User login
Arginine plays role in thalassemia complication
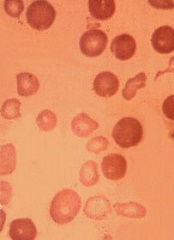
Low bioavailability of the amino acid arginine may contribute to cardiopulmonary dysfunction in patients with β-thalassemia, according to researchers.
Via previous work, the team found that arginine deficiency is a major factor in acute pain episodes in sickle cell disease.
With the current study, they discovered that arginase activity and concentration correlate with echocardiographic and cardiac-MRI measurements of cardiopulmonary function.
Claudia R. Morris, MD, of Emory University School of Medicine in Atlanta, Georgia, and her colleagues described this research in the British Journal of Haematology.
“We are finding that arginine dysregulation is an important hematologic mechanism beyond sickle cell disease,” Dr Morris said. “This new study shows that it plays a role in thalassemia patients as well and may contribute to cardiopulmonary dysfunction. Interventions aimed at restoring arginine bioavailability could be a promising area of focus for new therapeutics.”
Dr Morris and her colleagues noted that pulmonary hypertension (PH) is a common problem in patients with thalassemia. An elevated tricuspid-regurgitant jet-velocity (TRV) of 2.5 m/s or greater on Doppler echocardiography can identify patients with an increased risk of PH. But a right heart catheterization is required to confirm the condition.
The researchers wanted to provide a comprehensive description of the cardiopulmonary and biological profile of thalassemia patients at risk of developing PH. So they analyzed 27 patients with β-thalassemia.
Fourteen patients had an elevated TRV (≥ 2.5 m/s). According to echocardiography, these patients had a significantly larger right atrial size (P=0.03), left atrial size (P=0.002), left ventricular (LV) mass (P=0.03), and left septal-wall thickness (P=0.03) than patients with a TRV below 2.5 m/s.
According to MRI, patients with elevated TRV had significantly higher left atrial volume than patients with a TRV < 2.5 m/s (P=0.008).
Patients with an elevated TRV also had elevated lactate dehydrogenase (LDH) levels (P=0.03) and biomarkers of abnormal coagulation, including thrombin-antithrombin complex (P=0.04) and monoclonal prothrombin fragment 1.2 (P=0.02).
Patients with a TRV ≥ 2.5 m/s had significantly lower plasma arginine concentration (P<0.001) and biomarkers of global arginine bioavailability, including plasma arginine/ornithine ratio (P=0.01) and mean plasma global arginine bioavailability ratio (arginine/ornithine + citrulline ratio, P=0.04).
These patients also had significantly higher arginase concentration and activity than patients with TRV below 2.5 m/s (P=0.02).
These findings prompted the researchers to evaluate the relationship between arginase activity/concentration and clinical and laboratory markers of disease severity.
They found that arginase concentration was significantly correlated with several parameters of cardiovascular function, including left atrial volume (echo and MRI), right atrial volume (MRI), LV end systolic volume (echo and MRI), LV end diastolic volume (echo and MRI), LV mass (echo and MRI), and cardiac index (echo).
Arginase concentration was also significantly correlated with white blood cell count (P=0.03), plasma arginine (P=0.0002), arginine/ornithine (P=0.01) and arginine/ornithine + citrulline ratios (P=0.05), and hemoglobin (P=0.02), bilirubin (P=0.02), and LDH levels (P=0.001).
In multiple regression analysis, only cardiac index, bilirubin, and plasma arginine/ornithine ratio remained significantly associated with arginase concentration (P<0.05 for all).
Arginase activity was significantly correlated with several biomarkers of coagulation, including monoclonal prothrombin fragment 1.2 (P=0.04), thrombin-antithrombin complex (P=0.04), and tissue factor concentration (P=0.02).
Considering these results together, the researchers said it seems low arginine bioavailability contributes to cardiopulmonary dysfunction in patients with β-thalassemia. ![]()

Low bioavailability of the amino acid arginine may contribute to cardiopulmonary dysfunction in patients with β-thalassemia, according to researchers.
Via previous work, the team found that arginine deficiency is a major factor in acute pain episodes in sickle cell disease.
With the current study, they discovered that arginase activity and concentration correlate with echocardiographic and cardiac-MRI measurements of cardiopulmonary function.
Claudia R. Morris, MD, of Emory University School of Medicine in Atlanta, Georgia, and her colleagues described this research in the British Journal of Haematology.
“We are finding that arginine dysregulation is an important hematologic mechanism beyond sickle cell disease,” Dr Morris said. “This new study shows that it plays a role in thalassemia patients as well and may contribute to cardiopulmonary dysfunction. Interventions aimed at restoring arginine bioavailability could be a promising area of focus for new therapeutics.”
Dr Morris and her colleagues noted that pulmonary hypertension (PH) is a common problem in patients with thalassemia. An elevated tricuspid-regurgitant jet-velocity (TRV) of 2.5 m/s or greater on Doppler echocardiography can identify patients with an increased risk of PH. But a right heart catheterization is required to confirm the condition.
The researchers wanted to provide a comprehensive description of the cardiopulmonary and biological profile of thalassemia patients at risk of developing PH. So they analyzed 27 patients with β-thalassemia.
Fourteen patients had an elevated TRV (≥ 2.5 m/s). According to echocardiography, these patients had a significantly larger right atrial size (P=0.03), left atrial size (P=0.002), left ventricular (LV) mass (P=0.03), and left septal-wall thickness (P=0.03) than patients with a TRV below 2.5 m/s.
According to MRI, patients with elevated TRV had significantly higher left atrial volume than patients with a TRV < 2.5 m/s (P=0.008).
Patients with an elevated TRV also had elevated lactate dehydrogenase (LDH) levels (P=0.03) and biomarkers of abnormal coagulation, including thrombin-antithrombin complex (P=0.04) and monoclonal prothrombin fragment 1.2 (P=0.02).
Patients with a TRV ≥ 2.5 m/s had significantly lower plasma arginine concentration (P<0.001) and biomarkers of global arginine bioavailability, including plasma arginine/ornithine ratio (P=0.01) and mean plasma global arginine bioavailability ratio (arginine/ornithine + citrulline ratio, P=0.04).
These patients also had significantly higher arginase concentration and activity than patients with TRV below 2.5 m/s (P=0.02).
These findings prompted the researchers to evaluate the relationship between arginase activity/concentration and clinical and laboratory markers of disease severity.
They found that arginase concentration was significantly correlated with several parameters of cardiovascular function, including left atrial volume (echo and MRI), right atrial volume (MRI), LV end systolic volume (echo and MRI), LV end diastolic volume (echo and MRI), LV mass (echo and MRI), and cardiac index (echo).
Arginase concentration was also significantly correlated with white blood cell count (P=0.03), plasma arginine (P=0.0002), arginine/ornithine (P=0.01) and arginine/ornithine + citrulline ratios (P=0.05), and hemoglobin (P=0.02), bilirubin (P=0.02), and LDH levels (P=0.001).
In multiple regression analysis, only cardiac index, bilirubin, and plasma arginine/ornithine ratio remained significantly associated with arginase concentration (P<0.05 for all).
Arginase activity was significantly correlated with several biomarkers of coagulation, including monoclonal prothrombin fragment 1.2 (P=0.04), thrombin-antithrombin complex (P=0.04), and tissue factor concentration (P=0.02).
Considering these results together, the researchers said it seems low arginine bioavailability contributes to cardiopulmonary dysfunction in patients with β-thalassemia. ![]()

