User login
Diabetic Macular Edema: Is Your Patient Going Blind?
Diabetes mellitus (DM) affects about 347 million people worldwide, making it the new global epidemic.1 In the U.S alone, the number of adults with DM has more than tripled over the past 30 years. Now, almost 10% of the U.S. population has the disease and is at risk for serious systemic complications, including blindness.2
Diabetes is the leading cause of new cases of legal blindness in adults aged 18 to 74 years in the U.S. Diabetic retinopathy, seen as vascular changes in the retina related to DM, is found in almost half of all patients with DM.3 As the number of people with DM is expected to increase, so is the number of people affected with and blinded by diabetic retinopathy. Providers in both primary care and subspecialty settings have a critical role to play in the management and prevention of blindness in diabetic patients.
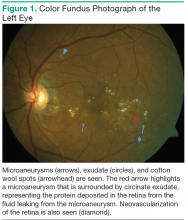
According to the Wisconsin Epidemiologic Study of Diabetic Retinopathy (WESDR), diabetic retinopathy will affect 99% of patients with type 1 DM (T1DM) and 60% of patients with type 2 DM (T2DM) after 20 years of having diabetes.4 Diabetic retinopathy is a result of the microvascular damage that occurs from diabetes. The most common findings seen on a dilated fundus exam in nonproliferative diabetic retinopathy are microaneurysms, intraretinal hemorrhages, hard exudates, and cotton wool spots (Figure 1). Cotton wool spots represent focal areas of retinal ischemia. Nonproliferative disease progresses to proliferative retinopathy when neovascularization develops.
One of the major causes of vision loss in the setting of DM is diabetic macular edema (DME). Between 4% and 7% of people with DM currently have DME.5,6 Diabetic macular edema is a result of the break down of the bloodretinal barrier, which is an extension of the blood-brain barrier. Hyperglycemia causes a disruption of the cellular tight junctions, pericyte loss, and thickening of the basement membrane. These changes cause weakness in the walls of the retinal blood vessels, allowing microaneurysms to form. Hyperglycemia also causes upregulation of the production of inflammatory markers such as vascular endothelial growth factor (VEGF), protein kinase C, prostaglandins, and cytokines, which increase retinal vascular permeability (Figure 2).7
Risk Factors for DME
Several studies have found the prevalence of DME to be higher in black (10.4%-15.6%) and Hispanic (18%) patients vs non-Hispanic white patients (6.3%-8.4%); Asian patients have the lowest prevalence of DME (5%).5,6,8,9 The modifiable risk factors for the development of DME include hyperglycemia, duration of disease, hypertension, and dyslipidemia. Patients who have had DM for a longer period of time (> 10 years) and those with a higher hemoglobin A1c (A1c ) are more likely to have DME.4,6 The WESDR showed that for each percentage point increase in baseline A1c, there was a 28% increase in the incidence of visual impairment at the 25-year follow-up.10 An A1c < 7% is recommended in patients, though some may benefit from an A1c < 6.5%.11
Epidemiologic studies have also found hypertension and dyslipidemia to be associated with an increased risk of DME.12-15 Both diseases lead to an increased vascular permeability, compounding the microvascular damage already present from DM. In hypertension, DME and retinal hemorrhages are thought to be influenced by the increased perfusion pressure in retinal vessels.16 For every 10 mm Hg increase in blood pressure over 160 mm Hg, the risk of DME increases by 25%.17 Dyslipidemia contributes to DME by damaging endothelial cells and causing increased vascular permeability through cytokine and VEGF upregulation.18
Other risk factors, such as nephropathy, anemia, sleep apnea, and thiazolidinedione (glitazone) use, may also affect the development of DME. Patients with microalbuminuria have a lower serum protein concentration and thus, a reduced plasma colloidal osmotic pressure. This decreased osmotic pressure allows fluid to exit the retinal blood vessels and causes DME.7 Serum osmolarity may also play a role in DME. Some patients were noted to have decreased DME after receiving hemodialysis.19 Retinal vascular permeability can be increased by ischemia caused by hypoxia from anemia or sleep apnea.7 Glitazones have been associated with an increased risk of developing DME, although the cause is unclear.7
Examination
The American Academy of Ophthalmology recommends annual diabetic retinopathy screening for all patients with DM. Screening exams should start at the time of diagnosis for patients with T2DM and at 5 years after diagnosis for patients with T1DM. Currently, patients without a history of diabetic retinopathy can be screened via an ophthalmologic exam or review of color fundus photographs, which can be taken by trained personnel in the primary care or subspecialty settings.
Unfortunately, only 60% of patients with DM are screened annually. It is important to emphasize to patients the importance of a screening eye exam. Many patients do not understand that diabetic retinopathy may be present even if they are not experiencing any changes in vision. The patient should be referred to an ophthalmologist immediately if he or she reports blurry vision, wavy lines, or dark spots in the vision, especially if those symptoms are acute. The goal of a screening program is early detection: to identify those patients who are at risk for vision loss from DM and to provide close follow-up and timely treatment. Any patient with a history of diabetic retinopathy should be followed at the interval recommended by the eye care provider.11
Patient history is an important part of the screening exam, including symptoms, duration of DM, A1c, medications, medical history (hypertension, nephropathy, dyslipidemia, obesity, pregnancy), and ocular history. If there is evidence of diabetic retinopathy or DME, recommendations for better systemic control of DM or its comorbidities can be made based on the patient history. During the screening exam, the patient’s visual acuity and intraocular pressure are measured. A basic examination of the anterior segment looking for neovascularization of the iris is also completed. Iris neovascularization is a sign of proliferative diabetic retinopathy that would indicate laser treatment, also known as panretinal photocoagulation, or intravitreal injection.
The patient’s pupils are dilated, which enables the eye care provider to examine the retina. Patients often dislike this portion of the examination, because the dilation drop causes their vision to be blurry for 4 to 6 hours. However, dilation ensures that the provider has a view of the entire retina and can detect early stages of diabetic retinopathy.
