User login
M. Alexander Otto began his reporting career early in 1999 covering the pharmaceutical industry for a national pharmacists' magazine and freelancing for the Washington Post and other newspapers. He then joined BNA, now part of Bloomberg News, covering health law and the protection of people and animals in medical research. Alex next worked for the McClatchy Company. Based on his work, Alex won a year-long Knight Science Journalism Fellowship to MIT in 2008-2009. He joined the company shortly thereafter. Alex has a newspaper journalism degree from Syracuse (N.Y.) University and a master's degree in medical science -- a physician assistant degree -- from George Washington University. Alex is based in Seattle.
Diffuse Nature of MRSA Abscesses Contribute to High Treatment Failures
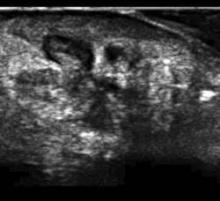
PHOENIX — Methicillin-resistant Staphylococcus aureus abscesses, when compared by ultrasound with those caused by other pathogens, are smaller and more likely to lack a defined edge.
They are also more likely to have edema in surrounding tissue planes as well as pus divided into multiple pockets within the abscess, according to an abscess ultrasound study presented at the Society for Academic Emergency Medicine’s annual meeting.
The characteristics could make it more likely that an abscess is caused by methicillin-resistant Staphylococcus aureus (MRSA), helping guide antibiotic selection pending culture and sensitivity reports, said the study’s presenter, Dr. Dana Resop, an emergency department physician at the University of Massachusetts, Worcester.
The findings also indicate that additional factors—in addition to thick pus—make MRSA abscesses harder to drain than those caused by other pathogens, according to Dr. Romolo J. Gaspari, also an emergency department physician at the school.
The doctors and their colleagues assessed ultrasound images of 254 abscess, blinded to culture results until after the assessments were complete; 124 were MRSA positive.
They found MRSA abscesses are more than twice as likely as those caused by other pathogens to have no defined edge, to have edema in surrounding tissue planes, and have extensions to the skin surface.
They were also smaller and less likely to have ultrasound edge artifacts, thin black lines extending from the abscess edge away from the ultrasound probe.
Though odds ratios were not significant for shape, 26.7% of MRSA abscesses were lobated or ill defined, compared with 11.3% of non-MRSA abscesses.
The less well-organized nature of MRSA abscesses makes it harder to evacuate purulent matter and could contribute to the higher treatment failure rates found in a second study by the team, presented by Dr. Gaspari.
The second study compared incision and drainage (I&D) to ultrasound-guided, 18-gauge needle aspiration for draining abscesses.
The study included 101 abscesses, 54 treated by I&D, the rest by needle aspiration. All patients were treated with antibiotics. Failure was defined as the need for a second drainage.
The team found that 39% of MRSA abscess failed I&D, compared with 11% of non-MRSA abscesses; 92% of MRSA abscesses failed needle aspiration, compared with 55% of non-MRSA abscesses.
Overall, “aspiration’s just not as good as I&D,” Dr. Gaspari said.
When asked by an audience member if he thought the high MRSA failure rates in the second study were related to the physical characteristics of MRSA abscesses found in the first, Dr. Gaspari said, “I would say yes.”
Dr. Gaspari and Dr. Resop reported that they had no disclosures. The studies had no outside funding.
PHOENIX — Methicillin-resistant Staphylococcus aureus abscesses, when compared by ultrasound with those caused by other pathogens, are smaller and more likely to lack a defined edge.
They are also more likely to have edema in surrounding tissue planes as well as pus divided into multiple pockets within the abscess, according to an abscess ultrasound study presented at the Society for Academic Emergency Medicine’s annual meeting.
The characteristics could make it more likely that an abscess is caused by methicillin-resistant Staphylococcus aureus (MRSA), helping guide antibiotic selection pending culture and sensitivity reports, said the study’s presenter, Dr. Dana Resop, an emergency department physician at the University of Massachusetts, Worcester.
The findings also indicate that additional factors—in addition to thick pus—make MRSA abscesses harder to drain than those caused by other pathogens, according to Dr. Romolo J. Gaspari, also an emergency department physician at the school.
The doctors and their colleagues assessed ultrasound images of 254 abscess, blinded to culture results until after the assessments were complete; 124 were MRSA positive.
They found MRSA abscesses are more than twice as likely as those caused by other pathogens to have no defined edge, to have edema in surrounding tissue planes, and have extensions to the skin surface.
They were also smaller and less likely to have ultrasound edge artifacts, thin black lines extending from the abscess edge away from the ultrasound probe.
Though odds ratios were not significant for shape, 26.7% of MRSA abscesses were lobated or ill defined, compared with 11.3% of non-MRSA abscesses.
The less well-organized nature of MRSA abscesses makes it harder to evacuate purulent matter and could contribute to the higher treatment failure rates found in a second study by the team, presented by Dr. Gaspari.
The second study compared incision and drainage (I&D) to ultrasound-guided, 18-gauge needle aspiration for draining abscesses.
The study included 101 abscesses, 54 treated by I&D, the rest by needle aspiration. All patients were treated with antibiotics. Failure was defined as the need for a second drainage.
The team found that 39% of MRSA abscess failed I&D, compared with 11% of non-MRSA abscesses; 92% of MRSA abscesses failed needle aspiration, compared with 55% of non-MRSA abscesses.
Overall, “aspiration’s just not as good as I&D,” Dr. Gaspari said.
When asked by an audience member if he thought the high MRSA failure rates in the second study were related to the physical characteristics of MRSA abscesses found in the first, Dr. Gaspari said, “I would say yes.”
Dr. Gaspari and Dr. Resop reported that they had no disclosures. The studies had no outside funding.
PHOENIX — Methicillin-resistant Staphylococcus aureus abscesses, when compared by ultrasound with those caused by other pathogens, are smaller and more likely to lack a defined edge.
They are also more likely to have edema in surrounding tissue planes as well as pus divided into multiple pockets within the abscess, according to an abscess ultrasound study presented at the Society for Academic Emergency Medicine’s annual meeting.
The characteristics could make it more likely that an abscess is caused by methicillin-resistant Staphylococcus aureus (MRSA), helping guide antibiotic selection pending culture and sensitivity reports, said the study’s presenter, Dr. Dana Resop, an emergency department physician at the University of Massachusetts, Worcester.
The findings also indicate that additional factors—in addition to thick pus—make MRSA abscesses harder to drain than those caused by other pathogens, according to Dr. Romolo J. Gaspari, also an emergency department physician at the school.
The doctors and their colleagues assessed ultrasound images of 254 abscess, blinded to culture results until after the assessments were complete; 124 were MRSA positive.
They found MRSA abscesses are more than twice as likely as those caused by other pathogens to have no defined edge, to have edema in surrounding tissue planes, and have extensions to the skin surface.
They were also smaller and less likely to have ultrasound edge artifacts, thin black lines extending from the abscess edge away from the ultrasound probe.
Though odds ratios were not significant for shape, 26.7% of MRSA abscesses were lobated or ill defined, compared with 11.3% of non-MRSA abscesses.
The less well-organized nature of MRSA abscesses makes it harder to evacuate purulent matter and could contribute to the higher treatment failure rates found in a second study by the team, presented by Dr. Gaspari.
The second study compared incision and drainage (I&D) to ultrasound-guided, 18-gauge needle aspiration for draining abscesses.
The study included 101 abscesses, 54 treated by I&D, the rest by needle aspiration. All patients were treated with antibiotics. Failure was defined as the need for a second drainage.
The team found that 39% of MRSA abscess failed I&D, compared with 11% of non-MRSA abscesses; 92% of MRSA abscesses failed needle aspiration, compared with 55% of non-MRSA abscesses.
Overall, “aspiration’s just not as good as I&D,” Dr. Gaspari said.
When asked by an audience member if he thought the high MRSA failure rates in the second study were related to the physical characteristics of MRSA abscesses found in the first, Dr. Gaspari said, “I would say yes.”
Dr. Gaspari and Dr. Resop reported that they had no disclosures. The studies had no outside funding.
Resistance Exercise May Preserve Muscle Mass in Arthritis
MARINA DEL REY, CALIF. — Rheumatoid arthritis patients with well-controlled disease may benefit from performing fat-burning exercises accompanied by resistance training—such as weight lifting—to preserve or even build muscle mass, according to Dr. Joan M. Bathon.
Such a regimen has the potential to address some of the elevated risks for cardiovascular disease found in this population.
A seemingly fit patient with well-controlled rheumatoid arthritis (RA) and a normal body mass index may still have excess body fat, elevated C-reactive protein levels, and increased coronary artery disease risk.
There is no easy way to assess body composition in the office. And even when body mass index is considered, “you don't know how healthy” an RA patient is, said Dr. Bathon, who is professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
The chronic inflammation of RA can waste muscles, and RA-associated disability can promote sedentary lifestyles, which in turn leads to excess fat. Sometimes muscle loss is offset by fat gain, leaving a patient with a normal or even low BMI, yet with an unhealthy body composition, Dr. Bathon explained at a rheumatology seminar that was sponsored by the University of California, Los Angeles.
Appendicular fat correlates with disability, and visceral fat correlates with coronary artery disease, the leading killer of patients with RA, she said.
When patients have well-controlled RA, their high C-reactive protein levels might be coming not from the inflamed joints, but rather from fat deposits, and might signal an increased risk of coronary artery disease.
Dr. Bathon reported supportive data from a published study.
She and her colleagues performed anthropomorphic measurements and dual-energy x-ray absorptiometry (DXA) scanning to assess fat:muscle ratio in 72 men and 117 women with RA and moderate disability. A single CT image of the abdomen in the axial plane was used to assess the amount of visceral fat. The subjects were then matched with 189 healthy controls.
Compared with the healthy controls, women with rheumatoid arthritis who had BMIs below 25 kg/m
Abnormal body composition was associated with increases in joint deformity, self-reported disability scores, C-reactive protein levels, rheumatoid factor seropositivity, and a lack of current treatment with disease-modifying antirheumatic drugs (Arthritis Rheum. 2008;59:807–15).
Disclosures: Dr. Bathon said that she had no relevant conflicts.
A seemingly fit patient with well-controlled rheumatoid arthritis and a normal body mass index may still have a number of heart disease risk factors.
Source ©Luc Ubaghs/iStockphoto.com
MARINA DEL REY, CALIF. — Rheumatoid arthritis patients with well-controlled disease may benefit from performing fat-burning exercises accompanied by resistance training—such as weight lifting—to preserve or even build muscle mass, according to Dr. Joan M. Bathon.
Such a regimen has the potential to address some of the elevated risks for cardiovascular disease found in this population.
A seemingly fit patient with well-controlled rheumatoid arthritis (RA) and a normal body mass index may still have excess body fat, elevated C-reactive protein levels, and increased coronary artery disease risk.
There is no easy way to assess body composition in the office. And even when body mass index is considered, “you don't know how healthy” an RA patient is, said Dr. Bathon, who is professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
The chronic inflammation of RA can waste muscles, and RA-associated disability can promote sedentary lifestyles, which in turn leads to excess fat. Sometimes muscle loss is offset by fat gain, leaving a patient with a normal or even low BMI, yet with an unhealthy body composition, Dr. Bathon explained at a rheumatology seminar that was sponsored by the University of California, Los Angeles.
Appendicular fat correlates with disability, and visceral fat correlates with coronary artery disease, the leading killer of patients with RA, she said.
When patients have well-controlled RA, their high C-reactive protein levels might be coming not from the inflamed joints, but rather from fat deposits, and might signal an increased risk of coronary artery disease.
Dr. Bathon reported supportive data from a published study.
She and her colleagues performed anthropomorphic measurements and dual-energy x-ray absorptiometry (DXA) scanning to assess fat:muscle ratio in 72 men and 117 women with RA and moderate disability. A single CT image of the abdomen in the axial plane was used to assess the amount of visceral fat. The subjects were then matched with 189 healthy controls.
