User login
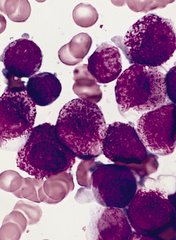
Factors appear to confer poor survival in AML

receiving chemotherapy
Photo by Rhoda Baer
A study published in the British Journal of Hematology has revealed factors that appear to affect survival in patients with acute myeloid leukemia (AML).
The research showed that death was more likely among AML patients treated at centers not affiliated with the National Cancer Institute (NCI).
Death was also more likely for black patients, older patients, those without health insurance, and those who lived in poorer neighborhoods.
“Our study reveals that survival inequalities persist among vulnerable patients with acute myeloid leukemia, such as the uninsured, those of black race/ethnicity, and adolescents and young adults,” said study author Renata Abrahão, MD, of Cancer Prevention Institute of California.
“This study can serve as a baseline to compare changes in survival that may result from potential improvements in health insurance coverage following the implementation of the Affordable Care Act.”
Dr Abrahão and her colleagues analyzed 3935 AML patients who were 39 or younger between 1988 and 2011. The team used data from the California Cancer Registry, which participates in the Surveillance, Epidemiology and End Results program of the NCI.
The data revealed an increase over time in the 5-year survival rate, from 32.9% in 1988–1995 to 50% in 2004–2011. However, 58% of the patients (n=2272) died during follow-up. The overall median follow-up was 10 years, and the median time to death was 0.9 years.
A multivariate analysis revealed several subgroups of patients with worse survival.
Older patients had a greater risk of death when compared to patients ages 0 to 9. The hazard ratio (HR) was 1.23 for patients ages 10 to 19, 1.34 for patients ages 20 to 29, and 1.55 for patients ages 30 to 39.
Black patients had an increased risk of death as well. When compared with white patients, the HR was 1.27 for black patients, 1.05 for Hispanic patients, and 0.98 for Asian/Pacific Islanders.
Patients living in the neighborhoods with the lowest socioeconomic status had an HR of 1.14. And patients who received their initial care at a hospital not affiliated with the NCI had an HR of 1.18.
Health insurance information was only available for patients diagnosed from 1996 to 2011. Among these patients, the risk of death was higher among uninsured patients (HR=1.34) than among privately insured patients, but there was no difference between privately and publicly insured patients.
Explaining the findings
The researchers said AML diagnosis in older children, adolescents, and young adults may require more intensive treatment than in young children, which may lead to a higher probability of treatment-related complications. And recent studies have shown the biology of pediatric AML differs from adult AML, which may lead to a favorable prognosis in younger patients.
In addition, older children, adolescents, and young adults are less likely to participate in clinical trials and more likely to receive treatment at hospitals not affiliated with the NCI, when compared to younger children.
The researchers said it is not clear what factors accounted for the inferior survival observed among black patients. The team speculated that genetics may contribute to the difference in chemotherapy response or that black patients had less access to chemotherapy and other treatments such as hematopoietic stem cell transplant.
The association between lower socioeconomic status and death suggests a lack of access to treatment. The same can be said for the association between death and a lack of insurance.
“[T]his study showed that survival after AML remains low among young patients and highlights the need for new therapeutic regimens to treat this disease with various subtypes,” Dr Abrahão said.
“We emphasized the importance of linking population-based data with genetic and clinical information contained in the patients’ medical records in order to better understand the causes of survival inequalities.” ![]()

receiving chemotherapy
Photo by Rhoda Baer
A study published in the British Journal of Hematology has revealed factors that appear to affect survival in patients with acute myeloid leukemia (AML).
The research showed that death was more likely among AML patients treated at centers not affiliated with the National Cancer Institute (NCI).
Death was also more likely for black patients, older patients, those without health insurance, and those who lived in poorer neighborhoods.
“Our study reveals that survival inequalities persist among vulnerable patients with acute myeloid leukemia, such as the uninsured, those of black race/ethnicity, and adolescents and young adults,” said study author Renata Abrahão, MD, of Cancer Prevention Institute of California.
“This study can serve as a baseline to compare changes in survival that may result from potential improvements in health insurance coverage following the implementation of the Affordable Care Act.”
Dr Abrahão and her colleagues analyzed 3935 AML patients who were 39 or younger between 1988 and 2011. The team used data from the California Cancer Registry, which participates in the Surveillance, Epidemiology and End Results program of the NCI.
The data revealed an increase over time in the 5-year survival rate, from 32.9% in 1988–1995 to 50% in 2004–2011. However, 58% of the patients (n=2272) died during follow-up. The overall median follow-up was 10 years, and the median time to death was 0.9 years.
A multivariate analysis revealed several subgroups of patients with worse survival.
Older patients had a greater risk of death when compared to patients ages 0 to 9. The hazard ratio (HR) was 1.23 for patients ages 10 to 19, 1.34 for patients ages 20 to 29, and 1.55 for patients ages 30 to 39.
Black patients had an increased risk of death as well. When compared with white patients, the HR was 1.27 for black patients, 1.05 for Hispanic patients, and 0.98 for Asian/Pacific Islanders.
Patients living in the neighborhoods with the lowest socioeconomic status had an HR of 1.14. And patients who received their initial care at a hospital not affiliated with the NCI had an HR of 1.18.
Health insurance information was only available for patients diagnosed from 1996 to 2011. Among these patients, the risk of death was higher among uninsured patients (HR=1.34) than among privately insured patients, but there was no difference between privately and publicly insured patients.
Explaining the findings
The researchers said AML diagnosis in older children, adolescents, and young adults may require more intensive treatment than in young children, which may lead to a higher probability of treatment-related complications. And recent studies have shown the biology of pediatric AML differs from adult AML, which may lead to a favorable prognosis in younger patients.
In addition, older children, adolescents, and young adults are less likely to participate in clinical trials and more likely to receive treatment at hospitals not affiliated with the NCI, when compared to younger children.
The researchers said it is not clear what factors accounted for the inferior survival observed among black patients. The team speculated that genetics may contribute to the difference in chemotherapy response or that black patients had less access to chemotherapy and other treatments such as hematopoietic stem cell transplant.
The association between lower socioeconomic status and death suggests a lack of access to treatment. The same can be said for the association between death and a lack of insurance.
“[T]his study showed that survival after AML remains low among young patients and highlights the need for new therapeutic regimens to treat this disease with various subtypes,” Dr Abrahão said.
“We emphasized the importance of linking population-based data with genetic and clinical information contained in the patients’ medical records in order to better understand the causes of survival inequalities.” ![]()

