the patients broke down their treatments.
This study was supported by Sanofi-Aventis, Breast Cancer Now, Bloodwise, Cancer Research UK, the Medical Research Council, the Cridlan Trust, and the Helen Rollason Cancer Charity. The authors’ institutions received funding from the National Health Service of the United Kingdom. ![]()

A form of the CYP3A7 gene is associated with poor outcomes in chronic lymphocytic leukemia (CLL) and other cancers, according to a study published in Cancer Research.
Among patients with CLL, breast cancer, or lung cancer, those with the CYP3A7*1C allele were more likely than those without it to experience disease progression or death.
Researchers believe this may be related to how patients metabolize treatment.
“The CYP3A7 gene encodes an enzyme that breaks down all sorts of naturally occurring substances—such as sex steroids like estrogen and testosterone—as well as a wide range of drugs that are used in the treatment of cancer,” said Olivia Fletcher, PhD, of The Institute of Cancer Research in London, UK.
“The CYP3A7 gene is normally turned on in an embryo and then turned off shortly after a baby is born, but individuals who have 1 or more copies of the CYP3A7*1C form of the gene turn on their CYP3A7 gene in adult life.”
“We found that individuals with breast cancer, lung cancer, or CLL who carry 1 or more copies of the CYP3A7*1C allele tend to have worse outcomes. One possibility is that these patients break down the drugs that they are given to treat their cancer too fast. However, further independent studies that replicate our findings in larger numbers of patients and rule out biases are needed before we could recommend any changes to the treatment that cancer patients with the CYP3A7*1C allele receive.”
To assess the impact of the CYP3A7*1C allele on patient outcomes, Dr Fletcher and her colleagues analyzed DNA samples from 1008 breast cancer patients, 1142 patients with lung cancer, and 356 patients with CLL.
The team looked for the presence of the single nucleotide polymorphism (SNP) rs45446698. Dr Fletcher explained that rs45446698 is 1 of 7 SNPs that cluster together to form the CYP3A7*1C allele.
The researchers found that, among CLL patients, rs45446698 (and, therefore, the CYP3A7*1C allele) was associated with a 62% increased risk of disease progression (P=0.03).
Among breast cancer patients, rs45446698 was associated with a 74% increased risk of breast cancer mortality (P=0.03). And among the lung cancer patients, the SNP was associated with a 43% increased risk of death from any cause (P=0.009).
The researchers also found borderline evidence of a statistical interaction between the CYP3A7*1C allele, treatment of patients with a cytotoxic agent that is a CYP3A substrate, and clinical outcome (P=0.06).
“Even though we did not see a statistically significant difference when stratifying patients by treatment with a CYP3A7 substrate, the fact that we see the same effect in 3 very different cancer types suggests to me that it is more likely to be something to do with treatment than the disease itself,” Dr Fletcher said.
“However, we are looking at ways of replicating these results in additional cohorts of patients and types of cancer, as well as overcoming the limitations of this study.”
Dr Fletcher explained that the main limitation of this study is that the researchers used samples and clinical information collected for other studies. So they did not have the same clinical information for each patient, and the samples were collected at different time points and for patients treated with various drugs.
She also noted that the researchers were not able to determine how quickly the patients broke down their treatments.
This study was supported by Sanofi-Aventis, Breast Cancer Now, Bloodwise, Cancer Research UK, the Medical Research Council, the Cridlan Trust, and the Helen Rollason Cancer Charity. The authors’ institutions received funding from the National Health Service of the United Kingdom. ![]()