Compared with the healthy controls, women with rheumatoid arthritis who had BMIs below 25 kg/m
Abnormal body composition was associated with increases in joint deformity, self-reported disability scores, C-reactive protein levels, rheumatoid factor seropositivity, and a lack of current treatment with disease-modifying antirheumatic drugs (Arthritis Rheum. 2008;59:807–15).
Disclosures: Dr. Bathon said that she had no relevant conflicts.
A seemingly fit patient with well-controlled rheumatoid arthritis and a normal body mass index may still have a number of heart disease risk factors.
Source ©Luc Ubaghs/iStockphoto.com
MARINA DEL REY, CALIF. — Rheumatoid arthritis patients with well-controlled disease may benefit from performing fat-burning exercises accompanied by resistance training—such as weight lifting—to preserve or even build muscle mass, according to Dr. Joan M. Bathon.
Such a regimen has the potential to address some of the elevated risks for cardiovascular disease found in this population.
A seemingly fit patient with well-controlled rheumatoid arthritis (RA) and a normal body mass index may still have excess body fat, elevated C-reactive protein levels, and increased coronary artery disease risk.
There is no easy way to assess body composition in the office. And even when body mass index is considered, “you don't know how healthy” an RA patient is, said Dr. Bathon, who is professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
The chronic inflammation of RA can waste muscles, and RA-associated disability can promote sedentary lifestyles, which in turn leads to excess fat. Sometimes muscle loss is offset by fat gain, leaving a patient with a normal or even low BMI, yet with an unhealthy body composition, Dr. Bathon explained at a rheumatology seminar that was sponsored by the University of California, Los Angeles.
Appendicular fat correlates with disability, and visceral fat correlates with coronary artery disease, the leading killer of patients with RA, she said.
When patients have well-controlled RA, their high C-reactive protein levels might be coming not from the inflamed joints, but rather from fat deposits, and might signal an increased risk of coronary artery disease.
Dr. Bathon reported supportive data from a published study.
She and her colleagues performed anthropomorphic measurements and dual-energy x-ray absorptiometry (DXA) scanning to assess fat:muscle ratio in 72 men and 117 women with RA and moderate disability. A single CT image of the abdomen in the axial plane was used to assess the amount of visceral fat. The subjects were then matched with 189 healthy controls.
Compared with the healthy controls, women with rheumatoid arthritis who had BMIs below 25 kg/m
Abnormal body composition was associated with increases in joint deformity, self-reported disability scores, C-reactive protein levels, rheumatoid factor seropositivity, and a lack of current treatment with disease-modifying antirheumatic drugs (Arthritis Rheum. 2008;59:807–15).
Disclosures: Dr. Bathon said that she had no relevant conflicts.
A seemingly fit patient with well-controlled rheumatoid arthritis and a normal body mass index may still have a number of heart disease risk factors.
Source ©Luc Ubaghs/iStockphoto.com
Febuxostat Tied to Hypersensitivity Reactions : Reports include two cases of Stevens-Johnson syndrome.
The Food and Drug Administration had received 11 reports of hypersensitivity reactions to febuxostat as of last month.
Among the 11 cases, there were 2 anaphylactic reactions, 1 case of angioedema, 2 of Stevens-Johnson syndrome, and 6 of rashes/allergies, the agency revealed in response to a request. Two patients were hospitalized; none died.
The information had not been made public until now.
At present, anaphylactic reactions and Stevens-Johnson syndrome are not mentioned in febuxostat (Uloric) labeling. Hypersensitivity is noted as a rare but possible adverse event.
The agency said in an e-mail that it is collecting febuxostat adverse event reports and will include them in a safety analysis to be completed in August.
The information is important because rheumatologists have been wondering how likely the drug is to cause hypersensitivity reactions, as it is typically used in patients who are intolerant of or allergic to allopurinol, which is associated with life-threatening, but rare, reactions.
The data do not answer that question, but do offer insight into it.
Takeda Pharmaceutical Co., febuxostat's maker, did not provide additional information when asked if the patients had kidney disease or allopurinol hypersensitivity. “We have been and will continue to work closely with the FDA to capture and monitor adverse events, as is our standard practice,” a Takeda spokesperson wrote in an e-mail.
Dr. Brian Mandell, a Cleveland Clinic rheumatologist, said that he is not surprised by the reports.
“Someone, somewhere is going to react to every drug. [There's] no free lunch,” Dr. Mandell said in an interview.
But, he added, “it is imperative that physicians realize that febuxostat can cause such reactions. There has been a tacit assumption that because it [has] a different molecular structure than allopurinol, such reactions will not occur.”
Febuxostat was approved by the agency in February 2009 for long-term management of chronic hyperuricemia in gout patients.
There were 139,565 prescriptions written for it in the United States in 2009, according to SDI Health LLC, a health care market insight and analytics firm.
No hypersensitivity reactions were attributed to febuxostat in trials comparing it to allopurinol, but allopurinol-sensitive patients were excluded, according to a briefing document that Takeda submitted to the FDA's Arthritis Advisory Committee in 2008 as well as the meeting transcript.
In trials, 1.6% of patients developed rashes in both the 80-mg febuxostat group and the allopurinol group, most of whom were on 300 mg. Among those in the placebo group, 0.7% developed rashes, according to febuxostat's label.
In early May, at a rheumatology conference sponsored by the University of California, Los Angeles, Dr. Mandell said, “the idea [that] you can give [febuxostat] to patients who are allopurinol hypersensitive is a guess. We just don't know.”
Allopurinol hypersensitivity syndrome, although rare, is a significant concern for physicians who use the drug to lower serum uric acid in patients with gout. Symptoms can include liver and kidney damage, Stevens-Johnson syndrome, and toxic epidermal necrolysis. It is fatal about 30% of the time. The majority of cases occur in patients who have chronic kidney disease.
While the medical community awaits additional febuxostat postmarketing safety data, Dr. Mandell said that he uses febuxostat in allopurinol-intolerant patients, but told attendees he doesn't think it's more effective.
He said that he doses febuxostat just as carefully as he doses allopurinol. As with allopurinol, he starts patients on a low dose and titrates up slowly, monitoring for tolerability and also to ensure that uric acid levels aren't dropped too precipitously, which can trigger gout attacks.
With both drugs, Dr. Mandell said that he tells patients to stop taking them if they develop a rash or any other hypersensitivity reaction symptoms, and to call him.
The treatment goal is to reduce the serum uric acid level to 6 mg/dL, with ongoing labs to ensure that it's reached.
He hasn't had a hypersensitivity reaction with febuxostat, “but my 'n' is 9,” he said in his presentation.
Although the lowest-dose febuxostat pill is 40 mg, he starts patients at 20 mg.
“Though the company suggests not to, I cut the pill,” he said.
Disclosures: Dr. Mandell disclosed that he is an advisor to Takeda and URL Pharma Inc., and he was a clinical investigator for Savient Pharmaceuticals Inc.
The Food and Drug Administration had received 11 reports of hypersensitivity reactions to febuxostat as of last month.
Among the 11 cases, there were 2 anaphylactic reactions, 1 case of angioedema, 2 of Stevens-Johnson syndrome, and 6 of rashes/allergies, the agency revealed in response to a request. Two patients were hospitalized; none died.
The information had not been made public until now.
At present, anaphylactic reactions and Stevens-Johnson syndrome are not mentioned in febuxostat (Uloric) labeling. Hypersensitivity is noted as a rare but possible adverse event.
The agency said in an e-mail that it is collecting febuxostat adverse event reports and will include them in a safety analysis to be completed in August.
The information is important because rheumatologists have been wondering how likely the drug is to cause hypersensitivity reactions, as it is typically used in patients who are intolerant of or allergic to allopurinol, which is associated with life-threatening, but rare, reactions.
The data do not answer that question, but do offer insight into it.
Takeda Pharmaceutical Co., febuxostat's maker, did not provide additional information when asked if the patients had kidney disease or allopurinol hypersensitivity. “We have been and will continue to work closely with the FDA to capture and monitor adverse events, as is our standard practice,” a Takeda spokesperson wrote in an e-mail.
Dr. Brian Mandell, a Cleveland Clinic rheumatologist, said that he is not surprised by the reports.
“Someone, somewhere is going to react to every drug. [There's] no free lunch,” Dr. Mandell said in an interview.
But, he added, “it is imperative that physicians realize that febuxostat can cause such reactions. There has been a tacit assumption that because it [has] a different molecular structure than allopurinol, such reactions will not occur.”
Febuxostat was approved by the agency in February 2009 for long-term management of chronic hyperuricemia in gout patients.
There were 139,565 prescriptions written for it in the United States in 2009, according to SDI Health LLC, a health care market insight and analytics firm.
No hypersensitivity reactions were attributed to febuxostat in trials comparing it to allopurinol, but allopurinol-sensitive patients were excluded, according to a briefing document that Takeda submitted to the FDA's Arthritis Advisory Committee in 2008 as well as the meeting transcript.
In trials, 1.6% of patients developed rashes in both the 80-mg febuxostat group and the allopurinol group, most of whom were on 300 mg. Among those in the placebo group, 0.7% developed rashes, according to febuxostat's label.
In early May, at a rheumatology conference sponsored by the University of California, Los Angeles, Dr. Mandell said, “the idea [that] you can give [febuxostat] to patients who are allopurinol hypersensitive is a guess. We just don't know.”
Allopurinol hypersensitivity syndrome, although rare, is a significant concern for physicians who use the drug to lower serum uric acid in patients with gout. Symptoms can include liver and kidney damage, Stevens-Johnson syndrome, and toxic epidermal necrolysis. It is fatal about 30% of the time. The majority of cases occur in patients who have chronic kidney disease.
While the medical community awaits additional febuxostat postmarketing safety data, Dr. Mandell said that he uses febuxostat in allopurinol-intolerant patients, but told attendees he doesn't think it's more effective.
He said that he doses febuxostat just as carefully as he doses allopurinol. As with allopurinol, he starts patients on a low dose and titrates up slowly, monitoring for tolerability and also to ensure that uric acid levels aren't dropped too precipitously, which can trigger gout attacks.
With both drugs, Dr. Mandell said that he tells patients to stop taking them if they develop a rash or any other hypersensitivity reaction symptoms, and to call him.
The treatment goal is to reduce the serum uric acid level to 6 mg/dL, with ongoing labs to ensure that it's reached.
He hasn't had a hypersensitivity reaction with febuxostat, “but my 'n' is 9,” he said in his presentation.
Although the lowest-dose febuxostat pill is 40 mg, he starts patients at 20 mg.
“Though the company suggests not to, I cut the pill,” he said.
Disclosures: Dr. Mandell disclosed that he is an advisor to Takeda and URL Pharma Inc., and he was a clinical investigator for Savient Pharmaceuticals Inc.
The Food and Drug Administration had received 11 reports of hypersensitivity reactions to febuxostat as of last month.
Among the 11 cases, there were 2 anaphylactic reactions, 1 case of angioedema, 2 of Stevens-Johnson syndrome, and 6 of rashes/allergies, the agency revealed in response to a request. Two patients were hospitalized; none died.
The information had not been made public until now.
At present, anaphylactic reactions and Stevens-Johnson syndrome are not mentioned in febuxostat (Uloric) labeling. Hypersensitivity is noted as a rare but possible adverse event.
The agency said in an e-mail that it is collecting febuxostat adverse event reports and will include them in a safety analysis to be completed in August.
The information is important because rheumatologists have been wondering how likely the drug is to cause hypersensitivity reactions, as it is typically used in patients who are intolerant of or allergic to allopurinol, which is associated with life-threatening, but rare, reactions.
The data do not answer that question, but do offer insight into it.
Takeda Pharmaceutical Co., febuxostat's maker, did not provide additional information when asked if the patients had kidney disease or allopurinol hypersensitivity. “We have been and will continue to work closely with the FDA to capture and monitor adverse events, as is our standard practice,” a Takeda spokesperson wrote in an e-mail.
Dr. Brian Mandell, a Cleveland Clinic rheumatologist, said that he is not surprised by the reports.