If the screening is taking place via color fundus photographs, a nonmydriatic fundus camera, which does not require dilation, can be used. The purpose of the screening examination is to assess changes that can lead to vision loss. Important features that must be detected if present are macular edema, extensive microvascular changes, vitreous hemorrhage, and neovascularization of the optic nerve, retina, or iris.11 It is important to remember that the diabetic screening examination does not take the place of a complete ophthalmologic examination for other ocular disease, such as glaucoma. The patient may need to schedule additional appointments with an eye care provider if other eye problems exist.
Imaging
Clinically significant macular edema (CSME) was first defined by the Early Treatment Diabetic Retinopathy Study (ETDRS) as macular edema that involves the center of vision, called the fovea, and can be visualized with clinical examination of the retina. The criteria for CSME are used to identify edema that is mostly likely to cause vision loss and to guide laser treatment.20 Today, adjunctive testing, such as optical coherence tomography (OCT) and fluorescein angiography (FA), aid in the earlier detection and diagnosis of DME. These imaging techniques are capable of detecting small amounts of macular edema that are vision-threatening but are not visible on exam. Center-involving DME is edema that involves the fovea; noncenter-involving DME is edema that does not involve the fovea but is found within the macula. Color and redfree fundus photographs, OCT, and FA are also used to guide treatment in patients with DME.
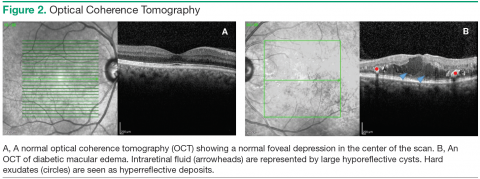
Color fundus photographs are helpful as a screening tool that identifies those patients who need to be seen by an eye care provider for a dilated fundus exam. Photographs are also helpful in documenting the changes in DME after treatment. Optical coherence tomography allows for high-resolution imaging of the retinal layers and objective measurement of the amount and location of DME. This information can be used to detect any change in DME between visits and evaluate the response to treatment. Further treatment decisions are frequently made based in part on the findings seen on OCT (Figure 2).
Fluorescein angiography is an imaging test that uses an IV dye called fluorescein to detect areas of retinal nonperfusion, edema, and neovascularization. Fluorescein is injected into a peripheral vein (frequently in the antecubital area or in the hand). Photographs are then taken
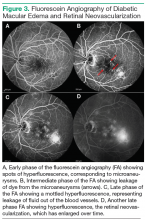
using special filters that allow only the wavelength corresponding to the fluorescein dye to be visible. The fluorescein can be seen filling the retinal arteries within 20 seconds of the peripheral injection. Photographs are usually taken intermittently for 15 minutes. As time passes, the dye will leak out of any blood vessels that have increased vascular permeability and highlight any microaneurysms, because the dye pools in the outpouching of the blood vessel. Leakage of the dye out of the vessels can be seen as an increase in fluorescence, or whitening, outside of the blood vessels in the photograph. This leakage leads to the accumulation of fluid in the retina, causing DME. Neovascularization is also very permeable, and areas of neovascularization, an indicator of proliferative diabetic retinopathy, are apparent on FA as spots of intense hyperfluorescence (Figure 3).
Treatment
In 1985, results from ETDRS revolutionized the treatment of DME. The study showed that by applying laser burns to leaking microaneurysms or in a grid pattern over an area of diffuse edema, severe vision loss could be reduced by 50%. In the past few years, the role of laser treatment has shifted so that it is now indicated for the treatment of noncenter-involving DME. The impetus for this change was the development of anti-VEGF therapy, which is now the first-line therapy for centerinvolving DME.
As aforementioned, VEGF causes increased vascular permeability and breakdown of the blood-retinal barrier. Patients with DME have been shown to have increased levels of VEGF in the vitreous when compared with nondiabetic controls.21 There are now 3 anti-VEGF agents that are commonly used in clinical practice for the treatment of DME: ranibizumab, aflibercept, and bevacizumab. Ranibizumab is an antibody fragment targeted against VEGF that is FDA approved for use in patients with DME. The Diabetic Retinopathy Clinical Research Network Protocol I showed that treatment with ranibizumab, paired with deferred laser treatment, results in greater visual improvement than does prompt laser treatment alone.22 Treatment with aflibercept is a recombinant fusion protein of VEGF receptors. It was shown to be superior in terms of visual improvement when compared with laser treatment.23 Bevacizumab is a full-length antibody that is more affordable than other anti-VEGF medications and is often used off label for the treatment of DME. All of the anti-VEGF therapies are intravitreal injections. After topical anesthesia, the medication is injected through the sclera into the vitreous cavity in the outpatient clinic setting.
A significant disadvantage of the anti-VEGF therapies is that many patients need monthly injections, especially in the first year of treatment, necessitating many office visits, which can decrease adherence. In some patients, the edema may not respond to anti-VEGF therapy. In these cases, steroid therapy may be helpful to suppress the inflammatory pathways that are independent of VEGF. Intravitreal triamcinolone in combination with laser treatment has been shown to be as effective as ranibizumab in a small group of patients.24 An intravitreal dexamethasone implant, which has a treatment effect lasting for 3 months, was also shown to improve visual acuity over sham treatment in patients with DME.25 Most recently, an intravitreal fluocinolone implant that lasts 3 years was approved by the FDA for treatment of DME.26 A significant benefit of the steroid implants is the long duration of treatment effect compared with that of the anti-VEGF injections. However, steroid therapy is associated with the development of cataracts and glaucoma, the rates of which are increased when treatment is prolonged. Because of these adverse effects, steroids are currently used as second-line or third-line treatment in DME. Retinal surgery may be indicated if there is vitreomacular traction that is exacerbating the DME. A vitrectomy is performed to remove the vitreous and relieve any adhesion to the surface of the retina.
Conclusion
Despite the new ophthalmic treatment modalities, it is important to remember that DME is a chronic condition that will require long-term follow-up. Many patients will not experience complete resolution of DME with a single therapy alone. Control of systemic risk factors, including blood sugar with a goal of A1c < 7%, blood pressure, and cholesterol, remains the key to a successful treatment program. Primary care physicians, endocrinologists, diabetologists, optometrists, comprehensive ophthalmologists, retina specialists, and patients must work together to create an individualized treatment regimen that will optimize the patient’s vision by preventing blindness and improving his/her quality of life for years to come.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. Danaei G, Finucane MM, Lu Y, et al; Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Blood Glucose). National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet. 2011;378(9785):31-40.
2. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimate of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014.
3. National Institutes of Health. Diabetic retinopathy: causes and risk factors. NIH Senior Health Website. http://nihseniorhealth.gov/diabeticretinopathy/causesandriskfactors/01.html. Updated February 2015. Accessed September 3, 2015.
4. Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. XV. The long-term incidence of macular edema. Ophthalmology. 1995;102(1):7-16.
5. Yau JW, Rogers SL, Kawasaki R, et al; Meta-Analysis for Eye Disease (META-EYE) Study Group. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556-564.
6. Varma R, Bressler NM, Doan QV, et al. Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol. 2014;132(11):1334-1340.
7. Diep TM, Tsui I. Risk factors associated with diabetic macular edema. Diabetes Res Clin Pract. 2013;100(3):298-305.
8. Varma R, Choudhury F, Klein R, Chung J, Torres M, Azen SP; Los Angeles Latino Eye Study Group. Four-year incidence and progression of diabetic retinopathy and macular edema: the Los Angeles Latino Eye Study. Am J Ophthalmol. 2010;149(5):752-761.e1-e3.
9. Emanuele N, Moritz T, Klein R, et al; Veterans Affairs Diabetes Trial Study Group. Ethnicity, race, and clinically significant macular edema in the Veterans Affairs Diabetes Trial (VADT). Diabetes Res Clin Pract. 2009;86(2):104-110.
10. Klein R, Lee KE, Gangnon RE, Klein BE. The 25-year incidence of visual impairment in type 1 diabetes mellitus: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Ophthalmology. 2010;117(1):63-70.
11. American Academy of Ophthalmology Retina/Vitreous Panel. Preferred Practice Pattern: Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; 2014.
12. Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. IV. Diabetic macular edema. Ophthalmology. 1984;91(12):1464-1474.
13. Klein R, Knudtson MD, Lee KE, Gangnon R, Klein BE. The Wisconsin Epidemiologic Study of Diabetic Retinopathy XXIII: the twenty-five-year incidence of macular edema in persons with type 1 diabetes. Ophthalmology. 2009;116(3):497-503.
14. Chew EY. Diabetic retinopathy and lipid abnormalities. Curr Opin Ophthalmol. 1997;8(3):59-62.
15. Klein R, Sharrett AR, Klein BE, et al; ARIC Group. The association of atherosclerosis, vascular risk factors, and retinopathy in adults with diabetes: the atherosclerosis risk in communities study. Ophthalmology. 2002;109(7):1225-1234.
16. Haefliger IO, Meyer P, Flammer J, Lüscher TF. The vascular endothelium as a regulator of the ocular circulation: a new concept in ophthalmology? Surv Ophthalmol. 1994;39(2):123-132.
17. Lopes de Faria JM, Jalkh AE, Trempe CL, McMeel JW. Diabetic macular edema: risk factors and concomitants. Acta Ophthalmol Scand. 1999;77(2):170-175.
18. Langeler EG, Snelting-Havinga I, van Hinsbergh VW. Passage of low density lipoproteins through monolayers of human arterial endothelial cells. Effects of vasoactive substances in an in vitro model. Arteriosclerosis. 1989;9(4):550-559.
19. Theodossiadis PG, Theodoropoulou S, Neamonitou G, et al. Hemodialysisinduced alterations in macular thickness measured by optical coherence tomography in diabetic patients with end-stage renal disease. Ophthalmologica. 2012;227(2):90-94.
20. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985;103(12):1796-1806.
21. Funatsu H, Noma H, Mimura T, Eguchi S, Hori S. Association of vitreous inflammatory factors with diabetic macular edema. Ophthalmology. 2009;116(1):73-79.
22. Diabetic Retinopathy Clinical Research Network; Elman MJ, Qin H, Aiello LP, et al. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment: three-year randomized trial results. Ophthalmology. 2012;119(11):2312-2318.
23. Korobelnik JF, Do DV, Schmidt-Erfurth U, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121(11):2247-2254.
24. Diabetic Retinopathy Clinical Research Network; Elman MJ, Aiello LP, Beck RW, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117(6):1064-1077.
25. Boyer DS, Yoon YH, Belfort R Jr, et al; Ozurdex MEAD Study Group. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology. 2014;121(10):1904-1914.
26. Campochiaro PA, Brown DM, Pearson A, et al; FAME Study Group. Long-term benefit of sustained-delivery fluocinolone acetonide vitreous inserts for diabetic macular edema. Ophthalmology. 2011;118(4):626-635.e2.
Diabetes mellitus (DM) affects about 347 million people worldwide, making it the new global epidemic.1 In the U.S alone, the number of adults with DM has more than tripled over the past 30 years. Now, almost 10% of the U.S. population has the disease and is at risk for serious systemic complications, including blindness.2
Diabetes is the leading cause of new cases of legal blindness in adults aged 18 to 74 years in the U.S. Diabetic retinopathy, seen as vascular changes in the retina related to DM, is found in almost half of all patients with DM.3 As the number of people with DM is expected to increase, so is the number of people affected with and blinded by diabetic retinopathy. Providers in both primary care and subspecialty settings have a critical role to play in the management and prevention of blindness in diabetic patients.
According to the Wisconsin Epidemiologic Study of Diabetic Retinopathy (WESDR), diabetic retinopathy will affect 99% of patients with type 1 DM (T1DM) and 60% of patients with type 2 DM (T2DM) after 20 years of having diabetes.4 Diabetic retinopathy is a result of the microvascular damage that occurs from diabetes. The most common findings seen on a dilated fundus exam in nonproliferative diabetic retinopathy are microaneurysms, intraretinal hemorrhages, hard exudates, and cotton wool spots (Figure 1). Cotton wool spots represent focal areas of retinal ischemia. Nonproliferative disease progresses to proliferative retinopathy when neovascularization develops.
