User login
Despite our success in reducing the number of deaths from influenza in the last half-century, we must remain vigilant, since influenza still can kill.1,2 Gene mutations and reassortment among different strains of influenza viruses pose a significant public health threat, especially in an increasingly mobile world.3,4
In this article, we will present an update on influenza to better prepare primary care providers to prevent and treat this ongoing threat.
H3N2v: SWINE FLU DÉJÀ VU?
Outbreaks of swine flu at state and county fairs in 2012 are unprecedented and have raised concerns.
From 1990 to 2010, human infections with swine-origin influenza viruses were sporadic, and the US Centers for Disease Control and Prevention (CDC) confirmed a total of only 27 cases during this period.5 However, the number has been increasing since 2011: as of August 31, 2012, a total of 309 cases had been reported.6
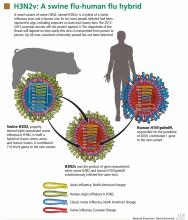
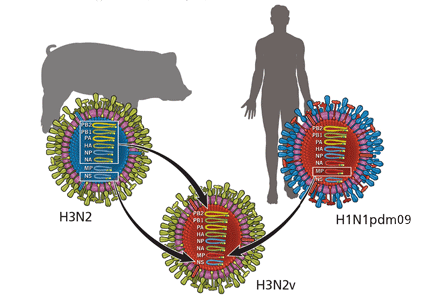
Analysis of viral RNA in clinical respiratory specimens from 12 cases in 2011 revealed a variant strain, called H3N2v, which is a hybrid containing genetic material from swine H3N2 and the 2009 human pandemic virus H1N1pdm09. The M gene in this new variant came from the human virus, while the other seven came from the swine virus when a host was infected with both viruses simultaneously (Figure 1). As a result of this genetic reassortment, this variant virus is genetically and antigenically different from seasonal H3N2.
Epidemiologic data showed that children under 10 years of age are especially susceptible to this new variant because they lack immunity, whereas adolescents and adults may have some immunity from cross-reacting antibodies.7 Most infected people had been exposed to swine in agriculture, including county and state fairs. So far, evidence suggests only limited human-to-human transmission.8 The clinical diagnosis of H3N2v infection relies on the epidemiologic link to exposure to pigs in the week before the onset of illness, since the symptoms are indistinguishable from those of seasonal influenza A or B infections.
In suspected cases, the clinician should notify the local or state public health department and arrange for a special test to be performed on respiratory specimens: the CDC Flu Real-Time Reverse Transcriptase Polymerase Chain Reaction Dx Panel. The reason is that a negative rapid influenza diagnostic test does not rule out influenza infection, and a positive immunofluorescence assay (direct fluorescent antibody staining) cannot specifically detect H3N2v.7
The current seasonal influenza vaccine will not protect against H3N2v. The isolates tested to date were susceptible to the neuraminidase inhibitor drugs oseltamivir (Tamiflu) and zanamivir (Relenza) but resistant to amantadine (Symmetrel) and rimantadine (Flumadine).9
Whether H3N2v will become a significant problem during the upcoming flu season largely depends on the extent of human-to-human transmission. We need to closely follow updates on this virus.
H5N1: THE LOOMING THREAT OF A BIRD FLU PANDEMIC
Since 2003, influenza A H5N1, a highly pathogenic avian virus, has broken out in Asia, Africa, and the Middle East, killing more than 100 million birds. It also has crossed the species barrier to infect humans, with an unusually high death rate.10
As of August 10, 2012, the World Health Organization had reported 608 confirmed cases of this virus infecting humans and 359 associated deaths.11 Most infected patients had a history of close contact with diseased poultry, but limited, nonsustained human-to-human transmission can occur during very close, unprotected contact with a severely ill patient.12
Molecular studies of this virus revealed further insights into its pathogenesis. Some of the viruses isolated from humans have had mutations that allow them to bind to human-type receptors.13 Amino acid substitutions in the polymerase basic protein 2 (PB2) gene are associated with mammalian adaptation, virulence in mice, and viral replication at temperatures present in the upper respiratory tract.14 Furthermore, higher plasma levels of macrophage- and neutrophil-attractant chemokines and both inflammatory and anti-inflammatory cytokines (interleukin 6, interleukin 10, and interferon gamma) have been observed in patients with H5N1 infection, especially in fatal cases.15 A recent study found that H5N1 causes significant perturbations in the host’s protein synthesis machinery as early as 1 hour after infection, suggesting that this virus gains an early advantage in replication by using the host’s proteome.16 The effects of unrestrained viral infection and inflammatory responses induced by H5N1 infection certainly contributed to the primary pathologic process and to death in human fulminant viral pneumonia. The up-regulation of inflammatory cytokines in these infections contributes to the development of sepsis syndrome, acute respiratory distress syndrome, and an increased risk of death, particularly in pregnant women.
Most experts predict that pandemic influenza is probably inevitable.17 If avian H5N1 and a human influenza virus swap genes in a host such as swine, the new hybrid virus will contain genetic material from both strains and will have surface antigens that the human immune system does not recognize. This could lead to a devastating avian flu pandemic with a very high death rate.18
An inactivated whole-virus H5N1 vaccine has been developed by the US government to prevent H5N1 infection.19 For treatment, the neuraminidase inhibitor oseltamivir is the drug of choice.10 Oseltamivir resistance remains uncommon. 20 Fortunately, zanamivir is still active against oseltamivir-resistant variants that have N1 neuraminidase mutations.21
THE 2009 H1N1 PANDEMIC KILLED MORE PEOPLE THAN WE THOUGHT
The fourth flu pandemic of the last 100 years occurred in 2009. (The other three were in 1918, 1957, and 1968.) It was caused by a novel strain, H1N1 of swine origin.22 This 2009 pandemic strain had six genes from the North American swine flu virus and two genes from the Eurasian swine flu virus. The pandemic affected more children and young people (who completely lacked prior immunity to this virus), while older people, who had cross-reacting antibodies, were less affected.
Worldwide, 18,500 people were reported initially to have died in this pandemic from April 2009 to August 2010.23 However, a recent modeling study estimated the number of respiratory and cardiovascular deaths associated with this pandemic at 283,500—about 15 times higher.24
AN AUSTRALIAN OUTBREAK OF OSELTAMIVIR-RESISTANT H1N1
Many strains of influenza A virus are resistant to amantadine and rimantadine, owing to amino acid substitutions in the M2 protein.25 In contrast, resistance to the neuraminidase inhibitors oseltamivir and zanamivir has been reported only occasionally.26
Until recently, most oseltamivir-resistant viruses were isolated from immunocompromised hosts treated with oseltamivir.27–29 All the resistant viral isolates contained an amino acid substitution of histidine (H) to tyrosine (Y) at position 275 of the viral neuraminidase.30 In general, transmission of these oseltamivir-resistant strains has been limited and unsustained, but it can occur in settings of close contact, such as hospitals, school camps, or long train rides.31–35 Oseltamivir-resistant strains were detected in fewer than 1% of isolates from the community during the 2010–2011 influenza season in the Northern Hemisphere and most countries in the Southern Hemisphere during the 2011 flu season.36,37
However, an outbreak of oseltamivir-resistant H1N1 occurred in Australia between June and August 2011.38 In that outbreak, the isolates from only 15% of the 191 people infected with this virus, designated H1N1pdm09, carried the H257Y neuraminidase substitution.39 Further, only 1 of the 191 patients had received oseltamivir before. More importantly, genetic analysis suggested that the infection spread from a single source.
This was the first reported sustained community transmission of oseltamivir-resistant H1N1 in a community previously unexposed to this drug. As such, it is a warning sign of the potential for a widespread outbreak of this virus. In the event of such an outbreak, inhaled zanamivir would be the only effective treatment available.
THIS SEASON’S TRIVALENT INACTIVATED VACCINE
The trivalent inactivated influenza vaccine for the 2012–2013 season contains three inactivated viruses40:
- Influenza A/California/7/2009(H1N1)-like
- Influenza A/Victoria/361/2011(H3N2)-like
- Influenza B/Wisconsin/1/2010-like (Yamagata lineage).
The influenza A H3N2 and influenza B antigens are different from those in the 2011–2012 vaccine.41 The H1N1 strain is derived from H1N1pdm09, which had been contained in the 2011–2012 seasonal vaccine. This vaccine will not protect against H3N2v or H5N1.
LATEST RECOMMENDATIONS ON VACCINATION
Since 2010, the Advisory Committee on Immunization Practices (ACIP) has recommended annual flu shots for all people older than 6 months in the United States.42
Vaccination should be done before the onset of influenza activity in the community as soon as vaccine is available for the season. However, one should continue offering vaccination throughout the influenza season as long as influenza viruses are circulating in the community.
Children ages 6 months through 8 years not previously vaccinated against influenza should receive two doses of influenza vaccine at least 4 weeks apart for an optimal immune response. The US-licensed Afluria vaccine (CSL Biotherapies, King of Prussia, PA), a trivalent inactivated vaccine, is not recommended for children under 9 years of age because of concern about febrile seizures.43,44
There is no contraindication to giving inactivated trivalent influenza vaccine to immunosuppressed people.
The live-attenuated influenza vaccine is indicated only for healthy, nonpregnant people age 2 through 49 years and not for people who care for severely immunosuppressed patients who require a protective environment.
For indications for and details about the different available influenza vaccines, see the ACIP’s current recommendations (www.cdc.gov/mmwr/pdf/wk/mm6132.pdf).40
Updated recommendations for people allergic to eggs
All currently available influenza vaccines are made by growing the virus in chicken eggs. Therefore, severe allergic and anaphylactic reactions can occur in people with egg allergy. The ACIP recommends that if people experienced only hives after egg exposure, they should still receive the trivalent inactivated vaccine. Recently, the ACIP reviewed data from the Vaccine Adverse Event Reporting System45 and issued the following recommendations for the 2012–2013 influenza season40:
- In people who are allergic to eggs, only trivalent inactivated vaccine should be used, not the live-attenuated vaccine, because of lack of data for use of the latter in this group.
- Vaccine should be given by providers who are familiar with the signs of egg allergy.
- Patients with a history of egg allergy who have experienced only hives after exposure to eggs should be observed for a minimum of 30 minutes after vaccination.
- Patients who experience lightheadedness, respiratory distress, angioedema, or recurrent emesis or who require epinephrine or emergency medical attention after egg exposure should be referred before vaccination to a physician who has expertise in managing allergic conditions.
- Tolerance to egg-containing foods does not exclude the possibility of egg allergy. Egg allergy can be confirmed by a consistent medical history of adverse reactions to eggs or egg-containing foods, plus skin or blood testing for immunoglobulin E antibodies to egg proteins.
A high-dose vaccine is available for people 65 years and older
The rates of hospitalization and death due to seasonal flu in elderly people have increased significantly in the last 20 years despite rising rates of vaccination.46–48 This is largely due to lower serologic response rates and vaccine efficacy in older adults with weaker immune systems.
