User login
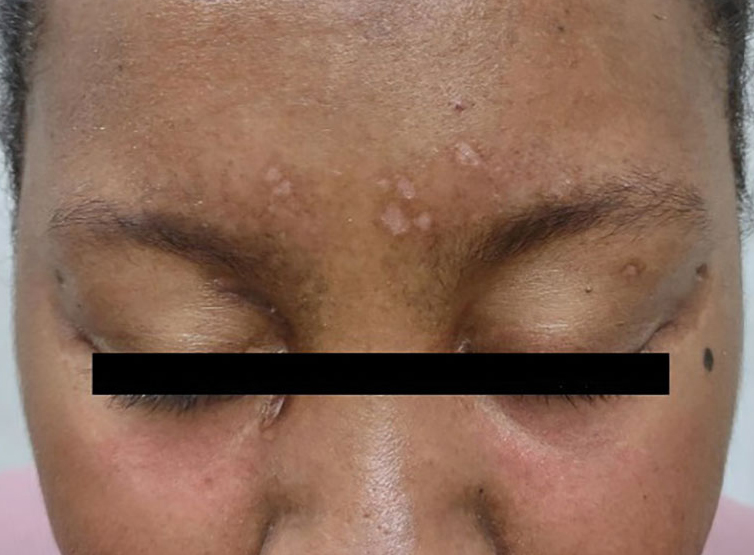
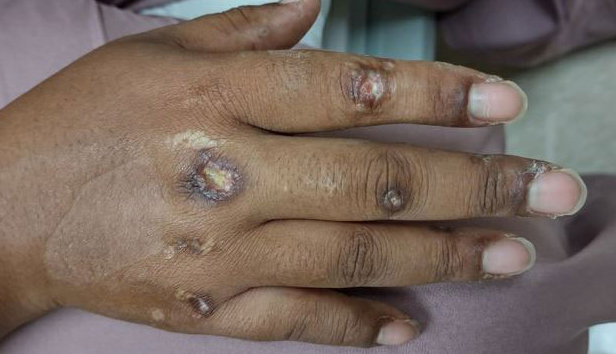
A previously healthy 31-year-old female active-duty Navy sailor working as a calibration technician developed a painful, erythematous, pruritic, indurated plaque on her left breast. The sailor was not lactating and had no known family history of malignancy. Initially, she was treated by her primary care practitioner for presumed mastitis with oral cephalexin and then with oral clindamycin with no symptom improvement. About 2 weeks after the completion of both antibiotic courses, she developed angioedema and periorbital edema (Figure 1), requiring highdose corticosteroids and antihistamines with a corticosteroid course of prednisone 40 mg daily tapered to 10 mg daily over 12 days and diphenhydramine 25 mg to use up to 4 times daily. Workup for both was acquired and hereditary angioedema was unremarkable. Two months later, the patient developed patches of alopecia, oral ulcerations, and hypopigmented plaques with a peripheral hyperpigmented rim on the central face and bilateral conchal bowls (Figure 2). She also developed hypopigmented papules with peripheral hyperpigmentation on the bilateral dorsal hands overlying the metacarpal and proximal interphalangeal joints, which eventually ulcerated (Figure 3). Laboratory evaluation, including tests for creatine kinase, aldolase, transaminases, lactate dehydrogenase, and autoantibodies (antiJo-1, anti-Mi-2, anti-MDA-5, anti-TIF-1, anti-NXP-2, and anti-SAEP), were unremarkable. A punch biopsy from a papule on the right dorsal hand showed superficial perivascular lymphohistiocytic inflammation with a subtle focal increase in dermal mucin, highlighted by the colloidal iron stain. Further evaluation of the left breast plaque revealed ER/PR+ HER2- stage IIIB inflammatory breast cancer.

DISCUSSION
Based on the clinical presentation and diagnosis of inflammatory breast cancer, the patient was diagnosed with paraneoplastic clinically amyopathic dermatomyositis (CADM). She was treated for her breast cancer with an initial chemotherapy regimen consisting of dose-dense cyclophosphamide and doxorubicin followed by paclitaxel. The patient underwent a mastectomy, axillary lymph node dissection, and 25 sessions of radiation therapy, and is currently continuing therapy with anastrozole 1 mg daily and ovarian suppression with leuprorelin 11.25 mg every 3 months. For the severe angioedema and dermatomyositis-like cutaneous findings, the patient was continued on high-dose corticosteroids at prednisone 60 mg daily with a prolonged taper to prednisone 10 mg daily. After about 10 months, she transitioned from prednisone 10 mg daily to hydrocortisone 30 mg daily and is currently tapering her hydrocortisone dosing. She was additionally started on monthly intravenous immunoglobulin, hydroxychloroquine 300 mg daily, and amlodipine 5 mg daily. The ulcerated papules on her hands were treated with topical clobetasol 0.05% ointment applied daily, topical tacrolimus 0.1% ointment applied daily, and multiple intralesional triamcinolone 5 mg/mL injections. With this regimen, the patient experienced significant improvement in her cutaneous symptoms.