A form of the CYP3A7 gene is associated with poor outcomes in chronic lymphocytic leukemia (CLL) and other cancers, according to a study published in Cancer Research.
Among patients with CLL, breast cancer, or lung cancer, those with the CYP3A7*1C allele were more likely than those without it to experience disease progression or death.
Researchers believe this may be related to how patients metabolize treatment.
“The CYP3A7 gene encodes an enzyme that breaks down all sorts of naturally occurring substances—such as sex steroids like estrogen and testosterone—as well as a wide range of drugs that are used in the treatment of cancer,” said Olivia Fletcher, PhD, of The Institute of Cancer Research in London, UK.
“The CYP3A7 gene is normally turned on in an embryo and then turned off shortly after a baby is born, but individuals who have 1 or more copies of the CYP3A7*1C form of the gene turn on their CYP3A7 gene in adult life.”
“We found that individuals with breast cancer, lung cancer, or CLL who carry 1 or more copies of the CYP3A7*1C allele tend to have worse outcomes. One possibility is that these patients break down the drugs that they are given to treat their cancer too fast. However, further independent studies that replicate our findings in larger numbers of patients and rule out biases are needed before we could recommend any changes to the treatment that cancer patients with the CYP3A7*1C allele receive.”
To assess the impact of the CYP3A7*1C allele on patient outcomes, Dr Fletcher and her colleagues analyzed DNA samples from 1008 breast cancer patients, 1142 patients with lung cancer, and 356 patients with CLL.
The team looked for the presence of the single nucleotide polymorphism (SNP) rs45446698. Dr Fletcher explained that rs45446698 is 1 of 7 SNPs that cluster together to form the CYP3A7*1C allele.
The researchers found that, among CLL patients, rs45446698 (and, therefore, the CYP3A7*1C allele) was associated with a 62% increased risk of disease progression (P=0.03).
Among breast cancer patients, rs45446698 was associated with a 74% increased risk of breast cancer mortality (P=0.03). And among the lung cancer patients, the SNP was associated with a 43% increased risk of death from any cause (P=0.009).
The researchers also found borderline evidence of a statistical interaction between the CYP3A7*1C allele, treatment of patients with a cytotoxic agent that is a CYP3A substrate, and clinical outcome (P=0.06).
“Even though we did not see a statistically significant difference when stratifying patients by treatment with a CYP3A7 substrate, the fact that we see the same effect in 3 very different cancer types suggests to me that it is more likely to be something to do with treatment than the disease itself,” Dr Fletcher said.
“However, we are looking at ways of replicating these results in additional cohorts of patients and types of cancer, as well as overcoming the limitations of this study.”
Dr Fletcher explained that the main limitation of this study is that the researchers used samples and clinical information collected for other studies. So they did not have the same clinical information for each patient, and the samples were collected at different time points and for patients treated with various drugs.
She also noted that the researchers were not able to determine how quickly the patients broke down their treatments.
This study was supported by Sanofi-Aventis, Breast Cancer Now, Bloodwise, Cancer Research UK, the Medical Research Council, the Cridlan Trust, and the Helen Rollason Cancer Charity. The authors’ institutions received funding from the National Health Service of the United Kingdom. ![]()
Product can reduce bleeding in hemophilia B

In a phase 3 study, prophylaxis with the recombinant factor IX product nonacog alfa (BeneFIX) reduced bleeding in patients with hemophilia B.
Once-weekly prophylaxis with nonacog alfa significantly reduced the annualized bleeding rate (ABR) when compared to on-demand treatment with the drug.
Nonacog alfa was generally well tolerated, and none of the patients who received it developed inhibitors or experienced thrombotic events.
These results were published in Haemophilia. The study was sponsored by Pfizer, the company developing nonacog alfa.
The study was a sequential-period trial with a 6-month period of on-demand treatment followed by a 12-month period of prophylaxis, with a mean total therapy duration of 550 days.
All 25 male participants had moderately severe or severe hemophilia B (factor IX activity of 2 IU/dL or less), and their mean age was 31.3.
Participants had experienced at least 12 bleeding events, 6 of them in joints, in the previous year. All participants received nonacog alfa, and no one discontinued treatment early.
The median ABR values were 2.0 (range, 0.0-13.8) for the prophylaxis period and 33.6 (range, 6.1-69.0) for the on-demand treatment period. The mean ABR values were 3.6 ± 4.6 and 32.9 ±17.4, respectively (P<0.0001).
Bleeding occurred in 16 patients (64%) during the prophylaxis period and all 25 patients (100%) during the on-demand treatment period.
During the prophylaxis period, 13 patients (52%) experienced spontaneous bleeding events, with a mean ABR of 2.6. Thirteen patients (52%) experienced traumatic bleeding events, with a mean ABR of 1.0.
During the on-demand treatment period, 21 patients (84%) experienced spontaneous bleeding events, with a mean ABR of 23.1. Eighteen patients (72.0%) experienced traumatic bleeding events, with a mean ABR of 9.9.
Most bleeds had “excellent” (53.5%) or “good” (34.9%) responses to the first infusion of nonacog alfa, 10.8% had “moderate” responses, and 0.6% had no response. All bleeds responded to follow-up infusions.
The incidence of treatment-emergent adverse events was 96% in all patients—96% during the prophylaxis period and 64% during the on-demand treatment period.
The most common adverse events, occurring in at least 10% of patients during either regimen, were arthralgia (24%), back pain (12%), headache (36%), joint swelling (20%), local swelling (12%), nasopharyngitis (12%), pharyngitis (20%), pyrexia (20%), toothache (24%), and upper respiratory tract infection (24%). ![]()