Several studies have shown that the development of protective antibody titers depends on the dose of antigen.49–53 A randomized, controlled clinical trial compared the immunogenicity of a high-dose vaccine and a standard-dose vaccine in older adults and found that the level of antibody response was significantly higher with the high-dose vaccine, and that the rate of adverse reactions was the same.54
In December 2009, the US Food and Drug Administration (FDA) licensed a new trivalent inactivated influenza vaccine with high doses of hemagglutinin antigens for adults over the age of 65.55 Postlicensure safety surveillance in 2010 revealed no serious safety concerns.56
At present, the ACIP expresses no preference for standard-dose or high-dose vaccine for adults 65 years of age and older.40 Importantly, if only the standard-dose vaccine is at hand, the opportunity for influenza vaccination should not be missed with the intention of giving high-dose vaccine at a later date.
A NEW QUADRIVALENT LIVE-ATTENUATED INFLUENZA VACCINE FOR THE 2013–2014 SEASON
In February 2012, the FDA approved the first quadrivalent live-attenuated influenza vaccine, which is expected to replace the currently available trivalent live-attenuated influenza vaccine in the 2013–2014 flu season. The quadrivalent vaccine will include both lineages of the circulating influenza B viruses (the Victoria and Yamagata lineages). The reasons for this inclusion is the difficulty in predicting which of these viruses will predominate in any given season, and the limited cross-resistance by immunization against one of the lineages.
A recent analysis57 estimated that such a vaccine is likely to further reduce influenza cases, related hospitalizations, and deaths compared with the current trivalent vaccine. Like the current trivalent live-attenuated vaccine, the quadrivalent vaccine is inhaled.
EVOLVING VACCINATION POLICY IN HEALTH CARE WORKERS
Starting in January 2013, the Centers for Medicare and Medicaid Services will require hospitals to report how many of their health care workers are vaccinated. These rates will be publicly reported as a measure of hospital quality. This has fueled the ongoing debate about mandating influenza vaccination for health care workers. Studies have shown that the most important factors in increasing influenza vaccination rates among health care workers are requiring vaccination as a condition for employment and making vaccination available on-site, for more than 1 day, at no cost to the worker.58
As an alternative, some institutions have implemented a “shot-or-mask” policy whereby a health care worker who elects not to be vaccinated because of medical or religious reasons would be asked to wear a mask during all faceto-face encounters with patients.
NEW ANTIVIRAL DRUGS ON THE HORIZON
The emergence of oseltamivir-resistant strains in recent years caused a great deal of concern in public health regarding the potential for outbreaks of drug-resistant influenza.34,35,59–61
A recent Asian randomized clinical trial reported the efficacy of a long-acting neuraminidase inhibitor, laninamivir octanoate, in the treatment of seasonal influenza.62 This study showed that a single inhalation of this drug is effective in treating seasonal influenza, including that caused by oseltamivir-resistant strains in adults. Laninamivir is currently approved in Japan.
CHALLENGES IN PREVENTING AND TREATING INFLUENZA
Despite the great advances that we have made in preventing and treating influenza in the last half-century, we still face many challenges. Each year in the United States, influenza infection results in an estimated 31 million outpatient visits, 226,000 hospital admissions, and 36,000 deaths.42
Antigenic drift and shift. Influenza viruses circulating among animals and humans vary genetically from season to season and within seasons. As a result of this changing viral antigenicity, the virus can evade the human immune system, causing widespread outbreaks.
One of the unique and most remarkable features of influenza virus is the antigenic variation: antigenic drift and antigenic shift. Antigenic drift is the relatively minor antigenic changes that occur frequently within an influenza subtype, typically resulting from genetic mutation of viral RNA coding for hemagglutinin or neuraminidase. This causes annual regional epidemics. In contrast, antigenic shift is the result of genetic material reassortment: the emerging of new viral RNA from different strains of different species. This often leads to global pandemics.
Therefore, it is challenging to accurately predict the antigenic makeup of influenza viruses for each season and to include new emerging antigens in the vaccine production, as we are facing a moving target. We prepare influenza vaccines each season based on past experience.63
Vaccination rates have hit a plateau of 60% to 70% in adults since the 1990s, in spite of greater vaccine supply and recommendations that all adults, regardless of underlying disease, be vaccinated annually.64 Similarly, only 51% of children age 6 months to 17 years were vaccinated in the 2010–2011 season.65 And vaccination rates are even lower in low-income populations.66,67
The emergence of oseltamivir-resistant strains in recent years, not only in people exposed to oseltamivir but also in those who haven’t been exposed to this drug, with sustained transmission, certainly raises the possibility of a more difficult epidemic to control.38
Global travel, global infection. Our last H1N1 pandemic in 2009 was an example of how easily the influenza virus can spread rapidly in today’s highly mobile global society.22
What we must do
As primary health care providers, we must closely monitor the community outbreak and the emergence of drug-resistant strains and strongly recommend vaccination for all patients older than 6 months, since timely vaccination is the cornerstone of influenza prevention. Although many have questioned the efficacy of influenza vaccination, a recent meta-analysis showed a 59% pooled efficacy of the trivalent inactivated vaccine in adults age 18 to 65 years in preventing virologically confirmed influenza, and 83% pooled efficacy of the live-attenuated influenza vaccine in children age 6 months to 7 years.68 Novel approaches for vaccination reminders, such as text messaging69 in addition to traditional mail or telephone reminders, can improve vaccination compliance in today’s highly mobile world that is more than ever connected.
With the lessons learned from four pandemics in the last century, updated recommendations for prevention, and adequate vaccine supply, we should be ready to face the challenge of another flu season.
- Doshi P. Trends in recorded influenza mortality: United States, 1900–2004. Am J Public Health 2008; 98:939–945.
- Centers for Disease Control and Prevention (CDC). Estimates of deaths associated with seasonal influenza — United States, 1976–2007. MMWR Morb Mortal Wkly Rep 2010; 59:1057–1062.
- Reid AH, Taubenberger JK, Fanning TG. Evidence of an absence: the genetic origins of the 1918 pandemic influenza virus. Nat Rev Microbiol 2004; 2:909–914.
- Lindstrom S, Garten R, Balish A, et al. Human infections with novel reassortant influenza A(H3N2)v viruses, United States, 2011. Emerg Infect Dis 2012; 18:834–837.
- Shu B, Garten R, Emery S, et al. Genetic analysis and antigenic characterization of swine origin influenza viruses isolated from humans in the United States, 1990–2010. Virology 2012; 422:151–160.
- Centers for Disease Control and Prevention (CDC). http://www.cdc.gov/flu/swineflu/h3n2v-outbreak.htm. Accessed September 27, 2012.
- Centers for Disease Control and Prevention (CDC). Evaluation of rapid influenza diagnostic tests for influenza A (H3N2)v virus and updated case count — United States, 2012. MMWR Morb Mortal Wkly Rep 2012; 61:619–621.
- Centers for Disease Control and Prevention (CDC). Update: Influenza A (H3N2)v transmission and guidelines — five states, 2011. MMWR Morb Mortal Wkly Rep 2012; 60:1741–1744.
- Centers for Disease Control and Prevention (CDC). Interim information for clinicians about human infections with H3N2v virus. http://www.cdc.gov/flu/swineflu/h3n2v-clinician.htm. Accessed September 27, 2012.
- Writing Committee of the Second World Health Organization Consultation on Clinical Aspects of Human Infection with Avian Influenza A (H5N1) Virus; Abdel-Ghafar AN, Chotpitayasunondh T, Gao Z, et al. Update on avian influenza A (H5N1) virus infection in humans. N Engl J Med 2008; 358:261–273.
- World Health Organization (WHO). http://www.who.int/influenza/human_animal_interface/H5N1_cumulative_table_archives/en/index.html. Accessed September 27, 2012.
- Ungchusak K, Auewarakul P, Dowell SF, et al. Probable person-to-person transmission of avian influenza A (H5N1). N Engl J Med 2005; 352:333–340.
- Yamada S, Suzuki Y, Suzuki T, et al. Haemagglutinin mutations responsible for the binding of H5N1 influenza A viruses to human-type receptors. Nature 2006; 444:378–382.
- Hatta M, Hatta Y, Kim JH, et al. Growth of H5N1 influenza A viruses in the upper respiratory tracts of mice. PLoS Pathog 2007; 3:1374–1379.
- de Jong MD, Simmons CP, Thanh TT, et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med 2006; 12:1203–1207.
- Cheung CY, Chan EY, Krasnoselsky A, et al. H5N1 virus causes significant perturbations in host proteome very early in influenza virus-infected primary human monocyte-derived macrophages. J Infect Dis 2012; 206:640–645.
- Gordon S. Avian influenza: a wake-up call from birds to humans. Cleve Clin J Med 2004; 71:273–274.
- Jin XW, Mossad SB. Avian influenza: an emerging pandemic threat. Cleve Clin J Med 2005; 72:1129–1234.
- Ehrlich HJ, Müller M, Oh HM, et al; Baxter H5N1 Pandemic Influenza Vaccine Clinical Study Team. A clinical trial of a whole-virus H5N1 vaccine derived from cell culture. N Engl J Med 2008; 358:2573–2584.
- de Jong MD, Tran TT, Truong HK, et al. Oseltamivir resistance during treatment of influenza A (H5N1) infection. N Engl J Med 2005; 353:2667–2672.
- Le QM, Kiso M, Someya K, et al. Avian flu: isolation of drug-resistant H5N1 virus. Nature 2005; 437:1108.
- Ison MG, Lee N. Influenza 2010–2011: lessons from the 2009 pandemic. Cleve Clin J Med 2010; 77:812–820.
- World Health Organization (WHO). Pandemic (H1N1) 2009 — update 112. http://www.who.int/csr/don/2010_08_06/en/index.html. Accessed September 27, 2012.
- Dawood FS, Iuliano AD, Reed C, et al. Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: a modelling study. Lancet Infect Dis 2012; 12:687–695.
- Bright RA, Shay DK, Shu B, Cox NJ, Klimov AI. Adamantane resistance among influenza A viruses isolated early during the 2005–2006 influenza season in the United States. JAMA 2006; 295:891–894.
- Nguyen HT, Fry AM, Gubareva LV. Neuraminidase inhibitor resistance in influenza viruses and laboratory testing methods. Antivir Ther 2012; 17:159–173.
- Graitcer SB, Gubareva L, Kamimoto L, et al. Characteristics of patients with oseltamivir-resistant pandemic (H1N1) 2009, United States. Emerg Infect Dis 2011; 17:255–257.
- Hurt AC, Deng YM, Ernest J, et al. Oseltamivir-resistant influenza viruses circulating during the first year of the influenza A(H1N1) 2009 pandemic in the Asia-Pacific region, March 2009 to March 2010. Euro Surveill 2011; 16:19770.
- Meijer A, Jonges M, Abbink F, et al. Oseltamivir-resistant pandemic A(H1N1) 2009 influenza viruses detected through enhanced surveillance in the Netherlands, 2009–2010. Antiviral Res 2011; 92:81–89.
- Gubareva LV, Kaiser L, Hayden FG. IInfluenza virus neuraminidase inhibitors. Lancet 2000; 355:827–835.
- Wolfe C, Greenwald I, Chen L. Pandemic (H1N1) 2009 and oseltamivir resistance in hematology/oncology patients. Emerg Infect Dis 2010; 16:1809–1811.
- Moore C, Galiano M, Lackenby A, et al. Evidence of person-to-person transmission of oseltamivir-resistant pandemic influenza A(H1N1) 2009 virus in a hematology unit. J Infect Dis 2011; 203:18–24.