“Someone, somewhere is going to react to every drug. [There's] no free lunch,” Dr. Mandell said in an interview.
But, he added, “it is imperative that physicians realize that febuxostat can cause such reactions. There has been a tacit assumption that because it [has] a different molecular structure than allopurinol, such reactions will not occur.”
Febuxostat was approved by the agency in February 2009 for long-term management of chronic hyperuricemia in gout patients.
There were 139,565 prescriptions written for it in the United States in 2009, according to SDI Health LLC, a health care market insight and analytics firm.
No hypersensitivity reactions were attributed to febuxostat in trials comparing it to allopurinol, but allopurinol-sensitive patients were excluded, according to a briefing document that Takeda submitted to the FDA's Arthritis Advisory Committee in 2008 as well as the meeting transcript.
In trials, 1.6% of patients developed rashes in both the 80-mg febuxostat group and the allopurinol group, most of whom were on 300 mg. Among those in the placebo group, 0.7% developed rashes, according to febuxostat's label.
In early May, at a rheumatology conference sponsored by the University of California, Los Angeles, Dr. Mandell said, “the idea [that] you can give [febuxostat] to patients who are allopurinol hypersensitive is a guess. We just don't know.”
Allopurinol hypersensitivity syndrome, although rare, is a significant concern for physicians who use the drug to lower serum uric acid in patients with gout. Symptoms can include liver and kidney damage, Stevens-Johnson syndrome, and toxic epidermal necrolysis. It is fatal about 30% of the time. The majority of cases occur in patients who have chronic kidney disease.
While the medical community awaits additional febuxostat postmarketing safety data, Dr. Mandell said that he uses febuxostat in allopurinol-intolerant patients, but told attendees he doesn't think it's more effective.
He said that he doses febuxostat just as carefully as he doses allopurinol. As with allopurinol, he starts patients on a low dose and titrates up slowly, monitoring for tolerability and also to ensure that uric acid levels aren't dropped too precipitously, which can trigger gout attacks.
With both drugs, Dr. Mandell said that he tells patients to stop taking them if they develop a rash or any other hypersensitivity reaction symptoms, and to call him.
The treatment goal is to reduce the serum uric acid level to 6 mg/dL, with ongoing labs to ensure that it's reached.
He hasn't had a hypersensitivity reaction with febuxostat, “but my 'n' is 9,” he said in his presentation.
Although the lowest-dose febuxostat pill is 40 mg, he starts patients at 20 mg.
“Though the company suggests not to, I cut the pill,” he said.
Disclosures: Dr. Mandell disclosed that he is an advisor to Takeda and URL Pharma Inc., and he was a clinical investigator for Savient Pharmaceuticals Inc.
Patterns Emerge on Fractures From Bisphosphonate Use
MARINA DEL REY, CALIF. — As the debate unfolds over both whether bisphosphonates cause femur fractures and the degree to which the benefits of drugs still outweigh the risks, a phenomenon has emerged.
Women who have taken bisphosphonates for years are being seen with thigh pain that is easy to mistake for hip or knee arthritis. They have a unique range of radiologic findings, and they either have permanent titanium rods placed in their thigh bones or go on to full femur fractures—sometimes bilaterally—and permanent disability.
“The thinking [among colleagues] is that this is novel and specific to bisphosphonate treatment, but only time will tell,” Dr. Benjamin C. Bengs, an orthopedic surgeon at the University of California, Los Angeles, said in an interview.
The possibility must be added to the hip pain differential in women with long-standing use of the drugs, according to Dr. Bengs and others who were interviewed for this story.
Unusual Radiology Findings
For many of the doctors who were interviewed, the question isn't so much whether bisphosphonates cause femur fractures, but rather how to care for women with a long history of using the drugs, and how to recognize signs and symptoms of impending trouble.
Any woman with pain over the thigh and those x-ray findings is a candidate for prophylactic rodding, said Dr. Bengs.
The unique findings on x-ray are cortical thickening that is most pronounced on the lateral side of the femur, accompanied by a beaking lesion, also on the lateral side. Intramedullary edema is often present.
The beaking lesion is the start of a horizontal or oblique stress fracture. In time, there generally develops a “little, lucent, dark line extending from the beak to the middle of the bone,” Dr. Joseph Robinson, a radiology fellow at Cedars-Sinai Medical Center in Los Angeles, said in an interview.
“Lateral stress fractures are unusual. In our area, they are all related to bisphosphonates,” Dr. Kambiz Motamedi, a diagnostic radiologist at UCLA Medical Center.
Both Legs Must be Examined
Standard hip x-rays don't go far enough down to detect the lesion, which is closer to the knee than the typical femur fracture would be, Dr. Robinson said. When they do a hip series, Cedars-Sinai radiologists are careful to image lower down so they don't miss it, he said, noting that they are also putting markers on skin to identify the source of pain.
If the lesion is found, it is imperative to image the other femur as well, said Dr. Stuart L. Silverman, a rheumatologist in private practice in Beverly Hills, Calif. The other femur can go on to fracture, often within 18 months, he said in an interview.
Dr. Bengs noted that in most cases, rodding is the usual treatment. It takes only a matter of weeks to recover from hip-to-knee rod placement, he said. It takes months to recover from a fracture, however, and older patients usually lose 10%–15% of strength and ambulation during convalescence, he added.
“The fractures are devastating,” said UCLA and Cedars-Sinai rheumatologist Dr. Solomon N. Forouzesh, who has seen two cases in his practice.
Who is at Risk?
Active women who have osteopenia—not osteoporosis—appear to be most at risk, Dr. Nancy Lane, a University of California, Davis, rheumatologist, said in an interview. “What I think is going on” is that bisphosphonates, by reducing bone turnover, lead to the overmineralization of cortical bone, she explained. “Over time, the bones become brittle [and] fail from too much mineralization. They cannot dissipate the load.”
It's “probably best not [to use bisphosphonates] in people with low risk of fractures who are very active,” she said, adding that use of the drugs might have been “too aggressive” in the past.
A Drug Holiday Is Advised
As the story unfolds, doctors are using bisphosphonates for shorter lengths of time, followed by a drug holiday and ongoing bone-density monitoring.
The risk of fracture seems to “start at about 3 years and peaks between 5 and 6 years,” Dr. Forouzesh said. To be ahead of the game, he advises not waiting until the risk peaks. “Back off ahead of time. I do a drug holiday in 3 or 4 years,” he said.
It's not clear at this point if the phenomenon—if it is truly real—is limited to alendronate or a bisphosphonate class effect.
Alendronate has been on the market the longest and has been the most widely used agent, Dr. Lane noted. “I am sure over time” it will emerge with other members of the class, as well, she said.
Disclosures: Dr. Motamedi, Dr. Robinson, and Dr. Forouzesh said that they had no conflicts to disclose. Dr. Bengs disclosed he is a paid consultant for Amgen Inc. Dr. Lane disclosed research grants, royalties, consulting fees from and positions of influence and ownership interests in Amgen, Eli Lilly & Co., and Pfizer Inc. Dr. Silverman disclosed he has served as a speaker, member of a speakers bureau, advisor for Eli Lilly, Novartis Pharmaceuticals Corp., Procter & Gamble, and Roche Inc., and that he has received research support from Eli Lilly, Procter & Gamble, Roche, and Novartis.
Cortical thickening, beak-like stress fracture are seen here.
Source Courtesy UCLA
MARINA DEL REY, CALIF. — As the debate unfolds over both whether bisphosphonates cause femur fractures and the degree to which the benefits of drugs still outweigh the risks, a phenomenon has emerged.
Women who have taken bisphosphonates for years are being seen with thigh pain that is easy to mistake for hip or knee arthritis. They have a unique range of radiologic findings, and they either have permanent titanium rods placed in their thigh bones or go on to full femur fractures—sometimes bilaterally—and permanent disability.
“The thinking [among colleagues] is that this is novel and specific to bisphosphonate treatment, but only time will tell,” Dr. Benjamin C. Bengs, an orthopedic surgeon at the University of California, Los Angeles, said in an interview.
The possibility must be added to the hip pain differential in women with long-standing use of the drugs, according to Dr. Bengs and others who were interviewed for this story.
Unusual Radiology Findings
For many of the doctors who were interviewed, the question isn't so much whether bisphosphonates cause femur fractures, but rather how to care for women with a long history of using the drugs, and how to recognize signs and symptoms of impending trouble.
Any woman with pain over the thigh and those x-ray findings is a candidate for prophylactic rodding, said Dr. Bengs.
The unique findings on x-ray are cortical thickening that is most pronounced on the lateral side of the femur, accompanied by a beaking lesion, also on the lateral side. Intramedullary edema is often present.
The beaking lesion is the start of a horizontal or oblique stress fracture. In time, there generally develops a “little, lucent, dark line extending from the beak to the middle of the bone,” Dr. Joseph Robinson, a radiology fellow at Cedars-Sinai Medical Center in Los Angeles, said in an interview.
“Lateral stress fractures are unusual. In our area, they are all related to bisphosphonates,” Dr. Kambiz Motamedi, a diagnostic radiologist at UCLA Medical Center.
Both Legs Must be Examined
Standard hip x-rays don't go far enough down to detect the lesion, which is closer to the knee than the typical femur fracture would be, Dr. Robinson said. When they do a hip series, Cedars-Sinai radiologists are careful to image lower down so they don't miss it, he said, noting that they are also putting markers on skin to identify the source of pain.
If the lesion is found, it is imperative to image the other femur as well, said Dr. Stuart L. Silverman, a rheumatologist in private practice in Beverly Hills, Calif. The other femur can go on to fracture, often within 18 months, he said in an interview.
Dr. Bengs noted that in most cases, rodding is the usual treatment. It takes only a matter of weeks to recover from hip-to-knee rod placement, he said. It takes months to recover from a fracture, however, and older patients usually lose 10%–15% of strength and ambulation during convalescence, he added.
“The fractures are devastating,” said UCLA and Cedars-Sinai rheumatologist Dr. Solomon N. Forouzesh, who has seen two cases in his practice.
Who is at Risk?
Active women who have osteopenia—not osteoporosis—appear to be most at risk, Dr. Nancy Lane, a University of California, Davis, rheumatologist, said in an interview. “What I think is going on” is that bisphosphonates, by reducing bone turnover, lead to the overmineralization of cortical bone, she explained. “Over time, the bones become brittle [and] fail from too much mineralization. They cannot dissipate the load.”
It's “probably best not [to use bisphosphonates] in people with low risk of fractures who are very active,” she said, adding that use of the drugs might have been “too aggressive” in the past.
A Drug Holiday Is Advised
As the story unfolds, doctors are using bisphosphonates for shorter lengths of time, followed by a drug holiday and ongoing bone-density monitoring.
The risk of fracture seems to “start at about 3 years and peaks between 5 and 6 years,” Dr. Forouzesh said. To be ahead of the game, he advises not waiting until the risk peaks. “Back off ahead of time. I do a drug holiday in 3 or 4 years,” he said.
It's not clear at this point if the phenomenon—if it is truly real—is limited to alendronate or a bisphosphonate class effect.
Alendronate has been on the market the longest and has been the most widely used agent, Dr. Lane noted. “I am sure over time” it will emerge with other members of the class, as well, she said.
Disclosures: Dr. Motamedi, Dr. Robinson, and Dr. Forouzesh said that they had no conflicts to disclose. Dr. Bengs disclosed he is a paid consultant for Amgen Inc. Dr. Lane disclosed research grants, royalties, consulting fees from and positions of influence and ownership interests in Amgen, Eli Lilly & Co., and Pfizer Inc. Dr. Silverman disclosed he has served as a speaker, member of a speakers bureau, advisor for Eli Lilly, Novartis Pharmaceuticals Corp., Procter & Gamble, and Roche Inc., and that he has received research support from Eli Lilly, Procter & Gamble, Roche, and Novartis.
Cortical thickening, beak-like stress fracture are seen here.
Source Courtesy UCLA
MARINA DEL REY, CALIF. — As the debate unfolds over both whether bisphosphonates cause femur fractures and the degree to which the benefits of drugs still outweigh the risks, a phenomenon has emerged.
