User login
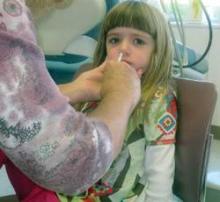
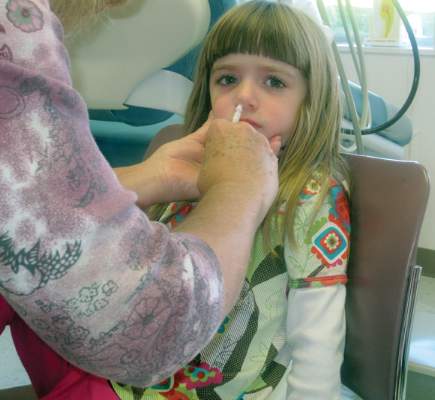
For the next influenza season, there will no longer be a preference for using the nasal spray influenza vaccine over the flu shot for healthy children aged 2-8 years, based on data from recent influenza seasons.
In a 14-0 vote, with one abstention, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) supported the following revision to the influenza vaccine recommendations, for the 2015-2016 influenza season: “For healthy children aged 2 through 8 years who have no contraindications or precautions, either LAIV [live attenuated influenza vaccine] or IIV [inactivated influenza vaccine] is an appropriate option. No preference is expressed for LAIV or IIV or any person aged 2 though 49 years for whom either vaccine is appropriate.”
The recommendation that all people aged 6 months and older should receive the influenza vaccine every year remains unchanged.
Currently, the recommendation is that when “immediately available, LAIV should be used for healthy children aged 2 through 8 years who have no contraindications or precautions,” which was approved in June 2014. This was based on evidence during several influenza seasons that LAIV provided better protection than the IIV for children in this age group. The decision to make the change regarding preference “was made based on new data from more recent seasons, which have not confirmed superior effectiveness of LAIV observed in earlier studies,” according to a statement issued by the CDC following the meeting.
For the next influenza season, there will no longer be a preference for using the nasal spray influenza vaccine over the flu shot for healthy children aged 2-8 years, based on data from recent influenza seasons.
In a 14-0 vote, with one abstention, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) supported the following revision to the influenza vaccine recommendations, for the 2015-2016 influenza season: “For healthy children aged 2 through 8 years who have no contraindications or precautions, either LAIV [live attenuated influenza vaccine] or IIV [inactivated influenza vaccine] is an appropriate option. No preference is expressed for LAIV or IIV or any person aged 2 though 49 years for whom either vaccine is appropriate.”
The recommendation that all people aged 6 months and older should receive the influenza vaccine every year remains unchanged.
Currently, the recommendation is that when “immediately available, LAIV should be used for healthy children aged 2 through 8 years who have no contraindications or precautions,” which was approved in June 2014. This was based on evidence during several influenza seasons that LAIV provided better protection than the IIV for children in this age group. The decision to make the change regarding preference “was made based on new data from more recent seasons, which have not confirmed superior effectiveness of LAIV observed in earlier studies,” according to a statement issued by the CDC following the meeting.
For the next influenza season, there will no longer be a preference for using the nasal spray influenza vaccine over the flu shot for healthy children aged 2-8 years, based on data from recent influenza seasons.
In a 14-0 vote, with one abstention, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) supported the following revision to the influenza vaccine recommendations, for the 2015-2016 influenza season: “For healthy children aged 2 through 8 years who have no contraindications or precautions, either LAIV [live attenuated influenza vaccine] or IIV [inactivated influenza vaccine] is an appropriate option. No preference is expressed for LAIV or IIV or any person aged 2 though 49 years for whom either vaccine is appropriate.”
The recommendation that all people aged 6 months and older should receive the influenza vaccine every year remains unchanged.
Currently, the recommendation is that when “immediately available, LAIV should be used for healthy children aged 2 through 8 years who have no contraindications or precautions,” which was approved in June 2014. This was based on evidence during several influenza seasons that LAIV provided better protection than the IIV for children in this age group. The decision to make the change regarding preference “was made based on new data from more recent seasons, which have not confirmed superior effectiveness of LAIV observed in earlier studies,” according to a statement issued by the CDC following the meeting.
FROM AN ACIP MEETING