- Chen LF, Dailey NJ, Rao AK, et al. Cluster of oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infections on a hospital ward among immunocompromised patients — North Carolina, 2009. J Infect Dis 2011; 203:838–846.
- Centers for Disease Control and Prevention (CDC). Oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infection in two summer campers receiving prophylaxis — North Carolina, 2009. MMWR Morb Mortal Wkly Rep 2009; 58:969–972.
- Le QM, Wertheim HF, Tran ND, van Doorn HR, Nguyen TH, Horby P; Vietnam H1N1 Investigation Team. A community cluster of oseltamivir-resistant cases of 2009 H1N1 influenza. N Engl J Med 2010; 362:86–87.
- Lackenby A, Moran Gilad J, Pebody R, et al. Continued emergence and changing epidemiology of oseltamivir-resistant influenza A(H1N1)2009 virus, United Kingdom, winter 2010/11. Euro Surveill 2011; 16:19784.
- World Health Organization (WHO). Summary of influenza antiviral susceptibility surveillance findings, September 2010 – March 2011. http://www.who.int/influenza/gisrs_laboratory/updates/antiviral_susceptibility/en/index.html. Accessed September 27, 2012.
- Hurt AC, Hardie K, Wilson NJ, et al. Community transmission of oseltamivir-resistant A(H1N1)pdm09 influenza. N Engl J Med 2011; 365:2541–2542.
- Hurt AC, Hardie K, Wilson NJ, et al. Characteristics of a widespread community cluster of H275Y oseltamivir-resistant A(H1N1)pdm09 influenza in Australia. J Infect Dis 2012; 206:148–157.
- Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP) — United States, 2012–13 Influenza Season. MMWR Morb Mortal Wkly Rep 2012; 61:613–618.
- Food and Drug Administration (FDA). Summary minutes: vaccines and related biological products advisory committee. February 28–29, 2012. Silver Spring, MD. http://www.fda.gov/downloads/Advisory-Committees/CommitteesMeetingMaterials/BloodVaccinesandOther-Biologics/VaccinesandRelatedBiologicalProductsAdvisoryCommittee/UCM296193.pdf. Accessed September 28, 2012.
- Fiore AE, Uyeki TM, Broder K, et al; Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Recomm Rep 2010; 59:1–62.
- Centers for Disease Control and Prevention (CDC). Update: recommendations of the Advisory Committee on Immunization Practices (ACIP) regarding use of CSL seasonal influenza vaccine (Afluria) in the United States during 2010–11. MMWR Morb Mortal Wkly Rep 2010; 59:989–992.
- Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2011. MMWR Morb Mortal Wkly Rep 2011; 60:1128–1132.
- Centers for Disease Control and Prevention (CDC). Advisory Committee on Immunization Practices: Update on influenza vaccine safety monitoring. June 20–21, 2012. Atlanta, GA. http://www.cdc.gov/vaccines/acip/meetings/downloads/slides-jun-2012/03-influenza-Shimabukuro.pdf. Accessed September 28, 2012.
- Simonsen L, Reichert TA, Viboud C, Blackwelder WC, Taylor RJ, Miller MA. Impact of influenza vaccination on seasonal mortality in the US elderly population. Arch Intern Med 2005; 165:265–272.
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004; 292:1333–1340.
- Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003; 289:179–186.
- Mostow SR, Schoenbaum SC, Dowdle WR, Coleman MT, Kaye HS. Inactivated vaccines. 1. Volunteer studies with very high doses of influenza vaccine purified by zonal ultracentrifugation. Postgrad Med J 1973; 49:152–158.
- Keitel WA, Atmar RL, Cate TR, et al. Safety of high doses of influenza vaccine and effect on antibody responses in elderly persons. Arch Intern Med 2006; 166:1121–1127.
- Ruben FL, Jackson GG. A new subunit influenza vaccine: acceptability compared with standard vaccines and effect of dose on antigenicity. J Infect Dis 1972; 125:656–664.
- Palache AM, Beyer WE, Sprenger MJ, et al. Antibody response after influenza immunization with various vaccine doses: a double-blind, placebo-controlled, multi-centre, dose-response study in elderly nursing-home residents and young volunteers. Vaccine 1993; 11:3–9.
- Couch RB, Winokur P, Brady R, et al. Safety and immunogenicity of a high dosage trivalent influenza vaccine among elderly subjects. Vaccine 2007; 25:7656–7663.
- Falsey AR, Treanor JJ, Tornieporth N, Capellan J, Gorse GJ. Randomized, double-blind controlled phase 3 trial comparing the immunogenicity of high-dose and standard-dose influenza vaccine in adults 65 years of age and older. J Infect Dis 2009; 200:172–180.
- US Food and Drug Administration. Vaccines, Blood & Biologics. December 23,2009 approval letter—Fluzone high-dose. http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm195481.htm. Accessed October 1, 2012.
- Moro PL, Arana J, Cano M, et al. Postlicensure safety surveillance for high-dose trivalent inactivated influenza vaccine in the Vaccine Adverse Event Reporting System, 1 July 2010–31 December 2010. Clin Infect Dis 2012; 54:1608–1614.
- Reed C, Meltzer MI, Finelli L, Fiore A. Public health impact of including two lineages of influenza B in a quadrivalent seasonal influenza vaccine. Vaccine 2012; 30:1993–1998.
- Centers for Disease Control and Prevention (CDC). Influenza vaccination coverage among health-care personnel — United States, 2010–11 influenza season. MMWR Morb Mortal Wkly Rep 2011; 60:1073–1077.
- Meijer A, Lackenby A, Hungnes O, et al; European Influenza Surveillance Scheme. Oseltamivir-resistant influenza virus A (H1N1), Europe, 2007–08 season. Emerg Infect Dis 2009; 15:552–560.
- Moscona A. Global transmission of oseltamivir-resistant influenza. N Engl J Med 2009; 360:953–956.
- World Health Organization (WHO). Influenza A virus resistance to oseltamivir. http://www.who.int/influenza/patient_care/antivirals/oseltamivir_summary/en/. Accessed September 28, 2012.
- Watanabe A, Chang SC, Kim MJ, Chu DW, Ohashi Y; MARVEL Study Group. Long-acting neuraminidase inhibitor laninamivir octanoate versus oseltamivir for treatment of influenza: a double-blind, randomized, noninferiority clinical trial. Clin Infect Dis 2010; 51:1167–1175.
- Deyde VM, Gubareva LV. Influenza genome analysis using pyro-sequencing method: current applications for a moving target. Expert Rev Mol Diagn 2009; 9:493–509.
- Schuchat A, Katz JM. Protecting adults from influenza: tis the season to learn from the pandemic. J Infect Dis 2012; 206:803–805.
- Centers for Disease Control and Prevention (CDC). Final state-level influenza vaccination coverage estimates for the 2010–11 season — United States, National Immunization Survey and Behavioral Risk Factor Surveillance System, August 2010 through May 2011. http://www.cdc.gov/flu/professionals/vaccination/coverage_1011estimates.htm. Accessed September 28, 2012.
- Bhatt P, Block SL, Toback SL, Ambrose CS. Timing of the availability and administration of influenza vaccine through the vaccines for children program. Pediatr Infect Dis J 2011; 30:100–106.
- Lee BY, Brown ST, Bailey RR, et al. The benefits to all of ensuring equal and timely access to influenza vaccines in poor communities. Health Aff (Millwood) 2011; 30:1141–1150.
- Osterholm MT, Kelley NS, Sommer A, Belongia EA. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis 2012; 12:36–44.
- Stockwell MS, Kharbanda EO, Martinez RA, Vargas CY, Vawdrey DK, Camargo S. Effect of a text messaging intervention on influenza vaccination in an urban, low-income pediatric and adolescent population: a randomized controlled trial. JAMA 2012; 307:1702–1708.
Despite our success in reducing the number of deaths from influenza in the last half-century, we must remain vigilant, since influenza still can kill.1,2 Gene mutations and reassortment among different strains of influenza viruses pose a significant public health threat, especially in an increasingly mobile world.3,4
In this article, we will present an update on influenza to better prepare primary care providers to prevent and treat this ongoing threat.
H3N2v: SWINE FLU DÉJÀ VU?
Outbreaks of swine flu at state and county fairs in 2012 are unprecedented and have raised concerns.
From 1990 to 2010, human infections with swine-origin influenza viruses were sporadic, and the US Centers for Disease Control and Prevention (CDC) confirmed a total of only 27 cases during this period.5 However, the number has been increasing since 2011: as of August 31, 2012, a total of 309 cases had been reported.6
Analysis of viral RNA in clinical respiratory specimens from 12 cases in 2011 revealed a variant strain, called H3N2v, which is a hybrid containing genetic material from swine H3N2 and the 2009 human pandemic virus H1N1pdm09. The M gene in this new variant came from the human virus, while the other seven came from the swine virus when a host was infected with both viruses simultaneously (Figure 1). As a result of this genetic reassortment, this variant virus is genetically and antigenically different from seasonal H3N2.
Epidemiologic data showed that children under 10 years of age are especially susceptible to this new variant because they lack immunity, whereas adolescents and adults may have some immunity from cross-reacting antibodies.7 Most infected people had been exposed to swine in agriculture, including county and state fairs. So far, evidence suggests only limited human-to-human transmission.8 The clinical diagnosis of H3N2v infection relies on the epidemiologic link to exposure to pigs in the week before the onset of illness, since the symptoms are indistinguishable from those of seasonal influenza A or B infections.
In suspected cases, the clinician should notify the local or state public health department and arrange for a special test to be performed on respiratory specimens: the CDC Flu Real-Time Reverse Transcriptase Polymerase Chain Reaction Dx Panel. The reason is that a negative rapid influenza diagnostic test does not rule out influenza infection, and a positive immunofluorescence assay (direct fluorescent antibody staining) cannot specifically detect H3N2v.7
The current seasonal influenza vaccine will not protect against H3N2v. The isolates tested to date were susceptible to the neuraminidase inhibitor drugs oseltamivir (Tamiflu) and zanamivir (Relenza) but resistant to amantadine (Symmetrel) and rimantadine (Flumadine).9
Whether H3N2v will become a significant problem during the upcoming flu season largely depends on the extent of human-to-human transmission. We need to closely follow updates on this virus.
H5N1: THE LOOMING THREAT OF A BIRD FLU PANDEMIC
Since 2003, influenza A H5N1, a highly pathogenic avian virus, has broken out in Asia, Africa, and the Middle East, killing more than 100 million birds. It also has crossed the species barrier to infect humans, with an unusually high death rate.10
As of August 10, 2012, the World Health Organization had reported 608 confirmed cases of this virus infecting humans and 359 associated deaths.11 Most infected patients had a history of close contact with diseased poultry, but limited, nonsustained human-to-human transmission can occur during very close, unprotected contact with a severely ill patient.12
Molecular studies of this virus revealed further insights into its pathogenesis. Some of the viruses isolated from humans have had mutations that allow them to bind to human-type receptors.13 Amino acid substitutions in the polymerase basic protein 2 (PB2) gene are associated with mammalian adaptation, virulence in mice, and viral replication at temperatures present in the upper respiratory tract.14 Furthermore, higher plasma levels of macrophage- and neutrophil-attractant chemokines and both inflammatory and anti-inflammatory cytokines (interleukin 6, interleukin 10, and interferon gamma) have been observed in patients with H5N1 infection, especially in fatal cases.15 A recent study found that H5N1 causes significant perturbations in the host’s protein synthesis machinery as early as 1 hour after infection, suggesting that this virus gains an early advantage in replication by using the host’s proteome.16 The effects of unrestrained viral infection and inflammatory responses induced by H5N1 infection certainly contributed to the primary pathologic process and to death in human fulminant viral pneumonia. The up-regulation of inflammatory cytokines in these infections contributes to the development of sepsis syndrome, acute respiratory distress syndrome, and an increased risk of death, particularly in pregnant women.