Women who have taken bisphosphonates for years are being seen with thigh pain that is easy to mistake for hip or knee arthritis. They have a unique range of radiologic findings, and they either have permanent titanium rods placed in their thigh bones or go on to full femur fractures—sometimes bilaterally—and permanent disability.
“The thinking [among colleagues] is that this is novel and specific to bisphosphonate treatment, but only time will tell,” Dr. Benjamin C. Bengs, an orthopedic surgeon at the University of California, Los Angeles, said in an interview.
The possibility must be added to the hip pain differential in women with long-standing use of the drugs, according to Dr. Bengs and others who were interviewed for this story.
Unusual Radiology Findings
For many of the doctors who were interviewed, the question isn't so much whether bisphosphonates cause femur fractures, but rather how to care for women with a long history of using the drugs, and how to recognize signs and symptoms of impending trouble.
Any woman with pain over the thigh and those x-ray findings is a candidate for prophylactic rodding, said Dr. Bengs.
The unique findings on x-ray are cortical thickening that is most pronounced on the lateral side of the femur, accompanied by a beaking lesion, also on the lateral side. Intramedullary edema is often present.
The beaking lesion is the start of a horizontal or oblique stress fracture. In time, there generally develops a “little, lucent, dark line extending from the beak to the middle of the bone,” Dr. Joseph Robinson, a radiology fellow at Cedars-Sinai Medical Center in Los Angeles, said in an interview.
“Lateral stress fractures are unusual. In our area, they are all related to bisphosphonates,” Dr. Kambiz Motamedi, a diagnostic radiologist at UCLA Medical Center.
Both Legs Must be Examined
Standard hip x-rays don't go far enough down to detect the lesion, which is closer to the knee than the typical femur fracture would be, Dr. Robinson said. When they do a hip series, Cedars-Sinai radiologists are careful to image lower down so they don't miss it, he said, noting that they are also putting markers on skin to identify the source of pain.
If the lesion is found, it is imperative to image the other femur as well, said Dr. Stuart L. Silverman, a rheumatologist in private practice in Beverly Hills, Calif. The other femur can go on to fracture, often within 18 months, he said in an interview.
Dr. Bengs noted that in most cases, rodding is the usual treatment. It takes only a matter of weeks to recover from hip-to-knee rod placement, he said. It takes months to recover from a fracture, however, and older patients usually lose 10%–15% of strength and ambulation during convalescence, he added.
“The fractures are devastating,” said UCLA and Cedars-Sinai rheumatologist Dr. Solomon N. Forouzesh, who has seen two cases in his practice.
Who is at Risk?
Active women who have osteopenia—not osteoporosis—appear to be most at risk, Dr. Nancy Lane, a University of California, Davis, rheumatologist, said in an interview. “What I think is going on” is that bisphosphonates, by reducing bone turnover, lead to the overmineralization of cortical bone, she explained. “Over time, the bones become brittle [and] fail from too much mineralization. They cannot dissipate the load.”
It's “probably best not [to use bisphosphonates] in people with low risk of fractures who are very active,” she said, adding that use of the drugs might have been “too aggressive” in the past.
A Drug Holiday Is Advised
As the story unfolds, doctors are using bisphosphonates for shorter lengths of time, followed by a drug holiday and ongoing bone-density monitoring.
The risk of fracture seems to “start at about 3 years and peaks between 5 and 6 years,” Dr. Forouzesh said. To be ahead of the game, he advises not waiting until the risk peaks. “Back off ahead of time. I do a drug holiday in 3 or 4 years,” he said.
It's not clear at this point if the phenomenon—if it is truly real—is limited to alendronate or a bisphosphonate class effect.
Alendronate has been on the market the longest and has been the most widely used agent, Dr. Lane noted. “I am sure over time” it will emerge with other members of the class, as well, she said.
Disclosures: Dr. Motamedi, Dr. Robinson, and Dr. Forouzesh said that they had no conflicts to disclose. Dr. Bengs disclosed he is a paid consultant for Amgen Inc. Dr. Lane disclosed research grants, royalties, consulting fees from and positions of influence and ownership interests in Amgen, Eli Lilly & Co., and Pfizer Inc. Dr. Silverman disclosed he has served as a speaker, member of a speakers bureau, advisor for Eli Lilly, Novartis Pharmaceuticals Corp., Procter & Gamble, and Roche Inc., and that he has received research support from Eli Lilly, Procter & Gamble, Roche, and Novartis.
Cortical thickening, beak-like stress fracture are seen here.
Source Courtesy UCLA
Unbranded Colchicine Getting Hard to Find
As URL Pharma Inc. and other drug companies battle in court over access to the U.S. colchicine market, the supply of unapproved colchicine is beginning to decline, and patients in at least one small town are scrambling to find what's left.
“It's harder to get,” Dr. Christopher Morris, a rheumatologist in Kingsport, Tenn., said in an interview. “Some [patients] have had to go to two or three different pharmacies to find it. Some are going online to find it. Some pharmacies only have Colcrys available,” he added.
The situation started last summer when the Food and Drug Administration approved URL's colchicine (Colcrys) for gout and familial Mediterranean fever.
URL was the first company to submit colchicine for approval and, under FDA regulations, the company was granted multiyear marketing exclusivity.
After the approval, URL went to court to end its competitors' sales on the grounds that by marketing their colchicine, they falsely imply that it is FDA approved. Instead, unapproved colchicine was grandfathered into the marketplace by the Food, Drug, and Cosmetic Act of 1938.
Two manufacturers of unbranded colchicine, Vision Pharma and Excellium Pharmaceuticals, were no longer supplying the U.S. market at the end of April, according to the American Society of Health System Pharmacists' drug shortage Web site. A third, West-Ward Pharmaceutical Corp., was still doing so, according to the Web site.
The unapproved colchicine business had been growing until now; 1.2 million retail prescriptions were written for it in the first 4 months of this year, compared with 1.1 million for the same period last year and 1 million during the first 4 months of 2008, according to SDI Health, a health care market insight and analytics firm.
Meanwhile, the price of unbranded colchicine has gone from a nationwide average of 19 cents per pill in 2008 to 44 cents, according to SDI.
West-Ward, Vision, and Excellium declined to comment for this story, citing ongoing litigation.
Several physicians said in interviews that although patients are complaining about the higher cost, they can still get the pills. But some physicians are writing larger-than-usual prescriptions to help patients stock up< while others say they will refer patients to the Internet and Canadian pharmacies if they run into trouble.
Two companies, Euro-Pharm International Canada and Odan Laboratories, sell colchicine in Canada, according to Gary Holub, a Health Canada spokesman.
Colcrys sells for about $5 per pill, according to DestinationRx.com
Doctors have said that their patients have safely used unapproved colchicine for decades, and that the higher price of Colcrys will limit patient access and drain public health programs (N. Engl. J. Med. 2010 April 21 [doi:10.1056/NEJMp1003126]).
There have also been calls for the FDA to tighten its marketing exclusivity regulations, and concern that the agency will remove unapproved colchicine from the market as part of its goal to ensure that drugs sold in the United States are tested for safety and efficacy.
In response, URL asserts that the marketing of unbranded colchicine is illegal.
The company also points to a March letter from Dr. Janet Woodcock, director of the FDA's Center for Drug Evaluation and Research, to the American College of Rheumatology. The letter stated that Colcrys approval studies generated important new information about how to safely use colchicine, and that the safety, purity, and efficacy of unapproved drugs cannot be guaranteed.
Because of the latter reason, unbranded colchicine was pulled from Medicare part D formularies this year and was replaced by Colcrys.
Whatever the outcome of the court battles, the situation isn't permanent.
URL's marketing exclusivity for Colcrys's gout indications expires July 30, 2012. The familial Mediterranean fever indication expires July 29, 2016, according to the FDA's Web site.
Disclosures: Dr. Morris disclosed that he is on the speakers' bureau for Takeda, Eli Lilly, and Forest Pharmaceuticals.
Information about URL's Colcrys patient assistance program and copayment coupons are available by visiting Colcrys.com
As URL Pharma Inc. and other drug companies battle in court over access to the U.S. colchicine market, the supply of unapproved colchicine is beginning to decline, and patients in at least one small town are scrambling to find what's left.
“It's harder to get,” Dr. Christopher Morris, a rheumatologist in Kingsport, Tenn., said in an interview. “Some [patients] have had to go to two or three different pharmacies to find it. Some are going online to find it. Some pharmacies only have Colcrys available,” he added.
The situation started last summer when the Food and Drug Administration approved URL's colchicine (Colcrys) for gout and familial Mediterranean fever.
URL was the first company to submit colchicine for approval and, under FDA regulations, the company was granted multiyear marketing exclusivity.
After the approval, URL went to court to end its competitors' sales on the grounds that by marketing their colchicine, they falsely imply that it is FDA approved. Instead, unapproved colchicine was grandfathered into the marketplace by the Food, Drug, and Cosmetic Act of 1938.
Two manufacturers of unbranded colchicine, Vision Pharma and Excellium Pharmaceuticals, were no longer supplying the U.S. market at the end of April, according to the American Society of Health System Pharmacists' drug shortage Web site. A third, West-Ward Pharmaceutical Corp., was still doing so, according to the Web site.
The unapproved colchicine business had been growing until now; 1.2 million retail prescriptions were written for it in the first 4 months of this year, compared with 1.1 million for the same period last year and 1 million during the first 4 months of 2008, according to SDI Health, a health care market insight and analytics firm.
Meanwhile, the price of unbranded colchicine has gone from a nationwide average of 19 cents per pill in 2008 to 44 cents, according to SDI.
West-Ward, Vision, and Excellium declined to comment for this story, citing ongoing litigation.
Several physicians said in interviews that although patients are complaining about the higher cost, they can still get the pills. But some physicians are writing larger-than-usual prescriptions to help patients stock up< while others say they will refer patients to the Internet and Canadian pharmacies if they run into trouble.
Two companies, Euro-Pharm International Canada and Odan Laboratories, sell colchicine in Canada, according to Gary Holub, a Health Canada spokesman.
Colcrys sells for about $5 per pill, according to DestinationRx.com
Doctors have said that their patients have safely used unapproved colchicine for decades, and that the higher price of Colcrys will limit patient access and drain public health programs (N. Engl. J. Med. 2010 April 21 [doi:10.1056/NEJMp1003126]).
There have also been calls for the FDA to tighten its marketing exclusivity regulations, and concern that the agency will remove unapproved colchicine from the market as part of its goal to ensure that drugs sold in the United States are tested for safety and efficacy.
In response, URL asserts that the marketing of unbranded colchicine is illegal.
The company also points to a March letter from Dr. Janet Woodcock, director of the FDA's Center for Drug Evaluation and Research, to the American College of Rheumatology. The letter stated that Colcrys approval studies generated important new information about how to safely use colchicine, and that the safety, purity, and efficacy of unapproved drugs cannot be guaranteed.
Because of the latter reason, unbranded colchicine was pulled from Medicare part D formularies this year and was replaced by Colcrys.
Whatever the outcome of the court battles, the situation isn't permanent.
URL's marketing exclusivity for Colcrys's gout indications expires July 30, 2012. The familial Mediterranean fever indication expires July 29, 2016, according to the FDA's Web site.
Disclosures: Dr. Morris disclosed that he is on the speakers' bureau for Takeda, Eli Lilly, and Forest Pharmaceuticals.
Information about URL's Colcrys patient assistance program and copayment coupons are available by visiting Colcrys.com
As URL Pharma Inc. and other drug companies battle in court over access to the U.S. colchicine market, the supply of unapproved colchicine is beginning to decline, and patients in at least one small town are scrambling to find what's left.
“It's harder to get,” Dr. Christopher Morris, a rheumatologist in Kingsport, Tenn., said in an interview. “Some [patients] have had to go to two or three different pharmacies to find it. Some are going online to find it. Some pharmacies only have Colcrys available,” he added.