In a phase 3 study, prophylaxis with the recombinant factor IX product nonacog alfa (BeneFIX) reduced bleeding in patients with hemophilia B.
Once-weekly prophylaxis with nonacog alfa significantly reduced the annualized bleeding rate (ABR) when compared to on-demand treatment with the drug.
Nonacog alfa was generally well tolerated, and none of the patients who received it developed inhibitors or experienced thrombotic events.
These results were published in Haemophilia. The study was sponsored by Pfizer, the company developing nonacog alfa.
The study was a sequential-period trial with a 6-month period of on-demand treatment followed by a 12-month period of prophylaxis, with a mean total therapy duration of 550 days.
All 25 male participants had moderately severe or severe hemophilia B (factor IX activity of 2 IU/dL or less), and their mean age was 31.3.
Participants had experienced at least 12 bleeding events, 6 of them in joints, in the previous year. All participants received nonacog alfa, and no one discontinued treatment early.
The median ABR values were 2.0 (range, 0.0-13.8) for the prophylaxis period and 33.6 (range, 6.1-69.0) for the on-demand treatment period. The mean ABR values were 3.6 ± 4.6 and 32.9 ±17.4, respectively (P<0.0001).
Bleeding occurred in 16 patients (64%) during the prophylaxis period and all 25 patients (100%) during the on-demand treatment period.
During the prophylaxis period, 13 patients (52%) experienced spontaneous bleeding events, with a mean ABR of 2.6. Thirteen patients (52%) experienced traumatic bleeding events, with a mean ABR of 1.0.
During the on-demand treatment period, 21 patients (84%) experienced spontaneous bleeding events, with a mean ABR of 23.1. Eighteen patients (72.0%) experienced traumatic bleeding events, with a mean ABR of 9.9.
Most bleeds had “excellent” (53.5%) or “good” (34.9%) responses to the first infusion of nonacog alfa, 10.8% had “moderate” responses, and 0.6% had no response. All bleeds responded to follow-up infusions.
The incidence of treatment-emergent adverse events was 96% in all patients—96% during the prophylaxis period and 64% during the on-demand treatment period.
The most common adverse events, occurring in at least 10% of patients during either regimen, were arthralgia (24%), back pain (12%), headache (36%), joint swelling (20%), local swelling (12%), nasopharyngitis (12%), pharyngitis (20%), pyrexia (20%), toothache (24%), and upper respiratory tract infection (24%). ![]()

In a phase 3 study, prophylaxis with the recombinant factor IX product nonacog alfa (BeneFIX) reduced bleeding in patients with hemophilia B.
Once-weekly prophylaxis with nonacog alfa significantly reduced the annualized bleeding rate (ABR) when compared to on-demand treatment with the drug.
Nonacog alfa was generally well tolerated, and none of the patients who received it developed inhibitors or experienced thrombotic events.
These results were published in Haemophilia. The study was sponsored by Pfizer, the company developing nonacog alfa.
The study was a sequential-period trial with a 6-month period of on-demand treatment followed by a 12-month period of prophylaxis, with a mean total therapy duration of 550 days.
All 25 male participants had moderately severe or severe hemophilia B (factor IX activity of 2 IU/dL or less), and their mean age was 31.3.
Participants had experienced at least 12 bleeding events, 6 of them in joints, in the previous year. All participants received nonacog alfa, and no one discontinued treatment early.
The median ABR values were 2.0 (range, 0.0-13.8) for the prophylaxis period and 33.6 (range, 6.1-69.0) for the on-demand treatment period. The mean ABR values were 3.6 ± 4.6 and 32.9 ±17.4, respectively (P<0.0001).
Bleeding occurred in 16 patients (64%) during the prophylaxis period and all 25 patients (100%) during the on-demand treatment period.
During the prophylaxis period, 13 patients (52%) experienced spontaneous bleeding events, with a mean ABR of 2.6. Thirteen patients (52%) experienced traumatic bleeding events, with a mean ABR of 1.0.
During the on-demand treatment period, 21 patients (84%) experienced spontaneous bleeding events, with a mean ABR of 23.1. Eighteen patients (72.0%) experienced traumatic bleeding events, with a mean ABR of 9.9.
Most bleeds had “excellent” (53.5%) or “good” (34.9%) responses to the first infusion of nonacog alfa, 10.8% had “moderate” responses, and 0.6% had no response. All bleeds responded to follow-up infusions.
The incidence of treatment-emergent adverse events was 96% in all patients—96% during the prophylaxis period and 64% during the on-demand treatment period.
The most common adverse events, occurring in at least 10% of patients during either regimen, were arthralgia (24%), back pain (12%), headache (36%), joint swelling (20%), local swelling (12%), nasopharyngitis (12%), pharyngitis (20%), pyrexia (20%), toothache (24%), and upper respiratory tract infection (24%). ![]()
Study elucidates MYC’s role in T-ALL