receiving chemotherapy
Photo by Rhoda Baer
A study published in the British Journal of Hematology has revealed factors that appear to affect survival in patients with acute myeloid leukemia (AML).
The research showed that death was more likely among AML patients treated at centers not affiliated with the National Cancer Institute (NCI).
Death was also more likely for black patients, older patients, those without health insurance, and those who lived in poorer neighborhoods.
“Our study reveals that survival inequalities persist among vulnerable patients with acute myeloid leukemia, such as the uninsured, those of black race/ethnicity, and adolescents and young adults,” said study author Renata Abrahão, MD, of Cancer Prevention Institute of California.
“This study can serve as a baseline to compare changes in survival that may result from potential improvements in health insurance coverage following the implementation of the Affordable Care Act.”
Dr Abrahão and her colleagues analyzed 3935 AML patients who were 39 or younger between 1988 and 2011. The team used data from the California Cancer Registry, which participates in the Surveillance, Epidemiology and End Results program of the NCI.
The data revealed an increase over time in the 5-year survival rate, from 32.9% in 1988–1995 to 50% in 2004–2011. However, 58% of the patients (n=2272) died during follow-up. The overall median follow-up was 10 years, and the median time to death was 0.9 years.
A multivariate analysis revealed several subgroups of patients with worse survival.
Older patients had a greater risk of death when compared to patients ages 0 to 9. The hazard ratio (HR) was 1.23 for patients ages 10 to 19, 1.34 for patients ages 20 to 29, and 1.55 for patients ages 30 to 39.
Black patients had an increased risk of death as well. When compared with white patients, the HR was 1.27 for black patients, 1.05 for Hispanic patients, and 0.98 for Asian/Pacific Islanders.
Patients living in the neighborhoods with the lowest socioeconomic status had an HR of 1.14. And patients who received their initial care at a hospital not affiliated with the NCI had an HR of 1.18.
Health insurance information was only available for patients diagnosed from 1996 to 2011. Among these patients, the risk of death was higher among uninsured patients (HR=1.34) than among privately insured patients, but there was no difference between privately and publicly insured patients.
Explaining the findings
The researchers said AML diagnosis in older children, adolescents, and young adults may require more intensive treatment than in young children, which may lead to a higher probability of treatment-related complications. And recent studies have shown the biology of pediatric AML differs from adult AML, which may lead to a favorable prognosis in younger patients.
In addition, older children, adolescents, and young adults are less likely to participate in clinical trials and more likely to receive treatment at hospitals not affiliated with the NCI, when compared to younger children.
The researchers said it is not clear what factors accounted for the inferior survival observed among black patients. The team speculated that genetics may contribute to the difference in chemotherapy response or that black patients had less access to chemotherapy and other treatments such as hematopoietic stem cell transplant.
The association between lower socioeconomic status and death suggests a lack of access to treatment. The same can be said for the association between death and a lack of insurance.
“[T]his study showed that survival after AML remains low among young patients and highlights the need for new therapeutic regimens to treat this disease with various subtypes,” Dr Abrahão said.
“We emphasized the importance of linking population-based data with genetic and clinical information contained in the patients’ medical records in order to better understand the causes of survival inequalities.” ![]()
Protein ‘map’ could aid development of malaria vaccine

Plasmodium vivax
Image by Mae Melvin
Researchers say they have determined the structure of the protein PvRBP2a, which is used by the malaria parasite Plasmodium vivax to infect human red blood cells.
This revealed that PvRBP2a is structurally similar to PfRh5, the essential erythrocyte-binding protein in the parasite P falciparum.
The researchers believe these findings could help scientists generate new tools to prevent infection with malaria parasites.
Wai-Hong Tham, PhD, of The Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and her colleagues conducted this research and reported the results in PNAS.
“We have produced the first 3-dimensional, atomic resolution structure of the protein [PvRBP2a] using the Australian Synchrotron in Melbourne,” Dr Tham said.
She and her colleagues found that PvRBP2a consists of 10 α-helices and 1 short β-hairpin. And although PvRBP2a is structurally similar to PfRh5, the 2 proteins have different surface properties.
“The 3-dimensional map showed us that the proteins are folded in the same way—like having similar origami instructions,” Dr Tham said. “The difference is actually in the electrical charge on the surface of the molecules.”
“Now that we have an atomic-resolution map, we hope to identify a common part of the protein that could be used to design a vaccine not only for Plasmodium vivax but potentially for both vivax and falciparum.”
“These two species of malaria are responsible for the majority of malaria infections worldwide, so a vaccine that targets both would be a critical addition to our arsenal.”
Dr Tham said there is growing evidence that developing better treatments or preventive strategies for P vivax malaria is imperative for malaria eradication.
“Not only is P vivax the most widespread species of malaria, it is also more difficult to treat because it can hide in the liver for long periods of time without symptoms,” she said. “In addition, studies show that effective treatment of falciparum malaria tends to be accompanied by a resurgence of P vivax, so it is critical to continue looking for better ways to manage this species.” ![]()

Plasmodium vivax
Image by Mae Melvin
Researchers say they have determined the structure of the protein PvRBP2a, which is used by the malaria parasite Plasmodium vivax to infect human red blood cells.
This revealed that PvRBP2a is structurally similar to PfRh5, the essential erythrocyte-binding protein in the parasite P falciparum.
The researchers believe these findings could help scientists generate new tools to prevent infection with malaria parasites.
Wai-Hong Tham, PhD, of The Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and her colleagues conducted this research and reported the results in PNAS.
“We have produced the first 3-dimensional, atomic resolution structure of the protein [PvRBP2a] using the Australian Synchrotron in Melbourne,” Dr Tham said.
She and her colleagues found that PvRBP2a consists of 10 α-helices and 1 short β-hairpin. And although PvRBP2a is structurally similar to PfRh5, the 2 proteins have different surface properties.
“The 3-dimensional map showed us that the proteins are folded in the same way—like having similar origami instructions,” Dr Tham said. “The difference is actually in the electrical charge on the surface of the molecules.”
“Now that we have an atomic-resolution map, we hope to identify a common part of the protein that could be used to design a vaccine not only for Plasmodium vivax but potentially for both vivax and falciparum.”
“These two species of malaria are responsible for the majority of malaria infections worldwide, so a vaccine that targets both would be a critical addition to our arsenal.”
Dr Tham said there is growing evidence that developing better treatments or preventive strategies for P vivax malaria is imperative for malaria eradication.
“Not only is P vivax the most widespread species of malaria, it is also more difficult to treat because it can hide in the liver for long periods of time without symptoms,” she said. “In addition, studies show that effective treatment of falciparum malaria tends to be accompanied by a resurgence of P vivax, so it is critical to continue looking for better ways to manage this species.” ![]()