Low bioavailability of the amino acid arginine may contribute to cardiopulmonary dysfunction in patients with β-thalassemia, according to researchers.
Via previous work, the team found that arginine deficiency is a major factor in acute pain episodes in sickle cell disease.
With the current study, they discovered that arginase activity and concentration correlate with echocardiographic and cardiac-MRI measurements of cardiopulmonary function.
Claudia R. Morris, MD, of Emory University School of Medicine in Atlanta, Georgia, and her colleagues described this research in the British Journal of Haematology.
“We are finding that arginine dysregulation is an important hematologic mechanism beyond sickle cell disease,” Dr Morris said. “This new study shows that it plays a role in thalassemia patients as well and may contribute to cardiopulmonary dysfunction. Interventions aimed at restoring arginine bioavailability could be a promising area of focus for new therapeutics.”
Dr Morris and her colleagues noted that pulmonary hypertension (PH) is a common problem in patients with thalassemia. An elevated tricuspid-regurgitant jet-velocity (TRV) of 2.5 m/s or greater on Doppler echocardiography can identify patients with an increased risk of PH. But a right heart catheterization is required to confirm the condition.
The researchers wanted to provide a comprehensive description of the cardiopulmonary and biological profile of thalassemia patients at risk of developing PH. So they analyzed 27 patients with β-thalassemia.
Fourteen patients had an elevated TRV (≥ 2.5 m/s). According to echocardiography, these patients had a significantly larger right atrial size (P=0.03), left atrial size (P=0.002), left ventricular (LV) mass (P=0.03), and left septal-wall thickness (P=0.03) than patients with a TRV below 2.5 m/s.
According to MRI, patients with elevated TRV had significantly higher left atrial volume than patients with a TRV < 2.5 m/s (P=0.008).
Patients with an elevated TRV also had elevated lactate dehydrogenase (LDH) levels (P=0.03) and biomarkers of abnormal coagulation, including thrombin-antithrombin complex (P=0.04) and monoclonal prothrombin fragment 1.2 (P=0.02).
Patients with a TRV ≥ 2.5 m/s had significantly lower plasma arginine concentration (P<0.001) and biomarkers of global arginine bioavailability, including plasma arginine/ornithine ratio (P=0.01) and mean plasma global arginine bioavailability ratio (arginine/ornithine + citrulline ratio, P=0.04).
These patients also had significantly higher arginase concentration and activity than patients with TRV below 2.5 m/s (P=0.02).
These findings prompted the researchers to evaluate the relationship between arginase activity/concentration and clinical and laboratory markers of disease severity.
They found that arginase concentration was significantly correlated with several parameters of cardiovascular function, including left atrial volume (echo and MRI), right atrial volume (MRI), LV end systolic volume (echo and MRI), LV end diastolic volume (echo and MRI), LV mass (echo and MRI), and cardiac index (echo).
Arginase concentration was also significantly correlated with white blood cell count (P=0.03), plasma arginine (P=0.0002), arginine/ornithine (P=0.01) and arginine/ornithine + citrulline ratios (P=0.05), and hemoglobin (P=0.02), bilirubin (P=0.02), and LDH levels (P=0.001).
In multiple regression analysis, only cardiac index, bilirubin, and plasma arginine/ornithine ratio remained significantly associated with arginase concentration (P<0.05 for all).
Arginase activity was significantly correlated with several biomarkers of coagulation, including monoclonal prothrombin fragment 1.2 (P=0.04), thrombin-antithrombin complex (P=0.04), and tissue factor concentration (P=0.02).
Considering these results together, the researchers said it seems low arginine bioavailability contributes to cardiopulmonary dysfunction in patients with β-thalassemia. ![]()
Mosquitoes can accumulate malaria infections
Photo courtesy of the CDC
Repeat infection with malaria parasites might make mosquitoes more dangerous, according to a study published in PLOS Pathogens.
The research showed that individual Anopheles mosquitoes can accumulate infections with different strains of malaria parasites, an existing malaria infection makes mosquitoes more susceptible to a second infection, and infections reach higher densities when another strain is already present.
These phenomena could promote the spread of drug-resistant malaria, according to investigators.
Laura Pollitt, PhD, of the University of Edinburgh in the UK, and her colleagues conducted this research.
They wanted to determine if and how mosquitoes can be infected with different strains of Plasmodium parasites, how such heterogeneous parasites interact in the insects, and whether such interactions affect the transmission of malaria to vertebrate hosts.
The investigators set up cages of female Anopheles stephensi mosquitoes and allowed them, at defined times, to feed on mice infected with 2 different Plasmodium chabaudi strains—AJ and ER.
This study design allowed the team to examine how the presence of a co-infecting strain affects parasites that enter the vector first and second, and to test whether co-infection impacts mosquito survival.
The investigators found that mosquitoes can accumulate mixed-strain malaria infections after feeding on multiple hosts. And parasites have a greater chance of establishing a secondary infection if another Plasmodium strain is already present in a mosquito.
Moreover, the presence of the primary infection facilitated replication of the secondary infection while the first infection developed as normal. This resulted in doubly infected mosquitoes having substantially higher parasite loads.
The investigators noted that the large parasite numbers do not appear to kill the insects. And, because it is expected that mosquitoes carrying more parasites are more likely to transmit malaria to vertebrates, mosquitoes taking multiple infective bites might disproportionally contribute to malaria transmission.
This, in turn, would increase rates of mixed infections in vertebrate hosts, with implications for the evolution of parasite virulence and the spread of drug-resistant strains. ![]()

Photo courtesy of the CDC
Repeat infection with malaria parasites might make mosquitoes more dangerous, according to a study published in PLOS Pathogens.
The research showed that individual Anopheles mosquitoes can accumulate infections with different strains of malaria parasites, an existing malaria infection makes mosquitoes more susceptible to a second infection, and infections reach higher densities when another strain is already present.
These phenomena could promote the spread of drug-resistant malaria, according to investigators.
Laura Pollitt, PhD, of the University of Edinburgh in the UK, and her colleagues conducted this research.
They wanted to determine if and how mosquitoes can be infected with different strains of Plasmodium parasites, how such heterogeneous parasites interact in the insects, and whether such interactions affect the transmission of malaria to vertebrate hosts.
The investigators set up cages of female Anopheles stephensi mosquitoes and allowed them, at defined times, to feed on mice infected with 2 different Plasmodium chabaudi strains—AJ and ER.
This study design allowed the team to examine how the presence of a co-infecting strain affects parasites that enter the vector first and second, and to test whether co-infection impacts mosquito survival.
The investigators found that mosquitoes can accumulate mixed-strain malaria infections after feeding on multiple hosts. And parasites have a greater chance of establishing a secondary infection if another Plasmodium strain is already present in a mosquito.
Moreover, the presence of the primary infection facilitated replication of the secondary infection while the first infection developed as normal. This resulted in doubly infected mosquitoes having substantially higher parasite loads.
The investigators noted that the large parasite numbers do not appear to kill the insects. And, because it is expected that mosquitoes carrying more parasites are more likely to transmit malaria to vertebrates, mosquitoes taking multiple infective bites might disproportionally contribute to malaria transmission.
This, in turn, would increase rates of mixed infections in vertebrate hosts, with implications for the evolution of parasite virulence and the spread of drug-resistant strains. ![]()

Photo courtesy of the CDC
Repeat infection with malaria parasites might make mosquitoes more dangerous, according to a study published in PLOS Pathogens.
The research showed that individual Anopheles mosquitoes can accumulate infections with different strains of malaria parasites, an existing malaria infection makes mosquitoes more susceptible to a second infection, and infections reach higher densities when another strain is already present.
These phenomena could promote the spread of drug-resistant malaria, according to investigators.
Laura Pollitt, PhD, of the University of Edinburgh in the UK, and her colleagues conducted this research.
They wanted to determine if and how mosquitoes can be infected with different strains of Plasmodium parasites, how such heterogeneous parasites interact in the insects, and whether such interactions affect the transmission of malaria to vertebrate hosts.
The investigators set up cages of female Anopheles stephensi mosquitoes and allowed them, at defined times, to feed on mice infected with 2 different Plasmodium chabaudi strains—AJ and ER.
This study design allowed the team to examine how the presence of a co-infecting strain affects parasites that enter the vector first and second, and to test whether co-infection impacts mosquito survival.
The investigators found that mosquitoes can accumulate mixed-strain malaria infections after feeding on multiple hosts. And parasites have a greater chance of establishing a secondary infection if another Plasmodium strain is already present in a mosquito.
Moreover, the presence of the primary infection facilitated replication of the secondary infection while the first infection developed as normal. This resulted in doubly infected mosquitoes having substantially higher parasite loads.
The investigators noted that the large parasite numbers do not appear to kill the insects. And, because it is expected that mosquitoes carrying more parasites are more likely to transmit malaria to vertebrates, mosquitoes taking multiple infective bites might disproportionally contribute to malaria transmission.
This, in turn, would increase rates of mixed infections in vertebrate hosts, with implications for the evolution of parasite virulence and the spread of drug-resistant strains. ![]()
New antimalarial progresses to clinical trials

falciparum
in a mouse liverImage courtesy of Seattle
Biomedical Research Institute
A new compound can fight multidrug-resistant malaria, according to preclinical research published in Science Translational Medicine.
The compound, DSM265, targets DHODH, an enzyme involved in the malaria parasite’s production of nucleotides.
Experiments showed that DSM265 can target the parasite at both the blood and liver stages of infection. And the compound appeared to be safe.
Based on these results, DSM265 has advanced to clinical trials.
Investigators believe the drug could potentially be partnered with other antimalarials for a single-dose treatment or once-weekly prophylaxis.
“This is the first of a new class of molecules that’s going into humans,” said study author Pradipsinh Rathod, PhD, of the University of Washington in Seattle.
“Until now, everything else in humans has been variations of drugs that have been developed in the distant past.”
The investigators tested DSM265 in multiple models, including human cells, rodents, dogs, and monkeys.
DSM265 arrested growth of the Plasmodium falciparum parasite at both the blood and liver stages. And the drug was active against P falciparum strains that were resistant to chloroquine and pyrimethamine.
DSM265 also proved to be long-acting. Experiments suggested a dose of 200 mg to 400 mg would maintain therapeutic concentrations in humans for at least 8 days.
Repeated doses of DSM265, given to mice and dogs, did not cause major side effects. The researchers said DSM265 was well tolerated at all dose levels given to mice (25 to 200 mg/kg per day once daily for 7 days) and dogs (30 to 480 mg/kg every other day for 10 days).
None of the animals died, and there were no clinical effects observed in the mice. Dogs receiving the highest dose of DSM265 experienced vomiting, apparent loss of appetite, and a slight increase in bilirubin that was not accompanied by changes in any other liver function markers.
Dr Rathod said he hopes the development and discovery pipeline for DSM265 will pave the way for a faster and more collaborative drug development process in “the long war against malaria.”
In an attempt to accelerate the drug’s development, the investigators transferred their patent rights for DSM265 to the Medicines for Malaria Venture, a Bill & Melinda Gates Foundation-supported nonprofit that is leading some of the clinical and field trials. ![]()

falciparum
in a mouse liverImage courtesy of Seattle
Biomedical Research Institute
A new compound can fight multidrug-resistant malaria, according to preclinical research published in Science Translational Medicine.
The compound, DSM265, targets DHODH, an enzyme involved in the malaria parasite’s production of nucleotides.
Experiments showed that DSM265 can target the parasite at both the blood and liver stages of infection. And the compound appeared to be safe.
Based on these results, DSM265 has advanced to clinical trials.
Investigators believe the drug could potentially be partnered with other antimalarials for a single-dose treatment or once-weekly prophylaxis.
“This is the first of a new class of molecules that’s going into humans,” said study author Pradipsinh Rathod, PhD, of the University of Washington in Seattle.
“Until now, everything else in humans has been variations of drugs that have been developed in the distant past.”
The investigators tested DSM265 in multiple models, including human cells, rodents, dogs, and monkeys.
DSM265 arrested growth of the Plasmodium falciparum parasite at both the blood and liver stages. And the drug was active against P falciparum strains that were resistant to chloroquine and pyrimethamine.
DSM265 also proved to be long-acting. Experiments suggested a dose of 200 mg to 400 mg would maintain therapeutic concentrations in humans for at least 8 days.
Repeated doses of DSM265, given to mice and dogs, did not cause major side effects. The researchers said DSM265 was well tolerated at all dose levels given to mice (25 to 200 mg/kg per day once daily for 7 days) and dogs (30 to 480 mg/kg every other day for 10 days).
None of the animals died, and there were no clinical effects observed in the mice. Dogs receiving the highest dose of DSM265 experienced vomiting, apparent loss of appetite, and a slight increase in bilirubin that was not accompanied by changes in any other liver function markers.
Dr Rathod said he hopes the development and discovery pipeline for DSM265 will pave the way for a faster and more collaborative drug development process in “the long war against malaria.”
In an attempt to accelerate the drug’s development, the investigators transferred their patent rights for DSM265 to the Medicines for Malaria Venture, a Bill & Melinda Gates Foundation-supported nonprofit that is leading some of the clinical and field trials. ![]()