Most experts predict that pandemic influenza is probably inevitable.17 If avian H5N1 and a human influenza virus swap genes in a host such as swine, the new hybrid virus will contain genetic material from both strains and will have surface antigens that the human immune system does not recognize. This could lead to a devastating avian flu pandemic with a very high death rate.18
An inactivated whole-virus H5N1 vaccine has been developed by the US government to prevent H5N1 infection.19 For treatment, the neuraminidase inhibitor oseltamivir is the drug of choice.10 Oseltamivir resistance remains uncommon. 20 Fortunately, zanamivir is still active against oseltamivir-resistant variants that have N1 neuraminidase mutations.21
THE 2009 H1N1 PANDEMIC KILLED MORE PEOPLE THAN WE THOUGHT
The fourth flu pandemic of the last 100 years occurred in 2009. (The other three were in 1918, 1957, and 1968.) It was caused by a novel strain, H1N1 of swine origin.22 This 2009 pandemic strain had six genes from the North American swine flu virus and two genes from the Eurasian swine flu virus. The pandemic affected more children and young people (who completely lacked prior immunity to this virus), while older people, who had cross-reacting antibodies, were less affected.
Worldwide, 18,500 people were reported initially to have died in this pandemic from April 2009 to August 2010.23 However, a recent modeling study estimated the number of respiratory and cardiovascular deaths associated with this pandemic at 283,500—about 15 times higher.24
AN AUSTRALIAN OUTBREAK OF OSELTAMIVIR-RESISTANT H1N1
Many strains of influenza A virus are resistant to amantadine and rimantadine, owing to amino acid substitutions in the M2 protein.25 In contrast, resistance to the neuraminidase inhibitors oseltamivir and zanamivir has been reported only occasionally.26
Until recently, most oseltamivir-resistant viruses were isolated from immunocompromised hosts treated with oseltamivir.27–29 All the resistant viral isolates contained an amino acid substitution of histidine (H) to tyrosine (Y) at position 275 of the viral neuraminidase.30 In general, transmission of these oseltamivir-resistant strains has been limited and unsustained, but it can occur in settings of close contact, such as hospitals, school camps, or long train rides.31–35 Oseltamivir-resistant strains were detected in fewer than 1% of isolates from the community during the 2010–2011 influenza season in the Northern Hemisphere and most countries in the Southern Hemisphere during the 2011 flu season.36,37
However, an outbreak of oseltamivir-resistant H1N1 occurred in Australia between June and August 2011.38 In that outbreak, the isolates from only 15% of the 191 people infected with this virus, designated H1N1pdm09, carried the H257Y neuraminidase substitution.39 Further, only 1 of the 191 patients had received oseltamivir before. More importantly, genetic analysis suggested that the infection spread from a single source.
This was the first reported sustained community transmission of oseltamivir-resistant H1N1 in a community previously unexposed to this drug. As such, it is a warning sign of the potential for a widespread outbreak of this virus. In the event of such an outbreak, inhaled zanamivir would be the only effective treatment available.
THIS SEASON’S TRIVALENT INACTIVATED VACCINE
The trivalent inactivated influenza vaccine for the 2012–2013 season contains three inactivated viruses40:
- Influenza A/California/7/2009(H1N1)-like
- Influenza A/Victoria/361/2011(H3N2)-like
- Influenza B/Wisconsin/1/2010-like (Yamagata lineage).
The influenza A H3N2 and influenza B antigens are different from those in the 2011–2012 vaccine.41 The H1N1 strain is derived from H1N1pdm09, which had been contained in the 2011–2012 seasonal vaccine. This vaccine will not protect against H3N2v or H5N1.
LATEST RECOMMENDATIONS ON VACCINATION
Since 2010, the Advisory Committee on Immunization Practices (ACIP) has recommended annual flu shots for all people older than 6 months in the United States.42
Vaccination should be done before the onset of influenza activity in the community as soon as vaccine is available for the season. However, one should continue offering vaccination throughout the influenza season as long as influenza viruses are circulating in the community.
Children ages 6 months through 8 years not previously vaccinated against influenza should receive two doses of influenza vaccine at least 4 weeks apart for an optimal immune response. The US-licensed Afluria vaccine (CSL Biotherapies, King of Prussia, PA), a trivalent inactivated vaccine, is not recommended for children under 9 years of age because of concern about febrile seizures.43,44
There is no contraindication to giving inactivated trivalent influenza vaccine to immunosuppressed people.
The live-attenuated influenza vaccine is indicated only for healthy, nonpregnant people age 2 through 49 years and not for people who care for severely immunosuppressed patients who require a protective environment.
For indications for and details about the different available influenza vaccines, see the ACIP’s current recommendations (www.cdc.gov/mmwr/pdf/wk/mm6132.pdf).40
Updated recommendations for people allergic to eggs
All currently available influenza vaccines are made by growing the virus in chicken eggs. Therefore, severe allergic and anaphylactic reactions can occur in people with egg allergy. The ACIP recommends that if people experienced only hives after egg exposure, they should still receive the trivalent inactivated vaccine. Recently, the ACIP reviewed data from the Vaccine Adverse Event Reporting System45 and issued the following recommendations for the 2012–2013 influenza season40:
- In people who are allergic to eggs, only trivalent inactivated vaccine should be used, not the live-attenuated vaccine, because of lack of data for use of the latter in this group.
- Vaccine should be given by providers who are familiar with the signs of egg allergy.
- Patients with a history of egg allergy who have experienced only hives after exposure to eggs should be observed for a minimum of 30 minutes after vaccination.
- Patients who experience lightheadedness, respiratory distress, angioedema, or recurrent emesis or who require epinephrine or emergency medical attention after egg exposure should be referred before vaccination to a physician who has expertise in managing allergic conditions.
- Tolerance to egg-containing foods does not exclude the possibility of egg allergy. Egg allergy can be confirmed by a consistent medical history of adverse reactions to eggs or egg-containing foods, plus skin or blood testing for immunoglobulin E antibodies to egg proteins.
A high-dose vaccine is available for people 65 years and older
The rates of hospitalization and death due to seasonal flu in elderly people have increased significantly in the last 20 years despite rising rates of vaccination.46–48 This is largely due to lower serologic response rates and vaccine efficacy in older adults with weaker immune systems.
Several studies have shown that the development of protective antibody titers depends on the dose of antigen.49–53 A randomized, controlled clinical trial compared the immunogenicity of a high-dose vaccine and a standard-dose vaccine in older adults and found that the level of antibody response was significantly higher with the high-dose vaccine, and that the rate of adverse reactions was the same.54
In December 2009, the US Food and Drug Administration (FDA) licensed a new trivalent inactivated influenza vaccine with high doses of hemagglutinin antigens for adults over the age of 65.55 Postlicensure safety surveillance in 2010 revealed no serious safety concerns.56
At present, the ACIP expresses no preference for standard-dose or high-dose vaccine for adults 65 years of age and older.40 Importantly, if only the standard-dose vaccine is at hand, the opportunity for influenza vaccination should not be missed with the intention of giving high-dose vaccine at a later date.
A NEW QUADRIVALENT LIVE-ATTENUATED INFLUENZA VACCINE FOR THE 2013–2014 SEASON
In February 2012, the FDA approved the first quadrivalent live-attenuated influenza vaccine, which is expected to replace the currently available trivalent live-attenuated influenza vaccine in the 2013–2014 flu season. The quadrivalent vaccine will include both lineages of the circulating influenza B viruses (the Victoria and Yamagata lineages). The reasons for this inclusion is the difficulty in predicting which of these viruses will predominate in any given season, and the limited cross-resistance by immunization against one of the lineages.
A recent analysis57 estimated that such a vaccine is likely to further reduce influenza cases, related hospitalizations, and deaths compared with the current trivalent vaccine. Like the current trivalent live-attenuated vaccine, the quadrivalent vaccine is inhaled.
EVOLVING VACCINATION POLICY IN HEALTH CARE WORKERS
Starting in January 2013, the Centers for Medicare and Medicaid Services will require hospitals to report how many of their health care workers are vaccinated. These rates will be publicly reported as a measure of hospital quality. This has fueled the ongoing debate about mandating influenza vaccination for health care workers. Studies have shown that the most important factors in increasing influenza vaccination rates among health care workers are requiring vaccination as a condition for employment and making vaccination available on-site, for more than 1 day, at no cost to the worker.58
As an alternative, some institutions have implemented a “shot-or-mask” policy whereby a health care worker who elects not to be vaccinated because of medical or religious reasons would be asked to wear a mask during all faceto-face encounters with patients.
NEW ANTIVIRAL DRUGS ON THE HORIZON
The emergence of oseltamivir-resistant strains in recent years caused a great deal of concern in public health regarding the potential for outbreaks of drug-resistant influenza.34,35,59–61
A recent Asian randomized clinical trial reported the efficacy of a long-acting neuraminidase inhibitor, laninamivir octanoate, in the treatment of seasonal influenza.62 This study showed that a single inhalation of this drug is effective in treating seasonal influenza, including that caused by oseltamivir-resistant strains in adults. Laninamivir is currently approved in Japan.
CHALLENGES IN PREVENTING AND TREATING INFLUENZA
Despite the great advances that we have made in preventing and treating influenza in the last half-century, we still face many challenges. Each year in the United States, influenza infection results in an estimated 31 million outpatient visits, 226,000 hospital admissions, and 36,000 deaths.42
Antigenic drift and shift. Influenza viruses circulating among animals and humans vary genetically from season to season and within seasons. As a result of this changing viral antigenicity, the virus can evade the human immune system, causing widespread outbreaks.
One of the unique and most remarkable features of influenza virus is the antigenic variation: antigenic drift and antigenic shift. Antigenic drift is the relatively minor antigenic changes that occur frequently within an influenza subtype, typically resulting from genetic mutation of viral RNA coding for hemagglutinin or neuraminidase. This causes annual regional epidemics. In contrast, antigenic shift is the result of genetic material reassortment: the emerging of new viral RNA from different strains of different species. This often leads to global pandemics.