Image by Juha Klefstrom
Research has revealed a relationship between the oncogene MYC and 2 cell-surface proteins that protect cancer cells from the immune system—CD47 and PD-L1.
Researchers discovered that MYC regulates the expression of CD47 and PD-L1 in T-cell acute lymphoblastic leukemia (T-ALL) and several solid tumor malignancies.
The team said this study is the first to link 2 critical steps in cancer development—uncontrolled cell growth (courtesy of mutated or misregulated MYC) and an ability to “outsmart” the immune molecules meant to stop it (via CD47 and PD-L1).
The study was published in Science.
“Our findings describe an intimate, causal connection between how oncogenes like MYC cause cancer and how those cancer cells manage to evade the immune system,” said study author Dean Felsher, MD, PhD, of the Stanford University School of Medicine in California.
Researchers in Dr Felsher’s lab have been studying MYC for more than a decade, focusing on oncogene addiction, in which tumor cells are completely dependent on the expression of the oncogene. Blocking the expression of MYC in these cases causes the complete regression of tumors in animals.
In 2010, Dr Felsher and his colleagues showed this regression could only occur in animals with an intact immune system, but it wasn’t clear why.
“Since then, I’ve had it in the back of my mind that there must be a relationship between MYC and the immune system,” Dr Felsher said.
So he and his colleagues decided to see if there was a link between MYC expression and the levels of CD47 and PD-L1 proteins on the surface of cancer cells. They investigated what would happen if they actively turned off MYC expression in tumor cells from mice or humans.
The researchers found that a reduction in MYC caused a similar reduction in the levels of CD47 and PD-L1 proteins on the surface of mouse and human T-ALL cells, mouse and human liver cancer cells, human skin cancer cells, and human non-small-cell lung cancer cells.
In contrast, levels of other immune regulatory molecules found on the surface of the cells were unaffected.
In gene expression data on tumor samples from hundreds of patients, the researchers found that levels of MYC expression correlated strongly with expression levels of CD47 and PD-L1 genes in liver, kidney, and colorectal tumors.
The team then looked directly at the regulatory regions in the CD47 and PD-L1 genes. They found high levels of the MYC protein bound directly to the promoter regions of CD47 and PD-L1 in mouse T-ALL cells and in a human osteosarcoma cell line.
The researchers were also able to verify that this binding increased the expression of CD47 in a human B cell line.
Finally, the team engineered mouse T-ALL cells to constantly express CD47 or PD-L1 regardless of MYC expression status.
These cells were better able than control cells to evade the detection of immune cells like macrophages and T cells. And, unlike in previous experiments, tumors arising from these cells did not regress when MYC expression was deactivated.
“What we’re learning is that if CD47 and PD-L1 are present on the surfaces of cancer cells, even if you shut down a cancer gene, the animal doesn’t mount an adequate immune response, and the tumors don’t regress,” Dr Felsher said.
Therefore, this work suggests a combination of therapies targeting the expression of both MYC and CD47 or PD-L1 could possibly have a synergistic effect by slowing or stopping tumor growth and waving a red flag at the immune system.
“There is a growing sense of tremendous excitement in the field of cancer immunotherapy,” Dr Felsher said. “In many cases, it’s working, but it’s not been clear why some cancers are more sensitive than others. Our work highlights a direct link between oncogene expression and immune regulation that could be exploited to help patients.” ![]()