CADM is a rare autoimmune inflammatory disease featuring classic dermatomyositis-like cutaneous findings such as a heliotrope rash and Gottron papules. Ulcerative Gottron papules are less common than the typical erythematous papules and are associated more strongly with amyopathic disease.1 Paraneoplastic myositis poses a diagnostic challenge because it presents like an idiopathic dermatomyositis and often has a heterogeneous clinical presentation with additional manifestations, including periorbital edema, myalgias, dysphagia, and shortness of breath. If clinically suspected, laboratory tests (eg, creatine kinase, aldolase, transaminases, and lactate dehydrogenase) can assist in diagnosing paraneoplastic myositis. Additionally, serologic testing for autoantibodies such as anti-CADM-140, anti-Jo-1, anti-Mi-2, antiMDA-5, anti-TIF-1, anti-NXP-2, and antiSAE can assist the diagnosis and predict disease phenotype.1,2
Malignancy can precede, occur during, or develop after the diagnosis of CADM.3 Malignancies most often associated with CADM include ovarian, breast, and lung cancers.4 Despite the strong correlation with malignancy, there are currently no screening guidelines for malignancy upon inflammatory myositis diagnosis. Therefore, it is important to consider the entirety of a patient’s clinical presentation in establishing further evaluation in the initial diagnostic workup.
There are numerous systemic complications associated with inflammatory myositis and imaging modalities can help to rule out some of these conditions. CADM is strongly associated with the development of interstitial lung disease, so chest radiography and pulmonary function testing are often checked.1 Though cardiac and esophageal involvement are more commonly associated with classic dermatomyositis, it may be useful to obtain an electrocardiogram to rule out conduction abnormalities from myocardial involvement, along with esophageal manometry to evaluate for esophageal dysmotility.1,5
In the management of paraneoplastic CADM, the underlying malignancy should be treated first.6 If symptoms persist after the cancer is in remission, then CADM is treated with immunosuppressive medications such as methotrexate, mycophenolate mofetil, or azathioprine. Physical therapy can also provide further symptom relief for those suffering from proximal weakness.
CONCLUSIONS
Presumed mastitis, angioedema, and eczematous lesions for this patient were dermatologic manifestations of an underlying inflammatory breast cancer. This case highlights the importance of early recognition, the diagnosis of CADM and awareness of its association with underlying malignancy, especially within the primary care setting where most skin concerns are addressed. Early clinical suspicion and a swift diagnostic workup can further optimize multidisciplinary management, which is often required to treat malignancies.
- Cao H, Xia Q, Pan M, et al. Gottron papules and gottron sign with ulceration: a distinctive cutaneous feature in a subset of patients with classic dermatomyositis and clinically amyopathic dermatomyositis. J Rheumatol. 2016;43(9):1735-1742. doi:10.3899/jrheum.160024
- Satoh M, Tanaka S, Ceribelli A, Calise SJ, Chan EK. A comprehensive overview on myositis-specific antibodies: new and old biomarkers in idiopathic inflammatory myopathy. Clin Rev Allergy Immunol. 2017;52(1):1-19. doi:10.1007/s12016-015-8510-y
- Zahr ZA, Baer AN. Malignancy in myositis. Curr Rheumatol Rep. 2011;13(3):208-215. doi:10.1007/s11926-011-0169-7
- Udkoff J, Cohen PR. Amyopathic dermatomyositis: a concise review of clinical manifestations and associated malignancies. Am J Clin Dermatol. 2016;17(5): 509-518. doi:10.1007/s40257-016-0199-z
- Fathi M, Lundberg IE, Tornling G. Pulmonary complications of polymyositis and dermatomyositis. Semin Respir Crit Care Med. 2007;28(4):451-458. doi:10.1055/s-2007-985666
- Hendren E, Vinik O, Faragalla H, Haq R. Breast cancer and dermatomyositis: a case study and literature review. Curr Oncol. 2017;24(5):e429-e433. doi:10.3747/co.24.3696