falciparum
in a mouse liverImage courtesy of Seattle
Biomedical Research Institute
A new compound can fight multidrug-resistant malaria, according to preclinical research published in Science Translational Medicine.
The compound, DSM265, targets DHODH, an enzyme involved in the malaria parasite’s production of nucleotides.
Experiments showed that DSM265 can target the parasite at both the blood and liver stages of infection. And the compound appeared to be safe.
Based on these results, DSM265 has advanced to clinical trials.
Investigators believe the drug could potentially be partnered with other antimalarials for a single-dose treatment or once-weekly prophylaxis.
“This is the first of a new class of molecules that’s going into humans,” said study author Pradipsinh Rathod, PhD, of the University of Washington in Seattle.
“Until now, everything else in humans has been variations of drugs that have been developed in the distant past.”
The investigators tested DSM265 in multiple models, including human cells, rodents, dogs, and monkeys.
DSM265 arrested growth of the Plasmodium falciparum parasite at both the blood and liver stages. And the drug was active against P falciparum strains that were resistant to chloroquine and pyrimethamine.
DSM265 also proved to be long-acting. Experiments suggested a dose of 200 mg to 400 mg would maintain therapeutic concentrations in humans for at least 8 days.
Repeated doses of DSM265, given to mice and dogs, did not cause major side effects. The researchers said DSM265 was well tolerated at all dose levels given to mice (25 to 200 mg/kg per day once daily for 7 days) and dogs (30 to 480 mg/kg every other day for 10 days).
None of the animals died, and there were no clinical effects observed in the mice. Dogs receiving the highest dose of DSM265 experienced vomiting, apparent loss of appetite, and a slight increase in bilirubin that was not accompanied by changes in any other liver function markers.
Dr Rathod said he hopes the development and discovery pipeline for DSM265 will pave the way for a faster and more collaborative drug development process in “the long war against malaria.”
In an attempt to accelerate the drug’s development, the investigators transferred their patent rights for DSM265 to the Medicines for Malaria Venture, a Bill & Melinda Gates Foundation-supported nonprofit that is leading some of the clinical and field trials. ![]()
Neutrophils cause hemorrhage in thrombocytopenia

Image by Volker Brinkmann
Researchers have found that hemorrhage occurs in the context of thrombocytopenia when neutrophils cross the endothelial barrier.
The team also identified ways to inhibit this diapedesis and prevent hemorrhage in mouse models.
If these results can be replicated in humans, such interventions could prevent bleeding in patients with immune thrombocytopenia and certain patients receiving chemotherapy or hematopoietic stem cell transplant.
Carina Hillgruber, PhD, of the University of Munster in Germany, and her colleagues conducted this research and detailed the results in the Journal of Experimental Medicine.
Previous studies showed that an absence of platelets alone was not sufficient to cause hemorrhage. Inflammation was required. But the exact cause of bleeding complications in thrombocytopenia was unknown.
With their research, Dr Hillgruber and her colleagues found that neutrophils are recruited to inflammatory sites to induce thrombocytopenic tissue hemorrhage. The process consists of Gαi2-mediated neutrophil transmigratory activity and opening of the endothelial barrier via VE-cadherin.
So the researchers speculated that preventing neutrophil diapedesis by either tightening the endothelial barrier or targeting neutrophil transmigratory activity would prevent hemorrhage.
The team showed they could inhibit the interaction between neutrophils and the endothelium by interfering with P-selectin, β2integrin-mediated adhesion, and chemokine signaling. This led to reduced inflammatory bleeding in thrombocytopenic mice.
The researchers also found they could interfere with Gαi signaling in neutrophils (which is critical for the cells’ transmigration) via treatment with pertussis toxin or gene ablation. And this protected thrombocytopenic mice from cutaneous hemorrhage.
Finally, the team showed that mice harboring the VE-cadherin mutation Y731F were protected from hemorrhage despite having low platelet counts and immune complex-mediated vasculitis.
The researchers said these findings suggest therapeutically targeting neutrophil diapedesis through the endothelial barrier could potentially prevent hemorrhage in patients with thrombocytopenia.
The team noted that platelets must seal the damage induced by neutrophil diapedesis, but they could only speculate as to how that occurs. ![]()

Image by Volker Brinkmann
Researchers have found that hemorrhage occurs in the context of thrombocytopenia when neutrophils cross the endothelial barrier.
The team also identified ways to inhibit this diapedesis and prevent hemorrhage in mouse models.
If these results can be replicated in humans, such interventions could prevent bleeding in patients with immune thrombocytopenia and certain patients receiving chemotherapy or hematopoietic stem cell transplant.
Carina Hillgruber, PhD, of the University of Munster in Germany, and her colleagues conducted this research and detailed the results in the Journal of Experimental Medicine.
Previous studies showed that an absence of platelets alone was not sufficient to cause hemorrhage. Inflammation was required. But the exact cause of bleeding complications in thrombocytopenia was unknown.
With their research, Dr Hillgruber and her colleagues found that neutrophils are recruited to inflammatory sites to induce thrombocytopenic tissue hemorrhage. The process consists of Gαi2-mediated neutrophil transmigratory activity and opening of the endothelial barrier via VE-cadherin.
So the researchers speculated that preventing neutrophil diapedesis by either tightening the endothelial barrier or targeting neutrophil transmigratory activity would prevent hemorrhage.
The team showed they could inhibit the interaction between neutrophils and the endothelium by interfering with P-selectin, β2integrin-mediated adhesion, and chemokine signaling. This led to reduced inflammatory bleeding in thrombocytopenic mice.
The researchers also found they could interfere with Gαi signaling in neutrophils (which is critical for the cells’ transmigration) via treatment with pertussis toxin or gene ablation. And this protected thrombocytopenic mice from cutaneous hemorrhage.
Finally, the team showed that mice harboring the VE-cadherin mutation Y731F were protected from hemorrhage despite having low platelet counts and immune complex-mediated vasculitis.
The researchers said these findings suggest therapeutically targeting neutrophil diapedesis through the endothelial barrier could potentially prevent hemorrhage in patients with thrombocytopenia.
The team noted that platelets must seal the damage induced by neutrophil diapedesis, but they could only speculate as to how that occurs. ![]()

Image by Volker Brinkmann
Researchers have found that hemorrhage occurs in the context of thrombocytopenia when neutrophils cross the endothelial barrier.
The team also identified ways to inhibit this diapedesis and prevent hemorrhage in mouse models.
If these results can be replicated in humans, such interventions could prevent bleeding in patients with immune thrombocytopenia and certain patients receiving chemotherapy or hematopoietic stem cell transplant.
Carina Hillgruber, PhD, of the University of Munster in Germany, and her colleagues conducted this research and detailed the results in the Journal of Experimental Medicine.
Previous studies showed that an absence of platelets alone was not sufficient to cause hemorrhage. Inflammation was required. But the exact cause of bleeding complications in thrombocytopenia was unknown.
With their research, Dr Hillgruber and her colleagues found that neutrophils are recruited to inflammatory sites to induce thrombocytopenic tissue hemorrhage. The process consists of Gαi2-mediated neutrophil transmigratory activity and opening of the endothelial barrier via VE-cadherin.
So the researchers speculated that preventing neutrophil diapedesis by either tightening the endothelial barrier or targeting neutrophil transmigratory activity would prevent hemorrhage.
The team showed they could inhibit the interaction between neutrophils and the endothelium by interfering with P-selectin, β2integrin-mediated adhesion, and chemokine signaling. This led to reduced inflammatory bleeding in thrombocytopenic mice.
The researchers also found they could interfere with Gαi signaling in neutrophils (which is critical for the cells’ transmigration) via treatment with pertussis toxin or gene ablation. And this protected thrombocytopenic mice from cutaneous hemorrhage.
Finally, the team showed that mice harboring the VE-cadherin mutation Y731F were protected from hemorrhage despite having low platelet counts and immune complex-mediated vasculitis.
The researchers said these findings suggest therapeutically targeting neutrophil diapedesis through the endothelial barrier could potentially prevent hemorrhage in patients with thrombocytopenia.
The team noted that platelets must seal the damage induced by neutrophil diapedesis, but they could only speculate as to how that occurs. ![]()
Drug granted orphan designation for MM