One of the major causes of vision loss in the setting of DM is diabetic macular edema (DME). Between 4% and 7% of people with DM currently have DME.5,6 Diabetic macular edema is a result of the break down of the bloodretinal barrier, which is an extension of the blood-brain barrier. Hyperglycemia causes a disruption of the cellular tight junctions, pericyte loss, and thickening of the basement membrane. These changes cause weakness in the walls of the retinal blood vessels, allowing microaneurysms to form. Hyperglycemia also causes upregulation of the production of inflammatory markers such as vascular endothelial growth factor (VEGF), protein kinase C, prostaglandins, and cytokines, which increase retinal vascular permeability (Figure 2).7
Risk Factors for DME
Several studies have found the prevalence of DME to be higher in black (10.4%-15.6%) and Hispanic (18%) patients vs non-Hispanic white patients (6.3%-8.4%); Asian patients have the lowest prevalence of DME (5%).5,6,8,9 The modifiable risk factors for the development of DME include hyperglycemia, duration of disease, hypertension, and dyslipidemia. Patients who have had DM for a longer period of time (> 10 years) and those with a higher hemoglobin A1c (A1c ) are more likely to have DME.4,6 The WESDR showed that for each percentage point increase in baseline A1c, there was a 28% increase in the incidence of visual impairment at the 25-year follow-up.10 An A1c < 7% is recommended in patients, though some may benefit from an A1c < 6.5%.11
Epidemiologic studies have also found hypertension and dyslipidemia to be associated with an increased risk of DME.12-15 Both diseases lead to an increased vascular permeability, compounding the microvascular damage already present from DM. In hypertension, DME and retinal hemorrhages are thought to be influenced by the increased perfusion pressure in retinal vessels.16 For every 10 mm Hg increase in blood pressure over 160 mm Hg, the risk of DME increases by 25%.17 Dyslipidemia contributes to DME by damaging endothelial cells and causing increased vascular permeability through cytokine and VEGF upregulation.18
Other risk factors, such as nephropathy, anemia, sleep apnea, and thiazolidinedione (glitazone) use, may also affect the development of DME. Patients with microalbuminuria have a lower serum protein concentration and thus, a reduced plasma colloidal osmotic pressure. This decreased osmotic pressure allows fluid to exit the retinal blood vessels and causes DME.7 Serum osmolarity may also play a role in DME. Some patients were noted to have decreased DME after receiving hemodialysis.19 Retinal vascular permeability can be increased by ischemia caused by hypoxia from anemia or sleep apnea.7 Glitazones have been associated with an increased risk of developing DME, although the cause is unclear.7
Examination
The American Academy of Ophthalmology recommends annual diabetic retinopathy screening for all patients with DM. Screening exams should start at the time of diagnosis for patients with T2DM and at 5 years after diagnosis for patients with T1DM. Currently, patients without a history of diabetic retinopathy can be screened via an ophthalmologic exam or review of color fundus photographs, which can be taken by trained personnel in the primary care or subspecialty settings.
Unfortunately, only 60% of patients with DM are screened annually. It is important to emphasize to patients the importance of a screening eye exam. Many patients do not understand that diabetic retinopathy may be present even if they are not experiencing any changes in vision. The patient should be referred to an ophthalmologist immediately if he or she reports blurry vision, wavy lines, or dark spots in the vision, especially if those symptoms are acute. The goal of a screening program is early detection: to identify those patients who are at risk for vision loss from DM and to provide close follow-up and timely treatment. Any patient with a history of diabetic retinopathy should be followed at the interval recommended by the eye care provider.11
Patient history is an important part of the screening exam, including symptoms, duration of DM, A1c, medications, medical history (hypertension, nephropathy, dyslipidemia, obesity, pregnancy), and ocular history. If there is evidence of diabetic retinopathy or DME, recommendations for better systemic control of DM or its comorbidities can be made based on the patient history. During the screening exam, the patient’s visual acuity and intraocular pressure are measured. A basic examination of the anterior segment looking for neovascularization of the iris is also completed. Iris neovascularization is a sign of proliferative diabetic retinopathy that would indicate laser treatment, also known as panretinal photocoagulation, or intravitreal injection.
The patient’s pupils are dilated, which enables the eye care provider to examine the retina. Patients often dislike this portion of the examination, because the dilation drop causes their vision to be blurry for 4 to 6 hours. However, dilation ensures that the provider has a view of the entire retina and can detect early stages of diabetic retinopathy.
If the screening is taking place via color fundus photographs, a nonmydriatic fundus camera, which does not require dilation, can be used. The purpose of the screening examination is to assess changes that can lead to vision loss. Important features that must be detected if present are macular edema, extensive microvascular changes, vitreous hemorrhage, and neovascularization of the optic nerve, retina, or iris.11 It is important to remember that the diabetic screening examination does not take the place of a complete ophthalmologic examination for other ocular disease, such as glaucoma. The patient may need to schedule additional appointments with an eye care provider if other eye problems exist.
Imaging
Clinically significant macular edema (CSME) was first defined by the Early Treatment Diabetic Retinopathy Study (ETDRS) as macular edema that involves the center of vision, called the fovea, and can be visualized with clinical examination of the retina. The criteria for CSME are used to identify edema that is mostly likely to cause vision loss and to guide laser treatment.20 Today, adjunctive testing, such as optical coherence tomography (OCT) and fluorescein angiography (FA), aid in the earlier detection and diagnosis of DME. These imaging techniques are capable of detecting small amounts of macular edema that are vision-threatening but are not visible on exam. Center-involving DME is edema that involves the fovea; noncenter-involving DME is edema that does not involve the fovea but is found within the macula. Color and redfree fundus photographs, OCT, and FA are also used to guide treatment in patients with DME.
Color fundus photographs are helpful as a screening tool that identifies those patients who need to be seen by an eye care provider for a dilated fundus exam. Photographs are also helpful in documenting the changes in DME after treatment. Optical coherence tomography allows for high-resolution imaging of the retinal layers and objective measurement of the amount and location of DME. This information can be used to detect any change in DME between visits and evaluate the response to treatment. Further treatment decisions are frequently made based in part on the findings seen on OCT (Figure 2).