Therefore, it is challenging to accurately predict the antigenic makeup of influenza viruses for each season and to include new emerging antigens in the vaccine production, as we are facing a moving target. We prepare influenza vaccines each season based on past experience.63
Vaccination rates have hit a plateau of 60% to 70% in adults since the 1990s, in spite of greater vaccine supply and recommendations that all adults, regardless of underlying disease, be vaccinated annually.64 Similarly, only 51% of children age 6 months to 17 years were vaccinated in the 2010–2011 season.65 And vaccination rates are even lower in low-income populations.66,67
The emergence of oseltamivir-resistant strains in recent years, not only in people exposed to oseltamivir but also in those who haven’t been exposed to this drug, with sustained transmission, certainly raises the possibility of a more difficult epidemic to control.38
Global travel, global infection. Our last H1N1 pandemic in 2009 was an example of how easily the influenza virus can spread rapidly in today’s highly mobile global society.22
What we must do
As primary health care providers, we must closely monitor the community outbreak and the emergence of drug-resistant strains and strongly recommend vaccination for all patients older than 6 months, since timely vaccination is the cornerstone of influenza prevention. Although many have questioned the efficacy of influenza vaccination, a recent meta-analysis showed a 59% pooled efficacy of the trivalent inactivated vaccine in adults age 18 to 65 years in preventing virologically confirmed influenza, and 83% pooled efficacy of the live-attenuated influenza vaccine in children age 6 months to 7 years.68 Novel approaches for vaccination reminders, such as text messaging69 in addition to traditional mail or telephone reminders, can improve vaccination compliance in today’s highly mobile world that is more than ever connected.
With the lessons learned from four pandemics in the last century, updated recommendations for prevention, and adequate vaccine supply, we should be ready to face the challenge of another flu season.
Despite our success in reducing the number of deaths from influenza in the last half-century, we must remain vigilant, since influenza still can kill.1,2 Gene mutations and reassortment among different strains of influenza viruses pose a significant public health threat, especially in an increasingly mobile world.3,4
In this article, we will present an update on influenza to better prepare primary care providers to prevent and treat this ongoing threat.
H3N2v: SWINE FLU DÉJÀ VU?
Outbreaks of swine flu at state and county fairs in 2012 are unprecedented and have raised concerns.
From 1990 to 2010, human infections with swine-origin influenza viruses were sporadic, and the US Centers for Disease Control and Prevention (CDC) confirmed a total of only 27 cases during this period.5 However, the number has been increasing since 2011: as of August 31, 2012, a total of 309 cases had been reported.6
Analysis of viral RNA in clinical respiratory specimens from 12 cases in 2011 revealed a variant strain, called H3N2v, which is a hybrid containing genetic material from swine H3N2 and the 2009 human pandemic virus H1N1pdm09. The M gene in this new variant came from the human virus, while the other seven came from the swine virus when a host was infected with both viruses simultaneously (Figure 1). As a result of this genetic reassortment, this variant virus is genetically and antigenically different from seasonal H3N2.
Epidemiologic data showed that children under 10 years of age are especially susceptible to this new variant because they lack immunity, whereas adolescents and adults may have some immunity from cross-reacting antibodies.7 Most infected people had been exposed to swine in agriculture, including county and state fairs. So far, evidence suggests only limited human-to-human transmission.8 The clinical diagnosis of H3N2v infection relies on the epidemiologic link to exposure to pigs in the week before the onset of illness, since the symptoms are indistinguishable from those of seasonal influenza A or B infections.
In suspected cases, the clinician should notify the local or state public health department and arrange for a special test to be performed on respiratory specimens: the CDC Flu Real-Time Reverse Transcriptase Polymerase Chain Reaction Dx Panel. The reason is that a negative rapid influenza diagnostic test does not rule out influenza infection, and a positive immunofluorescence assay (direct fluorescent antibody staining) cannot specifically detect H3N2v.7
The current seasonal influenza vaccine will not protect against H3N2v. The isolates tested to date were susceptible to the neuraminidase inhibitor drugs oseltamivir (Tamiflu) and zanamivir (Relenza) but resistant to amantadine (Symmetrel) and rimantadine (Flumadine).9
Whether H3N2v will become a significant problem during the upcoming flu season largely depends on the extent of human-to-human transmission. We need to closely follow updates on this virus.
H5N1: THE LOOMING THREAT OF A BIRD FLU PANDEMIC
Since 2003, influenza A H5N1, a highly pathogenic avian virus, has broken out in Asia, Africa, and the Middle East, killing more than 100 million birds. It also has crossed the species barrier to infect humans, with an unusually high death rate.10
As of August 10, 2012, the World Health Organization had reported 608 confirmed cases of this virus infecting humans and 359 associated deaths.11 Most infected patients had a history of close contact with diseased poultry, but limited, nonsustained human-to-human transmission can occur during very close, unprotected contact with a severely ill patient.12
Molecular studies of this virus revealed further insights into its pathogenesis. Some of the viruses isolated from humans have had mutations that allow them to bind to human-type receptors.13 Amino acid substitutions in the polymerase basic protein 2 (PB2) gene are associated with mammalian adaptation, virulence in mice, and viral replication at temperatures present in the upper respiratory tract.14 Furthermore, higher plasma levels of macrophage- and neutrophil-attractant chemokines and both inflammatory and anti-inflammatory cytokines (interleukin 6, interleukin 10, and interferon gamma) have been observed in patients with H5N1 infection, especially in fatal cases.15 A recent study found that H5N1 causes significant perturbations in the host’s protein synthesis machinery as early as 1 hour after infection, suggesting that this virus gains an early advantage in replication by using the host’s proteome.16 The effects of unrestrained viral infection and inflammatory responses induced by H5N1 infection certainly contributed to the primary pathologic process and to death in human fulminant viral pneumonia. The up-regulation of inflammatory cytokines in these infections contributes to the development of sepsis syndrome, acute respiratory distress syndrome, and an increased risk of death, particularly in pregnant women.
Most experts predict that pandemic influenza is probably inevitable.17 If avian H5N1 and a human influenza virus swap genes in a host such as swine, the new hybrid virus will contain genetic material from both strains and will have surface antigens that the human immune system does not recognize. This could lead to a devastating avian flu pandemic with a very high death rate.18
An inactivated whole-virus H5N1 vaccine has been developed by the US government to prevent H5N1 infection.19 For treatment, the neuraminidase inhibitor oseltamivir is the drug of choice.10 Oseltamivir resistance remains uncommon. 20 Fortunately, zanamivir is still active against oseltamivir-resistant variants that have N1 neuraminidase mutations.21
THE 2009 H1N1 PANDEMIC KILLED MORE PEOPLE THAN WE THOUGHT
The fourth flu pandemic of the last 100 years occurred in 2009. (The other three were in 1918, 1957, and 1968.) It was caused by a novel strain, H1N1 of swine origin.22 This 2009 pandemic strain had six genes from the North American swine flu virus and two genes from the Eurasian swine flu virus. The pandemic affected more children and young people (who completely lacked prior immunity to this virus), while older people, who had cross-reacting antibodies, were less affected.
Worldwide, 18,500 people were reported initially to have died in this pandemic from April 2009 to August 2010.23 However, a recent modeling study estimated the number of respiratory and cardiovascular deaths associated with this pandemic at 283,500—about 15 times higher.24
AN AUSTRALIAN OUTBREAK OF OSELTAMIVIR-RESISTANT H1N1
Many strains of influenza A virus are resistant to amantadine and rimantadine, owing to amino acid substitutions in the M2 protein.25 In contrast, resistance to the neuraminidase inhibitors oseltamivir and zanamivir has been reported only occasionally.26
Until recently, most oseltamivir-resistant viruses were isolated from immunocompromised hosts treated with oseltamivir.27–29 All the resistant viral isolates contained an amino acid substitution of histidine (H) to tyrosine (Y) at position 275 of the viral neuraminidase.30 In general, transmission of these oseltamivir-resistant strains has been limited and unsustained, but it can occur in settings of close contact, such as hospitals, school camps, or long train rides.31–35 Oseltamivir-resistant strains were detected in fewer than 1% of isolates from the community during the 2010–2011 influenza season in the Northern Hemisphere and most countries in the Southern Hemisphere during the 2011 flu season.36,37
However, an outbreak of oseltamivir-resistant H1N1 occurred in Australia between June and August 2011.38 In that outbreak, the isolates from only 15% of the 191 people infected with this virus, designated H1N1pdm09, carried the H257Y neuraminidase substitution.39 Further, only 1 of the 191 patients had received oseltamivir before. More importantly, genetic analysis suggested that the infection spread from a single source.
This was the first reported sustained community transmission of oseltamivir-resistant H1N1 in a community previously unexposed to this drug. As such, it is a warning sign of the potential for a widespread outbreak of this virus. In the event of such an outbreak, inhaled zanamivir would be the only effective treatment available.
THIS SEASON’S TRIVALENT INACTIVATED VACCINE
The trivalent inactivated influenza vaccine for the 2012–2013 season contains three inactivated viruses40:
- Influenza A/California/7/2009(H1N1)-like
- Influenza A/Victoria/361/2011(H3N2)-like
- Influenza B/Wisconsin/1/2010-like (Yamagata lineage).
The influenza A H3N2 and influenza B antigens are different from those in the 2011–2012 vaccine.41 The H1N1 strain is derived from H1N1pdm09, which had been contained in the 2011–2012 seasonal vaccine. This vaccine will not protect against H3N2v or H5N1.
LATEST RECOMMENDATIONS ON VACCINATION
Since 2010, the Advisory Committee on Immunization Practices (ACIP) has recommended annual flu shots for all people older than 6 months in the United States.42
Vaccination should be done before the onset of influenza activity in the community as soon as vaccine is available for the season. However, one should continue offering vaccination throughout the influenza season as long as influenza viruses are circulating in the community.
Children ages 6 months through 8 years not previously vaccinated against influenza should receive two doses of influenza vaccine at least 4 weeks apart for an optimal immune response. The US-licensed Afluria vaccine (CSL Biotherapies, King of Prussia, PA), a trivalent inactivated vaccine, is not recommended for children under 9 years of age because of concern about febrile seizures.43,44
There is no contraindication to giving inactivated trivalent influenza vaccine to immunosuppressed people.
The live-attenuated influenza vaccine is indicated only for healthy, nonpregnant people age 2 through 49 years and not for people who care for severely immunosuppressed patients who require a protective environment.
For indications for and details about the different available influenza vaccines, see the ACIP’s current recommendations (www.cdc.gov/mmwr/pdf/wk/mm6132.pdf).40
Updated recommendations for people allergic to eggs
All currently available influenza vaccines are made by growing the virus in chicken eggs. Therefore, severe allergic and anaphylactic reactions can occur in people with egg allergy. The ACIP recommends that if people experienced only hives after egg exposure, they should still receive the trivalent inactivated vaccine. Recently, the ACIP reviewed data from the Vaccine Adverse Event Reporting System45 and issued the following recommendations for the 2012–2013 influenza season40:
- In people who are allergic to eggs, only trivalent inactivated vaccine should be used, not the live-attenuated vaccine, because of lack of data for use of the latter in this group.
- Vaccine should be given by providers who are familiar with the signs of egg allergy.
- Patients with a history of egg allergy who have experienced only hives after exposure to eggs should be observed for a minimum of 30 minutes after vaccination.
- Patients who experience lightheadedness, respiratory distress, angioedema, or recurrent emesis or who require epinephrine or emergency medical attention after egg exposure should be referred before vaccination to a physician who has expertise in managing allergic conditions.
- Tolerance to egg-containing foods does not exclude the possibility of egg allergy. Egg allergy can be confirmed by a consistent medical history of adverse reactions to eggs or egg-containing foods, plus skin or blood testing for immunoglobulin E antibodies to egg proteins.
A high-dose vaccine is available for people 65 years and older
The rates of hospitalization and death due to seasonal flu in elderly people have increased significantly in the last 20 years despite rising rates of vaccination.46–48 This is largely due to lower serologic response rates and vaccine efficacy in older adults with weaker immune systems.