The US Food and Drug Administration (FDA) has granted orphan designation for the drug CB-5083 to treat multiple myeloma (MM).
CB-5083 works by inhibiting p97, an enzyme that controls various aspects of protein homeostasis.
Inhibiting p97 function in cancers has been shown to generate irresolvable endoplasmic reticulum stress that induces a lethal unfolded protein response, which leads to apoptosis and antitumor activity.
CB-5083 has exhibited anticancer activity in preclinical research, and the drug is currently under investigation in two phase 1 studies—one in MM and one in solid tumor malignancies.
Research in MM
Preclinical research presented at ASH 2014 showed that CB-5083 inhibits MM cell viability in vitro. The drug also inhibited tumor growth in xenograft models of MM and in an orthometastatic model of MM.
In addition, CB-5083 demonstrated synergy with carfilzomib, bortezomib, and combination lenalidomide-dexamethasone.
CB-5083 is now under investigation in a phase 1 dose-escalation/dose-expansion trial of MM patients.
Researchers are evaluating the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of the drug in patients who have relapsed/refractory or refractory MM after receiving 2 or more lines of therapy, including an immunomodulatory agent and a proteasome inhibitor.
The goal is to enroll up to 60 patients in this trial at multiple US cancer centers that are part of the Multiple Myeloma Research Consortium. More information about the trial is available at www.clinicaltrials.gov (NCT02223598).
About orphan designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US. Orphan designation provides the sponsor of a drug with various development incentives.
The orphan designation for CB-5083 provides Cleave Biosciences with opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, 7 years of US marketing exclusivity if the drug is approved, and other benefits. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation for the drug CB-5083 to treat multiple myeloma (MM).
CB-5083 works by inhibiting p97, an enzyme that controls various aspects of protein homeostasis.
Inhibiting p97 function in cancers has been shown to generate irresolvable endoplasmic reticulum stress that induces a lethal unfolded protein response, which leads to apoptosis and antitumor activity.
CB-5083 has exhibited anticancer activity in preclinical research, and the drug is currently under investigation in two phase 1 studies—one in MM and one in solid tumor malignancies.
Research in MM
Preclinical research presented at ASH 2014 showed that CB-5083 inhibits MM cell viability in vitro. The drug also inhibited tumor growth in xenograft models of MM and in an orthometastatic model of MM.
In addition, CB-5083 demonstrated synergy with carfilzomib, bortezomib, and combination lenalidomide-dexamethasone.
CB-5083 is now under investigation in a phase 1 dose-escalation/dose-expansion trial of MM patients.
Researchers are evaluating the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of the drug in patients who have relapsed/refractory or refractory MM after receiving 2 or more lines of therapy, including an immunomodulatory agent and a proteasome inhibitor.
The goal is to enroll up to 60 patients in this trial at multiple US cancer centers that are part of the Multiple Myeloma Research Consortium. More information about the trial is available at www.clinicaltrials.gov (NCT02223598).
About orphan designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US. Orphan designation provides the sponsor of a drug with various development incentives.
The orphan designation for CB-5083 provides Cleave Biosciences with opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, 7 years of US marketing exclusivity if the drug is approved, and other benefits. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation for the drug CB-5083 to treat multiple myeloma (MM).
CB-5083 works by inhibiting p97, an enzyme that controls various aspects of protein homeostasis.
Inhibiting p97 function in cancers has been shown to generate irresolvable endoplasmic reticulum stress that induces a lethal unfolded protein response, which leads to apoptosis and antitumor activity.
CB-5083 has exhibited anticancer activity in preclinical research, and the drug is currently under investigation in two phase 1 studies—one in MM and one in solid tumor malignancies.
Research in MM
Preclinical research presented at ASH 2014 showed that CB-5083 inhibits MM cell viability in vitro. The drug also inhibited tumor growth in xenograft models of MM and in an orthometastatic model of MM.
In addition, CB-5083 demonstrated synergy with carfilzomib, bortezomib, and combination lenalidomide-dexamethasone.
CB-5083 is now under investigation in a phase 1 dose-escalation/dose-expansion trial of MM patients.
Researchers are evaluating the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of the drug in patients who have relapsed/refractory or refractory MM after receiving 2 or more lines of therapy, including an immunomodulatory agent and a proteasome inhibitor.
The goal is to enroll up to 60 patients in this trial at multiple US cancer centers that are part of the Multiple Myeloma Research Consortium. More information about the trial is available at www.clinicaltrials.gov (NCT02223598).
About orphan designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US. Orphan designation provides the sponsor of a drug with various development incentives.
The orphan designation for CB-5083 provides Cleave Biosciences with opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, 7 years of US marketing exclusivity if the drug is approved, and other benefits. ![]()
IRB members and industry relationships

Institutional Review Boards (IRBs) at academic health centers have gotten better at managing members’ industry relationships in the last several years, but issues persist, according to a new study.
The research is a follow-up to a study published in 2006.
The new study showed improvements in the management of industry relationships, with increased levels of disclosure and fewer “problematic relationships” than were reported in the previous study.
However, nearly a third of IRB members in the new study said they were unaware of their board’s conflict of interest policies. And a quarter of IRB members reported voting on protocols with which they have conflicts of interest, which is a violation of federal regulations.
“IRBs are the primary mechanisms by which institutions oversee research that involves human participants, and industry relationships of IRB members have the potential to impact their IRB-related activities in both positive and negative ways,” said Eric G. Campbell, PhD, of the Mongan Institute for Health Policy at Massachusetts General Hospital in Boston.
“While our principal findings are that, compared to a decade ago, IRBs appear to be doing a much better job at managing their members’ industry relationships, there still are findings that are concerning.”
Dr Campbell and his colleagues described these findings in JAMA Internal Medicine.
Every US institution that conducts research involving human participants must have an IRB, which reviews proposed studies to ensure their design is scientifically valid and does not violate ethical and regulatory guidelines. IRBs also monitor ongoing studies to make sure they maintain appropriate practices.
The first study to examine industry relationships of IRB members, conducted by Dr Campbell and his colleagues, was based on a survey of IRB members at 100 institutions that was published in NEJM in November 2006.
That study showed that more than a third of IRB members had some sort of financial relationship with commercial firms. And while most respondents did not believe that such relationships had an inappropriate impact on IRB decision making, significant percentages were not aware of institutional guidelines for disclosing industry relationships or defining conflicts of interest.
The current study involved an identical survey administered in 2014 to nearly 500 IRB members at 115 medical schools and teaching hospitals around the country.
As in the 2006 study, respondents were asked about specific types of relationships with commercial companies, whether they received any industry funding and for what purposes, and how many protocols had come before their IRB that involved companies with which they had a relationship.
For protocols with which IRB members had a conflict—relating either to companies they had relationships with or to competitors—they were asked whether they had disclosed their relationships to the IRB, whether they participated in discussion of those protocols, and whether they voted on the protocols.
Results
There was no significant difference between the current study and the previous study in the percentage of IRB members who reported having industry relationships overall—37% and 32%, respectively.
However, the percentage of respondents who reported receiving payments for attending meetings and conferences or for serving on speakers’ bureaus—relationships that are considered problematic—dropped significantly. The percentages fell from 16% to 9% for meeting/conference participation and from 14% to 4% for speakers’ bureaus.
“We were encouraged to see that the prevalence of potentially beneficial relationships—such as industry funding to support research studies—was essentially unchanged, indicating IRBs have not tried to eliminate members’ industry relationships across the board,” said Christine Vogeli, PhD, also of the Mongan Institute.
Survey respondents were more likely in the current study than in the previous one to say that their IRB had a written definition of conflicts of interest—63% compared with 46%. In addition, the percentage of respondents who reported their IRB had no policy related to conflicts dropped from 14% to 5%.
However, the researchers were concerned by the fact that 32% of respondents still did not know whether or not their IRB had a policy on conflicts, even though that number had dropped from 41% in the previous study.
The percentage of respondents who handled their conflicts of interest in an appropriate manner increased. Eighty percent of respondents with conflicts reported them to the IRB, up from 55% in the previous study.
And 68% of respondents said they always left the room when a protocol with which they had a conflict was being discussed, up from 38% in the previous study.
However, a quarter of respondents with conflicts indicated they had voted on protocols with which they had a conflict. While that was a drop from what was reported in the previous study, it was not statistically significant.
The percentage of respondents who felt that at least one protocol had been presented to their IRB in a biased fashion because of another member’s industry relationships dropped from 14% in the previous study to 8% in this study.
And when asked about the types of bias they perceived in the presentation—questions not included in the previous survey—8% of respondents reported a pro-industry bias, and 14% reported an anti-industry bias.
“The fact that we found any bias—either pro- or anti-industry—is an issue, since bias is antithetical to research and should be eliminated,” Dr Campbell said. “IRBs should address that issue, along with increasing efforts to educate their members about what constitutes a conflict of interest and the inappropriateness of voting on protocols with which they have a conflict.” ![]()