Fluorescein angiography is an imaging test that uses an IV dye called fluorescein to detect areas of retinal nonperfusion, edema, and neovascularization. Fluorescein is injected into a peripheral vein (frequently in the antecubital area or in the hand). Photographs are then taken
using special filters that allow only the wavelength corresponding to the fluorescein dye to be visible. The fluorescein can be seen filling the retinal arteries within 20 seconds of the peripheral injection. Photographs are usually taken intermittently for 15 minutes. As time passes, the dye will leak out of any blood vessels that have increased vascular permeability and highlight any microaneurysms, because the dye pools in the outpouching of the blood vessel. Leakage of the dye out of the vessels can be seen as an increase in fluorescence, or whitening, outside of the blood vessels in the photograph. This leakage leads to the accumulation of fluid in the retina, causing DME. Neovascularization is also very permeable, and areas of neovascularization, an indicator of proliferative diabetic retinopathy, are apparent on FA as spots of intense hyperfluorescence (Figure 3).
Treatment
In 1985, results from ETDRS revolutionized the treatment of DME. The study showed that by applying laser burns to leaking microaneurysms or in a grid pattern over an area of diffuse edema, severe vision loss could be reduced by 50%. In the past few years, the role of laser treatment has shifted so that it is now indicated for the treatment of noncenter-involving DME. The impetus for this change was the development of anti-VEGF therapy, which is now the first-line therapy for centerinvolving DME.
As aforementioned, VEGF causes increased vascular permeability and breakdown of the blood-retinal barrier. Patients with DME have been shown to have increased levels of VEGF in the vitreous when compared with nondiabetic controls.21 There are now 3 anti-VEGF agents that are commonly used in clinical practice for the treatment of DME: ranibizumab, aflibercept, and bevacizumab. Ranibizumab is an antibody fragment targeted against VEGF that is FDA approved for use in patients with DME. The Diabetic Retinopathy Clinical Research Network Protocol I showed that treatment with ranibizumab, paired with deferred laser treatment, results in greater visual improvement than does prompt laser treatment alone.22 Treatment with aflibercept is a recombinant fusion protein of VEGF receptors. It was shown to be superior in terms of visual improvement when compared with laser treatment.23 Bevacizumab is a full-length antibody that is more affordable than other anti-VEGF medications and is often used off label for the treatment of DME. All of the anti-VEGF therapies are intravitreal injections. After topical anesthesia, the medication is injected through the sclera into the vitreous cavity in the outpatient clinic setting.
A significant disadvantage of the anti-VEGF therapies is that many patients need monthly injections, especially in the first year of treatment, necessitating many office visits, which can decrease adherence. In some patients, the edema may not respond to anti-VEGF therapy. In these cases, steroid therapy may be helpful to suppress the inflammatory pathways that are independent of VEGF. Intravitreal triamcinolone in combination with laser treatment has been shown to be as effective as ranibizumab in a small group of patients.24 An intravitreal dexamethasone implant, which has a treatment effect lasting for 3 months, was also shown to improve visual acuity over sham treatment in patients with DME.25 Most recently, an intravitreal fluocinolone implant that lasts 3 years was approved by the FDA for treatment of DME.26 A significant benefit of the steroid implants is the long duration of treatment effect compared with that of the anti-VEGF injections. However, steroid therapy is associated with the development of cataracts and glaucoma, the rates of which are increased when treatment is prolonged. Because of these adverse effects, steroids are currently used as second-line or third-line treatment in DME. Retinal surgery may be indicated if there is vitreomacular traction that is exacerbating the DME. A vitrectomy is performed to remove the vitreous and relieve any adhesion to the surface of the retina.
Conclusion
Despite the new ophthalmic treatment modalities, it is important to remember that DME is a chronic condition that will require long-term follow-up. Many patients will not experience complete resolution of DME with a single therapy alone. Control of systemic risk factors, including blood sugar with a goal of A1c < 7%, blood pressure, and cholesterol, remains the key to a successful treatment program. Primary care physicians, endocrinologists, diabetologists, optometrists, comprehensive ophthalmologists, retina specialists, and patients must work together to create an individualized treatment regimen that will optimize the patient’s vision by preventing blindness and improving his/her quality of life for years to come.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
Diabetes mellitus (DM) affects about 347 million people worldwide, making it the new global epidemic.1 In the U.S alone, the number of adults with DM has more than tripled over the past 30 years. Now, almost 10% of the U.S. population has the disease and is at risk for serious systemic complications, including blindness.2
Diabetes is the leading cause of new cases of legal blindness in adults aged 18 to 74 years in the U.S. Diabetic retinopathy, seen as vascular changes in the retina related to DM, is found in almost half of all patients with DM.3 As the number of people with DM is expected to increase, so is the number of people affected with and blinded by diabetic retinopathy. Providers in both primary care and subspecialty settings have a critical role to play in the management and prevention of blindness in diabetic patients.
According to the Wisconsin Epidemiologic Study of Diabetic Retinopathy (WESDR), diabetic retinopathy will affect 99% of patients with type 1 DM (T1DM) and 60% of patients with type 2 DM (T2DM) after 20 years of having diabetes.4 Diabetic retinopathy is a result of the microvascular damage that occurs from diabetes. The most common findings seen on a dilated fundus exam in nonproliferative diabetic retinopathy are microaneurysms, intraretinal hemorrhages, hard exudates, and cotton wool spots (Figure 1). Cotton wool spots represent focal areas of retinal ischemia. Nonproliferative disease progresses to proliferative retinopathy when neovascularization develops.