Plasmodium vivax
Image by Mae Melvin
Researchers say they have determined the structure of the protein PvRBP2a, which is used by the malaria parasite Plasmodium vivax to infect human red blood cells.
This revealed that PvRBP2a is structurally similar to PfRh5, the essential erythrocyte-binding protein in the parasite P falciparum.
The researchers believe these findings could help scientists generate new tools to prevent infection with malaria parasites.
Wai-Hong Tham, PhD, of The Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and her colleagues conducted this research and reported the results in PNAS.
“We have produced the first 3-dimensional, atomic resolution structure of the protein [PvRBP2a] using the Australian Synchrotron in Melbourne,” Dr Tham said.
She and her colleagues found that PvRBP2a consists of 10 α-helices and 1 short β-hairpin. And although PvRBP2a is structurally similar to PfRh5, the 2 proteins have different surface properties.
“The 3-dimensional map showed us that the proteins are folded in the same way—like having similar origami instructions,” Dr Tham said. “The difference is actually in the electrical charge on the surface of the molecules.”
“Now that we have an atomic-resolution map, we hope to identify a common part of the protein that could be used to design a vaccine not only for Plasmodium vivax but potentially for both vivax and falciparum.”
“These two species of malaria are responsible for the majority of malaria infections worldwide, so a vaccine that targets both would be a critical addition to our arsenal.”
Dr Tham said there is growing evidence that developing better treatments or preventive strategies for P vivax malaria is imperative for malaria eradication.
“Not only is P vivax the most widespread species of malaria, it is also more difficult to treat because it can hide in the liver for long periods of time without symptoms,” she said. “In addition, studies show that effective treatment of falciparum malaria tends to be accompanied by a resurgence of P vivax, so it is critical to continue looking for better ways to manage this species.” ![]()
Drug granted breakthrough designation for AML
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia. ![]()

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia. ![]()

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia. ![]()
Targeting EZH2 to treat ETP-ALL

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()
Health Canada approves ruxolitinib for PV

Image courtesy of AFIP
Health Canada has approved the JAK1/2 inhibitor ruxolitinib (Jakavi) for the control of hematocrit in adult patients with polycythemia vera (PV) that is resistant to or intolerant of a cytoreductive agent.
Ruxolitinib is the first targeted treatment approved to treat PV in Canada.
The approval is based on results of the phase 3 RESPONSE trial, which showed that ruxolitinib could provide hematocrit control without phlebotomy in patients with PV.
For RESPONSE, researchers compared ruxolitinib to best available therapy (BAT) for PV. The trial was sponsored by Incyte Corporation and Novartis Pharmaceuticals, the companies developing ruxolitinib.
The study’s primary endpoint was the proportion of patients who achieved hematocrit control and were not eligible for phlebotomy from weeks 8 through 32 (with no more than 1 instance of phlebotomy eligibility between randomization and week 8) and who saw a 35% or greater reduction in spleen volume from baseline, as assessed by imaging at week 32.
The primary endpoint was met by significantly more patients in the ruxolitinib arm than the BAT arm— 20.9% and 0.9%, respectively (P<0.0001).
Sixty percent of patients in the ruxolitinib arm achieved hematocrit control, as did 19.6% of patients in the BAT arm. The percentage of patients who had at least a 35% reduction in spleen volume was 38.2% in the ruxolitinib arm and 0.9% in the BAT arm.
The proportion of patients achieving a complete hematologic remission at week 32 was 23.6% in the ruxolitinib arm and 8.9% in the BAT arm (P=0.0028). The proportion of patients achieving a durable primary response at week 48 was 19.1% in the ruxolitinib arm and 0.9% in the BAT arm (P<0.0001).
At 80 weeks, the most common adverse events in the ruxolitinib arm were headache (22%), diarrhea (20%), pruritus (20%), and fatigue (17%). Grade 3 or 4 anemia and thrombocytopenia occurred in 2% and 6% of patients, respectively. Five percent of patients discontinued ruxolitinib due to adverse events. ![]()

Image courtesy of AFIP
Health Canada has approved the JAK1/2 inhibitor ruxolitinib (Jakavi) for the control of hematocrit in adult patients with polycythemia vera (PV) that is resistant to or intolerant of a cytoreductive agent.
Ruxolitinib is the first targeted treatment approved to treat PV in Canada.
The approval is based on results of the phase 3 RESPONSE trial, which showed that ruxolitinib could provide hematocrit control without phlebotomy in patients with PV.
For RESPONSE, researchers compared ruxolitinib to best available therapy (BAT) for PV. The trial was sponsored by Incyte Corporation and Novartis Pharmaceuticals, the companies developing ruxolitinib.
The study’s primary endpoint was the proportion of patients who achieved hematocrit control and were not eligible for phlebotomy from weeks 8 through 32 (with no more than 1 instance of phlebotomy eligibility between randomization and week 8) and who saw a 35% or greater reduction in spleen volume from baseline, as assessed by imaging at week 32.
The primary endpoint was met by significantly more patients in the ruxolitinib arm than the BAT arm— 20.9% and 0.9%, respectively (P<0.0001).
Sixty percent of patients in the ruxolitinib arm achieved hematocrit control, as did 19.6% of patients in the BAT arm. The percentage of patients who had at least a 35% reduction in spleen volume was 38.2% in the ruxolitinib arm and 0.9% in the BAT arm.
The proportion of patients achieving a complete hematologic remission at week 32 was 23.6% in the ruxolitinib arm and 8.9% in the BAT arm (P=0.0028). The proportion of patients achieving a durable primary response at week 48 was 19.1% in the ruxolitinib arm and 0.9% in the BAT arm (P<0.0001).
At 80 weeks, the most common adverse events in the ruxolitinib arm were headache (22%), diarrhea (20%), pruritus (20%), and fatigue (17%). Grade 3 or 4 anemia and thrombocytopenia occurred in 2% and 6% of patients, respectively. Five percent of patients discontinued ruxolitinib due to adverse events. ![]()