Institutional Review Boards (IRBs) at academic health centers have gotten better at managing members’ industry relationships in the last several years, but issues persist, according to a new study.
The research is a follow-up to a study published in 2006.
The new study showed improvements in the management of industry relationships, with increased levels of disclosure and fewer “problematic relationships” than were reported in the previous study.
However, nearly a third of IRB members in the new study said they were unaware of their board’s conflict of interest policies. And a quarter of IRB members reported voting on protocols with which they have conflicts of interest, which is a violation of federal regulations.
“IRBs are the primary mechanisms by which institutions oversee research that involves human participants, and industry relationships of IRB members have the potential to impact their IRB-related activities in both positive and negative ways,” said Eric G. Campbell, PhD, of the Mongan Institute for Health Policy at Massachusetts General Hospital in Boston.
“While our principal findings are that, compared to a decade ago, IRBs appear to be doing a much better job at managing their members’ industry relationships, there still are findings that are concerning.”
Dr Campbell and his colleagues described these findings in JAMA Internal Medicine.
Every US institution that conducts research involving human participants must have an IRB, which reviews proposed studies to ensure their design is scientifically valid and does not violate ethical and regulatory guidelines. IRBs also monitor ongoing studies to make sure they maintain appropriate practices.
The first study to examine industry relationships of IRB members, conducted by Dr Campbell and his colleagues, was based on a survey of IRB members at 100 institutions that was published in NEJM in November 2006.
That study showed that more than a third of IRB members had some sort of financial relationship with commercial firms. And while most respondents did not believe that such relationships had an inappropriate impact on IRB decision making, significant percentages were not aware of institutional guidelines for disclosing industry relationships or defining conflicts of interest.
The current study involved an identical survey administered in 2014 to nearly 500 IRB members at 115 medical schools and teaching hospitals around the country.
As in the 2006 study, respondents were asked about specific types of relationships with commercial companies, whether they received any industry funding and for what purposes, and how many protocols had come before their IRB that involved companies with which they had a relationship.
For protocols with which IRB members had a conflict—relating either to companies they had relationships with or to competitors—they were asked whether they had disclosed their relationships to the IRB, whether they participated in discussion of those protocols, and whether they voted on the protocols.
Results
There was no significant difference between the current study and the previous study in the percentage of IRB members who reported having industry relationships overall—37% and 32%, respectively.
However, the percentage of respondents who reported receiving payments for attending meetings and conferences or for serving on speakers’ bureaus—relationships that are considered problematic—dropped significantly. The percentages fell from 16% to 9% for meeting/conference participation and from 14% to 4% for speakers’ bureaus.
“We were encouraged to see that the prevalence of potentially beneficial relationships—such as industry funding to support research studies—was essentially unchanged, indicating IRBs have not tried to eliminate members’ industry relationships across the board,” said Christine Vogeli, PhD, also of the Mongan Institute.
Survey respondents were more likely in the current study than in the previous one to say that their IRB had a written definition of conflicts of interest—63% compared with 46%. In addition, the percentage of respondents who reported their IRB had no policy related to conflicts dropped from 14% to 5%.
However, the researchers were concerned by the fact that 32% of respondents still did not know whether or not their IRB had a policy on conflicts, even though that number had dropped from 41% in the previous study.
The percentage of respondents who handled their conflicts of interest in an appropriate manner increased. Eighty percent of respondents with conflicts reported them to the IRB, up from 55% in the previous study.
And 68% of respondents said they always left the room when a protocol with which they had a conflict was being discussed, up from 38% in the previous study.
However, a quarter of respondents with conflicts indicated they had voted on protocols with which they had a conflict. While that was a drop from what was reported in the previous study, it was not statistically significant.
The percentage of respondents who felt that at least one protocol had been presented to their IRB in a biased fashion because of another member’s industry relationships dropped from 14% in the previous study to 8% in this study.
And when asked about the types of bias they perceived in the presentation—questions not included in the previous survey—8% of respondents reported a pro-industry bias, and 14% reported an anti-industry bias.
“The fact that we found any bias—either pro- or anti-industry—is an issue, since bias is antithetical to research and should be eliminated,” Dr Campbell said. “IRBs should address that issue, along with increasing efforts to educate their members about what constitutes a conflict of interest and the inappropriateness of voting on protocols with which they have a conflict.” ![]()

Institutional Review Boards (IRBs) at academic health centers have gotten better at managing members’ industry relationships in the last several years, but issues persist, according to a new study.
The research is a follow-up to a study published in 2006.
The new study showed improvements in the management of industry relationships, with increased levels of disclosure and fewer “problematic relationships” than were reported in the previous study.
However, nearly a third of IRB members in the new study said they were unaware of their board’s conflict of interest policies. And a quarter of IRB members reported voting on protocols with which they have conflicts of interest, which is a violation of federal regulations.
“IRBs are the primary mechanisms by which institutions oversee research that involves human participants, and industry relationships of IRB members have the potential to impact their IRB-related activities in both positive and negative ways,” said Eric G. Campbell, PhD, of the Mongan Institute for Health Policy at Massachusetts General Hospital in Boston.
“While our principal findings are that, compared to a decade ago, IRBs appear to be doing a much better job at managing their members’ industry relationships, there still are findings that are concerning.”
Dr Campbell and his colleagues described these findings in JAMA Internal Medicine.
Every US institution that conducts research involving human participants must have an IRB, which reviews proposed studies to ensure their design is scientifically valid and does not violate ethical and regulatory guidelines. IRBs also monitor ongoing studies to make sure they maintain appropriate practices.
The first study to examine industry relationships of IRB members, conducted by Dr Campbell and his colleagues, was based on a survey of IRB members at 100 institutions that was published in NEJM in November 2006.
That study showed that more than a third of IRB members had some sort of financial relationship with commercial firms. And while most respondents did not believe that such relationships had an inappropriate impact on IRB decision making, significant percentages were not aware of institutional guidelines for disclosing industry relationships or defining conflicts of interest.
The current study involved an identical survey administered in 2014 to nearly 500 IRB members at 115 medical schools and teaching hospitals around the country.
As in the 2006 study, respondents were asked about specific types of relationships with commercial companies, whether they received any industry funding and for what purposes, and how many protocols had come before their IRB that involved companies with which they had a relationship.
For protocols with which IRB members had a conflict—relating either to companies they had relationships with or to competitors—they were asked whether they had disclosed their relationships to the IRB, whether they participated in discussion of those protocols, and whether they voted on the protocols.
Results
There was no significant difference between the current study and the previous study in the percentage of IRB members who reported having industry relationships overall—37% and 32%, respectively.
However, the percentage of respondents who reported receiving payments for attending meetings and conferences or for serving on speakers’ bureaus—relationships that are considered problematic—dropped significantly. The percentages fell from 16% to 9% for meeting/conference participation and from 14% to 4% for speakers’ bureaus.
“We were encouraged to see that the prevalence of potentially beneficial relationships—such as industry funding to support research studies—was essentially unchanged, indicating IRBs have not tried to eliminate members’ industry relationships across the board,” said Christine Vogeli, PhD, also of the Mongan Institute.
Survey respondents were more likely in the current study than in the previous one to say that their IRB had a written definition of conflicts of interest—63% compared with 46%. In addition, the percentage of respondents who reported their IRB had no policy related to conflicts dropped from 14% to 5%.
However, the researchers were concerned by the fact that 32% of respondents still did not know whether or not their IRB had a policy on conflicts, even though that number had dropped from 41% in the previous study.
The percentage of respondents who handled their conflicts of interest in an appropriate manner increased. Eighty percent of respondents with conflicts reported them to the IRB, up from 55% in the previous study.
And 68% of respondents said they always left the room when a protocol with which they had a conflict was being discussed, up from 38% in the previous study.
However, a quarter of respondents with conflicts indicated they had voted on protocols with which they had a conflict. While that was a drop from what was reported in the previous study, it was not statistically significant.
The percentage of respondents who felt that at least one protocol had been presented to their IRB in a biased fashion because of another member’s industry relationships dropped from 14% in the previous study to 8% in this study.
And when asked about the types of bias they perceived in the presentation—questions not included in the previous survey—8% of respondents reported a pro-industry bias, and 14% reported an anti-industry bias.
“The fact that we found any bias—either pro- or anti-industry—is an issue, since bias is antithetical to research and should be eliminated,” Dr Campbell said. “IRBs should address that issue, along with increasing efforts to educate their members about what constitutes a conflict of interest and the inappropriateness of voting on protocols with which they have a conflict.” ![]()
Prolonged sitting may increase cancer risk in women

Photo by Petr Kratochvil
Results of a large study indicate that women who spend their leisure time sitting for prolonged periods—6 or more hours a day—have an increased risk of developing certain cancers.
These women had a significant increase in the risk of overall cancer, multiple myeloma, breast cancer, and ovarian cancer.
The increased risks were present even after taking into account a woman’s body mass index (BMI), frequency of physical activity, and other factors.
For men, there was no association between leisure time spent sitting and any type of cancer.
Alpa Patel, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues conducted this research and reported the results in Cancer Epidemiology, Biomarkers & Prevention.
The researchers analyzed data on 69,260 men and 77,462 women who were initially cancer-free and enrolled in the American Cancer Society Cancer Prevention Study II Nutrition Cohort.
In addition to evaluating the amount of leisure time subjects spent sitting, the researchers assessed factors such as age, race, BMI, alcohol use, smoking status, diabetes status, diet, and frequency and type of physical activity.
At baseline, the men enrolled in this study were slightly older, leaner, and more likely to have ever smoked cigarettes compared to the women. Men and women who spent the most leisure time sitting were more likely than their peers to be obese, have type II diabetes mellitus or other chronic diseases, have a higher caloric intake, eat more red/processed meat, and have ever smoked.
Between 1992 and 2009, 18,555 men and 12,236 women were diagnosed with cancer.
In women, longer leisure time spent sitting was associated with a significantly increased risk of cancer overall, even after the researchers adjusted for potential confounders such as overall physical activity and BMI.
The relative risk (RR) of cancer was 1.10 for women who spent an average of 6 or more hours a day sitting, compared to those who spent less than 3 hours a day sitting.
Women who sat for 6 or more hours a day also had a significantly increased risk of multiple myeloma (RR=1.65), invasive breast cancer (RR=1.10), and ovarian cancer (RR=1.43).
Initially, there was a significant association between prolonged sitting and endometrial cancer, but this was attenuated after the researchers adjusted for BMI. The researchers also found positive, but not significant, associations between sitting time and esophageal, head and neck, and gallbladder cancers.
For men, there was no significant association between time spent sitting and overall cancer risk or the risk of individual cancers. The researchers said further study is warranted to better understand this difference between the sexes.
The team also noted that American Cancer Society guidelines for cancer prevention recommend reducing sitting time when possible. And, given the high rate of time spent sitting in the US, even a modest positive association with cancer could have broad public health implications. ![]()