Several studies have shown that the development of protective antibody titers depends on the dose of antigen.49–53 A randomized, controlled clinical trial compared the immunogenicity of a high-dose vaccine and a standard-dose vaccine in older adults and found that the level of antibody response was significantly higher with the high-dose vaccine, and that the rate of adverse reactions was the same.54
In December 2009, the US Food and Drug Administration (FDA) licensed a new trivalent inactivated influenza vaccine with high doses of hemagglutinin antigens for adults over the age of 65.55 Postlicensure safety surveillance in 2010 revealed no serious safety concerns.56
At present, the ACIP expresses no preference for standard-dose or high-dose vaccine for adults 65 years of age and older.40 Importantly, if only the standard-dose vaccine is at hand, the opportunity for influenza vaccination should not be missed with the intention of giving high-dose vaccine at a later date.
A NEW QUADRIVALENT LIVE-ATTENUATED INFLUENZA VACCINE FOR THE 2013–2014 SEASON
In February 2012, the FDA approved the first quadrivalent live-attenuated influenza vaccine, which is expected to replace the currently available trivalent live-attenuated influenza vaccine in the 2013–2014 flu season. The quadrivalent vaccine will include both lineages of the circulating influenza B viruses (the Victoria and Yamagata lineages). The reasons for this inclusion is the difficulty in predicting which of these viruses will predominate in any given season, and the limited cross-resistance by immunization against one of the lineages.
A recent analysis57 estimated that such a vaccine is likely to further reduce influenza cases, related hospitalizations, and deaths compared with the current trivalent vaccine. Like the current trivalent live-attenuated vaccine, the quadrivalent vaccine is inhaled.
EVOLVING VACCINATION POLICY IN HEALTH CARE WORKERS
Starting in January 2013, the Centers for Medicare and Medicaid Services will require hospitals to report how many of their health care workers are vaccinated. These rates will be publicly reported as a measure of hospital quality. This has fueled the ongoing debate about mandating influenza vaccination for health care workers. Studies have shown that the most important factors in increasing influenza vaccination rates among health care workers are requiring vaccination as a condition for employment and making vaccination available on-site, for more than 1 day, at no cost to the worker.58
As an alternative, some institutions have implemented a “shot-or-mask” policy whereby a health care worker who elects not to be vaccinated because of medical or religious reasons would be asked to wear a mask during all faceto-face encounters with patients.
NEW ANTIVIRAL DRUGS ON THE HORIZON
The emergence of oseltamivir-resistant strains in recent years caused a great deal of concern in public health regarding the potential for outbreaks of drug-resistant influenza.34,35,59–61
A recent Asian randomized clinical trial reported the efficacy of a long-acting neuraminidase inhibitor, laninamivir octanoate, in the treatment of seasonal influenza.62 This study showed that a single inhalation of this drug is effective in treating seasonal influenza, including that caused by oseltamivir-resistant strains in adults. Laninamivir is currently approved in Japan.
CHALLENGES IN PREVENTING AND TREATING INFLUENZA
Despite the great advances that we have made in preventing and treating influenza in the last half-century, we still face many challenges. Each year in the United States, influenza infection results in an estimated 31 million outpatient visits, 226,000 hospital admissions, and 36,000 deaths.42
Antigenic drift and shift. Influenza viruses circulating among animals and humans vary genetically from season to season and within seasons. As a result of this changing viral antigenicity, the virus can evade the human immune system, causing widespread outbreaks.
One of the unique and most remarkable features of influenza virus is the antigenic variation: antigenic drift and antigenic shift. Antigenic drift is the relatively minor antigenic changes that occur frequently within an influenza subtype, typically resulting from genetic mutation of viral RNA coding for hemagglutinin or neuraminidase. This causes annual regional epidemics. In contrast, antigenic shift is the result of genetic material reassortment: the emerging of new viral RNA from different strains of different species. This often leads to global pandemics.
Therefore, it is challenging to accurately predict the antigenic makeup of influenza viruses for each season and to include new emerging antigens in the vaccine production, as we are facing a moving target. We prepare influenza vaccines each season based on past experience.63
Vaccination rates have hit a plateau of 60% to 70% in adults since the 1990s, in spite of greater vaccine supply and recommendations that all adults, regardless of underlying disease, be vaccinated annually.64 Similarly, only 51% of children age 6 months to 17 years were vaccinated in the 2010–2011 season.65 And vaccination rates are even lower in low-income populations.66,67
The emergence of oseltamivir-resistant strains in recent years, not only in people exposed to oseltamivir but also in those who haven’t been exposed to this drug, with sustained transmission, certainly raises the possibility of a more difficult epidemic to control.38
Global travel, global infection. Our last H1N1 pandemic in 2009 was an example of how easily the influenza virus can spread rapidly in today’s highly mobile global society.22
What we must do
As primary health care providers, we must closely monitor the community outbreak and the emergence of drug-resistant strains and strongly recommend vaccination for all patients older than 6 months, since timely vaccination is the cornerstone of influenza prevention. Although many have questioned the efficacy of influenza vaccination, a recent meta-analysis showed a 59% pooled efficacy of the trivalent inactivated vaccine in adults age 18 to 65 years in preventing virologically confirmed influenza, and 83% pooled efficacy of the live-attenuated influenza vaccine in children age 6 months to 7 years.68 Novel approaches for vaccination reminders, such as text messaging69 in addition to traditional mail or telephone reminders, can improve vaccination compliance in today’s highly mobile world that is more than ever connected.
With the lessons learned from four pandemics in the last century, updated recommendations for prevention, and adequate vaccine supply, we should be ready to face the challenge of another flu season.
- Doshi P. Trends in recorded influenza mortality: United States, 1900–2004. Am J Public Health 2008; 98:939–945.
- Centers for Disease Control and Prevention (CDC). Estimates of deaths associated with seasonal influenza — United States, 1976–2007. MMWR Morb Mortal Wkly Rep 2010; 59:1057–1062.
- Reid AH, Taubenberger JK, Fanning TG. Evidence of an absence: the genetic origins of the 1918 pandemic influenza virus. Nat Rev Microbiol 2004; 2:909–914.
- Lindstrom S, Garten R, Balish A, et al. Human infections with novel reassortant influenza A(H3N2)v viruses, United States, 2011. Emerg Infect Dis 2012; 18:834–837.
- Shu B, Garten R, Emery S, et al. Genetic analysis and antigenic characterization of swine origin influenza viruses isolated from humans in the United States, 1990–2010. Virology 2012; 422:151–160.
- Centers for Disease Control and Prevention (CDC). http://www.cdc.gov/flu/swineflu/h3n2v-outbreak.htm. Accessed September 27, 2012.
- Centers for Disease Control and Prevention (CDC). Evaluation of rapid influenza diagnostic tests for influenza A (H3N2)v virus and updated case count — United States, 2012. MMWR Morb Mortal Wkly Rep 2012; 61:619–621.
- Centers for Disease Control and Prevention (CDC). Update: Influenza A (H3N2)v transmission and guidelines — five states, 2011. MMWR Morb Mortal Wkly Rep 2012; 60:1741–1744.
- Centers for Disease Control and Prevention (CDC). Interim information for clinicians about human infections with H3N2v virus. http://www.cdc.gov/flu/swineflu/h3n2v-clinician.htm. Accessed September 27, 2012.
- Writing Committee of the Second World Health Organization Consultation on Clinical Aspects of Human Infection with Avian Influenza A (H5N1) Virus; Abdel-Ghafar AN, Chotpitayasunondh T, Gao Z, et al. Update on avian influenza A (H5N1) virus infection in humans. N Engl J Med 2008; 358:261–273.
- World Health Organization (WHO). http://www.who.int/influenza/human_animal_interface/H5N1_cumulative_table_archives/en/index.html. Accessed September 27, 2012.
- Ungchusak K, Auewarakul P, Dowell SF, et al. Probable person-to-person transmission of avian influenza A (H5N1). N Engl J Med 2005; 352:333–340.
- Yamada S, Suzuki Y, Suzuki T, et al. Haemagglutinin mutations responsible for the binding of H5N1 influenza A viruses to human-type receptors. Nature 2006; 444:378–382.
- Hatta M, Hatta Y, Kim JH, et al. Growth of H5N1 influenza A viruses in the upper respiratory tracts of mice. PLoS Pathog 2007; 3:1374–1379.
- de Jong MD, Simmons CP, Thanh TT, et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med 2006; 12:1203–1207.
- Cheung CY, Chan EY, Krasnoselsky A, et al. H5N1 virus causes significant perturbations in host proteome very early in influenza virus-infected primary human monocyte-derived macrophages. J Infect Dis 2012; 206:640–645.
- Gordon S. Avian influenza: a wake-up call from birds to humans. Cleve Clin J Med 2004; 71:273–274.
- Jin XW, Mossad SB. Avian influenza: an emerging pandemic threat. Cleve Clin J Med 2005; 72:1129–1234.
- Ehrlich HJ, Müller M, Oh HM, et al; Baxter H5N1 Pandemic Influenza Vaccine Clinical Study Team. A clinical trial of a whole-virus H5N1 vaccine derived from cell culture. N Engl J Med 2008; 358:2573–2584.
- de Jong MD, Tran TT, Truong HK, et al. Oseltamivir resistance during treatment of influenza A (H5N1) infection. N Engl J Med 2005; 353:2667–2672.
- Le QM, Kiso M, Someya K, et al. Avian flu: isolation of drug-resistant H5N1 virus. Nature 2005; 437:1108.
- Ison MG, Lee N. Influenza 2010–2011: lessons from the 2009 pandemic. Cleve Clin J Med 2010; 77:812–820.
- World Health Organization (WHO). Pandemic (H1N1) 2009 — update 112. http://www.who.int/csr/don/2010_08_06/en/index.html. Accessed September 27, 2012.
- Dawood FS, Iuliano AD, Reed C, et al. Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: a modelling study. Lancet Infect Dis 2012; 12:687–695.
- Bright RA, Shay DK, Shu B, Cox NJ, Klimov AI. Adamantane resistance among influenza A viruses isolated early during the 2005–2006 influenza season in the United States. JAMA 2006; 295:891–894.
- Nguyen HT, Fry AM, Gubareva LV. Neuraminidase inhibitor resistance in influenza viruses and laboratory testing methods. Antivir Ther 2012; 17:159–173.
- Graitcer SB, Gubareva L, Kamimoto L, et al. Characteristics of patients with oseltamivir-resistant pandemic (H1N1) 2009, United States. Emerg Infect Dis 2011; 17:255–257.
- Hurt AC, Deng YM, Ernest J, et al. Oseltamivir-resistant influenza viruses circulating during the first year of the influenza A(H1N1) 2009 pandemic in the Asia-Pacific region, March 2009 to March 2010. Euro Surveill 2011; 16:19770.
- Meijer A, Jonges M, Abbink F, et al. Oseltamivir-resistant pandemic A(H1N1) 2009 influenza viruses detected through enhanced surveillance in the Netherlands, 2009–2010. Antiviral Res 2011; 92:81–89.
- Gubareva LV, Kaiser L, Hayden FG. IInfluenza virus neuraminidase inhibitors. Lancet 2000; 355:827–835.
- Wolfe C, Greenwald I, Chen L. Pandemic (H1N1) 2009 and oseltamivir resistance in hematology/oncology patients. Emerg Infect Dis 2010; 16:1809–1811.