Image courtesy of AFIP
Health Canada has approved the JAK1/2 inhibitor ruxolitinib (Jakavi) for the control of hematocrit in adult patients with polycythemia vera (PV) that is resistant to or intolerant of a cytoreductive agent.
Ruxolitinib is the first targeted treatment approved to treat PV in Canada.
The approval is based on results of the phase 3 RESPONSE trial, which showed that ruxolitinib could provide hematocrit control without phlebotomy in patients with PV.
For RESPONSE, researchers compared ruxolitinib to best available therapy (BAT) for PV. The trial was sponsored by Incyte Corporation and Novartis Pharmaceuticals, the companies developing ruxolitinib.
The study’s primary endpoint was the proportion of patients who achieved hematocrit control and were not eligible for phlebotomy from weeks 8 through 32 (with no more than 1 instance of phlebotomy eligibility between randomization and week 8) and who saw a 35% or greater reduction in spleen volume from baseline, as assessed by imaging at week 32.
The primary endpoint was met by significantly more patients in the ruxolitinib arm than the BAT arm— 20.9% and 0.9%, respectively (P<0.0001).
Sixty percent of patients in the ruxolitinib arm achieved hematocrit control, as did 19.6% of patients in the BAT arm. The percentage of patients who had at least a 35% reduction in spleen volume was 38.2% in the ruxolitinib arm and 0.9% in the BAT arm.
The proportion of patients achieving a complete hematologic remission at week 32 was 23.6% in the ruxolitinib arm and 8.9% in the BAT arm (P=0.0028). The proportion of patients achieving a durable primary response at week 48 was 19.1% in the ruxolitinib arm and 0.9% in the BAT arm (P<0.0001).
At 80 weeks, the most common adverse events in the ruxolitinib arm were headache (22%), diarrhea (20%), pruritus (20%), and fatigue (17%). Grade 3 or 4 anemia and thrombocytopenia occurred in 2% and 6% of patients, respectively. Five percent of patients discontinued ruxolitinib due to adverse events. ![]()
Blood collection set gets FDA clearance, CE mark

Photo by Graham Colm
A new blood collection set has received 510(k) clearance from the US Food and Drug Administration as well as the CE mark, which means it can be marketed within the European Economic Area.
The BD Vacutainer® UltraTouch™ Push Button Blood Collection Set is engineered to minimize patient discomfort during blood collection.
The set uses proprietary needle technology—Pentapoint™ Comfort and RightGauge™ Ultra-Thin Wall technology.
According to the manufacturer, BD, this technology can reduce penetration forces without compromising tube fill times or sample quality.
Research has shown that PentaPoint™ Comfort 5-bevel needle technology helps reduce the chance of a painful injection by creating a flatter, thinner surface to help penetrate the skin with significantly greater ease.1
When combined with RightGauge™ technology, which increases the needle’s inner diameter and enables clinicians to select a smaller gauge needle without sacrificing sample quality and blood flow, the BD Vacutainer® UltraTouch™ Push Button Blood Collection Set has been shown to reduce penetration forces by up to 32% when compared to another blood collection set.2
“The ability to use smaller gauge needles should also help clinicians access veins more successfully,” said Ana K. Stankovic, MD, PhD, worldwide vice president of Medical Affairs for BD Life Sciences – Preanalytical Systems and Global Health.
“This could prove especially valuable in patient populations—such as oncology, geriatric, and pediatric—that often have difficult or fragile veins.”
Dr Stankovic also noted that clinicians may be reluctant to use smaller gauge needles for fear of increasing hemolysis as the blood passes slowly through the narrow cannula.
“With BD Vacutainer® UltraTouch™ Push Button Blood Collection Sets, clinicians can select the gauge that is most appropriate for their patients, without compromising sample quality, testing accuracy, and their own efficiency,” she said. ![]()
1. Hirsch LJ, et al. Journal of Diabetes Science and Technology. 2012, 6(2):328-35.
2. 2015 BD bench testing versus BD Vacutainer® Push Button Blood Collection Sets.

Photo by Graham Colm
A new blood collection set has received 510(k) clearance from the US Food and Drug Administration as well as the CE mark, which means it can be marketed within the European Economic Area.
The BD Vacutainer® UltraTouch™ Push Button Blood Collection Set is engineered to minimize patient discomfort during blood collection.
The set uses proprietary needle technology—Pentapoint™ Comfort and RightGauge™ Ultra-Thin Wall technology.
According to the manufacturer, BD, this technology can reduce penetration forces without compromising tube fill times or sample quality.
Research has shown that PentaPoint™ Comfort 5-bevel needle technology helps reduce the chance of a painful injection by creating a flatter, thinner surface to help penetrate the skin with significantly greater ease.1
When combined with RightGauge™ technology, which increases the needle’s inner diameter and enables clinicians to select a smaller gauge needle without sacrificing sample quality and blood flow, the BD Vacutainer® UltraTouch™ Push Button Blood Collection Set has been shown to reduce penetration forces by up to 32% when compared to another blood collection set.2
“The ability to use smaller gauge needles should also help clinicians access veins more successfully,” said Ana K. Stankovic, MD, PhD, worldwide vice president of Medical Affairs for BD Life Sciences – Preanalytical Systems and Global Health.
“This could prove especially valuable in patient populations—such as oncology, geriatric, and pediatric—that often have difficult or fragile veins.”
Dr Stankovic also noted that clinicians may be reluctant to use smaller gauge needles for fear of increasing hemolysis as the blood passes slowly through the narrow cannula.
“With BD Vacutainer® UltraTouch™ Push Button Blood Collection Sets, clinicians can select the gauge that is most appropriate for their patients, without compromising sample quality, testing accuracy, and their own efficiency,” she said. ![]()
1. Hirsch LJ, et al. Journal of Diabetes Science and Technology. 2012, 6(2):328-35.
2. 2015 BD bench testing versus BD Vacutainer® Push Button Blood Collection Sets.