Photo by Petr Kratochvil
Results of a large study indicate that women who spend their leisure time sitting for prolonged periods—6 or more hours a day—have an increased risk of developing certain cancers.
These women had a significant increase in the risk of overall cancer, multiple myeloma, breast cancer, and ovarian cancer.
The increased risks were present even after taking into account a woman’s body mass index (BMI), frequency of physical activity, and other factors.
For men, there was no association between leisure time spent sitting and any type of cancer.
Alpa Patel, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues conducted this research and reported the results in Cancer Epidemiology, Biomarkers & Prevention.
The researchers analyzed data on 69,260 men and 77,462 women who were initially cancer-free and enrolled in the American Cancer Society Cancer Prevention Study II Nutrition Cohort.
In addition to evaluating the amount of leisure time subjects spent sitting, the researchers assessed factors such as age, race, BMI, alcohol use, smoking status, diabetes status, diet, and frequency and type of physical activity.
At baseline, the men enrolled in this study were slightly older, leaner, and more likely to have ever smoked cigarettes compared to the women. Men and women who spent the most leisure time sitting were more likely than their peers to be obese, have type II diabetes mellitus or other chronic diseases, have a higher caloric intake, eat more red/processed meat, and have ever smoked.
Between 1992 and 2009, 18,555 men and 12,236 women were diagnosed with cancer.
In women, longer leisure time spent sitting was associated with a significantly increased risk of cancer overall, even after the researchers adjusted for potential confounders such as overall physical activity and BMI.
The relative risk (RR) of cancer was 1.10 for women who spent an average of 6 or more hours a day sitting, compared to those who spent less than 3 hours a day sitting.
Women who sat for 6 or more hours a day also had a significantly increased risk of multiple myeloma (RR=1.65), invasive breast cancer (RR=1.10), and ovarian cancer (RR=1.43).
Initially, there was a significant association between prolonged sitting and endometrial cancer, but this was attenuated after the researchers adjusted for BMI. The researchers also found positive, but not significant, associations between sitting time and esophageal, head and neck, and gallbladder cancers.
For men, there was no significant association between time spent sitting and overall cancer risk or the risk of individual cancers. The researchers said further study is warranted to better understand this difference between the sexes.
The team also noted that American Cancer Society guidelines for cancer prevention recommend reducing sitting time when possible. And, given the high rate of time spent sitting in the US, even a modest positive association with cancer could have broad public health implications. ![]()

Photo by Petr Kratochvil
Results of a large study indicate that women who spend their leisure time sitting for prolonged periods—6 or more hours a day—have an increased risk of developing certain cancers.
These women had a significant increase in the risk of overall cancer, multiple myeloma, breast cancer, and ovarian cancer.
The increased risks were present even after taking into account a woman’s body mass index (BMI), frequency of physical activity, and other factors.
For men, there was no association between leisure time spent sitting and any type of cancer.
Alpa Patel, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues conducted this research and reported the results in Cancer Epidemiology, Biomarkers & Prevention.
The researchers analyzed data on 69,260 men and 77,462 women who were initially cancer-free and enrolled in the American Cancer Society Cancer Prevention Study II Nutrition Cohort.
In addition to evaluating the amount of leisure time subjects spent sitting, the researchers assessed factors such as age, race, BMI, alcohol use, smoking status, diabetes status, diet, and frequency and type of physical activity.
At baseline, the men enrolled in this study were slightly older, leaner, and more likely to have ever smoked cigarettes compared to the women. Men and women who spent the most leisure time sitting were more likely than their peers to be obese, have type II diabetes mellitus or other chronic diseases, have a higher caloric intake, eat more red/processed meat, and have ever smoked.
Between 1992 and 2009, 18,555 men and 12,236 women were diagnosed with cancer.
In women, longer leisure time spent sitting was associated with a significantly increased risk of cancer overall, even after the researchers adjusted for potential confounders such as overall physical activity and BMI.
The relative risk (RR) of cancer was 1.10 for women who spent an average of 6 or more hours a day sitting, compared to those who spent less than 3 hours a day sitting.
Women who sat for 6 or more hours a day also had a significantly increased risk of multiple myeloma (RR=1.65), invasive breast cancer (RR=1.10), and ovarian cancer (RR=1.43).
Initially, there was a significant association between prolonged sitting and endometrial cancer, but this was attenuated after the researchers adjusted for BMI. The researchers also found positive, but not significant, associations between sitting time and esophageal, head and neck, and gallbladder cancers.
For men, there was no significant association between time spent sitting and overall cancer risk or the risk of individual cancers. The researchers said further study is warranted to better understand this difference between the sexes.
The team also noted that American Cancer Society guidelines for cancer prevention recommend reducing sitting time when possible. And, given the high rate of time spent sitting in the US, even a modest positive association with cancer could have broad public health implications.
NICE recommends edoxaban for VTE

Image by Keven MacKenzie
The UK’s National Institute for Health and Care Excellence (NICE) has issued a draft guidance recommending the oral anticoagulant edoxaban tosylate (Lixiana) as an option for treating and preventing the recurrence of venous thromboembolism (VTE).
A committee advising NICE concluded that edoxaban is clinically effective, and the drug will be a cost-effective use of National Health Service resources.
“The newer oral anticoagulants like edoxaban tosylate are an alternative to warfarin as the mainstay of treatment to prevent recurrent blood clots,” said Carole Longson, NICE Health Technology Evaluation Centre Director.
“The committee concluded that patients value newer oral anticoagulants such as edoxaban tosylate, which cause less disruption to their day-to-day lives than warfarin. We are pleased, therefore, to be able to recommend edoxaban tosylate as a further cost-effective option for treating [VTE] and preventing further episodes in adults.”
Clinical effectiveness
Edoxaban was recently approved in the European Union to treat and prevent deep vein thrombosis (DVT) and pulmonary embolism (PE). This approval was based on results of the Hokusai-VTE trial. The committee advising NICE analyzed data from this trial when considering the clinical effectiveness of edoxaban.
For Hokusai-VTE, researchers evaluated edoxaban in 4921 patients with DVT and 3319 with PE. Patients received initial treatment with low-molecular-weight heparin and were then randomized to receive edoxaban or warfarin daily for 3 to 12 months.
Overall, edoxaban proved as effective as warfarin. Recurrent, symptomatic VTE occurred in 3.2% and 3.5% of patients, respectively (P<0.001 for non-inferiority).
In addition, the incidence of clinically relevant bleeding was significantly lower in the edoxaban arm than the warfarin arm—8.5% and 10.3%, respectively (P=0.004 for superiority).
Cost-effectiveness
The recommended dose of edoxaban is 60 mg once daily, or 30 mg once daily in specific patient groups—those with renal impairment, low body weight (60 kg or less), or concomitant use of potent permeability glycoprotein inhibitors—following treatment with a parenteral anticoagulant for at least 5 days.
Edoxaban costs £2.10 per 15 mg, 30 mg, or 60 mg tablet (excluding value-added tax). Costs may vary in different settings because of negotiated procurement discounts.
Taking into account the lack of any clear evidence that edoxaban is significantly different from the other new oral anticoagulants, as well as the testimony of experts, the committee advising NICE concluded that the most plausible incremental cost-effectiveness ratio for edoxaban was likely to be in line with the other oral anticoagulants already recommended by NICE.
The draft guidance for edoxaban is now with consultees, who have the opportunity to appeal against it. Once NICE issues its final guidance on a technology, it replaces local recommendations.

Image by Keven MacKenzie
The UK’s National Institute for Health and Care Excellence (NICE) has issued a draft guidance recommending the oral anticoagulant edoxaban tosylate (Lixiana) as an option for treating and preventing the recurrence of venous thromboembolism (VTE).
A committee advising NICE concluded that edoxaban is clinically effective, and the drug will be a cost-effective use of National Health Service resources.
“The newer oral anticoagulants like edoxaban tosylate are an alternative to warfarin as the mainstay of treatment to prevent recurrent blood clots,” said Carole Longson, NICE Health Technology Evaluation Centre Director.
“The committee concluded that patients value newer oral anticoagulants such as edoxaban tosylate, which cause less disruption to their day-to-day lives than warfarin. We are pleased, therefore, to be able to recommend edoxaban tosylate as a further cost-effective option for treating [VTE] and preventing further episodes in adults.”
Clinical effectiveness
Edoxaban was recently approved in the European Union to treat and prevent deep vein thrombosis (DVT) and pulmonary embolism (PE). This approval was based on results of the Hokusai-VTE trial. The committee advising NICE analyzed data from this trial when considering the clinical effectiveness of edoxaban.
For Hokusai-VTE, researchers evaluated edoxaban in 4921 patients with DVT and 3319 with PE. Patients received initial treatment with low-molecular-weight heparin and were then randomized to receive edoxaban or warfarin daily for 3 to 12 months.
Overall, edoxaban proved as effective as warfarin. Recurrent, symptomatic VTE occurred in 3.2% and 3.5% of patients, respectively (P<0.001 for non-inferiority).
In addition, the incidence of clinically relevant bleeding was significantly lower in the edoxaban arm than the warfarin arm—8.5% and 10.3%, respectively (P=0.004 for superiority).
Cost-effectiveness
The recommended dose of edoxaban is 60 mg once daily, or 30 mg once daily in specific patient groups—those with renal impairment, low body weight (60 kg or less), or concomitant use of potent permeability glycoprotein inhibitors—following treatment with a parenteral anticoagulant for at least 5 days.
Edoxaban costs £2.10 per 15 mg, 30 mg, or 60 mg tablet (excluding value-added tax). Costs may vary in different settings because of negotiated procurement discounts.
Taking into account the lack of any clear evidence that edoxaban is significantly different from the other new oral anticoagulants, as well as the testimony of experts, the committee advising NICE concluded that the most plausible incremental cost-effectiveness ratio for edoxaban was likely to be in line with the other oral anticoagulants already recommended by NICE.
The draft guidance for edoxaban is now with consultees, who have the opportunity to appeal against it. Once NICE issues its final guidance on a technology, it replaces local recommendations.