One of the major causes of vision loss in the setting of DM is diabetic macular edema (DME). Between 4% and 7% of people with DM currently have DME.5,6 Diabetic macular edema is a result of the break down of the bloodretinal barrier, which is an extension of the blood-brain barrier. Hyperglycemia causes a disruption of the cellular tight junctions, pericyte loss, and thickening of the basement membrane. These changes cause weakness in the walls of the retinal blood vessels, allowing microaneurysms to form. Hyperglycemia also causes upregulation of the production of inflammatory markers such as vascular endothelial growth factor (VEGF), protein kinase C, prostaglandins, and cytokines, which increase retinal vascular permeability (Figure 2).7
Risk Factors for DME
Several studies have found the prevalence of DME to be higher in black (10.4%-15.6%) and Hispanic (18%) patients vs non-Hispanic white patients (6.3%-8.4%); Asian patients have the lowest prevalence of DME (5%).5,6,8,9 The modifiable risk factors for the development of DME include hyperglycemia, duration of disease, hypertension, and dyslipidemia. Patients who have had DM for a longer period of time (> 10 years) and those with a higher hemoglobin A1c (A1c ) are more likely to have DME.4,6 The WESDR showed that for each percentage point increase in baseline A1c, there was a 28% increase in the incidence of visual impairment at the 25-year follow-up.10 An A1c < 7% is recommended in patients, though some may benefit from an A1c < 6.5%.11
Epidemiologic studies have also found hypertension and dyslipidemia to be associated with an increased risk of DME.12-15 Both diseases lead to an increased vascular permeability, compounding the microvascular damage already present from DM. In hypertension, DME and retinal hemorrhages are thought to be influenced by the increased perfusion pressure in retinal vessels.16 For every 10 mm Hg increase in blood pressure over 160 mm Hg, the risk of DME increases by 25%.17 Dyslipidemia contributes to DME by damaging endothelial cells and causing increased vascular permeability through cytokine and VEGF upregulation.18
Other risk factors, such as nephropathy, anemia, sleep apnea, and thiazolidinedione (glitazone) use, may also affect the development of DME. Patients with microalbuminuria have a lower serum protein concentration and thus, a reduced plasma colloidal osmotic pressure. This decreased osmotic pressure allows fluid to exit the retinal blood vessels and causes DME.7 Serum osmolarity may also play a role in DME. Some patients were noted to have decreased DME after receiving hemodialysis.19 Retinal vascular permeability can be increased by ischemia caused by hypoxia from anemia or sleep apnea.7 Glitazones have been associated with an increased risk of developing DME, although the cause is unclear.7
Examination
The American Academy of Ophthalmology recommends annual diabetic retinopathy screening for all patients with DM. Screening exams should start at the time of diagnosis for patients with T2DM and at 5 years after diagnosis for patients with T1DM. Currently, patients without a history of diabetic retinopathy can be screened via an ophthalmologic exam or review of color fundus photographs, which can be taken by trained personnel in the primary care or subspecialty settings.
Unfortunately, only 60% of patients with DM are screened annually. It is important to emphasize to patients the importance of a screening eye exam. Many patients do not understand that diabetic retinopathy may be present even if they are not experiencing any changes in vision. The patient should be referred to an ophthalmologist immediately if he or she reports blurry vision, wavy lines, or dark spots in the vision, especially if those symptoms are acute. The goal of a screening program is early detection: to identify those patients who are at risk for vision loss from DM and to provide close follow-up and timely treatment. Any patient with a history of diabetic retinopathy should be followed at the interval recommended by the eye care provider.11
Patient history is an important part of the screening exam, including symptoms, duration of DM, A1c, medications, medical history (hypertension, nephropathy, dyslipidemia, obesity, pregnancy), and ocular history. If there is evidence of diabetic retinopathy or DME, recommendations for better systemic control of DM or its comorbidities can be made based on the patient history. During the screening exam, the patient’s visual acuity and intraocular pressure are measured. A basic examination of the anterior segment looking for neovascularization of the iris is also completed. Iris neovascularization is a sign of proliferative diabetic retinopathy that would indicate laser treatment, also known as panretinal photocoagulation, or intravitreal injection.
The patient’s pupils are dilated, which enables the eye care provider to examine the retina. Patients often dislike this portion of the examination, because the dilation drop causes their vision to be blurry for 4 to 6 hours. However, dilation ensures that the provider has a view of the entire retina and can detect early stages of diabetic retinopathy.
If the screening is taking place via color fundus photographs, a nonmydriatic fundus camera, which does not require dilation, can be used. The purpose of the screening examination is to assess changes that can lead to vision loss. Important features that must be detected if present are macular edema, extensive microvascular changes, vitreous hemorrhage, and neovascularization of the optic nerve, retina, or iris.11 It is important to remember that the diabetic screening examination does not take the place of a complete ophthalmologic examination for other ocular disease, such as glaucoma. The patient may need to schedule additional appointments with an eye care provider if other eye problems exist.
Imaging
Clinically significant macular edema (CSME) was first defined by the Early Treatment Diabetic Retinopathy Study (ETDRS) as macular edema that involves the center of vision, called the fovea, and can be visualized with clinical examination of the retina. The criteria for CSME are used to identify edema that is mostly likely to cause vision loss and to guide laser treatment.20 Today, adjunctive testing, such as optical coherence tomography (OCT) and fluorescein angiography (FA), aid in the earlier detection and diagnosis of DME. These imaging techniques are capable of detecting small amounts of macular edema that are vision-threatening but are not visible on exam. Center-involving DME is edema that involves the fovea; noncenter-involving DME is edema that does not involve the fovea but is found within the macula. Color and redfree fundus photographs, OCT, and FA are also used to guide treatment in patients with DME.
Color fundus photographs are helpful as a screening tool that identifies those patients who need to be seen by an eye care provider for a dilated fundus exam. Photographs are also helpful in documenting the changes in DME after treatment. Optical coherence tomography allows for high-resolution imaging of the retinal layers and objective measurement of the amount and location of DME. This information can be used to detect any change in DME between visits and evaluate the response to treatment. Further treatment decisions are frequently made based in part on the findings seen on OCT (Figure 2).
Fluorescein angiography is an imaging test that uses an IV dye called fluorescein to detect areas of retinal nonperfusion, edema, and neovascularization. Fluorescein is injected into a peripheral vein (frequently in the antecubital area or in the hand). Photographs are then taken
using special filters that allow only the wavelength corresponding to the fluorescein dye to be visible. The fluorescein can be seen filling the retinal arteries within 20 seconds of the peripheral injection. Photographs are usually taken intermittently for 15 minutes. As time passes, the dye will leak out of any blood vessels that have increased vascular permeability and highlight any microaneurysms, because the dye pools in the outpouching of the blood vessel. Leakage of the dye out of the vessels can be seen as an increase in fluorescence, or whitening, outside of the blood vessels in the photograph. This leakage leads to the accumulation of fluid in the retina, causing DME. Neovascularization is also very permeable, and areas of neovascularization, an indicator of proliferative diabetic retinopathy, are apparent on FA as spots of intense hyperfluorescence (Figure 3).