Photo by Graham Colm
A new blood collection set has received 510(k) clearance from the US Food and Drug Administration as well as the CE mark, which means it can be marketed within the European Economic Area.
The BD Vacutainer® UltraTouch™ Push Button Blood Collection Set is engineered to minimize patient discomfort during blood collection.
The set uses proprietary needle technology—Pentapoint™ Comfort and RightGauge™ Ultra-Thin Wall technology.
According to the manufacturer, BD, this technology can reduce penetration forces without compromising tube fill times or sample quality.
Research has shown that PentaPoint™ Comfort 5-bevel needle technology helps reduce the chance of a painful injection by creating a flatter, thinner surface to help penetrate the skin with significantly greater ease.1
When combined with RightGauge™ technology, which increases the needle’s inner diameter and enables clinicians to select a smaller gauge needle without sacrificing sample quality and blood flow, the BD Vacutainer® UltraTouch™ Push Button Blood Collection Set has been shown to reduce penetration forces by up to 32% when compared to another blood collection set.2
“The ability to use smaller gauge needles should also help clinicians access veins more successfully,” said Ana K. Stankovic, MD, PhD, worldwide vice president of Medical Affairs for BD Life Sciences – Preanalytical Systems and Global Health.
“This could prove especially valuable in patient populations—such as oncology, geriatric, and pediatric—that often have difficult or fragile veins.”
Dr Stankovic also noted that clinicians may be reluctant to use smaller gauge needles for fear of increasing hemolysis as the blood passes slowly through the narrow cannula.
“With BD Vacutainer® UltraTouch™ Push Button Blood Collection Sets, clinicians can select the gauge that is most appropriate for their patients, without compromising sample quality, testing accuracy, and their own efficiency,” she said. ![]()
1. Hirsch LJ, et al. Journal of Diabetes Science and Technology. 2012, 6(2):328-35.
2. 2015 BD bench testing versus BD Vacutainer® Push Button Blood Collection Sets.
Risk of reproductive problems in male cancer survivors

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said.
SNPs may double risk of VTE in African Americans

Photo courtesy of NIGMS
Researchers say they have identified 3 genetic variants that may double the risk of venous thromboembolism (VTE) in African Americans.
The variants are single-nucleotide polymorphisms (SNPs) found on chromosome 20—rs2144940, rs2567617, and rs1998081.
The study suggests more than a third of African Americans may have at least 1 of these SNPs, but they are much less common among people of Asian or European descent.
Minoli Perera, PharmD, PhD, of the University of Chicago in Illinois, and her colleagues conducted this research and reported the results in Blood.
Dr Perera’s team noted that African Americans are 30% to 60% more likely to suffer from VTE than any other US population. However, well-known genetic risk factors for VTE, such as factor V Leiden, are common in Caucasians but occur infrequently in African Americans.
This realization led the researchers to hypothesize that there might be undiscovered genetic variants more specific to African Americans.
“While African Americans have a high risk for VTE, previous studies have not specifically focused on this population,” Dr Perera said. “If we are not looking for the correct genetic mutations when we run a laboratory test, we are doing a disservice to minority populations.”
To understand the genetic risk factors for VTE specific to African Americans, Dr Perera and her colleagues conducted a genome-wide association study in which they genotyped DNA samples from 578 African Americans, 146 of whom had a history of unprovoked VTE.
The team then confirmed the variants deemed highly prevalent in the first group by genotyping the DNA of an additional group of 159 African Americans, including 94 with VTE.
These analyses suggested a link between VTE and 3 SNPs in chromosome 20, which is associated with decreased expression of thrombomodulin—rs2144940, rs2567617, and rs1998081.
The researchers said the presence of 1 of these 3 SNPs doubles the risk of VTE. In the discovery cohort, the odds ratio was 2.18 for rs2144940, 2.17 for rs2567617, and 2.28 for rs1998081.
In the replication cohort, the odds ratio was 1.89 for rs2144940 and 1.94 for rs1998081. The researchers were not able to test for rs2567617 in this cohort due to high linkage disequilibrium.
The team said their data suggest approximately 36% of African Americans have at least 1 of the 3 SNPs. But the variants were found in much lower frequencies in other ethnicities from previous studies.
“This study not only brings us closer to understanding the cause of VTE in African Americans, it demonstrates the importance of conducting population-specific research in precision medicine,” Dr Perera said.
“Our next steps will involve investigating the predictiveness of these risk factors for VTE with the goal of reducing the high prevalence and burden of VTE in this disproportionately affected population.”

Photo courtesy of NIGMS
Researchers say they have identified 3 genetic variants that may double the risk of venous thromboembolism (VTE) in African Americans.
The variants are single-nucleotide polymorphisms (SNPs) found on chromosome 20—rs2144940, rs2567617, and rs1998081.
The study suggests more than a third of African Americans may have at least 1 of these SNPs, but they are much less common among people of Asian or European descent.
Minoli Perera, PharmD, PhD, of the University of Chicago in Illinois, and her colleagues conducted this research and reported the results in Blood.
Dr Perera’s team noted that African Americans are 30% to 60% more likely to suffer from VTE than any other US population. However, well-known genetic risk factors for VTE, such as factor V Leiden, are common in Caucasians but occur infrequently in African Americans.
This realization led the researchers to hypothesize that there might be undiscovered genetic variants more specific to African Americans.
“While African Americans have a high risk for VTE, previous studies have not specifically focused on this population,” Dr Perera said. “If we are not looking for the correct genetic mutations when we run a laboratory test, we are doing a disservice to minority populations.”
To understand the genetic risk factors for VTE specific to African Americans, Dr Perera and her colleagues conducted a genome-wide association study in which they genotyped DNA samples from 578 African Americans, 146 of whom had a history of unprovoked VTE.
The team then confirmed the variants deemed highly prevalent in the first group by genotyping the DNA of an additional group of 159 African Americans, including 94 with VTE.
These analyses suggested a link between VTE and 3 SNPs in chromosome 20, which is associated with decreased expression of thrombomodulin—rs2144940, rs2567617, and rs1998081.
The researchers said the presence of 1 of these 3 SNPs doubles the risk of VTE. In the discovery cohort, the odds ratio was 2.18 for rs2144940, 2.17 for rs2567617, and 2.28 for rs1998081.
In the replication cohort, the odds ratio was 1.89 for rs2144940 and 1.94 for rs1998081. The researchers were not able to test for rs2567617 in this cohort due to high linkage disequilibrium.
The team said their data suggest approximately 36% of African Americans have at least 1 of the 3 SNPs. But the variants were found in much lower frequencies in other ethnicities from previous studies.
“This study not only brings us closer to understanding the cause of VTE in African Americans, it demonstrates the importance of conducting population-specific research in precision medicine,” Dr Perera said.
“Our next steps will involve investigating the predictiveness of these risk factors for VTE with the goal of reducing the high prevalence and burden of VTE in this disproportionately affected population.”