The situation started last summer when the Food and Drug Administration approved URL's colchicine (Colcrys) for gout and familial Mediterranean fever.
URL was the first company to submit colchicine for approval and, under FDA regulations, the company was granted multiyear marketing exclusivity.
After the approval, URL went to court to end its competitors' sales on the grounds that by marketing their colchicine, they falsely imply that it is FDA approved. Instead, unapproved colchicine was grandfathered into the marketplace by the Food, Drug, and Cosmetic Act of 1938.
Two manufacturers of unbranded colchicine, Vision Pharma and Excellium Pharmaceuticals, were no longer supplying the U.S. market at the end of April, according to the American Society of Health System Pharmacists' drug shortage Web site. A third, West-Ward Pharmaceutical Corp., was still doing so, according to the Web site.
The unapproved colchicine business had been growing until now; 1.2 million retail prescriptions were written for it in the first 4 months of this year, compared with 1.1 million for the same period last year and 1 million during the first 4 months of 2008, according to SDI Health, a health care market insight and analytics firm.
Meanwhile, the price of unbranded colchicine has gone from a nationwide average of 19 cents per pill in 2008 to 44 cents, according to SDI.
West-Ward, Vision, and Excellium declined to comment for this story, citing ongoing litigation.
Several physicians said in interviews that although patients are complaining about the higher cost, they can still get the pills. But some physicians are writing larger-than-usual prescriptions to help patients stock up< while others say they will refer patients to the Internet and Canadian pharmacies if they run into trouble.
Two companies, Euro-Pharm International Canada and Odan Laboratories, sell colchicine in Canada, according to Gary Holub, a Health Canada spokesman.
Colcrys sells for about $5 per pill, according to DestinationRx.com
Doctors have said that their patients have safely used unapproved colchicine for decades, and that the higher price of Colcrys will limit patient access and drain public health programs (N. Engl. J. Med. 2010 April 21 [doi:10.1056/NEJMp1003126]).
There have also been calls for the FDA to tighten its marketing exclusivity regulations, and concern that the agency will remove unapproved colchicine from the market as part of its goal to ensure that drugs sold in the United States are tested for safety and efficacy.
In response, URL asserts that the marketing of unbranded colchicine is illegal.
The company also points to a March letter from Dr. Janet Woodcock, director of the FDA's Center for Drug Evaluation and Research, to the American College of Rheumatology. The letter stated that Colcrys approval studies generated important new information about how to safely use colchicine, and that the safety, purity, and efficacy of unapproved drugs cannot be guaranteed.
Because of the latter reason, unbranded colchicine was pulled from Medicare part D formularies this year and was replaced by Colcrys.
Whatever the outcome of the court battles, the situation isn't permanent.
URL's marketing exclusivity for Colcrys's gout indications expires July 30, 2012. The familial Mediterranean fever indication expires July 29, 2016, according to the FDA's Web site.
Disclosures: Dr. Morris disclosed that he is on the speakers' bureau for Takeda, Eli Lilly, and Forest Pharmaceuticals.
Information about URL's Colcrys patient assistance program and copayment coupons are available by visiting Colcrys.com
Doctors Concerned About Shrinking Supply of Unapproved Colchicine
As URL Pharma Inc. and other drug companies battle in court over access to the U.S. colchicine market, the supply of unapproved colchicine is beginning to decline, and patients in at least one small town are scrambling to find what’s left.
“It’s harder to get,” Dr. Christopher Morris, a rheumatologist in Kingsport, Tenn., said in an interview. “Some [patients] have had to go to two or three different pharmacies to find it. Some are going online to find it. Some pharmacies only have Colcrys available,” he added.
The situation started last summer when the Food and Drug Administration approved URL’s colchicine (Colcrys) for gout and familial Mediterranean fever.
URL was the first company to submit colchicine for approval and, under FDA regulations, the company was granted multiyear marketing exclusivity for its efforts.
After the approval, URL went to court to end its competitors’ sales on the grounds that by marketing their colchicine, they falsely imply that it is FDA approved.
Instead, unapproved colchicine was grandfathered into the marketplace by the Food, Drug, and Cosmetic Act of 1938.
Two manufacturers of unbranded colchicine, Vision Pharma LLC and Excellium Pharmaceuticals Inc., were no longer supplying the U.S. market at the end of April, according to the American Society of Health System Pharmacists’ drug shortage Web site.
A third, West-Ward Pharmaceutical Corp., was still doing so, according to the Web site.
The unapproved colchicine business had been growing until now; 1.2 million retail prescriptions were written for it in the first 4 months of this year, compared with 1.1 million for the same period last year and 1 million during the first 4 months of 2008, according to SDI Health LLC, a health care market insight and analytics firm.
Meanwhile, the price of unbranded colchicine has gone from a nationwide average of 19 cents per pill in 2008 to 44 cents, according to SDI.
West-Ward, Vision, and Excellium declined to comment for this story, citing ongoing litigation.
Stocking up
Physicians are bracing for an unbranded colchicine shortage.
Physicians in Cleveland; Austin, Tex.; and New York City said in interviews that although patients are complaining about the higher cost, they can still get the pills.
But anticipating dwindling supplies, Dr. Edward Fudman, a rheumatologist in Austin, said he is writing larger than usual prescriptions to help his patients stock up.
Dr. Brian Mandell, a Cleveland Clinic rheumatologist, said he will refer patients to the Internet and Canadian pharmacies if they run into trouble.
Two companies, Euro-Pharm International Canada Inc. and Odan Laboratories Ltd., sell colchicine in Canada, according to Gary Holub, a Health Canada spokesman.
Another Alternative
Colcrys sells for about $5 per pill, according to DestinationRx.com.
But URL has both a patient assistance program and a copay coupon program to help eligible patients. In this way, the company has taken steps to “ensure everyone in need of Colcrys therapy can get it regardless of income level,” a spokesperson said.
The Colcrys patient assistance program is tiered. A 30-day supply of Colcrys, for example, is free for households with an annual income of up to three times the federal poverty level ($66,150 per year for a family of four), a URL spokesperson said.
Assistance is available for households up to six times the federal poverty level ($132,000 per year for a four-member family).
Under the coupon program, the cost is no more than $25 for prescription of 30 pills or more, the spokesman said.
With Changes Come Tension
Even so, the changes in the U.S. colchicine market have brought tension, with strong opinions among those involved.
Doctors have said that their patients have safely used unapproved colchicine for decades, and that the higher price of Colcrys will limit patient access and drain public health programs (N. Engl. J. Med. 2010 April 21 [doi:10.1056/NEJMp 1003126]).
There have also been calls for the FDA to tighten its marketing exclusivity regulations, and concern that the agency will remove unapproved colchicine from the market as part of its goal to ensure that drugs sold in the United States are tested for safety and efficacy.
In response, URL asserts that the marketing of unbranded colchicine is illegal.
The company also points to a March letter from Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, to the American College of Rheumatology. The letter stated that Colcrys approval studies generated important new information about how to safely use colchicine, and that the safety, purity, and efficacy of unapproved drugs cannot be guaranteed.
Because of the latter reason, unbranded colchicine was pulled from Medicare part D formularies this year, and was replaced by Colcrys.
Whatever the outcome of the court battles, the situation isn’t permanent.
URL’s marketing exclusivity for Colcrys’s gout indications expires July 30, 2012. The familial Mediterranean fever indication expires July 29, 2016, according to the FDA’s Web site.
Information about URL’s Colcrys patient assistance program and copayment coupons are available by visiting Colcrys.com or by calling 888-811-8423. Copayment coupons can be downloaded at the Web site.
Dr. Fudman said that he had no relevant disclosures. Dr. Morris disclosed that he is on the speakers’ bureau for Takeda, Eli Lilly, and Forest Pharmaceuticals. Dr. Mandell disclosed that he is an advisor to Takeda and URL Pharma, and was a clinical investigator for Savient Pharmaceuticals.
As URL Pharma Inc. and other drug companies battle in court over access to the U.S. colchicine market, the supply of unapproved colchicine is beginning to decline, and patients in at least one small town are scrambling to find what’s left.
“It’s harder to get,” Dr. Christopher Morris, a rheumatologist in Kingsport, Tenn., said in an interview. “Some [patients] have had to go to two or three different pharmacies to find it. Some are going online to find it. Some pharmacies only have Colcrys available,” he added.
The situation started last summer when the Food and Drug Administration approved URL’s colchicine (Colcrys) for gout and familial Mediterranean fever.
URL was the first company to submit colchicine for approval and, under FDA regulations, the company was granted multiyear marketing exclusivity for its efforts.
After the approval, URL went to court to end its competitors’ sales on the grounds that by marketing their colchicine, they falsely imply that it is FDA approved.
Instead, unapproved colchicine was grandfathered into the marketplace by the Food, Drug, and Cosmetic Act of 1938.
Two manufacturers of unbranded colchicine, Vision Pharma LLC and Excellium Pharmaceuticals Inc., were no longer supplying the U.S. market at the end of April, according to the American Society of Health System Pharmacists’ drug shortage Web site.
A third, West-Ward Pharmaceutical Corp., was still doing so, according to the Web site.
The unapproved colchicine business had been growing until now; 1.2 million retail prescriptions were written for it in the first 4 months of this year, compared with 1.1 million for the same period last year and 1 million during the first 4 months of 2008, according to SDI Health LLC, a health care market insight and analytics firm.
Meanwhile, the price of unbranded colchicine has gone from a nationwide average of 19 cents per pill in 2008 to 44 cents, according to SDI.
West-Ward, Vision, and Excellium declined to comment for this story, citing ongoing litigation.
Stocking up
Physicians are bracing for an unbranded colchicine shortage.
Physicians in Cleveland; Austin, Tex.; and New York City said in interviews that although patients are complaining about the higher cost, they can still get the pills.
But anticipating dwindling supplies, Dr. Edward Fudman, a rheumatologist in Austin, said he is writing larger than usual prescriptions to help his patients stock up.
Dr. Brian Mandell, a Cleveland Clinic rheumatologist, said he will refer patients to the Internet and Canadian pharmacies if they run into trouble.
Two companies, Euro-Pharm International Canada Inc. and Odan Laboratories Ltd., sell colchicine in Canada, according to Gary Holub, a Health Canada spokesman.
Another Alternative
Colcrys sells for about $5 per pill, according to DestinationRx.com.
But URL has both a patient assistance program and a copay coupon program to help eligible patients. In this way, the company has taken steps to “ensure everyone in need of Colcrys therapy can get it regardless of income level,” a spokesperson said.
The Colcrys patient assistance program is tiered. A 30-day supply of Colcrys, for example, is free for households with an annual income of up to three times the federal poverty level ($66,150 per year for a family of four), a URL spokesperson said.
Assistance is available for households up to six times the federal poverty level ($132,000 per year for a four-member family).
Under the coupon program, the cost is no more than $25 for prescription of 30 pills or more, the spokesman said.
With Changes Come Tension
Even so, the changes in the U.S. colchicine market have brought tension, with strong opinions among those involved.
Doctors have said that their patients have safely used unapproved colchicine for decades, and that the higher price of Colcrys will limit patient access and drain public health programs (N. Engl. J. Med. 2010 April 21 [doi:10.1056/NEJMp 1003126]).
There have also been calls for the FDA to tighten its marketing exclusivity regulations, and concern that the agency will remove unapproved colchicine from the market as part of its goal to ensure that drugs sold in the United States are tested for safety and efficacy.
In response, URL asserts that the marketing of unbranded colchicine is illegal.
The company also points to a March letter from Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, to the American College of Rheumatology. The letter stated that Colcrys approval studies generated important new information about how to safely use colchicine, and that the safety, purity, and efficacy of unapproved drugs cannot be guaranteed.
Because of the latter reason, unbranded colchicine was pulled from Medicare part D formularies this year, and was replaced by Colcrys.
Whatever the outcome of the court battles, the situation isn’t permanent.
URL’s marketing exclusivity for Colcrys’s gout indications expires July 30, 2012. The familial Mediterranean fever indication expires July 29, 2016, according to the FDA’s Web site.