Image by Juha Klefstrom
Research has revealed a relationship between the oncogene MYC and 2 cell-surface proteins that protect cancer cells from the immune system—CD47 and PD-L1.
Researchers discovered that MYC regulates the expression of CD47 and PD-L1 in T-cell acute lymphoblastic leukemia (T-ALL) and several solid tumor malignancies.
The team said this study is the first to link 2 critical steps in cancer development—uncontrolled cell growth (courtesy of mutated or misregulated MYC) and an ability to “outsmart” the immune molecules meant to stop it (via CD47 and PD-L1).
The study was published in Science.
“Our findings describe an intimate, causal connection between how oncogenes like MYC cause cancer and how those cancer cells manage to evade the immune system,” said study author Dean Felsher, MD, PhD, of the Stanford University School of Medicine in California.
Researchers in Dr Felsher’s lab have been studying MYC for more than a decade, focusing on oncogene addiction, in which tumor cells are completely dependent on the expression of the oncogene. Blocking the expression of MYC in these cases causes the complete regression of tumors in animals.
In 2010, Dr Felsher and his colleagues showed this regression could only occur in animals with an intact immune system, but it wasn’t clear why.
“Since then, I’ve had it in the back of my mind that there must be a relationship between MYC and the immune system,” Dr Felsher said.
So he and his colleagues decided to see if there was a link between MYC expression and the levels of CD47 and PD-L1 proteins on the surface of cancer cells. They investigated what would happen if they actively turned off MYC expression in tumor cells from mice or humans.
The researchers found that a reduction in MYC caused a similar reduction in the levels of CD47 and PD-L1 proteins on the surface of mouse and human T-ALL cells, mouse and human liver cancer cells, human skin cancer cells, and human non-small-cell lung cancer cells.
In contrast, levels of other immune regulatory molecules found on the surface of the cells were unaffected.
In gene expression data on tumor samples from hundreds of patients, the researchers found that levels of MYC expression correlated strongly with expression levels of CD47 and PD-L1 genes in liver, kidney, and colorectal tumors.
The team then looked directly at the regulatory regions in the CD47 and PD-L1 genes. They found high levels of the MYC protein bound directly to the promoter regions of CD47 and PD-L1 in mouse T-ALL cells and in a human osteosarcoma cell line.
The researchers were also able to verify that this binding increased the expression of CD47 in a human B cell line.
Finally, the team engineered mouse T-ALL cells to constantly express CD47 or PD-L1 regardless of MYC expression status.
These cells were better able than control cells to evade the detection of immune cells like macrophages and T cells. And, unlike in previous experiments, tumors arising from these cells did not regress when MYC expression was deactivated.
“What we’re learning is that if CD47 and PD-L1 are present on the surfaces of cancer cells, even if you shut down a cancer gene, the animal doesn’t mount an adequate immune response, and the tumors don’t regress,” Dr Felsher said.
Therefore, this work suggests a combination of therapies targeting the expression of both MYC and CD47 or PD-L1 could possibly have a synergistic effect by slowing or stopping tumor growth and waving a red flag at the immune system.
“There is a growing sense of tremendous excitement in the field of cancer immunotherapy,” Dr Felsher said. “In many cases, it’s working, but it’s not been clear why some cancers are more sensitive than others. Our work highlights a direct link between oncogene expression and immune regulation that could be exploited to help patients.” ![]()