Image by Keven MacKenzie
The UK’s National Institute for Health and Care Excellence (NICE) has issued a draft guidance recommending the oral anticoagulant edoxaban tosylate (Lixiana) as an option for treating and preventing the recurrence of venous thromboembolism (VTE).
A committee advising NICE concluded that edoxaban is clinically effective, and the drug will be a cost-effective use of National Health Service resources.
“The newer oral anticoagulants like edoxaban tosylate are an alternative to warfarin as the mainstay of treatment to prevent recurrent blood clots,” said Carole Longson, NICE Health Technology Evaluation Centre Director.
“The committee concluded that patients value newer oral anticoagulants such as edoxaban tosylate, which cause less disruption to their day-to-day lives than warfarin. We are pleased, therefore, to be able to recommend edoxaban tosylate as a further cost-effective option for treating [VTE] and preventing further episodes in adults.”
Clinical effectiveness
Edoxaban was recently approved in the European Union to treat and prevent deep vein thrombosis (DVT) and pulmonary embolism (PE). This approval was based on results of the Hokusai-VTE trial. The committee advising NICE analyzed data from this trial when considering the clinical effectiveness of edoxaban.
For Hokusai-VTE, researchers evaluated edoxaban in 4921 patients with DVT and 3319 with PE. Patients received initial treatment with low-molecular-weight heparin and were then randomized to receive edoxaban or warfarin daily for 3 to 12 months.
Overall, edoxaban proved as effective as warfarin. Recurrent, symptomatic VTE occurred in 3.2% and 3.5% of patients, respectively (P<0.001 for non-inferiority).
In addition, the incidence of clinically relevant bleeding was significantly lower in the edoxaban arm than the warfarin arm—8.5% and 10.3%, respectively (P=0.004 for superiority).
Cost-effectiveness
The recommended dose of edoxaban is 60 mg once daily, or 30 mg once daily in specific patient groups—those with renal impairment, low body weight (60 kg or less), or concomitant use of potent permeability glycoprotein inhibitors—following treatment with a parenteral anticoagulant for at least 5 days.
Edoxaban costs £2.10 per 15 mg, 30 mg, or 60 mg tablet (excluding value-added tax). Costs may vary in different settings because of negotiated procurement discounts.
Taking into account the lack of any clear evidence that edoxaban is significantly different from the other new oral anticoagulants, as well as the testimony of experts, the committee advising NICE concluded that the most plausible incremental cost-effectiveness ratio for edoxaban was likely to be in line with the other oral anticoagulants already recommended by NICE.
The draft guidance for edoxaban is now with consultees, who have the opportunity to appeal against it. Once NICE issues its final guidance on a technology, it replaces local recommendations.
Prenatal test results linked to cancer in mothers

Photo by Nina Matthews
Researchers have again found evidence to suggest that tests used to identify chromosomal fetal disorders can detect occult malignancies in pregnant women.
In a study made public last month, non-invasive prenatal tests (NIPTs) revealed 2 cases of lymphoma and a case of ovarian cancer in expectant mothers.
In the new study, researchers showed that positive NIPT results were due to leukemia, lymphoma, or solid tumors in 10 expectant mothers.
The research was published in JAMA and presented at the 19th International Conference on Prenatal Diagnosis and Therapy in Washington, DC. Funding for the study was provided by Illumina, and company employees were involved in the research.
“We did this study because noninvasive prenatal testing using sequencing of cell-free DNA in the mother’s plasma is the fastest-growing area of prenatal testing and, indeed, of genomic medicine,” said study author Diana W. Bianchi, MD, of Tufts Medical Center in Boston, Massachusetts.
“As the volume of tests has expanded, we’ve become increasingly aware of the so-called “false-positive” cases. [A]pproximately 0.2% of the time, there is a discrepancy between the results of the prenatal test—in which an aneuploidy is reported—and the result from the diagnostic fetal procedure, the amniocentesis or the chorionic villus sampling.”
“So we’re interested in the situation where the fetal chromosomes are normal, but the prenatal test shows that there’s an aneuploidy detected. We’re interested in the possible explanations for that discrepancy.”
To gain some insight, Dr Bianchi and her colleagues evaluated 125,426 samples from asymptomatic pregnant women who underwent plasma cell-free DNA sequencing for clinical prenatal aneuploidy screening using Illumina’s verifi Prenatal Test.
In all, 3757 samples (3%) were positive for 1 or more aneuploidies involving chromosomes 13, 18, 21, X, or Y. These were reported to the ordering physician with recommendations for further evaluation.
“In a small minority of women, [subsequent tests analyzing only fetal DNA] showed that the fetal chromosomes were normal, and that disagreed with [results of the NIPT],” Dr Bianchi said. “We were examining whether cancer could explain the discrepancy between these two test results.”
The researchers found that 10 of the women with discordant test results were subsequently diagnosed with cancer. There were 3 cases of B-cell lymphoma and 1 case each of T-cell leukemia, Hodgkin lymphoma, unspecified adenocarcinoma, leiomyosarcoma, and neuroendocrine, colorectal, and anal carcinomas.
Dr Bianchi and her colleagues were able to obtain detailed clinical and sequencing data for 8 of these cases. In the other 2 cases (leiomyosarcoma and unspecified adenocarcinoma), the women were critically ill and were not approached about participating in the study.
The researchers found that maternal cancers most frequently occurred when the NIPT detected more than 1 aneuploidy. There were 7 known cancers among 39 cases of multiple aneuploidies by NIPT. In 1 case, blood was sampled after the patient completed treatment for colorectal cancer, and the abnormal pattern was no longer evident.
When the researchers examined additional genetic information for the women with cancer, they found unique patterns of nonspecific copy-number gains and losses across multiple chromosomes.
“[These women] had DNA imbalances all across the genome,” Dr Bianchi said. “The [NIPT] normally is only looking at DNA material from the chromosomes of clinical interest—chromosomes 13, 18, 21, X, and Y.”
“When we opened up their results to look at all of the chromosomes, there were multiple abnormalities in other places, such as chromosome 8, chromosome 6, etc. Each woman had a unique pattern that was abnormal in many places. This suggested that it was the tumor DNA that was being shed into her blood and was contributing to the abnormal pattern.”
Dr Bianchi stressed that the tumor DNA did not affect the babies. She said all were born healthy, although labor was induced early in one mother to facilitate her cancer treatment.

Photo by Nina Matthews
Researchers have again found evidence to suggest that tests used to identify chromosomal fetal disorders can detect occult malignancies in pregnant women.
In a study made public last month, non-invasive prenatal tests (NIPTs) revealed 2 cases of lymphoma and a case of ovarian cancer in expectant mothers.
In the new study, researchers showed that positive NIPT results were due to leukemia, lymphoma, or solid tumors in 10 expectant mothers.
The research was published in JAMA and presented at the 19th International Conference on Prenatal Diagnosis and Therapy in Washington, DC. Funding for the study was provided by Illumina, and company employees were involved in the research.
“We did this study because noninvasive prenatal testing using sequencing of cell-free DNA in the mother’s plasma is the fastest-growing area of prenatal testing and, indeed, of genomic medicine,” said study author Diana W. Bianchi, MD, of Tufts Medical Center in Boston, Massachusetts.
“As the volume of tests has expanded, we’ve become increasingly aware of the so-called “false-positive” cases. [A]pproximately 0.2% of the time, there is a discrepancy between the results of the prenatal test—in which an aneuploidy is reported—and the result from the diagnostic fetal procedure, the amniocentesis or the chorionic villus sampling.”
“So we’re interested in the situation where the fetal chromosomes are normal, but the prenatal test shows that there’s an aneuploidy detected. We’re interested in the possible explanations for that discrepancy.”
To gain some insight, Dr Bianchi and her colleagues evaluated 125,426 samples from asymptomatic pregnant women who underwent plasma cell-free DNA sequencing for clinical prenatal aneuploidy screening using Illumina’s verifi Prenatal Test.
In all, 3757 samples (3%) were positive for 1 or more aneuploidies involving chromosomes 13, 18, 21, X, or Y. These were reported to the ordering physician with recommendations for further evaluation.
“In a small minority of women, [subsequent tests analyzing only fetal DNA] showed that the fetal chromosomes were normal, and that disagreed with [results of the NIPT],” Dr Bianchi said. “We were examining whether cancer could explain the discrepancy between these two test results.”
The researchers found that 10 of the women with discordant test results were subsequently diagnosed with cancer. There were 3 cases of B-cell lymphoma and 1 case each of T-cell leukemia, Hodgkin lymphoma, unspecified adenocarcinoma, leiomyosarcoma, and neuroendocrine, colorectal, and anal carcinomas.
Dr Bianchi and her colleagues were able to obtain detailed clinical and sequencing data for 8 of these cases. In the other 2 cases (leiomyosarcoma and unspecified adenocarcinoma), the women were critically ill and were not approached about participating in the study.
The researchers found that maternal cancers most frequently occurred when the NIPT detected more than 1 aneuploidy. There were 7 known cancers among 39 cases of multiple aneuploidies by NIPT. In 1 case, blood was sampled after the patient completed treatment for colorectal cancer, and the abnormal pattern was no longer evident.
When the researchers examined additional genetic information for the women with cancer, they found unique patterns of nonspecific copy-number gains and losses across multiple chromosomes.
“[These women] had DNA imbalances all across the genome,” Dr Bianchi said. “The [NIPT] normally is only looking at DNA material from the chromosomes of clinical interest—chromosomes 13, 18, 21, X, and Y.”
“When we opened up their results to look at all of the chromosomes, there were multiple abnormalities in other places, such as chromosome 8, chromosome 6, etc. Each woman had a unique pattern that was abnormal in many places. This suggested that it was the tumor DNA that was being shed into her blood and was contributing to the abnormal pattern.”
Dr Bianchi stressed that the tumor DNA did not affect the babies. She said all were born healthy, although labor was induced early in one mother to facilitate her cancer treatment.