A previously healthy 31-year-old female active-duty Navy sailor working as a calibration technician developed a painful, erythematous, pruritic, indurated plaque on her left breast. The sailor was not lactating and had no known family history of malignancy. Initially, she was treated by her primary care practitioner for presumed mastitis with oral cephalexin and then with oral clindamycin with no symptom improvement. About 2 weeks after the completion of both antibiotic courses, she developed angioedema and periorbital edema (Figure 1), requiring highdose corticosteroids and antihistamines with a corticosteroid course of prednisone 40 mg daily tapered to 10 mg daily over 12 days and diphenhydramine 25 mg to use up to 4 times daily. Workup for both was acquired and hereditary angioedema was unremarkable. Two months later, the patient developed patches of alopecia, oral ulcerations, and hypopigmented plaques with a peripheral hyperpigmented rim on the central face and bilateral conchal bowls (Figure 2). She also developed hypopigmented papules with peripheral hyperpigmentation on the bilateral dorsal hands overlying the metacarpal and proximal interphalangeal joints, which eventually ulcerated (Figure 3). Laboratory evaluation, including tests for creatine kinase, aldolase, transaminases, lactate dehydrogenase, and autoantibodies (antiJo-1, anti-Mi-2, anti-MDA-5, anti-TIF-1, anti-NXP-2, and anti-SAEP), were unremarkable. A punch biopsy from a papule on the right dorsal hand showed superficial perivascular lymphohistiocytic inflammation with a subtle focal increase in dermal mucin, highlighted by the colloidal iron stain. Further evaluation of the left breast plaque revealed ER/PR+ HER2- stage IIIB inflammatory breast cancer.



DISCUSSION
Based on the clinical presentation and diagnosis of inflammatory breast cancer, the patient was diagnosed with paraneoplastic clinically amyopathic dermatomyositis (CADM). She was treated for her breast cancer with an initial chemotherapy regimen consisting of dose-dense cyclophosphamide and doxorubicin followed by paclitaxel. The patient underwent a mastectomy, axillary lymph node dissection, and 25 sessions of radiation therapy, and is currently continuing therapy with anastrozole 1 mg daily and ovarian suppression with leuprorelin 11.25 mg every 3 months. For the severe angioedema and dermatomyositis-like cutaneous findings, the patient was continued on high-dose corticosteroids at prednisone 60 mg daily with a prolonged taper to prednisone 10 mg daily. After about 10 months, she transitioned from prednisone 10 mg daily to hydrocortisone 30 mg daily and is currently tapering her hydrocortisone dosing. She was additionally started on monthly intravenous immunoglobulin, hydroxychloroquine 300 mg daily, and amlodipine 5 mg daily. The ulcerated papules on her hands were treated with topical clobetasol 0.05% ointment applied daily, topical tacrolimus 0.1% ointment applied daily, and multiple intralesional triamcinolone 5 mg/mL injections. With this regimen, the patient experienced significant improvement in her cutaneous symptoms.
CADM is a rare autoimmune inflammatory disease featuring classic dermatomyositis-like cutaneous findings such as a heliotrope rash and Gottron papules. Ulcerative Gottron papules are less common than the typical erythematous papules and are associated more strongly with amyopathic disease.1 Paraneoplastic myositis poses a diagnostic challenge because it presents like an idiopathic dermatomyositis and often has a heterogeneous clinical presentation with additional manifestations, including periorbital edema, myalgias, dysphagia, and shortness of breath. If clinically suspected, laboratory tests (eg, creatine kinase, aldolase, transaminases, and lactate dehydrogenase) can assist in diagnosing paraneoplastic myositis. Additionally, serologic testing for autoantibodies such as anti-CADM-140, anti-Jo-1, anti-Mi-2, antiMDA-5, anti-TIF-1, anti-NXP-2, and antiSAE can assist the diagnosis and predict disease phenotype.1,2
Malignancy can precede, occur during, or develop after the diagnosis of CADM.3 Malignancies most often associated with CADM include ovarian, breast, and lung cancers.4 Despite the strong correlation with malignancy, there are currently no screening guidelines for malignancy upon inflammatory myositis diagnosis. Therefore, it is important to consider the entirety of a patient’s clinical presentation in establishing further evaluation in the initial diagnostic workup.
There are numerous systemic complications associated with inflammatory myositis and imaging modalities can help to rule out some of these conditions. CADM is strongly associated with the development of interstitial lung disease, so chest radiography and pulmonary function testing are often checked.1 Though cardiac and esophageal involvement are more commonly associated with classic dermatomyositis, it may be useful to obtain an electrocardiogram to rule out conduction abnormalities from myocardial involvement, along with esophageal manometry to evaluate for esophageal dysmotility.1,5
In the management of paraneoplastic CADM, the underlying malignancy should be treated first.6 If symptoms persist after the cancer is in remission, then CADM is treated with immunosuppressive medications such as methotrexate, mycophenolate mofetil, or azathioprine. Physical therapy can also provide further symptom relief for those suffering from proximal weakness.