Image by Juha Klefstrom
Research has revealed a relationship between the oncogene MYC and 2 cell-surface proteins that protect cancer cells from the immune system—CD47 and PD-L1.
Researchers discovered that MYC regulates the expression of CD47 and PD-L1 in T-cell acute lymphoblastic leukemia (T-ALL) and several solid tumor malignancies.
The team said this study is the first to link 2 critical steps in cancer development—uncontrolled cell growth (courtesy of mutated or misregulated MYC) and an ability to “outsmart” the immune molecules meant to stop it (via CD47 and PD-L1).
The study was published in Science.
“Our findings describe an intimate, causal connection between how oncogenes like MYC cause cancer and how those cancer cells manage to evade the immune system,” said study author Dean Felsher, MD, PhD, of the Stanford University School of Medicine in California.
Researchers in Dr Felsher’s lab have been studying MYC for more than a decade, focusing on oncogene addiction, in which tumor cells are completely dependent on the expression of the oncogene. Blocking the expression of MYC in these cases causes the complete regression of tumors in animals.
In 2010, Dr Felsher and his colleagues showed this regression could only occur in animals with an intact immune system, but it wasn’t clear why.
“Since then, I’ve had it in the back of my mind that there must be a relationship between MYC and the immune system,” Dr Felsher said.
So he and his colleagues decided to see if there was a link between MYC expression and the levels of CD47 and PD-L1 proteins on the surface of cancer cells. They investigated what would happen if they actively turned off MYC expression in tumor cells from mice or humans.
The researchers found that a reduction in MYC caused a similar reduction in the levels of CD47 and PD-L1 proteins on the surface of mouse and human T-ALL cells, mouse and human liver cancer cells, human skin cancer cells, and human non-small-cell lung cancer cells.
In contrast, levels of other immune regulatory molecules found on the surface of the cells were unaffected.
In gene expression data on tumor samples from hundreds of patients, the researchers found that levels of MYC expression correlated strongly with expression levels of CD47 and PD-L1 genes in liver, kidney, and colorectal tumors.
The team then looked directly at the regulatory regions in the CD47 and PD-L1 genes. They found high levels of the MYC protein bound directly to the promoter regions of CD47 and PD-L1 in mouse T-ALL cells and in a human osteosarcoma cell line.
The researchers were also able to verify that this binding increased the expression of CD47 in a human B cell line.
Finally, the team engineered mouse T-ALL cells to constantly express CD47 or PD-L1 regardless of MYC expression status.
These cells were better able than control cells to evade the detection of immune cells like macrophages and T cells. And, unlike in previous experiments, tumors arising from these cells did not regress when MYC expression was deactivated.
“What we’re learning is that if CD47 and PD-L1 are present on the surfaces of cancer cells, even if you shut down a cancer gene, the animal doesn’t mount an adequate immune response, and the tumors don’t regress,” Dr Felsher said.
Therefore, this work suggests a combination of therapies targeting the expression of both MYC and CD47 or PD-L1 could possibly have a synergistic effect by slowing or stopping tumor growth and waving a red flag at the immune system.
“There is a growing sense of tremendous excitement in the field of cancer immunotherapy,” Dr Felsher said. “In many cases, it’s working, but it’s not been clear why some cancers are more sensitive than others. Our work highlights a direct link between oncogene expression and immune regulation that could be exploited to help patients.”
Program can predict drug side effects

Photo by Darren Baker
Scientists say they have developed a computer program that can predict whether or not a given pharmaceutical agent will produce certain side effects.
The software takes an “ensemble approach” to assessing the chemical structure of a drug molecule and can determine whether key substructures are present in the molecule that are known to give rise to side effects in other drugs.
Md Jamiul Jahid and Jianhua Ruan, PhD, both of the University of Texas at San Antonio, developed the computer program and described it in the International Journal of Computational Biology and Drug Design.
The pair tested the software’s ability to predict 1385 side effects associated with 888 marketed drugs and found that the program outperformed earlier software.
The team also used their new software to test 2883 uncharacterized compounds in the DrugBank database. The program proved capable of predicting a wide variety of side effects, including some effects that were missed by other screening methods.
The scientists believe their software could be used to alert regulatory authorities and healthcare workers as to what side effects might occur when a new drug enters late-stage clinical trials and is ultimately brought to market.
But the program may have an additional benefit as well. By identifying substructures that are associated with particular side effects, the software could be used to help medicinal chemists understand the underlying mechanism by which a side effect arises.
The chemists could then eliminate the offending substructures from drug molecules in the future, thereby reducing the number of drugs that go through the research and development pipeline and then fail in clinical trials due to severe side effects.

Photo by Darren Baker
Scientists say they have developed a computer program that can predict whether or not a given pharmaceutical agent will produce certain side effects.
The software takes an “ensemble approach” to assessing the chemical structure of a drug molecule and can determine whether key substructures are present in the molecule that are known to give rise to side effects in other drugs.
Md Jamiul Jahid and Jianhua Ruan, PhD, both of the University of Texas at San Antonio, developed the computer program and described it in the International Journal of Computational Biology and Drug Design.
The pair tested the software’s ability to predict 1385 side effects associated with 888 marketed drugs and found that the program outperformed earlier software.
The team also used their new software to test 2883 uncharacterized compounds in the DrugBank database. The program proved capable of predicting a wide variety of side effects, including some effects that were missed by other screening methods.
The scientists believe their software could be used to alert regulatory authorities and healthcare workers as to what side effects might occur when a new drug enters late-stage clinical trials and is ultimately brought to market.
But the program may have an additional benefit as well. By identifying substructures that are associated with particular side effects, the software could be used to help medicinal chemists understand the underlying mechanism by which a side effect arises.
The chemists could then eliminate the offending substructures from drug molecules in the future, thereby reducing the number of drugs that go through the research and development pipeline and then fail in clinical trials due to severe side effects.