Photo by Nina Matthews
Researchers have again found evidence to suggest that tests used to identify chromosomal fetal disorders can detect occult malignancies in pregnant women.
In a study made public last month, non-invasive prenatal tests (NIPTs) revealed 2 cases of lymphoma and a case of ovarian cancer in expectant mothers.
In the new study, researchers showed that positive NIPT results were due to leukemia, lymphoma, or solid tumors in 10 expectant mothers.
The research was published in JAMA and presented at the 19th International Conference on Prenatal Diagnosis and Therapy in Washington, DC. Funding for the study was provided by Illumina, and company employees were involved in the research.
“We did this study because noninvasive prenatal testing using sequencing of cell-free DNA in the mother’s plasma is the fastest-growing area of prenatal testing and, indeed, of genomic medicine,” said study author Diana W. Bianchi, MD, of Tufts Medical Center in Boston, Massachusetts.
“As the volume of tests has expanded, we’ve become increasingly aware of the so-called “false-positive” cases. [A]pproximately 0.2% of the time, there is a discrepancy between the results of the prenatal test—in which an aneuploidy is reported—and the result from the diagnostic fetal procedure, the amniocentesis or the chorionic villus sampling.”
“So we’re interested in the situation where the fetal chromosomes are normal, but the prenatal test shows that there’s an aneuploidy detected. We’re interested in the possible explanations for that discrepancy.”
To gain some insight, Dr Bianchi and her colleagues evaluated 125,426 samples from asymptomatic pregnant women who underwent plasma cell-free DNA sequencing for clinical prenatal aneuploidy screening using Illumina’s verifi Prenatal Test.
In all, 3757 samples (3%) were positive for 1 or more aneuploidies involving chromosomes 13, 18, 21, X, or Y. These were reported to the ordering physician with recommendations for further evaluation.
“In a small minority of women, [subsequent tests analyzing only fetal DNA] showed that the fetal chromosomes were normal, and that disagreed with [results of the NIPT],” Dr Bianchi said. “We were examining whether cancer could explain the discrepancy between these two test results.”
The researchers found that 10 of the women with discordant test results were subsequently diagnosed with cancer. There were 3 cases of B-cell lymphoma and 1 case each of T-cell leukemia, Hodgkin lymphoma, unspecified adenocarcinoma, leiomyosarcoma, and neuroendocrine, colorectal, and anal carcinomas.
Dr Bianchi and her colleagues were able to obtain detailed clinical and sequencing data for 8 of these cases. In the other 2 cases (leiomyosarcoma and unspecified adenocarcinoma), the women were critically ill and were not approached about participating in the study.
The researchers found that maternal cancers most frequently occurred when the NIPT detected more than 1 aneuploidy. There were 7 known cancers among 39 cases of multiple aneuploidies by NIPT. In 1 case, blood was sampled after the patient completed treatment for colorectal cancer, and the abnormal pattern was no longer evident.
When the researchers examined additional genetic information for the women with cancer, they found unique patterns of nonspecific copy-number gains and losses across multiple chromosomes.
“[These women] had DNA imbalances all across the genome,” Dr Bianchi said. “The [NIPT] normally is only looking at DNA material from the chromosomes of clinical interest—chromosomes 13, 18, 21, X, and Y.”
“When we opened up their results to look at all of the chromosomes, there were multiple abnormalities in other places, such as chromosome 8, chromosome 6, etc. Each woman had a unique pattern that was abnormal in many places. This suggested that it was the tumor DNA that was being shed into her blood and was contributing to the abnormal pattern.”
Dr Bianchi stressed that the tumor DNA did not affect the babies. She said all were born healthy, although labor was induced early in one mother to facilitate her cancer treatment.
YA cancer survivors hospitalized more often
Photo courtesy of
St. Michael’s Hospital
Up to 20 years after they are declared cancer-free, young adult (YA) cancer survivors are still hospitalized more often than the general population, according to research published in the Journal of Clinical Oncology.
Overall, the cancer survivors, who were ages 20 to 44 at diagnosis, were hospitalized about 1.5 times as often as control subjects.
“Even when young adults survive cancer, the cancer still has an impact on their lives and their long-term health, and this age group still has a lot of life to live,” said study author Nancy Baxter, MD, PhD, of St. Michael’s Hospital in Toronto, Ontario, Canada.
To conduct this study, Dr Baxter and her colleagues examined data from the Ontario Cancer Registry spanning the period from 1992 to 1999.
This included 20,275 patients who had their first cancer diagnosis between the ages of 20 and 44 and had lived for 5 years cancer-free. The researchers compared hospitalizations among these patients to hospitalizations in 101,344 non-cancer controls.
During the study period, 34.3% of cancer survivors (n=6948) were admitted to the hospital. The adjusted relative rate (ARR) of hospitalizations in survivors compared to controls was 1.51.
There was a significant decrease in hospitalizations among cancer survivors from the first time point the researchers analyzed to the last time point (P<0.0001).
But hospitalizations were more common among cancer survivors regardless of the time point. The ARR was 1.67 at 5 to 8 years after cancer diagnosis and 1.22 at 18 to 20 years after diagnosis.
When the researchers looked at individual malignancies, they found that survivors of melanoma or testicular cancer did not have higher rates of hospitalization than the control population. The ARRs were 0.97 and 1.07, respectively.
However, the rate of hospitalization was at least twice as high as the control population for survivors of leukemia (ARR=2.23) and lymphoma (ARR=2.02), as well as gastrointestinal (ARR=2.49), urologic (ARR=2.20), colorectal (ARR=2.10), and brain cancers (ARR=2.04).
Dr Baxter said having a better understanding of healthcare utilization and late effects in the YA cancer population may help healthcare providers counsel YA survivors on their future quality of life, identify areas where preventative strategies could be employed, and highlight the need to consider treatments that are not associated with long-term health consequences.

Photo courtesy of
St. Michael’s Hospital
Up to 20 years after they are declared cancer-free, young adult (YA) cancer survivors are still hospitalized more often than the general population, according to research published in the Journal of Clinical Oncology.
Overall, the cancer survivors, who were ages 20 to 44 at diagnosis, were hospitalized about 1.5 times as often as control subjects.
“Even when young adults survive cancer, the cancer still has an impact on their lives and their long-term health, and this age group still has a lot of life to live,” said study author Nancy Baxter, MD, PhD, of St. Michael’s Hospital in Toronto, Ontario, Canada.
To conduct this study, Dr Baxter and her colleagues examined data from the Ontario Cancer Registry spanning the period from 1992 to 1999.
This included 20,275 patients who had their first cancer diagnosis between the ages of 20 and 44 and had lived for 5 years cancer-free. The researchers compared hospitalizations among these patients to hospitalizations in 101,344 non-cancer controls.
During the study period, 34.3% of cancer survivors (n=6948) were admitted to the hospital. The adjusted relative rate (ARR) of hospitalizations in survivors compared to controls was 1.51.
There was a significant decrease in hospitalizations among cancer survivors from the first time point the researchers analyzed to the last time point (P<0.0001).
But hospitalizations were more common among cancer survivors regardless of the time point. The ARR was 1.67 at 5 to 8 years after cancer diagnosis and 1.22 at 18 to 20 years after diagnosis.
When the researchers looked at individual malignancies, they found that survivors of melanoma or testicular cancer did not have higher rates of hospitalization than the control population. The ARRs were 0.97 and 1.07, respectively.
However, the rate of hospitalization was at least twice as high as the control population for survivors of leukemia (ARR=2.23) and lymphoma (ARR=2.02), as well as gastrointestinal (ARR=2.49), urologic (ARR=2.20), colorectal (ARR=2.10), and brain cancers (ARR=2.04).
Dr Baxter said having a better understanding of healthcare utilization and late effects in the YA cancer population may help healthcare providers counsel YA survivors on their future quality of life, identify areas where preventative strategies could be employed, and highlight the need to consider treatments that are not associated with long-term health consequences.

Photo courtesy of
St. Michael’s Hospital
Up to 20 years after they are declared cancer-free, young adult (YA) cancer survivors are still hospitalized more often than the general population, according to research published in the Journal of Clinical Oncology.
Overall, the cancer survivors, who were ages 20 to 44 at diagnosis, were hospitalized about 1.5 times as often as control subjects.
“Even when young adults survive cancer, the cancer still has an impact on their lives and their long-term health, and this age group still has a lot of life to live,” said study author Nancy Baxter, MD, PhD, of St. Michael’s Hospital in Toronto, Ontario, Canada.
To conduct this study, Dr Baxter and her colleagues examined data from the Ontario Cancer Registry spanning the period from 1992 to 1999.
This included 20,275 patients who had their first cancer diagnosis between the ages of 20 and 44 and had lived for 5 years cancer-free. The researchers compared hospitalizations among these patients to hospitalizations in 101,344 non-cancer controls.
During the study period, 34.3% of cancer survivors (n=6948) were admitted to the hospital. The adjusted relative rate (ARR) of hospitalizations in survivors compared to controls was 1.51.
There was a significant decrease in hospitalizations among cancer survivors from the first time point the researchers analyzed to the last time point (P<0.0001).
But hospitalizations were more common among cancer survivors regardless of the time point. The ARR was 1.67 at 5 to 8 years after cancer diagnosis and 1.22 at 18 to 20 years after diagnosis.
When the researchers looked at individual malignancies, they found that survivors of melanoma or testicular cancer did not have higher rates of hospitalization than the control population. The ARRs were 0.97 and 1.07, respectively.
However, the rate of hospitalization was at least twice as high as the control population for survivors of leukemia (ARR=2.23) and lymphoma (ARR=2.02), as well as gastrointestinal (ARR=2.49), urologic (ARR=2.20), colorectal (ARR=2.10), and brain cancers (ARR=2.04).
Dr Baxter said having a better understanding of healthcare utilization and late effects in the YA cancer population may help healthcare providers counsel YA survivors on their future quality of life, identify areas where preventative strategies could be employed, and highlight the need to consider treatments that are not associated with long-term health consequences.