Information about URL’s Colcrys patient assistance program and copayment coupons are available by visiting Colcrys.com or by calling 888-811-8423. Copayment coupons can be downloaded at the Web site.
Dr. Fudman said that he had no relevant disclosures. Dr. Morris disclosed that he is on the speakers’ bureau for Takeda, Eli Lilly, and Forest Pharmaceuticals. Dr. Mandell disclosed that he is an advisor to Takeda and URL Pharma, and was a clinical investigator for Savient Pharmaceuticals.
As URL Pharma Inc. and other drug companies battle in court over access to the U.S. colchicine market, the supply of unapproved colchicine is beginning to decline, and patients in at least one small town are scrambling to find what’s left.
“It’s harder to get,” Dr. Christopher Morris, a rheumatologist in Kingsport, Tenn., said in an interview. “Some [patients] have had to go to two or three different pharmacies to find it. Some are going online to find it. Some pharmacies only have Colcrys available,” he added.
The situation started last summer when the Food and Drug Administration approved URL’s colchicine (Colcrys) for gout and familial Mediterranean fever.
URL was the first company to submit colchicine for approval and, under FDA regulations, the company was granted multiyear marketing exclusivity for its efforts.
After the approval, URL went to court to end its competitors’ sales on the grounds that by marketing their colchicine, they falsely imply that it is FDA approved.
Instead, unapproved colchicine was grandfathered into the marketplace by the Food, Drug, and Cosmetic Act of 1938.
Two manufacturers of unbranded colchicine, Vision Pharma LLC and Excellium Pharmaceuticals Inc., were no longer supplying the U.S. market at the end of April, according to the American Society of Health System Pharmacists’ drug shortage Web site.
A third, West-Ward Pharmaceutical Corp., was still doing so, according to the Web site.
The unapproved colchicine business had been growing until now; 1.2 million retail prescriptions were written for it in the first 4 months of this year, compared with 1.1 million for the same period last year and 1 million during the first 4 months of 2008, according to SDI Health LLC, a health care market insight and analytics firm.
Meanwhile, the price of unbranded colchicine has gone from a nationwide average of 19 cents per pill in 2008 to 44 cents, according to SDI.
West-Ward, Vision, and Excellium declined to comment for this story, citing ongoing litigation.
Stocking up
Physicians are bracing for an unbranded colchicine shortage.
Physicians in Cleveland; Austin, Tex.; and New York City said in interviews that although patients are complaining about the higher cost, they can still get the pills.
But anticipating dwindling supplies, Dr. Edward Fudman, a rheumatologist in Austin, said he is writing larger than usual prescriptions to help his patients stock up.
Dr. Brian Mandell, a Cleveland Clinic rheumatologist, said he will refer patients to the Internet and Canadian pharmacies if they run into trouble.
Two companies, Euro-Pharm International Canada Inc. and Odan Laboratories Ltd., sell colchicine in Canada, according to Gary Holub, a Health Canada spokesman.
Another Alternative
Colcrys sells for about $5 per pill, according to DestinationRx.com.
But URL has both a patient assistance program and a copay coupon program to help eligible patients. In this way, the company has taken steps to “ensure everyone in need of Colcrys therapy can get it regardless of income level,” a spokesperson said.
The Colcrys patient assistance program is tiered. A 30-day supply of Colcrys, for example, is free for households with an annual income of up to three times the federal poverty level ($66,150 per year for a family of four), a URL spokesperson said.
Assistance is available for households up to six times the federal poverty level ($132,000 per year for a four-member family).
Under the coupon program, the cost is no more than $25 for prescription of 30 pills or more, the spokesman said.
With Changes Come Tension
Even so, the changes in the U.S. colchicine market have brought tension, with strong opinions among those involved.
Doctors have said that their patients have safely used unapproved colchicine for decades, and that the higher price of Colcrys will limit patient access and drain public health programs (N. Engl. J. Med. 2010 April 21 [doi:10.1056/NEJMp 1003126]).
There have also been calls for the FDA to tighten its marketing exclusivity regulations, and concern that the agency will remove unapproved colchicine from the market as part of its goal to ensure that drugs sold in the United States are tested for safety and efficacy.
In response, URL asserts that the marketing of unbranded colchicine is illegal.
The company also points to a March letter from Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, to the American College of Rheumatology. The letter stated that Colcrys approval studies generated important new information about how to safely use colchicine, and that the safety, purity, and efficacy of unapproved drugs cannot be guaranteed.
Because of the latter reason, unbranded colchicine was pulled from Medicare part D formularies this year, and was replaced by Colcrys.
Whatever the outcome of the court battles, the situation isn’t permanent.
URL’s marketing exclusivity for Colcrys’s gout indications expires July 30, 2012. The familial Mediterranean fever indication expires July 29, 2016, according to the FDA’s Web site.
Information about URL’s Colcrys patient assistance program and copayment coupons are available by visiting Colcrys.com or by calling 888-811-8423. Copayment coupons can be downloaded at the Web site.
Dr. Fudman said that he had no relevant disclosures. Dr. Morris disclosed that he is on the speakers’ bureau for Takeda, Eli Lilly, and Forest Pharmaceuticals. Dr. Mandell disclosed that he is an advisor to Takeda and URL Pharma, and was a clinical investigator for Savient Pharmaceuticals.
Rheumatologists Learning US; Studies Show Diagnostic Usefulness
MARINA DEL REY, CALIF. — Thinking about learning ultrasound? You are not alone, according to Dr. Kambiz Motamedi of the radiology department at the University of California, Los Angeles.
In Europe, rheumatologists have been using ultrasound (US) in their offices for years. The use of diagnostic US is catching on among U.S. rheumatologists, too. The American College of Rheumatology is offering its first US course this summer.
Studies, mostly done overseas, suggest its diagnostic value (Rheumatology 2009;48:1,092-7).
Uptake has been slower in the United States because MRI is more readily available and US has a steep learning curve, Dr. Motamedi said during a presentation and workshop on US.
But US is “becoming more and more popular,” among U.S. rheumatologists, he said.
A recent, small study suggests that even self-taught rheumatologists can become proficient ultrasonographers (Arthritis Care Res. 2010;62:155-60).
US is useful to look at superficial structures, including muscles, tendons, ligaments, nerves, and blood vessels, said Dr. Mihaela Taylor of the division of rheumatology at UCLA, who collaborated with Dr. Motamedi on the presentation and workshop.
It has higher soft-tissue resolution than does CT, Dr. Motamedi noted, although CT remains the standard for visualizing bone.
US also can visualize superficial joint structures, and pick up bone erosions, Baker's cysts, fluid behind the patella, and even meniscal tears, if they are in the periphery of the meniscus, acacording to Dr. Motamedi.
Joints can also be seen in motion, meaning that US can help guide joint injections.
“Ultrasound can't replace all that MRI [or CT] does, but it helps diagnose a lot of pathology,” Dr. Motamedi said.
Painless, noninvasive, relatively inexpensive, and free of radiation, it's also readily accepted by patients, Dr. Taylor said.
Although magnetic resonance imaging remains the standard for visualizing deep anatomical structures, such as those of the knee, US is a valid alternative, especially for claustrophobic patients and those with pacemakers or other MRI contraindications. The general concept of US is easy to grasp: Sound waves emitted from a probe are bounced off body structures. Their reflections back to the probe indicate the structure's density. Bone reflects as white. Less-dense structures—those that contain more water—reflect as darker shades.
What's closest to the probe (usually skin) appears at the top of the screen. What's farther away appears lower down.
Anything below bone cortex is artifact. Ultrasound does not penetrate bone, Dr. Taylor said.
Higher sound-wave frequencies mean better resolution but less penetration; lower frequencies penetrate more deeply but give less resolution.
It's helpful to keep the probe in motion and tilt it from side to side to help differentiate structures, Dr. Taylor said.
Information about the American Colege of Rheumatology's US course is at www.rheumatology.org/education/clinicalsymposia/mus.asp
Disclosures: Dr. Motamedi and Dr. Taylor each reported having no relevant financial conflicts.
MARINA DEL REY, CALIF. — Thinking about learning ultrasound? You are not alone, according to Dr. Kambiz Motamedi of the radiology department at the University of California, Los Angeles.
In Europe, rheumatologists have been using ultrasound (US) in their offices for years. The use of diagnostic US is catching on among U.S. rheumatologists, too. The American College of Rheumatology is offering its first US course this summer.
Studies, mostly done overseas, suggest its diagnostic value (Rheumatology 2009;48:1,092-7).
Uptake has been slower in the United States because MRI is more readily available and US has a steep learning curve, Dr. Motamedi said during a presentation and workshop on US.
But US is “becoming more and more popular,” among U.S. rheumatologists, he said.
A recent, small study suggests that even self-taught rheumatologists can become proficient ultrasonographers (Arthritis Care Res. 2010;62:155-60).
US is useful to look at superficial structures, including muscles, tendons, ligaments, nerves, and blood vessels, said Dr. Mihaela Taylor of the division of rheumatology at UCLA, who collaborated with Dr. Motamedi on the presentation and workshop.
It has higher soft-tissue resolution than does CT, Dr. Motamedi noted, although CT remains the standard for visualizing bone.
US also can visualize superficial joint structures, and pick up bone erosions, Baker's cysts, fluid behind the patella, and even meniscal tears, if they are in the periphery of the meniscus, acacording to Dr. Motamedi.
Joints can also be seen in motion, meaning that US can help guide joint injections.
“Ultrasound can't replace all that MRI [or CT] does, but it helps diagnose a lot of pathology,” Dr. Motamedi said.
Painless, noninvasive, relatively inexpensive, and free of radiation, it's also readily accepted by patients, Dr. Taylor said.
Although magnetic resonance imaging remains the standard for visualizing deep anatomical structures, such as those of the knee, US is a valid alternative, especially for claustrophobic patients and those with pacemakers or other MRI contraindications. The general concept of US is easy to grasp: Sound waves emitted from a probe are bounced off body structures. Their reflections back to the probe indicate the structure's density. Bone reflects as white. Less-dense structures—those that contain more water—reflect as darker shades.
What's closest to the probe (usually skin) appears at the top of the screen. What's farther away appears lower down.
Anything below bone cortex is artifact. Ultrasound does not penetrate bone, Dr. Taylor said.
Higher sound-wave frequencies mean better resolution but less penetration; lower frequencies penetrate more deeply but give less resolution.
It's helpful to keep the probe in motion and tilt it from side to side to help differentiate structures, Dr. Taylor said.
Information about the American Colege of Rheumatology's US course is at www.rheumatology.org/education/clinicalsymposia/mus.asp
Disclosures: Dr. Motamedi and Dr. Taylor each reported having no relevant financial conflicts.
MARINA DEL REY, CALIF. — Thinking about learning ultrasound? You are not alone, according to Dr. Kambiz Motamedi of the radiology department at the University of California, Los Angeles.
In Europe, rheumatologists have been using ultrasound (US) in their offices for years. The use of diagnostic US is catching on among U.S. rheumatologists, too. The American College of Rheumatology is offering its first US course this summer.
Studies, mostly done overseas, suggest its diagnostic value (Rheumatology 2009;48:1,092-7).
Uptake has been slower in the United States because MRI is more readily available and US has a steep learning curve, Dr. Motamedi said during a presentation and workshop on US.
But US is “becoming more and more popular,” among U.S. rheumatologists, he said.
A recent, small study suggests that even self-taught rheumatologists can become proficient ultrasonographers (Arthritis Care Res. 2010;62:155-60).
US is useful to look at superficial structures, including muscles, tendons, ligaments, nerves, and blood vessels, said Dr. Mihaela Taylor of the division of rheumatology at UCLA, who collaborated with Dr. Motamedi on the presentation and workshop.
It has higher soft-tissue resolution than does CT, Dr. Motamedi noted, although CT remains the standard for visualizing bone.
US also can visualize superficial joint structures, and pick up bone erosions, Baker's cysts, fluid behind the patella, and even meniscal tears, if they are in the periphery of the meniscus, acacording to Dr. Motamedi.