Treatment
In 1985, results from ETDRS revolutionized the treatment of DME. The study showed that by applying laser burns to leaking microaneurysms or in a grid pattern over an area of diffuse edema, severe vision loss could be reduced by 50%. In the past few years, the role of laser treatment has shifted so that it is now indicated for the treatment of noncenter-involving DME. The impetus for this change was the development of anti-VEGF therapy, which is now the first-line therapy for centerinvolving DME.
As aforementioned, VEGF causes increased vascular permeability and breakdown of the blood-retinal barrier. Patients with DME have been shown to have increased levels of VEGF in the vitreous when compared with nondiabetic controls.21 There are now 3 anti-VEGF agents that are commonly used in clinical practice for the treatment of DME: ranibizumab, aflibercept, and bevacizumab. Ranibizumab is an antibody fragment targeted against VEGF that is FDA approved for use in patients with DME. The Diabetic Retinopathy Clinical Research Network Protocol I showed that treatment with ranibizumab, paired with deferred laser treatment, results in greater visual improvement than does prompt laser treatment alone.22 Treatment with aflibercept is a recombinant fusion protein of VEGF receptors. It was shown to be superior in terms of visual improvement when compared with laser treatment.23 Bevacizumab is a full-length antibody that is more affordable than other anti-VEGF medications and is often used off label for the treatment of DME. All of the anti-VEGF therapies are intravitreal injections. After topical anesthesia, the medication is injected through the sclera into the vitreous cavity in the outpatient clinic setting.
A significant disadvantage of the anti-VEGF therapies is that many patients need monthly injections, especially in the first year of treatment, necessitating many office visits, which can decrease adherence. In some patients, the edema may not respond to anti-VEGF therapy. In these cases, steroid therapy may be helpful to suppress the inflammatory pathways that are independent of VEGF. Intravitreal triamcinolone in combination with laser treatment has been shown to be as effective as ranibizumab in a small group of patients.24 An intravitreal dexamethasone implant, which has a treatment effect lasting for 3 months, was also shown to improve visual acuity over sham treatment in patients with DME.25 Most recently, an intravitreal fluocinolone implant that lasts 3 years was approved by the FDA for treatment of DME.26 A significant benefit of the steroid implants is the long duration of treatment effect compared with that of the anti-VEGF injections. However, steroid therapy is associated with the development of cataracts and glaucoma, the rates of which are increased when treatment is prolonged. Because of these adverse effects, steroids are currently used as second-line or third-line treatment in DME. Retinal surgery may be indicated if there is vitreomacular traction that is exacerbating the DME. A vitrectomy is performed to remove the vitreous and relieve any adhesion to the surface of the retina.
Conclusion
Despite the new ophthalmic treatment modalities, it is important to remember that DME is a chronic condition that will require long-term follow-up. Many patients will not experience complete resolution of DME with a single therapy alone. Control of systemic risk factors, including blood sugar with a goal of A1c < 7%, blood pressure, and cholesterol, remains the key to a successful treatment program. Primary care physicians, endocrinologists, diabetologists, optometrists, comprehensive ophthalmologists, retina specialists, and patients must work together to create an individualized treatment regimen that will optimize the patient’s vision by preventing blindness and improving his/her quality of life for years to come.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. Danaei G, Finucane MM, Lu Y, et al; Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Blood Glucose). National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet. 2011;378(9785):31-40.
2. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimate of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014.
3. National Institutes of Health. Diabetic retinopathy: causes and risk factors. NIH Senior Health Website. http://nihseniorhealth.gov/diabeticretinopathy/causesandriskfactors/01.html. Updated February 2015. Accessed September 3, 2015.
4. Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. XV. The long-term incidence of macular edema. Ophthalmology. 1995;102(1):7-16.
5. Yau JW, Rogers SL, Kawasaki R, et al; Meta-Analysis for Eye Disease (META-EYE) Study Group. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556-564.
6. Varma R, Bressler NM, Doan QV, et al. Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol. 2014;132(11):1334-1340.
7. Diep TM, Tsui I. Risk factors associated with diabetic macular edema. Diabetes Res Clin Pract. 2013;100(3):298-305.
8. Varma R, Choudhury F, Klein R, Chung J, Torres M, Azen SP; Los Angeles Latino Eye Study Group. Four-year incidence and progression of diabetic retinopathy and macular edema: the Los Angeles Latino Eye Study. Am J Ophthalmol. 2010;149(5):752-761.e1-e3.
9. Emanuele N, Moritz T, Klein R, et al; Veterans Affairs Diabetes Trial Study Group. Ethnicity, race, and clinically significant macular edema in the Veterans Affairs Diabetes Trial (VADT). Diabetes Res Clin Pract. 2009;86(2):104-110.
10. Klein R, Lee KE, Gangnon RE, Klein BE. The 25-year incidence of visual impairment in type 1 diabetes mellitus: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Ophthalmology. 2010;117(1):63-70.
11. American Academy of Ophthalmology Retina/Vitreous Panel. Preferred Practice Pattern: Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; 2014.
12. Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. IV. Diabetic macular edema. Ophthalmology. 1984;91(12):1464-1474.
13. Klein R, Knudtson MD, Lee KE, Gangnon R, Klein BE. The Wisconsin Epidemiologic Study of Diabetic Retinopathy XXIII: the twenty-five-year incidence of macular edema in persons with type 1 diabetes. Ophthalmology. 2009;116(3):497-503.
14. Chew EY. Diabetic retinopathy and lipid abnormalities. Curr Opin Ophthalmol. 1997;8(3):59-62.
15. Klein R, Sharrett AR, Klein BE, et al; ARIC Group. The association of atherosclerosis, vascular risk factors, and retinopathy in adults with diabetes: the atherosclerosis risk in communities study. Ophthalmology. 2002;109(7):1225-1234.
16. Haefliger IO, Meyer P, Flammer J, Lüscher TF. The vascular endothelium as a regulator of the ocular circulation: a new concept in ophthalmology? Surv Ophthalmol. 1994;39(2):123-132.
17. Lopes de Faria JM, Jalkh AE, Trempe CL, McMeel JW. Diabetic macular edema: risk factors and concomitants. Acta Ophthalmol Scand. 1999;77(2):170-175.