Photo by Darren Baker
Scientists say they have developed a computer program that can predict whether or not a given pharmaceutical agent will produce certain side effects.
The software takes an “ensemble approach” to assessing the chemical structure of a drug molecule and can determine whether key substructures are present in the molecule that are known to give rise to side effects in other drugs.
Md Jamiul Jahid and Jianhua Ruan, PhD, both of the University of Texas at San Antonio, developed the computer program and described it in the International Journal of Computational Biology and Drug Design.
The pair tested the software’s ability to predict 1385 side effects associated with 888 marketed drugs and found that the program outperformed earlier software.
The team also used their new software to test 2883 uncharacterized compounds in the DrugBank database. The program proved capable of predicting a wide variety of side effects, including some effects that were missed by other screening methods.
The scientists believe their software could be used to alert regulatory authorities and healthcare workers as to what side effects might occur when a new drug enters late-stage clinical trials and is ultimately brought to market.
But the program may have an additional benefit as well. By identifying substructures that are associated with particular side effects, the software could be used to help medicinal chemists understand the underlying mechanism by which a side effect arises.
The chemists could then eliminate the offending substructures from drug molecules in the future, thereby reducing the number of drugs that go through the research and development pipeline and then fail in clinical trials due to severe side effects.
Regimen can reduce risk of malaria during pregnancy

Photo by Nina Matthews
A 2-drug prophylactic regimen can reduce the risk of Plasmodium falciparum malaria during pregnancy, according to a study published in NEJM.
Monthly treatment with the regimen, dihydroartemisinin-piperaquine (DP), reduced the rate of symptomatic malaria, placental malaria, and parasitemia, when compared to less frequent dosing with DP or treatment with sulfadoxine-pyrimethamine (SP), the current standard prophylactic regimen.
Researchers therefore believe DP may be a feasible alternative to SP, which has become less effective over time.
“The malaria parasite’s resistance to SP is widespread, especially in sub-Saharan Africa,” said study author Abel Kakuru, MD, of the Infectious Diseases Research Collaboration in Kampala, Uganda.
“But we are still using the same drugs because we have no better alternatives.”
In an attempt to identify a better alternative, Dr Kakuru and his colleagues studied 300 pregnant women from Tororo, Uganda, from June 2014 through October 2014. All of the subjects were age 16 or older and anywhere from 12 to 20 weeks pregnant.
The women were randomized to receive 1 of 3 regimens for malaria prophylaxis:
- DP once a month
- DP at 20, 28, and 30 weeks of pregnancy
- SP at 20, 28, and 30 weeks of pregnancy.
Participants had monthly checkups at the study clinic, where they received regular blood tests for malaria. The researchers also assessed malaria infection in the placenta.
Placental malaria was confirmed in 50% of women in the SP group, 34% in the 3-dose-DP group (P=0.03), and 27% in the monthly DP group (P=0.001).
Forty-one percent of the women on SP had malaria parasites in their blood, compared to 17% in the 3-dose DP group (P<0.001), and 5% in the monthly DP group (P<0.001).
None of the women on monthly DP had symptomatic malaria during pregnancy. But there were 41 episodes of malaria during pregnancy in the SP group and 12 episodes in the 3-dose DP group.
The researchers also evaluated the women and infants in the study for a composite adverse birth outcome of spontaneous abortion, stillbirth, low birth weight, preterm delivery, or birth defects.
The risk of any adverse birth outcome was 9% in the monthly DP group, 21% in the 3-dose DP group (P=0.02), and 19% in the SP group (P=0.05).
The researchers concluded that monthly dosing of DP provided the best protection against malaria and called for additional studies to determine if the regimen would provide an effective alternative treatment in other parts of Uganda and elsewhere in Africa.