- Moore C, Galiano M, Lackenby A, et al. Evidence of person-to-person transmission of oseltamivir-resistant pandemic influenza A(H1N1) 2009 virus in a hematology unit. J Infect Dis 2011; 203:18–24.
- Chen LF, Dailey NJ, Rao AK, et al. Cluster of oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infections on a hospital ward among immunocompromised patients — North Carolina, 2009. J Infect Dis 2011; 203:838–846.
- Centers for Disease Control and Prevention (CDC). Oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infection in two summer campers receiving prophylaxis — North Carolina, 2009. MMWR Morb Mortal Wkly Rep 2009; 58:969–972.
- Le QM, Wertheim HF, Tran ND, van Doorn HR, Nguyen TH, Horby P; Vietnam H1N1 Investigation Team. A community cluster of oseltamivir-resistant cases of 2009 H1N1 influenza. N Engl J Med 2010; 362:86–87.
- Lackenby A, Moran Gilad J, Pebody R, et al. Continued emergence and changing epidemiology of oseltamivir-resistant influenza A(H1N1)2009 virus, United Kingdom, winter 2010/11. Euro Surveill 2011; 16:19784.
- World Health Organization (WHO). Summary of influenza antiviral susceptibility surveillance findings, September 2010 – March 2011. http://www.who.int/influenza/gisrs_laboratory/updates/antiviral_susceptibility/en/index.html. Accessed September 27, 2012.
- Hurt AC, Hardie K, Wilson NJ, et al. Community transmission of oseltamivir-resistant A(H1N1)pdm09 influenza. N Engl J Med 2011; 365:2541–2542.
- Hurt AC, Hardie K, Wilson NJ, et al. Characteristics of a widespread community cluster of H275Y oseltamivir-resistant A(H1N1)pdm09 influenza in Australia. J Infect Dis 2012; 206:148–157.
- Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP) — United States, 2012–13 Influenza Season. MMWR Morb Mortal Wkly Rep 2012; 61:613–618.
- Food and Drug Administration (FDA). Summary minutes: vaccines and related biological products advisory committee. February 28–29, 2012. Silver Spring, MD. http://www.fda.gov/downloads/Advisory-Committees/CommitteesMeetingMaterials/BloodVaccinesandOther-Biologics/VaccinesandRelatedBiologicalProductsAdvisoryCommittee/UCM296193.pdf. Accessed September 28, 2012.
- Fiore AE, Uyeki TM, Broder K, et al; Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Recomm Rep 2010; 59:1–62.
- Centers for Disease Control and Prevention (CDC). Update: recommendations of the Advisory Committee on Immunization Practices (ACIP) regarding use of CSL seasonal influenza vaccine (Afluria) in the United States during 2010–11. MMWR Morb Mortal Wkly Rep 2010; 59:989–992.
- Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2011. MMWR Morb Mortal Wkly Rep 2011; 60:1128–1132.
- Centers for Disease Control and Prevention (CDC). Advisory Committee on Immunization Practices: Update on influenza vaccine safety monitoring. June 20–21, 2012. Atlanta, GA. http://www.cdc.gov/vaccines/acip/meetings/downloads/slides-jun-2012/03-influenza-Shimabukuro.pdf. Accessed September 28, 2012.
- Simonsen L, Reichert TA, Viboud C, Blackwelder WC, Taylor RJ, Miller MA. Impact of influenza vaccination on seasonal mortality in the US elderly population. Arch Intern Med 2005; 165:265–272.
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004; 292:1333–1340.
- Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003; 289:179–186.
- Mostow SR, Schoenbaum SC, Dowdle WR, Coleman MT, Kaye HS. Inactivated vaccines. 1. Volunteer studies with very high doses of influenza vaccine purified by zonal ultracentrifugation. Postgrad Med J 1973; 49:152–158.
- Keitel WA, Atmar RL, Cate TR, et al. Safety of high doses of influenza vaccine and effect on antibody responses in elderly persons. Arch Intern Med 2006; 166:1121–1127.
- Ruben FL, Jackson GG. A new subunit influenza vaccine: acceptability compared with standard vaccines and effect of dose on antigenicity. J Infect Dis 1972; 125:656–664.
- Palache AM, Beyer WE, Sprenger MJ, et al. Antibody response after influenza immunization with various vaccine doses: a double-blind, placebo-controlled, multi-centre, dose-response study in elderly nursing-home residents and young volunteers. Vaccine 1993; 11:3–9.
- Couch RB, Winokur P, Brady R, et al. Safety and immunogenicity of a high dosage trivalent influenza vaccine among elderly subjects. Vaccine 2007; 25:7656–7663.
- Falsey AR, Treanor JJ, Tornieporth N, Capellan J, Gorse GJ. Randomized, double-blind controlled phase 3 trial comparing the immunogenicity of high-dose and standard-dose influenza vaccine in adults 65 years of age and older. J Infect Dis 2009; 200:172–180.
- US Food and Drug Administration. Vaccines, Blood & Biologics. December 23,2009 approval letter—Fluzone high-dose. http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm195481.htm. Accessed October 1, 2012.
- Moro PL, Arana J, Cano M, et al. Postlicensure safety surveillance for high-dose trivalent inactivated influenza vaccine in the Vaccine Adverse Event Reporting System, 1 July 2010–31 December 2010. Clin Infect Dis 2012; 54:1608–1614.
- Reed C, Meltzer MI, Finelli L, Fiore A. Public health impact of including two lineages of influenza B in a quadrivalent seasonal influenza vaccine. Vaccine 2012; 30:1993–1998.
- Centers for Disease Control and Prevention (CDC). Influenza vaccination coverage among health-care personnel — United States, 2010–11 influenza season. MMWR Morb Mortal Wkly Rep 2011; 60:1073–1077.
- Meijer A, Lackenby A, Hungnes O, et al; European Influenza Surveillance Scheme. Oseltamivir-resistant influenza virus A (H1N1), Europe, 2007–08 season. Emerg Infect Dis 2009; 15:552–560.
- Moscona A. Global transmission of oseltamivir-resistant influenza. N Engl J Med 2009; 360:953–956.
- World Health Organization (WHO). Influenza A virus resistance to oseltamivir. http://www.who.int/influenza/patient_care/antivirals/oseltamivir_summary/en/. Accessed September 28, 2012.
- Watanabe A, Chang SC, Kim MJ, Chu DW, Ohashi Y; MARVEL Study Group. Long-acting neuraminidase inhibitor laninamivir octanoate versus oseltamivir for treatment of influenza: a double-blind, randomized, noninferiority clinical trial. Clin Infect Dis 2010; 51:1167–1175.
- Deyde VM, Gubareva LV. Influenza genome analysis using pyro-sequencing method: current applications for a moving target. Expert Rev Mol Diagn 2009; 9:493–509.
- Schuchat A, Katz JM. Protecting adults from influenza: tis the season to learn from the pandemic. J Infect Dis 2012; 206:803–805.
- Centers for Disease Control and Prevention (CDC). Final state-level influenza vaccination coverage estimates for the 2010–11 season — United States, National Immunization Survey and Behavioral Risk Factor Surveillance System, August 2010 through May 2011. http://www.cdc.gov/flu/professionals/vaccination/coverage_1011estimates.htm. Accessed September 28, 2012.
- Bhatt P, Block SL, Toback SL, Ambrose CS. Timing of the availability and administration of influenza vaccine through the vaccines for children program. Pediatr Infect Dis J 2011; 30:100–106.
- Lee BY, Brown ST, Bailey RR, et al. The benefits to all of ensuring equal and timely access to influenza vaccines in poor communities. Health Aff (Millwood) 2011; 30:1141–1150.
- Osterholm MT, Kelley NS, Sommer A, Belongia EA. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis 2012; 12:36–44.
- Stockwell MS, Kharbanda EO, Martinez RA, Vargas CY, Vawdrey DK, Camargo S. Effect of a text messaging intervention on influenza vaccination in an urban, low-income pediatric and adolescent population: a randomized controlled trial. JAMA 2012; 307:1702–1708.
- Doshi P. Trends in recorded influenza mortality: United States, 1900–2004. Am J Public Health 2008; 98:939–945.
- Centers for Disease Control and Prevention (CDC). Estimates of deaths associated with seasonal influenza — United States, 1976–2007. MMWR Morb Mortal Wkly Rep 2010; 59:1057–1062.
- Reid AH, Taubenberger JK, Fanning TG. Evidence of an absence: the genetic origins of the 1918 pandemic influenza virus. Nat Rev Microbiol 2004; 2:909–914.
- Lindstrom S, Garten R, Balish A, et al. Human infections with novel reassortant influenza A(H3N2)v viruses, United States, 2011. Emerg Infect Dis 2012; 18:834–837.
- Shu B, Garten R, Emery S, et al. Genetic analysis and antigenic characterization of swine origin influenza viruses isolated from humans in the United States, 1990–2010. Virology 2012; 422:151–160.
- Centers for Disease Control and Prevention (CDC). http://www.cdc.gov/flu/swineflu/h3n2v-outbreak.htm. Accessed September 27, 2012.
- Centers for Disease Control and Prevention (CDC). Evaluation of rapid influenza diagnostic tests for influenza A (H3N2)v virus and updated case count — United States, 2012. MMWR Morb Mortal Wkly Rep 2012; 61:619–621.
- Centers for Disease Control and Prevention (CDC). Update: Influenza A (H3N2)v transmission and guidelines — five states, 2011. MMWR Morb Mortal Wkly Rep 2012; 60:1741–1744.
- Centers for Disease Control and Prevention (CDC). Interim information for clinicians about human infections with H3N2v virus. http://www.cdc.gov/flu/swineflu/h3n2v-clinician.htm. Accessed September 27, 2012.
- Writing Committee of the Second World Health Organization Consultation on Clinical Aspects of Human Infection with Avian Influenza A (H5N1) Virus; Abdel-Ghafar AN, Chotpitayasunondh T, Gao Z, et al. Update on avian influenza A (H5N1) virus infection in humans. N Engl J Med 2008; 358:261–273.
- World Health Organization (WHO). http://www.who.int/influenza/human_animal_interface/H5N1_cumulative_table_archives/en/index.html. Accessed September 27, 2012.
- Ungchusak K, Auewarakul P, Dowell SF, et al. Probable person-to-person transmission of avian influenza A (H5N1). N Engl J Med 2005; 352:333–340.
- Yamada S, Suzuki Y, Suzuki T, et al. Haemagglutinin mutations responsible for the binding of H5N1 influenza A viruses to human-type receptors. Nature 2006; 444:378–382.
- Hatta M, Hatta Y, Kim JH, et al. Growth of H5N1 influenza A viruses in the upper respiratory tracts of mice. PLoS Pathog 2007; 3:1374–1379.
- de Jong MD, Simmons CP, Thanh TT, et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med 2006; 12:1203–1207.
- Cheung CY, Chan EY, Krasnoselsky A, et al. H5N1 virus causes significant perturbations in host proteome very early in influenza virus-infected primary human monocyte-derived macrophages. J Infect Dis 2012; 206:640–645.
- Gordon S. Avian influenza: a wake-up call from birds to humans. Cleve Clin J Med 2004; 71:273–274.
- Jin XW, Mossad SB. Avian influenza: an emerging pandemic threat. Cleve Clin J Med 2005; 72:1129–1234.
