User login
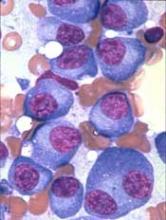
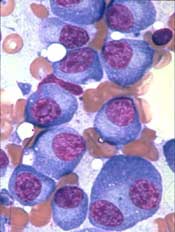
The US Food and Drug Administration (FDA) has granted orphan designation to PT-112 as a treatment for multiple myeloma (MM).
PT-112 is a small-molecule conjugate of pyrophosphate and platinum that promotes apoptosis with damage-associated molecular patterns, leading to downstream T-cell recruitment in the tumor microenvironment.
PT-112 is currently under investigation in a phase 1/2 study of patients with relapsed or refractory MM (NCT03288480).
Phosplatin Therapeutics LLC, the company developing PT-112, has enrolled the first cohort of patients in this trial.
In preclinical experiments, PT-112 demonstrated synergy with lenalidomide and bortezomib in RPMI-8226 cells and dexamethasone-resistant MM1R cells.
Single-agent PT-112 produced responses in mice with established MM. Researchers said PT-112 had “pronounced” activity against bortezomib-refractory Vk12598 tumors, which significantly improved overall survival in the mice.
This research was presented at the 2017 ASH Annual Meeting (abstract 1797).
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted orphan designation to PT-112 as a treatment for multiple myeloma (MM).
PT-112 is a small-molecule conjugate of pyrophosphate and platinum that promotes apoptosis with damage-associated molecular patterns, leading to downstream T-cell recruitment in the tumor microenvironment.
PT-112 is currently under investigation in a phase 1/2 study of patients with relapsed or refractory MM (NCT03288480).
Phosplatin Therapeutics LLC, the company developing PT-112, has enrolled the first cohort of patients in this trial.
In preclinical experiments, PT-112 demonstrated synergy with lenalidomide and bortezomib in RPMI-8226 cells and dexamethasone-resistant MM1R cells.
Single-agent PT-112 produced responses in mice with established MM. Researchers said PT-112 had “pronounced” activity against bortezomib-refractory Vk12598 tumors, which significantly improved overall survival in the mice.
This research was presented at the 2017 ASH Annual Meeting (abstract 1797).
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The US Food and Drug Administration (FDA) has granted orphan designation to PT-112 as a treatment for multiple myeloma (MM).
PT-112 is a small-molecule conjugate of pyrophosphate and platinum that promotes apoptosis with damage-associated molecular patterns, leading to downstream T-cell recruitment in the tumor microenvironment.
PT-112 is currently under investigation in a phase 1/2 study of patients with relapsed or refractory MM (NCT03288480).
Phosplatin Therapeutics LLC, the company developing PT-112, has enrolled the first cohort of patients in this trial.
In preclinical experiments, PT-112 demonstrated synergy with lenalidomide and bortezomib in RPMI-8226 cells and dexamethasone-resistant MM1R cells.
Single-agent PT-112 produced responses in mice with established MM. Researchers said PT-112 had “pronounced” activity against bortezomib-refractory Vk12598 tumors, which significantly improved overall survival in the mice.
This research was presented at the 2017 ASH Annual Meeting (abstract 1797).
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.