Photo courtesy of NIGMS
Researchers say they have identified 3 genetic variants that may double the risk of venous thromboembolism (VTE) in African Americans.
The variants are single-nucleotide polymorphisms (SNPs) found on chromosome 20—rs2144940, rs2567617, and rs1998081.
The study suggests more than a third of African Americans may have at least 1 of these SNPs, but they are much less common among people of Asian or European descent.
Minoli Perera, PharmD, PhD, of the University of Chicago in Illinois, and her colleagues conducted this research and reported the results in Blood.
Dr Perera’s team noted that African Americans are 30% to 60% more likely to suffer from VTE than any other US population. However, well-known genetic risk factors for VTE, such as factor V Leiden, are common in Caucasians but occur infrequently in African Americans.
This realization led the researchers to hypothesize that there might be undiscovered genetic variants more specific to African Americans.
“While African Americans have a high risk for VTE, previous studies have not specifically focused on this population,” Dr Perera said. “If we are not looking for the correct genetic mutations when we run a laboratory test, we are doing a disservice to minority populations.”
To understand the genetic risk factors for VTE specific to African Americans, Dr Perera and her colleagues conducted a genome-wide association study in which they genotyped DNA samples from 578 African Americans, 146 of whom had a history of unprovoked VTE.
The team then confirmed the variants deemed highly prevalent in the first group by genotyping the DNA of an additional group of 159 African Americans, including 94 with VTE.
These analyses suggested a link between VTE and 3 SNPs in chromosome 20, which is associated with decreased expression of thrombomodulin—rs2144940, rs2567617, and rs1998081.
The researchers said the presence of 1 of these 3 SNPs doubles the risk of VTE. In the discovery cohort, the odds ratio was 2.18 for rs2144940, 2.17 for rs2567617, and 2.28 for rs1998081.
In the replication cohort, the odds ratio was 1.89 for rs2144940 and 1.94 for rs1998081. The researchers were not able to test for rs2567617 in this cohort due to high linkage disequilibrium.
The team said their data suggest approximately 36% of African Americans have at least 1 of the 3 SNPs. But the variants were found in much lower frequencies in other ethnicities from previous studies.
“This study not only brings us closer to understanding the cause of VTE in African Americans, it demonstrates the importance of conducting population-specific research in precision medicine,” Dr Perera said.
“Our next steps will involve investigating the predictiveness of these risk factors for VTE with the goal of reducing the high prevalence and burden of VTE in this disproportionately affected population.”
Pinpointing the cells that cause CML relapse

Image by Difu Wu
Preclinical research suggests chronic myeloid leukemia (CML) patients have a heterogeneous population of long-term hematopoietic stem cells (LTHSCs)—some that can initiate leukemia and some that cannot.
Researchers found they could identify the leukemia-initiating cells by measuring expression of the thrombopoietin receptor MPL. Cells with high MPL expression could initiate CML in mice.
The team said these results suggest the leukemic LTHSCs are the cells responsible for relapse in CML.
“This shows that not all leukemia stem cells are equal,” said study author Ravi Bhatia, MD, of the University of Alabama Birmingham.
“Some are more prone to causing leukemia and relapses, while some others may just hang around without potential for contributing to relapse.”
Dr Bhatia and his colleagues reported these findings in The Journal of Clinical Investigation.
In their experiments, the team used an inducible transgenic mouse model of CML, where the BCR-ABL gene fusion is under the control of a tetracycline-regulated enhancer. This model creates a chronic myeloproliferative disorder that resembles chronic phase CML.
Previous work had shown that only cells with an LTHSC phenotype were capable of long-term repopulation and leukemia-initiating capacity after transplantation to another mouse.
When the researchers transplanted LTHSCs from CML-model mice to other mice, 11 of 20 recipients developed CML, and 9 of 20 showed engraftment with CML cells but did not develop the leukocytosis characteristic of leukemia.
When the LTHSCs from the primary-recipient mice were transferred to secondary recipients, 7 of 17 mice receiving cells from leukemic mice developed CML, and none of the secondary-recipient mice receiving cells from the non-leukemic mice developed CML.
The researchers tested these 2 groups of LTHSCs for differences in gene expression. They found significant differences between the leukemic and non-leukemic LTHSCs for the genes Mpl, c-Myc, CD47, Pten, Sirt1, Ptch1, and Tie2.
The team then decided to focus on Mpl. They used flow cytometry to select LTHSCs with either high or low Mpl expression from CML-model BCR-ABL mice.
Seven of 16 mice receiving Mpl-Hi LTHSCs developed leukemia after transplantation, compared with 1 of 17 receiving Mpl-Lo LTHSCs. This suggested an increased leukemogenic capacity for the Mpl-Hi LTHSCs.
The researchers also investigated the impact of cell-cycle status. They found that CML Mpl-Hi LTHSCs that were in a resting stage of the cell cycle had enhanced long-term engraftment and leukemogenic capacity compared with cycling Mpl-Hi LTHSCs.
The team used virus vectors and shRNA to create Mpl knockdown BCR-ABL LTHSCs and showed that the knockdown cells had a greatly reduced ability to produce leukemia in recipient mice.
The Mpl knockdown cells, after stimulation by the Mpl ligand thrombopoietin, also had reduced expression of the activated transcription factors p-STAT3 and p-STAT5, compared with controls.
Finally, the researchers examined human CML cells for differences between MPL-Hi LTHSCs and MPL-Lo LTHSCs. The results were similar to those observed in mice.
The human MPL-Hi LTHSCs had a higher rate of engraftment than the human MPL-Lo LTHSCs, as tested in a xenograft model using immunodeficient mice.
Additionally, the human MPL-Hi LTHSCs had reduced sensitivity to nilotinib compared with MPL-Lo LTHSCs. However, a Jak/STAT inhibitor significantly reduced cell growth and increased apoptosis in human MPL-Hi LTHSCs.
The researchers concluded that MPL expression is a marker and key regulator of leukemogenic potential and drug sensitivity of CML LTHSCs. They said their findings support further investigation of approaches to antagonize MPL signaling as a potential therapeutic strategy to eliminate leukemia-initiating LTHSCs.