- Ehrlich HJ, Müller M, Oh HM, et al; Baxter H5N1 Pandemic Influenza Vaccine Clinical Study Team. A clinical trial of a whole-virus H5N1 vaccine derived from cell culture. N Engl J Med 2008; 358:2573–2584.
- de Jong MD, Tran TT, Truong HK, et al. Oseltamivir resistance during treatment of influenza A (H5N1) infection. N Engl J Med 2005; 353:2667–2672.
- Le QM, Kiso M, Someya K, et al. Avian flu: isolation of drug-resistant H5N1 virus. Nature 2005; 437:1108.
- Ison MG, Lee N. Influenza 2010–2011: lessons from the 2009 pandemic. Cleve Clin J Med 2010; 77:812–820.
- World Health Organization (WHO). Pandemic (H1N1) 2009 — update 112. http://www.who.int/csr/don/2010_08_06/en/index.html. Accessed September 27, 2012.
- Dawood FS, Iuliano AD, Reed C, et al. Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: a modelling study. Lancet Infect Dis 2012; 12:687–695.
- Bright RA, Shay DK, Shu B, Cox NJ, Klimov AI. Adamantane resistance among influenza A viruses isolated early during the 2005–2006 influenza season in the United States. JAMA 2006; 295:891–894.
- Nguyen HT, Fry AM, Gubareva LV. Neuraminidase inhibitor resistance in influenza viruses and laboratory testing methods. Antivir Ther 2012; 17:159–173.
- Graitcer SB, Gubareva L, Kamimoto L, et al. Characteristics of patients with oseltamivir-resistant pandemic (H1N1) 2009, United States. Emerg Infect Dis 2011; 17:255–257.
- Hurt AC, Deng YM, Ernest J, et al. Oseltamivir-resistant influenza viruses circulating during the first year of the influenza A(H1N1) 2009 pandemic in the Asia-Pacific region, March 2009 to March 2010. Euro Surveill 2011; 16:19770.
- Meijer A, Jonges M, Abbink F, et al. Oseltamivir-resistant pandemic A(H1N1) 2009 influenza viruses detected through enhanced surveillance in the Netherlands, 2009–2010. Antiviral Res 2011; 92:81–89.
- Gubareva LV, Kaiser L, Hayden FG. IInfluenza virus neuraminidase inhibitors. Lancet 2000; 355:827–835.
- Wolfe C, Greenwald I, Chen L. Pandemic (H1N1) 2009 and oseltamivir resistance in hematology/oncology patients. Emerg Infect Dis 2010; 16:1809–1811.
- Moore C, Galiano M, Lackenby A, et al. Evidence of person-to-person transmission of oseltamivir-resistant pandemic influenza A(H1N1) 2009 virus in a hematology unit. J Infect Dis 2011; 203:18–24.
- Chen LF, Dailey NJ, Rao AK, et al. Cluster of oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infections on a hospital ward among immunocompromised patients — North Carolina, 2009. J Infect Dis 2011; 203:838–846.
- Centers for Disease Control and Prevention (CDC). Oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infection in two summer campers receiving prophylaxis — North Carolina, 2009. MMWR Morb Mortal Wkly Rep 2009; 58:969–972.
- Le QM, Wertheim HF, Tran ND, van Doorn HR, Nguyen TH, Horby P; Vietnam H1N1 Investigation Team. A community cluster of oseltamivir-resistant cases of 2009 H1N1 influenza. N Engl J Med 2010; 362:86–87.
- Lackenby A, Moran Gilad J, Pebody R, et al. Continued emergence and changing epidemiology of oseltamivir-resistant influenza A(H1N1)2009 virus, United Kingdom, winter 2010/11. Euro Surveill 2011; 16:19784.
- World Health Organization (WHO). Summary of influenza antiviral susceptibility surveillance findings, September 2010 – March 2011. http://www.who.int/influenza/gisrs_laboratory/updates/antiviral_susceptibility/en/index.html. Accessed September 27, 2012.
- Hurt AC, Hardie K, Wilson NJ, et al. Community transmission of oseltamivir-resistant A(H1N1)pdm09 influenza. N Engl J Med 2011; 365:2541–2542.
- Hurt AC, Hardie K, Wilson NJ, et al. Characteristics of a widespread community cluster of H275Y oseltamivir-resistant A(H1N1)pdm09 influenza in Australia. J Infect Dis 2012; 206:148–157.
- Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP) — United States, 2012–13 Influenza Season. MMWR Morb Mortal Wkly Rep 2012; 61:613–618.
- Food and Drug Administration (FDA). Summary minutes: vaccines and related biological products advisory committee. February 28–29, 2012. Silver Spring, MD. http://www.fda.gov/downloads/Advisory-Committees/CommitteesMeetingMaterials/BloodVaccinesandOther-Biologics/VaccinesandRelatedBiologicalProductsAdvisoryCommittee/UCM296193.pdf. Accessed September 28, 2012.
- Fiore AE, Uyeki TM, Broder K, et al; Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Recomm Rep 2010; 59:1–62.
- Centers for Disease Control and Prevention (CDC). Update: recommendations of the Advisory Committee on Immunization Practices (ACIP) regarding use of CSL seasonal influenza vaccine (Afluria) in the United States during 2010–11. MMWR Morb Mortal Wkly Rep 2010; 59:989–992.
- Centers for Disease Control and Prevention (CDC). Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2011. MMWR Morb Mortal Wkly Rep 2011; 60:1128–1132.
- Centers for Disease Control and Prevention (CDC). Advisory Committee on Immunization Practices: Update on influenza vaccine safety monitoring. June 20–21, 2012. Atlanta, GA. http://www.cdc.gov/vaccines/acip/meetings/downloads/slides-jun-2012/03-influenza-Shimabukuro.pdf. Accessed September 28, 2012.
- Simonsen L, Reichert TA, Viboud C, Blackwelder WC, Taylor RJ, Miller MA. Impact of influenza vaccination on seasonal mortality in the US elderly population. Arch Intern Med 2005; 165:265–272.
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004; 292:1333–1340.
- Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003; 289:179–186.
- Mostow SR, Schoenbaum SC, Dowdle WR, Coleman MT, Kaye HS. Inactivated vaccines. 1. Volunteer studies with very high doses of influenza vaccine purified by zonal ultracentrifugation. Postgrad Med J 1973; 49:152–158.
- Keitel WA, Atmar RL, Cate TR, et al. Safety of high doses of influenza vaccine and effect on antibody responses in elderly persons. Arch Intern Med 2006; 166:1121–1127.
- Ruben FL, Jackson GG. A new subunit influenza vaccine: acceptability compared with standard vaccines and effect of dose on antigenicity. J Infect Dis 1972; 125:656–664.
- Palache AM, Beyer WE, Sprenger MJ, et al. Antibody response after influenza immunization with various vaccine doses: a double-blind, placebo-controlled, multi-centre, dose-response study in elderly nursing-home residents and young volunteers. Vaccine 1993; 11:3–9.
- Couch RB, Winokur P, Brady R, et al. Safety and immunogenicity of a high dosage trivalent influenza vaccine among elderly subjects. Vaccine 2007; 25:7656–7663.
- Falsey AR, Treanor JJ, Tornieporth N, Capellan J, Gorse GJ. Randomized, double-blind controlled phase 3 trial comparing the immunogenicity of high-dose and standard-dose influenza vaccine in adults 65 years of age and older. J Infect Dis 2009; 200:172–180.
- US Food and Drug Administration. Vaccines, Blood & Biologics. December 23,2009 approval letter—Fluzone high-dose. http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm195481.htm. Accessed October 1, 2012.
- Moro PL, Arana J, Cano M, et al. Postlicensure safety surveillance for high-dose trivalent inactivated influenza vaccine in the Vaccine Adverse Event Reporting System, 1 July 2010–31 December 2010. Clin Infect Dis 2012; 54:1608–1614.
- Reed C, Meltzer MI, Finelli L, Fiore A. Public health impact of including two lineages of influenza B in a quadrivalent seasonal influenza vaccine. Vaccine 2012; 30:1993–1998.
- Centers for Disease Control and Prevention (CDC). Influenza vaccination coverage among health-care personnel — United States, 2010–11 influenza season. MMWR Morb Mortal Wkly Rep 2011; 60:1073–1077.
- Meijer A, Lackenby A, Hungnes O, et al; European Influenza Surveillance Scheme. Oseltamivir-resistant influenza virus A (H1N1), Europe, 2007–08 season. Emerg Infect Dis 2009; 15:552–560.
- Moscona A. Global transmission of oseltamivir-resistant influenza. N Engl J Med 2009; 360:953–956.
- World Health Organization (WHO). Influenza A virus resistance to oseltamivir. http://www.who.int/influenza/patient_care/antivirals/oseltamivir_summary/en/. Accessed September 28, 2012.
- Watanabe A, Chang SC, Kim MJ, Chu DW, Ohashi Y; MARVEL Study Group. Long-acting neuraminidase inhibitor laninamivir octanoate versus oseltamivir for treatment of influenza: a double-blind, randomized, noninferiority clinical trial. Clin Infect Dis 2010; 51:1167–1175.
- Deyde VM, Gubareva LV. Influenza genome analysis using pyro-sequencing method: current applications for a moving target. Expert Rev Mol Diagn 2009; 9:493–509.
- Schuchat A, Katz JM. Protecting adults from influenza: tis the season to learn from the pandemic. J Infect Dis 2012; 206:803–805.
- Centers for Disease Control and Prevention (CDC). Final state-level influenza vaccination coverage estimates for the 2010–11 season — United States, National Immunization Survey and Behavioral Risk Factor Surveillance System, August 2010 through May 2011. http://www.cdc.gov/flu/professionals/vaccination/coverage_1011estimates.htm. Accessed September 28, 2012.
- Bhatt P, Block SL, Toback SL, Ambrose CS. Timing of the availability and administration of influenza vaccine through the vaccines for children program. Pediatr Infect Dis J 2011; 30:100–106.
- Lee BY, Brown ST, Bailey RR, et al. The benefits to all of ensuring equal and timely access to influenza vaccines in poor communities. Health Aff (Millwood) 2011; 30:1141–1150.
- Osterholm MT, Kelley NS, Sommer A, Belongia EA. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis 2012; 12:36–44.
- Stockwell MS, Kharbanda EO, Martinez RA, Vargas CY, Vawdrey DK, Camargo S. Effect of a text messaging intervention on influenza vaccination in an urban, low-income pediatric and adolescent population: a randomized controlled trial. JAMA 2012; 307:1702–1708.
KEY POINTS
- A recent outbreak of swine flu in children exposed to pigs at agricultural fairs is unprecedented. Seasonal influenza vaccine does not protect against this strain, designated H3N2v. The neuraminidase inhibitors oseltamivir (Tamiflu) and zanamivir (Relenza) are the drugs of choice for treatment.
- A highly lethal bird flu, designated H5N1, is still a pandemic threat. In the event of an outbreak, an inactivated whole-virus vaccine is available.
- A community outbreak of oseltamivir-resistant H1N1 in Australia sounded an alarm for a potential drug-resistant flu epidemic. Inhaled zanamivir would be the only effective therapy available in the event of such an epidemic.
- An emerging new antiviral drug is effective against oseltamivir-resistant influenza.