CONCLUSIONS
Presumed mastitis, angioedema, and eczematous lesions for this patient were dermatologic manifestations of an underlying inflammatory breast cancer. This case highlights the importance of early recognition, the diagnosis of CADM and awareness of its association with underlying malignancy, especially within the primary care setting where most skin concerns are addressed. Early clinical suspicion and a swift diagnostic workup can further optimize multidisciplinary management, which is often required to treat malignancies.
A previously healthy 31-year-old female active-duty Navy sailor working as a calibration technician developed a painful, erythematous, pruritic, indurated plaque on her left breast. The sailor was not lactating and had no known family history of malignancy. Initially, she was treated by her primary care practitioner for presumed mastitis with oral cephalexin and then with oral clindamycin with no symptom improvement. About 2 weeks after the completion of both antibiotic courses, she developed angioedema and periorbital edema (Figure 1), requiring highdose corticosteroids and antihistamines with a corticosteroid course of prednisone 40 mg daily tapered to 10 mg daily over 12 days and diphenhydramine 25 mg to use up to 4 times daily. Workup for both was acquired and hereditary angioedema was unremarkable. Two months later, the patient developed patches of alopecia, oral ulcerations, and hypopigmented plaques with a peripheral hyperpigmented rim on the central face and bilateral conchal bowls (Figure 2). She also developed hypopigmented papules with peripheral hyperpigmentation on the bilateral dorsal hands overlying the metacarpal and proximal interphalangeal joints, which eventually ulcerated (Figure 3). Laboratory evaluation, including tests for creatine kinase, aldolase, transaminases, lactate dehydrogenase, and autoantibodies (antiJo-1, anti-Mi-2, anti-MDA-5, anti-TIF-1, anti-NXP-2, and anti-SAEP), were unremarkable. A punch biopsy from a papule on the right dorsal hand showed superficial perivascular lymphohistiocytic inflammation with a subtle focal increase in dermal mucin, highlighted by the colloidal iron stain. Further evaluation of the left breast plaque revealed ER/PR+ HER2- stage IIIB inflammatory breast cancer.



DISCUSSION
Based on the clinical presentation and diagnosis of inflammatory breast cancer, the patient was diagnosed with paraneoplastic clinically amyopathic dermatomyositis (CADM). She was treated for her breast cancer with an initial chemotherapy regimen consisting of dose-dense cyclophosphamide and doxorubicin followed by paclitaxel. The patient underwent a mastectomy, axillary lymph node dissection, and 25 sessions of radiation therapy, and is currently continuing therapy with anastrozole 1 mg daily and ovarian suppression with leuprorelin 11.25 mg every 3 months. For the severe angioedema and dermatomyositis-like cutaneous findings, the patient was continued on high-dose corticosteroids at prednisone 60 mg daily with a prolonged taper to prednisone 10 mg daily. After about 10 months, she transitioned from prednisone 10 mg daily to hydrocortisone 30 mg daily and is currently tapering her hydrocortisone dosing. She was additionally started on monthly intravenous immunoglobulin, hydroxychloroquine 300 mg daily, and amlodipine 5 mg daily. The ulcerated papules on her hands were treated with topical clobetasol 0.05% ointment applied daily, topical tacrolimus 0.1% ointment applied daily, and multiple intralesional triamcinolone 5 mg/mL injections. With this regimen, the patient experienced significant improvement in her cutaneous symptoms.
CADM is a rare autoimmune inflammatory disease featuring classic dermatomyositis-like cutaneous findings such as a heliotrope rash and Gottron papules. Ulcerative Gottron papules are less common than the typical erythematous papules and are associated more strongly with amyopathic disease.1 Paraneoplastic myositis poses a diagnostic challenge because it presents like an idiopathic dermatomyositis and often has a heterogeneous clinical presentation with additional manifestations, including periorbital edema, myalgias, dysphagia, and shortness of breath. If clinically suspected, laboratory tests (eg, creatine kinase, aldolase, transaminases, and lactate dehydrogenase) can assist in diagnosing paraneoplastic myositis. Additionally, serologic testing for autoantibodies such as anti-CADM-140, anti-Jo-1, anti-Mi-2, antiMDA-5, anti-TIF-1, anti-NXP-2, and antiSAE can assist the diagnosis and predict disease phenotype.1,2
Malignancy can precede, occur during, or develop after the diagnosis of CADM.3 Malignancies most often associated with CADM include ovarian, breast, and lung cancers.4 Despite the strong correlation with malignancy, there are currently no screening guidelines for malignancy upon inflammatory myositis diagnosis. Therefore, it is important to consider the entirety of a patient’s clinical presentation in establishing further evaluation in the initial diagnostic workup.