Image by Difu Wu
Preclinical research suggests chronic myeloid leukemia (CML) patients have a heterogeneous population of long-term hematopoietic stem cells (LTHSCs)—some that can initiate leukemia and some that cannot.
Researchers found they could identify the leukemia-initiating cells by measuring expression of the thrombopoietin receptor MPL. Cells with high MPL expression could initiate CML in mice.
The team said these results suggest the leukemic LTHSCs are the cells responsible for relapse in CML.
“This shows that not all leukemia stem cells are equal,” said study author Ravi Bhatia, MD, of the University of Alabama Birmingham.
“Some are more prone to causing leukemia and relapses, while some others may just hang around without potential for contributing to relapse.”
Dr Bhatia and his colleagues reported these findings in The Journal of Clinical Investigation.
In their experiments, the team used an inducible transgenic mouse model of CML, where the BCR-ABL gene fusion is under the control of a tetracycline-regulated enhancer. This model creates a chronic myeloproliferative disorder that resembles chronic phase CML.
Previous work had shown that only cells with an LTHSC phenotype were capable of long-term repopulation and leukemia-initiating capacity after transplantation to another mouse.
When the researchers transplanted LTHSCs from CML-model mice to other mice, 11 of 20 recipients developed CML, and 9 of 20 showed engraftment with CML cells but did not develop the leukocytosis characteristic of leukemia.
When the LTHSCs from the primary-recipient mice were transferred to secondary recipients, 7 of 17 mice receiving cells from leukemic mice developed CML, and none of the secondary-recipient mice receiving cells from the non-leukemic mice developed CML.
The researchers tested these 2 groups of LTHSCs for differences in gene expression. They found significant differences between the leukemic and non-leukemic LTHSCs for the genes Mpl, c-Myc, CD47, Pten, Sirt1, Ptch1, and Tie2.
The team then decided to focus on Mpl. They used flow cytometry to select LTHSCs with either high or low Mpl expression from CML-model BCR-ABL mice.
Seven of 16 mice receiving Mpl-Hi LTHSCs developed leukemia after transplantation, compared with 1 of 17 receiving Mpl-Lo LTHSCs. This suggested an increased leukemogenic capacity for the Mpl-Hi LTHSCs.
The researchers also investigated the impact of cell-cycle status. They found that CML Mpl-Hi LTHSCs that were in a resting stage of the cell cycle had enhanced long-term engraftment and leukemogenic capacity compared with cycling Mpl-Hi LTHSCs.
The team used virus vectors and shRNA to create Mpl knockdown BCR-ABL LTHSCs and showed that the knockdown cells had a greatly reduced ability to produce leukemia in recipient mice.
The Mpl knockdown cells, after stimulation by the Mpl ligand thrombopoietin, also had reduced expression of the activated transcription factors p-STAT3 and p-STAT5, compared with controls.
Finally, the researchers examined human CML cells for differences between MPL-Hi LTHSCs and MPL-Lo LTHSCs. The results were similar to those observed in mice.
The human MPL-Hi LTHSCs had a higher rate of engraftment than the human MPL-Lo LTHSCs, as tested in a xenograft model using immunodeficient mice.
Additionally, the human MPL-Hi LTHSCs had reduced sensitivity to nilotinib compared with MPL-Lo LTHSCs. However, a Jak/STAT inhibitor significantly reduced cell growth and increased apoptosis in human MPL-Hi LTHSCs.
The researchers concluded that MPL expression is a marker and key regulator of leukemogenic potential and drug sensitivity of CML LTHSCs. They said their findings support further investigation of approaches to antagonize MPL signaling as a potential therapeutic strategy to eliminate leukemia-initiating LTHSCs.