Joints can also be seen in motion, meaning that US can help guide joint injections.
“Ultrasound can't replace all that MRI [or CT] does, but it helps diagnose a lot of pathology,” Dr. Motamedi said.
Painless, noninvasive, relatively inexpensive, and free of radiation, it's also readily accepted by patients, Dr. Taylor said.
Although magnetic resonance imaging remains the standard for visualizing deep anatomical structures, such as those of the knee, US is a valid alternative, especially for claustrophobic patients and those with pacemakers or other MRI contraindications. The general concept of US is easy to grasp: Sound waves emitted from a probe are bounced off body structures. Their reflections back to the probe indicate the structure's density. Bone reflects as white. Less-dense structures—those that contain more water—reflect as darker shades.
What's closest to the probe (usually skin) appears at the top of the screen. What's farther away appears lower down.
Anything below bone cortex is artifact. Ultrasound does not penetrate bone, Dr. Taylor said.
Higher sound-wave frequencies mean better resolution but less penetration; lower frequencies penetrate more deeply but give less resolution.
It's helpful to keep the probe in motion and tilt it from side to side to help differentiate structures, Dr. Taylor said.
Information about the American Colege of Rheumatology's US course is at www.rheumatology.org/education/clinicalsymposia/mus.asp
Disclosures: Dr. Motamedi and Dr. Taylor each reported having no relevant financial conflicts.
Biomarkers Sought to Match RA Patients With Drug Therapy
MARINA DEL REY, CALIF. — Like rheumatologists everywhere, Dr. Joan M. Bathon, director of the Johns Hopkins Arthritis Center, sometimes has to rely on clinical trial and error to find the right medications for her patients.
It would be a tremendous help to be able to predict treatment response, and ways to do that may be coming to the clinic in the not-too-distant future, she said at rheumatology seminar sponsored by the University of California, Los Angeles.
Dr. Bathon said she usually starts RA patients on methotrexate. It generally takes 2-4 months to tell if the drug is working.
If methotrexate fails to bring RA under full control, Dr. Bathon said she will add a second agent. If a low to moderate level of disease activity remains, she'll add sulfasalazine, leflunomide, or hydroxychloroquine, depending on patient preference. With more severe residual disease activity, she's likely to add a tumor necrosis factor (TNF) inhibitor.
If one TNF inhibitor doesn't work, she'll try another. If the patient fails two TNF therapies, abatacept or rituximab are the next options, although rituximab is being used more cautiously these days since being linked with progressive multifocal leukoencephalopathy, she said.
Tocilizumab (Actemra)—which was approved by the Food and Drug Administration earlier this year as the first interleukin-6 receptor-inhibiting monoclonal antibody—is also an option if TNF inhibitors fail. The rheumatology community is still figuring out its place in the treatment paradigm, she said.
Research into biomarkers to predict treatment response may make that possible, Dr. Bathon said during her presentation.
While biomarkers for disease progression, treatment toxicity, and other aspects of RA care are also being sought, the biggest efforts are going into finding biomarkers for treatment response, according to Dr. Bathon.
Knowing, for instance, the particular molecular pathway—TNF, B cell, or T cell—that is most active in an individual RA patient would indicate if that patient would benefit from a TNF inhibitor such as etanercept, a B-cell depleter such as rituximab, or some other therapy.
Research into biomarkers for TNF inhibitor response is particularly active.
One study recently found that polymorphism in a TNF-alpha promoter gene (-308 G greater than A SNP) is associated with higher serum levels of TNF-alpha in RA patients, suggesting that the polymorphism is a weak predictor of response to anti-TNF therapy (Rheumatology Reports 2009 [doi:10.4081/rr.2009.e1]).
“If you have this, a TNF [inhibitor] may be the drug of choice,” Dr. Bathon said.
Another recent study measured an array of autoantibodies and cytokines in 93 patients from three different ethnic groups. A 24-biomarker signature was discovered that predicted good to excellent response, as well as lack of response, to etanercept (Arthritis Res. Ther. 2009;11:115).
Disclosures: Dr. Bathon said she had no relevant financial disclosures.
MARINA DEL REY, CALIF. — Like rheumatologists everywhere, Dr. Joan M. Bathon, director of the Johns Hopkins Arthritis Center, sometimes has to rely on clinical trial and error to find the right medications for her patients.
It would be a tremendous help to be able to predict treatment response, and ways to do that may be coming to the clinic in the not-too-distant future, she said at rheumatology seminar sponsored by the University of California, Los Angeles.
Dr. Bathon said she usually starts RA patients on methotrexate. It generally takes 2-4 months to tell if the drug is working.
If methotrexate fails to bring RA under full control, Dr. Bathon said she will add a second agent. If a low to moderate level of disease activity remains, she'll add sulfasalazine, leflunomide, or hydroxychloroquine, depending on patient preference. With more severe residual disease activity, she's likely to add a tumor necrosis factor (TNF) inhibitor.
If one TNF inhibitor doesn't work, she'll try another. If the patient fails two TNF therapies, abatacept or rituximab are the next options, although rituximab is being used more cautiously these days since being linked with progressive multifocal leukoencephalopathy, she said.
Tocilizumab (Actemra)—which was approved by the Food and Drug Administration earlier this year as the first interleukin-6 receptor-inhibiting monoclonal antibody—is also an option if TNF inhibitors fail. The rheumatology community is still figuring out its place in the treatment paradigm, she said.
Research into biomarkers to predict treatment response may make that possible, Dr. Bathon said during her presentation.
While biomarkers for disease progression, treatment toxicity, and other aspects of RA care are also being sought, the biggest efforts are going into finding biomarkers for treatment response, according to Dr. Bathon.
Knowing, for instance, the particular molecular pathway—TNF, B cell, or T cell—that is most active in an individual RA patient would indicate if that patient would benefit from a TNF inhibitor such as etanercept, a B-cell depleter such as rituximab, or some other therapy.
Research into biomarkers for TNF inhibitor response is particularly active.
One study recently found that polymorphism in a TNF-alpha promoter gene (-308 G greater than A SNP) is associated with higher serum levels of TNF-alpha in RA patients, suggesting that the polymorphism is a weak predictor of response to anti-TNF therapy (Rheumatology Reports 2009 [doi:10.4081/rr.2009.e1]).
“If you have this, a TNF [inhibitor] may be the drug of choice,” Dr. Bathon said.
Another recent study measured an array of autoantibodies and cytokines in 93 patients from three different ethnic groups. A 24-biomarker signature was discovered that predicted good to excellent response, as well as lack of response, to etanercept (Arthritis Res. Ther. 2009;11:115).
Disclosures: Dr. Bathon said she had no relevant financial disclosures.
MARINA DEL REY, CALIF. — Like rheumatologists everywhere, Dr. Joan M. Bathon, director of the Johns Hopkins Arthritis Center, sometimes has to rely on clinical trial and error to find the right medications for her patients.
It would be a tremendous help to be able to predict treatment response, and ways to do that may be coming to the clinic in the not-too-distant future, she said at rheumatology seminar sponsored by the University of California, Los Angeles.
Dr. Bathon said she usually starts RA patients on methotrexate. It generally takes 2-4 months to tell if the drug is working.
If methotrexate fails to bring RA under full control, Dr. Bathon said she will add a second agent. If a low to moderate level of disease activity remains, she'll add sulfasalazine, leflunomide, or hydroxychloroquine, depending on patient preference. With more severe residual disease activity, she's likely to add a tumor necrosis factor (TNF) inhibitor.
If one TNF inhibitor doesn't work, she'll try another. If the patient fails two TNF therapies, abatacept or rituximab are the next options, although rituximab is being used more cautiously these days since being linked with progressive multifocal leukoencephalopathy, she said.
Tocilizumab (Actemra)—which was approved by the Food and Drug Administration earlier this year as the first interleukin-6 receptor-inhibiting monoclonal antibody—is also an option if TNF inhibitors fail. The rheumatology community is still figuring out its place in the treatment paradigm, she said.
Research into biomarkers to predict treatment response may make that possible, Dr. Bathon said during her presentation.
While biomarkers for disease progression, treatment toxicity, and other aspects of RA care are also being sought, the biggest efforts are going into finding biomarkers for treatment response, according to Dr. Bathon.
Knowing, for instance, the particular molecular pathway—TNF, B cell, or T cell—that is most active in an individual RA patient would indicate if that patient would benefit from a TNF inhibitor such as etanercept, a B-cell depleter such as rituximab, or some other therapy.
Research into biomarkers for TNF inhibitor response is particularly active.
One study recently found that polymorphism in a TNF-alpha promoter gene (-308 G greater than A SNP) is associated with higher serum levels of TNF-alpha in RA patients, suggesting that the polymorphism is a weak predictor of response to anti-TNF therapy (Rheumatology Reports 2009 [doi:10.4081/rr.2009.e1]).
“If you have this, a TNF [inhibitor] may be the drug of choice,” Dr. Bathon said.
Another recent study measured an array of autoantibodies and cytokines in 93 patients from three different ethnic groups. A 24-biomarker signature was discovered that predicted good to excellent response, as well as lack of response, to etanercept (Arthritis Res. Ther. 2009;11:115).
Disclosures: Dr. Bathon said she had no relevant financial disclosures.
Resistance Exercise Protects Muscle Mass in Arthritis
MARINA DEL REY, CALIF. — Rheumatoid arthritis patients with well-controlled disease may benefit from performing fat-burning exercises accompanied by resistance training—such as weight lifting—to preserve or even build muscle mass, according to Dr. Joan M. Bathon.
A seemingly fit patient with well-controlled rheumatoid arthritis (RA) and a normal body mass index may still have excess body fat, elevated C-reactive protein (CRP) levels, and increased coronary artery disease risk, said Dr. Bathon, professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
RA's chronic inflammation can waste muscles, she explained at a rheumatology seminar sponsored by the University of California, Los Angeles. Appendicular fat correlates with disability, and visceral fat correlates with coronary artery disease, she said. When patients have well-controlled RA, their high CRP levels might be coming not from the inflamed joints, but rather from fat deposits, and might signal an increased risk of coronary artery disease.
Dr. Bathon and her colleagues performed anthropomorphic measurements and dual-energy x-ray absorptiometry (DXA) scanning to assess fat:muscle ratio in 72 men and 117 women with RA and moderate disability. A single CT image of the abdomen in the axial plane was used to assess the amount of visceral fat.
Women with RA and BMIs below 25 kg/m
Disclosures: Dr. Bathon said she had no relevant disclosures.
A seemingly fit RA patient with a normal body mass index may have a number of heart disease risk factors.
Source © Luc Ubaghs/iStockphoto.com
MARINA DEL REY, CALIF. — Rheumatoid arthritis patients with well-controlled disease may benefit from performing fat-burning exercises accompanied by resistance training—such as weight lifting—to preserve or even build muscle mass, according to Dr. Joan M. Bathon.
A seemingly fit patient with well-controlled rheumatoid arthritis (RA) and a normal body mass index may still have excess body fat, elevated C-reactive protein (CRP) levels, and increased coronary artery disease risk, said Dr. Bathon, professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
RA's chronic inflammation can waste muscles, she explained at a rheumatology seminar sponsored by the University of California, Los Angeles. Appendicular fat correlates with disability, and visceral fat correlates with coronary artery disease, she said. When patients have well-controlled RA, their high CRP levels might be coming not from the inflamed joints, but rather from fat deposits, and might signal an increased risk of coronary artery disease.
Dr. Bathon and her colleagues performed anthropomorphic measurements and dual-energy x-ray absorptiometry (DXA) scanning to assess fat:muscle ratio in 72 men and 117 women with RA and moderate disability. A single CT image of the abdomen in the axial plane was used to assess the amount of visceral fat.
Women with RA and BMIs below 25 kg/m
Disclosures: Dr. Bathon said she had no relevant disclosures.
A seemingly fit RA patient with a normal body mass index may have a number of heart disease risk factors.