There are numerous systemic complications associated with inflammatory myositis and imaging modalities can help to rule out some of these conditions. CADM is strongly associated with the development of interstitial lung disease, so chest radiography and pulmonary function testing are often checked.1 Though cardiac and esophageal involvement are more commonly associated with classic dermatomyositis, it may be useful to obtain an electrocardiogram to rule out conduction abnormalities from myocardial involvement, along with esophageal manometry to evaluate for esophageal dysmotility.1,5
In the management of paraneoplastic CADM, the underlying malignancy should be treated first.6 If symptoms persist after the cancer is in remission, then CADM is treated with immunosuppressive medications such as methotrexate, mycophenolate mofetil, or azathioprine. Physical therapy can also provide further symptom relief for those suffering from proximal weakness.
CONCLUSIONS
Presumed mastitis, angioedema, and eczematous lesions for this patient were dermatologic manifestations of an underlying inflammatory breast cancer. This case highlights the importance of early recognition, the diagnosis of CADM and awareness of its association with underlying malignancy, especially within the primary care setting where most skin concerns are addressed. Early clinical suspicion and a swift diagnostic workup can further optimize multidisciplinary management, which is often required to treat malignancies.
- Cao H, Xia Q, Pan M, et al. Gottron papules and gottron sign with ulceration: a distinctive cutaneous feature in a subset of patients with classic dermatomyositis and clinically amyopathic dermatomyositis. J Rheumatol. 2016;43(9):1735-1742. doi:10.3899/jrheum.160024
- Satoh M, Tanaka S, Ceribelli A, Calise SJ, Chan EK. A comprehensive overview on myositis-specific antibodies: new and old biomarkers in idiopathic inflammatory myopathy. Clin Rev Allergy Immunol. 2017;52(1):1-19. doi:10.1007/s12016-015-8510-y
- Zahr ZA, Baer AN. Malignancy in myositis. Curr Rheumatol Rep. 2011;13(3):208-215. doi:10.1007/s11926-011-0169-7
- Udkoff J, Cohen PR. Amyopathic dermatomyositis: a concise review of clinical manifestations and associated malignancies. Am J Clin Dermatol. 2016;17(5): 509-518. doi:10.1007/s40257-016-0199-z
- Fathi M, Lundberg IE, Tornling G. Pulmonary complications of polymyositis and dermatomyositis. Semin Respir Crit Care Med. 2007;28(4):451-458. doi:10.1055/s-2007-985666
- Hendren E, Vinik O, Faragalla H, Haq R. Breast cancer and dermatomyositis: a case study and literature review. Curr Oncol. 2017;24(5):e429-e433. doi:10.3747/co.24.3696
- Cao H, Xia Q, Pan M, et al. Gottron papules and gottron sign with ulceration: a distinctive cutaneous feature in a subset of patients with classic dermatomyositis and clinically amyopathic dermatomyositis. J Rheumatol. 2016;43(9):1735-1742. doi:10.3899/jrheum.160024
- Satoh M, Tanaka S, Ceribelli A, Calise SJ, Chan EK. A comprehensive overview on myositis-specific antibodies: new and old biomarkers in idiopathic inflammatory myopathy. Clin Rev Allergy Immunol. 2017;52(1):1-19. doi:10.1007/s12016-015-8510-y
- Zahr ZA, Baer AN. Malignancy in myositis. Curr Rheumatol Rep. 2011;13(3):208-215. doi:10.1007/s11926-011-0169-7
- Udkoff J, Cohen PR. Amyopathic dermatomyositis: a concise review of clinical manifestations and associated malignancies. Am J Clin Dermatol. 2016;17(5): 509-518. doi:10.1007/s40257-016-0199-z
- Fathi M, Lundberg IE, Tornling G. Pulmonary complications of polymyositis and dermatomyositis. Semin Respir Crit Care Med. 2007;28(4):451-458. doi:10.1055/s-2007-985666
- Hendren E, Vinik O, Faragalla H, Haq R. Breast cancer and dermatomyositis: a case study and literature review. Curr Oncol. 2017;24(5):e429-e433. doi:10.3747/co.24.3696