Image by Difu Wu
Preclinical research suggests chronic myeloid leukemia (CML) patients have a heterogeneous population of long-term hematopoietic stem cells (LTHSCs)—some that can initiate leukemia and some that cannot.
Researchers found they could identify the leukemia-initiating cells by measuring expression of the thrombopoietin receptor MPL. Cells with high MPL expression could initiate CML in mice.
The team said these results suggest the leukemic LTHSCs are the cells responsible for relapse in CML.
“This shows that not all leukemia stem cells are equal,” said study author Ravi Bhatia, MD, of the University of Alabama Birmingham.
“Some are more prone to causing leukemia and relapses, while some others may just hang around without potential for contributing to relapse.”
Dr Bhatia and his colleagues reported these findings in The Journal of Clinical Investigation.
In their experiments, the team used an inducible transgenic mouse model of CML, where the BCR-ABL gene fusion is under the control of a tetracycline-regulated enhancer. This model creates a chronic myeloproliferative disorder that resembles chronic phase CML.
Previous work had shown that only cells with an LTHSC phenotype were capable of long-term repopulation and leukemia-initiating capacity after transplantation to another mouse.
When the researchers transplanted LTHSCs from CML-model mice to other mice, 11 of 20 recipients developed CML, and 9 of 20 showed engraftment with CML cells but did not develop the leukocytosis characteristic of leukemia.
When the LTHSCs from the primary-recipient mice were transferred to secondary recipients, 7 of 17 mice receiving cells from leukemic mice developed CML, and none of the secondary-recipient mice receiving cells from the non-leukemic mice developed CML.
The researchers tested these 2 groups of LTHSCs for differences in gene expression. They found significant differences between the leukemic and non-leukemic LTHSCs for the genes Mpl, c-Myc, CD47, Pten, Sirt1, Ptch1, and Tie2.
The team then decided to focus on Mpl. They used flow cytometry to select LTHSCs with either high or low Mpl expression from CML-model BCR-ABL mice.
Seven of 16 mice receiving Mpl-Hi LTHSCs developed leukemia after transplantation, compared with 1 of 17 receiving Mpl-Lo LTHSCs. This suggested an increased leukemogenic capacity for the Mpl-Hi LTHSCs.
The researchers also investigated the impact of cell-cycle status. They found that CML Mpl-Hi LTHSCs that were in a resting stage of the cell cycle had enhanced long-term engraftment and leukemogenic capacity compared with cycling Mpl-Hi LTHSCs.
The team used virus vectors and shRNA to create Mpl knockdown BCR-ABL LTHSCs and showed that the knockdown cells had a greatly reduced ability to produce leukemia in recipient mice.
The Mpl knockdown cells, after stimulation by the Mpl ligand thrombopoietin, also had reduced expression of the activated transcription factors p-STAT3 and p-STAT5, compared with controls.
Finally, the researchers examined human CML cells for differences between MPL-Hi LTHSCs and MPL-Lo LTHSCs. The results were similar to those observed in mice.
The human MPL-Hi LTHSCs had a higher rate of engraftment than the human MPL-Lo LTHSCs, as tested in a xenograft model using immunodeficient mice.
Additionally, the human MPL-Hi LTHSCs had reduced sensitivity to nilotinib compared with MPL-Lo LTHSCs. However, a Jak/STAT inhibitor significantly reduced cell growth and increased apoptosis in human MPL-Hi LTHSCs.
The researchers concluded that MPL expression is a marker and key regulator of leukemogenic potential and drug sensitivity of CML LTHSCs. They said their findings support further investigation of approaches to antagonize MPL signaling as a potential therapeutic strategy to eliminate leukemia-initiating LTHSCs.
Study: Academic centers fail to report trial results

Photo by Daniel Sone
A study published in The BMJ indicates that leading US academic medical centers often fail to report clinical trial results in a timely manner, despite ethical obligations and even statutory requirements to do so.
Of the more than 4000 clinical trials studied, 67% had results disseminated at some point after trial completion, 29% were published within 2 years of completion, and 13% had results posted on ClinicalTrials.gov within 2 years of completion.
Harlan Krumholz, MD, of Yale School of Medicine in New Haven, Connecticut, and his colleagues conducted this study.
They evaluated 4347 registered trials conducted at 51 leading US academic institutions and completed between October 2007 and September 2010.
Overall, 67% of the trials (2892/4347) had been published or had results reported as of July 2014. Thirty-six percent (n=1560) had results disseminated within 2 years of trial completion.
Fifty-seven percent of trials (n=2458) had been published as of July 2014, and 29% (n=1245) were published within 2 years of completion.
Twenty-seven percent of trials (n=1166) had results reported on ClinicalTrials.gov as of July 2014, and 13% (n=547) had results reported within 2 years of completion.
There was marked variation in the dissemination of trial results across institutions. There was a more than 2-fold variation in the average time from study completion to dissemination of results and a more than 3-fold variation in the rate of dissemination across institutions.
Dr Krumholz and his colleagues noted that there are no repercussions to academic institutions or individual investigators for failing to report trial results, and there is no effective enforcement mechanism.

Photo by Daniel Sone
A study published in The BMJ indicates that leading US academic medical centers often fail to report clinical trial results in a timely manner, despite ethical obligations and even statutory requirements to do so.
Of the more than 4000 clinical trials studied, 67% had results disseminated at some point after trial completion, 29% were published within 2 years of completion, and 13% had results posted on ClinicalTrials.gov within 2 years of completion.
Harlan Krumholz, MD, of Yale School of Medicine in New Haven, Connecticut, and his colleagues conducted this study.
They evaluated 4347 registered trials conducted at 51 leading US academic institutions and completed between October 2007 and September 2010.
Overall, 67% of the trials (2892/4347) had been published or had results reported as of July 2014. Thirty-six percent (n=1560) had results disseminated within 2 years of trial completion.
Fifty-seven percent of trials (n=2458) had been published as of July 2014, and 29% (n=1245) were published within 2 years of completion.
Twenty-seven percent of trials (n=1166) had results reported on ClinicalTrials.gov as of July 2014, and 13% (n=547) had results reported within 2 years of completion.
There was marked variation in the dissemination of trial results across institutions. There was a more than 2-fold variation in the average time from study completion to dissemination of results and a more than 3-fold variation in the rate of dissemination across institutions.
Dr Krumholz and his colleagues noted that there are no repercussions to academic institutions or individual investigators for failing to report trial results, and there is no effective enforcement mechanism.

Photo by Daniel Sone
A study published in The BMJ indicates that leading US academic medical centers often fail to report clinical trial results in a timely manner, despite ethical obligations and even statutory requirements to do so.
Of the more than 4000 clinical trials studied, 67% had results disseminated at some point after trial completion, 29% were published within 2 years of completion, and 13% had results posted on ClinicalTrials.gov within 2 years of completion.
Harlan Krumholz, MD, of Yale School of Medicine in New Haven, Connecticut, and his colleagues conducted this study.
They evaluated 4347 registered trials conducted at 51 leading US academic institutions and completed between October 2007 and September 2010.
Overall, 67% of the trials (2892/4347) had been published or had results reported as of July 2014. Thirty-six percent (n=1560) had results disseminated within 2 years of trial completion.
Fifty-seven percent of trials (n=2458) had been published as of July 2014, and 29% (n=1245) were published within 2 years of completion.
Twenty-seven percent of trials (n=1166) had results reported on ClinicalTrials.gov as of July 2014, and 13% (n=547) had results reported within 2 years of completion.
There was marked variation in the dissemination of trial results across institutions. There was a more than 2-fold variation in the average time from study completion to dissemination of results and a more than 3-fold variation in the rate of dissemination across institutions.
Dr Krumholz and his colleagues noted that there are no repercussions to academic institutions or individual investigators for failing to report trial results, and there is no effective enforcement mechanism.