18. Langeler EG, Snelting-Havinga I, van Hinsbergh VW. Passage of low density lipoproteins through monolayers of human arterial endothelial cells. Effects of vasoactive substances in an in vitro model. Arteriosclerosis. 1989;9(4):550-559.
19. Theodossiadis PG, Theodoropoulou S, Neamonitou G, et al. Hemodialysisinduced alterations in macular thickness measured by optical coherence tomography in diabetic patients with end-stage renal disease. Ophthalmologica. 2012;227(2):90-94.
20. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985;103(12):1796-1806.
21. Funatsu H, Noma H, Mimura T, Eguchi S, Hori S. Association of vitreous inflammatory factors with diabetic macular edema. Ophthalmology. 2009;116(1):73-79.
22. Diabetic Retinopathy Clinical Research Network; Elman MJ, Qin H, Aiello LP, et al. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment: three-year randomized trial results. Ophthalmology. 2012;119(11):2312-2318.
23. Korobelnik JF, Do DV, Schmidt-Erfurth U, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121(11):2247-2254.
24. Diabetic Retinopathy Clinical Research Network; Elman MJ, Aiello LP, Beck RW, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117(6):1064-1077.
25. Boyer DS, Yoon YH, Belfort R Jr, et al; Ozurdex MEAD Study Group. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology. 2014;121(10):1904-1914.
26. Campochiaro PA, Brown DM, Pearson A, et al; FAME Study Group. Long-term benefit of sustained-delivery fluocinolone acetonide vitreous inserts for diabetic macular edema. Ophthalmology. 2011;118(4):626-635.e2.
1. Danaei G, Finucane MM, Lu Y, et al; Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Blood Glucose). National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet. 2011;378(9785):31-40.
2. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimate of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014.
3. National Institutes of Health. Diabetic retinopathy: causes and risk factors. NIH Senior Health Website. http://nihseniorhealth.gov/diabeticretinopathy/causesandriskfactors/01.html. Updated February 2015. Accessed September 3, 2015.
4. Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. XV. The long-term incidence of macular edema. Ophthalmology. 1995;102(1):7-16.
5. Yau JW, Rogers SL, Kawasaki R, et al; Meta-Analysis for Eye Disease (META-EYE) Study Group. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556-564.
6. Varma R, Bressler NM, Doan QV, et al. Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol. 2014;132(11):1334-1340.
7. Diep TM, Tsui I. Risk factors associated with diabetic macular edema. Diabetes Res Clin Pract. 2013;100(3):298-305.
8. Varma R, Choudhury F, Klein R, Chung J, Torres M, Azen SP; Los Angeles Latino Eye Study Group. Four-year incidence and progression of diabetic retinopathy and macular edema: the Los Angeles Latino Eye Study. Am J Ophthalmol. 2010;149(5):752-761.e1-e3.
9. Emanuele N, Moritz T, Klein R, et al; Veterans Affairs Diabetes Trial Study Group. Ethnicity, race, and clinically significant macular edema in the Veterans Affairs Diabetes Trial (VADT). Diabetes Res Clin Pract. 2009;86(2):104-110.
10. Klein R, Lee KE, Gangnon RE, Klein BE. The 25-year incidence of visual impairment in type 1 diabetes mellitus: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Ophthalmology. 2010;117(1):63-70.
11. American Academy of Ophthalmology Retina/Vitreous Panel. Preferred Practice Pattern: Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; 2014.
12. Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. IV. Diabetic macular edema. Ophthalmology. 1984;91(12):1464-1474.
13. Klein R, Knudtson MD, Lee KE, Gangnon R, Klein BE. The Wisconsin Epidemiologic Study of Diabetic Retinopathy XXIII: the twenty-five-year incidence of macular edema in persons with type 1 diabetes. Ophthalmology. 2009;116(3):497-503.
14. Chew EY. Diabetic retinopathy and lipid abnormalities. Curr Opin Ophthalmol. 1997;8(3):59-62.
15. Klein R, Sharrett AR, Klein BE, et al; ARIC Group. The association of atherosclerosis, vascular risk factors, and retinopathy in adults with diabetes: the atherosclerosis risk in communities study. Ophthalmology. 2002;109(7):1225-1234.
16. Haefliger IO, Meyer P, Flammer J, Lüscher TF. The vascular endothelium as a regulator of the ocular circulation: a new concept in ophthalmology? Surv Ophthalmol. 1994;39(2):123-132.
17. Lopes de Faria JM, Jalkh AE, Trempe CL, McMeel JW. Diabetic macular edema: risk factors and concomitants. Acta Ophthalmol Scand. 1999;77(2):170-175.
18. Langeler EG, Snelting-Havinga I, van Hinsbergh VW. Passage of low density lipoproteins through monolayers of human arterial endothelial cells. Effects of vasoactive substances in an in vitro model. Arteriosclerosis. 1989;9(4):550-559.
19. Theodossiadis PG, Theodoropoulou S, Neamonitou G, et al. Hemodialysisinduced alterations in macular thickness measured by optical coherence tomography in diabetic patients with end-stage renal disease. Ophthalmologica. 2012;227(2):90-94.
20. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985;103(12):1796-1806.
21. Funatsu H, Noma H, Mimura T, Eguchi S, Hori S. Association of vitreous inflammatory factors with diabetic macular edema. Ophthalmology. 2009;116(1):73-79.
22. Diabetic Retinopathy Clinical Research Network; Elman MJ, Qin H, Aiello LP, et al. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment: three-year randomized trial results. Ophthalmology. 2012;119(11):2312-2318.
23. Korobelnik JF, Do DV, Schmidt-Erfurth U, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121(11):2247-2254.
24. Diabetic Retinopathy Clinical Research Network; Elman MJ, Aiello LP, Beck RW, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117(6):1064-1077.
25. Boyer DS, Yoon YH, Belfort R Jr, et al; Ozurdex MEAD Study Group. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology. 2014;121(10):1904-1914.
26. Campochiaro PA, Brown DM, Pearson A, et al; FAME Study Group. Long-term benefit of sustained-delivery fluocinolone acetonide vitreous inserts for diabetic macular edema. Ophthalmology. 2011;118(4):626-635.e2.