Photo by Nina Matthews
A 2-drug prophylactic regimen can reduce the risk of Plasmodium falciparum malaria during pregnancy, according to a study published in NEJM.
Monthly treatment with the regimen, dihydroartemisinin-piperaquine (DP), reduced the rate of symptomatic malaria, placental malaria, and parasitemia, when compared to less frequent dosing with DP or treatment with sulfadoxine-pyrimethamine (SP), the current standard prophylactic regimen.
Researchers therefore believe DP may be a feasible alternative to SP, which has become less effective over time.
“The malaria parasite’s resistance to SP is widespread, especially in sub-Saharan Africa,” said study author Abel Kakuru, MD, of the Infectious Diseases Research Collaboration in Kampala, Uganda.
“But we are still using the same drugs because we have no better alternatives.”
In an attempt to identify a better alternative, Dr Kakuru and his colleagues studied 300 pregnant women from Tororo, Uganda, from June 2014 through October 2014. All of the subjects were age 16 or older and anywhere from 12 to 20 weeks pregnant.
The women were randomized to receive 1 of 3 regimens for malaria prophylaxis:
- DP once a month
- DP at 20, 28, and 30 weeks of pregnancy
- SP at 20, 28, and 30 weeks of pregnancy.
Participants had monthly checkups at the study clinic, where they received regular blood tests for malaria. The researchers also assessed malaria infection in the placenta.
Placental malaria was confirmed in 50% of women in the SP group, 34% in the 3-dose-DP group (P=0.03), and 27% in the monthly DP group (P=0.001).
Forty-one percent of the women on SP had malaria parasites in their blood, compared to 17% in the 3-dose DP group (P<0.001), and 5% in the monthly DP group (P<0.001).
None of the women on monthly DP had symptomatic malaria during pregnancy. But there were 41 episodes of malaria during pregnancy in the SP group and 12 episodes in the 3-dose DP group.
The researchers also evaluated the women and infants in the study for a composite adverse birth outcome of spontaneous abortion, stillbirth, low birth weight, preterm delivery, or birth defects.
The risk of any adverse birth outcome was 9% in the monthly DP group, 21% in the 3-dose DP group (P=0.02), and 19% in the SP group (P=0.05).
The researchers concluded that monthly dosing of DP provided the best protection against malaria and called for additional studies to determine if the regimen would provide an effective alternative treatment in other parts of Uganda and elsewhere in Africa.

Photo by Nina Matthews
A 2-drug prophylactic regimen can reduce the risk of Plasmodium falciparum malaria during pregnancy, according to a study published in NEJM.
Monthly treatment with the regimen, dihydroartemisinin-piperaquine (DP), reduced the rate of symptomatic malaria, placental malaria, and parasitemia, when compared to less frequent dosing with DP or treatment with sulfadoxine-pyrimethamine (SP), the current standard prophylactic regimen.
Researchers therefore believe DP may be a feasible alternative to SP, which has become less effective over time.
“The malaria parasite’s resistance to SP is widespread, especially in sub-Saharan Africa,” said study author Abel Kakuru, MD, of the Infectious Diseases Research Collaboration in Kampala, Uganda.
“But we are still using the same drugs because we have no better alternatives.”
In an attempt to identify a better alternative, Dr Kakuru and his colleagues studied 300 pregnant women from Tororo, Uganda, from June 2014 through October 2014. All of the subjects were age 16 or older and anywhere from 12 to 20 weeks pregnant.
The women were randomized to receive 1 of 3 regimens for malaria prophylaxis:
- DP once a month
- DP at 20, 28, and 30 weeks of pregnancy
- SP at 20, 28, and 30 weeks of pregnancy.
Participants had monthly checkups at the study clinic, where they received regular blood tests for malaria. The researchers also assessed malaria infection in the placenta.
Placental malaria was confirmed in 50% of women in the SP group, 34% in the 3-dose-DP group (P=0.03), and 27% in the monthly DP group (P=0.001).
Forty-one percent of the women on SP had malaria parasites in their blood, compared to 17% in the 3-dose DP group (P<0.001), and 5% in the monthly DP group (P<0.001).
None of the women on monthly DP had symptomatic malaria during pregnancy. But there were 41 episodes of malaria during pregnancy in the SP group and 12 episodes in the 3-dose DP group.
The researchers also evaluated the women and infants in the study for a composite adverse birth outcome of spontaneous abortion, stillbirth, low birth weight, preterm delivery, or birth defects.
The risk of any adverse birth outcome was 9% in the monthly DP group, 21% in the 3-dose DP group (P=0.02), and 19% in the SP group (P=0.05).
The researchers concluded that monthly dosing of DP provided the best protection against malaria and called for additional studies to determine if the regimen would provide an effective alternative treatment in other parts of Uganda and elsewhere in Africa.
Germline mutations linked to hematologic malignancies

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.”

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.”

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.”