Source © Luc Ubaghs/iStockphoto.com
MARINA DEL REY, CALIF. — Rheumatoid arthritis patients with well-controlled disease may benefit from performing fat-burning exercises accompanied by resistance training—such as weight lifting—to preserve or even build muscle mass, according to Dr. Joan M. Bathon.
A seemingly fit patient with well-controlled rheumatoid arthritis (RA) and a normal body mass index may still have excess body fat, elevated C-reactive protein (CRP) levels, and increased coronary artery disease risk, said Dr. Bathon, professor of medicine and director of the Johns Hopkins Arthritis Center in Baltimore.
RA's chronic inflammation can waste muscles, she explained at a rheumatology seminar sponsored by the University of California, Los Angeles. Appendicular fat correlates with disability, and visceral fat correlates with coronary artery disease, she said. When patients have well-controlled RA, their high CRP levels might be coming not from the inflamed joints, but rather from fat deposits, and might signal an increased risk of coronary artery disease.
Dr. Bathon and her colleagues performed anthropomorphic measurements and dual-energy x-ray absorptiometry (DXA) scanning to assess fat:muscle ratio in 72 men and 117 women with RA and moderate disability. A single CT image of the abdomen in the axial plane was used to assess the amount of visceral fat.
Women with RA and BMIs below 25 kg/m
Disclosures: Dr. Bathon said she had no relevant disclosures.
A seemingly fit RA patient with a normal body mass index may have a number of heart disease risk factors.
Source © Luc Ubaghs/iStockphoto.com
Cyclophosphamide Is of Most Benefit for Worst SSc-ILD
MARINA DEL REY, CALIF. — Cyclophosphamide is most likely to help scleroderma interstitial lung disease patients early in the course of their disease if they have extensive lung fibrosis, high Rodnan skin scores, and documented declines in forced vital capacity, according to findings from an unpublished subgroup analysis presented by Dr. Philip Clements, professor of medicine at the University of California, Los Angeles.
The news comes from ongoing analysis of the Scleroderma Lung Study, and is soon to be published, according to Dr. Clements. He was a lead investigator in the randomized, controlled clinical trial, which compared a 12-month course of cyclophosphamide given to 79 patients with systemic sclerosis interstitial lung disease (SSc-ILD) against placebo given to 79.
At 18 months, treated patients improved slightly over baseline forced vital capacity (FVC), whereas patients in the placebo group declined. The treatment difference between the two groups was 4.16% in favor of the cyclophosphamide patients (Am. J. Respir. Crit. Care. Med. 2007;176:1026-34).
The treatment differences “collapsed at 24 months, unfortunately,” Dr. Clements said.
However, in subsequent analysis, he and his colleagues identified a subset of patients who responded better to treatment: those with Rodnan skin thickness scores greater than 24 and fibrosis involving more than 50% of a lower-lung field.
Radiologists assessed the extent of lung fibrosis by visually inspecting high-resolution thoracic CT images. A software program has been developed to do the scoring, and should be available to clinicians within 3 years, Dr. Clements said.
For the subset of patients, FVC at 18 months in the treated group was 73% of predicted values for healthy, age-matched controls, but it was 63% of predicted values in the placebo group, although the treatment differences again collapsed at 24 months.
Even so, “the more fibrosis at baseline, the more likely [patients] are to respond,” Dr. Clements said. “Thick skin suggests their lungs are likely to respond to cyclophosphamide.” Additional analysis is planned to assess the clinical relevance of the findings, he said.
Patients from the Scleroderma Lung Study, which ran in 2000-2004, have been followed for an average of 8 years. So far, “cancer and death have not been associated with cyclophosphamide therapy,” Dr. Clements said.
Given the results, he said he treats SSc-ILD patients with cyclophosphamide if they have mild to moderate restrictive lung disease and are within 7 years of scleroderma diagnosis.
They must also have FVCs that are lower than 80% of predicted values, along with fibrosis involving 25% or more of any lung field accompanied by ground-glass opacifications and dyspnea involving difficulty in climbing two or three flights of stairs.
With those patients, “my treatment approach is similar to that of the National Institutes of Health's lupus nephritis protocol,” Dr. Clements said. The protocol includes the following:
▸ Pulse cyclophosphamide IV (500-750 mg/m
▸ Repeat pulmonary function tests every 3 months while patient is on cyclophosphamide.
▸ Upon completion of the infusion, switch to long-term mycophenolate mofetil (2-3 g/day orally).
Azathioprine (3-5 mg/kg per day) is an option if mycophenolate mofetil cannot be tolerated.
Mycophenolate mofetil is the subject of Scleroderma Lung Study II, which will compare a 2-year course of the drug in SSc-ILD patients against a 1-year course of cyclophosphamide, followed by placebo.
Mycophenolate mofetil “looks promising,” Dr. Clements said, based on several small, observational studies.
About 30 patients have enrolled in the trial since last November. “We need 150,” he said.
Information on the trial can be accessed on the Web at http://sls.med.ucla.educlinicaltrials.gov
Disclosures: Dr. Clements disclosed that he is a member of Gilead Sciences Inc.'s pulmonary hypertension advisory board.
MARINA DEL REY, CALIF. — Cyclophosphamide is most likely to help scleroderma interstitial lung disease patients early in the course of their disease if they have extensive lung fibrosis, high Rodnan skin scores, and documented declines in forced vital capacity, according to findings from an unpublished subgroup analysis presented by Dr. Philip Clements, professor of medicine at the University of California, Los Angeles.
The news comes from ongoing analysis of the Scleroderma Lung Study, and is soon to be published, according to Dr. Clements. He was a lead investigator in the randomized, controlled clinical trial, which compared a 12-month course of cyclophosphamide given to 79 patients with systemic sclerosis interstitial lung disease (SSc-ILD) against placebo given to 79.
At 18 months, treated patients improved slightly over baseline forced vital capacity (FVC), whereas patients in the placebo group declined. The treatment difference between the two groups was 4.16% in favor of the cyclophosphamide patients (Am. J. Respir. Crit. Care. Med. 2007;176:1026-34).
The treatment differences “collapsed at 24 months, unfortunately,” Dr. Clements said.
However, in subsequent analysis, he and his colleagues identified a subset of patients who responded better to treatment: those with Rodnan skin thickness scores greater than 24 and fibrosis involving more than 50% of a lower-lung field.
Radiologists assessed the extent of lung fibrosis by visually inspecting high-resolution thoracic CT images. A software program has been developed to do the scoring, and should be available to clinicians within 3 years, Dr. Clements said.
For the subset of patients, FVC at 18 months in the treated group was 73% of predicted values for healthy, age-matched controls, but it was 63% of predicted values in the placebo group, although the treatment differences again collapsed at 24 months.
Even so, “the more fibrosis at baseline, the more likely [patients] are to respond,” Dr. Clements said. “Thick skin suggests their lungs are likely to respond to cyclophosphamide.” Additional analysis is planned to assess the clinical relevance of the findings, he said.
Patients from the Scleroderma Lung Study, which ran in 2000-2004, have been followed for an average of 8 years. So far, “cancer and death have not been associated with cyclophosphamide therapy,” Dr. Clements said.
Given the results, he said he treats SSc-ILD patients with cyclophosphamide if they have mild to moderate restrictive lung disease and are within 7 years of scleroderma diagnosis.
They must also have FVCs that are lower than 80% of predicted values, along with fibrosis involving 25% or more of any lung field accompanied by ground-glass opacifications and dyspnea involving difficulty in climbing two or three flights of stairs.
With those patients, “my treatment approach is similar to that of the National Institutes of Health's lupus nephritis protocol,” Dr. Clements said. The protocol includes the following:
▸ Pulse cyclophosphamide IV (500-750 mg/m
▸ Repeat pulmonary function tests every 3 months while patient is on cyclophosphamide.
▸ Upon completion of the infusion, switch to long-term mycophenolate mofetil (2-3 g/day orally).
Azathioprine (3-5 mg/kg per day) is an option if mycophenolate mofetil cannot be tolerated.
Mycophenolate mofetil is the subject of Scleroderma Lung Study II, which will compare a 2-year course of the drug in SSc-ILD patients against a 1-year course of cyclophosphamide, followed by placebo.
Mycophenolate mofetil “looks promising,” Dr. Clements said, based on several small, observational studies.
About 30 patients have enrolled in the trial since last November. “We need 150,” he said.
Information on the trial can be accessed on the Web at http://sls.med.ucla.educlinicaltrials.gov
Disclosures: Dr. Clements disclosed that he is a member of Gilead Sciences Inc.'s pulmonary hypertension advisory board.
MARINA DEL REY, CALIF. — Cyclophosphamide is most likely to help scleroderma interstitial lung disease patients early in the course of their disease if they have extensive lung fibrosis, high Rodnan skin scores, and documented declines in forced vital capacity, according to findings from an unpublished subgroup analysis presented by Dr. Philip Clements, professor of medicine at the University of California, Los Angeles.
The news comes from ongoing analysis of the Scleroderma Lung Study, and is soon to be published, according to Dr. Clements. He was a lead investigator in the randomized, controlled clinical trial, which compared a 12-month course of cyclophosphamide given to 79 patients with systemic sclerosis interstitial lung disease (SSc-ILD) against placebo given to 79.
At 18 months, treated patients improved slightly over baseline forced vital capacity (FVC), whereas patients in the placebo group declined. The treatment difference between the two groups was 4.16% in favor of the cyclophosphamide patients (Am. J. Respir. Crit. Care. Med. 2007;176:1026-34).
The treatment differences “collapsed at 24 months, unfortunately,” Dr. Clements said.
However, in subsequent analysis, he and his colleagues identified a subset of patients who responded better to treatment: those with Rodnan skin thickness scores greater than 24 and fibrosis involving more than 50% of a lower-lung field.
Radiologists assessed the extent of lung fibrosis by visually inspecting high-resolution thoracic CT images. A software program has been developed to do the scoring, and should be available to clinicians within 3 years, Dr. Clements said.
For the subset of patients, FVC at 18 months in the treated group was 73% of predicted values for healthy, age-matched controls, but it was 63% of predicted values in the placebo group, although the treatment differences again collapsed at 24 months.
Even so, “the more fibrosis at baseline, the more likely [patients] are to respond,” Dr. Clements said. “Thick skin suggests their lungs are likely to respond to cyclophosphamide.” Additional analysis is planned to assess the clinical relevance of the findings, he said.
Patients from the Scleroderma Lung Study, which ran in 2000-2004, have been followed for an average of 8 years. So far, “cancer and death have not been associated with cyclophosphamide therapy,” Dr. Clements said.
Given the results, he said he treats SSc-ILD patients with cyclophosphamide if they have mild to moderate restrictive lung disease and are within 7 years of scleroderma diagnosis.
They must also have FVCs that are lower than 80% of predicted values, along with fibrosis involving 25% or more of any lung field accompanied by ground-glass opacifications and dyspnea involving difficulty in climbing two or three flights of stairs.
With those patients, “my treatment approach is similar to that of the National Institutes of Health's lupus nephritis protocol,” Dr. Clements said. The protocol includes the following:
▸ Pulse cyclophosphamide IV (500-750 mg/m
▸ Repeat pulmonary function tests every 3 months while patient is on cyclophosphamide.
▸ Upon completion of the infusion, switch to long-term mycophenolate mofetil (2-3 g/day orally).
Azathioprine (3-5 mg/kg per day) is an option if mycophenolate mofetil cannot be tolerated.
Mycophenolate mofetil is the subject of Scleroderma Lung Study II, which will compare a 2-year course of the drug in SSc-ILD patients against a 1-year course of cyclophosphamide, followed by placebo.
Mycophenolate mofetil “looks promising,” Dr. Clements said, based on several small, observational studies.
About 30 patients have enrolled in the trial since last November. “We need 150,” he said.
Information on the trial can be accessed on the Web at http://sls.med.ucla.educlinicaltrials.gov
Disclosures: Dr. Clements disclosed that he is a member of Gilead Sciences Inc.'s pulmonary hypertension advisory board.